Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
INSM similar filings
- 9 May 24 Insmed Reports First-Quarter 2024 Financial Results and Provides Business Update
- 11 Mar 24 Other Events
- 22 Feb 24 Insmed Reports Fourth-Quarter and Full-Year 2023 Financial Results and Provides Business Update
- 5 Jan 24 Insmed Provides Business Update at 42nd Annual J.P. Morgan Healthcare Conference
- 26 Oct 23 Insmed Reports Third-Quarter 2023 Financial Results and Provides Business Update
- 5 Sep 23 Regulation FD Disclosure
- 3 Aug 23 Insmed Reports Second-Quarter 2023 Financial Results and Provides Business Update
Filing view
External links
Exhibit 99.2

J.P. Morgan Healthcare Conference January 2024

Forward Looking Statement Forward-Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties. "Forward-looking statements," as that term is defined in the Private Securities Litigation Reform Act of 1995, are statements that are not historical facts and involve a number of risks and uncertainties. Words herein such as "may," "will," "should," "could," "would," "expects," "plans," "anticipates," "believes," "estimates," "projects," "predicts," "intends," "potential," "continues," and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) may identify forward-looking statements. The forward-looking statements in this presentation are based upon the Company's current expectations and beliefs, and involve known and unknown risks, uncertainties and other factors, which may cause the Company's actual results, performance and achievements and the timing of certain events to differ materially from the results, performance, achievements or timings discussed, projected, anticipated or indicated in any forward-looking statements. Such risks, uncertainties and other factors include, among others, the following: failure to obtain, or delays in obtaining, regulatory approvals for ARIKAYCE outside the United States (U.S.), Europe or Japan, or for the Company's product candidates in the U.S., Europe, Japan or other markets, including separate regulatory approval for the Lamira® Nebulizer System in each market and for each usage; failure to successfully commercialize ARIKAYCE, the Company's only approved product, in the U.S., Europe or Japan (amikacin liposome inhalation suspension, Liposomal 590 mg Nebuliser Dispersion, and amikacin sulfate inhalation drug product, respectively), or to maintain U.S., European or Japanese approval for ARIKAYCE, or failure to successfully commercialize any of the Company's product candidates in the future; business or economic disruptions due to catastrophes or other events, including natural disasters or public health crises; impact of the COVID-19 pandemic and efforts to reduce its spread on the Company's business, employees, including key personnel, patients, partners and suppliers; risk that brensocatib or TPIP does not prove to be effective or safe for patients in ongoing and future clinical studies, including, for brensocatib, the ASPEN study; uncertainties in the degree of market acceptance of ARIKAYCE by physicians, patients, third-party payors and others in the healthcare community; the Company's inability to obtain full approval of ARIKAYCE from the U.S. Food and Drug Administration, including the risk that the Company will not successfully or in a timely manner validate a PRO tool and complete the confirmatory post-marketing clinical trial required for full approval of ARIKAYCE; inability of the Company, PARI or the Company's other third-party manufacturers to comply with regulatory requirements related to ARIKAYCE or the Lamira® Nebulizer System; the Company's inability to obtain adequate reimbursement from government or third-party payors for ARIKAYCE or acceptable prices for ARIKAYCE or for the Company's other product candidates; development of unexpected safety or efficacy concerns related to ARIKAYCE, brensocatib, Treprostinil Palmitil Inhalation Powder (TPIP) or the Company's other product candidates; inaccuracies in the Company's estimates of the size of the potential markets for ARIKAYCE, brensocatib, TPIP or the Company's other product candidates or in data the Company has used to identify physicians, expected rates of patient uptake, duration of expected treatment, or expected patient adherence or discontinuation rates; the risks and uncertainties associated with, and the perceived benefits of, the Company's secured senior loan with certain funds managed by Pharmakon Advisors, LP and the Company's royalty financing with OrbiMed Royalty & Credit Opportunities IV, LP, including the Company's ability to maintain compliance with the covenants in the agreements for the senior secured loan and royalty financing and the perceived impact of the restrictions on the Company's operations under these agreements; the Company's inability to create an effective direct sales and marketing infrastructure or to partner with third parties that offer such an infrastructure for distribution of ARIKAYCE or any of the Company's product candidates