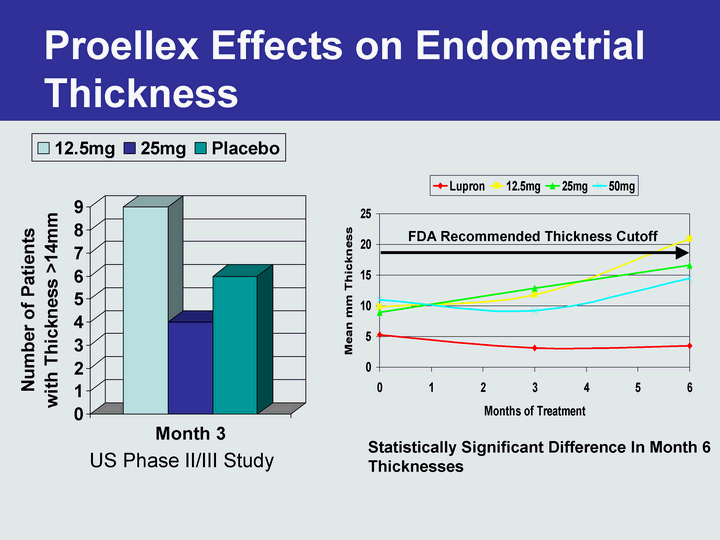
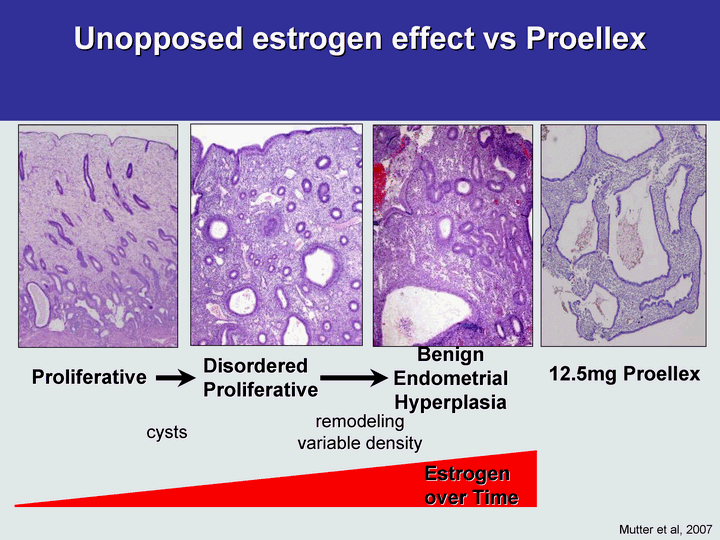
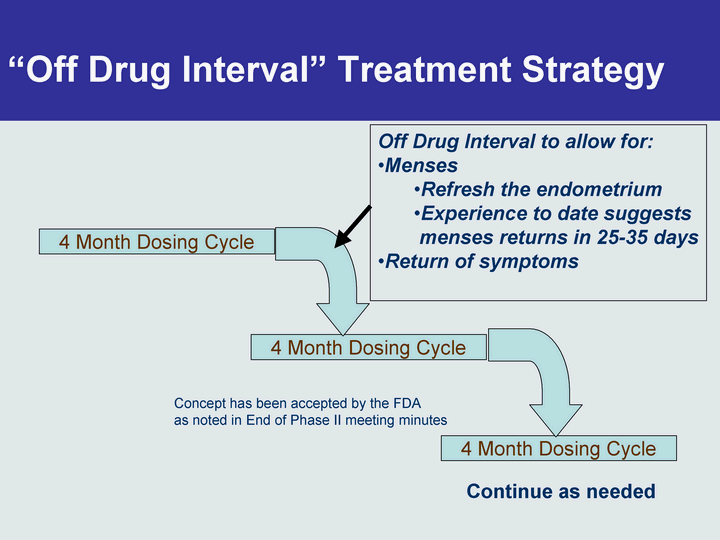
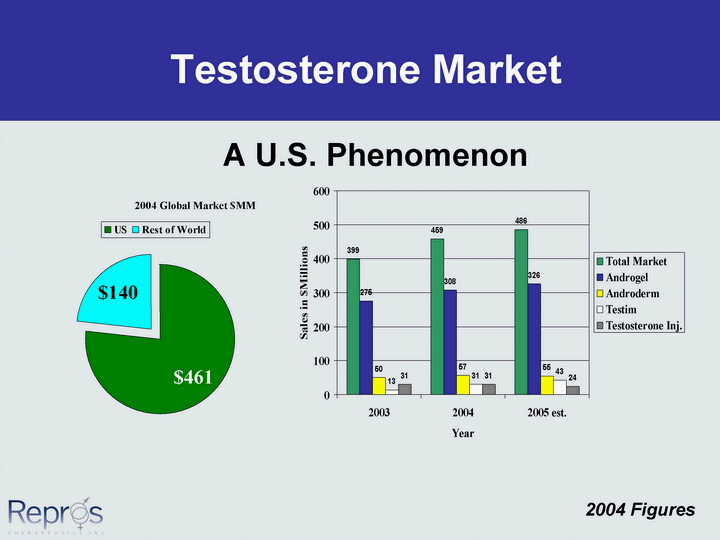
Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Royalty Pharma similar filings
- 12 Feb 08 Regulation FD Disclosure
- 11 Feb 08 Repros Presentation to Be Webcast from The 10th Annual Bio/CEO and Investor Conference 2008
- 1 Feb 08 Repros Announces 2008 Annual Shareholders’ Meeting
- 10 Jan 08 Regulation FD Disclosure
- 8 Jan 08 Regulation FD Disclosure
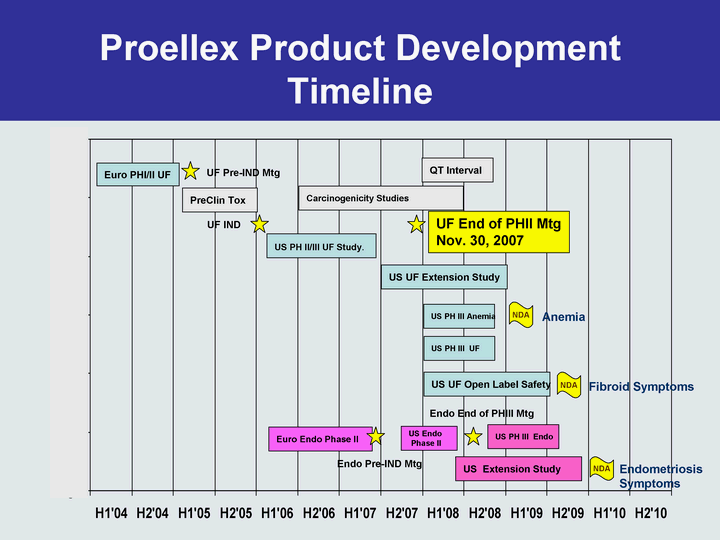
- 4 Dec 07 Repros’ Lead Product Proellex to Enter Phase 3 for the Treatment of Uterine Fibroid Indications
- 9 Nov 07 Repros Therapeutics Inc. Reports Third Quarter 2007 Financial Results and Provides Clinical Development Update
Filing view
External links