Exhibit 99.1
Shire RandD Day Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Shire RandD Day Jeff Poulton, Head of Investor Relations Our purpose We enable people with life-altering conditions to lead [GRAPHIC OMITTED] |  |
The "SAFE HARBOR" Statement Under the
Private Securities Litigation Reform Act of 1995
Statements included in this announcement that are not
historical facts are forward-looking statements.
Forward-looking statements can be identified by words such
as "aspiration", "will", "expect", "forecast", "aspiration",
"potential", "estimates", "may", "anticipate", "target",
"project" or similar expressions suitable for identifying
information that refers to future events. Forward-looking
statements involve a number of risks and uncertainties and
are subject to change at any time. In the event such risks
or uncertainties materialize, Shire's results could be
materially adversely affected. The risks and uncertainties
include, but are not limited to, that:
[] Shire's products may not be a commercial success;
[] revenues from ADDERALL XR are subject to generic
erosion and revenues from INTUNIV will become subject
to generic competition starting in December 2014;
[] the failure to obtain and maintain reimbursement, or an
adequate level of reimbursement, by third-party payors
in a timely manner for Shire's products may impact
future revenues, financial condition and results of
operations;
[] Shire conducts its own manufacturing operations for
certain of its products and is reliant on third party
contractors to manufacture other products and to
provide goods and services. Some of Shire's products or
ingredients are only available from a single approved
source for manufacture. Any disruption to the supply
chain for any of Shire's products may result in Shire
being unable to continue marketing or developing a
product or may result in Shire being unable to do so on
a commercially viable basis for some period of time.
[] the development, approval and manufacturing of Shire's
products is subject to extensive oversight by various
regulatory agencies. Submission of an application for
regulatory approval of any of our product candidates,
such as our planned submission of a New Drug
Application to the FDA for Lifitegrast as a treatment
for the signs and symptoms of dry eye disease in
adults, may be delayed for any number of reasons and,
once submitted, may be subjected to lengthy review and
ultimately rejected. Moreover, regulatory approvals or
interventions associated with changes to manufacturing
sites, ingredients or manufacturing processes could
lead to significant delays, increase in operating
costs, lost product sales, an interruption of research
activities or the delay of new product launches;
[] the actions of certain customers could affect Shire's
ability to sell or market products profitably.
Fluctuations in buying or distribution patterns by such
customers can adversely impact Shire's revenues,
financial conditions or results of operations;
[] investigations or enforcement action by regulatory
authorities or law enforcement agencies relating to
Shire's activities in the highly regulated markets in
which it operates may result in the distraction of
senior management, significant legal costs and the
payment of substantial compensation or fines;
[] adverse outcomes in legal matters and other disputes,
including Shire's ability to enforce and defend patents
and other intellectual property rights required for its
business, could have a material adverse effect on
Shire's revenues, financial condition or results of
operations;
[] Shire faces intense competition for highly qualified
personnel from other companies, academic institutions,
government entities and other organizations. Shire is
undergoing a corporate reorganization and the
consequent uncertainty could adversely impact Shire's
ability to attract and/or retain the highly skilled
personnel needed for Shire to meet its strategic
objectives;
[] failure to achieve Shire's strategic objectives with
respect to the acquisition of ViroPharma Incorporated
may adversely affect Shire's financial condition and
results of operations; and other risks and
uncertainties detailed from time to time in Shire's
filings with the US Securities and Exchange Commission,
including its most recent Annual Report on Form 10-K.
|  |
Investor Day Agenda and Outline Time 8:00-8:30am 8:30-8:45am 8:45-9:25am 9:25-10:00am 10:00-10:45am 10:45-11:15am 11:15-11:45am 11:45-12:15pm 12:15-1:15pm 1:15-1:30pm 1:30-2:00pm 2:00-2:45pm 2:45-3:00pm 3:00-3:30pm 3:30-4:30pm Topic Registration/Breakfast Corporate Overview RandD Strategy Overview Research Overview and Technology Platforms (mRNA, Protein Replacement, Gene Therapy, Antibody Platforms) Rare Diseases: GI/Hepatology (SHP625 / LUM001, SHP626 / LUM002) Morning Break Rare Diseases: Ophthalmology (SHP607 / ROP, SHP630 / BIKAM) Morning QandA Lunch Rare Diseases: Complement Pathway (SHP616 / CINRYZE new uses) Rare Diseases: CNS (SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen) Late Stage Update (SHP606 / Lifitegrast, BED, SHP465 / ADHD) Program Wrap-Up Afternoon QandA Reception Speaker Flemming Ornskov, M.D., MPH Phil Vickers, Ph.D. Albert Seymour, Ph.D. Ciara Kennedy, Ph.D. David Piccoli, M.D. Norman Barton, M.D., Ph.D. Howard Mayer, M.D. Howard Mayer, M.D. Howard Mayer, M.D. Randy Brenner Joe Tauber, M.D. Phil Vickers, Ph.D. |  |
Corporate Overview Flemming Ornskov, M.D., MPH, Chief Executive Officer Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] 5 |  |
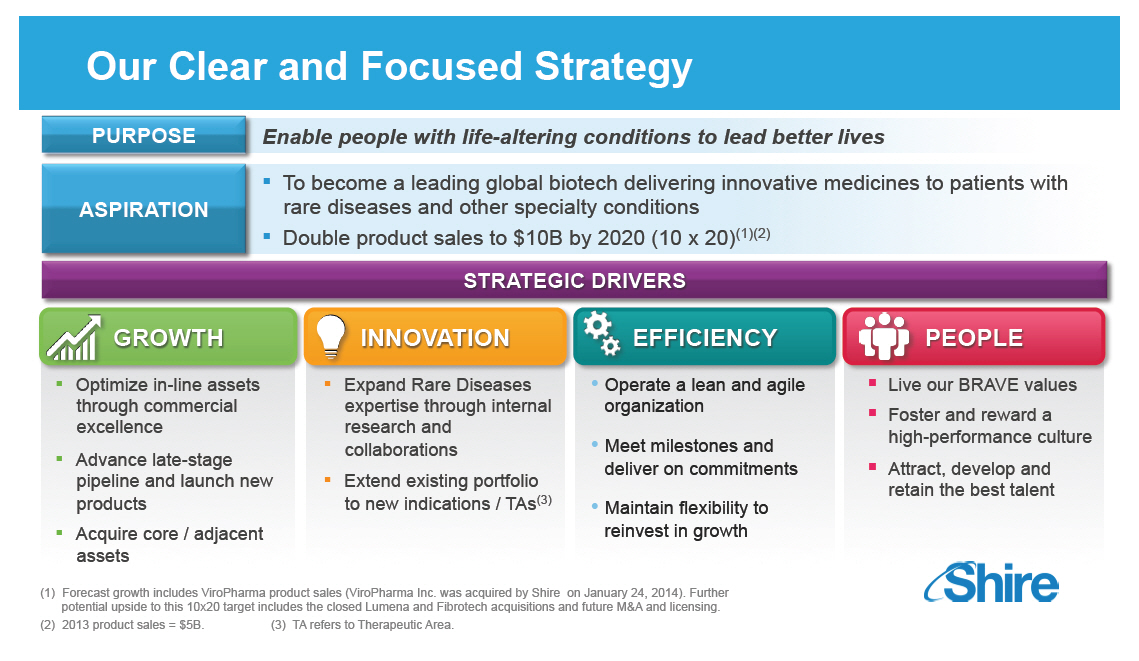
Our Clear and Focused Strategy
PURPOSE Enable people with life-altering conditions to lead
better lives
[] To become a leading global biotech delivering
innovative medicines to patients with ASPIRATION rare
diseases and other specialty conditions
[] Double product sales to $10B by 2020 (10 x 20)(1)(2)
STRATEGIC DRIVERS
GROWTH
[] Optimize in-line assets through commercial excellence
[] Advance late-stage pipeline and launch new products
[] Acquire core / adjacent assets
INNOVATION
[] Expand Rare Diseases expertise through internal
research and collaborations
[] Extend existing portfolio to new indications / TAs(3)
EFFICIENCY
[] Operate a lean and agile organization
[] Meet milestones and deliver on commitments
[] Maintain flexibility to reinvest in growth
PEOPLE
[] Live our BRAVE values
[] Foster and reward a high-performance culture
[] Attract, develop and retain the best talent
(1) Forecast growth includes ViroPharma
product sales
(ViroPharma Inc. was acquired by Shire on January 24,
2014). Further potential upside to this 10x20 target
includes the closed Lumena and Fibrotech acquisitions
and future MandA and licensing.
(2) 2013 product sales = $5B. (3) TA
refers to Therapeutic
Area.
[GRAPHIC OMITTED]
|  |
10 X 20: $10 BILLION IN PRODUCT SALES BY 2020 [GRAPHIC OMITTED] 10 x 20 Details In-Line: $7 billion expected from onmarket products Pipeline: $3 billion from existing pipeline Upside: Lumena, Fibrotech, BIKAM, early stage pipeline and future BD provide additional upside to 10 x 20 [GRAPHIC OMITTED] | 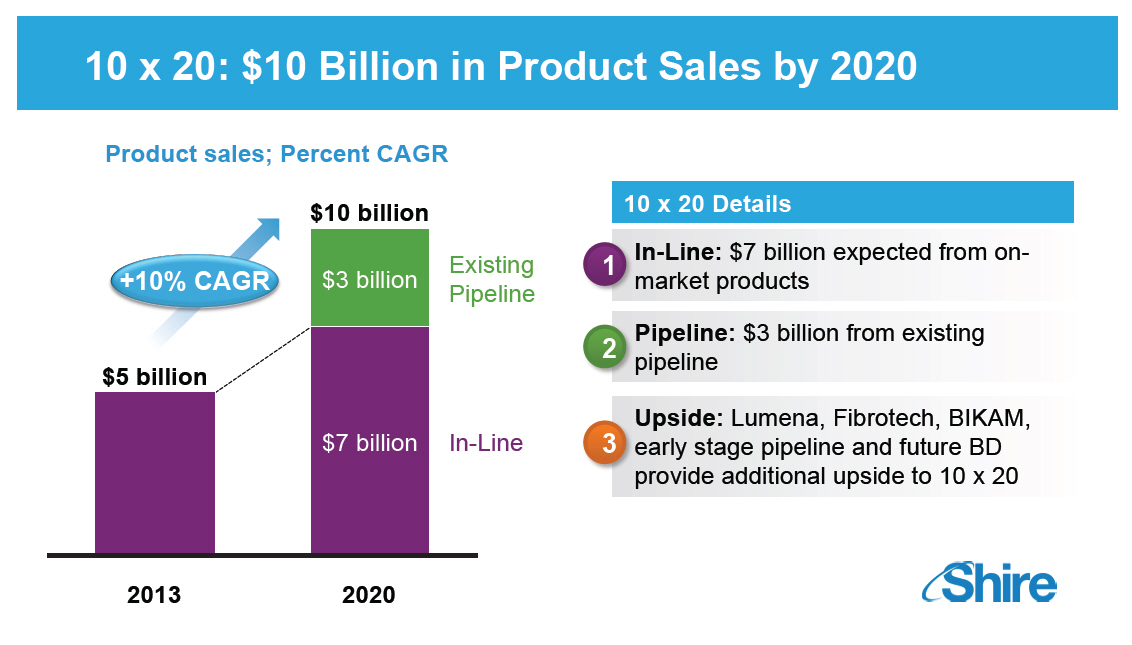 |
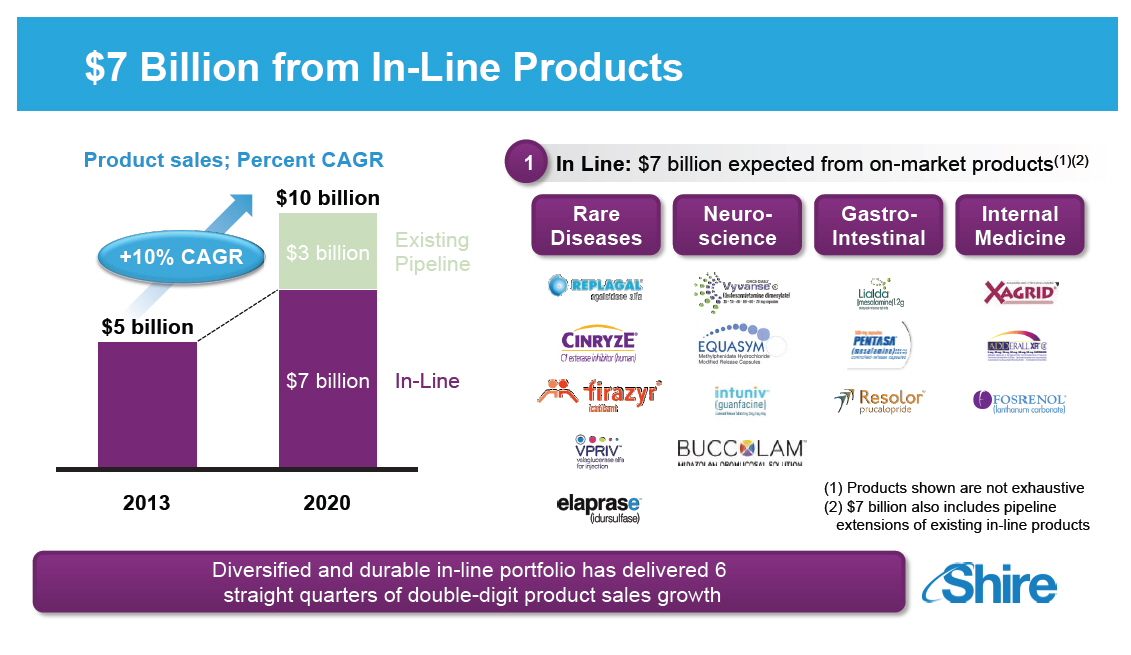
$7 Billion from In-Line Products
Product sales; Percent CAGR
[GRAPHIC OMITTED]
In Line: $7 billion expected 1 from on-market products(1)(2)
Rare
Diseases
Neuroscience
Gastro-
Intestinal
Internal
Medicine
[GRAPHIC OMITTED]
(1) Products shown are not exhaustive
(2) $7 billion also includes pipeline
extensions of
existing in-line products
[GRAPHIC OMITTED]
|  |
$3 Billion from Existing Pipeline [GRAPHIC OMITTED] Pipeline: $3 billion from existing pipeline Included in $3 Billion [GRAPHIC OMITTED] Balanced pipeline across stages of development with multiple upcoming milestones that will support long-term growth [GRAPHIC OMITTED] |  |
Upside from Recent and Future BD [GRAPHIC OMITTED] Upside: Lumena, Fibrotech, CINRYZE New Uses, early stage pipeline, and future BD provide additional upside to 10 x 20 Additional upside [GRAPHIC OMITTED] Rare Diseases domain expertise and significant cash generation creates an opportunity to become the industry's go-to-partner [GRAPHIC OMITTED] | 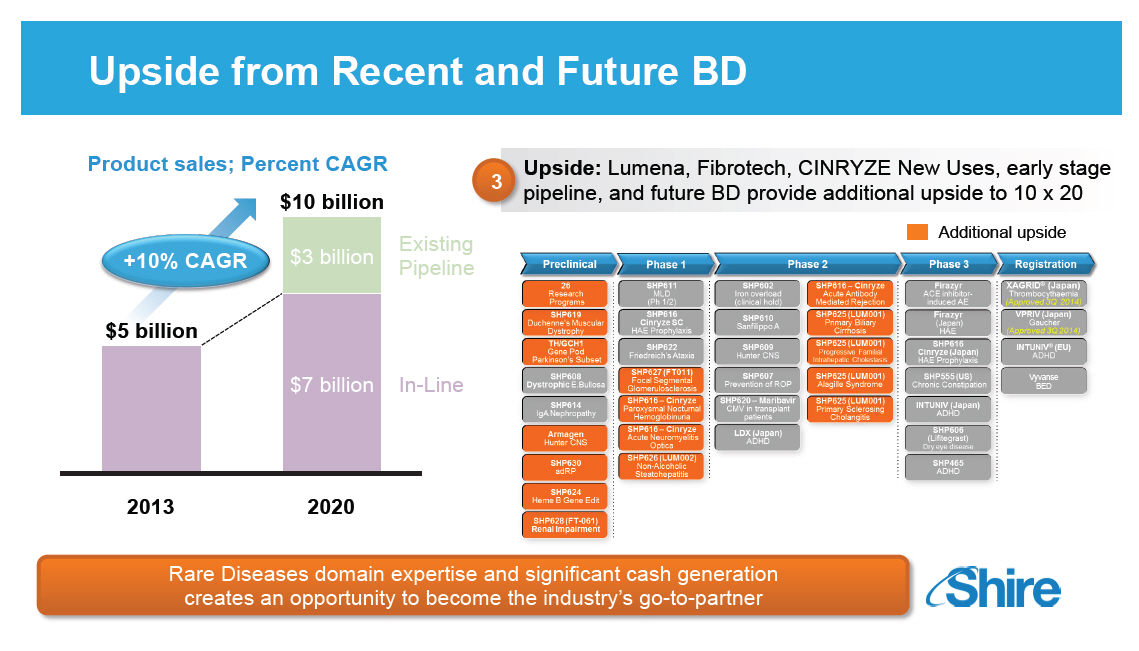 |
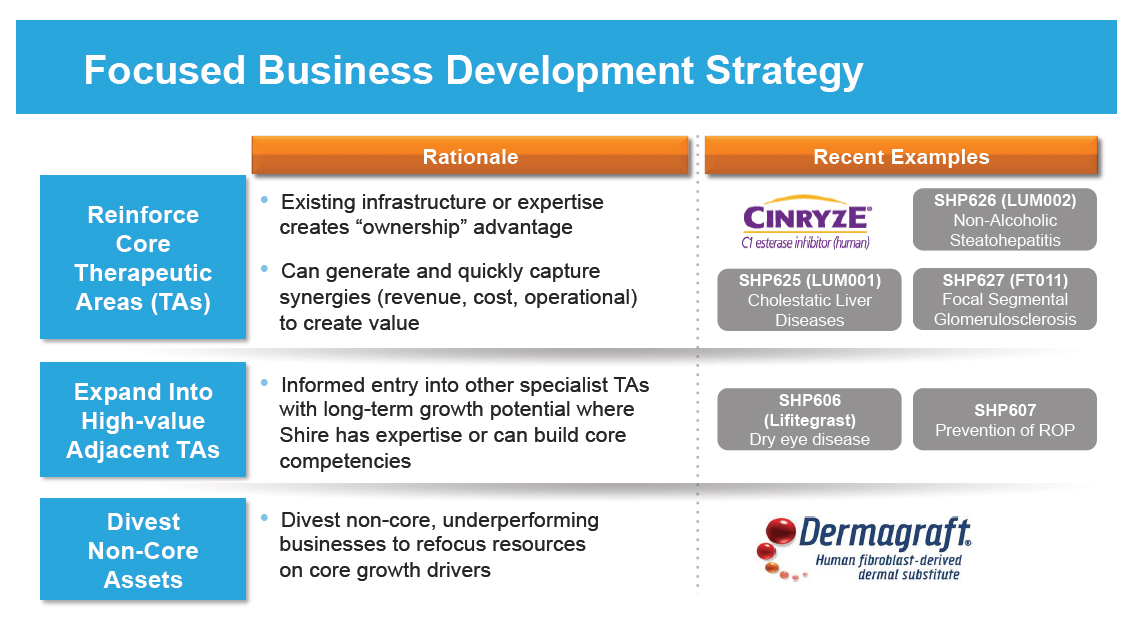
Focused Business Development Strategy
Rationale
Reinforce
Core
Therapeutic
Areas (TAs)
Expand Into
High-value
Adjacent TAs
Divest
Non-Core
Assets
o Existing infrastructure or expertise creates
"ownership" advantage
o Can generate and quickly capture synergies (revenue,
cost, operational) to create value
o Informed entry into other specialist TAs with long-term
growth potential where Shire has expertise or can build
core competencies
o Divest non-core, underperforming businesses to refocus
resources on core growth drivers
Recent Examples
[GRAPHIC OMITTED]
[GRAPHIC OMITTED]
|  |
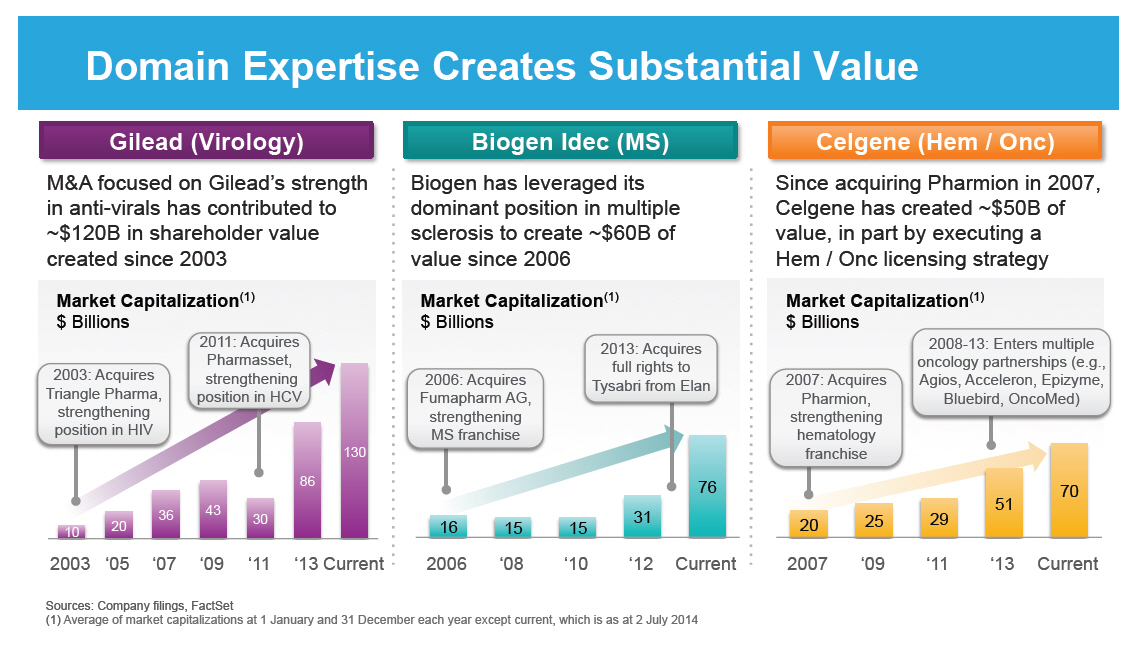
Domain Expertise Creates Substantial Value
Gilead (Virology)
M&A focused on Gilead's strength in anti-virals has
contributed to ~$120B in shareholder value created since
2003
Market Capitalization(1) $ Billions
[GRAPHIC OMITTED]
Biogen Idec (MS)
Biogen has leveraged its dominant position in multiple
sclerosis to create ~$60B of value since 2006
Market Capitalization(1)
$ Billions
[GRAPHIC OMITTED]
Celgene (Hem / Onc)
Since acquiring Pharmion in 2007, Celgene has created ~$50B
of value, in part by executing a Hem / Onc licensing
strategy
Market Capitalization(1)
$ Billions
[GRAPHIC OMITTED]
Sources: Company filings, FactSet
(1) Average of market capitalizations at
1 January
and 31 December each year except current, which
is as at 2 July 2014
|  |
Research and Development Pipeline Pre-One Shire Preclinical 46 Research Programs SPD604 GI SPD605 ADHD Phase 1 SPD535 ESRD SHP610 SanA SHP611 MLD SPD559 ADHD SPD554 ADHD Phase 2 SHP613 Acute Vascular Repair SHP609 Hunter CNS SHP602 Iron overload SPD557 GERD LDX NSS SPD554 ASD XAGRID(R) (Japan) 4510 DMD RESOLOR(R) (US) Chronic Constipation INTUNIV (Japan) ADHD Phase 3 DERMAGRAFT VLU ABH001 EB FIRAZYR ACE inhibitor-induced AE VIPRIV Japan Vyvanse BED Registration [GRAPHIC OMITTED] |  |
Preclinical 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 Heme B Gene Edit SHP628 (FT-061) Renal Impairment Phase 1 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze ) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis Phase 2 SHP602 Iron overload (clinical hold) SHP610 Sanfilippo A SHP609 Hunter CNS SHP607 Prevention of ROP SHP620 (Maribavir) CMV in transplant patients LDX (Japan) ADHD SHP616 (Cinryze) Acute Antibody Mediated Rejection SHP625 (LUM001) Primary Biliary Cirrhosis SHP625 (LUM001) Progressive Familial Intrahepatic Cholestasis SHP625 (LUM001) Alagille Syndrome SHP625 (LUM001) Primary Sclerosing Cholangitis Phase 3 Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD Registration XAGRID(R) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(R) (EU) ADHD Vyvanse BED [GRAPHIC OMITTED] |  |
RandD Strategy Overview Phil Vickers, Ph.D., Global Head of RandD Our purpose We enable people with life-altering conditions to lead better lives. [GRAPHIC OMITTED] |  |
RandD Pipeline Has Never Been Stronger 22 programs in the clinic, the most in the history of Shire Well-positioned to deliver on '10 x 20' expectations Many significant clinical milestones in the next 18 months On track to file at least 2 INDs from internal programs every year Establishing talent and capabilities appropriate to drive future growth Continued excellence in acquiring external assets with a strong strategic fit Establishing a leadership position in the treatment of Rare Diseases [GRAPHIC OMITTED] 16 |  |
Our Clear and Focused Strategy STRATEGIC DRIVERS GROWTH []Optimize in-line assets through commercial excellence []Advance late-stage pipeline and launch new products []Acquire core / adjacent assets INNOVATION []Expand Rare Diseases expertise through internal research and collaborations []Extend existing portfolio to new indications / TAs EFFICIENCY []Operate a lean and agile organization []Meet milestones and deliver on commitments []Maintain flexibility to reinvest in growth PEOPLE []Foster and reward a high-performance culture []Attract, develop and retain the best talent []Live our BRAVE values [GRAPHIC OMITTED] 17 |  |
Our Clear and Focused Strategy STRATEGIC DRIVERS GROWTH []Optimize in-line assets through commercial excellence []Advance late-stage pipeline and launch new products []Acquire core / adjacent assets INNOVATION []Expand Rare Diseases expertise through internal research and collaborations []Extend existing portfolio to new indications / TAs EFFICIENCY []Operate a lean and agile organization []Meet milestones and deliver on commitments []Maintain flexibility to reinvest in growth PEOPLE []Foster and reward a high-performance culture []Attract, develop and retain the best talent []Live our BRAVE values [GRAPHIC OMITTED] 18 |  |
RandD Strategy to Deliver Growth
GROWTH
INNOVATION
FOCUS IN AREAS OF HIGH
UNMET MEDICAL NEED
FOCUS IN AREAS
OF HIGH VALUE TO
PATIENTS, PAYORS
AND SHAREHOLDERS
FOCUS ON INNOVATION
ENSURE HIGH
PRODUCTIVITY,
OPERATIONAL
EXCELLENCE
[] Increasing emphasis on rare diseases with high
morbidity and / or mortality
[] Focus on specific platforms
[] Adapting existing therapeutic areas to a rare-disease
focus
[] Strategic focus, but flexibility in considering new
opportunities
[] Alignment with all key stakeholders, including patients
[] Focus in areas which drive attractive commercial return
[] Health economics assessments to support market access
[] Prioritize programs where a single approach can address
multiple rare diseases (e.g. Lumena, Cinryze) or expand
to common indications
[] Innovation associated with every pipeline program
[] Risk balance across pipeline
[] Foster a culture that ensures innovation and calculated
risk-taking
[] Creative partnerships and collaborations
[] Balance of internal programs and acquiring external
assets
[] Ensure industry-leading capabilities in operational
excellence
[GRAPHIC OMITTED]
19
|  |
Main Therapeutic Areas Increasingly Focused on Rare Diseases GROWTH INNOVATION Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 (ALGS, PSC, PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT Rare Diseases Leadership Also evaluating opportunities in 'Rare Hematological' malignancies [GRAPHIC OMITTED] 20 |  |
Preclinical 46 Research Programs SPD604 GI SPD605 ADHD Phase 1 SPD535 ESRD SHP610 SanA SHP611 MLD SPD559 ADHD SPD554 ADHD Phase 2 SHP613 Acute Vascular Repair SHP609 Hunter CNS SHP602 Iron overload SPD557 GERD LDX NSS SPD554 ASD XAGRID(R) (Japan) 4510 DMD RESOLOR(R) (US) Chronic Constipation INTUNIV (Japan) ADHD Phase 3 Registration DERMAGRAFT VLU ABH001 EB FIRAZYR ACE inhibitor-induced AE VIPRIV Japan LDX MDD Vyvanse BED Registration [GRAPHIC OMITTED] | 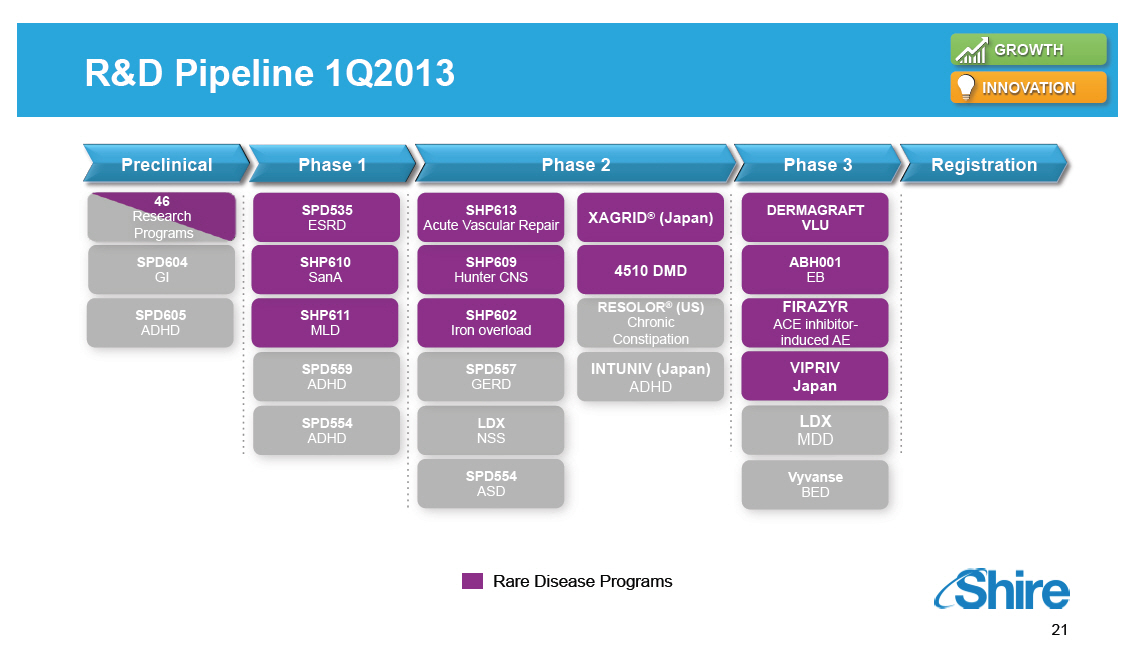 |
Preclinical 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 Heme B Gene Edit SHP628 (FT-061) Renal Impairment Phase 1 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis Phase 2 SHP602 Iron overload (clinical hold) SHP610 Sanfilippo A SHP609 Hunter CNS SHP607 Prevention of ROP SHP620 (Maribavir) CMV in transplant patients LDX (Japan) ADHD Phase 3 Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD Registration XAGRID(R) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(R) (EU) ADHD Vyvanse BED [GRAPHIC OMITTED] |  |
