
Issuer Free Writing Prospectus Filed Pursuant to Rule 433 Registration No. 333-189395 July 22, 2013
Precision Immunotherapy TM
Opexa Therapeutics, Inc.
NASDAQ: OPXA
July 2013 The Woodlands, TX

Forward-Looking Statements
This investor presentation contains forward-looking statements which are made pursuant to the safe harbor provisions of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Statements contained in this presentation, other than statements of historical fact, constitute “forward-looking statements.” The words “expects,” “believes,” “anticipates,” “estimates,” “may,” “could,” “intends,” and similar expressions are intended to identify forward-looking statements. Forward-looking statements do not constitute guarantees of future performance. Investors are cautioned that statements which are not strictly historical statements, including, without limitation, statements regarding the development of our product candidate, Tcelna (imilecleucel-T), constitute forward-looking statements.
Such forward-looking statements are subject to a number of risks and uncertainties that could cause actual results to differ materially from those anticipated, which include, but are not limited to, risks associated with: market conditions; our capital position; the rights and preferences provided to the Series A convertible preferred stock and investors in the July 2012 convertible secured notes; our ability to compete with larger, better financed pharmaceutical and biotechnology companies; new approaches to the treatment of our targeted diseases; our expectation of incurring continued losses; our ability to raise additional capital to continue our development programs or access the financing potentially available under the purchase agreements with Lincoln Park Capital Fund, LLC; our ability to maintain compliance with NASDAQ listing standards; the success of our clinical trials (including the Phase IIb trial for Tcelna in Secondary Progressive Multiple Sclerosis (“SPMS”) which, depending upon results, may determine whether Ares Trading SA (“Merck”) elects to exercise its option to obtain license rights to Tcelna in multiple sclerosis (the “Option”)); whether Merck exercises the Option and, if so, whether we receive any development or commercialization milestone payments or royalties from Merck pursuant to the Option; our dependence (if Merck exercises the Option) on the resources and abilities of Merck for the further development of Tcelna; the efficacy of Tcelna for any particular indication; our ability to develop and commercialize products; our ability to obtain required regulatory approvals; our compliance with all FDA regulations; our ability to obtain, maintain and protect intellectual property rights; the risk of litigation regarding our intellectual property rights or the rights of third parties; our limited manufacturing capabilities; our dependence on third-party manufacturers; our volatile stock price; and other risks detailed in our filings with the SEC.
These forward-looking statements speak only as of the date made. We assume no obligation or undertaking to update any forward-looking statements to reflect any changes in expectations with regard thereto or any change in events, conditions or circumstances on which any such statement is based. You should, however, review additional disclosures we make in our registration statement on Form S-1 initially filed on June 17, 2013 and as subsequently amended as well as in our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, and Current Reports on Form 8-K filed with the SEC.
2 |
|

Free Writing Prospectus Statement
This presentation highlights basic information about us and the offering. Being a summary document, this slide deck does not contain all the information that you should consider before investing.
We have filed a registration statement (including a preliminary prospectus) with the SEC for the offering to which this presentation relates. The registration statement has not yet become effective. Before you invest, you should read the preliminary prospectus in the registration statement (including the risk factors described therein) and other documents that we have filed with the SEC for more complete information about us and the offering. You may get these documents for free by visiting the “Search EDGAR” section on the SEC web site at http://www.sec.gov. The preliminary prospectus, dated July 22, 2013, is available on the SEC website. Alternatively, we or Aegis Capital Corp., the underwriter participating in this offering, will arrange to send you a preliminary prospectus if you contact Aegis Capital Corp., Prospectus Department, 810 Seventh Avenue, 18th Floor, New York, NY 10019; Telephone: (212) 813-1010; Email: prospectus@aegiscap.com
3 |
|
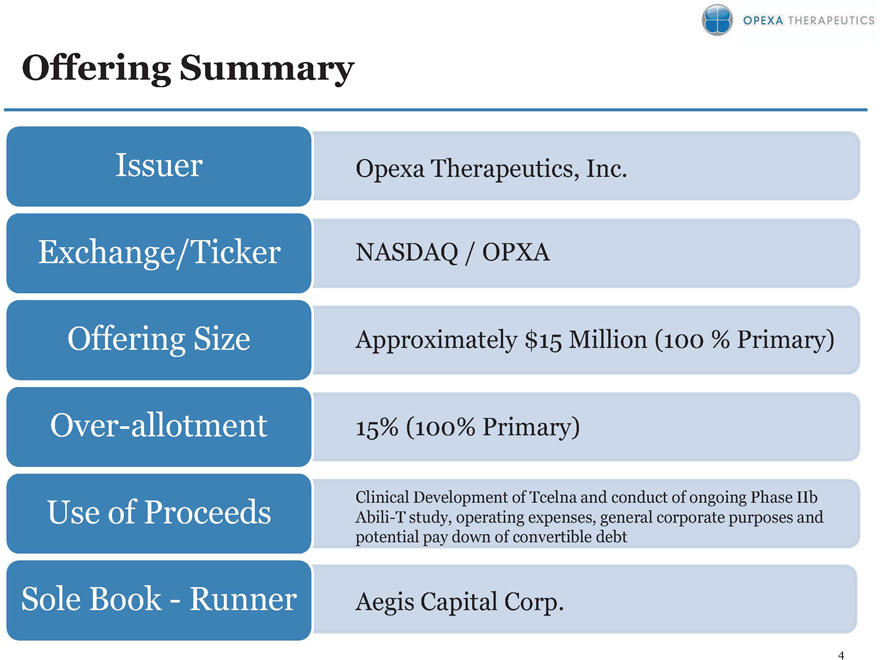
Offering Summary
Issuer
Exchange/Ticker
Offering Size
Over-allotment
Use of Proceeds
Sole Book—Runner
Opexa Therapeutics, Inc.
NASDAQ / OPXA
Approximately $15 Million (100% Primary)
Clinical Development of Tcelna and conduct of ongoing Phase IIb Abili-T study, operating expenses, general corporate purpose and potential pay down of convertible debt
Aegis Capital Corp.
4 |
|

Investment Thesis
T-cell platform company with Fast Track designation in SPMS
Esteemed Scientific Advisory Board
Precision Immunotherapy potentially optimizes benefit-risk profile
Targeting an unmet medical need in a potentially substantial market
Option Agreement with Merck Serono, a strong commercial partner
Replacement value of company is multiples of present market cap
Attractive potential risk-reward profile for long term/value investors
Goal-oriented management team focused on value creation
5 |
|

Experienced Management Team and Board of Directors
Neil Warma, President & CEO, Director
19+ years international healthcare experience with large Pharma and emerging biotechnology companies
Former Senior Management, Novartis Pharmaceuticals, Basel, Switzerland
Former CEO, Viron Therapeutics, Inc.
Co-founder and President of MedExact Inc., a company subsequently acquired
Karthik Radhakrishnan, Chief Financial Officer
10+ years of health care capital markets experience
Formerly, Vice President at ING Investment Management
MBA, MS in Engineering, CFA charter holder
Don Healey, Ph.D., Chief Scientific Officer
25+ years of experience in cellular immunology and immune regulation
Former Director of Immunology, Argos Therapeutics
Donna Rill, Chief Development Officer
30 years in cell and gene therapy research and clinical application
Designed and validated cGMP Cell & Gene Therapy Laboratories, Vector Production facilities, and Translational Research Labs
Kenny Frazier, VP of Clinical Dev.and Regulatory Affairs
24 years of extensive clinical and regulatory experience
Formerly, Head of Clinical Operations, Lexicon Pharmaceuticals and Tanox, Inc.
Board of Directors
Gail J. Maderis
CEO, BayBio, Former CEO of Five Prime Therapeutics, Founder of Genzyme Molecular Oncology
Michael S. Richman
CEO, Amplimmune
Scott B. Seaman
Executive Director, Alkek Foundation
David E. Jorden
Interim CEO, Nanospectra Biosciences
Neil K. Warma
CEO, Opexa
6 |
|

