
SECURITIES AND EXCHANGE COMMISSION
Form 20-F
4056 Basel, Switzerland
Group General Counsel
Novartis AG
CH 4056 Basel
Switzerland
Tel.: +41-61-324-1111
Fax: +41-61-324-7826
Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| American Depositary Shares each representing 1 share | NVS | New York Stock Exchange |
| Ordinary shares, nominal value CHF 0.50 per share* | NOVN | New York Stock Exchange* |
Large accelerated filer x | Accelerated filer o | Non-accelerated filer o | Emerging growth company o |
U.S. GAAP o | International Financial Reporting Standards as issued by the International Accounting Standards Board x | Other o |
Table of contents
Introduction and use of certain terms
Forward-looking statements
Part I
Item 1. Identity of Directors, Senior Management and Advisers
Item 2. Offer Statistics and Expected Timetable
Item 3. Key Information
3.A Selected financial data
| Year ended December 31, | |||||||||||
| (USD millions, except per share information) | 2019 | 2018 | 2017 | 2016 | 2015 | ||||||
INCOME STATEMENT DATA1 | |||||||||||
| Net sales to third parties from continuing operations | 47 445 | 44 751 | 42 338 | 41 975 | 42 641 | ||||||
| Operating income from continuing operations | 9 086 | 8 403 | 8 702 | 8 248 | 8 522 | ||||||
| Income from associated companies | 659 | 6 438 | 1 108 | 703 | 266 | ||||||
| Interest expense | – 850 | – 932 | – 750 | – 675 | – 637 | ||||||
| Other financial income and expense | 45 | 186 | 42 | – 385 | – 433 | ||||||
| Income before taxes from continuing operations | 8 940 | 14 095 | 9 102 | 7 891 | 7 718 | ||||||
| Taxes | – 1 793 | – 1 295 | – 1 603 | – 1 095 | – 1 066 | ||||||
| Net income from continuing operations | 7 147 | 12 800 | 7 499 | 6 796 | 6 652 | ||||||
| Net (loss) / income from discontinued operations before gain on distribution of Alcon Inc. to Novartis shareholders | – 101 | – 186 | 204 | – 98 | 376 | ||||||
| Gain on distribution of Alcon Inc. to Novartis AG shareholders | 4 691 | ||||||||||
| Net income related to portfolio transformation transactions | 10 766 | ||||||||||
| Net income from discontinued operations | 4 590 | – 186 | 204 | – 98 | 11 142 | ||||||
| Group net income | 11 737 | 12 614 | 7 703 | 6 698 | 17 794 | ||||||
| Attributable to: | |||||||||||
| Shareholders of Novartis AG | 11 732 | 12 611 | 7 703 | 6 712 | 17 783 | ||||||
| Non-controlling interests | 5 | 3 | 0 | – 14 | 11 | ||||||
| Basic earnings per share (USD) | |||||||||||
| Continuing operations | 3.12 | 5.52 | 3.20 | 2.86 | 2.77 | ||||||
| Discontinued operations | 2.00 | – 0.08 | 0.08 | – 0.04 | 4.63 | ||||||
| Total | 5.12 | 5.44 | 3.28 | 2.82 | 7.40 | ||||||
| Diluted earnings per share (USD) | |||||||||||
| Continuing operations | 3.08 | 5.46 | 3.17 | 2.84 | 2.72 | ||||||
| Discontinued operations | 1.98 | – 0.08 | 0.08 | – 0.04 | 4.57 | ||||||
| Total | 5.06 | 5.38 | 3.25 | 2.80 | 7.29 | ||||||
Cash dividends2 | 6 645 | 6 966 | 6 495 | 6 475 | 6 643 | ||||||
Cash dividends per share in CHF3 | 2.95 | 2.85 | 2.80 | 2.75 | 2.70 | ||||||
Personnel cost from continuing operations4, 5 | 13 843 | 13 515 | 12 009 | 11 950 | 11 336 | ||||||
Full-time equivalent associates of continuing operations at year-end5 | 103 914 | 104 780 | 102 467 | 99 747 | 99 624 | ||||||
1 Continuing operations include the businesses of the Innovative Medicines and Sandoz Divisions and Corporate activities. Discontinued operations include the Alcon business, which was divested in 2019; the Animal Health and Vaccines businesses divested in 2015; and the Consumer Health business, which was contributed also in 2015 into a new entity, GlaxoSmithKline Consumer Healthcare Holdings Ltd. (GSK Consumer Healthcare), where Novartis had a 36.5% interest. This newly created entity was sold during 2018 to GSK. To reflect these transactions, Novartis reported the Group’s financial results for 2019 to 2015 as “continuing operations” and “discontinued operations,” as required by IFRS. | |||||||||||
2 Cash dividends represent cash payments in the applicable year that generally relates to earnings of the previous year. | |||||||||||
3 Cash dividends per share represent dividends proposed that relate to earnings of the current year. Dividends for 2015 through 2018 were approved at the respective AGMs, and dividends for 2019 will be proposed to the Annual General Meeting on February 28, 2020, for approval. | |||||||||||
4 Personnel cost include wages, salaries, allowances, commissions and bonuses to staff, overtime, awards, holiday pay, severance payments and social welfare expenses. | |||||||||||
5 Own employees. | |||||||||||
| Year ended December 31, | |||||||||||
| (USD millions) | 2019 | 2018 | 2017 | 2016 | 2015 | ||||||
| BALANCE SHEET DATA | |||||||||||
| Cash, cash equivalents, and marketable securities and derivative financial instruments | 11 446 | 15 964 | 9 485 | 7 777 | 5 447 | ||||||
| Inventories | 5 982 | 6 956 | 6 867 | 6 255 | 6 226 | ||||||
| Other current assets | 11 235 | 11 836 | 11 856 | 10 899 | 11 172 | ||||||
| Non-current assets | 88 866 | 110 000 | 104 871 | 105 193 | 108 711 | ||||||
Assets of disposal group held for sale1 | 841 | 807 | |||||||||
| Total assets | 118 370 | 145 563 | 133 079 | 130 124 | 131 556 | ||||||
| Trade accounts payable | 5 424 | 5 556 | 5 169 | 4 873 | 5 668 | ||||||
| Other current liabilities | 22 809 | 24 000 | 18 234 | 17 336 | 18 040 | ||||||
| Non-current liabilities | 34 555 | 37 264 | 35 449 | 33 024 | 30 726 | ||||||
Liabilities of disposal group held for sale1 | 31 | 51 | |||||||||
| Total liabilities | 62 819 | 66 871 | 58 852 | 55 233 | 54 434 | ||||||
| Issued share capital and reserves attributable to shareholders of Novartis AG | 55 474 | 78 614 | 74 168 | 74 832 | 77 046 | ||||||
| Non-controlling interests | 77 | 78 | 59 | 59 | 76 | ||||||
| Total equity | 55 551 | 78 692 | 74 227 | 74 891 | 77 122 | ||||||
| Total liabilities and equity | 118 370 | 145 563 | 133 079 | 130 124 | 131 556 | ||||||
| Net assets | 55 551 | 78 692 | 74 227 | 74 891 | 77 122 | ||||||
| Outstanding share capital | 856 | 875 | 869 | 896 | 890 | ||||||
| Total outstanding shares (millions) | 2 265 | 2 311 | 2 317 | 2 374 | 2 374 | ||||||
1 The disposal group held for sale relate to the assets and liabilities of the pending divestment of the Sandoz US dermatology business and generic US oral solids portfolio to Aurobindo Pharma USA Inc., as announced on September 6, 2018 (see “Item 18. Financial Statements—Note 2. Significant pending transactions"). | |||||||||||
Year earned | Month and year paid | Total dividend per share (CHF) | Total dividend per share (USD) | ||||
| 2015 | March 2016 | 2.70 | 2.70 | ||||
| 2016 | March 2017 | 2.75 | 2.72 | ||||
| 2017 | March 2018 | 2.80 | 2.94 | ||||
| 2018 | March 2019 | 2.85 | 2.84 | ||||
2019 1 | March 2020 | 2.95 | 3.04 2 | ||||
1 Dividend to be proposed at the Annual General Meeting on February 28, 2020, and to be distributed March 5, 2020. | |||||||
2 Translated into US dollars at the December 31, 2019, rate of USD 1.032 to the Swiss franc. This translation is an example only, and should not be construed as a representation that the Swiss franc amount represents, or has been or could be converted into US dollars at that or any other rate. | |||||||
3.B Capitalization and indebtedness
3.C Reasons for the offer and use of proceeds
3.D Risk factors
Item 4. Information on the Company
4.A History and development of Novartis
4.B Business overview
Innovative Medicines
Project/ product | Common name | Mechanism of action | Potential indication | Business franchise | Formulation/ route of administration | Year project entered current development phase | Planned filing dates/current phase | ||||||||
| ABL001 | asciminib | BCR-ABL inhibitor | Chronic myeloid leukemia, 3rd line | Oncology | Oral | 2016 | 2021/III | ||||||||
| ACZ885 | canakinumab | Anti-interleukin-1 beta monoclonal antibody | 2nd line non-small cell lung cancer | Oncology | Subcutaneous injection | 2017 | 2021/III | ||||||||
1st line non-small cell lung cancer | Oncology | Subcutaneous injection | 2017 | 2021/III | |||||||||||
| Adjuvant non-small cell lung cancer | Oncology | Subcutaneous injection | 2017 | 2022/III | |||||||||||
AVXS-1011 | onasemno- gene abepar- vovec | Survival motor neuron (SMN) gene replacement therapy | Spinal muscular atrophy (IV formulation) | Neuroscience | Intravenous infusion | 2018 | US approved EU registration | ||||||||
Spinal muscular atrophy (IT formulation)2 | Neuroscience | Intrathecal injection | 2018 | 2020/I | |||||||||||
| AVXS-201 | TBD | Methyl-CpG binding protein 2 (MECP2) gene replacement therapy | Rett syndrome | Neuroscience | Intrathecal injection | 2018 | 2023/I | ||||||||
BYL7193 | alpelisib | PI3K-alpha inhibitor | PIK3CA mutant hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-negative (HER2-) postmenopausal advanced breast cancer, 2nd line (+ fulvestrant) | Oncology | Oral | 2018 | US approved EU registration | ||||||||
| PIK3CA-related overgrowth spectrum | Oncology | Oral | 2019 | 2020/III | |||||||||||
| Triple negative breast cancer | Oncology | Oral | 2019 | 2023/III | |||||||||||
| Hormone receptor-negative (HR-)/human epidermal growth factor receptor 2-positive (HER2+) advanced breast cancer | Oncology | Oral | 2019 | 2023/III | |||||||||||
| Ovarian cancer | Oncology | Oral | 2019 | 2023/III | |||||||||||
| Head and neck squamous cell carcinoma | Oncology | Oral | 2019 | ≥2024/III | |||||||||||
| CEE321 | TBD | Pan-JAK inhibitor | Atopic dermatitis | Immunology, Hepatology and Dermatology | Topical | 2019 | ≥2024/II | ||||||||
| CFZ533 | iscalimab | Blocking, non-depleting, anti-CD40 monoclonal antibody | Solid organ transplantation | Immunology, Hepatology and Dermatology | Intravenous infusion | 2017 | 2023/II | ||||||||
| Sjögren's syndrome | Immunology, Hepatology and Dermatology | Intravenous infusion | 2018 | ≥2024/II | |||||||||||
Cosentyx | secukinumab | Anti-interleukin-17 monoclonal antibody | Non-radiographic axial spondyloarthritis | Immunology, Hepatology and Dermatology | Subcutaneous injection | 2015 | US/EU registration | ||||||||
Psoriatic arthritis head-to-head study versus Humira® (adalimumab) | Immunology, Hepatology and Dermatology | Subcutaneous injection | 2015 | 2020/III | |||||||||||
Ankylosing spondylitis head-to-head study versus Sandoz biosimilarHyrimoz (adalimumab) | Immunology, Hepatology and Dermatology | Subcutaneous injection | 2015 | 2022/III | |||||||||||
| Hidradenitis suppurativa | Immunology, Hepatology and Dermatology | Intravenous infusion | 2017 | 2022/III | |||||||||||
| Giant cell arteritis | Immunology, Hepatology and Dermatology | Intravenous infusion | 2018 | ≥2024/II | |||||||||||
| Lichen planus | Immunology, Hepatology and Dermatology | Intravenous infusion | 2019 | ≥2024/II | |||||||||||
| CSJ117 | TBD | Anti-thymic stromal lymphopoietin monoclonal antibody fragment | Severe asthma | Respiratory | Inhalation | 2018 | 2023/II | ||||||||
| ECF843 | TBD | Boundary lubricant | Dry eye | Ophthalmology | Eye drops | 2017 | 2022/II | ||||||||
Entresto | valsartan and sacubitril (as sodium salt complex) | Angiotensin receptor/ neprilysin inhibitor | Chronic heart failure with preserved ejection fraction | Cardiovascular, Renal and Metabolism | Oral | 2012 | 2020/III | ||||||||
| Post-acute myocardial infarction | Cardiovascular, Renal and Metabolism | Oral | 2015 | 2021/III | |||||||||||
1 Approved in the US asZolgensma for spinal muscular atrophy (IV formulation) | |||||||||||||||
2 The FDA has placed a partial clinical hold on AVXS-101 intrathecal trials for spinal muscular atrophy patients based on findings in a small preclinical animal study. | |||||||||||||||
3 Approved in the US asPiqray for PIK3CA mutant HR+/HER2- postmenopausal advanced breast cancer, 2nd line (+ fulvestrant) | |||||||||||||||
Project/ product | Common name | Mechanism of action | Potential indication | Business franchise | Formulation/ route of administration | Year project entered current development phase | Planned filing dates/current phase | ||||||||
| INC280 | capmatinib | c-MET inhibitor | Non-small cell lung cancer | Oncology | Oral | 2014 | US registration | ||||||||
| Solid tumors | Oncology | Oral | 2019 | ≥2024/II | |||||||||||
Jakavi | ruxolitinib | JAK1/2 inhibitor | Acute graft-versus-host disease | Oncology | Oral | 2016 | 2021/III | ||||||||
| Chronic graft-versus-host disease | Oncology | Oral | 2016 | 2021/III | |||||||||||
| KAE609 | cipargamin | PfATP4 inhibitor | Malaria | Established Medicines | Oral | 2012 | ≥2024/II | ||||||||
| Severe malaria | Established Medicines | Oral | 2019 | ≥2024/II | |||||||||||
| KAF156 | ganaplacide | Imidazolopiperazines derivative | Malaria | Established Medicines | Oral | 2014 | ≥2024/II | ||||||||
Kisqali | ribociclib | CDK4/6 inhibitor | HR+/HER2- breast cancer (adjuvant) | Oncology | Oral | 2018 | 2022/III | ||||||||
| KJX839 | inclisiran | Small-interfering RNA (PCSK9) | Hyperlipidemia | Cardiovascular, Renal and Metabolism | Subcutaneous injection | 2019 | US/EU registration | ||||||||
| Secondary prevention of cardiovascular events in patients with elevated levels of LDL-C | Cardiovascular, Renal and Metabolism | Subcutaneous injection | 2019 | ≥2024/III | |||||||||||
Kymriah | tisagen- lecleucel | CD19-targeted chimeric antigen receptor T-cell immunotherapy | Relapsed/refractory follicular lymphoma | Oncology | Intravenous infusion | 2017 | 2021/II | ||||||||
Relapsed/refractory diffuse large B-cell lymphoma in 1st relapse | Oncology | Intravenous infusion | 2018 | 2021/III | |||||||||||
| Relapsed/refractory diffuse large B-cell lymphoma (+ pembrolizumab) | Oncology | Intravenous infusion | 2017 | ≥2024/II | |||||||||||
| LAM320 | clofazimine | Mycobacterial DNA binding | Multidrug-resistant tuberculosis | Established Medicines | Oral | 2016 | 2021/III | ||||||||
| LJC242 | tropifexor, cenicriviroc (in fixed-dose combination) | FXR agonist and CCR2/5 inhibitor | Nonalcoholic steatohepatitis | Immunology, Hepatology and Dermatology | Oral | 2017 | ≥2024/II | ||||||||
| LJN452 | tropifexor | FXR agonist | Nonalcoholic steatohepatitis | Immunology, Hepatology and Dermatology | Oral | 2015 | ≥2024/II | ||||||||
| LMI070 | branaplam | SMN2 RNA splicing modulator | Spinal muscular atrophy | Neuroscience | Oral | 2017 | ≥2024/II | ||||||||
| LNP023 | TBD | Factor B inhibitor | IgA nephropathy | Cardiovascular, Renal and Metabolism | Oral | 2018 | 2023/II | ||||||||
| C3 glomerulopathy | Cardiovascular, Renal and Metabolism | Oral | 2018 | 2023/II | |||||||||||
| Paroxysmal nocturnal hemoglobinuria | Cardiovascular, Renal and Metabolism | Oral | 2019 | 2023/II | |||||||||||
| Membranous nephropathy | Cardiovascular, Renal and Metabolism | Oral | 2018 | ≥2024/II | |||||||||||
| LOU064 | TBD | BTK inhibitor | Chronic spontaneous urticaria | Immunology, Hepatology and Dermatology | Oral | 2017 | 2023/II | ||||||||
177Lu- PSMA-617 | TBD | Targeted DNA destruction via beta-particle radiation | Metastatic castration-resistant prostate cancer | Oncology | Intravenous infusion | 2018 | 2020/III | ||||||||
| LXE408 | TBD | Kinetoplastid proteasome inhibitor | Visceral leishmaniasis | Established Medicines | Oral | 2019 | ≥2024/II | ||||||||
| MBG453 | TBD | TIM-3 antagonist | Myelodysplastic syndrome | Oncology | Intravenous infusion | 2018 | 2021/II | ||||||||
| Acute myeloid leukemia | Oncology | Intravenous infusion | 2019 | ≥2024/II | |||||||||||
| OMB157 | ofatumumab | Anti-CD20 monoclonal antibody | Relapsing multiple sclerosis | Neuroscience | Subcutaneous injection | 2015 | US/EU registration | ||||||||
| PDR001 | spartalizumab | Anti-PD-1 monoclonal antibody | Metastatic BRAF V600+ melanoma (w/ Tafinlar +Mekinist) | Oncology | Intravenous infusion | 2017 | 2020/III | ||||||||
| Metastatic melanoma (combo) | Oncology | Intravenous infusion | 2017 | 2023/II | |||||||||||
| QBW251 | TBD | CFTR potentiator | Chronic obstructive pulmonary disease | Respiratory | Oral | 2017 | ≥2024/II | ||||||||
| QGE031 | ligelizumab | High-affinity anti-IgE monoclonal antibody | Chronic spontaneous urticaria/ chronic idiopathic urticaria | Immunology, Hepatology and Dermatology | Subcutaneous injection | 2017 | 2021/III | ||||||||
| QMF149 | indacaterol, mometasone furoate (in fixed-dose combination) | Long-acting beta2- adrenergic agonist and inhaled corticosteroid | Asthma | Respiratory | Inhalation | 2019 | EU registration | ||||||||
Project/ product | Common name | Mechanism of action | Potential indication | Business franchise | Formulation/ route of administration | Year project entered current development phase | Planned filing dates/current phase | ||||||||
| QVM149 | indacaterol, mometasone furoate, glyco- pyrronium bromide (in fixed-dose combination) | Long-acting beta2- adrenergic agonist, long-acting muscarinic antagonist and inhaled corticosteroid | Asthma | Respiratory | Inhalation | 2019 | EU registration | ||||||||
RTH2584 | brolucizumab | Anti-VEGF single-chain antibody fragment | Neovascular (wet) age-related macular degeneration | Ophthalmology | Intravitreal injection | 2019 | US approved EU registration | ||||||||
| Diabetic macular edema | Ophthalmology | Intravitreal injection | 2017 | 2021/III | |||||||||||
| Retinal vein occlusion | Ophthalmology | Intravitreal injection | 2018 | 2023/III | |||||||||||
| Proliferative diabetic retinopathy | Ophthalmology | Intravitreal injection | 2019 | 2023/III | |||||||||||
| SAF312 | TBD | TRPV1 antagonist | Chronic ocular surface pain | Ophthalmology | Topical | 2019 | ≥2024/II | ||||||||
SEG1015 | crizanlizumab | P-selectin inhibitor | Sickle cell disease | Oncology | Intravenous infusion | 2019 | US approved EU registration | ||||||||
| TQJ230 | TBD | Anti-apo(a) antisense oligonucleotide | Secondary prevention of cardiovascular events in patients with elevated levels of lipoprotein(a) | Cardiovascular, Renal and Metabolism | Subcutaneous injection | 2018 | ≥2024/III | ||||||||
| UNR844 | TBD | Reduction of disulfide bonds | Presbyopia | Cardiovascular, Renal and Metabolism | Eye drops | 2017 | ≥2024/II | ||||||||
| VAY736 | ianalumab | Anti-BAFF (B-cell- activating factor) monoclonal antibody | Autoimmune hepatitis | Immunology, Hepatology and Dermatology | Subcutaneous injection | 2016 | ≥2024/II | ||||||||
| Primary Sjögren’s syndrome | Immunology, Hepatology and Dermatology | Subcutaneous injection | 2015 | ≥2024/II | |||||||||||
| VPM087 | TBD | Interleukin-1 beta neutralization monoclonal antibody | Colorectal cancer, 1st line; renal cell carcinoma, 1st line | Oncology | Intravenous infusion | 2018 | ≥2024/I | ||||||||
Xolair | omalizumab | Anti-IgE monoclonal antibody | Nasal polyps | Respiratory | Subcutaneous injection | 2017 | US/EU registration | ||||||||
| Food allergy | Respiratory | Subcutaneous injection | 2019 | 2021/III | |||||||||||
| ZPL389 | adriforant | Histamine H4 receptor antagonist | Atopic dermatitis | Immunology, Hepatology and Dermatology | Oral | 2017 | ≥2024/II | ||||||||
4 Approved in the US asBeovu for neovascular (wet) age-related macular degeneration | |||||||||||||||
5 Approved in the US asAdakveo for sickle cell disease | |||||||||||||||
| Project/product | Potential indication | Change | Reason | ||||
| ABL001 | Chronic myeloid leukemia, 1st line | Removed | Development discontinued | ||||
| AVXS-101 | Spinal muscular atrophy type 1 (IV formulation) | Now disclosed as spinal muscular atrophy (IV formulation) | |||||
| Spinal muscular atrophy type 2/3 (IT formulation) | Now disclosed as spinal muscular atrophy (IT formulation) | ||||||
| BAF312 | Secondary progressive multiple sclerosis | Commercialized asMayzent | |||||
| BYL719 | Hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-negative (HER2-) advanced breast cancer (postmenopausal women), 2nd line (+ fulvestrant) | Now disclosed as PIK3CA mutant hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-negative (HER2-) postmenopausal advanced breast cancer, 2nd line (+ fulvestrant) | |||||
| PIK3CA-related overgrowth spectrum | Added | Entered Confirmatory Development | |||||
| Triple negative breast cancer | Added | Entered Confirmatory Development | |||||
| Hormone receptor-negative (HR-)/human epidermal growth factor receptor 2-positive (HER2+) advanced breast cancer | Added | Entered Confirmatory Development | |||||
| Ovarian cancer | Added | Entered Confirmatory Development | |||||
| Head and neck squamous cell carcinoma | Added | Entered Confirmatory Development | |||||
| CAD106 | Alzheimer’s disease | Removed | Development discontinued | ||||
| CEE321 | Atopic dermatitis | Added | Entered Confirmatory Development | ||||
| CNP520 | Alzheimer’s disease | Removed | Development discontinued | ||||
Cosentyx | Giant cell arteritis | Added | Entered Confirmatory Development | ||||
| Lichen planus | Added | Entered Confirmatory Development | |||||
| EMA401 | Peripheral neuropathic pain | Removed | Development discontinued | ||||
| HDM201 | Acute myeloid lymphoma | Removed | Development discontinued | ||||
| INC280 | Non-small cell lung cancer (EGFR mutation) | Removed | Development discontinued | ||||
| Solid tumors | Added | Entered Confirmatory Development | |||||
| KAE609 | Severe malaria | Added | Entered Confirmatory Development | ||||
| KJX839 | Hyperlipidemia | Added | Acquired with acquisition of The Medicines Company | ||||
| Secondary prevention of cardiovascular events in patients with elevated levels of LDL-C | Added | Acquired with acquisition of The Medicines Company | |||||
Kymriah | Chronic lymphocytic leukemia | Removed | Development discontinued | ||||
| LCI699 | Cushing's disease | Removed | Divested to Recordati S.p.A. | ||||
| LNP023 | C3 glomerulopathy | Added | Entered Confirmatory Development | ||||
| Paroxysmal nocturnal hemoglobinuria | Added | Entered Confirmatory Development | |||||
Lucentis | Retinopathy of prematurity | Commercialized | |||||
| Diabetic retinopathy | Commercialized | ||||||
| LXE408 | Visceral leishmaniasis | Added | Entered Confirmatory Development | ||||
| MBG453 | Myelodysplastic syndrome | Added | Entered Confirmatory Development | ||||
| Acute myeloid leukemia | Added | Entered Confirmatory Development | |||||
| MOR106 | Atopic dermatitis | Removed | Development discontinued | ||||
| PDR001 | Malignant melanoma (combo) | Now disclosed as metastatic melanoma (combo) | |||||
Promacta/Revolade | Severe aplastic anemia, 1st line | Removed | Development discontinued | ||||
| QAW039 | Asthma | Removed | Development discontinued | ||||
| RTH258 | Neovascular age-related macular degeneration | Now disclosed as neovascular (wet) age-related macular degeneration | |||||
| Proliferative diabetic retinopathy | Added | Entered Confirmatory Development | |||||
Rydapt | Acute myeloid leukemia (FLT3 wild type) | Removed | Development discontinued | ||||
| SAF312 | Chronic ocular surface pain | Added | Entered Confirmatory Development | ||||
| TQJ230 | Secondary prevention of cardiovascular events in patients with elevated levels of lipoprotein(a) | Added | Entered Confirmatory Development | ||||
| VAY785 | Nonalcoholic steatohepatitis | Removed | Development discontinued | ||||
Xolair | Food allergy | Added | Entered Confirmatory Development | ||||
| 2019 net sales to third parties | |||||
| USD millions | % | ||||
| United States | 13 789 | 37 | |||
| Europe | 12 818 | 34 | |||
| Asia, Africa, Australasia | 8 458 | 22 | |||
| Canada and Latin America | 2 649 | 7 | |||
| Total | 37 714 | 100 | |||
Of which in Established Markets * | 28 573 | 76 | |||
Of which in Emerging Growth Markets * | 9 141 | 24 | |||
* Emerging Growth Markets comprise all markets other than the Established Markets of the US, Canada, Western Europe, Japan, Australia and New Zealand. | |||||
Sandoz
| Product | Originator drug | Description | |||
| Amoxicillin/clavulanic acid | Augmentin® | Antibiotic | |||
| Zoledronic acid | Aclasta | Osteoporosis treatment | |||
| Acetylsteine | Various | Mucolytic agent | |||
| Fentanyl | Various | Pain treatment | |||
| Active ingredients | Description | ||
| Oral and sterile penicillins | Anti-infectives | ||
| Oral and sterile cephalosporins | Anti-infectives | ||
| Clavulanic acid and mixtures with clavulanic acid | ß-lactam inhibitors | ||
| Classical and semisynthetic erythromycins | Anti-infectives | ||
| Intermediates | Description | ||
| Various cephalosporin intermediates | Anti-infectives | ||
| Erythromycin base | Anti-infectives | ||
| Various crude compounds produced by fermentation | Cyclosporine, ascomysin, rapamycin, mycophenolic acid, etc. | ||
| Product | Originator drug | Description | |||
| Omnitrope | Genotropin® | Recombinant human growth hormone | |||
Binocrit and Epoetin alfaHexal | Eprex®/Erypo® | Recombinant protein used for anemia | |||
Zarzio, Zarxio andFilgrastim Hexal | Neupogen® | Recombinant protein used in oncology | |||
| Glatopa | Copaxone® | Treatment for multiple sclerosis (MS) | |||
Erelzi 1 | Enbrel® | Treatment for multiple inflammatory diseases | |||
| Rixathon | MabThera® | Treatment for blood cancers and immunological diseases | |||
| Hyrimoz | Humira® | Treatment for multiple inflammatory diseases | |||
| Zessly | Remicade® | Treatment for gastroenterological, rheumatological and dermatological diseases | |||
| Ziextenzo | Neulasta® | Treatment to reduce duration of chemotherapy-induced neutropenia and incidence of chemotherapy-induced febrile neutropenia with the exception of chronic myeloid leukemia and myelodysplastic syndromes | |||
1 Approved in the US in 2016. Launch in the US pending final resolution of litigation with Amgen, which markets Enbrel®. The US District Court of New Jersey ruled against Sandoz on August 9, 2019; Sandoz respectfully disagrees with the ruling and submitted an appeal. The appeals court hearing is scheduled for March 4, 2020. | |||||
Project/ product 1 | Common name | Mechanism of action | Potential indication/indications | Therapeutic areas | Route of administration | Current phase | |||||||
| GP2017 | adalimumab | TNF-α inhibitor | Arthritides (rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis), plaque psoriasis and others (same as originator) | Immunology | Subcutaneous | EU: Approved US: Approved2 | |||||||
GP2411 3 | denosumab | RANKL inhibitor | Osteoporosis, bone loss, prevention of bone complications in cancer that has spread to the bone (indications vary in US and EU) | Endocrinology, Neurology | Subcutaneous | Phase III | |||||||
EGI014A1 4 | trastuzumab | Anti HER2 monoclonal antibody | Breast and gastric tumors | Oncology | Intravenous | Phase III | |||||||
DST356A1 5 | natalizumab | Anti-Alpha 4 (α4) integrin monoclonal antibody | Monotherapy for remitting relapsing forms of multiple sclerosis (RRMS); in US second line treatment for active Crohn’s disease | Neurology, Immunology (US only) | Intravenous | Phase III | |||||||
1 LA-EP2006 (pegfilgrastim) was approved and launched in the EU asZiextenzo in November 2018 and was approved and launched in the US in November 2019. | |||||||||||||
2 Launched asHyrimoz in the EU in October 2018. Also in October 2018, we announced a global resolution of all intellectual property-related litigation with AbbVie concerning adalimumab. Under the terms of the agreement, AbbVie grants us a non-exclusive license to AbbVie’s intellectual property relating to Humira®, beginning on certain dates in certain countries in which AbbVie has intellectual property. We are not entitled to launchHyrimoz in the US until the second half of 2023. | |||||||||||||
3 Development in collaboration with Hexal AG. | |||||||||||||
4 Development in collaboration with EirGenix, Inc. | |||||||||||||
5 Development in collaboration with Polpharma Biologics. | |||||||||||||
| 2019 net sales to third parties | |||||
| USD millions | % | ||||
| Europe | 5 115 | 53 | |||
| United States | 2 491 | 26 | |||
| Asia, Africa, Australasia | 1 341 | 14 | |||
| Canada and Latin America | 784 | 7 | |||
| Total | 9 731 | 100 | |||
Of which in Established Markets * | 7 111 | 73 | |||
Of which in Emerging Growth Markets * | 2 620 | 27 | |||
* Emerging Growth Markets comprise all markets other than the Established Markets of the US, Canada, Western Europe, Japan, Australia and New Zealand. | |||||
4.C Organizational structure
4.D Property, plants and equipment
Location | Size of site (in square meters) | Major activity | |||
| Basel, Switzerland – St. Johann | 589 000 | Global Group headquarters; global Innovative Medicines Division headquarters; Global Sandoz Division; research and development; production of drug substances and drug intermediates | |||
| Kundl and Schaftenau, Austria | 480 000 | Production of biotechnological products, drug products and finished products, anti-infectives, active drug substances, product development | |||
| East Hanover, New Jersey | 391 000 | Innovative Medicines Division US headquarters, research and development | |||
| Barleben, Germany | 340 000 | Production of broad range of generics finished dosage forms | |||
| Cambridge, Massachusetts | 205 000 | Research and development | |||
| Shanghai, China | 106 500 | Research and development | |||
| Ljubljana, Slovenia | 83 000 | Production of broad range of finished solid and sterile dosage forms | |||
| Hyderabad, India | 80 500 | General administrative and development global service center | |||
| Longmont, Colorado | 65 032 | Production, warehouse, and administrative offices for AveXis | |||
| Stein, Switzerland | 64 700 | Production of sterile vials, pre-filled syringes and ampoules; inhalation capsules, tablets and transdermals; active pharmaceutical ingredients, and cell and gene therapies | |||
| Holzkirchen, Germany | 64 200 | Global Sandoz Division headquarters, production of oral films, transdermal delivery systems, matrix patches, product development | |||
| Menges, Slovenia | 62 400 | Production of drug substances and drug intermediates | |||
| Stryków, Poland | 45 000 | Production of broad range of bulk oral solid forms and packaging | |||
| Huningue, France | 35 000 | Production of drug substances for clinical and commercial supply | |||
| Singapore | 35 000 | Production for Innovative Medicines solids and biologics | |||
| Barbera, Spain | 33 000 | Production of tablets, capsules and inhalation products | |||
| Basel, Switzerland – Schweizerhalle | 31 700 | Production of drug substances and drug intermediates | |||
| Rueil-Malmaison, France | 29 500 | Administrative offices for Innovative Medicines | |||
| Puurs, Belgium | 27 500 | Production for Innovative Medicines ophthalmic products | |||
| Tokyo, Japan | 20 000 | Administrative offices for Innovative Medicines and Sandoz | |||
| Morris Plains, New Jersey | 15 600 | Production for Innovative Medicines Division cell and gene therapies | |||
| Princeton, New Jersey | 14 300 | Sandoz Division US headquarters | |||
| Libertyville, Illinois | 9 800 | Production, warehouse, and administrative offices for AveXis | |||
| Targu Mures, Romania | 9 070 | Production of solids for Innovative Medicines and Sandoz | |||
| Les Ulis, France | 5 920 | Production for Innovative Medicines Division cell and gene therapies | |||
| Millburn, New Jersey | 1 400 | AAA primary production site for radioligand therapy | |||
| Colleretto Giacosa/Ivrea, Italy | 1 200 | AAA primary production site for radioligand therapy | |||
Item 4A. Unresolved Staff Comments
Item 5. Operating and Financial Review and Prospects
5.A Operating results
(USD millions unless indicated otherwise) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % 2 | |||||
| Net sales to third parties from continuing operations | 47 445 | 44 751 | 6 | 9 | |||||
| Sales to discontinued operations | 53 | 82 | – 35 | – 31 | |||||
| Net sales from continuing operations | 47 498 | 44 833 | 6 | 9 | |||||
| Other revenues | 1 179 | 1 266 | – 7 | – 7 | |||||
| Cost of goods sold | – 14 425 | – 14 510 | 1 | – 2 | |||||
| Gross profit from continuing operations | 34 252 | 31 589 | 8 | 12 | |||||
| Selling, general and administration | – 14 369 | – 13 717 | – 5 | – 8 | |||||
| Research and development | – 9 402 | – 8 489 | – 11 | – 13 | |||||
| Other income | 2 031 | 1 629 | 25 | 27 | |||||
| Other expense | – 3 426 | – 2 609 | – 31 | – 33 | |||||
| Operating income from continuing operations | 9 086 | 8 403 | 8 | 14 | |||||
| Return on net sales (%) | 19.2 | 18.8 | |||||||
| Income from associated companies | 659 | 6 438 | nm | nm | |||||
| Interest expense | – 850 | – 932 | 9 | 8 | |||||
| Other financial income and expense | 45 | 186 | – 76 | – 69 | |||||
| Income before taxes from continuing operations | 8 940 | 14 095 | – 37 | – 33 | |||||
| Taxes | – 1 793 | – 1 295 | – 38 | – 46 | |||||
| Net income from continuing operations | 7 147 | 12 800 | – 44 | – 41 | |||||
| Net loss from discontinued operations before gain on distribution of Alcon Inc. to Novartis AG shareholders | – 101 | – 186 | nm | nm | |||||
| Gain on distribution of Alcon Inc. to Novartis AG shareholders | 4 691 | ||||||||
| Net income/(loss) from discontinued operations | 4 590 | – 186 | nm | nm | |||||
| Net income | 11 737 | 12 614 | – 7 | – 3 | |||||
| Attributable to: | |||||||||
| Shareholders of Novartis AG | 11 732 | 12 611 | – 7 | – 3 | |||||
| Non-controlling interests | 5 | 3 | nm | nm | |||||
| Basic earnings per share from continuing operations (USD) | 3.12 | 5.52 | – 43 | – 40 | |||||
| Basic earnings per share from discontinued operations (USD) | 2.00 | – 0.08 | nm | nm | |||||
| Total basic earnings per share (USD) | 5.12 | 5.44 | – 6 | – 2 | |||||
| Net cash flows from operating activities from continuing operations | 13 547 | 13 049 | 4 | ||||||
Free cash flow from continuing operations 2 | 12 937 | 11 256 | 15 | ||||||
1 Continuing operations include the businesses of the Innovative Medicines and Sandoz divisions and the continuing Corporate activities and discontinued operations include the Alcon eye care devices business and certain Corporate activities attributable to the Alcon business prior to the spin-off, the gain on distribution of Alcon Inc. to Novartis AG shareholders in 2019 and certain other expenses related to the distribution. See “Item 18. Financial Statements—Note 1. Significant accounting principles”, “Item 18.Financial Statements—Note 2. Significant transactions—Significant transactions in 2019,” and “Item 18.Finanacial Statements—Note 30. Discontinued operations.” | |||||||||
2 For an explanation of non-IFRS measures and reconciliation tables, see "Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
| nm = not meaningful | |||||||||
(USD millions) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % | |||||
| Innovative Medicines | 37 714 | 34 892 | 8 | 11 | |||||
| Sandoz | 9 731 | 9 859 | – 1 | 2 | |||||
| Net sales to third parties from continuing operations | 47 445 | 44 751 | 6 | 9 | |||||
(USD millions) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % | |||||
| Total Novartis Oncology business unit | 14 370 | 13 428 | 7 | 10 | |||||
| Total Novartis Pharmaceuticals business unit | 23 344 | 21 464 | 9 | 12 | |||||
| Ophthalmology | 4 776 | 4 558 | 5 | 8 | |||||
| Immunology, Hepatology and Dermatology | 4 222 | 3 392 | 24 | 27 | |||||
| Neuroscience | 3 773 | 3 429 | 10 | 13 | |||||
| Respiratory | 1 825 | 1 767 | 3 | 9 | |||||
| Cardiovascular, Renal and Metabolism | 1 750 | 1 050 | 67 | 70 | |||||
| Established Medicines | 6 998 | 7 268 | – 4 | 0 | |||||
| Total Innovative Medicines | 37 714 | 34 892 | 8 | 11 | |||||
| US | Rest of world | Total | |||||||||||||||||||
Brands | Business franchise | Indication | USD m | % change USD/cc 2 | USD m | % change USD | % change cc 2 | USD m | % change USD | % change cc 2 | |||||||||||
| Cosentyx | Immunology, Hepatology and Dermatology | Psoriasis, ankylosing spondylitis and psoriatic arthritis | 2 220 | 33 | 1 331 | 14 | 20 | 3 551 | 25 | 28 | |||||||||||
| Gilenya | Neuroscience | Relapsing multiple sclerosis | 1 736 | – 2 | 1 487 | – 6 | 0 | 3 223 | – 4 | – 1 | |||||||||||
| Lucentis | Ophthalmology | Age-related macular degeneration | 2 086 | 2 | 7 | 2 086 | 2 | 7 | |||||||||||||
| Tasigna | Oncology | Chronic myeloid leukemia | 804 | 0 | 1 076 | 1 | 5 | 1 880 | 0 | 3 | |||||||||||
| Entresto | Cardiovascular, Renal and Metabolism | Chronic heart failure | 925 | 66 | 801 | 70 | 77 | 1 726 | 68 | 71 | |||||||||||
| Sandostatin | Oncology | Carcinoid tumors and acromegaly | 881 | 8 | 704 | – 9 | – 3 | 1 585 | 0 | 2 | |||||||||||
| Afinitor/Votubia | Oncology | Breast cancer/TSC | 1 003 | 8 | 536 | – 15 | – 10 | 1 539 | – 1 | 1 | |||||||||||
| Promacta/Revolade | Oncology | Immune thrombocytopenia (ITP), severe aplastic anemia (SAA) | 691 | 19 | 725 | 22 | 27 | 1 416 | 21 | 23 | |||||||||||
Tafinlar +Mekinist | Oncology | BRAF V600+ metastatic and adjuvant melanoma; advanced non-small cell lung cancer (NSCLC) | 481 | 5 | 857 | 23 | 30 | 1 338 | 16 | 20 | |||||||||||
Galvus Group | Established Medicines | Diabetes | 1 297 | 1 | 5 | 1 297 | 1 | 5 | |||||||||||||
| Gleevec/Glivec | Oncology | Chronic myeloid leukemia and GIST | 334 | – 24 | 929 | – 17 | – 14 | 1 263 | – 19 | – 17 | |||||||||||
Xolair 1 | Respiratory | Severe Allergic Asthma (SAA) and Chronic Spontaneous Urticaria (CSU) | 1 173 | 13 | 19 | 1 173 | 13 | 19 | |||||||||||||
| Jakavi | Oncology | Myelofibrosis (MF), polycytomia vera (PV) | 1 114 | 14 | 20 | 1 114 | 14 | 20 | |||||||||||||
Diovan Group | Established Medicines | Hypertension | 86 | 2 | 978 | 4 | 10 | 1 064 | 4 | 9 | |||||||||||
Exforge Group | Established Medicines | Hypertension | 13 | – 32 | 1��012 | 3 | 8 | 1 025 | 2 | 7 | |||||||||||
| Exjade/Jadenu | Oncology | Chronic iron overload | 450 | – 14 | 525 | – 9 | – 6 | 975 | – 11 | – 9 | |||||||||||
| Votrient | Oncology | Renal cell carcinoma | 332 | – 18 | 423 | 0 | 5 | 755 | – 9 | – 6 | |||||||||||
| Ilaris | Immunology, Hepatology and Dermatology | Auto-inflammatory (CAPS, TRAPS, HIDS/MKD, FMF, SJIA, AOSD and gout) | 304 | 16 | 367 | 26 | 33 | 671 | 21 | 25 | |||||||||||
| Zortress/Certican | Established Medicines | Transplantation | 169 | 17 | 316 | – 1 | 4 | 485 | 5 | 8 | |||||||||||
| Kisqali | Oncology | HR+/HER2- metastatic breast cancer | 250 | 45 | 230 | nm | nm | 480 | 104 | 111 | |||||||||||
| Top 20 products total | 10 679 | 11 | 17 967 | 5 | 11 | 28 646 | 7 | 11 | |||||||||||||
| Rest of portfolio | 3 110 | 39 | 5 958 | – 1 | 4 | 9 068 | 10 | 13 | |||||||||||||
| Total division sales | 13 789 | 16 | 23 925 | 4 | 9 | 37 714 | 8 | 11 | |||||||||||||
1 Net sales reflectXolair sales for all indications. | |||||||||||||||||||||
2 Constant currencies (cc) is a non-IFRS measure. For an explanation of non-IFRS measures, see " —Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||||||||||||||
(USD millions) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % | |||||
Retail Generics1 | 7 590 | 7 880 | – 4 | 0 | |||||
| Biopharmaceuticals | 1 607 | 1 436 | 12 | 16 | |||||
| Anti-Infectives (partner label/API) | 534 | 543 | – 2 | 2 | |||||
| Total Sandoz | 9 731 | 9 859 | – 1 | 2 | |||||
1 Of which USD 784 million (2018: USD 826 million) represents anti-infectives sold under the Sandoz name | |||||||||
(USD millions) | Year ended Dec 31, 2019 | % of net sales | Year ended Dec 31, 2018 | % of net sales | Change in USD % | Change in constant currencies % | |||||||
| Innovative Medicines | 9 287 | 24.6 | 7 871 | 22.6 | 18 | 24 | |||||||
| Sandoz | 551 | 5.7 | 1 332 | 13.5 | – 59 | – 53 | |||||||
| Corporate | – 752 | – 800 | 6 | 4 | |||||||||
| Operating income from continuing operations | 9 086 | 19.2 | 8 403 | 18.8 | 8 | 14 | |||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % | |||||
| Core gross profit from continuing operations | 37 392 | 34 886 | 7 | 10 | |||||
| Selling, general and administration | – 14 319 | – 13 690 | – 5 | – 7 | |||||
| Research and development | – 8 386 | – 8 154 | – 3 | – 5 | |||||
| Other income | 495 | 558 | – 11 | – 9 | |||||
| Other expense | – 1 070 | – 1 043 | – 3 | – 5 | |||||
| Core operating income from continuing operations | 14 112 | 12 557 | 12 | 17 | |||||
| As % of net sales | 29.7 | 28.1 | |||||||
1 For an explanation of non-IFRS measures and reconciliation tables, see "Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
(USD millions) | Year ended Dec 31, 2019 | % of net sales | Year ended Dec 31, 2018 | % of net sales | Change in USD % | Change in constant currencies % | |||||||
| Innovative Medicines | 12 650 | 33.5 | 11 151 | 32.0 | 13 | 18 | |||||||
| Sandoz | 2 094 | 21.5 | 2 002 | 20.3 | 5 | 10 | |||||||
| Corporate | – 632 | – 596 | – 6 | – 9 | |||||||||
| Core operating income from continuing operations | 14 112 | 29.7 | 12 557 | 28.1 | 12 | 17 | |||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % | |||||
| Research and exploratory development | – 2 855 | – 2 770 | – 3 | – 4 | |||||
| Confirmatory development | – 5 297 | – 4 905 | – 8 | – 10 | |||||
| Total Innovative Medicines Division research and development expense | – 8 152 | – 7 675 | – 6 | – 8 | |||||
| As % of Innovative Medicines net sales to third parties | 21.6 | 22.0 | |||||||
Core research and exploratory development1 | – 2 706 | – 2 665 | – 2 | – 2 | |||||
Core confirmatory development1 | – 4 879 | – 4 675 | – 4 | – 6 | |||||
| Total core Innovative Medicines Division research and development expense | – 7 585 | – 7 340 | – 3 | – 5 | |||||
| As % of Innovative Medicines net sales to third parties | 20.1 | 21.0 | |||||||
1 Core excludes impairments, amortization and certain other items. For an explanation of non-IFRS measures and reconciliation tables, see " —Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % | |||||
| Operating income from continuing operations | 9 086 | 8 403 | 8 | 14 | |||||
| Income from associated companies | 659 | 6 438 | nm | nm | |||||
| Interest expense | – 850 | – 932 | 9 | 8 | |||||
| Other financial income and expense | 45 | 186 | – 76 | – 69 | |||||
| Income before taxes from continuing operations | 8 940 | 14 095 | – 37 | – 33 | |||||
| Taxes | – 1 793 | – 1 295 | – 38 | – 46 | |||||
| Net income from continuing operations | 7 147 | 12 800 | – 44 | – 41 | |||||
| Net loss from discontinued operations before gain on distribution of Alcon Inc. to Novartis AG shareholders | – 101 | – 186 | nm | nm | |||||
| Gain on distribution of Alcon Inc. to Novartis AG shareholders | 4 691 | ||||||||
| Net income from discontinued operations | 4 590 | – 186 | nm | nm | |||||
| Net income | 11 737 | 12 614 | – 7 | – 3 | |||||
| Attributable to: | |||||||||
| Shareholders of Novartis AG | 11 732 | 12 611 | – 7 | – 3 | |||||
| Non-controlling interests | 5 | 3 | nm | nm | |||||
| Basic earnings per share from continuing operations (USD) | 3.12 | 5.52 | – 43 | – 40 | |||||
| Basic earnings per share from discontinued operations (USD) | 2.00 | – 0.08 | nm | nm | |||||
| Total basic earnings per share (USD) | 5.12 | 5.44 | – 6 | – 2 | |||||
| nm = not meaningful | |||||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2019 | Year ended Dec 31, 2018 | Change in USD % | Change in constant currencies % | |||||
| Core operating income from continuing operations | 14 112 | 12 557 | 12 | 17 | |||||
| Core income from associated companies | 1 086 | 1 113 | – 2 | – 2 | |||||
| Core interest expense | – 850 | – 932 | 9 | 8 | |||||
| Core other financial income and expense | 56 | 186 | – 70 | – 63 | |||||
| Core income before taxes from continuing operations | 14 404 | 12 924 | 11 | 16 | |||||
| Core taxes | – 2 300 | – 2 004 | – 15 | – 19 | |||||
| Core net income from continuing operations | 12 104 | 10 920 | 11 | 15 | |||||
| Core basic earnings per share from continuing operations (USD) | 5.28 | 4.71 | 12 | 17 | |||||
1 For an explanation of non-IFRS measures and reconciliation tables, see "Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % 2 | |||||
| Net sales to third parties from continuing operations | 44 751 | 42 338 | 6 | 5 | |||||
| Sales to discontinued operations | 82 | 43 | 91 | 76 | |||||
| Net sales from continuing operations | 44 833 | 42 381 | 6 | 5 | |||||
| Other revenues | 1 266 | 1 023 | 24 | 24 | |||||
| Cost of goods sold | – 14 510 | – 13 633 | – 6 | – 5 | |||||
| Gross profit from continuing operations | 31 589 | 29 771 | 6 | 6 | |||||
| Selling, general and administration | – 13 717 | – 12 465 | – 10 | – 9 | |||||
| Research and development | – 8 489 | – 8 389 | – 1 | 0 | |||||
| Other income | 1 629 | 1 922 | – 15 | – 16 | |||||
| Other expense | – 2 609 | – 2 137 | – 22 | – 21 | |||||
| Operating income from continuing operations | 8 403 | 8 702 | – 3 | – 3 | |||||
| Return on net sales (%) | 18.8 | 20.6 | |||||||
| Income from associated companies | 6 438 | 1 108 | nm | nm | |||||
| Interest expense | – 932 | – 750 | – 24 | – 27 | |||||
| Other financial income and expense | 186 | 42 | nm | nm | |||||
| Income before taxes from continuing operations | 14 095 | 9 102 | 55 | 55 | |||||
| Taxes | – 1 295 | – 1 603 | 19 | 19 | |||||
| Net income from continuing operations | 12 800 | 7 499 | 71 | 71 | |||||
| Net (loss)/income from discontinued operations | – 186 | 204 | nm | nm | |||||
| Net income | 12 614 | 7 703 | 64 | 64 | |||||
| Attributable to: | |||||||||
| Shareholders of Novartis AG | 12 611 | 7 703 | 64 | 64 | |||||
| Non-controlling interests | 3 | 0 | nm | nm | |||||
| Basic earnings per share from continuing operations (USD) | 5.52 | 3.20 | 73 | 73 | |||||
| Basic earnings per share from discontinued operations (USD) | – 0.08 | 0.08 | nm | nm | |||||
| Total basic earnings per share (USD) | 5.44 | 3.28 | 66 | 66 | |||||
| Net cash flows from operating activities from continuing operations | 13 049 | 11 419 | 14 | ||||||
Free cash flow from continuing operations 2 | 11 256 | 9 791 | 15 | ||||||
1 Continuing operations include the businesses of the Innovative Medicines and Sandoz divisions and the continuing Corporate activities and discontinued operations include the Alcon eye care devices business and certain Corporate activities attributable to the Alcon business prior to the spin-off and certain other expenses related to the distribution. See “Item 18. Financial Statements—Note 1. Significant accounting principles”, “Item 18.Financial Statements—Note 2. Significant transactions—Significant transactions in 2019,” and “Item 18.Finanacial Statements—Note 30. Discontinued operations.” | |||||||||
2 For an explanation of non-IFRS measures and reconciliation tables, see "Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
| nm = not meaningful | |||||||||
(USD millions) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % | |||||
| Innovative Medicines | 34 892 | 32 278 | 8 | 8 | |||||
| Sandoz | 9 859 | 10 060 | – 2 | – 3 | |||||
| Net sales to third parties from continuing operations | 44 751 | 42 338 | 6 | 5 | |||||
(USD millions) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % | |||||
| Total Novartis Oncology business unit | 13 428 | 12 274 | 9 | 9 | |||||
| Total Novartis Pharmaceuticals business unit | 21 464 | 20 004 | 7 | 7 | |||||
| Ophthalmology | 4 558 | 4 621 | – 1 | – 2 | |||||
| Neuroscience | 3 429 | 3 287 | 4 | 4 | |||||
| Immunology, Hepatology and Dermatology | 3 392 | 2 474 | 37 | 37 | |||||
| Respiratory | 1 767 | 1 617 | 9 | 8 | |||||
| Cardiovascular, Renal and Metabolism | 1 050 | 524 | 100 | 100 | |||||
| Established Medicines | 7 268 | 7 481 | – 3 | – 3 | |||||
| Total Innovative Medicines | 34 892 | 32 278 | 8 | 8 | |||||
| US | Rest of world | Total | |||||||||||||||||||
Brands | Business franchise | Indication | USD m | % change USD/cc 2 | USD m | % change USD | % change cc 2 | USD m | % change USD | % change cc 2 | |||||||||||
| Gilenya | Neuroscience | Relapsing multiple sclerosis | 1 765 | 3 | 1 576 | 7 | 5 | 3 341 | 5 | 4 | |||||||||||
| Cosentyx | Immunology, Hepatology and Dermatology | Psoriasis, ankylosing spondylitis and psoriatic arthritis | 1 674 | 31 | 1 163 | 46 | 44 | 2 837 | 37 | 36 | |||||||||||
| Lucentis | Ophthalmology | Age-related macular degeneration | 2 046 | 8 | 7 | 2 046 | 8 | 7 | |||||||||||||
| Tasigna | Oncology | Chronic myeloid leukemia | 806 | 0 | 1 068 | 4 | 3 | 1 874 | 2 | 1 | |||||||||||
| Sandostatin | Oncology | Carcinoid tumors and acromegaly | 817 | – 2 | 770 | – 1 | – 1 | 1 587 | – 2 | – 2 | |||||||||||
| Gleevec/Glivec | Oncology | Chronic myeloid leukemia and GIST | 440 | – 30 | 1 121 | – 15 | – 16 | 1 561 | – 20 | – 20 | |||||||||||
| Afinitor/Votubia | Oncology | Breast cancer/TSC | 929 | 13 | 627 | – 11 | – 12 | 1 556 | 2 | 2 | |||||||||||
Galvus Group | Established Medicines | Diabetes | 1 284 | 4 | 6 | 1 284 | 4 | 6 | |||||||||||||
| Promacta/Revolade | Oncology | Immune thrombocytopenia (ITP), severe aplastic anemia (SAA) | 581 | 30 | 593 | 41 | 40 | 1 174 | 35 | 35 | |||||||||||
Tafinlar +Mekinist | Oncology | BRAF V600+ metastatic and adjuvant melanoma; advanced non-small cell lung cancer (NSCLC) | 457 | 35 | 698 | 31 | 29 | 1 155 | 32 | 31 | |||||||||||
| Exjade/Jadenu | Oncology | Chronic iron overload | 521 | 1 | 578 | 6 | 5 | 1 099 | 4 | 3 | |||||||||||
Xolair 1 | Respiratory | Severe Allergic Asthma (SAA) and Chronic Spontaneous Urticaria (CSU) | 1 039 | 13 | 12 | 1 039 | 13 | 12 | |||||||||||||
| Entresto | Cardiovascular, Renal and Metabolism | Chronic heart failure | 556 | 87 | 472 | 125 | 124 | 1 028 | 103 | 102 | |||||||||||
Diovan Group | Established Medicines | Hypertension | 84 | – 3 | 939 | 8 | 8 | 1 023 | 7 | 7 | |||||||||||
Exforge Group | Established Medicines | Hypertension | 19 | – 32 | 983 | 5 | 5 | 1 002 | 4 | 4 | |||||||||||
| Jakavi | Oncology | Myelofibrosis (MF), polycytomia vera (PV) | 977 | 26 | 24 | 977 | 26 | 24 | |||||||||||||
| Votrient | Oncology | Renal cell carcinoma | 404 | – 1 | 424 | 6 | 5 | 828 | 2 | 2 | |||||||||||
| Ilaris | Immunology, Hepatology and Dermatology | Auto-inflammatory (CAPS, TRAPS, HIDS/MKD, FMF, SJIA, AOSD and gout) | 262 | 34 | 292 | 42 | 44 | 554 | 38 | 39 | |||||||||||
| Travoprost Group | Ophthalmology | Reduction of elevated intraocular pressure | 194 | – 10 | 323 | – 13 | – 13 | 517 | – 12 | – 12 | |||||||||||
| Zortress/Certican | Established Medicines | Transplantation | 145 | 12 | 319 | 12 | 12 | 464 | 12 | 12 | |||||||||||
| Top 20 products total | 9 654 | 11 | 17 292 | 10 | 9 | 26 946 | 10 | 10 | |||||||||||||
| Rest of portfolio | 2 210 | 4 | 5 736 | 0 | 0 | 7 946 | 1 | 1 | |||||||||||||
| Total division sales | 11 864 | 9 | 23 028 | 8 | 7 | 34 892 | 8 | 8 | |||||||||||||
1 Net sales reflectXolair sales for all indications. | |||||||||||||||||||||
2 Constant currencies (cc) is a non-IFRS measure. For an explanation of non-IFRS measures, see " —Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||||||||||||||
(USD millions) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % | |||||
Retail Generics1 | 7 880 | 8 409 | – 6 | – 7 | |||||
| Biopharmaceuticals | 1 436 | 1 135 | 27 | 24 | |||||
| Anti-Infectives (partner label/API) | 543 | 516 | 5 | 3 | |||||
| Total Sandoz | 9 859 | 10 060 | – 2 | – 3 | |||||
1 Of which USD 826 million (2017: USD 880 million) represents anti-infectives sold under the Sandoz name | |||||||||
(USD millions) | Year ended Dec 31, 2018 | % of net sales | Year ended Dec 31, 2017 | % of net sales | Change in USD % | Change in constant currencies % | |||||||
| Innovative Medicines | 7 871 | 22.6 | 7 595 | 23.5 | 4 | 4 | |||||||
| Sandoz | 1 332 | 13.5 | 1 368 | 13.6 | – 3 | – 2 | |||||||
| Corporate | – 800 | – 261 | nm | nm | |||||||||
| Operating income from continuing operations | 8 403 | 18.8 | 8 702 | 20.6 | – 3 | – 3 | |||||||
| nm = not meaningful | |||||||||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % | |||||
| Core gross profit from continuing operations | 34 886 | 32 374 | 8 | 7 | |||||
| Selling, general and administration | – 13 690 | – 12 468 | – 10 | – 9 | |||||
| Research and development | – 8 154 | – 7 808 | – 4 | – 4 | |||||
| Other income | 558 | 748 | – 25 | – 26 | |||||
| Other expense | – 1 043 | – 1 132 | 8 | 9 | |||||
| Core operating income from continuing operations | 12 557 | 11 714 | 7 | 7 | |||||
| As % of net sales | 28.1 | 27.7 | |||||||
1 For an explanation of non-IFRS measures and reconciliation tables, see "Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
(USD millions) | Year ended Dec 31, 2018 | % of net sales | Year ended Dec 31, 2017 | % of net sales | Change in USD % | Change in constant currencies % | |||||||
| Innovative Medicines | 11 151 | 32.0 | 10 019 | 31.0 | 11 | 11 | |||||||
| Sandoz | 2 002 | 20.3 | 2 080 | 20.7 | – 4 | – 3 | |||||||
| Corporate | – 596 | – 385 | – 55 | – 52 | |||||||||
| Core operating income from continuing operations | 12 557 | 28.1 | 11 714 | 27.7 | 7 | 7 | |||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % | |||||
| Research and exploratory development | – 2 770 | – 2 729 | – 2 | – 1 | |||||
| Confirmatory development | – 4 905 | – 4 886 | 0 | 0 | |||||
| Total Innovative Medicines Division research and development expense | – 7 675 | – 7 615 | – 1 | 0 | |||||
| As % of Innovative Medicines net sales to third parties | 22.0 | 23.6 | |||||||
Core research and exploratory development1 | – 2 665 | – 2 603 | – 2 | – 2 | |||||
Core confirmatory development1 | – 4 675 | – 4 431 | – 6 | – 5 | |||||
| Total core Innovative Medicines Division research and development expense | – 7 340 | – 7 034 | – 4 | – 4 | |||||
| As % of Innovative Medicines net sales to third parties | 21.0 | 21.8 | |||||||
1 Core excludes impairments, amortization and certain other items. For an explanation of non-IFRS measures and reconciliation tables, see " —Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % | |||||
| Operating income from continuing operations | 8 403 | 8 702 | – 3 | – 3 | |||||
| Income from associated companies | 6 438 | 1 108 | nm | nm | |||||
| Interest expense | – 932 | – 750 | – 24 | – 27 | |||||
| Other financial income and expense | 186 | 42 | nm | nm | |||||
| Income before taxes | 14 095 | 9 102 | 55 | 55 | |||||
| Taxes | – 1 295 | – 1 603 | 19 | 19 | |||||
| Net income from continuing operations | 12 800 | 7 499 | 71 | 71 | |||||
| Net (loss)/income from discontinued operations | – 186 | 204 | nm | nm | |||||
| Net income | 12 614 | 7 703 | 64 | 64 | |||||
| Attributable to: | |||||||||
| Shareholders of Novartis AG | 12 611 | 7 703 | 64 | 64 | |||||
| Non-controlling interests | 3 | 0 | nm | nm | |||||
| Basic earnings per share from continuing operations (USD) | 5.52 | 3.20 | 73 | 73 | |||||
| Basic earnings per share from discontinued operations (USD) | – 0.08 | 0.08 | nm | nm | |||||
| Total basic earnings per share (USD) | 5.44 | 3.28 | 66 | 66 | |||||
| nm = not meaningful | |||||||||
(USD millions unless indicated otherwise) | Year ended Dec 31, 2018 | Year ended Dec 31, 2017 | Change in USD % | Change in constant currencies % | |||||
| Core operating income from continuing operations | 12 557 | 11 714 | 7 | 7 | |||||
| Core income from associated companies | 1 113 | 1 335 | – 17 | – 17 | |||||
| Core interest expense | – 932 | – 750 | – 24 | – 27 | |||||
| Core other financial income and expense | 186 | 42 | nm | nm | |||||
| Core income before taxes from continuing operations | 12 924 | 12 341 | 5 | 5 | |||||
| Core taxes | – 2 004 | – 1 867 | – 7 | – 7 | |||||
| Core net income from continuing operations | 10 920 | 10 474 | 4 | 4 | |||||
| Core basic EPS from continuing operations (USD) | 4.71 | 4.46 | 6 | 5 | |||||
1 For an explanation of non-IFRS measures and reconciliation tables, see "Item 5.A Operating results—Non-IFRS measures as defined by Novartis." | |||||||||
| nm = not meaningful | |||||||||
(USD millions) | 2019 | In % of gross sales | 2018 | In % of gross sales | 2017 | In % of gross sales | |||||||
| Innovative Medicines gross sales subject to deductions | 52 956 | 100.0 | 47 785 | 100.0 | 43 127 | 100.0 | |||||||
| US-specific healthcare plans and program rebates | – 4 824 | – 9.1 | – 3 921 | – 8.2 | – 3 303 | – 7.7 | |||||||
| Non-US-specific healthcare plans and program rebates | – 3 438 | – 6.5 | – 3 140 | – 6.6 | – 2 652 | – 6.1 | |||||||
| Non-healthcare plans and program-related rebates, returns and other deductions | – 6 980 | – 13.2 | – 5 832 | – 12.2 | – 4 894 | – 11.4 | |||||||
| Total Innovative Medicines gross-to-net sales adjustments | – 15 242 | – 28.8 | – 12 893 | – 27.0 | – 10 849 | – 25.2 | |||||||
| Innovative Medicines net sales | 37 714 | 71.2 | 34 892 | 73.0 | 32 278 | 74.8 | |||||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Operating income from continuing operations | 9 086 | 8 403 | 8 702 | ||||
| Depreciation of property, plant and equipment | 1 345 | 1 482 | 1 303 | ||||
Depreciation of the right-of-use-assets 1 | 305 | ||||||
| Amortization of intangible assets | 2 836 | 2 587 | 2 624 | ||||
| Impairments of property, plant and equipment, and intangible assets | 1 340 | 1 142 | 809 | ||||
EBITDA from continuing operations 1 | 14 912 | 13 614 | 13 438 | ||||
| Operating income from discontinued operations | 71 | – 234 | – 73 | ||||
| Depreciation of property, plant and equipment | 42 | 235 | 217 | ||||
| Depreciation of the right-of-use-assets | 9 | ||||||
| Amortization of intangible assets | 174 | 1 052 | 1 066 | ||||
| Impairments of property, plant and equipment, and intangible assets | 394 | 57 | |||||
| EBITDA from discontinued operations | 296 | 1 447 | 1 267 | ||||
EBITDA Total Group 1 | 15 208 | 15 061 | 14 705 | ||||
1 In 2019, EBITDA is positively impacted through the adoption of IFRS 16 Leases on January 1, 2019, as lease expenses are classified as depreciation on right-of-use assets (USD 314 million, thereof USD 305 million continuing operations) and interest expense (USD 66 million). In the prior years, the lease expense was recognized as a functional expense within operating income (lease expense for total group was in 2018 USD 383 million, thereof USD 331 million from continuing operations and in 2017 USD 337 million thereof USD 292 million from continuing operations). | |||||||
| (USD millions unless indicated otherwise) | Dec 31, 2019 1 | Dec 31, 2018 | Dec 31, 2017 | ||||
| Market capitalization | 214 815 | 196 950 | 195 541 | ||||
| Non-controlling interests | 77 | 78 | 59 | ||||
| Non current financial debts | 20 353 | 22 470 | 23 224 | ||||
| Current financial debts and derivatives financial instruments | 7 031 | 9 678 | 5 308 | ||||
| Marketable securities, commodities, time deposits and derivative financial instruments | – 334 | – 2 693 | – 625 | ||||
| Cash and cash equivalents | – 11 112 | – 13 271 | – 8 860 | ||||
| Enterprise value | 230 830 | 213 212 | 214 647 | ||||
1 Decrmber 31, 2019 excludes the business of Alcon, which was spun off in April 2019 into a separately traded standalone company. For details see "Item 18. Financial Statements—Note 2. Significant transactions". | |||||||
| Innovative Medicines | Sandoz | Corporate | Group | ||||||||||||||
| (USD millions unless indicated otherwise) | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | |||||||||
| IFRS operating income from continuing operations | 9 287 | 7 871 | 551 | 1 332 | – 752 | – 800 | 9 086 | 8 403 | |||||||||
| Amortization of intangible assets | 2 447 | 2 158 | 314 | 363 | 2 761 | 2 521 | |||||||||||
| Impairments | |||||||||||||||||
| Intangible assets | 632 | 592 | 503 | 249 | 1 135 | 841 | |||||||||||
| Property, plant and equipment related to the Group-wide rationalization of manufacturing sites | 83 | 170 | 69 | 63 | 152 | 233 | |||||||||||
| Other property, plant and equipment | 10 | 65 | 33 | 43 | 65 | ||||||||||||
| Total impairment charges | 725 | 827 | 605 | 312 | 1 330 | 1 139 | |||||||||||
| Acquisition or divestment of businesses and related items | |||||||||||||||||
| - Income | – 8 | – 108 | – 21 | – 116 | – 21 | ||||||||||||
| - Expense | 87 | 126 | 115 | 29 | 202 | 155 | |||||||||||
| Total acquisition or divestment of businesses and related items, net | 79 | 126 | 7 | 8 | 86 | 134 | |||||||||||
| Other items | |||||||||||||||||
| Divestment gains | – 1 091 | – 482 | – 78 | 2 | – 56 | – 1 089 | – 616 | ||||||||||
| Financial assets - fair value adjustments | – 18 | – 107 | – 20 | 113 | – 38 | 6 | |||||||||||
| Restructuring and related items | |||||||||||||||||
| - Income | – 58 | – 25 | – 7 | – 12 | – 6 | – 2 | – 71 | – 39 | |||||||||
| - Expense | 509 | 665 | 390 | 179 | 113 | 106 | 1 012 | 950 | |||||||||
| Legal-related items | |||||||||||||||||
| - Income | – 1 | – 32 | – 63 | – 32 | – 64 | ||||||||||||
| - Expense | 999 | 36 | 156 | 90 | 1 155 | 126 | |||||||||||
| Additional income | – 316 | – 73 | – 4 | – 171 | – 95 | – 19 | – 415 | – 263 | |||||||||
| Additional expense | 87 | 156 | 121 | 50 | 119 | 54 | 327 | 260 | |||||||||
| Total other items | 112 | 169 | 624 | – 5 | 113 | 196 | 849 | 360 | |||||||||
| Total adjustments | 3 363 | 3 280 | 1 543 | 670 | 120 | 204 | 5 026 | 4 154 | |||||||||
| Core operating income from continuing operations | 12 650 | 11 151 | 2 094 | 2 002 | – 632 | – 596 | 14 112 | 12 557 | |||||||||
| as % of net sales | 33.5% | 32.0% | 21.5% | 20.3% | 29.7% | 28.1% | |||||||||||
| Income from associated companies | 1 | 1 | 2 | 5 | 656 | 6 432 | 659 | 6 438 | |||||||||
| Core adjustments to income from associated companies, net of tax | 427 | – 5 325 | 427 | – 5 325 | |||||||||||||
| Interest expense | – 850 | – 932 | |||||||||||||||
| Other financial income and expense | 45 | 186 | |||||||||||||||
| Core adjustments to other financial income and expense | 11 | ||||||||||||||||
| Taxes, adjusted for above items (core taxes) | – 2 300 | – 2 004 | |||||||||||||||
| Core net income from continuing operations | 12 104 | 10 920 | |||||||||||||||
Core net income from discontinued operations 1 | 278 | 1 018 | |||||||||||||||
| Core net income | 12 382 | 11 938 | |||||||||||||||
| Core net income attributable to shareholders of Novartis AG | 12 377 | 11 935 | |||||||||||||||
Core basic EPS from continuing operations (USD) 2 | 5.28 | 4.71 | |||||||||||||||
Core basic EPS from discontinued operations (USD) 2 | 0.12 | 0.44 | |||||||||||||||
Core basic EPS (USD) 2 | 5.40 | 5.15 | |||||||||||||||
1 For details on discontinued operations reconciliation from IFRS to core net income, please refer to page 109. | |||||||||||||||||
