Filed Pursuant to Rule 424(b)(5)
Registration No. 333-177793
Prospectus supplement
(to prospectus dated November 16, 2011)
14,300,000 shares

Common stock
We are offering 14,300,000 shares of our common stock.
Shares of our common stock trade on the NASDAQ Capital Market under the symbol “ZIOP.” The last reported sale price on October 23, 2013 was $4.03 per share.
| | | | | | | | |
| | | Per Share | | | Total | |
| | |
Public offering price | | $ | 3.50 | | | $ | 50,050,000 | |
| | |
Underwriting discounts and commissions | | $ | 0.21 | | | $ | 3,003,000 | |
| | |
Proceeds, before expenses, to us | | $ | 3.29 | | | $ | 47,047,000 | |
We have granted the underwriters an option for a period of up to 30 days from the date of this prospectus supplement to purchase up to 2,145,000 of additional shares of common stock at the public offering price less the underwriting discounts and commissions.
INVESTING IN OUR COMMON STOCK INVOLVES RISK. SEE “RISK FACTORS” BEGINNING ON PAGE S-11.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus supplement or the accompanying prospectus. Any representation to the contrary is a criminal offense.
Intrexon Corporation, or Intrexon, which is affiliated with Randal J. Kirk, who serves as one of our directors, has agreed to purchase 2,857,143 shares of our common stock in this offering at the public offering price. The underwriters will receive the same discount from such shares of our common stock as they will from any other shares of our common stock sold to the public in this offering.
The underwriters expect to deliver the shares on or about October 29, 2013.
Sole book-running manager
J.P. Morgan
Co-managers
| | |
| JMP Securities | | Griffin Securities, Inc. |
October 23, 2013
This prospectus contains references to our trademarks and to trademarks belonging to other entities. Solely for convenience, trademarks and trade names referred to in this prospectus, including logos, artwork and other visual displays, may appear without the® or TM symbols, but such references are not intended to indicate, in any way, that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. We do not intend our use or display of other companies’ trade names or trademarks to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
Table of contents
S-i
About this prospectus supplement
This document is in two parts. The first part is this prospectus supplement, which describes the specific terms of this common stock offering and also adds to and updates information contained in the accompanying prospectus and the documents incorporated by reference herein. The second part, the accompanying prospectus, provides more general information. Generally, when we refer to this prospectus, we are referring to both parts of this document combined. To the extent there is a conflict between the information contained in this prospectus supplement and the information contained in the accompanying prospectus or any document incorporated by reference therein filed prior to the date of this prospectus supplement, you should rely on the information in this prospectus supplement; provided that if any statement in one of these documents is inconsistent with a statement in another document having a later date—for example, a document incorporated by reference in the accompanying prospectus—the statement in the document having the later date modifies or supersedes the earlier statement.
We further note that the representations, warranties and covenants made by us in any agreement that is filed as an exhibit to any document that is incorporated by reference herein were made solely for the benefit of the parties to such agreement, including, in some cases, for the purpose of allocating risk among the parties to such agreements, and should not be deemed to be a representation, warranty or covenant to you. Moreover, such representations, warranties or covenants were accurate only as of the date specified in the relevant agreement. Accordingly, such representations, warranties and covenants should not be relied on as accurately representing the current state of our affairs.
You should rely only on the information contained in this prospectus supplement, the accompanying prospectus, or any free writing prospectus authorized by us, or incorporated by reference herein. We have not authorized, and the underwriters have not authorized, anyone to provide you with information that is different. The information contained in this prospectus supplement, the accompanying prospectus, or any free writing prospectus authorized by us, or incorporated by reference herein is accurate only as of the respective dates thereof, regardless of the time of delivery of this prospectus supplement, the accompanying prospectus, and any free writing prospectus authorized by us or of any sale of our common stock. It is important for you to read and consider all information contained in this prospectus supplement, the accompanying prospectus, and any free writing prospectus authorized by us, including the documents incorporated by reference herein and therein, in making your investment decision. You should also read and consider the information in the documents to which we have referred you in the sections entitled “Where you can find more information” and “Incorporation of information by reference” in this prospectus supplement and in the accompanying prospectus.
We are offering to sell, and seeking offers to buy, shares of our common stock only in jurisdictions where offers and sales are permitted. The distribution of this prospectus supplement and the accompanying prospectus and the offering of the common stock in certain jurisdictions may be restricted by law. Persons outside the United States who come into possession of this prospectus supplement and the accompanying prospectus must inform themselves about, and observe any restrictions relating to, the offering of the common stock and the distribution of this prospectus supplement and the accompanying prospectus outside the United States. This prospectus supplement and the accompanying prospectus do not constitute, and may not be used in connection with, an offer to sell, or a solicitation of an offer to buy, any securities offered by this prospectus supplement and the accompanying prospectus by any person in any jurisdiction in which it is unlawful for such person to make such an offer or solicitation.
Unless otherwise stated, all references in this prospectus to “we,” “us,” “our,” “ZIOPHARM,” the “Company” and similar designations refer to ZIOPHARM Oncology, Inc.
S-ii
Prospectus supplement summary
This summary highlights certain information about us, this offering and selected information contained elsewhere in or incorporated by reference in this prospectus supplement. This summary is not complete and does not contain all of the information that you should consider before deciding whether to invest in our common stock. For a more complete understanding of our company and this offering, we encourage you to read and consider carefully the more detailed information in this prospectus supplement and the accompanying prospectus, including the information referred to under the heading “Risk factors” in this prospectus supplement beginning on page S-11, the information incorporated by reference in this prospectus supplement and the accompanying prospectus, and the information included in any free writing prospectus that we have authorized for use in connection with this offering.
Company overview
ZIOPHARM Oncology, Inc. is a biopharmaceutical company that seeks to acquire, develop and commercialize, on its own or with commercial partners, a diverse portfolio of cancer therapies that can address unmet medical needs through synthetic biology. Pursuant to an exclusive channel agreement with Intrexon Corporation, or Intrexon, we obtained rights to Intrexon’s synthetic biology platform for use in the field of oncology, which included two existing clinical stage product candidates, Ad-RTS-IL-12 + Activator Ligand and DC-RTS-IL-12 + Activator Ligand. The synthetic biology platform employs an inducible gene-delivery system that enables controlled delivery of genes that produce therapeutic proteins to treat cancer. Ad-RTS-IL-12 is our lead drug candidate, which uses this gene delivery system to produce Interleukin-12, or IL-12, a potent, naturally occurring anti-cancer protein. We are currently studying Ad-RTS-IL-12 in two Phase 2 studies, the first for the treatment of metastatic melanoma, and the second for the treatment of metastatic breast cancer, and expect to announce early, preliminary data from these Phase 2 studies in the fourth quarter of 2013 and final data in 2014. We plan to continue to combine Intrexon’s synthetic biology platform with our capabilities to translate science to the patient setting to develop additional products to stimulate key pathways, including those used by the body’s immune system to inhibit the growth and metastasis of cancers. We have multiple programs under development and expect to file at least eight investigational new drug, or IND, applications through 2015. We also have a portfolio of small molecule drug candidates in early stages of development, which we are not actively developing ourselves but are seeking partners to pursue further development and potential commercialization.
Enabling technology
Synthetic biology entails the application of engineering principles to biological systems for the purpose of designing and constructing new biological systems or redesigning/modifying existing biological systems. Biological systems are governed by DNA, the building blocks of gene programs, which control cellular processes by coding for the production of proteins and other molecules that have a functional purpose and by regulating the activities of these molecules. This regulation occurs via complex biochemical and cellular reactions working through intricate cell signaling pathways, and control over these molecules modifies the output of biological systems. Synthetic biology has been enabled by the application of information technology and advanced statistical analysis, also known as bioinformatics, to genetic engineering, as well as by improvements in DNA synthesis. Synthetic biology aims to engineer gene-based programs or
S-1
codes to modify cellular function to achieve a desired biological outcome. Its application is intended to allow more precise control of drug concentration and dose, thereby improving the therapeutic index associated with the resulting drug.
On January 6, 2011, we entered into an Exclusive Channel Partner Agreement with Intrexon, which we refer to as the Channel Agreement, to develop and commercialize novel DNA-based therapeutics in the field of cancer treatment by combining Intrexon’s synthetic biology platform with our capabilities to translate science to the patient setting. As a result, our DNA synthetic biology platform employs an inducible gene-delivery system that enables controlled delivery of genes that produce therapeutic proteins to treat cancer. The first example of this regulated controlled delivery is achieved by producing IL-12, a potent, naturally occurring anti-cancer protein, under the control of Intrexon’s proprietary biological “switch” to turn on/off the therapeutic protein expression at the tumor site. We and Intrexon refer to this “switch” as the RheoSwitch Therapeutic System® or RTS® platform. Our initial drug candidates being developed using the synthetic biology platform are Ad-RTS-IL-12 and DC-RTS-IL-12, with a current focus on Ad-RTS-IL-12.
We have demonstrated that we are able to simultaneously express multiple effectors under control of the RTS® platform from the same construct. In the mouse, we have also shown that we are able to express multigenic DNA constructs in an embedded, controlled bioreactor, by injecting into skeletal muscle and measuring the DNA-coded proteins in the blood. Furthermore, we have also demonstrated the ability to express these same three genes under RTS® platform control in mesenchymal stem cells, or MSCs.
Recent developments
On October 21 and 22, 2013, we presented preclinical data from four studies at the 2013 joint meeting of the American Association for Cancer Research, the National Cancer Institute and the European Organization for Research and Treatment of Cancer, which we refer to as the 2013 AARC-NCI-EORTC. The reported data further supports the breadth of the Intrexon synthetic biology platform technology and the ability to express immunotherapies, and other therapies, in bothin vitro andin vivo models. The following summarizes the reported results:
The Controlled Local Expression of IL-12 as an Immunotherapeutic Treatment of Glioma study was designed to evaluate the viability of IL-12 expression-based therapeutic candidates in the treatment of glioma, or brain cancer. Two different RTS® controlled IL-12 expression-based therapeutic candidates were explored for the study, Ad-RTS-IL-12 (AD) and DC-RTS-IL-12 (DC), along with the orally-available small molecule activator ligand veledimex. Results demonstrated that the activator ligand achieved brain penetration in normal mouse and monkey models. Further, treatment with both AD and DC demonstrated dose-related increase in survival in a mouse model with no adverse clinical signs. Animals treated with DC > 5000 MOI (multiplicity of infection) or AD 5 x 109 viral particles survived throughout the duration of the study (100% survival at 75 days) with no adverse clinical signs observed. In contrast, the mean survival in the control groups was 22 (±3) days. We believe these findings support the utility of localized regulated IL-12 production as an approach for the treatment of malignant glioma.
The three additional abstracts presented at the 2013 AARC-NCI-EORTC meeting demonstrated (i) systemic expression of three distinct immune effectors from a single RTS® regulated multigenic
S-2
construct in mice; (ii) in vitro data supporting the potential use of MSCs for tumor-targeted delivery of single or multiple RTS® regulated cancer immunotherapies; and (iii) data supporting functional single chain variable fragment-Fc fusion proteins as an alternate approach to monoclonal antibodies which are more amenable for multi-genic therapies. These results highlight the potential use of skeletal muscle as an embedded controllable bioreactor to generate therapeutics for tumor-targeted delivery of single or multiple RTS® regulated cancer immunotherapies which could potentially be translated into an effective clinical regimen in the treatment of cancer. Furthermore, the potential use of MSCs for tumor-targeted delivery of single or multiple RTS® regulated cancer immunotherapies could potentially be translated into an effective clinical regimen for a variety of cancers. Furthermore, RTS® driven expression of trastuzumab- and cetuximab-based scFv-Fc constructs from an embedded controllable bioreactor have potential utility as DNA-based anticancer therapeutics.
Product candidates
The following chart identifies our current synthetic biology product candidates and their stage of development, each of which are described in more detail below.
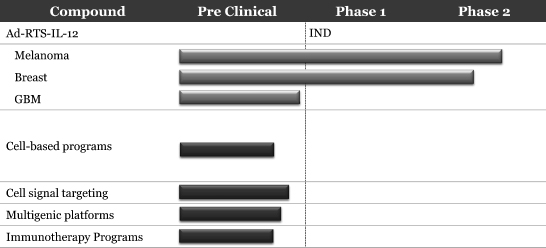
Synthetic biology programs:
Ad-RTS-IL-12 + Activator Ligand. Ad-RTS-IL-12 is currently being tested in two Phase 2 studies, the first for the treatment of metastatic melanoma, and the second for the treatment of non-resectable recurrent or metastatic breast cancer. Ad-RTS IL-12 is our lead drug candidate, which uses our gene delivery system to produce IL-12.
In March 2013, we announced the initiation of a randomized, open label Phase 2 clinical study of Ad-RTS-IL-12 to treat metastatic breast cancer. The two-part, multi-center U.S. study is enrolling patients with non-resectable, recurrent or metastatic breast cancer who have visible lesions or lesions accessible by injection. The primary endpoint of the study is rate of progression-free survival at 16 weeks. Secondary endpoints include objective response rate and duration of response. Initiation of the clinical study was followed by the presentation of results, from a study
S-3
in a breast cancer murine preclinical model, demonstrating the anti-tumor effects and tolerability of Ad-RTS-IL-12. The data were presented at the American Association for Cancer Research 2013 Annual Meeting in April.
In May 2013, we announced promising results from nonclinical studies and a Phase 1/2 study in metastatic melanoma using Ad-RTS-IL-12. In these studies, the controlled expression of IL-12, through a regulatable gene therapy strategy, was found to limit systemic toxicity while inducing biological and clinical activity in a dose-dependent fashion. The findings were presented in an oral session at the 16th Annual Meeting of the American Society of Gene and Cell Therapy, or ASGCT. In June, updated results were presented at the 2013 American Society for Clinical Oncology, or ASCO. Ad-RTS-IL-12 + Activator Ligand induce production of IL-12 mRNA in the tumor microenvironment (switch on). Upon removal of the oral activator ligand, IL-12 mRNA levels return to baseline (switch off). Following treatment with Ad-RTS-IL-12 + Activator Ligand, increases in TILs (CD8+, CD45RO+) were observed in the tumor microenvironment. Clinical activity was observed in injected and non-injected lesions primarily at the higher doses of the activator ligand. Inflammation, shrinkage, flattening, and depigmentation of lesions correlated with the highest serum levels of IFN-g. Ad-RTS-IL-12 + Activator Ligand therapy was generally well-tolerated and its safety profile is consistent with other immunotherapies.
The Phase 1 portion of the Phase 1/2 is study is complete, and the Phase 2 portion is on-going. This Phase 2 study, a multi-center, single-arm, open-label study, is enrolling patients with unresectable Stage III or IV melanoma and further evaluating the safety and efficacy of intratumoral injections of Ad-RTS-IL-12 in combination with an oral activator ligand. Data from this Phase 2 study is expected in the fourth quarter of 2013.
We are in the process of finalizing clinical protocol designs that will lead to the initiation of Phase 2 studies in the combination with standard of care, or SOC, in first quarter 2014 for the treatment of metastatic melanoma and metastatic breast cancer. Specifically, we expect to commence enrollment in a glioblastoma mulitforme Phase 1 dose-escalation study in the first quarter of 2014, with preliminary data expected near the end of 2014; in a melanoma Phase 2 combination study with SOC study in the first quarter of 2014, with preliminary data expected near the end of 2014; and in a breast cancer Phase 2 combination study with SOC in the first quarter of 2014, with preliminary data expected in the first quarter of 2015. Melanoma, breast cancer, and glioma (detailed below) represent significant market potentials with high unmet medical needs. The annual incidence in the United States of melanoma, breast cancer and glioblastoma is 76,690 patients, 234,580 patients and 18,000 patients, respectively, with the majority of those representing a high unmet medical need.
DC-RTS-IL-12 + Activator Ligand. We completed enrollment in a Phase 1 dose escalation study of DC-RTS-IL-12 in the second quarter of 2012 in the United States. DC-RTS-IL-12 employs intratumoral injection of modified dendritic cells from each patient and oral dosing of an activator ligand to turn on in vivo expression of IL-12. DC-RTS-IL-12, through the RTS® platform, controls the timing and level of transgene expression. The RTS® technology functions as a “gene switch” for the regulated expression of human IL-12 in the patients’ dendritic cells which are transduced with a replication incompetent adenoviral vector carrying the IL-12 gene under the control of the RTS® platform. Currently, there are no actively enrolling studies using DC-RTS-IL-12, as we have prioritized our clinical development efforts on Ad-RTS-IL-12.
S-4
Earlier stage programs. Preclinical mouse glioma studies evaluating either Ad-RTS-IL-12 orDC-RTS-IL-12 therapy demonstrated a survival benefit in all animals treated at higher doses with no adverse clinical signs and symptoms. Additional preclinical studies are currently ongoing with Ad-RTS-IL-12 to enable initiation of a Phase 1 clinical study in the first half of 2014. This Phase 1 clinical study will evaluate the safety and tolerability of the Ad-RTS-IL-12 therapy in patients with recurrent or progressive glioblastoma. Glioblastoma is by far the most frequent malignant glioma and is associated with a particularly aggressive course and dismal prognosis. The current standard of care is based on surgical resection to the maximum extent, followed by radiotherapy and concomitant adjuvant temozolomide. Such aggressive treatment is associated with only modest improvements in survival. Newly diagnosed glioblastoma patients have a median overall survival, or OS, of 11-17 month, 2 year OS rate, between 15-17%.
We are actively pursuing several synthetic biology approaches, including gene delivery with human MSCs, functional single chain variable fragment-Fc fusion proteins and multigenic approaches in our discovery pipeline to address unmet medical needs in cancer that are expected to result in multiple INDs in 2014 and beyond. Each of the candidates has been designed, built, and tested in our discovery and preclinical program, including significant progress made to date with multigenic approaches to cancer treatment that will target more than one biologic pathway. It is currently well accepted that combining multiple immunomodulatory therapeutic modalities should have a more profound impact promoting cancer remission than monotherapies.
Small molecule programs
Palifosfamide, ZIO-201. The small molecule palifosfamide, or isophosphoramide mustard, is a proprietary active metabolite of the pro-drug ifosfamide. Because palifosfamide is the stabilized active metabolite of ifosfamide and a distinct pharmaceutical composition without the acrolein or chloroacetaldehyde metabolites we believe that the administration of palifosfamide may be an effective and well-tolerated agent to treat cancer. In addition to anticipated lower toxicity, palifosfamide may have other advantages over ifosfamide and cyclophosphamide. Palifosfamide cross-links DNA differently than the active metabolite of cyclophosphamide, resulting in a different activity profile. We are seeking to out-license or otherwise monetize palifosfamide.
Soft Tissue Sarcoma. Previously we have studied palifosfamide in combination with doxorubicin in patients with soft tissue sarcoma. In March 2013, we announced that the Phase 3 study, PICASSO 3, did not meet its primary endpoint of progression-free survival, and that we would terminate our development program in metastatic soft tissue sarcoma. PICASSO 3 study data was presented at the 2013 European Cancer Congress.
Small-Cell Lung Cancer. Small-Cell Lung Cancer, or SCLC, is almost exclusively associated with smoking. Standard of care for SCLC, which is etoposide and platinum therapy, has changed little in decades. Published studies of ifosfamide in combination with standard of care have evidenced enhanced efficacy but also with enhanced side effects, providing for an unfavorable benefit to risk association. We believe that combining palifosfamide with standard of care could offer a separation of enhanced efficacy with reduced toxicity.
S-5
Data from a Phase 1 trial of palifosfamide in combination with etoposide and carboplatin informed appropriate dosing for initiating an adaptive Phase 3 trial in first-line, metastatic SCLC. In June 2012, the Company initiated an international, multi-center, open-label, adaptive, randomized study of palifosfamide in combination with carboplatin and etoposide, or PaCE, chemotherapy versus carboplatin and etoposide, or CE, alone in chemotherapy naïve patients with metastatic small cell lung cancer, which we refer to as MATISSE. The trial’s primary endpoint is overall survival.
Based on the outcome of PICASSO 3 in soft tissue sarcoma and the resulting revision in the company’s development plans for palifosfamide, enrollment in this study was suspended with 188 patients enrolled. The interim analysis of overall survival events in MATISSE is forecasted to be reached in the first half of 2014.
Darinaparsin, ZIO-101. Darinaparsin is an anti-mitochondrial (organic arsenic) compound (covered by issued patents and pending patent applications in the United States and in foreign countries). Phase 1 testing of the intravenous, or IV, form of darinaparsin in solid tumors and hematological cancers was completed. We reported clinical activity and a safety profile from these studies as predicted by preclinical results. We subsequently completed Phase 2 studies in advanced myeloma, primary liver cancer and in certain other hematological cancers. At the May 2009 annual meeting of ASCO, we reported favorable results from the IV trial in lymphoma, particularly peripheral T-cell lymphoma, or PTCL. A Phase 1 trial in solid tumors with an oral form of darinaparsin has completed enrollment. We have obtained Orphan Drug Designation for darinaparsin in the United States and Europe for the treatment of PTCL and have entered into a licensing agreement with Solasia Pharma K.K., or Solasia, for the Asia/Pacific territory with a focus on IV-administered darinaparsin in PTCL. Clinical studies are currently ongoing with Solasia. We are seeking to out-license or otherwise monetize darinaparsin for territories not covered by our agreement with Solasia.
Indibulin, ZIO-301. Indibulin is a novel, small molecule inhibitor of tubulin polymerization and is potentially safer than other tubulin inhibitors as no neurotoxicity has been observed in preclinical studies or in Phase 1 clinical trials. Indibulin has a different pharmacological profile from other tubulin inhibitors currently on the market as it binds to a unique site on tubulin and is active in multi-drug-resistant (MDR-1, MRP-1) and taxane-resistant tumors. A Phase 1 study was conducted in late stage metastatic breast cancer and was found to be safe and tolerable. We are seeking to out-license or otherwise monetize indibulin.
Development plans
We are currently pursuing several clinical development opportunities, principally in our synthetic biology programs. We are also evaluating additional potential preclinical candidates and continuing discovery efforts aimed at identifying other potential product candidates under our Channel Agreement with Intrexon. In addition, we may seek to enhance our pipeline in synthetic biology through highly focused strategic transactions, which may include acquisitions, partnerships and in-licensing activities. We are actively seeking to out-license some or all of our small molecule programs to further support our synthetic biology efforts.
Our current plans involve using our principal internal financial resources to develop the synthetic biology program, with the intention of ultimately partnering or otherwise raising additional
S-6
capital to support further development activities for our strategic product candidates. As of September 30, 2013, we had approximately $23.6 million of cash and cash equivalents. Based upon our current plans and without taking into account the net proceeds of this offering, we anticipate that our cash resources will be sufficient to fund our operations into the first quarter of 2014. This forecast of cash resources is forward-looking information that involves risks and uncertainties, and the actual amount of our expenses over the next six months could vary materially and adversely as a result of a number of factors, including the factors discussed in the “Risk Factors” section of this prospectus supplement and the uncertainties applicable to our forecast for the overall sufficiency of our capital resources. We have based our estimates on assumptions that may prove to be wrong, and our expenses could prove to be significantly higher than we currently anticipate.
Furthermore, the successful development of our product candidates is highly uncertain. Product development costs and timelines can vary significantly for each product candidate, are difficult to accurately predict, and will require us to obtain additional funding, either alone or in connection with partnering arrangements. Various statutes and regulations also govern or influence the development, manufacturing, safety, labeling, storage, record keeping and marketing of each product. The lengthy process of seeking approval and the subsequent compliance with applicable statutes and regulations require the expenditure of substantial resources. Any failure by us to obtain, or any delay in obtaining, regulatory approvals could materially, adversely affect our business. To date, we have not received approval for the sale of any product candidates in any market and, therefore, have not generated any revenues from our product candidates.
Risk factors
An investment in our common stock is subject to a number of risks and uncertainties. Before investing in our common stock, you should carefully consider the following, as well as the more detailed discussion of risk factors and other information included in this prospectus supplement.
| • | | We will require additional financial resources in order to continue on-going development of our product candidates; if we are unable to obtain these additional resources, we may be forced to delay or discontinue clinical testing of our product candidates. |
| • | | We need to raise additional capital to fund our operations. The manner in which we raise any additional funds may affect the value of your investment in our common stock. |
| • | | Clinical trials are very expensive, time-consuming, and difficult to design and implement, and may not support our product candidate claims. |
| • | | We may not be able to commercialize any products, generate significant revenues, or attain profitability. |
| • | | The technology underlying our Channel Arrangement with Intrexon is based in part on early stage technology in the field of human oncologic therapeutics, which makes it difficult to predict the time and cost of product candidate development and subsequently obtaining regulatory approval. |
| • | | Currently, no gene therapy products have been approved in the United States and only one product has been approved in Europe. |
S-7
| • | | Our use of synthetic biology to develop product candidates may become subject to increasing regulation, or may be limited by ethical, legal and social concerns about synthetic biologically engineered products. |
| • | | We have a limited operating history upon which to base an investment decision. |
Corporate information
We originally incorporated in Colorado in September 1998 (under the name Net Escapes, Inc.) and later changed our name to “EasyWeb, Inc.” in February 1999. We re-incorporated in Delaware on May 16, 2005 under the same name. On September 13, 2005, we completed a “reverse” acquisition of privately held ZIOPHARM, Inc., a Delaware corporation. To effect this transaction, we caused ZIO Acquisition Corp., our wholly-owned subsidiary, to merge with and into ZIOPHARM, Inc., with ZIOPHARM, Inc. surviving as our wholly owned subsidiary. In accordance with the terms of the merger, the outstanding common stock of ZIOPHARM, Inc. automatically converted into the right to receive an aggregate of approximately 97.3% of our outstanding common stock (after giving effect to the transaction). Following the merger, we caused ZIOPHARM, Inc. to merge with and into us and we changed our name to “ZIOPHARM Oncology, Inc.” Although EasyWeb, Inc. was the legal acquirer in the transaction, we accounted for the transaction as a reverse acquisition under generally accepted accounting principles. As a result, ZIOPHARM, Inc. became the registrant with the Commission and the historical financial statements of ZIOPHARM, Inc. became our historical financial statements.