SPMS Scientific Advisory Board
Dawn McGuire, M.D., FAAN (Chair)
Advisory Council of the Gill Heart Institute
Former Vice President of Clinical Research at Elan Pharmaceuticals
Hans-Peter Hartung, M.D
Chair of Neurology at Heinrich-Heine University, Düsseldorf
President ECTRIMS, World Health Organization Advisory Board on MS
Mark S. Freedman, M.D.
Director of the Multiple Sclerosis Research Unit at Ottawa Hospital
Multiple Sclerosis Society of Canada, National MS Society (USA)
ACTRIMS committee member
Clyde Markowitz, M.D.
Director of MS Center at the University of Pennsylvania
Doug Arnold, M.D.
James McGill Professor Neurology and Neurosurgery at the Montreal Neurological Institute
Edward Fox, M.D., Ph.D.
Director of Multiple Sclerosis Clinic of Central Texas
Advisory Committee, Lone Star Chapter of the National Multiple Sclerosis Society
7 |
|

Pipeline and Potential Areas of Investigation
Stage of Development
Indication Evaluation Preclinical Phase I Phase II Phase III Commercial
T-CELL PLATFORM
Multiple Sclerosis (Tcelna)*
Secondary Progressive MS* Phase IIb ongoing
Relapsing Remitting MS* Phase III not yet initiated
STEM CELL PLATFORM
Type 1 Diabetes
* Rights granted to Merck Serono under option and license agreement (Feb. 5, 2013)
8

Tcelna in Secondary Progressive Multiple Sclerosis
Patients initially experience a relapsing-remitting course then transition to SPMS
SPMS patients experience worsening QOL / disability with or without relapses
Over 450,000 patients in North America and 2 million world wide have MS
Approximately 30-45% of MS patients can be classified as Secondary Progressive
Tcelna is being pursued for this SPMS indication
Potential SPMS market in the North America alone could exceed $7 billion [150,000 SPMS patients at average cost of $50,000 per year of treatment]
No SPMS treatment approved by EMA; only one approved by FDA with limited use due to toxicity
Upon successful clinical development, Tcelna has the potential to be the treatment of choice in SPMS
9
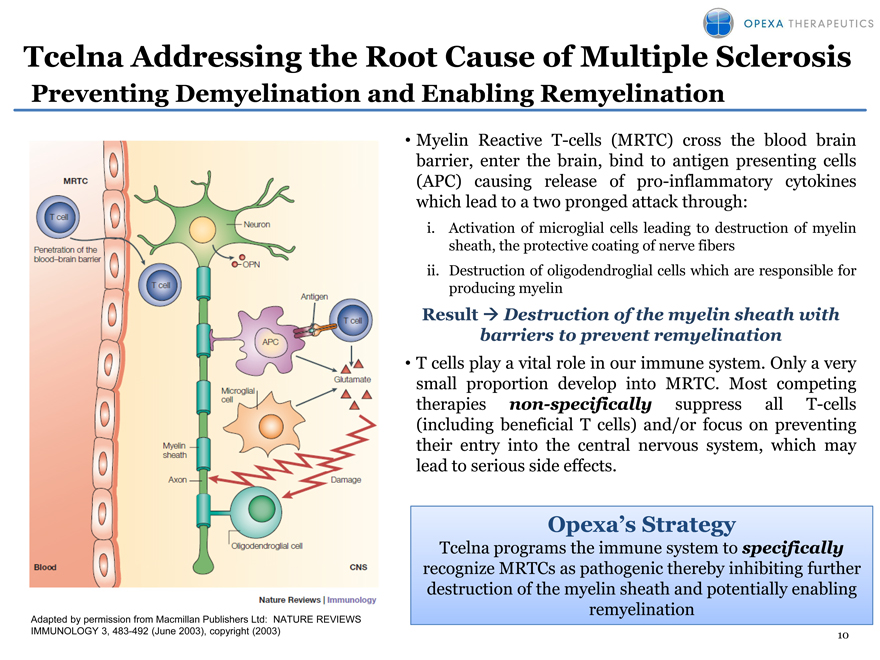
Tcelna Addressing the Root Cause of Multiple Sclerosis
Preventing Demyelination and Enabling Remyelination
Myelin Reactive T-cells (MRTC) cross the blood brain barrier, enter the brain, bind to antigen presenting cells (APC) causing release of pro-inflammatory cytokines which lead to a two pronged attack through: i. Activation of microglial cells leading to destruction of myelin sheath, the protective coating of nerve fibers ii. Destruction of oligodendroglial cells which are responsible for producing myelin
Result Destruction of the myelin sheath with barriers to prevent remyelination
T cells play a vital role in our immune system. Only a very small proportion develop into MRTC. Most competing therapies non-specifically suppress all T-cells (including beneficial T cells) and/or focus on preventing their entry into the central nervous system, which may lead to serious side effects.
Opexa’s Strategy
Tcelna programs the immune system to specifically recognize MRTCs as pathogenic thereby inhibiting further destruction of the myelin sheath and potentially enabling remyelination
Adapted by permission from Macmillan Publishers Ltd: NATURE REVIEWS
IMMUNOLOGY 3, 483-492 (June 2003), copyright (2003) 10
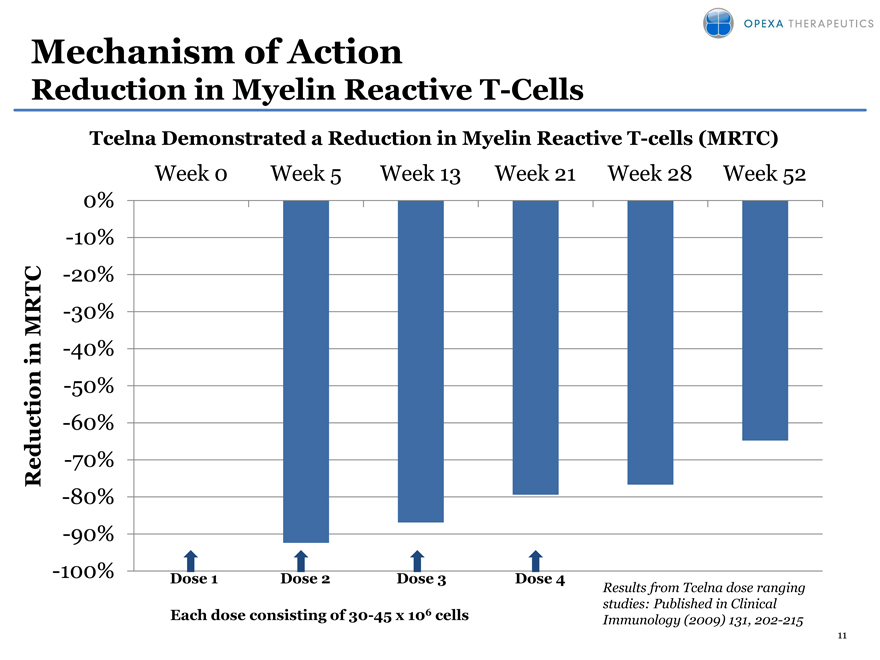
Mechanism of Action
Reduction in Myelin Reactive T-Cells
Tcelna Demonstrated a Reduction in Myelin Reactive T-cells (MRTC)
Week 0 Week 5 Week 13 Week 21 Week 28 Week 52
0%
-10%
C -20%
MRT -30%
in -40%
-50%
-60%
eduction -70%
R
-80%
-90%
-100% Dose 1 Dose 2 Dose 3 Dose 4
Each dose consisting of 30-45 x 106 cells
Results from Tcelna dose ranging studies: Published in Clinical Immunology (2009) 131, 202-215
11
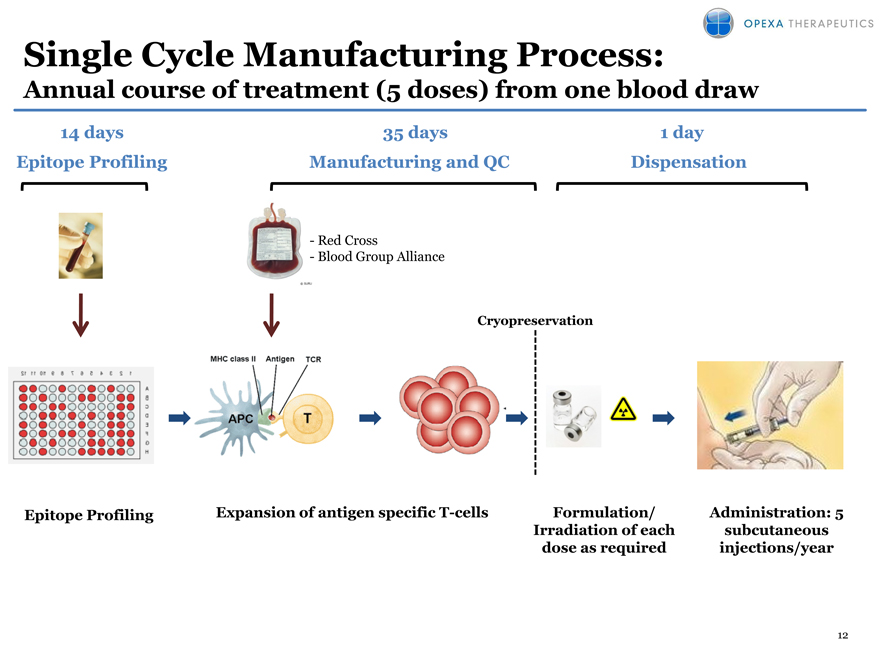
Single Cycle Manufacturing Process:
Annual course of treatment (5 doses) from one blood draw
14 days 35 days 1 day
Epitope Profiling Manufacturing and QC Dispensation
-Red Cross
—Blood Group Alliance
Cryopreservation
Epitope Profiling Expansion of antigen specific T-cells Formulation/ Administration: 5
Irradiation of each subcutaneous
dose as required injections/year
12

