UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
Form 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2008
OR
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from ____ to____
Commission file number: 000-52018
VIRTUALSCOPICS, INC.
(Exact name of registrant as specified in its charter)
| DELAWARE | 04- 3007151 |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
| 500 Linden Oaks, Rochester, New York | 14625 |
| (Address of principal executive offices) | (Zip Code) |
(585) 249-6231
(Registrant's Telephone Number, Including Area Code)
SECURITIES REGISTERED PURSUANT TO SECTION 12(b) OF THE EXCHANGE ACT:
Common Stock, $0.001 par value
NASDAQ Capital Market
SECURITIES REGISTERED PURSUANT TO SECTION 12(g) OF THE EXCHANGE ACT:
TITLE OF EACH CLASS:
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
¨ Yes x No |
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act.
¨ Yes x No |
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for past 90 days. Yes x or No ¨.
Indicate by check mark if disclosure of delinquent filers in response to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting” in Rule 12b-2 of the Exchange Act.
Larger accelerated filer ¨ | Accelerated filer ¨ |
Non-accelerated filer ¨ (Do not check if a smaller reporting company) | Smaller reporting company x |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ or No x
The aggregate market value of the issuer’s voting and non-voting common equity held by non-affiliates of the issuer as of as of February 27, 2009 was approximately $8,743,655 (calculated by excluding all shares held by executive officers, directors and holders known to the registrant of five percent or more of the voting power of the registrant's common stock, without conceding that such persons are “affiliates” of the registrant for purposes of the federal securities laws). This amount does not include any value for the issuer’s series A preferred stock or series B preferred stock, for which there is no established United States public trading market, or any value for the common stock issuable upon conversion of shares of such preferred stock.
As of February 27, 2009, there were outstanding 23,494,893 shares of the issuer’s common stock, $.001 par value.
Documents Incorporated By Reference: Portions of the Company's Proxy Statement to be delivered to the Company’s stockholders in connection with the Company’s 2009 Annual Meeting of Stockholders, which the Company plans to file with the Securities and Exchange Commission pursuant to Regulation 14A promulgated under the Securities Exchange Act of 1934, on or prior to April 30, 2009, are incorporated by reference in Part III (Items 9, 10, 11, 12 and 14) of this Form 10-K.
TABLE OF CONTENTS
| Page Numbers | ||
| PART I | ||
| ITEM 1: Business | 4 | |
| ITEM 1A: Risk Factors | 14 | |
| ITEM 2: Properties | 18 | |
| ITEM 3: Legal Proceedings | 18 | |
| ITEM 4: Submission of Matters to a Vote of Security Holders | 18 | |
| PART II | ||
| ITEM 5: Market For Registrant's Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities | 19 | |
| ITEM 7: Management's Discussion and Analysis of Financial Condition and Results of Operations | 20 | |
| ITEM 8: Financial Statements and Supplementary Data | 27 | |
| ITEM 9: Changes in and Disagreements with Accountants on Accounting and Financial Disclosure | 27 | |
| ITEM 9A: Controls and Procedures | 27 | |
| ITEM 9B: Other Information | 28 | |
| PART III | ||
| ITEM 10: Directors, Executive Officers and Corporate Governance | 29 | |
| ITEM 11: Executive Compensation | 29 | |
| ITEM 12: Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | 29 | |
| ITEM 13: Certain Relationships and Related Transactions, and Director Independence | 29 | |
| ITEM 14: Principal Accountant Fees and Services | 29 | |
| PART IV | ||
| ITEM 15: Exhibits | 29 |
PART I
FORWARD-LOOKING STATEMENTS
Some of the statements under the captions of this report on Form 10-K titled “Risk Factors,” “Management's Discussion and Analysis of Financial Condition and Results of Operations” or “Business,“ contained or incorporated by reference elsewhere in this report, and in our other reports filed with the Securities Exchange Commission (“SEC”) constitute “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements include statements that address activities, events or developments that we expect, believe or anticipate may occur in the future, including:
| · | adverse economic conditions; |
| · | unexpected costs, lower than expected sales and revenues, and operating defects; |
| · | adverse results of any legal proceedings; |
| · | the volatility of our operating results and financial condition; |
| · | inability to attract or retain qualified senior management personnel, including sales and marketing, and scientific personnel; |
| · | inability to raise sufficient additional capital to operate our business, if necessary, and; |
| · | other specific risks that may be referred to in this report. |
All statements, other than statements of historical facts, included in this report regarding our strategy, future operations, financial position, estimated revenue or losses, projected costs, prospects and plans and objectives of management are forward-looking statements. When used in this report, the words “may,” “believe,” “anticipate,” “intend,” “estimate,” “expect,” “project,” “plan,” “could,” “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain such identifying words. All forward-looking statements speak only as of the date of this report. We do not undertake any obligation to update any forward-looking statements or other information contained in this report. Existing stockholders and potential investors should not place undue reliance on these forward-looking statements. Although we believe that our plans, intentions and expectations reflected in or suggested by the forward-looking statements in this report are reasonable, we cannot assure our stockholders or potential investors that these plans, intentions or expectations will be achieved. We disclose important factors that could cause our actual results to differ materially from our expectations under “Risk Factors” and elsewhere in this report. These risk factors qualify all forward-looking statements attributable to us or persons acting on our behalf.
Information regarding market and industry statistics contained in this report is included based on information available to us that we believe is accurate. It is generally based on academic and other publications that are not produced for purposes of securities offerings or economic analysis. We have not reviewed or included data from all sources, and we cannot assure our stockholders or potential investors of the accuracy or completeness of the data included in this report. Forecasts and other forward-looking information obtained from these sources are subject to the same qualifications and the additional uncertainties accompanying any estimates of future market size, revenue and market acceptance of products and services. We have no obligation to update forward-looking information to reflect actual results or changes in assumptions or other factors that could affect those statements. See “Risk Factors” for a more detailed discussion of uncertainties and risks that may have an impact on future results.
ITEM 1: Business
VirtualScopics was formed in 2000 from research first carried out at the University of Rochester. As a result of this research, we have created a suite of image analysis software tools and applications which are used in detecting and measuring specific anatomical structures and metabolic activity using medical images. Our proprietary software and algorithms provide measurement capabilities designed to improve clinical research and development. We are headquartered in Rochester, New York.
Business Overview
We are a provider of quantitative imaging for clinical trials serving the pharmaceutical, biotechnology and medical device industries. We focus on applying our imaging technology to improve the efficiency and effectiveness of the pharmaceutical and medical device research and development processes. We believe our technology can also be used in improving the treatment planning for all patients.
Our image-based measurement and visualization tools enable automated, accurate and reproducible measurement of minute changes that occur in anatomic structures in musculoskeletal, oncological, cardiological and neurological diseases. For pharmaceutical, biotechnology and medical device manufacturers, these tools can significantly alleviate or reduce clinical development bottlenecks by dramatically increasing the speed, accuracy and reliability of the demonstration of a new compound’s efficacy. Further, these measurements can be used to assess the viability of continuing a drug development project and eliminate as soon as possible a drug that is doomed to fail. Early failure is critical to the pharmaceutical industry to prevent the expenditure of limited R&D funds on a drug that will not perform as expected. We believe that this is especially important today with the large number of compounds that are awaiting evaluation.
Our technology may also be applicable in the development of products for patient treatment and surgical planning. During 2009, we plan to explore the opportunities to pursue the market for our services within patient diagnostics. We believe the broadest demand for our services within patient diagnostics will be in Oncology but we will continue to pursue opportunities in cardiology, neurology, arthritis and other diseases. Diagnostic use of our services has not yet been cleared by the U.S. Food and Drug Administration.
Additionally, in 2007, we received two awards totaling approximately $2.1 million for the application of our technology with the United States Department of Defense in the area of oblique hyperspectral imagery, we anticipate concluding the work under these contracts by the end of 2009. In 2008 and 2007, we recognized approximately $860,000 and $671,000, respectively, in revenues related to our work with the Department of Defense.
Benefits to Pharmaceutical, Biotech and Medical Device Companies
The benefits to pharmaceutical companies from using our image analysis tools can include shorter clinical development time, and earlier determination of the effectiveness or ineffectiveness of a new drug or compound. Our technology helps to curtail trials that are not likely to be beneficial and to avoid mistaken termination of compounds that are likely to prove efficacious, through:
| · | improved precision in the measurement of existing biomarkers resulting in shorter observation periods, with beneficial cost savings within a clinical trial; |
| · | new biomarkers, which are better correlated with disease states, again reducing trial length and therefore costs; and |
| · | reduced processing time for image data analysis through automation. |
In addition, our technology reduces aggregate clinical development costs through:
| · | improved precision of existing biomarkers, thus requiring smaller patient populations and lower administrative costs; and |
4
| · | new biomarkers that serve as better correlates, leading to better early screening and elimination of weak drug candidates in pre-clinical trials. |
Benefits to Patients and Health Care Providers in a Diagnostic Setting
The specific diagnostic opportunities that we may pursue in the future are mostly related to the treatment monitoring of patients. Cancer is a leading cause of death throughout much of the developed world, and technologies for closely monitoring disease progression and response to treatment are currently lacking. We believe this presents us with a significant market opportunity.
In treatment monitoring our technology is designed to offer physicians and medical insurers better treatment planning of patients based on determination of patient response to compounds or other treatment options, for example, in oncology we have demonstrated the ability to determine whether patients are showing response to an anti-angiogenic drug after only 48 hours of treatment (Glenn Liu et al., “Dynamic Contrast-Enhanced Magnetic Resonance Imaging As a Pharmacodynamic Measure of Response After Acute Dosing of AG-013736, an Oral Angiogenesis Inhibitor, in Patients With Advanced Solid Tumors: Results From a Phase I Study,” Journal of Clinical Oncology, vol. 20, August 20, 2005).
We are the first company able to provide blood flow and volume measurements for cancer diagnosis and monitoring in a standardized and consistent way across multiple institutions (Jerry M. Collins, “Imaging and Other Biomarkers in Early Clinical Studies: One Step at a Time or Re-Engineering Drug Development?,” Journal of Clinical Oncology, vol. 20, August 20, 2005). These measurements are vital for assessing patient response to next-generation anti-angiogenic cancer drugs.
Our Technology Solution
Oncology Applications
Automated Measurement of Tumor Structure in Oncology
Rapid determination of drug efficacy depends on precise measurement of tumor structure and function. Yet current practices - direct measurement from films and computer-aided tracing - can be time-consuming, inaccurate and highly variable. Manual approaches often lead to false conclusions when tumors take on abnormal shapes; where a two-dimensional analysis may indicate no change, a three dimensional analysis may show a significant change in tumor volume. The RECIST standard, still the primary imaging endpoint for assessing disease progression or response to treatment in many types of cancer, measures structural changes in tumors through a simple summation of longest diameters, limited to the axial imaging plane. Originally developed for x-ray imaging, it fails to take advantage of the far richer three dimensional information set available with today's imaging technologies.
Our semi-automated, statistically driven feature analysis provides greater precision, higher throughput and less dependence on a particular reader than manual tracing does. In retrospective analysis for a leading pharmaceutical company, our volumetric measurement showed that tumors found to be stable under RECIST actually were growing significantly. With our semi-automated analysis we could have discovered the failure sooner and avoided the expense of funding the next phase of clinical research. Conversely, volumetric measurement can greatly accelerate clinical research by preventing mistaken kills and identifying efficacious compounds sooner.
Innovation in Image-Based Biomarkers
With a multidisciplinary team of medical professionals (including staff radiologists), scientists and software developers, we deliver unparalleled innovation in the analysis of specific biomarkers. Measurements may include specific FDA-accepted (RECIST and tumor volume) biomarkers as well as secondary or exploratory endpoints such as cavitation/necrosis, or shape. By extracting substantially more information from existing imaging modalities such as CT or MRI, we believe we offer a more definite and efficient basis for determining the course of clinical trials.
Measurement of Blood Flow and Metabolic Activity
A growing number of anti-cancer drugs both on the market (e.g., Iressa and Avastin) and under development are designed to reduce the blood supply available to tumors, thereby depriving them of the ability to grow and spread. During development, these compounds require the ability to accurately measure blood flow and vascular permeability in vivo, in order to determine dose-response relationships and compound efficacy. In the clinic, this same capability is necessary in order to determine whether a particular patient is responding to treatment. We have developed a method, using dynamic contrast enhanced magnetic resonance imaging (DCE-MRI), to accomplish this. This technique involves repeated imaging, generally every five to ten seconds, for a period of several minutes before and after the injection of a gadolinium-based, FDA-approved, contrast agent. Tracer concentration changes over time can then be measured both in normal and cancerous tissues, and based on this information parameters such as blood flow, blood volume and vascular permeability can be derived. These parameters have been shown to relate directly to the activity of anti-angiogenesis and anti-vascular cancer drugs, and to allow the prediction of response or failure after only a few days of treatment.
5
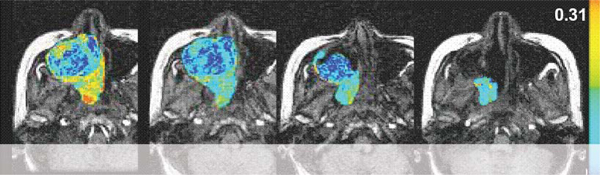
With dynamic contrast-enhanced series, changes in signal intensity can be related to tracer concentration in tissues. This information can be used to determine the blood flow to the tumor.
Musculoskeletal Applications
Our image analysis provides a degree of accuracy and reproducibility that cannot be duplicated by manual techniques. Standard endpoints, such as pain or functionality scoring are largely subjective and difficult to reproduce. Our quantitative imaging replaces subjective evaluation - knee pain ranked on a scale of 1 to 10 - with an objective quantification - volume of lost cartilage in cubic millimeters. Unlike manual assessment methods, our computer aided approach allows you to track the boundary location of each structure in a data set from one scan to another, even if the patient is not positioned in precisely the same way for each scan, or if there have been some anatomical changes between scans. For cartilage volumes and thickness measurements, the Coefficient of Variation (CV) typically falls between 2% and 4% - we can detect minute changes with statistical confidence, allowing you to reduce study populations or shorten study durations.
With our automated analysis, researchers can more confidently make the go/no go decision for a compound early in the evaluation process, allowing scarce resources to be allocated to the most promising candidates. In the evaluation of osteoarthritis, for example, MRI of the cartilage in the knee coupled with automated measurement of volume and composition shows disease changes in months; these changes would not be apparent for years using standard x-ray evaluation.
Reproducible medical image analysis is driven by computer image analysis algorithms that enable quantitative measurement of different structural parameters. Guided by the information present in the images, as well as embedded anatomical knowledge, the algorithms enable segmentation of different structures. From an MRI knee scan, for instance, it is possible to produce a three-dimensional reconstruction that graphically distinguishes cartilage from underlying bone, as well as from ligaments, fluid, degenerated menisci or inflamed synovium. This capability provides a valuable assessment tool for clinical research in osteoarthritis - a disease with multiple endpoints - because it allows sensitive and specific measurement of all the components of the knee joint and detects small changes in any of those components over time.
Medical Device and Biologics
New research continues to focus on the development of devices and/or biologics that will generate new and better cartilage for patients with osteoarthritis and knee injuries. Our technology uses a suite of tools to assist in the identification of cartilage lesions within the knee. These tools allow for the tracking of structural changes and the quality of new tissue being grown within those lesions. For example, we are currently working with leading biologic and medical device companies to determine the percent fill for lesions implanted with the device/biologic. This analysis serves as a useful tool in that it demonstrates the degree of success of the implant. It is presumed in the industry that the higher the percent fill the lower the degree of pain for the patient. We also provides quality of tissue assessments (i.e. T2 maps) to provide our customers with information on the composition of the repair tissue. It is also believed that the closer the repair tissue is to ‘normal’ tissue the longer the life span is of the repair tissue with the resulting benefit being the ultimate health and comfort of the patient.
6
Additionally, our motion tracking software capabilities allows us to more precisely measure changes in the structural and tissue quality measurements. It has been demonstrated that this technique can reduce the amount of variability inherent in these types of measurements, thereby, reducing the amount of patients necessary to demonstrate the effectiveness of the medical device and/or biologic.
Cardiovascular Applications
Cardiovascular disease is one of the leading causes of mortality within most developed countries. Early identification of the changes leading the disease can prompt early intervention which can result in longer and better quality of life as well as lower healthcare costs. Imaging provides a valuable tool for the assessment of early development of arterial plaques which can lead to arterial stenosis as well as stroke and myocardial infarction. The current primary imaging tool for screening cardiovascular patients is ultrasound but these carotid ultrasound scans produce a large amount of data which can be laborious and imprecise to analyze. We have developed a suite of patented semi-automated tools for the identification and measurement of carotid plaques which has proven to reduce analysis time to as little as 3 minutes per case compared to the current manual methods which can take over one hour. In addition, these tools have been tested against expert readers in the field and found to be highly precise and accurate and in many cases more sensitive to the appearance of small arterial plaques. This provides a valuable tool for screening of normal/healthy individuals as well as monitoring the use in patients enrolled in clinical trials.
More detailed information about plaque composition and progression can be obtained by using MRI. This modality has advantages over ultrasound in that it can precisely measure plaque volume as well as composition. This is important because it is widely recognized in the industry that certain plaques, in particular those with high lipid or necrotic cores, pose a much higher risk to the health of the patient, while those that are fibrous may pose a lower risk. Therefore, the ability to distinguish between benign and vulnerable plaques may enable treating physicians to better personalize the treatment for each patient. Additionally, certain drugs designed to reduce blood lipids may have greater effect on lipid rich plaques, making this a potentially beneficial screening tool for patients enrolled in clinical trials. Our patented semi-automated tools for the measurement of plaques in MRI and automated identification of lipids and calcification allows accurate and precise analysis of vulnerable plaques.
