Exhibit 99.3

Administering AML - directed DLIs to patients with AML or MDS Post - Allogeneic HSCT Relapse Premal Lulla, Swati Naik, Ifigeneia Tzannou, Shivani Mukhi, Manik Kuvalekar, Catherine Robertson, Carlos A Ramos, George Carrum, Rammurti Kamble, Jasleen Randhawa, Adrian P Gee, Bambi Grilley Malcolm K Brenner, Helen E Heslop, Juan F Vera and Ann M Leen

Acute Myeloid Leukemia 16% Koreth J et al, DeAngelo DJ JAMA 2009 20% Cured 80% Adverse risk/relapsed AML ~26,000/yr Allo - HSCT potentially curative Adverse risks >CR2 Complex cytogenetics Monosomy 7 MDS AML t - AML MLL - r FLT3/DNMT3A etc
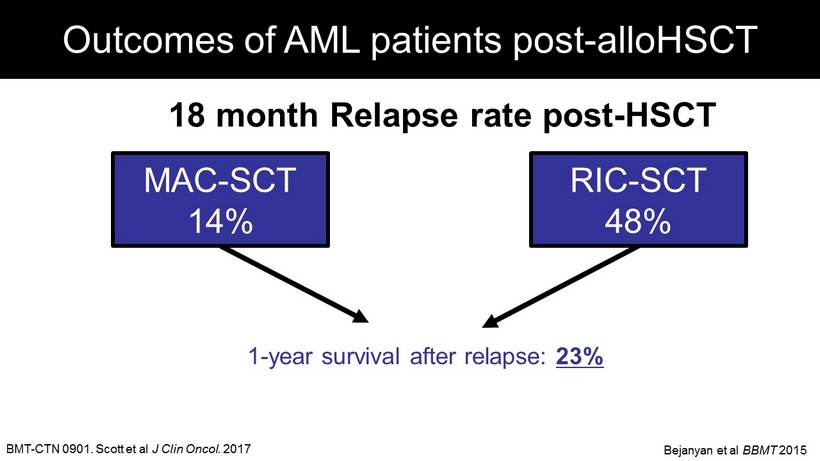
BMT - CTN 0901. Scott et al J Clin Oncol. 2017 Outcomes of AML patients post - alloHSCT 1 - year survival after relapse: 23% 18 month Relapse rate post - HSCT MAC - SCT 14% RIC - SCT 48% Bejanyan et al BBMT 2015
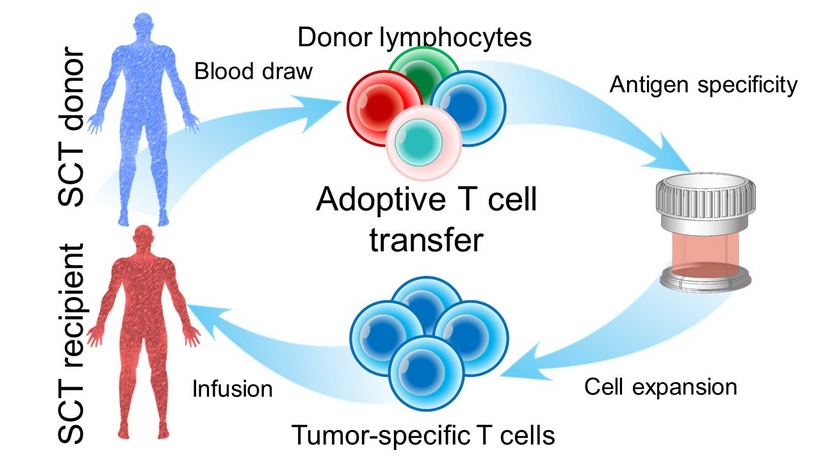
Infusion SCT recipient Cell expansion Tumor - specific T cells Antigen specificity Blood draw Donor lymphocytes Adoptive T cell transfer SCT donor
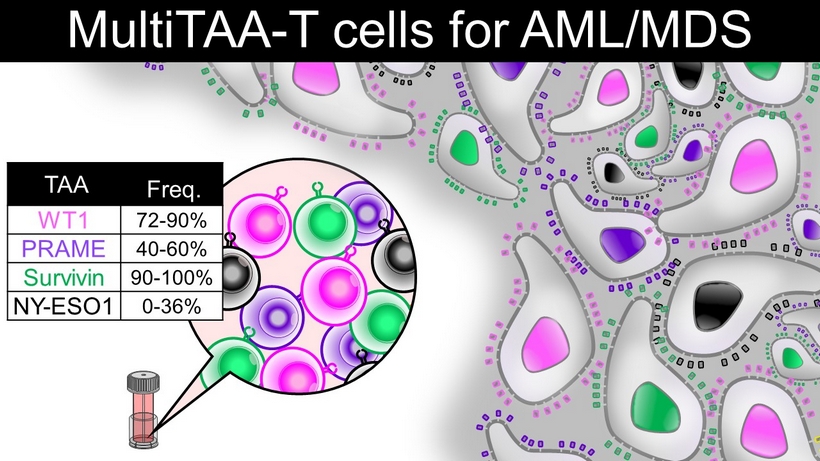
MultiTAA - T cells for AML/MDS TAA Freq. WT1 72 - 90% PRAME 40 - 60% Survivin 90 - 100% NY - ESO1 0 - 36%

