 Novel Vascular Disrupting Agents For Orphan Oncology Indications February 2014 Exhibit 99.1 |
 2 2 This presentation contains forward-looking statements under the meaning of the Private Securities Litigation Reform Act of 1995. These statements give our current expectations or forecasts and use words such as “anticipate,” “estimate,” "expect," “believe,” and other words of similar meaning. Any or all of the forward-looking statements in this presentation may turn out to be wrong. They can be affected by inaccurate assumptions we might make or by known or unknown risks and uncertainties including but not limited to, the efficacy of our product candidates, their efficacy at acceptable dosage levels, the ability to raise capital when needed and on reasonable terms, projections of potential commercial sales of company products, the results and progress of clinical trials, developing the necessary manufacturing processes and gaining all necessary regulatory approvals, both in the United States and internationally. Consequently, no forward-looking statement can be guaranteed and actual results may differ materially. Additional information concerning factors that could cause actual results to materially differ from those in the forward-looking statements are contained in our most recent reports to the Securities and Exchange Commission including our Form 10-Q, 8-K and 10-K reports. However, we undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future events or otherwise. We note these factors for investors as permitted by the Private Securities Litigation Reform Act of 1995. The information in this document has been prepared solely for informational purposes and does not constitute an offer to sell or the solicitation of an offer to purchase any securities from any entities described herein. Any such offer will be made solely by means of the prospectus contained in the registration statement (the "Registration Statement") filed by OXiGENE, Inc. (the "Company") with the Securities and Exchange Commission (the "SEC"). The information contained herein may not be used in connection with an offer or solicitation by anyone in any jurisdiction in which such offer or solicitation is not permitted by law or in which the person making the offer or solicitation is not qualified to do so or to any person to whom it is unlawful to much such offer or solicitation. INVESTING IS SPECULATIVE AND INVOLVES RISK OF LOSS. YOU SHOULD REVIEW CAREFULLY THE REGISTRATION STATEMENT, INCLUDING THE DESCRIPTION OF THE RISKS AND OTHER TERMS BEFORE MAKING A DECISION TO INVEST. The Company has filed a Registration Statement (including a prospectus) with the SEC for the offering to which this presentation relates. Before you invest, you should read the prospectus contained in the Registration Statement, the information incorporated by reference into the Registration Statement, and other documents the Company has filed