SCHEDULE 14A
(RULE 14a-101)
INFORMATION REQUIRED IN PROXY STATEMENT
SCHEDULE 14A INFORMATION
SCHEDULE 14A INFORMATION
Proxy Statement Pursuant to Section 14(a) of the Securities
Exchange Act of 1934 (Amendment No. )
Exchange Act of 1934 (Amendment No. )
Filed by the Registrant þ
Filed by a Party other than the Registrant o
Filed by a Party other than the Registrant o
Check the appropriate box:
o Preliminary Proxy Statement
o Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2))
o Definitive Proxy Statement
þ Definitive Additional Materials
o Soliciting Material Pursuant to Rule 14a-11c or Rule 14a-12
o Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2))
o Definitive Proxy Statement
þ Definitive Additional Materials
o Soliciting Material Pursuant to Rule 14a-11c or Rule 14a-12
Human Genome Sciences, Inc.
(Name of Registrant as Specified In Its Charter)
(Name of Person(s) Filing Proxy Statement)
Payment of Filing Fee (Check the appropriate box):
| þ | No fee required. | ||
| o | Fee computed on table below per Exchange Act Rules 14a-6(i)(1) and 0-11. |
| (1) | Title of each class of securities to which transaction applies: | ||
| (2) | Aggregate number of securities to which transaction applies: | ||
| (3) | Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): | ||
| (4) | Proposed maximum aggregate value of transaction: | ||
| (5) | Total fee paid: |
| o | Fee paid previously with preliminary materials. | ||
| o | Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. |
| (1) | Amount Previously Paid: | ||
| (2) | Form, Schedule or Registration Statement No.: | ||
| (3) | Filing Party: | ||
| (4) | Date Filed: |
EXPLANATORY NOTE
The following letter to stockholders and information about Human Genome Sciences, Inc. were included in the company's annual report to stockholders that was distributed on or about March 30, 2011.
Cautionary Statement Regarding Forward-Looking Statements
This report includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward-looking statements are based on our current intent, belief and expectations. These statements are not guarantees of future performance and are subject to certain risks and uncertainties that are difficult to predict. Actual results may differ materially from these forward-looking statements because of our unproven business model, our dependence on new technologies, the uncertainty and timing of clinical trials, our ability to develop and commercialize products, our dependence on collaborators for services and revenue, our substantial indebtedness and lease obligations, our changing requirements and costs associated with facilities, intense competition, the uncertainty of patent and intellectual property protection, our dependence on key management and key suppliers, the uncertainty of regulation of products, the impact of future alliances or transactions and other risks described in our other filings with the Securities and Exchange Commission. Existing and prospective investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today’s date. We undertake no obligation to update or revise the information contained in this report whether as a result of new information, future events or circumstances or otherwise.
TO OUR SHAREHOLDERS
OVER THE PAST SEVERAL YEARS, WE HAVE STEADILY MOVED HUMAN GENOME SCIENCES FORWARD TOWARD OUR GOAL OF BECOMING A FULLY COMMERCIAL BIOPHARMACEUTICAL COMPANY—WITH IMPORTANT PRODUCTS ON THE MARKET AND ABLE TO GENERATE SUSTAINABLE REVENUES AND SUSTAINABLE GROWTH. WITH THE MARCH 9, 2011 FDA APPROVAL OF BENLYSTA®, OUR GOAL IS FINALLY WITHIN REACH.
WE AND GLAXOSMITHKLINE (GSK) ARE HONORED TO BRING BENLYSTA FORWARD IN THE UNITED STATES AS THE FIRST NEW DRUG FOR SYSTEMIC LUPUS IN MORE THAN 50 YEARS. THIS NOVEL THERAPY IS NOW AVAILABLE TO PHYSICIANS AND PATIENTS IN THE U.S.
The approval of BENLYSTA opens the door to a promising future for our Company. Based on the opportunities we now have in hand and our expectation that decisions will be reached on European and other regulatory approvals in the second half of 2011, we believe that HGS will achieve sustainable multi-billion dollar revenues by 2015.
