 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Forward Looking Statements Any statements in this presentation about future expectations, plans and prospects for the Company, including statements containing the words "believes," "anticipates," "plans," "expects," and similar expressions, constitute forward looking statements, which are subject to the safe harbor for such statements in the Private Securities Litigation Reform Act of 1995. Future events could cause actual results to differ materially from those indicated by such statements. Reference is made to the factors discussed in the “Management Discussion and Analysis" and "Risk Factors" sections of the Company's most recent quarterly or annual report filed with the Securities and Exchange Commission. The forward-looking statements herein represent the Company's views as of the date of this presentation and should not be relied upon to represent the Company's views as of a subsequent date. While the Company anticipates that subsequent events may cause the Company's views to change, the Company disclaims any obligation to update such forward-looking statements. 2 * * |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Mission Advancing Drugs Through Glycoscience ® 3 * * |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Key Investment Points • Clinical stage pharmaceutical company – Completed Phase I/II cancer trials w/ DAVANAT ® – Stabilized 35% of end stage patients: 2-13 months – Two ongoing Phase II front line trials • DAVANAT ® increased the half life of 5-FU 8 fold with no increase in toxicity • DAVANAT ® /5-FU model can be applied to other chemotherapy agents 4 |
 Clinical Trial Program – Completed Phase I/II cancer trials w/ DAVANAT ® – Stabilized 35% of end stage patients: 2-13 months – Two ongoing Phase II front line trials * * 5 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Phase I/II End Stage Cancer Trials Summary • 35% stabilized at the highest dose level • Maximum Tolerated Dose & Dose Limiting Toxicity not reached • DAVANAT ® significantly increased half life of 5-FU with no increase in toxicity 6 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Phase I Clinical Trial Summary • DAVANAT ® was well tolerated • Maximum Tolerated Dose and Dose Limiting Toxicity of DAVANAT ® not reached – DAVANAT 280 mg/m² recommended Phase II dose • Pharmacokinetics – Half life of 5-FU alone is 6-22 minutes – Half life of 5-FU with DAVANAT ® is 28-137 minutes – No increase in 5-FU toxicity w/ increased exposure • Stable disease in 14 of 26 efficacy evaluable patients – 7/10 patients stabilized at the highest DAVANAT ® dose level 7 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W SD Stable Disease PD Progressive Disease NM Non-Measurable Disease Cycles Completed Dose, cycle 2 /m2 1+2 3 4 5 6 7 1001 PD 1002 SD 2001 Hepatocellular PD 2002 Hepatocellular SD 3001 PD 3002 SD 3004 PD 3003 PD 5001 SD 2004 SD 3005 PD 4001 PD 4002 Colorectal PD 3006 Prostate NM 2005 Colorectal SD 5003 Colorectal (appendix) NM 5004 SD 4003 PD 2007 Spindle Cell PD 5005 Pancreatic SD 2008 Colorectal SD 2009 Colorectal SD 5006 Billiary SD 2010 Colorectal (cecal) SD 2014 Breast SD 2016 Hepatic PD 2018 Cholangiocarcinoma SD 5008 Pancreatic PD 280 mg 150 mg Patient Number Tumor Type Outcome, end of cycle 2 (RECIST) C Y C L E S Colorectal Colorectal 210 mg 30 mg 60 mg Colorectal 100 mg Colorectal Phase l Patient Summary: Stabilized 70% at Highest Dose Level 8 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Phase II Colorectal Cancer Trial Summary • Trial conducted with end-stage patients • Anti-tumor activity was seen with DAVANAT ® /5-FU – 1 patient experienced Partial Response – 6 patients stabilized • No increase in 5-FU toxicity w/ increased exposure 9 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W DAVANAT ® /5-FU Enrolling Phase II Colorectal Cancer Trial • Indication: First-line treatment of patients who are unable to tolerate irinotecan or oxaliplatin • Regimen: DAVANAT ® /5-FU, Leucovorin, AVASTIN ® – Repeat cycles every 2 weeks to disease progression or toxicity • Objectives: Complete/Partial Response – Stable Disease; Progression Free Survival; Safety; Quality of Life • Design: Multi-center, open label study – Simon Optimal 2-stage design • Patients: Up to 50 patients – Begin enrolling/ dosing patients in Q4 2006 10 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W DAVANAT ® /5-FU