These proprietary ultrasound and MRI techniques for cardiovascular health are being used in large industry sponsored trials today.
Neurology Applications
Evaluating diseases such as multiple sclerosis (MS), epilepsy, and Alzheimer’s requires the identification and measurement of neurological structures and lesions. However, current methods for obtaining data points rely on subjective and error prone manual techniques. Manual tracing, especially of abnormal neurological structures, requires considerable expertise and time. Tracing introduces significant variability even when all measurements are made by one individual, an effect that is compounded with multiple operators. Intra- and inter-operator variability poses a major obstacle for researchers attempting to take advantage of the power of MRI analysis in the study of neurological disease. VirtualScopics eliminates these problems with automated, statistically driven feature analysis. Our algorithms employ the two types of knowledge that expert radiologists use to measure structures within the brain: differentiation of various tissue types and knowledge of structure, size, location, and shape. Our software incorporates an a priori model of neurological anatomy that enables the measurement of structures with indistinct boundaries such as the hippocampus. Knowledge of anatomical structures also improves reproducibility, allowing disease progression to be precisely monitored over time. To gain higher resolution and superior tissue separability, we reconstruct volumes by co-registering and fusing images from multiple imaging planes and pulse sequences. Moreover, its automatic reconstruction produces a smooth and continuous surface, much closer to actual shape than would result from manual segmentation.
7
Many neurological conditions can be detected and evaluated with quantitative measures of structures in MRI studies. While automated measurement tracks lesions in MS clinical trials, it also provides a critical tool in measuring hippocampal volume for diagnosing and monitoring both intractable temporal lobe epilepsy and Alzheimer’s disease. Validation studies prove that our automated approach provides greater speed, precision and accuracy in clinical trials than current manual methods do. In MS clinical trials, where current techniques to measure progress in drug development are largely manual, we provide an FDA-approved metric for quickly determining drug efficacy of MS compounds. A VirtualScopics validation study compared manual tracing using two VirtualScopics software algorithms for automated measurement: geometrically constrained region growth (GEORG) and directed clustering. Our Core Lab utilizes both algorithms to achieve an optimal system for quantification of MS lesions in multi-spectral MRI studies. In the MS validation study, mean processing time was 60 minutes for manual tracing, 10 minutes for GEORG, and 3 minutes for directed clustering. Intra- and inter-operator coefficients of variation were 5.1% and 16.5% for manual tracing, 1.4% and 2.3% for region growth and 1.5% and 5.2% for directed clustering. The study also compared our automated measurement and manual tracing from an expert radiologist against a phantom data set, obtained from the McConnell Brain Imaging Center. In all data sets, automated algorithms performed significantly better than manual tracing. Our automated measurements also proved more repeatable than manual methods, an important feature in multi-center clinical trials.
Sales and Marketing
Our sales and business development strategy is centered around the publication and presentation of our technology at targeted industry events along with an active calling effort aimed at pharmaceutical companies, medical device, biotechnology companies, principal investigators and clinical research organizations, (CROs). To date, we have made significant inroads by having contracts with 12 of the 15 leading pharmaceutical, biotechnology and medical device companies. During 2008, we had 33 active customers. We continue to grow our business by leveraging relationships with our current customers and referrals. As a result, our current customers have begun introducing us to other therapeutic groups within their organization. We continue to receive positive feedback from our customers, which has resulted in new projects.
In March 2004, we entered into two agreements, a consulting services agreement and co-marketing agreement with Chondrometrics GmbH and a consulting agreement with Dr. Felix Eckstein, the founder of that company. Dr. Eckstein is an internationally recognized authority in osteoarthritis research, especially as it relates to cartilage degeneration. These agreements were intended to leverage the research activities between us and Dr. Eckstein and co-market each party’s services. The consulting agreement expired on December 31, 2006 whereas the co-marketing agreement expired on December 31, 2008.
Complementing our sales and marketing effort, we actively participate in medical conferences to showcase our technology, as well as collaborating with principal investigators on their academic research, which often results in highly visible, published research. We have built a strong base of clinical collaborators across varied disease platforms.
We are continuing an active sales and marketing effort and are currently in discussions with a number of additional potential customers to form business and/or strategic alliances.
Pfizer Strategic Alliance
In July 2002, we entered into a multi-year strategic alliance under a clinical imaging and services agreement with Pfizer, which was expanded and renewed for two years in July 2005, and again in November 2006, to accelerate the discovery, validation and application of image-based biomarkers for clinical research. As part of the original agreement, Pfizer made an equity investment in VirtualScopics which is now represented by our common stock that Pfizer received in the exchange transaction. Pursuant to the terms of the agreement with Pfizer, we granted Pfizer a worldwide, non-exclusive, perpetual, royalty-free license to use, reproduce and modify “tool boxes” that we develop for Pfizer using our image analysis tools technology and that Pfizer will use for the research and development of its pharmaceutical products. The relationship enables Pfizer to apply our technology to ongoing clinical research in an effort to identify and validate biomarkers correlating to clinical outcomes. The biomarkers may then be used to assess the efficacy of new pharmaceutical compounds in the clinical trial process. The alliance continued until July 2008, and was automatically renewed.
We derived 9% and 28% of our revenue from Pfizer for the years ended December 31, 2008 and 2007, respectively. During 2008, we experienced a decline in the amount of projects from Pfizer as a result of their reorganizations as well as their stated intentions to not continue investments in the osteoarthritis field. As a result, our revenues with Pfizer have declined but the demand for our services in other therapeutic areas, largely oncology, have seen significant increases, thereby offsetting any impact from our Pfizer projects.
8
Industry Background and Market Trends
Market in Pharmaceutical and Medical Device Development
We estimate the current market for image analysis in clinical drug trials is approximately $500 million and is growing at over a 10 - 20% annual rate. The market is expected to exceed $1.0 billion annually by 2014. The use of MRI, CT and PET imaging to determine efficacy of drugs is expected to continue rapid growth.
Growth is a function of:
| · | the FDA’s desire to use MRI, CT and PET imaging to determine efficacy due to its non-invasive nature; |
| · | the pharmaceutical industry’s desire to accelerate the time to market and reduce the cost of clinical drug trials; and |
| · | an increase in the number of drugs undergoing clinical trials owing to significant and growing pharmaceutical R&D. |
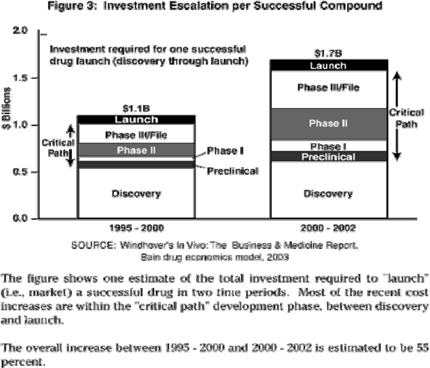
The table below outlines the estimated cost to develop a successful compound, from research to launch.
Quantitative Image Analysis Services
We have conducted research to determine the current size of the market for image analysis services in clinical trials supporting the pharmaceutical, biotech and medical device industries. The information was collected as part of our discussions with many of the major companies that currently serve the industry as well as with the assistance of Dawnbreaker, a strategic/business planning firm. Our discussions with companies in the industry and academic centers included Bio-Imaging Technologies, Synarc, Beacon Bioscience, Perceptive, Duke Image Analysis Laboratories and Boston University, as well as a number of other academic centers in the United States and Europe. We have found that the market is fragmented, with approximately $500 million in total annual revenues projected for 2008.
9
The industry is currently undergoing a rapid growth phase as the use of imaging end-points is becoming more accepted by the FDA and the number of compounds being tested by pharmaceutical companies continues to increase. We estimate the annual growth rate for the market at 10% to 20% for the next five years. Our estimates are based on a bottom-up calculation of the individual growth rates of the companies and academic centers within the industry. We believe that some of the largest players, which offer the broadest set of capabilities, are growing even faster. Specifically, Bio-Imaging Technologies, Perceptive (division of Parexel), Synarc and ICON.
Image Analysis Solutions in the Pharmaceutical and Medical Device Industries
It is well known that greater reproducibility of measurements can decrease the cost and time to market of compounds in development. The higher reproducibility of our automated analysis enables researchers to achieve statistically significant results with substantially smaller patient populations. Automated analysis greatly reduces the analyst variability and interaction time required to process clinical trial data. Published studies demonstrate that our automated analysis consistently yields a lower coefficient of variation than manual techniques. As measurement variation diminishes, so does the percentage change in a given structure necessary to determine whether a treatment is having the desired effect. In short, precise measurement allows companies to learn more from smaller populations earlier in the compound development process.
Drug discovery and development has been constrained by the lack of accurate image analysis tools and appropriate image-based biomarkers. In many musculoskeletal clinical studies, X-ray is the chosen modality for evaluating a compound’s efficacy. X-ray imaging in drug discovery has significant limitations, which include:
| · | partial or complete inability to detect changes in a region of interest due to poor contrast or occlusion; |
| · | the potential for inter/intra-observer variability - error in radiologist measurements can amount to upwards of 30% for small structures of interest; |
| · | the need for a radiologist to perform manual tracings is not only subject to error, but is also time consuming; and |
| · | reliance on a radiologist for biomarker measurements results in very limited throughput. |
The constraints mentioned above can add months and years to the drug discovery process.
The use of MRI and CT to determine drug efficacy is increasing, owing to its superior information content relative to X-ray. MRI and CT are more sensitive to pathology, provide higher contrast for soft tissue and are three-dimensional. These attributes improve the detection of disease and the ability to monitor disease progression over time. While MRI and CT are preferred modalities, they too suffer from the need to have a radiologist review the images, detect disease, monitor progression and, when necessary, perform manual calculations.
Intellectual Property
We depend on our ability to develop and maintain the proprietary aspects of our technology to distinguish our services from our competitors’ products and services. We consider our proprietary and patented technology and the technology for which we have applied for patent protection to be of importance to our business plan. We hold eleven patents issued by the United States Patent and Trademark Office. We have also applied for a number of other patents, both domestically and in foreign jurisdictions. To protect our proprietary technology, we rely primarily on a combination of confidentiality procedures, copyright, trademark and patent laws. Our policy is to require employees and consultants to execute confidentiality and invention assignment agreements upon the commencement of their relationship with us. These agreements provide that confidential information developed or made known during the course of a relationship with us must be kept confidential and not disclosed to third parties except in specific circumstances and for the assignment to us of intellectual property rights developed within the scope of the employment relationship.
10
Competition
Our competition is largely comprised of a limited number of university research centers that are working on developing the next generation of image analysis tools. Aside from the university centers, there are a few commercial entities that have a desire to provide these advanced imaging services; however, we believe they are constrained by their lack of technical capabilities.
One group of potential competitors is clinical research organizations (CROs) providing clinical trial services to pharmaceutical companies. As of the date of this report, we believe that none of the leading CROs have technology capabilities that are comparable to our technology. CROs typically provide manual and non-differentiated interpretation of medical images for the pharmaceutical industry.
We believe that currently there is an opportunity for us to establish a technology advantage and a set of differentiated services in the advanced image-based biomarker market.
Competitors in Accelerating Pharmaceutical and Medical Device Development
The main CROs which participate in imaging trials are Bio-Imaging Technologies, Synarc, Perceptive and ICON. It is our understanding that these companies use predominately manual approaches that are unable to quantify minute structures in medical images. As a result, it may be difficult for them to offer differentiated services to achieve higher profit margins and at the same levels of reproducibility as ours. Additionally, some academic centers have worked on software that has applications for neurological diseases. These academic centers include Duke Image Analysis Laboratory, University of Pennsylvania, University of Montreal and University of California at San Francisco. However, we believe these organizations lack the required FDA compliance standards and ability to scale their operations to meet customer demand and we believe they offer inferior technology.
We believe CROs recognize the pharmaceutical industry’s desire to have a quicker, less costly and more efficient means of determining efficacy through imaging, but they have not focused on developing the technology. It is highly likely that CROs will obtain the desired capabilities through the acquisition of a company that has developed the necessary image analysis technology. Given that there are a limited number of entities with automated segmentation and measurement technology, an acquisition of such an entity by a CRO would position the CRO at the forefront of a growing market.
Academic institutions such as the University of Pennsylvania, University of Montreal and University of California at San Francisco tend to have more advanced technology than their commercial peers. However, their commercial efforts are constrained by being part of an academic institution.
Government Regulation
Healthcare in the United States is heavily regulated by the federal government, and by state and local governments. The federal laws and regulations affecting healthcare change constantly, thereby increasing the uncertainty and risk associated with any healthcare-related company.
The federal government regulates healthcare through various agencies, including the following:
| · | the Food and Drug Administration, or FDA, which administers the Food, Drug, and Cosmetic Act, or FD&C Act, as well as other relevant laws; |
| · | Centers for Medicare & Medicaid Services, or CMS, which administers the Medicare and Medicaid programs; |
| · | the Office of Inspector General, or OIG, which enforces various laws aimed at curtailing fraudulent or abusive practices, including by way of example, the Anti-Kickback Law, the Anti-Physician Referral Law, commonly referred to as Stark, the Anti-Inducement Law, the Civil Money Penalty Law, and the laws that authorize the OIG to exclude health care providers and others from participating in federal healthcare programs; and |
11
| · | the Office of Civil Rights which administers the privacy aspects of the Health Insurance Portability and Accountability Act of 1996, or HIPAA. |
All of the aforementioned are agencies within the Department of Health and Human Services, or HHS. Healthcare is also provided or regulated, as the case may be, by the Department of Defense through its TriCare program, the Public Health Service within HHS under the Public Health Service Act, the Department of Justice through the Federal False Claims Act and various criminal statutes, and state governments under the Medicaid program and their internal laws regulating all healthcare activities.
FDA
We currently meet the requirements of Good Clinical Practices: Consolidated Guidance, which governs the conduct of clinical trials, and our software complies with the FDA’s Regulation 21 CFR Part 11 (Electronic Records; Signatures) and 21 CFR Part 820.30, which outline the requirements for design controls in medical devices.
The FDA regulates medical devices. A “medical device,” or device, is an article, including software and software associated with another medical device, which, among other things, is intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment or prevention of disease, in man or other animals. Computer software that complements a CT or MRI scan, such as VirtualScopics, is considered a medical device and is therefore subject to FDA regulation. To date, our sales have been to the pharmaceutical and medical device industries to support their clinical trials. We would need to obtain FDA clearance or approval, as discussed below, before using our technology and services for diagnostic or treatment planning in a clinical setting. When we begin pursuing the application of our services in patient diagnostics, no assurance can be given that such clearance or approval would be granted or that it would be granted in a timely manner.
Devices are subject to varying levels of regulatory control, the most comprehensive of which requires that a clinical evaluation be conducted before a device receives approval for commercial distribution. In the United States, we generally are able to obtain permission to distribute a new device in two ways. The first applies to any new device that is substantially equivalent to a device first marketed prior to May 1976. In this case, to obtain FDA permission to distribute the device, we generally must submit a premarket notification application (a section 510(k) submission), and receive an FDA order finding substantial equivalence to a device (first marketed prior to May 1976) and permitting commercial distribution of that device for its intended use. A 510(k) submission must provide information supporting its claim of substantial equivalence to the predicate device.
If clinical data from human experience are required to support the 510(k) submission, these data must be gathered in compliance with investigational device exemption (IDE) regulations for investigations performed in the United States. The 510(k) process is normally used for software products of the type that we propose distributing. The FDA review process for premarket notifications submitted pursuant to section 510(k) takes on average about 90 days, but it can take substantially longer if the agency has concerns, and there is no guarantee that the agency will “clear” the device for marketing, in which case the device cannot be used for diagnosis and distributed in the United States. Nor is there any guarantee that the agency will deem the article subject to the 510(k) process, as opposed to the more time-consuming and resource intensive and problematic, premarket approval, or PMA, process described below.
The second, more comprehensive, approval process applies to a new device that is not substantially equivalent to a pre-1976 product. In this case, two steps of FDA approval generally are required before we can market the product in the United States. First, we must comply with IDE regulations in connection with any human clinical investigation of the device. Second, the FDA must review our PMA application, which contains, among other things, clinical information acquired under the IDE. The FDA will approve the PMA application if it finds there is reasonable assurance the device is safe and effective for its intended use.
Certain changes to existing devices that do not significantly affect safety or effectiveness can be made with in vitro testing under reduced regulatory procedures, generally without human clinical trials and by filing a PMA supplement to a prior PMA. Exported devices are subject to the regulatory requirements of each country to which the device is exported, as well as certain FDA export requirements.
12
After approval or clearance to market is given, the FDA and foreign regulatory agencies, upon the occurrence of certain events, have the power to withdraw the clearance or require changes to a device, its manufacturing process, or its labeling or additional proof that regulatory requirements have been met.
A device manufacturer is also required to register with the FDA. As a result, we may be subject to periodic inspection by the FDA for compliance with the FDA’s Quality System Regulation requirements and other regulations. In the European Community, we are required to maintain certain International Organization for Standardization (ISO) certifications in order to sell product and to undergo periodic inspections by notified bodies to obtain and maintain these certifications. These regulations require us to manufacture products and maintain documents in a prescribed manner with respect to design, manufacturing, testing and control activities. Further, we are required to comply with various FDA and other agency requirements for labeling and promotion. The Medical Device Reporting regulations require that we provide information to the FDA whenever there is evidence to reasonably suggest that a device may have caused or contributed to a death or serious injury or, if a malfunction were to occur, could cause or contribute to a death or serious injury. In addition, the FDA prohibits us from promoting a medical device for unapproved indications.
