| PROXY STATEMENT | ||||||||
| INFORMATION CONCERNING SOLICITATION AND VOTING | ||||||||
| SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT | ||||||||
| PROPOSALS TO BE VOTED ON AT THE ANNUAL MEETING | ||||||||
| ADDITIONAL INFORMATION | ||||||||
Table of Contents
SCHEDULE 14A
INFORMATION REQUIRED IN PROXY STATEMENT
SCHEDULE 14A INFORMATION
Filed by the Registrant þ | ||
Filed by a Party other than the Registrant o | ||
| Check the appropriate box: | ||
o Preliminary Proxy Statement | o Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) | |
þ Definitive Proxy Statement | ||
o Definitive Additional Materials | ||
| o Soliciting Material Pursuant to Rule 14a-11(c) or Rule 14a-12 | ||
Human Genome Sciences, Inc.
Payment of Filing Fee (Check the appropriate box):
þ No fee required.
o Fee computed on table below per Exchange Act Rules 14a-6(i)(1) and 0-11.
(1) Title of each class of securities to which transaction applies:
(2) Aggregate number of securities to which transaction applies:
| (3) | Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (Set forth the amount on which the filing fee is calculated and state how it was determined): |
(4) Proposed maximum aggregate value of transaction:
(5) Total fee paid:
| o | Fee paid previously with preliminary materials. | |
| o | Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the form or schedule and the date of its filing. |
(1) Amount previously paid:
(2) Form, schedule or registration statement no.:
(3) Filing party:
(4) Date filed:
Table of Contents
HUMAN GENOME SCIENCES, INC.
NOTICE OF ANNUAL MEETING OF STOCKHOLDERS
to be held May 22, 2002
To the Stockholders of Human Genome Sciences, Inc.:
NOTICE IS HEREBY GIVEN that the 2002 Annual Meeting of Stockholders (the “Annual Meeting”) of Human Genome Sciences, Inc., a Delaware corporation (the “Company”), will be held at the University System of Maryland — Shady Grove Center, Germantown Room, 9630 Gudelsky Drive, Rockville, Maryland 20850 on Wednesday, May 22, 2002 at 9:30 a.m., local time, for the following purposes:
| 1. | To elect three directors for a three-year term ending in 2005. | |
| 2. | To ratify the selection of Ernst & Young LLP as the Company’s independent auditors. | |
| 3. | To act upon any other matter which may properly come before the Annual Meeting or any adjournment thereof. |
The Board of Directors of the Company has fixed the close of business on March 28, 2002 as the record date for determining stockholders of the Company entitled to notice of and to vote at the Annual Meeting. A list of the stockholders as of the record date will be available for inspection by stockholders at the Company’s corporate headquarters during business hours for a period of ten days prior to the Annual Meeting.
Your attention is directed to the attached Proxy Statement and the Annual Report of the Company for the fiscal year ended December 31, 2001.
| By Order of the Board of Directors, | |
| James H. Davis,Secretary |
Rockville, Maryland
EVEN IF YOU PLAN TO ATTEND THE MEETING, PLEASE PROMPTLY COMPLETE, SIGN, DATE AND RETURN THE ENCLOSED PROXY OR VOTE OVER THE INTERNET OR BY TELEPHONE. IF YOU ATTEND THE MEETING YOU MAY REVOKE YOUR PROXY AND VOTE IN PERSON.
Table of Contents
HUMAN GENOME SCIENCES, INC.
General
This Proxy Statement is being furnished to stockholders of Human Genome Sciences, Inc., a Delaware corporation (the “Company”), in connection with the solicitation by the Board of Directors of proxies for use at the Annual Meeting of Stockholders (the “Annual Meeting”) to be held at the University System of Maryland — Shady Grove Center, Germantown Room, 9630 Gudelsky Drive, Rockville, Maryland 20850 on Wednesday, May 22, 2002 at 9:30 a.m., local time, and at any adjournments thereof.
Solicitation
The solicitation is being made primarily by the use of the mails, but directors, officers and employees may also engage in the solicitation of proxies by telephone. The Company has retained the services of Georgeson Shareholder Communications, Inc. to assist it in soliciting proxies. Georgeson Shareholder Communications, Inc. will solicit proxies by personal interview, telephone, facsimile and mail. It is anticipated that the fee for those services will not exceed $7,000 plus reimbursement for out-of-pocket expenses. The cost of soliciting proxies will be borne by the Company. Other than the compensation of Georgeson Shareholder Communications, Inc., no compensation will be paid by the Company in connection with the solicitation of proxies, except that the Company may reimburse the brokers, custodians, nominees and other recordholders for their reasonable out-of-pocket expenses in forwarding proxy materials to the beneficial owners.
This Proxy Statement and the accompanying form of proxy are being sent to stockholders on or about April 19, 2002.
Revocation of Proxies
A proxy may be revoked at any time prior to its exercise by the filing of a written notice of revocation with the Secretary of the Company, by delivering to the Company a duly executed proxy bearing a later date or by attending the Annual Meeting and voting in person. However, if you are a stockholder whose shares are not registered in your own name, you will need documentation from your record holder stating your ownership as of March 28, 2002 in order to vote personally at the Annual Meeting.
Quorum and Voting Requirements
The close of business on March 28, 2002 has been fixed by the Board of Directors of the Company as the record date (the “Record Date”) for determining the stockholders of the Company entitled to notice of and to vote at the Annual Meeting. On the Record Date, there were 128,421,029 shares of the Company’s common stock, $0.01 par value per share (the “Common Stock”), outstanding. The presence at the Annual Meeting, in person or by a proxy relating to any matter to be acted upon at the meeting, of a majority of the outstanding shares, or 64,210,515 shares, is necessary to constitute a quorum for the Annual Meeting. Each outstanding share is entitled to one vote on all matters. For purposes of the quorum and the discussion below regarding the vote necessary to take stockholder action, stockholders of record who are present at the meeting in person or by proxy and who abstain, including brokers holding customers’ shares of record who cause abstentions to be recorded at the Annual Meeting, are considered stockholders who are present and entitled to vote and they count toward the quorum. In the event that there are not sufficient votes for a quorum or to approve any proposal at the Annual Meeting, the Annual Meeting may be adjourned in order to permit the further solicitation of proxies.
Table of Contents
Brokers holding shares of record for customers generally are not entitled to vote on certain matters unless they receive voting instructions from their customers. “Broker non-votes” mean the votes that could have been cast on the matter in question if the brokers had received their customers’ instructions, and as to which the broker has notified the Company on a proxy form in accordance with industry practice or has otherwise advised the Company that it lacks voting authority.
Directors are elected by a plurality and the three nominees who receive the most votes will be elected. Abstentions and broker non-votes will have no effect on the outcome of the election. On all other matters the affirmative vote of the majority of the shares present in person or by proxy at the meeting and entitled to vote on the matter is required to approve such matter. On such matters, broker non-votes are not considered shares entitled to vote on the matter and therefore will not be taken into account in determining the outcome of the vote on the matter. Abstentions are considered shares entitled to vote on the matter and therefore will have the effect of a vote against the matter.
All outstanding shares of the Company’s Common Stock represented by valid and unrevoked proxies received in time for the Annual Meeting will be voted. A stockholder may, with respect to the election of directors (1) vote for the election of the named director nominees, (2) withhold authority to vote for all such director nominees or (3) vote for the election of all such director nominees other than any nominee with respect to whom the stockholder withholds authority to vote by striking a line through such nominee’s name on the proxy. A stockholder may, with respect to each other matter specified in the notice of meeting (1) vote “FOR” the matter, (2) vote “AGAINST” the matter or (3) “ABSTAIN” from voting on the matter. Shares will be voted as instructed in the accompanying proxy on each matter submitted to stockholders. If no instructions are given, the shares will be voted FOR the election of the named director nominees and FOR the ratification of Ernst & Young LLP as the Company’s independent auditors.
