Exhibit 99.1
|
Corporate Overview
William D. Schwieterman, MD
President and Chief Executive Officer
October 2015
|
Safe Harbor Statement
This presentation contains forward-looking statements under the meaning of the Private Securities Litigation Reform Act of 1995. These statements give our current expectations or forecasts and use words such as “anticipate,” “estimate,” “expect,” “believe,” and other words of similar meaning. Any or all of the forward-looking statements in this presentation may turn out to be wrong. They can be affected by inaccurate assumptions we might make or by known or unknown risks and uncertainties including but not limited to, the efficacy of our product candidates, their efficacy at acceptable dosage levels, the ability to raise capital when needed and on reasonable terms, projections of potential commercial sales of company products, the results and progress of clinical trials, developing the necessary manufacturing processes and gaining all necessary regulatory approvals, both in the United States and internationally. Consequently, no forward-looking statement can be guaranteed and actual results may differ materially. Additional information concerning factors that could cause actual results to materially differ from those in the forward-looking statements are contained in our most recent reports to the Securities and Exchange Commission including our Form 10-Q, 8-K and 10-K reports. However, we undertake no obligation to publicly update forward-looking statements, whether as a result of new information, future events or otherwise. We note these factors for investors as permitted by the Private Securities Litigation Reform Act of 1995.
2 |
|
|
Management Team
Team Member Experience
William Schwieterman, MD Perceptive Advisors, Chelsea Therapeutics, FDA
President and Chief Executive Officer
David Chaplin, PhD Aventis, Rhône-Poulenc Rorer, Cancer Research
Chief Scientific Officer United Kingdom, University College London
Kathleen Lee, PhD, MBA InteKrin, Scios, Syntex
VP Chemistry, Manufacturing and Controls (CMC)
Matthew Loar KineMed, Neurobiological Technologies, Osteologix,
Chief Financial Officer Genelabs Technologies