Privacy Provisions of HIPAA
HIPAA, among other things, protects the privacy and security of individually identifiable health information by limiting its use and disclosure. HIPAA directly regulates “covered entities” (healthcare providers, insurers and clearinghouses) and indirectly regulates “business associates” with respect to the privacy of patients’ medical information. All entities that receive and process protected health information are required to adopt certain procedures to safeguard the security of that information. It is our policy to comply with HIPAA requirements.
Research and Development Costs
We incurred $941,193 and $1,440,312 in research and development costs for the years ended December 31, 2008 and 2007, respectively.
Customers
Three customers accounted for 10% or more of our revenue during the year ended December 31, 2008, two of which also accounted for more than 10% of our revenue during the year ended December 31, 2007. The following table sets forth information as to revenue and percentage of revenue for these years for our three largest customers:
| Years Ended December 31, | ||||||||||||||||
| Customer | 2008 | 2007 | ||||||||||||||
| Novartis | $ | 1,275,720 | (17.9 | )% | $ | 237,839 | (4.2 | )% | ||||||||
| $ | 859,915 | (12.1 | )% | $ | 671,138 | (11.9 | )% | |||||||||
| Abbott | $ | 849,951 | (11.9 | )% | $ | 618,188 | (10.9 | )% | ||||||||
Employees
As of December 31, 2008, we had 53 employees and six contract radiologists. Of our employees, 47 are full-time.
13
ITEM 1A: Risk Factors
You should carefully consider the following risk factors before making an investment decision. If any of the following risks actually occur, our business, financial condition, or results of operations could be materially adversely affected. In such cases, the trading price of our common stock could decline, and you may lose all or part of your investment.
We have a history of operating losses and uncertain future profitability.
VirtualScopics has incurred significant losses from operating activities since it began operations over the last seven years. We also may face risks and difficulties in our business of uncertainties of market penetration, competition, cost increases and delays in achieving business objectives. There can be no assurance that we will succeed in addressing any or all of these risks or that we will achieve future profitability and the failure to do so would have a material adverse effect on our business, financial condition and operating results.
If our products and services do not continue to attract interest from new and existing customers, we may not achieve future growth.
If we are unable to continue to attract interest in the industry for our services, we could fail to achieve future growth which would have a detrimental effect on our business. Our ability to generate revenues is highly dependent on building and maintaining relationships with leading pharmaceutical and biotechnology companies. No assurance can be given that a sufficient number of such companies will increase their demand for our services, thereby limiting the overall market for image-based biomarker services and not enable us to increase our revenue to the extent expected. In addition, the rate of the growth of MRI and CT image-based biomarkers is difficult to predict. Failure to attract and maintain a significant customer base would have a detrimental effect on our business, operating results and financial condition.
The majority of the contracts we have with customers are cancelable for any reason by giving 30 days advance notice.
Our customers typically engage us to perform services for them on a project-by-project basis and are required by us to enter into a written contractual agreement for the work, labor and services to be performed. Generally, our project contracts are terminable by the customer for any or no reason on 30 days’ advance notice to us. If a number of our customers were to exercise cancellation rights, our business and operating results would be materially and adversely affected.
Our operating results will be harmed if we are unable to manage and sustain our growth.
Our business is unproven on a large scale and actual operating margins may be less than expected. If we are unable to scale our capacity efficiently, we may fail to achieve expected operating margins, which would have a material and adverse effect on our operating results.
Our services may become obsolete if we do not effectively respond to rapid technological change on a timely basis.
Our services are new and our business model is evolving. Our services depend on the needs of our customers and their desire to utilize image-related services in drug development, new medical device development and clinical diagnosis and treatment. Since the image-based biomarker industry is characterized by evolving technologies, uncertain technology and limited availability of standards, we must respond to new research findings and technological changes affecting our customers. We may not be successful in developing and marketing, on a timely and cost-effective basis, new or modified products and services, which respond to technological changes, evolving customer requirements and competition. If we are unsuccessful in this regard, our business and operating results could be materially and adversely affected.
14
If we fail to protect our intellectual property, our current competitive strengths could be eroded and we could lose customers, market share and revenues.
Our viability may depend on our ability to develop and maintain the proprietary aspects of our technology to distinguish our service from our competitors’ products and services. To protect our proprietary technology, we rely primarily on a combination of confidentiality procedures, copyright, trademark and patent laws.
We hold United States patents which begin to expire in November 2018 through 2025. We have a number of foreign filings pending, or issued, which cover the technology that is the subject of our United States patents. We also have a number of pending United States patent applications with corresponding foreign filings. No assurance can be given that any of these patents will afford meaningful protection against a competitor or that any patent application will be issued. Patent applications filed in foreign countries are subject to laws, rules, regulations and procedures that differ from those of the United States, and thus there can be no assurance that foreign patent applications related to United States patents will issue. If these foreign patent applications issue, some foreign countries provide significantly less patent protection than the United States. In addition, in limited cases we have contractual relationships which give proprietary rights, including ownership rights, in proprietary technology to parties other than us. The status of patents involves complex legal and factual questions and the breadth of claims issued is uncertain. Accordingly, there can be no assurance that our patents, and any patents that may be issued to us in the future, will afford protection against competitors with similar technology. No assurance can be given that patents issued to us will not be infringed upon or designed around by others or that others will not obtain patents that we would need to license or design around. If others’ existing or future patents containing broad claims are upheld by the courts, the holders of such patents could require companies, including us, to obtain licenses or else to design around those patents. If we are found to be infringing third-party patents, there can be no assurance that any necessary licenses would be available on reasonable terms, if at all.
Despite our efforts to protect our proprietary rights, unauthorized parties may attempt to copy aspects of our products and services or obtain and use information that we regard as proprietary. Unauthorized use of our proprietary technology could harm our business. Litigation to protect our intellectual property rights can be costly and time-consuming to prosecute, and there can be no assurance that we will be able to enforce our rights or prevent other parties from developing similar technology or designing around our intellectual property.
Although we believe that our products and services do not and will not infringe upon the patents or violate the proprietary rights of others, it is possible such infringement or violation has occurred or may occur which could have a material adverse effect on our business.
Portions of our business are reliant upon patented and patentable systems and methods used in our image analysis and related intellectual property. In the event that products and services we sell are deemed to infringe upon the patents or proprietary rights of others, we could be required to modify our products and services or obtain a license for the manufacture and/or sale of such products and services. In such event, there can be no assurance that we would be able to do so in a timely manner, upon acceptable terms and conditions, or at all, and the failure to do any of the foregoing could have a material adverse effect upon our business. Moreover, there can be no assurance that we will have the financial or other resources necessary to enforce or defend a patent infringement or proprietary rights violation action. In addition, if our products and services or proposed products and services are deemed to infringe or likely to infringe upon the patents or proprietary rights of others, we could be subject to injunctive relief and, under certain circumstances, become liable for damages, which could also have a material adverse effect on our business.
We are subject to numerous pharmaceutical, medical device and healthcare industry regulations, which could adversely affect the nature and extent of the products and services we offer.
Many aspects of the pharmaceutical, medical device and healthcare industry are subject to regulation at the federal level. From time to time, the regulatory entities that have jurisdiction over the industry adopt new or modified regulations or take other actions as a result of their own regulatory processes or as directed by other governmental bodies. This changing regulatory environment could adversely affect the nature and extent of the services we are able to offer.
15
To date, our sales have been within the clinical trial industry. To significantly expand our business, we would need to obtain FDA clearance or approval before marketing our products into patient diagnostics. There can be no assurance that such clearance or approval would be granted or that it would be granted in a timely manner. To effectively market our products to physicians as a diagnostic and treatment aid, we would also need to obtain appropriate coverage and favorable reimbursement from third-party payors, such as Medicare. There can be no assurance that appropriate coverage would be granted or that reimbursement levels or conditions of coverage would be adequate to ensure acceptance among physicians.
We may in the future experience competition from academic sites and imaging CROs.
Competition in the development of image-based biomarker technologies may become more widespread. Competitors range from university-based research and development projects which would develop advanced tools to development stage companies and major domestic and international companies which would commercialize the tools. Many of these entities have financial, technical, marketing, sales, distribution and other resources significantly greater than ours. There can be no assurance that we can continue to develop our technologies or that present or future competitors will not develop technologies that render our image-based biomarker industry obsolete or less marketable or that we will be able to introduce new products and product enhancements that are competitive with other products marketed by industry participants.
We have experienced significant demand from one customer, thereby increasing our dependence on the customer until we can further diversify our customer base.
While we continue to serve a broad range of customers (33 customers serviced in 2008), because of the strong demand we’ve experienced from one of our customers, our dependence on the customer to sustain our continued growth has increased. We continue to see increasing demand from other customers, however, not to the same significance. We continue to invest on our sales and marketing efforts to further diversify our customers and more broadly penetrate the market, in order to minimize reliance on any one customer. As with all of our contracts, this customer may terminate its contractual relationship with us for any or no reason on 30 days’ advance notice. A decision by the customer to cancel all of its studies with us could have an adverse impact on the growth of our business.
The trading price of our stock may be adversely affected if we are not able to expand.
We intend to continue to use our cash on hand to broaden our market penetration of our technology within the industry. If our plans or assumptions with respect to our business change or prove to be inaccurate, we may be required to use part or all of the net proceeds to fund general operating expenses and/or reduce costs within the organization. This will depend on a number of factors, including, but not limited to:
| · | our ability to successfully market our products; |
| · | the growth and size of the image-based biomarker technology industry; |
| · | the market acceptance of our products and services; and |
| · | our ability to manage and sustain the growth of our business. |
We currently do not plan to raise additional capital, however, if we need to raise additional capital, it may not be available on acceptable terms, or at all. Our failure to obtain required capital, or the acquisition of capital on less favorable terms, would have a material adverse effect on our business. If we issue additional equity securities in the future, you could experience dilution or a reduction in priority of your securities.
The market price of our common stock may fluctuate significantly.
The market price of our common stock may fluctuate significantly in response to factors, some of which are beyond our control, such as the announcement of new products or product enhancements by us or our competitors; developments concerning intellectual property rights and regulatory approvals; quarterly variations in our competitors’ results of operations; changes in earnings estimates or recommendations by securities analysts; developments in our industry; product liability claims or other litigation; and general market conditions and other factors, including factors unrelated to our own operating performance.
16
Our common stock may be considered a “penny stock” and may be difficult to sell.
The SEC has adopted regulations which generally define “penny stock” to be an equity security that has a market or exercise price of less than $5.00 per share, subject to specific exemptions. The market price of our common stock is currently below $5.00 per share and therefore may be designated as a “penny stock” according to SEC rules. This designation requires any broker or dealer selling these securities to disclose certain information concerning the transaction, obtain a written agreement from the purchaser and determine that the purchaser is reasonably suitable to purchase the securities. These rules may restrict the ability of brokers or dealers to sell our common stock and may affect the ability of our stockholders to sell their shares.
Our common stock has traded at prices below $1.00 and is subject to delisting by NASDAQ.
As of the date of this report, the Company is out of compliance with section 4310(c)(8) of the Nasdaq Marketplace Rules, the minimum bid price requirements. Nasdaq has continue to extend the deadline due to general market conditions and the Company now has until August 11, 2009 to regain compliance by maintaining a bid price of our common stock at close of $1.00 or higher for a minimum of 10 consecutive business days, or such longer period as Nasdaq may determine to show the ability to maintain long-term compliance. There can be no assurance that we will be able to do so, or, maintain compliance with this or other listing requirements. The Company intends to take steps necessary to maintain its listing status on Nasdaq, however, if our common stock is delisted from Nasdaq, trading in our common stock could be conducted on the OTC Bulletin Board or in the over-the-counter market in what is commonly referred to as the "pink sheets." If this occurs, a shareholder will find it more difficult to dispose of our common stock or to obtain accurate quotations as to the price of our common stock. Lack of any active trading market would have an adverse effect on a shareholder's ability to liquidate an investment in our common stock easily and quickly at a reasonable price. It might also contribute to volatility in the market price of our common stock and could adversely affect our ability to raise additional equity or debt financing on acceptable terms or at all.
A significant number of the shares of our common stock are eligible for sale, and their sale could depress the market price of the our common stock.
Sales of a significant number of shares of our common stock in the public market or the possibility of such sales, could harm the market price of our common stock and impede our ability to raise capital through the issuance of equity securities. As of February 27, 2009, we had 23,494,893 shares of common stock outstanding. These shares are eligible for resale in the public market either immediately or subject to applicable volume, manner of sale, holding period and other limitations of Rule 144, except for approximately 16,050,914 shares which are subject to additional restrictions under lock-up agreements from our November 2005 private placement. These lock-up agreements provide for periodic releases of shares through October 2009. In addition to these outstanding shares of common stock, the series B convertible preferred stock and the warrants to purchase common stock issued in our 2007 private placement are initially convertible into 5,680,153 shares of our common stock and registered for resale under a registration statement on Form S-3. There are also 6,416,506 shares of our common stock issuable upon conversion of our series A convertible preferred stock and warrants sold in our November 2005 private placement, eligible for resale under Rule 144. Additionally, we have filed a registration statement on Form S-8 to register the sale of 2,500,000 shares issued pursuant to our 2006 Long-Term Incentive Plan. Finally, we have approximately 4.8 million shares of common stock underlying options issued under our 2001, 2005 and 2006 Long-Term Incentive Plans that may be eligible for resale in the public market pursuant to an exemption under Rule 701 of the Securities Act. Sales of our common stock in the public market may have a depressive effect on the market for the shares of our common stock.
Our principal stockholders have significant voting power and may take actions that may not be in the best interests of other stockholders.
Our officers, directors, principal stockholders (greater than 10%) and their affiliates control approximately 45% of our outstanding voting securities. If these stockholders act together, they will be able to exert significant control over our management and affairs requiring stockholder approval, including approval of significant corporate transactions. This concentration of ownership may have the effect of delaying or preventing a change in control and might adversely affect the market price of our common stock. This concentration of ownership may not be in the best interests of all our stockholders.
17
We do not anticipate paying dividends on our common stock in the foreseeable future, and the lack of dividends may have a negative effect on the stock price.
We currently intend to retain our future earnings to support operations and to finance expansion and meet dividend obligations on our series B convertible preferred stock. In addition, the terms of our series B preferred stock limit our ability to pay dividends to the holders of our common stock. Therefore, we do not anticipate paying any cash dividends on our common stock in the foreseeable future.
ITEM 2: Properties
In July, 2007 we began leasing approximately 19,500 square feet of office space at our corporate headquarters in Rochester, New York. The annual rent under the lease is $360,000, and increases three percent (3%) a year. The rent is paid in two portions: the cash portion of $156,000 annually, paid is equal monthly installments, increasing three percent (3%) annually, and the stock portion is $204,000 annually paid in equal monthly installments, increasing three percent (3%) annually. The stock portion is payable in shares of our common stock. At the option of either the landlord or us, upon 90 days written notice to the other party, the option of paying a portion of an installment of rent in stock may be rescinded and such party may elect to pay or receive, as applicable, such installment, or all future installment payments in cash only. In the event that we elect to rescind the stock portion of rent, then the rate per square foot shall be $18.00, increasing 3% annually. In the event that Landlord elects to rescind the stock portion of rent, then the rate per square foot shall be (i) $16.00 if said election is made during the first 24 months of the term, (ii) $17.00 if said election is made after the 24th month but before the 36th month, and (iii) $18.00 if said election is made after the 36th month. Notwithstanding the foregoing, the conversion to cash only rental shall not require 90 days prior written notice if the stock ceases to be publicly traded; in such event, the conversion shall begin with the next monthly installment of rent due and shall be based upon the rate of $18.00 per square foot increasing 3% annually. During 2008, we had issued 165,007 shares of the company’s common stock ranging from $1.20 to $2.50 per share for a total value of $221,000. As of December 31, 2008, there remains an accrued liability of $88,060 relating to the rent due in company stock. The Landlord has elected to receive their rent in all cash beginning on February 1, 2009. Management believes that the leased property is adequately covered by insurance.
ITEM 3: Legal Proceedings
None.
ITEM 4: Submission of Matters to Vote of Security Holders
None.
18
PART II
ITEM 5: Market For Registrant's Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Our shares of common stock are listed for trading on the NASDAQ Capital Market under the trading symbol “VSCP.” These prices represent inter-dealer quotations without retail markup, markdown or commission and may not necessarily represent actual transactions. Investors should not rely on historical stock price performance as an indication of future price performance.
| Fiscal Year Ended December 31, 2007 | ||||||||
| HIGH | LOW | |||||||
| First Quarter | $ | 2.33 | $ | 1.43 | ||||
| Second Quarter | 1.84 | 1.01 | ||||||
| Third Quarter | 1.80 | 1.06 | ||||||
| Fourth Quarter | 1.40 | 0.80 | ||||||
| Fiscal Year Ended December 31, 2008 | ||||||||
| HIGH | LOW | |||||||
| First Quarter | $ | 1.24 | $ | 0.37 | ||||
| Second Quarter | 0.79 | 0.40 | ||||||
| Third Quarter | 0.75 | 0.36 | ||||||
| Fourth Quarter | 0.60 | 0.37 | ||||||
As of February 27, 2009, we had approximately 100 registered holders of record of shares of our common stock.
Dividend Policy
We have never declared a cash dividend on our common stock. We intend to retain any earnings to fund future growth and the operation of our business and, therefore, we do not anticipate paying any cash dividends on our common stock in the foreseeable future. In addition, the terms of our series B preferred stock limit our ability to pay dividends to the holders of our common stock. Thereafter, dividends may be paid on our common stock only if and when declared by our board of directors and paid on an as-converted basis to the holders of our series A and series B convertible preferred stock.
