Filed by: Caliper Life Sciences, Inc.
Pursuant to Rule 425 under the Securities Act of 1933
and deemed filed pursuant to Rule 14a-12 of the
Securities Exchange Act of 1934
Subject Company: Xenogen Corporation
Exchanges Act File No. 000-32239
Pursuant to Rule 425 under the Securities Act of 1933
and deemed filed pursuant to Rule 14a-12 of the
Securities Exchange Act of 1934
Subject Company: Xenogen Corporation
Exchanges Act File No. 000-32239
Important Notice:
Caliper Life Sciences, Inc. intends to file a registration statement on Form S-4 in order to register shares of its common stock and warrants to be issued to the former stockholders of Xenogen Corporation in the proposed merger between Caliper and Xenogen., and Caliper and Xenogen will be filing a joint proxy statement with the Securities and Exchange Commission. Investors and security holders of Caliper and Xenogen are advised to read the registration statement on Form S-4 and the joint proxy statement regarding the proposed merger when they become available, because they will contain important information. Caliper and Xenogen expect to mail the joint proxy statement about the proposed merger to their respective stockholders. Investors and security holders may obtain a free copy of the proxy statement and any other documents filed by Caliper and Xenogen at the Securities and Exchange Commission’s website at http://www.sec.gov or directly from Caliper and Xenogen.
Caliper Life Sciences, Inc. intends to file a registration statement on Form S-4 in order to register shares of its common stock and warrants to be issued to the former stockholders of Xenogen Corporation in the proposed merger between Caliper and Xenogen., and Caliper and Xenogen will be filing a joint proxy statement with the Securities and Exchange Commission. Investors and security holders of Caliper and Xenogen are advised to read the registration statement on Form S-4 and the joint proxy statement regarding the proposed merger when they become available, because they will contain important information. Caliper and Xenogen expect to mail the joint proxy statement about the proposed merger to their respective stockholders. Investors and security holders may obtain a free copy of the proxy statement and any other documents filed by Caliper and Xenogen at the Securities and Exchange Commission’s website at http://www.sec.gov or directly from Caliper and Xenogen.
Caliper and its officers and directors may be deemed to be participants in the solicitation of proxies from stockholders of Caliper with respect to the proposed merger with Xenogen. Information regarding such officers and directors is included in Caliper’s annual report on Form 10-K for the fiscal year ended December 31, 2004 and in its proxy statement for its 2005 annual meeting filed with the SEC. This document is available free of charge at the Securities and Exchange Commission’s website at http://www.sec.gov or directly from Caliper.
Mr. Kevin Hrusovsky, the President and Chief Executive Officer of Caliper Life Sciences, Inc. (“Caliper”), and Mr. Thomas Higgins, the Executive Vice President and Chief Financial Officer of Caliper, plan to conduct a series of meetings with existing stockholders of Caliper and potential investors in Caliper to discuss Caliper and its business as well as Caliper’s proposed business combination with Xenogen Corporation. The following is a copy of the material that will be used by Mr. Hrusovsky and Mr. Higgins during these meetings.

| Transforming Life Sciences through Innovative Technologies Kevin Hrusovsky, President & CEO March 1, 2006 |

| Forward-Looking Statements This presentation includes forward-looking statements, including financial projections. Reference to what we expect, believe, intend to do, plan, hope, estimate or other statements referring to future events or results are intended to identify those as forward- looking statements. Forward-looking statements are inherently subject to risks and uncertainties. Further information on risks faced by Caliper are included under the caption Factors Affecting Operating Results in Caliper's quarterly report on Form 10-Q, filed with the Securities and Exchange Commission on November 9, 2005, and in our other SEC reports. These SEC filings are available on the website maintained by the Securities and Exchange Commission at www.SEC.gov. Caliper does not undertake any obligation to update forward-looking or other statements in this presentation to reflect any change in Caliper's expectations with regards to such statements or any change in events, conditions or circumstances on which any such statements are based. |

