 Company Update September 27, 2011 OTC: GALT Exhibit 99.1 |
 Forward Looking Statements This presentation contains, in addition to historical information, forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements relate to future events or future financial performance, and use words such as “may,” “estimate,” “could,” “expect” and others. They are based on our current expectations and are subject to factors and uncertainties which could cause actual results to differ materially from those described in the statements. Factors that could cause our actual performance to differ materially from those discussed in the forward-looking statements include, among others: incurrence of operating losses since our inception, uncertainty as to adequate financing of our operations, extensive and costly regulatory oversight that could restrict or prevent product commercialization, inability to achieve commercial product acceptance, inability to protect our intellectual property, dependence on strategic partnerships, product competition, and others stated in risk factors contained in our SEC filings. We cannot assure that we have identified all risks or that others may emerge which we do not anticipate. You should not place undue reliance on forward-looking statements. Although subsequent events may cause our views to change, we disclaim any obligation to update forward-looking statements. © 2011 Galectin Therapeutics OTC:GALT 2 |
 Recent Highlights • March 2011: New CEO • May 2011: Announced new strategic priorities, new web site • May 2011: Name changed to Galectin Therapeutics (Formally Pro-Pharmaceuticals) • July 2011: Analyst coverage initiated by Shiv Kapoor of Morgan Joseph TriArtisan • August 2011: Analyst coverage initiated by Vernon Bernardino of Dawson James Securities • September 2011: Presentation at Rodman & Renshaw Conference, New York, New York © 2011 Galectin Therapeutics OTC:GALT 3 |