Recent Changes to Internal Programs Increase
Portfolio Value
GROWTH
INNOVATION
Rare Diseases
Platform
[] Reinforced Rare Diseases leadership through internal
research and partnerships with world-class
organizations while extending the platform to new
indications and adapting current TAs
[] Future rare diseases strategy defined
Intrathecal
Programs
[] Progressed intrathecal programs to treat CNS
manifestations of Hunter, Sanfilippo A and MLD
[] Novel intrathecal device approved for use in US and EU
SHP465
ADHD
[] Planning to resubmit SHP465 for high- growth ADHD adult
segment
Vyvanse
BED
[] Positive Phase 3 results
[] Filed NDA in 3Q14
[] PDUFA date Feb 2015
[] Expect to launch in 1H15 subject to FDA review
[] Currently no approved Binge Eating Disorder treatments
Rationalization
[] Halted programs that did not fit scientific, strategic
or commercial criteria
[] Discovery programs now focused on rare diseases
[GRAPHIC OMITTED]
23
|  |
Recent Acquisitions Have Significant Potential
GROWTH
INNOVATION
[GRAPHIC OMITTED]
[] Acquired Viropharma 1Q14
[] Cinryze low volume SC program for HAE prophylaxis
(Phase 1) complement IV program
[] SHP620 (Maribavir) being developed for treatment of
cytomegalovirus infection in transplant patients (Phase
2)
[] Cinryze new use programs: PNH (Phase 1), NMO (Phase 1),
AMR (Phase 2)
[GRAPHIC OMITTED]
[] Acquired SARCode 2Q13
[] Lifitegrast being evaluated for signs and symptoms of
Dry Eye Disease
[] No currently approved product treats signs and symptoms
of disease
[] Plan to file NDA in 1Q15
[GRAPHIC OMITTED]
[] Acquired Lumena 2Q14
[] SHP625 (LUM001) for 4 rare cholestatic liver diseases
(Phase 2)
[] SHP626 (LUM002) for NASH (Phase 1)
[GRAPHIC OMITTED]
[] Acquired Premacure 1Q13
[] SHP607 (Premiplex) novel protein replacement therapy
with potential to prevent retinopathy of prematurity
[] Clinical program paused to allow optimization during
2014
[] Ongoing Phase 2 study, due to read out 2015
[GRAPHIC OMITTED]
[] Acquired Fibrotech 3Q14
[] SHP627 (FT011) antifibrotic agent with potential for
Focal Segmental Glomerulosclerosis (Phase 1 completed)
[] SHP628 (FT061)) with potential for renal impairment
(Preclinical)
[] Fibrotechs library of novel molecules targeting
fibrosis
[GRAPHIC OMITTED]
[] Acquired Lotus Tissue Repair 1Q13
[] SHP608 (recombinant collagen 7) novel protein
replacement therapy with potential for Dystrophic
Epidermolysis Bullosa
[GRAPHIC OMITTED]
[] AGT-182 licensed from Armagen in 3Q14
[] Molecular 'Trojan Horse' of ERT fused to human insulin
receptor designed to treat severe Hunter syndrome -
facilitates entry into CNS
[] Currently in preclinical development
[GRAPHIC OMITTED]
[] Acquired BIKAM 3Q14
[] SHP 630 is a pharmacological chaperone designed to
treat autosomal- dominant retinitis pigmentosa (adRP)
[] SHP 630 facilitates opsin folding, with the aim of
restoring retinal function in adRP
[] Currently in preclinical development
[GRAPHIC OMITTED]
24
|  |
Upcoming Anticipated Pipeline Milestones GROWTH INNOVATION Registration and Phase 4 Phase 3 Phase 2 Phase 1/2 SHP611 MLD Phase 1/2 head line data SHP625 (LUM001) ALGS Phase 2 head line data SHP606 Lifitegrast NDA Filing SHP625 (LUM001) PSC Phase 2 head line data SHP607 ROP Phase 2 head line data SHP606 Lifitegrast Launch(2) SHP609 Phase 2/3 head line data 20152015 VYVANSE BED Launch(2) SHP620 Maribavir Phase 2 head line data SHP625 (LUM001) PBC Phase 2 head line data SHP625 (LUM001) ALGS Phase 2 head line data 2016 SHP625 (LUM001) (1) PFIC Phase 2 head line data SHP610 Phase 2B head line data Notes (1)[] Interim 625 PFIC INDIGO data expected Q2 2015. (2)[] Subject to regulatory approval. [GRAPHIC OMITTED] 25 | 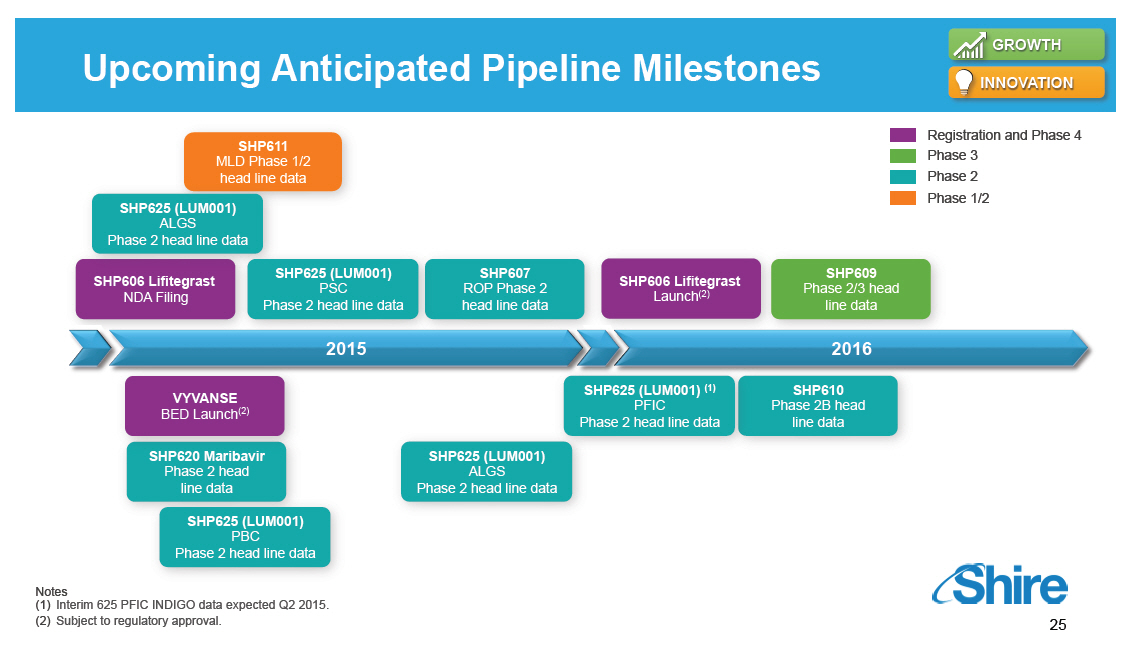 |
Our Clear and Focused Strategy STRATEGIC DRIVERS GROWTH []Optimize in-line assets through commercial excellence []Advance late-stage pipeline and launch new products []Acquire core / adjacent assets INNOVATION []Expand Rare Diseases expertise through internal research and collaborations []Extend existing portfolio to new indications / TAs(1) EFFICIENCY []Operate a lean and agile organization []Meet milestones and deliver on commitments []Maintain flexibility to reinvest in growth PEOPLE []Foster and reward a high-performance culture []Attract, develop and retain the best talent []Live our BRAVE values (1)[] TA refers to Therapeutic Area. [GRAPHIC OMITTED] 26 |  |
Unifying the Organization to Drive Efficiency EFFICIENCY 3 SEPARATE RandD UNITSFROM [] ONE INTEGRATED RandD ORG TO [] ONE Structure | Culture | Purpose [GRAPHIC OMITTED] [GRAPHIC OMITTED] 27 |  |
Effective Decision Making in RandD
EFFICIENCY
PEOPLE
Science and Technology Committee
Composition: Subset of board members with scientific
backgrounds
Role: Reviews scientific aspects of RandD and pipeline
progress
[] Evaluates in-licensing and acquisition opportunities
[] Reviews internal pipeline programs and assesses RandD
talent
[] Provides view to the full board of directors on the
above
Research Steering Committee
Composition: Internal stakeholders
responsible for transition from "R" to "D"
Role: Makes "go / no-go" decisions on
internal research programs
[] Reviews research program progress and manages to go /
no-go decisions
[] Engages externally for input (e.g. payors)
[] Makes recommendations to Pipeline Committee
Pipeline Committee
Composition: Internal stakeholders
responsible for the pipeline
Role: Makes major decisions on programs
in the development pipeline
[] Makes recommendations to the Board whether to pursue
external opportunities
[] Aligns on an RandD strategy and priorities
[] 'One stop shopping' for all major pipeline decisions
Scientific Advisory Board
Composition: External experts in science relevant to the
RandD pipeline
Role: Provides external perspective into science
underpinning RandD programs
[] Contributes independent advice on science and
technology
[] Provides ideas and challenges based on their experience
[GRAPHIC OMITTED]
28
|  |
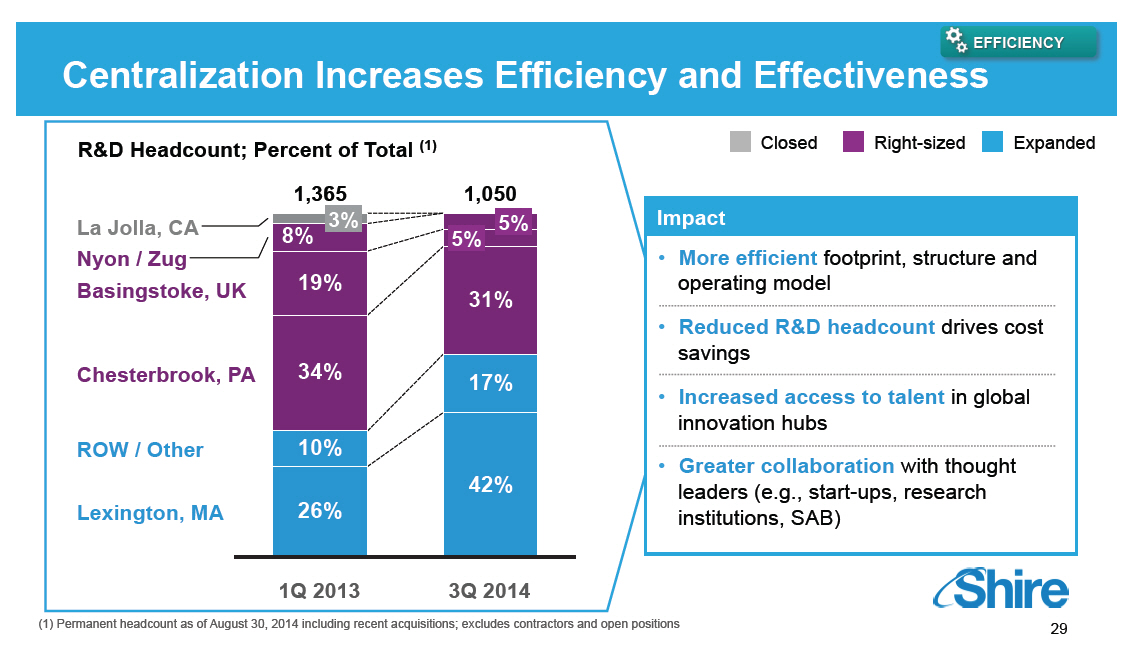
o More efficient footprint, structure and operating model
o Reduced R&D headcount drives cost savings
o Increased access to talent in global innovation hubs
o Greater collaboration with thought leaders (e.g.,
start-ups, research institutions, SAB)
[GRAPHIC OMITTED]
29
|  |
RandD is Supported by a Network of Alliances and Creative Collaborations EFFICIENCY [GRAPHIC OMITTED] [GRAPHIC OMITTED] 30 |  |
Experienced and Talented Team PEOPLE Presenting today [GRAPHIC OMITTED] Phil Vickers [] Head of RandD [] Former Merck, Pfizer, BI, Resolvyx [GRAPHIC OMITTED] Howard Mayer [] Head of Clinical Development [] Former CMO Merck Serono, ex-Pfizer [GRAPHIC OMITTED] Ciara Kennedy [] Head of ex-Lumena Programs [] Former Lumena COO [GRAPHIC OMITTED] Mike Heartlein [] Head of MRT Program [] Responsible for 3 marketed ERTs [GRAPHIC OMITTED] Norman Barton [] Head of ROP Program [] Responsible for industry's first ERT [GRAPHIC OMITTED] Albert Seymour [] Head of Research and Non-Clinical Development [] Former Pfizer Rare Diseases, Head of Human Genetics [GRAPHIC OMITTED] Randy Brenner [] Head of Regulatory Affairs [] Former Pfizer Head of Regulatory, Emerging Markets [GRAPHIC OMITTED] Rekha Abichandani [] Head of Intrathecal ERT Programs [] Former Genzyme, Rare Diseases and Hematological Malignancies [GRAPHIC OMITTED] Clark Pan [] Head of Discovery Therapeutics [] Former Genzyme, Head of Protein Engineering [GRAPHIC OMITTED] Jennifer Schranz [] Head of HAE Programs [] Former VP of Clinical Research, Viropharma [GRAPHIC OMITTED] 31 |  |
Positioned for Leadership in Rare Diseases PEOPLE Specific Rare Diseases RandD Challenges Research Clinical / Regulatory Medical Affairs / Post-Marketing [] Few scientific experts [] Incomplete knowledge of pathophysiology [] Genotype-phenotype unclear [] Inefficient diagnosis [] Few patients, geographically dispersed [] Clinical endpoints unclear [] Need for natural history studies [] Challenges with placebo-controlled studies [] Need for medical education [] Post-marketing commitments [] Patient registries / outcome surveys [] Need for early access programs [] Charitable access programs [] Health economics challenges Shire RandD Expertise [] Strong links to key opinion leaders [] Research grants and partnerships [] Ground-breaking RandD [] Clinical operations focus on specific challenges to open sites in novel therapeutic areas and recruit patients with rare diseases [] Early engagement with regulatory authorities [] Support for investigator-sponsored trials [] Natural history studies [] Strong links to patient associations [] Numerous early access programs [] Numerous registries / outcome surveys [] Health economics focus [GRAPHIC OMITTED] 32 |  |
Sessions for the Remainder of Today [GRAPHIC OMITTED] Topic Research Overview and Technology Platforms mRNA, Protein Replacement, Gene Therapy, Antibody Platforms Rare Diseases: GI / Metabolic SHP625 (LUM001), SHP626 (LUM002) Rare Diseases: Ophthalmics SHP607 / ROP, SHP630 / BIKAM Rare Diseases: Complement Biology and Renal / Fibrotic SHP616 / Cinryze new uses Rare Diseases: CNS SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen Late-Stage Update SHP606 / Lifitegrast, BED, SHP465 / ADHD Speaker Albert Seymour, Ph.D. Ciara Kennedy, Ph.D. David Piccoli, M.D. Norman Barton, M.D., Ph.D. Howard Mayer, M.D. Howard Mayer, M.D. Howard Mayer, M.D. Randy Brenner Joe Tauber, M.D. Time (EST) 9:25-10:00 10:00-10.45 11:15-11:45 1:15-1:30 1:30-2:00 2:00-2:45 [GRAPHIC OMITTED] 33 |  |
Research Overview and Technology Platforms Albert Seymour, PhD, Head of Research and Nonclinical Development Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Today's RandD Sessions [GRAPHIC OMITTED] Topic Research Overview and Technology Platforms mRNA, Protein Replacement, Gene Therapy, Antibody Platforms Rare Diseases: GI / Metabolic SHP625 (LUM001), SHP626 (LUM002) Rare Diseases: Ophthalmics SHP607 / ROP, SHP630 / BIKAM Rare Diseases: Complement Biology and Renal / Fibrotic SHP616 / Cinryze new uses Rare Diseases: CNS SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen Late-Stage Update SHP606 / Lifitegrast, BED, SHP465 / ADHD Speaker Albert Seymour, PhD Ciara Kennedy, PhD David Piccoli, MD Norman Barton, MD, PhD Howard Mayer, MD Howard Mayer, MD Howard Mayer, MD Randall Brenner Joe Tauber, MD Time (EST) 9:25-10:00 10:00-10.45 11:15-11:45 1:15-1:30 1:30-2:00 2:00-2:45 [GRAPHIC OMITTED] 35 |  |
Rare Diseases Leadership Underpins Multiple TAs Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 [] (ALGS, PSC, [] PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP 555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT Rare Diseases Leadership [GRAPHIC OMITTED] 36 | 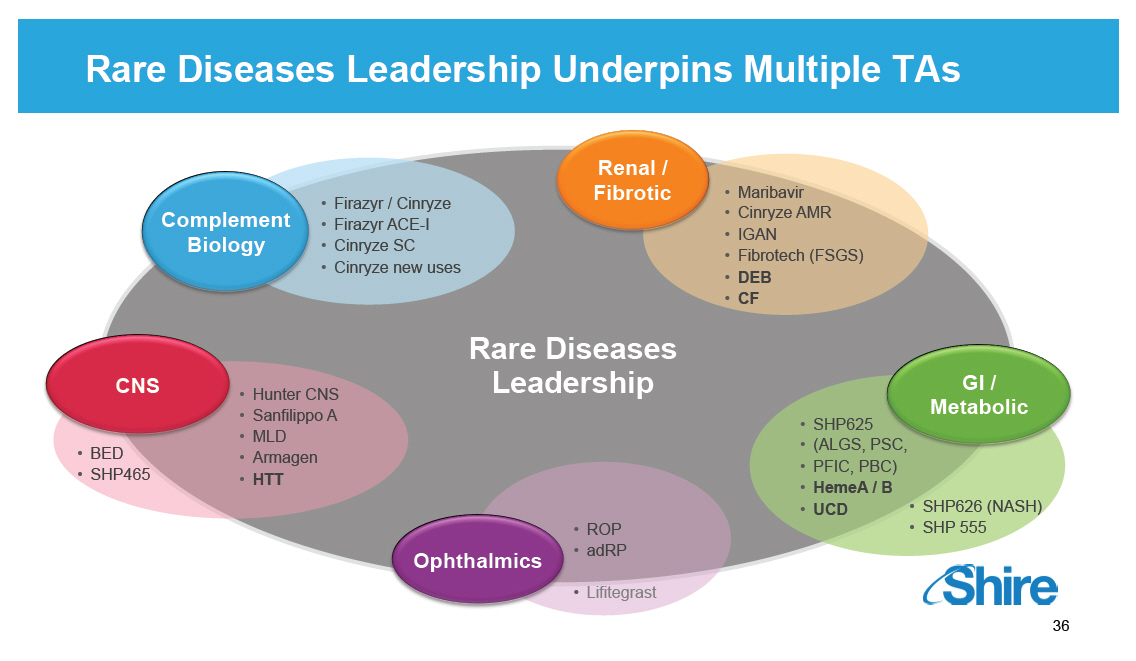 |
Genetic-based Drug Discovery and Development [GRAPHIC OMITTED] [GRAPHIC OMITTED] Drug Discovery and Early Development Patients and Families [GRAPHIC OMITTED] Description of Phenotype and Unmet Medical Need [GRAPHIC OMITTED] Identification of Underlying Genes and Disease Biology [GRAPHIC OMITTED] [GRAPHIC OMITTED] Characterizing Mechanism of Disease Biology Genetic Research Cycle [GRAPHIC OMITTED] 37 |  |
Research Model for Delivering the Portfolio in Rare Diseases
TIGEM
BCH
Research ventures
Internal
BD/Alliance
Access to tool
reagents to test
mechanism
Pre-defined
criteria
Data driven
decisions
DEB
HEMB
RP
Hunter CNS
CF
UCD
HTT
1. Research portfolio built around high confidence targets
in diseases with significant unmet medical need
2. Culture of rapid and clear data driven go/no go
decisions
3. Efficient and focused team integrating internal and
external flexible model to deliver early alignment with
process and clinical development
4. ~26 active programs with a goal of delivering 3
programs from research to early development per year
with a focus on quick to clinical POC
[GRAPHIC OMITTED]
38
|  |
Identifying Rare Diseases Opportunities [GRAPHIC OMITTED] ELICIT PHARMACOLOGY TARGET ENGAGED EXPOSURE IN TARGET TISSUE [GRAPHIC OMITTED] UNMET MEDICAL NEED [GRAPHIC OMITTED] 39 |  |
External Innovation is Core to Research [GRAPHIC OMITTED] [GRAPHIC OMITTED] Early Stage Opportunities [] Translate existing science into diseases of interest [] Develop lead assets [] Build relationships [GRAPHIC OMITTED] Co-Investment [] Externally fund programs and manage research and develop [] Create novel investment structures [GRAPHIC OMITTED] 40 | 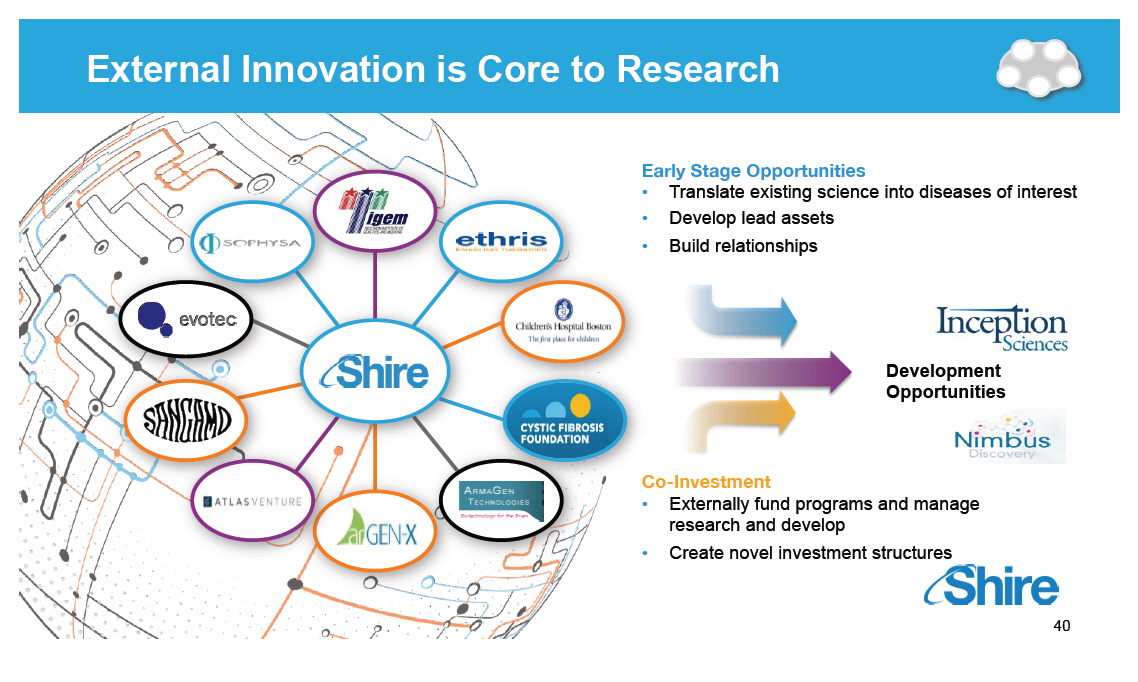 |
Rare Disease Leadership through Innovative Technology Platforms - Internal and External [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 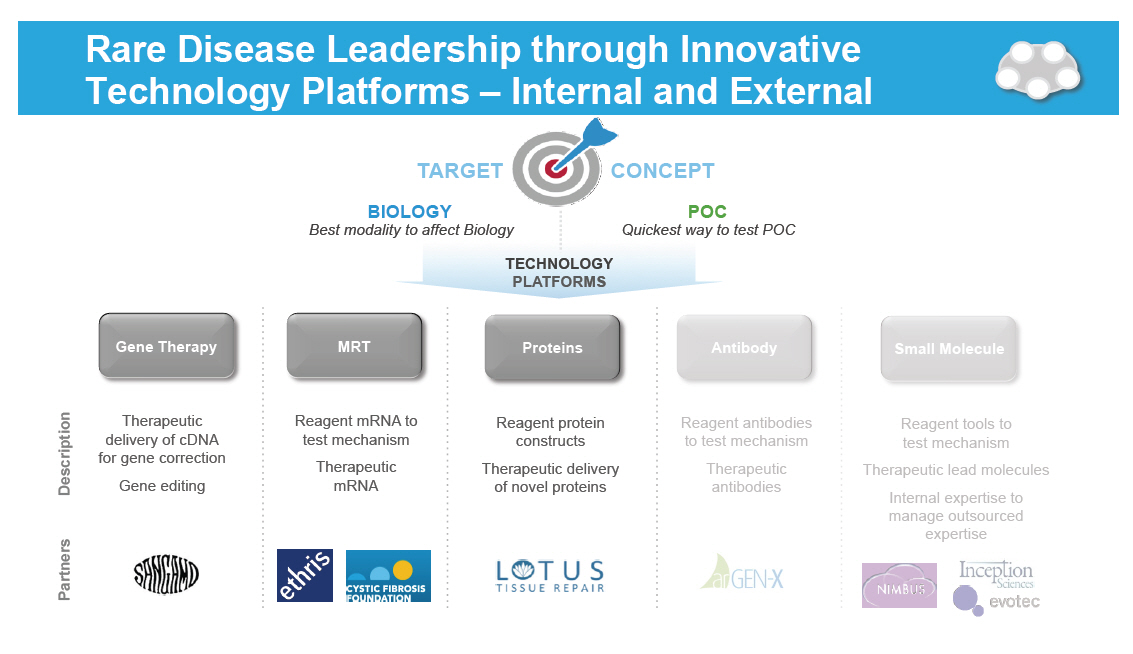 |
DNA, RNA, and Protein Technology Platforms [GRAPHIC OMITTED] Technology platforms Gene Therapy MRT Proteins Description [] Therapeutic delivery of cDNA for gene correction [] Gene editing [] Reagent mRNA to test mechanism [] Therapeutic mRNA [] Reagent protein constructs [] Therapeutic delivery of novel proteins [GRAPHIC OMITTED] 42 |  |
Gene Therapy Transformational Therapy for Monogenic Diseases [GRAPHIC OMITTED] Therapeutic delivery of cDNA for gene correction Reagent mRNA to test mechanism Therapeutic mRNA Reagent protein constructs Therapeutic delivery of novel proteins [GRAPHIC OMITTED] 43 |  |
Shire - Sangamo Collaboration Goal: Using Zinc Finger Protein (ZFP) Technology as Disease Therapeutics Shire innovative approach to address root-cause of life-threatening diseases Entry into gene editing therapies Cross-functional collaboration aimed to deliver multiple therapeutics Technology affords exploration of multiple approaches for disease-modifying therapies Initial focus on Hemophilia and Huntington Disease [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] 44 | 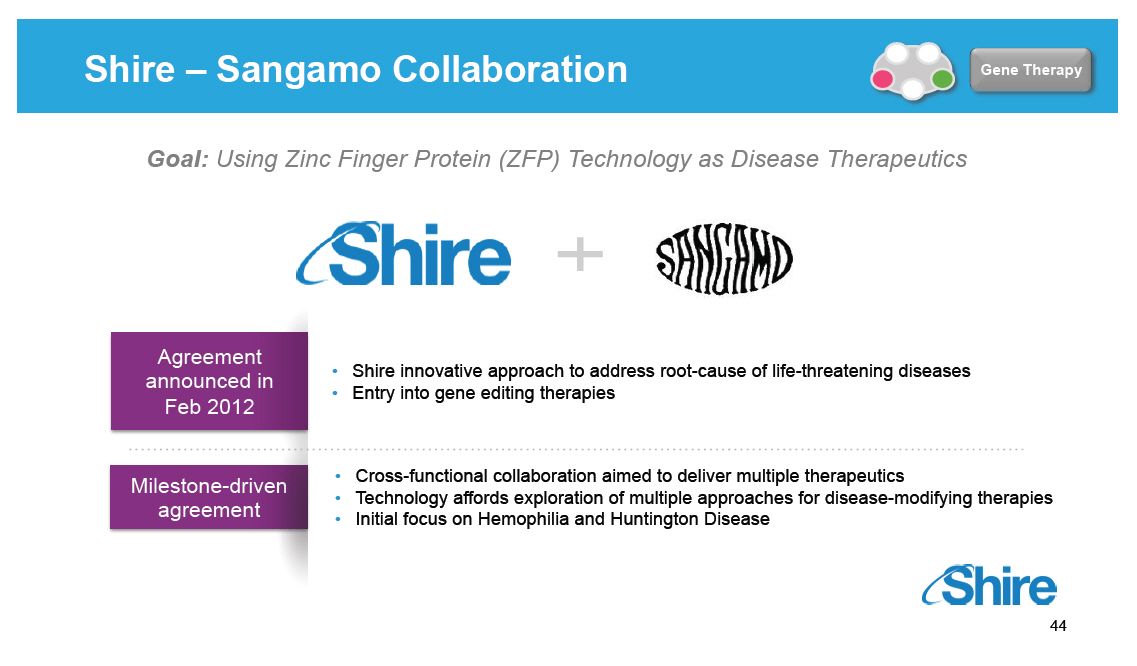 |
Engineered ZFPs Enables Sequence Specificity and Function [GRAPHIC OMITTED] Gene Therapy ZFP attached to a functional domain or nuclease [] Regulation of target gene expression (up-regulation or repression) [] Double strand break in the DNA (gene disruption or correction) [GRAPHIC OMITTED] [GRAPHIC OMITTED] 45 | 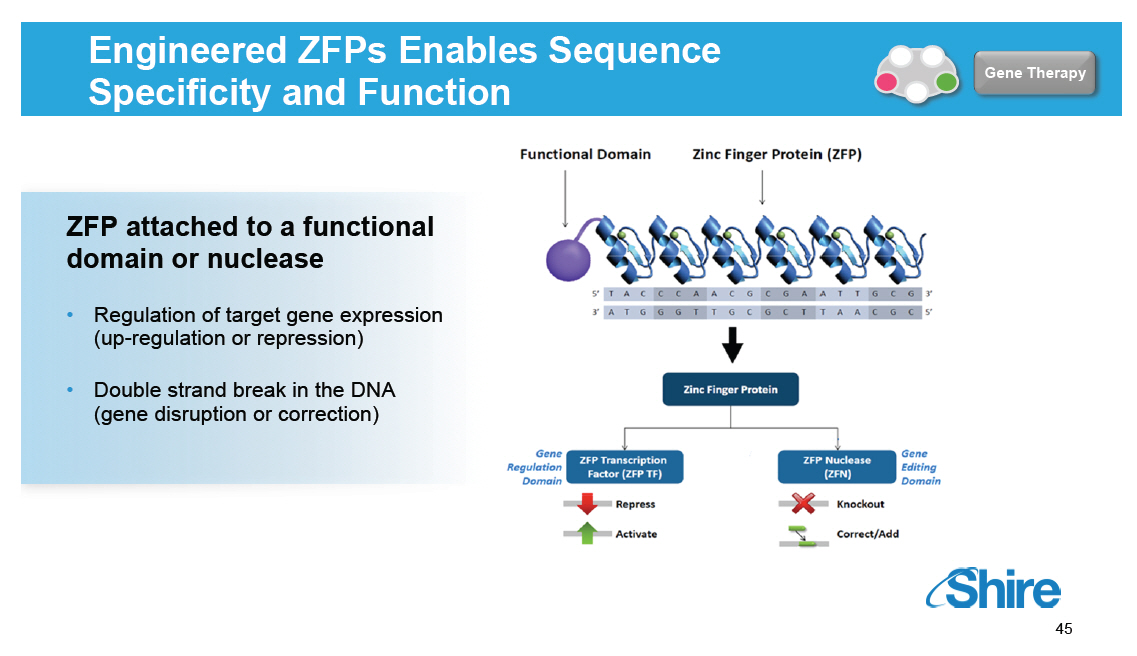 |
Gene Therapy in Albumin Safe Harbor Locus: Factor VIII and Factor IX [GRAPHIC OMITTED] Gene Therapy DISEASE [] Rare hereditary disorder in which the ability of patients' blood to clot is impaired due to impaired FIX or FVIII production -- leads to excessive and uncontrolled internal bleeding, pain and eventual permanent damage to joints and muscles [] Epidemiology: 1 / 5000 male births (~8 out of 10 people who have hemophilia have type A) [] Disease severity: severe, moderate, mild -- dependent on percentage of FVIII / FIX level in blood, (less than 1%, 1-5%, >5%) [GRAPHIC OMITTED] [GRAPHIC OMITTED] 46 | 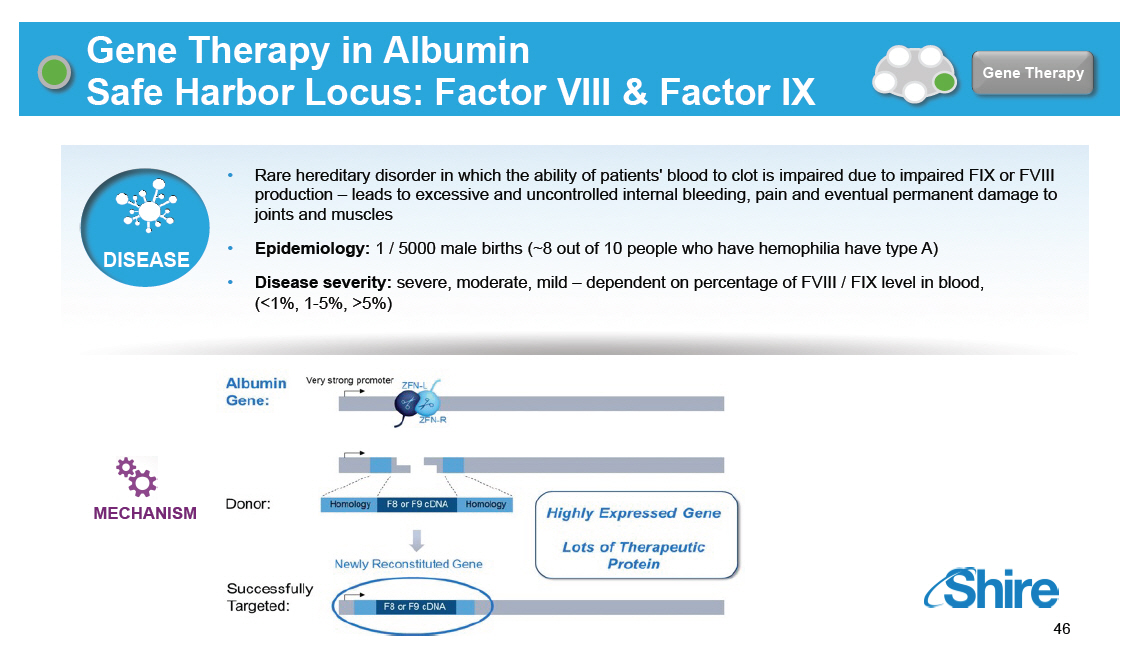 |
Systemic Delivery of ZFP Therapeutics([R]) via AAV Vectors [GRAPHIC OMITTED] Gene Therapy ZFN Pair and Corrective Gene Homology Corrective Gene Homology ZFN ZFN Nucleic Acids Coding for ZFNs and Corrective Gene in AAV Vectors AAV Vectors Peripheral IV Administration AAV Traffics to Liver And Permanently Corrects Gene Liver Cells Secrete Corrected Protein In Vivo Protein Replacement (e.g. Factor IX) [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 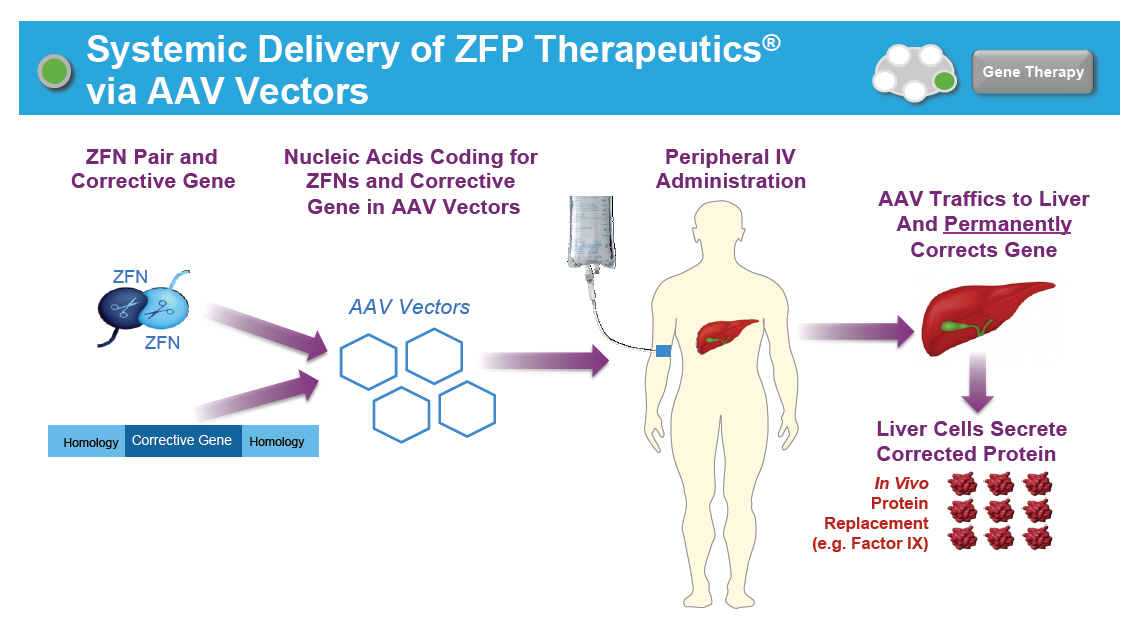 |