| Important Notice In addition to the foregoing, please note that Caliper intends to file a Registration Statement on Form S-4 in order to register the shares of its common stock and warrants to be issued to the former stockholders of Xenogen Corporation in the proposed merger between Caliper and Xenogen, and Caliper and Xenogen will be filing a joint proxy statement with the Securities and Exchange Commission. Investors and security holders of Caliper and Xenogen are advised to read the Registration Statement on Form S-4 and the joint proxy statement regarding the proposed merger when they become available because they will contain important information. Caliper and Xenogen expect to mail the joint proxy statement about the proposed merger to their respective stockholders. Investors and security holders may obtain a free copy of the proxy statement and any other documents filed by Caliper and Xenogen at the Securities and Exchange Commission's web site at http://www.sec.gov or directly from Caliper and Xenogen. Caliper and its officers and directors may be deemed to be participants in the solicitation of proxies from stockholders of Caliper with respect to the proposed merger with Xenogen. Information regarding such officers and directors is included in Caliper's Annual Report on Form 10-K for the fiscal year ended December 31, 2004 and in its proxy statement for its 2005 annual meeting, filed with the Securities and Exchange Commission. This document is available free of charge at the Securities and Exchange Commission's web site at http://www.sec.gov and directly from Caliper. |

| 2005: Another Year of Strong Financial Performance Exceeded Q4'05 goal of break even operating cash flow, $3M+ Exceeded year end cash goal with $32M vs. $25M street goal Achieved Q4 revenue guidance with $27M, 12% growth Exceeded EPS street consensus Completed accretive NS acquisition and announced Xenogen deal (Feb) Major agreements: Agilent, Affy, Bio-Rad, Cougar, Amphora ($60M, 5yr) Achieved strong direct channel LabChip adoption (over 100 installed - approx. 80% growth) |

| Building on Momentum Towards an Ambitious 2006 2003 - 2005 Accomplishments $35M EBITDA and $250M enterprise value gain Revenue growth 175%, reversed LabChip negative growth to positive 15%+ Cost Savings: $25M Revitalized R&D investment, target 18% of sales (40% funded) Mix shift toward aftermarket; 20% to 35% in '05 Met or exceeded all key financial measures for nine straight quarters 2006 Goals Close Xenogen transaction by mid year Q1 revenue forecast of $21 - 23M ( 20% growth) Full year revenue forecast of $120 - $128M, including Xenogen H2 (40% growth) Expect 50% growth in LabChip 3000 / LabChip 90 placements Controlled OPEX spending, targeted margin improvement |
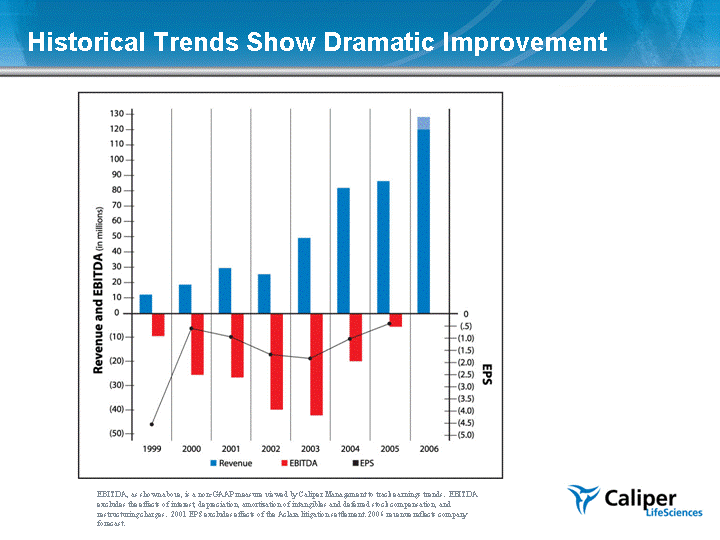
| Historical Trends Show Dramatic Improvement EBITDA, as shown above, is a non-GAAP measure viewed by Caliper Management to track earnings trends. EBITDA excludes the effects of interest, depreciation, amortization of intangibles and deferred stock compensation, and restructuring charges. 2001 EPS excludes effects of the Aclara litigation settlement. 2006 revenue reflects company forecast. |

| February 13, 2006 News: Caliper Life Sciences to Acquire Xenogen Xenogen profile Leader in in-vivo imaging systems and pre-clinical services Strong IP position in bio-photonics imaging 2005 revenues = $39.7M, 28.4% growth from 2004 Why Xenogen? Caliper entry into high-growth optical imaging market Catalyzes transformation to leading-edge drug discovery company bridging in vitro and in vivo experimentation Enhances revenue growth, critical mass and profit trajectory Key parameters All equity deal: 13.2M common shares, 5.1M warrants (5 year, $6.79 strike price) Post closing, Xenogen shareholders = 26% of Caliper stock, 32% fully diluted Value at announcement = $80M |

