Forward-Looking Statements 2 © 2019 Zynerba Pharmaceuticals, Inc. All rights reserved. Zynerba and Zygel are trademarks of Zynerba Pharmaceuticals, Inc. All other trademarks and registered trademarks are property of their respective owners. trademarks are property of their respective owners. THE STATEMENTS IN THIS PRESENTATION MAY INCLUDE FORWARD-LOOKING STATEMENTS WITHIN THE MEANING OF THE PRIVATE SECURITIES LITIGATION REFORM ACT OF 1995. THESE STATEMENTS, AMONG OTHER THINGS RELATE TO THE FUTURE OPERATIONS, OPPORTUNITIES OR FINANCIAL PERFORMANCE OF ZYNERBA PHARMACEUTICALS, INC. WE MAY, IN SOME CASES, USE TERMS SUCH AS “PREDICTS,” “BELIEVES,” “POTENTIAL,” “PROPOSED,” “CONTINUE,” “ESTIMATES,” “ANTICIPATES,” “EXPECTS,” “PLANS,” “INTENDS,” “MAY,” “COULD,” “MIGHT,” “WILL,” “SHOULD” OR OTHER WORDS THAT CONVEY UNCERTAINTY OF FUTURE EVENTS OR OUTCOMES TO IDENTIFY THESE FORWARD-LOOKING STATEMENTS. SUCH STATEMENTS ARE SUBJECT TO NUMEROUS IMPORTANT FACTORS, RISKS AND UNCERTAINTIES THAT MAY CAUSE ACTUAL EVENTS OR RESULTS TO DIFFER MATERIALLY FROM THE COMPANY’S CURRENT EXPECTATIONS, INCLUDING THE FOLLOWING: THE COMPANY’S CASH AND CASH EQUIVALENTS MAY NOT BE SUFFICIENT TO SUPPORT ITS OPERATING PLAN FOR AS LONG AS ANTICIPATED; THE RESULTS, COST AND TIMING OF THE COMPANY’S CLINICAL DEVELOPMENT PROGRAMS, INCLUDING ANY DELAYS TO SUCH CLINICAL TRIALS RELATING TO ENROLLMENT OR SITE INITIATION; CLINICAL RESULTS FOR THE COMPANY’S PRODUCT CANDIDATES MAY NOT BE REPLICATED OR CONTINUE TO OCCUR IN ADDITIONAL TRIALS AND MAY NOT OTHERWISE SUPPORT FURTHER DEVELOPMENT IN A SPECIFIED INDICATION OR AT ALL; ACTIONS OR ADVICE OF THE U.S. FOOD AND DRUG ADMINISTRATION AND FOREIGN REGULATORY AGENCIES MAY AFFECT THE DESIGN, INITIATION, TIMING, CONTINUATION AND/OR PROGRESS OF CLINICAL TRIALS OR RESULT IN THE NEED FOR ADDITIONAL CLINICAL TRIALS; THE COMPANY’S ABILITY TO OBTAIN AND MAINTAIN REGULATORY APPROVAL FOR ITS PRODUCT CANDIDATES, AND THE LABELING UNDER ANY SUCH APPROVAL; AND THE COMPANY’S EXPECTATIONS REGARDING ITS ABILITY TO OBTAIN AND ADEQUATELY MAINTAIN SUFFICIENT INTELLECTUAL PROPERTY PROTECTION FOR ITS PRODUCT CANDIDATES. THESE AND OTHER RISKS ARE DESCRIBED IN OUR FILINGS WITH THE SECURITIES AND EXCHANGE COMMISSION, AVAILABLE AT WWW.SEC.GOV. ANY FORWARD-LOOKING STATEMENTS THAT THE COMPANY MAKES IN THIS PRESENTATION SPEAK ONLY AS OF THE DATE OF THIS PRESENTATION. THE COMPANY ASSUMES NO OBLIGATION TO UPDATE FORWARD-LOOKING STATEMENTS WHETHER AS A RESULT OF NEW INFORMATION, FUTURE EVENTS OR OTHERWISE, AFTER THE DATE OF THIS PRESENTATION.