 Galectin Therapeutics Highlights • Leader in galectin science • Pipeline of carbohydrate-based drug compounds that inhibit galectins • Liver fibrosis program goal to be first therapy for this indication • Target validated in convincing pre-clinical data • Clinical trials expected to begin in 2012 • Cancer Therapy • Galectin inhibitor added to chemotherapy • Cancer immunotherapy program activates patient’s own immune system to kill tumor cells 4 © 2011 Galectin Therapeutics OTC:GALT |
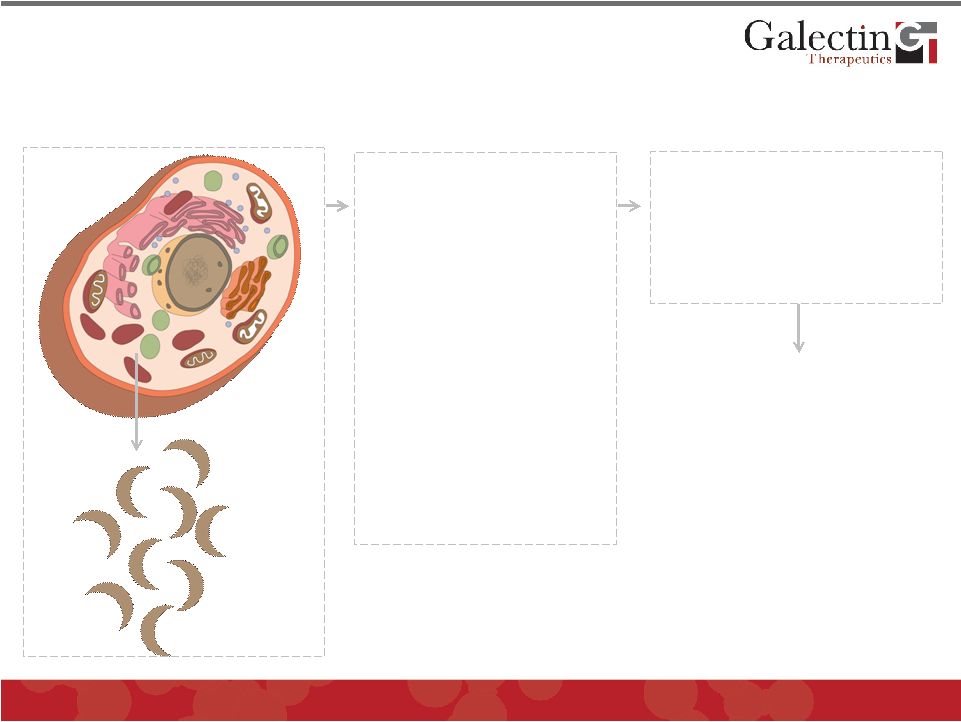 Galectin Proteins Are Important In Disease Pathogenesis © 2011 Galectin Therapeutics OTC:GALT 5 Secreted Galectin Proteins 1. Bind to cell surface and matrix glycoproteins (galactose residues) 2. Modulate cell signaling 3. Promote cell-cell interactions 4. Promote cell- matrix interactions PROMOTE PATHOLOGY Markedly Increased in: 1. Fibrosis 2. Cancer 3. Inflammation |
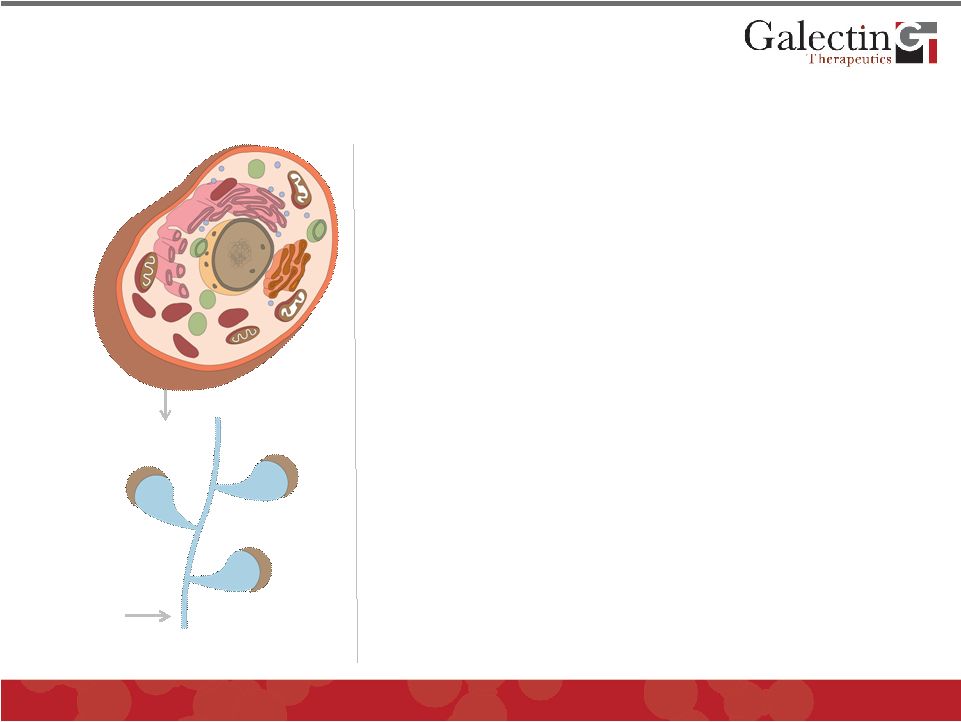 Galectin Proteins Galectin Inhibitor Our Galectin Inhibitors Are Novel Carbohydrate-Based Drug Compounds © 2011 Galectin Therapeutics OTC:GALT 6 • Target secreted galectins and those associated with cell membrane • Strong binding to multiple galectin proteins and multiple galectins per drug molecule • High molecular weight allows long exposure to galectin containing compartment • Low toxicity potential as a carbohydrate with no toxic metabolites • Low manufacturing costs • Strong patent protection with no licensing encumbrance • Two major classes of compounds under development: GM-CT and GR-MD |
 We Are The Leaders In Galectin Inhibitor Drug Development • Only company with galectin inhibitors in clinical development • Published authoritative books in the field 7 © 2011 Galectin Therapeutics OTC:GALT |
