Exhibit 99.4

Adoptive T cell therapy for ALL targeting multiple tumor associated antigens Swati Naik, Premal Lulla, Ifigeneia Tzannou, Shivani Mukhi, Manik Kuvalekar, Catherine Robertson, George Carrum , Rammurti Kamble , Adrian P Gee, Bambi Grilley, Robert Krance , Malcolm K Brenner, Helen E Heslop, Juan F Vera, Stephen Gottschalk and Ann M Leen

Fagioli Hematologica 2013 Porter et al , BBMT 2011 Arellano, BBMT 2006 ALL Relapse after HSCT • Leukemic relapse is major cause of treatment failure after HSCT – Incidence of relapse: 24 - 35% • Poor prognosis for pts who relapse – Particularly those who relapse early post - HSCT – Overall survival: 7 - 32%

• Strategies to prevent relapse – Prophylactic use of targeted agents (e.g. TKIs) – Modulation of immune suppression • Promote immune reconstitution resulting in GvL effect – Immunotherapeutic intervention with DLIs • Enhance GvL effect Wayne, Hematology 2017 De Lima,BBMT 2013 Alyea et al, BBMT, 2010 Prevention of ALL relapse
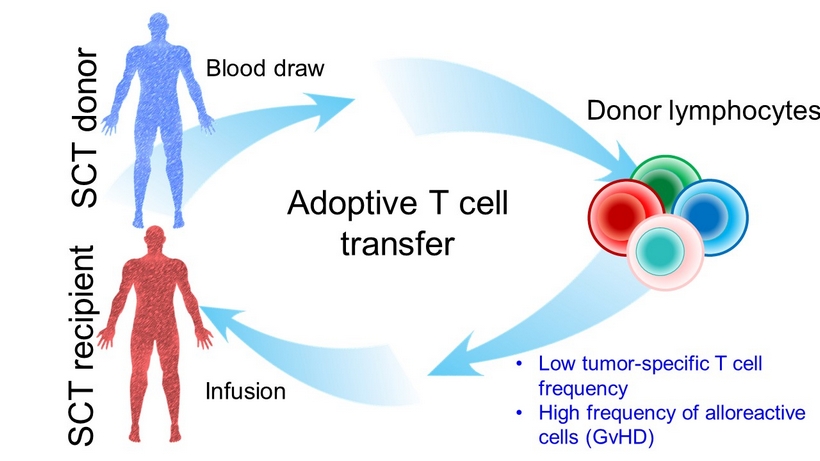
Infusion SCT recipient Blood draw Donor lymphocytes Adoptive T cell transfer SCT donor • Low tumor - specific T cell frequency • High frequency of alloreactive cells (GvHD)
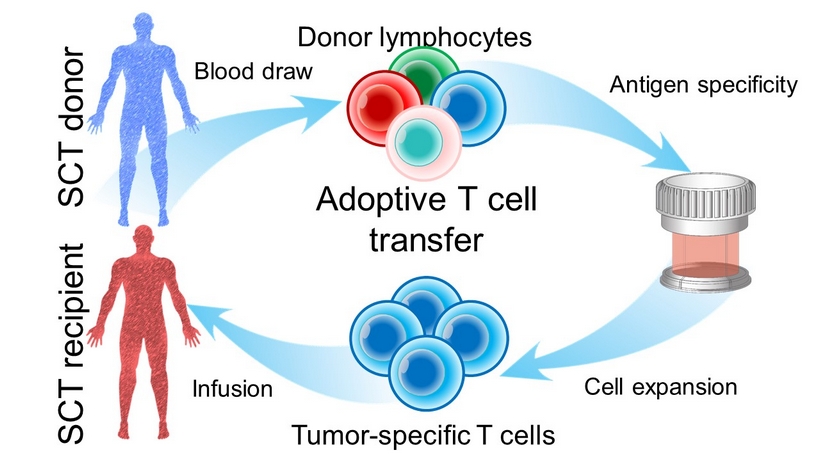
Infusion SCT recipient Cell expansion Tumor - specific T cells Antigen specificity Blood draw Donor lymphocytes Adoptive T cell transfer SCT donor
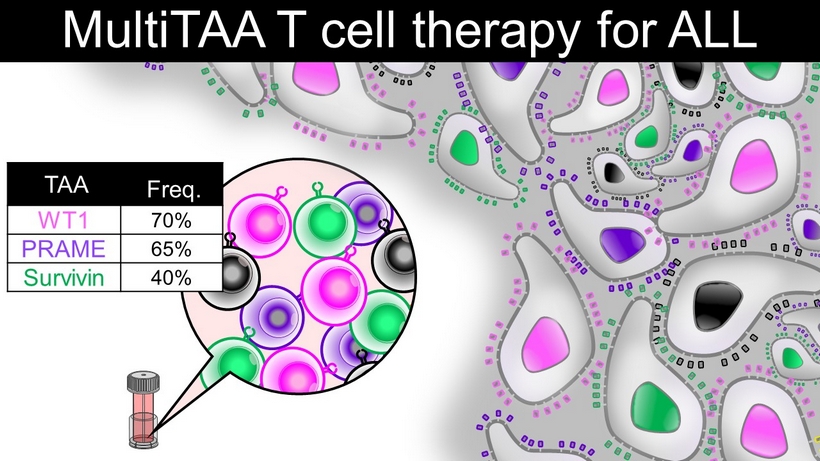
MultiTAA T cell therapy for ALL TAA Freq. WT1 70% PRAME 65% Survivin 40%