Jeff Nelson Axsome Therapeutics, Chelsea Therapeutics,
VP Program Management Ladenburg Thalmann, Cobalt Laboratories
Alice Varga, MS, MA Geron, Cell Genesys, Genentech/Roche,
VP Regulatory Affairs and Quality Assurance Protein Design Labs, SUGEN, Syntex
3
|
OXiGENE: Company Overview
Oncology focused biopharmaceutical company Developing vascular disrupting agents (VDAs)
VDAs selectively disrupt abnormal blood vessels that sustain tumors
Clinical development candidates
CA4P (fosbretabulin)
– Ovarian Cancer
– Glioblastoma Multiforme (GBM)
– Neuroendocrine Tumors (NET)
OXi4503
– Acute Myeloid Leukemia (AML) / Myelodysplastic Syndrome (MDS)
4
|
Product Pipeline Summary
Preclinical Phase 1 Phase 2 Phase 3
CA4P
Platinum resistant ovarian cancer
CA4P + bevacizumab + chemotherapy vs. Phase 2/3 initiating bevacizumab + chemotherapy
Glioblastoma multiforme (GBM)
Phase 2/3 initiating
CA4P + bevacizumab vs bevacizumab
Recurrent ovarian cancer
Phase 1b/2 ongoing
CA4P + pazopanib (PAZOFOS)
Neuroendocrine tumors (NETs)
Phase 2a ongoing
CA4P Monotherapy
OXi4503
AML
Phase 1/2 ongoing
OXi4503 + cytarabine
5
|
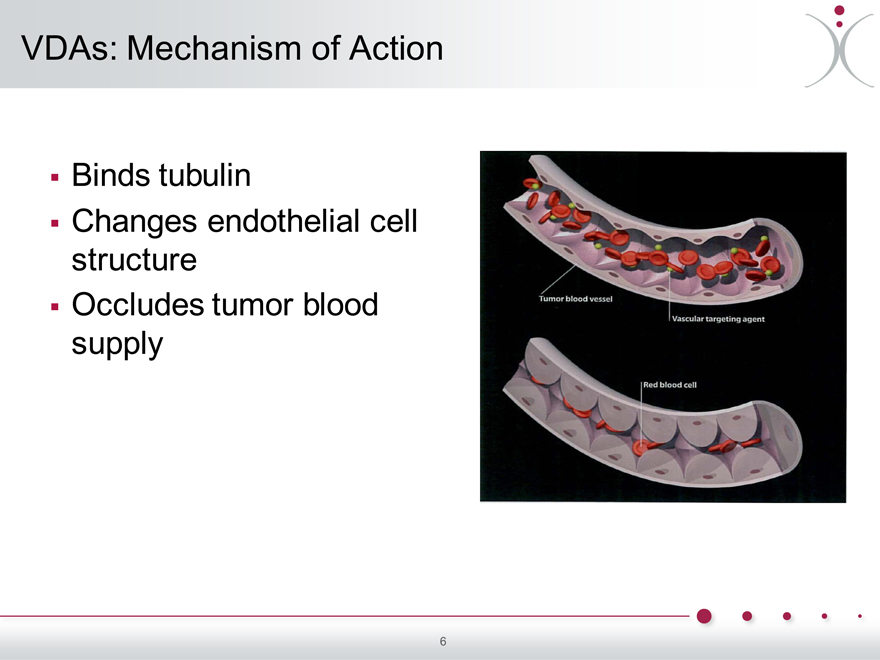
VDAs: Mechanism of Action
Binds tubulin
Changes endothelial cell structure Occludes tumor blood supply
6
|
VDAs: Selectively Targets Tumor Vasculature
Vascular Cast
Pericyte Immuno-fluorescence
Normal Tumor
Pericyte support No pericyte support
McDonald DM, Choyke PL. Nat Med. 2003;9(6):713-25. 7
|
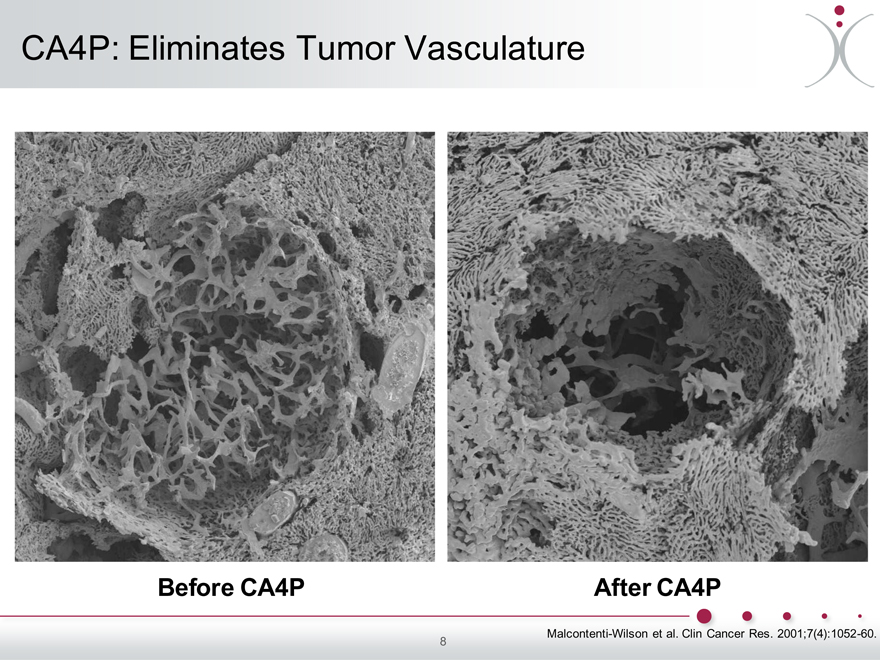
CA4P: Eliminates Tumor Vasculature
Before CA4P After CA4P
Malcontenti-Wilson et al. Clin Cancer Res. 2001;7(4):1052-60. 8
|
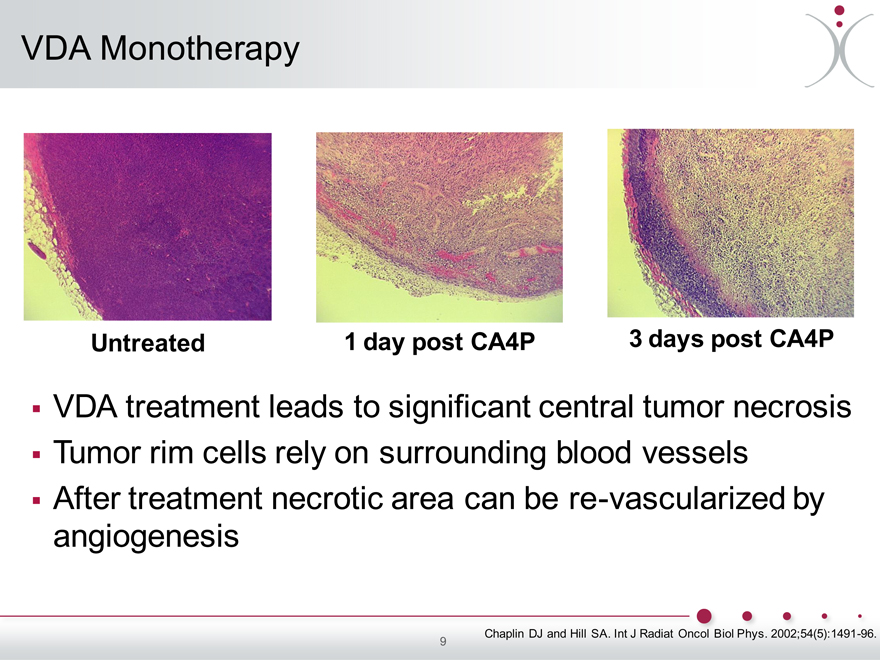
VDA Monotherapy
Untreated 1 day post CA4P 3 days post CA4P
VDA treatment leads to significant central tumor necrosis Tumor rim cells rely on surrounding blood vessels After treatment necrotic area can be re-vascularized by angiogenesis
Chaplin DJ and Hill SA. Int J Radiat Oncol Biol Phys. 2002;54(5):1491-96. 9
|
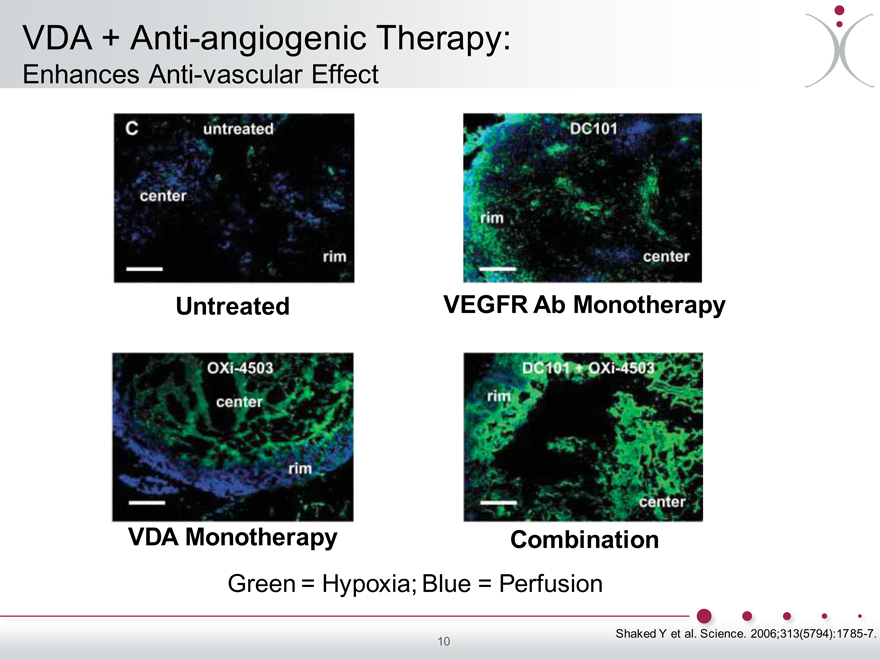
VDA + Anti-angiogenic Therapy:
Enhances Anti-vascular Effect
Untreated VEGFR Ab Monotherapy
VDA Monotherapy Combination
Green = Hypoxia; Blue = Perfusion
Shaked Y et al. Science. 2006;313(5794):1785-7.
