 Corporate Summary April 2012 NASDAQ: GALT Exhibit 99.1 |
 Forward Looking Statements This presentation contains, in addition to historical information, forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements relate to future events or future financial performance, and use words such as “may,” “estimate,” “could,” “expect” and others. They are based on our current expectations and are subject to factors and uncertainties which could cause actual results to differ materially from those described in the statements. Factors that could cause our actual performance to differ materially from those discussed in the forward-looking statements include, among others: incurrence of operating losses since our inception, uncertainty as to adequate financing of our operations, extensive and costly regulatory oversight that could restrict or prevent product commercialization, inability to achieve commercial product acceptance, inability to protect our intellectual property, dependence on strategic partnerships, product competition, and others stated in risk factors contained in our SEC filings. We cannot assure that we have identified all risks or that others may emerge which we do not anticipate. You should not place undue reliance on forward-looking statements. Although subsequent events may cause our views to change, we disclaim any obligation to update forward-looking statements. © 2012 Galectin Therapeutics GALT 2 |
 Proprietary Compounds • First in class, proprietary compounds that inhibit galectin proteins • Complex carbohydrate drugs with highly favorable safety profile • • GR-MD-02: Preclinical: Non-alcoholic steatohepatitis (NASH, fatty liver disease) and other causes of liver fibrosis Validated Science • Pre-clinical models show galectins are critical targets for intended diseases with mechanisms that would be novel in the market Large Market Opportunities • Enhancing the ability of immune system to kill cancer cells is synergistic with many current and experimental therapies • NASH and liver fibrosis indications would be first therapies for completely unmet medical needs, representing a multi-billion dollar market Intellectual Property • Sole ownership, no licenses granted • GM-CT-01: Matter and methods granted (expire 2023) • GR-MD-02: Matter and methods pending (priorities of 2006-2011) Experienced Management Team • Management team has collective experience in multiple biotech and Pharma companies and relevant scientific areas © 2012 Galectin Therapeutics GALT 3 Investment Highlights GM-CT-01: Phase II: Melanoma; Enhances ability of immune cells to kill cancer cells |