 Galectins Are Involved In The Pathogenesis Of Many Diseases • Fibrosis of organs • Nearly all cancers • Heart failure • Ischemic cardiovascular and cerebrovascular disease • Arthritis • Allergic disease • Eczema and skin inflammation • Inflammatory bowel disease • Eye inflammation • Inflammatory and autoimmune disorders • Response to infections • Kidney disease 8 Galectins implicated in: © 2011 Galectin Therapeutics OTC:GALT |
 So, How Do We Choose Diseases For Drug Development? • Galectins are proven important in the mechanism of disease • There are serious, life threatening consequences to patients • There are no, few, or ineffective therapies • Rapid development pathways are possible 9 Treat important diseases where: © 2011 Galectin Therapeutics OTC:GALT |
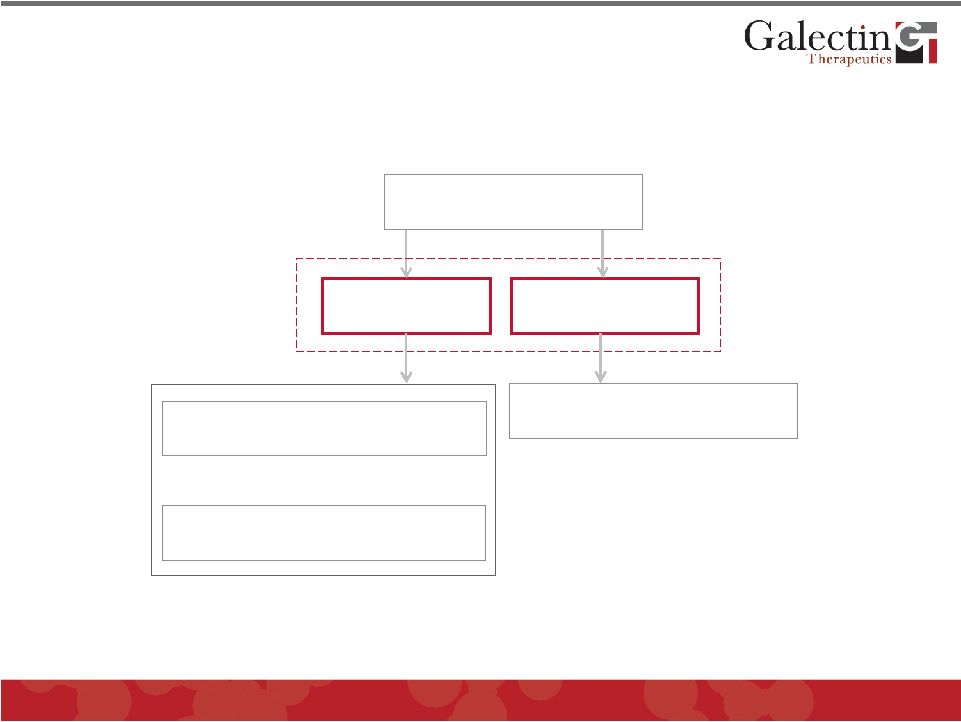 Disease Area Development Programs Cancer Fibrosis Liver Fibrosis Chemotherapy Immunotherapy GALECTINS 10 © 2011 Galectin Therapeutics OTC:GALT |
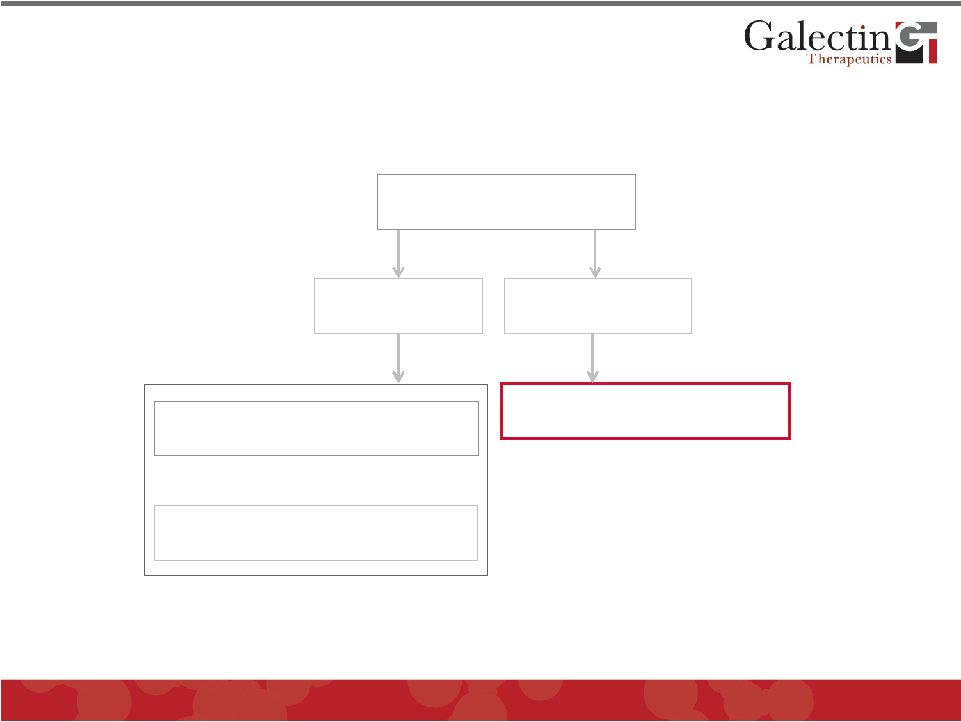 Disease Area Development Programs Cancer Fibrosis Liver Fibrosis Chemotherapy Immunotherapy GALECTINS 11 © 2011 Galectin Therapeutics OTC:GALT |
   Many Diseases Lead To Liver Fibrosis And Cirrhosis With Serious Medical Consequences Healthy Cirrhosis 12 Hepatitis C (57%) Alcoholic liver disease (24%) Non-alcoholic fatty liver (9.1%) Hepatitis B (4.4%) Miscellaneous (5.5%) Source: Burden of liver disease in the United States: Summary of a workshop. American Association for the Study of Liver Disease, May 2001 © 2011 Galectin Therapeutics OTC:GALT |
 Liver Cirrhosis Is A Major Problem In The United States Transplants (6,291*) Wait List (17,000**) Death (44,677 # ) Cirrhosis (400,000 ## ) Millions of people with liver disease that may progress to cirrhosis * Performed in US in 2010 (UNOS) * * Prevalence in US 2010 (UNOS) The ONLY current therapy is liver transplantation 13 # Deaths in 1998 (AASLD Workshop, 2001) ## Prevalence in US 1976-1980 (NIDDK) © 2011 Galectin Therapeutics OTC:GALT |