Zynerba Pharmaceuticals (NASDAQ: ZYNE) 3 A Rare/Near-Rare Neuropsychiatric Company Deep pipeline focused on high unmet medical needs; translating into multi-billion dollar market opportunity with Zygel™ Four clinical shots on goal: FXS, DEE, ASD, 22q Experienced team Proven development and commercialization track record in transdermal delivery, orphan diseases, neurology, psychiatry Well capitalized Cash runway expected into the second half of 2021 - beyond the expected NDA filing and potential approval in FXS Multiple expected near term milestones

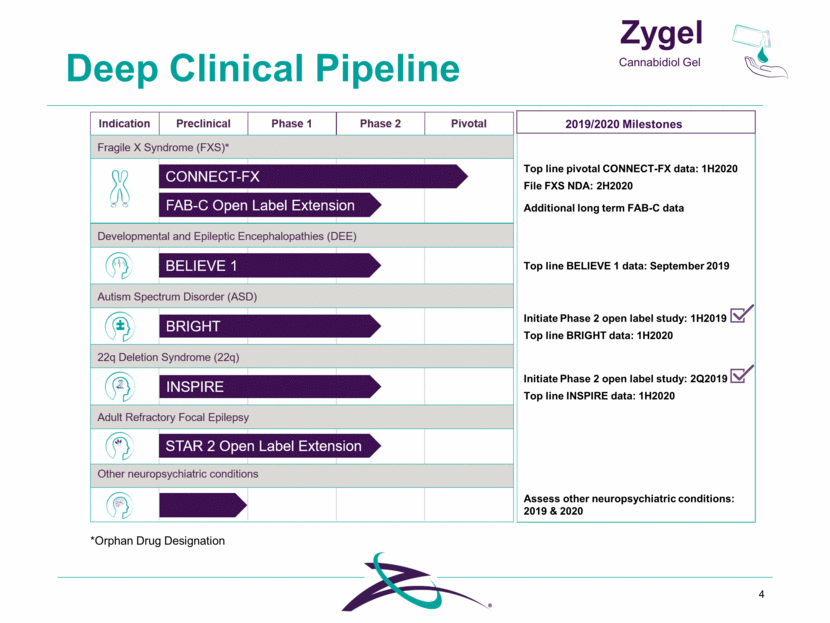
Deep Clinical Pipeline 4 *Orphan Drug Designation Zygel Cannabidiol Gel 2019/2020 Milestones Top line pivotal CONNECT-FX data: 1H2020 File FXS NDA: 2H2020 Additional long term FAB-C data Initiate Phase 2 open label study: 1H2019 Top line BRIGHT data: 1H2020 Initiate Phase 2 open label study: 2Q2019 Top line INSPIRE data: 1H2020 Top line BELIEVE 1 data: September 2019 Assess other neuropsychiatric conditions: 2019 & 2020
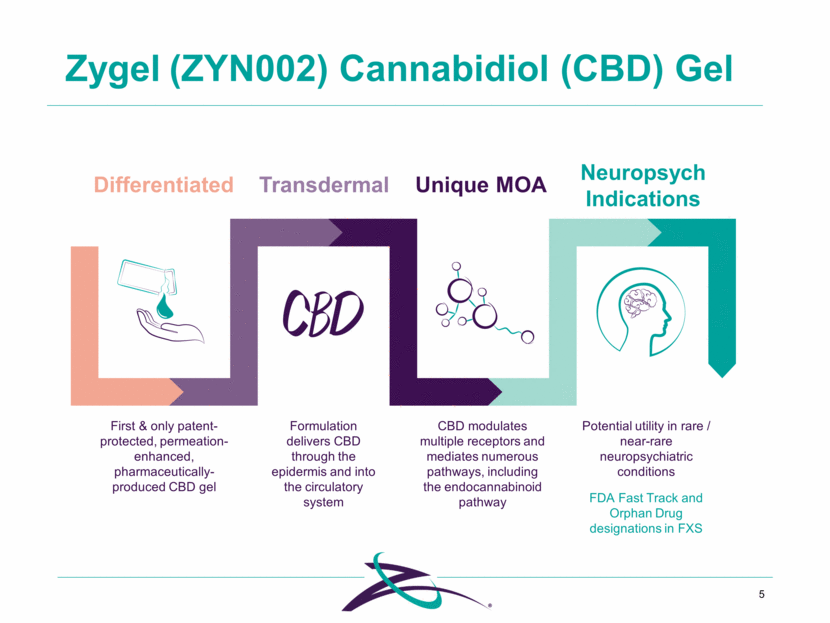
Neuropsych Indications Differentiated Formulation delivers CBD through the epidermis and into the circulatory system Zygel (ZYN002) Cannabidiol (CBD) Gel 5 Unique MOA First & only patent-protected, permeation-enhanced, pharmaceutically- produced CBD gel Transdermal CBD modulates multiple receptors and mediates numerous pathways, including the endocannabinoid pathway Potential utility in rare / near-rare neuropsychiatric conditions FDA Fast Track and Orphan Drug designations in FXS
Fragile X Syndrome (FXS) 6