 Experienced Management Team 4 Peter G. Traber, MD President, CEO, CMO • Over 25 years experience in biomedicine and pharmaceutical industries in research and development, clinical medicine, management and leadership, and business development. Medical expertise in liver disease • GlaxoSmithKline (CMO), Un of Pennsylvania (CEO), Baylor College of Medicine (CEO) Anatole Klyosov, PhD Chief Scientist • Over 35 years experience in biochemical reactions and their mechanisms, biotechnology, and carbohydrate research • Moscow University, Russian Academy of Sciences, Harvard Medical School Eliezer Zomer, PhD EVP, Product Development • Over 30 years experience in biotechnology engineering and regulatory in pharmaceuticals and diagnostics. • Koor Biotechnologies, Charm Sciences, Glycogenesis, HU Medical School (Jerusalem), Harvard University Thomas A. McGauley CFO (acting) • Over 10 years in accounting and finance with life science and technology companies • PricewaterhouseCoopers, Pro-Pharmaceuticals, deCode Genetics Maureen Foley COO • Over 30 years’ experience in business and operations management for public and private scientific, and biotech corporations and startup companies • eHealthDirect, Signatron, ArsDigita and Thermo Fibergen Elena Chekhova, PhD Program Manager • Over10 years of experience working in the biotech and life sciences industries, project management, manufacturing and business development. • Regis Tech., Decode, Zafgen, Boston College, Tokai Pharma, MIT, University of Dortmund, Harvard University © 2012 Galectin Therapeutics GALT |
 • Science of Galectins • Galectin Function • Galectin Inhibitors • Intellectual Property • Immune Enhancement in Cancer Therapy • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Liver Fibrosis • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Milestones 5 © 2012 Galectin Therapeutics GALT |
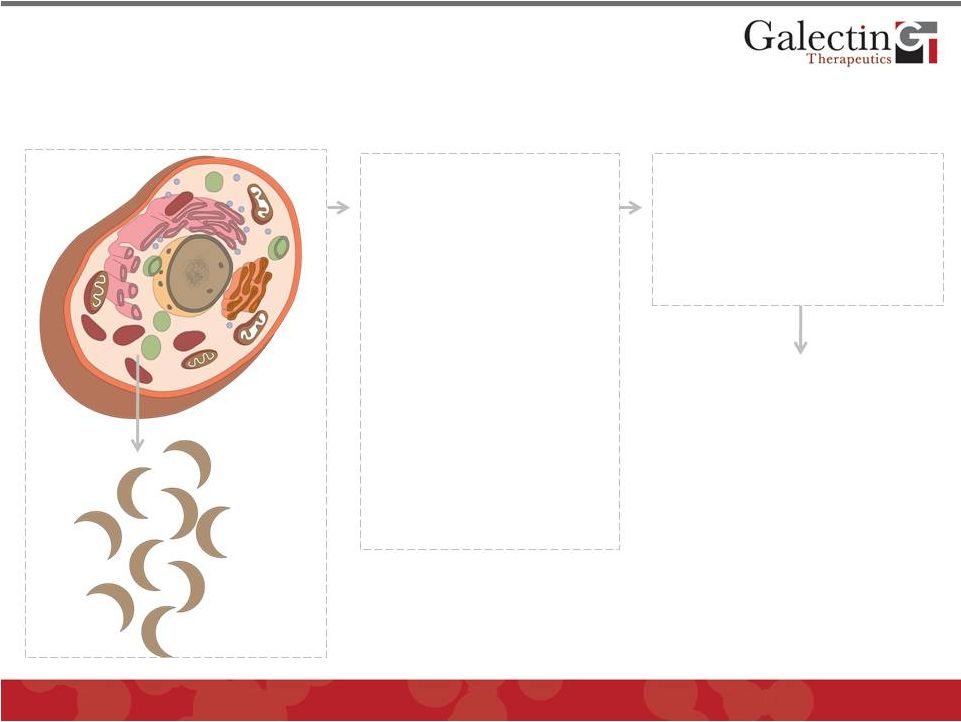 Galectin Proteins Are Critical Participants In Pathogenesis of Many Fibrotic and Neoplastic Diseases 6 © 2012 Galectin Therapeutics GALT Bind to cell surface and matrix glycoproteins (galactose residues) • Modulate cell signaling • Promote cell-cell interactions • Promote cell- matrix interactions Markedly Increased in: 1. Inflammation 2. Fibrosis 3. Cancer Galectin-3 is most prominent galectin secreted in disease *Secreted in small amounts normally by a number of cells, predominantly macrophages Secreted Galectin Proteins* GALECTINS PROMOTE PATHOLOGY |
