1Filed by: Caliper Life Sciences, Inc.
Pursuant to Rule 425 under the Securities Act of 1933
and deemed filed pursuant to Rule 14a-12 of the
Securities Exchange Act of 1934
Subject Company: Xenogen Corporation
Exchange Act File No. 000-32239
Important Notice:
Caliper Life Sciences, Inc. has filed a registration statement on Form S-4 in order to register the shares of its common stock and warrants to be issued to the former stockholders of Xenogen Corporation in the proposed merger between Caliper and Xenogen, and Caliper and Xenogen will be filing a joint proxy statement with the Securities and Exchange Commission. Investors and security holders of Caliper and Xenogen are advised to read the registration statement on Form S-4 and the joint proxy statement regarding the proposed merger when they become available, because they will contain important information. Caliper and Xenogen expect to mail the joint proxy statement about the proposed merger to their respective stockholders. Investors and security holders may obtain a free copy of the proxy statement and any other documents filed by Caliper and Xenogen at the Securities and Exchange Commission’s website at http://www.sec.gov or directly from Caliper and Xenogen.
Caliper and its officers and directors may be deemed to be participants in the solicitation of proxies from stockholders of Caliper with respect to the proposed merger with Xenogen. Information regarding such officers and directors is included in Caliper’s annual report on Form 10-K for the fiscal year ended December 31, 2005 and in its amendment to such Form 10-K on Form 10-K/A filed with the SEC. These documents are available free of charge at the Securities and Exchange Commission’s website at http://www.sec.gov or directly from Caliper.
On June 14, 2006, Mr. Thomas Higgins, the Executive Vice President and Chief Financial Officer of Caliper Life Sciences, Inc. (“Caliper”) and Dr. Mark Roskey, Vice President, Reagents and Applied Biology of Caliper made a presentation at the Pacific Growth Equities’ Life Sciences Growth Conference 2006. During this presentation, Mr. Higgins and Dr. Roskey discussed various business issues relating to Caliper, including Caliper’s proposed business combination with Xenogen Corporation. The following is that portion of the presentation made by Mr. Higgins and Dr. Roskey relating to Caliper’s proposed business combination with Xenogen Corporation. A copy of the full presentation is available on Caliper's website at www.caliperLS.com.

Transforming Life Sciences through Innovative Technologies Tom Higgins Executive Vice President & CFO Mark Roskey, Ph.D. Vice President, Reagents and Applied Biology June 14, 2006 |

Forward-Looking Statements This presentation includes forward-looking statements, including financial projections. Reference to what we expect, believe, intend to do, plan, hope, estimate or other statements referring to future events or results are intended to identify those as forward-looking statements. Forward-looking statements are inherently subject to risks and uncertainties. Further information on risks faced by Caliper are included under the caption Factors Affecting Operating Results in Caliper's annual report on Form 10-K, filed with the Securities and Exchange Commission on March 14, 2006, and in our other SEC reports. These SEC filings are available on the website maintained by the Securities and Exchange Commission at www.SEC.gov. Caliper does not undertake any obligation to update forward-looking or other statements in this presentation to reflect any change in Caliper's expectations with regards to such statements or any change in events, conditions or circumstances on which any such statements are based. |

Important Notice In addition to the foregoing, please note that Caliper has filed a draft Registration Statement on Form S-4 in order to register the shares of its common stock and warrants to be issued to the former stockholders of Xenogen Corporation in the proposed merger between Caliper and Xenogen, and Caliper and Xenogen will be filing a joint proxy statement with the Securities and Exchange Commission. Investors and security holders of Caliper and Xenogen are advised to read the draft Registration Statement on Form S-4 and the joint proxy statement regarding the proposed merger when it becomes available because they contain important information. Caliper and Xenogen expect to mail the joint proxy statement about the proposed merger to their respective stockholders. Investors and security holders may obtain a free copy of the proxy statement and any other documents filed by Caliper and Xenogen at the Securities and Exchange Commission's web site at http://www.sec.gov or directly from Caliper and Xenogen. Caliper and its officers and directors may be deemed to be participants in the solicitation of proxies from stockholders of Caliper with respect to the proposed merger with Xenogen. Information regarding such officers and directors is included in Caliper's Annual Report on Form 10-K for the fiscal year ended December 31, 2005, filed with the Securities and Exchange Commission. This document is available free of charge at the Securities and Exchange Commission's web site at http://www.sec.gov and directly from Caliper. |