The Board of Directors knows of no additional matters that will be presented for consideration at the Annual Meeting. Return of a valid proxy, however, confers on the designated proxyholders the discretionary authority to vote the shares in accordance with their best judgment on such other business, if any, that may properly come before the Annual Meeting or any adjournment thereof. Proxies solicited hereby will be tabulated by inspectors of election designated by the Board of Directors.
2
Table of Contents
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth information regarding the ownership of Common Stock of the Company as of March 28, 2002, unless otherwise indicated, by (1) all stockholders known by the Company to beneficially own more than five percent of the outstanding Common Stock, (2) each of the directors and nominees for director, (3) each executive officer of the Company, including those named in the Summary Compensation Table and (4) all directors and executive officers of the Company as a group.
| Number of | |||||||||
| Name and Address of Beneficial Owner(1) | Shares Owned | Percent Owned | |||||||
| FMR Corp. | 18,549,776 | (2) | 14.4 | % | |||||
| 82 Devonshire Street Boston, Massachusetts 02109-3614 | |||||||||
| Sid R. Bass and Lee M. Bass Group | 12,486,564 | (3) | 9.7 | % | |||||
| 201 Main Street Fort Worth, Texas 76201-3131 | |||||||||
| J.P. Morgan Chase & Co. | 7,899,070 | (4) | 6.2 | % | |||||
| 270 Park Avenue New York, New York 10017 | |||||||||
Directors, Nominees and Executive Officers | |||||||||
| William A. Haseltine, Ph.D. | 5,930,484 | (5) | 4.5 | % | |||||
| Ellen S. Baron, Ph.D. | 50 | (6) | * | ||||||
| Susan Bateson McKay | 160,313 | (7) | * | ||||||
| Richard J. Danzig | 13,499 | (8) | * | ||||||
| James H. Davis, Ph.D., J.D. | 727,189 | (9) | * | ||||||
| Jürgen Drews, M.D. | 59,999 | (10) | * | ||||||
| Richard C. Holbrooke | 11,999 | (11) | * | ||||||
| Robert D. Hormats | 50,999 | (12) | * | ||||||
| Argeris “Jerry” N. Karabelas, Ph.D. | — | * | |||||||
| Max Link, Ph.D. | 60,000 | (13) | * | ||||||
| Steven C. Mayer | 759,424 | (14) | * | ||||||
| Craig A. Rosen, Ph.D. | 1,419,583 | (15) | 1.1 | % | |||||
| Alan G. Spoon | 39,999 | (16) | * | ||||||
| David C. Stump, M.D. | 320,860 | (17) | * | ||||||
| Laura D’Andrea Tyson, Ph.D. | 55,999 | (18) | * | ||||||
| James B. Wyngaarden, M.D. | 75,999 | (19) | * | ||||||
| All directors and executive officers as a group (16 persons) | 9,686,396 | (20) | 7.2 | % | |||||
| * | Percentage is less than 1% of the total number of outstanding shares of the Company’s Common Stock. |
| (1) | Except as otherwise indicated, each party has sole voting and investment power over the shares beneficially owned. | |
| (2) | As reported on a Schedule 13G filed by FMR Corp. and certain of its affiliated entities on February 14, 2002. In some cases the entities have sole voting and investment power over their respective shares of the Company’s Common Stock; in other cases the entities share voting and/or investment power with other entities in the group. |
3
Table of Contents
| (3) | As reported on a Schedule 13G filed by Sid R. Bass and Lee M. Bass and 14 related and affiliated entities on February 14, 2002. In some cases the entities have sole voting and investment power over their respective shares of the Company’s Common Stock; in other cases the entities share voting and/or investment power with other entities in the group. | |
| (4) | As reported on a Schedule 13G filed by J.P. Morgan Chase & Co. and certain of its affiliated entities on February 14, 2002. In some cases the entities have sole voting and investment power over their respective shares of the Company’s Common Stock; in other cases the entities share voting and/or investment power or have no voting power. | |
| (5) | Includes 2,346,936 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 62,000 shares of Common Stock owned by Dr. Haseltine’s wife, as to which Dr. Haseltine disclaims beneficial ownership, and 1,663,064 shares of Common Stock issuable upon exercise of options held that are not exercisable within 60 days. | |
| (6) | Does not include 250,000 shares of Common Stock issuable upon exercise of options held that are not exercisable within 60 days. | |
| (7) | Includes 140,013 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 218,839 shares of Common Stock issuable upon exercise of options held that are not exercisable within 60 days. | |
| (8) | Includes 11,999 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 36,001 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. | |
| (9) | Includes 724,189 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 392,811 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. |
| (10) | Includes 59,999 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 44,001 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. |
| (11) | Includes 11,999 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 36,001 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. |
| (12) | Includes 44,999 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 24,001 shares of Common Stock issuable upon exercise of options held that are not exercisable within 60 days. |
| (13) | Includes 8,000 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. |
| (14) | Includes 671,092 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 380,476 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. |
| (15) | Includes 938,979 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Also includes 104,000 shares of Common Stock held in trust for Dr. Rosen’s minor children, as to which Dr. Rosen disclaims beneficial ownership. Does not include 790,961 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. |
| (16) | Includes 31,999 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 32,001 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. |
| (17) | Includes 316,284 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 519,140 shares of Common Stock issuable upon exercise of options that are not exercisable within 60 days. |
4
Table of Contents
| (18) | Includes 55,999 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 40,001 shares of Common Stock issuable upon exercise that are not exercisable within 60 days. |
| (19) | Includes 75,999 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 36,001 shares of Common Stock issuable upon exercise that are not exercisable within 60 days. |
| (20) | Includes 5,438,486 shares of Common Stock issuable upon exercise of options that are exercisable within 60 days. Does not include 4,463,298 shares issuable upon exercise of options that are not exercisable within 60 days. |
5
Table of Contents
PROPOSALS TO BE VOTED ON AT THE ANNUAL MEETING
PROPOSAL 1. ELECTION OF DIRECTORS WHOSE TERMS WILL EXPIRE IN 2005
Three directors, Drs. Link and Rosen and Amb. Holbrooke have a term of office expiring at the Annual Meeting, and at such time as their successors shall be elected and qualified. Drs. Link and Rosen and Amb. Holbrooke have each been nominated for a three-year term expiring at the annual meeting of stockholders in 2005 and until their successors shall be elected and qualified.
The persons named in the enclosed proxy intend to vote properly executed and returned proxies FOR the election of all nominees proposed by the Board of Directors unless authority to vote is withheld. In the event that any nominee is unable or unwilling to serve, the persons named in the proxy will vote for such substitute nominee or nominees as they, in their discretion, shall determine. The Board of Directors has no reason to believe that any nominee named herein will be unable or unwilling to serve.
Set forth below is information concerning the nominees for election and those directors whose term continues beyond the date of the Annual Meeting.