Our principal executive offices are located at One First Avenue, Parris Building 34, Navy Yard Plaza, Boston, Massachusetts 02129, and our telephone number is (617) 259-1970. Our Internet site iswww.ziopharm.com. Information found on, or accessible through, our website is not a part of, and is not incorporated into, this prospectus supplement or the accompanying prospectus, and you should not consider it part of this prospectus supplement or part of the accompanying prospectus.
S-8
The offering
Common stock offered
by us in this offering | 14,300,000 shares |
Option to purchase additional shares | We have granted the underwriters an option for a period of up to 30 days from the date of this prospectus supplement to purchase up to 2,145,000 additional shares of common stock at the public offering price less the underwriting discounts and commissions. |
Common stock to be outstanding immediately after this offering | 97,981,580 shares (or 100,126,580 shares if the underwriters exercise in full their option to purchase additional shares) |
Use of proceeds | We intend to use the net proceeds from this offering for the overall development of our drug candidates, including our synthetic biology candidates and small molecule candidates, and for general corporate and working capital purposes. See “Use of Proceeds.” |
Risk factors | See “Risk factors” beginning on page S-11 for a discussion of some of the factors you should carefully consider before deciding to invest in shares of our common stock. |
NASDAQ Capital Market symbol | ZIOP |
The number of shares of common stock to be outstanding immediately after this offering is based on 83,681,580 shares of common stock outstanding as of September 30, 2013 and does not include:
| • | | 5,730,169 shares of our common stock issuable upon the exercise of stock options outstanding as of September 30, 2013, having a weighted average exercise price of $3.73 per share; |
| • | | 1,565,070 shares of our common stock available as of September 30, 2013 for future issuance pursuant to our 2003 Stock Option Plan and our 2012 Equity Incentive Plan; and |
| • | | 10,392,387 shares of our common stock issuable upon the exercise of outstanding warrants as of September 30, 2013 with a weighted-average exercise price of $3.57 per share. |
Except as otherwise indicated, all information in this prospectus supplement assumes no exercise by the underwriters of their option to purchase additional shares.
S-9
Indication of interest
Intrexon, which is affiliated with Randal J. Kirk, who serves as one of our directors, has agreed to purchase 2,857,143 shares of our common stock in this offering at the public offering price. The underwriters will receive the same discount from such shares of our common stock as they will from any other shares of our common stock sold to the public in this offering.
S-10
Risk factors
An investment in our common stock involves a high degree of risk. Before deciding whether to invest in our common stock, you should consider carefully the risks described below and discussed under the section captioned “Risk Factors” contained in our Quarterly Report on Form 10-Q for the period ended September 30, 2013, which is incorporated by reference in this prospectus supplement and the accompanying prospectus, in its entirety, together with other information in this prospectus supplement, the accompanying prospectus, the information and documents incorporated by reference, and in any free writing prospectus that we have authorized for use in connection with this offering. If any of these risks actually occurs, our business, financial condition, results of operations or cash flow could be harmed. This could cause the trading price of our common stock to decline, resulting in a loss of all or part of your investment. The risks described below and in the documents referenced above are not the only ones that we face. Additional risks not presently known to us or that we currently deem immaterial may also affect our business.
Risks related to our business
We will require additional financial resources in order to continue ongoing development of our product candidates; if we are unable to obtain these additional resources, we may be forced to delay or discontinue clinical testing of our product candidates.
We have not generated significant revenue and have incurred significant net losses in each year since our inception. For the nine months ended September 30, 2013, we had a net loss of $48.2 million, and, as of September 30, 2013, we had incurred approximately $331.9 million of cumulative net losses since our inception in 2003. We expect to continue to incur significant operating expenditures and net losses. Further development of our product candidates, including product candidates that we may develop under our Channel Agreement with Intrexon, will likely require substantial increases in our expenses as we:
| • | | continue to undertake clinical trials for product candidates; |
| • | | scale-up the formulation and manufacturing of our product candidates; |
| • | | seek regulatory approvals for product candidates; |
| • | | implement additional internal systems and infrastructure; and |
| • | | hire additional personnel. |
We continue to seek additional financial resources to fund the further development of our product candidates. If we are unable to obtain sufficient additional capital, one or more of these programs could be placed on hold. Because we are currently devoting a significant portion of our resources to the development of synthetic biology and our adaptive Phase 3 trial for first-line SCLC for IV palifosfamide, MATISSE, further progress with the development of our other candidates may be significantly delayed and may depend on the licensing of those compounds to third parties.
We anticipate that our cash resources without giving effect to the proceeds of this offering will be sufficient to fund our operations into the first quarter of 2014, and we have no current committed sources of additional capital. As a result, our independent registered public accounting firm has expressed a substantial doubt about our ability to continue as a going concern in their report on our financial statements. We do not know whether additional financing will be available on terms favorable or acceptable to us when needed, if at all. Our
S-11
business is highly cash-intensive and our ability to continue operations after our current cash resources are exhausted depends on our ability to obtain additional financing and/or achieve profitable operations, as to which no assurances can be given. If adequate additional funds are not available when required, or if we are unsuccessful in entering into partnership agreements for the further development of our products, we will be required to delay, reduce or eliminate planned preclinical and clinical trials and may be forced to terminate the approval process for our product candidates from the FDA or other regulatory authorities. In addition, we could be forced to discontinue product development, forego attractive business opportunities or pursue merger or divestiture strategies. In the event we are unable to obtain additional financing, we may be forced to cease operations altogether.
We need to raise additional capital to fund our operations. The manner in which we raise any additional funds may affect the value of your investment in our common stock.
As of September 30, 2013, we had incurred approximately $331.9 million of cumulative net losses and had approximately $23.6 million of cash and cash equivalents. We anticipate that our cash resources without giving effect to the proceeds of this offering will be sufficient to fund our operations into the first quarter of 2014. Following negative results in our PICASSO 3 pivotal trial in first-line metastatic soft tissue sarcoma, or STS, in March 2013, we implemented a workforce reduction plan and other cost-cutting measures in an attempt to extend our cash resources as long as possible, though there are no assurances that such efforts will be effective. In addition, changes may occur that would consume our existing capital prior to the first quarter of 2014, including expansion of the scope of, and/or slower than expected progress of, our research and development efforts and changes in governmental regulation. As a result, our independent registered public accounting firm has expressed a substantial doubt about our ability to continue as a going concern in their report on our financial statements. Actual costs may ultimately vary from our current expectations, which could materially impact our use of capital and our forecast of the period of time through which our financial resources will be adequate to support our operations. We have estimated the sufficiency of our cash resources based in part on the discontinuation of the PICASSO 3 pivotal trial for first-line metastatic STS and our adaptive Phase 3 trial for first-line SCLC for IV palifosfamide and our current timing expectations for the interim analysis of data in the MATISSE trial. Also our estimates include the advancement of our synthetic biology product candidates in the clinic under our Channel Agreement with Intrexon, and we expect that the costs associated with these and additional product candidates will increase the level of our overall research and development expenses significantly going forward.
In addition to above factors, our actual cash requirements may vary materially from our current expectations for a number of other factors that may include, but are not limited to, changes in the focus and direction of our development programs, competitive and technical advances, costs associated with the development of our product candidates, our ability to secure partnering arrangements, and costs of filing, prosecuting, defending and enforcing our intellectual property rights. If we exhaust our capital reserves more quickly than anticipated, regardless of the reason, and we are unable to obtain additional financing on terms acceptable to us or at all, we will be unable to proceed with development of some or all of our product candidates on expected timelines and will be forced to prioritize among them.
The unpredictability of the capital markets may severely hinder our ability to raise capital within the time periods needed or on terms we consider acceptable, if at all. Moreover, if we fail to advance one or more of our current product candidates to later-stage clinical trials, successfully
S-12
commercialize one or more of our product candidates, or acquire new product candidates for development, we may have difficulty attracting investors that might otherwise be a source of additional financing.
Our need for additional capital and limited capital resources may force us to accept financing terms that could be significantly dilutive to existing stockholders. To the extent that we raise additional capital by issuing equity securities, our stockholders may experience dilution. In addition, we may grant future investors rights superior to those of our existing stockholders. If we raise additional funds through collaborations and licensing arrangements, it may be necessary to relinquish some rights to our technologies, product candidates or products, or grant licenses on terms that are not favorable to us. If we raise additional funds by incurring debt, we could incur significant interest expense and become subject to covenants in the related transaction documentation that could affect the manner in which we conduct our business.
Clinical trials are very expensive, time-consuming, and difficult to design and implement.
Human clinical trials are very expensive and difficult to design and implement, in part because they are subject to rigorous regulatory requirements. The clinical trial process itself is also time-consuming and results are inherently uncertain. We estimate that clinical trials of our product candidates will take at least several years to complete. Furthermore, failure can occur at any stage of the trials, and we could encounter problems that cause us to abandon or repeat clinical trials. The commencement and completion of clinical trials may be delayed by several factors, including:
| • | | unforeseen safety issues; |
| • | | determination of dosing issues; |
| • | | lack of effectiveness during clinical trials; |
| • | | slower than expected rates of patient recruitment and enrollment; |
| • | | inability to monitor patients adequately during or after treatment; |
| • | | inability or unwillingness of medical investigators to follow our clinical protocols; and |
| • | | regulatory determinations to temporarily or permanently cease enrollment for other reasons not related to patient safety. |
Success in preclinical testing and early clinical trials does not ensure that later clinical trials will be successful. For example, despite positive findings in earlier clinical trials, our product candidate palifosfamide failed to meet the primary endpoint of the Phase 3 PICASSO 3 trail. In addition, we or the FDA may suspend our clinical trials at any time if it appears that we are exposing participants to unacceptable health risks or if the FDA finds deficiencies in our Investigational New Drug, or IND, submission or in the conduct of these trials.
See also “—Our product candidates are in various stages of clinical trials, which are very expensive and time-consuming. We cannot be certain when we will be able to file an NDA or BLA, with the FDA and any failure or delay in completing clinical trials for our product candidates could harm our business.”
We may not be able to obtain or maintain orphan drug exclusivity for our product candidates.
We have received Orphan Drug designations for darinaparsin for the treatment of peripheralT-cell lymphoma in both the United States and Europe, and we may be able to receive additional
S-13
Orphan Drug designation from the FDA and the European Medicines Agency, or EMA, for other product candidates. In the United States, orphan designation is available to drugs intended to treat, diagnose or prevent a rare disease or condition that affects fewer than 200,000 people in the United States at the time of application for orphan designation. Orphan designation qualifies the sponsor of the product for a tax credit and marketing incentives. The first sponsor to receive FDA marketing approval for a drug with an orphan designation is entitled to a seven-year exclusive marketing period in the United States for that product for that indication and, typically, a waiver of the prescription drug user fee for its marketing application. However, a drug that the FDA considers to be clinically superior to, or different from, the approved orphan drug, even though for the same indication, may also obtain approval in the United States during the seven-year exclusive marketing period. Orphan drug exclusive marketing rights may also be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug. There is no guarantee that any of our other product candidates will receive Orphan Drug designation or that, even if such product candidate is granted such status, the product candidate’s clinical development and regulatory approval process will not be delayed or will be successful.
We may not be able to commercialize any products, generate significant revenues, or attain profitability.
To date, none of our product candidates have been approved for commercial sale in any country. The process to develop, obtain regulatory approval for, and commercialize potential drug candidates is long, complex, and costly. Unless and until we receive approval from the FDA and/or other regulatory authorities for our product candidates, we cannot sell our drugs and will not have product revenues. Even if we obtain regulatory approval for one or more of our product candidates, if we are unable to successfully commercialize our products, we may not be able to generate sufficient revenues to achieve or maintain profitability, or to continue our business without raising significant additional capital, which may not be available. Our failure to achieve or maintain profitability could negatively impact the trading price of our common stock.
Ethical, legal and social concerns about synthetic biologically engineered products could limit or prevent the use of our product candidates.
Our products candidates use a synthetic biology platform. Public perception about the safety and environmental hazards of, and ethical concerns over, genetically engineered products could influence public acceptance of our product candidates. If we are not able to overcome the ethical, legal and social concerns relating to synthetic biological engineering, our product candidates may not be accepted. These concerns could result in increased expenses, regulatory scrutiny, delays or other impediments to the public acceptance and commercialization of our product candidates. Our ability to develop and commercialize products could be limited by public attitudes and governmental regulation.
The subject of genetically modified organisms has received negative publicity, which has aroused public debate. This adverse publicity could lead to greater regulation and trade restrictions on imports of genetically altered products. Further, there is a risk that our product candidates could cause adverse health effects or other adverse events, which could also lead to negative publicity.
The synthetic biological platform that we use may have significantly enhanced characteristics compared to those found in naturally occurring organisms, enzymes or microbes. While we
S-14
believe we produce synthetic biological technologies only for use in a controlled laboratory and industrial environment, the release of such synthetic biological technologies into uncontrolled environments could have unintended consequences. Any adverse effect resulting from such a release could have a material adverse effect on our business and financial condition, and we may have exposure to liability for any resulting harm.
Our use of synthetic biology to develop product candidates may become subject to increasing regulation in the future.
Most of the laws and regulations concerning synthetic biology relate to the end products produced using synthetic biology, but that may change. For example, the Presidential Commission for the Study of Bioethical Issues in December 2010 recommended that the federal government oversee, but not regulate, synthetic biology research. The Presidential Commission also recommended that the government lead an ongoing review of developments in the synthetic biology field. Synthetic biology may become subject to additional government regulations as a result of the recommendations, which could require us to incur significant additional capital and operating expenditures and other costs in complying with these laws and regulations.
The technology on which our Channel Agreement with Intrexon Corporation is based in part on early stage technology in the field of human oncologic therapeutics.
Our Channel Agreement with Intrexon contemplates our using Intrexon’s advanced transgene engineering platform for the controlled and precise cellular production of anti-cancer effectors. The synthetic biology effector platform in which we have acquired rights represents early-stage technology in the field of human oncologic biotherapeutics, with DC-RTS-IL-12 having completed a Phase 1 study in melanoma and Ad-RTS-IL-12 currently in two Phase 2 studies, in melanoma and breast cancer. Although we plan to leverage Intrexon’s synthetic biology platform for additional products targeting key pathways used by cancers to grow and metastasize, we may not be successful in developing and commercializing these products for a variety of reasons. The risk factors set forth herein that apply to our small molecule drug candidates, which are in various stages of development, also apply to product candidates that we seek to develop under our Channel Agreement with Intrexon.
We will incur additional expenses in connection with our Channel Agreement with Intrexon Corporation.
The synthetic biology platform, in which we have acquired rights for cancer from Intrexon, includes two existing product candidates, DC-RTS-IL-12 and Ad-RTS-IL-12. Upon entry into the Channel Agreement with Intrexon, we assumed responsibility for the clinical development of these product candidates, which we expect will increase the level of our overall research and development expenses significantly going forward. Although all human clinical trials are expensive and difficult to design and implement, we believe that due to complexity, costs associated with clinical trials for synthetic biology products are greater than the corresponding costs associated with clinical trials for small molecule candidates. In addition to increased research and development costs, prior to the adoption of our March 2013 workforce reduction plan, we added headcount in part to support our Channel Agreement endeavors, and we may need to do so again in the future which would add to our general and administrative expenses going forward.
S-15
Although our forecasts for expenses and the sufficiency of our capital resources takes into account our plans to develop the Intrexon products, we assumed development responsibility for these products on January 6, 2011, and the actual costs associated therewith may be significantly in excess of forecasted amounts. In addition to the amount and timing of expenses related to the clinical trials, our actual cash requirements may vary materially from our current expectations for a number of other factors that may include, but are not limited to, changes in the focus and direction of our development programs, competitive and technical advances, costs associated with the development of our product candidates and costs of filing, prosecuting, defending and enforcing our intellectual property rights. If we exhaust our capital reserves more quickly than anticipated, regardless of the reason, and we are unable to obtain additional financing on terms acceptable to us or at all, we will be unable to proceed with development of some or all of our product candidates on expected timelines and will be forced to prioritize among them.
We may not be able to retain the exclusive rights licensed to us by Intrexon Corporation to develop and commercialize products involving DNA administered to humans for expression of anti-cancer effectors for the purpose of treatment or prophylaxis of cancer.
Under the Channel Agreement, we use Intrexon’s technology directed towards in vivo expression of effectors in connection with the development of DC-RTS-IL-12 and Ad-RTS-IL-12 and generally to research, develop and commercialize products, in each case in which DNA is administered to humans for expression of anti-cancer effectors for the purpose of treatment or prophylaxis of cancer, which we collectively refer to as the Cancer Program. The Channel Agreement grants us a worldwide license to use patents and other intellectual property of Intrexon in connection with the research, development, use, importing, manufacture, sale, and offer for sale of products involving DNA administered to humans for expression of anti-cancer effectors for the purpose of treatment or prophylaxis of cancer, which we refer to collectively as the ZIOPHARM Products. Such license is exclusive with respect to any clinical development, selling, offering for sale or other commercialization of ZIOPHARM Products, and otherwise is non-exclusive. Subject to limited exceptions, we may not sublicense the rights described without Intrexon’s written consent. Under the Channel Agreement, and subject to certain exceptions, we are responsible for, among other things, the performance of the Cancer Program, including development, commercialization and certain aspects of manufacturing of ZIOPHARM Products.
Intrexon may terminate the Channel Agreement if we fail to use diligent efforts to develop and commercialize ZIOPHARM Products or if we elect not to pursue the development of a Cancer Program identified by Intrexon that is a “Superior Therapy” as defined in the Channel Agreement. We may voluntarily terminate the Channel Agreement upon 90 days written notice to Intrexon. Upon termination of the Channel Agreement, we may continue to develop and commercialize any ZIOPHARM Product that, at the time of termination:
| • | | is being commercialized by us; |
| • | | has received regulatory approval; |
| • | | is a subject of an application for regulatory approval that is pending before the applicable regulatory authority; or |
| • | | is the subject of at least an ongoing Phase 2 clinical trial (in the case of a termination by Intrexon due to an uncured breach or a voluntary termination by us), or an ongoing Phase 1 clinical trial in the field (in the case of a termination by us due to an uncured breach or a |
S-16
| | termination by Intrexon following an unconsented assignment by us or our election not to pursue development of a Superior Therapy). |
Our obligation to pay 50% of net profits or revenue as described further in our Annual Report on Form 10-K under the heading “Business—License Agreements, Intellectual Property and Other Agreements—Exclusive Channel Partner Agreement with Intrexon Corporation” with respect to these “retained” products will survive termination of the Channel Agreement.
There can be no assurance that we will be able to successfully perform under the Channel Agreement and if the Channel Agreement is terminated it may prevent us from achieving our business objectives.
We have a limited operating history upon which to base an investment decision.
We are a development-stage company that was incorporated in September 2003. To date, we have not demonstrated an ability to perform the functions necessary for the successful commercialization of any product candidates. The successful commercialization of any product candidates will require us to perform a variety of functions, including:
| • | | continuing to undertake preclinical development and clinical trials; |
| • | | participating in regulatory approval processes; |
| • | | formulating and manufacturing products; and |
| • | | conducting sales and marketing activities. |
Our operations have been limited to organizing and staffing our company, acquiring, developing and securing our proprietary product candidates, and undertaking preclinical and clinical trials of our product candidates. These operations provide a limited basis for you to assess our ability to commercialize our product candidates and the advisability of investing in our securities.
Because we currently neither have nor intend to establish internal research capabilities, we are dependent upon pharmaceutical and biotechnology companies and academic and other researchers to sell or license us their product candidates and technology.
Proposing, negotiating, and implementing an economically viable product acquisition or license is a lengthy and complex process. We compete for partnering arrangements and license agreements with pharmaceutical, biopharmaceutical, and biotechnology companies, many of which have significantly more experience than we do, and have significantly more financial resources. Our competitors may have stronger relationships with certain third parties including academic research institutions, with whom we are interested in collaborating and may have, therefore, a competitive advantage in entering into partnering arrangements with those third parties. We may not be able to acquire rights to additional product candidates on terms that we find acceptable, or at all.
We expect that any product candidate to which we acquire rights will require significant additional development and other efforts prior to commercial sale, including extensive clinical testing and approval by the FDA and applicable foreign regulatory authorities. All drug product candidates are subject to the risks of failure inherent in pharmaceutical product development, including the possibility that the product candidate will not be shown to be sufficiently safe or effective for approval by regulatory authorities. Even if our product candidates are approved, they may not be economically manufactured or produced, or be successfully commercialized.
S-17
We actively evaluate additional product candidates to acquire for development. Such additional product candidates, if any, could significantly increase our capital requirements and place further strain on the time of our existing personnel, which may delay or otherwise adversely affect the development of our existing product candidates. We must manage our development efforts and clinical trials effectively, and hire, train and integrate additional management, administrative, and sales and marketing personnel. We may not be able to accomplish these tasks, and our failure to accomplish any of them could prevent us from successfully growing.
We may not be able to successfully manage our growth.
In the future, if we are able to advance our product candidates to the point of, and thereafter through, clinical trials, we will need to expand our development, regulatory, manufacturing, marketing and sales capabilities or contract with third parties to provide for these capabilities. Any future growth will place a significant strain on our management and on our administrative, operational, and financial resources. Therefore, our future financial performance and our ability to commercialize our product candidates and to compete effectively will depend, in part, on our ability to manage any future growth effectively. To manage this growth, we must expand our facilities, augment our operational, financial and management systems, and hire and train additional qualified personnel. If we are unable to manage our growth effectively, our business may be harmed.
Our business will subject us to the risk of liability claims associated with the use of hazardous materials and chemicals.
Our contract research and development activities may involve the controlled use of hazardous materials and chemicals. Although we believe that our safety procedures for using, storing, handling and disposing of these materials comply with federal, state and local laws and regulations, we cannot completely eliminate the risk of accidental injury or contamination from these materials. In the event of such an accident, we could be held liable for any resulting damages and any liability could have a materially adverse effect on our business, financial condition, and results of operations. In addition, the federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposal of hazardous or radioactive materials and waste products may require our contractors to incur substantial compliance costs that could materially adversely affect our business, financial condition, and results of operations.
We rely on key executive officers and scientific and medical advisors, and their knowledge of our business and technical expertise would be difficult to replace.
We are highly dependent on Dr. Jonathan Lewis, our Chief Executive Officer, Caesar J. Belbel, our Executive Vice President and Chief Legal Officer and our principal scientific, regulatory, and medical advisors. Dr. Lewis’ and Mr. Belbel’s employment are governed by written employment agreements. The employment agreement with Dr. Lewis provides for a term that expires in January 2014. Dr. Lewis and Mr. Belbel may terminate their employment with us at any time, subject, however, to certain non-compete and non-solicitation covenants. The loss of the technical knowledge and management and industry expertise of Dr. Lewis and Mr. Belbel, or any of our other key personnel, could result in delays in product development, loss of customers and sales, and diversion of management resources, which could adversely affect our operating results. We do not carry “key person” life insurance policies on any of our officers or key employees.
S-18
If we are unable to hire additional qualified personnel, our ability to grow our business may be harmed.
We will need to hire additional qualified personnel with expertise in preclinical and clinical research and testing, government regulation, formulation and manufacturing, and eventually, sales and marketing. We compete for qualified individuals with numerous biopharmaceutical companies, universities, and other research institutions. Competition for such individuals is intense and we cannot be certain that our search for such personnel will be successful. Attracting and retaining qualified personnel will be critical to our success. If we are unable to hire additional qualified personnel, our ability to grow our business may be harmed.
We may incur substantial liabilities and may be required to limit commercialization of our products in response to product liability lawsuits.
The testing and marketing of medical products entail an inherent risk of product liability. If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit commercialization of our products, if approved. Even a successful defense would require significant financial and management resources. Regardless of the merit or eventual outcome, liability claims may result in:
| • | | decreased demand for our product candidates; |
| • | | injury to our reputation; |
| • | | withdrawal of clinical trial participants; |
| • | | withdrawal of prior governmental approvals; |
| • | | costs of related litigation; |
| • | | substantial monetary awards to patients; |
| • | | the inability to commercialize our product candidates. |
We currently carry clinical trial insurance and product liability insurance. However, an inability to renew our policies or to obtain sufficient insurance at an acceptable cost could prevent or inhibit the commercialization of pharmaceutical products that we develop, alone or with collaborators.
