Table of Contents
As filed with the Securities and Exchange Commission on March 30, 2015
Registration No. 333-202665
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
AMENDMENT NO. 2 TO
FORM F-1
REGISTRATION STATEMENT UNDER THE SECURITIES ACT OF 1933
TRILLIUM THERAPEUTICS INC.
(Exact name of Registrant as specified in its charter)
| Ontario, Canada | 2834 | Not applicable | ||
(State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) |
96 Skyway Avenue, Toronto, Ontario, Canada M9W 4Y9
Telephone: (416) 595-0627
(Address, including zip code, and telephone number, including area code, of Registrant’s principal executive offices)
Puglisi & Associates, 850 Library Avenue, Suite 204, Newark, Delaware 19711
Telephone: (302) 738-6680
(Name, address, including zip code, and telephone number, including area code, of agent for service)
| Copies to: | ||
| Daniel M. Miller Dorsey & Whitney LLP Suite 1605, Pacific Centre 777 Dunsmuir Street Vancouver, British Columbia Canada V7Y 1K4 (604) 687-5151 | Thomas S. Levato Goodwin Procter LLP The New York Times Building 620 Eighth Avenue New York, NY 10018 (212) 813-8800 | |
Approximate date of commencement of proposed sale to the public: As soon as practicable after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box. [ ]
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. [ ]
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. [ ]
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. [ ]
CALCULATION OF REGISTRATION FEE
Title of each class of securities to be registered | Proposed maximum aggregate offering price(1)(3)(4) | Amount of registration fee(2) | ||
| Common shares, no par value | ||||
| Series II First Preferred shares, no par value | ||||
| Total | US$57,500,000 | US$6,682 |
(1) Estimated solely for the purpose of determining the amount of registration fee in accordance with Rule 457(o) under the Securities Act of 1933, as amended.
(2) Previously paid.
(3) Includes shares that the underwriters may purchase pursuant to their option to purchase additional common shares, if any.
(4) Includes the common shares issuable upon conversion of the Series II First Preferred shares being registered.
The Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the Registration Statement shall become effective on such date as the Commission, acting pursuant to such Section 8(a), may determine.
Table of Contents
The information in this preliminary prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the United States Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION, DATED MARCH 30, 2015
PROSPECTUS
US$50,000,000

Common Shares
Series II First Preferred Shares
Trillium Therapeutics Inc. is offering US$50,000,000 of common shares and Series II Non-Voting Convertible First Preferred Shares, or the Series II First Preferred Shares, including the common shares issuable from time to time upon conversion of the Series II First Preferred Shares, pursuant to this prospectus. The Series II First Preferred Shares are being offered to certain of our existing shareholders whose purchase of common shares in this offering may result in such shareholder, together with its affiliates and certain related parties, beneficially owning more than 4.99% of our outstanding common shares following the consummation of this offering.
Our common shares are listed for trading on the Toronto Stock Exchange under the symbol “TR” and on the NASDAQ Capital Market under the symbol “TRIL”. On March 27, 2015, the closing price of the common shares on the Toronto Stock Exchange was $23.00 and on the NASDAQ Capital Market was US$18.37. There is no established public trading market for our Series II First Preferred Shares, and we do not expect a market to develop. In addition, we do not intend to apply for listing of our Series II First Preferred Shares on any national securities exchange or other nationally recognized trading system.
Each Series II First Preferred Share is convertible into one common share, subject to adjustment, at any time at the option of the holder, provided that the holder will be prohibited from converting Series II First Preferred Shares into common shares if, as a result of such conversion, the holder, together with its affiliates, would own more than 4.99% of the total number of our common shares then issued and outstanding, unless the holder gives us at least 61 days prior notice of an intent to convert into common shares that would cause the holder to own more than 4.99% of the total number of our common shares then issued and outstanding. See the discussion in the section of this prospectus entitled “Description of Share Capital – First Preferred Shares — Series II First Preferred Shares”.
We are an “emerging growth company” under the U.S. Jumpstart Our Business Startups Act of 2012 and as such have elected to comply with certain reduced public company disclosure requirements. See “Management’s Discussion and Analysis of Financial Condition and Results of Operations — Implications of Being an Emerging Growth Company”.
Investing in our securities involves risks. See “Risk Factors” beginning on page 9 of this prospectus.
| Per Share | Total | |||||||
Price to the public, common shares | US$ | US$ | ||||||
Underwriting discount, common shares(1) | US$ | US$ | ||||||
Proceeds to us (before expenses), common shares | US$ | US$ | ||||||
Price to the public, Series II First Preferred Shares | US$ | US$ | ||||||
Underwriting discount, Series II First Preferred Shares (1) | US$ | US$ | ||||||
Proceeds to us (before expenses), Series II First Preferred Shares | US$ | US$ | ||||||
Aggregate proceeds, before expenses, to us | US$ | US$ | ||||||
(1) Please see the section of this prospectus entitled “Underwriting” for a complete description of the compensation payable to the underwriters.
We have granted the underwriters an option to purchase up to an additional 15% of the total number of common shares sold in this offering. The underwriters can exercise this option at any time within 30 days after the date of this prospectus.
Neither the United States Securities and Exchange Commission nor any state securities commission has approved or disapproved of the securities or passed upon the accuracy or adequacy of this prospectus. Any representation to the contrary is a criminal offense.
The underwriters expect to deliver the offered shares on or about , 2015.
Joint Book-Running Managers
| Leerink Partners | Cowen and Company |
Co-Manager
Oppenheimer & Co.
The date of this prospectus is , 2015.
Table of Contents
i
Table of Contents
All references in this prospectus to “the Company”, “Trillium”, “we”, “us”, or “our” refer to Trillium Therapeutics Inc. and the subsidiaries through which it conducts its business, unless otherwise indicated.
Neither we nor the underwriters have authorized anyone to provide information different from that contained in this prospectus, any amendment or supplement to this prospectus or in any free writing prospectus prepared by us or on our behalf. Neither we nor the underwriters take any responsibility for, and can provide no assurance as to the reliability of, any information other than the information in this prospectus, any amendment or supplement to this prospectus, and any free writing prospectus prepared by us or on our behalf. Neither the delivery of this prospectus nor the sale of our securities means that information contained in this prospectus is correct after the date of this prospectus. This prospectus is not an offer to sell or the solicitation of an offer to buy our securities in any circumstances under which such offer or solicitation is unlawful.
For investors outside the United States, neither we nor any of the underwriters have done anything that would permit this offering or possession or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. You are required to inform yourselves about and to observe any restrictions relating to this offering and the distribution of this prospectus. The offered shares have not been and will not be qualified for distribution pursuant to a prospectus filed with the securities regulatory authorities in any of the provinces or territories of Canada and may not be offered or sold in Canada except pursuant to an exemption from the prospectus requirements of applicable Canadian securities laws.
This prospectus includes statistical data, market data and other industry data and forecasts, which we obtained from market research, publicly available information and independent industry publications and reports that we believe to be reliable sources.
All references in this prospectus to the number of common shares, stock options and deferred share units, or DSUs, the conversion ratio of our Series II First Preferred Shares, our Series I Non-Convertible First Preferred Shares, or the Series I First Preferred Shares, and warrants, and the market price of our common shares prior to November 14, 2014 are presented on a pre-consolidated basis, and on or after November 14, 2014 on a post-consolidation basis, unless otherwise noted.
Unless otherwise indicated, all references to “dollars” or the use of the symbol “$” are to Canadian dollars, and all references to “US dollars” or “US$” are to United States dollars.
ii
Table of Contents
The following table sets forth, for each period indicated, the high, low and average exchange rates for Canadian dollars expressed in United States dollars, provided by the Bank of Canada. The exchange rates set forth below demonstrate trends in exchange rates, but the actual exchange rates used throughout this prospectus may vary. The average exchange rate is calculated by using the average of the closing prices on the last day of each month during the relevant period. On March 27, 2015, the noon exchange rate for one Canadian dollar expressed in United States dollars as reported by the Bank of Canada, was $1.00 = US$0.7949. The high and low exchange rates are intra-day values rather than noon or closing rates.
|
| |||||||||||
$1 Canadian dollar equivalent in United States dollars | High | Low | Average | |||||||||
Year ended December 31, 2010 | US$ | 1.0069 | US$ | 0.9218 | US$ | 0.9657 | ||||||
Year ended December 31, 2011 | 1.0630 | 0.9383 | 1.0136 | |||||||||
Year ended December 31, 2012 | 1.0371 | 0.9576 | 1.0010 | |||||||||
Year ended December 31, 2013 | 1.0188 | 0.9314 | 0.9662 | |||||||||
Year ended December 31, 2014 | 0.9444 | 0.8568 | 0.9021 | |||||||||
September 2014 | 0.9241 | 0.8913 | ||||||||||
October 2014 | 0.9017 | 0.8783 | ||||||||||
November 2014 | 0.8936 | 0.8732 | ||||||||||
December 2014 | 0.8839 | 0.8568 | ||||||||||
January 2015 | 0.8562 | 0.7813 | ||||||||||
February 2015 | 0.8095 | 0.7876 | ||||||||||
iii
Table of Contents
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements within the meaning of applicable securities laws. All statements contained herein that are not clearly historical in nature are forward-looking, and the words ���anticipate”, “believe”, “expect”, “estimate”, “may”, “will”, “could”, “leading”, “intend”, “contemplate”, “shall” and similar expressions are generally intended to identify forward-looking statements. Forward-looking statements in this prospectus include, but are not limited to, statements with respect to:
| • | our expected future loss and accumulated deficit levels; |
| • | our projected financial position and estimated cash burn rate; |
| • | our expectations about the timing of achieving milestones and the cost of our development programs; |
| • | our observations and expectations regarding the binding profile of SIRPaFc with red blood cells compared to anti-CD47 monoclonal antibodies and proprietary CD47-blocking agents and the potential benefits to patients; |
| • | our requirements for, and the ability to obtain, future funding on favorable terms or at all; |
| • | our projections for the SIRPaFc development plan and progress of each of our products and technologies, particularly with respect to the timely and successful completion of studies and trials and availability of results from such studies and trials; |
| • | our expectations about our products’ safety and efficacy; |
| • | our expectations regarding our ability to arrange for the manufacturing of our products and technologies; |
| • | our expectations regarding the progress, and the successful and timely completion, of the various stages of the regulatory approval process; |
| • | our ability to secure strategic partnerships with larger pharmaceutical and biotechnology companies; |
| • | our strategy to acquire and develop new products and technologies and to enhance the capabilities of existing products and technologies; |
| • | our plans to market, sell and distribute our products and technologies; |
| • | our expectations regarding the acceptance of our products and technologies by the market; |
| • | our ability to retain and access appropriate staff, management, and expert advisers; |
| • | our expectations with respect to existing and future corporate alliances and licensing transactions with third parties, and the receipt and timing of any payments to be made by us or to us in respect of such arrangements; and |
| • | our strategy with respect to the protection of our intellectual property. |
All forward-looking statements reflect our beliefs and assumptions based on information available at the time the assumption was made. These forward-looking statements are not based on historical facts but rather on management’s expectations regarding future activities, results of operations, performance, future capital and other expenditures (including the amount, nature and sources of funding thereof), competitive advantages, business prospects and opportunities. By its nature, forward-looking information involves numerous assumptions, inherent risks and uncertainties, both general and specific, known and unknown, that contribute to the possibility that the predictions, forecasts, projections or other forward-looking statements will not occur. Factors which could cause future outcomes to differ materially from those set forth in the forward-looking statements include, but are not limited to:
| • | the effect of continuing operating losses on our ability to obtain, on satisfactory terms, or at all, the capital required to maintain us as a going concern; |
| • | the ability to obtain sufficient and suitable financing to support operations, preclinical development, clinical trials, and commercialization of products; |
| • | the risks associated with the development of novel compounds at early stages of development in our intellectual property portfolio; |
| • | the risks of reliance on third-parties for the planning, conduct and monitoring of clinical trials and for the manufacture of drug product; |
| • | the risks associated with the development of our product candidates including the demonstration of efficacy and safety; |
| • | the risks related to clinical trials including potential delays, cost overruns and the failure to demonstrate efficacy and safety; |
iv
Table of Contents
| • | the risks of delays and inability to complete clinical trials due to difficulties enrolling patients; |
| • | risks associated with our inability to successfully develop companion diagnostics for our development candidates; |
| • | delays or negative outcomes from the regulatory approval process; |
| • | our ability to successfully compete in our targeted markets; |
| • | our ability to attract and retain key personnel, collaborators and advisors; |
| • | risks relating to the increase in operating costs from expanding existing programs, acquisition of additional development programs and increased staff; |
| • | risk of negative results of clinical trials or adverse safety events by us or others related to our product candidates; |
| • | the potential for product liability claims; |
| • | our ability to achieve our forecasted milestones and timelines on schedule; |
| • | financial risks related to the fluctuation of foreign currency rates and expenses denominated in foreign currencies; |
| • | our ability to adequately protect proprietary information and technology from competitors; |
| • | risks related to changes in patent laws and their interpretations; |
| • | our ability to source and maintain licenses from third-party owners; and |
| • | the risk of patent-related litigation and the ability to protect trade secrets, |
all as further and more fully described under the section of this prospectus entitled “Risk Factors”.
Although the forward-looking statements contained in this prospectus are based upon what our management believes to be reasonable assumptions, we cannot assure readers that actual results will be consistent with these forward-looking statements.
Any forward-looking statements represent our estimates only as of the date of this prospectus and should not be relied upon as representing our estimates as of any subsequent date. We undertake no obligation to update any forward-looking statement or statements to reflect events or circumstances after the date on which such statement is made or to reflect the occurrence of unanticipated events, except as may be required by securities legislation.
v
Table of Contents
This summary highlights information contained elsewhere in this prospectus. This summary may not contain all of the information that you should consider before deciding to invest in our common shares or Series II First Preferred Shares. You should carefully read this entire prospectus, including the sections of this prospectus entitled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations”, and the financial statements and related notes included in this prospectus, before investing.
Overview
We are an immuno-oncology company developing innovative therapies for the treatment of cancer. Our lead program, SIRPaFc, is a novel, antibody-like protein that harnesses the innate immune system by blocking the activity of CD47, a molecule whose expression is increased on cancer cells to evade the host immune system. Expressed at high levels on the cell surface of a variety of liquid and solid tumors, CD47 functions as a signal that inhibits the destruction of tumor cells by macrophages via phagocytosis. By blocking the activity of CD47, we believe SIRPaFc has the ability to promote the macrophage-mediated killing of tumor cells in a broad variety of cancers both as a monotherapy and in combination with other immune therapies. We expect to file an investigational new drug, or IND, application in the third quarter of 2015, and shortly thereafter commence phase I studies in acute myeloid leukemia, or AML, and myelodysplastic syndrome, or MDS. In the first phase I study we are also considering dosing patients with solid tumors or lymphomas who have normal bone marrow function in order to more clearly assess the potential hematological toxicity of SIRPaFc. We also plan to continue to conduct preclinical studies in other liquid and solid tumors to identify future clinical indications.
The immune system is the body’s mechanism to identify and eliminate pathogens, and can be divided into the innate immune system and the adaptive immune system. The innate immune system is the body’s first line of defense and consists of proteins and cells, such as macrophages, that identify and provide an immediate response to pathogens. The adaptive immune system is activated by, and adapts to, pathogens, creating a targeted and durable response. Cancer cells often have the ability to reduce the immune system’s ability to recognize and destroy them. We believe SIRPaFc plays a critical role in enhancing the innate immune system and importantly, because of its ability to target macrophages, also has an important downstream effect on the adaptive immune system.
SIRPaFc Key Attributes
| • | Potential efficacy in a broad range of cancers. SIRPaFc blocks the tumor’s ability to transmit a “do not eat” signal allowing macrophages to destroy tumor cells; a mechanism that we believe has broad applicability. | |||
| • | Potential for use as monotherapy and in combination with other therapies. We intend to develop SIRPaFc as a monotherapy as well as potentially for use in combination with other cancer immuno-therapies. | |||
| • | Differentiated pharmacokinetic and safety profile. We believe SIRPaFc’s low level of binding to red blood cells lowers the risk of anemia and lowers the loss of drug from circulation. This pharmacokinetic profile potentially allows for lower dosing and a positive safety profile. | |||
| • | May enhance both innate and adaptive immune response. SIRPaFc may enhance stimulation of tumor attacking T-cells since macrophages, in addition to their role in phagocytosis, can also prime T-cells through antigen-presentation. |
1
Table of Contents
Our Strategy
Our goal is to become a leading innovator in the field of immuno-oncology by targeting immune-regulatory pathways that tumor cells exploit to evade the host immune system. We believe that SIRPaFc has the potential to improve survival and quality of life for cancer patients.
| • | Rapidly advance the clinical development of SIRPaFc. Following the completion of ongoing toxicology studies and current Good Manufacturing Practice, or cGMP, production of our clinical lot, we plan to file an IND in the third quarter of 2015 for a first-in-human trial of SIRPaFc in AML and MDS patients, and possibly solid tumor or lymphoma patients with normal bone marrow function. | |||
| • | Expand our SIRPaFc clinical program to include additional cancer indications. Because CD47 is highly expressed by multiple liquid and solid tumors, and high expression is correlated with worse clinical outcomes, we believe SIRPaFc has potential to be effective in a wide variety of tumors. We plan to carry out the preclinical work necessary to select additional, high potential cancer indications for clinical development. | |||
| • | Maximize value of SIRPaFc through scientific collaborations. SIRPaFc has broad potential applicability in various cancer indications, and we believe it is well suited for use as both a monotherapy and in combination with other agents. We plan to selectively and opportunistically pursue scientific collaborations with academic researchers as well as other companies, in order to realize the full value proposition of SIRPaFc. | |||
| • | To become a leading integrated immuno-oncology company. Developing cancer immunotherapies requires significant experience and development expertise. Our experienced management team, board of directors, scientific advisory board and in-house capabilities enable us to execute against our product development plan. We intend to continue to invest in the infrastructure and personnel to support our continued growth that will enable us to become a leading immuno-oncology company. |
Risk Factors
Our business is subject to a number of risks of which you should be aware before making an investment decision. These risks are discussed more fully in the section of this prospectus entitled “Risk Factors.” These risks include, but are not limited to, the following:
| • | We expect to incur future losses and we may never become profitable. |
| • | We currently have no product revenue and will not be able to maintain our operations and research and development without additional funding. |
| • | We will require additional capital to finance our operations, which may not be available to us on acceptable terms, or at all. As a result, we may not complete the development and commercialization of our product candidates or develop new product candidates. |
| • | Our prospects depend on the success of our product candidates, which are at early stages of development, and we may not generate revenue for several years, if at all, from these products. |
| • | We rely on third parties to plan, conduct and monitor our preclinical studies and clinical trials, and their failure to perform as required would interfere with our product development. |
| • | We expect to be classified as a passive foreign investment company, or PFIC, for 2015 and certain of our U.S. shareholders may suffer adverse tax consequences as a result. |
Implications of Being an Emerging Growth Company
We are an “emerging growth company” under the U.S. Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and will continue to qualify as an “emerging growth company” until the earliest to occur of: (a) the last day of the fiscal year during which we have total annual gross revenues of $1,000,000,000 (as
2
Table of Contents
such amount is indexed for inflation every 5 years by the United States Securities and Exchange Commission, or SEC) or more; (b) the last day of our fiscal year following the fifth anniversary of the date of the first sale of our common shares pursuant to an effective registration statement under the U.S. Securities Act of 1933, as amended, or the Securities Act; (c) the date on which we have, during the previous 3-year period, issued more than $1,000,000,000 in non-convertible debt; or (d) the date on which we are deemed to be a “large accelerated filer”, as defined in Rule 12b–2 of the U.S. Securities Exchange Act of 1934, as amended, or the Exchange Act.
Generally, a company that registers any class of its securities under Section 12 of the Exchange Act is required to include in the second and all subsequent annual reports filed by it under the Exchange Act, a management report on internal control over financial reporting and, subject to an exemption available to companies that meet the definition of a “smaller reporting company” in Rule 12b-2 under the Exchange Act, an auditor attestation report on management’s assessment of the company’s internal control over financial reporting. However, for so long as we continue to qualify as an emerging growth company, we will be exempt from the requirement to include an auditor attestation report in our annual reports filed under the Exchange Act, even if we do not qualify as a “smaller reporting company”. In addition, Section 103(a)(3) of the Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act, has been amended by the JOBS Act to provide that, among other things, auditors of an emerging growth company are exempt from any rules of the Public Company Accounting Oversight Board requiring mandatory audit firm rotation or a supplement to the auditor’s report in which the auditor would be required to provide additional information about the audit and the financial statements of the company.
Any U.S. domestic issuer that is an emerging growth company is able to avail itself of the reduced disclosure obligations regarding executive compensation in periodic reports and proxy statements, and to not present to its shareholders a non-binding advisory vote on executive compensation, obtain approval of any golden parachute payments not previously approved, or present the relationship between executive compensation actually paid and our financial performance. So long as we are a foreign private issuer, we are not subject to such requirements, and will not become subject to such requirements even if we were to cease to be an emerging growth company.
As a reporting issuer under the securities legislation of the Canadian provinces of Ontario, British Columbia, Manitoba, Nova Scotia and Alberta, we are required to comply with all new or revised accounting standards that apply to Canadian public companies. Pursuant to Section 107(b) of the JOBS Act, an emerging growth company may elect to utilize an extended transition period for complying with new or revised accounting standards for public companies until such standards apply to private companies. We have elected not to utilize this extended transition period.
Implications of Being a Foreign Private Issuer
We are also considered a “foreign private issuer” pursuant to Rule 405 promulgated under the Securities Act. In our capacity as a foreign private issuer, we are exempt from certain rules under the Exchange Act that impose certain disclosure obligations and procedural requirements for proxy solicitations under Section 14 of the Exchange Act. In addition, our officers, directors and principal shareholders are exempt from the reporting and “short-swing” profit recovery provisions of Section 16 of the Exchange Act and the rules under the Exchange Act with respect to their purchases and sales of our common shares. Moreover, we are not required to file periodic reports and financial statements with the SEC as frequently or as promptly as United States companies whose securities are registered under the Exchange Act. In addition, we are not required to comply with Regulation FD, which restricts the selective disclosure of material information.
We may take advantage of these exemptions until such time as we are no longer a foreign private issuer. We would cease to be a foreign private issuer at such time as more than 50% of our outstanding voting securities
3
Table of Contents
are held by United States residents and any of the following three circumstances applies: (1) the majority of our executive officers or directors are United States citizens or residents; (2) more than 50% of our assets are located in the United States; or (3) our business is administered principally in the United States.
We have taken advantage of certain reduced reporting and other requirements in this prospectus. Accordingly, the information contained herein may be different than the information you receive from other public companies in which you hold equity securities.
Trading Markets
Our common shares began trading on the Toronto Stock Exchange, or TSX, under the symbol “TR” in April 2014. As of December 2014, our common shares also trade on the NASDAQ Capital Market under the symbol “TRIL”. There is no established public trading market for our Series II First Preferred Shares, and we do not expect a market to develop. In addition, we do not intend to apply for listing of our Series II First Preferred Shares on any national securities exchange or other nationally recognized trading system.
Corporate Information
We were incorporated under the Business Corporations Act (Alberta) on March 31, 2004 as Neurogenesis Biotech Corp. On October 19, 2004, we amended our articles of incorporation to change our name from Neurogenesis Biotech Corp. to Stem Cell Therapeutics Corp., or SCT. On November 7, 2013 SCT was continued under the Business Corporations Act (Ontario), or OBCA. On June 1, 2014 we filed articles of amalgamation to amalgamate SCT with our wholly-owned subsidiary, Trillium Therapeutics Inc., or Trillium Privateco, and renamed the combined company Trillium Therapeutics Inc. We are a company domiciled in Ontario, Canada.
On March 26, 2015, we incorporated a subsidiary, Trillium Therapeutics USA Inc., under the laws of Delaware.
We had one wholly-owned subsidiary, Stem Cell Therapeutics Inc., which was incorporated under the Business Corporations Act (Alberta) on December 22, 1999 and was continued under the OBCA on November 12, 2003 and then extra-provincially registered in Alberta on January 11, 2005. This inactive subsidiary was dissolved on September 17, 2014.
Our head and registered offices are located at 96 Skyway Avenue, Toronto, Ontario, Canada M9W 4Y9. Our telephone number is (416) 595-0627. Our agent for service of process in the United States is Puglisi & Associates, 850 Library Avenue, Suite 204, Newark, Delaware 19711, Telephone: (302) 738-6680. Our website address is www.trilliumtherapeutics.com. Our website and the information contained on, or that can be accessed through, our website will not be deemed to be incorporated by reference in, and are not considered part of, this prospectus. You should not rely on our website or any such information in making your decision whether to purchase our securities.
4
Table of Contents
Issuer | Trillium Therapeutics Inc. | |
Offering Amount | US$50,000,000 of common shares and Series II First Preferred Shares (including the common shares issuable from time to time upon conversion of the Series II First Preferred Shares) | |
Common Shares | ||
Common shares being offered | common shares | |
Common shares to be outstanding immediately after this offering | common shares | |
Option to purchase additional shares | We have granted the underwriters an option for a period of up to 30 days from the date of this prospectus to purchase up to an additional 15% of the total number of common shares sold in this offering at the public offering price less the underwriting discounts and commissions. | |
TSX symbol | TR | |
NASDAQ Capital Market symbol | TRIL | |
Series II First Preferred Shares | ||
Series II First Preferred Shares being offered |
Series II First Preferred Shares (including the common shares issuable from time to time upon conversion of the Series II First Preferred Shares). The Series II First Preferred Shares are being offered to certain of our existing shareholders whose purchase of common shares in this offering may result in such shareholder, together with its affiliates and certain related parties, beneficially owning more than 4.99% of our outstanding common shares following the consummation of this offering. | |
Conversion | Each Series II First Preferred Share is convertible into one common share at any time at the option of the holder, provided that the holder will be prohibited from converting Series II First Preferred Shares into common shares if, as a result of such conversion, the holder, together with its affiliates, would own more than 4.99% of the total number of our common shares then issued and outstanding, unless the holder gives us at least 61 days prior notice of an intent to convert into common shares that would cause the holder to own more than 4.99% of the total number of our common shares then issued and outstanding.See “Description of Share Capital – First Preferred Shares — Series II First Preferred Shares”. | |
Liquidation preference | In the event of our liquidation, dissolution or winding-up, whether voluntary or involuntary, or in the event of any other distribution of our assets among our shareholders for the purpose of winding-up our affairs, or in the event of a reduction or redemption of our capital stock, the holders of Series II First Preferred Shares are entitled to receive an amount per share equal to that amount of money that we received as consideration for such Series II First Preferred Shares divided by the | |
5
Table of Contents
| number of Series II First Preferred Shares issued. After such payment, the holders of Series II First Preferred Shares are not entitled to share in any further distribution of our property or assets. See “Description of Share Capital – First Preferred Shares – Series II First Preferred Shares”. | ||
Voting rights | The holders of our Series II First Preferred Shares will generally have no voting rights, except with respect to such matters and in the manner as to which voting rights are accorded to the holders of specified classes of shares pursuant to the Business Corporations Act (Ontario). | |
Dividends | Holders of Series II First Preferred Shares are entitled to receive dividends as determined and declared at the discretion of our board of directors equally on a one-for-one basis with the holders of the other series of First Preferred Shares and, at the discretion of our board of directors, either in priority to, or equally on a one-for-one basis with, the holders of our common shares and Class B shares. See “Description of Share Capital – First Preferred Shares – Series II First Preferred Shares”. | |
Market for Series II First Preferred Shares | There is no established public trading market for our Series II First Preferred Shares, and we do not expect a market to develop. In addition, we do not intend to apply for listing of our Series II First Preferred Shares on any national securities exchange or other nationally recognized trading system. | |
Offering restrictions | The common shares and Series II First Preferred Shares described in this prospectus are not being offered in Canada, and each purchaser of common shares and Series II First Preferred Shares from the underwriters is deemed to agree, upon acceptance of delivery of the purchased common shares and Series II First Preferred Shares, that the purchaser will not resell the purchased common shares or Series II First Preferred Shares to any resident of Canada or (in the case of the common shares) over the Toronto Stock Exchange or otherwise in Canada for a period of 90 days following the completion of this offering, and to represent that it is not a resident of Canada or purchasing the offered shares on behalf of or as agent for any person that is in or a resident of Canada. See “Underwriting – International Selling Restrictions – Canada”. | |
Use of proceeds | We estimate that we will receive net proceeds from this offering of approximately US$ million, or US$ million if the underwriters exercise in full their option to purchase additional common shares, after deducting underwriting discounts and commissions and estimated offering expenses.
We currently intend to use the net proceeds we receive from this offering for the development of SIRPaFc in AML, MDS, and other potential indications; working capital; and other general corporate purposes.
See the section of this prospectus entitled “Use of Proceeds” for additional information. | |
6
Table of Contents
Dividend policy | We have not declared any dividends since our inception and do not anticipate that we will do so in the foreseeable future. We currently intend to retain future earnings, if any, to finance the development of our business. Any future payment of dividends or distributions will be determined by our board of directors on the basis of our earnings, financial requirements and other relevant factors. | |
Risk factors | Investing in our securities involves a high degree of risk and purchasers of our securities may lose part or all of their investment. See the section of this prospectus entitled “Risk Factors” for a discussion of factors you should carefully consider before deciding to invest in our securities. | |
The number of our common shares to be outstanding after this offering is based on 5,018,139 of our common shares outstanding as of March 20, 2015. The number of common shares to be outstanding after this offering excludes the following:
| • | 64,904,689 Series I First Preferred Shares convertible into 2,163,490 common shares; |
| • | 125,797,904 common share purchase warrants convertible into 4,193,263 common shares at aweighted-average exercise price of $8.66 per common share; |
| • | 580,475 stock options with a weighted-average exercise price of $9.68 per common share; |
| • | 28,777 DSUs; and |
| • | Series II First Preferred Shares being offered pursuant to this prospectus convertible into common shares. |
Except as otherwise indicated herein, all information in this prospectus assumes no exercise of the underwriters’ option to purchase additional common shares.
7
Table of Contents
SUMMARY CONSOLIDATED FINANCIAL INFORMATION
The following tables summarize financial data as at and for the fiscal years ended December 31, 2014, 2013, 2012, 2011, and 2010 prepared in accordance with International Financial Reporting Standards, or IFRS, as issued by the International Accounting Standards Board, or IASB. The financial information in the tables below as at December 31, 2014, 2013 and 2012 and for the years then ended has been derived from our audited consolidated financial statements and related notes included in this prospectus. The financial information in the tables below as at December 31, 2011 and 2010 and for the years then ended has been derived from our audited consolidated financial statements and related notes for those years.
The summary financial data below should be read in conjunction with the financial statements included in this prospectus beginning on page F-1 of this prospectus and with the information appearing in the section of this prospectus entitled “Selected Consolidated Financial Information” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations”. Our historical results do not necessarily indicate results expected for any future period.
Consolidated statement of loss and comprehensive loss data | Year ended December 31, | |||||||||||||||||||
| 2014 | 2013 | 2012 | 2011 | 2010 | ||||||||||||||||
| Net sales | $ | - | $ | - | $ | - | $ | - | $ | - | ||||||||||
| Net loss and comprehensive loss | 12,881,820 | 4,289,308 | 1,061,502 | 3,173,947 | 4,305,867 | |||||||||||||||
| Loss from continuing operations per share(1) | 3.06 | 3.16 | 1.71 | 5.22 | 8.24 | |||||||||||||||
| Net loss per common share(1) | 3.06 | 3.16 | 1.71 | 5.22 | 8.24 | |||||||||||||||
| Fully dilute net loss per common share(1) | 3.06 | 3.16 | 1.71 | 5.22 | 8.24 | |||||||||||||||
| As at December 31, 2014 | ||||||||
Consolidated statement of financial position data | (actual) | (as adjusted)(4) | ||||||
Total assets | $ | 28,186,032 | $ | |||||
Net assets | $ | 24,304,294 | $ | |||||
Capital stock - common | $ | 49,505,792 | $ | |||||
Number of common shares outstanding(2) | 4,427,244 | |||||||
Capital stock - preferred | $ | 10,076,151 | $ | |||||
Number of preferred shares outstanding(3) | 69,504,689 | |||||||
Dividends declared per share | - | - | ||||||
Notes:
| (1) | The per share figures have been restated to reflect a share consolidation ratio of one post-consolidated common share for each 30 pre-consolidation common shares on November 14, 2014. |
| (2) | The number of common shares has been restated to reflect a share consolidation ratio of 1 post-consolidated common share for each 30 pre-consolidation common shares on November 14, 2014. |
| (3) | 30 preferred shares are convertible into one common share. |
| (4) | The as adjusted column reflects the sale by us of common shares at a price of US$ per share and Series II First Preferred Shares at a price of US$ per Series II First Preferred Share in this offering. |
8
Table of Contents
An investment in our securities involves a high degree of risk and should be considered speculative. An investment in our securities should only be undertaken by those persons who can afford the total loss of their investment. You should carefully consider the risks and uncertainties described below, as well as other information contained in this prospectus. The risks and uncertainties below are not the only ones we face. Additional risks and uncertainties not presently known to us or that we believe to be immaterial may also adversely affect our business. If any of the following risks occur, our business, financial condition and results of operations could be seriously harmed and you could lose all or part of your investment. Further, if we fail to meet the expectations of the public market in any given period, the market price of our common shares could decline, which could cause the value of our Series II First Preferred Shares to decline. We operate in a highly competitive environment that involves significant risks and uncertainties, some of which are outside of our control.
Risks Related to our Business and our Industry
We expect to incur future losses and we may never become profitable.
We have incurred losses of $12.9 million, $4.3 million and $1.1 million during 2014, 2013 and 2012, respectively, and expect to incur an operating loss in 2015. We have an accumulated deficit since inception through December 31, 2014 of $50.6 million. We believe that operating losses will continue in and beyond 2015 because we are planning to incur significant costs associated with the preclinical and clinical development of SIRPaFc. Our net losses have had and will continue to have an adverse effect on, among other things, our shareholders’ equity, total assets and working capital. We expect that losses will fluctuate from quarter to quarter and year to year, and that such fluctuations may be substantial. We cannot predict when we will become profitable, if at all.
We currently have no product revenue and will not be able to maintain our operations and research and development without additional funding.
To date, we have generated no product revenue and cannot predict when and if we will generate product revenue. Our ability to generate product revenue and ultimately become profitable depends upon our ability, alone or with partners, to successfully develop our product candidates, obtain regulatory approval and commercialize products, including any of our current product candidates, or other product candidates that we may develop, in-license or acquire in the future. We do not anticipate generating revenue from the sale of products for the foreseeable future. We expect our research and development expenses to increase substantially in connection with our ongoing activities, particularly as we advance our product candidates into clinical trials.
Our prospects depend on the success of our product candidates, which are at early stages of development, and we may not generate revenue for several years, if at all, from these products.
Given the early stage of our product development, we can make no assurance that our research and development programs will result in regulatory approval or commercially viable products. To achieve profitable operations, we, alone or with others, must successfully develop, gain regulatory approval and market our future products. We currently have no products that have been approved by the U.S. Food and Drug Administration, or FDA, Health Canada, or HC, or any similar regulatory authority. To obtain regulatory approvals for our product candidates being developed and to achieve commercial success, clinical trials must demonstrate that the product candidates are safe for human use and that they demonstrate efficacy. While SIRPaFc has begun preclinical studies to support an IND application, we have not yet completed cGMP manufacturing, formal toxicology studies, a phase I clinical trial or subsequent required clinical trials. Our CD200 program is ready to enter formal IND-enabling preclinical studies, but we do not plan to initiate these studies until we have a development partner to share in the cost and responsibility of the program.
Many product candidates never reach the stage of clinical testing and even those that do have only a small chance of successfully completing clinical development and gaining regulatory approval. Product candidates may fail for
9
Table of Contents
a number of reasons, including, but not limited to, being unsafe for human use or due to the failure to provide therapeutic benefits equal to or better than the standard of treatment at the time of testing. Unsatisfactory results obtained from a particular study relating to a research and development program may cause us or our collaborators to abandon commitments to that program. Positive results of early preclinical research may not be indicative of the results that will be obtained in later stages of preclinical or clinical research. Similarly, positive results from early- stage clinical trials may not be indicative of favorable outcomes in later-stage clinical trials. We can make no assurance that any future studies, if undertaken, will yield favorable results. We can make no assurances that toxicology studies in non-human primates will yield results that will allow us to proceed with clinical trials in humans.
The early stage of our product development makes it particularly uncertain whether any of our product development efforts will prove to be successful and meet applicable regulatory requirements, and whether any of our product candidates will receive the requisite regulatory approvals, be capable of being manufactured at a reasonable cost or be successfully marketed. If successful in developing our current and future product candidates into approved products, we will still experience many potential obstacles such as the need to develop or obtain manufacturing, marketing and distribution capabilities. If we are unable to successfully commercialize any of our products, our financial condition and results of operations may be materially and adversely affected.
We rely and will continue to rely on third parties to plan, conduct and monitor our preclinical studies and clinical trials, and their failure to perform as required could cause substantial harm to our business.
We rely and will continue to rely on third parties to conduct a significant portion of our preclinical and clinical development activities. Preclinical activities include in vivo studies providing access to specific disease models, pharmacology and toxicology studies, and assay development. Clinical development activities include trial design, regulatory submissions, clinical patient recruitment, clinical trial monitoring, clinical data management and analysis, safety monitoring and project management. If there is any dispute or disruption in our relationship with third parties, or if they are unable to provide quality services in a timely manner and at a feasible cost, our active development programs will face delays. Further, if any of these third parties fails to perform as we expect or if their work fails to meet regulatory requirements, our testing could be delayed, cancelled or rendered ineffective.
We rely on contract manufacturers over whom we have limited control. If we are subject to quality, cost or delivery issues with the preclinical and clinical grade materials supplied by contract manufacturers, our business operations could suffer significant harm.
We have limited manufacturing experience and rely on contract manufacturing organizations, or CMOs, to manufacture our product candidates for larger preclinical studies and clinical trials. We produce small quantities of our product candidates at bench scale in our laboratory facilities for use in smaller preclinical studies. We rely on CMOs for manufacturing, filling, packaging, storing and shipping of drug product in compliance with cGMP regulations applicable to our products. The FDA ensures the quality of drug products by carefully monitoring drug manufacturers’ compliance with cGMP regulations. The cGMP regulations for drugs contain minimum requirements for the methods, facilities and controls used in manufacturing, processing and packing of a drug product.
We have contracted with Catalent Pharma Solutions, LLC, or Catalent, for the manufacture of the SIRPaFc protein needed for our pre-IND toxicology and pharmacology studies and to supply drug product for our phase I clinical trial. The manufacture of recombinant proteins uses well established processes including a protein expression system. Catalent is producing SIRPaFc using their proprietary GPEx® expression system. We believe that Catalent has the capacity, the systems and the experience to supply SIRPaFc for our planned phase I clinical trial and we may consider using Catalent for manufacturing for later clinical trials. However, since the Catalent manufacturing facility where SIRPaFc is being produced was only recently established, it has not been inspected by the FDA. Any manufacturing failures or delays or compliance issues could cause delays in the completion of our preclinical studies of SIRPaFc, and delays in the submission or approval of our IND and the initiation of our phase I clinical trial.
10
Table of Contents
There can be no assurances that CMOs will be able to meet our timetable and requirements. We have not contracted with alternate suppliers in the event Catalent is unable to scale up production, or if Catalent otherwise experiences any other significant problems. If we are unable to arrange for alternative third-party manufacturing sources on commercially reasonable terms or in a timely manner, we may be delayed in the development of our product candidates. Further, contract manufacturers must operate in compliance with cGMP and failure to do so could result in, among other things, the disruption of product supplies. Our dependence upon third parties for the manufacture of our products may adversely affect our profit margins and our ability to develop and deliver products on a timely and competitive basis.
If clinical trials of our product candidates fail to demonstrate safety and efficacy to the satisfaction of regulatory authorities or do not otherwise produce positive results, we would incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our product candidates.
Before obtaining marketing approval from regulatory authorities for the sale of our product candidates, we must conduct preclinical studies in animals and extensive clinical trials in humans to demonstrate the safety and efficacy of the product candidates. Clinical testing is expensive and difficult to design and implement, can take many years to complete and has uncertain outcomes. The outcome of preclinical studies and early clinical trials may not predict the success of later clinical trials, and interim results of a clinical trial do not necessarily predict final results. A number of companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in advanced clinical trials due to lack of efficacy or unacceptable safety profiles, notwithstanding promising results in earlier trials. We do not know whether the clinical trials we may conduct will demonstrate adequate efficacy and safety to result in regulatory approval to market any of our product candidates in any jurisdiction. A product candidate may fail for safety or efficacy reasons at any stage of the testing process. A major risk we face is the possibility that none of our product candidates under development will successfully gain market approval from the FDA or other regulatory authorities, resulting in us being unable to derive any commercial revenue from them after investing significant amounts of capital in multiple stages of preclinical and clinical testing.
If we experience delays in clinical testing, we will be delayed in commercializing our product candidates, and our business may be substantially harmed.
We cannot predict whether any clinical trials will begin as planned, will need to be restructured or will be completed on schedule, or at all. Our product development costs will increase if we experience delays in clinical testing. Significant clinical trial delays could shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before us, which would impair our ability to successfully commercialize our product candidates and may harm our financial condition, results of operations and prospects. The commencement and completion of clinical trials for our products, including our planned SIRPaFc phase I clinical trial, may be delayed for a number of reasons, including delays related, but not limited, to:
| • | failure by regulatory authorities to grant permission to proceed or placing the clinical trial on hold; |
| • | patients failing to enroll or remain in our trials at the rate we expect; |
| • | suspension or termination of clinical trials by regulators for many reasons, including concerns about patient safety or failure of our contract manufacturers to comply with cGMP requirements; |
| • | any changes to our manufacturing process that may be necessary or desired; |
| • | delays or failure to obtain clinical supply from contract manufacturers of our products necessary to conduct clinical trials; |
| • | product candidates demonstrating a lack of safety or efficacy during clinical trials; |
| • | patients choosing an alternative treatment for the indications for which we are developing any of our product candidates or participating in competing clinical trials; |
| • | patients failing to complete clinical trials due to dissatisfaction with the treatment, side effects or other reasons; |
11
Table of Contents
| • | reports of clinical testing on similar technologies and products raising safety and/or efficacy concerns; |
| • | competing clinical trials and scheduling conflicts with participating clinicians; |
| • | clinical investigators not performing our clinical trials on their anticipated schedule, dropping out of a trial, or employing methods not consistent with the clinical trial protocol, regulatory requirements or other third parties not performing data collection and analysis in a timely or accurate manner; |
| • | failure of our contract research organizations, or CROs, to satisfy their contractual duties or meet expected deadlines; |
| • | inspections of clinical trial sites by regulatory authorities or Institutional Review Boards, or IRBs, or ethics committees finding regulatory violations that require us to undertake corrective action, resulting in suspension or termination of one or more sites or the imposition of a clinical hold on the entire study; |
| • | one or more IRBs or ethics committees rejecting, suspending or terminating the study at an investigational site, precluding enrollment of additional subjects, or withdrawing its approval of the trial; or |
| • | failure to reach agreement on acceptable terms with prospective clinical trial sites. |
Our product development costs will increase if we experience delays in testing or approval or if we need to perform more or larger clinical trials than planned. Additionally, changes in regulatory requirements and policies may occur, and we may need to amend study protocols to reflect these changes. Amendments may require us to resubmit our study protocols to regulatory authorities or IRBs or ethics committees for re-examination, which may impact the cost, timing or successful completion of that trial. Delays or increased product development costs may have a material adverse effect on our business, financial condition and prospects.
If we have difficulty enrolling patients in clinical trials, the completion of the trials may be delayed or cancelled.
As our product candidates advance from preclinical testing to clinical testing, and then through progressively larger and more complex clinical trials, we will need to enroll an increasing number of patients that meet our eligibility criteria. There is significant competition for recruiting cancer patients in clinical trials, and we may be unable to enroll the patients we need to complete clinical trials on a timely basis or at all. The factors that affect our ability to enroll patients are largely uncontrollable and include, but are not limited to, the following:
| • | size and nature of the patient population; |
| • | eligibility and exclusion criteria for the trial; |
| • | design of the study protocol; |
| • | competition with other companies for clinical sites or patients; |
| • | the perceived risks and benefits of the product candidate under study; |
| • | the patient referral practices of physicians; and |
| • | the number, availability, location and accessibility of clinical trial sites. |
If we are unable to successfully develop companion diagnostics for our therapeutic product candidates, or experience significant delays in doing so, we may not achieve marketing approval or realize the full commercial potential of our therapeutic product candidates.
We plan to develop companion diagnostics for our therapeutic product candidates. We expect that, at least in some cases, regulatory authorities may require the development and regulatory approval of a companion diagnostic as a condition to approving our therapeutic product candidates. We have limited experience and capabilities in developing or commercializing diagnostics and plan to rely in large part on third parties to perform these functions. We do not currently have any agreement in place with any third party to develop or commercialize companion diagnostics for any of our therapeutic product candidates.
Companion diagnostics are subject to regulation by the FDA, HC and comparable foreign regulatory authorities as medical devices and may require separate regulatory approval or clearance prior to commercialization. If we,
12
Table of Contents
or any third parties that we engage to assist us, are unable to successfully develop companion diagnostics for our therapeutic product candidates, or experience delays in doing so, our business may be substantially harmed.
Regulatory approval processes are lengthy, expensive and inherently unpredictable. Our inability to obtain regulatory approval for our product candidates would substantially harm our business.
Our development and commercialization activities and product candidates are significantly regulated by a number of governmental entities, including the FDA, HC and by comparable authorities in other countries. Regulatory approvals are required prior to each clinical trial and we may fail to obtain the necessary approvals to commence or continue clinical testing. We must comply with regulations concerning the manufacture, testing, safety, effectiveness, labeling, documentation, advertising and sale of products and product candidates and ultimately must obtain regulatory approval before we can commercialize the product candidate. The time required to obtain approval by such regulatory authorities is unpredictable but typically takes many years following the commencement of preclinical studies and clinical trials. Any analysis of data from clinical activities we perform is subject to confirmation and interpretation by regulatory authorities, which could delay, limit or prevent regulatory approval. Even if we believe results from our clinical trials are favorable to support the marketing of our product candidates, the FDA or other regulatory authorities may disagree. In addition, approval policies, regulations, or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s clinical development and may vary among jurisdictions. We have not obtained regulatory approval for any product candidate and it is possible that none of our existing product candidates or any future product candidates will ever obtain regulatory approval.
We could fail to receive regulatory approval for our product candidates for many reasons, including, but not limited to:
| • | disagreement with the design or implementation of our clinical trials; |
| • | failure to demonstrate that a product candidate is safe and effective for its proposed indication; |
| • | failure of clinical trials to meet the level of statistical significance required for approval; |
| • | failure to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks; |
| • | disagreement with our interpretation of data from preclinical studies or clinical trials; |
| • | the insufficiency of data collected from clinical trials of our product candidates to support the submission and filing of a biologics license application, or BLA, or other submission or to obtain regulatory approval; |
| • | deficiencies in the manufacturing processes or the failure of facilities of CMOs with whom we contract for clinical and commercial supplies to pass a pre-approval inspection; or |
| • | changes in the approval policies or regulations that render our preclinical and clinical data insufficient for approval. |
A regulatory authority may require more information, including additional preclinical or clinical data to support approval, which may delay or prevent approval and our commercialization plans, or we may decide to abandon the development program. If we were to obtain approval, regulatory authorities may approve any of our product candidates for fewer or more limited indications than we request, may grant approval contingent on the performance of costly post-marketing clinical trials, or may approve a product candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product candidate. Moreover, depending on any safety issues associated with our product candidates that garner approval, the FDA may impose a risk evaluation and mitigation strategy, thereby imposing certain restrictions on the sale and marketability of such products.
We will require additional capital to finance our operations, which may not be available to us on acceptable terms, or at all. As a result, we may not complete the development and commercialization of our product candidates or develop new product candidates.
As a research and development company, our operations have consumed substantial amounts of cash since inception. We expect to spend substantial funds to continue the research, development and testing of our product
13
Table of Contents
candidates that are in the preclinical testing stages of development, and to prepare to commercialize products subject to FDA approval in the U.S. and similar approvals in other jurisdictions. We will also require significant additional funds if we expand the scope of our current clinical plans for the SIRPaFc program or if we were to acquire any new assets and advance those towards clinical testing. Therefore, for the foreseeable future, we will have to fund all of our operations and development expenditures from cash on hand, equity or debt financings, through collaborations with other biotechnology or pharmaceutical companies or through financings from other sources. We expect that our existing cash at December 31, 2014 of $26,165,056 will enable us to fund our current operating plan requirements into the fourth quarter of 2016. Additional financing will be required to meet our long term liquidity needs. If we do not succeed in raising additional funds on acceptable terms, we might not be able to complete planned preclinical studies and clinical trials or pursue and obtain approval of any product candidates from the FDA and other regulatory authorities. It is possible that future financing will not be available or, if available, may not be on favorable terms. The availability of financing will be affected by the achievement of our corporate goals, the results of scientific and clinical research, the ability to obtain regulatory approvals, the state of the capital markets generally and with particular reference to drug development companies, the status of strategic alliance agreements and other relevant commercial considerations. If adequate funding is not available, we may be required to delay, reduce or eliminate one or more of our product development programs, or obtain funds through corporate partners or others who may require us to relinquish significant rights to product candidates or obtain funds on less favorable terms than we would otherwise accept. To the extent that external sources of capital become limited or unavailable or available on onerous terms, our intangible assets and our ability to continue our clinical development plans may become impaired, and our assets, liabilities, business, financial condition and results of operations may be materially or adversely affected.
We face competition from other biotechnology and pharmaceutical companies and our financial condition and operations will suffer if we fail to effectively compete.
The biotechnology and pharmaceutical industries are intensely competitive and subject to rapid and significant technological change. Our competitors include large, well-established pharmaceutical companies, biotechnology companies, and academic and research institutions developing cancer therapeutics for the same indications we are targeting and competitors with existing marketed therapies. Many other companies are developing or commercializing therapies to treat the same diseases or indications for which our product candidates may be useful. Although there are no approved therapies that specifically target the CD47 pathway, some competitors use therapeutic approaches that may compete directly with our product candidates. For example, SIRPaFc is in direct competition with CD47 blocking antibodies from Stanford University, Celgene Corporation and Novimmune SA.
Many of our competitors have substantially greater financial, technical and human resources than we do and have significantly greater experience than us in conducting preclinical testing and human clinical trials of product candidates, scaling up manufacturing operations and obtaining regulatory approvals of products. Accordingly, our competitors may succeed in obtaining regulatory approval for products more rapidly than we do. Our ability to compete successfully will largely depend on:
| • | the efficacy and safety profile of our product candidates relative to marketed products and other product candidates in development; |
| • | our ability to develop and maintain a competitive position in the product categories and technologies on which we focus; |
| • | the time it takes for our product candidates to complete clinical development and receive marketing approval; |
| • | our ability to obtain required regulatory approvals; |
| • | our ability to commercialize any of our product candidates that receive regulatory approval; |
| • | our ability to establish, maintain and protect intellectual property rights related to our product candidates; and |
| • | acceptance of any of our product candidates that receive regulatory approval by physicians and other healthcare providers and payers. |
14
Table of Contents
If we are not able to compete effectively against our current and future competitors, our business will not grow and our financial condition and operations will substantially suffer.
We heavily rely on the capabilities and experience of our key executives and scientists and the loss of any of them could affect our ability to develop our products.
The loss of Dr. Niclas Stiernholm, our President and Chief Executive Officer, or other key members of our staff, including Dr. Robert Uger, our Chief Scientific Officer, James Parsons, our Chief Financial Officer, or Dr. Penka Petrova, our Vice President, Drug Development, could harm us. We have employment agreements with Drs. Stiernholm, Uger and Petrova and Mr. Parsons, although such employment agreements do not guarantee their retention. We also depend on our scientific and clinical collaborators and advisors, all of whom have outside commitments that may limit their availability to us. In addition, we believe that our future success will depend in large part upon our ability to attract and retain highly skilled scientific, managerial, medical, clinical and regulatory personnel, particularly as we expand our activities and seek regulatory approvals for clinical trials. We routinely enter into consulting agreements with our scientific and clinical collaborators and advisors, key opinion leaders and academic partners in the ordinary course of our business. We also enter into contractual agreements with physicians and institutions who will recruit patients into our clinical trials on our behalf in the ordinary course of our business. Notwithstanding these arrangements, we face significant competition for these types of personnel from other companies, research and academic institutions, government entities and other organizations. We cannot predict our success in hiring or retaining the personnel we require for continued growth. The loss of the services of any of our executive officers or other key personnel could potentially harm our business, operating results or financial condition.
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have a material adverse effect on our business.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include failures to comply with FDA regulations, provide accurate information to the FDA, comply with manufacturing standards we have established, comply with federal and state health-care fraud and abuse laws and regulations, report financial information or data accurately or disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a substantial impact on our business and results of operations, including the imposition of substantial fines or other sanctions.
We may expand our business through the acquisition of companies or businesses or by entering into collaborations or by in-licensing product candidates, each of which could disrupt our business and harm our financial condition.
We have in the past and may in the future seek to expand our pipeline and capabilities by acquiring one or more companies or businesses, entering into collaborations or in-licensing one or more product candidates. For example, in April 2013, we acquired Trillium Privateco to acquire novel cancer therapeutics, an experienced scientific management team and internal development capabilities. We licensed intellectual property relating to methods and compounds for the modulation of the SIRPa-CD47 interaction for therapeutic cancer applications from University Health Network, or UHN, and Hospital for Sick Children, or HSC. In April 2013, we also in-licensed tigecycline which was subsequently returned to the UHN in 2014. Acquisitions, collaborations and in-licenses involve numerous risks, including, but not limited to:
| • | substantial cash expenditures; |
15
Table of Contents
| • | technology development risks; |
| • | potentially dilutive issuances of equity securities; |
| • | incurrence of debt and contingent liabilities, some of which may be difficult or impossible to identify at the time of acquisition; |
| • | difficulties in assimilating the operations of the acquired companies; |
| • | potential disputes regarding contingent consideration; |
| • | diverting our management’s attention away from other business concerns; |
| • | entering markets in which we have limited or no direct experience; and |
| • | potential loss of our key employees or key employees of the acquired companies or businesses. |
We have experience in making acquisitions, entering collaborations and in-licensing product candidates, however, we cannot provide assurance that any acquisition, collaboration or in-license will result in short-term or long-term benefits to us. We may incorrectly judge the value or worth of an acquired company or business or in-licensed product candidate. In addition, our future success would depend in part on our ability to manage the rapid growth associated with some of these acquisitions, collaborations and in-licenses. We cannot assure you that we would be able to successfully combine our business with that of acquired businesses, manage a collaboration or integrate in-licensed product candidates. Furthermore, the development or expansion of our business may require a substantial capital investment by us.
Negative results from clinical trials or studies of others and adverse safety events involving the targets of our products may have an adverse impact on our future commercialization efforts.
From time to time, studies or clinical trials on various aspects of biopharmaceutical products are conducted by academic researchers, competitors or others. The results of these studies or trials, when published, may have a significant effect on the market for the biopharmaceutical product that is the subject of the study. The publication of negative results of studies or clinical trials or adverse safety events related to our product candidates, or the therapeutic areas in which our product candidates compete, could adversely affect our share price and our ability to finance future development of our product candidates, and our business and financial results could be materially and adversely affected.
We face the risk of product liability claims, which could exceed our insurance coverage and produce recalls, each of which could deplete our cash resources.
We are exposed to the risk of product liability claims alleging that use of our product candidates caused an injury or harm. These claims can arise at any point in the development, testing, manufacture, marketing or sale of our product candidates and may be made directly by patients involved in clinical trials of our product candidates, by consumers or healthcare providers or by individuals, organizations or companies selling our products. Product liability claims can be expensive to defend, even if the product or product candidate did not actually cause the alleged injury or harm.
Insurance covering product liability claims becomes increasingly expensive as a product candidate moves through the development pipeline to commercialization. We currently maintain clinical trial liability insurance coverage of $10 million. However, there can be no assurance that such insurance coverage is or will continue to be adequate or available to us at a cost acceptable to us or at all. We may choose or find it necessary under our collaborative agreements to increase our insurance coverage in the future. We may not be able to secure greater or broader product liability insurance coverage on acceptable terms or at reasonable costs when needed. Any liability for damages resulting from a product liability claim could exceed the amount of our coverage, require us to pay a substantial monetary award from our own cash resources and have a material adverse effect on our business, financial condition and results of operations. Moreover, a product recall, if required, could generate substantial negative publicity about our products and business, inhibit or prevent commercialization of other products and product candidates or negatively impact existing or future collaborations.
16
Table of Contents
In addition, some of our licensing and other agreements with third parties require or might require us to maintain product liability insurance. If we cannot maintain acceptable amounts of coverage on commercially reasonable terms in accordance with the terms set forth in these agreements, the corresponding agreements would be subject to termination, which could have a material adverse impact on our operations.
We may not achieve our publicly announced milestones according to schedule, or at all.
From time to time, we may announce the timing of certain events we expect to occur, such as the anticipated timing of results from our clinical trials. These statements are forward-looking and are based on the best estimates of management at the time relating to the occurrence of such events. However, the actual timing of such events may differ from what has been publicly disclosed. The timing of events such as initiation or completion of a clinical trial, filing of an application to obtain regulatory approval, or announcement of additional clinical trials for a product candidate may ultimately vary from what is publicly disclosed. For example, we cannot provide assurances that the SIRPaFc phase I clinical trial will be initiated on schedule, if at all, or that we will make regulatory submissions or receive regulatory approvals as planned. These variations in timing may occur as a result of different events, including the nature of the results obtained during a clinical trial or during a research phase, problems with a CMO or a CRO or any other event having the effect of delaying the publicly announced timeline. We undertake no obligation to update or revise any forward-looking information, whether as a result of new information, future events or otherwise, except as otherwise required by law. Any variation in the timing of previously announced milestones could have a material adverse effect on our business plan, financial condition or operating results and the trading price of common shares.
We are exposed to the financial risk related to the fluctuation of foreign exchange rates and the degrees of volatility of those rates.
We may be adversely affected by foreign currency fluctuations. To date, we have been primarily funded through issuances of equity, proceeds from the exercise of warrants and stock options and from interest income on funds available for investment, which are all denominated in Canadian dollars. A growing portion of our expenditures are in U.S. dollars, however, and we are, therefore, subject to foreign currency fluctuations which may, from time to time, impact our financial position and results of operations.
Risks Related to Intellectual Property
If we are unable to adequately protect and enforce our intellectual property, our competitors may take advantage of our development efforts or acquired technology and compromise our prospects of marketing and selling our key products.
We own or hold licenses to a number of U.S.-issued patents and U.S. and Canadian pending patent applications, as well as foreign patents and foreign counterparts. In the U.S., we have our own rights to five key issued patents and two pending key patent applications. In addition, we have numerous patent applications pending in Europe, Australia, China, India and Japan. Our success will depend in part upon our ability to protect our intellectual property and proprietary technologies and upon the nature of the intellectual property protection we receive. For example, some of our patent portfolio covers primarily methods of use but not compositions of matter. The ability to compete effectively and to achieve partnerships will depend on our ability to develop and maintain proprietary aspects of our technology and to operate without infringing on the proprietary rights of others. The presence of such proprietary rights of others could severely limit our ability to develop and commercialize our products, to conduct our existing research and/or require financial resources to defend litigation, which may be in excess of our ability to raise such funds. There is no assurance that our pending patent applications or those that we intend to acquire will be approved in a form that will be sufficient to protect our proprietary technology and gain or keep any competitive advantage that we may have.
The patent positions of pharmaceutical companies can be highly uncertain and involve complex legal, scientific and factual questions for which important legal principles remain unresolved. Patents issued to us or our
17
Table of Contents
respective licensors may be challenged, invalidated or circumvented. To the extent our intellectual property, including licensed intellectual property, offers inadequate protection, or is found to be invalid or unenforceable, we are exposed to a greater risk of direct competition. If our intellectual property does not provide adequate protection against our competitors’ products, our competitive position could be adversely affected, as could our business, financial condition and results of operations. Both the patent application process and the process of managing patent disputes can be time consuming and expensive, and the laws of some foreign countries may not protect our intellectual property rights to the same extent as do the laws of Canada and the United States.
We will be able to protect our intellectual property from unauthorized use by third parties only to the extent that our proprietary technologies, key products and any future products are covered by valid and enforceable patents or are effectively maintained as trade secrets and provided we have the funds to enforce our rights, if necessary.
If we lose our licenses from third-party owners we may be unable to continue a substantial part of our business.
We are party to a number of licenses that give us rights to intellectual property that is necessary or useful for a substantial part of our business. Pursuant to our exclusive license agreement with UHN and HSC under which we license certain patent rights for our key products and their uses, we are required to use commercially reasonable efforts to commercialize products based on the licensed rights and pay certain royalties and sublicensing revenue to UHN and HSC. These licenses require that we pay development milestone payments, regulatory milestone payments, royalties on net sales and sublicensing revenues as well as annual maintenance fees. See “Business – General Development of the Business – Acquisition of Trillium Privateco” in this prospectus for additional information regarding this license agreement.
We have also entered into agreements allowing us to manufacture SIRPaFc using Catalent’s proprietary GPEx® expression system. The consideration includes payments at the time we successfully reach a series of development and sales milestones. We may also enter into licenses in the future to access additional third-party intellectual property.
If we fail to pay annual maintenance fees, development and sales milestones, or it is determined that we did not use commercially reasonable efforts to commercialize licensed products, we could lose our licenses, which could have a material adverse effect on our business and financial condition.
We may require additional third-party licenses to effectively develop and manufacture our key products and are currently unable to predict the availability or cost of such licenses.
A substantial number of patents have already been issued to other biotechnology and pharmaceutical companies. To the extent that valid third-party patent rights cover our products or services, we or our strategic collaborators would be required to seek licenses from the holders of these patents in order to manufacture, use or sell these products and services, and payments under them would reduce our profits from these products and services. We are currently unable to predict the extent to which we may wish or be required to acquire rights under such patents and the availability and cost of acquiring such rights, or whether a license to such patents will be available on acceptable terms or at all. There may be patents in the U.S. or in foreign countries or patents issued in the future that are unavailable to license on acceptable terms. Our inability to obtain such licenses may hinder or eliminate our ability to manufacture and market our products.
Changes in patent law and its interpretation could diminish the value of patents in general, thereby impairing our ability to protect our product candidates.
As is the case with other biotechnology and pharmaceutical companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biopharmaceutical industry involves technological and legal complexity, and obtaining and enforcing biopharmaceutical patents is costly,
18
Table of Contents
time-consuming and inherently uncertain. The U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to our and our licensors’ or collaborators’ ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the federal courts, and the U.S. Patent and Trademark Office, or USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our and our licensors’ or collaborators’ ability to obtain new patents or to enforce existing patents and patents we and our licensors or collaborators may obtain in the future.
Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our and our licensors’ or collaborators’ patent applications and the enforcement or defense of our or our licensors’ or collaborators’ issued patents. On September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are prosecuted and may also affect patent litigation. The USPTO recently developed new regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, and in particular, the first to file provisions, only became effective on March 16, 2013. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our or our licensors’ or collaborators’ patent applications and the enforcement or defense of our or our licensors’ or collaborators’ issued patents, all of which could have a material adverse effect on our business and financial condition.
Litigation regarding patents, patent applications and other proprietary rights may be expensive, time consuming and cause delays in the development and manufacturing of our key products.
Our success will depend in part on our ability to operate without infringing the proprietary rights of third parties. The pharmaceutical industry is characterized by extensive patent litigation. Other parties may have, or obtain in the future, patents and allege that the use of our technologies infringes these patent claims or that we are employing their proprietary technology without authorization.
In addition, third parties may challenge or infringe upon our existing or future patents. Proceedings involving our patents or patent applications or those of others could result in adverse decisions regarding:
| • | the patentability of our inventions relating to our key products; and/or |
| • | the enforceability, validity or scope of protection offered by our patents relating to our key products. |
If we are unable to avoid infringing the patent rights of others, we may be required to seek a license, defend an infringement action or challenge the validity of the patents in court. Regardless of the outcome, patent litigation is costly and time consuming. In some cases, we may not have sufficient resources to bring these actions to a successful conclusion. In addition, if we do not obtain a license, develop or obtain non-infringing technology, fail to defend an infringement action successfully or have infringed patents declared invalid, we may:
| • | incur substantial monetary damages; |
| • | encounter significant delays in bringing our key products to market; and/or |
| • | be precluded from participating in the manufacture, use or sale of our key products or methods of treatment requiring licenses. |
Even if we are successful in these proceedings, we may incur substantial costs and divert management time and attention in pursuing these proceedings, which could have a material adverse effect on us.
19
Table of Contents
Our reliance on third parties requires us to share our trade secrets, which increases the possibility that a competitor will discover them.
Because we rely on third parties to develop our products, we must share trade secrets with them. We seek to protect our proprietary technology in part by entering into confidentiality agreements and, if applicable, material transfer agreements, collaborative research agreements, consulting agreements or other similar agreements with our collaborators, advisors, employees and consultants prior to beginning research or disclosing proprietary information. These agreements typically restrict the ability of our collaborators, advisors, employees and consultants to publish data potentially relating to our trade secrets. Our academic collaborators typically have rights to publish data, provided that we are notified in advance and may delay publication for a specified time in order to secure our intellectual property rights arising from the collaboration. In other cases, publication rights are controlled exclusively by us, although in some cases we may share these rights with other parties. We also conduct joint research and development programs which may require us to share trade secrets under the terms of research and development collaboration or similar agreements. Despite our efforts to protect our trade secrets, our competitors may discover our trade secrets, either through breach of these agreements, independent development or publication of information including our trade secrets in cases where we do not have proprietary or otherwise protected rights at the time of publication. A competitor’s discovery of our trade secrets would impair our competitive position and could have a material adverse effect on our business and financial condition.
Risks Related to Our Common Shares and/or our Series II First Preferred Shares
Our common share price has been volatile in recent years, and may continue to be volatile.
The market prices for securities of biopharmaceutical companies, including ours, have historically been volatile. In the year ended December 31, 2013, our common shares traded on the TSX Venture Exchange, or TSXV, at a high of $0.600 and a low of $0.140 per share. In the year ended December 31, 2014, on the TSXV/TSX our common shares traded at a high of $22.20 and a low of $6.30 per share. A number of factors could influence the volatility in the trading price of our common shares, including changes in the economy or in the financial markets, industry related developments, the results of product development and commercialization, changes in government regulations, and developments concerning proprietary rights, litigation and cash flow. Our quarterly losses may vary because of the timing of costs for manufacturing and initiating and completing preclinical and clinical trials. Also, the reporting of adverse safety events involving our products and public rumors about such events could cause our share price to decline or experience periods of volatility. Each of these factors could lead to increased volatility in the market price of our common shares. In addition, changes in the market prices of the securities of our competitors may also lead to fluctuations in the trading price of our common shares.
The value of our Series II First Preferred Shares is directly tied to the value of our common shares, and any change in the value of our common shares will be reflected in the value of our Series II First Preferred Shares.
There is no established public trading market for our Series II First Preferred Shares, and we do not expect a market to develop. In addition, we do not intend to apply for listing of our Series II First Preferred Shares on any national securities exchange or other nationally recognized trading system. As a result, because each offered Series II First Preferred Shares will be convertible into one common share, subject to adjustment and to certain limitations, we expect the value of our Series II First Preferred Shares to have a value directly tied to the value of our common shares. Accordingly, any change in the trading price of our common shares will be reflected in the value of our Series II First Preferred Shares, and the price of our common shares may be volatile, as described above.
We have never paid dividends and do not expect to do so in the foreseeable future.
We have not declared or paid any cash dividends on our common or preferred shares to date. The payment of dividends in the future will be dependent on our earnings and financial condition in addition to such other factors as our board of directors considers appropriate. Unless and until we pay dividends, shareholders may not receive a return on their shares. There is no present intention by our board of directors to pay dividends on our shares.
20
Table of Contents
Future sales or issuances of equity securities and the conversion of outstanding securities to common shares could decrease the value of the common shares, dilute investors’ voting power and reduce our earnings per share.
We may sell additional equity securities in future offerings, including through the sale of securities convertible into equity securities, to finance operations, acquisitions or projects, and issue additional common shares if outstanding warrants or stock options are exercised, or preferred shares are converted to common shares, including the Series II First Preferred Shares offered by us in this offering, which may result in dilution. See the information in the section of this prospectus entitled “Description of Share Capital” for details of our outstanding securities convertible into common shares. We filed a base shelf prospectus with securities commissions in Canada on May 26, 2014 that provides that we may sell under the prospectus from time to time over the following 25 months up to $50 million, in one or more offerings, of common shares, preferred shares, warrants to purchase common shares, or units comprising a combination of common shares, preferred shares and/or warrants. Subject to receipt of any required regulatory approvals, subscribers of the December 2013 private placement who purchased a minimum of 10% of the securities sold under the offering received rights to purchase our securities in future financings to enable each such shareholder to maintain their percentage holding in our common shares for so long as the subscriber holds at least 10% of the outstanding common shares on a fully-diluted basis. Shareholders who do not have this future financing participation right may be disadvantaged in participating in such financings.
Our board of directors has the authority to authorize certain offers and sales of additional securities without the vote of, or prior notice to, shareholders. Based on the need for additional capital to fund expected expenditures and growth, it is likely that we will issue additional securities to provide such capital. Such additional issuances may involve the issuance of a significant number of common shares at prices less than the current market price for our common shares.