As we move forward, we have three over-arching strategic priorities: first, the successful launch and commercialization of BENLYSTA; second, achieving the full therapeutic and commercial potential of BENLYSTA by developing it for other B-cell mediated diseases; and third, to continue to build strength at every stage of our pipeline.
BENLYSTA Launch and Commercialization
In June 2010, HGS and GSK submitted regulatory applications seeking approval to market BENLYSTA (belimumab) in the United States and Europe, respectively. In November 2010, the FDA Arthritis Advisory Committee voted 13 to 2 to recommend that the FDA approve BENLYSTA in the U.S. for the treatment of autoantibody-positive adult patients with active systemic lupus erythematosus who are receiving standard therapy. In March 2011, the FDA approved BENLYSTA for marketing in the U.S. with the indication we requested, limited only by the inclusion/exclusion criteria of the Phase 3 studies.
With BENLYSTA now approved by FDA, our first strategic priority is to achieve a world-class launch and the successful commercialization of a drug that we believe represents a potential improvement in the standard of care for systemic lupus. Over the past year, we have hired and trained a specialized commercial team that we believe is second to none in their experience and familiarity with rheumatology, biologics and infused products. We are ready to hit the ground running alongside GlaxoSmithKline to help ensure that BENLYSTA realizes its full therapeutic and commercial potential. The combined HGS and GSK team, including a U.S. sales
HUMAN GENOME SCIENCES ANNUAL REPORT 2010 1

H. THOMAS WATKINS
President and Chief Executive Officer
President and Chief Executive Officer

ARGERIS (JERRY) N. KARABELAS, PH.D.
Chairman of the Board
Chairman of the Board
force of approximately 150, will work closely together to communicate effectively and appropriately with rheumatologists and other stakeholders and ensure that patients with systemic lupus who need BENLYSTA will have access to it. HGS and GSK will also support patients with appropriate reimbursement and access support programs.
Under the terms of our 2006 co-development and co-commercialization agreement with GSK, HGS has responsibility for the global supply of BENLYSTA. We are fully prepared to meet that responsibility. We believe we already have the inventory to meet global market needs for at least one year, and our large-scale manufacturing facility has sufficient capacity to provide worldwide supply for the first two to three years following launch. In addition, in July 2010, we announced an agreement with Lonza that will eventually triple our manufacturing capacity with the new production expected to come on line by mid-2012.
We and GSK expect to have a decision on the European approval of BENLYSTA in the second half of 2011. Regulatory applications have also been submitted and are currently under consideration in Canada, Australia, Switzerland, Russia, Brazil and The Philippines — and regulatory submissions are planned in additional countries in the coming months. HGS is building its own commercialization team to work alongside GSK in Europe, with headquarters in Switzerland and local organizations in Germany, France and Spain. In other countries, GSK will lead local implementation, with HGS sharing costs and profits equally with GSK.
Achieving the Full Potential of BENLYSTA
BENLYSTA is clearly the most important driver of our Company’s growth over the next several years. Our second strategic priority is to achieve the full therapeutic and commercial potential of BENLYSTA by developing it for other B-cell mediated diseases where patients need new treatment options and strong scientific evidence supports therapeutic and commercial potential. We and GSK are examining a range of potential additional indications that might be explored in new clinical trials.
We plan to initiate a Phase 2 trial of BENLYSTA in vasculitis in the second half of 2011. We also plan a trial in lupus nephritis, with trial design and timing dependent on further conversations with the FDA. In addition, a number of small exploratory studies are already ongoing or imminent in Sjögren’s syndrome, Waldenstrom’s disease, idiopathic thrombocytopenic purpura, myasthenia gravis, idiopathic membranous nephropathy and others.
“WITH BENLYSTA NOW APPROVED BY FDA, OUR FIRST STRATEGIC PRIORITY IS TO ACHIEVE A WORLD-CLASS LAUNCH AND THE SUCCESSFUL COMMERCIALIZATION OF A DRUG THAT WE BELIEVE REPRESENTS A POTENTIAL IMPROVEMENT IN THE STANDARD OF CARE FOR SYSTEMIC LUPUS.”