| The Merger Offers Strategic and Financial Benefits Revenue Synergies Customer synergy: academic / commercial and geographic Total company growth rate expected at 10% - 15% Critical Mass Expands base over 40% (2005 pro forma) Leverage manufacturing, commercial, G&A and aftermarket infrastructure Expect 200 basis point improvement in OPEX and 100 b.p. in gross margin Profitability Identified "high probability" cost synergies = $10 million. EBITDA per share expected accretive in 2006/2007 EPS expected accretive in 2008 Cash flow and Funding Cash resources expected adequate for integration and operations |
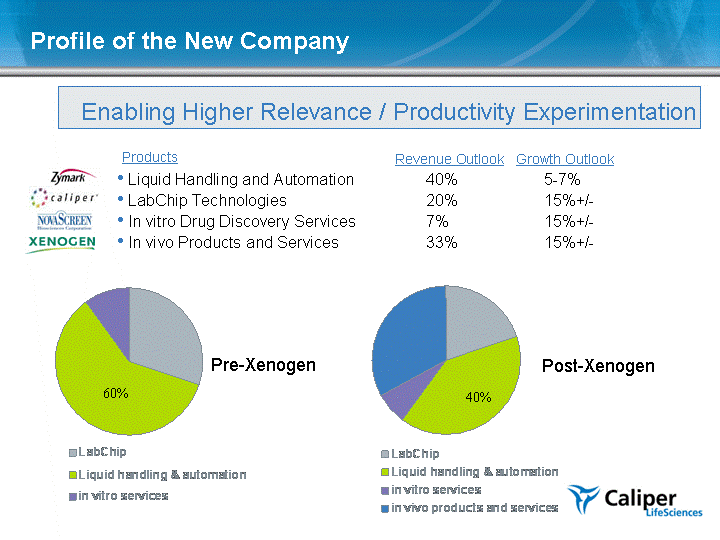
| Enabling Higher Relevance / Productivity Experimentation Products Liquid Handling and Automation LabChip Technologies In vitro Drug Discovery Services In vivo Products and Services Profile of the New Company LabChip Liquid handling & automation in vitro services Pre-Xenogen 30 60 10 Revenue Outlook Growth Outlook 40% 5-7% 20% 15%+/- 7% 15%+/- 33% 15%+/- LabChip Liquid handling & automation in vitro services in vivo products and services 20 40 7 33 Pre-Xenogen Post-Xenogen 40% 60% |

| Enabling Higher Relevance / Productivity Experimentation Markets Drug Discovery and Development $1.7B market, growing 5-8%, approx 5%+ share 2005 revenue approx. $80M; Expect 15%+/- growth in 2006 (40% w/ XGEN) Molecular Diagnostics $1.5B market growing 20%, new entrant 2005 license, contract revenue approx $7M, expect product revenue in '07 Customers Complementary market strength: Commercial represent 80% of Caliper revenue; academic and government represent 80% of Xenogen revenue International distribution capability (35% CALP; 10% XGEN) Profile of the New Company (cont'd) |
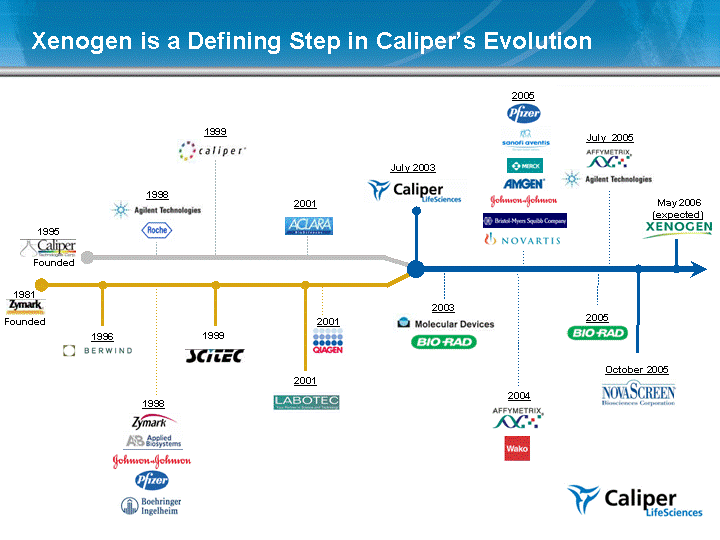
| Xenogen is a Defining Step in Caliper's Evolution Founded 1981 1998 1999 Founded 1998 1999 2001 July 2003 2003 October 2005 1995 2001 1996 2004 July 2005 2005 2001 2005 May 2006 (expected) |

