 Salix Pharmaceuticals, Ltd. The Name Gastroenterologists Trust Exhibit 99.2 |
 2 Forward-Looking Statement Statements presented in this overview that are not historical facts are forward-looking statements that involve risks and uncertainties that could cause actual results to differ materially from projected results. Factors that could cause actual results to differ materially include anticipated write-offs and losses and our expectation to return to profitability, intellectual property risks specifically patent protection, competition from generic products and otherwise, the risks and uncertainties of clinical trials and regulatory review, market acceptance of approved products, and the need to acquire new products. These and other relevant risks are detailed in the Company’s Securities and Exchange Commission filings. |
 3 Generic Balsalazide December 28, 2007 Office of Generic Drugs Approved 3 Generic Balsalazide Products Salix Announced Watson Authorized Generic |
 4 Current Situation • Dramatic price erosion for COLAZAL – $125 million COLAZAL market now a $30-42 million balsalazide market • Impact on price of future IBD products – Balsalazide tablet and granulated mesalamine • Substantial non-cash charge in 4Q07 • Not profitable in 2008 – Financial guidance to be provided March 3, 2008 |
 5 Future Outlook • Based on current projections – – No need to raise money to execute business plan – Sufficient cash reserve to return to profitability • Expect to be cash flow positive in 2009 • OSMOPREP and MOVIPREP well-positioned in a growing market – TRx market growth of 18% in 2007* – Osmoprep/Moviprep TRx growth of 353% in 2007* – Expect products to be a $100 million franchise in approx. 3 years • Stable revenue from XIFAXAN and “Other Products” * Wolters Kluwer Health PHAST |