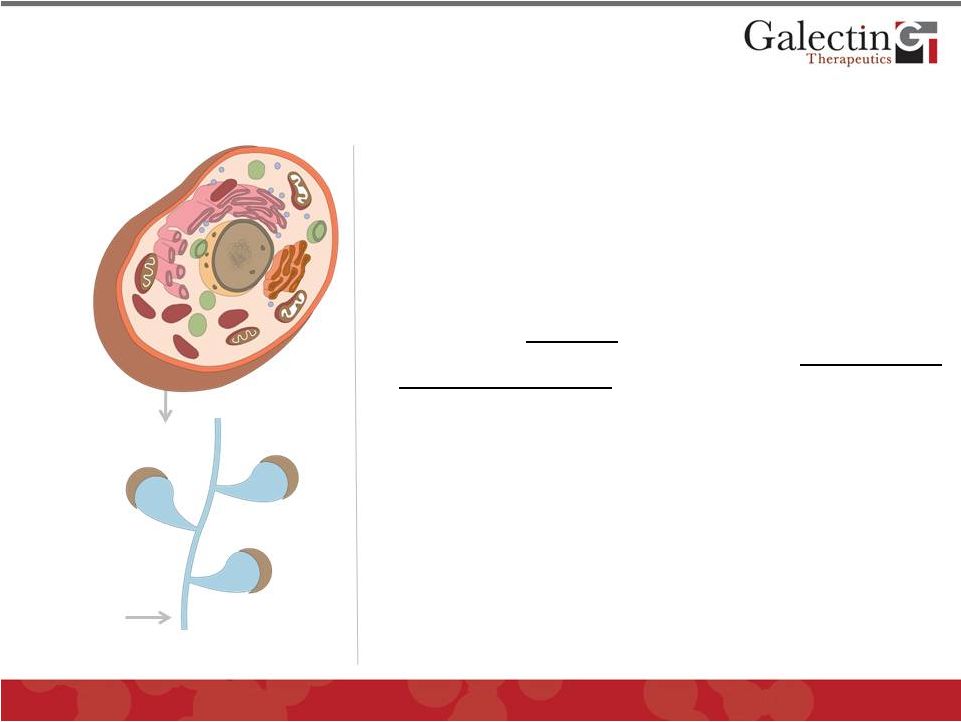 Galectin Inhibitors: A New Class of Pathology Modulators 7 • Novel complex carbohydrate drugs that target secreted and membrane-associated galectins by virtue of high molecular weight • Strongest binding to galectin-3, most prominent galectin in disease processes • Binding to galectins disrupts function and modulates multiple cellular pathways in pathology representing a potential new class of therapeutic agents • Low toxicity potential as a carbohydrate with no toxic metabolites • Two classes of compounds under development • GM-CT • GR-MD • Low manufacturing costs; abundant natural plant product starting materials © 2012 Galectin Therapeutics GALT Galectin Proteins Galectin Inhibitor |
 Intellectual Property • GM-CT Class (current NCE is GM-CT-01) • US Composition of matter patent Issued 2011 (priority 2003) • Five US issued method of use patents in combination with cancer therapy for increased efficacy and reduced side effects • International Patents: 13 granted and 5 pending • Method of use in liver fibrosis patent pending (priority 2006) • Method of use in NASH patent pending (priority 2011) • GR-MD Class (current NCE is GR-MD-02) • Composition of matter patent pending (priority 2011) • Method of use in liver fibrosis patent pending (priority 2006) • Method of use in NASH patent pending (priority 2011) • All intellectual property generated in house with no encumbrances • No established generic pathway for such complex carbohydrate drugs 8 © 2012 Galectin Therapeutics GALT |
 • Science of Galectins • Galectin Function • Galectin Inhibitors • Intellectual Property • Immune Enhancement in Cancer Therapy • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Liver Fibrosis • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Milestones 9 © 2012 Galectin Therapeutics GALT |
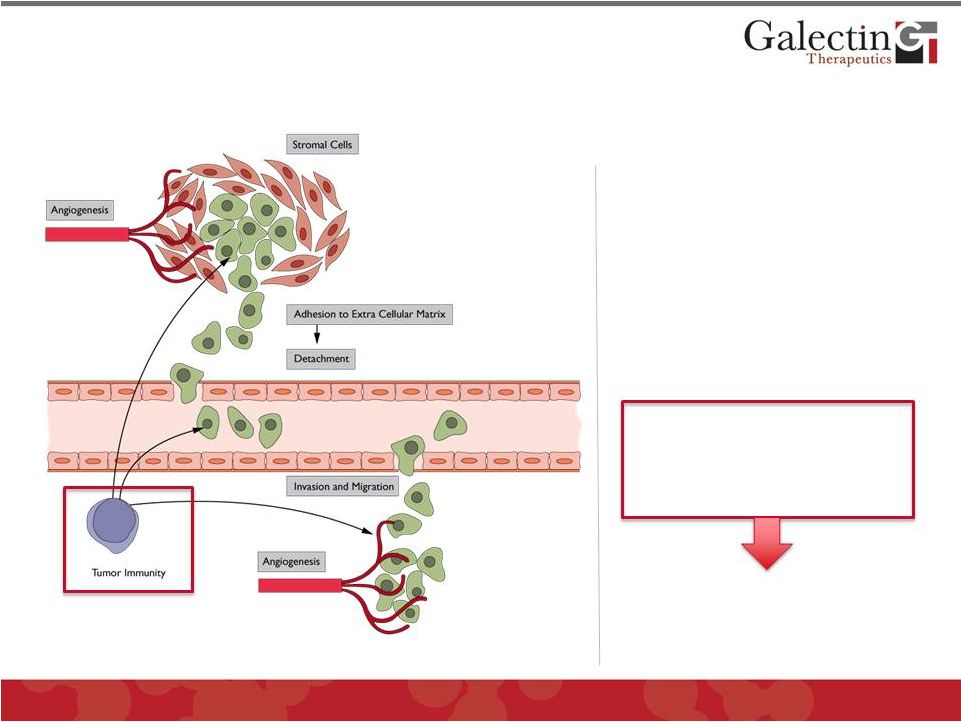 The Vast Majority of Cancers Secrete Large Amounts of Galectins Which Have Multiple Roles in Tumor Pathogenesis • Tumor cell invasion: extracellular matrix adhesion & detachment • Metastasis: cell invasion and migration • Angiogenesis • Tumor immunity has recently been shown to be critically affected by galectins 10 The “Galectin Effect” protects tumors from immune system © 2012 Galectin Therapeutics GALT |
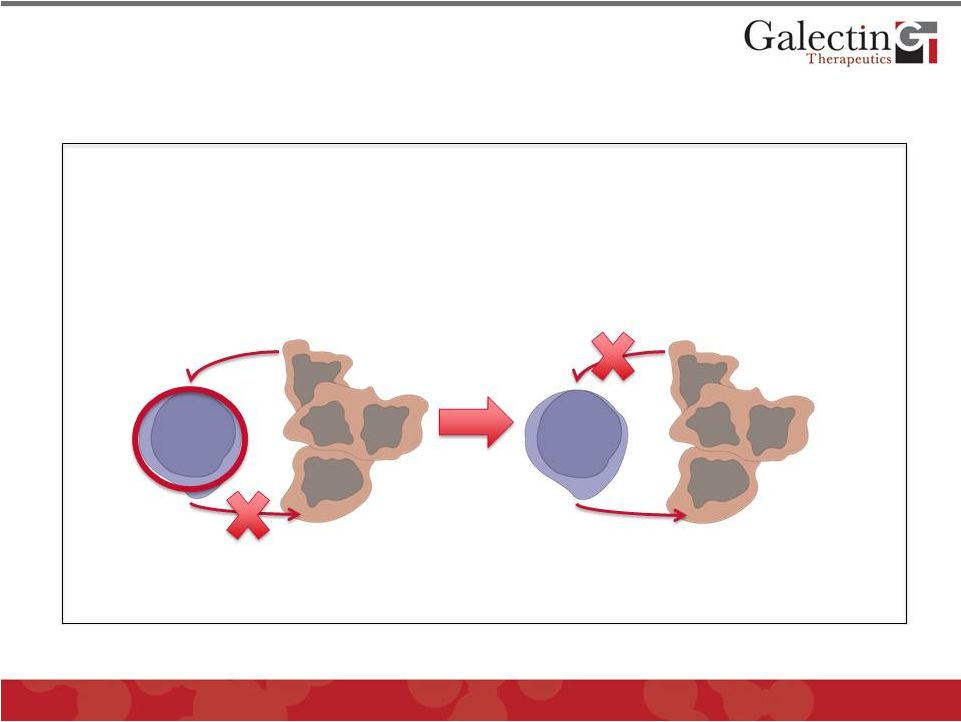 11 Experiments performed by Dr. Pierre van der Bruggen of the Ludwig Institute in Brussels, Belgium in collaboration with Galectin Therapeutics Tumors Evade the Immune System Using the “Galectin Effect” and GM-CT-01 Reverses This Effect © 2012 Galectin Therapeutics GALT Galectin-3 Cytokines Kill tumor cells GM-CT-01 Galectin-3 secreted by tumor cells binds to surface of T-cells and inhibits cytokine secretion Treatment with GM-CT-01 blocks Galectin-3 and restores T-cell cytokine secretion and tumor killing Tumor Cells T-Cells Tumor Cells Cytokines T-Cells |