Rapid Evolution of our Business and Strategy Transformative Event – Xenogen merger Molecular imaging Bridging in vitro/in vivo experimentation Revenue, cost synergies 2006 Forward Accelerating revenue growth Critical mass Drive to profitability Transformative Event – Zymark merger New management team Commercial orientation Established distribution channels 2003/2004 “Old” Caliper Development stage - R&D focus, $25M rev, customer concentration ’02 OPEX - $61M, 235% of rev Operating loss of $47M in 2003 Negative $40M enterprise value Positioning NovaScreen acquisition for in vitro services “Caliper Driven” collaborations with Agilent, Affymetrix and Bio-Rad. New diagnostic collaborations 2005 Financial Improvement Completed 2.5 years restructuring $87M Revenue, 9% growth OPEX $49M, 57% of revenue Op. loss $15M, Q4 cash flow positive |
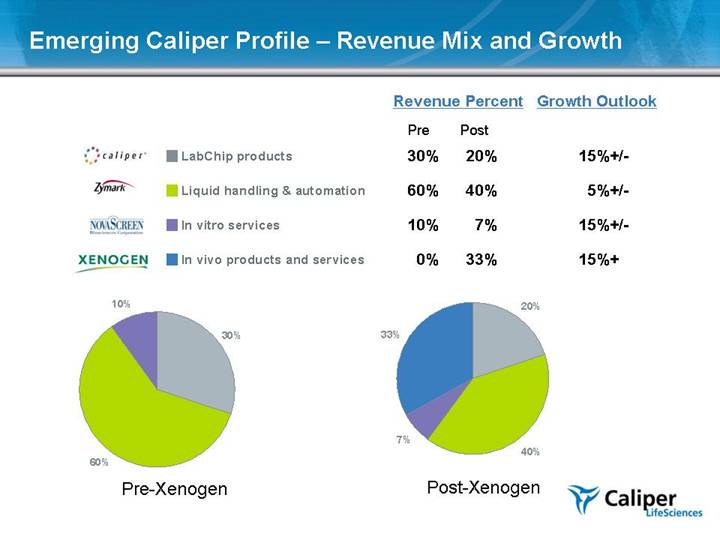
Emerging Caliper Profile – Revenue Mix and Growth Revenue Percent Growth Outlook Pre Post 30% 20% 15%+/- 60% 40% 5%+/- 10% 7% 15%+/- 0% 33% 15%+ Pre-Xenogen Post-Xenogen LabChip products Liquid handling & automation In vitro services In vivo products and services 30% 60% 10% 20% 40% 7% 33% |

Markets Drug Discovery and Development $2.0B market, growing 5-8%; approx 6%+ share w/ Xenogen 2005 revenue approx. $80M; expect 2006 growth above market rate Molecular Diagnostics $1.5B market, growing 20%; new entrant 2005 license & contract revenue approx. $7M; expect product revenue in 2007 Profile of the New Company (cont’d) Customers Revenue Distribution by Customer Segment 0 20 40 60 80 100 120 Caliper Xenogen % of Revenue (approx.) Gov't/Academia Commercial Revenue Distribution by Geography 0 20 40 60 80 100 120 Caliper Xenogen % of Revenue (approx.) International Domestic |

