Table of Contents
| Delaware | 2834 | 20-8099512 | ||
| (State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification No.) |
| Marc Rubenstein, Esq. Ropes & Gray LLP One International Place Boston, MA 02110-2624 Phone: (617) 951-7000 Fax: (617) 951-7050 | Benjamin S. Levin, Esq. CytRx Corporation 11726 San Vicente Boulevard, Suite 650 Los Angeles, CA 90049 Phone: (310) 826-5648 Fax: (310) 826-6139 | Dale E. Short, Esq. Troy & Gould Professional Corporation 1801 Century Park East, 16thFloor Los Angeles, CA 90067 Phone: (310) 789-1259 Fax: (310) 789-1459 |
| Proposed Maximum | Proposed Maximum | |||||||||||
| Title of Each Class of | Amount to be | Offering Price | Aggregate Offering | Amount of | ||||||||
| Securities to be Registered | Registered | per Share(1) | Price(1) | Registration Fee | ||||||||
| Common Stock, $0.0001 par value per share | 10,823,199(2) | $1.02 | $11,039,663 | $338.92 | ||||||||
| (1) | Estimated solely for the purpose of calculating the amount of the registration fee pursuant to Rule 457(o) under the Securities Act, and assumes that the value of the shares to be issued in the distribution described herein will be determined by the currently estimated net book value of the registrant at the time of the distribution. | |
| (2) | Represents the aggregate number of shares of Common Stock to be distributed by CytRx Corporation to holders of CytRx Corporation common stock, to be awarded by CytRx Corporation to directors, officers and employees of CytRx Corporation and registered for resale by CytRx Corporation. |
Table of Contents
| • | a prospectus (the “Distribution Prospectus”) to be used for the distribution by CytRx Corporation of 4,526,624 shares of the Registrant’s common stock to the holders of common stock of CytRx Corporation. | |
| • | a prospectus (the “Award Prospectus”) to be used for the award by CytRx Corporation of up to 83,714 shares of the Registrant’s common stock to some of CytRx Corporation’s directors, officers and employees. | |
| • | a prospectus (the “Resale Prospectus”) to be used for the resale by CytRx Corporation of up to 6,212,861 shares of the Registrant’s common stock owned by CytRx Corporation after the distribution and award transactions pursuant to the Distribution Prospectus and the Award Prospectus, respectively. |
| • | they contain different outside and inside front covers and back covers; | |
| • | they contain different introductory paragraphs to the “Summary” section beginning onpage A-1 andpage B-1, respectively, and different discussions of the transactions covered by the Award Prospectus and Resale Prospectus in the “Summary” section onpage A-1 andpage B-1, respectively; | |
| • | they contain different “Risk Factors” beginning onpage A-10 andpage B-8, respectively; | |
| • | each prospectus has a different section describing the transaction and plan of distribution related thereto: the Distribution Prospectus contains a section entitled “The Distribution” beginning on page 27, the Award Prospectus instead has a section entitled “The Award” beginning onpage A-26, and the Resale Prospectus contains a section entitled “Plan of Distribution” beginning onpage B-81; | |
| • | the Award Prospectus refers throughout to the Distribution and the Resale Prospectus, and the Resale Prospectus refers throughout to the Distribution Prospectus and the Award Prospectus; | |
| • | the Award Prospectus and the Resale Prospectus contain different “Use of Proceeds” sections onpage A-29 andpage B-23, respectively; | |
| • | the Award Prospectus contains a different “Material Federal Income Tax Consequences” section than the Distribution Prospectus, beginning onpage A-90, and the Resale Prospectus omits this section; and | |
| • | the Resale Prospectus includes a “Selling Stockholder” section beginning onpage B-80. |
Table of Contents
| We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities, and we are not soliciting an offer to buy these securities in any state where the offer or sale is not permitted. |

Table of Contents
| • | Neurology. Initially, we are pursuing research in ALS (amyotrophic lateral sclerosis, commonly known as Lou Gehrig’s Disease). Some forms of ALS are caused by defects in a gene called SOD1. Early preclinical studies conducted by RXi advisors, Dr. Tariq Rana and Dr. Zuoshang Xu at UMMS, showed promising results in animals using an RNAi compound to selectively inhibit the SOD1 gene. We are refining and extending this work and, if successful, will move into formal preclinical development. We also intend to leverage our experience related to the delivery of RNAi therapeutics in the central nervous system to explore development of RNAi-based treatments for neurodegenerative diseases other than ALS, including Alzheimer’s Disease. | |
| • | Metabolic disease. One of our scientific co-founders and scientific advisory board members, Dr. Michael Czech, is a leading metabolic disease researcher. We have in-licensed intellectual property developed by Dr. Czech on genes that appear to be important regulators of metabolism. Studies conducted in Dr. Czech’s laboratory at UMMS and by others at Imperial College of London have demonstrated that inactivation of one of these genes, called RIP140, can cause fat cells to metabolize rather than store fat. Mice in these studies that did not express RIP140 remained lean and non-diabetic even when maintained on a high fat diet. We are currently designing RNAi compounds targeting |
1
Table of Contents
| RIP140 as a potential treatment for obesity and obesity-related type 2 diabetes. We also continue to evaluate genes in Dr. Czech’s database for candidate targets. |
| • | Oncology. We are initiating a program to develop RNAi drugs for use in oncology. This strategy is led by key RXi scientific advisors, Dr. Greg Hannon and Dr. Nicholas Dean, both of whom are leading researchers in targeting oncogene pathways. Additionally, our management team has expertise in developing programs targeting genes involved in cancer. Dr. Pamela Pavco, our Vice President for Pharmaceutical Development, previously managed the pre-clinical programs targeting genes involved with cancer while at Sirna Therapeutics, Inc. (recently acquired by Merck & Co.). | |
| • | Additional indications. There are many well-studied genes associated with numerous diseases that have been identified but have been difficult to target with normal medicinal chemistry. We believe RNAi technology may play an important role in targeting these genes and potentially treating these diseases. With the pioneering work being done in developing the RXi technology platform, we believe that we will discover many more drug candidates than can be advanced into clinical trials. For research on target genes in our portfolio that are not funded internally, we will seek to identify and work with partners in the discovery and development process to build our development pipeline. |
| • | World-class scientific advisors with extraordinary experience in RNAi, including Dr. Craig Mello, recipient of the 2006 Nobel Prize in Medicine for co-discovering RNAi. | |
| • | Strong early intellectual property position covering |
| • | Key therapeutic targets, | |
| • | Proprietary, potent nanotransporter delivery of active RNAi to tissues, and | |
| • | Novel approaches to RNAi chemistry. |
| • | Initial focus on significant unmet medical needs, including |
| • | Neurological disorders, | |
| • | Metabolic disease, and | |
| • | Oncology. |
| • | Management that is experienced in commercializing products, including our President and Chief Executive Officer, Tod Woolf, Ph.D. | |
| • | Potentially rapid route to the clinic for ALS therapeutics due to the high mortality rate. |
| • | We intend to finance the initial development of a limited number of RNAi drug candidates with our own capital resources and any financial resources that we may obtain from capital markets and partners. Our key therapeutic areas of interest are neurology, metabolic disease and oncology. We intend to |
2
Table of Contents
| develop drugs in these areas internally to establish significant value, at which point we may seek to partner them. |
| • | We intend to seek partnerships with large pharmaceutical and biotechnology companies to leverage our intellectual property and expand our development pipeline. Such partnerships may include traditionally structured drug development and commercialization licenses, discovery and development collaborations, research and technology collaborations, and intellectual property licenses. | |
| • | We intend to maintain and continue to develop and enhance our RNAi technology platform by expanding our intellectual property position in RNAi compound chemistry, delivery and target sequences. To date, we have in-licensed RNAi technologies from various institutions and companies, including UMMS, Imperial College of London, Cold Spring Harbor Laboratories and TriLink Biotechnologies. We intend to continue to enhance our technology platform through in-licensing in combination with internal and collaborative research and development programs. | |
| • | We intend to develop future RNAi technology improvements and believe we are well positioned to do so. Our management and advisors have developed much of the core technology in the field of oligonucleotide therapeutics, and more specifically RNAi. For example, our Scientific Advisory Board member, Dr. Nassim Usman, developed the standard synthesis chemistry used to manufacture RNAi throughout the world while a fellow at Massachusetts Institute of Technology (MIT). Our CEO, Dr. Tod Woolf, co-developed STEALTH RNAi, which is one of the most commonly employed second generation RNAi chemistries, and our Vice President of Pharmaceutical Development, Dr. Pamela Pavco, developed the first modified RNAi tested in humans while she was at Sirna Therapeutics. Our advisors and scientists routinely meet to discuss novel approaches and improvements in our RNAi technology platforms to enhance our intellectual property portfolio. | |
| • | We may also seek to collaborate with government and charitable institutions through grants and funded research for our development programs. |
| • | we may be unable to achieve some or all of the benefits that we expect to achieve from our separation from CytRx; | |
| • | we may not be able to obtain sufficient funding and may not be able to commercialize our product candidates; | |
| • | the approach we are taking to discover and develop novel therapeutics using RNAi is unproven and may never lead to marketable products; | |
| • | we may not be able to maintain the third-party relationships that are necessary to develop or commercialize some or all of our product candidates; | |
| • | we currently have no compounds in pre-clinical toxicology studies, and we may not be able to advance any product candidate through the pre-clinical stage into clinical trials; | |
| • | if our pre-clinical testing does not produce successful results or our clinical trials do not demonstrate safety and efficacy in humans, our products may not receive approval by the FDA or similar foreign governmental agencies and we will not be able to commercialize our drug candidates; | |
| • | even if we receive regulatory approval to market our product candidates, our product candidates may not be accepted commercially, which may prevent us from becoming profitable; and | |
| • | we are dependent on technologies we license, and if we lose the right to license such technologies or we fail to license new technologies in the future, our ability to develop new products would be harmed. |
3
Table of Contents
4
Table of Contents
| Distributing Company | CytRx Corporation, a Delaware corporation. | |
| Distributed Company | RXi Pharmaceuticals Corporation, a Delaware corporation. | |
| Securities to Be Distributed | A fixed number of 4,526,624 shares of our common stock will be distributed to CytRx stockholders, which constitutes approximately 36% of the outstanding shares of our common stock. | |
| Other Transactions | Concurrently with the distribution, CytRx is awarding approximately 83,714 outstanding shares of RXi common stock to some of its directors, officers and employees by means of the award prospectus. Immediately following the distribution made under this prospectus and the award made under the award prospectus, CytRx will own approximately 6,212,861 shares of our common stock, or approximately 49% of our outstanding shares, all of which shares are concurrently being registered for resale by CytRx by means of the resale prospectus. | |
| Reasons for the Distribution | CytRx’s lead therapeutic programs are based on small molecule drugs, while ours are focused on RNAi. The discovery and development of small molecule drugs requires skills, patents and management expertise that are different from the skills, patents and management expertise that are required for RNAi drug development and discovery. Because of the difference in focus with respect to therapeutic programs, and the difficulties that arise in adequately funding and managing programs in both of these areas, the CytRx board of directors determined that the best strategy for realizing the potential value of CytRx’s RNAi assets was to contribute them to us, with our concentration on developing and commercializing RNAi therapeutics. In furtherance of this strategy, the CytRx board believes that the partial distribution of our common stock will, among other things: |
| • | allow us direct access to public capital markets to finance our RNAi drug development activities, | |
| • | establish us as one of the leading companies dedicated to developing proprietary RNAi therapeutics and enhance our ability to compete with other companies in this field, | |
| • | allow our management and management of CytRx to each pursue their own separate business strategies and strategic relationships, | |
| • | enhance the ability of CytRx and us to attract advisors and collaborators, including collaborators who may be competitors of the other company, | |
| • | facilitate acquisitions, joint ventures and partnerships by CytRx and us with other companies focusing on the same or complementary technologies, | |
| • | provide CytRx stockholders with a direct ownership interest in us in addition to the approximately 49% indirect interest in us that they will have as CytRx stockholders immediately following the distribution under this prospectus and the award under the award prospectus, |
5
Table of Contents
| • | enhance public disclosures regarding CytRx and us and improve investor understanding of the two companies, and | |
| • | allow for our own common stock options and other equity securities in order to enable us to provide incentives for management and other key employees that are directly related to the market performance of our publicly traded shares. |
| The CytRx board believes that the partial distribution will also satisfy CytRx’s obligations under its existing agreements with UMMS, our principal RNAi collaborator, and with our scientific advisors and other founding shareholders, to reduce its ownership of RXi to less than 50%. | ||
| Distribution Ratio | Based on shares of CytRx common stock outstanding on the date of this prospectus, holders of CytRx common stock as of the close of business (Eastern time) on , 2007, the record date for the distribution, which we anticipate will be approximately ten days after the effectiveness of the registration statement of which this prospectus is a part, will receive a dividend of one share of our common stock for approximately every shares of CytRx common stock they hold. This distribution ratio assumes that no outstanding common stock options or stock purchase warrants of CytRx will be exercised between the date of this prospectus and the record date. The actual distribution ratio may be less than one-for- , depending on the number of shares of CytRx common stock outstanding on the record date. | |
| Manner of Effecting the Distribution | The distribution will consist of CytRx’s payment of a dividend to CytRx stockholders in the form of shares of RXi common stock. No vote or approval of CytRx stockholders is required in connection with the distribution. You will not be required to make any payment or to surrender or exchange your shares of CytRx common stock or take any other action to receive your shares of our common stock. If you own CytRx common stock as of the close of business on the record date, CytRx, with the assistance of Computershare, the distribution agent, will electronically issue shares of our common stock to you or to your brokerage firm on your behalf by way of direct registration in book-entry form. Computershare will mail to you a book-entry account statement that reflects your shares of RXi common stock, or your bank or brokerage firm will credit your account for the shares. If you sell shares of CytRx common stock in the “regular-way” market up to and including through the distribution date, you will be selling your right to receive shares of RXi common stock in the distribution. Following the distribution, stockholders whose shares of RXi common stock are held in book-entry form may request that their shares be transferred to a brokerage or other account at any time, without charge. Please see “The Distribution — Direct Registration System” for a more detailed description of the direct registration system and how shares of CytRx common stock may be sold and transferred. | |
| Fractional Shares | No fractional shares of RXi common stock will be distributed to CytRx shareholders. Instead, fractional shares will be sold in the public market by the distribution agent. The aggregate net proceeds of these sales will be distributed ratably, after making certain |
6
Table of Contents
| deductions, to those CytRx stockholders who would otherwise have received fractional shares. For additional information, see “The Distribution — Treatment of Fractional Shares.” | ||
| Record Date | The record date for distribution is the close of business (Eastern time) on , 2007 which we anticipate will be approximately ten days after the effectiveness of the registration statement of which this prospectus is a part. | |
| Expected Distribution Date | , 2007. | |
| Distribution Agent, Transfer Agent andRegistrar for theShares | Computershare Investor Services will be the distribution agent for the distribution and the transfer agent and registrar for RXi common stock following the distribution. | |
| United States Federal Income TaxConsequences of theDistribution | CytRx expects that the distribution will be treated as a taxable distribution in an amount equal to the sum of the fair market value of our shares on the distribution date and any cash received in lieu of fractional shares. This sum will be treated as a dividend to the extent of any current year earnings and profits of CytRx, including gain resulting from the distribution, with the excess treated as a non-taxable return of capital to the extent of a holder’s tax basis in CytRx common stock and any remaining excess treated as capital gain. For a discussion of the tax consequences to CytRx and CytRx stockholders of the distribution, see the “Material United States Federal Income Tax Considerations” section of this prospectus. CytRx stockholders should consult with their own individual tax advisors regarding the tax consequences of the distribution. | |
| Stock Exchange Listing | There is not currently a public market for our common stock. We have applied for listing of our common stock on the NASDAQ Capital Market under the symbol “RXIP.” We anticipate that trading will commence on , 2007. We cannot predict the trading prices for our common stock before or after the distribution date. | |
| Relationship With CytRx after theDistribution | Immediately following the distribution made under this prospectus and the award made under the award prospectus, CytRx will own approximately 49% of our common stock. We are a party to a number of agreements with CytRx relating to its ownership of our common stock and other matters. Please see “Arrangements With CytRx Corporation.” | |
| Post-Distribution Dividend Policy | RXi has never declared a dividend on its common stock and does not plan to do so for the foreseeable future. | |
| Risk Factors | You should carefully consider the matters discussed under the heading “Risk Factors.” |
7
Table of Contents
| Where CytRx Stockholders CanObtain MoreInformation | If you have any questions relating to the separation of RXi from CytRx, you should contact: | |
| Benjamin S. Levin General Counsel, Vice President — Legal Affairs CytRx Corporation 11726 San Vicente Boulevard, Suite 650 Los Angeles, California 90049 Tel:(310) 826-5648 x313 Fax:(310) 826-6139 | ||
| If you have any questions relating to the distribution of the RXi shares to CytRx stockholders, you should contact: | ||
| Computershare Investor Services 250 Royall Street Canton, MA 02021 Tel: (781) [ ] Fax: (781) [ ] |
8
Table of Contents
9
Table of Contents
| Period from | ||||||||||||||||||||||||||||
| Period from | January 1, 2003 | |||||||||||||||||||||||||||
| April 3, 2006 | (Date of | |||||||||||||||||||||||||||
| (Date of | Inception) | |||||||||||||||||||||||||||
| Incorporation) | through | |||||||||||||||||||||||||||
| through June 30, | Six Months Ended June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||
| 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||||||||
| (Successor) | (Successor) | (Predecessor) | (Predecessor) | (Predecessor) | ||||||||||||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||||||||||||||
| (All amounts in thousands, except share and earnings per share data) | ||||||||||||||||||||||||||||
Statements of Expenses Data: | ||||||||||||||||||||||||||||
| Revenue | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Expenses: | ||||||||||||||||||||||||||||
| Research and development | 4,594 | 4,594 | 907 | 8,541 | 1,772 | 2,080 | 2,814 | |||||||||||||||||||||
| General and administrative | 1,710 | 1,710 | 155 | 1,500 | 633 | 129 | 458 | |||||||||||||||||||||
| Operating loss | (6,304 | ) | (6,304 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Interest income | 133 | 133 | — | — | — | — | — | |||||||||||||||||||||
| Loss before income taxes | (6,171 | ) | (6,171 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Income taxes | — | — | — | — | — | — | — | |||||||||||||||||||||
| Net loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | $ | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | |||||||
| Basic and diluted loss per share | $ | (0.64 | ) | $ | (0.64 | ) | NA | NA | NA | NA | NA | |||||||||||||||||
| Weighted average shares, basic and diluted | 9,671,849 | 9,671,849 | NA | NA | NA | NA | NA | |||||||||||||||||||||
Cash Flows Data: | ||||||||||||||||||||||||||||
| Cash flows used in operating activities | $ | (1,713 | ) | $ | (1,713 | ) | $ | (1,254 | ) | $ | (6,712 | ) | $ | (2,112 | ) | $ | (2,527 | ) | $ | (1,280 | ) | |||||||
| Cash flows used in investing activities | $ | (19 | ) | $ | (19 | ) | $ | — | $ | (50 | ) | $ | — | $ | (50 | ) | $ | — | ||||||||||
| Cash flows provided by financing activities | $ | 16,695 | $ | 16,694 | $ | 1,248 | $ | 6,762 | $ | 2,112 | $ | 2,577 | $ | 1,280 | ||||||||||||||
| As of | As of | |||||||||||||||||||||||
| June 30, | December, | As of December 31, | ||||||||||||||||||||||
| 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | |||||||||||||||||||
| (Successor) | (Predecessor) | |||||||||||||||||||||||
| (Unaudited) | (Successor) | (Predecessor) | ||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||
Balance Sheets and Statements of Assets, Liabilities and Parent Company’s Net Deficit: | ||||||||||||||||||||||||
| Cash and cash equivalents | $ | 14,965 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Total current assets | $ | 14,980 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Equipment and furnishings, net | $ | 59 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||
| Total assets | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | ||||||||||||
| Total current liabilities | $ | 2,490 | $ | 202 | $ | — | $ | 325 | $ | 500 | $ | 968 | ||||||||||||
| Total stockholders’ equity | $ | 12,599 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Total liabilities and stockholders’ equity | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | ||||||||||||
| Parent company’s net deficit | $ | — | $ | (152 | ) | $ | — | $ | (268 | ) | $ | (450 | ) | $ | (968 | ) | ||||||||
10
Table of Contents
| • | Prior to the contribution of our RNAi assets from CytRx, our RNAi research and development activities were conducted by CytRx as part of its broader operations, rather than as an independent division or subsidiary, and were primarily conducted through sponsored research arrangements rather than through internal activities. CytRx also performed various corporate functions relating to our business, including governance functions such as compliance with the Sarbanes-Oxley Act of 2002 and external reporting. Our historical financial information reflect allocations of corporate expenses from CytRx for these and similar functions. We believe that these allocations are comparable to the expenses we would have incurred had we operated as a separate publicly traded company, although we will likely incur higher expenses as an independent company; | |
| • | Our working capital requirements and capital for our general corporate purposes have historically been provided by CytRx. Following the distribution, CytRx will not be obligated to provide us with funds to finance our working capital or other cash requirements. Without financing from CytRx, we will need to obtain additional financing through public offerings or private placements of debt or equity securities, strategic relationships or other arrangements; and | |
| • | Other significant changes may occur in our cost structure, management, financing and business operations as a result of our operating as a company separate from CytRx. |
11
Table of Contents
| • | to conduct research and development to successfully develop our RNAi technologies, | |
