
Developing clinical stage small molecule therapeutics to treat hormonal and reproductive system disorders

Repros Disclaimer Any statements made by the Company that are not historical facts contained in these slides (or in any oral accompanying discussion) are forward - looking statements that involve risks and uncertainties that could cause actual results to differ materially from the results expressed or implied by such statements, including the ability to raise additional needed capital on a timely basis in order for it to continue to fund development of its Androxal ® and Proellex ® programs, the ability to have success in the clinical development of its technologies, the reliability of interim results to predict final study outcomes, and such other risks which are identified in the Company's most recent Annual Report on Form 10 - K and the subsequent quarterly report on Form 10 - Q and in the prospectus supplement and the accompanying prospectus included in the registration statement mentioned below. These documents are available on request from Repros Therapeutics or at www.sec.gov . Repros disclaims any intention or obligation to update or revise any forward - looking statements, whether as a result of new information, future events or otherwise. In this presentation, we rely on and refer to information and statistics regarding the pharmaceutical industry. We obtained this information and these statistics from third - party sources, which we have supplemented where necessary with information from publicly available sources and our own internal estimates. Industry publications and surveys generally state that they have obtained information from sources believed to be reliable, but do not guarantee the accuracy and completeness of such information. While we believe that each of these studies and publications is reliable, we have not independently verified such data, and we make no any representation as to the accuracy of such information. Similarly, we believe our internal research is reliable, but it has not been verified by any independent sources. 1

Investment Highlights • Focused strategy: small molecule therapeutics for reproductive disorders • Two late stage clinical programs each with +$1B sales potential • Androxal ® : PHASE 3 (SPA) oral treatment for Low Testosterone – Patented and pending patent’s life to the mid 2020’s – Growing +$2B market – Restoration of testicular function and testosterone levels in treatment of 2º hypogonadism (most common cause of low T) • Proellex: PHASE 2 treatment for uterine fibroids and endometriosis – Pending patent/patent life to the mid 2020’s – +$5B market – Fibroid de - bulking and chronic relief of symptoms associated with uterine fibroids, endometriosis – Potential breast cancer intervention • Key late stage clinical & regulatory events driven news flow in 2013 2