with the SEC for more complete information about the Company and the offering. You may get these documents for free by visiting EDGAR on the SEC website at http://www.sec.gov Alternatively, the Company or the placement agent participating in the offering will arrange to send you the prospectus contained in the Registration Statement if you request it by calling H.C. Wainwright & Co., LLC at (212) 356-0500. Safe Harbor And Free Writing Prospectus Statement |
 3 OXiGENE, Inc. (OXGN) Corporate Snapshot Development-stage biotechnology company Lead product candidate: ZYBRESTAT ® A vascular disrupting agent (VDA) being developed for ovarian cancer and other solid tumors primarily in combination with other anti-cancer agents which has shown utility and tolerability to date in combination with Avastin ® and other cancer drugs Follow-on product: OXi4503 A 2 nd – generation, dual-mechanism VDA being developed for acute myeloid leukemia (AML) Robust intellectual property (IP) portfolio consists of more than 120 patents worldwide Balance Sheet (as of YE 2013) No debt: cash $7.0M, projected to last into mid 3Q14 Equity Metrics (January 2014) Mkt Cap: $14.6M Shr Out: 5.6M (11.0M fully diluted) |
 OXiGENE (OXGN) Investment Thesis Compelling Valuation Currently trading at a market cap of approximately $14 million Leveraging $200+ million previously invested primarily in VDA development Cost-Efficient and Risk-Mitigated Development Strategy Ongoing and planned Ph 2 trials supported by foundations, non-profit research institutions and larger pharmaceutical companies Significant Potential Value-Creating Events on the Horizon Readout of Avastin ® +/- ZYBRESTAT ® Ph 2 in ovarian cancer – primary endpoint results expected 1H14 Potential initiation of FDA interaction and planning of pivotal NDA program for Avastin ® + ZYBRESTAT ® combination Initiation of the planned Phase 2 trial in neuroendocrine tumors (NETs) Demonstrate clinical activity for OXi4503 Phase 1 trial in AML Actively pursuing development and commercialization agreements with established industry leaders 4 |
 OXiGENE Leadership Team TEAM MEMBER EXPERIENCE Peter Langecker MD PhD Chief Executive Officer CIBA GEIGY (Novartis), Schering-Plough (Merck), Coulter (GSK), SUGEN (Pfizer), Intarcia, DURECT Barbara Riching CPA Chief Financial Officer Abgenix (Amgen), ALZA (J&J), Natural Wonders, Ernst & Young Alice Varga MS/MA VP Regulatory Affairs and Quality Assurance Syntex (Roche), SUGEN (Pfizer), PDL, Genentech/Roche, Cell Genesys (Bio Sante), Geron Kathleen Lee PhD MBA VP Chemistry, Manufacturing and Controls (CMC) Syntex (Roche), Scios (J&J), InteKrin Jai Balkissoon MD, FACS Clinical Development Consultant NCI Bethesda, Chiron (Novartis), Genentech/Roche, PPD Dai Chaplin PhD Scientific Advisor and Board Member University College, London, Cancer Research Campaign UK, RPR, Aventis 5 |