| • | to obtain regulatory approval for our products, | |
| • | to file and prosecute patent applications and to defend and assess patents to protect our technologies, | |
| • | to retain qualified employees, particularly in light of intense competition for qualified scientists, | |
| • | to manufacture products ourselves or through third parties, | |
| • | to market our products, either through building our own sales and distribution capabilities or relying on third parties, and | |
| • | to acquire new technologies, licenses or products. |
12
Table of Contents
13
Table of Contents
14
Table of Contents
| • | delays in filing initial drug applications, | |
| • | difficulty in securing centers to conduct trials, | |
| • | conditions imposed on us by the FDA or comparable foreign authorities regarding the scope or design of our clinical trials, | |
| • | problems in engaging IRBs to oversee trials or problems in obtaining or maintaining IRB approval of studies, | |
| • | difficulty in enrolling patients in conformity with required protocols or projected timelines, | |
| • | unexpected adverse reactions by patients in trials, | |
| • | difficulty in obtaining clinical supplies of the product, | |
| • | negative or inconclusive results from our clinical trials or the clinical trials of others for drug candidates similar to our own or inability to generate statistically significant data confirming the efficacy of the product being tested, | |
| • | changes in the FDA’s requirements for our testing during the course of that testing, | |
| • | modification of the drug during testing, | |
| • | reallocation of our limited financial and other resources to other clinical programs, and | |
| • | adverse results obtained by other companies developing RNAi drugs. |
15
Table of Contents
16
Table of Contents
| • | regulators or IRBs may not authorize us to commence a clinical trial or conduct a clinical trial at a prospective trial site, | |
| • | our pre-clinical tests or clinical trials may produce negative or inconclusive results, and we may decide, or regulator may require us, to conduct additional pre-clinical testing or clinical trials, or we may abandon projects that we previously expected to be promising, | |
| • | enrollment in our clinical trials may be slower than we anticipate or participants may drop out of our clinical trials at a higher rate than we anticipate, resulting in significant delays, | |
| • | our third party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, | |
| • | our drug candidates may have very different chemical and pharmacological properties in humans than in laboratory testing and it may interact with human biological systems in unforeseen, ineffective or harmful ways. | |
| • | we might have to suspend or terminate our clinical trials if the participants are being exposed to unacceptable health risks, | |
| • | IRBs or regulators, including the FDA, may require that we hold, suspend or terminate clinical research for various reasons, including noncompliance with regulatory requirements, | |
| • | the cost of our clinical trials may be greater than we anticipate, | |
| • | the supply or quality of our drug candidates or other necessary materials necessary to conduct our clinical trials may be insufficient or inadequate, and | |
| • | effects of our drug candidates may not be the desired effects or may include undesirable side effects or the drug candidates may have other unexpected characteristics. |
17
Table of Contents
| • | the timing of our receipt of any marketing approvals, the terms of any approvals and the countries in which approvals are obtained, | |
| • | the safety, efficacy and ease of administration of our product candidates, | |
| • | the advantages of our product candidates over those of our competitors, | |
| • | the willingness of patients to accept relatively new therapies, | |
| • | the success of our physician education programs, | |
| • | the availability of government and third-party payor reimbursement, | |
| • | the pricing of our products, particularly as compared to alternative treatments, and | |
| • | the availability of effective alternative treatments and the relative risksand/or benefits of the treatments. |
18
Table of Contents
19
Table of Contents
| • | they are “incidental” to a physician’s services, | |
| • | they are “reasonable and necessary” for the diagnosis or treatment of the illness or injury for which they are administered according to accepted standard of medical practice, | |
| • | they are not excluded as immunizations, and | |
| • | they have been approved by the FDA. |
20
Table of Contents
21
Table of Contents
| • | announcements of regulatory developments or technological innovations by us or our competitors, | |
| • | changes in our relationship with our licensors and other strategic partners, | |
| • | changes in our ownership or other relationships with CytRx, | |
| • | our quarterly operating results, | |
| • | developments in patent or other technology ownership rights, | |
| • | public concern regarding the safety of our products, | |
| • | government regulation of drug pricing, and | |
| • | general changes in the economy, the financial markets or the pharmaceutical or biotechnology industries. |
22
Table of Contents
23
Table of Contents
| • | delaying, deferring or preventing a change in control of our company, | |
| • | impeding a merger, consolidation, takeover or other business combination involving our company, or | |
| • | discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of our company. |
| • | divide our board of directors into three classes, with members of each class to be elected for staggered three-year terms, | |
| • | limit the right of stockholders to remove directors, | |
| • | regulate how stockholders may present proposals or nominate directors for election at annual meetings of stockholders, and | |
| • | authorize our board of directors to issue preferred stock in one or more series, without stockholder approval. |
24
Table of Contents
25
Table of Contents
| • | development of our RNAi-based therapeutics may be delayed or may not proceed as planned, and we may not be able to complete development of any RNAi-based product, | |
| • | higher costs associated with being a separate, publicly trade company, | |
| • | the difficulty in evaluating our financial information due to the distribution, | |
| • | the inability to raise additional future financing and lack of financial and other resources to us as a separate company, | |
| • | our ability to control product development costs, | |
| • | we may not be able to attract and retain key employees, | |
| • | we may not be able to compete effectively, | |
| • | we may not be able enter into new strategic collaborations, | |
| • | changes in government regulation affecting our RNAi-based therapeutics could increase our development costs, | |
| • | our involvement in patent and other intellectual property litigation could be expensive and could divert management’s attention, | |
| • | the possibility that there will be no market acceptance for our products, and | |
| • | changes in third-party reimbursement policies could adversely affect potential future sales of any of our products that are approved for marketing. |
26
Table of Contents
| • | allow us direct access to capital markets to finance our RNAi drug development activities; | |
| • | establish us as one of the leading companies dedicated to developing proprietary RNAi therapeutics and enhance our ability to compete with other companies pursuing RNAi therapeutics; | |
| • | allow our management and management of CytRx to each pursue their own separate business strategies and strategic relationships based on their specific technologies and assets; | |
| • | enhance the ability of CytRx and us to attract advisors and collaborators such as Dr. Mello and our other scientific advisors who are leaders in the particular fields of research and development being pursued by CytRx and us, including collaborators who may be competitors of the other company; | |
| • | facilitate acquisitions, joint ventures and partnerships by CytRx and us with other companies focusing on the same or complementary technologies some of who may be competitors of the other company; | |
| • | provide CytRx stockholders with a direct ownership interest in us in addition to the approximately 49% indirect interest in us that they will have as a CytRx stockholders immediately following the distribution pursuant to this prospectus and the award pursuant to the award pursuant to the award prospectus. The CytRx board also believes that some investors invest only in companies focused on particular technologies such as RNAi, and that there may be greater collective investment demand for the publicly traded shares of CytRx and us, separately, than for CytRx shares alone. | |
| • | enhance public disclosures regarding CytRx and us and improve investor understanding of the two companies; and | |
| • | allow for common stock options and other equity securities in our company with a value related directly to the performance of our business. Such equity securities will enable us to provide incentives for our management and other key employees that are directly related to the market performance of our publicly traded shares and improve our ability to attract, retain and motivate additional qualified personnel. |
27
Table of Contents
28
Table of Contents
29
Table of Contents
30
Table of Contents
| • | a registration statement that the SEC has declared effective under the Securities Act, or | |
| • | an exemption from registration under the Securities Act, such as the exemption afforded by Rule 144, which imposes certain restrictions on such sales, including volume limitations. |
31
Table of Contents
32
Table of Contents
33
Table of Contents
| Period from | Period from | |||||||||||||||||||||||||||||||
| April 3, | January 1, | |||||||||||||||||||||||||||||||
| 2006 (Date of | 2003 (Date of | |||||||||||||||||||||||||||||||
| Incorporation) | Inception) | |||||||||||||||||||||||||||||||
| Through | Through | |||||||||||||||||||||||||||||||
| June 30, | Six Months Ended June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||||||
| 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | 2003 | |||||||||||||||||||||||||
| (Successor) | (Successor) | (Predecessor) | (Predecessor) | (Predecessor) | ||||||||||||||||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||||||||||||||||||
| (All amounts are in thousands, except shares and earnings per share data) | ||||||||||||||||||||||||||||||||
Statements of Expenses Data: | �� | |||||||||||||||||||||||||||||||
| Revenue | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||||
| Expenses: | ||||||||||||||||||||||||||||||||
| Research and development | 4,594 | 4,594 | 907 | 8,541 | 1,772 | 2,080 | 2,814 | 1,877 | ||||||||||||||||||||||||
| General and administrative | 1,710 | 1,710 | 155 | 1,500 | 633 | 129 | 458 | 278 | ||||||||||||||||||||||||
| Operating loss | (6,304 | ) | (6,304 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | (2,155 | ) | ||||||||||||||||
| Interest income | 133 | 133 | — | — | — | — | — | — | ||||||||||||||||||||||||
| Loss before income taxes | (6,171 | ) | (6,171 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | (2,155 | ) | ||||||||||||||||
| Income taxes | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||
| Net loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | $ | (2,155 | ) | |||||||||
| Basic and diluted loss per share | $ | (0.64 | ) | $ | (0.64 | ) | NA | NA | NA | NA | NA | NA | ||||||||||||||||||||
| Weighted average shares, basic and diluted | 9,671,849 | 9,671,849 | NA | NA | NA | NA | NA | NA | ||||||||||||||||||||||||
| Balance Sheets and | ||||||||||||||||||||||||||||
| Statements of Assets, | As of | |||||||||||||||||||||||||||
| Liabilities and Parent | As of June 30, | December 31, | As of December 31, | |||||||||||||||||||||||||
Company’s Net Deficit: | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | 2003 | |||||||||||||||||||||
| (Successor) | (Predecessor) | (Successor) | (Predecessor) | |||||||||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 14,965 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Total current assets | $ | 14,980 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Working capital | $ | 12,490 | $ | (202 | ) | $ | 2 | $ | (318 | ) | $ | (500 | ) | $ | (968 | ) | $ | (89 | ) | |||||||||
| Total assets | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | $ | — | ||||||||||||||
| Due to Parent | $ | (1,678 | ) | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||
| Total stockholders’ equity | $ | 12,599 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Parent company’s net deficit | $ | — | $ | (152 | ) | $ | — | $ | (268 | ) | $ | (450 | ) | $ | (968 | ) | $ | (89 | ) | |||||||||
34
Table of Contents
35
Table of Contents
| • | our ability to advance product candidates into pre-clinical research and clinical trials; | |
| • | the scope and rate of progress of our pre-clinical program and other research and development activities; | |
| • | the scope, rate of progress and cost of any clinical trials we commence; | |
| • | the cost of filing, prosecuting, defending and enforcing patent claims and other intellectual property rights; | |
| • | clinical trial results; | |
| • | the terms and timing of any collaborative, licensing and other arrangements that we may establish; | |
| • | the cost and timing of regulatory approvals; | |
| • | the cost of establishing clinical and commercial supplies of our product candidates and any products that we may develop; | |
| • | the cost and timing of establishing sales, marketing and distribution capabilities; | |
| • | the effect of competing technological and market developments; and | |
| • | the effect of government regulation and insurance industry efforts to control healthcare costs through reimbursement policy and other cost management strategies. |
36
Table of Contents
37
Table of Contents
38
Table of Contents
39
Table of Contents
40
Table of Contents
41
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| June 30, | Years Ended December 31, | |||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | ||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| Research and development expense | $ | 1,271 | $ | 802 | $ | 1,298 | $ | 1,929 | $ | 1,700 | ||||||||||
| Research and development non-employee stock-based compensation expense | 1,012 | 105 | 212 | 151 | 1,006 | |||||||||||||||
| Fair value of common stock issued in exchange for licensing rights | 2,311 | — | 262 | — | 108 | |||||||||||||||
| Total research and development expense | $ | 4,594 | $ | 907 | $ | 1,772 | $ | 2,080 | $ | 2,814 | ||||||||||
42
Table of Contents
43
Table of Contents
| Six Months Ended June 30, | Years Ended December 31, | |||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | ||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| General and administrative expense | $ | 1,381 | $ | 155 | $ | 633 | $ | 129 | $ | 458 | ||||||||||
| Common stock and stock options issued for general and administrative expense | 329 | — | — | — | — | |||||||||||||||
| Total general and administrative expense | $ | 1,710 | $ | 155 | $ | 633 | $ | 129 | $ | 458 | ||||||||||
44
Table of Contents
45
Table of Contents
| Non-Cancelable | Cancelable | |||||||||||||||||||
| Operating | Employment | License | ||||||||||||||||||
Years Ending December 31, | Leases(1) | Agreements(2) | Subtotal | Agreements(3) | Total | |||||||||||||||
| 2007 | $ | 250 | $ | 951 | $ | 1,201 | $ | 2,892 | $ | 4,093 | ||||||||||
| 2008 | 9 | 810 | 819 | 433 | 1,252 | |||||||||||||||
| 2009 | — | 710 | 710 | 383 | 1,093 | |||||||||||||||
| 2010 | — | 460 | 460 | 333 | 793 | |||||||||||||||
| 2011 | — | 255 | 255 | 333 | 588 | |||||||||||||||
| Thereafter | — | 170 | 170 | 7,568 | 7,738 | |||||||||||||||
| Total | $ | 259 | $ | 3,356 | $ | 3,615 | $ | 11,942 | $ | 15,557 | ||||||||||
| (1) | Operating leases are primarily facility and equipment related obligations with third-party vendors. Facility lease expenses during the six months ended June 30, 2007 and 2006 were approximately $68,000 and $1,000, respectively. Facility lease expense during the years ended December 31, 2006, 2005 and 2004 was $2,000, $2,000 and $3,000 respectively. | |
| (2) | Employment agreement obligations include management contracts, as well as scientific advisory board member compensation agreements. Certain agreements provide for minimum salary levels, adjusted annually at the discretion of RXi’s Board of Directors, as well as for minimum annual bonuses. | |
| (3) | License agreements primarily relate to our obligations with UMMS associated with RNAi and, for future periods, represent minimum annual royalty payment obligations. Included in the 2007 license obligations was $2,310,000 that was paid to UMMS in the second quarter of 2007 in the form of shares of common stock of RXi. |
46
Table of Contents
47
Table of Contents
| • | Neurology. Initially, we are pursuing research in ALS (amyotrophic lateral sclerosis, commonly known as Lou Gehrig’s Disease). Some forms of ALS are caused by defects in a gene called SOD1. Early preclinical studies conducted by our advisors, Dr. Tariq Rana and Dr. Zuoshang Xu at UMMS, showed promising results in animals using an RNAi compound to selectively inhibit the SOD1 gene. We are refining and extending this work and, if successful, will move into formal preclinical development. We also intend to leverage our experience related to the delivery of RNAi therapeutics in the central nervous system to explore development of RNAi-based treatments for neurodegenerative diseases other than ALS, including Alzheimer’s Disease. | |
| • | Metabolic disease. One of our scientific co-founders and scientific advisory board members, Dr. Michael Czech, is a leading metabolic disease researcher. We have in-licensed intellectual property developed by Dr. Czech on genes that appear to be important regulators of metabolism. Studies conducted in Dr. Czech’s laboratory at UMMS and by others at Imperial College of London have demonstrated that inactivation of one of these genes, called RIP140, can cause fat cells to metabolize rather than store fat. Mice in these studies that did not express RIP140 remained lean and non-diabetic even when maintained on a high-fat diet. We are currently designing RNAi compounds targeting RIP140 as a potential treatment for obesity and obesity-related type 2 diabetes. We also continue to evaluate genes in Dr. Czech’s database for candidate targets. | |
| • | Oncology. We are initiating a program to develop RNAi drugs for use in oncology. This strategy is led by key RXi scientific advisors, Dr. Greg Hannon and Dr. Nicholas Dean, both of whom are leading researchers in targeting oncogene pathways. Additionally, our management team has expertise in developing programs targeting genes involved in cancer. Dr. Pamela Pavco, our Vice President for Pharmaceutical Development, previously managed the pre-clinical programs targeting genes involved with cancer while at Sirna Therapeutics, Inc. (recently acquired by Merck & Co.). | |
| • | Additional indications. There are many well-studied genes associated with numerous diseases that have been identified but have been difficult to target with normal medicinal chemistry. We believe RNAi technology may play an important role in targeting these genes and potentially treating these diseases. With the pioneering work being done in developing the RXi technology platform, we believe |
48
Table of Contents
| that we will discover many more drug candidates than can be advanced into clinical trials. For research on target genes in our portfolio that are not funded internally, we will seek to identify and work with partners in the discovery and development process to build our development pipeline. |
| • | World-class scientific advisors with extraordinary experience in RNAi, including Dr. Craig Mello, recipient of the 2006 Nobel Prize in Medicine for co-discovering RNAi. | |
| • | Strong early intellectual property position covering |
| • | Key therapeutic targets, | |
| • | Proprietary, potent nanotransporter delivery of active RNAi to tissues, and | |
| • | Novel approaches to RNAi chemistry. |
| • | Initial focus on significant unmet medical needs, including |
| • | Neurological disorders, | |
| • | Metabolic disease, and | |
| • | Oncology. |
| • | Management that is experienced in commercializing products, including our President and Chief Executive Officer, Tod Woolf, Ph.D. | |
| • | Potentially rapid route to the clinic for ALS therapeutics due to the high mortality rate. |
49
Table of Contents
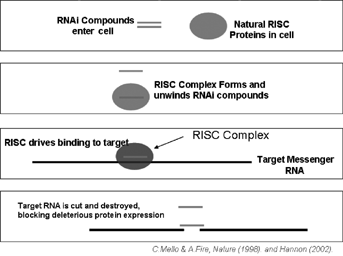
| • | high specificity for targeted genes; | |
| • | high potency (low doses); | |
| • | potential interference with the expression of any gene; and | |
| • | accelerated development of lead compounds. |
50
Table of Contents

| • | up to x100 more active than conventional siRNA (depending on the target site), | |
| • | nuclease resistant, | |
| • | readily manufactured, | |
| • | comprised of elements from RXi’s IP portfolio, and | |
| • | potentially more specific for the target gene. |
51
Table of Contents
| • | inhibition of liver target with 1 mg RNAi compound per kg of body weight; | |
| • | no immune stimulation detected; | |
| • | defined particle size; and | |
| • | readily formulated. |
52
Table of Contents
53
Table of Contents
| • | We intend to finance the initial development of a limited number of RNAi drug candidates with our own capital resources and any financial resources that we may obtain from capital markets and partners. Our key therapeutic areas of interest are neurology, metabolic disease and oncology. We intend to |
54
Table of Contents
| develop drugs in these areas internally to establish significant value, at which point we may seek to partner them. |
| • | We intend to seek partnerships with large pharmaceutical and biotechnology companies to leverage our intellectual property and expand our development pipeline. Such partnerships may include traditionally structured drug development and commercialization licenses, discovery and development collaborations, research and technology collaborations, and intellectual property licenses. | |
| • | We intend to maintain and continue to develop and enhance our RNAi technology platform by expanding our intellectual property position in RNAi compound chemistry, delivery and target sequences. To date, we have in-licensed RNAi technologies from various institutions and companies, including UMMS, Imperial College of London, Cold Spring Harbor Laboratories and TriLink Biotechnologies. We intend to continue to enhance our technology platform through in-licensing in combination with internal and collaborative research and development programs. | |
| • | We intend to develop future RNAi technology improvements and believe we are well positioned to do so. Our management and advisors have developed much of the core technology in the field of oligonucleotide therapeutics, and more specifically RNAi. For example, our Scientific Advisory Board member, Dr. Nassim Usman, developed the standard synthesis chemistry used to manufacture RNAi compounds throughout the world while a fellow at MIT. Our CEO, Dr. Tod Woolf, co-developed STEALTH RNAi, which is one of the most commonly employed second generation RNAi chemistries, and our Vice President of Pharmaceutical Development, Dr. Pamela Pavco, developed the first modified RNAi tested in humans while she was at Sirna Therapeutics. Our advisors and scientists routinely meet to discuss novel approaches and improvements in our RNAi technology platforms to enhance our intellectual property portfolio. | |
| • | We may also seek to collaborate with government and charitable institutions through grants and funded research for our development programs. |
| • | therapeutic targets, | |
| • | chemistry and configurations of RNAi compounds, and | |
| • | delivery technology for RNAi. |
55
Table of Contents
| • | Dr. Rana’s inventions which provide fundamental rules for designing chemically modified RNAi sequences suitable forin vivo gene silencing, to which we have co-exclusive licenses in the therapeutic areas of type 2 diabetes, obesity, and ALS by targeting mutant SOD1, and CMV retinitis; | |
| • | Dr. Tuschl’s invention regarding RNAi therapeutics using double-stranded RNAs in the areas of type 2 diabetes, obesity, and ALS by targeting mutant SOD1, and CMV retinitis; | |
| • | Drs. Mello and Zamore’s invention regardingin vivoproduction of siRNA; and | |
| • | methods of triggering RNAiin vivo, based on particular RNA structural characteristics. |
| • | methods and compositions, including use of nanotransporters, for RNAi compound delivery which enable therapeutic gene silencing in cells and animals; and | |
| • | inhibition of gene expression in fat cells using RNAi. |
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Drug discovery for diabetes and obesity | Michael P. Czech Silvia Corvera | 09/27/1993 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | US Pat. 5,989,893 Pending elsewhere | ||||
| RNAi in general | Craig C. Mello Andrew Fire Stephen Kostas Mary Montgomery Lisa Timmons SiQun Zu Hiroaki Tabara Samuel E. Driver | 12/23/1997 | Non-exclusive license to fundamental RNAi from Carnegie Institute of Washington. | US Pat. 6,506,559 Pending elsewhere |
56
Table of Contents
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Drug discovery using genomic databases for diabetes and obesity | Michael P. Czech Andrew D. Cherniack Adelson L. Guilherme | 10/20/2000 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending | ||||
| dsRNA in general | Thomas Tuschl, Philip A. Sharp Phillip D. Zamore David P. Bartel | 12/01/2000 | Non-exclusive license for RNAi use to inhibit (1) HCMV immediate early gene in retinitis (2) mutant SOD1 gene in ALS, and (3) gene targets in type II diabetes & obesity from UMMS, a co-owner of the patent. | Pending | ||||