C
Untreated center rim DC101 center rim OXi-4503 center rim
DC101 + OXi-4503 center rim
10
|
Previously Conducted Studies
|
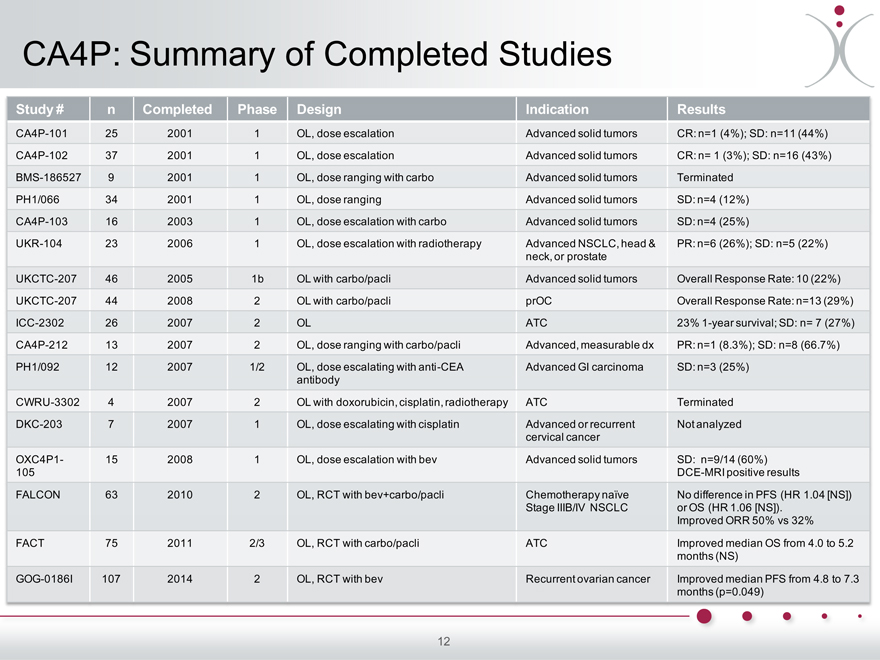
CA4P: Summary of Completed Studies
Study # n Completed Phase Design Indication Results
CA4P-101 25 2001 1 OL, dose escalation Advanced solid tumors CR: n=1 (4%); SD: n=11 (44%)
CA4P-102 37 2001 1 OL, dose escalation Advanced solid tumors CR: n= 1 (3%); SD: n=16 (43%)
BMS-186527 9 2001 1 OL, dose ranging with carbo Advanced solid tumors Terminated
PH1/066 34 2001 1 OL, dose ranging Advanced solid tumors SD: n=4 (12%)
CA4P-103 16 2003 1 OL, dose escalation with carbo Advanced solid tumors SD: n=4 (25%)
UKR-104 23 2006 1 OL, dose escalation with radiotherapy Advanced NSCLC, head & PR: n=6 (26%); SD: n=5 (22%)
neck, or prostate
UKCTC-207 46 2005 1b OL with carbo/pacli Advanced solid tumors Overall Response Rate: 10 (22%)
UKCTC-207 44 2008 2 OL with carbo/pacli prOC Overall Response Rate: n=13 (29%)
ICC-2302 26 2007 2 OL ATC 23% 1-year survival; SD: n= 7 (27%)
CA4P-212 13 2007 2 OL, dose ranging with carbo/pacli Advanced, measurable dx PR: n=1 (8.3%); SD: n=8 (66.7%)
PH1/092 12 2007 1/2 OL, dose escalating with anti-CEA Advanced GI carcinoma SD: n=3 (25%)
antibody
CWRU-3302 4 2007 2 OL with doxorubicin, cisplatin, radiotherapy ATC Terminated
DKC-203 7 2007 1 OL, dose escalating with cisplatin Advanced or recurrent Not analyzed
cervical cancer
OXC4P1- 15 2008 1 OL, dose escalation with bev Advanced solid tumors SD: n=9/14 (60%)
105 DCE-MRI positive results