Merck Serono Agreement for MS indication
February 2013 option and license agreement with Merck Serono
Up to $220 million in additional payments
Option exercise $25 million for starting Phase III/ or $15 million if another Phase II
$35 million FDA filing, approval and commercialized in US
$30 million for EU filing, approval and commercialization in at least three countries
RRMS development and commercialization of up to $40 million
One time commercial milestones of up to $85 million
Royalties ranging from 8% to 15% of annual net sales with step-ups occurring when net sales exceed $500 million, $1 B & $2 B
Opexa maintains key rights:
Development and commercialization rights to Tcelna in Japan
Certain manufacturing rights
Co-development funding option in exchange for increased royalties
Rights to all other disease indications
13
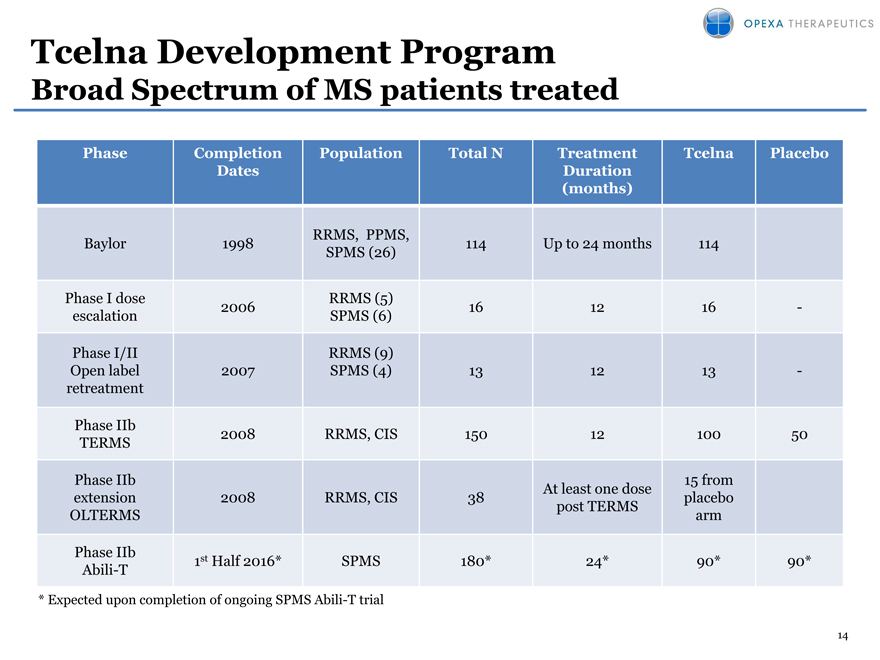
Tcelna Development Program
Broad Spectrum of MS patients treated
Phase Completion Population Total N Treatment Tcelna Placebo
Dates Duration
(months)
RRMS, PPMS,
Baylor 1998 114 Up to 24 months 114
SPMS (26)
Phase I dose RRMS (5)
2006 16 12 16 —
escalation SPMS (6)
Phase I/II RRMS (9)
Open label 2007 SPMS (4) 13 12 13 —
retreatment
Phase IIb 2008 RRMS, CIS 150 12 100 50
TERMS
Phase IIb 15 from
At least one dose
extension 2008 RRMS, CIS 38 placebo
post TERMS
OLTERMS arm
Phase IIb
Abili-T 1st Half 2016* SPMS 180* 24* 90* 90*
* |
| Expected upon completion of ongoing SPMS Abili-T trial |
14
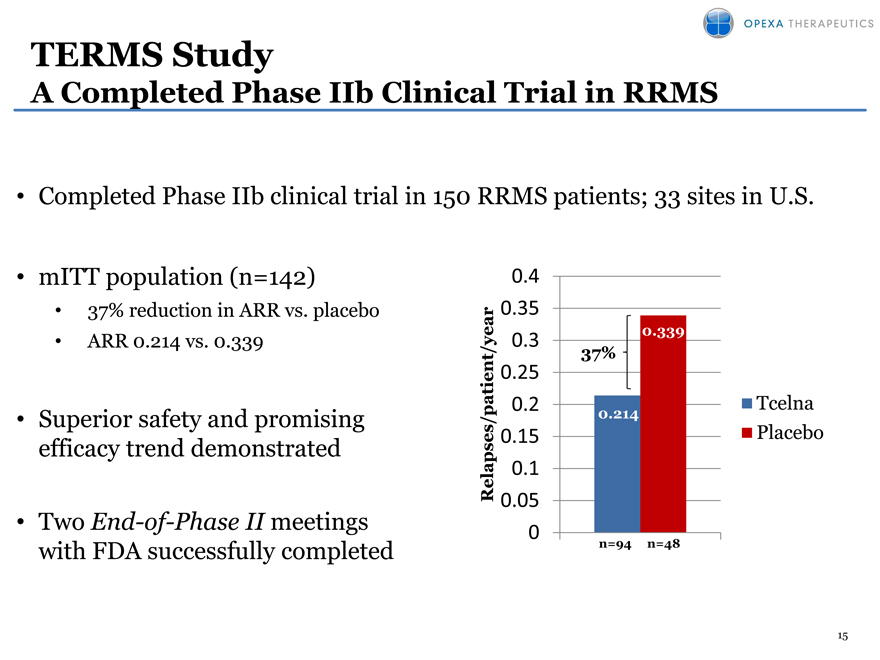
TERMS Study
A Completed Phase IIb Clinical Trial in RRMS
Completed Phase IIb clinical trial in 150 RRMS patients; 33 sites in U.S.
mITT population (n=142)
37% reduction in ARR vs. placebo
ARR 0.214 vs. 0.339
Superior safety and promising efficacy trend demonstrated
Two End-of-Phase II meetings with FDA successfully completed
0.4
0.35
0.3 0.339
nt/year 37%
0.25
0.2 Tcelna
0.214
apses/patie 0.15 Placebo
Rel 01.
0.05
0
n=94 n=48
15
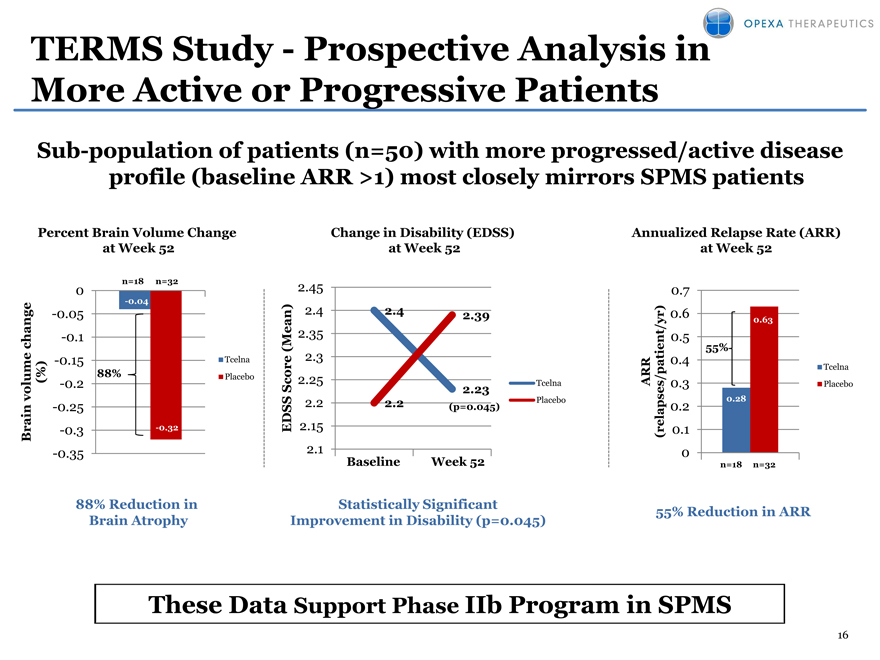
TERMS Study—Prospective Analysis in More Active or Progressive Patients
Sub-population of patients (n=50) with more progressed/active disease profile (baseline ARR >1) most closely mirrors SPMS patients
Percent Brain Volume Change Change in Disability (EDSS) Annualized Relapse Rate (ARR)
at Week 52 at Week 52 at Week 52
0 n=18 n=32 2.45 0.7
-0.04
-0.05 2.4 2.4 2.39 0.6 0.63
-0.1 2.35 0.5
55%
-0.15 Tcelna 2.3 0.4
change (Mean) patient/yr) Tcelna
me %) 88% Placebo ore RR
volu ( -0.2 Sc 2.25 2.23 Tcelna A 0.3 Placebo
-0.25 2.2 2.2 (p=0.045) Placebo 0.2 0.28
Brain -0.3 -0.32 EDSS 2.15 (relapses/ 0.1
-0.35 2.1 0
Baseline Week 52 n=18 n=32
88% Reduction in Statistically Significant 55% Reduction in ARR
Brain Atrophy Improvement in Disability (p=0.045)
These Data Support Phase IIb Program in SPMS
16
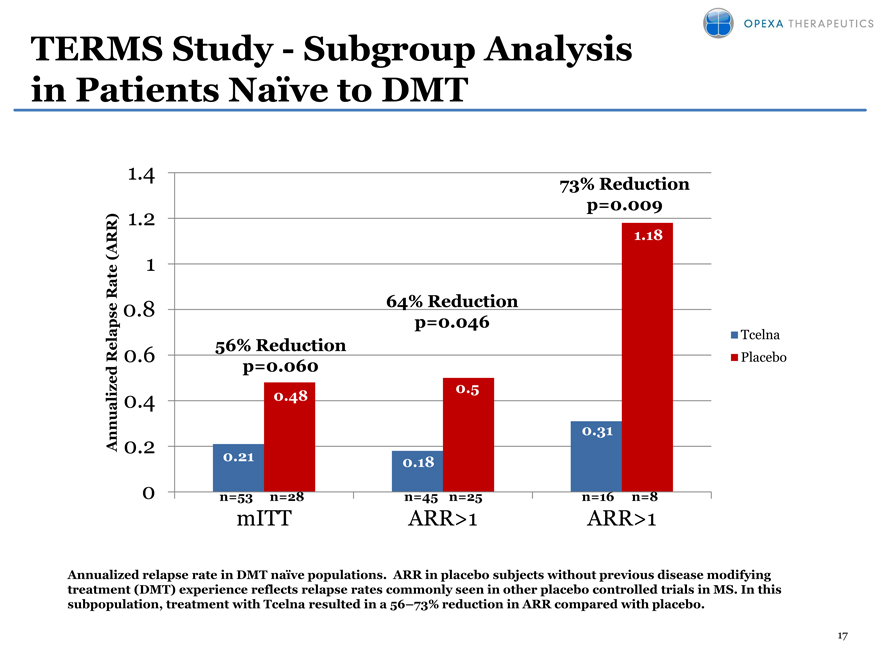
TERMS Study—Subgroup Analysis in Patients Naïve to DMT
1.4 73% Reduction
1.2 p=0.009
(ARR) 1.18
te 1
Ra 0.8 64% Reduction
p=0.046
Tcelna
Relapse 0.6 56% Reduction Placebo
d p=0.060
0.4 0.48 0.5
Annualize 0.31
0.2
0.21 0.18
0 n=53 n=28 n=45 n=25 n=16 n=8
mITT ARR>1 ARR>1
Annualized relapse rate in DMT naïve populations. ARR in placebo subjects without previous disease modifying treatment (DMT) experience reflects relapse rates commonly seen in other placebo controlled trials in MS. In this subpopulation, treatment with Tcelna resulted in a 56 73% reduction in ARR compared with placebo.
17

