Filed Pursuant to Rule 424(b)(3) | |
Registration No. 333-142355 |
PROSPECTUS
3,500,000 SHARES
LOTUS PHARMACEUTICALS, INC.
COMMON STOCK
Our Common Stock is registered under Section 12(g) of the Securities Exchange Act of 1934, as amended, and is quoted on the over-the-counter market and prices are reported on the OTC Bulletin Board under the symbol “LTUS.” On April 24, 2007, the closing price as reported was $2.00 per share. Since our Common Stock trades below $5.00 per share, it is considered a "penny stock" and is subject to SEC rules and regulations that impose limitations on the manner in which it can be publicly traded.
The Selling Security-holders, and any participating broker-dealers may be deemed to be “underwriters” within the meaning of the Securities Act of 1933, as amended, and any commissions or discounts given to any such broker-dealer may be regarded as underwriting commissions or discounts under the Securities Act of 1933. The Selling Security-holders have informed us that they do not have any agreement or understanding, directly or indirectly, with any person to distribute their common stock. We agreed to pay the expenses of registering the foregoing shares of our Common Stock.
INVESTMENT IN THE COMMON STOCK OFFERED BY THIS PROSPECTUS INVOLVES A HIGH DEGREE OF RISK. YOU MAY LOSE YOUR ENTIRE INVESTMENT. CONSIDER CAREFULLY THE “RISK FACTORS” BEGINNING ON PAGE 4 OF THIS PROSPECTUS BEFORE INVESTING.
NEITHER THE SECURITIES AND EXCHANGE COMMISSION NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED OF THESE SECURITIES OR DETERMINED IF THIS PROSPECTUS IS TRUTHFUL OR COMPLETE. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENSE.
The date of this prospectus is May 7, 2007.
You should rely only on the information contained in or incorporated by reference in this prospectus. We have not, and the Selling Security-holders have not, authorized anyone, including any salesperson or broker, to give oral or written information about this offering, Lotus Pharmaceuticals, Inc., or the shares of common stock offered hereby that is different from the information included in this prospectus. If anyone provides you with different information, you should not rely on it. We are not, and the Selling Security-holders are not, making an offer to sell these securities in any jurisdiction where the offer or sale is not permitted. You should assume that the information contained in this prospectus is accurate only as of the date on the front cover of this prospectus. Our business, financial condition, results of operations and prospects may have changed since that date.
This prospectus is not an offer to sell any securities other than the shares of common stock offered hereby. This prospectus is not an offer to sell securities in any circumstances in which such an offer is unlawful.
TABLE OF CONTENTS
| Special Note Regarding Forward-Looking Statements | 1 |
| Prospectus Summary | 2 |
| Summary Financial Information | 3 |
| Risk Factors | 4 |
| Use of Proceeds | 15 |
| Selling Security-holders | 15 |
| Plan of Distribution | 16 |
| Legal Proceedings | 18 |
| Directors, Executive Officers, Promoters, and Control Persons | 18 |
| Security Ownership of Certain Beneficial Owners and Management | 20 |
| Description of Securities | 22 |
| Legal Matters | 22 |
| Experts | 22 |
| Disclosure of Commission Position on Indemnification for Securities Act Liabilities | 22 |
| Business | 22 |
| Management’s Discussion and Analysis of Financial Condition and Results of Operations | 29 |
| Description of Property | 38 |
| Certain Relationships and Related Transactions; Director Independence | 38 |
| Market for Common Equity and Related Stockholder Matters | 40 |
| Executive Compensation | 40 |
| Financial Statements | 41 |
| Changes in and Disagreements with Accountants | 41 |
| Index to Financial Statements | F-1 |
i
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
| · | adverse economic conditions; | |
| · | inability to raise sufficient additional capital to implement our business plan; | |
| · | intense competition, from providers of services similar to those offered by us; | |
| · | unexpected costs and operating deficits, and lower than expected sales and revenues; |
| · | adverse results of any legal proceedings; | |
| · | inability to satisfy government and commercial customers using our technology; | |
| · | the volatility of our operating results and financial condition; | |
· | inability to attract or retain qualified senior management personnel, including sales and marketing, and technology personnel; and |
| · | other specific risks that may be alluded to in this Registration Statement on Form SB-2. |
All statements, other than statements of historical facts, included in this Registration Statement on Form SB-2 regarding our strategy, future operations, financial position, estimated revenue or losses, projected costs, prospects and plans and objectives of management are forward-looking statements. When used in this Registration Statement on Form SB-2, the words ‘‘will,’’ ‘‘may,’’ ‘‘believe,’’ ‘‘anticipate,’’ ‘‘intend,’’ ‘‘estimate,’’ ‘‘expect,’’ ‘‘project,’’ ‘‘plan’’ and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain such identifying words. All forward-looking statements speak only as of the date of this Registration Statement on Form SB-2. We do not undertake any obligation to update any forward-looking statements or other information contained herein. Potential investors should not place undue reliance on these forward-looking statements. Although we believe that our plans, intentions and expectations reflected in or suggested by the forward-looking statements in this Registration Statement on Form SB-2 are reasonable, no one can assure investors that these plans, intentions or expectations will be achieved. Important factors that could cause actual results to differ materially from expectations expressed herein are described under ‘‘Risk Factors’’ and elsewhere in this Registration Statement on Form SB-2. These cautionary statements and risk factors qualify all forward-looking statements attributable to information provided in this Registration Statement on Form SB-2 and on behalf of us or persons acting on our behalf.
Information regarding market and industry statistics contained in this Registration Statement on Form SB-2 is included based on information available to us that we believe is accurate. It is generally based on academic and other publications that are not produced for purposes of securities offerings or economic analysis. Forecasts and other forward-looking information obtained from these sources are subject to the same qualifications and the additional uncertainties accompanying any estimates of future market size, revenue and market acceptance of products and services. We have no obligation to update forward-looking information to reflect actual results or changes in assumptions or other factors that could affect those statements. See ‘‘Risk Factors’’ for a more detailed discussion of uncertainties and risks that may have an impact on future results.
We file reports with the Securities and Exchange Commission (“SEC”). You can read and copy any materials we file with the SEC at the SEC’s Public Reference Room, 100 F. Street, NE, Washington, D.C. 20549. You can obtain additional information about the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. In addition, the SEC maintains an Internet site (www.sec.gov) that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC, including us.
1
This summary highlights some information from this prospectus, and may not contain all of the information that is important to you. You should read the entire prospectus carefully, including the more detailed information regarding our company, the risks of purchasing our common stock discussed under “risk factors,” and our financial statements and the accompanying notes. In this prospectus, “we”, “us,” “Company” and “our”, refer to Lotus Pharmaceuticals, Inc., unless the context otherwise requires. Unless otherwise indicated, the term “year,” “fiscal year” or “fiscal” refers to our fiscal year ending December 31st. Unless we tell you otherwise, the term “common stock” as used in this prospectus refers to our Common Stock.
Our Business
We are a pharmaceutical company focused on developing, manufacturing and distributing drugs in the People's Republic of China (“China” or PRC”). Additionally, through our 10 retail pharmacy locations in Beijing, China, we sell Western and traditional Chinese medications as well as medical treatment equipment.
We originally became duly incorporated in the State of Nevada on January 28, 2004 under the name S.E. Asia Trading Company, Inc. (“SEAA”) to sell jewelry and home accessories. On September 6, 2006, we entered into a definitive Share Exchange Agreement with Lotus Pharmaceutical International, Inc. (“Lotus International”), a company incorporated under the laws of the State of Nevada on August 28, 2006 to develop and market pharmaceutical products in China. Pursuant to the Share Exchange Agreement, we acquired all of the outstanding common stock of Lotus International in exchange for issuing stock to Lotus International’s shareholders. On September 28, 2006, Lotus International became our wholly-owned subsidiary, and Lotus International’s shareholders became the majority owners of our voting stock. On December 6, 2006, we changed our name to Lotus Pharmaceuticals, Inc. (“Lotus”).
PRC law currently has limits on foreign ownership of certain companies. To comply with these foreign ownership restrictions, we operate our pharmaceutical business in PRC through Beijing Liang Fang Pharmaceutical Co., Ltd. (“Liang Fang”) and an affiliate of Liang Fang, Beijing En Zhe Jia Shi Pharmaceutical Co., Ltd. (“En Zhe Jia”), both of which are pharmaceutical companies headquartered in the PRC and organized under the laws of the PRC (hereinafter, referred to together as “Lotus East”). We entered into consulting service agreements and equity-related arrangements with Lotus East and its shareholders pursuant to which we provide technology consulting and other general business operation services to Lotus East. Through these contractual arrangements, we also have the ability to substantially influence Lotus East’s daily operations and financial affairs, appoint its senior executives and approve all matters requiring shareholder approval. As a result of these contractual arrangements, which enable us to control Lotus East, we are considered the primary beneficiary of Lotus East. Accordingly, we consolidate Lotus East’s results, assets and liabilities in our financial statements.
The Offering
Shares Outstanding | 41,280,000 | |
Prior to Offering | ||
Common Stock, $0.001 | 3,500,000 | |
par value | ||
Use of Proceeds | We will not receive any proceeds from the sale of the shares by the selling shareholders. | |
Risk Factors | See “Risk Factors.” | |
OTC Symbol | LTUS | |
Executive Offices | Our executive offices are located at Boca Corporate Plaza, 7900 Glades Road, Suite 420, Boca Raton, FL 33434. Our telephone number is (877) 801-0344 and our website is www.lotuseast.com. The information on our websites is not part of this prospectus. |
2
SUMMARY FINANCIAL INFORMATION
The following tables set forth the summary financial information for our Company and our subsidiaries as provided in our year end financial statements. You should read this information together with the financial statements and the notes thereto appearing elsewhere in this prospectus and the information under “Management's Discussion and Analysis of Financial Condition and Results of Operations.”
For the Year Ended December 31, 2006 (Audited) | For the Year Ended December 31, 2005 (Audited) | ||||||
| Revenues | $ | 36,207,689 | $ | 18,345,841 | |||
| Cost of Sales | $ | 26,523,039 | $ | 14,584,682 | |||
| Operating Expenses | $ | 5,258,089 | $ | 3,149,120 | |||
| Net Income (Loss) | $ | 4,100,737 | $ | 1,831,712 | |||
| Basic Net Income (Loss) per Share | $ | 0.10 | $ | 0.05 | |||
| Weighted Average Common Shares Outstanding | 40,360,530 | 40,041,600 | |||||
Condensed Consolidated Balance Sheet Data
As of December 31, 2006 (Audited) | ||||
| Total Current Assets | $ | 12,752,003 | ||
| Current Liabilities | $ | 4,105,927 | ||
| Total Liabilities | $ | 8,835,807 | ||
| Stockholders' Equity | $ | 11,764,926 | ||
3
RISK FACTORS
You should carefully consider the risks described below before buying shares of our Common Stock in this offering. Additional risks and uncertainties not currently known to us or that we currently deem immaterial may impair our business operations. If any of the adverse events described in this risk factors section actually occur, our business, results of operations and financial condition could be materially adversely affected, the trading price of our common stock could decline and you might lose all or part of your investment. We have had operating losses to date and cannot assure that we will be profitable in the foreseeable future. We make various statements in this section which constitute “forward-looking” statements under Section 27A of the Securities Act.
RISKS RELATED TO OPERATIONS
Our limited operating history makes it difficult to evaluate our future prospects and results of operations.
We have a limited operating history. We operate our pharmaceutical business in PRC through Beijing Liang Fang Pharmaceutical Co., Ltd. (“Liang Fang”) and an affiliate of Liang Fang, Beijing En Zhe Jia Shi Pharmaceutical Co., Ltd. (“En Zhe Jia”), both of which are limited liability companies headquartered in PRC and organized under the laws of PRC (hereinafter, referred to together as “Lotus East”), pursuant to certain contractual arrangements with Lotus East more fully described under "Business" below. Lotus East commenced operations in 1999. Accordingly, you should consider our future prospects in light of the risks and uncertainties experienced by early stage companies in evolving industries such as the pharmaceutical industry in China. Some of these risks and uncertainties relate to our ability to:
| · | maintain our market position in the pharmaceuticals business in China; | |
| · | offer new and innovative products to attract and retain a larger customer base; | |
| · | attract additional customers and increase spending per customer; | |
| · | increase awareness of our brand and continue to develop user and customer loyalty |
| · | respond to competitive market conditions; | |
| · | respond to changes in our regulatory environment; | |
| · | manage risks associated with intellectual property rights; |
| · | maintain effective control of our costs and expenses; |
| · | raise sufficient capital to sustain and expand our business; | |
| · | attract, retain and motivate qualified personnel; and | |
| · | upgrade our technology to support additional research and development of new products. |
If we are unsuccessful in addressing any of these risks and uncertainties, our business may be materially and adversely affected.
We may need additional financing to execute our business plan.
The revenues from the production and sale of pharmaceutical products and the projected revenues from these products are not adequate to support our expansion and product development programs. We will need substantial additional funds to build additional production facilities, pursue further research and development, obtain regulatory approvals, file, prosecute, defend and enforce our intellectual property rights, market our products, expand our presence in the retail market by opening additional pharmacy locations, and the acquisition of similar businesses. We will seek additional funds through public or private equity or debt financing, strategic transactions and/or from other sources. We could enter into collaborative arrangements for the development of particular products that would lead to our relinquishing some or all rights to the related technology or products.
There are no assurances that future funding will be available on favorable terms or at all. If additional funding is not obtained, we will need to reduce, defer or cancel development programs, planned initiatives or overhead expenditures, to the extent necessary. The failure to fund our capital requirements would have a material adverse effect on our business, financial condition and results of operations.
4
Our success depends on our relationships with third parties over whom we have limited control.
Due to the complexity of the process of developing pharmaceuticals, our core business depends on arrangements we might enter from time to time with other companies for the research, development, clinical testing, technology rights, manufacturing, marketing and commercialization of our products. There are no assurances that we will be able to establish or maintain collaborations or agreements with others, as may be required, that are important to our business on favorable terms, or at all.
A number of risks arise from our dependence on collaborative agreements with third parties and potential for failure to achieve such agreements. Product development and commercialization efforts could be adversely affected if any prospective collaborative partner:
| · | fails to engage in an agreement as we require or terminates or suspends its agreement with us; | |
| · | causes delays; | |
| · | fails to timely develop or manufacture in adequate quantities a substance needed in order to conduct clinical trials; |
| · | fails to adequately perform clinical trials; |
| · | determines not to develop, manufacture or commercialize a product to which it has rights; or | |
| · | otherwise fails to meet its contractual obligations. |
The profitability of our products will depend in part on our ability to obtain and maintain patents and manufacturing rights and preserve trade secrets, and the period our intellectual property remains protected. We must also operate without infringing the proprietary rights of third parties and without third parties circumventing our rights. The patent positions of pharmaceutical and biotechnology enterprises, including ours, are uncertain and involve complex legal and factual questions for which important legal principles are largely unresolved. The biotechnology patent situation outside the U.S. is uncertain, is currently undergoing review and revision in many countries, and may not protect our intellectual property rights to the same extent as the laws of the U.S. Because patent applications are maintained in secrecy in some cases, we cannot be certain that we or our licensors are the first creators of inventions described in our pending patent applications or patents or the first to file patent applications for such inventions.
Most of our drug products have been approved by the PRC’s Food and Drug Administration (SFDA) but have not received patent protection. For instance, Valsartan, one of our most profitable products, is produced by other companies in China, including Novartis. If Novartis or any other company were to obtain patent protection for Valsartan in China, or for any of our other drug products, it would have a material adverse effect on our revenue.
Other companies may independently develop similar products and design around any patented products we develop. We cannot assure you that:
| · | any of our patent applications will result in the issuance of patents; | |
| · | we will develop patentable products; |
| · | the manufacturing rights we have been issued will provide us with any competitive advantages; | |
| · | the patents of others will not impede our ability to do business; or |
| · | third parties will not be able to circumvent our manufacturing rights. |
A number of pharmaceutical, biotechnology, research and academic companies and institutions have developed technologies, filed patent applications or received patents on technologies that may relate to our business. If these technologies, applications or patents conflict with ours, the scope of any future patents could be limited or our patent applications could be denied. Our business may be adversely affected if competitors independently develop competing technologies, especially if we do not obtain, or obtain only narrow, patent protection. If patents that cover our activities are issued to other companies, we may not be able to obtain manufacturing rights at a reasonable cost, or at all; develop our technology; or introduce, manufacture or sell the products we have planned.
5
Patent litigation is becoming widespread in the biotechnology industry. Such litigation may affect our efforts to form collaborations, to conduct research or development, to conduct clinical testing or to manufacture or market any products under development. There are no assurances that our patents would be held valid or enforceable by a court or that a competitor’s technology or product would be found to infringe our patents in the event of patent litigation. Our business could be materially affected by an adverse outcome to such litigation. Similarly, we may need to participate in interference proceedings declared by the U.S. Patent and Trademark Office or equivalent international authorities to determine priority of invention. We could incur substantial costs and devote significant management resources to defend our patent position or to seek a declaration that another company’s patents are invalid.
Much of our know-how and technology may not be patentable, though it may constitute trade secrets. There are no assurances that we will be able to meaningfully protect our trade secrets. We cannot assure you that any of our existing confidentiality agreements with employees, consultants, advisors or collaborators will provide meaningful protection for our trade secrets, know-how or other proprietary information in the event of any unauthorized use or disclosure. Collaborators, advisors or consultants may dispute the ownership of proprietary rights to our technology, for example by asserting that they developed the technology independently.
Before our products can be profitable, they must be produced in commercial quantities in a cost-effective manufacturing process that complies with regulatory requirements, including GMP, production and quality control regulations. If we cannot arrange for or maintain commercial-scale manufacturing on acceptable terms, or if there are delays or difficulties in the manufacturing process, we may not be able to conduct clinical trials, obtain regulatory approval or meet demand for our products. Production of our products could require raw materials which are scarce or which can be obtained only from a limited number of sources. If we are unable to obtain adequate supplies of such raw materials, the development, regulatory approval and marketing of our products could be delayed.
We could need more clinical trials or take more time to complete our clinical trials than we have planned.
Clinical trials vary in design by factors including dosage, end points, length, and controls. We may need to conduct a series of trials to demonstrate the safety and efficacy of our products. The results of these trials may not demonstrate safety or efficacy sufficiently for regulatory authorities to approve our products. Further, the actual schedules for our clinical trials could vary dramatically from the forecasted schedules due to factors including changes in trial design, conflicts with the schedules of participating clinicians and clinical institutions, and changes affecting product supplies for clinical trials.
We rely on collaborators, including academic institutions, governmental agencies and clinical research organizations, to conduct, supervise, monitor and design some or all aspects of clinical trials involving our products. Since these trials depend on governmental participation and funding, we have less control over their timing and design than trials we sponsor. Delays in or failure to commence or complete any planned clinical trials could delay the ultimate timelines for our product releases. Such delays could reduce investors’ confidence in our ability to develop products, likely causing our share price to decrease.
We may not be able to obtain the regulatory approvals or clearances that are necessary to commercialize our products.
The PRC and other countries impose significant statutory and regulatory obligations upon the manufacture and sale of pharmaceutical products. Each regulatory authority typically has a lengthy approval process in which it examines pre-clinical and clinical data and the facilities in which the product is manufactured. Regulatory submissions must meet complex criteria to demonstrate the safety and efficacy of the ultimate products. Addressing these criteria requires considerable data collection, verification and analysis. We may spend time and money preparing regulatory submissions or applications without assurances as to whether they will be approved on a timely basis or at all.
Our product candidates, some of which are currently in the early stages of development, will require significant additional development and pre-clinical and clinical testing prior to their commercialization. These steps and the process of obtaining required approvals and clearances can be costly and time-consuming. If our potential products are not successfully developed, cannot be proven to be safe and effective through clinical trials, or do not receive applicable regulatory approvals and clearances, or if there are delays in the process:
| · | the commercialization of our products could be adversely affected; |
6
| · | any competitive advantages of the products could be diminished; and | |
| · | revenues or collaborative milestones from the products could be reduced or delayed. |
Governmental and regulatory authorities may approve a product candidate for fewer indications or narrower circumstances than requested or may condition approval on the performance of post-marketing studies for a product candidate. Even if a product receives regulatory approval and clearance, it may later exhibit adverse side effects that limit or prevent its widespread use or that force us to withdraw the product from the market.
Any marketed product and its manufacturer will continue to be subject to strict regulation after approval. Results of post-marketing programs may limit or expand the further marketing of products. Unforeseen problems with an approved product or any violation of regulations could result in restrictions on the product, including its withdrawal from the market and possible civil actions.
In manufacturing our products we will be required to comply with applicable good manufacturing practices regulations, which include requirements relating to quality control and quality assurance, as well as the maintenance of records and documentation. We cannot comply with regulatory requirements, including applicable good manufacturing practice requirements, we may not be allowed to develop or market the product candidates. If we or our manufacturers fail to comply with applicable regulatory requirements at any stage during the regulatory process, we may be subject to sanctions, including fines, product recalls or seizures, injunctions, refusal of regulatory agencies to review pending market approval applications or supplements to approve applications, total or partial suspension of production, civil penalties, withdrawals of previously approved marketing applications and criminal prosecution.
Competitors may develop and market pharmaceutical products that are less expensive, more effective or safer, making our products obsolete or uncompetitive.
Some of our competitors and potential competitors have greater product development capabilities and financial, scientific, marketing and human resources than we do. Technological competition from pharmaceutical companies and biotechnology companies is intense and is expected to increase. Other companies have developed technologies that could be the basis for competitive products. Some of these products have an entirely different approach or means of accomplishing the desired curative effect than products we are developing. Alternative products may be developed that are more effective, work faster and are less costly than our products. Competitors may succeed in developing products earlier than us, obtaining approvals and clearances for such products more rapidly than us, or developing products that are more effective than ours. In addition, other forms of treatment may be competitive with our products. Over time, our technology or products may become obsolete or uncompetitive.
Our products may not gain market acceptance.
Our products may not gain market acceptance in the pharmaceutical community. The degree of market acceptance of any product depends on a number of factors, including establishment and demonstration of clinical efficacy and safety, cost-effectiveness, clinical advantages over alternative products, and marketing and distribution support for the products. Limited information regarding these factors is available in connection with our products or products that may compete with ours.
To directly market and distribute our pharmaceutical products, we or our collaborators require a marketing and sales force with appropriate technical expertise and supporting distribution capabilities. We may not be able to further establish sales, marketing and distribution capabilities or enter into arrangements with third parties on acceptable terms. If we or our partners cannot successfully market and sell our products, our ability to generate revenue will be limited.
Our operations and the use of our products could subject us to damages relating to injuries or accidental contamination.
Our research and development processes involve the controlled use of hazardous materials. We are subject to federal, provincial and local laws and regulations governing the use, manufacture, storage, handling and disposal of such materials and waste products. The risk of accidental contamination or injury from handling and disposing of such materials cannot be completely eliminated. In the event of an accident involving hazardous materials, we could be held liable for resulting damages. We are not insured with respect to this liability. Such liability could exceed our resources. In the future we could incur significant costs to comply with environmental laws and regulations.
7
If we were successfully sued for product liability, we could face substantial liabilities that may exceed our resources.
We may be held liable if any product we develop, or any product which is made using our technologies, causes injury or is found unsuitable during product testing, manufacturing, marketing, sale or use. These risks are inherent in the development of agricultural and pharmaceutical products. We currently do not have product liability insurance. We are not insured with respect to this liability. If we choose to obtain product liability insurance but cannot obtain sufficient insurance coverage at an acceptable cost or otherwise protect against potential product liability claims, the commercialization of products that we develop may be prevented or inhibited. If we are sued for any injury caused by our products, our liability could exceed our total assets.