 Galectin-3 Is A Critical Target For Therapy of Liver Fibrosis 1. Galectin-3 is produced in large amounts by human fibrotic liver 2. Galectin-3 is essential in mice for the development of liver fibrosis 3. Galectin inhibitors block production of fibrogenic markers in the key human cell responsible for liver fibrosis 4. Galectin inhibitors reverse experimental fibrosis in rats © 2011 Galectin Therapeutics OTC:GALT 14 Key Evidence: Key Evidence: |
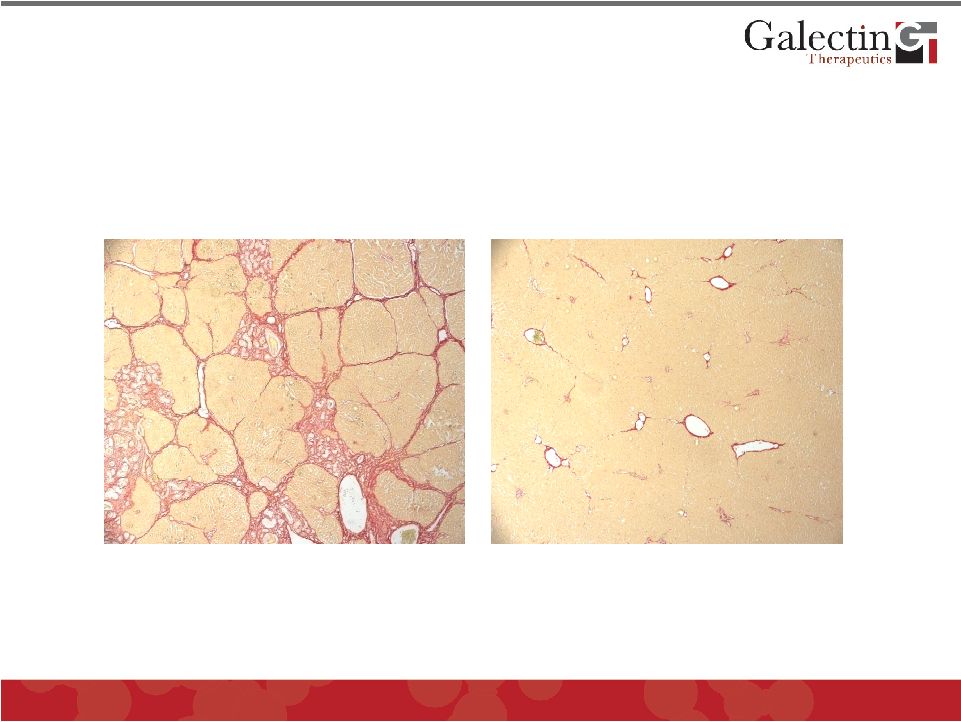 Galectin Inhibitors Effectively Treat Liver Fibrosis in Rats Liver Fibrosis, induced by injection of chemical toxin for 8 weeks Regression of Fibrosis after 4 weeks of treatment with GR-MD-01 15 © Galectin Therapeutics OTC : GALT |
 Summary Of Development Program Rationale In Liver Fibrosis • Liver fibrosis represents a very large unmet medical need • Galectin-3 protein is proven target • Drugs reverse liver fibrosis in animals and show efficacy in human cell culture models of fibrosis • Non toxic drugs with little likelihood of drug interactions • Rapid clinical development pathways • Initial indication provides opportunity for orphan disease status, fast track, priority review, and potentially accelerated approval © Galectin Therapeutics OTC : GALT 16 |
 Development Program & Markets • Initial Indication: Post transplant recurrent Hepatitis C with fibrosis • Focus of first phase II clinical trial • Orphan disease designation possible • Additional peri-transplant indications: • Established cirrhosis of various etiologies but not eligible for transplantation • Established cirrhosis of various etiologies, on transplant list • Expansion of indications • Non-alcoholic fatty liver disease (NASH) • Viral hepatitis C and B with fibrosis • Alcoholic fibrosis © 2011 Galectin Therapeutics OTC:GALT 17 |