Fragile X Syndrome (FXS) Overview 7 Rare genetic developmental disability Leading known cause of both inherited intellectual disability and autism spectrum disorder Symptoms linked to deficiencies in the endocannabinoid (EC) system System of neurotransmitters regulating emotional responses, behavioral reactivity to context, social interaction FMR1 mutation causes dysregulation of the EC system Results in core cognitive, social, and behavioral symptoms of FXS CBD may modulate EC system Increases availability of endocannabinoids (anandamide, 2-AG) by inhibiting metabolism Affects ~71K people in U.S. No approved drugs indicated for FXS

Recent Development Progress in FXS 8 Received FDA Fast Track designation for treatment of behavioral symptoms associated with FXS Recently issued US patent directed to methods of treating FXS with cannabidiol extends IP protection to 2038 Presented 12-month FAB-C open label Phase 2 data at American Psychiatric Association meeting (APA; May 2019) Statistical improvement from baseline in FXS phenotypic behaviors including social avoidance, anxiety, and irritability Three month improvements sustained through 12 months of treatment Excellent tolerability profile Enrollment ongoing in CONNECT-FX: a pivotal trial in pediatric and adolescent patients with FXS Top line results expected in 1H2020

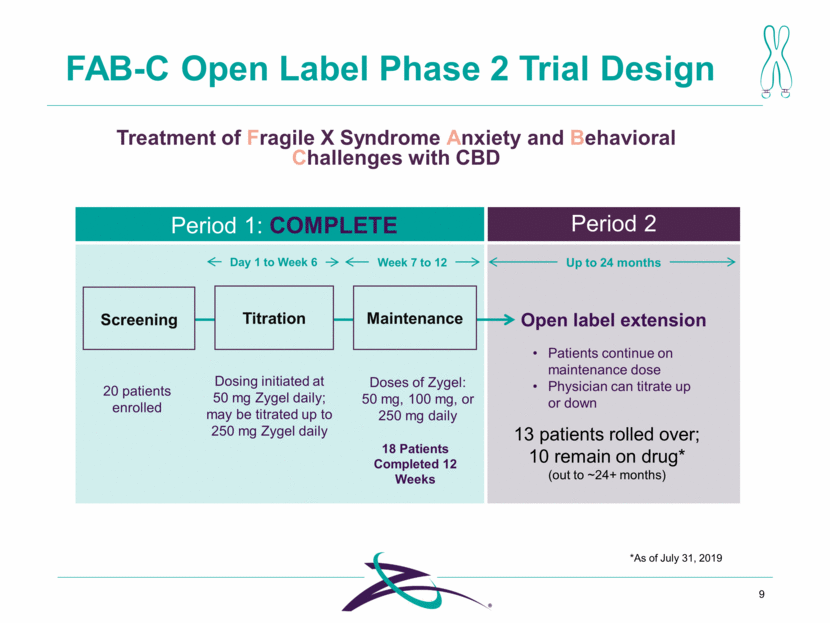
Day 1 to Week 6 Week 7 to 12 Up to 24 months FAB-C Open Label Phase 2 Trial Design 9 Treatment of Fragile X Syndrome Anxiety and Behavioral Challenges with CBD Titration Maintenance Screening Open label extension Dosing initiated at 50 mg Zygel daily; may be titrated up to 250 mg Zygel daily Doses of Zygel: 50 mg, 100 mg, or 250 mg daily 20 patients enrolled Patients continue on maintenance dose Physician can titrate up or down 18 Patients Completed 12 Weeks 13 patients rolled over; 10 remain on drug* (out to ~24+ months) Period 1: COMPLETE Period 2 *As of July 31, 2019
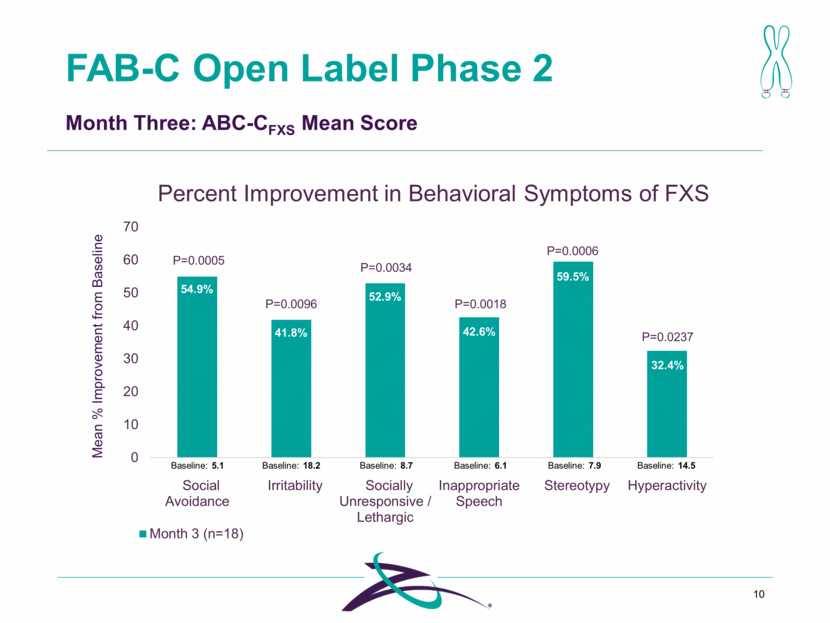
FAB-C Open Label Phase 2 10 Baseline: 18.2 Baseline: 14.5 Baseline: 8.7 Baseline: 5.1 Baseline: 7.9 Baseline: 6.1 41.8% 52.9% 54.9% 32.4% 59.5% 42.6% P=0.0005 P=0.0096 P=0.0034 P=0.0018 P=0.0006 P=0.0237 Mean % Improvement from Baseline Month Three: ABC-CFXS Mean Score Percent Improvement in Behavioral Symptoms of FXS 0 10 20 30 40 50 60 70 Social Avoidance Irritability Socially Unresponsive / Lethargic Inappropriate Speech Stereotypy Hyperactivity Month 3 (n=18)
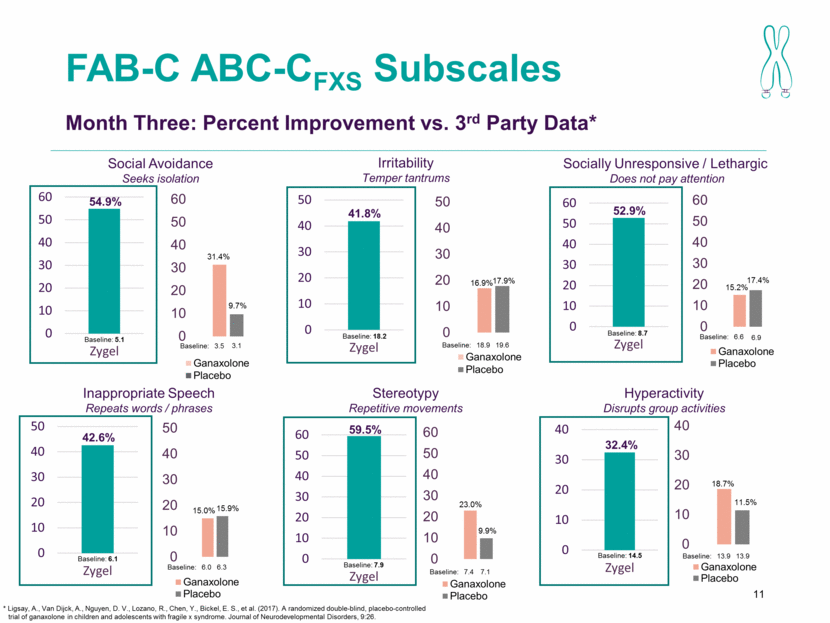