2 Earnings per share (EPS) is calculated on the amount of net income attributable to shareholders of Novartis AG. | |||||||||||||||||
| Innovative Medicines | Sandoz | Corporate | Group | ||||||||||||||
| (USD millions unless indicated otherwise) | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | |||||||||
| IFRS operating income from continuing operations | 7 871 | 7 595 | 1 332 | 1 368 | – 800 | – 261 | 8 403 | 8 702 | |||||||||
| Amortization of intangible assets | 2 158 | 2 119 | 363 | 454 | 2 521 | 2 573 | |||||||||||
| Impairments | |||||||||||||||||
| Intangible assets | 592 | 591 | 249 | 61 | 841 | 652 | |||||||||||
| Property, plant and equipment related to the Group-wide rationalization of manufacturing sites | 170 | 7 | 63 | 60 | 233 | 67 | |||||||||||
| Other property, plant and equipment | 65 | 77 | 13 | 65 | 90 | ||||||||||||
Financial assets 1 | 197 | 197 | |||||||||||||||
| Total impairment charges | 827 | 675 | 312 | 134 | 197 | 1 139 | 1 006 | ||||||||||
| Acquisition or divestment of businesses and related items | |||||||||||||||||
| - Income | – 2 | – 21 | – 115 | – 21 | – 117 | ||||||||||||
| - Expense | 126 | 32 | 29 | 130 | 155 | 162 | |||||||||||
| Total acquisition or divestment of businesses and related items, net | 126 | 30 | 8 | 15 | 134 | 45 | |||||||||||
| Other items | |||||||||||||||||
| Divestment gains | – 482 | – 368 | – 78 | – 56 | – 616 | – 368 | |||||||||||
Financial assets - fair value adjustments 1 | – 107 | 113 | 6 | ||||||||||||||
| Restructuring and related items | |||||||||||||||||
| - Income | – 25 | – 53 | – 12 | – 7 | – 2 | – 1 | – 39 | – 61 | |||||||||
| - Expense | 665 | 268 | 179 | 134 | 106 | – 9 | 950 | 393 | |||||||||
| Legal-related items | |||||||||||||||||
| - Income | – 1 | – 21 | – 63 | – 64 | – 21 | ||||||||||||
| - Expense | 36 | 35 | 90 | 126 | 35 | ||||||||||||
| Additional income | – 73 | – 534 | – 171 | – 3 | – 19 | – 372 | – 263 | – 909 | |||||||||
| Additional expense | 156 | 273 | 50 | 54 | 46 | 260 | 319 | ||||||||||
| Total other items | 169 | – 400 | – 5 | 124 | 196 | – 336 | 360 | – 612 | |||||||||
| Total adjustments | 3 280 | 2 424 | 670 | 712 | 204 | – 124 | 4 154 | 3 012 | |||||||||
| Core operating income from continuing operations | 11 151 | 10 019 | 2 002 | 2 080 | – 596 | – 385 | 12 557 | 11 714 | |||||||||
| as % of net sales | 32.0% | 31.0% | 20.3% | 20.7% | 28.1% | 27.7% | |||||||||||
| Income from associated companies | 1 | – 1 | 5 | 23 | 6 432 | 1 086 | 6 438 | 1 108 | |||||||||
| Core adjustments to income from associated companies, net of tax | 1 | – 5 325 | 226 | – 5 325 | 227 | ||||||||||||
| Interest expense | – 932 | – 750 | |||||||||||||||
| Other financial income and expense | 186 | 42 | |||||||||||||||
| Taxes, adjusted for above items (core taxes) | – 2 004 | – 1 867 | |||||||||||||||
| Core net income from continuing operations | 10 920 | 10 474 | |||||||||||||||
Core net income from discontinued operations 2 | 1 018 | 917 | |||||||||||||||
| Core net income | 11 938 | 11 391 | |||||||||||||||
| Core net income attributable to shareholders of Novartis AG | 11 935 | 11 391 | |||||||||||||||
Core basic EPS from continuing operations (USD) 3 | 4.71 | 4.46 | |||||||||||||||
Core basic EPS from discontinued operations (USD) 3 | 0.44 | 0.40 | |||||||||||||||
Core basic EPS (USD) 3 | 5.15 | 4.86 | |||||||||||||||
1 For financial instruments accounted for as fair value through profit and loss, as of January 1, 2018, unrealized gains/losses on financial assets are shown under "Financial assets - fair value adjustments," due to the change in IFRS 9. | |||||||||||||||||
2 For details on discontinued operations reconciliation from IFRS to core net income, please refer to page 110. | |||||||||||||||||
3 Earnings per share (EPS) is calculated on the amount of net income attributable to shareholders of Novartis AG. | |||||||||||||||||
2019 (USD millions unless indicated otherwise) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items 3 | Other items 4 | Core results | |||||||
| Gross profit from continuing operations | 34 252 | 2 711 | 85 | 48 | 296 | 37 392 | |||||||
| Operating income from continuing operations | 9 086 | 2 761 | 1 330 | 86 | 849 | 14 112 | |||||||
| Income before taxes from continuing operations | 8 940 | 3 188 | 1 330 | 86 | 860 | 14 404 | |||||||
Taxes from continuing operations 5 | – 1 793 | – 2 300 | |||||||||||
| Net income from continuing operations | 7 147 | 12 104 | |||||||||||
Net income from discontinued operations 6 | 4 590 | 278 | |||||||||||
| Net income | 11 737 | 12 382 | |||||||||||
Basic EPS from continuing operations (USD) 7 | 3.12 | 5.28 | |||||||||||
Basic EPS from discontined operations (USD) 7 | 2.00 | 0.12 | |||||||||||
Basic EPS (USD) 7 | 5.12 | 5.40 | |||||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Other revenues | 1 179 | – 66 | 1 113 | ||||||||||
| Cost of goods sold | – 14 425 | 2 711 | 85 | 48 | 362 | – 11 219 | |||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 14 369 | 10 | 40 | – 14 319 | |||||||||
| Research and development | – 9 402 | 50 | 1 078 | 10 | – 122 | – 8 386 | |||||||
| Other income | 2 031 | – 2 | – 116 | – 1 418 | 495 | ||||||||
| Other expense | – 3 426 | 169 | 134 | 2 053 | – 1 070 | ||||||||
| The following are adjustments to arrive at core income before taxes | |||||||||||||
| Income from associated companies | 659 | 427 | 1 086 | ||||||||||
| Other financial income and expense | 45 | 11 | 56 | ||||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the amortization of acquired rights for technologies; income from associated companies includes USD 427 million for the Novartis share of the estimated Roche core items | |||||||||||||
2 Impairments: cost of goods sold, and research and development include impairment charges related to intangible assets; research and development also includes the reversal of an impairment charge; cost of goods sold, other income and other expense include net impairment charges related to property, plant and equipment | |||||||||||||
3 Acquisition or divestment of businesses and related items, including restructuring and integration charges: cost of goods sold, selling, general and administration, research and development, other income and other expense include net charges related to acquisitions; other income and other expense also include transitional service fee income and expenses related to the portfolio transformation and the Alcon spin-off | |||||||||||||
4 Other items: other revenues includes income from an outlicensing agreement, and income related to an amendment of a collaboration agreement; cost of goods sold, other income and other expense include net restructuring and other charges related to the Group-wide rationalization of manufacturing sites; cost of goods sold, research and development, selling, general and administration, other income and other expense include other restructuring income and charges and related items; cost of goods sold, and research and development also include fair value adjustments of contingent consideration liabilties; cost of goods sold also includes inventory write-offs and other provisions; selling, general and administration includes receivable expected credit loss provisions and other provisions; other income and other expense include fair value adjustments and divestment gains and losses on financial assets and legal-related items as well as environmental provisions; other income also includes net gains from the divestment of products and property, plant and equipment, and provision releases; other expense includes a provision for onerous contracts and other provisions; other financial income and expense includes a revaluation impact of a financial liability incurred through the Alcon distribution | |||||||||||||
5 Taxes on the adjustments between IFRS and core results take into account, for each individual item included in the adjustment, the tax rate that will finally be applicable to the item based on the jurisdiction where the adjustment will finally have a tax impact. Generally, this results in amortization and impairment of intangible assets and acquisition-related restructuring and integration items having a full tax impact. There is usually a tax impact on other items, although this is not always the case for items arising from legal settlements in certain jurisdictions. Adjustments related to income from associated companies are recorded net of any related tax effect. Due to these factors and the differing effective tax rates in the various jurisdictions, the tax on the total adjustments for continuing operations of USD 5.5 billion to arrive at the core results before tax amounts to USD 507 million. The average tax rate on the adjustments is 9.3%. | |||||||||||||
6 For details on discontinued operations reconciliation from IFRS to core net income please refer to page 109. | |||||||||||||
7 Earnings per share (EPS) is calculated on the amount of net income, attributable to shareholders of Novartis AG. | |||||||||||||
2018 (USD millions unless indicated otherwise) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items 3 | Other items 4 | Core results | |||||||
| Gross profit from continuing operations | 31 589 | 2 342 | 488 | 5 | 462 | 34 886 | |||||||
| Operating income from continuing operations | 8 403 | 2 521 | 1 139 | 134 | 360 | 12 557 | |||||||
| Income before taxes from continuing operations | 14 095 | 2 965 | 1 139 | – 5 656 | 381 | 12 924 | |||||||
Taxes from continuing operations 5 | – 1 295 | – 2 004 | |||||||||||
| Net income from continuing operations | 12 800 | 10 920 | |||||||||||
Net income from discontinued operations 6 | – 186 | 1 018 | |||||||||||
| Net income | 12 614 | 11 938 | |||||||||||
Basic EPS from continuing operations (USD) 7 | 5.52 | 4.71 | |||||||||||
Basic EPS from discontined operations (USD) 7 | – 0.08 | 0.44 | |||||||||||
Basic EPS (USD) 7 | 5.44 | 5.15 | |||||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 14 510 | 2 342 | 488 | 5 | 462 | – 11 213 | |||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 13 717 | 28 | – 1 | – 13 690 | |||||||||
| Research and development | – 8 489 | 179 | 167 | 23 | – 34 | – 8 154 | |||||||
| Other income | 1 629 | – 21 | – 1 050 | 558 | |||||||||
| Other expense | – 2 609 | 484 | 99 | 983 | – 1 043 | ||||||||
| The following are adjustments to arrive at core income before taxes | |||||||||||||
| Income from associated companies | 6 438 | 444 | – 5 790 | 21 | 1 113 | ||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products, and other production-related intangible assets; research and development includes the amortization of acquired rights, including technology platforms; income from associated companies includes USD 444 million for the Novartis share of the estimated Roche core items | |||||||||||||
2 Impairments: cost of goods sold, and research and development include impairment charges related to intangible assets; research and development also includes impairment reversals of property, plant and equipment; other expense includes impairment charges related to property, plant and equipment; cost of goods sold and other expense include impairment charges related to a disposal group held for sale for goodwill and currently marketed products | |||||||||||||
3 Acquisition or divestment of businesses and related items, including restructuring and integration charges: cost of goods sold, selling, general and administration, research and development and other expense include charges related to acquisitions; other income and other expense include transitional service fee income and expenses, and other items related to the portfolio transformation; income from associated companies includes the pre-tax gain of USD 5.8 billion on the sale of the 36.5% investment in GSK Consumer Healthcare Holdings Ltd. | |||||||||||||
4 Other items: cost of goods sold, other income and other expense include net restructuring and other charges related to the Group-wide rationalization of manufacturing sites; cost of goods sold, selling, general and administration, research and development, other income and other expense include other restructuring income and charges and related items; cost of goods sold and other expense include charges related to changes in a contractual agreement; cost of goods sold also includes inventory write-off and other product recall-related costs; selling, general and administration includes a reversal of a provision; research and development includes fair value adjustments of contingent consideration liabilities and a charge for onerous contracts; other income and other expense include fair value adjustments and divestment gains and losses on financial assets; other income also includes product divestment gains, divestment gains on property, plant and equipment, releases of accruals and a legal settlement gain; other expense includes legal-related items and restructuring charges; income from associated companies includes an adjustment of USD 21 million for the Novartis share of the estimated GSK Consumer Healthcare Holdings Ltd. core items | |||||||||||||
5 Taxes on the adjustments between IFRS and core results take into account, for each individual item included in the adjustment, the tax rate that will finally be applicable to the item based on the jurisdiction where the adjustment will finally have a tax impact. Generally, this results in amortization and impairment of intangible assets and acquisition-related restructuring and integration items having a full tax impact. There is usually a tax impact on other items, although this is not always the case for items arising from legal settlements in certain jurisdictions. Adjustments related to income from associated companies are recorded net of any related tax effect. Due to these factors and the differing effective tax rates in the various jurisdictions, the tax on the total adjustments of USD 1.2 billion to arrive at the core results before tax amounts to USD 709 million. Excluding the gain on the sale of the 36.5% investment in GSK Cosumer Healthcare Holdings Ltd., the tax on the total adjustments of USD 4.6 billion to arrive at the core results before tax amounts to USD 770 billion. The average tax rate on the adjustments excluding this transaction is 16.7%. | |||||||||||||
6 For details on discontinued operations reconciliation from IFRS to core net income please refer to page 110. | |||||||||||||
7 Earnings per share (EPS) is calculated on the amount of net income, attributable to shareholders of Novartis AG. | |||||||||||||
2017 (USD millions unless indicated otherwise) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items 3 | Other items 4 | Core results | |||||||
| Gross profit | 29 771 | 2 386 | 92 | 125 | 32 374 | ||||||||
| Operating income | 8 702 | 2 573 | 1 006 | 45 | – 612 | 11 714 | |||||||
| Income before taxes | 9 102 | 2 949 | 1 007 | 45 | – 762 | 12 341 | |||||||
Taxes 5 | – 1 603 | – 1 867 | |||||||||||
| Net income from continuing operations | 7 499 | 10 474 | |||||||||||
Net income from discontinued operations 6 | 204 | 917 | |||||||||||
| Net income | 7 703 | 11 391 | |||||||||||
Basic EPS from continuing operations (USD) 7 | 3.20 | 4.46 | |||||||||||
Basic EPS from discontined operations (USD) 7 | 0.08 | 0.40 | |||||||||||
Basic EPS (USD) 7 | 3.28 | 4.86 | |||||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 13 633 | 2 386 | 92 | 125 | – 11 030 | ||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 12 465 | – 3 | – 12 468 | ||||||||||
| Research and development | – 8 389 | 187 | 594 | – 200 | – 7 808 | ||||||||
| Other income | 1 922 | – 9 | – 117 | – 1 048 | 748 | ||||||||
| Other expense | – 2 137 | 329 | 162 | 514 | – 1 132 | ||||||||
| The following are adjustments to arrive at core income before taxes | |||||||||||||
| Income from associated companies | 1 108 | 376 | 1 | – 150 | 1 335 | ||||||||
1 Amortization of intangible assets: cost of goods sold includes the recurring amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the recurring amortization of acquired rights for technology platforms; income from associated companies includes USD 376 million for the Novartis share of the estimated Roche core items | |||||||||||||
2 Impairments: cost of goods sold, and research and development include impairment charges related to intangible assets; research and development, other income and other expense include reversals and charges related to the impairment of property, plant and equipment; other expense also includes impairment charges related to financial assets | |||||||||||||
3 Acquisition or divestment of businesses and related items, including restructuring and integration charges: other income and other expense include transitional service fee income and expenses, and other items related to the portfolio transformation | |||||||||||||
4 Other items: cost of goods sold, other income and other expense include net restructuring and other charges related to the Group-wide rationalization of manufacturing sites; cost of goods sold, research and development, selling, general and administration, other income and other expense include other restructuring income and charges and related items; selling, general and administration includes an income from the release of a provision; research and development includes fair value adjustments to contingent consideration liabilities; other income and other expense include legal-related items; other income also includes a gain from a Swiss pension plan amendment, product and financial asset divestment gains, income from a settlement of a contract dispute and a fair value adjustment to contingent consideration sales milestone receivables; other expense also includes a provision for contract termination costs, a charge for onerous contracts, and an amendment to the Swiss pension plan; income from associated companies includes an adjustment of USD 150 million for the Novartis share of the estimated GSK Consumer Healthcare Holdings Ltd. core items | |||||||||||||
5 Taxes on the adjustments between IFRS and core results take into account, for each individual item included in the adjustment, the tax rate that will finally be applicable to the item based on the jurisdiction where the adjustment will finally have a tax impact. Generally, this results in amortization and impairment of intangible assets and acquisition-related restructuring and integration items having a full tax impact. There is usually a tax impact on other items, although this is not always the case for items arising from legal settlements in certain jurisdictions. Adjustments related to income from associated companies are recorded net of any related tax effect. Due to these factors and the differing effective tax rates in the various jurisdictions, the tax on the total adjustments of USD 3.2 billion to arrive at the core results before tax amounts to USD 264 million. The average tax rate on the adjustments is 8.2%. | |||||||||||||
6 For details on discontinued operations reconciliation from IFRS to core net income please refer to page 110. | |||||||||||||
7 Earnings per share (EPS) is calculated on the amount of net income, attributable to shareholders of Novartis AG. | |||||||||||||
2019 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items 3 | Other items 4 | Core results | |||||||
| Gross profit | 29 539 | 2 397 | 48 | 116 | 32 100 | ||||||||
| Operating income | 9 287 | 2 447 | 725 | 79 | 112 | 12 650 | |||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Other revenues | 1 092 | – 66 | 1 026 | ||||||||||
| Cost of goods sold | – 10 050 | 2 397 | 48 | 182 | – 7 423 | ||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 11 617 | 10 | 25 | – 11 582 | |||||||||
| Research and development | – 8 152 | 50 | 632 | 10 | – 125 | – 7 585 | |||||||
| Other income | 1 586 | – 1 | – 8 | – 1 230 | 347 | ||||||||
| Other expense | – 2 069 | 94 | 19 | 1 326 | – 630 | ||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the amortization of acquired rights for technologies | |||||||||||||
2 Impairments: research and development includes impairment charges and a reversal of impairment charges related to intangible assets; other income and other expense include net impairment charges related to property, plant and equipment | |||||||||||||
3 Acquisition or divestment of businesses and related items, including restructuring and integration charges: cost of goods sold, selling, general and administration, research and development, other income and other expense include net charges related to acquisitions; other income and other expense also include transitional service-fee income and expenses related to the portfolio transformation and the Alcon spin-off | |||||||||||||
4 Other items: other revenues includes a net income from an outlicensing agreement and an income related to an amendment of a collaboration agreement; cost of goods sold, other income and other expense include restructuring and other charges related to the Group-wide rationalization of manufacturing sites; cost of goods sold, research and development, other income and other expense include other restructuring income and charges and related items; cost of goods sold, and research and development also include fair value adjustments of contingent consideration liabilities; selling, general and administration includes other provisions; other income and other expense include fair value adjustments and divestment gains and losses on financial assets; other income also includes net gains from the divestment of products and property, plant and equipment, and provision releases; other expense includes legal-related items | |||||||||||||
2018 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items 3 | Other items 4 | Core results | |||||||
| Gross profit | 26 951 | 1 979 | 423 | 5 | 329 | 29 687 | |||||||
| Operating income | 7 871 | 2 158 | 827 | 126 | 169 | 11 151 | |||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 9 870 | 1 979 | 423 | 5 | 329 | – 7 134 | |||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 10 907 | 28 | – 11 | – 10 890 | |||||||||
| Research and development | – 7 675 | 179 | 167 | 23 | – 34 | – 7 340 | |||||||
| Other income | 977 | – 671 | 306 | ||||||||||
| Other expense | – 1 475 | 237 | 70 | 556 | – 612 | ||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the amortization of acquired rights, including technology platforms | |||||||||||||
2 Impairments: cost of goods sold, and research and development include impairment charges related to intangible assets; research and development also includes impairment reversals of property, plant and equipment; other expense includes impairment charges related to property, plant and equipment | |||||||||||||
3 Acquisition or divestment of businesses and related items, including restructuring and integration charges: cost of goods sold, selling, general and administration, research and development and other expense include charges related to acquisitions; other expense also includes items related to the portfolio transformation | |||||||||||||
4 Other items: cost of goods sold and other expense include restructuring and other charges related to the Group-wide rationalization of manufacturing sites, and charges related to changes in a contractual agreement; cost of goods sold, research and development, other income and other expense include other restructuring income and charges and related items; cost of goods sold, and research and development also include fair value adjustments of contingent consideration liabilities; cost of goods sold also includes an inventory write-off; selling, general and administration includes a reversal of a provision; research and development includes a charge for onerous contracts; other income and other expense include fair value adjustments on financial assets and legal-related items; other income also includes product divestment gains and releases of accruals | |||||||||||||
2017 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items 3 | Other items 4 | Core results | |||||||
| Gross profit | 25 194 | 1 932 | 31 | 56 | 27 213 | ||||||||
| Operating income | 7 595 | 2 119 | 675 | 30 | – 400 | 10 019 | |||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 8 650 | 1 932 | 31 | 56 | – 6 631 | ||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 9 887 | – 3 | – 9 890 | ||||||||||
| Research and development | – 7 615 | 187 | 594 | – 200 | – 7 034 | ||||||||
| Other income | 1 027 | – 9 | – 2 | – 665 | 351 | ||||||||
| Other expense | – 1 124 | 59 | 32 | 412 | – 621 | ||||||||
1 Amortization of intangible assets: cost of goods sold includes the recurring amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the recurring amortization of acquired rights for technology platforms | |||||||||||||
2 Impairments: cost of goods sold, and research and development include impairment charges related to intangible assets; research and development, other income and other expense include reversals and charges related to the impairment of property, plant and equipment | |||||||||||||
3 Acquisition or divestment of businesses and related items, including restructuring and integration charges: other income includes transitional service fee income; other expense includes items related to the portfolio transformation and costs related to an acquisition | |||||||||||||
4 Other items: cost of goods sold, other income and other expense include net restructuring and other charges related to the Group-wide rationalization of manufacturing sites; costs of goods sold, research and development, selling, general and administration, other income and other expense include other restructuring income and charges and related items; selling, general and administration includes an income from the release of a provision; research and development includes fair value adjustments to contingent consideration liabilities; other income and other expense include legal-related items; other income also includes a gain from a Swiss pension plan amendment, income from a settlement of a contract dispute, as well as product and financial asset divestment gains; other expense also includes a provision for contract termination costs, an amendment to the Swiss pension plan, a charge for onerous contracts, and other charges | |||||||||||||
2019 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items | Other items 3 | Core results | |||||||
| Gross profit | 4 601 | 314 | 85 | 180 | 5 180 | ||||||||
| Operating income | 551 | 314 | 605 | 624 | 2 094 | ||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 5 334 | 314 | 85 | 180 | – 4 755 | ||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 2 218 | 15 | – 2 203 | ||||||||||
| Research and development | – 1 250 | 446 | 3 | – 801 | |||||||||
| Other income | 167 | – 1 | – 39 | 127 | |||||||||
| Other expense | – 749 | 75 | 465 | – 209 | |||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products and other production-related intangible assets | |||||||||||||
2 Impairments: cost of goods sold, and research and development include impairment charges related to intangible assets; cost of goods sold, other income and other expense include net impairment charges related to property, plant and equipment | |||||||||||||
3 Other items: cost of goods sold and other expense include net restructuring and other charges related to the Group-wide rationalization of manufacturing sites; cost of goods sold, selling, general and administration, other income and other expense include restructuring income and charges and related items; cost of goods sold also includes inventory write-offs and other provisions; selling, general and administration includes receivable expected credit loss provisions and other provisions; other income and other expense also include legal-related items; other expense also includes an environmental provision, a provision for onerous contracts and other provisions | |||||||||||||
2018 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items | Other items 3 | Core results | |||||||
| Gross profit | 4 568 | 363 | 65 | 133 | 5 129 | ||||||||
| Operating income | 1 332 | 363 | 312 | – 5 | 2 002 | ||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 5 530 | 363 | 65 | 133 | – 4 969 | ||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 2 305 | 10 | – 2 295 | ||||||||||
| Other income | 505 | – 295 | 210 | ||||||||||
| Other expense | – 622 | 247 | 147 | – 228 | |||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products and other production-related intangible assets | |||||||||||||
2 Impairments: cost of goods sold includes impairment charges related to intangible assets, and impairment charges for currently marketed products related to a disposal group held for sale; other expense includes impairment charges related to property, plant and equipment, and goodwill impairment charges related to a disposal group held for sale | |||||||||||||
3 Other items: cost of goods sold, other income and other expense include net restructuring and other charges related to the Group-wide rationalization of manufacturing sites; cost of goods sold also includes inventory write-off and other product recall-related costs; cost of goods sold, selling, general and administration, other income and other expense include other restructuring income and charges and related items; other income also includes product divestment gains, a legal settlement gain, and fair value adjustments of contingent consideration liabilities; other expense includes legal-related items and restructuring charges | |||||||||||||
2017 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items | Other items 3 | Core results | |||||||
| Gross profit | 4 415 | 454 | 61 | 69 | 4 999 | ||||||||
| Operating income | 1 368 | 454 | 134 | 124 | 2 080 | ||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 5 800 | 454 | 61 | 69 | – 5 216 | ||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Other income | 204 | – 10 | 194 | ||||||||||
| Other expense | – 351 | 73 | 65 | – 213 | |||||||||
1 Amortization of intangible assets: cost of goods sold includes the recurring amortization of acquired rights to in-market products and other production-related intangible assets | |||||||||||||
2 Impairments: cost of goods sold includes impairment charges related to intangible assets; other expense includes impairment charges related to property, plant and equipment | |||||||||||||
3 Other items: cost of goods sold, other income and other expense include net restructuring and other charges related to the Group-wide rationalization of manufacturing sites, and other restructuring income and charges and related items; other income also includes a gain from a Swiss pension plan amendment | |||||||||||||
2019 (USD millions) | IFRS results | Amortization of intangible assets | Impairments | Acquisition or divestment of businesses and related items 1 | Other items 2 | Core results | |||||||
| Gross profit | 112 | 112 | |||||||||||
| Operating loss | – 752 | 7 | 113 | – 632 | |||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Other income | 278 | – 108 | – 149 | 21 | |||||||||
| Other expense | – 608 | 115 | 262 | – 231 | |||||||||
1 Acquisition or divestment of businesses and related items, including restructuring and integration charges: other income and other expense include transitional service fee income and expenses related to the portfolio transformation and the Alcon spin-off | |||||||||||||
2 Other items: other income and other expense include fair value adjustments and divestment gains and losses on financial assets, restructuring income and charges and related items, as well as environmental provisions | |||||||||||||
2018 (USD millions) | IFRS results | Amortization of intangible assets | Impairments | Acquisition or divestment of businesses and related items 1 | Other items 2 | Core results | |||||||
| Gross profit | 70 | 70 | |||||||||||
| Operating loss | – 800 | 8 | 196 | – 596 | |||||||||
| The following are adjustments to arrive at core operating loss | |||||||||||||
| Other income | 147 | – 21 | – 84 | 42 | |||||||||
| Other expense | – 512 | 29 | 280 | – 203 | |||||||||
1 Acquisition or divestment of businesses and related items, including restructuring and integration charges: other income and other expense include transitional service fee income and expenses, and other items related to the portfolio transformation | |||||||||||||
2 Other items: other income and other expense include fair value adjustments and divestment gains and losses on financial assets, as well as restructuring income and charges and related items; other income also includes divestment gains on property, plant and equipment | |||||||||||||
2017 (USD millions) | IFRS results | Amortization of intangible assets | Impairments 1 | Acquisition or divestment of businesses and related items 2 | Other items 3 | Core results | |||||||
| Gross profit | 162 | 162 | |||||||||||
| Operating loss | – 261 | 197 | 15 | – 336 | – 385 | ||||||||
| The following are adjustments to arrive at core operating loss | |||||||||||||
| Other income | 691 | – 115 | – 373 | 203 | |||||||||
| Other expense | – 662 | 197 | 130 | 37 | – 298 | ||||||||
1 Impairments: other expense includes impairment charges related to financial assets | |||||||||||||
2 Acquisition or divestment of businesses and related items, including restructuring and integration charges: other income and other expense include transitional service fee income and expenses, and other items related to the portfolio transformation | |||||||||||||
3 Other items: other income includes a fair value adjustment to contingent consideration sales milestone receivables, a Swiss pension plan amendment and other items; other income and other expense include restructuring income and charges and related items; other expense also includes an amendment to the Swiss pension plan | |||||||||||||
2019 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments | Acquisition or divestment of businesses and related items 2 | Other items 3 | Core results | |||||||
| Gross profit | 949 | 165 | 9 | 1 123 | |||||||||
| Operating income of discontinued operations | 71 | 167 | 112 | 350 | |||||||||
| Income before taxes of discontinued operations | 58 | 337 | |||||||||||
Taxes 4 | – 159 | – 59 | |||||||||||
| Net loss/income from discontinued operations before gain on distribution of Alcon Inc. to Novartis AG shareholders | – 101 | 278 | |||||||||||
| Gain on distribution of Alcon Inc. to Novartis AG shareholders | 4 691 | – 4 691 | |||||||||||
| Net income from discontinued operations | 4 590 | 278 | |||||||||||
Basic EPS (USD) 5 | 2.00 | 0.12 | |||||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 860 | 165 | 9 | – 686 | |||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 638 | 14 | – 624 | ||||||||||
| Research and development | – 142 | 2 | 4 | – 136 | |||||||||
| Other income | 15 | – 3 | 12 | ||||||||||
| Other expense | – 113 | 88 | – 25 | ||||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the amortization of acquired rights for technologies | |||||||||||||
2 Acquisition or divestment of businesses and related items represents the non-taxable, non-cash gain adjustment related to the distribution of Alcon Inc. (spin-off) to Novartis AG shareholders | |||||||||||||
3 Other items: cost of goods sold, selling, general and administration, research and development and other expense include other restructuring charges and related items; research and development also includes amortization of option rights and the fair value adjustment of a contingent consideration liability; other income includes fair value adjustments on a financial asset; other expense also includes legal-related items | |||||||||||||
4 Taxes on the adjustments between IFRS and core results take into account, for each individual item included in the adjustment, the tax rate that will finally be applicable to the item based on the jurisdiction where the adjustment will finally have a tax impact. Generally, this results in amortization and impairment of intangible assets and acquisition-related restructuring and integration items having a full tax impact. There is usually a tax impact on other items, although this is not always the case for items arising from legal settlements in certain jurisdictions. Due to these factors and the differing effective tax rates in the various jurisdictions, the tax on the total adjustments, excluding the non-taxable, non-cash gain on the distribution (spin-off) of Alcon Inc. to Novartis AG shareholders of USD 279 million to arrive at the core results before tax amounts to USD 100 million. The 2019 core tax rate, excluding the effect of the gain on the distribution of Alcon Inc. to Novartis AG shareholders, is 17.5%. | |||||||||||||
5 Earnings per share (EPS) is calculated on the amount of net income attributable to shareholders of Novartis AG. | |||||||||||||
2018 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items | Other items 3 | Core results | |||||||
| Gross profit | 3 170 | 996 | 389 | – 23 | 4 532 | ||||||||
| Operating loss/income of discontinued operations | – 234 | 1 007 | 391 | 102 | 1 266 | ||||||||
| Loss/income before taxes of discontinued operations | – 260 | 1 240 | |||||||||||
Taxes 4 | 74 | – 222 | |||||||||||
| Net loss/income from discontinued operations | – 186 | 1 018 | |||||||||||
Basic EPS (USD) 5 | – 0.08 | 0.44 | |||||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 3 983 | 996 | 389 | – 23 | – 2 621 | ||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Selling, general and administration | – 2 754 | 2 | 13 | – 2 739 | |||||||||
| Research and development | – 585 | 11 | 47 | – 527 | |||||||||
| Other income | 61 | – 23 | 38 | ||||||||||
| Other expense | – 126 | 88 | – 38 | ||||||||||
1 Amortization of intangible assets: cost of goods sold includes the amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the amortization of acquired rights for technology platforms | |||||||||||||
2 Impairments: cost of goods sold and selling, general and administration include impairment charges related to intangible assets | |||||||||||||
3 Other items: cost of goods sold, selling, general and administration and research and development include charges and reversal of charges related to a product's voluntary market withdrawal; cost of goods sold, selling, general and administration, research and development, other income and other expense also include other restructuring income and charges and related items; research and development also includes amortization of option rights and the fair value adjustment of a contingent consideration liability; other income includes fair value adjustments on a financial asset; other expense includes legal-related items | |||||||||||||
4 Taxes on the adjustments between IFRS and core results take into account, for each individual item included in the adjustment, the tax rate that will finally be applicable to the item based on the jurisdiction where the adjustment will finally have a tax impact. Generally, this results in amortization and impairment of intangible assets and acquisition-related restructuring and integration items having a full tax impact. There is usually a tax impact on other items, although this is not always the case for items arising from legal settlements in certain jurisdictions. Due to these factors and the differing effective tax rates in the various jurisdictions, the tax on the total adjustments of USD 1.5 billion to arrive at the core results before tax amounts to USD 296 million. The 2018 core tax rate is 17.9%. | |||||||||||||
5 Earnings per share (EPS) is calculated on the amount of net income attributable to shareholders of Novartis AG. | |||||||||||||
2017 (USD millions) | IFRS results | Amortization of intangible assets 1 | Impairments 2 | Acquisition or divestment of businesses and related items | Other items 3 | Core results | |||||||
| Gross profit | 3 189 | 1 015 | 4 204 | ||||||||||
| Operating loss/income of discontinued operations | – 73 | 1 025 | 86 | 98 | 1 136 | ||||||||
| Loss/income before taxes of discontinued operations | – 103 | 1 106 | |||||||||||
Taxes 4 | 307 | – 189 | |||||||||||
| Net income from discontinued operations | 204 | 917 | |||||||||||
Basic EPS (USD) 5 | 0.08 | 0.40 | |||||||||||
| The following are adjustments to arrive at core gross profit | |||||||||||||
| Cost of goods sold | – 3 588 | 1 015 | – 2 573 | ||||||||||
| The following are adjustments to arrive at core operating income | |||||||||||||
| Research and development | – 583 | 10 | 86 | – 18 | – 505 | ||||||||
| Other income | 47 | – 17 | 30 | ||||||||||
| Other expense | – 194 | 133 | – 61 | ||||||||||
1 Amortization of intangible assets: cost of goods sold includes the recurring amortization of acquired rights to in-market products and other production-related intangible assets; research and development includes the recurring amortization of acquired rights for technology platforms | |||||||||||||
2 Impairments: research and development includes impairment charges related to intangible and financial assets | |||||||||||||
3 Other items: research and development includes fair value adjustments to contingent consideration liabilities; other income and other expense include restructuring income and charges and related items; other income also includes a gain from a Swiss pension plan amendment and the partial reversal of a prior-period charge; other expense also includes legal-related items | |||||||||||||
4 Taxes on the adjustments between IFRS and core results take into account, for each individual item included in the adjustment, the tax rate that will finally be applicable to the item based on the jurisdiction where the adjustment will finally have a tax impact. Generally, this results in amortization and impairment of intangible assets and acquisition-related restructuring and integration items having a full tax impact. There is usually a tax impact on other items, although this is not always the case for items arising from legal settlements in certain jurisdictions. Due to these factors and the differing effective tax rates in the various jurisdictions, the tax on the total adjustments of USD 1.2 billion to arrive at the core results before tax amounts to USD 496 million. The 2017 core tax rate is 17.1%. | |||||||||||||
5 Earnings per share (EPS) is calculated on the amount of net income attributable to shareholders of Novartis AG. | |||||||||||||
5.B Liquidity and capital resources
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Net cash flows from operating activities of continuing operations | 13 547 | 13 049 | 11 419 | ||||
| Net cash flows from operating activities of discontinued operations | 78 | 1 223 | 1 202 | ||||
| Net cash flows used in investing activities of continuing operations | – 1 067 | – 4 590 | – 2 344 | ||||
| Net cash flows used in investing activities from discontinued operations | – 1 159 | – 1 001 | – 775 | ||||
| Net cash flows used in financing activities of continuing operations | – 16 884 | – 4 077 | – 7 318 | ||||
| Net cash flows from/used in financing activities of discontinued operations | 3 257 | – 167 | – 415 | ||||
| Effect of exchange rate changes on cash and cash equivalents | 69 | – 26 | 84 | ||||
| Net change in cash and cash equivalents | – 2 159 | 4 411 | 1 853 | ||||
| Change in marketable securities, commodities, time deposits and derivative financial instruments | – 2 359 | 2 068 | – 145 | ||||
| Change in current and non-current financial debts and derivative financial instruments | 4 764 | – 3 616 | – 4 730 | ||||
| Change in net debt | 246 | 2 863 | – 3 022 | ||||
| Net debt at January 1 | – 16 184 | – 19 047 | – 16 025 | ||||
| Net debt at December 31 | – 15 938 | – 16 184 | – 19 047 | ||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Non-current financial debts | – 20 353 | – 22 470 | – 23 224 | ||||
| Current financial debts and derivative financial instruments | – 7 031 | – 9 678 | – 5 308 | ||||
| Total financial debts | – 27 384 | – 32 148 | – 28 532 | ||||
| Less liquidity | |||||||
| Cash and cash equivalents | 11 112 | 13 271 | 8 860 | ||||
| Marketable securities, commodities, time deposits and derivative financial instruments | 334 | 2 693 | 625 | ||||
| Total liquidity | 11 446 | 15 964 | 9 485 | ||||
| Net debt at December 31 | – 15 938 | – 16 184 | – 19 047 | ||||
| 2019 | |||||||||||||
(USD millions) | Due within one month | Due later than one month but less than three months | Due later than three months but less than one year | Due later than one year but less than five years | Due after five years | Total | |||||||
| Current assets | |||||||||||||
| Marketable securities, time deposits and short-term investments with original maturity more than 90 days | 20 | 26 | 16 | 3 | 57 | 122 | |||||||
| Commodities | 110 | 110 | |||||||||||
| Derivative financial instruments and accrued interest | 14 | 79 | 3 | 3 | 3 | 102 | |||||||
| Cash and cash equivalents | 9 712 | 1 400 | 11 112 | ||||||||||
| Total current financial assets | 9 746 | 1 505 | 19 | 6 | 170 | 11 446 | |||||||
| Non-current liabilities | |||||||||||||
| Financial debt | – 9 110 | – 11 243 | – 20 353 | ||||||||||
| Financial debt - undiscounted | – 9 150 | – 11 355 | – 20 505 | ||||||||||
| Total non-current financial debt | – 9 110 | – 11 243 | – 20 353 | ||||||||||
| Current liabilities | |||||||||||||
| Financial debt | – 4 243 | – 1 373 | – 1 230 | – 6 846 | |||||||||
| Financial debt - undiscounted | – 4 243 | – 1 373 | – 1 230 | – 6 846 | |||||||||
| Derivative financial instruments | – 130 | – 29 | – 26 | – 185 | |||||||||
| Total current financial debt | – 4 373 | – 1 402 | – 1 256 | – 7 031 | |||||||||
| Net debt | 5 373 | 103 | – 1 237 | – 9 104 | – 11 073 | – 15 938 | |||||||
| 2018 | |||||||||||||
(USD millions) | Due within one month | Due later than one month but less than three months | Due later than three months but less than one year | Due later than one year but less than five years | Due after five years | Total | |||||||
| Current assets | |||||||||||||
| Marketable securities, time deposits and short-term investments with original maturity more than 90 days | 39 | 56 | 2 091 | 198 | 63 | 2 447 | |||||||
| Commodities | 104 | 104 | |||||||||||
| Derivative financial instruments and accrued interest | 40 | 75 | 27 | 142 | |||||||||
| Cash and cash equivalents | 3 571 | 9 700 | 13 271 | ||||||||||
| Total current financial assets | 3 650 | 9 831 | 2 118 | 198 | 167 | 15 964 | |||||||
| Non-current liabilities | |||||||||||||
| Financial debt | – 8 980 | – 13 490 | – 22 470 | ||||||||||
| Financial debt - undiscounted | – 9 025 | – 13 623 | – 22 648 | ||||||||||
| Total non-current financial debt | – 8 980 | – 13 490 | – 22 470 | ||||||||||
| Current liabilities | |||||||||||||
| Financial debt | – 5 217 | – 4 084 | – 319 | – 9 620 | |||||||||
| Financial debt - undiscounted | – 5 217 | – 4 084 | – 319 | – 9 620 | |||||||||
| Derivative financial instruments | – 16 | – 34 | – 8 | – 58 | |||||||||
| Total current financial debt | – 5 233 | – 4 118 | – 327 | – 9 678 | |||||||||
| Net debt | – 1 583 | 5 713 | 1 791 | – 8 782 | – 13 323 | – 16 184 | |||||||
Liquidity in % 2019 1 | Liquidity in % 2018 1 | Liquidity in % 2017 1 | Financial debt in % 2019 2 | Financial debt in % 2018 2 | Financial debt in % 2017 2 | ||||||||
| USD | 72 | 83 | 77 | 53 | 60 | 63 | |||||||
| CHF | 14 | 7 | 5 | 12 | 10 | 11 | |||||||
| EUR | 7 | 6 | 8 | 29 | 25 | 20 | |||||||
| JPY | 1 | 1 | 3 | 3 | 4 | ||||||||
| Other | 6 | 4 | 9 | 3 | 2 | 2 | |||||||
| 100 | 100 | 100 | 100 | 100 | 100 | ||||||||
1 Liquidity includes cash and cash equivalents, marketable securities, commodities and time deposits. | |||||||||||||
2 Financial debt includes non-current and current financial debt. | |||||||||||||
| 2019 | 2018 | 2017 | |||||||||||
Currency | Net sales % | Operating expenses % 1 | Net sales % | Operating expenses % 1 | Net sales % | Operating expenses % 1 | |||||||
| US dollar (USD) | 37 | 36 | 35 | 31 | 36 | 37 | |||||||
| Euro (EUR) | 28 | 26 | 29 | 27 | 28 | 24 | |||||||
| Swiss franc (CHF) | 2 | 16 | 2 | 20 | 2 | 17 | |||||||
| Japanese yen (JPY) | 6 | 3 | 6 | 3 | 6 | 5 | |||||||
| Chinese yuan (CNY) | 5 | 4 | 4 | 3 | 4 | 3 | |||||||
| Canadian dollar (CAD) | 3 | 2 | 3 | 2 | 3 | 1 | |||||||
| British pound (GBP) | 2 | 2 | 2 | 2 | 2 | 2 | |||||||
| Brazilian real (BRL) | 2 | 1 | 2 | 1 | 2 | 1 | |||||||
| Russian ruble (RUB) | 2 | 1 | 2 | 1 | 2 | 1 | |||||||
| Australian dollar (AUD) | 1 | 1 | 1 | 1 | 2 | 1 | |||||||
| Other currencies | 12 | 8 | 14 | 9 | 13 | 8 | |||||||
1 Operating expenses include cost of goods sold; selling, general and administration; research and development; other income and other expense. | |||||||||||||
| Average for year | Year-end | ||||||||||||
| USD per unit | 2019 | 2018 | Change in % | 2019 | 2018 | Change in % | |||||||
| Australian dollar (AUD) | 0.695 | 0.748 | – 7 | 0.701 | 0.707 | – 1 | |||||||
| Brazilian real (BRL) | 0.254 | 0.275 | – 8 | 0.249 | 0.258 | – 3 | |||||||
| Canadian dollar (CAD) | 0.754 | 0.772 | – 2 | 0.767 | 0.735 | 4 | |||||||
| Swiss franc (CHF) | 1.006 | 1.023 | – 2 | 1.032 | 1.014 | 2 | |||||||
| Chinese yuan (CNY) | 0.145 | 0.151 | – 4 | 0.144 | 0.145 | – 1 | |||||||
| Euro (EUR) | 1.120 | 1.181 | – 5 | 1.121 | 1.144 | – 2 | |||||||
| British pound (GBP) | 1.277 | 1.336 | – 4 | 1.313 | 1.274 | 3 | |||||||
| Japanese yen (JPY (100)) | 0.918 | 0.906 | 1 | 0.920 | 0.907 | 1 | |||||||
| Russian ruble (RUB (100)) | 1.546 | 1.600 | – 3 | 1.613 | 1.437 | 12 | |||||||
| Average for year | Year-end | ||||||||||||
| USD per unit | 2018 | 2017 | Change in % | 2018 | 2017 | Change in % | |||||||
| Australian dollar (AUD) | 0.748 | 0.766 | – 2 | 0.707 | 0.779 | – 9 | |||||||
| Brazilian real (BRL) | 0.275 | 0.313 | – 12 | 0.258 | 0.302 | – 15 | |||||||
| Canadian dollar (CAD) | 0.772 | 0.771 | 0 | 0.735 | 0.797 | – 8 | |||||||
| Swiss franc (CHF) | 1.023 | 1.016 | 1 | 1.014 | 1.024 | – 1 | |||||||
| Chinese yuan (CNY) | 0.151 | 0.148 | 2 | 0.145 | 0.154 | – 6 | |||||||
| Euro (EUR) | 1.181 | 1.129 | 5 | 1.144 | 1.195 | – 4 | |||||||
| British pound (GBP) | 1.336 | 1.288 | 4 | 1.274 | 1.347 | – 5 | |||||||
| Japanese yen (JPY (100)) | 0.906 | 0.892 | 2 | 0.907 | 0.888 | 2 | |||||||
| Russian ruble (RUB (100)) | 1.600 | 1.715 | – 7 | 1.437 | 1.734 | – 17 | |||||||
Change in USD % 2019 | Change in constant currencies % 2019 | Percentage point currency impact 2019 | Change in USD % 2018 | Change in constant currencies % 2018 | Percentage point currency impact 2018 | ||||||||
| Total Group | |||||||||||||
| Net sales to third parties from continuing operations | 6 | 9 | – 3 | 6 | 5 | 1 | |||||||
| Operating income from continuing operations | 8 | 14 | – 6 | – 3 | – 3 | 0 | |||||||
| Net income from continuing operations | – 44 | – 41 | – 3 | 71 | 71 | 0 | |||||||
| Basic earnings per share from continuing operations (USD) | – 43 | – 40 | – 3 | 73 | 73 | 0 | |||||||
| Core operating income from continuing operations | 12 | 17 | – 5 | 7 | 7 | 0 | |||||||
| Core net income from continuing operations | 11 | 15 | – 4 | 4 | 4 | 0 | |||||||
| Core basic earnings per share from continuing operations (USD) | 12 | 17 | – 5 | 6 | 5 | 1 | |||||||
| Innovative Medicines | |||||||||||||
| Net sales to third parties | 8 | 11 | – 3 | 8 | 8 | 0 | |||||||
| Operating income | 18 | 24 | – 6 | 4 | 4 | 0 | |||||||
| Core operating income | 13 | 18 | – 5 | 11 | 11 | 0 | |||||||
| Sandoz | |||||||||||||
| Net sales to third parties | – 1 | 2 | – 3 | – 2 | – 3 | 1 | |||||||
| Operating income | – 59 | – 53 | – 6 | – 3 | – 2 | – 1 | |||||||
| Core operating income | 5 | 10 | – 5 | – 4 | – 3 | – 1 | |||||||
| Corporate | |||||||||||||
| Operating loss | 6 | 4 | 2 | nm | nm | nm | |||||||
| Core operating loss | – 6 | – 9 | 3 | – 55 | – 52 | – 3 | |||||||
| nm = not meaningful | |||||||||||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Operating income from continuing operations | 9 086 | 8 403 | 8 702 | ||||
| Adjustments for non-cash items | |||||||
| Depreciation, amortization and impairments | 5 788 | 5 217 | 4 963 | ||||
| Change in provisions and other non-current liabilities | 1 871 | 895 | 86 | ||||
| Other | – 476 | – 229 | – 465 | ||||
| Operating income adjusted for non-cash items | 16 269 | 14 286 | 13 286 | ||||
| Dividends received from associated companies and others | 463 | 719 | 987 | ||||
| Interest and other financial receipts | 242 | 459 | 97 | ||||
| Interest and other financial payments | – 826 | – 847 | – 967 | ||||
| Taxes paid | – 1 876 | – 1 506 | – 1 487 | ||||
| Payments out of provisions and other net cash movements in non-current liabilities | – 924 | – 638 | – 829 | ||||
| Change in inventory and trade receivables less trade payables | – 809 | – 679 | – 776 | ||||
| Change in other net current assets and other operating cash flow items | 1 008 | 1 255 | 1 108 | ||||
| Net cash flows from operating activities from continuing operations | 13 547 | 13 049 | 11 419 | ||||
| Purchase of property, plant and equipment | – 1 379 | – 1 254 | – 1 325 | ||||
| Proceeds from sales of property, plant and equipment | 857 | 102 | 91 | ||||
| Purchase of intangible assets | – 878 | – 1 394 | – 969 | ||||
| Proceeds from sales of intangible assets | 973 | 823 | 640 | ||||
| Purchase of financial assets | – 302 | – 205 | – 354 | ||||
Proceeds from sales of financial assets 1 | 176 | 165 | 328 | ||||
| Purchase of other non-current assets | – 60 | – 39 | – 40 | ||||
| Proceeds from sales of other non-current assets | 3 | 9 | 1 | ||||
| Free cash flow from continuing operations | 12 937 | 11 256 | 9 791 | ||||
Free cash flow from discontinued operations 2 | – 62 | 461 | 637 | ||||
| Total free cash flow | 12 875 | 11 717 | 10 428 | ||||
1 For the free cash flow, proceeds from the sales of financial assets exclude the cash inflows from the sale of a portion of the Alcon Inc. shares received by certain consolidated foundations through the Alcon spin-off, which amounted to USD 976 million. See "Item 18. Financial Statements - Note 2 Significant transactions in 2019." | |||||||
2 In 2019, the free cash flow from discontinued operations was a cash outflow of USD 62 million (2018: USD 461 million cash inflow, 2017: USD 637 million cash inflow) consisting of USD 78 million net cash inflows from operating activities from discontinued operations (2018 and 2017: USD 1.2 billion), USD 1.2 billion net cash flows used in investing activities from discontinued operations (2018: USD 1.0 billion, 2017: USD 775 million) adjusted by USD 362 million of net cash outflows for acquisition and divestments of businesses (2018: USD 239 million, 2017: USD 210 million) and by USD 657 million for cash outflows attributable to the spin-off of the Alcon business (2018 and 2017: nil). | |||||||
| (USD millions) | Dec 31, 2019 | Dec 31, 2018 | Change | ||||
| Assets | |||||||
| Property, plant and equipment | 12 069 | 15 696 | – 3 627 | ||||
| Right-of-use assets | 1 677 | 1 677 | |||||
| Goodwill | 26 524 | 35 294 | – 8 770 | ||||
| Intangible assets other than goodwill | 28 787 | 38 719 | – 9 932 | ||||
| Financial and other non-current assets | 19 809 | 20 291 | – 482 | ||||
| Total non-current assets | 88 866 | 110 000 | – 21 134 | ||||
| Inventories | 5 982 | 6 956 | – 974 | ||||
| Trade receivables | 8 301 | 8 727 | – 426 | ||||
| Other current assets and income tax receivable | 2 934 | 3 109 | – 175 | ||||
| Cash, marketable securities, commodities, time deposits and derivative financial instruments | 11 446 | 15 964 | – 4 518 | ||||
| Assets of disposal group held for sale | 841 | 807 | 34 | ||||
| Total current assets | 29 504 | 35 563 | – 6 059 | ||||
| Total assets | 118 370 | 145 563 | – 27 193 | ||||
| Equity and liabilities | |||||||
| Total equity | 55 551 | 78 692 | – 23 141 | ||||
| Financial debts | 20 353 | 22 470 | – 2 117 | ||||
| Lease liabilities | 1 703 | 1 703 | |||||
| Deferred tax liabilities | 5 867 | 7 475 | – 1 608 | ||||
| Provisions and other non-current liabilities | 6 632 | 7 319 | – 687 | ||||
| Total non-current liabilities | 34 555 | 37 264 | – 2 709 | ||||
| Trade payables | 5 424 | 5 556 | – 132 | ||||
| Financial debts and derivative financial instruments | 7 031 | 9 678 | – 2 647 | ||||
| Lease liabilities | 246 | 246 | |||||
| Provisions and other current liabilities and current income tax liabities | 15 532 | 14 322 | 1 210 | ||||
| Liabilities of disposal group held for sale | 31 | 51 | – 20 | ||||
| Total current liabilities | 28 264 | 29 607 | – 1 343 | ||||
| Total liabilities | 62 819 | 66 871 | – 4 052 | ||||
| Total equity and liabilities | 118 370 | 145 563 | – 27 193 | ||||
| (USD millions) | 2019 | 2018 | |||
| Not overdue | 7 763 | 7 916 | |||
| Past due for not more than one month | 161 | 296 | |||
| Past due for more than one month but less than three months | 123 | 194 | |||
| Past due for more than three months but less than six months | 103 | 136 | |||
| Past due for more than six months but less than one year | 96 | 98 | |||
| Past due for more than one year | 150 | 213 | |||
| Provisions for doubtful trade receivables | – 95 | – 126 | |||
| Total trade receivables, net | 8 301 | 8 727 | |||
| Number of outstanding shares (in millions) | Issued share capital and reserves attributable to Novartis AG shareholders | ||||||||||||
2019 | 2018 | Change | 2019 USD millions | 2018 USD millions | Change USD millions | ||||||||
| Balance at beginning of year | 2 311.2 | 2 317.5 | – 6.3 | 78 614 | 74 168 | 4 446 | |||||||
Impact of change in accounting policy 1 | 3 | 60 | – 57 | ||||||||||
| Restated equity at January 1 | 78 617 | 74 228 | 4 389 | ||||||||||
| Shares acquired to be canceled | – 60.3 | – 23.3 | – 37.0 | – 5 351 | – 1 859 | – 3 492 | |||||||
| Other share purchases | – 1.7 | – 1.2 | – 0.5 | – 160 | – 114 | – 46 | |||||||
| Exercise of options and employee transactions | 5.5 | 7.8 | – 2.3 | 210 | 434 | – 224 | |||||||
| Other share sales | 3.0 | – 3.0 | 263 | – 263 | |||||||||
| Equity-based compensation | 9.4 | 7.4 | 2.0 | 833 | 756 | 77 | |||||||
| Shares delivered to Alcon employees as a result of the Alcon spin-off | 0.9 | 0.9 | 18 | 18 | |||||||||
Taxes on treasury share transactions 2 | – 189 | – 189 | |||||||||||
| Decrease/(increase) of treasury share repurchase obligation under a share buyback trading plan | 284 | – 284 | 568 | ||||||||||
Transaction costs, net of taxes 3 | – 253 | – 79 | – 174 | ||||||||||
| Dividends | – 6 645 | – 6 966 | 321 | ||||||||||
Dividend in kind to effect the spin-off of Alcon Inc. 4 | – 23 434 | – 23 434 | |||||||||||
| Net income of the year attributable to shareholders of Novartis AG | 11 732 | 12 611 | – 879 | ||||||||||
| Impact of change in ownership of consolidated entities | – 3 | – 13 | 10 | ||||||||||
| Other comprehensive income attributable to shareholders of Novartis AG | – 207 | – 401 | 194 | ||||||||||
Other movements 5 | 22 | 38 | – 16 | ||||||||||
| Balance at end of year | 2 265.0 | 2 311.2 | – 46.2 | 55 474 | 78 614 | – 23 140 | |||||||
1 In 2019, the impact of change in accounting policy includes USD 3 million related to the implementation of IFRS 16 Leases (see “Item 18. Financial Statements—Note 1. Significant accounting policies"). In 2018, the impact of change in accounting policy includes USD 60 million related to the implementation of IFRS 15, and USD 177 million related to the implementation of IFRS 9. | |||||||||||||
2 Included in 2019 is a USD 69 million impact related to the revaluation of deferred tax liability on treasury shares. This revaluation resulted from the Swiss federal tax reform enacted in May 2019 (see “Item 18. Financial Statements—Note 12. Deferred tax assets and liabilities"). | |||||||||||||
3 Transaction costs, net of tax of USD 36 million (2018: USD 20 milion), directly attributable to the potential distribution (spin-off) of Alcon to Novartis shareholders (see “Item 18. Financial Statements—Note 1. Significant accounting policies"). | |||||||||||||
4 Fair value of the dividend in kind of Alcon Inc. shares to Novartis AG shareholders and ADR (American Depositary Receipt) holders approved at the 2019 Annual General Meeting held on February 28, 2019. Distribution was effected on April 8, 2019, whereby each Novartis AG shareholder and ADR holder received one Alcon Inc. share for every five Novartis AG shares/ADRs they held on April 8, 2019, close of business (see “Item 18. Financial Statements—Note 1. Significant accounting policies"). | |||||||||||||
5 Impact of hyperinflationary economies (see “Item 18. Financial Statements—Note 1. Significant accounting policies"). | |||||||||||||
5.C Research and development, patents and licenses
5.D Trend information
5.E Off-balance sheet arrangements
5.F Tabular disclosure of contractual obligations
| Payments due by period | |||||||||||
(USD millions) | Total | Less than 1 year | 2–3 years | 4–5 years | After 5 years | ||||||
| Non-current financial debt, including current portion | 22 355 | 2 002 | 4 650 | 4 460 | 11 243 | ||||||
| Interest on non-current financial debt, including current portion | 5 434 | 464 | 845 | 686 | 3 439 | ||||||
| Lease liabilities, non-current and current portion | 1 949 | 246 | 365 | 257 | 1 081 | ||||||
| Interest on lease liabilities, non-current and current portion | 1 536 | 49 | 83 | 66 | 1 338 | ||||||
| Unfunded pensions and other post-employment benefit plans | 1 748 | 101 | 201 | 208 | 1 238 | ||||||
| Research and development potential milestone commitments | 4 404 | 809 | 761 | 891 | 1 943 | ||||||
| Contingent consideration liabilities | 1 036 | 71 | 352 | 230 | 383 | ||||||
| Property, plant and equipment purchase commitments | 220 | 177 | 43 | ||||||||
Acquisition of business commitments 1 | 10 164 | 10 130 | 34 | ||||||||
| Total contractual cash obligations | 48 846 | 14 049 | 7 334 | 6 798 | 20 665 | ||||||
1 For acquisition of business commitments, please refer to "Item 18. Financial Statements - Note 2 Significant transactions - significant pending transactions". | |||||||||||
Item 6. Directors, Senior Management and Employees
6.A Directors and senior management
6.B Compensation