 Fibrosis Pipeline: Drug Advancement December 2011 Pre-Clinical Phase 1 Phase Phase Registration Submitted Liver Fibrosis GM-CT-01 GM-CT-02 GR-MD-01 GR-MD-02 © 2011 Galectin Therapeutics OTC:GALT 18 More advanced than typical pre-clinical development program: Efficacy shown in human cells as well as animals Proven safety in animals and humans (GM-CT-01) Low potential for toxicity for all compounds Low potential for drug interactions 2 3 |
 Fibrosis Clinical Program (First Indication Clinical Trials) © 2011 Galectin Therapeutics OTC:GALT 19 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 2011 2012 2013 Pre-IND Meeting Initiate P-II Initiate P-I Submit IND GM-CT-01 Others |
 Disease Area Development Programs Cancer Fibrosis Liver Fibrosis Immunotherapy Chemotherapy GALECTINS © 2011 Galectin Therapeutics OTC:GALT 20 |
 Roles Of Secreted Galectins In Cancer The vast majority of cancers secrete large amounts of galectins • Galectins impact cancer growth at many points • Interfering with galectin function has been shown to have beneficial effects • Galectin Therapeutics has proprietary compounds that leverage this benefit 21 © 2011 Galectin Therapeutics OTC:GALT |
 Phase 2 Clinical Trial Performed in Metastatic Colorectal Cancer (DAVFU-003) Cancer Trial (DAVFU-003) Day 0 Day 28 GM-CT-01 Chemo (5-FU) Phase 2 trial of 5-FU plus GM-CT-01 in line 3/4 therapy of metastatic colorectal cancer Twenty (20) patients enrolled who all had 3 to 4 previous courses of chemotherapy, including 5-FU and biologicals Overall median survival was 6.7 months. In similar patients, Erbitux ® had a 6.1 month survival compared to 4.6 months with no therapy Suggests efficacy of regimen |
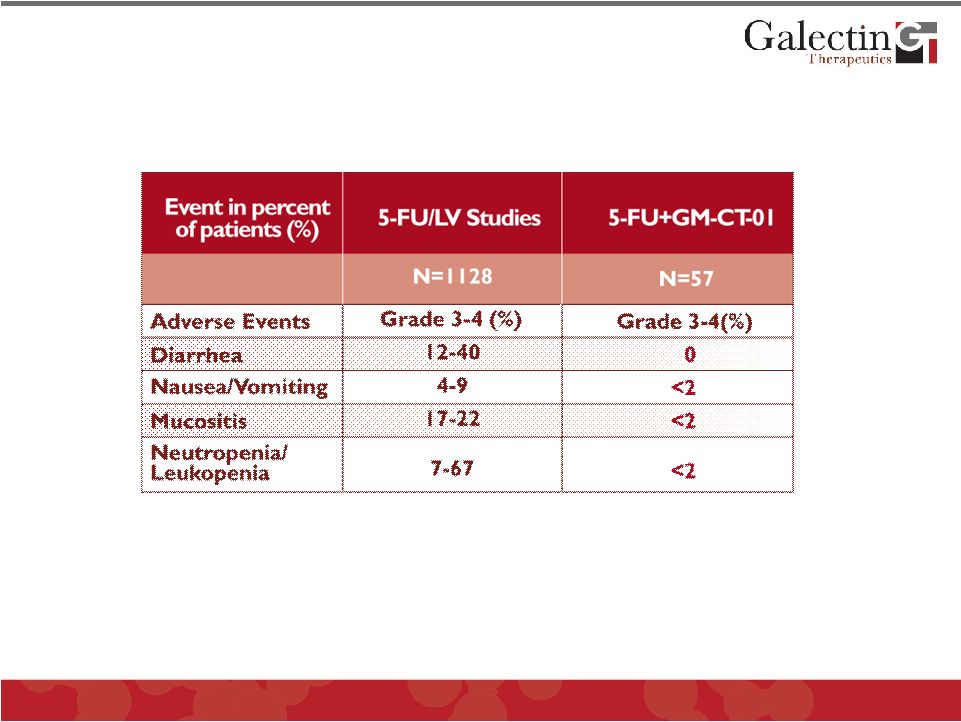 GM-CT-01 Reduces 5-FU Chemotherapy Related Side Effects Simultaneous improved efficacy with reduction in side effects of standard chemotherapy would be desirable in cancer therapeutics Data on 5-FU+GM-CT-01 compiled from patients receiving full dose therapy in studies DAVFU-001, 003, 006, and 007 © 2011 Galectin Therapeutics OTC:GALT 23 |