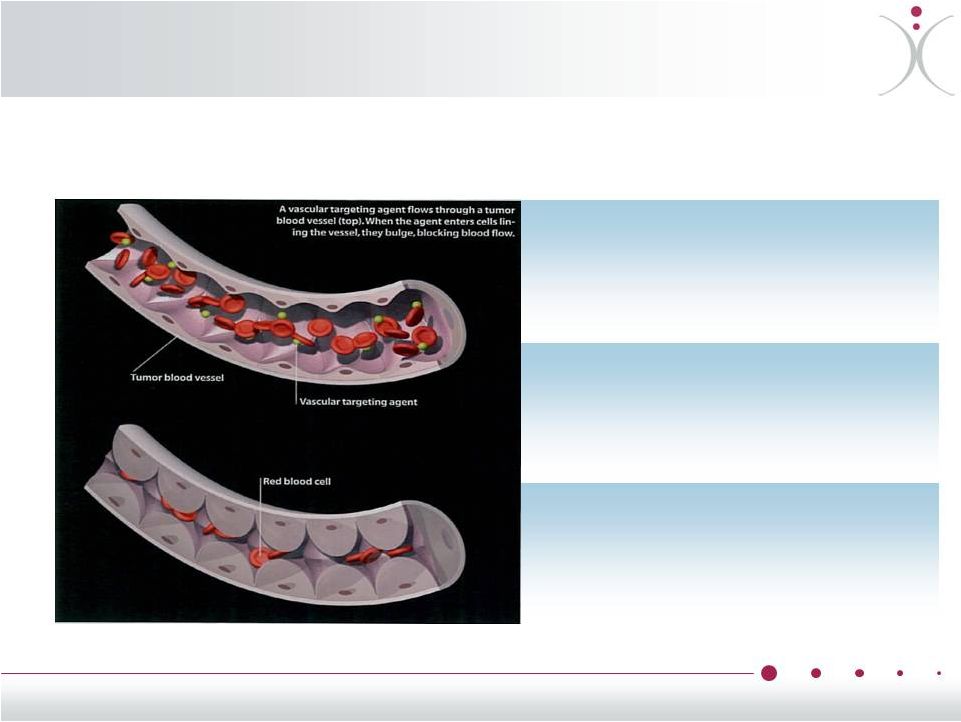 ZYBRESTAT ® : Reversible Tubulin Depolymerizing Agent Current chemotherapy is notorious for killing healthy cells along with cancerous ones – ZYBRESTAT® is designed to change that What is ZYBRESTAT? ZYBRESTAT ® , (fosbretabulin, CA4P), kills tumor cells by selectively blocking those blood vessels that carry vital oxygen and nutrients to tumors What does that do? ZYBRESTAT disrupts the scaffolding, and within hours the normally flat cells become round, blocking the blood flow to the tumor and causing massive tumor cell death How does it work? ZYBRESTAT preferentially binds to a protein called “Tubulin” within the cells that line the interior of tumor blood vessels 6 |
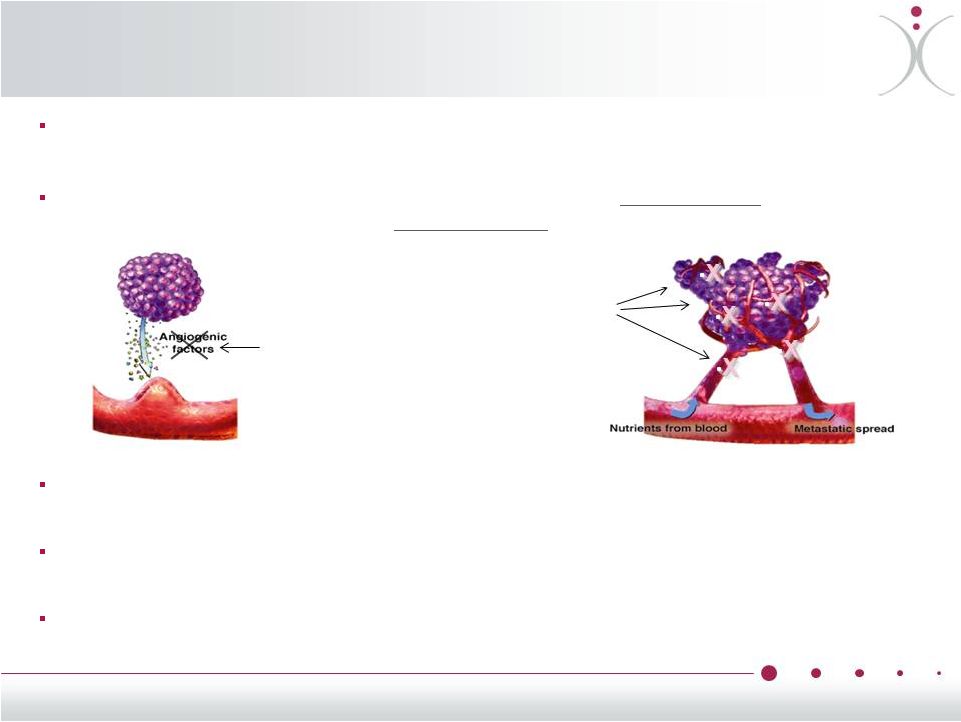 VDAs: A Targeted Approach to Tumor Necrosis Reducing tumor access to blood, oxygen and nutrients is very effective anti-cancer therapy as demonstrated by anti-VEGF therapy like bevacizumab (Avastin ® ) VDAs reduced blood flow to the center of the tumors by > 70% in 7/7 clinical trials as shown using 4 different imaging modalities Blood flow reduction leads to tumor necrosis of those regions of tumors believed to be associated with worse prognosis, often resistant to therapy Combining VDAs and Anti-VEGFs as well as other anti-cancer drugs have demonstrated enhanced preclinical and clinical activity and are subject of ongoing clinical research • Anti-VEGF • VDA Tubulin-targeted ascular isrupting gents (VDAs) work from the inside of the tumor while anti-VEGF compounds work from the outside of the tumor V D A 7 |
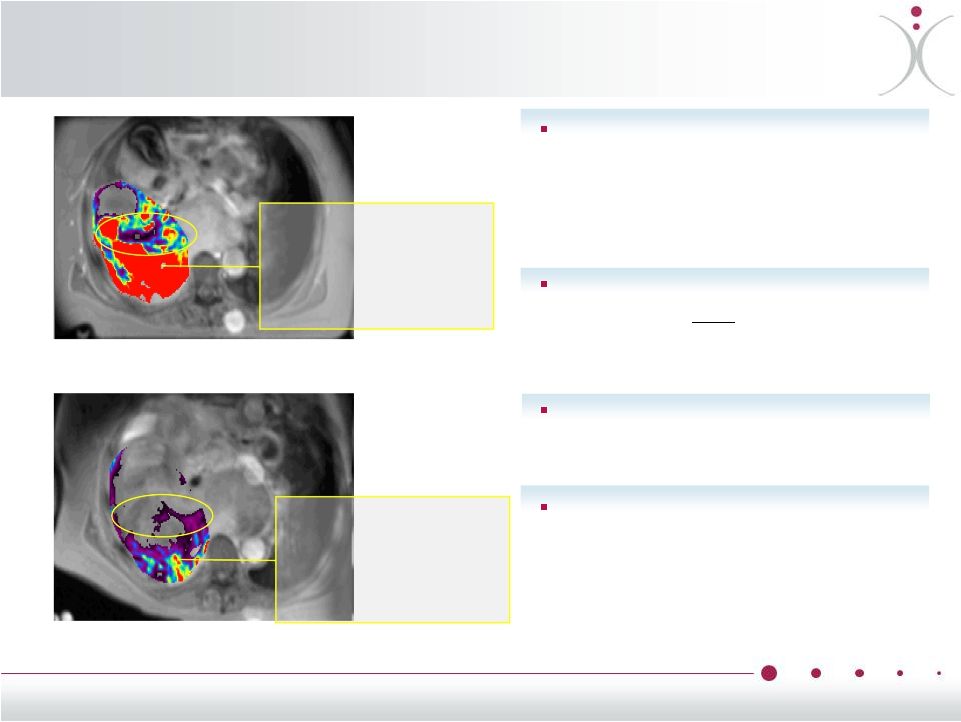 About Vascular Disrupting Agents (VDAs) Tumor flood flow before VDA Tumor blood flow 1 hour after VDA 8 Reducing tumor blood supply is a proven mechanism used by commercialized anti-VEGF drugs such as Avastin ® (bevacizumab) VDAs disrupt blood flow and vasculature only within tumors, sparing normal cells and organs VDAs are potentially useful in a wide range of tumors VDAs potentially improve clinical outcomes when used in combination with other types of anti-cancer therapies Red areas have high blood flow Blood flow significantly reduced |
 After many years of research, we believe ZYBRESTAT ® may become the first commercialized VDA… 9 |
 … attracting the support of numerous prestigious foundations, non-profit research institutions and “big pharma” companies. 10 |
 ZYBRESTAT ® : Dual Track to Potential Commercialization ZYBRESTAT ® – Clinically validated VDA Clinical activity in ATC, advanced ovarian cancer and other indications Good tolerability observed to date as mono- and combination therapy in 400+ patients Orphan Drug status for ATC and ovarian cancer in US and EU US/WW - Ovarian cancer clinical lead indication Activity and tolerability observed to date in combination with taxane-based chemotherapy or anti-VEGF therapy for ovarian cancer in Phase 1 and 2 clinical studies Combination with anti-VEGF (Avastin ® ) – randomized phase 2 data expected 1H2014 If Phase 