Nominees for Director for a Three-Year Term
| Director | Principal Occupation and Business | |||||||||
| Name | Age | Since | Experience During the Past Five Years | |||||||
| Max Link, Ph.D. | 61 | 1995 | Member of the Executive, Compensation and Nominating Committees of the Company. Dr. Link has held a number of executive positions with pharmaceutical and healthcare companies. Most recently he served as Chief Executive Officer of Corange Limited, from May 1993 until June 1994. Prior to joining Corange Limited, Dr. Link served in a number of positions within Sandoz Pharma Ltd., including Chief Executive Officer from 1987 until April 1992, and Chairman from April 1992 until May 1993. Dr. Link currently serves on the Boards of Directors of Access Pharmaceuticals, Inc.; Alexion Pharmaceuticals, Inc.; Cell Therapeutics, Inc.; Celsion Corporation; Columbia Laboratories Inc.; CytRx Corporation; Discovery Laboratories, Inc.; Osiris Therapeutics, Inc.; Protein Design Labs, Inc. and Sulzer Medica Ltd. Dr. Link received his doctorate in Economics from the University of St. Gallen. | |||||||
| Richard C. Holbrooke | 60 | 2001 | Member of the Ethics Committee of the Company. Served as U.S. Ambassador to the United Nations and as a member of the U.S. Cabinet from 1999 to early 2001. Served as Assistant Secretary of State for Europe from 1994 to 1996 and was the chief architect of the 1995 Dayton peace agreement that ended the war in Bosnia. Served as the President’s Special Envoy to Bosnia and Kosovo and as Special Envoy to Cyprus on a pro-bono basis while a private citizen. Served as the U.S. Ambassador to Germany from 1993 to 1994. Serves on the Boards of Directors of AIG and the National Endowment for Democracy and is Chairman of the International Rescue Committee and Refugees International. Founding Chairman of the American Academy, in Berlin, and Vice Chairman of Perseus, L.L.C. | |||||||
6
Table of Contents
| Director | Principal Occupation and Business | |||||||||
| Name | Age | Since | Experience During the Past Five Years | |||||||
| Craig A. Rosen, Ph.D. | 44 | 1992 | Executive Vice President of Research and Development and Director of the Company. Employed by the Roche Institute of Molecular Biology from 1987 to December 1992, serving as Chairman of the Department of Gene Regulation from 1991 to December 1992 and in varying positions in the Department of Molecular Oncology and Virology from 1987 to 1991. Member of the Board and Chairman of the Scientific Advisory Council for the American Foundation for AIDS Research from 1990 to 1995 and serves on the Scientific Advisory Boards for the Wistar Institute and the Institute for Human Virology. Dr. Rosen serves as a Director of Vascular Genetics, Inc. Author of approximately 130 publications and an editorial board member of several scientific publications. | |||||||
The Board of Directors recommends a vote FOR
7
Table of Contents
Directors Continuing in Office.
Directors whose term will expire at the 2003 annual meeting:
| Director | Principal Occupation and Business Experience | |||||||||
| Name | Age | Since | During the Past Five Years | |||||||
| William A. Haseltine, Ph.D. | 57 | 1993 | Chairman of the Board, Chief Executive Officer and member of the Executive and Nominating Committees of the Company. Consultant to the Company from December 1992 until May 1993. Chairman of the Board of Trustees of the National Health Museum, member of The Brookings Institution Board of Trustees and member of The Trilateral Commission. Member of the faculty of Harvard Medical School and the Dana Farber Cancer Institute from 1976 to May 1993. Member of the faculty of the Harvard School of Public Health from 1977 to May 1993 and Chief of Human Retrovirology at the Dana Farber Cancer Institute from 1988 to May 1993. A founder of several biotechnology companies. Author of approximately 250 scientific publications. | |||||||
| Laura D’Andrea Tyson, Ph.D. | 54 | 1998 | Member of the Audit and Ethics Committees of the Company. Dean of the London Business School since January 2002. Dean of the Walter A. Haas School of Business at the University of California, Berkeley from 1998 to 2001. Served as the President’s National Economic Advisor from 1995 to 1996 and as Chairman of the White House Council of Economic Advisors from 1993 to 1995. Dr. Tyson was a member of the National Bipartisan Commission on the Future of Medicare and a principal of the Law & Economics Consulting Group from June 1997 to December 2000. Dr. Tyson is a member of the Boards of Directors of SBC Communications Inc.; the Council on Foreign Relations; Eastman Kodak Company; Fox Entertainment Group, Inc.; Morgan Stanley and the Institute of International Economics. She also serves on the Board of Trustees of The Asia Foundation; the Advisory Boards of E.M. Warburg, Pincus & Co., LLP; E-piphany; the G7 Group, Inc.; H & Q Asia Pacific, Ltd. and the Board of Editors of The American Prospect. She is a Senior Director of Sebago Associates, Inc., an economic consulting firm, and an Economic Viewpoint columnist for Business Week magazine. | |||||||
8
Table of Contents
| Director | Principal Occupation and Business Experience | |||||||||
| Name | Age | Since | During the Past Five Years | |||||||
| Robert D. Hormats | 59 | 1996 | Member of the Executive, Audit and Compensation Committees of the Company. Vice Chairman of Goldman Sachs (International) since 1987. Served in various capacities with Goldman, Sachs & Co. since 1982. Served as a Senior Staff Member for International Economic Affairs on the National Security Council from 1974 to 1977. Served as Deputy Assistant Secretary for Economic and Business Affairs at the Department of State from 1977 to 1979, Ambassador and Deputy U.S. Trade Representative from 1979 to 1981 and Assistant Secretary of State for Economic and Business Affairs from 1981 to 1982. Appointed by former President Clinton in 1993 to the Board of the U.S. — Russia Investment Fund. | |||||||
| Alan G. Spoon | 50 | 1998 | Member of the Executive, Audit and Compensation Committees of the Company. Mr. Spoon is Managing General Partner at Polaris Venture Partners, a venture capital firm that he joined in May 2000. He was Chief Operating Officer and a Director of The Washington Post Company, a public company, from 1991 until his retirement in March 2000; he also served as President from May 1993 until his retirement. From 1989 to 1991, Mr. Spoon was President of Newsweek, Inc. During that time he was also responsible for Post-Newsweek television stations. From 1987 to 1989, he was The Washington Post Company’s Chief Financial Officer. Mr. Spoon presently serves as a Director of American Management Systems, Inc., Ticketmaster and Danaher Corporation, each of which is a public company. He is a Director of International Data Group (IDG), a private company, and serves on the boards of various Polaris portfolio companies. In addition, Mr. Spoon is a Regent of The Smithsonian Institution. | |||||||
9
Table of Contents
Directors whose term will expire at the 2004 annual meeting:
| Director | Principal Occupation and Business Experience | |||||||||
| Name | Age | Since | During the Past Five Years | |||||||
| Richard J. Danzig | 57 | 2001 | Member of the Nominating Committee of the Company. Chairman of the Board of the Center for Strategic and Budgetary Assessments. Served as Secretary of the Navy from 1998 to 2001 and as Under Secretary of the Navy from 1993 to 1997. Traveling Fellow of the Center for International Political Economy and an Adjunct Professor at Syracuse University’s Maxwell School of Citizenship & Public Affairs between 1997 and 1998. A partner in the law firm of Latham and Watkins from 1981 to 1993. Senior Fellow at the Center for Naval Analyses. Serves on the Boards of Directors of National Semiconductor Corporation, Public Agenda and the Partnership for Public Service. | |||||||
| Jürgen Drews, M.D. | 68 | 1998 | Member of the Executive and Nominating Committees of the Company. Chairman and partner of International Biomedicine Management Partners, Basel, Switzerland. Managing Partner of Bear Stearns Health Innoventures, LLC. Member of the Executive Committee of the Roche Group, Hoffman-La Roche, Inc., from 1986 until his retirement in 1998. Dr. Drews also served as President, Global Research for the Roche Group from 1996 until 1998. He was President, International Research and Development at the Roche Group from 1991 until 1996. Before joining Roche in 1985, Dr. Drews was Head of International Pharmaceutical Research and Development of Sandoz, Ltd. in Basel, Switzerland. Dr. Drews serves as Chairman of the Board of Directors of Genaissance Pharmaceuticals Inc. and is a Director of Protein Design Labs, Inc., of Mountain View, CA, and MorphoSys GmbH as well as Genomics Pharmaceutical Company, both in Munich, Germany. | |||||||
| Argeris “Jerry” N. Karabelas, Ph.D. | 49 | 2002 | Dr. Karabelas is Operating Partner of Care Capital LLC. Founder and Chairman of Novartis BioVenture Fund from 2000 to 2001; Head of Healthcare and CEO of Worldwide Pharmaceuticals at Novartis, AG from 1998 to 2000; Executive Vice-President, Pharmaceuticals at SmithKline Beecham from 1997 to 1998; President, North American Pharmaceuticals at SmithKline Beecham from 1993 to 1997; Vice President of U.S. Marketing, SmithKline Beecham, from 1990 to 1993. Dr. Karabelas is a visiting Committee Member of MIT Health Studies & Technology, a member of the Scientific Advisory Committee of Massachusetts General Hospital, a Director of Skye Pharma PLC, a Director of Nitromed, Inc., and a Trustee of the University of the Sciences of Philadelphia and the Fox Chase Cancer Center. | |||||||
10
Table of Contents
| Director | Principal Occupation and Business Experience | |||||||||
| Name | Age | Since | During the Past Five Years | |||||||
| James B. Wyngaarden, M.D. | 77 | 1993 | Member of the Ethics Committee of the Company. Founding Principal of the Washington Advisory Group, LLC.; Emeritus Professor of Medicine, Duke University; Professor of Medicine and Sr. Associate Dean, International Programs, University of Pennsylvania Medical Center, 1995-1997. Foreign Secretary, National Academy of Sciences and Institute of Medicine and Associate Vice Chancellor for Health Affairs at Duke University, 1990 to 1994. Served as Director of the National Institutes of Health from 1982 to 1989 and as Associate Director for Life Sciences, Office of Science and Technology Policy, Executive Office of the President from 1989 to 1990. Served as Director of Human Genome Organization from 1990 to 1991. Serves as Chairman of the Board of Directors of Hybridon, Inc., and as a Director of Genaera Corporation and Origenics Technologies. Author of approximately 250 scientific publications. | |||||||
Information Regarding the Board of Directors and Certain Committees
The Board of Directors held three regular meetings and one telephonic meeting during 2001. Richard Holbrooke attended fewer than 75% of the total number of meetings of the Board and of the Committees of which he was a member during 2001. Dr. Karabelas was appointed to the Board of Directors in April 2002 and thus attended no meetings of the Board during 2001. The Board of Directors has an Executive Committee, an Audit Committee, a Compensation Committee, an Ethics Committee and a Nominating Committee.