 Development Approach In Colorectal Cancer • Studies demonstrate potential utility of galectin inhibitors in combination with chemotherapy in cancer • FDA has confirmed that preclinical and clinical data are adequate to proceed with large clinical trials • Our colorectal cancer program remains active, but we are deferring new clinical trials pending data from the tumor immunology clinical trial that may improve the design of future studies • More rapid international registration is an approach that may provide revenue to support development programs and gain additional clinical experience with GM-CT-01 © 2011 Galectin Therapeutics OTC:GALT 24 |
 Registration And Marketing GM-CT-01 In Colombia And Latin America • The government of Colombia, and oncology key opinion leaders in that country, expressed an interest in making GM-CT-01 available for use in Colombia for patients with metastatic colorectal cancer • Equally interested in the increased tumor efficacy and reduction in 5- FU related side effects • Our partner Pro-Caps has submitted a marketing application to INVIMA (FDA equivalent) and has indicated our clinical data should be sufficient for approval • With approval, Pro-Caps expects sales to begin in 2012 • Upon success in Colombia, we have the opportunity to seek approval in other Latin American countries (reciprocity with 12 other countries) © 2011 Galectin Therapeutics OTC:GALT 25 |
 Disease Area Development Programs Cancer Fibrosis Liver Fibrosis Immunotherapy GALECTINS Chemotherapy © 2011 Galectin Therapeutics OTC:GALT 26 |
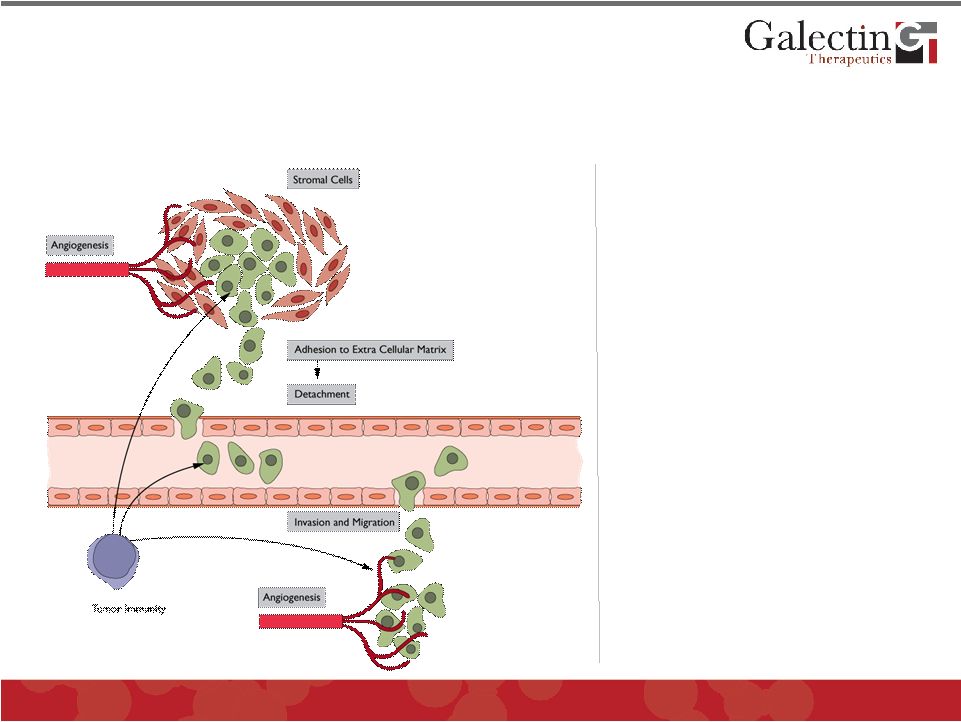
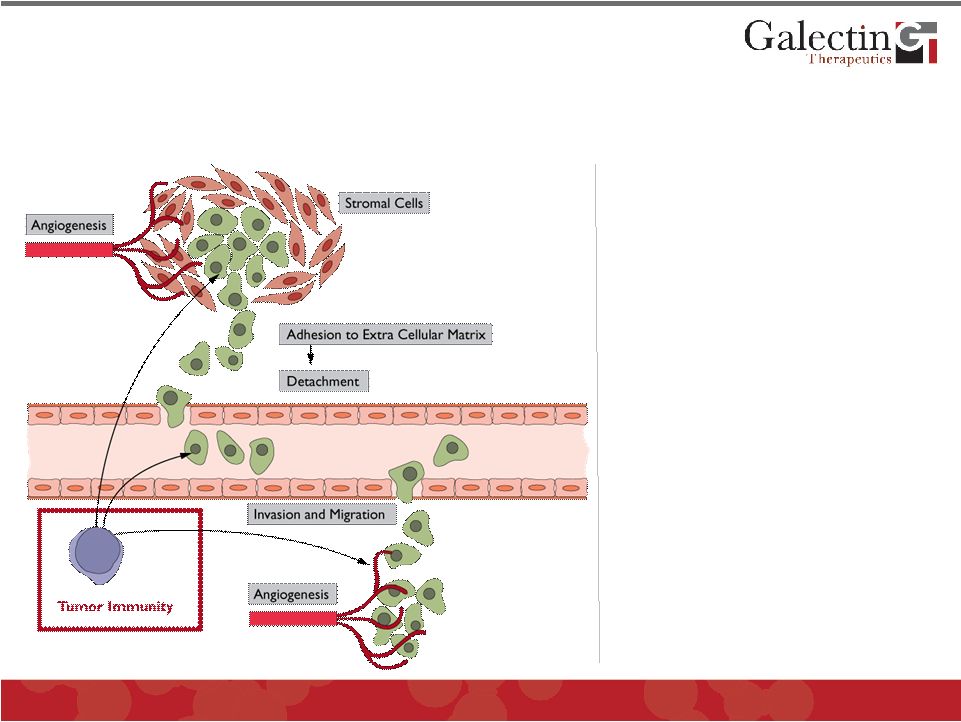 Enhancing Anti-Tumor Immunity Is A Promising Effect Of Blocking Galectins © 2011 Galectin Therapeutics OTC:GALT 27 • Tumor cell invasion: extracellular matrix adhesion & detachment • Stromal cell function • Metastasis: cell invasion and migration • Angiogenesis • Tumor immunity |