that are approved in the future; failure to obtain regulatory approval to expand ARIKAYCE's indication to a broader patient population; risk that the Company's competitors may obtain orphan drug exclusivity for a product that is essentially the same as a product the Company is developing for a particular indication; failure to successfully predict the time and cost of development, regulatory approval and commercialization for novel gene therapy products; failure to successfully conduct future clinical trials for ARIKAYCE, brensocatib, TPIP and the Company's other product candidates due to the Company's limited experience in conducting preclinical development activities and clinical trials necessary for regulatory approval and its potential inability to enroll or retain sufficient patients to conduct and complete the trials or generate data necessary for regulatory approval, among other things; risks that the Company's clinical studies will be delayed, that serious side effects will be identified during drug development, or that any protocol amendments submitted will be rejected; risks that interim or partial data sets are not representative of a complete or larger data set or that blinded data will not be predictive of unblinded data; failure of third parties on which the Company is dependent to manufacture sufficient quantities of ARIKAYCE or the Company's product candidates for commercial or clinical needs, to conduct the Company's clinical trials, or to comply with the Company's agreements or laws and regulations that impact the Company's business or agreements with the Company; the Company's inability to attract and retain key personnel or to effectively manage the Company's growth; the Company's inability to successfully integrate its recent acquisitions and appropriately manage the amount of management's time and attention devoted to integration activities; risks that the Company's acquired technologies, products and product candidates are not commercially successful; the Company's inability to adapt to its highly competitive and changing environment; risk that the Company is unable to maintain its significant customers; risk that government healthcare reform materially increases the Company's costs and damages its financial condition; deterioration in general economic conditions in the U.S., Europe, Japan and globally, including the effect of prolonged periods of inflation, affecting the Company, its suppliers, third-party service providers and potential partners; the Company's inability to adequately protect its intellectual property rights or prevent disclosure of its trade secrets and other proprietary information and costs associated with litigation or other proceedings related to such matters; restrictions or other obligations imposed on the Company by agreements related to ARIKAYCE or the Company's product candidates, including its license agreements with PARI and AstraZeneca AB, and failure of the Company to comply with its obligations under such agreements; the cost and potential reputational damage resulting from litigation to which the Company is or may become a party, including product liability claims; risk that the Company's operations are subject to a material disruption in the event of a cybersecurity attack or issue; the Company's limited experience operating internationally; changes in laws and regulations applicable to the Company's business, including any pricing reform, and failure to comply with such laws and regulations; the Company's history of operating losses, and the possibility that the Company may never achieve or maintain profitability; goodwill impairment charges affecting the Company's results of operations and financial condition; inability to repay the Company's existing indebtedness and uncertainties with respect to the Company's ability to access future capital; and delays in the execution of plans to build out an additional third-party manufacturing facility approved by the appropriate regulatory authorities and unexpected expenses associated with those plans. The Company may not actually achieve the results, plans, intentions or expectations indicated by the Company's forward-looking statements because, by their nature, forward-looking statements involve risks and uncertainties because they relate to events and depend on circumstances that may or may not occur in the future. For additional information about the risks and uncertainties that may affect the Company's business, please see the factors discussed in Item 1A, "Risk Factors," in the Company's Annual Report on Form 10-K for the year ended December 31, 2022 and any subsequent Company filings with the Securities and Exchange Commission (SEC). The Company cautions readers not to place undue reliance on any such forward-looking statements, which speak only as of the date of this presentation. The Company disclaims any obligation, except as specifically required by law and the rules of the SEC, to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements.