FAB-C ABC-CFXS Subscales 11 Month Three: Percent Improvement vs. 3rd Party Data* Socially Unresponsive / Lethargic Does not pay attention Inappropriate Speech Repeats words / phrases Stereotypy Repetitive movements Hyperactivity Disrupts group activities Social Avoidance Seeks isolation Irritability Temper tantrums Baseline: 18.2 Baseline: 18.9 Baseline: 14.5 Baseline: 13.9 Baseline: 8.7 Baseline: 6.6 Baseline: 5.1 Baseline: 3.5 Baseline: 7.9 Baseline: 7.4 Baseline: 6.1 Baseline: 6.0 3.1 19.6 13.9 6.9 7.1 6.3 41.8% 17.9% 16.9% 32.4% 11.5% 18.7% 52.9% 17.4% 15.2% 54.9% 9.7% 31.4% 59.5% 9.9% 23.0% 42.6% 15.9% 15.0% * Ligsay, A., Van Dijck, A., Nguyen, D. V., Lozano, R., Chen, Y., Bickel, E. S., et al. (2017). A randomized double-blind, placebo-controlled trial of ganaxolone in children and adolescents with fragile x syndrome. Journal of Neurodevelopmental Disorders, 9:26. Baseline: 18.9 0 10 20 30 40 50 60 Zygel 0 10 20 30 40 50 Zygel 0 10 20 30 40 50 60 Zygel 0 10 20 30 40 50 Zygel 0 10 20 30 40 50 60 Zygel 0 10 20 30 40 Zygel 0 10 20 30 40 50 60 Ganaxolone Placebo 0 10 20 30 40 50 60 Ganaxolone Placebo 0 10 20 30 40 50 Ganaxolone Placebo 0 10 20 30 40 50 60 Ganaxolone Placebo 0 10 20 30 40 Ganaxolone Placebo 0 10 20 30 40 50 Ganaxolone Placebo
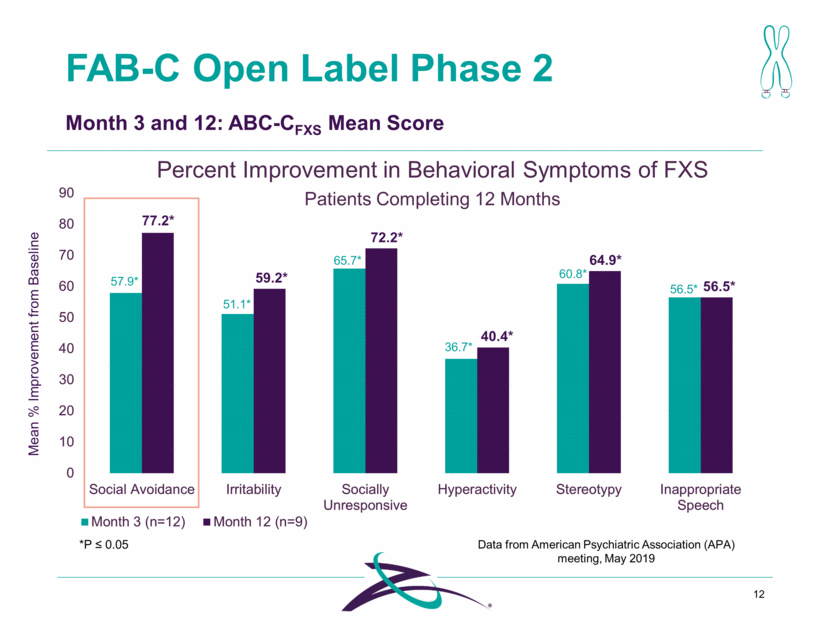
FAB-C Open Label Phase 2 12 Month 3 and 12: ABC-CFXS Mean Score 57.9* 51.1* 65.7* 36.7* 60.8* 56.5* *P 0.05 Percent Improvement in Behavioral Symptoms of FXS Mean % Improvement from Baseline 77.2* 59.2* 72.2* 40.4* 64.9* 56.5* Patients Completing 12 Months Data from American Psychiatric Association (APA) meeting, May 2019 0 10 20 30 40 50 60 70 80 90 Social Avoidance Irritability Socially Unresponsive Hyperactivity Stereotypy Inappropriate Speech Month 3 (n=12) Month 12 (n=9)
FAB-C Open Label Phase 2 13 Well tolerated, consistent with previously reported data; no SAEs No clinically meaningful trends in vital signs, ECG, or clinical safety labs including LFTs; no THC detected in plasma Discontinuations Two siblings discontinued in Period 1 One for worsening of pre-existing eczema (not considered Tx-related) One due to administrative reasons Three patients discontinued in Period 2 (administrative reasons; non-compliance) Little to no redness at application site One patient developed moderate application site rash (resolved, did not recur); remains in the study TEAEs mild or moderate Most common: Gastroenteritis (14%), URTI (12%) All resolved during study period Zygel Safety Summary Through 12 Months