Factor IX Gene Therapy Development Candidate for Hemophilia B: Murine POC [GRAPHIC OMITTED] Gene Therapy Factor 9 Expression Elevated 1500 ) (-1) 1000 hF.IX (ng ml 500 Plasma 50 25 0 Baseline Week 2 Clotting Time Reduced 100 * * 80 60 aPTT (s) 40 20 0 n . s . Baseline Week 2 Wild Type Duration of Expression Beyond 60 Weeks 6000 ) (-1) hF.IX (ng/ml 4000 Plasma 2000 0 0 4 8 1230 4050 60 Time (weeks after Injection) [GRAPHIC OMITTED] 48 |  |
Huntington's Disease: Selective Inhibition of Disease Causing Mutation [GRAPHIC OMITTED] Gene Therapy DISEASE [] Huntington's disease is an autosomal dominant neurodegenerative disease characterized by cognitive, behavioral and motor dysfunction. It is a progressive disease initiating typically at mid-life, with an average death 15 years after onset. [] Caused by expansion of a CAG nucleotide repeat within the first exon of the huntington gene causing a misfolding and pathologic conformation of the huntington protein [] Epidemiology: 8 / 100,000 (prevalence) MECHANISM [GRAPHIC OMITTED] Mutant allele selectively inhibited [GRAPHIC OMITTED] 49 |  |
Huntington's Disease Gene Therapy: Murine POC [GRAPHIC OMITTED] Gene Therapy CURRENT DATA 61% knock-down of HD mutant allele in ZFP-treated striata 7 weeks after injection NS [] *** Normalized Expression ZFP Mouse R6/2 ZFP GFP Htt Transgene GFP qPCR %GFP 102 39 *** P < 0.001 Reduced clasping (primary motor phenotype) In HD model of showing Proportion in 30s clasping mice AAV-ZFP delivery Statistical longitudinal analysis: Generalized Estimated Equation (GEE) * GFP vs. ZFP = OR 2.4, p = 0.015 [GRAPHIC OMITTED] 50 | 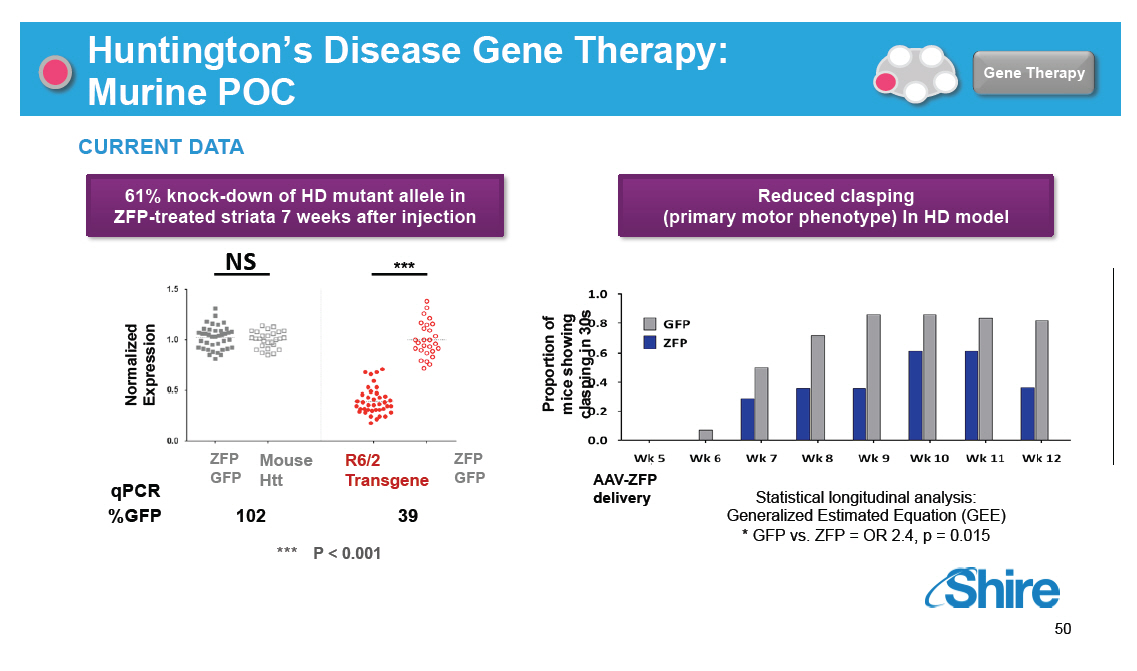 |
Huntington's Disease Gene Therapy: Murine POC [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
mRNA Therapeutics Platform Overview [GRAPHIC OMITTED] MRT Transformational Technology Broad Applicability Proof-of-Principle In Multiple Disease Models Scalable Manufacturing In Place Other [] Novel therapeutic -- Messenger RNA designed to enable the in vivo production of both intracellular and secreted proteins -- ability to treat diseases that cannot be addressed with current technologies [] As patient's own cells produce endogenous protein, downstream processes (e.g., glycosylation, protein processing and trafficking) remain unaffected [] Unlike gene therapy, does not alter the genome [] Potent, dose-dependent pharmacology is reversible upon cessation of treatment in animal models [] Rapid normalization of clinical biomarkers in liver disease models [] Successful delivery of therapeutic protein to lungs via nebulization in pulmonary model [] Successful delivery of mRNA in mouse models via intrathecal, intra-ocular and intra-articular administration; efficient production of therapeutic antibodies [] "Plug-and-play" platform with ability to use same reagents, same cell-free production process and same proprietary delivery vehicle -- rapid, cost-effective, small footprint manufacturing [] Protected by extensive IP portfolio: 26 patent families and 70+ pending applications [] Repeat dosing treatment algorithm -- no need to define new commercial model (e.g., gene therapy "one-and-done") [GRAPHIC OMITTED] 52 |  |
MRT has demonstrated Potential for Broad Therapeutic Applicability [GRAPHIC OMITTED] MRT Proteins produced systemically 4000 (ng/mL) 3500 Serum 3000 in 2500 Protein 2000 IX 1500 Factor 1000 Mean 500 0 3h 6h 12h 24h 48h 72h Time (Post-Administration) Mean Human EPO Protein in Mouse Serum (Single IV Injection) 16 (ug/mL) 14 12 Protein 10 EPO 8 Human 6 4 Mean 2 0 1 2 4 6 8 24 48 Time (h) GLA Expression In Mouse Serum Over Time After Treament via GLA MRT 6000 5000 Serum 4000 GLA/mL 3000 ng 2000 1000 0 3 6 12 24 48 72 Time Post-Treatment Factor IX OTC EPO MRT Technology CFTR AGAL ASS1 Proteins produced locally Mitochondrial Transmembrane- Lung 1200 Total 1000 protein/mg 800 Protein [] 600 encoded Cytoplasmic 400 ng MRT-[] 200 0 n= 4 per group Time (Post-[]Administration) MRT demonstrated across species: Mouse, Rat, Rabbit, Pig, Non-human Primate [GRAPHIC OMITTED] 53 |  |
MRT CFTR: Development Candidate to Treat Cystic Fibrosis [GRAPHIC OMITTED] Autosomal recessive disorder DF508 is most common mutation (>70% of patient population) Results in improper folding and mis-trafficking of protein away from membrane surface Results in clogging of the airways due to mucus build-up ~60,000 Patients in US and EU Recent treatments focus on small subset of patients Significant unmet medical need remains [GRAPHIC OMITTED] 54 |  |
Pulmonary Delivery: Functional Translation of CFTR MRT In Vitro and In Vivo in mice Nanoparticle delivered hCFTR mRNA can be observed in target epithelial cells in vivo1... 1High magnification (40x) representation of in situ hybridization analysis of lungs from CFTR KO mice treated with hCFTR mRNA nanoparticles. hCFTR mRNA is observed in apical cytoplasm of target epithelial cells within bronchus. Tissues were harvested 24 hours post-administration. ....and creates functional ion channels in vitro2 2Current-voltage plot comparing chloride ion-generated current of various treated HEK293T cells 24 hours after transfection of hCFTR mRNA. The light blue represents cells that have been transfected with hCFTR mRNA 24 hours prior, followed by treatment with activator 8-Br-cAMP (4 min prior to analysis). The dark blue line represents similarly treated cells which were subjected to further exposure of 10 mM CFTRinh-172. The black line represents untreated HEK 293T cells (untreated). [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Animal Proof-of-Principle for Lung Delivery of CFTR Shows Broad Biodistribution of Functional Protein [GRAPHIC OMITTED] [GRAPHIC OMITTED] 56 |  |
IHC in Normal Pig Lung: Anti-hCFTR Antibody Detection of MRT-Derived hCFTR [GRAPHIC OMITTED] MRT Immunohistochemistry images of one representative pig from either vehicle treated (A) or 10 mg human CFTR mRNA dose group (B) A) [GRAPHIC OMITTED] B) Positive stain [GRAPHIC OMITTED] Vehicle 10 mg human CFTR mRNA [GRAPHIC OMITTED] 57 |  |
Summary of Pulmonary Delivery Results [GRAPHIC OMITTED] MRT [] CFTR MRT offers potential for disease modification in an area of significant unmet medical need [] Pulmonary delivery and expression of hCFTR:FFL to the porcine lung by nebulized mRNA formulation was demonstrated [] Tissue regions with expression of Luciferase also co-expressed hCFTR [] Luciferase negative regions lacked hCFTR expression [] Provides evidence that target lung tissue, i.e. bronchial epithelial cells, expresses hCFTR following mRNA delivery [] Tolerance to 5 doses at weekly intervals in pigs demonstrated [] No tissue pathology detected [] No adverse clinical signs observed [] No increase in liver enzymes or inflammatory cytokines detected [] Early development to date has demonstrated: [] Strong preclinical data package [] Clear development strategy [GRAPHIC OMITTED] 58 |  |
MRT ASS1: Development Candidate to Treat Citrullinemia [GRAPHIC OMITTED] Citrullenemia is an autosomal recessive metabolic disorder Human urea cycle pathway Due to mutations in argininosuccinate synthetase (ASS1) ~14% of all urea cycle disorders Results in high levels of plasma ammonia, leading to lethargy, vomiting, seizures, and failure to thrive 1: 57,000 live births Expressed in liver and kidney No disease modifying therapies available [GRAPHIC OMITTED] |  |
Immunohistochemical (IHC) Detection of Human ASS1 Protein Encoded by MRT [GRAPHIC OMITTED] MRT Pharmacokinetic profile studied after single dose of MRT 1.0 mg / kg MRT, single dose, IV, WT mice 30 min 12h 72h 3h 24h 1 Week 6h 48h Untreated [] IHC staining of MRT- encoded protein [] Positive staining in hepatocytes and Kupffer cells [] Widespread distribution in liver [] Protein detectable up to 1 week post-dose [GRAPHIC OMITTED] 60 |  |
Evidence of Efficacy in Citrullinemia Mouse Model [GRAPHIC OMITTED] MRT Initial efficacy study performed in ASS1 deficiency model; hypomorphic mutation with ~10% of normal ASS1 activity [] Single dose (1.0 mg / kg) of either ASS1 mRNA loaded LNP or empty LNP (control) [] Measure plasma ammonia levels pre- and post-dose [] Compare model with WT levels Plasma Ammonia Levels Pre- and Post-Treatment with ASS1 MRT Pre-Dose Day 16 (24h post dose) 24 hr Day 22 Day 8 Day 29 (pre-dose) Day 15 (pre-dose) Day 30 700.00 600.00 mol/L) 500.00 [] ( 3 400.00 NH 300.00 Plasma 200.00 100.00 0.00 N=6 / group D0 D15 D29 ASS1 MRT1 Wild Type Ass1 (fold) (T389I) mutant [GRAPHIC OMITTED] 61 | 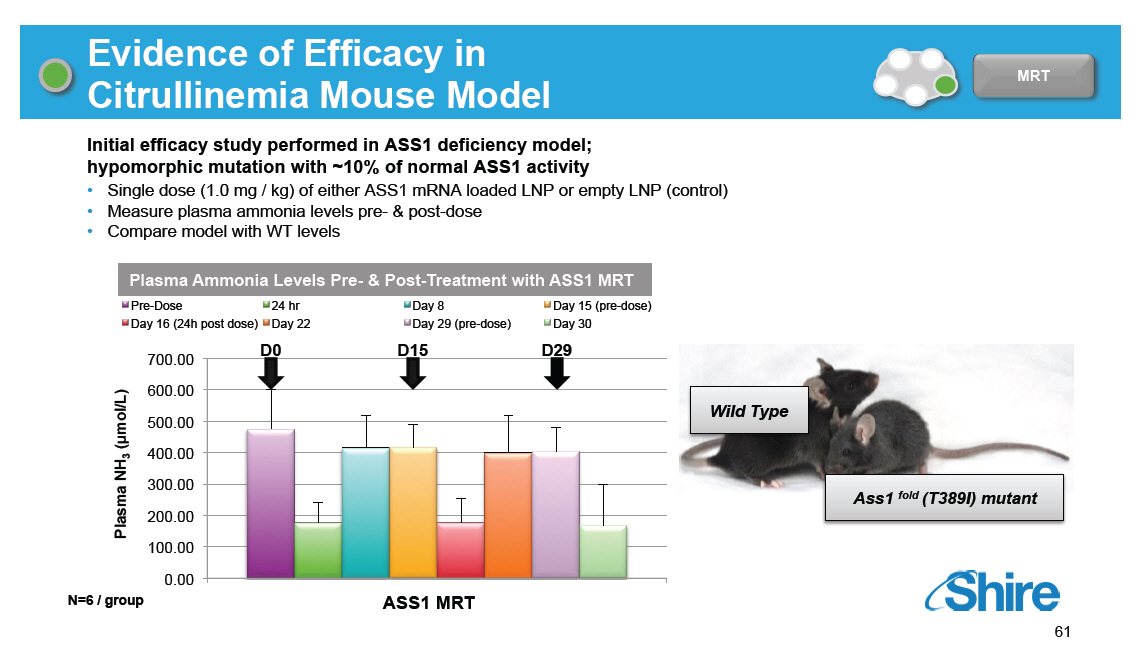 |
Summary of Intravenous MRT for Urea Cycle Diseases [GRAPHIC OMITTED] MRT [] We have focused initially on urea cycle defects for IV MRT as they allow rapid clinical proof-of-concept and compelling market opportunity [] Clinical path clarity [] High unmet need in a sizeable market [] Clear MRT advantage [] Mouse model results demonstrate approximately 50-100% of normal liver ASS1 and ammonia reduction following MRT [] Human ASS1 mutational spectrum in Citrullinemia suggests activity >10% would be disease-modifying [GRAPHIC OMITTED] 62 |  |
mRNA Replacement Therapy (MRT) MRT for Monogenic Diseases Therapeutic delivery of cDNA for gene correction Gene editing Reagent mRNA to test mechanism Therapeutic mRNA Reagent protein constructs Therapeutic delivery of novel proteins [GRAPHIC OMITTED] [GRAPHIC OMITTED] 63 |  |
Novel Therapeutics via Protein Engineering [GRAPHIC OMITTED] Proteins [GRAPHIC OMITTED] [GRAPHIC OMITTED] 64 |  |
Human rCollagen 7: Development Candidate to Treat Dystrophic Epidermolysis Bullosa (DEB) Rare genetic disease: fragile blistering skin, deformed limbs, numerous co-morbidities, early death Mutations in gene encoding Collagen Type VII (C7): DISEASE autosomal dominant (DDEB) & autosomal recessive (RDEB) Aberrant function / absence of C7 at dermal-epidermal junction affect attachment of epidermis to dermis ~5000 diagnosed patients: 2,000 addressable patients (base case) with IV protein replacement therapy; all genders and races affected No disease modifying treatment available: only recurrent, symptomatic treatments; painful and costly disease IV rC7 as protein replacement therapy will: Reach the lamina densa at dermal-epidermal surfaces Incorporate into matrix to normalize function Correct blistering abnormalities and complications [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 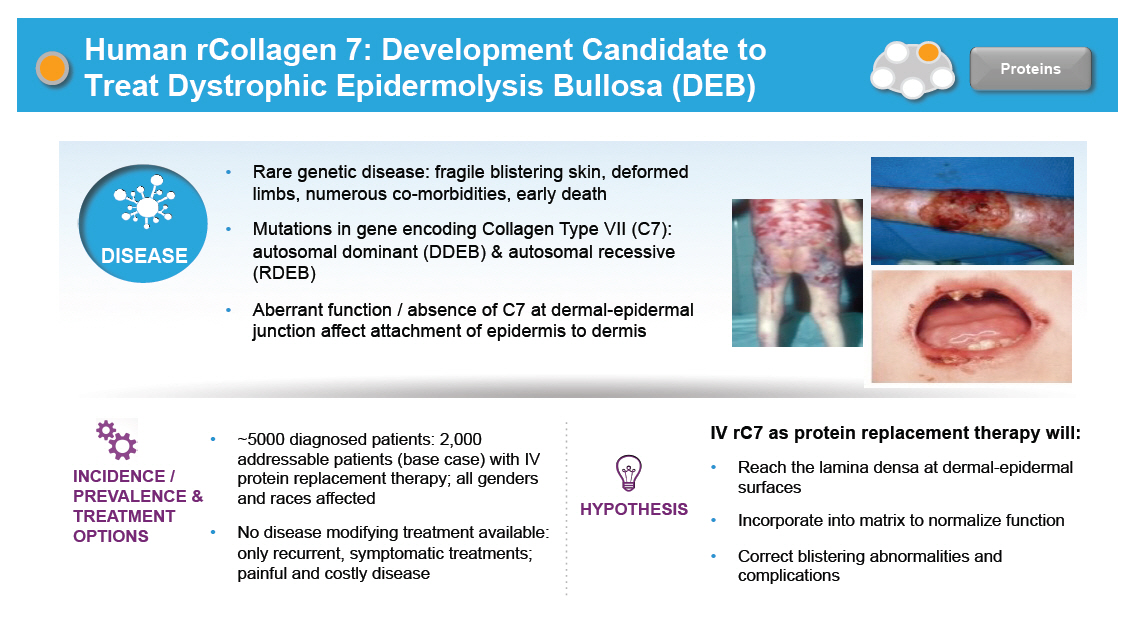 |
rC7 Forms Anchoring Fibrils Critical for Dermal-Epidermal Adhesion in Animal Models C7 forms anchoring fibrils, attachment structures in the basement membrane zone (BMZ) responsible for adhering the epidermis to the dermis Loss-of-function mutations in COL7A1 lead to abnormal, decreased or absent anchoring fibrils [GRAPHIC OMITTED] Recombinant C7 (rC7) incorporates into basement membrane zone forming anchoring fibrils, reversing separation of dermal-epidermal junction in animal models | 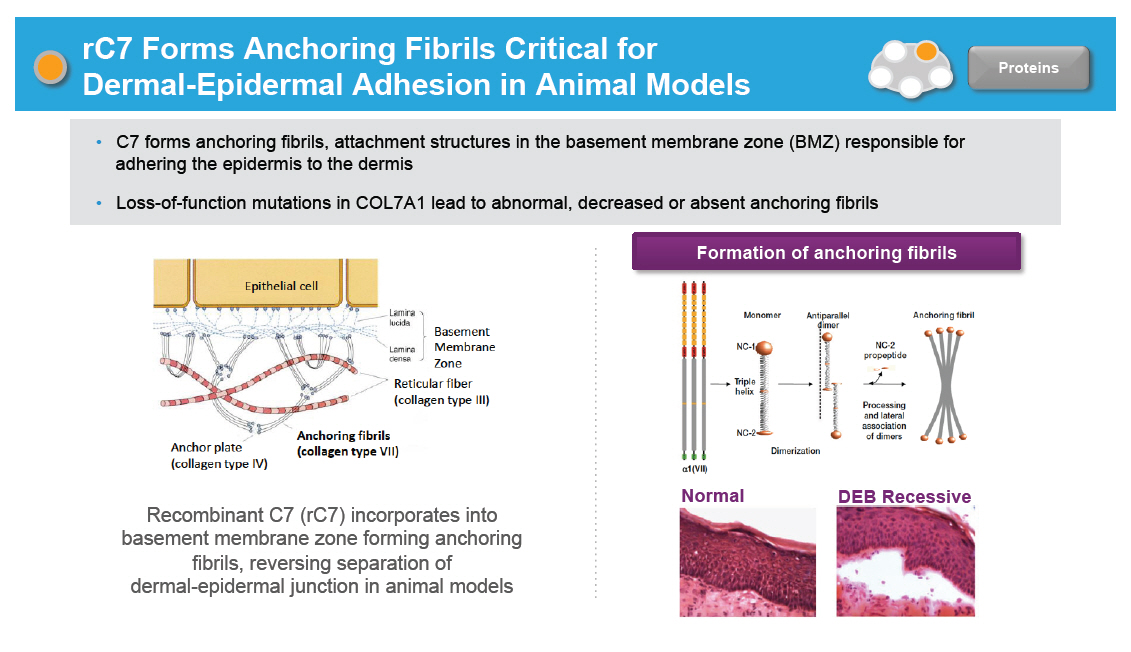 |
Tissue Distribution of Shire Collagen 7 [GRAPHIC OMITTED] Shire Collagen 7 was Distributed to the Proper Location within the Dermal-Epidermal Junction in Multiple Tissues in Mouse Models Control WT Shire CHO-C7 Mice Treated with a Single Injection of Recombinant Collagen 7 Lived Longer Shire Collagen 7 Reversed Dermal-Epidermal Separation in a Dose-Dependent Manner in Mouse Models [GRAPHIC OMITTED] |  |
Research Model for Delivering the Portfolio in Rare Diseases TIGEM BCH Research ventures Internal BD/Alliance Access to tool reagents to test mechanism Pre-defined criteria Data driven decisions DEB /HEMB /RP /Hunter CNS /CF /UCD /HTT 1. Research portfolio built around high confidence targets in diseases with significant unmet medical need 2. Culture of rapid and clear data driven go/no go decisions 3. Efficient and focused team integrating internal and external flexible model to deliver early alignment with process and clinical development 4. ~26 active programs with a goal of delivering 3 programs from research to early development per year with a focus on quick to clinical POC [GRAPHIC OMITTED] 68 | 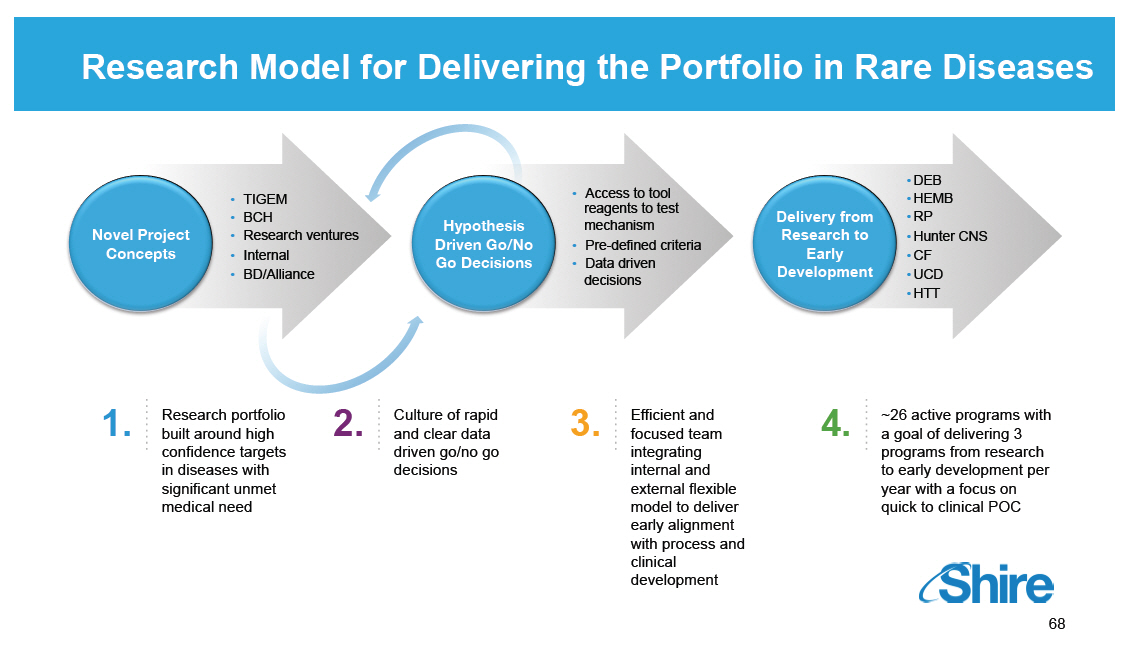 |
Rare Diseases: GI / Metabolic Ciara Kennedy, PhD, MBA -- Head of Cholestatic Liver Disease David Piccoli, MD -- Chief of Gastroenterology, Hepatology and Nutrition, Children's Hospital Of Philadelphia Our purpose We enable people with life-altering conditions to lead better lives. [GRAPHIC OMITTED] |  |
Today's RandD sessions [GRAPHIC OMITTED] Topic Research Overview and Technology Platforms mRNA, Protein Replacement, Gene Therapy, Antibody Platforms Rare Diseases: GI / Metabolic SHP625 (LUM001), SHP626 (LUM002) Rare Diseases: Ophthalmics SHP607 / ROP, SHP630 / BIKAM Rare Diseases: Complement Biology and Renal / Fibrotic SHP616 / Cinryze new uses Rare Diseases: CNS SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen Late-Stage Update SHP606 / Lifitegrast, BED, SHP465 / ADHD Speaker Albert Seymour, PhD Ciara Kennedy, PhD David Piccoli, MD Norman Barton, MD, PhD Howard Mayer, MD Howard Mayer, MD Howard Mayer, MD Randall Brenner Joe Tauber, MD Time (EST) 9:25-10:00 10:00-10.45 11:15-11:45 1:15-1:30 1:30-2:00 2:00-2:45 [GRAPHIC OMITTED] 70 |  |
Multiple Rare Diseases Programs in GI / Metabolic Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 (ALGS, PSC, PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP 555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT Rare Diseases Leadership [GRAPHIC OMITTED] 71 | 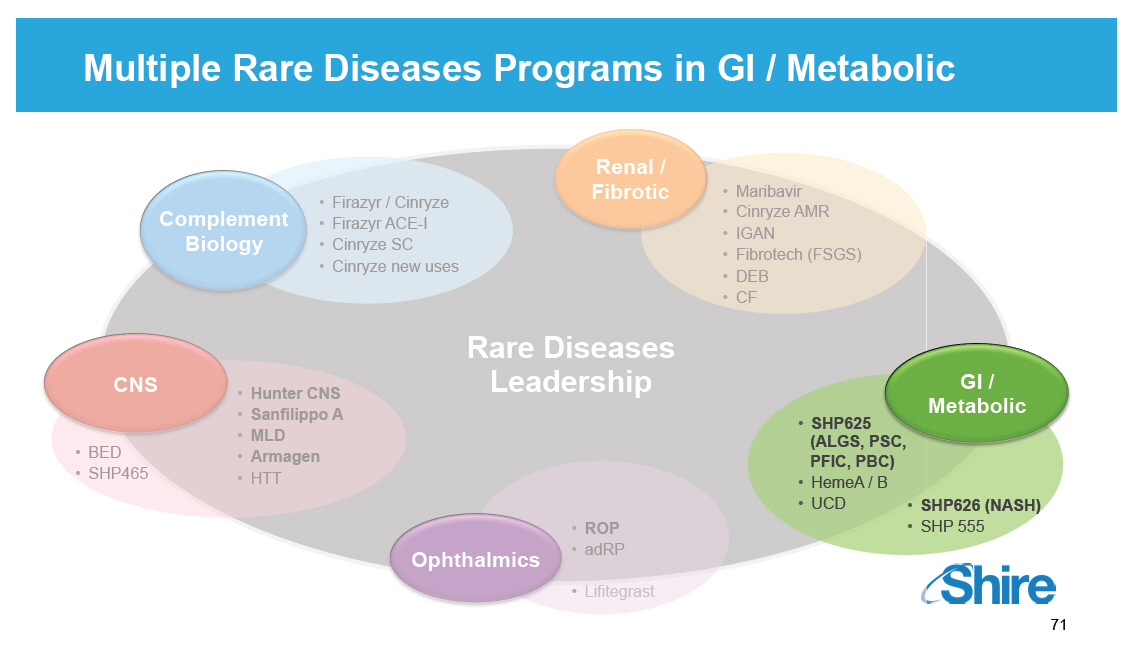 |
Multiple Rare Diseases Programs in GI / Metabolic [GRAPHIC OMITTED] |  |
The Role of Bile Acids [GRAPHIC OMITTED] [] Bile acids are synthesized in the liver from cholesterol [] Recovered from GI tract via the apical sodium bile acids transporter (ASBT) and returned to liver [] Facilitate digestion and absorption of dietary fats and fat-soluble vitamins [] Regulate lipid and glucose metabolism [] Excess bile acids are associated with liver damage and pruritus [] ASBT inhibition: [] Reduces serum bile acid levels [] Decreases serum and hepatic cholesterol [] Lowers plasma glucose [] Reduces insulin resistance [GRAPHIC OMITTED] 73 | 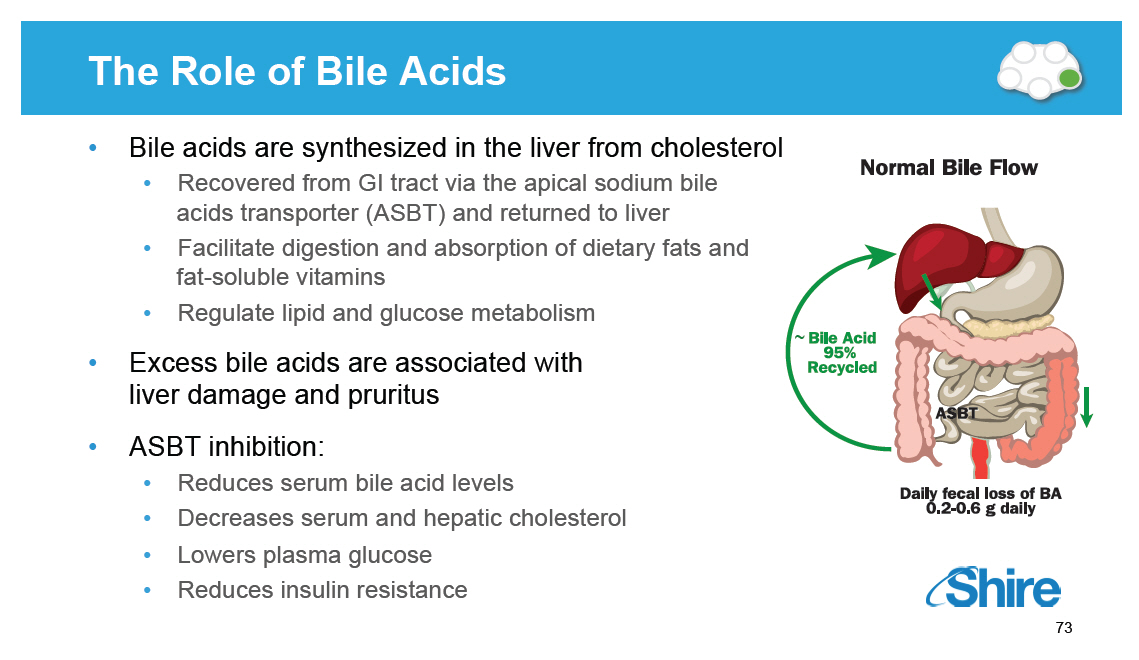 |
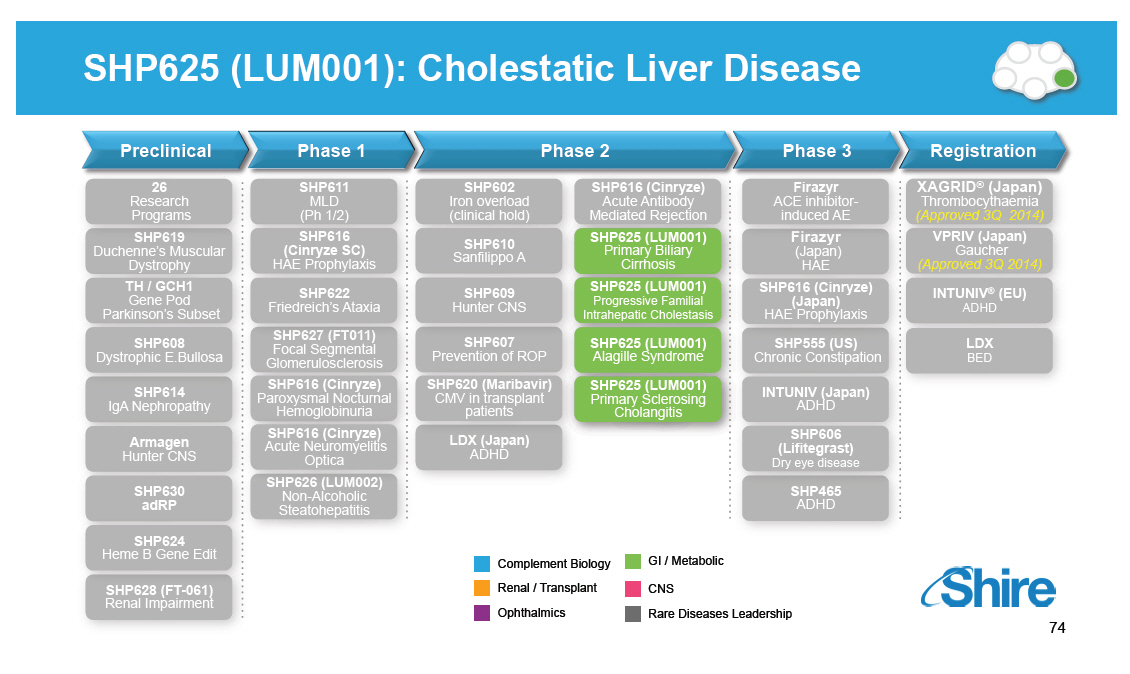
SHP625 (LUM001): Cholestatic Liver Disease
[GRAPHIC OMITTED]
Preclinical
26
Research
Programs
SHP619
Duchenne's Muscular
Dystrophy
TH / GCH1
Gene Pod
Parkinson's Subset
SHP608
Dystrophic E.Bullosa
SHP614
IgA Nephropathy
Armagen
Hunter CNS
SHP630 adRP
SHP624
Phase 1
SHP611
MLD (Ph 1/2)
SHP616
(Cinryze SC)
HAE Prophylaxis
SHP622
Friedreich's Ataxia
SHP627 (FT011)
Focal Segmental
Glomerulosclerosis
SHP616 (Cinryze)
Paroxysmal Nocturnal
Hemoglobinuria
SHP616 (Cinryze)
Acute Neuromyelitis
Optica
SHP626 (LUM002)
Non-Alcoholic
Steatohepatitis
Phase 2
SHP602 SHP616 (Cinryze)
Iron overload Acute Antibody
(clinical hold) Mediated Rejection
SHP610 SHP625 (LUM001)
Primary Biliary
Sanfilippo A
Cirrhosis
SHP609 SHP625 (LUM001)
Progressive Familial
Hunter CNS
Intrahepatic
Cholestasis
SHP607 SHP625 (LUM001)
Prevention of ROP Alagille Syndrome
SHP620 (Maribavir) SHP625 (LUM001)
CMV in transplant Primary Sclerosing
patients Cholangitis
LDX (Japan)
ADHD
Phase 3
Firazyr
ACE inhibitor-induced AE
Firazyr
(Japan) HAE
SHP616 (Cinryze) (Japan)
HAE Prophylaxis
SHP555 (US)
Chronic Constipation
INTUNIV (Japan)
ADHD
SHP606
(Lifitegrast)
Dry eye disease
SHP465
ADHD
Registration
XAGRID(R) (Japan)
Thrombocythaemia
(Approved 3Q 2014)
VPRIV (Japan)
Gaucher
(Approved 3Q 2014)
INTUNIV(R) (EU)
ADHD
LDX
BED
Complement Biology GI / Metabolic
Renal / Transplant CNS
Rare Diseases
Ophthalmics Leadership
[GRAPHIC OMITTED]
74