One Reason for the Productivity Gap is “Over-reliance” on Early Stage In Vitro Testing Current industry trends which may help close the gap: Improved in vitro data quality Better correlation between in vitro and in vivo results, esp. clinical Identification of relevant biomarkers, and use of imaging technologies, to accelerate endpoints New, validated in vivo models Biochemical Inhibition Cellular Functional Curing Mice Affecting Primary Cells Curing People |
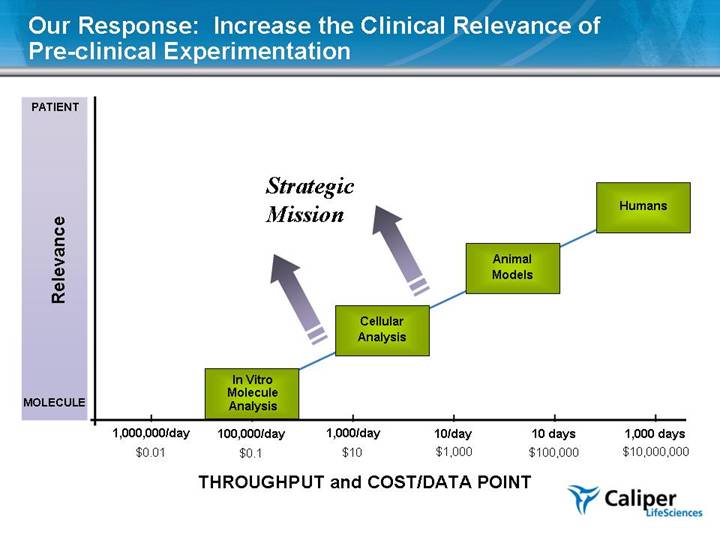
In Vitro Molecule Analysis Cellular Analysis Humans Animal Models 1,000 days 10 days 10/day 1,000,000/day THROUGHPUT and COST/DATA POINT Relevance 1,000/day $10,000,000 $100,000 $1,000 $10 $0.01 PATIENT MOLECULE Our Response: Increase the Clinical Relevance of Pre-clinical Experimentation 100,000/day $0.1 Strategic Mission |
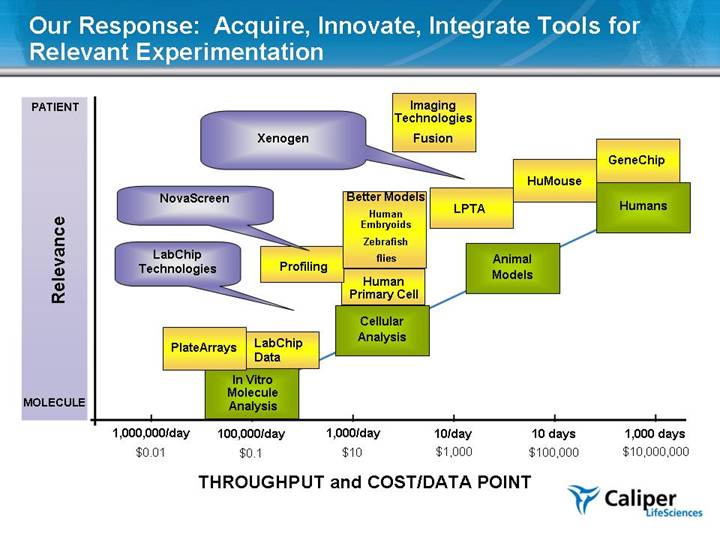
In Vitro Molecule Analysis Cellular Analysis Humans Animal Models 1,000 days 10 days 10/day 1,000,000/day THROUGHPUT and COST/DATA POINT Relevance 1,000/day $10,000,000 $100,000 $1,000 $10 $0.01 PATIENT MOLECULE Our Response: Acquire, Innovate, Integrate Tools for Relevant Experimentation 100,000/day $0.1 GeneChip Human Primary Cell Profiling LPTA HuMouse PlateArrays Better Models Human Embryoids Zebrafish flies Xenogen NovaScreen LabChip Data LabChip Technologies Imaging Technologies Fusion |
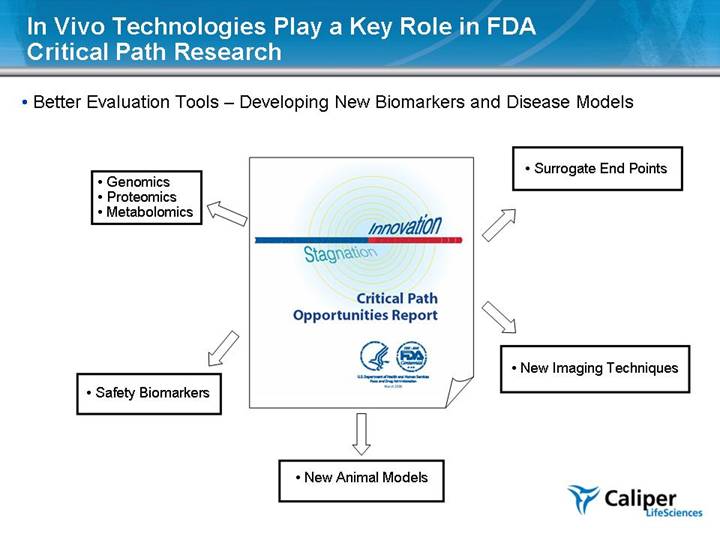
In Vivo Technologies Play a Key Role in FDA Critical Path Research Better Evaluation Tools – Developing New Biomarkers and Disease Models Genomics Proteomics Metabolomics Safety Biomarkers Surrogate End Points New Imaging Techniques New Animal Models |
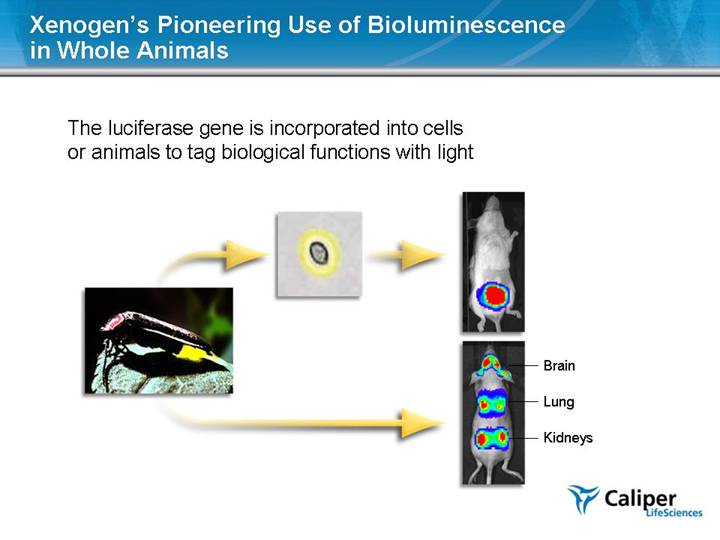
Xenogen’s Pioneering Use of Bioluminescence in Whole Animals The luciferase gene is incorporated into cells or animals to tag biological functions with light Brain Lung Kidneys |