2 is positive potential for subsequent partnered pivotal FDA NDA program Neuroendocrine tumors – potential novel indication for VDAs Positive preclinical results presented at AACR Potentially rapid development pathway, robust intellectual property Phase 2 study in patients with carcinoid tumors in planning EU approval path targeted for anaplastic thyroid cancer (ATC) Potential for 2016 commercialization of ZYBRESTAT ® Approval possible based on current data under “Exceptional Circumstances” Ongoing compassionate use program in EU and other countries 11 |
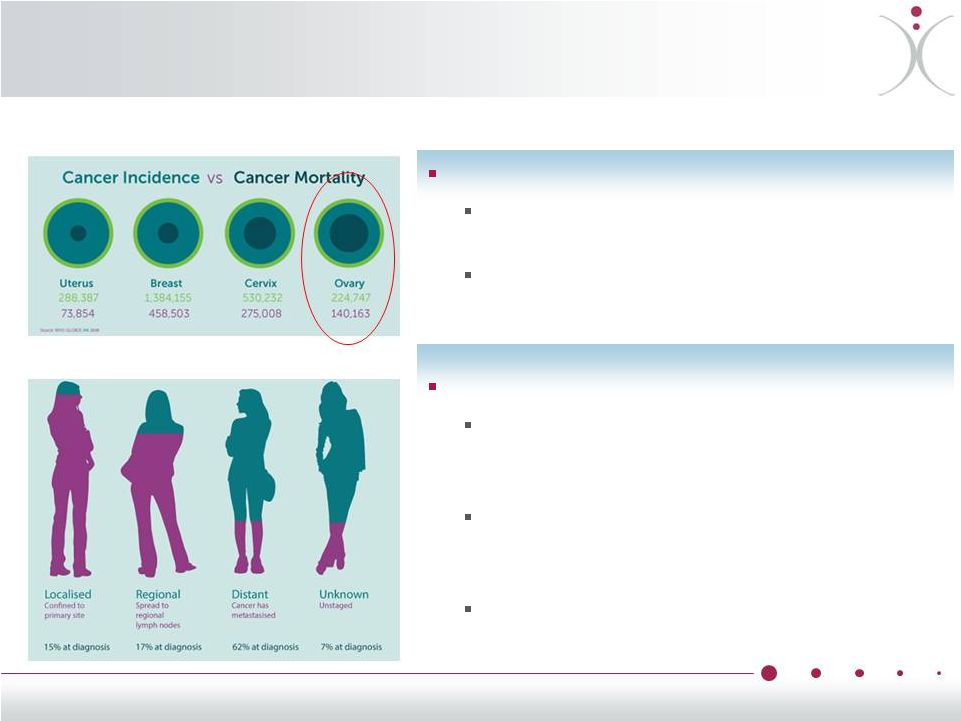 Why Ovarian Cancer? Aggressive, stealthy cancer ~22,000 new cases in the U.S. annually, ~220,000 new cases worldwide Over 60% of patients have distant spread at diagnosis resulting in poor prognosis Dire need for better therapies Highly lethal cancer that is responsible for the deaths of approximately 14,000 women in the U.S. each year Rapid resistance to existing FDA- approved treatments ultimately limits their effectiveness 5-year survival rates of only 47%, largely unchanged since the 1990s • Annual Incidence And Mortality Rates of Female Cancers Worldwide • Ovarian Cancer Spread at Diagnosis 12 |
 Collaborative effort between OXGN, Gynecologic Oncology Group (GOG), NCI (CTEP), Genentech / Roche Ongoing Phase 2 trial (GOG0186I) in ovarian cancer sponsored by GOG/CTEP Based on compelling preclinical data of bevacizumab/ZYBRESTAT combination Positive results from earlier Phase 1 trial 9/14 treated patients (60%) with ovarian cancer and other solid tumors had disease stabilization (m = 55 days) 2/4 patients with ovarian cancer had stable disease including a CA125 response lasting > 1 year Longer blood-flow shutdown shown via functional imaging with ZYBRESTAT Avastin ® +/- ZYBRESTAT ® in Solid Tumors 13 |
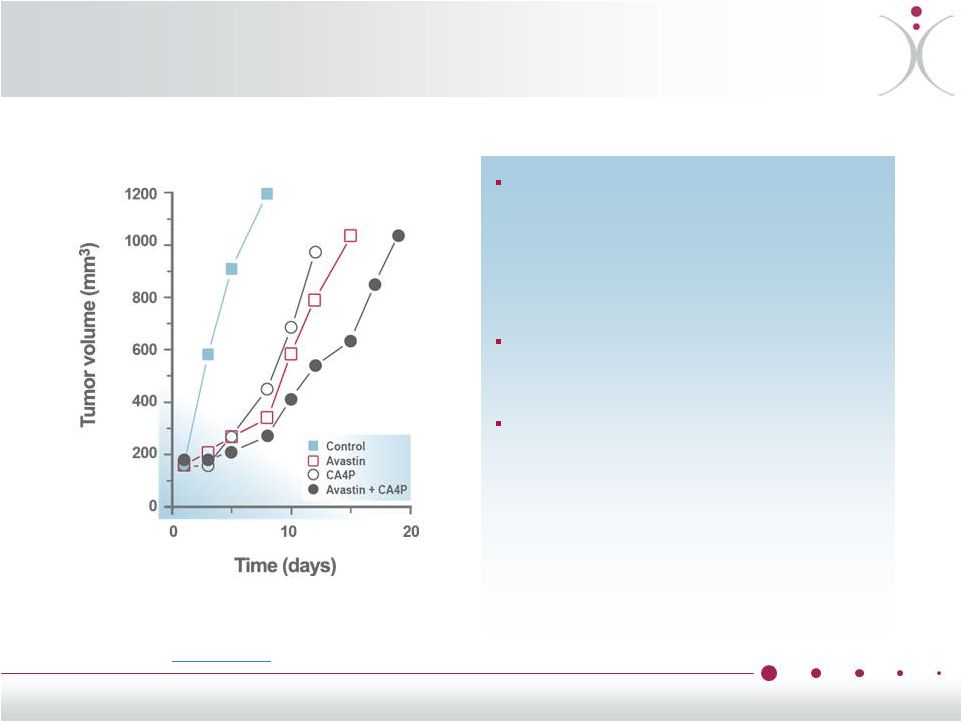 Preclinical Data Support Effects of ZYBRESTAT (CA4P) + Bevacizumab Combination Response of Caki-1 (renal cell) tumors to bevacizumab (2 mg/kg, twice a week for 2 weeks) and ZYBRESTAT (CA4P) 100 mg/kg, 3 times a week for 2 weeks or the combination of the ZYBRESTAT + bevacizumab Data shown represent the median tumor responses of groups of 8-10 mice The combination of ZYBRESTAT + bevacizumab resulted in greater reduction of tumor growth rates than in either compound alone Siemann et al., Anticancer Res. 2008 Jul-Aug;28(4B):2027-31 14 |
 GOG 0186I: Ongoing Phase 2 Study in Ovarian Cancer First randomized study to test anti-VEGF + VDA in advanced ovarian cancer – no cytotoxic chemotherapy involved 2nd-line, 3rd-line platinum-sensitive and platinum-resistant ovarian cancer at 67 clinical sites, 107 patients, controlled, 1:1 randomization Dosing: Bevacizumab +/- ZYBRESTAT, q 3 weeks Primary endpoint: demonstrate median progression free survival (PFS) increase from 50% to 65% Enrollment complete, two successful interim safety analyses and successful futility analysis for primary endpoint reported Primary endpoint readout expected 1H2014 Event-driven based on PFS Go/no go for potential pivotal study and potential end-of-Phase-2 meeting with FDA 2H2014 Potential path to NDA/ EU MAA filings in ovarian cancer 15 |