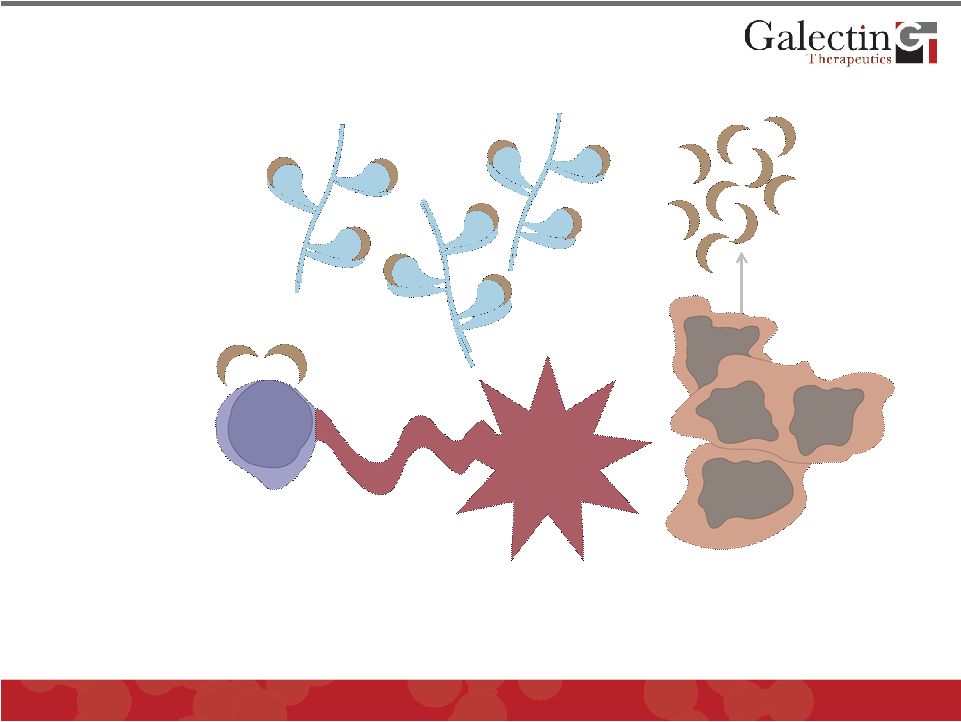 Tumor-Specific Cytotoxic T-Cell Lymphocytes Blocking Galectins Enhances Tumor Killing By Immune System Studies done in collaboration with Ludwig Institute, Brussels, Belgium © 2011 Galectin Therapeutics OTC:GALT 28 Tumor Cells Cytotoxicity |
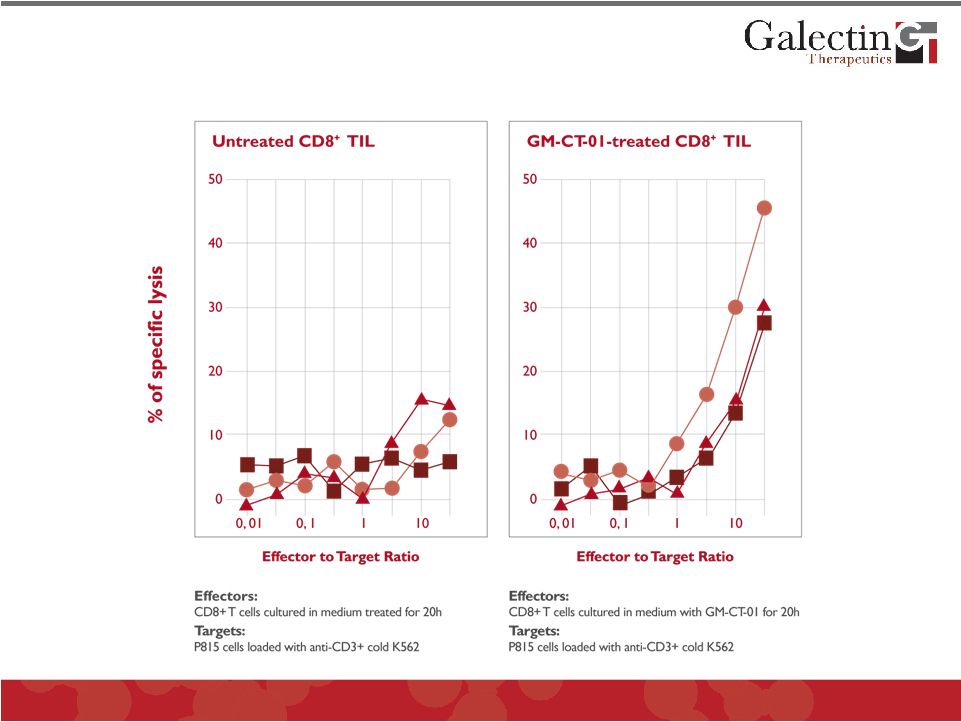 GM-CT-01 Restores Ability of Immune Cells to Kill Tumor Cells © 2011 Galectin Therapeutics OTC:GALT 29 |
 GM-CT-01 In Tumor Immunotherapy • A Phase 1/2 study is scheduled to begin in Q4 2011 • IMPD (Investigational Medicinal Product Dossier) was submitted to the EMA (European Medicines Agency) on September 20, 2011 • Patients with advanced metastatic melanoma • Treatment Regimen: • Tumor-specific peptide vaccination (previously tested) • GM-CT-01 administered between peptide vaccinations • Primary endpoint: Partial or complete response • Historical controls who received same peptide vaccine • Galectin Therapeutics provides study drug • The Ludwig Institute and Cancer Center funds Stage 1 of the trial • Trial conducted in 6 centers in Europe (Belgium, France, Luxembourg) © 2011 Galectin Therapeutics OTC:GALT 30 |
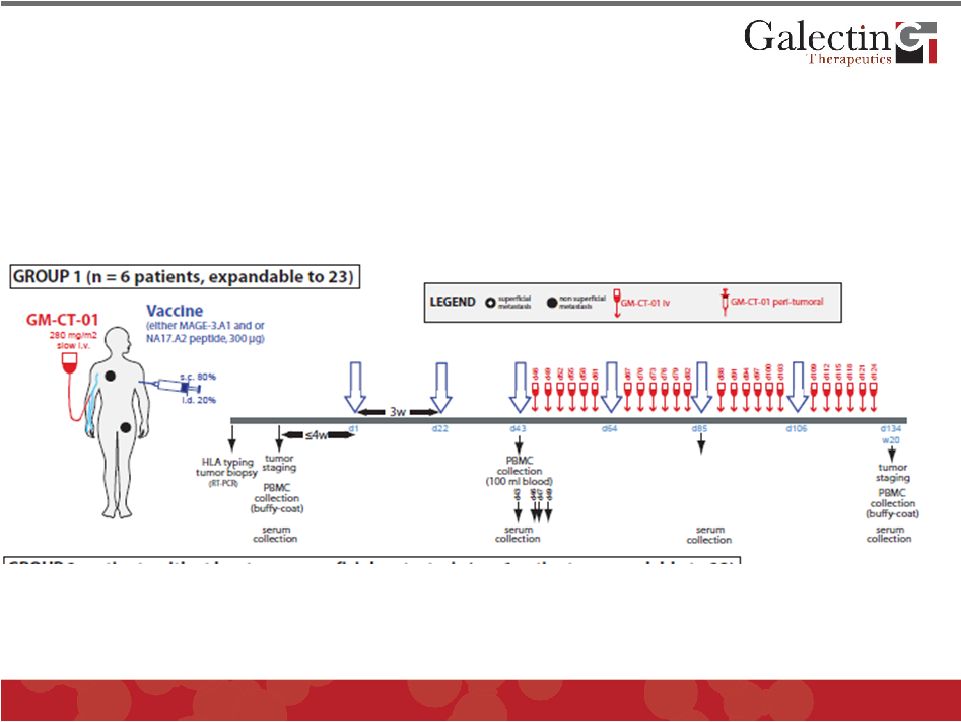 Melanoma Clinical Trial Design (I) © 2011 Galectin Therapeutics OTC:GALT 31 Phase I/II study of peptide vaccination associated with GM-CT-01, a galactomannan oligomer that inhibits galectin-3, in patients with advanced metastatic melanoma |
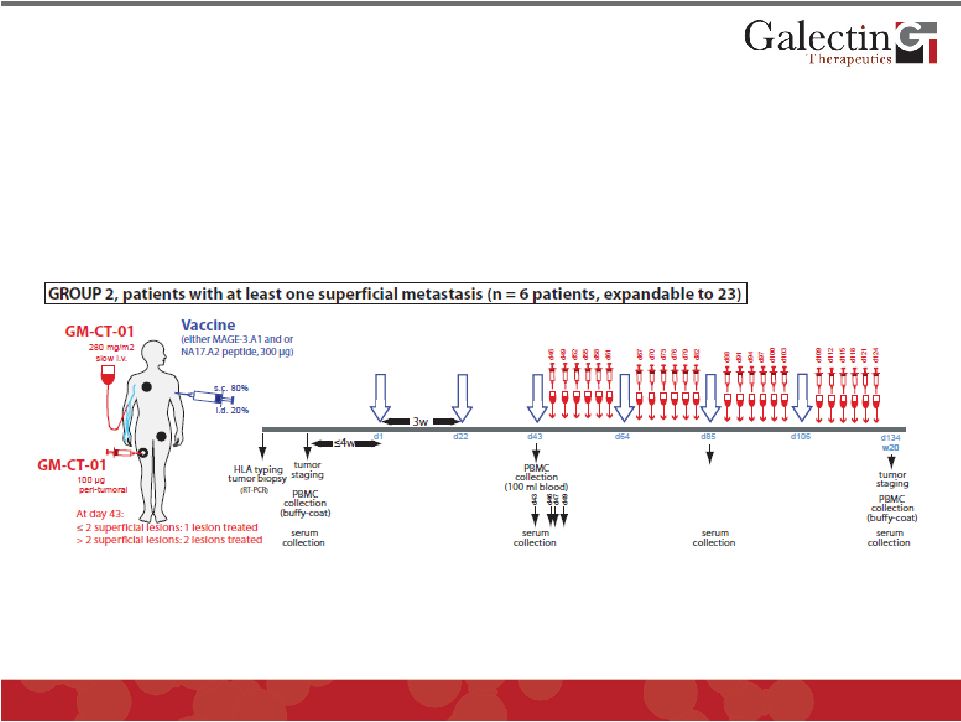 Melanoma Clinical Trial Design (II) © 2011 Galectin Therapeutics OTC:GALT 32 Phase I/II study of peptide vaccination associated with GM-CT-01, a galactomannan oligomer that inhibits galectin-3, in patients with advanced metastatic melanoma |
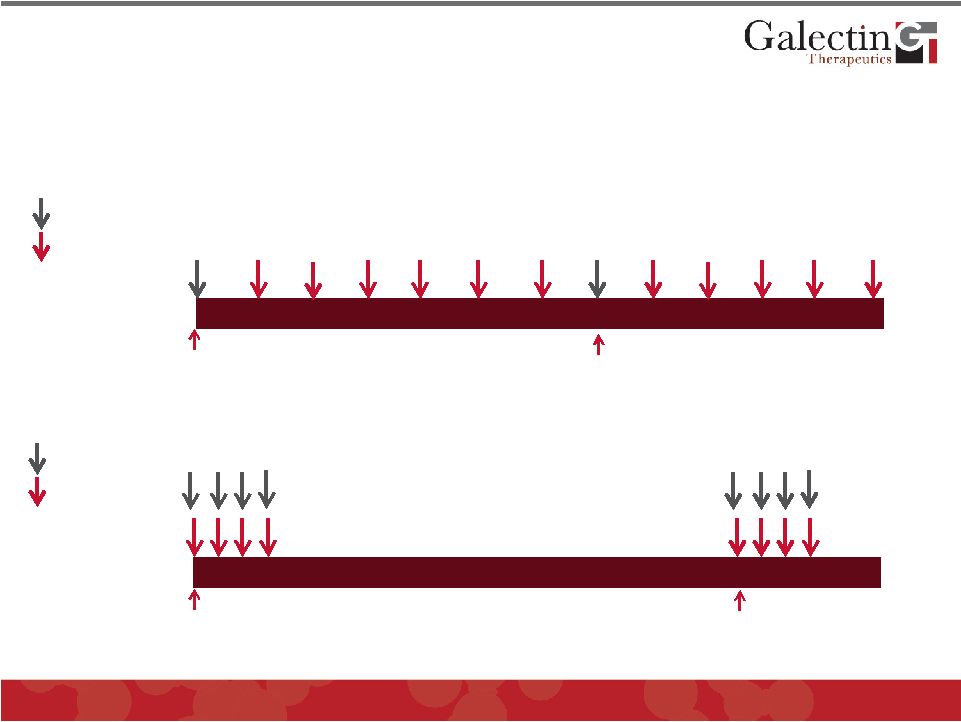 Regimen of GM-CT-01 in Melanoma Trial Versus Colorectal Cancer Trial © 2011 Galectin Therapeutics OTC:GALT 33 GM-CT-01 Melanoma Trial Day 0 Day 21 Vaccine Cancer Trial (DAVFU-003) Day 0 Day 28 GM-CT-01 Chemo (5-FU) |