Our business and operations would suffer in the event of system failures.
Despite the implementation of security measures, our internal computer systems and those of our current and future contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we are not aware of any such material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations. For example, the loss of clinical trial data from completed or future clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. Likewise, we rely on third parties to manufacture our drug candidates and conduct clinical trials, and similar events relating to their computer systems could also have a material adverse effect on our business. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development and commercialization of our drug candidates could be delayed.
S-19
Risks related to the clinical testing, regulatory approval and manufacturing of our product candidates
If we are unable to obtain the necessary U.S. or worldwide regulatory approvals to commercialize any product candidate, our business will suffer.
We may not be able to obtain the approvals necessary to commercialize our product candidates, or any product candidate that we may acquire or develop in the future for commercial sale. We will need FDA approval to commercialize our product candidates in the United States and approvals from regulatory authorities in foreign jurisdictions equivalent to the FDA to commercialize our product candidates in those jurisdictions. In order to obtain FDA approval of any product candidate, we must submit to the FDA a New Drug Application, or NDA, or Biologics License Application, or BLA, demonstrating that the product candidate is safe for humans and effective for its intended use. This demonstration requires significant research and animal tests, which are referred to as preclinical studies, as well as human tests, which are referred to as clinical trials. Satisfaction of the FDA’s regulatory requirements typically takes many years, depending upon the type, complexity, and novelty of the product candidate, and will require substantial resources for research, development, and testing. We cannot predict whether our research, development, and clinical approaches will result in drugs that the FDA will consider safe for humans and effective for their intended uses. The FDA has substantial discretion in the drug approval process and may require us to conduct additional preclinical and clinical testing or to perform post-marketing studies. The approval process may also be delayed by changes in government regulation, future legislation, or administrative action or changes in FDA policy that occur prior to or during our regulatory review. Delays in obtaining regulatory approvals may:
| • | | delay commercialization of, and our ability to derive product revenues from, our product candidates; |
| • | | impose costly procedures on us; and |
| • | | diminish any competitive advantages that we may otherwise enjoy. |
Even if we comply with all FDA requests, the FDA may ultimately reject one or more of our NDAs or BLAs. We cannot be sure that we will ever obtain regulatory approval for any of our product candidates. Failure to obtain FDA approval for our product candidates will severely undermine our business by leaving us without a saleable product, and therefore without any potential revenue source, until another product candidate can be developed. There is no guarantee that we will ever be able to develop or acquire another product candidate or that we will obtain FDA approval if we are able to do so.
In foreign jurisdictions, we similarly must receive approval from applicable regulatory authorities before we can commercialize any drugs. Foreign regulatory approval processes generally include all of the risks associated with the FDA approval procedures described above.
Our product candidates are in various stages of clinical trials, which are very expensive and time-consuming. We cannot be certain when we will be able to submit an NDA or BLA to the FDA and any failure or delay in completing clinical trials for our product candidates could harm our business.
Our product candidates are in various stages of development and require extensive clinical testing. Notwithstanding our current clinical trial plans for each of our existing product
S-20
candidates, we may not be able to commence additional trials or see results from these trials within our anticipated timelines. As such, we cannot predict with any certainty if or when we might submit an NDA or BLA for regulatory approval of our product candidates or whether such an NDA or BLA will be accepted. Because we do not anticipate generating revenues unless and until we submit one or more NDAs or BLAs and thereafter obtain requisite FDA approvals, the timing of our NDA or BLA submissions and FDA determinations regarding approval thereof, will directly affect if and when we are able to generate revenues.
The results of our clinical trials may not support our product candidate claims.
Even if our clinical trials are completed as planned, we cannot be certain that their results will support approval of our product candidates. The FDA normally expects two randomized, well-controlled Phase 3 pivotal studies in support of approval of an NDA or BLA. Success in preclinical testing and early clinical trials does not ensure that later clinical trials will be successful, and we cannot be certain that the results of later clinical trials will replicate the results of prior clinical trials and preclinical testing. For example, despite positive findings in earlier clinical trials, our product candidate palifosfamide failed to meet the primary endpoints of the Phase 3 PICASSO 3 trial, causing us to suspend clinical development of palifosfamide in soft tissue sarcoma. The clinical trial process may fail to demonstrate that our product candidates are safe for humans and effective for the indicated uses. This failure would cause us to abandon a product candidate and may delay development of other product candidates. Any delay in, or termination of, our clinical trials will delay the submission of our NDAs or BLAs with the FDA and, ultimately, our ability to commercialize our product candidates and generate product revenues. In addition, our clinical trials involve small patient populations. Because of the small sample size, the results of these clinical trials may not be indicative of future results.
Our synthetic biology product candidates are based on a novel technology, which makes it difficult to predict the time and cost of product candidate development and subsequently obtaining regulatory approval. Currently, no gene therapy products have been approved in the United States and only one product has been approved in Europe.
We have recently focused our product research and development efforts on our synthetic biology product candidates under our Channel Agreement with Intrexon. These products, including DC-RTS-IL-12 and Ad-RTS-IL-12, are based on gene therapy technology. Due to the novelty of this medical technology, there can be no assurance that any development problems we experience in the future related to our synthetic biology platform will not cause significant delays or unanticipated costs, or that such development problems can be solved. We may also experience unanticipated problems or delays in expanding our manufacturing capacity or transferring our manufacturing process to commercial partners, which may prevent us from completing our clinical studies or commercializing our synthetic biology product candidates on a timely or profitable basis, if at all.
In addition, the clinical study requirements of the FDA, the EMA and other regulatory agencies and the criteria these regulators use to determine the safety and efficacy of a product candidate vary substantially according to the type, complexity, novelty and intended use and market of the potential products. The regulatory approval process for novel product candidates such as ours can be more expensive and take longer than for other, better known or extensively studied pharmaceutical or other product candidates. Currently, only one gene therapy product, UniQure’s Glybera, which received marketing authorization from the EMA in 2012, has been
S-21
approved in Europe but has not yet been launched for commercial sale, which makes it difficult to determine how long it will take or how much it will cost to obtain regulatory approvals for our product candidates in either the United States or Europe. Approvals by the EMA may not be indicative of what the FDA may require for approval.
Regulatory requirements governing gene and cell therapy products have changed frequently and may continue to change in the future. For example, the FDA has established the Office of Cellular, Tissue and Gene Therapies within its Center for Biologics Evaluation and Research, or CBER, to consolidate the review of gene therapy and related products, and the Cellular, Tissue and Gene Therapies Advisory Committee to advise CBER on its review. Gene therapy clinical studies conducted at institutions that receive funding for recombinant DNA research from the U.S. National Institutes of Health, or the NIH, are also subject to review by the NIH Office of Biotechnology Activities’ Recombinant DNA Advisory Committee, or the RAC. Although the FDA decides whether individual gene therapy protocols may proceed, the RAC review process can impede the initiation of a clinical trial, even if the FDA has reviewed the trial and approved its initiation. Conversely, the FDA can put an IND on clinical hold even if the RAC has provided a favorable review. Also, before a clinical trial can begin at an NIH-funded institution, that institution’s institutional review board, or IRB, and its Institutional Biosafety Committee will have to review the proposed clinical trial to assess the safety of the trial. In addition, adverse developments in clinical trials of gene therapy products conducted by others may cause the FDA or other regulatory bodies to change the requirements for approval of any of our product candidates.
These regulatory review committees and advisory groups and the new guidelines they promulgate may lengthen the regulatory review process, require us to perform additional studies, increase our development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of these treatment candidates or lead to significant post-approval limitations or restrictions. As we advance our synthetic biology product candidates, we will be required to consult with these regulatory and advisory groups, and comply with applicable guidelines. If we fail to do so, we may be required to delay or discontinue development of our product candidates. These additional processes may result in a review and approval process that is longer than we otherwise would have expected for oncology product candidates. Delay or failure to obtain, or unexpected costs in obtaining, the regulatory approval necessary to bring a potential product to market could decrease our ability to generate sufficient product revenue to maintain our business.
Because we are dependent upon clinical research institutions and other contractors for clinical testing and for research and development activities, the results of our clinical trials and such research activities are, to a certain extent, beyond our control.
We materially rely upon independent investigators and collaborators, such as universities and medical institutions, to conduct our preclinical and clinical trials under agreements with us. These collaborators are not our employees and we cannot control the amount or timing of resources that they devote to our programs. These investigators may not assign as great a priority to our programs or pursue them as diligently as we would if we were undertaking such programs ourselves. If outside collaborators fail to devote sufficient time and resources to our drug development programs, or if their performance is substandard, the approval of our FDA applications, if any, and our introduction of new products, if any, will be delayed. These collaborators may also have relationships with other commercial entities, some of whom may
S-22
compete with us. If our collaborators assist our competitors to our detriment, our competitive position would be harmed.
Our reliance on third parties to formulate and manufacture our product candidates exposes us to a number of risks that may delay the development, regulatory approval and commercialization of our products or result in higher product costs.
We do not have experience in drug formulation or manufacturing of drugs or biologics and do not intend to establish our own manufacturing facilities. Although we will work closely with and rely upon Intrexon on the manufacturing and scale-up of Intrexon product candidates, we lack the resources and expertise to formulate or manufacture our own product candidates. We currently are contracting for the manufacture of our product candidates. We intend to contract with one or more manufacturers to manufacture, supply, store, and distribute drug supplies for our clinical trials. If a product candidate we develop or acquire in the future receives FDA approval, we will rely on one or more third-party contractors or Intrexon to manufacture our products. Our anticipated future reliance on a limited number of third-party manufacturers exposes us to the following risks:
| • | | We may be unable to identify manufacturers on acceptable terms or at all because the number of potential manufacturers is limited and the FDA must approve any replacement contractor. This approval would require new testing and compliance inspections. In addition, a new manufacturer would have to be educated in, or develop substantially equivalent processes for, production of our products after receipt of FDA approval, if any. |
| • | | Our third-party manufacturers might be unable to formulate and manufacture our products in the volume and of the quality required to meet our clinical needs and commercial needs, if any. |
| • | | Our future contract manufacturers may not perform as agreed or may not remain in the contract manufacturing business for the time required to supply our clinical trials or to successfully produce, store, and distribute our products. |
| • | | Drug manufacturers are subject to ongoing periodic unannounced inspection by the FDA and corresponding state and foreign agencies to ensure strict compliance with current good manufacturing practices, or cGMP, and other government regulations and corresponding foreign standards. We do not have control over third-party manufacturers’ compliance with these regulations and standards. |
| • | | If any third-party manufacturer makes improvements in the manufacturing process for our products, we may not own, or may have to share, the intellectual property rights to the innovation. |
| • | | Our third-party manufacturers may not be able to comply with cGMP regulations or similar regulatory requirements outside the United States. Our failure, or the failure of our third-party manufacturers, to comply with applicable regulations could result in sanctions being imposed on us, including clinical holds, fines, injunctions, civil penalties, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of drug candidates or products, operating restrictions and criminal prosecutions, any of which could significantly and adversely affect supplies of our products. |
S-23
Each of these risks could delay our clinical trials, the approval, if any, of our product candidates by the FDA or the commercialization of our product candidates or result in higher costs or deprive us of potential product revenues.
Any drug candidate for which we obtain marketing approval could be subject to post-marketing restrictions or withdrawal from the market and we may be subject to penalties if we fail to comply with regulatory requirements or if we experience unanticipated problems with our products, when and if any of them are approved.
Any drug candidate for which we obtain marketing approval, along with the manufacturing processes, post-approval clinical data, labeling, advertising and promotional activities for such product, will be subject to continual requirements of and review by the FDA and other regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports, registration and listing requirements, cGMP requirements relating to manufacturing, quality control, quality assurance and corresponding maintenance of records and documents, requirements regarding the distribution of samples to physicians and recordkeeping. Even if marketing approval of a drug candidate is granted, the approval may be subject to limitations on the indicated uses for which the product may be marketed or to the conditions of approval, including the requirement to implement a risk evaluation and mitigation strategy, or REMS, which could include requirements for a restricted distribution system. If any of our drug candidates receives marketing approval, the accompanying label may limit the approved use of our drug, which could limit sales of the product.
The FDA may also impose requirements for costly post-marketing studies or clinical trials and surveillance to monitor the safety or efficacy of our approved products. The FDA closely regulates the post-approval marketing and promotion of drugs to ensure drugs are marketed only for the approved indications and in accordance with the provisions of the approved labeling. The FDA imposes stringent restrictions on manufacturers’ communications regarding off-label use and if we market our products outside of their approved indications, we may be subject to enforcement action for off-label marketing. Violations of the FDCA relating to the promotion of prescription drugs may lead to investigations alleging violations of federal and state health care fraud and abuse laws, as well as state consumer protection laws.
In addition, later discovery of previously unknown adverse events or other problems with our products, manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may yield various results, including:
| • | | litigation involving patients taking our drug; |
| • | | restrictions on such products, manufacturers or manufacturing processes; |
| • | | restrictions on the labeling or marketing of a product; |
| • | | restrictions on product distribution or use; |
| • | | requirements to conduct post-marketing studies or clinical trials; |
| • | | withdrawal of the products from the market; |
| • | | refusal to approve pending applications or supplements to approved applications that we submit; |
S-24
| • | | fines, restitution or disgorgement of profits or revenues; |
| • | | suspension or withdrawal of marketing approvals; |
| • | | damage to relationships with existing and potential collaborators; |
| • | | unfavorable press coverage and damage to our reputation; |
| • | | refusal to permit the import or export of our products; |
| • | | injunctions or the imposition of civil or criminal penalties. |
Noncompliance with similar European Union requirements regarding safety monitoring or pharmacovigilance can also result in significant financial penalties. Similarly, failure to comply with U.S. and foreign regulatory requirements regarding the development of products for pediatric populations and the protection of personal health information can also lead to significant penalties and sanctions.
Risks related to our ability to commercialize our product candidates
If we are unable either to create sales, marketing and distribution capabilities or enter into agreements with third parties to perform these functions, we will be unable to commercialize our product candidates successfully.
We currently have no marketing, sales, or distribution capabilities. If and when we become reasonably certain that we will be able to commercialize our current or future product candidates, we anticipate allocating resources to the marketing, sales and distribution of our proposed products in North America and in certain other countries; however, we cannot assure that we will be able to market, sell, and distribute our products successfully. Our future success also may depend, in part, on our ability to enter into and maintain collaborative relationships for such capabilities and to encourage the collaborator’s strategic interest in the products under development, and such collaborator’s ability to successfully market and sell any such products. Although we intend to pursue certain collaborative arrangements regarding the sale and marketing of certain of our product candidates, there are no assurances that we will be able to establish or maintain collaborative arrangements or, if we are able to do so, whether we would be able to conduct our own sales efforts. There can also be no assurance that we will be able to establish or maintain relationships with third-party collaborators or develop in-house sales and distribution capabilities. To the extent that we depend on third parties for marketing and distribution, any revenues we receive will depend upon the efforts of such third parties, and there can be no assurance that such efforts will be successful. In addition, there can also be no assurance that we will be able to market and sell our product candidates in the United States or overseas.
If we are not able to partner with a third party and are not successful in recruiting sales and marketing personnel or in building a sales and marketing infrastructure, we will have difficulty commercializing our product candidates, which would harm our business. If we rely on pharmaceutical or biotechnology companies with established distribution systems to market our products, we will need to establish and maintain partnership arrangements, and we may not be
S-25
able to enter into these arrangements on acceptable terms or at all. To the extent that we enter into co-promotion or other arrangements, any revenues we receive will depend upon the efforts of third parties that may not be successful and that will be only partially in our control.
If we cannot compete successfully for market share against other drug companies, we may not achieve sufficient product revenues and our business will suffer.
The market for our product candidates is characterized by intense competition and rapid technological advances. If a product candidate receives FDA approval, it will compete with a number of existing and future drugs and therapies developed, manufactured and marketed by others. Existing or future competing products may provide greater therapeutic convenience or clinical or other benefits for a specific indication than our products, or may offer comparable performance at a lower cost. If our products fail to capture and maintain market share, we may not achieve sufficient product revenues and our business will suffer.
We will compete against fully integrated pharmaceutical companies and smaller companies that are collaborating with larger pharmaceutical companies, academic institutions, government agencies and other public and private research organizations. Many of these competitors have products already approved or in development. In addition, many of these competitors, either alone or together with their collaborative partners, operate larger research and development programs or have substantially greater financial resources than we do, as well as significantly greater experience in:
| • | | developing drugs and biopharmaceuticals; |
| • | | undertaking preclinical testing and human clinical trials; |
| • | | obtaining FDA and other regulatory approvals of drugs and biopharmaceuticals; |
| • | | formulating and manufacturing drugs and biopharmaceuticals; and |
| • | | launching, marketing, and selling drugs and biopharmaceuticals. |
Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are more effective, have fewer or less severe side effects, are more convenient or are less expensive than any products that we may develop. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market. In addition, our ability to compete may be affected in many cases by insurers or other third-party payors seeking to encourage the use of generic products.
If physicians and patients do not accept and use our product candidates, our ability to generate revenue from sales of our products will be materially impaired.
Even if the FDA approves our product candidates, physicians and patients may not accept and use them. Acceptance and use of our products will depend upon a number of factors including:
| • | | perceptions by members of the healthcare community, including physicians, about the safety and effectiveness of our drugs; |
| • | | pharmacological benefit and cost-effectiveness of our products relative to competing products; |
| • | | availability of coverage and adequate reimbursement for our products from government or other healthcare payors; |
S-26
| • | | effectiveness of marketing and distribution efforts by us and our licensees and distributors, if any; and |
| • | | the price at which we sell our products. |
Because we expect sales of our current product candidates, if approved, to generate substantially all of our product revenues for the foreseeable future, the failure of a drug to find market acceptance would harm our business and could require us to seek additional financing in order to fund the development of future product candidates.
Our ability to generate product revenues will be diminished if our drugs do not obtain coverage or adequate reimbursement from payors.
Our ability to commercialize our drugs, alone or with collaborators, will depend in part on the extent to which coverage and reimbursement will be available from government and health administration authorities, private health maintenance organizations and health insurers and other third-party payors.
Patients who are prescribed medicine for the treatment of their conditions generally rely on third-party payors to reimburse all or part of the costs associated with their prescription drugs. Adequate coverage and reimbursement from governmental healthcare programs, such as Medicare and Medicaid, and commercial payors is critical to new product acceptance. Coverage decisions may depend upon clinical and economic standards that disfavor new drug products when more established or lower cost therapeutic alternatives are already available or subsequently become available. Even if we obtain coverage for our product candidates, the resulting reimbursement payment rates might not be adequate or may require co-payments that patients find unacceptably high. Patients are unlikely to use our product candidates unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of our product candidates.
In addition, the market for our product candidates for which we may receive regulatory approval will depend significantly on access to third-party payors’ drug formularies, or lists of medications for which third-party payors provide coverage and reimbursement. The industry competition to be included in such formularies often leads to downward pricing pressures on pharmaceutical companies. Also, third-party payors may refuse to include a particular branded drug in their formularies or otherwise restrict patient access to a branded drug when a less costly generic equivalent or other alternative is available.
Third-party payors, whether foreign or domestic, or governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. In addition, in the United States, no uniform policy of coverage and reimbursement for drug products exists among third-party payors. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that requires us to provide scientific and clinical support for the use of our products to each payor separately, with no assurance that approval will be obtained. If we are unable to obtain coverage of and adequate payment levels for our product candidates from third-party payors, physicians may limit how much or under what circumstances they will prescribe or administer them and patients may decline to purchase them. This in turn could affect our ability to successfully commercialize our products and impact our profitability, results of operations, financial condition, and future success.
S-27
In addition, in many foreign countries, particularly the countries of the European Union, the pricing of prescription drugs is subject to government control. In some non-U.S. jurisdictions, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, the EU provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A member state may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. We may face competition for our product candidates from lower-priced products in foreign countries that have placed price controls on pharmaceutical products. In addition, there may be importation of foreign products that compete with our own products, which could negatively impact our profitability.
Healthcare legislative reform measures may have a material adverse effect on our business and results of operations.
In both the United States and certain foreign jurisdictions, there have been a number of legislative and regulatory proposals in recent years that change the healthcare system in ways that could impact our future ability to sell our product candidates profitably. For example, the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 established a new Part D prescription drug benefit, which became effective January 1, 2006. Under the prescription drug benefit, Medicare beneficiaries can obtain prescription drug coverage from private sector plans that are permitted to limit the number of prescription drugs that are covered in each therapeutic category and class on their formularies. If any of our product candidates that are approved by the FDA are not widely included on the formularies of these plans, our ability to market our products to the Medicare population could suffer.
Furthermore, there have been and continue to be a number of initiatives at the federal and state level that seek to reduce healthcare costs. Most recently, in March 2010, President Obama signed into law the Patient Protection and Affordable Health Care Act, as amended by the Health Care and Education Reconciliation Act, or collectively the PPACA, which includes measures that significantly change the way healthcare is financed by both governmental and private insurers. Among the provisions of the PPACA of importance to the pharmaceutical industry are the following:
| • | | an annual, nondeductible fee on any entity that manufactures or imports certain branded prescription drugs and biologic agents, apportioned among these entities according to their market share in certain government healthcare programs, beginning in 2011; |
| • | | an increase in the statutory minimum rebates a manufacturer must pay under the Medicaid Drug Rebate Program, retroactive to January 1, 2010, to 23% and 13% of the average manufacturer price for most branded and generic drugs, respectively; |
| • | | an extension of manufacturers’ Medicaid rebate liability to covered drugs dispensed to individuals who are enrolled in Medicaid managed care organizations, effective March 23, 2010; |
| • | | new methodologies by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected, and for drugs that are line extensions; |
S-28
| • | | expansion of eligibility criteria for Medicaid programs by, among other things, allowing states to offer Medicaid coverage to additional individuals beginning in April 2010 and by adding new mandatory eligibility categories for certain individuals with income at or below 133% of the Federal Poverty Level beginning in 2014, thereby potentially increasing both the volume of sales and manufacturers’ Medicaid rebate liability; |
| • | | a new Medicare Part D coverage gap discount program, in which manufacturers must agree to offer 50% point-of-sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D, beginning in 2011; |
| • | | expansion of the entities eligible for discounts under the Public Health Service pharmaceutical pricing program, effective in January 2010; |
| • | | a new requirement to annually report drug samples that manufacturers and distributors provide to physicians, effective April 1, 2012; |
| • | | expansion of healthcare fraud and abuse laws, including the False Claims Act and the Anti-Kickback Statute, new government investigative powers, and enhanced penalties for noncompliance; |
| • | | a licensure framework for follow-on biologic products; |
| • | | a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research; |
| • | | creation of the Independent Payment Advisory Board which, beginning in 2014, will have authority to recommend certain changes to the Medicare program that could result in reduced payments for prescription drugs and those recommendations could have the effect of law even if Congress does not act on the recommendations; and |
| • | | establishment of a Center for Medicare Innovation at CMS to test innovative payment and service delivery models to lower Medicare and Medicaid spending, potentially including prescription drug spending beginning by January 1, 2011. |
In addition, other legislative changes have been proposed and adopted since the PPACA was enacted. In August 2011, President Obama signed into law the Budget Control Act of 2011, which, among other things, created the Joint Select Committee on Deficit Reduction to recommend proposals in spending reductions to Congress. The Joint Select Committee on Deficit Reduction did not achieve its targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, triggering the legislation’s automatic reductions to several government programs. These reductions include aggregate reductions to Medicare payments to providers of up to 2% per fiscal year, which went into effect on April 1, 2013. In January 2013, President Obama signed into law the American Taxpayer Relief Act of 2012, which, among other things, further reduced Medicare payments to several providers and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. The full impact of these new laws, as well as laws and other reform and cost containment measures that may be proposed and adopted in the future, remains uncertain, but may result in additional reductions in Medicare and other healthcare funding, which could have a material adverse effect on our future customers and accordingly, our ability to generate revenue, attain profitability, or commercialize our products.
S-29
If we fail to comply with federal and state healthcare laws, including fraud and abuse and health information privacy and security laws, we could face substantial penalties and our business, results of operations, financial condition and prospects could be adversely affected.