 Attractive Potential Commercial Opportunity Target patient population: Recurrent ovarian cancer – 80% of all ovarian cancer patients ZYBRESTAT in Ovarian Cancer: Use of anti-VEGF agents has been shown to be effective across different tumor types – potential for greater activity in combination with VDA like ZYBRESTAT® AVASTIN® already approved as single agent for ovarian cancer in EU and other countries Absence of chemotherapy and good tolerability would make the combination regimen with anti-VEGF agents attractive to patients Orphan drug status for ovarian cancer in US and EU for ZYBRESTAT® Potential for pivotal program exploring novel approach targeting tumor blood supply ® 16 |
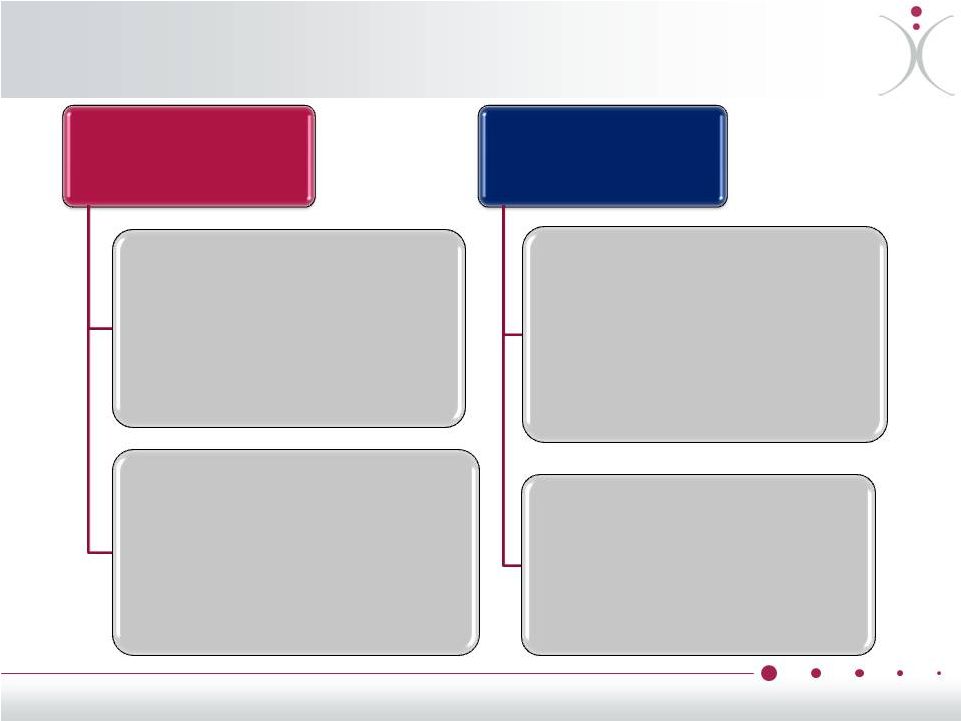 Risk-Mitigated, Cost Effective Development Pivotal Anti-VEGF Combination Program Potential Future Exploratory Combinations Ongoing Phase 2 with Avastin ® (bevacizumab) With Votrient ® (pazopanib) Potential Future Study with Avastin ® (bevacizumab) With weekly Taxol ® (paclitaxel) ZYBRESTAT in Ovarian Cancer – ® • Daily Votrient ® p.o. +/- ZYBRESTAT ® i.v., weekly x 3, q 4 weeks • Potentially Sponsored by UK Non-Profit Cancer Research Organization and Involved Drug Manufacturers and OXiGENE • Multicenter study, up to 120 patients • Primary endpoint: PFS • Initiation dependent on future funding and external partner • Potential Pivotal Phase 3 study • Avastin ® q 3 weeks +/- ZYBRESTAT ® i.v., q 3 weeks • Sample size, Primary Endpoint: based on FDA feedback if Phase 2 study is positive • Potential for CTEP/GOG support of follow-on study • Weekly paclitaxel +/- weekly ZYBRESTAT ® i.v., q 4 weeks • Multicenter study, up to 120 patients • Primary endpoint: PFS • Initiation dependent on future funding and external partner Avastin ® q 3 weeks +/- ZYBRESTAT ® i.v., q 3 weeks Sponsored by Genentech / Roche, GOG, NCI 107 patients enrolled at 67 clinical sites Primary Endpoint: PFS Primary endpoint data expected in 1H2014 • • • • • 17 |
 Neuroendocrine Tumors Including Carcinoid Slow-Growing, Biologically Active Tumors Neuroendocrine tumors (NETs) overproduce biologically active substances such as serotonin or insulin These can cause debilitating symptoms, including episodic flushing, diarrhea, wheezing, potentially carcinoid heart disease or hypoglycemia Low mitotic index often limits effectiveness of chemotherapy Limited Therapeutic Options Somatostatin analogues such as Sandostatin ® (octreotide) help to control symptoms, but the effect is often only temporary Serotonin inhibitors in development (only effective in serotonin-producing tumors). Non-responders are limited to surgical resection - if possible Chemotherapy only effective if mitotic index is high 18 |