| Engineered precursor to siRNA for in vivo production of siRNA | Phillip D. Zamore Gyorgy Hutvagner Juanita McLachlan Craig C. Mello Alla Grishok | 07/12/2001 | Exclusive license for RNAi use to inhibit (1) HCMV immediate early gene in retinitis (2) mutant SOD1 gene in ALS, and (3) gene targets in type II diabetes & obesity. | Pending | ||||
| In vivo gene silencing by chemically modified and stable siRNA | Tariq M. Rana Ya-Lin Chiu | 09/25/2002 | Exclusive worldwide license to the treatment of CMV, ALS, diabetes & obesity for therapeutics, prophylactics or diagnostics. | Pending | ||||
| Allele specific inhibition by siRNA, especially SOD inhibition for the treatment of ALS | Zuoshang Xu Phillip D. Zamore | 11/04/2002 | Exclusive license on inhibition of SOD1 expression for the treatment of ALS including therapeutics, prophylactics and diagnostics. | Pending | ||||
| Delivery of siRNA using peptide conjugate | Tariq M. Rana | 11/26/2002 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans, including for ALS, diabetes and obesity. | Pending | ||||
| Allele specific inhibition by chemically modified siRNA | Tariq M. Rana | 11/26/2002 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Delivery of siRNA into adipocytes for treatment of diabetes and obesity ; | Michael P. Czech Qiong L. Zhou Zhen Y. Jiang | 12/11/2002 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending |
57
Table of Contents
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Screening for modulators of fat storage | Malcolm Parker Roger White Goran Leonardsson | 02/03/2003 | Exclusive license for screening methods for identifying compounds that are useful as modulators of fat storage from Imperial College, UK. | Pending | ||||
| Inhibition of CMV using siRNA | Timothy F. Kowalik | 02/05/2003 | Exclusive license to develop RNAi therapeutics for cytomegalovirus related diseases (HCMV- immediate early gene). (Note that original license only for CMV retinitis; amended 01/04 to include all CMV) | Pending | ||||
| Modulation of insulin sensitivity through stress proteins | Silvia Corvera | 06/19/2003 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending | ||||
| Chemically modified siRNA and their uses | Tariq M. Rana Ya-Lin Chiu | 08/05/2003 | Co-Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications of RNAi for inhibition of human CMV immediate early gene expression, mutant SOD1 (ALS treatment), genes implicated in diabetes and obesity. | Pending | ||||
| Increase of insulin stimulated glucose regulation by a small molecule inhibitor of RIP140 | Michael P. Czech Aimee Powelka Adilson L. Guilherme Andrew D. Cherniack | 03/05/2004 | Exclusive license for drug discovery in type 2 diabetes and/or obesity with therapeutic, prophylactic, or diagnostic applications and products for treatment, prevention or diagnosis of type 2 diabetes and/or obesity. | Pending | ||||
| Efficient delivery of siRNA into cells and animals | Tariq M. Rana | 08/11/2005 | Nonexclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Delivery of chemically modified siRNA using nanotransporters | Tariq M. Rana (Zuoshang Xu) | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending |
58
Table of Contents
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Delivery of chemically modified siRNA | Tariq M. Rana | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Gene silencing of cholesterol biosynthesis and other metabolic genes by chemically modified siRNA | Tariq M. Rana | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Microwave assisted nucleic acid delivery system | Tariq M. Rana | Not yet filed | Nonexclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Invention disclosure | ||||
| Methods and compositions for RNA interference in suspended cells or in whole body | Scott Hammond Gregory Hannon David Beach Amy Caudy Emily Bernstein | 03/16/2001 | Non-exclusive license from Cold Spring Harbor Laboratory for use of short hairpin RNA for drug discovery and development. | Pending |
| * | Unless otherwise noted, the licenses are from UMMS. |
59
Table of Contents
60
Table of Contents
61
Table of Contents
62
Table of Contents
63
Table of Contents
64
Table of Contents
Name | Position | Age | ||||
| Sanford J. Hillsberg(1)(2) | Director and Chairman of the Board | 59 | ||||
| Tod Woolf, Ph.D. | President and Chief Executive Officer, Director | 43 | ||||
| Mark J. Ahn, Ph.D.(1)(2)(3) | Director | 44 | ||||
| Stephen S. Galliker(1)(2)(3) | Director | 60 | ||||
| Steven A. Kriegsman(3) | Director | 65 | ||||
| Stephen J. DiPalma | Chief Financial Officer and Secretary | 48 | ||||
| Pamela Pavco, Ph.D. | Vice President of Pharmaceutical Development | 51 | ||||
| Dmitry Samarsky, Ph.D. | Vice President of Technology and Business Development | 40 | ||||
| James Warren | Former Chief Financial Officer and Secretary | 62 | ||||
| (1) | Member of our Nominating and Governance Committee | |
| (2) | Member of our Audit Committee | |
| (3) | Member of our Compensation Committee |
Name | Professional Affiliation | |
| Craig C. Mello, Ph.D (Chairman) | • Professor and Blaise University Chair in Molecular Medicine at the University of Massachusetts Medical School | |
| • Investigator, Howard Hughes Medical Institute | ||
| • Member of the National Academy of Sciences | ||
| Tariq Rana, Ph.D | • Professor of Biochemistry & Molecular Pharmacology and Founding Director of the Program in Chemical Biology at the University of Massachusetts Medical School | |
| Gregory Hannon, Ph.D | • Investigator, Howard Hughes Medical Institute | |
| Michael Czech, Ph.D | • Professor and Chair in Molecular Medicine at the University of Massachusetts Medical School | |
| Nicholas Dean, Ph.D | • Founder and Chief Scientific Officer of Excaliard Pharmaceuticals | |
| Nassim Usman, Ph.D | • Chief Executive Officer and Board Member of Catalyst Biosciences |
65
Table of Contents
66
Table of Contents
67
Table of Contents
68
Table of Contents
69
Table of Contents
70
Table of Contents
| • | an annual retainer fee of $10,000, payable quarterly, | |
| • | an annual retainer fee of $12,000 for the chairperson of each committee of our board of directors other than the audit committee, payable quarterly, | |
| • | an annual retainer fee of $20,000 for the chairperson of the audit committee of our board of directors, payable quarterly, | |
| • | an annual retainer fee of $32,000 for the Chairman of the board of directors, payable quarterly, | |
| • | a fee of $2,000 per board meeting attended by the director ($1,000 if attendance is telephonic), and | |
| • | a fee of $1,500 per committee meeting attended by the director ($750 if attendance is telephonic). |
71
Table of Contents
72
Table of Contents
| • | base salary, | |
| • | performance-based cash compensation, and | |
| • | long-term equity incentive compensation. |
| • | the negotiated terms of each executive employment agreement, | |
| • | internal review of the executive’s compensation, both individually and relative to other executive officers, and | |
| • | individual performance of the executive. |
73
Table of Contents
| • | establish the link between the creation of stockholder value and long-term executive incentive compensation, | |
| • | provide an opportunity for increased equity ownership by executives, | |
| • | function as a retention tool because of the vesting features included in all options granted by the Committee, and | |
| • | maintain competitive levels of total compensation. |
74
Table of Contents
75
Table of Contents
| Value of | ||||||||||||||||||||
| Value of | Accelerated | |||||||||||||||||||
| Accelerated | Option | |||||||||||||||||||
| Salary | Benefit | Option | Vesting upon | |||||||||||||||||
| Continuation | Continuation | Vesting | Total | a Change in | ||||||||||||||||
Name and Principal Position | (Termination) | (Termination) | (Termination) | (Termination) | Control | |||||||||||||||
| Tod Woolf | $ | 250,000 | $ | 14,500 | $ | 449,000 | $ | 714,400 | $ | 1,087,100 | ||||||||||
| President and Chief Executive Officer | ||||||||||||||||||||
| Stephen J. DiPalma | $ | 110,000 | $ | 7,300 | — | $ | 117,300 | $ | 406,400 | |||||||||||
| Chief Financial Officer | ||||||||||||||||||||
| Pamela Pavco(1) | $ | 198,000 | $ | 2,100 | $ | 157,200 | $ | 357,300 | $ | 511,000 | ||||||||||
| Vice President of Research and Development | ||||||||||||||||||||
| Dmitry Samarsky(1) | $ | 85,000 | $ | 1,800 | $ | 57,200 | $ | 144,000 | $ | 400,400 | ||||||||||
| Vice President of Technology and Business Development | ||||||||||||||||||||
| (1) | Each of Drs. Pavco and Samarsky are entitled to specified benefits in the event his or her employment is terminated without cause or he or she terminates his or her employment for good reason following a change in control, as further described below. |
76
Table of Contents
77
Table of Contents
78
Table of Contents
79
Table of Contents
80
Table of Contents
81
Table of Contents
82
Table of Contents
83
Table of Contents
84
Table of Contents
85
Table of Contents
86
Table of Contents
| • | any person known by us to be the beneficial owner of 5% or more of our common stock, including any “group” as that term is defined in the Exchange Act, | |
| • | each director, our named executive officers identified in the “Management-Executive Compensation” section above, and | |
| • | all of our directors and executive officers as a group. |
| After the Distribution | ||||||||||||||||
| and Award Transactions | ||||||||||||||||
| Before the Distribution | Described in this Prospectus | |||||||||||||||
| Number | Number | |||||||||||||||
| of | of | |||||||||||||||
Name | Shares | Percent | Shares | Percent | ||||||||||||
5% Stockholders: | ||||||||||||||||
| CytRx Corporation(1) | 10,823,199 | 85.8 | % | 6,212,861 | 49.3 | % | ||||||||||
Executive Officers and Directors: | ||||||||||||||||
| Tod Woolf, Ph.D.(2) | 52,832 | # | 52,832 | # | ||||||||||||
| Stephen J. DiPalma(3) | 6,250 | # | 6,250 | # | ||||||||||||
| James Warren(4) | 62,743 | # | 62,743 | # | ||||||||||||
| Pamela Pavco, Ph.D.(5) | 18,163 | # | 18,163 | # | ||||||||||||
| Dmitry Samarsky, Ph.D.(6) | 6,597 | # | 6,597 | # | ||||||||||||
| Mark J. Ahn, Ph.D.(7) | 47,500 | # | 47,500 | # | ||||||||||||
| Stephen S. Galliker(7) | 47,500 | # | 47,500 | # | ||||||||||||
| Sanford J. Hillsberg(7)(8) | 47,500 | # | 47,500 | # | ||||||||||||
| Steven A. Kriegsman(9) | 37,500 | # | 278,268 | 2.2 | % | |||||||||||
| All executive officers and directors as a group (8 persons)(10) | 296,585 | 2.50 | % | 567,353 | 4.4 | % | ||||||||||
| # | Represents less than 1% of the outstanding shares of our common stock. |
87
Table of Contents
| (1) | The address for CytRx is 11726 San Vicente Boulevard, Suite 650, Los Angeles, California 90049. | |
| (2) | Consists of 52,832 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (3) | Consists of 6,250 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (4) | Consists of 62,743 shares of common stock underlying stock options currently exercisable until November 30, 2007. | |
| (5) | Consists of 18,163 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (6) | Includes 6,597 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007 | |
| (7) | Includes 37,500 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (8) | The shares shown do not include shares owned by Troy & Gould Professional Corporation. | |
| (9) | Includes 37,500 shares of common stock underlying stock options and exercisable within 60 days of October 1, 2007 and, following the distribution and award transactions described in this prospectus, approximately 240,768 shares that Mr. Kriegsman will receive as a result of distribution under this prospectus and the award under the award prospectus. Please see “The Distribution” for a further explanation of the allocation of shares in the distribution. Mr. Kriegsman is the CEO and a director of CytRx, but acting alone, he has neither voting nor investment power with respect to the shares beneficially owned by CytRx. As a result, Mr. Kriegsman disclaims beneficial ownership of such shares. |
| (10) | Includes 293,282 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. |
88
Table of Contents
89
Table of Contents
90
Table of Contents
| • | prior to such date, the board of directors of the corporation approved either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder, | |
| • | upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, other than statutorily excluded shares, or | |
| • | on or subsequent to such date, the business combination is approved by the board of directors of the corporation and authorized at an annual or special meeting of the stockholders by the affirmative vote of at least 662/3% of the outstanding voting stock which is not owned by the interested stockholder. |
| • | any person that is the owner of 15% or more of the outstanding voting stock of the corporation, or is an affiliate or associate of the corporation and was the owner of 15% or more of the outstanding voting stock of the corporation at any time within three years immediately prior to the date of determination, and | |
| • | the affiliates and associates of any such person. |
91
Table of Contents
92
Table of Contents
93
Table of Contents
| • | an individual citizen or resident of the United States, | |
| • | a corporation (including any entity treated as a corporation for U.S. federal income tax purposes), partnership or other entity created or organized in or under the laws of the United States, any state or any political subdivision thereof, | |
| • | an estate, the income of which is subject to United States federal income taxation regardless of the source of the income, or | |
| • | a trust whose administration is subject to the primary supervision of a United States court and which has one or more United States persons who have the authority to control all of its substantial decisions or which has elected to be treated as a United States person. |
94
Table of Contents
95
Table of Contents
96
Table of Contents
Investor Relations
One Innovation Drive
Worcester, Massachusetts 01605
Telephone:(508) 767-3861
97
Table of Contents
| Page | ||||
| F-3 | ||||
| F-4 | ||||
| F-5 | ||||
| F-6 | ||||
| F-7 | ||||
F-1
Table of Contents
F-2
Table of Contents
| As of | As of | As of | ||||||||||||||||||
| June 30, | December 31, | December 31, | ||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | |||||||||||||||||
| (Successor) | (Predecessor) | |||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (Amounts in thousands, except share data) | ||||||||||||||||||||
ASSETS | ||||||||||||||||||||
| Current assets: | ||||||||||||||||||||
| Cash and cash equivalents | $ | 14,965 | $ | 2 | $ | — | $ | — | ||||||||||||
| Prepaid expenses | 15 | — | 7 | — | ||||||||||||||||
| Total current assets | 14,980 | 2 | 7 | — | ||||||||||||||||
| Equipment and furnishings, net | 59 | — | — | — | ||||||||||||||||
| Deposits | 50 | — | 50 | 50 | ||||||||||||||||
| Total assets | $ | 15,089 | $ | 2 | $ | 57 | $ | 50 | ||||||||||||
| LIABILITIES, STOCKHOLDERS’ EQUITY AND PARENT COMPANY’S NET DEFICIT | ||||||||||||||||||||
| Current liabilities: | ||||||||||||||||||||
| Accounts payable | $ | 438 | $ | — | $ | 134 | $ | 367 | ||||||||||||
| Accrued expenses and other current liabilities | 374 | — | 191 | 133 | ||||||||||||||||
| Due to Parent | 1,678 | — | — | — | ||||||||||||||||
| Total current liabilities | 2,490 | — | 325 | 500 | ||||||||||||||||
| Commitment and contingencies | — | — | — | — | ||||||||||||||||
| Stockholders’ Equity: | ||||||||||||||||||||
| Common stock, $0.0001 par value, 50,000,000 shares authorized, 12,400,000 and 1,624,278 shares issued and outstanding at June 30, 2007 and December 31, 2006, respectively | 1 | — | — | — | ||||||||||||||||
| Additional paid-in capital | 18,769 | 2 | — | — | ||||||||||||||||
| Deficit accumulated during the development stage | (6,171 | ) | — | — | — | |||||||||||||||
| Total stockholders’ equity | 12,599 | 2 | — | — | ||||||||||||||||
| Parent company’s net deficit | — | — | (268 | ) | (450 | ) | ||||||||||||||
| Total liabilities, stockholders’ equity and parent company’s net deficit | $ | 15,089 | $ | 2 | $ | 57 | $ | 50 | ||||||||||||
F-3
Table of Contents
| Period | ||||||||||||||||||||||||||||
| Period | from | |||||||||||||||||||||||||||
| from | January 1, | |||||||||||||||||||||||||||
| April 3, | 2003 | |||||||||||||||||||||||||||
| 2006 (Date of | (Date of | |||||||||||||||||||||||||||
| Incorporation) | Inception) to | |||||||||||||||||||||||||||
| to June 30, | Six Months Ended June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||
| 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||||||||
| (Successor) | (Successor) | (Predecessor) | (Predecessor) | |||||||||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||||||||||
| (Amounts in thousands, except shares and earnings per share data) | ||||||||||||||||||||||||||||
| Revenue | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Expenses: | ||||||||||||||||||||||||||||
| Research and development expense | $ | 1,271 | $ | 1,271 | $ | 802 | $ | 5,531 | $ | 1,298 | $ | 1,929 | $ | 1,700 | ||||||||||||||
| Research and development non-employee stock-based compensation expense | 1,012 | 1,012 | 105 | 1,367 | 212 | 151 | 1,006 | |||||||||||||||||||||
| Fair value of common stock issued in exchange for licensing rights | 2,311 | 2,311 | — | 1,643 | 262 | — | 108 | |||||||||||||||||||||
| Total research and development expense | 4,594 | 4,594 | 907 | 8,541 | 1,772 | 2,080 | 2,814 | |||||||||||||||||||||
| General and administrative expense | 1,381 | 1,381 | 155 | 1,500 | 633 | 129 | 458 | |||||||||||||||||||||
| General and administrative employee stock-based compensation | 329 | 329 | — | — | — | — | — | |||||||||||||||||||||
| Total general and administrative expense | 1,710 | 1,710 | 155 | 1,500 | 633 | 129 | 458 | |||||||||||||||||||||
| Operating loss | (6,304 | ) | (6,304 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Interest income | 133 | 133 | — | — | — | — | — | |||||||||||||||||||||
| Loss before income taxes | (6,171 | ) | (6,171 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Income taxes | — | — | — | — | — | — | — | |||||||||||||||||||||
| Net loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | $ | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | |||||||
| Basic and diluted loss per share | $ | (0.64 | ) | $ | (0.64 | ) | NA | NA | NA | NA | NA | |||||||||||||||||
| Weighted average shares, basic and diluted | 9,671,849 | 9,671,849 | NA | NA | NA | NA | NA | |||||||||||||||||||||
F-4
Table of Contents
| Deficit | ||||||||||||||||||||||||
| Additional | Accumulated | Parent | ||||||||||||||||||||||
| Common Stock | Paid-In | During | Company’s | |||||||||||||||||||||
| Shares Issued | Amount | Capital | Development Stage | Net Deficit | Total | |||||||||||||||||||
| (Amounts in thousands, except share data) | ||||||||||||||||||||||||
Predecessor | ||||||||||||||||||||||||
| Balance at December 31, 2003 | — | $ | — | $ | — | $ | (89 | ) | $ | (89 | ) | |||||||||||||
| Net loss | — | — | — | (3,272 | ) | (3,272 | ) | |||||||||||||||||
| Net transactions with Parent | — | — | — | 2,393 | 2,393 | |||||||||||||||||||
| Balance at December 31, 2004 | — | — | — | (968 | ) | (968 | ) | |||||||||||||||||
| Net loss | — | — | — | (2,209 | ) | (2,209 | ) | |||||||||||||||||
| Net transactions with Parent | — | — | — | 2,727 | 2,727 | |||||||||||||||||||
| Balance at December 31, 2005 | — | — | — | (450 | ) | (450 | ) | |||||||||||||||||
| Net loss | — | — | — | (2,405 | ) | (2,405 | ) | |||||||||||||||||
| Net transactions with Parent | — | — | — | 2,587 | 2,587 | |||||||||||||||||||
| Balance at December 31, 2006 | $ | — | $ | — | $ | — | $ | (268 | ) | $ | (268 | ) | ||||||||||||
Successor | ||||||||||||||||||||||||
| Balance at April 3, 2006 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Issuance of common stock | 1,624,278 | — | 2 | — | 2 | |||||||||||||||||||
| Balance at December 31, 2006 | 1,624,278 | — | 2 | — | 2 | |||||||||||||||||||
| Common stock issued to CytRx for contribution of RXi and other assets | 7,040,318 | 1 | 47 | — | 48 | |||||||||||||||||||
| Common stock issued to CytRx for cash | 3,273,292 | — | 15,348 | — | 15,348 | |||||||||||||||||||
| Common stock issued to UMMS for additional intellectual properties | 462,112 | — | 2,311 | — | 2,311 | |||||||||||||||||||
| Stock based compensation expense for directors and employees | — | — | 329 | — | 329 | |||||||||||||||||||
| Stock based compensation expense for services | — | — | 732 | — | 732 | |||||||||||||||||||
| Net loss | — | — | — | (6,171 | ) | (6,171 | ) | |||||||||||||||||
| Balance at June 30, 2007 (Unaudited) | 12,400,000 | $ | 1 | $ | 18,769 | $ | (6,171 | ) | $ | 12,599 | ||||||||||||||
F-5
Table of Contents
STATEMENTS OF CASH FLOWS
(A Development Stage Company)
| Period | ||||||||||||||||||||||||||||
| from | ||||||||||||||||||||||||||||
| Period | January 1, | |||||||||||||||||||||||||||
| from | 2003 | |||||||||||||||||||||||||||
| April 3, | (Date of | |||||||||||||||||||||||||||
| 2006 (Date of | Inception) | |||||||||||||||||||||||||||
| Incorporation) | through | |||||||||||||||||||||||||||
| through | Six Months Ended June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||
| June 30, 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||||||||
| (Successor) | (Successor) | (Predecessor) | (Predecessor) | (Predecessor) | ||||||||||||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||||||||||||||
| (Amounts in thousands) | ||||||||||||||||||||||||||||
| Cash flows from operating activities: | ||||||||||||||||||||||||||||
| Net Loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | $ | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | |||||||
| Adjustment to reconcile net loss to net cash used in operating activities: | ||||||||||||||||||||||||||||
| Depreciation expense | 12 | 12 | — | — | — | — | — | |||||||||||||||||||||
| Stock option expense | 1,341 | 1,341 | 105 | 1,369 | 212 | 151 | 1,006 | |||||||||||||||||||||
| Fair value of common stock issued in exchange for licensing rights | 2,311 | 2,311 | — | 1,643 | 262 | — | 108 | |||||||||||||||||||||
| Changes in assets and liabilities: | ||||||||||||||||||||||||||||
| Prepaid expenses | (15 | ) | (15 | ) | — | (7 | ) | (7 | ) | — | — | |||||||||||||||||
| Accounts payable | 435 | 435 | (245 | ) | 134 | (232 | ) | (421 | ) | 764 | ||||||||||||||||||
| Accrued expenses and other current liabilities | 374 | 374 | (52 | ) | 191 | 58 | (47 | ) | 114 | |||||||||||||||||||
| Total adjustments | 4,458 | 4,458 | (192 | ) | 3,330 | 293 | (317 | ) | 1,992 | |||||||||||||||||||
| Net cash used in operating activities | (1,713 | ) | (1,713 | ) | (1,254 | ) | (6,711 | ) | (2,112 | ) | (2,526 | ) | (1,280 | ) | ||||||||||||||
| Cash flows from investing activities: | ||||||||||||||||||||||||||||
| Cash paid for purchase of equipment and furnishings | (19 | ) | (19 | ) | — | — | — | — | — | |||||||||||||||||||
| Cash paid for lease deposit | — | — | — | (50 | ) | — | (50 | ) | — | |||||||||||||||||||
| Net cash used in investing activities | (19 | ) | (19 | ) | — | (50 | ) | — | (50 | ) | — | |||||||||||||||||
| Cash flows from financing activities: | ||||||||||||||||||||||||||||
| Net proceeds from issuance of common stock | 15,348 | 15,348 | — | — | — | — | — | |||||||||||||||||||||
| Cash advances from Parent, net | 1,347 | 1,347 | 1,254 | 6,761 | 2,112 | 2,576 | 1,280 | |||||||||||||||||||||
| Net cash provided by financing activities | 16,695 | 16,695 | 1,254 | 6,761 | 2,112 | 2,576 | 1,280 | |||||||||||||||||||||
| Net increase in cash and cash equivalents | 14,963 | 14,963 | — | — | — | — | — | |||||||||||||||||||||
| Cash and cash equivalents at beginning of period | 2 | 2 | — | — | — | — | — | |||||||||||||||||||||
| Cash and cash equivalents at end of period | $ | 14,965 | $ | 14,965 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Supplemental disclosure of cash flow information: | ||||||||||||||||||||||||||||
| Cash received during the period for interest | $ | 133 | $ | 133 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Supplemental disclosure of non-cash investing and financing activities: | ||||||||||||||||||||||||||||
| Equipment and furnishings exchanged for common stock | $ | 49 | $ | 49 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Equipment and furnishings purchased and included in current liabilities | $ | 3 | $ | 3 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
F-6
Table of Contents
| 1. | Nature of Business |
F-7
Table of Contents
| 2. | Summary of Significant Accounting Policies |
F-8
Table of Contents
F-9
Table of Contents
| Years Ended December 31, | ||||||||
| 2005 | 2004 | |||||||
| Net loss, as reported | $ | (2,209 | ) | $ | (3,272 | ) | ||
| Total stock-based employee compensation expense determined under fair-value based method for all awards | (54 | ) | (110 | ) | ||||
| Pro forma net loss | $ | (2,263 | ) | $ | (3,382 | ) | ||
| 2005 | 2004 | |||||||
| Weighted average risk free interest rate | 4.10 | % | 3.65 | % | ||||
| Volatility factors of the expected market price of CytRx’s common stock | 109 | % | 117 | % | ||||
| Expected lives (years) | 8 | 8 | ||||||
| Weighted average years outstanding | 4.8 | 5.8 | ||||||
| Dividend yields | 0 | % | 0 | % | ||||
F-10
Table of Contents
F-11
Table of Contents
F-12
Table of Contents
| For the Six Months Ended June 30, | For The Years Ended December 31, | |||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | ||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| Executive | $ | 217 | $ | 61 | $ | 115 | $ | 60 | $ | 98 | ||||||||||
| Accounting | 31 | 13 | 24 | 14 | 15 | |||||||||||||||
| Legal | 56 | 24 | 39 | 15 | 15 | |||||||||||||||
| Total | 304 | 98 | 178 | 89 | 128 | |||||||||||||||
| 3. | Recent Accounting Pronouncements |
F-13
Table of Contents
| 4. | Development Stage Supplemental Equity Disclosure |
| Price per | ||||||||||||||||||||||||||||
| Share or | ||||||||||||||||||||||||||||
| Shares of | Dollar | Exercise | Counter | Nature of | Basis for | |||||||||||||||||||||||
| Date of | Common | Amount of | Price per | Party to | Non-Cash | Assigning | ||||||||||||||||||||||