The Executive Committee, currently consisting of Drs. Haseltine, Link and Drews and Messrs. Spoon and Hormats, has the authority to act on most matters during the intervals between meetings of the Board of Directors. The Executive Committee held two regular meetings during 2001.
The Audit Committee, currently consisting of Messrs. Hormats and Spoon and Dr. Tyson, provides the opportunity for direct contact between the Company’s independent auditors and the Board of Directors. Each member of the Audit Committee is independent in accordance with the Nasdaq listing standards. The Audit Committee makes recommendations concerning the engagement of independent auditors, reviews with the independent auditors the plans and results of the audit engagement, reviews the adequacy of the Company’s internal accounting controls and oversees the Company’s financial reporting process. The Audit Committee held seven meetings during 2001.
The Compensation Committee, currently consisting of Dr. Link and Messrs. Hormats and Spoon, determines all compensation paid or awarded to the Company’s executive officers and senior officers (those with the rank of vice president or above) and administers the Company’s 2000 Stock Incentive Plan and Employee Stock Purchase Plan. The Compensation Committee held eight regular meetings and one telephonic meeting during 2001.
The Ethics Committee, currently consisting of Drs. Wyngaarden and Tyson and Amb. Holbrooke, is responsible for considering ethical issues raised by the Company’s research and development. The Ethics Committee did not meet during 2001.
The Nominating Committee, currently consisting of Drs. Haseltine, Link and Drews and Mr. Danzig, is responsible for proposing a slate of directors for election by the stockholders at each annual meeting and proposing candidates to fill any vacancies on the Board of Directors. The committee will consider nominees for Board membership recommended by stockholders. Any stockholder wishing to propose a
11
Table of Contents
Identification of Executive Officers
Set forth below is certain information regarding the positions and business experience of each executive officer of the Company who is not also a director of the Company.
| Executive Officer | Age | Positions | ||||
| Susan Bateson McKay | 47 | Ms. Bateson McKay has served as Senior Vice President, Human Resources, of the Company since December 2000 and served as Vice President, Human Resources, of the Company from January 1997 to December 2000. Prior to joining the Company, Ms. Bateson McKay served as Director of Human Resources and Administration at Finnegan, Henderson, Farabow, Garrett & Dunner, L.L.P., from May 1994 to December 1996. From 1983 to 1994, Ms. Bateson McKay was employed by J.P. Morgan & Co., Incorporated and was appointed Vice President, Human Resources, in 1985. Ms. Bateson McKay holds a masters degree in Business Administration from New York University and a bachelors degree, cum laude, in Economics from Mount Holyoke College. | ||||
| Ellen S. Baron, Ph.D. | 49 | Dr. Baron has served as Senior Vice President, Business Development, of the Company since August 2001. Prior to joining the Company, Dr. Baron had more than twenty years of experience in the pharmaceutical industry including expertise in translating basic science into commercial products. Dr. Baron served previously as Senior Director, Business Development/ Biotechnology, at Schering-Plough Corporation from 1993 to 2001. Previously, Dr. Baron spent twelve years in Research and Development at Schering-Plough in positions of increasing responsibility, including Director of Biotechnology Development, Fermentation Process Development and Cell Culture. Dr. Baron holds a doctorate degree in Microbiology from Georgetown University School of Medicine. | ||||
| James H. Davis, Ph.D., J.D. | 51 | Dr. Davis has served as Senior Vice President, General Counsel and Secretary of the Company since May 1997. From 1995 to 1997, Dr. Davis was Of Counsel to the Washington D.C. law firm of Finnegan, Henderson, Farabow, Garrett & Dunner, L.L.P. Prior to this time, Dr. Davis served in a number of capacities with an agricultural biotechnology company, Crop Genetics International, including General Counsel from 1988 to 1995, Vice President of Research and Development from 1990 to 1995, Secretary from 1990 to 1995 and a member of the Board of Directors from 1992 to 1995. Prior to joining Crop Genetics, Dr. Davis was a partner in the Washington, D.C. office of Weil, Gotshal & Manges. Dr. Davis holds a doctorate degree in Organic and Theoretical Chemistry from the California Institute of Technology and a law degree from the University of Virginia. | ||||
12
Table of Contents
| Executive Officer | Age | Positions | ||||
| Steven C. Mayer | 48 | Mr. Mayer has served as Senior Vice President and Chief Financial Officer of the Company since September 1996. From 1995 to 1996, Mr. Mayer was Vice President and Chief Financial Officer of GenVec, Inc., an early-stage gene therapy company. From 1991 to 1995, he served as Vice President (subsequently Senior Vice President) and Chief Financial Officer of TheraTech, Inc. Prior to joining TheraTech, Inc., Mr. Mayer was involved in the formation of Myriad Genetics and was Vice President and Chief Financial Officer of NPI, an agricultural biotechnology company. Mr. Mayer holds a masters degree in Business Administration from Stanford University. | ||||
| David C. Stump, M.D. | 52 | Dr. Stump has served as Senior Vice President, Drug Development of the Company since November 1999. From October 1995 to October 1999, he served as Vice President, Clinical Research and Genentech Fellow at Genentech, Inc. Dr. Stump first joined Genentech in 1989 as Director, Clinical Research and leader of its thrombolytic therapy drug development program. Prior to joining Genentech he was Associate Professor of Medicine and Biochemistry at the University of Vermont. He received his medical degree at Indiana University and his postgraduate training at the University of Iowa as well as the University of Leuven, Belgium. He is board certified in Internal Medicine, Hematology and Medical Oncology and is a Fellow of the American Colleges of Physicians and the Council on Arteriosclerosis, Thrombosis and Vascular Biology of the American Heart Association. He is the author of approximately 60 scientific publications. | ||||
13
Table of Contents
PROPOSAL 2 — RATIFICATION OF INDEPENDENT AUDITORS
The Board of Directors has selected the firm of Ernst & Young LLP to serve as independent auditors for the fiscal year ending December 31, 2002, subject to the ratification of such appointment by the stockholders. A representative of Ernst & Young LLP is expected to be present at the Annual Meeting and is expected to be available to respond to appropriate questions from stockholders. Ernst & Young LLP currently serves as the Company’s independent auditors.
Unless marked to the contrary, the shares represented by the enclosed proxy, if properly executed and returned, will be voted FOR the ratification of the appointment of Ernst & Young LLP as the independent auditors of the Company for the fiscal year ending December 31, 2002.
The Board of Directors recommends a vote FOR ratification of Ernst & Young LLP.