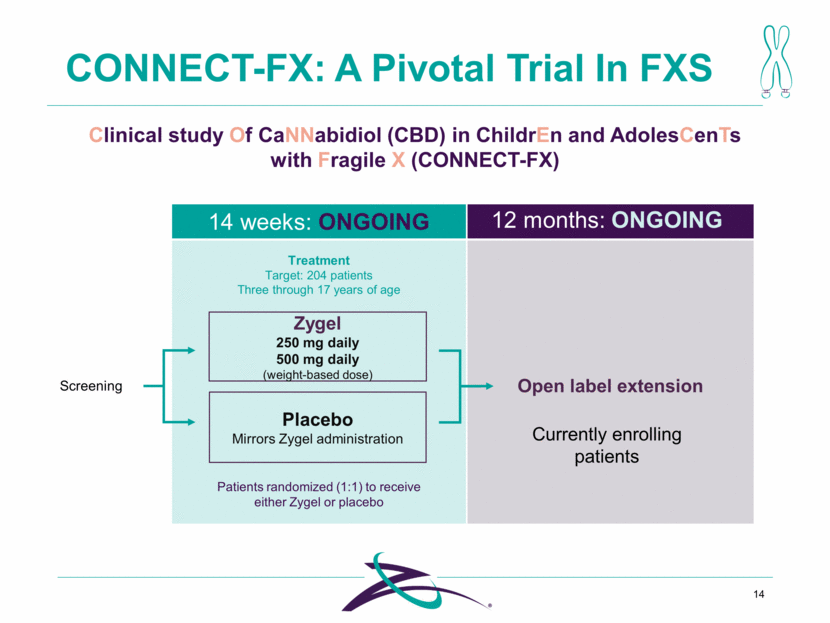
CONNECT-FX: A Pivotal Trial In FXS 14 Clinical study Of CaNNabidiol (CBD) in ChildrEn and AdolesCenTs with Fragile X (CONNECT-FX) Zygel 250 mg daily 500 mg daily (weight-based dose) Placebo Mirrors Zygel administration Patients randomized (1:1) to receive either Zygel or placebo Treatment Target: 204 patients Three through 17 years of age Screening Open label extension 14 weeks: ONGOING 12 months: ONGOING Currently enrolling patients
CONNECT-FX: A Pivotal Trial In FXS 15 Primary endpoint: Change from baseline to end of treatment in ABC-CFXS Social Avoidance subscale Key secondary endpoints: Change from baseline to end of the treatment in ABC-CFXS Irritability subscale score ABC-CFXS Socially Unresponsive/Lethargic subscale score Improvement in CGI-I (anchored to FXS behaviors) at end of treatment Aligned with FDA’s ‘Voice of the Patient’ Guidance Capturing qualitative data on clinical relevance of FXS behaviors