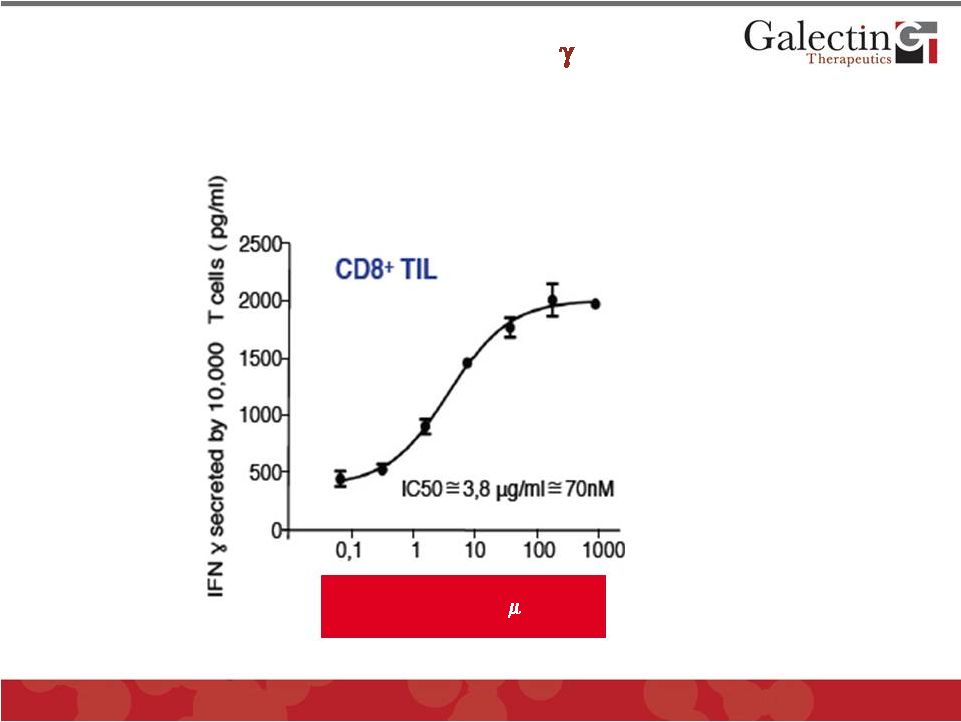 GM-CT-01 Activates Secretion of INF- in a Dose- Dependent Manner In Tumor Infiltrating CD8+ T- Cells From Human Patients 12 © 2012 Galectin Therapeutics GALT GM-CT-01 in g/ml |
 GM-CT-01 Restores Ability of Human CD8 T-Cells to Kill Tumor Cells Through Inhibition of “Galectin Effect” 13 Effectors: CD8+ T Cells Target: Tumor Cells © 2012 Galectin Therapeutics GALT |
 GM-CT-01 Has Demonstrated Safety in over 100 Human Subjects in Phase I and Partially Completed Phase II Clinical Trials with Some Evidence of Efficacy • Phase I trial (DAVFU-001) in 40 subjects with end stage cancer showed GM-CT-01 was safe alone and in combination with the chemotherapy 5-FU • Three Phase II trials were conducted, but only partially completed • One Phase II trial (DAVFU-001) of 5-FU plus GM-CT-01 in line 3/4 therapy of metastatic colorectal cancer showed 6.7 months median survival. In similar patients, Erbitux ®* had a 6.1 month survival compared to 4.6 months with no therapy • When the data from the three partially completed Phase II trials were pooled , the serious adverse events associated with 5-FU were reduced when compared to historical controls • The company is seeking partners with significant chemotherapy business for pursuing an indication for reduction in side effects for 5-FU and leucovorin containing chemotherapy regimens. • Our preclinical efficacy and clinical safety data were strong enough to obtain an IMPD for Phase I/II trial in metastatic melanoma with a combination of a tumor vaccine and GM-CT-01 to test the efficacy of blocking the “Galectin Effect” [Study not being conducted under FDA IND, but there is open IND for GM-]CT-01 14 *“Erbitux® is a registered trademark of ImClone LLC, a wholly-owned subsidiary of Eli Lilly and Company.“ © 2012 Galectin Therapeutics GALT |
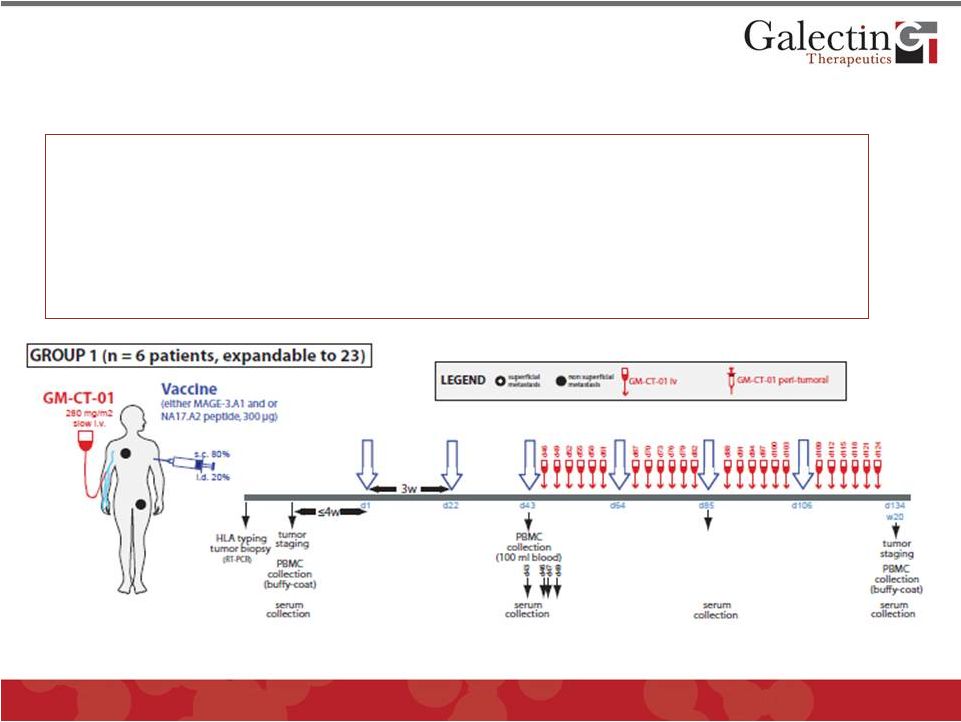 Preclinical efficacy and clinical safety data sufficient to obtain an IMPD for treatment of metastatic melanoma to test the efficacy of blocking the “Galectin Effect” 15 Melanoma “Proof of Concept” Trial: Patients: Advanced metastatic melanoma Design: Two Stage (12 in stage 1 and 46 in stage 2) Regimen: Prime with melanoma specific peptide vaccine Treat with GM-CT-01 to block “Galectin Effect” Endpoint: Partial or complete response by imaging Group 2 patients have additional injection of GM-CT-01 in cutaneous tumors © 2012 Galectin Therapeutics GALT |