Type of Security | Issuance | Stock | Consideration | Share | Transaction | Consideration | Cost | |||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||||||
| Common Stock | April 3, 2006 | 1,624,278 | $ | 2 | $ | 0.002 | Founders | NA | Cash | |||||||||||||||||||
| Common Stock | January 8, 2007 | 7,040,318 | 48 | $ | 0.007 | (A) | CytRx | Contributed Assets | Predecessor Cost | |||||||||||||||||||
| Common Stock | April 30, 2007 | 3,273,292 | 15,348 | $ | 4.69 | (B) | CytRx | NA | Cash | |||||||||||||||||||
| Common Stock | April 30, 2007 | 462,112 | 2,311 | $ | 5.00 | UMMS | Intellectual properties | Independent third-party valuation | ||||||||||||||||||||
| Sub-total of Common Stock issued | 12,400,000 | 17,709 | ||||||||||||||||||||||||||
| Common Stock Options | May 23, 2007 | $ | 5.00 | Employees | Professional & Employee | Independent third-party | ||||||||||||||||||||||
| 313,000 | (C) | 1,061 | services | valuation | ||||||||||||||||||||||||
| $ | 18,770 | |||||||||||||||||||||||||||
| (A) | Transactions between related parties are accounted for at the historical cost of CytRx. with the intellectual property which was previously expensed on CytRx’s books being recorded at zero cost and equipment and furnishings being recorded at $48,000. | |
| (B) | RXi received gross proceeds of $17.0 million for the issuance of the 3,273,292 shares of common stock which equals $5.19 per share. The gross proceeds were reduced by a reimbursement to CytRx of (1) $1.3 million for RXi’s pro rata share of offering costs and (2) $363,000 of expenses incurred on behalf of RXi for the year ended December 31, 2006. Net proceeds to RXi after these charges was $15.3 million. | |
| (C) | Common stock underlying options granted is 1,177,000 and the vested portion of the common stock option grants at June 30, 2007 was 313,000. |
F-14
Table of Contents
| 5. | Deposits |
| 6. | Equipment and Furnishings, net |
| As of | As of | |||||||||||||||
| June 30, | December 31, | As of December 31, | ||||||||||||||
| 2007 | 2006 | 2006 | 2005 | |||||||||||||
| (Successor) | ||||||||||||||||
| (unaudited) | (Predecessor) | |||||||||||||||
| (In thousands) | ||||||||||||||||
| Equipment and furnishings | $ | 116 | $ | — | $ | — | $ | — | ||||||||
| Less — accumulated depreciation | (57 | ) | — | — | — | |||||||||||
| Equipment and furnishings, net | $ | 59 | $ | — | $ | — | $ | — | ||||||||
| 7. | Accrued Expenses and Other Current Liabilities |
| As of | As of | |||||||||||||||
| June 30, | December 31, | As of December 31, | ||||||||||||||
| 2007 | 2006 | 2006 | 2005 | |||||||||||||
| (Successor) | ||||||||||||||||
| (unaudited) | (Predecessor) | |||||||||||||||
| (In thousands) | ||||||||||||||||
| Professional fees | $ | 166 | $ | — | $ | 176 | $ | 71 | ||||||||
| Research and development costs | 196 | — | 10 | 62 | ||||||||||||
| Other | 12 | — | 5 | — | ||||||||||||
| Total accrued expenses and other current liabilities | $ | 374 | $ | — | $ | 191 | $ | 133 | ||||||||
| 8. | Commitments and Contingencies |
F-15
Table of Contents
| Non-Cancelable | Cancelable | |||||||||||||||||||
| Operating | Employment | License | ||||||||||||||||||
Years Ending December 31, | Leases(1) | Agreements(2) | Subtotal | Agreements(3) | Total | |||||||||||||||
| 2007 | $ | 250 | $ | 951 | $ | 1,201 | $ | 2,892 | $ | 4,093 | ||||||||||
| 2008 | 9 | 810 | 819 | 433 | 1,252 | |||||||||||||||
| 2009 | — | 710 | 710 | 383 | 1,093 | |||||||||||||||
| 2010 | — | 460 | 460 | 333 | 793 | |||||||||||||||
| 2011 | — | 255 | 255 | 333 | 588 | |||||||||||||||
| Thereafter | — | 170 | 170 | 7,568 | 7,738 | |||||||||||||||
| Total | $ | 259 | $ | 3,356 | $ | 3,615 | $ | 11,942 | $ | 15,557 | ||||||||||
| (1) | Operating leases are primarily facility and equipment related obligations with third party vendors. Facility lease expenses during the six months ended June 30, 2007 and 2006, were approximately $68,000 and $1,000, respectively. Facility lease expenses during the twelve months ended December 31, 2006, 2005 and 2004 were $2,000, $2,000 and $3,000, respectively. | |
| (2) | Employment agreement obligations include management contracts, as well as scientific advisory board member compensation agreements. Certain agreements, which have been revised from time to time, provide for minimum salary levels, adjusted annually at the discretion of the Compensation Committee, as well as for minimum bonuses that are payable. | |
| (3) | License agreements generally relate to our obligations with UMMS associated with RNAi and, for future periods, represent minimum annual royalty payment obligations. Included in the 2007 license obligations was a $2.3 million payment made in RXi common stock to UMMS during the second quarter of 2007. |
| 9. | Stock Based Compensation |
F-16
Table of Contents
| Six Months Ended | ||
| June 30, 2007 | ||
| Risk-free interest rate | 4.51% — 4.55% | |
| Expected volatility | 108.7% | |
| Expected lives (years) | 6 | |
| Expected dividend yield | 0.00% |
F-17
Table of Contents
| Stock Options Six Months Ended June 30, 2007 | ||||||||||||||||
| Number of Shares | Number of Shares | Total Number | Weighted Average | |||||||||||||
| (Employees) | (Non-Employees) | of Shares | Exercise Price | |||||||||||||
| Outstanding at January 1, 2007 | — | — | — | — | ||||||||||||
| Granted | 829,000 | 348,000 | 1,177,000 | $ | 5.00 | |||||||||||
| Exercised | — | — | — | — | ||||||||||||
| Forfeited | — | — | — | — | ||||||||||||
| Outstanding at June 30, 2007 | 829,000 | 348,000 | 1,177,000 | $ | 5.00 | |||||||||||
| Options exercisable at June 30, 2007 | 98,000 | 215,000 | 313,000 | $ | 5.00 | |||||||||||
| Weighted Average | ||||||||||||||||
| Number of Shares | Number of Shares | Total Number of | Grant Date Fair | |||||||||||||
| (Employees) | (Non-Employees) | Shares | Value per Share | |||||||||||||
| Unvested at January 1, 2007 | — | — | — | — | ||||||||||||
| Granted | 829,000 | 348,000 | 1,177,000 | $ | 3.52 | |||||||||||
| Forfeited | — | — | — | — | ||||||||||||
| Vested | (97,000 | ) | (216,000 | ) | (313,000 | ) | $ | 3.37 | ||||||||
| Unvested at June 30, 2007 | 732,000 | 132,000 | 864,000 | $ | 3.57 | |||||||||||
| Weighted Average | ||||||||||||||||||||||||
| Remaining | Number of | |||||||||||||||||||||||
| Range of | Number of | Contractual Life | Weighted Average | Options | Weighted Average | Weighted Average | ||||||||||||||||||
Exercise Prices | Options | (Years) | Exercise Price | Exercisable | Contractual Life | Exercise Price | ||||||||||||||||||
| $5.00 | 1,177,000 | 9.87 | $ | 5.00 | 313,000 | 9.88 | $ | 5.00 | ||||||||||||||||
| 10. | Income Taxes |
F-18
Table of Contents
| 11. | License Agreements |
| 12. | Related Party Transactions |
F-19
Table of Contents
F-20
Table of Contents
| 13. | Subsequent Events |
F-21
Table of Contents
F-22
Table of Contents
Table of Contents
| We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities, and we are not soliciting an offer to buy these securities in any state where the offer or sale is not permitted. |

| Page | ||
| A-1 | ||
| A-10 | ||
| A-25 | ||
| A-26 | ||
| A-29 | ||
| A-29 | ||
| A-30 | ||
| A-32 | ||
| A-45 | ||
| A-62 | ||
| A-69 | ||
| A-80 | ||
| A-82 | ||
| A-84 | ||
| A-86 | ||
| A-89 | ||
| A-90 | ||
| A-91 | ||
| A-91 | ||
| A-92 | ||
| F-1 |
A-i
Table of Contents
| • | Neurology. Initially, we are pursuing research in ALS (amyotrophic lateral sclerosis, commonly known as Lou Gehrig’s Disease). One form of ALS is caused by defects in a gene called SOD1. Early preclinical studies conducted by RXi advisors, Dr. Tariq Rana and Dr. Zuoshang Xu at UMMS, showed promising results in animals using an RNAi compound to selectively inhibit the SOD1 gene. We are refining and extending this work and, if successful, will move into formal preclinical development. We also intend to leverage our experience related to the delivery of RNAi therapeutics in the central nervous system to explore development of RNAi-based treatments for neurodegenerative diseases other than ALS, including Alzheimer’s Disease. | |
| • | Metabolic disease. One of our scientific co-founders and scientific advisory board members, Dr. Michael Czech, is a leading metabolic disease researcher. We have in-licensed intellectual property developed by Dr. Czech on genes that appear to be important regulators of metabolism. Studies conducted in Dr. Czech’s laboratory at UMMS and by others at Imperial College of London have demonstrated that inactivation of one of these genes, called RIP140, can cause fat cells to metabolize rather than store fat. Mice in these studies that did not express RIP140 remained lean and non-diabetic even when maintained on a high-fat diet. We are currently designing RNAi compounds targeting |
A-1
Table of Contents
| RIP140 as a potential treatment for obesity and obesity-related type 2 diabetes. We also continue to evaluate genes in Dr. Czech’s database for candidate targets. |
| • | Oncology. We are is initiating a program to develop RNAi drugs for use in oncology. This strategy is led by key RXi scientific advisors, Dr. Greg Hannon and Dr. Nicholas Dean, both of whom are leading researchers in targeting oncogene pathways. Additionally, our management team has expertise in developing programs targeting genes involved in cancer. Dr. Pamela Pavco, our Vice President for Pharmaceutical Development, previously managed the pre-clinical programs targeting genes involved with cancer while at Sirna Therapeutics, Inc. (recently acquired by Merck & Co.). | |
| • | Additional indications. There are many well-studied genes associated with numerous diseases that have been identified but have been difficult to target with normal medicinal chemistry. We believe RNAi technology may play an important role in targeting these genes and potentially treating these diseases. With the pioneering work being done in developing the RXi technology platform, we believe that we will discover many more drug candidates than can be advanced into clinical trials. For research on target genes in our portfolio that are not funded internally, we will seek to identify and work with partners in the discovery and development process to build our development pipeline. |
| • | World-class scientific advisors with extraordinary experience in RNAi, including Dr. Craig Mello, recipient of the 2006 Nobel Prize in Medicine for co-discovering RNAi. | |
| • | Strong early intellectual property position covering |
| • | Key therapeutic targets, | |
| • | Proprietary, potent nanotransporter delivery of active RNAi to tissues, and | |
| • | Novel approaches to RNAi chemistry. |
| • | Initial focus on significant unmet medical needs, including |
| • | Neurological disorders, | |
| • | Metabolic disease, and | |
| • | Oncology. |
| • | Management that is experienced in commercializing products, including our President and Chief Executive Officer, Tod Woolf, Ph.D. | |
| • | Potentially rapid route to the clinic for ALS therapeutics due to the high mortality rate. |
| • | We intend to finance the initial development of a limited number of RNAi drug candidates with our own capital resources and any financial resources that we may obtain from capital markets and partners. Our key therapeutic areas of interest are neurology, metabolic disease and oncology. We intend to |
A-2
Table of Contents
| develop drugs in these areas internally to establish significant value, at which point we may seek to partner them. |
| • | We intend to seek partnerships with large pharmaceutical and biotechnology companies to leverage our intellectual property and expand our development pipeline. Such partnerships may include traditionally structured drug development and commercialization licenses, discovery and development collaborations, research and technology collaborations, and intellectual property licenses. | |
| • | We intend to maintain and continue to develop and enhance our RNAi technology platform by expanding our intellectual property position in RNAi compound chemistry, delivery and target sequences. To date, we have in-licensed RNAi technologies from various institutions and companies, including UMMS, Imperial College of London, Cold Spring Harbor Laboratories and TriLink Biotechnologies. We intend to continue to enhance our technology platform through in-licensing in combination with internal and collaborative research and development programs. | |
| • | We intend to develop future RNAi technology improvements and believe we are well positioned to do so. Our management and advisors have developed much of the core technology in the field of oligonucleotide therapeutics, and more specifically RNAi. For example, our Scientific Advisory Board member, Dr. Nassim Usman, developed the standard synthesis chemistry used to manufacture RNAi throughout the world while a fellow at Massachusetts Institute of Technology (MIT). Our CEO, Dr. Tod Woolf, co-developed STEALTH RNAi, which is one of the most commonly employed second generation RNAi chemistries, and our Vice President of Pharmaceutical Development, Dr. Pamela Pavco, developed the first modified RNAi tested in humans while she was at Sirna Therapeutics. Our advisors and scientists routinely meet to discuss novel approaches and improvements in our RNAi technology platforms to enhance our intellectual property portfolio. | |
| • | We may also seek to collaborate with government and charitable institutions through grants and funded research for our development programs. |
| • | we may be unable to achieve some or all of the benefits that we expect to achieve from our separation from CytRx; | |
| • | we may not be able to obtain sufficient funding and may not be able to commercialize our product candidates; | |
| • | the approach we are taking to discover and develop novel therapeutics using RNAi is unproven and may never lead to marketable products; | |
| • | we may not be able to maintain the third-party relationships that are necessary to develop or commercialize some or all of our product candidates; | |
| • | we currently have no compounds in pre-clinical toxicology studies, and we may not be able to advance any product candidate through the pre-clinical stage into clinical trials; | |
| • | if our pre-clinical testing does not produce successful results or our clinical trials do not demonstrate safety and efficacy in humans, our products may not receive approval by the FDA or similar foreign governmental agencies and we will not be able to commercialize our drug candidates; | |
| • | even if we receive regulatory approval to market our product candidates, our product candidates may not be accepted commercially, which may prevent us from becoming profitable; and | |
| • | we are dependent on technologies we license, and if we lose the right to license such technologies or we fail to license new technologies in the future, our ability to develop new products would be harmed. |
A-3
Table of Contents
A-4
Table of Contents
| Issuer | RXi Pharmaceuticals Corporation, a Delaware corporation. | |
| Awarding Company | CytRx Corporation, a Delaware corporation. | |
| Securities to Be Awarded | Approximately 83,714 shares of common stock, $0.0001 par value per share, of RXi. | |
| Other Transactions | Concurrently with the award, CytRx is distributing by means of the distribution prospectus, 4,526,624 outstanding shares of RXi common stock to CytRx stockholders as of , 2007, the record date for the distribution, which we anticipate will be approximately ten days after the effectiveness of the registration statement of which this prospectus is a part. Immediately following the distributions made under this prospectus and the award made under the award prospectus, CytRx will own approximately 49% of the outstanding shares of RXi common stock, all of which shares are concurrently being registered for future resale by means of the resale prospectus. | |
| Reasons for the Award | The board of directors of CytRx determined to award RXi shares in connection with the separation of RXi from CytRx. The purpose is to compensate directors, officers and other employees of CytRx for services performed in connection with the separation above and beyond their usual responsibilities to CytRx. | |
| Recipients and Method of Computing Awards | CytRx will award each of its directors, officers and other employees who hold stock options to purchase CytRx common stock as of the date of this prospectus, the number of RXi shares that such individual would have received in the distribution of RXi shares to CytRx stockholders, assuming such individual had, on the record date for the distribution, exercised, in full, on a “net-exercise” basis, all such stock options to the extent then exercisable. The award will be based on the market price of CytRx common stock on the record date and the actual distribution ratio in the distribution. Based on the market price of CytRx common stock of $ on , 2007 and the estimated distribution ratio of one share of our common stock for approximately every shares of CytRx common stock held by CytRx stockholders, CytRx expects to award a total of 83,714 RXi shares under this prospectus. This distribution ratio assumes that no outstanding common stock options or stock purchase warrants of CytRx will be exercised between the date of this prospectus and the record date. The actual market price of CytRx common stock on the record date may be different than $ , and the actual distribution ratio may be less than one-for- , depending on the number of shares of CytRx common stock outstanding on the record date. For these reasons, the actual number of RXi shares awarded by CytRx may be different. | |
| Manner of Effecting the Award | The award will consist of CytRx’s payment to you of a one-time bonus in the form of shares of RXi common stock. No vote or approval of CytRx stockholders is required in connection with the award. You will not be required to pay for the RXi shares awarded |
A-5
Table of Contents
| to you or to exercise, surrender or exchange your CytRx stock options to receive your shares of our common stock. Unless you are a non-employee director, you will be required, as a condition of the award, (i) to pay CytRx an amount equal to the taxes required to be withheld (federal and state income tax, social security tax and health care tax) or (ii) to have such taxes withheld from your other compensation from CytRx or (iii) to have such taxes withheld by CytRx withholding from the award RXi shares with a value, based on the market price of RXi common stock as of the distribution date, equal to such taxes; provided, however, that the manner of withholding described in clauses (ii) and (iii) will only be available if CytRx agrees, in its discretion, to withhold in such manner. CytRx, with the assistance of Computershare, the transfer agent for RXi common stock, will electronically transfer shares of our common stock to you by way of direct registration in book-entry form. Computershare will mail to you a book-entry account statement that reflects your shares of RXi common stock. Following the award, you may request that your RXi shares be transferred to a brokerage or other account at any time, without charge. Please see “The Award — Direct Registration System” for a more detailed description of the direct registration system. | ||
| Fractional Shares | No fractional shares of RXi common stock will be awarded; instead, fractional shares will be rounded up to the nearest whole number of shares. For additional information, see “The Award — Treatment of Fractional Shares.” | |
| Record Date | The record date for the distribution is the close of business (Eastern time) on , 2007. The award of our shares to you will be based on the market price of CytRx common stock on the record date which we anticipate will be approximately ten days after the effectiveness of the registration statement of which this prospectus is a part. | |
| Expected Payment Date | The expected payment date for the award is , 2007, the expected distribution date for the distribution of our shares to CytRx stockholders pursuant to the distribution prospectus. | |
| Transfer Agent and Registrar for the Shares | Computershare Investor Services is the transfer agent and registrar for RXi common stock. | |
| United States Federal Income Tax Consequences of the Award | The award will be taxable to you as ordinary income in an amount equal to the fair market value on the payment date of the RXi shares awarded to you, and, if you are an employee, will be subject to withholding as described in the “Material Federal Income Tax Considerations” section of this prospectus. You should consult with your own individual tax advisor regarding the tax consequences of your award. | |
| Stock Exchange Listing | There is not currently a public market for our common stock. We have applied for listing of our common stock on the NASDAQ Capital Market under the symbol “RXIP.” We anticipate that trading will commence on , 2007. We cannot predict the |
A-6
Table of Contents
| trading prices for our common stock before or after the distribution date. | ||
| Relationship With CytRx after the Distribution | Immediately following the award made under this prospectus and the distribution made under the distribution prospectus, CytRx will own approximately 6,212,861, or 49%, of the outstanding shares of our common stock. CytRx is concurrently registering these shares for resale pursuant to the resale prospectus. We are a party to a number of agreements with CytRx relating to its ownership of our common stock and other matters. Please see “Arrangements With CytRx Corporation.” | |
| Post-Award Dividend Policy | RXi has never declared a dividend on its common stock and does not plan to do so for the foreseeable future. | |
| Risk Factors | You should carefully consider the matters discussed under the heading “Risk Factors.” | |
| Where You Can Obtain More Information | If you have any questions relating to the award of RXi shares, you should contact: | |
| Benjamin S. Levin Vice President — Legal Affairs CytRx Corporation 11726 San Vicente Boulevard, Suite 650 Los Angeles, California 90049 Tel:(310) 826-5648 x313 Fax:(310) 826-6139 | ||
| If you have any questions relating to the delivery of your RXi shares to you, you should contact: | ||
| Computershare Investor Services 250 Royall Street Canton, MA 02021 Tel: (781) [ ] Fax: (781) [ ] |
A-7
Table of Contents
A-8
Table of Contents
| Period from | ||||||||||||||||||||||||||||
| January 1, | ||||||||||||||||||||||||||||
| Period from | 2003 | |||||||||||||||||||||||||||
| April 3, 2006 | (Date of | |||||||||||||||||||||||||||
| (Date of | Inception) | |||||||||||||||||||||||||||
| Incorporation) | Through | |||||||||||||||||||||||||||
| Through June 30, | Six Months Ended June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||
| 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||||||||
| (Successor) | (Successor) | (Predecessor) | (Predecessor) | (Predecessor) | ||||||||||||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||||||||||||||
| (All amounts in thousands, except share and earnings per share data) | ||||||||||||||||||||||||||||
Statements of Expenses Data: | ||||||||||||||||||||||||||||
| Revenue | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Expenses: | ||||||||||||||||||||||||||||
| Research and development | 4,594 | 4,594 | 907 | 8,541 | 1,772 | 2,080 | 2,814 | |||||||||||||||||||||
| General and administrative | 1,710 | 1,710 | 155 | 1,500 | 633 | 129 | 458 | |||||||||||||||||||||
| Operating loss | (6,304 | ) | (6,304 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Interest income | 133 | 133 | — | — | — | — | — | |||||||||||||||||||||
| Loss before income taxes | (6,171 | ) | (6,171 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Income taxes | — | — | — | — | — | — | — | |||||||||||||||||||||
| Net loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | $ | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | |||||||
| Basic and diluted loss per share | $ | (0.64 | ) | $ | (0.64 | ) | NA | NA | NA | NA | NA | |||||||||||||||||
| Weighted average shares, basic and diluted | 9,671,849 | 9,671,849 | NA | NA | NA | NA | NA | |||||||||||||||||||||
Cash Flows Data: | ||||||||||||||||||||||||||||
| Cash flows used in operating activities | $ | (1,713 | ) | $ | (1,713 | ) | $ | (1,254 | ) | $ | (6,712 | ) | $ | (2,112 | ) | $ | (2,527 | ) | $ | (1,280 | ) | |||||||
| Cash flows used in investing activities | $ | (19 | ) | $ | (19 | ) | $ | — | $ | (50 | ) | $ | — | $ | (50 | ) | $ | — | ||||||||||
| Cash flows provided by financing activities | $ | 16,695 | $ | 16,694 | $ | 1,248 | $ | 6,762 | $ | 2,112 | $ | 2,577 | $ | 1,280 | ||||||||||||||
| As of | As of | |||||||||||||||||||||||
| June 30, | December, | As of December 31, | ||||||||||||||||||||||
| 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | |||||||||||||||||||
| (Successor) | (Predecessor) | |||||||||||||||||||||||
| (Unaudited) | (Successor) | (Predecessor) | ||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||
Balance Sheets and Statements of Assets, Liabilities and Parent Company’s Net Deficit: | ||||||||||||||||||||||||
| Cash and cash equivalents | $ | 14,965 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Total current assets | $ | 14,980 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Equipment and furnishings, net | $ | 59 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||
| Total assets | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | ||||||||||||
| Total current liabilities | $ | 2,490 | $ | 202 | $ | — | $ | 325 | $ | 500 | $ | 968 | ||||||||||||
| Total stockholders’ equity | $ | 12,599 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Total liabilities and stockholders’ equity | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | ||||||||||||
| Parent company’s net deficit | $ | — | $ | (152 | ) | $ | — | $ | (268 | ) | $ | (450 | ) | $ | (968 | ) | ||||||||
A-9
Table of Contents