Secondary Progressive MS:
Overview of Phase I/II Patients treated with Tcelna
36 SPMS patients treated in three clinical studies
35 of these patients were treated with Tcelna for two years
26 patients treated at Baylor; 10 patients treated by Opexa
Promising efficacy observed
Disease stabilization as measured by EDSS was observed in 80% of patients at two years (compared to a historical control data that shows progression rate of 40%)
10 patients in Opexa sponsored study showed significant reduction in relapse rates from a baseline ARR of 0.5 to an ARR of 0.1 at two years
No worsening in key Quality of Life Indicators (physical and psychological condition) at two years
Well-tolerated with a favorable safety profile
No Serious Adverse Events
18
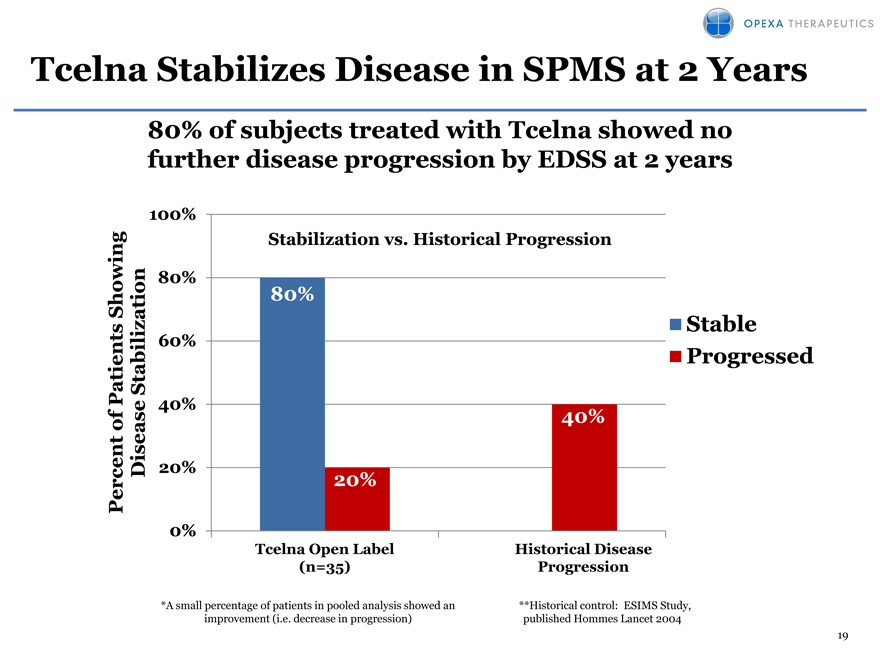
Tcelna Stabilizes Disease in SPMS at 2 Years
80% of subjects treated with Tcelna showed no further disease progression by EDSS at 2 years
100%
wing Stabilization vs. Historical Progression
n 80%
Sho 80%
Stable
60%
tients abilizatio Progressed
Pa St 40%
of 40%
ent Disease 20%
Perc 20%
0%
Tcelna Open Label Historical Disease
(n=35) Progression
*A small percentage of patients in pooled analysis showed an **Historical control: ESIMS Study,
improvement (i.e. decrease in progression) published Hommes Lancet 2004
19
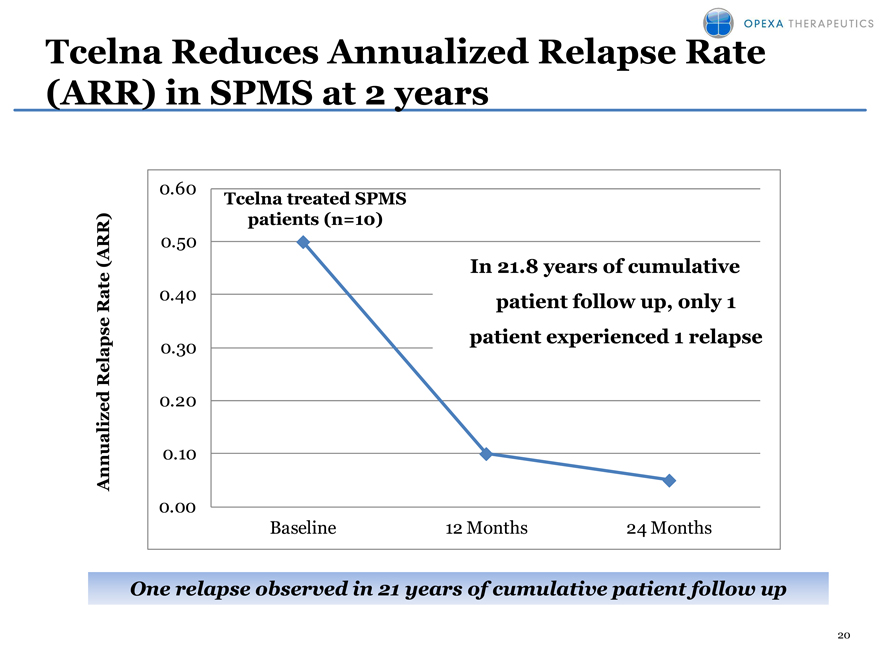
Tcelna Reduces Annualized Relapse Rate (ARR) in SPMS at 2 years
060. Tcelna treated SPMS
patients (n=10)
(ARR) 0.50
In 21.8 years of cumulative
e
Rat 0.40 patient follow up, only 1
elapse 0.30 patient experienced 1 relapse
R
0.20
nualized 0.10
An
0.00
Baseline 12 Months 24 Months
One relapse observed in 21 years of cumulative patient follow up
20
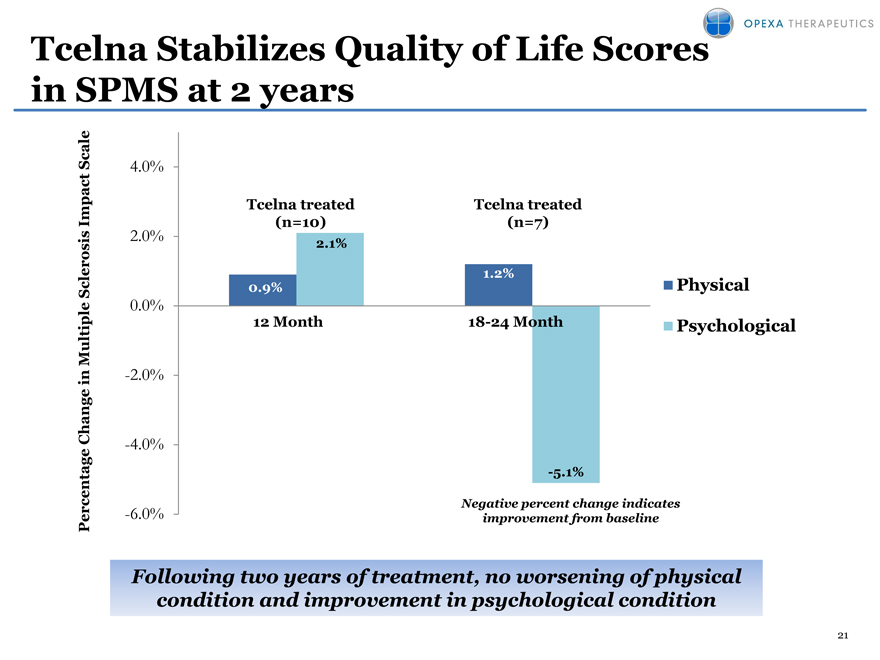
Tcelna Stabilizes Quality of Life Scores in SPMS at 2 years
Scale 4.0%
ct
Impa Tcelna treated Tcelna treated
(n=10) (n=7)
2.0% 2.1%
lerosis 1.2%
Sc 0.9% Physical
0.0%
Multiple 12 Month 18-24 Month Psychological
n -20%.
i
Change -4.0%
age -51%.
Negative percent change indicates
Percent -6.0% improvement from baseline
Following two years of treatment, no worsening of physical
condition and improvement in psychological condition
21