Integrated Solution for Biophotonic Imaging Proprietary System Biological Solutions Imaging Hardware Imaging Software Tag Cell Tag Gene |
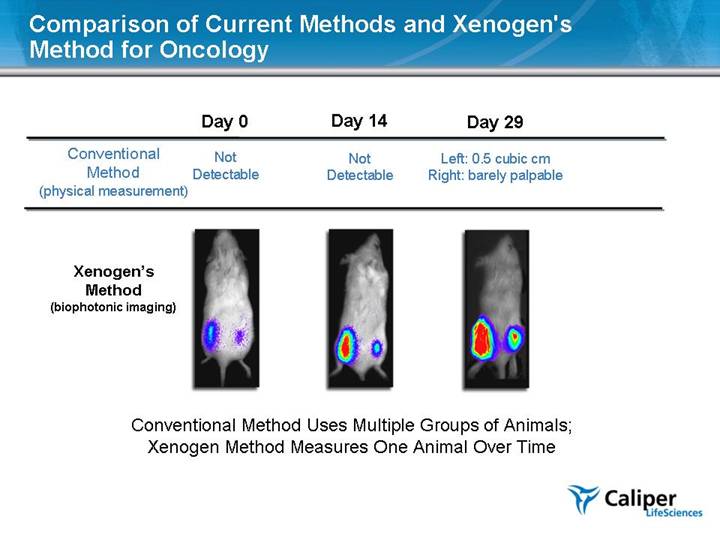
Comparison of Current Methods and Xenogen's Method for Oncology Conventional Method (physical measurement) Xenogen’s Method (biophotonic imaging) Day 0 Not Detectable Day 14 Not Detectable Day 29 Left: 0.5 cubic cm Right: barely palpable Conventional Method Uses Multiple Groups of Animals; Xenogen Method Measures One Animal Over Time |