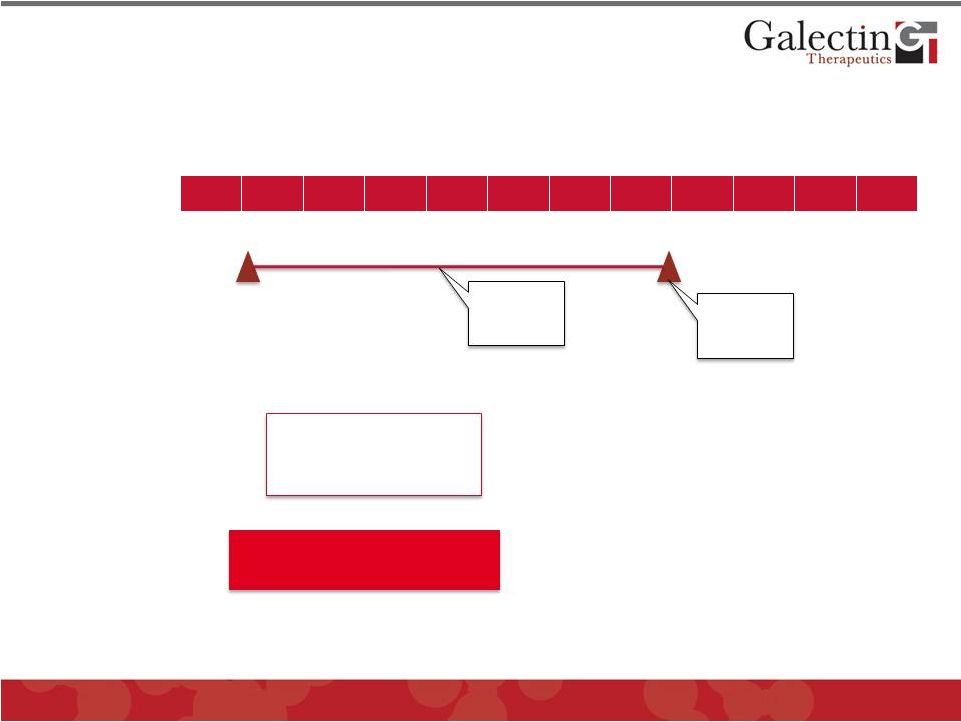 Tumor Immune Enhancement Development Program 16 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 2012 2013 2014 Preclinical Efficacy Studies GM-CT-01 Phase I/II Melanoma Trial* *Conducted in Belgium under an IMPD. Not conducted under FDA IND, but there is an open IND for GM-CT-01 At conclusion of Stage 1 of study: • • • • © 2012 Galectin Therapeutics GALT Pursue Partnering Discussions Efficacy in immune competent mice with syngeneic tumors Top line results in Stage 1 Top line results in Stage 2 Seek partnership with company with marketed or late stage cancer immunotherapy Consider Phase IIb controlled trial with strong efficacy Continue to Stage 2 with some efficacy Stop for lack of efficacy |
 Immune Enhancement by Blocking “Galectin Effect” is Synergistic With Many Emerging Cancer Immunotherapies • Enhancing the ability of the immune system to recognize and kill tumor cells is a very active area in the personalized approach to cancer therapy. The “Galectin Effect” inhibits the immune system • Two agents have been approved for use to date • Dendritic cell vaccine: Provenge® (Dendreon) • T-cell activator (CTLA4 receptor mAb): Yervoy® (Ipilimumab, BMS) • Many more vaccines and activators in development • Our drugs reverse the “Galectin Effect” by which tumors inhibit the immune system and may be synergistic with all tumor immunotherapies. May be effective with unaltered immune system • While tumor vaccines are patient and tumor specific, reversal of the “Galectin Effect” appears to be universal. Initial Phase I/II clinical trial in Belgium in combination with a vaccine to treat metastatic melanoma • The tumor vaccine market is forecast to be over $7 billion by 2015 17 © 2012 Galectin Therapeutics GALT |
 Best Positioned to Advance Tumor Immunotherapy with Galectin Inhibitor • Market for tumor vaccines is expected to grow to $7B by 2015. If ipilimumab (Yervoy ®, BMS) is included, market is even larger • Blocking the “Galectin Effect” would be synergistic with all types of tumor vaccines or immune stimulatory approaches • In this regard, competition will come from other galectin-inhibitors • Galecto Biotech AG (Sweden): Discovery phase focusing on small molecule inhibitors • LaJolla Pharmaceuticals (CA): In Jan. 2012, they purchased GCS-100 from Solana Therapeutics (formally Prospect Therapeutics, formally Glycogenesis). GCS-100 is a natural product compound with claims for binding galectins; focused on blood cancers; significant side effects reported. • Mandel Med (Oakland, CA): Truncated galectin-3 protein. Not progressed into human trials and no active program currently. • Stelic Institute (Japan): Pre-clinical RNAi approach in inflammatory bowel disease • Galectin Therapeutics is best positioned with a human trial in cancer immunotherapy; GM-CT-01 has proved safe in Phase I and three partially completed Phase II trials. 18 © 2012 Galectin Therapeutics GALT |