Audit Fees
For the year ended December 31, 2001, the Company incurred from Ernst & Young LLP an aggregate of $104,000 for audit fees, covering professional services rendered for (1) the audit of the Company’s annual financial statements included in the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2001 and (2) the reviews of the financial statements included in the Company’s quarterly reports on Form 10-Q for the first three quarters of 2001.
Financial Information Systems Design and Implementation Fees
Ernst & Young LLP did not perform any financial information systems design, implementation or related services for the Company during the year ended December 31, 2001.
All Other Fees
For the year ended December 31, 2001, Ernst & Young LLP billed the Company an aggregate of $135,297 for all other services, including $91,349 for non-audit accounting services and $43,948 for tax related services.
Audit Committee Report
The Audit Committee of the Board of Directors consists of three independent directors, as required by Nasdaq listing standards. The Audit Committee operates under a written charter adopted by the Board of Directors, and is responsible for overseeing the Company’s financial reporting process on behalf of the Board of Directors. The members of the Audit Committee are Messrs. Hormats and Spoon and Dr. Tyson. Each year, the Audit Committee recommends to the Board of Directors, subject to stockholder ratification, the selection of the Company’s independent auditors.
Management is responsible for the Company’s financial statements and the financial reporting process, including internal controls. The independent auditors are responsible for performing an independent audit of the Company’s consolidated financial statements in accordance with auditing standards generally accepted in the United States and for issuing a report thereon. The Audit Committee’s responsibility is to monitor and oversee these processes.
In this context, the Audit Committee has met and held discussions with management and Ernst & Young LLP, the Company’s independent auditors. Management represented to the Audit Committee that the Company’s consolidated financial statements were prepared in accordance with accounting principles generally accepted in the United States, and the Audit Committee has reviewed and discussed the consolidated financial statements with management and the independent auditors. The Audit Committee discussed with Ernst & Young LLP the matters required to be discussed by Statement on Auditing Standards No. 61 (Communication with Audit Committees). These matters included a discussion of Ernst & Young’s judgments about the quality (not just the acceptability) of the Company’s accounting principles as applied to financial reporting.
14
Table of Contents
Ernst & Young also provided the Audit Committee with the written disclosures and letter required by Independence Standards Board Standard No. 1 (Independence Discussions with Audit Committees), and the Audit Committee discussed with Ernst & Young LLP that firm’s independence. The Audit Committee further considered whether the provision by Ernst & Young LLP of the non-audit services described above is compatible with maintaining the auditors’ independence.
Based upon the Audit Committee’s discussion with management and the independent auditors and the Audit Committee’s review of the representation of management and the disclosures by the independent auditors to the Audit Committee, the Audit Committee recommended to the Board of Directors that the Company’s audited consolidated financial statements be included in the Company’s Annual Report on Form 10-K for the year ended December 31, 2001, for filing with the Securities and Exchange Commission. The Audit Committee and the Board of Directors have also recommended the selection of Ernst & Young LLP as the Company’s independent auditors for 2002, subject to stockholder ratification.
| Audit Committee | |
| Robert D. Hormats, Chairman | |
| Alan G. Spoon | |
| Laura D’Andrea Tyson, Ph.D. |
15
Table of Contents
EXECUTIVE COMPENSATION AND OTHER MATTERS
Compensation of Directors
In 2001, each director who was not an employee of the Company was eligible to receive a director’s fee of $25,000 per year and a fee of $1,000 for participation in each meeting of a committee of the Board of Directors. Other directors received no compensation for their services to the Company as directors. Beginning in 2000, each non-employee director was also entitled to receive an automatic grant of options to purchase 48,000 shares of Common Stock on the date that such non-employee director was first elected or appointed or re-elected as a director. In each year before a non-employee director receives the automatic grant noted above, such non-employee director will receive an annual automatic grant of an option to purchase 8,000 shares of Common Stock. All directors are reimbursed for expenses incurred in connection with attending meetings of the Board of Directors.
Executive Compensation
The employees named in the following table were the Company’s Chief Executive Officer and the four highest-paid executive officers during 2001 (the “Named Executive Officers”).
SUMMARY COMPENSATION TABLE
| Long-Term | |||||||||||||||||||||||||||||
| Annual Compensation | Compensation Awards | ||||||||||||||||||||||||||||
| Other Annual | Restricted Stock | All Other | |||||||||||||||||||||||||||
| Salary(1) | Bonus | Compensation(2) | Awards(3) | Options(4) | Compensation(5) | ||||||||||||||||||||||||
| Name and Principal Position | Year | ($) | ($) | ($) | ($) | (#) | ($) | ||||||||||||||||||||||
| William A. Haseltine, Ph.D. | 2001 | 483,654 | 210,000 | 63,824 | 520,000 | 2,625 | |||||||||||||||||||||||
| Chief Executive Officer | 2000 | 448,077 | 200,000 | 82,615 | 650,000 | 2,625 | |||||||||||||||||||||||
| 1999 | 398,654 | 175,000 | 82,845 | 520,000 | 2,500 | ||||||||||||||||||||||||
| Craig A. Rosen, Ph.D. | 2001 | 364,038 | 200,000 | 280,000 | 2,625 | ||||||||||||||||||||||||
| Executive Vice President | 2000 | 338,846 | 170,000 | 350,000 | 2,493 | ||||||||||||||||||||||||
| 1999 | 309,115 | 160,000 | 300,000 | 2,493 | |||||||||||||||||||||||||
| Steven C. Mayer | 2001 | 294,231 | 135,000 | 140,000 | 2,573 | ||||||||||||||||||||||||
| Senior Vice President | 2000 | 273,654 | 105,000 | 200,000 | 2,573 | ||||||||||||||||||||||||
| 1999 | 238,808 | 165,000 | 120,000 | 2,500 | |||||||||||||||||||||||||
| James H. Davis, Ph.D., J.D. | 2001 | 294,231 | 125,000 | 140,000 | 2,625 | ||||||||||||||||||||||||
| Senior Vice President | 2000 | 273,654 | 105,000 | 200,000 | 2,625 | ||||||||||||||||||||||||
| 1999 | 239,000 | 85,000 | 160,000 | 2,500 | |||||||||||||||||||||||||
| David C. Stump, M.D.(6) | 2001 | 294,231 | 110,000 | 140,000 | 2,625 | ||||||||||||||||||||||||
| Senior Vice President | 2000 | 275,000 | 105,000 | 81,530 | 200,000 | 2,625 | |||||||||||||||||||||||
| 1999 | 37,019 | 50,000 | 500,000 | ||||||||||||||||||||||||||
| (1) | Includes amounts earned but deferred at the election of the Named Executive Officer, such as salary deferrals under the Company’s 401(k) Plan established under Section 401(k) of the Internal Revenue Code. |
| (2) | As permitted by rules promulgated by the Securities and Exchange Commission, no amounts are shown with respect to certain perquisites (such as car and housing allowances), where such amounts do not exceed the lesser of (i) 10% of the sum of the salary and bonus of the Named Executive Officer, or (ii) $50,000. Amounts reported for Dr. Haseltine include $36,890, $55,680 and $44,101 in imputed interest for 2001, 2000 and 1999, respectively, on an interest-free loan (See “—Loan to Executive Officer”). Also included in the amounts reported for Dr. Haseltine are $26,934, $26,934 and $22,143 for car allowances for 2001, 2000 and 1999, respectively. The amount reported for Dr. Stump for 2000 represents relocation expenses for which he was reimbursed, as well as gross-up for income tax purposes on these expenses. |
16
Table of Contents
| (3) | No cash dividends were paid on stock or restricted stock for the fiscal years ended December 31, 2001, 2000 and 1999. |
| (4) | The Company has awarded no Stock Appreciation Rights. Share amounts for all years presented have been restated to reflect two two-for-one stock splits paid in the form of a stock dividend on January 28, 2000 and October 5, 2000. |
| (5) | Except as otherwise indicated, the amounts reported represent the Company’s contributions to the Named Executive Officer’s account under its 401(k) Plan. |
| (6) | Dr. Stump joined the Company in November 1999. |
The following table sets forth information concerning grants to the Named Executive Officers of options to purchase shares of the Company’s Common Stock granted in 2001.