CONNECT-FX 16 With positive results, Zynerba intends to request a meeting with the FDA to: Determine acceptability of data as basis for NDA filing Seek advice on marketing authorization preparation Zynerba believes indication may include the treatment of behavioral symptoms associated with FXS Evaluating opportunities for FDA breakthrough status and/or priority review Top Line Results Expected in 1H2020

DEE Developmental and Epileptic Encephalopathies 17

DEE Overview 18 Doose Syndrome Dravet Syndrome Early Myoclonic Encephalopathy Juvenile Myoclonic Epilepsy (JME) Landau-Kleffner Syndrome Lennox-Gastaut Syndrome (LGS) Ohtahara Syndrome West Syndrome / Infantile Spasms Heterogeneous group of rare / ultra rare epilepsy syndromes Severe cognitive impairment and behavioral disturbances Affects ~45K U.S. children & adolescents Syndromes involve: Impaired development (developmental encephalopathies) Regression of developmental progress (epileptic encephalopathies) Often progressive; highly resistant to treatment Improved seizure control may positively impact development and quality of life DEE includes syndromes such as:

Developing Zygel in DEE 19 Enrollment Complete in BELIEVE 1 Trial Compelling rationale for utility of CBD in DEE Third party clinical data show impact of CBD on seizures and behavioral issues in children Patient enrollment in BELIEVE 1 Phase 2 study complete Six month multi-dose study in DEE patients (3 through 17 years) Being conducted in Australia and New Zealand Inclusion criteria require 5 generalized motor seizures during baseline ~27% have Dravet or LGS Primary efficacy assessment: change in seizure frequency Top line results expected in September 2019

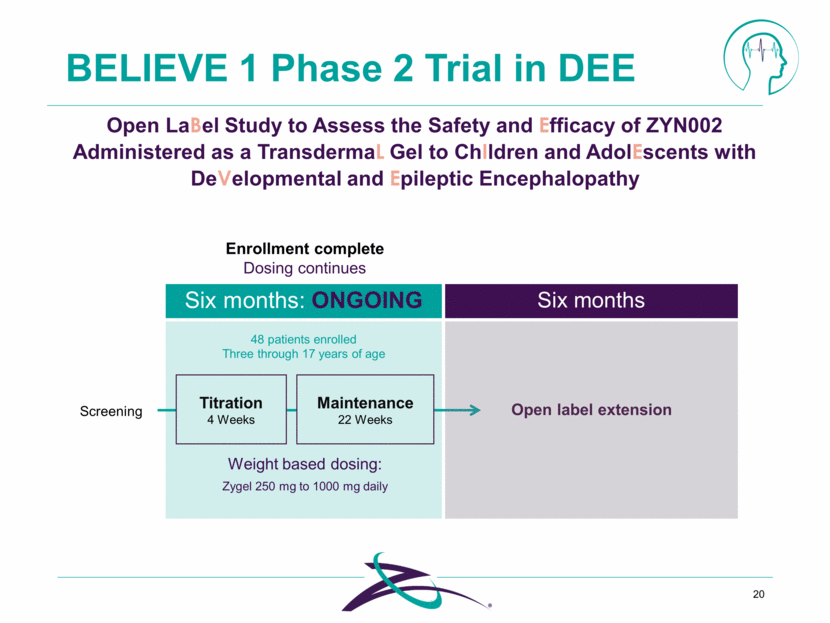
BELIEVE 1 Phase 2 Trial in DEE 20 Open LaBel Study to Assess the Safety and Efficacy of ZYN002 Administered as a TransdermaL Gel to ChIldren and AdolEscents with DeVelopmental and Epileptic Encephalopathy 48 patients enrolled Three through 17 years of age Screening Maintenance 22 Weeks Titration 4 Weeks Open label extension Weight based dosing: Six months: ONGOING Six months Zygel 250 mg to 1000 mg daily Enrollment complete Dosing continues
21 Autism Spectrum Disorder (ASD) in pediatric patients