Testosterone Market and Androxal Overview • US market for low testosterone exceeds $1.5 billion • Only approved non invasive therapies are hormone replacements • Repros believes 85% of hypogonadal men experience low T due to an endocrine disorder – These men have functional but un - stimulated testes – Hypothalamic - pituitary suppression due to estrogen • Androxal is the only oral medication in development that treats the underlying disorder for the majority of hypogonadal men 3
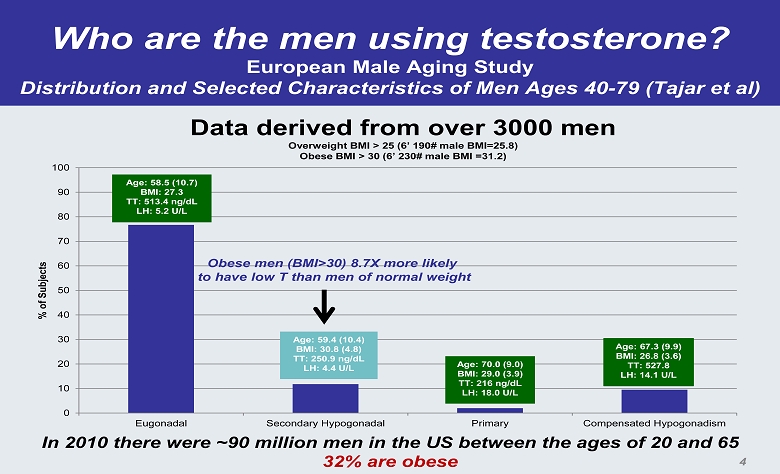
Who are the men using testosterone? European Male Aging Study Distribution and Selected Characteristics of Men Ages 40 - 79 (Tajar et al) 0 10 20 30 40 50 60 70 80 90 100 Eugonadal Secondary Hypogonadal Primary Compensated Hypogonadism % of Subjects Age: 58.5 (10.7) BMI: 27.3 TT: 513.4 ng/dL LH: 5.2 U/L Age: 59.4 (10.4) BMI: 30.8 (4.8) TT: 250.9 ng/dL LH: 4.4 U/L Age: 70.0 (9.0) BMI: 29.0 (3.9) TT: 216 ng/dL LH: 18.0 U/L Age: 67.3 (9.9) BMI: 26.8 (3.6) TT: 527.8 LH: 14.1 U/L Data derived from over 3000 men Overweight BMI > 25 (6’ 190# male BMI=25.8) Obese BMI > 30 (6’ 230# male BMI =31.2) Obese men (BMI>30) 8.7X more likely to have low T than men of normal weight In 2010 there were ~90 million men in the US between the ages of 20 and 65 32% are obese 4
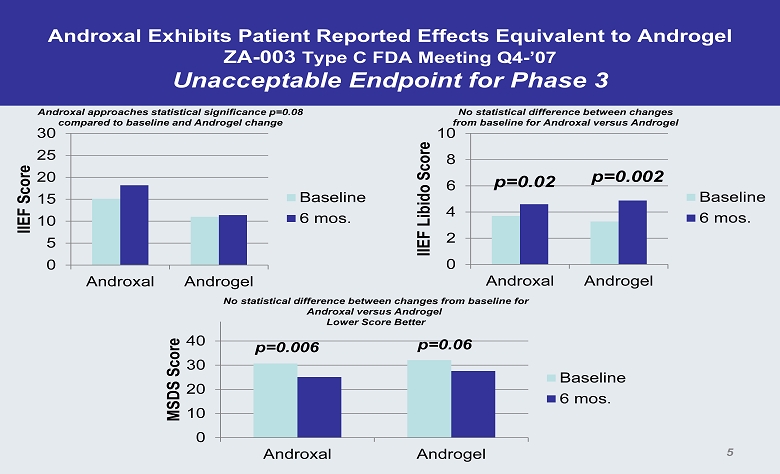
Androxal Exhibits Patient Reported Effects Equivalent to Androgel ZA - 003 Type C FDA Meeting Q4 - ’07 Unacceptable Endpoint for Phase 3 0 5 10 15 20 25 30 Androxal Androgel IIEF Score Baseline 6 mos. Androxal approaches statistical significance p=0.08 compared to baseline and Androgel change 0 2 4 6 8 10 Androxal Androgel IIEF Libido Score Baseline 6 mos. No statistical difference between changes from baseline for Androxal versus Androgel p=0.02 p=0.002 0 10 20 30 40 Androxal Androgel MSDS Score Baseline 6 mos. p=0.006 p=0.06 No statistical difference between changes from baseline for Androxal versus Androgel Lower Score Better 5
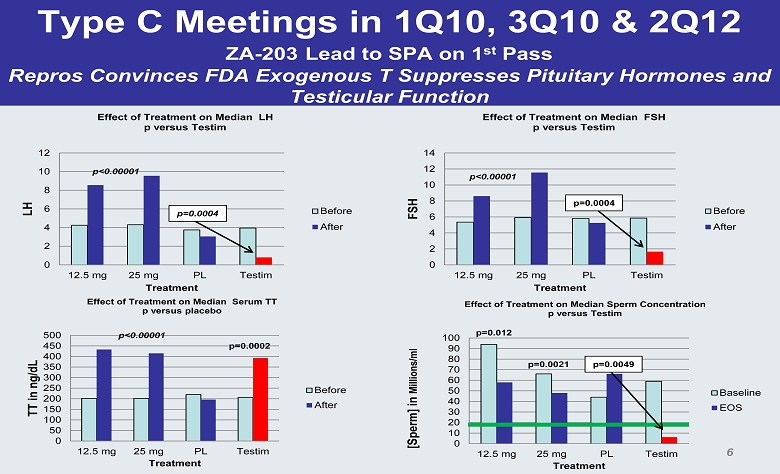
Type C Meetings in 1Q10, 3Q10 & 2Q12 ZA - 203 Lead to SPA on 1 st Pass Repros Convinces FDA Exogenous T Suppresses Pituitary Hormones and Testicular Function 0 2 4 6 8 10 12 14 12.5 mg 25 mg PL Testim FSH Treatment Effect of Treatment on Median FSH p versus Testim Before After p<0.00001 p=0.0004 0 10 20 30 40 50 60 70 80 90 100 12.5 mg 25 mg PL Testim [Sperm] in Millions/ml Treatment Effect of Treatment on Median Sperm Concentration p versus Testim Baseline EOS p=0.012 p=0.0021 p=0.0049 0 2 4 6 8 10 12 12.5 mg 25 mg PL Testim LH Treatment Effect of Treatment on Median LH p versus Testim Before After p=0.0004 0 50 100 150 200 250 300 350 400 450 500 12.5 mg 25 mg PL Testim TT in ng/dL Treatment Effect of Treatment on Median Serum TT p versus placebo Before After p<0.00001 p=0.0002 p<0.00001 6