 • Science of Galectins • Galectin Function • Galectin Inhibitors • Intellectual Property • Immune Enhancement in Cancer Therapy • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Liver Fibrosis • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Milestones 19 © 2012 Galectin Therapeutics GALT |
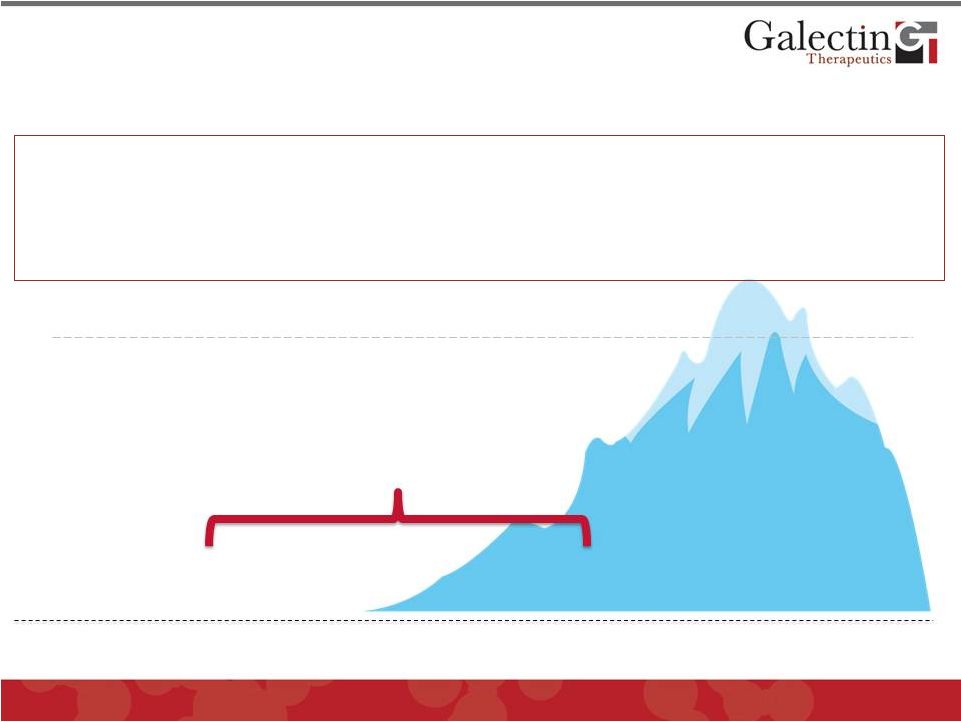 NASH and Liver Fibrosis are Multi- Billion Dollar Markets In US Alone * Performed in US in 2010 (UNOS) * * Prevalence in US 2010 (UNOS) 20 # Deaths in 1998 (AASLD Workshop, 2001) ## Prevalence in US 1976-1980 (NIDDK) & Prevalence in US 2011 (NIH) © 2012 Galectin Therapeutics GALT • The ONLY current therapy for advanced fibrosis (cirrhosis) is liver transplantation • No approved medical therapy for fibrosis • While there are treatments for some underlying etiologies (Hepatitis C and B), there is no approved therapy for NASH Transplants (6,291*) Wait List (17,000**) Death From Cirrhosis (44,677 # ) Cirrhosis (400,000 ## ) NASH: 9-15 Million & Hepatitis C, Hepatitis B, Alcohol |
 Galectin-3 Is A Critical Protein Target For Therapy of Liver Fibrosis Galectin-3 is produced in large amounts by fibrotic liver (animal and human) Galectin-3 is essential in mice for the development of liver fibrosis Fibrosis due to toxin exposure or fatty liver does not occur in mice that lack the galectin-3 gene Galectin inhibitors block production of fibrogenic markers in the key human cell (stellate cells) responsible for liver fibrosis Galectin inhibitors reverse experimental fibrosis in rats induced by both fibrosis and fatty liver 21 Key Evidence: Key Evidence: © 2012 Galectin Therapeutics GALT |
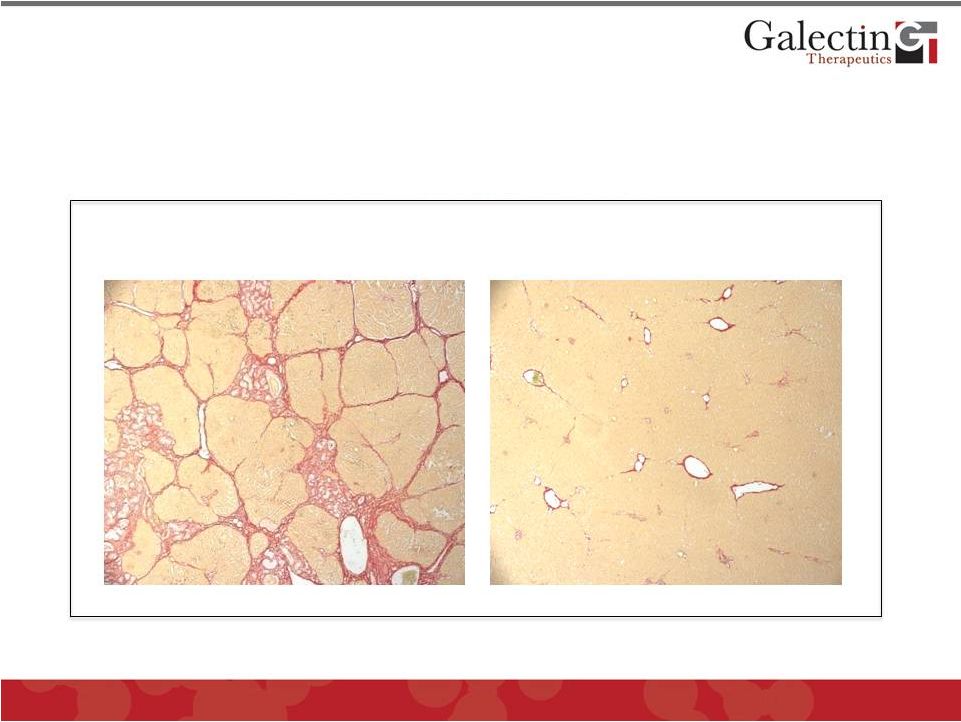 Galectin Inhibitor GR-MD-02 Effectively Treats Toxin-Induced Liver Fibrosis in Rats 22 Galectin Therapeutics Data Liver fibrosis induced in all rats by injection of chemical toxin (thioacetimide) for 8 weeks © 2012 Galectin Therapeutics GALT Treatment with vehicle alone for four weeks (Control) shows robust fibrosis Treatment with GR-MD-02 for four weeks shows dramatic regression of fibrosis |
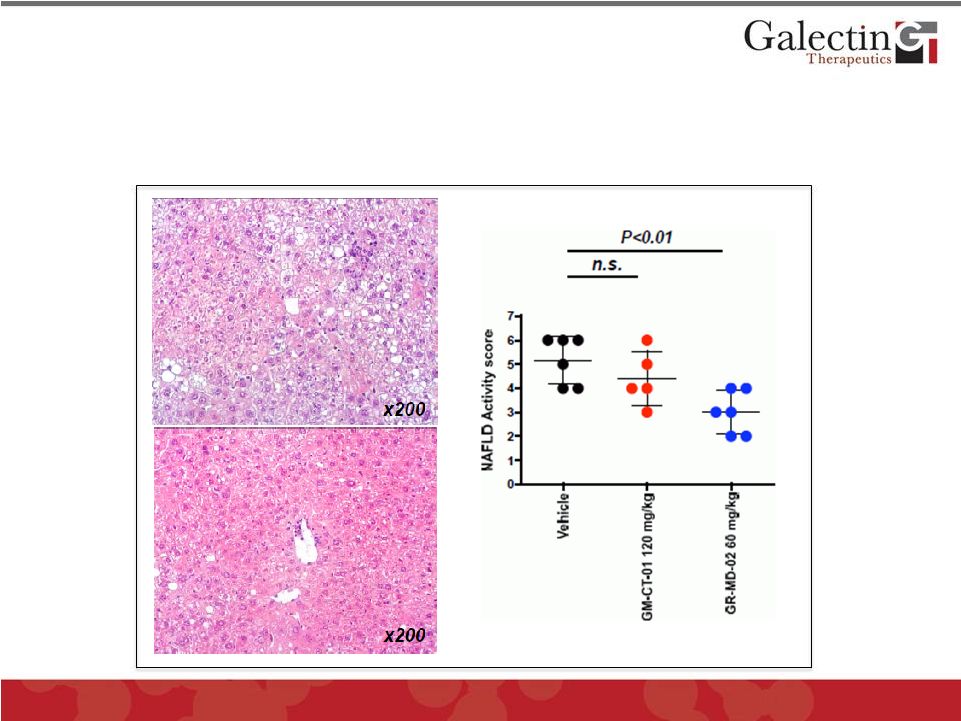 GR-MD-02 Markedly Improved NASH (Non-Alcoholic Steatohepatitis) in a Mouse Model Vehicle GR-MD-02 NASH was induced in mice by rendering them diabetic and feeding them a high fat diet © 2012 Galectin Therapeutics GALT |
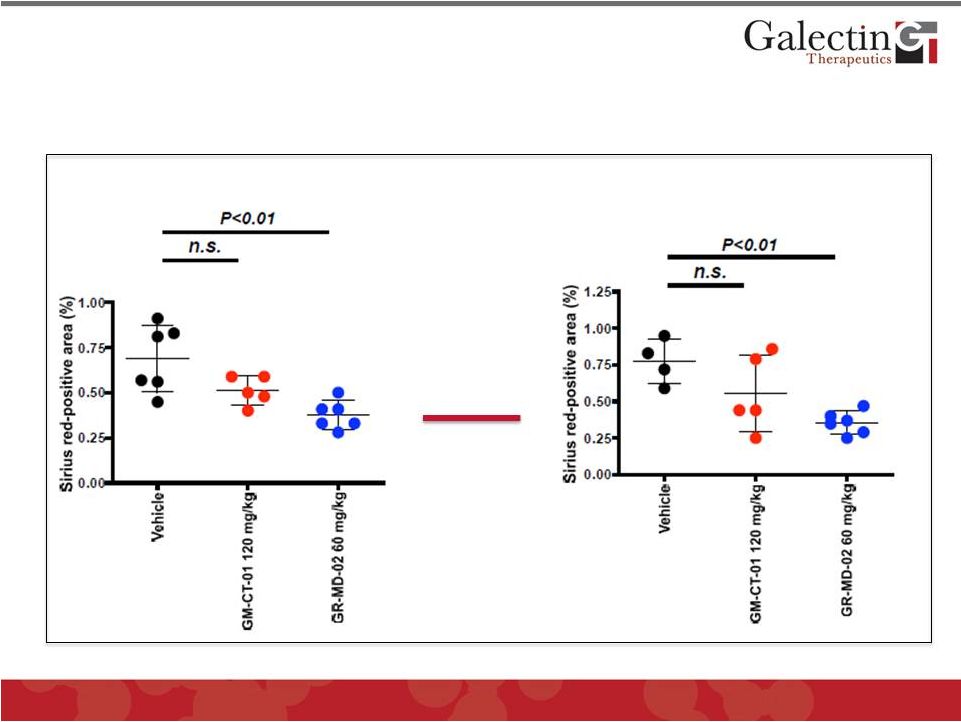 GR-MD-02 Prevented and Completely Reversed Fibrosis in NASH Mouse Model 24 Early Treatment Late Treatment normal mouse 0.33 © 2012 Galectin Therapeutics GALT |
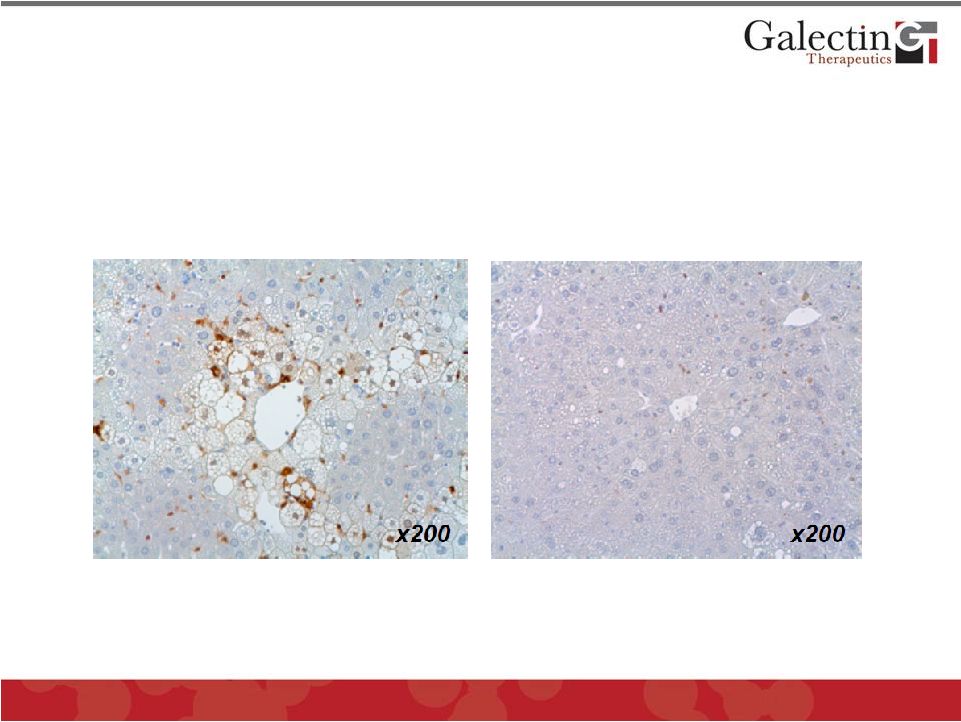 Treatment with GR-MD-02 Markedly Reduces Galectin-3 in NASH Mice 25 Vehicle GR-MD-02 Immunohistochemistry for detection of Galectin-3 © 2012 Galectin Therapeutics GALT |