Sales of substantial amounts of our securities, or the availability of such securities for sale, as well as the issuance of substantial amounts of our common shares upon conversion of outstanding convertible equity securities, including the Series II First Preferred Shares offered by us in this offering, could adversely affect the prevailing market prices for our securities and dilute investors’ earnings per share. A decline in the market prices of our securities could impair our ability to raise additional capital through the sale of securities should we desire to do so.
We are likely a “passive foreign investment company”, which may have adverse U.S. federal income tax consequences for U.S. shareholders.
U.S. investors should be aware that we believe we were classified as a passive foreign investment company, or PFIC, during the tax year ended December 31, 2014, and based on current business plans and financial expectations, we expect that we will be a PFIC for the current tax year and may be a PFIC in future tax years. If we are a PFIC for any year during a U.S. shareholder’s holding period of our offered shares, then such U.S. shareholder generally will be required to treat any gain realized upon a disposition of our offered shares, or any so-called “excess distribution” received on our offered shares, as ordinary income, and to pay an interest charge on a portion of such gain or distributions, unless the shareholder makes a timely and effective “qualified electing fund” election, or QEF Election, or a “mark-to-market” election with respect to our shares. A U.S. shareholder who makes a QEF Election generally must report on a current basis its share of our net capital gain and ordinary earnings for any year in which we are a PFIC, whether or not we distribute any amounts to our shareholders. A U.S. shareholder who makes the mark-to-market election generally must include as ordinary income each year the excess of the fair market value of the offered shares over the shareholder’s adjusted tax basis therein. This paragraph is qualified in its entirety by the discussion in the section of this prospectus entitled “Certain U.S. Federal Income Tax Considerations”. Each U.S. shareholder should consult its own tax advisors regarding the PFIC rules and the U.S. federal income tax consequences of the acquisition, ownership and disposition of our offered shares.
21
Table of Contents
Risks Related to the Offering
Because the public offering price is substantially higher than the pro forma net tangible book value per share, you will experience immediate and substantial dilution.
The public offering price will be substantially higher than thepro forma net tangible book value per share of our common shares immediately following this offering. Therefore, if you purchase our common shares in this offering, you will experience immediate and substantial dilution of approximately US$ per share (or approximately $ per share) in the price you pay for our common shares as compared to its pro forma net tangible book value, assuming full conversion of our Series II First Preferred Shares sold in this offering. If you purchase our Series II First Preferred Shares in this offering, and assuming that you convert your Series II First Preferred Shares into common shares, you will experience the same dilution per common share. To the extent outstanding options, DSUs and warrants to purchase common shares are exercised, there will be further dilution. For further information on this calculation, see the section of this prospectus entitled “Dilution”.
It may be difficult for non-Canadian investors to obtain and enforce judgments against us because of our Canadian incorporation and presence.
We are a corporation existing under the laws of the Province of Ontario, Canada. Several of our directors and officers, and several of the experts named in this prospectus, are residents of Canada, and all or a substantial portion of their assets, and a substantial portion of our assets, are located outside the United States. Consequently, although we have appointed an agent for service of process in the United States, it may be difficult for holders of our securities who reside in the United States to effect service within the United States upon those directors and officers, and the experts who are not residents of the United States. It may also be difficult for holders of our securities who reside in the United States to realize in the United States upon judgments of courts of the United States predicated upon our civil liability and the civil liability of our directors, officers and experts under the United States federal securities laws. Investors should not assume that Canadian courts (i) would enforce judgments of United States courts obtained in actions against us or such directors, officers or experts predicated upon the civil liability provisions of the United States federal securities laws or the securities or “blue sky” laws of any state or jurisdiction of the United States or (ii) would enforce, in original actions, liabilities against us or such directors, officers or experts predicated upon the United States federal securities laws or any securities or “blue sky” laws of any state or jurisdiction of the United States. In addition, the protections afforded by Canadian securities laws may not be available to investors in the United States.
If there are substantial sales of our common shares, the market price of our common shares could decline.
Sales of substantial numbers of our common shares could cause a decline in the market price of our common shares. Any sales by existing shareholders or holders who exercise their warrants or stock options may have an adverse effect on our ability to raise capital and may adversely affect the market price of our common shares.
We have broad discretion in how we use the net proceeds of the offering, and we may not use these proceeds in a manner desired by our security holders.
We will have broad discretion with respect to the use of the net proceeds from the offering and investors will be relying on the judgment of our management regarding the application of these proceeds. We could spend most of the net proceeds from the offering in ways that our shareholders may not desire or that do not yield a favorable return. You will not have the opportunity, as part of your investment in our securities, to influence the manner in which the net proceeds of the offering are used. We currently intend to use the proceeds of the offering as described in the section of this prospectus entitled “Use of Proceeds”. However, our needs may change as our business and our industry evolve. As a result, the proceeds we receive in the offering may be used in a manner significantly different from our current expectations.
We are an “emerging growth company,” and we cannot be certain if the reduced reporting requirements applicable to emerging growth companies will make our common shares less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are
22
Table of Contents
applicable to other public companies that are not emerging growth companies, including not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We could be an emerging growth company for up to five years, although circumstances could cause us to lose that status earlier. We cannot predict if investors will find our common shares less attractive because we may rely on these exemptions. If some investors find our common shares less attractive as a result, there may be a less active trading market for our common shares and our share price may be more volatile.
Any failure to maintain an effective system of internal controls may result in material misstatements of our consolidated financial statements or cause us to fail to meet our reporting obligations or fail to prevent fraud; and in that case, our shareholders could lose confidence in our financial reporting, which would harm our business and could negatively impact the price of our common shares.
Effective internal controls are necessary for us to provide reliable financial reports and prevent fraud. If we fail to maintain an effective system of internal controls, we might not be able to report our financial results accurately or prevent fraud; and in that case, our shareholders could lose confidence in our financial reporting, which would harm our business and could negatively impact the price of our common shares. While we believe that we have sufficient personnel and review procedures to allow us maintain an effective system of internal controls, we cannot assure you that we will not experience potential material weaknesses in our internal control. Even if we conclude that our internal control over financial reporting provides reasonable assurance regarding the reliability of financial reporting and the preparation of consolidated financial statements for external purposes in accordance with International Financial Reporting Standards, as issued by the International Accounting Standards Board, because of its inherent limitations, internal control over financial reporting may not prevent or detect fraud or misstatements. Failure to implement required new or improved controls, or difficulties encountered in their implementation, could harm our results of operations or cause us to fail to meet our future reporting obligations.
If we fail to timely achieve and maintain the adequacy of our internal control over financial reporting, we may not be able to produce reliable financial reports or help prevent fraud. Our failure to achieve and maintain effective internal control over financial reporting could prevent us from complying with our reporting obligations on a timely basis, which could result in the loss of investor confidence in the reliability of our consolidated financial statements, harm our business and negatively impact the trading price of our common shares.
As a foreign private issuer, we are not subject to certain United States securities law disclosure requirements that apply to a domestic United States issuer, which may limit the information which would be publicly available to our shareholders.
As a foreign private issuer, we are not required to comply with all the periodic disclosure requirements of the Exchange Act, and therefore, there may be less publicly available information about us than if we were a United States domestic issuer. For example, we are not subject to the proxy rules in the United States and disclosure with respect to our annual meetings will be governed by Canadian requirements.
Our charter documents and certain Canadian legislation could delay or deter a change of control, limit attempts by our shareholders to replace or remove our current management and limit the market price of our common shares.
Our authorized preferred shares are available for issuance from time to time at the discretion of our board of directors, without shareholder approval. Our articles grant our board of directors the authority to determine the special rights and restrictions granted to or imposed on any unissued series of preferred shares, and those rights may be superior to those of our common shares. Further, the Investment Canada Act subjects any acquisition of control of a company by a non-Canadian to government review if the value of the assets as calculated pursuant to the legislation exceeds a threshold amount or in other circumstances determined at the discretion of the Canadian government. A reviewable acquisition may not proceed unless the relevant minister is satisfied that the investment is likely to be of net benefit to Canada and the Canadian government is satisfied that no other important concerns arise from the acquisition of control. Any of the foregoing could prevent or delay a change of control and may deprive or limit strategic opportunities to our shareholders to sell their shares.
23
Table of Contents
We estimate that we will receive net proceeds of approximately US$ million (or approximately US$ million if the underwriters’ option to purchase additional common shares is exercised in full) from the sale of common shares and Series II First Preferred Shares offered by us in this offering and after deducting the estimated underwriting discounts and estimated offering expenses payable by us.
We anticipate that we will use the net proceeds of this offering, together with our cash on hand for the development of SIRPaFc in AML, MDS, and other potential indications; working capital; and other general corporate purposes.
We may also use a portion of the net proceeds from this offering to in-license, acquire or invest in complementary businesses, technologies, products or assets. However, we have no current plan, commitments or obligations to do so.
We believe that the net proceeds from this offering and our existing cash and cash equivalents, together with interest thereon, will be sufficient to fund our operations for the next months. Even with the expected net proceeds from this offering, we do not expect to have sufficient cash to complete the clinical development of any of our product candidates or, if applicable, to prepare for commercializing any product candidate that is approved.
Our expected use of net proceeds from this offering represents our current intentions based upon our present plans and business condition. As of the date of this prospectus, we cannot predict with certainty all of the particular uses for the net proceeds to be received upon the completion of this offering, or the amounts that we will actually spend on the uses set forth above. The amounts and timing of our actual use of the net proceeds will vary depending on numerous factors, including our ability to obtain additional financing, the progress, cost and results of our preclinical and clinical development programs, and whether we are able to enter into future product development partnerships and technology license arrangements. As a result, our management will have broad discretion in the application of the net proceeds, and investors will be relying on our judgment regarding the application of the net proceeds from this offering.
Pending their use, we plan to invest the net proceeds from this offering in short- and intermediate-term, interest-bearing obligations, investment-grade instruments, certificates of deposit or direct or guaranteed obligations of the U.S. and Canadian governments.
24
Table of Contents
Overview
We are an immuno-oncology company developing innovative therapies for the treatment of cancer. Our lead program, SIRPaFc, is a novel, antibody-like protein that harnesses the innate immune system by blocking the activity of CD47, a molecule whose expression is increased on cancer cells to evade the host immune system. Expressed at high levels on the cell surface of a variety of liquid and solid tumors, CD47 functions as a signal that inhibits the destruction of tumor cells by macrophages via phagocytosis. By blocking the activity of CD47, we believe SIRPaFc has the ability to promote the macrophage-mediated killing of tumor cells in a broad variety of cancers both as a monotherapy and in combination with other immune therapies. We expect to file an investigational new drug, or IND, application in the third quarter of 2015, and shortly thereafter we expect to commence a phase I clinical trial in acute myeloid leukemia, or AML, and myelodysplastic syndrome, or MDS. In the first phase I study we are also considering dosing patients with solid tumors or lymphomas who have normal bone marrow function in order to more clearly assess the potential hematological toxicity of SIRPaFc. We also plan to continue to conduct preclinical studies in other liquid and solid tumors to identify future clinical indications.
The immune system is the body’s mechanism to identify and eliminate pathogens, and can be divided into the innate immune system and the adaptive immune system. The innate immune system is the body’s first line of defense and consists of proteins and cells, such as macrophages, that identify and provide an immediate response to pathogens. The adaptive immune system is activated by, and adapts to, pathogens, creating a targeted and durable response. Cancer cells often have the ability to reduce the immune system’s ability to recognize and destroy them. We believe SIRPaFc plays a critical role in enhancing the innate immune system and importantly, because of its ability to target macrophages, also has an important downstream effect on the adaptive immune system.
Immuno-oncology is altering cancer treatment
While the concept of using the immune system to treat cancer can trace its roots back to late nineteenth century, it was only recently that the field of immuno-oncology received validation with the approval of antibodies that block CTLA-4 (ipilimumab) and PD-1 (pembrolizumab and nivolumab). These drugs have established the clinical principle that anti-tumor responses can be induced by interfering with the negative signals that normally shut down immune responses. The field of immuno-oncology is now regarded as causing a “paradigm shift” in cancer treatment and has been predicted by equity research analysts to generate US$20 to US$44 billion in revenue by the mid-2020s.
Blocking the CD47 “do not eat” signal using the SIRPaFc decoy receptor
Macrophages are a type of white blood cell that can ingest and destroy (phagocytose) other cells. They are part of the innate immune system that helps to protect the body from infection. More recently, a role for macrophages in the control of tumors has been described.
Macrophage activity is controlled by both positive “eat” and negative “do not eat” signals. Tumor cells may express “eat” signals (e.g., calreticulin) that make themselves visible to macrophages. To counterbalance this increased visibility the tumor cells often express high levels of CD47, which transmits a “do not eat” signal by binding the signal regulatory protein alpha, or SIRPa, on the surface of macrophages. We believe that the higher expression of CD47 on the tumor cell helps it evade destruction by the macrophage by overwhelming any activating “eat” signals.
SIRPaFc is an antibody-like protein that consists of the CD47-binding domain of human SIRPa linked to the Fc region of a human immunoglobulin. It is designed to act as a soluble decoy receptor, preventing CD47 from
25
Table of Contents
delivering its inhibitory signal. Neutralization of the inhibitory CD47 signal enables the activation of macrophage anti-tumor effects by the pro-phagocytic “eat” signals. The Fc region of SIRPaFc may, depending on its identity, also assist in the activation of macrophages by engaging Fc receptors.
Figure 1, below, illustrates how SIRPaFc blocks the CD47 “do not eat” signal.
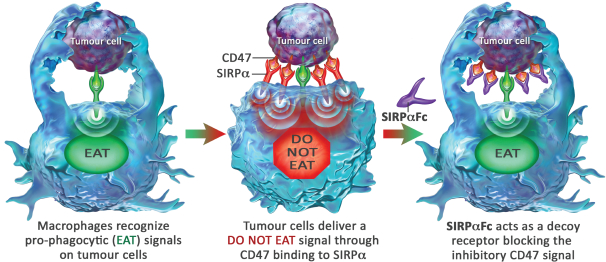
In addition to their direct anti-tumor activity, macrophages can also function as antigen-presenting cells and stimulate antigen-specific T cells. Recent data in mice have demonstrated that CD47 antibody blockade increases antigen presentation by macrophage and stimulates the development of anti-tumor cytotoxic T cell responses. We hypothesize that increasing macrophage phagocytosis by SIRPaFc treatment may also enhance tumor-specific T cell responses. Thus, SIRPaFc may enhance both the innate (macrophage) and adaptive (T cell) components of an anti-tumor immune response.
SIRPaFc has broad clinical potential
We believe that SIRPaFc has broad clinical potential in both hematological and solid tumors. High expression of the CD47 “do not eat” signal on tumor cells has been observed in acute myeloid leukemia, or AML, myelodysplastic syndrome, or MDS, chronic myeloid leukemia, or CML, acute lymphoblastic leukemia, or ALL, diffuse large B cell lymphoma, chronic lymphocytic leukemia, follicular lymphoma, mantle cell lymphoma, marginal zone lymphoma, multiple myeloma and in the following solid tumors: bladder, brain, breast, colon, leiomyosarcoma, liver, melanoma, ovarian and prostate. In a number of these cancers high CD47 expression was shown to have negative clinical consequences, correlating with more aggressive disease and poor survival. In normal karyotype AML patients, for example, high CD47 expression was correlated with worse event-free survival (6.8 vs. 17.1 months) and worse overall survival (9.1 vs. 22.1 months) compared to low CD47 expression. These data are consistent with CD47 providing a survival advantage to tumor cells. Furthermore, numerous studies have shown that antibody blockade of CD47 has demonstrated activity in mice engrafted with human tumors.
In vitro studies with blood cancer lines derived from ALL, B lymphoma, AML, CML and multiple myeloma patients demonstrated that SIRPaFc frequently triggered significant macrophage-mediated tumor cell phagocytosis compared to control treatment.
26
Tumour cell EAT Macrophages recognize pro-phagocytic (EAT) signals on tumour cells Tumour cell CD47 SIRPa DO NOT EAT Tumour cells deliver a DO NOT EAT signal through CD47 binding to SIRPa SIRPaFc Tumour cell EAT SIRPaFC acts as a decoy receptor blocking the inhibitory CD47 signal
Table of Contents
Figure 2, below, shows how SIRPaFc triggers macrophage-mediated phagocytosis of many different human blood cancer cell lines.
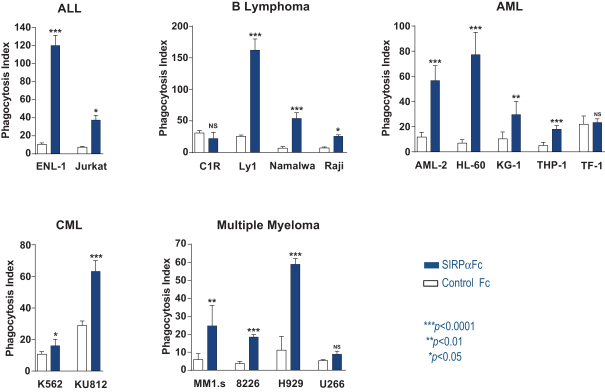
***p<0.0001; **p<0.01; *p<0.05; NS: Thep value is a probability value, with values <0.05 considered statistically significant versus control treatment. NS indicatesp>0.05, which is considered not statistically significant versus control treatment.
In August 2014, we entered into a collaboration with academic investigators to explore the therapeutic potential of SIRPaFc in a variety of solid tumor models. The research is being conducted in the laboratories of Drs. James Koropatnick and Ting-Yim Lee, at the Lawson Health Research Institute and the Robarts Research Institute, University of Western Ontario. Our funding is matched 1:1 by a grant from the Ontario Research Fund – Research Excellence providing the collaboration with a research budget approximating $600,000. We are also working with collaborators at from University Health Network, or UHN, and Hospital for Sick Children, or HSC, and plan to further expand our collaboration network in 2015.
SIRPaFc for the Treatment of AML and MDS
We currently expect our first clinical evaluation of a potential indication for SIRPaFc will be for the treatment of AML and MDS patients. We may also add solid tumor or lymphoma patients with normal bone marrow function. AML is the most common type of acute leukemia in adults, with approximately 13,000 new cases diagnosed each year in the United States. The majority of AML patients receive induction chemotherapy. In patients under 60 years of age, remission rates of up to 75% can be achieved, and patients with good-risk or standard-risk chromosomal changes will typically receive post-remission therapy with high dose cytarabine. However, relapses are common, and the majority of patients will die from their disease. AML in patients over 60 years of age is very difficult to treat, with five-year survival rates reported of less than 10%. There is ample evidence that AML is sustained by leukemic stem cells, or LSCs, and the resistance of these LSCs to conventional chemotherapy is thought to be responsible for disease relapse. CD47 is overexpressed by both bulk differentiated AML cancer cells as well as LSCs.
27
ALL Phagocytosis Index 140 120 100 80 60 40 20 0 *** * ENL-1 Jurkat B Lymphoma Phagocytosis Index 200 150 100 50 0 C1R Ly1 Namalwa Raji NS *** *** * AML Phagocytosis Index 100 80 60 40 20 0 *** *** ** *** NS AML-2 HL-60 KG-1 THP-1 TF-1 CML Phagocytosis 0 20 40 60 80 * *** K562 KU812 Multiple Myeloma Phagocytosis Index 70 60 50 40 30 20 10 0 ** *** *** NS MM1.s 8226 H929 U266 SIRPaFc Control Fc ***p<0.0001 **p<0.01 *p<0.05
Table of Contents
MDS is a heterogeneous group of closely related blood disorders characterized by one or more peripheral blood cytopenias secondary to bone marrow dysfunction. It is considered a premalignant condition and often progresses to AML when additional genetic abnormalities are acquired. There are approximately 13,000 new cases of MDS diagnosed in the United States each year. The median survival times for MDS patients range from 5.7 years (low risk) to 5 months (high risk).
As described above, AML tumor cells have been shown to have high levels of CD47, and CD47 expression has been shown to correlate with worse clinical outcome. Antibody blockade of CD47 has been shown to promote phagocytosis of AML cells in vitro and promote anti-leukemic activity in mouse models. CD47 expression has also been shown to be elevated in bone marrow progenitor cells from high risk MDS patients compared to low risk.
In cell-based experiments, SIRPaFc has been shown to promote the phagocytosis of patient-derived AML tumor samples. In the absence of SIRPaFc treatment, human macrophages are poorly phagocytic. However, treatment of tumor cells with SIRPaFc at doses equal or greater to 10 nanomolar (nM) results in a dramatic increase in tumor cell phagocytosis. This effect is seen visually using confocal microscopy and quantified by expressing a phagocytosis index (number of engulfed tumor cells per 100 macrophages).
Figure 3, below, illustrates how SIRPaFc enables macrophages to kill human AML tumor cells.
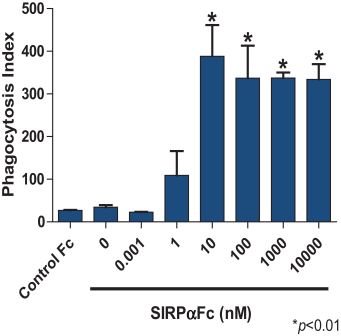
*p<0.01: Thep value is a probability value, with values <0.05 considered statistically significant versus control treatment.
Since AML is a heterogeneous disease, we assessed the impact of SIRPaFc on a panel of 10 AML tumor samples isolated from a diverse patient population at various disease stages and different genetic abnormalities. SIRPaFc was shown to be active against 9 of these 10 samples, indicating that the pro-phagocytic effect of SIRPaFc is not restricted to a subset of AML tumors.
28
Phagocytosis Index 500 400 300 200 100 0 * * * * Control Fc 0 0.001 1 10 100 1000 10000 SIRPaFc (nM) *p<0.01
Table of Contents
Figure 4, below, illustrates how SIRPaFc is active against a diverse panel of AML samples.
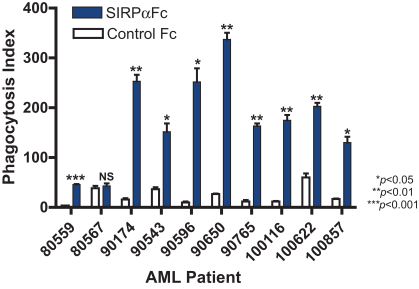
***p<0.0001; **p<0.01; *p<0.05; NS: The p value is a probability value, with values <0.05 considered statistically significant versus control treatment. NS indicatesp>0.05, which is considered not statistically significant versus control treatment.
The below table sets forth the AML patient characteristics supporting the information in Figure 4, above.
Patient | Age | Sex | FAB Subtype(1) | Blast (%)(2) | ||||||||
80559 | 68 | M | M5a | 20 | ||||||||
|
|
|
|
|
|
| ||||||
80567 | 69 | F | M5a | 90 | ||||||||
|
|
|
|
|
|
| ||||||
90174 | 41 | M | M4 | N/A | ||||||||
|
|
|
|
|
|
| ||||||
90543 | 33 | M | M2 | 82 | ||||||||
|
|
|
|
|
|
| ||||||
90596 | 69 | M | M0 | 97 | ||||||||
|
|
|
|
|
|
| ||||||
90650 | 67 | M | M1 | 90 | ||||||||
|
|
|
|
|
|
| ||||||
90765 | 94 | F | M2 | 90 | ||||||||
|
|
|
|
|
|
| ||||||
100116 | 66 | F | M4 | N/A | ||||||||
|
|
|
|
|
|
| ||||||
100622 | 65 | F | M4Eo | 40 | ||||||||
|
|
|
|
|
|
| ||||||
100857 | 73 | M | M2 | 10 | ||||||||
|
|
|
|
|
|
| ||||||
Notes:
| (1) | The French-American-British, or FAB, classification system divides AML into eight subtypes, M0 through to M7, based on the type of cell from which the leukemia developed and its degree of maturity. |
| (2) | Blast (%) refers to the percentage of leukemic myeloblasts, or blood cells affected by disease, compared to all blood cells in the sample. |
The ability of macrophages to kill normal cells in the presence of SIRPaFc was also assessed by confocal microscopy. SIRPaFc enabled macrophages to phagocytose AML tumor cells but spared normal peripheral blood-derived mononuclear cells. This indicates that SIRPaFc-mediated phagocytosis is tumor cell-specific, and strongly suggests that a therapeutic window exists in which SIRPaFc can promote the killing of tumor cells while sparing normal hematopoietic targets. These results are consistent with similar published data using CD47
29
Phagocytosis Index 400 300 200 100 0 SIRPaFc Control Fc *** NS ** * * ** ** ** ** * 80559 80567 90174 90543 90596 90650 90765 100116 100622 100857 *p<0.05 **p<0.01 ***p<0.0001 AML Patient
Table of Contents
antibodies. The strong selectivity for tumor cells may be due to the expression of pro-phagocytic “eat” signals such as calreticulin on malignant cells but not on normal cells.
Figure 5, below, illustrates how SIRPaFc induces tumor cell-specific phagocytosis.
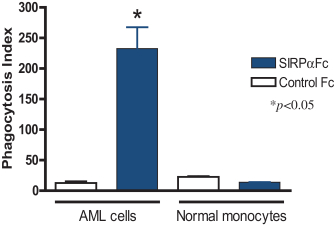 | ||
*Thep value is a probability value, with values <0.05 considered statistically significant versus control treatment.
The activity of SIRPaFc was assessed by engrafting AML patient tumor cells into nonobese diabetic NOD.SCID, or NS, mice. In this model, tumor cells were injected directly into the bone marrow of an NS mouse and three weeks later therapy was initiated with SIRPaFc or a control Fc protein. After treating for 4 weeks, the mice were sacrificed and leukemia in the bone marrow (harvested from the injected and non-injected femurs) and spleen was measured by flow cytometry. Treatment with SIRPaFc in this model resulted in the reduction of leukemia to non-detectable levels in all three tissues in most mice. These results are further validated by independent research demonstrating activity of CD47 blockade using monoclonal antibodies.
Figure 6, below, illustrates how SIRPaFc has potent anti-leukemic activityin vivo.The information presented in Figure 6 is based on the treatment of mice with a human SIRPaFc at a dose of 8 mg/kg intraperitoneal, or IP, three times per week for four weeks, starting 21 days after engraftment.
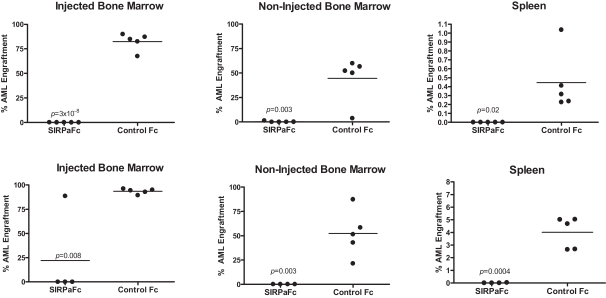
Thep value is a probability value, with values <0.05 considered statistically significant versus control treatment.
30
Phagocytosis Index 300 250 200 150 100 50 0 AML cells Normal monocytes SIRPaFc Control Fc * p<0.05 Injected Bone Marrow % AML Engraftment 100 75 50 25 0 p=3x10-8 SIRPaFc Control Fc Spleen %AML Engraftment 1.1 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0.0 SIRPaFc Control Fc p=0.02 Injected Bone Marrow % AML Engraftment 100 75 50 25 0 P=0.008 SIRPaFc Control Fc Non-Injected Bone Marrow % AML Engraftment 100 75 50 25 P=0.003 SIRPaFc Control Fc Spleen SIRPaFc Control Fc
Table of Contents
The human SIRPaFc used in the above model does not bind mouse CD47 and thus does not model a potential “antigen sink” effect, whereby drug is rapidly removed from circulation by binding target on non-tumor (host) cells. To overcome this limitation, tumor studies were conducted using a mouse surrogate drug that contains the SIRPa sequence from the NS mouse strain, which has the unusual property of binding both mouse and human CD47 and thus can be used in studies in which human tumor cells are transplanted into mice. The mouse surrogate drug was shown to mediate strong anti-leukemic activity in AML tumor studies, even at relatively low doses (<1 mg/kg). These data demonstrate that the presence of CD47 target on normal cells does not interfere with drug activity.
Figure 7, below, illustrates how SIRPaFc has strong anti-leukemic activity even at low doses. The information presented in Figure 7 is based on the treatment of mice with a mouse SIRPaFc at doses of 0.2, 1 or 5 mg/kg IP for three times per week for four weeks, starting 21 days after engraftment.
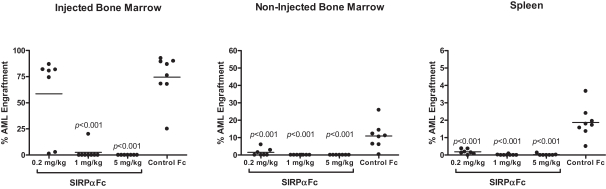
Thep value is a probability value, with values <0.05 considered statistically significant versus control treatment.
Additional AML tumor studies have been conducted, examining various therapeutic dosing regimens, as well as the effect of different Fc regions. The data continue to demonstrate significant anti-tumor activity at doses below 1 mg/kg. The results have also shown that the identity of the Fc region directly impactsin vivo potency.
Several preliminary non-good laboratory practice safety studies have been conducted in cynomolgus monkeys. The results have provided supportive safety and pharmacokinetic data and provided valuable guidance for the design of our formal IND-enabling toxicology program which commenced in February 2015.
31
Injected Bone Marrow % AML Engraftment 100 75 50 25 0 p<0.001 p<0.001 0.2 mg/kg 1 mg/kg 5 mg/kg Control Fc SIRPaFc Non-Injected Bone Marrow % AML Engraftment 60 50 40 30 20 10 0 p<0.001 p<0.001 p<0.001 0.2 mg/kg 1 mg/kg 5 mg/kg Control Fc SIRPaFc Spleen % AML Engraftment 6 5 4 3 2 1 p<0.001 p<0.001 p<0.001 0.2 mg/kg 1 mg/kg 5 mg/kg Control Fc SIRPaFc
Table of Contents
Clinical development plan
Figure 8, below, illustrates the SIRPaFc preclinical development plan. We have completed production of the SIRPaFc toxicology lots and commenced formal IND-enabling toxicology studies. We expect to complete current Good Manufacturing Practice, or cGMP, production of our clinical lot in the second quarter of 2015 and file an IND in the third quarter of 2015 for a first-in-human trial of SIRPaFc in AML and MDS patients. The first clinical study may also involve patients with solid tumors or lymphoma who have normal bone marrow function.
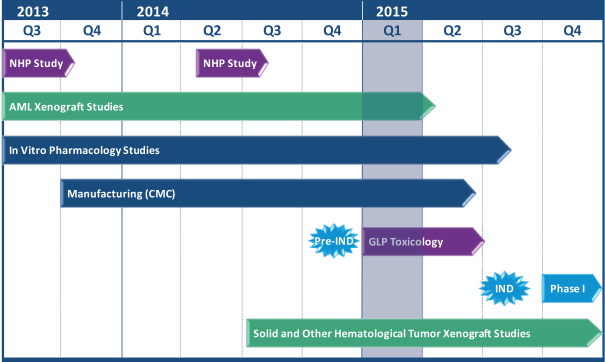
Combination Therapy
SIRPaFc is currently being developed as a monotherapy. However, we believe that SIRPaFc enhancement of macrophage activity and possibly T cell responses could be synergistic with other immune-mediated therapies. Studies conducted by third parties provide evidence that SIRPaFc may be useful in combination with approved anti-cancer antibodies (e.g., Rituxan®, Herceptin®, Campath®). Since many cancer antibodies work at least in part by activating cells of the innate immune system, it may be possible to enhance the potency of these agents by blocking the negative “do not eat” CD47 signal that tumor cells deliver to macrophages. We hypothesize that SIRPaFc may act synergistically with other immunological agents, including T cell checkpoint inhibitors, cancer vaccines, oncolytic viruses or chimeric antigen receptor, or CAR, T cells. We therefore plan to explore SIRPaFc in combination studies using preclinical tumor models.
Our Strategy
Our goal is to become a leading innovator in the field of immuno-oncology by targeting immune-regulatory pathways that tumor cells exploit to evade the host immune system. We believe that SIRPaFc has the potential to improve survival and quality of life for cancer patients.
| • | Rapidly advance the clinical development of SIRPaFc. Following the completion of ongoing toxicology studies and cGMP production of our clinical lot, we plan to file an IND in the third quarter of 2015 for a first-in-human trial of SIRPaFc in AML and MDS patients, and possibly solid tumor or lymphoma patients with normal bone marrow function. |
32
2013 2014 2015 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 NHP Study NHP Study AML Xenograft Studies In Vitro Pharmacology Studies In Vitro Pharmacology Studies Manufacturing (CMC) Pre-IND GLP Toxicology IND Phase I Solid and Other Hematological Tumor Xenograft Studies
Table of Contents
| • | Expand our SIRPaFc clinical program to include additional cancer indications. Because CD47 is highly expressed by multiple liquid and solid tumors, and high expression is correlated with worse clinical outcomes, we believe SIRPaFc has potential to be effective in a wide variety of tumors. We plan to carry out the preclinical work necessary to select additional, high potential cancer indications for clinical development. |
| • | Maximize value of SIRPaFc through scientific collaborations. SIRPaFc has broad potential applicability in various cancer indications, and we believe it is well suited for use as both a monotherapy and in combination with other agents. We plan to selectively and opportunistically pursue scientific collaborations with academic researchers as well as other companies, in order to realize the full value proposition of SIRPaFc. |
| • | To become a leading integrated immuno-oncology company. Developing cancer immunotherapies requires significant experience and development expertise. Our experienced management team, board of directors, scientific advisory board and in-house capabilities enable us to execute our product development plan. We intend to continue to invest in the infrastructure and personnel to support our continued growth that will enable us to become a leading immuno-oncology company. |
Competition - CD47/ AML
SIRPaFc competes directly with CD47 blocking antibodies from Stanford University and Celgene Corporation, both of which have entered early clinical development. Novimmune SA also has a bispecific (anti-CD47/anti-CD19) antibody program, although it is still in the discovery stage.
We believe that our approach of using SIRPa, the natural binding partner for CD47, may have important advantages over treatment with CD47-specific antibodies. In April 2014, we presented data at the 105th American Association for Cancer Research annual meeting demonstrating that our SIRPaFc fusion proteins bind very poorly to human red blood cells, or RBCs, compared to both commercial anti-CD47 monoclonal antibodies and proprietary CD47-blocking agents. In Figure 9, below, on the left, we demonstrate the poor binding of SIRPaFc to RBCs compared to four anti-CD47 antibodies. This profound difference in binding was not seen with other cell types, including an AML cell line (AML-2, right side), suggesting it is an RBC-specific phenomenon. Our observation is consistent with a prior report demonstrating a notable difference in human RBC binding between CD47 antibody and recombinant SIRPa. Furthermore, the lack of significant binding of SIRPaFc to RBCs is unique to humans, since SIRPaFc bound strongly to mouse and monkey RBCs. While the mechanism behind this observation is still under investigation, our preliminary data suggest that it may relate to unique structural features of CD47 in the human RBC membrane.
33
Table of Contents
Figure 9, below, illustrates how SIRPaFc binds very poorly to human RBCs compared to CD47 antibodies. The information presented in Figure 9 is based on the binding of commercially available anti-CD47 monoclonal antibodies and SIRPaFc to human RBCs (n=14 donors) and AML-2 cells.
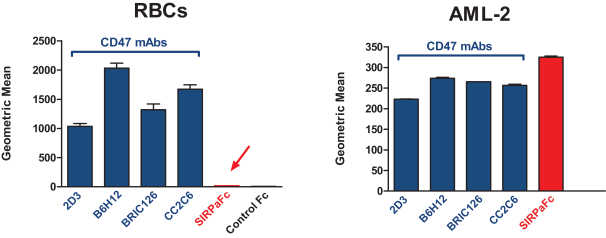
The very low RBC binding profile of our SIRPaFc proteins may provide two important advantages over other CD47 blocking agents. First, there may be a lower risk of RBC toxicity (anemia) with SIRPaFc. Anemia is a condition in which the blood is deficient in red blood cells, in hemoglobin, or in total volume. This potential advantage would translate into improved patient care, particularly in diseases such as AML where patients are predisposed to developing anemia. Second, there may be much lower loss of SIRPaFc from circulation in treated patients compared to CD47 antibodies, which bind strongly to circulating RBCs. The ability of RBCs to absorb and remove circulating CD47-specific antibodies, a so called “antigen-sink effect”, may necessitate high doses of drug in order to effectively target the tumor cells, which could lead to potentially higher off-target toxicity. Therefore, SIRPaFc may have a better safety profile and drug kinetics than CD47-specific antibodies based on these preclinical analyses.
While there is significant competitive clinical activity in AML, there are very few agents in development that are pursuing the same unique mechanism of action (enhancement of macrophage phagocytic activity) as SIRPaFc. To our knowledge, there are a few parties pursuing CD47-blocking antibodies, but no one else developing a SIRPaFc protein. In addition, there are few agents in development targeting AML LSCs. Similar to our targeting of CD47, we believe other agents in development targeting AML LSCs are largely focused on cell surface proteins such as CD123 and CD44.
Plan of Operations
Over the next 12 months, our primary focus is the advancement of our SIRPaFc development program with the intent to initiate the first phase I clinical trial of SIRPaFc in AML and MDS patients. This study may also involve patients with solid tumors or lymphomas who have normal bone marrow function. The major tasks to be performed to accomplish this include completion of manufacturing of cGMP material suitable for clinical testing, completion of IND-enabling toxicology and pharmacology studies, submission of our IND application for FDA approval to start the trial in the third quarter of 2015 and the initiation of the clinical trial in the fourth quarter of 2015. We will also continue preclinical studies in other blood cancers and solid tumors to identify potential future clinical indications. See the sections of this prospectus entitled “Business – Overview” and “Use of Proceeds” for additional information.
Product for Development with Partners – CD200 Monoclonal Antibodies
Our CD200 monoclonal antibody program also targets a key pathway that tumor cells use to evade attack from the immune system. There is evidence that CD200 is highly expressed by many different types of blood cell and
34
RBCs Genometric Mean CD47 mAbs 2500 2000 1500 1000 500 0 2D3 B6H12 BRIC126 CC2C6 SIRPaFc Control Fc AML-2 Genometric Mean CD47 mAbs 350 300 250 200 150 100 50 0 2D3 B6H12 BRIC126 CC2C6 SIRPaFc
Table of Contents
solid tumors, and in numerous cases this high expression correlates with disease progression and poor clinical outcome. This is consistent with tumor cells using CD200 as a means of evading immune-mediated destruction. Tumor-expressed CD200 can modulate anti-tumor responses in vitro and in vivo, and antibodies that block CD200 have been shown to promote anti-tumor immunity in animal models of cancer.
Our CD200 monoclonal antibody is a fully human monoclonal antibody that blocks the activity of CD200. We have conducted in vitro and in vivo preclinical oncology-focused studies. Our antibodies bind strongly to human CD200, potently neutralizing CD200 function in vitro, and have shown anti-tumor activity in a transplanted human tumor cell model. We are seeking a development partner to advance this program into formal preclinical IND-enabling studies.
General Development of the Business
Acquisition of Trillium Privateco
During 2013, we refocused our product pipeline through the strategic acquisition of Trillium Privateco, a private biopharmaceutical company specializing in immune regulation and the development of cancer therapeutics which included the SIRPaFc and CD200 preclinical cancer programs. On April 9, 2013, we completed a merger with Trillium Privateco, to access its SIRPaFc immuno-oncology program. Prior to this merger, we were focused on regenerative stem cell technologies. Following the merger, in July 2013, we ceased all activities associated with our regenerative neurology programs and abandoned all related intellectual property filings.
As consideration for the merger, we paid $1,200,000 in cash and issued 2,779,180 common shares and 3,300,000 units. Each unit consisted of one common share and one common share purchase warrant, with each common share purchase warrant allowing its holder to acquire one additional common share at an exercise price of $0.40 until March 15, 2018. Cash used in the investment of $647,996 was determined by subtracting cash acquired of $552,004 from cash consideration of $1,200,000. Post-closing, Trillium Privateco shareholders held approximately 16% of the issued and outstanding common shares, and Trillium Privateco became our wholly-owned subsidiary.
Trillium Privateco’s SIRPaFc program originated from leading researchers in the field, including Drs. John Dick and Jean Wang of the UHN and Dr. Jayne Danska of HSC. Exclusive rights to SIRPaFc have been licensed from UHN and HSC pursuant to a license agreement amended and restated as of June 1, 2012. The licensed intellectual property relates to methods and compounds used in the modulation of SIRPa-CD47 interaction for therapeutic cancer applications. The license agreement requires us to use commercially reasonable efforts to commercialize the licensed technology. The license agreement will terminate on a country-by-country basis, in countries where a valid claim exists, when the last valid claim expires in such country, or if no valid claim exists, when the last valid claim expires in the U.S.
The license includes commitments to pay an annual maintenance fee of $25,000, as well as payments on patent issuances, development milestone payments ranging from $100,000 to $300,000 on the initiation of phase I, II and III clinical trials, and payments on the achievement of certain regulatory milestones as well as low single digit royalties on commercial sales. The payments due on submission of the first biologics license application, or BLA, and receipt of a first regulatory approval in the U.S. are $1,000,000 for each milestone. The aggregate milestones payable on their first achievement under the agreement in the major markets of the U.S., Europe and Asia combined are $5,660,000. We are also required to pay 20% of any sublicensing revenues to the licensors on the first $50 million of sublicensing revenues received and 15% thereafter.
Tigecycline License
On April 16, 2013, we signed an agreement with UHN to gain rights to intellectual property related to the use of tigecycline for the treatment of leukemia which included a UHN sponsored, open label phase I multicenter dose-escalation tigecycline trial in patients with relapsed or refractory AML. The initial consideration for the UHN
35
Table of Contents
license of $1,085,714 was satisfied by the issuance of 5,028,571 common shares and 1,600,000 common share purchase warrants, each warrant allowing its holder to acquire one additional common share at an exercise price of $0.40 until March 15, 2018. Additional consideration under the UHN license included an annual license maintenance fee and future development milestones. The fair value of the common shares issued was based on the closing price of our common shares of $0.20 on April 16, 2013 and $0.25 per unit based on the fair value of the common share and common share purchase warrant components as of the date of acquisition.
Since acquiring these rights, we monitored the results of the phase I trial and assessed alternate development strategies for the technology. In August 2014, we completed an evaluation of this program with respect to its scientific merit, commercial potential, strength of intellectual property, as well as its overall fit with our current focus and expertise. We concluded that we should focus our business plan on expanding the SIRPaFc program, rather than continue development of tigecycline, and returned the licensed rights to UHN.
Capital Expenditures
Prior to the April 2013 merger with Trillium Privateco, we incurred minimal capital expenditures in 2012. Capital expenditures are required mainly for laboratory equipment, office equipment, computers and leasehold improvements. Capital expenditures in the last three fiscal years are set out in the following table.
| Year ended December 31, | ||||||||||||||||||
| 2014 | 2013 | 2012 | ||||||||||||||||
Capital expenditures | $ | 173,603 | $ | 34,017 | $ | - | ||||||||||||
Agreements with Catalent Pharma Solutions
In connection with our development of SIRPaFc, we entered into two agreements on August 12, 2014 with Catalent pursuant to which we acquired the right to use two of Catalent’s proprietary GPEx® expression cell lines for the manufacture of SIRPaFc proteins: TTI-621 and TTI-622. One agreement relates to the manufacture of TTI-621 and the other agreement relates to the manufacture of TTI-622. In consideration for the purchase of the expression cell lines, each agreement provides that we will pay Catalent up to US$875,000 upon reaching certain pre-marketing approval milestones and up to an additional US$28.8 million for reaching certain sales milestones. We will also pay Catalent an annual product maintenance fee until the first product derived from the expression cell lines receives a regulatory approval other than a pricing or reimbursement approval.
Under the agreements, we may use the two expression cell lines to secure such regulatory approvals and to develop, test, market and otherwise commercially exploit products originating from the cell lines. We may transfer the expression cell lines to a third party contract manufacturer who may utilize the cell lines in a similar fashion. We, or a third-party, cannot use or modify the cell lines, or any portions of the cell lines, to create a new cell line.
We plan to further develop the expression cell lines for use in our pre-IND toxicology and pharmacology studies, as well as to supply our phase I clinical trial. We will be required to indemnify Catalent for any costs Catalent incurs related to regulatory filings and related claims or proceedings, for the conduct of any clinical trials and for any manufacture, packaging, sale, promotion, distribution, use of or exposure to the expression cell lines or products. As a result of this risk, we are obligated to maintain several designated insurance policies throughout the term of the agreements.
We may terminate the agreements upon 90 days’ written notice to Catalent, upon their bankruptcy or upon their material breach and failure to cure within 30 days. Similarly, Catalent may terminate the agreements upon our bankruptcy or upon our material breach and failure to cure within 30 days. If our material breach is for nonpayment, however, we will only have 10 days to cure before Catalent may terminate the agreement.
36
Table of Contents
Intellectual Property
We own or control patent rights covering our key products and their therapeutic end uses. The patents and patent applications are either granted or pending in major pharmaceutical markets. In all, the patent estate includes five key patent families, including five key issued patents and two key pending patent applications in the U.S. and various other patent applications pending in Europe, Canada, Australia, China, India and Japan. These include four granted patents in the U.S. relating to the immune modulating use of CD200 monoclonal antibodies, and a granted patent in the U.S. for an assay useful to detect CD200 in cancer patients, as well as a filing for novel human antibodies to CD200.
In connection specifically with patent applications relating to SIRPaFc, we control two patent families that comprise eight individual filings. One family has claims that embrace species of SIRPaFc found to have certain therapeutic properties and their use for the treatment of cancer. These patent rights are owned outright by Trillium and national patent filings are planned. Patents emerging from this family will expire in 2033. A second SIRPa patent family was in-licensed on an exclusive basis from co-owners UHN and HSC. This family has been filed in major markets, including the U.S., Europe, Japan, Canada, Australia, China and India. The claims cover the use of various forms of SIRPa to treat CD47-positive cancers. Patents issuing from this family begin to expire in the year 2030.
In connection specifically with CD200 monoclonal antibody patents, we have four granted U.S. patents that cover the use of CD200 antibodies for the treatment of autoimmune and related disorders that will begin to expire in 2018. We more recently made a patent filing that covers particular species of human antibodies to human CD200. A patent family based on this filing would expire in 2035.
We intend to protect additional intellectual property developed by us through the filing of patent applications within appropriate jurisdictions throughout the world.
Regulatory Process
Government authorities in the United States, including federal, state, and local authorities, and in other countries, extensively regulate, among other things, the manufacturing, research and clinical development, marketing, labeling and packaging, storage, distribution, post-approval monitoring and reporting, advertising and promotion, and export and import of biological products, such as those we are developing. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local, and foreign statutes and regulations require the expenditure of substantial time and financial resources.
Securing final regulatory approval for the manufacture and sale of biological products in the U.S., Europe, Canada and other commercial territories, is a long and costly process that is controlled by that particular territory’s regulatory agency. The regulatory agency in the U.S. is the FDA, in Canada it is HC, and in Europe it is the European Medicines Agency, or EMA. Other regulatory agencies have similar regulatory approval processes, but each regulatory agency has its own approval processes. Approval in the U.S., Canada or Europe does not assure approval by other regulatory agencies, although often test results from one country may be used in applications for regulatory approval in another country.
None of our products have been completely developed or tested and, therefore, we are not yet in a position to seek final regulatory approval to market any of our products.
U.S. Government Regulation
In the U.S., the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act, or FDCA, and its implementing regulations, and biologics under the FDCA and the Public Health Service Act, or PHSA, and its implementing regulations. FDA approval is required before any new unapproved drug or biologic or dosage form, including a new use of a previously approved drug, can be marketed in the United States. Drugs and
37
Table of Contents
biologics are also subject to other federal, state, and local statutes and regulations. If we fail to comply with applicable FDA or other requirements at any time during the product development process, clinical testing, the approval process or after approval, we may become subject to administrative or judicial sanctions. These sanctions could include the FDA’s refusal to approve pending applications, license suspension or revocation, withdrawal of an approval, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, civil monetary penalties or criminal prosecution. Any FDA enforcement action could have a material adverse effect on us.
The process required by the FDA before product candidates may be marketed in the United States generally involves the following:
| • | completion of extensive preclinical laboratory tests and preclinical animal studies, all performed in accordance with the Good Laboratory Practices regulations; |
| • | submission to the FDA of an investigational new drug application, or IND, which must become effective before human clinical trials may begin and must be updated annually; |
| • | approval by an independent institutional review board, or IRB, or ethics committee representing each clinical site before each clinical trial may be initiated; |
| • | performance of adequate and well-controlled human clinical trials to establish the safety and efficacy of the product candidate for each proposed indication; |
| • | preparation of and submission to the FDA of a new drug application, or NDA, or biologics license application, or BLA, after completion of all pivotal clinical trials; |
| • | potential review of the product application by an FDA advisory committee, where appropriate and if applicable; |
| • | a determination by the FDA within 60 days of its receipt of an NDA or BLA to file the application for review; |
| • | satisfactory completion of an FDA pre-approval inspection of the manufacturing facilities where the proposed product is produced to assess compliance with current Good Manufacturing Practices, or cGMP; |
| • | a potential FDA audit of the preclinical research and clinical trial sites that generated the data in support of the NDA or BLA; and |
| • | FDA review and approval of an NDA or BLA prior to any commercial marketing or sale of the product in the U.S. |
The preclinical research and clinical testing and approval process require substantial time, effort, and financial resources, and we cannot be certain that any approvals for our product candidates will be granted on a timely basis, if at all.
An IND is a request for authorization from the FDA to administer an investigational new drug product to humans in clinical trials. The central focus of an IND submission is on the general investigational plan and the protocol(s) for human clinical trials. The IND also includes results of animal studies assessing the toxicology, pharmacokinetics, pharmacology, and pharmacodynamic characteristics of the product; chemistry, manufacturing, and controls information; and any available human data or literature to support the use of the investigational new drug. An IND must become effective before human clinical trials may begin. An IND will automatically become effective 30 days after receipt by the FDA, unless before that time the FDA raises
38
Table of Contents
concerns or questions related to the proposed clinical trials. In such a case, the IND may be placed on clinical hold and the IND sponsor and the FDA must resolve any outstanding concerns or questions before clinical trials can begin. Accordingly, submission of an IND may or may not result in the FDA allowing clinical trials to commence.
Clinical Trials
Clinical trials involve the administration of the investigational new drug to human subjects under the supervision of qualified investigators in accordance with Good Clinical Practices, or GCPs, which include the requirement that all research subjects provide their informed consent for their participation in any clinical trial. Clinical trials are conducted under protocols detailing, among other things, the objectives of the study, the parameters to be used in monitoring safety, and the efficacy criteria to be evaluated. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the FDA as part of the IND. Additionally, approval must also be obtained from each clinical trial site’s IRB or ethics committee, before the trials may be initiated, and the IRB or ethics committee must monitor the trial until completed. There are also requirements governing the reporting of ongoing clinical trials and clinical trial results to public registries.
The clinical investigation of a drug is generally divided into three or four phases. Although the phases are usually conducted sequentially, they may overlap or be combined.
| • | Phase I. The drug is initially introduced into healthy human subjects or patients with the target disease or condition. These studies are designed to evaluate the safety, dosage tolerance, metabolism and pharmacologic actions of the investigational new drug in humans, the side effects associated with increasing doses, and if possible, to gain early evidence on effectiveness. |
| • | Phase II. The drug is administered to a limited patient population to evaluate dosage tolerance and optimal dosage, identify possible adverse side effects and safety risks, and preliminarily evaluate efficacy. |
| • | Phase III. The drug is administered to an expanded patient population, generally at geographically dispersed clinical trial sites to generate enough data to statistically evaluate dosage, clinical effectiveness and safety, to establish the overall benefit-risk relationship of the investigational new drug product, and to provide an adequate basis for physician labeling. |
| • | Phase IV. In some cases, the FDA may condition approval of an NDA or BLA for a product candidate on the sponsor’s agreement to conduct additional clinical trials after approval. In other cases, a sponsor may voluntarily conduct additional clinical trials after approval to gain more information about the drug. Such post-approval studies are typically referred to as phase IV clinical trials. |
Clinical trial sponsors must also report to the FDA, within certain timeframes, serious and unexpected adverse reactions, any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator’s brochure, or any findings from other studies or animal testing that suggest a significant risk in humans exposed to the product candidate. The FDA, the IRB or ethics committee, or the clinical trial sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the research subjects are being exposed to an unacceptable health risk. Additionally, some clinical trials are overseen by an independent group of qualified experts organized by the clinical trial sponsor, known as a data safety monitoring board or committee. This group provides authorization for whether or not a trial may move forward at designated check points based on access to certain data from the trial.
The clinical trial process can take years to complete, and there can be no assurance that the data collected will support FDA approval or licensure of the product. Results from one trial are not necessarily predictive of results from later trials. We may also suspend or terminate a clinical trial based on evolving business objectives and/or competitive climate.
39
Table of Contents
Submission of an NDA or BLA to the FDA
Assuming successful completion of all required preclinical studies and clinical testing in accordance with all applicable regulatory requirements, detailed investigational new drug product information is submitted to the FDA in the form of an NDA or BLA requesting approval to market the product for one or more indications. Under federal law, the submission of most NDAs and BLAs is subject to an application user fee. For fiscal year 2015, the application user fee exceeds $2.3 million, and the sponsor of an approved NDA or BLA is also subject to annual product and establishment user fees, set at $110,370 per product and $569,200 per establishment. These fees are typically increased annually. Applications for orphan drug products are exempted from the NDA and BLA application user fee, unless the application includes an indication for other than a rare disease or condition, and may be exempted from product and establishment user fees under certain conditions.
An NDA or BLA must include all relevant data available from pertinent preclinical studies and clinical trials, including negative or ambiguous results as well as positive findings, together with detailed information relating to the product’s chemistry, manufacturing, controls, and proposed labeling, among other things. Data comes from company-sponsored clinical trials intended to test the safety and effectiveness of a use of a product, and may also come from a number of alternative sources, including trials initiated by investigators. To support marketing approval, the data submitted must be sufficient in quality and quantity to establish the safety and effectiveness of the investigational new drug product to the satisfaction of the FDA.
Once an NDA or BLA has been submitted, the FDA’s goal is to review the application within ten months after it accepts the application for filing, or, if the application relates to an unmet medical need in a serious or life-threatening indication, six months after the FDA accepts the application for filing. The review process is often significantly extended by the FDA’s requests for additional information or clarification.
Before approving an NDA or BLA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving an NDA or BLA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP.
The FDA is required to refer an NDA or BLA for a novel drug (in which no active ingredient has been approved in any other application) to an advisory committee or explain why such referral was not made. Typically, an advisory committee is a panel of independent experts, including clinicians and other scientific experts, that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
The FDA’s Decision on an NDA or BLA
After the FDA evaluates the NDA or BLA and conducts inspections of manufacturing facilities where the product will be produced, the FDA will issue either an approval letter or a Complete Response Letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. A Complete Response Letter indicates that the review cycle of the application is complete and the application is not ready for approval. In order to satisfy deficiencies identified in a Complete Response Letter, additional clinical data and/or an additional phase III clinical trial(s), and/or other significant, expensive and time-consuming requirements related to clinical trials, preclinical studies or manufacturing may be required for the product candidate. Even if such additional information is submitted, the FDA may ultimately decide that the NDA or BLA does not satisfy the criteria for approval. The FDA could also approve the NDA or BLA with a risk evaluation and mitigation strategy, plan to mitigate risks, which could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA also may condition approval on, among other things, changes to
40
Table of Contents
proposed labeling, development of adequate controls and specifications, or a commitment to conduct one or more post-market studies or clinical trials. Such post-market testing may include phase IV clinical trials and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization. New government requirements, including those resulting from new legislation, may be established, or the FDA’s policies may change, which could delay or prevent regulatory approval of our products under development.
Patent Term Restoration
Depending upon the timing, duration, and specifics of the FDA approval of the use of our product candidates, some of our U.S. patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, commonly referred to as the Hatch-Waxman Amendments. The Hatch-Waxman Amendments permit a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one-half the time between the effective date of an IND and the submission date of an NDA or BLA, plus the time between the submission date and the approval of that application. Only one patent applicable to an approved product is eligible for the extension and the application for the extension must be submitted prior to the expiration of the patent and within 60 days of the product’s approval. The U.S. Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we may apply for restoration of patent term for one of our currently owned or licensed patents to add patent life beyond its current expiration date, depending on the expected length of the clinical trials and other factors involved in the filing of the relevant NDA or BLA.
Companion Diagnostics
In its August 6, 2014, guidance document entitled “In Vitro Companion Diagnostic Devices,” the FDA defines an IVD companion diagnostic device to be an in vitro diagnostic device that provides information that is essential for the safe and effective use of a corresponding therapeutic product. Use of an IVD companion diagnostic device is considered essential when its use is required in the labeling of a therapeutic product, for example, to select appropriate patients for a product or those who should not use the product, or to monitor patients to achieve safety or effectiveness. In most circumstances, the IVD companion diagnostic device should be approved or cleared by FDA under the device authorities of the FDCA contemporaneously with the therapeutic product’s approval under section 505 of the FDCA for a drug or section 351 of the PHSA for a biological product. FDA expects the therapeutic product sponsor to address the need for an approved or cleared IVD companion diagnostic device in its therapeutic product development plan. The therapeutic product sponsor may develop its own IVD companion diagnostic device, partner with a diagnostic device sponsor to develop an IVD companion diagnostic device, or explore modifying an existing IVD diagnostic device to develop a new intended use. The FDA explains if a diagnostic device and a therapeutic device are studied together to support their respective approvals, both products can be studied in the same investigational study that meets both the requirements of the Investigational Device Exemption, or IDE, regulations and the IND regulations. Depending on the study plan and participants, a sponsor may seek to submit an IND alone, or both an IND and IDE.
Raw Materials, Manufacturing, and Supply
We have limited experience in manufacturing products for clinical or commercial purposes. We produce small quantities of SIRPaFc and CD200 monoclonal antibody in our laboratories for internal use. We believe that sources of raw materials pertinent to our laboratory operations and for manufacturing of our SIRPaFc product by our CMO are generally available.
We have established a contract manufacturing relationship for the supply of SIRPaFc that we believe will provide sufficient material for early clinical trials. In addition, we are establishing the basis for long-term commercial production capabilities. However, there can be no assurance that our contract manufacturer will be successful at scaling up and producing our product with the required quality and in the quantities and timelines that we will need for clinical and/or commercial purposes.
41
Table of Contents
We expect to similarly rely on contract manufacturing relationships for any products that we may further develop, or in-license or acquire in the future. However, there can be no assurance that we will be able to successfully contract with such manufacturers on terms acceptable to us, or at all.
Contract manufacturers are subject to ongoing periodic and unannounced inspections by the FDA, the U.S. Drug Enforcement Administration and corresponding state agencies to ensure strict compliance with cGMP and other state and federal regulations. We do not have control over third-party manufacturers’ compliance with these regulations and standards, other than through contractual obligations and periodic auditing. If they are deemed out of compliance with such regulations, approvals could be delayed, product recalls could result, inventory could be destroyed, production could be stopped and supplies could be delayed or otherwise disrupted.
If we need to change manufacturers after commercialization, the FDA and corresponding foreign regulatory agencies must approve these new manufacturers in advance, which will involve testing and additional inspections to ensure compliance with FDA regulations and standards and may require significant lead times and delay, and disruption of supply. Furthermore, switching manufacturers may be difficult because the number of potential manufacturers is limited. It may be difficult or impossible for us to find a replacement manufacturer quickly or on terms acceptable to us, or at all.
Property, Plant and Equipment
We operate from approximately 10,000 square feet of leased laboratory and office space at 96 Skyway Avenue, Toronto, Ontario, Canada, M9W 4Y9. We perform research and development in our facility and use qualified vendors and collaborators to conduct research and development and manufacturing on our behalf. We incur capital expenditures mainly for laboratory equipment, office equipment, computer equipment and leaseholds in the operation of our business. As at December 31, 2014 the net carrying value of our property and equipment was $235,402.
Employees
As at March 27, 2015, we had nineteen full-time employees including five senior management, twelve research and development staff and two finance and administrative staff. All employees are located at our head office and lab facilities in Toronto, Ontario, Canada.
We also use consultants and outside contractors to carry on many of our activities, including preclinical testing and validation, formulation, assay development, manufacturing, clinical and regulatory affairs, toxicology and clinical trials.
Legal Proceedings
To our knowledge, there have not been any legal or arbitration proceedings, including those relating to bankruptcy, receivership or similar proceedings, those involving any third party, and governmental proceedings pending or known to be contemplated, which may have, or have had in the recent past, significant effect our financial position or profitability.
Also, to our knowledge, there have been no material proceedings in which any director, any member of senior management, or any of our affiliates is either a party adverse to us or any of our subsidiaries or has a material interest adverse to us or any of our subsidiaries.
Recent Developments
Dr. Eric L. Sievers joins Trillium as the Chief Medical Officer, effective April 1, 2015.
On March 26, 2015, we incorporated a subsidiary in the U.S., Trillium Therapeutics USA Inc., through which we will employ Dr. Sievers.
42
Table of Contents
SELECTED CONSOLIDATED FINANCIAL INFORMATION
The following tables present selected financial data as at and for the fiscal years ended December 31, 2014, 2013, 2012, 2011, and 2010 prepared in accordance with IFRS, as issued by the IASB. The financial information in the tables below as at December 31, 2014, 2013 and 2012 and for the years then ended has been derived from our audited consolidated financial statements and related notes included in this prospectus. The financial information in the tables below as at December 31, 2011 and 2010 and for the years then ended has been derived from our audited consolidated financial statements and related notes for those years.
The selected financial data below should be read in conjunction with the financial statements included in this prospectus beginning on page F-1 of this prospectus and with the information appearing in the section of this prospectus entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations”. Our historical results do not necessarily indicate results expected for any future period.
Consolidated statement of loss and comprehensive loss data | Year ended December 31, | |||||||||||||||||||||||||||||
| 2014 | 2013 | 2012 | 2011 | 2010 | ||||||||||||||||||||||||||
| Net sales | $ | - | $ | - | $ | - | $ | - | $ | - | ||||||||||||||||||||
| Net loss and comprehensive loss | 12,881,820 | 4,289,308 | 1,061,502 | 3,173,947 | 4,305,867 | |||||||||||||||||||||||||
| Loss from continuing operations per share(1) | 3.06 | 3.16 | 1.71 | 5.22 | 8.24 | |||||||||||||||||||||||||
| Net loss per common share(1) | 3.06 | 3.16 | 1.71 | 5.22 | 8.24 | |||||||||||||||||||||||||
| Fully dilute net loss per common share(1) | 3.06 | 3.16 | 1.71 | 5.22 | 8.24 | |||||||||||||||||||||||||
Consolidated statement of financial position date | As at December 31, | |||||||||||||||||||
| 2014 | 2013 | 2012 | 2011 | 2010 | ||||||||||||||||
| Total assets | $ | 28,186,032 | $ | 35,087,386 | $ | 1,567,728 | $ | 2,763,580 | $ | 3,904,256 | ||||||||||
| Net assets | $ | 24,304,294 | $ | 33,908,447 | $ | 1,382,470 | $ | 2,409,466 | $ | 3,568,240 | ||||||||||
| Capital stock - common | $ | 49,505,792 | $ | 47,191,303 | $ | 31,388,959 | $ | 31,388,959 | $ | 30,212,225 | ||||||||||
| Number of common shares outstanding(2) | 4,427,244 | 4,058,408 | 622,060 | 622,060 | 561,121 | |||||||||||||||
| Capital stock - preferred | $ | 10,076,151 | $ | 11,292,525 | - | - | - | |||||||||||||
| Number of preferred shares outstanding(3) | 69,504,689 | 77,895,165 | - | - | - | |||||||||||||||
| Dividends declared per share | - | - | - | - | - | |||||||||||||||
Notes:
| (1) | The per share figures have been restated to reflect a share consolidation ratio of 1 post-consolidated common share for each 30 pre-consolidation common shares on November 14, 2014. |
| (2) | The number of common shares has been restated to reflect a share consolidation ratio of 1 post-consolidated common share for each 30 pre-consolidation common shares on November 14, 2014. |
| (3) | 30 preferred shares are convertible into one common share. |
43
Table of Contents
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF
OPERATIONS
You should read the following discussion of our financial condition and results of operations in conjunction with the financial statements and the notes thereto included elsewhere in this prospectus. The following discussion contains forward looking statements that reflect our plans, estimates and beliefs. Our actual results could differ materially from those discussed in the forward-looking statements. Factors that could cause or contribute to these differences include those discussed below and elsewhere in this prospectus, particularly those in the section of this prospectus entitled “Risk Factors.”
The following discussion and analysis of our financial condition and results of operations for the years ended December 31, 2014, 2013 and 2012, should be read in conjunction with our consolidated financial statements and related notes included in this prospectus. Our consolidated financial statements were prepared in accordance with IFRS as issued by the IASB. See the financial statements included as part of this prospectus and the notes thereto for a discussion of the significant accounting policies and significant estimates and judgments required to be made by management.
Operating Results
For the years ended December 31, 2014 and 2013
Net loss for the year ended December 31, 2014 of $12,881,820 exceeded the loss of $4,289,308 for 2013 due mainly to the inclusion of Trillium Privateco results for the whole fiscal year in 2014, including personnel and the SIRPaFc program costs, compared to approximately nine months in 2013, higher share-based compensation on the issuance of stock options and DSUs, higher professional fees, and due to the impairment loss on the return of the tigecycline rights to UHN, partially offset by higher interest income, higher tax credits and lower patent costs.
Research and Development
Components of research and development expenses for the years ended December 31, 2014 and 2013 were as follows:
| 2014 | 2013 | |||||||||||
| Research and development programs excluding the below items | $ | 5,893,030 | $ | 1,411,264 | ||||||||
Salaries, fees and short-term benefits | 2,311,755 | 1,299,055 | ||||||||||
Share-based compensation | 1,626,824 | 211,419 | ||||||||||
Amortization of intangible assets | 610,776 | 632,779 | ||||||||||
Impairment of intangible assets | 429,763 | - | ||||||||||
Depreciation of property and equipment | 47,208 | 16,010 | ||||||||||
Tax credits | (323,548 | ) | (233,821 | ) | ||||||||
|
|
|
| |||||||||
| $ | 10,595,808 | $ | 3,336,706 | |||||||||
|
|
|
| |||||||||
Our research and development expenses consist primarily of personnel-related costs, manufacturing and preclinical study services costs for external service providers, patent fees, share-based compensation and amortization of intangible assets.
The increase in research and development expenses for the year ended December 31, 2014 over the prior year was due mainly to the inclusion of Trillium Privateco in 2014 for the full fiscal year compared to just under nine months in 2013. Research and development program expenses increased for the year ended December 31, 2014
44
Table of Contents
due mainly to an increase in SIRPaFc expenses of $4,275,605 related to manufacturing, preclinical studies, and clinical and regulatory work in preparation for an expected IND filing in 2015. Program costs were also higher due to an increase in tigecycline costs of $283,260, and costs for our laboratory and office space for three additional months compared to 2013. Salaries, fees and short-term benefits increased due mainly to Trillium Privateco’s research and development staff for the full fiscal year in 2014 compared to nine months in 2013, as well as due to an increase in staff. Share-based compensation was higher due to a larger number of options issued in 2014 compared to 2013. Non-cash intangible asset related costs were higher due to the impairment loss of $429,763 in the second quarter of 2014 on the return of the tigecycline rights. Research and development tax credits recorded were $323,548 for the year ended December 31, 2014, commensurate with the higher level of development activity compared to 2013.
General and Administrative
Components of general and administrative expenses for the years ended December 31, 2014 and 2013 were as follows:
| 2014 | 2013 | |||||||||||||
| General and administrative expenses excluding the below items | $ | 1,198,181 | $ | 599,785 | ||||||||||
Salaries, fees and short-term benefits | 694,849 | 254,952 | ||||||||||||
DSU units issued for director compensation | 240,000 | - | ||||||||||||
Share-based compensation | 444,430 | 107,463 | ||||||||||||
|
|
|
| |||||||||||
| $ | 2,577,460 | $ | 962,200 | |||||||||||
|
|
|
| |||||||||||
General and administrative expenses consist mainly of professional fees, personnel costs related to corporate activities, directors’ fees, investor relations, stock exchange fees and share-based compensation.
General and administrative costs for the year ended December 31, 2014 were higher than the prior year due mainly to listing fees on graduation to the TSX of $162,000 and higher professional fees. Personnel costs increased due mainly to higher directors’ fees and full-time administrative staff in 2014. Share-based compensation was higher in 2014 due mainly to a larger number of options issued in 2014 and payment of directors’ fees in the form of DSUs in 2014 compared to no DSUs issued in 2013.
Finance income and costs
Finance income for the year ended December 31, 2014 was higher than the prior year due mainly to investment income earned on higher cash balances in 2014.
Finance costs for the year ended December 31, 2014 were higher than the prior year due mainly to interest accretion related to the loan payable and long-term liability.
For the years ended December 31, 2013 and 2012
Net loss for the year ended December 31, 2013 of $4,289,308 exceeded the loss of $1,061,502 for the year ended December 31, 2012 due mainly to higher personnel and facility costs with the inclusion of Trillium Privateco results from April 9, 2013 with a staff of 12 people, higher research and development program costs mainly due to the SIRPaFc program, higher professional fees and shareholder relations costs, higher amortization of intangible assets acquired with Trillium Privateco, and higher share-based compensation expenses.