We have limited business insurance coverage.
The insurance industry in China is still at an early stage of development. Insurance companies in China offer limited business insurance products. We do not have any business liability or disruption insurance coverage for our operations in China. Any business disruption, litigation or natural disaster may result in our incurring substantial costs and the diversion of our resources.
Dependence on key personnel
We depend on our key management and technological personnel. The unavailability or departure of such key personnel may seriously disrupt and harm our operations, business and the implementation of our business strategy and plans. Although most of these personnel are founders and shareholders of our company, there can be no assurance that we can be successful in retaining them.
Our success depends on attracting and retaining qualified personnel.
We depend on a core management and scientific team. The loss of any of these individuals could prevent us from achieving our business objective of commercializing our product candidates. Our future success will depend in large part on our continued ability to attract and retain other highly qualified scientific, technical and management personnel, as well as personnel with expertise in clinical testing and government regulation. We face competition for personnel from other companies, universities, public and private research institutions, government entities and other organizations. If our recruitment and retention efforts are unsuccessful, our business operations could suffer.
RISKS RELATED TO OUR CORPORATE STRUCTURE
PRC laws and regulations governing our businesses and the validity of certain of our contractual arrangements are uncertain. If we are found to be in violation, we could be subject to sanctions. In addition, changes in such PRC laws and regulations may materially and adversely affect our business.
There are substantial uncertainties regarding the interpretation and application of PRC laws and regulations, including, but not limited to, the laws and regulations governing our business, or the enforcement and performance of our contractual arrangements with our affiliated Chinese entity, Lotus East, and its shareholders. We are considered a foreign person or foreign invested enterprise under PRC law. As a result, we are subject to PRC law limitations on foreign ownership of Chinese companies.
These laws and regulations are relatively new and may be subject to change, and their official interpretation and enforcement may involve substantial uncertainty. The effectiveness of newly enacted laws, regulations or amendments may be delayed, resulting in detrimental reliance by foreign investors. New laws and regulations that affect existing and proposed future businesses may also be applied retroactively.
The PRC government has broad discretion in dealing with violations of laws and regulations, including levying fines, revoking business and other licenses and requiring actions necessary for compliance. In particular, licenses and permits issued or granted to us by relevant governmental bodies may be revoked at a later time by higher regulatory bodies. We cannot predict the effect of the interpretation of existing or new PRC laws or regulations on our businesses. We cannot assure you that our current ownership and operating structure would not be found in violation of any current or future PRC laws or regulations. As a result, we may be subject to sanctions, including fines, and could be required to restructure our operations or cease to provide certain services. Any of these or similar actions could significantly disrupt our business operations or restrict us from conducting a substantial portion of our business operations, which could materially and adversely affect our business, financial condition and results of operations.
8
We may be adversely affected by complexity, uncertainties and changes in PRC regulation of pharmaceutical business and companies, including limitations on our ability to own key assets.
The PRC government regulates the pharmaceutical industry including foreign ownership of, and the licensing and permit requirements pertaining to, companies in the pharmaceutical industry. These laws and regulations are relatively new and evolving, and their interpretation and enforcement involve significant uncertainty. As a result, in certain circumstances it may be difficult to determine what actions or omissions may be deemed to be a violation of applicable laws and regulations. Issues, risks and uncertainties relating to PRC government regulation of the pharmaceutical industry include the following:
| · | we only have contractual control over Lotus East. We do not own it due to the restriction of foreign investment in Chinese businesses; and | |
| · | uncertainties relating to the regulation of the pharmaceutical business in China, including evolving licensing practices, means that permits, licenses or operations at our company may be subject to challenge. This may disrupt our business, or subject us to sanctions, requirements to increase capital or other conditions or enforcement, or compromise enforceability of related contractual arrangements, or have other harmful effects on us. |
The interpretation and application of existing PRC laws, regulations and policies and possible new laws, regulations or policies have created substantial uncertainties regarding the legality of existing and future foreign investments in, and the businesses and activities of, pharmaceutical businesses in China, including our business.
In order to comply with PRC laws limiting foreign ownership of Chinese companies, we conduct our pharmaceutical business through Lotus East by means of contractual arrangements. If the PRC government determines that these contractual arrangements do not comply with applicable regulations, our business could be adversely affected.
The PRC government restricts foreign investment in pharmaceutical businesses in China. Accordingly, we operate our business in China through Lotus East. Lotus East holds the licenses and approvals necessary to operate our pharmaceutical business in China. We have contractual arrangements with Lotus East and its shareholders that allow us to substantially control Lotus East. We cannot assure you, however, that we will be able to enforce these contracts.
Although we believe we comply with current PRC regulations, we cannot assure you that the PRC government would agree that these operating arrangements comply with PRC licensing, registration or other regulatory requirements, with existing policies or with requirements or policies that may be adopted in the future. If the PRC government determines that we do not comply with applicable law, it could revoke our business and operating licenses, require us to discontinue or restrict our operations, restrict our right to collect revenues, require us to restructure our operations, impose additional conditions or requirements with which we may not be able to comply, impose restrictions on our business operations or on our customers, or take other regulatory or enforcement actions against us that could be harmful to our business.
Our contractual arrangements with Lotus East and its shareholders may not be as effective in providing control over these entities as direct ownership.
Since PRC law limits foreign equity ownership in companies in China, we operate our pharmaceutical business through two affiliated Chinese companies, collectively referred to as Lotus East. We have no equity ownership interest in Lotus East and rely on contractual arrangements to control and operate such business. These contractual arrangements may not be as effective in providing control over Lotus East as direct ownership. For example, Lotus East could fail to take actions required for our business despite its contractual obligation to do so. If Lotus East fails to perform under their agreements with us, we may have to rely on legal remedies under PRC law, which may not be effective. In addition, we cannot assure you that Lotus East’s shareholders would always act in our best interests.
The Chairman of the Board of Directors of Lotus East has potential conflicts of interest with us, which may adversely affect our business.
Liu Zhong Yi, our Chief Executive Officer, is also the Chairman of the Board of Directors of the two affiliated companies that comprise Lotus East. Conflicts of interests between his duties to our company and Lotus East may arise. As Dr. Liu is a director and executive officer of our company, he has a duty of loyalty and care to us under Nevada law when there are any potential conflicts of interests between our company and Lotus East. We cannot assure you, however, that when conflicts of interest arise, Dr. Liu will act completely in our interests or that conflicts of interests will be resolved in our favor. In addition, Dr. Liu could violate his legal duties by diverting business opportunities from us to others. If we cannot resolve any conflicts of interest between us and Dr. Liu, we would have to rely on legal proceedings, which could result in the disruption of our business.
9
RISKS RELATED TO DOING BUSINESS IN CHINA
Adverse changes in economic and political policies of the PRC government could have a material adverse effect on the overall economic growth of China, which could adversely affect our business.
Substantially all of our business operations are conducted in China. Accordingly, our results of operations, financial condition and prospects are subject to a significant degree to economic, political and legal developments in China. China’s economy differs from the economies of most developed countries in many respects, including with respect to the amount of government involvement, level of development, growth rate, control of foreign exchange and allocation of resources. While the PRC economy has experienced significant growth in the past 20 years, growth has been uneven across different regions and among various economic sectors of China. The PRC government has implemented various measures to encourage economic development and guide the allocation of resources. Some of these measures benefit the overall PRC economy, but may also have a negative effect on us. For example, our financial condition and results of operations may be adversely affected by government control over capital investments or changes in tax regulations that are applicable to us. Since early 2004, the PRC government has implemented certain measures to control the pace of economic growth. Such measures may cause a decrease in the level of economic activity in China, which in turn could adversely affect our results of operations and financial condition.
If PRC law were to phase out the preferential tax benefits currently being extended to foreign invested enterprises and “new or high-technology enterprises” located in a high-tech zone, we would have to pay more taxes, which could have a material and adverse effect on our financial condition and results of operations.
Under PRC laws and regulations, a foreign invested enterprise may enjoy preferential tax benefits if it is registered in a high-tech zone and also qualifies as “new or high-technology enterprise”. As a foreign invested enterprise as well as a certified “new or high-technology enterprise” located in a high-tech zone in Beijing, Lotus is registered in the Liangxiang Economic Tech Development Zone and is entitled to an exemption from enterprise income tax until December 12, 2008. If the new foreign investment company is founded before that and purchases Lotus, the new company will enjoy exemption of income tax for three additional years, which continues to 2010. Following the tax exemption period, we will enjoy a 50% tax break which means a tax rate of 7.5%.
If the PRC law were to phase out preferential tax benefits currently granted to “new or high-technology enterprises” and technology consulting services, we would be subject to the standard statutory tax rate, which currently is 33%, and we would be unable to obtain business tax refunds for our provision of technology consulting services. Loss of these preferential tax treatments could have a material and adverse effect on our financial condition and results of operations.
Lotus East is subject to restrictions on making payments to us.
We are a holding company incorporated in the State of Nevada and do not have any assets or conduct any business operations other than our investments in our affiliated entity in China, Lotus East. As a result of our holding company structure, we rely entirely on payments from Lotus East under our contractual arrangements. The PRC government also imposes controls on the conversion of RMB into foreign currencies and the remittance of currencies out of China. We may experience difficulties in completing the administrative procedures necessary to obtain and remit foreign currency. See “Government control of currency conversion may affect the value of your investment.”
Furthermore, if our affiliated entity in China incurs debt on its own in the future, the instruments governing the debt may restrict its ability to make payments. If we are unable to receive all of the revenues from our operations through these contractual or dividend arrangements, we may be unable to pay dividends on our ordinary shares.
Uncertainties with respect to the PRC legal system could adversely affect us.
We conduct our business primarily through our affiliated Chinese entity, Lotus East. Our operations in China are governed by PRC laws and regulations. We are generally subject to laws and regulations applicable to foreign investments in China and, in particular, laws applicable to wholly foreign-owned enterprises. The PRC legal system is based on written statutes. Prior court decisions may be cited for reference but have limited precedential value.
10
Since 1979, PRC legislation and regulations have significantly enhanced the protections afforded to various forms of foreign investments in China. However, China has not developed a fully integrated legal system and recently enacted laws and regulations may not sufficiently cover all aspects of economic activities in China. In particular, because these laws and regulations are relatively new, and because of the limited volume of published decisions and their nonbinding nature, the interpretation and enforcement of these laws and regulations involve uncertainties. In addition, the PRC legal system is based in part on government policies and internal rules (some of which are not published on a timely basis or at all) that may have a retroactive effect. As a result, we may not be aware of our violation of these policies and rules until some time after the violation. In addition, any litigation in China may be protracted and result in substantial costs and diversion of resources and management attention.
You may experience difficulties in effecting service of legal process, enforcing foreign judgments or bringing original actions in China based on United States or other foreign laws against us, our management or the experts named in the prospectus.
We conduct substantially all of our operations in China and substantially all of our assets are located in China. In addition, most of our senior executive officers reside within China. As a result, it may not be possible to effect service of process within the United States or elsewhere outside China upon our senior executive officers, including with respect to matters arising under U.S. federal securities laws or applicable state securities laws. Moreover, our PRC counsel has advised us that the PRC does not have treaties with the United States or many other countries providing for the reciprocal recognition and enforcement of judgment of courts.
Governmental control of currency conversion may affect the value of your investment.
The PRC government imposes controls on the convertibility of RMB into foreign currencies and, in certain cases, the remittance of currency out of China. We receive substantially all of our revenues in RMB. Under our current structure, our income is primarily derived from payments from Lotus East. Shortages in the availability of foreign currency may restrict the ability of our PRC affiliated entity to remit sufficient foreign currency to pay dividends or other payments to us, or otherwise satisfy their foreign currency denominated obligations. Under existing PRC foreign exchange regulations, payments of current account items, including profit distributions, interest payments and expenditures from trade-related transactions, can be made in foreign currencies without prior approval from the PRC State Administration of Foreign Exchange by complying with certain procedural requirements. However, approval from appropriate government authorities is required where RMB is to be converted into foreign currency and remitted out of China to pay capital expenses such as the repayment of bank loans denominated in foreign currencies.
The PRC government may also at its discretion restrict access in the future to foreign currencies for current account transactions. If the foreign exchange control system prevents us from obtaining sufficient foreign currency to satisfy our currency demands, we may not be able to pay dividends in foreign currencies to our shareholders.
Fluctuation in the value of RMB may have a material adverse effect on your investment.
The value of RMB against the U.S. dollar and other currencies may fluctuate and is affected by, among other things, changes in political and economic conditions. Our revenues and costs are mostly denominated in RMB, while a significant portion of our financial assets are denominated in U.S. dollars. We rely entirely on fees paid to us by our affiliated entity in China. Any significant fluctuation in value of RMB may materially and adversely affect our cash flows, revenues, earnings and financial position, and the value of, and any dividends payable on, our stock in U.S. dollars. For example, an appreciation of RMB against the U.S. dollar would make any new RMB denominated investments or expenditures more costly to us, to the extent that we need to convert U.S. dollars into RMB for such purposes. An appreciation of RMB against the U.S. dollar would also result in foreign currency translation losses for financial reporting purposes when we translate our U.S. dollar denominated financial assets into RMB, as RMB is our reporting currency.
We face risks related to health epidemics and other outbreaks.
Our business could be adversely affected by the effects of SARS or another epidemic or outbreak. China reported a number of cases of SARS in April 2004. Any prolonged recurrence of SARS or other adverse public health developments in China may have a material adverse effect on our business operations. For instance, health or other government regulations adopted in response may require temporary closure of our production facilities or of our offices. Such closures would severely disrupt our business operations and adversely affect our results of operations. We have not adopted any written preventive measures or contingency plans to combat any future outbreak of SARS or any other epidemic.
11
RISKS RELATED TO HOLDING OUR SECURITIES
To date, we have not paid any cash dividends, and no cash dividends will be paid in the foreseeable future.
We do not anticipate paying cash dividends on our common stock in the foreseeable future and we may not have sufficient funds legally available to pay dividends. Even if the funds are legally available for distribution, we may nevertheless decide not to pay any dividends. We intend to retain all earnings for the company's operations.
The application of the "penny stock" rules could adversely affect the market price of our common stock and increase your transaction costs to sell those shares.
As long as the trading price of our common shares is below $5 per share, the open-market trading of our common shares will be subject to the "penny stock" rules. The "penny stock" rules impose additional sales practice requirements on broker-dealers who sell securities to persons other than established customers and accredited investors (generally those with assets in excess of $1,000,000 or annual income exceeding $200,000 or $300,000 together with their spouse). For transactions covered by these rules, the broker-dealer must make a special suitability determination for the purchase of securities and have received the purchaser's written consent to the transaction before the purchase. Additionally, for any transaction involving a penny stock, unless exempt, the broker-dealer must deliver, before the transaction, a disclosure schedule prescribed by the Securities and Exchange Commission relating to the penny stock market. The broker-dealer also must disclose the commissions payable to both the broker-dealer and the registered representative and current quotations for the securities. Finally, monthly statements must be sent disclosing recent price information on the limited market in penny stocks. These additional burdens imposed on broker-dealers may restrict the ability or decrease the willingness of broker-dealers to sell our common shares, and may result in decreased liquidity for our common shares and increased transaction costs for sales and purchases of our common shares as compared to other securities.
Our common shares are thinly traded and you may be unable to sell at or near ask prices or at all if you need to sell your shares to raise money or otherwise desire to liquidate your shares.
The Company cannot predict the extent to which an active public market for its common stock will develop or be sustained. However, the Company does not rule out the possibility of applying for listing on the NASDAQ National Market or other exchanges.
Our common shares have historically been sporadically or "thinly-traded" on the “Over-the-Counter Bulletin Board”, meaning that the number of persons interested in purchasing our common shares at or near bid prices at any given time may be relatively small or non-existent. This situation is attributable to a number of factors, including the fact that we are a small company which is relatively unknown to stock analysts, stock brokers, institutional investors and others in the investment community that generate or influence sales volume, and that even if we came to the attention of such persons, they tend to be risk-averse and would be reluctant to follow an unproven company such as ours or purchase or recommend the purchase of our shares until such time as we became more seasoned and viable. As a consequence, there may be periods of several days or more when trading activity in our shares is minimal or non-existent, as compared to a seasoned issuer which has a large and steady volume of trading activity that will generally support continuous sales without an adverse effect on share price. We cannot give you any assurance that a broader or more active public trading market for our common stock will develop or be sustained, or that current trading levels will be sustained.
The market price for our common stock is particularly volatile given our status as a relatively small company with a small and thinly traded “float” and lack of current revenues that could lead to wide fluctuations in our share price. The price at which you purchase our common stock may not be indicative of the price that will prevail in the trading market. You may be unable to sell your common stock at or above your purchase price if at all, which may result in substantial losses to you.
The market for our common shares is characterized by significant price volatility when compared to seasoned issuers, and we expect that our share price will continue to be more volatile than a seasoned issuer for the indefinite future. The volatility in our share price is attributable to a number of factors. First, as noted above, our common shares are sporadically and/or thinly traded. As a consequence of this lack of liquidity, the trading of relatively small quantities of shares by our shareholders may disproportionately influence the price of those shares in either direction. The price for our shares could, for example, decline precipitously in the event that a large number of our common shares are sold on the market without commensurate demand, as compared to a seasoned issuer which could better absorb those sales without adverse impact on its share price. Secondly, we are a speculative or "risky" investment due to our uncertainty of future market acceptance for our current and potential products.
12
As a consequence of this enhanced risk, more risk-adverse investors may, under the fear of losing all or most of their investment in the event of negative news or lack of progress, be more inclined to sell their shares on the market more quickly and at greater discounts than would be the case with the stock of a seasoned issuer. The following factors may add to the volatility in the price of our common shares: actual or anticipated variations in our quarterly or annual operating results; adverse outcomes; the termination of our contractual agreements with Lotus East; and additions or departures of our key personnel, as well as other items discussed under this "Risk Factors" section, as well as elsewhere in this Current Report. Many of these factors are beyond our control and may decrease the market price of our common shares, regardless of our operating performance. We cannot make any predictions or projections as to what the prevailing market price for our common shares will be at any time, including as to whether our common shares will sustain their current market prices, or as to what effect that the sale of shares or the availability of common shares for sale at any time will have on the prevailing market price. However, the Company does not rule out the possibility of applying for listing on the NASDAQ National Market or other exchanges.
Shareholders should be aware that, according to SEC Release No. 34-29093, the market for penny stocks has suffered in recent years from patterns of fraud and abuse. Such patterns include (1) control of the market for the security by one or a few broker-dealers that are often related to the promoter or issuer; (2) manipulation of prices through prearranged matching of purchases and sales and false and misleading press releases; (3) boiler room practices involving high-pressure sales tactics and unrealistic price projections by inexperienced sales persons; (4) excessive and undisclosed bid-ask differential and markups by selling broker-dealers; and (5) the wholesale dumping of the same securities by promoters and broker-dealers after prices have been manipulated to a desired level, along with the resulting inevitable collapse of those prices and with consequent investor losses. Our management is aware of the abuses that have occurred historically in the penny stock market. Although we do not expect to be in a position to dictate the behavior of the market or of broker-dealers who participate in the market, management will strive within the confines of practical limitations to prevent the described patterns from being established with respect to our securities. The occurrence of these patterns or practices could increase the volatility of our share price.
Volatility in our common share price may subject us to securities litigation.
The market for our common stock is characterized by significant price volatility when compared to seasoned issuers, and we expect that our share price will continue to be more volatile than a seasoned issuer for the indefinite future. In the past, plaintiffs have often initiated securities class action litigation against a company following periods of volatility in the market price of its securities. We may, in the future, be the target of similar litigation. Securities litigation could result in substantial costs and liabilities and could divert management's attention and resources.
Our corporate actions are substantially controlled by our principal shareholders and affiliated entities.
Our principal shareholders and their affiliated entities own approximately 62% of our outstanding ordinary shares, representing approximately 62% of our voting power. These shareholders, acting individually or as a group, could exert substantial influence over matters such as electing directors and approving mergers or other business combination transactions. In addition, because of the percentage of ownership and voting concentration in these principal shareholders and their affiliated entities, elections of our board of directors will generally be within the control of these shareholders and their affiliated entities.
While all of our shareholders are entitled to vote on matters submitted to our shareholders for approval, the concentration of shares and voting control presently lies with these principal shareholders and their affiliated entities. As such, it would be difficult for shareholders to propose and have approved proposals not supported by management. There can be no assurances that matters voted upon by our officers and directors in their capacity as shareholders will be viewed favorably by all shareholders of the company.
The elimination of monetary liability against our directors, officers and employees under Nevada law and the existence of indemnification rights to our directors, officers and employees may result in substantial expenditures by our Company and may discourage lawsuits against our directors, officers and employees.
Our articles of incorporation do not contain any specific provisions that eliminate the liability of our directors for monetary damages to our company and shareholders, however we are prepared to give such indemnification to our directors and officers to the extent provided by Nevada law. We may also have contractual indemnification obligations under our employment agreements with our officers. The foregoing indemnification obligations could result in our company incurring substantial expenditures to cover the cost of settlement or damage awards against directors and officers, which we may be unable to recoup. These provisions and resultant costs may also discourage our company from bringing a lawsuit against directors and officers for breaches of their fiduciary duties, and may similarly discourage the filing of derivative litigation by our shareholders against our directors and officers even though such actions, if successful, might otherwise benefit our company and shareholders.
13
Legislative actions, higher insurance costs and potential new accounting pronouncements may impact our future financial position and results of operation.
There have been regulatory changes, including the Sarbanes-Oxley Act of 2002, and there may potentially be new accounting pronouncements or additional regulatory rulings that will have an impact on our future financial position and results of operations. The Sarbanes-Oxley Act of 2002 and other rule changes as well as proposed legislative initiatives following the Enron bankruptcy are likely to increase general and administrative costs and expenses. In addition, insurers are likely to increase premiums as a result of high claims rates over the past several years, which we expect will increase our premiums for insurance policies. Further, there could be changes in certain accounting rules. These and other potential changes could materially increase the expenses we report under generally accepted accounting principles, and adversely affect our operating results.
Past activities of the Company and its Affiliates may lead to future liability for the Company.
Prior to our entry into the Share Exchange Agreement with Lotus International on September 6, 2006, the Company engaged in businesses unrelated to its current operations. Although certain of our shareholders who were connected with such prior business are providing certain indemnifications against any loss, liability, claim, damage or expense arising out of or based on any breach of or inaccuracy in any of their representations and warranties made regarding such acquisition, any liabilities relating to such prior business against which we are not completely indemnified may have a material adverse effect on the Company.
The market price for our stock may be volatile.
The market price for our stock may be volatile and subject to wide fluctuations in response to factors including the following:
| · | actual or anticipated fluctuations in our quarterly operating results; | |
| · | announcements by us or our competitors of new products, acquisitions, strategic partnerships, joint ventures or capital commitments; | |
| · | addition or departure of key personnel; | |
| · | fluctuations of exchange rates between RMB and the U.S. dollar; | |
| · | intellectual property litigation; | |
| · | general economic or political conditions in China. |
In addition, the securities market has from time to time experienced significant price and volume fluctuations that are not related to the operating performance of particular companies. These market fluctuations may also materially and adversely affect the market price of our stock.
We may need additional capital, and the sale of additional shares or other equity securities could result in additional dilution to our shareholders.