| • | Prior to the contribution of our RNAi assets from CytRx, our RNAi research and development activities were conducted by CytRx as part of its broader operations, rather than as an independent division or subsidiary, and were primarily conducted through sponsored research arrangements rather than through internal activities. CytRx also performed various corporate functions relating to our business, including governance functions such as compliance with the Sarbanes-Oxley Act of 2002 and external reporting. Our historical financial information reflect allocations of corporate expenses from CytRx for these and similar functions. We believe that these allocations are comparable to the expenses we would have incurred had we operated as a separate publicly traded company, although we will likely incur higher expenses as an independent company; | |
| • | Our working capital requirements and capital for our general corporate purposes have historically been provided by CytRx. Following the distribution, CytRx will not be obligated to provide us with funds to finance our working capital or other cash requirements. Without financing from CytRx, we will need to obtain additional financing through public offerings or private placements of debt or equity securities, strategic relationships or other arrangements; and | |
| • | Other significant changes may occur in our cost structure, management, financing and business operations as a result of our operating as a company separate from CytRx. |
A-10
Table of Contents
| • | to conduct research and development to successfully develop our RNAi technologies, | |
| • | to obtain regulatory approval for our products, | |
| • | to file and prosecute patent applications and to defend and assess patents to protect our technologies, | |
| • | to retain qualified employees, particularly in light of intense competition for qualified scientists, | |
| • | to manufacture products ourselves or through third parties, | |
| • | to market our products, either through building our own sales and distribution capabilities or relying on third parties, and | |
| • | to acquire new technologies, licenses or products. |
A-11
Table of Contents
A-12
Table of Contents
A-13
Table of Contents
| • | delays in filing initial drug applications, | |
| • | difficulty in securing centers to conduct trials, | |
| • | conditions imposed on us by the FDA or comparable foreign authorities regarding the scope or design of our clinical trials, | |
| • | problems in engaging IRBs to oversee trials or problems in obtaining or maintaining IRB approval of studies, | |
| • | difficulty in enrolling patients in conformity with required protocols or projected timelines, | |
| • | unexpected adverse reactions by patients in trials, | |
| • | difficulty in obtaining clinical supplies of the product, | |
| • | negative or inconclusive results from our clinical trials or the clinical trials of others for drug candidates similar to our own or inability to generate statistically significant data confirming the efficacy of the product being tested, | |
| • | changes in the FDA’s requirements for our testing during the course of that testing, | |
| • | modification of the drug during testing, | |
| • | reallocation of our limited financial and other resources to other clinical programs, and | |
| • | adverse results obtained by other companies developing RNAi drugs. |
A-14
Table of Contents
A-15
Table of Contents
| • | regulators or IRBs may not authorize us to commence a clinical trial or conduct a clinical trial at a prospective trial site, | |
| • | our pre-clinical tests or clinical trials may produce negative or inconclusive results, and we may decide, or regulator may require us, to conduct additional pre-clinical testing or clinical trials, or we may abandon projects that we previously expected to be promising, | |
| • | enrollment in our clinical trials may be slower than we anticipate or participants may drop out of our clinical trials at a higher rate than we anticipate, resulting in significant delays, | |
| • | our third party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, | |
| • | our drug candidates may have very different chemical and pharmacological properties in humans than in laboratory testing and it may interact with human biological systems in unforeseen, ineffective or harmful ways. | |
| • | we might have to suspend or terminate our clinical trials if the participants are being exposed to unacceptable health risks, | |
| • | IRBs or regulators, including the FDA, may require that we hold, suspend or terminate clinical research for various reasons, including noncompliance with regulatory requirements, | |
| • | the cost of our clinical trials may be greater than we anticipate, | |
| • | the supply or quality of our drug candidates or other necessary materials necessary to conduct our clinical trials may be insufficient or inadequate, and | |
| • | effects of our drug candidates may not be the desired effects or may include undesirable side effects or the drug candidates may have other unexpected characteristics. |
A-16
Table of Contents
| • | the timing of our receipt of any marketing approvals, the terms of any approvals and the countries in which approvals are obtained, | |
| • | the safety, efficacy and ease of administration of our product candidates, | |
| • | the advantages of our product candidates over those of our competitors, | |
| • | the willingness of patients to accept relatively new therapies, | |
| • | the success of our physician education programs, | |
| • | the availability of government and third-party payor reimbursement, | |
| • | the pricing of our products, particularly as compared to alternative treatments, and | |
| • | the availability of effective alternative treatments and the relative risksand/or benefits of the treatments. |
A-17
Table of Contents
A-18
Table of Contents
| • | they are “incidental” to a physician’s services, | |
| • | they are “reasonable and necessary” for the diagnosis or treatment of the illness or injury for which they are administered according to accepted standard of medical practice, | |
| • | they are not excluded as immunizations, and | |
| • | they have been approved by the FDA. |
A-19
Table of Contents
A-20
Table of Contents
| • | announcements of regulatory developments or technological innovations by us or our competitors, | |
| • | changes in our relationship with our licensors and other strategic partners, | |
| • | changes in our ownership or other relationships with CytRx, | |
| • | our quarterly operating results, | |
| • | developments in patent or other technology ownership rights, | |
| • | public concern regarding the safety of our products, | |
| • | government regulation of drug pricing, and | |
| • | general changes in the economy, the financial markets or the pharmaceutical or biotechnology industries. |
A-21
Table of Contents
A-22
Table of Contents
| • | delaying, deferring or preventing a change in control of our company, | |
| • | impeding a merger, consolidation, takeover or other business combination involving our company, or | |
| • | discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of our company. |
| • | divide our board of directors into three classes, with members of each class to be elected for staggered three-year terms, | |
| • | limit the right of stockholders to remove directors, | |
| • | regulate how stockholders may present proposals or nominate directors for election at annual meetings of stockholders, and | |
| • | authorize our board of directors to issue preferred stock in one or more series, without stockholder approval. |
A-23
Table of Contents
A-24
Table of Contents
| • | development of our RNAi-based therapeutics may be delayed or may not proceed as planned, and we may not be able to complete development of any RNAi-based product, | |
| • | higher costs associated with being a separate, publicly trade company, | |
| • | the difficulty in evaluating our financial information due to the distribution, | |
| • | the inability to raise additional future financing and lack of financial and other resources to us as a separate company, | |
| • | our ability to control product development costs, | |
| • | we may not be able to attract and retain key employees, | |
| • | we may not be able to compete effectively, | |
| • | we may not be able enter into new strategic collaborations, | |
| • | changes in government regulation affecting our RNAi-based therapeutics could increase our development costs, | |
| • | our involvement in patent and other intellectual property litigation could be expensive and could divert management’s attention, | |
| • | the possibility that there will be no market acceptance for our products, and | |
| • | changes in third-party reimbursement policies could adversely affect potential future sales of any of our products that are approved for marketing. |
A-25
Table of Contents
A-26
Table of Contents
A-27
Table of Contents
| • | a registration statement that the SEC has declared effective under the Securities Act, or | |
| • | an exemption from registration under the Securities Act, such as the exemption afforded by Rule 144. |
A-28
Table of Contents
A-29
Table of Contents
A-30
Table of Contents
| Period from | Period from | |||||||||||||||||||||||||||||||
| April 3, | January 1, | |||||||||||||||||||||||||||||||
| 2006 (Date of | 2003 (Date of | |||||||||||||||||||||||||||||||
| Incorporation) | Inception) | |||||||||||||||||||||||||||||||
| Through | Through | |||||||||||||||||||||||||||||||
| June 30, | Six Months Ended June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||||||
| 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | 2003 | |||||||||||||||||||||||||
| (Successor) | (Successor) | (Predecessor) | (Predecessor) | (Predecessor) | ||||||||||||||||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||||||||||||||||||
| (All amounts are in thousands, except shares and earnings per share data) | ||||||||||||||||||||||||||||||||
Statements of Expenses Data: | ||||||||||||||||||||||||||||||||
| Revenue | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||||
| Expenses: | ||||||||||||||||||||||||||||||||
| Research and development | 4,594 | 4,594 | 907 | 8,541 | 1,772 | 2,080 | 2,814 | 1,877 | ||||||||||||||||||||||||
| General and administrative | 1,710 | 1,710 | 155 | 1,500 | 633 | 129 | 458 | 278 | ||||||||||||||||||||||||
| Operating loss | (6,304 | ) | (6,304 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | (2,155 | ) | ||||||||||||||||
| Interest income | 133 | 133 | — | — | — | — | — | — | ||||||||||||||||||||||||
| Loss before income taxes | (6,171 | ) | (6,171 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | (2,155 | ) | ||||||||||||||||
| Income taxes | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||
| Net loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | $ | (2,155 | ) | |||||||||
| Basic and diluted loss per share | $ | (0.64 | ) | $ | (0.64 | ) | NA | NA | NA | NA | NA | NA | ||||||||||||||||||||
| Weighted average shares, basic and diluted | 9,671,849 | 9,671,849 | NA | NA | NA | NA | NA | NA | ||||||||||||||||||||||||
| Balance Sheets and | ||||||||||||||||||||||||||||
| Statements of Assets, | As of | |||||||||||||||||||||||||||
| Liabilities and Parent | As of June 30, | December 31, | As of December 31, | |||||||||||||||||||||||||
Company’s Net Deficit: | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | 2003 | |||||||||||||||||||||
| (Successor) | (Predecessor) | (Successor) | (Predecessor) | |||||||||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 14,965 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Total current assets | $ | 14,980 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Working capital | $ | 12,490 | $ | (202 | ) | $ | 2 | $ | (318 | ) | $ | (500 | ) | $ | (968 | ) | $ | (89 | ) | |||||||||
| Total assets | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | $ | — | ||||||||||||||
| Due to Parent | $ | (1,678 | ) | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||
| Total stockholders’ equity | $ | 12,599 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Parent company’s net deficit | $ | — | $ | (152 | ) | $ | — | $ | (268 | ) | $ | (450 | ) | $ | (968 | ) | $ | (89 | ) | |||||||||
A-31
Table of Contents
A-32
Table of Contents
| • | our ability to advance product candidates into pre-clinical research and clinical trials; | |
| • | the scope and rate of progress of our pre-clinical program and other research and development activities; | |
| • | the scope, rate of progress and cost of any clinical trials we commence; | |
| • | the cost of filing, prosecuting, defending and enforcing patent claims and other intellectual property rights; | |
| • | clinical trial results; | |
| • | the terms and timing of any collaborative, licensing and other arrangements that we may establish; | |
| • | the cost and timing of regulatory approvals; | |
| • | the cost of establishing clinical and commercial supplies of our product candidates and any products that we may develop; | |
| • | the cost and timing of establishing sales, marketing and distribution capabilities; | |
| • | the effect of competing technological and market developments; and | |
| • | the effect of government regulation and insurance industry efforts to control healthcare costs through reimbursement policy and other cost management strategies. |
A-33
Table of Contents
A-34
Table of Contents
A-35
Table of Contents
A-36
Table of Contents
A-37
Table of Contents
A-38
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| June 30, | Years Ended December 31, | |||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | ||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| Research and development expense | $ | 1,271 | $ | 802 | $ | 1,298 | $ | 1,929 | $ | 1,700 | ||||||||||
| Research and development non-employee stock-based compensation expense | 1,012 | 105 | 212 | 151 | 1,006 | |||||||||||||||
| Fair value of common stock issued in exchange for licensing rights | 2,311 | — | 262 | — | 108 | |||||||||||||||
| Total research and development expense | $ | 4,594 | $ | 907 | $ | 1,772 | $ | 2,080 | $ | 2,814 | ||||||||||
A-39
Table of Contents
A-40
Table of Contents
| Years Ended | ||||||||||||||||||||
| Six Months Ended June 30, | December 31, | |||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | ||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| General and administrative expense | $ | 1,381 | $ | 155 | $ | 633 | $ | 129 | $ | 458 | ||||||||||
| Common stock, and stock options issued for general and administrative expense | 329 | — | — | — | — | |||||||||||||||
| Total general and administrative expense | $ | 1,710 | $ | 155 | $ | 633 | $ | 129 | $ | 458 | ||||||||||
A-41
Table of Contents
A-42
Table of Contents
| Non-Cancelable | Cancelable | |||||||||||||||||||
| Operating | Employment | License | ||||||||||||||||||
Years Ending December 31, | Leases(1) | Agreements(2) | Subtotal | Agreements(3) | Total | |||||||||||||||
| 2007 | $ | 250 | $ | 951 | $ | 1,201 | $ | 2,892 | $ | 4,093 | ||||||||||
| 2008 | 9 | 810 | 819 | 433 | 1,252 | |||||||||||||||
| 2009 | — | 710 | 710 | 383 | 1,093 | |||||||||||||||
| 2010 | — | 460 | 460 | 333 | 793 | |||||||||||||||
| 2011 | — | 255 | 255 | 333 | 588 | |||||||||||||||
| Thereafter | — | 170 | 170 | 7,568 | 7,738 | |||||||||||||||
| Total | $ | 259 | $ | 3,356 | $ | 3,615 | $ | 11,942 | $ | 15,557 | ||||||||||
| (1) | Operating leases are primarily facility and equipment related obligations with third-party vendors. Facility lease expenses during the six months ended June 30, 2007 and 2006 were approximately $68,000 and $1,000, respectively. Facility lease expense during the years ended December 31, 2006, 2005 and 2004 was $2,000, $2,000 and $3,000 respectively. | |
| (2) | Employment agreement obligations include management contracts, as well as scientific advisory board member compensation agreements. Certain agreements provide for minimum salary levels, adjusted annually at the discretion of RXi’s Board of Directors, as well as for minimum annual bonuses. | |
| (3) | License agreements primarily relate to our obligations with UMMS associated with RNAi and, for future periods, represent minimum annual royalty payment obligations. Included in the 2007 license obligations was $2,310,000 that was paid to UMMS in the second quarter of 2007 in the form of shares of common stock of RXi. |
A-43
Table of Contents
A-44
Table of Contents
| • | Neurology. Initially, we are pursuing research in ALS (amyotrophic lateral sclerosis, commonly known as Lou Gehrig’s Disease). Some forms of ALS are caused by defects in a gene called SOD1. Early preclinical studies conducted by our advisors, Dr. Tariq Rana and Dr. Zuoshang Xu at UMMS, showed promising results in animals using an RNAi compound to selectively inhibit the SOD1 gene. We are refining and extending this work and, if successful, will move into formal preclinical development. We also intend to leverage our experience related to the delivery of RNAi therapeutics in the central nervous system to explore development of RNAi-based treatments for neurodegenerative diseases other than ALS, including Alzheimer’s Disease. | |
| • | Metabolic disease. One of our scientific co-founders and scientific advisory board members, Dr. Michael Czech, is a leading metabolic disease researcher. We have in-licensed intellectual property developed by Dr. Czech on genes that appear to be important regulators of metabolism. Studies conducted in Dr. Czech’s laboratory at UMMS and by others at Imperial College of London have demonstrated that inactivation of one of these genes, called RIP140, can cause fat cells to metabolize rather than store fat. Mice in these studies that did not express RIP140 remained lean and non-diabetic even when maintained on a high-fat diet. We are currently designing RNAi compounds targeting RIP140 as a potential treatment for obesity and obesity-related type 2 diabetes. We also continue to evaluate genes in Dr. Czech’s database for candidate targets. | |
| • | Oncology. We are initiating a program to develop RNAi drugs for use in oncology. This strategy is led by key RXi scientific advisors, Dr. Greg Hannon and Dr. Nicholas Dean, both of whom are leading researchers in targeting oncogene pathways. Additionally, our management team has expertise in developing programs targeting genes involved in cancer. Dr. Pamela Pavco, our Vice President for Pharmaceutical Development, previously managed the pre-clinical programs targeting genes involved with cancer while at Sirna Therapeutics, Inc. (recently acquired by Merck & Co.). | |
| • | Additional indications. There are many well-studied genes associated with numerous diseases that have been identified but have been difficult to target with normal medicinal chemistry. We believe RNAi technology may play an important role in targeting these genes and potentially treating these diseases. With the pioneering work being done in developing the RXi technology platform, we believe |
A-45
Table of Contents
| that we will discover many more drug candidates than can be advanced into clinical trials. For research on target genes in our portfolio that are not funded internally, we will seek to identify and work with partners in the discovery and development process to build our development pipeline. |
| • | World-class scientific advisors with extraordinary experience in RNAi, including Dr. Craig Mello, recipient of the 2006 Nobel Prize in Medicine for co-discovering RNAi. | |
| • | Strong early intellectual property position covering |
| • | Key therapeutic targets, | |
| • | Proprietary, potent nanotransporter delivery of active RNAi to tissues, and | |
| • | Novel approaches to RNAi chemistry. |
| • | Initial focus on significant unmet medical needs, including |
| • | Neurological disorders, | |
| • | Metabolic disease, and | |
| • | Oncology. |
| • | Management that is experienced in commercializing products, including our President and Chief Executive Officer, Tod Woolf, Ph.D. | |
| • | Potentially rapid route to the clinic for ALS therapeutics due to the high mortality rate. |
A-46
Table of Contents

| • | high specificity for targeted genes; | |
| • | high potency (low doses); | |
| • | potential interference with the expression of any gene; and | |
| • | accelerated development of lead compounds. |
A-47
Table of Contents

| • | up to x100 more active than conventional siRNA (depending on the target site), | |
| • | nuclease resistant, | |
| • | readily manufactured, | |
| • | comprised of elements from RXi’s IP portfolio, and | |
| • | potentially more specific for the target gene. |
A-48
Table of Contents

| • | inhibition of liver target with 1 mg RNAi compound per kg of body weight; | |
| • | no immune stimulation detected; | |
| • | defined particle size; and | |
| • | readily formulated. |
A-49
Table of Contents
A-50
Table of Contents
| • | We intend to finance the initial development of a limited number of RNAi drug candidates with our own capital resources and any financial resources that we may obtain from capital markets and partners. Our key therapeutic areas of interest are neurology, metabolic disease and oncology. We intend to develop drugs in these areas internally to establish significant value, at which point we may seek to partner them. |
A-51
Table of Contents
| • | We intend to seek partnerships with large pharmaceutical and biotechnology companies to leverage our intellectual property and expand our development pipeline. Such partnerships may include traditionally structured drug development and commercialization licenses, discovery and development collaborations, research and technology collaborations, and intellectual property licenses. | |
| • | We intend to maintain and continue to develop and enhance our RNAi technology platform by expanding our intellectual property position in RNAi compound chemistry, delivery and target sequences. To date, we have in-licensed RNAi technologies from various institutions and companies, including UMMS, Imperial College of London, Cold Spring Harbor Laboratories and TriLink Biotechnologies. We intend to continue to enhance our technology platform through in-licensing in combination with internal and collaborative research and development programs. | |
| • | We intend to develop future RNAi technology improvements and believe we are well-positioned to do so. Our management and advisors have developed much of the core technology in the field of oligonucleotide therapeutics, and more specifically RNAi. For example, our Scientific Advisory Board member, Dr. Nassim Usman, developed the standard synthesis chemistry used to manufacture RNAi compounds throughout the world while a fellow at MIT. Our CEO, Dr. Tod Woolf, co-developed STEALTH RNAi, which is one of the most commonly employed second generation RNAi chemistries, and our Vice President of Pharmaceutical Development, Dr. Pamela Pavco, developed the first modified RNAi tested in humans while she was at Sirna Therapeutics. Our advisors and scientists routinely meet to discuss novel approaches and improvements in our RNAi technology platforms to enhance our intellectual property portfolio. | |
| • | We may also seek to collaborate with government and charitable institutions through grants and funded research for our development programs. |
| • | therapeutic targets, | |
| • | chemistry and configurations of RNAi compounds, and | |
| • | delivery technology for RNAi. |
A-52
Table of Contents
| • | Dr. Rana’s inventions which provide fundamental rules for designing chemically modified RNAi sequences suitable forin vivo gene silencing, to which we have co-exclusive licenses in the therapeutic areas of type 2 diabetes, obesity, and ALS by targeting mutant SOD1, and CMV retinitis; | |
| • | Dr. Tuschl’s invention regarding RNAi therapeutics using double-stranded RNAs in the areas of type 2 diabetes, obesity, and ALS by targeting mutant SOD1, and CMV retinitis; | |
| • | Drs. Mello and Zamore’s invention regardingin vivoproduction of siRNA; and | |
| • | methods of triggering RNAiin vivo, based on particular RNA structural characteristics. |
| • | methods and compositions, including use of nanotransporters, for RNAi compound delivery which enable therapeutic gene silencing in cells and animals; and | |
| • | inhibition of gene expression in fat cells using RNAi. |
Subject Field | Inventor(s) | Priority Date | License* | Status | ||||
| Drug discovery for diabetes and obesity | Michael P. Czech Silvia Corvera | 09/27/1993 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | US Pat. 5,989,893 Pending elsewhere | ||||
| RNAi in general | Craig C. Mello Andrew Fire Stephen Kostas Mary Montgomery Lisa Timmons SiQun Zu Hiroaki Tabara Samuel E. Driver | 12/23/1997 | Non-exclusive license to fundamental RNAi from Carnegie Institute of Washington. | US Pat. 6,506,559 Pending elsewhere | ||||
| Drug discovery using genomic databases for diabetes and obesity | Michael P. Czech Andrew D. Cherniack Adelson L. Guilherme | 10/20/2000 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending |
A-53
Table of Contents
Subject Field | Inventor(s) | Priority Date | License* | Status | ||||
| dsRNA in general | Thomas Tuschl, Philip A. Sharp Phillip D. Zamore David P. Bartel | 12/01/2000 | Non-exclusive license for RNAi use to inhibit (1) HCMV immediate early gene in retinitis (2) mutant SOD1 gene in ALS, and (3) gene targets in type II diabetes & obesity from UMMS, a co-owner of the patent. | Pending | ||||
| Engineered precursor to siRNA for in vivo production of siRNA | Phillip D. Zamore Gyorgy Hutvagner Juanita McLachlan Craig C. Mello Alla Grishok | 07/12/2001 | Exclusive license for RNAi use to inhibit (1) HCMV immediate early gene in retinitis (2) mutant SOD1 gene in ALS, and (3) gene targets in type II diabetes & obesity. | Pending | ||||
| In vivo gene silencing by chemically modified and stable siRNA | Tariq M. Rana Ya-Lin Chiu | 09/25/2002 | Exclusive worldwide license to the treatment of CMV, ALS, diabetes & obesity for therapeutics, prophylactics or diagnostics. | Pending | ||||
| Allele specific inhibition by siRNA, especially SOD inhibition for the treatment of ALS | Zuoshang Xu Phillip D. Zamore | 11/04/2002 | Exclusive license on inhibition of SOD1 expression for the treatment of ALS including therapeutics, prophylactics and diagnostics. | Pending | ||||