Equity Compensation Plan Information
The following table summarizes information, as of December 31, 2008, relating to our equity compensation plans:
Number of Securities to be Issued Upon Exercise of Outstanding Options | Weighted-Average Exercise Price of Outstanding Options | Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans (Excluding Securities Reflected in Column (a) | ||||||||||
| Plan Category | (a) | (b) | (c) | |||||||||
| Equity compensation plans approved by security holders | 5,297,980 | (1) | $ | 1.83 | 20,658 | |||||||
| Equity compensation plans not approved by security holders | 350,000 | (2) | $ | 2.50 | - | |||||||
| Total | 5,647,980 | $ | 1.87 | 20,658 | ||||||||
19
(1) This amount includes shares under the plans of VirtualScopics, LLC, pursuant to the November 2005 reverse acquisition in connection with which we agreed to issue 532,490 shares of our common stock to holders of warrants granted by VirtualScopics, LLC, in exchange for consideration in the form of goods and services. Also pursuant to the transaction, we agreed to issue 2,327,937 shares of our common stock to holders of options granted by VirtualScopics, LLC pursuant to its 2001 Long-Term Incentive Plan and its 2005 Long-Term Incentive Plan. No further options are available for future issuance under these plans. Also included are shares granted under our 2006 Plan approved by security holders in May 2007. Options to purchase 669,640 shares have been cancelled pursuant to their terms and options to purchase 25,000 shares have been exercised. Also in connection with our November 2005 private placement, we issued warrants to purchase 280,000 shares of our common stock to the placement agents for such transaction. As of February 28, 2009, 50,598 shares under the placement agent warrants were exercised. It also includes 67,530 shares of common stock underlying warrants we issued to the placement agent in connection with our September 2007 private placement, which was approved by stockholders in November 2007.
(2) In November 2005, our Board of Directors granted to our Chairman and former CEO, Robert. Klimasewski, an option to purchase 350,000 shares of our common stock at $2.50 per share.
Recent Sales of Unregistered Securities
During the quarter ended December 31, 2008, there were no conversions of the Company’s series A convertible preferred stock and series B convertible preferred stock. The Company did not receive any cash or other consideration in connection with the conversions. Additionally, no commission or other remuneration was paid by the Company in connection with such conversions. The issuance of common stock upon conversions of the series A and series B convertible preferred stock was made in reliance on the exemption provided in Section 3(a)(9) of the Securities Act of 1933, as amended.
Issuer Repurchases of Equity Securities
None.
ITEM 7: Management’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion should be read in conjunction with VirtualScopics’ consolidated balance sheet, and related consolidated statements of operations, consolidated changes in stockholders’ equity and cash flow for the years ended December 31, 2008 and 2007, included elsewhere in this report. This discussion contains forward-looking statements, the accuracy of which involves risks and uncertainties. Our actual results could differ materially from those anticipated in the forward-looking statements for many reasons including, but not limited to, those discussed in “Risk Factors” and elsewhere in this report. We disclaim any obligation to update information contained in any forward-looking statements.
Overview
VirtualScopics, Inc. is a leading provider of imaging solutions to accelerate drug and medical device development. We have developed a robust software platform for analysis and modeling of both structural and functional medical images. In combination with our industry-leading experience and expertise in advanced imaging biomarker measurement, this platform provides a uniquely clear window into the biological activity of drugs and devices in clinical trial patients, allowing our customers to make better decisions faster.
In July 2000, VirtualScopics was formed after being spun out of the University of Rochester. In June 2002, we purchased the underlying technology and patents created by VirtualScopics’ founders from the University of Rochester. We own all rights to the patents underlying its technology. Since our inception, our principal activities have consisted of:
| · | research and development; |
| · | providing imaging related services within the pharmaceutical industry; |
| · | business development of customer and strategic relationships; and |
20
| · | raising capital. |
Revenue over the past seven years has been derived primarily from image processing services in connection with pharmaceutical drug trials. For these services, we have been concentrating in the areas of oncology and osteoarthritis. We have also derived a small portion of revenue from consulting services, and pharmaceutical drug trials in the neurology and cardiovascular areas. We expect that the concentration of our revenue will continue in these services and in those areas in 2009. Revenues are recognized as the MRI and CT images that we process are quantified and delivered to our customers and/or the services are performed.
Once we enter into a new contract for participation in a drug trial, there are several factors that can effect whether we will realize the full benefits under the contract, and the time over which we will realize that revenue. Customers may not continue our services due to performance reasons with their compounds in development. Furthermore, the contracts may contemplate performance over multiple years. Therefore, revenue may not be realized in the fiscal year in which the contract is signed. Recognition of revenue under the contract may also be affected by the timing of patient recruitment and image site identification and training.
Results of Operations
Results of Operations for Year Ended December 31, 2008 Compared to Year Ended December 31, 2007
Revenue
We had revenues of $7,131,000 for the year ended December 31, 2008 compared to $5,647,000 for the year ended December 31, 2007, representing a 26% increase. The increase in revenues is directly related to the growing demand for our services in the industry. We continue to see growing demand for our Oncology analysis services as well as demand within the medical device industry. During 2008 we performed work for 33 customers, representing 90 different projects, in connection with their pharmaceutical drug trials primarily in the fields of oncology and musculoskeletal diseases (osteoarthritis and rheumatoid arthritis) along with various other projects. As of December 31, 2008, we had active projects with 12 of the leading 15 pharmaceutical companies in the world. The majority of the pharmaceutical projects for which we have performed work to date are in pre-clinical, Phase I or Phase II studies. In 2009, we expect that a majority of our work on pharmaceutical trial projects will continue to be focused in Phase II and III. During 2008, we generated approximately $5.0 million in revenues for our customers in Phase I, II, and III, this compares to approximately $3.3 million in 2007, a 52% increase.
Gross Profit
We had a gross profit of $3,099,000 for the year ended December 31, 2008 compared to $1,967,000 for the comparable period in 2007. Our gross profit improved year over year due to the greater amount of work performed on later stage clinical trials, efficiencies made within our operations, and the reduction in work force at the end of 2007. Later stage trials (Phase II/III) typically have more analysis due to the patient population of the study, as a result, we tend to have greater economies of scale because of the repetitive nature of the work we perform. We believe we can continue to generate near 50% gross margins as we scale the business and therefore, we do not anticipate significant additional investments to be made within our operational infrastructure in 2009.
Research and Development
Research and development costs decreased in 2008 by $499,000, or 35%, to $941,000, when compared to 2007. The decrease was largely attributed to the reduction in employees with our research and development area. During 2007, we began seeing heightened demand for our services in later stage clinical trials and less heavily on research projects as our technology and services became more widely used in the industry and less early stage/proof of concept work was necessary. As a larger percentage of our projects were in later stage clinical trials (as opposed to research/pre-clinical studies) less time was required of the research group to work on customer projects. Additionally, we determined at that time that additional internal funding of research projects was not necessary. As a result, at the end of 2007, we realigned our research group to better reflect the market demand for our services. As of December 31, 2008, there were nine employees in our research and development group, this includes the algorithm and software development groups. Our research and development efforts are centered around improving the functionality of our existing algorithms and software platform as well as the refinement of our algorithms to new therapeutic areas.
21
Sales and Marketing
Sales and marketing costs increased in 2008 by $471,000, to $1,220,000, when compared to 2007. The increase was a result of marketing efforts during the first half of 2008. Due to the changing nature of our business and the customers we target, we re-branded our marketing materials in order to have a consistent and representative reflection of the services we provide and the value it generates for our customers. Our previous marketing efforts were centered around the technology as opposed to the benefits for our customers. As of the date of this report, there are four individuals in our sales and marketing department.
General and Administrative
General and administrative expenses for the year ended December 31, 2008 were $3,158,000, a decrease of $596,000 or 16%, when compared to 2007. The decrease is mainly due to the $380,000 decrease in stock compensation expense due to the timing and valuation of the vesting of outstanding stock options. Also contributing to the decrease in general and administrative costs was a reduction in our spending on public and investor relations during 2008 compared to 2007. In 2007, the Company raised $4.35 million in a convertible series B offering which led to higher costs incurred on investor outreach. Legal costs were also lower in 2008 as a result of settling of a legal matter with a former executive in early 2007.
Depreciation and Amortization
Depreciation and amortization charges decreased for the year ended December 31, 2008 by $20,000 or 4%, to $466,000, when compared to 2007. This decrease is attributed to slight declines in the amortization and depreciation of patents and computer equipment. The amortization and depreciation schedules are based on the timing and life of the patent costs and equipment. We continue to invest in our patent portfolio and computer systems, however, do not anticipate significant expenditures necessary in either area to support our current business.
Interest (Expense)/Income, net
Interest income for the year ended December 31, 2008 was $74,000, representing interest derived on the Company’s operating and savings accounts, compared to interest income of $154,000 in 2007. The decrease in interest income was due to lower rates of return earned on our investment accounts. Other expense for the years ended December 31, 2008 and 2007 was $16,000 and $13,000, respectively. This decrease in interest expense was due to quarterly payments on loans from certain VirtualScopics stockholders. The loans were fully paid as of December 31, 2007.
Net Loss
Our net loss for the year ended December 31, 2008 was $2,629,000 compared to a net loss of $4,321,000 for the year ended December 31, 2007. The decrease in our net loss over the prior period was related to higher revenues, better gross margin and reduced spending in research and development, as planned and outlined above.
In accordance with EITF 98-5, “Accounting for Convertible Securities with Beneficial Conversion Features or Contingently Adjustable Conversion Ratios” and EITF 00-27, “Application of Issue No. 98-5 to Certain Convertible Instruments,” the series B convertible preferred stock was considered to have an embedded beneficial conversion feature because the conversion price was less than the fair value of the Company’s common stock at the issuance date. This beneficial conversion feature is calculated after the warrants have been valued with proceeds allocated on a relative value basis. The series B convertible preferred stock was fully convertible beginning on November 23, 2007 and the value of the beneficial conversion feature was recorded as a deemed dividend. As a result, the net loss attributable to common stockholders for the year ended December 31, 2007 was $5,801,980, including series B preferred stock deemed dividend of $1,381,361. No similar beneficial conversion feature was recorded in 2008.
22
Liquidity and Capital Resources
Our working capital as of December 31, 2008 and 2007 was approximately $2,923,000 and $4,239,000, respectively. The decrease in working capital was a result of the $4.35 million private placement of newly issued series B preferred stock that occurred September 2007, thereby increasing the cash position at the end of 2007. The private placement resulted in net proceeds to the company of approximately $3.9 million. A portion of the capital was spent in 2008 on sales and marketing efforts as well as supporting some of the development efforts.
On September 17, 2007, as mentioned above, we completed a private placement of 4,350 shares of series B convertible preferred stock, par value $0.001 per share, and warrants to purchase the Company’s common stock, par value $0.001 per share, for an aggregate purchase price of $4,350,000. At the time of the closing, we were required to restrict cash equal to ten percent of the stated value of the series B convertible preferred stock that are outstanding, or $435,000, for the payment of dividends plus $200,000 for the payment of investor relation services over the next two years. As of December 31, 2007, the Company had $498,799 in cash restricted for dividend payments and investor relations services. As of December 31, 2008, the restricted balance has been fully spent and no further restrictions of our cash on hand exist.
Net cash used in operating activities totaled $846,000 in 2008 compared to $2,814,000 in 2007. This significant decrease in the amount of cash used on operating activities was primarily the result of the increase in our revenues and reduction in expenses in 2008 as compared to 2007. Also impacting the change from prior year was the timing of our receivables, accounts payable and accrued expenses and advance billings from customers, which is reflected in deferred revenue. As we perform the services to our customers, revenue is recognized and offset against deferred revenue.
We invested $127,000 in the purchase of equipment and the acquisition of patents in 2008, compared to $343,000 for the investment of these items in 2007. This decrease largely represents a decrease in furniture and leasehold improvement costs in 2008 due to a relocation of facilities in 2007 which created higher than average costs. We anticipate that our IT related costs will increase in 2009 as we continue to experience greater demand from our customers. During 2008, we incurred $100,000 in patent costs associated with filing costs for intellectual property, as compared to $138,000 in 2007.
Net cash provided by our financing activities in 2008 was $161,000, compared to net cash provided by our financing activities of $3,211,000 in 2007. This decrease was due to the private placement of our series B convertible preferred stock issued in September 2007, as described above.
We currently expect that existing cash and cash equivalents will be sufficient to fund our existing operations for the next 12 months. If in the next 12 months our plans or assumptions change or prove to be inaccurate, we may be required to seek additional capital through public or private debt or equity financings. If we need to raise additional funds, we may not be able to do so on terms favorable to us, or at all. If we cannot raise sufficient funds on acceptable terms, we may have to curtail our level of expenditures and our rate of expansion.
Off Balance Sheet Arrangements
We have no off-balance sheet arrangements, other than the consulting agreements and operating leases (as described in “Contractual Obligations” below) that have or are reasonably likely to have a current or future effect that is material to investors on our financial condition, revenues or expenses, results of operations, liquidity, capital resources or capital expenditures.
Contractual Obligations
The following table summarizes our contractual obligations at December 31, 2008 which we expect to have an effect on our liquidity and cash flow in future periods. (See Item 2: Description of Property for a full description of our lease obligations which includes a portion of the lease to be paid in our common stock.)
23
| Payments Due by Period | ||||||||||||
| Less than | ||||||||||||
| Total | 1 Year | 1-4 Years | ||||||||||
| Operating Leases | $ | 1,146,956 | $ | 309,674 | $ | 837,282 | ||||||
The Company entered into a Services and Co-Marketing Agreement dated March 1, 2004, in which it agreed to pay Chondrometrics GmbH, a German limited liability company, fees equal to approximately 7% of the gross revenues it derived from certain services each year throughout the term of the agreement. The Company was obligated to make minimum payments to Chondrometrics for the first three years of the agreement. Payments made to Chondrometrics in 2008 and 2007 amounted to $15,000 and $37,000, respectively. There was no minimum payment required in 2008. The agreement terminated on December 31, 2008.
Critical Accounting Policies
Our management’s discussion and analysis of our financial condition and results of operations is based on our financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States of America. The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements as well as the revenues and expenses during the reporting periods. We evaluate our estimates and judgments on an ongoing basis. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
Our significant accounting policies are more fully described in Note 2 to our financial statements appearing elsewhere in this report. The following accounting policies are important in fully understanding and evaluating our reported financial results.
Patents
Costs incurred to acquire and file for patents, including legal costs, are capitalized as long-lived assets and amortized on a straight-line basis over the lower of the estimated useful life or legal life of the patent, which is 20 years.
Impairment of Long-Lived Assets
The Company adopted SFAS No. 144 “Accounting for the Impairment or Disposal of Long-Lived Assets.” Long-lived assets held for use are subject to an impairment assessment if the carrying value is no longer recoverable based upon the undiscounted cash flows of the assets. The amount of the impairment is the difference between the carrying amount and the fair value of the asset. Management does not believe that there is any impairment of long-lived assets at December 31, 2008.
Revenue Recognition
The Company applies the revenue recognition principles set forth under the Securities and Exchange Commission Staff Accounting Bulletin No. 104, “Revenue Recognition,” with respect to its revenues from image analysis, consulting and project/data management services, and recognizes revenue when it is realized or realizable and earned. The Company considers revenue realized or realizable and earned when an agreement exists, services and products are provided to the customer, prices are fixed or determinable, and collectibility is reasonably assured. Revenues are reduced for estimated discounts and other allowances, if any.
The Company provides advanced medical image analysis on a per image basis, and recognizes revenue when the image analysis is completed and delivered to the customer. Revenue related to project, data and site management services is recognized as the services are rendered and in accordance with the terms of the contract. Consulting revenue is recognized once the services are rendered and typically charged as an hourly rate.
24
Occasionally, the Company provides software development services to its customers, which may require significant development, modification, and customization. Software development revenue is billed on a fixed price basis and recognized upon delivery of the software and acceptance by the customer on a completed contract basis in accordance with the American Institute of Certified Public Accountants Statement of Position 81-1, “Accounting for Performance of Construction-Type and Certain Production-Type Contracts.” The Company does not sell software licenses, upgrades or enhancements, or post-contract customer services.
Reimbursements received for out-of-pocket expenses incurred are reported as revenue in the financial statements in accordance with Emerging Issues Task Force No. 01-14, “Income Statement Characterization of Reimbursements received for ‘Out-of-Pocket’ Expenses Incurred.”
Research and Development
Research and development expenses consist of costs incurred to further our research and development activities and include salaries and related employee benefits, consultants and research-related overhead expenses. Research and development costs are expensed as incurred.
Stock-Based Compensation
On January 1, 2006, the Company adopted the fair value recognition provisions of SFAS No. 123R, “Share-Based Payment,” using the modified prospective method. Consequently, for the year ended December 31, 2008, the Company’s results of operations reflect compensation expense for new stock options granted and vested under its stock incentive plans during the fiscal year 2008 and the unvested portion of previous stock option grants which vested during the fiscal year 2008.
The Company accounts for its stock-based payments to non-employees under the guidance of Emerging Issues Task Force (“EITF”) 96-18, “Accounting for Equity Instruments that are Issued to Other than Employees for Acquiring or in Connection with Selling Goods or Services,” which states that the transaction should be valued based on the fair value of the services provided or the fair value of the equity received, whichever is more reliably measurable.
Recent Accounting Pronouncements
In February 2007, the Financial Accounting Standards Board (“FASB”) issued SFAS No. 159, “The Fair Value Option for Financial Assets and Financial Liabilities—Including an amendment of FASB Statement No. 115” (“SFAS No. 159”). This Statement permits entities to choose to measure many financial instruments and certain other items at fair value. The objective is to improve financial reporting by providing entities with the opportunity to mitigate volatility in reported earnings caused by measuring related assets and liabilities differently without having to apply complex hedge accounting provisions. This Statement is expected to expand the use of fair value measurement, which is consistent with the Board’s long-term measurement objectives for accounting for financial instruments. SFAS No. 159 is effective as of the beginning of an entity’s first fiscal year that begins after November 15, 2007. Early adoption is permitted as of the beginning of a fiscal year that begins on or before November 15, 2007, provided the entity also elects to apply the provisions of SFAS No. 157. SFAS No. 159 did not have an impact on its consolidated financial statements.