|  |
SHP625 (LUM001): Novel Therapy with Potential to Address Four Rare Hepatic Conditions [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease Significant unmet need Alagille Syndrome (ALGS) [] Present at 3 months [] Markedly elevated bile acids and cholesterol [] Very intense pruritus [] No approved therapy [] ~13% bile diversion surgery, 21-31% liver transplant [] ~25K prevalence in U.S. / EU Progressive Familial Intrahepatic Cholestasis (PFIC) [] Present at 3-6 months [] Very intense pruritus [] No approved therapy [] ~35% bile diversion surgery, ~50% liver transplant [] Without surgery, fatal by 2nd decade [] ~13K prevalence in U.S. / EU Primary Biliary Cirrhosis (PBC) [] >40 years old, 90% female [] ~50% of patients respond to approved therapy (UDCA(*)) [] Intense pruritus [] Slow progression [] ~275K prevalence U.S. / EU Primary Sclerosing Cholangitis (PSC) [] Mean age at diagnosis: 40 years, 70% Male [] Intense pruritus [] No approved therapy [] Aggressive, life expectancy 8-10 years from diagnosis [] ~60K prevalence in U.S. / EU * Ursodeoxycholic acid (UDCA) Pediatric Adult [GRAPHIC OMITTED] 75 |  |
Cholestatic Pruritus: Not Simply Itching [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease [GRAPHIC OMITTED] John would sleep about 20-40 min at a time, then he would be up with us holding him for 1-2 hours itching. This was all night long. -- Robin (Mother of 2 children with PFIC) [GRAPHIC OMITTED] 76 | 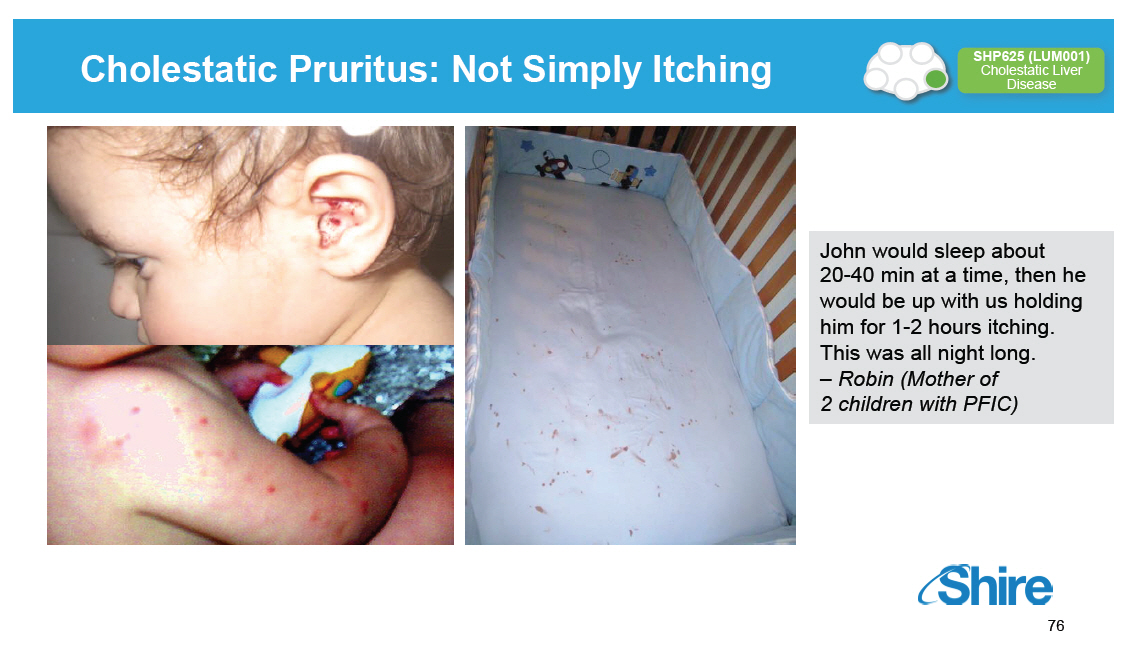 |
Xanthomathosis Manifestation of Elevated Cholesterol [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease [GRAPHIC OMITTED] In severe cases xanthomas can be "disfiguring" causing distortion of the face or extremities, and "disabling" interfering with function (such as hand use or ability to walk) [GRAPHIC OMITTED] 77 | 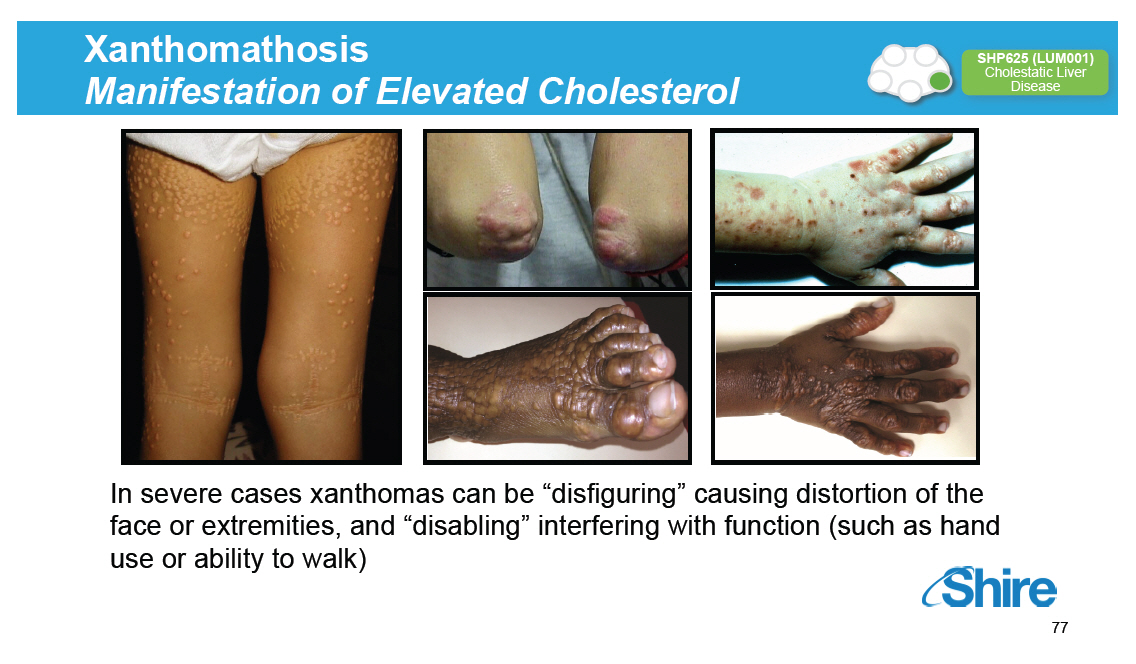 |
Management of Cholestatic Liver Disease [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease [] Limited options for pharmaceutical management of cholestasis [] UDCA only approved for treatment of PBC [] Bile acid resins can reduce pruritus in some patients [] Rifampicin may alleviate pruritus in some cases [] In a 15 year retrospective review of Alagille syndrome patients from Kings College London; <20% of patients experience relief of pruritus following treatment with UDCA and with cholestyramine: Treatment Effect None -- Some Good -- Very Good Adverse Effects UDCA Frequency (n = 40) 32 8 3 Percentage (%) 80 20 7.5 Cholestyramine Frequency (n = 18) 15 3 6 Percentage (%) 83.4 16.7 33.3 [] Many patients resort to invasive interventions to manage disease Kronsten, JPGN (2013); 149-154 78 |  |
Reduction of Serum Bile Acid Levels Associated With Pruritus Control in a Patient with PFIC [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease Serum Bile Salt Concentration and Degree of Itch in a PFIC Patient Over a 4-year Course 4+ Itch O Naso-duodenal Cholecystectomy drainage tube SBS Accidental removal of cholecystectomy tube I-lM Whitington, Gastroenterology 1988 Age (Years) [GRAPHIC OMITTED] 79 |  |
Lowering Bile Acids Results in Significant Clinical Benefits Removing bile acids through surgical intervention: Reduces serum bile acids Improves biochemical markers of liver disease Rapidly reduces itching Slows disease progression Disfiguring and associated with serious complications ALGS After Before Surgery(1) Surgery(1) Bile Acids ((proportional to)mol/L) 115 28 Bilirubin (mg/dL) 2.4 1.6 Itching (0 no scratching- 4 1 4 cutaneous mutilation) PFIC Before Surgery(2) After Surgery(2) Bile Acids ((proportional to)mol/L) 337 11 Bilirubin (mg/dL) 2.4 1.5 Itching (0-4) 3 1 [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Treatment of Severe Pruritus in Patients With Cholestasis With Extracorporeal Albumin Dialysis (PBC, PSC, ALGS) [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease Molecular adsorbent recirculating system (MARS) reduces serum bile acid levels and controls pruritus [] 20 patients (12 females), mean age: 51 [] 3.4 years with chronic cholestatic liver disease or chronic liver-graft rejection [] Pruritus assessed with VAS before and after MARS, and 30 days thereafter [] Liver tests, including total bilirubin, ALP, GGT and total bile acid levels were determined (r =-0.82, p<0.001) [GRAPHIC OMITTED] Pares A. Journal of Hepatology 2010 81 |  |
Animal Models of Cholestasis [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease Partial bile duct ligation (pBDL) [] Mimics the paucity of bile ducts or narrowing of bile ducts observed in clinical cases of cholestasis MDR2 knockout mouse model [] Model for PFIC3 (MDR2 deficiency) [] Primary sclerosing cholangitis (PSC) [GRAPHIC OMITTED] 82 |  |
SHP265 (LUM001) Increased Total Fecal Bile Acid Excretion 10 Days of Treatment in Rat Model of Cholestasis [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease 100 P<0.0001 P<0.0001 80 60 mol/24 hr) 40 [] ( Total Fecal Bile Acids 20 0 Sham Vehicle SHP625 LUM001 - .3 mg/kg LU LUM001 01 (0.3 mg/kg) (10 mg/kg) LUM001 causes a 4.8 - and 5.9 -fold increase in total FBA after 10 days treatment with 0.3 and 10 mg/kg/d, respectively Data Presented at AASLD [GRAPHIC OMITTED] 83 | 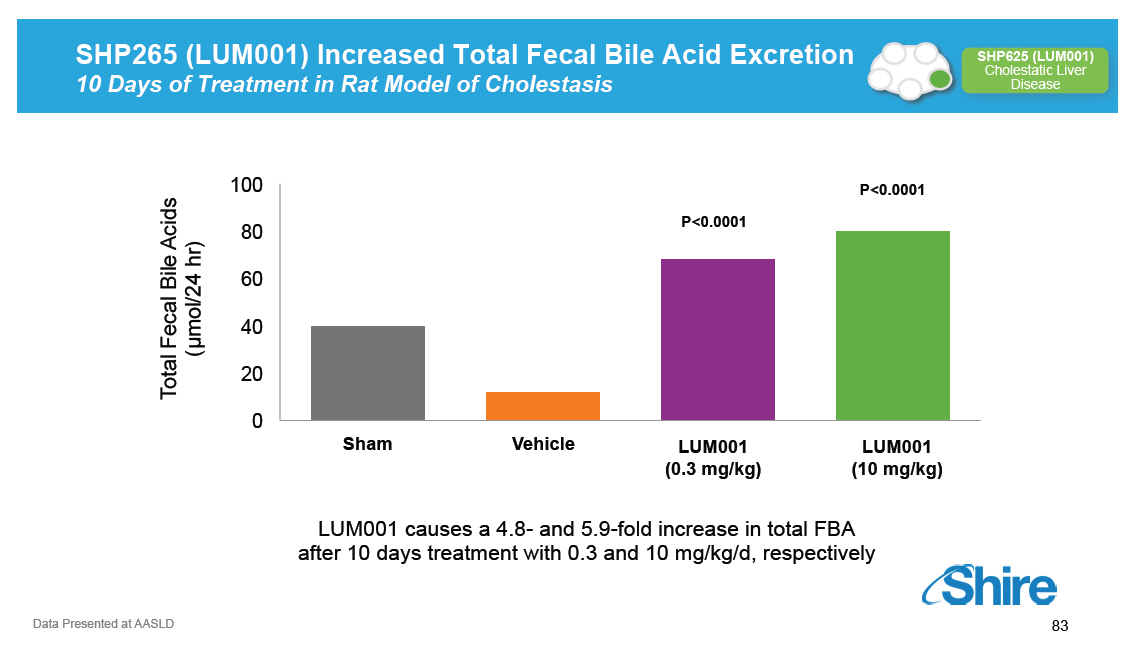 |
SHP625 (LUM001) Improved Biochemical Markers of Liver Damage pBDL Rat Cholestasis Model [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease Total Serum Bile Acids 500 mol/L 400 [] 300 SBA - 200 P=0.055 100 P=0.012 0 g-Glutamyl Transpeptidase 16 12 P=0.103 GGT -- IU/L 8 4 P=0.022 0 Total Bilirubin 16 12 dL Total Bilirubin -- mg/ 8 P=0.103 4 P=0.022 0 Alkaline Phosphatase 10 8 6 ALP -- IU/L 4 2 P=0.033 P=0.032 0 Sham n=5 Vehicle n=4 LUM001-- 0.3 mg/kg n=3 LUM001-- 10 mg/kg n=3 Aspartate Aminotransferase 400 300 200 AST -- IU/L P=0.034 100 P=0.018 0 P value: LUM001-treated vs. Vehicle Group Alanine Aminotransferase 400 300 ALT -- IU/L 200 100 P=0.149 P=0.021 0 Data Presented at AASLD [GRAPHIC OMITTED] 84 |  |
SHP625 (LUM001) Reduced Liver Injury pBDL Rat Cholestasis Model [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease Vehicle [GRAPHIC OMITTED] Moderate bile duct epithelial cell proliferation (arrow), cell necrosis and inflammatory cell infiltration (arrowhead) LUM001 (10 mg/kg) [GRAPHIC OMITTED] Normal bile duct morphology within the portal region with minimal epithelial cell proliferation [GRAPHIC OMITTED] Data Presented at AASLD 85 | 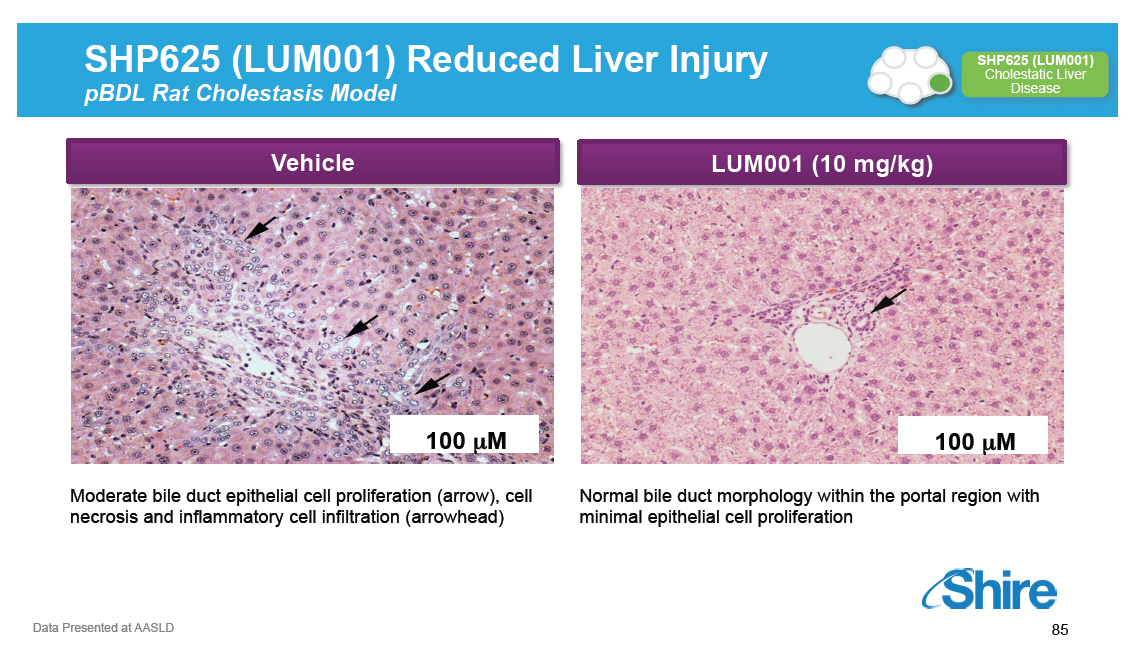 |
Animal Models of Cholestasis [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease Partial bile duct ligation (pBDL) [] Mimics the paucity of bile ducts or narrowing of bile ducts observed in clinical cases of cholestasis MDR2 knockout mouse model [] Model for PFIC3 (MDR2 deficiency) [] Primary sclerosing cholangitis (PSC) [GRAPHIC OMITTED] 86 |  |
ASBTi* Promoted Fecal Bile Acid Losses and Reduced Serum Bile Acid Levels in MDR2-/- Model *ASBTi used is SC-435, a research analogue of LUM001 Data Presented at AASLD [GRAPHIC OMITTED] 87 [GRAPHIC OMITTED] |  |
ASBTi* Blocked Wasting and Attenuated Hepatocellular Injury and Cholestasis in MRD2-/- Model [GRAPHIC OMITTED] *ASBTi used is SC-435, a research analogue of LUM001 Data Presented at AASLD [GRAPHIC OMITTED] 88 | 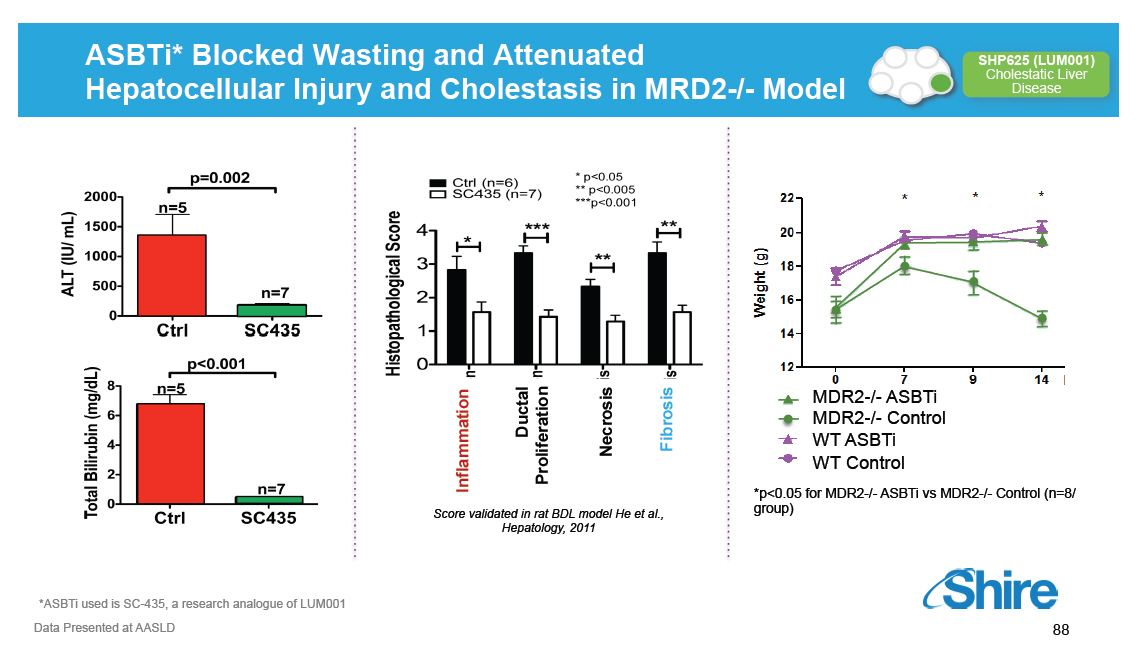 |
SHP625 (LUM001): Safety Profile
Z SHP625 (LUM001)
Cholestatic Liver
Disease
/ SHP625 (LUM001) was designed to be minimally absorbed
/ Extensive non-clinical data package with good safety
margins
/ Experience in over 1,400 human subjects in 12 clinical
studies
/ Most common AEs in completed studies were gastrointestinal
in nature; 1 possibly related SAE
[GRAPHIC OMITTED]
|  |
SHP625 (LUM001): Increased Fecal Bile Acids Excretion Z and Lowered Serum Bile Acids in Clinical Trials / SHP625 (LUM001) once daily dosing for 28 days in healthy volunteers (n=167) / Data are shown as the mean (mmol/24 hours) for fecal bile acids and mean AUC(0-15 hr) (mmol/ 24 hours) for serum bile acids / AUC, or area under the curve, is a measure of drug concentration in the blood [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
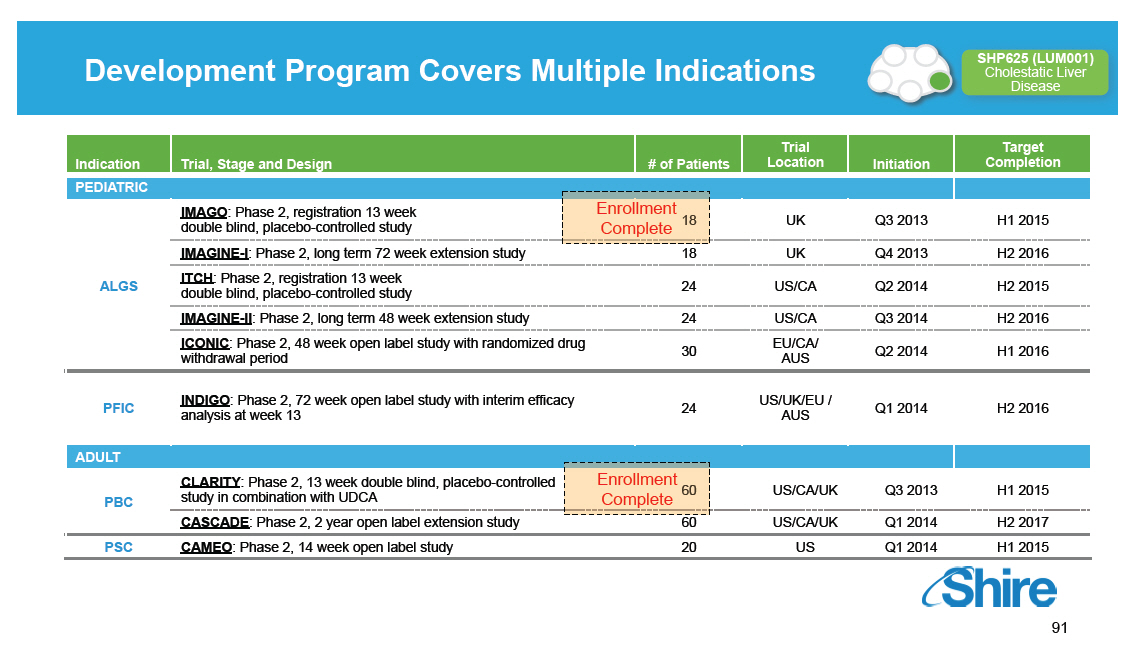
Development Program Covers Multiple Indications
Indication
PEDIATRIC
ALGS
-----------------
PFIC
ADULT
PBC
-----------------
PSC
-----------------
Trial, Stage and Design
IMAGO: Phase 2, registration 13 week
double blind, placebo-controlled study
IMAGINE-I: Phase 2, long term 72 week extension study ITCH:
Phase 2, registration 13 week double blind,
placebo-controlled study
IMAGINE-II: Phase 2, long term 48 week extension study
ICONIC: Phase 2, 48 week open label study with randomized
drug withdrawal period
---------------------------------------------------------------
INDIGO: Phase 2, 72 week open label study with interim
efficacy analysis at week 13
CLARITY: Phase 2, 13 week double blind, placebo-controlled
study in combination with UDCA
CASCADE: Phase 2, 2 year open label extension study
---------------------------------------------------------------
CAMEO: Phase 2, 14 week open label study
-------------------------------------------------------------------
# of Patients
Enrollment
Complete
18 24
24 30
24
Enrollment Complete
60 20
Trial Target
Location Initiation Completion
UK Q3 2013 H1 2015
UK Q4 2013 H2 2016
US/CA Q2 2014 H2 2015
US/CA Q3 2014 H2 2016
EU/CA/ Q2 2014 H1 2016
AUS
-------------------------------------------------------
US/UK/EU / Q1 2014 H2 2016
AUS
US/CA/UK Q3 2013 H1 2015
US/CA/UK Q1 2014 H2 2017
-------------------------------------------------------
US Q1 2014 H1 2015
-------------------------------------------------------
[GRAPHIC OMITTED]
[GRAPHIC OMITTED]
|  |
SHP625 (LUM001): Summary [GRAPHIC OMITTED] SHP625 (LUM001) Cholestatic Liver Disease [] SHP625 (LUM001) is a highly potent and selective, minimally- absorbed ASBT inhibitor [] Orphan drug designation for ALGS, PFIC, PBC, PSC in US and EU [] Parallel development in all 4 high unmet need indications [] Data from Phase 2 studies in first half of 2015 [] Phase 3 studies in PBC and PSC will be required for approval [] Plan to file NDA/MAA for pediatric indications (ALGS/PFIC) using Phase 2 registration studies in the first half of 2016 [GRAPHIC OMITTED] 92 |  |
SHP626 (LUM002): Non-Alcoholic Steatohepatitis (NASH) Preclinical 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 Heme B Gene Edit SHP628 (FT-061) Renal Impairment Phase 1 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis Phase 2 Phase 3 Registration [GRAPHIC OMITTED] [GRAPHIC OMITTED] 93 |  |
NASH -- A Growing Problem [GRAPHIC OMITTED] SHP626 (LUM002) Non-Alcoholic Steatohepatitis Disease Overview [] Non-alcoholic Fatty Liver Disease (NAFLD) is the hepatic manifestation of metabolic syndrome; NAFLD affects ~27% of adults in U.S. / EU [] Non-Alcoholic Steatohepatitis (NASH) is the progressive form of NAFLD characterized by accumulation of fat, fibrous tissue, inflammation and damage to the liver; characteristics resemble those of alcoholic steatohepatitis [] Underlying cause of NASH-associated liver injury is not fully known; strong association with obesity, Type 2 diabetes, high cholesterol and triglycerides [] ~10% of the NAFLD population has NASH (1) [] Estimated ~6 million individuals in the U.S. have NASH and ~600K have NASH-related cirrhosis [] NASH projected to surpass Hepatitis C and alcoholic liver disease to become leading cause for liver transplant by 2020 [] Despite increasing incidence of nonalcoholic fatty liver disease (NAFLD) and NASH, there are no treatments currently approved for these common liver disorders (1) National Digestive Diseases Information Clearinghouse 94 |  |
Rationale for SHP626 (LUM002) in NASH [GRAPHIC OMITTED] SHP626 (LUM002) Non-Alcoholic Steatohepatitis Lipid Lowering [] Blocking bile acid reabsorption decreases level of bile acids returning to the liver via the enterohepatic circulation [] Stimulating bile acid synthesis from cholesterol [] Reduction of hepatic cholesterol reduces oxidative stress [] Preclinical data demonstrates reduction of serum and hepatic LDL-cholesterol [] Clinical data demonstrates reduction of serum LDL-cholesterol in healthy volunteers Regulation of Metabolic Function [] Blocking bile acid reabsorption increases colonic bile acids levels [] Bile acids have a signaling function in the intestine, liver and other tissues that is mediated by receptors including TGR5; these signaling pathways have key functions in regulating insulin homeostasis [] Preclinical data supports improved metabolic function [] Clinical data shows that SHP626 (LUM002) reduced fasting glucose levels, and suggested improvements in glucose homeostasis 95 |  |
SHP626 (LUM002) Demonstrated Efficacy in Cholesterol-fed Hamsters; Reducing Serum LDL and Hepatic Cholesterol Levels SHP626 (LUM002) lowered Serum LDL and Hepatic Cholesterol in Cholesterol-fed hamsters after 3 weeks Normal chow group received standard chow, control group and all other groups a cholesterol-enriched diet (0.1%, w/w) [n = 6] Mean SEM, (1) p less than 0.05 normal chow versus control, (2) p less than 0.05 versus control LDL-Cholesterol Liver Cholesterol [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
SHP626 (LUM002) Demonstrated Reduction in Plasma Glucose and HbA1c in ZDF Rats [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 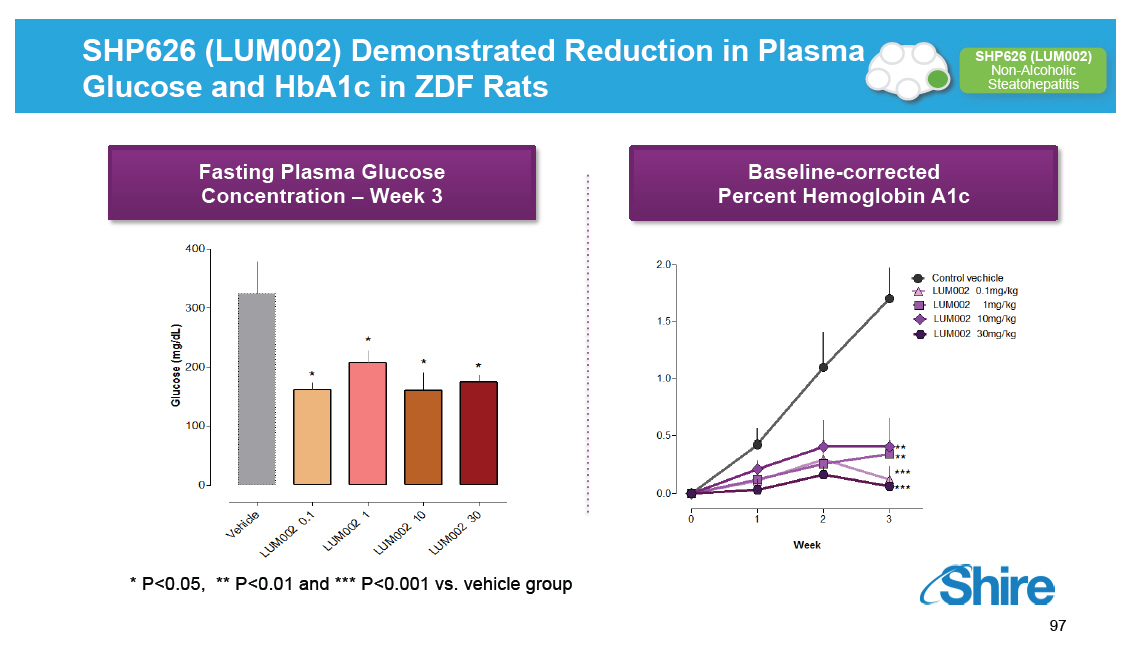 |
SHP626 (LUM002) Treatment Lowered LDL Cholesterol After 14 Days Oral Administration in Healthy Subjects [GRAPHIC OMITTED] SHP626 (LUM002) Non-Alcoholic Steatohepatitis Data expressed as percent change from the baseline value (Day -1) LUM002 50 mg PIB Subjects (n=12) were dosed once daily with SHP626 (LUM002) (50 mg/kg) for 14 days and followed for an additional 7-day period after the termination of dosing [GRAPHIC OMITTED] 98 | 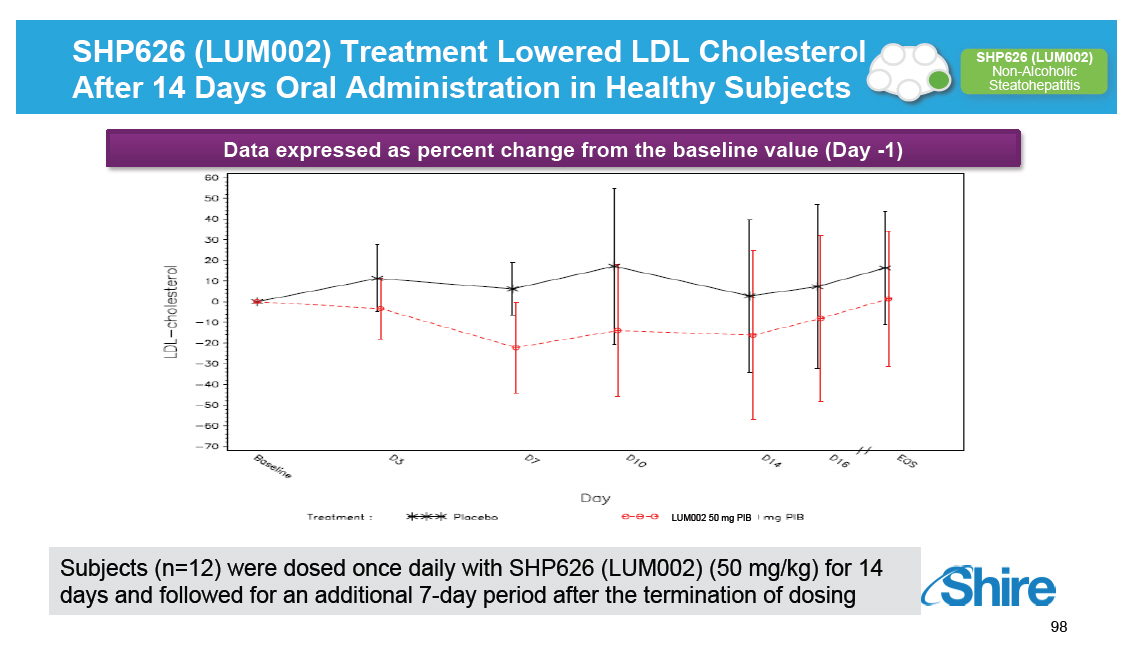 |
SHP626 (LUM002) Increased Fecal Bile Acid Levels in Healthy Volunteers, as Well as in T2DM(1) Patients [GRAPHIC OMITTED] SHP626 (LUM002) Non-Alcoholic Steatohepatitis Total fecal bile acids days 26-28 [48 hrs] in T2DM patients (mean [] SD) Healthy Volunteers T2DM (n-=48) (n=11) 3000 [] 2500 [] hrs) 2000 [] (umol/24 1500 [] 1000 [] Total Fecal Bile Acids 500 [] [] 2.2X [] 1.7X [] 2.0X [] 3.2X [] 8.1X 0 [] 0 mg 0.5 mg 1 mg 5 mg 10 mg 0 10 mg SHP626 (LUM002) Dose (1) Type 2 diabetes mellitus Pre-specified analysis *P [] 0.05, ** P []0.01 vs. placebo [GRAPHIC OMITTED] 99 |  |
SHP626 (LUM002) Showed Statistically Significant Reduction in Fasting Blood Glucose Levels at Day 14 and Day 28 in T2DM Patients [GRAPHIC OMITTED] SHP626 (LUM002) Non-Alcoholic Steatohepatitis Fasting blood glucose, absolute change from baseline (Day-1) SHP626 (LUM002) in T2DM patients (n=11) (mean [] SD) Fasting from (mmol/L) Change Glucose Absolute on Day -1 Baseline Placebo SHP626 (LUM002) (10 mg/day) Pre-specified analysis *P [] 0.05, ** P []0.01 vs. placebo [GRAPHIC OMITTED] 100 |  |
SHP626 (LUM002): Safety Profile [GRAPHIC OMITTED] SHP626 (LUM002) Non-Alcoholic Steatohepatitis [] SHP626 (LUM002) is minimally absorbed [] Single and multiple ascending dose and food effect studies complete (n = 153) [] 28 day study in T2DM patients (n = 11) [] Adverse events were mostly mild GI events [] The total rate of AEs decreased following first 2 weeks of treatment 60 Weeks 1 and 2 of GI AEs 40 Weeks 3 and 4 Incidence 20 0 Placebo 0.5 mg/ 1 mg/day 5 mg/day 10 mg/ Placebo 10 mg/ day day (T2DM) day (T2DM) [GRAPHIC OMITTED] 101 | 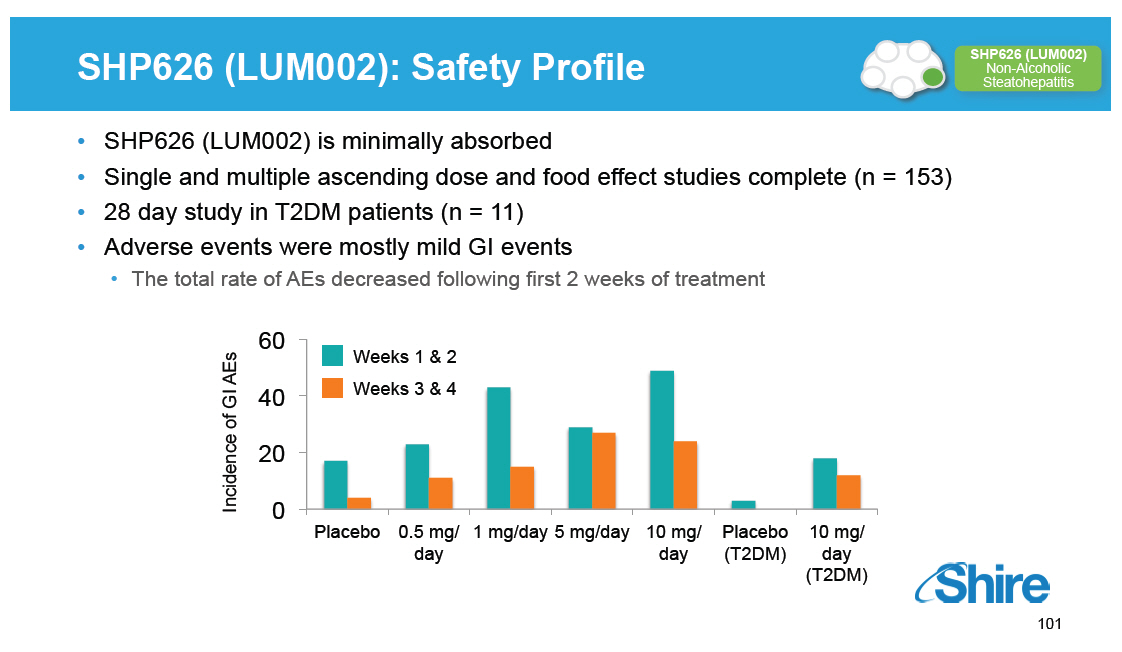 |