We believe that our current cash and cash equivalents, anticipated cash flow from operations and the net proceeds from this offering will be sufficient to meet our anticipated cash needs for the near future. We may, however, require additional cash resources due to changed business conditions or other future developments, including any investments or acquisitions we may decide to pursue. If our resources are insufficient to satisfy our cash requirements, we may seek to sell additional equity or debt securities or obtain a credit facility. The sale of additional equity securities could result in additional dilution to our shareholders. The incurrence of indebtedness would result in increased debt service obligations and could result in operating and financing covenants that would restrict our operations. We cannot assure you that financing will be available in amounts or on terms acceptable to us, if at all.
14
If we fail to maintain an effective system of internal controls, we may not be able to accurately report our financial results or prevent fraud.
We will be subject to reporting obligations under the U.S. securities laws. The Securities and Exchange Commission, or the SEC, as required by Section 404 of the Sarbanes-Oxley Act of 2002, adopted rules requiring every public company to include a management report on such company’s internal controls over financial reporting in its annual report, which contains management’s assessment of the effectiveness of the company’s internal controls over financial reporting. In addition, an independent registered public accounting firm must attest to and report on management’s assessment of the effectiveness of the company’s internal controls over financial reporting. Our management may conclude that our internal controls over our financial reporting are not effective.
Moreover, even if our management concludes that our internal controls over financial reporting are effective, our independent registered public accounting firm may still decline to attest to our management’s assessment or may issue a report that is qualified if it is not satisfied with our controls or the level at which our controls are documented, designed, operated or reviewed, or if it interprets the relevant requirements differently from us. Our reporting obligations as a public company will place a significant strain on our management, operational and financial resources and systems for the foreseeable future. Effective internal controls, particularly those related to revenue recognition, are necessary for us to produce reliable financial reports and are important to help prevent fraud. As a result, our failure to achieve and maintain effective internal controls over financial reporting could result in the loss of investor confidence in the reliability of our financial statements, which in turn could harm our business and negatively impact the trading price of our stock. Furthermore, we anticipate that we will incur considerable costs and use significant management time and other resources in an effort to comply with Section 404 and other requirements of the Sarbanes-Oxley Act.
We will incur increased costs as a result of being a public company.
As a public company, we will incur significant legal, accounting and other expenses that we did not incur as a private company. In addition, the Sarbanes-Oxley Act, as well as new rules subsequently implemented by SEC have required changes in corporate governance practices of public companies. We expect these new rules and regulations to increase our legal, accounting and financial compliance costs and to make certain corporate activities more time-consuming and costly. In addition, we will incur additional costs associated with our public company reporting requirements. We are currently evaluating and monitoring developments with respect to these new rules, and we cannot predict or estimate the amount of additional costs we may incur or the timing of such costs.
We agreed to register for resale shares of common stock by the Selling Security-holders listed below. The Selling Security-holders may from time to time offer and sell any or all of their shares that are registered under this prospectus. All expenses incurred with respect to the registration of the common stock will be borne by us, but we will not be obligated to pay any underwriting fees, discounts, commissions or other expenses incurred by the Selling Security-holders in connection with the sale of such shares.
The following table sets forth information with respect to the maximum number of shares of common stock beneficially owned by the Selling Security-holders named below and as adjusted to give effect to the sale of the shares offered hereby. The shares beneficially owned have been determined in accordance with rules promulgated by the SEC, and the information is not necessarily indicative of beneficial ownership for any other purpose. The information in the table below is current as of the date of this prospectus. All information contained in the table below is based upon information provided to us by the Selling Security-holders, and we have not independently verified this information. The Selling Security-holders are not making any representation that any shares covered by the prospectus will be offered for sale. The Selling Security-holders may from time to time offer and sell pursuant to this prospectus any or all of the common stock being registered.
15
Except as indicated below, none of the Selling Security-holders have held any position or office with us, nor are any of the Selling Security-holders associates or affiliates of any of our officers or directors. Except as indicated below, no Selling Security-holder is the beneficial owner of any additional shares of common stock or other equity securities issued by us or any securities convertible into, or exercisable or exchangeable for, our equity securities. No Selling Security-holder is a registered broker-dealer or an affiliate of a broker-dealer.
For purposes of this table, beneficial ownership is determined in accordance with SEC rules, and includes voting power and investment power with respect to shares and shares owned pursuant to warrants exercisable within 60 days. The “Number of Shares Beneficially Owned After the Offering” column assumes the sale of all shares offered.
As explained below under “Plan of Distribution,” we have agreed with the Selling Security-holders to bear certain expenses (other than broker discounts and commissions, if any) in connection with this Registration Statement, which includes this prospectus.
Name | Number of Shares Beneficially Owned Prior to Offering | Number of Shares Offered | Number of Shares Beneficially Owned After the Offering | Percentage of Class of Shares Beneficially Owned After the Offering (4) | |||||||||
| Longview Fund LP | 4,125,000 (1 | ) | 2,750,000 | 1,375,000 | 3.22 | % | |||||||
| Alpha Capital Anstalt | 375,000 (2 | ) | 250,000 | 125,000 | * | ||||||||
| Genesis Technology Group, Inc. | 6,736,896 (3 | ) | 500,000 | 6,236,896 | 15.1 | % | |||||||
| Totals | 3,500,000 | ||||||||||||
(1) These shares represent 1,375,000 shares of common stock issuable upon exercise of the Selling Security-holder’s warrants at an exercise price of $1.50 per share and expiring on February 12, 2012, and 2,750,000 shares of common stock issuable upon conversion of a secured convertible note maturing on February 12, 2008, of which only the 2,750,000 conversion shares are being registered at this time. The convertible debt and warrants may not be converted or exercised if such conversion or exercise would cause the selling security-holder to hold more than 4.99% of our outstanding common stock. The natural person with voting and investment control over these shares is S. Michael Rudolph.
(2) These shares represent 125,000 shares of common stock issuable upon exercise of the Selling Security-holder’s warrants at an exercise price of $1.50 per share and expiring on February 12, 2012, and 250,000 shares of common stock issuable upon conversion of a secured convertible note maturing on February 12, 2008, of which only the 250,000 conversion shares are being registered at this time. The natural person with voting and investment control over these shares is Konrad Ackerman.
(3) These shares represent a total 6,736,896 common stock shares issued to the holder, 500,000 of which are being registered in this Registration Statement. The natural person with voting and investment control over these shares is Gary Wolfson. Genesis Equity Partners LLC, a 51% owned subsidiary of Genesis Technology Group, is currently a consultant to the Company.
(4) Percentage based upon 41,280,000 shares outstanding as of April 25, 2007.
PLAN OF DISTRIBUTION
The Selling Security-holders and any of their respective pledges, donees, assignees and other successors-in-interest may, from time to time, sell any or all of their shares of common stock on any stock exchange, market or trading facility on which the shares are traded or in private transactions. These sales may be at fixed or negotiated prices. The Selling Security-holders may use any one or more of the following methods when selling shares:
| o | ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
16
| o | block trades in which the broker-dealer will attempt to sell the shares as agent, but may position and resell a portion of the block as principal to facilitate the transaction; |
| o | purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
| o | an exchange distribution in accordance with the rules of the applicable exchange; |
| o | privately negotiated transactions; |
| o | short sales after this registration statement becomes effective; |
| o | broker-dealers may agree with the Selling Security-holders to sell a specified number of such shares at a stipulated price per share; |
| o | through the writing of options on the shares; |
| o | a combination of any such methods of sale; and |
| o | any other method permitted pursuant to applicable law. |
The Selling Security-holders may also sell shares under Rule 144 under the Securities Act of 1933, if available, rather than under this prospectus. The Selling Security-holders will have the sole and absolute discretion not to accept any purchase offer or make any sale of shares if they deem the purchase price to be unsatisfactory at any particular time.
The Selling Security-holders may also engage in short sales against the box after this registration statement becomes effective, puts and calls and other transactions in our securities or derivatives of our securities and may sell or deliver shares in connection with these trades.
The Selling Security-holders or their respective pledgees, donees, transferees or other successors in interest, may also sell the shares directly to market makers acting as principals and/or broker-dealers acting as agents for themselves or their customers. Such broker-dealers may receive compensation in the form of discounts, concessions or commissions from the Selling Security-holders and/or the purchasers of shares for whom such broker-dealers may act as agents or to whom they sell as principal or both, which compensation as to a particular broker-dealer might be in excess of customary commissions. Market makers and block purchasers purchasing the shares will do so for their own account and at their own risk. It is possible that a selling stockholder will attempt to sell shares of common stock in block transactions to market makers or other purchasers at a price per share which may be below the then market price. The Selling Security-holders cannot assure that all or any of the shares offered in this prospectus will be issued to, or sold by, the Selling Security-holders. The Selling Security-holders and any brokers, dealers or agents, upon effecting the sale of any of the shares offered in this prospectus, may be deemed to be "underwriters" as that term is defined under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, or the rules and regulations under such acts. In such event, any commissions received by such broker-dealers or agents and any profit on the resale of the shares purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act.
Discounts, concessions, commissions and similar selling expenses, if any, attributable to the sale of shares will be borne by the Selling Security-holder. The Selling Security-holders may agree to indemnify any agent, dealer or broker-dealer that participates in transactions involving sales of the shares if liabilities are imposed on that person under the Securities Act of 1933.
The Selling Security-holders may from time to time pledge or grant a security interest in some or all of the shares of common stock owned by them and, if they default in the performance of their secured obligations, the pledgee or secured parties may offer and sell the shares of common stock from time to time under this prospectus after we have filed an amendment to this prospectus under Rule 424(b)(3) or any other applicable provision of the Securities Act of 1933 amending the list of Selling Security-holders to include the pledgee, transferee or other successors in interest as Selling Security-holders under this prospectus.
17
The Selling Security-holders also may transfer the shares of common stock in other circumstances, in which case the transferees, pledgees or other successors in interest will be the selling beneficial owners for purposes of this prospectus and may sell the shares of common stock from time to time under this prospectus after we have filed an amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the Securities Act of 1933 amending the list of Selling Security-holders to include the pledgee, transferee or other successors in interest as Selling Security-holders under this prospectus.
We are required to pay all fees and expenses incident to the registration of the shares of common stock. We have agreed to indemnify the Selling Security-holders against certain losses, claims, damages and liabilities, including liabilities under the Securities Act of 1933.
Each of the Selling Security-holders acquired the securities offered hereby in the ordinary course of business and have advised us that they have not entered into any agreements, understandings or arrangements with any underwriters or broker-dealers regarding the sale of their shares of common stock, nor is there an underwriter or coordinating broker acting in connection with a proposed sale of shares of common stock by any selling stockholder. If we are notified by any selling stockholder that any material arrangement has been entered into with a broker-dealer for the sale of shares of common stock, if required, we will file a supplement to this prospectus. If the Selling Security-holders use this prospectus for any sale of the shares of common stock, they will be subject to the prospectus delivery requirements of the Securities Act of 1933.
The anti-manipulation rules of Regulation M under the Securities Exchange Act of 1934 may apply to sales of our common stock and activities of the Selling Security-holders.
LEGAL PROCEEDINGS
From time to time, we may be involved in litigation or other business disputes including patent infringement, defamation and unfair competition. The Company’s management is not aware of any material legal proceedings pending against the Company.
DIRECTORS, EXECUTIVE OFFICERS, PROMOTERS, AND CONTROL PERSONS
The following table sets forth the name, age, and position of each of the members of our board of directors, executive officers and promoters as of April 25, 2007:
Name | Age | Position | ||
| Dr. Liu Zhong Yi | 45 | Chief Executive Officer and Chairman of the Board | ||
| Mr. Adam Wasserman | 42 | Chief Financial Officer | ||
| Dr. Ian Ashley | 37 | Director | ||
| Mr. Li Ping | 44 | Director | ||
| Mr. Liu Jin | 67 | Director | ||
| Mr. Mel Rothberg | 59 | Director | ||
| Ms. Caeli Widger | 32 | Director | ||
| Ms. Xian Xuemei | 38 | Director | ||
| Mrs. Song Zhenghong | 39 | Director |
Current Management
Dr. Liu Zhong Yi is the Chairman of the Board, Chief Executive Officer and Founder of Lotus Pharmaceutical International, Inc. in the United States and its Chinese antecedent, Liang Fang Pharmaceuticals Co., Ltd. in Beijing. Dr. Liu holds the title of Deputy Chief Physician, as a professional apothecary, in operating Lotus (Liang Fang) as one of China’s most innovative and fastest growing pharmaceutical companies. As a researcher and medical student, Dr. Liu excelled in the development of many new drugs in wide use in China today. While working for the Chinese Government in 1992, he established the Research Center of Space Flight Biological Engineering Technology, and continued his renowned research related to incretion diseases. Four years later, entering the private sector and invigorated by burgeoning capitalism in China, Dr. Liu started his first pharmaceutical company in Beijing. He earned his Master’s Degree in Beijing hospitals, after doing his undergraduate studies in his homeland of Inner Mongolia. The Company does not currently have terms for its directors, and Dr. Liu will serve as a director of the Company until the next annual meeting and until a successor is elected and qualified.
18
Mr. Adam Wasserman has served as Chief Financial Officer for Lotus. He also serves as Chief Executive Officer of CFO Oncall, Inc. since October 1999. CFO Oncall, Inc. provides chief financial officer and controller services to companies on an outsourced basis. Mr. Wasserman currently serves as Chief Financial Officer for Transax International Limited since May 2005 and Chief Financial Officer for Genesis Technology Group, Inc. since 2000. From June 1991 to September 1999, Mr. Wasserman was an audit manager at American Express Tax & Business in Fort Lauderdale, Florida. During his tenure as manager, Mr. Wasserman successfully acted as an outsourced CFO and advisor to a diversified clientele in the wholesale, technology, distribution, medical, retail, and service industries in both the private and public sector. Prior to this, Mr. Wasserman served as senior auditor and staff accountant with Deloitte & Touche, LLP from June 1986 to May 1991. Mr. Wasserman holds a Bachelor of Administration from the New York State University at Albany, is a member of The American Institute of Certified Public Accountants and is the Treasurer and an Executive Board Member of the Gold Coast Venture Capital Association.
Dr. Ian Ashley is a Director on the founding Lotus Board of Directors. Previously he worked for Merck & Co. in Research and Development with focus on hypertension and calcium channel blockers. He is ABEM Board Certified in Emergency Medicine after finishing a residency in Emergency Medicine at Loma Linda University Medical Center in Southern California. Since 2002, Dr. Ashley has served as Attending Physician at Providence Hospital in Waco, Texas. Dr. Ashley graduated Summa Cum Laude with degrees in Chemistry and Biochemistry from Oberlin College before graduating from Baylor College of Medicine in Houston, Texas in 1996. The Company does not currently have terms for its directors, and Dr. Ashley will serve as a director of the Company until the next annual meeting and until a successor is elected and qualified.
Mr. Li Ping is a Director on the founding Lotus Board of Directors. Mr. Li served as salesman and deputy manager of the sales department of Beijing Dongcheng Medicine Wholesale Company in 1984-1999. Since 2000, he served as director of Lotus Pharmaceuticals and is responsible for medicine and clinic promotions. Mr. Li graduated from the Beijing Medical School, apothecary. The Company does not currently have terms for its directors, and Mr. Li will serve as a director of the Company until the next annual meeting and until a successor is elected and qualified.
Mr. Liu Jin is a Director on the founding Lotus Board of Directors. Mr. Liu served as an accountant for the Finance Bureau of Liangcheng County from 1958-1970 and the as accountant and accountant general for Finance Bureau of Chayouqiqnqi of Inner Mongolia. Since 2000, Mr. Liu has served as a director of Lotus Pharmaceuticals and has been responsible for production cost control. Mr. Liu graduated from the Middling Finance School of Wulanchabu City of Inner Mongolia in 1958. The Company does not currently have terms for its directors, and Mr. Liu will serve as a director of the Company until the next annual meeting and until a successor is elected and qualified.
Mr. Mel Rothberg is a Director on the founding Lotus Board of Directors. Mr. Rothberg has, over the last 30 years, participated in the development of the South Florida bioscience industry and is presently Chairman of Bioflorida’s South Florida Bioscience Consortium. Since October 2005, Mr. Rothberg has been Chairman and CEO of Advanced Processing and Imaging, CEO of Dharma Biomedical and CEO of Rothberg Associates, Inc. From March 1998 to April 2005, Mr. Rothberg was Executive Vice President, Operations of Viragen Inc., Plantation, Florida (VRA: AMEX) and Director of Viragen International Inc. (VGNI: OTCBB) and Viranative AB (Sweden), where he was responsible for manufacturing, regulatory, business development, marketing and sales of Viragen’s lead drug, Multiferon. Prior to Viragen, Mr. Rothberg was Vice President of Manufacturing and Business Development for Althin Medical a Swedish publicly traded company focused on the hemodialysis machine and disposables market. He previously held multiple roles with Dow Chemical and Cordis Dow and was instrumental in selling Dow’s dialysis division to Althin Medical. He held both international and domestic positions within Dow’s subsidiary CD Medical including responsibility for offices in Australia, Singapore and Japan and was Managing Director of Cordis Dow’s European manufacturing operations located in the Netherlands. The Company does not currently have terms for its directors, and Mr. Rothberg will serve as a director of the Company until the next annual meeting and until a successor is elected and qualified.
Ms. Caeli Widger is a Director on the founding Lotus Board of Directors. Since April 2000, Ms. Widger has directed large-scale staffing projects, ranging from sales force expansion to executive searches, for young technology companies on both coasts of the U.S. She is currently managing a talent search for Mimeo.com (www.mimeo.com), an international print-on-demand solutions provider named one of America's fastest-growing technology companies by Red Herring and the Deloitte Fast 50 List. Ms. Widger co-directs The Sackett School, a renowned creative writing institute in New York City (www.sackettworkshop.com). She holds a BA from Wellesley College and an MFA from the University of Montana. The Company does not currently have terms for its directors, and Ms. Widger will serve as a director of the Company until the next annual meeting and until a successor is elected and qualified.
19
Ms. Xian Xuemei is a Director since December 11, 2006. Ms. Xian served as clinical pharmacist of Chengdu Spaceflight Hospital from 1997 to 2000. Since 2001, she has been working with Liang Fang Pharmacy Co., Ltd, and is responsible for medical quality inspections. Ms. Xian graduated from the School of Pharmacy, West China University of Medical Sciences with a bachelor’s degree in 1996 with excellent academic results. The Company does not currently have terms for its directors, and Ms. Xian will serve as a director of the Company until the next annual meeting and until a successor is elected and qualified.
Mrs. Song Zhenghong is a Director on the founding Lotus Board of Directors and the spouse of Dr. Liu Zhong Yi. Since 1991 and currently, Mrs. Song Zhenghong has been a teacher, assistant teacher and senior teacher at Yungang Second Middle School in Fengtai, Beijing.
Family Relationships
There are no family relationships among our executive officers and directors except for Liu Zhong Yi and Song Zhenghong who are husband and wife.
Involvement in Certain Legal Proceedings
None of the directors or executive officers has, during the past five years:
| (a) | Had any bankruptcy petition filed by or against any business of which such person was a general partner or executive officer either at the time of the bankruptcy or within two years prior to that time; |
| (b) | Been convicted in a criminal proceeding or subject to a pending criminal proceeding; |
| (c) | Been subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining, barring, suspending or otherwise limiting his involvement in any type of business, securities, futures, commodities or banking activities; and |
| (d) | Been found by a court of competent jurisdiction (in a civil action), the Securities and Exchange Commission or the Commodity Futures Trading Commission to have violated a federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated. |
Audit Committee Financial Expert
We are not a “listed company” under SEC rules and are therefore not required to have an audit committee comprised of independent directors. We do not currently have an audit committee, however, for certain purposes of the rules and regulations of the SEC, the Company’s board of directors is deemed to be its audit committee. The board of directors has determined that each of its members is able to read and understand fundamental financial statements and has substantial business experience that results in that member’s financial sophistication. However, none of our directors qualifies as an “audit committee financial expert” within the meaning of the rules and regulations of the SEC.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth, as of April 25, 2007, certain information regarding the ownership of the Company’s capital stock by the following persons on such date: each of the directors and executive officers, each person who is known to be a beneficial owner of more than 5% of any class of our voting stock, and all of our officers and directors as a group. Unless otherwise indicated below, to our knowledge, all persons listed below had sole voting and investing power with respect to their shares of capital stock, except to the extent authority was shared by spouses under applicable community property laws.
20
Beneficial ownership is determined in accordance with the rules of the Securities and Exchange Commission and generally includes voting or investment power with respect to securities. Shares of our common stock subject to options, warrants or convertible securities exercisable or convertible within 60 days of April 25, 2007 were deemed outstanding for computing the percentage of the person or entity holding such options, warrants or convertible securities but are not deemed outstanding for computing the percentage of any other person, and was based on 41,280,000 shares of the Common Stock issued and outstanding on a fully diluted basis, as of April 25, 2007.
Title of Class | Name and Address Of Beneficial Owners (1) | Amount and Nature Of Beneficial Ownership | Percent Of Class (2) | |||||||
| Common Stock | Dr. Liu Zhong Yi (5 | ) | 18,782,400 | 45.50 | % | |||||
| Common Stock | Mrs. Song Zhenghong (5 | ) | 6,708,000 | 16.25 | % | |||||
| Common Stock | Genesis Technology Group, Inc. | 6,736,896 | 16.32 | % | ||||||
| Common Stock | China West, LLC (3 | ) | 3,302,400 | 8.00 | % | |||||
| Common Stock | Mr. Shaohua Tan (4 | ) | 3,170,304 | 7.68 | % | |||||
| Common Stock | Mr. Adam Wasserman | 0 | 0 | % | ||||||
| Common Stock | Dr. Ian Ashley | 0 | 0 | % | ||||||
| Common Stock | Mr. Li Ping | 0 | 0 | % | ||||||
| Common Stock | Mr. Liu Jin | 0 | 0 | % | ||||||
| Common Stock | Mr. Mel Rothberg | 0 | 0 | % | ||||||
| Common Stock | Ms. Caeli Widger | 0 | 0 | % | ||||||
| Common Stock | Ms. Xian Xuemei | 0 | 0 | % | ||||||
| All officers and directors as a group (9 persons) | 25,490,400 | 61.75 | % | |||||||
| (1) | Unless otherwise noted, the address for each of the named beneficial owners is: 7900 Glades Road, Suite 420, Boca Raton, Florida 33434. |
| (2) | Percentage based upon 41,280,000 shares outstanding as of April 25, 2007. |
| (3) | Charles Tamburello is the manager of China West, LLC and exercises sole voting and investment control over such shares. |
| (4) | Mr. Shaohua Tan’s address is Suite 310, Building B, Ideal Plaza, No. 3, Danling Street, Haidian District, Beijing 100080, China. |
| (5) | Dr. Liu Zhong Yi and Mrs. Song Zhenghong are husband and wife. |
Change-in-Control Arrangements
On February 12, 2007, we entered into a financing agreement with two of the Selling Security-holders (the “Purchasers”) pursuant to which we issued Secured Convertible Notes (the “Notes”) with an aggregate principle value of $3,000,000, plus interest on the unpaid principal balance at a rate equal to 14.0% per annum.