Brensocatib ASPEN Readout in Bronchiectasis First Phase 2 TPIP Readout in PH-ILD Advance ARIKAYCE® Toward Label Expansion Continued Progress on Cutting-Edge Early Pipeline The Stage is Set for a Breakout Year Rarely Seen in Biotech 2024: The Year of Insmed’s Transformation
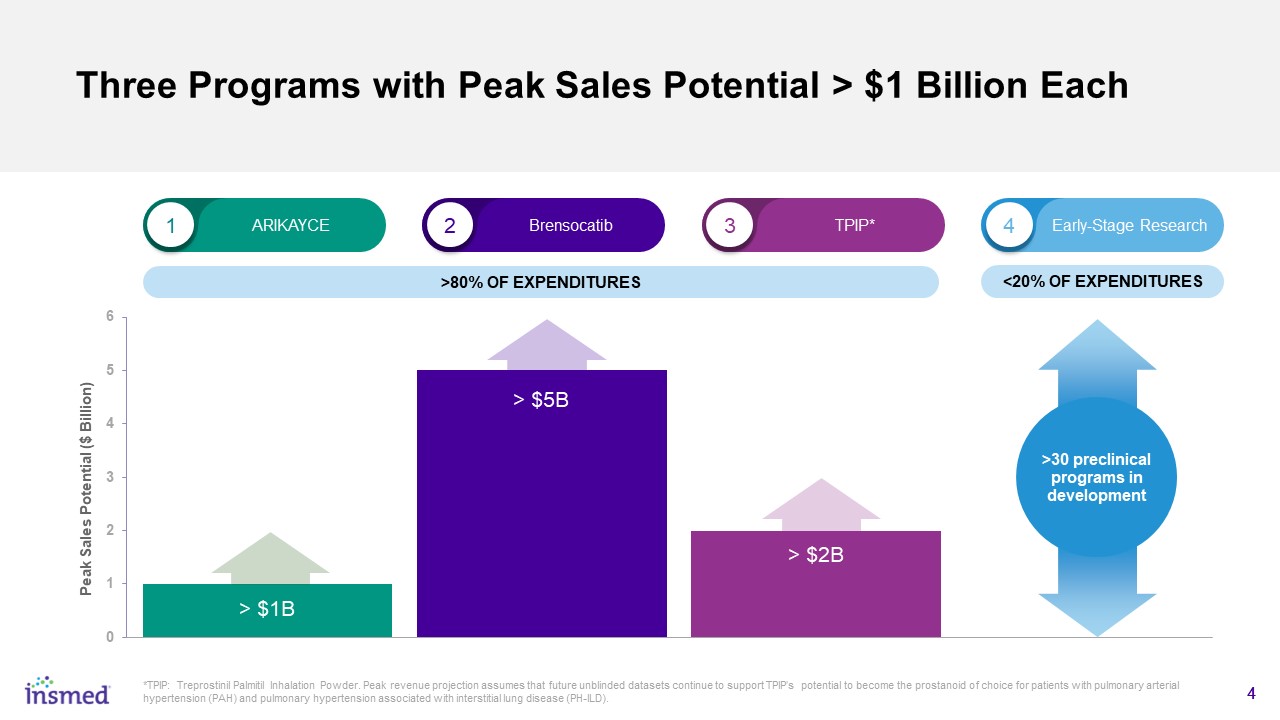
Three Programs with Peak Sales Potential > $1 Billion Each 1 2 3 4 ARIKAYCE Brensocatib TPIP* Early-Stage Research <20% OF EXPENDITURES >80% OF EXPENDITURES *TPIP: Treprostinil Palmitil Inhalation Powder. Peak revenue projection assumes that future unblinded datasets continue to support TPIP’s potential to become the prostanoid of choice for patients with pulmonary arterial hypertension (PAH) and pulmonary hypertension associated with interstitial lung disease (PH-ILD). 4 >30 preclinical programs in development

ARIKAYCE Expected to Continue to Deliver Strong Double-Digit Revenue Growth Globally in 2024 ARIKAYCE Full-Year 2024 Revenue Guidance ($340M - $360M) * Slower Growth Period During COVID-19 Pandemic Growth Reacceleration 2024 Guidance Represents ~15% YoY Growth at the Midpoint ($20-$30M sales growth/year) Launch (Q4 2018) First Full Year of Revenues *Revenues for 2023 are unaudited.