SHP626 (LUM002): Summary [GRAPHIC OMITTED] SHP626 (LUM002) Non-Alcoholic Steatohepatitis [] Highly potent and selective, minimally absorbed ASBT inhibitor [] Safety: [] Adverse events were mostly mild GI events [] No clinically significant elevations in lipids or triglycerides [] Phase 1 data supports: [] Increased fecal bile acid excretion [] Lowering of LDL [] Reduction in fasting blood glucose and trends towards insulin sensitivity in T2DM [] Phase 2 NASH study in planning: [] 52-week double-blind, randomized, placebo-controlled study [] Endpoints include: biopsy, biochemical markers, and imaging [GRAPHIC OMITTED] 102 |  |
Upcoming GI / Metabolic Rare Diseases Milestones [GRAPHIC OMITTED] Registration and Phase 4 Phase 3 Phase 2 Phase 1/2 SHP611 MLD Phase 1/2 head line data SHP625 (LUM001) ALGS Phase 2 head line data SHP606 Lifitegrast NDA Filing SHP625 (LUM001) PSC Phase 2 head line data SHP607 ROP Phase 2 head line data SHP606 Lifitegrast Launch(2) SHP609 Phase 2/3 head line data 20152015 VYVANSE BED Launch(2) SHP620 Maribavir Phase 2 head line data SHP625 (LUM001) PBC Phase 2 head line data SHP625 (LUM001) ALGS Phase 2 head line data 2016 SHP625 (LUM001) (1) PFIC Phase 2 head line data SHP610 Phase 2B head line data Notes (1)[] Interim 625 PFIC INDIGO data expected Q2 2015. (2)[] Subject to regulatory approval. [GRAPHIC OMITTED] 103 |  |
Break Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Rare Diseases Ophthalmics Norman Barton, M.D., PhD, Global Development Team Leader Our purpose We enable people with life-altering conditions to lead better lives. [GRAPHIC OMITTED] |  |
Today's RandD Sessions [GRAPHIC OMITTED] Topic Research Overview and Technology Platforms mRNA, Protein Replacement, Gene Therapy, Antibody Platforms Rare Diseases: GI / Metabolic SHP625 (LUM001), SHP626 (LUM002) Rare Diseases: Ophthalmics SHP607 / ROP, SHP630 / BIKAM Rare Diseases: Complement Biology and Renal / Fibrotic SHP616 / Cinryze new uses Rare Diseases: CNS SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen Late-Stage Update SHP606 / Lifitegrast, BED, SHP465 / ADHD Speaker Albert Seymour, Ph.D Ciara Kennedy, Ph.D David Piccoli, M.D. Norman Barton, M.D., Ph.D Howard Mayer, M.D. Howard Mayer, M.D. Howard Mayer, M.D. Randy Brenner Joe Tauber, M.D. Time (EST) 9:25-10:00 10:00-10.45 11:15-11:45 1:15-1:30 1:30-2:00 2:00-2:45 [GRAPHIC OMITTED] 106 |  |
Rare Diseases Programs in Ophthalmics Rare Diseases Leadership Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 [] (ALGS, PSC, [] PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP 555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT [GRAPHIC OMITTED] 107 |  |
SHP607: Prevention of Retinopathy of Prematurity (ROP) IGF-1 / IGFBP3 [GRAPHIC OMITTED] Preclinical 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 Phase 1 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis Phase 2 SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Phase 3 Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD Registration XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 108 [GRAPHIC OMITTED] | 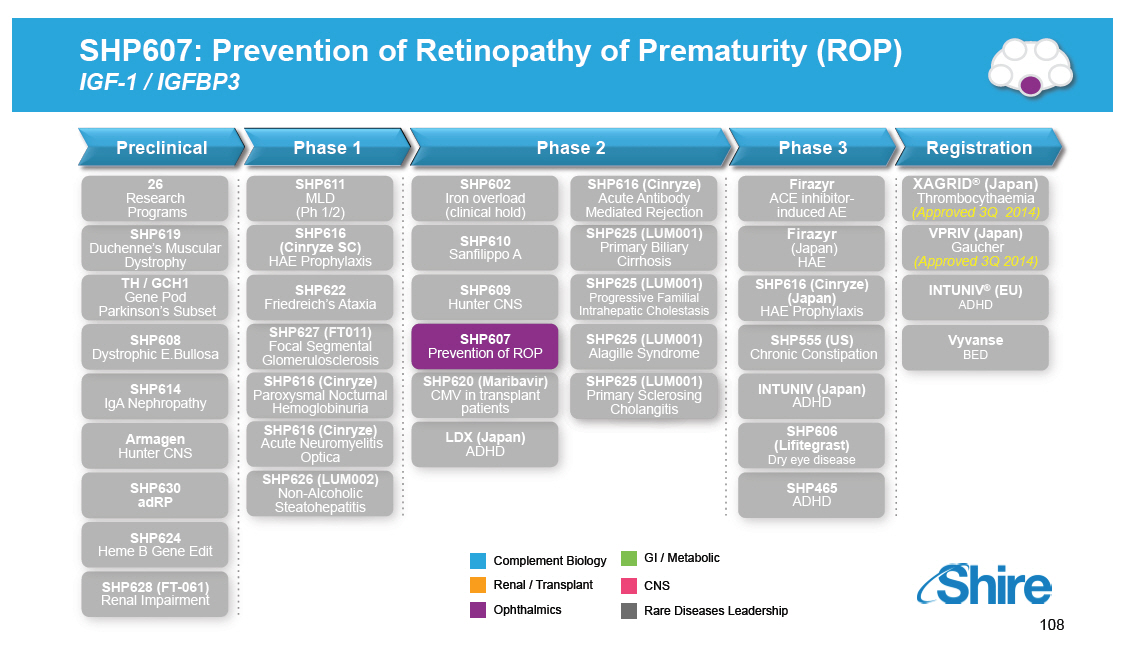 |
Prevention of Retinopathy of Prematurity (ROP) [GRAPHIC OMITTED] SHP607 Prevention of ROP Patients Product Progress Potential [] ~30K patients < 28 weeks gestational age (GA) in the US and a similar number in the EU per year [] Surgery is currently only widely recognized treatment option [] IGF-1 protein replacement therapy administered preventatively by continuous IV infusion beginning within the first 24-48 hours of life [] Delivered until endogenous production of IGF-1 begins at ~30 weeks GA [] Phase 2 studies ongoing with headline data expected 2H 2015 [] Dose selection completed [] Significant opportunity to treat a serious unmet need [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] 109 |  |
Retinopathy of Prematurity (ROP): Extent of Disease (Stages) [GRAPHIC OMITTED] SHP607 Prevention of ROP ROP STAGES [GRAPHIC OMITTED] Stage 1 Demarcation line [GRAPHIC OMITTED] Stage 2 Demarcation ridge [GRAPHIC OMITTED] Stage 3 Neovascularization [GRAPHIC OMITTED] Stage 4 Subtotal retinal detachment [GRAPHIC OMITTED] Stage 5 Total retinal detachment [GRAPHIC OMITTED] 110 |  |
ROP Images Z SHP607 Prevention of ROP Premature Retina with ROP Mature Healthy Retina (34 Weeks PMA /Birth at 28 Weeks GA) (40 Week Term Infant) [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Compelling Pre-clinical Evidence [GRAPHIC OMITTED] SHP607 Prevention of ROP [] Retinal vascularization is inhibited in IGF-1 KO mice despite the presence of other growth factors (VEGF) [] IGF-1 supplementation preserves retinal vasculature in oxygen induced retinopathy model [] IGF-1 receptor blockade in mice prevents retinal vascularization despite the presence of IGF-1 IGF-I -/- mouse [GRAPHIC OMITTED] WT mouse [GRAPHIC OMITTED] [GRAPHIC OMITTED] 112 | 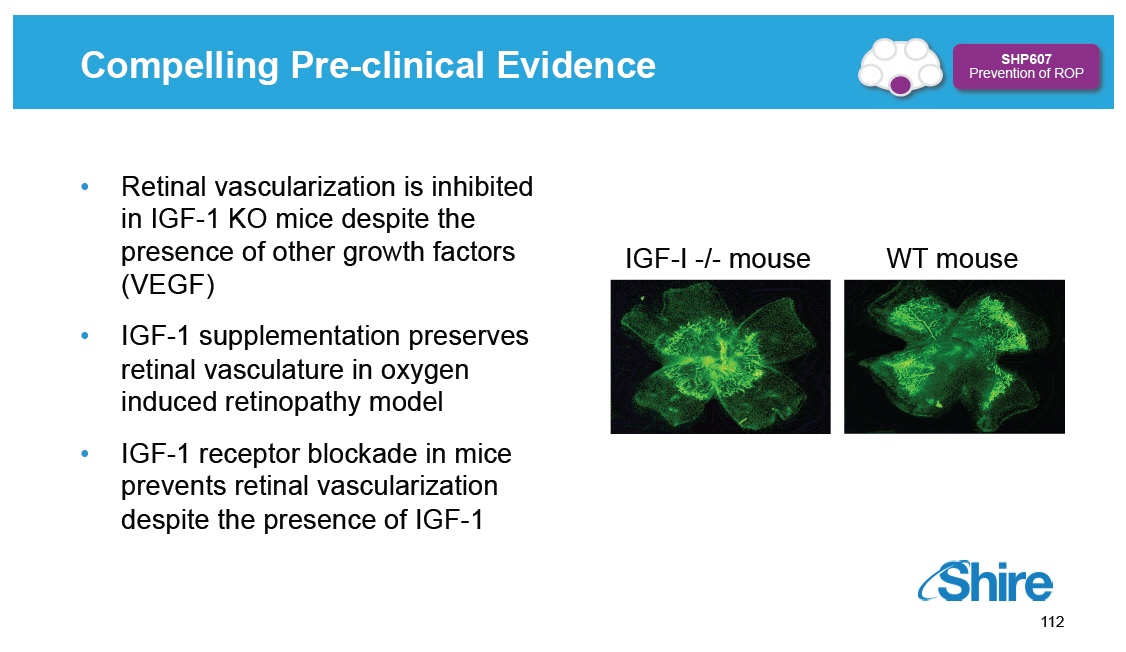 |
Intra-uterine IGF-1 Levels and the Correlation Between ROP and Serum IGF-1 Levels in Premature Infants [GRAPHIC OMITTED] SHP607 Prevention of ROP [GRAPHIC OMITTED] | 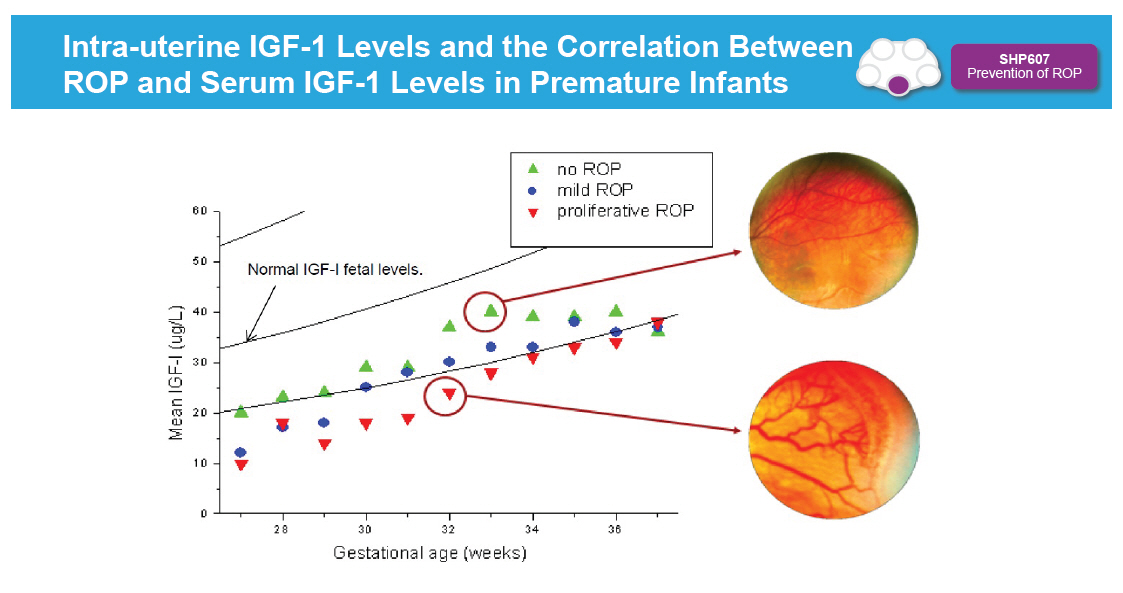 |
Retinopathy of Prematurity (ROP) IGF-1 and VEGF Roles in Development [GRAPHIC OMITTED] SHP607 Prevention of ROP In utero Normal vessel growth IGF-1 nl VEGF nl Premature birth Vessel growth stops IGF-1 nl ( VEGF) IGF-1 Maturing retina Hypoxia Slow IGF-1 VEGF Retinal neovascularization IGF-1 to "threshold" VEGF NI vessel growth in retina VEGF Resolution of ROP VEGF Proliferative retinopathy Retinal detachment VEGF IGF-1 [GRAPHIC OMITTED] 114 |  |
Phase II Trial Design [GRAPHIC OMITTED] SHP607 Prevention of ROP A Phase II, Open-Label, Multicenter, Dose Evaluation Study to Determine Safety and Efficacy of rhIGF-1 / rhIGFBP-3 in Premature Infants (Sections A, B, and C) COMPLETED Section A with MPA------------ N=5 GA 26+0 -- 27+6 Consultation Lund COMPLETED Section B N=5 GA 24+2 -- 27+2 Randomization Lund with MPA------------ Consultation Controls N=3 GA 23+3 -- 27+4 Lund ----------- COMPLETED Section C N=4 GA 23+0 -- 27+6 Randomization Lund and Karolinska SECTION D Controls N=7 GA 23+0 -- 27+6 Lund and Karolinska Trial designed in phased sections -- sections A, B and C completed by Premacure [] 24 patients included in Sections A, B and C [] Dosing per individualized algorithm and intense serum IGF-1 monitoring [GRAPHIC OMITTED] 115 | 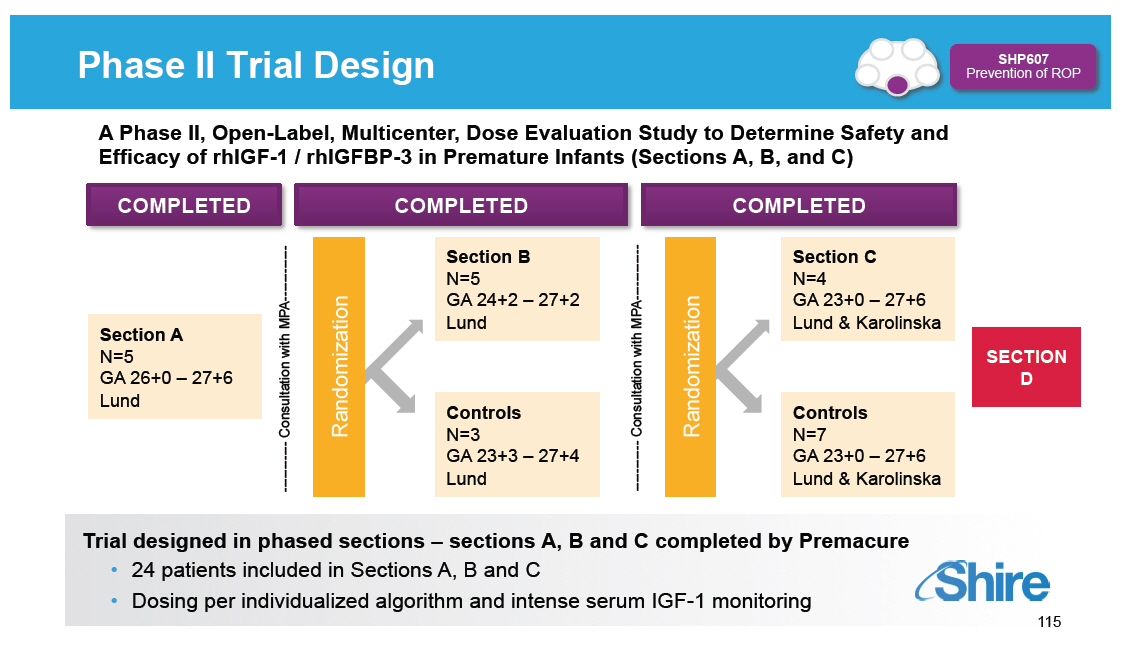 |
Results: Phase 1 and Phase 2 Sections A, B and C [] Number of exposures [] Phase 1: 5 active [] Phase 2: Sections A, B and C: 9 active and 10 SOC controls [] Results [] Elimination is rapid (T[] < 1hour); IGF-1/IGFBP-3 placement requires continuous IV infusion [] Average administered dose (~100 []g/kg/day) was insufficient to achieve physiologic replacement (range: 21-124 []g/kg/day) [] No safety signals [] Fewer total days of NICU care required for active treatment vs SOC [] One SOC control required laser treatment for ROP; none in the active treatment group [GRAPHIC OMITTED] 116 |  |
Simulated IGF-1 Levels Versus Dose Superimposed on Normal in Utero Levels [GRAPHIC OMITTED] SHP607 Prevention of ROP Dose of [] 250 ug/kg/24h is necessary to achieve therapeutic target 117 |  |
Next Steps: Ongoing Phase II in ROP [GRAPHIC OMITTED] SHP607 Prevention of ROP A Phase II, Open-Label, Multicenter, Dose Evaluation Study to Determine Safety and Efficacy rhIGF-1 / rhIGFBP-3 in Premature Infants (Section D) [] Randomized, treated versus standard of care control, assessor-masked trial [] Extremely pre-term infants (23 weeks -- 27 weeks + 6 days GA) included in trial [] Standardized dose developed: continuous IV infusion (250[]g/kg/24hrs) of rhIGF-1/rhIGFBP [] Primary endpoint: maximum severity of ROP stage across all retinal examinations (assessed at 40 weeks corrected gestational age) [] Key secondary endpoint: time to discharge from neonatal intensive care [] Additional secondary endpoints: incidence of BPD at 36 weeks PMA and brain volume by MRI at 40 weeks term equivalent [] 120 patients at 15-20 sites across Europe, US and Canada [] MOH approval in UK, Sweden, Italy, Netherlands, Poland, Canada; US IND accepted Nov 17, 2014 and fast track designation granted in December Headline data expected 2H2015 [GRAPHIC OMITTED] 118 |  |
SHP630: Autosomal Dominant Retinitis Pigmentosa (adRP) Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 119 [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 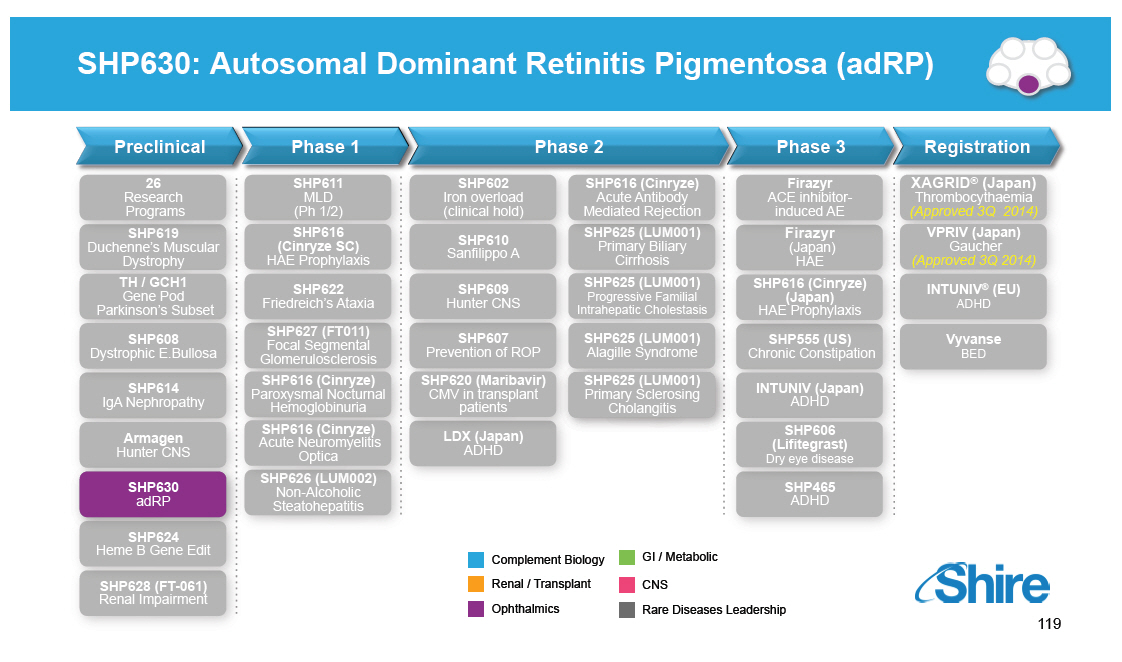 |
Autosomal Dominant Retinitis Pigmentosa (adRP) [GRAPHIC OMITTED] SHP630 adRP The Disease Prevalence and Gene Mutations Diagnosis Treatment Strategy [] adRP is an orphan genetic disease of the eye characterized by onset of night blindness in late childhood or adolescence followed by progressive loss of peripheral vision [] Most patients meet legal criteria for blindness between 40 and 70 [] Genetically heterogeneous with several disease causing genes; mutations in the opsin gene are a frequent cause of adRP [] ~ 75,000 patients with adRP worldwide [] ~ 15,000 carry a Class II mutation (mis-folded opsin) [] P23H, T17M, R135W account for 75% of Class II opsin mutations [] Retinal specialist (fundoscopic exam, visual field testing, electroretinogram and optical coherence tomography) [] Genotype provides confirmation of diagnosis [] No approved treatments for adRP [] SHP630 is designed to stabilize misfolded opsin, facilitate trafficking to the cell membrane and restore function [GRAPHIC OMITTED] 120 |  |
Opsin is Critical for Rod Photoreceptor Function Z SHP630 adRP Opsin binds to cis-retinal in membrane discs located in rod outer segment (ROS); opsin comprises 90% of all ROS protein Binding of opsin to cis-retinal is the first step in the visual cycle Class II mutations in opsin lead to reduced protein in ROS, leading to loss of ROS followed by rod photoreceptor cell death SHP630 is an orally available, non-retinal small molecule chaperone designed to facilitate opsin trafficking to the correct cellular location [GRAPHIC OMITTED] |  |
SHP630 is Designed to Preserve Rod Photoreceptor Structure and Function [GRAPHIC OMITTED] | 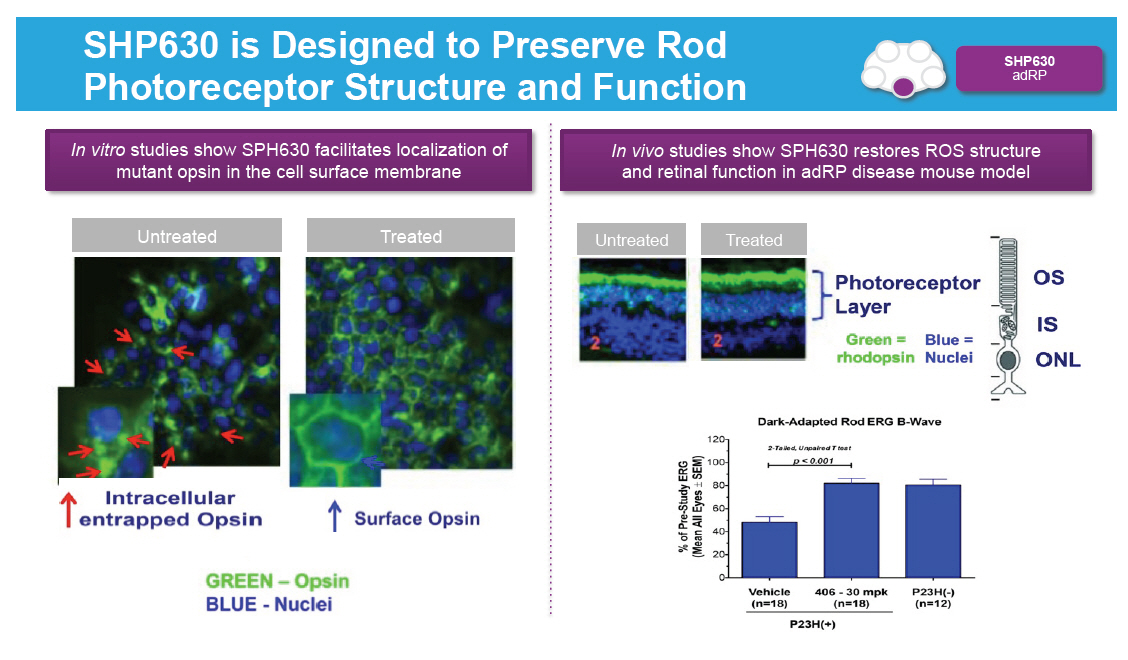 |
Upcoming Anticipated Ophthalmics Rare Diseases Milestones [GRAPHIC OMITTED] Registration and Phase 4 Phase 3 Phase 2 Phase 1/2 SHP611 MLD Phase 1/2 head line data SHP625 (LUM001) ALGS Phase 2 head line data SHP606 Lifitegrast NDA Filing SHP625 (LUM001) PSC Phase 2 head line data SHP607 ROP Phase 2 head line data SHP606 Lifitegrast Launch(2) SHP609 Phase 2/3 head line data 20152015 VYVANSE BED Launch(2) SHP620 Maribavir Phase 2 head line data SHP625 (LUM001) PBC Phase 2 head line data SHP625 (LUM001) ALGS Phase 2 head line data SHP625 (LUM001) (1) PFIC Phase 2 head line data SHP610 Phase 2B head line data Notes (1)[] Interim 625 PFIC INDIGO data expected Q2 2015. (2)[] Subject to regulatory approval. [GRAPHIC OMITTED] 123 |  |
Question and Answer Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Lunch Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Rare Diseases Complement Biology and Renal / Fibrotic CINRYZE([R]) New Uses[] [] Howard Mayer, M.D., Head of Clinical Development Our purpose We enable people with life-altering conditions to lead [GRAPHIC OMITTED] |  |
Today's RandD Sessions [GRAPHIC OMITTED] Topic Research Overview and Technology Platforms mRNA, Protein Replacement, Gene Therapy, Antibody Platforms Rare Diseases: GI / Metabolic SHP625 (LUM001), SHP626 (LUM002) Rare Diseases: Ophthalmology SHP607 / ROP, SHP630 / BIKAM Rare Diseases: Complement Biology and Renal / Fibrotic SHP616 / Cinryze new uses Rare Diseases: CNS SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen Late-Stage Update SHP606 / Lifitegrast, BED, SHP465 / ADHD Speaker Albert Seymour, Ph.D. Ciara Kennedy, Ph.D. David Piccoli, M.D. Norman Barton, M.D., Ph.D. Howard Mayer, M.D. Howard Mayer, M.D. Howard Mayer, M.D. Randy Brenner Joe Tauber, M.D. Time (EST) 9:25-10:00 10:00-10.45 11:15-11:45 1:15-1:30 1:30-2:00 2:00-2:45 [GRAPHIC OMITTED] 127 |  |
Cinryze New Use Programs in Complement and Renal Rare Diseases Leadership Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 [] (ALGS, PSC, [] PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP 555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT [GRAPHIC OMITTED] 128 |  |
CINRYZE (C1 INH):
Regulator of Inflammatory Cascades
[] Member of serine proteinase inhibitors ("serpins")
[] Heavily glycosylated 478 amino acid protein
[] Regulates several inflammatory cascades
[] Contact system: inhibition of factor XIIa and
kallikrein
[] Complement: inhibition of C1s, C1r and MASPs
[] Amplification loop of coagulation: inhibition of
factor XIa
[] Inhibition of Factor VII activating protease (FSAP)
[] Physiological role: to mitigate the pro-inflammatory
phase
of the acute phase response
[] Doubling of plasma concentrations later in acute phase
reaction
[GRAPHIC OMITTED]
[GRAPHIC OMITTED]
[GRAPHIC OMITTED]
129
|  |
CINRYZE Reduces Frequency of HAE Attacks by Regulating Contact Inflammatory Pathway Preventing Excess Bradykinin [GRAPHIC OMITTED] Triggering Event (toxins, injury, inflammation, ischemia, infection) Prekallikrein Factor XIIa Kallikrein HMWK Bradykinin HAE Deficiency of C1-INH Vascular permeability, vasodilation, pain [GRAPHIC OMITTED] [GRAPHIC OMITTED] Normal Condition [GRAPHIC OMITTED] Mild/Moderate [GRAPHIC OMITTED] Severe [GRAPHIC OMITTED] Very Severe [GRAPHIC OMITTED] 130 |  |
CINRYZE (C1 INH) Also Has Inhibitory Activity On All Three Complement Inflammatory Pathways* [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 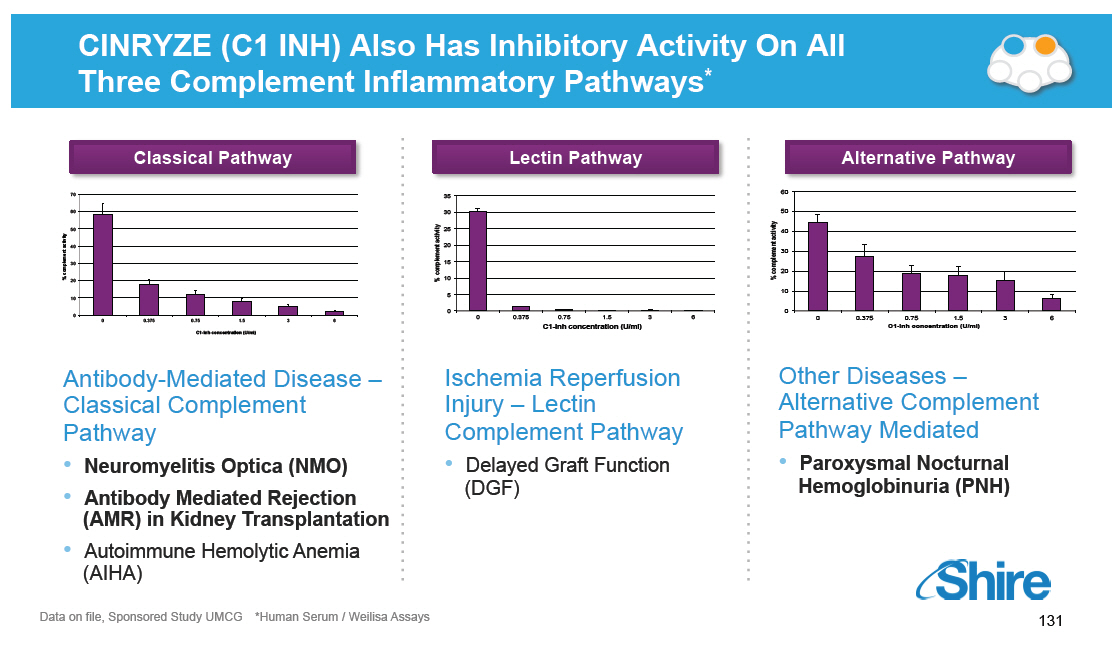 |
SHP616 (Cinryze): Acute Neuromyelitis Optica Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Neuromyelitis Optica (NMO) [GRAPHIC OMITTED] SHP616 (Cinryze) Acute Neuromyelitis Optica [] Inflammatory disease of the CNS that selectively attacks the optic nerve and spinal cord [] NMO disability accumulates through relapses; even a single relapse can have severe clinical consequence [] Prevalence: 1 -- 3 / 100,000 [] Associated with circulating IgG auto-antibodies against the astrocyte water channel protein aquaporin-4 (AQP4) "NMO-IgG" [] Results in antibody-mediated classical complement activation [] No approved treatment [] Steroids and plasmapheresis for acute attacks [] Immunosuppressants for prevention Wingerchuk D. AAN Continuum. 2010 [GRAPHIC OMITTED] 133 [GRAPHIC OMITTED] |  |
NMO Pilot Clinical Study Design [GRAPHIC OMITTED] SHP616 (Cinryze) Acute Neuromyelitis Optica Study enrolled between January -- August 2013 N = 10 patients with NMO / NMOSD Dosing Regimen Outcomes Top Line Results [] CINRYZE 2000 units/day on days 1-3 at onset of acute NMO attack [] Added on to 'standard of care': IV Solumedrol [] Primary: safety/tolerability [] Secondary: effectiveness as measured by EDSS [] No SAEs reported [] Generally well tolerated; no patients discontinued from study [] AE profile c/w known product profile *Expanded Disability Status Scale quantifies disability across eight functional systems with an overall score ranging from 0 (normal) to 10 (death). Scores above 5 indicate impaired ambulation. Levy M,Mealy MA. Purified human C1-esterase inhibitor is safe in acute relapses of neuromyelitis optica. Neurol Neuroimmunol Neuroinflammation vol 1 no.1 Published online April 24,2014 [GRAPHIC OMITTED] 134 |  |