Abili-T : Landmark trial in SPMS
Abili-T Phase IIb clinical trial in SPMS is ongoing
Double-blind, 1:1 randomized, placebo-controlled
Inclusion criteria: Secondary Progressive MS with EDSS of 3 to 6
53 patients enrolled at June 14, 2013
Immune Monitoring program conducted on a blinded basis
Fast Track designation granted by FDA for Tcelna in SPMS
180 Patients expected to be enrolled
SPMS population
30 sites in USA and Canada
2 |
| annual courses of personalized therapy |
22

Efficacy Assessments
Primary Endpoint
Whole-brain atrophy
Secondary Endpoints
Sustained progression measured by EDSS
Time to sustained progression
T2 lesions progressing to hypointense lesions (black holes)
Change in EDSS
Annualized Relapse Rate (ARR)
Change in MSFC Assessment of disability
Change in Symbol Digit Modality Test (SDMT)
Exploratory Endpoints
Quality of life assessment by MSQLI
Gd-enhancing lesion volume with increasing MTR
Gd-enhancing lesion volume with decreasing MTR
Change in MTR in normal-appearing white matter
Changes in T-regulatory cell repertoire and function
23

Immune Monitoring Program
Trial initiation 40 patients at dose 5 90 patients at dose 5 180 patients at 10 doses End of Study
2012 2013 2014 2015 2016
Immuno vigilance Phenotypical Analysis Plausible indicators of
(CD4 and CD8) Tcelna efficacy
Whole blood analysis:
Absolute frequency of T- Reduction in Th1/TH17
cells, B-cells, Dendritic cells Pro-inflammatory Anti-inflammatory Increase in Treg/Tr1 cells
and Monocytes TH1 Treg Reduction in proinflammatory
TH17 Tr1 cytokines (IL-12, IL-23, IFN ?,
TH2 TNF?)
Goal is to show no systemic
impact on immune Increase in anti-inflammatory
IL-2 IL-4 cytokines (IL-10, IL-27)
response, i.e. Tcelna is a IL-6 IL-5
Precision Immunotherapy IL-12 IL-10 Loss of proinflammatory
IL-17E, IL-17F monocyte markers
Tcelna differentiates by not (CD16/HLA-DR, CCR5)
IL-23 IL-27
depleting a broad spectrum TNF? TGF? Gain in anti-inflammatory
of T-cells IFN BDNF monocyte markers (PDL-1,
?
HLA-G, ICOS)
24

Financials
Pro forma as of June 30, 2013
Cash and Cash Equivalents (MM) $ 5.0
Convertible Debt (MM) (1) $ 3.2
Shares Outstanding (MM) ~8.1
Warrants (MM) (2) ~3.1
Stock Options (MM) (3) ~1.1
(1) Principal outstanding on convertible debt (2) Weighted average exercise price =$4.12 (3) Weighted average exercise price = $4.46
25

Goals and Milestones
Present Immune Monitoring base line data at ACTRIMS Conference (2013)
Activate Canadian sites and initiate enrollment in Canada (2013)
Activate and initiate all Abili-T clinical sites in U.S. (2013)
Advance and complete enrollment in Phase IIb Abili-T study in SPMS (2013-14)
Evaluate and present interim immune monitoring data (2013-15)
Evaluate potential of T-cell platform in additional autoimmune diseases (2013-14)
26

Investment Highlights
Lead product, Tcelna®, a T-cell immunotherapy for Multiple Sclerosis (MS)
Currently conducting a Phase IIb clinical trial in Secondary Progressive MS (SPMS)
Limited treatment options currently available for SPMS
Potential SPMS market in North America alone could exceed $7 Billion
Tcelna positioned as potential first-to-market personalized T-cell immunotherapy
