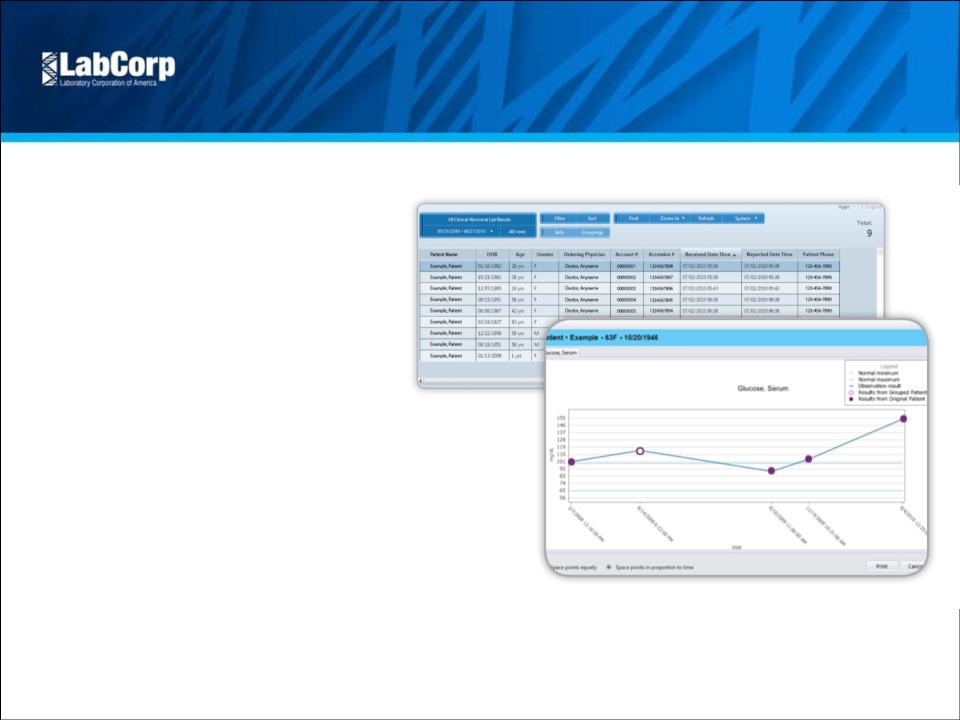
- LH Dashboard
- Financials
- Filings
-
Holdings
- Transcripts
- ETFs
- Insider
- Institutional
- Shorts
-
8-K Filing
Laboratory Corporation of America (LH) 8-KRegulation FD Disclosure
Filed: 10 Nov 11, 12:00am
| Delaware | 1-11353 | 13-3757370 | ||
| (State or other jurisdiction of Incorporation) | (Commission File Number) | (I.R.S. Employer Identification No.) |
| 358 South Main Street, | ||||
| Burlington, North Carolina | 27215 | 336-229-1127 | ||
| (Address of principal executive offices) | (Zip Code) | (Registrant’s telephone number including area code) |
| [ ] | Written communication pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| [ ] | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| [ ] | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| [ ] | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
| Item 7.01 | Regulation FD Disclosure |
| By: | /s/ F. SAMUEL EBERTS III | |
| F. Samuel Eberts III | ||
| Chief Legal Officer and Secretary |














Partner | Clinical Area |
ARCA biopharma | Companion Diagnostics (Cardiovascular Disease) |
BG Medicine | Cardiovascular Disease |
Duke University | Joint Venture in biomarker development |
Duke University | Lung Cancer |
Exact Sciences | Colon Cancer |
Intema Ltd. | Prenatal Testing |
Johns Hopkins | Melanoma |
MDxHealth | Companion Diagnostics (Oncology) |
Medco Health Solutions | Companion Diagnostics (Research) |
Merck | Companion Diagnostics (Infectious Disease) |
University of Minnesota | Lupus |
Veridex | Prostate Cancer |
Yale University | Ovarian Cancer (exclusive) |




Three Months Ended Sep 30, | Nine Months Ended Sep 30, | ||||||||||||||
2011 | 2010 | +/(-) | 2011 | 2010 | +/(-) | ||||||||||
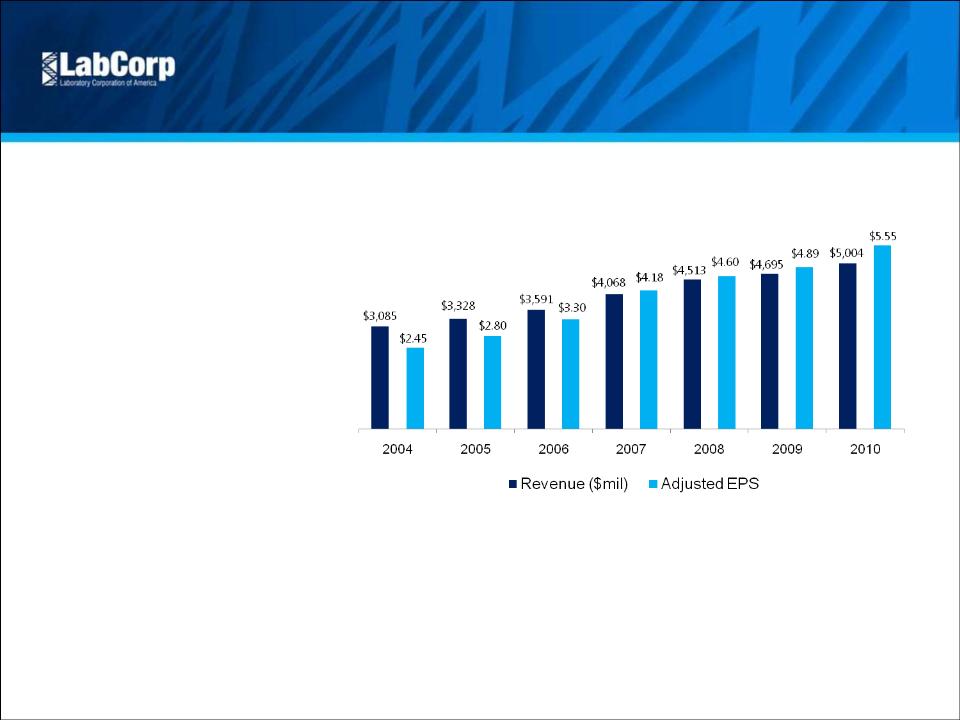
Revenue | $ 1,404.5 | $ 1,276.5 | 10.0% | $ 4,176.2 | $ 3,708.5 | 12.6% | |||||||||
Adjusted Operating Income (1) | $ 263.5 | $ 250.1 | 5.4% | $ 806.8 | $ 764.1 | 5.6% | |||||||||
Adjusted Operating Income Margin (1) | 18.8% | 19.6% | -80 | bp | 19.3% | 20.6% | -130 | bp | |||||||
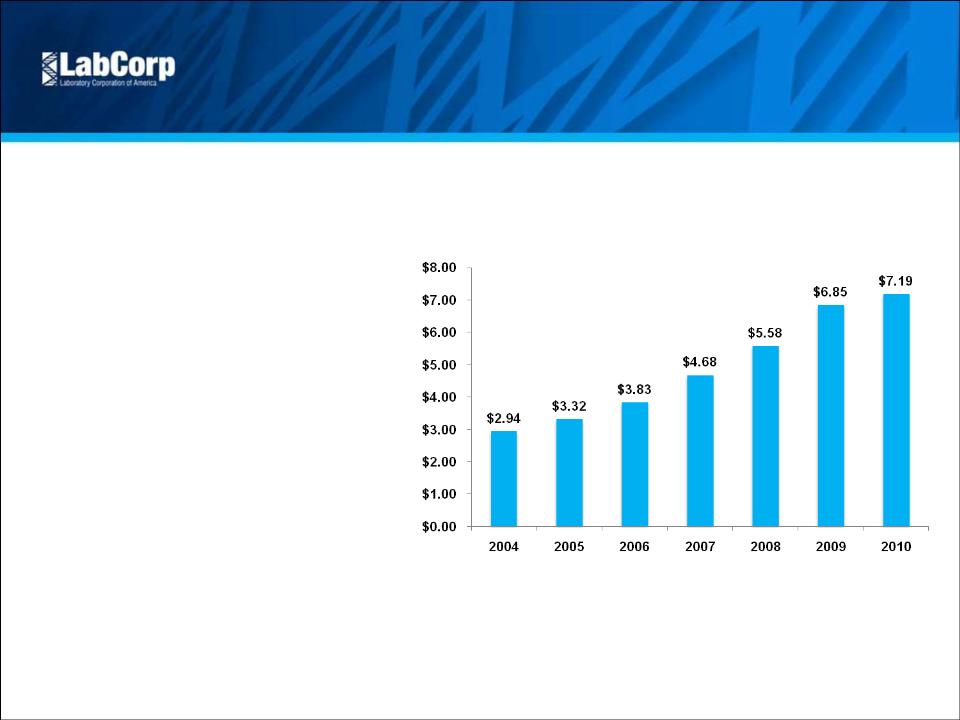
Adjusted EPS Excluding Amortization (1) | $ 1.61 | $ 1.58 | 1.9% | $ 4.80 | $ 4.54 | 5.7% | |||||||||
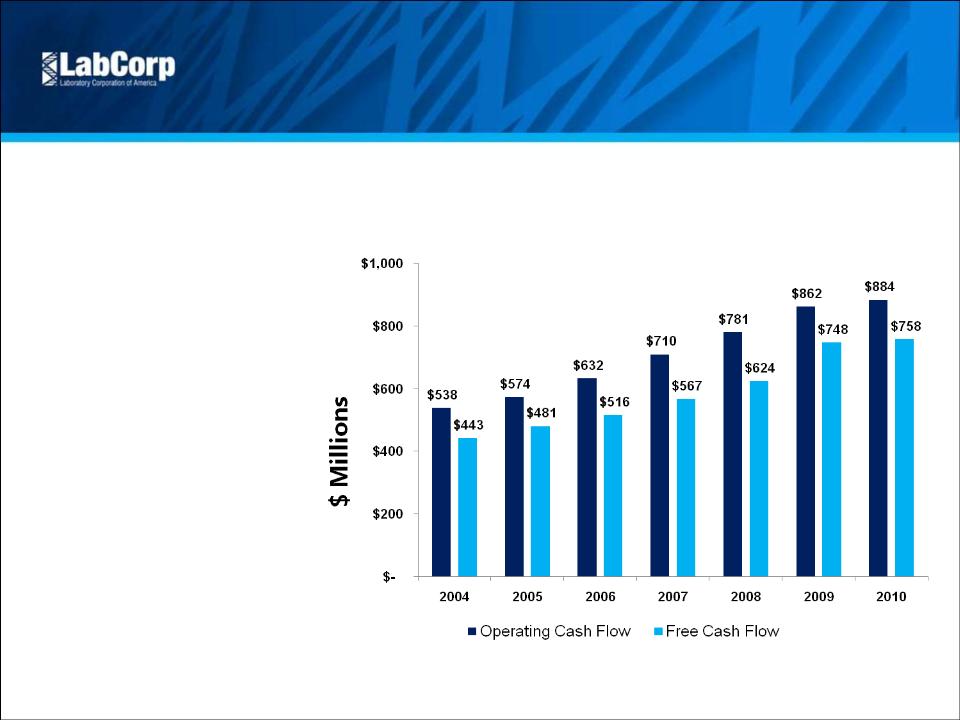
Operating Cash Flow (2) | $ 176.8 | $ 176.2 | 0.3% | $ 577.0 | $ 624.4 | -7.6% | |||||||||
Less: Capital Expenditures | $ (40.4) | $ (34.3) | 17.8% | $ (115.6) | $ (93.3) | 23.9% | |||||||||
Free Cash Flow | $ 136.4 | $ 141.9 | -3.9% | $ 461.4 | $ 531.1 | -13.1% | |||||||||
