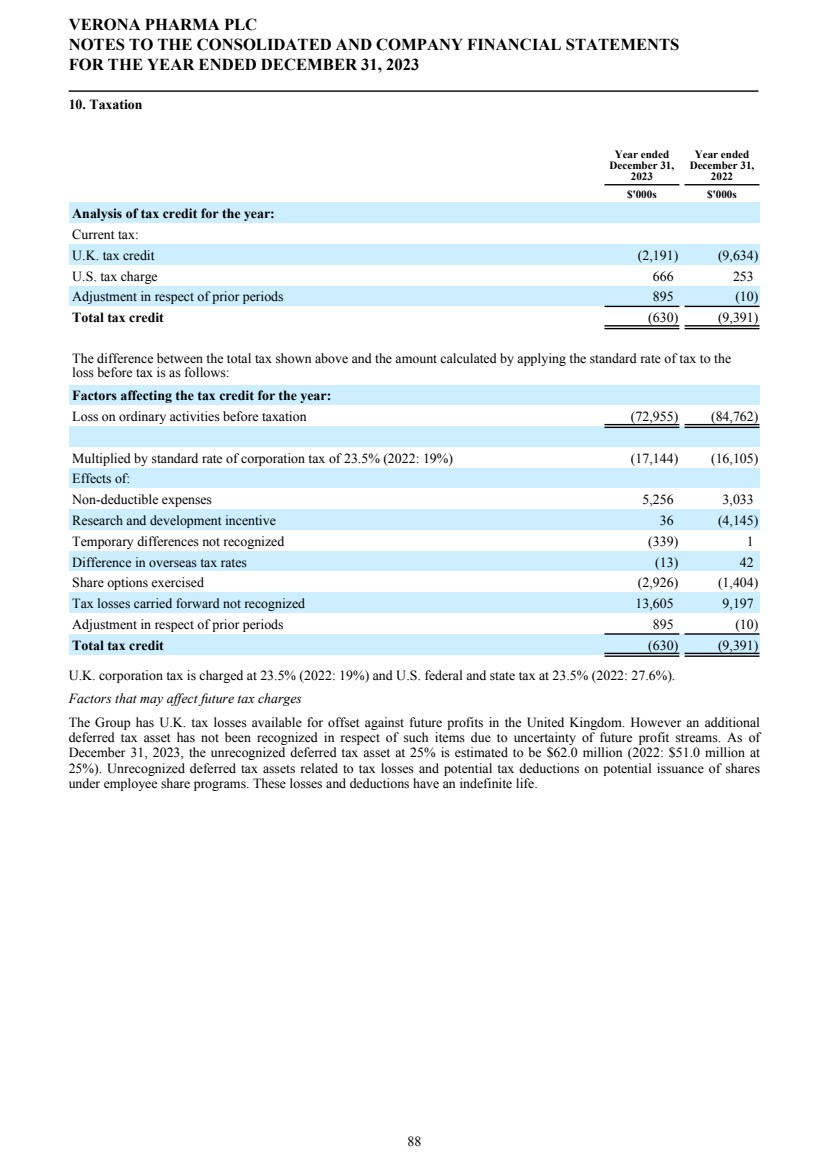
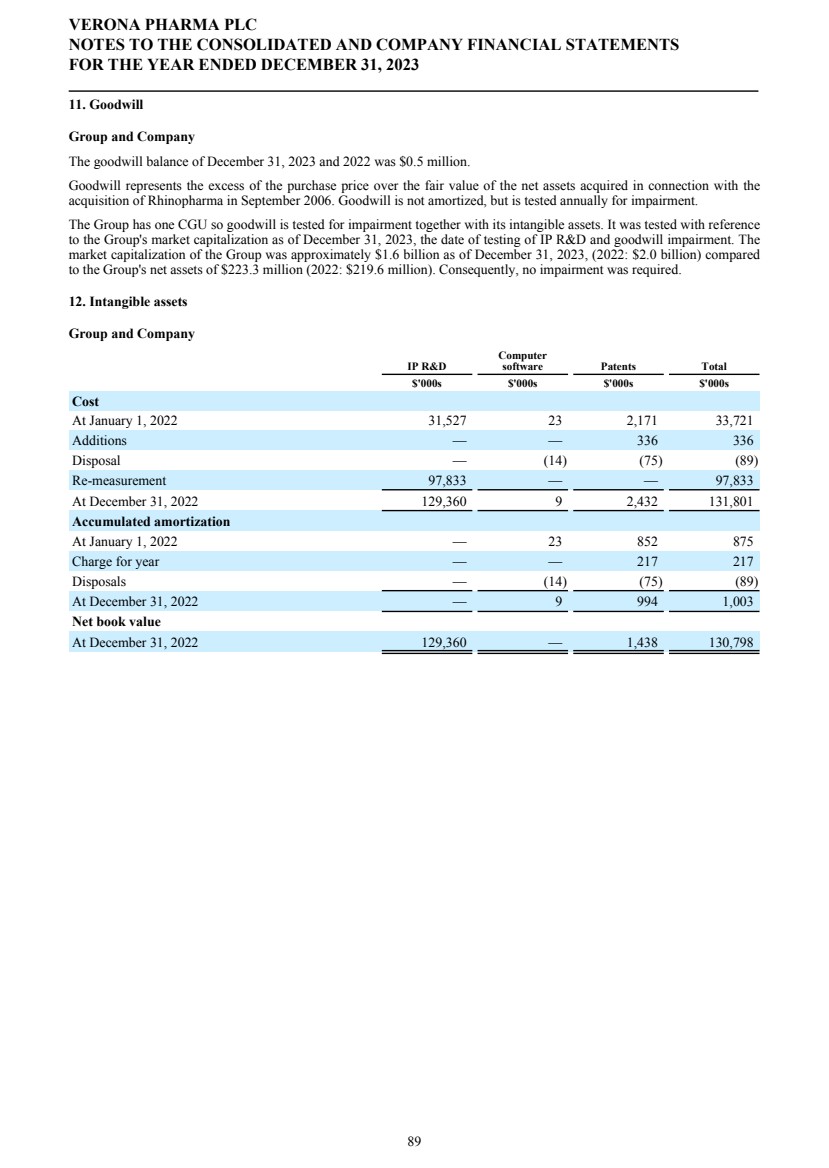