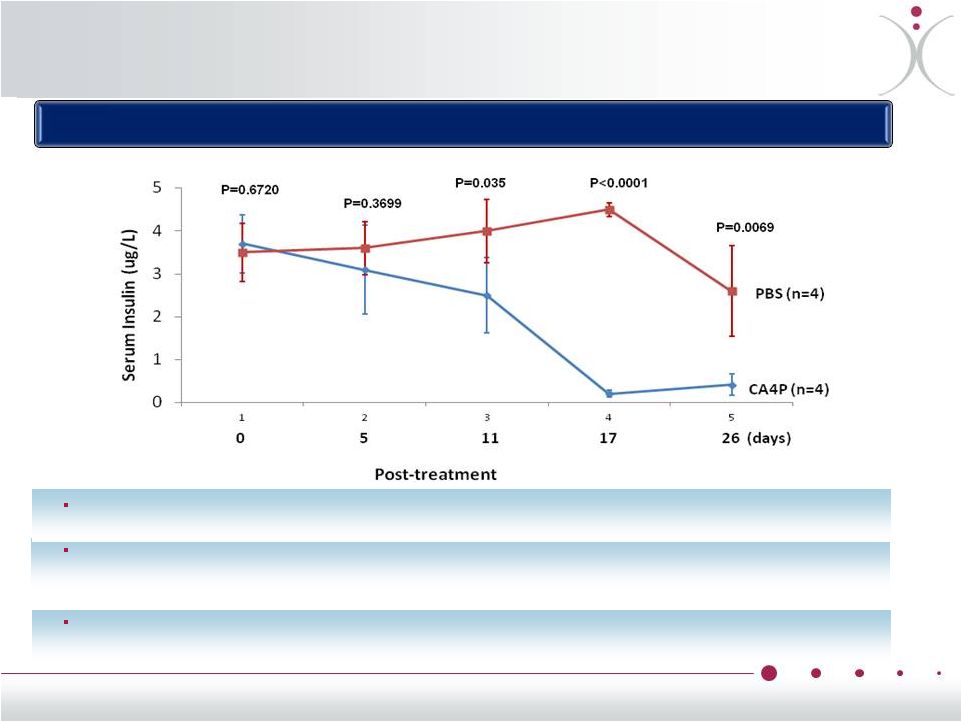 ZYBRESTAT in Pancreatic NET •* ZiQiang Yuan et al., Albert Einstein College of Medicine, Bronx, NY, 2013 AACR AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics Effect of CA4P on serum insulin level in Men1 KO mice with IP injection of CA4P or PBS for four weeks ZYBRESTAT ® decreased insulin levels in functional transgenic mouse model * ® Treatment with ZYBRESTAT ® resulted in a significant and sustained decrease in circulating insulin, with maximum effect seen by day 17 (CA4P group 0.213 ± 0.075 µg/L, versus PBS group 4.578 ± 0.161 µg/L, p<0.0001) The reduction in insulin was accompanied by a significantly (p=0.0128) reduced tumor size in the CA4P group (1.83 ± 1.27 mm 2 ) compared to the PBS group (9.88 ± 2.99 mm 2 ). 19 |
 Sandostatin ® (octreotide) +/- ZYBRESTAT ® Planned Phase 2 Self-Controlled Study 20 Sandostatin®-refractory GI- NET patients with increased biomarker levels (5-HIAA and/or chromogranin A) or clinical symptoms Primary endpoints: Biomarker (5-HIAA and/or chromogranin A levels) Secondary endpoints: Symptom control, Quality of Life Rationale: Positive Preclinical Results, Potentially Rapid Development Pathway, Robust Intellectual Property Positive preclinical data in prolactinoma and insulinoma models Potentially rapid readout and development based on biomarkers Exclusive, worldwide licensing agreement with Angiogene for the use of VDAs in the treatment of carcinoid syndrome and other neuroendocrine tumors (NETs) Additional IP filed 20 |
 ZYBRESTAT ® in Anaplastic Thyroid Cancer Completed Randomized Ph. 2/3 (FACT) Trial Results 80 patients with biopsy-proven ATC at 40 clinical sites Paraplatin® (carboplatin) / Taxol® (paclitaxel) +/- ZYBRESTAT® Primary endpoint: Median survival: 5.2 vs. 4.0 months (NS), HR 0.72 Secondary endpoints: 1-year survival 26 vs. 9%; OR rate 20 vs. 16% Target 2016 EMA marketing authorization in EU Potential for approval via “Exceptional Circumstances” pathway May not require additional clinical data Anaplastic Thyroid Cancer (ATC) Highly aggressive and lethal cancer with limited treatment options ~2,000 total patients annually in the US and EU Orphan drug status in both the US and EU 21 |
 ZYBRESTAT ® in ATC: Data Suggest Clinical Benefit •* Remick S. et al; Thyroid. 2009 March; 19(3): 233–240; **Sosa et al; Thyroid 2013 May 30. [Epub ahead of print] Phase 1 study in solid tumors – single agent therapy Including 7 ATC patients: 1 SD, 1 PR, and one long-lasting CR (14 years) Open label Phase 2 study in ATC patients – single agent therapy 26 patients with biopsy-proven ATC Median overall survival: 4.7 months 1-year survival: 23%, comparing favorably to historical control (<10%)* FACT Study – randomized, controlled Phase 2/3 study in ATC patients 80 patients with biopsy-proven ATC Carboplatin/paclitaxel +/- ZYBRESTAT ® randomized 2:1; q 3 weeks x 6, followed by ZYBRESTAT ® maintenance therapy or observation, until evidence of progression 22 |