(1) See Reconciliation of non-GAAP Financial Measures (included herein) | |||||||||||||||
(2) Operating Cash Flow was reduced by $49.5 million as a result of the Hunter Labs settlement | |||||||||||||||
Operating Cash Flow | $ 176.8 | $ 577.0 | |||||||||||||
Hunter Labs settlement | $ 49.5 | $ 49.5 | |||||||||||||
Adjusted Operating Cash Flow | $ 226.3 | $ 626.5 | |||||||||||||


Reconciliation of non-GAAP Financial Measures | |||||
(In millions, except per share data) | |||||
Three Months Ended Sep 30, | |||||
Adjusted Operating Income | 2011 | 2010 | |||
Operating income | $ 239.4 | $ 235.3 | |||
Restructuring and other special charges (1) (2) | 24.1 | 14.8 | |||
Adjusted operating income | $ 263.5 | $ 250.1 | |||
Adjusted EPS Excluding Amortization | |||||
Diluted earnings per common share | $ 1.31 | $ 1.34 | |||
Impact of restructuring and other special charges (1) (2) | 0.17 | 0.13 | |||
Amortization expense | 0.13 | 0.11 | |||
Adjusted EPS Excluding Amortization (3) | $ 1.61 | $ 1.58 | |||
1) During the third quarter of 2011, the Company recorded net restructuring and other special charges of $24.1 million, consisting of $7.9 million in severance related liabilities and $16.2 million in net facility-related costs primarily associated with ongoing integration of the Clearstone, Genzyme Genetics and Westcliff acquisitions. The after tax impact of these charges decreased net earnings for the three months ended September 30, 2011, by $16.9 million and diluted earnings per share by $0.17 ($16.9 million divided by 102.2 million shares). |
During the first two quarters of 2011, the Company recorded restructuring and other special charges of $81.8 million. The restructuring charges included $10.9 million in net severance and other personnel costs along with $20.5 million in net facility-related costs primarily associated with the ongoing integration of the Genzyme Genetics and Westcliff acquisitions. The special charges also include $34.5 million ($49.5 million, net of previously recorded reserves of $15.0 million) relating to the settlement of the Hunter Labs litigation in California, along with $1.1 million for legal costs associated with the planned acquisition of Orchid Cellmark incurred during the second quarter of 2011, both of which were recorded in Selling, General and Administrative Expenses in the Company’s Statement of Operations. The charges also included a $14.8 million write-off of an investment made in a prior year. |