• Simultaneously target multiple TAAs • Target multiple epitopes (CD4 and CD8) within each antigen • T cells with native T cell receptor specificity (non - engineered) Our approach

MultiTAA Manufacture DC Overlapping pepmixes PBMCs MultiTAA T cells
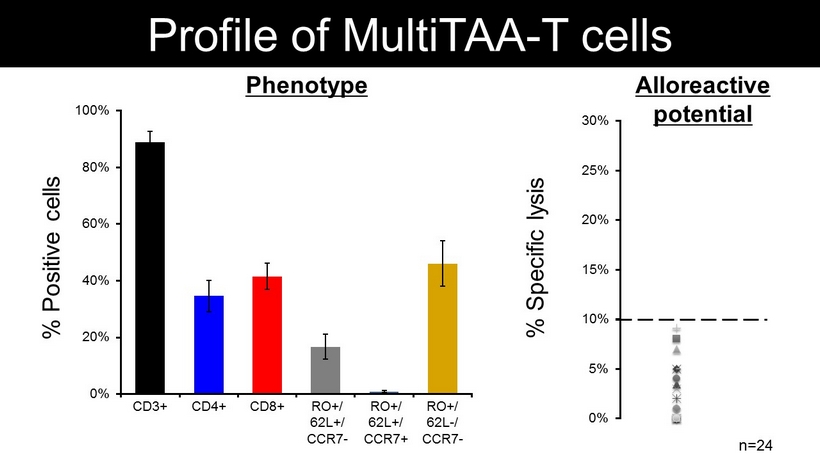
CD3+ CD4+ CD8+ RO+/ 62L+/ CCR7 - Profile of MultiTAA - T cells % Positive cells Phenotype % Specific lysis n=24 0% 5% 10% 15% 20% 25% 30% 0% 20% 40% 60% 80% 100% RO+/ 62L+/ CCR7+ RO+/ 62L - / CCR7 - Alloreactive potential
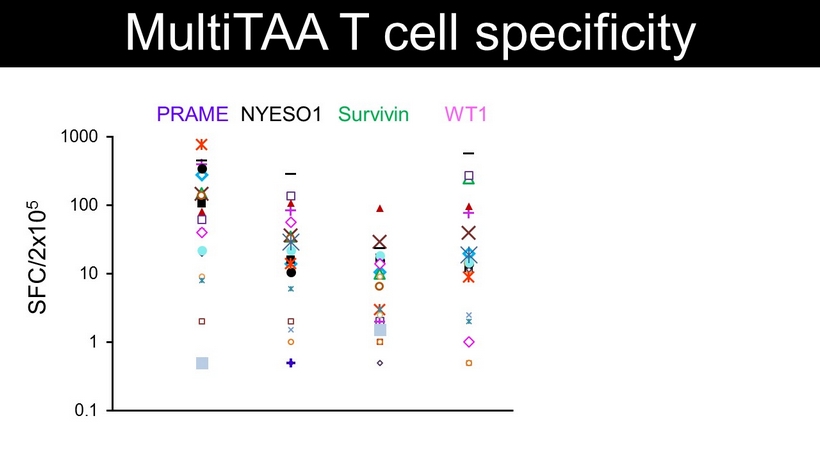
PRAME WT1 NYESO1 Survivin 0.1 1 10 100 1000 MultiTAA T cell specificity SFC/2x10 5
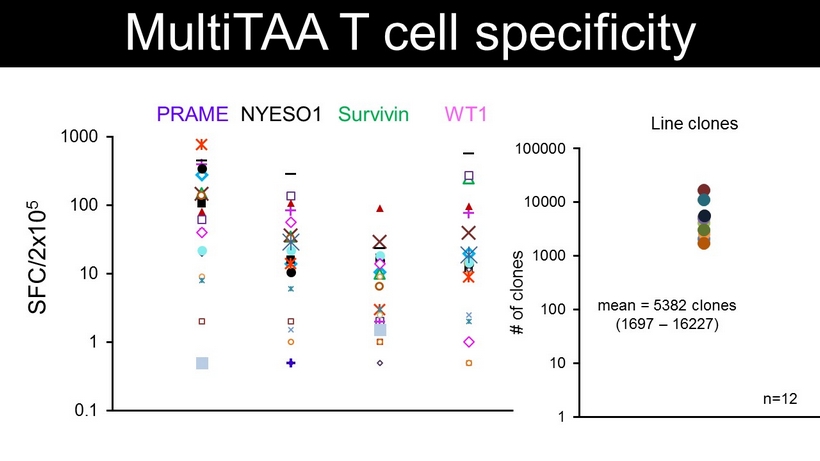
PRAME WT1 NYESO1 Survivin 0.1 1 10 100 1000 MultiTAA T cell specificity SFC/2x10 5 Line clones 1 10 100 1000 10000 100000 # of clones mean = 5382 clones (1697 – 16227) n=12