The Notes are secured by a Security Agreement, a Pledge Agreement, a Guaranty, and Share Pledge Agreements (collectively, the “Security Agreements”). In the event of a default, the Security Agreements grant to S. Michael Rudolph, as collateral agent for the Purchasers, a security interest in all right, title and interest of Lotus, Lotus International and Lotus East to and in respect of all Accounts, Goods, real or personal property, all present and future books and records relating to the foregoing and all products and Proceeds of the foregoing, as set forth therein, and all security interests, as set forth therein, of Lotus, Lotus International and Lotus East. In addition, Liu Zhong Yi, the Company’s president and chief executive officer, pledged his 18,782,400 shares of the Company’s common stock, Song Zhenghong, a director of the Company, pledged his 6,708,000 shares of the Company’s common stock, and the Company pledged its 38,800 shares of Lotus International’s common stock in the event of a default under the Notes. Events of default include, but are not limited to, (i) the Company’s failure to pay any installment of principal, interest or other sum due under the Notes when due and such failure continues for a period of 5 business days after the due date, (ii) the commencement of a voluntary or involuntary liquidation, or other relief with respect to the Company or its debts under bankruptcy, insolvency or other similar law, that is not dismissed with 45 days of initiation (iii) final judgments for the payment in excess of $100,000 are rendered against the Company, and remain unpaid, unvacated, unbonded or unstayed for a period of 45 days (iv) or any breach or failure to comply in any respect with the terms of the Notes or any agreements entered into in connection with the transactions contemplated by the Agreements.
21
The Notes and Security Agreements are attached as exhibits to our Current Report on Form 8-K filed with the SEC on February 15, 2007.
DESCRIPTION OF SECURITIES
Our Company’s Articles of Incorporation, as amended, provide for authority to issue 200,000,000 shares of Common Stock with par value of $0.001 per Share. As of April 23, 2007, the capitalization of Lotus consists of 41,280,000 outstanding shares of Common Stock. The holders of the Common Stock are entitled to receive dividends when and as declared by the Board of Directors, out of funds legally available therefore. The Company has not paid cash dividends in the past and does not expect to pay any within the foreseeable future since any earnings are expected to be reinvested in the Company. In the event of liquidation, dissolution or winding up of the Company, either voluntarily or involuntarily, each outstanding share of the Common Stock is entitled to share equally in the Company's assets. Each outstanding share of the Common Stock is entitled to equal voting rights, consisting of one vote per share.
We have designated and authorized 10,000,000 Preferred Stock in our Articles of Incorporation, as amended. We have no Preferred Stock issued at this time, and there are no material rights attached to our Preferred Stock.
The validity of the common stock to be sold by the selling stockholders under this prospectus will be passed upon for us by Casale Alliance, LLP.
The financial statements of Lotus for the years ended December 31, 2006 and 2005 appearing in this prospectus have been audited by Sherb & Co., LLP, as set forth in their reports thereon appearing elsewhere herein, and are included in reliance upon such reports given upon the authority of such firms as experts in accounting and auditing.
DISCLOSURE OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Our bylaws provide that we will indemnify directors, officers, employees, and agents of the Company to the full extent permitted by law. We have been advised that in the opinion of the Securities and Exchange Commission indemnification for liabilities arising under the Securities Act of 1933 is against public policy as expressed in the Securities Act of 1933, and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities is asserted by one of our directors, officers, or controlling persons in connection with the securities being registered, we will, unless in the opinion of our legal counsel the matter has been settled by controlling precedent, submit the question of whether such indemnification is against public policy to a court of appropriate jurisdiction. We will then be governed by the court's decision.
BUSINESS
Unless otherwise indicated, all references to ‘‘we’’, ‘‘us’’, ‘‘our’’, the ‘‘Company’’, “Lotus”, and similar terms, as well as references to the ‘‘Registrant’’ in this Registration Statement on Form SB-2, refer to Lotus Pharmaceuticals, Inc. (including its subsidiaries).
Overview
We are a pharmaceutical company focused on developing, manufacturing and distributing drugs in the People's Republic of China (“China” or PRC”). Additionally, through our 10 retail pharmacy locations in Beijing, China, we sell Western and traditional Chinese medications as well as medical treatment equipment.
We originally became duly incorporated in the State of Nevada on January 28, 2004 under the name S.E. Asia Trading Company, Inc. (“SEAA”) to sell jewelry and home accessories. On September 6, 2006, we entered into a definitive Share Exchange Agreement with Lotus Pharmaceutical International, Inc. (“Lotus International”), a company incorporated under the laws of the State of Nevada on August 28, 2006, to develop and market pharmaceutical products in China. Pursuant to the Share Exchange Agreement, we acquired all of the outstanding common stock of Lotus International in exchange for issuing stock to Lotus International’s shareholders. On September 28, 2006, Lotus International became our wholly-owned subsidiary, and Lotus International’s shareholders became the majority owners of our voting stock. On December 6, 2006, we changed our name to Lotus Pharmaceuticals, Inc. (“Lotus”).
22
PRC law currently has limits on foreign ownership of certain companies. To comply with these foreign ownership restrictions, we operate our pharmaceutical business in PRC through Beijing Liang Fang Pharmaceutical Co., Ltd. (“Liang Fang”) and an affiliate of Liang Fang, Beijing En Zhe Jia Shi Pharmaceutical Co., Ltd. (“En Zhe Jia”), both of which are pharmaceutical companies headquartered in the PRC and organized under the laws of the PRC (hereinafter, referred to together as “Lotus East”). We entered into consulting service agreements and equity-related arrangements with Lotus East and its shareholders pursuant to which we provide technology consulting and other general business operation services to Lotus East. Through these contractual arrangements, we also have the ability to substantially influence Lotus East’s daily operations and financial affairs, appoint its senior executives and approve all matters requiring shareholder approval. As a result of these contractual arrangements, which enable us to control Lotus East, we are considered the primary beneficiary of Lotus East. Accordingly, we consolidate Lotus East’s results, assets and liabilities in our financial statements. Our headquarters are located in Boca Raton, Florida at Boca Corporate Plaza, 7900 Glades Road, Suite 420, Boca Raton, FL 33434, and our telephone number at that address is (877) 801-0344. We maintain a web site at www.lotuseast.com. Information on our web site is not a part of this Registration Statement.
Contractual Arrangements with Lotus East and Its Shareholders
On September 6, 2006, we entered into the following contractual arrangements:
Consulting Services Agreement. Pursuant to the exclusive consulting services agreements between Lotus International and Lotus East, Lotus International has the exclusive right to provide to Lotus East general pharmaceutical business operations services as well as consulting services related to the technological research and development of pharmaceutical products as well as general business operation advice and strategic planning (the “Services”). Under this agreement, Lotus International owns the intellectual property rights developed or discovered through research and development, in the course of providing the Services, or derived from the provision of the Services. Lotus East shall pay a quarterly consulting service fees in Renminbi (“RMB”) to Lotus that is equal to Lotus East’s revenue, as defined, for such quarter.
Operating Agreement. Pursuant to the operating agreement among Lotus International, Lotus East and the shareholders of Lotus East, (collectively “Lotus East’s Shareholders”), Lotus International provides guidance and instructions on Lotus East’s daily operations, financial management and employment issues. Lotus East’s Shareholders must designate the candidates recommended by Lotus International as their representatives on Lotus East’s Board of Directors. Lotus International has the right to appoint senior executives of Lotus East. In addition, Lotus International agreed to guarantee Lotus East’s performance under any agreements or arrangements relating to Lotus East’s business arrangements with any third party. Lotus East, in return, agreed to pledge its accounts receivable and all of its assets to Lotus International. Moreover, Lotus East agreed that without the prior consent of Lotus International, Lotus East would not engage in any transaction that could materially affect the assets, liabilities, rights or operations of Lotus East, including, without limitation, incurrence or assumption of any indebtedness, sale or purchase of any assets or rights, incurrence of any encumbrance on any of its assets or intellectual property rights in favor of a third party or transfer of any agreements relating to its business operation to any third party. The term of this agreement is ten (10) years from September 6, 2006 and may be extended only upon Lotus International’s written confirmation prior to the expiration of the this agreement, with the extended term to be mutually agreed upon by the parties.
Equity Pledge Agreement. Under the equity pledge agreement between the shareholders of Lotus East and Lotus International, the shareholders of Lotus East pledged all of their equity interests in Lotus East to Lotus International to guarantee Lotus East’s performance of its obligations under the technology consulting agreement. If Lotus East or Lotus East’s Shareholders breaches its respective contractual obligations, Lotus International, as pledgee, will be entitled to certain rights, including the right to sell the pledged equity interests. Lotus East’s Shareholders also agreed that upon occurrence of any event of default, Lotus International shall be granted an exclusive, irrevocable power of attorney to take actions in the place and stead of Lotus East’s Shareholders to carry out the security provisions of the equity pledge agreement and take any action and execute any instrument that Lotus International may deem necessary or advisable to accomplish the purposes of the equity pledge agreement. The shareholders of Lotus East agreed not to dispose of the pledged equity interests or take any actions that would prejudice Lotus International’s interest. The equity pledge agreement will expire two (2) years after Lotus East’s obligations under the exclusive consulting services agreements have been fulfilled.
Option Agreement. Under the option agreement between Lotus East’s Shareholders and Lotus International, Lotus East’s Shareholders irrevocably granted Lotus International or its designated person an exclusive option to purchase, to the extent permitted under PRC law, all or part of the equity interests in Lotus East for the cost of the initial contributions to the registered capital or the minimum amount of consideration permitted by applicable PRC law. Lotus International or its designated person has sole discretion to decide when to exercise the option, whether in part or in full. The term of this agreement is ten (10) years from September 6, 2006 and may be extended prior to its expiration by written agreement of the parties.
23
Proxy Agreement. Pursuant to the proxy agreement among Lotus International and Lotus East’s Shareholders, Lotus East’s Shareholders agreed to irrevocably grant a person to be designated by Lotus International with the right to exercise Lotus East’s Shareholders’ voting rights and their other rights, including the attendance at and the voting of Lotus East’s Shareholders’ shares at the shareholders’ meetings (or by written consent in lieu of such meetings) in accordance with applicable laws and its Article of Association, including but not limited to the rights to sell or transfer all or any of his equity interests of Lotus East, and appoint and vote for the directors and Chairman as the authorized representative of Lotus East’s Shareholders. The term of this Proxy Agreement is ten (10) years from September 6, 2006 and may be extended prior to its expiration by written agreement of the parties.
Lotus East
As discussed above, our operations are conducted through Liang Fang and its affiliated company, En Zhe Jia, both of which are limited liability companies headquartered in PRC and organized under the laws of PRC.
Based in Beijing, China, Liang Fang is engaged in the production, trade and retailing of pharmaceuticals. Further, Liang Fang is focused on the development of innovative medicines to address various medical needs for patients worldwide. Liang Fang owns and operates 10 drug stores throughout Beijing, China. These drug stores sell Western and traditional Chinese medications, medical equipment and accessories, and lease space. Liang Fang also leases part of its retail space to licensed physicians to administer medical treatment, to third party vendors and as advertising locations. Liang Fang has 3 shareholders: Wen Li Xian, Song Zhenghong, and Liu Zhong Yi.
En Zhe Jia is the sole manufacturer for Liang Fang and maintains facilities for the production of Western medicines, patented Chinese medicine, as well as the research and production of new medicines. En Zhe Jia has 2 shareholders: our CEO, Liu Zhong Yi and Song Zhenghong, Liu Zhong Yi’s spouse.
Business Description
Our business is composed of four parts: (1) Manufacturing and distribution of pharmaceutical products, including the manufacture of pharmaceutical products for other distributors (OEM manufacturing), (2) retailing of Western and traditional Chinese medications and accessories, (3) leasing retail space to licensed physicians and other parties and (4) research and development as outlined below:
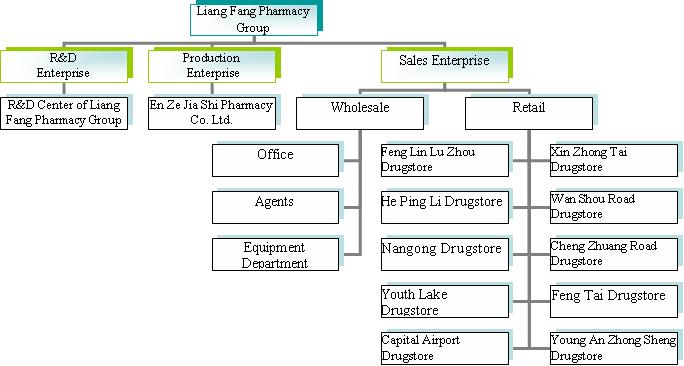
24
Manufacturing and distribution of pharmaceutical products
Our production enterprise is located in the Chaoyang District of Beijing and covers a floor space of approximately 50,000 square feet. We possess liquid phase, gas phase, spectrum, and mass spectrum equipment and all kinds of purification and distilling equipment, all of which can be used for production and research and development of biochemical medicines, Chinese traditional medicines, chemical compound medicines, antibiotics and other new medicines. As one of the first enterprises to be authenticated by the National China Good Manufacturing Practices (GMP), we have an advanced automatic pharmacy product line which is GMP authenticated under certificate numbers C0849, C0850, D1645, and G3452. Medicines produced by the Company include material medicine, troches, capsules, granule medicaments; freeze-dried powder for injections, injections of small capacities and eye drops. Different production control zones are used with separated air conditioning system according to different production and control needs.
Production of pharmaceuticals is Lotus’ largest and most profitable business and accounts for approximately 83% of our revenues. Under various manufacturing contracts, we provide manufacturing services for several drug companies in which they supply the raw materials and we manufacture their product to their specifications for a fee.
Our principal pharmaceutical manufactured products include:
Valsartan
In 2000, we obtained approval from the State Food and Drug Administration ("SFDA") of China to sell Valsartan as a raw material and as a capsule (Maixin). Among our best selling products, Valsartan is a drug that treats hypertension or high blood pressure. This product is globally recognized as the ideal anti high blood pressure medication by the medical industry due to its most stable and longest lasting treatment results and minimum side-effects.
Valsartan works by blocking a substance in the body that causes blood vessels to tighten. As a result, Valsartan relaxes blood vessels. This lowers blood pressure and increases the supply of blood and oxygen to the heart.
Our goal is to make Valsartan the number one prescribed brand in its class of high blood pressure medications in the PRC.
Through October 29, 2006, we held protective manufacturing rights to produce Valsartan. We estimate that there are currently 10 enterprises producing single regent dosage and fixed-dose combinations of Valsartan in China. The foreign pharmacy Novartis Pharma sells Valsartan under its brand name, Diovan. According to clinical verification, the two products, Maixin and Diovan, have the same clinical effects. We believe that the longer our product is on the market, our market share will increase year by year due to brand recognition and continued sales efforts. We believe that even if the Chinese government does not take any measures to protect these products, other enterprises still can not replicate our products before February 2009 because it takes at least 28 months to replicate any orally taken medicine. By then, even if the market has fiercer competition, our market will be relatively mature and we believe that our production cost will be lower than those new products. Accordingly, we believe that our sales revenue from Valsartan can stay relatively stable.
Brimonidine Tartrate Eyes Drops
Brimonidine Tartrate is a drug used to constrict adrenaline receptors, an important step in treating glaucoma. We sell Brimonidine Tartrate eye drops under our brand name “Muxin”. The drug was first put into market in the U.S. in 1998 and in August 2004, we received the rights to manufacture and release the drug in the Chinese market. A fast and obvious curative effect, very few side effects, a very high exponent of cure and high endurement are prime features of the drug. It will produce no harmful effects of reducing blood pressure, resulting in calmness and so on, much like diazepam. The imported product of its kind is Alphagan and is produced by Allergan (Hangzhou) Pharmaceutical Co., Ltd. According to clinical experiments of People's Hospital of Peking University and Tianjin Eye Hospital, our Muxin and imported Alphagan have exactly the same curative effect but the side-effects of Muxin are fewer than Alphagan, so our product has a stronger competitive advantage.
To date, we have done minimal advertising on this product, with sales increasing only by the customer’s natural needs. We hold protective manufacturing rights to produce Brimonidine Tartrate through September 2007. We estimate that other enterprises will not be able to replicate this product until March 2010, because we believe it takes at least 30 months to replicate an eye drop medicine. Currently, we have minimal competition and we believe our market share will not decrease. However, we anticipate lowers profits after 2010.
25
Leflunomide
Leflunomide is a medicine for the treatment of rheumatoid arthritis, systematically erythematic lupus, and psoriasis. It also has functions such as anti-rejection.
Levofloxacin Lactate
Levofloxacin is a popular anti-bacterial drug with indications of all kinds of bacterial inflections and used for medical care.
Nicergoline for Injection
Nicergoline for Injection is a national medical insurance product. It is an a-receptor blockage nerve system blood-brain medicine with remarkable curative effect.
With the implementation in China of macro economic policy, the profit margin of medicines shall decrease. As of the date of this report, we can not estimate the effect of these economic policies on our results of operations.
Currently, we market and sell the products only in PRC.
Retail Drugstores
Lotus owns and operates 10 drug stores throughout many different districts of Beijing, including:
| · | Xin Zhong Tai Drugstore | |
| · | Nangong Drugstore | |
| · | Wan Shou Road Drugstore | |
| · | He Ping Li Drugstore |
| · | Feng Lin Lu Zhou | |
| · | Youth Lake Drugstore | |
| · | Capital Airport Drugstore | |
| · | Young An Zhong Sheng Drugstore |
| · | Cheng Zhuang Road Drugstore | |
| · | Feng Tai Drugstore |
These 10 drugstores all deal in over 5,000 types of Western and traditional Chinese medications, and medical treatment equipment. We have over 200 employees including registered pharmacists. The drugstores attempt to compete according to lower pricing and more efficient distribution and management practices. All of the drugstores are located in Beijing, China.
Through our retail operations, we also obtain revenue through the leasing of office space to licensed physicians, the lease of store front areas to various merchants, and the lease of advertising space in the retail pharmacies.
Research and Development Enterprise
We have a research and development ("R&D") team focused on discovering new drugs as well as developing generic and improved drugs based on existing products already on the market and traditional Chinese medicinal products. Our R&D team consists of experts in the fields of medical technology, biotechnology, and pharmaceuticals.
Distribution Methods of the Products or Services and our Customers
Many of our drug products are sold through our retail drug stores. In addition, we sell our drugs through drugs distribution companies in China covering Beijing, Shanghai, GuangDong, Inner Mongolia, Ningxia, Henan, Hubei, LiaoNing, HeiLongjiang, Guangxi, Jiangsu, Hebei, Anhui provinces in the PRC. Currently, we have approximately 69 distribution agents throughout the PRC.
26
We work with various distribution companies to distribute our products. The demand for new drugs in China is substantial as the drug distribution companies suffer from very low profit margins from the distribution of old generic drugs. Additionally, we have a network of connections with hospitals in the Beijing and surrounding areas. We have established contacts that provide direct access to hospitals in these areas.
We will continue to establish more representative offices and engage additional distribution agents in order to strengthen our distribution network.
We recognize the importance of branding as well as packaging. All of our products bear a uniform brand but we also brand and package our products with specialized designs to differentiate the various categories of our products.
We conduct promotional marketing activities to publicize and enhance our image as well as to reinforce the recognition of the our brand name, including:
| 1. | publishing advertisements and articles in national as well as specialized and provincial newspapers, magazines, and in other media, including the Internet; |
| 2. | participation in national meetings, seminars, symposiums, exhibitions for bio-pharmaceutical and other related industries; |
| 3. | organizing cooperative promotional activities with distributors; and |
| 4. | sending direct mail to major physician offices and laboratories. |
We currently have over 100 customers, including over 30 major direct customers in Beijing, Shanghai, GuangDong, Inner Mongolia, Ningxia, Henan, Hubei, LiaoNing, HeiLongjiang, Guangxi, Jiangsu, Hebei, Anhui provinces in the PRC.
Competition
Vertically integrated pharmaceutical operations are still at an early stage of development in China due to heavy state involvement in the past. The industry is fragmented. We face competition from domestic drug R&D and manufacturing companies, which are growing rapidly. Our direct competitors are Beijing Venture Biopharma Technology Co., Ltd., Fosun Group Co., Ltd., Chongqing Pharmaceutical Research Institute, Co. Ltd., Zhuhai Lizhu and Beijing Nohua We also face competition from foreign companies that have strong proprietary pipelines and strong financial resources. These companies have significantly greater assets than we do and have a larger current market share. Our advantage is our local concentration in research and development as well as our local distribution network, along with lower prices. Our focus is on quality and establishing long-term cooperative relations with the Chinese Academy of Medical Sciences and the China Academy of Traditional Chinese Medicine, as well as medicine test organizations. We are in a competitive position to seize substantial market opportunities as the pharmaceutical industry in China rapidly moves toward consolidation, privatization, and commercialization.
Sources and Availability of Raw Materials and the Principal Suppliers
Our principal raw materials include Brimonidine Tartrate, Brimonidine Tartrate and Valsartan. Currently, we purchase our raw materials used to manufacture our products from a variety of suppliers of which approximately 95% of our raw materials are provided by three suppliers. These suppliers account for 60%, 20% and 15%, respectively, of our total raw material purchases. Management is aware of similar raw materials supplies that would be available from other sources, if required, and has the ability to immediately engage in purchasing transactions with such sources, if and as necessary.
We design, create prototypes and manufacture our products at our manufacturing facilities located at Beijing, PRC. The prices for these raw materials are subject to market forces largely beyond our control, including energy costs, organic chemical prices, market demand, and freight costs. The prices for these raw materials have varied significantly in the past and may vary significantly in the future.
Patents, Trademarks, Licenses, Franchises, Concessions. Royalty Agreements or Labor Contracts
We rely on a combination of trademark, copyright and trade secret protection laws in PRC and other jurisdictions, as well as confidentiality procedures and contractual provisions to protect our intellectual property and our brand. We have an issued patented special technology in PRC, valid for 10 years, and we intend to apply for more patents to protect our core technologies. We also enter into confidentiality, non-compete and invention assignment agreements with our employees and consultants and nondisclosure agreements with third parties. “Maixing” and “Liang Fang” are our registered trademarks in the PRC.
27
Bio-pharmaceutical companies are at times involved in litigation based on allegations of infringement or other violations of intellectual property rights. Furthermore, the application of laws governing intellectual property rights in the PRC and abroad is uncertain and evolving and could involve substantial risks to us.
Government Approval and Regulation of the Company’s Principal Products or Services
Our principal sales market is presently in China. We are subject to the Drug Administration Law of China, which governs the licensing, manufacturing, marketing and distribution of pharmaceutical products in China and sets penalties for violations of the law. Additionally, we are also subject to various regulations and permit systems by the Chinese government.
The State Food & Drug Administration of the PRC regulates and implements our drug laws. The State FDA has granted the Company six government permits for us to produce the following products: Valsartan Capsules, the material of Valsartan, Levofloxacin Lactate for Injection, the material of Levofloxacin Lactate, Brimonidine Tartrate Eyes Drops with a density of 0.1, Brimonidine Tartrate Eyes Drops with the density of 0.3.
The approval process takes about two years, including local SFDA approval, Local SFDA test, State SFDA processing, state SFDA expert valuation, clinical trial, and final approval.
No enterprise may start production at its facilities until it receives approval from the Ministry of Agriculture to begin operations. The Company currently has obtained the requisite approval and licenses from the Ministry of Agriculture in order to operate our production facilities.