For the nine months ended September 30, 2011, the after tax impact of these combined charges decreased net earnings by $66.3 million and diluted earnings per share by $0.65 ($66.3 million divided by 102.3 million shares). |
2) During the third quarter of 2010, the Company recorded restructuring and other special charges of $21.8 million, consisting of $10.9 million in professional fees and expenses associated with acquisitions; $7.0 million in bridge financing fees associated with the signing of an asset purchase agreement for Genzyme Genetics; and $3.9 million in severance related liabilities associated with workforce reduction initiatives. The after tax impact of these charges decreased net earnings for the three months ended September 30, 2010, by $13.4 million and diluted earnings per share by $0.13 ($13.4 million divided by 104.1 million shares). |
During the first quarter of 2010, the Company recorded net charges of $9.3 million relating to severance payments and the closing of redundant and underutilized facilities as well as the write-off of development costs incurred on systems abandoned during the quarter. |
For the nine months ended September 30, 2010, the after tax impact of these combined charges decreased net earnings by $19.1 million and diluted earnings per share by $0.18 ($19.1 million divided by 105.4 million shares). |
3) The Company continues to grow its business through acquisitions and uses Adjusted EPS Excluding Amortization as a measure of operational performance, growth and shareholder returns. The Company believes adjusting EPS for amortization will provide investors with better insight into the operating performance of the business. For the quarters ended September 30, 2011 and 2010, intangible amortization was $21.2 million and $18.0 million, respectively ($13.0 million and $11.0 million net of tax, respectively) and decreased EPS by $0.13 ($13.0 million divided by 102.2 million shares) and $0.11 ($11.0 million divided by 104.1 million shares), respectively. For the nine months ended September 30, 2011 and 2010, intangible amortization was $64.6 million and $53.1 million respectively ($39.5 million and $32.5 million net of tax, respectively) and decreased EPS by $0.39 ($39.5 million divided by 102.3 million shares) and $0.31 ($32.5 million divided by 105.4 million shares), respectively. |

Reconciliation of non-GAAP Financial Measures | |||||
(In millions, except per share data) | |||||