Any patient with AML/MDS post allo - HSCT Phase I trial - ADSPAM AML/MDS patients 30 days post allo - HSCT AML/MDS patients 30 days post allo - HSCT GROUP B – Active disease GROUP A - Adjuvant DL1 5x10 6 cells/m 2 DL2 1x10 7 cells/m 2 DL3 2x10 7 cells/m 2 Dose Escalation Donor - derived multiTAA T cells

Clinical trial: Current status 27 patients (24 AML and 3 MDS) Patients Enrolled: 20 patients (0.5 - 2x10 7 cells/m 2 ) Patients Treated: Grade II or lesser Grade III 3 (all grade I elevation in LFTs 1 grade III LFT elevation (resolved with 0.5 mg/kg prednisone)
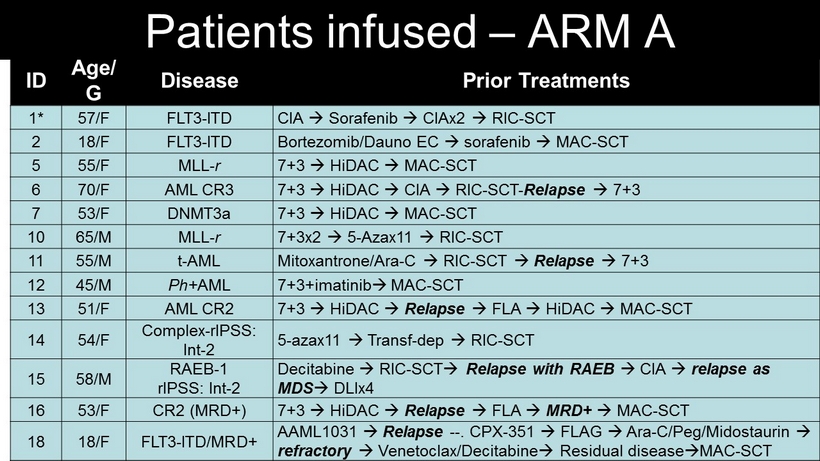
ID Age/ G Disease Prior Treatments 1* 57/F FLT3 - ITD CIA Sorafenib CIAx2 RIC - SCT 2 18/F FLT3 - ITD Bortezomib / Dauno EC sorafenib MAC - SCT 5 55/F MLL - r 7+3 HiDAC MAC - SCT 6 70/F AML CR3 7+3 HiDAC CIA RIC - SCT - Relapse 7+3 7 53/F DNMT3a 7+3 HiDAC MAC - SCT 10 65/M MLL - r 7+3x2 5 - Azax11 RIC - SCT 11 55/M t - AML Mitoxantrone /Ara - C RIC - SCT Relapse 7+3 12 45/M Ph+ AML 7+3+imatinib MAC - SCT 13 51/F AML CR2 7+3 HiDAC Relapse FLA HiDAC MAC - SCT 14 54/F Complex - rIPSS : Int - 