OPTIONS GRANTED IN 2001
| Potential realizable value | ||||||||||||||||||||||||
| Individual Grants | at assumed annual rates | |||||||||||||||||||||||
| of stock price | ||||||||||||||||||||||||
| Percent of total | appreciation for option | |||||||||||||||||||||||
| options granted to | Exercise price | term(1) | ||||||||||||||||||||||
| Options | all employees in | per share | Expiration | |||||||||||||||||||||
| Name | (#) | fiscal year | ($) | date | 5%($) | 10%($) | ||||||||||||||||||
| William A. Haseltine, Ph.D. | 520,000 | 7.7 | % | 38.63 | 12/05/2011 | 12,632,984 | 32,014,461 | |||||||||||||||||
| Craig A. Rosen, Ph.D. | 280,000 | 4.1 | % | 38.63 | 12/05/2001 | 6,802,376 | 17,238,556 | |||||||||||||||||
| Steven C. Mayer | 140,000 | 2.1 | % | 38.63 | 12/05/2011 | 3,401,188 | 8,619,278 | |||||||||||||||||
| James H. Davis, Ph.D., J.D. | 140,000 | 2.1 | % | 38.63 | 12/05/2011 | 3,401,188 | 8,619,278 | |||||||||||||||||
| David C. Stump, M.D. | 140,000 | 2.1 | % | 38.63 | 12/05/2011 | 3,401,188 | 8,619,278 | |||||||||||||||||
| (1) | The assumed annual rates of stock price appreciation of 5% and 10% are required by the Securities and Exchange Commission to be used for illustration purposes and are not intended to forecast possible future appreciation, if any, of the Company’s Common Stock. |
The following table sets forth information with respect to option exercises by and year-end values during 2001 for the Named Executive Officers.
AGGREGATED OPTION EXERCISES IN LAST FISCAL YEAR
| Number of unexercised | Value of unexercised in-the- | |||||||||||||||||||||||
| options at | money options at fiscal | |||||||||||||||||||||||
| Shares | fiscal year-end(#) | year-end($)(1) | ||||||||||||||||||||||
| acquired on | Value | |||||||||||||||||||||||
| Name | exercise(#) | Realized($) | Exercisable | Unexercisable | Exercisable | Unexercisable | ||||||||||||||||||
| William A. Haseltine, Ph.D. | 600,000 | 26,772,060 | 1,834,547 | 2,175,453 | 26,155,754 | 17,349,046 | ||||||||||||||||||
| Craig A. Rosen, Ph.D. | 130,060 | 6,100,854 | 824,534 | 905,406 | 16,813,312 | 5,441,945 | ||||||||||||||||||
| Steven C. Mayer | 44,608 | 1,272,837 | 620,259 | 431,309 | 14,147,347 | 2,110,682 | ||||||||||||||||||
| James H. Davis, Ph.D., J.D. | 0 | 0 | 620,352 | 496,648 | 13,789,347 | 3,326,000 | ||||||||||||||||||
| David C. Stump, M.D. | 0 | 0 | 253,775 | 581,649 | 2,420,095 | 3,463,685 | ||||||||||||||||||
| (1) | Value is based on the difference between the stock option exercise price and the closing price of the Company’s Common Stock on The Nasdaq National Market on December 31, 2001 of $33.72 per share. |
Employment Agreements, Termination of Employment and Change in Control Arrangements
In February 1997, the Company entered into an employment agreement with Dr. Haseltine that superseded his May 1993 employment agreement. The employment agreement is for an initial term that
17
Table of Contents
In October 1992, the Company entered into an employment agreement with Dr. Rosen in which Dr. Rosen agreed to serve as Senior Vice President, Research and Development of the Company. Since 2000, he has served as Executive Vice President, Research and Development. The employment agreement was for an initial term that ended in October 1995 and has been and will be automatically extended for additional one-year periods unless terminated by either party prior to the end of the applicable term. Dr. Rosen is entitled to an annual base salary, as determined by the Board of Directors ($390,000 as of January 1, 2002), an annual bonus as determined by the Board of Directors, and an annual car allowance. The Company may terminate the employment agreement without cause, and upon such termination, Dr. Rosen will be entitled to receive up to six months’ base salary, but in no event less than three months’ base salary. In April 1993, pursuant to the agreement, the Company sold to Dr. Rosen 563,908 shares of restricted Common Stock at a purchase price of $0.0025 per share. All of the shares are subject to rights of first refusal by the Company on sale (at a price per share equal to $0.0475 less than the price of the proposed sale).
In August 1996, the Company entered into an employment agreement with Mr. Mayer in which Mr. Mayer agreed to serve as Senior Vice President and Chief Financial Officer of the Company. Mr. Mayer is entitled to an annual base salary, as determined by the Board of Directors ($315,000 as of January 1, 2002) and an annual bonus as determined by the Board of Directors. Upon termination without cause, Mr. Mayer will be entitled to receive base salary until the earlier of (1) 12 months after termination of employment or (2) commencement of other regular full-time employment.
In April 1997, the Company entered into an employment agreement with Dr. Davis in which Dr. Davis agreed to serve as Senior Vice President, General Counsel and Secretary of the Company. Dr. Davis is entitled to an annual base salary, as determined by the Board of Directors ($315,000 as of January 1, 2002) and an annual bonus as determined by the Board of Directors. Upon termination without cause, Dr. Davis will be entitled to receive base salary until the earlier of (1) 12 months after termination of employment or (2) commencement of other regular full-time employment.
In September 1999, the Company entered into an employment agreement with Dr. Stump in which Dr. Stump agreed to serve as Senior Vice President, Drug Development of the Company, effective November 1999. Dr. Stump is entitled to an annual base salary, as determined by the Board of Directors ($315,000 as of January 1, 2002) and an annual bonus as determined by the Board of Directors. Upon termination without cause, Dr. Stump will be entitled to receive base salary until the earlier of (1) 12 months after termination of employment or (2) commencement of other regular full-time employment.
In August 2001, the Company entered into an employment agreement with Dr. Baron in which Dr. Baron agreed to serve as Senior Vice President, Business Development of the Company. Dr. Baron is entitled to an annual base salary, as determined by the Board of Directors ($280,000 as of January 1, 2002) and an annual bonus as determined by the Board of Directors. Upon termination without cause, Dr. Baron will be entitled to receive base salary until the earlier of (1) 12 months after termination of employment or (2) commencement of other regular full-time employment.
18
Table of Contents
In July 1998, the Company established a Key Executive Severance Plan for the Chief Executive Officer, the President and other key employees of the Company, and pursuant to that plan, the Company entered into agreements with the Named Executive Officers. The agreements provide that in the event the executive’s employment is terminated by the Company without cause or by the executive for good reason, in either case within 18 months of a Change in Control of the Company (as defined in the Key Executive Severance Plan), the Company shall make a cash payment to the executive equal to 1.5 times the sum of the executive’s annual salary plus bonus (2.0 times in the case of the Chief Executive Officer) and the executive will be entitled to continue to participate in the Company’s group medical, dental, life and disability programs for a period of eighteen months (twenty-four months in the case of the Chief Executive Officer). In addition, the Key Executive Severance Plan provides that upon a Change in Control, all option plans will vest unless the options are assumed or replaced in connection with the Change in Control and the assumed or replacement options will vest in the event the executive’s employment is terminated without cause or the executive resigns for good reason, in either case within 18 months of the Change in Control. Each executive also agreed to certain confidentiality and non-solicitation provisions as a condition to participation in the Key Executive Severance Plan.