As a pharmaceutical company, even though we do not and will not control referrals of healthcare services or bill directly to Medicare, Medicaid or other third-party payors, certain federal and state healthcare laws and regulations pertaining to fraud and abuse and patients’ rights are and will be applicable to our business. We could be subject to healthcare fraud and abuse and patient privacy regulation by both the federal government and the states in which we conduct our business. The laws that may affect our ability to operate include:
| • | | the federal Anti-Kickback Statute, which constrains our marketing practices, educational programs, pricing policies, and relationships with healthcare providers or other entities, by prohibiting, among other things, soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce, or in return for, either the referral of an individual or the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
| • | | federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third-party payors that are false or fraudulent; |
| • | | the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created new federal criminal statutes that prohibit executing a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters; |
| • | | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, or HITECH, and its implementing regulations, which imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information; |
| • | | new requirements to report certain financial arrangements with physicians and teaching hospitals, as defined in the PPACA and its implementing regulations, including reporting any “transfer of value” made or distributed to teaching hospitals, prescribers, and other healthcare providers and reporting any ownership and investment interests held by physicians and their immediate family members and applicable group purchasing organizations during the preceding calendar year, with data collection required as of August 1, 2013 and reporting to the Centers for Medicare & Medicaid Services, or CMS, to be required by March 31, 2014 and by the 90th day of each subsequent calendar year; and |
| • | | state and foreign law equivalents of each of the above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payor, including commercial insurers; state laws that require pharmaceutical companies to comply with the industry’s voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government that otherwise restricts certain payments that may be made to healthcare providers; state laws that require drug manufacturers to report information related to payments and other transfer of value to physicians and other healthcare providers; and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
S-30
Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws. In addition, recent health care reform legislation has further strengthened these laws. For example, the PPACA, among other things, amends the intent requirement of the federal anti-kickback and criminal healthcare fraud statutes. A person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it. Moreover, the PPACA provides that the government may assert that a claim including items or services resulting from a violation of the federal anti-kickback statute constitutes a false or fraudulent claim for purposes of the False Claims Act.
To the extent that any of our product candidates is ultimately sold in a foreign country, we may be subject to similar foreign laws and regulations. If we or our operations are found to be in violation of any of the laws described above or any other governmental regulations that apply to us, we may be subject to penalties, including civil and criminal penalties, damages, fines, exclusion from participation in United States federal or state health care programs, and the curtailment or restructuring of our operations. Any penalties, damages, fines, curtailment or restructuring of our operations could materially adversely affect our ability to operate our business and our financial results. Although compliance programs can mitigate the risk of investigation and prosecution for violations of these laws, the risks cannot be entirely eliminated. Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. Moreover, achieving and sustaining compliance with applicable federal and state privacy, security and fraud laws may prove costly.
Our ability to use net operating loss carryforwards to reduce future tax payments may be limited or restricted.
We have generated significant net operating loss carryforwards, or NOLs, as a result of our incurrence of losses since inception. We generally are able to carry NOLs forward to reduce taxable income in future years. However, our ability to utilize the NOLs is subject to the rules of Section 382 of the Internal Revenue Code. Section 382 generally restricts the use of NOLs after an “ownership change.” An ownership change occurs if, among other things, the stockholders (or specified groups of stockholders) who own or have owned, directly or indirectly, 5% or more of a corporation’s common stock or are otherwise treated as 5% stockholders under Section 382 and the United States Treasury Department regulations promulgated thereunder increase their aggregate percentage ownership of that corporation’s stock by more than 50 percentage points over the lowest percentage of the stock owned by these stockholders over the applicable testing period. In the event of an ownership change, Section 382 imposes an annual limitation on the amount of taxable income a corporation may offset with NOL carry forwards. This annual limitation is generally equal to the product of the value of the corporation’s stock on the date of the ownership change, multiplied by the long-term tax-exempt rate published monthly by the Internal Revenue Service. Any unused annual limitation may be carried over to later years until the applicable expiration date for the respective NOL carry forwards. We may have experienced an “ownership change” within the meaning of Section 382 in the past and there can be no assurance that we have not experienced additional ownership changes, or that we will not experience an ownership change as a result of this offering. As a result, our NOLs may be subject to limitations and we may be required to pay taxes earlier and in larger amounts than would be the case if our NOLs were freely usable.
S-31
Our synthetic biology product candidates may face competition in the future from follow-on biologics.
With the enactment of the Biologics Price Competition and Innovation Act of 2009, or BPCIA, as part of the Patient Protection and Affordable Care Act, an abbreviated pathway for the approval of follow-on biological products was created. The new abbreviated regulatory pathway establishes legal authority for the FDA to review and approve biosimilar biologics, including the possible designation of a biosimilar as “interchangeable” with an existing brand product. Under the BPCIA, an application for a biosimilar product cannot be approved by the FDA until 12 years after the original branded product was approved under a BLA. The new law is complex and is only beginning to be interpreted and implemented by the FDA. As a result, its ultimate impact is subject to uncertainty, and could have a material adverse effect on the future commercial prospects for our biological products.
Risks related to our intellectual property
If we or our licensors fail to adequately protect or enforce our intellectual property rights or secure rights to patents of others, the value of our intellectual property rights would diminish and our ability to successfully commercialize our products may be impaired.
Our success, competitive position, and future revenues will depend in part on our ability and the abilities of our licensors to obtain and maintain patent protection for our products, methods, processes and other technologies, to preserve our trade secrets, to prevent third parties from infringing on our proprietary rights, and to operate without infringing the proprietary rights of third parties.
To date, we have exclusive rights to certain United States and foreign intellectual property with respect to our small molecule product candidates and with respect to the Intrexon technology, including the existing Intrexon product candidates. Under our Channel Agreement with Intrexon, Intrexon has the sole right to conduct and control the filings, prosecution and maintenance of the patents and patent applications licensed to us. Although under the agreement Intrexon has agreed to consider in good faith and consult with us regarding any comments we may have regarding these patents and patent applications, we cannot guarantee that our comments will be solicited or followed. Without direct control of the channel program patents and patent applications, we are dependent on Intrexon to keep us advised of prosecution, particularly in foreign jurisdictions where prosecution information may not be publicly available. We anticipate that we and Intrexon will file additional patent applications both in the United States and in other countries. However, we cannot predict or guarantee:
| • | | the degree and range of protection any patents will afford us against competitors, including whether third parties will find ways to invalidate or otherwise circumvent our patents; |
| • | | if and when patents will be issued; |
| • | | whether or not others will obtain patents claiming subject matter related to or relevant to our product candidates; or |
| • | | whether we will need to initiate litigation or administrative proceedings that may be costly whether we win or lose. |
The patent prosecution process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost, in a timely
S-32
manner, or in all jurisdictions. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. Moreover, in some circumstances, we do not have the right to control the preparation, filing and prosecution of patent applications, or to maintain the patents, covering technology that we license from third parties. We may also require the cooperation of our licensors in order to enforce the licensed patent rights, and such cooperation may not be provided. Therefore, these patents and applications may not be prosecuted and enforced in a manner consistent with the best interests of our business.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions and has in recent years been the subject of much litigation. In addition, the laws of foreign countries may not protect our rights to the same extent as the laws of the United States and we may fail to seek or obtain patent protection in all major markets. For example, European patent law restricts the patentability of methods of treatment of the human body more than United States law does. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all.
Changes in patent laws or in interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property or narrow the scope of our patent protection. In September 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law, resulting in a number of significant changes to United States patent law. These changes include provisions that affect the way patent applications will be prosecuted and may also affect patent litigation. In addition, the United States Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. This combination of events has created uncertainty with respect to the value of patents, once obtained, and with regard to our ability to obtain patents in the future. As the United States Patent and Trademark Office continues to implement the Leahy-Smith Act, and as the federal courts have the opportunity to interpret the Leahy-Smith Act, the laws and regulations governing patents, and the rules regarding patent procurement could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and patents that we might obtain in the future.
Certain technologies utilized in our research and development programs are already in the public domain. Moreover, a number of our competitors have developed technologies, filed patent applications or obtained patents on technologies, compositions and methods of use that are related to our business and may cover or conflict with our owned or licensed patent applications, technologies or product candidates. Such conflicts could limit the scope of the patents that we may be able to obtain or may result in the rejection of claims in our patent applications. Because patent applications in the United States and many foreign jurisdictions are typically not published until eighteen months after filing, or in some cases not at all, and because publications of discoveries in the scientific literature often lag behind actual discoveries, neither we nor our licensors can be certain that others have not filed or maintained patent applications for technology used by us or covered by our pending patent applications without our being aware of these applications. Therefore, we cannot know with certainty whether we were the first to make the inventions claimed in our owned patents or pending patent applications, or that we were the first to file for patent protection of such inventions, nor can we know whether those from whom
S-33
we license patents were the first to make the inventions claimed or were the first to file. As a result, the issuance, scope, validity, enforceability and commercial value of our patent rights are highly uncertain. Our pending and future patent applications may not result in patents being issued which protect our technology or products, in whole or in part, or which effectively prevent others from commercializing competitive technologies and products. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of our patents or narrow the scope of our patent protection. In addition, our own earlier filed patents and applications or those of Intrexon may limit the scope of later patents we obtain or may result in the rejection of claims in our later filed patent applications. If third parties filed patent applications or obtained patents on technologies, compositions and methods of use that are related to our business and that cover or conflict with our owned or licensed patent applications, technologies or product candidates, we may be required to challenge such protection, terminate or modify our programs impacted by such protection or obtain licenses from such third parties, which might not be available on acceptable terms, or at all.
Even if our owned and licensed patent applications issue as patents, they may not issue in a form that will provide us with any meaningful protection, prevent competitors from competing with us or otherwise provide us with any competitive advantage. Our competitors may be able to circumvent our owned or licensed patents by developing similar or alternative technologies or products in a non-infringing manner.
The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our owned and licensed patents may be challenged in the courts or patent offices in the United States and abroad. Such challenges may result in loss of exclusivity or freedom to operate or in patent claims being narrowed, invalidated or held unenforceable, in whole or in part, which could limit our ability to stop others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection of our technology and products. Given the amount of time required for the development, testing and regulatory review of new drug candidates, patents protecting such candidates might expire before or shortly after such candidates are commercialized. As a result, our owned and licensed patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
Our success also depends upon the skills, knowledge, and experience of our scientific and technical personnel, our consultants and advisors, as well as our licensors and contractors. To help protect our proprietary know-how and our inventions for which patents may be unobtainable or difficult to obtain, and to maintain our competitive position, we rely on trade secret protection and confidentiality agreements. To this end, it is our general policy to require our employees, consultants, advisors, and contractors to enter into agreements that prohibit the disclosure of confidential information and, where applicable, require disclosure and assignment to us of the ideas, developments, discoveries, and inventions important to our business. These agreements may not provide adequate protection for our trade secrets, know-how or other proprietary information in the event of any unauthorized use or disclosure or the lawful development by others of such information. Moreover, we may not be able to obtain adequate remedies for any breaches of these agreements. Our trade secrets may also be obtained by third parties by other
S-34
means, such as breaches of our physical or computer security systems. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them, or those to whom they communicate it, from using that technology or information to compete with us. If any of our trade secrets, know-how or other proprietary information is disclosed, the value of our trade secrets, know-how and other proprietary rights would be significantly impaired and our business and competitive position would suffer.
Third-party claims of intellectual property infringement would require us to spend significant time and money and could prevent us from developing or commercializing our products.
In order to protect or enforce patent rights, we, or Intrexon, may initiate patent infringement litigation against third parties. Similarly, we may be sued by others for patent infringement. We also may become subject to proceedings conducted in the United States Patent and Trademark Office, including interference proceedings to determine the priority or derivation of inventions, or post-grant review, inter partes review, or reexamination proceedings reviewing the patentability of our patented claims. In addition, any foreign patents that are granted may become subject to opposition, nullity, or revocation proceedings in foreign jurisdictions having such proceedings. The defense and prosecution, if necessary, of intellectual property actions are costly and divert technical and management personnel away from their normal responsibilities.
Our commercial success depends upon our ability, and the ability of our collaborators, to develop, manufacture, market and sell our drug candidates without infringing the proprietary rights of third parties. There is considerable intellectual property litigation in the biotechnology and pharmaceutical industries. While no such litigation has been brought against us and we have not been held by any court to have infringed a third party’s intellectual property rights, we cannot guarantee that our products or use of our products do not infringe third-party patents. It is also possible that we have failed to identify relevant third-party patents or applications. For example, applications filed before November 29, 2000 and certain applications filed after that date that will not be filed outside the United States remain confidential until patents issue. Patent applications in the United States and elsewhere are published approximately 18 months after the earliest filing, which is referred to as the priority date. Therefore, patent applications covering our products or technology could have been filed by others without our knowledge. Additionally, pending patent applications which have been published can, subject to certain limitations, be later amended in a manner that could cover our products or the use of our products.
Our research, development and commercialization activities, as well as any product candidates or products resulting from these activities, may infringe or be claimed to infringe patents or patent applications under which we do not hold licenses or other rights. Patents do not protect its owner from a claim of infringement of another owner’s patent. Therefore, our patent position cannot and does not provide any assurance that we are not infringing the patent rights of another.
The patent landscape in the field of synthetic biology, which we are pursuing under our Channel Agreement with Intrexon, is particularly complex. We are aware of numerous United States and foreign patents and pending patent applications of third parties that cover compositions,
S-35
methods of use and methods of manufacture of synthetic biology, including biotherapeutics involving the in vivo expression of human IL-12. In addition, there may be patents and patent applications in the field of which we are not aware. The technology we license from Intrexon is early-stage technology and we are just beginning the process of designing and developing products using this technology. Although we will seek to avoid pursuing the development of products that may infringe any patent claims that we believe to be valid and enforceable, we may fail to do so. Moreover, given the breadth and number of claims in patents and pending patent applications in the field of synthetic biology and the complexities and uncertainties associated with them, third parties may allege that we are infringing upon patent claims even if we do not believe such claims to be valid and enforceable.
If a claim for patent infringement is asserted, there can be no assurance that the resolution of the claim would permit us to continue marketing the relevant product on commercially reasonable terms, if at all. We may not have sufficient resources to bring these actions to a successful conclusion. If we do not successfully defend any infringement actions to which we become a party or are unable to have infringed patents declared invalid or unenforceable, we may have to pay substantial monetary damages, which can be tripled if the infringement is deemed willful, or be required to discontinue or significantly delay commercialization and development of the affected products.
Any legal action against us or our collaborators claiming damages and seeking to enjoin developmental or marketing activities relating to affected products could, in addition to subjecting us to potential liability for damages, require us or our collaborators to obtain licenses to continue to develop, manufacture, or market the affected products. Such a license may not be available to us on commercially reasonable terms, if at all.
An adverse determination in a proceeding involving our owned or licensed intellectual property may allow entry of generic substitutes for our products.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic maintenance fees on any issued patent are due to be paid to the U.S. PTO and foreign patent agencies in several stages over the lifetime of the patent. The U.S. PTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Noncompliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. In such an event, our competitors might be able to enter the market, which would have a material adverse effect on our business.
S-36
If we breach any of the agreements under which we license rights to products or technology from others, we could lose license rights that are material to our business or be subject to claims by our licensors.
We license rights to products and technology that are important to our business, and we expect to enter into additional licenses in the future. For instance, we have exclusively licensed patents and patent applications under our Channel Agreement with Intrexon. Under these agreements, we are subject to a range of commercialization and development, sublicensing, royalty, patent prosecution and maintenance, insurance and other obligations.
Any failure by us to comply with any of these obligations or any other breach by us of our license agreements could give the licensor the right to terminate the license in whole, terminate the exclusive nature of the license or bring a claim against us for damages. Any such termination or claim could have a material adverse effect on our financial condition, results of operations, liquidity or business. Even if we contest any such termination or claim and are ultimately successful, such dispute could lead to delays in the development or commercialization of potential products and result in time-consuming and expensive litigation or arbitration. On termination we may be required to license to the licensor any related intellectual property that we developed.
In addition, in certain cases, the rights licensed to us are rights of a third party licensed to our licensor. In such instances, if our licensors do not comply with their obligations under such licenses, our rights under our license agreements with our licensor may be adversely affected.
We may be subject to claims by third parties asserting that our employees or we have misappropriated their intellectual property, or claiming ownership of what we regard as our own intellectual property.
Many of our employees were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that these employees or we have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former employer. Litigation may be necessary to defend against these claims.
In addition, while it is our policy to require our employees and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own. Our and their assignment agreements may not be self-executing or may be breached, and we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property.
If we fail in prosecuting or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and be a distraction to management.
S-37
Other risks related to our company
Our stock price has been, and may continue to be, volatile.
The market price of our common stock has been highly volatile. The stock market from time to time experiences significant price and volume fluctuations unrelated to the operating performance of particular companies. In addition, factors such as fluctuations in our operating results, future sales of our common stock, announcements of the timing and amount of product sales, announcements of the status of development of our products, announcements of technological innovations or new therapeutic products by us or our competitors, announcements regarding collaborative agreements, laboratory or clinical trial results, government regulation, FDA determinations on the approval of a product candidate NDA submission, developments in patent or other proprietary rights, public concern as to the safety of drugs developed by us or others, changes in reimbursement policies, comments made by securities analysts and general market conditions may have a substantial effect on the market price of our common stock.
We are subject to Sarbanes-Oxley and the reporting requirements of federal securities laws, which can be expensive.
As a public reporting company, we are subject to the Sarbanes-Oxley Act of 2002, as well as to the information and reporting requirements of the Securities Exchange Act of 1934, as amended, or the Exchange Act, and other federal securities laws. As a result, we incur significant legal, accounting, and other expenses that we would not incur as a private company, including costs associated with our public company reporting requirements and corporate governance requirements. As an example of public reporting company requirements, we evaluate the effectiveness of disclosure controls and procedures and of our internal control over financing reporting in order to allow management to report on such controls. Sarbanes-Oxley generally requires that a public reporting company’s independent registered public accounting firm attest to the effectiveness of the company’s internal control over financial reporting as of the end of each fiscal year in the company’s Annual Report on Form 10-K. In addition, any updates to our finance and accounting systems, procedures and controls, which may be required as a result of our ongoing analysis of internal controls, or results of testing by our independent auditor, may require significant time and expense. As a company with limited accounting resources, a significant amount of management’s time and attention has been and will continue to be diverted from our business to ensure compliance with these regulatory requirements. This diversion of management’s time and attention may have a material adverse effect on our business, financial condition and results of operations.
Management is working to continuously monitor and improve internal controls and has set in place controls to mitigate the potential segregation of duties risk. In the event significant deficiencies or material weaknesses are identified in our internal control over financial reporting that we cannot remediate in a timely manner, or if we are unable to receive a positive attestation from our independent registered public accounting firm with respect to our internal controls over financial reporting, investors and others may lose confidence in the reliability of our financial statements and the trading price of our common stock and ability to obtain any necessary equity or debt financing could suffer. In addition, in the event that our independent registered public accounting firm is unable to rely on our internal controls over financial reporting in connection with its audit of our financial statements, and in the further event that it is unable to devise alternative procedures in order to satisfy itself as to the material accuracy of
S-38
our financial statements and related disclosures, we may be unable to file our periodic reports with the Securities and Exchange Commission, or SEC. This would likely have an adverse effect on the trading price of our common stock and our ability to secure any necessary additional equity or debt financing, and could result in the delisting of our common stock from the NASDAQ Capital Market, which would severely limit the liquidity of our common stock.
Anti-takeover provisions in our charter documents and under Delaware law may make an acquisition of us, which may be beneficial to our stockholders, more difficult.
Provisions of our amended and restated certificate of incorporation and bylaws, as well as provisions of Delaware law, could make it more difficult for a third party to acquire us, even if doing so would benefit our stockholders. These provisions authorize the issuance of “blank check” preferred stock that could be issued by our board of directors to increase the number of outstanding shares and hinder a takeover attempt, and limit who may call a special meeting of stockholders. In addition, Section 203 of the Delaware General Corporation Law generally prohibits a publicly-held Delaware corporation from engaging in a business combination with a party that owns at least 15% of its common stock unless the business combination is approved by the company’s board of directors before the person acquires the 15% ownership stake or later by its board of directors and two-thirds of its stockholders. In connection with our January 2011 issuance of shares of common stock to Intrexon in a private placement transaction, our board of directors waived the Section 203 prohibition with respect to a future business combination with Intrexon. However, the Stock Purchase Agreement governing such issuance contains a standstill provision that generally prohibits Intrexon from seeking, initiating, offering or proposing to effect such a transaction prior to January 6, 2014 without our inviting them to do so. Section 203 and this standstill provision could have the effect of delaying, deferring or preventing a change in control that our stockholders might consider to be in their best interests.
Because we do not expect to pay dividends, you will not realize any income from an investment in our common stock unless and until you sell your shares at profit.
We have never paid dividends on our capital stock and we do not anticipate that we will pay any dividends for the foreseeable future. Accordingly, any return on an investment in us will be realized, if at all, only when you sell shares of our common stock.
Risks related to this offering
Management will have broad discretion as to the use of the proceeds from this offering, and we may not use the proceeds effectively.
Our management will have broad discretion in the application of the net proceeds from this offering and could spend the proceeds in ways that do not improve our results of operations or enhance the value of our common stock. Our failure to apply these funds effectively could have a material adverse effect on our business, delay the development of our product candidates and cause the price of our common stock to decline.
You will experience immediate and substantial dilution in the net tangible book value per share of the common stock you purchase.
Since the price per share of our common stock being offered is substantially higher than the net tangible book value per share of our common stock, you will suffer substantial dilution in the net
S-39
tangible book value of the common stock you purchase in this offering. Based on the public offering price of $3.50 per share, and after deducting underwriting discounts and commissions and estimated offering expenses payable by us, and based on a net tangible book value of our common stock of $0.35 per share as of September 30, 2013, if you purchase shares of common stock in this offering, you will suffer immediate and substantial dilution of $2.72 per share in the net tangible book value of common stock. See the section entitled “Dilution” below for a more detailed discussion of the dilution you will incur if you purchase common stock in this offering.
You may experience future dilution as a result of future equity offerings.
In order to raise additional capital, we may in the future offer additional shares of our common stock or other securities convertible into or exchangeable for our common stock at prices that may not be the same as the price per share in this offering. We cannot assure you that we will be able to sell shares or other securities in any other offering at a price per share that is equal to or greater than the price per share paid by investors in this offering, and investors purchasing shares or other securities in the future could have rights superior to existing stockholders, including investors who purchase shares of common stock in this offering. The price per share at which we sell additional shares of our common stock or securities convertible into common stock in future transactions may be higher or lower than the price per share in this offering.
If securities and/or industry analysts fail to continue publishing research about our business, if they change their recommendations adversely or if our results of operations do not meet their expectations, our stock price and trading volume could decline.
The trading market for our common stock will be influenced by the research and reports that industry or securities analysts publish about us or our business. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, we could lose visibility in the financial markets, which in turn could cause our stock price or trading volume to decline. In addition, it is likely that in some future period our operating results will be below the expectations of securities analysts or investors. If one or more of the analysts who cover us downgrade our stock, or if our results of operations do not meet their expectations, our stock price could decline.
Our stock price is volatile and may decline regardless of our operating performance, and you may not be able to resell your shares at or above the price at which you purchased such shares.
The market price for our common stock is volatile and may fluctuate significantly in response to a number of factors, most of which we cannot control, including:
| • | | price and volume fluctuations in the overall stock market; |
| • | | market conditions or trends in our industry or the economy as a whole; |
| • | | changes in operating performance and stock market valuations of other biopharmaceutical companies generally, or those that develop and commercialize cancer drugs in particular; |
| • | | the financial projections we may provide to the public, any changes in these projections or our failure to meet these projections; |
S-40
| • | | changes in financial estimates or ratings by any securities analysts who follow our common stock, our failure to meet these estimates or failure of those analysts to initiate or maintain coverage of our common stock; |
| • | | the public’s response to press releases or other public announcements by us or third parties, including our filings with the SEC and announcements relating to product development, litigation and intellectual property impacting us or our business; |
| • | | the sustainability of an active trading market for our common stock; |
| • | | future sales of our common stock by our executive officers, directors and significant stockholders; |
| • | | announcements of mergers or acquisition transactions; |
| • | | our inclusion or deletion from certain stock indices; |
| • | | announcements of medical innovations or new products by our competitors; |
| • | | announcements of changes in our senior management; |
| • | | other events or factors, including those resulting from war, incidents of terrorism, natural disasters or responses to these events; and |
| • | | changes in accounting principles. |
In addition, the stock markets, and in particular the NASDAQ Capital Market, have experienced extreme price and volume fluctuations that have affected and continue to affect the market prices of equity securities of many biopharmaceutical companies. Stock prices of many biopharmaceutical companies have fluctuated in a manner unrelated or disproportionate to the operating performance of those companies. In the past, stockholders have instituted securities class action litigation following periods of market volatility. If we were involved in securities litigation, we could incur substantial costs and our resources and the attention of management could be diverted from our business.
Our principal stockholders, executive officers and directors have substantial control over the company, which may prevent you and other stockholders from influencing significant corporate decisions and may harm the market price of our common stock.