FALCON 63 2010 2 OL, RCT with bev+carbo/pacli Chemotherapy naïve No difference in PFS (HR 1.04 [NS])
Stage IIIB/IV NSCLC or OS (HR 1.06 [NS]).
Improved ORR 50% vs 32%
FACT 75 2011 2/3 OL, RCT with carbo/pacli ATC Improved median OS from 4.0 to 5.2
months (NS)
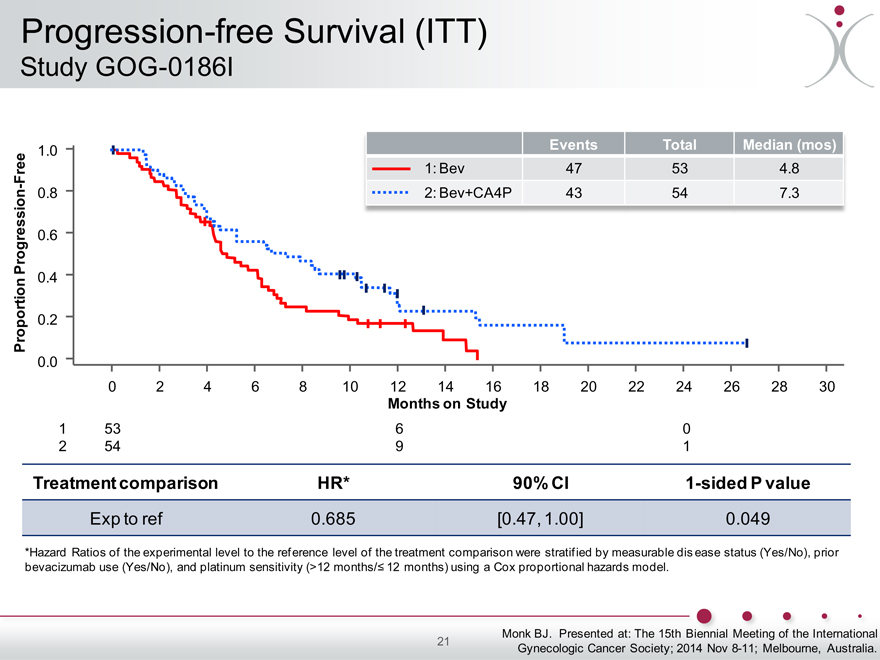
GOG-0186I 107 2014 2 OL, RCT with bev Recurrent ovarian cancer Improved median PFS from 4.8 to 7.3
months (p=0.049)
12
|
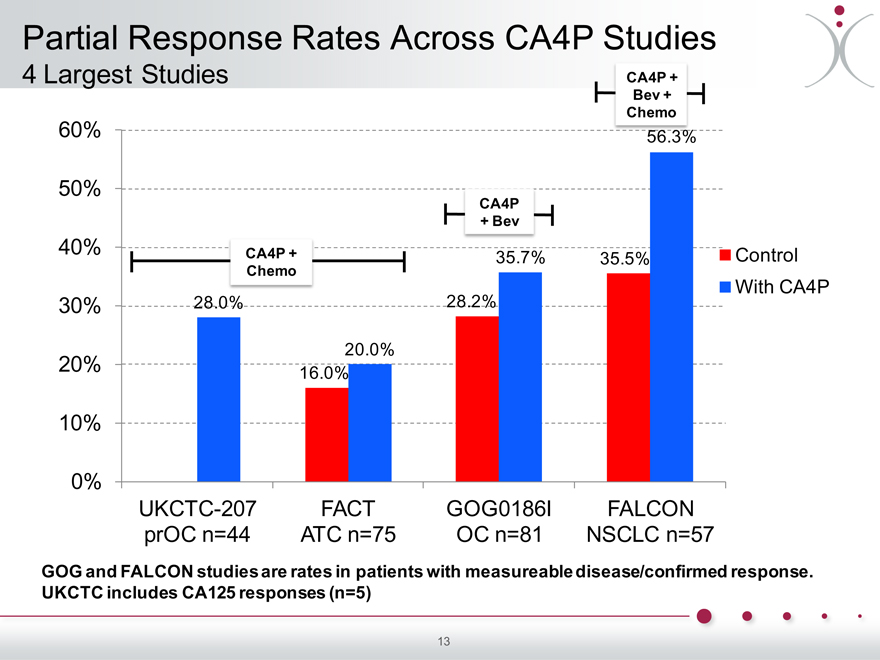
Partial Response Rates Across CA4P Studies
4 Largest Studies CA4P +
Bev + Chemo
60% 56.3%
50%
CA4P
+ Bev
40% CA4P + Control
35.7% 35.5%
Chemo With CA4P
30% 28.0% 28.2%
20.0% 20% 16.0%
10%
0%
UKCTC-207 FACT GOG0186I FALCON prOC n=44 ATC n=75 OC n=81 NSCLC n=57
GOG and FALCON studies are rates in patients with measureable disease/confirmed response. UKCTC includes CA125 responses (n=5)
13
|
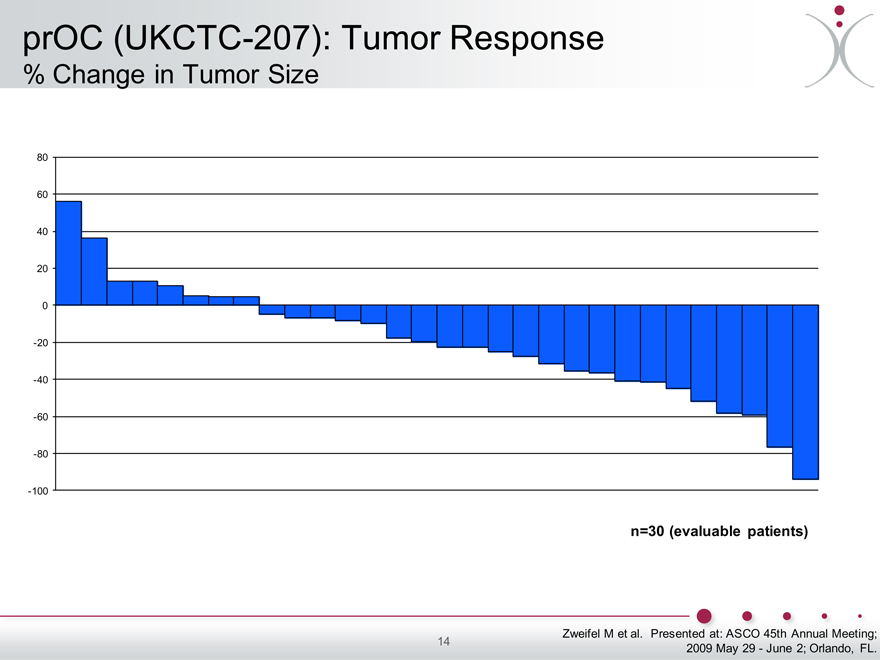
prOC (UKCTC-207): Tumor Response
% Change in Tumor Size
80 60 40 20 0 -20 -40 -60 -80 -100
n=30 (evaluable patients)
Zweifel M et al. Presented at: ASCO 45th Annual Meeting; 2009 May 29—June 2; Orlando, FL.
14
|
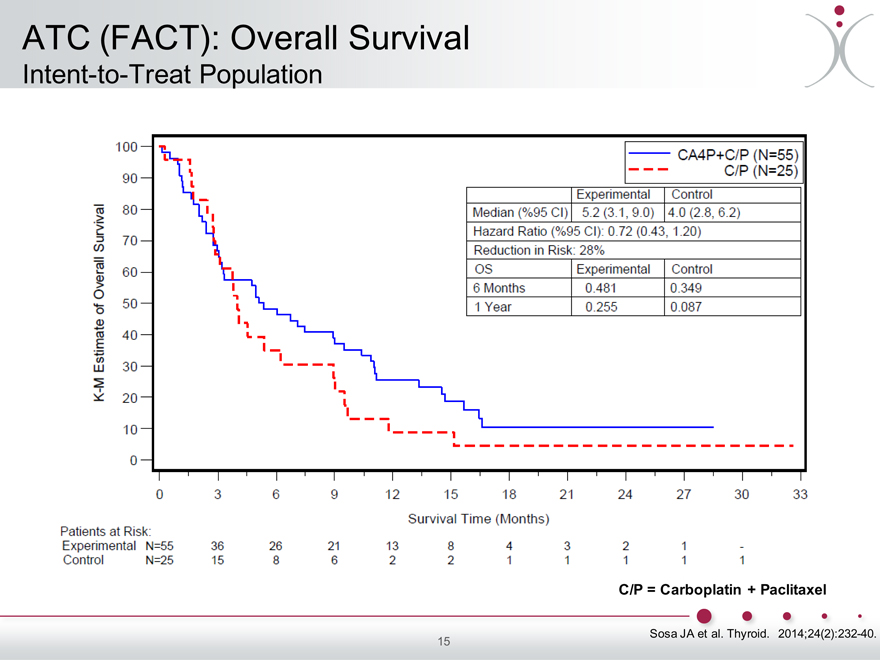
ATC (FACT): Overall Survival
Intent-to-Treat Population
K-M Estimate of Overall Survival
100 90 80 70 60 50 40 30 20 10 0
0 3 6 9 12 15 18 21 24 27 30 33
CA4P+C/P (N=55) C/P (N=25)
Experimental Control
Median (%95 Cl)
Sosa JA et al. Thyroid. 2014;24(2):232-40.
15
|
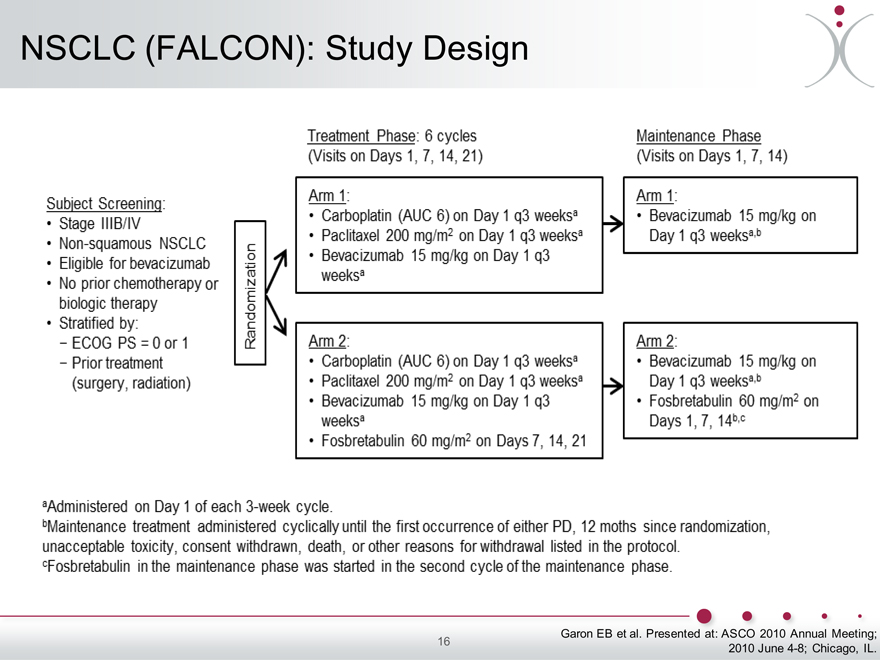
NSCLC (FALCON): Study Design
Garon EB et al. Presented at: ASCO 2010 Annual Meeting;