2 HUMAN GENOME SCIENCES ANNUAL REPORT 2010
“OUR SECOND STRATEGIC PRIORITY IS TO ACHIEVE THE FULL THERAPEUTIC AND COMMERCIAL POTENTIAL OF BENLYSTA BY DEVELOPING IT FOR OTHER B-CELL MEDIATED DISEASES WHERE PATIENTS NEED NEW TREATMENT OPTIONS AND STRONG SCIENTIFIC EVIDENCE SUPPORTS THERAPEUTIC AND COMMERCIAL POTENTIAL.”
Further study of BENLYSTA in systemic lupus is planned as well, including a Phase 3 trial in Asia, and a Phase 3 trial of a subcutaneous formulation—both scheduled for initiation in 2011. In the coming months, we will also be discussing with FDA the timing and design of studies for which we have made Phase 4 commitments.
Building Strength at Every Stage of the Pipeline
Our third strategic priority is to continue to build strength at every stage of our product pipeline. We will selectively pursue external opportunities to acquire high-potential innovative products and we will develop investigational products based on our own discoveries.
We will give priority to programs that address large markets or markets on the verge of escalating growth.
We want products that can be first-in-class or highly differentiated best-in-class. We expect to maintain our primary therapeutic focus on immunology and oncology—indications in which we have special expertise and which can be commercialized with small specialty sales forces. While we are recognized for our core competencies in biologics, we are open to both biologics and small molecule opportunities.
We underscored our commitment to developing novel targeted therapies that address significant unmet medical needs when we announced on March 16, 2011 a collaboration with FivePrime Therapeutics to develop and commercialize FP-1039, a first-in-class biologic that targets multiple fibroblast growth factor ligands and is
PRODUCT PIPELINE
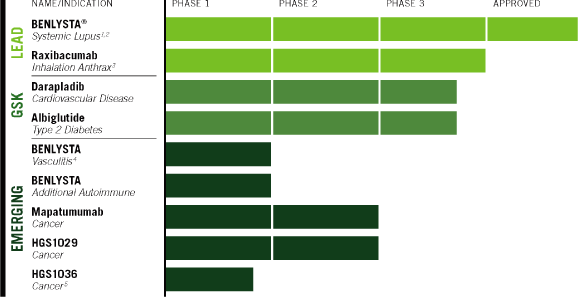
| 1 | Initiation of Phase 3 trial in Asia planned 2011 | |
| 2 | Initiation of Phase 3 trial of subcutaneous formulation planned 2011 | |
| 3 | Sales to Strategic National Stockpile under U.S. Government contract | |
| 4 | Initiation of Phase 2 trial planned 2011 | |
| 5 | Formerly FP-1039; rights acquired March 2011 from FivePrime Therapeutics |
HUMAN GENOME SCIENCES ANNUAL REPORT 2010 3
“OUR THIRD STRATEGIC PRIORITY IS TO CONTINUE TO BUILD STRENGTH AT EVERY STAGE OF OUR PRODUCT PIPELINE.”
being studied in early-stage clinical trials in a variety of cancers. Under the terms of the agreement, HGS will have rights to develop and commercialize FP-1039 in the United States, Canada and Europe.
We will also initiate the randomization stage of our trial of mapatumumab with Nexavar in hepatocellular cancer. Mapatumumab is an investigational product that we have developed based on our own discoveries. In addition, we plan to initiate Phase 1B combination trials of our in-licensed small-molecule oncology drug, HGS1029.
We also look forward to GSK’s continuing progress with late-stage products to which we have substantial financial rights. In two pivotal Phase 3 trials, GSK is currently evaluating whether darapladib can reduce the risk of adverse cardiovascular events such as heart attack or stroke in patients with chronic coronary heart disease and acute coronary syndrome, respectively. In addition, GSK has eight Phase 3 trials in progress to evaluate the long-term efficacy, safety and tolerability of albiglutide as monotherapy and add-on therapy for patients with type 2 diabetes mellitus. Darapladib was discovered by GSK based on HGS technology. HGS will receive 10% royalties on worldwide sales if darapladib is commercialized, and has a 20% co-promotion option in North America and Europe. Albiglutide was created by HGS using its proprietary albumin-fusion technology, and the product was licensed to GSK in 2004. HGS is entitled to fees and milestone payments that could amount to as much as $183 million—including $33 million received to date—in addition to single-digit royalties on worldwide sales if albiglutide is commercialized.