As of September 30, 2013, our executive officers, directors and holders of five percent or more of our outstanding common stock, beneficially owned, in the aggregate, 30.0% of our outstanding common stock. These stockholders may have interests that conflict with our other stockholders and, if acting together, have the ability to influence the outcome of matters submitted to our stockholders for approval, including the election and removal of directors and any merger, consolidation or sale of all or substantially all of our assets. Accordingly, this concentration of ownership may harm the market price of our common stock by:
| • | | delaying, deferring or preventing a change in control; |
| • | | impeding a merger, consolidation, takeover or other business combination involving us; or |
| • | | discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of us. |
S-41
Special note regarding forward-looking statements
This prospectus supplement and the accompanying prospectus contain, and the documents incorporated by reference herein and therein and any free writing prospectus that we have authorized for use in connection with this offering may contain, forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Exchange Act. These statements relate to future events or to our future operating or financial performance and involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements to be materially different from any future results, performances or achievements expressed or implied by the forward-looking statements. Forward-looking statements may include, but are not limited to statements about:
| • | | the progress, timing and results of preclinical and clinical trials involving our drug candidates; |
| • | | the progress of our research and development programs; |
| • | | our plans or others’ plans to conduct future clinical trials or research and development efforts; |
| • | | the risk that final trial data may not support interim analysis of the viability of our drug candidates; |
| • | | our plans and expectations regarding partnering our drug candidates; |
| • | | the benefits to be derived from relationships with our collaborators; |
| • | | the receipt or anticipated receipt of regulatory clearances and approvals; |
| • | | estimates of the potential markets for our drug candidates; |
| • | | our ability to adequately protect our intellectual property rights; |
| • | | the use of proceeds from this offering; |
| • | | our estimates of future revenues and profitability; |
| • | | our estimates regarding our capital requirements and our ability to control costs; and |
| • | | our need for additional funding and the period through which we anticipate our resources will sufficient to fund operations. |
In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “could,” “would,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “projects,” “predicts,” “potential” and similar expressions intended to identify forward-looking statements. These statements reflect our current views with respect to future events and are based on assumptions and subject to risks and uncertainties. Given these uncertainties, you should not place undue reliance on these forward-looking statements. We discuss many of these risks in greater detail under the heading “Risk factors” in this prospectus supplement and in our SEC filings. Also, these forward-looking statements represent our estimates and assumptions only as of the date of the document containing the applicable statement.
You should read this prospectus supplement, the accompanying prospectus, the documents we have filed with the SEC that are incorporated by reference and any free writing prospectus that we have authorized for use in connection with this offering completely and with the
S-42
understanding that our actual future results may be materially different from what we expect. We qualify all of the forward-looking statements in the foregoing documents by these cautionary statements.
Unless required by law, we undertake no obligation to update or revise any forward-looking statements to reflect new information or future events or developments. Thus, you should not assume that our silence over time means that actual events are bearing out as expressed or implied in such forward-looking statements.
S-43
Use of proceeds
We estimate that the net proceeds to us from the sale of our common stock offered hereby will be approximately $46.7 million, or approximately $53.8 million if the underwriters exercise in full their option to purchase additional shares of common stock, after deducting the underwriting discounts and commissions and our estimated offering expenses.
We currently intend to use the net proceeds from this offering for the overall development of our drug candidates, including our synthetic biology candidates and our small molecule candidates, and for general corporate and working capital purposes.
The amount and timing of these expenditures will depend on a number of factors, including the progress of our research and development efforts, the progress of any partnering efforts, technological advances and the competitive environment for our product candidates. Accordingly, our management will have broad discretion in the application of these proceeds, and investors will be relying on the judgment of our management with regard to the use of these proceeds. Pending application of the net proceeds as described above, we intend to invest the proceeds in investment grade interest bearing instruments.
S-44
Dilution
Our net tangible book value as of September 30, 2013 was approximately $29.5 million, or $0.35 per share. Net tangible book value per share is determined by dividing our total tangible assets, less total liabilities, by the number of shares of our common stock outstanding as of September 30, 2013. Dilution in net tangible book value per share represents the difference between the amount per share paid by purchasers of shares of common stock in this offering and the net tangible book value per share of our common stock immediately after this offering.
After taking into account the sale by us of 14,300,000 shares of our common stock in this offering at the public offering price of $3.50 per share, less the underwriting discounts and commissions and estimated offering expenses payable by us, our as adjusted net tangible book value as of September 30, 2013 would have been approximately $76.2 million, or $0.78 per share. This would represent an immediate increase in net tangible book value of $0.43 per share to existing stockholders and in immediate dilution of $2.72 per share to investors purchasing our common stock in this offering at the public offering price. The following table illustrates this dilution on a per share basis:
| | | | | | | | |
Public offering price per share | | | | | | $ | 3.50 | |
Net tangible book value per share as of September 30, 2013 | | $ | 0.35 | | | | | |
Increase per share attributable to investors purchasing our common stock in this offering | | $ | 0.43 | | | | | |
As adjusted net tangible book value per share as of September 30, 2013, after giving effect to this offering | | | | | | $ | 0.78 | |
Dilution in net tangible book value per share to investors purchasing our common stock in this offering | | | | | | $ | 2.72 | |
| |
If the underwriters exercise in full their option to purchase 2,145,000 additional shares of common stock at the public offering price of $3.50 per share, the pro forma as adjusted net tangible book value after this offering would be $0.83 per share, representing an increase in net tangible book value of $0.48 per share to existing stockholders and immediate dilution in net tangible book value of $2.67 per share to investors purchasing our common stock in this offering at the public offering price.
S-45
The amounts above are based on 83,681,580 shares of common stock outstanding as of September 30, 2013 and assume no exercise of outstanding options or warrants since that date. The number of common stock expected to be outstanding after this offering excludes:
| • | | 5,730,169 shares of our common stock issuable upon the exercise of stock options outstanding as of September 30, 2013, having a weighted average exercise price of $3.73 per share; |
| • | | 1,565,070 shares of our common stock available as of September 30, 2013 for future issuance pursuant to our 2003 Stock Option Plan and our 2012 Equity Incentive Plan; and |
| • | | 10,392,387 shares of our common stock issuable upon the exercise of outstanding warrants as of September 30, 2013 with a weighted-average exercise price of $3.57 per share. |
To the extent options or warrants outstanding as of September 30, 2013 have been or may be exercised or other shares have been issued, there may be further dilution to investors.
S-46
Material United States federal income and estate tax
consequences for certain non-U.S. holders
The following discussion describes the material U.S. federal income and estate tax consequences of the acquisition, ownership and disposition of our common stock acquired in this offering by a Non-U.S. Holder (as defined below). This discussion does not address all aspects of U.S. federal income and estate taxes, does not discuss the potential application of the 3.8% Medicare tax on net investment income, and does not deal with non-U.S., state and local consequences that may be relevant to Non-U.S. Holders in light of their particular circumstances, nor does it address U.S. federal tax consequences other than income and estate taxes. Rules different from those described below may apply to certain Non-U.S. Holders that are subject to special treatment under the Internal Revenue Code of 1986, as amended (the “Code”), including, without limitation, banks, thrifts and other financial institutions, insurance companies, tax-exempt organizations, broker-dealers and traders in securities, U.S. expatriates, regulated investment companies, real estate investment trusts, “controlled foreign corporations,” “passive foreign investment companies,” corporations that accumulate earnings to avoid U.S. federal income tax, persons that hold our common stock as part of a “straddle,” “hedge,” “conversion transaction,” “synthetic security,” integrated investment or other risk reduction strategy, partnerships and other pass-through entities, and investors in such pass-through entities or entities that are treated as disregarded entities for U.S. federal income tax purposes (regardless of their places of organization or formation), persons subject to the alternative minimum tax, persons that hold or receive shares of our common stock pursuant to the exercise of any employee stock option or otherwise as compensation, persons that own, or are deemed to own, more than 5% of our outstanding common stock (except to the extent specifically set forth below), and persons deemed to sell shares of our common stock under the constructive sale provisions of the Code. Such Non-U.S. Holders are urged to consult their own tax advisors to determine the U.S. federal, state, local and other tax consequences that may be relevant to them. Furthermore, the discussion below is based upon the provisions of the Code, and Treasury regulations, rulings and judicial decisions thereunder as of the date hereof, and such authorities may be repealed, revoked or modified, perhaps retroactively, so as to result in U.S. federal income or estate tax consequences different from those discussed below. No ruling has been or will be sought from the United States Internal Revenue Service (the “IRS”) with respect to the statements made and the conclusions reached in the following discussion, and there can be no assurance that the IRS will not take a contrary position regarding the tax consequences of the acquisition, ownership or disposition of shares of our common stock, or that any such contrary position would not be sustained by a court. This discussion is limited to Non-U.S. Holders who hold our common stock as a capital asset within the meaning of Section 1221 of the Code (generally, property held for investment).
The following discussion is for general information only and is not tax advice for any Non-U.S. Holders under their particular circumstances.Persons considering the purchase of our common stock should consult their own tax advisors concerning the U.S. federal income and estate tax consequences of acquiring, owning and disposing of our common stock in light of their particular situations as well as any consequences arising under the laws of any other taxing jurisdiction, including any state, local or non-U.S. tax consequences and the possible application of tax treaties that might change the general provisions discussed below.
Except as otherwise described in the discussion of estate tax below, a “Non-U.S. Holder” is a beneficial owner of our common stock that is not a U.S. Holder. A “U.S. Holder” means a
S-47
beneficial owner of our common stock that is for U.S. federal income tax purposes (i) an individual who is a citizen or resident of the United States, (ii) a corporation or other entity treated as a corporation created or organized in or under the laws of the United States, any state thereof or the District of Columbia, (iii) an estate the income of which is subject to U.S. federal income taxation regardless of its source or (iv) a trust if it (x) is subject to the primary supervision of a court within the United States and one or more U.S. persons have the authority to control all substantial decisions of the trust or (y) has a valid election in effect under applicable U.S. Treasury regulations to be treated as a U.S. person.
If a partnership (including any entity or arrangement treated as a partnership for U.S. federal income tax purposes) acquires our common stock, the tax treatment of a partner in the partnership will generally depend upon the status of the partner and the activities of the partnership. Persons who are partners of partnerships holding our common stock are urged to consult their tax advisors. For purposes of the following discussion, entities treated as partnerships or disregarded entities for U.S. federal income tax purposes are not addressed by this discussion and are, therefore, not considered to be Non-U.S. Holders for the purposes of this discussion.
Distributions
Subject to the discussion below, distributions, if any, of cash or property made on our common stock to a Non-U.S. Holder of our common stock, to the extent made out of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles), generally will constitute dividends for U.S. tax purposes and generally will be subject to withholding tax at a 30% rate or such lower rate as may be specified by an applicable income tax treaty. To obtain a reduced rate of withholding under a treaty, a Non-U.S. Holder generally will be required to provide us or our paying agent with a properly-executed IRS Form W-8BEN, or other appropriate form, certifying the Non-U.S. Holder’s entitlement to benefits under that treaty. Such certificate must be provided prior to the payment of dividends and must be updated periodically. Treasury regulations provide special rules to determine whether, for purposes of determining the applicability of a tax treaty, dividends paid to a Non-U.S. Holder that is an entity should be treated as paid to the entity or to those holding an interest in that entity. If a Non-U.S. Holder holds stock through a financial institution or other agent acting on the holder’s behalf, the holder will be required to provide appropriate documentation to such agent. The holder’s agent will then be required to provide certification to us or our paying agent, either directly or through other intermediaries, of the Non-U.S. Holder’s entitlement to treaty benefits. If you are eligible for a reduced rate of U.S. federal withholding tax pursuant to an applicable income tax treaty but do not timely provide us or our paying agent with the required certification, you should consult with your tax advisor to determine whether you may obtain a refund of any excess amounts withheld by timely filing an appropriate claim for refund with the IRS.
We generally are not required to withhold tax on dividends paid to a Non-U.S. Holder that are effectively connected with the Non-U.S. Holder’s conduct of a trade or business within the United States (and, if required by an applicable income tax treaty, are attributable to a permanent establishment that such Non-U.S. Holder maintains in the United States) if a properly-executed IRS Form W-8ECI, or other appropriate form, stating that the dividends are so connected, is furnished to us or our paying agent (or, if stock is held through a financial institution or other agent, to such agent). In general, such effectively connected dividends will be subject to U.S. federal income tax, on a net income basis generally at regular graduated rates,
S-48
unless a specific treaty exemption applies. A corporate Non-U.S. Holder receiving effectively connected dividends may also be subject to an additional “branch profits tax”, which is imposed, under certain circumstances, at a rate of 30% (or such lower rate as may be specified by an applicable treaty) on the corporate Non-U.S. Holder’s effectively connected earnings and profits, subject to certain adjustments.
To the extent distributions on our common stock, if any, exceed our current and accumulated earnings and profits, they will first reduce your adjusted basis in our common stock as a non-taxable return of capital, but not below zero, and then any excess will be treated as gain from the sale of stock and taxed in the same manner as gain realized from a sale or other disposition of common stock as described in the next section.
Gain on disposition of our common stock
Subject to the discussion below regarding backup withholding and foreign accounts, a Non-U.S. Holder generally will not be subject to U.S. federal income tax with respect to gain realized on a sale or other disposition of our common stock unless (i) the gain is effectively connected with a trade or business of such holder in the United States (and, if required by an applicable income tax treaty, is attributable to a permanent establishment that such Non-U.S. Holder maintains in the United States), (ii) in the case of Non-U.S. Holders who are nonresident alien individuals, such individuals are present in the United States for 183 or more days in the taxable year of the disposition and certain other conditions are met, or (iii) we are or have been a “United States real property holding corporation” within the meaning of Code Section 897(c)(2) at any time within the shorter of the five-year period preceding such disposition or such holder’s holding period.
If you are a Non-U.S. Holder described in (i) above, you will be required to pay tax on the net gain derived from the sale at regular graduated U.S. federal income tax rates, unless a specific treaty exemption applies, and corporate Non-U.S. Holders described in (i) above may be subject to the additional branch profits tax at a 30% rate or such lower rate as may be specified by an applicable income tax treaty. If you are an individual Non-U.S. Holder described in (ii) above, you will be required to pay a flat 30% tax on the gain derived from the sale, which tax may be offset by U.S. source capital losses if you have timely filed tax returns with respect to such losses (even though you are not considered a resident of the United States). With respect to (iii) above, in general, we would be a United States real property holding corporation if interests in U.S. real estate comprised (by fair market value) at least half of our business assets. We believe that we are not, and do not anticipate becoming, a United States real property holding corporation; however, there can be no assurance that we will not become a U.S. real property holding corporation in the future. Even if we are treated as a U.S. real property holding corporation, gain realized by a Non-U.S. Holder on a disposition of our common stock will not be subject to U.S. federal income tax so long as (1) the Non-U.S. Holder owned directly, indirectly and constructively, no more than five percent of our common stock at all times within the shorter of (a) the five year period preceding the disposition or (b) the holder’s holding period and (2) our common stock is regularly traded on an established securities market. There can be no assurance that our common stock will continue to qualify as regularly traded on an established securities market. If gain realized by you on the sale of our common stock is taxable because we are a U.S. real property holding corporation and your ownership of our common stock exceeded the 5% threshold in the period noted above, you will be taxed on such disposition generally in the same manner applicable to U.S. persons.
S-49
Information reporting requirements and backup withholding
Generally, we or certain financial middlemen must report information to the IRS with respect to any dividends we pay on our stock including the amount of any such dividends, the name and address of the recipient, and the amount, if any, of tax withheld. A similar report is sent to the holder to whom any such dividends are paid. Pursuant to tax treaties or certain other agreements, the IRS may make its reports available to tax authorities in the country in which the Non-U.S. Holder resides or is established.
Proceeds from a disposition of our stock and dividends paid by us (or our paying agents) to a Non-U.S. Holder may also be subject to United States backup withholding. United States backup withholding generally will not apply to a Non-U.S. Holder who provides a properly-executed IRS Form W-8BEN, or W-8ECI or otherwise establishes an exemption. The current backup withholding rate is 28%. Notwithstanding the foregoing, backup withholding may apply if either we or our paying agent has actual knowledge, or reason to know, that the holder of our common stock is a U.S. person that is not an exempt recipient.
Backup withholding is not an additional tax. Rather, the tax liability of persons subject to backup withholding will be reduced by the amount of tax withheld. If withholding results in an overpayment of taxes, a refund or credit may generally be obtained with respect to such backup withholding, provided that the required information is timely furnished to the IRS.
Legislation relating to foreign accounts
A U.S. federal withholding tax of 30% may apply to dividends and the gross proceeds of a disposition of our common stock paid to a foreign financial institution (as specifically defined by applicable rules), including when the foreign financial institution holds our common stock on behalf of a Non-U.S. Holder, unless such institution enters into an agreement with the U.S. government to withhold on certain payments and to collect and provide to the U.S. tax authorities substantial information regarding U.S. account holders of such institution (which includes certain equity holders of such institution, as well as certain account holders that are foreign entities with U.S. owners), or otherwise qualifies for an exemption from these rules. Foreign financial institutions located in jurisdictions that have an intergovernmental agreement with the United States governing these withholding and reporting requirements may be subject to different rules. This U.S. federal withholding tax of 30% will also apply to dividends and the gross proceeds of a disposition of our common stock paid to a non-financial foreign entity (as specifically defined by applicable rules), unless such entity provides the withholding agent with either a certification that it does not have any substantial direct or indirect U.S. owners or provides information regarding direct and indirect U.S. owners of the entity, or otherwise qualifies for an exemption from these rules. The withholding tax described above will not apply if the foreign financial institution or non-financial foreign entity otherwise qualifies for an exemption from the rules. Under certain circumstances, a Non-U.S. Holder might be eligible for refunds or credits of such taxes. Holders are encouraged to consult with their own tax advisors regarding the possible implications of the legislation on their investment in our common stock.
The withholding provisions described above will generally apply to payments of dividends made on or after July 1, 2014 and to payments of gross proceeds from a sale or other disposition of common stock on or after January 1, 2017.
S-50
Federal estate tax
An individual who at the time of death is not a citizen or resident of the United States and who is treated as the owner of, or has made certain lifetime transfers of, an interest in our common stock will be required to include the value thereof in his or her taxable estate for U.S. federal estate tax purposes, and may be subject to U.S. federal estate tax unless an applicable estate tax treaty provides otherwise. The test for whether an individual is a resident of the United States for federal estate tax purposes differs from the test used for U.S. federal income tax purposes. Some individuals, therefore, may be “Non-U.S. Holders” for U.S. federal income tax purposes, but not for U.S. federal estate tax purposes, and vice versa.
THE PRECEDING DISCUSSION OF UNITED STATES FEDERAL INCOME TAX CONSIDERATIONS IS FOR GENERAL INFORMATION ONLY. IT IS NOT TAX ADVICE. EACH PROSPECTIVE INVESTOR SHOULD CONSULT ITS OWN TAX ADVISOR REGARDING THE TAX CONSEQUENCES OF PURCHASING, HOLDING AND DISPOSING OF OUR COMMON STOCK, INCLUDING THE CONSEQUENCES OF ANY PROPOSED CHANGE IN APPLICABLE LAW.
S-51
Underwriting
We are offering the shares of common stock described in this prospectus supplement through a number of underwriters. J.P. Morgan Securities LLC is acting as sole book-running manager and representative of the underwriters. We have entered into an underwriting agreement with the underwriters. Subject to the terms and conditions of the underwriting agreement, we have agreed to sell to the underwriters, and each underwriter has severally agreed to purchase, at the public offering price less the underwriting discounts and commissions set forth on the cover page of this prospectus supplement, the number of shares of common stock listed next to its name in the following table:
| | | | |
| Name | | Number
of shares | |
| |
J.P. Morgan Securities LLC | | | 11,440,000 | |
JMP Securities LLC | | | 1,430,000 | |
Griffin Securities, Inc. | | | 1,430,000 | |
| | | | |
Total | | | 14,300,000 | |
| |
The underwriters are committed to purchase all shares of common stock offered by us if they purchase any shares. The underwriting agreement also provides that if an underwriter defaults, the purchase commitments of non-defaulting underwriters may also be increased or the offering may be terminated.
The underwriters propose to offer the shares of common stock directly to the public at the public offering price set forth on the cover page of this prospectus supplement and to certain dealers at that price less a concession not in excess of $0.1260 per share. After the public offering of the shares, the offering price and other selling terms may be changed by the underwriters. Sales of shares made outside of the United States may be made by affiliates of the underwriters.
The underwriters have an option to buy up to 2,145,000 additional shares of common stock from us to cover sales of shares by the underwriters which exceed the number of shares specified in the table above. The underwriters have 30 days from the date of this prospectus supplement to exercise this option. If any shares are purchased with this option, the underwriters will purchase shares in approximately the same proportion as shown in the table above. If any additional shares of common stock are purchased, the underwriters will offer the additional shares on the same terms as those on which the shares are being offered.
The underwriting fee is equal to the public offering price per share of common stock less the amount paid by the underwriters to us per share of common stock. The underwriting fee is $0.21 per share. The following table shows the per share and total underwriting discounts and commissions to be paid to the underwriters assuming both no exercise and full exercise of the underwriters’ option to purchase additional shares.
| | | | | | | | |
| | | Without option
exercise | | | With option
exercise | |
| |
Per Share | | $ | 0.21 | | | $ | 0.21 | |
Total | | $ | 3,003,000 | | | $ | 3,453,450 | |
| |
We estimate that the total expenses of this offering, including registration, filing and listing fees, printing fees and legal and accounting expenses, but excluding the underwriting discounts and commissions, will be approximately $300,000.
S-52
A prospectus in electronic format may be made available on the websites maintained by one or more underwriters, or selling group members, if any, participating in the offering. The underwriters may agree to allocate a number of shares to underwriters and selling group members for sale to their online brokerage account holders. Internet distributions will be allocated by the underwriters to selling group members that may make Internet distributions on the same basis as other allocations.
We have agreed that we will not (i) offer, pledge, announce the intention to sell, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase or otherwise transfer or dispose of, directly or indirectly, or file with SEC a registration statement under the Securities Act relating to, any shares of our common stock or securities convertible into or exercisable or exchangeable for any shares of our common stock, or publicly disclose the intention to make any offer, sale, pledge, disposition or filing, or (ii) enter into any swap or other arrangement that transfers, in whole or in part, any portion of the economic consequences associated with the ownership of any shares of common stock or any such other securities (regardless of whether any of these transactions are to be settled by the delivery of shares of common stock or such other securities, in cash or otherwise), in each case without the prior written consent of J.P. Morgan Securities LLC on behalf of the underwriters for a period of 90 days after the date of this prospectus supplement.
Notwithstanding the foregoing, if (1) during the last 17 days of the 90-day restricted period, we issue an earnings release or material news or a material event relating to our company occurs; or (2) prior to the expiration of the 90-day restricted period, we announce that we will release earnings results during the 16-day period beginning on the last day of the 90-day period, the restrictions described above shall continue to apply until the expiration of the 18-day period beginning on the issuance of the earnings release or the occurrence of the material news or material event unless J.P. Morgan Securities LLC waives, in writing, such an extension. The foregoing sentence will not apply if, within three business days prior to the 15th calendar day before the last day of the 90-day period, we deliver a certificate, signed by our Chief Financial Officer or Chief Executive Officer, certifying on our behalf that the shares of common stock are “actively traded securities” (as defined in Regulation M) and that we meet the requirements set forth in paragraph (a)(1) of Rule 139 under the Securities Act in the manner contemplated by NASD Conduct Rule 2711(f)(4).
The restrictions described in the immediately preceding paragraph do not apply, subject to certain conditions, to the following:
| • | | the sale of shares of common stock pursuant to the underwriting agreement; |
| • | | the issuance of shares of our common stock pursuant to our stock plans; |
| • | | the issuance of shares of our common stock upon the exercise of warrants outstanding on the date hereof; |
| • | | the filing by us of any Registration Statement on Form S-8 or a successor form thereto; or |
| • | | the issuance of shares of common stock or securities convertible into or exercisable or exchangeable for shares of common stock representing in the aggregate no more than 5% of our issued and outstanding shares of common stock immediately following the completion of this offering, which may be sold only to one or more counterparties in connection with the consummation of a strategic partnership, joint venture, collaboration, merger or the |
S-53
| | acquisition or license of any business products or technology, so long as the recipients of such securities sign and deliver a lock-up agreement. |
Our directors and executive officers have entered into lock-up agreements with the underwriters prior to the commencement of this offering pursuant to which each of these persons or entities, with limited exceptions described below, for a period of 90 days after the date of this prospectus, may not, without the prior written consent of J.P. Morgan Securities LLC on behalf of the underwriters, (i) offer, pledge, announce the intention to sell, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, or otherwise transfer or dispose of, directly or indirectly, any shares of our common stock or any securities convertible into or exercisable or exchangeable for our common stock (including, without limitation, common stock or such other securities which may be deemed to be beneficially owned by such persons or entities in accordance with the rules and regulations of the SEC and securities which may be issued upon exercise of a stock option or warrant), (ii) enter into any swap or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of the common stock or such other securities, whether any such transaction described in clause (i) or (ii) above is to be settled by delivery of common stock or such other securities, in cash or otherwise, or (iii) make any demand for or exercise any right with respect to the registration of any shares of our common stock or any security convertible into or exercisable or exchangeable for our common stock. Notwithstanding the foregoing, if (1) during the last 17 days of the 90-day restricted period, we issue an earnings release or material news or a material event relating to our company occurs; or (2) prior to the expiration of the 90-day restricted period, we announce that we will release earnings results during the 16-day period beginning on the last day of the 90-day period, the restrictions described above shall continue to apply until the expiration of the 18-day period beginning on the issuance of the earnings release or the occurrence of the material news or material event unless J.P. Morgan Securities LLC waives, in writing, such an extension. The foregoing sentence will not apply if, within three business days prior to the 15th calendar day before the last day of the 90-day period, we deliver a certificate, signed by our Chief Financial Officer or Chief Executive Officer, certifying on our behalf that the shares of common stock are “actively traded securities” (as defined in Regulation M) and that we meet the requirements set forth in paragraph (a)(1) of Rule 139 under the Securities Act in the manner contemplated by NASD Conduct Rule 2711(f)(4).