 6 Future Outlook • 3 NDA approvals anticipated in 2008 – Balsalazide tab, granulated mesalamine, and metoclopramide-ZYDIS • Focus 2008 R&D spend on largest potential indications • Rifaximin Irritable Bowel Syndrome (IBS) program – Mid-2008: Initiate Phase III trials – 2008: Publication/presentation of Phase IIb data – Mid-2010: Anticipate NDA submission • Rifaximin Hepatic Encephalopathy (HE) program – Orphan Drug Designation – 4Q08/1Q09 NDA submission – Potential 6 month review • Numerous products with patent coverage |
 7 Mission Statement Salix Pharmaceuticals is committed to being the leading U.S. specialty pharmaceutical company licensing, developing and marketing innovative products to health care professionals to prevent or treat gastrointestinal disorders. |
 8 • In-license selective late-stage/marketed product candidates – U.S. gastroenterology focus – Reduce risk and capital commitment • Complete development and secure FDA approval • Sell products through specialty sales force – Establish franchise with U.S. gastroenterologists – Sell products that serve unmet needs Strategy |
 9 An Overall Compelling Market Opportunity • An influential and demanding target audience (13,400 specialty physicians) – 30.5 million patient visits annually – 40 million prescriptions annually – $6 billion Rx sales annually Source: Verispan PDDA and Wolters Kluwer Health (1/08) |