Raxibacumab
In July 2009, the U.S. Government exercised its option to purchase 45,000 additional doses of raxibacumab for the U.S. Strategic National Stockpile for emergency use in treating inhalation anthrax, with delivery to be completed over a three-year period. HGS expects to receive approximately $142 million from this second award as deliveries are completed, including approximately $51 million received in 2010.
Raxibacumab is being developed under a contract entered into in 2006 with the Biomedical Advanced Research and Development Authority (BARDA) of the Office of the Assistant Secretary for Preparedness and Response (ASPR), U.S. Department of Health and Human Services (HHS). HGS continues to work closely with FDA to achieve licensure of raxibacumab. HGS is entitled to receive $20 million under its contract with the U.S. Government upon FDA licensure.
Leadership Team
We are delighted to welcome three new members to our Board of Directors: Dr. Colin Goddard, former Chief Executive Officer of OSI Pharmaceuticals; Greg Norden, former Senior Vice President and Chief Financial Officer of Wyeth; and George J. Morrow, former Executive Vice President and Chief Commercial Officer of Amgen, Inc. We are honored to strengthen our Board with the addition of these proven leaders in our industry, and we look forward to benefitting from their guidance and insight.
We expect 2011 to be another transformational year for HGS, as we focus, along with GSK, on the successful launch and commercialization of BENLYSTA. The approval of BENLYSTA opens the door to many new opportunities. Our cash position remains strong, and we are well prepared to seize and capitalize on our opportunities and continue to drive growth in the years to come.
We thank you for your continued support.
H. Thomas Watkins
President and Chief Executive Officer
President and Chief Executive Officer
Argeris (Jerry) N. Karabelas, Ph.D.
Chairman of the Board
Chairman of the Board
4 HUMAN GENOME SCIENCES ANNUAL REPORT 2010

The last drugs approved to treat lupus in the United States were Plaquenil, originally developed to treat malaria, and corticosteroids, a class of drugs widely used to treat inflammatory illnesses. Both were approved by FDA in 1955. Aspirin was approved to treat lupus in 1948.
Before 2009, no drug developed specifically for lupus had ever made it through a Phase 3 clinical trial with positive results. During 2009, BENLYSTA achieved positive results in two pivotal Phase 3 trials that subsequently became the primary basis of regulatory submissions in the U.S. and around the world. This was the largest clinical trial program ever completed in patients with systemic lupus.
Primary U.S. Target Market: 200,000 Patients
HGS believes BENLYSTA has the potential to improve the standard of care for patients with systemic lupus. It also represents a very substantial market opportunity. HGS market research data suggests that there are currently about 325,000 patients in the U.S. who are diagnosed with systemic lupus and under the care of a rheumatologist. About two thirds of these patients will have moderate-to-severe disease at some point. It is these 200,000 patients who are likely to benefit most from BENLYSTA. They represent the primary target market in the U.S.
The FDA approved BENLYSTA for use in the treatment of adult patients with active, autoantibody-positive systemic lupus erythematosus (SLE) who are receiving standard therapy. The indication statement accurately describes the patient population in the BENLYSTA Phase 3 trials. It includes a real-world spectrum of patients with active SLE disease and is not specific to any particular manifestations.1
The limitations of use are consistent with the inclusion/ exclusion criteria applied in the BENLYSTA studies. BENLYSTA is not recommended for the treatment of patients taking other biologics or intravenous cyclophosphamide or those withsevere active lupus nephritis orsevere activeCNS lupus—both of which
| 1 | Full BENLYSTA (belimumab) prescribing information is available at www.benlysta.com. |
HUMAN GENOME SCIENCES ANNUAL REPORT 2010 5
HGS BELIEVES BENLYSTA HAS THE POTENTIAL TO IMPROVE THE STANDARD OF CARE FOR PATIENTS WITH SYSTEMIC LUPUS. IT ALSO REPRESENTS A VERY SUBSTANTIAL MARKET OPPORTUNITY.
are acute manifestations occurring at a point in time during the course of a patient’s disease. Patients experiencing these severe manifestations of disease were excluded from the Phase 3 trials because they had the most immediate need for acute intervention with highly aggressive therapies.
