Exhibit 99.3
| REVA’s Bioresorbable Pipeline Fantom™ Sirolimus-Eluting Bioresorbable Scaffold Dr. Alexandre Abizaid Instituto Dante Pazzanese de Cardiologia Sao Paulo, Brazil |
| Potential conflicts of interest Speaker's name: Dr. Alexandre Abizaid I do not have any potential conflict of interest I have the following potential conflicts of interest to report: Institutional grant/research support: REVA Medical, Inc. Consultant: REVA Medical, Inc. |
| REVA’s Technology History 1998: Company founded around novel steel slide & lock design For minimal recoil 2002: Initiated development of tyrosine-derived polycarbonate polymer Rutgers University Foundation for REVA’s distinctive attributes for polymer scaffolds 2003: 100% effort on bioresorbable scaffolds Evaluated in > 1,000 animals and advanced bench tests Evaluated in > 150 patients beginning in 2007 Proved acute performance, safe resorption, visibility, drug delivery 2014: Introduction of Fantom™ |
| Transition to Fantom ReZolve elegantly designed...but Design required relatively thicker strut segments Up to 230 microns in locking sections A scaffold with -0- recoil is not necessary REVA polymer advancements led to increased design options Physician’s demands for ease of use are increasing More deliverable and lower profile devices Devices that require standard inflation techniques Devices that can be visualized more readily |
| Foundation of the Fantom Platform Fantom™ Sirolimus-Eluting Bioresorbable Scaffold |
| REVA polymer family High-performance biomaterials Based on phenyl ring structure Competitive Polymers Polylactide-based Based on Alkane structure REVA Polymers vs. Competitive Polymers Key Structural Polymer Building Blocks Phenyl ring structure provides high strength, radiopaque materials without compromising structural properties |
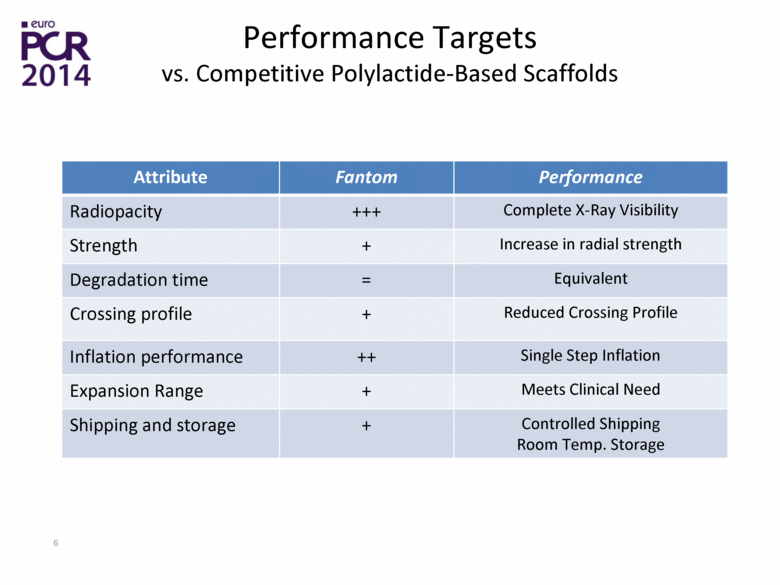
| Performance Targets vs. Competitive Polylactide-Based Scaffolds Attribute Fantom Performance Radiopacity +++ Complete X-Ray Visibility Strength + Increase in radial strength Degradation time = Equivalent Crossing profile + Reduced Crossing Profile Inflation performance ++ Single Step Inflation Expansion Range + Meets Clinical Need Shipping and storage + Controlled Shipping Room Temp. Storage |