Loan to Executive Officer
In November 1993, the Board of Directors authorized the extension of an interest-free loan (the “Loan”) to Dr. Haseltine, in an amount up to $1,000,000, to enable Dr. Haseltine to pay certain tax liabilities relating to his purchase of Common Stock in May 1993. The Loan was made on December 29, 1993 and was evidenced by a promissory note dated March 4, 1994 in the original amount of $872,845, replacing a promissory note dated December 29, 1993 (as replaced, the “Note”). The principal amount of the Loan was increased to $891,488 on December 16, 1994. The Loan was secured initially by 457,888 shares of issued and outstanding Common Stock of the Company, with a market value at that time equal to 200% of the principal sum of the loan (the “Collateral”) and is now secured by 128,504 shares of Common Stock. The Loan is repayable in full on the earliest to occur of (1) twenty business days after demand for repayment has been made by the Company, (2) twenty business days after Dr. Haseltine’s employment has been terminated, (3) on the date Dr. Haseltine defaults on the Note, or (4) December 29, 2003. Dr. Haseltine is required to repay the Loan in full, or in some instances, in part, if he sells any of the Collateral.
Compensation Committee Interlocks and Insider Participation
None of the members of the Compensation Committee is a current or former officer or employee of the Company.
Compensation Committee Report on Executive Compensation
The Compensation Committee of the Board of Directors consists entirely of non-employee directors. The Compensation Committee is responsible for setting and administering the policies that govern annual executive salaries, bonuses and stock ownership programs. The Committee annually evaluates the performance, and determines the compensation, of the Chief Executive Officer and the other executive officers of Human Genome Sciences based upon a mix of the achievement of the corporate goals, individual performance and comparisons with other pharmaceutical and biotechnology companies.
The Compensation Committee’s goals with respect to executive officers, including the CEO, are to provide compensation sufficient to attract, motivate and retain executives of outstanding ability and potential, and to establish and maintain an appropriate relationship between executive compensation and the creation of shareholder value. When determining adjustments to an individual’s compensation package, the Compensation Committee evaluates the importance to stockholders of that person’s continued service.
The Compensation Committee retains a leading executive compensation consultant to provide industry-specific competitive intelligence and advice about executive compensation program design and competitive compensation levels.
19
Table of Contents
The executive officers’ compensation structure consists of: (1) base salary, (2) cash bonus and (3) stock options.
Base Salary.Salaries for 2001 were set based on the above factors and after review of industry comparables and discussion with a leading compensation consultant.
The Company’s philosophy is to maintain executive base salary at a competitive level sufficient to recruit individuals possessing the skills and values necessary to achieve the Company’s vision and mission over the long term. Each individual’s base salary is determined by the Compensation Committee after considering a variety of factors that make up market value and prospective value to the Company, including the knowledge, experience and accomplishments of the individual, the individual’s level of responsibility, and the typical compensation levels for individuals with similar credentials. The Compensation Committee may, considering the advice of Company management, change the salary of an individual on the basis of its judgment for any reason, including the performance of the individual or the Company, changes in responsibility, and changes in the market for executives with similar credentials. Determinations of appropriate base salary levels and other compensation elements are generally made through consideration of a variety of industry surveys and studies, as well as by monitoring developments in relevant industries such as the pharmaceutical and biotechnology industries.
Cash Bonus.Bonuses are awarded for accomplishments during the past year. Bonuses are determined by the Compensation Committee with advice from Company management, based upon the Committee’s assessment of the individual’s contributions during the year, compared to, but not limited to, a list of individualized goals previously approved by management and the Compensation Committee. In determining bonuses for 2001, the Compensation Committee considered in addition to the individualized goals, the Company’s successful integration of Principia’s technology and assets, completion of an antibody manufacturing facility, design and initial site development for a large-scale manufacturing plant, progress in conducting clinical trials of new drug candidates and in identifying new therapeutic proteins for internal development or out-licensing, and continued progress within collaborations previously established in the area of antibodies.
Stock Options.Stock options are a fundamental element in the HGS executive compensation program because they emphasize long-term Company performance, as measured by creation of stockholder value, and foster a commonality of interest between stockholders and employees. Options are granted to all regular full-time employees, and particularly to key employees likely to contribute significantly to the Company. In determining the size of an option grant to an executive officer, the Compensation Committee considers competitive factors, changes in responsibility, and the executive officer’s achievement of individual pre-established goals. In addition, the Company usually makes a significant grant of options when an executive officer joins the Company. Options are granted at 100% of the fair market value on the date of grant. The Company generally awards options to officers upon the commencement of employment and at regular intervals, but other awards may be made as well. Some of the Company’s stock option plans also provide for option grants to members of the Board. Options granted to employees generally vest over periods ranging from two to five years after grant.
Executive officers other than the Chief Executive Officer received options for 1,050,000 shares in 2001.
Chief Executive Officer’s Compensation.The Compensation Committee awarded Dr. Haseltine a bonus of $210,000 for 2001. In addition, effective January 1, 2002, Dr. Haseltine’s base salary was increased from $485,000 per year to $500,000 per year. The bonus and increase in salary are based on the Compensation Committee’s assessments of Dr. Haseltine’s role in the Company’s performance in 2001. Under Dr. Haseltine’s leadership, the Company’s capabilities and opportunities were significantly enhanced by completion of an antibody manufacturing facility, design and initial site development for a large-scale manufacturing plant, the Company’s successful integration of Principia’s technology and assets, and additional integration and construction at our research campus. At the same time, the Company has made significant progress in developing new drug candidates. Organizational development is keeping pace with the expanding opportunities.
20
Table of Contents
Compensation Deduction Limit.The Compensation Committee has considered the $1 million limit for federal income tax purposes on deductible executive compensation that is not performance-based, and believes all executive compensation expenses will be deductible by the Company for the foreseeable future.
| Compensation Committee | |
| Max Link, Ph.D., Chairman | |
| Robert D. Hormats | |
| Alan G. Spoon |
Performance Graph
As part of the proxy statement disclosure requirements mandated by the Securities and Exchange Commission, the Company is required to provide a comparison of the cumulative total stockholder return on its Common Stock with that of a broad equity market index and either a published industry index or a company-constructed peer group index. The following graph compares the performance of the Company’s Common Stock for the periods indicated with the performance of the Nasdaq U.S. Stock Market Total Return Index (the “TRI”) and the Nasdaq Pharmaceutical Index (the “NPI”). The comparison assumes $100 was invested on December 31, 1996 in the Company’s Common Stock and in each of the foregoing indices and assumes the reinvestment of dividends.
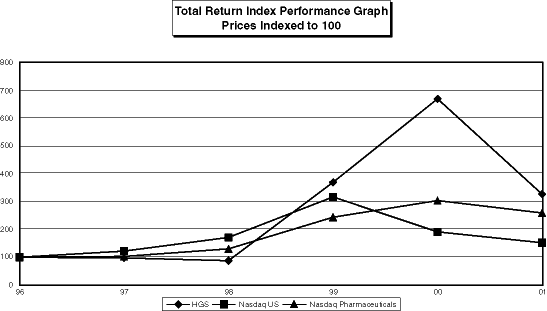
[LINE GRAPH]
| HGS | Nasdaq US | Nasdaq Pharmaceuticals | ||||||||||
| 1996 | 100.00 | 100.00 | 100.00 | |||||||||
| 1997 | 97.546 | 122.477 | 103.05 | |||||||||
| 1998 | 87.2699 | 172.68 | 130.807 | |||||||||
| 1999 | 374.54 | 320.832 | 246.642 | |||||||||
| 2000 | 680.368 | 192.977 | 307.651 | |||||||||
| 2001 | 330.994 | 153.121 | 262.189 | |||||||||
ADDITIONAL INFORMATION
Compliance with Section 16(a) of the Exchange Act
Section 16(a) of the Exchange Act requires that the Company’s executive officers and directors, and persons who own more than ten percent of a registered class of the Company’s equity securities, file reports of ownership and changes in ownership with the SEC and provide the Company with copies of such reports. The Company has reviewed such reports received by it and written representations from its directors and executive officers. Based solely on such review, the Company believes that all ownership filing requirements were timely during 2001. Mr. Mayer filed an amended Form 5 in 2001 reporting an inadvertent omission of a gift of shares to his children in 2000.
21
Table of Contents
Other Matters
The Board of Directors of the Company knows of no other business which will be presented for consideration at the Annual Meeting. Return of a valid proxy, however, confers on the designated proxyholders discretionary authority to vote the shares in accordance with their best judgment on such other business, if any, that may properly come before the Annual Meeting or any adjournments thereof.