2010 June 4-8; Chicago, IL.
16
|
NSCLC (FALCON): Clinical Summary
CA4P + Bev + Chemo Bev + Chemo
n (%) n (%)
Total number of subjects 32 31
Response Rate
Complete Response 0 0
Partial Response 18 (56.3) 11 (35.5)
Stable Disease 8 (25.0) 13 (41.9)
Progressive Disease 1 (3.1) 3 (9.7)
Not Evaluable 5 (15.6) 4 (12.9)
Confirmed Partial Response 16 (50) 10 (32.3)
95% Cl 31.9%, 68.1% 16.7%, 51.4%
Disposition
Discontinued Prior to end of 15 (46.8) 12 (38.7)
Treatment Phase (4.5 months)*
Median Time to Progression 8.6 months 9.0 months
Garon EB et al. Presented at: ASCO 2010 Annual Meeting;
2010 June 4-8; Chicago, IL.
17
|
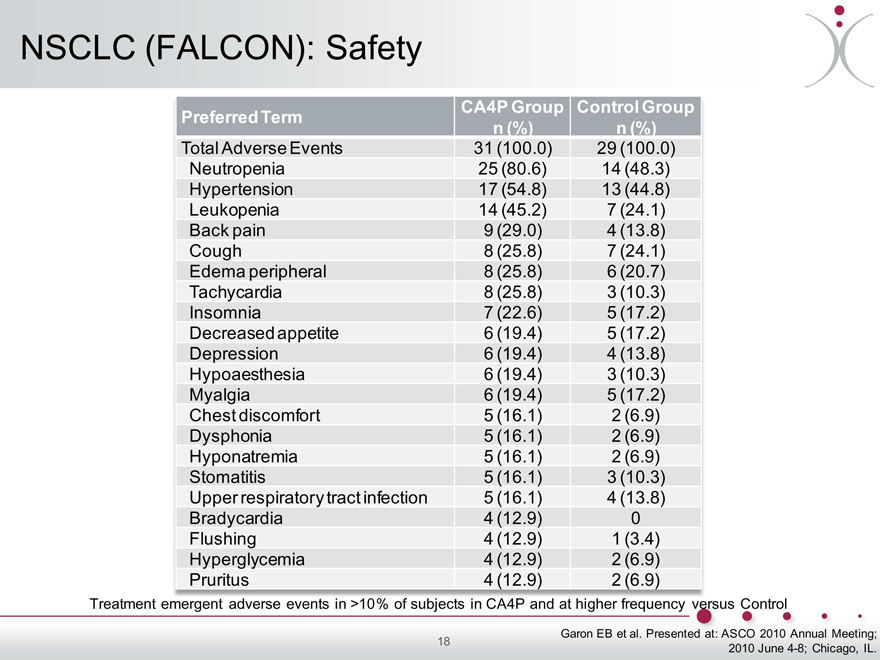
NSCLC (FALCON): Safety
Preferred Term CA4P Group Control Group
n (%) n (%)
Total Adverse Events 31 (100.0) 29 (100.0)
Neutropenia 25 (80.6) 14 (48.3)
Hypertension 17 (54.8) 13 (44.8)
Leukopenia 14 (45.2) 7 (24.1)
Back pain 9 (29.0) 4 (13.8)
Cough 8 (25.8) 7 (24.1)
Edema peripheral 8 (25.8) 6 (20.7)
Tachycardia 8 (25.8) 3 (10.3)
Insomnia 7 (22.6) 5 (17.2)
Decreased appetite 6 (19.4) 5 (17.2)
Depression 6 (19.4) 4 (13.8)
Hypoaesthesia 6 (19.4) 3 (10.3)
Myalgia 6 (19.4) 5 (17.2)
Chest discomfort 5 (16.1) 2 (6.9)
Dysphonia 5 (16.1) 2 (6.9)
Hyponatremia 5 (16.1) 2 (6.9)
Stomatitis 5 (16.1) 3 (10.3)
Upper respiratory tract infection 5 (16.1) 4 (13.8)
Bradycardia 4 (12.9) 0
Flushing 4 (12.9) 1 (3.4)
Hyperglycemia 4 (12.9) 2 (6.9)
Pruritus 4 (12.9) 2 (6.9)
Treatment emergent adverse events in >10% of subjects in CA4P and at higher frequency versus Control
Garon EB et al. Presented at: ASCO 2010 Annual Meeting;
2010 June 4-8; Chicago, IL.
18
|
GOG-0186I Combination Study: Recurrent Ovarian Cancer
|
Phase 2 Study in Recurrent Ovarian Cancer
Study GOG-0186I
n=107 (67 sites); randomized controlled study CA4P combined with bevacizumab
Bevacizumab (Avastin?) 15 mg/kg IV q3 weeks (n=54) Bevacizumab 15 mg/kg+CA4P 60 mg/m2 IV q3 wks (n=53)
Patient Population: 2nd- and 3rd-line ovarian cancer
Both platinum-sensitive and platinum-resistant
Primary endpoint: PFS
Ongoing follow-up for overall survival
Monk BJ. Presented at: The 15th Biennial Meeting of the International
Gynecologic Cancer Society; 2014 Nov 8-11; Melbourne, Australia.
20
|
Progression-free Survival (ITT)
Study GOG-0186I
Events Total Median (mos)
1.0
Free 1: Bev 47 53 4.8
- 0.8
2: Bev+CA4P 43 54 7.3
Progression 0.6
0.4
Proportion 0.2
0.0
0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30
Months on Study
1 53 6 0
2 54 9 1
Treatment comparison HR* 90% CI 1-sided P value
Exp to ref 0.685 [0.47, 1.00] 0.049
*Hazard Ratios of the experimental level to the reference level of the treatment comparison were stratified by measurable disease status (Yes/No), prior bevacizumab use (Yes/No), and platinum sensitivity (>12 months/? 12 months) using a Cox proportional hazards model.
Monk BJ. Presented at: The 15th Biennial Meeting of the International
Gynecologic Cancer Society; 2014 Nov 8-11; Melbourne, Australia.