ARIKAYCE Expected to be a >$1 Billion Peak Sales Product Assuming Label Expansion to Include All Patients with MAC Lung Disease Newly diagnosed NTM MAC could expand the ARIKAYCE commercial opportunity by 3-5 times ARIKAYCE Refractory MAC Lung Patients ~1,400* EU** *Source: Internal analysis of published NTM epidemiology, primary market research with treating HCPs, and anonymized patient level claims data in US. **EU: European 5 comprised of France, Germany, Italy, Spain and the United Kingdom. Refractory MAC Lung Patients 12-17K* Total MAC Lung Patients 95-115K* U.S. Japan Refractory MAC Lung Patients 15-18K* Total MAC Lung Patients 125-145K* Total MAC Lung Patients 14K* 3-5x 3-5x 3-5x

Brensocatib: ASPEN on Track to Read Out in 2Q24 Need One Dose to Win for Regulatory and Commercial Success Clear Win 5th and Final DSMC4 Meeting Held in November 2023 ONE DOSE (either 10mg or 25mg) achieves a p-value1 < 0.01 Anticipated commercial launch timing: Mid-20252 Brensocatib Can File3 ONE DOSE (either 10mg or 25mg) achieves a p-value1 < 0.05 1Refers to the p-value for the primary endpoint, adjusted for multiplicity. 2Timing assumes one dose (either 10mg or 25mg) achieves a p-value <0.01. Brensocatib has been granted Breakthrough Therapy Designation in the US and PRIME designation in the EU. 3FDA indicated that this data result could be filed for review. 4DSMC: Data Safety Monitoring Committee.

If ASPEN Succeeds, Brensocatib Expected to Become a $5+ Billion Peak Sales Product Brensocatib Timeline for Next Steps BiRCh P2 Trial Readout (CRSsNP) ASPEN P3 Trial Readout (Bronchiectasis) P2 Trial Initiation (HS) Launch (Bronchiectasis) 1Assumes indication for non-cystic fibrosis bronchiectasis and approval in US, European 5, and Japan. 2Weycker, et al. Prevalence and incidence of NCFBE among US adults in 2013. Chronic Respiratory Disease. 2017. 3Insmed: Patient Level Claims Data Analysis and Internal Market Research. 