Compensation at a glance

| Decision on | Decision-making authority | ||
| Compensation of Chairman and other Board members | Board of Directors | ||
| Compensation of CEO | Board of Directors | ||
| Compensation of other Executive Committee members | Compensation Committee | ||
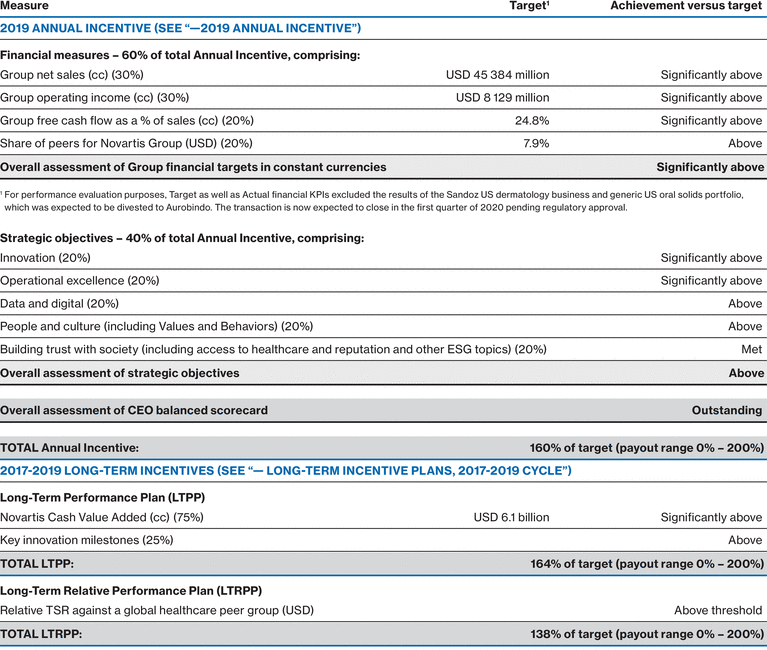

CHF 000s | AGM 2019-2020 annual fee | ||
| Chairman of the Board | 3 800 | ||
| Board membership | 280 | ||
| Vice Chairman | 50 | ||
| Chair of the Audit and Compliance Committee | 130 | ||
| Chair of the Compensation Committee | 90 | ||
| Chair of the following committees: • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee | 70 | ||
| Membership of the Audit and Compliance Committee | 70 | ||
| Membership of the following committees: • Compensation Committee • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee | 40 | ||
CHF 000s | 2019 total compensation 1 | ||
| Chairman of the Board | 3 804 | ||
| Other 12 members of the Board | 4 387 | ||
| Total | 8 191 | ||
1 Includes an amount of CHF 21'002 for mandatory employer contributions for all Board members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 413'985 , and provides a right to the maximum future insured government pension benefit for the Board member. | |||
Executive Committee compensation philosophy and principles



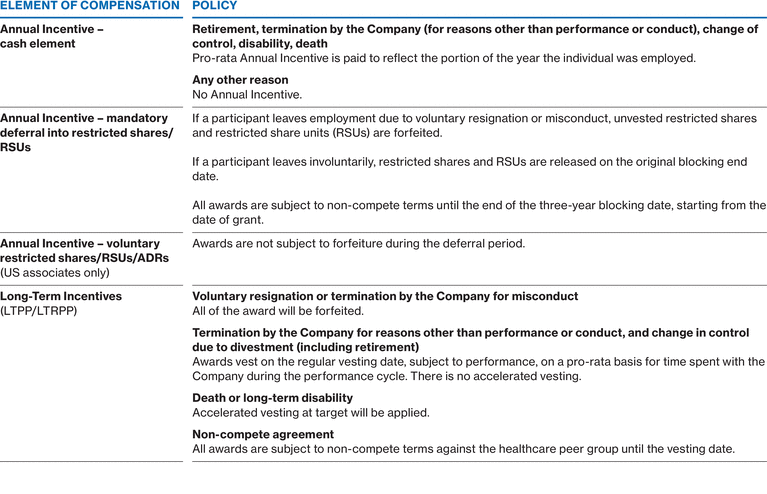
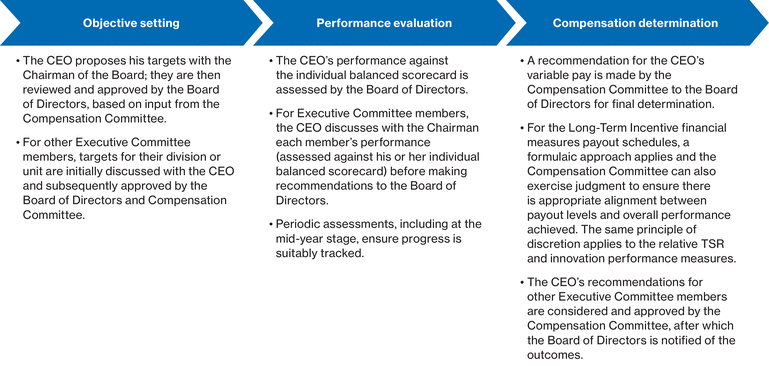
2019 Executive Committee compensation