| Blue-Chip Customers have Guided Caliper's Development Top 15 customers are less than 50% of total revenues Largest customer is 10% of total revenues Human Genome Project Representative list of top 15 customers |

| Our Customers Face Declining Productivity "Low hanging fruit" exhausted Current opportunities require more complex biology/MOA Diseases for investigation are more complex Regulatory oversight is more stringent Primary failure mode: Safety & Side Effects # NME Approvals Productivity Gap Sources: FDA, NIH, PhRMA, BIO, Frost & Sullivan $Billions Total R&D Spending vs. NME Approvals NIH Spending Biotech R&D Pharma R&D NME Approvals More causes to fail than succeed |
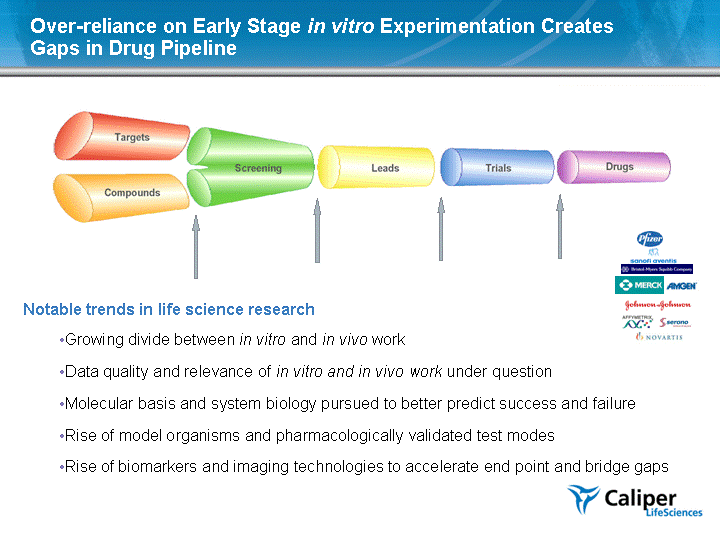
| Over-reliance on Early Stage in vitro Experimentation Creates Gaps in Drug Pipeline Notable trends in life science research Growing divide between in vitro and in vivo work Data quality and relevance of in vitro and in vivo work under question Molecular basis and system biology pursued to better predict success and failure Rise of model organisms and pharmacologically validated test modes Rise of biomarkers and imaging technologies to accelerate end point and bridge gaps |

| Life Science Researchers are Focused on Bridging in vitro and in vivo Experimentation to Close these Gaps "We want as much consistency from in vitro to in vivo studies as possible, so we can be assured that what we see happening in the test tube is the same as what we see in animals. For example, we want to be able to use the same biomarker both in vitro and in vivo." Research Scientist, Major Pharmaceutical Company |
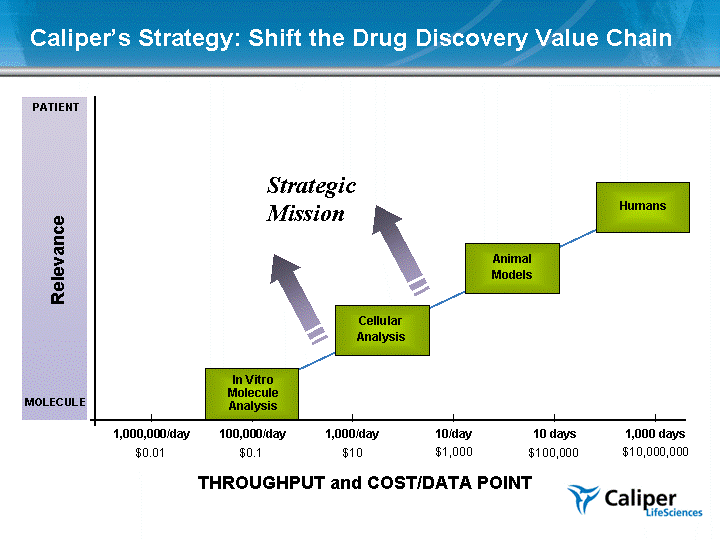
| In Vitro Molecule Analysis Cellular Analysis Humans Animal Models 1,000 days 10 days 10/day 1,000,000/day THROUGHPUT and COST/DATA POINT Relevance 1,000/day $10,000,000 $100,000 $1,000 $10 $0.01 PATIENT MOLECULE Caliper's Strategy: Shift the Drug Discovery Value Chain 100,000/day $0.1 Strategic Mission |