The restrictions described in the immediately preceding paragraph do not apply, subject to certain conditions, to the following:
| • | | the transfer of shares of our common stock or other securities as abona fide gift or gifts; |
| • | | the transfer of share of our common stock withheld for tax purposes in connection with the vesting and distribution of our common stock; |
| • | | transactions relating to shares of common stock or other securities acquired in open market transactions after the completion of this offering; |
| • | | the transfer of shares of common stock or any security convertible into or exercisable or exchangeable for common stock to the immediate family of the signatory, to a trust the beneficiaries of which are exclusively the signatory and/or a member or members of the immediate family of the signatory or to any corporation, partnership, limited liability company or other entity all of the beneficial ownership interests of which are held exclusively by the signatory and/or a member or members of the immediate family of the signatory; |
S-54
| • | | if the signatory is an entity, the transfer of shares of our common stock or other securities to an affiliate (as defined in Rule 12b-2 of the Exchange Act) in a transaction not involving a distribution for value; |
| • | | the distribution of shares of our common stock or other securities to members, partners or stockholders of the signatory; |
| • | | the transfer of shares of common stock or any security convertible or exercisable or exchangeable for common stock upon death by will or intestate succession; or |
| • | | the entry into any trading plan established pursuant to Rule 10b5-1 of the Exchange Act, provided that no sales or other dispositions may occur under such plan until the expiration of the 90-day restricted period and that no filing or other public announcement, whether under the Exchange Act or otherwise, shall be required or shall be made by the signatory or the Company in connection with the trading plan during such restricted period and, before the trading plan is established, the Company shall have provided to J.P. Morgan Securities LLC on behalf of the underwriters written confirmation that no such filing or public announcement shall be required or shall be made by the Company in connection with the trading plan during such period. |
We have agreed to indemnify the underwriters against certain liabilities, including liabilities under the Securities Act.
Our common stock is listed on the NASDAQ Capital Market under the symbol “ZIOP.”
In connection with this offering, the underwriters may engage in stabilizing transactions, which involve making bids for, purchasing and selling shares of common stock in the open market for the purpose of preventing or retarding a decline in the market price of the common stock while this offering is in progress. These stabilizing transactions may include making short sales of the common stock, which involves the sale by the underwriters of a greater number of shares of common stock than they are required to purchase in this offering, and purchasing shares of common stock on the open market to cover positions created by short sales. Short sales may be “covered” shorts, which are short positions in an amount not greater than the underwriters’ option referred to above, or may be “naked” shorts, which are short positions in excess of that amount. The underwriters may close out any covered short position either by exercising their option, in whole or in part, or by purchasing shares in the open market. In making this determination, the underwriters will consider, among other things, the price of shares available for purchase in the open market compared to the price at which the underwriters may purchase shares through the option. A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of the common stock in the open market that could adversely affect investors who purchase in this offering. To the extent that the underwriters create a naked short position, they will purchase shares in the open market to cover the position.
The underwriters have advised us that, pursuant to Regulation M under the Exchange Act, they may also engage in other activities that stabilize, maintain or otherwise affect the price of the common stock, including the imposition of penalty bids. This means that if the representative of the underwriters purchase common stock in the open market in stabilizing transactions or to cover short sales, the representative can require the underwriters that sold those shares as part of this offering to repay the underwriting discount received by them.
S-55
These activities may have the effect of raising or maintaining the market price of the common stock or preventing or retarding a decline in the market price of the common stock, and, as a result, the price of the common stock may be higher than the price that otherwise might exist in the open market. If the underwriters commence these activities, they may discontinue them at any time. The underwriters may carry out these transactions on the NASDAQ Capital Market, in the over-the-counter market or otherwise.
In addition, in connection with this offering certain of the underwriters (and selling group members) may engage in passive market making transactions in our common stock on the NASDAQ Capital Market prior to the pricing and completion of this offering. Passive market making consists of displaying bids on the NASDAQ Capital Market no higher than the bid prices of independent market makers and making purchases at prices no higher than these independent bids and effected in response to order flow. Net purchases by a passive market maker on each day are generally limited to a specified percentage of the passive market maker’s average daily trading volume in the common stock during a specified period and must be discontinued when such limit is reached. Passive market making may cause the price of our common stock to be higher than the price that otherwise would exist in the open market in the absence of these transactions. If passive market making is commenced, it may be discontinued at any time.
If you purchase shares of common stock offered in this prospectus, you may be required to pay stamp taxes and other charges under the laws and practices of the country of purchase, in addition to the offering price listed on the cover page of this prospectus.
Other than in the United States, no action has been taken by us or the underwriters that would permit a public offering of the securities offered by this prospectus in any jurisdiction where action for that purpose is required. The securities offered by this prospectus may not be offered or sold, directly or indirectly, nor may this prospectus or any other offering material or advertisements in connection with the offer and sale of any such securities be distributed or published in any jurisdiction, except under circumstances that will result in compliance with the applicable rules and regulations of that jurisdiction. Persons into whose possession this prospectus comes are advised to inform themselves about and to observe any restrictions relating to the offering and the distribution of this prospectus. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities offered by this prospectus in any jurisdiction in which such an offer or a solicitation is unlawful.
Certain of the underwriters and their affiliates may provide from time to time in the future certain commercial banking, financial advisory, investment banking and other services for us and our affiliates in the ordinary course of their business, for which they may receive customary fees and commissions. In addition, from time to time, certain of the underwriters and their affiliates may effect transactions for their own account or the account of customers, and hold on behalf of themselves or their customers, long or short positions in our debt or equity securities or loans.
S-56
Selling restrictions
European economic area
In relation to each Member State of the European Economic Area which has implemented the Prospectus Directive (each, a Relevant Member State) an offer to the public of any securities which are the subject of the offering contemplated by this prospectus may not be made in that Relevant Member State, except that an offer to the public in that Relevant Member State of any securities may be made at any time under the following exemptions under the Prospectus Directive, if they have been implemented in that Relevant Member State:
| • | | to any legal entity which is a qualified investor as defined in the Prospectus Directive; |
| • | | to fewer than 100 or, if the Relevant Member State has implemented the relevant provision of the 2010 PD Amending Directive, 150, natural or legal persons (other than qualified investors as defined in the Prospectus Directive), as permitted under the Prospectus Directive, subject to obtaining the prior consent of the joint book-running managers for any such offer; or |
| • | | in any other circumstances falling within Article 3(2) of the Prospectus Directive, provided that no such offer of the securities shall result in a requirement for the publication by us or any underwriter of a prospectus pursuant to Article 3 of the Prospectus Directive. |
For the purposes of this provision, the expression an “offer to the public” in relation to any securities in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and any securities to be offered so as to enable an investor to decide to purchase any securities, as the same may be varied in that Member State by any measure implementing the Prospectus Directive in that Member State, the expression “Prospectus Directive” means Directive 2003/71/EC (and amendments thereto, including the 2010 PD Amending Directive, to the extent implemented in the Relevant Member State), and includes any relevant implementing measure in the Relevant Member State, and the expression “2010 PD Amending Directive” means Directive 2010/73/EU.
United Kingdom
Each underwriter has represented and agreed that:
| • | | it has only communicated or caused to be communicated and will only communicate or cause to be communicated an invitation or inducement to engage in investment activity within the meaning of Section 21 of the Financial Services and Markets Act 2000, or the FSMA, received by it in connection with the issue or sale of the securities in circumstances in which Section 21(1) of the FSMA does not apply to us; and |
| • | | it has complied and will comply with all applicable provisions of the FSMA with respect to anything done by it in relation to the securities in, from or otherwise involving the United Kingdom. |
Switzerland
This document, as well as any other material relating to the shares of our common stock, which are the subject of the offering contemplated by this prospectus, does not constitute an issue prospectus pursuant to Article 652a of the Swiss Code of Obligations. The shares will not be listed
S-57
on the SIX Swiss Exchange and, therefore, the documents relating to the shares, including, but not limited to, this document, do not claim to comply with the disclosure standards of the listing rules of SIX Swiss Exchange and corresponding prospectus schemes annexed to the listing rules of the SIX Swiss Exchange.
The shares are being offered in Switzerland by way of a private placement, i.e., to a small number of selected investors only, without any public offer and only to investors who do not purchase the shares with the intention to distribute them to the public. The investors will be individually approached by us from time to time.
This document, as well as any other material relating to the shares, is personal and confidential and does not constitute an offer to any other person. This document may only be used by those investors to whom it has been handed out in connection with the offering described herein and may neither directly nor indirectly be distributed or made available to other persons without our express consent. It may not be used in connection with any other offer and shall in particular not be copied and/or distributed to the public in (or from) Switzerland.
S-58
Legal matters
Cooley LLP, Boston, Massachusetts, will pass upon the validity of the issuance of the common stock offered hereby. The underwriters are being represented by Latham & Watkins LLP, Costa Mesa, California.
Experts
The financial statements and the effectiveness of internal control over financial reporting incorporated in this Prospectus by reference to the Annual Report on Form 10-K for the year ended December 31, 2012, have been audited by McGladrey LLP (formerly McGladrey & Pullen, LLP), an independent registered public accounting firm, as stated in their report incorporated by reference herein (which report expressed an unqualified opinion and includes an explanatory paragraph relating to the Company’s ability to continue as a going concern, and also noted the reliance on other auditors for cumulative amounts from September 9, 2013 (date of inception) through December 31, 2009), and have been so incorporated in reliance upon such reports and upon the authority of such firm as experts in accounting and auditing.
The statements of operations, changes in preferred stock and stockholders’ equity (deficit) and cash flows for the period from September 9, 2003 (date of inception) through December 31, 2009, incorporated by reference in this Prospectus but not separately presented in the Annual Report on Form 10-K for the year ended December 31, 2012, have been audited by Caturano and Company, P.C. (whose name has since been changed to Caturano and Company, Inc.), an independent registered public accounting firm, as stated in their report incorporated by reference herein, and is incorporated by reference in reliance upon such report and upon the authority of such firm as experts in accounting and auditing.
Where you can find more information
We are a reporting company and file annual, quarterly and current reports, proxy statements and other information with the SEC. You may read and copy these reports, proxy statements and other information at the SEC’s public reference room at 100 F Street, N.E., Washington, D.C. 20549 or at the SEC’s other public reference facilities. Please call the SEC at 1-800-SEC-0330 for more information about the operation of the public reference rooms. You can request copies of these documents by writing to the SEC and paying a fee for the copying costs. In addition, the SEC maintains an Internet site athttp://www.sec.gov that contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC. Our SEC filings are available on the SEC’s Internet site. We maintain a website athttp://www.ziopharm.com. Information found on, or accessible through, our website is not a part of, and is not incorporated into, this prospectus supplement or the accompanying prospectus, and you should not consider it part of this prospectus supplement or part of the accompanying prospectus.
S-59
Incorporation of information by reference
We are allowed to incorporate by reference information contained in documents that we file with the SEC. This means that we can disclose important information to you by referring you to those documents and that the information in this prospectus supplement is not complete and you should read the information incorporated by reference for more detail. Information in this prospectus supplement supersedes information incorporated by reference that we filed with the SEC prior to the date of this prospectus supplement, while information that we file later with the SEC will automatically update and supersede the information in this prospectus supplement.
We incorporate by reference the documents listed below and any future filings we will make with the SEC under Section 13(a), 13(c), 14 or 15(d) of the Exchange Act from the date of this prospectus supplement but prior to the termination of the offering of the securities covered hereby (other than Current Reports or portions thereof furnished under Item 2.02 or 7.01 of Form 8-K):
| • | | Annual Report on Form 10-K for the fiscal year ended December 31, 2012, filed on March 18, 2013 (as amended by Amendment No. 1 on Form 10-K/A filed on June 28, 2013); |
| • | | Quarterly Reports on Form 10-Q for the quarters ended March 31, 2013, June 30, 2013, and September 30, 2013, filed on May 7, 2013, August 8, 2013, and October 22, 2013, respectively; |
| • | | Current Reports on Form 8-K filed on January 8, 2013, January 31, 2013, February 12, 2013, March 13, 2013, March 26, 2013; April 5, 2013, May 7, 2013, May 29, 2013, May 31, 2013, June 21, 2013, August 8, 2013 and September 27, 2013; |
| • | | The description of our common stock set forth in the registration statement on Form 8-A registering our common stock under Section 12 of the Exchange Act, which was filed with the SEC on September 20, 2006, including any amendments or reports filed for purposes of updating such description. |
We will provide to each person, including any beneficial owner, to whom a prospectus is delivered a copy of any or all of the documents that are incorporated by reference in this prospectus supplement but not delivered with this prospectus, including exhibits that are specifically incorporated by reference in such documents. You may request a copy of such documents at no cost, by writing or telephoning us at the following address or telephone number:
ZIOPHARM Oncology, Inc.
One First Avenue, Parris Building 34, Navy Yard Plaza
Boston, Massachusetts 02129
Attention: Chief Executive Officer
Telephone: (617) 259-1970
S-60
Prospectus
$150,000,000

ZIOPHARM Oncology, Inc.
Common Stock
Preferred Stock
Warrants
Debt Securities
From time to time, we may offer and sell any combination of common stock, preferred stock, warrants and debt securities, with a total initial offering price of up to $150,000,000. We may also offer common stock or preferred stock upon conversion of debt securities, common stock upon conversion of preferred stock, or common stock, preferred stock or debt securities upon the exercise of warrants.
We will provide the specific terms of these offerings and securities in one or more supplements to this prospectus. We may also authorize one or more free writing prospectuses to be provided to you in connection with these offerings. The prospectus supplement and any related free writing prospectus may also add, update or change information contained in this prospectus. You should carefully read this prospectus, the applicable prospectus supplement and any related free writing prospectus, as well as any documents incorporated by reference, before buying any of the securities being offered.
Our common stock is listed on the NASDAQ Capital Market under the symbol “ZIOP.” On November 4, 2011, the closing price of our common stock, as reported on the NASDAQ Capital Market, was $4.68.
This prospectus may not be used to consummate a sale of any securities unless accompanied by a prospectus supplement.
The securities may be sold directly by us to investors, through agents designated from time to time or to or through underwriters or dealers, on a continuous or delayed basis. For additional information on the methods of sale, you should refer to the section entitled “Plan of Distribution” in this prospectus. If any agents or underwriters are involved in the sale of any securities with respect to which this prospectus is being delivered, the names of such agents or underwriters and any applicable fees, commissions, discounts and over-allotment options will be set forth in a prospectus supplement. The price to the public of such securities and the net proceeds that we expect to receive from such sale will also be set forth in a prospectus supplement.
Investing in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described under the heading “Risk Factors” contained in the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other documents that are incorporated by reference into this prospectus.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved these securities or determined if this prospectus is truthful or complete. A representation to the contrary is a criminal offense.
The date of this Prospectus is November 16, 2011.
Table of contents
i
About this prospectus
This prospectus is part of a registration statement that we filed with the Securities and Exchange Commission, or the Commission, using a “shelf” registration process. Under this shelf registration process, from time to time, we may sell any combination of the securities described in this prospectus in one or more offerings, up to a total dollar amount of $150,000,000. This prospectus provides you with a general description of the securities we may offer. Each time we offer and sell securities under this prospectus, we will provide a prospectus supplement that will contain more specific information about the terms of the applicable offering. We may also authorize one or more free writing prospectuses to be provided to you that may contain material information relating to these offerings. The prospectus supplement, and any related free writing prospectus that we may authorize to be provided to you, may also add, update or change the information contained in this prospectus or in the documents incorporated by reference into this prospectus. We urge you to carefully read this prospectus, any applicable prospectus supplement and any related free writing prospectus, together with the information incorporated herein by reference as described under the headings “Where You Can Find More Information” and “Incorporation of Information by Reference” before buying any of the securities being offered.THIS PROSPECTUS MAY NOT BE USED TO CONSUMMATE A SALE OF SECURITIES UNLESS IT IS ACCOMPANIED BY A PROSPECTUS SUPPLEMENT.
You should rely only on the information contained in, or incorporated by reference into, this prospectus or any applicable prospectus supplement, along with the information contained in any related free writing prospectus that we have authorized for use in connection with a specific offering. We have not authorized anyone to provide you with different information. No dealer, salesperson or other person is authorized to give any information or to represent anything not contained in this prospectus, any applicable prospectus supplement or any related free writing prospectus that we may authorize to be provided to you. You must not rely on any unauthorized information or representation. This prospectus is an offer to sell only the securities offered hereby, but only under circumstances and in jurisdictions where it is lawful to do so. The information in this prospectus, any applicable prospectus supplement or any related free writing prospectus is accurate only as of the date on the front of the document, and any information we have incorporated by reference is accurate only as of the date of the document incorporated by reference, regardless of the time of delivery of this prospectus, any applicable prospectus supplement or any related free writing prospectus, or any sale of a security. Our business, financial condition, results of operations and prospects may have changed since those dates.
ii
Prospectus summary
This summary highlights selected information contained elsewhere in this prospectus or incorporated by reference into this prospectus, and does not contain all of the information that you need to consider in making your investment decision. You should carefully read the entire prospectus, the applicable prospectus supplement and any related free writing prospectus, including the risks of investing in our securities discussed under the heading “Risk Factors” contained in the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other documents that are incorporated by reference into this prospectus. You should also carefully read the information incorporated by reference into this prospectus, including our financial statements, and the exhibits to the registration statement of which this prospectus is a part. Unless otherwise indicated, “ZIOPHARM,” our “Company,” “we,” “us,” “our” and similar terms refer to ZIOPHARM Oncology, Inc.
Our company
ZIOPHARM Oncology, Inc. is a biopharmaceutical company that is seeking to develop and commercialize a diverse portfolio of in-licensed cancer drugs that can address unmet medical needs. Our principal focus has been on the licensing and development of proprietary small molecule drug candidates that are related to cancer therapeutics already on the market or in development and that can be administered by intravenous, or IV, and/or oral dosing. Our clinical programs for our small molecule candidates include palifosfamide (Zymafos TM or ZIO-201) darinaparsin (Zinapar TM or ZIO-101) and indibulin (Zybulin TM or ZIO-301). We are also pursuing the development of novel DNA-based biotherapeutics in the field of cancer pursuant to a partnering arrangement with Intrexon Corporation, or Intrexon. Under the arrangement, we obtained rights to Intrexon’s effector platform for use in the field of oncology, which includes two existing clinical stage product candidates, ZIN-CTI-001 (or DC-RTS-IL-12 + AL) and ZIN-ATI-001 (or Ad-RTS-IL-12 + AL). We plan to leverage Intrexon’s synthetic biology platform to develop products to stimulate key pathways used by the body’s immune system to inhibit the growth and metastasis of cancers, adding significantly to our small molecule drug development portfolio utilizing our global capabilities to translate science to the patient setting. Descriptions of our current clinical development plans for palifosfamide, darinaparsin, indibulin, ZIN-CTI-001 and ZIN-ATI-001 are set forth below. More detailed descriptions of these product candidates and our clinical development plans for each are also set forth in the our Annual Report on Form 10-K for the fiscal year ended December 31, 2010, incorporated by reference herein, and in other reports that we file from time to time with the Securities and Exchange Commission, or the Commission.
We believe that our strategy will result in expedited drug development programs for product candidates with a cost of manufacturing that, upon successful commercialization, would help to address changing worldwide product reimbursement requirements. We are currently in Phase 1, 2, and/or Phase 3 studies for our product candidates with a particular emphasis on completing a global palifosfamide pivotal Phase 3 trial to support registration in combination with doxorubicin in the front-line setting of metastatic soft tissue sarcoma.
Product candidates
ZIO-101, Darinaparsin, Zinapar TM
Darinaparsin is a novel mitochondrial-targeted agent (organic arsenic) in development with IV and oral administrations. Phase 1 testing of the IV form of darinaparsin in solid tumors and
1
hematological cancers was completed and we reported clinical activity and, importantly, a safety profile from these studies as predicted by preclinical results. We subsequently completed Phase 2 studies in advanced myeloma, primary liver cancer and in certain other hematological cancers. At the May 2009 annual meeting of the American Society of Clinical Oncology, or ASCO, we reported favorable results from the trial with IV-administered darinaparsin in lymphoma, particularly peripheral T-cell lymphoma, or PTCL. With focus on the relapsed setting, a Phase 1 study of darinaparsin in combination with the treatment regimen called “CHOP” in the front-line setting of PTCL was ended. A Phase 1 trial with an oral form of darinaparsin is currently in progress in a solid tumor Phase 1 study. Data from the completion of this study will guide further study in solid tumors and/or PTCL. We have obtained Orphan Drug Designation for darinaparsin in the United States and Europe for the treatment of PTCL.
ZIO-201, Palifosfamide, Zymafos TM
Palifosfamide is a novel DNA cross-linker (stabilized active metabolite of ifosfamide) in class with bendemustine, ifosfamide, and cyclophosphamide and currently in development with IV administration (oral in late preclinical). Following Phase 1 study, we completed Phase 2 testing of the IV form of palifosfamide as a single agent to treat advanced sarcoma. In both Phase 1 and Phase 2 testing, palifosfamide has been administered without the “uroprotectant” mesna as is required with ifosfamide, and the toxicities associated with other ifosfamide metabolites, acrolein and chloroacetaldehyde, have not been observed. We reported clinical activity of palifosfamide when used alone in the Phase 2 study addressing advanced sarcoma. Following review of preclinical combination studies, we initiated a Phase 1 dose escalation study of palifosfamide in combination with doxorubicin, primarily in patients with soft tissue sarcoma. We reported favorable results and safety profile from this study at ASCO’s 2009 annual meeting. In light of reported favorable Phase 2 single agent clinical activity data and with the combination being well tolerated in the Phase 1 trial, we initiated a Phase 2 randomized controlled trial, entitled PICASSO, in the second half of 2008 to compare doxorubicin plus palifosfamide to doxorubicin alone in patients with front- and second-line metastatic or unresectable soft tissue sarcoma. The study generated positive top line interim data in 2009. Upon successfully reaching a pre-specified efficacy milestone and following safety and efficacy data review by the Data Committee, sarcoma experts, and our Medical Advisory Board, we elected to suspend enrollment in the trial in October 2009. We subsequently presented further positive interim data from the trial at the 15th Annual Connective Tissue Oncology Society meeting held in November 2009 and again at the 2010 ASCO annual meeting in June 2010, where the presentation was selected for “Best of ASCO.” In July 2010, we announced the initiation of a worldwide registration trial on a protocol design developed through a FDA End-of-Phase 2 meeting and the Special Protocol Assessment, or SPA, process. Although we did engage in the SPA process, we, with guidance from the United States Food and Drug Administration, or FDA, elected to initiate the trial without having obtained SPA agreement from the FDA. The Phase 3 trial is in front-line metastatic soft tissue sarcoma, entitled PICASSO 3, and is an international, randomized, double-blinded, placebo-controlled trial with a targeted enrollment of 424 patients. The study is designed to evaluate the safety and efficacy of palifosfamide administered with doxorubicin compared with doxorubicin administered with placebo, with no cross-over between the arms. Progression-free survival is the primary endpoint for accelerated approval, with overall survival as the primary endpoint for full approval PICASSO 3 has no interim data analysis, although the trial is monitored by a Data Monitoring Committee, or DMC, of outside, independent expects. The DMC has met twice to review trial data for safety and futility and on both occasions has recommended trial
2
continuation. Orphan Drug Designation for palifosfamide has been obtained in both the United States and the European Union for the treatment of soft tissue sarcomas.
A Phase 1 trial is also ongoing with palifosfamide in combination with etoposide and carboplatin to determine appropriate safety for initiating a potentially pivotal, adaptive trial in front-line small-cell lung cancer, or SCLC. An oral form of palifosfamide has been the subject of preclinical studies necessary for an Investigational New Drug, or IND, application to support commencing Phase 1 study. Based on an initial review, the FDA has requested that we repeat an animal study, that is currently underway, in order to support the planned Phase 1 protocol.
ZIO-301, Indibulin, Zybulin TM
Indibulin is a novel orally administered tubulin binding agent. Phase 1 study as a single agent in patients with advanced solid tumors has been completed. We have reported clinical activity at well-tolerated doses using a continuous dosing scheme without the development of clinically relevant peripheral neuropathy. Following encouraging preclinical results obtained with indibulin in combination with other chemotherapies, two Phase 1 combination studies were initiated with Tarceva TM and Xeloda TM , respectively. The favorable activity and safety profile of oral indibulin with oral Xeloda TM was reported at ASCO’s annual meeting in May 2009. In all studies, a maximum tolerated dose, or MTD, was not established. Preclinical work with our consultant, Dr. Larry Norton, established a dosing schedule to enhance activity and reduce toxicity, which is presently five days on drug and nine days off in a Phase 1 study in late stage metastatic breast cancer. In light of the lack of establishing an MTD and the need to administer many capsules several times a day, we have recently modified the dosage form to administer once a day dosing in the Phase 1 trial.