| Name | Date of appointment | Cash payments (CHF) | Equity awards | Total value at grant | ||
| Richard Saynor | July 15, 2019 | 157 087 | 1 034 803 | 1 191 890 | ||
| CEO, Sandoz | equal to 4'172 PSUs and 7'280 RSUs, vesting over the period 2020-2022 | |||||
| 2019 annual base salary | 2019 pension benefits1 | 2019 Annual Incentive | Long-Term Incentives | Other 2019 compensation | |||||||||||||||
| LTPP 2017-2019 cycle | LTRPP 2017-2019 cycle | ||||||||||||||||||
Currency | Cash (amount) | Amount | Cash | Equity2 | Equity (value at vesting date)3 | Equity (value at vesting date)3 | Amount2,4,5 | Total realized compensation (incl. share price movement)6 | |||||||||||
| Executive Committee members | |||||||||||||||||||
| Vasant Narasimhan (CEO) | CHF | 1 653 333 | 165 547 | 2 008 800 | 2 008 839 | 3 510 963 | 1 107 806 | 160 452 | 10 615 740 | ||||||||||
Aggregate realized compensation of the other 14 Executive Committee members, including the two members who stepped down during financial year 2019 7, 8 | CHF | 9 370 547 | 2 131 905 | 5 809 455 | 7 013 842 | 17 932 704 | 6 383 700 | 7 233 594 | 55 875 748 | ||||||||||
| Total | CHF | 11 023 880 | 2 297 452 | 7 818 255 | 9 022 682 | 21 443 667 | 7 491 506 | 7 394 046 | 66 491 488 | ||||||||||
| See “—2018 realized compensation for the CEO and other Executive Committee members” for 2018 comparative figures. | |||||||||||||||||||
1 Includes mandatory employer contributions of CHF 4'373 for the CEO and CHF 63'461 for the other Executive Committee members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 3'923'070 paid in 2019 for all Executive Committee members, and provides a right to the maximum future insured government pension benefit for the Executive Committee member. | |||||||||||||||||||
2 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 21, 2020) of CHF 92.89 per Novartis share and USD 95.19 per ADR. | |||||||||||||||||||
3 The amounts represent the underlying share value of the 232'425 LTPP PSUs and 77'904 LTRPP PSUs vesting on January 17, 2020, to the CEO and other Executive Committee members for the performance cycle 2017-2019, inclusive of earned dividend equivalents for the three-year cycle (details in “—2017-2019 performance cycle LTPP and 2017-2019 performance cycle LTRPP").The taxable value is determined using the closing share price on the day the Novartis Board of Directors approved the final LTPP and LTRPP performance factors (i.e., January 21, 2020) of 92.89 per Novartis share and USD 95.19 per ADR. Shannon Thyme Klinger, Stefan Lang, Susanne Schaffert and Marie-France Tschudin were promoted to the Executive Committee during the course of the performance period 2017-2019, and as such, the information disclosed reflects their pro-rata LTPP & LTRPP 2017-2019 payouts attributable to the period they were a member of the Executive Committee. Bertrand Bodson, Klaus Moosmayer, John Tsai, Robert Weltevreden and Richard Saynor joined Novartis post the 2017 LTI awards being made and hence did not receive LTPP & LTRPP awards for the 2017-2019 performance period. | |||||||||||||||||||
4 Includes any other perquisites, benefits in kind, international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization) as well as vested shares under LTPP & LTRPP after the step down date. | |||||||||||||||||||
5 Includes 5 430 vested RSUs (CHF 502 003) on July 28, 2019, to John Tsai, in lieu of the LTI that he forfeited when leaving his previous employer and 1 323 vested RSUs (CHF 123 092) and 14 470 vested PSUs (CHF 1 346 289) on March 24, 2019, to Paul Hudson in lieu of the LTI that he forfeited when leaving his previous employer. The PSUs had the same performance measures as the LTPP for the 2016-2018 performance cycle (NCVA and long-term innovation). | |||||||||||||||||||
6 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member. | |||||||||||||||||||
7 Comprises the compensation of Richard Francis, the former CEO of Sandoz including the vesting of his Long-Term Incentives for performance cycle 2017-2019, as per the plan rules. Unvested shares for Paul Hudson were forfeited upon his departure from the Company. See “—Executive Committee member departures" for details. | |||||||||||||||||||
8 Amounts for Executive Committee members paid in USD were converted at a rate of USD 1.00 = CHF 0.9938, which is the same average exchange rate used in the Group's 2019 consolidated financial statements. | |||||||||||||||||||
| PSUs at grant | Shares delivered at vesting | ||||||||||||||||
PSUs (target number) | PSUs (target value at grant date) (CHF) 2 | Payout factor for LTPP (% of target) | Performance shares delivered at vesting (number) | Performance shares delivered at vesting (value at vesting date) (CHF) 3 | Dividend equivalent shares delivered at vesting (number) 4 | Dividend equivalent shares delivered at vesting (value at vesting date) (CHF) | Total shares delivered at vesting (value at vesting date) (CHF) | ||||||||||
Executive Committee members 1 | |||||||||||||||||
| Vasant Narasimhan | 21 323 | 1 980 693 | 164% | 34 970 | 3 248 363 | 2 827 | 262 600 | 3 510 963 | |||||||||
Other 14 Executive Committee members, including the two members who stepped down during financial year 2019 5 | 109 616 | 10 247 244 | 164% | 178 984 | 16 735 046 | 13 151 | 1 227 397 | 17 932 704 | |||||||||
| Total | 130 939 | 12 227 938 | 213 954 | 19 983 409 | 15 978 | 1 489 997 | 21 443 667 | ||||||||||
1 Shannon Thyme Klinger, Stefan Lang, Susanne Schaffert and Marie-France Tschudin joined the Executive Committee during the course of the performance period 2017-2019. As such, the information disclosed reflects their pro-rata LTPP 2017-2019 payout attributable to the period they were a member of the Executive Committee. Bertrand Bodson, Klaus Moosmayer, John Tsai, Robert Weltevreden and Richard Saynor joined Novartis post the 2017 LTPP awards being made and hence did not receive an LTPP award for the 2017-2019 performance period. | |||||||||||||||||
2 The shown amounts represent the underlying share value of the target number of PSUs granted to each Executive Committee member for the performance period 2017-2019, based on the closing share price on the grant date (January 17, 2017) of CHF 71.35 per Novartis share and USD 71.99 per ADR. | |||||||||||||||||
3 The shown amounts represent the underlying share value of the target number of PSUs vested for the performance period 2017-2019, based on the last closing share price on the day the Novartis Board of Directors approved the final LTPP and LTRPP performance payout factors (i.e., January 21, 2020) of CHF 92.89 per Novartis share and USD 95.19 per ADR. | |||||||||||||||||
4 Dividend equivalent shares are calculated on the dividend each member of the Executive Committee would have received, based on the actual number of shares delivered at the end of the performance period 2017-2019. At vesting, the dividend equivalents are credited in shares or ADRs. | |||||||||||||||||
5 Includes the LTPP vesting for Richard Francis, the former CEO, Sandoz for performance cycle 2017-2019, as per the plan rules. The LTPP vesting for the former CEO, Novartis Pharmaceuticals, Paul Hudson were forfeited on August 31, 2019, at his departure from the Company. See “—LTPP perfomance outcomes" for further details. | |||||||||||||||||
| PSUs at grant | Shares delivered at vesting | ||||||||||||||||
PSUs (target number) | PSUs (target value at grant date) (CHF) 2 | Payout factor for LTRPP (% of target) | Performance shares delivered at vesting (number) | Performance shares delivered at vesting (value at vesting date) (CHF) 3 | Dividend equivalent shares delivered at vesting (number) 4 | Dividend equivalent shares delivered at vesting (value at vesting date) (CHF) | Total shares delivered at vesting (value at vesting date) (CHF) | ||||||||||
Executive Committee members 1 | |||||||||||||||||
| Vasant Narasimhan | 7 996 | 742 748 | 138% | 11 034 | 1 024 948 | 892 | 82 858 | 1 107 806 | |||||||||
Other 14 Executive Committee members, including the two members who stepped down during financial year 2019 5 | 45 858 | 4 290 344 | 138% | 63 283 | 5 920 579 | 5 077 | 474 933 | 6 383 700 | |||||||||
| Total | 53 854 | 5 033 093 | 74 317 | 6 945 527 | 5 969 | 557 791 | 7 491 506 | ||||||||||
1 Shannon Thyme Klinger, Stefan Lang, Susanne Schaffert and Marie-France Tschudin joined the Executive Committee during the course of the performance period 2017-2019. As such, the information disclosed reflects their pro-rata LTRPP 2017-2019 payout attributable to the period they were a member of the Executive Committee. Bertrand Bodson, Klaus Moosmayer, John Tsai, Robert Weltevreden and Richard Saynor joined Novartis post the 2017 LTRPP awards being made and hence did not receive an LTRPP award for the 2017-2019 performance period. | |||||||||||||||||
2 The shown amounts represent the underlying share value of the target number of PSUs granted to each Executive Committee member for the performance period 2017-2019, based on the closing share price on the grant date (January 17, 2017) of CHF 71.35 per Novartis share and USD 71.99 per ADR. | |||||||||||||||||
3 The shown amounts represent the underlying share value of the target number of PSUs vested for the performance period 2017-2019, based on the last closing share price on the day the Novartis Board of Directors approved the final LTPP and LTRPP performance payout factors (i.e., January 21, 2020) of CHF 92.89 per Novartis share and USD 95.19 per ADR. | |||||||||||||||||
4 Dividend equivalent shares are calculated on the dividend each member of the Executive Committee would have received, based on the actual number of shares delivered at the end of the performance period 2017-2019. At vesting, the dividend equivalents are credited in shares or ADRs. | |||||||||||||||||
5 Includes the LTRPP vesting for the CEO of Sandoz for performance cycle 2017-2019, as per the plan rules. The LTRPP vesting for Paul Hudson were forfeited on August 31, 2019, at his departure from the Company. See “—LTRPP perfomance outcomes" for further details. | |||||||||||||||||
| 2018 annual base salary | 2018 pension benefits1 | 2018 Annual Incentive | Long-Term Incentives | Other 2018 compensation | |||||||||||||||
| LTPP 2016-2018 cycle | LTRPP 2016-2018 cycle | ||||||||||||||||||
Currency | Cash (amount) | Amount | Cash | Equity2 | Equity (value at vesting date)3 | Equity (value at vesting date) | Amount2,4 | Total realized compensation (incl. share price movement)5 | |||||||||||
| Executive Committee members | |||||||||||||||||||
| Vasant Narasimhan (CEO from February 1, 2018) | CHF | 1 491 667 | 168 233 | 1 594 801 | 1 594 805 | 1 796 381 | 0 | 34 401 | 6 680 288 | ||||||||||
Aggregate realized compensation of the other 16 Executive Committee members, including the four members who stepped down during financial year 2018 6, 7 | CHF | 9 297 021 | 1 874 671 | 5 727 765 | 5 532 316 | 24 079 974 | 0 | 13 131 653 | 59 643 400 | ||||||||||
| Total | CHF | 10 788 688 | 2 042 904 1 | 7 322 566 | 7 127 121 | 25 876 355 | 0 | 13 166 054 | 66 323 688 | ||||||||||
1 Includes mandatory employer contributions of CHF 4 336 for the CEO and CHF 78 403 for the other Executive Committee members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 2 847 422 paid in 2018 for all Executive Committee members, and provides a right to the maximum future insured government pension benefit for the Executive Committee member. | |||||||||||||||||||
2 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 22, 2019) of CHF 88.14 per Novartis share and USD 88.32 per ADR. | |||||||||||||||||||
3 The amounts represent the underlying share value of the 294 971 PSUs vesting on January 22, 2019, to the CEO and other Executive Committee members for the performance cycle 2016-2018, inclusive of earned dividend equivalents for the three-year cycle. The taxable value is determined using the closing share price on the day the Novartis Board approved the final LTPP and LTRPP performance factors (i.e., January 22, 2019) of CHF 88.14 per Novartis share and USD 88.32 per ADR. Vasant Narasimhan, Shannon Thyme Klinger, Stefan Lang and André Wyss joined the Executive Committee during the course of the performance period 2016-2018, and as such, the information disclosed reflects their pro-rata LTPP 2016-2018 payout attributable to the period they were a member of the Executive Committee. Elizabeth Barrett, Bertrand Bodson, Paul Hudson, Klaus Moosmayer, John Tsai and Robert Weltevreden joined post the 2016 LTPP awards being made and hence did not receive an LTPP award for the 2016-2018 performance period. | |||||||||||||||||||
4 Includes any other perquisites, benefits in kind, international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization) as well as vested shares under LTPP after the step down date. | |||||||||||||||||||
5 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member. | |||||||||||||||||||
6 Comprises the compensation of the outgoing CEO, General Counsel, CEO of Alcon, and President of Novartis Operations and Country President Switzerland, including the vesting of their Long-Term Incentives for performance cycle 2016-2018, as per the plan rules. | |||||||||||||||||||
7 Amounts for Executive Committee members paid in USD were converted at a rate of UDS 1.00 = CHF 0.978, which is the same average exchange rate used in the Group's 2018 consolidated financial statements. | |||||||||||||||||||
| Fixed compensation and pension benefits | Variable compensation | ||||||||||||||||
| Actual compensation paid or granted for 2019 | Long-Term Incentive 2019-2021 cycle grants at target | ||||||||||||||||
| 2019 annual base salary | 2019 pension benefits | 2019 Annual Incentive (performance achieved) | LTPP 2019-2021 cycle | Other 2019 compensation | Total compensation paid, promised or granted 2019 | ||||||||||||
Currency | Cash (amount) | Amount 1 | Cash (amount) | Equity (value at grant date) 2 | PSUs (target value at grant date) 3 | Amount 4 | Amount 5 | ||||||||||
| Executive Committee members active on December 31, 2019 | |||||||||||||||||
| Vasant Narasimhan | CHF | 1 653 333 | 165 547 | 2 008 800 | 2 008 839 | 5 440 530 | 160 452 | 11 437 501 | |||||||||
| Steven Baert | CHF | 789 750 | 161 454 | 633 360 | 633 417 | 1 662 585 | 124 979 | 4 005 545 | |||||||||
| Bertrand Bodson | CHF | 607 500 | 170 178 | 341 040 | 341 092 | 974 476 | 137 826 | 2 572 111 | |||||||||
James Bradner 6 | USD | 1 126 781 | 359 961 | 951 720 | 951 805 | 2 832 511 | 85 498 | 6 308 275 | |||||||||
| Harry Kirsch | CHF | 1 053 000 | 164 467 | 1 045 044 | 1 045 105 | 2 744 591 | 27 658 | 6 079 865 | |||||||||
| Shannon Thyme Klinger | CHF | 783 333 | 188 990 | 468 000 | 468 073 | 1 600 005 | 111 375 | 3 619 776 | |||||||||
| Steffen Lang | CHF | 745 000 | 167 815 | 408 000 | 612 145 | 1 200 026 | 9 810 | 3 142 796 | |||||||||
| Klaus Moosmayer | CHF | 500 000 | 125 483 | 260 000 | 260 092 | 800 047 | 171 749 | 2 117 371 | |||||||||
Richard Saynor (from July 15, 2019) 7 | CHF | 356 021 | 87 118 | 179 315 | 179 371 | – | 1 950 908 | 2 752 732 | |||||||||
Susanne Schaffert 7 | CHF | 850 000 | 167 096 | 459 000 | 1 071 115 | 1 870 066 | 160 252 | 4 577 528 | |||||||||
| John Tsai | CHF | 858 333 | 181 048 | 602 000 | 602 020 | 2 064 063 | 377 544 | 4 685 007 | |||||||||
Marie-France Tschudin (from June 7, 2019) 7 | CHF | 481 667 | 92 090 | 290 630 | 290 653 | 968 249 | – | 2 123 289 | |||||||||
| Robert Weltevreden | CHF | 607 500 | 157 423 | 158 340 | 475 132 | 974 476 | 4 860 | 2 377 731 | |||||||||
| Subtotal | 10 405 223 | 2 186 434 | 7 799 341 | 8 932 950 | 23 114 040 | 3 322 378 | 55 760 366 | ||||||||||
| Executive Committee members who stepped down during 2019 | |||||||||||||||||
Richard Francis (until March 19, 2019) 8, 9 | CHF | 179 315 | 36 025 | 18 914 | 37 435 | 720 460 | 3 808 445 | 4 800 594 | |||||||||
Paul Hudson (until June 7, 2019) 9, 10 | CHF | 439 342 | 74 994 | – | – | 270 451 | 2 885 164 | 3 669 950 | |||||||||
| Subtotal | 618 657 | 111 018 | 18 914 | 37 435 | 990 910 | 6 693 609 | 8 470 543 | ||||||||||
| Total | 11 023 880 | 2 297 452 | 7 818 255 | 8 970 384 | 24 104 951 | 10 015 988 | 64 230 910 | ||||||||||
| Based on assumption of 100% payout at target. Actual payout (0-200% of target) will be known at the end of the three-year cycle in January 2022. | |||||||||||||||||
| See next page for 2018 comparative figures. | |||||||||||||||||
1 Includes mandatory employer contributions of CHF 4'373 for the CEO and CHF 63'461 for the other Executive Committee members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 3'923'070 paid in 2019 for all Executive Committee members, and provides a right to the maximum future insured government pension benefit for the Executive Committee member. | |||||||||||||||||
2 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 21, 2020) of CHF 92.89 per Novartis share and USD 95.19 per ADR. | |||||||||||||||||
3 The amounts represent the underlying share value of the target number of PSUs granted to Executive Committee members for the performance cycle 2019-2021, based on the closing share price on the grant date (January 22, 2019) of CHF 88.14 per Novartis share and USD 88.32 per ADR for all members except for Mr. Richard Saynor, who was not part of the Company at the annual grant date and hence did not receive an LTPP award. | |||||||||||||||||
4 Includes any other perquisites, benefits in kind, and international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization) | |||||||||||||||||
5 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member. | |||||||||||||||||
6 Amounts in USD for James Bradner were converted at a rate of CHF 1.00 = USD 1.006 , which is the average rate used in the Group’s 2019 consolidated financial statements. | |||||||||||||||||
7 For those members who joined the Executive Committee in 2019, the information under the columns “Actual compensation paid or granted for 2019” and “Long-Term Incentive 2019-2021 cycle grants at target” includes their pro-rata compensation from the date they joined the Executive Committee to December 31, 2019 or to the end of the performance cycle in the case of the “Long-Term Incentive 2019-2021 cycle grants at target”. | |||||||||||||||||
8 Richard Francis stepped down as CEO, Sandoz on March 19, 2019 and will leave the company on 31 March, 2020 in line with his contractual notice period. Until the end of the notice period, he will receive further contractual compensation that includes the base salary, Annual Incentive and pension benefits. In accordance with the plan rules, the LTPP 2019-2021 cycle grant (21 217 PSUs), included in full in the above table, will vest on the normal vesting date pro-rata based on the number of months of Novartis employment during the performance cycle. The vesting of this grant is subject to performance conditions assessed at the end of the period. | |||||||||||||||||
9 For those members leaving the Executive Committee, the columns under “Actual compensation paid or granted for 2019” and “Long-Term Incentive 2019-2021 cycle grants at target” reflect the pro-rata compensation for their period as Executive Committee member. The column “Other 2019 compensation” includes inter alia their pro-rata compensation from the date they left the Executive Committee to December 31, 2019 or to the end of the performance cycle in the case of the “Long-Term Incentive 2019-2021 cycle grants at target”. See “—2019 Executive Committee member departures" for details. | |||||||||||||||||
10 Paul Hudson stepped down as CEO, Novartis Pharmaceuticals on June 7, 2019 and left the company on August 31, 2019 in line with his reduced contractual notice period (see for more details “—2019 Executive Committee member departures”). The Annual Incentive and LTPP 2019-2021 cycle grant (31 553 PSUs) included in the table above, were forfeited in full upon his departure. | |||||||||||||||||
| Fixed compensation and pension benefits | Variable compensation | |||||||||||||||||||
| Actual compensation paid or granted for 2018 | Long-Term Incentive 2018-2020 cycle grants at target | |||||||||||||||||||
| 2018 annual base salary | 2018 pension benefits | 2018 Annual Incentive (performance achieved) | LTPP 2018-2020 cycle | LTRPP 2018-2020 cycle | Other 2018 compensation | Total compensation paid, promised or granted 2018 | ||||||||||||||
Currency | Cash (amount) | Amount 1 | Cash (amount) | Equity (value at grant date) 2 | PSUs (target value at grant date) 3 | PSUs (target value at grant date) 3 | Amount 4 | Amount 5 | ||||||||||||
| Executive Committee members active on December 31, 2018 | ||||||||||||||||||||
Vasant Narasimhan (CEO from February 1, 2018) 6 | CHF | 1 491 667 | 168 233 | 1 594 801 | 1 594 805 | 3 100 046 | 1 937 539 | 34 401 | 9 921 491 | |||||||||||
| Steven Baert | CHF | 780 000 | 152 914 | 585 000 | 585 073 | 1 170 051 | 468 053 | 77 550 | 3 818 642 | |||||||||||
Elizabeth Barrett (from February 1, 2018, to December 31, 2018) 7 | CHF | 779 167 | 174 274 | 0 | 0 | 1 360 040 | 510 057 | 2 747 859 | 5 571 397 | |||||||||||
Bertrand Bodson (from April 1, 2018) 8 | CHF | 450 000 | 97 666 | 216 986 | 217 001 | 440 614 | 110 174 | 146 478 | 1 678 918 | |||||||||||
James Bradner 9 | USD | 1 094 462 | 257 018 | 924 000 | 924 004 | 1 870 085 | 880 086 | 63 313 | 6 012 967 | |||||||||||
| Richard Francis | CHF | 850 000 | 176 368 | 382 500 | 382 528 | 1 360 057 | 510 001 | 1 790 428 | 5 451 882 | |||||||||||
| Paul Hudson | CHF | 985 000 | 180 771 | 1 007 325 | 1 007 352 | 1 683 036 | 792 027 | 94 355 | 5 749 866 | |||||||||||
| Harry Kirsch | CHF | 1 040 000 | 173 499 | 858 000 | 858 043 | 1 768 008 | 832 067 | 58 814 | 5 588 431 | |||||||||||
Shannon Thyme Klinger (from April 1, 2018) 8 | CHF | 520 833 | 103 448 | 275 770 | 275 790 | 619 595 | 185 862 | 37 118 | 2 018 416 | |||||||||||
Steffen Lang (from April 1, 2018) 8 | CHF | 540 000 | 99 535 | 260 384 | 260 454 | 596 631 | 179 064 | 8 595 | 1 944 663 | |||||||||||
| Klaus Moosmayer (from December 1, 2018) | CHF | 41 667 | 9 704 | 16 986 | 17 011 | 0 | 0 | 808 821 | 894 189 | |||||||||||
| John Tsai (from May 1, 2018) | CHF | 566 667 | 126 845 | 313 801 | 313 867 | 0 | 0 | 4 590 950 | 5 912 129 | |||||||||||
| Robert Weltevreden (from June 1, 2018) | CHF | 350 000 | 70 950 | 77 392 | 232 337 | 671 702 | 155 003 | 3 715 | 1 561 099 | |||||||||||
| Subtotal | 9 464 855 | 1 785 446 | 6 492 171 | 6 647 490 | 14 597 819 | 6 540 145 | 10 460 974 | 55 988 900 | ||||||||||||
Executive Committee members who stepped down during 2018 10 | ||||||||||||||||||||
| Joseph Jimenez (CEO until January 31, 2018) | CHF | 178 601 | 19 146 | 133 767 | 0 | 0 | 0 | 2 357 371 | 2 688 885 | |||||||||||
F. Michael Ball (until June 30, 2018) 9 | USD | 555 397 | 126 594 | 333 238 | 333 231 | 888 640 | 388 845 | 2 970 642 | 5 596 587 | |||||||||||
| Felix R. Ehrat (until May 31, 2018) | CHF | 384 740 | 68 918 | 153 896 | 153 892 | 654 081 | 230 877 | 2 346 072 | 3 992 477 | |||||||||||
André Wyss (until March 31, 2018) 11 | CHF | 217 582 | 45 646 | 216 986 | 0 | 116 060 | 43 523 | 1 375 802 | 2 015 599 | |||||||||||
| Subtotal | 1 323 833 | 257 458 | 830 395 | 479 632 | 1 638 802 | 654 503 | 8 983 098 | 14 167 721 | ||||||||||||
| Total | 10 788 688 | 2 042 904 | 7 322 566 | 7 127 122 | 16 236 621 | 7 194 648 | 19 444 072 | 70 156 621 | ||||||||||||
| Based on assumption of 100% payout at target. Actual payout (0-200% of target) will be known at the end of the three-year cycle in January 2021. | ||||||||||||||||||||
1 Includes mandatory employer contributions of CHF 4 336 for the CEO and CHF 78 403 for the other Executive Committee members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 2 847 422 paid in 2018 for all Executive Committee members, and provides a right to the maximum future insured government pension benefit for the Executive Committee member. | ||||||||||||||||||||
2 The portion of the Annual Incentive delivered in equity is rounded up to the nearest share, based on the closing share price on the grant date (January 22, 2019) of CHF 88.14 per Novartis share and USD 88.32 per ADR. | ||||||||||||||||||||
3 The amounts represent the underlying share value of the target number of PSUs granted to Executive Committee members for the performance cycle 2018-2020, based on the closing share price on the grant date (January 18, 2018) of CHF 82.90 per Novartis share and USD 86.41 per ADR for all members except Elizabeth Barrett and Robert Weltevreden. For Ms. Barrett and Mr. Weltevreden, the closing share price on the grant date was respectively CHF 83.52 on February 1, 2018, and CHF 74.70 on June 1, 2018, per Novartis share. | ||||||||||||||||||||
4 Includes any other perquisites, benefits in kind, and international assignment benefits as per the global mobility policy (e.g., housing, international health insurance, children's school fees, tax equalization) | ||||||||||||||||||||
5 All amounts are before deduction of the social security contribution and income tax due by the Executive Committee member. | ||||||||||||||||||||
6 The figures include Vasant Narasimhan's compensation of January 2018 as Head of Global Drug Development. | ||||||||||||||||||||
7 Elizabeth Barrett stepped down from the role of CEO, Novartis Oncology and from the Executive Committee as at the end of the 2018 business year. The LTPP and LTRPP grants (16 284 and 6 107 PSUs, respectively) for the 2018-2020 performance cycle, and the 2018 buyout award of 21 267 performance shares, reflected in other compensation, both included in the table above, were forfeited in full upon her departure on December 31, 2018. | ||||||||||||||||||||
8 For those members who joined the Executive Committee in 2018, the information under the columns “2018 annual base salary,” “2018 pension benefits,” “2018 Annual Incentive,” “LTPP” and “LTRPP” includes their pro-rata compensation from the date they joined the Executive Committee to December 31, 2018. | ||||||||||||||||||||
9 Amounts in USD for F. Michael Ball and James Bradner were converted at a rate of CHF 1.00 = USD 0.978, which is the same average exchange rate used in the Group's 2018 consolidated financial statements. | ||||||||||||||||||||
10 For those members who left the Executive Committee in 2018, the information under the columns “2018 annual base salary,” “2018 pension benefits,” “2018 Annual Incentive,” “LTPP” and “LTRPP” reflects the pro-rata compensation for the period they were an Executive Committee member in 2018. The information under the column “Other 2018 compensation” also includes, inter alia, their pro-rata compensation from the date they stepped down from the Executive Committee to December 31, 2018. | ||||||||||||||||||||
11 The full number of PSUs under LTPP and LTRPP 2018-2020 granted to André Wyss were 16 985 and 6 370, respectively. The amounts included under LTPP and LTRPP in the table above are disclosed on a pro-rata basis to the end of his notice period (i.e., September 30, 2018), per his contractual agreement and subject to the plan rules. | ||||||||||||||||||||
Variable compensation1 | |||||||
| 2019 Annual Incentive (performance achieved) | LTPP 2019-2021 cycle | Other | |||||
Equity (number) 2 | PSUs (target number) 3 | Equity/PSUs (number) | |||||
| Executive Committee members active on December 31, 2019 | |||||||
| Vasant Narasimhan | 21 626 | 61 726 | 0 | ||||
| Steven Baert | 6 819 | 18 863 | 0 | ||||
| Bertrand Bodson | 3 672 | 11 056 | 0 | ||||
| James Bradner | 9 999 | 32 071 | 0 | ||||
| Harry Kirsch | 11 251 | 31 139 | 0 | ||||
| Shannon Thyme Klinger | 5 039 | 18 153 | 0 | ||||
| Steffen Lang | 6 590 | 13 615 | 0 | ||||
| Klaus Moosmayer | 2 800 | 9 077 | 0 | ||||
Richard Saynor (from July 15, 2019) 4 | 1 931 | 0 | 11 452 | ||||
| Susanne Schaffert | 11 531 | 21 217 | 0 | ||||
| John Tsai | 6 481 | 23 418 | 0 | ||||
Marie-France Tschudin (from June 7, 2019) 4 | 3 129 | 11 085 | 0 | ||||
| Robert Weltevreden | 5 115 | 11 056 | 0 | ||||
| Subtotal | 95 983 | 262 476 | 11 452 | ||||
| Executive Committee members who stepped down during 2019 | |||||||
Richard Francis (until March 19, 2019) 5, 6 | 403 | 8 841 | 0 | ||||
Paul Hudson (until June 7, 2019) 5, 7 | 0 | 4 525 | 0 | ||||
| Subtotal | 403 | 13 366 | 0 | ||||
| Total | 96 386 | 275 842 | 11 452 | ||||
| See next page for 2018 comparative figures. | |||||||
1 The values of the awards are reported in the table "2019 compensation at grant value for the CEO and other Executive Committee members" in “—2019 compensation at grant value for the CEO and other Executive Committee members." | |||||||
2 Vested shares, restricted shares and/or RSUs granted under the Annual Incentive for performance period 2019 | |||||||
3 Target number of PSUs granted under the LTPP as applicable for the performance cycle 2019-2021 | |||||||
4 For those members who joined the Executive Committee in 2019, the information under the column “Variable compensation” includes their pro-rata number of equity instruments from the date they joined the Executive Committee to December 31, 2019 or to the end of the performance cycle in case of the "LTPP 2019-2021 cycle". | |||||||
5 For those members leaving the Executive Committee, the column under “Variable compensation” reflects the pro-rata number of equity instruments for their period as Executive Committee member. The column “Other” includes their pro-rata compensation from the date they left the Executive Committee to December 31, 2019 or to the end of the performance cycle in the case of the “LTPP 2019-2021 cycle”. See “—2019 Executive Committee member departures” for details. | |||||||
6 Richard Francis stepped down as CEO, Sandoz on March 19, 2019 and will leave the company on 31 March, 2020 in line with his contractual notice period. In accordance with the plan rules, the LTPP 2019-2021 cycle grant (21 217 PSUs), included in full in the above table, will vest on the normal vesting date pro-rata based on the number of months of Novartis employment during the performance cycle. The vesting of this grant is subject to performance conditions assessed at the end of the period. | |||||||
7 Paul Hudson stepped down as CEO, Novartis Pharmaceuticals on June 7, 2019 and left the company on August 31, 2019 in line with his reduced contractual notice period (see for more details “—2019 Executive Committee member departures”). The 2019 Annual Incentive and LTPP 2019-2021 cycle grant (31 553 PSUs) included in the table above, were forfeited in full upon his departure. | |||||||
Variable compensation1 | |||||||||
| 2018 Annual Incentive (performance achieved) | LTPP 2018-2020 cycle | LTRPP 2018-2020 cycle | Other | ||||||
Equity (number) 2 | PSUs (target number) 3 | PSUs (target number) 3 | Equity/PSUs (number) | ||||||
| Executive Committee members active on December 31, 2018 | |||||||||
| Vasant Narasimhan (CEO from February 1, 2018) | 18 094 | 37 395 | 23 372 | 0 | |||||
| Steven Baert | 6 638 | 14 114 | 5 646 | 0 | |||||
Elizabeth Barrett (from February 1, 2018, to December 31, 2018) 4 | 0 | 16 284 | 6 107 | 21 267 | |||||
| Bertrand Bodson (from April 1, 2018) | 2 462 | 5 315 | 1 329 | 0 | |||||
| James Bradner | 10 462 | 21 642 | 10 185 | 0 | |||||
| Richard Francis | 4 340 | 16 406 | 6 152 | 0 | |||||
| Paul Hudson | 11 429 | 20 302 | 9 554 | 0 | |||||
| Harry Kirsch | 9 735 | 21 327 | 10 037 | 0 | |||||
| Shannon Thyme Klinger (from April 1, 2018) | 3 129 | 7 474 | 2 242 | 0 | |||||
| Steffen Lang (from April 1, 2018) | 2 955 | 7 197 | 2 160 | 0 | |||||
| Klaus Moosmayer (from December 1, 2018) | 193 | 0 | 0 | 8 857 | |||||
| John Tsai (from May 1, 2018) | 3 561 | 0 | 0 | 27 381 | |||||
| Robert Weltevreden (from June 1, 2018) | 2 636 | 8 992 | 2 075 | 0 | |||||
| Subtotal | 75 634 | 176 448 | 78 859 | 57 505 | |||||
| Executive Committee members who stepped down during 2018 | |||||||||
Joseph Jimenez (CEO until January 31, 2018) 5 | 0 | 0 | 0 | 0 | |||||
| F. Michael Ball (until June 30, 2018) | 7 609 | 10 284 | 4 500 | 18 865 | |||||
| Felix R. Ehrat (until May 31, 2018) | 4 221 | 7 890 | 2 785 | 17 603 | |||||
André Wyss (until March 31, 2018) 6 | 0 | 1 400 | 525 | 3 915 | |||||
| Subtotal | 11 830 | 19 574 | 7 810 | 40 383 | |||||
| Total | 87 464 | 196 022 | 86 669 | 97 888 | |||||
1 The values of the awards are reported in the table "2018 compensation at grant value for the CEO and other Executive Committee members" in “—2018 compensation at grant value for the CEO and Executive Committee members." | |||||||||
2 Vested shares, restricted shares and/or RSUs granted under the Annual Incentive for performance period 2018 | |||||||||
3 Target number of PSUs granted under the LTPP and LTRPP as applicable for the performance cycle 2018-2020 | |||||||||
4 Elizabeth Barrett stepped down from the role of CEO, Novartis Oncology and from the Executive Committee as at the end of the 2018 business year. The LTPP and LTRPP grants (16 284 and 6 107 PSUs, respectively) for the 2018-2020 performance cycle, and the 2018 buyout award of 21 267 performance shares, reflected in other compensation, both included in the table above, were forfeited in full upon her departure on December 31, 2018. | |||||||||
5 Joseph Jimenez received his 2018 Annual Incentive 100% in cash and was not granted LTPP and LTRPP awards for the performance cycle 2018-2020. | |||||||||
6 André Wyss stepped down from the Executive Committee on March 31, 2018, and ended his notice period on September, 30 2018. He received his 2018 Annual Incentive 100% in cash on a pro-rata basis, and the LTPP and LTRPP grants for the 2018-2020 performance cycle, included in the table above, will vest at the end of the performance cycle on a pro-rata basis per his contractual agreement and subject to the plan rules. | |||||||||
| Function | Ownership level | ||
| CEO | 5 x base compensation | ||
| Other Executive Committee members | 3 x base compensation | ||
Vested shares and ADRs 1 | Unvested shares and other equity rights 2 | Equity ownership level as a multiple of annual base salary 3 | Unvested target PSUs (e.g., LTPP/LTRPP) 4 | Matching shares under the LSSP 5 | Total at December 31, 2019 | ||||||||
| Vasant Narasimhan | 59 983 | 89 381 | 8x | 115 896 | 4 657 | 269 917 | |||||||
| Steven Baert | 39 785 | 31 890 | 8x | 64 538 | 0 | 136 213 | |||||||
| Bertrand Bodson | 4 600 | 11 492 | 2x | 15 037 | 0 | 31 129 | |||||||
| James Bradner | 21 794 | 46 531 | 5x | 104 379 | 0 | 172 704 | |||||||
| Harry Kirsch | 108 193 | 40 968 | 12x | 102 484 | 0 | 251 645 | |||||||
| Shannon Thyme Klinger | 12 193 | 22 028 | 3x | 35 117 | 1 488 | 70 826 | |||||||
| Steffen Lang | 56 063 | 20 248 | 9x | 26 782 | 4 535 | 107 628 | |||||||
| Klaus Moosmayer | 0 | 3 016 | 0x | 12 034 | 0 | 15 050 | |||||||
| Richard Saynor (from July 15, 2019) | 0 | 9 211 | 1x | 1 790 | 0 | 11 001 | |||||||
| Susanne Schaffert | 43 770 | 26 123 | 7x | 36 224 | 1 735 | 107 852 | |||||||
| John Tsai | 11 859 | 29 570 | 4x | 12 487 | 0 | 53 916 | |||||||
| Marie-France Tschudin (from June 7, 2019) | 5 500 | 24 715 | 3x | 45 078 | 0 | 75 293 | |||||||
| Robert Weltevreden | 150 | 7 751 | 1x | 11 386 | 0 | 19 287 | |||||||
Total 6 | 363 890 | 362 924 | 583 232 | 12 415 | 1 322 461 | ||||||||
1 Includes holdings of "persons closely linked" to Executive Committee members (see definition “—Persons closely linked.") | |||||||||||||
2 Includes unvested shares and ADRs as well as other equity rights applicable for the determination of equity amounts for the share ownership requirements, as per the definition above. Also includes unvested keep-whole shares received in connection to the Alcon spin-off. | |||||||||||||
3 The multiple is calculated based on the full-year annual base salary and the closing share price as at the end of the 2019 financial year. The share price on the final trading day of 2019 was CHF 91.90 / USD 94.69 as at December 31, 2019. | |||||||||||||
4 The target number of PSUs is disclosed pro-rata to December 31, 2019, unless the award qualified for full vesting under the relevant plan rules. | |||||||||||||
5 Matching shares under the Leveraged Share Savings Plan (LSSP) are disclosed pro-rata to December 31, 2019, unless the award qualified for full vesting under the plan rules. LSSP participation for Executive Committee members ceased in 2014 although some current members received later grants under this plan prior to becoming members of the Executive Committee. Outstanding awards will vest five years from the grant date, subject to the LSSP plan rules. | |||||||||||||
6 Paul Hudson and Richard Francis stepped down from the Executive Committee in 2019. At the time they stepped down from the Executive Committee, Mr. Hudson owned zero vested shares, and 140'121 unvested shares and other equity rights and Mr. Francis owned 50'615 vested shares and 86'740 unvested shares and other equity rights. | |||||||||||||
Annual base salary 1 | Variable compensation 2 | ||||
| Vasant Narasimhan | 14.7% | 85.3% | |||
| Steven Baert | 20.5% | 79.5% | |||
| Bertrand Bodson | 25.3% | 74.7% | |||
| James Bradner | 18.9% | 81.1% | |||
| Harry Kirsch | 17.8% | 82.2% | |||
| Shannon Thyme Klinger | 22.8% | 77.2% | |||
| Steffen Lang | 25.0% | 75.0% | |||
| Klaus Moosmayer | 25.1% | 74.9% | |||
| Richard Saynor | 13.4% | 86.6% | |||
| Susanne Schaffert | 19.3% | 80.7% | |||
| John Tsai | 19.1% | 80.9% | |||
| Marie-France Tschudin | 23.7% | 76.3% | |||
| Robert Weltevreden | 27.4% | 72.6% | |||
Total 3 | 19.4% | 80.6% | |||
1 Excludes pension and other benefits | |||||
2 See table “2019 compensation at grant value for the CEO and other Executive Committee members" with regard to the disclosure principles of variable compensation. | |||||
3 Excludes Richard Francis, who stepped down from the Executive Committee during 2019 and Paul Hudson, who resigned. | |||||
| Performance measures | Tracking | ||
| LTPP NCVA (75%) | Ahead of target | ||
| LTPP innovation (25%) | Ahead of target | ||
| LTRPP relative TSR (100%) | Ahead of peer group median | ||
2020 Executive Committee compensation system
2020 Executive Committee compensation
2019 Board compensation
CHF 000s | AGM 2019-2020 annual fee | ||
| Chairman of the Board | 3 800 | ||
| Board membership | 280 | ||
| Vice Chairman | 50 | ||
| Chair of the Audit and Compliance Committee | 130 | ||
| Chair of the Compensation Committee | 90 | ||
| Chair of the following committees: • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee | 70 | ||
| Membership of the Audit and Compliance Committee | 70 | ||
| Membership of the following committees: • Compensation Committee • Governance, Nomination and Corporate Responsibilities Committee • Research & Development Committee • Risk Committee | 40 | ||
| Board membership | Audit and Compliance Committee | Compensation Committee | Governance, Nomination and Corporate Responsibilities Committee | Research & Development Committee | Risk Committee | Shares (number) 1 | Cash (CHF) (A) | Shares (CHF) (B) | Other (CHF) (C) 2 | Total (CHF) (A)+(B)+(C) 3 | |||||||||||||
| Board members active on December 31, 2019 | |||||||||||||||||||||||
Joerg Reinhardt 4 | Chair | Chair | 21 498 | 1 900 000 | 1 900 000 | 4 373 | 3 804 373 | ||||||||||||||||
| Enrico Vanni | Vice Chair | • | Chair | • | 4 494 | 220 833 | 309 166 | 3 512 | 533 511 | ||||||||||||||
| Nancy Andrews | • | • | • | 2 035 | 180 000 | 180 000 | – | 360 000 | |||||||||||||||
| Ton Buechner | • | • | 2 967 | 145 833 | 204 166 | 4 373 | 354 372 | ||||||||||||||||
Patrice Bula 5 | • | • | 1 813 | – | 266 667 | 4 373 | 271 040 | ||||||||||||||||
| Srikant Datar | • | • | • | Chair | 2 602 | 230 000 | 230 000 | – | 460 000 | ||||||||||||||
| Elizabeth Doherty | • | Chair | • | 2 544 | 225 000 | 225 000 | – | 450 000 | |||||||||||||||
| Ann Fudge | • | • | • | • | 2 262 | 200 000 | 200 000 | – | 400 000 | ||||||||||||||
| Frans van Houten | • | • | 2 716 | 26 667 | 293 334 | – | 320 001 | ||||||||||||||||
| Andreas von Planta | • | • | Chair | • | 2 602 | 230 000 | 230 000 | 4 373 | 464 373 | ||||||||||||||
| Charles L. Sawyers | • | • | • | 2 035 | 180 000 | 180 000 | – | 360 000 | |||||||||||||||
| William T. Winters | • | • | • 5 | 3 620 | – | 353 333 | – | 353 333 | |||||||||||||||
| Subtotal | 51 188 | 3 538 333 | 4 571 666 | 21 002 | 8 131 001 | ||||||||||||||||||
| Board members who stepped down at the 2019 AGM | |||||||||||||||||||||||
Dimitri Azar 6 | • | • | • | 1 016 | 30 000 | 30 000 | – | 60 000 | |||||||||||||||
| Subtotal | 1 016 | 30 000 | 30 000 | – | 60 000 | ||||||||||||||||||
| Total | 52 204 | 3 568 333 | 4 601 666 | 21 002 | 8 191 001 | ||||||||||||||||||
| See next page for 2018 comparative figures. | |||||||||||||||||||||||
1 The shown amounts represent the gross number of shares delivered to each Board member in 2019 for the respective Board member’s service period. The number of shares reported in this column represent: (i) the second and final equity installment delivered in February 2019 for the services from the 2018 AGM to the 2019 AGM, and (ii) the first of two equity installments delivered in August 2019 for the services from the 2019 AGM to the 2020 AGM. The second and final equity installment for the services from the 2019 AGM to the 2020 AGM will take place in February 2020. | |||||||||||||||||||||||
2 Includes an amount of CHF 21'002 for mandatory employer contributions for all Board members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 413'985 , and provides a right to the maximum future insured government pension benefit for the Board member. | |||||||||||||||||||||||
3 All amounts are before deduction of the social security contribution and income tax due by the Board member. | |||||||||||||||||||||||
4 No additional committee fees for chairing the Research & Development Committee were delivered to Joerg Reinhardt. | |||||||||||||||||||||||
5 From February 28, 2019 | |||||||||||||||||||||||
6 Until February 28, 2019 | |||||||||||||||||||||||
| Board membership | Audit and Compliance Committee | Compensation Committee | Governance, Nomination and Corporate Responsibilities Committee | Research & Development Committee | Risk Committee | Shares (number) 1 | Cash (CHF) (A) | Shares (CHF) (B) | Other (CHF) (C) 2 | Total (CHF) (A)+(B)+(C) 3 | |||||||||||||
| Board members active on December 31, 2018 | |||||||||||||||||||||||
Joerg Reinhardt 4 | Chair | Chair | 23 889 | 1 900 000 | 1 900 000 | 4 336 | 3 804 336 | ||||||||||||||||
| Enrico Vanni | Vice Chair | • | Chair | • | 4 854 | 41 667 | 483 334 | 3 475 | 528 476 | ||||||||||||||
| Nancy Andrews | • | • | • | 2 262 | 180 000 | 180 000 | – | 360 000 | |||||||||||||||
| Dimitri Azar | • | • 5 | • | 2 359 | 182 500 | 182 500 | – | 365 000 | |||||||||||||||
| Ton Buechner | • | • 5 | • 6 | 4 270 | – | 346 667 | 4 336 | 351 003 | |||||||||||||||
| Srikant Datar | • | • | • | Chair | 2 859 | 229 167 | 229 167 | – | 458 334 | ||||||||||||||
| Elizabeth Doherty | • | Chair | • | 2 828 | 225 000 | 225 000 | – | 450 000 | |||||||||||||||
| Ann Fudge | • | • | • | • | 2 481 | 199 167 | 199 167 | – | 398 334 | ||||||||||||||
| Frans van Houten | • | • 5 | 2 334 | 148 333 | 168 333 | – | 316 666 | ||||||||||||||||
| Andreas von Planta | • | • | Chair | • | 2 859 | 229 167 | 229 167 | 4 336 | 462 670 | ||||||||||||||
| Charles L. Sawyers | • | • | • | 2 262 | 180 000 | 180 000 | – | 360 000 | |||||||||||||||
| William T. Winters | • | • | 4 087 | – | 321 667 | – | 321 667 | ||||||||||||||||
| Subtotal | 57 344 | 3 515 001 | 4 645 002 | 16 483 | 8 176 486 | ||||||||||||||||||
| Board members who stepped down at the 2018 AGM | |||||||||||||||||||||||
Pierre Landolt (until March 2, 2018) 7 | • | • 6 | 2 131 | – | 55 000 | 3 475 | 58 475 | ||||||||||||||||
| Subtotal | 2 131 | – | 55 000 | 3 475 | 58 475 | ||||||||||||||||||
| Total | 59 475 | 3 515 001 | 4 700 002 | 19 958 | 8 234 961 | ||||||||||||||||||
1 The shown amounts represent the gross number of shares delivered to each Board member in 2018 for the respective Board member’s service period. The number of shares reported in this column represent: (i) the second and final equity installment delivered in February 2018 for the services from the 2017 AGM to the 2018 AGM, and (ii) the first of two equity installments delivered in August 2018 for the services from the 2018 AGM to the 2019 AGM. The second and final equity installment for the services from the 2018 AGM to the 2019 AGM will take place in February 2019. | |||||||||||||||||||||||
2 Includes an amount of CHF 19 958 for mandatory employer contributions for all Board members paid by Novartis to governmental social security systems. This amount is out of total employer contributions of CHF 383 864, and provides a right to the maximum future insured government pension benefit for the Board member. | |||||||||||||||||||||||
3 All amounts are before deduction of the social security contribution and income tax due by the Board member. | |||||||||||||||||||||||
4 No additional committee fees for chairing the Research & Development Committee were delivered to Joerg Reinhardt. | |||||||||||||||||||||||
5 From March 2, 2018 | |||||||||||||||||||||||
6 Until March 2, 2018 | |||||||||||||||||||||||
7 According to Pierre Landolt, the Sandoz Family Foundation is the economic beneficiary of the compensation. | |||||||||||||||||||||||
Number of shares at December 31, 2019 1,2 | |||
| Joerg Reinhardt | 563 697 | ||
| Enrico Vanni | 26 645 | ||
| Nancy Andrews | 7 265 | ||
| Ton Buechner | 10 950 | ||
| Patrice Bula | 1 946 | ||
| Srikant Datar | 41 334 | ||
| Elizabeth Doherty | 6 765 | ||
| Ann Fudge | 14 114 | ||
| Frans van Houten | 4 764 | ||
| Andreas von Planta | 161 035 | ||
| Charles L. Sawyers | 10 986 | ||
| William T. Winters | 18 170 | ||
Total 3 | 867 671 | ||
1 Includes holdings of "persons closely linked" to Board members (see definition “—Persons closely linked") | |||
2 Each share provides entitlement to one vote. | |||
3 Dimitri Azar stepped down from the Board of Directors on February 28, 2019. On February 28, 2019, Mr. Azar owned 18 750 shares. His shares are not included in the total. | |||
Compensation governance

| Decision on | Decision-making authority | ||
| Compensation of Chairman and other Board members | Board of Directors | ||
| Compensation of CEO | Board of Directors | ||
| Compensation of other Executive Committee members | Compensation Committee | ||
6.C Board practices


| % holding of share capital Dec 31, 2019 | |||
| Shareholders registered for their own account: | |||
| Emasan AG, Basel | 3.5 | ||
| Novartis Foundation for Employee Participation, Basel | 2.1 | ||
| UBS Fund Management (Switzerland) AG, Basel | 2.1 | ||
| % holding of share capital Dec 31, 2019 | |||
| Shareholders registered as nominees: | |||
| Chase Nominees Ltd., London | 10.4 | ||
| The Bank of New York Mellon, New York | 3.8 | ||
| Through The Bank of New York Mellon, Everett | 2.0 | ||
| Through The Bank of New York Mellon, New York | 1.2 | ||
| Through The Bank of New York Mellon, SA/NV, Brussels | 0.6 | ||
| Nortrust Nominees Ltd., London | 3.9 | ||
| Shareholder acting as American Depositary Share (ADS) depositary: | |||
| JPMorgan Chase Bank, N.A., New York | 12.5 | ||
As of December 31, 2019 1 | Number of registered shareholders | % of registered share capital | |||
| 1–100 | 25 442 | 0.06 | |||
| 101–1 000 | 97 161 | 1.59 | |||
| 1 001–10 000 | 34 884 | 3.84 | |||
| 10 001–100 000 | 3 080 | 3.16 | |||
| 100 001–1 000 000 | 451 | 5.45 | |||
| 1 000 001–5 000 000 | 64 | 4.96 | |||
5 000 001 or more 2 | 29 | 50.66 | |||
| Total registered shareholders/shares | 161 111 | 69.72 | |||
| Unregistered shares | 30.28 | ||||
| Total | 100.00 | ||||
1 At the record date of the Annual General Meeting (AGM) 2019, unregistered shares amounted to 15% | |||||
2 Including significant registered shareholders as listed above | |||||
| As of December 31, 2019 | Shareholders in % | Shares in % | |||
| Individual shareholders | 96.43 | 12.99 | |||
Legal entities 1 | 3.52 | 32.77 | |||
| Nominees, fiduciaries and ADS depositary | 0.05 | 54.24 | |||
| Total | 100.00 | 100.00 | |||
1 Excluding 5.7% of the share capital held as treasury shares by Novartis AG or its fully owned subsidiaries | |||||
| As of December 31, 2019 | Shareholders in % | Shares in % | |||
| Belgium | 0.13 | 1.13 | |||
| France | 2.06 | 0.29 | |||
| Germany | 5.66 | 1.64 | |||
| Japan | 0.21 | 0.70 | |||
| Luxembourg | 0.05 | 0.42 | |||
Switzerland 2 | 87.68 | 42.38 | |||
| United Kingdom | 0.56 | 26.15 | |||
| United States | 0.32 | 25.41 | |||
| Other countries | 3.33 | 1.88 | |||
| Total | 100.00 | 100.00 | |||
1 Registered shares held by nominees are shown in the country where the company/affiliate entered in the Share Register as shareholder has its registered seat. | |||||
2 Excluding 5.7% of the share capital held as treasury shares by Novartis AG or its fully owned subsidiaries | |||||
AGM | Shareholder decision | Shares cancelled | Average repurchase share price (CHF) 1 | ||||
| 2017 | • Capital reduction by CHF 5.1 million (from CHF 1 313 557 410 to CHF 1 308 422 410) | 10 270 000 | 74.67 | ||||
| 2018 | • Capital reduction by CHF 33.11 million (from CHF 1 308 422 410 to CHF 1 275 312 410) | 66 220 000 | 78.34 | ||||
| 2019 | • Capital reduction by CHF 11.63 million (from CHF 1 275 312 410 to CHF 1 263 687 410) • Authorization of the Board to repurchase shares up to a maximum of CHF 10 billion until the AGM 2022 under an eighth share repurchase program | 23 250 000 | 79.08 | ||||
AGM | Proposal to the shareholders | Shares to be cancelled | Average repurchase share price (CHF) 1 | ||||
| 2020 | • Capital reduction by CHF 30.16 million (from CHF 1 263 687 410 to CHF 1 233 530 460) | 60 313 900 | 88.18 | ||||
1 All shares were repurchased on the SIX Swiss Exchange second trading line. | |||||||
| 2019 | 2018 | 2017 | |||||
| Issued shares | 2 527 374 820 | 2 550 624 820 | 2 616 844 820 | ||||
Treasury shares 1 | 262 366 332 | 239 453 391 | 299 388 321 | ||||
| Outstanding shares at December 31 | 2 265 008 488 | 2 311 171 429 | 2 317 456 499 | ||||
| Weighted average number of shares outstanding | 2 290 792 782 | 2 319 322 369 | 2 345 783 843 | ||||
1 Approximately 118 million treasury shares (2018: 122 million; 2017: 131 million) are held in Novartis entities that restrict their availability for use. | |||||||
| 2019 | 2018 | 2017 | |||||
| Basic earnings per share from continuing operations (USD) | 3.12 | 5.52 | 3.20 | ||||
| Basic earnings per share from discontinued operations (USD) | 2.00 | – 0.08 | 0.08 | ||||
| Total basic earnings per share (USD) | 5.12 | 5.44 | 3.28 | ||||
| Diluted earnings per share from continuing operations (USD) | 3.08 | 5.46 | 3.17 | ||||
| Diluted earnings per share from discontinued operations (USD) | 1.98 | – 0.08 | 0.08 | ||||
| Total diluted earnings per share (USD) | 5.06 | 5.38 | 3.25 | ||||
| Net cash flow from operating activities of continuing operations (USD) | 5.91 | 5.63 | 4.87 | ||||
| Year-end equity for Novartis AG shareholders (USD) | 24.49 | 34.01 | 32.00 | ||||
Dividend (CHF) 2 | 2.95 | 2.85 | 2.80 | ||||
1 Calculated on the weighted average number of shares outstanding, except year-end equity | |||||||
2 2019: proposal to shareholders for approval at the AGM on February 28, 2020 | |||||||
| 2019 | 2018 | 2017 | |||||
Price/earnings ratio 1 | 18.5 | 15.7 | 25.7 | ||||
Price/earnings ratio from continuing operations 1 | 30.4 | 15.4 | 25.7 | ||||
Dividend yield (%) 1 | 3.2 | 3.4 | 3.4 | ||||
1 Based on the Novartis share price at December 31 of each year | |||||||
2019 1 | 2018 | 2017 | |||||
| Year-end ADR price (USD) | 94.69 | 85.81 | 83.96 | ||||
High 2 | 96.14 | 93.91 | 86.65 | ||||
Low 2 | 75.40 | 72.44 | 70.03 | ||||
Number of ADRs outstanding 3 | 315 073 094 | 338 641 387 | 320 833 039 | ||||
1 2019 excludes the business of Alcon, which was spun off in April 2019 into a separately traded standalone company. | |||||||
2 Based on the daily closing prices | |||||||
3 The depositary, JPMorgan Chase Bank, N.A., holds one Novartis AG share for every ADR issued. | |||||||
2019 1 | 2018 | 2017 | |||||
| Year-end share price | 91.90 | 84.04 | 82.40 | ||||
High 2 | 96.04 | 91.84 | 85.15 | ||||
Low 2 | 77.03 | 72.42 | 69.55 | ||||
Year-end market capitalization (USD billions) 3 | 214.8 | 197.0 | 195.5 | ||||
Year-end market capitalization (CHF billions) 3 | 208.2 | 194.2 | 191.0 | ||||
1 2019 excludes the business of Alcon, which was spun off in April 2019 into a separately traded standalone company. | |||||||
2 Based on the daily closing prices | |||||||
3 Market capitalization is calculated based on the number of shares outstanding (excluding treasury shares). Market capitalization in USD is based on the market capitalization in CHF converted at the year-end CHF/USD exchange rate. | |||||||
Shareholder participation

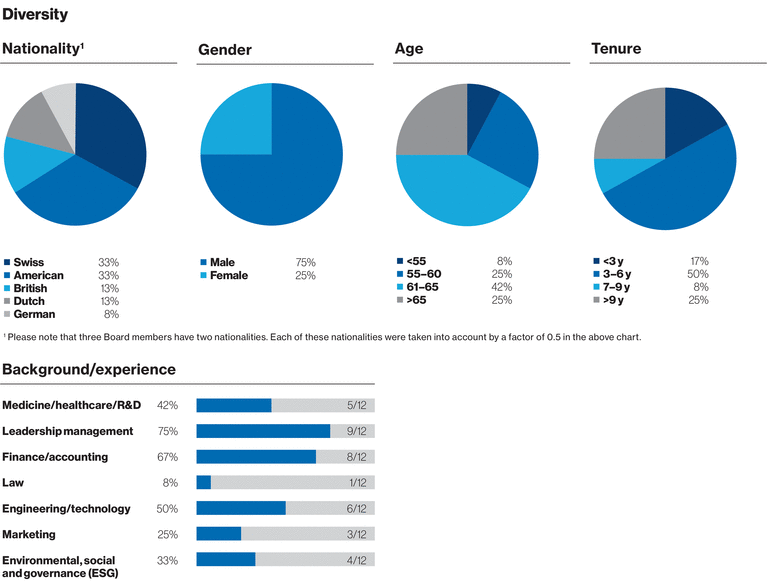
Members of the Board of Directors

 Medicine/healthcare/R&D
Medicine/healthcare/R&D  Leadership management
Leadership management
 Medicine/healthcare/R&D
Medicine/healthcare/R&D  Finance/accounting
Finance/accounting  Engineering/technology
Engineering/technology
 Medicine/healthcare/R&D
Medicine/healthcare/R&D  Leadership management
Leadership management
 Leadership management
Leadership management  Finance/accounting
Finance/accounting  Environmental, social and governance (ESG)
Environmental, social and governance (ESG)  Engineering/technology
Engineering/technology
 Leadership management
Leadership management  Marketing
Marketing  Finance/accounting
Finance/accounting  Engineering/technology
Engineering/technology
Audit Committee Financial Expert
 Finance/accounting
Finance/accounting
Audit Committee Financial Expert
 Leadership management
Leadership management  Marketing
Marketing  Finance/accounting
Finance/accounting  Engineering/technology
Engineering/technology
 Leadership management
Leadership management  Marketing
Marketing  Environmental, social and governance (ESG)
Environmental, social and governance (ESG)
 Medicine/healthcare/R&D
Medicine/healthcare/R&D  Leadership management
Leadership management  Finance/accounting
Finance/accounting  Engineering/technology
Engineering/technology
 Finance/accounting
Finance/accounting  Law
Law  Environmental, social and governance (ESG)
Environmental, social and governance (ESG)
 Medicine/healthcare/R&D
Medicine/healthcare/R&D  Leadership management
Leadership management  Environmental, social and governance (ESG)
Environmental, social and governance (ESG)
 Leadership management
Leadership management  Finance/accounting
Finance/accounting  Engineering/technology
Engineering/technology






| Maximum number of mandates | |||
| Mandates | 10 | ||
Other listed companies 1 | 4 | ||
1 Chairmanship of the board of directors in other listed companies counts as two mandates. | |||
| Maximum number of mandates | |||
| Mandates in companies that are controlled by Novartis AG | No limit | ||
| Mandates held at the request of Novartis AG or companies controlled by it | 5 | ||
| Mandates in associations, charitable organizations, foundations, trusts and employee welfare foundations | 10 | ||


Executive Committee

| Maximum number of mandates | |||
| Mandates | 6 | ||
Other listed companies 1 | 2 | ||
1 Chairmanship of the board of directors in other listed companies is not allowed. | |||
| Maximum number of mandates | |||
| Mandates in companies that are controlled by Novartis AG | No limit | ||
| Mandates held at the request of Novartis AG or companies controlled by it | 5 | ||
| Mandates in associations, charitable organizations, foundations, trusts and employee welfare foundations | 10 | ||
Members of the Executive Committee