45
Table of Contents
Research and Development
Components of research and development expenses for the years ended December 31, 2013 and 2012 were as follows:
| 2013 | 2012 | |||||||||||
Research and development programs, excluding the below | $ | 1,411,264 | $ | 518,030 | ||||||||
Salaries, fees and short-term benefits | 1,299,055 | 209,588 | ||||||||||
Share-based compensation | 211,419 | 38,011 | ||||||||||
Amortization of intangible assets | 632,779 | 2,000 | ||||||||||
Depreciation of property and equipment | 16,010 | 4,382 | ||||||||||
Net proceeds from patent interference settlement | - | (99,551 | ) | |||||||||
Tax credits | (233,821 | ) | (48,912 | ) | ||||||||
|
|
|
| |||||||||
| $ | 3,336,706 | $ | 623,548 | |||||||||
|
|
|
| |||||||||
The increase in research and development expenses for the year ended December 31, 2013 over the prior year was due mainly to the inclusion of the Trillium Privateco results from April 9, 2013. Program costs increased with preclinical and lab expenses for the SIRPaFc program of $402,558 compared to $139,110 in the prior year for legacy programs, higher patent fees of $230,126 due in part to a favorable patent interference settlement which provided gross proceeds of US$250,000 in Q4-2012 related to legacy programs (which lowered program expenses), and lab and office facility costs of $192,155 not in the prior year. Personnel costs increased due to Trillium Privateco’s research and development staff and amortization of intangible assets increased due to the Trillium Privateco and UHN intangible assets acquired in Q2-2013.
General and Administrative
Components of general and administrative expenses for the years ended December 31, 2013 and 2012 were as follows:
| 2013 | 2012 | |||||||||||
General and administrative, excluding the below | $ | 599,785 | $ | 269,849 | ||||||||
Salaries, fees and short-term benefits | 254,952 | 199,943 | ||||||||||
Share-based compensation | 107,463 | (3,505 | ) | |||||||||
|
|
|
| |||||||||
| $ | 962,200 | $ | 466,287 | |||||||||
|
|
|
| |||||||||
General and administrative costs for the year ended December 31, 2013 were higher than the comparable prior year due mainly to higher professional fees related to the Trillium Privateco merger and UHN license agreements, stock exchange and transfer agent fees related to the issuances of shares and warrants related to the merger and UHN license, higher share-based compensation expenses, costs of participating on the OTCQX International over-the-counter trading market, and slightly higher personnel costs.
Finance Income and Costs
Finance income for the year ended December 31, 2013 was higher than the prior year due mainly to investment income earned on the $33 million financing proceeds received in December 2013. Finance costs for the year ended December 31, 2013 were higher than the comparable period due mainly to interest accretion on the loan payable and long-term liability.
46
Table of Contents
Liquidity and Capital Resources
Since inception, we have financed our operations primarily from sales of equity, proceeds from the exercise of warrants and stock options, and from interest income on funds available for investment.
Our primary capital needs are for funds to support our scientific research and development activities including manufacturing, preclinical studies and clinical trials, administrative costs and for working capital.
We have experienced operating losses and cash outflows from operations since incorporation, will require ongoing financing in order to continue our research and development activities, and we have not earned significant revenue or reached successful commercialization of our products. Our future operations are dependent upon our ability to finance our cash requirements which will allow us to continue our research and development activities and the commercialization of our products. There can be no assurance that we will be successful in continuing to finance our operations.
We filed a base shelf prospectus with the British Columbia, Alberta, Manitoba, Ontario and Nova Scotia securities commissions in Canada on May 26, 2014 that provides that we may sell under the prospectus from time to time over the following 25 months up to $50 million, in one or more offerings, of common shares, First Preferred shares, warrants to purchase common shares, or units comprising a combination of common shares, First Preferred shares and/or warrants.
There are a significant number of warrants outstanding, some of which are in-the-money and may provide future liquidity. See “Description of Share Capital—Warrants”.
December 31, 2014 Compared to December 31, 2013
Our cash and marketable securities totaled $26,165,056 at December 31, 2014 compared to $32,983,104 at December 31, 2013. As at December 31, 2014, our working capital of $23,989,252 was lower compared to the amount of $32,772,281 at December 31, 2013 mainly due to cash used in operations for development programs. Accounts payable and accrued liabilities as at December 31, 2014 were $3,248,984 compared to $669,860 at December 31, 2013. The increase in accounts payable was due mainly to higher development costs, insurance and professional fees. Amounts receivable as at December 31, 2014 were $344,416 compared to $427,234 at December 31, 2013. The decrease in amounts receivable was due mainly to lower harmonized sales tax refunds expected in 2014.
We are indebted to the Federal Economic Development Agency for Southern Ontario, or FedDev, under a non-interest-bearing contribution agreement dated February 1, 2012. The period of contribution ended on December 31, 2013. As at December 31, 2014, we had repayable contributions of $555,968. The original principal balance is repayable in equal monthly installments for 60 months, which began on December 1, 2014. As at December 31, 2014 two installment payments have been made. The loan payable bears no interest. On the acquisition of Trillium Privateco, the fair value of the loan payable was determined by discounting the loan payable using an estimated market interest rate of 15%. Interest expense accretes on the discounted loan amount until it reaches its face value at maturity.
We have a long-term liability of $69,941 related to certain discontinued technologies. This liability has been discounted using an estimated market interest rate of 15% and interest expense is accreting.
As at December 31, 2014 and in the normal course of business, we had obligations to make future payments, representing research and development contracts and other commitments that are known and committed in the amount of $2,587,000 over the next 12 months, $99,000 from 12 to 24 months, $26,000 from 24-36 months, and $25,000 each year thereafter.
47
Table of Contents
We enter into research, development and license agreements in the ordinary course of business where we receive research services and rights to proprietary technologies. Milestone and royalty payments that may become due under various agreements are dependent on, among other factors, clinical trials, regulatory approvals and ultimately the successful development of a new drug, the outcome and timing of which is uncertain. Under the license agreement for SIRPaFc, we have future contingent milestones payable of $60,000 related to successful patent grants, and $100,000, $200,000 and $300,000 payable on the first patient dosed in phase I, II and III trials respectively. The regulatory milestone payments amount to $1 million on each of the submission of a first BLA in the U.S. and receipt of first regulatory approval in the U.S. and proportionate payments in other territories worldwide. The aggregate milestones payable on their first achievement under the agreement in the major markets of the U.S., Europe and Asia combined are $5,660,000. Royalties of either 3% or 1% of net revenues are due on commercial sales. Under the license agreement, Trillium is required to pay 20% of any sublicensing revenues to the licensors on the first $50 million of sublicensing revenues, and pay 15% of any sublicensing revenues to the licensors after the first $50 million of sublicensing revenue received.
We entered into two agreements with Catalent in August 2014 pursuant to which we acquired the right to use a proprietary expression system for the manufacture of two SIRPaFc constructs. Consideration for each license includes potential pre-marketing approval milestones of up to US$875,000 and aggregate sales milestone payments of up to US$28.8 million.
We periodically enter into research and license agreements with third parties that include indemnification provisions customary in the industry. These guarantees generally require us to compensate the other party for certain damages and costs incurred as a result of claims arising from research and development activities undertaken by or on our behalf. In some cases, the maximum potential amount of future payments that could be required under these indemnification provisions could be unlimited. These indemnification provisions generally survive termination of the underlying agreement. The nature of the indemnification obligations prevents us from making a reasonable estimate of the maximum potential amount it could be required to pay. Historically, we have not made any indemnification payments under such agreements and no amount has been accrued in our financial statements with respect to these indemnification obligations.
December 31, 2013 Compared to December 31, 2012
Our cash and marketable securities totaled $32,983,104 at December 31, 2013 compared to $1,375,015 at December 31, 2012. The increase in our cash resources resulted from two financings in 2013 where we raised gross proceeds of $36 million.
As at December 31, 2013 our working capital was $32,772,281. Accounts payable and accrued liabilities as at December 31, 2013 were $669,860 compared to $185,258 at December 31, 2012. The increase in accounts payable was due mainly to the addition of the Trillium Privateco operations.
Research and Development, Patents and Licenses, etc.
During 2013, we refocused our product pipeline through the strategic acquisition of Trillium Privateco on April 9, 2013, which included the SIRPaFc and CD200 monoclonal antibody preclinical cancer programs. On April 16, 2013, we completed a license agreement with UHN to gain rights to intellectual property related to the use of tigecycline for the treatment of leukemia. In August 2014, we returned these rights to UHN and expanded our SIRPaFc research and development program to include preclinical work on additional cancer indications and combinations with other therapies. Substantially all of our research and development resources are focused on the SIRPaFc program.
Prior to the April 2013 merger with Trillium Privateco, we were developing regenerative stem cell products and sought new technologies to develop. In July 2013, we ceased all activities associated with our pre-merger regenerative medicine neurology programs and abandoned all related intellectual property filings.
48
Table of Contents
We have also determined not to pursue our TTI-1612 project. TTI-1612 is a soluble form of heparin-binding epidermal-growth-factor-like growth factor, or HB-EGF. HB-EGF is a naturally occurring protein that triggers cell growth and stimulates wound healing. We completed a 28-patient phase I trial in 2013 designed to assess the safety and tolerability of single ascending doses of TTI-1612 in interstitial cystitis/bladder pain syndrome, or IC/BPS, patients. IC/BPS is a chronic bladder disease characterized by low urinary HB-EGF levels and a dysfunctional, “leaky” bladder epithelium. Drug levels and changes in disease symptoms were also evaluated. While clinical data indicated that TTI-1612 was well tolerated and exhibited a favorable drug absorption profile, our TTI-1612 project is outside of our core interest in immuno-oncology. Accordingly, we have allowed our license for HB-EGF to lapse and we do not intend to renew our intellectual property associated with the project.
Research and development expenditures for the three years ended December 31, 2014, 2013 and 2012 were as follows:
| Year ended December 31, | ||||||||||||||||||||||||
| 2014 | 2013 | 2012 | ||||||||||||||||||||||
| SIRPaFc | $ | 9,372,467 | $ | 1,913,051 | $ | - | ||||||||||||||||||
| Tigecycline | 1,091,297 | 642,879 | - | |||||||||||||||||||||
| TTI-1612 | 44,844 | 328,459 | - | |||||||||||||||||||||
| Regenerative medicine programs discontinued in 2013 | - | 409,278 | 623,548 | |||||||||||||||||||||
| Other programs | 87,200 | 43,039 | - | |||||||||||||||||||||
|
|
|
|
|
| |||||||||||||||||||
Total(1) | $ | 10,595,808 | $ | 3,336,706 | $ | 623,548 | ||||||||||||||||||
|
|
|
|
|
| |||||||||||||||||||
Note:
| (1) | Research and development expenditures in the above table include all direct and indirect costs for the programs, personnel costs, intellectual property related costs net of recoveries, share-based compensation and research and development overhead, and is net of government assistance. |
We rely on patents and licenses to enable the commercialization of our novel technologies. See the section of this prospectus entitled “Business – Intellectual Property”.
Trend Information
Historical patterns of expenditures cannot be taken as an indication of future expenditures. The amount and timing of expenditures and therefore liquidity and capital resources vary substantially from period to period depending on the number of research and development programs being undertaken at any one time, the stage of the development programs, the timing of significant expenditures for manufacturing, toxicology and pharmacology studies and clinical trials, and the availability of funding from investors and prospective commercial partners.
Expenses for the three months ended March 31, 2013 were lower due to low development activity levels while we sought new products for development. Subsequent to the April 9, 2013 Trillium Privateco merger, expenses increased significantly as we acquired twelve personnel and an approximately 10,000 square foot leased laboratory and office facility, and thereafter licensed additional technology assets from UHN. In December 2013, we completed a private placement raising gross proceeds of $33 million to advance our SIRPaFc program through phase I clinical trials. In 2014, we advanced the manufacturing and preclinical development of our SIRPaFc program. Management expects that 2015 expenditures will increase from 2014 levels as we expect to advance our SIRPaFc program through IND-enabling studies, complete manufacturing for and initialize the phase I clinical trial, hire additional personnel, and carry out an expanded preclinical program that includes additional cancer indications and combination studies.
49
Table of Contents
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements that have, or are reasonably likely to have, a current or future effect on our financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that are material to investors.
Tabular Disclosure of Contractual Obligations
Other than as disclosed below, we did not have any contractual obligations as at December 31, 2014 relating to long-term debt obligations, capital (finance) lease obligations, operating lease obligations, purchase obligations or other long-term liabilities reflected on our balance sheet as at December 31, 2014:
Contractual Obligations(1)(2) | Payment due by period | |||||||||||||||||||||||||||||
| Total | Less than 1 year | 1 to 3 years | 3 to 5 years | More than 5 years | ||||||||||||||||||||||||||
| Long-Term Debt Obligations(3) | $ | 555,968 | $ | 105,446 | $ | 230,064 | $ | 220,458 | $ | - | ||||||||||||||||||||
| Capital (Finance) Lease Obligations | - | - | - | - | - | |||||||||||||||||||||||||
| Operating Lease Obligations(4) | 130,768 | 78,746 | 52,022 | - | - | |||||||||||||||||||||||||
| Purchase Obligations(5) | 2,279,518 | 2,279,518 | - | - | - | |||||||||||||||||||||||||
| Other Long-Term Liabilities Reflected on our Balance Sheet(6) | 232,020 | 58,005 | 174,015 | - | - | |||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
| $ | 3,198,274 | $ | 2,521,715 | $ | 456,101 | $ | 220,458 | $ | - | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
| |||||||||||||||||||||
Notes:
| (1) | Contractual obligations in the above table do not include amounts in accounts payable and accrued liabilities on our balance sheet as at December 31, 2014. Annual technology license fees currently approximating $50,000 are not included in the above table. |
| (2) | We are party to a license agreement for our SIRPaFc technology with UHN and HSC that has future milestones where the certainty and timing of reaching the milestones are unknown and are not included in the above table. Aggregate milestones under this agreement, related to major markets on their first achievement, are $5,660,000 of which management estimates that $360,000 may occur in 1 to 3 years, $300,000 may occur in 3 to 5 years and the balance more than five years, if the milestones are reached at all. |
| (3) | Amounts due to FedDev repayable in equal monthly installments for 60 months beginning on December 1, 2014. |
| (4) | Operating lease obligations for laboratory and office facilities currently expire in August 2016. |
| (5) | Long-term liability related to the cessation of the development of our regenerative medicine products. |
We entered into two agreements on August 12, 2014 with Catalent pursuant to which we acquired the right to use two of Catalent’s proprietary GPEx® expression cell lines for the manufacture of SIRPaFc. In consideration for the purchase of the expression cell lines, we will pay Catalent up to US$875,000 upon reaching certain pre-marketing approval milestones and up to an additional US$28.8 million for reaching certain sales milestones.
50
Table of Contents
Quantitative & Qualitative Disclosures About Market Risk
Fair Value
IFRS 13,Fair Value Measurement, provides a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable or unobservable. Observable inputs are those which reflect market data obtained from independent sources, while unobservable inputs reflects our assumptions with respect to how market participants would price an asset or liability. These two inputs used to measure fair value fall into the following three different levels of the fair value hierarchy:
Level 1 Quoted prices in active markets for identical instruments that are observable.
Level 2 Quoted prices in active markets for similar instruments; inputs other than quoted prices that are observable and derived from or corroborated by observable market data.
Level 3 Valuations derived from valuation techniques in which one or more significant inputs are unobservable.
The hierarchy requires the use of observable market data when available.
We have classified cash and marketable securities as Level 1. The loan payable has been classified as Level 2.
Cash, marketable securities, amounts receivable, accounts payable and accrued liabilities, and other current liabilities, due within one year, are all short-term in nature and, as such, their carrying values approximate fair values. The fair value of the non-current loan payable and long-term liability is estimated by discounting the expected future cash flows at the cost of money to us, which is equal to its carrying value.
Risks
We are exposed to credit risk, liquidity risk, interest rate risk and currency risk. Our Board of Directors has overall responsibility for the establishment and oversight of our risk management framework. The Audit Committee is responsible for reviewing our risk management policies.
Credit risk
Credit risk is the risk of financial loss to us if a counterparty to a financial instrument fails to meet its contractual obligations, and arises principally from our cash and cash equivalents and amounts receivable. The carrying amount of these financial assets represents the maximum credit exposure. We follow an investment policy to mitigate against the deterioration of principal and to enhance our ability to meet its liquidity needs. Cash and cash equivalents are on deposit with a major Canadian chartered bank. Amounts receivable are primarily comprised of amounts due from the federal government.
Liquidity risk
Liquidity risk is the risk that we will not be able to meet its financial obligations as they fall due. We are a development stage company and are reliant on external fundraising to support its operations. Once funds have been raised, we manage our liquidity risk by investing in cash and cash equivalents to provide regular cash flow for current operations. We also manage liquidity risk by continuously monitoring actual and projected cash flows. The Board of Directors reviews and approves our operating and capital budgets, as well as any material transactions not in the ordinary course of business. The majority of our accounts payable and accrued liabilities have maturities of less than three months.
51
Table of Contents
Interest rate risk
Interest rate risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market interest rates. We hold our cash in bank accounts or high interest savings accounts which have a variable rate of interest. We manage our interest rate risk by holding highly liquid short-term instruments and by holding our investments to maturity, where possible. For the years ended December 31, 2014, 2013 and 2012, we earned interest income of $378,692, $44,113 and $25,740, respectively. Therefore, a 1% change in the average interest rate for the years ended December 31, 2014, 2013 and 2012, would have a net impact on finance income of $3,787, $441 and $257, respectively.
Currency risk
We are exposed to currency risk related to the fluctuation of foreign exchange rates and the degree of volatility of those rates. Currency risk is limited to the portion of our business transactions denominated in currencies other than the Canadian dollar which are primarily expenses in U.S. dollars. As at December 31, 2014, 2013 and 2012, we held U.S. dollar cash in the amount of US$142,558, US$493,031 and US$157,383, respectively, and had U.S. dollar denominated accounts payable and accrued liabilities in the amount of US$1,910,430, US$88,063 and US$30,396, respectively. Therefore, a 1% change in the foreign exchange rate would have had a net impact on finance costs of $17,679, $4,050 and $1,270 as at December 31, 2014, 2013 and 2012, respectively.
U.S. dollar expenses for the years ended December 31, 2014, 2013 and 2012 were approximately US$3,260,000, US$518,000 and US$532,000, respectively. Varying the US exchange rate for the year to reflect a 5% strengthening of the Canadian dollar would have decreased the net loss by approximately $163,000, $26,000 and $27,000 for the three years ended December 31, 2014, 2013 and 2012, respectively, assuming that all other variables remained constant.
Significant Changes
We are not aware of any significant change that has occurred since December 31, 2014 included in this prospectus and that has not been disclosed elsewhere in this prospectus.
Implications of Being an Emerging Growth Company
We are an “emerging growth company” under the U.S. Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and will continue to qualify as an “emerging growth company” until the earliest to occur of: (a) the last day of the fiscal year during which we have total annual gross revenues of $1,000,000,000 (as such amount is indexed for inflation every 5 years by the United States Securities and Exchange Commission, or SEC) or more; (b) the last day of our fiscal year following the fifth anniversary of the date of the first sale of our common shares pursuant to an effective registration statement under the Securities Act; (c) the date on which we have, during the previous 3-year period, issued more than $1,000,000,000 in non-convertible debt; or (d) the date on which we are deemed to be a “large accelerated filer”, as defined in Rule 12b–2 of the Exchange Act.
Generally, a company that registers any class of its securities under Section 12 of the Exchange Act is required to include in the second and all subsequent annual reports filed by it under the Exchange Act, a management report on internal control over financial reporting and, subject to an exemption available to companies that meet the definition of a “smaller reporting company” in Rule 12b-2 under the Exchange Act, an auditor attestation report on management’s assessment of the company’s internal control over financial reporting. However, for so long as we continue to qualify as an emerging growth company, we will be exempt from the requirement to include an auditor attestation report in our annual reports filed under the Exchange Act, even if we do not qualify as a “smaller reporting company”. In addition, Section 103(a)(3) of the Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act, has been amended by the JOBS Act to provide that, among other things, auditors of an emerging growth company are exempt from any rules of the Public Company Accounting Oversight Board requiring
52
Table of Contents
mandatory audit firm rotation or a supplement to the auditor’s report in which the auditor would be required to provide additional information about the audit and the financial statements of the company.
Any U.S. domestic issuer that is an emerging growth company is able to avail itself of the reduced disclosure obligations regarding executive compensation in periodic reports and proxy statements, and to not present to its shareholders a non-binding advisory vote on executive compensation, obtain approval of any golden parachute payments not previously approved, or present the relationship between executive compensation actually paid and our financial performance. So long as we are a foreign private issuer, we are not subject to such requirements, and will not become subject to such requirements even if we were to cease to be an emerging growth company.
As a reporting issuer under the securities legislation of the Canadian provinces of Ontario, British Columbia, Manitoba, Nova Scotia and Alberta, we are required to comply with all new or revised accounting standards that apply to Canadian public companies. Pursuant to Section 107(b) of the JOBS Act, an emerging growth company may elect to utilize an extended transition period for complying with new or revised accounting standards for public companies until such standards apply to private companies. We have elected not to utilize this extended transition period.
53
Table of Contents
The table below sets forth our total indebtedness and shows our capitalization as at December 31, 2014:
| • | on an actual basis; and |
| • | on an as adjusted basis, to give further effect to the issuance and sale of common shares at a price of US$ per share and Series II First Preferred Shares at a price of US$ per share, after deducting underwriting discounts and estimated offering expenses. We also assumed that the exchange rate for Canadian dollars expressed in United States dollars of the offering was $1.00 = US$0.7949 (the noon exchange rate for one Canadian dollar expressed in United States dollars as reported by the Bank of Canada on March 27, 2015). |
You should read this table in conjunction with our consolidated financial statements included in this prospectus, together with the accompanying notes and the other information appearing in the sections of this prospectus entitled “Selected Consolidated Financial Information” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.”
| As at December 31, 2014 | ||||||||
| (actual) | (as adjusted) | |||||||
Liabilities | ||||||||
Accounts payable and accrued liabilities | $ | 3,248,984 | $ | 3,248,984 | ||||
Other current liabilities(1) | 279,461 | 279,461 | ||||||
Total current liabilities | 3,528,445 | 3,528,445 | ||||||
Loan payable(2) | 283,352 | 283,352 | ||||||
Long-term liability(3) | 69,941 | 69,941 | ||||||
Total non-current liabilities | 353,293 | 353,293 | ||||||
Equity | ||||||||
Common shares(4) | 49,505,792 | |||||||
Preferred shares(4) | 10,076,151 | |||||||
Warrants(4) | 9,283,332 | 9,283,332 | ||||||
Contributed surplus(5)(6) | 5,995,055 | 5,995,055 | ||||||
Deficit | (50,556,036 | ) | (50,556,036 | ) | ||||
Total equity | 24,304,294 | |||||||
Notes:
| (1) | The current portions of the Loan payable and Long-term liability are included in Other current liabilities. |
| (2) | We are indebted under a contribution agreement with FedDev. The period of contribution ended on December 31, 2013. As at December 31, 2014, we have repayable contributions of $555,968. The original principal balance is repayable in equal monthly installments for 60 months, which began on December 1, 2014. As at December 31, 2014 two installment payments have been made. The loan payable bears no interest. On acquisition of Trillium Privateco in April 2013, the fair value of the loan payable was determined by discounting the loan payable using an estimated market interest rate of 15%. Interest expense accretes on the discounted loan amount until it reaches its face value at maturity. |
| (3) | We have a long-term liability of $69,941 related to certain discontinued technologies. This liability has been discounted using an estimated market interest rate of 15% and interest expense is accreting. |
| (4) | As at December 31, 2014, 4,427,244 common shares, 69,504,689 Series I First Preferred Shares, and 138,724,781 common share purchase warrants were outstanding. |
| (5) | As at December 31, 2014, there were 590,141 stock options outstanding. Share-based compensation expense recognized as stock options vest is recorded in contributed surplus. |
| (6) | As at December 31, 2014, there were 28,777 DSUs issued. Share-based compensation expense recorded on the issuance of DSUs is recorded in contributed surplus. |
54
Table of Contents
The preceding table takes into account the consolidation of our common shares in a ratio of one post-consolidated common share for each 30 pre-consolidation common shares on November 14, 2014. The consolidation resulted in the following changes to our equity:
| • | the number of common shares decreased by a factor of 30; |
| • | the number of Series I First Preferred Shares was unchanged but the conversion ratio changed such that each preference share is convertible into 1/30th of a common share; |
| • | the number of DSUs decreased by a factor of 30; |
| • | the number of warrants was unchanged but the conversion ratio changed such that 30 warrants are needed to purchase one common share and the exercise price increased by a factor of 30; and |
| • | the number of stock options decreased by a factor of 30 and the exercise price increased by a factor of 30. |
55
Table of Contents
If you invest in our common shares and Series II First Preferred Shares in this offering, your ownership interest will be immediately diluted to the extent of the difference between the public offering price per share and the pro forma net tangible book value per share after this offering.
Our historical net tangible book value as of December 31, 2014 was approximately $23.9 million, or $5.39 per common share. Our historical net tangible book value is the amount of our total tangible assets less our liabilities. Net historical tangible book value per share is our historical net tangible book value divided by the number of common shares outstanding as of December 31, 2014. Our pro forma net tangible book value as of December 31, 2014 was $23.9 million, or $3.54 per common share. Pro forma net tangible book value (deficit) gives effect to the conversion of all of our outstanding Series I First Preferred Shares as of December 31, 2014, into an aggregate of 2,316,823 common shares.
Pro forma as adjusted net tangible book value is our pro forma net tangible book value (deficit), plus the effect of the sale of common shares in this offering at a public offering price of US$ per share and of Series II First Preferred Shares in this offering at a public offering price of US$ per share, and after deducting underwriting discounts and estimated offering expenses payable by us (assuming full conversion of the Series II First Preferred Shares in this offering into common shares). This amount represents an immediate increase in pro forma as adjusted net tangible book value of approximately $ per share to our existing shareholders, and an immediate dilution of approximately $ per share to new investors participating in this offering.
The following table illustrates this dilution on a per share basis:
Public offering price per share | US$ | |||||||||
Historical net tangible book value (deficit) per share as of December 31, 2014 | $ | ( | ) | |||||||
Pro forma increase in net tangible book value per share as of December 31, 2014 attributable to the conversion of Series II First Preferred Shares | ||||||||||
|
| |||||||||
Pro forma net tangible book value per share as of December 31, 2013, before giving effect to this offering | ( | ) | ||||||||
Increase in pro forma net tangible book value per share attributable to new investors purchasing our common shares (and our Series II First Preferred Shares on an as converted basis per common share) in this offering | ||||||||||
|
| |||||||||
Pro forma as adjusted net tangible book value per share after this offering | ______ | |||||||||
Dilution per share to new investors purchasing our common shares (and our Series II First Preferred Shares on an as converted basis per common share) in this offering | US$ |
If the underwriters exercise their option to purchase additional common shares in full, the pro forma as adjusted net tangible book value will increase to $ per share, (assuming full conversion of the Series II First Preferred Shares in this offering into common shares) representing an immediate dilution of US$ per share to new investors purchasing our common shares (and our Series II First Preferred Shares on an as converted basis per common share) in this offering.
The foregoing discussion is based on 4,427,244 common shares outstanding as of December 31, 2014, and excludes:
| • | 69,504,689 Series I First Preferred Shares; |
| • | 138,724,781 common share purchase warrants; |
| • | 590,141 stock options; and |
| • | 28,777 DSUs. |
Furthermore, we may choose to raise additional capital through the sale of equity or convertible debt securities due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans. To the extent that any of these options are exercised, new options or DSUs are issued under our equity incentive plans or we issue additional common shares or other equity or convertible debt securities in the future, there will be further dilution to investors participating in this offering.
56
Table of Contents
BOARD OF DIRECTORS AND MANAGEMENT
The following table and summary of business experience set forth the name, office held, and functions and areas of experience in the Company, principal business activities and other principal directorships of each of our directors and senior management:
Name Position Held Since | Principal Business Activities, Other Principal Directorships and Function | |
Luke Beshar Director(1)
March 10, 2014 | Mr. Beshar was the Executive/Senior Vice President and Chief Financial Officer of NPS Pharmaceuticals, Inc., a global biopharmaceutical company from November 2007 to February 2015.
As an independent director, Mr. Beshar supervises our management and helps to ensure compliance with our corporate governance policies and standards. | |
Henry Friesen Director(1)(2)
June 28, 2011 | Dr. Friesen is a Distinguished University Professor Emeritus at University of Manitoba since October 2000.
As an independent director, Dr. Friesen supervises our management and helps to ensure compliance with our corporate governance policies and standards. | |
Robert Kirkman Director(1) (3)
December 17, 2013 | Dr. Kirkman is President and Chief Executive Officer and director of Oncothyreon Inc., an oncology-focused biotechnology company since September 2006.
As an independent director, Dr. Kirkman supervises our management and helps to ensure compliance with our corporate governance policies and standards. | |
Michael Moore Director(2)(3)
April 9, 2013 | Dr. Moore is the Executive Chair ofMISSIONTherapeutics Ltd. since 2012 and a Board Chair or Director of several private biopharmaceutical companies in the UK. From 2004 to 2013, Dr. Moore was the Chair of Trillium Privateco, and from 2003 to 2008, Dr. Moore was the Chief Executive Officer and Director of PIramed Ltd, a UK- based oncology company sold to Roche in 2008.
As an independent director, Dr. Moore supervises our management and helps to ensure compliance with our corporate governance policies and standards. | |
Thomas Reynolds Director(2)(3)
March 10, 2014 | Dr. Reynolds is an independent biotechnology consultant since February 2013, and was Chief Medical Officer of Seattle Genetics, Inc., a biotechnology company focused on antibody-based therapies for the treatment of cancer from March 2007 to January 2013. Dr. Reynolds also sits on the board of MEI Pharma, Inc.
As an independent director, Dr. Reynolds supervises our management and helps to ensure compliance with our corporate governance policies and standards. | |
Calvin Stiller Director, Chair of the Board
July 18, 2011 | Dr. Stiller is the Chair of Ontario Institute for Cancer Research and Professor Emeritus at Western University. Dr. Stiller also sits on the board of Revera Corporation and Magor Corporation.
As an independent director, Dr. Stiller supervises our management and helps to ensure compliance with our corporate governance policies and standards. |
57
Table of Contents
Name Position Held Since | Principal Business Activities, Other Principal Directorships and Function | |
Niclas Stiernholm President and Chief Executive Officer, Director
Director since July 18, 2011; President and CEO since April 9, 2013 | Dr. Stiernholm is the President and Chief Executive Officer of Trillium since April 9, 2013 and was the President and Chief Executive Officer of Trillium Privateco since 2002. He joined Trillium from YM BioSciences where he was Executive Vice President and Chief Scientific Officer.
As President and Chief Executive Officer, Dr. Stiernholm is responsible for overseeing our strategic direction, executing business development plans and ensuring that our scientific programs remain funded and advance on schedule. As a director, Dr. Stiernholm participates in management oversight and helps to ensure compliance with our corporate governance policies and standards. | |
Robert Uger Chief Scientific Officer
April 9, 2013 | Dr. Uger is the Chief Scientific Officer of Trillium since April 9, 2013 and was the Vice President, Research of Trillium Privateco since 2003. He joined Trillium from Aventis Pasteur where he was a Senior Research Scientist involved in cancer vaccine research.
As Chief Scientific Officer, Dr. Uger is responsible for developing and implementing our scientific direction, and oversees both internal product development and external research and development programs. | |
Eric L. Sievers Chief Medical Officer
April 1, 2015 | Dr. Sievers is the Chief Medical Officer of Trillium effective April 1, 2015. He joins Trillium from Seattle Genetics where he was Senior Vice President, Clinical Development.
As Chief Medical Officer, Dr. Sievers is responsible for evaluating and implementing clinical development strategies for advancement and approval of our clinical programs, with initial emphasis on our SIRPaFc development program. | |
James Parsons Chief Financial Officer
August 25, 2011 | Mr. Parsons is the Chief Financial Officer of Trillium since August 25, 2011 and was also the Director, Finance of Trillium Privateco. He was previously the Vice President, Finance of DiaMedica Inc. from October 2010 to May 2014, and Chief Financial Officer of Amorfix Life Sciences Ltd. from 2006 to 2010. Mr. Parsons sits on the board of Sernova Corp.
As Chief Financial Officer, Mr. Parsons is responsible for financial and risk management, investor relations, corporate governance and administration. | |
Penka Petrova Vice-President, Drug Development
April 9, 2013 | Dr. Petrova is the Vice President, Drug Development of Trillium since April 9, 2013 and was the Director, Drug Development of Trillium Privateco from June 2011 to March 2013. Dr. Petrova joined Trillium Privateco from Prescient Neuropharma in 2003.
As Vice President, Drug Development, Dr. Petrova is responsible for managing our formal drug development efforts, including all outsourced activities to contract manufacturers and contract research organizations. |
Notes:
| (1) | Member of our audit committee. |
| (2) | Member of our corporate governance and nominating committee. |
| (3) | Member of our compensation committee. |
58
Table of Contents
The business address for our directors and senior management is Trillium Therapeutics Inc., 96 Skyway Avenue, Toronto, Ontario, Canada, M9W 4Y9.
Summary of Business Experience and Functions within the Company
Luke Beshar, CPA -Director, Chair of the Audit Committee
Mr. Beshar was Executive Vice President and Chief Financial Officer of NPS Pharmaceuticals until February 2015 when the company was sold to Shire plc. He joined NPS Pharmaceuticals in 2007 and has been responsible for financial management, investor relations, information technology, technical operations, supply-chain management, facilities, project management, contracts and outsourcing and strategic and alliance management. Prior to joining NPS, Mr. Beshar served as Executive Vice President and Chief Financial Officer of Cambrex Corporation, a global life sciences company. Mr. Beshar began his career with Arthur Andersen & Co. and is a certified public accountant.
He obtained his bachelor’s degree in Accounting and Finance from Michigan State University and is a graduate of The Executive Program at the Darden Graduate School of Business at the University of Virginia.
Dr. Henry Friesen -Director
Dr. Friesen was the President of the Canadian Government’s Medical Research Council, and the architect and lead champion for the creation the Canadian Institutes for Health Research, President of the National Cancer Institute of Canada and President of the Canadian Society for Clinical Investigation. He is the Past Founding Chair of Genome Canada. A Fellow of the Royal Society of Canada, Dr. Friesen was named a Companion of the Order of Canada and was inducted into the Canadian Medical Hall of Fame in 2001 and, later the Order of Manitoba. He was also awarded the Gairdner Foundation Wightman Award, the McLaughlin Medal of the Royal Society of Canada, and the Koch Medal, the highest award of the Endocrine Society. He was presented with the Frederic Newton Gisborne Starr Award by the Canadian Medical Association, the association’s highest award, in 2006. Dr. Friesen also holds eight Honorary Doctorates from Canadian universities.
Dr. Robert Kirkman -Director
Dr. Kirkman has served as Oncothyreon’s President and Chief Executive Officer since September 2006. From 2005 to 2006, he was acting President and Chief Executive Officer of Xcyte Therapies, which concluded a merger with Cyclacel Pharmaceuticals, both development-stage biopharmaceutical companies, in March of 2006. From 2004 to 2005, Dr. Kirkman was Chief Business Officer and Vice President of Xcyte. From 1998 to 2003, Dr. Kirkman was Vice President, Business Development and Corporate Communications of Protein Design Labs, a biopharmaceutical company. Dr. Kirkman holds a M.D. degree from Harvard Medical School and a B.A. in economics from Yale University.
Dr. Michael Moore -Director
Dr. Moore is currently Executive Chairman ofMISSIONTherapeutics Limited, a UK drug discovery company targeting deubiquitinating enzymes for multiple disease indications. He also holds non-executive positions with other UK biopharmaceutical companies. From 2004 to 2013, Dr. Moore was non-executive chair of Trillium Privateco and from 2003-2008 Chief Executive Officer and Director of PIramed Ltd, a UK-based biotechnology company developing small molecule drugs against the PI-3 kinase superfamily, which was acquired by Roche. Prior to PIramed, he held several progressive positions at Xenova Group plc (1988-2003), including Chief Scientific Officer and Research Director. Dr. Moore held a tenured Cancer Research Campaign position (1980) at the Paterson Institute for Cancer Research, was Honorary Reader in Immunology and Oncology at the University of Manchester Medical School (1986), and Editor-in-Chief of the British Journal of Cancer (1983). Dr. Moore
59
Table of Contents
received his PhD (Faculty of Pure Science) and DSc (Faculty of Medicine) from Nottingham University and has more than 150 scientific publications.
Dr. Thomas Reynolds -Director
Dr. Reynolds served as Chief Medical Officer of Seattle Genetics from March 2007 until his retirement in February, 2013. While at Seattle Genetics, he was responsible for building and leading an integrated clinical development, regulatory and medical affairs organization, highlighted by the development and approval of ADCETRIS. From 2002 to 2007, Dr. Reynolds served at ZymoGenetics (acquired by Bristol-Myers Squibb in 2010), most recently as Vice President, Medical Affairs, where he oversaw the clinical development and regulatory filing of RECOTHROM. Previously, he was Vice President, Clinical Affairs at Targeted Genetics, and before that he was at Somatix Therapy (acquired by Cell Genesys in 1997). Dr. Reynolds received his M.D., and Ph.D. in Biophysics, from Stanford University and a B.A. in Chemistry from Dartmouth College.
Dr. Calvin Stiller -Director, Chair of the Board of Directors
Dr. Stiller is a Member of the Order of Canada and the Order of Ontario, was the recipient of the Canada Gairdner Wightman Award in 2011 (awarded to a Canadian who has demonstrated outstanding leadership in medicine and medical science) and was inducted into the Canadian Medical Hall of Fame in 2010. Dr. Stiller is chair of the Ontario Institute for Cancer Research, the former chair of Genome Canada and is Professor Emeritus in the Departments of Medicine, and Immunology and Bacteriology at the University of Western Ontario. Dr. Stiller founded the J. Allyn Taylor International Prize in Medicine, co-founded the Medical and Related Sciences Research District, was the Chair of the Ontario Research and Development Challenge Fund Board and was the co-founder of four venture capital funds of over $500 million. He serves on the boards of a number of private and public companies, was founding Chair of Trillium Privateco and was chair of Verio Therapeutics, a Canadian stem cell company that was acquired in 2010 by Fate Corporation, a California-based regeneration company. Together with Robert Klein (the founder of the California Institute of Regenerative Medicine, a state agency responsible for granting approximately $3 billion in stem cell research funding), he co-founded the Cancer Stem Cell Initiative, a Canada-California consortium that has been productive in the search for and identification of cancer stem cells. He serves on the board of Revera Corporation, one of the nation’s largest seniors’ accommodation, health and long-term care and services companies.
Dr. Niclas Stiernholm -President and Chief Executive Officer, Director
Dr. Stiernholm became the President and Chief Executive Officer on our merger with Trillium Privateco in April 2013. Previously, as President and Chief Executive Officer of Trillium Privateco since 2002, Dr. Stiernholm spearheaded the in-licensing of our development technologies, raised over $23 million in venture capital financing, and raised non-dilutive funding from several out-licensing transactions with pharmaceutical partners. Dr. Stiernholm joined Trillium Privateco from YM BioSciences where he was Executive Vice President and Chief Scientific Officer. While there, he played a significant role in the success of their Initial Public Offering in 2002. Dr. Stiernholm began his industry career as a member of Allelix Biopharmaceuticals’ business development office. He currently serves on the boards of AvidBiologics and Vasomune Therapeutics. He received his Ph.D. in Immunology from the University of Toronto, where he also completed his postdoctoral training.
Dr. Robert Uger -Chief Scientific Officer
Dr. Uger became the Chief Scientific Officer on our merger with Trillium Privateco in April 2013. Dr. Uger is responsible for developing and implementing our scientific direction, and overseeing both internal product development and external research discovery programs. He also acts as our scientific liaison with respect to global collaborations with academic and hospital research scientists. Dr. Uger joined Trillium Privateco in 2003
60
Table of Contents
from Aventis Pasteur where he was a Senior Research Scientist involved in cancer vaccine research. He received his Ph.D. in Immunology from the University of Toronto.
Dr. Eric L. Sievers - Chief Medical Officer
Dr. Sievers is the Chief Medical Officer effective April 1, 2015. He brings more than 25 years’ experience in the biotechnology industry and academia. His expertise spans across multiple cancer indications including acute myeloid leukemia, myelodysplastic syndromes, Hodgkin and non-Hodgkin lymphoma, melanoma, and renal cell carcinoma. Dr. Sievers most recently served as Senior Vice President, Clinical Development at Seattle Genetics, where he also held several other senior clinical leadership positions over the past nine years. Notably, during his tenure at Seattle Genetics he played a key role in the development and approval of ADCETRIS®. Dr. Sievers started his industry career as Medical Director at ZymoGenetics. Prior to joining the biotechnology industry, Dr. Sievers spent over a decade at the Fred Hutchinson Cancer Research Center practicing medicine in the areas of hematology and oncology. He received his Medical and Bachelor of Arts degrees from Brown University.
James Parsons, CPA-CA -Chief Financial Officer
Mr. Parsons joined us in August 2011 and Trillium Privateco in 2003 on a part-time basis, and became full-time in June 2014. Mr. Parsons has an extensive background in the life sciences industry and over 25 years of financial management experience. Mr. Parsons was the Vice-President, Finance for DiaMedica Inc. from October 2010 to May 2014, and the Chief Financial Officer and Corporate Secretary for Amorfix Life Sciences Ltd. from 2006 to 2010 where his responsibilities included finance, administration, commercialization, risk management and corporate governance. Mr. Parsons has been a CFO and advisor in the life sciences industry since 2000 with early-stage to late-clinical stage biotechnology companies across many therapeutic, diagnostic and device areas. Mr. Parsons has a Master of Accounting degree from the University of Waterloo and is a Chartered Professional Accountant and Chartered Accountant.
Dr. Penka Petrova -Vice President, Drug Development
Dr. Petrova became the Vice President, Drug Development on our merger with Trillium Privateco in April 2013. Dr. Petrova is responsible for managing our formal drug development efforts, including all outsourced activities to contract research organizations. In addition, she continues to serve as the Head of Trillium’s cell biology group and oversees all thein vivo studies. Dr. Petrova joined Trillium Privateco from Prescient Neuropharma in 2003, where she was a Research Scientist and was involved in identifying and characterizing novel proteins involved in neuroprotection. Dr. Petrova received her Ph.D. in Microbiology from Saarland University in Saarbruecken, Germany, where she also conducted her postdoctoral studies.
Family Relationships
There are no family relationships among our directors and senior management.
Other Arrangements
There are no arrangements or understanding with major shareholders, customers, suppliers or others, pursuant to which any person referred to above was selected as a director or member of senior management.
Board Composition; Term of Office
Our board of directors currently consists of seven members. Our board of directors are elected at each annual general meeting of our shareholders and serve until their successors are elected or appointed, unless their office is vacated earlier. Our current board of directors was elected at the annual general meeting of shareholders on May 27, 2014.
61
Table of Contents
Director Independence
As a foreign private issuer, under the listing requirements and rules of the NASDAQ Capital Market, we are not required to have independent directors on our board of directors, except to the extent that our audit committee is required to consist of independent directors, subject to certain phase-in schedules. Nevertheless, our board of directors has undertaken a review of the independence of the directors and considered whether any director has a material relationship with us that could compromise his or her ability to exercise independent judgment in carrying out his or her responsibilities. Our board of directors determined that six of our seven directors are “independent directors” as defined under current rules and regulations of the SEC and the listing standards of the NASDAQ Capital Market. In making these determinations, our board of directors considered the current and prior relationships that each non-employee director has with our company and all other facts and circumstances that our board of directors deemed relevant in determining their independence.
Board Committees
We have an audit committee, a corporate governance and nominating committee and a compensation committee. Each of our committee charters is available on our website at www.trilliumtherapeutics.com.
Audit Committee
Our audit committee is comprised of a minimum of three members, each of whom, in the determination of our board of directors, satisfies the independence, financial literacy and experience requirements of applicable U.S. and Canadian securities laws, rules and guidelines, any applicable stock exchange requirements or guidelines and any other applicable regulatory rules.
In particular:
| • | each member shall be (a) an “Independent Director,” as defined in NASDAQ Marketplace Rule 5605(a)(2), and (b) “independent” within the meaning of Rule 10A-3 under the Exchange Act, and the determination of independence will be affirmatively made by our board annually, provided that our board may elect to take advantage of any exemption from such requirements provided in the rules of NASDAQ or the Exchange Act; |
| • | each member shall meet the independence and financial literacy requirements set forth in Canadian National Instrument 52- 110 Audit Committees; |
| • | each member shall not have participated in the preparation of the financial statements of ours (or any then current subsidiary of ours) at any time during the past three years; |
| • | each member shall be able to read and understand fundamental financial statements in accordance with the audit committee requirements for companies listed on NASDAQ in NASDAQ Marketplace Rule 5605(c)(2)(A)(iv); and |
| • | at least one (1) member shall, in the judgment of our board, be an “audit committee financial expert” within the meaning of such term in Item 407(d) of Regulation S-K under the Securities Act. |
Our audit committee members are Mr. Luke Beshar (Chair), Dr. Henry Friesen and Dr. Robert Kirkman, each of whom is a non-executive member of our board of directors. Our board of directors has determined that each of the members of our audit committee is financially literate and has sufficient financial expertise, and is independent within the meaning of such term in the rules of NASDAQ, the SEC and Canadian provincial securities regulatory authorities. Our board of directors has determined that Mr. Luke Beshar is a financial expert in accordance with the rules and regulations of the SEC.
The purpose of our audit committee is to assist our board of directors in:
| • | overseeing the integrity of our financial statements and our accounting and financial reporting processes and financial statement audits; |
62
Table of Contents
| • | overseeing our compliance with legal and regulatory requirements; |
| • | overseeing the qualifications and independence of our registered public accounting firm (independent auditor); |
| • | overseeing the performance of our independent auditor; and |
| • | overseeing the design, implementation and ongoing effectiveness of our systems of disclosure controls and procedures, risk management systems, internal control over financial reporting and compliance with ethical standards adopted by us. |
Corporate Governance and Nominating Committee
Our corporate governance and nominating committee shall be composed of at least two members of our board, all of whom are “independent directors” within the meaning of NASDAQ Rule 5605(a)(2). In affirmatively determining the independence of any member of our corporate governance and nominating committee, our board must consider all factors specifically relevant to determining whether a director has a relationship to us that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
All members of our corporate governance and nominating committee shall be “independent” as contemplated in National Instrument 58-101 – Disclosure of Corporate Governance Practices, such that all members of our corporate governance and nominating committee will have no direct or indirect relationship with us that could, in the view of our board of directors, be reasonably expected to interfere with the exercise of his or her independent judgment.
The purpose of our corporate governance and nominating committee is to:
| • | assist our board of directors in identifying prospective director nominees and recommend to our board of directors the director nominees for each annual meeting of shareholders; |
| • | recommend members for each board committee; |
| • | ensure that our board of directors is properly constituted to meet its fiduciary obligations to the Company and its shareholders and that the Company follows appropriate governance standards; |
| • | develop and recommend to our board of directors governance principles applicable to the Company; |
| • | oversee the succession planning for senior management; and |
| • | oversee the evaluation of our board of directors and management. |
and nomination committee members are Dr. Henry Friesen (Chair), Dr. Michael Moore and Dr. Thomas Reynolds. Our board of directors has determined that each member of our corporate governance and nomination committee is independent within the meaning of such term in the rules of NASDAQ and Canadian provincial securities regulatory authorities.
Compensation Committee
Our compensation committee shall be composed of at least two members of our board, all of whom are considered “independent” of the management of the Company in accordance with the provisions of Rule 10C-1(b)(1) under the Exchange Act and NASDAQ Rule 5605(a)(2). In affirmatively determining the independence of any member of our compensation committee, our board of directors must consider all factors specifically relevant to determining whether a director has a relationship to the Company that is material to that director’s ability to be independent from management in connection with the duties of a compensation committee member, including, but not limited to: (i) the source of compensation of such director, including any consulting, advisory or other compensatory fee paid by the Company to such director; and (ii) whether such director is affiliated with the Company, a subsidiary of the Company or an affiliate of a subsidiary of the Company.
The purpose of our compensation committee shall be to ensure that the compensation programs and values transferred to management through cash pay, share and share-based awards, whether immediate, deferred, or
63
Table of Contents
contingent are fair and appropriate to attract, retain and motivate management and are reasonable in view of company economics and of the relevant practices of other similar companies. Our compensation committee will also recommend to our board of directors compensation arrangements for board members.
Our compensation committee members are Dr. Michael Moore (Chair), Dr. Robert Kirkman and Dr. Thomas Reynolds. Our board of directors has determined that each member of our compensation committee is independent within the meaning of such term in the rules of NASDAQ and Canadian provincial securities regulatory authorities.
Compensation
For the year ended December 31, 2014, our directors and members of our administrative, supervisory or management bodies received compensation for services, as follows:
Name and Principal Position | Salary/Fees earned(1) | Share-based awards | Option- based awards(2) | Non-equity incentive plan com- pensation(3) | Total | |||||||||||||||||||||||||
Niclas Stiernholm(4) | $ | 390,000 | $ | - | $ | 2,035,768 | $ | 204,750 | $ | 2,630,518 | ||||||||||||||||||||
President & Chief Executive Officer and Director | ||||||||||||||||||||||||||||||
Robert Uger | 270,000 | - | 525,057 | 85,050 | 880,107 | |||||||||||||||||||||||||
Chief Scientific Officer | ||||||||||||||||||||||||||||||
James Parsons(5) | 218,750 | - | 439,393 | 65,625 | 723,768 | |||||||||||||||||||||||||
Chief Financial Officer | ||||||||||||||||||||||||||||||
Penka Petrova | 205,000 | - | 321,467 | 53,813 | 580,280 | |||||||||||||||||||||||||
| Vice President, Drug Development | ||||||||||||||||||||||||||||||
Luke Beshar(6) | 49,333 | 40,000 | 92,257 | - | 181,590 | |||||||||||||||||||||||||
Director | ||||||||||||||||||||||||||||||
Henry Friesen | 58,000 | 40,000 | -- | - | 98,000 | |||||||||||||||||||||||||
Director | ||||||||||||||||||||||||||||||
Robert Kirkman(6) | 53,000 | 40,000 | 74,693 | -- | 167,693 | |||||||||||||||||||||||||
Director | ||||||||||||||||||||||||||||||
Michael Moore(6) | 55,000 | 40,000 | - | - | 95,000 | |||||||||||||||||||||||||
Director | ||||||||||||||||||||||||||||||
Thomas Reynolds(6) | 43,333 | 40,000 | 92,257 | - | 175,590 | |||||||||||||||||||||||||
Director | ||||||||||||||||||||||||||||||
Calvin Stiller | 80,000 | 40,000 | - | - | 120,000 | |||||||||||||||||||||||||
Director, Chair | ||||||||||||||||||||||||||||||
David Allan(7) | 10,416 | - | - | - | 10,416 | |||||||||||||||||||||||||
Executive Chair and Director | ||||||||||||||||||||||||||||||
James DeMesa(8) | 5,528 | - | - | - | 5,528 | |||||||||||||||||||||||||
Director | ||||||||||||||||||||||||||||||
Dean Peterson(8) | 5,000 | - | - | - | 5,000 | |||||||||||||||||||||||||
Director | ||||||||||||||||||||||||||||||
Notes:
| (1) | For the year ended December 31, 2014, we compensated each director with an annual cash retainer of $40,000 and annual DSU’s in the amount of $40,000. Directors also received the following annual |
64
Table of Contents
fees: chair of the board of directors $40,000; chair of the audit committee $16,000; chair of the corporate governance and nominating committee and chair of the compensation committee $10,000; member of the audit committee $8,000; and member of the corporate governance and nominating committee and member of the compensation committee $5,000. |
| (2) | The option-based awards value is the grant date fair value of stock options granted in the year calculated in accordance with IFRS using the Black-Scholes option pricing model. |
| (3) | These payments reflect cash bonuses on the achievement of the 2014 corporate objectives. |
| (4) | Dr. Stiernholm was not compensated as a director. |
| (5) | Mr. Parsons worked on a part-time basis until June 1, 2014 when he joined full-time. Mr. Parsons’ annual salary for 2014 was $250,000. |
| (6) | Dr. Moore, Dr. Kirkman, Mr. Beshar and Dr. Reynolds were appointed to our board of directors on April 9, 2013, December 17, 2013, March 10, 2014, and March 10, 2014, respectively. |
| (7) | Mr. Allan was an officer to May 31, 2013, and a consultant thereafter. Mr. Allan received $2,500 in compensation as a director and $7,916 as a consultant. Mr. Allan resigned as a director and ceased being a consultant on January 25, 2014. |
| (8) | Dr. DeMesa resigned as a director on March 7, 2014. Mr. Peterson resigned as a director on March 6, 2014. |
Employment Agreements
Effective June 3, 2014, we entered into an employment agreement with Niclas Stiernholm which has an indefinite term and provides for his employment as Chief Executive Officer. The agreement provides for compensation with respect to Dr. Stiernholm’s annual base salary of $390,000 and participation in our bonus plan and stock option plan. Dr. Stiernholm’s agreement provides for severance pay of 15 months of salary plus bonus if terminated without cause and 18 months of salary plus bonus if terminated on a change in control. If the severance is as a result of a change in control, all unvested stock options will also immediately vest.
Effective June 3, 2014, we entered into an employment agreement with Robert Uger which has an indefinite term and provides for his employment as Chief Scientific Officer. The agreement provides for compensation with respect to Dr. Uger’s annual base salary of $270,000 and participation in our bonus plan and stock option plan. Dr. Uger’s agreement provides for severance pay of nine months base salary remuneration. If the severance is as a result of a change in control, all unvested stock options will also immediately vest.
Effective June 3, 2014, we entered into an employment agreement with James Parsons which has an indefinite term and provides for his employment as Chief Financial Officer. The agreement provides for compensation with respect to Mr. Parsons annual base salary of $250,000 and participation in our bonus plan and stock option plan. Mr. Parsons’ agreement provides for severance pay of nine months base salary remuneration. If the severance is as a result of a change in control, all unvested stock options will also immediately vest.
Effective June 3, 2014, we entered into an employment agreement with Penka Petrova which has an indefinite term and provides for her employment as Vice President, Drug Development. The agreement provides for compensation with respect to Dr. Petrova’s annual base salary of $205,000 and participation in our bonus plan and stock option plan. Dr. Petrova’s agreement provides for severance pay of six months base salary remuneration. If the severance is as a result of a change in control, all unvested stock options will also immediately vest.
We entered into an employment agreement with Eric Sievers effective April 1, 2015, which has an indefinite term and provides for his employment as Chief Medical Officer. The agreement provides for compensation with respect to Dr. Sievers annual base salary of US$375,000 and participation in our bonus plan and stock option plan. Dr. Siever’s agreement provides for severance pay of twelve months base salary remuneration. If the severance is as a result of a change in control, all unvested stock options will also immediately vest.
65
Table of Contents
Stock Option Plan
We have adopted a stock option plan, or the 2014 Stock Option Plan, that provides for the granting of stock options to officers, directors, employees and consultants of ours and our affiliates. The purpose of the 2014 Stock Option Plan is to advance our interests by encouraging our directors, officers and key employees and consultants retained to acquire common shares, thereby: (a) increasing the proprietary interests of such persons in us; (b) aligning the interests of such persons with the interests of our shareholders generally; (c) encouraging such persons to remain associated with us; and (d) furnishing such persons with an additional incentive in their efforts on behalf of us. As at December 31, 2014, pursuant to the 2014 Stock Option Plan, we were entitled to issue an additional 55,488 options.
The following is a summary only, and is qualified in its entirety by the terms and conditions of the 2014 Stock Option Plan, which is filed as an exhibit to the registration statement of which this prospectus forms a part.
Administration by the Board of Directors
The 2014 Stock Option Plan is administered by our board of directors which has final authority and discretion, subject to the express provisions of the 2014 Stock Option Plan, to interpret the 2014 Stock Option Plan, to prescribe, amend and rescind rules and regulations relating to it and to make all other determinations deemed necessary or advisable for the administration of the 2014 Stock Option Plan. This includes the discretion of our board of directors to decide who will participate in the 2014 Stock Option Plan, including directors, officers, employees or consultants, each a Participant. Our board of directors also has authority to delegate its duties to the compensation committee.
Expiry
Options granted under the 2014 Stock Option Plan, or the Options, are non-transferable, expire not later than ten years from the date of issuance and are exercisable as determined by our board of directors. In addition, notwithstanding the expiration date applicable to any Option, if an Option would otherwise expire during or immediately after a Blackout Period (as defined in the 2014 Stock Option Plan), then the expiration date of such Option shall be the 10th business day following the expiration of the Blackout Period, provided that in no event shall the period during which said Option is exercisable be extended beyond 10 years from the date such Option is granted to the Participant.
Exercise Price
The exercise price payable in respect of each Option may not be lower than the volume weighted average trading price of the common shares on the TSX over a period of five days preceding the date of grant.
Insider Participation Limit
The aggregate number of common shares issuable (or reserved for issuance) to insider Participants under the 2014 Stock Option Plan or any other security-based compensation arrangement of ours and our affiliates (including, without limitation, our 2014 Deferred Share Unit Plan, or the 2014 DSU Plan, may not at any time exceed 10% of the combined total number of common shares issued and outstanding (on a non-diluted basis) and the total number of common shares into which the outstanding preferred shares may be converted. Common shares issued to insider Participants under the 2014 Stock Option Plan or any other security-based arrangement of ours within a one-year period may not exceed 10% of the total number of common shares issued and outstanding (on a non-diluted basis) plus 10% of the total number of common shares into which the outstanding preferred shares may be converted.
Amendment Provisions
Our board of directors has the discretion to make amendments to the 2014 Stock Option Plan and any Options granted thereunder which it may deem necessary, without having to obtain shareholder approval. Such changes include, without limitation:
| • | minor changes of a “housekeeping” nature; |
66
Table of Contents
| • | amending Options under the 2014 Stock Option Plan, including with respect to the Option period (provided that the period during which an Option is exercisable does not exceed ten years from the date the Option is granted and that such Option is not held by an insider Participant), vesting period, exercise method and frequency, exercise price of an Option (provided that such Option is not held by an insider Participant) and method of determining the exercise price, assignability and effect of termination of a Participant’s employment or cessation of the Participant’s directorship; |
| • | changing the class of Participants eligible to participate under the 2014 Stock Option Plan; |
| • | changing the terms and conditions of any financial assistance which may be provided by us to Participants to facilitate the purchase of common shares under the 2014 Stock Option Plan; and |
| • | adding a cashless exercise feature, payable in cash or securities, whether or not providing for a full deduction of the number of underlying common shares from the 2014 Stock Option Plan reserve. |
Shareholder approval will be required in the case of: (i) any amendment to the amendment provisions of the 2014 Stock Option Plan; (ii) any increase in the maximum number of common shares issuable under the 2014 Stock Option Plan; and (iii) any reduction in the exercise price or extension of the Option period benefiting an insider Participant, in addition to such other matters that may require shareholder approval under the rules and policies of the TSX.
Termination, Resignation, Death, etc.
Options granted under the 2014 Stock Option Plan are, and will be, evidenced by an option agreement entered between us and the Participant. Options granted under the plan terminate immediately if a Participant is dismissed with cause. If a Participant ceases to hold any position as a Participant, by reason of retirement, resignation or termination, the vested options terminate within 120 days from cessation (or until the normal expiry of the Options if earlier). If a Participant ceases to hold any position as a Participant, by reason of retirement, resignation or termination, the vested options terminate within 120 days from cessation (or until the normal expiry of the Options if earlier). Under the 2014 Stock Option Plan, if a Participant dies, his options may be exercised by his legal representatives during the period of (and they shall terminate) one year following the date to the extent they were exercisable on the date of death. If a Participant ceases to be a director, officer or employee of, or consultant to, the Company or of one of our subsidiaries as a result of disability or illness preventing the Participant from performing the duties routinely performed by such Participant, vested options terminate 180 days from the date that the Participant ceases to serve in such capacities (or until the normal expiry date of the Options if earlier).
Change of Control
The 2014 Stock Option Plan provides that any Options outstanding immediately prior to the occurrence of a Change of Control (as defined in the plan), but which are not then exercisable, shall immediately vest and become fully exercisable upon the occurrence of a Change of Control.
Maximum Limit
The 2014 Stock Option Plan is a “rolling” and “reloading” stock option plan, meaning that Options may be granted for no more than 10% of the total number of common shares issued and outstanding (on a non-diluted basis) plus 10% of the total number of common shares into which the outstanding preferred shares may be converted in accordance with their terms less the number of any common shares reserved for issuance to insiders of ours pursuant to the 2014 DSU Plan. Each exercise of Options will result in a corresponding increase in the number of Options available for granting under the 2014 Stock Option Plan, up to the 10% aggregate maximum limit. There are additional restrictions in the 2014 Stock Option Plan with regard to the number of Options that may be granted which include the restriction that the aggregate number of Options granted in any 12 month period to any one person cannot exceed 5% of the aggregate total number of issued and outstanding common shares (on a non-diluted basis) and common shares into which the outstanding preferred shares may be converted in accordance with their terms.
67
Table of Contents
Other Terms
Any consolidation or subdivision of common shares will be reflected in an adjustment to the stock options.
Option Grant Summary
The following table sets forth the outstanding option-based awards outstanding for each of our directors and officers as at March 9, 2015:
| Option-based Awards | ||||||||||||||||
| Name and Office Held | Number of securities underlying unexercised options | Option exercise price | Option expiration date | Value of unexercised in- the-money options(1) | ||||||||||||
Niclas Stiernholm
|
| 1,667 42,505 159,768 134,848 |
|
| $30.00 $7.50 $10.35 $8.34 |
|
| Jul 18, 2016 Apr 8, 2023 Apr 27, 2024 May 27, 2024 |
|
| Nil $629,074 $1,909,228 $1,882,478 |
| ||||
Robert Uger
|
| 8,501 42,067 33,712 |
|
| $7.50 $10.35 $8.34 |
|
| Apr 8, 2023 Apr 27, 2024 May 27, 2024 |
|
| $125,815 $502,701 $470,620 |
| ||||
James Parsons
|
| 417 4,250 36,204 26,970 |
|
| $30.00 $7.50 $10.35 $8.34 |
|
| Aug 25, 2016 Apr 8, 2023 Apr 27, 2024 May 27, 2024 |
|
| Nil $62,900 $432,638 $376,501 |
| ||||
Penka Petrova |
| 4,250 26,090 20,227 |
|
| $7.50 $10.35 $8.34 |
|
| Apr 8, 2023 Apr 27, 2024 May 27, 2024 |
|
| $62,900 $311,776 $202,369 |
| ||||
Luke Beshar | 6,667 | $18.90 | Mar 6, 2024 | $22,668 | ||||||||||||
Henry Friesen |
| 1,667 4,500 |
|
| $30.00 $7.50 |
|
| Jun 28, 2016 Apr 8, 2023 |
|
| Nil $66,600 |
| ||||
Robert Kirkman | 6,667 | $15.30 | Jan 29, 2024 | $46,669 | ||||||||||||
Michael Moore | 4,000 | $7.50 | Apr 8, 2023 | $59,200 | ||||||||||||
Thomas Reynolds | 6,667 | $18.90 | Mar 6, 2024 | $22,668 | ||||||||||||
Calvin Stiller |
| 1,667 4,000 |
|
| $30.00 7.50 |
| | Jul 18, 2016 Apr 8, 2023 | |
| Nil $59,200 |
| ||||
Note:
| (1) | The value of the unexercised “in-the-money” options as at March 20, 2015 has been determined based on the excess of the closing price on such date of the common shares on the TSX of $22.30 per common share over the exercise price of such options. |
68
Table of Contents
Deferred Share Unit Plan
Our shareholders approved the 2014 DSU Plan on May 27, 2014. The 2014 DSU Plan is intended to promote a greater alignment of long-term interests between non-executive directors and executive officers of ours and its shareholders through the issuance of DSUs. Since the value of a DSU increases or decreases with the market
price of the common shares, DSUs reflect a philosophy of aligning the interests of directors and executive officers with those of the shareholders by tying compensation to share price performance. Our board of directors intends to use DSUs issued under the 2014 DSU Plan, as well as Options issued under the 2014 Stock Option Plan, as part of our overall director and executive officer compensation program. A total of 28,777 units were issued and outstanding as at December 31, 2014.
Overview of the 2014 DSU Plan
The 2014 DSU Plan provides that, subject to the terms of the 2014 DSU Plan and such other conditions as our board of directors (or compensation committee of our board of directors after delegation by authority from our board of directors) may impose, an executive officer or director of ours, each an Eligible Person, shall receive his or her Total Compensation in the form of DSUs. The term “Total Compensation” includes annual and special bonuses payable to directors and executive officers and, in the case of directors, directors fees (including annual board retainers, fees for serving as chair of our board of directors and/or as a chair or member of any committee of our board of directors, for attending meetings of our board of directors or any committee thereof, and any other fees payable to directors) in the form of DSUs. Our board of directors may use DSUs to pay bonuses and directors fees either alone or in conjunction with cash, or any combination of DSUs and cash.
The number of DSUs (including fractional DSUs, computed to three digits) to be credited to an Eligible Person for services will be determined by dividing the awarded amount by the fair market value as at the last trading day before the date the awarded amount is declared by our board of directors. “Fair Market Value” of the common shares is the volume weighted average trading price of the common shares on the TSX for the five days immediately preceding the date in question.
An Eligible Person who has ceased to be a director or executive officer (other than as a result of death) may elect to receive one common share in respect of each whole DSU credited to the Eligible Person’s account by filing with us a notice of redemption in the form and by the time stipulated in the 2014 DSU Plan.
If the Eligible Person does not make the election on a timely basis, the Eligible Person will be deemed to have elected to redeem all of his or her DSUs.
The issuance of the common shares will be made by us as soon as reasonably possible following the election to redeem the DSUs, or being deemed to have been made, by the Eligible Person.
Maximum Number of Shares Issuable under the Plan
The “Outstanding Issue” means the combined total of the number of common shares outstanding and the number of common shares into which the preferred shares outstanding (on a non-diluted basis) may be converted in accordance with their terms. The maximum number of common shares reserved for issuance under the 2014 DSU Plan is 66,667 on a post-consolidated basis, which is approximately 1.0% of the current Outstanding Issue, subject to adjustment.
The 2014 DSU Plan provides that the maximum number of common shares that may be reserved for issuance to Insiders (as that term is defined in the TSX��s rules) pursuant to the 2014 DSU Plan, together with any common shares issuable pursuant to any other securities-based compensation arrangement of ours (including the 2014 Stock Option Plan), will not exceed 10% of the Outstanding Issue.
69
Table of Contents
In addition, the maximum number of common shares that may be issued to Insiders under the 2014 DSU Plan, together with any common shares issued to Insiders pursuant to any other securities-based compensation arrangement of ours (including the 2014 Stock Option Plan), within any one year period, will not exceed 10% of the Outstanding Issue. Also, in no event, may the number of common shares reserved for issuance to any one person pursuant to the 2014 DSU Plan and the 2014 Stock Option Plan exceed 5% of the Outstanding Issue.
Transferability
DSUs and any other rights, benefits or interests in the 2014 DSU Plan are non-transferable, except that if the Eligible Person dies, the legal representatives of the Eligible Person will be entitled to receive the amount of any payment otherwise payable to the Eligible Person in accordance with the provisions 2014 DSU Plan.
Amendments to the 2014 DSU Plan
Our board of directors has the discretion to make amendments to the 2014 DSU Plan and any DSUs granted thereunder which it may deem necessary, without having to obtain shareholder approval. Such changes may include, without limitation:
| • | minor changes of a “housekeeping” nature; |
| • | amending the terms of DSUs under the 2014 DSU Plan and method of determining the awarded amount and the number of DSUs that may be issued to an Eligible Person, and the assignability and effect of terminated service of an Eligible Person; |
| • | changing the class of Eligible Persons; and |
| • | changing the method and procedures to be followed with regard to the issuance of DSUs under the 2014 DSU Plan. |
Shareholder approval will be required in the case of: (i) any amendment to the amendment provisions of the 2014 DSU Plan; (ii) any increase in the maximum number of common shares issuable under the 2014 DSU Plan; and (iii) such other matters that may require shareholder approval under the rules and policies of the TSX.
Termination of Service
An Eligible Person who has terminated service may elect to receive one common share in respect of each whole DSU credited to the Eligible Person’s account, by filing a notice of redemption in the form prescribed from time to time by us on or before December 15 of the first calendar year commencing after the date on which the Eligible Person has terminated service. If the Eligible Person fails to file such notice on or before that December 15, the Eligible Person will be deemed to have filed a notice of redemption on that December 15 and will be deemed to have elected to redeem all of his or her DSUs. The date on which a notice is filed or deemed to be filed with the Secretary of the Company is the “Filing Date”. We may defer the Filing Date to any other date if such deferral is, in the sole opinion of ours, desirable to ensure compliance the 2014 DSU Plan. There are no causes of cessation of entitlement under the 2014 DSU Plan, including termination for or without cause.
In the event of the death of an Eligible Person, we will, within two months of the Eligible Person’s death, pay cash equal to the Fair Market Value of the shares which would be deliverable to the Eligible Person if the Eligible Person had terminated service in respect of the DSUs credited to the deceased Eligible Person’s account (net of any applicable withholding tax) to or for the benefit of the legal representative of the Eligible Person. The Fair Market Value will be calculated on the date of death of the Eligible Person.
The foregoing is a summary only, and is qualified in its entirety by the terms and conditions of the 2014 DSU Plan which is filed as an exhibit to the registration statement of which this prospectus forms a part.
70
Table of Contents
DSU Grant Summary
As of March 23, 2015, Mr. Beshar, Dr. Friesen, Dr. Kirkman, Dr. Moore, Dr. Reynolds and Dr. Stiller each held 4,796 DSUs. Dr. Stiernholm, Dr. Uger, Mr. Parsons and Dr. Petrova held no DSUs as of March 23, 2015. Deferred share units are redeemable on a one-for-one basis for common shares only after termination of service with us.