| Fantom Bioresorbable Scaffold Radiopacity Fantom’s complete (x-ray) visibility increases confidence during the procedure Precise scaffold placement Complete lesion coverage No geographic miss Confirmation of apposition to vessel wall Reduces need for IVUS & OCT catheter use Saving costs to hospital No permanent metal markers left behind Competitive Scaffolds Visualized Only by Small Permanent Markers (Scaffold Itself is Invisible) Absorb Fantom Xience |
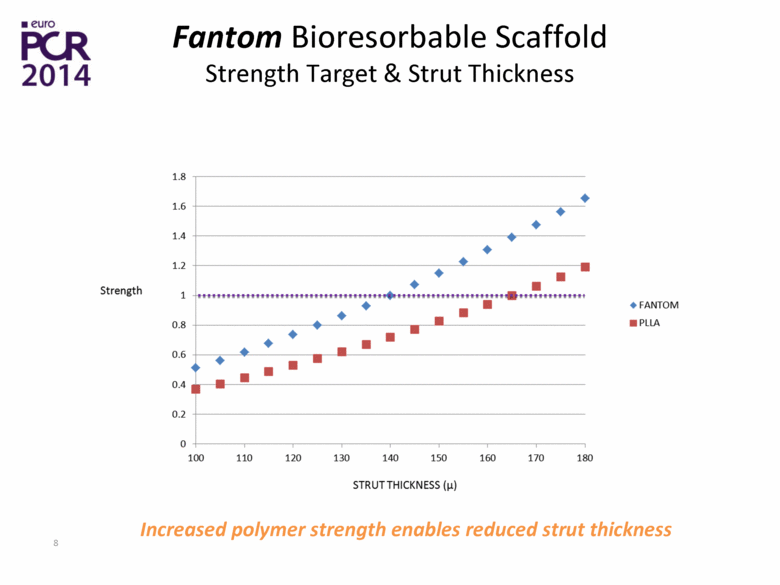
| Fantom Bioresorbable Scaffold Strength Target & Strut Thickness Increased polymer strength enables reduced strut thickness |
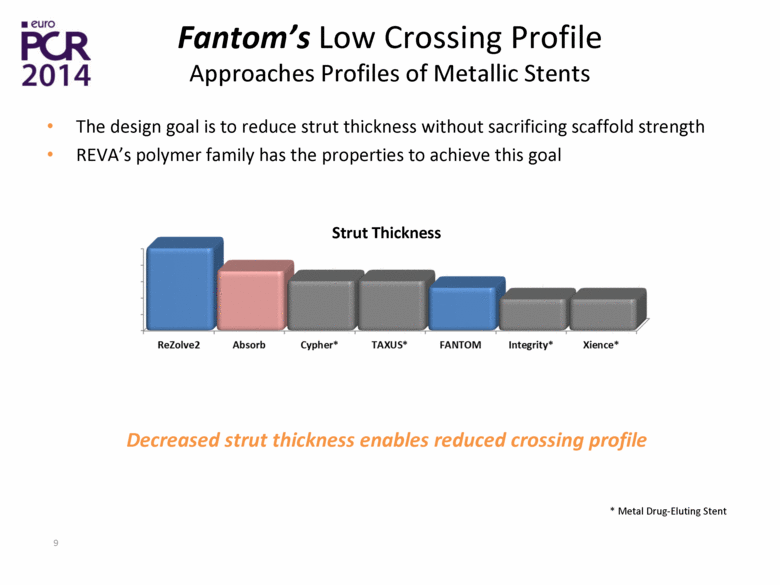
| Fantom’s Low Crossing Profile Approaches Profiles of Metallic Stents The design goal is to reduce strut thickness without sacrificing scaffold strength REVA’s polymer family has the properties to achieve this goal Decreased strut thickness enables reduced crossing profile Strut Thickness * Metal Drug-Eluting Stent |
| Effect of Strut Thickness on Crossing Profile A Small Change Has a Big Impact A reduction in strut thickness of only 25 µm achieves 10% decrease in profile and significant improvements in deliverability PLLA Scaffold 1.4 mm (.055”) Fantom <1.27 mm (.050”) |
| Fantom Bioresorbable Scaffold Single Step Inflation REVAs Advanced Polymer enables single step inflation |
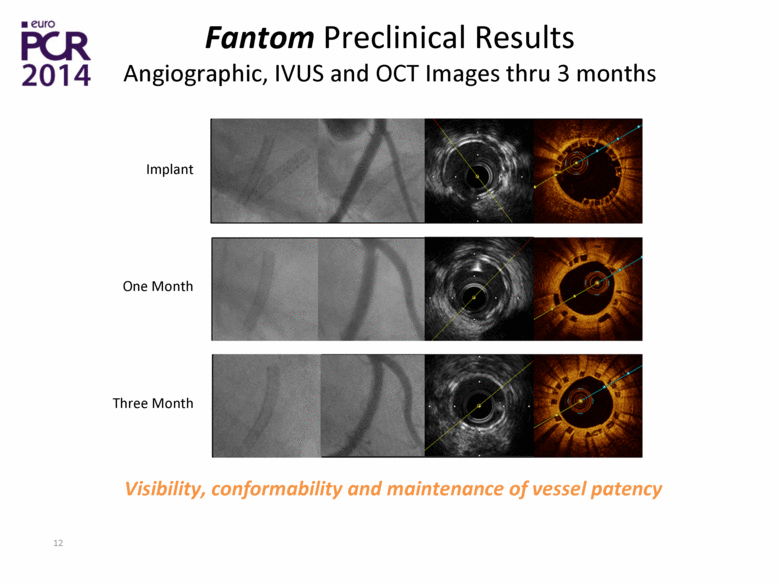
| Fantom Preclinical Results Angiographic, IVUS and OCT Images thru 3 months Implant One Month Three Month Visibility, conformability and maintenance of vessel patency |
| Fantom Clinical Plan First Patient Implants anticipated Q4’14 All CE sites fully enrolling by early 2015 Up to 125 patients Over 20 clinical investigational centers BE, BR, DK, FR, DE, NL and PL Primary endpoints MACE and late lumen loss at six months |
| Fantom Bioresorbable Scaffold Visibility Strength Ease-of-Use Superior Scaffold visibility Scaffold strength to treat challenging lesions Improved deliverability with reduced profile Expansion with one smooth and continuous inflation No requirement for “stepped” inflation No procedural time limitations No special storage requirements Human Clinical Evaluation Q4 2014 |
| Thank you |