Year of birth: 1972







Year of birth: 1967


Auditors
| 2019 USD million | 2018 USD million | ||||
| Audit services | 21.2 | 25.6 | |||
| Audit-related services | 1.0 | 13.4 | |||
| Tax services | 0.7 | 0.7 | |||
| Other services | 1.4 | 2.4 | |||
| Total | 24.3 | 42.1 | |||
Information policy
| Topic | Information | ||
| Share capital | Articles of Incorporation of Novartis AG www.novartis.com/investors/company-overview/corporate-governance Novartis key share data www.novartis.com/key-share-data | ||
| Shareholder rights | Articles of Incorporation of Novartis AG www.novartis.com/investors/company-overview/corporate-governance | ||
| Annual General Meeting of Shareholders | Annual General Meeting of Shareholders www.novartis.com/investors/shareholder-information/annual-general-meeting | ||
| Board Regulations | Board Regulations www.novartis.com/investors/company-overview/corporate-governance | ||
| Novartis code for senior financial officers | Novartis Code of Ethical Conduct for CEO and Senior Financial Officers www.novartis.com/investors/company-overview/corporate-governance | ||
| Novartis in Society ESG Report | Novartis in Society ESG Report www.novartis.com/nisreport2019 | ||
| Novartis financial data | Novartis financial data www.novartis.com/investors/financial-data | ||
| Press releases | Press releases www.novartis.com/news/news-archive?type=press_release Free email service www.novartis.com/news/stay-up-to-date | ||
| Additional information (including Novartis investors event calendar, registered office, contact and email addresses, phone numbers, etc.) | Novartis Investor Relations www.novartis.com/investors | ||
6.D Employees
| For the year ended December 31, 2019 (full-time equivalents) | Marketing and sales | Production and supply | Research and development | NBS 1 | General and administration | Total | |||||||
| USA | 5 360 | 2 830 | 5 412 | 614 | 763 | 14 979 | |||||||
| Canada and Latin America | 3 396 | 838 | 480 | 864 | 397 | 5 975 | |||||||
| Europe | 16 395 | 19 386 | 9 988 | 4 352 | 2 666 | 52 787 | |||||||
| Asia/Africa/Australasia | 17 455 | 3 163 | 4 296 | 4 233 | 1 026 | 30 173 | |||||||
| Total | 42 606 | 26 217 | 20 176 | 10 063 | 4 852 | 103 914 | |||||||
| For the year ended December 31, 2018 (full-time equivalents) | Marketing and sales | Production and supply | Research and development | NBS 1 | General and administration | Total | |||||||
| USA | 6 825 | 7 524 | 6 700 | 1 467 | 911 | 23 427 | |||||||
| Canada and Latin America | 4 584 | 960 | 508 | 899 | 490 | 7 441 | |||||||
| Europe | 19 608 | 21 397 | 10 049 | 4 845 | 2 780 | 58 679 | |||||||
| Asia/Africa/Australasia | 20 099 | 6 636 | 3 977 | 3 613 | 1 289 | 35 614 | |||||||
| Total | 51 116 | 36 517 | 21 234 | 10 824 | 5 470 | 125 161 | |||||||
Thereof continuing operations2 | 43 954 | 25 862 | 19 803 | 10 824 | 4 337 | 104 780 | |||||||
Thereof discontinued operations2 | 7 162 | 10 655 | 1 431 | 0 | 1 133 | 20 381 | |||||||
| For the year ended December 31, 2017 (full-time equivalents) | Marketing and sales | Production and supply | Research and development | NBS 1 | General and administration | Total | |||||||
| USA | 6 563 | 7 095 | 6 803 | 1 680 | 726 | 22 867 | |||||||
| Canada and Latin America | 4 477 | 1 305 | 557 | 900 | 471 | 7 710 | |||||||
| Europe | 18 665 | 20 412 | 10 173 | 4 903 | 2 469 | 56 622 | |||||||
| Asia/Africa/Australasia | 19 005 | 6 970 | 3 883 | 3 386 | 1 154 | 34 398 | |||||||
| Total | 48 710 | 35 782 | 21 416 | 10 869 | 4 820 | 121 597 | |||||||
Thereof continuing operations2 | 42 115 | 25 564 | 20 060 | 10 869 | 3 859 | 102 467 | |||||||
Thereof discontinued operations2 | 6 595 | 10 218 | 1 356 | 0 | 961 | 19 130 | |||||||
1 NBS relates to full-time equivalent employees from our Novartis Business Services organizational unit. | |||||||||||||
2 Continuing operations include the businesses of the Innovative Medicines and Sandoz Divisions and the continuing Corporate activities, and discontinued operations include the Alcon eye care devices business and certain corporate activities attributable to Alcon prior to the spin-off. See “Item 18.Financial Statements—Note 2. Significant transactions—Significant transactions in 2019.” | |||||||||||||
6.E Share ownership
Item 7. Major Shareholders and Related Party Transactions
7.A Major shareholders
| % of respective share capital beneficially owned as of: | |||||||||
| Ordinary shares beneficially owned as of Dec 31, 2019 | Dec 31, 2019 | Dec 31, 2018 | Dec 31, 2017 | ||||||
| Shareholders registered for their own account: | |||||||||
| Emasan AG, Basel, Switzerland | 89 193 765 | 3.5 | 3.5 | 3.4 | |||||
Novartis Foundation for Employee Participation, Basel, Switzerland 1 | 53 983 628 | 2.1 | 2.3 | 2.5 | |||||
| UBS Fund Management (Switzerland) AG, Basel, Switzerland | 52 845 411 | 2.1 | 2.2 | 2.0 | |||||
1 The Novartis Foundation for Employee Participation (the "Employee Foundation") is a special purpose entity that was founded by, but is independent from, Novartis. | |||||||||
| % of respective share capital held as of: | |||||||||
| Ordinary shares held as of Dec 31, 2019 | Dec 31, 2019 | Dec 31, 2018 | Dec 31, 2017 | ||||||
| Shareholders registered as nominees: | |||||||||
| Chase Nominees Ltd., London, England | 264 073 363 | 10.4 | 9.8 | 7.8 | |||||
| The Bank of New York Mellon, New York, NY | 95 456 296 | 3.8 | 4.1 | 4.3 | |||||
| Through The Bank of New York Mellon, Everett, MA | 50 207 591 | 2.0 | 2.1 | 2.0 | |||||
| Through The Bank of New York Mellon, New York, NY | 29 356 938 | 1.2 | 1.3 | – | |||||
| Through The Bank of New York Mellon, SA/NV, Brussels, Belgium | 15 891 767 | 0.6 | 0.7 | 2.3 | |||||
| Nortrust Nominees Ltd., London, England | 98 354 215 | 3.9 | 3.6 | 3.8 | |||||
| Shareholder acting as American Depositary Share (ADS) depositary: | |||||||||
| JPMorgan Chase Bank, N.A., New York, NY | 314 717 099 | 12.5 | 13.3 | 12.3 | |||||
7.B Related party transactions
7.C Interests of experts and counsel
Item 8. Financial Information
8.A Consolidated statements and other financial information
8.B Significant changes
Item 9. The Offer and Listing
9.A Offer and listing details
9.B Plan of distribution
9.C Markets
9.D Selling shareholders
9.E Dilution
9.F Expenses of the issue
Item 10. Additional Information
10.A Share capital
10.B Memorandum and articles of association
10.C Material contracts
10.D Exchange controls
10.E Taxation
10.F Dividends and paying agents
10.G Statement by experts
10.H Documents on display
10.I Subsidiary information
Item 11. Quantitative and Qualitative Disclosures About Market Risk
Item 12. Description of Securities Other Than Equity Securities
12.A Debt securities
12.B Warrants and rights
12.C Other securities
12.D American Depositary Shares
| Category | Depositary actions | Associated fee | |||
| Depositing or substituting underlying shares | Acceptance of shares surrendered, and issuance of ADRs in exchange, including surrenders and issuances in respect of: — Share distributions — Stock split — Rights — Merger — Exchange of shares or any other transaction or event or other distribution affecting the ADSs or the deposited shares | USD 5.00 for each 100 ADSs (or portion thereof) evidenced by the new ADRs delivered | |||
| Withdrawing underlying shares | Acceptance of ADRs surrendered for withdrawal of deposited shares | USD 5.00 for each 100 ADSs (or portion thereof) evidenced by the ADRs surrendered | |||
| Selling or exercising rights | Distribution or sale of shares, the fee being in an amount equal to the fee for the execution and delivery of ADRs that would have been charged as a result of the deposit of such shares | USD 5.00 for each 100 ADSs (or portion thereof) | |||
| Transferring, splitting or grouping receipts | Transfers, combining or grouping of depositary receipts | USD 1.50 per ADR | |||
| Expenses of the depositary | Expenses incurred on behalf of holders in connection with: — Compliance with foreign exchange control regulations or any law or regulation relating to foreign investment — The depositary’s or its custodian’s compliance with applicable law, rule or regulation — Stock transfer or other taxes and other governmental charges — Cable, telex and facsimile transmission and delivery — Expenses of the depositary in connection with the conversion of foreign currency into US dollars (which are paid out of such foreign currency) — Any other charge payable by any of the depositary or its agents | Expenses payable at the sole discretion of the depositary by billing holders or by deducting charges from one or more cash dividends or other cash distributions | |||
| Advance tax relief | Tax relief/reclamation process for qualified holders | A depositary service charge of USD 0.008 per ADS | |||
PART II
Item 13. Defaults, Dividend Arrearages and Delinquencies
Item 14. Material Modifications to the Rights of Security Holders and Use of Proceeds
Item 15. Controls and Procedures
Item 16A. Audit Committee Financial Expert
Item 16B. Code of Ethics
Item 16C. Principal Accountant Fees and Services
Item 16D. Exemptions from the Listing Standards for Audit Committees
Item 16E. Purchases of Equity Securities by the Issuer and Affiliated Purchasers
2019 | Total Number of Shares Purchased (a) 1 | Average price paid per share in USD (b) | Total number of shares purchased as part of publicly announced plans or programs (c) 2 | Maximum approximate value of shares that may yet be purchased under the plans or programs (CHF millions) (d) | Maximum approximate value of shares that may yet be purchased under the plans or programs (USD millions) (e) 3 | ||||||
| Jan. 1-31 | 1 913 205 | 87.38 | 830 000 | 2 137 | 2 152 | ||||||
| Feb. 1-28 | 203 399 | 88.31 | 0 | 10 000 | 10 027 | ||||||
| Mar. 1-31 | 81 551 | 92.09 | 0 | 10 000 | 10 042 | ||||||
| Apr. 1-30 | 3 687 215 | 80.82 | 3 452 000 | 9 716 | 9 539 | ||||||
| May 1-31 | 15 719 319 | 83.55 | 15 695 000 | 8 392 | 8 338 | ||||||
| Jun. 1-30 | 12 852 939 | 90.27 | 12 840 000 | 7 247 | 7 433 | ||||||
| Jul. 1-31 | 23 550 117 | 92.10 | 23 538 500 | 5 106 | 5 156 | ||||||
| Aug. 1-31 | 3 966 974 | 91.76 | 3 958 400 | 4 752 | 4 809 | ||||||
| Sep. 1-30 | 36 197 | 89.69 | 0 | 4 752 | 4 790 | ||||||
| Oct. 1-31 | 2 031 | 84.92 | 0 | 4 752 | 4 811 | ||||||
| Nov. 1-30 | 16 065 | 87.12 | 0 | 4 752 | 4 758 | ||||||
| Dec. 1-31 | 18 567 | 90.12 | 0 | 4 752 | 4 903 | ||||||
| Total | 62 047 579 | 88.70 | 60 313 900 | ||||||||
1 Column (a) shows shares repurchased on the SIX Swiss Exchange second trading line plus shares we purchased from employees who had obtained the shares through a Novartis Employee Ownership Plan. See "Item 18. Financial Statements - Note 26 Equity-based participation plans for associates." | |||||||||||
2 Column (c) shows shares repurchased on the SIX Swiss Exchange second trading line under the seventh CHF 10 billion share buyback authority approved at the 2016 Annual General Meeting (AGM) for transactions before February 28, 2019 and under the eighth CHF 10 billion share buyback authority approved at the 2019 AGM for transactions after such date. See "Item 6. Directors, Senior Management and Employees - Item 6C. Board Practices - Our capital structure - Changes in capital." | |||||||||||
3 Column (e) shows the Swiss franc amount from column (d) converted into US dollars as of the month-end, using the Swiss franc/US dollar exchange rate at the applicable month-end | |||||||||||
Item 16F. Change in Registrant’s Certifying Accountant
Item 16G. Corporate Governance
Item 16H. Mine Safety Disclosure
PART III
Item 17. Financial Statements
Item 18. Financial Statements
Item 19. Exhibits
SIGNATURES
Novartis Group consolidated financial statements
| (USD millions unless indicated otherwise) | Note | 2019 | 2018 | 2017 | |||||
| Net sales to third parties from continuing operations | 3 | 47 445 | 44 751 | 42 338 | |||||
| Sales to discontinued segment | 53 | 82 | 43 | ||||||
| Net sales from continuing operations | 47 498 | 44 833 | 42 381 | ||||||
| Other revenues | 3 | 1 179 | 1 266 | 1 023 | |||||
| Cost of goods sold | – 14 425 | – 14 510 | – 13 633 | ||||||
| Gross profit from continuing operations | 34 252 | 31 589 | 29 771 | ||||||
| Selling, general and administration | – 14 369 | – 13 717 | – 12 465 | ||||||
| Research and development | – 9 402 | – 8 489 | – 8 389 | ||||||
| Other income | 2 031 | 1 629 | 1 922 | ||||||
| Other expense | – 3 426 | – 2 609 | – 2 137 | ||||||
| Operating income from continuing operations | 9 086 | 8 403 | 8 702 | ||||||
| Income from associated companies | 4 | 659 | 6 438 | 1 108 | |||||
| Interest expense | 5 | – 850 | – 932 | – 750 | |||||
| Other financial income and expense | 5 | 45 | 186 | 42 | |||||
| Income before taxes from continuing operations | 8 940 | 14 095 | 9 102 | ||||||
| Taxes | 6 | – 1 793 | – 1 295 | – 1 603 | |||||
| Net income from continuing operations | 7 147 | 12 800 | 7 499 | ||||||
| Net (loss)/income from discontinued operations before gain on distribution of Alcon Inc. to Novartis AG shareholders | 30 | – 101 | – 186 | 204 | |||||
| Gain on distribution of Alcon Inc. to Novartis AG shareholders | 2 | 4 691 | |||||||
| Net income/(loss) from discontinued operations | 30 | 4 590 | – 186 | 204 | |||||
| Net income | 11 737 | 12 614 | 7 703 | ||||||
| Attributable to: | |||||||||
| Shareholders of Novartis AG | 11 732 | 12 611 | 7 703 | ||||||
| Non-controlling interests | 5 | 3 | 0 | ||||||
| Basic earnings per share (USD) from continuing operations | 3.12 | 5.52 | 3.20 | ||||||
| Basic earnings per share (USD) from discontinued operations | 2.00 | – 0.08 | 0.08 | ||||||
| Total basic earnings per share (USD) | 7 | 5.12 | 5.44 | 3.28 | |||||
| Diluted earnings per share (USD) from continuing operations | 3.08 | 5.46 | 3.17 | ||||||
| Diluted earnings per share (USD) from discontinued operations | 1.98 | – 0.08 | 0.08 | ||||||
| Total diluted earnings per share (USD) | 7 | 5.06 | 5.38 | 3.25 | |||||
| The accompanying Notes form an integral part of the consolidated financial statements. | |||||||||
| (USD millions) | Note | 2019 | 2018 | 2017 | |||||
| Net income | 11 737 | 12 614 | 7 703 | ||||||
| Other comprehensive income to be eventually recycled into the consolidated income statement: | |||||||||
| Fair value adjustments on marketable securities, net of taxes | 8.1 | 39 | |||||||
| Fair value adjustments on debt securities, net of taxes | 8.1 | 1 | – 1 | ||||||
| Fair value adjustments on deferred cash flow hedges, net of taxes | 8.1 | 1 | 12 | 12 | |||||
| Total fair value adjustments on financial instruments, net of taxes | 2 | 12 | 50 | ||||||
| Novartis share of other comprehensive income recognized by associated companies, net of taxes | 4 | – 94 | – 482 | – 37 | |||||
| Net investment hedge | 8 | 44 | 95 | – 237 | |||||
| Currency translation effects | 8.2 | 352 | 315 | 2 210 | |||||
| Total of items to eventually recycle | 304 | – 60 | 1 986 | ||||||
| Other comprehensive income never to be recycled into the consolidated income statement: | |||||||||
| Actuarial (losses)/gains from defined benefit plans, net of taxes | 8.3 | – 467 | – 359 | 851 | |||||
| Fair value adjustments on equity securities, net of taxes | 8.1 | – 47 | 13 | ||||||
| Total of items never to be recycled | – 514 | – 346 | 851 | ||||||
| Total comprehensive income | 11 527 | 12 208 | 10 540 | ||||||
| Attributable to: | |||||||||
| Shareholders of Novartis AG | 11 525 | 12 210 | 10 538 | ||||||
| Continuing operations | 6 948 | 12 417 | 10 211 | ||||||
| Discontinued operations | 4 577 | – 207 | 327 | ||||||
| Non-controlling interests | 2 | – 2 | 2 | ||||||
| The accompanying Notes form an integral part of the consolidated financial statements. | |||||||||
| (USD millions) | Note | 2019 | 2018 | ||||
| Assets | |||||||
| Non-current assets | |||||||
| Property, plant and equipment | 9 | 12 069 | 15 696 | ||||
| Right-of-use assets | 10 | 1 677 | |||||
| Goodwill | 11 | 26 524 | 35 294 | ||||
| Intangible assets other than goodwill | 11 | 28 787 | 38 719 | ||||
| Investments in associated companies | 4 | 8 644 | 8 352 | ||||
| Deferred tax assets | 12 | 7 909 | 8 699 | ||||
| Financial assets | 13 | 2 518 | 2 345 | ||||
| Other non-current assets | 13 | 738 | 895 | ||||
| Total non-current assets | 88 866 | 110 000 | |||||
| Current assets | |||||||
| Inventories | 14 | 5 982 | 6 956 | ||||
| Trade receivables | 15 | 8 301 | 8 727 | ||||
| Income tax receivables | 254 | 248 | |||||
| Marketable securities, commodities, time deposits and derivative financial instruments | 16 | 334 | 2 693 | ||||
| Cash and cash equivalents | 16 | 11 112 | 13 271 | ||||
| Other current assets | 17 | 2 680 | 2 861 | ||||
| Total current assets without disposal group | 28 663 | 34 756 | |||||
| Assets of disposal group held for sale | 2 | 841 | 807 | ||||
| Total current assets | 29 504 | 35 563 | |||||
| Total assets | 118 370 | 145 563 | |||||
| Equity and liabilities | |||||||
| Equity | |||||||
| Share capital | 18 | 936 | 944 | ||||
| Treasury shares | 18 | – 80 | – 69 | ||||
| Reserves | 54 618 | 77 739 | |||||
| Issued share capital and reserves attributable to Novartis AG shareholders | 55 474 | 78 614 | |||||
| Non-controlling interests | 77 | 78 | |||||
| Total equity | 55 551 | 78 692 | |||||
| Liabilities | |||||||
| Non-current liabilities | |||||||
| Financial debts | 19 | 20 353 | 22 470 | ||||
| Lease liabilities | 10 | 1 703 | |||||
| Deferred tax liabilities | 12 | 5 867 | 7 475 | ||||
| Provisions and other non-current liabilities | 20 | 6 632 | 7 319 | ||||
| Total non-current liabilities | 34 555 | 37 264 | |||||
| Current liabilities | |||||||
| Trade payables | 5 424 | 5 556 | |||||
| Financial debts and derivative financial instruments | 21 | 7 031 | 9 678 | ||||
| Lease liabilities | 10 | 246 | |||||
| Current income tax liabilities | 2 194 | 2 038 | |||||
| Provisions and other current liabilities | 22 | 13 338 | 12 284 | ||||
| Total current liabilities without disposal group | 28 233 | 29 556 | |||||
| Liabilities of disposal group held for sale | 2 | 31 | 51 | ||||
| Total current liabilities | 28 264 | 29 607 | |||||
| Total liabilities | 62 819 | 66 871 | |||||
| Total equity and liabilities | 118 370 | 145 563 | |||||
| The accompanying Notes form an integral part of the consolidated financial statements. | |||||||
(USD millions) | Note | Share capital | Treasury shares | Retained earnings | Total value adjustments | Issued share capital and reserves attributable to Novartis shareholders | Non- controlling interests | Total equity | |||||||||
| Total equity at January 1, 2017 | 972 | – 76 | 81 148 | – 7 212 | 74 832 | 59 | 74 891 | ||||||||||
| Net income | 7 703 | 7 703 | 7 703 | ||||||||||||||
| Other comprehensive income | 8 | – 37 | 2 872 | 2 835 | 2 | 2 837 | |||||||||||
| Total comprehensive income | 7 666 | 2 872 | 10 538 | 2 | 10 540 | ||||||||||||
| Dividends | 18.1 | – 6 495 | – 6 495 | – 6 495 | |||||||||||||
| Purchase of treasury shares | 18.2 | – 36 | – 5 538 | – 5 574 | – 5 574 | ||||||||||||
| Reduction of share capital | 18 | – 3 | 5 | – 2 | |||||||||||||
| Exercise of options and employee transactions | 18.2 | 2 | 253 | 255 | 255 | ||||||||||||
| Equity-based compensation | 18.2 | 5 | 607 | 612 | 612 | ||||||||||||
| Changes in non-controlling interests | 18.6 | – 2 | – 2 | ||||||||||||||
| Total of other equity movements | – 3 | – 24 | – 11 175 | – 11 202 | – 2 | – 11 204 | |||||||||||
| Total equity at December 31, 2017, as previously reported | 969 | – 100 | 77 639 | – 4 340 | 74 168 | 59 | 74 227 | ||||||||||
| Impact of change in accounting policies | 1 | 237 | – 177 | 60 | 60 | ||||||||||||
| Restated equity at January 1, 2018 | 969 | – 100 | 77 876 | – 4 517 | 74 228 | 59 | 74 287 | ||||||||||
| Net income | 12 611 | 12 611 | 3 | 12 614 | |||||||||||||
| Other comprehensive income | 8 | – 482 | 81 | – 401 | – 5 | – 406 | |||||||||||
| Total comprehensive income | 12 129 | 81 | 12 210 | – 2 | 12 208 | ||||||||||||
| Dividends | 18.1 | – 6 966 | – 6 966 | – 6 966 | |||||||||||||
| Purchase of treasury shares | 18.2 | – 13 | – 1 960 | – 1 973 | – 1 973 | ||||||||||||
| Reduction of share capital | 18 | – 25 | 34 | – 9 | |||||||||||||
| Exercise of options and employee transactions | 18.2 | 4 | 430 | 434 | 434 | ||||||||||||
| Other share sales | 18.2 | 2 | 261 | 263 | 263 | ||||||||||||
| Equity-based compensation | 18.2 | 4 | 752 | 756 | 756 | ||||||||||||
| Increase of treasury share repurchase obligation under a share buyback trading plan | 18.3 | – 284 | – 284 | – 284 | |||||||||||||
| Transaction costs, net of taxes | 18.4 | – 79 | – 79 | – 79 | |||||||||||||
| Fair value adjustments on financial assets sold | 8 | 16 | – 16 | ||||||||||||||
| Impact of change in ownership of consolidated entities | 18.5 | – 13 | – 13 | 22 | 9 | ||||||||||||
| Changes in non-controlling interests | 18.6 | – 1 | – 1 | ||||||||||||||
| Other movements | 18.7 | 38 | 38 | 38 | |||||||||||||
| Total of other equity movements | – 25 | 31 | – 7 814 | – 16 | – 7 824 | 21 | – 7 803 | ||||||||||
| Total equity at December 31, 2018, as previously reported | 944 | – 69 | 82 191 | – 4 452 | 78 614 | 78 | 78 692 | ||||||||||
| Impact of change in accounting policies | 1 | 3 | 3 | 3 | |||||||||||||
| Restated equity at January 1, 2019 | 944 | – 69 | 82 194 | – 4 452 | 78 617 | 78 | 78 695 | ||||||||||
| Net income | 11 732 | 11 732 | 5 | 11 737 | |||||||||||||
| Other comprehensive income | 8 | – 94 | – 113 | – 207 | – 3 | – 210 | |||||||||||
| Total comprehensive income | 11 638 | – 113 | 11 525 | 2 | 11 527 | ||||||||||||
| Dividends | 18.1 | – 6 645 | – 6 645 | – 6 645 | |||||||||||||
| Dividend in kind to effect the spin-off of Alcon Inc. | 2 | – 23 434 | – 23 434 | – 23 434 | |||||||||||||
| Purchase of treasury shares | 18.2 | – 31 | – 5 480 | – 5 511 | – 5 511 | ||||||||||||
| Reduction of share capital | 18 | – 8 | 12 | – 4 | |||||||||||||
| Exercise of options and employee transactions | 18.2 | 3 | 207 | 210 | 210 | ||||||||||||
| Equity-based compensation | 18.2 | 5 | 828 | 833 | 833 | ||||||||||||
| Shares delivered to Alcon employees as a result of the Alcon spin-off | 18.2 | 18 | 18 | 18 | |||||||||||||
| Taxes on treasury share transactions | – 189 | – 189 | – 189 | ||||||||||||||
| Decrease of treasury share repurchase obligation under a share buyback trading plan | 18.3 | 284 | 284 | 284 | |||||||||||||
| Transaction costs, net of taxes | 18.4 | – 253 | – 253 | – 253 | |||||||||||||
| Fair value adjustments on financial assets sold | 8 | 95 | – 95 | ||||||||||||||
| Impact of change in ownership of consolidated entities | 18.5 | – 3 | – 3 | – 2 | – 5 | ||||||||||||
| Changes in non-controlling interests | 18.6 | – 1 | – 1 | ||||||||||||||
| Fair value adjustments related to divestments | 8 | – 3 | 3 | ||||||||||||||
| Other movements | 18.7 | 22 | 22 | 22 | |||||||||||||
| Total of other equity movements | – 8 | – 11 | – 34 557 | – 92 | – 34 668 | – 3 | – 34 671 | ||||||||||
| Total equity at December 31, 2019 | 936 | – 80 | 59 275 | – 4 657 | 55 474 | 77 | 55 551 | ||||||||||
| The accompanying Notes form an integral part of the consolidated financial statements. | |||||||||||||||||
| (USD millions) | Note | 2019 | 2018 | 2017 | |||||
| Net income from continuing operations | 7 147 | 12 800 | 7 499 | ||||||
| Adjustments to reconcile net income from continuing operations to net cash flows from operating activities from continuing operations | |||||||||
| Reversal of non-cash items and other adjustments | 23.1 | 9 122 | 1 486 | 5 787 | |||||
| Dividends received from associated companies and others | 463 | 719 | 987 | ||||||
| Interest received | 214 | 241 | 97 | ||||||
| Interest paid | – 793 | – 816 | – 697 | ||||||
| Other financial receipts | 28 | 218 | |||||||
| Other financial payments | – 33 | – 31 | – 270 | ||||||
| Taxes paid | 23.2 | – 1 876 | – 1 506 | – 1 487 | |||||
| Net cash flows from operating activities from continuing operations before working capital and provision changes | 14 272 | 13 111 | 11 916 | ||||||
| Payments out of provisions and other net cash movements in non-current liabilities | – 924 | – 638 | – 829 | ||||||
| Change in net current assets and other operating cash flow items | 23.3 | 199 | 576 | 332 | |||||
| Net cash flows from operating activities from continuing operations | 13 547 | 13 049 | 11 419 | ||||||
| Net cash flows from operating activities from discontinued operations | 78 | 1 223 | 1 202 | ||||||
| Total net cash flows from operating activities | 13 625 | 14 272 | 12 621 | ||||||
| Purchase of property, plant and equipment | – 1 379 | – 1 254 | – 1 325 | ||||||
| Proceeds from sales of property, plant and equipment | 857 | 102 | 91 | ||||||
| Purchase of intangible assets | – 878 | – 1 394 | – 969 | ||||||
| Proceeds from sales of intangible assets | 973 | 823 | 640 | ||||||
| Purchase of financial assets | – 302 | – 205 | – 354 | ||||||
| Proceeds from sales of financial assets | 1 152 | 165 | 328 | ||||||
| Purchase of other non-current assets | – 60 | – 39 | – 40 | ||||||
| Proceeds from sales of other non-current assets | 3 | 9 | 1 | ||||||
| Acquisitions and divestments of interests in associated companies, net | 23.4 | – 6 | 12 854 | 29 | |||||
| Acquisitions and divestments of businesses, net | 23.5 | – 3 760 | – 13 683 | – 714 | |||||
| Purchase of marketable securities and commodities | – 228 | – 2 440 | – 580 | ||||||
| Proceeds from sales of marketable securities and commodities | 2 561 | 472 | 549 | ||||||
| Net cash flows used in investing activities from continuing operations | – 1 067 | – 4 590 | – 2 344 | ||||||
| Net cash flows used in investing activities from discontinued operations | 30 | – 1 159 | – 1 001 | – 775 | |||||
| Total net cash flows used in investing activities | – 2 226 | – 5 591 | – 3 119 | ||||||
| Dividends paid to shareholders of Novartis AG | – 6 645 | – 6 966 | – 6 495 | ||||||
| Acquisition of treasury shares | – 5 533 | – 2 036 | – 5 490 | ||||||
| Proceeds from exercise of options and other treasury share transactions | 201 | 700 | 252 | ||||||
| Increase in non-current financial debts | 23.6 | 93 | 2 856 | 4 933 | |||||
| Repayment of non-current financial debts | 23.6 | – 3 195 | – 366 | – 188 | |||||
| Change in current financial debts | 23.6 | – 1 582 | 1 687 | – 644 | |||||
| Payments of lease liabilities, net | 23.6 | – 273 | |||||||
| Impact of change in ownership of consolidated entities | – 6 | – 19 | 0 | ||||||
| Other financing cash flows, net | 56 | 67 | 314 | ||||||
| Net cash flows used in financing activities from continuing operations | – 16 884 | – 4 077 | – 7 318 | ||||||
| Net cash flows from/used in financing activities from discontinued operations | 30 | 3 257 | – 167 | – 415 | |||||
| Total net cash flows used in financing activities | – 13 627 | – 4 244 | – 7 733 | ||||||
| Net change in cash and cash equivalents before effect of exchange rate changes | – 2 228 | 4 437 | 1 769 | ||||||
| Effect of exchange rate changes on cash and cash equivalents | 69 | – 26 | 84 | ||||||
| Total net change in cash and cash equivalents | – 2 159 | 4 411 | 1 853 | ||||||
| Cash and cash equivalents at January 1 | 13 271 | 8 860 | 7 007 | ||||||
| Cash and cash equivalents at December 31 | 11 112 | 13 271 | 8 860 | ||||||
| The accompanying Notes form an integral part of the consolidated financial statements. | |||||||||
Notes to the Novartis Group consolidated financial statements
| Useful life | |||
| Buildings | 20 to 40 years | ||
| Machinery and other equipment | |||
| Machinery and equipment | 7 to 20 years | ||
| Furniture and vehicles | 5 to 10 years | ||
| Computer hardware | 3 to 7 years | ||
Useful life | Income statement location for amortization and impairment charges | ||||
| Currently marketed products | 5 to 20 years | "Cost of goods sold" | |||
| Marketing know-how | 25 years | "Cost of goods sold" | |||
| Technologies | 10 to 20 years | "Cost of goods sold" or "Research and development" | |||
| Other (including computer software) | 3 to 7 years | In the respective functional expense | |||
| Alcon brand name | Not amortized, indefinite useful life | "Other expense" | |||
(USD millions) | April 8, 2019 | ||
Net assets derecognized 1 | – 20 025 | ||
| Derecognition of distribution liability | 23 434 | ||
| Difference between net assets and distribution liability | 3 409 | ||
| Recognition of Alcon Inc. shares obtained through consolidated foundations | 1 273 | ||
| Currency translation gains recycled into the consolidated income statement | 123 | ||
| Transaction costs recognized in the consolidated income statement | – 114 | ||
| Gain on distribution of Alcon Inc. to Novartis AG shareholders | 4 691 | ||
1 See Note 30 for additional information. | |||
(USD millions) | December 31, 2019 | December 31, 2018 | |||
| Assets of disposal group classified as held for sale | |||||
| Property, plant and equipment | 169 | 148 | |||
| Intangible assets other than goodwill | 475 | 478 | |||
| Deferred tax assets | 11 | 8 | |||
| Other non-current assets | 2 | 1 | |||
| Inventories | 181 | 165 | |||
| Other current assets | 3 | 7 | |||
| Total | 841 | 807 | |||
(USD millions) | December 31, 2019 | December 31, 2018 | |||
| Liabilities of disposal group classified as held for sale | |||||
| Deferred tax liabilities | 2 | 2 | |||
| Provisions and other non-current liabilities | 4 | 4 | |||
| Provisions and other current liabilities | 25 | 45 | |||
| Total | 31 | 51 | |||
| Innovative Medicines | Sandoz | Corporate (including eliminations) | Group | ||||||||||||||
| (USD millions) | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | |||||||||
| Net sales to third parties from continuing operations | 37 714 | 34 892 | 9 731 | 9 859 | 47 445 | 44 751 | |||||||||||
| Sales to continuing and discontinued segments | 783 | 741 | 141 | 177 | – 871 | – 836 | 53 | 82 | |||||||||
| Net sales from continuing operations | 38 497 | 35 633 | 9 872 | 10 036 | – 871 | – 836 | 47 498 | 44 833 | |||||||||
| Other revenues | 1 092 | 1 188 | 63 | 62 | 24 | 16 | 1 179 | 1 266 | |||||||||
| Cost of goods sold | – 10 050 | – 9 870 | – 5 334 | – 5 530 | 959 | 890 | – 14 425 | – 14 510 | |||||||||
| Gross profit from continuing operations | 29 539 | 26 951 | 4 601 | 4 568 | 112 | 70 | 34 252 | 31 589 | |||||||||
| Selling, general and administration | – 11 617 | – 10 907 | – 2 218 | – 2 305 | – 534 | – 505 | – 14 369 | – 13 717 | |||||||||
| Research and development | – 8 152 | – 7 675 | – 1 250 | – 814 | – 9 402 | – 8 489 | |||||||||||
| Other income | 1 586 | 977 | 167 | 505 | 278 | 147 | 2 031 | 1 629 | |||||||||
| Other expense | – 2 069 | – 1 475 | – 749 | – 622 | – 608 | – 512 | – 3 426 | – 2 609 | |||||||||
| Operating income from continuing operations | 9 287 | 7 871 | 551 | 1 332 | – 752 | – 800 | 9 086 | 8 403 | |||||||||
| Income from associated companies | 1 | 1 | 2 | 5 | 656 | 6 432 | 659 | 6 438 | |||||||||
| Interest expense | – 850 | – 932 | |||||||||||||||
| Other financial income and expense | 45 | 186 | |||||||||||||||
| Income before taxes from continuing operations | 8 940 | 14 095 | |||||||||||||||
| Taxes | – 1 793 | – 1 295 | |||||||||||||||
| Net income from continuing operations | 7 147 | 12 800 | |||||||||||||||
| Net loss from discontinued operations before gain on distribution of Alcon Inc. to Novartis AG shareholders | – 101 | – 186 | |||||||||||||||
| Gain on distribution of Alcon Inc. to Novartis AG shareholders | 4 691 | ||||||||||||||||
| Net income/(loss) from discontinued operations | 4 590 | – 186 | |||||||||||||||
| Net income | 11 737 | 12 614 | |||||||||||||||
| Attributable to: | |||||||||||||||||
| Shareholders of Novartis AG | 11 732 | 12 611 | |||||||||||||||
| Non-controlling interests | 5 | 3 | |||||||||||||||
| Included in net income from continuing operations are: | |||||||||||||||||
| Interest income | 245 | 292 | |||||||||||||||
| Depreciation of property, plant and equipment | – 952 | – 1 075 | – 283 | – 285 | – 110 | – 122 | – 1 345 | – 1 482 | |||||||||
Depreciation of right-of-use assets 1 | – 247 | – 41 | – 17 | – 305 | |||||||||||||
| Amortization of intangible assets | – 2 509 | – 2 214 | – 315 | – 366 | – 12 | – 7 | – 2 836 | – 2 587 | |||||||||
| Impairment charges on property, plant and equipment, net | – 100 | – 239 | – 101 | – 60 | – 1 | – 2 | – 202 | – 301 | |||||||||
| Impairment charges on intangible assets, net | – 632 | – 592 | – 506 | – 249 | – 1 138 | – 841 | |||||||||||
| Impairment charges and fair value changes on financial assets, net | 18 | 107 | 20 | – 113 | 38 | – 6 | |||||||||||
| Additions to restructuring provisions | – 229 | – 395 | – 165 | – 32 | – 98 | – 94 | – 492 | – 521 | |||||||||
| Equity-based compensation of Novartis equity plans | – 761 | – 645 | – 67 | – 53 | – 239 | – 220 | – 1 067 | – 918 | |||||||||
1 Depreciation of right-of-use assets recognized from January 1, 2019, the date of implementation of IFRS 16 leases. Notes 1 and 10 provide additional disclosures. | |||||||||||||||||
| Innovative Medicines | Sandoz | Corporate (including eliminations) | Group | ||||||||||||||
| (USD millions) | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | |||||||||
| Net sales to third parties from continuing operations | 34 892 | 32 278 | 9 859 | 10 060 | 44 751 | 42 338 | |||||||||||
| Sales to continuing and discontinued segments | 741 | 668 | 177 | 118 | – 836 | – 743 | 82 | 43 | |||||||||
| Net sales from continuing operations | 35 633 | 32 946 | 10 036 | 10 178 | – 836 | – 743 | 44 833 | 42 381 | |||||||||
| Other revenues | 1 188 | 898 | 62 | 37 | 16 | 88 | 1 266 | 1 023 | |||||||||
| Cost of goods sold | – 9 870 | – 8 650 | – 5 530 | – 5 800 | 890 | 817 | – 14 510 | – 13 633 | |||||||||
| Gross profit from continuing operations | 26 951 | 25 194 | 4 568 | 4 415 | 70 | 162 | 31 589 | 29 771 | |||||||||
| Selling, general and administration | – 10 907 | – 9 887 | – 2 305 | – 2 126 | – 505 | – 452 | – 13 717 | – 12 465 | |||||||||
| Research and development | – 7 675 | – 7 615 | – 814 | – 774 | – 8 489 | – 8 389 | |||||||||||
| Other income | 977 | 1 027 | 505 | 204 | 147 | 691 | 1 629 | 1 922 | |||||||||
| Other expense | – 1 475 | – 1 124 | – 622 | – 351 | – 512 | – 662 | – 2 609 | – 2 137 | |||||||||
| Operating income from continuing operations | 7 871 | 7 595 | 1 332 | 1 368 | – 800 | – 261 | 8 403 | 8 702 | |||||||||
| Income from associated companies | 1 | – 1 | 5 | 23 | 6 432 | 1 086 | 6 438 | 1 108 | |||||||||
| Interest expense | – 932 | – 750 | |||||||||||||||
| Other financial income and expense | 186 | 42 | |||||||||||||||
| Income before taxes from continuing operations | 14 095 | 9 102 | |||||||||||||||
| Taxes | – 1 295 | – 1 603 | |||||||||||||||
| Net income from continuing operations | 12 800 | 7 499 | |||||||||||||||
| Net (loss)/income from discontinued operations | – 186 | 204 | |||||||||||||||
| Net income | 12 614 | 7 703 | |||||||||||||||
| Attributable to: | |||||||||||||||||
| Shareholders of Novartis AG | 12 611 | 7 703 | |||||||||||||||
| Non-controlling interests | 3 | 0 | |||||||||||||||
| Included in net income from continuing operations are: | |||||||||||||||||
| Interest income | 292 | 110 | |||||||||||||||
| Depreciation of property, plant and equipment | – 1 075 | – 916 | – 285 | – 270 | – 122 | – 117 | – 1 482 | – 1 303 | |||||||||
| Amortization of intangible assets | – 2 214 | – 2 167 | – 366 | – 447 | – 7 | – 10 | – 2 587 | – 2 624 | |||||||||
| Impairment charges on property, plant and equipment, net | – 239 | – 84 | – 60 | – 73 | – 2 | – 301 | – 157 | ||||||||||
| Impairment charges on intangible assets, net | – 592 | – 591 | – 249 | – 61 | – 841 | – 652 | |||||||||||
| Impairment charges and fair value changes on financial assets, net | 107 | – 42 | – 113 | – 185 | – 6 | – 227 | |||||||||||
| Additions to restructuring provisions | – 395 | – 122 | – 32 | – 61 | – 94 | – 3 | – 521 | – 186 | |||||||||
| Equity-based compensation of Novartis equity plans | – 645 | – 593 | – 53 | – 52 | – 220 | – 208 | – 918 | – 853 | |||||||||
| Innovative Medicines | Sandoz | Alcon1 | Corporate (including eliminations) | Group | |||||||||||||||||
| (USD millions) | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | |||||||||||
| Total assets | 71 225 | 67 055 | 16 468 | 17 328 | 25 971 | 30 677 | 35 209 | 118 370 | 145 563 | ||||||||||||
| Total liabilities | – 15 332 | – 13 056 | – 3 804 | – 3 377 | – 1 964 | – 43 683 | – 48 474 | – 62 819 | – 66 871 | ||||||||||||
| Total equity | 55 551 | 78 692 | |||||||||||||||||||
| Net debt | 15 938 | 16 184 | |||||||||||||||||||
| Net operating assets | 55 893 | 53 999 | 12 664 | 13 951 | 24 007 | 71 489 | 94 876 | ||||||||||||||
| Included in assets and liabilities are: | |||||||||||||||||||||
| Total property, plant and equipment | 9 632 | 10 098 | 1 888 | 2 159 | 2 878 | 549 | 561 | 12 069 | 15 696 | ||||||||||||
Additions to property, plant and equipment 2 | 1 114 | 822 | 217 | 294 | 519 | 143 | 139 | 1 474 | 1 774 | ||||||||||||
Total right-of-use assets 3 | 1 487 | 136 | 54 | 1 677 | |||||||||||||||||
Additions to right-of-use assets 2, 3 | 454 | 49 | 34 | 537 | |||||||||||||||||
| Total goodwill and intangible assets | 46 336 | 44 593 | 8 892 | 9 712 | 19 578 | 83 | 130 | 55 311 | 74 013 | ||||||||||||
Additions to goodwill and intangible assets 2 | 647 | 1 265 | 68 | 107 | 196 | 52 | 24 | 767 | 1 592 | ||||||||||||
| Total investment in associated companies | 128 | 81 | 7 | 7 | 8 509 | 8 264 | 8 644 | 8 352 | |||||||||||||
| Additions to investment in associated companies | 44 | 18 | 11 | 11 | 55 | 29 | |||||||||||||||
| Cash and cash equivalents, marketable securities, commodities, time deposits and derivative financial instruments | 11 446 | 15 964 | 11 446 | 15 964 | |||||||||||||||||
| Financial debts and derivative financial instruments | 27 384 | 32 148 | 27 384 | 32 148 | |||||||||||||||||
| Current income tax and deferred tax liabilities | 8 061 | 9 513 | 8 061 | 9 513 | |||||||||||||||||
1 From February 28, 2019, the Alcon Division was reported as discontinued operations (see Notes 1, 2 and 30). In accordance with IFRS, the December 31, 2018, consolidated balance sheet includes the assets and liabilities of the Alcon eye care devices business and certain Corporate assets and liabilities attributable to the Alcon business. Note 30 provides additional information on discontinued operations. | |||||||||||||||||||||
2 Excluding the impact of business combinations. | |||||||||||||||||||||
3 Total right-of-use assets and additions to right-of-use assets recognized in 2019 with the implementation of IFRS 16 Leases on January 1, 2019. See Notes 1 and 10 for additional disclosures. | |||||||||||||||||||||
Net sales1 | Total of selected non-current assets2 | ||||||||||||||||||||
| (USD millions) | 2019 | % | 2018 | % | 2017 | % | 2019 | % | 2018 | % | |||||||||||
| Country | |||||||||||||||||||||
| Switzerland | 848 | 2 | 795 | 2 | 780 | 2 | 33 032 | 43 | 41 972 | 43 | |||||||||||
| United States | 16 280 | 34 | 14 618 | 33 | 14 135 | 33 | 28 893 | 37 | 39 082 | 40 | |||||||||||
| France | 2 442 | 5 | 2 505 | 6 | 2 289 | 5 | 3 933 | 5 | 3 976 | 4 | |||||||||||
| Germany | 4 120 | 9 | 3 972 | 9 | 3 484 | 8 | 2 554 | 3 | 3 124 | 3 | |||||||||||
| Japan | 2 656 | 6 | 2 575 | 6 | 2 617 | 6 | 309 | 144 | |||||||||||||
| Other | 21 099 | 44 | 20 286 | 44 | 19 033 | 46 | 8 980 | 12 | 9 763 | 10 | |||||||||||
| Group | 47 445 | 100 | 44 751 | 100 | 42 338 | 100 | 77 701 | 100 | 98 061 | 100 | |||||||||||
| Region | |||||||||||||||||||||
| Europe | 17 933 | 38 | 17 259 | 39 | 15 760 | 37 | 46 103 | 59 | 55 913 | 57 | |||||||||||
| Americas | 19 713 | 41 | 18 032 | 39 | 17 485 | 41 | 29 389 | 38 | 39 082 | 40 | |||||||||||
| Asia/Africa/Australasia | 9 799 | 21 | 9 460 | 22 | 9 093 | 22 | 2 209 | 3 | 3 066 | 3 | |||||||||||
| Group | 47 445 | 100 | 44 751 | 100 | 42 338 | 100 | 77 701 | 100 | 98 061 | 100 | |||||||||||
1 Net sales from operations by location of third-party customer | |||||||||||||||||||||
2 Total of property, plant and equipment; goodwill; intangible assets; and investment in associated companies, and in 2019 right-of-use assets recognized with the implementation of IFRS 16 Leases on January 1, 2019. See Notes 1 and 10 for additional disclosures. | |||||||||||||||||||||
2019 USD m | 2018 USD m | Change (2018 to 2019) USD % | 2017 USD m | Change (2017 to 2018) USD % | |||||||
| Innovative Medicines | |||||||||||
| Europe | 12 818 | 12 296 | 4 | 11 127 | 11 | ||||||
| US | 13 789 | 11 864 | 16 | 10 857 | 9 | ||||||
| Asia/Africa/Australasia | 8 458 | 8 097 | 4 | 7 702 | 5 | ||||||
| Canada and Latin America | 2 649 | 2 635 | 1 | 2 592 | 2 | ||||||
| Total | 37 714 | 34 892 | 8 | 32 278 | 8 | ||||||
| Of which in Established Markets | 28 573 | 26 258 | 9 | 24 174 | 9 | ||||||
| Of which in Emerging Growth Markets | 9 141 | 8 634 | 6 | 8 104 | 7 | ||||||
| Sandoz | |||||||||||
| Europe | 5 115 | 4 963 | 3 | 4 633 | 7 | ||||||
| US | 2 491 | 2 754 | – 10 | 3 278 | – 16 | ||||||
| Asia/Africa/Australasia | 1 341 | 1 363 | – 2 | 1 391 | – 2 | ||||||
| Canada and Latin America | 784 | 779 | 1 | 758 | 3 | ||||||
| Total | 9 731 | 9 859 | – 1 | 10 060 | – 2 | ||||||
| Of which in Established Markets | 7 111 | 7 233 | – 2 | 7 383 | – 2 | ||||||
| Of which in Emerging Growth Markets | 2 620 | 2 626 | 0 | 2 677 | – 2 | ||||||
| Group | |||||||||||
| Europe | 17 933 | 17 259 | 4 | 15 760 | 10 | ||||||
| US | 16 280 | 14 618 | 11 | 14 135 | 3 | ||||||
| Asia/Africa/Australasia | 9 799 | 9 460 | 4 | 9 093 | 4 | ||||||
| Canada and Latin America | 3 433 | 3 414 | 1 | 3 350 | 2 | ||||||
| Total | 47 445 | 44 751 | 6 | 42 338 | 6 | ||||||
| Of which in Established Markets | 35 684 | 33 491 | 7 | 31 557 | 6 | ||||||
| Of which in Emerging Growth Markets | 11 761 | 11 260 | 4 | 10 781 | 4 | ||||||
1 Net sales from operations by location of third-party customer. Emerging Growth Markets comprise all markets other than the Established Markets of the US, Canada, Western Europe, Japan, Australia and New Zealand. | |||||||||||
2019 USD m | 2018 USD m | Change (2018 to 2019) USD % | 2017 USD m | Change (2017 to 2018) USD % | |||||||
| Oncology | |||||||||||
| Tasigna | 1 880 | 1 874 | 0 | 1 841 | 2 | ||||||
| Sandostatin | 1 585 | 1 587 | 0 | 1 612 | – 2 | ||||||
| Afinitor/Votubia | 1 539 | 1 556 | – 1 | 1 525 | 2 | ||||||
| Promacta/Revolade | 1 416 | 1 174 | 21 | 867 | 35 | ||||||
Tafinlar +Mekinist | 1 338 | 1 155 | 16 | 873 | 32 | ||||||
| Gleevec/Glivec | 1 263 | 1 561 | – 19 | 1 943 | – 20 | ||||||
| Jakavi | 1 114 | 977 | 14 | 777 | 26 | ||||||
| Exjade/Jadenu | 975 | 1 099 | – 11 | 1 059 | 4 | ||||||
| Votrient | 755 | 828 | – 9 | 808 | 2 | ||||||
| Kisqali | 480 | 235 | 104 | 76 | nm | ||||||
| Lutathera | 441 | 167 | 164 | 0 | nm | ||||||
| Kymriah | 278 | 76 | nm | 6 | nm | ||||||
| Piqray | 116 | nm | nm | ||||||||
| Other | 1 190 | 1 139 | 4 | 887 | 28 | ||||||
| Total Novartis Oncology business unit | 14 370 | 13 428 | 7 | 12 274 | 9 | ||||||
| Ophthalmology | |||||||||||
| Lucentis | 2 086 | 2 046 | 2 | 1 888 | 8 | ||||||
| Travoprost Group | 433 | 517 | – 16 | 589 | – 12 | ||||||
| Xiidra | 192 | nm | nm | ||||||||
| Beovu | 35 | nm | nm | ||||||||
| Other | 2 030 | 1 995 | 2 | 2 144 | – 7 | ||||||