Termination and Change of Control Benefits
Except as disclosed above, we have no plans or arrangements in respect of remuneration received or that may be received by our directors and senior management in respect of compensating such person in the event of termination of employment (as a result of, for example, resignation, retirement, change of control) or a change in responsibilities.
Pension, Retirement or Similar Benefits
We have not set aside or accrued any amounts to provide pension, retirement or similar benefit for our directors or senior management.
Service Contracts
See the disclosure in the section of this prospectus entitled “Board of Directors and Management – Employment Agreements” for particulars of Dr. Stiernholm’s service contract. Other than as disclosed herein, we do not have any service contracts with directors which provide for benefits upon termination of employment.
71
Table of Contents
The following table and accompanying footnotes sets forth, as of March 20, 2015, information regarding beneficial ownership of our common shares by:
| • | each person, or group of affiliated persons, known by us to beneficially own more than 5% of our common shares; |
| • | each of our executive officers; |
| • | each of our directors; and |
| • | all of our executive officers and directors as a group. |
Beneficial ownership is determined according to the rules of the SEC and generally means that a person has beneficial ownership of a security if he, she or it possesses sole or shared voting or investment power of that security, including options and warrants that are currently exercisable or exercisable within 60 days of March 20, 2015. Common shares subject to options and warrants currently exercisable or exercisable within 60 days of March 20, 2015 are deemed to be outstanding for computing the percentage ownership of the person holding these options and/or warrants and the percentage ownership of any group of which the holder is a member, but are not deemed outstanding for computing the percentage of any other person.
Except as indicated by the footnotes below, we believe, based on the information furnished to us, that the persons named in the table below have sole voting and investment power with respect to all common shares shown that they beneficially own, subject to community property laws where applicable. The table is based upon information supplied by officers, directors and principal stockholders and Schedule 13Gs filed with the SEC.
Our calculation of the percentage of beneficial ownership prior to this offering is based on 5,018,139 common shares issued and outstanding as at March 20, 2015. Beneficial ownership representing less than 1% is denoted with an asterisk (*).
Unless otherwise indicated, the address of each beneficial owner listed in the table below is c/o Trillium Therapeutics Inc., 96 Skyway Avenue, Toronto, Ontario, Canada M9W 4Y9.
| Number of shares beneficially owned | Percentage of shares beneficially owned
| |||||||||||
|
| |||||||||||
Name of Beneficial Owner | Before offering | After offering(1) | ||||||||||
5% or greater shareholders | ||||||||||||
Merlin Nexus IV, LP(2) | 708,472 | 13.3% | % | |||||||||
Thomas D. Mottola(3) | 421,240 | 8.2% | % | |||||||||
Entities affiliated with Special Situations Fund(4) | 404,841 | 8.1% | % | |||||||||
Opaleye Management Inc.(5) | 330,900 | 6.6% | % | |||||||||
James H. D. Stebbins(6) | 236,450 | 4.7% | % | |||||||||
Deerfield Special Situations Fund, L.P.(7) | 226,046 | 4.5% | % | |||||||||
Executive officers and directors | ||||||||||||
Niclas Stiernholm | 154,836 | 3.0% | % | |||||||||
Calvin Stiller(8) | 59,584 | 1.2% | % | |||||||||
Robert Uger | 37,938 | * | % | |||||||||
James Parsons | 30,342 | * | % | |||||||||
Penka Petrova | 22,620 | * | % | |||||||||
Henry Friesen | 4,667 | * | % | |||||||||
Luke Beshar | 3,333 | * | % | |||||||||
72
Table of Contents
| Number of shares beneficially owned | Percentage of shares beneficially owned
| |||||||||||
|
| |||||||||||
Name of Beneficial Owner | Before offering | After offering(1) | ||||||||||
Robert Kirkman | 3,333 | * | % | |||||||||
Thomas Reynolds | 3,333 | * | % | |||||||||
Michael Moore | 2,667 | * | % | |||||||||
| All executive officers and directors as a group (10 persons)(9) | 322,653 | 6.1% | % | |||||||||
Notes:
| (1) | Assumes no exercise of the underwriters’ option to purchase additional shares. |
| (2) | Based on information set forth in a Schedule 13G/A filed with the SEC on February 17, 2015 by Merlin Biomed Private Equity Advisors, LLC (Merlin Biomed), Merlin Nexus IV, L.P. (Merlin IV) and Dominique Sémon with respect to both common shares held and warrants exercisable for common shares. Merlin BioMed is the investment adviser to Merlin IV. Dominique Sémon is the Managing Member of Merlin BioMed. Merlin IV, Merlin BioMed and Dominique Sémon have shared voting power and dispositive power over the common shares held by Merlin IV. The principal address of Merlin IV is 424 West 33rd Street, Suite 520, New York, NY 10001. |
| (3) | Based on information set forth in a Schedule 13G filed with the SEC on March 20, 2015 by Thomas D. Mottola. Consists of 275,560 common shares and warrants exercisable for 145,680 common shares. Mr. Mottola has sole voting power and dispositive power over such securities. The principal address of Mr. Mottola is 150 East 52nd Street, 21st Floor, New York, NY 10022. |
| (4) | Consists of (i) 125,501 common shares owned by Special Situations Fund III QP, L.P., (ii) 36,436 common shares owned by Special Situations Cayman Fund L.P., (iii) 161,937 common shares owned by Special Situations Life Sciences Fund, L.P. and (iv) 80,968 common shares owned by Special Situations Private Equity Fund, L.P. The principal address of Special Situations Fund is 527 Madison Avenue, Suite 2600, New York, New York 10022. |
| (5) | Based on information set forth in a Schedule 13G filed with the SEC on February 23, 2015 by James Silverman and Opaleye Management Inc. James Silverman, the President of Opaleye Management Inc., has voting and disposition power with respect to the 330,900 common shares owned by Opaleye Management Inc. The principal address of Opaleye Management Inc. is 9B Russell Street, Cambridge, MA 02140. |
| (6) | Based on information set forth in a Schedule 13G filed with the SEC on February 16, 2015 by James H. D. Stebbins. Mr. Stebbins has sole voting and dispositive power over the common shares. The principal address of Mr. Stebbins is 222 East Wisconsin Avenue, Suite 303B, Lake Forest, IL 60045. |
| (7) | Based on information set forth in a Schedule 13G filed with the SEC on February 18, 2015 by James E. Flynn, Deerfield Mgmt, L.P., Deerfield Management Company, L.P., and Deerfield Special Situations Fund, L.P. (the Fund). Deerfield Mgmt, L.P. is the general partner of the Fund. Deerfield Management Company, L.P. is the investment manager of the Fund. James E. Flynn is the sole member of the general partner of Deerfield Mgmt, L.P. and Deerfield Management Company, L.P. Deerfield Mgmt, L.P. may be deemed to beneficially own the shares held by the Fund. Each of Deerfield Management Company, L.P. and Mr. Flynn may be deemed to beneficially own the shares held by the Fund. The address of the Fund is c/o Deerfield Management Company, L.P., 780 Third Avenue, 37th Floor, New York, NY 10017. |
| (8) | Includes 31,000 common shares and 727,500 warrants exercisable for common shares that Dr. Stiller exercises direct or indirect control or direction. |
| (9) | Includes an aggregate of 267,403 common shares underlying options exercisable within 60 days of March 20, 2015. |
73
Table of Contents
Other than as disclosed in this prospectus, since January 1, 2012, there have been no transactions or loans between us and:
| (a) | enterprises that directly or indirectly through one or more intermediaries, control or are controlled by, or are under common control with, us; |
| (b) | associates, meaning unconsolidated enterprises in which we have a significant influence or which have significant influence over us; |
| (c) | individuals owning, directly or indirectly, an interest in the voting power of us that gives them significant influence over our us, and close members of any such individual’s family; |
| (d) | key management personnel, that is, those persons having authority and responsibility for planning, directing and controlling the activities of ours, including directors and senior management of us and close members of such individuals’ families; and |
| (e) | enterprises in which a substantial interest in the voting power is owned, directly or indirectly, by any person described in (c) or (d) or over which such a person is able to exercise significant influence, including enterprises owned by directors or major shareholders of us and enterprises that have a member of key management in common with us. |
For the years ended December 31, 2014 and 2013, $7,916 and $138,750, respectively, was paid to our former Executive Chair, a director, for consulting fees. These transactions were in the normal course of operations and were measured at the exchange amount, which is the amount of consideration established and agreed to by the parties.
74
Table of Contents
PRICE RANGE OF COMMON SHARES AND TRADING MARKETS
Price History
Our common shares were listed on the TSXV until April 22, 2014 when we delisted from the TSXV and began trading on the TSX. Our common shares traded under the symbol “SSS” until June 6, 2014 when the symbol was changed to “TR”. Our common shares were listed on the OTCQX International under the symbol “SCTPF” from May 20, 2013 until we delisted and began trading on the NASDAQ Capital Market under the symbol “TRIL” on December 19, 2014.
Five Most Recent Financial Years
The annual high and low market prices of our common shares for the five most recent full financial years on the TSXV/TSX and since May 20, 2013 on the OTCQX International and from December 19, 2014 on the NASDAQ Capital Market were as follows:
| Year ended December 31, | TSXV/TSX(1)(3) | OTCQX International(2)(3) | NASDAQ(2)(3) | |||||||||||||||||||||||||||||||||||
| High | Low | High | Low | High | Low | |||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| ||||||||||||||||||||||||||||
2014 | $ | 22.20 | $ | 6.30 | US$ | 19.60 | US$ | 5.64 | US$ | 9.10 | US$ | 7.01 | ||||||||||||||||||||||||||
2013 | 18.00 | 4.20 | 14.27 | 4.16 | ||||||||||||||||||||||||||||||||||
2012 | 18.00 | 4.50 | ||||||||||||||||||||||||||||||||||||
2011 | 42.00 | 12.00 | ||||||||||||||||||||||||||||||||||||
2010 | 165.00 | 13.50 | ||||||||||||||||||||||||||||||||||||
Notes:
| (1) | Our common shares began trading on the TSX on April 22, 2014. |
| (2) | Our common shares began trading on the OTCQX International on May 20, 2013 and on the NASDAQ Capital Market on December 19, 2014. |
| (3) | Common share market prices are restated to reflect the 30 for 1 share consolidation completed in November 2014. |
Full Financial Quarters
The high and low market prices of our common shares for each full financial quarter for the two most recent full financial years on the TSXV/TSX, the OTCQX Marketplace and the NASDAQ Capital Market were as follows:
| Quarter ended | TSXV/TSX(1)(3) | OTCQX International(2)(3) | NASDAQ(2)(3) | |||||||||||||||||||||||||||||||||||
| High | Low | High | Low | High | Low | |||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| ||||||||||||||||||||||||||||
December 31, 2014 | $ | 10.50 | $ | 6.30 | US$ | 9.09 | US$ | 5.64 | US$ | 9.10 | US$ | 7.01 | ||||||||||||||||||||||||||
September 30, 2014 | 10.35 | 7.20 | 9.96 | 6.30 | ||||||||||||||||||||||||||||||||||
June 30, 2014 | 15.60 | 7.95 | 14.24 | 7.14 | ||||||||||||||||||||||||||||||||||
March 31, 2014 | 22.20 | 11.40 | 19.60 | 10.61 | ||||||||||||||||||||||||||||||||||
December 31, 2013 | 13.20 | 7.80 | 12.42 | 7.26 | ||||||||||||||||||||||||||||||||||
September 30, 2013 | 14.85 | 6.60 | 14.27 | 6.53 | ||||||||||||||||||||||||||||||||||
June 30, 2013 | 7.50 | 4.20 | 7.22 | 4.16 | ||||||||||||||||||||||||||||||||||
March 31, 2013 | 18.00 | 5.55 | ||||||||||||||||||||||||||||||||||||
Notes:
| (1) | Our common shares began trading on the TSX on April 22, 2014. |
75
Table of Contents
| (2) | Our common shares began trading on the OTCQX International on May 20, 2013 and on the NASDAQ Capital Market on December 19, 2014. |
| (3) | Common share market prices are restated to reflect the 30 for 1 share consolidation completed in November 2014. |
Most Recent Six Months
The high and low market prices of our common shares for each month for the most recent six months on the TSXV/TSX, OTCQX Marketplace and the NASDAQ Capital Market were as follows:
| Month ended | TSXV/TSX(1)(2) | OTCQX International(1)(2) | NASDAQ(1) | |||||||||||||||||||||||||||||||||
| High | Low | High | Low | High | Low | |||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
| ||||||||||||||||||||||||||
March 31, 2015 (through March 27) | $ | 23.00 | $ | 17.00 | US$ | US$ | US$ | 18.37 | US$ | 13.60 | ||||||||||||||||||||||||||
February 28, 2015 | 20.21 | 15.14 | 16.50 | 12.61 | ||||||||||||||||||||||||||||||||
January 31, 2015 | 18.37 | 10.50 | 15.65 | 9.05 | ||||||||||||||||||||||||||||||||
December 31, 2014 | 10.50 | 7.02 | 8.35 | 6.02 | 9.10 | 7.01 | ||||||||||||||||||||||||||||||
November 30, 2014 | 10.35 | 8.25 | 9.09 | 7.35 | ||||||||||||||||||||||||||||||||
October 30, 2014 | 8.40 | 6.30 | 7.65 | 5.64 | ||||||||||||||||||||||||||||||||
Notes:
| (1) | Our common shares began trading on the OTCQX International on May 20, 2013 and on the NASDAQ Capital Market on December 19, 2014. |
| (2) | Common share market prices are restated to reflect the 30 for 1 share consolidation completed in November 2014. |
Shareholders
As at March 26, 2015, approximately 52% of common shares and 100% of Series I First Preferred Shares were held by shareholders in the United States. As at March 26, 2015, there were 82 record holders in the United States.
Transfer Agent
Our common shares, with no par value, are in registered form and the transfer of our common shares is managed by our transfer agent, Computershare Investor Services Inc., 8th floor, University Avenue, Toronto, Ontario, Canada (Tel: (800) 564-6253).
76
Table of Contents
We are offering the securities described in this prospectus through a number of underwriters. Leerink Partners LLC and Cowen and Company, LLC are acting as joint book-running managers of the offering and as representatives of the underwriters. We have entered into an underwriting agreement with the underwriters. Subject to the terms and conditions of the underwriting agreement, we have agreed to sell to the underwriters, and each underwriter has severally agreed to purchase, at the public offering price less the underwriting discounts and commissions set forth on the cover page of this prospectus, the number of common shares and Series II First Preferred Shares listed next to its name in the following table:
Name | Number of Common Shares | Number of Series II First Preferred Shares | ||
Leerink Partners LLC | ||||
Cowen and Company, LLC | ||||
Oppenheimer & Co. Inc. | ||||
|
| |||
Total | ||||
|
|
The underwriters are committed to purchase all the common shares and Series II First Preferred Shares offered by us if they purchase any shares. The underwriting agreement also provides that if an underwriter defaults, the purchase commitments of non-defaulting underwriters may also be increased or the offering may be terminated.
The underwriters propose to offer the common shares and Series II First Preferred Shares directly to the public at the public offering price set forth on the cover page of this prospectus and to certain dealers at that price less a concession not in excess of US$ per share. After the public offering of the shares, the offering price and other selling terms may be changed by the underwriters. Sales of shares made outside of the United States may be made by affiliates of the underwriters.
The underwriters have an option to buy from us up to an additional 15% of the number of common shares offered hereby. The underwriters have 30 days from the date of this prospectus to exercise this option to purchase additional common shares. If any common shares are purchased with this option to purchase additional common shares, the underwriters will purchase common shares in approximately the same proportion as shown in the table above. If any additional common shares are purchased, the underwriters will offer the additional common shares on the same terms as those on which the common shares are being offered.
The underwriting fee is equal to the public offering price per share less the amount paid by the underwriters to us per share. The underwriting fee is US$ per common share and US$ per Series II First Preferred Share. The following table shows the per share and total underwriting discounts and commissions to be paid to the underwriters assuming both no exercise and full exercise of the underwriters’ option to purchase additional shares.
| No Exercise | Full Exercise | |||||||||||||||||||||||
| Common Shares | Series II First Preferred Shares | Common Shares | Series II First Preferred Shares | |||||||||||||||||||||
Per Share | US$ | US$ | US$ | US$ | ||||||||||||||||||||
|
|
|
| |||||||||||||||||||||
Total | US$ | US$ | US$ | US$ | ||||||||||||||||||||
|
|
|
| |||||||||||||||||||||
We estimate that the total expenses of this offering, including registration, filing and listing fees, printing fees and legal and accounting expenses, but excluding the underwriting discounts and commissions, will be approximately US$ .
77
Table of Contents
We have agreed that we will not (i) offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase or otherwise dispose of, directly or indirectly, or file with the Securities and Exchange Commission a registration statement under the Securities Act relating to, any of our common shares or securities convertible into or exchangeable or exercisable for any of our common shares, or publicly disclose the intention to make any offer, sale, pledge, disposition or filing, or (ii) enter into any swap or other arrangement that transfers all or a portion of the economic consequences associated with the ownership of any common shares or any such other securities (regardless of whether any of these transactions are to be settled by the delivery of common shares or such other securities, in cash or otherwise), in each case without the prior written consent of Leerink Partners LLC and Cowen and Company, LLC for a period of 180 days after the date of this prospectus, other than our common shares and Series II First Preferred Shares (including the common shares issuable upon conversion of the Series II First Preferred Shares) to be sold hereunder and any of our common shares issued upon the exercise of options granted under our existing share-based compensation plans.
Our directors and executive officers have entered into lock-up agreements with the underwriters prior to the commencement of this offering pursuant to which each of these persons, with limited exceptions, for a period of 180 days after the date of this prospectus, may not, without the prior written consent of Leerink Partners LLC and Cowen and Company, LLC, (1) offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, lend or otherwise transfer or dispose of, directly or indirectly, any of our common shares or any securities convertible into, or exercisable or exchangeable for, our common shares (including, without limitation, common shares or such other securities which may be deemed to be beneficially owned by such directors and executive officers in accordance with the rules and regulations of the SEC and securities which may be issued upon exercise of an option or warrant) or publicly disclose the intention to make any offer, sale, pledge or disposition or (2) enter into any swap or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of the common shares or such other securities, whether any such transaction described in clause (1) or (2) above is to be settled by delivery of common shares or such other securities, in cash or otherwise, or (3) make any demand for or exercise any right with respect to the registration of any of our common shares or any security convertible into or exercisable or exchangeable for our common shares subject to limited exceptions which include:
| • | sales of securities acquired in open market transactions after the completion of this offering, |
| • | transfers of securities (i) as a bona fide gift or gifts or (ii) by will or intestacy to the legal representative, heir, beneficiary or a member of the immediate family of the undersigned in a transaction not involving a disposition for value, |
| • | transfers of our common shares or any security convertible into our common shares to any trust for the benefit of the holder or the immediate family of the undersigned, or limited partnerships the partners of which are the holder and/or the immediate family members of the holder, in each case for estate planning purposes, |
| • | if the holder is a trust, distributions of our common shares or any security convertible into our common shares to its beneficiaries in a transaction not involving a disposition for value, |
| • | if the holder is a corporation, limited liability company, partnership or other entity, distribution of our common shares or any security convertible into our common shares to members, shareholders, limited partners, subsidiaries or affiliates (as defined in Rule 405 promulgated under the Securities Act) of the holder or to any investment fund or other entity that controls or manages the holder in a transaction not involving a disposition for value, |
| • | transfers to us pursuant to agreements under which we have the option to repurchase such shares or securities upon termination of service of the holder, and |
| • | the receipt by the holder from us of our common shares upon the exercise of options. |
We have agreed to indemnify the underwriters against certain liabilities, including liabilities under the Securities Act.
Our common shares are listed on the Toronto Stock Exchange under the symbol “TR” and on the NASDAQ Capital Market under the symbol “TRIL”. There is no established public trading market for our Series II First
78
Table of Contents
Preferred Shares, and we do not expect a market to develop. In addition, we do not intend to apply for listing of our Series II First Preferred Shares on any national securities exchange or other nationally recognized trading system.
In connection with this offering, the underwriters may engage in stabilizing transactions, which involves making bids for, purchasing and selling common shares in the open market for the purpose of preventing or retarding a decline in the market price of the common shares while this offering is in progress. These stabilizing transactions may include making short sales of the common shares, which involves the sale by the underwriters of a greater number of common shares than they are required to purchase in this offering, and purchasing common shares on the open market to cover positions created by short sales. Short sales may be “covered” shorts, which are short positions in an amount not greater than the underwriters’ option to purchase additional shares referred to above, or may be “naked” shorts, which are short positions in excess of that amount. The underwriters may close out any covered short position either by exercising their option to purchase additional shares, in whole or in part, or by purchasing shares in the open market. In making this determination, the underwriters will consider, among other things, the price of shares available for purchase in the open market compared to the price at which the underwriters may purchase shares through the option to purchase additional shares. A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of the common shares in the open market that could adversely affect investors who purchase in this offering. To the extent that the underwriters create a naked short position, they will purchase shares in the open market to cover the position.
The underwriters have advised us that, pursuant to Regulation M of the Securities Act, they may also engage in other activities that stabilize, maintain or otherwise affect the price of the common shares, including the imposition of penalty bids. This means that if the representatives of the underwriters purchase common shares in the open market in stabilizing transactions or to cover short sales, the representatives can require the underwriters that sold those shares as part of this offering to repay the underwriting discount received by them.
These activities may have the effect of raising or maintaining the market price of the common shares or preventing or retarding a decline in the market price of the common shares, and, as a result, the price of the common shares may be higher than the price that otherwise might exist in the open market. If the underwriters commence these activities, they may discontinue them at any time. The underwriters may carry out these transactions on the NASDAQ Capital Market, in the over-the-counter market or otherwise.
In addition, in connection with this offering certain of the underwriters (and selling group members) may engage in passive market making transactions in our common shares on the NASDAQ Capital Market prior to the pricing and completion of this offering. Passive market making consists of displaying bids on the NASDAQ Capital Market no higher than the bid prices of independent market makers and making purchases at prices no higher than these independent bids and effected in response to order flow. Net purchases by a passive market maker on each day are generally limited to a specified percentage of the passive market maker’s average daily trading volume in the common shares during a specified period and must be discontinued when such limit is reached. Passive market making may cause the price of our common shares to be higher than the price that otherwise would exist in the open market in the absence of these transactions. If passive market making is commenced, it may be discontinued at any time.
Certain of the underwriters and their affiliates have provided in the past to us and our affiliates and may provide from time to time in the future certain commercial banking, financial advisory, investment banking and other services for us and such affiliates in the ordinary course of their business, for which they have received and may continue to receive customary fees and commissions. In addition, from time to time, certain of the underwriters and their affiliates may effect transactions for their own account or the account of customers, and hold on behalf of themselves or their customers, long or short positions in our debt or equity securities or loans, and may do so in the future.
79
Table of Contents
International Selling Restrictions
Other than in the United States, no action has been taken by us or the underwriters that would permit a public offering of the securities offered by this prospectus in any jurisdiction where action for that purpose is required. The securities offered by this prospectus may not be offered or sold, directly or indirectly, nor may this prospectus or any other offering material or advertisements in connection with the offer and sale of any such securities be distributed or published in any jurisdiction, except under circumstances that will result in compliance with the applicable rules and regulations of that jurisdiction. Persons into whose possession this prospectus comes are advised to inform themselves about and to observe any restrictions relating to the offering and the distribution of this prospectus. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities offered by this prospectus in any jurisdiction in which such an offer or a solicitation is unlawful.
Canada
The common shares and Series II First Preferred Shares described in this prospectus are not being offered, sold or delivered, directly or indirectly, to any resident of Canada or over the Toronto Stock Exchange or otherwise in Canada, and each purchaser of common shares and Series II First Preferred Shares from the underwriters is deemed to agree, upon acceptance of delivery of the purchased common shares or Series II First Preferred Shares by the purchaser or its dealer or other representative, that the purchaser will not resell the purchased common shares or Series II First Preferred Shares, directly or indirectly, to any resident of Canada or (in the case of the common shares) over the Toronto Stock Exchange or otherwise in Canada for a period of 90 days following the completion of this offering, and to represent that it is not a resident of Canada or purchasing the offered shares on behalf of or as agent for any person that is in or a resident of Canada. Each underwriter has agreed that it will not knowingly, directly or indirectly, offer, sell or deliver any common shares or Series II First Preferred Shares purchased by it to any resident of Canada or over the Toronto Stock Exchange or otherwise in Canada, and that it will include a comparable provision in any selling group member agreement that it may enter into.
United Kingdom
This document is only being distributed to and is only directed at (i) persons who are outside the United Kingdom or (ii) investment professionals falling within Article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005 (the “Order”) or (iii) high net worth entities, and other persons to whom it may lawfully be communicated, falling with Article 49(2)(a) to (d) of the Order (all such persons together being referred to as “relevant persons”). The securities are only available to, and any invitation, offer or agreement to subscribe, purchase or otherwise acquire such securities will be engaged in only with, relevant persons. Any person who is not a relevant person should not act or rely on this document or any of its contents.
European Economic Area
In relation to each Member State of the European Economic Area which has implemented the Prospectus Directive (each, a “Relevant Member State”), from and including the date on which the European Union Prospectus Directive (the “EU Prospectus Directive”) was implemented in that Relevant Member State (the “Relevant Implementation Date”) an offer of securities described in this prospectus may not be made to the public in that Relevant Member State prior to the publication of a prospectus in relation to the shares which has been approved by the competent authority in that Relevant Member State or, where appropriate, approved in another Relevant Member State and notified to the competent authority in that Relevant Member State, all in accordance with the EU Prospectus Directive, except that, with effect from and including the Relevant Implementation Date, an offer of securities described in this prospectus may be made to the public in that Relevant Member State at any time:
| • | to any legal entity which is a qualified investor as defined under the EU Prospectus Directive; |
80
Table of Contents
| • | to fewer than 100 or, if the Relevant Member State has implemented the relevant provision of the 2010 PD Amending Directive, 150 natural or legal persons (other than qualified investors as defined in the EU Prospectus Directive); or |
| • | in any other circumstances falling within Article 3(2) of the EU Prospectus Directive; |
provided that no such offer of securities described in this prospectus shall result in a requirement for the publication by us of a prospectus pursuant to Article 3 of the EU Prospectus Directive.
For the purposes of this provision, the expression an “offer of securities to the public” in relation to any securities in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and the securities to be offered so as to enable an investor to decide to purchase or subscribe for the securities, as the same may be varied in that Member State by any measure implementing the EU Prospectus Directive in that Member State. The expression “EU Prospectus Directive” means Directive 2003/71/EC (and any amendments thereto, including the 2010 PD Amending Directive, to the extent implemented in the Relevant Member State) and includes any relevant implementing measure in each Relevant Member State, and the expression “2010 PD Amending Directive” means Directive 2010/73/EU.
Israel
In the State of Israel this prospectus shall not be regarded as an offer to the public to purchase common shares under the Israeli Securities Law, 5728 – 1968, which requires a prospectus to be published and authorized by the Israel Securities Authority, if it complies with certain provisions of Section 15 of the Israeli Securities Law, 5728–1968, including, inter alia, if: (i) the offer is made, distributed or directed to not more than 35 investors, subject to certain conditions (the “Addressed Investors”); or (ii) the offer is made, distributed or directed to certain qualified investors defined in the First Addendum of the Israeli Securities Law, 5728 – 1968, subject to certain conditions (the “Qualified Investors”). The Qualified Investors shall not be taken into account in the count of the Addressed Investors and may be offered to purchase securities in addition to the 35 Addressed Investors. The company has not and will not take any action that would require it to publish a prospectus in accordance with and subject to the Israeli Securities Law, 5728 – 1968. We have not and will not distribute this prospectus or make, distribute or direct an offer to subscribe for our common shares to any person within the State of Israel, other than to Qualified Investors and up to 35 Addressed Investors.
Qualified Investors may have to submit written evidence that they meet the definitions set out in of the First Addendum to the Israeli Securities Law, 5728 – 1968. In particular, we may request, as a condition to be offered common shares, that Qualified Investors will each represent, warrant and certify to us and/or to anyone acting on our behalf: (i) that it is an investor falling within one of the categories listed in the First Addendum to the Israeli Securities Law, 5728 – 1968; (ii) which of the categories listed in the First Addendum to the Israeli Securities Law, 5728 – 1968 regarding Qualified Investors is applicable to it; (iii) that it will abide by all provisions set forth in the Israeli Securities Law, 5728 – 1968 and the regulations promulgated thereunder in connection with the offer to be issued common shares; (iv) that the common shares that will be issued are, subject to exemptions available under the Israeli Securities Law, 5728 – 1968: (a) for its own account; (b) for investment purposes only; and (c) not issued with a view to resale within the State of Israel, other than in accordance with the provisions of the Israeli Securities Law, 5728 – 1968; and (v) that it is willing to provide further evidence of its Qualified Investor status. Addressed Investors may have to submit written evidence in respect of their identity and may have to sign and submit a declaration containing, inter alia, the Addressed Investor’s name, address and passport number or Israeli identification number.
81
Table of Contents
EXPENSES RELATING TO THIS OFFERING
We estimate our expenses related to this offering to be as follows:
SEC registration fee | US$ | 6,682 | ||||
Printing expenses | 85,000 | |||||
Accounting fees and expenses | 125,000 | |||||
Legal fees and expenses | 335,000 | |||||
Financial Industry Regulatory Authority, Inc. filing fee | 9,125 | |||||
| ||||||
Total | US$ | 560,807 | ||||
|
82
Table of Contents
Overview
Our authorized share capital consists of an unlimited number of common shares, Class B Shares, First Preferred Shares, Series I First Preferred Shares and Series II First Preferred Shares, in each case without nominal or par value.
Common Shares
The holders of common shares are entitled to receive notice of and to attend all annual and special meetings of our shareholders and to one vote per share held at each such meeting, and they are entitled to receive dividends as determined and declared by our board of directors.
Subject to the rights of the holders of any other class of our shares entitled to receive dividends in priority to or concurrently with the holders of the common shares, our board of directors may in its sole discretion declare dividends on the common shares to the exclusion of any other class of shares of the Company.
In the event of our liquidation, dissolution or winding up or other distribution of our assets among our shareholders for the purpose of winding up our affairs, the holders of the common shares shall, subject to the rights of the holders of any other class of shares entitled to receive our assets upon such a distribution in priority to or concurrently with the holders of the common shares, be entitled to participate in the distribution. Such distribution shall be made in equal amounts per share on all the common shares at the time outstanding without preference or distinction.
Class B Shares
The holders of the Class B Shares are entitled to receive notice of and to attend any meeting of our shareholders but shall not be entitled to vote any of their Class B Shares at any such meeting. Each issued and fully paid Class B Share may at any time be converted, at the option of the holder, into one common share.
First Preferred Shares
The First Preferred Shares may at any time and from time to time be issued in one or more series and our board of directors may before the issue thereof fix the number of shares in, and determine the designation, rights, privileges, restrictions and conditions attaching to the shares of, each series of First Preferred Shares.
The First Preferred Shares are entitled to priority over the common shares and Class B Shares and all other shares ranking junior to the First Preferred Shares with respect to the payment of dividends and the distribution of our assets in the event of our liquidation, dissolution or winding up or other distribution of our assets among our shareholders for the purpose of winding up our affairs.
The First Preferred Shares of each series rank on a parity with the First Preferred Shares of every other series with respect to priority in the payment of dividends and in the distribution of our assets in the event of our liquidation, dissolution or winding up or other distribution of our assets among our shareholders for the purpose of winding up our affairs.
Series I First Preferred Shares
During 2013, we created a new series of First Preferred Shares, our Series I First Preferred Shares. The holders of Series I First Preferred Shares are not entitled to vote at any meeting of our shareholders (except in limited circumstances provided for in the Business Corporations Act (Ontario)).
The holders of Series I First Preferred Shares are entitled to receive dividends as determined and declared at the discretion of our board of directors equally on a one-for-one basis with the holders of shares of the other series of
83
Table of Contents
First Preferred Shares and, at the discretion of our board of directors, either in priority to, or equally on a share-for-share basis with, holders of our common shares or Class B shares.If any amount of cumulative dividends, whether or not declared, or declared non-cumulative dividends, with respect to shares of a series of our First Preferred Shares is not paid in full, the shares of the series will participate on a pro rata basis with the shares of all other series of that class of shares with respect to all accumulated cumulative dividends, whether or not declared, and all declared non-cumulative dividends.
Each issued and fully paid Series I First Preferred Share may at any time be converted, at the option of the holder, into one common share, subject to adjustment. Following the 30 for 1 share consolidation completed in November 2014, each presently outstanding Series I First Preferred Share may be converted, at the option of the holder into one thirtieth (1/30th) of a common share, subject to further adjustment. Notwithstanding the foregoing, holders of Series I First Preferred Shares will be prohibited from converting Series I First Preferred Shares into common shares if, as a result of such conversion, the holder, together with its affiliates, would own more than 4.99%(which the holder may elect to increase or decrease by written notice to us to any other percentage specified in such notice, provided that any increase (but not decrease) will not be effective until the 61st day after such notice) of the total number of our common shares then issued and outstanding, unless the holder gives us at least 61 days prior notice of an intent to convert into common shares that would cause the holder to own more than 4.99% (or another percentage elected by the holder) of the total number of our common shares then issued and outstanding.
In addition, we will not be required to deliver to a holder any common shares upon a conversion of our Series I First Preferred Shares into common shares if our common shares are then listed and posted for trading on the Toronto Stock Exchange (or the TSX Venture Exchange) and to the extent that the conversion would result in the holder, together with any person acting jointly or in concert with the holder within the meaning of the Securities Act (Ontario), beneficially owning or exercising control or direction over common shares representing more than:
| 1. | 9.99% of our outstanding common shares unless the holder (or, where the holder is not an individual, any director, officer or insider of the holder) has first provided: |
| (a) | the stock exchange with a personal information form pursuant to the rules of that stock exchange and the form has been approved by the stock exchange; and |
| (b) | a copy of the approval of the personal information form by the stock exchange to us; and |
| 2. | 19.99% of our outstanding common shares, unless we have received approval from the stock exchange and the holders of our common shares of the issuance of common shares at a meeting of holders of common shares which we will call, at our expense, in accordance with the applicable policies of the stock exchange. |
In the event of our liquidation, dissolution or winding-up, whether voluntary or involuntary, or in the event of any other distribution of our assets among our shareholders for the purpose of winding-up our affairs, or in the event of a reduction or redemption of our capital stock, the holders of Series I First Preferred Shares are entitled to receive an amount per share equal to that amount of money that we received as consideration for such Series I First Preferred Shares or, in the event that Series I First Preferred Shares were not issued for money, then the amount equal to the fair value of any property we received as consideration for the issuance of such Series I First Preferred Shares divided by the number of Series I First Preferred Shares issued, the whole before any amount shall be paid by us or any of our assets shall be distributed to holders of our common shares and Class B Shares. After such payment, the holders of Series I First Preferred Shares are not entitled to share in any further distribution of our property or assets.If any amount payable on return of capital in the event of our liquidation, dissolution or winding-up in respect of shares of a series of our First Preferred Shares is not paid in full, the shares of the series will participate on a pro rata basis with the shares of all other series of that class of shares with respect to all amounts payable on return of capital in the event of our liquidation, dissolution or winding-up.
84
Table of Contents
Series II First Preferred Shares
During 2015, we created a new series of First Preferred Shares, our Series II First Preferred Shares. The holders of Series II First Preferred Shares are not entitled to vote at any meeting of our shareholders (except in limited circumstances provided for in the Business Corporations Act (Ontario)).
The holders of Series II First Preferred Shares are entitled to receive dividends as determined and declared at the discretion of our board of directors on a parity basis with the holders of shares of the other series of First Preferred Shares and, at the discretion of our board of directors, either in priority to, or equally on a share-for-share basis with, holders of our common shares or Class B shares.If any amount of cumulative dividends, whether or not declared, or declared non-cumulative dividends, with respect to shares of a series of our First Preferred Shares is not paid in full, the shares of the series will participate on a pro rata basis with the shares of all other series of that class of shares with respect to all accumulated cumulative dividends, whether or not declared, and all declared non-cumulative dividends.
Each issued and fully paid Series II First Preferred Share may at any time be converted, at the option of the holder, into one common share, subject to adjustment. Notwithstanding the foregoing, holders of Series II First Preferred Shares will be prohibited from converting Series II First Preferred Shares into common shares if, as a result of such conversion, the holder, together with its affiliates, would own more than 4.99%(which the holder may elect to increase or decrease by written notice to us to any other percentage specified in such notice, provided that any increase (but not decrease) will not be effective until the 61st day after such notice) of the total number of our common shares then issued and outstanding, unless the holder gives us at least 61 days prior notice of an intent to convert into common shares that would cause the holder to own more than 4.99% of the total number of our common shares then issued and outstanding.
In addition, we will not be required to deliver to a holder any common shares upon a conversion of our Series II First Preferred Shares into common shares if our common shares are then listed and posted for trading on the Toronto Stock Exchange (or the TSX Venture Exchange) and to the extent that the conversion would result in the holder, together with any person acting jointly or in concert with the holder within the meaning of the Securities Act (Ontario), beneficially owning or exercising control or direction over common shares representing more than:
| 1. | 9.99% of our outstanding common shares unless the holder (or, where the holder is not an individual, any director, officer or insider of the holder) has first provided: |
| (a) | the stock exchange with a personal information form pursuant to the rules of that stock exchange and the form has been approved by the stock exchange; and |
| (b) | a copy of the approval of the personal information form by the stock exchange to us; and |
| 2. | 19.99% of our outstanding common shares, unless we have received approval from the stock exchange and the holders of our common shares of the issuance of common shares at a meeting of holders of common shares which we will call, at our expense, in accordance with the applicable policies of the stock exchange. |
In the event of our liquidation, dissolution or winding-up, whether voluntary or involuntary, or in the event of any other distribution of our assets among our shareholders for the purpose of winding-up our affairs, or in the event of a reduction or redemption of our capital stock, the holders of Series II First Preferred Shares are entitled to receive an amount per share equal to that amount of money that we received as consideration for such Series II First Preferred Shares or, in the event that Series II First Preferred Shares were not issued for money, then the amount equal to the fair value of any property we received as consideration for the issuance of such Series II First Preferred Shares divided by the number of Series II First Preferred Shares issued, the whole before any amount shall be paid by us or any of our assets shall be distributed to holders of our common shares and Class B
85
Table of Contents
Shares. After such payment, the holders of Series II First Preferred Shares are not entitled to share in any further distribution of our property or assets. If any amount payable on return of capital in the event of our liquidation, dissolution or winding-up in respect of shares of a series of our First Preferred Shares is not paid in full, the shares of the series will participate on a pro rata basis with the shares of all other series of that class of shares with respect to all amounts payable on return of capital in the event of our liquidation, dissolution or winding-up.
Outstanding Securities
As at March 20, 2015, 5,018,139 common shares were outstanding, 64,904,689 Series I First Preferred Shares were outstanding and convertible into 2,163,490 common shares, no Series II First Preferred Shares were outstanding, and no Class B Shares were outstanding. As at March 20, 2015, 125,797,904 common share purchase warrants were outstanding and convertible into 4,193,263 common shares at a weighted average exercise price of $8.66 per common share.
As at March 20, 2015, there were 580,475 stock options outstanding to purchase common shares with aweighted-average exercise price of $9.68 per common share. The terms and conditions of such stock options are contained in the 2014 Stock Option Plan. See “Board of Directors and Management – Stock Option Plan” for a summary of the 2014 Stock Option Plan.
As at March 20, 2015, we have issued 28,777 deferred share units. The terms and conditions of such deferred share units are contained in the 2014 DSU Plan. See “Board of Directors and Management – Deferred Share Unit Plan” for a summary of the 2014 DSU Plan.
Warrants
Our board of directors authorized or ratified the issuances of the warrants set forth in the table below and the issuance of one common share upon the due exercise of each 30 warrants in accordance with its terms and the receipt by us of the designated exercise price payable in respect of the share prior to the time of expiry on the designated expiry date.
As at March 20, 2015, we had the following outstanding warrants (on a post-consolidation basis):
Number of Warrants Outstanding | Exercise Price | Expiry Date | ||
5,014,839 | $6.30 | December 13, 2015 | ||
|
|
| ||
11,659,240 | $12.00 | March 15, 2018 | ||
|
|
| ||
420,000 | $12.00 | March 27, 2018 | ||
|
|
| ||
108,703,825 | $8.40 | December 13, 2018 | ||
|
|
| ||
Total: 125,797,904 | ||||
| ||||
Stock Options
Our board of directors authorized or ratified the issuances of the options set forth in the table below and the issuance of one common share upon the due exercise of each option in accordance with its terms and the receipt by us of the designated exercise price payable in respect of the share prior to the time of expiry on the designated expiry date.
86
Table of Contents
As at March 20, 2015, we had the following outstanding stock options (on a post-consolidation basis):
Number of Stock | Number Exercisable | Exercise Price | Expiry Date | |||
1,667 | 1,667 | $30.00 | June 28, 2016 | |||
|
|
|
| |||
3,334 | 3,334 | $30.00 | July 18, 2016 | |||
|
|
|
| |||
417 | 417 | $30.00 | August 25, 2016 | |||
|
|
|
| |||
333 | 333 | $30.00 | June 1, 2017 | |||
|
|
|
| |||
73,675 | 36,842 | $7.50 | April 8, 2023 | |||
|
|
|
| |||
1,166 | 584 | $7.50 | May 23, 2023 | |||
|
|
|
| |||
6,666 | 2,222 | $15.30 | January 29, 2024 | |||
|
|
|
| |||
13,332 | 4,444 | $18.90 | March 6, 2024 | |||
|
|
|
| |||
264,127 | 88,043 | $10.35 | April 17, 2024 | |||
|
|
|
| |||
215,758 | 71,918 | $8.34 | May 27, 2024 | |||
|
|
|
| |||
Total: 580,475 | Total: 209,804 | |||||
| ||||||
Deferred Share Units
As at March 20, 2015, we had 28,777 (post-consolidated) issued and outstanding DSUs which are convertible into common shares.
Issuances of Securities
Below is information regarding securities sold by us since January 1, 2012. None of the securities sold by us since January 1, 2012 were registered under the Securities Act, and we have made no public offerings in the U.S. Except as noted below, all offers and sales of securities by us were made in offshore transactions pursuant to the exclusion from registration provided by Regulation S under the Securities Act.
Share capital issued – subsequent to December 31, 2014
From January 1, 2015 to March 20, 2015, 12,926,877 warrants were exercised for proceeds of $4,041,928 and 6,666 stock options were exercised for proceeds of $49,995.
Share capital issued – for the year ended December 31, 2014
We consolidated our outstanding common shares issuing one post-consolidated share for each 30 pre-consolidated shares effective November 14, 2014 and the common shares began trading on a post-consolidated basis on November 20, 2014.
2014 Exercise of Warrants and Stock Options and Conversion of Series I Preferred Shares
During the year ended December 31, 2014, 2,596,251 warrants were exercised for proceeds of $946,813 and 2,614 post-consolidated stock options were exercised for proceeds of $19,600. Also, 909,091 warrants issued on March 14, 2011 expired unexercised.
During the year ended December 31, 2014, 8,390,476 post-consolidated Series I Preferred Shares were converted into 279,682 post-consolidated common shares.
Share capital issued – for the year ended December 31, 2013
We consolidated our outstanding common shares issuing one post-consolidated share for each 10 pre-consolidated shares and the common shares began trading on a post-consolidated basis on February 6, 2013.
87
Table of Contents
March 2013 Offering
In March 2013, we completed an offering for a total of 12,735,000 units at a price of $0.25 per unit, for aggregate gross proceeds of $3,185,080 ($2,615,240 net of issuance costs). Each unit consisted of one common share and one common share purchase warrant. Each warrant entitles the holder to purchase one common share at a price of $0.40 per share at any time prior to expiry on March 15, 2018 (12,315,000 warrants) and March 27, 2018 (420,000 warrants).
Euro Pacific Canada Inc. acted as placement agent for the offering and received a commission of $206,603. In connection with the financing, we issued 814,051 compensation warrants to the placement agents having an aggregate fair value of $48,843 estimated using the Black-Scholes option pricing model. Each compensation warrant entitles the holder to acquire one common share at an exercise price of $0.25 per share prior to expiry on March 16, 2015.
The allocation of the $0.25 common share unit issue price to the common shares and unit warrants was based on the relative fair values of the common shares and the warrants. The fair value of the warrants was determined using the Black-Scholes option pricing model. The common shares were allocated a price of $0.20 per share and the warrants were allocated a price of $0.05 per warrant. The costs of the issue were allocated on a pro rata basis to the common shares and warrants. Accordingly, $2,092,192 was allocated to common shares and $523,048 to warrants, net of issuance costs. Assumptions used to determine the value of the unit warrants were: dividend yield of 0%, risk-free interest rate of 1.1%, expected volatility of 64% and average expected life of 3 years. Assumptions used to determine the value of the compensation warrants were: dividend yield of 0%, risk-free interest rate of 1.0%; expected volatility of 80% and average expected life of two years.
The offering was completed pursuant to Regulation S under the Securities Act and Section 4(a)(2) of the Securities Act.
April 2013 Acquisition of Trillium Privateco
On April 9, 2013, we issued 6,079,180 common shares and 3,300,000 common share purchase warrants for partial consideration for the acquisition of Trillium Privateco. See the section of this prospectus entitled “Business – General Development of the Business – Acquisition of Trillium Privateco.”
April 2013 Issuance to UHN
On April 16, 2013, we issued 5,028,571 common shares and 1,600,000 common share purchase warrants as consideration for the acquisition of certain rights related to tigecycline from UHN.
December 2013 Private Placement
On December 13, 2013, we completed a private placement of 79,247,693 common share units (each common share unit consisting of one common share and three-quarters of a common share purchase warrant) at a price of $0.21 per unit and 77,895,165 preferred share units (each preferred share unit consisting of one Series I First Preferred Share and three-quarters of a common share purchase warrant) at a price of $0.21 per unit for gross proceeds of $32,866,025 ($30,713,841 net of issuance costs). Each whole warrant entitles the holder to purchase one common share at a price of $0.28 at any time prior to expiry on December 13, 2018.
Bloom Burton & Co. acted as lead agent for the private placement and ROTH Capital Partners, LLC acted as placement agent in the U.S. The placement agents received a commission of $1,051,616. In connection with the financing, we issued 5,014,839 compensation warrants to the placement and other agents having an aggregate fair value of $651,929 estimated using the Black-Scholes option pricing model. Each compensation warrant entitles the holder to acquire one common share at an exercise price of $0.21 per share prior to expiry on December 13, 2015.
88
Table of Contents
The allocation of the $0.21 unit issue price to the common shares, Series I First Preferred Shares and unit warrants was based on the relative fair values of the common shares, Series I First Preferred Shares and the warrants. The fair value of the warrants was determined using the Black-Scholes option pricing model. The common shares were allocated a price of $0.16 per share, the Series I First Preferred Shares were allocated a price of $0.16 per share, and the warrants were allocated a price of $0.05 per three-quarter warrant. The costs of the issue were allocated on a pro rata basis. Accordingly, $11,488,602 was allocated to common shares, $11,292,525 to the Series I First Preferred Shares and $7,932,714 to warrants, net of issuance costs. Assumptions used to determine the value of the unit warrants were: dividend yield of 0%; risk-free interest rate of 1.2%, expected volatility of 70% and average expected life of 3 years. Assumptions used to determine the value of the compensation warrants were: dividend yield of 0%, risk-free interest rate of 1.1%, expected volatility of 66% and average expected life of two years.
All securities issued under the offering (including the compensation warrants), in Canada were subject to a four month hold and resale restrictions under Canadian securities law, and in the United States are subject to statutory resale restrictions under U.S. securities laws. The offering was completed pursuant to Regulation S under the Securities Act and to the exemption from registration provided by Rule 506(b) of Regulation D under the Securities Act.
In the December 2013 private placement, subscribers who purchased preferred share units and certain subscribers who purchased common share units were subject to restrictions on the conversion and exercise of securities of ours convertible into common shares. Such subscribers cannot convert or exercise securities of ours convertible or exercisable into common shares if, after giving effect to the exercise of conversion, the subscriber and its joint actors would have beneficial ownership or direction or control over common shares in excess of 4.99% of the then outstanding common shares. This limit may be increased at the option of the subscriber on 61 days prior written notice: (i) up to 9.99%, (ii) up to 19.99%, subject to stock exchange clearance of a personal information form submitted by the subscriber and (iii) above 19.99%, subject to stock exchange approval and shareholder approval.
Subject to receipt of any required regulatory approvals, subscribers who purchased a minimum of 10% of the securities sold under the offering have been given rights to purchase securities of ours in future financings to enable each such subscriber to maintain its percentage holding in us for so long as the subscriber holds at least 10% of the outstanding common shares on a fully-diluted basis.
Share capital issued – for the year ended December 31, 2012
There were no common or preferred shares issued in the year ended December 31, 2012.
Shareholder Rights Plan
On October 17, 2013 our shareholders adopted a shareholder rights plan, or the 2013 Rights Plan, and approved certain amendments on May 27, 2014, or the Rights Plan Amendment, and which together with the 2013 Rights Plan we refer to as the Rights Plan.
Purpose of the Rights Plan
Many public companies in Canada have shareholder rights plans in effect. While securities legislation in Canada requires a take-over bid to be open for at least 35 days, our board of directors is concerned that this is too short a time for companies that are subject to unsolicited take-over bids to be able to respond to ensure that shareholders are offered full and fair value for their shares. The Rights Plan is designed to give our shareholders sufficient time to properly assess a take-over bid without undue pressure and to give our board of directors time to consider alternatives designed to allow our shareholders to receive full and fair value for their common shares.
89
Table of Contents
Our board of directors is also concerned that current Canadian take-over bid rules permit a person or company to obtain control or effective control of the Company without treating all shareholders equally. The Rights Plan is not intended to prevent a take-over bid or deter offers for common shares. It is designed to encourage any bidder to provide shareholders with equal treatment and full and fair value for their common shares.
Summary of the Rights Plan
The following is a summary of the principal terms of the Rights Plan, which is qualified in its entirety by reference to the text of the Rights Plan which is filed as an exhibit to this registration statement. All capitalized terms used in this summary without definition have the meanings attributed to them in the Rights Plan.
Effective Date and Term
The 2013 Rights Plan came into effect on September 16, 2013 and the Rights Plan Amendment was approved by the shareholders on May 27, 2014. Subject to periodic confirmation by shareholders as discussed below, the Rights Plan will remain in effect until the termination of our annual meeting in the year 2016.
Issue of Rights
Immediately upon the Rights Plan coming into effect, one right, each a Right, was issued and attached to each common share outstanding and will attach to each common share subsequently issued.
Rights Exercise Privilege
The Rights will separate from the common shares and will be exercisable on the close of business on the tenth trading day, referred to as the Separation Time, after the earlier of the date on which a person has acquired 20% or more of, or a person commences or announces a take-over bid for, our outstanding common shares, other than by an acquisition pursuant to a Permitted Bid (as defined in the Rights Plan) or a Competing Permitted Bid (as defined in the Rights Plan). The acquisition by a person, or Acquiring Person, of 20% or more of the common shares is referred to as a “Flip-in Event”. When a Flip-in Event occurs each Right (except for Rights beneficially owned by an Acquiring Person or certain transferees of an Acquiring Person, which Rights will be void pursuant to the Rights Plan) becomes a right to purchase from us, upon exercise thereof in accordance with the terms of the Rights Plan, that number of common shares having an aggregate market price on the date of consummation or occurrence of such Flip-in Event equal to twice the exercise price for an amount in cash equal to the exercise price. The exercise price for the Rights provided for in the Rights Plan is $100. As an example, if at the time of the Flip-in Event the common shares have a market price of $25.00, the holder of each Right would be entitled to receive $200 (twice the exercise price) in market value of the common shares (8 common shares) for $100, i.e. at a 50% discount.
The issue of the Rights is not initially dilutive. However, upon a Flip-in Event occurring and the Rights separating from the common shares, reported earnings per share may be affected. Holders of Rights not exercising their Rights upon the occurrence of a Flip-in Event may suffer substantial dilution.
Any Rights held by an Acquiring Person will become void upon the occurrence of a Flip-in Event. Any offer other than a Permitted Bid, a competing Permitted Bid or a bid for which our board of directors has waived the application of the Rights Plan to a particular Flip-in Event (see “Waiver” below) will become prohibitively expensive for the Acquiring Person. The Rights Plan is therefore designed to require any person interested in acquiring more than 20% of the common shares to do so by way of a Permitted Bid or a Competing Permitted Bid or to make an offer which our board of directors considers to represent the full and fair value of the common shares.
90
Table of Contents
Exemptions for Portfolio Managers, etc.
Portfolio managers (for fully managed accounts), mutual funds and their managers, trust companies (acting in their capacities as trustees and administrators), statutory bodies whose business includes the management of funds, administrators of registered pension plans and crown agents acquiring greater than 20% of the common shares are exempted from triggering a Flip-in Event, provided that they are not making, and are not part of a group making, a take-over bid.
Grandfathered Person
A person who was the beneficial owner of more than 20% of the outstanding common shares on September 16, 2013 is deemed not to be an Acquiring Person until it ceases to own more than 20% of the common shares or increases its beneficial ownership by more than 1% of the outstanding common shares on September 16, 2013 except in specified circumstances. To the knowledge of our senior officers, we do not have any such grandfathered persons.
Certificates and Transferability
Prior to the Separation Time, the Rights will be evidenced by a legend imprinted on our common share certificates and will not be transferable separately from the common shares. Common share certificates do not need to be exchanged to entitle a shareholder to these Rights. The legend will be on all new certificates we issue after the effective date of the Rights Plan. From and after the Separation Time, the Rights will be evidenced by Rights certificates and will be transferable separately from the common shares.
Permitted Bid Requirements
The Permitted Bid requirements include the following:
| (a) | the take-over bid must be made by way of a take-over bid circular; |
| (b) | the take-over bid must be made to all holders of common shares (other than the bidder); |
| (c) | the take-over bid provides that no common shares tendered pursuant to the take-over bid may be taken up prior to the expiry of a 60 day period following the date of the bid and unless at such date more than 50% of the common shares held by the independent shareholders (i.e. the shareholders, other than the bidder, its affiliates and persons acting jointly or in concert and certain other persons), have been tendered to the take-over bid and not withdrawn; |
| (d) | the take-over bid must be open for acceptance for a minimum period of 60 days; |
| (e) | the common shares deposited pursuant to the bid may be withdrawn until taken up or paid for; and |
| (f) | if the minimum deposit condition described in (iii) above has been satisfied, the bidder must make a public announcement of that fact and the take-over bid must remain open for deposits of common shares for an additional 10 business days from the date of such public announcement. |
The Rights Plan allows for a Competing Permitted Bid, which we refer to as a Competing Permitted Bid to be made while a Permitted Bid is in existence. A Competing Permitted Bid must satisfy all of the requirements of a Permitted Bid except that no common shares will be taken up or paid for pursuant to the Competing Permitted Bid prior to the close of business on a date that is no earlier than the later of:
| (a) | 35 days after the date of the Competing Permitted Bid; and |
91
Table of Contents
| (b) | the 60th day after the earliest date on which any other Permitted Bid that is then in existence was made. |
Waiver
Our board of directors, acting in good faith may, prior to the occurrence of a Flip-in Event, waive the application of the Rights Plan to a particular Flip-in Event where the take-over bid is made by a take-over bid circular to all holders of common shares. Where our board of directors exercises the waiver power for one take-over bid, the waiver will also apply to any other take-over bid for us made by a take-over bid circular to all holders of common shares prior to the expiry of any other bid for which the Rights Plan has been waived.
Redemption
Our board of directors, with the approval of the majority of votes cast by shareholders (or the holders of the Rights if the Separation Time has occurred) voting in person and by proxy, at a meeting duly called for that purpose, may redeem all of the then outstanding Rights at $0.000001 per Right as adjusted by the terms of the Rights Plan. Rights shall be automatically redeemed following completion of a Permitted Bid, Competing Permitted Bid or Exempt Acquisition.
Protection Against Dilution
The Rights Plan contains detailed provisions regarding adjustments to the Exercise Price and the number and nature of the securities that may be purchased upon exercise of Rights outstanding to prevent dilution in the event of certain declarations of dividends, or consolidation of outstanding common shares, issuances of common shares (or other securities or rights) in respect of or in lieu of an exchange for existing common shares or other changes in the common shares.
Amendment
Our board of directors may amend the Rights Plan with the approval of a majority of votes cast by shareholders (or the holders of the Rights if the Separation Time has occurred) voting in person and by proxy at a meeting duly called for that purpose. Our board of directors, without such approval, may correct clerical or typographical errors and, subject to the subsequent approval as noted above at the next meeting of the shareholders (or holders of Rights, as the case may be), may make amendments to the Rights Plan to maintain its validity due to changes in applicable legislation.
92
Table of Contents
MEMORANDUM AND ARTICLES OF INCORPORATION
Incorporation
On November 7, 2013, we were continued, and we became a corporation subsisting, under the OBCA, named Stem Cell Therapeutics Corp. On June 1, 2014, we filed articles of amalgamation to amalgamate with our wholly-owned subsidiary, Trillium Therapeutics Inc., and renamed the combined company Trillium Therapeutics Inc. Our Ontario corporation number is 1916667 and our business number is 864092275.
Objects and Purposes of the Company
Our articles do not contain and are not required to contain a description of our objects and purposes. There is no restriction contained in our articles on the business that we may carry on.
Voting on Certain Proposal, Arrangement, Contract or Compensation by Directors
Other than as disclosed below, neither our articles nor our corporate by-laws restrict our directors’ power to (a) vote on a proposal, arrangement or contract in which the directors are materially interested or (b) to vote with regard to compensation payable to themselves or any other members of their body in the absence of an independent quorum.
Our corporate by-laws provide that a director or officer who: (a) is a party to; or (b) is a director or an officer of, or has a material interest in, any person who is a party to; a material contract or transaction or proposed material contract or transaction with us shall disclose the nature and extent of such director’s or officer’s interest at the time and in the manner provided by the OBCA. Any such contract or transaction or proposed material contract or transaction shall be referred to our board of directors or shareholders for approval in accordance with the OBCA even if such contract or proposed material contract or transaction is one that in the ordinary course of our business would not require approval by our board of directors or shareholders, and a director interested in a contract or transaction so referred to our board of directors shall not attend any part of a meeting of our board of directors during which the contract or transaction is discussed and shall not vote on any resolution to approve such contract or transaction except as provided by the OBCA.
Subject to our articles and any unanimous shareholder agreement, our directors shall be paid such remuneration for their services as our board of directors may from time to time determine. Our directors shall also be entitled to be reimbursed for travelling and other expenses properly incurred by them in attending meetings of our board of directors or any committee thereof.
The OBCA provides that a director who holds a disclosable interest in a contract or transaction into which we have entered or propose to enter shall not attend any part of a meeting of directors during which the contract or transaction is discussed and shall not vote on any resolution to approve the contract or transaction unless it is a contract or transaction: (i) relating primarily to such director’s remuneration as a director of the company or one of our affiliates; (ii) for indemnity or insurance for the benefit of such director in his/her capacity as a director; or (iii) with one of our affiliates.
A director or officer who holds a disclosable interest in a contract or transaction into which we have entered or propose to enter is not accountable to us or our shareholders for any profit or gain realized from the contract or transaction and the contract or transaction is neither void nor voidable by reason only of that relationship or by reason only that the director is present at or is counted to determine the presence of a quorum at the meeting of directors that authorized the contract or transaction, if the director or officer disclosed his or her interest in accordance with the OBCA and the contract or transaction was reasonable and fair to us at the time it was approved.
The OBCA provides that a director or officer generally holds a disclosable interest in a contract or transaction if either (a) the director or officer is a party to the contract or transaction with us and such contract or transaction is
93
Table of Contents
material to us; or (b) the director or officer is a director or an officer of, or has a material interest in, any person who is a party to a material contract or transaction or proposed material contract or transaction with us.
Borrowing Powers of Directors
Our corporate by-laws provide that, if authorized by our directors, we may:
| • | borrow money upon our credit; |
| • | issue, reissue, sell or pledge debt obligations, including bonds, debentures, notes or other evidences of indebtedness or guarantees, whether secured or unsecured; |
| • | give a guarantee on our behalf to secure performance of an obligation of any person; and |
| • | mortgage, hypothecate, pledge or otherwise create a security interest in all or any currently owned or subsequently acquired real or personal, movable or immovable, property of the Company including book debts, rights, powers, franchises and undertakings, to secure any such bonds, debentures, notes or other evidences of indebtedness or guarantee or any other present or future indebtedness, liability or obligation of the Company. |
Amendment to the borrowing powers described above requires an amendment to our corporate by-laws. Our corporate by-laws do not contain any provisions in connection with amending the by-laws. The OBCA provides that our board of directors may by resolution, make, amend or repeal any by-laws that regulate our business and affairs and that our board of directors will submit such by-law, amendment or repeal to our shareholders at the next meeting of shareholders and the shareholders may, by ordinary resolution, confirm, reject or amend the by-law, amendment or repeal.
Qualifications of Directors
Under our articles and corporate by-laws, a director is not required to hold a share in our capital as qualification for his or her office but must be qualified as required by the OBCA to become, act or continue to act as a director. The OBCA provides that the following persons are disqualified from being a director of a corporation: (i) a person who is less than 18 years of age; (ii) a person who has been found under the Substitute Decisions Act, 1992 or under the Mental Health Act to be incapable of managing property or who has been found to be incapable by a court in Canada or elsewhere; (iii) a person who is not an individual; and (iv) a person who has the status of a bankrupt.
Share Rights
See the discussion in the section of this prospectus entitled “Description of Share Capital” for a summary of our authorized capital and the rights attached to our common shares and preferred shares.
Procedures to Change the Rights of Shareholders
The rights, privileges, restrictions and conditions attaching to our shares are contained in our articles and such rights, privileges, restrictions and conditions may be changed by amending our articles. In order to amend our articles, the OBCA requires a resolution to be passed by a majority of not less than two-thirds of the votes cast by the shareholders entitled to vote thereon. In addition, if we resolve to make particular types of amendments to our articles, a holder of our shares may dissent with regard to such resolution and, if such shareholder so elects, we would have to pay such shareholder the fair value of the shares held by the shareholder in respect of which the shareholder dissents as of the close of business on the day before the resolution was adopted. The types of
94
Table of Contents
amendments that would be subject to dissent rights include without limitation: (i) to add, remove or change restrictions on the issue, transfer or ownership of shares of a class or series of our shares; and (ii) to add, remove or change any restriction upon the business that we may carry on or upon the powers that we may exercise.
Meetings
Each director holds office until our next annual general meeting or until his office is earlier vacated in accordance with our articles or with the provisions of the OBCA. A director appointed or elected to fill a vacancy on our board also holds office until our next annual general meeting.
Annual meetings of our shareholders must be held at such time in each year not more than 15 months after the last annual meeting, as our board of directors may determine. Notice of the time and place of a meeting of shareholders must be sent not less than twenty-one days and not more than fifty days, before the meeting.
Meetings of our shareholders shall be held at our registered office or, if our board of directors shall so determine, at some other place in Ontario or, at some place outside Ontario if all the shareholders entitled to vote at the meeting so agree.
Our board of directors, the chair of our board, our chief executive officer, or our president shall have power to call a special meeting of our shareholders at any time.
The OBCA provides that our shareholders may requisition a special meeting in accordance with the OBCA. The OBCA provides that the holders of not less than five percent of our issued shares that carry the right to vote at a meeting may requisition our directors to call a special meeting of shareholders for the purposes stated in the requisition.
Under our by-laws, the quorum for the transaction of business at a meeting of our shareholders is two or more persons, present in person or by proxy and holding in aggregate not less than 33 1/3% of our issued shares entitled to vote at such meeting.
Limitations on Ownership of Securities
Except as provided in the Investment Canada Act (Canada), there are no limitations specific to the rights of non-Canadians to hold or vote our shares under the laws of Canada or Ontario, or in our charter documents.
Change in Control
We refer you to the Rights Plan described in the section of this prospectus entitled “Description of Share Capital – Shareholder Rights Plan”. There are no provisions in our articles or by-laws that would have the effect of delaying, deferring or preventing a change in control of the Company, and that would operate only with respect to a merger, acquisition or corporate restructuring involving the Company or our subsidiaries.
Ownership Threshold
Neither our by-laws nor our articles contain any provisions governing the ownership threshold above which shareholder ownership must be disclosed. In addition, securities legislation in Canada requires that we disclose in our proxy information circular for our annual meeting and certain other disclosure documents filed by us under such legislation, holders who beneficially own more than 10% of our issued and outstanding shares.
United States federal securities laws require us to disclose, in our annual reports on Form 20-F, holders who own 5% or more of our issued and outstanding voting shares.