Research and Development
We place great emphasis on product research and development. We have two research and development centers. We have a product pipeline containing approximately seven products in different dosage forms which are ready for commercialization in China for the treatment of diseases. We intend to commercialize or license these drugs during 2007 and 2008. Major projects currently being undertaken at these centers focus on the following:
| Drug Name | Target Treatment | Status | ||
| Calcium Dibutyryl Adenosine Monophosphate for Injection | used to cure angina and acute myocardial infarction | (A) Expected approval date February 2008 | ||
| Rabeprazole Sodium raw material and Rabeprazole Sodium Enteric-coated Tablets | used for curing gastric ulcer, duodenal ulcer, stomal ulcer, reflux esophagitis, Zollinger-Ellison symptom complex and gastrinoma | (A) Expected approval date August 2007 | ||
| Compound Allantoin Dispersible Tablets | used for curing gastrelcosis, duodenal bulbar ulcer and chronic gastritis | (A) Expected approval date April 2008 | ||
| Gatifloxacin Lactate for Injection | used for curing acute nasosinusitis, chronic bronchitis, pneumonia, gonorrhea, and rectum infection | (A) Expected approval date September 2007 | ||
| Sodium Aescinate for Injection | use for curing hydrocephalus, swelling caused by wounds or surgery operation and for venous return disorder. | (A) Expected approval date September 2007 | ||
| Candesartan Pills | used for curing essential hypertension | (A) Expected approval date March 2008 | ||
| Nicergoline Pills | used for the curing feeling dullness, impaired concentration, memory deterioration, gloom and restlessness caused by cerebral infarction sequela | (A) Expected approval date May 2008 |
(A) Subject to SFDA. Pending Administrative Protection and approval.
28
In 2004, we spent approximately $1.15 million or approximately 9% of the Company’s 2004 revenue on research and development of various bio-pharmaceutical products. In 2005, we spent approximately $1.76 million or approximately 10% of the Company’s 2005 revenue on research and development of products. In 2006, research and development costs were incurred for the improvement of technology on existing drugs and to improve our production process for which we incurred research and development expenses of approximately $387,000 or approximately 1% of the Company’s 2006 revenue. We expect to begin new research and development projects in the late part of 2007.
Compliance with Environmental Law
We comply with the Environmental Protection Law of China and its local regulations. In addition to statutory and regulatory compliance, we actively ensure the environmental sustainability of our operations.
To comply with PRC environmental regulations, the Company spent approximately $50,000 in 2004, $50,000 in 2005, and $150,000 in 2006, mainly for waste discharge processing and dust cleaning.
Employees
In 2004, the Company had 218 employees, of which 155 worked as full time employees. In 2005, the Company had 230 employees, of which 193 worked as full time employees. In 2006, the Company had 266 employees, of which 215 worked as full time employees. Currently, the Company has 268 employees, 215 of which are full time employees. Additionally, we employ our chief financial officer on an outsourced basis.
MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
Overview
The following discussion of the financial condition and results of operation of Lotus Pharmaceuticals, Inc. (“Lotus”) should be read in conjunction with the financial statements and the notes to those statements included herein. This discussion includes forward-looking statements that involve risk and uncertainties. As a result of many factors, such as those set forth under “Risk Factors”, actual results may differ materially from those anticipated in the forward-looking statements.
Lotus’ revenues and results of operations could differ materially from those projected in the forward-looking statements as a result of numerous factors, including, but not limited to, the following: the risk of significant natural disaster, the inability of Lotus to insure against certain risks, inflationary and deflationary conditions and cycles, currency exchange rates, changing government regulations domestically and internationally affecting the pharmaceutical industry, competition and pricing, changes in external competitive market factors, termination of certain agreements, protocol, or inability to enter into strategic agreements, inability to satisfy anticipated working capital or other cash shortage requirements, changes in or developments under domestic or foreign laws, regulations, or changes in Lotus’ business strategy or an inability to execute its strategy due to unanticipated changes in the market. In light of these risks and uncertainties, there can be no assurance that actual results, performance or achievements of Lotus will not differ materially from any future results, performance or achievements expressed or implied by such forward-looking statements.
For purposes of the following discussion and analysis, references to ‘‘we’’, ‘‘our’’, ‘‘us’’ refers to Lotus.
On September 6, 2006, we entered into a definitive Share Exchange Agreement with Lotus Pharmaceutical International, Inc. (“Lotus International”), whereby we acquired all of the outstanding common stock of Lotus International in exchange for newly-issued shares of our common stock to Lotus International’s shareholders. Lotus International was incorporated under the laws the State of Nevada on August 28, 2006 to develop and market pharmaceutical products in the People's Republic of China (“PRC” or “China”). On September 28, 2006 (the closing date), Lotus International became our wholly-owned subsidiary and Lotus International’s shareholders became owners of a majority of our voting stock. The acquisition of Lotus International by us is accounted for as a reverse merger because on a post-merger basis, the former shareholders of Lotus International held a majority of our outstanding common stock on a voting and fully-diluted basis. As a result, Lotus International is deemed to be the acquirer for accounting purposes.
29
Additionally, on September 6, 2006, Lotus International entered into various agreements with Liang Fang Pharmaceuticals Co. Ltd. (“Liang Fang”) and Beijing En Zhe Jia Pharmaceutical Co., Ltd. (“En Zhe Jia”) (herein referred to as “Lotus East”) whereby the shareholders of Lotus East transferred all operating and voting control of Lotus East to Lotus International. As a result of these agreements, Lotus East was deemed to be the acquirer of Lotus International for accounting purposes. Accordingly, the financial statement data presented are those of Lotus East for all periods prior to our acquisition of Lotus International on September 28, 2006, and the financial statements of the consolidated companies from the acquisition date forward.
Liang Fang is a Chinese limited liability company and was formed under laws of the People’s Republic of China on June 21, 2000 and is engaged in the production, trade and retailing of pharmaceuticals. Further, Liang Fang is focused on development of innovative medicines and investing strategic growth to address various medical needs for patients worldwide. Liang Fang’s operations are based in Beijing, China.
Liang Fang owns and operates several drug stores throughout Beijing, China. These drugstores sell Western and traditional Chinese medicine, and medical treatment equipment.
Liang Fang’s affiliate, En Zhe Jia, is a Chinese limited liability company and was formed under the laws of the People’s Republic of China on September 17, 1999. En Zhe Jia is the sole manufacturer of pharmaceuticals for the Company and maintains facilities for the production of Western medicines, Chinese medicines, as well as the research and production of other new medicines.
Current Business Operations
As discussed above, our operations are conducted through Liang Fang and its affiliated company, En Zhe Jia, both of which are limited liability companies headquartered in PRC and organized under the laws of PRC. Our business is composed of four parts: (1) Manufacturing and distribution of pharmaceutical products, including the manufacture of pharmaceutical products for other distributors (OEM manufacturing), (2) retailing of Western and traditional Chinese medications and accessories, (3) leasing retail space to licensed physicians and other parties and (4) research and development.
The Company currently has 268 employees, but we do not have any employees at our Boca Raton, Florida headquarters offices located at Boca Corporate Plaza, 7900 Glades Road, Suite 420, Boca Raton, Florida 33434, except our Chief Financial Officer who is hired on an outsourced basis.
Results of Operations
For the Year Ended December 31, 2006 Versus Year Ended December 31, 2005
Total Revenues
Total revenues for the year ended December 31, 2006 were $36,207,689 as compared to total revenues of $18,345,841 for the year ended December 31, 2005, an increase of $17,861,848 or approximately 97%. For the year ended December 31, 2006 and 2005, net revenues consisted of the following:
| 2006 | 2005 | ||||||
| Wholesale | $ | 25,430,069 | $ | 16,099,036 | |||
| Retail | 3,532,623 | 1,629,385 | |||||
| Other revenues | 7,244,997 | 617,420 | |||||
| Total Revenues | $ | 36,207,689 | $ | 18,345,841 | |||
| · | For the year ended December 31, 2006, wholesale revenues increased by $9,331,033 or 58%. The significant increase in tangible product revenues is mainly attributed to continued strong sales of our best selling product, VALSARTAN Capsules, a medicine for primary hypertension or high blood pressure. |
| · | For the year ended December 31, 2006, retail revenues increased by $1,903,238 or 116.8%. In 2006, due to the gradual implementation of local medication policy reforms, Beijing started to change the way the patient may obtain medication and separated the treatment of the patient from the obtaining of the medicine. Before 2005, most civil servants could only get treatment and buy medicines in the hospital, and the expenses could not get reimbursed if they bought medicines from pharmacy stores. Since 2005, patients from many companies and organizations have been allowed to purchase medicines from approved pharmacy stores after they get prescriptions in the hospital, and receive expense reimbursement. The convenience, better service and lower prices encourages patients to purchase medicines at pharmacy stores. In March 2006, the Beijing government started approving medicine expense reimbursements based on receipts from our retail pharmacy stores. As a result, our retail revenues increased substantially. |
30
| · | For the year ended December 31, 2006, other revenues increased by $6,627,577. The significant increase in other revenues is attributed to the following: |
| 2006 | 2005 | ||||||
| Leasing revenues | $ | 696,637 | $ | 617,420 | |||
| Third-party manufacturing | 4,855,402 | - | |||||
| Advertising revenues | 571,293 | - | |||||
| Research and development and lab testing services | 1,121,665 | - | |||||
| Total other revenues | $ | 7,244,997 | $ | 617,420 | |||
| · | During 2006, we began performing third party contract manufacturing projects. In connection with third-party manufacturing, the customer supplies the raw materials and we are paid a fee for manufacturing their product. |
| · | In 2006, we entered into an advertisement contract whereby we receive approximately $50,000 per month for the lease of counter and other space at our retail locations. We expect this revenue to increase in the future as we sign similar contracts. |
| · | In 2006, we performed research and development and lab testing projects for various third parties and performed drug testing and analysis. |
Cost of Sales
Cost of sales includes raw materials, packing materials, shipping, and manufacturing costs, which includes allocated portion of overhead expenses such as utilities and depreciation directly related to product production. For the year ended December 31, 2006, cost of sales amounted to $26,523,039 or approximately 73.3% of total revenues as compared to cost of sales of $14,584,682 or approximately 79.5% of total revenues for the year ended December 31, 2005. The decrease in cost of sales as a percentage of total revenues for the year ended December 31, 2006 as compared to the year ended December 31, 2005 is attributable to increases in other service oriented revenues from third-party manufacturing, advertising revenues and research and development services and the increase in leasing revenues offset by a reduction in drug prices due to governmental pricing policies.
Gross Profit
Gross profit for the year ended December 31, 2006 was $9,684,650 or 26.7% of total revenues, as compared to $3,761,159 or 20.5% of revenues for the year ended December 31, 2005. The increases in gross profits is attributable to increases in other revenues from third-party manufacturing, advertising revenues and research and development services and the increase in leasing revenues offset by a reduction in drug prices due to governmental pricing policies. Although we recognized higher than average gross profits during the year ended December 31, 2006, there could be no assurances that we will continue to recognize similar gross profit margin in the future.
Operating Expenses
Total operating expenses for the year ended December 31, 2006 were $5,258,089, an increase of $2,108,969 or 67.0%, from total operating expenses in the year ended December 31, 2005 of $3,149,120. This increase included the following:
For the year ended December 31, 2006, selling expenses amounted to $2,154,216 as compared to $414,361 for the year ended December 31, 2005, an increase of $1,739,855 or 420%. This increase is attributable to increase in commission paid of $1,045,448 in 2006 compared to $0 in 2005, an increase in advertising costs of approximately $82,000, and an increase in other selling expenses of $612,407 such as travel and entertainment, and sales conferences and training, We expect our selling expenses to increase as our revenues increase and expect to spend increased funds on adverting and promotion of our products as well as sales training. During fiscal 2007, we intend to expand our marketing efforts related to our products.
31
For the year ended December 31, 2006, research and development costs amounted to $387,337 as compared to $1,759,756 for the year ended December 31, 2005, a decrease of $1,372,419 or 78%. In fiscal 2006, we decreased our research and development activities to concentrate on the sale and marketing of developed products. We currently have 4 drugs in the pipeline that are in the process of obtaining government approval. As of the date of this filing, we can not predict when we will begin the development of new pharmaceutical products or the related costs.
For the year ended December 31, 2006, general and administrative expenses were $2,716,536 as compared to $975,003 for the year ended December 31, 2005, an increase of $1,741,533, or 179.0% as summarized below:
| Year ended December 31, | |||||||
| 2006 | 2005 | ||||||
| Salaries and related benefits | $ | 1,012,438 | $ | 342,150 | |||
| Amortization | 169,517 | 178,577 | |||||
| Bad debt | 482,853 | 26,865 | |||||
| Depreciation | 22,102 | 19,958 | |||||
| Rent | 206,965 | 238,610 | |||||
| Travel and entertainment | 164,804 | 13,172 | |||||
| Professional fees | 105,081 | - | |||||
| Other | 552,776 | 155,671 | |||||
| Total | $ | 2,716,536 | $ | 975,003 | |||
The changes in these expenses from the year ended December 31, 2006 as compared to the year ended December 31, 2005 included the following:
| · | Salaries and related benefits increased $670,288 or 196% due the increase in hours worked and overtime and the increased use of our part time employees. | |
| · | Amortization of our manufacturing rights decreased by $9,060 or approximately 5%. |
| · | An increase of $455,988 or 1,697%, in bad debt expenses due to an increase in our sales and accounts receivable balances during the year ended December 31, 2006. | |
| · | Depreciation increased by $2,144 or approximately 10.7%. |
| · | Rent decreased by $31,645 or approximately 13.3%. | |
| · | Travel and entertainment expenses increased by $151,632 or 1,151% due to increased sales-related travel. | |
| · | Professional fees increased $105,081 or 100% due to an increase in accounting and legal fees activities related to being a public entity in the United States. |
| · | Other selling, general and administrative expenses, which includes utilities, office supplies and expenses increased by $397,105 or 255.1%. |
Income from Operations
We reported income from operations of $4,426,561 for the year ended December 31, 2006 as compared to income from operations of $612,039 for the year ended December 31, 2005, an increase of $3,814,522 or approximately 623.2%.
Other Income (Expense)
For the year ended December 31, 2006, total other expense amounted to $(325,824) as compared to other income of $1,219,673 for the year ended December 31, 2005, a decrease of $1,545,497. This change is primarily attributable to:
| · | The sale of patent to a third party for a drug formula that we had developed in 2005 for approximately $1.2 million for the year ended December 31, 2005. |
| · | Interest expense was $325,824 as compared to $0 for the year ended December 31, 2005, an increase of $325,824 due to our notes payable to related parties issued on December 31, 2005. |
Net Income
As a result of these factors, we reported net income of $4,100,737 for the year ended December 31, 2006 as compared to net income of $1,831,712 for the year ended December 31, 2005. This translates to net income per common share of $0.10 and $.05 for the years ended December 31, 2006 and 2005, respectively.
32
Liquidity and Capital Resources
Liquidity is the ability of a company to generate funds to support its current and future operations, satisfy its obligations and otherwise operate on an ongoing basis.
At December 31, 2006, we had a cash balance of $2,089,156. Substantially all of the Company’s cash is deposited in financial institutions in China.
Our working capital position increased $2,694,840 to $8,646,076 at December 31, 2006 from $5,951,236 at December 31, 2005. This increase in working capital is primarily attributable to an increase of approximately $6.3 million in accounts receivable due from third parties offset by an increase in taxes payable. The increase in accounts receivable and corresponding decrease inventories reflects the effects of increased sales during fiscal 2006 and the corresponding receivables generated by those sales.
At December 31, 2006, our inventories of raw materials and finished goods totaled $3,133,677, a decrease of approximately $3.8 million, or 120.9%, from December 31, 2005. During fiscal 2005 we introduced several new products which had been in development for a period of time; these new products were partially attributable for our increased sales in fiscal 2006. We expect to maintain higher inventory levels to accommodate for anticipated future sales growth as well as a wider variety of products.
At December 31, 2006, we maintain an allowance for doubtful accounts on accounts receivable balances of $539,627 as compared to $45,205 at December 31, 2005, an increase of $494,422 and reflects our best estimate of probable losses. In determining the allowance for doubtful accounts, our management reviews our accounts receivable aging as well as the facts and circumstances of specific customers which may indicate the collection of specific amounts are at risk. As is customary in the PRC, we extend relatively long payment terms to our customers. Our terms of sale generally require payment within four months to a year, which is considerably longer than customary terms offered in the United States, however, we believe that our terms of sale are customary amongst our competitors for a company our size within our industry. We also occasionally offer established customers longer payment terms on new products as an incentive to purchase these products, which has served to further increase the average days outstanding for accounts receivable. As the market for these new products is established, we will discontinue offering this sales incentive. Occasionally we will request a customer to prepay an order prior to shipment. At December 31, 2006 our balance sheet reflected advances from customers of $231,340, an increase of $46,142, or approximately 25%, from December 31, 2005.
Our balance sheet at December 31, 2006 also reflects a balance due to related parties of $695,231 which was a working capital advances made to us by our President, vice-president and an officer of the Company and a Board member. These loans are non-interest bearing and are due on demand. We are currently repaying these balances as operating cash become available.
Our balance sheet at December 31, 2006 also reflects notes payable to related parties of approximately $4.7 million due on December 30, 2015 which is a working capital loan made to us by the Company’s Chief Executive Officer, two employees of the Company and a Board member. These loans bear a variable annual interest at 80% of current bank rate and are unsecured. During fiscal 2006, we repaid approximately $3.3 million of these loan balances.
Net cash provided by (used in) operating activities for the year ended December 31, 2006 was $4,778,264 as compared to ($343,280) for the year ended December 31, 2005. For the year ended December 31, 2006, net cash provided by operating activities was attributable primarily to our net income of $4,100,737, the add back of depreciation and amortization of $567,469, a decrease in inventory balance of $3,928,236, an increase in taxes payable of $1,999,675, and an increase in other liabilities of $446,660 offset by a net increase in accounts receivable of $6,189,358 (net of increases in allowance for doubtful accounts and sales returns of $2,732,889) and prepaid expenses of $75,155. For the year ended December 31, 2005, net cash used in operating activities was attributable primarily to our net income of $1,831,712, a decrease in prepaid and other current assets of $622,007, an increase accounts payable and accrued expense of $137,331, and the add back of non-cash items such as depreciation and amortization of $429,633, bad debt of $26,865, and changes in other assets and liabilities and non-cash items of $182,188, offset by cash used to fund a net increase in accounts receivable of $597,584 and an increase in our inventory of $2,975,432.
33
Net cash provided by investing activities for the year ended December 31, 2006 was $1,242,262 attributable to payment received on due from related parties. Net cash used in investing activities for the year ended December 31, 2005 amounted to $4,208,338 which consisted of $2,163,128 attributable to advances made to an affiliated company owned by one of our officers and purchases of equipment of $2,045,210.
Net cash used in financing activities was $4,137,109 for the year ended December 31, 2006 and was attributable to payments on related party advances and loans as compared to net cash provided by financing activities of $4,624,531 for the year ended December 31, 2005 and reflects proceeds received from related party advances.
We reported a net increase in cash for the year ended December 31, 2006 of $1,928,222 as compared to a net increase in cash of $76,277 for the year ended December 31, 2005.
We currently have no material commitments for capital expenditures. Other than working capital and loans from related parties, and from our recent capital raising transaction as discussed below, we presently have no other alternative source of working capital. We may not have sufficient working capital to fund the expansion of our production line, an upgrade of our manufacturing facilities and technologies, expand our retail operations, and fund research and development projects as well as providing working capital necessary for our ongoing operations and obligations. We will need to raise additional working capital to complete these projects. We may seek to raise additional capital through the sale of equity securities. No assurances can be given that we will be successful in obtaining additional capital, or that such capital will be available in terms acceptable to our company. At this time, we have no commitments or plans to obtain additional capital.
Recent Capital Raising Transactions
On February 12, 2007, we entered into Subscription Agreements with two of the Selling Security-holders (the "Purchasers"). Pursuant to the Agreements, we issued to the Purchasers Secured Convertible Notes, ("Notes"), with an aggregate principle value of $3,000,000, plus interest on the unpaid principal balance at a rate equal to 14.0% per annum. The maturity date of the Notes is February 12, 2008. Interest on the Notes commences accruing on February 12, 2007 and is payable on April 30, 2007 and on the last business day of each calendar quarter thereafter. Interest is payable in cash, except that upon notice to the Purchasers in writing not less than 15 trading days prior to the relevant interest payment date, we may deliver common stock that were registered exclusively for the purpose of satisfying the payment of interest and not principal (“Interest Shares”) valued at 75% of the average of the three lowest closing bid prices of the common stock as report by Bloomberg L.P. for the 20 trading days ending on the trading day preceding the relevant interest payment date. In the event of default under the terms of the Notes, at the option of the Purchaser, all sums of principal and interest remaining unpaid will become immediately due and payable. The notes are convertible into shares of common stock of the Company a rate equal to $1.00 per share, subject to adjustment. The transaction closed on February 12, 2007.
The Purchasers also received Common Stock Purchase Warrants (the "Warrants"). Pursuant to the terms of the Warrants, the Purchasers are entitled to purchase up to 1,500,000 shares of our common stock at an exercise price of $1.50 per share. The Warrants have a term of 5 years after the issue date of February 12, 2007.
The Notes are secured by a Security Agreement, a Pledge Agreement, a Guaranty, and Share Pledge Agreements (collectively, the “Security Agreements”). In the event of a default, the Security Agreements grant to S. Michael Rudolph, as collateral agent for the Purchasers, a security interest in all right, title and interest of Debtors, as that term is defined therein, to and in respect of all Accounts, Goods, real or personal property, all present and future books and records relating to the foregoing and all products and Proceeds of the foregoing, as set forth therein, and all security interests, as set forth therein, of the Company, Lotus Pharmaceutical International, Inc. Beijing Liang Fang Pharmaceutical Co., Ltd. and Beijing En Ze Jia Shi Pharmaceutical Co., Ltd. In addition, Liu Zhong Yi, the Company’s president and chief executive officer, pledged his 18,782,400 shares of the Company’s common stock, Song Zhenghong, a director of the Company and spouse of Liu Zhong Yi, pledged her 6,708,000 shares of the Company’s common stock, and the Company pledged its 38,800 shares of Lotus’s common stock in the event of a default under the Notes. Events of default include, but are not limited to, (i) the Company’s failure to pay any installment of principal, interest or other sum due under the Notes when due and such failure continues for a period of 5 business days after the due date, (ii) the commencement of a voluntary or involuntary liquidation, or other relief with respect to the Company or its debts under bankruptcy, insolvency or other similar law, that is not dismissed with 45 days of initiation (iii) final judgments for the payment in excess of $100,000 are rendered against the Company, and remain unpaid, unvacated, unbonded or unstayed for a period of 45 days (iv) or any breach or failure to comply in any respect with the terms of the Notes or any agreements entered into in connection with the transactions contemplated by the Agreements.
34
The financing was completed through a private placement to accredited investors and is exempt from registration pursuant to Section 4(2) of the Securities Act of 1933, as amended ("Securities Act"). The securities sold in the financing have not yet been registered under the Securities Act of 1933, as amended, and may not be offered or sold in the United States in the absence of an effective registration statement or exemption from registration requirements.
We granted registration rights to holders of the Securities. Generally, the Company shall file with the Commission a Form SB-2 registration statement (the “Registration Statement”) (or such other form that it is eligible to use) in order to register the Registrable Securities for resale and distribution under the 1933 Act within forty-five (45) calendar days after the Closing Date (the “Filing Date”), and cause the Registration Statement to be declared effective not later than one hundred and fifty (150) calendar days after the Closing Date (the “Effective Date”). If the Registration Statement is not filed on or before the Filing Date or is not declared effective on or before the Effective Date, then we shall deliver to the holder of Registrable Securities, as Liquidating Damages, an amount equal to two percent (2%) for the first day of such Non-Registration Event and two percent (2%) for each thirty (30) days (or such lesser pro-rata amount for any period of less than thirty (30) days) of the Purchase Price of the outstanding Notes. The Company must pay the Liquidated Damages in cash. The Liquidated Damages must be paid within ten (10) days after the end of each thirty (30) day period or shorter part thereof for which Liquidated Damages are payable.