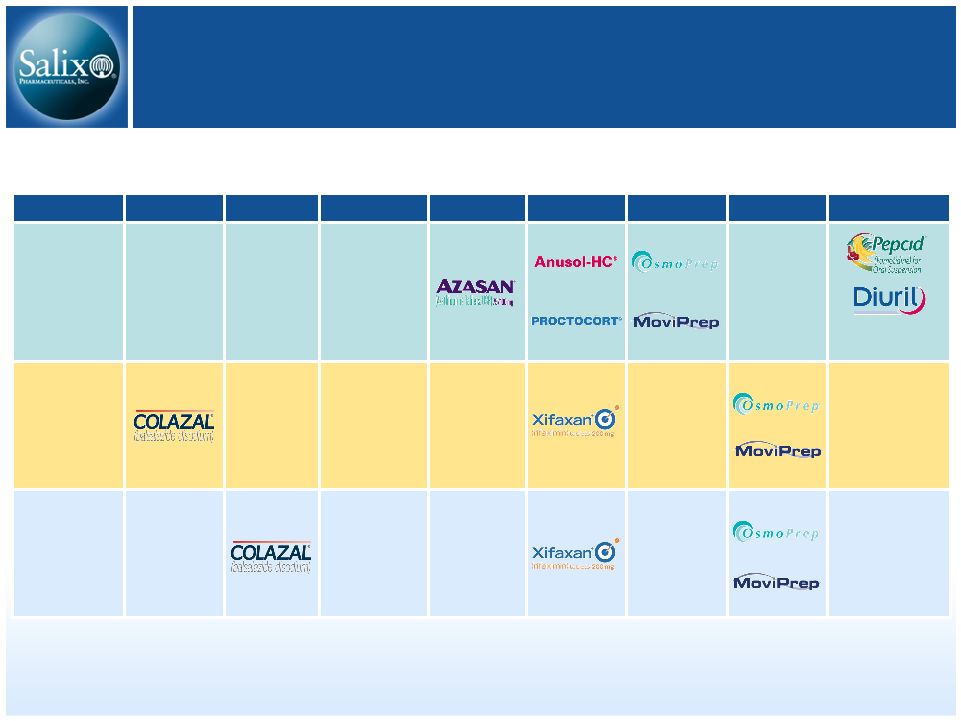 10 History of Success Product Launches NDA Approvals Metoclopramide- ZYDIS ® Vapreotide acetate Granulated Mesalamine Acquisitions 2007 2006 2005 2004 2003 2002 2001 2000 |
 11 Anticipated Approvals • INFLAMMATORY BOWEL DISEASE – Balsalazide tablet (1100 mg) 2Q08 – Granulated mesalamine (1.5 gm) 4Q08 • METOCLOPRAMIDE-ZYDIS 4Q08 • RIFAXIMIN – Hepatic Encephalopathy (550 mg) 3Q09 – Irritable Bowel Syndrome (550 mg) Mid-2011 |
 12 |
 13 Xifaxan • Unique, Gastrointestinal-Specific Antibiotic – Broad spectrum – Reduced risks – No stable resistance • 2004 FDA approval for treatment of travelers’ diarrhea • Patent coverage: May 24, 2024* – Orange Book listed *Patent expiration dates listed herein are for US patents and assume there are no patent term adjustments, extensions or other events that could affect the term or scope of a patent. |
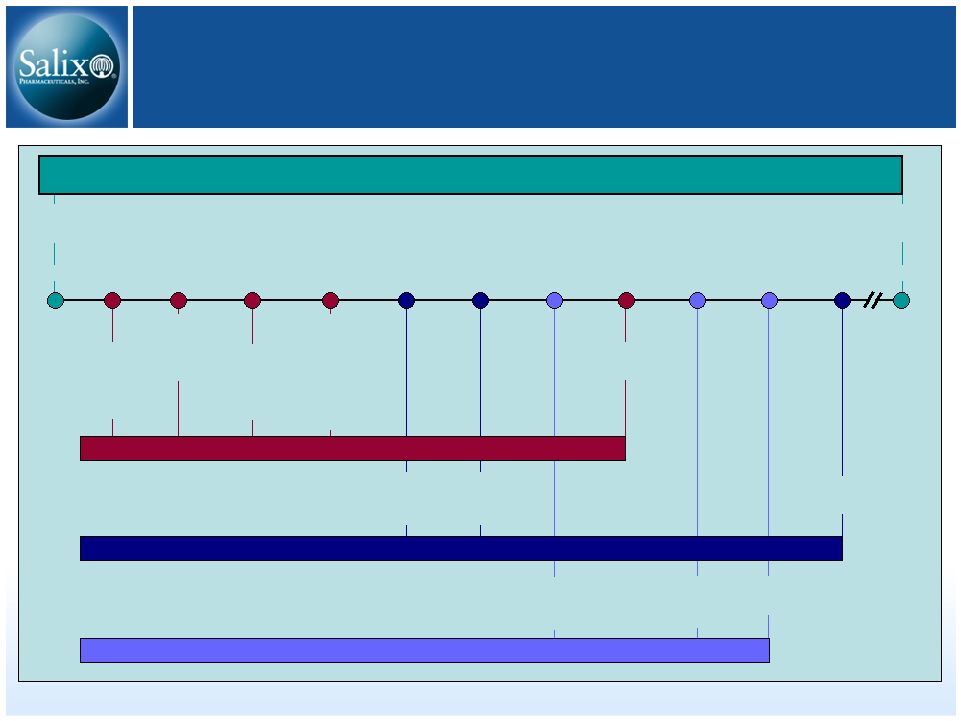 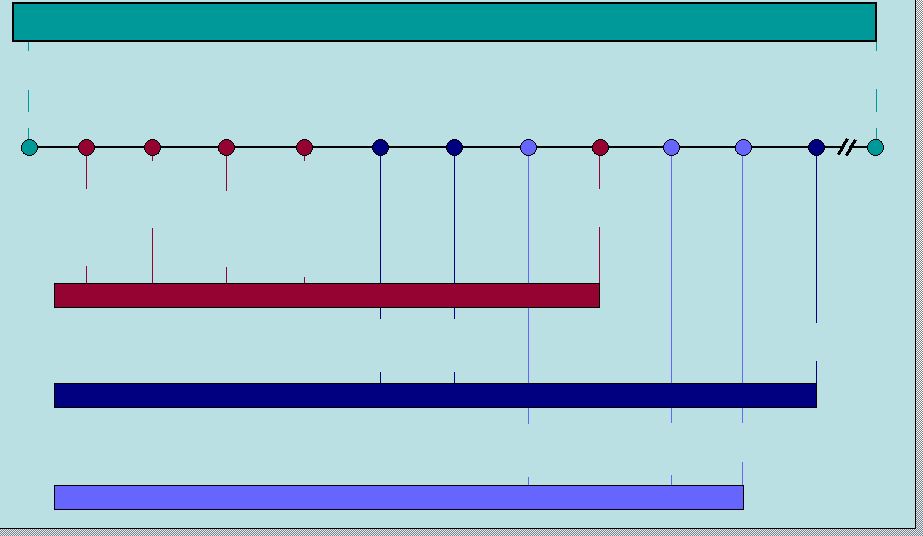 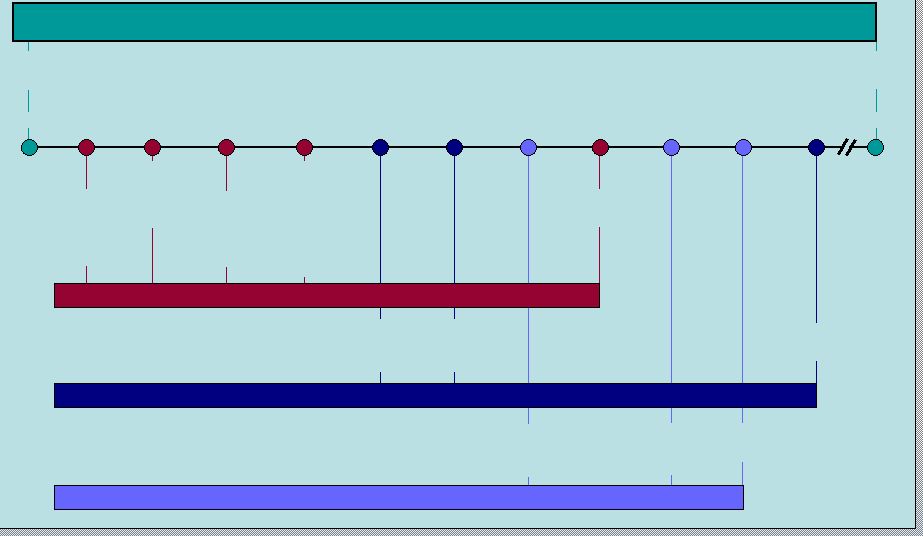 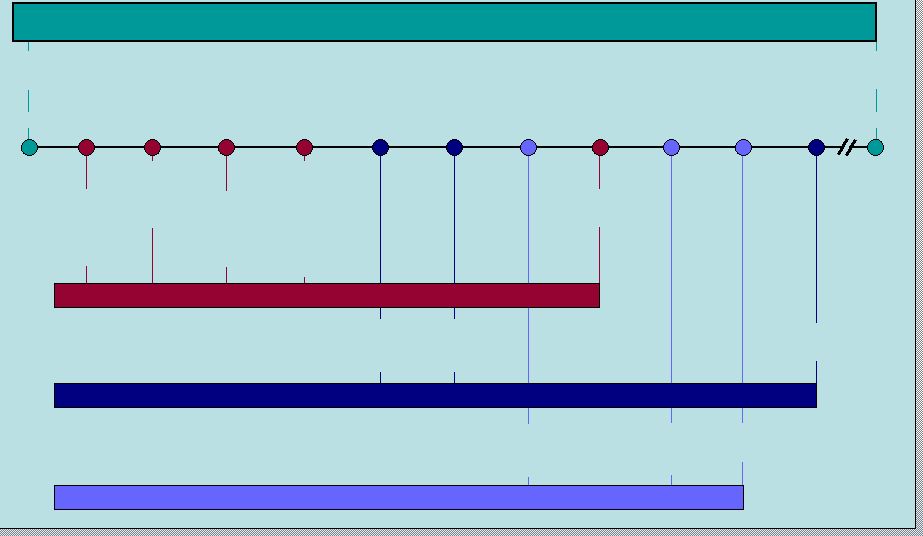 14 RIFAXIMIN Intellectual Property Q2 2011 Q1 2009 Q3 2009 Q2 2010 2014 2016 3-28-11 HE anticipated filing 7 year orphan exclusivity HE anticipated approval 9-28-08 7-25-08 8-14-08 5-26-08 First possible ANDA filing (includes paragraph 4 certification) IBS anticipated approval IBS anticipated filing 3 year exclusivity Patent expiration Patent issued Salix has 45 days to respond and file lawsuit (entitled up to 30 month stay; average is 28.5 months according to Paragraph 4 database) FDA has 60 days to accept the ANDA ANDA filer (20 days to inform Salix) 30 month stay expiration RIFAXIMIN 550mg (HE) 5-26-06 5-22-24 RIFAXIMIN 550mg (IBS) XIFAXAN 200mg (TD) Polymorphous forms of RIFAXIMAN, processes for their production and use thereof in medicinal preparations |