NMO Pilot Clinical Study Results [GRAPHIC OMITTED] SHP616 (Cinryze) Acute Neuromyelitis Optica Expanded disability scale score (EDSS) P(1)=0.016 P(1)=0.0142 10 9 8 7 6 5 4 3 2 1 0 Baseline Actual Post- 30-day attack hospitali- follow-up zation Data suggest that C1 INH may be effective to limit neurologic damage and clinical disability from acute relapses: [] Majority of patients returned to their pre-attack level of neurologic function [] Only 2 patients required plasmapheresis (historical rate of about 40 -- 50%) Clinical Development next steps Meet with FDA in 1Q2015 to agree on the design of a Phase 2/3 placebo controlled trial in patients with acute NMO relapses *Expanded Disability Status Scale quantifies disability across eight functional systems with an overall score ranging from 0 (normal) to 10 (death). Scores above 5 indicate impaired ambulation. Levy M,Mealy MA. Purified human C1-esterase inhibitor is safe in acute relapses of neuromyelitis optica. Neurol Neuroimmunol Neuroinflammation vol 1 no.1 Published online April 24,2014 [GRAPHIC OMITTED] 135 |  |
SHP616 (Cinryze): Antibody-Mediated Rejection (AMR) in Kidney Transplantation Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 136 [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 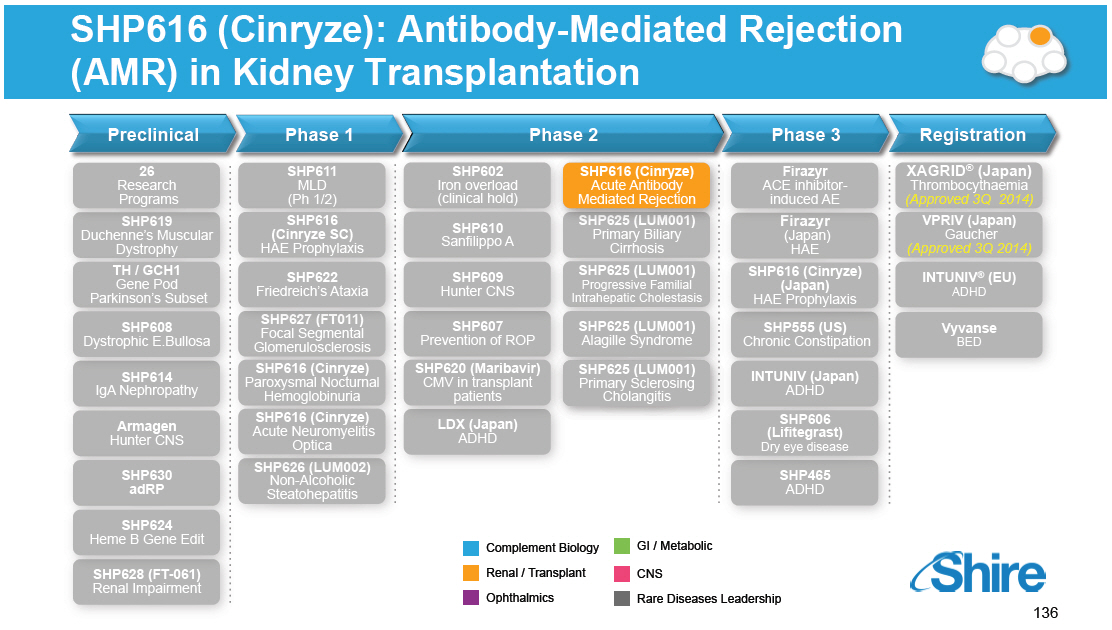 |
Antibody-Mediated Rejection (AMR) in Kidney Transplantation [GRAPHIC OMITTED] SHP616 (Cinryze) Acute Antibody Mediated Rejection [] Kidney transplant patients with donor specific antibody (DSA) have worse outcomes because of the high rate of AMR and resultant transplant glomerulopathy (tg) [] Patients with tg have a lower graft survival at one year (67% vs 97%; p<0.001) [] Loss of graft = poor quality of life, significant costs related to dialysis and related complications of renal failure [] 6,000 US patients have willing live donor but are DSA+ and do not get transplanted [] Acute AMR affects ~2,500 in US and EU5 [] 2.5% of standard transplants; >25% of DSA+ patients [] No approved treatments for AMR and current treatments not sufficient [] 50% fail standard of care (Plasmapheresis and IVIG) Sharif et al. Transplantation 2014;97: 541-547, Montgomery RA. Am J Transpl 2010;10: 449-457, Bentall et al. Am J Transpl 2013;13: 76-85, Haririan et al. Am J Transpl 2009;9: 2758-2767. [GRAPHIC OMITTED] 137 |  |
Antibody-Mediated Rejection in Kidney Transplantation Phase 2 Study Design [GRAPHIC OMITTED] SHP616 (Cinryze) Acute Antibody Mediated Rejection Double-blind, randomized, placebo-controlled, multicenter Phase 2 study of acute AMR in kidney transplant patients with donor specific antibody 18 patients enrolled [CINRYZE (n=9); Placebo (n=9)] Dosing Regimen Objectives Top Line Results [] CINRYZE 20,000 Units IV over 13 days (5,000 U bolus followed by 2,500 U on Days 3, 5, 7, 9, 11, 13) [] Placebo: IV 0.9% sodium chloride solution for infusion administered on Days 1, 3, 5, 7, 9, 11, and 13 [] Add on to standard of care: IVIG and / or plasmapheresis [] Safety, PK, Clinical Effect [] No discontinuations, study drug-related SAEs or deaths [] CINRYZE-treated subjects achieved higher exposure of C1 INH functional activity on Day 13, with baseline-corrected steady-state Cmax and AUClast for functional activity ranging from 3.7 - to 8.8 -fold higher than placebo subjects [] No difference in renal histopathological outcome 7 days after last dose 138 |  |
Antibody-Mediated Rejection in Kidney Transplantation Phase 2 Study Results (1/2) SHP616 (Cinryze) Acute Antibody Mediated Rejection Renal function (mean by cohort), measured as mean change in Creatinine Clearance (mL/min) 20 15 Mean change in Creatinine 10 Clearance mL/min 5 C1 INH 0 Placebo Base- Day Day Day Day Day Day line 3 5 7 9 11 13 Montgomery R, et al. Am J Transplant. 2014; 14(S3):129-130. In: Supplement: 2014 World Transplant Congress Abstracts, 26-31 July, 2014; San Francisco, CA. Abstract 2252. 139 [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 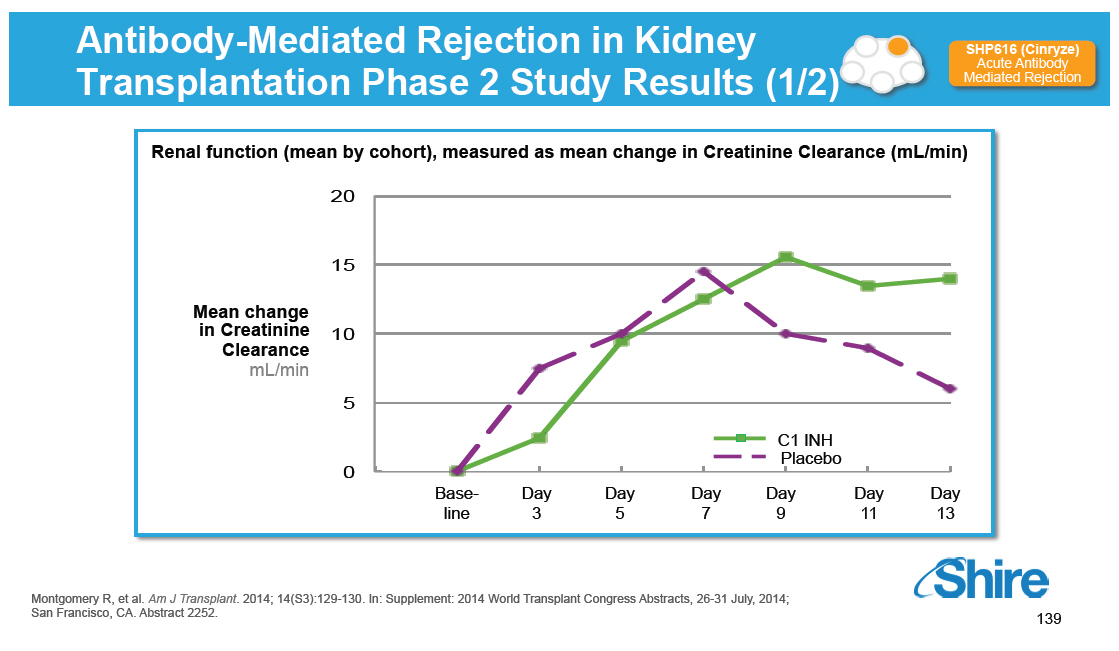 |
Transplant Glomerulopathy (tg) Seen in 3/7 PBO Patients and 0/7 CINRYZE Patients at 6 Months [GRAPHIC OMITTED] SHP616 (Cinryze) Acute Antibody Mediated Rejection [GRAPHIC OMITTED] normal CINRYZE subject [GRAPHIC OMITTED] [GRAPHIC OMITTED] tg Placebo subject [GRAPHIC OMITTED] WBC(2): inflammation Multilayered GBM (1) Chronic rejection = new onset transplant glomerulopathy (tg), a clinically accepted surrogate for accelerated loss of a kidney allograft (1) GBM refers to the glomerular basement membrane (2) White Blood Cells [GRAPHIC OMITTED] 140 |  |
Study Results (2/2): tg May be Related to Lower C1 INH Antigen Levels During Therapy for AMR [GRAPHIC OMITTED] SHP616 (Cinryze) Acute Antibody Mediated Rejection Patients developing post-transplantation glomerulopathy in the placebo arm had no C1 INH level above baseline Placebo Patient N[] C1 INH concentration (mg/dL) C1 INH Patient N[] C1 INH concentration (mg/dL) [] None of the patients who received C1 INH had post- transplant glomerulopathy [] This suggests that C1 INH may reduce the risks of AMR Clinical Development Next Steps [] Positive EOP2 feedback from FDA on the use of the accelerated approval pathway based on tg, with longer term confirmatory clinical data in the same study [] Pivotal registrational trial to start in 2015 Montgomery R, et al. Am J Transplant. 2014; 14(S3):129-130. In: Supplement: 2014 World Transplant Congress Abstracts, 26-31 July, 2014; San Francisco, CA. Abstract 2252. [GRAPHIC OMITTED] 141 | 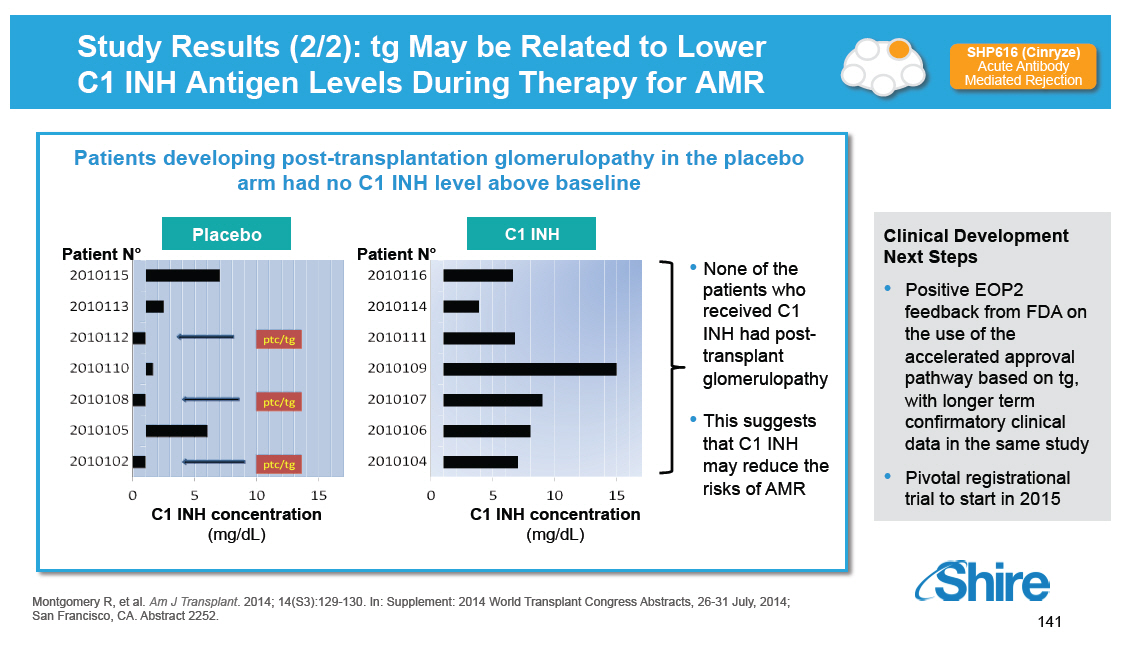 |
SHP616 (Cinryze): Paroxysmal Nocturnal Hemoglobinuria [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Paroxysmal Nocturnal Hemoglobinuria (PNH) [GRAPHIC OMITTED] SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria [] Clonal hematopoietic stem cell disorder [] Worldwide prevalence estimated at 1-5 cases / million [] Hemolytic anemia, bone marrow failure, thrombosis [] Acquired mutation of PIG-A gene [] PIG-A required for GPI--anchored protein biosynthesis [] CD55 inhibits C3 convertase [] CD59 blocks membrane attack complex (MAC) formation [] PNH cells have deficiency or absence of both CD 55/59 [] Eculizumab is an FDA-approved monoclonal antibody vs C5 protein that compensates for CD59 deficiency and reduces intravascular hemolysis and thrombosis risk in PNH patients [] Eculizumab does not compensate for CD55 deficiency which may result in extravascular hemolysis and persistent anemia DeZern AE et al, Exp Hematol 2014;42:857-861 Brodsky RA, Blood 2014;124:2804-2811 [GRAPHIC OMITTED] 143 |  |
PNH ex vivo Study Design [GRAPHIC OMITTED] SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria Sera from 6 PNH patients with type III proportion >5% All patients had been treated with eculizumab Clinical criteria for hemolysis noted at time of sampling Study Design [] PNH Erythrocytes were incubated with 25% acidified human serum with or without CINRYZE [] Part A -- optical density by spectrophotometry at 415 nm was used to calculate the percentage PNH RBC lysis [] Part B -- Flow cytometry was used to analyze deposition of C3 activation fragments on intact and lysed PNH erythrocytes (ghosts) DeZern AE et al, Exp Hematol 2014;42:857-861 [GRAPHIC OMITTED] 144 |  |
PNH ex vivo Study Results SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria [GRAPHIC OMITTED] [GRAPHIC OMITTED] 145 | 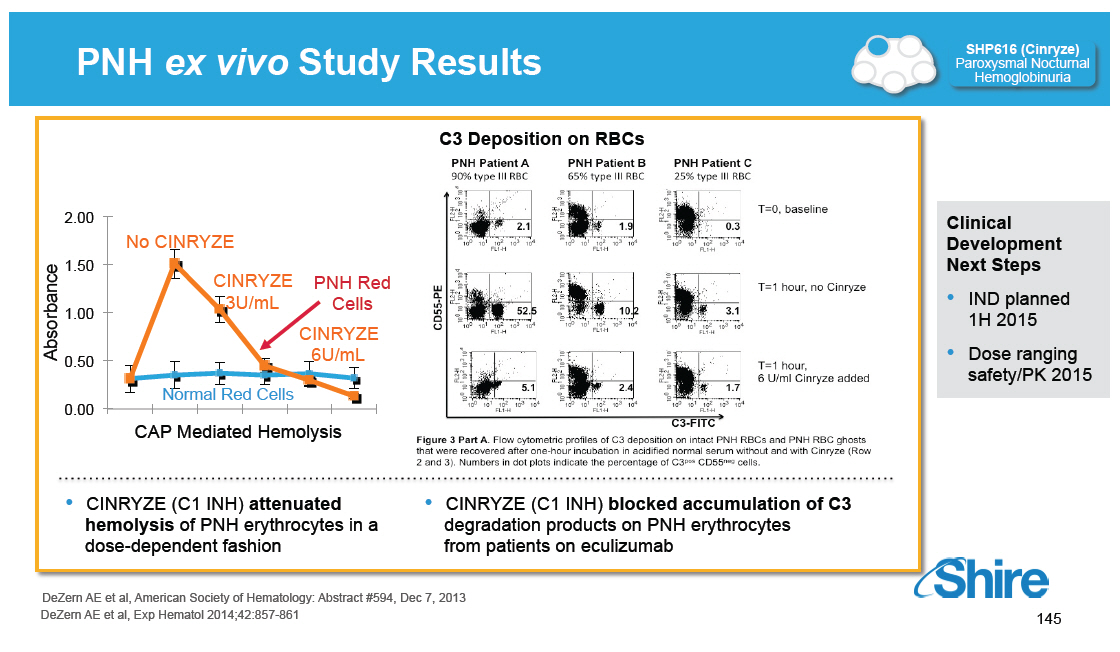 |
PNH ex vivo Study Results SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria [GRAPHIC OMITTED] [GRAPHIC OMITTED] 146 | 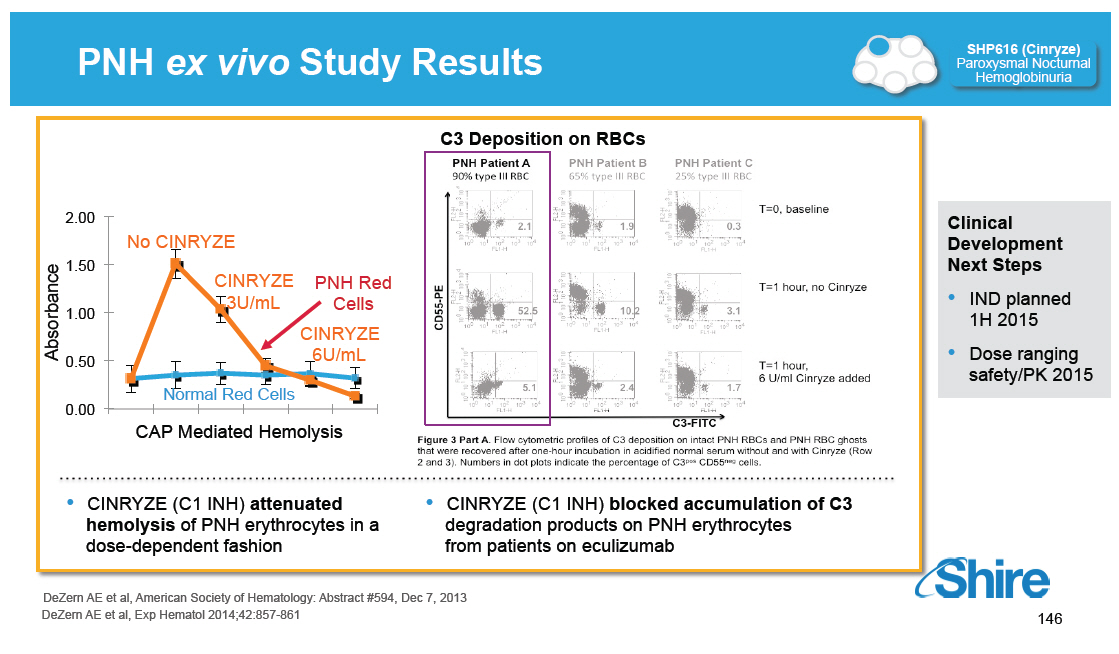 |
Next Steps for CINRYZE([R]) New Uses Programs Acute Neuromyelitis Optica Acute Antibody Mediated Rejection Paroxysmal Nocturnal Hemoglobinuria [] Meet with FDA in 1Q2015 on the design of a Phase 2/3 trial in patients with acute NMO relapses [] Positive feedback from FDA on accelerated approval pathway based on transplant glomerulopathy [] Pivotal registrational trial to start in 2015 [] IND planned 1H 2015 [] Dose ranging safety/PK study planned for 2015 [GRAPHIC OMITTED] 147 |  |
Rare Diseases: CNS[] Howard Mayer, M.D., Head of Clinical Development Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Today's RandD Sessions [GRAPHIC OMITTED] Research Overview and Technology Platforms mRNA, Protein Replacement, Gene Therapy, Antibody Platforms Rare Diseases: GI / Metabolic SHP625 (LUM001), SHP626 (LUM002) Rare Diseases: Ophthalmology SHP607 / ROP, SHP630 / BIKAM Rare Diseases: Complement Biology and Renal / Fibrotic SHP616 / Cinryze new uses Rare Diseases: CNS SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen Late-Stage Update SHP606 / Lifitegrast, BED, SHP465 / ADHD Albert Seymour, Ph.D. Ciara Kennedy, Ph.D. David Piccoli, M.D. Norman Barton, M.D., Ph.D. Howard Mayer, M.D. Howard Mayer, M.D. Howard Mayer, M.D. Randy Brenner Joe Tauber, M.D. 9:25-10:00 10:00-10.45 11:15-11:45 1:15-1:30 1:30-2:00 2:00-2:45 [GRAPHIC OMITTED] 149 |  |
Multiple Rare Diseases Programs in CNS Rare Diseases Leadership Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 [] (ALGS, PSC, [] PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP 555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT [GRAPHIC OMITTED] 150 | 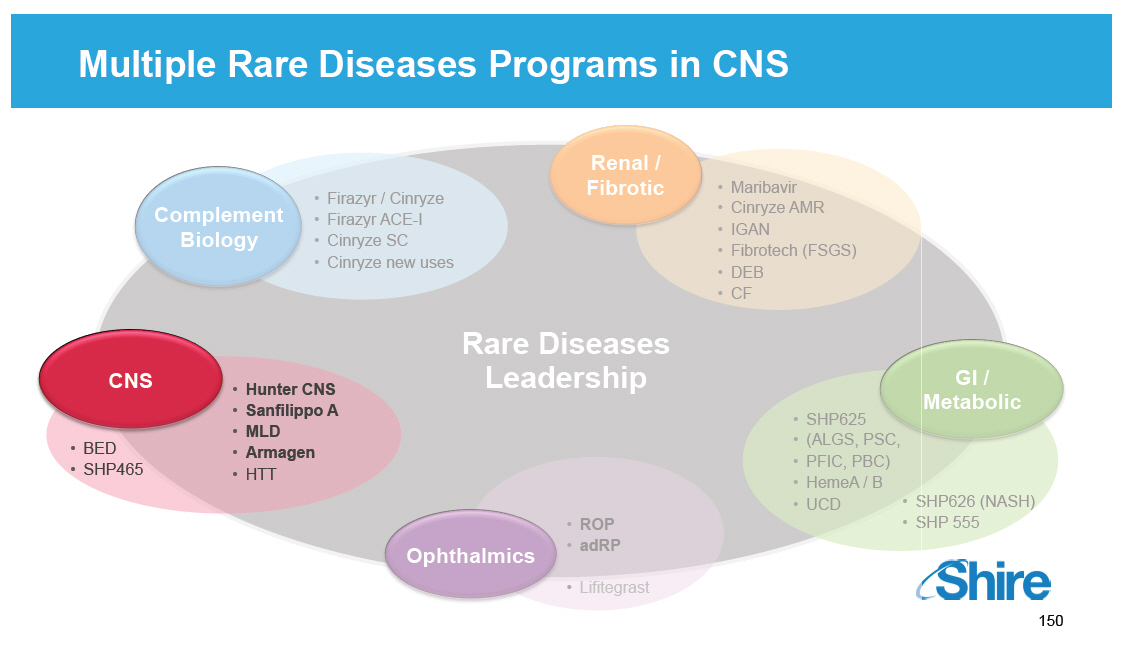 |
CNS Involvement by Lysosomal Storage Diseases (LSDs) [GRAPHIC OMITTED] Significant or severe No or minimal CNS CNS involvement involvement (~ 54%) (~ 46%) Meikle et al., JAMA 1999 [GRAPHIC OMITTED] 151 |  |
Intrathecal Drug Delivery Device (IDDD) [GRAPHIC OMITTED] [] To circumvent the Blood Brain Barrier, recombinant enzymes are administered into the subarachnoid space, via a transcutaneously accessible indwelling intrathecal drug delivery device (IDDD) [] Soph-A-Port([R]) Mini S is an implantable access system designed to provide repeated access to the intrathecal space for drug delivery [] It is CE marked in Europe and approved for investigational use in the US [] It is currently being used in Shire's Hunter-IT, Sanfilippo A and MLD clinical development programs [GRAPHIC OMITTED] [GRAPHIC OMITTED] 152 |  |
SHP609: Hunter Syndrome (MPS II) Recombinant human idursulfase-IT [GRAPHIC OMITTED] [GRAPHIC OMITTED] 153 | 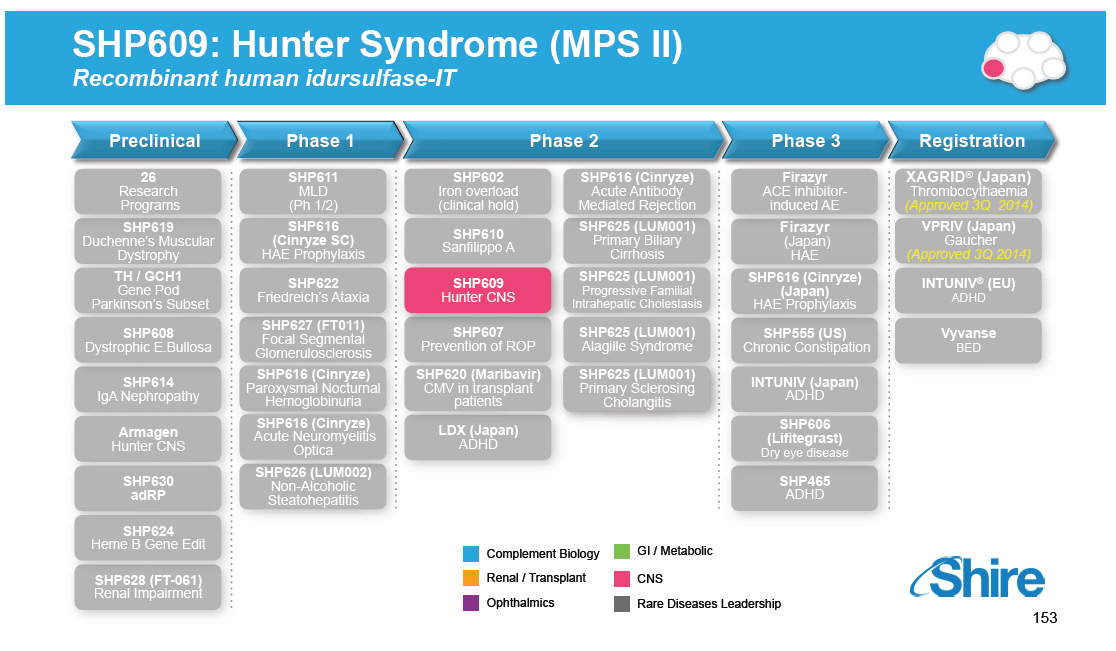 |
Mucopolysaccharidosis II -- Hunter Syndrome [GRAPHIC OMITTED] SHP609 Hunter CNS [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] A series of photographs showing the progression of the characteristic facial features of Hunter syndrome. The ages of the boy from left to right are 6 months and 5, 9 and 30 years. [] Extremely rare X-linked lysosomal storage disease [] Incidence ~ 1 in 170,000 male births [] Absence or deficient activity of the lysosomal enzyme iduronate-2-sulfatase (I2S) [] Diagnosis typically at 2-6 years, prompted by typical appearance (coarse facial features), organomegaly or developmental delays [] Multiple physical issues caused by deposits of glycosaminoglycans in the soft tissues of upper respiratory tract, joints, heart, liver and spleen [] 2/3 of patients experience progressive developmental delay and cognitive decline, usually leading to death in the teenage years [] Idursulfase (ELAPRASE) is an intravenous enzyme replacement therapy which addresses some of the somatic issues but does not address the cognitive issues Muenzer et al., Pediatrics 2009; Martin et al., 2008 |  |
Spectrum of Disease in Hunter Syndrome [GRAPHIC OMITTED] SHP609 Hunter CNS Attenuated [GRAPHIC OMITTED] [] Insidious onset [] Normal intelligence [] Variable life expectancy [GRAPHIC OMITTED] [GRAPHIC OMITTED] [] Onset by 2 to 4 years of age [] Impaired intelligence [] Life expectancy 10 to 15 years Severe |  |
Uptake of Labeled Iduronate-2-Sulfatase (I2S) in Cynomolgus Monkey After Intrathecal and ICV(1) Delivery [GRAPHIC OMITTED] SHP609 Hunter CNS In vivo distribution of 124I -labeled I2S (3 mg/animal) in cynomolgus monkeys by PET [GRAPHIC OMITTED] [GRAPHIC OMITTED] Distribution of I2S administered through the lumbar (left) and ICV (right) catheters 30 minutes after the administration as demonstrated by a projection PET image (sum of all slices). Relative linear color scale. The distribution of I2S in the brain at 0.5, 2.5, 5 and 24 hours after lumbar administration; PET image, 1.2 mm slice through the corpus callosum region in the plane parallel to the occipital bone. The color scale is calibrated in mg/ml of I2S. 1 Intracerebroventricular Papisov et al., PLOS 2012 [GRAPHIC OMITTED] 156 | 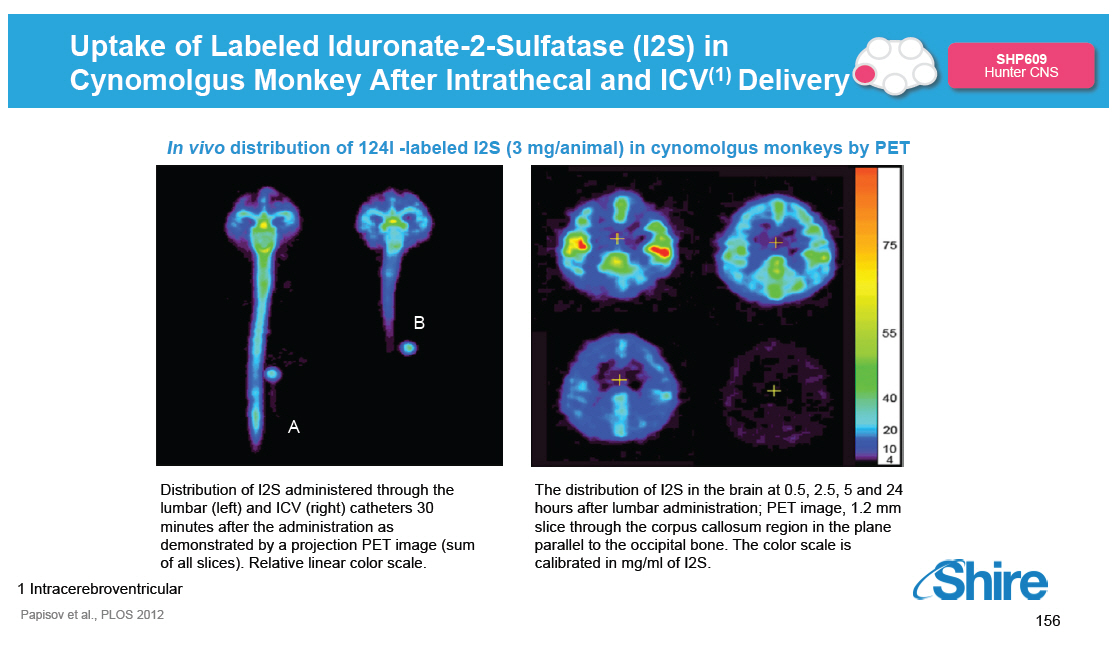 |
Phase 1/2: Multiple-dose Dose-escalation Study in MPS II [GRAPHIC OMITTED] SHP609 Hunter CNS [] Study drug was idursulfase-IT, a formulation of recombinant iduronate-2-sulfatase that is different from that used for IV idursulfase treatment [] Four patients per treatment arm (n=16) Treatment Arms Inclusion Criteria Primary Endpoint Secondary Endpoint Exploratory Efficacy Endpoints [] 10, 30 and 1 mg of idursulfase-IT and no-treatment [] Administered via IDDD or lumbar puncture [] Dosed monthly for 6 months [] All patients received weekly IV idursulfase 0.5 mg/kg IV [] Ages 3 to18 years [] Cognitive impairment due to MPS II (GCA [] 77) [] History of tolerating weekly IV idursulfase 0.5 mg/kg for at least 6 months [] Safety and tolerability of ascending doses of idursulfase-IT [] Change From Baseline in CSF Glycosaminoglycans [GAGs] at 6 months [] Cognitive / adaptive behavioral testing at 0 and 6 months [] Differential Abilities Scale-II (DAS-II) [] Preferred method in the study and can be performed with cognitively impaired children [] Yields the General Conceptual Ability (GCA), which is constructed like an IQ [] All eligible patients rolled over into an extension trial, currently there are 14 patients in the extension trial |  |
MPSII: Effect of Idursulfase-IT Administration on Biomarkers in a Phase I-II Study and Extension [GRAPHIC OMITTED] SHP609 Hunter CNS Note: Time 0 is the phase [] (HGT-HIT-045) randomization date for treated patients in HGT-HIT-045 and is the date of receipt of the informed consent form for the extension study (HGT-HIT-046) from untreated patients in HGT-HIT-045. Week 3 is the date of the first dose of idursulfase-IT. CSF, cerebrospinal fluid; GAG, glycosaminoglycan; IT, intrathecal. [GRAPHIC OMITTED] Investigational Intrathecal Enzyme Replacement Therapy for the Severe Form of Hunter Syndrome (Mucopolysaccharidosis II) Joseph Muenzer et al., poster 172, LDN 2014 158 |  |
MPSII: Effect of Idursulfase-IT on Cognitive Performance in a Phase I-II Study and Extension [GRAPHIC OMITTED] SHP609 Hunter CNS [] At Month 9, Patient 9 was switched from 1 mg idursulfase-IT monthly in the phase [] trial to 10mg monthly in the extension study. Note: Time 0 is the phase [] (HGT-HIT-045) randomization date for treated patients in HGT-HIT-045 and is the date of receipt of the informed consent form for the extension study (HGT-HIT-046) from untreated patients in HFT-HIT-045. Week 3 is the date of the the first dose of idursulfase-IT. DAS-II, Differential Ability Scales-Second Edition; GCA, general conceptual ability; IT, intrathecal. [GRAPHIC OMITTED] Investigational Intrathecal Enzyme Replacement Therapy for the Severe Form of Hunter Syndrome (Mucopolysaccharidosis II) Joseph Muenzer et al., poster 172, LDN 2014 159 | 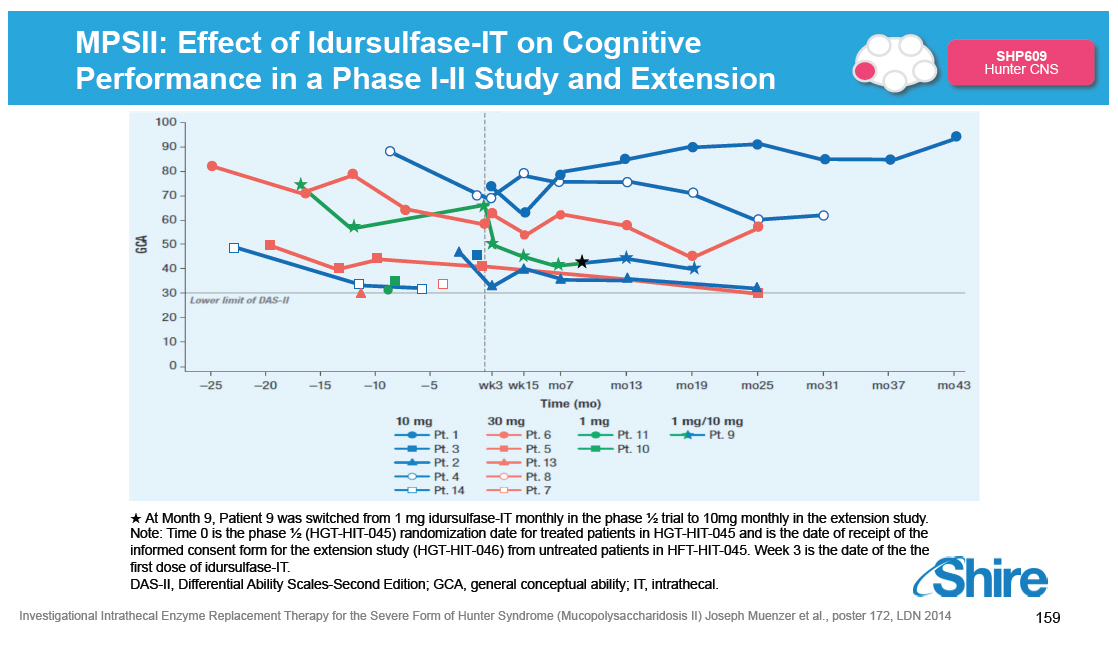 |