The BENLYSTA trials did include a limited number of patients with renal or CNS manifestations that were not severely active, and HGS expects that physicians will consider BENLYSTA for these types of patients.1
Encouraging Appropriate Use of BENLYSTA
HGS and GSK have detailed plans in place for bringing BENLYSTA to patients and the medical professionals who treat them. The objective, consistent with the labeling received from FDA, is to work with healthcare professionals, patients and payers to encourage appropriate use of BENLYSTA.
HGS and GSK have built a high-quality sales team— totaling approximately 150 account managers—who will focus on rheumatology accounts, both in hospitals and in the community. They are well trained and prepared to share the clinical data from the BENLYSTA studies and encourage appropriate patient selection. The sales force, along with payer and reimbursement experts, will also provide education and support for the complicated reimbursement process for infused buy-and-bill biologics.
People living with lupus tend to be very interested in information about their disease and treatment options. So, education and support programs will be provided for patients—largely through educational materials in physicians’ offices and through on-line programs—to help encourage dialogue between patients and their healthcare providers.
Pricing and Access
Pricing and access are key components of the BENLYSTA commercialization plan. The price for the average patient will be about $35,000 per year, which is in the range of biologic therapies for other chronic autoimmune diseases. For example, most of the biologics for multiple sclerosis are priced at around $40,000, and most of the biologics for rheumatoid arthritis are priced at more than $20,000.
Based on significant market research we expect payers to provide broad coverage for BENLYSTA under the medical benefit. Prior authorization requirements will be implemented by payers for BENLYSTA as they are for most biologics.
The HGS and GSK goal in working with payers is to ensure that the requirements for prior therapies allow
BASED ON SIGNIFICANT MARKET RESEARCH WE EXPECT PAYERS TO PROVIDE BROAD COVERAGE FOR BENLYSTA UNDER THE MEDICAL BENEFIT.
| 1 | Full BENLYSTA (belimumab) prescribing information is available at www.benlysta.com. |
6 HUMAN GENOME SCIENCES ANNUAL REPORT 2010

access to BENLYSTA for patients who have persistent disease activity despite treatment with standard therapy.
To the extent that patients are responsible for part of their drug costs, HGS and GSK will have programs available to assist. Uninsured low-income patients who meet certain eligibility requirements will have access to free drug through the BENLYSTA patient assistance program, and a copay program will help cover out-of-pocket costs for eligible commercially insured patients.
The BENLYSTA distribution strategy is now being implemented and product is now available to physicians and patients in the United States.
Europe and Other Regions of the World
HGS and GSK look forward to a decision on European approval of BENLYSTA in the second half of 2011. Regulatory applications have also been submitted and are currently under consideration in Canada, Australia, Switzerland, Russia, Brazil and The Philippines.
HGS is building its own commercialization team to work alongside GSK in Europe. HGS Europe is headquartered in Switzerland and HGS is creating local organizations in Germany, France and Spain. In these three key countries, HGS teams will work alongside GSK to commercialize BENLYSTA. Strong relationships have already been established by HGS teams with the local GSK country teams as well as the regional headquarters. In other countries, HGS will rely upon GSK for local implementation, with HGS and GSK sharing costs and profits equally.
The BENLYSTA commercialization team is in place and highly energized—a very talented group of people at both HGS and GSK who are well prepared and passionate about making sure that patients who can benefit from BENLYSTA get that opportunity.
The commercialization plans are well defined and the team is committed to achieving the world-class launch that BENLYSTA deserves.
HUMAN GENOME SCIENCES ANNUAL REPORT 2010 7