21
|
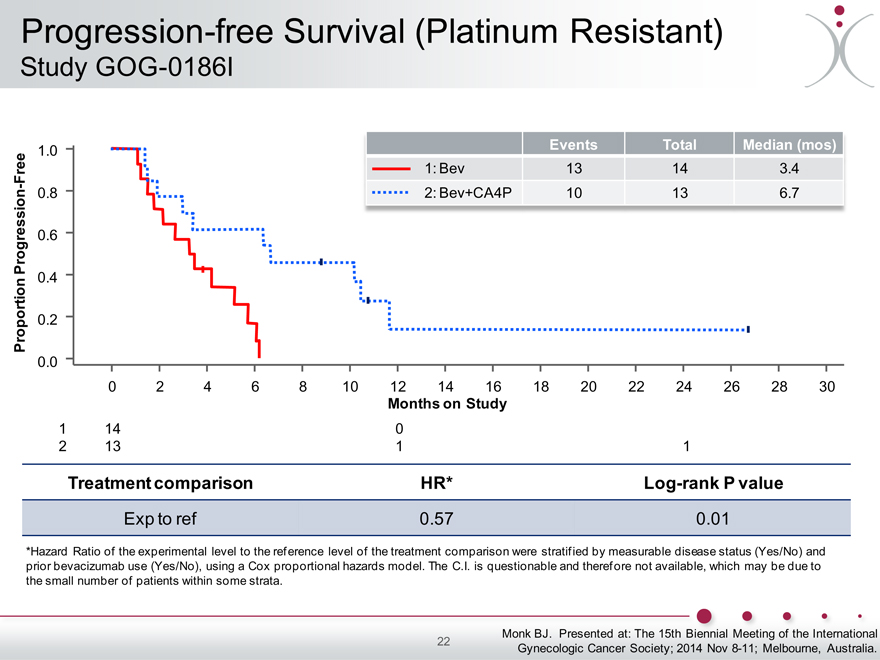
Progression-free Survival (Platinum Resistant)
Study GOG-0186I
Events Total Median (mos) 1.0 Free 1: Bev 13 14 3.4
- 0.8
2: Bev+CA4P 10 13 6.7
Progression 0.6
0.4
Proportion 0.2
0.0
0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30
Months on Study
1 14 0
2 13 1 1
Treatment comparison HR* Log-rank P value
Exp to ref 0.57 0.01
*Hazard Ratio of the experimental level to the reference level of the treatment comparison were stratified by measurable disease status (Yes/No) and prior bevacizumab use (Yes/No), using a Cox proportional hazards model. The C.I. is questionable and therefore not available, which may be due to the small number of patients within some strata.
Monk BJ. Presented at: The 15th Biennial Meeting of the International
Gynecologic Cancer Society; 2014 Nov 8-11; Melbourne, Australia.
22
|
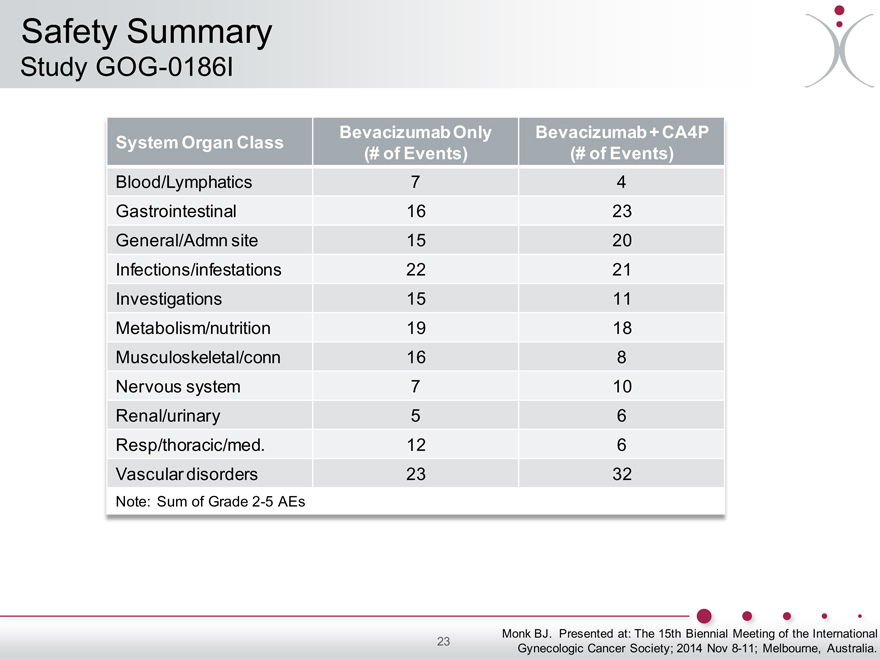
Safety Summary
Study GOG-0186I
System Organ Class Bevacizumab Only Bevacizumab + CA4P
(# of Events) (# of Events)
Blood/Lymphatics 7 4
Gastrointestinal 16 23
General/Admn site 15 20
Infections/infestations 22 21
Investigations 15 11
Metabolism/nutrition 19 18
Musculoskeletal/conn 16 8
Nervous system 7 10
Renal/urinary 5 6
Resp/thoracic/med. 12 6
Vascular disorders 23 32
Note: Sum of Grade 2-5 AEs
Monk BJ. Presented at: The 15th Biennial Meeting of the International
Gynecologic Cancer Society; 2014 Nov 8-11; Melbourne, Australia.
23
|
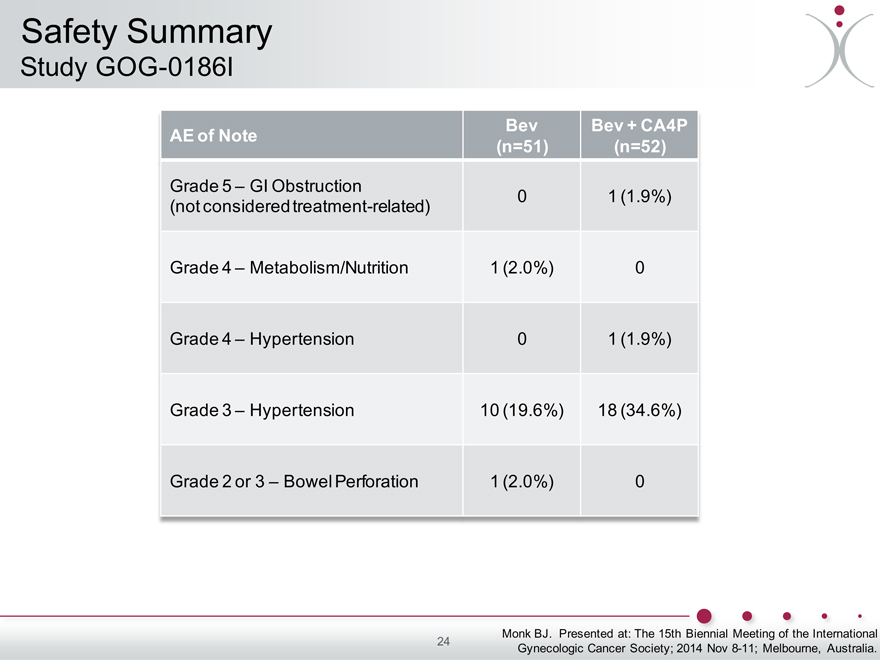
Safety Summary
Study GOG-0186I
Bev Bev + CA4P
AE of Note (n=51) (n=52)
Grade 5 – GI Obstruction
(not considered treatment-related) 0 1 (1.9%)
Grade 4 – Metabolism/Nutrition 1 (2.0%) 0
Grade 4 – Hypertension 0 1 (1.9%)
Grade 3 – Hypertension 10 (19.6%) 18 (34.6%)
Grade 2 or 3 – Bowel Perforation 1 (2.0%) 0
Monk BJ. Presented at: The 15th Biennial Meeting of the International
Gynecologic Cancer Society; 2014 Nov 8-11; Melbourne, Australia.