DC Pepmix spanning full length WT1, PRAME, Survivin PBMCs MultiTAA T cells Activation Expansion MultiTAA - T Cell manufacture

% Positive cells MultiTAA T cell profile CD3 CD4 CD8 CD3+/ RO+/ 62L+ CD3+/ RO+/ 62L - Phenotype n=11 -10% 0% 10% 20% 30% 40% 20:1 % Specific lysis Safety
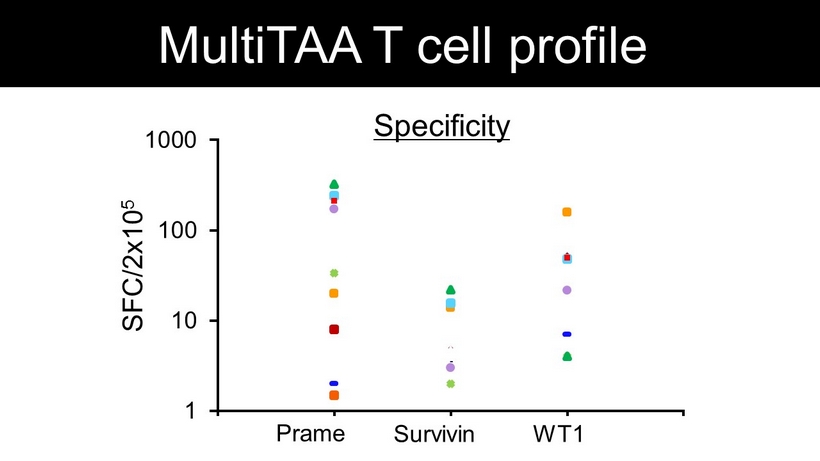
1 10 100 1000 0 1 2 3 4 Specificity Prame Survivin WT1 SFC/2x10 5 MultiTAA T cell profile

DL1 5x10 6 cells/m 2 DL2 1x10 7 cells/m 2 DL3 2x10 7 cells/m 2 Any patient with ALL who received an allogeneic SCT from a family donor Given after day +30 post - transplant Study design (STELLA)
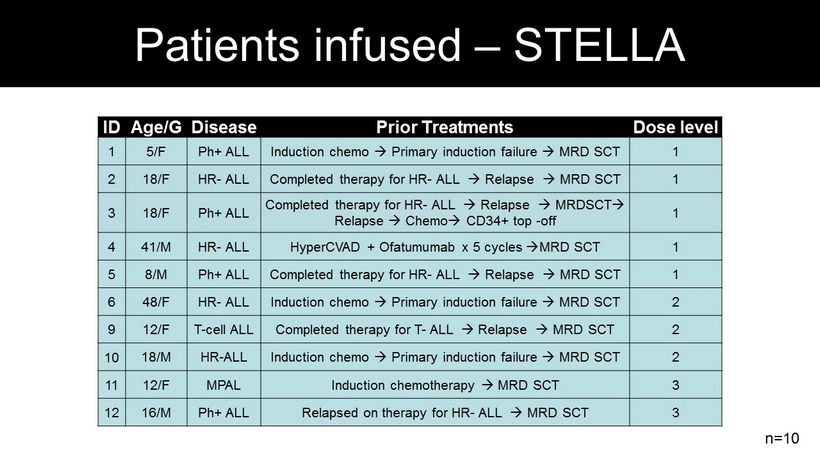
ID Age/G Disease Prior Treatments Dose level 1 5/F Ph + ALL Induction chemo Primary induction failure MRD SCT 1 2 18/F HR - ALL Completed therapy for HR - ALL Relapse MRD SCT 1 3 18/F Ph + ALL Completed therapy for HR - ALL Relapse MRDSCT Relapse Chemo CD34+ top - off 1 4 41/M HR - ALL HyperCVAD + Ofatumumab x 5 cycles MRD SCT 1 5 8/M Ph + ALL Completed therapy for HR - ALL Relapse MRD SCT 1 6 48/F HR - ALL Induction chemo Primary induction failure MRD SCT 2 9 12/F T - cell ALL Completed therapy for T - ALL Relapse MRD SCT 2 10 18/M HR - ALL Induction chemo Primary induction failure MRD SCT 2 11 12/F MPAL Induction chemotherapy MRD SCT 3 12 16/M Ph + ALL Relapsed on therapy for HR - ALL MRD SCT 3 n=10 Patients infused – STELLA