 Inhibition of Gal-3 May Have Multiple Sites of Action in Therapy of NASH 26 Cause of Liver Injury Mediators Inflammation Fibrosis Resolution Metabolic Syndrome Glucose Intolerance Fat Accumulation Impaired Lipid Metabolism Hyperglycemia Adipocytokines Free Fatty Acids Extracellular Matrix Cytokines ECM Dissolution MMPs Inflammation ceases Stellate Cells Macrophages Gal-3 Gal-3 Gal-3 Gal-3 Stellate Cell Release of multiple inflammatory cytokines, including TGF- 1, a critical mediator of fibrosis Quiescent Activated Hepatocyte Macrophage (Kupffer Cell) T-Cell Bile Duct Cell Lipo-peroxidation Products Scavenger receptor increases cellular uptake of toxic products Inhibit scavenger mechanism and reduce cellular toxicity Galectin Functions GR-MD-02 Putative Mechanisms Increases TGF- receptor signaling Reduce cell membrane TGF- receptor activity Promotes cell-cell and cell matrix interactions which enable inflammatory networks Disrupt cell-cell signaling to reduce inflammatory cascade Enhance interactions between matrix protein components to stabilize matrix Destabilize matrix to make more available for degradation Increase expression of MMPs Modulate expression of Metaloproteases (MMPs) © 2012 Galectin Therapeutics GALT |
 27 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 2012 2013 2014 Phase I1 NASH NASH NASH Development Program: GR-MD-02 Phase 2 NASH Trial File Fast Track Designation Jan Jul Sep IND © 2012 Galectin Therapeutics GALT Phase I Trial: Patient Inclusion: Design: Primary Endpoint: Secondary Endpoints: Biopsy proven NASH with fibrosis Single dose escalation to target dose and then additional 10 patient extension for two months of therapy Patient safety Serum markers; MR-fat and elastography Phase II Trial: Patient Inclusion: Design: Primary Endpoint: Secondary Endpoints: Biopsy proven NASH with fibrosis Randomized, controlled, double blinded study with six months of therapy Liver biopsy NASH score and percent area collagen Safety; Serum markers; MR-fat and elastography Pre-Clinical (Target SQ) Top line results First patient enrolled Top line results |
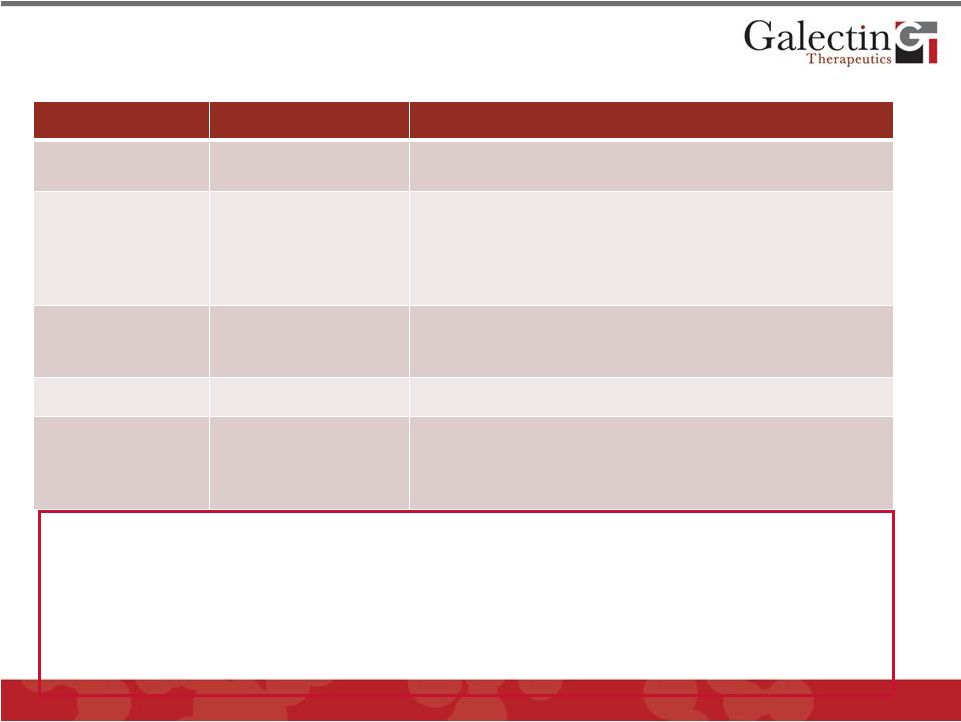 Among Competition Company is Best Positioned For a Successful Development Program in NASH General Mechanism Examples Comments Treat Diabetes and Insulin Resistance • Pioglitazone • Failed to achieve significant endpoints in phase II and phase III clinical trials Inhibit Lipid Metabolism • Aramchol • Colesevelam • Cholesterol inhibition, no clinical results; weak mechanism • Intestinal bile salt binder, no clinical results, weak mechanism Modulate the Immune System • EGS21 (Enzo) • Pentoxifylline • Abandoned after phase II trial • Non significant phase III results