| Delivery of siRNA using peptide conjugate | Tariq M. Rana | 11/26/2002 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans, including for ALS, diabetes and obesity. | Pending | ||||
| Allele specific inhibition by chemically modified siRNA | Tariq M. Rana | 11/26/2002 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending |
A-54
Table of Contents
Subject Field | Inventor(s) | Priority Date | License* | Status | ||||
| Delivery of siRNA into adipocytes for treatment of diabetes and obesity; | Michael P. Czech Qiong L. Zhou Zhen Y. Jiang | 12/11/2002 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending | ||||
| Screening for modulators of fat storage | Malcolm Parker Roger White Goran Leonardsson | 02/03/2003 | Exclusive license for screening methods for identifying compounds that are useful as modulators of fat storage from Imperial College, UK. | Pending | ||||
| Inhibition of CMV using siRNA | Timothy F. Kowalik | 02/05/2003 | Exclusive license to develop RNAi therapeutics for cytomegalovirus related diseases (HCMV- immediate early gene). (Note that original license only for CMV retinitis; amended 01/04 to include all CMV) | Pending | ||||
| Modulation of insulin sensitivity through stress proteins | Silvia Corvera | 06/19/2003 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending | ||||
| Chemically modified siRNA and their uses | Tariq M. Rana Ya-Lin Chiu | 08/05/2003 | Co-Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications of RNAi for inhibition of human CMV immediate early gene expression, mutant SOD1 (ALS treatment), genes implicated in diabetes and obesity. | Pending | ||||
| Increase of insulin stimulated glucose regulation by a small molecule inhibitor of RIP140 | Michael P. Czech Aimee Powelka Adilson L. Guilherme Andrew D. Cherniack | 03/05/2004 | Exclusive license for drug discovery in type 2 diabetes and/or obesity with therapeutic, prophylactic, or diagnostic applications and products for treatment, prevention or diagnosis of type 2 diabetes and/or obesity. | Pending |
A-55
Table of Contents
Subject Field | Inventor(s) | Priority Date | License* | Status | ||||
| Efficient delivery of siRNA into cells and animals | Tariq M. Rana | 08/11/2005 | Nonexclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Delivery of chemically modified siRNA using nanotransporters | Tariq M. Rana (Zuoshang Xu) | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Delivery of chemically modified siRNA | Tariq M. Rana | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Gene silencing of cholesterol biosynthesis and other metabolic genes by chemically modified siRNA | Tariq M. Rana | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Microwave assisted nucleic acid delivery system | Tariq M. Rana | Not yet filed | Nonexclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Invention disclosure | ||||
| Methods and compositions for RNA interference in suspended cells or in whole body | Scott Hammond Gregory Hannon David Beach Amy Caudy Emily Bernstein | 03/16/2001 | Non-exclusive license from Cold Spring Harbor Laboratory for use of short hairpin RNA for drug discovery and development. | Pending |
| * | Unless otherwise noted, the licenses are from UMMS. |
A-56
Table of Contents
A-57
Table of Contents
A-58
Table of Contents
A-59
Table of Contents
A-60
Table of Contents
A-61
Table of Contents
Name | Position | Age | ||||||
| Sanford J. Hillsberg(1)(2) | Director and Chairman of the Board | 59 | ||||||
| Tod Woolf, Ph.D. | President and Chief Executive Officer, Director | 43 | ||||||
| Mark J. Ahn, Ph.D.(1)(2)(3) | Director | 44 | ||||||
| Stephen S. Galliker(1)(2)(3) | Director | 60 | ||||||
| Steven A. Kriegsman(3) | Director | 65 | ||||||
| Stephen J. DiPalma | Chief Financial Officer and Secretary | 48 | ||||||
| Pamela Pavco, Ph.D. | Vice President of Pharmaceutical Development | 51 | ||||||
| Dmitry Samarsky, Ph.D. | Vice President of Technology and Business Development | 40 | ||||||
| James Warren | Former Chief Financial Officer and Secretary | 62 | ||||||
| (1) | Member of our Nominating and Governance Committee | |
| (2) | Member of our Audit Committee | |
| (3) | Member of our Compensation Committee |
Name | Professional Affiliation | |||
| Craig C. Mello, Ph.D (Chairman) | • | Professor and Blaise University Chair in Molecular Medicine at the University of Massachusetts Medical School | ||
| • | Investigator, Howard Hughes Medical Institute | |||
| • | Member of the National Academy of Sciences | |||
| Tariq Rana, Ph.D | • | Professor of Biochemistry & Molecular Pharmacology and Founding Director of the Program in Chemical Biology at the University of Massachusetts Medical School | ||
| Gregory Hannon, Ph.D | • | Investigator, Howard Hughes Medical Institute | ||
| Michael Czech, Ph.D | • | Professor and Chair in Molecular Medicine at the University of Massachusetts Medical School | ||
| Nicholas Dean, Ph.D | • | Founder and Chief Scientific Officer of Excaliard Pharmaceuticals | ||
| Nassim Usman, Ph.D | • | Chief Executive Officer and Board Member of Catalyst Biosciences | ||
A-62
Table of Contents
A-63
Table of Contents
A-64
Table of Contents
A-65
Table of Contents
A-66
Table of Contents
A-67
Table of Contents
| • | an annual retainer fee of $10,000, payable quarterly, | |
| • | an annual retainer fee of $12,000 for the chairperson of each committee of our board of directors other than the audit committee, payable quarterly, | |
| • | an annual retainer fee of $20,000 for the chairperson of the audit committee of our board of directors, payable quarterly, | |
| • | an annual retainer fee of $32,000 for the Chairman of the board of directors, payable quarterly, | |
| • | a fee of $2,000 per board meeting attended by the director ($1,000 if attendance is telephonic), and | |
| • | a fee of $1,500 per committee meeting attended by the director ($750 if attendance is telephonic). |
A-68
Table of Contents
A-69
Table of Contents
| • | base salary, | |
| • | performance-based cash compensation, and | |
| • | long-term equity incentive compensation. |
| • | the negotiated terms of each executive employment agreement, | |
| • | internal review of the executive’s compensation, both individually and relative to other executive officers, and | |
| • | individual performance of the executive. |
A-70
Table of Contents
| • | establish the link between the creation of stockholder value and long-term executive incentive compensation, | |
| • | provide an opportunity for increased equity ownership by executives, | |
| • | function as a retention tool because of the vesting features included in all options granted by the Committee, and | |
| • | maintain competitive levels of total compensation. |
A-71
Table of Contents
A-72
Table of Contents
| Value of | Value of | |||||||||||||||||||
| Salary | Benefit | Accelerated Option | Accelerated Option | |||||||||||||||||
| Name and Principal | Continuation | Continuation | Vesting | Total | Vesting Upon a | |||||||||||||||
Position | (Termination) | (Termination) | (Termination) | (Termination) | Change in Control | |||||||||||||||
| Tod Woolf | $ | 250,000 | $ | 14,500 | $ | 449,000 | $ | 714,400 | $ | 1,087,100 | ||||||||||
| President and Chief Executive Officer | ||||||||||||||||||||
| Stephen J. DiPalma | $ | 110,000 | $ | 7,300 | — | $ | 117,300 | $ | 406,400 | |||||||||||
| Chief Financial Officer | ||||||||||||||||||||
| Pamela Pavco(1) | $ | 198,000 | $ | 2,100 | $ | 157,200 | $ | 357,300 | $ | 511,000 | ||||||||||
| Vice President of Research and Development | ||||||||||||||||||||
| Dmitry Samarsky(1) | $ | 85,000 | $ | 1,800 | $ | 57,200 | $ | 144,000 | $ | 400,400 | ||||||||||
| Vice President of Technology and Business Development | ||||||||||||||||||||
| (1) | Each of Drs. Pavco and Samarsky are entitled to specified benefits in the event his or her employment is terminated without cause or he or she terminates his or her employment for good reason following a change in control, as further described below. |
A-73
Table of Contents
A-74
Table of Contents
A-75
Table of Contents
A-76
Table of Contents
A-77
Table of Contents
A-78
Table of Contents
A-79
Table of Contents
A-80
Table of Contents
A-81
Table of Contents
A-82
Table of Contents
A-83
Table of Contents
| • | any person known by us to be the beneficial owner of 5% or more of our common stock, including any “group” as that term is defined in the Exchange Act, | |
| • | each director, our named executive officers identified in the “Management-Executive Compensation” section above, and | |
| • | all of our directors and executive officers as a group. |
| After the Distribution and | ||||||||||||||||
| Award Transactions | ||||||||||||||||
| Described in this | ||||||||||||||||
| Before the Distribution | Prospectus | |||||||||||||||
| Number | Number | |||||||||||||||
| of | of | |||||||||||||||
Name | Shares | Percent | Shares | Percent | ||||||||||||
5% Stockholders: | ||||||||||||||||
| CytRx Corporation(1) | 10,823,199 | 85.8 | % | 6,212,861 | [49.3 | %] | ||||||||||
Executive Officers and Directors: | ||||||||||||||||
| Tod Woolf, Ph.D.(2) | 52,832 | # | 52,832 | # | ||||||||||||
| Stephen J. DiPalma(3) | 6,250 | # | 6,250 | # | ||||||||||||
| James Warren(4) | 62,743 | # | 62,743 | # | ||||||||||||
| Pamela Pavco, Ph.D.(5) | 18,163 | # | 18,163 | # | ||||||||||||
| Dmitry Samarsky, Ph.D.(6) | 6,597 | # | 6,597 | # | ||||||||||||
| Mark J. Ahn, Ph.D.(7) | 47,500 | # | 47,500 | # | ||||||||||||
| Stephen S. Galliker(7) | 47,500 | # | 47,500 | # | ||||||||||||
| Sanford J. Hillsberg(7)(8) | 47,500 | # | 47,500 | # | ||||||||||||
| Steven A. Kriegsman(9) | 37,500 | # | 278,268 | 2.2 | % | |||||||||||
| All executive officers and directors as a group (8 persons)(10) | 296,585 | 2.50 | % | 567,353 | 4.4 | % | ||||||||||
| # | Represents less than 1% of the outstanding shares of our common stock. |
A-84
Table of Contents
| (1) | The address for CytRx is 11726 San Vicente Boulevard, Suite 650, Los Angeles, California 90049. | |
| (2) | Consists of 52,832 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (3) | Consists of 6,250 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (4) | Consists of 62,743 shares of common stock underlying stock options currently exercisable until November 30, 2007. | |
| (5) | Consists of 18,163 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (6) | Includes 6,597 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (7) | Includes 37,500 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (8) | The shares shown do not include shares owned by Troy & Gould Professional Corporation. | |
| (9) | Includes 37,500 shares of common stock underlying stock options and exercisable within 60 days of October 1, 2007 and, following the distribution and award transactions described in this prospectus, approximately 240,768 shares that Mr. Kriegsman will receive as a result of distribution under this prospectus and the award under the award prospectus. Please see “The Distribution” for a further explanation of the allocation of shares in the distribution. Mr. Kriegsman is the CEO and a director of CytRx, but acting alone, he has neither voting nor investment power with respect to the shares beneficially owned by CytRx. As a result, Mr. Kriegsman disclaims beneficial ownership of such shares. | |
| (10) | Includes 293,282 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. |
A-85
Table of Contents
A-86
Table of Contents
A-87
Table of Contents
| • | prior to such date, the board of directors of the corporation approved either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder, | |
| • | upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, other than statutorily excluded shares, or | |
| • | on or subsequent to such date, the business combination is approved by the board of directors of the corporation and authorized at an annual or special meeting of the stockholders by the affirmative vote of at least 662/3% of the outstanding voting stock which is not owned by the interested stockholder. |
| • | any person that is the owner of 15% or more of the outstanding voting stock of the corporation, or is an affiliate or associate of the corporation and was the owner of 15% or more of the outstanding voting stock of the corporation at any time within three years immediately prior to the date of determination, and | |
| • | the affiliates and associates of any such person. |
A-88
Table of Contents
A-89
Table of Contents
A-90
Table of Contents
A-91
Table of Contents
A-92
Table of Contents
Table of Contents
| We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities, and we are not soliciting an offer to buy these securities in any state where the offer or sale is not permitted. |

Table of Contents
| Page | ||||
| B-1 | ||||
| B-8 | ||||
| B-22 | ||||
| B-23 | ||||
| B-23 | ||||
| B-24 | ||||
| B-26 | ||||
| B-39 | ||||
| B-56 | ||||
| B-63 | ||||
| B-74 | ||||
| B-76 | ||||
| B-78 | ||||
| B-80 | ||||
| B-81 | ||||
| B-84 | ||||
| B-87 | ||||
| B-87 | ||||
| B-87 | ||||
| B-88 | ||||
| F-1 | ||||
B-i
Table of Contents
| • | Neurology. Initially, we are pursuing research in ALS (amyotrophic lateral sclerosis, commonly known as Lou Gehrig’s Disease). Some forms of ALS are caused by defects in a gene called SOD1. Early preclinical studies conducted by RXi advisors, Dr. Tariq Rana and Dr. Zuoshang Xu at UMMS, showed promising results in animals using an RNAi compound to selectively inhibit the SOD1 gene. We are refining and extending this work and, if successful, will move into formal preclinical development. We also intend to leverage our experience related to the delivery of RNAi therapeutics in the central nervous system to explore development of RNAi-based treatments for neurodegenerative diseases other than ALS, including Alzheimer’s Disease. | |
| • | Metabolic disease. One of our scientific co-founders and scientific advisory board members, Dr. Michael Czech, is a leading metabolic disease researcher. We have in-licensed intellectual property developed by Dr. Czech on genes that appear to be important regulators of metabolism. Studies conducted in Dr. Czech’s laboratory at UMMS and by others at Imperial College of London have demonstrated that inactivation of one of these genes, called RIP140, can cause fat cells to metabolize rather than store fat. Mice in these studied that did not express RIP140 remained lean and non-diabetic even when maintained on a high fat diet. We are currently designing RNAi compounds targeting |
B-1
Table of Contents
| RIP140 as a potential treatment for obesity and obesity-related type 2 diabetes. We also continue to evaluate genes in Dr. Czech’s database for candidate targets. |
| • | Oncology. We are initiating a program to develop RNAi drugs for use in oncology. This strategy is led by key RXi scientific advisors, Dr. Greg Hannon and Dr. Nicholas Dean, both of whom are leading researchers in targeting oncogene pathways. Additionally, our management team has expertise in developing programs targeting genes involved in cancer. Dr. Pamela Pavco, our Vice President for Pharmaceutical Development, previously managed the pre-clinical programs targeting genes involved with cancer while at Sirna Therapeutics, Inc. (recently acquired by Merck & Co.). | |
| • | Additional indications. There are many well-studied genes associated with numerous diseases that have been identified but have been difficult to target with normal medicinal chemistry. We believe RNAi technology may play an important role in targeting these genes and potentially treating these diseases. With the pioneering work being done in developing the RXi technology platform, we believe that we will discover many more drug candidates than can be advanced into clinical trials. For research on target genes in our portfolio that are not funded internally, we will seek to identify and work with partners in the discovery and development process to build our development pipeline. |
| • | World-class scientific advisors with extraordinary experience in RNAi, including Dr. Craig Mello, recipient of the 2006 Nobel Prize in Medicine for co-discovering RNAi. | |
| • | Strong early intellectual property position covering |
| • | Key therapeutic targets, | |
| • | Proprietary, potent nanotransporter delivery of active RNAi to tissues, and | |
| • | Novel approaches to RNAi chemistry. |
| • | Initial focus on significant unmet medical needs, including |
| • | Neurological disorders, | |
| • | Metabolic disease, and | |
| • | Oncology. |
| • | Management that is experienced in commercializing products, including President and Chief Executive Officer, Tod Woolf, Ph.D. | |
| • | Potentially rapid route to the clinic for ALS therapeutics due to the high mortality rate. |
| • | We intend to finance the initial development of a limited number of RNAi drug candidates with our own capital resources and any financial resources that we may obtain from capital markets and partners. Our key therapeutic areas of interest are neurology, metabolic disease and oncology. We intend to |
B-2
Table of Contents
| develop drugs in these areas internally to establish significant value, at which point we may seek to partner them. |
| • | We intend to seek partnerships with large pharmaceutical and biotechnology companies to leverage our intellectual property and expand our development pipeline. Such partnerships may include traditionally structured drug development and commercialization licenses, discovery and development collaborations, research and technology collaborations, and intellectual property licenses. | |
| • | We intend to maintain and continue to develop and enhance our RNAi technology platform by expanding our intellectual property position in RNAi compound chemistry, delivery and target sequences. To date, we have in-licensed RNAi technologies from various institutions and companies, including UMMS, Imperial College of London, Cold Spring Harbor Laboratories and TriLink Biotechnologies. We intend to continue to enhance our technology platform through in-licensing in combination with internal and collaborative research and development programs. | |
| • | We intend to develop future RNAi technology improvements and believe we are well positioned to do so. Our management and advisors have developed much of the core technology in the field of oligonucleotide therapeutics, and more specifically RNAi. For example, our Scientific Advisory Board member, Dr. Nassim Usman, developed the standard synthesis chemistry used to manufacture RNAi throughout the world while a fellow at Massachusetts Institute of Technology (MIT). Our CEO, Dr. Tod Woolf, co-developed STEALTH RNAi, which is one of the most commonly employed second generation RNAi chemistries, and our Vice President of Pharmaceutical Development, Dr. Pamela Pavco, developed the first modified RNAi tested in humans while she was at Sirna Therapeutics. Our advisors and scientists routinely meet to discuss novel approaches and improvements in our RNAi technology platforms to enhance our intellectual property portfolio. | |
| • | We may also seek to collaborate with government and charitable institutions through grants and funded research for our development programs. |
| • | we may be unable to achieve some or all of the benefits that we expect to achieve from our separation from CytRx; | |
| • | we may not be able to obtain sufficient funding and may not be able to commercialize our product candidates; | |
| • | the approach we are taking to discover and develop novel therapeutics using RNAi is unproven and may never lead to marketable products; | |
| • | we may not be able to maintain the third-party relationships that are necessary to develop or commercialize some or all of our product candidates; | |
| • | we currently have no compounds in pre-clinical toxicology studies, and we may not be able to advance any product candidate through the pre-clinical stage into clinical trials; | |
| • | if our pre-clinical testing does not produce successful results or our clinical trials do not demonstrate safety and efficacy in humans, our products may not receive approval by the FDA or similar foreign governmental agencies and we will not be able to commercialize our drug candidates; | |
| • | even if we receive regulatory approval to market our product candidates, our product candidates may not be accepted commercially, which may prevent us from becoming profitable; and | |
| • | we are dependent on technologies we license, and if we lose the right to license such technologies or we fail to license new technologies in the future, our ability to develop new products would be harmed. |
B-3
Table of Contents
B-4
Table of Contents
| Issuer | RXi Pharmaceuticals Corporation, a Delaware corporation. | |
| Selling Stockholder | CytRx Corporation, a Delaware corporation. | |
| Shares Offered | 6,212,861 shares. | |
| Shares to be Outstanding beforeand after theOffering | 12,618,387 shares, excluding 1,413,427 shares subject to outstanding stock options. | |
| Use of Proceeds | CytRx will receive all proceeds from the sale of shares under this prospectus. We will not receive any proceeds from the sale of the shares by CytRx. | |
| Other Transactions | Concurrently with the sale of the shares being offered by this prospectus, CytRx is distributing approximately 4,526,624 outstanding shares of our common stock to CytRx stockholders as of , 2007, the record date for the distribution which we anticipate will be approximately ten days after the effectiveness of the registration statement of which this prospectus is a part, and is awarding to some of its directors, officers and other employees up to approximately 83,714 additional outstanding shares of our common stock. The distribution and award by CytRx of our shares are being made by means of the distribution prospectus and the award prospectus, respectively. | |
| Stock Exchange Listing | There is not currently a public market for our common stock. We have applied for listing of our common stock on the NASDAQ Capital Market under the symbol “RXIP.” We anticipate that trading will commence on , 2007, the expected distribution date for the distribution of our shares to CytRx stockholders. We cannot predict the trading prices for our common stock before or after the distribution date. | |
| Relationship With CytRx | We are a party to a number of agreements with CytRx relating to its ownership of our common stock and other matters. Please see “Arrangements With CytRx Corporation” for a discussion of these agreements. | |
| Risk Factors | An investment in our shares involves a high degree of risk. You should consider carefully the risks described in the “Risk Factors” section of this prospectus before purchasing any shares. |
B-5
Table of Contents
B-6
Table of Contents
| Period from | ||||||||||||||||||||||||||||
| Period from | January 1, 2003 | |||||||||||||||||||||||||||
| April 3, 2006 | (Date of | |||||||||||||||||||||||||||
| (Date of | Inception) | |||||||||||||||||||||||||||
| Incorporation) | Through | |||||||||||||||||||||||||||
| Through June 30, | Six Months Ended June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||
| 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | (Predecessor) | |||||||||||||||||||||||||
| (Successor) | ||||||||||||||||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||||||||||||||
| (All amounts in thousands, except share and earnings per share data) | ||||||||||||||||||||||||||||
Statements of Expenses Data: | ||||||||||||||||||||||||||||
| Revenue | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| �� | ||||||||||||||||||||||||||||
| Expenses: | ||||||||||||||||||||||||||||
| Research and development | 4,594 | 4,594 | 907 | 8,541 | 1,772 | 2,080 | 2,814 | |||||||||||||||||||||
| General and administrative | 1,710 | 1,710 | 155 | 1,500 | 633 | 129 | 458 | |||||||||||||||||||||
| Operating loss | (6,304 | ) | (6,304 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Interest income | 133 | 133 | — | — | — | — | — | |||||||||||||||||||||
| Loss before income taxes | (6,171 | ) | (6,171 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | ||||||||||||||
| Income taxes | — | — | — | — | — | — | — | |||||||||||||||||||||
| Net loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | $ | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | |||||||
| Basic and diluted loss per share | $ | (0.64 | ) | $ | (0.64 | ) | NA | NA | NA | NA | NA | |||||||||||||||||
| Weighted average shares, basic and diluted | 9,671,849 | 9,671,849 | NA | NA | NA | NA | NA | |||||||||||||||||||||
Cash Flows Data: | ||||||||||||||||||||||||||||
| Cash flows used in operating activities | $ | (1,713 | ) | $ | (1,713 | ) | $ | (1,254 | ) | $ | (6,712 | ) | $ | (2,112 | ) | $ | (2,527 | ) | $ | (1,280 | ) | |||||||
| Cash flows used in investing activities | $ | (19 | ) | $ | (19 | ) | $ | — | $ | (50 | ) | $ | — | $ | (50 | ) | $ | — | ||||||||||
| Cash flows provided by financing activities | $ | 16,695 | $ | 16,694 | $ | 1,248 | $ | 6,762 | $ | 2,112 | $ | 2,577 | $ | 1,280 | ||||||||||||||
| As of | As of | |||||||||||||||||||||||
| June 30, | December, | As of December 31, | ||||||||||||||||||||||
| 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | |||||||||||||||||||
| (Successor) | (Predecessor) | |||||||||||||||||||||||
| (Unaudited) | (Successor) | (Predecessor) | ||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||
Balance Sheets and Statements of Assets, Liabilities and Parent Company’s Net Deficit: | ||||||||||||||||||||||||
| Cash and cash equivalents | $ | 14,965 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Total current assets | $ | 14,980 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Equipment and furnishings, net | $ | 59 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||
| Total assets | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | ||||||||||||
| Total current liabilities | $ | 2,490 | $ | 202 | $ | — | $ | 325 | $ | 500 | $ | 968 | ||||||||||||
| Total stockholders’ equity | $ | 12,599 | $ | — | $ | 2 | $ | — | $ | — | $ | — | ||||||||||||
| Total liabilities and stockholders’ equity | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | ||||||||||||