• No Dose Limiting Toxicities (DLTs) • No GVHD • No CRS/neurotoxicity or other adverse events • Three patients not evaluable per protocol: – received >0.5mg/kg steroids within 4 weeks of infusion – Pt 1 and 12: Stress dose steroids for sepsis – Pt 6: Steroids for elevation of AST/ALT, GVHD ruled out Safety
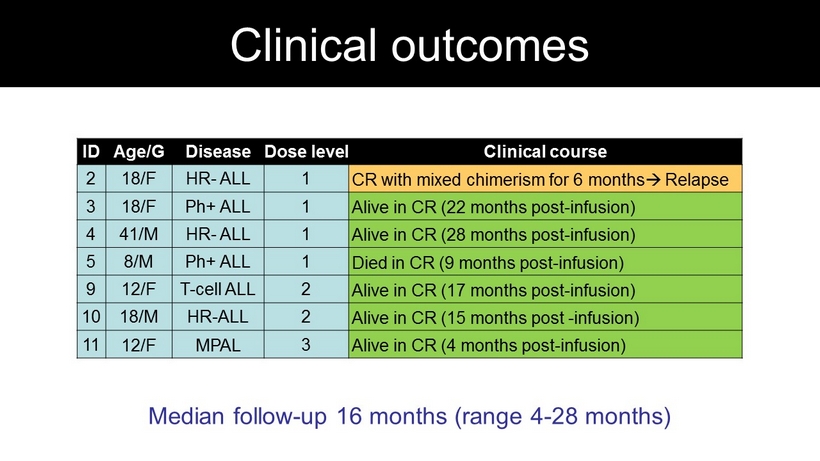
ID Age/G Disease Dose level Clinical course 2 18/F HR - ALL 1 CR with mixed chimerism for 6 months Relapse 3 18/F Ph + ALL 1 Alive in CR ( 22 months post - infusion) 4 41/M HR - ALL 1 Alive in CR ( 28 months post - infusion) 5 8/M Ph + ALL 1 Died in CR ( 9 months post - infusion) 9 12/F T - cell ALL 2 Alive in CR ( 17 months post - infusion) 10 18/M HR - ALL 2 Alive in CR ( 15 months post - infusion) 11 12/F MPAL 3 Alive in CR ( 4 months post - infusion) Median follow - up 16 months (range 4 - 28 months) Clinical outcomes
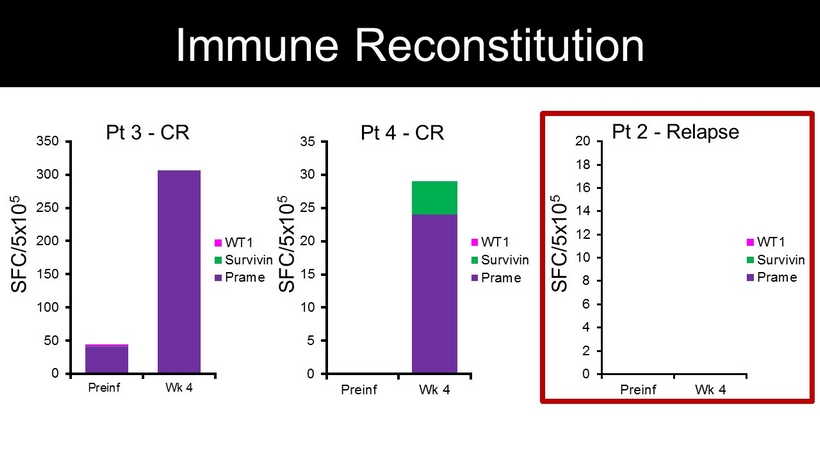