Critical Accounting Policies and Estimates
Our discussion and analysis of our financial condition and results of operations are based on our financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States of America. The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities and expenses. On an ongoing basis, we evaluate our estimates and judgments, including those related to accrued expenses, fair valuation of stock related to stock-based compensation and income taxes. We based our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
Revenue Recognition
Product sales. Product sales are generally recognized when title to the product has transferred to customers in accordance with the terms of the sale. The Company recognizes revenue in accordance with the Securities and Exchange Commission’s (SEC) Staff Accounting Bulletin (SAB) No. 101, “Revenue Recognition in Financial Statements” as amended by SAB No. 104 (together, “SAB 104”), and Statement of Financial Accounting Standards (SFAS) No. 48 “Revenue Recognition When Right of Return Exists.” SAB 104 states that revenue should not be recognized until it is realized or realizable and earned. In general, the Company records revenue when persuasive evidence of an arrangement exists, services have been rendered or product delivery has occurred, the sales price to the customer is fixed or determinable, and collectibility is reasonably assured.
SFAS No. 48 states that revenue from sales transactions where the buyer has the right to return the product shall be recognized at the time of sale only if the seller’s price to the buyer is substantially fixed or determinable at the date of sale, the buyer has paid the seller, or the buyer is obligated to pay the seller and the obligation is not contingent on resale of the product, the buyer’s obligation to the seller would not be changed in the event of theft or physical destruction or damage of the product, the buyer acquiring the product for resale has economic substance apart from that provided by the seller, the seller does not have significant obligations for future performance to directly bring about resale of the product by the buyer, and the amount of future returns can be reasonably estimated.
We recognize revenue for the sale of pharmaceutical products and for payments received, if any, under reimbursement of development costs as follows:
Product Sales. Revenue from product sales, net of estimated provisions, is recognized when there is persuasive evidence that an arrangement exists, delivery has occurred, the selling price is fixed or determinable, and collectibility is reasonably probable. Our customers consist of pharmaceutical wholesalers who sell directly into the retail channel, hospitals, and retail customers. Provisions for sales discounts, and estimates for chargebacks, and product returns are established as a reduction of product sales revenue at the time revenues are recognized, based on historical experience adjusted to reflect known changes in the factors that impact these reserves. Factors include current contract prices and terms, estimated wholesaler inventory levels, remaining shelf life of product, and historical information for similar products in the same distribution channel. These revenue reductions are generally reflected either as a direct reduction to accounts receivable through an allowance, or as an addition to accrued expenses if the payment is due to a party other than the customer.
35
Product returns. In the pharmaceutical industry, customers are normally granted the right to return product for a refund if the product has not been used prior to its expiration date. Our return policy typically allows product returns for products within an twelve-month window from six months prior to the expiration date and up to six months after the expiration date. We estimate the level of sales which will ultimately be returned pursuant to our return policy, and record a related reserve at the time of sale. These amounts are deducted from our gross sales to determine our net revenues. Our estimates take into consideration historical returns of our products and our future expectations. We periodically review the reserves established for returns and adjust them based on actual experience. The amount of actual product returns could be either higher or lower than the amounts we accrued. Changes in our estimates would be recorded in the income statement in the period of the change. If we over or under estimate the quantity of product which will ultimately be returned, there may be a material impact to our financial statements.
Other revenues. Other revenues consist of (i) rental income received for the lease of retail space to various retail merchants; (ii) advertising revenues from the lease of counter space at our retail locations; (iii) rental income from the lease of retail space to licensed medical practitioners; and (iv) revenues received by us for research and development projects. We recognize revenues upon performance of such funded research. We recognize revenues from leasing of space as earned from contracting third parties. Revenues received in advance are reflected as deferred revenue on the accompanying balance sheet. Additionally, we receive income from the sale of developed drug formulas. Income from the sale of drug formulas are recognized upon performance of all of our obligations under the respective sales contract and are included in other income on the accompanying consolidated statement of operations.
Accounts Receivable
Accounts receivable are carried at original invoice amount less an estimate made for doubtful receivables based on a review of all outstanding amounts on a monthly basis. Management judgment and estimates are made in connection with establishing the allowance for doubtful accounts. Specifically, we analyze the aging of accounts receivable balances, historical bad debts, customer concentrations, customer credit-worthiness, current economic trends and changes in our customer payment terms. Significant changes in customer concentration or payment terms, deterioration of customer credit-worthiness or weakening in economic trends could have a significant impact on the collectibility of receivables and our operating results. If the financial condition of our customers were to deteriorate, resulting in an impairment of their ability to make payments, additional allowances may be required. The reserve for bad debts increased to $539,627 at December 31, 2006 from $45,205 at December 31, 2005. This increase represents 1.4% of total revenues and is due to the increasing level of sales revenue and its corresponding exposure. At December 31, 2006, accounts receivable, net of allowance for doubtful accounts and sales returns, amounted to $7,277,067.
Inventories
Inventory is stated at the lower of cost or market with cost determined under the weighted-average method. Inventory consists of finished capsules, liquids, finished oral suspension powder and other western and traditional Chinese medicines and medical equipment. At least on a quarterly basis, we review our inventory levels and write down inventory that has become obsolete or has a cost basis in excess of its expected net realizable value or is in excess of expected requirements. Inventory levels are evaluated by management relative to product demand, remaining shelf life, future marketing plans and other factors, and reserves for obsolete and slow-moving inventories are recorded for amounts which may not be realizable.
Property and Equipment
Property and equipment are carried at cost. The cost of repairs and maintenance is expensed as incurred; major replacements and improvements are capitalized. When assets are retired or disposed of, the cost and accumulated depreciation are removed from the accounts, and any resulting gains or losses are included in income in the year of disposition. In accordance with Statement of Financial Accounting Standards (SFAS) No. 144, "Accounting for the Impairment or Disposal of Long-Lived Assets", the Company examines the possibility of decreases in the value of fixed assets when events or changes in circumstances reflect the fact that their recorded value may not be recoverable.
Depreciation is calculated on a straight-line basis over the estimated useful lives of the assets. The useful lives for property and equipment are as follows:
| Buildings and leasehold improvement | 20 to 40 years | |||
| Manufacturing equipment | 10 to 15 years | |||
| Office equipment and furniture | 5 to 8 years |
36
Income Taxes
Taxes are calculated in accordance with taxation principles currently effective in the United States and PRC. The Company accounts for income taxes using the liability method. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized as income in the period that includes the enactment date. A valuation allowance is provided for the amount of deferred tax assets that, based on available evidence, are not expected to be realized.
Recent Accounting Pronouncements
In July 2006, the Financial Accounting Standards Board (“FASB”) issued FASB Interpretation No. 48 (“FIN 48”), “Accounting for Uncertainty in Income Taxes,” an interpretation of FASB Statement No. 109 (“SFAS 109”). The interpretation clarifies the accounting for uncertainty in income taxes recognized in an entity’s financial statements in accordance with SFAS 109, “Accounting for Income Taxes.” It prescribes a recognition threshold and measurement attribute for financial statement recognition and measurement of a tax position taken or expected to be taken on a tax return. FIN 48 is effective for fiscal years beginning after December 15, 2006. We are currently evaluating the impact, if any, of FIN 48 on our financial statements.
In September 2006, the FASB issued SFAS No. 157, “Fair Value Measurements” (SFAS 157), which provides guidance for how companies should measure fair value when required to use a fair value measurement for recognition or disclosure purposes under generally accepted accounting principle (GAAP). SFAS 157 is effective for fiscal years beginning after November 15, 2007. We are currently assessing the impact, if any, the adoption of SFAS 157 will have on our financial statements.
In December 2006, FASB Staff Position No. EITF 00-19-2, “Accounting for Registration Payment Arrangements,” was issued. The FSP specifies that the contingent obligation to make future payments or otherwise transfer consideration under a registration payment arrangement, whether issued as a separate agreement or included as a provision of a financial instrument or other agreement, should be separately recognized and measured in accordance with SFAS No. 5, “Accounting for Contingencies.” We believe that our current accounting is consistent with the FSP. Accordingly, adoption of the FSP had no effect on our financial statements.
In February 2007, the FASB issued SFAS No. 159, “The Fair Value Option for Financial Assets and Financial Liabilities, Including an Amendment of FASB Statement No. 115”, under which entities will now be permitted to measure many financial instruments and certain other assets and liabilities at fair value on an instrument-by-instrument basis. This Statement is effective as of the beginning of an entity’s first fiscal year that begins after November 15, 2007. Early adoption is permitted as of the beginning of a fiscal year that begins on or before November 15, 2007, provided the entity also elects to apply the provisions of SFAS 157. We are currently assessing the impact, if any, the adoption of SFAS 159 will have on our financial statements.
Other accounting standards that have been issued or proposed by the FASB or other standards-setting bodies that do not require adoption until a future date are not expected to have a material impact on the consolidated financial statements upon adoption.
Off Balance Sheet Arrangements
As of the date of this Registration Statement, we do not have any off-balance sheet arrangements that have or are reasonably likely to have a current or future effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that are material to investors. The term "off-balance sheet arrangement" generally means any transaction, agreement or other contractual arrangement to which an entity unconsolidated with us is a party, under which we have: (i) any obligation arising under a guarantee contract, derivative instrument or variable interest; or (ii) a retained or contingent interest in assets transferred to such entity or similar arrangement that serves as credit, liquidity or market risk support for such assets.
37
We also lease the following properties in Beijing, China:
Property Location (District of Beijing, China) | Area (sq. ft) | Lease Expiration Period | Purpose | |||
| Fengtai District | 4,413 | August 31, 2009 | Liang Fang warehouse | |||
| Fengtai District | 11,345 | December 31, 2009 | Liang Fang headquarters | |||
| Fengtai District | 12,917 | July 3, 2019 | Retail - Xin Zhong Tai Pharmacy | |||
| Fengtai District | 2,067 | December 31, 2007 | Retail - Nangong Pharmacy | |||
| Fengtai District | 2,153 | December 31, 2010 | Retail - Chen Zhuang Rd. Pharmacy | |||
| Haidian District | 3,660 | December 31, 2009 | Retail - Wan Shou Rd. Pharmacy | |||
| Dongcheng District | 2,153 | May 31, 2009 | Retail - Qingnianhu Pharmacy | |||
| Dongcheng District | 807 | Month-to-Month | Retail - He Ping Li Pharmacy | |||
| Chaoyang District | 1,550 | December 31, 2008 | Retail - Capital Airport Pharmacy | |||
| Chaoyang District | 2,691 | Month-to-Month (related party) | Retail - Feng Lin Lu Zhou Pharmacy | |||
| Fangshan District | 1,615 | December 31, 2010 | Retail - Yong An Zhong Shen Pharmacy | |||
| Liujia Village | 2,153 | October 9, 2007 | Retail - Youth Lake Pharmacy | |||
| Chaoyan District | 72,118 | Property Owned by Lotus | En Zhe Jia Shi production and manufacturing facility |
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS; DIRECTOR INDEPENDENCE
Certain Relationships and Related Transactions
Lotus’ Contractual Arrangements with Lotus East and Its Shareholders
PRC law currently limits foreign equity ownership of Chinese companies. To comply with these foreign ownership restrictions, we operate our business in China through a series of contractual arrangements between Lotus International and Lotus East and its majority shareholders that were executed on September 6, 2006. For a description of these contractual arrangements, see “Description of Business.”
Related Party Transactions of Lotus East
Set forth below are the related party transactions between Lotus East’s shareholders, officers and/or directors, and Lotus East, with whom Lotus International has contractual arrangements which give us the ability to substantially influence Lotus East’s daily operations and financial affairs, appoint its senior executives and approve all matters requiring shareholder approval.
Revolving Line of Credit.
On December 31, 2005, Lotus East entered into a Loan Agreement with Dr. Liu Zhong Yi, the Company’s Chief Executive Officer and Chairman of the Board, whereby Dr. Liu agreed to grant Lotus East a revolving line of credit of approximately US$1,268,477 for the purpose of purchasing raw materials and equipment. The term of the loan is 10 years and the interest rate is at a 20% discount to the PRC banking loan interest rate, payable annually.
38
On December 31, 2005, Lotus East entered into a Loan Agreement with Song Zhenghong, a director of the Company, whereby Mr. Song agreed to grant Lotus East a revolving line of credit of approximately US$3,035,170 for the purpose of purchasing raw materials and equipment. The term of the loan is 10 years and the interest rate is at a 20% discount to the PRC banking loan interest rate, payable annually.
Lease Agreement
Dr. Liu leases to Lotus East a 249 square meter retail space for no charge on a month-to-month basis.
General Partnership Agreement
On March 15, 2006, Lotus East entered into a General Partnership Agreement (the “Partnership Agreement”) with Genesis Equity Partners, LLC (“GEP”) to engage GEP as a non-exclusive external consultant in connection with financial and related matters, including assisting Lotus East in becoming a publicly traded company in the United States. The term of the Partnership Agreement is eighteen (18) months. Pursuant to the Partnership Agreement, GEP received 35% of the outstanding shares of Lotus International in connection with the signing of the Management Agreements between Lotus International and Lotus East and the Exchange Agreement between Lotus and SEAA. GEP opted to receive the shares in the following manner: 8.52% to GEP, 16.82% to Genesis Technology Group, Inc. (“GTEC”), GEP’s parent company, and 7.92% to Joshua Tan, one of GTEC’s directors. The Partnership Agreement also provides for GEP to provide “post-registration” services to Lotus East, including assisting the Company with its SEC filing obligations, for a general service fee of US$10,000 per month; provided, however, that the service fee payment shall not start until the Company receives funding of a minimum of US$1,000,000.
Chief Financial Officer
Adam Wasserman, our Chief Financial Officer, is also the Chief Financial Officer of GTEC, which owns more than 10% of the issued and outstanding common stock of the Company. GTEC is also the parent company of GEP, with which Lotus East has a Partnership Agreement, as described above.
Other Related Party Transactions
On October 10, 2006, we entered into a five-year loan agreement and contract (“Loan Agreement and Contract”) with Wu Lan Cha Bu Emergency Hospital (“Wu Lan”), whereby we agreed to lend to Wu Lan approximately $3,840,000 for the construction of a hospital ward in Inner Mongolia, China (the “Loan”). In exchange for the Loan, Wu Lan agreed that the Company would be the exclusive provider for all medicines and disposable medical treatment apparatus to Wu Lan for a period of twenty (20) years. In October 2006, Liu Zhong Yi, our Chief Executive Officer, loaned these funds to Wu Lan on behalf of the Company. Accordingly, on October 10, 2006 we entered into an Assignment Agreement whereby we assigned all of our rights, obligations, and receipts under the Loan Agreement to Dr. Liu, except for the rights to revenues from the sale of medical and disposable medical treatment apparatus which will remain with us. As compensation to Dr. Liu for accepting the assignment under the loan agreement including all of the risks and obligations and for Dr. Liu not accepting the rights to revenues from the sale of medical and disposable medical treatment apparatus which will remain with the Company, we agreed to pay Dr. Liu an aggregate of approximately $1,150,000 to be paid in five (5) equal annual installments of approximately $230,000.
Director Independence
The following information concerning director independence is based on the director independence standards of The NASDAQ Stock Market Corporate Governance Rules.
The Board has determined that directors Mel Rothberg, Dr. Ian Ashley, and Caeli Widger are independent directors within The NASDAQ Stock Market's director independence standards. Directors Song Zhenghong, Xian Xuemei, Li Ping, and Liu Jin are not independent. In determining independence, the Board reviews and seeks to determine whether directors have any material relationship with the Company, direct or indirect, which would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. The Board reviews business, professional, charitable and familial relationships of the directors in determining independence. The Board has not designated a separate compensation or nominating committee.
39
MARKET FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Our common stock is traded over-the-counter on the Over-the-Counter Electronic Bulletin Board under the symbol "LTUS.OB". The following table sets forth the high and low bid information for the common stock for each quarter within the last two fiscal years, as reported by the Over-the-Counter Electronic Bulletin Board. The bid prices reflect inter-dealer quotations, do not include retail markups, markdowns or commissions and do not necessarily reflect actual transactions.
| LOW | HIGH | ||||||
| 2007 | |||||||
| First Quarter | $ | 1.55 | $ | 2.50 | |||
| 2006 | |||||||
| Fourth Quarter | $ | 0.40 | $ | 2.50 | |||
| Third Quarter | $ | 0.40 | $ | 0.40 | |||
| Second Quarter | $ | 0.40 | $ | 0.40 | |||
| First Quarter | $ | 0.40 | $ | 0.40 | |||
| 2005 | |||||||
| Fourth Quarter | $ | 0.40 | $ | 0.40 | |||
| Third Quarter | $ | 0.10 | $ | 0.40 | |||
| Second Quarter | n/a | n/a | |||||
| First Quarter | n/a | n/a | |||||
There was no trading of our common stock prior to August 2005.
As of April 25, 2007, there were approximately 91 stockholders of record of our common stock.
Dividends
We have never paid any dividends on the Common Stock or the Preferred Stock. We currently anticipate that any future earnings will be retained for the development of our business and do not anticipate paying any dividends on the Common Stock or the Preferred Stock in the foreseeable future.
Transfer Agent
Our transfer agent is Signature Stock Transfer, Inc., One Preston Park, 2301 Ohio Drive, Suite 100, Plano, Texas 75093. Their telephone number is (972) 612-4120.
Equity Compensation Plan Information
EXECUTIVE COMPENSATION
The following table summarizes all compensation recorded by us in the last completed fiscal year for our principal executive officer, each other executive officer serving as such whose annual compensation exceeded $100,000, and up to two additional individuals for whom disclosure would have been made in this table but for the fact that the individual was not serving as an executive officer of our company at December 31, 2006. The value attributable to any option awards is computed in accordance with FAS 123R.
40
Summary Annual Compensation Table
Name and Principal Position | Fiscal Year | Salary ($) | Bonus ($) | Stock Awards ($) | Option Awards ($) | Non-Equity Incentive Plan Compensation ($) | Nonqualified Deferred Compensation Earnings ($) | All Other Compensation ($) | Total ($) | |||||||||||||||||||
| Dr. Liu Zhong Yi | 2006 | $ | 37,500 | 0 | 0 | 0 | 0 | 0 | $ | 100,000 | $ | 137,500 | ||||||||||||||||
| current President and CEO (1) | ||||||||||||||||||||||||||||
| Thomas G. Miller | 2006 | $ | 12,500 | 0 | $ | 10,900 | 0 | 0 | 0 | 0 | $ | 23,400 | ||||||||||||||||
| former President, CEO and CFO | ||||||||||||||||||||||||||||
(1) Dr. Liu has served as our Chief Executive Officer and President since September 28, 2006. Dr. Liu’s fiscal 2006 other compensation includes fees paid for car allowances and personal expenses.
Outstanding Equity Awards at Fiscal Year-End
None of our named executive officers had any unexercised options, stock that has not vested, or equity incentive plan awards outstanding as of December 31, 2006.
Employment Agreements, Termination of Employment, and Change-in-Control Arrangements for Lotus
We currently have no employment agreements with any of our executive officers, nor any compensatory plans or arrangements resulting from the resignation, retirement or any other termination of any of our executive officers, from a change-in-control, or from a change in any executive officer's responsibilities following a change-in-control. However, in December 2006, the Board of Directors approved compensation for Dr. Liu Zhong Yi and Adam Wasserman.
Dr. Liu Zhong Yi, our Chief Executive Officer, has a compensation package that provides for base compensation of $150,000 per year. Dr. Liu’s compensation began on October 1, 2006. Dr. Liu’s salary will be reviewed annually.
Adam Wasserman, our Chief Financial Officer, has a compensation package that provides for base compensation of $93,600 per year. Mr. Wasserman's compensation began on October 1, 2006. Mr. Wasserman's salary will be reviewed annually. Mr. Wasserman began with the Company on October 1, 2006. Mr. Wasserman’s salary is payable as follows: $50,000 in cash and $43,600 in our common stock.
Compensation of Directors
In 2006, members of the Board of Directors did not currently receive any cash compensation for their services as Directors. To attract and keep Board members the Company will grant options to Board members in fiscal 2007.
See our Financial Statements beginning on page F-1.
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS
In November, 2006, we replaced LBB & Associates, Ltd., LLP (formerly Lopez, Blevins, Bork & Associates, LLP) (“Lopez”) as the Company’s independent accountant with Sherb & Co., LLP as the Company’s Independent Registered Public Accounting Firm to audit the Company’s financial statements for the fiscal year ending December 31, 2006.
Lopez audited our financial statements for the periods ended December 31, 2005 and December 31, 2004. Lopez did not resign or decline to stand for re-election.
Lopez's report dated March 1, 2006, on our balance sheet as of December 31, 2005 and the related statements of operations, stockholders' deficit, and cash flows for the year ended December 31, 2005 and for the period from January 28, 2004 (inception) to December 31, 2004, did not contain an adverse opinion or a disclaimer of opinion, nor was qualified or modified as to uncertainty, audit scope, or accounting principles, except that the report indicated we had suffered losses in 2005 and 2004 and that we were required to raise additional capital; and the report raised substantial doubts on our ability to continue as a going concern.
41
During our two most recent fiscal years and the subsequent interim period preceding our decision not to re-appoint Lopez, we had no disagreements with them on any matter of accounting principles or practices, financial statement disclosure, or auditing scope of procedure, which disagreement if not resolved to the satisfaction of Lopez, would have caused it to make reference to the subject matter of the disagreement in connection with its report.
During our two most recent fiscal years and the subsequent interim period prior to retaining Sherb & Co., LLP: (1) neither we nor anyone on our behalf consulted Sherb & Co., LLP regarding (a) either the application of accounting principles to a specified transaction, either completed or proposed, or the type of audit opinion that might be rendered on our consolidated financial statements, or (b) any matter that was the subject of a disagreement or a reportable event as set forth in Item 304(a)(1)(iv) and (v), respectively, of Regulation S-B, and (2) Sherb & Co., LLP did not provide us with a written report or oral advice that they concluded was an important factor considered by us in reaching a decision as to accounting, auditing or financial reporting issue.
We have provided LBB with a copy of the disclosure made in response to Item 304(a) of Regulation S-B in this Registration Statement. LBB has furnished the Company with a letter addressed to the Commission, which is incorporated by reference hereto as Exhibit 16.1.
During the past two fiscal years, there have been no disagreements with the Company’s independent auditors.
