Exhibit 99.2
Corplex™ Memantine TDS Phase 1 Interim Topline Results

Forward-looking Statements This presentation and the accompanying oral presentation contain forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, including statements regarding our business strategy, clinical trial timing and plans, the achievement of clinical and commercial milestones, and the advancement of our technologies and our proprietary, co-developed and partnered products and product candidates. Forward-looking statements are based on management’s current expectations and projections and are subject to risks and uncertainties, which may cause Corium’s actual results to differ materially from the statements contained herein. Further information on potential risk factors that could affect Corium’s business and its results are detailed in Corium's Quarterly Report on Form 10-Q for the quarter ended December 31, 2015, filed with the Securities and Exchange Commission on February 12, 2016, and other reports as filed from time to time with the Securities and Exchange Commission. Undue reliance should not be placed on forward-looking statements, especially guidance on future financial or operating performance, which speaks only as of the date they are made. Corium undertakes no obligation to update publicly any forward-looking statements to reflect new information, events or circumstances after the date they were made or to reflect the occurrence of unanticipated events.

Phase 1 Study Overview Open label, partial crossover study Three treatments Memantine TDS, single-dose, once-weekly patch* targeted to deliver 28 mg/day Memantine TDS, single-dose, 3-day patch* targeted to deliver 28 mg/day Seven consecutive days of once-daily oral Namenda XR 28 mg capsule as comparator 11 healthy volunteers (50-80 years of age) Not all subjects completed all treatments Primary endpoint Assess transdermal feasibility Compare pharmacokinetics (PK) of Memantine TDS and Namenda XR Secondary endpoints: safety and skin tolerability *The same formulation was used for the 3-day and once-weekly Memantine TDS product candidates

Phase 1 Study Overview (cont.) Follow-on clinical wear study Evaluate effect of modified patch application procedure and configuration Skin tolerability Adhesion