 FACT Study – Summary of Results FACT Study results One-year survival rate 25.5% vs. 8.7% Median Overall Survival 5.2 vs. 4.0 months (HR 0.72) 25% higher Objective Response rate (20% vs. 16%) Forest Plot of Hazard Ratios for OS in subgroups Lower HR with use of ZYBRESTAT ® for patients 60 years of age, prior thyroid surgery, prior chemotherapy, prior radiation therapy, stage IVC disease and tumor size >6 cm; suggesting greater antitumor activity in more advanced tumors Safety Addition of ZYBRESTAT ® to standard chemotherapy was observed to be well tolerated with AEs primarily related to ATC and disease progression Treatment-related AEs were easily clinically managed 23 |
 ZYBRESTAT ® Clinical Safety Profile Observed in Clinical trials To Date No cumulative toxicities or cytotoxic-like side-effects Most AEs related to underlying disease or disease progression Adverse events are mainly low grade, reversible, transient, and manageable Absence of significant cardiac side effects - transient mild to moderate hypertension effectively managed through antihypertensive prophylaxis Transient myelosuppression in association with chemotherapy, manageable without stopping therapy Sequence of administration relevant for myelosuppression – less if ZYBRESTAT given after chemotherapy 400+ patient safety data set 24 |
 Unique, Dual Mechanism of Action Tubulin-mediated cell shape change of endothelial and leukemic cells Metabolized by oxidative enzymes (e.g., tyrosinases and peroxidases) into an orthoquinone species with direct cytotoxic effects on tumor cells Compelling Preclinical and Early Clinical Data Recommended dose, objective responses and significant blood flow reductions shown in Ph 1/2 studies in refractory solid tumors Demonstrated near-complete elimination of FLT-3 mutated human AML (leukemic clone) in systemic xenograft in SCID mouse model Ongoing Dose-Escalating Phase 1 Study in Hematologic Malignancies Robust Intellectual Property (IP) Protected by both composition-of-matter and method-of-use patents Exclusive, worldwide license to the combretastatins discovered and isolated by researchers at Arizona State University (ASU) OXiGENE’s OXi4503: A 2nd-Generation VDA 25 |
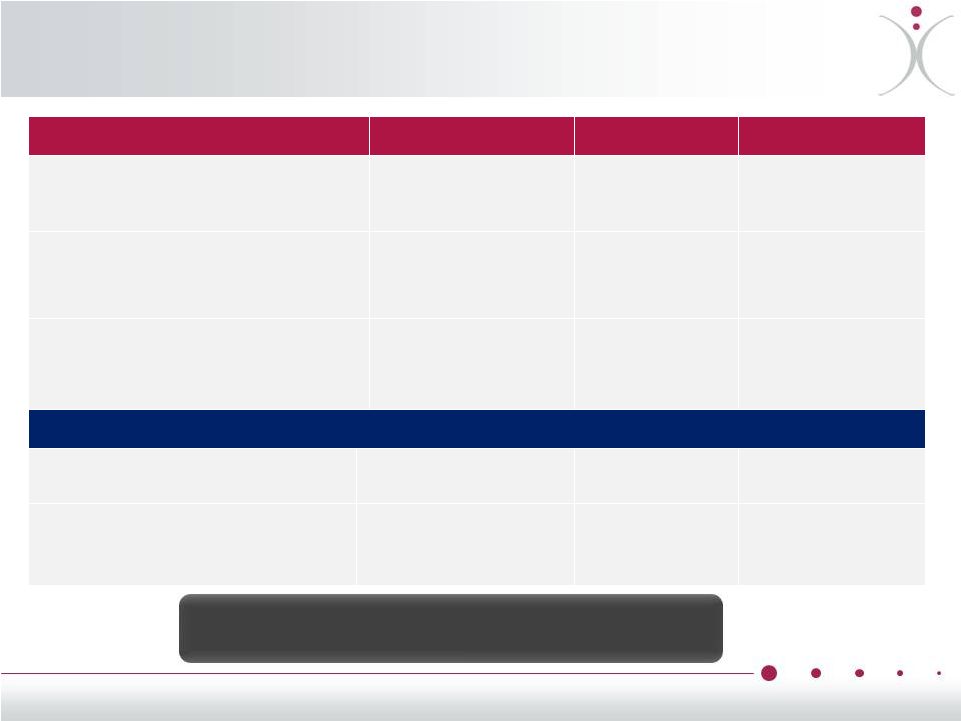 Robust Intellectual Property Portfolio ZYBRESTAT® United States Europe Japan Composition of Matter: fosbretabulin tromethamine September 2021 US 7659261, 7524832 & 7659262 September 2021 EPO 1320534 September 2021 JP 4149804 Method of modulating tumor growth or metastasis by administration of fosbretabulin and paclitaxel December 2021 US 6,538,038 N/A N/A Method of treating NET including carcinoid tumor symptoms by administering fosbretabulin June 2033 PCT* pending June 2033 PCT *pending June 2033 PCT* pending OXi4503 Composition of Matter: OXi4503 – salt form including potassium October 2021 US 7,078,552 April 2021 EPO 1278758 February 2020 pending Method of treating myeloid neoplasm by administering OXi4503 November 2028 November 2028 November 2028 JP 5302328 • *A Patent Cooperation Treaty (PCT) application establishes a filing date in all 148 contracting states. OXiGENE is the owner or exclusive licensee of more than 120 patents worldwide 26 |