FDA Accepted Phase 3 Protocols Under an SPA Goal to Submit NDA Mid 2014 • Phase 3 pivotal studies conducted under SPA – 2 identical trials (BMI > 25, Age ≤ 60) • ZA - 301 152 subjects (114 on Androxal, 38 on placebo) • ZA - 301 181 subjects ( 134 on Androxal, 47 on placebo) – Men with morning T<300 ng/dL assessed twice on two separate days – Men with sperm concentration > 15 x10 6 per milliliter assessed on 2 separate days separated by at least 2 days • Up - titration from 12.5 to 25 • 3 month duration (men up - titrated on study for additional 6 weeks) – Both Co - primary Endpoints Met in Both Studies • 75% of men achieved T in normal range (300 - 1040 ng/dL) – 24 hr average at week 12 • Non inferior to placebo regarding change in sperm counts (average of week 12 & 13) – All protocol changes have been reviewed with FDA before incorporating into study • Safety Requirements of Phase 3 program – >100 for one year (~100 completed, ~200 at approval) – >800 for 6 months (~800 completed, ~850 at approval) – 100 subject one year DEXA study (bone marker data suggests Androxal builds bone) 7
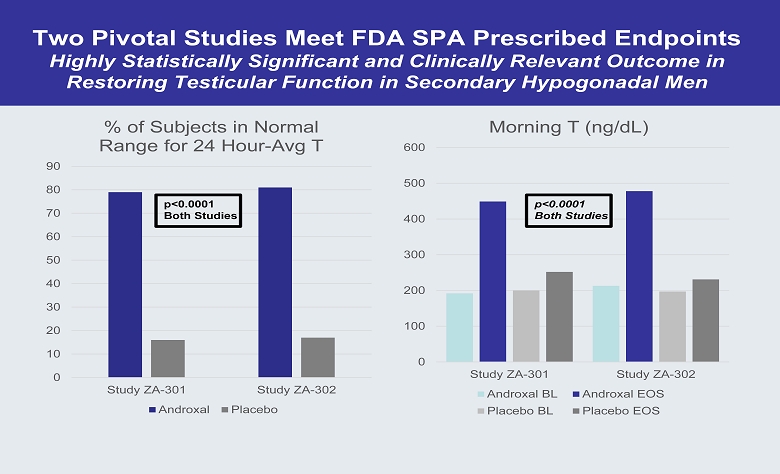
Two Pivotal Studies Meet FDA SPA Prescribed Endpoints Highly Statistically Significant and Clinically Relevant Outcome in Restoring Testicular Function in Secondary Hypogonadal Men 0 10 20 30 40 50 60 70 80 90 Study ZA-301 Study ZA-302 % of Subjects in Normal Range for 24 Hour - Avg T Androxal Placebo p<0.0001 Both Studies 0 100 200 300 400 500 600 Study ZA-301 Study ZA-302 Morning T (ng/dL) Androxal BL Androxal EOS Placebo BL Placebo EOS p<0.0001 Both Studies
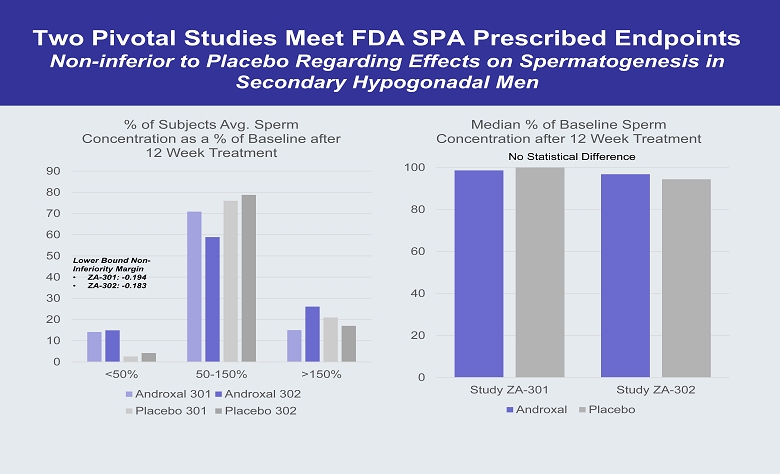
Two Pivotal Studies Meet FDA SPA Prescribed Endpoints Non - inferior to Placebo Regarding Effects on Spermatogenesis in Secondary Hypogonadal Men 0 10 20 30 40 50 60 70 80 90 <50% 50-150% >150% % of Subjects Avg. Sperm Concentration as a % of Baseline after 12 Week Treatment Androxal 301 Androxal 302 Placebo 301 Placebo 302 0 20 40 60 80 100 Study ZA-301 Study ZA-302 Median % of Baseline Sperm Concentration after 12 Week Treatment Androxal Placebo No Statistical Difference Lower Bound Non - Inferiority Margin • ZA - 301: - 0.194 • ZA - 302: - 0.183
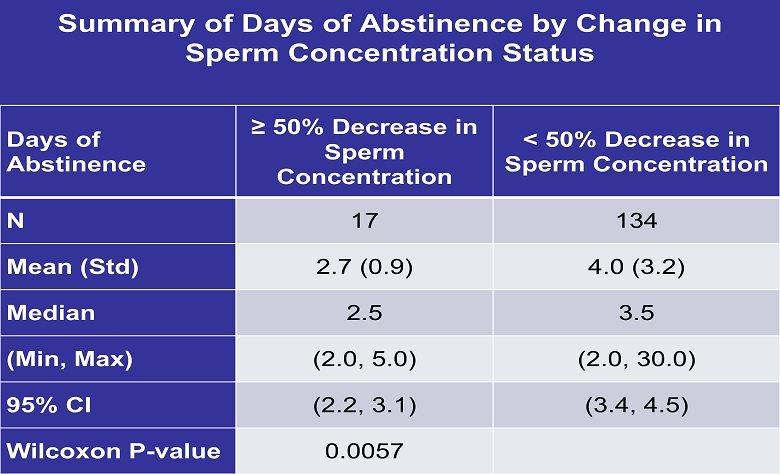
Summary of Days of Abstinence by Change in Sperm Concentration Status Days of Abstinence ≥ 50% Decrease in Sperm Concentration < 50% Decrease in Sperm Concentration N 17 134 Mean (Std) 2.7 (0.9) 4.0 (3.2) Median 2.5 3.5 (Min, Max) (2.0, 5.0) (2.0, 30.0) 95 % CI (2.2, 3.1) (3.4, 4.5) Wilcoxon P - value 0.0057
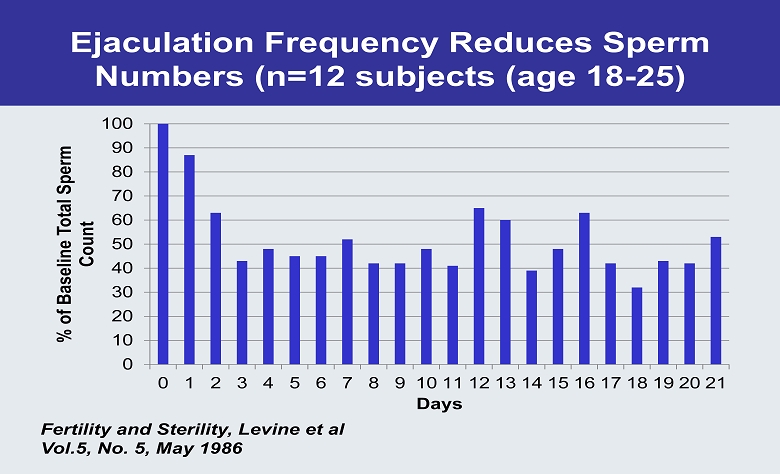
Ejaculation Frequency Reduces Sperm Numbers (n=12 subjects (age 18 - 25) 0 10 20 30 40 50 60 70 80 90 100 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 % of Baseline Total Sperm Count Days Fertility and Sterility, Levine et al Vol.5, No. 5, May 1986
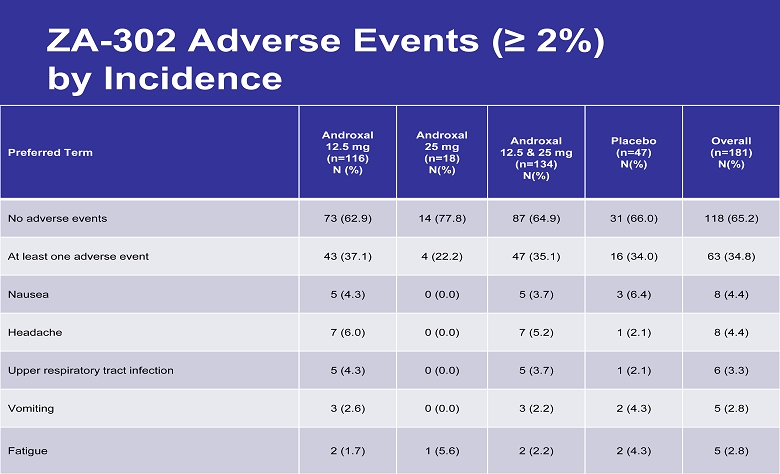