3Ex-US estimates based on published epidemiology research, Insmed market research, and extrapolation of US-focused claims and epi data analysis (sourced from swoop/ipm.ai). 4Cho et. al, Chronic Rhinosinusitis without Nasal Polyps J Allergy Clin Immunol Pract. 2016 ; 4(4): 575–582. doi:10.1016/j.jaip.2016.04.015. 5Benjamin et. al, Clinical Characteristics of Patients with Chronic Rhinosinusitis without Nasal Polyps in an Academic Setting, J ALLERGY CLIN IMMUNOL PRACT VOLUME 7, NUMBER 3, MARCH 2019. 6Patient level claims data analysis US ONLY (Komodo Health), proportion of actively managed CRS patients with no Dx codes for Nasal Polyps in patient history; Extrapolated to European 5 and Japan. 7Patient level claims data analysis US ONLY (Komodo Health), proportion of actively managed CRSsNP patients with ESS; Extrapolated to European 5 and Japan. 8Phan et al, Global prevalence of hidradenitis suppurativa and geographical variation—systematic review and meta-analysis Biomedical Dermatology (2020) 4:2. 9Puri, Ajay: Hidradenitis Suppurativa Executive Insights, DRG Nov 2019. Total Addressable Market (global) Current Treatment Landscape Bronchiectasis1,2,3 ~1M No approved treatments Chronic Rhinosinusitis w/o Nasal Polyps (CRSsNP)4,5,6,7 ~400K Hidradenitis Suppurativa (HS)8,9 ~250K Treatment includes multiple therapies and/or surgery 2024 2025

TPIP TPIP: Multiple Clinical Readouts Expected in 2024 and 2025 PAH 45 patients enrolled in Phase 2 study (YE 2023) ~47% average PVR reduction among responders* Phase 2 topline data in 2025 Doubling dose ceiling to 1,280μg from 640μg PH-ILD Phase 2 study fully enrolled (Nov. 2023) Topline readout expected pre-ASPEN (Q2 2024) *Based on blinded data shared on 10/26/2023. KOL: Key Opinion Leader. PVR: Pulmonary Vascular Resistance. PAH: Pulmonary arterial hypertension. PH-ILD: Pulmonary hypertension associated with interstitial lung disease. “The hemodynamic changes are stunning…” KOL comment on the blinded results shared in Oct. 2023 >80% of all study participants reached the maximum dose in just 5 weeks*