2 5 - azax11 Transf - dep RIC - SCT 15 58/M RAEB - 1 rIPSS : Int - 2 Decitabine RIC - SCT Relapse with RAEB CIA relapse as MDS DLIx4 16 53/F CR2 (MRD+) 7+3 HiDAC Relapse FLA MRD+ MAC - SCT 18 18/F FLT3 - ITD/MRD+ AAML1031 Relapse -- . CPX - 351 FLAG Ara - C/Peg/ Midostaurin refractory Venetoclax / Decitabine Residual disease MAC - SCT Patients infused – ARM A
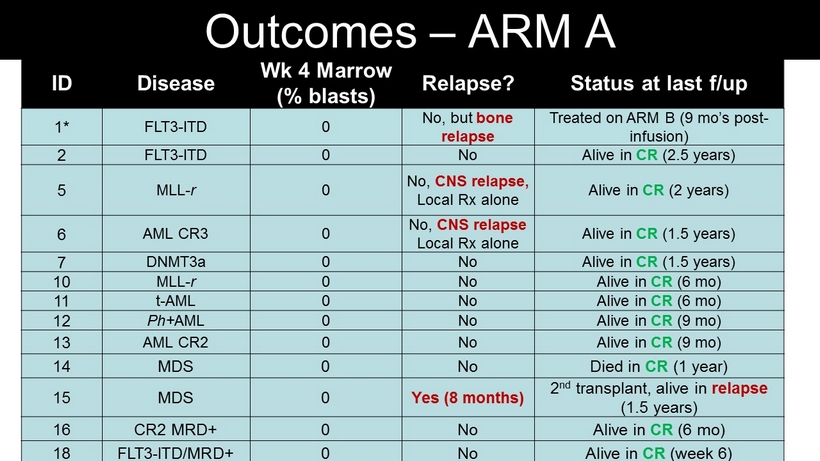
ID Disease Wk 4 Marrow (% blasts) Relapse? Status at last f/up 1* FLT3 - ITD 0 No, but bone relapse Treated on ARM B (9 mo’s post - infusion) 2 FLT3 - ITD 0 No Alive in CR (2.5 years) 5 MLL - r 0 No, CNS relapse, Local Rx alone Alive in CR (2 years) 6 AML CR3 0 No, CNS relapse Local Rx alone Alive in CR (1.5 years) 7 DNMT3a 0 No Alive in CR (1.5 years) 10 MLL - r 0 No Alive in CR (6 mo ) 11 t - AML 0 No Alive in CR (6 mo ) 12 Ph+ AML 0 No Alive in CR (9 mo ) 13 AML CR2 0 No Alive in CR (9 mo ) 14 MDS 0 No Died in CR (1 year) 15 MDS 0 Yes (8 months ) 2 nd transplant, alive in relapse (1.5 years) 16 CR2 MRD+ 0 No Alive in CR (6 mo ) 18 FLT3 - ITD/MRD+ 0 No Alive in CR (week 6) Outcomes – ARM A