Option and license agreement secured with Merck Serono for Tcelna in MS indications only, worldwide excluding Japan
Opexa’s in-house cGMP manufacturing enables close control of process and COGS
In previous MS clinical studies, Tcelna has demonstrated good safety and potential indications of clinical efficacy
FDA has granted Opexa Fast Track Designation for Tcelna in SPMS
27

APPENDIX
28

Tcelna® Highlights
Clinical studies conducted in Relapsing Remitting and Secondary Progressive MS
Progress made on manufacturing with focus on commercially viable process
? Over 850 Tcelna preparations have been successfully manufactured, reproducible and consistent
Safety demonstrated and trends in clinical efficacy across broad spectrum of patients for relapsing remitting and secondary progressive MS
Dose and regimen for ongoing clinical development has been selected based on dose ranging studies
Commercial opportunities in MS:
Secondary Progressive MS
Abili-T Phase II SPMS trial ongoing with 180 patients expected in U.S. and Canada
Fast track designation
Relapsing Remitting MS
Formal End of Phase II meetings have been conducted
FDA feedback obtained for potential Phase III studies
29

Tcelna Manufacturing:
Personalization followed by Expansion Step
The Epitope Profiling Assay (EPA)
Screen peripheral blood for Myelin-Reactive T-cells (MRTCs), and mapping of immunodominant epitopes to MBP, MOG and PLP
109 overlapping peptides encompassing MBP, MOG and PLP
Interferon gamma response to individual peptide pools defines positive response in 7 day assay
ImmPathTM Process
Procure unit of blood from which up to six T-cell lines reactive with immunodominant myelin peptides are generated and pooled as a patient-specific Tcelna product
Manufacturing performed under GMP/GTPs in functionally closed system
Process generates a year of Tcelna doses from a single unit of blood
30
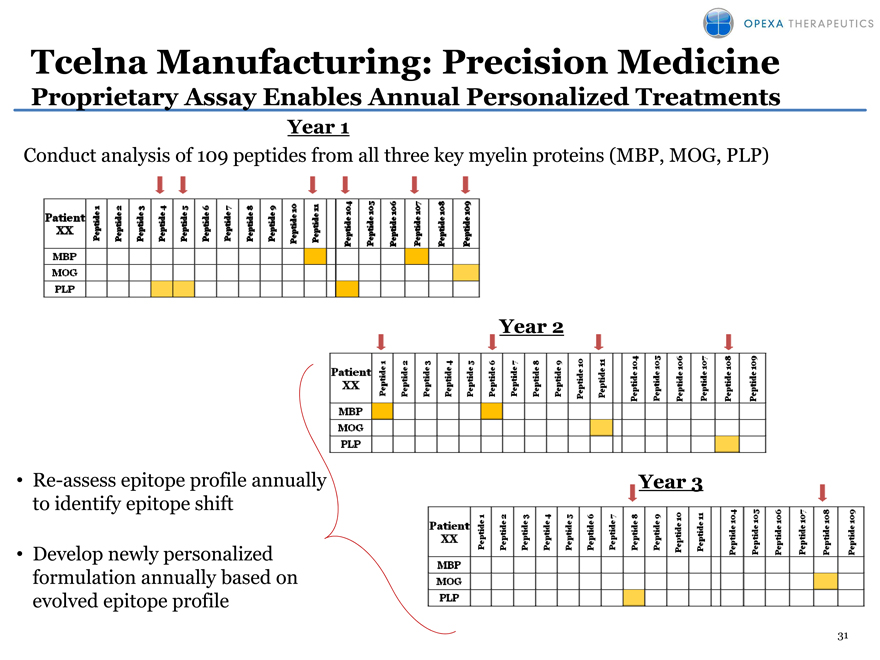
Tcelna Manufacturing: Precision Medicine
Proprietary Assay Enables Annual Personalized Treatments
Year 1
Conduct analysis of 109 peptides from all three key myelin proteins (MBP, MOG, PLP)
Re-assess epitope profile annually to identify epitope shift
Develop newly personalized formulation annually based on evolved epitope profile
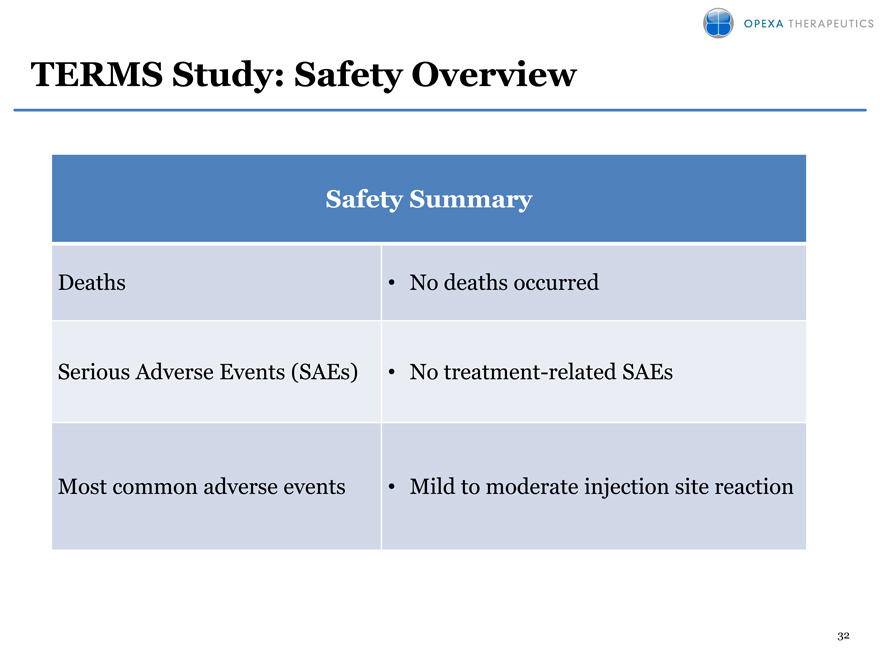
TERMS Study: Safety Overview
Safety Summary
Deaths No deaths occurred
Serious Adverse Events (SAEs) No treatment-related SAEs
Most common adverse events Mild to moderate injection site reaction
32
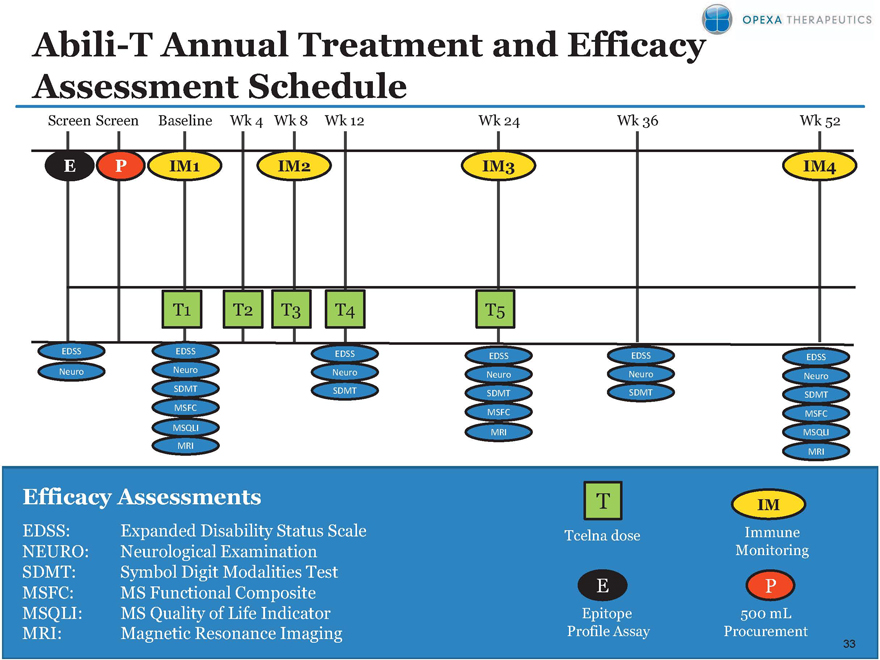
Abili-T Annual Treatment and Efficacy Assessment Schedule
Screen Screen Baseline Wk 4 Wk 8 Wk 12 Wk 24 Wk 36 Wk 52
P IM1 IM2 IM3 IM4
T1 T2 T3 T4 T5
EDSS EDSS EDSS EDSS EDSS EDSS
Neuro Neuro Neuro Neuro Neuro Neuro
SDMT SDMT SDMT SDMT SDMT
MSFC MSFC MSFC
MSQLI MRI MSQLI
MRI
MRI
Efficacy Assessments T IM
EDSS: Expanded Disability Status Scale Tcelna dose Immune
NEURO: Neurological Examination Monitoring
SDMT: Symbol Digit Modalities Test
MSFC: MS Functional Composite P
MSQLI: MS Quality of Life Indicator Epitope 500 mL
MRI: Magnetic Resonance Imaging Profile Assay Procurement
33