ZA - 302 Adverse Events (≥ 2 %) by Incidence Preferred Term Androxal 12.5 mg (n=116) N (%) Androxal 25 mg (n=18) N(%) Androxal 12.5 & 25 mg (n=134) N(%) Placebo (n=47) N(%) Overall (n=181) N(%) No adverse events 73 (62.9) 14 (77.8) 87 (64.9) 31 (66.0) 118 (65.2) At least one adverse event 43 (37.1) 4 (22.2) 47 (35.1) 16 (34.0) 63 (34.8) Nausea 5 (4.3) 0 (0.0) 5 (3.7) 3 (6.4) 8 (4.4) Headache 7 (6.0) 0 (0.0) 7 (5.2) 1 (2.1) 8 (4.4) Upper respiratory tract infection 5 (4.3) 0 (0.0) 5 (3.7) 1 (2.1) 6 (3.3) Vomiting 3 (2.6) 0 (0.0) 3 (2.2) 2 (4.3) 5 (2.8) Fatigue 2 (1.7) 1 (5.6) 2 (2.2) 2 (4.3) 5 (2.8)

ZA - 300 Population Summary • 499 Subjects Enrolled – 12.5 mg (n = 234) – 25.0 mg (n = 265), these subjects experienced a morning Testosterone less than 450 ng/dL and were up - titrated • Race/Ethnicity – 84.5% Caucasian (includes Hispanic ethnicity) – 11.3% African American – 4.2% Other • Mean Age 49 years • 33.7% Previous T Users • Baseline Testosterone 212 ng/dL • Mean BMI at baseline 33

ZA - 300 Testosterone Response 500 Subject Six Month Safety Study • Over 90% of subjects had a maximum morning Testosterone above 300 ng/dL • Almost 67% of subjects had a maximum morning Testosterone above 450 ng/dL • 53% of subjects were up - titrated (T < 450 ng/dL) • Only 3 subjects experienced a maximum morning Testosterone above 1,040 ng/dL ( 1720, 1479, 1264) – Two subjects reported concomitant testosterone replacement use – LH values of third subject suggest concomitant testosterone use • Subjects who required up - titration were less - likely to respond to treatment – 13.6% of subjects treated with 25 mg had a maximum morning Testosterone less than 300 ng/dL – 3.6% of subjects treated with 12.5 mg had a maximum morning Testosterone less than 300 ng/dL
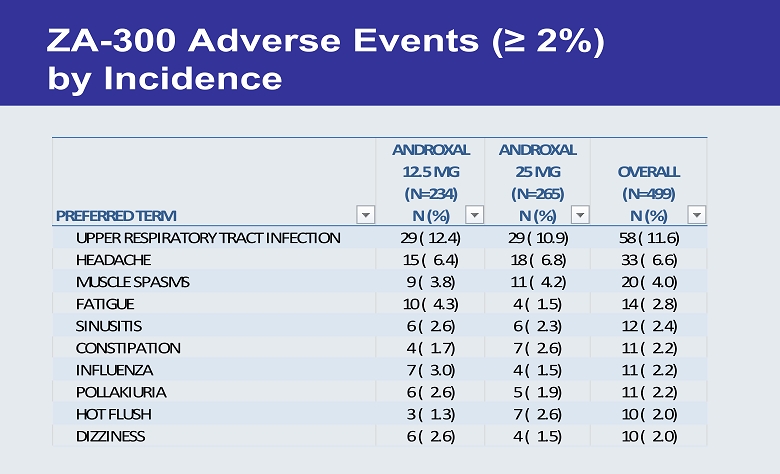
ZA - 300 Adverse Events (≥ 2 %) by Incidence PREFERRED TERM ANDROXAL 12.5 MG (N=234) N (%) ANDROXAL 25 MG (N=265) N (%) OVERALL (N=499) N (%) UPPER RESPIRATORY TRACT INFECTION 29 ( 12.4) 29 ( 10.9) 58 ( 11.6) HEADACHE 15 ( 6.4) 18 ( 6.8) 33 ( 6.6) MUSCLE SPASMS 9 ( 3.8) 11 ( 4.2) 20 ( 4.0) FATIGUE 10 ( 4.3) 4 ( 1.5) 14 ( 2.8) SINUSITIS 6 ( 2.6) 6 ( 2.3) 12 ( 2.4) CONSTIPATION 4 ( 1.7) 7 ( 2.6) 11 ( 2.2) INFLUENZA 7 ( 3.0) 4 ( 1.5) 11 ( 2.2) POLLAKIURIA 6 ( 2.6) 5 ( 1.9) 11 ( 2.2) HOT FLUSH 3 ( 1.3) 7 ( 2.6) 10 ( 2.0) DIZZINESS 6 ( 2.6) 4 ( 1.5) 10 ( 2.0)
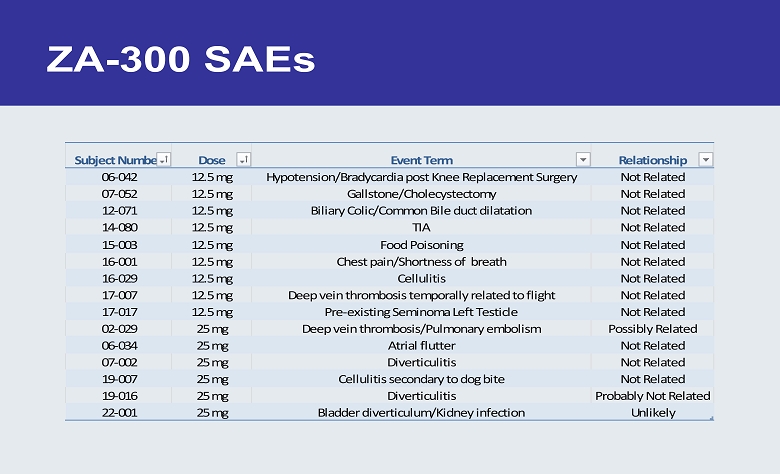
ZA - 300 SAEs Subject Number Dose Event Term Relationship 06-042 12.5 mg Hypotension/Bradycardia post Knee Replacement Surgery Not Related 07-052 12.5 mg Gallstone/Cholecystectomy Not Related 12-071 12.5 mg Biliary Colic/Common Bile duct dilatation Not Related 14-080 12.5 mg TIA Not Related 15-003 12.5 mg Food Poisoning Not Related 16-001 12.5 mg Chest pain/Shortness of breath Not Related 16-029 12.5 mg Cellulitis Not Related 17-007 12.5 mg Deep vein thrombosis temporally related to flight Not Related 17-017 12.5 mg Pre-existing Seminoma Left Testicle Not Related 02-029 25 mg Deep vein thrombosis/Pulmonary embolism Possibly Related 06-034 25 mg Atrial flutter Not Related 07-002 25 mg Diverticulitis Not Related 19-007 25 mg Cellulitis secondary to dog bite Not Related 19-016 25 mg Diverticulitis Probably Not Related 22-001 25 mg Bladder diverticulum/Kidney infection Unlikely