ID Ag e/G Disease Prior Treatments 3 70/M IDH1 mut 7+3 decitabine IDH inhib cutis relapse CIA RIC - SCT Relapse 4 16/M MDS AML Double cord SCT AML Relapse C haplo - SCTx2 Relapse 1* 57/F FLT3 - ITD CIA Sorafenib CIAx2 RIC - SCT mTAA - T cells steroids Relapse 8 55/M Induc . failure 7+3 HiDAC x4 RIC - SCT Relapse DLIx4 MEC 5 - aza Relapse 9 23/M Del 17p CIAx3 haplo - SCT Relapse CIA - decitabine haplo - SCT 5 - aza Nivolumab CD123 BiTE MEC - decitabine midostaurin Relapse 9* 23/M Del 17p CIAx3 haplo - SCT#1 Relapse CIA - decitabine haplo - SCT#2 5 - aza Nivolumab CD123 BiTE MEC - decitabine midostaurin Relapse mTAA T cells haplo - SCT#3 Relapse 17 20/F FLT3 - ITD 7+3 HiDAC MAC - SCT Relapse CIA Relapse GROUP B: Active AML: 7 patients treated for active AML Patients infused – ARM B
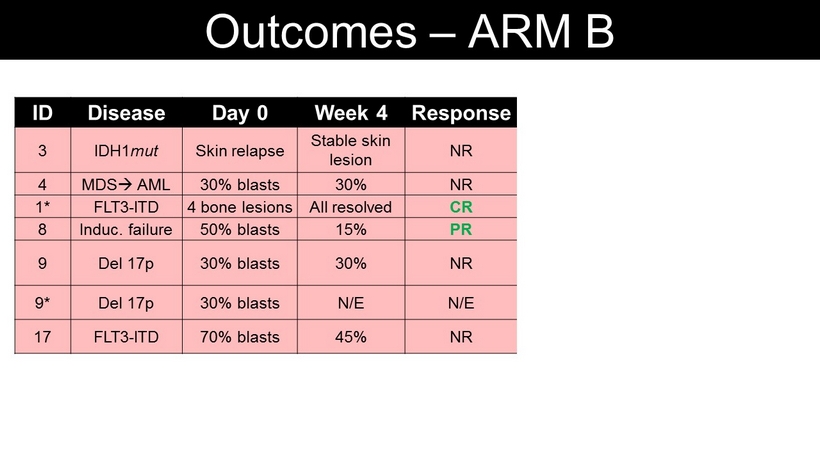
Outcomes – ARM B ID Disease Day 0 Week 4 Response Status at last f/up 3 IDH1 mut Skin relapse Stable skin lesion NR PD (3 months) HiDAC chemo 4 MDS AML 30% blasts 30% NR PD (4 weeks) Hospice (4 mo ) 1* FLT3 - ITD 4 bone lesions All resolved CR Relapse (13 mo ) 7+3 chemo 8 Induc . failure 50% blasts 15% PR PR (4 mo ) 2 nd donor SCT (6 mo ) 9 Del 17p 30% blasts 30% NR SD (2 mo ) Chemo/ venetoclax Same donor SCT (6 mo ) 9* Del 17p 30% blasts N/E N/E PD (3 weeks) Ara - C chemo 17 FLT3 - ITD 70% blasts 45% NR SD (2 mo ) chemo/ venetoclax
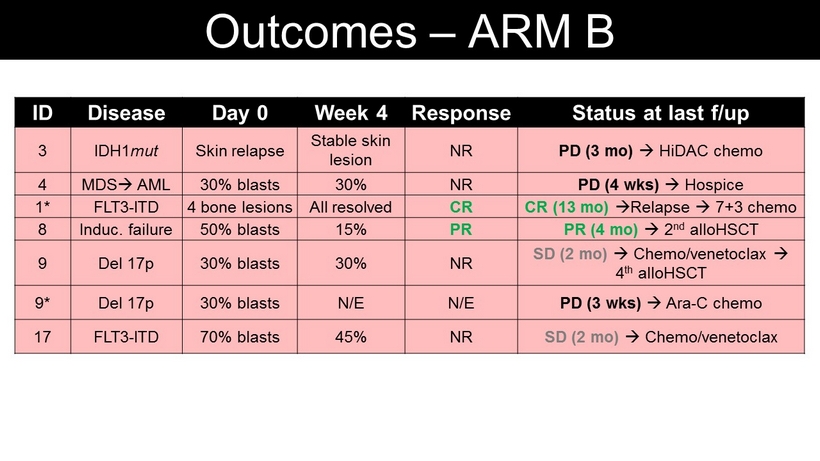
ID Disease Day 0 Week 4 Response Status at last f/up 3 IDH1 mut Skin relapse Stable skin lesion NR PD (3 mo ) HiDAC chemo 4 MDS AML 30% blasts 30% NR PD (4 wks ) Hospice 1* FLT3 - ITD 4 bone lesions All resolved CR CR (13 mo ) Relapse 7+3 chemo 8 Induc . failure 50% blasts 15% PR PR (4 mo ) 2 nd alloHSCT 9 Del 17p 30% blasts 30% NR SD (2 mo ) Chemo/ venetoclax 4 th alloHSCT 9* Del 17p 30% blasts N/E N/E PD (3 wks ) Ara - C chemo 17 FLT3 - ITD 70% blasts 45% NR SD (2 mo ) Chemo/ venetoclax Outcomes – ARM B