42
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
CONSOLIDATED FINANCIAL STATEMENTS
December 31, 2006
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
CONTENTS
| Report of Independent Registered Public Accounting Firm | F-2 | |
| Consolidated Financial Statements: | ||
| Consolidated Balance Sheet | F-3 | |
| Consolidated Statements of Operations | F-4 | |
| Consolidated Statements of Shareholders’ Equity | F-5 | |
| Consolidated Statements of Cash Flows | F-6 | |
| Notes to Consolidated Financial Statements | F-7 to F-22 |
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors
Lotus Pharmaceuticals, Inc. and Subsidiaries
Beijing, China
We have audited the accompanying consolidated balance sheet of Lotus Pharmaceuticals, Inc. and Subsidiaries as of December 31, 2006 and the related consolidated statements of operations, changes in shareholders' equity, and cash flows for the years ended December 31, 2006 and 2005. These consolidated financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purposes of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit includes examining on a test basis, evidence supporting the amount and disclosures in the combined financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Lotus Pharmaceuticals, Inc. and Subsidiaries as of December 31, 2006, and the results of their operations and their cash flows for the years ended December 31, 2006 and 2005, in conformity with accounting principles generally accepted in the United States of America.
| By: | /s/ Sherb & Co., LLP | |
Certified Public Accountants | ||
Boca Raton, Florida
March 17, 2007
F-2
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
CONSOLIDATED BALANCE SHEET
December 31, 2006
| ASSETS | ||||
| CURRENT ASSETS: | ||||
| Cash | $ | 2,089,156 | ||
| Accounts receivable, net of allowance for doubtful accounts of $539,627 | ||||
| and sales returns of $2,297,399 | 7,277,067 | |||
| Inventories, net of reserve for obsolete inventory of $280,251 | 3,133,677 | |||
| Prepaid expenses | 252,103 | |||
| Total Current Assets | 12,752,003 | |||
| PROPERTY AND EQUIPMENT - Net | 5,813,935 | |||
| OTHER ASSETS | ||||
| Intangible assets, net of accumulated amortization | 208,169 | |||
| Due from related party | 1,826,626 | |||
| Total Assets | $ | 20,600,733 | ||
| LIABILITIES AND SHAREHOLDERS' EQUITY | ||||
| CURRENT LIABILITIES: | ||||
| Accounts payable and accrued expenses | $ | 705,147 | ||
| VAT and service taxes payable | 2,047,851 | |||
| Advances from customers | 231,340 | |||
| Unearned revenue | 426,358 | |||
| Due to related parties | 695,231 | |||
| Total Current Liabilities | 4,105,927 | |||
| LONG-TERM LIABILITIES: | ||||
| Notes payable - related parties | 4,729,880 | |||
| Total Liabilities | 8,835,807 | |||
| SHAREHOLDERS' EQUITY: | ||||
| Common stock ($.001 par value; 50,000,000 shares authorized; | ||||
| 41,280,000 shares issued and outstanding) | 41,280 | |||
| Additional paid-in capital | 5,965,323 | |||
| Retained earnings | 5,258,007 | |||
| Other comprehensive gain - cumulative foreign currency translation adjustment | 500,316 | |||
| Total Shareholders' Equity | 11,764,926 | |||
| Total Liabilities and Shareholders' Equity | $ | 20,600,733 | ||
See notes to consolidated financial statements
F-3
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
CONSOLIDATED STATEMENTS OF OPERATIONS
| For the Year Ended | |||||||
| December 31, | |||||||
| 2006 | 2005 | ||||||
| NET REVENUES: | |||||||
| Wholesale | $ | 25,430,069 | $ | 16,099,036 | |||
| Retail | 3,532,623 | 1,629,385 | |||||
| Other revenues | 7,244,997 | 617,420 | |||||
| Total Revenues | 36,207,689 | 18,345,841 | |||||
| COST OF SALES | 26,523,039 | 14,584,682 | |||||
| GROSS PROFIT | 9,684,650 | 3,761,159 | |||||
| OPERATING EXPENSES: | |||||||
| Selling expenses | 2,154,216 | 414,361 | |||||
| Research and development | 387,337 | 1,759,756 | |||||
| General and administrative | 2,716,536 | 975,003 | |||||
| Total Operating Expenses | 5,258,089 | 3,149,120 | |||||
| INCOME FROM OPERATIONS | 4,426,561 | 612,039 | |||||
| OTHER INCOME (EXPENSE): | |||||||
| Other income | - | 1,219,512 | |||||
| Interest income | - | 161 | |||||
| Interest expense | (325,824 | ) | - | ||||
| Total Other Income (Expense) | (325,824 | ) | 1,219,673 | ||||
| INCOME BEFORE INCOME TAXES | 4,100,737 | 1,831,712 | |||||
| INCOME TAXES | - | - | |||||
| NET INCOME | $ | 4,100,737 | $ | 1,831,712 | |||
| COMPREHENSIVE INCOME: | |||||||
| NET INCOME | $ | 4,100,737 | $ | 1,831,712 | |||
| OTHER COMPREHENSIVE INCOME: | |||||||
| Unrealized foreign currency translation gain | 326,679 | 168,543 | |||||
| COMPREHENSIVE INCOME | $ | 4,427,416 | $ | 2,000,255 | |||
| NET INCOME PER COMMON SHARE: | |||||||
| Basic | $ | 0.10 | $ | 0.05 | |||
| Diluted | $ | 0.10 | $ | 0.05 | |||
| WEIGHTED AVERAGE COMMON SHARES OUTSTANDING: | |||||||
| Basic | 40,360,530 | 40,041,600 | |||||
| Diluted | 40,360,530 | 40,041,600 | |||||
See notes to consolidated financial statements
F-4
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
CONSOLIDATED STATEMENTS OF SHAREHOLDERS' EQUITY
For the Years Ended December 31, 2006 and 2005
| Common Stock, $.001 Par Value | |||||||||||||||||||
| Number of Shares | Amount | Additional Paid-in Capital | Retained Earnings (Deficit) | Other Comprehensive Income | Total Shareholders' Equity | ||||||||||||||
| Balance, December 31, 2004 | 40,041,600 | $ | 40,042 | $ | 5,972,243 | $ | (674,442 | ) | $ | 5,094 | $ | 5,342,937 | |||||||
| Member Contributions | - | - | 818 | - | - | 818 | |||||||||||||
| Comprehensive income: | |||||||||||||||||||
| Net income for the year | - | - | - | 1,831,712 | - | 1,831,712 | |||||||||||||
| Foreign currency translation adjustment | - | - | - | - | 168,543 | 168,543 | |||||||||||||
| �� | |||||||||||||||||||
| Total comprehensive income | - | - | - | - | - | 2,000,255 | |||||||||||||
| Balance, December 31, 2005 | 40,041,600 | 40,042 | 5,973,061 | 1,157,270 | 173,637 | 7,344,010 | |||||||||||||
| Recapitalization of Company | 1,238,400 | 1,238 | (7,738 | ) | - | - | (6,500 | ) | |||||||||||
| Comprehensive income: | |||||||||||||||||||
| Net income for the year | - | - | - | 4,100,737 | - | 4,100,737 | |||||||||||||
| Foreign currency translation adjustment | - | - | - | - | 326,679 | 326,679 | |||||||||||||
| Total comprehensive income | - | - | - | - | - | 4,427,416 | |||||||||||||
| Balance, December 31, 2006 | 41,280,000 | $ | 41,280 | $ | 5,965,323 | $ | 5,258,007 | $ | 500,316 | $ | 11,764,926 | ||||||||
See notes to consolidated financial statements
F-5
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
CONSOLIDATED STATEMENTS OF CASH FLOWS
| For the Year Ended | |||||||
| December 31, | |||||||
| 2006 | 2005 | ||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | |||||||
| Net income | $ | 4,100,737 | $ | 1,831,712 | |||
| Adjustments to reconcile net income from operations to net cash provided by (used in) operating activities: | |||||||
| Depreciation and amortization | 567,469 | 429,633 | |||||
| Increase in allowance for doubtful accounts and sales returns | 2,732,889 | 26,865 | |||||
| Changes in assets and liabilities: | |||||||
| Accounts receivable | (8,922,247 | ) | (597,584 | ) | |||
| Inventories | 3,928,236 | (2,975,432 | ) | ||||
| Prepaid and other current assets | (75,155 | ) | 622,007 | ||||
| Other assets | - | 30,006 | |||||
| Accounts payable and accrued expenses | 206,516 | 137,331 | |||||
| VAT and service taxes payable | 1,999,675 | 2,519 | |||||
| Unearned revenue | 200,811 | 38,992 | |||||
| Advances from customers | 39,333 | 110,671 | |||||
| NET CASH PROVIDED BY (USED IN) OPERATING ACTIVITIES | 4,778,264 | (343,280 | ) | ||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | |||||||
| Decrease (increase) in due from related parties | 1,243,742 | (2,163,128 | ) | ||||
| Purchase of property and equipment | (1,480 | ) | (2,045,210 | ) | |||
| NET CASH PROVIDED BY (USED IN) INVESTING ACTIVITIES | 1,242,262 | (4,208,338 | ) | ||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | |||||||
| Capital contributions | - | 818 | |||||
| Proceeds from related party advances | - | 4,623,713 | |||||
| Repayments of related party advances | (680,913 | ) | - | ||||
| Repayments of notes payable - related parties | (3,456,196 | ) | - | ||||
| NET CASH PROVIDED BY (USED IN) FINANCING ACTIVITIES | (4,137,109 | ) | 4,624,531 | ||||
| EFFECT OF EXCHANGE RATE ON CASH | 44,805 | 3,364 | |||||
| NET INCREASE IN CASH | 1,928,222 | 76,277 | |||||
| CASH - beginning of year | 160,934 | 84,657 | |||||
| CASH - end of year | $ | 2,089,156 | $ | 160,934 | |||
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: | |||||||
| Cash paid for: | |||||||
| Interest | $ | 325,824 | $ | - | |||
| Income taxes | $ | - | $ | - | |||
See notes to consolidated financial statements.
F-6
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Organization
Lotus Pharmaceuticals, Inc. (“Lotus” or the “Company”), formerly S.E. Asia Trading Company, Inc. (“SEAA”), was incorporated on January 28, 2004 under the laws of the State of Nevada. SEAA operated as a retailer of jewelry, framed art and home accessories. In December 2006, SEAA changed its name to Lotus Pharmaceuticals, Inc.
On September 6, 2006, the Company entered into a definitive Share Exchange Agreement with Lotus Pharmaceutical International, Inc. (“Lotus International”), whereby the Company acquired all of the outstanding common stock of Lotus International in exchange for newly-issued stock of the Company to Lotus International’s shareholders. On September 28, 2006 (the closing date), Lotus International became a wholly-owned subsidiary of the Company and Lotus International’s shareholders own the majority of the Company’s voting stock. The acquisition of Lotus International by the Company was accounted for as a reverse merger because on a post-merger basis, the former shareholders of Lotus International hold a majority of the outstanding common stock of the Company on a voting and fully-diluted basis. As a result, Lotus International is deemed to be the acquirer for accounting purposes.
Lotus International was incorporated under the laws the State of Nevada on August 28, 2006 to develop and market pharmaceutical products in the People's Republic of China (“PRC” or “China”). PRC law currently has limits on foreign ownership of certain companies. To comply with these foreign ownership restrictions, Lotus operates its pharmaceutical business in China through Lotus East. Lotus International has contractual arrangements with Lotus East and its shareholders pursuant to which Lotus International will provide technology consulting and other general business operation services to Lotus East. Through these contractual arrangements, Lotus International also has the ability to substantially influence Lotus East’s daily operations and financial affairs, appoint its senior executives and approve all matters requiring shareholder approval. As a result of these contractual arrangements, which enable Lotus International to control Lotus East, Lotus International is considered the primary beneficiary of Lotus East.
On September 6, 2006, Lotus International entered into the following contractual arrangements:
Consulting Services Agreement. Pursuant to the exclusive consulting services agreements between Lotus and Lotus East, Lotus has the exclusive right to provide to Lotus East general pharmaceutical business operations services as well as consulting services related to the technological research and development of pharmaceutical products as well as general business operation advice and strategic planning (the “Services”). Under this agreement, Lotus owns the intellectual property rights developed or discovered through research and development, in the course of providing the Services, or derived from the provision of the Services. Lotus East pays a quarterly consulting service fees in Renminbi (“RMB”) to Lotus that is equal to Lotus East’s revenue, as defined, for such quarter.
F-7
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Organization (continued)
Operating Agreement. Pursuant to the operating agreement among Lotus, Lotus East and the shareholders of Lotus East, (collectively “Lotus East’s Shareholders”), Lotus provides guidance and instructions on Lotus East’s daily operations, financial management and employment issues. The shareholders of Lotus East must designate the candidates recommended by Lotus as their representatives on Lotus East’s Board of Directors. Lotus has the right to appoint senior executives of Lotus East. In addition, Lotus agreed to guarantee Lotus East’s performance under any agreements or arrangements relating to Lotus East’s business arrangements with any third party. Lotus East, in return, agreed to pledge its accounts receivable and all of its assets to Lotus. Moreover, Lotus East agreed that without the prior consent of Lotus, Lotus East would not engage in any transaction that could materially affect the assets, liabilities, rights or operations of Lotus East, including, without limitation, incurrence or assumption of any indebtedness, sale or purchase of any assets or rights, incurrence of any encumbrance on any of its assets or intellectual property rights in favor of a third party or transfer of any agreements relating to its business operation to any third party. The term of this agreement is ten (10) years from September 6, 2006 and may be extended only upon Lotus’s written confirmation prior to the expiration of the this agreement, with the extended term to be mutually agreed upon by the parties.
Equity Pledge Agreement. Under the equity pledge agreement between the shareholders of Lotus East and Lotus, the shareholders of Lotus East pledged all of their equity interests in Lotus East to Lotus to guarantee Lotus East’s performance of its obligations under the technology consulting agreement. If Lotus East or Lotus East’s Shareholders breaches its respective contractual obligations, Lotus, as pledgee, will be entitled to certain rights, including the right to sell the pledged equity interests. Lotus East’s Shareholders also agreed that upon occurrence of any event of default, Lotus shall be granted an exclusive, irrevocable power of attorney to take actions in the place and stead of Lotus East’s Shareholders to carry out the security provisions of the equity pledge agreement and take any action and execute any instrument that Lotus may deem necessary or advisable to accomplish the purposes of the equity pledge agreement. The shareholders of Lotus East agreed not to dispose of the pledged equity interests or take any actions that would prejudice Lotus’ interest. The equity pledge agreement will expire two (2) years after Lotus East’s obligations under the exclusive consulting services agreements have been fulfilled.
Option Agreement. Under the option agreement between the shareholders of Lotus East and Lotus, the shareholders of Lotus East irrevocably granted Lotus or its designated person an exclusive option to purchase, to the extent permitted under PRC law, all or part of the equity interests in Lotus East for the cost of the initial contributions to the registered capital or the minimum amount of consideration permitted by applicable PRC law. Lotus or its designated person has sole discretion to decide when to exercise the option, whether in part or in full. The term of this agreement is ten (10) years from September 6, 2006 and may be extended prior to its expiration by written agreement of the parties.
Proxy Agreement. Pursuant to the proxy agreement among Lotus and Lotus East’s Shareholders, Lotus East’s Shareholders agreed to irrevocably grant a person to be designated by Lotus with the right to exercise Lotus East’s Shareholders’ voting rights and their other rights, including the attendance at and the voting of Lotus East’s Shareholders’ shares at the shareholders’ meetings (or by written consent in lieu of such meetings) in accordance with applicable laws and its Article of Association, including but not limited to the rights to sell or transfer all or any of his equity interests of Lotus East, and appoint and vote for the directors and Chairman as the authorized representative of the shareholders of Lotus East. The term of this Proxy Agreement is ten (10) years from September 6, 2006 and may be extended prior to its expiration by written agreement of the parties.
F-8
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Organization (continued)
Liang Fang Pharmaceuticals Co. Ltd. (“Liang Fang”) is a Chinese limited liability company and was formed under laws of the People’s Republic of China on June 21, 2000. Liang Fang is engaged in the production, trade and retailing of pharmaceuticals. Further, Liang Fang is focused on development of innovative medicines and investing strategic growth to address various medical needs for patients worldwide. Liang Fang’s operations are based in Beijing, China.
Liang Fang owns and operates several drug stores throughout Beijing, China. These drugstores sell Western and traditional Chinese medicines, and medical treatment accessories.
Liang Fang’s affiliate, Beijing En Zhe Jia Pharmaceutical Co., Ltd. (“En Zhe Jia”) is a Chinese limited liability company and was formed under laws of the People’s Republic of China on September 17, 1999. En Zhe Jia is the sole manufacturer for Liang Fang and maintains facilities for the production of medicines, patented Chinese medicine, as well as the research and production of other new medicines.
As a result of the management agreements between Lotus International and Lotus East, Lotus East was deemed to be the acquirer of Lotus International for accounting purposes. Accordingly, the financial statement data presented are those of Lotus East for all periods prior to the Company’s acquisition of Lotus International on September 28, 2006, and the financial statements of the consolidated companies from the acquisition date forward.
Basis of presentation
The financial statements are prepared in accordance with generally accepted accounting principles in the United States of America ("US GAAP"). The consolidated statements include the accounts of Lotus Pharmaceuticals, Inc. and its wholly-owned subsidiary, Lotus and companies under its control (Liang Fang and En Zhe Jia). All significant inter-company balances and transactions have been eliminated.
Use of estimates
The preparation of financial statements in conformity with US GAAP requires management to make estimates and assumptions that affect certain reported amounts and disclosures. Accordingly, actual results could differ from those estimates. Significant estimates in 2006 and 2005 include the allowance for doubtful accounts and sales returns, the allowance for obsolete inventory, the useful life of property and equipment and intangible assets, and accruals for taxes due.
Fair value of financial instruments
The carrying amounts reported in the balance sheet for cash, accounts receivable, accounts payable and accrued expenses, customer advances, and amounts due from related parties approximate their fair market value based on the short-term maturity of these instruments.
F-9
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - BASIS OF PRESENTATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Cash and cash equivalents
For purposes of the combined statements of cash flows, the Company considers all highly liquid instruments purchased with a maturity of three months or less and money market accounts to be cash equivalents.
Accounts receivable
The Company has a policy of reserving for uncollectible accounts based on its best estimate of the amount of probable credit losses in its existing accounts receivable. The Company periodically reviews its accounts receivable to determine whether an allowance is necessary based on an analysis of past due accounts and other factors that may indicate that the realization of an account may be in doubt. Account balances deemed to be uncollectible are charged to the allowance after all means of collection have been exhausted and the potential for recovery is considered remote. At December 31, 2006, the Company has established, based on a review of its outstanding balances, an allowance for doubtful accounts in the amount of $539,627.
The Company accounts for sales returns in accordance with Statements of Financial Accounting Standards (SFAS) No. 48, Revenue Recognition When Right of Return Exists, by establishing an accrual in an amount equal to its estimate of sales recorded for which the related products are expected to be returned. The Company determines the estimate of the sales return accrual primarily based on historical experience regarding sales return, but also by considering other factors that could impact sales returns. These factors include levels of inventory in the distribution channel, estimated shelf life, product discontinuances, price changes of competitive products, introductions of generic products and introductions of competitive new products. The allowance for product returns at December 31, 2006 was $2,297,399.
Inventories
Inventories, consisting of raw materials and finished goods related to the Company’s products are stated at the lower of cost or market utilizing the weighted average method. An allowance is established when management determines that certain inventories may not be saleable. If inventory costs exceed expected market value due to obsolescence or quantities in excess of expected demand, the Company will record reserves for the difference between the cost and the market value. These reserves are recorded based on estimates. The Company recorded an inventory reserve of $280,251 for the year ended December 31, 2006.
Property and equipment
Property and equipment are carried at cost and are depreciated on a straight-line basis over the estimated useful lives of the assets. The cost of repairs and maintenance is expensed as incurred; major replacements and improvements are capitalized. When assets are retired or disposed of, the cost and accumulated depreciation are removed from the accounts, and any resulting gains or losses are included in income in the year of disposition. In accordance with Statement of Financial Accounting Standards (SFAS) No. 144, "Accounting for the Impairment or Disposal of Long-Lived Assets", the Company examines the possibility of decreases in the value of fixed assets when events or changes in circumstances reflect the fact that their recorded value may not be recoverable.
F-10
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Impairment of long-lived assets
In accordance with Statement of Financial Accounting Standards (SFAS) No. 144, "Accounting for the Impairment or Disposal of Long-Lived Assets," The Company periodically reviews its long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of the assets may not be fully recoverable. The Company recognizes an impairment loss when the sum of expected undiscounted future cash flows is less than the carrying amount of the asset. The amount of impairment is measured as the difference between the asset’s estimated fair value and its book value. The Company did not consider it necessary to record any impairment charges during the year ended December 31, 2006 and 2005.
Advances from customers
Income taxes
The Company is governed by the Income Tax Law of the People’s Republic of China and the United States. Income taxes are accounted for under Statement of Financial Accounting Standards No. 109, "Accounting for Income Taxes," which is an asset and liability approach that requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been recognized in the Company's financial statements or tax returns.
Revenue recognition
Product sales
Product sales are generally recognized when title to the product has transferred to customers in accordance with the terms of the sale. The Company recognizes revenue in accordance with the Securities and Exchange Commission’s (SEC) Staff Accounting Bulletin (SAB) No. 101, “Revenue Recognition in Financial Statements” as amended by SAB No. 104 (together, “SAB 104”), and Statement of Financial Accounting Standards (SFAS) No. 48 “Revenue Recognition When Right of Return Exists.” SAB 104 states that revenue should not be recognized until it is realized or realizable and earned. In general, the Company records revenue when persuasive evidence of an arrangement exists, services have been rendered or product delivery has occurred, the sales price to the customer is fixed or determinable, and collectibility is reasonably assured.
SFAS No. 48 states that revenue from sales transactions where the buyer has the right to return the product shall be recognized at the time of sale only if the seller’s price to the buyer is substantially fixed or determinable at the date of sale, the buyer has paid the seller, or the buyer is obligated to pay the seller and the obligation is not contingent on resale of the product, the buyer’s obligation to the seller would not be changed in the event of theft or physical destruction or damage of the product, the buyer acquiring the product for resale has economic substance apart from that provided by the seller, the seller does not have significant obligations for future performance to directly bring about resale of the product by the buyer, and the amount of future returns can be reasonably estimated.
F-11
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Revenue recognition (continued)
The Company’s net product revenues represent total product revenues less allowances for returns.
Allowance for returns — In general, for wholesale sales, the Company provides credit for product returns that are returned six months prior to and up to six months after the product expiration date. Upon sale, the Company estimates an allowance for future product returns. The Company provides additional reserves for contemporaneous events that were not known and knowable at the time of shipment. In order to reasonably estimate future returns, the Company analyzed both quantitative and qualitative information including, but not limited to, actual return rates, the level of product manufactured by the Company, the level of product in the distribution channel, expected shelf life of the product, current and projected product demand, the introduction of new or generic products that may erode current demand, and general economic and industry wide indicators. The Company also utilizes the guidance provided in SFAS No. 48 and SAB 104 in establishing its return estimates. The allowance for product returns at December 31, 2006 was $2,297,399. This allowance has been netted with accounts receivable on the Company’s balance sheet.
Other revenues
Other revenues consist of (i) leasing revenues received for the lease of retail space to various retail merchants; (ii) advertising revenues from the lease of counter space at the Company’s retail locations; (iii) leasing revenue from the lease of retail space to licensed medical practitioners; (iv) revenues received by the Company for research and development projects and lab testing jobs conducted on behalf of third party companies, and; (v) revenues received for performing third party contract manufacturing projects. In connection with third-party manufacturing, the customer supplies the raw materials and we are paid a fee for manufacturing their product and revenue is recognized at the completion of the manufacturing job. The Company recognizes revenues from leasing of space and advertising revenues as earned from contracting third parties. The Company recognizes revenues upon performance of a any research or lab testing jobs. Revenues received in advance are reflected as deferred revenue on the accompanying balance sheet. Additionally, the Company receives income from the sale of developed drug formulas. Income from the sale of drug formulas are recognized upon performance of all of the Company’s obligations under the respective sales contract and are included in other income on the accompanying consolidated statement of operations.