 15 Bowel Cleansing |
 16 Improved Preps for Colonoscopy • OsmoPrep Proprietary tablet formulation Launched June 2006 Patent coverage: May 2013* • MoviPrep Proprietary, state-of-the-art 2 liter liquid PEG with ascorbic acid Launched October 2006 Patent coverage: September 2024* • Bowel Prep TRx Market Growth in 2007 Total market 18% OsmoPrep/MoviPrep 353% *Patent expiration dates listed herein are for US patents and assume there are no patent term adjustments, extensions or other events that could affect the term or scope of a patent. |
 17 Next Generation IBD Products • Balsalazide Tablet (1100mg) PDUFA* action date – May 16, 2008 Patent coverage: March 2018** Expected launch July 2008 • Granulated Mesalamine (375mg capsule) PDUFA action date – 4Q08 (estimate) Patent coverage: April 2018** Expected launch Jan/Feb 2009 *Prescription Drug User Fee Act **Patent expiration dates listed herein are for US patents and assume there are no patent term adjustments, extensions or other events that could affect the term or scope of a patent. |
 18 Products in Development • Metoclopramide-ZYDIS ® Gastroparesis/Gastroesophageal reflux Expected approval 4Q08 Expected launch Jan/Feb 2009 Patent coverage: July 2017* • Vapreotide acetate Treatment of acute esophageal variceal bleeding » Serious complication of late-stage liver cirrhosis – Ongoing confirmatory Phase III trial Orphan Drug Designation *Patent expiration dates listed herein are for US patents and assume there are no patent term adjustments, extensions or other events that could affect the term or scope of a patent. |