| Total Ophthalmology | 4 776 | 4 558 | 5 | 4 621 | – 1 | ||||||
| Immunology, Hepatology and Dermatology | |||||||||||
| Cosentyx | 3 551 | 2 837 | 25 | 2 071 | 37 | ||||||
| Ilaris | 671 | 554 | 21 | 402 | 38 | ||||||
| Other | 1 | nm | 1 | 0 | |||||||
| Total Immunology, Hepatology and Dermatology | 4 222 | 3 392 | 24 | 2 474 | 37 | ||||||
| Neuroscience | |||||||||||
| Gilenya | 3 223 | 3 341 | – 4 | 3 185 | 5 | ||||||
| Zolgensma | 361 | nm | nm | ||||||||
| Aimovig | 103 | 8 | nm | nm | |||||||
| Mayzent | 26 | nm | nm | ||||||||
| Other | 60 | 80 | – 25 | 102 | – 22 | ||||||
| Total Neuroscience | 3 773 | 3 429 | 10 | 3 287 | 4 | ||||||
2019 USD m | 2018 USD m | Change (2018 to 2019) USD % | 2017 USD m | Change (2017 to 2018) USD % | |||||||
| Respiratory | |||||||||||
Xolair 1 | 1 173 | 1 039 | 13 | 920 | 13 | ||||||
| Ultibro Breezhaler | 427 | 454 | – 6 | 411 | 10 | ||||||
| Seebri Breezhaler | 121 | 148 | – 18 | 151 | – 2 | ||||||
| Onbrez Breezhaler | 82 | 101 | – 19 | 112 | – 10 | ||||||
| Other | 22 | 25 | – 12 | 23 | 9 | ||||||
| Total Respiratory | 1 825 | 1 767 | 3 | 1 617 | 9 | ||||||
| Cardiovascular, Renal and Metabolism | |||||||||||
| Entresto | 1 726 | 1 028 | 68 | 507 | 103 | ||||||
| Other | 24 | 22 | 9 | 17 | 29 | ||||||
| Total Cardiovascular, Renal and Metabolism | 1 750 | 1 050 | 67 | 524 | 100 | ||||||
| Established Medicines | |||||||||||
Galvus Group | 1 297 | 1 284 | 1 | 1 233 | 4 | ||||||
Diovan Group | 1 064 | 1 023 | 4 | 957 | 7 | ||||||
Exforge Group | 1 025 | 1 002 | 2 | 960 | 4 | ||||||
| Zortress/Certican | 485 | 464 | 5 | 414 | 12 | ||||||
| Neoral/Sandimmun(e) | 419 | 463 | – 10 | 488 | – 5 | ||||||
| Voltaren/Cataflam | 417 | 445 | – 6 | 465 | – 4 | ||||||
| Other | 2 291 | 2 587 | – 11 | 2 964 | – 13 | ||||||
| Total Established Medicines | 6 998 | 7 268 | – 4 | 7 481 | – 3 | ||||||
| Total Novartis Pharmaceuticals business unit | 23 344 | 21 464 | 9 | 20 004 | 7 | ||||||
| Total division net sales | 37 714 | 34 892 | 8 | 32 278 | 8 | ||||||
1 Net sales reflectXolair sales for all indications. | |||||||||||
| nm = not meaningful | |||||||||||
Brands | Business franchise | Indication | US USD m | Rest of world USD m | Total USD m | ||||||
| Cosentyx | Immunology, Hepatology and Dermatology | Psoriasis, ankylosing spondylitis and psoriatic arthritis | 2 220 | 1 331 | 3 551 | ||||||
| Gilenya | Neuroscience | Relapsing multiple sclerosis | 1 736 | 1 487 | 3 223 | ||||||
| Lucentis | Ophthalmology | Age-related macular degeneration | 2 086 | 2 086 | |||||||
| Tasigna | Oncology | Chronic myeloid leukemia | 804 | 1 076 | 1 880 | ||||||
| Entresto | Cardiovascular, Renal and Metabolism | Chronic heart failure | 925 | 801 | 1 726 | ||||||
| Sandostatin | Oncology | Carcinoid tumors and acromegaly | 881 | 704 | 1 585 | ||||||
| Afinitor/Votubia | Oncology | Breast cancer/TSC | 1 003 | 536 | 1 539 | ||||||
| Promacta/Revolade | Oncology | Immune thrombocytopenia (ITP), severe aplastic anemia (SAA) | 691 | 725 | 1 416 | ||||||
Tafinlar +Mekinist | Oncology | BRAF V600+ metastatic and adjuvant melanoma; advanced non-small cell lung cancer (NSCLC) | 481 | 857 | 1 338 | ||||||
Galvus Group | Established Medicines | Diabetes | 1 297 | 1 297 | |||||||
| Gleevec/Glivec | Oncology | Chronic myeloid leukemia and GIST | 334 | 929 | 1 263 | ||||||
Xolair 1 | Respiratory | Severe Allergic Asthma (SAA) and Chronic Spontaneous Urticaria (CSU) | 1 173 | 1 173 | |||||||
| Jakavi | Oncology | Myelofibrosis (MF), polycytomia vera (PV) | 1 114 | 1 114 | |||||||
Diovan Group | Established Medicines | Hypertension | 86 | 978 | 1 064 | ||||||
Exforge Group | Established Medicines | Hypertension | 13 | 1 012 | 1 025 | ||||||
| Exjade/Jadenu | Oncology | Chronic iron overload | 450 | 525 | 975 | ||||||
| Votrient | Oncology | Renal cell carcinoma | 332 | 423 | 755 | ||||||
| Ilaris | Immunology, Hepatology and Dermatology | Auto-inflammatory (CAPS, TRAPS, HIDS/MKD, FMF, SJIA, AOSD and gout) | 304 | 367 | 671 | ||||||
| Zortress/Certican | Established Medicines | Transplantation | 169 | 316 | 485 | ||||||
| Kisqali | Oncology | HR+/HER2- metastatic breast cancer | 250 | 230 | 480 | ||||||
| Top 20 products total | 10 679 | 17 967 | 28 646 | ||||||||
| Rest of portfolio | 3 110 | 5 958 | 9 068 | ||||||||
| Total division sales | 13 789 | 23 925 | 37 714 | ||||||||
1 Net sales reflectXolair sales for all indications. | |||||||||||
Brands | Business franchise | Indication | US USD m | Rest of world USD m | Total USD m | ||||||
| Gilenya | Neuroscience | Relapsing multiple sclerosis | 1 765 | 1 576 | 3 341 | ||||||
| Cosentyx | Immunology, Hepatology and Dermatology | Psoriasis, ankylosing spondylitis and psoriatic arthritis | 1 674 | 1 163 | 2 837 | ||||||
| Lucentis | Ophthalmology | Age-related macular degeneration | 2 046 | 2 046 | |||||||
| Tasigna | Oncology | Chronic myeloid leukemia | 806 | 1 068 | 1 874 | ||||||
| Sandostatin | Oncology | Carcinoid tumors and acromegaly | 817 | 770 | 1 587 | ||||||
| Gleevec/Glivec | Oncology | Chronic myeloid leukemia and GIST | 440 | 1 121 | 1 561 | ||||||
| Afinitor/Votubia | Oncology | Breast cancer/TSC | 929 | 627 | 1 556 | ||||||
Galvus Group | Established Medicines | Diabetes | 1 284 | 1 284 | |||||||
| Promacta/Revolade | Oncology | Immune thrombocytopenia (ITP), severe aplastic anemia (SAA) | 581 | 593 | 1 174 | ||||||
Tafinlar +Mekinist | Oncology | BRAF V600+ metastatic and adjuvant melanoma; advanced non-small cell lung cancer (NSCLC) | 457 | 698 | 1 155 | ||||||
| Exjade/Jadenu | Oncology | Chronic iron overload | 521 | 578 | 1 099 | ||||||
Xolair 1 | Respiratory | Severe Allergic Asthma (SAA) and Chronic Spontaneous Urticaria (CSU) | 1 039 | 1 039 | |||||||
| Entresto | Cardiovascular, Renal and Metabolism | Chronic heart failure | 556 | 472 | 1 028 | ||||||
Diovan Group | Established Medicines | Hypertension | 84 | 939 | 1 023 | ||||||
Exforge Group | Established Medicines | Hypertension | 19 | 983 | 1 002 | ||||||
| Jakavi | Oncology | Myelofibrosis (MF), polycytomia vera (PV) | 977 | 977 | |||||||
| Votrient | Oncology | Renal cell carcinoma | 404 | 424 | 828 | ||||||
| Ilaris | Immunology, Hepatology and Dermatology | Auto-inflammatory (CAPS, TRAPS, HIDS/MKD, FMF, SJIA, AOSD and gout) | 262 | 292 | 554 | ||||||
| Travoprost Group | Ophthalmology | Reduction of elevated intraocular pressure | 194 | 323 | 517 | ||||||
| Zortress/Certican | Established Medicines | Transplantation | 145 | 319 | 464 | ||||||
| Top 20 products total | 9 654 | 17 292 | 26 946 | ||||||||
| Rest of portfolio | 2 210 | 5 736 | 7 946 | ||||||||
| Total division sales | 11 864 | 23 028 | 34 892 | ||||||||
1 Net sales reflectXolair sales for all indications. | |||||||||||
Brands | Business franchise | Indication | US USD m | Rest of world USD m | Total USD m | ||||||
| Gilenya | Neuroscience | Relapsing multiple sclerosis | 1 709 | 1 476 | 3 185 | ||||||
| Cosentyx | Immunology, Hepatology and Dermatology | Psoriasis, ankylosing spondylitis and psoriatic arthritis | 1 275 | 796 | 2 071 | ||||||
| Gleevec/Glivec | Oncology | Chronic myeloid leukemia and GIST | 627 | 1 316 | 1 943 | ||||||
| Lucentis | Ophthalmology | Age-related macular degeneration | 1 888 | 1 888 | |||||||
| Tasigna | Oncology | Chronic myeloid leukemia | 810 | 1 031 | 1 841 | ||||||
| Sandostatin | Oncology | Carcinoid tumors and acromegaly | 832 | 780 | 1 612 | ||||||
| Afinitor/Votubia | Oncology | Breast cancer/TSC | 819 | 706 | 1 525 | ||||||
Galvus Group | Established Medicines | Diabetes | 1 233 | 1 233 | |||||||
| Exjade/Jadenu | Oncology | Chronic iron overload | 515 | 544 | 1 059 | ||||||
Exforge Group | Established Medicines | Hypertension | 28 | 932 | 960 | ||||||
Diovan Group | Established Medicines | Hypertension | 87 | 870 | 957 | ||||||
Xolair 1 | Respiratory | Severe Allergic Asthma (SAA) and Chronic Spontaneous Urticaria (CSU) | 920 | 920 | |||||||
Tafinlar +Mekinist | Oncology | BRAF V600+ metastatic and adjuvant melanoma; advanced non-small cell lung cancer (NSCLC) | 339 | 534 | 873 | ||||||
| Promacta/Revolade | Oncology | Immune thrombocytopenia (ITP), severe aplastic anemia (SAA) | 446 | 421 | 867 | ||||||
| Votrient | Oncology | Renal cell carcinoma | 407 | 401 | 808 | ||||||
| Jakavi | Oncology | Myelofibrosis (MF), polycytomia vera (PV) | 777 | 777 | |||||||
| Travoprost Group | Ophthalmology | Reduction of elevated intraocular pressure | 216 | 373 | 589 | ||||||
| Entresto | Cardiovascular, Renal and Metabolism | Chronic heart failure | 297 | 210 | 507 | ||||||
| Neoral/Sandimmun(e) | Immunology, Hepatology and Dermatology | Transplantation | 38 | 450 | 488 | ||||||
| Voltaren/Cataflam | Established Medicines | Inflammation/pain | 465 | 465 | |||||||
| Top 20 products total | 8 445 | 16 123 | 24 568 | ||||||||
| Rest of portfolio | 2 412 | 5 298 | 7 710 | ||||||||
| Total division sales | 10 857 | 21 421 | 32 278 | ||||||||
1 Net sales reflectXolair sales for all indications. | |||||||||||
2019 USD m | 2018 USD m | Change (2018 to 2019) USD % | 2017 USD m | Change (2017 to 2018) USD % | |||||||
Retail Generics 1 | 7 590 | 7 880 | – 4 | 8 409 | – 6 | ||||||
| Biopharmaceuticals | 1 607 | 1 436 | 12 | 1 135 | 27 | ||||||
| Anti-Infectives | 534 | 543 | – 2 | 516 | 5 | ||||||
| Total division net sales | 9 731 | 9 859 | – 1 | 10 060 | – 2 | ||||||
1 Of which USD 784 million (2018: USD 826 million; 2017: USD 880 million) represents anti-infectives sold under the Sandoz name | |||||||||||
| Innovative Medicines | Sandoz | Corporate (including eliminations) | Group | ||||||||||||||
| (USD millions) | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | |||||||||
| Profit-sharing income | 732 | 874 | 2 | 3 | 734 | 877 | |||||||||||
| Royalty income | 104 | 162 | 19 | 10 | 24 | 16 | 147 | 188 | |||||||||
| Milestone income | 201 | 128 | 30 | 45 | 231 | 173 | |||||||||||
Other 1 | 55 | 24 | 12 | 4 | 67 | 28 | |||||||||||
| Total other revenues | 1 092 | 1 188 | 63 | 62 | 24 | 16 | 1 179 | 1 266 | |||||||||
1 Other includes revenue from activities such as manufacturing or other services rendered, to the extent such revenue is not recorded under net sales. | |||||||||||||||||
| Innovative Medicines | Sandoz | Corporate (including eliminations) | Group | ||||||||||||||
| (USD millions) | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | 2018 | 2017 | |||||||||
| Profit-sharing income | 874 | 648 | 3 | 4 | 877 | 652 | |||||||||||
| Royalty income | 162 | 186 | 10 | 24 | 16 | 88 | 188 | 298 | |||||||||
| Milestone income | 128 | 28 | 45 | 173 | 28 | ||||||||||||
Other 1 | 24 | 36 | 4 | 9 | 28 | 45 | |||||||||||
| Total other revenues | 1 188 | 898 | 62 | 37 | 16 | 88 | 1 266 | 1 023 | |||||||||
1 Other includes revenue from activities such as manufacturing or other services rendered, to the extent such revenue is not recorded under net sales. | |||||||||||||||||
| Net income statement effect | Other comprehensive income effect 1 | Total comprehensive income effect | |||||||||||||||||
| (USD millions) | 2019 | 2018 | 2017 | 2019 | 2018 | 2017 | 2019 | 2018 | 2017 | ||||||||||
| Roche Holding AG, Switzerland | 662 | 526 | 456 | – 94 | 75 | 108 | 568 | 601 | 564 | ||||||||||
| GlaxoSmithKline Consumer Healthcare Holdings Ltd., UK | 5 910 | 629 | – 557 | – 145 | 5 353 | 484 | |||||||||||||
| Others | – 3 | 2 | 23 | – 3 | 2 | 23 | |||||||||||||
| Associated companies related to continuing operations | 659 | 6 438 | 1 108 | – 94 | – 482 | – 37 | 565 | 5 956 | 1 071 | ||||||||||
1 In 2018, Novartis share of other comprehensive income recognized by associated companies, net of taxes of USD 511 million was recycled into the consolidated income statement as a result of the divestment of the investment in GSK Consumer Healthcare Holdings Ltd. No Novartis share of other comprehensive income recognized by associated companies, net of taxes was recycled into the consolidated income statement in 2019 and 2017. | |||||||||||||||||||
| Balance sheet value | |||||
(USD millions) | December 31, 2019 | December 31, 2018 | |||
| Roche Holding AG, Switzerland | 8 445 | 8 195 | |||
| Others | 199 | 157 | |||
| Total | 8 644 | 8 352 | |||
(CHF billions) | Current assets | Non-current assets | Current liabilities | Non-current liabilities | |||||
| December 31, 2018 | 32.2 | 53.7 | 23.0 | 25.1 | |||||
| June 30, 2019 | 32.3 | 53.8 | 23.4 | 25.7 | |||||
(CHF billions) | Revenue | Net income | Other comprehen- sive income | Total comprehen- sive income | |||||
| December 31, 2018 | 59.5 | 8.6 | – 0.1 | 8.5 | |||||
| June 30, 2019 | 31.8 | 8.3 | – 0.6 | 7.7 | |||||
(USD millions) | December 31, 2019 | ||
| Novartis share of Roche’s estimated net assets | 2 404 | ||
| Novartis share of reappraised intangible assets | 297 | ||
| Implicit Novartis goodwill | 2 939 | ||
| Current value of share in net identifiable assets and goodwill | 5 640 | ||
| Accumulated equity accounting adjustments and translation effects less dividends received | 2 805 | ||
| Balance sheet value | 8 445 | ||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Novartis share of Roche's estimated current-year consolidated net income | 910 | 799 | 669 | ||||
| Prior-year adjustment | – 129 | – 125 | – 67 | ||||
| Amortization of fair value adjustments relating to intangible assets, net of taxes of USD 24 million (2018: USD 40 million; 2017: USD 42 million) | – 162 | – 148 | – 146 | ||||
| Partial release of deferred tax liability recognized | 43 | ||||||
| Net income effect | 662 | 526 | 456 | ||||
| (USD millions) | 2018 | 2017 | |||
| Novartis share of GSK Consumer Healthcare's estimated current-year consolidated net income | 119 | 589 | |||
| Prior-year adjustment | 4 | 47 | |||
| Amortization of fair value adjustments relating to intangible assets and inventory, net of taxes of USD 1 million (2017: USD 1 million) | – 3 | – 7 | |||
| Pre-tax gain on divestment of GSK Consumer Healthcare | 5 790 | ||||
| Net income effect | 5 910 | 629 | |||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Interest expense | – 714 | – 877 | – 741 | ||||
| Interest expense on lease liabilities | – 66 | ||||||
| Expense arising from discounting long-term liabilities and capitalized borrowing costs | – 70 | – 55 | – 9 | ||||
| Total interest expense from continuing operations | – 850 | – 932 | – 750 | ||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Interest income | 245 | 292 | 110 | ||||
| Other financial income | 12 | 1 | 1 | ||||
| Financial expense | – 52 | – 39 | – 11 | ||||
| Currency result, net | – 160 | – 68 | – 58 | ||||
| Total other financial income and expense from continuing operations | 45 | 186 | 42 | ||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Switzerland | 8 097 | 11 887 | 5 385 | ||||
| Foreign | 843 | 2 208 | 3 717 | ||||
| Income before taxes from continuing operations | 8 940 | 14 095 | 9 102 | ||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Switzerland | – 1 186 | – 615 | – 462 | ||||
| Foreign | – 961 | – 988 | – 1 451 | ||||
| Current income tax expense | – 2 147 | – 1 603 | – 1 913 | ||||
| Switzerland | – 93 | – 120 | – 305 | ||||
| Foreign | 447 | 428 | 615 | ||||
| Deferred tax income | 354 | 308 | 310 | ||||
| Income tax expense from continuing operations | – 1 793 | – 1 295 | – 1 603 | ||||
| (As a percentage) | 2019 | 2018 | 2017 | ||||
| Applicable tax rate | 11.7 | 14.3 | 14.3 | ||||
| Effect of disallowed expenditures | 4.8 | 1.7 | 3.1 | ||||
| Effect of utilization of tax losses brought forward from prior periods | – 0.1 | – 0.1 | – 0.1 | ||||
| Effect of income taxed at reduced rates | – 0.7 | – 0.4 | – 0.2 | ||||
Effect of income not subject to tax 1 | 0.0 | – 3.7 | 0.0 | ||||
| Effect of tax credits and allowances | – 2.3 | – 2.3 | – 2.1 | ||||
| Effect of release of contingent consideration liability | – 0.5 | – 0.2 | – 1.3 | ||||
Effect of tax rate change on current and deferred tax assets and liabilities 2 | – 1.4 | – 0.1 | 4.8 | ||||
Effect of write-off of deferred tax assets 3 | 4.0 | 0.2 | 0.0 | ||||
| Effect of write-down and reversal of write-down of investments in subsidiaries | – 0.6 | 0.0 | – 1.1 | ||||
| Effect of tax benefits expiring in 2017 | 0.0 | 0.0 | – 0.9 | ||||
| Effect of prior-year items | 2.2 | – 0.5 | 1.2 | ||||
Effect of other items 4 | 3.0 | 0.3 | – 0.1 | ||||
| Effective tax rate for continuing operations | 20.1 | 9.2 | 17.6 | ||||
1 Included in 2018 is the effect of income not subject to tax (-3.7%) arising from the portion of the non-taxable gain on the divestment of the Group's investment in GSK Consumer Healthcare Holdings Ltd. attributable to Switzerland. | |||||||
2 2019 is mainly related to the revaluation of the deferred tax assets and liabilities resulting from the tax reforms enacted in Switzerland in 2019, refer to Note 12 for additional disclosures. Included in 2017 is a 4.8% impact related to the revaluation of the deferred tax assets and liabities and a portion of current tax payables. This revaluation resulted from the US tax reform legislation enacted on December 22, 2017, refer to Note 12 for additional disclosures. | |||||||
3 2019 is primarily related to a non-cash, one-time deferred tax expense for the write-off of a deferred tax asset resulting from legal entity reorganizations. | |||||||
4 In 2019, other items (+3.0%) include changes in uncertain tax positions (+2.6%) and other items (+0.4%). | |||||||
| 2019 | 2018 | 2017 | |||||
| Net income attributable to shareholders of Novartis AG (USD millions) | |||||||
| - Continuing operations | 7 142 | 12 797 | 7 499 | ||||
| - Discontinued operations | 4 590 | – 186 | 204 | ||||
| Total | 11 732 | 12 611 | 7 703 | ||||
| Number of shares (in millions) | |||||||
| Weighted average number of shares outstanding used in basic earnings per share | 2 291 | 2 319 | 2 346 | ||||
| Adjustment for vesting of restricted shares, restricted share units and dilutive shares from options | 28 | 25 | 25 | ||||
| Weighted average number of shares in diluted earnings per share | 2 319 | 2 344 | 2 371 | ||||
| Basic earnings per share (USD) | |||||||
| - Continuing operations | 3.12 | 5.52 | 3.20 | ||||
| - Discontinued operations | 2.00 | – 0.08 | 0.08 | ||||
| Total | 5.12 | 5.44 | 3.28 | ||||
| Diluted earnings per share (USD) | |||||||
| - Continuing operations | 3.08 | 5.46 | 3.17 | ||||
| - Discontinued operations | 1.98 | – 0.08 | 0.08 | ||||
| Total | 5.06 | 5.38 | 3.25 | ||||
(USD millions) | Fair value adjustments on marketable securities | Fair value adjustments on debt securities | Fair value adjustments on deferred cash flow hedges | Fair value adjustments on equity securities | Actuarial gains/(losses) from defined benefit plans | Cumulative currency translation effects | Total value adjustments | ||||||||
| Value adjustments at January 1, 2017 | 349 | – 1 | – 3 | – 5 915 | – 1 642 | – 7 212 | |||||||||
| Fair value adjustments on financial instruments | 39 | – 1 | 12 | 50 | |||||||||||
| Net investment hedge | – 237 | – 237 | |||||||||||||
| Net actuarial losses from defined benefit plans | 851 | 851 | |||||||||||||
| Currency translation effects | 2 208 | 2 208 | |||||||||||||
| Total value adjustments in 2017 | 39 | – 1 | 12 | 851 | 1 971 | 2 872 | |||||||||
| Value adjustments at December 31, 2017, as previously reported | 388 | – 2 | 9 | – 5 064 | 329 | – 4 340 | |||||||||
Impact of adoption of IFRS 9 on retained earnings and OCI 1 | – 177 | – 177 | |||||||||||||
Reclassification to presentation required under IFRS 9 1 | – 211 | 211 | |||||||||||||
| Restated value adjustments at January 1, 2018 | – 2 | 9 | 211 | – 5 064 | 329 | – 4 517 | |||||||||
| Fair value adjustments on financial instruments | 12 | 13 | 25 | ||||||||||||
| Fair value adjustments on financial assets sold | – 16 | – 16 | |||||||||||||
| Net investment hedge | 95 | 95 | |||||||||||||
| Net actuarial gains from defined benefit plans | – 359 | – 359 | |||||||||||||
| Currency translation effects | 320 | 320 | |||||||||||||
| Total value adjustments in 2018 | 12 | – 3 | – 359 | 415 | 65 | ||||||||||
| Value adjustments at December 31, 2018 | – 2 | 21 | 208 | – 5 423 | 744 | – 4 452 | |||||||||
| Fair value adjustments on financial instruments | 1 | 1 | – 47 | – 45 | |||||||||||
| Fair value adjustments on financial assets sold | – 95 | – 95 | |||||||||||||
| Net investment hedge | 44 | 44 | |||||||||||||
| Net actuarial gains from defined benefit plans | – 466 | – 466 | |||||||||||||
| Currency translation effects | 354 | 354 | |||||||||||||
| Total value adjustments in 2019 | 1 | 1 | – 142 | – 466 | 398 | – 208 | |||||||||
| Fair value adjustments related to divestments | 33 | – 30 | 3 | ||||||||||||
| Value adjustments at December 31, 2019 | – 1 | 22 | 99 | – 5 919 | 1 142 | – 4 657 | |||||||||
1 Note 1 provides additional disclosures related to the impact of adoption of IFRS 9 Financial Instruments. OCI: other comprehensive income | |||||||||||||||
(USD millions) | Fair value adjustments on equity securities 1 | Fair value adjustments on debt securities | Fair value adjustments on deferred cash flow hedges | Total | |||||||
| Fair value adjustments at January 1, 2019 | 208 | – 2 | 21 | 227 | |||||||
| Changes in fair value: | |||||||||||
| – Debt securities sold | 1 | 1 | |||||||||
| – Equity securities | – 94 | – 94 | |||||||||
| Amortized net losses on cash flow hedges transferred to the consolidated income statement | 1 | 1 | |||||||||
| Deferred tax on above items | 47 | 47 | |||||||||
| Realized net gains reclassified to the retained earnings: | |||||||||||
| – Other financial assets sold | – 95 | – 95 | |||||||||
| Fair value adjustments during the year | – 142 | 1 | 1 | – 140 | |||||||
| Fair value adjustments related to divestments | 33 | 33 | |||||||||
| Fair value adjustments at December 31, 2019 | 99 | – 1 | 22 | 120 | |||||||
1 Includes fair value adjustments on equity securities designated as financial assets valued at fair value through other comprehensive income with no subsequent recycling into the consolidated income statement | |||||||||||
(USD millions) | Fair value adjustments on marketable securities | Fair value adjustments on equity securities 1 | Fair value adjustments on debt securities | Fair value adjustments on deferred cash flow hedges | Total | ||||||
| Fair value adjustments at January 1, 2018, as previously reported | 388 | – 2 | 9 | 395 | |||||||
Impact of adoption of IFRS 9 on retained earnings and other comprehensive income 2 | – 177 | – 177 | |||||||||
| Reclassification to presentation required under IFRS 9 | – 211 | 211 | |||||||||
| Restated fair value adjustments at January 1, 2018 | 211 | – 2 | 9 | 218 | |||||||
| Changes in fair value: | |||||||||||
| – Equity securities | 18 | 18 | |||||||||
| Amortized net losses on cash flow hedges transferred to the consolidated income statement | 13 | 13 | |||||||||
| Deferred tax on above items | – 5 | – 1 | – 6 | ||||||||
| Realized net gains reclassified to the retained earnings: | |||||||||||
| – Other financial assets sold | – 16 | – 16 | |||||||||
| Fair value adjustments during the year | – 3 | 12 | 9 | ||||||||
| Fair value adjustments at December 31, 2018 | 208 | – 2 | 21 | 227 | |||||||
1 Includes fair value adjustments on equity securities designated as financial assets valued at fair value through other comprehensive income with no subsequent recycling into the consolidated income statement | |||||||||||
2 Note 1 provides aditional disclosures on the impact of adoption of IFRS 9 Financial Instruments. | |||||||||||
(USD millions) | Fair value adjustments on marketable securities | Fair value adjustments on debt securities | Fair value adjustments on deferred cash flow hedges | Total | |||||
| Fair value adjustments at January 1, 2017 | 349 | – 1 | – 3 | 345 | |||||
| Changes in fair value: | |||||||||
| – Available-for-sale marketable securities | 12 | – 1 | 11 | ||||||
| – Available-for-sale financial investments | 47 | 47 | |||||||
| Realized net gains transferred to the consolidated income statement: | |||||||||
| – Other financial assets sold | – 109 | – 109 | |||||||
| Amortized net losses on cash flow hedges transferred to the consolidated income statement | 13 | 13 | |||||||
| Impaired financial assets transferred to the consolidated income statement | 102 | 102 | |||||||
Deferred tax on above items 1 | – 13 | – 1 | – 14 | ||||||
| Fair value adjustments during the year | 39 | – 1 | 12 | 50 | |||||
| Fair value adjustments at December 31, 2017 | 388 | – 2 | 9 | 395 | |||||
1 Included is a USD 18 million impact related to the revaluation of deferred tax liabilities on available-for-sale financial investments held in the US that were previously recognized through other comprehensive income related to continuing operrations. This revaluation resulted from the US tax reform legislation enacted on December 22, 2017. Refer to Note 12 for additional disclosures. | |||||||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Defined benefit pension plans before tax | – 119 | – 482 | 1 367 | ||||
| Other post-employment benefit plans before tax | – 35 | 54 | 76 | ||||
Taxation on above items 1 | – 313 | 69 | – 592 | ||||
| Total after tax | – 467 | – 359 | 851 | ||||
| Attributable to: | |||||||
| Shareholders of Novartis AG | – 466 | – 359 | 851 | ||||
| Non-controlling interests | – 1 | ||||||
1 Included in 2019 is a USD -358 million impact related to the revaluation of deferred tax assets on Swiss post-employment benefits that were previously recognized through other comprehensive income. This revaluation resulted from the Swiss tax reforms enacted by the voters in 2019. Refer to Note 12 for additional disclosures. Included in 2017 is a USD -272 million impact related to the revaluation of deferred tax assets on US post-employment benefits that were previously recognized through other comprehensive income (continuing operations USD-259 million and discontinued operations USD -13 million). This revaluation resulted from the US tax reform legislation enacted on December 22, 2017. Refer to Note 12 for additional disclosures. | |||||||
(USD millions) | Land | Buildings | Construction in progress | Machinery and other equipment | Total | ||||||
| Cost | |||||||||||
| January 1, 2019 | 696 | 14 135 | 2 042 | 17 155 | 34 028 | ||||||
Cost of assets related to discontinued operations 1 | – 61 | – 1 615 | – 655 | – 2 678 | – 5 009 | ||||||
Reclassification to right-of-use assets 2 | – 122 | – 3 | – 2 | – 127 | |||||||
Cost of assets related to disposal group held for sale 3 | – 3 | – 12 | – 8 | – 23 | |||||||
| Impact of business combinations | 10 | 24 | 1 | 9 | 44 | ||||||
Reclassifications 4 | 57 | 332 | – 1 019 | 630 | |||||||
Additions 5 | 6 | 112 | 1 001 | 355 | 1 474 | ||||||
Disposals and derecognitions 6 | – 75 | – 1 551 | – 9 | – 1 774 | – 3 409 | ||||||
| Currency translation effects | 1 | 32 | 1 | – 13 | 21 | ||||||
| December 31, 2019 | 512 | 11 463 | 1 350 | 13 674 | 26 999 | ||||||
| Accumulated depreciation | |||||||||||
| January 1, 2019 | – 43 | – 6 328 | – 37 | – 11 924 | – 18 332 | ||||||
Accumulated depreciation on assets related to discontinued operations 1 | 8 | 562 | 7 | 1 541 | 2 118 | ||||||
Reclassification to right-of-use assets 2 | 26 | 26 | |||||||||
Accumulated depreciation on assets related to disposal group held for sale 3 | 2 | 2 | |||||||||
Accumulated depreciation on disposals and derecognitions 6 | 1 170 | 2 | 1 674 | 2 846 | |||||||
Depreciation charge 7 | – 447 | – 898 | – 1 345 | ||||||||
Impairment charge 8 | – 10 | – 51 | – 34 | – 110 | – 205 | ||||||
| Reversal of impairment charge | 1 | 2 | 3 | ||||||||
| Currency translation effects | – 1 | – 33 | – 9 | – 43 | |||||||
| December 31, 2019 | – 20 | – 5 124 | – 60 | – 9 726 | – 14 930 | ||||||
| Net book value at December 31, 2019 | 492 | 6 339 | 1 290 | 3 948 | 12 069 | ||||||
| Commitments for purchases of property, plant and equipment | 220 | ||||||||||
| Capitalized borrowing costs | 4 | ||||||||||
1 Represents the cost of assets and accumulated depreciation at January 1, 2019 related to the Alcon business reported as discontinued operations. Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||||||
2 Reclassification to right-of-use assets at January 1, 2019, upon adoption of IFRS 16 Leases. Refer to Notes 1 and 10 for additional disclosure. | |||||||||||
3 Note 2 provides additional disclosures related to disposal group held for sale. | |||||||||||
4 Reclassifications between various asset categories due to completion of plant and other equipment under construction | |||||||||||
5 Additions in the disposal group held for sale for the period from January 1, 2019, to December 31, 2019 were USD 23 million. | |||||||||||
6 Derecognition of assets that are no longer used and are not considered to have a significant disposal value or other alternative use | |||||||||||
7 No depreciation charge in the disposal group held for sale for the period from January 1, 2019, to December 31, 2019 was recorded. | |||||||||||
8 Impairments in the disposal group held for sale for the period from January 1, 2019, to December 31, 2019 were USD 2 million. | |||||||||||
(USD millions) | Land | Buildings | Construction in progress | Machinery and other equipment | Total | ||||||
| Cost | |||||||||||
| January 1, 2018 | 720 | 14 064 | 2 368 | 16 858 | 34 010 | ||||||
Cost of assets related to disposal group held for sale 1 | – 11 | – 114 | – 24 | – 160 | – 309 | ||||||
| Impact of business combinations | 2 | 40 | 15 | 80 | 137 | ||||||
Reclassifications 2 | 1 | 538 | – 1 470 | 931 | |||||||
Additions 3 | 7 | 110 | 1 250 | 407 | 1 774 | ||||||
Disposals and derecognitions 4 | – 7 | – 212 | – 21 | – 457 | – 697 | ||||||
| Currency translation effects | – 16 | – 291 | – 76 | – 504 | – 887 | ||||||
| December 31, 2018 | 696 | 14 135 | 2 042 | 17 155 | 34 028 | ||||||
| Accumulated depreciation | |||||||||||
| January 1, 2018 | – 40 | – 5 983 | – 38 | – 11 485 | – 17 546 | ||||||
Accumulated depreciation on assets related to disposal group held for sale 1 | 56 | 4 | 101 | 161 | |||||||
Depreciation charge 5 | – 3 | – 574 | – 1 140 | – 1 717 | |||||||
Accumulated depreciation on disposals and derecognitions 4 | 180 | 3 | 412 | 595 | |||||||
| Impairment charge | – 1 | – 122 | – 16 | – 185 | – 324 | ||||||
| Reversal of impairment charge | 8 | 12 | 20 | ||||||||
| Currency translation effects | 1 | 115 | 2 | 361 | 479 | ||||||
| December 31, 2018 | – 43 | – 6 328 | – 37 | – 11 924 | – 18 332 | ||||||
| Net book value at December 31, 2018 | 653 | 7 807 | 2 005 | 5 231 | 15 696 | ||||||
| Net book value of property, plant and equipment under finance lease contracts | 79 | 79 | |||||||||
| Commitments for purchases of property, plant and equipment | 289 | ||||||||||
| Capitalized borrowing costs | 6 | ||||||||||
1 Note 2 provides additional disclosures related to disposal group held for sale. | |||||||||||
2 Reclassifications between various asset categories due to completion of plant and other equipment under construction | |||||||||||
3 Additions in the disposal group held for sale were USD 21 million. | |||||||||||
4 Derecognition of assets that are no longer used and are not considered to have a significant disposal value or other alternative use | |||||||||||
5 Depreciation charge in the disposal group held for sale for the period from January 1, 2018, to the date of reclassification to assets held for sale was USD 15 million. For depreciation charge related to discontinued operations, refer to Note 30. | |||||||||||
| (USD millions) | |||
Operating lease commitments December 31, 2018 1 | 3 612 | ||
| Operating lease commitments December 31, 2018 related to discontinued operations | – 222 | ||
| Operating lease commitments December 31, 2018 related to continuing operations | 3 390 | ||
| Recognition exemption for short-term leases | – 30 | ||
| Recognition exception for low-value leases | – 12 | ||
| Lease arrangements with commencement date after December 31, 2018 | – 65 | ||
| Undiscounted future lease payments continuing operations as of January 1, 2019 | 3 283 | ||
| Effect of discounting | – 1 547 | ||
Lease liabilities recognized as of January 1, 2019 2 | 1 736 | ||
1 As reported in Annual Report 2018 Note 27 | |||
2 The weighted average incremental borrowing rate at January 1, 2019, the date of implementation of IFRS 16 Leases, was 3.5%. | |||
| (USD millions) | January 1, 2019 | ||
| Land | 536 | ||
| Buildings | 848 | ||
| Vehicles | 147 | ||
| Machinery and equipment, and other assets | 23 | ||
Right-of-use assets 1 | 1 554 | ||
1 Right-of-use assets were lower than the lease liability at the date of implementation of IFRS 16 by USD 182 million, due to adjustments made for recognition of sublease receivables, prepayments and accrued lease payments and transfers from leased assets recorded in property, plant and equipment at December 31, 2018. | |||
| (USD millions) | January 1, 2019 | ||
| Assets | |||
| Non-current assets | |||
| Property, plant and equipment | – 101 | ||
| Right-of-use assets | 1 554 | ||
| Other non-current assets | 74 | ||
| Total non-current assets | 1 527 | ||
| Total assets | 1 527 | ||
| Equity and liabilities | |||
| Equity | |||
| Reserves | 3 | ||
| Total equity | 3 | ||
| Liabilities | |||
| Non-current liabilities | |||
| Financial debts | – 2 | ||
| Lease liabilities | 1 471 | ||
| Provision and other non-current liabilities | – 212 | ||
| Total non-current liabilities | 1 257 | ||
| Current liabilities | |||
| Financial debts and derivative financial instruments | – 1 | ||
| Lease liabilities | 268 | ||
| Total current liabilities | 267 | ||
| Total liabilities | 1 524 | ||
| Total equity and liabilities | 1 527 | ||
| (USD millions) | |||
| Right-of-use assets at January 1, 2019 | 1 554 | ||
| Additions | 537 | ||
| Depreciation charge | – 305 | ||
Lease contract terminations 1 | – 98 | ||
| Impact of divestments | – 17 | ||
| Currency translation effects | 6 | ||
| Total right-of-use assets at December 31, 2019 | 1 677 | ||
No impairments were recorded in the period. | |||
1 Lease contract terminations also includes modifications to existing leases that result in reductions to the right-of-use assets, and reductions due to sub-leasing. | |||
(USD millions) | December 31, 2019 carrying value | Depreciation charge 2019 | |||
| Land | 537 | 14 | |||
| Buildings | 990 | 194 | |||
| Vehicles | 129 | 87 | |||
| Machinery and equipment, and other assets | 21 | 10 | |||
| Total right-of-use assets | 1 677 | 305 | |||
(USD millions) | Lease liabilities 2019 | Lease liabilities undiscounted 2019 | ||
| Less than one year | 246 | 295 | ||
| Between one and two years | 202 | 246 | ||
| Between two and three years | 163 | 202 | ||
| Between three and four years | 138 | 173 | ||
| Between four and five years | 119 | 150 | ||
| After five years | 1 081 | 2 419 | ||
| Total lease liabilities | 1 949 | 3 485 | ||
| Less current portion of lease liabilities | – 246 | – 295 | ||
| Non-current portion of lease liabilities | 1 703 | 3 190 | ||
| (USD millions) | 2019 | ||
Interest expense on lease liabilities 1 | 66 | ||
| Expense on short-term leases | 7 | ||
| Expense on low-value leases | 8 | ||
| Total cash outflows for leases | 339 | ||
| Thereof: | |||
Cash outflows for short-term leases and low-value leases 2 | 15 | ||
Payments of interest 3 | 51 | ||
Payments of lease liabilities 4 | 273 | ||
1 The weighted average interest rate is 3.9%. | |||
2 Cash flows from short-term and low-value leases are included within total net cash flows from operating activities. The portfolio of short-term leases to which the Group is committed to at December 31, 2019, is similar to the portfolio of short-term leases the Group entered into during 2019. | |||
3 Included within total net cash flows from operating activities. | |||
4 Reported as cash outflows used in financing activities net of lease incentives received of USD 33 million. | |||
| Goodwill | Intangible assets other than goodwill | ||||||||||||||||
(USD millions) | Total | In-process research and development | Alcon brand name | Technologies | Currently marketed products | Marketing know-how | Other intangible assets | Total | |||||||||
| Cost | |||||||||||||||||
| January 1, 2019 | 35 700 | 16 167 | 2 980 | 6 253 | 35 412 | 5 960 | 2 253 | 69 025 | |||||||||
Cost of assets related to discontinued operations 1 | – 9 000 | – 249 | – 2 980 | – 5 369 | – 4 440 | – 5 960 | – 572 | – 19 570 | |||||||||
Cost of assets related to disposal group held for sale, net 2 | – 1 | 4 | 3 | ||||||||||||||
| Impact of business combinations | 186 | 342 | 3 550 | 22 | 3 914 | ||||||||||||
Reclassifications 3 | – 9 069 | 9 069 | |||||||||||||||
Additions 4 | 265 | 243 | 259 | 767 | |||||||||||||
Disposals and derecognitions 5 | – 75 | – 544 | – 436 | – 1 055 | |||||||||||||
| Currency translation effects | – 61 | 49 | 254 | 32 | 335 | ||||||||||||
| December 31, 2019 | 26 825 | 7 429 | 884 | 43 548 | 1 558 | 53 419 | |||||||||||
| Accumulated amortization | |||||||||||||||||
| January 1, 2019 | – 406 | – 1 120 | – 4 758 | – 21 218 | – 1 906 | – 1 304 | – 30 306 | ||||||||||
Accumulated amortization/impairments on assets related to discontinued operations 1 | 101 | 3 | 4 184 | 2 592 | 1 906 | 128 | 8 813 | ||||||||||
Amortization charge 6 | – 42 | – 2 657 | – 137 | – 2 836 | |||||||||||||
Accumulated impairments on disposals and derecognitions 5 | 70 | 494 | 419 | 983 | |||||||||||||
Impairment charge 6 | – 984 | – 105 | – 54 | – 32 | – 1 175 | ||||||||||||
| Reversal of impairment charge | 37 | 37 | |||||||||||||||
| Currency translation effects | 4 | – 11 | – 126 | – 11 | – 148 | ||||||||||||
| December 31, 2019 | – 301 | – 2 005 | – 721 | – 20 969 | – 937 | – 24 632 | |||||||||||
| Net book value at December 31, 2019 | 26 524 | 5 424 | 163 | 22 579 | 621 | 28 787 | |||||||||||
1 Represents the cost of assets and accumulated amortization at January 1, 2019, related to the Alcon business reported as discontinued operations. Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||||||||||||
2 Note 2 provides additional disclosures related to assets of disposal group held for sale. | |||||||||||||||||
3 Reclassifications between various asset categories as a result of product launches of acquired in-process research and development, and completion of software development | |||||||||||||||||
4 No addition in the disposal group held for sale for the period from January 1, 2019 to December 31, 2019 | |||||||||||||||||
5 Derecognitions of assets that are no longer used or being developed and are not considered to have a significant disposal value or other alternative use | |||||||||||||||||
6 No amortization or impairment charges related to the disposal group held for sale for the period from January 1, 2019, to December 31, 2019. | |||||||||||||||||
| Goodwill | Intangible assets other than goodwill | ||||||||||||||||
(USD millions) | Total | In-process research and development | Alcon brand name | Technologies | Currently marketed products | Marketing know-how | Other intangible assets | Total | |||||||||
| Cost | |||||||||||||||||
| January 1, 2018 | 32 179 | 6 462 | 2 980 | 6 638 | 34 105 | 5 960 | 1 852 | 57 997 | |||||||||
Cost of assets related to disposal group held for sale 1 | – 9 | – 276 | – 1 116 | – 2 | – 1 403 | ||||||||||||
| Impact of business combinations | 4 084 | 10 224 | 2 531 | 1 | 12 756 | ||||||||||||
Reclassifications 2 | – 697 | 479 | 218 | ||||||||||||||
Additions 3 | 477 | 2 | 728 | 385 | 1 592 | ||||||||||||
Disposals and derecognitions 4 | – 214 | – 70 | – 928 | – 183 | – 1 395 | ||||||||||||
Impairment charge 5 | – 183 | ||||||||||||||||
| Currency translation effects | – 380 | – 76 | – 41 | – 387 | – 18 | – 522 | |||||||||||
| December 31, 2018 | 35 700 | 16 167 | 2 980 | 6 253 | 35 412 | 5 960 | 2 253 | 69 025 | |||||||||
| Accumulated amortization | |||||||||||||||||
| January 1, 2018 | – 429 | – 1 170 | – 4 268 | – 19 631 | – 1 668 | – 1 263 | – 28 000 | ||||||||||
Accumulated amortization/impairments on assets related to disposal group held for sale 1 | 2 | 107 | 816 | 925 | |||||||||||||
Amortization charge 6 | – 570 | – 2 521 | – 238 | – 310 | – 3 639 | ||||||||||||
Accumulated impairments on disposals and derecognitions4 | 209 | 791 | 257 | 1 257 | |||||||||||||
Impairment charge 5 | – 167 | – 53 | – 825 | – 4 | – 1 049 | ||||||||||||
| Currency translation effects | 23 | 6 | 26 | 152 | 16 | 200 | |||||||||||
| December 31, 2018 | – 406 | – 1 120 | – 4 758 | – 21 218 | – 1 906 | – 1 304 | – 30 306 | ||||||||||
| Net book value at December 31, 2018 | 35 294 | 15 047 | 2 980 | 1 495 | 14 194 | 4 054 | 949 | 38 719 | |||||||||
1 Note 2 provides additional disclosures related to assets of disposal group held for sale. | |||||||||||||||||
2 Reclassifications between various asset categories as a result of product launches of acquired in-process research and development, and completion of software development | |||||||||||||||||
3 No addition in the disposal group held for sale for the period from January 1, 2018, to the date of reclassification to assets held for sale | |||||||||||||||||
4 Derecognitions of assets that are no longer used or being developed and are not considered to have a significant disposal value or other alternative use | |||||||||||||||||
5 Impairment charges related to the disposal group held for sale for the write-down of the allocated goodwill were USD 183 million and for the currently marketed products were USD 37 million (thereof USD 9 million recognized for the period from January 1, 2018, to the date of reclassification to assets held for sale). For amortization related to discontinued operations, refer to Note 30. | |||||||||||||||||
6 Amortization related to the disposal group held for sale for the period from January 1, 2018, to the date of reclassification to assets held for sale was USD 45 million. | |||||||||||||||||
| Goodwill | Intangible assets other than goodwill | ||||||||||||
(USD millions) | Total | In-process research and development | Technologies | Currently marketed products | Other intangible assets | Total | |||||||
| Innovative Medicines | 18 750 | 5 339 | 7 | 21 720 | 520 | 27 586 | |||||||
| Sandoz | 7 767 | 85 | 156 | 859 | 25 | 1 125 | |||||||
| Corporate | 7 | 76 | 76 | ||||||||||
| Net book value at December 31, 2019 | 26 524 | 5 424 | 163 | 22 579 | 621 | 28 787 | |||||||
| Goodwill | Intangible assets other than goodwill | ||||||||||||||||
(USD millions) | Total | In-process research and development | Alcon brand name | Technologies | Currently marketed products | Marketing know-how | Other intangible assets | Total | |||||||||
| Innovative Medicines | 18 551 | 14 377 | 6 | 11 228 | 431 | 26 042 | |||||||||||
| Sandoz | 7 837 | 419 | 304 | 1 115 | 37 | 1 875 | |||||||||||
Alcon 1 | 8 899 | 246 | 2 980 | 1 185 | 1 851 | 4 054 | 363 | 10 679 | |||||||||
| Corporate | 7 | 5 | 118 | 123 | |||||||||||||
| Net book value at December 31, 2018 | 35 294 | 15 047 | 2 980 | 1 495 | 14 194 | 4 054 | 949 | 38 719 | |||||||||
1 From February 28, 2019, the Alcon Division was reported as discontinued operations (see Notes 1, 2 and 30). In accordance with IFRS, the December 31, 2018, consolidated balance sheet includes the assets and liabilities of the Alcon eye care devices business and certain Corporate assets and liabilities attributable to the Alcon business. Note 30 provides additional information on discontinued operations. | |||||||||||||||||