TPIP: Upcoming Phase 2 Data in PH-ILD Primarily Meant to Characterize Safety Profile Favorable tolerability profile in patients with PH-ILD Highest dose (640μg) achieved in majority of patients Treatment-emergent adverse events consistent with underlying disease Pharmacokinetic exploratory endpoints are consistent with preclinical data (not to be included with topline results) TPIP The PH-ILD Phase 2 is a SAFETY study and is not powered to show statistical differences Clear Win
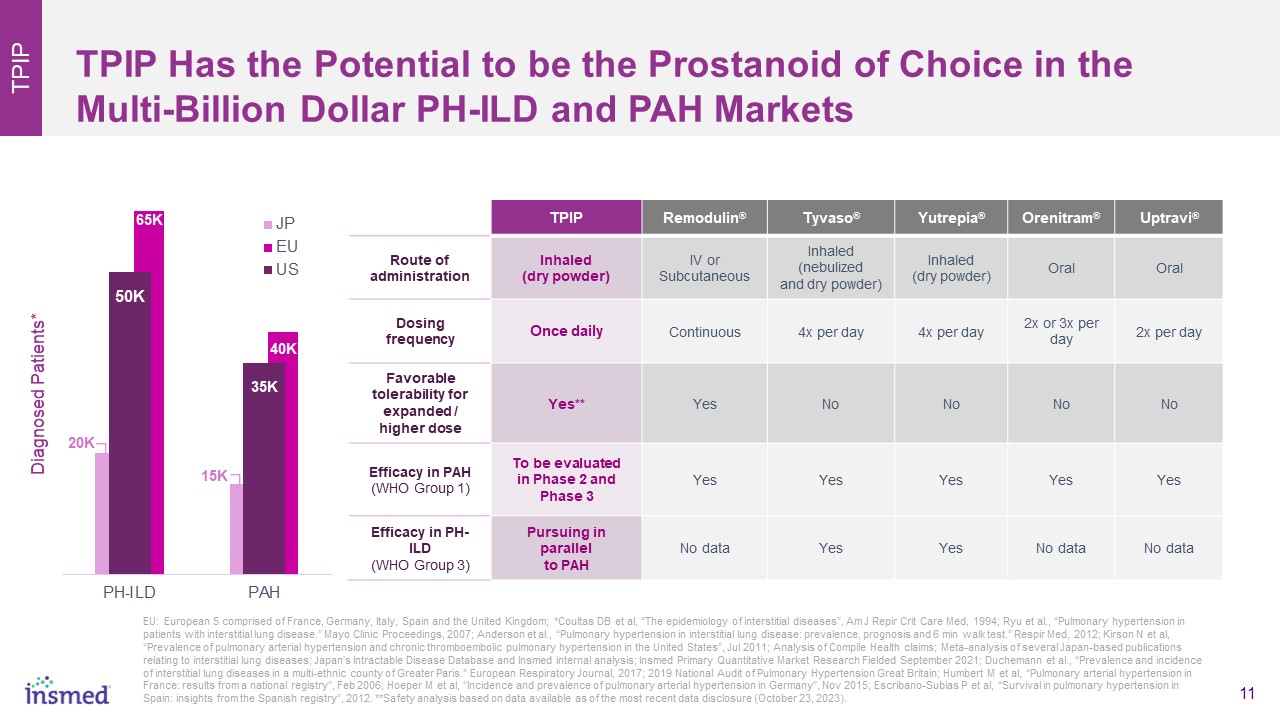
TPIP Has the Potential to be the Prostanoid of Choice in the Multi-Billion Dollar PH-ILD and PAH Markets TPIP TPIP Remodulin® Tyvaso® Yutrepia® Orenitram® Uptravi® Route of administration Inhaled (dry powder) IV orSubcutaneous Inhaled (nebulized and dry powder) Inhaled (dry powder) Oral Oral Dosing frequency Once daily Continuous 4x per day 4x per day 2x or 3x per day 2x per day Favorable tolerability for expanded / higher dose Yes** Yes No No No No Efficacy in PAH (WHO Group 1) To be evaluated in Phase 2 and Phase 3 Yes Yes Yes Yes Yes Efficacy in PH-ILD (WHO Group 3) Pursuing in parallel to PAH No data Yes Yes No data No data EU: European 5 comprised of France, Germany, Italy, Spain and the United Kingdom; *Coultas DB et al, “The epidemiology of interstitial diseases”, Am J Repir Crit Care Med, 1994; Ryu et al., “Pulmonary hypertension in patients with interstitial lung disease.” Mayo Clinic Proceedings, 2007; Anderson et al., “Pulmonary hypertension in interstitial lung disease: prevalence, prognosis and 6 min walk test.” Respir Med, 2012; Kirson N et al, “Prevalence of pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension in the United States”, Jul 2011; Analysis of Compile Health claims; Meta-analysis of several Japan-based publications relating