| In Vitro Molecule Analysis Cellular Analysis Humans Animal Models 1,000 days 10 days 10/day 1,000,000/day THROUGHPUT and COST/DATA POINT Relevance 1,000/day $10,000,000 $100,000 $1,000 $10 $0.01 PATIENT MOLECULE Caliper's Strategy: Shift the Drug Discovery Value Chain 100,000/day $0.1 GeneChip Human Primary Cell Zebrafish LPTA HuMouse PlateArrays Human Embryoids Xenogen NovaScreen LabChip Data LabChip Technologies |
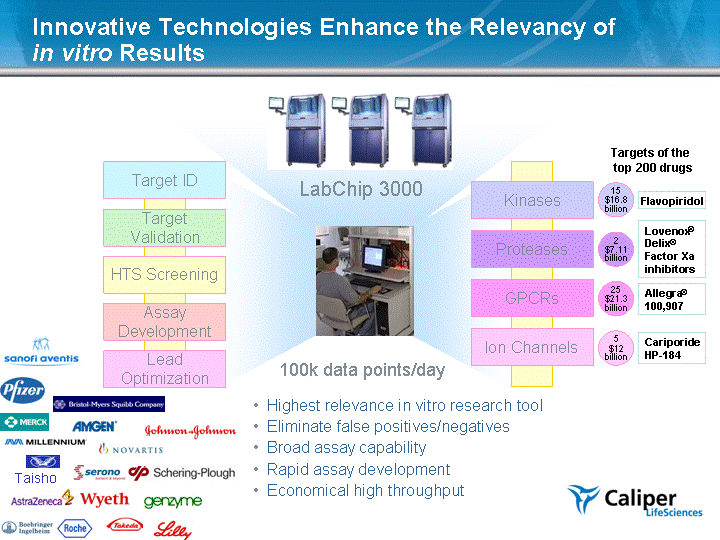
| 100k data points/day Innovative Technologies Enhance the Relevancy of in vitro Results Ion Channels GPCRs Proteases Kinases Allegra (r) 100,907 Flavopiridol Lovenox (r) Delix (r) Factor Xa inhibitors Cariporide HP - - 184 Targets of the top 200 drugs 25 $21.3 billion 15 $16.8 billion 2 $7.11 billion 5 $12 billion Assay Development HTS Screening Target Validation Target ID Lead Optimization LabChip 3000 Highest relevance in vitro research tool Eliminate false positives/negatives Broad assay capability Rapid assay development Economical high throughput Taisho |

| T+ T+ T+ T+ T+ T+ 384 Technology With LabChip 384 Technology |
| T + T+ T+ T+ T+ T+ x1000 plates per day |

| 1997 1998 1999 2000 2001 2002 2003 2004 2005 Number of Citations 5 5 16 47 91 146 260 620 760 Number of Citations Industry Attention to Microfluidic Technology has Accelerated |

| Rapidly Growing LabChip Installed Base Demonstrates Caliper Direct Channel Success *Includes LabChip 3000, Caliper 250, Caliper 220, AMS 90, AMS 90 SE, LabChip 90; excludes Amphora Caliper 250 systems Total Units Placed* Year |

| "Caliper Driven" Program is Expanding the Reach of our Technologies Experion launched in Q4 '04 New system under development Diagnostic license in 2005. Expect product sales in 2007 More than 1.5 million chips sold - New 5 year supply deal 2100 Bioanalyzer over 4000 units installed Launched 5100 ALP in Q1'05 "GCAS" target preparation Commercial launch Q3 '05 |