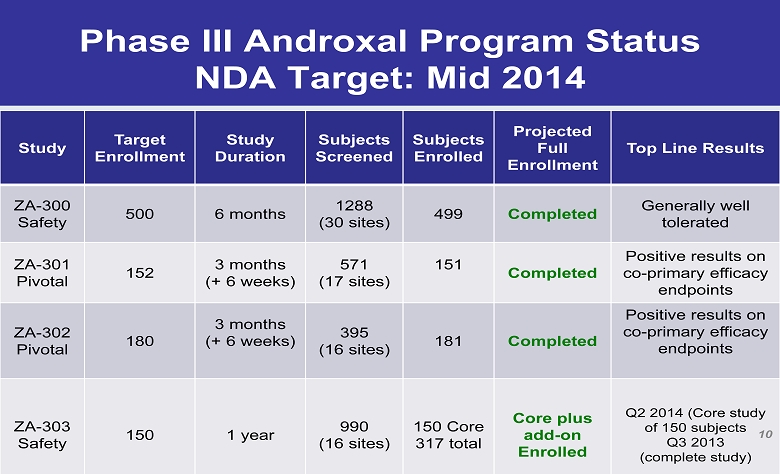
Phase III Androxal Program Status NDA Target: Mid 2014 Study Target Enrollment Study Duration Subjects Screened Subjects Enrolled Projected Full Enrollment Top Line Results ZA - 300 Safety 500 6 months 1288 (30 sites) 499 Completed Generally well tolerated ZA - 301 Pivotal 152 3 months (+ 6 weeks) 571 (17 sites) 151 Completed Positive results on co - primary efficacy endpoints ZA - 302 Pivotal 180 3 months (+ 6 weeks) 395 (16 sites) 181 Completed Positive results on co - primary efficacy endpoints ZA - 303 Safety 150 1 year 990 (16 sites) 150 Core 317 total Core plus add - on Enrolled Q2 2014 (Core study of 150 subjects Q3 2013 (complete study) 10

How do we maximize shareholder return? • All Repros Assets are Unencumbered • Androxal asset should be +90% de - risked during Q1 - ’14 • Androxal should open T treatment into primary care – Outside the scope of Repros capability • Measured launch of Androxal into specialty space • KOL board established (first meeting scheduled for Oct., ‘13) • Recent follow - on offering enables this option – Scope of effort controlled by Company • Third party assessment suggests feasibility of Repros marketing effort • Company to add seasoned marketing executive to Board of Directors to assess this option • Repros will remain opportunistic regarding transactions that maximize shareholder return 14
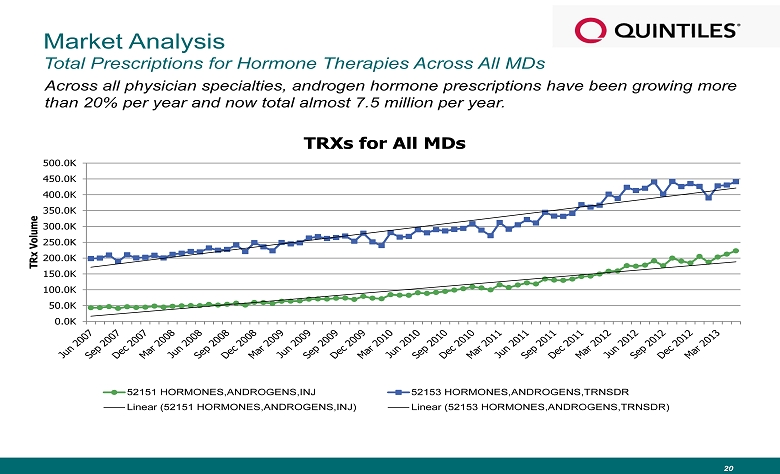
Market Analysis Total Prescriptions for Hormone Therapies Across All MDs Across all physician specialties, androgen hormone prescriptions have been growing more than 20% per year and now total almost 7.5 million per year. 20 0.0K 50.0K 100.0K 150.0K 200.0K 250.0K 300.0K 350.0K 400.0K 450.0K 500.0K TRx Volume TRXs for All MDs 52151 HORMONES,ANDROGENS,INJ 52153 HORMONES,ANDROGENS,TRNSDR Linear (52151 HORMONES,ANDROGENS,INJ) Linear (52153 HORMONES,ANDROGENS,TRNSDR)
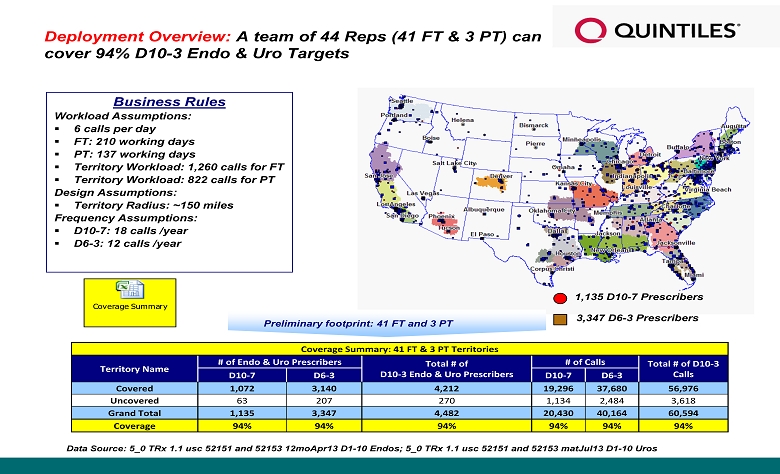
Preliminary footprint : 41 FT and 3 PT Business Rules Workload Assumptions : ▪ 6 calls per day ▪ FT: 210 working days ▪ PT: 137 working days ▪ Territory Workload: 1,260 calls for FT ▪ Territory Workload: 822 calls for PT Design Assumptions : ▪ Territory Radius: ~150 miles Frequency Assumptions: ▪ D10 - 7: 18 calls /year ▪ D6 - 3: 12 calls /year Deployment Overview : A team of 44 Reps (41 FT & 3 PT) can cover 94% D10 - 3 Endo & Uro Targets 1,135 D10 - 7 Prescribers 3,347 D6 - 3 Prescribers