Mechanism of Action
Attenuated, patient-specific (autologous) myelin reactive T-cells (MRTC)
MRTC expanded ex vivo in response to immunodominant peptides of MBP, MOG and PLP
Therapeutic sc dosing (30-45 x 106 cells) stimulates host reactivity to the ‘over-represented’ MRTC inducing a dominant negative ‘regulatory T-cell’ response leading to:
Down-regulation of similar endogenous disease-causing myelin reactive T-cells
Potential to induce up-regulation of regulatory cells (Foxp3+ and Tr1 cells) to reduce inflammation and provide possible neuroprotection, should these gain entry to the CNS
Phase I/II studies conducted with Tcelna in RRMS and SPMS showed a substantial reduction in MRTC, and improved clinical outcomes
lower relapse rates and stabilization of disease as defined by EDSS
34
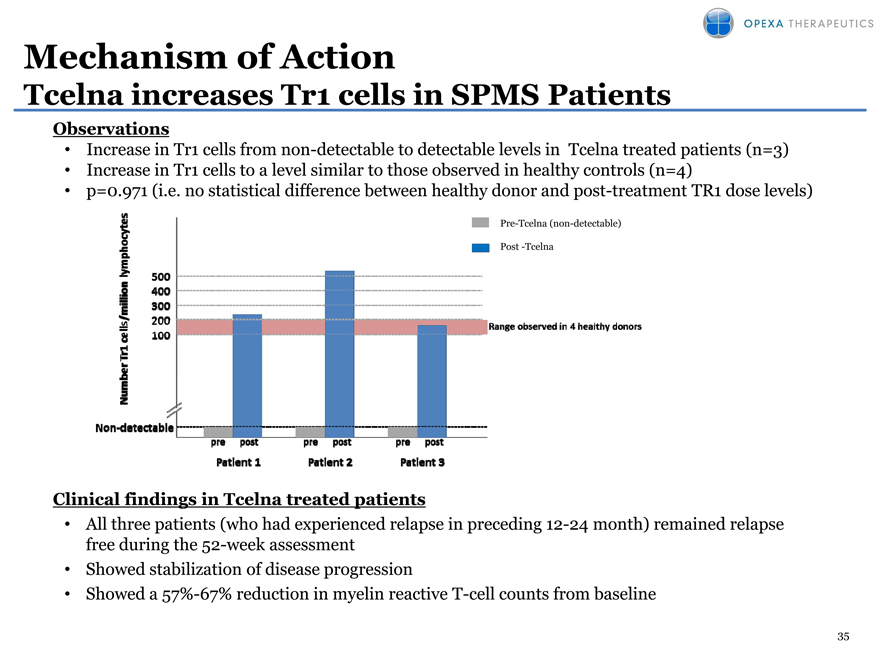
Mechanism of Action
Tcelna increases Tr1 cells in SPMS Patients
Observations
Increase in Tr1 cells from non-detectable to detectable levels in Tcelna treated patients (n=3)
Increase in Tr1 cells to a level similar to those observed in healthy controls (n=4)
p=0.971 (i.e. no statistical difference ifference between etween healthy donor onor and post ost-treatment TR1 dose ose levels)
Pre-Tcelna (non-detectable)
Post -Tcelna
Clinical findings in Tcelna treated patients
All three patients (who had experienced relapse in preceding 12-24 month) remained relapse free during the 52-week assessment
Showed stabilization of disease progression
Showed a 57%-67% reduction in myelin reactive T-cell counts from baseline
35