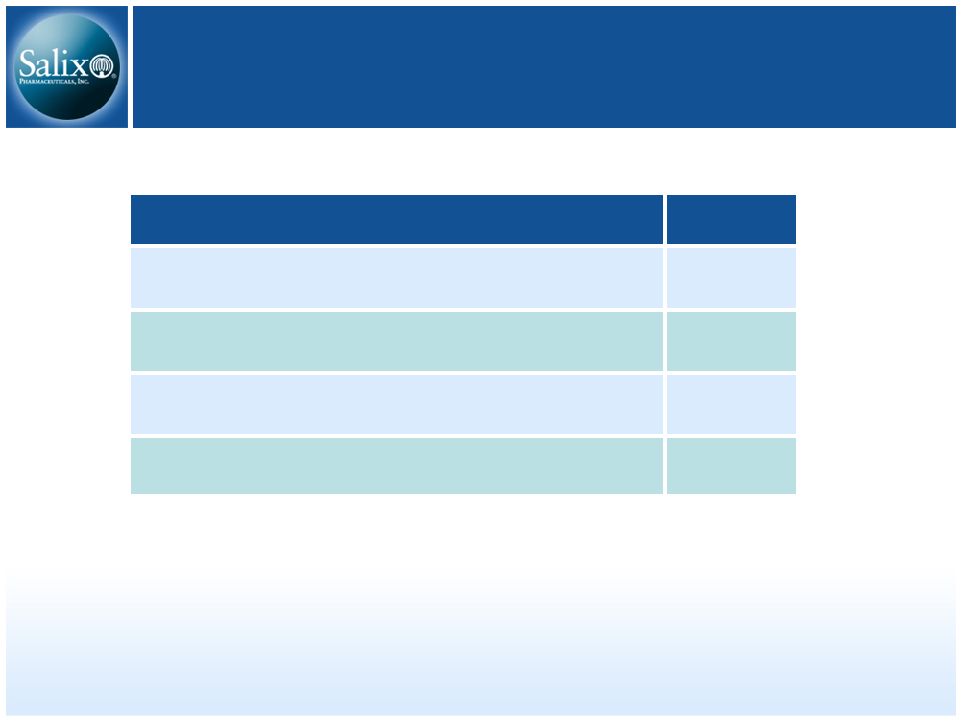 19 Capitalization – 9/30/07 (in millions) 48.6 Fully diluted common shares (Treasury method) 47.2 Common shares outstanding $15.0 Long term debt $70.2 Cash, cash equivalents & investments Actual |
 20 Current Run Rates for Base Business ~ $28 Other Products ~$5** Colazal ~ $60 MoviPrep OsmoPrep Visicol ~ $64 Xifaxan (rifaximin) Revenue Run Rate (in millions)* *Run rate based on current Rx data **Estimate for 2008 |
    21 2008 Anticipated Milestones Q2 2008 Q1 2008 • Complete Rifaximin HE Phase III enrollment • Balsalazide tablet PDUFA action date • Digestive Disease Week – Rifaximin IBS Phase IIb data and balsalazide tablet data (abstracts submitted) • Initiate Rifaximin IBS Phase III trials (Mid-2008) Q3 2008 • Launch balsalazide tablet • American College Gastroenterology – Rifaximin IBS Phase IIb data and granulated mesalamine data (abstracts to be submitted) Q4 2008 • Granulated mesalamine PDUFA action date (estimated) • Metoclopramide-ZYDIS PDUFA action date (estimated) • File Rifaximin HE NDA (4Q08/1Q09) |
 22 2009 Anticipated Milestones • File Rifaximin HE NDA (4Q08/1Q09) • Launch granulated mesalamine • Launch metoclopramide-ZYDIS • Compete Rifaximin IBS Phase III enrollment • Expect to be cash flow positive |
 23 Salix Pharmaceuticals, Ltd. Providing products to gastroenterologists and their patients |