Data Source: 5_0 TRx 1.1 usc 52151 and 52153 12moApr13 D1 - 10 Endos; 5_0 TRx 1.1 usc 52151 and 52153 matJul13 D1 - 10 Uros Coverage Summary Coverage Summary: 41 FT & 3 PT Territories Territory Name # of Endo & Uro Prescribers Total # of D10 - 3 Endo & Uro Prescribers # of Calls Total # of D10 - 3 Calls D10 - 7 D6 - 3 D10 - 7 D6 - 3 Covered 1,072 3,140 4,212 19,296 37,680 56,976 Uncovered 63 207 270 1,134 2,484 3,618 Grand Total 1,135 3,347 4,482 20,430 40,164 60,594 Coverage 94% 94% 94% 94% 94% 94%
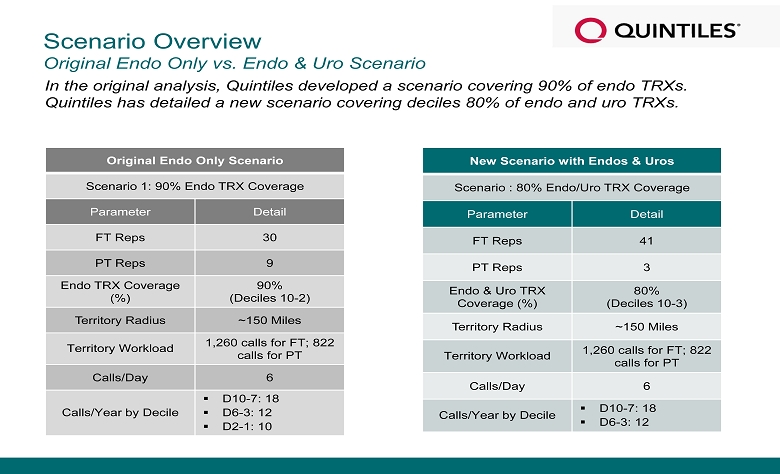
Scenario Overview Original Endo Only vs. Endo & Uro Scenario In the original analysis, Quintiles developed a scenario covering 90% of endo TRXs. Quintiles has detailed a new scenario covering deciles 80% of endo and uro TRXs. Original Endo Only Scenario Scenario 1: 90% Endo TRX Coverage Parameter Detail FT Reps 30 PT Reps 9 Endo TRX Coverage (%) 90% (Deciles 10 - 2) Territory Radius ~150 Miles Territory Workload 1,260 calls for FT; 822 calls for PT Calls/Day 6 Calls/Year by Decile ▪ D10 - 7: 18 ▪ D6 - 3: 12 ▪ D2 - 1: 10 New Scenario with Endos & Uros Scenario : 80% Endo/Uro TRX Coverage Parameter Detail FT Reps 41 PT Reps 3 Endo & Uro TRX Coverage (%) 80% (Deciles 10 - 3) Territory Radius ~150 Miles Territory Workload 1,260 calls for FT; 822 calls for PT Calls/Day 6 Calls/Year by Decile ▪ D10 - 7: 18 ▪ D6 - 3: 12
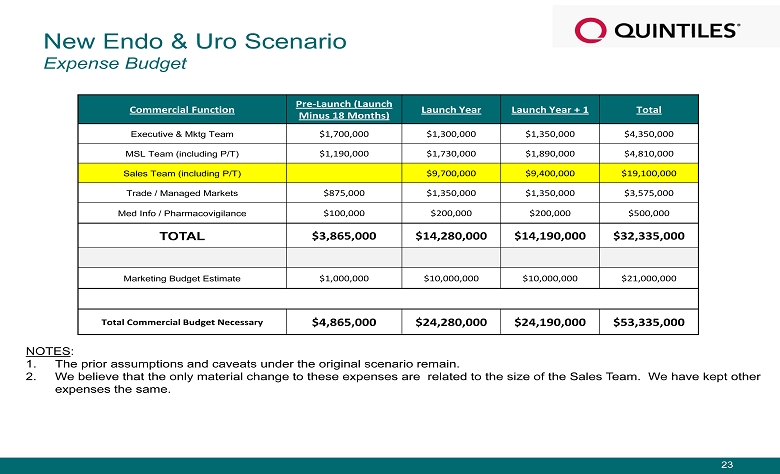
NOTES : 1. The prior assumptions and caveats under the original scenario remain. 2. We believe that the only material change to these expenses are related to the size of the Sales Team. We have kept other expenses the same. Commercial Function Pre - Launch (Launch Minus 18 Months) Launch Year Launch Year + 1 Total Executive & Mktg Team $1,700,000 $1,300,000 $1,350,000 $4,350,000 MSL Team (including P/T) $1,190,000 $1,730,000 $1,890,000 $4,810,000 Sales Team (including P/T) $9,700,000 $9,400,000 $19,100,000 Trade / Managed Markets $875,000 $1,350,000 $1,350,000 $3,575,000 Med Info / Pharmacovigilance $100,000 $200,000 $200,000 $500,000 TOTAL $3,865,000 $ 14,280,000 $ 14,190,000 $32,335,000 Marketing Budget Estimate $1,000,000 $10,000,000 $10,000,000 $21,000,000 Total Commercial Budget Necessary $4,865,000 $24,280,000 $24,190,000 $53,335,000 New Endo & Uro Scenario Expense Budget 23
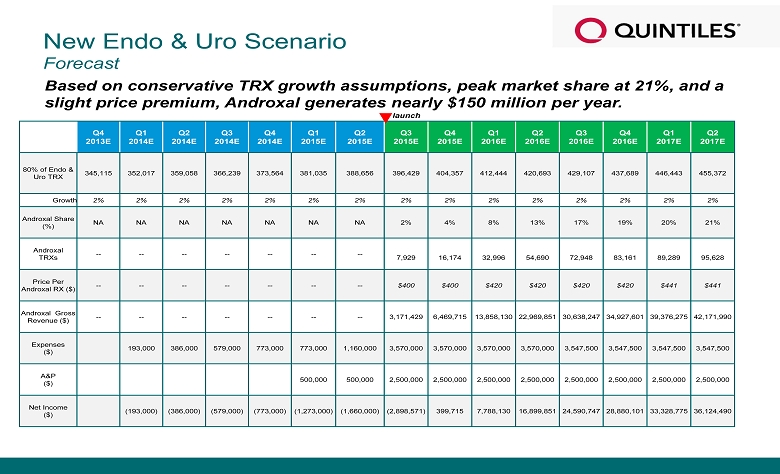
New Endo & Uro Scenario Forecast Based on conservative TRX growth assumptions, peak market share at 21%, and a slight price premium, Androxal generates nearly $150 million per year. Q4 2013E Q1 2014E Q2 2014E Q3 2014E Q4 2014E Q1 2015E Q2 2015E Q3 2015E Q4 2015E Q1 2016E Q2 2016E Q3 2016E Q4 2016E Q1 2017E Q2 2017E 80% of Endo & Uro TRX 345,115 352,017 359,058 366,239 373,564 381,035 388,656 396,429 404,357 412,444 420,693 429,107 437,689 446,443 455,372 Growth 2% 2% 2% 2% 2% 2% 2% 2% 2% 2% 2% 2% 2% 2% 2% Androxal