24
|
Summary of Completed Studies
CA4P shows signs of efficacy in multiple studies
Efficacy seen across range of tumor types
Safety profile and dosing well characterized
CA4P + anti-angiogenics maximizes tumor ischemia CA4P + bevacizumab improves PFS in ovarian cancer
Largest effect in platinum-resistant (GOG-0186I)
25
|
GBM as a New Target Indication for CA4P
Significant unmet medical need Few effective therapies High mortality rate
Most patients have recurrent disease
Rapid disease progression—shorter time to determine treatment effects Expected fast enrollment period Bevacizumab—standard treatment for recurrent disease Combination anti-vascular approach for highly vascular tumor CA4P has demonstrated efficacy in preclinical GBM model
26
|
Clinical Development Plan
Combine VDAs with products with complementary mechanisms of action Take multiple shots on goal
Conduct two stage Phase 2/3 trials: CA4P + bevacizumab
Platinum-resistant ovarian cancer (prOC)
– First stage – randomized controlled trial, site blinded, continual interim analyses (n=80)
– Second stage – randomized controlled trial, double-blind confirmatory efficacy trial (n = 300)
Glioblastoma multiforme (GBM)
– First stage – randomized controlled trial, site blinded, continual interim analyses (n=80)
– Second stage – randomized controlled trial, double-blind confirmatory efficacy trial (n = 300)
27
|
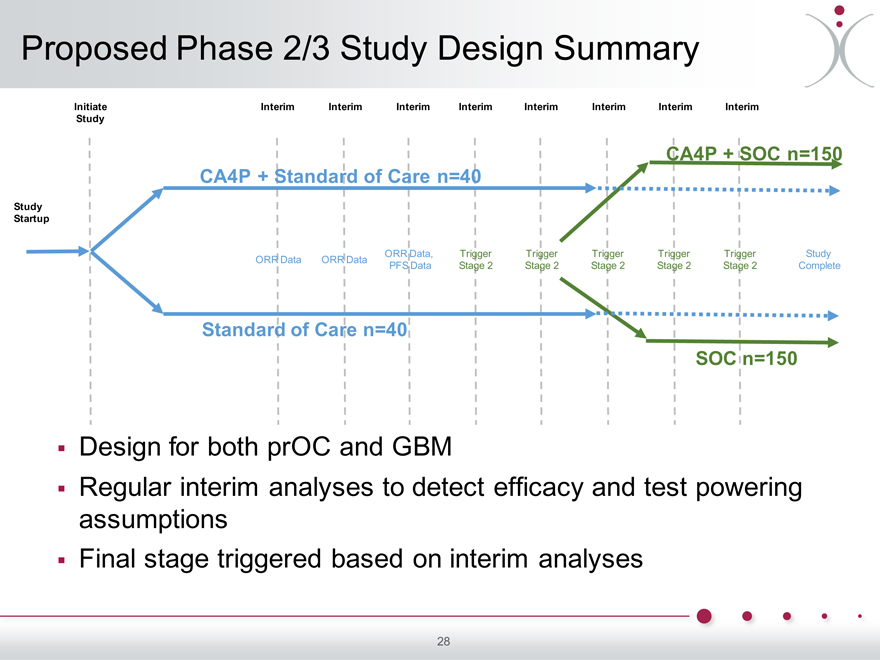
Proposed Phase 2/3 Study Design Summary
Initiate Interim Interim Interim Interim Interim Interim Interim Interim Study
CA4P + SOC n=150 CA4P + Standard of Care n=40
Study Startup
ORR Data, Trigger Trigger Trigger Trigger Trigger Study ORR Data ORR Data PFS Data Stage 2 Stage 2 Stage 2 Stage 2 Stage 2 Complete
Standard of Care n=40
SOC n=150
Design for both prOC and GBM
Regular interim analyses to detect efficacy and test powering assumptions Final stage triggered based on interim analyses
28
|
OXi4503 Program Summary
|
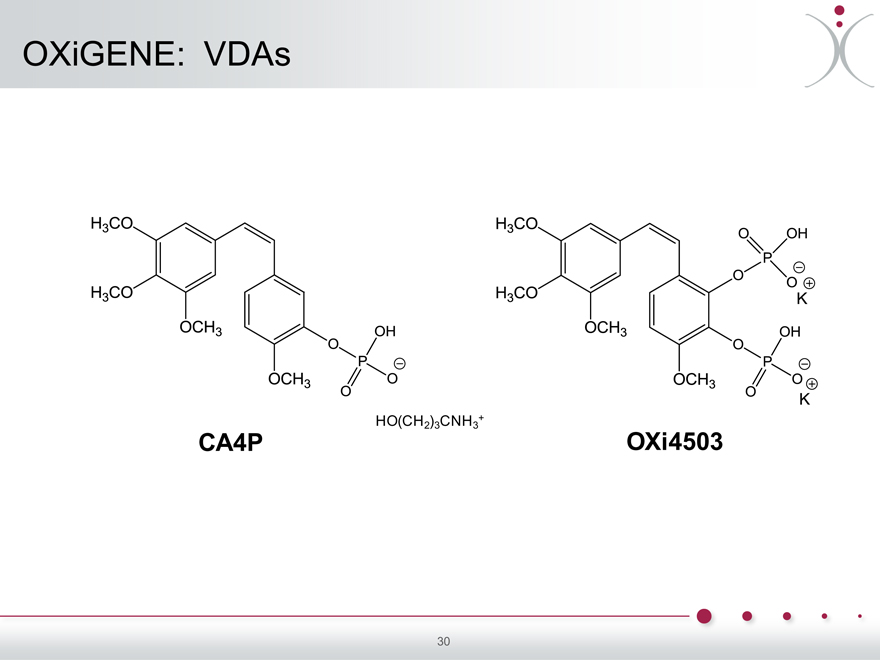
OXiGENE: VDAs
HO(CH ) CNH +
2 3 3
CA4P OXi4503
30
|
OXi4503: Summary of Completed Studies
Study # n Completed Phase Design Indication Results
PH1/098 43 2010 1 OL, dose escalation Advanced solid CR: n=1 (4%); SD: n=11 tumors (48%)* OXC101- 16 2011 1b OL, dose escalation 1o and 2o SD: n=1 (6%) 100 hepatic tumors
*Based on evaluable patients