95
Table of Contents
Differences in Corporate Law
We are governed by the OBCA, which is generally similar to laws applicable to United States corporations. Significant differences between the OBCA and the Delaware General Corporate Law, or DGCL, which governs companies incorporated in the State of Delaware, include the following:
Number and Election of Directors | ||
Delaware | Ontario | |
| Under the DGCL, the board of directors must consist of at least one number. The number of directors shall be fixed by the bylaws of the corporation, unless the certificate of incorporation fixes the number of directors, in which case a change in the number of directors shall only be made by an amendment of the certificate of incorporation. Under the DGCL, directors are elected at annual stockholder meetings by plurality vote of the stockholders, unless a shareholder- adopted bylaw prescribes a different required vote. | Under the OBCA, the board of directors must consist of at least three members so long as Trillium remains an “offering corporation” for purposes of the OBCA, which includes a corporation whose securities are listed on a recognized stock exchange such as the NASDAQ Stock Exchange or Toronto Stock Exchange. Under the OBCA, the shareholders of a corporation elect directors by ordinary resolution at each annual meeting of shareholders at which such an election is required. | |
| Removal of Directors | ||
| Delaware | Ontario | |
| Under the DGCL, any or all directors may be removed with or without cause by the holders of a majority of shares entitled to vote at an election of directors unless the certificate of incorporation otherwise provides or in certain other circumstances if the corporation has cumulative voting. | Under the OBCA, the shareholders of a corporation may, by resolution passed by a majority of the vote cast thereon at a meeting of shareholders, remove a director and may elect any qualified person to fill the resulting vacancy. | |
| Vacancies on the Board of Directors | ||
| Delaware | Ontario | |
| Under the DGCL, vacancies and newly created directorships resulting from an increase in the authorized number of directors, may be filled by a majority of the directors then in office, although less than a quorum, or by a sole remaining director. | Under the OBCA, vacancies that exist on the board of directors may generally be filled by the board if the remaining directors constitute a quorum. In the absence of a quorum, the remaining directors shall call a meeting of shareholders to fill the vacancy. | |
| Board of Director Quorum and Vote Requirements | ||
| Delaware | Ontario | |
| Under the DGCL, a majority of the total number of directors shall constitute a quorum for the transaction of business unless the certificate or bylaws require a greater number. The bylaws may lower the number required for a quorum to one-third the number of directors, but no less. | Under the OBCA, subject to an Ontario corporation’s articles or bylaws, a majority of the number of directors or minimum number of directors required by the articles constitutes a quorum at any meeting of directors, but in no case shall a quorum be less than two-fifths of the number of directors or minimum number of directors, as the case may be. Where a corporation has fewer than three directors, all directors must directors must be present at any meeting to constitute a quorum. | |
96
Table of Contents
| Under the DGCL, the board of directors may take action by the majority vote of the directors present at a meeting at which a quorum is present unless the certificate of incorporation or bylaws require a greater vote. | Under the OBCA, subject to an Ontario corporation’s articles or bylaws, where there is a vacancy or vacancies in the board of directors, the remaining directors may exercise all the powers of the board so long as a quorum of the board remains in office. | |
| Transactions with Directors and Officers | ||
| Delaware | Ontario | |
| The DGCL generally provides that no transaction between a corporation and one or more of its directors or officers, or between a corporation and any other corporation or other organization in which one or more of its directors or officers, are directors or officers, or have a financial interest, shall be void or voidable solely for this reason, or solely because the director or officer is present at or participates in the meeting of the board or committee which authorizes the transaction, or solely because any such director’s or officer’s votes are counted for such purpose, if (i) the material facts as to the director’s or officer’s interest and as to the transaction are known to the board of directors or the committee, and the board or committee in good faith authorizes the transaction by the affirmative votes of a majority of the disinterested directors, even though the disinterested directors be less than a quorum (ii) the material facts as to the director’s or officer’s interest and as to the transaction are disclosed or are known to the stockholders entitled to vote thereon, and the transaction is specifically approved in good faith by vote of the stockholders; or (iii) the transaction is fair as to the corporation as of the time it is authorized, approved or ratified, by the board of directors, a committee or the stockholders. | The OBCA requires that a director or officer of a corporation who is: (i) a party to a material contract or transaction or proposed material contract or transaction with the corporation; or (ii) a director or an officer of, or has a material interest in, any person who is a party to a material contract to or transaction or proposed material contract or transaction with the corporation shall disclose in writing to the corporation or request to have entered in the minutes of meetings of directors the nature and extent of his or her interest. An interested director is prohibited from attending the part of the meeting during which the contract or transaction is discussed and is prohibited from voting on a resolution to approve the contract or transaction except in specific circumstances, such as a contract or transaction relating primarily to his or her remuneration as a director, a contract or transaction for indemnification or liability insurance of the director, or a contract or transaction with an affiliate of the corporation. If a director or officer has disclosed his or her interest in accordance with the OBCA and the contract or transaction was reasonable and fair to the corporation at the time it was approved, the director or officer is not accountable to the corporation or its shareholders for any profit or gain realized from the contract or transaction and the contract or transaction is neither void nor voidable by reason only of the interest of the director or officer or that the director is present at or is counted to determine the presence of a quorum at the meeting of directors that authorized the contract or transaction. | |
| The OBCA further provides that even if a director or officer does not disclose his or her interest in accordance with the OBCA, or (in the case of a director) votes in respect of a resolution on a contract or transaction in which he or she is interested contrary to the OBCA, if the director or officer acted honestly and in good faith and the contract or transaction was reasonable and fair to the corporation at the time it was approved, the director or officer is | ||
97
Table of Contents
| not accountable to the corporation or to its shareholders for any profit or gain realized from the contract or transaction by reason only of his or her holding the office of the director or officer and the contract or transaction is not by reason only of the director’s or officer’s interest therein void or voidable, if the contract or transaction has been confirmed or approved by the shareholders by special resolution, on the basis of disclosure in reasonable detail of the nature and extent of the director’s or officer’s interest in the notice of meeting or management information circular. | ||
| Limitation on Liability of Directors | ||
| Delaware | Ontario | |
| The DGCL permits a corporation to include a provision in its certificate of incorporation eliminating or limiting the personal liability of a director to the corporation or its stockholders for monetary damages for a breach of the director’s fiduciary duty as a director, except for liability: | The OBCA does not permit the limitation of a director’s liability as the DGCL does. | |
• for breach of the director’s duty of loyalty to the corporation or its stockholders; | ||
• for acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of the law; | ||
• under Section 174 of the DGCL, which concerns unlawful payment of dividends, stock purchases or redemptions; or | ||
• for any transaction from which the director derived an improper personal benefit | ||
Indemnification of Directors and Officers
| Delaware | Ontario | |
| The DGCL permits indemnification for derivative suits only for expenses (including legal fees) and only if the person is not found liable, unless a court determines the person is fairly and reasonably entitled to the indemnification. | Under the OBCA, an Ontario corporation may also, with the approval of a court, indemnify or advance moneys to an Indemnified Person in respect of an action by or on behalf of the corporation to obtain a judgment in its favor, to which the Indemnified Person is made a party because of his or her association with the corporation or other entity, against all costs, charges and expenses reasonably incurred by the Indemnified Person in connection | |
98
Table of Contents
| with such action, if he or she acted honestly and in good faith with a view to the best interests of the corporation or, as the case may be, to the best interests of any other entity for which the Indemnified Person acted as a director or officer or in a similar capacity at the corporation’s request. However, any such Indemnified Person is entitled under the OBCA to indemnity from the corporation in respect of all costs, charges and expenses reasonably incurred by the Indemnified Person in connection with the defense of any civil, criminal, administrative, investigative or other proceeding to which he or she is subject because of his or her association with the corporation or other entity, if such Indemnified Person (i) was not judged by a court or other competent authority to have committed any fault or omitted to do anything that the individual ought to have done, and (ii) acted honestly and in good faith with a view to the best interests of the corporation or other entity and had reasonable grounds for believing that his or her conduct was lawful. | ||
| Call and Notice of Stockholder Meetings | ||
| Delaware | Ontario | |
| Under the DGCL, an annual or special stockholder meeting is held on such date, at such time and at such place as may be designated by the board of directors or any other person authorized to call such meeting under the corporation’s certificate of incorporation or bylaws. If an annual meeting for election of directors is not held on the date designated or an action by written consent to elect directors in lieu of an annual meeting has not been taken within 30 days after the date designated for the annual meeting, or if no date has been designated, for a period of 13 months after the later of the last annual meeting or the last action by written consent to elect directors in lieu of an annual meeting, the Delaware Court of Chancery may summarily order a meeting to be held upon the application of any stockholder or director. | Under the OBCA, the directors of a corporation are required to call an annual meeting of shareholders no later than fifteen months after holding the last preceding annual meeting. Under the OBCA, the directors of a corporation may call a special meeting at any time. In addition, holders of not less than five percent of the issued shares of a corporation that carry the right to vote at a meeting sought to be held may requisition the directors to call a meeting of shareholders. | |
Stockholder Action by Written Consent
| Delaware | Ontario | |
| Under the DGCL, a majority of the stockholders of a corporation may act by written consent without a meeting unless such action is prohibited by the corporation’s certificate of incorporation. | Under the OBCA, a written resolution signed by all the shareholders of a corporation who would have been entitled to vote on the resolution at a meeting is effective to approve the resolution. | |
99
Table of Contents
| Stockholder Nominations and Proposals | ||
| Delaware | Ontario | |
| Not applicable. | Under the OBCA, a shareholder entitled to vote at a shareholders’ meeting may submit a shareholder proposal relating to matters which the shareholder wishes to propose and discuss at a shareholders’ meeting and, subject to such shareholder’s compliance with the prescribed time periods and other requirements of the OBCA pertaining to shareholder proposals, the corporation is required to include such proposal in the information circular pertaining to any meeting at which it solicits proxies, subject to certain exceptions. Notice of such a proposal must be provided to the corporation at least 60 days before the anniversary date of the last annual shareholders’ meeting, or at least 60 days before any other meeting at which the matter is proposed to be raised. | |
| In addition, the OBCA requires that any shareholder proposal that includes nominations for the election of directors must be signed by one or more holders of shares representing in the aggregate not less than five per cent of the shares or five per cent of the shares of a class or series of shares of the corporation entitled to vote at the meeting to which the proposal is to be presented. | ||
Stockholder Quorum and Vote Requirements
| Delaware | Ontario | |
| Under the DGCL, quorum for a stock corporation is a majority of the shares entitled to vote at the meeting unless the certificate of incorporation or bylaws specify a different quorum, but in no event may a quorum be less than one-third of the shares entitled to vote. Unless the DGCL, certificate of incorporation or bylaws provide for a greater vote, generally the required vote under the DGCL is a majority of the shares present in person or represented by proxy, except for the election of directors which requires a plurality of the votes cast. | Under the OBCA, unless the bylaws otherwise provide, the holders of a majority of the shares of an OBCA corporation entitled to vote at a meeting of shareholders, whether present in person or represented by proxy, constitute a quorum. | |
100
Table of Contents
Amendment of Governing Instrument
| Delaware | Ontario | |
| Amendment of Certificate of Incorporation. Generally, under the DGCL, the affirmative vote of the holders of a majority of the outstanding stock entitled to vote is required to approve a proposed amendment to the certificate of incorporation, following the adoption of the amendment by the board of directors of the corporation, provided that the certificate of incorporation may provide for a greater vote. Under the DGCL, holders of outstanding shares of a class or series are entitled to vote separately on an amendment to the certificate of incorporation if the amendment would have certain consequences, including changes that adversely affect the rights and preferences of such class or series. | Amendment of Articles. Under the OBCA, amendments to the articles of incorporation generally require the approval of not less than two-thirds of the votes cast by shareholders entitled to vote on the resolution. | |
| Amendment of Bylaws. Under the DGCL, after a corporation has received any payment for any of its stock, the power to adopt, amend or repeal bylaws shall be vested in the stockholders entitled to vote; provided, however, that any corporation nay, in its certificate of incorporation, provide that bylaws may be adopted, amended or repealed by the board of directors. The fact that such power has been conferred upon the board of directors shall not divest the stockholders of the power nor limit their power to adopt, amend or repeal the bylaws. | Amendment of Bylaws. Under the OBCA, the directors may, by resolution, make, amend or repeal any bylaws that regulate the business or affairs of a corporation and they must submit the bylaw, amendment or repeal to the shareholders at the next meeting of shareholders, and the shareholders may confirm, reject or amend the bylaw, amendment or repeal. | |
| Votes on Mergers, Consolidations and Sales of Assets | ||
| Delaware | Ontario | |
| The DGCL provides that, unless otherwise provided in the certificate of incorporation or bylaws, the adoption of a merger agreement requires the approval of a majority of the outstanding stock of the corporation entitled to vote thereon. | Under the OBCA, the approval of at least two-thirds of votes cast by shareholders entitled to vote on the resolution is required for extraordinary corporate actions. Extraordinary corporate actions include: amalgamations; continuances; sales, leases or exchanges of all or substantially all of the property of a corporation; liquidations and dissolutions. | |
| Dissenter’s Rights of Appraisal | ||
| Delaware | Ontario | |
| Under the DGCL, a stockholder of a Delaware corporation generally has the right to dissent flume, merger or consolidation in which the Delaware corporation is participating, subject to specified procedural requirements, including that such dissenting | Under the OBCA each of the following matters listed will entitle shareholders to exorcise rights of dissent and to be paid the fair value of their shares: (i) any amalgamation with another corporation (other than with certain affiliated corporations); (ii) an | |
101
Table of Contents
| stockholder does not vote in favor of the merger or consolidation. However, the DGCL does not confer appraisal rights, in certain circumstances, including if the dissenting stockholder owns shares traded on a national securities exchange and will receive publicly traded shares in the merger or consolidation. Under the DGCL, a stockholder asserting appraisal rights does not receive any payment for his or her shares until the court determines the fair value or the parties otherwise agree to a value. The costs of the proceeding may be determined by the court and assessed against the parties as the court deems equitable under the circumstances. | amendment to the corporation’s articles to add, change or remove any provisions restricting the issue, transfer or ownership of that class of shares; (iii) an amendment to the corporation’s articles to add, change or remove any restriction upon the business or businesses that the corporation may carry on; (iv) a continuance under the laws of another jurisdiction; (v) a sale, lease or exchange of all or substantially all the property of the corporation other than in the ordinary course of business; and (vi) where a court order permits a shareholder to dissent in connection with an application to the court for an order approving an arrangement. | |
| However, a shareholder is not entitled to dissent if an amendment to the articles is effected by a court order approving a reorganization or by a court order made in connection with an action for an oppression remedy, unless otherwise authorized by the court. The OBCA provides these dissent rights for both listed and unlisted shares. | ||
| Under the OBCA, a shareholder may, in addition to exercising dissent rights, seek an oppression remedy for any act or omission of a corporation which is oppressive or unfairly prejudicial to or that unfairly disregards a shareholder’s interests. | ||
| Anti-Takeover and Ownership Provisions | ||
| Delaware | Ontario | |
| Unless an issuer opts out of the provisions of Section 203 of the DGCL, Section 203 generally prohibits a public Delaware corporation from engaging in a “business combination” with a holder of 15% or more of the corporation’s voting stock (as defined in Section 203), referred to as an interested stockholder, for a period of three years after the date of the transaction in which the interested stockholder became an interested stockholder, except as otherwise provided in Section 203. For these purposes, the term “business combination” includes mergers, assets sales and other similar transactions with an interested stockholder. | While the OBCA does not contain specific anti- takeover provisions with respect to “business combinations”, roles and policies of certain Canadian securities regulatory authorities, including Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions, or Multilateral Instrument 61-101, contain requirements in connection with, among other things, ‘related party transactions” and “business combinations”, including, among other things, any transaction by which an issuer directly or indirectly engages in the following with a related party: acquires, sells, leases or transfers an asset, acquires the related party, acquires or issues treasury securities, amends the terms of a security if the security is owned by the related party or assumes or becomes subject to a liability or takes certain other actions with respect to debt. | |
102
Table of Contents
| The term “related party” includes directors, senior officers and holders of more than 10% of the voting rights attached to all outstanding voting securities of the issuer or holders of a sufficient number of any securities of the issuer to materially affect control of the issuer. | ||
| Multilateral Instrument 61-101 requires, subject to certain exceptions, the preparation of a formal valuation relating to certain aspects of the transaction and more detailed disclosure in the proxy material sent to security holders in connection with a related party transaction including related to the valuation. Multilateral Instrument 61-101 also requires, subject to certain exceptions, that an issuer not engage in a related party transaction unless the shareholders of the issuer, other than the related parties, approve the transaction by a simple majority of the votes cast. | ||
103
Table of Contents
CERTAIN U.S. FEDERAL INCOME TAX CONSIDERATIONS
The following is a general summary of certain material U.S. federal income tax considerations applicable to a U.S. Holder, as defined below, arising from and relating to the acquisition, ownership, and disposition of our offered shares acquired in the offering.
This summary is for general information purposes only and does not purport to be a complete analysis or listing of all potential U.S. federal income tax considerations that may apply to a U.S. Holder arising from and relating to the acquisition, ownership, and disposition of our offered shares. In addition, this summary does not take into account the individual facts and circumstances of any particular U.S. Holder that may affect the U.S. federal income tax consequences to such U.S. Holder, including, without limitation, specific tax consequences to a U.S. Holder under an applicable income tax treaty. Accordingly, this summary is not intended to be, and should not be construed as, legal or U.S. federal income tax advice with respect to any U.S. Holder. This summary does not address the U.S. federal alternative minimum, U.S. federal estate and gift, U.S. state and local, and non-U.S. tax consequences to U.S. Holders of the acquisition, ownership, and disposition of our offered shares. In addition, except as specifically set forth below, this summary does not discuss applicable tax reporting requirements. Each prospective U.S. Holder should consult its own tax advisors regarding the U.S. federal, U.S. federal alternative minimum, U.S. federal estate and gift, U.S. state and local, and non-U.S. tax consequences relating to the acquisition, ownership and disposition of our offered shares.
No ruling from the Internal Revenue Service, or IRS, has been requested, or will be obtained, regarding the U.S. federal income tax consequences of the acquisition, ownership, and disposition of our offered shares. This summary is not binding on the IRS, and the IRS is not precluded from taking a position that is different from, and contrary to, the positions taken in this summary. In addition, because the authorities on which this summary is based are subject to various interpretations, the IRS and the U.S. courts could disagree with one or more of the conclusions described in this summary.
Scope of this Summary
Authorities
This summary is based on the Internal Revenue Code of 1986, as amended, or the Code, Treasury Regulations (whether final, temporary, or proposed), published rulings of the IRS, published administrative positions of the IRS, the Convention Between Canada and the United States of America with Respect to Taxes on Income and Capital, signed September 26, 1980, as amended, or the Treaty, and U.S. court decisions that are applicable, and, in each case, as in effect and available, as of the date hereof. Any of the authorities on which this summary is based could be changed in a material and adverse manner at any time, and any such change could be applied retroactively. This summary does not discuss the potential effects, whether adverse or beneficial, of any proposed legislation.
U.S. Holders
For purposes of this summary, the term “U.S. Holder” means a beneficial owner of our offered shares acquired in the offering that is for U.S. federal income tax purposes:
| • | an individual who is a citizen or resident of the United States; |
| • | a corporation (or other entity treated as a corporation for U.S. federal income tax purposes) organized under the laws of the United States, any state thereof or the District of Columbia; |
| • | an estate whose income is subject to U.S. federal income taxation regardless of its source; or |
| • | a trust that (a) is subject to the primary supervision of a court within the United States and the control of one or more U.S. persons for all substantial decisions or (b) has a valid election in effect under applicable Treasury Regulations to be treated as a U.S. person. |
104
Table of Contents
U.S. Holders Subject to Special U.S. Federal Income Tax Rules Not Addressed
This summary does not address the U.S. federal income tax considerations applicable to U.S. Holders that are subject to special provisions under the Code, including, but not limited to, U.S. Holders that: (a) are tax-exempt organizations, qualified retirement plans, individual retirement accounts, or other tax-deferred accounts; (b) are financial institutions, underwriters, insurance companies, real estate investment trusts, or regulated investment companies; (c) are broker-dealers, dealers, or traders in securities or currencies that elect to apply a mark-to-market accounting method; (d) have a “functional currency” other than the U.S. dollar; (e) own our offered shares as part of a straddle, hedging transaction, conversion transaction, constructive sale, or other arrangement involving more than one position; (f) acquire our offered shares in connection with the exercise of employee stock options or otherwise as compensation for services; (g) hold our offered shares other than as a capital asset within the meaning of Section 1221 of the Code (generally, property held for investment purposes); or (h) own, have owned or will own (directly, indirectly, or by attribution) 10% or more of the total combined voting power of our outstanding shares. This summary also does not address the U.S. federal income tax considerations applicable to U.S. Holders who are: (a) U.S. expatriates or former long-term residents of the U.S.; (b) persons that have been, are, or will be a resident or deemed to be a resident in Canada for purposes of the Income Tax Act (Canada), or the ITA; (c) persons that use or hold, will use or hold, or that are or will be deemed to use or hold our offered shares in connection with carrying on a business in Canada; (d) persons whose offered shares constitute “taxable Canadian property” under the ITA; or (e) persons that have a permanent establishment in Canada for the purposes of the Treaty. U.S. Holders that are subject to special provisions under the Code, including, but not limited to, U.S. Holders described immediately above, should consult their own tax advisors regarding the U.S. federal, U.S. federal alternative minimum, U.S. federal estate and gift, U.S. state and local, and non-U.S. tax consequences relating to the acquisition, ownership and disposition of our offered shares.
If an entity or arrangement that is classified as a partnership (or other “pass-through” entity) for U.S. federal income tax purposes holds our offered shares, the U.S. federal income tax consequences to such entity or arrangement and the partners (or other owners or participants) of such entity or arrangement generally will depend on the activities of the entity or arrangement and the status of such partners (or owners or participants). This summary does not address the tax consequences to any such partner (or owner or participants). Partners (or other owners or participants) of entities or arrangements that are classified as partnerships or as “pass-through” entities for U.S. federal income tax purposes should consult their own tax advisors regarding the U.S. federal income tax consequences arising from and relating to the acquisition, ownership, and disposition of our offered shares.
Passive Foreign Investment Company Rules
PFIC Status
If we were to constitute a “passive foreign investment company” under the meaning of Section 1297 of the Code, or PFIC, for any year during a U.S. Holder’s holding period, then certain potentially adverse rules may affect the U.S. federal income tax consequences to a U.S. Holder as a result of the acquisition, ownership and disposition of our offered shares. We believe that we were classified as a PFIC during the tax year ended December 31, 2014, and, based on current business plans and financial expectations, expect that we will be a PFIC for the current tax year and may be a PFIC in future tax years. The determination of whether any corporation was, or will be, a PFIC for a tax year depends, in part, on the application of complex U.S. federal income tax rules, which are subject to differing interpretations. In addition, whether any corporation will be a PFIC for any tax year depends on the assets and income of such corporation over the course of each such tax year and, as a result, cannot be predicted with certainty as of the date hereof. Accordingly, there can be no assurance that the IRS will not challenge any determination made by us (or any subsidiary) concerning its PFIC status. Each U.S. Holder should consult its own tax advisors regarding our PFIC status and the PFIC status of our subsidiaries.
In any year in which we are classified as a PFIC, a U.S. Holder will be required to file an annual report with the IRS containing such information as Treasury Regulations and/or other IRS guidance may require. In addition to
105
Table of Contents
penalties, a failure to satisfy such reporting requirements may result in an extension of the time period during which the IRS can assess a tax. U.S. Holders should consult their own tax advisors regarding the requirements of filing such information returns under these rules, including the requirement to file an IRS Form 8621.
We generally will be a PFIC if, for a tax year, (a) 75% or more of our gross income is passive income, or the “income test”, or (b) 50% or more of the value of our assets either produce passive income or are held for the production of passive income, based on the quarterly average of the fair market value of such assets, or the “asset test”. “Gross income” generally includes all sales revenues less the cost of goods sold, plus income from investments and from incidental or outside operations or sources, and “passive income” generally includes, for example, dividends, interest, certain rents and royalties, certain gains from the sale of stock and securities, and certain gains from commodities transactions.
For purposes of the PFIC income test and asset test described above, if we own, directly or indirectly, 25% or more of the total value of the outstanding shares of another corporation, we will be treated as if we (a) held a proportionate share of the assets of such other corporation and (b) received directly a proportionate share of the income of such other corporation. In addition, for purposes of the PFIC income test and asset test described above, and assuming certain other requirements are met, “passive income” does not include certain interest, dividends, rents, or royalties that are received or accrued by us from certain “related persons” (as defined in Section 954(d)(3) of the Code) also organized in Canada, to the extent such items are properly allocable to the income of such related person that is not passive income.
Under certain attribution rules, if we are a PFIC, U.S. Holders will generally be deemed to own their proportionate share of our direct or indirect equity interest in any company that is also a PFIC, or a Subsidiary PFIC, and will generally be subject to U.S. federal income tax on their proportionate share of (a) any “excess distributions,” as described below, on the stock of a Subsidiary PFIC and (b) a disposition or deemed disposition of the stock of a Subsidiary PFIC by us or another Subsidiary PFIC, both as if such U.S. Holders directly held the shares of such Subsidiary PFIC. In addition, U.S. Holders may be subject to U.S. federal income tax on any indirect gain realized on the stock of a Subsidiary PFIC on the sale or disposition of our offered shares. Accordingly, U.S. Holders should be aware that they could be subject to tax under the PFIC rules even if no distributions are received and no redemptions or other dispositions of our offered shares are made.
Default PFIC Rules Under Section 1291 of the Code
If we are a PFIC for any tax year during which a U.S. Holder owns our offered shares, the U.S. federal income tax consequences to such U.S. Holder of the acquisition, ownership, and disposition of our offered shares will depend on whether and when such U.S. Holder makes an election to treat us and each Subsidiary PFIC, if any, as a “qualified electing fund” or “QEF” under Section 1295 of the Code, or QEF Election, or makes a mark-to-market election under Section 1296 of the Code, or Mark-to-Market Election. A U.S. Holder that does not make either a QEF Election or a Mark-to-Market Election will be referred to in this summary as a “Non-Electing U.S. Holder.”
A Non-Electing U.S. Holder will be subject to the rules of Section 1291 of the Code (described below) with respect to (a) any gain recognized on the sale or other taxable disposition of our offered shares and (b) any “excess distribution” received on our offered shares. A distribution generally will be an “excess distribution” to the extent that such distribution (together with all other distributions received in the current tax year) exceeds 125% of the average distributions received during the three preceding tax years (or during a U.S. Holder’s holding period for our offered shares, if shorter).
Under Section 1291 of the Code, any gain recognized on the sale or other taxable disposition of our offered shares (including an indirect disposition of the stock of any Subsidiary PFIC), and any “excess distribution” received on our offered shares or with respect to the stock of a Subsidiary PFIC, must be ratably allocated to each day in a Non-Electing U.S. Holder’s holding period for the respective offered shares. The amount of any such gain or excess distribution allocated to the tax year of disposition or distribution of the excess distribution
106
Table of Contents
and to years before the entity became a PFIC, if any, would be taxed as ordinary income (and not eligible for certain preferred rates). The amounts allocated to any other tax year would be subject to U.S. federal income tax at the highest tax rate applicable to ordinary income in each such year, and an interest charge would be imposed on the tax liability for each such year, calculated as if such tax liability had been due in each such year. A Non-Electing U.S. Holder that is not a corporation must treat any such interest paid as “personal interest,” which is not deductible.
If we are a PFIC for any tax year during which a Non-Electing U.S. Holder holds our offered shares, we will continue to be treated as a PFIC with respect to such Non-Electing U.S. Holder, regardless of whether we cease to be a PFIC in one or more subsequent tax years. A Non-Electing U.S. Holder may terminate this deemed PFIC status by electing to recognize gain (which will be taxed under the rules of Section 1291 of the Code discussed above), but not loss, as if such offered shares were sold on the last day of the last tax year for which we were a PFIC.
QEF Election
A U.S. Holder that makes a timely and effective QEF Election for the first tax year in which the holding period of its offered shares begins generally will not be subject to the rules of Section 1291 of the Code discussed above with respect to its offered shares. A U.S. Holder that makes a timely and effective QEF Election will be subject to U.S. federal income tax on such U.S. Holder’s pro rata share of (a) our net capital gain, which will be taxed as long-term capital gain to such U.S. Holder, and (b) our ordinary earnings, which will be taxed as ordinary income to such U.S. Holder. Generally, “net capital gain” is the excess of (a) net long-term capital gain over (b) net short-term capital loss, and “ordinary earnings” are the excess of (a) “earnings and profits” over (b) net capital gain. The IRS has not issued rules regarding the allocation of net capital gain and ordinary earnings amounts to multiple classes of stock. Accordingly, the proper manner for allocating such items between our common shares and Series II First Preferred Shares is not certain. A U.S. Holder that makes a QEF Election will be subject to U.S. federal income tax on such amounts for each tax year in which we are a PFIC, regardless of whether such amounts are actually distributed to such U.S. Holder by us. However, for any tax year in which we are a PFIC and have no net income or gain, U.S. Holders that have made a QEF Election would not have any income inclusions as a result of the QEF Election. If a U.S. Holder that made a QEF Election has an income inclusion, such a U.S. Holder may, subject to certain limitations, elect to defer payment of current U.S. federal income tax on such amounts, subject to an interest charge. If such U.S. Holder is not a corporation, any such interest paid will be treated as “personal interest,” which is not deductible.
A U.S. Holder that makes a timely and effective QEF Election with respect to us generally (a) may receive a tax-free distribution from us to the extent that such distribution represents our “earnings and profits” that were previously included in income by the U.S. Holder because of such QEF Election and (b) will adjust such U.S. Holder’s tax basis in our offered shares to reflect the amount included in income or allowed as a tax-free distribution because of such QEF Election. In addition, a U.S. Holder that makes a QEF Election generally will recognize capital gain or loss on the sale or other taxable disposition of our offered shares.
The procedure for making a QEF Election, and the U.S. federal income tax consequences of making a QEF Election, will depend on whether such QEF Election is timely. A QEF Election will be treated as “timely” if such QEF Election is made for the first year in the U.S. Holder’s holding period for our offered shares in which we were a PFIC. A U.S. Holder may make a timely QEF Election by filing the appropriate QEF Election documents at the time such U.S. Holder files a U.S. federal income tax return for such year. If a U.S. Holder does not make a timely and effective QEF Election for the first year in the U.S. Holder’s holding period for our offered shares, the U.S. Holder may still be able to make a timely and effective QEF Election in a subsequent year if such U.S. Holder meets certain requirements and makes a “purging” election to recognize gain (which will be taxed under the rules of Section 1291 of the Code discussed above) as if such offered shares were sold for their fair market value on the day the QEF Election is effective. If a U.S. Holder makes a QEF Election but does not make a “purging” election to recognize gain, as discussed in the preceding sentence, then such U.S. Holder shall continue to be subject to tax under the rules of Section 1291 discussed above with respect to its offered shares. If a U.S. Holder owns PFIC stock indirectly through another PFIC, separate QEF Elections must be made for the PFIC in which the U.S. Holder is a direct shareholder and the Subsidiary PFIC for the QEF rules to apply to both PFICs.
107
Table of Contents
A QEF Election will apply to the tax year for which such QEF Election is timely made and to all subsequent tax years, unless such QEF Election is invalidated or terminated or the IRS consents to revocation of such QEF Election. If a U.S. Holder makes a QEF Election and, in a subsequent tax year, we cease to be a PFIC, the QEF Election will remain in effect (although it will not be applicable) during those tax years in which we are not a PFIC. Accordingly, if we become a PFIC in another subsequent tax year, the QEF Election will be effective and the U.S. Holder will be subject to the QEF rules described above during any subsequent tax year in which we qualify as a PFIC.
We will (a) use commercially reasonable efforts to make available to U.S. Holders, upon their written request after the end of a tax year, information as to our status as a PFIC, and (b) for each year in which we are a PFIC, provide to a U.S. Holder, upon written request, all information and documentation that a U.S. Holder making a QEF Election with respect to us is required to obtain for U.S. federal income tax purposes. We may elect to provide such information on our website. However, U.S. Holders should be aware that we can provide no assurances that we will provide any such information relating to any Subsidiary PFIC. Because we may own shares in one or more Subsidiary PFICs at any time, U.S. Holders will continue to be subject to the rules discussed above with respect to the taxation of gains and excess distributions with respect to any Subsidiary PFIC for which the U.S. Holders do not obtain the required information. Each U.S. Holder should consult its own tax advisors regarding the availability of, and procedure for making, a QEF Election with respect to us and any Subsidiary PFIC.
A U.S. Holder makes a QEF Election by attaching a completed IRS Form 8621, including a PFIC Annual Information Statement, to a timely filed United States federal income tax return. However, if we do not provide the required information with regard to us or any of our Subsidiary PFICs, U.S. Holders will not be able to make a QEF Election for such entity and will continue to be subject to the rules of Section 1291 of the Code, discussed above, that apply to Non-Electing U.S. Holders with respect to the taxation of gains and excess distributions.
Mark-to-Market Election
A U.S. Holder may make a Mark-to-Market Election only if our offered shares are marketable stock. Our offered shares generally will be “marketable stock” if our offered shares are regularly traded on (a) a national securities exchange that is registered with the Securities and Exchange Commission, (b) the national market system established pursuant to section 11A of the Securities and Exchange Act of 1934, or (c) a foreign securities exchange that is regulated or supervised by a governmental authority of the country in which the market is located, provided that (i) such foreign exchange has trading volume, listing, financial disclosure, and surveillance requirements, and meets other requirements and the laws of the country in which such foreign exchange is located, together with the rules of such foreign exchange, ensure that such requirements are actually enforced and (ii) the rules of such foreign exchange effectively promote active trading of listed stocks. If such stock is traded on such a qualified exchange or other market, such stock generally will be “regularly traded” for any calendar year during which such stock is traded, other than in de minimis quantities, on at least 15 days during each calendar quarter. We believe that our offered shares were “regularly traded” in the fourth calendar quarter of 2014 and we expect that our offered shares will be “regularly traded” in the first calendar quarter of 2015. However, there can be no assurance that our common shares will be “regularly traded” in subsequent calendar quarters. U.S. Holders should consult their own tax advisors regarding the marketable stock rules.
A U.S. Holder that makes a Mark-to-Market Election with respect to its offered shares generally will not be subject to the rules of Section 1291 of the Code discussed above with respect to such offered shares. However, if a U.S. Holder does not make a Mark-to-Market Election beginning in the first tax year of such U.S. Holder’s holding period for our offered shares for which we are a PFIC or such U.S. Holder has not made a timely QEF Election, the rules of Section 1291 of the Code discussed above will apply to certain dispositions of, and distributions on, our offered shares.
A U.S. Holder that makes a Mark-to-Market Election will include in ordinary income, for each tax year in which we are a PFIC, an amount equal to the excess, if any, of (a) the fair market value of our offered shares, as of the close of such tax year over (b) such U.S. Holder’s adjusted tax basis in such offered shares. A U.S. Holder that
108
Table of Contents
makes a Mark-to-Market Election will be allowed a deduction in an amount equal to the excess, if any, of (a) such U.S. Holder’s adjusted tax basis in our offered shares, over (b) the fair market value of such offered shares (but only to the extent of the net amount of previously included income as a result of the Mark-to-Market Election for prior tax years).
A U.S. Holder that makes a Mark-to-Market Election generally also will adjust such U.S. Holder’s tax basis in our offered shares to reflect the amount included in gross income or allowed as a deduction because of such Mark-to-Market Election. In addition, upon a sale or other taxable disposition of our offered shares, a U.S. Holder that makes a Mark-to-Market Election will recognize ordinary income or ordinary loss (not to exceed the excess, if any, of (a) the amount included in ordinary income because of such Mark-to-Market Election for prior tax years over (b) the amount allowed as a deduction because of such Mark-to-Market Election for prior tax years). Losses that exceed this limitation are subject to the rules generally applicable to losses provided in the Code and Treasury Regulations.
A U.S. Holder makes a Mark-to-Market Election by attaching a completed IRS Form 8621 to a timely filed United States federal income tax return. A Mark-to-Market Election applies to the tax year in which such Mark-to-Market Election is made and to each subsequent tax year, unless our offered shares cease to be “marketable stock” or the IRS consents to revocation of such election. Each U.S. Holder should consult its own tax advisors regarding the availability of, and procedure for making, a Mark-to-Market Election.
Although a U.S. Holder may be eligible to make a Mark-to-Market Election with respect to our offered shares, no such election may be made with respect to the stock of any Subsidiary PFIC that a U.S. Holder is treated as owning, because such stock is not marketable. Hence, the Mark-to-Market Election will not be effective to avoid the application of the default rules of Section 1291 of the Code described above with respect to deemed dispositions of Subsidiary PFIC stock or excess distributions from a Subsidiary PFIC.
Other PFIC Rules
Under Section 1291(f) of the Code, the IRS has issued proposed Treasury Regulations that, subject to certain exceptions, would cause a U.S. Holder that had not made a timely QEF Election to recognize gain (but not loss) upon certain transfers of our offered shares that would otherwise be tax-deferred (e.g., gifts and exchanges pursuant to corporate reorganizations). However, the specific U.S. federal income tax consequences to a U.S. Holder may vary based on the manner in which offered shares are transferred.
Certain additional adverse rules may apply with respect to a U.S. Holder if we are a PFIC, regardless of whether such U.S. Holder makes a QEF Election. For example, under Section 1298(b)(6) of the Code, a U.S. Holder that uses our common shares as security for a loan will, except as may be provided in Treasury Regulations, be treated as having made a taxable disposition of such offered shares.
Special rules also apply to the amount of foreign tax credit that a U.S. Holder may claim on a distribution from a PFIC. Subject to such special rules, foreign taxes paid with respect to any distribution in respect of stock in a PFIC are generally eligible for the foreign tax credit. The rules relating to distributions by a PFIC and their eligibility for the foreign tax credit are complicated, and a U.S. Holder should consult with its own tax advisors regarding the availability of the foreign tax credit with respect to distributions by a PFIC.
The PFIC rules are complex, and each U.S. Holder should consult its own tax advisors regarding the PFIC rules and how the PFIC rules may affect the U.S. federal income tax consequences of the acquisition, ownership, and disposition of our offered shares.
General Rules Applicable to the Ownership and Disposition of Offered Shares
The following discussion describes the general rules applicable to the ownership and disposition of our offered shares but is subject in its entirety to the special rules described above under the heading “Passive Foreign Investment Company Rules.”
109
Table of Contents
Distributions on Offered Shares
A U.S. Holder that receives a distribution, including a constructive distribution, with respect to an offered share will be required to include the amount of such distribution in gross income as a dividend (without reduction for any Canadian income tax withheld from such distribution) to the extent of our current and accumulated “earnings and profits,” as computed for U.S. federal income tax purposes. A dividend generally will be taxed to a U.S. Holder at ordinary income tax rates if we are a PFIC for the tax year of such distribution or the preceding tax year. To the extent that a distribution exceeds our current and accumulated “earnings and profits,” such distribution will be treated first as a tax-free return of capital to the extent of a U.S. Holder’s tax basis in our offered shares and thereafter as gain from the sale or exchange of such offered shares. (See “Sale or Other Taxable Disposition of Offered Shares,” below.) However, we may not maintain the calculations of its earnings and profits in accordance with U.S. federal income tax principles, and each U.S. Holder may have to assume that any distribution by us with respect to our offered shares will constitute ordinary dividend income. Dividends received on our offered shares by corporate U.S. Holders generally will not be eligible for the “dividends received deduction.” Subject to applicable limitations and provided we are eligible for the benefits of the Treaty, dividends paid by us to non-corporate U.S. Holders, including individuals, generally will be eligible for the preferential tax rates applicable to long-term capital gains for dividends, provided certain holding period and other conditions are satisfied, including that we not be classified as a PFIC in the tax year of distribution or in the preceding tax year. The dividend rules are complex, and each U.S. Holder should consult its own tax advisors regarding the application of such rules.
Sale or Other Taxable Disposition of Offered Shares
Subject to the discussion above under “Passive Foreign Investment Company Rules,” upon the sale or other taxable disposition of our offered shares, a U.S. Holder generally will recognize capital gain or loss in an amount equal to the difference between the U.S. dollar value of cash received plus the fair market value of any property received and such U.S. Holder’s tax basis in such offered shares sold or otherwise disposed of. A U.S. Holder’s tax basis in our offered shares generally will be such holder’s U.S. dollar cost for such offered shares. Gain or loss recognized on such sale or other disposition generally will be long-term capital gain or loss if, at the time of the sale or other disposition, our offered shares have been held for more than one year.
Preferential tax rates currently apply to long-term capital gain of a U.S. Holder that is an individual, estate, or trust. There are currently no preferential tax rates for long-term capital gain of a U.S. Holder that is a corporation. Deductions for capital losses are subject to significant limitations under the Code.
Certain Adjustments to the Series II First Preferred Shares
Under Section 305 of the Code, an adjustment to the number of common shares that will be issued on the conversion of Series II First Preferred Shares, or an adjustment to the conversion ratio of the Series II First Preferred Shares, may be treated as a constructive distribution to a U.S. Holder of Series II First Preferred Shares if, and to the extent that, such adjustment has the effect of increasing such U.S. Holder’s proportionate interest in our “earnings and profits” or our assets, depending on the circumstances of such adjustment (for example, if such adjustment is to compensate for a distribution of cash or other property to the shareholders). Adjustments to the conversion ratio of the Series II First Preferred Shares made pursuant to a bona fide reasonable adjustment formula that has the effect of preventing dilution of the interest of the holders of Series II First Preferred Shares should generally not be considered to result in a constructive distribution. Any such constructive distribution would be taxable whether or not there is an actual distribution of cash or other property. (See more detailed discussion of the rules applicable to distributions made by us at “Passive Foreign Investment Company Rules” and “Distributions on Offered Shares” above).
110
Table of Contents
Conversion of the Series II First Preferred Shares into Common Shares
Subject to the PFIC rules and Section 305 rules discussed below, the conversion of Series II First Preferred Shares into our common shares pursuant to the right of conversion should qualify as a tax-deferred “recapitalization” within the meaning of Section 368(a)(1)(E) of the Code, or “Recapitalization”. If there are dividend arrearages (i.e., accumulated, undeclared and unpaid dividends) on the Series II First Preferred Shares, then the common shares received as consideration for such dividend arrearages will be treated as a distribution on the Series II First Preferred Shares (see “Passive Foreign Investment Company Rules” and “Distributions on Offered Shares” above). Subject to the PFIC rules and Section 305 rules discussed below, assuming that such conversion qualifies as a recapitalization under Section 368(a)(1)(E) of the Code and that there are no dividend arrearages, upon the conversion of the Series II First Preferred Shares into our common shares pursuant to the right of conversion, a U.S. Holder: (a) should not recognize any gain or loss; (b) the aggregate tax basis of our common shares received in the conversion will equal the aggregate tax basis in the Series II First Preferred Shares surrendered in the conversion; and (c) the holding period in our common shares received in the conversion will include the holding period of the Series II First Preferred Shares surrendered in the conversion.
Section 1291(f) of the Code provides that, to the extent provided in Treasury Regulations, any normally available non-recognition provision will not apply to a U.S. Holder’s disposition (including, in particular, any disposition of shares that occurs pursuant to a reorganization under Section 368(a) of the Code) of shares of a foreign corporation if the corporation was a PFIC for any taxable year that is included in whole or in part during the U.S. Holder’s holding period for the Series II First Preferred Shares. The U.S. Treasury Department has issued proposed Treasury Regulations (which are not yet effective), but no final or temporary Treasury Regulations, under Section 1291(f). However, it is impossible to predict at this time whether, in what form, and with what effective date, final Treasury Regulations will be adopted. There is uncertainty about whether Section 1291(f) is self-executing. However, the IRS’s position appears to be that Section 1291(f) of the Code is self-executing notwithstanding the absence of final or temporary Treasury Regulations. The proposed Treasury Regulations, however, provide that non-recognition treatment is not precluded if the U.S. Holder’s conversion of Series II First Preferred Shares are converted into our common shares pursuant to a “recapitalization” under Section 368(a)(1)(E) of the Code. U.S. Holders of Series II First Preferred Shares should consult its own tax advisors regarding Section 1291(f) of the Code and the proposed Treasury Regulations issued thereunder.
Under Section 305(b)(4) of the Code, the conversion of the Series II First Preferred Shares into our common shares pursuant to the right of conversion may be treated as a constructive distribution to a U.S. Holder of Series II First Preferred Shares if, and to the extent that, such conversion has the effect of increasing such U.S. Holder’s proportionate interest in our “earnings and profits” or our assets. It is unclear whether any such conversion will result in a constructive distribution under Section 305(b)(4). Any such constructive distribution would be taxable whether or not there is an actual distribution of cash or other property. (See more detailed discussion of the rules applicable to distributions made by us at “Passive Foreign Investment Company Rules” and “Distributions on Offered Shares” above).
Each U.S. Holder should consult its own tax advisors regarding the tax consequences of the conversion of the Series II First Preferred Shares into our common shares pursuant to the right of conversion.
Additional Considerations
Additional Tax on Passive Income
Certain U.S. Holders that are individuals, estates or trusts (other than trusts that are exempt from tax) will be subject to a 3.8% tax on all or a portion of their “net investment income,” which includes dividends on our offered shares, and net gains from the disposition of our offered shares. Further, excess distributions treated as dividends, gains treated as excess distributions, and mark-to-market inclusions and deductions are all included in the calculation of net investment income.
111
Table of Contents
Treasury Regulations provide, subject to the election described in the following paragraph, that solely for purposes of this additional tax, distributions of previously taxed income will be treated as dividends and included in net investment income subject to the additional 3.8% tax. Additionally, to determine the amount of any capital gain from the sale or other taxable disposition of our common shares that will be subject to the additional tax on net investment income, a U.S. Holder who has made a QEF Election will be required to recalculate its basis in our common shares excluding QEF basis adjustments.
Alternatively, a U.S. Holder may make an election which will be effective with respect to all interests in a PFIC for which a QEF Election has been made and which is held in that year or acquired in future years. Under this election, a U.S. Holder pays the additional 3.8% tax on QEF income inclusions and on gains calculated after giving effect to related tax basis adjustments. U.S. Holders that are individuals, estates or trusts should consult their own tax advisors regarding the applicability of this tax to any of their income or gains in respect of our offered shares.
Receipt of Foreign Currency
The amount of any distribution paid to a U.S. Holder in foreign currency, or on the sale, exchange or other taxable disposition of our offered shares, generally will be equal to the U.S. dollar value of such foreign currency based on the exchange rate applicable on the date of receipt (regardless of whether such foreign currency is converted into U.S. dollars at that time). A U.S. Holder will have a basis in the foreign currency equal to its U.S. dollar value on the date of receipt. Any U.S. Holder who converts or otherwise disposes of the foreign currency after the date of receipt may have a foreign currency exchange gain or loss that would be treated as ordinary income or loss, and generally will be U.S. source income or loss for foreign tax credit purposes. Different rules apply to U.S. Holders who use the accrual method with respect to foreign currency received upon the sale, exchange or other taxable disposition of our offered shares. Each U.S. Holder should consult its own U.S. tax advisors regarding the U.S. federal income tax consequences of receiving, owning, and disposing of foreign currency.
Foreign Tax Credit
Subject to the PFIC rules discussed above, a U.S. Holder that pays (whether directly or through withholding) Canadian income tax with respect to dividends paid on our offered shares generally will be entitled, at the election of such U.S. Holder, to receive either a deduction or a credit for such Canadian income tax. Generally, a credit will reduce a U.S. Holder’s U.S. federal income tax liability on a dollar-for-dollar basis, whereas a deduction will reduce a U.S. Holder’s income that is subject to U.S. federal income tax. This election is made on a year-by-year basis and applies to all foreign taxes paid (whether directly or through withholding) by a U.S. Holder during a year.
Complex limitations apply to the foreign tax credit, including the general limitation that the credit cannot exceed the proportionate share of a U.S. Holder’s U.S. federal income tax liability that such U.S. Holder’s “foreign source” taxable income bears to such U.S. Holder’s worldwide taxable income. In applying this limitation, a U.S. Holder’s various items of income and deduction must be classified, under complex rules, as either “foreign source” or “U.S. source.” Generally, dividends paid by a foreign corporation should be treated as foreign source for this purpose, and gains recognized on the sale of stock of a foreign corporation by a U.S. Holder should be treated as U.S. source for this purpose, except as otherwise provided in an applicable income tax treaty, and if an election is properly made under the Code. However, the amount of a distribution with respect to our offered shares that is treated as a “dividend” may be lower for U.S. federal income tax purposes than it is for Canadian federal income tax purposes, resulting in a reduced foreign tax credit allowance to a U.S. Holder. In addition, this limitation is calculated separately with respect to specific categories of income. The foreign tax credit rules are complex, and each U.S. Holder should consult its own U.S. tax advisors regarding the foreign tax credit rules.
112
Table of Contents
Backup Withholding and Information Reporting
Under U.S. federal income tax law, certain categories of U.S. Holders must file information returns with respect to their investment in, or involvement in, a foreign corporation. For example, U.S. return disclosure obligations (and related penalties) are imposed on individuals who are U.S. Holders that hold certain specified foreign financial assets in excess of certain thresholds. The definition of specified foreign financial assets includes not only financial accounts maintained in foreign financial institutions, but also, unless held in accounts maintained by a financial institution, any stock or security issued by a non-U.S. person, any financial instrument or contract held for investment that has an issuer or counterparty other than a U.S. person and any interest in a foreign entity. U.S. Holders may be subject to these reporting requirements unless their offered shares are held in an account at certain financial institutions. Penalties for failure to file certain of these information returns are substantial. U.S. Holders should consult with their own tax advisors regarding the requirements of filing information returns, including the requirement to file an IRS Form 8938.
Payments made within the U.S., or by a U.S. payor or U.S. middleman, of dividends on, and proceeds arising from the sale or other taxable disposition of, our offered shares will generally be subject to information reporting and backup withholding tax, at the rate of 28%, if a U.S. Holder (a) fails to furnish such U.S. Holder’s correct U.S. taxpayer identification number (generally on Form W-9), (b) furnishes an incorrect U.S. taxpayer identification number, (c) is notified by the IRS that such U.S. Holder has previously failed to properly report items subject to backup withholding tax, or (d) fails to certify, under penalty of perjury, that such U.S. Holder has furnished its correct U.S. taxpayer identification number and that the IRS has not notified such U.S. Holder that it is subject to backup withholding tax. However, certain exempt persons generally are excluded from these information reporting and backup withholding rules. Backup withholding is not an additional tax. Any amounts withheld under the U.S. backup withholding tax rules will be allowed as a credit against a U.S. Holder’s U.S. federal income tax liability, if any, or will be refunded, if such U.S. Holder furnishes required information to the IRS in a timely manner.
The discussion of reporting requirements set forth above is not intended to constitute a complete description of all reporting requirements that may apply to a U.S. Holder. A failure to satisfy certain reporting requirements may result in an extension of the time period during which the IRS can assess a tax, and, under certain circumstances, such an extension may apply to assessments of amounts unrelated to any unsatisfied reporting requirement. Each U.S. Holder should consult its own tax advisors regarding the information reporting and backup withholding rules.
113
Table of Contents
CERTAIN CANADIAN FEDERAL INCOME TAX CONSIDERATIONS
The following is, as of the date hereof, a summary of the principal Canadian federal income tax considerations generally applicable under theIncome Tax Act(Canada) and the regulations promulgated thereunder, or the Tax Act, to a Non-Resident Holder (as hereinafter defined), who acquires, as beneficial owner, the offered shares under this offering, and who, for purposes of the Tax Act and at all relevant times, holds the offered shares as capital property and deals at arm’s length with, and is not affiliated with, Trillium or the Underwriters. Generally, the offered shares will be considered to be capital property to a Non-Resident Holder provided the Non-Resident Holder does not hold the offered shares in the course of carrying on a business of trading or dealing in securities and has not acquired them in one or more transactions considered to be an adventure or concern in the nature of trade.
This summary is based upon the provisions of the Tax Act in force as of the date hereof, all specific proposals to amend the Tax Act that have been publicly and officially announced by or on behalf of the Minister of Finance (Canada) prior to the date hereof, or the Proposed Amendments, and Counsels’ understanding of the current administrative policies and assessing practices of the Canada Revenue Agency (the “CRA”), published in writing by it prior to the date hereof. This summary assumes the Proposed Amendments will be enacted in the form proposed. However, no assurance can be given that the Proposed Amendments will be enacted in their current form, or at all. This summary is not exhaustive of all possible Canadian federal income tax considerations and, except for the Proposed Amendments, does not take into account or anticipate any changes in the law or any changes in the CRA’s administrative policies or assessing practices, whether by legislative, governmental or judicial action or decision, nor does it take into account or anticipate any other federal or any provincial, territorial or foreign tax considerations, which may differ significantly from those discussed herein.
This summary is not applicable to a Non-Resident Holder: (i) that is a “financial institution”, as defined in the Tax Act for purposes of the mark-to-market rules; (ii) an interest in which would be a “tax shelter investment”; (iii) that is a “specified financial institution”; (iv) who makes or has made a “functional currency” reporting election; or (v) that has entered or enters into a “derivative forward agreement” with respect to the offered shares (each as defined in the Tax Act).Any such Non-Resident Holder should consult its own tax advisor with respect to an investment in the offered shares. This summary is of a general nature only and is not intended to be, nor should it be construed to be, legal or tax advice to any prospective purchaser or holder of the offered shares, and no representations with respect to the income tax consequences to any prospective purchaser or holder are made. Consequently, prospective purchasers or holders of the offered shares should consult their own tax advisors with respect to their particular circumstances.
A “Non-Resident Holder” at all relevant times for purposes of the Tax Act and any relevant income tax treaty or convention: (i) is not, and is not deemed to be resident in Canada; and (ii) does not use or hold and will not be deemed to use or hold, the offered shares in a business carried on in Canada. Special rules, which are not discussed in this summary, may apply to an insurer that carries on an insurance business in Canada.Non-Resident Holders of the offered shares should seek advice from their own tax advisors.
Currency Conversion
Generally, for purposes of the Tax Act, all amounts relating to the acquisition, holding or disposition of the offered shares must be converted into Canadian dollars based on the exchange rates as determined in accordance with the Tax Act. The amounts subject to withholding tax and any capital gains or capital losses realized by a Non-Resident Holder may be affected by fluctuations in the Canadian-U.S. dollar exchange rate.
Disposition of Common Shares
A Non-Resident Holder will not generally be subject to tax under the Tax Act on a disposition of a common share, unless the common share constitutes “taxable Canadian property” (as defined in the Tax Act) of the Non-Resident Holder at the time of disposition and the Non-Resident Holder is not entitled to relief under an applicable income tax treaty or convention.
114
Table of Contents
Provided the common shares are listed on a “designated stock exchange”, as defined in the Tax Act (which currently includes the TSX and NASDAQ) at the time of disposition, the common shares will generally not constitute taxable Canadian property of a Non-Resident Holder at that time, unless at any time during the60-month period immediately preceding the disposition the following two conditions are satisfied concurrently: (i) (a) the Non-Resident Holder; (b) persons with whom the Non-Resident Holder did not deal at arm’s length; (c) partnerships in which the Non-Resident Holder or a person described in (b) holds a membership interest directly or indirectly through one or more partnerships; or (d) any combination of the persons and partnerships described in (a) through (c), owned 25% or more of the issued shares of any class or series of the shares of Trillium; and (ii) more than 50% of the fair market value of the shares of Trillium was derived directly or indirectly from one or any combination of: real or immovable property situated in Canada, “Canadian resource properties”, “timber resource properties” (each as defined in the Tax Act), and options in respect of, or interests in or for civil law rights in, such properties. Notwithstanding the foregoing, in certain circumstances set out in the Tax Act, the common shares could be deemed to be taxable Canadian property. Even if the common shares are taxable Canadian property to a Non-Resident Holder, such Non-Resident Holder may be exempt from tax under the Tax Act on the disposition of such common shares by virtue of an applicable income tax treaty or convention.A Non Resident Holder contemplating a disposition of common shares that may constitute taxable Canadian property should consult a tax advisor prior to such disposition.
Disposition and Conversion of Preferred Shares
The conversion of Series II First Preferred Shares into common shares of Trillium pursuant to the exercise of the conversion privilege will be deemed not to constitute a disposition of property for purposes of the Tax Act and, accordingly, will not give rise to a capital gain or capital loss.
A Non-Resident Holder will not generally be subject to tax under the Tax Act on a disposition of a Series II First Preferred Shares, unless the Series II First Preferred Shares constitutes “taxable Canadian property” (as defined in the Tax Act) of the Non-Resident Holder at the time of disposition and the Non-Resident Holder is not entitled to relief under an applicable income tax treaty or convention.
The Series II First Preferred shares will generally not constitute taxable Canadian property to a Non-Resident at that time, unless at any time during the 60-month period immediately preceding the disposition more than 50% of the fair market value of Trillium was derived directly or indirectly (otherwise than through a corporation, partnership or trust the shares or interests in which were not themselves taxable Canadian property at the particular time) from one or any combination of: real or immovable property situated in Canada, “Canadian resource properties”, “timber resource properties” (each as defined in the Tax Act), and options in respect of, or interests in or for civil law rights in, such properties. Notwithstanding the foregoing, in certain circumstances set out in the Tax Act, the Series II First Preferred Shares could be deemed to be taxable Canadian property. Even if the Series II First Preferred Shares are taxable Canadian property to a Non-Resident Holder, such Non-Resident Holder may be exempt from tax under the Tax Act on the disposition of such Series II First Preferred Shares by virtue of an applicable income tax treaty or convention.A Non Resident Holder contemplating a disposition of common shares that may constitute taxable Canadian property should consult a tax advisor prior to such disposition.
Receipt of Dividends on Common Shares or the Series II First Preferred Shares
Dividends received or deemed to be received by a Non-Resident Holder on the common shares or the Series II First Preferred Shares will be subject to Canadian withholding tax under the Tax Act. The general rate of withholding tax is 25%, although such rate may be reduced under the provisions of an applicable income tax convention between Canada and the Non-Resident Holder’s country of residence, for example, under theCanada-United States Income Tax Convention (1980) as amended, the rate is generally reduced to 15%.
115
Table of Contents
There are no other contracts, other than those disclosed in this prospectus and those entered into in the ordinary course of our business, that are material to us and which were entered into in the last two completed fiscal years or which were entered into before the two most recently completed fiscal years but are still in effect as of the date of this prospectus:
| 1. | License Agreement between Trillium Privateco, UHN and HSC dated February 1, 2010 pursuant to which we licensed intellectual property relating to methods and compounds for the modulation of the SIRPa-CD47 interaction for therapeutic cancer applications. The license agreement requires us to use commercially reasonable efforts to commercialize the licensed technology. The license agreement will terminate on a country-by-country basis, in countries where a valid claim exists, when the last valid claim expires in such country, or if no valid claim exists, when the last valid claim expires in the U.S. We paid an up-front license fee of $150,000 and committed to pay an annual maintenance fee of $25,000, as well as payments on patent issuances, development milestone payments ranging from $100,000 to $300,000 on the initiation of phase I, II and III clinical trials respectively, and payments upon the achievement of certain regulatory milestones as well as royalties of either 3% or 1% of net revenues on commercial sales. The regulatory milestone payments amount to $1 million on each of the submission of a first BLA in the U.S. and receipt of first regulatory approval in the U.S. and proportionate payments in other territories worldwide. The aggregate milestones payable on their first achievement under the agreement in the major markets of the U.S., Europe and Asia combined are $5,660,000. Under the license agreement, Trillium is required to pay 20% of any sublicensing revenues to the licensors on the first $50 million of sublicensing revenues, and pay 15% of any sublicensing revenues to the licensors after the first $50 million of sublicensing revenue received. |
| 2. | Debenture Purchase Agreement and Merger Agreement between the Company, Trillium Privateco, 2364556 Ontario Limited, and the holders of debentures issued by Trillium Privateco dated March 25, 2013 pursuant to which we acquired Trillium Privateco. See the disclosure of the terms of the merger with Trillium Privateco in the section of this prospectus entitled “Business – General Development of the Business – Acquisition of Trillium Privateco”. |
| 3. | GPEx®-Derived Cell Line Sale Agreement between Trillium Therapeutics Inc. and Catalent Pharma Solutions, LLC dated August 12, 2014 pursuant to which we acquired the right to use the GPEx® expression system for the manufacture of TTI-621 (SIRPaFc). Consideration for the license includes potential pre-marketing approval milestones of up to US$875,000 and aggregate sales milestone payments of up to US$28.8 million. |
| 4. | GPEx®-Derived Cell Line Sale Agreement between Trillium Therapeutics Inc. and Catalent Pharma Solutions, LLC dated August 12, 2014 pursuant to which we acquired the right to use the GPEx® expression system for the manufacture of TTI-622 (SIRPaFc). Consideration for the license includes potential pre-marketing approval milestones of up to US$875,000 and aggregate sales milestone payments of up to US $28.8 million. |
| 5. | Shareholder rights plan between Trillium and Computershare Investor Services Inc. dated September 16, 2013 and amended June 3, 2014. This plan was approved by our shareholders on May 27, 2014. See the discussion in the section of this prospectus entitled “Description of Share Capital – Shareholder Rights Plan”. |
| 6. | 2014 Stock Option Plan that was approved by our shareholders on May 27, 2014. See the discussion in the section of this prospectus entitled “Board of Directors and Management – Stock Option Plan”. |
| 7. | 2014 DSU Plan that was approved by our shareholders on May 27, 2014. See the discussion in the section of this prospectus entitled “Board of Directors and Management – Deferred Share Unit Plan”. |
116
Table of Contents
| 8. | Warrant Indenture between the Company and Computershare Trust Company of Canada, or Computershare, dated March 15, 2013. This indenture provides that Computershare will act as the trust agent for the administration of the issued warrants. |
| 9. | Warrant Indenture between the Company and Computershare dated April 8, 2013. This indenture provides that Computershare will act as the trust agent for the administration of the issued warrants. |
| 10. | Warrant Indenture between the Company and Computershare dated December 13, 2013. This indenture provides that Computershare will act as the trust agent for the administration of the issued warrants. |
| 11. | Agency Agreement between the Company, Bloom Burton & Co. Inc. and Roth Capital Partners, LLC dated December 13, 2013. Bloom Burton & Co. Inc. and Roth Capital Partners acted as the sole and exclusive agents in our December 2013 private placement to solicit, on a commercially reasonable best efforts basis, orders for the Offered Units from Purchasers resident in the Offering Jurisdictions (capitalized terms having their meaning as set out in the agreement which is filed as an exhibit to the registration statement of which this prospectus forms a part). For their services, the agents received a commission equal to 6% of the total proceeds of the offering sold by the agents, and compensation warrants equal to 6% of the number of units sold by the agents, with an exercise price of $0.21 and a term of two years. |
| 12. | Executive Employment Agreement between the Company and Dr. Niclas Stiernholm effective June 3, 2014 for the position of Chief Executive Officer. |
| 13. | Executive Employment Agreement between the Company and Dr. Robert Uger effective June 3, 2014 for the position of Chief Scientific Officer. |
| 14. | Executive Employment Agreement between the Company and James Parsons effective June 3, 2014 for the position of Chief Financial Officer. |
| 15. | Executive Employment Agreement between the Company and Dr. Eric Sievers effective April 1, 2015 for the position of Chief Medical Officer. |
| 16. | Executive Employment Agreement between the Company and Dr. Penka Petrova effective June 3, 2014 for the position of Vice President, Drug Development. |
117
Table of Contents
There is no dividend restriction; however, we have not declared any dividends since our inception and do not anticipate that we will do so in the foreseeable future. We currently intend to retain future earnings, if any, to finance the development of our business. Any future payment of dividends or distributions will be determined by our board of directors on the basis of our earnings, financial requirements and other relevant factors. There is no special procedure for Non-Resident Holders to claim dividends. Any remittances of dividends to United States residents and to other non-residents are, however, subject to withholding tax. See the sections of this prospectus entitled “Certain U.S. Federal Income Tax Considerations” and “Certain Canadian Federal Income Tax Considerations” for additional information.
ENFORCEABILITY OF CIVIL LIABILITIES
We are incorporated under the laws of the Province of Ontario. Substantially all of our assets are located outside the United States. In addition, several of our directors and officers are nationals and/or residents of countries other than the United States, and all or a substantial portion of such persons’ assets may be located outside the United States. As a result, it may be difficult for investors to effect service of process within the United States upon us or such persons or to enforce against them or against us, judgments obtained in United States courts, including judgments predicated upon the civil liability provisions of the securities laws of the United States or any state thereof. In addition, investors should not assume that the courts of Canada (i) would enforce judgments of U.S. courts obtained in actions against us, our officers or directors, or other said persons, predicated upon the civil liability provisions of the U.S. federal securities laws or other laws of the United States or (ii) would enforce, in original actions, liabilities against us or such directors, officers or experts predicated upon the United States federal securities laws or any securities or other laws of any state or jurisdiction of the United States.
Notwithstanding this, we have also been advised by Borden Ladner Gervais LLP, that there is doubt whether an action could be brought in Canada in the first instance on the basis of liability predicated solely upon United States federal securities laws.
We have appointed Puglisi & Associates as our agent to receive service of process with respect to any action brought against us in the United States.
Our consolidated financial statements as at and for the years ended December 31, 2014 and 2013, and as at and for the years ended December 31, 2013 and 2012 included in this prospectus have been audited by Ernst & Young LLP, Independent Registered Public Accounting Firm, as stated in their report appearing in this prospectus, and have been so included in reliance upon the report of such firm given their authority as experts in accounting and auditing.
The financial statements of Trillium Privateco as at and for the years ended June 30, 2012 and 2011 included in this prospectus have been audited by Ernst & Young LLP, Independent Auditors, as stated in their report appearing in this prospectus, and have been so included in reliance upon the report of such firm given their authority as experts in accounting and auditing.
The validity of the common shares being offered by this prospectus and other legal matters concerning this offering relating to Canadian law will be passed upon for us by Borden Ladner Gervais LLP, Toronto, Ontario, Canada. Certain legal matters in connection with this offering relating to U.S. law will be passed upon for us by Dorsey & Whitney LLP, Vancouver, British Columbia, Canada and Seattle, Washington. Certain legal matters in connection with this offering will be passed upon for the underwriters by Blake, Cassels & Graydon LLP, Vancouver, British Columbia, Canada, with respect to Canadian law, and by Goodwin Procter LLP, New York, New York, with respect to U.S. law.
118
Table of Contents
There are no government laws, decrees or regulations in Canada that restrict the export or import of capital or that affect the remittance of dividends, interest or other payments to Non-Resident Holders of our common shares. Any remittances of dividends to United States residents and to other non-residents are, however, subject to withholding tax. See the sections of this prospectus entitled “Certain U.S. Federal Income Tax Considerations” and “Certain Canadian Federal Income Tax Considerations”.
We are subject to the informational requirements of the Exchange Act, and we file reports and other information with the SEC. You may read and copy any of our reports and other information at, and obtain copies upon payment of prescribed fees from, the Public Reference Room maintained by the SEC at 100 F Street, N.E., Washington, DC 20549. In addition, the SEC maintains a web site that contains reports and other information regarding registrants that file electronically with the SEC at http://www.sec.gov. The public may obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330.
As a foreign private issuer, we are exempt under the Exchange Act from, among other things, certain rules prescribing the furnishing and content of proxy statements, and our executive officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. In addition, we are not required under the Exchange Act to file periodic reports and financial statements with the SEC as frequently or as promptly as U.S. companies whose securities are registered under the Exchange Act.
We are also subject to the full informational requirements of the securities commissions in all provinces of Canada, and you are also invited to read and copy any reports, statements or other information, other than confidential filings, that we file with the Canadian provincial securities commissions. These filings are also electronically available from the Canadian System for Electronic Document Analysis and Retrieval (SEDAR) at www.sedar.com, the Canadian equivalent of the SEC’s electronic document gathering and retrieval system.
We maintain a corporate website at http://trilliumtherapeutics.com. Information contained on, or that can be accessed through, our website does not constitute a part of this prospectus.
119
Table of Contents
INDEX TO THE FINANCIAL STATEMENTS
Trillium Therapeutics Inc. (formerly Stem Cell Therapeutics Corp.)
For the years ended December 31, 2014 and 2013
| F-2 | ||||
| F-3 | ||||
| F-4 | ||||
| F-5 | ||||
| F-6 | ||||
| F-7 |
Trillium Therapeutics Inc. (formerly Stem Cell Therapeutics Corp.)
For the years ended December 31, 2013 and 2012
| F-34 | ||||
| F-35 | ||||
| F-36 | ||||
| F-37 | ||||
| F-39 | ||||
| F-40 |
Trillium Therapeutics Inc. (Trillium Privateco)
For the nine months ended March 31, 2013 and 2012
| F-68 | ||||
| F-69 | ||||
| F-70 | ||||
| F-71 |
Trillium Therapeutics Inc. (Trillium Privateco)
For the years ended June 30, 2012 and 2011
| F-82 | ||||
| F-83 | ||||
| F-84 | ||||
| F-85 | ||||
| F-86 |
Trillium Therapeutics Inc. (formerly Stem Cell Therapeutics Corp.)
For the year ended December 31, 2013
Pro forma Consolidated Statement of Loss and Comprehensive Loss | F-103 |
F-i
Table of Contents

(formerly Stem Cell Therapeutics Corp.)
CONSOLIDATED FINANCIAL STATEMENTS
FOR THE YEARS ENDED
DECEMBER 31, 2014 AND 2013
96 Skyway Avenue
Toronto, Ontario M9W 4Y9
www.trilliumtherapeutics.com
F-1
Table of Contents
INDEPENDENT AUDITORS’ REPORT OF REGISTERED PUBLIC ACCOUNTING FIRM
To the Directors of
Trillium Therapeutics Inc.
We have audited the accompanying consolidated financial statements ofTrillium Therapeutics Inc.(formerly Stem Cell Therapeutics Corp.), which comprise the consolidated statements of financial position as at December 31, 2014 and 2013, and the consolidated statements of loss and comprehensive loss, changes in equity and cash flows for the years then ended, and a summary of significant accounting policies and other explanatory information.
Management’s responsibility for the consolidated financial statements
Management is responsible for the preparation and fair presentation of these consolidated financial statements in accordance with International Financial Reporting Standards, as issued by the International Accounting Standards Board, and for such internal control as management determines is necessary to enable the preparation of consolidated financial statements that are free from material misstatement, whether due to fraud or error.
Auditors’ responsibility
Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We conducted our audits in accordance with Canadian generally accepted auditing standards and the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we comply with ethical requirements and plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free from material misstatement. We were not engaged to perform an audit of the Company’s internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
An audit involves performing procedures to obtain audit evidence about the amounts and disclosures in the consolidated financial statements. The procedures selected depend on the auditors’ judgment, including the assessment of the risks of material misstatement of the consolidated financial statements, whether due to fraud or error. An audit also includes, on a test basis, evidence supporting the amounts and disclosures in the consolidated financial statements, evaluating the appropriateness of accounting policies used and the reasonableness of accounting estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements.
We believe that the audit evidence we have obtained in our audits is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position ofTrillium Therapeutics Inc. as at December 31, 2014 and 2013 and its financial performance and its cash flows for the years then ended in accordance with International Financial Reporting Standards, as issued by the International Accounting Standards Board.