Share (%) NA NA NA NA NA NA NA 2% 4% 8% 13% 17% 19% 20% 21% Androxal TRXs -- -- -- -- -- -- -- 7,929 16,174 32,996 54,690 72,948 83,161 89,289 95,628 Price Per Androxal RX ($) -- -- -- -- -- -- -- $400 $400 $420 $420 $420 $420 $441 $441 Androxal Gross Revenue ($) -- -- -- -- -- -- -- 3,171,429 6,469,715 13,858,130 22,969,851 30,638,247 34,927,601 39,376,275 42,171,990 Expenses ($ ) 193,000 386,000 579,000 773,000 773,000 1,160,000 3,570,000 3,570,000 3,570,000 3,570,000 3,547,500 3,547,500 3,547,500 3,547,500 A&P ($) 500,000 500,000 2,500,000 2,500,000 2,500,000 2,500,000 2,500,000 2,500,000 2,500,000 2,500,000 Net Income ($) (193,000) (386,000) (579,000) (773,000) (1,273,000) (1,660,000) (2,898,571) 399,715 7,788,130 16,899,851 24,590,747 28,880,101 33,328,775 36,124,490 launch
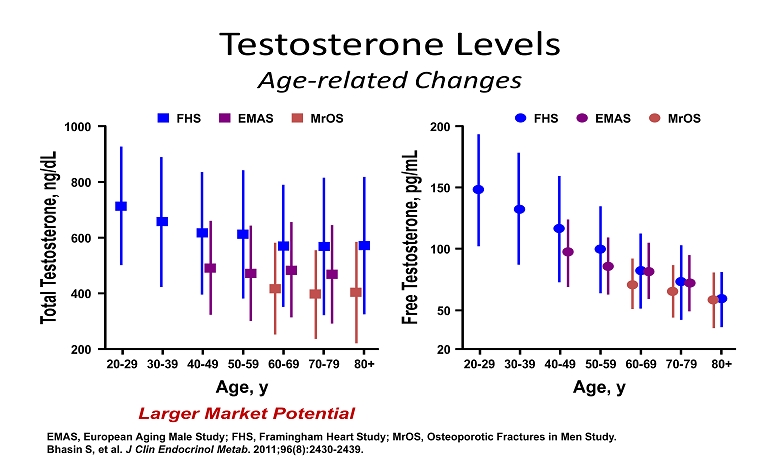
Testosterone Levels Age - related Changes EMAS, European Aging Male Study; FHS, Framingham Heart Study; MrOS, Osteoporotic Fractures in Men Study. Bhasin S, et al. J Clin Endocrinol Metab . 2011;96(8):2430 - 2439. Total Testosterone, ng/dL 20 - 29 30 - 39 40 - 49 50 - 59 60 - 69 70 - 79 1000 800 200 600 400 80+ Age, y FHS EMAS MrOS Free Testosterone, pg/mL 20 - 29 30 - 39 40 - 49 50 - 59 60 - 69 70 - 79 200 150 20 100 50 80+ Age, y FHS EMAS MrOS Larger Market Potential

Androxal Exhibits Unique Profile with Numerous Advantages vs Approved Hormone Replacement • The Androxal Advantages – Oral – Not controlled substance, cannot be abused – No transference risk – Restores normal function (no loss of testicular function) – Does not develop dependency – Avoids withdrawal symptoms – With lifestyle change can reverse disorder and result in no need for therapy
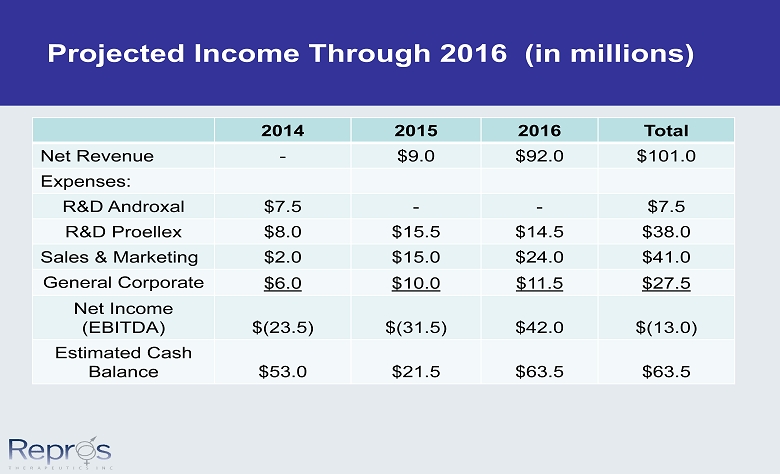
Projected Income Through 2016 (in millions) 2014 2015 2016 Total Net Revenue - $9.0 $92.0 $101.0 Expenses: R&D Androxal $7.5 - - $7.5 R&D Proellex $8.0 $15.5 $14.5 $38.0 Sales & Marketing $2.0 $15.0 $24.0 $41.0 General Corporate $6.0 $10.0 $11.5 $27.5 Net Income (EBITDA) $(23.5) $(31.5) $42.0 $(13.0) Estimated Cash Balance $53.0 $21.5 $63.5 $63.5

Proellex for the Treatment of Uterine Fibroids and Endometriosis • Over 30 million women of reproductive age in the US afflicted with symptomatic uterine fibroids or endometriosis • Over 300,000 hysterectomies performed every year in the US to treat these two disorders • No acceptable chronic therapeutic options available today 19
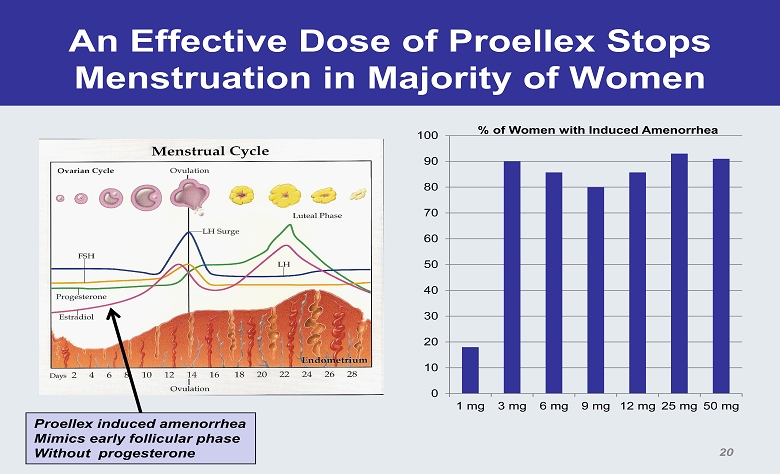
An Effective Dose of Proellex Stops Menstruation in Majority of Women 0 10 20 30 40 50 60 70 80 90 100 1 mg 3 mg 6 mg 9 mg 12 mg 25 mg 50 mg % of Women with Induced Amenorrhea Proellex induced amenorrhea Mimics early follicular phase Without progesterone 20
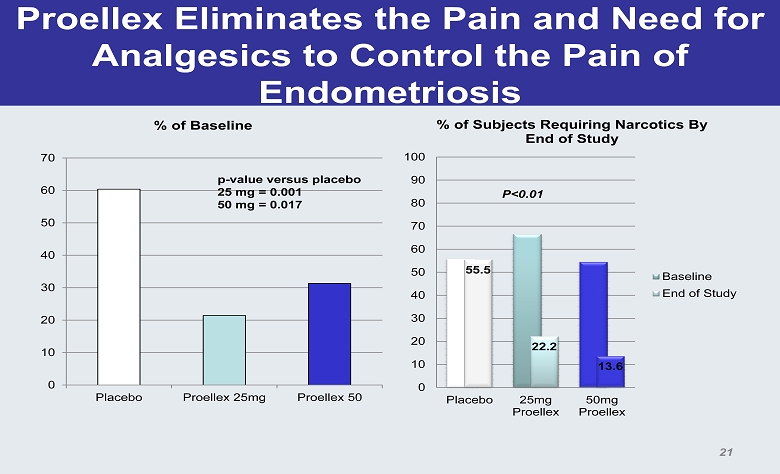