In December 2007, the FASB issued SFAS No. 141R, “Business Combinations” (“SFAS No. 141R”), which replaces SFAS No. 141, “Business Combinations.” SFAS No. 141R establishes principles and requirements for determining how an enterprise recognizes and measures the fair value of certain assets and liabilities acquired in a business combination, including noncontrolling interests, contingent consideration, and certain acquired contingencies. SFAS No. 141R also requires acquisition-related transaction expenses and restructuring costs be expensed as incurred rather than capitalized as a component of the business combination. SFAS No. 141R will be applicable prospectively to business combinations for which the acquisition date is on or after January 1, 2009. SFAS No. 141R would have an impact on accounting for any businesses acquired after the effective date of this pronouncement.
25
In December 2007, the FASB issued SFAS No. 160, “Noncontrolling Interests in Consolidated Financial Statements—An Amendment of ARB No. 51” (“SFAS No. 160”). SFAS No. 160 establishes accounting and reporting standards for the noncontrolling interest in a subsidiary (previously referred to as minority interests). SFAS No. 160 also requires that a retained noncontrolling interest upon the deconsolidation of a subsidiary be initially measured at its fair value. Upon effectiveness of SFAS No. 160, the Company would be required to report any noncontrolling interests as a separate component of consolidated stockholders’ equity. The Company would also be required to present any net income allocable to noncontrolling interests and net income attributable to the stockholders of the Company separately in its consolidated statements of operations. SFAS No. 160 is effective for fiscal years, and interim periods within those fiscal years, beginning on or after January 1, 2009. SFAS No. 160 requires retroactive adoption of the presentation and disclosure requirements for existing minority interests. All other requirements of SFAS No. 160 shall be applied prospectively. SFAS No. 160 would have an impact on the presentation and disclosure of the noncontrolling interests of any non wholly-owned businesses acquired in the future.
In March 2008, the Financial Accounting Standards Board (“FASB”) issued Statement of Financial Accounting Standards No. 161, “Disclosures about Derivative Instruments and Hedging Activities—an amendment of FASB Statement No. 133” (“SFAS No. 161”). SFAS No. 161 changes the disclosure requirements for derivative instruments and hedging activities. Entities are required to provide enhanced disclosures about (a) how and why an entity uses derivative instruments, (b) how derivative instruments and related hedged items are accounted for under SFAS No. 133 and its related interpretations, and (c) how derivative instruments and related hedged items affect an entity’s financial position, financial performance and cash flows. The guidance in SFAS No. 161 is effective for financial statements issued for fiscal years and interim periods beginning after November 15, 2008, with early application encouraged. This Statement encourages, but does not require, comparative disclosures for earlier periods at initial adoption. Management has concluded that the implementation of SFAS No. 161 will have no impact on the Company’s consolidated financial statements.
In April 2008, the FASB issued FASB Staff Position SFAS 142-3, Determination of the Useful Life of Intangible Assets (“FSP SFAS 142-3”). FSP SFAS 142-3 amends the factors that should be considered in developing renewal or extension assumptions used to determine the useful life of a recognized intangible asset under FASB Statement No. 142, Goodwill and Other Intangible Assets. The objective of this FSP is to improve the consistency between the useful life of a recognized intangible asset under Statement 142 and the period of expected cash flows used to measure the fair value of the asset under SFAS 141R, Business Combinations, and other U.S. GAAP principles. FSP SFAS 142-3 is effective for fiscal years beginning after December 15, 2008. The adoption of FSP SFAS 142-3 is effective January 1, 2009 and is not expected to have a material impact on the Company’s consolidated financial statements.
In June 2008, the FASB ratified Emerging Issue Task Force (“EITF”) 07-5, “Determining Whether an Instrument (or an Embedded Feature) is Indexed to an Entity’s Own Stock” (“EITF 07-5”). EITF 07-5 provides framework for determining whether an instrument is indexed to an entity’s own stock. EITF 07-5 is effective for fiscal years beginning after December 15, 2008. The implementation of EITF 07-5 is not expected to have a material effect on the Company’s consolidated financial statements.
In October 2008, The FASB issued FSP 157-3 “Determining Fair Value of a Financial Asset in a Market That is Not Active” (FSP 157-3). FSP 157-3 classified the application of SFAS No. 157 in an inactive market. It demonstrated how the fair value of a financial asset is determined when the market for that financial asset is inactive. FSP 157-3 was effective upon issuance, including prior periods for which financial statements had not been issued. The implementation of FSP 157-3 did not have a material effect on the Company’s consolidated financial statements.
In September 2006, the FASB issued SFAS No. 157, “Fair Value Measurements” (“SFAS No. 157”). This Statement defines fair value, establishes a framework for measuring fair value and expands disclosure of fair value measurements. SFAS No. 157 applies under other accounting pronouncements that require or permit fair value measurements and accordingly, does not require any new fair value measurements. We adopted SFAS No. 157 as of January 1, 2008. In February 2008, the FASB issued FASB Staff Position No. FAS 157-2, "Effective Date of FASB Statement No. 157", which provides a one year deferral of the effective date of SFAS No.157 for non-financial assets and non-financial liabilities, except those that are recognized or disclosed in the financial statements at fair value at least annually. Therefore, the Company has adopted the provisions of SFAS No. 157 with respect to its financial assets and liabilities only. The adoption of SFAS No. 157 did not have a material effect on the Company’s consolidated financial statements.
26
ITEM 8: Financial Statements and Supplementary Data
The financial statements required hereby are located on pages F-1 through F-19 of this report.
ITEM 9: Changes In and Disagreements with Accountants on Accounting and Financial Disclosure
Not applicable.
ITEM 9A: Controls and Procedures
Evaluation of Disclosure Controls and Procedures
We maintain a set of disclosure controls and procedures, as defined in Section 13a-15(e) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), that are designed to ensure that information required to be disclosed by us in the reports filed by us under the Securities Exchange Act of 1934, as amended (the “Exchange Act”) is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. We carried out an evaluation, under the supervision and with the participation of our Chief Executive Officer and our Chief Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures pursuant to Rule 13a-15(b) of the Exchange Act. Based on that evaluation, our Chief Executive Officer and our Chief Financial Officer concluded that our disclosure controls and procedures were effective as of the end of the period covered by this report.
Notwithstanding the foregoing, there can be no assurance that the Company’s disclosure controls and procedures will detect or uncover all failures of persons within the Company to disclose material information otherwise required to be set forth in the Company’s periodic reports. There are inherent limitations to the effectiveness of any system of disclosure controls and procedures, including the possibility of human error and the circumvention or overriding of the controls and procedures. Accordingly, even effective disclosure controls and procedures can only provide reasonable, not absolute, assurance of achieving their control objectives.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rule 13a-15(f). Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of the financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures are being made only with proper authorizations; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Our management, under the supervision of and with the participation of the Chief Executive Officer and the Chief Financial Officer, assessed the effectiveness of our internal control over financial reporting as of December 31, 2008 based on criteria for effective control over financial reporting described in Internal Control — Integrated Framework issued by the COSO. Based on this assessment, our management concluded that our internal control over financial reporting was effective as of December 31, 2008.
27
This annual report does not include an attestation report of the Company's independent registered public accounting firm regarding internal control over financial reporting. Management's report was not subject to attestation by the Company's registered public accounting firm pursuant to temporary rules of the Securities and Exchange Commission that permit the Company to provide only management's report in this annual report.
Changes in Internal Controls over Financial Reporting
An evaluation was performed under the supervision of the Company’s management, including the Chief Executive Officer and Chief Financial Officer, as required under Exchange Act Rule 13a-15(d) and 15d-15(d), of whether any change in the Company’s internal control over financial reporting occurred during the fiscal quarter ended December 31, 2008. Based on that evaluation, the Company’s management, including the Chief Executive Officer and Chief Financial Officer, concluded that no change in the Company’s internal controls over financial reporting occurred during the fiscal quarter ended December 31, 2008 that has materially affected or is reasonably likely to materially affect, the Company’s internal control over financial reporting.
ITEM 9B: Other Information
None.
28
PART III
ITEM 10: Directors, Executive Officers, Promoters and Control Persons; Compliance with Section 16(a) of the Exchange Act
The information required by this Item regarding our directors and executive officers is incorporated in this report by reference to our Proxy Statement for our 2009 Annual Meeting of Stockholders where such information appears under the heading “Directors and Executive Officers” in our Proxy Statement for our 2009 Annual Meeting of Stockholders.
Section 16(a) Beneficial Ownership Reporting Compliance
The discussion under the heading “Security Ownership of Certain Beneficial Owners and Management” in our definitive proxy statement for the 2009 Annual Meeting of Stockholders is incorporated herein by reference to such proxy statement.
Code of Ethics
The Company has adopted a Code of Ethics that is applicable to our principal executive officer and principal financial officer and can be viewed on our website www.virtualscopics.com.
ITEM 11. Executive Compensation
The information required by this Item is incorporated in this report by reference to our definitive Proxy Statement referred to in Item 10 above where such information appears under the heading “Executive Compensation and Other Matters.”
ITEM 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this Item regarding security ownership of certain beneficial owners and management is incorporated in this report by reference to our definitive Proxy Statement referred to in Item 10 above where such information appears under the heading “Security Ownership of Certain Beneficial Owners and Management” and “Equity Compensation Plans” is.
ITEM 13. Certain Relationships and Related Transactions
The information required by this Item is incorporated in this report by reference to our definitive Proxy Statement referred to in Item 10 above where such information appears under the heading “Certain Relationships and Related Transactions.”
ITEM 14. Principal Accountant Fees and Services
The information required by this Item is incorporated in this report by reference to our definitive Proxy Statement referred to in Item 10 above where such information appears under the heading “Principal Accounting Fees and Services.”
ITEM 15: Exhibits
The list of exhibits required by this Item is incorporated in this Item by reference to the exhibit index attached after the signature page to this report.
29
SIGNATURES
In accordance with Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| March 19, 2009 | VirtualScopics, Inc. (Registrant) |
| /s/ Jeffrey Markin | |
| President and Chief Executive Officer |
POWER OF ATTORNEY
KNOW ALL BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Jeffrey Markin and Molly Henderson, or either of them, as his or her true and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution, for him and in his name, place and stead, in any and all capacities, to sign any and all amendments to this Annual Report on Form 10-K and any documents related to this report and filed pursuant to the Securities and Exchange Act of 1934, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or their substitute or substitutes may lawfully do or cause to be done by virtue hereof.
In accordance with the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
| DATE | SIGNATURE | TITLE | |||
| March 19, 2009 | /s/ Jeffrey Markin | President and Chief Executive Officer | |||
| (Jeffrey Markin) | (Principal Executive Officer) | ||||
| March 19, 2009 | /s/ Molly Henderson | Chief Business and Financial Officer and | |||
| (Molly Henderson) | Sr. Vice President | ||||
| (Principal Financial and Accounting Officer) | |||||
| March 19, 2009 | /s/ Robert Klimasewski | Chairman of the Board of Directors | |||
| (Robert Klimasewski) | |||||
| March 19, 2009 | /s/ Norman Mintz | Director | |||
| (Norman Mintz) | |||||
| March 19, 2009 | /s/ Colby Chandler | Director | |||
| (Colby Chandler) | |||||
| March 19, 2009 | /s/ Charles Phelps | Director | |||
| (Charles Phelps) | |||||
| March 19, 2009 | /s/ Sidney Knafel | Director | |||
| (Sidney Knafel) | |||||
| March 19, 2009 | /s/ Dan Kerpelman | Director | |||
| (Dan Kerpelman) | |||||
| March 19, 2009 | /s/ Terence Walts | Director | |||
| (Terence Walts) | |||||
| March 19, 2009 | /s/ Mostafa Analoui | Director | |||
| (Mostafa Analoui) |
30
Exhibit Index
| 2.1 | Securities Purchase Agreement dated September 12, 2007 by and among the VirtualScopics, Inc. and the Buyers listed on the Schedule of Buyers attached thereto (Incorporated herein by reference to Exhibit 10.1 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on September 17, 2007 (File No. 000-52018)). (Schedules and exhibits have been omitted pursuant to Regulation S-B Item 601(b)(2) and will be made available to the Commission upon request). |
| 3.1 | Certificate of Incorporation of VirtualScopics, Inc. dated April 21, 1988 (Incorporated herein by reference to Exhibit 3.1 of the VirtualScopics, Inc.’s Registration Statement on Form SB-2 filed with the Securities and Exchange Commission on November 4, 2004 (File No. 333-120253)). |
| 3.2 | Certificate of Amendment of Certificate of Incorporation of VirtualScopics, Inc. dated February 2, 1989 (Incorporated herein by reference to Exhibit 3.1a of the VirtualScopics, Inc.’s Registration Statement on Form SB-2 filed with the Securities and Exchange Commission on November 4, 2004 (File No. 333-120253)). |
| 3.3 | Certificate for Renewal and Revival of Certificate of Incorporation of VirtualScopics, Inc. dated February 23, 2004 (Incorporated herein by reference to Exhibit 3.1b of the VirtualScopics, Inc.’s Registration Statement on Form SB-2 filed with the Securities and Exchange Commission on November 4, 2004 (File No. 333-120253)). |
| 3.4 | Certificate of Amendment of Certificate of Incorporation of VirtualScopics, Inc. dated August 20, 2004 (Incorporated herein by reference to Exhibit 3.1c of the VirtualScopics, Inc.’s Registration Statement on Form SB-2 filed with the Securities and Exchange Commission on November 4, 2004 (File No. 333-120253)). |
| 3.5 | Certificate of Amendment of Certificate of Incorporation of VirtualScopics, Inc. dated October 7, 2005 (Incorporated by reference to Exhibit 3.5 the VirtualScopics, Inc.’s Annual Report on Form 10-KSB filed with the Securities and Exchange Commission on March 30, 2006 (File No. 333-120253)). |
| 3.6 | Certificate of Amendment to Certificate of Incorporation of VirtualScopics, Inc. dated November 4, 2005 (Incorporated herein by reference to Exhibit 3.1 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 3.7 | Certificate of Designations, Powers, Preferences and Other Rights and Qualifications of Series A Convertible Preferred Stock of VirtualScopics, Inc. dated November 4, 2005 (Incorporated herein by reference to Exhibit 3.2 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 3.8 | Certificate of Designation of Rights and Preferences of the Series B Preferred Stock of VirtualScopics, Inc. dated September 12, 2007 (Incorporated herein by reference to Exhibit 3.1 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on September 17, 2007 (File No. 000-52018)). |
| 3.9 | Certificate of Amendment to Certificate of Designation of Rights and Preferences of the Series B Preferred Stock of VirtualScopics, Inc. dated November 27, 2007 (Incorporated herein by reference to Exhibit 3.1 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 29, 2007 (File No. 000-52018)). |
| 3.10 | Amended and Restated Bylaws of VirtualScopics, Inc. dated August 28, 2007 (Incorporated herein by reference to Exhibit 3.9 to the VirtualScopics, Inc.’s Registration Statement of Form S-3 filed with the Securities Exchange Commission on October 11, 2007 (File No. 333-146635)). |
| 4.1 | Form of VirtualScopics, Inc. Four-Year Warrant to Purchase Common Stock at $4.00 per share (Incorporated herein by reference to Exhibit 4.1 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
31
| 4.2 | Form of VirtualScopics, Inc. Four-Year Warrant to Purchase Common Stock at $2.50 per share (Incorporated herein by reference to Exhibit 4.2 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 4.3 | Registration Rights Agreement Dated September 12, 2007 between the VirtualScopics, Inc. and the Buyers listed on the Schedule of Buyers thereto (Incorporated herein by reference to Exhibit 10.3 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on September 17, 2007 (File No. 000-52018)). |
| 4.4 | Form of Warrant to Purchase Common Stock of VirtualScopics (Incorporated herein by reference to Exhibit 10.2 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on September 17, 2007 (File No. 000-52018)). |
| 10.1 | VirtualScopics, Inc. 2005 Long Term Incentive Plan (Incorporated herein by reference to Exhibit 10.1 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)).* |
| 10.2 | Form of Lock-Up Agreement between VirtualScopics, LLC and certain of its former members (Incorporated herein by reference to Exhibit 10.2 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 10.3 | Form of Lock-Up Agreement between VirtualScopics, LLC and its employees, director and certain of its former members (Incorporated herein by reference to Exhibit 10.3 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 10.4 | Clinical Imaging Development and Services Agreement dated July 26, 2005 by and between VirtualScopics, LLC and Pfizer, Inc. (Incorporated herein by reference to Exhibit 10.6 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 10.5 | Amendment to Clinical Imaging Development and Services Agreement dated October 5, 2006 by and between VirtualScopics, Inc. and Pfizer, Inc. (Incorporated herein by reference to Exhibit 10.1 to the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 28, 2006 (File No. 000-52018)). |
| 10.6 | Equipment Purchase Agreement dated December, 2003 by and between VirtualScopics, LLC and the University of Rochester Medical Center. (Incorporated herein by reference to Exhibit 10.9 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 10.7 | Determination of Intellectual Property Rights Agreement dated July 1, 2002 by and between VirtualScopics, LLC and the University of Rochester (Incorporated herein by reference to Exhibit 10.10 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on November 10, 2005 (File No. 333-120253)). |
| 10.8 | Option Agreements with Robert Klimasewski dated November 5, 2005 (Incorporated herein by reference to Exhibit 10.18 to the VirtualScopics, Inc. Registration Statement on Form SB-2 filed with the Securities and Exchange Commission on May 2, 2006 (File No. 333-133747)).* |
| 10.9 | Form of April 28, 2006 Indemnification Agreement by and among VirtualScopics, Inc. and the directors and officers of the VirtualScopics, Inc. (Incorporated herein by reference to Exhibit 10.19 to the VirtualScopics, Inc. Registration Statement on Form SB-2 filed with the Securities and Exchange Commission on May 2, 2006 (File No. 333-133747)).* |
32
| 10.10 | VirtualScopics, Inc. 2006 Long Term Incentive Plan (incorporated herein by reference to Exhibit A to the Registrant’s Definitive Proxy Statement on Schedule 14A, filed with the Commission on April 26, 2007) |
| 10.11 | 2007 Bonus Plan (Incorporated herein by reference to Exhibit 10.21 of the VirtualScopics, Inc. Annual Report on Form 10-KSB filed with the Securities and Exchange Commission on April 2, 2007 (File No. 000-52018)).* |
| 10.12 | Independent Director Compensation Plan (Incorporated herein by reference to Exhibit 10.22 of the VirtualScopics, Inc. Annual Report on Form 10-KSB filed with the Securities and Exchange Commission on April 2, 2007 (File No. 000-52018))* |
| 10.13 | Indemnification Agreement by and among VirtualScopics, Inc. and Norman Mintz, dated as of August 1, 2007. (Reference is made to the VirtualScopics, Inc. Form of Indemnification Agreement by and among VirtualScopics, Inc., and the directors and officers of the VirtualScopics, Inc. filed as Exhibit 10.19 to the VirtualScopics, Inc. Registration Statement on Form SB-2 filed with the Securities and Exchange Commission on May 2, 2006 (File No. 333-133747)).* |
| 10.14 | Escrow Agreement by and between VirtualScopics, Inc. and each buyer identified on Schedule of Buyers attached thereto and Woods Oviatt Gilman LLP, dated September 12, 2007, with the Amendment and Waiver, dated September 13, 2007. (Incorporated herein by reference to Exhibit 10.5 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on September 17, 2007 (File No. 000-52018)). |
| 10.15 | Standstill Agreement by and between VirtualScopics, Inc. Jeffrey Markin, dated September 12, 2007. (Incorporated herein by reference to Exhibit 10.4 of the VirtualScopics, Inc. Report on Form 8-K filed with the Securities and Exchange Commission on September 17, 2007 (File No. 000-52018)).* |
| 10.16 | Non-Employee Director Compensation Plan (Incorporated herein by reference to Exhibit 10.1 to the VirtualScopics, Inc. Report on Form 10-Q filed with the Securities and Exchange Commission on August12, 2008 (File No.000-52018)).* |
| 10.17 | Employment Agreement dated February 24, 2009, by and between Jeffrey Markin and VirtualScopics, Inc.* |
| 10.18 | Employment Agreement dated February 24, 2009, by and between Molly Henderson and VirtualScopics, Inc.* |
| 21 | Subsidiaries of VirtualScopics, Inc. |
| 23.1 | Consent of Marcum & Kliegman, LLP |
| 24 | Power of Attorney (included on the signature page to this report) |
| 31.1 | Certification of Chief Executive Officer as required by Rule 13a-14 Or 15d-14 of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 Of The Sarbanes-Oxley Act of 2002 |
| 31.2 | Certification of Chief Financial Officer as required by Rule 13a-14 Or 15d-14 of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 Of The Sarbanes-Oxley Act of 2002 |
| 32.1 | Certification pursuant to 18 U.S.C. Section 1350 by the Chief Executive Officer, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| 32.2 | Certification pursuant to 18 U.S.C. Section 1350 by the Chief Financial Officer, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 |
| * | Management contract or compensatory plan or arrangement. |
33
VirtualScopics, Inc. and Subsidiary
Index to Consolidated Financial Statements
| F-2 | ||
| Consolidated Balance Sheets as of December 31, 2008 and 2007 | F-3 | |
| Consolidated Statements of Operations for the Years Ended December 31, 2008 and 2007 | F-4 | |
| Consolidated Statements of Changes in Stockholders’ Equity for the Years Ended December 31, 2008 and 2007 | F-5 | |
| Consolidated Statements of Cash Flows for the Years Ended December 31, 2008 and 2007 | F-6 | |
| Notes to Consolidated Financial Statements | F-7 - F-19 |
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Audit Committee of the
Board of Directors and Stockholders of
VirtualScopics, Inc. and Subsidiary
We have audited the accompanying consolidated balance sheets of VirtualScopics, Inc. and Subsidiary (the “Company”) as of December 31, 2008 and 2007, and the related consolidated statements of operations, changes in stockholders’ equity and cash flows for the years then ended. These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the consolidated financial position of VirtualScopics, Inc. and Subsidiary as of December 31, 2008 and 2007, and the results of their operations and their cash flows for the years then ended in conformity with accounting principles generally accepted in the United States of America.
