 Issuer Free Writing Prospectus Filed Pursuant to Rule 433 Registration No. 333-189395 July 29, 2013 |
 2 EXPLANATORY NOTE: The attached pages update the corporate presentation filed as an issuer free writing prospectus on July 22, 2013: • Slide 16, number of patients identified above the left graph for Percent Brain Volume Change at Week 52: – n=32 corresponds with bar showing -0.04 percent brain volume change; and – n=18 corresponds with bar showing -0.32 percent brain volume change. [Note: the corporate presentation prior to this update had the patient numbers (i.e., n=32 and n=18) reversed.] • Slide 16, number of patients identified below the right graph for Annualized Relapse Rate (ARR) at Week 52: – n=32 corresponds with bar showing 0.28 ARR; and – n=18 corresponds with bar showing 0.63 ARR. [Note: the corporate presentation prior to this update had the patient numbers (i.e., n=32 and n=18) reversed.] • Slide 17, ARR below the middle graph is > 1. [Note: the corporate presentation prior to this update reflected “>1” rather than “>1.”] • Slide 22, 65 patients were enrolled in the Abili-T trial at July 19, 2013. [Note: the corporate presentation prior to this update reflected 53 patients enrolled at June 14, 2013.] |
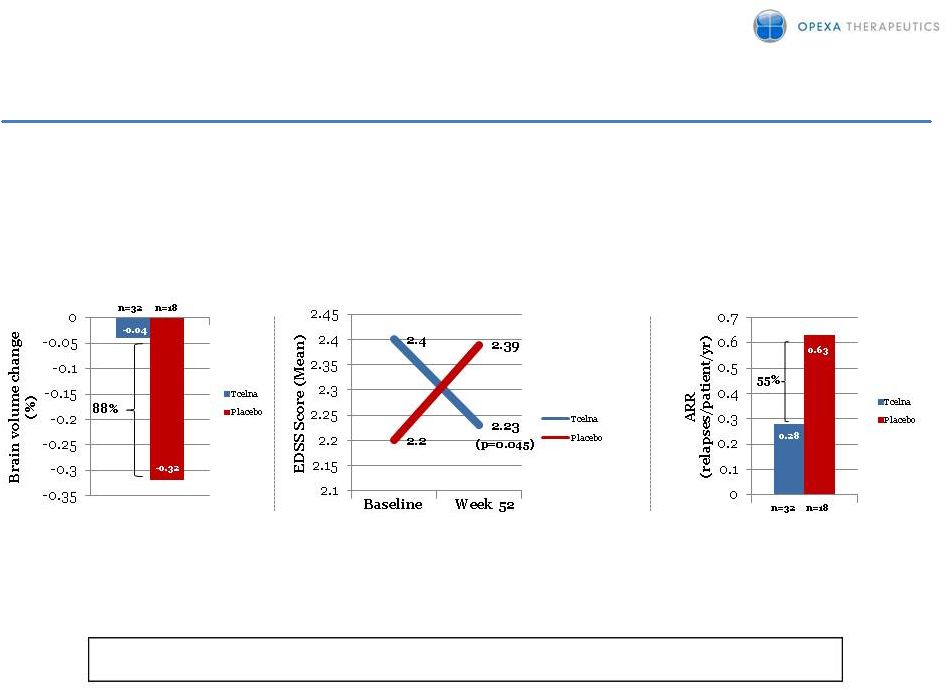 3 TERMS Study - Prospective Analysis in More Active or Progressive Patients Sub-population of patients (n=50) with more progressed/active disease profile (baseline ARR >1) most closely mirrors SPMS patients 88% Reduction in Brain Atrophy Percent Brain Volume Change at Week 52 Statistically Significant Improvement in Disability (p=0.045) 55% Reduction in ARR Annualized Relapse Rate (ARR) at Week 52 Change in Disability (EDSS) at Week 52 These Data Support Phase IIb Program in SPMS |
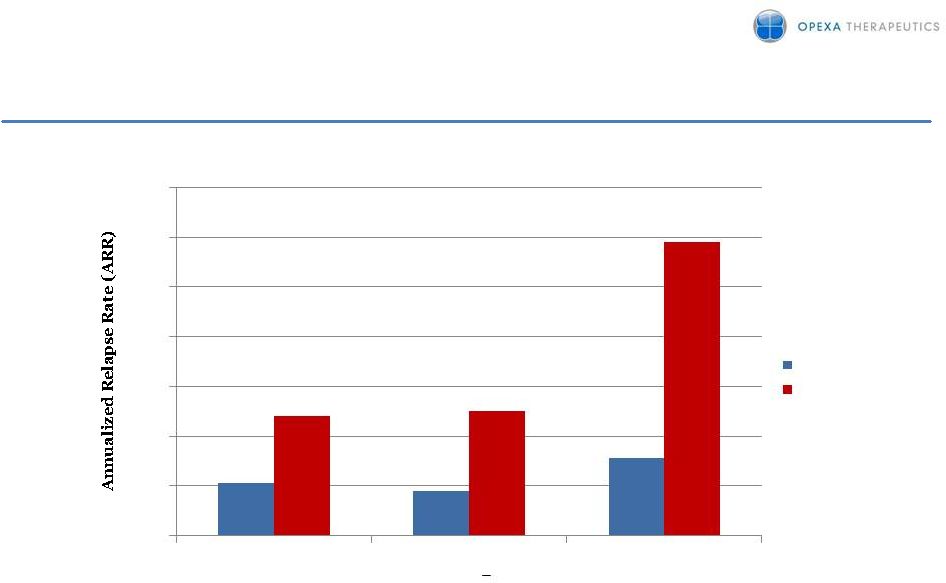 4 TERMS Study - Subgroup Analysis in Patients Naïve to DMT Annualized relapse rate in DMT naïve populations. ARR in placebo subjects without previous disease modifying treatment (DMT) experience reflects relapse rates commonly seen in other placebo controlled trials in MS. In this subpopulation, treatment with Tcelna™ resulted in a 56–73% reduction in ARR compared with placebo. 64% Reduction p=0.046 73% Reduction p=0.009 56% Reduction p=0.060 n=25 n=45 n=8 n=16 n=28 n=53 mITT ARR > 1 ARR>1 Tcelna Placebo 0 0.2 0.4 0.6 0.8 1 1.2 1.4 0.21 0.18 0.31 0.48 0.5 1.18 |
 5 Abili-T : Landmark trial in SPMS • Abili-T Phase IIb clinical trial in SPMS is ongoing – Double-blind, 1:1 randomized, placebo-controlled – Inclusion criteria: Secondary Progressive MS with EDSS of 3 to 6 – 65 patients enrolled at July 19, 2013 – Immune Monitoring program conducted on a blinded basis • Fast Track designation granted by FDA for Tcelna in SPMS • 180 Patients expected to be enrolled – SPMS population – 30 sites in USA and Canada • 2 annual courses of personalized therapy |