ZIN-CTI-001 (or DC-RTS-IL-12 + AL) and ZIN-ATI-001 (or Ad-RTS-IL-12 + AL)
We are also pursuing the development of novel DNA-based therapeutics in the field of cancer pursuant to an exclusive channel partnership with Intrexon. The partnership includes two existing clinical-stage product candidates. ZIN-CTI-001 is in a Phase 1b trial in the United States and employs intratumoral injection of modified dendritic cells from each patient and oral dosing of an activator ligand to turn onin vivo expression of interleukin-12, or IL-12. ZIN-CTI-001 uses a RheoSwitch Therapeutic System® (RTS) to control the timing and level of transgene expression for gene and cell therapy. The RTS technology functions as a “gene switch” for the regulated expression of human IL-12 in the patients’ dendritic cells, which are transduced with a replication deficient adenoviral vector carrying the IL-12 gene under the control of the RTS and in this study injected intratumorally for the treatment of patients with stage III or IV melanoma. The binding of the small molecule activator to the fusion proteins of RTS is intended to regulate the timing and level of IL-12 expression. In the absence of the activator ligand, the level of IL-12 is below detectable levels.
The activator ligand has been the subject of a number of preclinical, safety and pharmacology studies under FDA and International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use guidelines. Preclinical studies in the B16 mouse melanoma model consistently induced regression of established melanoma lesions, both in those directly injected and those elsewhere in the body. Preclinical studies have shown DC-RTS-IL-12, in combination with an activator ligand, to have strong activity against a broad array of cancers, including brain, colon, renal, and pancreatic cancers and melanoma.
3
A Phase 1a clinical study of the activator ligand was conducted in 65 healthy volunteers, with the two most common side effects being dysgeusia (impairment of taste) and throat irritation. A subsequent Phase 1b trial, which is ongoing in patients with advanced melanoma, has been amended to study efficacy and immunological and biological effects in addition to safety with cohort-based dose escalation of the activator ligand during repeated treatment cycles. Initial positive clinical results from the Phase 1b trial were presented at the June 2011 ASCO annual meeting. The trial enrolled ten patients (median age 61) with unresectable Stage III or IV melanoma. Among eight evaluable patients, partial or complete regression of injected and some uninjected lesions was observed by computed axial tomography scans in three patients, with one patient having a RECIST PR of >11 months and three patients demonstrating stable disease by RECIST, for an overall disease control rate of 50%. Treatment was generally well tolerated, and maximum tolerated dose has not yet been reached. Adverse events were mild to moderate, with one to two patients each experiencing nausea, vomiting, anorexia, arthralgia, fever or chills. One severe adverse event was reported 18 hours after treatment onset with 60 mg AL + ZIN-CTI-001, and included diarrhea, followed by hypotension and reversible acute renal failure, which completely resolved.
The FDA has recently accepted our IND application to begin clinical study of ZIN-ATI-001 in oncology and patient enrollment is underway. The Phase 1 study will evaluate safety in addition to immunological and biological effects of the therapeutic candidate in patients with melanoma.
We intend to evaluate both ZIN-CTI-001 and ZIN-ATI-001 with the intent either to further develop both candidates or to select one of the two candidates for further study. ZIN-ATI-001 is identical to ZIN- CTI-001 except that the autologous dendritic cell component is omitted. Both product candidates are targeted for further development in different indications.
Development plans
We are currently pursuing several clinical programs which include:
| • | | palifosfamide (Zymafos TM or ZIO-201)—completing our Phase 3 pivotal trial in front-line metastatic soft tissue sarcoma, entitled PICASSO 3, and completing our recently initiated Phase 1 trial with palifosfamide in combination with etoposide and carboplatin to determine appropriate safety for initiating a subsequent randomized trial in front-line small-cell lung cancer. |
| • | | darinaparsin (Zinapar TM or ZIO-101)—completing an ongoing Phase 1 study with an oral form. |
| • | | indibulin (Zybulin TM or ZIO-301)—entering the Phase 2 portion of the Phase 1/2 trial having established the MTD in Phase 1 with once daily dosing. |
| • | | ZIN-CTI-001—completing a Phase 1b trial in patients with advanced melanoma that is on-going in the United States. |
| • | | ZIN-ATI-001—completing the Phase 1 trial treatment of patients with late-stage malignant melanoma that is the subject of an IND application recently accepted by FDA. |
We are also in late preclinical evaluation with respect to several additional potential product candidates under our channel partnership with Intrexon and we anticipate continuing evaluation to select product candidates for clinical study, which could commence as early as 2012. We also anticipate continuing discovery efforts aimed at identifying additional potential product candidates under the Intrexon channel partnership for study thereafter.
4
Our current plans involve using internal financial resources to develop palifosfamide and pursue the clinical work outlined above, with the intention of ultimately partnering or otherwise raising additional resources to support further development activities for all of our product candidates. The successful development of our product candidates is highly uncertain. Product development costs and timelines can vary significantly for each product candidate, are difficult to accurately predict, and will require us to obtain additional funding, either alone or in connection with partnering arrangements. Various statutes and regulations also govern or influence the manufacturing, safety, labeling, storage, record keeping and marketing of each product. The lengthy process of seeking approval and the subsequent compliance with applicable statutes and regulations require the expenditure of substantial resources. Any failure by us to obtain, or any delay in obtaining, regulatory approvals could materially, adversely affect our business. To date, we have not received approval for the sale of any product candidates in any market and, therefore, have not generated any revenues from our product candidates.
Corporate information
We originally incorporated in Colorado in September 1998 (under the name Net Escapes, Inc.) and later changed our name to “EasyWeb, Inc.” in February 1999. We re-incorporated in Delaware on May 16, 2005 under the same name. On September 13, 2005, we completed a “reverse” acquisition of privately held ZIOPHARM, Inc., a Delaware corporation. To effect this transaction, we caused ZIO Acquisition Corp., our wholly-owned subsidiary, to merge with and into ZIOPHARM, Inc., with ZIOPHARM, Inc. surviving as our wholly owned subsidiary. In accordance with the terms of the merger, the outstanding common stock of ZIOPHARM, Inc. automatically converted into the right to receive an aggregate of approximately 97.3% of our outstanding common stock (after giving effect to the transaction). Following the merger, we caused ZIOPHARM, Inc. to merge with and into us and we changed our name to “ZIOPHARM Oncology, Inc.” Although EasyWeb, Inc. was the legal acquirer in the transaction, we accounted for the transaction as a reverse acquisition under generally accepted accounting principles. As a result, ZIOPHARM, Inc. became the registrant with the Commission and the historical financial statements of ZIOPHARM, Inc. became our historical financial statements.
Our principal executive offices are located at 1180 Avenue of the Americas, 19th Floor, New York, NY 10036, and our telephone number is (646) 214-0700. Our Internet site iswww.ziopharm.com . Information found on, or accessible through, our website is not a part of, and is not incorporated into, this prospectus, and you should not consider it part of this prospectus or part of any prospectus supplement.
5
Risk factors
Investing in our securities involves a high degree of risk. You should carefully review the risks and uncertainties described under the heading “Risk Factors” contained in the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other documents that are incorporated by reference into this prospectus, before deciding whether to purchase any of the securities being offered. Each of the risk factors could adversely affect our business, operating results and financial condition, as well as adversely affect the value of an investment in our securities, and the occurrence of any of these risks might cause you to lose all or part of your investment. Moreover, the risks described are not the only ones that we face. Additional risks not presently known to us or that we currently believe are immaterial may also significantly impair our business operations.
Special note regarding
forward-looking statements
This prospectus and the documents incorporated by reference herein contain, and any prospectus supplement or free writing prospectus may contain, forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended, or the Exchange Act. These statements relate to future events or to our future financial performance and involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements to be materially different from any future results, performances or achievements expressed or implied by the forward-looking statements. Forward-looking statements may include, but are not limited to statements about:
| • | | the progress, timing and results of preclinical and clinical trials involving our drug candidates; |
| • | | the progress of our research and development programs; |
| • | | our plans or others’ plans to conduct future clinical trials or research and development efforts; |
| • | | the risk that final trial data may not support interim analysis of the viability of our drug candidates; |
| • | | our plans and expectations regarding partnering our drug candidates; |
| • | | the benefits to be derived from relationships with our collaborators; |
| • | | the receipt or anticipated receipt of regulatory clearances and approvals; |
| • | | estimates of the potential markets for our drug candidates; |
| • | | our ability to adequately protect our intellectual property rights; |
| • | | the use of proceeds from this offering; |
| • | | our estimates of future revenues and profitability; and |
| • | | our estimates regarding our capital requirements, our ability to control our costs and our need for additional funding. |
6
In some cases, you can identify forward-looking statements by terms such as “may”, “will”, “should”, “could”, “would”, “expects”, “plans”, “anticipates”, “believes”, “estimates”, “projects”, “predicts”, “potential” and similar expressions intended to identify forward-looking statements. These statements reflect our current views with respect to future events and are based on assumptions and subject to risks and uncertainties. Given these uncertainties, you should not place undue reliance on these forward-looking statements. We discuss many of these risks in greater detail under the heading “Risk Factors” in the applicable prospectus supplement or free writing prospectus and in our reports filed from time to time under the Securities Act and/or the Exchange Act. We encourage you to read these filings as they are made. Also, these forward-looking statements represent our estimates and assumptions only as of the date of the document containing the applicable statement.
You should read this prospectus, the documents incorporated by reference herein, and any prospectus supplement or free writing prospectus that we have authorized for use in connection with this offering completely and with the understanding that our actual future results may be materially different from what we expect. We qualify all of the forward-looking statements in the foregoing documents by these cautionary statements.
Unless required by law, we undertake no obligation to update or revise any forward-looking statements to reflect new information or future events or developments. Thus, you should not assume that our silence over time means that actual events are bearing out as expressed or implied in such forward-looking statements.
7
Ratio of earnings to fixed charges and to
combined fixed charges and preference dividends
The following table shows our ratio of earnings to fixed charges and the ratio of earnings to combined fixed charges and preference dividends for the periods indicated.
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | Year ended December 31, | | | Nine months ended September 30, 2011 | |
| | | 2006 | | | 2007 | | | 2008 | | | 2009 | | | 2010 | | |
| |
Ratio of earnings to fixed charges | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | |
Ratio of earnings to combined fixed charges and preference dividends | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | |
| |
For purposes of computing the ratio of earnings to fixed charges and the ratio of earnings to our combined fixed charges and preference dividends, earnings consist of our net income (loss) before tax benefit (provision) for the period plus fixed charges. Fixed charges consist of interest expense and amortized premiums, discounts and capitalized expenses related to indebtedness. The ratio of earnings to fixed charges and the ratio of earnings to combined fixed charges and preference dividends were less than one-to-one for each of the periods presented. Earnings were insufficient to cover fixed charges by $17.9 million in 2006, $26.6 million in 2007, $25.2 million in 2008, $7.6 million in 2009, $32.7 million in 2010 and $50.5 million in the nine months ended September 30, 2011.
Use of proceeds
We will retain broad discretion over the use of the net proceeds from the sale of our securities offered by this prospectus. Unless we indicate otherwise in the applicable prospectus supplement or in any related free writing prospectus we have authorized for use in connection with a specific offering, we anticipate that any net proceeds will be used for working capital and general corporate purposes. We will set forth in the applicable prospectus supplement or free writing prospectus our intended use for the net proceeds received from the sale of securities sold pursuant to that prospectus supplement or free writing prospectus.
8
Description of capital stock
As of the date of this prospectus, our authorized capital stock consists of 280,000,000 shares, comprised of 250,000,000 shares of common stock, par value $.001 per share, and 30,000,000 shares of preferred stock, par value $.001 per share. As of October 28, 2011, there were 68,451,324 shares of common stock and no shares of preferred stock issued and outstanding. Our common stock is traded on the NASDAQ Capital Market under the symbol “ZIOP”.
The following description summarizes the material terms of our capital stock. This summary is, however, subject to the provisions of our certificate of incorporation and bylaws. For greater detail about our capital stock, please refer to our certificate of incorporation and bylaws.
Common stock
Voting. The holders of our common stock are entitled to one vote for each outstanding share of common stock owned by such stockholder on every matter properly submitted to the stockholders for their vote. Stockholders are not entitled to vote cumulatively for the election of directors. Because of this, the holders of a majority of the shares of common stock entitled to vote in any election of directors can elect all of the directors standing for election, if they should so choose. At any meeting of the stockholders, a quorum as to any matter shall consist of a majority of the votes entitled to be cast on the matter, except where a larger quorum is required by law, by our certificate of incorporation or by our bylaws.
Dividend rights. Holders of our common stock are entitled to receive ratably dividends and other distributions of cash or any other right or property as may be declared by our board of directors out of our assets or funds legally available for such dividends or distributions. The dividend rights of holders of common stock are subject to the dividend rights of the holders of any series of preferred stock that may be issued and outstanding from time to time.
Liquidation rights. In the event of any voluntary or involuntary liquidation, dissolution or winding up of our affairs, holders of our common stock would be entitled to share ratably in our assets that are legally available for distribution to stockholders after payment of liabilities. If we have any preferred stock outstanding at such time, the holders of such preferred stock may be entitled to distribution and/or liquidation preferences that require us to pay the applicable distribution to the holders of preferred stock before paying distributions to the holders of common stock.
Rights and preferences. Holders of our common stock have no preemptive, conversion or subscription rights, and there are no redemption or sinking fund provisions applicable to our common stock. The rights, preferences and privileges of the holders of common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of our preferred stock that we may designate and issue in the future.
The transfer agent and registrar for our common stock is American Stock Transfer & Trust Company.
See “Certain Provisions of Delaware Law, the Certificate of Incorporation and Bylaws” for a description of provisions of our certificate of incorporation and bylaws which may have the effect of delaying, deferring or preventing changes in the our control.
9
Preferred stock
Pursuant to our amended and restated certificate of incorporation, our board of directors has the authority, without stockholder approval, subject to limitations prescribed by law, to provide for the issuance of up to 30,000,000 shares of preferred stock in one or more series, and by filing a certificate pursuant to the applicable law of the State of Delaware, to establish from time to time the number of shares to be included in each such series, and to fix the designation, powers, preferences and rights of the shares of each series and any qualifications, limitations or restrictions thereof, and to increase or decrease the number of shares of any such series, but not below the number of shares of such series then outstanding.
We will fix the designations, voting powers, preferences and rights of the preferred stock of each series, as well as the qualifications, limitations or restrictions thereof, in the certificate of designation relating to that series. We will file as an exhibit to the registration statement of which this prospectus is a part, or will incorporate by reference from reports that we file with the Commission, the form of any certificate of designation that describes the terms of the series of preferred stock we are offering before the issuance of that series of preferred stock. This description will include:
| • | | the title and stated value; |
| • | | the number of shares offered; |
| • | | the liquidation preference per share; |
| • | | the purchase price per share; |
| • | | the dividend rate(s), period(s) and/or payment date(s) or method(s) of calculation for dividends; |
| • | | whether dividends are cumulative or non-cumulative and, if cumulative, the date from which dividends will accumulate; |
| • | | our right, if any, to defer payment of dividends and the maximum length of any such deferral period; |
| • | | the procedures for any auction and remarketing, if any; |
| • | | the provisions for a sinking fund, if any; |
| • | | the provision for redemption or repurchase, if applicable, and any restrictions on our ability to exercise those redemption and repurchase rights; |
| • | | any listing of the preferred stock on any securities exchange or market; |
| • | | the terms and conditions, if applicable, upon which the preferred stock will be convertible into common stock, including the conversion price (or manner of calculation) and conversion period; |
| • | | whether the preferred stock will be exchangeable into debt securities, and, if applicable, the exchange price, or how it will be calculated, and the exchange period; |
| • | | voting rights, if any, of the preferred stock; |
| • | | preemptive rights, if any; |
10
| • | | restrictions on transfer, sale or other assignment, if any; |
| • | | whether interests in the preferred stock will be represented by depositary shares; |
| • | | a discussion of any material and/or special United States federal income tax considerations applicable to the preferred stock; |
| • | | the relative ranking and preferences of the preferred stock as to dividend rights and rights upon the liquidation, dissolution or winding up of our affairs; |
| • | | any limitations on issuance of any class or series of preferred stock ranking senior to or on a parity with the class or series of preferred stock as to dividend rights and rights upon liquidation, dissolution or winding up of our affairs; and |
| • | | any other specific terms, preferences, rights, limitations or restrictions of the preferred stock. |
Our board of directors could authorize the issuance of shares of preferred stock with terms and conditions that could have the effect of discouraging a takeover or other transaction that might involve a premium price for holders of the shares or which holders might believe to be in their best interests. The issuance of preferred stock could adversely affect the voting power, conversion or other rights of holders of common stock and reduce the likelihood that common stockholders will receive dividend payments and payments upon liquidation.
The laws of the state of Delaware, the state of our incorporation, provide that the holders of preferred stock will have the right to vote separately as a class on any proposal involving fundamental changes in the rights of holders of such preferred stock. This right is in addition to any voting rights that may be provided for in the applicable certificate of designation.
The transfer agent and registrar for any series of preferred stock will be set forth in the applicable prospectus supplement.
11
Description of debt securities
We may issue debt securities from time to time, in one or more series, as either senior or subordinated debt or as senior or subordinated convertible debt. The terms of each series of debt securities will be established by or pursuant to a resolution of our board of directors and set forth or determined in the manner provided in an officers’ certificate or by a supplemental indenture. The particular terms of each series of debt securities will be described in more detail in the applicable prospectus supplement or any related free writing prospectus. The terms of any debt securities offered under a prospectus supplement may differ from the terms described below.
Unless the context requires otherwise, whenever we refer to the indenture, we also are referring to any supplemental indentures that specify the terms of a particular series of debt securities.
We will issue the debt securities under an indenture that we will enter into with the trustee named in the indenture. The indenture will be qualified under the Trust Indenture Act of 1939. We have filed a form of the indenture as an exhibit to the registration statement of which this prospectus is a part, and supplemental indentures and forms of debt securities containing the terms of the debt securities being offered will be filed as exhibits to the registration statement of which this prospectus is a part or will be incorporated by reference from reports that we file with the Commission.
The following summaries of material provisions of the debt securities and the indenture are subject to, and qualified in their entirety by reference to, all of the provisions of the indenture, including any supplemental indentures applicable to a particular series of debt securities. We urge you to read the applicable prospectus supplements and any related free writing prospectuses related to the debt securities that we may offer under this prospectus, as well as the complete indenture that contains the terms of the debt securities.
General
The indenture does not limit the amount of debt securities that we may issue. It provides that we may issue debt securities up to the principal amount that we may authorize and may be in any currency or currency unit that we may designate. Except for the limitations on consolidation, merger and sale of all or substantially all of our assets contained in the indenture, the terms of the indenture do not contain any covenants or other provisions designed to give holders of any debt securities protection against changes in our operations, financial condition or transactions involving us.
We will describe in the applicable prospectus supplement the terms of the series of debt securities being offered, including:
| • | | any limit on the aggregate principal amount; |
| • | | the person who shall be entitled to receive interest, if other than the record holder on the record date; |
| • | | the date the principal will be payable; |
12
| • | | the interest rate, if any, the date interest will accrue, the interest payment dates and the regular record dates; |
| • | | the place where payments may be made; |
| • | | any mandatory or optional redemption provisions; |
| • | | if applicable, the method for determining how the principal, premium, if any, or interest will be calculated by reference to an index or formula; |
| • | | if other than United States currency, the currency or currency units in which principal, premium, if any, or interest will be payable and whether we or the holder may elect payment to be made in a different currency; |
| • | | the portion of the principal amount that will be payable upon acceleration of stated maturity, if other than the entire principal amount; |
| • | | any defeasance provisions if different from those described below under “Satisfaction and Discharge; Defeasance”; |
| • | | any conversion or exchange provisions; |
| • | | any obligation to redeem or purchase the debt securities pursuant to a sinking fund; |
| • | | whether the debt securities will be issuable in the form of a global security; |
| • | | any applicable subordination provisions for any subordinated debt securities; |
| • | | any deletions of, or changes or additions to, the events of default or covenants; and |
| • | | any other specific terms of such debt securities. |
Unless otherwise specified in the prospectus supplement, the debt securities will be registered debt securities. Debt securities may be sold at a substantial discount below their stated principal amount, bearing no interest or interest at a rate which at the time of issuance is below market rates.
Exchange and transfer
Debt securities may be transferred or exchanged at the office of the security registrar or at the office of any transfer agent designated by us.
We will not impose a service charge for any transfer or exchange, but we may require holders to pay any tax or other governmental charges associated with any transfer or exchange.
In the event of any potential redemption of debt securities of any series, we will not be required to:
| • | | issue, register the transfer of, or exchange, any debt security of that series during a period beginning at the opening of business 15 days before the day of mailing of a notice of redemption and ending at the close of business on the day of the mailing; or |
| • | | register the transfer of, or exchange, any debt security of that series selected for redemption, in whole or in part, except the unredeemed portion being redeemed in part. |
13
We may initially appoint the trustee as the security registrar. Any transfer agent, in addition to the security registrar, initially designated by us will be named in the prospectus supplement. We may designate additional transfer agents or change transfer agents or change the office of the transfer agent. However, we will be required to maintain a transfer agent in each place of payment for the debt securities of each series.
Global securities
The debt securities of any series may be represented, in whole or in part, by one or more global securities. Each global security will:
| • | | be registered in the name of a depositary that we will identify in a prospectus supplement; |
| • | | be deposited with the depositary or nominee or custodian; and |
| • | | bear any required legends. |
No global security may be exchanged in whole or in part for debt securities registered in the name of any person other than the depositary or any nominee unless:
| • | | the depositary has notified us that it is unwilling or unable to continue as depositary or has ceased to be qualified to act as depositary; |
| • | | an event of default is continuing; or |
| • | | the Company executes and delivers to the trustee an officers’ certificate stating that the global security is exchangeable. |
As long as the depositary, or its nominee, is the registered owner of a global security, the depositary or nominee will be considered the sole owner and holder of the debt securities represented by the global security for all purposes under the indenture. Except in the above limited circumstances, owners of beneficial interests in a global security:
| • | | will not be entitled to have the debt securities registered in their names; |
| • | | will not be entitled to physical delivery of certificated debt securities; and |
| • | | will not be considered to be holders of those debt securities under the indenture. |
Payments on a global security will be made to the depositary or its nominee as the holder of the global security. Some jurisdictions have laws that require that certain purchasers of securities take physical delivery of such securities in definitive form. These laws may impair the ability to transfer beneficial interests in a global security.
Institutions that have accounts with the depositary or its nominee are referred to as “participants.” Ownership of beneficial interests in a global security will be limited to participants and to persons that may hold beneficial interests through participants. The depositary will credit, on its book-entry registration and transfer system, the respective principal amounts of debt securities represented by the global security to the accounts of its participants.
Ownership of beneficial interests in a global security will be shown on and effected through records maintained by the depositary, with respect to participants’ interests, or any participant, with respect to interests of persons held by participants on their behalf.
Payments, transfers and exchanges relating to beneficial interests in a global security will be subject to policies and procedures of the depositary.
14
The depositary policies and procedures may change from time to time. Neither we nor the trustee will have any responsibility or liability for the depositary’s or any participant’s records with respect to beneficial interests in a global security.
Payment and paying agent
The provisions of this paragraph will apply to the debt securities unless otherwise indicated in the prospectus supplement. Payment of interest on a debt security on any interest payment date will be made to the person in whose name the debt security is registered at the close of business on the regular record date. Payment on debt securities of a particular series will be payable at the office of a paying agent or paying agents designated by us. However, at our option, we may pay interest by mailing a check to the record holder. Except as described in the prospectus supplement, the corporate trust office of the trustee will be designated as our sole paying agent.
We may also name any other paying agents in the prospectus supplement. We may designate additional paying agents, change paying agents or change the office of any paying agent. However, we will be required to maintain a paying agent in each place of payment for the debt securities of each series.
All moneys paid by us to a paying agent for payment on any debt security that remain unclaimed at the end of two years after such payment was due will be repaid to us. Thereafter, the holder may look only to us for such payment.
Consolidation, merger and sale of assets
Except as otherwise set forth in the applicable prospectus supplement, we may not consolidate with or merge into any other person, in a transaction in which we are not the surviving corporation, or sell, convey, transfer or lease all or substantially all of our assets to, any person, unless:
| • | | the successor, if any, is a United States corporation, limited liability company, partnership, trust or other entity; |
| • | | the successor assumes our obligations on the debt securities and under the indenture; |
| • | | immediately after giving effect to the transaction, no default or event of default shall have occurred and be continuing; and |
| • | | certain other conditions are met. |
Events of default
Unless we inform you otherwise in the applicable prospectus supplement, the indenture will define an event of default with respect to any series of debt securities as one or more of the following events:
| (1) | | failure to pay principal of or any premium on any debt security of that series when due; |
| (2) | | failure to pay any interest on any debt security of that series for 30 days when due; |
| (3) | | failure to deposit any sinking fund payment when due; |
| (4) | | failure to perform any other covenant in the indenture continued for 90 days after being given the notice required in the indenture; |
15
| (5) | | our bankruptcy, insolvency or reorganization; and |
| (6) | | any other event of default specified in the applicable prospectus supplement. |
An event of default of one series of debt securities is not necessarily an event of default for any other series of debt securities.