/s/ Marcum & Kliegman LLP
New York, New York
March 18, 2009
F-2
VirtualScopics, Inc. and Subsidiary
Consolidated Balance Sheets
| December 31, | ||||||||
| 2008 | 2007 | |||||||
| Assets | ||||||||
| Current assets | ||||||||
| Cash and cash equivalents | $ | 3,143,904 | $ | 3,955,835 | ||||
| Restricted cash | - | 455,583 | ||||||
| Accounts receivable | 1,021,110 | 648,300 | ||||||
| Prepaid expenses and other assets | 263,297 | 306,301 | ||||||
| Total current assets | 4,428,311 | 5,366,019 | ||||||
| Patents, net | 1,920,446 | 1,948,785 | ||||||
| Property and equipment, net | 355,479 | 542,679 | ||||||
| Other assets | 156,788 | 280,317 | ||||||
| Total assets | $ | 6,861,024 | $ | 8,181,016 | ||||
| Liabilities and Stockholders’ Equity | ||||||||
| Current liabilities | ||||||||
| Accounts payable and accrued expenses | $ | 659,009 | $ | 626,693 | ||||
| Accrued payroll | 554,425 | 221,013 | ||||||
| Unearned revenue | 291,594 | 279,275 | ||||||
| Total current liabilities | 1,505,028 | 1,126,981 | ||||||
| Commitments and Contingencies | ||||||||
| Stockholders’ Equity | ||||||||
| Convertible preferred stock, $0.001 par value; 15,000,000 shares authorized; 8,400 shares designated Series A; issued and outstanding; 3,976 and 4,001 at December 31, 2008 and 2007, respectively, liquidation preference $1,000 per share | 4 | 4 | ||||||
| 6,000 shares designated Series B; issued and outstanding; 4,226 and 4,230 at December 31, 2008 and 2007, respectively, liquidation preference $1,000 per share | 4 | 4 | ||||||
| Common stock, $0.001 par value; 85,000,000 shares authorized; issued and outstanding, 23,502,352 shares at December 31, 2008 and $23,225,664 shares at December 31, 2007 | 23,503 | 23,226 | ||||||
| Additional paid-in capital | 16,546,550 | 15,616,124 | ||||||
| Accumulated deficit | (11,214,065 | ) | (8,585,323 | ) | ||||
| Total stockholders’ equity | 5,355,996 | 7,054,035 | ||||||
| Total liabilities and stockholders’ equity | $ | 6,861,024 | $ | 8,181,016 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
F-3
VirtualScopics, Inc. and Subsidiary
Consolidated Statements of Operations
| For the Years Ended December 31, | ||||||||
| 2008 | 2007 | |||||||
| Revenue | $ | 7,130,518 | $ | 5,647,049 | ||||
| Cost of services | 4,031,198 | 3,680,409 | ||||||
| Gross profit | 3,099,320 | 1,966,640 | ||||||
| Operating expenses | ||||||||
| Research and development | 941,193 | 1,440,312 | ||||||
| Sales and marketing | 1,219,882 | 749,329 | ||||||
| General and administrative | 3,158,359 | 3,753,552 | ||||||
| Depreciation and amortization | 466,150 | 485,520 | ||||||
| Total operating expenses | 5,785,584 | 6,428,713 | ||||||
| Operating loss | (2,686,264 | ) | (4,462,073 | ) | ||||
| Other income (expense) | ||||||||
| Interest income | 73,599 | 153,968 | ||||||
| Other expense | (16,077 | ) | (13,279 | ) | ||||
| Total other income | 57,522 | 140,689 | ||||||
| Net loss | (2,628,742 | ) | (4,321,384 | ) | ||||
| Series B preferred stock deemed dividend | - | 1,381,163 | ||||||
| Series B preferred stock cash dividend | 338,827 | 99,433 | ||||||
| Net loss attributable to common stockholders | $ | (2,967,569 | ) | $ | (5,801,980 | ) | ||
| Basic and diluted net loss per common share | $ | (0.13 | ) | $ | (0.25 | ) | ||
| Weighted average number of common shares outstanding basic and diluted | 23,389,705 | 23,058,820 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-4
VirtualScopics, Inc. and Subsidiary
Consolidated Statements of Changes in Stockholders’ Equity
For the Years Ended December 31, 2008 and 2007
Series A | Series B | |||||||||||||||||||||||||||||||||||
Preferred Stock | Preferred Stock | Common Stock | Additional | |||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Shares | Amount | Paid-In Capital | Accumulated Deficit | Total | ||||||||||||||||||||||||||||
| Balances at January 1, 2007 | 4,346 | $ | 4 | - | - | 22,962,826 | $ | 22,963 | $ | 10,420,893 | $ | (4,263,939 | ) | $ | 6,179,921 | |||||||||||||||||||||
| Conversion of Series A Preferred to Common Stock | (345 | ) | - | - | - | 138,195 | 138 | (138 | ) | - | - | |||||||||||||||||||||||||
| Conversion of Series B Preferred to Common Stock | - | - | (120 | ) | - | 99,643 | 100 | (100 | ) | - | - | |||||||||||||||||||||||||
| Exercise of Employee Stock Options | - | - | - | - | 25,000 | 25 | 18,350 | - | 18,375 | |||||||||||||||||||||||||||
| Amortization of stock option costs | - | - | - | - | - | - | 1,403,129 | - | 1,403,129 | |||||||||||||||||||||||||||
| Stock options issued to non-employees for services | - | - | - | - | - | - | 1,818 | - | 1,818 | |||||||||||||||||||||||||||
| Series B preferred stock issued in private placement, net of issuance costs of $478,391 | - | - | 4,350 | 4 | - | - | 3,871,605 | - | 3,871,609 | |||||||||||||||||||||||||||
| Series B preferred stock cash dividends based on 8% annual rate | - | - | - | - | - | - | (99,433 | ) | - | (99,433 | ) | |||||||||||||||||||||||||
| Net loss | (4,321,384 | ) | $ | (4,321,384 | ) | |||||||||||||||||||||||||||||||
| Balances at December 31, 2007 | 4,001 | $ | 4 | 4,230 | $ | 4 | 23,225,664 | $ | 23,226 | $ | 15,616,124 | $ | (8,585,323 | ) | $ | 7,054,035 | ||||||||||||||||||||
| Conversion of Series A Preferred to Common Stock | (25 | ) | - | - | - | 20,759 | 21 | (21 | ) | - | - | |||||||||||||||||||||||||
| Conversion of Series B Preferred to Common Stock | - | - | (4 | ) | - | 3,321 | 3 | (3 | ) | - | - | |||||||||||||||||||||||||
| Exercise of warrants | - | - | - | - | 87,601 | 88 | 788 | - | 876 | |||||||||||||||||||||||||||
| Amortization of stock option costs | - | - | - | - | - | - | 1,047,654 | - | 1,047,654 | |||||||||||||||||||||||||||
| Common stock issued for rent | - | - | - | - | 165,007 | 165 | 220,835 | - | 221,000 | |||||||||||||||||||||||||||
| Series B preferred stock cash dividends based on 8% annual rate | - | - | - | - | - | - | (338,827 | ) | - | (338,827 | ) | |||||||||||||||||||||||||
| Net loss | (2,628,742 | ) | $ | (2,628,742 | ) | |||||||||||||||||||||||||||||||
| Balances at December 31, 2008 | 3,976 | $ | 4 | 4,226 | $ | 4 | 23,502,352 | $ | 23,503 | $ | 16,546,550 | $ | (11,214,065 | ) | $ | 5,355,996 | ||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-5
VirtualScopics, Inc. and Subsidiary
Consolidated Statements of Cash Flows
| For the Years Ended December 31, | ||||||||
| 2008 | 2007 | |||||||
| Cash flows from operating activities | ||||||||
| Net loss | $ | (2,628,742 | ) | $ | (4,321,384 | ) | ||
| Adjustments to reconcile change in net loss to net cash used in operating activities: | ||||||||
| Depreciation and amortization | 466,150 | 485,520 | ||||||
| Stock-based compensation expense | 1,123,283 | 1,484,884 | ||||||
| Issuance of equity instruments for rent | 221,000 | - | ||||||
| Issuance of equity instruments to non-employees for services | - | 1,818 | ||||||
Accrual for future issuance of equity instruments for rent Changes in operating assets and liabilities | 207,060 | - | ||||||
| Accounts receivable | (372,810 | ) | 13,719 | |||||
| Prepaid expenses and other assets | 43,004 | (83,109 | ) | |||||
| Unearned revenue | 12,319 | (235,744 | ) | |||||
| Accounts payable and accrued expenses | (250,373 | ) | 107,457 | |||||
| Accrued payroll | 333,412 | (267,052 | ) | |||||
| Total adjustments | 1,783,045 | 1,507,493 | ||||||
| Net cash used in operating activities | (845,697 | ) | (2,813,891 | ) | ||||
| Cash flows from investing activities | ||||||||
| Purchase of equipment | (26,731 | ) | (204,791 | ) | ||||
| Acquisition of patents | (100,351 | ) | (137,942 | ) | ||||
| Net cash used in investing activities | (127,082 | ) | (342,733 | ) | ||||
| Cash flows from financing activities | ||||||||
| Proceeds from issuance of preferred stock and warrants, net of issuance costs of $478,391 | - | 3,871,609 | ||||||
| Proceeds from the exercise of warrants and stock options | 876 | 18,375 | ||||||
| Restricted cash | 498,799 | (498,799 | ) | |||||
| Cash dividends on series B preferred stock | (338,827 | ) | (99,433 | ) | ||||
| Repayments of notes payable - related parties | - | (80,446 | ) | |||||
| Net cash provided by financing activities | 160,848 | 3,211,306 | ||||||
| Net (decrease) increase in cash and cash equivalents | (811,931 | ) | 54,682 | |||||
| Cash and cash equivalents | ||||||||
| Beginning of year | 3,955,835 | 3,901,153 | ||||||
| End of year | $ | 3,143,904 | $ | 3,955,835 | ||||
| Supplemental disclosure of cash flow information | ||||||||
| Cash paid during the year for: | ||||||||
| Interest | $ | - | $ | 2,912 | ||||
| Issuance of common stock for partial payment of rent in equity | $ | 221,0000 | $ | - | ||||
The accompanying notes are an integral part of these consolidated financial statements.
F-6
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
NOTE 1 - Organization and Basis of Presentation
Nature of Business
The Company’s headquarters are located in Rochester, New York. The Company’s business evolved from research first carried out at the University of Rochester, a related party (Note 9). As a result of this research, the Company has created a suite of image analysis software tools and applications which are used in detecting and analyzing specific structures in medical images. The Company’s developed software provides measurement and visualization capabilities designed to improve clinical research and development.
NOTE 2 - Summary of Significant Accounting Policies
Principles of Consolidation
The accompanying consolidated financial statements include the accounts of the Company and its wholly-owned subsidiary, VirtualScopics, LLC. All significant intercompany balances and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the year. Actual results could differ from those estimates.
Cash and Cash Equivalents
The Company considers all highly liquid investments purchased with a maturity of three months or less to be cash equivalents.
Accounts Receivable
Accounts receivable are stated at estimated net realizable value. Accounts receivable are comprised of balances due from customers net of estimated allowances for uncollectible accounts, if any. In determining collectability, historical trends are evaluated and specific customer issues are reviewed to arrive at appropriate allowances, if any.
Right to Use Equipment
In April 2004, the Company obtained the right to use a Magnetic Resonance Imaging (“MRI”) machine owned by the University of Rochester for a period of seven years (Note 9). The Company has recorded the value of the right as an other asset in the accompanying consolidated balance sheets and is amortizing the asset based on usage over the life of the agreement.
For the years ended December 31, 2008 and 2007, the total amount charged to amortization on this asset which is included in the accompanying consolidated statements of operations was $123,529 and $140,159, respectively. As of December 31, 2008 and 2007, the unamortized balance of this asset is $280,317 and $403,846, respectively, of which $123,529 is classified within prepaid expenses and other assets for 2008 and 2007.
Patents
Costs incurred to acquire and file for patents, including legal costs, are capitalized as long-lived assets and amortized on a straight-line basis over the lower of the estimated useful life or legal life of the patent, which is 20 years.
Impairment of Long-Lived Assets
In the event that facts and circumstances indicate that the carrying amounts of long-lived assets may be impaired, an evaluation of recoverability would be performed. If an evaluation is required, the estimated future undiscounted cash flows associated with the asset would be compared to the asset’s carrying amount to determine if a write-down is required. If the evaluation indicates that the assets will not be recoverable, the carrying value of the Company’s assets would be reduced to their estimated market value. The Company evaluated the carrying amount of its long-lived assets as of December 31, 2008 and determined that the long-lived assets are not impaired.
F-7
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
Property and Equipment
Property and equipment are carried at cost less accumulated depreciation. When retired or otherwise disposed of, the related cost and accumulated depreciation are removed from the respective accounts and any resulting gain or loss is recognized and included in income. Repairs and maintenance costs are expensed as incurred.