Enrolling Phase II Biliary Cancer Trial • Indication: First line treatment of patients with biliary tract cancer • Regimen: DAVANAT ® (280 mg/m²) + 5-FU (600 mg/m²) IV daily x 4 days – Repeat cycles every 28 days to disease progression or toxicity • Objectives: Complete/Partial Response – Stable Disease; Progression Free Survival; Safety; Quality of Life • Design: Multi-center, open label study – Simon Optimal 2-stage design • Patients: Up to 35 – Begin enrolling/dosing patients in Q4 2006 11 |
 DAVANAT ® Increases the Half Life of 5-FU, 8 Fold with No Increase in Toxicity * * 12 |
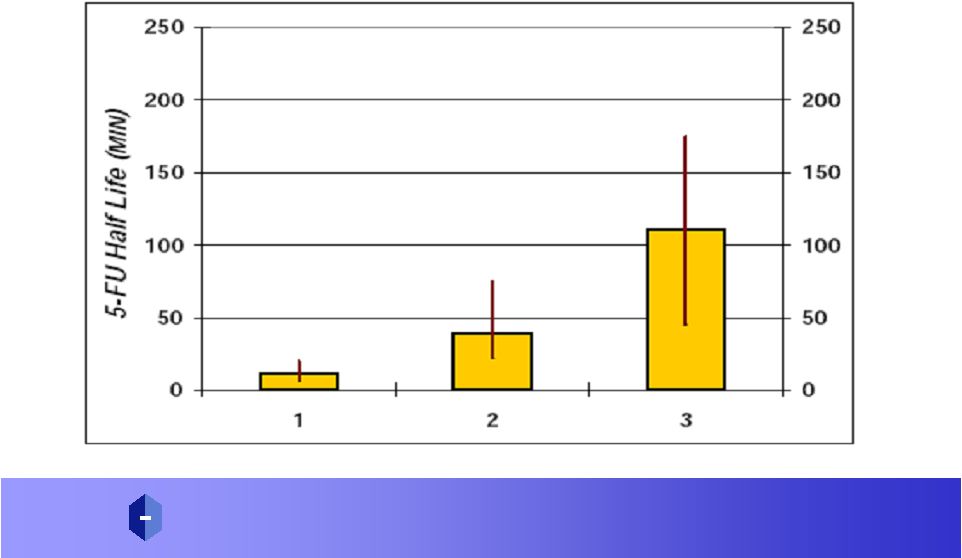 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W DAVANAT ® Increases Half Life of 5-FU in Patients, 8 times 5-FU / DAVANAT – Day 4 5-FU / DAVANAT – Day 1 5-FU – Historical |
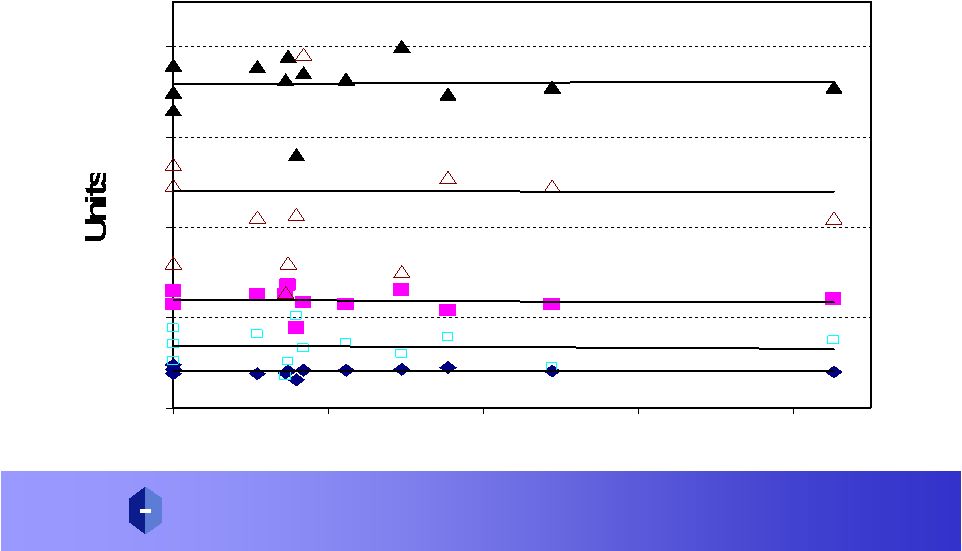 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W 5-FU in the presence of DAVANAT ® Does Not Change Key Toxicity Markers 0 10 20 30 40 0 10 20 30 40 AUC Hematocrit Hemoglobin Red blood cells White blood cells Platelets |
 The DAVANAT ® /5-FU Model Can Be Applied To Increasing Efficacy/ Decreasing Toxicity of Other Chemotherapy Agents 15 |
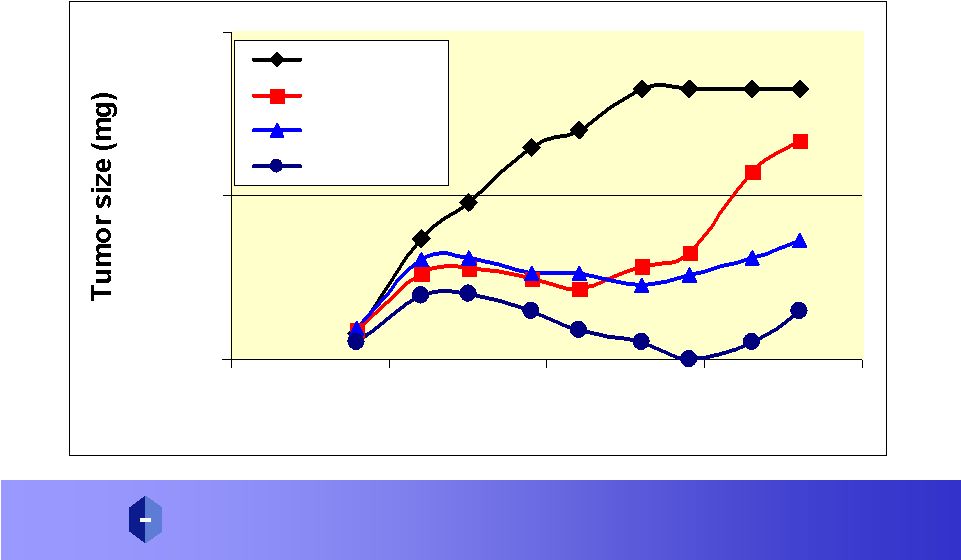 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W DAVANAT ® with another Chemotherapy Agent 100 1000 10000 0 10 20 30 40 Time post implanatation (Days) Control IR40/DAV6 IR40/DAV30 IR40/DAV120 16 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Mechanism of Action: Targeting Lectins on Cancer Cells • DAVANAT ® binds to lectins • Galectins are a type of lectin that are over- expressed on cancer cells • Galectins affect cell development, differentiation, apoptosis and tumor metastasis 17 |
 P R O P H A R M A C E U T I C A L S , I N C. A D V A N C I N G D R U G S T H R O U G H G L Y C O S C I E N C E ® Amex: P R W Key Investment Points • Clinical stage pharmaceutical company – Completed Phase I/II cancer trials w/ DAVANAT ® – Stabilized 35% of end stage patients: 2-13 months – Two ongoing Phase II front line trials • DAVANAT ® increased the half life of 5-FU 8 fold with no increase in toxicity • DAVANAT ® /5-FU model can be applied to other chemotherapy agents 18 |