Tracking infused clones in vivo Rationale: • Infused lymphocytes are not gene modified • Leukemia specific T cell clones enriched in infused line Approach: • TCR Deep sequencing of donor - T cell product • Track expansion of line - derived clones in vivo

In vivo expansion of line clones 0.5 1 1.5 2 By week 4 Overall Fold change (from baseline) 382 line - exclusive clones 0% 20% 40% 60% Marrow n=4 Week 4 n=12 Productive Frequency 0.5% line - exclusive clones
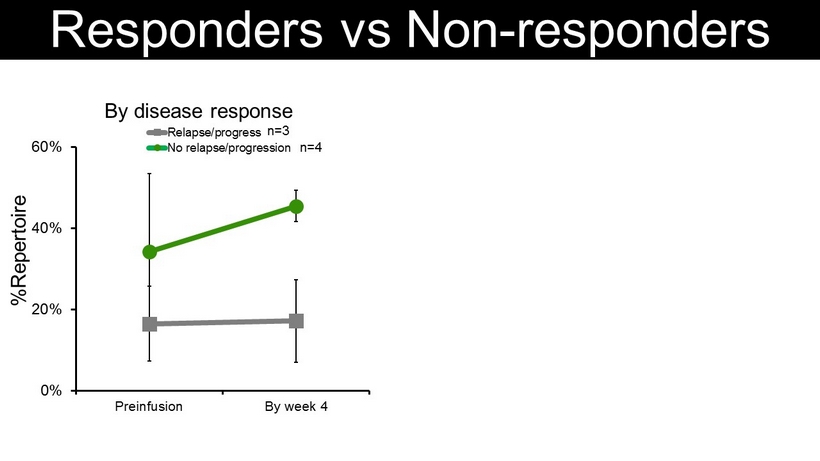
0% 20% 40% 60% Preinfusion By week 4 By disease response Relapse/progress No relapse/progression Responders vs Non - responders 0 40 80 120 Pre Wk4 Responders Non - responders IFN γ ELISPOT %Repertoire n=3 n=4 n=9 n=7 SFC/5x10 5
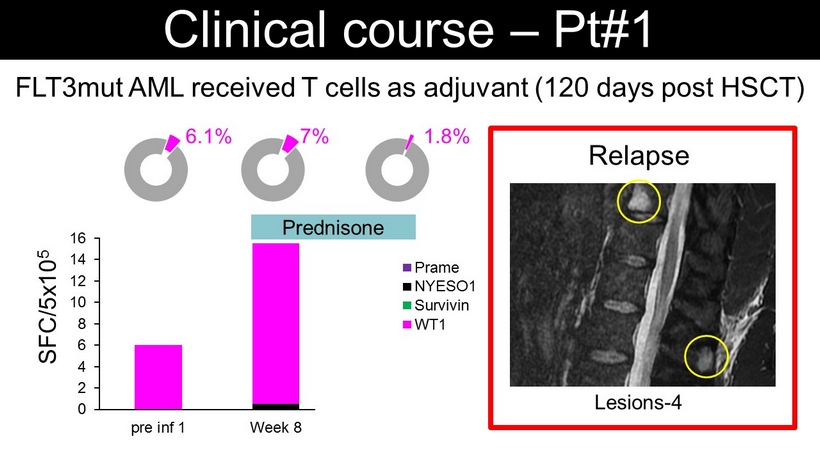
Relapse Lesions - 4 FLT3mut AML received T cells as adjuvant (120 days post HSCT) Clinical course – Pt#1 SFC/5x10 5 0 2 4 6 8 10 12 14 16 pre inf 1 Week 8 Mo5 Prame NYESO1 Survivin WT1 6.1% 7% 1.8% Prednisone