Depreciation is computed using the straight-line method over the following useful lives:
| Years | |||
| Office/computer equipment | 3-5 | ||
| Furniture and fixtures | 5-7 | ||
| Software | 3 |
Leasehold improvements, which are included in property and equipment, are recorded at cost less accumulated depreciation. Depreciation on leasehold improvements is computed using the straight-line method over the shorter of their estimated useful lives or the lease term, whichever is shorter.
Revenue Recognition
The Company applies the revenue recognition principles set forth under the Securities and Exchange Commission Staff Accounting Bulletin No. 104, “Revenue Recognition,” with respect to its revenues from image analysis, consulting and project/data management services, and recognizes revenue when it is realized or realizable and earned. The Company considers revenue realized or realizable and earned when an agreement exists, services and products are provided to the customer, prices are fixed or determinable, and collectibility is reasonably assured. Revenues are reduced for estimated discounts and other allowances, if any.
The Company provides advanced medical image analysis on a per image basis, and recognizes revenue when the image analysis is completed and delivered to the customer. Revenue related to project, data and site management services is recognized as the services are rendered and in accordance with the terms of the contract. Consulting revenue is recognized once the services are rendered and typically charged as an hourly rate.
Occasionally, the Company provides software development services to its customers, which may require significant development, modification, and customization. Software development revenue is billed on a fixed price basis and recognized upon delivery of the software and acceptance by the customer on a completed contract basis in accordance with the American Institute of Certified Public Accountants Statement of Position 81-1, “Accounting for Performance of Construction-Type and Certain Production-Type Contracts.” The Company does not sell software license, upgrades or enhancements, or post-contract customer services.
Reimbursements received for out-of-pocket expenses incurred are reported as revenue in the financial statements in accordance with Emerging Issues Task Force No. 01-14, “Income Statement Characterization of Reimbursements received for ‘Out-of-Pocket’ Expenses Incurred.”
Income Taxes
In accordance with Statement of Financial Accounting Standards No.109 “Accounting for Income Taxes,” we account for deferred income taxes under the liability method. Under this method, we recognize deferred tax assets and liabilities based on the tax effects of temporary differences between the financial statement and tax bases of assets and liabilities, as measured by current enacted tax rates. A valuation allowance is recorded to reduce deferred tax assets when necessary.
Research and Development
Research and development expense relates to the development of new products and processes including significant improvements to existing products. These costs are expensed as incurred.
F-8
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
Fair Value of Financial Instruments
The carrying amounts of cash and cash equivalents, restricted cash, accounts receivables, accounts payable and accrued expenses, accrued payroll, and unearned revenue approximate fair value due to the short-term maturities of these instruments.
Stock-Based Compensation
On January 1, 2006, the Company adopted the fair value recognition provisions of Statement of Financial Accounting Standards (“SFAS”) No. 123R, “Share-Based Payment,” using the modified prospective method. Consequently, for the years ended December 31, 2008 and 2007, the Company’s consolidated statements of operations reflect stock-based compensation expense for stock options granted under its long-term stock incentive plans amounting to $1,123,283 and $1,484,884, respectively.
Stock options issued under the Company’s long-term incentive plans are granted with an exercise price equal to no less than the market price of the Company’s stock at the date of grant and expire up to ten years from the date of grant. These options generally vest over a three- or four-year period. The fair value of stock options granted is determined on the grant date using assumptions for risk free interest rate, the expected term, expected volatility, and expected dividend yield. The risk free interest rate is based on U.S. Treasury zero-coupon yield curve over the expected term of the option. The expected term assumption is primarily based on the Company’s historical data related to exercise and post-vesting cancellation information, which is expected to be similar to future results. Since the Company has limited historical volatility information, it bases its expected volatility on the historical volatility of similar entities whose share prices are publicly available. In making its determination as to similarity, the Company considered the industry, stage of life cycle, size, and financial leverage of such other entities. The Company’s model includes a zero dividend yield assumption, as the Company has not historically paid nor does it anticipate paying dividends on its common stock. The Company’s model does not include a discount for post-vesting restrictions, as the Company has not issued awards with such restrictions. The periodic expense is then determined based on the valuation of the options, and at that time an estimated forfeiture rate is used to reduce the expense recorded. The Company’s estimate of pre-vesting forfeitures is primarily based on the Company’s historical experience and is adjusted to reflect actual forfeitures as the options vest. The following assumptions were used to estimate the fair value of options granted for the years ended December 31, 2008 and 2007 using the Black-Scholes option-pricing model:
To estimate compensation expense which would be recognized under SFAS No. 123R, the Company used the Black-Scholes option-pricing model with the following weighted-average assumptions:
| December 31, | ||||||||
| 2008 | 2007 | |||||||
| Risk free interest rate | 3.85 | % | 4.35 | % | ||||
| Expected term (years) | 9.71 | 9.06 | ||||||
| Expected volatility | 89.7 | % | 92.7 | % | ||||
| Expected dividend yield | - | - | ||||||
The Company accounts for its stock-based payments to non-employees under the guidance of EITF 96-18, “Accounting for Equity Instruments that are Issued to Other than Employees for Acquiring or in Connection with Selling Goods or Services,” which states that the transaction should be valued based on the fair value of the services provided or the fair value of the equity received, whichever is more reliably measurable.
Loss Per Share
Basic loss per share is computed by dividing the net loss applicable to common shares by the weighted average number of common shares outstanding during the period. Diluted loss attributable to common shares adjusts basic loss per share for the effects of convertible securities, warrants, stock options and other potentially dilutive financial instruments only in the periods in which such effect is dilutive. The shares issuable upon the conversion of preferred stock, the exercise of stock options and warrants are excluded from the calculation of net loss per share as their effect would be antidilutive.
Securities that could potentially dilute earnings per share in the future that were not included in the computation of diluted loss per share consist of the following as of December 31,:
F-9
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
| 2008 | 2007 | |||||||
| Series A convertible preferred stock | 3,301,098 | 3,321,857 | ||||||
| Series B convertible preferred stock | 3,509,093 | 3,512,414 | ||||||
| Warrants to purchase common stock | 4,309,055 | 4,396,656 | ||||||
| Options to purchase common stock | 4,818,558 | 3,932,421 | ||||||
| Total | 15,937,804 | 15,163,348 | ||||||
Recent Accounting Pronouncements
In February 2007, the Financial Accounting Standards Board (“FASB”) issued SFAS No. 159, “The Fair Value Option for Financial Assets and Financial Liabilities—Including an amendment of FASB Statement No. 115” (“SFAS No. 159”). This Statement permits entities to choose to measure many financial instruments and certain other items at fair value. The objective is to improve financial reporting by providing entities with the opportunity to mitigate volatility in reported earnings caused by measuring related assets and liabilities differently without having to apply complex hedge accounting provisions. This Statement is expected to expand the use of fair value measurement, which is consistent with the Board’s long-term measurement objectives for accounting for financial instruments. SFAS No. 159 is effective as of the beginning of an entity’s first fiscal year that begins after November 15, 2007. Early adoption is permitted as of the beginning of a fiscal year that begins on or before November 15, 2007, provided the entity also elects to apply the provisions of SFAS No. 157. SFAS No. 159 did not have an impact on the Company’s consolidated financial statements.
In December 2007, the FASB issued SFAS No. 141R, “Business Combinations” (“SFAS No. 141R”), which replaces SFAS No. 141, “Business Combinations.” SFAS No. 141R establishes principles and requirements for determining how an enterprise recognizes and measures the fair value of certain assets and liabilities acquired in a business combination, including noncontrolling interests, contingent consideration, and certain acquired contingencies. SFAS No. 141R also requires acquisition-related transaction expenses and restructuring costs be expensed as incurred rather than capitalized as a component of the business combination. SFAS No. 141R will be applicable prospectively to business combinations for which the acquisition date is on or after January 1, 2009. SFAS No. 141R would have an impact on accounting for any businesses acquired after the effective date of this pronouncement.
In December 2007, the FASB issued SFAS No. 160, “Noncontrolling Interests in Consolidated Financial Statements—An Amendment of ARB No. 51” (“SFAS No. 160”). SFAS No. 160 establishes accounting and reporting standards for the noncontrolling interest in a subsidiary (previously referred to as minority interests). SFAS No. 160 also requires that a retained noncontrolling interest upon the deconsolidation of a subsidiary be initially measured at its fair value. Upon effectiveness of SFAS No. 160, the Company would be required to report any noncontrolling interests as a separate component of consolidated stockholders’ equity. The Company would also be required to present any net income allocable to noncontrolling interests and net income attributable to the stockholders of the Company separately in its consolidated statements of operations. SFAS No. 160 is effective for fiscal years, and interim periods within those fiscal years, beginning on or after January 1, 2009. SFAS No. 160 requires retroactive adoption of the presentation and disclosure requirements for existing minority interests. All other requirements of SFAS No. 160 shall be applied prospectively. SFAS No. 160 would have an impact on the presentation and disclosure of the noncontrolling interests of any non wholly-owned businesses acquired in the future.
In March 2008, the Financial Accounting Standards Board (“FASB”) issued Statement of Financial Accounting Standards No. 161, “Disclosures about Derivative Instruments and Hedging Activities—an amendment of FASB Statement No. 133” (“SFAS No. 161”). SFAS No. 161 changes the disclosure requirements for derivative instruments and hedging activities. Entities are required to provide enhanced disclosures about (a) how and why an entity uses derivative instruments, (b) how derivative instruments and related hedged items are accounted for under SFAS No. 133 and its related interpretations, and (c) how derivative instruments and related hedged items affect an entity’s financial position, financial performance and cash flows. The guidance in SFAS No. 161 is effective for financial statements issued for fiscal years and interim periods beginning after November 15, 2008, with early application encouraged. This Statement encourages, but does not require, comparative disclosures for earlier periods at initial adoption. Management has concluded that the implementation of SFAS No. 161 will have no impact on the Company’s consolidated financial statements.
F-10
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
In April 2008, the FASB issued FASB Staff Position SFAS 142-3, Determination of the Useful Life of Intangible Assets (“FSP SFAS 142-3”). FSP SFAS 142-3 amends the factors that should be considered in developing renewal or extension assumptions used to determine the useful life of a recognized intangible asset under FASB Statement No. 142, Goodwill and Other Intangible Assets. The objective of this FSP is to improve the consistency between the useful life of a recognized intangible asset under Statement 142 and the period of expected cash flows used to measure the fair value of the asset under SFAS 141R, Business Combinations, and other U.S. GAAP principles. FSP SFAS 142-3 is effective for fiscal years beginning after December 15, 2008. The adoption of FSP SFAS 142-3 is effective January 1, 2009 and is not expected to have a material impact on the Company’s consolidated financial statements.
In June 2008, the FASB ratified Emerging Issue Task Force (“EITF”) 07-5, “Determining Whether an Instrument (or an Embedded Feature) is Indexed to an Entity’s Own Stock” (“EITF 07-5”). EITF 07-5 provides framework for determining whether an instrument is indexed to an entity’s own stock. EITF 07-5 is effective for fiscal years beginning after December 15, 2008. The implementation of EITF 07-5 is not expected to have a material effect on the Company’s consolidated financial statements.
In October 2008, The FASB issued FSP 157-3 “Determining Fair Value of a Financial Asset in a Market That is Not Active” (FSP 157-3). FSP 157-3 classified the application of SFAS No. 157 in an inactive market. It demonstrated how the fair value of a financial asset is determined when the market for that financial asset is inactive. FSP 157-3 was effective upon issuance, including prior periods for which financial statements had not been issued. The implementation of FSP 157-3 did not have a material effect on the Company’s consolidated financial statements.
In September 2006, the FASB issued SFAS No. 157, “Fair Value Measurements” (“SFAS No. 157”). This Statement defines fair value, establishes a framework for measuring fair value and expands disclosure of fair value measurements. SFAS No. 157 applies under other accounting pronouncements that require or permit fair value measurements and accordingly, does not require any new fair value measurements. We adopted SFAS No. 157 as of January 1, 2008. In February 2008, the FASB issued FASB Staff Position No. FAS 157-2, "Effective Date of FASB Statement No. 157", which provides a one year deferral of the effective date of SFAS No.157 for non-financial assets and non-financial liabilities, except those that are recognized or disclosed in the financial statements at fair value at least annually. Therefore, the Company has adopted the provisions of SFAS No. 157 with respect to its financial assets and liabilities only. The adoption of SFAS No. 157 did not have a material effect on the Company’s consolidated financial statements, and the adoption of SFAS 157-2 is not expected to have a material effect on the Company’s consolidated financial statements.
NOTE 3 - Property and Equipment
Property and equipment consisted of the following as of December 31:
| 2008 | 2007 | |||||||
| Office/computer equipment | $ | 861,901 | $ | 856,066 | ||||
| Furniture and fixtures | 114,462 | 106,222 | ||||||
| Software | 147,650 | 147,650 | ||||||
| Leasehold improvements | 99,375 | 86,719 | ||||||
| 1,223,388 | 1,196,657 | |||||||
| Less: accumulated depreciation | (867,909 | ) | (653,978 | ) | ||||
| $ | 355,479 | $ | 542,679 | |||||
Depreciation expense amounted to $213,931 and $222,144 for the years ended December 31, 2008 and 2007, respectively.
F-11
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
NOTE 4 - Patents
On May 24, 2002, the Company purchased from the University of Rochester, a related party, certain patents developed by the Company’s founders and previously licensed by the Company under an Exclusive Right Agreement. The Company paid $1,500,000 and issued warrants to acquire 357,075 shares of common stock to the University of Rochester for the full right and title to the patents. The warrants were recorded at fair value which totaled $157,000. This amount was capitalized and is included in the costs of the patents and was charged as an increase to additional paid-in capital. In addition, the Company capitalized $100,351 of legal expenses and filing fees associated with these acquired and other subsequently filed patents during the year ended December 31, 2008.
Accumulated amortization on the patents amounted to $705,406 as of December 31, 2008. Amortization expense for the years ended December 31, 2008 and 2007 amounted to $128,690 and $123,217, respectively. The estimated amortization expense on the patents for the next five years and thereafter is as follows:
| For the Years Ending | ||||
| December 31, | Amount | |||
| 2009 | 130,282 | |||
| 2010 | 130,282 | |||
| 2011 | 130,282 | |||
| 2012 | 130,282 | |||
| 2013 | 130,282 | |||
| Thereafter | 1,269,036 | |||
| Total | $ | 1,920,446 | ||
NOTE 5 - Commitments and Contingencies
Operating Leases
The Company leases approximately 19,500 square feet of office space at its corporate headquarters in Rochester, New York. The annual Base Rent under the lease is $360,000, and increases 3% annually and expires in July 2012. The Base Rent is paid in two portions: the cash portion of $156,000 annually, paid in equal monthly installments, increasing 3% annually, and the stock portion is $204,000 annually, paid in equal monthly installments, increasing 3% annually. The stock portion is payable in common stock, par value $.001 per share. At the option of either the landlord or the Company, upon 90 days written notice to the other party, the option of paying a portion of an installment of Base Rent in stock may be rescinded and such party may elect to pay or receive, as applicable, such installment, or all future installment payments in cash only. In the event that the Company elects to rescind the Stock Portion of Base Rent, then the rate per square foot shall be $18.00, increasing 3% annually. In the event that Landlord elects to rescind the Stock Portion of Base Rent, then the rate per square foot shall be (i) $16.00 if said election is made during the first 24 months of the Term, (ii) $17.00 if said election is made after the 24th month but before the 36th month, and (iii) $18.00 if said election is made after the 36th month. Notwithstanding the foregoing, the conversion to cash only rental shall not require 90 days prior written notice if the stock ceases to be publicly traded; in such event, the conversion shall begin with the next monthly installment of Base Rent due and shall be based upon the rate of $18.00 per square foot increasing 3% annually. During 2008, we had issued 165,007 shares of the company’s common stock ranging from $1.20 to $2.50 per share for a total value of $221,000. As of December 31, 2008, there remains an accrued liability of $88,060 relating to the rent due in company stock that is included within accounts payable and accrued expenses. The Landlord has elected to receive their rent in all cash beginning on February 1, 2009.
In October 2007, the Company entered into a lease agreement for certain equipment. The lease is for 36 months and will expire in November 2010.
Total rent expense for the years ended December 31, 2008 and 2007 was $371,004 and $277,999, respectively.
F-12
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
Future minimum rental commitments under non-cancelable operating leases are as follows:
| For the Years Ending | ||||
| December 31, | Amount | |||
| 2009 | $ | 327,184 | ||
| 2010 | 330,851 | |||
| 2011 | 335,966 | |||
| 2012 | 170,465 | |||
| 2013 | - | |||
| Total | $ | 1,164,466 | ||
Services and Co-Marketing Agreement
The Company entered into a Consulting Services and Co-Marketing Agreement dated March 1, 2004, in which it agreed to pay Chondrometrics GmbH, a German limited liability company, fees equal to approximately 7% of the gross revenues it derived from certain services each year throughout the term of the agreement. The Company was obligated to make minimum payments to Chondrometrics for the first three years of the agreement. Payments made to Chondrometrics in 2008 and 2007 amounted to approximately $15,000 and $37,000, respectively. The Consulting Services agreement expired on December 31, 2006 whereas the Co-Marketing Agreement expired on December 31, 2008.
NOTE 6 - Equity Transactions
Warrants
Warrants to purchase common stock have been issued to various individuals at exercise prices ranging from $0.01 to $2.50 per common unit in connection with the acquisition of patents and private placements, see below.
Common Stock
The Company has authorized 85,000,000 shares of common stock, par value $0.001. As of December 31, 2008, the Company had reserved 2,327,937 shares of common stock for issuance under its 2001 and 2005 long-term incentive plans, another 350,000 shares of common stock issued to a previous CEO outside of one of its long-term incentive plans, and 2,500,000 shares for its 2006 Long-term Incentive Plan.