 OXiGENE - Where is the Opportunity? Significant Therapeutic and Commercial Potential Potential to significantly improve outcomes in solid tumor indications Example: Antisoma received $75M upfront and $915 M in potential development-based milestones for ASA404 from Novartis Challenging Development History Limits Competition Apart from OXiGENE only 3 other companies are known to be currently developing VDAs, none of which have progressed to the submission stage OXiGENE believes it has determined the optimal way ZYBRESTAT ® should be developed and brought to the market ZYBRESTAT® May Become the First Commercialized VDA Clinically meaningful activity with no significant safety issues to date Previously identified cardiovascular response was found to be easily managed with no abrogation of the anti-tumor effects 27 |
 OXiGENE - Financial Overview Cost-Efficient, “Virtual Company” Infrastructure (as of YE 2013) No debt, cash: $7.0M, projected to last into mid 3Q14 – Raised $9.1M in 2013 via ATM and two PIPEs, including proceeds from exercise of PIPE warrants, net of redemption and expenses. Common Stock: 5.6M outstanding (11.0M fully diluted) Use of Proceeds from Offering Completion of the ongoing GOG0186I trial in ovarian cancer – most of the costs were borne by NCI/CTEP If GOG0186I is positive, initiate pursuit of pivotal program with FDA input and potential partner If GOG0186I is positive, partial funding of potential Phase 1/2 trial in ovarian cancer with ® in combination with Votrient® EU MAA filing for ® ® combo in ATC including manufacturing of mandatory registration lots of ® Initiate Phase 2 study in gastrointestinal neuroendocrine tumors / ZYBRESTAT ZYBRESTAT Paraplatin Taxol 28 |
 OXiGENE (OXGN) Investment Thesis Compelling Valuation Currently trading at a market cap of approximately $14 million Leveraging $200+ million previously invested primarily in VDA development Cost-Efficient and Risk-Mitigated Development Strategy Ongoing and planned Ph 2 trials supported by foundations, non-profit research institutions and larger pharmaceutical companies Significant Potential Value-Creating Events on the Horizon Readout of Avastin ® +/- ZYBRESTAT ® Ph 2 in ovarian cancer – primary endpoint results expected 1H14 Potential initiation of FDA interaction and planning of pivotal NDA program for Avastin ® + ZYBRESTAT ® combination Initiation of the planned Phase 2 trial in neuroendocrine tumors (NETs) Demonstrate clinical activity for OXi4503 Phase 1 trial in AML Actively pursuing development and commercialization agreements with established industry leaders 29 |