7-Day Delivery Demonstrated* Oral Namenda XR: administered once daily for 7 days; plasma sampling during Day 1 and on Days 7 through 15 Memantine TDS: single application of once-weekly TDS; plasma sampling during patch application (Days 1-7) and post patch removal (Days 8-15) Memantine TDS: single application of 3-day TDS; plasma sampling during patch application (Days 1-3) and post patch removal (Days 4-11) *The same formulation was used for the 3-day and once-weekly Memantine TDS product candidates **Namenda XR: Marketed in 4 doses: 7, 14, 21 and 28 mg/day Corplex Memantine achieves concentrations comparable to Namenda XR 28 mg Patch size can be scaled to achieve delivery of various doses of Namenda XR** Once-weekly Patch Removed 3-day Patch Removed 0 40 80 120 160 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Memantine Plasma Concentration (ng/ml) Time (days) Once-weekly TDS (n=5) 3-day TDS (n=7) Oral Namenda XR 28 mg Day 1 (n=7) Oral Namenda XR 28 mg Day 7 (n=7) TDS Day 7 expected average concentration equivalent to 28 mg Namenda XR (97 ng/ml)
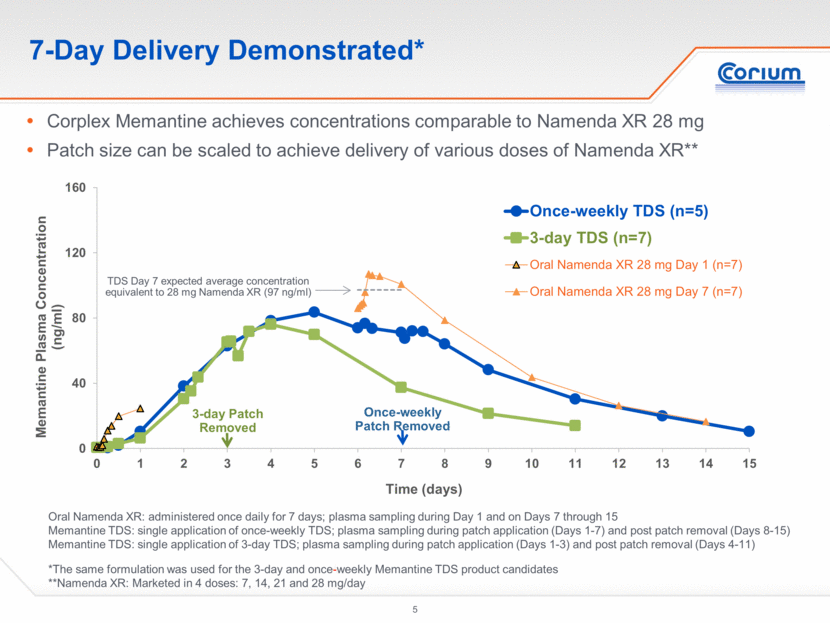
Sustained and Controlled Delivery Demonstrated Sustained PK profile on Day 7 Controlled delivery with minimal peak to trough fluctuation Namenda XR: administered once daily for 7 days; plasma sampling on Day 7 (0-24 hours) Memantine TDS: single application of once-weekly patch with plasma sampling on Day 7 (0-24 hours) Observed Plasma Concentrations throughout Day 7 0 40 80 120 160 0 4 8 12 16 20 24 Mematine Plasma Concentration (ng/ml) Time (hours within day 7) Once-weekly TDS (n=5) Oral Namenda XR 28 mg QD (n=7)
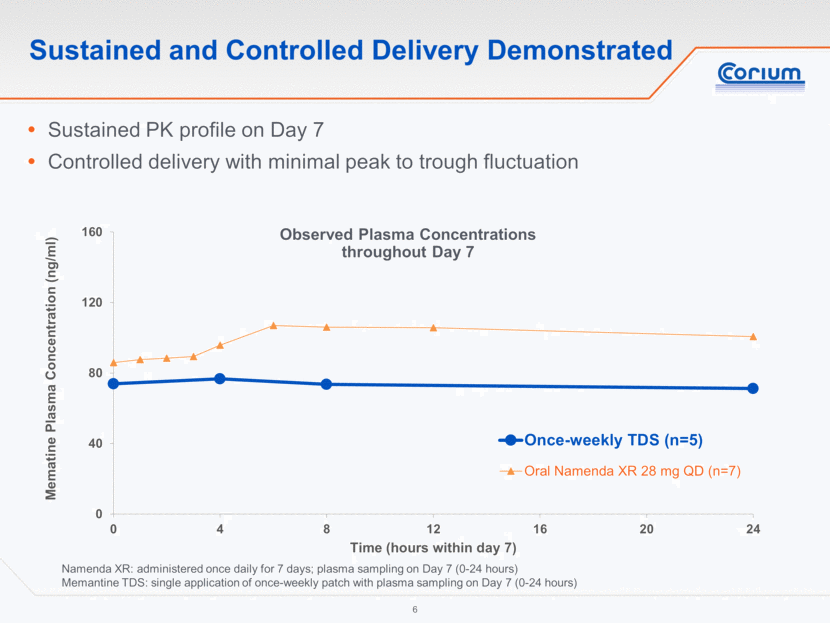
3-Day TDS: Similar PK to Oral Projected at Steady State *TDS steady-state concentrations projected for Days 24-27 immediately after nine consecutive 3-day patch applications **28 mg oral projection based on observed data; 28 mg TDS projection based on observed data with patch size scaled for bioequivalence Sustained and controlled TDS delivery at steady state* Corplex Memantine exposure at steady state highly similar to oral** Supports potential PK based justification for product approval Mean Projected Plasma Concentrations over Steady State in Week 4 0 40 80 120 160 24 25 26 27 Memantine Plasma Concentration (ng/ml) Time (days after first dose) 3-day TDS (n=7) Oral Namenda XR 28 mg QD (n=7)
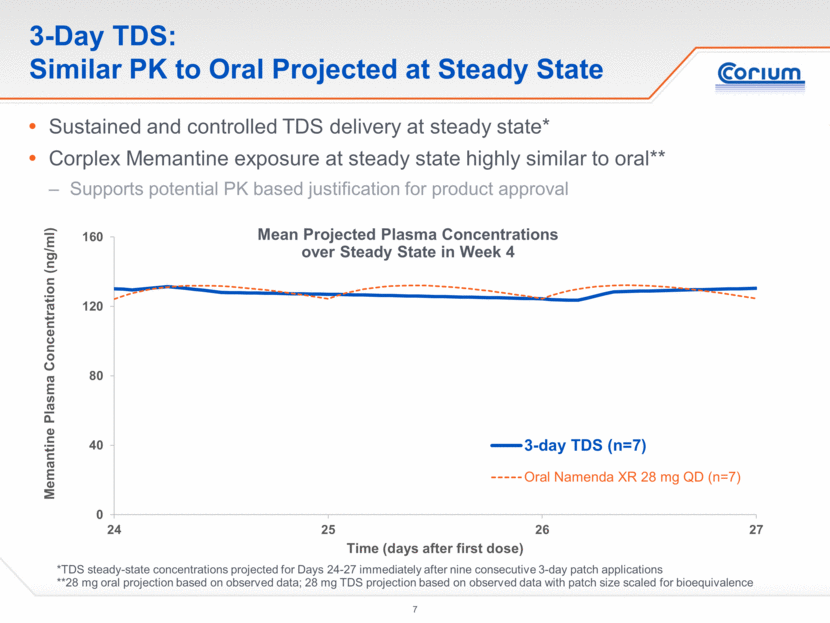