TFF1/pS2 (trefoil peptide-1) Mup1 (major urinary protein-1, or alpha 2u globulin) ER1 (estrogen receptor-alpha) KAP (kidney androgen regulated protein) VEGF (vascular endothelial growth factor) KDR or VEGFR2 in NUDE (Nu/Nu) background KDR or VEGFR2 (vascular endothelial growth factor receptor-2) Angiogenesis/Oncology CRP (C-reactive protein, human promoter) VCAM1 (vascular cell adhesion molecule-1) MMP1 (matrix metalloproteinase-1, human promoter) RETN (resistin) SCYAA2 or MCP1 (small inducible cytokine A2 or monocyte colonizing protein-1) INS2 (rat insulin gene promoter) Metabolic Disease GAL (galanin) VGCC (alpha 2 delta 1Ca++ channel subunit) PSA (prostate specific antigen driving luc and T antigen) PAI1 (plasminogen activator inhibitor type 1) eNOS (endothelial nitroc oxide synthase) GFAP (glial fibrillary acidic protein) BMP4 (bone morphogenetic protein-4) CMV (cytomeglovirus promoter) CadioVascular, CNS, IMID LPTA® Animal Models IL6 (interleukin-6) TNFa (tumor necrosis factor-alpha) IB (inhibitor protein for NF-B) NF-kB-RE (Nf-kappa B response element construct) Cox2 or Ptgs2 (cyclooxygenase-2) IL2 (interleukin-2) SAA1 (serum amyloid A-1) Epx (eosinophil peroxidase) iNOS or NOS2 (macrophage nitric oxide synthase) Inflammation |

CP2C (Cytochrome p450, isoform 2C, human promoter) GADD153 (growth arrest and DNA damage inducible 153) GADD45B (growth arrest and DNA damage inducible 45-beta) GADD45a (growth arrest and DNA damage inducible 45-alpha) MDR1 (multiple drug resistance-1, human promoter) SOD1 (superoxide dismutase) lGCS (gamma-glutamylcysteine synthetase) CYP19 or Aro (Aromatase) CYP1A2 (Cytochrome p450, isoform 1A2-mouse promoter CYP1A2 (Cytochrome p450, isoform 1A2-human promoter CYP3A4 RAT (Cytochrome p450, isoform 3A-human promoter) CYP3A11 (Cytochrome p450 isoform 3A-mouse promoter) CYP3A4 (cytochrome p450 isoform 3A-human promoter) Ho1 or Hmox1 (heme oxygenase-1) Animal Model Drug Metabolism/Toxicology LPTA Animal Models (cont’d) |
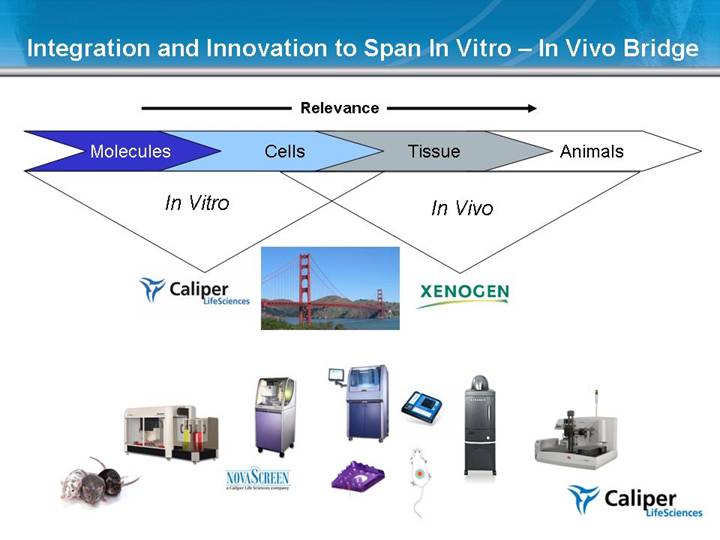
Integration and Innovation to Span In Vitro – In Vivo Bridge In Vitro In Vivo Cells Molecules Animals Tissue Relevance |