| Parent company’s net deficit | $ | — | $ | (152 | ) | $ | — | $ | (268 | ) | $ | (450 | ) | $ | (968 | ) | ||||||||
B-7
Table of Contents
| • | Prior to the contribution of our RNAi assets from CytRx, our RNAi research and development activities were conducted by CytRx as part of its broader operations, rather than as an independent division or subsidiary, and were primarily conducted through sponsored research arrangements rather than through internal activities. CytRx also performed various corporate functions relating to our business, including governance functions such as compliance with the Sarbanes-Oxley Act of 2002 and external reporting. Our historical financial information reflect allocations of corporate expenses from CytRx for these and similar functions. We believe that these allocations are comparable to the expenses we would have incurred had we operated as a separate publicly traded company, although we will likely incur higher expenses as an independent company; | |
| • | Our working capital requirements and capital for our general corporate purposes have historically been provided by CytRx. Following the distribution, CytRx will not be obligated to provide us with funds to finance our working capital or other cash requirements. Without financing from CytRx, we will need to obtain additional financing through public offerings or private placements of debt or equity securities, strategic relationships or other arrangements; and | |
| • | Other significant changes may occur in our cost structure, management, financing and business operations as a result of our operating as a company separate from CytRx. |
B-8
Table of Contents
| • | to conduct research and development to successfully develop our RNAi technologies, | |
| • | to obtain regulatory approval for our products, | |
| • | to file and prosecute patent applications and to defend and assess patents to protect our technologies, | |
| • | to retain qualified employees, particularly in light of intense competition for qualified scientists, | |
| • | to manufacture products ourselves or through third parties, | |
| • | to market our products, either through building our own sales and distribution capabilities or relying on third parties, and | |
| • | to acquire new technologies, licenses or products. |
B-9
Table of Contents
B-10
Table of Contents
B-11
Table of Contents
| • | delays in filing initial drug applications, | |
| • | difficulty in securing centers to conduct trials, | |
| • | conditions imposed on us by the FDA or comparable foreign authorities regarding the scope or design of our clinical trials, | |
| • | problems in engaging IRBs to oversee trials or problems in obtaining or maintaining IRB approval of studies, | |
| • | difficulty in enrolling patients in conformity with required protocols or projected timelines, | |
| • | unexpected adverse reactions by patients in trials, | |
| • | difficulty in obtaining clinical supplies of the product, | |
| • | negative or inconclusive results from our clinical trials or the clinical trials of others for drug candidates similar to our own or inability to generate statistically significant data confirming the efficacy of the product being tested, | |
| • | changes in the FDA’s requirements for our testing during the course of that testing, | |
| • | modification of the drug during testing, | |
| • | reallocation of our limited financial and other resources to other clinical programs, and | |
| • | adverse results obtained by other companies developing RNAi drugs. |
B-12
Table of Contents
B-13
Table of Contents
| • | regulators or IRBs may not authorize us to commence a clinical trial or conduct a clinical trial at a prospective trial site, | |
| • | our pre-clinical tests or clinical trials may produce negative or inconclusive results, and we may decide, or regulator may require us, to conduct additional pre-clinical testing or clinical trials, or we may abandon projects that we previously expected to be promising, | |
| • | enrollment in our clinical trials may be slower than we anticipate or participants may drop out of our clinical trials at a higher rate than we anticipate, resulting in significant delays, | |
| • | our third party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, | |
| • | our drug candidates may have very different chemical and pharmacological properties in humans than in laboratory testing and it may interact with human biological systems in unforeseen, ineffective or harmful ways. | |
| • | we might have to suspend or terminate our clinical trials if the participants are being exposed to unacceptable health risks, | |
| • | IRBs or regulators, including the FDA, may require that we hold, suspend or terminate clinical research for various reasons, including noncompliance with regulatory requirements, | |
| • | the cost of our clinical trials may be greater than we anticipate, | |
| • | the supply or quality of our drug candidates or other necessary materials necessary to conduct our clinical trials may be insufficient or inadequate, and | |
| • | effects of our drug candidates may not be the desired effects or may include undesirable side effects or the drug candidates may have other unexpected characteristics. |
B-14
Table of Contents
| • | the timing of our receipt of any marketing approvals, the terms of any approvals and the countries in which approvals are obtained, | |
| • | the safety, efficacy and ease of administration of our product candidates, | |
| • | the advantages of our product candidates over those of our competitors, | |
| • | the willingness of patients to accept relatively new therapies, | |
| • | the success of our physician education programs, | |
| • | the availability of government and third-party payor reimbursement, | |
| • | the pricing of our products, particularly as compared to alternative treatments, and | |
| • | the availability of effective alternative treatments and the relative risksand/or benefits of the treatments. |
B-15
Table of Contents
B-16
Table of Contents
| • | they are “incidental” to a physician’s services, | |
| • | they are “reasonable and necessary” for the diagnosis or treatment of the illness or injury for which they are administered according to accepted standard of medical practice, | |
| • | they are not excluded as immunizations, and | |
| • | they have been approved by the FDA. |
B-17
Table of Contents
B-18
Table of Contents
| • | announcements of regulatory developments or technological innovations by us or our competitors, | |
| • | changes in our relationship with our licensors and other strategic partners, | |
| • | changes in our ownership or other relationships with CytRx, | |
| • | our quarterly operating results, | |
| • | developments in patent or other technology ownership rights, | |
| • | public concern regarding the safety of our products, | |
| • | government regulation of drug pricing, and | |
| • | general changes in the economy, the financial markets or the pharmaceutical or biotechnology industries. |
B-19
Table of Contents
| • | delaying, deferring or preventing a change in control of our company, | |
| • | impeding a merger, consolidation, takeover or other business combination involving our company, or | |
| • | discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of our company. |
| • | divide our board of directors into three classes, with members of each class to be elected for staggered three-year terms, | |
| • | limit the right of stockholders to remove directors, | |
| • | regulate how stockholders may present proposals or nominate directors for election at annual meetings of stockholders, and | |
| • | authorize our board of directors to issue preferred stock in one or more series, without stockholder approval. |
B-20
Table of Contents
B-21
Table of Contents
| • | development of our RNAi-based therapeutics may be delayed or may not proceed as planned, and we may not be able to complete development of any RNAi-based product, | |
| • | higher costs associated with being a separate, publicly trade company, | |
| • | the difficulty in evaluating our financial information due to the distribution, | |
| • | the inability to raise additional future financing and lack of financial and other resources to us as a separate company, | |
| • | our ability to control product development costs, | |
| • | we may not be able to attract and retain key employees, | |
| • | we may not be able to compete effectively, | |
| • | we may not be able enter into new strategic collaborations, | |
| • | changes in government regulation affecting our RNAi-based therapeutics could increase our development costs, | |
| • | our involvement in patent and other intellectual property litigation could be expensive and could divert management’s attention, | |
| • | the possibility that there will be no market acceptance for our products, and | |
| • | changes in third-party reimbursement policies could adversely affect potential future sales of any of our products that are approved for marketing. |
B-22
Table of Contents
B-23
Table of Contents
B-24
Table of Contents
| Period from | Period from | |||||||||||||||||||||||||||||||
| April 3, | January 1, | |||||||||||||||||||||||||||||||
| 2006 (Date of | 2003 (Date of | |||||||||||||||||||||||||||||||
| Incorporation) | Inception) | |||||||||||||||||||||||||||||||
| Through | Six Months Ended | Through | ||||||||||||||||||||||||||||||
| June 30, | June 30, | December 31, | Years Ended December 31, | |||||||||||||||||||||||||||||
| 2007 | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | 2003 | |||||||||||||||||||||||||
| (Successor) | (Successor) | (Predecessor) | (Predecessor) | (Predecessor) | ||||||||||||||||||||||||||||
| (Unaudited) | (Unaudited) | |||||||||||||||||||||||||||||||
| (All amounts are in thousands, except shares and earnings per share data) | ||||||||||||||||||||||||||||||||
Statements of Expenses Data: | �� | |||||||||||||||||||||||||||||||
| Revenue | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||||||||
| Expenses: | ||||||||||||||||||||||||||||||||
| Research and development | 4,594 | 4,594 | 907 | 8,541 | 1,772 | 2,080 | 2,814 | 1,877 | ||||||||||||||||||||||||
| General and administrative | 1,710 | 1,710 | 155 | 1,500 | 633 | 129 | 458 | 278 | ||||||||||||||||||||||||
| Operating loss | (6,304 | ) | (6,304 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | (2,155 | ) | ||||||||||||||||
| Interest income | 133 | 133 | — | — | — | — | — | — | ||||||||||||||||||||||||
| Loss before income taxes | (6,171 | ) | (6,171 | ) | (1,062 | ) | (10,041 | ) | (2,405 | ) | (2,209 | ) | (3,272 | ) | (2,155 | ) | ||||||||||||||||
| Income taxes | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||
| Net loss | $ | (6,171 | ) | $ | (6,171 | ) | $ | (1,062 | ) | (10,041 | ) | $ | (2,405 | ) | $ | (2,209 | ) | $ | (3,272 | ) | $ | (2,155 | ) | |||||||||
| Basic and diluted loss per share | $ | (0.64 | ) | $ | (0.64 | ) | NA | NA | NA | NA | NA | NA | ||||||||||||||||||||
| Weighted average shares, basic and diluted | 9,671,849 | 9,671,849 | NA | NA | NA | NA | NA | NA | ||||||||||||||||||||||||
| Balance Sheets and | ||||||||||||||||||||||||||||
| Statements of Assets, | As of | |||||||||||||||||||||||||||
| Liabilities and Parent | As of June 30, | December 31, | As of December 31, | |||||||||||||||||||||||||
Company’s Net Deficit: | 2007 | 2006 | 2006 | 2006 | 2005 | 2004 | 2003 | |||||||||||||||||||||
| (Successor) | (Predecessor) | (Successor) | (Predecessor) | |||||||||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 14,965 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Total current assets | $ | 14,980 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Working capital | $ | 12,490 | $ | (202 | ) | $ | 2 | $ | (318 | ) | $ | (500 | ) | $ | (968 | ) | $ | (89 | ) | |||||||||
| Total assets | $ | 15,089 | $ | 50 | $ | 2 | $ | 57 | $ | 50 | $ | — | $ | — | ||||||||||||||
| Due to parent | $ | (1,678 | ) | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||
| Total stockholders’ equity | $ | 12,599 | $ | — | $ | 2 | $ | — | $ | — | $ | — | $ | — | ||||||||||||||
| Parent company’s net deficit | $ | — | $ | (152 | ) | $ | — | $ | (268 | ) | $ | (450 | ) | $ | (968 | ) | $ | (89 | ) | |||||||||
B-25
Table of Contents
B-26
Table of Contents
| • | our ability to advance product candidates into pre-clinical research and clinical trials; | |
| • | the scope and rate of progress of our pre-clinical program and other research and development activities; | |
| • | the scope, rate of progress and cost of any clinical trials we commence; | |
| • | the cost of filing, prosecuting, defending and enforcing patent claims and other intellectual property rights; | |
| • | clinical trial results; | |
| • | the terms and timing of any collaborative, licensing and other arrangements that we may establish; | |
| • | the cost and timing of regulatory approvals; | |
| • | the cost of establishing clinical and commercial supplies of our product candidates and any products that we may develop; | |
| • | the cost and timing of establishing sales, marketing and distribution capabilities; | |
| • | the effect of competing technological and market developments; and | |
| • | the effect of government regulation and insurance industry efforts to control healthcare costs through reimbursement policy and other cost management strategies. |
B-27
Table of Contents
B-28
Table of Contents
B-29
Table of Contents
B-30
Table of Contents
B-31
Table of Contents
B-32
Table of Contents
| Six Months Ended | ||||||||||||||||||||
| June 30, | Years Ended December 31, | |||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | ||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| Research and development expense | $ | 1,271 | $ | 802 | $ | 1,298 | $ | 1,929 | $ | 1,700 | ||||||||||
| Research and development non-employee stock-based compensation expense | 1,012 | 105 | 212 | 151 | 1,006 | |||||||||||||||
| Fair value of common stock issued in exchange for licensing rights | 2,311 | — | 262 | — | 108 | |||||||||||||||
| Total research and development expense | $ | 4,594 | $ | 907 | $ | 1,772 | $ | 2,080 | $ | 2,814 | ||||||||||
B-33
Table of Contents
B-34
Table of Contents
| Six Months Ended June 30, | Years Ended December 31, | |||||||||||||||||||
| 2007 | 2006 | 2006 | 2005 | 2004 | ||||||||||||||||
| (Successor) | (Predecessor) | (Predecessor) | ||||||||||||||||||
| (Unaudited) | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| General and administrative expense | $ | 1,381 | $ | 155 | $ | 633 | $ | 129 | $ | 458 | ||||||||||
| Common stock and stock options issued for general and administrative expense | 329 | — | — | — | — | |||||||||||||||
| Total general and administrative expense | $ | 1,710 | $ | 155 | $ | 633 | $ | 129 | $ | 458 | ||||||||||
B-35
Table of Contents
B-36
Table of Contents
| Non-Cancelable | Cancelable | |||||||||||||||||||
| Operating | Employment | License | ||||||||||||||||||
Years Ending December 31, | Leases(1) | Agreements(2) | Subtotal | Agreements(3) | Total | |||||||||||||||
| 2007 | $ | 250 | $ | 951 | $ | 1,201 | $ | 2,892 | $ | 4,093 | ||||||||||
| 2008 | 9 | 810 | 819 | 433 | 1,252 | |||||||||||||||
| 2009 | — | 710 | 710 | 383 | 1,093 | |||||||||||||||
| 2010 | — | 460 | 460 | 333 | 793 | |||||||||||||||
| 2011 | — | 255 | 255 | 333 | 588 | |||||||||||||||
| Thereafter | — | 170 | 170 | 7,568 | 7,738 | |||||||||||||||
| Total | $ | 259 | $ | 3,356 | $ | 3,615 | $ | 11,942 | $ | 15,557 | ||||||||||
| (1) | Operating leases are primarily facility and equipment related obligations with third-party vendors. Facility lease expenses during the six months ended June 30, 2007 and 2006 were approximately $68,000 and $1,000, respectively. Facility lease expense during the years ended December 31, 2006, 2005 and 2004 was $2,000, $2,000 and $3,000 respectively. | |
| (2) | Employment agreement obligations include management contracts, as well as scientific advisory board member compensation agreements. Certain agreements provide for minimum salary levels, adjusted annually at the discretion of RXi’s Board of Directors, as well as for minimum annual bonuses. | |
| (3) | License agreements primarily relate to our obligations with UMMS associated with RNAi and, for future periods, represent minimum annual royalty payment obligations. Included in the 2007 license obligations was $2,310,000 that was paid to UMMS in the second quarter of 2007 in the form of shares of common stock of RXi. |
B-37
Table of Contents
B-38
Table of Contents
| • | Neurology. Initially, we are pursuing research in ALS (amyotrophic lateral sclerosis, commonly known as Lou Gehrig’s Disease). Some forms of ALS are caused by defects in a gene called SOD1. Early preclinical studies conducted by our advisors, Dr. Tariq Rana and Dr. Zuoshang Xu at UMMS, showed promising results in animals using an RNAi compound to selectively inhibit the SOD1 gene. We are refining and extending this work and, if successful, will move into formal preclinical development. We also intend to leverage our experience related to the delivery of RNAi therapeutics in the central nervous system to explore development of RNAi-based treatments for neurodegenerative diseases other than ALS, including Alzheimer’s Disease. | |
| • | Metabolic disease. One of our scientific co-founders and scientific advisory board members, Dr. Michael Czech, is a leading metabolic disease researcher. We have in-licensed intellectual property developed by Dr. Czech on genes that appear to be important regulators of metabolism. Studies conducted in Dr. Czech’s laboratory at UMMS and by others at Imperial College of London have demonstrated that inactivation of one of these genes, called RIP140, can cause fat cells to metabolize rather than store fat. Mice in these studies that did not express RIP140 remained lean and non-diabetic even when maintained on a high-fat diet. We are currently designing RNAi compounds targeting RIP140 as a potential treatment for obesity and obesity-related type 2 diabetes. We also continue to evaluate genes in Dr. Czech’s database for candidate targets. | |
| • | Oncology. We are initiating a program to develop RNAi drugs for use in oncology. This strategy is led by key RXi scientific advisors, Dr. Greg Hannon and Dr. Nicholas Dean, both of whom are leading researchers in targeting oncogene pathways. Additionally, our management team has expertise in developing programs targeting genes involved in cancer. Dr. Pamela Pavco, our Vice President for Pharmaceutical Development, previously managed the pre-clinical programs targeting genes involved with cancer while at Sirna Therapeutics, Inc. (recently acquired by Merck & Co.). | |
| • | Additional indications. There are many well-studied genes associated with numerous diseases that have been identified but have been difficult to target with normal medicinal chemistry. We believe RNAi technology may play an important role in targeting these genes and potentially treating these diseases. With the pioneering work being done in developing the RXi technology platform, we believe |
B-39
Table of Contents
| that we will discover many more drug candidates than can be advanced into clinical trials. For research on target genes in our portfolio that are not funded internally, we will seek to identify and work with partners in the discovery and development process to build our development pipeline. |
| • | World-class scientific advisors with extraordinary experience in RNAi, including Dr. Craig Mello, recipient of the 2006 Nobel Prize in Medicine for co-discovering RNAi. | |
| • | Strong early intellectual property position covering |
| • | Key therapeutic targets, | |
| • | Proprietary, potent nanotransporter delivery of active RNAi to tissues, and | |
| • | Novel approaches to RNAi chemistry. |
| • | Initial focus on significant unmet medical needs, including |
| • | Neurological disorders, | |
| • | Metabolic disease, and | |
| • | Oncology. |
| • | Management that is experienced in commercializing products, including our President and Chief Executive Officer, Tod Woolf, Ph.D. | |
| • | Potentially rapid route to the clinic for ALS therapeutics due to the high mortality rate. |
B-40
Table of Contents

| • | high specificity for targeted genes; | |
| • | high potency (low doses); | |
| • | potential interference with the expression of any gene; and | |
| • | accelerated development of lead compounds. |
B-41
Table of Contents

| • | up to x100 more active than conventional siRNA (depending on the target site), | |
| • | nuclease resistant, | |
| • | readily manufactured, | |
| • | comprised of elements from RXi’s IP portfolio, and | |
| • | potentially more specific for the target gene. |
B-42
Table of Contents

| • | inhibition of liver target with 1 mg RNAi compound per kg of body weight; | |
| • | no immune stimulation detected; | |
| • | defined particle size; and | |
| • | readily formulated. |
B-43
Table of Contents
B-44
Table of Contents
| • | We intend to finance the initial development of a limited number of RNAi drug candidates with our own capital resources and any financial resources that we may obtain from capital markets and partners. Our key therapeutic areas of interest are neurology, metabolic disease and oncology. We intend to develop drugs in these areas internally to establish significant value, at which point we may seek to partner them. |
B-45
Table of Contents
| • | We intend to seek partnerships with large pharmaceutical and biotechnology companies to leverage our intellectual property and expand our development pipeline. Such partnerships may include traditionally structured drug development and commercialization licenses, discovery and development collaborations, research and technology collaborations, and intellectual property licenses. | |
| • | We intend to maintain and continue to develop and enhance our RNAi technology platform by expanding our intellectual property position in RNAi compound chemistry, delivery and target sequences. To date, we have in-licensed RNAi technologies from various institutions and companies, including UMMS, Imperial College of London, Cold Spring Harbor Laboratories and TriLink Biotechnologies. We intend to continue to enhance our technology platform through in-licensing in combination with internal and collaborative research and development programs. | |
| • | We intend to develop future RNAi technology improvements and believe we are well-positioned to do so. Our management and advisors have developed much of the core technology in the field of oligonucleotide therapeutics, and more specifically RNAi. For example, our Scientific Advisory Board member, Dr. Nassim Usman, developed the standard synthesis chemistry used to manufacture RNAi compounds throughout the world while a fellow at MIT. Our CEO, Dr. Tod Woolf, co-developed STEALTH RNAi, which is one of the most commonly employed second generation RNAi chemistries, and our Vice President of Pharmaceutical Development, Dr. Pamela Pavco, developed the first modified RNAi tested in humans while she was at Sirna Therapeutics. Our advisors and scientists routinely meet to discuss novel approaches and improvements in our RNAi technology platforms to enhance our intellectual property portfolio. | |
| • | We may also seek to collaborate with government and charitable institutions through grants and funded research for our development programs. |
| • | therapeutic targets, | |
| • | chemistry and configurations of RNAi compounds, and | |
| • | delivery technology for RNAi. |
B-46
Table of Contents
| • | Dr. Rana’s inventions which provide fundamental rules for designing chemically modified RNAi sequences suitable forin vivo gene silencing, to which we have co-exclusive licenses in the therapeutic areas of type 2 diabetes, obesity, and ALS by targeting mutant SOD1, and CMV retinitis; | |
| • | Dr. Tuschl’s invention regarding RNAi therapeutics using double-stranded RNAs in the areas of type 2 diabetes, obesity, and ALS by targeting mutant SOD1, and CMV retinitis; | |
| • | Drs. Mello and Zamore’s invention regardingin vivoproduction of siRNA; and | |
| • | methods of triggering RNAiin vivo, based on particular RNA structural characteristics. |
| • | methods and compositions, including use of nanotransporters, for RNAi compound delivery which enable therapeutic gene silencing in cells and animals; and | |
| • | inhibition of gene expression in fat cells using RNAi. |
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Drug discovery for diabetes and obesity | Michael P. Czech Silvia Corvera | 09/27/1993 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | US Pat. 5,989,893 Pending elsewhere | ||||
| RNAi in general | Craig C. Mello Andrew Fire Stephen Kostas Mary Montgomery Lisa Timmons SiQun Zu Hiroaki Tabara Samuel E. Driver | 12/23/1997 | Non-exclusive license to fundamental RNAi from Carnegie Institute of Washington. | US Pat. 6,506,559 Pending elsewhere |
B-47
Table of Contents
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Drug discovery using genomic databases for diabetes and obesity | Michael P. Czech Andrew D. Cherniack Adelson L. Guilherme | 10/20/2000 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending | ||||
| dsRNA in general | Thomas Tuschl, Philip A. Sharp Phillip D. Zamore David P. Bartel | 12/01/2000 | Non-exclusive license for RNAi use to inhibit (1) HCMV immediate early gene in retinitis (2) mutant SOD1 gene in ALS, and (3) gene targets in type II diabetes & obesity from UMMS, a co-owner of the patent. | Pending | ||||
| Engineered precursor to siRNA for in vivo production of siRNA | Phillip D. Zamore Gyorgy Hutvagner Juanita McLachlan Craig C. Mello Alla Grishok | 07/12/2001 | Exclusive license for RNAi use to inhibit (1) HCMV immediate early gene in retinitis (2) mutant SOD1 gene in ALS, and (3) gene targets in type II diabetes & obesity. | Pending | ||||
| In vivo gene silencing by chemically modified and stable siRNA | Tariq M. Rana Ya-Lin Chiu | 09/25/2002 | Exclusive worldwide license to the treatment of CMV, ALS, diabetes & obesity for therapeutics, prophylactics or diagnostics. | Pending | ||||