Mechanism of Action
The Immunopathology of SPMS
SPMS is associated with compartmentalized CNS inflammatory cells
- Microglia activation suggests ongoing chronic innate immune responses
SPMS is associated with inflammatory processes localized within the central nervous system
- Mechanisms responsible for continued neurodegeneration in SPMS are distinct from those of RRMS
- In SPMS, it is believed that the Blood Brain Barrier (BBB) remains closed enabling only those products that are able to cross the BBB to be potentially effective
- SPMS is characterized by a chronic inflammatory process (vs. acute episodes for RRMS)
Immunological defects associated with progression to SPMS
- Levels of the anti-inflammatory cytokine IL-10 are reduced in patients with SPMS
- A therapeutic that has the potential to restore local production of IL-10 would be expected to alleviate chronic inflammation, and thereby reduce neurodegeneration
36
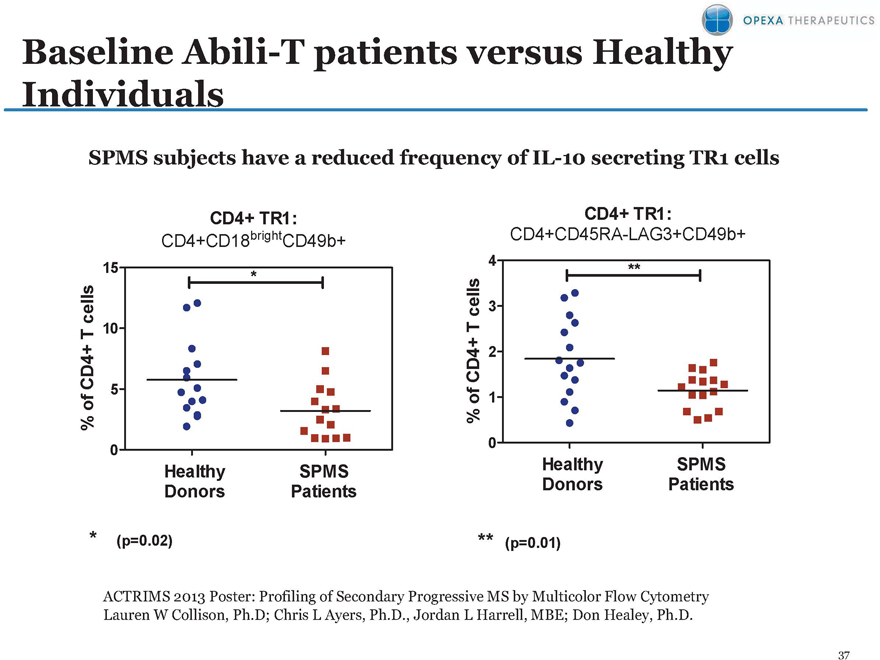
Baseline Abili-T patients versus Healthy Individuals
SPMS subjects have a reduced frequency of IL-10 secreting TR1 cells
CD4+ TR1: CD4+ TR1:
CD4+CD18brightCD49b+ CD4+CD45RA-LAG3+CD49b+
4 |
|
15 * **
s
cells cell 3
T 10 T
CD4+ CD4+ 2
5 |
|
of of 1
% %
0 0
Healthy SPMS Healthy SPMS
Donors Patients Donors Patients
** (p=0.02) ** (p=0.01)
ACTRIMS 2013 Poster: Profiling of Secondary Progressive MS by Multicolor Flow Cytometry Lauren W Collison, Ph.D; Chris L Ayers, Ph.D., Jordan L Harrell, MBE; Don Healey, Ph.D.
37
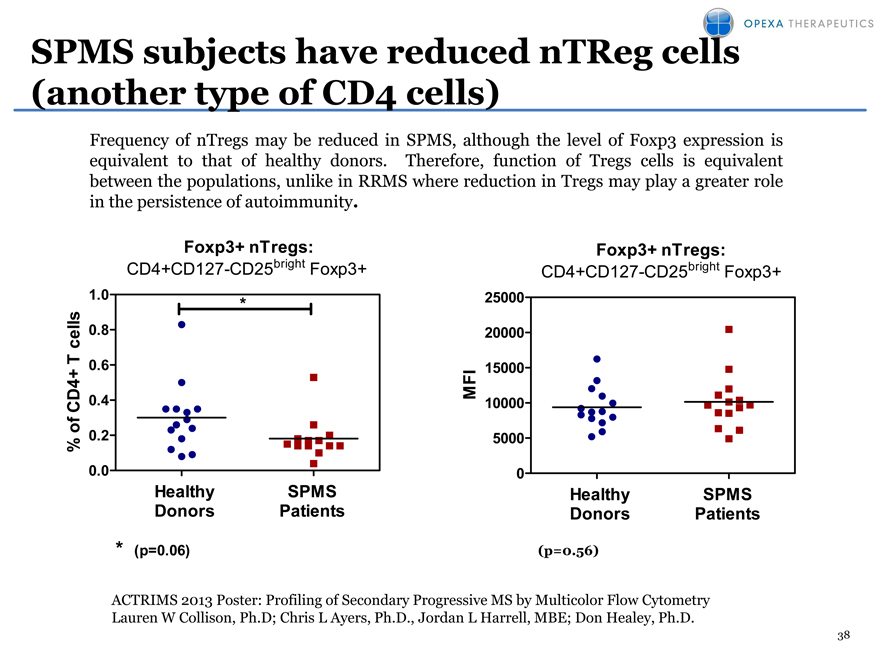
SPMS subjects have reduced nTReg cells (another type of CD4 cells)
Frequency of nTregs may be reduced in SPMS, although the level of Foxp3 expression is equivalent to that of healthy donors. Therefore, function of Tregs cells is equivalent between the populations, unlike in RRMS where reduction in Tregsmay play a greater role in the persistence of autoimmunity.
Foxp3+ nTregs: Foxp3+ nTregs:
CD4+CD127-CD25bright Foxp3+ CD4+CD127-CD25bright Foxp3+
1.0 * 25000
cells 0.8 20000
T 0.6 15000
+ I
CD4 0.4 MF 10000
% of 0.2 5000
00. |
| 0 |
Healthy SPMS Healthy SPMS
Donors Patients Donors Patients
* |
| (p=0.06) (p=0.56) |
ACTRIMS 2013 Poster: Profiling of Secondary Progressive MS by Multicolor Flow Cytometry Lauren W Collison, Ph.D; Chris L Ayers, Ph.D., Jordan L Harrell, MBE; Don Healey, Ph.D.
38
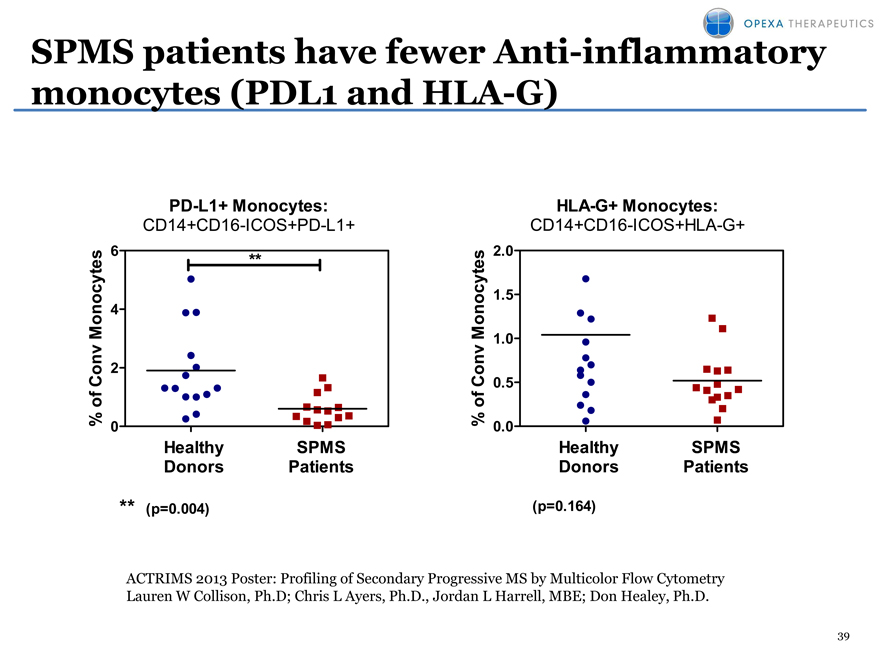
SPMS patients have fewer Anti-inflammatory monocytes (PDL1 and HLA-G)
PD-L1+ Monocytes: HLA-G+ Monocytes:
CD14+CD16-ICOS+PD-L1+ CD14+CD16-ICOS+HLA-G+
6 |
| 2.0 |
ytes ** ytes
1.5
Monoc 4 Monoc
1.0
onv 2 onv
C C 0.5
of of
% 0 % 0.0
Healthy SPMS Healthy SPMS
Donors Patients Donors Patients
** (p=0.004) (p=0.164)
ACTRIMS 2013 Poster: Profiling of Secondary Progressive MS by Multicolor Flow Cytometry Lauren W Collison, Ph.D; Chris L Ayers, Ph.D., Jordan L Harrell, MBE; Don Healey, Ph.D.
39