/s/ Ernst & Young LLP | ||
Toronto, Canada | Chartered Professional Accountants | |
March 23, 2015 | Licensed Public Accountants |
F-2
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Consolidated Statements of Financial Position
Amounts in Canadian Dollars
| Note | As at December 31, 2014 $ | As at December 31, 2013 $ | ||||||||
ASSETS | ||||||||||
Current | ||||||||||
Cash | 26,165,056 | 32,456,506 | ||||||||
Marketable securities | - | 526,598 | ||||||||
Amounts receivable | 5 | 344,416 | 427,234 | |||||||
Prepaid expenses and other | 1,008,225 | 94,569 | ||||||||
Total current assets | 27,517,697 | 33,504,907 | ||||||||
Property and equipment | 6 | 235,402 | 109,007 | |||||||
Intangible assets | 7 | 432,933 | 1,473,472 | |||||||
Total non-current assets | 668,335 | 1,582,479 | ||||||||
Total assets | 28,186,032 | 35,087,386 | ||||||||
LIABILITIES | ||||||||||
Current | ||||||||||
Accounts payable and accrued liabilities | 8 | 3,248,984 | 669,860 | |||||||
Other current liabilities | 9 | 279,461 | 62,766 | |||||||
Total current liabilities | 3,528,445 | 732,626 | ||||||||
Loan payable | 9 | 283,352 | 341,884 | |||||||
Long-term liability | 9 | 69,941 | 104,429 | |||||||
Total non-current liabilities | 353,293 | 446,313 | ||||||||
Total liabilities | 3,881,738 | 1,178,939 | ||||||||
EQUITY | ||||||||||
Common shares | 10 | 49,505,792 | 47,191,303 | |||||||
Preferred shares | 10 | 10,076,151 | 11,292,525 | |||||||
Warrants | 10 | 9,283,332 | 9,818,179 | |||||||
Contributed surplus | 10 | 5,995,055 | 3,280,656 | |||||||
Deficit | (50,556,036 | ) | (37,674,216 | ) | ||||||
Total equity | 24,304,294 | 33,908,447 | ||||||||
Total liabilities and equity | 28,186,032 | 35,087,386 | ||||||||
Commitments and contingencies[note 15]
Approved by the Board and authorized for issue on March 23, 2015:
| (signed) Luke Beshar, Director | (signed) Henry Friesen, Director |
See accompanying notes to the consolidated financial statements
F-3
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Consolidated Statements of Loss and Comprehensive Loss
Amounts in Canadian Dollars
Note
| Year ended $ | Year ended $ | ||||||||
EXPENSES | ||||||||||
Research and development | 12 | 10,595,808 | 3,336,706 | |||||||
General and administrative | 13 | 2,577,460 | 962,200 | |||||||
Operating expenses | 13,173,268 | 4,298,906 | ||||||||
Finance income | 14 | (378,692 | ) | (54,028 | ) | |||||
Finance costs | 14 | 87,244 | 44,430 | |||||||
Net finance income | (291,448 | ) | (9,598 | ) | ||||||
Net loss and comprehensive loss for the year | 12,881,820 | 4,289,308 | ||||||||
Basic and diluted loss per share | 10(c) | (3.06 | ) | (3.16 | ) | |||||
See accompanying notes to the consolidated financial statements
F-4
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Consolidated Statements of Changes in Equity
Amounts in Canadian Dollars
| Common shares | Preferred shares | Warrants | Contributed | |||||||||||||||||||||||||||||||||
Number # | Amount $ | Number # | Amount $ | Number # | Amount $ | surplus $ | Deficit $ | Total $ | ||||||||||||||||||||||||||||
| (note 10 | ) | (note 10 | ) | (note 10 | ) | (note 10 | ) | |||||||||||||||||||||||||||||
Balance, December 31, 2013 | 4,058,408 | 47,191,303 | 77,895,165 | 11,292,525 | 142,230,123 | 9,818,179 | 3,280,656 | (37,674,216 | ) | 33,908,447 | ||||||||||||||||||||||||||
Net loss and comprehensive loss for the year | - | - | - | - | - | - | - | (12,881,820 | ) | (12,881,820 | ) | |||||||||||||||||||||||||
Transactions with owners of the Company, recognized directly in equity | ||||||||||||||||||||||||||||||||||||
Exercise of warrants | 86,540 | 1,065,015 | - | - | (2,596,251 | ) | (118,202 | ) | - | - | 946,813 | |||||||||||||||||||||||||
Exercise of stock options | 2,614 | 33,100 | - | - | - | - | (13,500 | ) | - | 19,600 | ||||||||||||||||||||||||||
Conversion of preferred shares | 279,682 | 1,216,374 | (8,390,476 | ) | (1,216,374 | ) | - | - | - | - | - | |||||||||||||||||||||||||
Expiry of warrants | - | - | - | - | (909,091 | ) | (416,645 | ) | 416,645 | - | - | |||||||||||||||||||||||||
Share-based compensation | - | - | - | - | - | - | 2,311,254 | - | 2,311,254 | |||||||||||||||||||||||||||
Total transactions with owners of the Company | 368,836 | 2,314,489 | (8,390,476 | ) | (1,216,374 | ) | (3,505,342 | ) | (534,847 | ) | 2,714,399 | - | 3,277,667 | |||||||||||||||||||||||
Balance, December 31, 2014 | 4,427,244 | 49,505,792 | 69,504,689 | 10,076,151 | 138,724,781 | 9,283,332 | 5,995,055 | (50,556,036 | ) | 24,304,294 | ||||||||||||||||||||||||||
| Common shares | Preferred shares | Warrants | Contributed surplus $ | |||||||||||||||||||||||||||||||||
Number # | Amount $ | Number # | Amount $ | Number # | Amount $ | Deficit $ | Total $ | |||||||||||||||||||||||||||||
| (note 10 | ) | (note 10 | ) | (note 10 | ) | (note 10 | ) | |||||||||||||||||||||||||||||
Balance, December 31, 2012 | 622,060 | 31,388,959 | - | - | 1,036,364 | 508,281 | 2,870,138 | (33,384,908 | ) | 1,382,470 | ||||||||||||||||||||||||||
Net loss and comprehensive loss for the year | - | - | - | - | - | - | - | (4,289,308 | ) | (4,289,308 | ) | |||||||||||||||||||||||||
Transactions with owners of the Company, recognized directly in equity | ||||||||||||||||||||||||||||||||||||
Share units issued, net of issue costs | 3,066,090 | 13,580,794 | 77,895,165 | 11,292,525 | 130,592,142 | 8,455,762 | - | - | 33,329,081 | |||||||||||||||||||||||||||
Compensation warrants | - | - | - | - | 5,828,890 | 700,772 | - | - | 700,772 | |||||||||||||||||||||||||||
Issued to acquire Trillium Privateco (notes 4 and 10) | 202,639 | 1,215,836 | - | - | 3,300,000 | 165,000 | - | - | 1,380,836 | |||||||||||||||||||||||||||
Issued to acquire technology rights (notes 7 and 10) | 167,619 | 1,005,714 | - | - | 1,600,000 | 80,000 | - | - | 1,085,714 | |||||||||||||||||||||||||||
Expiry of warrants | - | - | - | - | (127,273 | ) | (91,636 | ) | 91,636 | - | - | |||||||||||||||||||||||||
Share-based compensation | - | - | - | - | - | - | 318,882 | - | 318,882 | |||||||||||||||||||||||||||
Total transactions with owners of the Company | 3,436,348 | 15,802,344 | 77,895,165 | 11,292,525 | 141,193,759 | 9,309,898 | 410,518 | - | 36,815,285 | |||||||||||||||||||||||||||
Balance, December 31, 2013 | 4,058,408 | 47,191,303 | 77,895,165 | 11,292,525 | 142,230,123 | 9,818,179 | 3,280,656 | (37,674,216 | ) | 33,908,447 | ||||||||||||||||||||||||||
See accompanying notes to the consolidated financial statements
F-5
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Consolidated Statements of Cash Flows
Amounts in Canadian Dollars
Note
| Year ended $ | Year ended $ | ||||||||||
OPERATING ACTIVITIES | ||||||||||||
Net loss for the year | (12,881,820 | ) | (4,289,308 | ) | ||||||||
Adjustments for items not affecting cash | ||||||||||||
Share-based compensation | 10 | 2,311,254 | 318,882 | |||||||||
Interest accretion | 9,14 | 69,770 | 40,133 | |||||||||
Amortization of intangible assets | 7 | 610,776 | 632,779 | |||||||||
Impairment of intangible assets | 7 | 429,763 | - | |||||||||
Depreciation of property and equipment | 6 | 47,208 | 16,010 | |||||||||
| (9,413,049 | ) | (3,281,504 | ) | |||||||||
Changes in non-cash working capital balances | ||||||||||||
Amounts receivable | 82,818 | 554,918 | ||||||||||
Prepaid expenses | (913,656 | ) | (21,264 | ) | ||||||||
Accounts payable and accrued liabilities | 2,579,124 | 202,424 | ||||||||||
Other current liabilities | 216,695 | 62,766 | ||||||||||
Cash used in operating activities | (7,448,068 | ) | (2,482,660 | ) | ||||||||
FINANCING ACTIVITIES | ||||||||||||
Change in loan payable | 9 | (115,031 | ) | 13,284 | ||||||||
Change in long-term liability | 9 | (47,759 | ) | 98,915 | ||||||||
Issue of share capital, net of issuance costs | 10 | 966,413 | 34,029,853 | |||||||||
Cash provided by financing activities | 803,623 | 34,142,052 | ||||||||||
INVESTING ACTIVITIES | ||||||||||||
Purchase of property and equipment | 6 | (173,603 | ) | (34,017 | ) | |||||||
Acquisition of Trillium Privateco, net of cash acquired | 4 | - | (647,996 | ) | ||||||||
Proceeds from marketable securities | 526,598 | 104,112 | ||||||||||
Cash provided by (used) in investing activities | 352,995 | (577,901 | ) | |||||||||
Net increase (decrease) in cash during the year | (6,291,450 | ) | 31,081,491 | |||||||||
Cash, beginning of year | 32,456,506 | 1,375,015 | ||||||||||
Cash, end of year | 26,165,056 | 32,456,506 | ||||||||||
Supplemental cash flow information | ||||||||||||
Common share purchase warrants issued as agent’s compensation (note 10) | - | 700,772 | ||||||||||
Common shares and warrants issued on acquisition of Trillium Privateco (note 4) | - | 1,380,836 | ||||||||||
Common shares and warrants issued on acquisition of technology rights (note 7) | - | 1,085,714 | ||||||||||
Preferred shares converted to common shares (note 10) | 1,216,374 | - | ||||||||||
See accompanying notes to the consolidated financial statements
F-6
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 1. | Corporate information |
Trillium Therapeutics Inc. (formerly Stem Cell Therapeutics Corp.) (the “Company” or “Trillium”) is a Canadian public immuno-oncology company developing innovative therapies for the treatment of cancer. The Company was incorporated under the laws of the Province of Alberta on March 31, 2004 with nominal share capital. On October 19, 2004, the Company changed its name from Neurogenesis Biotech Corp. to Stem Cell Therapeutics Corp. On November 7, 2013, the Company filed Articles of Continuance to change its jurisdiction to Ontario. On June 1, 2014, the Company amalgamated with its wholly-owned subsidiary Trillium Therapeutics Inc. (“Trillium Privateco”) and changed its name to Trillium Therapeutics Inc.
The Company’s head office is located at 96 Skyway Avenue, Toronto, Ontario, M9W 4Y9 and is listed on the Toronto Stock Exchange under the symbol TR and on the NASDAQ Stock Exchange under the symbol TRIL.
| 2. | Basis of presentation |
| (a) | Statement of compliance |
The consolidated financial statements have been prepared in accordance with International Financial Reporting Standards (“IFRS”), as issued by the International Accounting Standards Board (“IASB”).
These consolidated financial statements were approved by the Company’ Board of Directors on March 23, 2015.
| (b) | Basis of measurement |
These consolidated financial statements have been prepared on the historical cost basis, except for held-for-trading financial assets which are measured at fair value.
| (c) | Functional and presentation currency |
These consolidated financial statements are presented in Canadian dollars, which is the Company’s functional currency.
| (d) | Use of significant estimates and assumptions |
The preparation of financial statements in conformity with IFRS requires management to make judgments, estimates and assumptions that affect the application of accounting policies and the reported amounts of assets and liabilities, revenue and expenses and the related disclosures of contingent assets and liabilities and the determination of the Company’s ability to continue as a going concern. Actual results could differ materially from these estimates and assumptions. The Company reviews its estimates and underlying assumptions on an ongoing basis. Revisions are recognized in the period in which the estimates are revised and may impact future periods.
Management has applied significant estimates and assumptions to the following:
Valuation of share-based compensation and warrants
F-7
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 2. | Basis of presentation (continued) |
Management measures the costs for share-based compensation and warrants using market-based option valuation techniques. Assumptions are made and estimates are used in applying the valuation techniques. These include estimating the future volatility of the share price, expected dividend yield, expected risk-free interest rate, future employee turnover rates, future exercise behaviours and corporate performance. Such estimates and assumptions are inherently uncertain. Changes in these assumptions affect the fair value estimates of share-based payments and warrants.
Impairment of long lived assets
Long-lived assets are reviewed for impairment upon the occurrence of events or changes in circumstances indicating that the carrying value of the asset may not be recoverable. For the purpose of measuring recoverable amounts, assets are grouped at the lowest levels for which there are separately identifiable cash flows (cash-generating units). The recoverable amount is the higher of an asset’s fair value less costs to sell and value in use (being the present value of the expected future cash flows of the relevant asset or cash-generating unit). An impairment loss is recognized for the amount by which the asset’s carrying amount exceeds its recoverable amount. Management evaluates impairment losses for potential reversals when events or circumstances warrant such consideration.
Intangible assets
The Company estimates the useful lives of intangible assets from the date they are available for use in the manner intended by management and at least annually reviews the useful lives to reflect management’s intent about developing and commercializing the assets.
| 3. | Significant accounting policies |
The accounting policies set out below have been applied consistently to all periods presented in these consolidated financial statements.
| (a) | Basis of consolidation |
These consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries, Stem Cell Therapeutics Inc. to the date of its dissolution on September 17, 2014, and Trillium Privateco from April 9, 2013, the date of acquisition (see note 4) to the date of its amalgamation with the Company on June 1, 2014.
Investments in entities where the Company is exposed, or has rights, to variable returns from its involvement with the investee and has the ability to affect those returns through its power over the investee, are considered subsidiaries due to the control exercised over the investee by the Company. Subsidiaries are fully consolidated from the date at which control is determined to have occurred and are de-consolidated from the date that the Company no longer controls the entity.
Intercompany transactions, balances and unrealized gains and losses on transactions between subsidiaries are eliminated.
F-8
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
The financial statements of the subsidiaries are prepared for the same reporting period as the Company, using consistent accounting policies.
| (b) | Foreign currency |
Transactions in foreign currencies are translated to the functional currency at the rate on the date of the transactions. Monetary assets and liabilities denominated in foreign currencies are retranslated at the spot rate of exchange as at the reporting date. All differences are taken to profit or loss. Non-monetary items that are measured in terms of historical cost in a foreign currency are translated using the exchange rate as at the date of the initial transaction. Non-monetary items measured at fair value in a foreign currency are translated using the exchange rate at the date when the fair value was determined.
| (c) | Financial instruments |
Financial assets
A financial asset is classified at fair value through profit or loss if it is held for trading or is designated as such upon initial recognition. Attributable transaction costs are recognized in profit or loss as incurred. Financial assets at fair value through profit or loss are measured at fair value and changes therein are recognized in profit or loss.
Marketable securities
Marketable securities are comprised of short-term debt instruments with an original maturity of more than 90 days on purchase.
Loans and receivables
Loans and receivables are non-derivative financial assets with fixed or determinable payments that are not quoted in an active market. Loans and receivables are initially recognized at fair value plus transaction costs and subsequently measured at amortized cost using the effective interest rate method less any impairment losses. The Company has classified its amounts receivable as loans and receivables.
Derecognition
A financial asset is derecognized when the rights to receive cash flows from the asset have expired or when the Company has transferred its rights to receive cash flows from the asset.
Financial liabilities
Financial liabilities are recognized initially at fair value plus any directly attributable transaction costs, and subsequently at amortized cost using the effective interest method. The Company has classified its accounts payable and accrued liabilities, and loan payable as financial liabilities.
F-9
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
Derecognition
A financial liability is derecognized when its contractual obligations are discharged, cancelled or expired.
Equity
Common shares, preferred shares and warrants to purchase common shares are classified as equity. Incremental costs directly attributable to the issue of common shares, preferred shares and warrants are recognized as a deduction from equity, net of any tax effects.
| (d) | Property and equipment |
Recognition and measurement
Items of property and equipment are measured at cost less accumulated depreciation and accumulated impairment losses. Cost includes the expenditure that is directly attributable to the acquisition of the asset. When parts of an item of property and equipment have different useful lives, they are accounted for as separate items (major components) of property and equipment. Gains and losses on disposal of an item of property and equipment are determined by comparing the proceeds from disposal with the carrying amount of property and equipment, and are recognized in profit or loss.
Subsequent costs
The cost of replacing a part of an item of property and equipment is recognized in the carrying amount of the item if it is probable that the future economic benefits embodied within the part will flow to the Company, and its cost can be measured reliably. The carrying amount of the replaced part is then derecognized. The costs of the day-to-day servicing of property and equipment are recognized in profit or loss as incurred.
Depreciation
The estimated useful lives and the methods of depreciation for the current and comparative periods are as follows:
| Asset | Basis | |
Lab equipment | 20% declining balance | |
Computer equipment | Straight-line over 3 years | |
Office equipment | Straight-line over 5 years | |
Leaseholds | Straight-line over lease term | |
Estimates for depreciation methods, useful lives and residual values are reviewed at each reporting period-end and adjusted if appropriate. Depreciation expense is recognized in research and development expenses.
F-10
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
| (e) | Intangible assets |
Research and development
Expenditures on research activities, undertaken with the prospect of gaining new scientific or technical knowledge and understanding, are recognized in profit or loss as incurred.
Development activities involve a plan or design for the production of new or substantially improved products and processes. Development expenditures are capitalized only if development costs can be measured reliably, the product or process is technically and commercially feasible, future economic benefits are probable, and the Company intends to complete development and has sufficient resources to complete development and to use or sell the asset. Other development expenditures are expensed as incurred. No internal development costs have been capitalized to date.
Research and development expenses include all direct and indirect operating expenses supporting the products in development. The costs incurred in establishing and maintaining patents are expensed as incurred.
Intangible assets
Intangible assets that are acquired separately and have finite useful lives are measured at cost less accumulated amortization and accumulated impairment losses. Subsequent expenditures are capitalized only when they increase the future economic benefits embodied in the specific asset to which it relates. All other expenditure is recognized in profit or loss as incurred.
Amortization is recognized in profit or loss on a straight-line basis over the estimated useful lives of intangible assets from the date they are available for use in the manner intended by management. The period that the technologies acquired in the Trillium Privateco acquisition are available for use is estimated at three years, which reflects management’s intent about developing and commercializing the assets.
The amortization method and amortization period of an intangible asset with a finite life is reviewed at least annually. Changes in the expected useful life or the expected pattern of consumption of future economic benefits embodied in the asset are accounted for by changing the amortization period or method, as appropriate, and are treated as changes in accounting estimates. The amortization expense on intangible assets with finite lives is recognized in research and development expenses.
| (f) | Impairment |
Financial assets
A financial asset not carried at fair value through profit or loss is assessed at each reporting date to determine whether there is objective evidence that it is impaired. A financial asset is impaired if objective evidence indicates that a loss event has occurred after the initial recognition of the asset, and that the loss event had a negative effect on the estimated future cash flows of that asset that can be estimated reliably.
F-11
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
An impairment test is performed, on an individual basis, for each material financial asset. Other individually non-material financial assets are tested as groups of financial assets with similar risk characteristics. Impairment losses are recognized in profit or loss.
An impairment loss in respect of a financial asset measured at amortized cost is calculated as the difference between its carrying amount and the present value of the estimated future cash flows discounted at the asset’s original effective interest rate. Losses are recognized in profit or loss and reflected in an allowance account against the respective financial asset. Interest on the impaired asset continues to be recognized through the unwinding of the discount. When a subsequent event causes the amount of impairment loss to decrease, the decrease in impairment loss is reversed through profit or loss.
Non-financial assets
The carrying amounts of the Company’s non-financial assets are reviewed at each reporting date to determine whether there is any indication of impairment. If such an indication exists, the recoverable amount is estimated.
The recoverable amount of an asset or a cash-generating unit is the greater of its value in use and its fair value less costs to sell. In assessing value in use, the estimated future cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset or cash-generating unit. For the purpose of impairment testing, assets that cannot be tested individually are grouped together into the smallest group of assets that generates cash inflows from continuing use that are largely independent of cash inflows of other assets or cash-generating units. An impairment loss is recognized if the carrying amount of an asset or its related cash-generating unit exceeds its estimated recoverable amount. Impairment losses for intangible assets are recognized in research and development expenses.
Impairment losses recognized in prior periods are assessed at each reporting date for any indications that the loss has decreased or no longer exists. An impairment loss is reversed if there has been a change in the estimates used to determine the recoverable amount. An impairment loss is reversed only to the extent that the asset’s carrying amount does not exceed the carrying amount that would have been determined, net of depreciation or amortization, if no impairment loss had been recognized.
| (g) | Provisions |
A provision is recognized if, as a result of a past event, the Company has a present legal or constructive obligation that can be estimated reliably, and it is probable that an outflow of economic benefits will be required to settle the obligation. Provisions are assessed by discounting the expected future cash flows at a pre-tax rate that reflects current market assessments of the time value of money and the risks specific to the liability. The unwinding of the discount on provisions is recognized in finance costs.
A provision for onerous contracts is recognized when the unavoidable costs of meeting the obligations under the contract exceed the economic benefits expected to be received under it. The provision is measured at the present value of the lower of the expected cost of terminating the contract and the expected net cost of continuing with the contract.
F-12
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
| (h) | Government assistance |
Government assistance relating to research and development is recorded as a reduction of expenses when the related expenditures are incurred.
| (i) | Share-based compensation |
The grant-date fair value of share-based payment awards granted to employees is recognized as personnel costs, with a corresponding increase in contributed surplus, over the period that the employees unconditionally become entitled to the awards. The amount recognized as an expense is adjusted to reflect the number of awards for which the related service and non-market vesting conditions are expected to be met, such that the amount ultimately recognized as an expense is based on the number of awards that met the related service and non-market performance conditions at the vesting date.
For equity-settled share-based payment transactions, the Company measures the goods or services received, and the corresponding increase in contributed surplus, directly, at the fair value of the goods or services received, unless that fair value cannot be estimated reliably. If the Company cannot estimate reliably the fair value of the goods or services received, it measures their value by reference to the fair value of the equity instruments granted. Transactions measured by reference to the fair value of the equity instruments granted have their fair values remeasured at each vesting and reporting date until fully vested.
| (j) | Income taxes |
Deferred tax is recognized in respect of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for taxation purposes. Deferred tax is not recognized for temporary differences on the initial recognition of assets or liabilities in a transaction that is not a business combination and that affects neither accounting nor taxable income or loss.
Deferred tax assets and liabilities are offset if there is a legally enforceable right to offset current tax assets and liabilities, and they relate to income taxes levied by the same tax authority on the same taxable entity.
Deferred tax is measured at the tax rates that are expected to be applied to temporary differences when they reverse, based on the laws that have been enacted or substantively enacted at the reporting date. A deferred tax asset is recognized for unused tax losses, tax credits and deductible temporary differences, to the extent that it is probable that future taxable profits will be available against which they can be utilized.
Investment tax credits earned from scientific research and development expenditures are recorded when collectability is reasonably assured.
| (k) | Loss per share |
Basic loss per share is computed by dividing the net loss available to common shareholders by the weighted average number of shares outstanding during the reporting period. Diluted loss per share is
F-13
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
computed similar to basic loss per share except that the weighted average number of shares outstanding are increased to include additional shares for the assumed exercise of stock options, deferred share units, warrants, and conversion of preferred shares, if dilutive. The number of additional shares is calculated by assuming that outstanding preferred shares would convert to common shares and that outstanding stock options and warrants were exercised and that the proceeds from such exercises were used to acquire common stock at the average market price during the reporting period. The inclusion of the Company’s stock options, deferred share units, warrants and preferred shares in the computation of diluted loss per share has an anti-dilutive effect on the loss per share and therefore, they have been excluded from the calculation of diluted loss per share.
| (l) | New standards, amendments, and interpretations adopted during 2014 |
IAS 32Financial Instruments: Presentation
The amendments to IAS 32Financial Instruments: Presentation (“IAS 32”), clarify the criteria that should be considered in determining whether an entity has a legally enforceable right of set off in respect of its financial instruments. Amendments to IAS 32 were applicable to annual periods beginning on or after January 1, 2014, with retrospective application required. The adoption of these amendments did not have a material impact on the consolidated financial statements.
IFRIC 21Levies
In May 2013, International Financial Reporting Standards Interpretations Committee Interpretation 21Levies (“IFRIC 21”) was issued. IFRIC 21 provides guidance on when to recognize a liability to pay a levy imposed by a government that is accounted for in accordance with IAS 37Provisions, Contingent Liabilities and Contingent Assets. IFRIC 21 was effective for annual periods beginning on or after January 1, 2014 with retrospective application required. The adoption of IFRIC 21 did not have a material impact on the consolidated financial statements.
IAS 36Impairment of Assets
In May 29, 2013, the IASB published amendments to IAS 36Impairment of Assets (“IAS 36”) which reduce the circumstances in which the recoverable amount of cash-generating units is required to be disclosed and clarifies the disclosures required when an impairment loss has been recognized or reversed in the period. This amendment was effective for annual periods beginning on or after January 1, 2014. The adoption of these amendments did not have a material impact on the consolidated financial statements.
| (m) | New standards and interpretations not yet effective |
IFRS 9Financial Instruments
In October 2010, the IASB published amendments to IFRS 9Financial Instruments(“IFRS 9”) which provide added guidance on the classification and measurement of financial liabilities. In July 2014, the
F-14
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
IASB issued its final version of IFRS 9, which completes the classification and measurement, impairment and hedge accounting phases of the IASB’s project to replace IAS 39Financial Instruments: Recognition and Measurement. The final standard is mandatorily effective for annual periods beginning on or after January 1, 2018, with earlier application permitted. The Company is currently assessing the impact that the adoption of this standard may have on the consolidated financial statements.
IFRS 15Revenue from Contracts with Customers
In May 2014, the IASB issued IFRS 15Revenue from Contracts with Customers(“IFRS 15”), which covers principles for reporting about the nature, amount, timing and uncertainty of revenue and cash flows arising from contracts with customers. IFRS 15 is effective for annual periods beginning on or after January 1, 2017. The Company is reviewing the standard to determine the impact that the adoption of the standard may have on the consolidated financial statements.
Other accounting standards or amendments to existing accounting standards that have been issued, but have future effective dates, are either not applicable or are not expected to have a significant impact on the Company’s consolidated financial statements.
| 4. | Acquisition of Trillium Privateco |
On April 9, 2013, the Company completed a merger with Trillium Privateco, a private biopharmaceutical company specializing in immune regulation and the development of cancer therapeutics. The Company applied the acquisition method of accounting for the business combination and the losses have been consolidated from the acquisition date. The consideration was allocated to the underlying assets acquired and liabilities assumed based upon their fair values at the date of acquisition. The purchase price allocation
F-15
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 4. | Acquisition of Trillium Privateco (continued) |
was assigned to the net identifiable assets based on their estimated fair values. The final purchase price allocation was as follows:
| $ | ||||
Fair value of consideration paid: | ||||
Cash | 1,200,000 | |||
92,639 common shares and 110,000 units | 1,380,836 | |||
| 2,580,836 | ||||
Assets acquired: | ||||
Cash and marketable securities | 1,182,714 | |||
Amounts receivable | 820,944 | |||
Prepaid expenses | 44,300 | |||
Equipment | 91,000 | |||
Acquired technology | 1,018,037 | |||
| 3,156,995 | ||||
Liabilities assumed: | ||||
Accounts payable and accrued liabilities | 282,178 | |||
Loan payable | 293,981 | |||
| 576,159 | ||||
Net identifiable assets acquired | 2,580,836 | |||
As consideration, the Company paid $1,200,000 in cash and issued 92,639 common shares and 110,000 units. Each unit consisted of one common share and 30 common share purchase warrants, with each 30 common share purchase warrants allowing their holder to acquire one additional common share at an exercise price of $12.00 until March 15, 2018. The fair value of the unit was determined based on the stand-alone value of the common share and the common share purchase warrant, in accordance with IFRS 13Fair Value Measurement and IFRS 3Business Combinations. Accordingly, the fair value of the aggregate 202,639 common shares issued was based on the closing price of the Company’s common shares of $6.00 on April 9, 2013. The fair value of the 3,300,000 common share purchase warrants was determined using the Black-Scholes option pricing model. Assumptions used to determine the value of the common share purchase warrant were: dividend yield of 0%; risk-free interest rate of 1.1%; expected volatility of 68%; and average expected life of 3 years. Post-closing, Trillium Privateco shareholders held approximately 16% of the issued and outstanding common shares, and Trillium Privateco became a wholly-owned subsidiary of the Company.
Cash used in the investment was determined as follows:
| $ | ||||
Cash consideration | 1,200,000 | |||
Less cash acquired | 552,004 | |||
| 647,996 | ||||
F-16
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 4. | Acquisition of Trillium Privateco (continued) |
Acquisition costs incurred by the Company and included in general and administrative expenses for the year ended December 31, 2013, were approximately $113,000. From the date of the acquisition to December 31, 2013, Trillium Privateco contributed revenue of nil and a loss of $2,236,262.
| 5. | Amounts receivable |
December 31, $ | December 31, $ | |||||||
Government programs receivable | 344,416 | 426,946 | ||||||
Other amounts receivable | - | 288 | ||||||
| 344,416 | 427,234 | |||||||
| 6. | Property and equipment |
Lab $ | Computer $ | Office and $ | Total $ | |||||||||||||
Cost | ||||||||||||||||
Balance, December 31, 2012 | - | 13,500 | - | 13,500 | ||||||||||||
Additions | 111,025 | 4,611 | 9,381 | 125,017 | ||||||||||||
Balance, December 31, 2013 | 111,025 | 18,111 | 9,381 | 138,517 | ||||||||||||
Additions | 141,051 | 21,917 | 10,635 | 173,603 | ||||||||||||
Balance, December 31, 2014 | 252,076 | 40,028 | 20,016 | 312,120 | ||||||||||||
Accumulated depreciation | ||||||||||||||||
Balance, December 31, 2012 | - | 13,500 | - | 13,500 | ||||||||||||
Depreciation | 14,723 | 504 | 783 | 16,010 | ||||||||||||
Balance, December 31, 2013 | 14,723 | 14,004 | 783 | 29,510 | ||||||||||||
Depreciation | 33,366 | 9,554 | 4,288 | 47,208 | ||||||||||||
Balance December 31, 2014 | 48,089 | 23,558 | 5,071 | 76,718 | ||||||||||||
Net carrying amounts | ||||||||||||||||
December 31, 2013 | 96,302 | 4,107 | 8,598 | 109,007 | ||||||||||||
December 31, 2014 | 203,987 | 16,470 | 14,945 | 235,402 | ||||||||||||
F-17
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 6. | Property and equipment (continued) |
The Company acquired lab equipment with a fair value of $91,000 on its acquisition of Trillium Privateco in 2013.
| 7. | Intangible assets |
Total $ | ||||
Cost | ||||
Balance, December 31, 2012 | 2,431,279 | |||
Additions | 2,103,751 | |||
Disposals | (2,431,279 | ) | ||
Balance December 31, 2013 | 2,103,751 | |||
Disposals | (1,085,714 | ) | ||
Balance December 31, 2014 | 1,018,037 | |||
Accumulated amortization | ||||
Balance, December 31, 2012 | 2,428,779 | |||
Amortization | 632,779 | |||
Disposals | (2,431,279 | ) | ||
Balance, December 31, 2013 | 630,279 | |||
Amortization | 610,776 | |||
Disposals | (655,951 | ) | ||
Balance, December 31, 2014 | 585,104 | |||
Net carrying amounts | ||||
December 31, 2013 | 1,473,472 | |||
December 31, 2014 | 432,933 | |||
On April 9, 2013, licensed patent rights acquired of Trillium Privateco amounted to $1,018,037 (see note 4).
On April 16, 2013, the Company signed an agreement with the University Health Network (“UHN”) to gain rights to intellectual property related to the use of tigecycline for the treatment of leukemia. The initial consideration for the UHN license of $1,085,714 was satisfied by the issuance of 167,619 common shares and 1,600,000 common share purchase warrants, with each 30 warrants allowing their holder to acquire one additional common share at an exercise price of $12.00 until March 15, 2018. Additional consideration under the UHN license included an annual license maintenance fee and future development milestones. The fair value of the common shares issued was based on the closing price of the Company’s common shares of $6.00 on April 16, 2013 and $7.50 per unit based on the fair value of the common share and common share purchase warrant components as of the date of acquisition. The Company returned the rights back to UHN and recorded an impairment loss of $429,763 in the second quarter of 2014.
F-18
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 7. | Intangible assets (continued) |
In 2013, the Company ceased all activities related to the regenerative stem cell products and retired the fully amortized assets in the amount of $2,431,279.
| 8. | Accounts payable and accrued liabilities |
December 31, $ | December 31, $ | |||||||
Trade and other payables | 1,604,533 | 113,003 | ||||||
Accrued liabilities | 1,585,823 | 221,580 | ||||||
Due to related parties (note 16) | 58,628 | 335,277 | ||||||
| 3,248,984 | 669,860 | |||||||
| 9. | Non-current liabilities |
| (a) | Trillium is indebted to the Federal Economic Development Agency for Southern Ontario under a non-interest-bearing contribution agreement dated February 1, 2012. The period of contribution ended on December 31, 2013. As at December 31, 2014, the Company has repayable contributions of $555,968. The original principal balance is repayable in equal monthly instalments for 60 months, which began on December 1, 2014. As at December 31, 2014 two instalment payments have been made. The loan payable bears no interest. The fair value of the loan payable was determined by discounting the loan payable using an estimated market interest rate of 15%. Interest expense accretes on the discounted loan amount until it reaches its face value at maturity. |
| (b) | The Company has a long-term liability of $69,941 related to certain discontinued technologies. This liability has been discounted using an estimated market interest rate of 15% and interest expense is accreting. |
| The current portions of the loan payable and long-term liability are included in other current liabilities in the statements of financial position. |
| 10. | Share capital |
| (a) | Authorized |
The authorized share capital of the Company consists of an unlimited number of common shares, Class B shares and First Preferred Shares, in each case without nominal or par value. Common shares are voting and may receive dividends as declared at the discretion of the Board of Directors. Class B shares are non-voting and convertible to common shares at the holder’s discretion, on a one-for-one basis. Upon dissolution or wind-up of the Company, Class B shares participate rateably with the common shares in the distribution of the Company’s assets. Preferred shares have voting rights as decided upon by the Board of |
F-19
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
| Directors at the time of grant. Upon dissolution or wind-up of the Company, First Preferred Shares are entitled to priority over common and Class B shares. |
| In December 2013, the Company created the Series I First Preferred Shares that are non-voting, may receive dividends as declared at the discretion of the Board of Directors, and are convertible to common shares at the holder’s discretion, on the basis of 30 Series I First Preferred Shares for each common share. |
| (b) | Share capital issued – year ended December 31, 2014 |
| On November 14, 2014, the Company consolidated its outstanding common shares issuing one post-consolidated share for each 30 pre-consolidated shares. All references in these consolidated financial statements and notes to the number of common shares, deferred share units and stock options have been adjusted to the post-consolidation amounts. |
| In the year ended December 31, 2014, 2,596,251 warrants were exercised for 86,540 common shares and for proceeds of $946,813 and 2,614 stock options were exercised for proceeds of $19,600. Also, 909,091 warrants issued in March 2011 expired unexercised. |
| During the year ended December 31, 2014, 8,390,476 Series I First Preferred Shares were converted into 279,682 common shares. |
Share capital issued – year ended December 31, 2013
| On February 6, 2013, the Company consolidated its outstanding common shares issuing one post-consolidated share for each 10 pre-consolidated shares. |
In March 2013, the Company completed an offering pursuant to a base shelf prospectus and prospectus supplement and in the U.S. pursuant to a private placement memorandum for a total of 424,500 units at a price of $7.50 per unit, for aggregate gross proceeds to the Company of $3,185,080 ($2,615,240 net of issuance costs). Each unit consisted of one common share and 30 common share purchase warrants. Each 30 warrants entitle the holder to purchase one common share at a price of $12.00 per share at any time prior to expiry on March 15, 2018 (12,315,000 warrants) and March 27, 2018 (420,000 warrants). In connection with the financing, the Company issued 814,051 compensation warrants having an aggregate fair value of $48,843 estimated using the Black-Scholes option pricing model. Each 30 compensation warrants entitle the holder to acquire one common share at an exercise price of $7.50 per share prior to expiry on March 16, 2015.
The allocation of the $7.50 common share unit issue price to the common shares and unit warrants was based on the relative fair values of the common shares and the warrants. The fair value of the warrants was determined using the Black-Scholes option pricing model. The common shares were allocated a price of $6.00 per share and the warrants were allocated a price of $1.50 per 30 warrants. The costs of the issue were allocated on a pro rata basis to the common shares and warrants. Accordingly, $2,092,192 was
F-20
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
allocated to common shares and $523,048 to warrants, net of issuance costs. Assumptions used to determine the value of the unit warrants were: dividend yield of 0%; risk-free interest rate of 1.1%; expected volatility of 64%; and average expected life of 3 years. Assumptions used to determine the value of the compensation warrants were: dividend yield of 0%; risk-free interest rate of 1.0%; expected volatility of 80%; and average expected life of 2 years.
On April 9, 2013, the Company issued 202,639 common shares and 3,300,000 common share purchase warrants for partial consideration for the acquisition of Trillium Privateco (see note 4).
On April 16, 2013, the Company issued 167,619 common shares and 1,600,000 common share purchase warrants as consideration for the acquisition of certain rights related to tigecycline from UHN (see note 7).
On December 13, 2013, the Company completed a private placement of 2,641,590 common share units (each common share unit consisting of one common share and 22.5 common share purchase warrants) at a price of $6.30 per unit and 77,895,165 preferred share units (each preferred share unit consisting of one Series I First Preferred Share and three-quarters of a common share purchase warrant) at a price of $0.21 per unit for gross proceeds of $32,866,025 ($30,713,841 net of issuance costs). Each 30 warrants entitle the holder to purchase one common share at a price of $8.40 at any time prior to expiry on December 13, 2018. In connection with the financing, the Company issued 5,014,839 compensation warrants having an aggregate fair value of $651,929 estimated using the Black-Scholes option pricing model. Each 30 compensation warrants entitle the holder to acquire one common share at an exercise price of $6.30 per share prior to expiry on December 13, 2015.
The allocation of the $6.30 unit issue price to the common shares, 30 Series I First Preferred Shares and 22.5 unit warrants was based on the relative fair values of the common shares, Series I First Preferred Shares and the warrants. The fair value of the warrants was determined using the Black-Scholes option pricing model. The common shares were allocated a price of $4.80 per share, the Series I First Preferred Shares were allocated a price of $4.80 per each 30 shares, and the warrants were allocated a price of $1.50 per each 22.5 warrants. The costs of the issue were allocated on a pro rata basis. Accordingly, $11,488,602 was allocated to common shares, $11,292,525 to the Series I First Preferred Shares and $7,932,714 to warrants, net of issuance costs. Assumptions used to determine the value of the unit warrants were: dividend yield of 0%; risk-free interest rate of 1.2%; expected volatility of 70%; and average expected life of 3 years. Assumptions used to determine the value of the compensation warrants were: dividend yield of 0%; risk-free interest rate of 1.1%; expected volatility of 66%; and average expected life of 2 years.
All securities issued under the offering (including the compensation warrants) are subject to statutory resale restrictions.
In the December 2013 private placement, subscribers who purchased preferred share units and certain subscribers who purchased common share units are subject to restrictions on the conversion and exercise of securities of the Company convertible into common shares. Such subscribers cannot convert or exercise securities of the Company convertible or exercisable into common shares if, after giving effect to the
F-21
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
exercise of conversion, the subscriber and its joint actors would have beneficial ownership or direction or control over common shares in excess of 4.99% of the then outstanding common shares. This limit can be raised at the option of the subscriber on 61 days prior written notice: (i) up to 9.99%, (ii) up to 19.99%, subject to stock exchange clearance of a personal information form submitted by the subscriber, and (iii) above 19.99%, subject to stock exchange approval and shareholder approval.
Subject to receipt of any required regulatory approvals, subscribers who purchased a minimum of 10% of the securities sold under the offering were given rights to purchase securities of the Company in future financings to enable each such subscriber to maintain its percentage holding in the Company for so long as the subscriber holds at least 10% of the outstanding common shares on a fully-diluted basis.
| (c) | Weighted average number of common shares |
The weighted average number of common shares outstanding for the purposes of calculating earnings per share have been adjusted for 2014 and 2013 to the post-consolidated number. The post-consolidated weighted average number of common shares outstanding for the years ended December 31, 2014 and 2013 were 4,202,900 and 1,356,924, respectively. The Company has not adjusted its weighted average number of common shares outstanding in the calculation of diluted loss per share, as any adjustment would be antidilutive.
| (d) | Warrants |
Warrants outstanding at December 31, 2014 were as follows:
| Expiry dates | Number of warrants | Exercise price | ||||||
March 16, 2015 | 202,800 | $ | 7.50 | |||||
December 13, 2015 | 5,014,839 | $ | 6.30 | |||||
March 15, 2018 | 15,230,000 | $ | 12.00 | |||||
March 27, 2018 | 420,000 | $ | 12.00 | |||||
December 13, 2018 | 117,857,142 | $ | 8.40 | |||||
| 138,724,781 | ||||||||
All warrants were exercisable on issuance. The exercise price above and the weighted average exercise price below are the amounts the warrantholder would pay to exercise 30 warrants and receive one
F-22
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
common share. Changes in the number of warrants outstanding during the years ended December 31 were as follows:
| 2014 | 2013 | |||||||||||||||
| Number of warrants | Weighted price | Number of warrants | Weighted average exercise price | |||||||||||||
Balance, beginning of year | 142,230,123 | $ | 9.02 | 1,036,364 | $ | 46.16 | ||||||||||
Issued | - | - | 141,321,032 | 8.77 | ||||||||||||
Exercised | (2,596,251 | ) | 10.94 | - | - | |||||||||||
Expired | (909,091 | ) | 48.00 | (127,273 | ) | 33.00 | ||||||||||
Balance, end of year | 138,724,781 | $ | 8.73 | 142,230,123 | $ | 9.02 | ||||||||||
| (e) | Stock option plan |
The Company has a 10% rolling stock option plan (the “2014 Stock Option Plan”) that was approved by the Company’s shareholders at its annual general meeting held on May 27, 2014. Pursuant to the 2014 Stock Option Plan, the Company may grant stock options to purchase up to an aggregate of 10% of the Company’s issued and outstanding common shares plus 10% of the total number of common shares into which the outstanding Series I First Preferred Shares may be converted. Options granted under the 2014 Stock Option plan are equity-settled, have a vesting period of four years and have a maximum term of ten years. As at December 31, 2014, the Company was entitled to issue an additional 55,488 stock options under the 2014 Stock Option Plan.
Changes in the number of options outstanding during the years ended December 31 were as follows:
| 2014 | 2013 | |||||||||||||||
| ||||||||||||||||
| Number of options | Weighted average exercise price | Number of options | Weighted average exercise price | |||||||||||||
Balance, beginning of year | 97,372 | $ | 9.94 | 23,332 | $ | 31.02 | ||||||||||
Granted | 499,883 | 9.78 | 87,454 | 7.50 | ||||||||||||
Exercised | (2,614 | ) | 7.50 | - | - | |||||||||||
Cancelled/forfeited | (4,500 | ) | 16.58 | (13,414 | ) | 30.69 | ||||||||||
| ||||||||||||||||
Balance, end of year | 590,141 | $ | 9.76 | 97,372 | $ | 9.94 | ||||||||||
| ||||||||||||||||
Options exercisable, end of year | 219,470 | $ | 10.13 | 39,667 | $ | 13.44 | ||||||||||
F-23
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
The following table reflects stock options outstanding at December 31, 2014:
| Stock options outstanding | Stock options exercisable | |||||||||||||||||||
| Exercise prices | Number outstanding | Weighted average (in years) | Weighted average | Number exercisable | Weighted average | |||||||||||||||
$7.50 | 81,507 | 7.6 | $ | 7.50 | 44,092 | $ | 7.50 | |||||||||||||
$8.34 | 215,758 | 9.4 | $ | 8.34 | 71,918 | $ | 8.34 | |||||||||||||
$10.35 | 264,127 | 9.3 | $ | 10.35 | 88,043 | $ | 10.35 | |||||||||||||
$15.30 | 6,666 | 9.1 | $ | 15.30 | 2,222 | $ | 15.30 | |||||||||||||
$18.90 | 13,332 | 9.2 | $ | 18.90 | 4,444 | $ | 18.90 | |||||||||||||
$30.00 | 8,751 | 1.1 | $ | 30.00 | 8,751 | $ | 30.00 | |||||||||||||
| 590,141 | 9.0 | $ | 9.76 | 219,470 | $ | 10.13 | ||||||||||||||
Share-based compensation expense was determined based on the fair value of the options at the date of measurement using theBlack-Scholes option pricing model with the weighted average assumptions for the years ended December 31 as follows:
| 2014 | 2013 | |||||||
Expected option life | 6 years | 10 years | ||||||
Risk-free interest rate | 1.7% | 1.7% | ||||||
Dividend yield | 0% | 0% | ||||||
Expected volatility | 90% | 118% | ||||||
TheBlack-Scholes option pricing model was developed to estimate the fair value of freely tradable, fully transferable options without vesting restrictions, which significantly differs from the Company’s stock option awards. This model also requires highly subjective assumptions, including future stock price volatility and average option life, which significantly affect the calculated values.
The risk-free interest rate is based on the implied yield on a Government of Canada zero-coupon issue with a remaining term equal to the expected term of the option. The volatility for 2013 is based solely on historical volatility equal to the expected life of the option. Due to the significant changes in the operations of the Company in 2013, the Company concluded that for 2014 the sole use of historical volatility would not reflect the best estimate of forward looking volatility. For 2014, the Company compiled a peer group of biotechnology companies and their historical volatilities and integrated the Company’s own historical
F-24
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
volatility to determine a reasonable estimate of forward looking volatility. The life of the options is estimated considering the vesting period at the grant date, the life of the option and the average length of time similar grants have remained outstanding in the past. The forfeiture rate is an estimate based on historical evidence and future expectations. The dividend yield was excluded from the calculation since it is the present policy of the Company to retain all earnings to finance operations and future growth.
During years ended December 31, 2014 and 2013, the Company issued 499,883 and 87,454 stock options with a fair value of $3,580,892 and $535,781 and a weighted average grant date fair value of $7.16 and $6.13, respectively.
| (f) | Deferred Share Unit Plan |
The shareholders of the Company approved the 2014 Deferred Share Unit Plan (the “2014 DSU Plan”) on May 27, 2014. The 2014 DSU Plan is intended to promote a greater alignment of long-term interests between non-executive directors and executive officers of the Company and its shareholders through the issuance of deferred share units (“DSUs”). Since the value of a DSU increases or decreases with the market price of the common shares, DSUs reflect a philosophy of aligning the interests of directors and executive officers with those of the shareholders by tying compensation to share price performance. The Board of Directors intends to use DSUs issued under the 2014 DSU Plan, as well as stock options issued under the 2014 Stock Option Plan, as part of the Company’s overall director and executive officer compensation program. A total of 28,777 units were issued during the year ended December 31, 2014 for payment of directors’ fees and were outstanding as at December 31, 2014. The Company has reserved for issuance up to 66,667 common shares under the 2014 DSU Plan.
| (g) | Shareholder Rights Plan |
On October 17, 2013 the Company’s shareholders adopted a shareholder rights plan (the “2013 Rights Plan”) and approved certain amendments on May 27, 2014 (the “Rights Plan Amendment” which together with the 2013 Rights Plan may be referred to as the “Rights Plan”). The Rights Plan is designed to provide adequate time for the Board of Directors and the shareholders to assess an unsolicited takeover bid for the Company, to provide the Board of Directors with sufficient time to explore and develop alternatives for maximizing shareholder value if a takeover bid is made, and to provide shareholders with an equal opportunity to participate in a takeover bid and receive full and fair value for their common shares. The Rights Plan will expire at the close of the Company’s annual meeting of shareholders in 2016.
The rights issued under the Rights Plan initially attach to and trade with the common shares and no separate certificates will be issued unless an event triggering these rights occurs. The rights will become exercisable only when a person, including any party related to it, acquires or attempts to acquire 20% or more of the outstanding common shares without complying with the “Permitted Bid” provisions of the Rights Plan or without approval of the Board of Directors. Should such an acquisition occur or be announced, each right would, upon exercise, entitle a rights holder, other than the acquiring person and related persons, to purchase common shares at an approximate 50% discount to the market price at the time.
F-25
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
Under the Rights Plan, a Permitted Bid is a bid made to all holders of the common shares and which is open for acceptance for not less than 60 days. If at the end of 60 days at least 50% of the outstanding common shares, other than those owned by the offeror and certain related parties have been tendered, the offeror may take up and pay for the common shares but must extend the bid for a further 10 days to allow other shareholders to tender. The issuance of common shares upon the exercise of the rights is subject to receipt of certain regulatory approvals.
| 11. | Income taxes |
Income taxes have not been recognized in the consolidated statements of loss and comprehensive loss, as the Company has been incurring losses since inception, and it is not probable that future taxable profits will be available against which the accumulated tax losses can be utilized.
| (a) | Unrecognized deferred tax assets |
As at December 31, 2014 and 2013, deferred tax assets have not been recognized with respect to the following items:
2014 $ | 2013 $ | |||||||
Non-capital losses carried forward | 9,234,460 | 8,515,351 | ||||||
Tax credits carryforward | 2,607,496 | 2,097,043 | ||||||
Tax basis of property and equipment and intangible assets in excess of accounting basis | 1,413,171 | 1,138,196 | ||||||
Scientific research and experimental development expenditures | 4,801,014 | 4,101,322 | ||||||
Share issue costs and other | 436,795 | 540,886 | ||||||
| 18,492,936 | 16,392,798 | |||||||
| (b) | As at December 31, 2014 and 2013, the Company has available research and development expenditures of approximately $18,117,000 and $15,477,000, respectively, for income tax purposes which may be carried forward indefinitely to reduce future years’ taxable income. As at December 31, 2014 and 2013, the Company also has unclaimed Canadian scientific research and development tax credits of $3,293,000 and $2,643,000, respectively, which are available to reduce future taxes payable with expiries from 2017 through 2034. The benefit of these expenditures and tax credits has not been recorded in the accounts. |
| (c) | As at December 31, 2014, the Company has accumulated non-capital losses for federal and provincial income tax purposes in Canada which are available for application against future taxable income. The benefit of these losses has not been recorded in the accounts. |
F-26
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 11. | Income taxes (continued) |
The non-capital tax losses expire as follows:
Federal $ | ||||
2025 | 3,213,000 | |||
2026 | 6,457,000 | |||
2027 | 4,659,000 | |||
2028 | 4,144,000 | |||
2029 | 3,736,000 | |||
2030 | 1,819,000 | |||
2031 | 1,387,000 | |||
2032 | 2,450,000 | |||
2033 | 1,868,000 | |||
2034 | 5,113,000 | |||
| 34,846,000 | ||||
| (d) | The reconciliation of the Canadian statutory income tax rate applied to the net loss for the year to the income tax recovery is as follows: |
2014 $ | 2013 $ | |||||||
Statutory income tax rate | 26.5% | 26.5% | ||||||
Income tax recovery based on statutory income tax rate | (3,413,682 | ) | (1,136,667 | ) | ||||
Investment tax credits | (1,091,870 | ) | (207,850 | ) | ||||
Share-based compensation and other | 657,494 | 84,902 | ||||||
Change in unrecognized tax assets | 3,848,058 | 1,259,615 | ||||||
Income tax recovery | - | - | ||||||
F-27
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 12. | Research and development |
Components of research and development expenses for the years ended December 31 were as follows:
2014 $ | 2013 $ | |||||||
Research and development programs, excluding the below items | 5,893,030 | 1,411,264 | ||||||
Salaries, fees and short-term benefits | 2,311,755 | 1,299,055 | ||||||
Share-based compensation | 1,626,824 | 211,419 | ||||||
Amortization of intangible assets | 610,776 | 632,779 | ||||||
Impairment of intangible assets | 429,763 | - | ||||||
Depreciation of property and equipment | 47,208 | 16,010 | ||||||
Tax credits | (323,548 | ) | (233,821 | ) | ||||
| 10,595,808 | 3,336,706 | |||||||
| 13. | General and administrative |
Components of general and administrative expenses for the years ended December 31 were as follows:
2014 $ | 2013 $ | |||||||
General and administrative expenses, excluding the below items | 1,198,181 | 599,785 | ||||||
Salaries, fees and short-term benefits | 694,849 | 254,952 | ||||||
DSU units issued for director compensation | 240,000 | - | ||||||
Share-based compensation | 444,430 | 107,463 | ||||||
| 2,577,460 | 962,200 | |||||||
| 14. | Finance income and finance costs |
Finance income for the years ended December 31 was as follows:
2014 $ | 2013 $ | |||||||
Interest income | 378,692 | 44,113 | ||||||
Net foreign currency gain | - | 9,915 | ||||||
| 378,692 | 54,028 | |||||||
F-28
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 14. | Finance income and finance costs (continued) |
Finance costs for the years ended December 31 were as follows:
2014 $ | 2013 $ | |||||||
Bank charges | 7,212 | 4,297 | ||||||
Accreted interest | 69,770 | 40,133 | ||||||
Net foreign currency loss | 10,262 | - | ||||||
| 87,244 | 44,430 | |||||||
| 15. | Commitments and contingencies |
As at December 31, 2014 and in the normal course of business, the Company had obligations to make future payments, representing research and development contracts and other commitments that are known and committed in the amount of $2,587,000 over the next 12 months, $99,000 from 12 to 24 months, $26,000 from 24 to 36 months, and $25,000 each year thereafter.
The Company enters into research, development and license agreements in the ordinary course of business where the Company receives research services and rights to proprietary technologies. Milestone and royalty payments that may become due under various agreements are dependent on, among other factors, clinical trials, regulatory approvals and ultimately the successful development of a new drug, the outcome and timing of which is uncertain. Under the license agreement for SIRPaFc, the Company has future contingent milestones payable of $60,000 related to successful patent grants, $100,000, $200,000 and $300,000 on the first patient dosed in phase I, II and III trials respectively, and regulatory milestones on their first achievement totalling $5,000,000.
The Company entered into two agreements with Catalent Pharma Solutions in August 2014 pursuant to which Trillium acquired the right to use a proprietary expression system for the manufacture of two SIRPaFc constructs. Consideration for each license includes potential pre-marketing approval milestones of up to U.S. $875,000 and aggregate sales milestone payments of up to U.S. $28.8 million.
The Company periodically enters into research and license agreements with third parties that include indemnification provisions customary in the industry. These guarantees generally require the Company to compensate the other party for certain damages and costs incurred as a result of claims arising from research and development activities undertaken by or on behalf of the Company. In some cases, the maximum potential amount of future payments that could be required under these indemnification provisions could be unlimited. These indemnification provisions generally survive termination of the underlying agreement. The nature of the indemnification obligations prevents the Company from making a reasonable estimate of the maximum potential amount it could be required to pay. Historically, the Company has not made any indemnification payments under such agreements and no amount has been accrued in the consolidated financial statements with respect to these indemnification obligations.
F-29
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 16. | Related parties |
For the years ended December 31, 2014 and 2013, the key management personnel of the Company were the Board of Directors, former Executive Chairman, Chief Executive Officer, Chief Financial Officer, Chief Scientific Officer and Vice President, Drug Development.
Compensation for key management personnel of the Company for the years ended December 31 was as follows:
2014 $ | 2013 $ | |||||||
Salaries, fees and short-term benefits | 1,708,717 | 844,425 | ||||||
Share-based compensation | 2,281,561 | 316,069 | ||||||
Total | 3,990,278 | 1,160,494 | ||||||
Executive officers and directors participate in the 2014 Stock Option Plan and the 2014 DSU Plan, and officers participate in the Company’s benefit plans. Directors receive annual fees for their services. As at December 31, 2014, the key management personnel controlled approximately 1% of the voting shares of the Company.
Outstanding balances with related parties at the year-end are unsecured, interest free and settlement occurs in cash. There have been no guarantees provided or received for any related party receivables or payables. For the years ended December 31, 2014 and 2013, a former director was paid consulting fees of $7,916 and $138,750, respectively.
| 17. | Operating segment |
The Company has a single operating segment, the research and development of immunotherapy drugs for the treatment of cancer. Substantially all of the Company’s operations, assets, and employees are in Canada.
| 18. | Management of capital |
The Company defines its capital as share capital, warrants and contributed surplus. The Company’s objectives when managing capital are to ensure there are sufficient funds available to carry out its research and development programs. To date, these programs have been funded primarily through the sale of equity securities and the exercise of common share purchase warrants. The Company also sources non-dilutive funding by accessing grants, government assistance and tax incentives, and through partnerships with corporations and research institutions. The Company uses budgets and purchasing controls to manage its costs. The Company is not exposed to any externally imposed capital requirements.
F-30
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 19. | Financial instruments |
Fair value
IFRS 13 Fair Value Measurement provides a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable or unobservable. Observable inputs are those which reflect market data obtained from independent sources, while unobservable inputs reflect the Company’s assumptions with respect to how market participants would price an asset or liability. These two inputs used to measure fair value fall into the following three different levels of the fair value hierarchy:
Level 1 Quoted prices in active markets for identical instruments that are observable.
Level 2 Quoted prices in active markets for similar instruments; inputs other than quoted prices that are observable and derived from or corroborated by observable market data.
Level 3 Valuations derived from valuation techniques in which one or more significant inputs are unobservable.
The hierarchy requires the use of observable market data when available.
The Company has classified cash and marketable securities as Level 1. The loan payable has been classified as Level 2.
Cash, marketable securities, amounts receivable, accounts payable and accrued liabilities, and other current liabilities, due within one year, are all short-term in nature and, as such, their carrying values approximate fair values. The fair value of the non-current loan payable is estimated by discounting the expected future cash flows at the cost of money to the Company, which is equal to its carrying value.
Risks
The Company has exposure to credit risk, liquidity risk, interest rate risk and currency risk. The Company’s Board of Directors has overall responsibility for the establishment and oversight of the Company’s risk management framework. The Audit Committee of the Board is responsible for reviewing the Company’s risk management policies.
| (a) | Credit risk |
Credit risk is the risk of financial loss to the Company if a counterparty to a financial instrument fails to meet its contractual obligations, and arises principally from the Company’s cash, marketable securities, and amounts receivable. The carrying amount of these financial assets represents the maximum credit exposure. The Company follows an investment policy to mitigate against the deterioration of principal and to enhance the Company’s ability to meet its liquidity needs. Cash is on deposit with major Canadian chartered banks and the Company invests in high grade fixed income securities. Amounts receivable are primarily comprised of amounts due from the federal government.
F-31
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly Stem Cell Therapeutics Corp.)
Notes to the Consolidated Financial Statements
For the years ended December 31, 2014 and 2013
Amounts in Canadian Dollars
| 19. | Financial instruments (continued) |
| (b) | Liquidity risk |
Liquidity risk is the risk that the Company will not be able to meet its financial obligations as they fall due. The Company is a development stage company and is reliant on external fundraising to support its operations. Once funds have been raised, the Company manages its liquidity risk by investing in cash and marketable securities to provide regular cash flow for current operations. It also manages liquidity risk by continuously monitoring actual and projected cash flows. The Board of Directors reviews and approves the Company’s operating and capital budgets, as well as any material transactions not in the ordinary course of business. The majority of the Company’s accounts payable and accrued liabilities have maturities of less than three months.
| (c) | Interest rate risk |
Interest rate risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market interest rates. The Company holds its cash in bank accounts or high interest savings accounts which have a variable rate of interest. The Company manages its interest rate risk by holding highly liquid short-term instruments and by holding its investments to maturity, where possible. For the years ended December 31, 2014 and 2013, the Company earned interest income of $378,692 and $44,113, respectively. Therefore, a 1% change in the average interest rate for the years ended December 31, 2014 and 2013, would have a net impact on finance income of $3,787 and $441, respectively.
| (d) | Currency risk |
The Company is exposed to currency risk related to the fluctuation of foreign exchange rates and the degree of volatility of those rates. Currency risk is limited to the portion of the Company’s business transactions denominated in currencies other than the Canadian dollar which are primarily expenses in US dollars. As at December 31, 2014 and 2013, the Company held US dollar cash in the amount of US$142,558 and US$493,031 and had US dollar denominated accounts payable and accrued liabilities in the amount of US$1,910,430 and US$88,063, respectively. Therefore, a 1% change in the foreign exchange rate would have a net impact on finance costs as at December 31, 2014 and 2013 of $17,679 and $4,050, respectively.
US dollar expenses for the years ended December 31, 2014 and 2013 were approximately US$3,260,000 and US$518,000, respectively. Varying the US exchange rate for the years ended December 31, 2014 and 2013 to reflect a 5% strengthening of the Canadian dollar would have decreased the net loss by approximately $163,000 and $26,000, respectively, assuming that all other variables remained constant.
F-32
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
CONSOLIDATED FINANCIAL STATEMENTS
FOR THE YEARS ENDED
DECEMBER 31, 2013 AND 2012
96 Skyway Avenue
Toronto, Ontario M9W 4Y9
www.trilliumtherapeutics.com
F-33
Table of Contents
INDEPENDENT AUDITORS’ REPORT OF REGISTERED PUBLIC ACCOUNTING FIRM
To the Directors of
Trillium Therapeutics Inc.
We have audited the accompanying consolidated financial statements ofTrillium Therapeutics Inc. (formerly Stem Cell Therapeutics Corp.), which comprise the consolidated statements of financial position as at December 31, 2013 and 2012, and the consolidated statements of loss and comprehensive loss, changes in equity and cash flows for the years then ended, and a summary of significant accounting policies and other explanatory information.
Management’s responsibility for the consolidated financial statements
Management is responsible for the preparation and fair presentation of these consolidated financial statements in accordance with International Financial Reporting Standards, as issued by the International Accounting Standards Board, and for such internal control as management determines is necessary to enable the preparation of consolidated financial statements that are free from material misstatement, whether due to fraud or error.
Auditors’ responsibility
Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We conducted our audits in accordance with Canadian generally accepted auditing standards and the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we comply with ethical requirements and plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free from material misstatement. We were not engaged to perform an audit of the Company’s internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
An audit involves performing procedures to obtain audit evidence about the amounts and disclosures in the consolidated financial statements. The procedures selected depend on the auditors’ judgment, including the assessment of the risks of material misstatement of the consolidated financial statements, whether due to fraud or error. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the consolidated financial statements, evaluating the appropriateness of accounting policies used and the reasonableness of accounting estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements.
We believe that the audit evidence we have obtained in our audits is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position ofTrillium Therapeutics Inc. as at December 31, 2013 and 2012 and its financial performance and its cash flows for the years then ended in accordance with International Financial Reporting Standards, as issued by the International Accounting Standards Board.
| /s/ Ernst & Young LLP | ||
Toronto, Canada | Chartered Accountants | |
March 27, 2014 | Licensed Public Accountants |
F-34
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Consolidated Statements of Financial Position
Amounts in Canadian Dollars
| Note | As at December 31, 2013 $ | As at December 31, 2012 $ | ||||||||
ASSETS | ||||||||||
Current | ||||||||||
Cash | 32,456,506 | 1,375,015 | ||||||||
Marketable securities | 526,598 | - | ||||||||
Amounts receivable | 5 | 427,234 | 161,208 | |||||||
Prepaid expenses | 94,569 | 29,005 | ||||||||
Total current assets | 33,504,907 | 1,565,228 | ||||||||
Property and equipment | 6 | 109,007 | - | |||||||
Intangible assets | 7 | 1,473,472 | 2,500 | |||||||
Total non-current assets | 1,582,479 | 2,500 | ||||||||
Total assets | 35,087,386 | 1,567,728 | ||||||||
LIABILITIES | ||||||||||
Current | ||||||||||
Accounts payable and accrued liabilities | 8 | 669,860 | 185,258 | |||||||
Other current liabilities | 9 | 62,766 | - | |||||||
Total current liabilities | 732,626 | 185,258 | ||||||||
Loan payable | 9 | 341,884 | - | |||||||
Long-term liability | 9 | 104,429 | - | |||||||
Total non-current liabilities | 446,313 | - | ||||||||
Total liabilities | 1,178,939 | 185,258 | ||||||||
EQUITY | ||||||||||
Common shares | 10 | 47,191,303 | 31,388,959 | |||||||
Preferred shares | 10 | 11,292,525 | - | |||||||
Warrants | 10 | 9,818,179 | 508,281 | |||||||
Contributed surplus | 10 | 3,280,656 | 2,870,138 | |||||||
Deficit | (37,674,216 | ) | (33,384,908 | ) | ||||||
Total equity | 33,908,447 | 1,382,470 | ||||||||
Total liabilities and equity | 35,087,386 | 1,567,728 | ||||||||
Commitments and contingencies [note 15]
Approved by the Board and authorized for issue on March 27, 2014:
(signed) Henry Friesen, Director (signed) Calvin Stiller, Director
See accompanying notes to the consolidated financial statements
F-35
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Consolidated Statements of Loss and Comprehensive Loss
Amounts in Canadian Dollars
| Note | Year ended December 31, 2013 | Year ended December 31, 2012 | ||||||||||||||||
| $ | $ | |||||||||||||||||
EXPENSES | ||||||||||||||||||
Research and development | 12 | 3,336,706 | 623,548 | |||||||||||||||
General and administrative | 13 | 962,200 | 466,287 | |||||||||||||||
Operating expenses | 4,298,906 | 1,089,835 | ||||||||||||||||
Finance income | 14 | (54,028 | ) | (30,730 | ) | |||||||||||||
Finance costs | 14 | 44,430 | 2,397 | |||||||||||||||
Net finance income | (9,598 | ) | (28,333 | ) | ||||||||||||||
Net loss and comprehensive loss for the year | 4,289,308 | 1,061,502 | ||||||||||||||||
Basic and diluted loss per common share | 10 | (0.11 | ) | (0.06 | ) | |||||||||||||
See accompanying notes to the consolidated financial statements
F-36
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Consolidated Statements of Changes in Equity
Amounts in Canadian Dollars
| Common shares | Preferred shares | Warrants | Contributed | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number | Amount | Number | Amount | Number | Amount | surplus | Deficit | Total | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| # | $ | # | $ | # | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| (note 10 | ) | (note 10 | ) | (note 10 | ) | (note 10 | ) | |||||||||||||||||||||||||||||||||||||||||||||||||||||
Balance, December 31, 2012 | 18,661,936 | 31,388,959 | - | - | 1,036,364 | 508,281 | 2,870,138 | (33,384,908 | ) | 1,382,470 | ||||||||||||||||||||||||||||||||||||||||||||||||||
Net loss and comprehensive loss for the year | - | - | - | - | - | - | - | (4,289,308 | ) | (4,289,308 | ) | |||||||||||||||||||||||||||||||||||||||||||||||||
Transactions with owners of the Company, recognized directly in equity | - | - | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Share units issued, net of issue costs | 91,982,693 | 13,580,794 | 77,895,165 | 11,292,525 | 130,592,142 | 8,455,762 | - | - | 33,329,081 | |||||||||||||||||||||||||||||||||||||||||||||||||||
Compensation warrants | - | - | - | - | 5,828,890 | 700,772 | - | - | 700,772 | |||||||||||||||||||||||||||||||||||||||||||||||||||
To acquire Trillium Privateco (notes 4 and 10) | 6,079,180 | 1,215,836 | - | - | 3,300,000 | 165,000 | - | - | 1,380,836 | |||||||||||||||||||||||||||||||||||||||||||||||||||
To acquire technology rights (notes 7 and 10) | 5,028,571 | 1,005,714 | - | - | 1,600,000 | 80,000 | - | - | 1,085,714 | |||||||||||||||||||||||||||||||||||||||||||||||||||
Expiry of warrants | - | - | - | - | (127,273 | ) | (91,636 | ) | 91,636 | - | - | |||||||||||||||||||||||||||||||||||||||||||||||||
Share-based compensation | - | - | - | - | - | - | 318,882 | - | 318,882 | |||||||||||||||||||||||||||||||||||||||||||||||||||
Total transactions with owners of the Company | 103,090,444 | 15,802,344 | 77,895,165 | 11,292,525 | 141,193,759 | 9,309,898 | 410,518 | 36,815,285 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
Balance, December 31, 2013 | 121,752,380 | 47,191,303 | 77,895,165 | 11,292,525 | 142,230,123 | 9,818,179 | 3,280,656 | (37,674,216 | ) | 33,908,447 | ||||||||||||||||||||||||||||||||||||||||||||||||||
F-37
Table of Contents
| Common Shares | Preffered Share | Warrants | Contributed | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number | Amount | Number | Surplus | Number | Amount | Surplus | Deficit | Total | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| # | $ | # | $ | # | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||||
Balance, December 31, 2011 | 18,661,936 | 31,388,959 | - | - | 1,036,364 | 508,281 | 2,835,632 | (32,323,406 | ) | 2,409,466 | ||||||||||||||||||||||||||||||||||||||||||||||||||
Net loss and comprehensive loss for the year | - | - | - | - | - | - | (1,061,502 | ) | (1,061,502 | ) | ||||||||||||||||||||||||||||||||||||||||||||||||||
Transactions with owners of the Company, recognized directly in equity | - | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Share-based compensation | - | - | - | - | - | - | 34,506 | - | 34,506 | |||||||||||||||||||||||||||||||||||||||||||||||||||
Total transactions with owners of the Company | - | - | - | - | - | - | 34,506 | - | 34,506 | |||||||||||||||||||||||||||||||||||||||||||||||||||
Balance, December 31, 2012 | 18,661,936 | 31,388,959 | - | 1,036,364 | 508,281 | - | 2,870,138 | (33,384,908 | ) | 1,382,470 | ||||||||||||||||||||||||||||||||||||||||||||||||||
See accompanying notes to the consolidated financial statements
F-38
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Consolidated Statements of Cash Flows
Amounts in Canadian Dollars
Note
| Year ended December 31, 2013 $ | Year ended December 31, 2012 $ | ||||||||||
OPERATING ACTIVITIES | ||||||||||||
Net loss for the year | (4,289,308 | ) | (1,061,502 | ) | ||||||||
Adjustments for items not affecting cash | ||||||||||||
Share-based compensation | 10 | 318,882 | 34,506 | |||||||||
Interest accretion | 9 | 40,133 | - | |||||||||
Amortization of intangible assets | 7 | 632,779 | 2,000 | |||||||||
Depreciation of property and equipment | 6 | 16,010 | 4,382 | |||||||||
| (3,281,504 | ) | (1,020,614 | ) | |||||||||
Changes in non-cash working capital balances | ||||||||||||
Amounts receivable | 554,918 | (119,594 | ) | |||||||||
Prepaid expenses | (21,264 | ) | (12,685 | ) | ||||||||
Other current liabilities | 62,766 | - | ||||||||||
Accounts payable and accrued liabilities | 202,424 | (168,856 | ) | |||||||||
Cash used in operating activities | (2,482,660 | ) | (1,321,749 | ) | ||||||||
FINANCING ACTIVITIES | ||||||||||||
Increase in loan payable | 9 | 13,284 | - | |||||||||
Increase in long-term liability | 9 | 98,915 | - | |||||||||
Issue of share capital, net of issuance costs | 10 | 34,029,853 | - | |||||||||
Cash provided by financing activities | 34,142,052 | - | ||||||||||
INVESTING ACTIVITIES | ||||||||||||
Purchase of property and equipment | 6 | (34,017 | ) | - | ||||||||
Net change in marketable securities | 104,112 | - | ||||||||||
Acquisition of Trillium Privateco , net of cash acquired | 4 | (647,996 | ) | - | ||||||||
Cash used in investing activities | (577,901 | ) | - | |||||||||
Net increase (decrease) in cash during the year | 31,081,491 | (1,321,749 | ) | |||||||||
Cash, beginning of year | 1,375,015 | 2,696,764 | ||||||||||
Cash, end of year | 32,456,506 | 1,375,015 | ||||||||||
Supplemental cash flow information | ||||||||||||
Common share purchase warrants issued as agent’s compensation (note 10) | 700,772 | - | ||||||||||
Common shares and warrants issued on acquisition of Trillium Privateco (note 10) | 1,380,836 | - | ||||||||||
Common shares and warrants issued on acquisition of technology rights (note 10) | 1,085,714 | - | ||||||||||
See accompanying notes to the consolidated financial statements
F-39
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 1. | Corporate information |
Trillium Therapeutics Inc. (formerly Stem Cell Therapeutics Corp.) (the “Company” or “Trillium”) is a Canadian public immuno-oncology company advancing cancer stem cell discoveries into novel and innovative cancer therapies.