MPSII: Effect of Idursulfase-IT on Cognitive Performance in Mild-Moderate Impaired Patients [GRAPHIC OMITTED] SHP609 Hunter CNS 94 (BL 74) 62 (SC/BL 70/69) 57 (SC/BL 59/63) [] At Month 9, Patient 9 was switched from 1 mg idursulfase-IT monthly in the phase [] trial to 10mg monthly in the extension study. Note: Time 0 is the phase [] (HGT-HIT-045) randomization date for treated patients in HGT-HIT-045 and is the date of receipt of the informed consent form for the extension study (HGT-HIT-046) from untreated patients in HFT-HIT-045. Week 3 is the date of the the first dose of idursulfase-IT. DAS-II, Differential Ability Scales-Second Edition; GCA, general conceptual ability; IT, intrathecal. [GRAPHIC OMITTED] Investigational Intrathecal Enzyme Replacement Therapy for the Severe Form of Hunter Syndrome (Mucopolysaccharidosis II) Joseph Muenzer et al., poster 172, LDN 2014 160 |  |
Hunter-IT Current Phase II/III Pivotal Study Actively Enrolling, Headline Data Expected Mid 2016* [GRAPHIC OMITTED] SHP609 Hunter CNS Phase II-III (094 study) [] Multicenter, international, randomized, assessor-blinded [] 42 patients, to be randomized 2:1 (28 treated, 14 untreated) Inclusion Criteria Dosage Primary Endpoint Key Secondary Endpoint [] Ages 3 to18 years [] Hunter Syndrome and mild to moderate cognitive impairment (between 85 and 55 GCA as measured by DAS-II) [] 12 monthly doses via IDDD [] Dose level of idursulfase-IT - 10 mg [] Change in General Conceptual Ability (GCA) obtained by the Differential Abilities Scale (DAS-II) [] Independent / adaptive function, measured by Vineland Adaptive Behavior Scales (VABS) * Subject to enrollment timelines and interactions with Regulatory Authorities |  |
SHP610: SANFILIPPO A (MPS IIIA) Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 162 [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
MPSIIIA Disease Overview [GRAPHIC OMITTED] SHP610 Sanfilippo A [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] [GRAPHIC OMITTED] [] Autosomal recessive lysosomal storage disease: mutations in SGSH, encoding heparan N sulfatase (HNS) [] Live birth incidence ~ 1 in 100,000 [] Enzyme defect causes accumulation of heparan sulfate [] Clinical features are overwhelmingly neurological [] Normal early infancy [] Developmental delays often first manifestations [] Severe behavioral disturbances are a prominent feature of middle childhood [] Progressive dementia leads to a "quiet phase" of withdrawal and developmental regression [] Survival to late teens / early 20s [] Primary accumulation of the glycosaminoglycan (GAG) heparan sulfate triggers poorly understood pathological cascade with primarily CNS manifestations Wijburg, Acta Pediatrica 2013 [GRAPHIC OMITTED] 163 |  |
Phase 1/2 Study with Once-Monthly IT-rHNS [GRAPHIC OMITTED] SHP610 Sanfilippo A Pharmacodynamic Analysis Revealed Dose-dependent Suppression of CSF GAG* in MPS IIIA [] Pharmacodynamic data demonstrated in vivo biological activity and provide basis for Phase IIB dose selection [] Safety results [] No deaths [] No discontinuations [] No SAEs related to investigational drug *GAG: glycosaminoglycans Wijburg et al. Am Coll Med Human Genetics Meeting 2013, poster 392 [GRAPHIC OMITTED] 164 |  |
SanA Phase IIB Study Focusing on Patient Population Believed Most Likely to Benefit from IT rhHNS [GRAPHIC OMITTED] SHP610 Sanfilippo A 1 0 0 t n tie o u Q l DQ = 60 ta n e 5 0 m p lo e v e D 0 0 1 0 0 2 0 0 3 0 0 A g e ( M o n t h s ) 10mg monthly 45 mg monthy 90 mg monthly Natural History Phase IIB entry criteria: Age [] 48 months, DQ [] 60 [] No children in Phase I/II (SAN-055) met these criteria vs 8/25 in Natural history study [] Median decline in DQ: 23 points in 12 months among the 8/25 patients in Natural history study [GRAPHIC OMITTED] 165 | 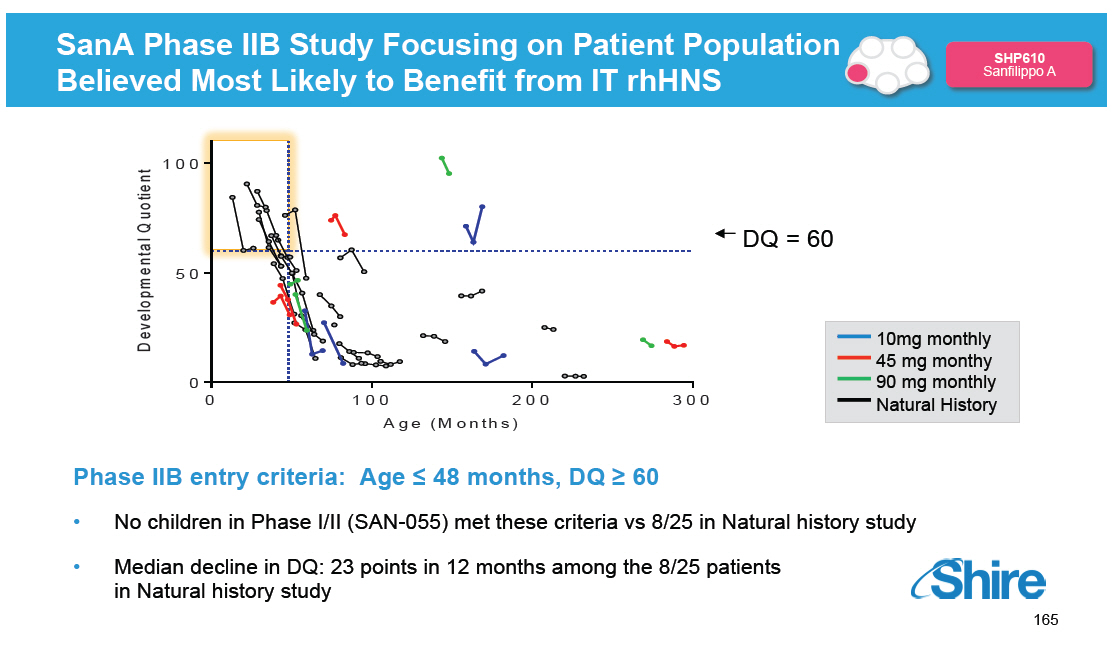 |
SHP610: Phase IIb Proof-of-Concept Study Actively Enrolling, Headline Data Expected Mid 2016* [GRAPHIC OMITTED] SHP610 Sanfilippo A [] A randomized, open-label, parallel group, controlled, multicenter study [] 45 mg rHNS-IT administered either every 2 weeks (Q2W) or every 4 weeks (Q4W) via an IDDD, versus no treatment -- 6 patients per group Primary Objective To assess the potential clinical efficacy of rHNS administered via a surgically implanted IDDD in patients with MPS IIIA. Efficacy will be measured as the number of patients with no more than 10 point loss over 12 months as measured by the Bayley Scales of Infant and Toddler Development, 3rd Edition (BSID-III). * Subject to enrollment timelines and interactions with Regulatory Authorities [GRAPHIC OMITTED] 166 |  |
SHP611: METACHROMATIC LEUKODYSTROPHY (MLD) Recombinant human arylsulfatase A (rhASA) Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 167 [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Metachromatic Leukodystrophy: Disease Overview [GRAPHIC OMITTED] SHP611 MLD Manifestations of MLD [] MLD results from deficiency of the lysosomal enzyme arylsulfatase-A [] Arylsulfatase-A breaks down sulfatides and accumulation causes nerve demyelination Disease Summary [] An inherited leukodystrophy [] Inheritance is autosomal recessive [] Birth incidence 1 in 100,000 [] Three different phenotypic presentations classified by age of onset: late-infantile, juvenile, and adult [] Motor weakness and cognitive loss the most prominent symptoms [] Uniformly fatal; earlier onset correlates with more rapid decline [] Management focused on palliative care [] No treatments currently available [] Significant negative impact on patients and caregiver quality of life [] High economic costs [GRAPHIC OMITTED] Haberlandt et al., Eur. J. Ped Neuro 2009 168 |  |
Distribution and PK of (124)l-labeled ARSA [GRAPHIC OMITTED] SHP611 MLD In vivo Distribution and PK of (124)l-labeled ARSA (6 mg) at t=1hr Post-dose in Cynomolgus Monkeys by PET / CT Imaging (124)I-labeled rhASA IT-L dose = 6 mg 1 hr post-dose Image courtesy of Papisov group at MGH 169 | 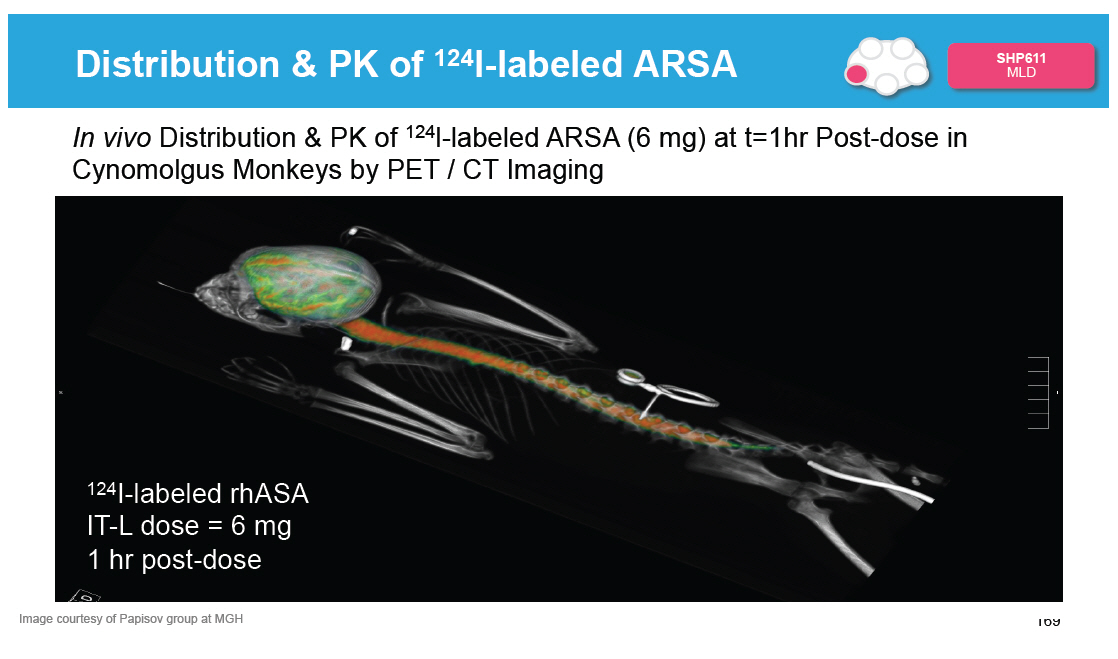 |
SHP611: Phase I/II Dose Escalation Study with rASA Fully Enrolled (1), Headline Data Expected Mid 2015 [GRAPHIC OMITTED] SHP611 MLD [] Dose escalation study to evaluate safety of 3 doses administered EOW [] Study duration is 40 weeks [] 18 patients have been enrolled in 3 dosing cohorts (10, 30, 100 mg every other week) Primary Objective Secondary Objectives Key Inclusion Criteria Key Exclusion Criteria [] Safety of ascending doses in children [] Clinical activity on gross motor function [] Effects on other key clinical signs and symptoms [] On serum / CSF pharmacokinetics [] Appearance of first symptoms at or before 30 months of age [] Able to walk at the time of screening [] Neurological signs must be present at screening [] History of bone marrow transplantation (1) All But 1 Eligible Patient In Extension Study * Subject to enrollment timelines and interactions with Regulatory Authorities |  |
AGT-182: HUNTER SYNDROME Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 171 [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Armagen's Platform For Delivery to Target the Brain: IgG molecular Trojan-Horse Fusion Protein [GRAPHIC OMITTED] Armagen Hunter CNS Fusion protein consisting of chimeric MAb against the human insulin receptor(HIR) fused with a recombinant protein (e.g. iduronate-2-sulfatase, L-iduronidase) HEAD [GRAPHIC OMITTED] Molecular Trojan Horse A genetically engineered monoclonal antibody (MAb) against the human insulin receptor (HIR) TAIL [GRAPHIC OMITTED] Recombinant Protein [GRAPHIC OMITTED] IgG Fusion Protein: A new chemical entity Data provided by Armagen [GRAPHIC OMITTED] 172 | 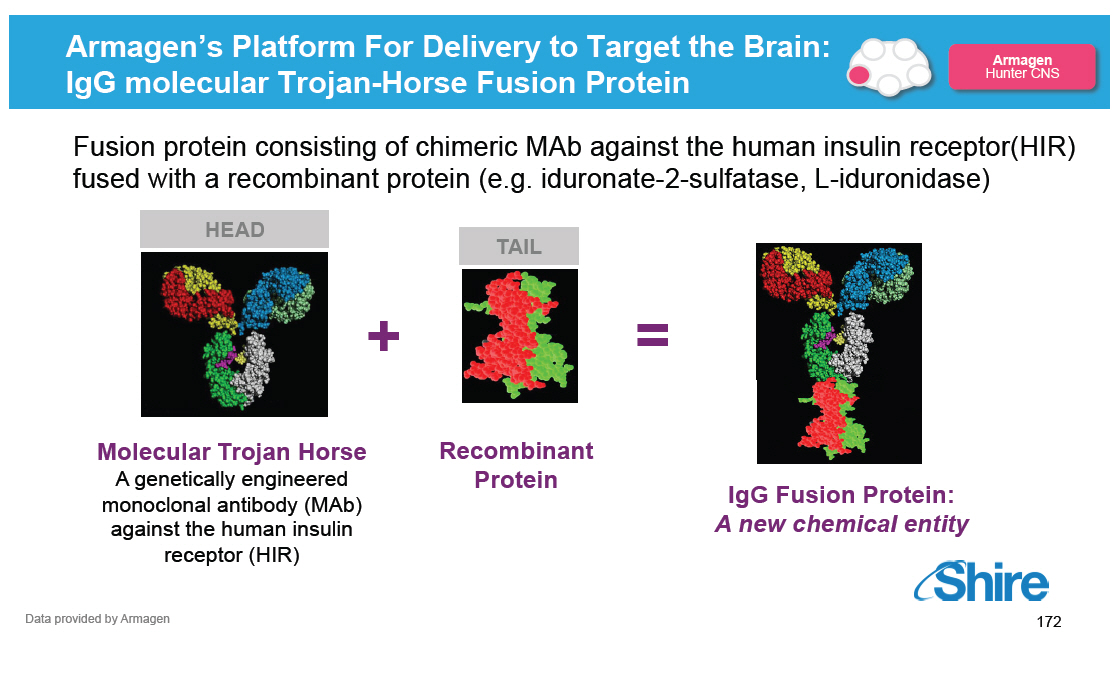 |
Utilizing Endogenous Transporter To Cross Blood Brain Barrier [GRAPHIC OMITTED] Armagen Hunter CNS Fusion protein consisting of chimeric MAb against the human insulin receptor(HIR) fused with a recombinant protein (e.g. iduronate-2-sulfatase, L-iduronidase). 173 |  |
Data to Support MOA: Distribution to Brain Tissue in Rhesus Monkey [GRAPHIC OMITTED] Armagen Hunter CNS [] Systemic distribution similar to ELAPRASE([R]) [] Autoradiography demonstrated the extensive distribution of drug into all regions of the brain (at 2 hours after IV injection) versus ELAPRASE [GRAPHIC OMITTED] ELAPRASE-alone [GRAPHIC OMITTED] AGT-182 Published Bioconjugate Chem. 2013, 24, 1741[]1749 [GRAPHIC OMITTED] 174 |  |
AGT-182: Next Steps Phase 1/2 Expected to Start Early 2015* [GRAPHIC OMITTED] Armagen Hunter CNS [] US IND submitted October 2014 [] A Phase 1/2, Dose-Exploratory, Clinical Study of Human Insulin Receptor Monoclonal Antibody-Human Iduronate-2-Sulfatase (IDS) Fusion Protein, AGT-182 in adult patients with MPSII is being planned to start in early 2015 [] Next study -- is expected to be in children with cognitive impairment due to Hunter Syndrome * Subject to enrollment timelines and interactions with Regulatory Authorities [GRAPHIC OMITTED] 175 |  |
Upcoming Anticipated CNS Rare Diseases Milestones [GRAPHIC OMITTED] Registration and Phase 4 Phase 3 Phase 2 Phase 1/2 SHP611 MLD Phase 1/2 head line data SHP625 (LUM001) ALGS Phase 2 head line data SHP606 Lifitegrast NDA Filing SHP625 (LUM001) PSC Phase 2 head line data SHP607 ROP Phase 2 head line data SHP606 Lifitegrast Launch(2) SHP609 Phase 2/3 head line data 20152015 VYVANSE BED Launch(2) SHP620 Maribavir Phase 2 head line data SHP625 (LUM001) PBC Phase 2 head line data SHP625 (LUM001) ALGS Phase 2 head line data 2016 SHP625 (LUM001) (1) PFIC Phase 2 head line data SHP610 Phase 2B head line data Notes (1)[] Interim 625 PFIC INDIGO data expected Q2 2015. (2)[] Subject to regulatory approval. [GRAPHIC OMITTED] 176 |  |
Late-Stage Pipeline Update Howard Mayer, M.D., Head of Clinical Development Randy Brenner, Head of Regulatory Affairs Joe Tauber, M.D., Tauber Eye Center, Kansas City, MO Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Today's RandD Sessions [GRAPHIC OMITTED] Topic Research Overview and Technology Platforms mRNA, Protein Replacement, Gene Therapy, Antibody Platforms Rare Diseases: GI / Metabolic SHP625 (LUM001), SHP626 (LUM002) Rare Diseases: Ophthalmics SHP607 / ROP, SHP630 / BIKAM Rare Diseases: Complement Biology and Renal / Fibrotic SHP616 / Cinryze new uses Rare Diseases: CNS SHP609 / Hunter CNS, SHP610 / Sanfilippo A, SHP611 / MLD, Armagen Late-Stage Update SHP606 / Lifitegrast, BED, SHP465 / ADHD Speaker Albert Seymour, Ph.D. Ciara Kennedy, Ph.D. David Piccoli, M.D. Norman Barton, M.D., Ph.D. Howard Mayer, M.D. Howard Mayer, M.D. Howard Mayer, M.D. Randy Brenner Joe Tauber, M.D. Time (EST) 9:25-10:00 10:00-10.45 11:15-11:45 1:15-1:30 1:30-2:00 2:00-2:45 178 |  |
Late-Stage Programs in CNS and Ophthalmics Rare Diseases Leadership Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 [] (ALGS, PSC, [] PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP 555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT [GRAPHIC OMITTED] 179 |  |
Late-Stage Pipeline Update -- Agenda Dry Eye 2014 Lifitegrast for the treatment of Dry Eye Disease Late-Stage Regulatory Update [] Vyvanse for Binge Eating Disorder [] SHP465 for Attention Deficit Hyperactivity Disorder Joe Tauber, M.D. Howard Mayer, M.D. Randy Brenner [GRAPHIC OMITTED] 180 |  |
Dry Eye 2014 JOSEPH TAUBER, M.D. TAUBER EYE CENTER KANSAS CITY, MO [GRAPHIC OMITTED] |  |
The Eye Surface Requires Lubrication to Adequately Perform its Primary Functions of: 1.[] Barrier protection 2.[] Visual function [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Dry Eye is a Significant Market Opportunity[] It is the Primary or Related Cause of 40% of Eye Care Visits (1) Women: 4% @ 40 [] 15% @ 65 Men: 4% @ 50 [] 7.7% @ 80+ u[] 1/10 dry eye patients in U.S. has Sjogren's syndrome u[] 1.5 M Rheumatoid Arthritis (CDC, 2005) u[] 37% of diabetics (Canadian Dry Eye Epidem. Study) u[] $4B in health care costs (Yu J, et al, Cornea 2011) (1) Shire has used the following prevalence estimates: patients diagnosed with DED from U.S. studies vary from 0.39% in 1998 to 18.8% of patients seeking treatment in VA eye clinics from 2006-2011. Overall U.S. prevalence by self-reported symptoms has been estimated to be 7.8% of females aged 50 and older and 4.34% of males aged 50 and older. Using this self-reported symptom data and U.S. census estimates for 2012, over 2.1 M males and over 4.3 M females over the age of 50 have either been clinically diagnosed with DED or have severe dry eye symptoms. Market Scope. 2010 Comprehensive Report on The Global Dry Eye Products Market. | 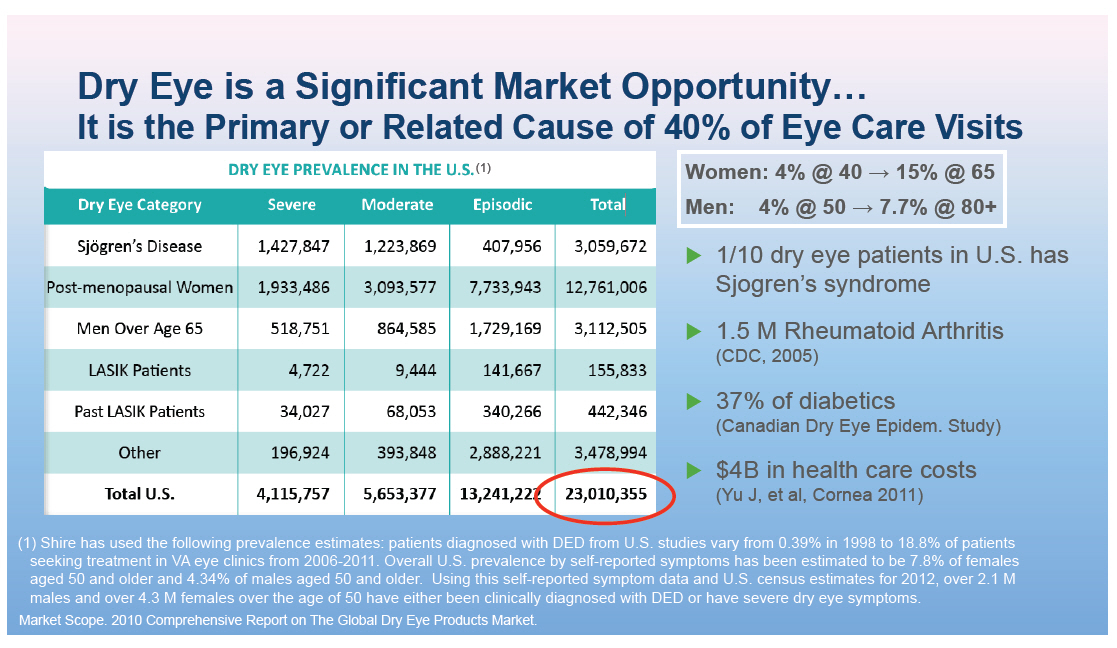 |
For all Eye Care Providers[] Contact lens discomfort Blurred vision Address pre-surgical risk proactively Dry Eye Rx Skill is Critical Better surgical outcomes Happy patients referrals Practice Building ($) If you found it before surgery and there are postop issues -- it is because of the disease. If you didn't -- patients will believe it's your fault. |  |
For Patients, Dry Eye is not a Trivial Matter
Mean
Health State Score
Moderate Dry Eye Disease* 0.78
Moderate Angina* 0.75
L[] LASIK
patients:
48% have
dry eye
[GRAPHIC OMITTED]
[GRAPHIC OMITTED]
[GRAPHIC OMITTED]
[GRAPHIC OMITTED]
Schiffman RM, et al. Ophthalmology 2003 Jul;110(7):1412-9.
Salom[]o MQ et al. J Cataract Refract Surg. 2009
Oct;35(10):1756-60. doi: 10.1016/j. jcrs.2009.05.032.
Raoof D et al. Semin Ophthalmol. 2014 Nov;29(5-6):358-62.
doi: 10.3109/08820538.2014.962663.
|  |
Unmet Needs in Dry Eye u[] Cure is rare or non-existent u[] Disease incidence may be growing, independent of improved diagnosis u[] Aging population, increasing incidence with age, menopause u[] Growth of prostaglandin eye drop Rx for glaucoma u[] Increasing patient demand for better control of symptoms u[] Growth of premium IOL cataract surgery, with increased insistence on high grade vision after more expensive surgery |  |
Dry Eye is an Inflammatory Disease CD4+ T Cell Infiltration [GRAPHIC OMITTED] CHRONIC INFLAMMATION [GRAPHIC OMITTED] MHC Class II Upregulation [GRAPHIC OMITTED] ICAM-1 Upregulation [GRAPHIC OMITTED] T Cell Homing to the Ocular Surface Antigen Presentation T Cell Activation |  |
ABC's of "First Line" Treatment In "Doctor speak" Supplementation Add tears Stimulation Stimulate more tears Retention Block tears from leaving in Patient's Terms NB -- Only 1/3 of these strategies reduces inflammation |  |
 |
How Do We Measure Symptoms? CATEGORICAL SCALES 0 - 3 Dryness Pain, soreness Burning Sandiness, grittiness Blurred vision Discharge Itching None, mild, moderate severe None, mild, moderate severe None, mild, moderate severe None, mild, moderate severe None, mild, moderate severe None, mild, moderate severe None, mild, moderate severe VISUAL ANALOGUE SCALES 0 100 VISUAL ANALOGUE SCALES WITH ANCHORS 0 50 100 |  |
How Do We Measure Symptoms? 1.[] McMonnies Questionnaire 2.[] DEQ, The Dry Eye Questionnaire 3.[] VFQ-25 NEI-devised Visual Functioning Questionnaire 4.[] VT-HRQ, Vision-Targeted Health-Related Quality of Life, a questionnaire that evaluates QOL activities related to or dependent upon vision 5.[] NEI-VFQ NEI, Visual Function Questionnaire, a questionnaire developed by the National Eye Institute to evaluate vision function inactivities of daily life 6.[] OSDI, Ocular Surface Disease Index, a set of questions assessing the level of discomfort and interference with activities of daily living produced by ocular surface disease. Gothwal et al. Invest Ophthamol Vis Sci 2010;51:1401-1407. 2007 Report of the International Dry Eye Workshop (DEWS). The Ocular Surface 2007; 5:65-204. |  |
How Do We Measure Signs?