ASD in Pediatrics Overview 22 Near-rare disorder affecting <1MM pediatric and adolescent pts DSM-5 diagnosis Includes Autistic disorder, Asperger’s syndrome, and Pervasive Development Disorder-not otherwise specified (PDD-NOS) Symptoms include Anxiety Restricted, repetitive patterns of behavior Impairments in social communication Deficits in verbal and non-verbal communication Deficits in developing, understanding and maintaining relationships Most diagnosed after age 4; can be diagnosed as early as age 2 Significant unmet medical need Accelerating rate of diagnosis but only two FDA approved products Both atypical antipsychotics have significant side effect profile Neither approved to address the key symptoms of social impairment and anxiety

Developing Zygel in ASD 23 Newer studies suggest ASD is linked to disruption in the EC system Altered anandamide signaling may contribute to ASD-related social and communication impairments EC system modulates many cellular functions and molecular pathways altered in ASD: imbalanced GABAergic, glutamatergic transmission, oxidative stress, immune dysregulation and altered energy metabolism Clinical and anecdotal data show improvement in social avoidance and anxiety in children with CBD CBD may modulate the EC system and improve certain autism-related behaviors Recent US patent directed to methods of treating ASD with synthetic cannabidiol provides IP protection to 2038 Phase 2 study underway in pediatric and adolescent patients with ASD Top line results expected in 1H2020

BRIGHT Phase 2 Trial in ASD 24 Open-LaBel ToleRabIlity and Efficacy Study of ZYN002 Administered as a Transdermal Gel to CHildren and AdolescenTs with Autism Spectrum Disorder Target: 36 patients Four through 17 years of age Screening Zygel 14 Weeks Weight based dosing: 14 Weeks: ONGOING 250 mg to 500 mg daily Enrollment ongoing Follow up assessments Week 16 / 17 Efficacy assessments (week 14 vs baseline) include: Aberrant Behavior Checklist Parent Rated Anxiety Scale – Autism Spectrum Disorder Autism Impact Measure Clinical Global Impression – Severity and Improvement

22q11.2 Deletion Syndrome (22q) 25

22q Overview 26 Most common contiguous gene deletion syndrome Rare disorder: ~81K patients in US Midline condition with abnormalities affecting palate, face, heart and other organs; surgically corrected in infancy Neuropsychiatric illnesses (anxiety disorders, ASD) and learning disabilities common and impactful 22q associated with increased anxiety, withdrawn behavior and social interaction problems Early onset of neuropsychiatric symptoms disrupts development and QOL, and heightens risk of later psychotic disorders 25-fold increased risk of developing schizophrenia vs. 1% lifetime risk in general population

22q Patient Management 27 Two primary stages of 22q patient management: During infancy, doctors address acute physical concerns, such as anomalies of heart and palate, with surgery Once the physical concerns are stabilized, focus shifts to managing neuropsychiatric symptoms, such as anxiety and autistic behaviors No approved drugs indicated for 22q

Developing Zygel in 22q 28 CBD may treat neuropsychiatric symptoms in 22q due to activity as: Agonist at serotonin 1A receptors Antagonist at GPR55 receptors Modulator of endocannabinoid system Early control of anxiety may delay the development of psychosis Phase 2 study underway in pediatric and adolescent patients with 22q Top line results expected in 1H2020