The Company was incorporated under the laws of the Province of Alberta on March 31, 2004 with nominal share capital. On October 19, 2004, the Company changed its name from Neurogenesis Biotech Corp. to Stem Cell Therapeutics Corp. On November 7, 2013, the Company filed Articles of Continuance to change its jurisdiction to Ontario. On June 1, 2014, the Company amalgamated with its wholly-owned subsidiary Trillium Therapeutics Inc. (“Trillium Privateco”) and changed its name to Trillium Therapeutics Inc.
The Company’s head office is located at 96 Skyway Avenue, Toronto, Ontario, M9W 4Y9 and is listed on the TSX Venture Exchange under the symbol SSS.
| 2. | Basis of presentation |
| (a) | Statement of compliance |
The consolidated financial statements have been prepared in accordance with International Financial Reporting Standards (“IFRS”), as issued by the International Accounting Standards Board (“IASB”).
The policies applied in these consolidated financial statements are based on IFRS issued and in effect as of March 27, 2014, the date the Board of Directors approved the consolidated financial statements.
| (b) | Basis of measurement |
These consolidated financial statements have been prepared on the historical cost basis, except for held-for-trading financial assets which are measured at fair value.
| (c) | Functional and presentation currency |
These consolidated financial statements are presented in Canadian dollars, which is the Company’s functional currency.
| (d) | Use of significant estimates and assumptions |
The preparation of financial statements in conformity with IFRS requires management to make judgments, estimates and assumptions that affect the application of accounting policies and the reported amounts of assets and liabilities, revenue and expenses and the related disclosures of contingent assets and liabilities and the determination of the Company’s ability to continue as a going concern. Actual results could differ materially from these estimates and assumptions. The Company reviews its estimates and underlying assumptions on an ongoing basis. Revisions are recognized in the period in which the estimates are revised and may impact future periods.
F-40
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 2. | Basis of presentation (continued) |
Management has applied significant estimates and assumptions to:
Valuation of share-based compensation and warrants
Management measures the costs for share-based compensation and warrants using market-based option valuation techniques. Assumptions are made and estimates are used in applying the valuation techniques. These include estimating the future volatility of the share price, expected dividend yield, expected risk-free interest rate, future employee turnover rates, future exercise behaviours and corporate performance. Such estimates and assumptions are inherently uncertain. Changes in these assumptions affect the fair value estimates of share-based payments and warrants.
Impairment of long lived assets
Long-lived assets are reviewed for impairment upon the occurrence of events or changes in circumstances indicating that the carrying value of the asset may not be recoverable. For the purpose of measuring recoverable amounts, assets are grouped at the lowest levels for which there are separately identifiable cash flows (cash-generating units). The recoverable amount is the higher of an asset’s fair value less costs to sell and value in use (being the present value of the expected future cash flows of the relevant asset or cash-generating unit.) An impairment loss is recognized for the amount by which the asset’s carrying amount exceeds its recoverable amount. We evaluate impairment losses for potential reversals when events or circumstances warrant such consideration.
Intangible assets
The Company estimates the useful lives of intangible assets from the date they are available for use in the manner intended by management and periodically reviews the useful lives to reflect management’s intent about developing and commercializing the assets.
| 3. | Significant accounting policies |
The accounting policies set out below have been applied consistently to all periods presented in these consolidated financial statements.
| (a) | Basis of consolidation |
These consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries, Stem Cell Therapeutics Inc, and Trillium Privateco (“Trillium”) from April 9, 2013, the date of acquisition (see note 4).
Investments in other entities where the Company is exposed, or has rights, to variable returns from its involvement with the investee and has the ability to affect those returns through its power over the investee, are considered subsidiaries due to the control exercised over the investee by the Company. Subsidiaries are fully consolidated from the date at which control is determined to have occurred and are de-consolidated from the date that the Company no longer controls the entity.
Intercompany transactions, balances and unrealized gains and losses on transactions between subsidiaries are eliminated.
F-41
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
The financial statements of the subsidiaries are prepared for the same reporting period as the Company, using consistent accounting policies.
| (b) | Foreign currency |
Transactions in foreign currencies are translated to the functional currency at the rate on the date of the transactions. Monetary assets and liabilities denominated in foreign currencies are retranslated at the spot rate of exchange as at the reporting date. All differences are taken to profit or loss. Non-monetary items that are measured in terms of historical cost in a foreign currency are translated using the exchange rate as at the date of the initial transaction. Non-monetary items measured at fair value in a foreign currency are translated using the exchange rate at the date when the fair value was determined.
The functional currency of each entity is separately determined. The assets and liabilities of each entity are translated at the rate of exchange in effect as at the year-end while revenue and expense items, including depreciation and amortization, are translated at the rates of exchange prevailing at the dates of the transactions. Foreign exchange gains and losses from the translation of such financial statements are deferred and recognized in other comprehensive income. On disposal of a foreign operation, the foreign currency translation reserve relating to that foreign operation is taken to profit or loss.
Financial instruments
| (c) | Financial assets |
A financial asset is classified at fair value through profit or loss if it is held for trading or is designated as such upon initial recognition. Attributable transaction costs are recognized in profit or loss as incurred. Financial assets at fair value through profit or loss are measured at fair value and changes therein are recognized in profit or loss.
Marketable securities
Marketable securities are comprised of short-term debt instruments with an original maturity of less than 90 days on purchase.
Loans and receivables
Loans and receivables are non-derivative financial assets with fixed or determinable payments that are not quoted in an active market. Loans and receivables are initially recognized at fair value plus transaction costs and subsequently measured at amortized cost using the effective interest rate method less any impairment losses. The Company has classified its amounts receivable as loans and receivables.
Derecognition
A financial asset is derecognized when the rights to receive cash flows from the asset have expired or when the Company has transferred its rights to receive cash flows from the asset.
F-42
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
Financial liabilities
Financial liabilities are recognized initially at fair value plus any directly attributable transaction costs, and subsequently at amortized cost using the effective interest method. The Company has classified its accounts payable and accrued liabilities and loan payable as financial liabilities.
Derecognition
A financial liability is derecognized when its contractual obligations are discharged or cancelled or expire.
Equity
Common shares, preferred shares and warrants to purchase common shares are classified as equity. Incremental costs directly attributable to the issue of common shares, preferred shares and warrants are recognized as a deduction from equity, net of any tax effects.
| (d) | Property and equipment |
Recognition and measurement
Items of property and equipment are measured at cost less accumulated depreciation and accumulated impairment losses. Cost includes the expenditure that is directly attributable to the acquisition of the asset. When parts of an item of property and equipment have different useful lives, they are accounted for as separate items (major components) of property and equipment. Gains and losses on disposal of an item of property and equipment are determined by comparing the proceeds from disposal with the carrying amount of property and equipment, and are recognized in profit or loss.
Subsequent costs
The cost of replacing a part of an item of property and equipment is recognized in the carrying amount of the item if it is probable that the future economic benefits embodied within the part will flow to the Company, and its cost can be measured reliably. The carrying amount of the replaced part is then derecognized. The costs of the day-to-day servicing of property and equipment are recognized in profit or loss as incurred.
F-43
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
Depreciation
The estimated useful lives and the methods of depreciation for the current and comparative periods are as follows:
| Asset | Basis | |
Lab equipment | 20% declining balance | |
Computer equipment | Straight-line over 3 years | |
Office equipment | Straight-line over 5 years | |
Leaseholds | Straight-line over lease term | |
Estimates for depreciation methods, useful lives and residual values are reviewed at each reporting period-end and adjusted if appropriate. Depreciation expense is recognized in research and development expenses.
| (e) | Intangible assets |
Research and development
Expenditures on research activities, undertaken with the prospect of gaining new scientific or technical knowledge and understanding, is recognized in profit or loss as incurred.
Development activities involve a plan or design for the production of new or substantially improved products and processes. Development expenditures are capitalized only if development costs can be measured reliably, the product or process is technically and commercially feasible, future economic benefits are probable, and the Company intends to complete development and has sufficient resources to complete development and to use or sell the asset. Other development expenditures are expensed as incurred. No internal development costs have been capitalized to date.
Research and development expenses include all direct and indirect operating expenses supporting the products in development. The costs incurred in establishing and maintaining patents are expensed as incurred.
Intangible assets
Intangible assets that are acquired separately and have finite useful lives are measured at cost less accumulated amortization and accumulated impairment losses. Subsequent expenditures are capitalized only when they increase the future economic benefits embodied in the specific asset to which it relates. All other expenditure is recognized in profit or loss as incurred.
Amortization is recognized in profit or loss on a straight-line basis over the estimated useful lives of intangible assets from the date they are available for use in the manner intended by management. The
F-44
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
period that the technologies acquired in the Trillium Privateco acquisition are available for use is estimated at three years, and the period that the technologies acquired from the University Health Network (“UHN”) are available for use is estimated at two years, which reflects management’s intent about developing and commercializing the assets.
The amortization method and amortization period of an intangible asset with a finite life is reviewed at least annually. Changes in the expected useful life or the expected pattern of consumption of future economic benefits embodied in the asset are accounted for by changing the amortization period or method, as appropriate, and are treated as changes in accounting estimates. The amortization expense on intangible assets with finite lives is recognized in research and development expenses.
| (f) | Impairment |
Financial assets
A financial asset not carried at fair value through profit or loss is assessed at each reporting date to determine whether there is objective evidence that it is impaired. A financial asset is impaired if objective evidence indicates that a loss event has occurred after the initial recognition of the asset, and that the loss event had a negative effect on the estimated future cash flows of that asset that can be estimated reliably.
An impairment test is performed, on an individual basis, for each material financial asset. Other individually non-material financial assets are tested as groups of financial assets with similar risk characteristics. Impairment losses are recognized in profit or loss.
An impairment loss in respect of a financial asset measured at amortized cost is calculated as the difference between its carrying amount and the present value of the estimated future cash flows discounted at the asset’s original effective interest rate. Losses are recognized in net profit or loss and reflected in an allowance account against the respective financial asset. Interest on the impaired asset continues to be recognized through the unwinding of the discount. When a subsequent event causes the amount of impairment loss to decrease, the decrease in impairment loss is reversed through profit or loss.
Non-financial assets
The carrying amounts of the Company’s non-financial assets are reviewed at each reporting date to determine whether there is any indication of impairment. If such an indication exists, the recoverable amount is estimated.
The recoverable amount of an asset or a cash-generating unit is the greater of its value in use and its fair value less costs to sell. In assessing value in use, the estimated future cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset or cash-generating unit. For the purpose of impairment testing, assets that cannot be tested individually are grouped together into the smallest group of assets that generates cash inflows from continuing use that are largely independent of cash inflows of other assets or cash-generating units. An impairment loss is recognized if the carrying amount of an asset or its related
F-45
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
cash-generating unit exceeds its estimated recoverable amount. Impairment losses for intangible assets are recognized in research and development expenses.
Impairment losses recognized in prior periods are assessed at each reporting date for any indications that the loss has decreased or no longer exists. An impairment loss is reversed if there has been a change in the estimates used to determine the recoverable amount. An impairment loss is reversed only to the extent that the asset’s carrying amount does not exceed the carrying amount that would have been determined, net of depreciation or amortization, if no impairment loss had been recognized.
| (g) | Provisions |
A provision is recognized if, as a result of a past event, the Company has a present legal or constructive obligation that can be estimated reliably, and it is probable that an outflow of economic benefits will be required to settle the obligation. Provisions are assessed by discounting the expected future cash flows at a pre-tax rate that reflects current market assessments of the time value of money and the risks specific to the liability. The unwinding of the discount on provisions is recognized in finance costs. No provisions have been recognized.
| (h) | Government assistance |
Government assistance relating to research and development is recorded as a reduction of expenses when the related expenditures are incurred.
| (i) | Share-based compensation |
The grant-date fair value of share-based payment awards granted to employees is recognized as personnel costs, with a corresponding increase in contributed surplus, over the period that the employees unconditionally become entitled to the awards. The amount recognized as an expense is adjusted to reflect the number of awards for which the related service and non-market vesting conditions are expected to be met, such that the amount ultimately recognized as an expense is based on the number of awards that met the related service and non-market performance conditions at the vesting date.
For equity-settled share-based payment transactions, the Company measures the goods or services received, and the corresponding increase in contributed surplus, directly, at the fair value of the goods or services received, unless that fair value cannot be estimated reliably. If the Company cannot estimate reliably the fair value of the goods or services received, it measures their value by reference to the fair value of the equity instruments granted. Transactions measured by reference to the fair value of the equity instruments granted have their fair values remeasured at each vesting and reporting date until fully vested.
| (j) | Income taxes |
Deferred tax is recognized in respect of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for taxation purposes. Deferred tax is not recognized for temporary differences on the initial recognition of assets or liabilities in a transaction that is not a business combination and that affects neither accounting nor taxable income or loss.
F-46
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
Deferred tax assets and liabilities are offset if there is a legally enforceable right to offset current tax assets and liabilities, and they relate to income taxes levied by the same tax authority on the same taxable entity.
Deferred tax is measured at the tax rates that are expected to be applied to temporary differences whey they reverse, based on the laws that have been enacted or substantively enacted at the reporting date. A deferred tax asset is recognized for unused tax losses, tax credits and deductible temporary differences, to the extent that it is probable that future taxable profits will be available against which they can be utilized.
Investment tax credits earned from scientific research and development expenditures are recorded when collectability is reasonably assured.
| (k) | Loss per share |
Basic loss per share is computed by dividing the net loss available to common shareholders by the weighted average number of shares outstanding during the reporting period. Diluted loss per share is computed similar to basic loss per share except that the weighted average number of shares outstanding are increased to include additional shares for the assumed exercise of stock options and warrants and conversion of preferred shares, if dilutive. The number of additional shares is calculated by assuming that outstanding preferred shares would convert to common shares and that outstanding stock options and warrants were exercised and that the proceeds from such exercises were used to acquire common stock at the average market price during the reporting period. The inclusion of the Company’s stock options, warrants and preferred shares in the computation of diluted loss per share has an anti-dilutive effect on the loss per share and therefore, they have been excluded from the calculation of diluted loss per share.
| (l) | New standards and interpretations adopted during 2013 |
IFRS 10Consolidated Financial Statements
IFRS 10 requires an entity to consolidate an investee when it is exposed, or has rights, to variable returns from its involvement with the investee and has the ability to affect those returns through its power over the investee. IFRS 10 supersedes SIC-12
Consolidations – Special Purpose Entitiesand replaces parts of IAS 27Consolidated and Separate Financial Statements. The effective date of this amendment is for annual periods beginning on or after January 1, 2013. The Company adopted IFRS 10, including the amendments issued in June 2012, in its financial statements for the annual period beginning on January 1, 2013. The adoption did not have a material impact on the consolidated financial statements.
IFRS 12Disclosure of Interests in Other Entities
IFRS 12 establishes disclosure requirements for interest in other entities such as subsidiaries, joint arrangements, associates and unconsolidated structured entities. The standard carries forward existing disclosures and also introduces significant additional disclosure requirements that address the nature of,
F-47
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
and risks associated with, an entity’s interest in other entities. IFRS 12 replaces the previous disclosure requirements included in IAS 27Consolidated and Separate Financial Statements, IAS 31Joint Venturesand IAS 28Investment in Associates. The effective date of this amendment is for annual periods beginning on or after January 1, 2013. The Company adopted IFRS 12 in its consolidated financial statements for the annual period beginning on January 1, 2013. The adoption did not have a material impact on the consolidated financial statements.
IFRS 13Fair Value Measurement
IFRS 13 is a comprehensive standard for fair value measurement and disclosure requirements for use across all IFRS standards. IFRS 13 defines fair value and establishes disclosures about fair value measurement. The effective date of this amendment is for annual periods beginning on or after January 1, 2013. The Company adopted IFRS 13 in its consolidated financial statements for the annual period beginning on January 1, 2013. The adoption did not have a material impact on the consolidated financial statements.
IAS 1Presentation of Financial Statements
The IASB amended IAS 1 by revising how certain items are presented in other comprehensive income (“OCI”). Items within OCI that may be reclassified to income will be separated from items that will not. The standard is effective for fiscal years beginning on or after July 1, 2012 with early adoption permitted. The Company has adopted IAS 1 retrospectively and there are minimal disclosure differences.
Annual Improvements to IFRSs 2009-2011 Cycle – various standards
In May 2012, the IASB published Annual Improvements to IFRSs – 2009-2011 Cycle as part of its annual improvements process to make non-urgent but necessary amendments to IFRS. These amendments are effective for annual periods beginning on or after January 1, 2013 with retrospective application. The Company adopted the amendments to the standards in its consolidated financial statements for the annual period beginning on January 1, 2013. The adoption did not have a material impact on the consolidated financial statements.
| (m) | New standards and interpretations not yet effective |
IFRS 9 Financial Instruments
In October 2010, the IASB published amendments to IFRS 9 which provides added guidance on the classification and measurement of financial liabilities. IFRS 9 will replace IAS 39 and will be completed in three phases: classification and measurement of financial assets and liabilities, impairment of financial assets, and general hedge accounting. This was the first phase of the project on classification and measurement of financial assets and liabilities. The IASB is discussing proposed limited amendments related to this phase of the project. The standard on general hedge accounting was issued and included as part of IFRS 9 in November 2013. The accounting for macro hedging is expected to be issued as a separate standard outside of IFRS 9. The impairment of financial assets phase of the project is currently in
F-48
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 3. | Significant accounting policies (continued) |
development. In November 2013, the mandatory effective date of IFRS 9 of January 1, 2015 was removed and the effective date will be determined when the remaining phases of IFRS 9 are finalized. The Company is currently monitoring the developments of this standard and assessing the impact that the adoption of this standard may have on the consolidated financial statements.
IFRIC 21 Levies
In May 2013, International Financial Reporting Standards Interpretations Committee Interpretation 21, Levies (“IFRIC 21”) was issued. IFRIC 21 provides guidance on when to recognize a liability to pay a levy imposed by a government that is accounted for in accordance with IAS 37Provisions, Contingent Liabilities and Contingent Assets. IFRIC 21 is effective for annual periods beginning on or after January 1, 2014 and is to be applied retrospectively. The Company is currently assessing the impact the adoption of this interpretation may have on the consolidated financial statements.
IAS 36Impairment of Assets
In May 29, 2013, IASB published amendments to IAS 36, Impairment of Assets which reduce the circumstances in which the recoverable amount of cash-generating units is required to be disclosed and clarifies the disclosures required when an impairment loss has been recognized or reversed in the period. This amendment is effective for annual periods beginning on or after January 1, 2014. The Company does not expect the adoption of this amendment to have a material impact on its consolidated financial statements.
F-49
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 4. | Acquisition of Trillium Privateco |
On April 9, 2013, the Company completed a merger with Trillium Privateco, a private biopharmaceutical company specializing in immune regulation and the development of cancer stem cell-related therapeutics. The Company applied the acquisition method of accounting for the business combination and the losses have been consolidated from the acquisition date. The consideration was allocated to the underlying assets acquired and liabilities assumed based upon their fair values at the date of acquisition. The purchase price allocation was assigned to the net identifiable assets based on their estimated fair values. The final purchase price allocation is listed below:
Fair value of consideration paid: | ||||
Cash | $ | 1,200,000 | ||
2,779,180 common shares and 3,300,000 units | 1,380,836 | |||
| 2,580,836 | ||||
Assets acquired: | ||||
Cash and marketable securities | $ | 1,182,714 | ||
Amounts receivable | 820,944 | |||
Prepaid expenses | 44,300 | |||
Equipment | 91,000 | |||
Acquired technology | 1,018,037 | |||
| 3,156,995 | ||||
Liabilities assumed: | ||||
Accounts payable and accrued liabilities | 282,178 | |||
Loan payable | 293,981 | |||
| 576,159 | ||||
Net identifiable assets acquired | $ | 2,580,836 | ||
As consideration, the Company paid $1,200,000 in cash and issued 2,779,180 common shares and 3,300,000 units. Each unit consisted of one common share and one common share purchase warrant, with each common share purchase warrant allowing its holder to acquire one additional common share at an exercise price of $0.40 until March 15, 2018. The fair value of the unit was determined based on the stand-alone value of the common share and the common share purchase warrant, in accordance with IFRS 13, Fair value measurement and IFRS 3, Business combinations. Accordingly, the fair value of the aggregate 6,079,180 common shares issued was based on the closing price of the Company’s common shares of $0.20 on April 9, 2013. The fair value of the 3,300,000 common share purchase warrants was determined using the Black-Scholes option pricing model. Assumptions used to determine the value of the common share purchase warrant were: dividend yield of 0%; risk-free interest rate of 1.1%; expected volatility of 68%; and average expected life of 3 years. Post-closing, Trillium Privateco shareholders held approximately 16% of the issued and outstanding common shares, and Trillium Privateco became a wholly-owned subsidiary of the Company.
F-50
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 4. | Acquisition of Trillium Privateco (continued) |
Cash used in the investment is determined as follows:
| $ | ||||
Cash consideration | 1,200,000 | |||
Less cash acquired | 552,004 | |||
| 647,996 | ||||
Acquisition costs incurred by the Company for the Trillium Privateco merger, and included in general and administrative expenses for the year ended December 31, 2013, were approximately $113,000. From the date of the acquisition to December 31, 2013, Trillium Privateco contributed revenue of nil and a loss of $2,236,262. If the acquisition had occurred on January 1, 2013, management estimates that consolidated revenue would have been nil, and consolidated loss for the year would have been $5,368,637. In determining these amounts, management has assumed that the fair value adjustments that arose on the date of acquisition would have been the same if the acquisition had occurred on January 1, 2013.
The acquisition gave rise to a deferred tax liability related to the acquired technology and a deferred tax asset related to the tax effect of the Trillium Privateco non-capital losses and the scientific research and experimental development tax pools. The Company has offset the tax liabilities and tax assets as it has a legally enforceable right to set them off. A net residual tax asset has not been recognized as it is not probable that future taxable profits will be available against which the accumulated tax assets can be utilized.
| 5. | Amounts receivable |
| December 31, 2013 $ | December 31, 2012 $ | |||||||
Government programs receivable | 426,946 | 83,458 | ||||||
Other amounts receivable | 288 | 77,750 | ||||||
| 427,234 | 161,208 | |||||||
F-51
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 6. | Property and equipment |
| Lab equipment $ | Computer equipment $ | Office equipment and leaseholds $ | Total $ | |||||||||||||
Cost | ||||||||||||||||
Balance, December 31, 2011 | - | 154,706 | 33,889 | 188,595 | ||||||||||||
Disposals | - | (141,206 | ) | (33,889 | ) | (175,095 | ) | |||||||||
Balance, December 31, 2012 | - | 13,500 | - | 13,500 | ||||||||||||
Additions | 111,025 | 4,611 | 9,381 | 125,017 | ||||||||||||
Balance, December 31, 2013 | 111,025 | 18,111 | 9,381 | 138,517 | ||||||||||||
Accumulated depreciation | ||||||||||||||||
Balance, December 31, 2011 | - | 150,324 | 33,889 | 184,213 | ||||||||||||
Disposals | - | (141,206 | ) | (33,889 | ) | (175,095 | ) | |||||||||
Depreciation | - | 4,382 | - | 4,382 | ||||||||||||
Balance, December 31, 2012 | - | 13,500 | - | 13,500 | ||||||||||||
Depreciation | 14,723 | 504 | 783 | 16,010 | ||||||||||||
Balance December 31, 2013 | 14,723 | 14,004 | 783 | 29,510 | ||||||||||||
Net carrying amounts | ||||||||||||||||
December 31, 2012 | - | - | - | - | ||||||||||||
December 31, 2013 | 96,302 | 4,107 | 8,598 | 109,007 | ||||||||||||
During the first quarter of 2012, the Company closed its Calgary office and retired computer and office equipment that was fully depreciated in the amount of $175,095. In the second quarter of 2013, the Company acquired lab equipment in its merger with Trillium Privateco with a fair value of $91,000.
F-52
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 7. | Intangible assets |
Total $ | ||||
Cost | ||||
Balance, December 31, 2011 and December 31, 2012 | 2,431,279 | |||
Additions | 2,103,751 | |||
Disposals | (2,431,279 | ) | ||
Balance December 31, 2013 | 2,103,751 | |||
Accumulated amortization | ||||
Balance, December 31, 2011 | 2,426,779 | |||
Amortization | 2,000 | |||
Balance, December 31, 2012 | 2,428,779 | |||
Amortization | 632,779 | |||
Disposals | (2,431,279 | ) | ||
Balance, December 31, 2013 | 630,279 | |||
Net carrying amounts | ||||
December 31, 2012 | 2,500 | |||
December 31, 2013 | 1,473,472 | |||
On April 9, 2013, the Company completed a merger with Trillium Privateco. Licensed patent rights of Trillium Privateco acquired amounted to $1,018,037 (see note 4).
On April 16, 2013, the Company signed an agreement with UHN for an exclusive world-wide license to an innovative cancer stem cell program. The initial consideration for the UHN license of $1,085,714 was satisfied by the issuance of 5,028,571 common shares and 1,600,000 common share purchase warrants, each warrant allowing its holder to acquire one additional common share at an exercise price of $0.40 until March 15, 2018. Additional consideration under the UHN license includes an annual license maintenance fee and future development milestones. The fair value of the common shares issued was based on the closing price of the Company’s common shares of $0.20 on April 16, 2013 and $0.25 per unit based on the fair value of the common share and common share purchase warrant components as of the date of acquisition.
During the third quarter of 2013, the Company ceased all activities related to the regenerative stem cell products and retired the fully amortized assets in the amount of $2,431,279.
F-53
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 8. | Accounts payable and accrued liabilities |
December 31, $ | December 31, $ | |||||||
Trade and other payables | 113,003 | 55,548 | ||||||
Accrued liabilities | 221,580 | 95,484 | ||||||
Due to related parties (note 16) | 335,277 | 34,226 | ||||||
| 669,860 | 185,258 | |||||||
| 9. | Non-current liabilities |
| (a) | On February 1, 2012, Trillium Privateco entered into a contribution agreement with the Federal Economic Development Agency for Southern Ontario where it received a repayable contribution to support the clinical development of its TTI-1612 program. The period of contribution ended on December 31, 2013. As at December 31, 2013, Trillium Privateco has repayable contributions of $575,139 representing the outstanding principal balance, repayable in equal monthly installments for 60 months beginning on December 1, 2014. The loan payable bears no interest. On acquisition of Trillium Privateco, the fair value of the loan payable was determined by discounting the loan payable using an estimated market interest rate of 15%. Interest expense will accrete on the discounted loan amount until it reaches its face value at maturity. |
| (b) | The Company incurred a discounted long-term liability of $157,609 related to the cessation of the development of its regenerative medicine products. This liability has been discounted using an estimated market interest rate of 15% and interest expense is accreting. |
The current portions of the loan payable and long-term liability are included in other current liabilities in the statements of financial position.
| 10. | Share capital |
| (a) | Authorized |
The authorized share capital of the Company consists of an unlimited number of common shares, Class B shares and First Preferred Shares, in each case without nominal or par value. Common shares are voting and may receive dividends as declared at the discretion of the Board of Directors. Class B shares are non-voting and convertible to common shares at the holder’s discretion, on a one-for-one basis. Upon dissolution or wind-up of the Company, Class B shares participate rateably with the common shares in the distribution of the Company’s assets. Preferred shares have voting rights as decided upon by the Board of Directors at the time of grant. Upon dissolution or wind-up of the Company, First Preferred Shares are entitled to priority over common and Class B shares.
During 2013, the Company created a new series of preferred shares being the Series I Non-Voting Convertible First Preferred Shares. These shares are non-voting, may receive dividends as declared at the discretion of the Board of Directors, and are convertible to common shares at the holder’s discretion, on a one-for-one basis.
F-54
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
| (b) | Common and preferred shares issued – year ended December 31, 2013 |
The Company consolidated its outstanding common shares issuing one post-consolidated share for each 10 pre-consolidated shares and the common shares began trading on a post-consolidated basis on February 6, 2013. All references in the consolidated financial statements and notes to the number of common shares, warrants and stock options have been adjusted to the post-consolidation amounts.
In March 2013, the Company completed a capital raise as part of a shelf prospectus supplement and a U.S. private placement for a total of 12,735,000 units at a price of $0.25 per unit, for aggregate gross proceeds to the Company of $3,185,080 ($2,615,240 net of issuance costs). Each unit consisted of one common share and one common share purchase warrant. Each warrant entitles the holder to purchase one common share at a price of $0.40 per share at any time prior to expiry on March 15, 2018 (12,315,000 warrants) and March 27, 2018 (420,000 warrants). In connection with the financing, the Company issued 814,050 compensation warrants having an aggregate fair value of $48,843 estimated using the Black- Scholes option pricing model. Each compensation warrant entitles the holder to acquire one common share at an exercise price of $0.25 per share prior to expiry on March 16, 2015.
The allocation of the $0.25 common share unit issue price to the common shares and unit warrants was based on the relative fair values of the common shares and the warrants. The fair value of the warrants was determined using the Black-Scholes option pricing model. The common shares were allocated a price of $0.20 per share and the warrants were allocated a price of $0.05 per warrant. The costs of the issue were allocated on a pro rata basis to the common shares and warrants. Accordingly, $2,092,192 was allocated to common shares and $523,048 to warrants, net of issuance costs. Assumptions used to determine the value of the unit warrants were: dividend yield of 0%; risk-free interest rate of 1.1%; expected volatility of 64%; and average expected life of 3 years. Assumptions used to determine the value of the compensation warrants were: dividend yield of 0%; risk-free interest rate of 1.0%; expected volatility of 80%; and average expected life of 2 years.
On April 9, 2013, the Company issued 6,079,180 common shares and 3,300,000 common share purchase warrants for partial consideration for the acquisition of Trillium Privateco (see note 4).
On April 16, 2013, the Company issued 5,028,571 common shares and 1,600,000 common share purchase warrants on the acquisition of certain cancer stem cell assets from UHN (see note 7).
On December 13, 2013, the Company completed a private placement financing issuing 79,247,693 common share units (each common share unit consisting of one common share and three-quarters of a common share purchase warrant) at a price of $0.21 per unit and 77,895,165 preferred share units (each preferred share unit consisting of one Series 1 Non-Voting First Preferred Share and three-quarters of a common share purchase warrant) at a price of $0.21 per unit for gross proceeds of $32,866,025 ($30,713,841 net of issuance costs). Each whole warrant entitles the holder to purchase one common share at a price of $0.28 at any time prior to expiry on December 13, 2018. In connection with the financing, the Company issued 5,014,839 compensation warrants having an aggregate fair value of $651,929 estimated using the Black-Scholes option pricing model. Each compensation warrant entitles the holder to acquire one common share at an exercise price of $0.21 per share prior to expiry on December 13, 2015.
F-55
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
The allocation of the $0.21 unit issue price to the common shares, Series 1 Non-Voting First Preferred Shares and unit warrants was based on the relative fair values of the common shares, Series 1 Non-Voting First Preferred Shares and the warrants. The fair value of the warrants was determined using the Black-Scholes option pricing model. The common shares were allocated a price of $0.16 per share, the Series 1 Non-Voting First Preferred Shares were allocated a price of $0.16 per share, and the warrants were allocated a price of $0.05 per three-quarter warrant. The costs of the issue were allocated on a pro rata basis. Accordingly, $11,488,602 was allocated to common shares, $11,292,525 to the Series 1 Non-Voting First Preferred Shares and $7,932,714 to warrants, net of issuance costs. Assumptions used to determine the value of the unit warrants were: dividend yield of 0%; risk-free interest rate of 1.2%; expected volatility of 70%; and average expected life of 3 years. Assumptions used to determine the value of the compensation warrants were: dividend yield of 0%; risk-free interest rate of 1.1%; expected volatility of 66%; and average expected life of 2 years.
All securities issued under the offering (including the compensation warrants), in Canada are subject to a four month hold and resale restrictions under Canadian securities law, and in the United States are subject to statutory resale restrictions under U.S. securities laws. All securities issued under the offering are also subject to a four month hold imposed under the policies of the TSX Venture Exchange.
In the December 2013 private placement, subscribers who purchased Preferred Share Units and certain subscribers who purchased Common Share Units are subject to restrictions on the conversion and exercise of securities of the Company convertible in common shares. Such subscribers cannot convert or exercise securities of the Company convertible or exerciseable into common shares if, after giving effect to the exercise of conversion, the subscriber and its joint actors would have beneficial ownership or direction or control over common shares in excess of 4.99% of the then outstanding common shares. This limit can be raised at the option of the subscriber on 61 days prior written notice: (i) up to 9.99%, (ii) up to 19.99%, subject to stock exchange clearance of a personal information form submitted by the subscriber, and (iii) above 19.99%, subject to stock exchange approval and shareholder approval.
Subject to receipt of any required regulatory approvals, subscribers who purchased a minimum of 10% of the securities sold under the offering have been given rights to purchase securities of the Company in future financings to enable each such subscriber to maintain its percentage holding in the Company for so long as the subscriber holds at least 10% of the outstanding common shares on a fully-diluted basis.
There were no common or preferred shares issued in the year ended December 31, 2012.
| (c) | Weighted average number of common shares |
The weighted average number of common shares outstanding for the purposes of calculating earnings per share have been adjusted for 2013 and 2012 to the post-consolidated number. The post-consolidated weighted average number of common shares outstanding for year ended December 31, 2013 was 40,707,868 (2012 – 18,661,936).
F-56
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
| (d) | Warrants |
Warrants outstanding at December 31, 2013 were as follows:
| Expiry dates | Number of warrants | Exercise price | ||||||
March 14, 2014 | 909,091 | $ | 1.60 | |||||
March 16, 2015 | 814,051 | $ | 0.25 | |||||
December 13, 2015 | 5,014,839 | $ | 0.21 | |||||
March 15, 2018 | 17,215,000 | $ | 0.40 | |||||
March 27, 2018 | 420,000 | $ | 0.40 | |||||
December 13, 2018 | 117,857,142 | $ | 0.28 | |||||
| 142,230,123 | ||||||||
All warrants were exercisable on issuance. Changes in the number of warrants outstanding during the years ended December 31 were as follows:
| 2013 | 2012 | |||||||||||||||
| Number of warrants | Weighted average exercise price | Number of warrants | Weighted average exercise price | |||||||||||||
Balance, beginning of year | 1,036,364 | $ | 1.54 | 1,036,364 | $ | 1.54 | ||||||||||
Issued | 141,321,032 | 0.29 | - | - | ||||||||||||
Expired | (127,273 | ) | 1.10 | - | - | |||||||||||
Balance, end of year | 142,230,123 | $ | 0.30 | 1,036,364 | $ | 1.54 | ||||||||||
| (e) | Stock option plan |
The Company has a 10% rolling stock option plan (the “Plan”) that was approved by the Company’s shareholders at its annual general meeting held on October 17, 2013. Pursuant to the Plan, the Company may grant stock options to purchase up to an aggregate of 10% of the Company’s issued and outstanding share capital. As at December 31, 2013, the Company was entitled to issue an additional 9,254,141 stock options under the Plan.
F-57
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
Changes in the number of options outstanding during the years ended December 31 were as follows:
| 2013 | 2012 | |||||||||||||||
| Number of options | Weighted average exercise price | Number of options | Weighted average exercise price | |||||||||||||
Balance, beginning of year | 700,000 | $ | 1.03 | 1,110,194 | $ | 1.39 | ||||||||||
Granted | 2,623,597 | 0.25 | 10,000 | 1.00 | ||||||||||||
Cancelled/forfeited | (402,500 | ) | 1.02 | (420,194 | ) | 1.98 | ||||||||||
Balance, end of year | 2,921,097 | $ | 0.33 | 700,000 | $ | 1.03 | ||||||||||
Options exercisable, end of year | 1,189,918 | $ | 0.45 | 596,806 | $ | 1.04 | ||||||||||
The following table reflects stock options outstanding at December 31, 2013:
| Stock options outstanding | Stock options exercisable | |||||||||||||||||||
| Exercise prices | Number outstanding | Weighted average remaining contractual life | Weighted average exercise price | Exercisable number | Weighted average exercise price | |||||||||||||||
$0.25 | 2,623,597 | 9.2 years | $ | 0.25 | 895,196 | $ | 0.25 | |||||||||||||
$1.00 | 274,500 | 2.3 years | $ | 1.00 | 271,722 | $ | 1.00 | |||||||||||||
$1.20 | 3,000 | 0.7 years | $ | 1.20 | 3,000 | $ | 1.20 | |||||||||||||
$1.70 | 20,000 | 0.9 years | $ | 1.70 | 20,000 | $ | 1.70 | |||||||||||||
| 2,921,097 | 8.5 years | $ | 0.33 | 1,189,918 | $ | 0.45 | ||||||||||||||
Share-based compensation expense was determined based on the fair value of the options at the date of measurement using the Black-Scholes option pricing model with the weighted average assumptions for the years ended December 31 as follows:
| 2013 | 2012 | |||||||
Expected option life | 10 years | 5 years | ||||||
Risk-free interest rate | 1.7% | 1.1% | ||||||
Forfeiture rate | 11% | 10% | ||||||
Dividend yield | 0% | 0% | ||||||
Expected volatility | 118% | 137% | ||||||
F-58
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
The Black-Scholes option pricing model was developed to estimate the fair value of freely tradable, fully transferable options without vesting restrictions, which significantly differs from the Company’s stock option awards. This model also requires highly subjective assumptions, including future stock price volatility and average option life, which significantly affect the calculated values.
The risk-free interest rate is based on the implied yield on a Government of Canada zero-coupon issue with a remaining term equal to the expected term of the option. The volatility is based solely on historical volatility equal to the expected life of the option. The life of the options is estimated considering the vesting period at the grant date, the life of the option and the average length of time similar grants have remained outstanding in the past. The forfeiture rate is an estimate based on historical evidence and future expectations. The dividend yield was excluded from the calculation since it is the present policy of the Company to retain all earnings to finance operations and future growth.
During the year ended December 31, 2013, the Company issued 2,623,597 (2012 – 10,000) stock options with a fair value of $535,781 (2012 – $2,300). The weighted average grant date fair value of the stock options granted during the year ended December 31, 2013 was $0.20 (2012 - $0.02) .
| (f) | Deferred Share Units Plan |
The shareholders of the Company approved the adoption of a deferred share units plan (the “DSU Plan”) on October 17, 2013 reserving for issuance up to 2,000,000 common shares under the DSU Plan. The purpose of the DSU Plan is to provide an alternative form of compensation to satisfy director fees and annual and special bonuses payable to senior officers and directors of the Company. No units have been issued to date.
| (g) | Shareholder Rights Plan |
The shareholders of the Company approved the adoption of a shareholder rights plan agreement (the “SRP Plan”) on October 17, 2013. The SRP Plan is designed to provide adequate time for the Board of Directors and the shareholders to assess an unsolicited takeover bid for the Company, to provide the Board of Directors with sufficient time to explore and develop alternatives for maximizing shareholder value if a takeover bid is made, and to provide shareholders with an equal opportunity to participate in a takeover bid and receive full and fair value for their common shares. The SRP Plan will expire at the close of the Company’s annual meeting of shareholders in 2016.
The rights issued under the SRP Plan initially attach to and trade with the common shares and no separate certificates will be issued unless an event triggering these rights occurs. The rights will become exercisable only when a person, including any party related to it, acquires or attempts to acquire 20 percent or more of the outstanding common shares without complying with the “Permitted Bid” provisions of the SRP Plan or without approval of the Board of Directors. Should such an acquisition occur or be announced, each right would, upon exercise, entitle a rights holder, other than the acquiring person and related persons, to purchase common shares at a approximate 50 percent discount to the market price at the time.
F-59
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 10. | Share capital (continued) |
Under the SRP Plan, a Permitted Bid is a bid made to all holders of the common shares and which is open for acceptance for not less than 60 days. If at the end of 60 days at least 50 percent of the outstanding common shares, other than those owned by the offeror and certain related parties have been tendered, the offeror may take up and pay for the common shares but must extend the bid for a further 10 days to allow other shareholders to tender. The issuance of common shares upon the exercise of the rights is subject to receipt of certain regulatory approvals.
| 11. | Income taxes |
The Company recognized no income taxes in the consolidated statements of loss and comprehensive loss, as it has been incurring losses since inception, and it is not probable that future taxable profits will be available against which the accumulated tax losses can be utilized.
| (a) | Unrecognized deferred tax assets: |
As at December 31, 2013 and 2012, deferred tax assets have not been recognized with respect to the following items:
| 2013 | 2012 | |||||||
Non-capital losses carried forward | $ | 8,515,351 | $ | 6,130,941 | ||||
Federal investment tax credits carryforward | 1,515,626 | 947,496 | ||||||
Carrying value of property and equipment and intangible assets in excess of accounting basis | 1,138,196 | 961,313 | ||||||
Scientific research and experimental development expenditures | 4,101,322 | 1,100,271 | ||||||
Share issue costs and other | 540,886 | 53,975 | ||||||
| $ | 15,811,381 | $ | 9,193,996 | |||||
| (b) | As at December 31, 2013, the Company has available research and development expenditures of approximately $15,477,000 (2012 – $4,152,000), for income tax purposes which may be carried forward indefinitely to reduce future years’ taxable income. As at December 31, 2013, the Company also has unclaimed Canadian federal scientific research and development investment tax credits of $2,062,000 (2012 – $1,287,000) which are available to reduce future federal taxes payable with expiries from 2014 through 2033. The benefit of these expenditures and investment tax credits has not been recorded in the accounts. |
| (c) | As at December 31, 2013, the Company has accumulated non-capital losses for federal and provincial income tax purposes in Canada which are available for application against future taxable income. The benefit of these losses has not been recorded in the accounts. |
F-60
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 11. | Income taxes (continued) |
The non-capital tax losses expire as follows:
| Federal | ||||
| $ | ||||
2014 | 971,000 | |||
2015 | 2,968,000 | |||
2026 | 3,320,000 | |||
2027 | 6,645,000 | |||
2028 | 4,659,000 | |||
2029 | 4,144,000 | |||
2030 | 3,736,000 | |||
2031 | 1,819,000 | |||
2032 | 1,387,000 | |||
2033 | 2,450,000 | |||
| 32,099,000 | ||||
| (d) | The reconciliation of the Canadian statutory income tax rate applied to the net loss for the year to the income tax recovery is as follows: |
| 2013 | 2012 | |||||||
Statutory income tax rate | 26.5% | 26.5% | ||||||
Income tax recovery based on statutory income tax rate | $ | (1,136,667 | ) | $ | (281,298 | ) | ||
Change in income tax rates | - | (431,758 | ) | |||||
Investment tax credits | (207,850 | ) | (23,188 | ) | ||||
Share-based compensation and other | 84,902 | 9,429 | ||||||
Change in valuation allowance | 1,259,615 | 726,815 | ||||||
Income tax recovery | $ | - | $ | - | ||||
F-61
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 12. | Research and development |
Components of research and development expenses for the years ended December 31 were as follows:
| 2013 | 2012 | |||||||
| $ | $ | |||||||
Research and development, excluding the below | 1,411,264 | 518,030 | ||||||
Salaries, fees and short-term benefits | 1,299,055 | 209,588 | ||||||
Share-based compensation | 211,419 | 38,011 | ||||||
Amortization of intangible assets | 632,779 | 2,000 | ||||||
Depreciation of property and equipment | 16,010 | 4,382 | ||||||
Net proceeds from patent interference settlement | - | (99,551 | ) | |||||
Investment tax credits | (233,821 | ) | (48,912 | ) | ||||
| 3,336,706 | 623,548 | |||||||
| 13. | General and Administrative |
Components of general and administrative expenses for the years ended December 31 were as follows:
| 2013 | 2012 | |||||||
| $ | $ | |||||||
General and administrative, excluding the below | 599,785 | 269,849 | ||||||
Salaries, fees and short-term benefits | 254,952 | 199,943 | ||||||
Share-based compensation | 107,463 | (3,505 | ) | |||||
| 962,200 | 466,287 | |||||||
| 14. | Finance income and finance costs |
Finance income for the years ended December 31 was as follows:
| 2013 | 2012 | |||||||
| $ | $ | |||||||
Interest income | 44,113 | 25,740 | ||||||
Net foreign currency gain | 9,915 | 4,990 | ||||||
| 54,028 | 30,730 | |||||||
F-62
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 14. | Finance income and finance costs (continued) |
Finance costs for the years ended December 31 were as follows:
| 2013 | 2012 | |||||||
| $ | $ | |||||||
Bank charges | 4,297 | 2,397 | ||||||
Accreted interest | 40,133 | - | ||||||
| 44,430 | 2,397 | |||||||
| 15. | Commitments and contingencies |
As at December 31, 2013 and in the normal course of business, the Company had obligations to make future payments, representing research and development contracts and other commitments that are known and committed in the amount of $247,000 over the next 12 months, $182,000 from 12 to 24 months, $127,000 from 24-36 months, and $78,000 each year thereafter.
The Company enters into research, development and license agreements in the ordinary course of business where the Company receives research services and rights to proprietary technologies. Milestone and royalty payments that may become due under various agreements are dependent on, among other factors, clinical trials, regulatory approvals and ultimately the successful development of a new drug, the outcome and timing of which is uncertain. Under the license agreement for SIRPaFc, the Company has future contingent milestones payable of $60,000 related to successful patent grants, $100,000, $200,000 and $300,000 on the first patient dosed in phase I, II and III trials respectively, and regulatory milestones on their first achievement totaling $5,000,000.
The Company periodically enters into research and license agreements with third parties that include indemnification provisions customary in the industry. These guarantees generally require the Company to compensate the other party for certain damages and costs incurred as a result of claims arising from research and development activities undertaken by or on behalf of the Company. In some cases, the maximum potential amount of future payments that could be required under these indemnification provisions could be unlimited. These indemnification provisions generally survive termination of the underlying agreement. The nature of the indemnification obligations prevents the Company from making a reasonable estimate of the maximum potential amount it could be required to pay. Historically, the Company has not made any indemnification payments under such agreements and no amount has been accrued in the consolidated financial statements with respect to these indemnification obligations.
| 16. | Related parties |
For the years ended December 31, 2013 and 2012, the key management personnel of the Company were the Board of Directors, the Executive Chairman, the Chief Executive Officer, the Chief Financial Officer, the Chief Scientific Officer and the Vice-President Drug Development.
F-63
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 16. | Related parties (continued) |
Compensation for key management personnel of the Company for the years ended December 31 was as follows:
| 2013 | 2012 | |||||||
| $ | $ | |||||||
Salaries, fees and short-term benefits | 844,425 | 396,400 | ||||||
Share-based compensation | 316,069 | 55,272 | ||||||
Total | 1,160,494 | 451,672 | ||||||
Executive officers and directors participate in the stock option plan and officers participate in the Company’s benefit plans. Directors receive annual fees for their services. As at December 31, 2013, the key management personnel controlled approximately 4% of the voting shares of the Company.
Outstanding balances with related parties at the year-end are unsecured, interest free and settlement occurs in cash. There have been no guarantees provided or received for any related party receivables or payables. For the year ended December 31, 2013, $138,750 was paid to a director for consulting fees (2012 - nil).
| 17. | Operating segment |
The Company has a single operating segment, the research and development of immunotherapy drugs for the treatment of cancer. Substantially all of the Company’s operations, assets, and employees are in Canada.
| 18. | Management of capital |
The Company defines its capital as share capital, warrants and contributed surplus. The Company’s objectives when managing capital are to ensure there are sufficient funds available to carry out its research and development programs. To date, these programs have been funded primarily through the sale of equity securities and the exercise of common share purchase warrants.
The Company also sources non-dilutive funding by accessing grants, government assistance and tax incentives, and through partnerships with corporations and research institutions. The Company uses budgets and purchasing controls to manage its costs.
The Company is not exposed to any externally imposed capital requirements.
| 19. | Financial instruments |
Fair value
IFRS 13, Fair Value Measurement, provides a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable or unobservable. Observable inputs are those which
F-64
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 19. | Financial instruments (continued) |
reflect market data obtained from independent sources, while unobservable inputs reflects the Company’s assumptions with respect to how market participants would price an asset or liability. These two inputs used to measure fair value fall into the following three different levels of the fair value hierarchy:
Level 1 Quoted prices in active markets for identical instruments that are observable.
Level 2 Quoted prices in active markets for similar instruments; inputs other than quoted prices that are observable and derived from or corroborated by observable market data.
Level 3 Valuations derived from valuation techniques in which one or more significant inputs are unobservable.
The hierarchy requires the use of observable market data when available.
The Company has classified cash and marketable securities as Level 1. The loan payable has been classified as Level 2.
Cash, marketable securities, amounts receivable, accounts payable and accrued liabilities, and other current liabilities, due within one year, are all short-term in nature and, as such, their carrying values approximate fair values. The fair value of the non-current loan payable and long-term liability is estimated by discounting the expected future cash flows at the cost of money to the Company, which is equal to its carrying value.
Risks
The Company has exposure to credit risk, liquidity risk, interest rate risk and currency risk. The Company’s Board of Directors has overall responsibility for the establishment and oversight of the Company’s risk management framework. The Audit Committee of the Board is responsible to review the Company’s risk management policies.
| (a) | Credit risk |
Credit risk is the risk of financial loss to the Company if a counterparty to a financial instrument fails to meet its contractual obligations, and arises principally from the Company’s cash and cash equivalents and amounts receivable. The carrying amount of these financial assets represents the maximum credit exposure. The Company follows an investment policy to mitigate against the deterioration of principal and to enhance the Company’s ability to meet its liquidity needs. Cash and cash equivalents are on deposit with a major Canadian chartered bank. Amounts receivable are primarily comprised of amounts due from the federal government.
| (b) | Liquidity risk |
Liquidity risk is the risk that the Company will not be able to meet its financial obligations as they fall due. The Company is a development stage company and is reliant on external fundraising to support its
F-65
Table of Contents
TRILLIUM THERAPEUTICS INC.
(formerly STEM CELL THERAPEUTICS CORP.)
Notes to the Consolidated Financial Statements
December 31, 2013
Amounts in Canadian Dollars
| 19. | Financial instruments (continued) |
operations. Once funds have been raised, the Company manages its liquidity risk by investing in cash and cash equivalents to provide regular cash flow for current operations. It also manages liquidity risk by continuously monitoring actual and projected cash flows. The Board of Directors reviews and approves the Company’s operating and capital budgets, as well as any material transactions not in the ordinary course of business. The majority of the Company’s accounts payable and accrued liabilities have maturities of less than three months.
| (c) | Interest rate risk |
Interest rate risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market interest rates. The Company’s cash and cash equivalents are highly liquid holdings in bank accounts or high interest savings accounts which have a variable rate of interest. The Company manages its interest rate risk by holding highly liquid short-term instruments and by holding its investments to maturity, where possible. For the year ended December 31, 2013, the Company earned interest income of $44,113 (2012 - $25,740). Therefore, a 1% change in the average interest rate for the year would have a net impact on finance income of $441.
| (d) | Currency risk |
The Company is exposed to currency risk related to the fluctuation of foreign exchange rates and the degree of volatility of those rates. Currency risk is limited to the portion of the Company’s business transactions denominated in currencies other than the Canadian dollar which are primarily expenses in US dollars. The Company manages its exposure to currency fluctuations by holding cash and cash equivalents denominated in US dollars in amounts approximating current US dollar financial liabilities and US dollar planned expenditures. As at December 31, 2013, the Company held US dollar cash and cash equivalents in the amount of US$493,031 (2012 - US$157,383) and had US dollar denominated accounts payable and accrued liabilities in the amount of US$88,063 (2012 - US$30,396). Therefore, a 1% change in the foreign exchange rate would have a net impact on finance costs of $4,050 (2012 – $1,270).
US dollar expenses for the year ended December 31, 2013 were approximately US$518,000 (2012 – US$532,000). Varying the US exchange rate for the year to reflect a 5% strengthening of the Canadian dollar would have decreased the net loss by approximately $26,000 (2012 - $27,000) assuming that all other variables remained constant.
F-66
Table of Contents
TRILLIUM THERAPEUTICS INC.
[A development stage company]
INTERIM FINANCIAL STATEMENTS
FOR THE NINE MONTHS ENDED
MARCH 31, 2013 AND 2012
(UNAUDITED)
96 Skyway Avenue,
Toronto, Ontario M9W 4Y9
www.trilliumtherapeutics.com
F-67
Table of Contents
[A development stage company]
Incorporated under the laws of Ontario
Balance Sheets
[See note 1 – Basis of presentation]
Amounts in Canadian Dollars
(Unaudited)
| As at | ||||||||
March 31, 2013 $ | June 30, 2012 $ | |||||||
ASSETS | ||||||||
Current | ||||||||
Cash and cash equivalents | 633,845 | 118,953 | ||||||
Marketable securities | 630,486 | 1,807,471 | ||||||
Accounts receivable | 140,538 | 154,945 | ||||||
Prepaid expenses | 25,045 | 57,508 | ||||||
Tax credits receivable | 499,509 | 1,315,000 | ||||||
Contract research deposits | 8,333 | 15,974 | ||||||
Total current assets | 1,937,756 | 3,469,851 | ||||||
Property and equipment, net[note 3] | 115,681 | 139,820 | ||||||
| 2,053,437 | 3,609,671 | |||||||
LIABILITIES AND SHAREHOLDERS’ DEFICIENCY | ||||||||
Current | ||||||||
Accounts payable and accrued liabilities | 198,581 | 203,186 | ||||||
Deferred revenue | — | 3,786 | ||||||
Total current liabilities | 198,581 | 206,972 | ||||||
Loan payable[note 4] | 530,154 | 292,363 | ||||||
Convertible debentures[note 5] | 10,000,000 | 9,710,122 | ||||||
Interest payable[note 6] | 5,014,337 | 22,650,198 | ||||||
Preference share liability[note 6] | 4 | 13,500,000 | ||||||
Total liabilities | 15,743,076 | 46,359,655 | ||||||
Shareholders’ deficiency | ||||||||
Common shares[note 7] | 15,139,864 | 1,639,868 | ||||||
Stock-based compensation[note 8] | 613,534 | 607,899 | ||||||
Contributed surplus[notes 5, 6 and 9] | 32,783,141 | 11,885,765 | ||||||
Deficit | (62,226,178 | ) | (56,883,516 | ) | ||||
Total shareholders’ deficiency | (13,689,639 | ) | (42,749,984 | ) | ||||
| 2,053,437 | 3,609,671 | |||||||
Commitments and contingencies[note 11]
Subsequent event[note 12]
Differences between Canadian and United States Generally Accepted Accounting Principles[note 13]
See accompanying notes to the interim financial statements
F-68
Table of Contents
Statements of Operations and Deficit
Amounts in Canadian Dollars
(Unaudited)
For the nine $ | For the nine $ | |||||||
REVENUES | ||||||||
Licensing | 3,786 | 11,359 | ||||||
Contract | — | — | ||||||
Investment income | 14,796 | 27,334 | ||||||
| 18,582 | 38,693 | |||||||
EXPENSES | ||||||||
Research and development, net[notes 2 and 11] | 1,420,498 | 1,643,681 | ||||||
General and administrative | 307,034 | 148,569 | ||||||
Interest accretion on convertible debentures | 289,878 | 1,220,839 | ||||||
Accrued interest on convertible debentures | 1,293,190 | 1,102,919 | ||||||
Interest accretion on preference share liability | — | — | ||||||
Accrued interest on preference share liability | 1,968,325 | 2,094,872 | ||||||
Sublicense | 58,180 | 127,512 | ||||||
Amortization | 24,139 | 121,475 | ||||||
| 5,361,244 | 6,459,867 | |||||||
Loss before income taxes | (5,342,662 | ) | (6,421,174 | ) | ||||
Provision for income taxes | — | — | ||||||
Net loss for the period | (5,342,662 | ) | (6,421,174 | ) | ||||
Deficit, beginning of period | (56,883,516 | ) | (48,418,833 | ) | ||||
Sale of technology rights in exchange for common shares | — | — | ||||||
Deficit, end of period | (62,226,178 | ) | (54,840,007 | ) | ||||
See accompanying notes to the interim financial statements
F-69
Table of Contents
Statements of Cash Flows
Amounts in Canadian Dollars
(Unaudited)
For the nine $ | For the nine $ | |||||||
OPERATING ACTIVITIES | ||||||||
Net loss for the period | (5,342,662 | ) | (6,421,174 | ) | ||||
Add (deduct) items not affecting cash | ||||||||
Amortization of property and equipment | 24,139 | 33,975 | ||||||
Amortization of technology rights | — | 87,500 | ||||||
Amortization of deferred revenue | (3,786 | ) | (11,359 | ) | ||||
Stock-based compensation | 5,635 | 10,202 | ||||||
Interest accretion on convertible debentures | 289,878 | 1,220,839 | ||||||
Accrued interest on convertible debentures | 1,293,190 | 1,102,919 | ||||||
Interest accretion on preference share liability | — | — | ||||||
Accrued interest on preference share liability | 1,968,325 | 2,094,872 | ||||||
Write-down of technology rights | — | — | ||||||
Loss on disposal of property and equipment | — | — | ||||||
Gain on sale of marketable securities | — | — | ||||||
| (1,765,281 | ) | (1,882,226 | ) | |||||
Net change in non-cash working capital balances related to operations[note 10] | 865,397 | 12,720 | ||||||
Cash used in operating activities | (899,884 | ) | (1,869,506 | ) | ||||
INVESTING ACTIVITIES | ||||||||
Purchase of property and equipment | — | (11,149 | ) | |||||
Purchase of technology rights | — | — | ||||||
Purchase of marketable securities | (1,000,000 | ) | (1,500,000 | ) | ||||
Proceeds from sale of marketable securities | 2,176,985 | (6,684 | ) | |||||
Cash provided by (used in) investing activities | 1,176,985 | (1,517,833 | ) | |||||
FINANCING ACTIVITIES | ||||||||
Issuance of common shares | — | — | ||||||
Issuance of convertible debentures, net of costs | — | 1,993,634 | ||||||
Issuance of Class A preference shares, net of costs | — | — | ||||||
Increase in loan payable | 237,791 | 108,133 | ||||||
Cash provided by financing activities | 237,791 | 2,101,767 | ||||||
Net increase (decrease) in cash and cash equivalents during the period | 514,892 | (1,285,572 | ) | |||||
Cash and cash equivalents, beginning of period | 118,953 | 1,906,787 | ||||||
Cash and cash equivalents, end of period | 633,845 | 621,215 | ||||||
See accompanying notes to the interim financial statements
F-70
Table of Contents
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 1. | Corporate information and basis of presentation |
Trillium Therapeutics Inc. [“Trillium” or the “Company”] is a privately held biotechnology company specializing in the discovery and development of innovative therapies that restore balance to the immune system in conditions associated with aberrant and harmful immune responses, such as autoimmune and inflammatory disorders, cancer and viral disease.
The Company has earned product licensing revenues as a result of agreements with development partners. Given the early stage of the technologies under these development agreements and risks associated with continued development of individual technologies in the biotechnology industry in general, the Company is considered to be in the development stage.
The success of the Company is dependent on completing product development and commercializing or entering into agreements with third parties to commercialize its products, and obtaining financing for its ongoing operations. It is not possible to predict either the commercial success of the Company’s products or the Company’s ability to fund these programs going forward. The Company incurred a net loss of $5,342,662 for the nine months ended March 31, 2013 and has a deficit of $62,226,178 as at March 31, 2013 and, without an additional source of funding, it will have inadequate funds to continue its existing operations for the coming year. These circumstances raise substantial doubt as to the ability of the Company to continue as a going concern. The Company’s ability to continue as a going concern is dependent on receipt of additional convertible debenture financing, sourcing alternative financing or its ability to generate revenues from its products through licensing activities; however, there can be no assurance that these activities will be successful.
These financial statements have been prepared on a going concern basis, which assumes the Company will be able to realize its assets and discharge its liabilities in the normal course of business for the foreseeable future. These financial statements do not include adjustments that would be necessary should the Company be unable to continue as a going concern.
| 2. | Significant accounting policies |
These financial statements were prepared in accordance with Part II of the Canadian Institute of Chartered Accountants [“CICA”] Accounting Handbook - Accounting Standards for Private Enterprises, which sets out generally accepted accounting principles for non-publicly accountable enterprises in Canada [“Canadian GAAP”].
The Company’s principal accounting policies were outlined in the Company’s annual audited financial statements for the year ended June 30, 2012 and have been applied consistently to all periods presented in these financial statements. The notes presented in these financial statements include only significant events and transactions occurring since our last fiscal year end and are not fully inclusive of all matters required to be disclosed in our annual audited financial statements. These statements should be read in conjunction with the annual audited financial statements for the year ended June 30, 2012.
F-71
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 2. | Significant accounting policies (continued) |
Research and development costs
Research and development costs are charged to income as incurred, net of grants or related tax credits, unless they meet the criteria under Canadian GAAP for deferral and amortization. No development costs have been deferred to date.
The Company records investment tax credits when there is reasonable assurance that the assistance will be realized. Government assistance recognized as a credit to research and development costs for the nine months ended March 31, 2013 was $595,787 [2012 - $967,147].
| 3. | Property and equipment |
Property and equipment consist of the following:
Cost $ | March 31, 2013 Accumulated amortization $ | Net $ | Cost $ | June 30, 2012 Accumulated amortization $ | Net $ | |||||||||||||||||||
Research equipment | 865,203 | 762,557 | 102,646 | 865,203 | 744,282 | 120,921 | ||||||||||||||||||
| Computer hardware and software | 70,555 | 63,897 | 6,658 | 70,555 | 61,946 | 8,609 | ||||||||||||||||||
Office equipment | 38,574 | 33,871 | 4,703 | 38,574 | 33,034 | 5,540 | ||||||||||||||||||
Leasehold improvements | 52,452 | 50,778 | 1,674 | 52,452 | 47,702 | 4,750 | ||||||||||||||||||
| 1,026,784 | 911,103 | 115,681 | 1,026,784 | 886,964 | 139,820 | |||||||||||||||||||
During the year ended June 30, 2012, the Company revised its estimate of the remaining useful life of certain research equipment and computer hardware and recognized $40,977 of additional depreciation expense. The Company also wrote off $20,118 of research equipment and $50,754 of computer hardware which was fully depreciated and no longer in use.
| 4. | Loan payable |
On February 1, 2012 the Company entered into a contribution agreement with the Federal Economic Development Agency for Southern Ontario where the Company can receive a repayable contribution of up to $965,000 to support the clinical development of its TTI-1612 program. The period of contribution is up to December 31, 2013. As at March 31, 2013, the Company has repayable contributions of $530,154 representing the outstanding principal balance, repayable in monthly installments of $16,083 for 60 months beginning on December 1, 2014. The loan payable bears no interest.
F-72
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 5. | Convertible debentures |
Amount $ | ||||
Balance at July 1, 2011 | 6,786,593 | |||
Debt portion of convertible debentures issued | 1,293,712 | |||
Accreted interest | 1,629,817 | |||
Balance at June 30, 2012 | 9,710,122 | |||
Accreted interest | 289,878 | |||
Balance at March 31, 2013 | 10,000,000 | |||
On September 15, 2011, the Company issued $2,000,000 of 12% convertible debentures with a maturity date of August 31, 2012 and received net proceeds of $1,993,634 after cash issue costs. The debentures plus accrued and unpaid interest are convertible at any time at the option of the holder, and under certain conditions, into Class A preference shares of the Company at the Class A Conversion Price, established by the Company’s shareholders’ agreement, divided by 1.5. Under the terms of the debentures subscription agreement, the Company issued 451,128 Class A preference share purchase warrants entitling the holders to purchase one Class A preference share at a price of $1.33 per share, subject to adjustment at any time up to September 16, 2016.
Consistent with CICA Handbook Part II Section 3856, the debentures were split into separate debt and equity components on the balance sheet. The fair value of the debt, initially determined to be $1,293,712 and described as “convertible debentures” on the balance sheet, has been estimated as the present value of future payments based on the Company’s estimated incremental borrowing rates for similar debt agreements. The fair value of the equity portion of the debentures of $699,922 was included in contributed surplus and was estimated using the Black-Scholes option pricing model with the following weighted-average assumptions for 2012: risk-free interest rate of 0.9%; dividend yield of 0%; expected volatility of 93%; and an expected life of 1.0 year. The warrants were issued for nominal consideration.
In the first quarter, the debenture holders unanimously extended the maturity date of all outstanding convertible debentures to October 31, 2013. Subsequent to March 31, 2013, Trillium merged with Stem Cell Therapeutics Corp. which does not intend to call the debentures in the next 12 months (see note 12).
| 6. | Preference share liability |
The Company is authorized to issue an unlimited number of Class A preference shares. Based on the terms of these shares, the consideration received on their issuance was split into debt and equity components.
Continuity of issued preference shares
Shares # | Amount $ | |||||||
Balance at June 30, 2011 and 2012 | 10,150,377 | 13,500,000 | ||||||
Converted to common shares | (10,150,374 | ) | (13,499,996 | ) | ||||
Balance at March 31, 2013 | 3 | 4 | ||||||
F-73
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 6. | Preference share liability (continued) |
The equity component of the issued preference shares of $7,610,125 is included in contributed surplus.
The Class A preference shares are convertible into common shares at the Class A Conversion Price, initially at $1.33 per share, which is subject to adjustment. Each holder of a Class A preference share is entitled to vote for each common share into which the holder’s Class A preference shares are convertible. Dividends on these shares are cumulative at 8% per annum.
The preference shareholders converted all but 3 preference shares to common shares on March 20, 2013. The undeclared and unpaid cumulative dividends on the Class A preference shares of $15,209,062 and the Part VI.1 tax liability on these dividends of $5,688,314 on March 20, 2013 was reversed and recognized as contributed surplus at the time of the conversion to common shares. This amount of cumulative dividends was described as “interest payable” on the balance sheet. Included in the “interest payable” balance as at March 31, 2013 is $nil [June 30, 2012 – 13,663,608] cumulative preference share dividends, the Part VI.1 tax liability on these dividends of $nil [June 30, 2012 - $5,265,443] and accrued interest on the Debentures of $5,014,337 [June 30, 2012 - $3,721,147].
Warrants
The Company issued Class A preference share purchase warrants in connection with its Class A preference share and convertible debentures financings. Each warrant is exercisable into one Class A preference share at a price of $1.33 per share, subject to adjustment. As at March 31, 2013, there were 5,300,752 [June 30, 2012 - 5,300,752] warrants issued and outstanding. All warrants issued to purchase Class A preference shares were issued for nominal consideration and all warrants expire on September 16, 2016.
| 7. | Share capital |
Authorized
The Company is authorized to issue an unlimited number of common shares and an unlimited number of Class A preference shares[note 6].
Continuity of issued common shares
Shares # | Amount $ | |||||||
Balance at June 30, 2011 and 2012 | 6,116,667 | 1,639,868 | ||||||
Issued on conversion of Class A Preference shares[note 6] | 10,150,374 | 13,499,996 | ||||||
Balance at March 31, 2013 | 16,267,041 | 15,139,864 | ||||||
| 8. | Stock-based compensation |
Under the Company’s stock option plan enacted April 10, 2003, options may be granted to directors, officers, employees and consultants of the Company to purchase up to 2,429,129 common shares. Options
F-74
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 8. | Stock-based compensation (continued) |
are granted at the fair market value of the common shares on the date of grant. Options vest at various rates and have a term not exceeding 10 years.
For the nine months ended March 31, 2013 no options were granted, cancelled or expired. For the nine months ended March 31, 2012, 20,000 stock options were granted with an exercise price of $0.43 and a fair value of $0.38. For the nine months ended March 31, 2013 the Company recorded stock-based compensation expense of $5,635 (2012 - $10,202).
At March 31, 2013, there were 2,236,673 (June 30, 2012 – 2,236,673) stock options outstanding with a weighted average exercise price of $0.76, and 2,204,280 (June 30, 2012 – 2,173,339) stock options exercisable with a weighted average exercise price of $0.77.
| 9. | Contributed surplus |
Amount $ | ||||
Balance at June 30, 2011 | 11,185,843 | |||
Equity portion of convertible debentures issued[note 5] | 699,922 | |||
Balance at June 30, 2012 | 11,885,765 | |||
Recognized on conversion of Class A Preference shares[note 6] | 20,897,376 | |||
Balance at March 31, 2013 | 32,783,141 | |||
| 10. | Statements of cash flows |
The net change in non-cash working capital balances related to operations consists of the following:
2013 $ | 2012 $ | |||||||
Decrease (increase) in current assets | ||||||||
Accounts receivable | 14,407 | (82,397 | ) | |||||
Prepaid expenses | 32,463 | (51,822 | ) | |||||
Tax credits receivable | 815,491 | 355,000 | ||||||
Contract research deposits | 7,641 | 47,294 | ||||||
| 870,002 | 268,075 | |||||||
Increase (decrease) in current liabilities | ||||||||
Accounts payable and accrued liabilities | (4,605 | ) | (255,355 | ) | ||||
| (4,605 | ) | (255,355 | ) | |||||
| 865,397 | 12,720 | |||||||
F-75
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 11. | Commitments and contingencies |
[a] The Company enters into research, development and license agreements with various parties in the ordinary course of business whereby the Company receives research services and rights to proprietary technologies. The agreements require compensation to be paid by the Company, typically by a combination of the following methods:
| [i] | Fees comprising amounts due initially upon entering into the agreements as well as additional amounts due either on specified timelines or defined services to be provided. |
| [ii] | Milestone payments that are dependent on products developed under the agreements proceeding towards specified plans of clinical trials and commercial development. |
| [iii] | Royalty payments calculated as a percentage of net sales commencing upon commercial sales of any product candidates developed from the technologies. |
As at March 31, 2013, the Company has commitments under agreements to fund research, pre-clinical studies and manufacturing in the amount of $57,000 over the next year [June 30, 2012 - $497,000]. Included in these commitments is $nil [June 30, 2012 - $100,000] to the University Health Network [“UHN”] which is a shareholder. During the nine months ended March 31, 2013, the Company incurred $75,000 [2012 - $233,334] in expenses to shareholders for research and scientific advisory services and license fees which were recorded at the agreed upon exchange amount.