THE COMPANY WILL PROVIDE WITHOUT CHARGE A COPY OF THE COMPANY’S ANNUAL REPORT ON FORM 10-K FOR THE FISCAL YEAR ENDED DECEMBER 31, 2001, INCLUDING FINANCIAL STATEMENTS AND SCHEDULES, TO EACH OF THE COMPANY’S STOCKHOLDERS OF RECORD ON MARCH 28, 2002, AND TO EACH BENEFICIAL OWNER OF STOCK ON THAT DATE, UPON RECEIPT OF A WRITTEN REQUEST THEREFOR MAILED TO THE COMPANY’S OFFICES, 9410 KEY WEST AVENUE, ROCKVILLE, MARYLAND 20850-3338, ATTENTION: INVESTOR RELATIONS OFFICE. IN THE EVENT THAT EXHIBITS TO SUCH FORM 10-K ARE REQUESTED, A FEE WILL BE CHARGED FOR REPRODUCTION OF SUCH EXHIBITS. REQUESTS FROM BENEFICIAL OWNERS OF COMMON STOCK MUST SET FORTH A GOOD FAITH REPRESENTATION AS TO SUCH OWNERSHIP.
Proposals for the 2003 Annual Meeting
The deadline for submission of stockholder proposals to be considered for inclusion in the proxy statement and form of proxy relating to the 2003 annual meeting of stockholders is December 26, 2002. Any such proposal received by the Company’s principal executive offices in Rockville, Maryland after such date will be considered untimely and may be excluded from the proxy statement and form of proxy.
The deadline for submission of stockholder proposals to be presented at the 2003 annual meeting of stockholders, but which will not be included in the proxy statement and form of proxy relating to such meeting, is March 25, 2003. Any such proposal received by the Company’s principal executive offices in Rockville, Maryland after such date may be considered untimely and excluded. If such proposal is presented at the 2003 annual meeting of stockholders, the persons named in the proxy for such meeting may exercise their discretionary voting power with respect to such proposal.
| By Order of the Board of Directors, | |
| James H. Davis,Secretary |
April 19, 2002
THE BOARD OF DIRECTORS HOPES THAT YOU WILL ATTEND THE MEETING. WHETHER OR NOT YOU PLAN TO ATTEND, PLEASE PROMPTLY COMPLETE, DATE, SIGN AND RETURN THE ENCLOSED PROXY OR VOTE OVER THE INTERNET OR BY TELEPHONE. IF YOU ATTEND THE MEETING, YOU MAY WITHDRAW YOUR PROXY AND VOTE YOUR OWN SHARES.
22
Table of Contents
Annual Meeting of Stockholders
HUMAN GENOME SCIENCES, INC.
May 22, 2002
Please date, sign and mail your proxy card back
as soon as possible or submit your proxy by telephone or
Internet. If you submit your proxy by telephone or Internet,
do not return your proxy card.
HUMAN GENOME SCIENCES, INC.
PROXY SOLICITED ON BEHALF OF THE BOARD OF DIRECTORS FOR THE
ANNUAL MEETING OF STOCKHOLDERS TO BE HELD ON MAY 22, 2002
The undersigned hereby appoints WILLIAM A. HASELTINE, Ph.D. and JAMES H. DAVIS, Ph.D., J.D., and each of them, with full power of substitution to each, as attorneys and proxies of the undersigned, to vote all shares which the undersigned is entitled to vote at the Annual Meeting of Stockholders of Human Genome Sciences, Inc. (the “Company”) to be held at the University System of Maryland, Shady Grove Center, Germantown Room, 9630 Gudelsky Drive, Rockville, MD 20850, on May 22, 2002 at 9:30 a.m., local time, and at any adjournments thereof, upon and in respect of the following matters, and in accordance with the following instructions, with discretionary authority as to any and all other matters that may properly come before the meeting.
The undersigned hereby acknowledges receipt of a copy of the Company’s 2001 Annual Report and Notice of Annual Meeting and Proxy Statement relating to such Annual Meeting. The undersigned revokes all proxies heretofore given for said Annual Meeting and any adjournment(s) thereof.
THIS PROXY IS SOLICITED ON BEHALF OF THE BOARD OF DIRECTORS. IT MAY BE REVOKED AT ANY TIME PRIOR TO ITS EXERCISE BY SENDING WRITTEN NOTICE TO THE SECRETARY OF THE COMPANY, BY DELIVERING TO THE COMPANY A DULY EXECUTED PROXY BEARING A LATER DATE OR BY ATTENDING THE ANNUAL MEETING AND VOTING IN PERSON.
THIS PROXY WILL BE VOTED IN THE MANNER DIRECTED HEREIN BY THE PERSON(S) SIGNING IT. IF NO DIRECTION IS MADE, THIS PROXY WILL BE VOTED “FOR” THE ELECTION OF THE NOMINEES INDICATED AND “FOR” THE OTHER PROPOSAL.
(Continued on other side)
Table of Contents
| [HUMAN GENOME SCIENCES LOGO] | ||
| C/O AMERICAN STOCK TRANSFER & TRUST COMPANY 59 MAIDEN LANE NEW YORK, NY 10038 | VOTE BY INTERNET — www.proxyvote.com Use the Internet to transmit your voting instructions and for electronic delivery of information until 11:59 P.M. Eastern Time the day before the meeting date. Have your proxy card in hand when you access the web site. You will be prompted to enter your 12-digit Control Number which is located below to obtain your records and to create an electronic voting instruction form. | |
| VOTE BY PHONE — 1-800-690-6903 Use any touch-tone telephone to transmit your voting instructions until 11:59 P.M. Eastern Time the day before the meeting date. Have your proxy card in hand when you call. You will be prompted to enter your 12-digit Control Number which is located below and then follow the simple instructions the Vote Voice provides to you. | ||
| VOTE BY MAIL Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Human Genome Sciences, Inc., c/o ADP, 51 Mercedes Way, Edgewood, NY 11717. |
| TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: HMNGS1 KEEP THIS PORTION FOR YOUR RECORDS |
| DETACH AND RETURN THIS PORTION ONLY |
| THIS PROXY CARD IS VALID ONLY WHEN SIGNED AND DATED |
| HUMAN GENOME SCIENCES, INC. | ||||||||
| MANAGEMENT RECOMMENDS A VOTE “FOR” THE NOMINEES FOR DIRECTOR LISTED BELOW AND “FOR” PROPOSAL 2. 1. To elect 3 directors. | For | Withhold | For All | To withhold authority to vote for any nominee(s) mark “For All Except” and write the nominee’s number on the line below. | ||||
| Nominees: 01) Max Link, Ph.D. | All | All | Except | |||||
| 02) Ambassador Richard C. Holbrooke | [ ] | [ ] | [ ] | |||||
| 03) Craig A. Rosen, Ph.D. |
| For | Against | Abstain | ||||
| 2. To ratify the appointment of Ernst & Young LLP as the Company’s independent auditors for the fiscal year ending December 31, 2002. | [ ] | [ ] | [ ] | |||
| 3. To act upon such other matters which may properly come before the Annual Meeting or any adjournment(s) thereof. | ||||||
| This proxy, when properly executed, will be voted in the manner directed herein by the stockholder. If no direction is made, this proxy will be voted “FOR” the election of the nominees and “FOR” the appointment of Ernst & Young LLP. | ||||||
| Please sign, date and promptly return this proxy in the enclosed envelope. No postage is required if mailed within the United States. (If you submit your proxy by telephone or Internet, do not return your proxy card.) | ||||||
| Note: Please sign exactly as your name appears hereon. If the shares are in the names of two or more persons, each should sign. Executors, administrators, trustees, guardians and attorneys-in-fact should give their full titles. If a signatory is a corporation, please give the full corporate name and have a duly authorized officer sign, stating his or her title. If a signatory is a partnership, please sign in partnership name by an authorized person. | ||||||
| If you plan to attend the meeting, please check the box to the right. [ ] |
| Signature (PLEASE SIGN WITHIN BOX) Date | Signature (Joint Owners) Date |