If an event of default, other than an event of default described in clause (5) above, shall occur and be continuing, either the trustee or the holders of at least 25% in aggregate principal amount of the outstanding securities of that series may declare the principal amount of the debt securities of that series to be due and payable immediately.
If an event of default described in clause (5) above shall occur, the principal amount of all the debt securities of that series will automatically become immediately due and payable. Any payment by us on subordinated debt securities following any such acceleration will be subject to the subordination provisions described below under “Subordinated Debt Securities.”
After acceleration, the holders of a majority in aggregate principal amount of the outstanding securities of that series may, under certain circumstances, rescind and annul such acceleration if all events of default, other than the non-payment of accelerated principal, or other specified amount, have been cured or waived.
Other than the duty to act with the required care during an event of default, the trustee will not be obligated to exercise any of its rights or powers at the request of the holders unless the holders shall have offered to the trustee reasonable indemnity. Generally, the holders of a majority in aggregate principal amount of the outstanding debt securities of any series will have the right to direct the time, method and place of conducting any proceeding for any remedy available to the trustee or exercising any trust or power conferred on the trustee.
A holder will not have any right to institute any proceeding under the indenture, or for the appointment of a receiver or a trustee, or for any other remedy under the indenture, unless:
| (1) | | the holder has previously given to the trustee written notice of a continuing event of default with respect to the debt securities of that series; |
| (2) | | the holders of at least 25% in aggregate principal amount of the outstanding debt securities of that series have made a written request and have offered reasonable indemnity to the trustee to institute the proceeding; and |
| (3) | | the trustee has failed to institute the proceeding and has not received direction inconsistent with the original request from the holders of a majority in aggregate principal amount of the outstanding debt securities of that series within 90 days after the original request. |
Holders may, however, sue to enforce the payment of principal or interest on any debt security on or after the due date without following the procedures listed in (1) through (3) above.
Modification and waiver
Except as provided in the next two succeeding paragraphs, the applicable trustee and we may make modifications and amendments to the indenture (including, without limitation, through consents obtained in connection with a tender offer or exchange offer for, outstanding securities) and may waive any existing default or event of default (including, without limitation,
16
through consents obtained in connection with a tender offer or exchange offer for, outstanding securities) with the consent of the holders of a majority in aggregate principal amount of the outstanding securities of each series affected by the modification or amendment.
However, neither we nor the trustee may make any amendment or waiver without the consent of the holder of each outstanding security of that series affected by the amendment or waiver if such amendment or waiver would, among other things:
| • | | change the amount of securities whose holders must consent to an amendment, supplement or waiver; |
| • | | change the stated maturity of any debt security; |
| • | | reduce the principal on any debt security or reduce the amount of, or postpone the date fixed for, the payment of any sinking fund; |
| • | | reduce the principal of an original issue discount security on acceleration of maturity; |
| • | | reduce the rate of interest or extend the time for payment of interest on any debt security; |
| • | | make a principal or interest payment on any debt security in any currency other than that stated in the debt security; |
| • | | impair the right to enforce any payment after the stated maturity or redemption date; |
| • | | waive any default or event of default in payment of the principal of, premium or interest on any debt security (except certain rescissions of acceleration); or |
| • | | waive a redemption payment or modify any of the redemption provisions of any debt security. |
Notwithstanding the preceding, without the consent of any holder of outstanding securities, we and the trustee may amend or supplement the indenture:
| • | | to cure any ambiguity, defect or inconsistency; |
| • | | to provide for the issuance of and establish the form and terms and conditions of debt securities of any series as permitted by the indenture; |
| • | | to provide for uncertificated securities in addition to or in place of certificated securities; |
| • | | to provide for the assumption of our obligations to holders of any debt security in the case of a merger, consolidation, transfer or sale of all or substantially all of our assets; |
| • | | to make any change that does not materially adversely affect the legal rights under the indenture of any such holder; |
| • | | to comply with requirements of the SEC in order to effect or maintain the qualification of the indenture under the Trust Indenture Act of 1939; or |
| • | | to evidence and provide for the acceptance of appointment by a successor trustee with respect to the debt securities of one or more series and to add to or change any of the provisions of the indenture as shall be necessary to provide for or facilitate the administration of the trusts by more than one trustee. |
The consent of holders is not necessary under the indenture to approve the particular form of any proposed amendment. It is sufficient if such consent approves the substance of the proposed amendment.
17
Satisfaction and discharge; defeasance
We may be discharged from our obligations on the debt securities of any series that have matured or will mature or be redeemed within one year if we deposit with the trustee enough cash to pay all the principal, interest and any premium due to the stated maturity date or redemption date of the debt securities.
The indenture contains a provision that permits us to elect:
| • | | to be discharged from all of our obligations, subject to limited exceptions, with respect to any series of debt securities then outstanding; and/or |
| • | | to be released from our obligations under certain covenants and from the consequences of an event of default resulting from a breach of certain covenants, including covenants as to payment of taxes and maintenance of corporate existence. |
To make either of the above elections, we must deposit in trust with the trustee enough money to pay in full the principal and interest on the debt securities. This amount may be made in cash and/or United States government obligations. As a condition to either of the above elections, we must deliver to the trustee an opinion of counsel that the holders of the debt securities will not recognize income, gain or loss for federal income tax purposes as a result of the action.
If any of the above events occurs, the holders of the debt securities of the series will not be entitled to the benefits of the indenture, except for the rights of holders to receive payments on debt securities or the registration of transfer and exchange of debt securities and replacement of lost, stolen or mutilated debt securities.
Subordinated debt securities
If the trustee or any holder of the notes receives any payment or distribution of our assets in contravention of the subordination provisions on subordinated debt securities before all senior indebtedness is paid in full in cash, property or securities, including by way of set-off, or other payment satisfactory to holders of senior indebtedness, then such payment or distribution will be held in trust for the benefit of holders of senior indebtedness or their representatives to the extent necessary to make payment in full in cash or payment satisfactory to the holders of senior indebtedness of all unpaid senior indebtedness.
In the event of our bankruptcy, dissolution or reorganization, holders of senior indebtedness may receive more, ratably, and holders of subordinated debt securities may receive less, ratably, than our other creditors (including our trade creditors). This subordination will not prevent the occurrence of any event of default under the indenture.
Notices
Notices to holders will be given by mail to the addresses of the holders in the security register.
Governing law
The indenture and the debt securities will be governed by, and construed under, the law of the State of New York, except to the extent that the Trust Indenture Act of 1939 is applicable.
18
Description of warrants
General
We may issue warrants for the purchase of our debt securities, preferred stock or common stock, or any combination thereof. Warrants may be issued independently or together with our debt securities, preferred stock or common stock and may be attached to or separate from any offered securities. Each series of warrants will be issued under a separate warrant agreement. We may enter into a warrant agreement with a bank or trust company, as warrant agent. We will indicate the name and address and other information regarding the warrant agent in the applicable prospectus supplement or free writing prospectus relating to a particular series of warrants. The warrant agent will act solely as our agent in connection with the warrants. The warrant agent will not have any obligation or relationship of agency or trust for or with any holders or beneficial owners of warrants.
This summary of certain provisions of the warrants is not complete. For the terms of a particular series of warrants, you should refer to the prospectus supplement or free writing prospectus for that series of warrants and the warrant agreement for that particular series. The terms of any warrants we offer under a prospectus supplement or free writing prospectus may differ from the terms we describe below.
Debt warrants
The prospectus supplement or free writing prospectus relating to a particular issue of warrants to purchase debt securities will describe the terms of the debt warrants, including the following:
| • | | the title of the debt warrants; |
| • | | the offering price for the debt warrants, if any; |
| • | | the aggregate number of the debt warrants; |
| • | | the designation and terms of the debt securities, including any conversion rights, purchasable upon exercise of the debt warrants; |
| • | | if applicable, the date from and after which the debt warrants and any debt securities issued with them will be separately transferable; |
| • | | the principal amount of debt securities that may be purchased upon exercise of a debt warrant and the exercise price for the warrants, which may be payable in cash, securities or other property; |
| • | | the dates on which the right to exercise the debt warrants will commence and expire; |
| • | | if applicable, the minimum or maximum amount of the debt warrants that may be exercised at any one time; |
| • | | whether the debt warrants represented by the debt warrant certificates or debt securities that may be issued upon exercise of the debt warrants will be issued in registered or bearer form; |
| • | | information with respect to book-entry procedures, if any; |
| • | | the currency or currency units in which the offering price, if any, and the exercise price are payable; |
19
| • | | if applicable, a discussion of material United States federal income tax considerations; |
| • | | the antidilution provisions of the debt warrants, if any; |
| • | | the redemption or call provisions, if any, applicable to the debt warrants; |
| • | | any provisions with respect to the holder’s right to require us to repurchase the warrants upon a change in control or similar event; and |
| • | | any additional terms of the debt warrants, including procedures, and limitations relating to the exchange, exercise and settlement of the debt warrants. |
Debt warrant certificates will be exchangeable for new debt warrant certificates of different denominations. Debt warrants may be exercised at the corporate trust office of the warrant agent or any other office indicated in the prospectus supplement or free writing prospectus.
Prior to the exercise of their debt warrants, holders of debt warrants will not have any of the rights of holders of the debt securities purchasable upon exercise and will not be entitled to payment of principal or any premium, if any, or interest on the debt securities purchasable upon exercise or to enforce covenants in the indenture.
Equity warrants
The prospectus supplement or free writing prospectus relating to a particular series of warrants to purchase our common stock or preferred stock will describe the terms of the warrants, including the following:
| • | | the title of the warrants; |
| • | | the offering price for the warrants, if any; |
| • | | the aggregate number of warrants; |
| • | | the designation and terms of the common stock or preferred stock that may be purchased upon exercise of the warrants; |
| • | | if applicable, the designation and terms of the securities with which the warrants are issued and the number of warrants issued with each security; |
| • | | if applicable, the date from and after which the warrants and any securities issued with the warrants will be separately transferable; |
| • | | the number of shares of common stock or preferred stock that may be purchased upon exercise of a warrant and the exercise price for the warrants; |
| • | | the dates on which the right to exercise the warrants shall commence and expire; |
| • | | if applicable, the minimum or maximum amount of the warrants that may be exercised at any one time; |
| • | | the currency or currency units in which the offering price, if any, and the exercise price are payable; |
| • | | if applicable, a discussion of material United States federal income tax considerations; |
| • | | the antidilution provisions of the warrants, if any; |
20
| • | | the redemption or call provisions, if any, applicable to the warrants; |
| • | | any provisions with respect to holder’s right to require us to repurchase the warrants upon a change in control or similar event; and |
| • | | any additional terms of the warrants, including procedures, and limitations relating to the exchange, exercise and settlement of the warrants. |
Holders of equity warrants will not be entitled:
| • | | to vote, consent or receive dividends; |
| • | | to receive notice as stockholders with respect to any meeting of stockholders for the election of our directors or any other matter; or |
| • | | to exercise any rights as stockholders of the Company. |
Exercise of warrants
Each warrant will entitle the holder to purchase the securities that we specify in the applicable prospectus supplement or free writing prospectus at the exercise price that we describe in the applicable prospectus supplement or free writing prospectus. The warrants may be exercised as set forth in the prospectus supplement or free writing prospectus relating to the warrants offered. Unless we otherwise specify in the applicable prospectus supplement or free writing prospectus, warrants may be exercised at any time up to the close of business on the expiration date set forth in the prospectus supplement or free writing prospectus relating to the warrants offered thereby. After the close of business on the expiration date, unexercised warrants will become void.
Upon receipt of payment and the warrant certificate or agreement, as applicable, properly completed and duly executed at the corporate trust office of the warrant agent, if any, or any other office, including ours, indicated in the prospectus supplement or free writing prospectus, we will, as soon as practicable, issue and deliver the securities purchasable upon such exercise. If less than all of the warrants represented by such warrant certificate or agreement are exercised, a new warrant certificate or agreement will be issued for the remaining warrants.
Governing law
Unless we provide otherwise in the applicable prospectus supplement or free writing prospectus, the warrants and warrant agreements will be governed by and construed in accordance with the laws of the State of New York.
Enforceability of rights by holders of warrants
Each warrant agent, if any, will act solely as our agent under the applicable warrant agreement and will not assume any obligation or relationship of agency or trust with any holder of any warrant. A single bank or trust company may act as warrant agent for more than one issue of warrants. A warrant agent will have no duty or responsibility in case of any default by us under the applicable warrant agreement or warrant, including any duty or responsibility to initiate any proceedings at law or otherwise, or to make any demand upon us. Any holder of a warrant may, without the consent of the related warrant agent or the holder of any other warrant, enforce by appropriate legal action its right to exercise, and receive the securities purchasable upon exercise of, its warrants.
21
Certain provisions of delaware law, the certificate of incorporation and bylaws
Limitations on directors’ liability
Our amended and restated certificate of incorporation and our bylaws contain provisions indemnifying our directors and officers to the fullest extent permitted by law. In addition, as permitted by Delaware law, our amended and restated certificate of incorporation provides that no director will be liable to us or our stockholders for monetary damages for breach of certain fiduciary duties as a director. The effect of this provision is to restrict our rights and the rights of our stockholders in derivative suits to recover monetary damages against a director for breach of certain fiduciary duties as a director, except that a director will be personally liable for:
| • | | the benefits to be derived from relationships with our collaborators; |
| • | | any breach of his or her duty of loyalty to the registrant or its stockholders; |
| • | | acts or omissions not in good faith which involve intentional misconduct or a knowing violation of law; |
| • | | the payment of dividends or the redemption or purchase of stock in violation of Delaware law; or |
| • | | any transaction from which the director derived an improper personal benefit. |
This provision does not affect a director’s liability under the federal securities laws.
To the extent that our directors, officers and controlling persons are indemnified under the provisions contained in our amended and restated certificate of incorporation, Delaware law or contractual arrangements against liabilities arising under the Securities Act, we have been advised that in the opinion of the Commission such indemnification is against public policy as expressed in the Securities Act, and is therefore unenforceable.
Provisions that may have an anti-takeover effect
Certain provisions set forth in our amended and restated certificate of incorporation, bylaws and in Delaware law, which are summarized below, are intended to enhance the likelihood of continuity and stability in the composition of our board of directors and in the policies formulated by our board of directors and to discourage certain types of transactions that may involve an actual or threatened change of control. In that regard, these provisions are designed to reduce our vulnerability to an unsolicited acquisition proposal. The provisions also are intended to discourage certain tactics that may be used in proxy fights. However, such provisions could have the effect of discouraging others from making tender offers for our shares and, as a consequence, they also may inhibit fluctuations in the market price of our common stock that could result from actual or rumored takeover attempts. Such provisions also may have the effect of preventing changes in our management.
Blank check preferred stock. Our amended and restated certificate of incorporation contains provisions that permit our board of directors to issue, without any further vote or action by the stockholders, up to 30,000,000 shares of preferred stock in one or more series and, with respect to each such series, to fix the number of shares constituting the series and the designation of the series, the voting powers (if any) of the shares of the series, and the preferences and relative, participating, optional and other special rights, if any, and any qualifications, limitations or
22
restrictions, of the shares of such series. As a result, our board of directors could authorize the issuance of shares of preferred stock with terms and conditions that could have the effect of delaying, deferring or preventing a transaction or a change in control that might involve a premium price for holders of the registrant’s common stock or otherwise be in their best interest.
Special meetings of stockholders. Our bylaws provide that special meetings of stockholders may be called only by the board of directors. Stockholders are not permitted to call a special meeting of stockholders or to require that the board of directors call such a special meeting.
Delaware takeover statute.
We are subject to Section 203 of the Delaware General Corporation Law, or DGCL, which regulates acquisitions of some Delaware corporations. In general, Section 203 prohibits, with some exceptions, a Delaware corporation that is a public company from engaging in any “business combination” with any “interested stockholder” for a period of three years following the date that such stockholder became an interested stockholder, unless:
| • | | prior to such date, the board of directors of the corporation approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder; |
| • | | on consummation of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the number of shares outstanding those shares owned (x) by persons who are directors and also officers and (y) by employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or |
| • | | on or subsequent to such date, the business combination is approved by the board of directors and authorized at an annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66 2/3% of the outstanding voting stock that is not owned by the interested stockholder. |
Section 203 of the DGCL defines “business combination” to include:
| • | | any merger or consolidation involving the corporation and the interested stockholder; |
| • | | any sale, transfer, pledge or other disposition of 10% or more of the assets of the corporation involving the interested stockholder; |
| • | | subject to certain exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder; |
| • | | any transaction involving the corporation that has the effect of increasing the proportionate share of the stock of any class or series of the corporation beneficially owned by the interested stockholder; or |
| • | | the receipt by the interested stockholder of the benefit of any loans, advances, guarantees, pledges or other financial benefits provided by or through the corporation. |
In general, Section 203 defines an “interested shareholder” as an entity or person beneficially owning 15% or more of the outstanding voting stock of the corporation and any entity or person affiliated with such entity or person.
23
Plan of distribution
We may sell the securities from time to time pursuant to underwritten public offerings, negotiated transactions, block trades or a combination of these methods. We may sell the securities to or through underwriters or dealers, through agents, or directly to one or more purchasers. We may distribute securities from time to time in one or more transactions:
| • | | at a fixed price or prices, which may be changed; |
| • | | at market prices prevailing at the times of sale; |
| • | | at prices related to such prevailing market prices; or |
A prospectus supplement or supplements (and any related free writing prospectus that we may authorize to be provided to you) will describe the terms of the offering of the securities, including, to the extent applicable:
| • | | the name or names of the underwriters, if any; |
| • | | the purchase price of the securities or other consideration therefore, and the proceeds, if any, we will receive from the sale; |
| • | | any over-allotment options under which underwriters may purchase additional securities from us; |
| • | | any agency fees or underwriting discounts and other items constituting agents’ or underwriters’ compensation; |
| • | | any public offering price; |
| • | | any discounts or concessions allowed or reallowed or paid to dealers; and |
| • | | any securities exchange or market on which the securities may be listed. |
Only underwriters named in the prospectus supplement will be underwriters of the securities offered by the prospectus supplement.
If underwriters are used in the sale, they will acquire the securities for their own account and may resell the securities from time to time in one or more transactions at a fixed public offering price or at varying prices determined at the time of sale. The obligations of the underwriters to purchase the securities will be subject to the conditions set forth in the applicable underwriting agreement. We may offer the securities to the public through underwriting syndicates represented by managing underwriters or by underwriters without a syndicate. Subject to certain conditions, the underwriters will be obligated to purchase all of the securities offered by the prospectus supplement, other than securities covered by any over-allotment option. Any public offering price and any discounts or concessions allowed or reallowed or paid to dealers may change from time to time. We may use underwriters with whom we have a material relationship. We will describe in the prospectus supplement, naming the underwriter, the nature of any such relationship.
We may sell securities directly or through agents we designate from time to time. We will name any agent involved in the offering and sale of securities and we will describe any commissions we will pay the agent in the prospectus supplement. Unless the prospectus supplement states otherwise, our agent will act on a best-efforts basis for the period of its appointment.
24
We may authorize agents or underwriters to solicit offers by certain types of institutional investors to purchase securities from us at the public offering price set forth in the prospectus supplement pursuant to delayed delivery contracts providing for payment and delivery on a specified date in the future. We will describe the conditions to these contracts and the commissions we must pay for solicitation of these contracts in the prospectus supplement.
We may provide agents and underwriters with indemnification against civil liabilities, including liabilities under the Securities Act, or contribution with respect to payments that the agents or underwriters may make with respect to these liabilities. Agents and underwriters may engage in transactions with, or perform services for, us in the ordinary course of business.
All securities we may offer, other than common stock, will be new issues of securities with no established trading market. Any underwriters may make a market in these securities, but will not be obligated to do so and may discontinue any market making at any time without notice. We cannot guarantee the liquidity of the trading markets for any securities.
Any underwriter may engage in over-allotment, stabilizing transactions, short-covering transactions and penalty bids in accordance with Regulation M under the Exchange Act. Over-allotment involves sales in excess of the offering size, which create a short position. Stabilizing transactions permit bids to purchase the underlying security so long as the stabilizing bids do not exceed a specified maximum price. Syndicate-covering or other short-covering transactions involve purchases of the securities, either through exercise of the over-allotment option or in the open market after the distribution is completed, to cover short positions. Penalty bids permit the underwriters to reclaim a selling concession from a dealer when the securities originally sold by the dealer are purchased in a stabilizing or covering transaction to cover short positions. Those activities may cause the price of the securities to be higher than it would otherwise be. If commenced, the underwriters may discontinue any of the activities at any time.
25
Legal matters
The validity of the securities offered hereby will be passed upon by Cooley LLP, Boston, Massachusetts.
Experts
The financial statements as of and for the year ended December 31, 2010 and for the period from September 9, 2003 (date of inception) through December 31, 2010, incorporated by reference into this prospectus and registration statement, and the effectiveness of internal control over financial reporting as of December 31, 2010, have been audited by McGladrey & Pullen, LLP, an independent registered public accounting firm, as stated in their report incorporated by reference herein, and are included in reliance upon such reports and upon the authority of such firm as experts in accounting and auditing.
The balance sheet as of December 31, 2009 and the related statements of operations, changes in preferred stock and stockholders’ equity (deficit) and cash flows for each of the years in thetwo-year period ended December 31, 2009, incorporated by reference into the registration statement of which this prospectus is a part, have been included herein in reliance on the report, dated March 17, 2010, of Caturano and Company, P.C. (whose name has since been changed to Caturano and Company, Inc.), independent registered public accounting firm, given on the authority of that firm as experts in auditing and accounting.
Where you can find more information
We are a reporting company and file annual, quarterly and current reports, proxy statements and other information with the Commission. You may read and copy these reports, proxy statements and other information at the Commission’s public reference room at 100 F Street, N.E., Washington, D.C. 20549 or at the Commission’s other public reference facilities. Please call the Commission at 1-800-SEC-0330 for more information about the operation of the public reference rooms. You can request copies of these documents by writing to the Commission and paying a fee for the copying costs. In addition, the Commission maintains an Internet site athttp://www.sec.gov that contains reports, proxy and information statements and other information regarding issuers that file electronically with the Commission. Our Commission filings are available on the Commission’s Internet site. We maintain a website athttp://www.ziopharm.com . Information found on, or accessible through, our website is not a part of, and is not incorporated into, this prospectus, and you should not consider it part of this prospectus or part of any prospectus supplement.
26
Incorporation of information by reference
We are allowed to incorporate by reference information contained in documents that we file with the Commission. This means that we can disclose important information to you by referring you to those documents and that the information in this prospectus is not complete and you should read the information incorporated by reference for more detail. Information in this prospectus supersedes information incorporated by reference that we filed with the Commission prior to the date of this prospectus, while information that we file later with the Commission will automatically update and supersede the information in this prospectus.
We incorporate by reference the documents listed below and any future filings we will make with the Commission under Section 13(a), 13(c), 14 or 15(d) of the Exchange Act (i) after the date of the initial filing of the registration statement of which this prospectus is a part and prior to effectiveness of such registration statement, and (ii) from the date of this prospectus but prior to the termination of the offering of the securities covered by this prospectus (other than Current Reports or portions thereof furnished under Item 2.02 or 7.01 of Form 8-K):
| • | | Annual Report on Form 10-K for the fiscal year ended December 31, 2010, filed on March 1, 2011; |
| • | | Quarterly Reports on Form 10-Q for the quarters ended March 31, 2011, June 30, 2011, and September 30, 2011, filed on May 5, 2011, August 1, 2011 (as amended by Amendment No. 1 on Form 10-Q/A filed on August 9, 2011), and November 3, 2011, respectively; |
| • | | Current Reports on Form 8-K filed on January 5, January 12, January 26, February 3 (excluding Item 2.02), February 7 (two filings), February 8, March 7, May 9, May 13, May 19, May 23, June 3, June 6, June 10, June 22, July 15, 2011, September 6, 2011, October 28, 2011 and November 3, 2011; and |
| • | | The description of our common stock set forth in the registration statement on Form 8-A registering our common stock under Section 12 of the Exchange Act, which was filed with the Commission on September 20, 2006, including any amendments or reports filed for purposes of updating such description. |
We will provide to each person, including any beneficial owner, to whom a prospectus is delivered a copy of any or all of the documents that are incorporated by reference in this prospectus but not delivered with this prospectus, including exhibits that are specifically incorporated by reference in such documents. You may request a copy of such documents at no cost, by writing or telephoning us at the following address or telephone number:
ZIOPHARM Oncology, Inc.
1180 Avenue of the Americas, 19th Floor
New York, NY 10036
Attention: President
Telephone: (646) 214-0700
27
14,300,000 shares

Common stock
Prospectus supplement
J.P. Morgan
| | | | |
| JMP Securities | | | | Griffin Securities, Inc. |
October 23, 2013