Nine Months Ended Sep 30, | |||||
Adjusted Operating Income | 2011 | 2010 | |||
Operating income | $ 700.9 | $ 740.0 | |||
Restructuring and other special charges (1) (2) | 105.9 | 24.1 | |||
Adjusted operating income | $ 806.8 | $ 764.1 | |||
Adjusted EPS Excluding Amortization | |||||
Diluted earnings per common share | $ 3.76 | $ 4.05 | |||
Impact of restructuring and other special charges (1) (2) | 0.65 | 0.18 | |||
Amortization expense | 0.39 | 0.31 | |||
Adjusted EPS Excluding Amortization (3) | $ 4.80 | $ 4.54 | |||
1) During the third quarter of 2011, the Company recorded net restructuring and other special charges of $24.1 million, consisting of $7.9 million in severance related liabilities and $16.2 million in net facility-related costs primarily associated with ongoing integration of the Clearstone, Genzyme Genetics and Westcliff acquisitions. The after tax impact of these charges decreased net earnings for the three months ended September 30, 2011, by $16.9 million and diluted earnings per share by $0.17 ($16.9 million divided by 102.2 million shares). |
During the first two quarters of 2011, the Company recorded restructuring and other special charges of $81.8 million. The restructuring charges included $10.9 million in net severance and other personnel costs along with $20.5 million in net facility-related costs primarily associated with the ongoing integration of the Genzyme Genetics and Westcliff acquisitions. The special charges also include $34.5 million ($49.5 million, net of previously recorded reserves of $15.0 million) relating to the settlement of the Hunter Labs litigation in California, along with $1.1 million for legal costs associated with the planned acquisition of Orchid Cellmark incurred during the second quarter of 2011, both of which were recorded in Selling, General and Administrative Expenses in the Company’s Statement of Operations. The charges also included a $14.8 million write-off of an investment made in a prior year. |
For the nine months ended September 30, 2011, the after tax impact of these combined charges decreased net earnings by $66.3 million and diluted earnings per share by $0.65 ($66.3 million divided by 102.3 million shares). |