Tumor – antigen expression WT1 H+E Clinical course – Pt#1

Post - T cell (Mo.11) Clinical course – Pt#1 0 5 10 15 20 Post decitabine Post - decitabine (Mo.10) 0 80 160 240 CR NYESO - 1 PRAME WT1 Survivin 0 80 160 240 Marrow • CASSSGQAYEQYF • CASSQVFPNTGELFF 1.45% 2.47% 3.07%
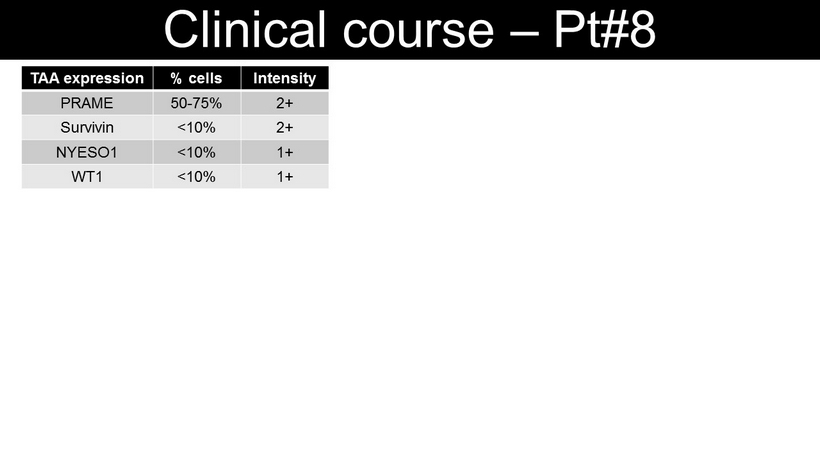
Clinical course – Pt#8 TAA expression % cells Intensity PRAME 50 - 75% 2+ Survivin <10% 2+ NYESO1 <10% 1+ WT1 <10% 1+ ANC SFC/5x10 5 Blasts%

Summary course - SM • Leukemia - directed donor T cell infusions are safe • Mediate anti - tumor effects • In vivo expansion superior in responders • Antigen spreading studies ongoing • Investigation of immune escape mechanisms

Administering AML - directed DLIs to patients with AML or MDS Post - Allogeneic HSCT Relapse Premal Lulla, Swati Naik, Ifigeneia Tzannou, Shivani Mukhi, Manik Kuvalekar, Catherine Robertson, Carlos A Ramos, George Carrum, Rammurti Kamble, Jasleen Randhawa, Adrian P Gee, Bambi Grilley Malcolm K Brenner, Helen E Heslop, Juan F Vera and Ann M Leen Funding : Evans MDS discovery research grant, Leukemia Texas, Leukemia and Lymphoma SCOR, Lymphoma SPORE, ASBMT New Investigator Award, ASH Scholar Award, BCM Junior Faculty Seed Funding Award, EPCRS - DLDCC, LLS/Rising Tide, ARC - Coalition