 Development Program In Cancer Immunotherapy • Galectin proteins secreted by tumor cells are directly responsible for inhibiting the ability of immune cells to kill tumors • GM-CT-01 restores the ability of immune cells to kill tumor cells • Initial clinical trial for treatment of metastatic malignant melanoma • Market for tumor vaccines is expected to grow to $7B by 2015 • Potential important therapy for many cancers © 2011 Galectin Therapeutics OTC:GALT 34 |
 Pre-Clinical Phase 1 Phase 2 Phase 3 Registration Submitted Colorectal Cancer GM-CT-01 • International (Colombia) • United States Tumor Vaccine GM-CT-01 Liver Fibrosis GM-CT-01 GM-CT-02 GR-MD-01 GR-MD-02 Pipeline 35 © 2011 Galectin Therapeutics OTC:GALT |
 Catalyst Milestones • Fibrosis Program • Complete pre-clinical assessment and announce drug or drugs to take into clinical development: December 2011 • Commence phase 2 trial Q2 2012 with top line results Q2/Q3 2013 (GM-CT-01) • Commence phase 1 trial Q3 2012 and phase 2 trial Q4 2012 with top line results Q4 2013 (GR-MD series) • Tumor Immunology Program • Commence phase 1/2 trial Q4 2011 • Top line results on first stage second half of 2012 • Chemotherapy Program • Colombia final approval to market GM-CT-01 by Q2 2012 • Sales initiated 2012 © 2011 Galectin Therapeutics OTC:GALT 36 |
 Galectin Therapeutics Highlights • Leader in galectin science • Pipeline of carbohydrate-based drug compounds that inhibit galectins • Liver fibrosis program goal to be first therapy for this indication • Target validated in convincing pre-clinical data • Clinical trials expected to begin in 2012 • Cancer Therapy • Galectin inhibitor added to chemotherapy • Cancer immunotherapy program activates patient’s own immune system to kill tumor cells 37 © 2011 Galectin Therapeutics OTC:GALT |
 Thank You OTC: GALT |