Milestone and royalty-related amounts that may become due under various agreements are dependent on, among other factors, pre-clinical safety and efficacy, clinical trials, regulatory approvals and ultimately the successful development of a new drug, the outcome and timing of which are uncertain. Amounts due per the various agreements for milestone payments will be accrued once the occurrence of a milestone is likely. Amounts due as royalty payments will be accrued as commercial revenues from the product are earned.
| [i] | On February 1, 2010, the Company entered into a license agreement with UHN and The Hospital For Sick Children to acquire intellectual property relating to the methods and compounds for the modulation of the SIRP±-CD47 interaction for use in therapeutic cancer applications. The Company paid an up-front license fee of $150,000 and committed to pay annual maintenance fees of $25,000 in addition to patent issuance, development and regulatory milestones, and royalties on commercial sales. As at March 31, 2013, no future contingent consideration had been paid. These technology rights have been fully amortized. |
| [ii] | On March 26, 2008, the Company entered into a license agreement with the University of Maryland, Baltimore [“UMB”] wherein UMB granted the Company an exclusive license and non-exclusive license under the UMB patent rights and a non-exclusive sublicense under the Children’s Medical Center Corporation [“CMCC”] patent rights related to the Company’s TTI-1612 technology. |
The Company has a commitment to pay U.S.$10,000 in annual maintenance fees and there is future contingent consideration in the form of milestone payments of up to $760,000, and royalties. During
F-76
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 11. | Commitments and contingencies (continued) |
the year ended June 30, 2012, the Company paid a $25,000 milestone fee to UMB on the initiation of its Phase I trial for TTI-1612. As at March 31, 2013, no other future contingent consideration had been paid. These technology rights have been fully amortized.
[b] The Company’s future commitments under operating leases for premises and equipment are $27,500 for 2013.
[c] The Company enters into license agreements with third parties that include indemnification provisions in the ordinary course of business that are customary in the industry. Those guarantees generally require the Company to compensate the other party for certain damages and costs incurred as a result of third-party claims or damages arising from these transactions. In some cases, the maximum potential amount of future payments that could be required under these indemnification provisions is unlimited. The nature of the indemnification obligations prevents the Company from making a reasonable estimate of the maximum potential amount that it could be required to pay. Historically, the Company has not made any indemnification payments under such agreements and no amount has been accrued in the accompanying financial statements with respect to these indemnification obligations.
| 12. | Subsequent event |
On April 9, 2013, the Company was merged into Stem Cell Therapeutics Corp. (“SCT”) by way of an amalgamation with a newly-created SCT subsidiary. On closing of the transaction, the security holders of Trillium received an aggregate consideration of $2,850,000 comprised of $1.2 million in cash and $1.65 million in SCT common shares.
| 13. | Differences between Canadian and United States Generally Accepted Accounting Principles |
The financial statements have been prepared in accordance with Canadian GAAP, which differ in certain respects from those principles that the Company would have followed if its financial statements had been prepared in accordance with generally accepted accounting principles in the United States (US GAAP).
A reconciliation of net earnings from Canadian GAAP to conform with US GAAP is as follows:
Note
| March 31, $ | March 31, $ | ||||||||||
Net loss for the period based on Canadian GAAP | (5,342,662 | ) | (6,421,174 | ) | ||||||||
Acquired research and development | B | - | 87,500 | |||||||||
Preference shares – accrued dividends | C | 1,545,454 | 1,496,337 | |||||||||
Preference shares – part VI.1 tax (interest expense) | C | 422,871 | - | |||||||||
Preference shares – income tax recovery | C | 5,265,443 | - | |||||||||
Convertible debentures – accreted interest expense | D | 289,878 | 1,220,839 | |||||||||
Convertible debentures – issue costs | D | (13,043 | ) | (22,769 | ) | |||||||
| Net income (loss) and comprehensive income (loss) for the period based on US GAAP | 2,167,941 | (3,639,267 | ) | |||||||||
F-77
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 13. | Differences between Canadian and United States Generally Accepted Accounting Principles (continued) |
A reconciliation of shareholders’ deficiency from Canadian GAAP to conform with US GAAP is as follows:
Note
| March 31, $ | June 30, $ | ||||||||||
Shareholders’ deficiency based on Canadian GAAP | (13,689,639 | ) | (42,749,984 | ) | ||||||||
Preference shares – accreted interest | C | 7,610,125 | 7,610,125 | |||||||||
Preference shares – equity component | C | (7,610,125 | ) | (7,610,125 | ) | |||||||
Convertible debentures – accreted interest | D | 4,480,421 | 4,190,543 | |||||||||
Convertible debentures – equity component | D | (4,275,640 | ) | (4,275,640 | ) | |||||||
Convertible debentures – issue costs | D | 7,134 | 20,178 | |||||||||
Shareholders’ deficiency based on US GAAP | (13,477,724 | ) | (42,814,903 | ) | ||||||||
Reconciling items
A) Government assistance – investment tax credits
Under Canadian GAAP, the Company records investment tax credits arising from research and development activities on a net basis against costs to which they relate. Under US GAAP, the Company recognizes investment tax credits as a reduction of tax expense. Accordingly, to reconcile to US GAAP, research and development costs would increase and income tax recovery would increase for the nine months ended March 31, 2013 and 2012 by $595,787 and $967,147, respectively. Since both adjustments are on the statement of operations and deficit, there is no impact on net loss.
B) Acquired research and development
Under Canadian GAAP, the Company capitalized acquired research and development assets on the balance sheet as technology rights and amortized these assets over their estimated useful lives. Under US GAAP, these research and development assets are expensed at the time of acquisition because they do not have alternative uses. Accordingly, the amount of technology rights would be nil as at March 31, 2013 and June 30, 2012 and the amortization reflected in the statement of operations and deficit for the nine months ended March 31, 2013 and 2012 would be lower by $0 and $87,500, respectively.
C) Preference shares
Under Canadian GAAP, the preference shares are initially bifurcated into debt and equity components. Interest is accreted on the debt component up to the original subscription amount of the shares and both accrued cumulative dividends and Part IV.1 tax are recognized as interest expense and included in interest payable. Since the shares have reached their redemption date, they are recorded at their original subscription amount in the financial statements. Upon conversion, the pro rata share of the debt
F-78
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 13. | Differences between Canadian and United States Generally Accepted Accounting Principles (continued) |
component was reclassified to common shares and the pro rata share of the accrued cumulative dividends and related Part IV.1 tax was reclassified to contributed surplus. Under US GAAP, the preference shares are classified as temporary equity and adjusted to their maximum redemption amount each balance sheet date once they become redeemable. Accrued cumulative dividends are recorded as a charge to equity and included in temporary equity and Part VI.1 tax is recognized as income tax expense and included in taxes payable. Upon conversion, the pro rata share of the amount recorded in temporary equity is reclassified to common shares and the cumulative Part VI.1 tax payable is reversed as an income tax recovery.
To reconcile to US GAAP as at and for the nine-months ended March 31, 2013 (a) the $4 recorded as the preference share liability under Canadian GAAP (which is equal to the shares’ maximum redemption amount) is reclassified to temporary equity, (b) the $7,610,125 set up in contributed surplus on the original bifurcation under Canadian GAAP is deducted from shareholders’ deficiency and $7,610,125 of cumulative accreted interest included in the deficit is added back to shareholders’ deficiency, (c) the $15,209,062 of accrued cumulative dividends classified as contributed surplus under Canadian GAAP is reclassified to common shares and the $1,545,454 of accrued cumulative dividends recognized as interest expense is added back to net loss, and (d) the $5,688,314 of Part VI.1 tax liability classified as contributed surplus under Canadian GAAP is reclassified to the deficit, the $422,871 of Part VI.1 tax recognized as interest expense is reversed and $5,265,443 of income tax recovery is recorded.
To reconcile to US GAAP as at June 30, 2012 and for the nine-months ended March 31, 2012 (a) the $13,500,000 recorded as a non-current liability under Canadian GAAP (which is currently equal to the shares’ maximum redemption amount) is reclassified to temporary equity, (b) the $7,610,125 set up in contributed surplus on the original bifurcation under Canadian GAAP is deducted from shareholders’ deficiency and $7,610,125 of cumulative accreted interest included in the deficit is added back to shareholders’ deficiency, (c) the $13,663,608 of accrued cumulative dividends classified as interest payable under Canadian GAAP is reclassified to temporary equity under US GAAP and the $1,496,337 of accrued cumulative dividends recognized as interest expense is added back to net loss, and (d) the $5,265,443 of Part VI.1 tax liability classified as interest payable under Canadian GAAP is reclassified to taxes payable and the $598,535 of Part VI.1 tax recognized as interest expense is reclassified to income tax expense.
Preference share subscribers also received warrants exercisable into preference shares. Under Canadian GAAP, the Company did not account for the warrants in the financial statements as it was determined that the fair value of the warrants at the time of issuance is nominal. Under US GAAP, the warrants are classified as a liability and carried at fair value, both initially and on an on-going basis. Consistent with Canadian GAAP, the Company determined the initial fair value of the warrants to be nominal. The Company also determined that the fair value of the warrants has remained nominal since the time of issuance, resulting in no measurement difference between Canadian and US GAAP.
D) Convertible debentures
Under Canadian GAAP, the convertible debentures are initially bifurcated into debt and equity components and interest is accreted on the debt component up to its face amount of the shares. Under
F-79
Table of Contents
TRILLIUM THERAPEUTICS INC.
Notes to the Financial Statements
March 31, 2013
Amounts in Canadian Dollars
(Unaudited)
| 13. | Differences between Canadian and United States Generally Accepted Accounting Principles (continued) |
US GAAP, the convertible debenture is classified as debt and carried at amortized cost. Accordingly, to reconcile to US GAAP, accreted interest expense recorded of $4,480,421 and $4,190,543 as at March 31, 2013 and June 30, 2012, respectively, must be added back to shareholders’ deficiency and the amount set up in equity on the original bifurcation of the convertible debt under Canadian GAAP needs to be deducted from shareholders’ deficiency and added to the convertible debt liability in the amounts of $4,275,640 and $4,275,640 as at March 31, 2013 and June 30, 2012, respectively.
Similarly, accreted interest expense recorded under Canadian GAAP would need to be added back to the net loss under US GAAP in the amounts of $289,878 and $1,220,839 for the nine months ended March 31, 2013 and 2012, respectively.
There is also a balance sheet classification difference related to the costs incurred to issue the convertible debt. Under Canadian GAAP the costs were allocated to the debt and equity components on initial bifurcation and only the portion allocated to the debt component was subsequently amortized, whereas US GAAP requires all of the costs to be set up as a separate asset and amortized. Accordingly, the issue costs allocated to equity under Canadian GAAP are amortized under US GAAP, resulting in a decrease in net income of $13,043 and $22,769 for the nine months ended March 31, 2013 and 2012, respectively. Additionally, the total balance of unamortized issue costs is reclassified to deferred financing costs, including the unamortized portion of the issue costs allocated to equity under Canadian GAAP in the amounts of $7,134 and 20,178 as at March 31, 2013 and June 30, 2012, respectively.
Convertible debt subscribers also received warrants exercisable into preference shares. Under Canadian GAAP, the Company did not account for the warrants in the financial statements as it was determined that the fair value of the warrants at the time of issuance is nominal. Under US GAAP, the warrants are classified as a liability and carried at fair value, both initially and on an on-going basis. Consistent with Canadian GAAP, the Company determined the initial fair value of the warrants to be nominal. The Company also determined that the fair value of the warrants has remained nominal since the time of issuance, resulting in no measurement difference between Canadian and US GAAP.
Cash flow
In addition to the differences between Canadian and US GAAP related to the recognition and measurement of transactions by the Company, there are differences in the manner in which items are classified in the statement of cash flows. These classification differences have no impact on cash used in operating, investing and financing activities.
F-80
Table of Contents
TRILLIUM THERAPEUTICS INC.
[A development stage company]
JUNE 30, 2012
F-81
Table of Contents
REPORT OF INDEPENDENT AUDITORS
To the Directors of
Trillium Therapeutics Inc.
We have audited the accompanying financial statements ofTrillium Therapeutics Inc., which comprise the balance sheets as of June 30, 2012 and 2011, and July 1, 2010, and the related statements of operations and deficit and cash flows for the years ended June 30, 2012 and 2011. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with auditing standards generally accepted in the United States. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. We were not engaged to perform an audit of the Company’s internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position ofTrillium Therapeutics Inc. at June 30, 2012 and 2011, and July 1, 2010, and the results of its operations and its cash flows for the years ended June 30, 2012 and 2011 in conformity with Canadian accounting standards for private enterprises.
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company incurred a net loss of $8,464,683 during the year ended June 30, 2012 and, as at that date the Company has a deficit of $56,883,516. These conditions raise substantial doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to these matters also are described in Note 1. The financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from the outcome of this uncertainty.
| /s/ Ernst & Young LLP | ||
| Toronto, Canada, | Chartered Accountants | |
| September 27, 2012, except Note 16 as to which the date is August 11, 2014 | Licensed Public Accountants |
F-82
Table of Contents
[A development stage company]
Incorporated under the laws of Ontario
BALANCE SHEETS
[See note 1 - Basis of Presentation]
As at
June 30, 2012 $ | June 30, 2011 $ | July 1, 2010 $ | ||||||||||
ASSETS | ||||||||||||
Current | ||||||||||||
Cash and cash equivalents | 118,953 | 1,906,787 | 1,527,621 | |||||||||
Marketable securities[note 2] | 1,807,471 | 400,148 | 400,148 | |||||||||
Accounts receivable | 154,945 | 67,680 | 67,070 | |||||||||
Prepaid expenses | 57,508 | 33,325 | 38,703 | |||||||||
Tax credits receivable | 1,315,000 | 1,275,000 | 970,000 | |||||||||
Contract research deposits | 15,974 | 66,068 | 33,333 | |||||||||
Total current assets | 3,469,851 | 3,749,008 | 3,036,875 | |||||||||
Property and equipment, net[note 3] | 139,820 | 216,213 | 267,439 | |||||||||
Technology rights, net[note 4] | — | 87,500 | 249,811 | |||||||||
| 3,609,671 | 4,052,721 | 3,554,125 | ||||||||||
LIABILITIES AND SHAREHOLDERS’ DEFICIENCY | ||||||||||||
Current | ||||||||||||
Accounts payable and accrued liabilities | 203,186 | 427,228 | 239,809 | |||||||||
Deferred revenue | 3,786 | 15,145 | 269,993 | |||||||||
Total current liabilities | 206,972 | 442,373 | 509,802 | |||||||||
Deferred revenue | — | 3,786 | 29,208 | |||||||||
Loan payable[note 5] | 292,363 | — | — | |||||||||
Convertible debentures[note 6] | 9,710,122 | 6,786,593 | 3,861,425 | |||||||||
Interest payable[note 7] | 22,650,198 | 18,319,309 | 14,693,493 | |||||||||
Preference share liability[note 7] | 13,500,000 | 13,500,000 | 13,500,000 | |||||||||
Total liabilities | 46,359,655 | 39,052,061 | 32,593,928 | |||||||||
Shareholders’ deficiency | ||||||||||||
Share capital[note 8] | 1,639,868 | 1,639,868 | 1,639,868 | |||||||||
Stock-based compensation[note 9] | 607,899 | 593,782 | 566,368 | |||||||||
Contributed surplus[notes 6, 7 and 10] | 11,885,765 | 11,185,843 | 9,851,922 | |||||||||
Deficit | (56,883,516 | ) | (48,418,833 | ) | (41,097,961 | ) | ||||||
Total shareholders’ deficiency | (42,749,984 | ) | (34,999,340 | ) | (29,039,803 | ) | ||||||
| 3,609,671 | 4,052,721 | 3,554,125 | ||||||||||
Commitments and contingencies[notes 4 and 13]
Subsequent event[note 15]
Differences between Canadian and United States Generally Accepted Accounting Principles[note 16]
See accompanying notes
On behalf of the Board:
| Director | Director |
F-83
Table of Contents
[A development stage company]
STATEMENTS OF OPERATIONS AND DEFICIT
Year ended June 30
2012 $ | 2011 $ | |||||||
REVENUES | ||||||||
Licensing | 15,145 | 280,270 | ||||||
Contract | — | 10,092 | ||||||
Investment income | 38,902 | 29,751 | ||||||
| 54,047 | 320,113 | |||||||
EXPENSES | ||||||||
Research and development, net[notes 2 and 13] | 2,042,205 | 2,194,408 | ||||||
General and administrative | 213,265 | 207,163 | ||||||
Interest accretion on convertible debentures | 1,629,817 | 1,311,257 | ||||||
Accrued interest on convertible debentures | 1,507,278 | 1,018,768 | ||||||
Interest accretion on preference share liability | — | — | ||||||
Accrued interest on preference share liability | 2,823,611 | 2,607,048 | ||||||
Sublicense[note 13[a]] | 127,512 | 83,114 | ||||||
Amortization | 175,042 | 219,227 | ||||||
| 8,518,730 | 7,640,985 | |||||||
Loss before income taxes | (8,464,683 | ) | (7,320,872 | ) | ||||
Provision for income taxes | — | — | ||||||
Net loss for the period | (8,464,683 | ) | (7,320,872 | ) | ||||
Deficit, beginning of period | (48,418,833 | ) | (41,097,961 | ) | ||||
Sale of technology rights in exchange for common shares | — | — | ||||||
Deficit, end of period | (56,883,516 | ) | (48,418,833 | ) | ||||
See accompanying notes
F-84
Table of Contents
[A development stage company]
STATEMENTS OF CASH FLOWS
Year ended June 30
2012 $ | 2011 $ | |||||||
OPERATING ACTIVITIES | ||||||||
Net loss for the period | (8,464,683 | ) | (7,320,872 | ) | ||||
Add (deduct) items not affecting cash | ||||||||
Amortization of property and equipment | 87,542 | 56,916 | ||||||
Amortization of technology rights | 87,500 | 162,311 | ||||||
Amortization of deferred revenue | (3,786 | ) | (25,422 | ) | ||||
Stock-based compensation[note 9] | 14,117 | 27,414 | ||||||
Interest accretion on convertible debentures | 1,629,817 | 1,311,257 | ||||||
Accrued interest on convertible debentures | 1,507,278 | 1,018,768 | ||||||
Interest accretion on preference share liability | — | — | ||||||
Accrued interest on preference share liability | 2,823,611 | 2,607,048 | ||||||
Write-down of technology rights | — | — | ||||||
Loss on disposal of property and equipment | — | — | ||||||
Gain on sale of marketable securities | (1,500 | ) | — | |||||
| (2,320,104 | ) | (2,162,580 | ) | |||||
Net change in non-cash working capital balances related to operations[note 12] | (336,755 | ) | (400,396 | ) | ||||
Cash used in operating activities | (2,656,859 | ) | (2,562,976 | ) | ||||
INVESTING ACTIVITIES | ||||||||
Purchase of property and equipment | (11,149 | ) | (5,690 | ) | ||||
Purchase of technology rights | — | — | ||||||
Purchase of marketable securities | (1,500,000 | ) | (400,000 | ) | ||||
Proceeds from sale of marketable securities | 94,177 | 400,000 | ||||||
Cash used in investing activities | (1,416,972 | ) | (5,690 | ) | ||||
FINANCING ACTIVITIES | ||||||||
Issuance of common shares | — | — | ||||||
Issuance of convertible debentures, net of costs | 1,993,634 | 2,947,832 | ||||||
Issuance of Class A preference shares, net of costs | — | — | ||||||
Increase in loan payable | 292,363 | — | ||||||
Cash provided by financing activities | 2,285,997 | 2,947,832 | ||||||
Net increase (decrease) in cash and cash equivalents during the year | (1,787,834 | ) | 379,166 | |||||
Cash and cash equivalents, beginning of year | 1,906,787 | 1,527,621 | ||||||
Cash and cash equivalents, end of year | 118,953 | 1,906,787 | ||||||
See accompanying notes
F-85
Table of Contents
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
1. BASIS OF PRESENTATION
Trillium Therapeutics Inc. [“Trillium” or the “Company”] is a privately held biotechnology company specializing in the discovery and development of innovative therapies that restore balance to the immune system in conditions associated with aberrant and harmful immune responses, such as autoimmune and inflammatory disorders, cancer and viral disease.
The Company has earned product licensing revenues as a result of agreements with development partners. Given the early stage of the technologies under these development agreements and risks associated with continued development of individual technologies in the biotechnology industry in general, the Company is considered to be in the development stage.
The success of the Company is dependent on completing product development and commercializing or entering into agreements with third parties to commercialize its products, and obtaining financing for its ongoing operations. It is not possible to predict either the commercial success of the Company’s products or the Company’s ability to fund these programs going forward. The Company incurred a loss of $8,464,683 for the year ended June 30, 2012 and has a deficit of $56,883,516 as at June 30, 2012 and, without an additional source of funding, it will have inadequate funds to continue its existing operations for the coming year. These circumstances raise substantial doubt as to the ability of the Company to continue as a going concern. The Company’s ability to continue as a going concern is dependent on receipt of additional convertible debenture financing, sourcing alternative financing or its ability to generate revenues from its products through licensing activities; however, there can be no assurance that these activities will be successful.
These financial statements have been prepared on a going concern basis, which assumes the Company will be able to realize its assets and discharge its liabilities in the normal course of business for the foreseeable future. These financial statements do not include adjustments that would be necessary should the Company be unable to continue as a going concern.
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
| A. | SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES AND FIRST-TIME ADOPTION OF ACCOUNTING STANDARDS FOR PRIVATE ENTERPRISES |
These financial statements were prepared in accordance with Part II of the Canadian Institute of Chartered Accountants [“CICA”] Accounting Handbook Accounting Standards for Private Enterprises [“Part II”], which sets out generally accepted accounting principles for non-publicly accountable enterprises in Canada [“Canadian GAAP”] and include the significant accounting policies described hereafter:
Use of estimates
The preparation of financial statements in conformity with Canadian GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. The most significant estimates include tax credits receivable, the valuation allowance for future income tax assets, and the fair values used to account for equity and convertible debt transactions including stock-based compensation expense, and the fair values determined in connection with acquiring technology rights. Actual results could differ from those estimates.
F-86
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Cash equivalents, marketable securities and investment income
Trillium invests in high-quality government and corporate issuers with low credit risk. Cash equivalents consist of highly liquid investments with a maturity of 90 days or less at the time of purchase.
Marketable securities consist of fixed income securities with a maturity date beyond 90 days from the date of purchase and are recorded at their accreted value as they are held-to-maturity instruments. The estimated market value of marketable securities as at June 30, 2012 was $1,813,471 [2011 - $402,148]. Investment income includes interest income on marketable securities. Interest income is accrued as earned.
Property and equipment
Property and equipment are recorded at cost less accumulated amortization. Amortization is provided at rates that are expected to charge operations with the cost of the assets over their estimated useful lives as follows:
| Research and office equipment | 20% declining balance | |
| Computer hardware | 30% declining balance | |
| Computer software | 100% declining balance | |
| Leasehold improvements | Straight-line over lease term |
Each year, the Company assesses its property and equipment to determine if there has been an impairment in their value. There is an impairment loss when the carrying value of an asset exceeds the sum of the undiscounted cash flow expected from this asset. An impairment loss is measured as the amount by which the carrying amount of the asset exceeds its fair value.
Technology rights
At the time of acquisition of intangible assets, the Company makes an assessment to determine if they have an estimated useful life or whether they have an indefinite life. The Company has determined that the technology rights have finite lives and, accordingly, they are being amortized over their estimated useful lives, from two to four years, on a straight-line basis.
The Company annually reviews the carrying value of the technology rights for evidence of facts or changes in circumstance that might indicate a condition of impairment. An impairment loss would be recognized when estimates of undiscounted future cash flows expected to result from the use of an asset and its eventual disposition are less than the carrying amount. An impairment loss is measured as the amount by which the carrying amount of the asset exceeds its fair value.
Revenue recognition
Research and development funding and license fees
Non-refundable, up-front fees received for access to the Company’s proprietary technology are deferred and recognized as revenue on a systematic basis over the term of the research collaboration with the development
F-87
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
partner. Research and development funding is recognized as an offset to research and development expenses on a systematic basis over the term of the research collaboration. Contingent revenue attributable to the achievement of development milestones is recognized only on the achievement of the applicable milestone, consideration to be received is fixed or determinable, and collectability is reasonably assured.
Contract revenue
The Company recognizes revenue from research and development contracts as expenses are incurred. Funding received for services to be rendered in the future is reflected as deferred revenue.
Research and development costs
Research and development costs are charged to income as incurred, net of grants or related tax credits, unless they meet the criteria under Canadian GAAP for deferral and amortization. No development costs have been deferred to date.
The Company records investment tax credits when there is reasonable assurance that the assistance will be realized. Government assistance recognized as a credit to research and development costs for the year ended June 30, 2012 was $1,362,147 [2011 - $1,371,702].
Foreign currency translation
Transactions denominated in foreign currencies have been translated into Canadian dollars at the average rates of exchange prevailing at the time of the respective transactions. Monetary assets and liabilities have been translated into Canadian dollars at the year-end exchange rate. All gains and losses are included in the statement of operations and deficit.
Income taxes
Income taxes are recorded using the asset and liability method whereby future tax assets and liabilities are recorded based on the differences between the financial statement carrying amounts of assets and liabilities and their respective tax bases. Future tax assets and liabilities are measured using the substantively enacted tax rates and laws expected to be in effect when the differences are expected to reverse. A valuation allowance is recorded for the portion of the future tax assets where the realization of any value is uncertain.
Stock-based compensation
The Company uses the fair value method of accounting for stock options granted to employees and non-employees. The Company incorporates an expected volatility assumption in fair valuing stock options awarded to employees which is expensed over the vesting period and for non-employees is expensed as the services are received.
B. FIRST-TIME ADOPTION OF ACCOUNTING STANDARDS FOR PRIVATE ENTERPRISES
These financial statements are the first financial statements which the Company has prepared in accordance with Part II of the CICA Handbook – Accounting Standards for Private Enterprises, which constitutes generally
F-88
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
accepted accounting principles for non-publicly accountable enterprises in Canada. In preparing its opening balance sheet as at July 1, 2010, the Company has applied Section 1500, First-time Adoption, retrospectively using the following four principles such that it has:
| • | Recognized all assets and liabilities whose recognition is required by GAAP; |
| • | Not recognized items as assets or liabilities if GAAP does not permit such recognition; |
| • | Reclassified items recognized previously as one type of asset, liability or component of equity, but are now recognized as a different type of asset, liability or component of equity; and |
| • | Applied GAAP in measuring all recognized assets and liabilities. |
The accounting policies that the Company has used in the preparation of its opening balance sheet through application of these principles has not resulted in adjustments to balances which were presented in the balance sheet prepared in accordance with Part V of the CICA Handbook - Accounting XFI [“Previous GAAP”].
Section 1500 provides a number of elective exemptions from the retrospective adoption of GAAP. The Company did not elect to use any of the transitional exemptions.
There are no reconciling items for the deficit as at July 1, 2010 and net loss for the year ended June 30, 2011 and the deficit as presented under Previous GAAP with those computed under GAAP.
3. PROPERTY AND EQUIPMENT
Property and equipment consist of the following:
June 30, 2012 | June 30, 2011 | July 1, 2010 | ||||||||||||||||||||||||||
Cost $ | Accumulated amortization $ | Net $ | Cost $ | Accumulated amortization $ | Net $ | Net $ | ||||||||||||||||||||||
Research equipment | 865,203 | 744,282 | 120,921 | 877,633 | 686,317 | 191,316 | 234,550 | |||||||||||||||||||||
| Computer hardware and software | 70,555 | 61,946 | 8,609 | 117,848 | 107,001 | 10,847 | 13,546 | |||||||||||||||||||||
Office equipment | 38,574 | 33,034 | 5,540 | 38,574 | 31,649 | 6,925 | 8,656 | |||||||||||||||||||||
| Leasehold improvements | 52,452 | 47,702 | 4,750 | 52,452 | 45,327 | 7,125 | 10,687 | |||||||||||||||||||||
| 1,026,784 | 886,964 | 139,820 | 1,086,507 | 870,294 | 216,213 | 267,439 | ||||||||||||||||||||||
During 2012, the Company revised its estimate of the remaining useful life of certain research equipment and computer hardware and recognized $40,977 of additional amortization expense. The Company also wrote off $20,118 of research equipment and $50,754 of computer hardware which was fully amortized and no longer in use. The write-downs have been included in amortization expense on the statements of operations and deficit.
F-89
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
4. TECHNOLOGY RIGHTS
Technology rights consist of the following:
June 30, $ | June 30, $ | July 1, $ | ||||||||||
Cost | 2,469,062 | 2,469,062 | 2,469,062 | |||||||||
Less accumulated amortization | 2,469,062 | 2,381,562 | 2,219,251 | |||||||||
| — | 87,500 | 249,811 | ||||||||||
| [a] | On February 1, 2010, the Company entered into two separate license agreements with the University Health Network and The Hospital For Sick Children to acquire intellectual property relating to the methods and compounds for the modulation of the SIRP -CD47 interaction for use in human hematopoietic stem cell transplantation and therapeutic cancer applications. The Company paid two up-front license fees of $150,000 and committed to pay annual maintenance fees of $25,000 under each agreement in addition to patent issuance, development and regulatory milestones, and royalties on commercial sales. As at June 30, 2012, no future contingent consideration[note 13[a]]had been paid. These technology rights have been fully amortized. |
| [b] | On March 26, 2008, the Company entered into a license agreement with the University of Maryland, Baltimore [“UMB”] wherein UMB granted the Company an exclusive license and non-exclusive license under the UMB patent rights and a non- exclusive sublicense under the Children’s Medical Center Corporation patent rights related to the Company’s TTI-1612 technology. |
The Company has a commitment to pay U.S.$10,000 in annual maintenance fees and there is future contingent consideration in the form of milestone payments and royalties. During the year ended June 30, 2012, the Company paid a $25,000 milestone fee to UMB on the initiation of its Phase I trial for TTI-1612. As at June 30, 2012, no other future contingent consideration[note 13[a]]had been paid. These technology rights have been fully amortized.
| [c] | In December 2005, the Company acquired an exclusive worldwide license from the Children’s Hospital, Inc., Columbus, Ohio to a patent application entitled “Methods of Treating Intestinal Ischemia Using Heparin-Binding Epidermal Growth Factor” [TTI-1612]. The Company has a commitment to pay annual license fees in the range of U.S.$12,500 to U.S.$22,500 during the terms of the agreements and paid U.S.$22,500 in fiscal 2012 and 2011. In addition, there is future contingent consideration in the form of milestone payments and royalties that may be paid by the Company in relation to clinical trial enrollment, regulatory filings and commercial sales. As at June 30, 2012, no future contingent consideration[note 13[a]]had been paid. These technology rights have been fully amortized. |
5. LOAN PAYABLE
On February 1, 2012, the Company entered into a contribution agreement with the Federal Economic Development Agency for Southern Ontario where the Company can receive a repayable contribution of up to
F-90
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
5. LOAN PAYABLE (continued)
$965,000 to support the clinical development of its TTI-1612 program. The period of contribution is up to December 31, 2013. As at June 30, 2012, the Company has repayable contributions of $292,363 representing the outstanding principal balance, repayable in monthly installments of $16,083 for 60 months beginning on December 1, 2014. The loan payable bears no interest.
6. CONVERTIBLE DEBENTURES
Continuity of convertible debentures
Amount $ | ||||
Balance at July 1, 2010 | 3,861,425 | |||
Debt portion of convertible debentures issued | 1,613,911 | |||
Accreted interest | 1,311,257 | |||
Balance at June 30, 2011 | 6,786,593 | |||
Debt portion of convertible debentures issued | 1,293,712 | |||
Accreted interest | 1,629,817 | |||
Balance at June 30, 2012 | 9,710,122 | |||
On September 15, 2011, the Company issued $2,000,000 of 12% convertible debentures with a maturity date of August 31, 2012 and received net proceeds of $1,993,634 after cash issue costs. The debentures plus accrued and unpaid interest are convertible at any time at the option of the holder, and under certain conditions, into Class A preference shares of the Company at the Class A Conversion Price, established by the Company’s shareholders’ agreement, divided by 1.5. Under the terms of the debentures subscription agreement, the Company issued 451,128 Class A preference share purchase warrants entitling the holders to purchase one Class A preference share at a price of $1.33 per share, subject to adjustment at any time up to September 16, 2016.
In three separate transactions in fiscal 2011, the Company issued an aggregate of $3,000,000 of 12% convertible debentures with a maturity date of August 31, 2012 and received net proceeds of $2,947,832 after cash issue costs. Under the terms of the debentures subscription agreement, the Company issued 676,692 Class A preference share purchase warrants entitling the holders to purchase one Class A preference share at a price of $1.33 per share, subject to adjustment at any time up to September 16, 2016.
Consistent with CICA Handbook Part II Section 3856, the debentures were split into separate debt and equity components on the balance sheet. The fair value of the debt, initially determined to be $1,293,712[2011 - $1,613,911] and described as “convertible debentures” on the balance sheet, has been estimated as the present value of future payments based on the Company’s estimated incremental borrowing rates for similar debt agreements. The fair value of the equity portion of the debentures of $699,922 [2011 - $1,333,921] was included in contributed surplus and was estimated using the Black-Scholes option pricing model with the following weighted-average assumptions: risk-free interest rate of 0.9% [2011 - 1.5%]; dividend yield of 0% [2011 - 0%]; expected volatility of 93% [2011 - 90%]; and an expected life of 1.0 year [2011 - 1.8 years]. The warrants were issued for nominal consideration.
F-91
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
6. CONVERTIBLE DEBENTURES (continued)
Subsequent to June 30, 2012, the debenture holders unanimously extended the maturity date of all outstanding convertible debentures to August 31, 2013.
7. PREFERENCE SHARE LIABILITY
The Company is authorized to issue an unlimited number of Class A preference shares. Based on the terms of these shares, the consideration received on their issuance was split into debt and equity components.
Continuity of issued preference shares
Shares # | Amount $ | |||||||
Balance at July 1, 2010, June 30, 2011 and 2012 | 10,150,377 | 13,500,000 | ||||||
The equity component of the issued preference shares of $7,610,125 is included in contributed surplus.
The Class A preference shares are convertible into common shares at the Class A Conversion Price, initially at $1.33 per share, which is subject to adjustment. Each holder of a Class A preference share is entitled to vote for each common share into which the holder’s Class A preference shares are convertible. Dividends on these shares are cumulative at 8% per annum. The cumulative dividends on the Class A preference shares amount to $13,663,608 [2011 - $11,646,743; July 1, 2010 - $9,784,566], none of which has been paid as at June 30, 2012. This amount of cumulative dividends is described as “interest payable” on the balance sheet. Also included in the “interest payable” balance is the Part VI.1 tax liability on these dividends of $5,265,443 [2011 - $4,458,697; July 1, 2010 - $3,713,826] and accrued interest on the debentures of $3,721,147 [2011 - $2,213,869; July 1, 2010 - $1,195,101]. All warrants issued to purchase Class A preference shares were issued for nominal consideration.
The Class A preference shares are redeemable at the option of the holder five years after issue if holders of at least 67% of the then outstanding Class A preference shares request the Company to redeem, at which time all Class A preference shares shall be redeemed at the greater of the original purchase price plus any cumulative dividends and the fair market value of the shares. The Class A preference shareholders do not intend to redeem these shares in the next 12 months.
Upon the closing of a qualified public offering, immediately prior to a qualified acquisition, or upon the approval of holders of 67% of the then outstanding Class A preference shares, all outstanding Class A preference shares shall automatically be converted into common shares.
Warrants
The Company issued Class A preference share purchase warrants in connection with its Class A preference share and convertible debenture financings. Each warrant is exercisable into one Class A preference share at a price of $1.33 per share, subject to adjustment. As at June 30, 2012, there were 5,300,752 [2011 - 4,849,624] warrants issued and outstanding. All warrants expire on September 16, 2016.
F-92
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
8. SHARE CAPITAL Authorized
The Company is authorized to issue an unlimited number of common shares and an unlimited number of Class A preference shares[note 7].
Continuity of issued common shares
Shares # | Amount $ | |||||||
Balance at July 1, 2010, June 30, 2011 and 2012 | 6,116,667 | 1,639,868 | ||||||
9. STOCK-BASED COMPENSATION
| [a] | Stock options |
Under the Company’s stock option plan enacted April 10, 2003, options may be granted to directors, officers, employees and consultants of the Company to purchase up to 2,429,129 common shares. Options are granted at the fair market value of the common shares on the date of grant. Options vest at various rates and have a term not exceeding 10 years.
The Company issued 20,000 [2011 - 20,000] options with an exercise price of $0.43 [2011 - $0.43] per common share during the year ended June 30, 2012. During the year, 20,000 [2011 - nil] options with an exercise price of $0.62 expired.
The following table summarizes information about stock options outstanding as at June 30, 2012:
| Options outstanding | Options exercisable | |||||||||||||||
Exercise price $ | Options outstanding # | Weighted average remaining contractual life [years] | Weighted $ | Options exercisable # | Weighted $ | |||||||||||
0.43 | 165,000 | 7.51 | 0.43 | 101,666 | 0.43 | |||||||||||
0.77 | 1,332,673 | 1.08 | 0.77 | 1,332,673 | 0.77 | |||||||||||
0.83 | 739,000 | 4.48 | 0.83 | 739,000 | 0.83 | |||||||||||
| 2,236,673 | 2.68 | 0.76 | 2,173,339 | 0.77 | ||||||||||||
F-93
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
9. STOCK-BASED COMPENSATION (continued)
| [b] | Stock-based compensation |
During the year ended June 30, 2012, the Company issued options with a fair value of $7,500 [2011 - $7,440] and recorded stock-based compensation expense of $14,117 [2011 - $27,414]. The fair value of the options granted was estimated using the Black-Scholes option pricing model with the following assumptions:
2012 % | 2011 % | |||||||
Risk-free interest rate | 2.1 | 3.4 | ||||||
Dividend yield | 0.0 | 0.0 | ||||||
Expected volatility | 93 | 89 | ||||||
Expected life of options | 10 years | 10 years | ||||||
10. CONTRIBUTED SURPLUS
Continuity of contributed surplus
Amount $ | ||||
Balance at July 1, 2010 | 9,851,922 | |||
Equity portion of convertible debentures issued[note 6] | 1,333,921 | |||
Balance at June 30, 2011 | 11,185,843 | |||
Equity portion of convertible debentures issued[note 6] | 699,922 | |||
Balance at June 30, 2012 | 11,885,765 | |||
11. INCOME TAXES
Income tax recoveries attributable to losses from operations differ from the amounts computed by applying the combined Canadian federal and provincial income tax rates to pre-tax losses from operations primarily as a result of the provision of a valuation allowance on net future income tax benefits.
F-94
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
11. INCOME TAXES (continued)
Significant components of future tax assets are as follows:
June 30, 2012 $ | June 30, 2011 $ | July 1, 2010 $ | ||||||||||
Future tax assets | ||||||||||||
Non-capital losses carried forward | 6,898,398 | 5,641,887 | 4,809,126 | |||||||||
Research and development expenditures | 2,752,162 | 2,330,572 | 2,012,133 | |||||||||
Investment tax credits | 485,532 | 364,020 | 296,811 | |||||||||
| Carrying value of property and equipment and technology rights in excess of accounting basis | 1,000,626 | 861,135 | 769,062 | |||||||||
Ontario harmonization tax credit | 501,594 | 501,594 | 561,786 | |||||||||
Convertible debenture interest not deducted for tax | 986,358 | 553,591 | 298,842 | |||||||||
Debt issuance costs | 17,395 | 25,058 | 21,937 | |||||||||
License fees deductible for tax purposes [previously taxed] | 1,107 | 5,538 | 87,535 | |||||||||
Total future tax assets | 12,643,172 | 10,283,395 | 8,857,232 | |||||||||
Valuation allowance | (12,643,172 | ) | (10,283,395 | ) | (8,857,232 | ) | ||||||
Net future tax assets | — | — | — | |||||||||
The Company has available $10,385,000 of federal research and development expenditures for income tax purposes, which may be carried forward indefinitely to reduce future years’ taxable income, and $10,235,000 of federal non-capital losses available to reduce future years’ taxable income that expire from 2027 to 2032.
F-95
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
11. INCOME TAXES (continued)
The Company also has accumulated unclaimed scientific research and experimental development investment tax credits which can be used to offset future years’ taxable income. The investment tax credits expire as follows:
| $ | ||||
2020 | 32,911 | |||
2021 | 223 | |||
2022 | 7,893 | |||
2023 | 8,819 | |||
2024 | 43,049 | |||
2025 | 81,560 | |||
2026 | 62,285 | |||
2027 | 107,947 | |||
2028 | 52,234 | |||
2029 | 17,460 | |||
2030 | 5,053 | |||
2031 | 1,147 | |||
2032 | 2,248 | |||
| 422,829 | ||||
12. STATEMENTS OF CASH FLOWS
The net change in non-cash working capital balances related to operations consists of the following:
2012 $ | 2011 $ | |||||||
Decrease (increase) in current assets | ||||||||
Accounts receivable | (87,265 | ) | (610 | ) | ||||
Prepaid expenses | (24,183 | ) | 5,378 | |||||
Tax credits receivable | (40,000 | ) | (305,000 | ) | ||||
Contract research deposits | 50,094 | (32,735 | ) | |||||
| (101,354 | ) | (332,967 | ) | |||||
Increase (decrease) in current liabilities | ||||||||
Accounts payable and accrued liabilities | (224,042 | ) | 187,419 | |||||
Deferred revenue | (11,359 | ) | (254,848 | ) | ||||
| (235,401 | ) | (67,429 | ) | |||||
| (336,755 | ) | (400,396 | ) | |||||
F-96
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
13. COMMITMENTS AND CONTINGENCIES
| [a] | The Company enters into research, development and license agreements with various parties in the ordinary course of business whereby the Company receives research services and rights to proprietary technologies[note 4]. The agreements require compensation to be paid by the Company, typically by a combination of the following methods: |
| [i] | Fees comprising amounts due initially upon entering into the agreements as well as additional amounts due either on specified timelines or defined services to be provided. |
| [ii] | Milestone payments that are dependent on products developed under the agreements proceeding towards specified plans of clinical trials and commercial development. |
| [iii] | Royalty payments calculated as a percentage of net sales commencing upon commercial sales of any product candidates developed from the technologies. |
During the year ended June 30, 2012, the Company made sublicense payments under the above agreements in the amount of $127,512 [2011 - $83,114].
As at June 30, 2012, the Company has commitments under agreements to fund research, pre-clinical studies and manufacturing in the amount of $497,211 over the next year [2011 - $1,105,601]. During the year ended June 30, 2012, the Company incurred $250,000 [2011 - $400,000] in expenses to shareholders for research and scientific advisory services and license fees which were recorded at the agreed-upon exchange amount.
Milestone and royalty-related amounts that may become due under various agreements are dependent on, among other factors, pre-clinical safety and efficacy, clinical trials, regulatory approvals and ultimately the successful development of a new drug, the outcome and timing of which are uncertain. Amounts due per the various agreements for milestone payments will be accrued once the occurrence of a milestone is likely. Amounts due as royalty payments will be accrued as commercial revenues from the product are earned.
| [b] | The Company’s future minimum annual commitments under operating leases for premises and equipment are as follows: |
| $ | ||||
2013 | 69,416 | |||
| [c] | The Company enters into license agreements with third parties that include indemnification provisions in the ordinary course of business that are customary in the industry. Those guarantees generally require the Company to compensate the other party for certain damages and costs incurred as a result of third-party claims or damages arising from these transactions. In some cases, the maximum potential amount of future payments that could be required under these indemnification provisions is unlimited. The nature of the indemnification obligations prevents the Company from making a reasonable estimate of the maximum potential amount that it could be required to pay. Historically, the Company has not made any |
F-97
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
13. COMMITMENTS AND CONTINGENCIES (continued)
| indemnification payments under such agreements and no amount has been accrued in the accompanying financial statements with respect to these indemnification obligations. |
14. FINANCIAL INSTRUMENTS
Financial instruments of the Company consist of cash and cash equivalents, marketable securities, accounts receivable, accounts payable and accrued liabilities, and loan payable. As at June 30, 2012, there was no significant difference between the carrying values of the cash and cash equivalents, marketable securities, accounts receivable, and accounts payable and accrued liabilities and their estimated fair values due to their short-term nature. The fair value of the loan payable approximates its carrying value as it is non-interest bearing, consistent with loans of this nature. The Company manages its cash and cash equivalents and marketable securities in accordance with an investment policy that establishes guidelines for investment eligibility, credit quality, liquidity and foreign currency exposure.
| [a] | Credit risk |
Financial instruments that potentially subject the Company to credit risk consist primarily of cash and cash equivalents and marketable securities. The Company manages its exposure to credit risk by placing its cash with major financial institutions and investing in high-quality government and corporate issuers with low credit risk. Cash and cash equivalents held by the Company are not subject to any external restrictions.
| [b] | Liquidity risk |
The Company’s exposure to liquidity risk is dependent on purchasing obligations and the raising of funds to meet commitments and sustain operations. The Company is a development stage company and is reliant on external fundraising to support its operations[note 1]. Once funds have been raised, the Company manages its liquidity risk by investing in highly liquid corporate and government bonds with staggered or short-term maturities to provide regular cash flow for current operations. It also manages liquidity risk by monitoring actual and projected cash flows. The Board of Directors reviews and approves the Company’s operating and capital budgets, as well as any material transactions not in the ordinary course of business. The majority of the Company’s accounts payable and accrued liabilities have maturities of less than three months.
| [c] | Market risk |
Interest rate risk arises due to the Company’s marketable securities bearing fixed interest rates, and foreign exchange risk arises on its holdings of U.S. dollar denominated cash and cash equivalents. The Company manages its interest rate risk by holding its investments to maturity, where possible. The Company manages its exposure to currency fluctuations by holding cash and cash equivalents denominated in U.S. dollars in amounts not exceeding current U.S. dollar financial liabilities and U.S. dollar planned expenditures. As at June 30, 2012, the Company held U.S. dollar cash and cash equivalents in the amount of $38,303.
F-98
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
15. SUBSEQUENT EVENT
Subsequent to June 30, 2012, the debenture holders unanimously extended the maturity date of all outstanding convertible debentures to August 31, 2013[note 1].
16. DIFFERENCES BETWEEN CANADIAN AND UNITED STATES GENERALLY ACCEPTED ACCOUNTING PRINCIPLES
The financial statements have been prepared in accordance with Canadian GAAP, which differ in certain respects from those principles that the Company would have followed if its financial statements had been prepared in accordance with generally accepted accounting principles in the United States (US GAAP).
A reconciliation of net earnings from Canadian GAAP to conform with US GAAP is as follows:
Note
| June 30, 2012 $ | June 30, $ | ||||||||
Net loss for the year based on Canadian GAAP | (8,464,683 | ) | (7,320,872 | ) | ||||||
Acquired research and development | B | 87,500 | 162,311 | |||||||
Preference shares – accrued dividends | C | 2,016,865 | 1,862,177 | |||||||
Convertible debentures – accreted interest expense | D | 1,629,817 | 1,311,257 | |||||||
Convertible debentures – issue costs | D | (30,494 | ) | (21,062 | ) | |||||
Net loss and comprehensive loss for the year based on US GAAP | (4,760,995 | ) | (4,006,189 | ) | ||||||
A reconciliation of shareholders’ deficiency from Canadian GAAP to conform with US GAAP is as follows:
Note
| June 30, $ | June 30, $ | July 1, $ | |||||||||||
Shareholders’ deficiency based on Canadian GAAP | (42,749,984 | ) | (34,999,340 | ) | (29,039,803 | ) | ||||||||
Acquired research and development | B | - | 87,500 | 249,811 | ||||||||||
Preference shares – accreted interest | C | 7,610,125 | 7,610,125 | 7,610,125 | ||||||||||
Preference shares – equity component | C | (7,610,125 | ) | (7,610,125 | ) | (7,610,125 | ) | |||||||
Convertible debentures – accreted interest | D | 4,190,543 | 2,560,727 | 1,249,470 | ||||||||||
Convertible debentures – equity component | D | (4,275,640 | ) | (3,575,719 | ) | (2,241,797 | ) | |||||||
Convertible debentures – issue costs | D | 20,178 | 48,437 | 46,374 | ||||||||||
Shareholders’ deficiency based on US GAAP | (42,814,903 | ) | (35,878,395 | ) | (29,735,945 | ) | ||||||||
F-99
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
16. DIFFERENCES BETWEEN CANADIAN AND UNITED STATES GENERALLY ACCEPTED ACCOUNTING PRINCIPLES (continued)
Reconciling items
A) Government assistance – investment tax credits
Under Canadian GAAP, the Company records its investment tax credits arising from research and development activities on a net basis against costs to which they relate. Under US GAAP, the Company recognizes its investment tax credits as a reduction of tax expense. Accordingly, to reconcile to US GAAP, research and development costs would increase and income tax recovery would increase for the years ended June 30, 2012 and 2011 by $1,362,147 and $1,371,702, respectively. Since both adjustments are on the statement of operations and deficit, there is no impact on net loss.
B) Acquired research and development
Under Canadian GAAP, the technology rights of $0, $87,500 and $249,811 as at June 30, 2012, June 30, 2011 and July 1, 2010, respectively, are acquired research and development assets that have been capitalized on the balance sheet and are being amortized over their estimated useful lives. Under US GAAP, research and development assets acquired without alternative uses are expensed at the time of acquisition, and the amount of technology rights would be nil as at June 30, 2012, June 30, 2011 and July 1, 2010.
As a result of immediately expensing the acquired research and development under US GAAP, the amortization reflected in the statement of operations and deficit for the years ended June 30, 2012 and 2011 would be lower by $87,500 and $162,311, respectively.
C) Preference shares
Under Canadian GAAP, the preference shares are initially bifurcated into debt and equity components. Interest is accreted on the debt component up to the original subscription amount of the shares and both accrued cumulative dividends and Part IV.1 tax are recognized as interest expense and included in interest payable. Since the shares have reached their redemption date, they are recorded at their original subscription amount in the financial statements. Under US GAAP, the preference shares are classified as temporary equity and adjusted to their maximum redemption amount each balance sheet date once they become redeemable. Accrued cumulative dividends are recorded as a charge to equity and included in temporary equity. Part VI.1 tax is recognized as income tax expense and included in taxes payable. Accordingly, to reconcile to US GAAP, (a) the $13,500,000 recorded as a non-current liability under Canadian GAAP as at June 30, 2012, June 30, 2011 and July 1, 2010 (which is currently equal to the shares’ maximum redemption amount) is reclassified to temporary equity, (b) $7,610,125 set up in contributed surplus on the original bifurcation under Canadian GAAP as at June 30, 2012, June 30, 2011 and July 1, 2010 is deducted from shareholders’ deficiency and $7,610,125 of cumulative accreted interest included in the deficit as at June 30, 2012, June 30, 2011 and July 1, 2010 is added back to shareholders’ deficiency, (c) the $13,663,608, $11,646,743, and $9,784,566 of accrued cumulative dividends classified as interest payable under Canadian GAAP is reclassified to temporary equity under US GAAP as at June 30, 2012, June 30, 2011 and July 1, 2010, respectively, and the $2,016,865 and $1,862,177 of accrued cumulative dividends recognized as interest expense for the years ended June 30, 2012 and 2011, respectively, is added back to net loss, and (d) the $5,265,443, $4,458,697, and $3,713,826 of Part VI.1 tax liability classified as
F-100
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
16. DIFFERENCES BETWEEN CANADIAN AND UNITED STATES GENERALLY ACCEPTED ACCOUNTING PRINCIPLES (continued)
interest payable under Canadian GAAP as at June 30, 2012, June 30, 2011 and July 1, 2010, respectively, is reclassified to taxes payable and the $806,746 and $744,871 of Part VI.1 tax recognized as interest expense for the years ended June 30, 2012 and 2011, respectively, is reclassified to income tax expense.
Preference share subscribers also received warrants exercisable into preference shares. Under Canadian GAAP, the Company did not account for the warrants in the financial statements as it was determined that the fair value of the warrants at the time of issuance is nominal. Under US GAAP, the warrants are classified as a liability and carried at the fair value both initially and on an on-going basis. Consistent with Canadian GAAP, the Company determined the initial fair value of the warrants to be nominal. The Company also determined the fair value of the warrants has remained nominal since the time of issuance resulting in no measurement difference between Canadian and US GAAP.
D) Convertible debentures
Under Canadian GAAP, the convertible debentures are initially bifurcated into debt and equity components and interest is accreted on the debt component up to its face amount of the shares. Under US GAAP, the convertible debenture is classified as debt and carried at amortized cost. Accordingly, to reconcile to US GAAP, accreted interest expense recorded of $4,190,543, $2,560,727 and $1,249,470 as at June 30, 2012, June 30, 2011, and July 1, 2010, respectively, must be added back to shareholders’ deficiency and the amount set up in equity on the original bifurcation of the convertible debt under Canadian GAAP needs to be deducted from shareholders’ deficiency and added to the convertible debt liability in the amounts of $4,275,640, $3,575,719 and $2,241,797 as at June 30, 2012, June 30, 2011, and July 1, 2010, respectively.
Similarly, accreted interest expense recorded under Canadian GAAP would need to be added back to the net loss under US GAAP in the amounts of $1,629,817 and $1,311,257 for the years ended June 30, 2012 and 2011, respectively.
There is also a balance sheet classification difference related to the costs incurred to issue the convertible debt. Under Canadian GAAP the costs were allocated to the debt and equity components on initial bifurcation and only the portion allocated to the debt component was subsequently amortized, whereas US GAAP requires all of the costs to be set up as a separate asset and amortized. Accordingly, the issue costs allocated to equity under Canadian GAAP are amortized under US GAAP, resulting in a decrease in net income of $30,494 and $21,062 for the years ended June 30, 2012 and 2011, respectively. Additionally, the total balance of unamortized issue costs is reclassified to deferred financing costs, including the unamortized portion of the issue costs allocated to equity under Canadian GAAP in the amounts of $20,178, $48,437 and $46,374 as at June 30, 2012, June 30, 2011 and July 1, 2010, respectively.
Convertible debt subscribers also received warrants exercisable into preference shares. Under Canadian GAAP, the Company did not account for the warrants in the financial statements as it was determined that the fair value of the warrants at the time of issuance is nominal. Under US GAAP, the warrants are classified as a liability and carried at fair value, both initially and on an on-going basis. Consistent with Canadian GAAP, the Company
F-101
Table of Contents
Trillium Therapeutics Inc.
[A development stage company]
NOTES TO FINANCIAL STATEMENTS
June 30, 2012
16. DIFFERENCES BETWEEN CANADIAN AND UNITED STATES GENERALLY ACCEPTED ACCOUNTING PRINCIPLES (continued)
determined the initial fair value of the warrants to be nominal. The Company also determined that the fair value of the warrants has remained nominal since the time of issuance, resulting in no measurement difference between Canadian and US GAAP
Cash flow
In addition to the differences between Canadian and US GAAP related to the recognition and measurement of transactions by the Company, there are differences in the manner in which items are classified in the statement of cash flows. These classification differences have no impact on cash used in operating, investing and financing activities.
F-102
Table of Contents
Pro formaConsolidated Statement of Loss and Comprehensive Loss
The following table sets forth Trillium Therapeutics Inc.’s (or “Trillium”, formerly Stem Cell Therapeutics Corp.) unauditedpro forma consolidated statement of loss and comprehensive loss for the year ended December 31, 2013 after giving effect to the acquisition of the private company Trillium Therapeutics Inc. (“Trillium Privateco”) as if it had occurred on January 1, 2013 rather than on April 9, 2013.
The unauditedpro forma consolidated statement of loss and comprehensive loss for the year ended December 31, 2013 is not necessarily indicative of the results of operations that would have occurred in the year ended December 31, 2013, had the acquisition been effective on the assumed date, or of the results of operations expected in 2014 and future years.
The unauditedpro forma consolidated statement of loss and comprehensive loss for the year ended December 31, 2013 has been derived from and should be read in conjunction with:
| • | the description of the acquisition of Trillium Privateco contained under the heading “Item 4.A History and Development of the Company” of this registration statement; |
| • | Trillium’s audited annual consolidated financial statements for the year ended December 31, 2013 and 2012 (in the name of Stem Cell Therapeutics Corp.), which are included in this registration statement; |
| • | pro formaadjustments giving effect to the acquisition and the related assumptions as described in the notes to thepro forma consolidated statement of loss and comprehensive loss below. |
The unauditedpro forma consolidated statement of loss and comprehensive loss has been prepared in accordance with International Financial Reporting Standards (“IFRS”) as issued by the International Accounting Standards Board (“IASB”) as described in the audited consolidated financial statements of Trillium for the year ended December 31, 2013 and 2012.
The financial statements of Trillium Privateco were prepared in accordance with Part II of the CICA Handbook - Accounting Standards for Private Enterprises (“ASPE”). These accounting policies were reviewed by management for conformity with IFRS. It was determined that there were measurement differences with respect to share-based compensation, however, as the Trillium Privateco stock options were cancelled at the time of the acquisition, there was no impact on conversion to IFRS on a consolidated basis.
The accounting policies used in the preparation of the unauditedpro forma consolidated statement of loss and comprehensive loss are those set out in the Trillium audited consolidated financial statements as at and for the year ended December 31, 2013.
F-103
Table of Contents
UnauditedPro FormaConsolidated Statement of Loss and Comprehensive Loss
For the year ended December 31, 2013
All amounts are expressed in Canadian dollars unless otherwise indicated.
Trillium $ | Trillium $ | Pro forma Adjustments | Note
| Pro forma Consolidated $ | ||||||||||||||||
EXPENSES | ||||||||||||||||||||
Research and development | 3,336,706 | 810,422 | 88,907 | i,ii,iii | 4,236,035 | |||||||||||||||
General and administrative | 962,200 | 212,032 | (68,835) | iii,iv | 1,105,397 | |||||||||||||||
Operating expenses | 4,298,906 | 1,022,454 | 20,072 | 5,341,432 | ||||||||||||||||
Finance income | (54,028) | (5,895) | - | (59,923) | ||||||||||||||||
Finance costs | 44,430 | 972,035 | (970,442) | v | 46,023 | |||||||||||||||
Net finance (income) costs | (9,598) | 966,140 | (970,442) | (13,900) | ||||||||||||||||
Net loss and comprehensive loss | 4,289,308 | 1,988,594 | (950,370) | 5,327,532 | ||||||||||||||||
Basic and diluted loss per common share | (3.16) | (3.77) | ||||||||||||||||||
The unauditedpro forma consolidated statement of loss and comprehensive loss reflects the following adjustments as if the acquisition had occurred on January 1, 2013:
| (i) | On applying the acquisition method of accounting, the purchase consideration of $2,850,836 exceeded the net tangible assets acquired of Trillium Privateco in the amount of $1,018,037 which was attributed to the licensed patent rights. Amortization has been recognized on a straight-line basis over the estimated useful lives of the intangible assets estimated at three years. For thepro formaconsolidated statement of loss and comprehensive loss an additional 99 days of amortization have been reflected in research and development expenses for the period January 1, 2013 to April 9, 2013 in the amount of $92,042; |
| (ii) | A reduction in the net book value of Trillium Privateco property and equipment to the estimated fair value of the assets of $91,000 at January 1, 2013 results in lower depreciation expense on Trillium Privateco assets of $2,844 and is reflected in research and development expenses; |
| (iii) | On acquisition all previously issued stock options of Trillium Privateco were cancelled and accordingly the share-based compensation expenses recorded are deducted in the amount of $291 from research and development expenses and $1,585 from general and administrative expenses; |
| (iv) | Legal costs in the amount of $67,250 related to the acquisition recorded in the Trillium Privateco statement of operations for the period from January 1, 2013 to April 9, 2013 were deducted from general and administrative expenses; |
| (v) | On the elimination of the equity and debt securities of Trillium Privateco on consolidation, the interest on convertible debt and accrued dividends on the preference shares are eliminated for the period from January 1, 2013 to April 9, 2013 in the aggregate amount of $970,442; |
| (vi) | Pro forma loss per share has been calculated using the weighted average number of shares that would have been outstanding for the year ended December 31, 2013, after giving effect to the acquisition as if it had occurred on January 1, 2013. Thepro formaweighted average number of common shares outstanding is 1,411,336. Thepro forma loss per share and weighted average number of common shares incorporate the impact of the reverse split in November 2014. |
F-104
Table of Contents
US$50,000,000

COMMON SHARES
SERIES II FIRST PREFERRED SHARES
Prospectus
| Joint Book-Running Managers | ||
| Leerink Partners | Cowen and Company | |
| Co-Manager | ||
| Oppenheimer & Co. | ||
, 2015
Table of Contents
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 6. Indemnification of Directors and Officers
Directors’ and officers’ liability insurance has been purchased for the benefit of the directors and officers of the registrant, to back up the registrant’s indemnification of them against liability incurred in their capacity as directors and officers, subject to certain limitations under applicable law.
In accordance with the provisions of the OBCA, the by–laws of the registrant also provide that the registrant will indemnify a director or officer, a former director or officer, or an individual who acts or acted at the registrant’s request as a director or officer or an individual acting in a similar capacity of another entity, and such person’s heirs and legal representatives, against all costs, charges and expenses, including an amount paid to settle an action or satisfy a judgment, reasonably incurred by such individual in respect of any civil, criminal, administrative investigative or other proceeding in which the individual is involved because of that association with the registrant or other entity, provided however that the registrant shall not so indemnify an individual unless the individual (i) acted honestly and in good faith with a view to the best interests of the registrant or, as the case may be, to the best interests of the other entity for which the individual acted as a director or officer or in a similar capacity at the registrant’s request, and (ii) if the matter is a criminal or administrative action or proceeding that is enforced by a monetary penalty, had reasonable grounds for believing that the individual’s conduct was lawful.
In addition, the registrant may advance money to a director, officer or other individual for the costs, charges and expenses of a proceeding referred to above, but the individual shall repay the registrant if the individual does not fulfil the conditions set out in (i) and (ii) above.
If the registrant becomes liable under the terms of its by–laws, the insurance coverage discussed above will extend to its liability; however, each claim will be subject to a per claim retention of nil or $15,000, depending on the nature of the claim.
Item 7. Recent Sales of Unregistered Securities
See “Description of Share Capital—Share Capital—Issuances of Securities” for the sales of all securities by the registrant since January 1, 2012 and up to the date of this registrant statement which were not registered under the Securities Act.
Item 8. Exhibits and Financial Statement Schedules.
a. See the Exhibit Index to this registration statement.
b. Financial statement schedules.
All financial statement schedules have been omitted because either they are not required, are not applicable or the information required therein is otherwise set forth in the registrant’s financial statements and related notes thereto.
Item 9. Undertakings.
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the undersigned registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act of 1933 and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the
II-1
Table of Contents
registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act of 1933 and will be governed by the final adjudication of such issue.
The undersigned registrant hereby undertakes that:
1. For purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective.
2. For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
II-2
Table of Contents
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the registrant certifies that it has reasonable grounds to believe that it meets all of the requirements for filing on Form F-1 and has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Toronto, Province of Ontario on March 30, 2015.
| TRILLIUM THERAPEUTICS INC. | ||
By:
| /s/ Niclas Stiernholm
| |
Name: Niclas Stiernholm | ||
| Title: President and Chief Executive Officer | ||
Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities and on the dates indicated.
| Signature | Title | Date | ||
/s/ Niclas Stiernholm | President and Chief Executive | March 30, 2015 | ||
| Niclas Stiernholm | Officer (principal executive officer), Director | |||
/s/ James Parsons | Chief Financial Officer (principal | March 30, 2015 | ||
| James Parsons | financial and accounting officer) | |||
* | Director | March 30, 2015 | ||
| Calvin Stiller | ||||
* | Director | March 30, 2015 | ||
| Luke Beshar | ||||
* | Director | March 30, 2015 | ||
| Henry Friesen | ||||
* | Director | March 30, 2015 | ||
| Robert Kirkman | ||||
* | Director | March 30, 2015 | ||
| Michael Moore | ||||
* | Director | March 30, 2015 | ||
Thomas Reynolds | ||||
| *By: | /s/ James Parsons | |
| James Parsons | ||
| Attorney-in-fact | ||
II-3
Table of Contents
AUTHORIZED REPRESENTATIVE
Pursuant to the requirements of Section 6(a) of the Securities Act of 1933, the undersigned has signed this Registration Statement, solely in the capacity of the duly authorized representative of Trillium Therapeutics Inc. in the United States, on March 30, 2015.
| PUGLISI & ASSOCIATES | ||
By:
| /s/ Donald J. Puglisi
| |
Name: Donald J. Puglisi | ||
| Title: Managing Director | ||
II-4
Table of Contents
EXHIBIT INDEX
Exhibit | Description
| |
| 1.1 | Form of underwriting agreement | |
| 3.1 | Articles of Incorporation dated March 31, 2004 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 3.2 | Articles of Amendment dated October 19, 2004 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 3.3 | Articles of Amendment dated February 6, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 3.4 | Articles of Continuance dated November 7, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 3.5 | Articles of Amendment dated December 12, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 3.6 | Articles of Amalgamation dated June 1, 2014 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 3.7 | By-law No. 1 of Trillium Therapeutics Inc. amended and restated as of May 27, 2014 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 3.8 | Articles of Amendment dated November 14, 2014 (incorporated by reference to the registrant’s registration statement on Form 20-F (Amendment No. 2) filed with the SEC on November 26, 2014) | |
| 3.9 | Form of Articles of Amendment | |
| 4.1 | Rights Agreement between Trillium Therapeutics Inc. and Computershare Investor Services Inc. dated September 16, 2013 and amended on June 3, 2014, including the form of rights certificate (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 4.2 | Form of Subscription Agreement between Stem Cell Therapeutics Corp. and U.S. purchasers who acquired common share units and preferred share units in December 2013 | |
| 5.1 | Opinion of Borden Ladner Gervais LLP regarding validity of the securities offered | |
| 8.1 | Opinion of Dorsey & Whitney LLP as to certain United States tax matters | |
| 8.2 | Opinion of Borden Ladner Gervais LLP as to certain Canadian tax matters | |
| 10.1 | Amended and Restated License Agreement between Trillium Privateco, the University Health Network and The Hospital for Sick Children effective February 1, 2010 and amended June 1, 2012 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 10.2 | Debenture Purchase Agreement and Merger Agreement among Stem Cell Therapeutics Corp., Trillium Privateco, 2364556 Ontario Limited, and the Trillium Privateco debenture holders dated March 25, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 10.3* | GPEx®-Derived Cell Line Sale Agreement between Trillium Therapeutics Inc. and Catalent Pharma Solutions, LLC dated August 12, 2014 for TTI-621 (incorporated by reference to the registrant’s registration statement on Form 20-F (Amendment No. 1) filed with the SEC on October 3, 2014) | |
| 10.4* | GPEx -Derived Cell Line Sale Agreement between Trillium Therapeutics Inc. and Catalent Pharma Solutions, LLC dated August 12, 2014 for TTI-622 (incorporated by reference to the registrant’s registration statement on Form 20-F (Amendment No. 1) filed with the SEC on October 3, 2014) | |
| 10.5# | 2014 Stock Option Plan (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 10.6# | 2014 Deferred Share Unit Plan (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 10.7 | Warrant Indenture between Stem Cell Therapeutics Corp. and Computershare Trust Company of Canada dated March 15, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) |
Table of Contents
Exhibit | Description
| |
| 10.8 | Warrant Indenture between Stem Cell Therapeutics Corp. and Computershare Trust Company of Canada dated April 8, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 10.9 | Warrant Indenture between Stem Cell Therapeutics Corp. and Computershare Trust Company of Canada dated December 13, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 10.10 | Agency Agreement among Stem Cell Therapeutics Corp. and Bloom Burton & Co. Inc. and Roth Capital Partners, LLC dated December 13, 2013 (incorporated by reference to the registrant’s registration statement on Form 20-F filed with the SEC on August 12, 2014) | |
| 10.11# | Form of Executive Employment Agreement between Trillium Therapeutics Inc. and Dr. Niclas Stiernholm effective June 3, 2014 | |
| 10.12# | Form of Executive Employment Agreement between Trillium Therapeutics Inc. and Dr. Robert Uger effective June 3, 2014 | |
| 10.13# | Form of Executive Employment Agreement between Trillium Therapeutics Inc. and James Parsons effective June 3, 2014 | |
| 10.14# | Form of Executive Employment Agreement between the Trillium Therapeutics USA Inc. and Dr. Eric Sievers effective April 1, 2015 | |
| 10.15# | Form of Executive Employment Agreement between Trillium Therapeutics Inc. and Dr. Penka Petrova effective June 3, 2014 | |
| 23.1 | Consent of Ernst & Young LLP | |
| 23.2 | Consent of Ernst & Young LLP | |
| 23.3 | Consent of Dorsey & Whitney LLP (included in Exhibit 8.1) | |
| 23.4 | Consent of Borden Ladner Gervais LLP (included in Exhibit 5.1 and Exhibit 8.2) | |
| 24.1† | Powers of Attorney |
* Portions of this exhibit have been omitted and filed separately with the Securities and Exchange Commission pursuant to a confidential treatment request.
# Denotes management compensation plan or contract.
† Previously Filed.