Preferred Stock
The Company has authorized 15,000,000 shares of preferred stock, par value $0.001 per share, of which 8,400 are designated as Series A Convertible Preferred Stock (“Series A Preferred”) and 6,000 are designated as Series B Convertible Preferred Stock (“Series B Preferred”) as specified in the Certificate of Designation (the “Certificate”).
On November 4, 2005, November 23, 2005 and December 2, 2005, the Company completed a private placement totaling 7,000 units at a purchase price of $1,000 per unit. Each unit consisted of one share of Series A Preferred, convertible into 400 shares of common stock, and a detachable warrant to purchase 200 shares of common stock at an exercise price of $4.00 per share. Gross proceeds from the private placement amounted to $7,000,000 and net proceeds amounted to approximately $6,000,000. As a result of the subsequent private placement, in September 2007 (see below), a Series A Preferred is now convertible into 830.36 shares of the Company’s common stock and the detachable warrant to purchase 200 shares of common stock now has an exercise price of $1.92 per share.
On September 17, 2007, the Company completed a private placement of 4,350 shares of Series B convertible preferred stock, par value $0.001 per share, and warrants to purchase the Company’s common stock, par value $0.001 per share, for an aggregate purchase price of $4,350,000. At the time of the closing, the Company was required to restrict cash equal to ten percent of the stated value of the Series B convertible preferred stock that are outstanding, or $435,000, for the payment of dividends plus $200,000 for the payment of investor relation services over the next two years. As of December 31, 2008, the Company no longer has cash restricted for dividend payments and investor relations services. Dividend payments will continue to be made as long as the Series B convertible preferred shares are outstanding.
F-13
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
Each share of the Series B convertible preferred stock is initially convertible, at the holder’s election, into approximately 830.36 shares of common stock and has a liquidation preference that is pari passu with the Company’s Series A convertible preferred stock and senior to the Company’s common stock. Cumulative dividends on the Series B convertible preferred stock accrue on the initial stated value of $1,000 per share at an annual rate of 8%, payable monthly in cash and/or shares of the Company’s common stock, at the option of the Company. As of December 31, 2008, cash dividend payments amounted to $338,827 and there were no accrued but unpaid dividends to Series B convertible preferred stockholders.
The warrants have a seven-year term and are initially exercisable into 2,167,232 shares of common stock. The warrants are exercisable, at the holder’s election, for shares of the Company’s common stock in either a cash or cashless exercise. Half of the warrants have an initial exercise price equal to $1.2043 per share and the other half have an initial exercise price of $1.3849 per share. The Company also issued warrants to the financial advisor in the transaction, Canaccord Adams, Inc., to purchase 67,530 shares of common stock, which was recorded at fair value of approximately $57,000 and was recognized as additional paid in capital. The value of the warrants was computed using the Black-Scholes option-pricing model with the following assumptions: risk free rate of 4.21%, contractual term of seven years, expected volatility of 69.90%, 0% expected dividend yield, stock price of $1.23 per share, and exercise prices of $1.2043 and $1.3849 per share.
In accordance with EITF Topic D-98, “Classification and Measurement of Redeemable Securities,” the Series A and B convertible preferred stock was classified as an equity instrument since all events requiring redemption are within the control of the Company, and therefore, would not result in the security being classified outside of permanent equity.
The conversion feature of the Series A and B convertible preferred stock and warrants to purchase common stock were evaluated in accordance with SFAS 133, “Accounting for Derivative Instruments and Hedging Activities” and EITF 00-19, “Accounting for Derivative Financial Instruments Indexed to, and Potentially Settled in, a Company’s Own Stock,” and were determined to be equity instruments. The characteristics of the Series A and B warrants which allowed for equity treatment are as follows: (1) the contract permits the Company to settle in unregistered shares; (2) the Company has sufficient authorized and unissued shares available to settle the warrants after considering all other commitments that may require the issuance of stock during the maximum period of the warrant; (3) the warrant contains an explicit limit on the number of shares to be delivered in a share settlement; and, (4) there are no required cash payments to the counterparty in the event the Company fails to make timely filings with the SEC.
In accordance with EITF 98-5, “Accounting for Convertible Securities with Beneficial Conversion Features or Contingently Adjustable Conversion Ratios” and EITF 00-27, “Application of Issue No. 98-5 to Certain Convertible Instruments,” the Series B convertible preferred stock was considered to have an embedded beneficial conversion feature because the conversion price was less than the fair value of the Company’s common stock at the issuance date. This beneficial conversion feature is calculated after the warrants have been valued with proceeds allocated on a relative fair value basis. A deemed dividend of approximately $1,381,163 relating to the beneficial conversion feature was recognized in the Company’s results of operations for the period ended December 31, 2007.
Stock Options
The Company’s 2001 Long-Term Incentive Plan, 2005 Long-Term Incentive Plan and 2006 Long-Term Incentive Plan have a total of 4,676,868 stock option grants made to directors, officers and employees of the Company. As of December 31, 2008, 196,202 stock options remained eligible for grant under the 2006 Long-Term Incentive Plan. The 2001 and 2005 Long-Term Incentive Plans have been closed for additional grants.
During the years ended December 31, 2008 and 2007, the Company granted options to purchase 1,124,432 and 735,460 common stock, respectively, to employees at exercise prices of $0.48, $0.62, $0.69, and $1.01, which approximated the fair value on the respective grant dates. Shares granted after November 4, 2005 (the exchange transaction date) were made under the 2006 VirtualScopics, Inc., Long Term Incentive Plan. These options generally vest ratably during the first four years following their issuance and have a ten-year life.
F-14
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
A summary of the option activity for the year ended December 31, 2007 and 2008 are as follows:
Number of Shares | Weighted Average Exercise Price | Weighted- Average Remaining Contractual Term | Aggregate Intrinsic Value | |||||||||||||
| Options outstanding at January 1, 2007 | 3,129,051 | $ | 2.22 | |||||||||||||
| Granted | 735,460 | $ | 1.49 | |||||||||||||
| Exercised | (25,000 | ) | $ | (0.74 | ) | |||||||||||
| Cancelled | (48,780 | ) | $ | (3.62 | ) | |||||||||||
| Options outstanding at December 31, 2007 | 3,790,731 | $ | 2.12 | 6.92 | $ | 128,155 | ||||||||||
| Granted | 1,124,432 | $ | 0.95 | |||||||||||||
| Cancelled | (238,295 | ) | $ | (1.70 | ) | |||||||||||
| Options outstanding at December 31, 2008 | 4,676,868 | $ | 1.64 | 6.53 | $ | 4,200 | ||||||||||
| Options exercisable at December 31, 2008 | 2,988,866 | 1.98 | 5.97 | $ | 0.00 | |||||||||||
Additional information with respect to the outstanding options as of December 31, 2008 is as follows:
| Options Outstanding | Options Exercisable | |||||||||||||||||||
| Exercise Prices | Number Outstanding at December 31, 2008 | Weighted Average Remaining Life (Years) | Weighted Average Exercise Price | Number Exercisable at December 31, 2008 | Weighted Average Exercise Price | |||||||||||||||
| $ 0.48 - 0.74 | 1,782,672 | 6.47 | .86 | 803,422 | 0.77 | |||||||||||||||
| $ 1.20 - 1.40 | 519,460 | 7.61 | 1.24 | 250,210 | 1.29 | |||||||||||||||
| $ 1.88 - 1.90 | 254,330 | 3.65 | 1.90 | 245,329 | 1.90 | |||||||||||||||
| $ 2.36 - 2.50 | 1,446,406 | 6.32 | 2.40 | 1,352,905 | 2.39 | |||||||||||||||
| $ 3.03 - 6.70 | 674,000 | 7.39 | 3.81 | 337,000 | 3.81 | |||||||||||||||
| 4,676,868 | 6.53 | 1.64 | 2,988,866 | 1.98 | ||||||||||||||||
The weighted-average grant-date fair value of options granted during the year ended December 31, 2008 and 2007 was $1,065,431 and $1,097,392, respectively.
For the year ended December 31, 2008, the Company recorded a total of $1,123,283 in stock-based compensation expense, which is included in the general and administrative expenses in the consolidated financial statements.
F-15
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
A summary of the status of the non-vested shares as of December 31, 2008 and changes during the year ended December 31, 2008, is presented below:
| Non-vested Shares | Shares | Weighted-Average Grant-Date Fair Value Per Share | ||||||
| Non-vested at January 1, 2008 | 1,323,153 | $ | 2.53 | |||||
| Granted | 1,124,432 | 0.95 | ||||||
| Vested | (521,287 | ) | (2.36 | ) | ||||
| Cancelled Grants | (238,295 | ) | (1.70 | ) | ||||
| Non-vested at December 31, 2008 | 1,688,003 | |||||||
As of December 31, 2008, there was $1,732,222 of total unrecognized compensation cost related to non-vested share-based compensation arrangements. This cost is expected to be recognized over a weighted-average period of 2.67 years. The total fair value of shares vested during the year ended December 31, 2008 amounted to $1,230,866.
During the years ended December 31, 2008 and 2007, the Company issued 0 and 1,196 options under the 2006 Long-Term Incentive Plan to non-employee consultants valued at $0 and $1,818, respectively, for radiological services performed. These options to non-employees vest immediately, have exercise prices ranging from $1.12 to $1.81 and a term of seven or six years from the date of grant. The value of the options was based on the fair value of the services performed and is included in the Company’s statements of operations.
The total amount of stock options outstanding as of December 31, 2008 is:
| Stock options granted to employees | 4,676,868 | |||
| Stock options granted to consultants | 141,690 | |||
| Total outstanding | 4,818,558 |
Restricted Stock Awards
Under the provisions of the 2006 Long Term Incentive Plan, the Company may grant restricted stock to its employees, Board members and consultants. During 2006, the Board of Directors Compensation Committee approved an equity based compensation structure for the Board members under a plan to be created. As a result, the Company has reserved $229,634 related to restricted stock awards that will be given to certain Board members, on pre-determined dates, in lieu of cash for their services as Board members during 2006, 2007 and 2008.
NOTE 7 - Benefit Plan
The Company has a defined contribution plan which covers all of its full-time employees. The employees’ annual contributions are limited to the maximum allowed under the Internal Revenue Code. Currently, there is no Company match for employee contributions.
NOTE 8 - Income Taxes
Effective January 1, 2007, the Company adopted the provisions of FASB Interpretation No. 48, “Accounting for Uncertainty in Income Taxes, an interpretation of FASB Statement No. 109” (“FIN No. 48”). FIN No. 48 clarifies the accounting for uncertainty in income taxes recognized in an enterprise’s financial statements in accordance with FASB Statement 109, “Accounting for Income Taxes,” and prescribes a recognition threshold and measurement process for financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return.
F-16
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
The Company has identified its federal tax return and its state tax return in New York as “major” tax jurisdictions, as defined. Based on the Company’s evaluation, it has been concluded that there are no significant uncertain tax positions requiring recognition in the Company’s consolidated financial statements. The Company’s evaluation was performed for the tax years ended 2001 through 2008, the only periods subject to examination. The Company believes that its income tax positions and deductions will be sustained on audit and does not anticipate any adjustments that will result in a material change to its financial position. In addition, the Company did not record a cumulative effect adjustment related to the adoption of FIN 48. The Company does not expect its unrecognized tax benefit to change during the next 12 months. As of December 31, 2008, all of the Company’s deferred tax assets were fully reserved by a valuation allowance equal to 100% of the net deferred tax assets. The Company has never been profitable and has not paid any income taxes.
The Company has significant net operating loss and business credit carryovers which are subject to a valuation allowance due to the uncertain nature of the realization of the losses. The Internal Revenue Code imposes certain limitations on the utilization of net operating loss carryovers and other tax attributes after a change in control. The Company encountered ownership changes which could significantly limit the possible utilization of such carryovers. The Company has not performed a detailed analysis to determine the effect of any such ownership changes on its ability to use these net operating loss and credit carryforwards. However, it is not anticipated that limitations, if any, may have a material impact on the consolidated balance sheet as a result of offsetting changes in the deferred tax valuation allowance.
The Company will recognize interest and penalties accrued related to unrecognized tax benefits as components of its income tax provision. The Company did not have any interest and penalties accrued upon the adoption of FIN No. 48 and as of December 31, 2008, the Company does not have any interest and penalties accrued related to unrecognized tax benefits.
The Company has net operating loss carryforwards (“NOLs”) of approximately $9,145,000 as of December 31, 2008 that will be available to offset future taxable income. Approximately $9,000 of the NOL carryforward, if realized, will result in a benefit to be recorded in APIC. The NOLs are due to expire in 2025 - 2028. The Company has concluded that a full valuation allowance was appropriate for the NOLs as they are more likely than not to be utilized prior to their expiration.
The total net deferred tax asset as of December 31, 2008 and 2007 consists of the following:
| 2008 | 2007 | |||||||
| Net operating loss carryforwards | $ | 3,537,789 | $ | 2,361,923 | ||||
| Intangible assets | 1,274,223 | 1,729,297 | ||||||
| Accrued expenses | 114,704 | 84,142 | ||||||
| Stock-based compensation | 1,204,814 | 1,098,484 | ||||||
| Total deferred tax asset | 6,131,530 | 5,273,846 | ||||||
| Deferred tax liability: | ||||||||
| Property and equipment | (18,517 | ) | (35,167 | ) | ||||
| Subtotal | 6,113,013 | 5,238,679 | ||||||
| Less: valuation allowance | (6,113,013 | ) | (5,238,679 | ) | ||||
| Total net deferred tax asset | $ | - | $ | - | ||||
F-17
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
The difference between the federal statutory and effective income tax rates for the years ended December 31, 2008 and 2007 is as follows:
| 2008 | 2007 | |||||||
| Federal statutory tax rate | 34.00 | % | 34.00 | % | ||||
| State and local income taxes, net of federal benefit | 4.95 | % | 4.95 | % | ||||
| Effect of permanent differences | (4.78 | )% | 1.93 | % | ||||
| Other | (0.30 | )% | - | |||||
| 33.87 | % | 40.88 | % | |||||
| Less: valuation allowance | (33.87 | )% | (40.88 | )% | ||||
| Provision for income taxes | 0.00 | % | 0.00 | % | ||||
NOTE 9 - Related Parties
On June 26, 2002, the Company entered into a multi-year strategic relationship with Pfizer Inc. (“Pfizer”) to accelerate the discovery, validation and application of image-based biomarkers for clinical research. Under the terms of the agreement, Pfizer invested $2,500,000 in the Company, for a 10% ownership interest. Of the Pfizer investment, $750,000 was paid in advance in conjunction with the signing of the agreement. The Company used $1,500,000 of the investment to purchase the intellectual property that it was licensing from the University of Rochester. In August 2005, the agreement was extended for another two years and provided for termination by Pfizer by giving a 30-day advance written notice. Additionally, in November 2006, the agreement was further extended until July 2008, with automatic renewals unless either party provides written notice 60 days prior to the next anniversary.
In December 2002, the Company received an investment of $2,450,000 from GE Medical Systems. Upon receipt of the proceeds of this investment, the Company purchased an MRI machine from this investor for $2,300,000. During 2003, the equipment was sold to the University of Rochester, a related party, for $2,300,000. The Company retained the right to use the machine exclusively one day a week for the next seven years. The balance of $1,050,000 was applied as an advance payment for use of the equipment and is recorded as an other asset on the balance sheet as of December 31, 2008 and is being amortized based on usage over the life of the agreement. The equipment is being used by the Company for research and to broaden its ability to service its customers.
NOTE 10 - Major Customers
The Company derived 9% and 28% of its revenue from one of its customers, which is also a stockholder, for the years ended December 31, 2008 and 2007, respectively. This customer was the Company’s largest customer in 2007. The Company’s largest three customer in 2008 accounted for 18%, 12% and 12% of the 2008 revenues and 23%, 6% and 10%, respectively of the Company’s accounts receivable at December 31, 2008.
NOTE 11 - Concentration of Credit Risk
Financial instruments that subject the Company to concentrations of credit risk consist primarily of cash, cash equivalents, restricted cash and accounts receivable. The Company maintains all of its cash, cash equivalents, and restricted cash in two financial institutions, although substantially all of the balance is within one institution. The Company performs periodic evaluations of the relative credit standing of the two institutions. The cash balances are insured by either the FDIC, up to $250,000 per depositor, or the U.S. Department of Treasury’s Temporary Guarantee Program for Money Market Funds. The Company has cash balances on deposit with two financial institutions at December 31, 2008 that exceed the insured limit by the amount of $2.9 million. The Company’s customers are primarily concentrated in the United States. The Company performs ongoing credit evaluations, generally does not require collateral, and establishes an allowance for doubtful accounts based upon factors surrounding the credit risk of customers, historical trends and other information.
F-18
VirtualScopics, Inc. and Subsidiary
Notes to Consolidated Financial Statements
NOTE 12 – Restricted Cash
As part of the private placement of Series B convertible preferred stock, in September 2007, the Company reserved ten percent of the stated value of the Series B convertible preferred stock that were outstanding, or $435,000, for the payment of dividends. At that time, the Company also reserved $200,000 for the payment of investor relation services over the next two years. The restricted cash balance as of December 31, 2008 and 2007 amounted to $0 and $498,799, respectively, of which $43,216 was classified as a non-current asset as of December 31, 2007.
NOTE 13 – Other Matters
In February 2008, the Company received a written notice from the Nasdaq Stock Market indicating that its common stock was not in compliance with minimum bid price required for continued listing on the Nasdaq Capital Market. Nasdaq has granted the company additional time to regain compliance and as of the date of this report, the Company has until August 11, 2009 to regain compliance by maintaining a bid price of common stock at a close of $1.00 or higher for a minimum of 10 consecutive business days, or such longer period as Nasdaq may determine to show the ability to maintain long-term compliance. The Company’s intention is to maintain its listing on Nasdaq and will continue to assess its options in maintaining its listing.
F-19