Proellex Eliminates the Pain and Need for Analgesics to Control the Pain of Endometriosis 0 10 20 30 40 50 60 70 Placebo Proellex 25mg Proellex 50 % of Baseline p - value versus placebo 25 mg = 0.001 50 mg = 0.017 55.5 22.2 13.6 0 10 20 30 40 50 60 70 80 90 100 Placebo 25mg Proellex 50mg Proellex % of Subjects Requiring Narcotics By End of Study Baseline End of Study P<0.01 21

ZPE - 202 Phase 2 Endometriosis Study • 90 subject double blind placebo controlled study balanced between placebo, 6 and 12 mg oral Proellex – Subject population (confirmed endometriosis) • Severe endometriosis as determined by BBSS score • Requiring narcotics or prescription analgesics to control endometriosis related pain – Study Duration: 4 months – Study endpoints: • Reduction in need for analgesics from baseline • Change from baseline in BBSS pain scores – Status: enrolling sites and subjects 22

Vaginal Proellex to Eliminate the Need for Hysterectomy in Most Situations • Initial Phase 2 study tested four doses of vaginal administration in the treatment of uterine fibroids completed – Assess reduction of fibroid size and elimination of symptoms – Top line data reported • FDA requires additional Phase 2b study before proceeding to Phase 3 to insure proper dose selection – Propose 90 subject Phase 2b study – Separate IND from low dose oral 23
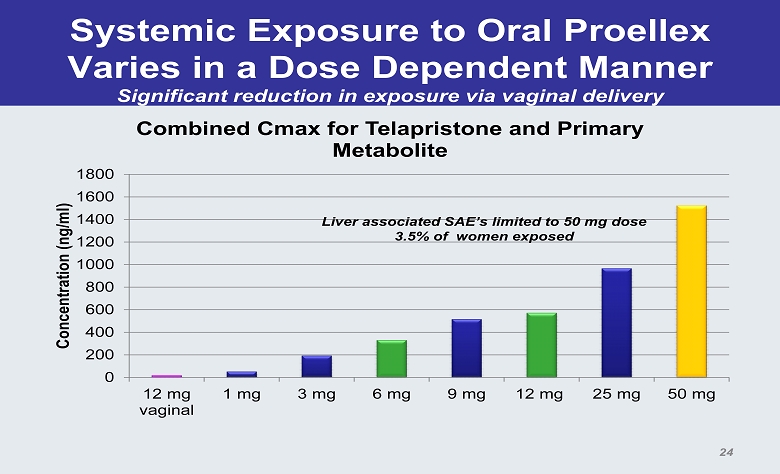
Systemic Exposure to Oral Proellex Varies in a Dose Dependent Manner Significant reduction in exposure via vaginal delivery 0 200 400 600 800 1000 1200 1400 1600 1800 12 mg vaginal 1 mg 3 mg 6 mg 9 mg 12 mg 25 mg 50 mg Concentration (ng/ml) Combined Cmax for Telapristone and Primary Metabolite Liver associated SAE’s limited to 50 mg dose 3.5% of women exposed 24
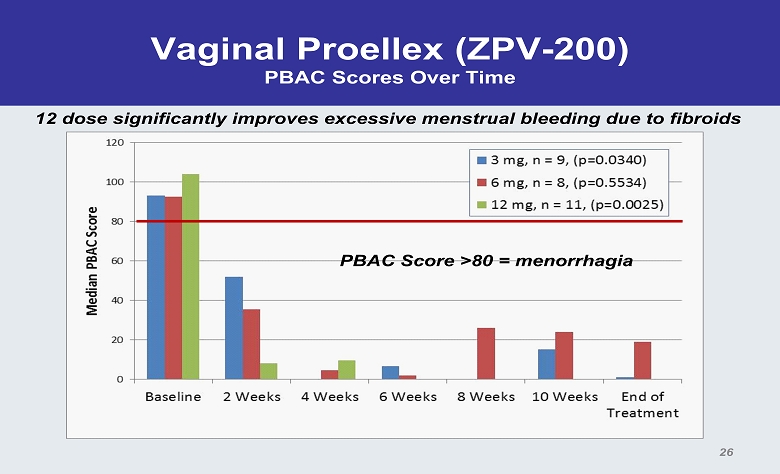
Vaginal Proellex (ZPV - 200) PBAC Scores Over Time PBAC Score >80 = menorrhagia 26 12 dose significantly improves excessive menstrual bleeding due to fibroids
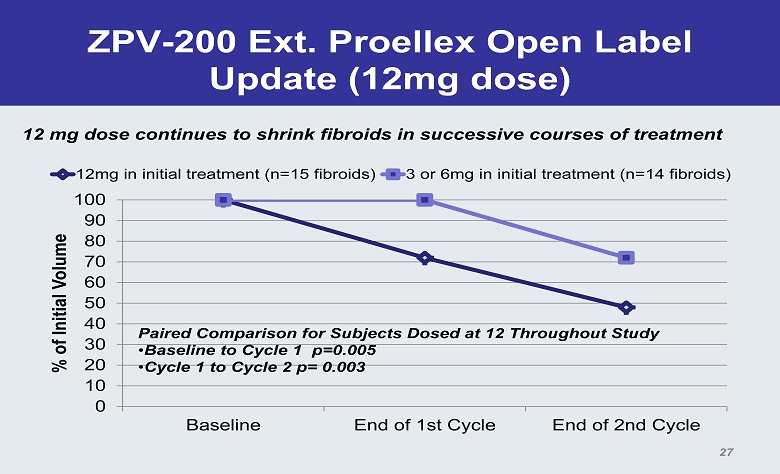
ZPV - 200 Ext. Proellex Open Label Update (12mg dose) 0 10 20 30 40 50 60 70 80 90 100 Baseline End of 1st Cycle End of 2nd Cycle % of Initial Volume 12mg in initial treatment (n=15 fibroids) 3 or 6mg in initial treatment (n=14 fibroids) Paired Comparison for Subjects Dosed at 12 Throughout Study • Baseline to Cycle 1 p=0.005 • Cycle 1 to Cycle 2 p= 0.003 27 12 mg dose continues to shrink fibroids in successive courses of treatment

Proellex Clinical Goals • Enter Phase 3 • Uterine Fibroids: Second half 2014 • Endometriosis: Fourth Quarter 2014 • Submit 1 st Proellex NDA • Second Half 2016 • Pre - clinical complete • Phase 1 complete except TQtc

Financial Summary • Cash and equivalents (as of 9/20/13) $82.0 M • Cash runway: 2016 – Both drugs’ NDAs filed – Anticipate Androxal approved and launched • Current shares outstanding: 23.0 M shares – Warrants Outstanding – Series A – 877,137 (purchased in unit deal @ $2.45); Series B – 810,109 @ $2.49 exercise price. 29