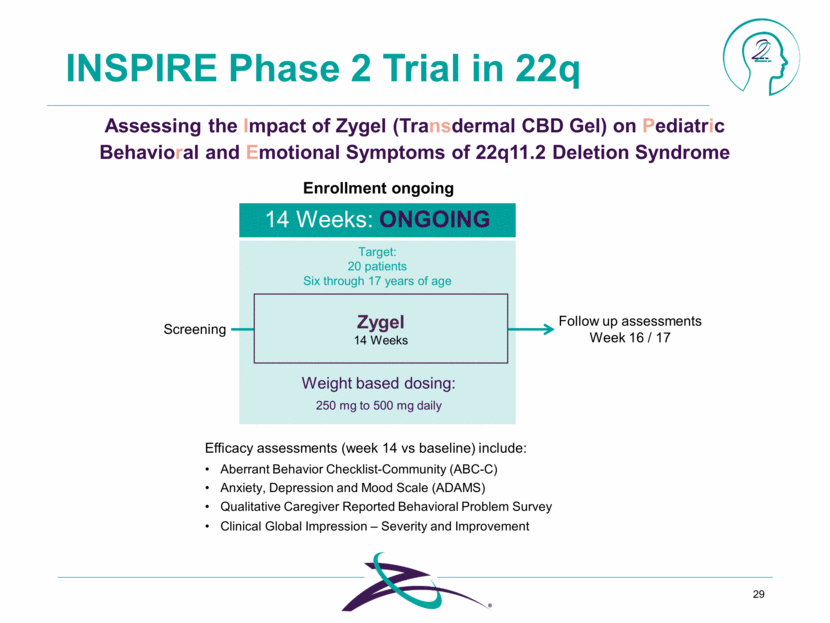
INSPIRE Phase 2 Trial in 22q 29 Target: 20 patients Six through 17 years of age Screening Zygel 14 Weeks Weight based dosing: 14 Weeks: ONGOING 250 mg to 500 mg daily Enrollment ongoing Follow up assessments Week 16 / 17 Efficacy assessments (week 14 vs baseline) include: Aberrant Behavior Checklist-Community (ABC-C) Anxiety, Depression and Mood Scale (ADAMS) Qualitative Caregiver Reported Behavioral Problem Survey Clinical Global Impression – Severity and Improvement Assessing the Impact of Zygel (Transdermal CBD Gel) on Pediatric Behavioral and Emotional Symptoms of 22q11.2 Deletion Syndrome
Financial Strength 30 Clean balance sheet No debt, 23.2M shares outstanding (as of August 6, 2019) Cash and cash equivalent position of $88.7M as of June 30, 2019 Includes net proceeds of $27.0M from 2.1M shares sold and issued at a weighted average selling price of $13.50 per share during the second quarter of 2019 under ATM Advance Overseas Finding expected to generate an incremental $7.0 to $9.0 million over the next 18 to 24 months Cash expected to be sufficient to fund operations and capital requirements into the second half of 2021 - beyond the expected NDA submission and potential approval in FXS

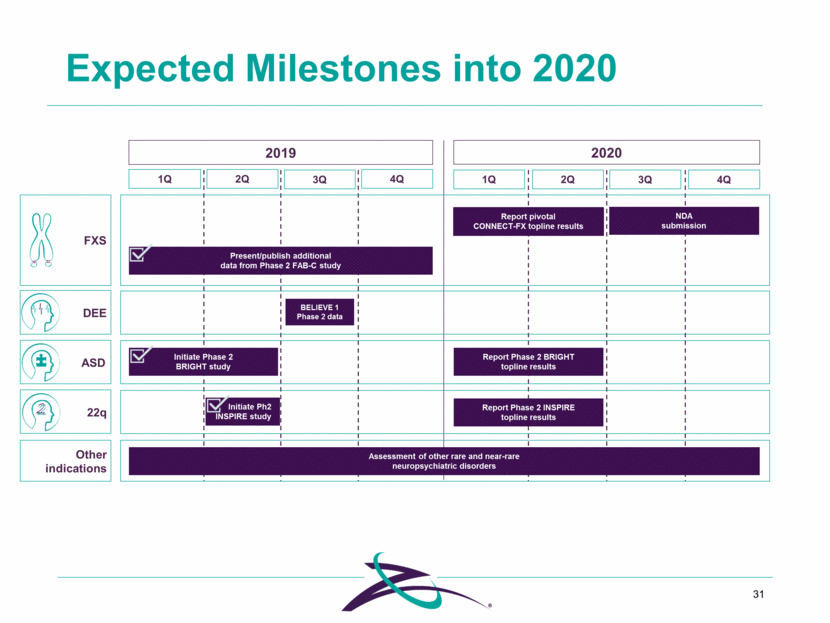
Expected Milestones into 2020 31 FXS DEE Other indications 1Q 2Q 3Q 4Q 2019 Present/publish additional data from Phase 2 FAB-C study Report pivotal CONNECT-FX topline results ASD Initiate Phase 2 BRIGHT study 22q Initiate Ph2 INSPIRE study BELIEVE 1 Phase 2 data 1Q 2Q 2020 Report Phase 2 BRIGHT topline results Report Phase 2 INSPIRE topline results Assessment of other rare and near-rare neuropsychiatric disorders 3Q 4Q NDA submission
Corporate Overview August 2019