SFC/5x10 5 0 50 100 150 200 250 300 350 Preinf Wk 4 WT1 Survivin Prame Pt 3 - CR 0 5 10 15 20 25 30 35 Preinf Wk 4 WT1 Survivin Prame Pt 4 - CR SFC/5x10 5 0 2 4 6 8 10 12 14 16 18 20 Preinf Wk 4 WT1 Survivin Prame Pt 2 - Relapse SFC/5x10 5 Immune Reconstitution
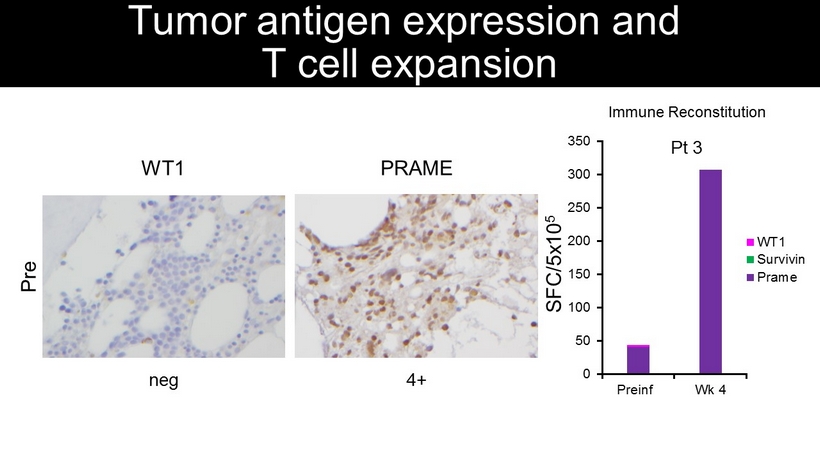
PRAME 4+ WT1 Pre neg Tumor antigen expression and T cell expansion SFC/5x10 5 0 50 100 150 200 250 300 350 Preinf Wk 4 WT1 Survivin Prame Pt 3 Immune Reconstitution
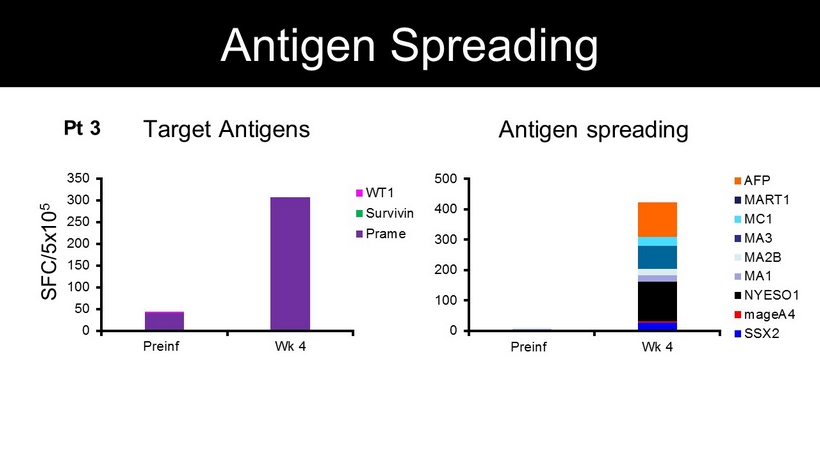
SFC/5x10 5 Target Antigens Antigen spreading 0 50 100 150 200 250 300 350 Preinf Wk 4 WT1 Survivin Prame 0 100 200 300 400 500 Preinf Wk 4 AFP MART1 MC1 MA3 MA2B MA1 NYESO1 mageA4 SSX2 Pt 3 Antigen Spreading

• Feasible for both B - cell and T - cell ALL • Safe to date, well - tolerated • In vivo expansion of tumor - antigen associated T - cells directed to target antigens • Evidence of antigen spreading which may contribute to relapse prevention • May present a safe and effective strategy to prevent leukemic relapse post - HSCT Summary