[GRAPHIC OMITTED]
Fluorescein
cornea
[GRAPHIC OMITTED]
Lissamine
Green
conjunctiva
Grading Schemes for Staining
Oxford 0-3, entire cornea
NEI 0-4, 5 zones
Modified 0-4, 5 zones
NEI micro, macropunctate
ORA unique system
of conj/cornea zones
Degree of staining increases over time (1-2-3-4 min)
-- only recent trials have specified time in grading method
|  |
Dry Eye Development -- Ten+ Years of Terminated Clinical Trials u[]Diquafasol u[]Rebamipide u[]Hyaluronidase tears u[]Androgen tears u[]Ecabet sodium |  |
ORA's Controlled Adverse Environment (CAE) for Dry Eye u[] Controlled airflow u[] Controlled humidity u[] Monitored blink rate u[] Quantitative tear film breakup u[] Functional visual assessment |  |
Ocular Surface Disease Rx Algorithm Tear Underproduction Supplement Uncontrolled Higher viscosity Tears P.F. Tears Supradose Restasis Serum Tears Secretagogue (oral, SL) Clinical Trials Stimulate Retain Controlled Lid Disease Posterior Lid Hygiene Oral Rx (macrolides) OM-3 supplements MG Duct Probing Anti-keratin Rx (Vitamin A, "scraping") Lipiflow Anterior Soaks / Scrubs Demodex Vitamin A |  |
Late-Stage Pipeline Update -- Agenda Dry Eye 2014 Lifitegrast for the treatment of Dry Eye Disease Late-Stage Regulatory Update [] Vyvanse for Binge Eating Disorder [] SHP465 for Attention Deficit Hyperactivity Disorder Joe Tauber, MD Howard Mayer, M.D. Randy Brenner [GRAPHIC OMITTED] 196 |  |
SHP606 (Lifitegrast): Dry Eye Disease Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 197 [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 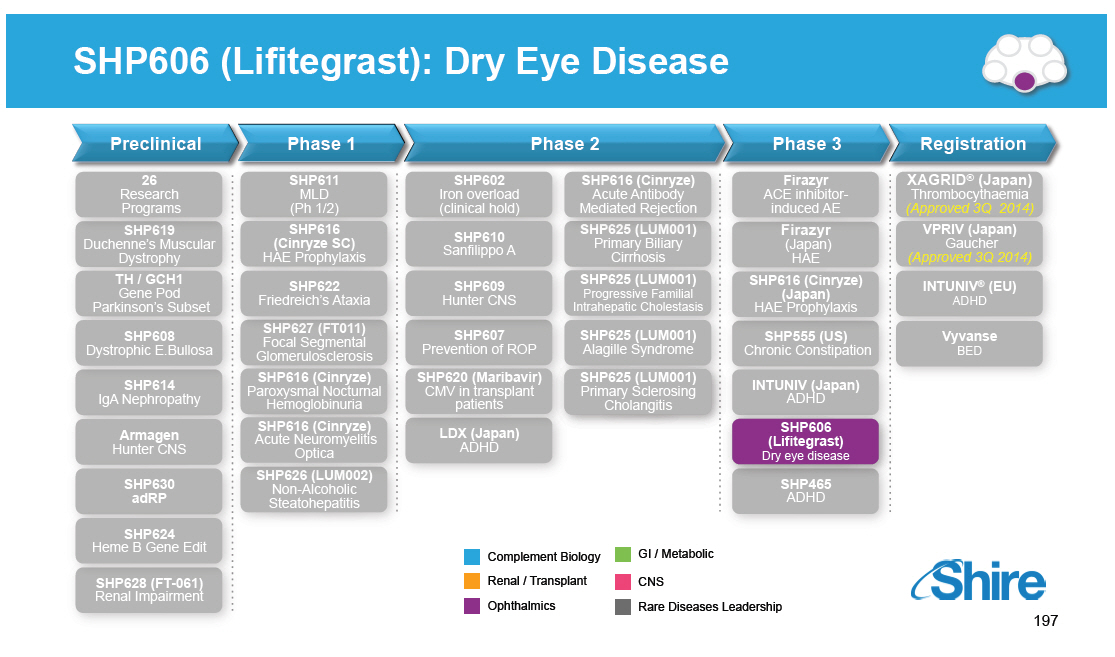 |
Lifitegrast -- A Purpose-Built Molecule [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease [] Selected from 1500 candidate molecules [] Specifically engineered as topical ophthalmic [] Built to potently block LFA-1 / ICAM-1 interaction [] Adhesion to ICAM-1 (IC50 = 2 nM) [] Cytokine release (SEB stimulated IL-2) IC50 >> 60nM [] Highly stable and hydrophilic [] water/ saline/ pH 7 >>2 years [] >200 mgs/mL at pH 7 [] No systemic accumulation with rapid clearance [] Selectively targets LFA-1 on infiltrating leukocytes [] Selected based on its properties for further development as a candidate to treat Dry Eye Disease [GRAPHIC OMITTED] Lifitegrast (formerly SAR 1118) MW 610 g/mol 1.[] Ref.:USAN/INN (Jan 2012) 2.[] Zhong et al., Med Chem Lett (2012) [GRAPHIC OMITTED] 198 |  |
Summary of the Overall Lifitegrast Program [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease Phase 1a 1b 2 2 3 3 3 Study Number SAR1118-001 1118-DME-100 (Johns Hopkins, IST 1118-ACJ-100 Phase 2 OPUS-1 OPUS-2 SONATA Indication Healthy Volunteers, Safety, PK PK Vitrectomy Allergic Conjunctivitis Dry Eye Dry Eye Dry Eye Dry Eye Subjects 28 30 60 230 588 718 332 Status Complete Complete Complete Complete Complete Complete Complete [GRAPHIC OMITTED] 199 |  |
Results and Findings From 3 Key Efficacy Studies [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease Phase 2 [] Dose response relationship for signs and symptoms; 5% solution chosen for further evaluation [] Met pre-specified secondary sign endpoint change from baseline to Day 84 in ICSS; established as the sign endpoint for future studies [] Several pre-specified symptom endpoints trended or met significance particularly in patients with history of artificial tear use Learnings and development progression OPUS-1 [] Co-primary sign endpoint met significance validating Phase 2 findings (p=0.0007) [] Co-primary symptom endpoint (Visual Related Function Subscale) not met [] Pre-specified subpopulation of subjects with history of AT use showed greater treatment effect on EDS [] Post hoc analysis of more symptomatic AT subgroup with EDS[]40 met significance vs placebo (nominal p=0.0178) Learnings and development progression OPUS-2 [] Co-primary symptom endpoint met significance validating OPUS-1 findings in this population of moderate to severely symptomatic subjects (EDS[]40) with history of AT use (p<0.0001) [] All pre-specified secondary symptom endpoints achieved statistical significance [] Co-primary sign endpoint failed to separate from placebo in this more symptomatic population and did not demonstrate significance vs placebo ICSS: Inferior Corneal Staining Score EDS: Eye Disease Score |  |
Why Different Outcomes in Different Studies? Multifactorial Etiologies but Likely Influenced by Population Differences [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease DESIGN SIGN SYMPTOM KEY OBSERVATION Phase 2 and OPUS-1 [] CAE(1) for subject selection [] Inclusion and exclusion [] Similar sites (New England) [] Similar seasonality (fall / winter) Mild-moderate(2) Mild-moderate(2) OPUS-2 [] No CAE(1) [] Thresholds [] West / South [] All year Moderate-severe(3) Moderate-severe(3) [] Mild-to-moderate subjects enhance detection of the sign [] Moderate-to-severe subjects enhance detection of symptoms [] Enriching population for one variable lead to loss of detection of the co-variable [] Co-primary may not be practical 1.[] Studies 1 and 2 were conducted in collaboration with ORA and utilized ORA's Controlled Adverse Environment (CAE[]) as a clinical model to study the treatment of dry eye disease with lifitegrast. 2.[] As defined per protocol, subjects with ICSS of greater than 3.0 were not allowed in the trial. As defined per protocol, subjects had to have worsening of ODS by +3 points and as such severely symptomatic subjects could not enroll. 3.[] As defined per protocol, subjects with EDS of greater than or equal to 40 could only enroll into the trial and there were no outer limits to the ICSS and as such subjects with the highest degree of staining (+4) could also enroll. [GRAPHIC OMITTED] 201 |  |
Feedback from FDA [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease Pre-NDA meeting held on May 15 with the division of Ophthalmology and Transplant Medicine of the FDA [] With respect to the evidence collected from the current program to date and its suitability for an NDA submission, the Agency acknowledged that: The clinical portion of an NDA with the current clinical data package was likely fileable. Approvability of a submitted NDA would be a review issue. [] Regarding totality of clinical evidence and the paradoxical relationship between the sign and symptom co-variables, FDA acknowledged that: Safety and efficacy is recommended to be demonstrated in at least two adequate and well-controlled, multi-center, independent trials. You may wish to consider demonstrating efficacy based on subjective findings in a different patient group or in a different clinical study than the patient group or clinical study which demonstrates efficacy based on objective findings. [GRAPHIC OMITTED] 202 |  |
Evidence of Replication in Sign
Inferior Corneal Staining Score
[GRAPHIC OMITTED]
SHP606
(Lifitegrast)
Dry eye disease
Phase 2
Phase 2 (ATU)
OPUS-1
OPUS-1 (ATU)
OPUS-1 (EDS <40)
Table 1. Inferior Corneal Staining Score (ICSS)
All Subjects
Phase 2 OPUS-1
PBO vs. 5.0% LIF PBO vs. 5.0% LIF
(N=55:N=54) (a) (N=294:N=293) (b)
Treatment Effect 0.35 0.24
(95% CI) (0.05, 0.65) (0.10, 0.38)
P-value 0.0209 0.0007
Artificial Tear Users
Phase 2 OPUS-1
PBO vs. 5.0% LIF PBO vs. 5.0% LIF
(N=29:N=28) (c) (N=128:N=128) (d)
0.76 0.28
(0.38, 1.14) (0.06, 0.5)
0.0002 0.0127
Phase 2 OPUS-1
All Subjects Subjects with
PBO vs. 5.0% LIF Baseline EDS<40
(N=55:N=54) (a) (N=147:N=137) (c)
0.35 0.41
(0.05, 0.65) (0.21, 0.60)
0.0209 <0.0001
| 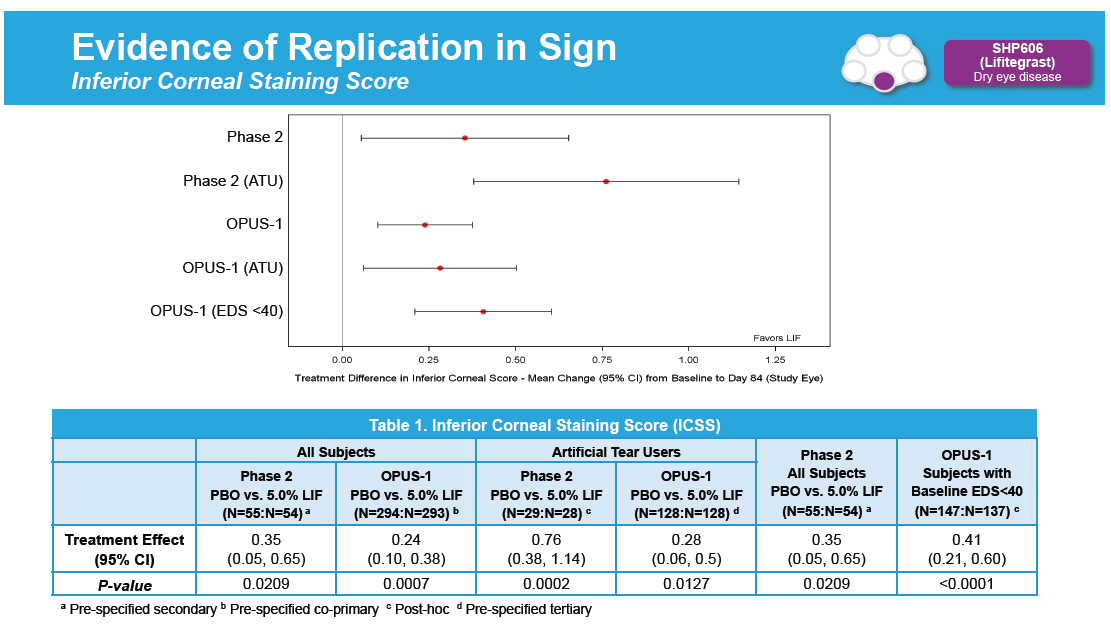 |
Treatment Effect on Inferior Corneal Staining Score Over the Course of Studies [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease Phase 2 ITT Population with LOCF Mean ([]SE) Change from Baseline OPUS-1Study 2 ITT Population with LOCF Mean ([]SE) Change from Baseline [GRAPHIC OMITTED] 204 |  |
Evidence of Replication in Symptom
Eye Dryness Score
[GRAPHIC OMITTED]
SHP606
(Lifitegrast)
Dry eye disease
OPUS-1 (ATU)
OPUS-1 (ATU, EDS >40)
OPUS-2
Table 2. Eye Dryness Score (EDS)
Artificial Tear Users
OPUS-1 OPUS-2
PBO vs. 5.0% LIF PBO vs. 5.0% LIF
(N=129:N=128) (a) (N=294:N=293) (b)
6.96 12.61
Treatment Effect (95% CI) (-0.79, 14.71) (8.51, 16.70)
P-value 0.0783 <0.0001
Artificial Tear Users, EDS [] 40
OPUS-1 OPUS-2
PBO vs. 5.0% LIF PBO vs. 5.0% LIF
(N=67:N=63) (c) (N=294:N=293) (b)
13.34 12.61
(2.35, 24.33) (8.51, 16.70)
0.0178 <0.0001
(a) Pre-specified tertiary (b) Pre-specified co-primary (c)
Post-hoc
| 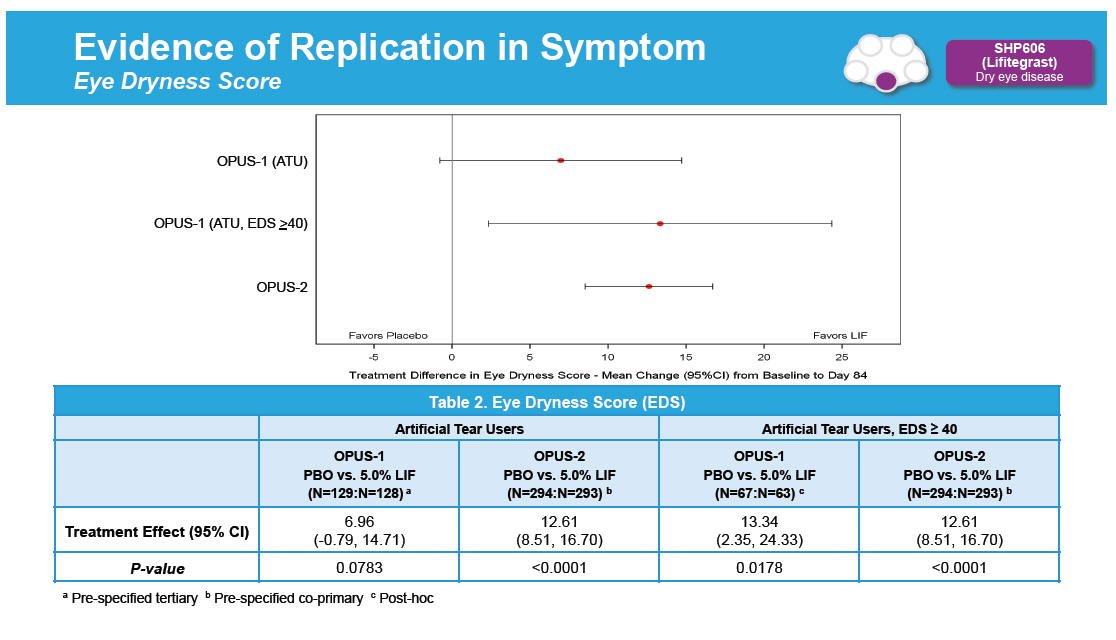 |
Treatment Effect on Eye Dryness Score Over the Course of the Study [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease OPUS-1 ITT Population Observed Data plus ITTOPUS-1 with LOCF at Day 84 Mean ([]SE) Change from BaselineOPUS-1 OPUS-2 ITT Population Observed Data plus ITTOPUS-2 with LOCF at Day 84 Mean ([]SE) Change from Baseline [GRAPHIC OMITTED] 206 | 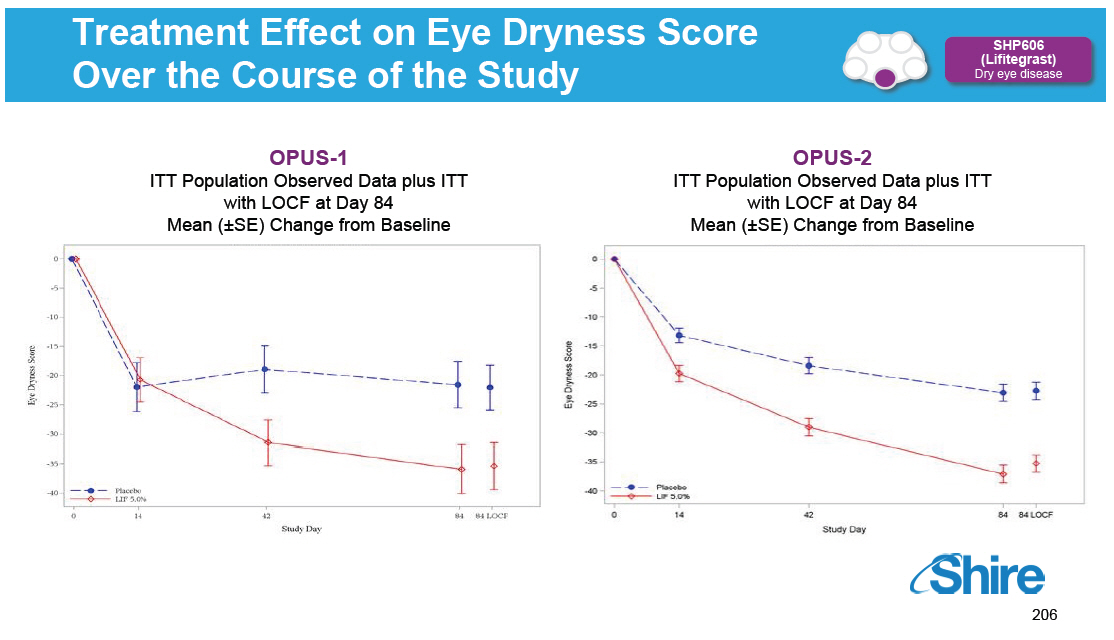 |
Regulatory Position Based on our Pre-NDA Meeting - May 15, 2014 [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease [] We are confident that the totality of the data from our existing clinical development program supports the submission of an NDA for Lifitegrast [] However, we are continuing to gather clinical data in support of US and potential international regulatory submissions [] OPUS-3 will not delay our plans to submit an NDA for Lifitegrast for signs and symptoms of dry eye disease in the first quarter of 2015 [GRAPHIC OMITTED] 207 |  |
Why Conduct OPUS-3? [GRAPHIC OMITTED] SHP606 (Lifitegrast) Dry eye disease [] OPUS-3 (a Phase 3 safety and efficacy study) is to bolster our potential US label and further support international markets and will be conducted concurrent to the US NDA review [] Identical to OPUS-2 Population: DED, history of artificial tear use, and Eye Dryness Score [] 40 [] Study designed to strengthen our existing efficacy data by evaluating the following symptom endpoints: [] Primary efficacy endpoint: Superiority against placebo on eye dryness score as shown by change from baseline to day 84 [] Key Secondary efficacy endpoints: Superiority against placebo on eye dryness score as shown by change from baseline to day 42 and day 14 [GRAPHIC OMITTED] 208 |  |
Late Stage Pipeline Update -- Agenda Dry Eye 2014 Lifitegrast for the treatment of Dry Eye Disease Late-Stage Regulatory Update [] Vyvanse for Binge Eating Disorder [] SHP465 for Attention Deficit Hyperactivity Disorder Joe Tauber, MD Howard Mayer, MD Randy Brenner [GRAPHIC OMITTED] 209 |  |
Vyvanse for Binge Eating Disorder (BED) Preclinical Phase 1 Phase 2 Phase 3 Registration 26 Research Programs SHP619 Duchenne's Muscular Dystrophy TH / GCH1 Gene Pod Parkinson's Subset SHP608 Dystrophic E.Bullosa SHP614 IgA Nephropathy Armagen Hunter CNS SHP630 adRP SHP624 SHP611 MLD (Ph 1/2) SHP616 (Cinryze SC) HAE Prophylaxis SHP622 Friedreich's Ataxia SHP627 (FT011) Focal Segmental Glomerulosclerosis SHP616 (Cinryze) Paroxysmal Nocturnal Hemoglobinuria SHP616 (Cinryze) Acute Neuromyelitis Optica SHP626 (LUM002) Non-Alcoholic Steatohepatitis SHP602 SHP616 (Cinryze) Iron overload Acute Antibody (clinical hold) Mediated Rejection SHP610 SHP625 (LUM001) Primary Biliary Sanfilippo A Cirrhosis SHP609 SHP625 (LUM001) Progressive Familial Hunter CNS Intrahepatic Cholestasis SHP607 SHP625 (LUM001) Prevention of ROP Alagille Syndrome SHP620 (Maribavir) SHP625 (LUM001) CMV in transplant Primary Sclerosing patients Cholangitis LDX (Japan) ADHD Firazyr ACE inhibitor-induced AE Firazyr (Japan) HAE SHP616 (Cinryze) (Japan) HAE Prophylaxis SHP555 (US) Chronic Constipation INTUNIV (Japan) ADHD SHP606 (Lifitegrast) Dry eye disease SHP465 ADHD XAGRID(r) (Japan) Thrombocythaemia (Approved 3Q 2014) VPRIV (Japan) Gaucher (Approved 3Q 2014) INTUNIV(r) (EU) ADHD Vyvanse BED Heme B Gene Edit SHP628 (FT-061) Renal Impairment Complement Biology GI / Metabolic Renal / Transplant CNS Ophthalmics Rare Diseases Leadership 210 [GRAPHIC OMITTED] [GRAPHIC OMITTED] |  |
Vyvanse for BED [GRAPHIC OMITTED] LDX BED Development Program Status [] SPD489-208: Fixed-dose study (completed, included in sNDA) [] SPD489-343: Dose optimization study #1 (completed, included in sNDA) [] SPD489-344: Dose optimization study #2 (completed, included in sNDA) [] SPD489-345: Open-label long-term safety study (Interim data cuts included in sNDA with final CSR planned March 2015) [] SPD489-346: Long term maintenance of efficacy study (ongoing, final CSR planned for September 2015, not included in sNDA) [GRAPHIC OMITTED] 211 |  |
SPD489-343/344: Primary Efficacy Endpoint*: LS Mean ([]SEM) Change from Baseline in the Number of Binge Days/Week (Full Analysis Set) 0 P la c e b o S P D 4 8 9 k e - 1 e e W 3 g 4 n r a e 3 h p - 2 - 9 C s 8 y n a 4 a e D D - 3 P M e g S S in L B - 4 in - 5 0 P la c e b o S P D 4 8 9 k e - 1 e e W 4 g 4 n r 3 a e - h p - 2 9 C s 8 y n 4 a a e D D - 3 M e P g S S in L B - 4 in - 5 V i s i t 1 V i s i t 2 V i s i t 3 V i s i t 4 V i s i t 5 V i s i t 6 V i s i t 7 V i s i t 8 w k 1 w k 2 w k 3 w k 4 w k 5 / 6 w k 7 / 8 w k 9 / 1 0 w k 1 1 / 1 2 *p-value <0.001 at Visit 8 (Weeks 11 and 12) for both studies. Data Source: Table 3.1.1.1 and 3.1.1.2 [GRAPHIC OMITTED] 212 |  |
BED Regulatory Update [GRAPHIC OMITTED] LDX BED Site inspections complete Timeline August 1 Submission Sept 30 Accepted for filing PRIORITY REVIEW No filing issues identified Mid Cycle Review No AC currently planned Ongoing QandA Nov 21 Safety Update submission Feb 1 PDUFA [GRAPHIC OMITTED] 213 |  |
SHP465: ADHD [GRAPHIC OMITTED] [GRAPHIC OMITTED] | 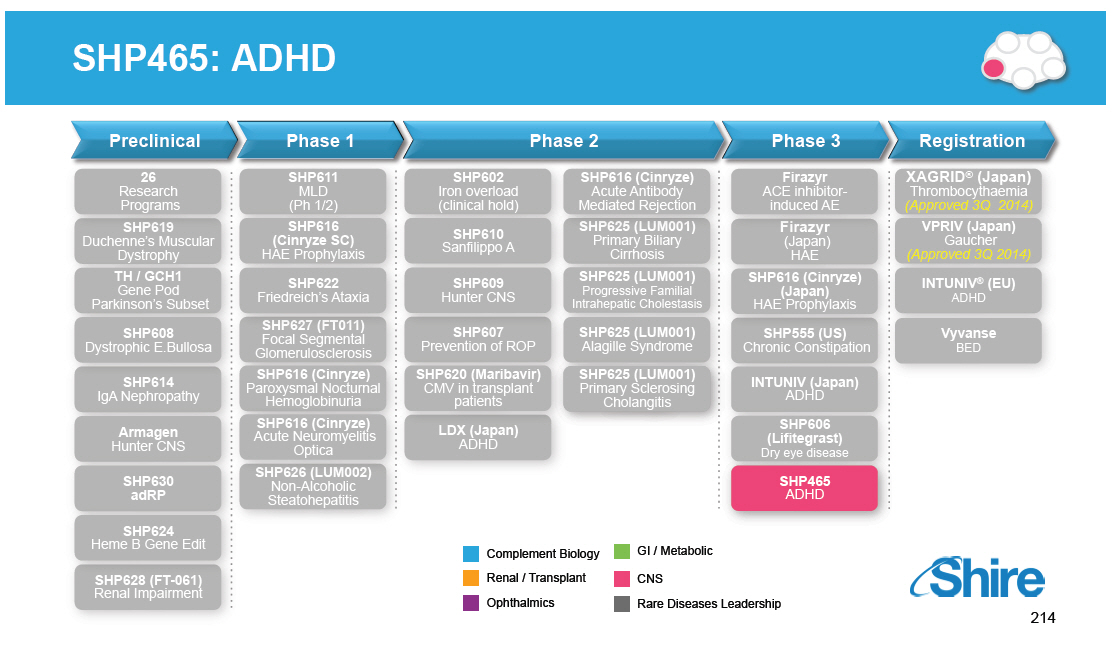 |
SHP465: ADHD [GRAPHIC OMITTED] SHP465 ADHD [] A three bead formulation of mixed amphetamine salts provides ADHD symptom control at 16 hours post-dose (Adult ADHD). [GRAPHIC OMITTED] [GRAPHIC OMITTED] 215 |  |
SHP465: ADHD Development Status [GRAPHIC OMITTED] SHP465 ADHD Development Program Status [] SPD465-201: Adult workplace laboratory study (completed) [] SPD465-202: Adolescent analog classroom study (completed) [] SPD465-203: Adult workplace laboratory study (completed) [] SPD465-301: Adult dose-optimization study (completed) [] SPD465-303: Adult fixed-dose study (completed) [] SPD465-304: Adult open-label long-term safety study (completed) [] SHP465-305: Pediatric and adolescent dose-optimization study (planning) Part of initial submission in 2006 [GRAPHIC OMITTED] 216 |  |
SHP465 Regulatory Update [GRAPHIC OMITTED] SHP465 ADHD Initial NDA Review Timeline Slide 2006 Submission 2007 Approvable letter Ongoing dialogue regarding Peds requirements Feb 2014 Reinitiation with FDA discussions started April Initial FDA comments received. began discussions on peds requirements October General alignment on plan and peds plan submitted Nov Full protocol provided to FDA [GRAPHIC OMITTED] 217 | 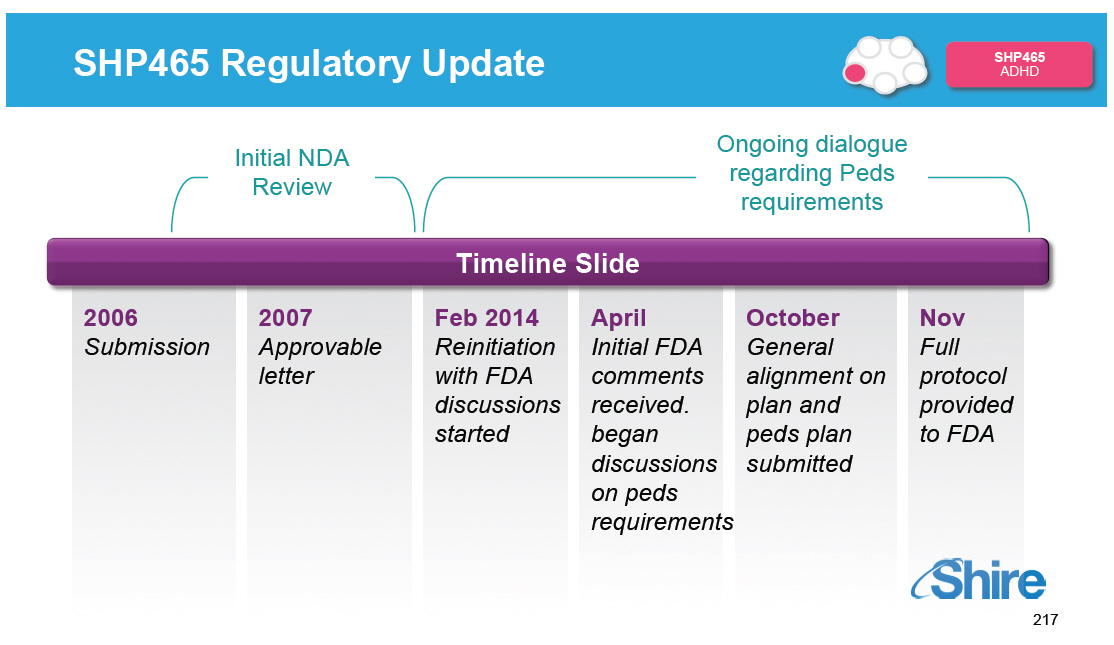 |
Upcoming Anticipated Late-Stage Pipeline Milestones [GRAPHIC OMITTED] Registration and Phase 4 Phase 3 Phase 2 Phase 1/2 SHP611 MLD Phase 1/2 head line data SHP625 (LUM001) ALGS Phase 2 head line data SHP606 Lifitegrast NDA Filing SHP625 (LUM001) PSC Phase 2 head line data SHP607 ROP Phase 2 head line data SHP606 Lifitegrast Launch(2) SHP609 Phase 2/3 head line data 20152015 VYVANSE BED Launch(2) SHP620 Maribavir Phase 2 head line data SHP625 (LUM001) PBC Phase 2 head line data SHP625 (LUM001) ALGS Phase 2 head line data 2016 SHP625 (LUM001) (1) PFIC Phase 2 head line data SHP610 Phase 2B head line data Notes (1)[] Interim 625 PFIC INDIGO data expected Q2 2015. (2)[] Subject to regulatory approval. [GRAPHIC OMITTED] 218 |  |
Program Wrap-Up Phil Vickers, Ph.D., Global Head of RandD Our purpose We enable people with life-altering conditions to lead better lives [GRAPHIC OMITTED] |  |
RandD Focused on Five Therapeutic Areas Rare Diseases Leadership Complement Biology [] Firazyr / Cinryze [] Firazyr ACE-I [] Cinryze SC [] Cinryze new uses Renal / Fibrotic [] Maribavir [] Cinryze AMR [] IGAN [] Fibrotech (FSGS) [] DEB [] CF GI / Metabolic [] SHP625 (ALGS, PSC, PFIC, PBC) [] HemeA / B [] UCD [] SHP626 (NASH) [] SHP555 Ophthalmics [] ROP [] adRP [] Lifitegrast CNS [] BED [] SHP465 [] Hunter CNS [] Sanfilippo A [] MLD [] Armagen [] HTT Also evaluating opportunities in 'Rare Hematological' malignancies [GRAPHIC OMITTED] 220 | 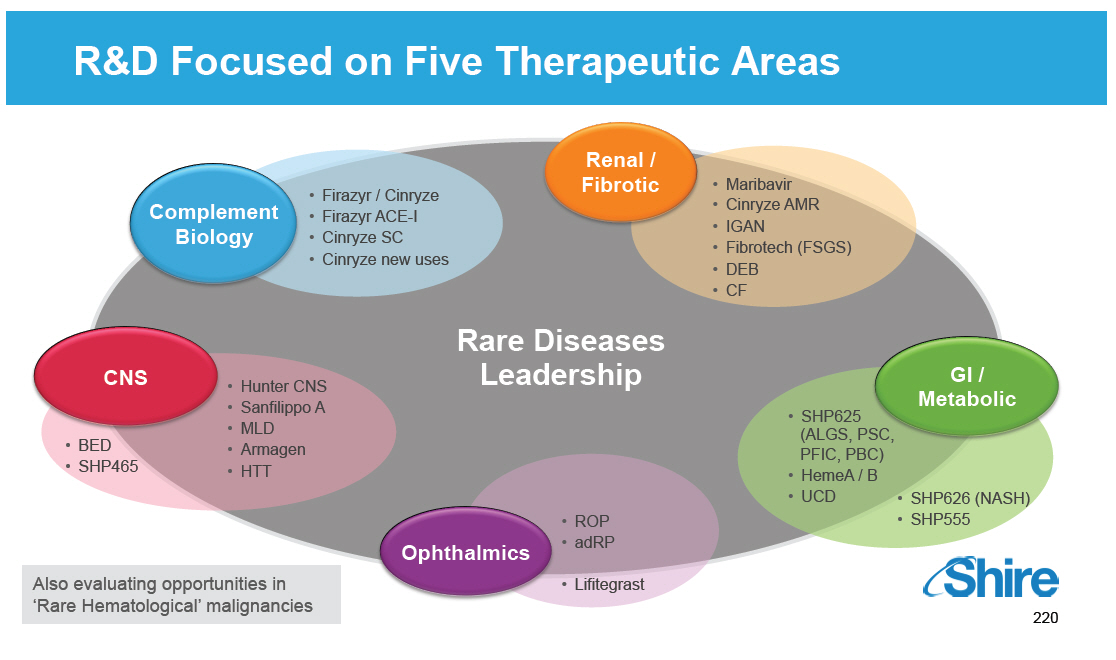 |
Pipeline Increasingly Focused on Rare Diseases
26
Research
Programs
SHP619
Duchenne's Muscular
Dystrophy
TH / GCH1
Gene Pod
Parkinson's Subset
SHP608
Dystrophic E.Bullosa
SHP614
IgA Nephropathy
Armagen
Hunter CNS
SHP630 adRP
SHP624
Heme B Gene Edit
SHP628 (FT-061)
Renal Impairment
SHP611
MLD (Ph 1/2)
SHP616
(Cinryze SC)
HAE Prophylaxis
SHP622
Friedreich's Ataxia
SHP627 (FT011)
Focal Segmental
Glomerulosclerosis
SHP616 (Cinryze )
Paroxysmal Nocturnal
Hemoglobinuria
SHP616 (Cinryze)
Acute Neuromyelitis
Optica
SHP626 (LUM002)
Non-Alcoholic
Steatohepatitis
SHP602
SHP616 (Cinryze)
Iron overload
Acute Antibody
(clinical hold)
Mediated Rejection
SHP610
SHP625 (LUM001)
Primary Biliary
Sanfilippo A
Cirrhosis
SHP609
SHP625 (LUM001)
Progressive Familial
Hunter CNS
Intrahepatic Cholestasis
SHP607
SHP625 (LUM001)
Prevention of ROP
Alagille Syndrome
SHP620 (Maribavir)
SHP625 (LUM001)
CMV in transplant
Primary Sclerosing
patients
Cholangitis
LDX (Japan)
ADHD
Rare Diseases Programs
Firazyr
ACE inhibitor-induced AE
Firazyr
(Japan) HAE
SHP616 (Cinryze)
(Japan) HAE Prophylaxis
SHP555 (US)
Chronic Constipation
INTUNIV (Japan)
ADHD
SHP606
(Lifitegrast)
Dry eye disease
SHP465
ADHD
XAGRID(r) (Japan)
Thrombocythaemia
(Approved 3Q 2014)
VPRIV (Japan)
Gaucher
(Approved 3Q 2014)
INTUNIV(r) (EU)
ADHD
Vyvanse
BED
221
[GRAPHIC OMITTED]
|  |
Programs Reviewed Today
GROWTH
INNOVATION
26
Research
Programs
SHP619
Duchenne's Muscular
Dystrophy
TH / GCH1
Gene Pod
Parkinson's Subset
SHP608
Dystrophic E.Bullosa
SHP614
IgA Nephropathy
Armagen
Hunter CNS
SHP630 adRP
SHP611
MLD (Ph 1/2)
SHP616
(Cinryze SC)
HAE Prophylaxis
SHP622
Friedreich's Ataxia
SHP627 (FT011)
Focal Segmental
Glomerulosclerosis
SHP616 (Cinryze)
Paroxysmal Nocturnal
Hemoglobinuria
SHP616 (Cinryze)
Acute Neuromyelitis
Optica
SHP626 (LUM002)
Non-Alcoholic
Steatohepatitis
SHP602
SHP616 (Cinryze)
Iron overload
Acute Antibody
(clinical hold)
Mediated Rejection
SHP610
SHP625 (LUM001)
Primary Biliary
Sanfilippo A
Cirrhosis
SHP609
SHP625 (LUM001)
Progressive Familial
Hunter CNS
Intrahepatic Cholestasis
SHP607
SHP625 (LUM001)
Prevention of ROP
Alagille Syndrome
SHP620 (Maribavir)
SHP625 (LUM001)
CMV in transplant
Primary Sclerosing
patients
Cholangitis
LDX (Japan)
ADHD
Firazyr
ACE inhibitor-induced AE
Firazyr
(Japan) HAE
SHP616 (Cinryze)
(Japan) HAE Prophylaxis
SHP555 (US)
Chronic Constipation
INTUNIV (Japan)
ADHD
SHP606
(Lifitegrast)
Dry eye disease
SHP465
ADHD
XAGRID(r) (Japan)
Thrombocythaemia
(Approved 3Q 2014)
VPRIV (Japan)
Gaucher
(Approved 3Q 2014)
INTUNIV(r) (EU)
ADHD
Vyvanse
BED
SHP624
Heme B Gene Edit
SHP628 (FT-061)
Renal Impairment
Complement Biology
GI / Metabolic
Renal / Transplant
CNS
Ophthalmics
Rare Diseases Leadership
Programs not specifically discussed today
222
[GRAPHIC OMITTED]
| 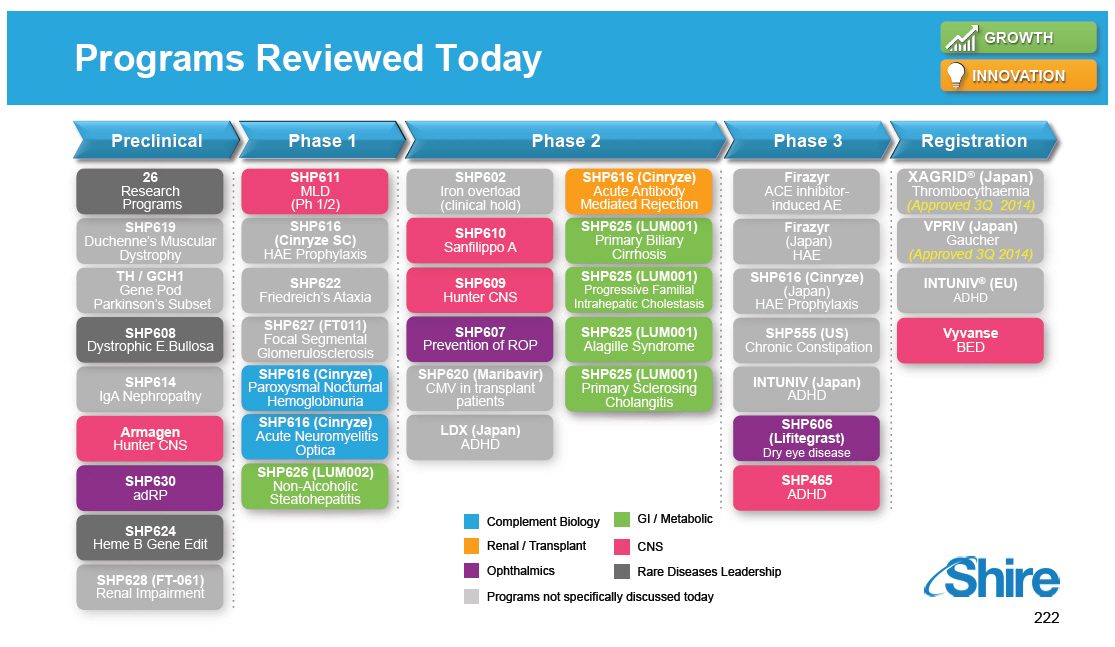 |
Pipeline Balances Innovation and Risk 223 | 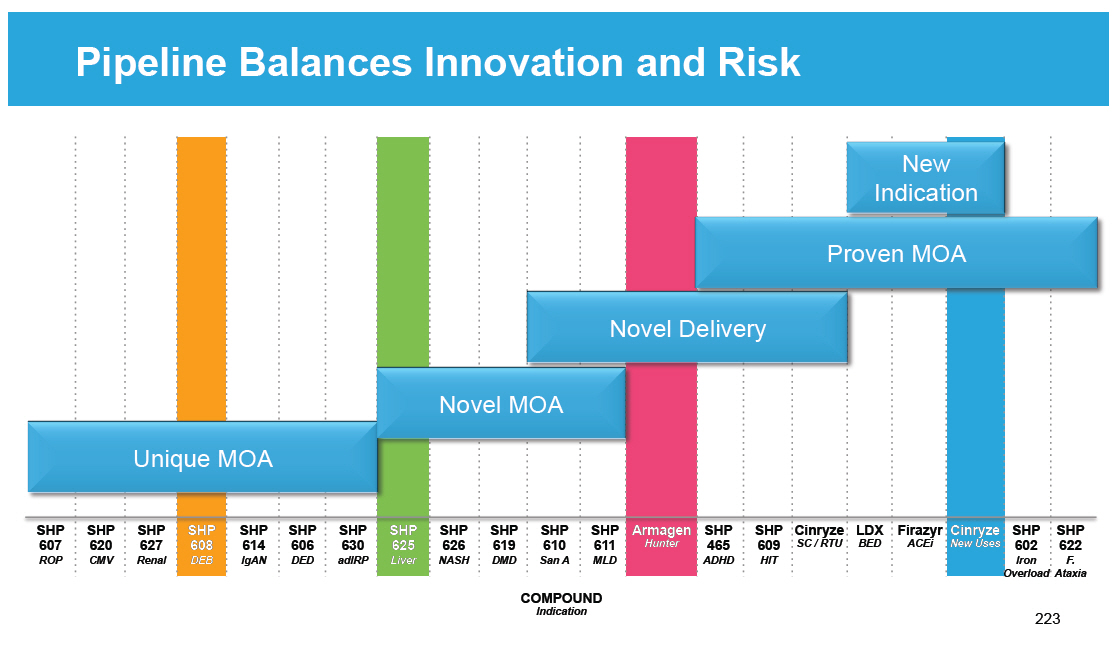 |
Upcoming Anticipated Pipeline Milestones Registration and Phase 4 Phase 3 Phase 2 Phase 1/2 SHP611 MLD Phase 1/2 head line data SHP625 (LUM001) ALGS Phase 2 head line data SHP606 Lifitegrast NDA Filing SHP625 (LUM001) PSC Phase 2 head line data SHP607 ROP Phase 2 head line data SHP606 Lifitegrast Launch(2) SHP609 Phase 2/3 head line data 20152015 VYVANSE BED Launch(2) SHP620 Maribavir Phase 2 head line data SHP625 (LUM001) PBC Phase 2 head line data SHP625 (LUM001) ALGS Phase 2 head line data 2016 SHP625 (LUM001) (1) PFIC Phase 2 head line data SHP610 Phase 2B head line data Notes (1)[] Interim 625 PFIC INDIGO data expected Q2 2015. (2)[] Subject to regulatory approval. [GRAPHIC OMITTED] 224 |  |
Pipeline Has Never Been Stronger 22 programs in the clinic, the most in the history of Shire Well-positioned to deliver on '10 x 20' expectations Many significant clinical milestones in the next 18 months On track to file at least 2 INDs from internal programs every year Establishing talent and capabilities appropriate to drive future growth Continued excellence in acquiring external assets with a strong strategic fit Establishing a leadership position in the treatment of Rare Diseases [GRAPHIC OMITTED] 225 |  |
Question and Answer Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |
Executing on our Corporate Strategy through RandD [] We plan to increase product sales to $10 Billion by 2020 -- our 10 x 20 plan [] $3 Billion expected to come from our pipeline, excluding recent and future MandA [] Highly focused RandD organization [] Prioritizing Rare Diseases (18 of 22 independent clinical programs) [] Attracting, developing and retaining best talent [] Ensuring organizational simplicity through two major geographic hubs [] Our RandD strategy is driving shareholder value [] Addressing indications with high unmet medical need [] Delivering a high-value late-stage pipeline to market [] Optimizing asset value across the portfolio [] Expanding expertise and access to innovation through collaborations [GRAPHIC OMITTED] |  |
[] Shire RandD Day [] Our purpose We enable people with life-altering conditions to lead better [GRAPHIC OMITTED] |  |