31
|
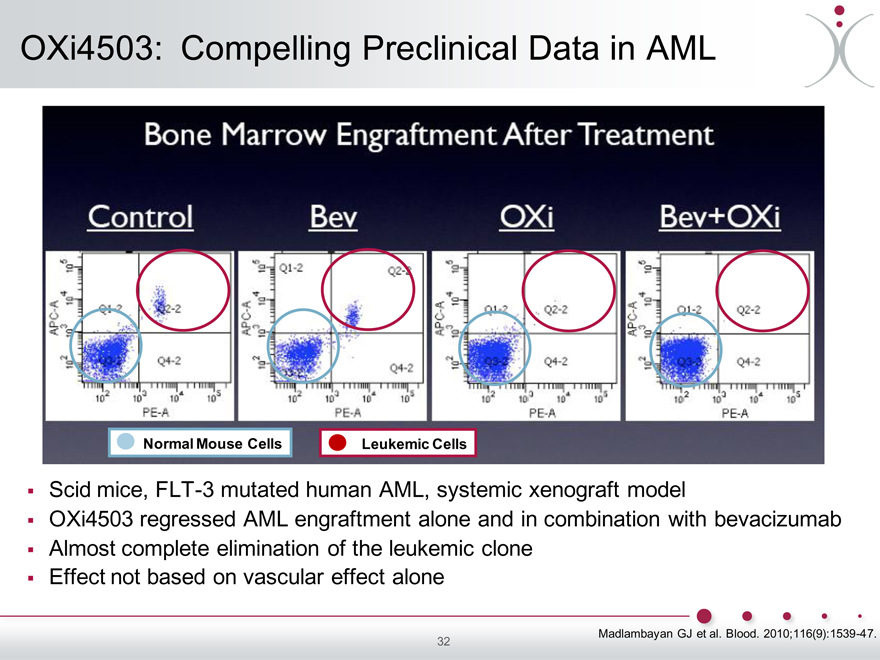
OXi4503: Compelling Preclinical Data in AML
Scid mice, FLT-3 mutated human AML, systemic xenograft model
OXi4503 regressed AML engraftment alone and in combination with bevacizumab
Almost complete elimination of the leukemic clone
Effect not based on vascular effect alone
Madlambayan GJ et al. Blood. 2010;116(9):1539-47.
32
|
AML (UF OXi4503 AML MDS Ph1):
Preliminary Data
n=18 (1 site); open-label study
CA4P as a monotherapy infused on Days 1, 8, 15, and 21 of a 28-day cycle
Dose cohorts: 2.5 mg/m2 (n=2), 3.75 mg/m2 (n=2), 5 mg/m2 (n=9), 6.25 mg/m2 (n=3), 7.81 mg/m2 (n=2)
Patient Population: Refractory AML (16/18 patients) and MDS (2/18 patients) CR n=1 (6%) PR n=1 (6%), response was sustained for 10 months
Data on File
33
|
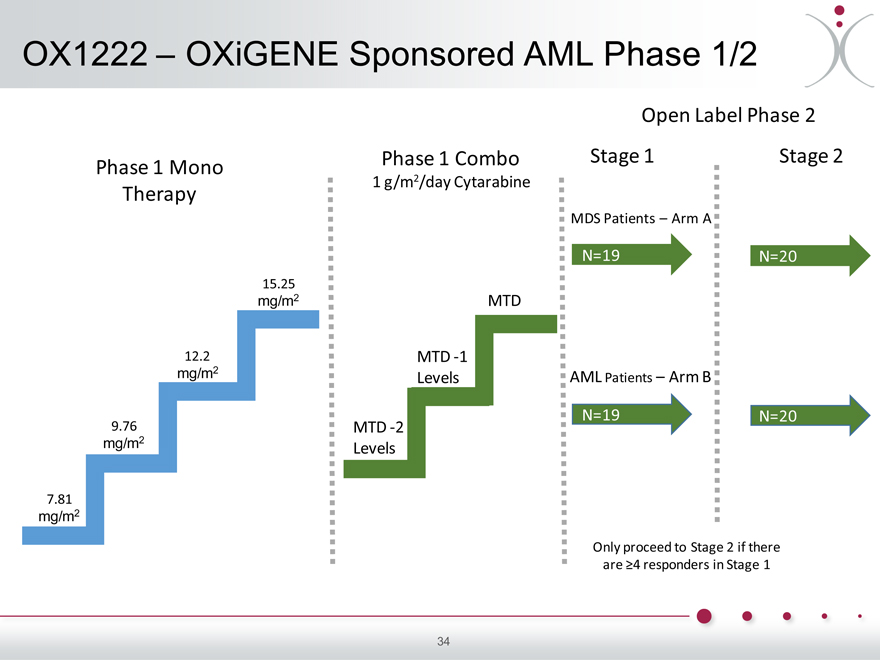
OX1222 – OXiGENE Sponsored AML Phase 1/2
Open Label Phase 2
Phase 1 Combo Stage 1 Stage 2
Phase 1 Mono
1 g/m2/day Cytarabine
Therapy
MDS Patients – Arm A
N=19 N=20
15.25 mg/m2 MTD
12.2 MTD -1
mg/m2
Levels AML Patients – Arm B
N=19 N=20
9.76 MTD -2
mg/m2
Levels
7.81 mg/m2
Only proceed to Stage 2 if there are ?4 responders in Stage 1
34
|
Finances
Stock listed on Nasdaq Capital Market: OXGN $33 million cash at June 30, 2015
Approximately $30 million estimated for September 30, 2015 No preferred stock or debt outstanding
Approximately 26.5 million shares outstanding
Additional 11.7 million warrants and options outstanding at average exercise price of $2.51
35
|
Milestones
4Q2015
Phase 1b/2 Ovarian study – interim data Phase 2 NET study – complete enrollment Phase 1/2a OXi4503 AML – initiate study Phase 2 NET study – interim data
1H2016
Phase 2/3 prOC – initiate study
Phase 1 OXi4503 AML study – interim data
Additional CA4P preclinical data: melanoma, GBM, colon cancer
2H2016
Phase 2/3 GBM – initiate study Phase 2 NET study – final data
Phase 1 AML study – end of monotherapy arm
36
|
Summary
CA4P alone shows activity across multiple tumor types CA4P + anti-angiogenics – enhances activity Multiple shots on goal
prOC: CA4P + bevacizumab + chemotherapy GBM: CA4P + bevacizumab AML: OXi4503 + cytarabine Phase 1/2 study NET: CA4P monotherapy Phase 2 study Recurrent OC: CA4P + pazopanib
New management team
Focus on larger randomized trials
37
|
Thank you
www.oxigene.com 650-635-7000