3-Day TDS: Potential Basis for Bioequivalence Approach Geometric Mean Ratio TDS:Oral for PK parameters were between 95%-105% Intra-subject coefficient of variation was 60% Acceptance criteria for bioequivalence are 90% confidence intervals within 80%-125% limits 134 subjects in a two-way crossover study design would be needed for 80% power 181 subjects would be required for 90% power PK Parameters at Steady State 3-day TDS Namenda XR 28 mg QD Geometric Mean Ratio (TDS:Oral) Geometric mean Cmax (ng/ml) 138.3 132.2 1.046 Geometric mean Cmin (ng/ml) 119.9 124.4 0.964 Geometric mean AUCweek (ng-hr/ml) 9186 9319* 0.986 *AUC 0-24 hour x 7 days
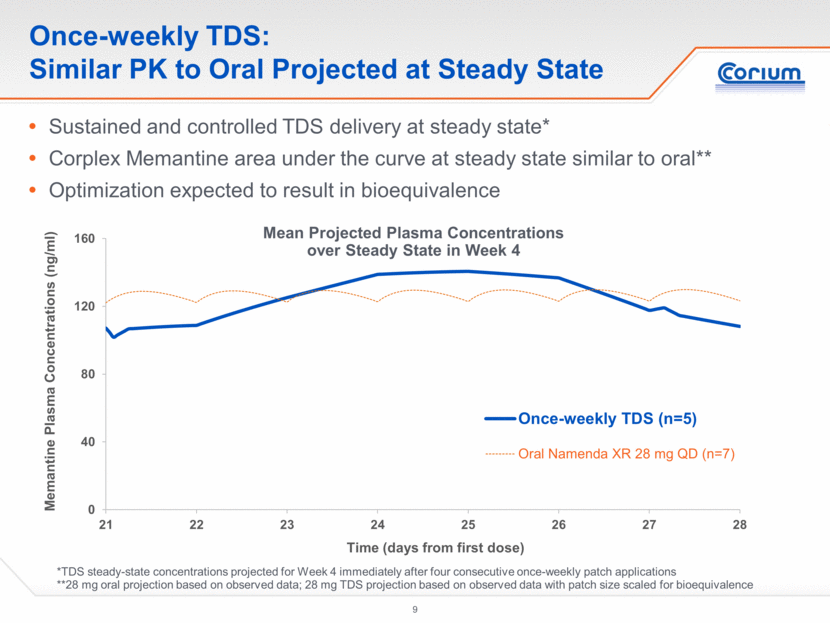
Once-weekly TDS: Similar PK to Oral Projected at Steady State Sustained and controlled TDS delivery at steady state* Corplex Memantine area under the curve at steady state similar to oral** Optimization expected to result in bioequivalence *TDS steady-state concentrations projected for Week 4 immediately after four consecutive once-weekly patch applications **28 mg oral projection based on observed data; 28 mg TDS projection based on observed data with patch size scaled for bioequivalence Mean Projected Plasma Concentrations over Steady State in Week 4 0 40 80 120 160 21 22 23 24 25 26 27 28 Memantine Plasma Concentrations (ng/ml) Time (days from first dose) Once-weekly TDS (n=5) Oral Namenda XR 28 mg QD (n=7)
Safety and Skin Tolerability Local skin tolerability in PK portion of the study Moderate to high skin irritation for 3-day and once-weekly patch 4 subjects discontinued Systemic safety Similarly infrequent for both treatments No systemic adverse events unique to the transdermal delivery Follow-on wear study in eleven new subjects using modified patch application procedure and configuration Significant improvement in skin tolerability for 3-day wear Mild to slightly moderate irritation No subjects discontinued 7-day wear to be determined

Key Phase 1 Conclusions Sustained and controlled PK in target age group (50-80) Achievement of plasma concentrations comparable to Namenda XR 28 mg Patch size can be adjusted to deliver various doses equivalent to Namenda XR Acceptable safety and skin tolerability for 3-day TDS based on wear study Once-weekly TDS skin tolerability to be determined Steady-state TDS projections show comparable memantine exposure to oral Potential for PK-based product approval for 3-day TDS Patch optimization planned for bioequivalence for once-weekly TDS Results support proceeding to confirmatory PK and tolerability study for 3-day Memantine TDS In parallel, pursue optimization of once-weekly TDS Anticipate start of next study for both 3-day and once-weekly TDS as early as Q3 2016

Nasdaq: CORI