Protease Inhibition • GS-9450 (Gilead) • Liver Toxicity: abandoned (caspase inhibitor) Anti-Oxidant • MND-21 (Mochida) • Cysteamine (Raptor) • Omega-3 fatty acid (phase II trial) • Increase glutathione in liver cells, single point of action (phase II) • GR-MD-02 is well positioned with respect to competition • Most attractive mechanism: multiple sites of action in disease • Independent of hyperglycemia or hyperlipidemia • May reverse established fibrosis • Low toxicity potential as a carbohydrate with no toxic metabolites 28 © 2012 Galectin Therapeutics GALT |
 • Science of Galectins • Galectin Function • Galectin Inhibitors • Intellectual Property • Immune Enhancement in Cancer Therapy • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Liver Fibrosis • Mechanism of Action • Regulatory and Clinical Plan • Competitive Positioning • Milestones 29 © 2012 Galectin Therapeutics GALT |
 Company Milestones • General • 09/12: Host Global Conference on Galectins in Disease and Therapy; planned second edition of book on Galectins • Cancer • 03/12: Operational start of Phase I/II melanoma trial • 04/12: First patient infusedPhase I/II melanoma trial • 01/13: Stage 1 top line results (12 patients) • 01/14: Stage 2 top line results (46 patients) • Fibrosis • 05/12: NASH Presentation at DDW (Digestive Disease Week) • 12/12: NASH FDA IND • 01/13: Initiate Phase I NASH trial • 07/13: Phase I NASH trial results • 09/13: Initiate Phase II NASH trial • 10/14: Phase II NASH top line results 30 © 2012 Galectin Therapeutics GALT |
 Proprietary Compounds • First in class, proprietary compounds that inhibit galectin proteins • Complex carbohydrate drugs with highly favorable safety profile • GM-CT-01: Enhanced ability of immune cells to kill of cancer cells • GR-MD-02: Potential to treat non-alcoholic steatohepatitis (NASH) and other causes of liver fibrosis Validated Science • Pre-clinical models show galectins are critical targets for intended diseases with mechanisms that would be novel in the market Large Market Opportunities • Enhancing the ability of immune system to kill cancer cells is synergistic with many current and experimental therapies • NASH and liver fibrosis indications would be first therapies for completely unmet medical needs, representing a multi-billion dollar market Intellectual Property • Sole ownership, no licenses granted • GM-CT-01: Matter and methods granted (expire 2023) • GR-MD-02: Matter and methods pending (priorities of 2006-2011) Experienced Management Team • Management team has collective experience in multiple biotech and Pharma companies and relevant scientific areas 31 Investment Highlights © 2012 Galectin Therapeutics GALT |