(As a percentage) | Innovative Medicines | Sandoz | |||
| Terminal growth rate | 1.5 | 2.0 | |||
| Discount rate (post-tax) | 6.5 | 6.5 | |||
| (USD millions) | 2019 | 2018 | 2017 | ||||
Innovative Medicines 1 | – 669 | – 592 | – 591 | ||||
Sandoz 2 | – 506 | – 249 | – 61 | ||||
| Total | – 1 175 | – 841 | – 652 | ||||
1 2019 includes an impairment of USD 416 million related to the write-down of IPR&D acquired through the 2015 Spinifex Pharmaceuticals Inc. acquisition and USD 108 million write-down related to cessation of clinical development program MOR106 for atopic dermatitis. 2018 includes an impairment of USD 400 million related to a partial write-down of theVotrient currently marketed product. 2017 includes an impairment of USD 465 million related to the write-down of the Serelaxin IPR&D. | |||||||
2 2019 includes impairment of USD 442 million related to the write-down of IPR&D related to the discontinuation of the generic Advair® development program. 2018 includes impairments of USD 220 million related to the write-down of the allocated goodwill (USD 183 million) and the currently marketed products (USD 37 million) related to the pending divestment of the Sandoz US dermatology business and generic US oral solids portfolio. See Note 2. | |||||||
(USD millions) | Property, plant and equipment | Intangible assets | Pensions and other benefit obligations of associates | Inventories | Tax loss carry- forwards | Other assets, provisions and accruals | Total | ||||||||
| Gross deferred tax assets at January 1, 2019 | 191 | 1 233 | 1 188 | 3 722 | 273 | 2 175 | 8 782 | ||||||||
| Gross deferred tax liabilities at January 1, 2019 | – 622 | – 5 384 | – 273 | – 474 | – 805 | – 7 558 | |||||||||
| Net deferred tax balance at January 1, 2019 | – 431 | – 4 151 | 915 | 3 248 | 273 | 1 370 | 1 224 | ||||||||
| At January 1, 2019 | – 431 | – 4 151 | 915 | 3 248 | 273 | 1 370 | 1 224 | ||||||||
Net deferred tax balance related to discontinued operations 1 | 82 | 1 403 | – 123 | – 248 | – 39 | – 217 | 858 | ||||||||
| Credited/(charged) to income | 74 | 605 | 308 | – 818 | – 113 | 298 | 354 | ||||||||
| Charged to equity | 8 | 75 | – 166 | – 83 | |||||||||||
| Charged to other comprehensive income | – 313 | 24 | – 289 | ||||||||||||
| Impact of business combinations | 3 | – 45 | 21 | – 26 | – 47 | ||||||||||
Other movements 2 | – 10 | 39 | – 23 | 31 | – 12 | 25 | |||||||||
| Net deferred tax balance at December 31, 2019 | – 282 | – 2 141 | 787 | 2 159 | 248 | 1 271 | 2 042 | ||||||||
| Gross deferred tax assets at December 31, 2019 | 108 | 1 469 | 1 078 | 2 446 | 255 | 2 596 | 7 952 | ||||||||
| Gross deferred tax liabilities at December 31, 2019 | – 390 | – 3 610 | – 291 | – 287 | – 7 | – 1 325 | – 5 910 | ||||||||
| Net deferred tax balance at December 31, 2019 | – 282 | – 2 141 | 787 | 2 159 | 248 | 1 271 | 2 042 | ||||||||
| After offsetting the following amount of deferred tax assets and liabilities within the same tax jurisdiction, the balance amounts to: | 43 | ||||||||||||||
| Deferred tax assets at December 31, 2019 | 7 909 | ||||||||||||||
| Deferred tax liabilities at December 31, 2019 | – 5 867 | ||||||||||||||
| Net deferred tax balance at December 31, 2019 | 2 042 | ||||||||||||||
| Gross deferred tax assets at January 1, 2018 | 137 | 1 287 | 1 090 | 3 786 | 97 | 1 983 | 8 380 | ||||||||
| Gross deferred tax liabilities at January 1, 2018 | – 613 | – 2 985 | – 254 | – 455 | – 9 | – 1 003 | – 5 319 | ||||||||
| Net deferred tax balance at January 1, 2018 | – 476 | – 1 698 | 836 | 3 331 | 88 | 980 | 3 061 | ||||||||
| At January 1, 2018 | – 476 | – 1 698 | 836 | 3 331 | 88 | 980 | 3 061 | ||||||||
| Net deferred tax balance related to disposal group held for sale | 1 | 1 | – 6 | – 1 | – 1 | – 6 | |||||||||
| Credited/(charged) to income | 31 | 378 | 4 | – 86 | – 113 | 368 | 582 | ||||||||
| Charged to equity | – 17 | – 17 | |||||||||||||
| Charged to other comprehensive income | 69 | 8 | 77 | ||||||||||||
| Impact of business combinations | – 2 874 | 298 | 83 | – 2 493 | |||||||||||
| Other movements | 13 | 42 | 6 | 9 | 1 | – 51 | 20 | ||||||||
| Net deferred tax balance at December 31, 2018 | – 431 | – 4 151 | 915 | 3 248 | 273 | 1 370 | 1 224 | ||||||||
| Gross deferred tax assets at December 31, 2018 | 191 | 1 233 | 1 188 | 3 722 | 273 | 2 175 | 8 782 | ||||||||
| Gross deferred tax liabilities at December 31, 2018 | – 622 | – 5 384 | – 273 | – 474 | – 805 | – 7 558 | |||||||||
| Net deferred tax balance at December 31, 2018 | – 431 | – 4 151 | 915 | 3 248 | 273 | 1 370 | 1 224 | ||||||||
| After offsetting the following amount of deferred tax assets and liabilities within the same tax jurisdiction, the balance amounts to: | 83 | ||||||||||||||
| Deferred tax assets at December 31, 2018 | 8 699 | ||||||||||||||
| Deferred tax liabilities at December 31, 2018 | – 7 475 | ||||||||||||||
| Net deferred tax balance at December 31, 2018 | 1 224 | ||||||||||||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||||||||||
2 Includes USD 3 million net deferred tax movement related to disposal group held for sale | |||||||||||||||
| (USD billions) | 2019 | 2018 | |||
| Expected to have an impact on current tax payable after more than 12 months | |||||
| – Deferred tax assets | 4.3 | 3.9 | |||
| – Deferred tax liabilities | 5.2 | 6.7 | |||
| (USD billions) | 2019 | 2018 | |||
| Unremitted earnings that have been retained by consolidated entities for reinvestment | 61 | 73 | |||
| (USD billions) | 2019 | 2018 | |||
| Investments in subsidiaries | 3 | 3 | |||
| Goodwill from acquisitions | – 24 | – 33 | |||
| (USD millions) | Not capitalized | Capitalized | 2019 total | ||||
| One year | 14 | 0 | 14 | ||||
| Two years | 28 | 0 | 28 | ||||
| Three years | 28 | 6 | 34 | ||||
| Four years | 16 | 46 | 62 | ||||
| Five years | 127 | 37 | 164 | ||||
| More than five years | 435 | 2 249 | 2 684 | ||||
| Total | 648 | 2 338 | 2 986 | ||||
| (USD millions) | Not capitalized | Capitalized | 2018 total | ||||
| One year | 23 | 4 | 27 | ||||
| Two years | 14 | 0 | 14 | ||||
| Three years | 27 | 12 | 39 | ||||
| Four years | 65 | 5 | 70 | ||||
| Five years | 345 | 36 | 381 | ||||
| More than five years | 522 | 2 288 | 2 810 | ||||
| Total | 996 | 2 345 | 3 341 | ||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Tax losses carried forward that expired | 9 | 8 | 1 | ||||
(USD millions) | Income statement continuing operations | Equity | Total | ||||
| Deferred tax asset and liability revaluation | |||||||
| Items previously recognized in consolidated income statement | 234 | 234 | |||||
Items previously recognized in other comprehensive income 1 | – 358 | – 358 | |||||
| Total revaluation of deferred tax assets and liabilities | 234 | – 358 | – 124 | ||||
1 Related to post-employment benefits | |||||||
(USD millions) | Income statement continuing operations | Equity | Total | ||||
| Deferred tax asset and liability revaluation | |||||||
Items previously recognized in consolidated income statement 1 | – 440 | – 440 | |||||
Items previously recognized in other comprehensive income 2 | – 254 | – 254 | |||||
Items previously recognized in retained earnings 3 | – 71 | – 71 | |||||
| Total revaluation of deferred tax assets and liabilities | – 440 | – 325 | – 765 | ||||
Total revaluation of current tax payables 4 | – 34 | – 34 | |||||
| Total revaluation of deferred tax assets and liabilities, and current income tax liabilities | – 474 | – 325 | – 799 | ||||
1 Items previously recognized in discontinued operations amounted to USD 416 million | |||||||
2 Related to post-employment benefits and available-for-sale financial investments (attributable to continuing operations USD -241 million and attributable to discontinued operations USD -13 million) | |||||||
3 Related to equity-based compensation plans (attributable to continuing operations USD -66 million and attributable to discontinued operations USD -5 million) | |||||||
4 Revaluation of current tax payable attributable to discontinued operations amounted to USD -3 million. | |||||||
| (USD millions) | 2019 | 2018 | |||
| Equity securities | 1 524 | 1 155 | |||
| Debt securities | 33 | 31 | |||
| Fund investments | 233 | 251 | |||
| Total financial investments | 1 790 | 1 437 | |||
| Long-term receivables from customers | 164 | ||||
Minimum lease payments from finance lease agreements 1 | 91 | ||||
| Long-term receivables from finance subleases | 66 | ||||
| Other long-term receivables | 104 | 3 | |||
Contingent consideration receivables 2 | 399 | 396 | |||
| Long-term loans, advances and security deposits | 159 | 254 | |||
| Total financial assets | 2 518 | 2 345 | |||
1 Note 30 provides additional disclosures on minimal lease payments from finance lease agreements that relate to discontinued operations. | |||||
2 Note 29 provides additional disclosures related to contingent considerations. | |||||
| (USD millions) | 2019 | 2018 | |||
| Deferred compensation plans | 414 | 468 | |||
| Prepaid post-employment benefit plans | 148 | 137 | |||
| Other non-current assets | 176 | 290 | |||
| Total other non-current assets | 738 | 895 | |||
| (USD millions) | 2019 | 2018 | |||
| Raw material, consumables | 751 | 931 | |||
| Work in progress | 3 024 | 3 087 | |||
| Finished products | 2 207 | 2 938 | |||
| Total inventories | 5 982 | 6 956 | |||
| (USD billions) | 2019 | 2018 | 2017 | ||||
| Cost of goods sold | – 8.5 | – 8.3 | – 8.2 | ||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Inventory provisions | – 752 | – 603 | – 416 | ||||
| Reversals of inventory provisions | 218 | 216 | 172 | ||||
| (USD millions) | 2019 | 2018 | |||
| Total gross trade receivables | 8 396 | 8 853 | |||
| Provisions for doubtful trade receivables | – 95 | – 126 | |||
| Total trade receivables, net | 8 301 | 8 727 | |||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| January 1 | – 126 | – 190 | – 162 | ||||
Provisions related to discontinued operations 1 | 54 | ||||||
| Impact of divestments | 12 | ||||||
| Impact of business combination | – 1 | ||||||
Provisions for doubtful trade receivables charged to the consolidated income statement 2 | – 89 | – 47 | – 119 | ||||
| Utilization of provisions for doubtful trade receivables | 12 | 39 | 12 | ||||
Reversal of provisions for doubtful trade receivables credited to the consolidated income statement 3 | 53 | 61 | 76 | ||||
| Currency translation effects | 1 | 12 | – 9 | ||||
| December 31 | – 95 | – 126 | – 190 | ||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||
2 Provisions charged to the consolidated income statement from continuing operations were USD 30 million in 2018 and USD 94 million in 2017. | |||||||
3 Reversal of provisions credited to the consolidated income statement from continuing operations were USD 44 million in 2018 and USD 60 million in 2017. | |||||||
| (USD millions) | 2019 | 2018 | |||
| Not overdue | 7 763 | 7 916 | |||
| Past due for not more than one month | 161 | 296 | |||
| Past due for more than one month but less than three months | 123 | 194 | |||
| Past due for more than three months but less than six months | 103 | 136 | |||
| Past due for more than six months but less than one year | 96 | 98 | |||
| Past due for more than one year | 150 | 213 | |||
| Provisions for doubtful trade receivables | – 95 | – 126 | |||
| Total trade receivables, net | 8 301 | 8 727 | |||
| (USD millions) | 2019 | 2018 | |||
| Total balance of gross trade receivables from closely monitored countries | 1 588 | 1 729 | |||
| Past due for more than one year | 61 | 97 | |||
| Provisions | 24 | 44 | |||
| (USD millions) | 2019 | 2018 | |||
| US dollar (USD) | 3 466 | 3 510 | |||
| Euro (EUR) | 1 384 | 1 551 | |||
| Japanese yen (JPY) | 466 | 658 | |||
| Russian ruble (RUB) | 341 | 247 | |||
| Chinese yuan (CNY) | 279 | 282 | |||
| British pound (GBP) | 202 | 183 | |||
| Brazilian real (BRL) | 165 | 206 | |||
| Canadian dollar (CAD) | 129 | 136 | |||
| Australian dollar (AUD) | 125 | 161 | |||
| Swiss franc (CHF) | 89 | 100 | |||
| Other currencies | 1 655 | 1 693 | |||
| Total trade receivables, net | 8 301 | 8 727 | |||
| (USD millions) | 2019 | 2018 | |||
| Debt securities | 24 | 325 | |||
| Fund investments | 37 | 35 | |||
| Total marketable securities | 61 | 360 | |||
| Commodities | 110 | 104 | |||
| Time deposits and short-term investments with original maturity more than 90 days | 61 | 2 087 | |||
| Derivative financial instruments | 102 | 130 | |||
| Accrued interest on debt securities, time deposits and short-term investments | 12 | ||||
| Total marketable securities, commodities, time deposits and derivative financial instruments | 334 | 2 693 | |||
| (USD millions) | 2019 | 2018 | |||
| US dollar (USD) | 302 | ||||
| Euro (EUR) | 13 | 12 | |||
| Japanese yen (JPY) | 11 | 11 | |||
| Total debt securities | 24 | 325 | |||
| (USD millions) | 2019 | 2018 | |||
| Current accounts | 3 247 | 3 121 | |||
| Time deposits and short-term investments with original maturity less than 90 days | 7 865 | 10 150 | |||
| Total cash and cash equivalents | 11 112 | 13 271 | |||
| (USD millions) | 2019 | 2018 | |||
| VAT receivable | 508 | 588 | |||
| Withholding tax recoverable | 108 | 99 | |||
| Prepaid expenses | |||||
| – Third parties | 898 | 811 | |||
| – Associated companies | 1 | ||||
| Receivables from associated companies | 1 | 2 | |||
| Other receivables and current assets | 1 165 | 1 360 | |||
| Total other current assets | 2 680 | 2 861 | |||
(USD millions) | Jan 1, 2017 | Movement in year | Dec 31, 2017 | Movement in year | Dec 31, 2018 | Movement in year | Dec 31, 2019 | ||||||||
| Share capital | 972 | – 3 | 969 | – 25 | 944 | – 8 | 936 | ||||||||
| Treasury shares | – 76 | – 24 | – 100 | 31 | – 69 | – 11 | – 80 | ||||||||
| Outstanding share capital | 896 | – 27 | 869 | 6 | 875 | – 19 | 856 | ||||||||
| 2019 | 2018 | 2017 | |||||||||||||||||||
Number of outstanding shares (in millions) | Note | Total Novartis shares | Total treasury shares 1 | Total outstanding shares | Total Novartis shares | Total treasury shares 1 | Total outstanding shares | Total Novartis shares | Total treasury shares 1 | Total outstanding shares | |||||||||||
| Balance at beginning of year | 2 550.6 | – 239.4 | 2 311.2 | 2 616.8 | – 299.3 | 2 317.5 | 2 627.1 | – 253.0 | 2 374.1 | ||||||||||||
Shares canceled for capital reduction 2 | – 23.3 | 23.3 | – 66.2 | 66.2 | – 10.3 | 10.3 | |||||||||||||||
Shares acquired to be canceled 3 | – 60.3 | – 60.3 | – 23.3 | – 23.3 | – 66.2 | – 66.2 | |||||||||||||||
Other share purchases 4 | – 1.7 | – 1.7 | – 1.2 | – 1.2 | – 3.8 | – 3.8 | |||||||||||||||
Exercise of options and employee transactions 5 | 18.8 | 5.5 | 5.5 | 7.8 | 7.8 | 4.6 | 4.6 | ||||||||||||||
Equity-based compensation 5 | 9.4 | 9.4 | 7.4 | 7.4 | 8.8 | 8.8 | |||||||||||||||
| Shares delivered to Alcon employees | 0.9 | 0.9 | |||||||||||||||||||
| Other share sales | 3.0 | 3.0 | |||||||||||||||||||
| Total movements | – 23.3 | – 22.9 | – 46.2 | – 66.2 | 59.9 | – 6.3 | – 10.3 | – 46.3 | – 56.6 | ||||||||||||
| Balance at end of year | 2 527.3 | – 262.3 | 2 265.0 | 2 550.6 | – 239.4 | 2 311.2 | 2 616.8 | – 299.3 | 2 317.5 | ||||||||||||
1 Approximately 117.6 million treasury shares (2018: 121.6 million; 2017: 131.3 million) are held in Novartis entities that restrict their availability for use. | |||||||||||||||||||||
2 Novartis reduced its share capital by canceling shares that were repurchased on the SIX Swiss Exchange second trading line during previous years. | |||||||||||||||||||||
3 Shares repurchased on the SIX Swiss Exchange second trading line under a CHF 10 billion share buyback authority approved at the 2016 Annual General Meeting (AGM) for transactions before February 28, 2019, and under a new CHF 10 billion share buyback authority approved at the 2019 AGM for transactions after such date | |||||||||||||||||||||
4 Shares acquired from employees, which were previously granted to them under the respective programs | |||||||||||||||||||||
5 Shares delivered as a result of options being exercised and physical share deliveries related to equity-based participation plans | |||||||||||||||||||||
| 2019 | 2018 | 2017 | |||||
| Dividend per share (in CHF) | 2.85 | 2.80 | 2.75 | ||||
| Total dividend payment (in USD billion) | 6.6 | 7.0 | 6.5 | ||||
| 2019 | 2018 | 2017 | |||||||||||||
Note | Number of outstanding shares (in millions) | Equity impact USD m | Number of outstanding shares (in millions) | Equity impact USD m | Number of outstanding shares (in millions) | Equity impact USD m | |||||||||
Shares acquired to be canceled 1 | – 60.3 | – 5 351 | – 23.3 | – 1 859 | – 66.2 | – 5 270 | |||||||||
Other share purchases 2 | – 1.7 | – 160 | – 1.2 | – 114 | – 3.8 | – 304 | |||||||||
| Purchase of treasury shares | – 62.0 | – 5 511 | – 24.5 | – 1 973 | – 70.0 | – 5 574 | |||||||||
Exercise of options and employee transactions 3 | 18.8 | 5.5 | 210 | 7.8 | 434 | 4.6 | 255 | ||||||||
Equity-based compensation 4,5 | 9.4 | 833 | 7.4 | 756 | 8.8 | 612 | |||||||||
| Shares delivered to Alcon employees | 0.9 | 18 | |||||||||||||
| Other share sales | 3.0 | 263 | |||||||||||||
| Total | – 46.2 | – 4 450 | – 6.3 | – 520 | – 56.6 | – 4 707 | |||||||||
1 Shares repurchased on the SIX Swiss Exchange second trading line under a CHF 10 billion share buyback authority approved at the 2016 AGM for transactions before February 28, 2019, and under a new CHF 10 billion share buyback authority approved at the 2019 AGM for transactions after such date | |||||||||||||||
2 Shares acquired from employees, which were previously granted to them under the respective programs | |||||||||||||||
3 Shares delivered as a result of options being exercised related to equity-based participation plans and the delivery of treasury shares. The average share price of the shares delivered was significantly below market price, reflecting the strike price of the options exercised. | |||||||||||||||
4 Equity-settled share-based compensation is expensed in the consolidated income statement in accordance with the vesting period of the share-based compensation plans. The value for the shares and options granted is credited to consolidated equity over the respective vesting period. In addition, tax benefits arising from tax-deductible amounts exceeding the expense recognized in the income statement are credited to equity. | |||||||||||||||
5 Included in 2017 is a USD 71 million impact related to the revaluation of deferred tax assets on equity-based compensation that were previously recognized through retained earnings. This revaluation resulted from the US tax reform legislation enacted on December 22, 2017. Refer to Note 12 for additional disclosures. | |||||||||||||||
| (USD millions) | 2019 | 2018 | |||
| Straight bonds | 22 167 | 25 283 | |||
Liabilities to banks and other financial institutions 1 | 188 | 285 | |||
| Finance lease obligations | 92 | ||||
| Total, including current portion of non-current financial debt | 22 355 | 25 660 | |||
| Less current portion of non-current financial debt | – 2 002 | – 3 190 | |||
| Total non-current financial debt | 20 353 | 22 470 | |||
1 Average interest rate 0.2% (2018: 0.3%) | |||||
Coupon | Currency | Nominal amount | Issuance year | Maturity year | Issuer | Issue price | 2019 (USD millions) | 2018 (USD millions) | |||||||||
| 5.125% | USD | 3 000 | 2009 | 2019 | Novartis Securities Investment Ltd., Hamilton, Bermuda | 99.822% | 3 000 | ||||||||||
| 4.400% | USD | 1 000 | 2010 | 2020 | Novartis Capital Corporation, New York, United States | 99.237% | 1 000 | 998 | |||||||||
| 2.400% | USD | 1 500 | 2012 | 2022 | Novartis Capital Corporation, New York, United States | 99.225% | 1 495 | 1 493 | |||||||||
| 3.700% | USD | 500 | 2012 | 2042 | Novartis Capital Corporation, New York, United States | 98.325% | 489 | 489 | |||||||||
| 3.400% | USD | 2 150 | 2014 | 2024 | Novartis Capital Corporation, New York, United States | 99.287% | 2 139 | 2 137 | |||||||||
| 4.400% | USD | 1 850 | 2014 | 2044 | Novartis Capital Corporation, New York, United States | 99.196% | 1 825 | 1 825 | |||||||||
| 0.750% | EUR | 600 | 2014 | 2021 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.134% | 670 | 683 | |||||||||
| 1.625% | EUR | 600 | 2014 | 2026 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.697% | 670 | 684 | |||||||||
| 0.250% | CHF | 500 | 2015 | 2025 | Novartis AG, Basel, Switzerland | 100.640% | 517 | 508 | |||||||||
| 0.625% | CHF | 550 | 2015 | 2029 | Novartis AG, Basel, Switzerland | 100.502% | 568 | 558 | |||||||||
| 1.050% | CHF | 325 | 2015 | 2035 | Novartis AG, Basel, Switzerland | 100.479% | 336 | 330 | |||||||||
| 3.000% | USD | 1 750 | 2015 | 2025 | Novartis Capital Corporation, New York, United States | 99.010% | 1 735 | 1 732 | |||||||||
| 4.000% | USD | 1 250 | 2015 | 2045 | Novartis Capital Corporation, New York, United States | 98.029% | 1 219 | 1 219 | |||||||||
| 0.125% | EUR | 1 250 | 2016 | 2023 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.127% | 1 392 | 1 419 | |||||||||
| 0.625% | EUR | 500 | 2016 | 2028 | Novartis Finance S.A., Luxembourg, Luxembourg | 98.480% | 553 | 563 | |||||||||
| 1.800% | USD | 1 000 | 2017 | 2020 | Novartis Capital Corporation, New York, United States | 99.609% | 1 000 | 998 | |||||||||
| 2.400% | USD | 1 000 | 2017 | 2022 | Novartis Capital Corporation, New York, United States | 99.449% | 996 | 995 | |||||||||
| 3.100% | USD | 1 000 | 2017 | 2027 | Novartis Capital Corporation, New York, United States | 99.109% | 990 | 989 | |||||||||
| 0.000% | EUR | 1 250 | 2017 | 2021 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.133% | 1 396 | 1 421 | |||||||||
| 1.125% | EUR | 600 | 2017 | 2027 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.874% | 670 | 684 | |||||||||
| 0.500% | EUR | 750 | 2018 | 2023 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.655% | 837 | 853 | |||||||||
| 1.375% | EUR | 750 | 2018 | 2030 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.957% | 838 | 856 | |||||||||
| 1.700% | EUR | 750 | 2018 | 2038 | Novartis Finance S.A., Luxembourg, Luxembourg | 99.217% | 832 | 849 | |||||||||
| Total straight bonds | 22 167 | 25 283 | |||||||||||||||
| (USD millions) | 2019 | 2018 | |||
| 2019 | 3 190 | ||||
| 2020 | 2 002 | 2 006 | |||
| 2021 | 2 067 | 2 111 | |||
| 2022 | 2 583 | 2 585 | |||
| 2023 | 2 321 | 2 278 | |||
| 2024 | 2 139 | 2 137 | |||
| After 2024 | 11 243 | 11 353 | |||
| Total | 22 355 | 25 660 | |||
| (USD millions) | 2019 | 2018 | |||
| US dollar (USD) | 12 889 | 15 964 | |||
| Euro (EUR) | 7 861 | 8 028 | |||
| Japanese yen (JPY) | 184 | 272 | |||
| Swiss franc (CHF) | 1 421 | 1 396 | |||
| Total | 22 355 | 25 660 | |||
(USD millions) | 2019 Balance sheet | 2019 Fair values | 2018 Balance sheet | 2018 Fair values | |||||
| Straight bonds | 22 167 | 23 701 | 25 283 | 25 438 | |||||
| Others | 188 | 188 | 377 | 377 | |||||
| Total | 22 355 | 23 889 | 25 660 | 25 815 | |||||
| (USD millions) | 2019 | 2018 | |||
| Total net book value of property, plant and equipment pledged as collateral for non-current financial debts | 3 | 96 | |||
| (USD millions) | 2019 | 2018 | |||
| Accrued liability for employee benefits: | |||||
Defined benefit pension plans 1 | 3 469 | 3 546 | |||
| Other long-term employee benefits and deferred compensation | 546 | 600 | |||
Other post-employment benefits 1 | 612 | 954 | |||
| Environmental remediation provisions | 592 | 634 | |||
| Provisions for product liabilities, governmental investigations and other legal matters | 200 | 214 | |||
Contingent consideration 2 | 958 | 874 | |||
| Other non-current liabilities | 255 | 497 | |||
| Total provisions and other non-current liabilities | 6 632 | 7 319 | |||
1 Note 25 provides additional disclosures related to post-employment benefits. | |||||
2 Note 29 provides additional disclosures related to contingent consideration. | |||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| January 1 | 692 | 761 | 773 | ||||
| Cash payments | – 30 | – 48 | – 46 | ||||
Releases 1 | – 83 | – 21 | – 153 | ||||
Additions 2 | 124 | 7 | 154 | ||||
| Currency translation effects | 11 | – 7 | 33 | ||||
| December 31 | 714 | 692 | 761 | ||||
| Less current provision | – 122 | – 58 | – 55 | ||||
| Non-current environmental remediation provisions at December 31 | 592 | 634 | 706 | ||||
1 Releases of provisions credited to the consolidated income statement from continuing operations were USD 21 million in 2018 and USD 153 million in 2017. | |||||||
2 Provisions charged to the consolidated income statement from continuing operations were USD 7 million in 2018 and USD 154 million in 2017. | |||||||
(USD millions) | Expected cash outflows | ||
| Due within two years | 141 | ||
| Due later than two years, but within five years | 210 | ||
| Due later than five years, but within 10 years | 258 | ||
| Due after 10 years | 105 | ||
| Total environmental remediation liability provisions | 714 | ||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| January 1 | 340 | 351 | 395 | ||||
Provisions related to discontinued operations 1 | – 42 | ||||||
| Impact of business combinations | 10 | ||||||
| Cash payments | – 116 | – 118 | – 69 | ||||
Releases of provisions 2 | – 52 | – 107 | – 70 | ||||
Additions to provisions 3 | 1 230 | 220 | 93 | ||||
| Currency translation effects | – 1 | – 6 | 2 | ||||
| December 31 | 1 369 | 340 | 351 | ||||
| Less current portion | – 1 169 | – 126 | – 121 | ||||
| Non-current product liabilities, governmental investigations and other legal matters provisions at December 31 | 200 | 214 | 230 | ||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||
2 Releases of provisions credited to the consolidated income statement from continuing operations were USD 107 million in 2018 and USD 63 million in 2017. | |||||||
3 Provisions charged to the consolidated income statement from continuing operations were USD 220 million in 2018 and USD 47 million in 2017. | |||||||
| (USD millions) | 2019 | 2018 | |||
Interest-bearing accounts of associates payable on demand 1 | 1 836 | 1 778 | |||
Bank and other financial debt 2 | 719 | 701 | |||
| Commercial paper | 2 289 | 3 951 | |||
| Current portion of non-current financial debt | 2 002 | 3 190 | |||
| Fair value of derivative financial instruments | 185 | 58 | |||
| Total current financial debt and derivative financial instruments | 7 031 | 9 678 | |||
1 Weighted average interest rate 0.5% (2018: 0.5%) | |||||
2 Weighted average interest rate 12.9% (2018: 9.6%) | |||||
| (USD millions) | 2019 | 2018 | |||
| Taxes other than income taxes | 471 | 528 | |||
| Restructuring provisions | 438 | 507 | |||
| Accrued expenses for goods and services received but not invoiced | 1 046 | 970 | |||
| Accruals for royalties | 653 | 651 | |||
| Accrued interests on financial debt | 98 | 156 | |||
| Provisions for deductions from revenue | 5 595 | 5 262 | |||
| Accruals for compensation and benefits, including social security | 2 464 | 2 527 | |||
| Environmental remediation liabilities | 122 | 58 | |||
| Deferred income | 114 | 236 | |||
Provisions for product liabilities, governmental investigations and other legal matters 1 | 1 169 | 126 | |||
| Accrued share-based payments | 326 | 273 | |||
Contingent considerations 2 | 78 | 33 | |||
Commitment for repurchase of own shares 3 | 284 | ||||
| Other payables | 764 | 673 | |||
| Total provisions and other current liabilities | 13 338 | 12 284 | |||
1 Note 20 provides additional disclosures related to legal provisions. | |||||
2 Note 29 provides additional disclosures related to contingent considerations. | |||||
3 Note 18 provides additional disclosures related to commitment for repurchase of own shares. | |||||
Income statement charge2 | |||||||||||||||||
(USD millions) | Revenue deductions provisions at January 1 | Revenue deductions provisions related to discontinued operations1 | Effect of currency translation and business combinations | Payments/ utilizations | Adjustments of prior years | Current year | Change in provisions offset against gross trade receivables | Revenue deductions provisions at December 31 | |||||||||
| 2019 | |||||||||||||||||
| US-specific healthcare plans and program rebates | 1 883 | 0 | – 5 183 | – 193 | 5 474 | 1 981 | |||||||||||
| Non-US-specific healthcare plans and program rebates | 1 625 | – 28 | – 19 | – 2 467 | – 2 | 2 659 | 1 | 1 769 | |||||||||
| Non-healthcare plans and program-related rebates, returns and other deductions | 1 754 | – 166 | 9 | – 11 698 | – 25 | 11 868 | 103 | 1 845 | |||||||||
| Total 2019 | 5 262 | – 194 | – 10 | – 19 348 | – 220 | 20 001 | 104 | 5 595 | |||||||||
| 2018 | |||||||||||||||||
| US-specific healthcare plans and program rebates | 1 590 | – 4 158 | – 90 | 4 541 | 1 883 | ||||||||||||
| Non-US-specific healthcare plans and program rebates | 1 356 | – 78 | – 2 182 | 83 | 2 555 | – 109 | 1 625 | ||||||||||
| Non-healthcare plans and program-related rebates, returns and other deductions | 1 726 | – 51 | – 12 227 | – 91 | 11 956 | 441 | 1 754 | ||||||||||
| Total 2018 | 4 672 | – 129 | – 18 567 | – 98 | 19 052 | 332 | 5 262 | ||||||||||
| 2017 | |||||||||||||||||
| US-specific healthcare plans and program rebates | 1 461 | – 3 684 | – 62 | 3 875 | 1 590 | ||||||||||||
| Non-US-specific healthcare plans and program rebates | 1 020 | 131 | – 1 954 | 80 | 2 186 | – 107 | 1 356 | ||||||||||
| Non-healthcare plans and program-related rebates, returns and other deductions | 1 702 | 65 | – 11 814 | – 127 | 12 045 | – 145 | 1 726 | ||||||||||
| Total 2017 | 4 183 | 196 | – 17 452 | – 109 | 18 106 | – 252 | 4 672 | ||||||||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||||||||||||
2 Charges to the consolidated income statement from continuing operations were USD 18 248 million in 2018 and USD 17 772 million in 2017. | |||||||||||||||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| January 1 | 507 | 153 | 222 | ||||
Provisions related to discontinued operations 1 | – 8 | ||||||
Additions 2 | 492 | 534 | 194 | ||||
| Cash payments | – 479 | – 145 | – 200 | ||||
Releases 3 | – 72 | – 33 | – 64 | ||||
| Transfers | – 7 | ||||||
| Currency translation effects | – 2 | – 2 | 8 | ||||
| December 31 | 438 | 507 | 153 | ||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||
2 Provisions charged to the consolidated income statement from continuing operations were USD 521 million in 2018 and USD 186 million in 2017. | |||||||
3 Reversal of provisions credited to the consolidated income statement from continuing operations were USD 31 million in 2018 and USD 59 million in 2017. | |||||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Depreciation, amortization and impairments on: | |||||||
| Property, plant and equipment | 1 547 | 1 783 | 1 460 | ||||
Right-of-use assets 1 | 305 | ||||||
| Intangible assets | 3 974 | 3 428 | 3 276 | ||||
Financial assets 2 | – 38 | 6 | 227 | ||||
| Non-cash change in provisions and other non-current liabilities | 1 871 | 895 | 86 | ||||
| Gains on disposal and other adjustments on property, plant and equipment; intangible assets; financial assets; and other non-current assets, net | – 1 234 | – 902 | – 1 077 | ||||
| Equity-settled compensation expense | 758 | 673 | 612 | ||||
Income from associated companies 3 | – 659 | – 6 438 | – 1 108 | ||||
| Taxes | 1 793 | 1 295 | 1 603 | ||||
| Net financial expense | 805 | 746 | 708 | ||||
| Total | 9 122 | 1 486 | 5 787 | ||||
1 Depreciation of right-of-use assets recognized from January 1, 2019, the date of implementation of IFRS 16 leases. Notes 1 and 10 provide additional disclosures. | |||||||
2 Includes fair value adjustments | |||||||
3 2018 includes a reversal of a pre-tax gain (USD 5.8 billion) recognized from the divestment of the investment in GSK Consumer Healthcare Holdings Ltd. (see Note 2). The net cash proceed of USD 13.0 billion from the divestment was included in the consolidated statements of cash flows in the line "Acquisitions and divestments of interests in associated companies, net." | |||||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| (Increase) in inventories | – 382 | – 387 | – 203 | ||||
| (Increase) in trade receivables | – 980 | – 544 | – 655 | ||||
| Increase in trade payables | 553 | 252 | 82 | ||||
| Change in other current assets | – 160 | 316 | – 303 | ||||
| Change in other current liabilities | 1 167 | 941 | 1 410 | ||||
| Other adjustments, net | 1 | – 2 | 1 | ||||
| Total | 199 | 576 | 332 | ||||
| (USD millions) | Note | 2019 | 2018 | 2017 | |||||
| Net assets recognized as a result of business combinations | 24 | – 4 124 | – 13 660 | – 874 | |||||
| Fair value of previously held equity interests | 33 | ||||||||
| Receivables and payables contingent consideration, net | 242 | – 5 | 151 | ||||||
| Payments, deferred consideration and other adjustments, net | – 2 | – 36 | – 36 | ||||||
| Cash flows used for acquisitions of businesses | – 3 851 | – 13 701 | – 759 | ||||||
Cash flows from divestments of businesses, net 1 | 91 | 18 | 45 | ||||||
| Cash flows used for acquisitions and divestments of businesses, net | – 3 760 | – 13 683 | – 714 | ||||||
1 In 2019, the USD 91 million included USD 4 million net cash outflows from previous years divestments and USD 95 million net cash inflows from business divestments in 2019. The net identifiable assets of the 2019 divested businesses amounted to USD 196 million, comprised of non-current asset of USD 159 million, current assets of USD 96 million including USD 11 million cash and cash equivalents, non-current liabilities USD 18 million and current liabilities of USD 41 million. In 2018, USD 18 million represented the net cash inflows from previous years divestments. In 2017, the USD 45 million net cash inflows related to the net identifiable assets from a 2017 divestment. The 2017 divested business amounted to USD 48 million, comprised of non-current assets of USD 29 million, current assets of USD 34 million and current liabilities of USD 15 million. | |||||||||
(USD millions) | Non-current financial debts | Current financial debts and derivative financial instruments | Non-current lease liabilities | Current lease liabilities | |||||
| January 1, 2019 | 22 470 | 9 678 | |||||||
Impact of adoption of IFRS 16 Leases continuing operations 1 | – 2 | – 1 | 1 471 | 268 | |||||
Impact of adoption of IFRS 16 Leases discontinued operations 2 | – 89 | 246 | 40 | ||||||
Financial debts and lease liabilities related to discontinued operations 3 | – 47 | – 246 | – 40 | ||||||
| Increase in non-current financial debts | 93 | ||||||||
| Repayment of non-current financial debts | – 3 195 | ||||||||
| Change in current financial debts | – 1 582 | ||||||||
| Payments of lease liabilities, net | – 273 | ||||||||
| Interest payments for amounts included in lease liabilities classified as cash flows from operating activities | – 51 | ||||||||
| New leases | 362 | 131 | |||||||
| Impact of business combinations and divestments | 2 | – 11 | – 6 | ||||||
| Changes in fair values, and other changes, net | 129 | 33 | 20 | ||||||
| Amortization of bonds discount | 25 | ||||||||
| Currency translation effects | – 141 | 44 | 4 | 1 | |||||
| Reclassification from non-current to current, net | – 2 003 | 2 003 | – 156 | 156 | |||||
| December 31, 2019 | 20 353 | 7 031 | 1 703 | 246 | |||||
1 Lease liabilties recognized on January 1, 2019, the date of implementation of IFRS 16 Leases. Note 10 provides additional disclosure. | |||||||||
2 In 2018, financial debts included USD 89 million for previously reported finance lease obligations of the Alcon business that have been reclassified on January 1, 2019, to lease liabilities, with the adoption of IFRS 16 Leases. Note 30 provides additional disclosures. | |||||||||
3 Represents the financial debts and lease liabilities at January 1, 2019, related to the Alcon business reported as discontinued operations. See Notes 1, 2 and 30. | |||||||||
(USD millions) | Non-current financial debts | Current financial debts and derivative financial instruments | |||
| January 1, 2018 | 23 224 | 5 308 | |||
Increase in non-current financial debts 1 | 2 856 | ||||
Repayment of non-current financial debts 2 | – 366 | ||||
Change in current financial debts 3 | 1 681 | ||||
| Impact of business combinations | 10 | 4 | |||
| Changes in fair values, and other changes | 5 | – 48 | |||
| Amortization of bonds discount | 27 | 2 | |||
| Currency translation effects | – 462 | – 93 | |||
| Current portion of non-current financial debt | – 3 190 | 3 190 | |||
| December 31, 2018 | 22 470 | 9 678 | |||
1 Increases in non-current financial debts were only recorded in the consolidated statements of cash flows from continuing operations. | |||||
2 Repayment of non-current financial debts were only recorded in the consolidated statements of cash flows from continuing operations. | |||||
3 Changes in current financial debts included in the consolidated statements of cash flows from continuing operations were USD 1 687 million. | |||||
(USD millions) | Non-current financial debts | Current financial debts and derivative financial instruments | |||
| January 1, 2017 | 17 897 | 5 905 | |||
Increase in non-current financial debts 1 | 4 933 | ||||
Repayment of non-current financial debts 2 | – 1 | – 187 | |||
Change in current financial debts 3 | – 755 | ||||
| Changes in fair values, and other changes | – 6 | – 140 | |||
| Amortization of bonds discount | 16 | ||||
| Currency translation effects | 744 | 126 | |||
| Current portion of non-current financial debt | – 359 | 359 | |||
| December 31, 2017 | 23 224 | 5 308 | |||
1 Increases in non-current financial debts were only recorded in the consolidated statements of cash flows from continuing operations. | |||||
2 Repayment of non-current financial debts were only recorded in the consolidated statements of cash flows from continuing operations. | |||||
3 Changes in current financial debts included in the consolidated statements of cash flows from continuing operations were USD 644 million. | |||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Property, plant and equipment | 44 | 137 | |||||
| Currently marketed products | 3 550 | 2 531 | |||||
| Acquired research and development | 342 | 10 224 | 1 223 | ||||
| Other intangible assets | 22 | 1 | |||||
| Deferred tax assets | 60 | 381 | 8 | ||||
| Financial and other assets | 8 | 19 | |||||
| Inventories | 195 | 20 | |||||
| Trade receivables and other current assets | 4 | 90 | |||||
| Cash and cash equivalents | 1 112 | 20 | |||||
| Deferred tax liabilities | – 107 | – 2 874 | – 325 | ||||
| Current and non-current financial debts | – 2 | – 14 | |||||
| Trade payables and other liabilities | – 178 | – 627 | – 1 | ||||
| Net identifiable assets acquired | 3 938 | 11 000 | 925 | ||||
| Cash and cash equivalents | – 1 112 | – 20 | |||||
| Non-controlling interests | – 26 | ||||||
| Goodwill | 186 | 4 084 | 94 | ||||
Net assets recognized as a result of business combinations 1 | 4 124 | 13 946 | 999 | ||||
1 Net assets recognized as a result of business combinations in the consolidated balance sheet from continuing operations were USD 13 660 million in 2018 and USD 874 million in 2017. | |||||||
| Pension plans | Other post-employment benefit plans | ||||||||
| (USD millions) | 2019 | 2018 | 2019 | 2018 | |||||
| Benefit obligation at January 1 | 22 179 | 23 210 | 1 073 | 1 115 | |||||
Benefit obligations related to discontinued operations 1 | – 662 | – 385 | |||||||
| Current service cost | 336 | 378 | 13 | 34 | |||||
| Interest cost | 330 | 321 | 29 | 39 | |||||
| Past service costs and settlements | – 168 | – 1 | |||||||
| Administrative expenses | 24 | 26 | |||||||
| Remeasurement losses/(gains) arising from changes in financial assumptions | 1 791 | – 567 | 76 | – 31 | |||||
| Remeasurement (gains)/losses arising from changes in demographic assumptions | – 193 | 5 | – 9 | 1 | |||||
| Experience-related remeasurement losses/(gains) | 184 | 264 | – 22 | – 32 | |||||
| Currency translation effects | 283 | – 374 | – 7 | ||||||
| Benefit payments | – 1 256 | – 1 263 | – 30 | – 46 | |||||
| Contributions of associates | 169 | 169 | |||||||
| Effect of acquisitions, divestments or transfers | 49 | 11 | 1 | ||||||
| Benefit obligation at December 31 | 23 066 | 22 179 | 746 | 1 073 | |||||
| Fair value of plan assets at January 1 | 18 838 | 20 275 | 119 | 162 | |||||
Plan assets related to discontinued operations 1 | – 424 | – 40 | |||||||
| Interest income | 257 | 249 | 3 | 5 | |||||
| Return on plan assets excluding interest income | 1 656 | – 805 | 10 | – 8 | |||||
| Currency translation effects | 304 | – 310 | |||||||
| Novartis Group contributions | 420 | 520 | 74 | 6 | |||||
| Contributions of associates | 169 | 169 | |||||||
| Settlements | – 193 | – 3 | |||||||
| Benefit payments | – 1 256 | – 1 263 | – 30 | – 46 | |||||
| Effect of acquisitions, divestments or transfers | 39 | 6 | – 2 | ||||||
| Fair value of plan assets at December 31 | 19 810 | 18 838 | 134 | 119 | |||||
| Funded status | – 3 256 | – 3 341 | – 612 | – 954 | |||||
| Limitation on recognition of fund surplus at January 1 | – 68 | – 89 | |||||||
| Change in limitation on recognition of fund surplus (incl. exchange rate differences) | 7 | 25 | |||||||
| Interest income on limitation of fund surplus | – 4 | – 4 | |||||||
| Limitation on recognition of fund surplus at December 31 | – 65 | – 68 | |||||||
| Net liability in the balance sheet at December 31 | – 3 321 | – 3 409 | – 612 | – 954 | |||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||||
| Pension plans | Other post-employment benefit plans | ||||||||
| (USD millions) | 2019 | 2018 | 2019 | 2018 | |||||
| Net liability at January 1 | – 3 409 | – 3 024 | – 954 | – 953 | |||||
Less: net liability related to discontinued operations 1 | 238 | 345 | |||||||
| Current service cost | – 336 | – 378 | – 13 | – 34 | |||||
| Net interest expense | – 77 | – 76 | – 26 | – 34 | |||||
| Administrative expenses | – 24 | – 26 | |||||||
| Past service costs and settlements | – 25 | – 2 | |||||||
| Remeasurements | – 126 | – 507 | – 35 | 54 | |||||
| Currency translation effects | 21 | 64 | 7 | ||||||
| Novartis Group contributions | 420 | 520 | 74 | 6 | |||||
| Effect of acquisitions, divestments or transfers | – 10 | – 5 | – 3 | ||||||
| Change in limitation on recognition of fund surplus | 7 | 25 | |||||||
| Net liability at December 31 | – 3 321 | – 3 409 | – 612 | – 954 | |||||
| Amounts recognized in the consolidated balance sheet | |||||||||
| Prepaid benefit cost | 148 | 137 | |||||||
| Accrued benefit liability | – 3 469 | – 3 546 | – 612 | – 954 | |||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | |||||||||
| 2019 | 2018 | ||||||||||||||||
(USD millions) | Switzerland | United States | Rest of the world | Total | Switzerland | United States | Rest of the world | Total | |||||||||
| Benefit obligation at December 31 | 15 106 | 3 552 | 4 408 | 23 066 | 14 263 | 3 348 | 4 568 | 22 179 | |||||||||
| Thereof unfunded | 670 | 466 | 1 136 | 649 | 491 | 1 140 | |||||||||||
| By type of member | |||||||||||||||||
| Active | 6 167 | 630 | 1 400 | 8 197 | 5 618 | 653 | 1 616 | 7 887 | |||||||||
| Deferred pensioners | 1 205 | 1 517 | 2 722 | 1 131 | 1 531 | 2 662 | |||||||||||