to interstitial lung diseases; Japan’s Intractable Disease Database and Insmed internal analysis; Insmed Primary Quantitative Market Research Fielded September 2021; Duchemann et al., “Prevalence and incidence of interstitial lung diseases in a multi-ethnic county of Greater Paris.” European Respiratory Journal, 2017; 2019 National Audit of Pulmonary Hypertension Great Britain; Humbert M et al, “Pulmonary arterial hypertension in France: results from a national registry”, Feb 2006; Hoeper M et al, “Incidence and prevalence of pulmonary arterial hypertension in Germany”, Nov 2015; Escribano-Subias P et al, “Survival in pulmonary hypertension in Spain: insights from the Spanish registry”, 2012. **Safety analysis based on data available as of the most recent data disclosure (October 23, 2023). 65K

LOW UPFRONT COSTS to acquire the technologies HIGH POTENTIAL IMPACT >30 pre-clinical programs LOW ONGOING EXPENSE <20% of expenditures Early-Stage Research: An Assembly of Complementary Platform Technologies to Answer the Question: “What’s Next?” MULTIPLE CUTTING-EDGE TECHNOLOGIES UNDERPIN INSMED’S EARLY-STAGE PIPELINE Deimmunized Protein Engineering Using AI RNA End-Joining Algae-Based Protein Manufacturing Targeting diseases caused by larger genes Lower cost, higher yield; Potential for licensing Synthetic Rescue Gene Therapy Using Intrathecal Delivery Increased safety with decreased viral load Re-design / re-dose immunogenic drugs Addressing previously untreatable diseases Early Research
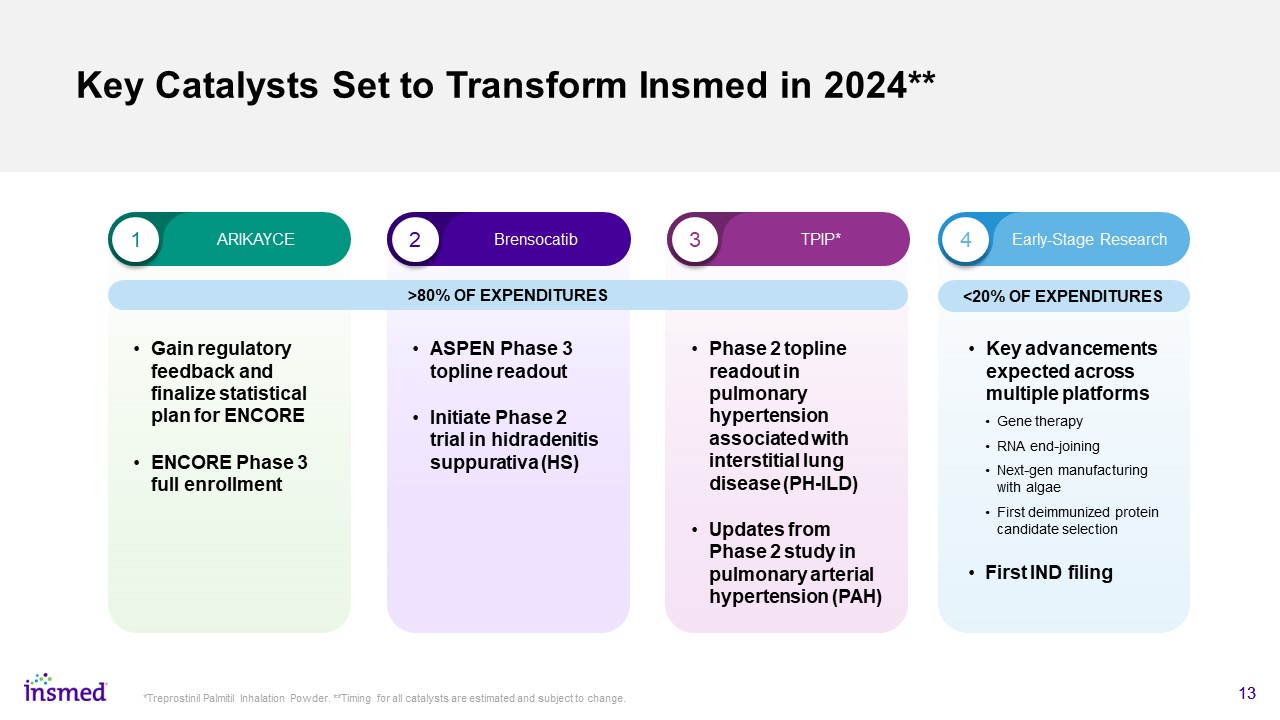
Key Catalysts Set to Transform Insmed in 2024** Gain regulatory feedback and finalize statistical plan for ENCORE ENCORE Phase 3 full enrollment 13 *Treprostinil Palmitil Inhalation Powder. **Timing for all catalysts are estimated and subject to change. 