Concentrations of credit risk
Financial instruments which potentially subject the Company to concentrations of credit risk consist principally of cash and trade accounts receivable. Substantially all of the Company’s cash is maintained with state-owned banks within the People’s Republic of China of which no deposits are covered by insurance. The Company has not experienced any losses in such accounts and believes it is not exposed to any risks on its cash in bank accounts. A significant portion of the Company's sales are credit sales which are primarily to customers whose ability to pay is dependent upon the industry economics prevailing in these areas; however, concentrations of credit risk with respect to trade accounts receivables is limited due to generally short payment terms. The Company also performs ongoing credit evaluations of its customers to help further reduce credit risk.
Shipping costs
Shipping costs are included in cost of sales and totaled $963,504 and $236,237 for the years ended December 31, 2006 and 2005, respectively.
F-12
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Advertising
Advertising is expensed as incurred. Advertising expenses amounted to $81,888 and $683 for the years ended December 31, 2006 and 2005, respectively.
Foreign currency translation
The reporting currency is the U.S. dollar. The functional currency of the Company is the local currency, the Chinese Renminbi (“RMB”). The financial statements of the Company are translated into United States dollars using year-end rates of exchange for assets and liabilities, and average rates of exchange for the period for revenues, costs, and expenses. Translation adjustments resulting from the process of translating the local currency financial statements into U.S. dollars are included in determining comprehensive income. The cumulative translation adjustment and effect of exchange rate changes on cash at December 31, 2006 was $44,805.
Research and development
Research and development costs are expensed as incurred. These costs primarily consist of cost of material used and salaries paid for the development of the Company’s products and fees paid to third parties. Research and development costs for the years ended December 31, 2006 and 2005 were approximately $387,000 and $1,760,000, respectively, and are included in operating expenses.
Accumulated other comprehensive income
Accumulated other comprehensive income consisted of unrealized gains on currency translation adjustments from the translation of financial statements from Chinese RMB to US dollars. For the years ended December 31, 2006 and 2005, accumulated other comprehensive income was $326,679 and $168,543, respectively.
Recent Accounting Pronouncements
In July 2006, the Financial Accounting Standards Board (“FASB”) issued FASB Interpretation No. 48 (“FIN 48”), “Accounting for Uncertainty in Income Taxes,” an interpretation of FASB Statement No. 109 (“SFAS 109”). The interpretation clarifies the accounting for uncertainty in income taxes recognized in an entity’s financial statements in accordance with SFAS 109, “Accounting for Income Taxes.” It prescribes a recognition threshold and measurement attribute for financial statement recognition and measurement of a tax position taken or expected to be taken on a tax return. FIN 48 is effective for fiscal years beginning after December 15, 2006. The Company is currently evaluating the impact, if any, of FIN 48 on its financial statements.
In September 2006, the FASB issued SFAS No. 157, “Fair Value Measurements” (SFAS 157), which provides guidance for how companies should measure fair value when required to use a fair value measurement for recognition or disclosure purposes under generally accepted accounting principle (GAAP). SFAS 157 is effective for fiscal years beginning after November 15, 2007. The Company is currently assessing the impact, if any, the adoption of SFAS 157 will have on its financial statements.
F-13
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 1 - ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Recent Accounting Pronouncements (continued)
In December 2006, FASB Staff Position No. EITF 00-19-2, “Accounting for Registration Payment Arrangements,” was issued. The FSP specifies that the contingent obligation to make future payments or otherwise transfer consideration under a registration payment arrangement, whether issued as a separate agreement or included as a provision of a financial instrument or other agreement, should be separately recognized and measured in accordance with SFAS No. 5, “Accounting for Contingencies.” The Company believes that its current accounting is consistent with the FSP. Accordingly, adoption of the FSP had no effect on its financial statements.
In February 2007, the FASB issued SFAS No. 159, “The Fair Value Option for Financial Assets and Financial Liabilities, Including an Amendment of FASB Statement No. 115”, under which entities will now be permitted to measure many financial instruments and certain other assets and liabilities at fair value on an instrument-by-instrument basis. This Statement is effective as of the beginning of an entity’s first fiscal year that begins after November 15, 2007. Early adoption is permitted as of the beginning of a fiscal year that begins on or before November 15, 2007, provided the entity also elects to apply the provisions of SFAS 157. The Company is currently assessing the impact, if any, the adoption of SFAS 159 will have on its financial statements.
Reclassifications
Certain prior period balances have been reclassified to conform to the current period's financial statement presentation. These reclassifications had no impact on previously reported financial position, results of operations or cash flows.
NOTE 2 - INVENTORIES
At December 31, 2006, inventories consisted of the following:
| Raw materials | $ | 2,500,564 | ||
| Finished goods | 913,364 | |||
| 3,413,928 | ||||
| Less: Reserve for obsolete inventory | (280,251 | ) | ||
| $ | 3 ,133,677 |
NOTE 3 - PROPERTY AND EQUIPMENT
At December 31, 2006, property and equipment consist of the following:
| Useful Life | |||||||
| Office equipment and furniture | 5-8 Years | $ | 136,609 | ||||
| Manufacturing equipment | 10 - 15 Years | 4,708,531 | |||||
| Building and building improvements | 20 - 40 Years | 2,385,656 | |||||
| 7,230,796 | |||||||
| Less: accumulated depreciation | (1,416,861 | ) | |||||
| $ | 5,813,935 | ||||||
For the year ended December 31, 2006 and 2005, depreciation expense amounted to $397,951 and $251,056, respectively.
F-14
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 4 - INTANGIBLE ASSETS
At December 31, 2006, the Company holds manufacturing rights for two issued drugs. The manufacturing rights issued are in connection with the Company’s products Valsartan and Brimonidine. The manufacturing rights for Valsartan became effective in November 2000 and have a life of 6.5 years. The manufacturing rights for Brimonidine became effective on August 27, 2005 and have a life of 5 years.
At December 31, 2006, intangible assets consist of the following:
| Manufacturing rights | $ | 1,111,097 | ||
| Less: accumulated amortization | (902,928 | ) | ||
| $ | 208,169 |
Amortization expense amounted to approximately $170,000 and $179,000 for the year ended December 31, 2006 and 2005.
Amortization expense attributable to future periods is as follows:
| Year ending December 31: | ||||
| 2007 | $ | 79,309 | ||
| 2008 | 76,751 | |||
| 2009 | 52,109 | |||
| $ | 208,169 | |||
NOTE 5 - RELATED PARTY TRANSACTIONS
Due from related parties
At December 31, 2006, the Company had a receivable from affiliated entity owned by the Company’s chief executive officer. During fiscal 2006, this related party repaid $1,243,742 of these advances and is currently repaying this balance monthly at approximately $84,000 per month until paid in full. At December 31, 2006, the Company has a receivable from this related party amounting to $1,826,626. These advances are payable on demand and were personally guaranteed by the officer. The Company is aware that the receivable from this related party may be in violation of the Sarbanes-Oxley Act of 2002. Through monthly payments, the Company intends on rectifying this situation during fiscal 2007.
F-15
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 5 - RELATED PARTY TRANSACTIONS (continued)
Notes payable - related parties
Notes payable - related parties consisted of the following at December 31, 2006:
| Note to Song Guoan, father of Song Zheng Hong, director and spouse of Liu Zhong Yi, due on December 30, 2015 with variable annual interest at 80% of current bank rate (5.1% at December 31, 2006), and unsecured | $ | 1,880,397 | ||
| Note to Zheng Gui Xin, employee, due on December 30, 2015 with with variable annual interest at 80% of current bank rate (5.1% at December 31, 2006), and unsecured | 1,446,114 | |||
| Note to Ma Zhao Zhao, employee, due on December 30, 2015 with variable annual interest at 80% of current bank rate (5.1% at December 31, 2006), and unsecured | 579,165 | |||
| Note to Liu Zhong Yi, officer and director, due on December 30, 2015 with variable annual interest at 80% of current bank rate (5.1% at December 31, 2006), and unsecured | 568,367 | |||
| Note to Song Zheng Hong, director and spouse of the Company chief executive officer, Liu Zhong Yi, due on December 30, 2015 with variable annual interest at 80% of current bank rate (5.1% at December 31, 2006), and unsecured | 255,837 | |||
Total notes payable - related parties, long term | $ | 4,729,880 |
For the years ended December 31, 2006 and 2005, interest expense related to these related loans amounted to $325,824 and $0, respectively.
Due to related parties
Prior to fiscal 2006, the chief executive officer of the Company and his spouse, from time to time, provided advances to the Company for operating expenses. During fiscal 2006, the Company repaid $302,871 of these advances. At December 31, 2006, the Company had a payable to the chief executive officer and his spouse amounting to $547,481. These advances are short-term in nature and non-interest bearing.
Prior to fiscal 2006, an employee of the Company, from time to time, provided advances to the Company for operating expenses. During fiscal 2006, the Company repaid $378,042 of these advances. At December 31, 2006, the Company had a payable to this employee amounting to $147,750. These advances are short-term in nature and non-interest bearing.
F-16
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 5 - RELATED PARTY TRANSACTIONS (continued)
Other
On October 9, 2006, the Company entered into a five-year loan agreement and a contract with Wu Lan Cha Bu Emergency Hospital (“Wu Lan”), whereby the Company agreed to lend to Wu Lan approximately $3,840,000 for the construction of a hospital ward in Inner Mongolia, China. In exchange for agreeing to lend to Wu Lan the funds, Wu Lan agreed that the Company will be the exclusive provider for all medicines and disposable medical treatment apparatus to Wu Lan for a period of twenty (20) years. In October 2006, the Company’s chief executive officer, Mr. Liu, lent these funds to Wu Lan on behalf of the Company. Accordingly, the Company entered into an assignment agreement whereby the Company assigned all of its rights, obligations, and receipts under the Loan Agreement to Mr. Liu, except for the rights to revenues from the sale of medical and disposable medical treatment apparatus which will remain with the Company. As compensation to Mr. Liu for accepting the assignment under the loan agreement including all of the risks and obligations and for Liu not accepting the rights to revenues from the sale of medical and disposable medical treatment apparatus which will remain with the Company, the Company agreed to pay Mr. Liu an aggregate of approximately $1,150,000 to be paid in 5 equal annual installments of approximately $230,000.
For the year ended December 31, 2006, assignment fee expense, which was paid during fiscal 2006, amounted to $56,378 and is included in general and administrative expenses on the accompanying consolidated statements of operations.
NOTE 6 - OTHER INCOME
In 2005, the Company recorded other income of $1,219,512 related to the sale of a patent to a third party for a drug formula that it had developed.
NOTE 7 - INCOME TAXES
The Company accounts for income taxes under Statement of Financial Accounting Standards No. 109, ‘‘Accounting for Income Taxes’’ (‘‘SFAS 109’’). SFAS 109 requires the recognition of deferred tax assets and liabilities for both the expected impact of differences between the financial statements and the tax basis of assets and liabilities, and for the expected future tax benefit to be derived from tax losses and tax credit carryforwards. SFAS 109 additionally requires the establishment of a valuation allowance to reflect the likelihood of realization of deferred tax assets. Realization of deferred tax assets, including those related to the U.S. net operating loss carryforwards, are dependent upon future earnings, if any, of which the timing and amount are uncertain. Accordingly, the net deferred tax asset related to the U.S. net operating loss carryforward has been fully offset by a valuation allowance. The Company is governed by the Income Tax Law of the People’s Republic of China and the United States.
The Company has a net operating loss carryforward for United States income tax purposes totaling approximately $153,800 at December 31, 2006, expiring through the year 2026 subject to the Internal Revenue Code Section 382, which places a limitation on the amount of taxable income that can be offset by net operating losses after a change in ownership.
The PRC local government has provided various incentives to companies in order to encourage economic development. Such incentives include reduced tax rates and other measures. Since 2002, the Company has been entitled to an income tax exemption for a six-year period and was exempted from substantially all of its income tax in the 2006 and 2005 period. The estimated tax savings due to the tax exemption for the years ending December 31, 2006 and 2005 amounted to approximately $1,400,000 and $603,000, respectively. The net effect on earnings per share if the income tax had been applied would decrease earnings per share for December 31, 2006 and 2005 to $0.03 and $0.02, respectively.
F-17
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 7 - INCOME TAXES (continued)
The table below summarizes the differences between the U.S. statutory federal rate and the Company’s effective tax rate and as follows for years ended December 31, 2006 and 2005:
| 2006 | 2005 | ||||||
| Computed "expected" provision | 34.0 | % | 34.0 | % | |||
| State tax provision, net of federal effect | 4.0 | % | 4.0 | % | |||
| Effect of foreign income tax exemption | (39.0 | )% | (38 | )% | |||
| Change in valuation allowance | 1.0 | % | 0.0 | % | |||
| 0.0 | % | 0.0 | % | ||||
Deferred tax assets and liabilities are provided for significant income and expense items recognized in different years for tax and financial reporting purposes. Temporary differences, which give rise to a net deferred tax asset is as follows:
| 2006 | ||||
| Tax benefit of net operating loss carryforward | $ | 58,444 | ||
| Valuation allowance | (58,444 | ) | ||
Net deferred tax asset | $ | - | ||
After consideration of all the evidence, both positive and negative, management has recorded a valuation allowance at December 31, 2006, due to the uncertainty of realizing the deferred income tax assets related to the U.S. net operating loss carryforward.
Business Tax and Value Added Tax
The Company is subject to value added tax (“VAT”) for manufacturing products and business tax for services provided. The applicable VAT tax rate is 17% for products sold in the PRC, and the business tax rate is 5% for services provided in PRC. The amount of VAT liability is determined by applying the applicable tax rate to the invoiced amount of goods sold (output VAT) less VAT paid on purchases made with the relevant supporting invoices (input VAT). Under the commercial practice of the PRC, the Company paid value added taxes (“VAT”) and business tax based on tax invoices issued. The tax invoices may be issued subsequent to the date on which revenue is recognized, and there may be a considerable delay between the date on which the revenue is recognized and the date on which the tax invoice is issued. In the event that the PRC tax authorities dispute the date of which revenue is recognized for tax purposes, the PRC tax office has the right to assess a penalty, which can range from zero to five times the amount of the taxes which are determined to be late or deficient. According to the PRC tax laws, any potential tax penalty payable on late or deficient payments of this tax could be between zero and five times the amount of the late or deficient tax payable, and will be expensed as a period expense if and when a determination has been made by the taxing authorities that a penalty is due. At December 31, 2006, the Company has accrued $2,047,851 of underpaid business and value-added taxes.
F-18
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 8 - SEGMENT INFORMATION
The following information is presented in accordance with SFAS No. 131, Disclosure about Segments of an Enterprise and Related Information. In the year ended December 31, 2006 and 2005, the Company operated in two reportable business segments - (1) the manufacture and distribution of pharmaceutical products and (2) the retailing of traditional and Chinese medicines and supplies through ten drug stores located in Beijing China and other ancillary revenues generated from retail location such as advertising income, rental income, and examination income. The Company's reportable segments are strategic business units that offer different products. They are managed separately based on the fundamental differences in their operations.
Information with respect to these reportable business segments for the year ended December 31, 2006 and 2005 is as follows:
Wholesale and third-party manufacturing | Retail Operations | Unallocated | Total | ||||||||||
2006 | |||||||||||||
Net Revenues | $ | 30,285,471 | $ | 4,800,553 | $ | 1,121,665 | $ | 36,207,689 | |||||
Cost of Sales (excluding depreciation) | 21,807,867 | 4,309,542 | 29,780 | 26,147,189 | |||||||||
Operating expenses (excluding depreciation and amortization) | - | - | 5,066,470 | 5,066,470 | |||||||||
Depreciation and Amortization | 357,058 | 18,792 | 191,619 | 567,469 | |||||||||
Interest Expense | - | - | 325,824 | 325,824 | |||||||||
Net Income | $ | 8,120,546 | $ | 472,219 | $ | (4,492,028 | ) | $ | 4,100,737 | ||||
2005 | |||||||||||||
Net Revenues | $ | 16,099,036 | $ | 2,246,805 | $ | - | $ | 18,345,841 | |||||
Cost of Sales (excluding depreciation) | 12,487,753 | 1,865,831 | - | 14,353,584 | |||||||||
Operating expenses (excluding depreciation and amortization) | - | - | 2,950,585 | 2,950,585 | |||||||||
Depreciation and Amortization | 212,128 | 18,970 | 198,535 | 429,633 | |||||||||
Other income | - | - | (1,219,673 | ) | (1,219,673 | ) | |||||||
Interest Expense | - | - | - | - | |||||||||
Net Income | $ | 3,399,155 | $ | 362,004 | $ | (1,929,447 | ) | $ | 1,831,712 | ||||
The Company does not allocate research and development, selling and general and administrative expenses to its reportable segments, because these activities are managed at a corporate level.
Asset information by reportable segment is not reported to or reviewed by the chief operating decision maker and, therefore, the Company has not disclosed asset information for each reportable segment. All of the Company’s assets are located in China.
F-19
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 9 - COMMITMENTS AND CONTINGENCIES
Operating Leases
The Company leases office and manufacturing space under leases in Beijing, China that expire through July 2019.
Future minimum rental payments required under these operating leases are as follows:
| Year Ended December 31, | ||||
| 2007 | $ | 241,057 | ||
| 2008 | 212,689 | |||
| 2009 | 184,346 | |||
| 2010 | 95,939 | |||
| 2011 and thereafter | 830,828 | |||
| Total minimum lease payments | $ | 1,564,859 | ||
For the years ended December 31, 2006 and 2005, rent expense amounted to $206,965 and $238,610, respectively.
NOTE 10 - OPERATING RISK
(a) Country risk
Currently, the Company’s revenues are primarily derived from the sale of pharmaceutical products to customers in the Peoples Republic of China (PRC). The Company hopes to expand its operations to countries outside the PRC, however, such expansion has not been commenced and there are no assurances that the Company will be able to achieve such an expansion successfully. Therefore, a downturn or stagnation in the economic environment of the PRC could have a material adverse effect on the Company’s financial condition.
(b) Products risk
In addition to competing with other manufacturers of pharmaceutical product offerings, the Company competes with larger Chinese companies who may have greater funds available for expansion, marketing, research and development and the ability to attract more qualified personnel. These Chinese companies may be able to offer products at a lower price. There can be no assurance that the Company will remain competitive should this occur.
(c) Political risk
Currently, PRC is in a period of growth and is openly promoting business development in order to bring more business into PRC. Additionally PRC allows a Chinese corporation to be owned by a United States corporation. If the laws or regulations are changed by the PRC government, the Company's ability to operate the PRC companies could be affected.
F-20
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 11 - SUBSEQUENT EVENT
On February 12, 2007, the Company entered into Subscription Agreements with the purchasers (the "Purchasers"). Pursuant to the Agreements, the Company issued to the Purchasers Secured Convertible Notes, ("Notes"), with an aggregate principle value of $3,000,000, plus interest on the unpaid principal balance at a rate equal to 14.0% per annum. The maturity date of the Notes is February 12, 2008. Interest on the Notes commences accruing on February 12, 2007 and is payable on April 30, 2007 and on the last business day of each calendar quarter thereafter. Interest is payable in cash, except that upon notice to the Purchasers in writing not less than 15 trading days prior to the relevant interest payment date, the Company may deliver common stock that were registered exclusively for the purpose of satisfying the payment of interest and not principal (“Interest Shares”) valued at 75% of the average of the three lowest closing bid prices of the common stock as report by Bloomberg L.P. for the 20 trading days ending on the trading day preceding the relevant interest payment date. In the event of default under the terms of the Notes, at the option of the Purchaser, all sums of principal and interest remaining unpaid will become immediately due and payable. The notes are convertible into shares of common stock of the Company a rate equal to $1.00 per share, subject to adjustment. The transaction closed on February 12, 2007.
The Purchasers also received Common Stock Purchase Warrants (the "Warrants"). Pursuant to the terms of the Warrants, the Purchasers are entitled to purchase up to 1,500,000 shares of common stock of the Company at an exercise price of $1.50 per share. The Warrants have a term of 5 years after the issue date of February 12, 2007.
The Notes are secured by a Security Agreement, a Pledge Agreement, a Guaranty, and Share Pledge Agreements (collectively, the “Security Agreements”). In the event of a default, the Security Agreements grant to S. Michael Rudolph, as collateral agent for the Purchasers, a security interest in all right, title and interest of Debtors, as that term is defined therein, to and in respect of all Accounts, Goods, real or personal property, all present and future books and records relating to the foregoing and all products and Proceeds of the foregoing, as set forth therein, and all security interests, as set forth therein, of the Company, Lotus Pharmaceutical International, Inc. Beijing Liang Fang Pharmaceutical Co., Ltd. and Beijing En Ze Jia Shi Pharmaceutical Co., Ltd. In addition, Liu Zhong Yi, the Company’s president and chief executive officer, pledged his 18,782,400 shares of the Company’s common stock, Song Zhenghong, a director of the Company and spouse of Liu Zhong Yi, pledged her 6,708,000 shares of the Company’s common stock, and the Company pledged its 38,800 shares of Lotus’s common stock in the event of a default under the Notes. Events of default include, but are not limited to, (i) the Company’s failure to pay any installment of principal, interest or other sum due under the Notes when due and such failure continues for a period of 5 business days after the due date, (ii) the commencement of a voluntary or involuntary liquidation, or other relief with respect to the Company or its debts under bankruptcy, insolvency or other similar law, that is not dismissed with 45 days of initiation (iii) final judgments for the payment in excess of $100,000 are rendered against the Company, and remain unpaid, unvacated, unbonded or unstayed for a period of 45 days (iv) or any breach or failure to comply in any respect with the terms of the Notes or any agreements entered into in connection with the transactions contemplated by the Agreements.
The financing was completed through a private placement to accredited investors and is exempt from registration pursuant to Section 4(2) of the Securities Act of 1933, as amended ("Securities Act"). The securities sold in the financing have not yet been registered under the Securities Act of 1933, as amended, and may not be offered or sold in the United States in the absence of an effective registration statement or exemption from registration requirements.
F-21
LOTUS PHARMACEUTICALS, INC. AND SUBSIDIARIES
(FORMERLY S.E. ASIA TRADING COMPANY, INC.)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2006
NOTE 11 - SUBSEQUENT EVENT (continued)
The Company granted registration rights to holders of the Securities. Generally, the Company shall file with the Commission a Form SB-2 registration statement (the “Registration Statement”) (or such other form that it is eligible to use) in order to register the Registrable Securities for resale and distribution under the 1933 Act within forty-five (45) calendar days after the Closing Date (the “Filing Date”), and cause the Registration Statement to be declared effective not later than one hundred and fifty (150) calendar days after the Closing Date (the “Effective Date”). If the Registration Statement is not filed on or before the Filing Date or is not declared effective on or before the Effective Date, then the Company shall deliver to the holder of Registrable Securities, as Liquidating Damages, an amount equal to two percent (2%) for the first day of such Non-Registration Event and two percent (2%) for each thirty (30) days (or such lesser pro-rata amount for any period of less than thirty (30) days) of the Purchase Price of the outstanding Notes. The Company must pay the Liquidated Damages in cash. The Liquidated Damages must be paid within ten (10) days after the end of each thirty (30) day period or shorter part thereof for which Liquidated Damages are payable.
F-22
3,500,000 SHARES
COMMON STOCK
PROSPECTUS
May 7, 2007
You should rely only on the information contained in this prospectus to make your investment decision. We have not authorized anyone to provide you with different information. This prospectus may be used only where it is legal to sell these securities. You should not assume that the information in this prospectus is accurate as of any date other than the date of this prospectus.