The Xenogen Merger - Strategic and Financial Benefits Revenue Synergies Customer synergies: academic / commercial and geographic Post-merger growth rate for combined companies expected at 10% - 15% Critical Mass Expands revenue base over 40% (2005 pro forma) Leverages manufacturing, commercial, G&A and aftermarket infrastructure Expect 200 basis point improvement in OPEX and 100 b.p. in gross margin Profitability Identified “high probability” cost synergies = $10 million. Transaction should be accretive in 2008 Key Parameters All equity deal: 13.2M shares, 5.1M warrants (5yr, $6.79 strike) - $80M at announcement Post closing, XGEN shareholders own 26% of Caliper, 32% fully diluted Cash resources expected adequate for integration and operations |

An Ambitious 2006 2006 Goals Complete Xenogen diligence Evaluate Q1 results and revenue outlook Update full year guidance Q3 target closing Achieve 50% growth in LabChip 3000 / LabChip 90 placements Control OPEX spending and improve gross margins Achieve Q2 revenue guidance of $22 – 24M (8 - 18% growth) Implement Xenogen integration plan to position Caliper for 2007 |
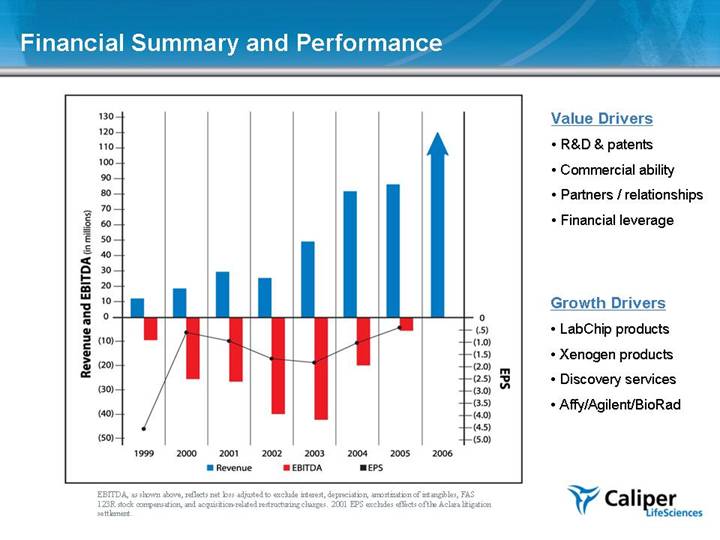
Financial Summary and Performance EBITDA, as shown above, reflects net loss adjusted to exclude interest, depreciation, amortization of intangibles, FAS 123R stock compensation, and acquisition-related restructuring charges. 2001 EPS excludes effects of the Aclara litigation settlement. Value Drivers R&D & patents Commercial ability Partners / relationships Financial leverage Growth Drivers LabChip products Xenogen products Discovery services Affy/Agilent/BioRad |