1 2 3 4 ARIKAYCE Brensocatib TPIP* Early-Stage Research <20% OF EXPENDITURES >80% OF EXPENDITURES ASPEN Phase 3 topline readout Initiate Phase 2 trial in hidradenitis suppurativa (HS) Phase 2 topline readout in pulmonary hypertension associated with interstitial lung disease (PH-ILD) Updates from Phase 2 study in pulmonary arterial hypertension (PAH) Key advancements expected across multiple platforms Gene therapy RNA end-joining Next-gen manufacturing with algae First deimmunized protein candidate selection First IND filing
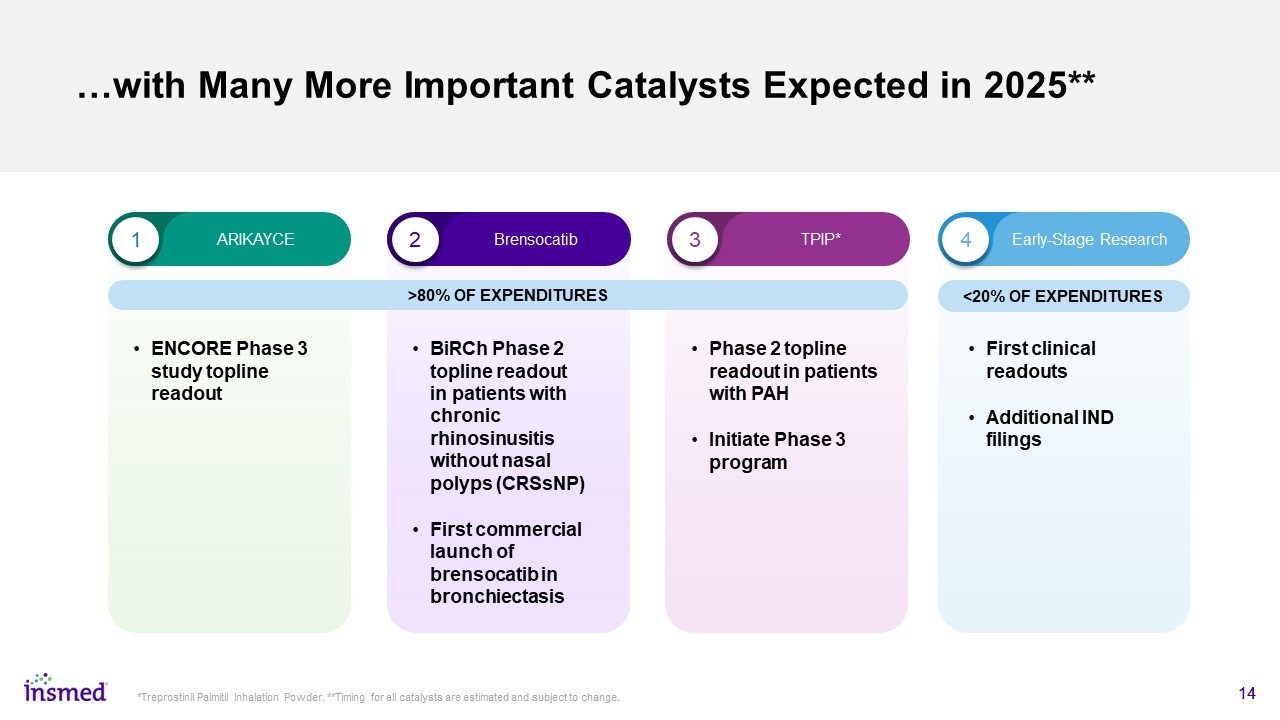
…with Many More Important Catalysts Expected in 2025** 14 *Treprostinil Palmitil Inhalation Powder. **Timing for all catalysts are estimated and subject to change. ENCORE Phase 3 study topline readout 1 2 3 4 ARIKAYCE Brensocatib TPIP* Early-Stage Research <20% OF EXPENDITURES >80% OF EXPENDITURES BiRCh Phase 2 topline readout in patients with chronic rhinosinusitis without nasal polyps (CRSsNP) First commercial launch of brensocatib in bronchiectasis Phase 2 topline readout in patients with PAH Initiate Phase 3 program First clinical readouts Additional IND filings

Shareholder Value Will Be Key Consideration Current Cash Position Offers Insmed Flexibility on the Timing and Form of Any Future Capital Raise Rich event pipeline in next two years Financing options available under all scenarios Convert or Issue Debt Sell Royalty Equity Raise Monetize TPIP Many Financing Options Exist, Regardless of ASPEN Result

Our Purpose and Values Define our Culture at Insmed Where Employees Are Empowered to do their Best Work on Behalf of Patients in Need “I am proud to work at Insmed.” “I am inspired by the work we do.” “I understand how my job helps Insmed achieve success.” “I believe Insmed will be successful in the future.” In a recent survey* >90% of employees responded that… *The 2023 annual Insmed Pulse Survey included 91% participation across the organization. #1 on Science’s Top Biopharma Employers List (3rd Year in a Row) Certified as a Great Place to Work (3rd Year in a Row)