| Agilent and Bio-Rad Combined Chip Sales Relative to Installed Base *Estimate |
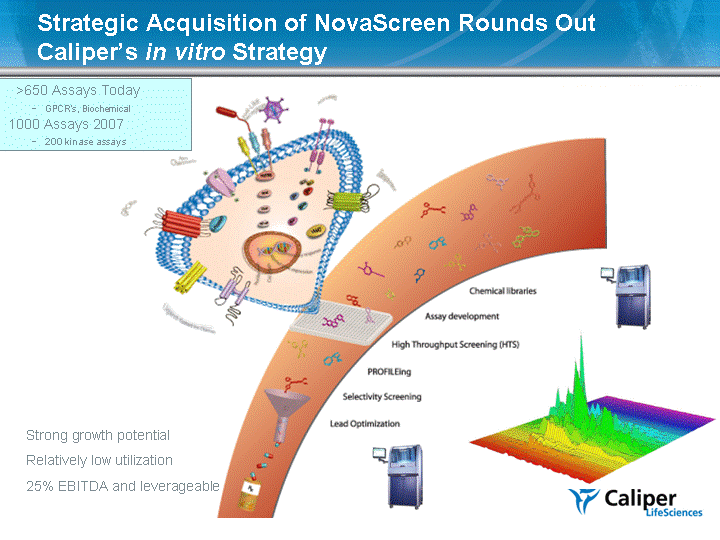
| Strategic Acquisition of NovaScreen Rounds Out Caliper's in vitro Strategy Caliper's in vitro Strategy Caliper's in vitro Strategy >650 Assays Today - GPCR's, Biochemical 1000 Assays 2007 - 200 kinase assays Strong growth potential Relatively low utilization 25% EBITDA and leverageable |

| Xenogen: Advanced Optical Imaging Systems that Accelerate Drug Discovery and Development |

| Xenogen's in vivo Technologies Offer Unique Capabilities for Integrated Molecular Level Solutions |
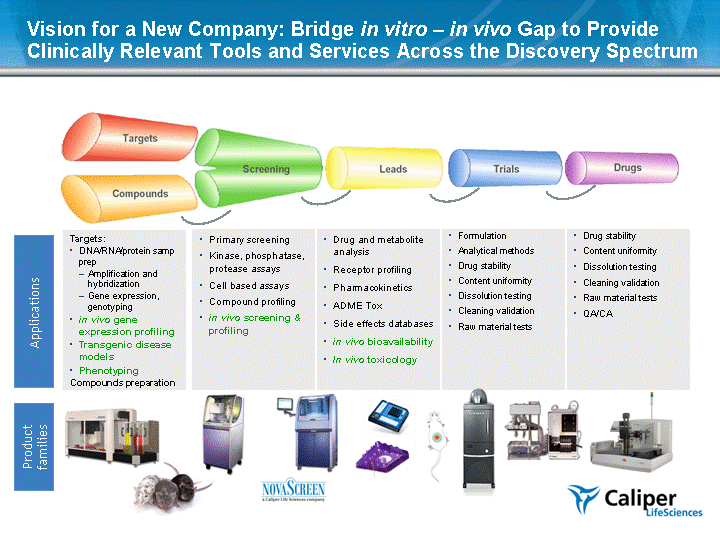
| Vision for a New Company: Bridge in vitro - in vivo Gap to Provide Clinically Relevant Tools and Services Across the Discovery Spectrum Product families Applications Primary screening Kinase, phosphatase, protease assays Cell based assays Compound profiling in vivo screening & profiling Drug and metabolite analysis Receptor profiling Pharmacokinetics ADME Tox Side effects databases in vivo bioavailability In vivo toxicology Formulation Analytical methods Drug stability Content uniformity Dissolution testing Cleaning validation Raw material tests Drug stability Content uniformity Dissolution testing Cleaning validation Raw material tests QA/CA Targets: DNA/RNA/protein samp prep Amplification and hybridization Gene expression, genotyping in vivo gene expression profiling Transgenic disease models Phenotyping Compounds preparation |

| Caliper's 2006 Priorities Close the Xenogen acquisition by mid-year Phased integration of Xenogen operations to achieve cost synergies Exploit distribution channel synergies, customer and geographic, to drive Imaging Products and LabChip revenues Expand reagent strategy and disease models to address targeted research areas Accelerate growth of aftermarket and services 50% growth of LabChip 3000/LabChip 90 placements Controlled OPEX spending, targeted margin improvements Achieve 2006 revenue forecast of $120 - $128 million |

| Financial Summary and Performance 2005 performance is unaudited and presented for illustrative purposes only. EBITDA, as shown above, is a non-GAAP measure viewed by Caliper Management to track earnings trends. EBITDA excludes the effects of interest, depreciation, amortization of intangibles and deferred stock compensation, and restructuring charges. 2001 EPS excludes effects of the Aclara litigation settlement. 2006 revenue reflects company forecast. Value Drivers R&D & patents Commercial abilities Partners / relationships Financial leverage Growth Drivers LabChip products Xenogen products Discovery services Affy/Agilent/BioRad |