2) During the third quarter of 2010, the Company recorded restructuring and other special charges of $21.8 million, consisting of $10.9 million in professional fees and expenses associated with acquisitions; $7.0 million in bridge financing fees associated with the signing of an asset purchase agreement for Genzyme Genetics; and $3.9 million in severance related liabilities associated with workforce reduction initiatives. The after tax impact of these charges decreased net earnings for the three months ended September 30, 2010, by $13.4 million and diluted earnings per share by $0.13 ($13.4 million divided by 104.1 million shares). |
During the first quarter of 2010, the Company recorded net charges of $9.3 million relating to severance payments and the closing of redundant and underutilized facilities as well as the write-off of development costs incurred on systems abandoned during the quarter. |
For the nine months ended September 30, 2010, the after tax impact of these combined charges decreased net earnings by $19.1 million and diluted earnings per share by $0.18 ($19.1 million divided by 105.4 million shares). |
3) The Company continues to grow its business through acquisitions and uses Adjusted EPS Excluding Amortization as a measure of operational performance, growth and shareholder returns. The Company believes adjusting EPS for amortization will provide investors with better insight into the operating performance of the business. For the quarters ended September 30, 2011 and 2010, intangible amortization was $21.2 million and $18.0 million, respectively ($13.0 million and $11.0 million net of tax, respectively) and decreased EPS by $0.13 ($13.0 million divided by 102.2 million shares) and $0.11 ($11.0 million divided by 104.1 million shares), respectively. For the nine months ended September 30, 2011 and 2010, intangible amortization was $64.6 million and $53.1 million respectively ($39.5 million and $32.5 million net of tax, respectively) and decreased EPS by $0.39 ($39.5 million divided by 102.3 million shares) and $0.31 ($32.5 million divided by 105.4 million shares), respectively. |

Laboratory Corporation of America | ||||||||||||||||||||||
Other Financial Information | ||||||||||||||||||||||
FY 2009, FY 2010 and Q1-Q3 2011 | ||||||||||||||||||||||
Q1 09 | Q2 09 | Q3 09 | Q4 09 | Q1 10 | Q2 10 | Q3 10 | Q4 10 | Q1 11 | Q2 11 | Q3 11 | ||||||||||||
Bad debt as a percentage of sales | 5.3% | 5.3% | 5.3% | 5.3% | 5.0% | 4.8% | 4.8% | 4.7% | 4.7% | 4.7% | 4.5% | |||||||||||
Days sales outstanding | 52 | 50 | 48 | 44 | 46 | 45 | 44 | 46 | 47 | 46 | 46 | |||||||||||
A/R coverage (Allow. for Doubtful Accts. / A/R) | 19.5% | 20.6% | 21.9% | 23.2% | 21.7% | 20.7% | 20.4% | 18.5% | 19.4% | 20.6% | 21.1% | |||||||||||