| Allele specific inhibition by siRNA, especially SOD inhibition for the treatment of ALS | Zuoshang Xu Phillip D. Zamore | 11/04/2002 | Exclusive license on inhibition of SOD1 expression for the treatment of ALS including therapeutics, prophylactics and diagnostics. | Pending | ||||
| Delivery of siRNA using peptide conjugate | Tariq M. Rana | 11/26/2002 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans, including for ALS, diabetes and obesity. | Pending | ||||
| Allele specific inhibition by chemically modified siRNA | Tariq M. Rana | 11/26/2002 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending |
B-48
Table of Contents
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Delivery of siRNA into adipocytes for treatment of diabetes and obesity ; | Michael P. Czech Qiong L. Zhou Zhen Y. Jiang | 12/11/2002 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending | ||||
| Screening for modulators of fat storage | Malcolm Parker Roger White Goran Leonardsson | 02/03/2003 | Exclusive license for screening methods for identifying compounds that are useful as modulators of fat storage from Imperial College, UK. | Pending | ||||
| Inhibition of CMV using siRNA | Timothy F. Kowalik | 02/05/2003 | Exclusive license to develop RNAi therapeutics for cytomegalovirus related diseases (HCMV- immediate early gene). (Note that original license only for CMV retinitis; amended 01/04 to include all CMV) | Pending | ||||
| Modulation of insulin sensitivity through stress proteins | Silvia Corvera | 06/19/2003 | Exclusive license for products in the field of drug discovery in type 2 diabetes and/or obesity. | Pending | ||||
| Chemically modified siRNA and their uses | Tariq M. Rana Ya-Lin Chiu | 08/05/2003 | Co-Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications of RNAi for inhibition of human CMV immediate early gene expression, mutant SOD1 (ALS treatment), genes implicated in diabetes and obesity. | Pending | ||||
| Increase of insulin stimulated glucose regulation by a small molecule inhibitor of RIP140 | Michael P. Czech Aimee Powelka Adilson L. Guilherme Andrew D. Cherniack | 03/05/2004 | Exclusive license for drug discovery in type 2 diabetes and/or obesity with therapeutic, prophylactic, or diagnostic applications and products for treatment, prevention or diagnosis of type 2 diabetes and/or obesity. | Pending |
B-49
Table of Contents
| Priority | ||||||||
Subject Field | Inventor(s) | Date | License* | Status | ||||
| Efficient delivery of siRNA into cells and animals | Tariq M. Rana | 08/11/2005 | Nonexclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Delivery of chemically modified siRNA using nanotransporters | Tariq M. Rana (Zuoshang Xu) | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Delivery of chemically modified siRNA | Tariq M. Rana | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Gene silencing of cholesterol biosynthesis and other metabolic genes by chemically modified siRNA | Tariq M. Rana | 01/26/2006 | Exclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Pending | ||||
| Microwave assisted nucleic acid delivery system | Tariq M. Rana | Not yet filed | Nonexclusive license for therapeutic, prophylactic or diagnostic healthcare applications in humans including for ALS, diabetes and obesity. | Invention disclosure | ||||
| Methods and compositions for RNA interference in suspended cells or in whole body | Scott Hammond Gregory Hannon David Beach Amy Caudy Emily Bernstein | 03/16/2001 | Non-exclusive license from Cold Spring Harbor Laboratory for use of short hairpin RNA for drug discovery and development. | Pending |
| * | Unless otherwise noted, the licenses are from UMMS. |
B-50
Table of Contents
B-51
Table of Contents
B-52
Table of Contents
B-53
Table of Contents
B-54
Table of Contents
B-55
Table of Contents
Name | Position | Age | ||||
| Sanford J. Hillsberg(1)(2) | Director and Chairman of the Board | 59 | ||||
| Tod Woolf, Ph.D. | President and Chief Executive Officer, Director | 43 | ||||
| Mark J. Ahn, Ph.D.(1)(2)(3) | Director | 44 | ||||
| Stephen S. Galliker(1)(2)(3) | Director | 60 | ||||
| Steven A. Kriegsman(3) | Director | 65 | ||||
| Stephen J. DiPalma | Chief Financial Officer and Secretary | 48 | ||||
| Pamela Pavco, Ph.D. | Vice President of Pharmaceutical Development | 51 | ||||
| Dmitry Samarsky, Ph.D. | Vice President of Technology and Business Development | 40 | ||||
| James Warren | Former Chief Financial Officer and Secretary | 62 | ||||
| (1) | Member of our Nominating and Governance Committee | |
| (2) | Member of our Audit Committee | |
| (3) | Member of our Compensation Committee |
Name | Professional Affiliation | |||
| Craig C. Mello, Ph.D (Chairman) | • | Professor and Blaise University Chair in Molecular Medicine at the University of Massachusetts Medical School | ||
| • | Investigator, Howard Hughes Medical Institute | |||
| • | Member of the National Academy of Sciences | |||
| Tariq Rana, Ph.D | • | Professor of Biochemistry & Molecular Pharmacology and Founding Director of the Program in Chemical Biology at the University of Massachusetts Medical School | ||
| Gregory Hannon, Ph.D | • | Investigator, Howard Hughes Medical Institute | ||
| Michael Czech, Ph.D | • | Professor and Chair in Molecular Medicine at the University of Massachusetts Medical School | ||
| Nicholas Dean, Ph.D | • | Founder and Chief Scientific Officer of Excaliard Pharmaceuticals | ||
| Nassim Usman, Ph.D | • | Chief Executive Officer and Board Member of Catalyst Biosciences | ||
B-56
Table of Contents
B-57
Table of Contents
B-58
Table of Contents
B-59
Table of Contents
B-60
Table of Contents
B-61
Table of Contents
| • | an annual retainer fee of $10,000, payable quarterly, | |
| • | an annual retainer fee of $12,000 for the chairperson of each committee of our board of directors other than the audit committee, payable quarterly, | |
| • | an annual retainer fee of $20,000 for the chairperson of the audit committee of our board of directors, payable quarterly, | |
| • | an annual retainer fee of $32,000 for the Chairman of the board of directors, payable quarterly, | |
| • | a fee of $2,000 per board meeting attended by the director ($1,000 if attendance is telephonic), and | |
| • | a fee of $1,500 per committee meeting attended by the director ($750 if attendance is telephonic). |
B-62
Table of Contents
B-63
Table of Contents
| • | base salary, | |
| • | performance-based cash compensation, and | |
| • | long-term equity incentive compensation. |
| • | the negotiated terms of each executive employment agreement, | |
| • | internal review of the executive’s compensation, both individually and relative to other executive officers, and | |
| • | individual performance of the executive. |
B-64
Table of Contents
| • | establish the link between the creation of stockholder value and long-term executive incentive compensation, | |
| • | provide an opportunity for increased equity ownership by executives, | |
| • | function as a retention tool because of the vesting features included in all options granted by the Committee, and | |
| • | maintain competitive levels of total compensation. |
B-65
Table of Contents
B-66
Table of Contents
| Value of | ||||||||||||||||||||
| Accelerated | ||||||||||||||||||||
| Value of | Option | |||||||||||||||||||
| Accelerated | Vesting | |||||||||||||||||||
| Salary | Benefit | Option | Upon a | |||||||||||||||||
| Name and Principal | Continuation | Continuation | Vesting | Total | Change in | |||||||||||||||
Position | (Termination) | (Termination) | (Termination) | (Termination) | Control | |||||||||||||||
| Tod Woolf | $ | 250,000 | $ | 14,500 | $ | 449,000 | $ | 714,400 | $ | 1,087,100 | ||||||||||
| President and Chief Executive Officer | ||||||||||||||||||||
| Stephen J. DiPalma | $ | 110,000 | $ | 7,300 | — | $ | 117,300 | $ | 406,400 | |||||||||||
| Chief Financial Officer | ||||||||||||||||||||
| Pamela Pavco(1) | $ | 198,000 | $ | 2,100 | $ | 157,200 | $ | 357,300 | $ | 511,000 | ||||||||||
| Vice President of Research and Development | ||||||||||||||||||||
| Dmitry Samarsky(1) | $ | 85,000 | $ | 1,800 | $ | 57,200 | $ | 144,000 | $ | 400,400 | ||||||||||
| Vice President of Technology and Business Development | ||||||||||||||||||||
| (1) | Each of Drs. Pavco and Samarsky are entitled to specified benefits in the event his or her employment is terminated without cause or he or she terminates his or her employment for good reason following a change in control, as further described below. |
B-67
Table of Contents
B-68
Table of Contents
B-69
Table of Contents
B-70
Table of Contents
B-71
Table of Contents
B-72
Table of Contents
B-73
Table of Contents
B-74
Table of Contents
B-75
Table of Contents
B-76
Table of Contents
B-77
Table of Contents
| • | any person known by us to be the beneficial owner of 5% or more of our common stock, including any “group” as that term is defined in the Exchange Act, | |
| • | each director, our named executive officers identified in the “Management-Executive Compensation” section above, and | |
| • | all of our directors and executive officers as a group. |
| After the Distribution | ||||||||||||||||
| and Award Transactions | ||||||||||||||||
| Described in this | ||||||||||||||||
| Before the Distribution | Prospectus | |||||||||||||||
| Number | Number | |||||||||||||||
| of | of | |||||||||||||||
Name | Shares | Percent | Shares | Percent | ||||||||||||
5% Stockholders: | ||||||||||||||||
| CytRx Corporation(1) | 10,823,199 | 85.8 | % | 6,212,861 | 49.3 | % | ||||||||||
Executive Officers and Directors: | ||||||||||||||||
| Tod Woolf, Ph.D.(2) | 52,832 | # | 52,832 | # | ||||||||||||
| Stephen J. DiPalma(3) | 6,250 | # | 6,250 | # | ||||||||||||
| James Warren(4) | 62,743 | # | 62,743 | # | ||||||||||||
| Pamela Pavco, Ph.D.(5) | 18,163 | # | 18,163 | # | ||||||||||||
| Dmitry Samarsky, Ph.D.(6) | 6,597 | # | 6,597 | # | ||||||||||||
| Mark J. Ahn, Ph.D.(7) | 47,500 | # | 47,500 | # | ||||||||||||
| Stephen S. Galliker(7) | 47,500 | # | 47,500 | # | ||||||||||||
| Sanford J. Hillsberg(7)(8) | 47,500 | # | 47,500 | # | ||||||||||||
| Steven A. Kriegsman(9) | 37,500 | # | 278,268 | 2.2 | % | |||||||||||
| All executive officers and directors as a group (8 persons)(10) | 296,585 | 2.50 | % | 567,353 | 4.4 | % | ||||||||||
| # | Represents less than 1% of the outstanding shares of our common stock. |
B-78
Table of Contents
| (1) | The address for CytRx is 11726 San Vicente Boulevard, Suite 650, Los Angeles, California 90049. | |
| (2) | Consists of 52,832 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (3) | Consists of 6,250 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (4) | Consists of 62,743 shares of common stock underlying stock options currently exercisable until November 30, 2007. | |
| (5) | Consists of 18,163 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (6) | Includes 6,597 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (7) | Includes 37,500 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. | |
| (8) | The shares shown do not include shares owned by Troy & Gould Professional Corporation. | |
| (9) | Includes 37,500 shares of common stock underlying stock options and exercisable within 60 days of October 1, 2007 and, following the distribution and award transactions described in this prospectus, approximately 240,768 shares that Mr. Kriegsman will receive as a result of distribution under this prospectus and the award under the award prospectus. Please see “The Distribution” for a further explanation of the allocation of shares in the distribution. Mr. Kriegsman is the CEO and a director of CytRx, but acting alone, he has neither voting nor investment power with respect to the shares beneficially owned by CytRx. As a result, Mr. Kriegsman disclaims beneficial ownership of such shares. | |
| (10) | Includes 293,282 shares of common stock underlying stock options exercisable within 60 days of October 1, 2007. |
B-79
Table of Contents
| Beneficial Ownership | ||||||||||||||||||||||||||||
| Beneficial Ownership | After Distribution and | Number of | Beneficial Ownership | |||||||||||||||||||||||||
| Before Distribution and | Award Transactions and Before this Offering | Shares | After this Offering | |||||||||||||||||||||||||
| Award Transactions | Number of | Being | Number of | |||||||||||||||||||||||||
Name | Number of Shares | Percent | Shares | Percent | Offered | Shares | Percent | |||||||||||||||||||||
| CytRx Corporation | 10,823,199 | 85.77 | % | 6,212,861 | 49.3 | % | 6,212,861 | 0 | -0- | |||||||||||||||||||
B-80
Table of Contents
| • | ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers, | |
| • | block trades in which the broker-dealer will attempt to sell the shares as agent but may position and resell a portion of the block as principal to facilitate the transaction, | |
| • | purchases by a broker-dealer as principal and resale by the broker-dealer for its account, | |
| • | an exchange distribution in accordance with the rules of the applicable exchange, | |
| • | privately negotiated transactions, | |
| • | settlement of short sales entered into after the effective date of the registration statement of which this prospectus is a part, | |
| • | broker-dealers may agree with CytRx to sell a specified number of such shares at a stipulated price per share, | |
| • | through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise, | |
| • | any combination of any of the foregoing methods of sale, and | |
| • | any other method permitted by applicable law. |
B-81
Table of Contents
B-82
Table of Contents
B-83
Table of Contents
B-84
Table of Contents
B-85
Table of Contents
| • | prior to such date, the board of directors of the corporation approved either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder, | |
| • | upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, other than statutorily excluded shares, or | |
| • | on or subsequent to such date, the business combination is approved by the board of directors of the corporation and authorized at an annual or special meeting of the stockholders by the affirmative vote of at least 662/3% of the outstanding voting stock which is not owned by the interested stockholder. |
| • | any person that is the owner of 15% or more of the outstanding voting stock of the corporation, or is an affiliate or associate of the corporation and was the owner of 15% or more of the outstanding voting stock of the corporation at any time within three years immediately prior to the date of determination, and | |
| • | the affiliates and associates of any such person. |
B-86
Table of Contents
B-87
Table of Contents
Investor Relations
One Innovation Drive
Worcester, Massachusetts 01605
Telephone:(508) 767-3861
B-88
Table of Contents
Table of Contents
| Item 13. | Other expenses of issuance and distribution |
| Total | ||||
| SEC registration fee | $ | 338.92 | ||
| NASDAQ Capital Market listing fee | $ | 5,000.00 | ||
| Printing expenses | $ | |||
| Legal fees and expenses | $ | |||
| Accounting fees and expenses | $ | |||
| Transfer agent and registrar fees and expenses | $ | |||
| Miscellaneous fees and expenses | $ | |||
| Total | $ | |||
| Item 14. | Indemnification of officers and directors |
| • | any breach of the director’s duty of loyalty to the corporation or its stockholders, | |
| • | acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law, | |
| • | payments of unlawful dividends or unlawful stock repurchases or redemptions, or | |
| • | any transaction from which the director derived an improper personal benefit. |
II-1
Table of Contents
| Item 15. | Recent sales of unregistered securities |
II-2
Table of Contents
| Item 16. | Exhibits and financial statement schedules |
| (a) | Exhibits |
| Exhibit | ||||
Number | Description | |||
| 2 | .1 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated January 8, 2007(2) | ||
| 2 | .2 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated April 30, 2007(2) | ||
| 2 | .3 | Reimbursement Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated January 8, 2007(2) | ||
| 3 | .1 | Form of Amended and Restated Certificate of Incorporation of RXi Pharmaceuticals Corporation(2) | ||
| 3 | .2 | Form of Amended and Restated By-laws of RXi Pharmaceuticals Corporation(2) | ||
| 4 | .1 | Specimen common stock certificate(1) | ||
| 4 | .2 | Stockholders Agreement between CytRx Corporation, RXi Pharmaceuticals Corporation, the other Stockholders and the Scientific Advisory Board Members, dated February 23, 2007(2) | ||
| 4 | .3 | Exhibit A to Contribution Agreement — Registration Rights Terms between CytRx Corporation and RXi Pharmaceuticals Corporation, dated April 30, 2007(2) | ||
| 4 | .4 | Annex I to form of Subscription Agreement — Registration Rights Terms between RXi Pharmaceuticals Corporation and Stephen Galliker, Mark Ahn and Sanford Hillsberg(2) | ||
II-3
Table of Contents
| Exhibit | ||||
Number | Description | |||
| 5 | .1 | Opinion of Ropes & Gray LLP, counsel to the Registrant, with respect to the legality of securities being registered(1) | ||
| 10 | .1 | Voting Agreement between CytRx Corporation and the University of Massachusetts Medical School, dated January 10, 2007(2) | ||
| 10 | .2 | License Agreement between Cold Spring Harbor Laboratory and RXi Pharmaceuticals Corporation, dated March 15, 2007(1) | ||
| 10 | .3 | Invention Disclosure Agreement between the University of Massachusetts Medical School and RXi Pharmaceuticals Corporation, dated January 8, 2007(1) | ||
| 10 | .4 | Exclusive License Agreement (No.: UMMC06-21-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .5 | Exclusive License Agreement (No.: UMMC03-68-02) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .6 | Exclusive License Agreement (No.: UMMC03-75-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .7 | Non-Exclusive License Agreement (No.: UMMC06-08-03) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .8 | Non-Exclusive License Agreement, between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number01-36, dated April 15, 2003, as amended February 1, 2004(1) | ||
| 10 | .9 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number02-01, dated April 15, 2003, as amended September 10, 2004(1) | ||
| 10 | .10 | Amended and Restated Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-05,00-37,01-31,03-134,93-09 and02-38, dated September 15, 2003, as amended September 17, 2003 and February 1, 2004(1) | ||
| 10 | .11 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-17, dated April 15, 2003, as amended January 7, 2004 and February 1, 2004(1) | ||
| 10 | .12 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-60, dated April 15, 2003 as amended February 1, 2004(1) | ||
| 10 | .13 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-33, and all amendments thereto, dated May 18, 2006(1) | ||
| 10 | .14 | License Agreement between CytRx Corporation, Imperial College Innovations Limited and Imperial College of Science and Technology, dated May 19, 2004(1) | ||
| 10 | .15 | Employment Agreement between RXi Pharmaceuticals Corporation and Tod Woolf, Ph.D., dated February 22, 2007*(2) | ||
| 10 | .16 | Employment Agreement between RXi Pharmaceuticals Corporation and Pamela Pavco, dated March 7, 2007*(2) | ||
| 10 | .17 | Employment Agreement between RXi Pharmaceuticals Corporation and Dmitry Samarsky, dated June 25, 2007*(2) | ||
| 10 | .18 | Employment Agreement between RXi Pharmaceuticals Corporation and Stephen J. DiPalma, dated August 28, 2007*(2) | ||
| 10 | .19 | RXi Pharmaceuticals Corporation’s 2007 Incentive Plan*(2) | ||
| 10 | .20 | Form of Incentive Stock Option*(2) | ||
| 10 | .21 | Form of Non-qualified Stock Option*(2) | ||
II-4
Table of Contents
| Exhibit | ||||
Number | Description | |||
| 10 | .22 | Lease between RXi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts, 01605, dated September 25, 2007(1) | ||
| 23 | .1 | Consent of BDO Seidman, LLP, Independent Registered Public Accounting Firm(2) | ||
| 23 | .2 | Consent of Ropes & Gray LLP (included in Exhibit 5.1) | ||
| 24 | .1 | Power of Attorney (included onpage II-6) | ||
| (1) | To be filed by amendment | |
| (2) | Filed herewith. | |
| * | Indicates a management contract or compensatory plan or arrangement. | |
| + | Confidential treatment has been requested or granted for certain portions which have been blanked out in the copy of the exhibit filed with the Securities and Exchange Commission. The omitted information has been filed separately with the Securities and Exchange Commission. |
| (b) | Financial Statement Schedules |
| Item 17. | Undertakings |
II-5
Table of Contents
| By: | /s/ Tod Woolf |
President and Chief Executive Officer
Signatures | Title | Date | ||||
/s/ Tod Woolf Tod Woolf, Ph.D. | President, Chief Executive Officer and Director (Principal Executive Officer) | October 30, 2007 | ||||
/s/ Stephen J. DiPalma Stephen J. DiPalma | Chief Financial Officer (Principal Financial and Accounting Officer) | October 30, 2007 | ||||
/s/ Sanford J. Hillsberg Sanford J. Hillsberg | Director | October 30, 2007 | ||||
/s/ Mark J. Ahn Mark J. Ahn | Director | October 30, 2007 | ||||
/s/ Stephen S. Galliker Stephen S. Galliker | Director | October 30, 2007 | ||||
/s/ Steven A. Kriegsman Steven A. Kriegsman | Director | October 30, 2007 | ||||
II-6
Table of Contents
| Exhibit | ||||
Number | Description | |||
| 2 | .1 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated January 8, 2007(2) | ||
| 2 | .2 | Contribution Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated April 30, 2007(2) | ||
| 2 | .3 | Reimbursement Agreement between CytRx Corporation and RXi Pharmaceuticals Corporation, dated January 8, 2007(2) | ||
| 3 | .1 | Form of Amended and Restated Certificate of Incorporation of RXi Pharmaceuticals Corporation(2) | ||
| 3 | .2 | Form of Amended and Restated By-laws of RXi Pharmaceuticals Corporation(2) | ||
| 4 | .1 | Specimen common stock certificate(1) | ||
| 4 | .2 | Stockholders Agreement between CytRx Corporation, RXi Pharmaceuticals Corporation, the other Stockholders and the Scientific Advisory Board Members, dated February 23, 2007(2) | ||
| 4 | .3 | Exhibit A to Contribution Agreement — Registration Rights Terms between CytRx Corporation and RXi Pharmaceuticals Corporation, dated April 30, 2007(2) | ||
| 4 | .4 | Annex I to form of Subscription Agreement — Registration Rights Terms between RXi Pharmaceuticals Corporation and Stephen Galliker, Mark Ahn and Sanford Hillsberg(2) | ||
| 5 | .1 | Opinion of Ropes & Gray LLP, counsel to the Registrant, with respect to the legality of securities being registered(1) | ||
| 10 | .1 | Voting Agreement between CytRx Corporation and the University of Massachusetts Medical School, dated January 10, 2007(2) | ||
| 10 | .2 | License Agreement between Cold Spring Harbor Laboratory and RXi Pharmaceuticals Corporation, dated March 15, 2007(1) | ||
| 10 | .3 | Invention Disclosure Agreement between the University of Massachusetts Medical School and RXi Pharmaceuticals Corporation, dated January 8, 2007(1) | ||
| 10 | .4 | Exclusive License Agreement (No.: UMMC06-21-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .5 | Exclusive License Agreement (No.: UMMC03-68-02) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .6 | Exclusive License Agreement (No.: UMMC03-75-01) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .7 | Non-Exclusive License Agreement (No.: UMMC06-08-03) between the University of Massachusetts and RXi Pharmaceuticals Corporation, dated January 10, 2007(1) | ||
| 10 | .8 | Non-Exclusive License Agreement, between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number01-36, dated April 15, 2003, as amended February 1, 2004(1) | ||
| 10 | .9 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number02-01, dated April 15, 2003, as amended September 10, 2004(1) | ||
| 10 | .10 | Amended and Restated Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-05,00-37,01-31,03-134,93-09 and02-38, dated September 15, 2003, as amended September 17, 2003 and February 1, 2004(1) | ||
| 10 | .11 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-17, dated April 15, 2003, as amended January 7, 2004 and February 1, 2004(1) | ||
| 10 | .12 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-60, dated April 15, 2003 as amended February 1, 2004(1) | ||
| 10 | .13 | Exclusive License Agreement between CytRx Corporation and the University of Massachusetts Medical School related to UMMS disclosure number03-33, and all amendments thereto, dated May 18, 2006(1) | ||
Table of Contents
| Exhibit | ||||
Number | Description | |||
| 10 | .14 | License Agreement between CytRx Corporation, Imperial College Innovations Limited and Imperial College of Science and Technology, dated May 20, 2004(1) | ||
| 10 | .15 | Employment Agreement between RXi Pharmaceuticals Corporation and Tod Woolf, Ph.D., dated February 22, 2007*(2) | ||
| 10 | .16 | Employment Agreement between RXi Pharmaceuticals Corporation and Pamela Pavco, dated March 7, 2007*(2) | ||
| 10 | .17 | Employment Agreement between RXi Pharmaceuticals Corporation and Dmitry Samarsky, dated June 25, 2007*(2) | ||
| 10 | .18 | Employment Agreement between RXi Pharmaceuticals Corporation and Stephen J. DiPalma, dated August 28, 2007*(2) | ||
| 10 | .19 | RXi Pharmaceuticals Corporation’s 2007 Incentive Plan*(2) | ||
| 10 | .20 | Form of Incentive Stock Option*(2) | ||
| 10 | .21 | Form of Non-qualified Stock Option*(2) | ||
| 10 | .22 | Lease between RXi Pharmaceuticals Corporation and Newgate Properties, LLC for One Gateway Place, Worcester, Massachusetts, 01605, dated September 25, 2007(1) | ||
| 23 | .1 | Consent of BDO Seidman, LLP, Independent Registered Public Accounting Firm | ||
| 23 | .2 | Consent of Ropes & Gray LLP (included in Exhibit 5.1) | ||
| 24 | .1 | Power of Attorney (included onpage II-6) | ||
| (1) | To be filed by amendment | |
| (2) | Filed herewith. | |
| * | Indicates a management contract or compensatory plan or arrangement. | |
| + | Confidential treatment has been requested or granted for certain portions which have been blanked out in the copy of the exhibit filed with the Securities and Exchange Commission. The omitted information has been filed separately with the Securities and Exchange Commission. |