| Pensioners | 8 939 | 1 717 | 1 491 | 12 147 | 8 645 | 1 564 | 1 421 | 11 630 | |||||||||
| Fair value of plan assets at December 31 | 14 457 | 2 311 | 3 042 | 19 810 | 13 470 | 2 160 | 3 208 | 18 838 | |||||||||
| Funded status | – 649 | – 1 241 | – 1 366 | – 3 256 | – 793 | – 1 188 | – 1 360 | – 3 341 | |||||||||
| 2019 | 2018 | ||||||||||||
(USD millions) | United States | Rest of the world | Total | United States | Rest of the world | Total | |||||||
| Benefit obligation at December 31 | 658 | 88 | 746 | 1 001 | 72 | 1 073 | |||||||
| Thereof unfunded | 524 | 88 | 612 | 882 | 72 | 954 | |||||||
| By type of member | |||||||||||||
| Active | 121 | 36 | 157 | 270 | 25 | 295 | |||||||
| Deferred pensioners | 15 | 0 | 15 | 18 | 0 | 18 | |||||||
| Pensioners | 522 | 52 | 574 | 713 | 47 | 760 | |||||||
| Fair value of plan assets at December 31 | 134 | 0 | 134 | 119 | 0 | 119 | |||||||
| Funded status | – 524 | – 88 | – 612 | – 882 | – 72 | – 954 | |||||||
| Pension plans | Other post-employment benefit plans | ||||||||||||
| 2019 | 2018 | 2017 | 2019 | 2018 | 2017 | ||||||||
| Weighted average assumptions used to determine benefit obligations at December 31 | |||||||||||||
| Discount rate | 1.0% | 1.6% | 1.5% | 3.6% | 4.4% | 3.7% | |||||||
| Expected rate of pension increase | 0.3% | 0.4% | 0.5% | ||||||||||
| Expected rate of salary increase | 2.8% | 2.8% | 2.8% | ||||||||||
| Interest on savings account | 0.3% | 0.8% | 0.6% | ||||||||||
| Current average life expectancy for a 65-year-old male in years | 22 | 22 | 22 | 21 | 21 | 21 | |||||||
| Current average life expectancy for a 65-year-old female in years | 24 | 24 | 24 | 23 | 23 | 23 | |||||||
(USD millions) | Change in 2019 year-end defined benefit pension obligation | ||
| 25 basis point increase in discount rate | – 787 | ||
| 25 basis point decrease in discount rate | 837 | ||
| One-year increase in life expectancy | 848 | ||
| 25 basis point increase in rate of pension increase | 546 | ||
| 25 basis point decrease in rate of pension increase | – 135 | ||
| 25 basis point increase of interest on savings account | 62 | ||
| 25 basis point decrease of interest on savings account | – 60 | ||
| 25 basis point increase in rate of salary increase | 54 | ||
| 25 basis point decrease in rate of salary increase | – 55 | ||
| 2019 | 2018 | 2017 | |||||
| Healthcare cost trend rate assumed for next year | 6.5% | 7.0% | 6.5% | ||||
| Rate to which the cost trend rate is assumed to decline | 4.5% | 4.5% | 4.5% | ||||
| Year that the rate reaches the ultimate trend rate | 2028 | 2028 | 2025 | ||||
| Pension plans | |||||||||
(as a percentage) | Long-term target minimum | Long-term target maximum | 2019 | 2018 | |||||
| Equity securities | 15 | 40 | 27 | 28 | |||||
| Debt securities | 20 | 60 | 36 | 35 | |||||
| Real estate | 5 | 20 | 17 | 17 | |||||
| Alternative investments | 0 | 20 | 15 | 16 | |||||
| Cash and other investments | 0 | 15 | 5 | 4 | |||||
| Total | 100 | 100 | |||||||
| December 31, 2019 | December 31, 2018 | ||||
| Investment in shares of Novartis AG | |||||
| Number of shares (in millions) | 2.3 | 11.0 | |||
| Market value (in USD billions) | 0.2 | 0.9 | |||
(USD millions) | Pension plans | Other post- employment benefit plans | |||
| Novartis Group contributions | |||||
| 2020 (estimated) | 410 | 46 | |||
| Expected future benefit payments | |||||
| 2020 | 1 201 | 46 | |||
| 2021 | 1 116 | 47 | |||
| 2022 | 1 106 | 48 | |||
| 2023 | 1 096 | 48 | |||
| 2024 | 1 087 | 48 | |||
| 2025–2029 | 5 270 | 231 | |||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Contributions for defined contribution plans continuing operations | 422 | 443 | 307 | ||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Expense related to equity-based participation plans | 1 067 | 918 | 853 | ||||
| Liabilities arising from equity-based payment transactions | 326 | 273 | 261 | ||||
| 2019 | 2018 | ||||||||
Options (millions) | Weighted average exercise price (USD) | Options (millions) | Weighted average exercise price (USD) | ||||||
| Options outstanding at January 1 | 5.6 | 59.9 | 7.4 | 59.5 | |||||
| Sold or exercised | – 2.2 | 58.4 | – 1.8 | 58.2 | |||||
| Outstanding at December 31 | 3.4 | 60.9 | 5.6 | 59.9 | |||||
| Exercisable at December 31 | 3.4 | 60.9 | 5.6 | 59.9 | |||||
Options outstanding | Total/ weighted average | ||||||||||
| Number outstanding (millions) | 0.3 | 0.7 | 0.9 | 1.5 | 3.4 | ||||||
| Remaining contractual life (years) | 0.0 | 1.0 | 2.0 | 3.0 | 2.1 | ||||||
| Exercise price (USD) | 54.5 | 57.0 | 57.6 | 66.0 | 60.9 | ||||||
| 2019 | 2018 | ||||||||
ADR options (millions) | Weighted average exercise price (USD) | ADR options (millions) | Weighted average exercise price (USD) | ||||||
| Options outstanding at January 1 | 15.2 | 60.7 | 20.3 | 59.9 | |||||
| Sold or exercised | – 5.6 | 58.6 | – 5.1 | 57.4 | |||||
| Outstanding at December 31 | 9.6 | 61.9 | 15.2 | 60.7 | |||||
| Exercisable at December 31 | 9.6 | 61.9 | 15.2 | 60.7 | |||||
ADR options outstanding | Total/ weighted average | ||||||||||
| Number outstanding (millions) | 0.4 | 1.2 | 2.9 | 5.1 | 9.6 | ||||||
| Remaining contractual life (years) | 0.0 | 1.0 | 2.0 | 3.0 | 2.3 | ||||||
| Exercise price (USD) | 54.5 | 57.0 | 57.6 | 66.0 | 61.9 | ||||||
| 2019 | 2018 | ||||||||||||
Number of shares in millions | Weighted average fair value at grant date in USD | Fair value at grant date in USD millions | Number of shares in millions | Weighted average fair value at grant date in USD | Fair value at grant date in USD millions | ||||||||
| Non-vested shares at January 1 | 25.7 | 77.1 | 1 981 | 23.9 | 80.6 | 1 926 | |||||||
| Granted | |||||||||||||
| – Annual Incentive | 1.1 | 78.4 | 86 | 1.3 | 83.9 | 109 | |||||||
| – Share savings plans | 4.2 | 83.0 | 349 | 4.1 | 84.9 | 348 | |||||||
| – Select North America | 5.3 | 64.0 | 339 | 3.9 | 77.8 | 303 | |||||||
| – Select outside North America | 2.6 | 67.4 | 175 | 2.1 | 79.7 | 167 | |||||||
| – Long-Term Performance Plan | 2.5 | 68.9 | 172 | 1.5 | 85.8 | 129 | |||||||
| – Long-Term Relative Performance Plan | 0.1 | 0.0 | 0 | 0.3 | 52.0 | 16 | |||||||
| – Other share awards | 1.9 | 67.7 | 129 | 1.2 | 77.9 | 93 | |||||||
| Vested | – 13.3 | 80.3 | – 1 068 | – 10.7 | 90.2 | – 965 | |||||||
| Forfeited | – 4.3 | 76.3 | – 328 | – 1.9 | 76.4 | – 145 | |||||||
| Non-vested shares at December 31 | 25.8 | 71.1 | 1 835 | 25.7 | 77.1 | 1 981 | |||||||
| Executive Officers | Non-Executive Directors | Total | |||||||||||||||||
| (USD millions) | 2019 | 2018 | 2017 | 2019 | 2018 | 2017 | 2019 | 2018 | 2017 | ||||||||||
| Cash and other compensation | 20.7 | 22.5 | 18.4 | 4.1 | 4.0 | 4.0 | 24.8 | 26.5 | 22.4 | ||||||||||
| Post-employment benefits | 2.6 | 2.5 | 2.0 | 2.6 | 2.5 | 2.0 | |||||||||||||
| Equity-based compensation | 40.6 | 42.5 | 49.9 | 4.6 | 4.8 | 4.8 | 45.2 | 47.3 | 54.7 | ||||||||||
| Total | 63.9 | 67.5 | 70.3 | 8.7 | 8.8 | 8.8 | 72.6 | 76.3 | 79.1 | ||||||||||
| Currency | 2019 | 2018 | 2017 | ||||||
| Dr. Krauer | CHF | 60 000 | 60 000 | 60 000 | |||||
| Dr. Vasella | CHF | 0 | 18 228 | 26 279 | |||||
| (USD millions) | 2019 | ||
| 2020 | 809 | ||
| 2021 | 442 | ||
| 2022 | 319 | ||
| 2023 | 724 | ||
| 2024 | 167 | ||
| Thereafter | 1 943 | ||
| Total | 4 404 | ||
| (USD millions) | Note | 2019 1 | 2018 1 | ||||
| Cash and cash equivalents | 16 | 11 112 | 13 271 | ||||
| Financial assets - measured at fair value through other comprehensive income | |||||||
| Marketable securities | |||||||
| Debt securities | 16 | 24 | 325 | ||||
| Long-term financial investments | |||||||
| Equity securities | 13 | 1 158 | 802 | ||||
| Debt securities | 13 | 33 | 31 | ||||
| Total long-term financial investments - fair value through other comprehensive income | 1 191 | 833 | |||||
| Total financial assets - measured at fair value through other comprehensive income | 1 215 | 1 158 | |||||
| Financial assets - measured at amortized costs | |||||||
| Trade receivables, income tax receivables and other current assets (excluding pre-payments) | 15/17 | 10 337 | 11 024 | ||||
| Accrued interest on debt securities, time deposits and short-term investments | 16 | 12 | |||||
| Time deposits and short-term investments with original maturity more than 90 days | 16 | 61 | 2 087 | ||||
| Long-term loans, advances, security deposits and other long-term receivables | 13 | 329 | 512 | ||||
| Total financial assets - measured at amortized costs | 10 727 | 13 635 | |||||
| Financial assets - measured at fair value through the consolidated income statement | |||||||
| Equity securities | 13 | 366 | 353 | ||||
| Fund investments | 13/16 | 270 | 286 | ||||
| Associated companies at fair value through profit and loss | 186 | 145 | |||||
| Derivative financial instruments | 16 | 102 | 130 | ||||
| Contingent consideration receivables | 13 | 399 | 396 | ||||
| Total financial assets - measured at fair value through the consolidated income statement | 1 323 | 1 310 | |||||
| Total financial assets | 24 377 | 29 374 | |||||
| Financial liabilities - measured at amortized costs | |||||||
| Current financial debt | |||||||
| Interest-bearing accounts of associates payable on demand | 21 | 1 836 | 1 778 | ||||
| Bank and other financial debt | 21 | 719 | 701 | ||||
| Commercial paper | 21 | 2 289 | 3 951 | ||||
| Current portion of non-current debt | 21 | 2 002 | 3 190 | ||||
| Total current financial debt | 6 846 | 9 620 | |||||
| Non-current financial debt | |||||||
| Straight bonds | 19 | 22 167 | 25 283 | ||||
| Liabilities to banks and other financial institutions | 19 | 188 | 285 | ||||
| Finance lease obligations | 19 | 92 | |||||
| Current portion of non-current debt | 19 | – 2 002 | – 3 190 | ||||
| Total non-current financial debt | 20 353 | 22 470 | |||||
Trade payables and commitment for repurchase of own shares 2 | 5 424 | 5 840 | |||||
| Total financial liabilities - measured at amortized costs | 32 623 | 37 930 | |||||
| Financial liabilities - measured at fair value through the consolidated income statement | |||||||
| Contingent consideration (see Note 20/22) and other financial liabilities | 1 065 | 917 | |||||
| Derivative financial instruments | 21 | 185 | 58 | ||||
| Total financial liabilities - measured at fair value through the consolidated income statement | 1 250 | 975 | |||||
| Lease liabilities | 10 | 1 949 | |||||
| Total financial liabilities | 35 822 | 38 905 | |||||
1 Except for straight bonds (see Note 19), the carrying amount is a reasonable approximation of fair value. | |||||||
2 Notes 18 and 22 provide additional disclosures related to commitment for repurchase of own shares. | |||||||
| Contract or underlying principal amount | Positive fair values | Negative fair values | |||||||||||
| (USD millions) | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | |||||||
| Forward foreign exchange rate contracts | 10 779 | 10 823 | 96 | 130 | – 75 | – 58 | |||||||
| Commodity purchase contract | 9 | 6 | |||||||||||
| Options on equity securities | 269 | – 110 | |||||||||||
| Total derivative financial instruments included in marketable securities and in current financial debts | 11 057 | 10 823 | 102 | 130 | – 185 | – 58 | |||||||
| 2019 | |||||||||
| (USD millions) | EUR | USD | Other | Total | |||||
| Forward foreign exchange rate contracts | 1 373 | 7 760 | 1 646 | 10 779 | |||||
| Commodity purchase contract | 9 | 9 | |||||||
| Options on equity securities | 250 | 19 | 269 | ||||||
| Total derivative financial instruments | 1 373 | 8 019 | 1 665 | 11 057 | |||||
| 2018 | |||||||||
| (USD millions) | EUR | USD | Other | Total | |||||
| Forward foreign exchange rate contracts | 2 989 | 6 558 | 1 276 | 10 823 | |||||
| Total derivative financial instruments | 2 989 | 6 558 | 1 276 | 10 823 | |||||
| 2019 | |||||||||||
(USD millions) | Level 1 | Level 2 | Level 3 | Valued at amortized cost | Total | ||||||
| Financial assets | |||||||||||
| Debt securities | 24 | 24 | |||||||||
| Fund investments | 37 | 37 | |||||||||
| Total marketable securities | 37 | 24 | 61 | ||||||||
| Time deposits and short term investments with original maturity more than 90 days | 61 | 61 | |||||||||
| Derivative financial instruments | 102 | 102 | |||||||||
| Total marketable securities, time deposits and derivative financial instruments | 37 | 126 | 61 | 224 | |||||||
| Debt and equity securities | 976 | 581 | 1 557 | ||||||||
| Fund investments | 233 | 233 | |||||||||
| Contingent consideration receivables | 399 | 399 | |||||||||
| Long-term loans, advances, security deposits and other long-term receivables | 329 | 329 | |||||||||
| Total financial investments and long-term loans | 976 | 1 213 | 329 | 2 518 | |||||||
| Associated companies at fair value through profit and loss | 186 | 186 | |||||||||
| Financial liabilities | |||||||||||
| Contingent consideration payables | – 1 036 | – 1 036 | |||||||||
| Other financial liabilities | – 29 | – 29 | |||||||||
| Derivative financial instruments | – 185 | – 185 | |||||||||
| Total financial liabilities at fair value | – 185 | – 1 065 | – 1 250 | ||||||||
| 2018 | |||||||||||
(USD millions) | Level 1 | Level 2 | Level 3 | Valued at amortized cost | Total | ||||||
| Financial assets | |||||||||||
| Debt securities | 302 | 23 | 325 | ||||||||
| Fund investments | 35 | 35 | |||||||||
| Total marketable securities | 337 | 23 | 360 | ||||||||
| Time deposits and short term investments with original maturity more than 90 days | 2 087 | 2 087 | |||||||||
| Derivative financial instruments | 130 | 130 | |||||||||
| Accrued interest on debt securities, time deposits and short-term investments | 12 | 12 | |||||||||
| Total marketable securities, time deposits and derivative financial instruments | 337 | 153 | 2 099 | 2 589 | |||||||
| Debt and equity securities | 698 | 488 | 1 186 | ||||||||
| Fund investments | 251 | 251 | |||||||||
| Contingent consideration receivables | 396 | 396 | |||||||||
| Long-term loans and receivables from customers and finance lease, advances, security deposits | 512 | 512 | |||||||||
| Total financial investments and long-term loans | 698 | 1 135 | 512 | 2 345 | |||||||
| Associated companies at fair value through profit and loss | 145 | 145 | |||||||||
| Financial liabilities | |||||||||||
| Contingent consideration payables | – 907 | – 907 | |||||||||
| Other financial liabilities | – 10 | – 10 | |||||||||
| Derivative financial instruments | – 58 | – 58 | |||||||||
| Total financial liabilities at fair value | – 58 | – 917 | – 975 | ||||||||
| 2019 | ||||||||||||||
(USD millions) | Associated companies at fair value through profit and loss | Fund investments | Long-term financial investments | Contingent consideration receivables | Contingent consideration payables | Other financial liabilities | ||||||||
| January 1 | 145 | 251 | 488 | 396 | – 907 | – 10 | ||||||||
Impact from discontinued operations 1 | – 28 | – 19 | 163 | |||||||||||
| Fair value gains and other adjustments, including from divestments recognized in the consolidated income statement | 12 | 6 | 35 | 195 | 1 | |||||||||
| Fair value losses (including impairments and amortizations) and other adjustments recognized in the consolidated income statement | – 15 | – 89 | – 48 | |||||||||||
| Fair value adjustments recognized in the consolidated statement of comprehensive income | – 6 | |||||||||||||
| Purchases | 49 | 28 | 229 | – 401 | – 5 | |||||||||
| Cash receipts and payments | – 32 | 3 | 33 | |||||||||||
| Disposals | – 3 | – 30 | – 53 | |||||||||||
| Reclassification | 10 | – 64 | ||||||||||||
| December 31 | 186 | 233 | 581 | 399 | – 1 036 | – 29 | ||||||||
| Total of fair value gains and losses recognized in the consolidated income statement for assets and liabilities held at December 31, 2019 | – 15 | 12 | 6 | 35 | 106 | – 47 | ||||||||
1 Notes 1, 2 and 30 provide information related to discontinued operations. | ||||||||||||||
| 2018 | ||||||||||||||
(USD millions) | Associated companies at fair value through profit and loss | Fund investments | Long-term financial investments | Contingent consideration receivables | Contingent consideration payables | Other financial liabilities | ||||||||
| January 1 | 188 | 166 | 437 | 844 | – 852 | – 72 | ||||||||
| Fair value gains and other adjustments, including from divestments recognized in the consolidated income statement | 93 | 36 | 213 | |||||||||||
| Fair value losses (including impairments and amortizations) and other adjustments recognized in the consolidated income statement | – 22 | – 5 | – 100 | |||||||||||
| Fair value adjustments recognized in the consolidated statement of comprehensive income | – 10 | |||||||||||||
| Purchases | 24 | 22 | 123 | – 182 | ||||||||||
| Cash receipts and payments | – 484 | 11 | 62 | |||||||||||
| Disposals | – 6 | – 30 | – 25 | |||||||||||
| Contingent consideration payable related to disposal group held for sale | 3 | |||||||||||||
| Reclassification | – 39 | – 32 | ||||||||||||
| December 31 | 145 | 251 | 488 | 396 | – 907 | – 10 | ||||||||
| Total of fair value gains and losses recognized in the consolidated income statement for assets and liabilities held at December 31, 2018 | – 22 | 93 | – 5 | 36 | 113 | |||||||||
| (USD millions) | 2019 | 2018 | |||
| Listed equity securities | 843 | 597 | |||
| Non-listed equity securities | 315 | 205 | |||
| Total equity securities | 1 158 | 802 | |||
| 2019 | |||||||||||||
(USD millions) | Due within one month | Due later than one month but less than three months | Due later than three months but less than one year | Due later than one year but less than five years | Due after five years | Total | |||||||
| Current assets | |||||||||||||
| Marketable securities, time deposits and short-term investments with original maturity more than 90 days | 20 | 26 | 16 | 3 | 57 | 122 | |||||||
| Commodities | 110 | 110 | |||||||||||
| Derivative financial instruments and accrued interest | 14 | 79 | 3 | 3 | 3 | 102 | |||||||
| Cash and cash equivalents | 9 712 | 1 400 | 11 112 | ||||||||||
| Total current financial assets | 9 746 | 1 505 | 19 | 6 | 170 | 11 446 | |||||||
| Non-current liabilities | |||||||||||||
| Financial debt | – 9 110 | – 11 243 | – 20 353 | ||||||||||
| Financial debt - undiscounted | – 9 150 | – 11 355 | – 20 505 | ||||||||||
| Total non-current financial debt | – 9 110 | – 11 243 | – 20 353 | ||||||||||
| Current liabilities | |||||||||||||
| Financial debt | – 4 243 | – 1 373 | – 1 230 | – 6 846 | |||||||||
| Financial debt - undiscounted | – 4 243 | – 1 373 | – 1 230 | – 6 846 | |||||||||
| Derivative financial instruments | – 130 | – 29 | – 26 | – 185 | |||||||||
| Total current financial debt | – 4 373 | – 1 402 | – 1 256 | – 7 031 | |||||||||
| Net debt | 5 373 | 103 | – 1 237 | – 9 104 | – 11 073 | – 15 938 | |||||||
| 2018 | |||||||||||||
(USD millions) | Due within one month | Due later than one month but less than three months | Due later than three months but less than one year | Due later than one year but less than five years | Due after five years | Total | |||||||
| Current assets | |||||||||||||
| Marketable securities, time deposits and short-term investments with original maturity more than 90 days | 39 | 56 | 2 091 | 198 | 63 | 2 447 | |||||||
| Commodities | 104 | 104 | |||||||||||
| Derivative financial instruments and accrued interest | 40 | 75 | 27 | 142 | |||||||||
| Cash and cash equivalents | 3 571 | 9 700 | 13 271 | ||||||||||
| Total current financial assets | 3 650 | 9 831 | 2 118 | 198 | 167 | 15 964 | |||||||
| Non-current liabilities | |||||||||||||
| Financial debt | – 8 980 | – 13 490 | – 22 470 | ||||||||||
| Financial debt - undiscounted | – 9 025 | – 13 623 | – 22 648 | ||||||||||
| Total non-current financial debt | – 8 980 | – 13 490 | – 22 470 | ||||||||||
| Current liabilities | |||||||||||||
| Financial debt | – 5 217 | – 4 084 | – 319 | – 9 620 | |||||||||
| Financial debt - undiscounted | – 5 217 | – 4 084 | – 319 | – 9 620 | |||||||||
| Derivative financial instruments | – 16 | – 34 | – 8 | – 58 | |||||||||
| Total current financial debt | – 5 233 | – 4 118 | – 327 | – 9 678 | |||||||||
| Net debt | – 1 583 | 5 713 | 1 791 | – 8 782 | – 13 323 | – 16 184 | |||||||
| 2019 | |||||||||
(USD millions) | Due within one month | Due later than one month but less than three months | Due later than three months but less than one year | Total | |||||
| Derivative financial instruments and accrued interest on derivative financial instruments | |||||||||
| Potential outflows in various currencies - from financial derivative liabilities | – 814 | – 4 624 | – 952 | – 6 390 | |||||
| Potential inflows in various currencies - from financial derivative assets | 807 | 4 656 | 922 | 6 385 | |||||
| 2018 | |||||||||
(USD millions) | Due within one month | Due later than one month but less than three months | Due later than three months but less than one year | Total | |||||
| Derivative financial instruments and accrued interest on derivative financial instruments | |||||||||
| Potential outflows in various currencies - from financial derivative liabilities | – 1 305 | – 2 949 | – 598 | – 4 852 | |||||
| Potential inflows in various currencies - from financial derivative assets | 1 328 | 2 974 | 593 | 4 895 | |||||
| 2019 | |||||||||||
(USD millions) | Due later than one month but less than three months | Due later than three months but less than one year | Due later than one year but less than five years | Due after five years | Total | ||||||
| Contractual interest on non-current liabilities | – 36 | – 428 | – 1 531 | – 3 439 | – 5 434 | ||||||
| Lease liabilities | – 65 | – 181 | – 622 | – 1 081 | – 1 949 | ||||||
| Trade payables | – 5 222 | – 202 | – 5 424 | ||||||||
| Contingent consideration liabilities | – 62 | – 9 | – 582 | – 383 | – 1 036 | ||||||
| 2018 | |||||||||||
(USD millions) | Due later than one month but less than three months | Due later than three months but less than one year | Due later than one year but less than five years | Due after five years | Total | ||||||
| Contractual interest on non-current liabilities | – 113 | – 459 | – 1 667 | – 3 755 | – 5 994 | ||||||
| Trade payables | – 5 556 | – 5 556 | |||||||||
| Contingent consideration liabilities | – 98 | – 470 | – 339 | – 907 | |||||||
| (USD millions) | 2019 | 2018 | |||
| All financial instruments | 355 | 337 | |||
| Analyzed by components: | |||||
| Instruments sensitive to foreign currency exchange rates | 89 | 217 | |||
| Instruments sensitive to equity market movements | 31 | 122 | |||
| Instruments sensitive to interest rates | 187 | 221 | |||
| 2019 | |||||||
| (USD millions) | Average | High | Low | ||||
| All financial instruments | 348 | 385 | 303 | ||||
| Analyzed by components: | |||||||
| Instruments sensitive to foreign currency exchange rates | 143 | 195 | 86 | ||||
| Instruments sensitive to equity market movements | 36 | 81 | 16 | ||||
| Instruments sensitive to interest rates | 233 | 303 | 187 | ||||
| 2018 | |||||||
| (USD millions) | Average | High | Low | ||||
| All financial instruments | 443 | 553 | 337 | ||||
| Analyzed by components: | |||||||
| Instruments sensitive to foreign currency exchange rates | 324 | 473 | 217 | ||||
| Instruments sensitive to equity market movements | 60 | 122 | 22 | ||||
| Instruments sensitive to interest rates | 253 | 361 | 169 | ||||
| (USD millions) | 2019 1 | 2018 | 2017 | ||||
| Net sales to third parties from discontinued operations | 1 777 | 7 149 | 6 771 | ||||
| Sales to continuing segments | 32 | 4 | 3 | ||||
| Net sales from discontinued operations | 1 809 | 7 153 | 6 774 | ||||
| Other revenues | 3 | ||||||
| Cost of goods sold | – 860 | – 3 983 | – 3 588 | ||||
| Gross profit from discontinued operations | 949 | 3 170 | 3 189 | ||||
| Selling, general and administration | – 638 | – 2 754 | – 2 532 | ||||
| Research and development | – 142 | – 585 | – 583 | ||||
| Other income | 15 | 61 | 47 | ||||
| Other expense | – 113 | – 126 | – 194 | ||||
| Operating income/(loss) from discontinued operations | 71 | – 234 | – 73 | ||||
| Interest expense | – 10 | – 25 | – 27 | ||||
| Other financial income and expense | – 3 | – 1 | – 3 | ||||
| Income/(loss) before taxes from discontinued operations | 58 | – 260 | – 103 | ||||
| Taxes | – 159 | 74 | 307 | ||||
| Net (loss)/income from discontinued operations before gain on distribution of Alcon Inc. to Novartis AG shareholders | – 101 | – 186 | 204 | ||||
Gain on distribution of Alcon Inc. to Novartis AG shareholders 2 | 4 691 | ||||||
| Net income/(loss) from discontinued operations | 4 590 | – 186 | 204 | ||||
1 The consolidated income statement amounts are for the period from January 1, 2019, to the completion of the spin-off. | |||||||
2 See Note 2 for further details on the non-taxable non-cash gain on distribution of Alcon Inc. to Novartis AG shareholders. | |||||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Interest income | 2 | ||||||
| Depreciation of property, plant and equipment | – 42 | – 235 | – 217 | ||||
Depreciation of right-of-use assets 1 | – 9 | ||||||
| Amortization of intangible assets | – 174 | – 1 052 | – 1 066 | ||||
| Impairment charges on property, plant and equipment | – 3 | ||||||
Impairment charges on intangible assets 2 | – 391 | – 57 | |||||
| Additions to restructuring provisions | – 13 | – 8 | |||||
| Equity-based compensation of Novartis equity plans | – 9 | – 93 | – 71 | ||||
1 Depreciation of right-of-use assets recognized from January 1, 2019, the date of implementation of IFRS 16 leases. See Note 1 for additional disclosures. | |||||||
2 2018 includes an impairment of USD 337 million related to the write-down of the CyPass currently marketed product, which was acquired with the Alcon Division 2016 acquisition of Transcend Medical, Inc. | |||||||
| (USD millions) | 2019 | 2018 | |||
| Additions to property, plant and equipment | 113 | 519 | |||
Additions to right-of-use assets 1 | 3 | ||||
| Additions to goodwill and intangible assets | 36 | 196 | |||
1 Additions to right-of-use assets recognized in 2019 with the implementation of IFRS 16 Leases on January 1, 2019. See Note 1 for additional disclosures. | |||||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Payments out of provisions for transaction costs attributable to the spin-off of the Alcon business | – 29 | ||||||
| Divested cash and cash equivalents | – 628 | ||||||
| Cash flows attributable to the spin-off of the Alcon business | – 657 | ||||||
| Other cash flows used in investing activities, net | – 502 | – 1 001 | – 775 | ||||
| Net cash flows used in investing activities from discontinued operations | – 1 159 | – 1 001 | – 775 | ||||
| 2018 | |||||||||||
(USD millions) | Total future payments | Unearned finance income | Present value | Provision | Net book value | ||||||
Not later than one year 1 | 64 | – 5 | 59 | – 2 | 57 | ||||||
| Between one and five years | 117 | – 9 | 108 | – 28 | 80 | ||||||
| Later than five years | 48 | – 2 | 46 | – 35 | 11 | ||||||
| Total | 229 | – 16 | 213 | – 65 | 148 | ||||||
1 The current portion of the minimum lease payments was recorded in trade receivables or other current assets (to the extent not invoiced). | |||||||||||
| (USD millions) | 2019 | ||
| Property, plant and equipment | 2 858 | ||
| Right-of-use assets | 269 | ||
| Goodwill | 8 906 | ||
| Intangible assets other than goodwill | 11 121 | ||
| Deferred tax assets | 732 | ||
| Financial and other non-current assets | 526 | ||
| Inventories | 1 469 | ||
| Trade receivables and other current assets | 1 787 | ||
| Cash and cash equivalents | 628 | ||
| Deferred tax liabilities | – 1 713 | ||
| Current and non-current lease liabilities | – 269 | ||
| Current and non-current financial debts | – 3 538 | ||
| Trade payables, provisions and other liabilities | – 2 751 | ||
| Net assets derecognized | 20 025 | ||
| (USD millions) | 2019 | 2018 | 2017 | ||||
| Contributions for defined contribution plans discontinued operations | 33 | 104 | 99 | ||||
As at December 31, 2019 | Share capital 1 | Equity interest | |||||||
| Algeria | |||||||||
| Société par actions SANDOZ | Algiers | DZD | 650.0 | m | 100% | ||||
| Argentina | |||||||||
| Novartis Argentina S.A. | Buenos Aires | ARS | 906.1 | m | 100% | ||||
| Australia | |||||||||
| Novartis Australia Pty Ltd | Macquarie Park, NSW | AUD | 2 | 100% | |||||
| Novartis Pharmaceuticals Australia Pty Ltd | Macquarie Park, NSW | AUD | 3.8 | m | 100% | ||||
| Sandoz Pty Ltd | Macquarie Park, NSW | AUD | 11.6 | m | 100% | ||||
| Austria | |||||||||
| Novartis Austria GmbH | Vienna | EUR | 1.0 | m | 100% | ||||
| Novartis Pharma GmbH | Vienna | EUR | 1.1 | m | 100% | ||||
| Sandoz GmbH | Kundl | EUR | 32.7 | m | 100% | ||||
| EBEWE Pharma Ges.m.b.H Nfg. KG | Unterach am Attersee | EUR | 1.0 | m | 100% | ||||
| Bangladesh | |||||||||
| Novartis (Bangladesh) Limited | Gazipur | BDT | 162.5 | m | 60% | ||||
| Belgium | |||||||||
| Novartis Pharma NV | Vilvoorde | EUR | 7.1 | m | 100% | ||||
| Sandoz NV | Vilvoorde | EUR | 19.2 | m | 100% | ||||
| Alcon - Couvreur NV | Puurs | EUR | 110.6 | m | 100% | ||||
| Bermuda | |||||||||
| Novartis Investment Ltd. | Hamilton 3 | USD | 12 000 | 100% | |||||
| Novartis Securities Investment Ltd. | Hamilton | CHF | 30 000 | 100% | |||||
| Novartis Finance Services Ltd. | Hamilton | CHF | 20 000 | 100% | |||||
| Triangle International Reinsurance Limited | Hamilton | CHF | 1.0 | m | 100% | ||||
| Trinity River Insurance Co Ltd. | Hamilton | USD | 370 000 | 100% | |||||
| Brazil | |||||||||
| Novartis Biociências S.A. | São Paulo | BRL | 265.0 | m | 100% | ||||
| Sandoz do Brasil Indústria Farmacêutica Ltda. | Cambé, PR | BRL | 190.0 | m | 100% | ||||
| Canada | |||||||||
| Novartis Pharmaceuticals Canada Inc. | Dorval, Quebec | CAD | 1.2 | m | 100% | ||||
| Sandoz Canada Inc. | Boucherville, Quebec | CAD | 80.8 | m | 100% | ||||
| CIBA Vision Canada Inc. | Mississauga, Ontario | CAD | 82 886 | 100% | |||||
| Chile | |||||||||
| Novartis Chile S.A. | Santiago de Chile | CLP | 2.0 | bn | 100% | ||||
| China | |||||||||
| Beijing Novartis Pharma Co., Ltd. | Beijing | USD | 30.0 | m | 100% | ||||
| Novartis Pharmaceuticals (HK) Limited | Hong Kong | HKD | 200 | 100% | |||||
| China Novartis Institutes for BioMedical Research Co., Ltd. | Shanghai | USD | 320.0 | m | 100% | ||||
| Suzhou Novartis Technical Development Co., Ltd. | Changshu | USD | 12.0 | m | 100% | ||||
| Shanghai Novartis Trading Ltd. | Shanghai | USD | 3.2 | m | 100% | ||||
| Sandoz (China) Pharmaceutical Co., Ltd. | Zhongshan | USD | 57.6 | m | 100% | ||||
| Colombia | |||||||||
| Novartis de Colombia S.A. | Santafé de Bogotá | COP | 7.9 | bn | 100% | ||||
| Croatia | |||||||||
| Sandoz d.o.o. farmaceutska industrija | Zagreb | HRK | 25.6 | m | 100% | ||||
| Czech Republic | |||||||||
| Novartis s.r.o. | Prague | CZK | 51.5 | m | 100% | ||||
| Sandoz s.r.o. | Prague | CZK | 44.7 | m | 100% | ||||
| Denmark | |||||||||
| Novartis Healthcare A/S | Copenhagen | DKK | 14.0 | m | 100% | ||||
| Sandoz A/S | Copenhagen | DKK | 12.0 | m | 100% | ||||
| Ecuador | |||||||||
| Novartis Ecuador S.A. | Quito | USD | 4.0 | m | 100% | ||||
| Egypt | |||||||||
| Novartis Pharma S.A.E. | Cairo | EGP | 193.8 | m | 99.77% | ||||
| Sandoz Egypt Pharma S.A.E. | New Cairo City | EGP | 250 000 | 100% | |||||
| Finland | |||||||||
| Novartis Finland Oy | Espoo | EUR | 459 000 | 100% | |||||
As at December 31, 2019 | Share capital 1 | Equity interest | |||||||
| France | |||||||||
| Novartis Groupe France S.A. | Rueil-Malmaison | EUR | 903.0 | m | 100% | ||||
| Novartis Pharma S.A.S. | Rueil-Malmaison | EUR | 43.4 | m | 100% | ||||
| Advanced Accelerator Applications S.A. | Saint-Genis-Pouilly | EUR | 9.6 | m | 99.2% | ||||
| CELLforCURE | Les Ulis | EUR | 4.2 | m | 100% | ||||
| Sandoz S.A.S. | Levallois-Perret | EUR | 5.4 | m | 100% | ||||
| Germany | |||||||||
| Novartis Deutschland GmbH | Nuremberg | EUR | 155.5 | m | 100% | ||||
| Novartis Pharma GmbH | Nuremberg | EUR | 25.6 | m | 100% | ||||
| Novartis Pharma Produktions GmbH | Wehr | EUR | 2.0 | m | 100% | ||||
| Novartis Manufacturing GmbH | Marburg | EUR | 25 000 | 100% | |||||
| Sandoz International GmbH | Holzkirchen | EUR | 100 000 | 100% | |||||
| 1 A Pharma GmbH | Oberhaching | EUR | 26 000 | 100% | |||||
| HEXAL AG | Holzkirchen | EUR | 93.7 | m | 100% | ||||
| Salutas Pharma GmbH | Barleben | EUR | 42.1 | m | 100% | ||||
| Aeropharm GmbH | Rudolstadt | EUR | 26 000 | 100% | |||||
| Greece | |||||||||
| Novartis (Hellas) S.A.C.I. | Metamorphosis / Athens | EUR | 23.4 | m | 100% | ||||
| Hungary | |||||||||
| Novartis Hungary Healthcare Limited Liability Company | Budapest | HUF | 545.6 | m | 100% | ||||
| Sandoz Hungary Limited Liability Company | Budapest | HUF | 883.0 | m | 100% | ||||
| India | |||||||||
| Novartis India Limited | Mumbai | INR | 123.5 | m | 70.68% | ||||
| Novartis Healthcare Private Limited | Mumbai | INR | 60.0 | m | 100% | ||||
| Sandoz Private Limited | Mumbai | INR | 32.0 | m | 100% | ||||
| Indonesia | |||||||||
| PT. Novartis Indonesia | Jakarta | IDR | 7.7 | bn | 100% | ||||
| Ireland | |||||||||
| Novartis Ireland Limited | Dublin | EUR | 25 000 | 100% | |||||
| Novartis Ringaskiddy Limited | Ringaskiddy, County Cork | EUR | 2.0 | m | 100% | ||||
| Israel | |||||||||
| Novartis Israel Ltd. | Tel Aviv | ILS | 1 000 | 100% | |||||
| Italy | |||||||||
| Novartis Farma S.p.A. | Origgio | EUR | 18.2 | m | 100% | ||||
| Advanced Accelerator Applications (Italy) S.r.l. | Pozzilli | EUR | 119 000 | 99.2% | |||||
| Sandoz S.p.A. | Origgio | EUR | 1.7 | m | 100% | ||||
| Japan | |||||||||
| Novartis Holding Japan K.K. | Tokyo | JPY | 10.0 | m | 100% | ||||
| Novartis Pharma K.K. | Tokyo | JPY | 6.0 | bn | 100% | ||||
| Ciba-Geigy Japan Limited | Tokyo | JPY | 8.5 | m | 100% | ||||
| Sandoz K.K. | Tokyo | JPY | 100.0 | m | 100% | ||||
| Latvia | |||||||||
| Novartis Baltics SIA | Riga | EUR | 3.0 | m | 100% | ||||
| Luxembourg | |||||||||
| Novartis Investments S.à r.l. | Luxembourg City | USD | 100.0 | m | 100% | ||||
| Novartis Finance S.A. | Luxembourg City | USD | 100 000 | 100% | |||||
| Malaysia | |||||||||
| Novartis Corporation (Malaysia) Sdn. Bhd. | Kuala Lumpur | MYR | 3.3 | m | 100% | ||||
| Mexico | |||||||||
| Novartis Farmacéutica, S.A. de C.V. | Mexico City | MXN | 205.0 | m | 100% | ||||
| Sandoz, S.A. de C.V. | Mexico City | MXN | 468.2 | m | 100% | ||||
| Morocco | |||||||||
| Novartis Pharma Maroc SA | Casablanca | MAD | 80.0 | m | 100% | ||||
| Netherlands | |||||||||
| Novartis Netherlands B.V. | Amsterdam | EUR | 1.4 | m | 100% | ||||
| Novartis Pharma B.V. | Amsterdam | EUR | 4.5 | m | 100% | ||||
| IDB Holland BV | Baarle-Nassau | EUR | 18 000 | 99.2% | |||||
| Sandoz B.V. | Almere | EUR | 907 560 | 100% | |||||
| New Zealand | |||||||||
| Novartis New Zealand Ltd | Auckland | NZD | 820 000 | 100% | |||||
As at December 31, 2019 | Share capital 1 | Equity interest | |||||||
| Norway | |||||||||
| Novartis Norge AS | Oslo | NOK | 1.5 | m | 100% | ||||
| Pakistan | |||||||||
| Novartis Pharma (Pakistan) Limited | Karachi | PKR | 6.7 | bn | 99.99% | ||||
| Panama | |||||||||
| Novartis Pharma (Logistics), Inc. | Panama City | USD | 10 000 | 100% | |||||
| Peru | |||||||||
| Novartis Biosciences Perú S.A. | Lima | PEN | 6.1 | m | 100% | ||||
| Philippines | |||||||||
| Novartis Healthcare Philippines, Inc. | Makati City | PHP | 298.8 | m | 100% | ||||
| Sandoz Philippines Corporation | Makati City | PHP | 30.0 | m | 100% | ||||
| Poland | |||||||||
| Novartis Poland Sp. z o.o. | Warsaw | PLN | 44.2 | m | 100% | ||||
| Sandoz Polska Sp. z o.o. | Warsaw | PLN | 25.6 | m | 100% | ||||
| Lek S.A. | Strykow | PLN | 11.4 | m | 100% | ||||
| Portugal | |||||||||
| Novartis Portugal SGPS Lda. | Porto Salvo | EUR | 500 000 | 100% | |||||
| Novartis Farma - Produtos Farmacêuticos S.A. | Porto Salvo | EUR | 2.4 | m | 100% | ||||
| Sandoz Farmacêutica Lda. | Porto Salvo | EUR | 499 900 | 100% | |||||
| Romania | |||||||||
| Novartis Pharma Services Romania S.R.L. | Bucharest | RON | 3.0 | m | 100% | ||||
| Sandoz S.R.L. | Targu-Mures | RON | 105.2 | m | 100% | ||||
| Russian Federation | |||||||||
| Novartis Pharma LLC | Moscow | RUB | 20.0 | m | 100% | ||||
| Novartis Neva LLC | St. Petersburg | RUB | 500.0 | m | 100% | ||||
| ZAO Sandoz | Moscow | RUB | 57.4 | m | 100% | ||||
| Saudi Arabia | |||||||||
| Novartis Saudi Ltd. | Riyadh | SAR | 26.8 | m | 75% | ||||
| Singapore | |||||||||
| Novartis (Singapore) Pte Ltd. | Singapore | SGD | 100 000 | 100% | |||||
| Novartis Singapore Pharmaceutical Manufacturing Pte Ltd | Singapore | SGD | 45.0 | m | 100% | ||||
| Novartis Asia Pacific Pharmaceuticals Pte Ltd | Singapore | SGD | 39.0 | m | 100% | ||||
| Slovakia | |||||||||
| Novartis Slovakia s.r.o. | Bratislava | EUR | 2.0 | m | 100% | ||||
| Slovenia | |||||||||
| Lek Pharmaceuticals d.d. | Ljubljana | EUR | 48.4 | m | 100% | ||||
| Sandoz Pharmaceuticals d.d. | Ljubljana | EUR | 1.5 | m | 100% | ||||
| South Africa | |||||||||
| Novartis South Africa (Pty) Ltd | Midrand | ZAR | 86.3 | m | 100% | ||||
| Sandoz South Africa (Pty) Ltd | Kempton Park | ZAR | 3.0 | m | 100% | ||||
| South Korea | |||||||||
| Novartis Korea Ltd. | Seoul | KRW | 24.5 | bn | 98.55% | ||||
| Spain | |||||||||
| Novartis Farmacéutica, S.A. | Barcelona | EUR | 63.0 | m | 100% | ||||
| Advanced Accelerator Applications Iberica, S.L.U. | Esplugues de Llobregat | EUR | 22.6 | m | 99.2% | ||||
| Sandoz Farmacéutica S.A. | Madrid | EUR | 270 450 | 100% | |||||
| Sandoz Industrial Products S.A. | Les Franqueses del Vallés / Barcelona | EUR | 9.3 | m | 100% | ||||
| Alcon Cusi S.A. | El Masnou / Barcelona | EUR | 10.1 | m | 100% | ||||
| Abadia Retuerta S.A. | Sardón de Duero / Valladolid | EUR | 6.0 | m | 100% | ||||
| Sweden | |||||||||
| Novartis Sverige AB | Stockholm | SEK | 5.0 | m | 100% | ||||
| Switzerland | |||||||||
| Novartis International AG | Basel | CHF | 10.0 | m | 100% | ||||
| Novartis Holding AG | Basel 3 | CHF | 100.2 | m | 100% | ||||
| Novartis International Pharmaceutical Investment AG | Basel | CHF | 100 000 | 100% | |||||
| Novartis Bioventures AG | Basel | CHF | 100 000 | 100% | |||||
| Novartis Forschungsstiftung | Basel | -- | -- | 100% | |||||
| Novartis Stiftung für Kaderausbildung | Basel | -- | -- | 100% | |||||
| Novartis Mitarbeiterbeteiligungsstiftung | Basel | -- | -- | 100% | |||||
| Novartis Stiftung für Mensch und Umwelt | Basel | -- | -- | 100% | |||||
| Stiftung der Novartis AG für Erziehung, Ausbildung und Bildung | Basel | -- | -- | 100% | |||||
| Novartis Overseas Investments AG | Basel | CHF | 1.0 | m | 100% | ||||
| Japat AG | Basel | CHF | 50 000 | 100% | |||||
| Novartis Pharma AG | Basel 3 | CHF | 350.0 | m | 100% | ||||
| Novartis International Pharmaceutical AG | Basel 3 | CHF | 100 000 | 100% | |||||
| Novartis Pharma Services AG | Basel | CHF | 20.0 | m | 100% | ||||
| Novartis Pharma Schweizerhalle AG | Muttenz | CHF | 18.9 | m | 100% | ||||
| Novartis Pharma Stein AG | Stein | CHF | 251 000 | 100% | |||||
| Novartis Pharma Schweiz AG | Risch | CHF | 5.0 | m | 100% | ||||
| Novartis Ophthalmics AG | Fribourg | CHF | 100 000 | 100% | |||||
| Advanced Accelerator Applications International SA | Geneva | CHF | 9.3 | m | 99.2% | ||||
| Sandoz AG | Basel | CHF | 5.0 | m | 100% | ||||
| Sandoz Pharmaceuticals AG | Risch | CHF | 100 000 | 100% | |||||
| Roche Holding AG | Basel | CHF | 160.0 | m | 33/6 2 | ||||
As at December 31, 2019 | Share capital 1 | Equity interest | |||||||
| Taiwan | |||||||||
| Novartis (Taiwan) Co., Ltd. | Taipei | TWD | 170.0 | m | 100% | ||||
| Thailand | |||||||||
| Novartis (Thailand) Limited | Bangkok | THB | 302.0 | m | 100% | ||||
| Turkey | |||||||||
| Novartis Saglik, Gida ve Tarim Ürünleri Sanayi ve Ticaret A.S. | Istanbul | TRY | 98.0 | m | 100% | ||||
| Farmanova Saglik Hizmetleri Ltd. Sti. | Istanbul | TRY | 6.7 | m | 100% | ||||
| Sandoz Ilaç Sanayi ve Ticaret A.S. | Istanbul | TRY | 165.2 | m | 99.99% | ||||
| Sandoz Grup Saglik Ürünleri Ilaçlari Sanayi ve Ticaret A.S. | Gebze - Kocaeli | TRY | 50.0 | m | 100% | ||||
| Ukraine | |||||||||
| Sandoz Ukraine LLC | Kyiv | UAH | 8.0 | m | 100% | ||||
| United Arab Emirates | |||||||||
| Novartis Middle East FZE | Dubai | AED | 7.0 | m | 100% | ||||
| United Kingdom | |||||||||
| Novartis UK Limited | London | GBP | 25.5 | m | 100% | ||||
| Novartis Pharmaceuticals UK Limited | London | GBP | 5.4 | m | 100% | ||||
| Novartis Grimsby Limited | London | GBP | 250.0 | m | 100% | ||||
| Imaging Equipment Ltd | London | GBP | 100 | 99.2% | |||||
| Vivacta Limited | Frimley / Camberley | GBP | 2.9 | m | 100.0% | ||||
| Ziarco Group Limited | London | GBP | 3 904 | 100% | |||||
| Sandoz Limited | Frimley / Camberley | GBP | 2.0 | m | 100% | ||||
| United States of America | |||||||||
| Novartis Corporation | East Hanover, NJ | USD | 72.2 | m | 100% | ||||
| Novartis Finance Corporation | East Hanover, NJ 3 | USD | 1 000 | 100% | |||||
| Novartis Capital Corporation | East Hanover, NJ | USD | 1 | 100% | |||||
| Novartis Services, Inc. | East Hanover, NJ | USD | 1 | 100% | |||||
| Novartis US Foundation | East Hanover, NJ | -- | -- | 100% | |||||
| Novartis Pharmaceuticals Corporation | East Hanover, NJ | USD | 650 | 100% | |||||
| Advanced Accelerator Applications USA, Inc. | Millburn, NJ | USD | 1 | 99.2% | |||||
| AveXis, Inc. | Bannockburn, IL | USD | 1 | 100% | |||||
| Novartis Inflammasome Research, Inc. | East Hanover, NJ | USD | 1 | 100% | |||||
| Novartis Technology LLC | East Hanover, NJ | -- | -- | 100% | |||||
| Novartis Institutes for BioMedical Research, Inc. | Cambridge, MA | USD | 1 | 100% | |||||
| CoStim Pharmaceuticals Inc. | Cambridge, MA | USD | 1 | 100% | |||||
| Encore Vision, Inc. | East Hanover, NJ | USD | 1 | 100% | |||||
| Endocyte, Inc. | East Hanover, NJ | USD | 1 | 100% | |||||
| Navigate BioPharma Services, Inc. | Carlsbad, CA | USD | 1 | 100% | |||||
| Reprixys Pharmaceuticals Corporation | East Hanover, NJ | USD | 1 | 100% | |||||
| Spinifex Pharmaceuticals, Inc. | East Hanover, NJ | USD | 1 | 100% | |||||
| Novartis Institute for Functional Genomics, Inc. | San Diego, CA | USD | 1 000 | 100% | |||||
| Sandoz Inc. | Princeton, NJ | USD | 25 000 | 100% | |||||
| Oriel Therapeutics, Inc. | Durham, NC | USD | 1 | 100% | |||||
| Fougera Pharmaceuticals Inc. | Melville, NY | USD | 1 | 100% | |||||
| Eon Labs, Inc. | Princeton, NJ | USD | 1 | 100% | |||||
| Novartis Vaccines and Diagnostics, Inc. | East Hanover, NJ | USD | 3 | 100% | |||||
| Venezuela | |||||||||
| Novartis de Venezuela, S.A. | Caracas | VES | 14 | 100% | |||||
| In addition, the Group is represented by subsidiaries and associated companies with total assets or net sales to third parties below USD 25 million in the following countries: Bosnia/Herzegovina, Bulgaria, Dominican Republic, Guatemala, Kenya, North Macedonia, Nigeria, Puerto Rico, Uruguay and Vietnam | |||||||||
1 Share capital may not reflect the taxable share capital and does not include any paid-in surplus. | |||||||||
2 Approximately 33% of voting shares; approximately 6% of total net income and equity attributable to Novartis. | |||||||||
3 Significant subsidiary under SEC Regulation S-X Rule 1-02(w) | |||||||||
| m = million; bn = billion | |||||||||
Report of Independent Registered Public Accounting Firm
