change the estimated incidence or prevalence of these diseases. The number of patients may turn out to be lower than expected. Additionally, the potentially addressable patient population for each of our drug or biologic candidates may be limited or may not be amenable to treatment with our drug or biologic candidates, and new patients may become increasingly difficult to identify or gain access to, which would adversely affect our business, financial condition, results of operations and prospects.
We face substantial competition, and our competitors may discover, develop or commercialize drugs faster or more successfully than we do.
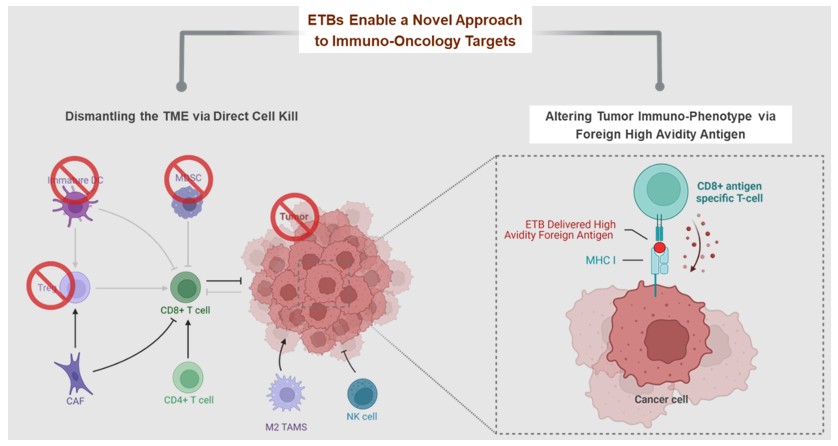
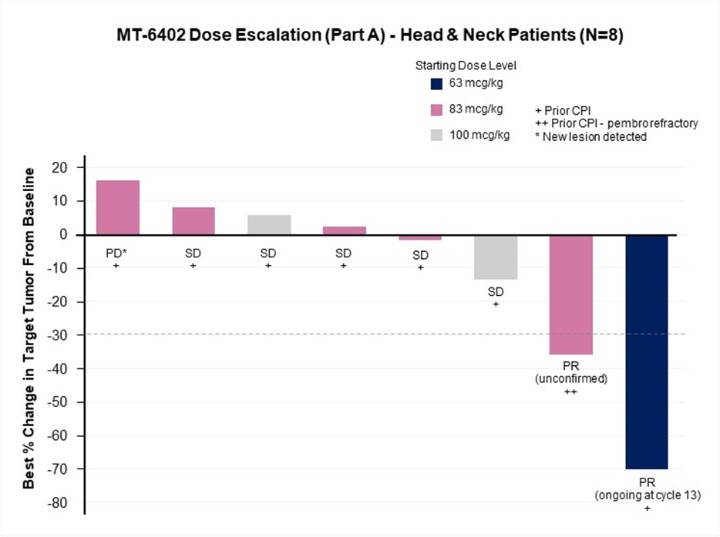
The development and commercialization of new drug products is highly competitive. We face competition from large pharmaceutical companies, specialty pharmaceutical companies, biotechnology companies, universities and other research institutions worldwide with respect to MT-6402, MT-8421, MT-0169, and the other drug or biologic candidates that we may seek to develop or commercialize in the future. We are aware that companies including the following have products marketed or in development that could compete directly or indirectly with ETBs: Merck, Bayer, Takeda, AbbVie, Immunogen, Morphosys, Genmab, Bristol-Myers Squibb, Novartis, Regeneron, Janssen, Xencor, Amgen, AstraZeneca, Lilly, Merck KGaA, Pfizer, Sanofi, Spectrum Pharmaceuticals, Cogent Biosciences, Karyopharm, ADC Therapeutics, 2seventy bio, Gilead, GlaxoSmithKline, Incyte, TG Therapeutics, Mersana Therapeutics, Seagen, and Verastem. Our competitors may succeed in developing, acquiring or licensing technologies or drug or biological products that are more effective or less costly than MT-6402, MT-8421, MT-0169, or any other drug or biologic candidates that we are currently developing or that we may develop, which could render our drug or biologic candidates obsolete and noncompetitive.
Many of our competitors have materially greater name recognition and financial, manufacturing, marketing, research and drug development resources than we do. Additional mergers and acquisitions in the biotechnology and pharmaceutical industries may result in even more resources being concentrated in our competitors. Large pharmaceutical companies in particular have extensive expertise in preclinical and clinical testing and in obtaining regulatory approvals for drugs, including biologics. In addition, academic institutions, government agencies, and other public and private organizations conducting research may seek patent protection with respect to potentially competitive products or technologies. These organizations may also establish exclusive collaborative or licensing relationships with our competitors.
If our competitors obtain marketing approval from the FDA or comparable foreign regulatory authorities for their drug or biologic candidates more rapidly than we do, it could result in our competitors establishing a strong market position before we are able to enter the market. In addition, third-party payors, including governmental and private insurers, also may encourage the use of generic products. For example, if MT-6402, MT-8421, or MT-0169 is ultimately approved, it may be priced at a significant premium over other competitive products. This may make it difficult for, MT-6402, MT-8421, MT-0169, or any other of our future drugs or biologics to compete with these products. Failure of MT-6402, MT-8421, MT-0169 or any other of our drug or biologic candidates to effectively compete against established treatment options or in the future with new products currently in development would harm our business, financial condition, results of operations and prospects.
The commercial success of any of our current or future drug or biologic candidates will depend upon the degree of market acceptance by physicians, patients, third-party payors, and others in the medical community.
Even with the approvals from the FDA and comparable foreign regulatory authorities, the commercial success of our drugs will depend in part on the health care providers, patients and third-party payors accepting our drug or biologic candidates as medically useful, cost-effective and safe. Any product that we bring to the market may not gain market acceptance by physicians, patients or third-party payors. The degree of market acceptance of any of our drug candidates will depend on a number of factors, including but not limited to:
| ● | the efficacy of the product as demonstrated in clinical trials and potential advantages over competing treatments; |
| ● | the prevalence and severity of the disease and any side effects of the product; |