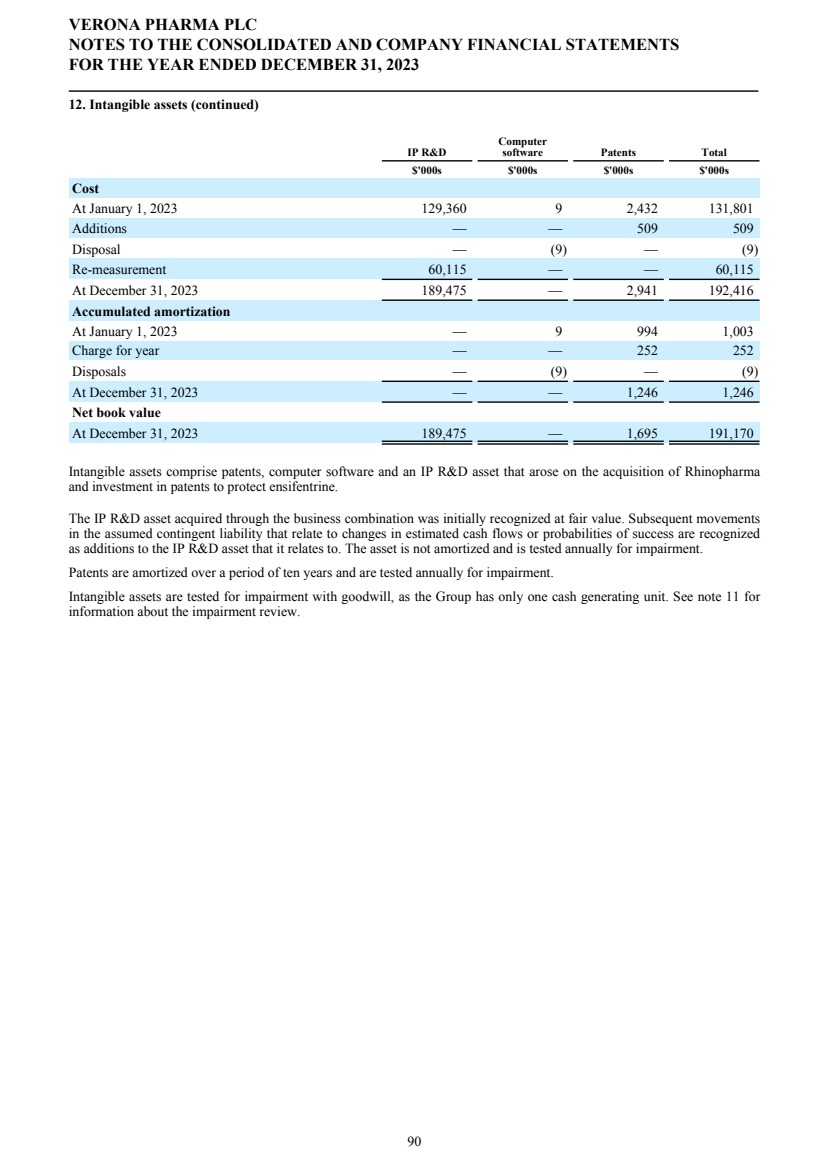
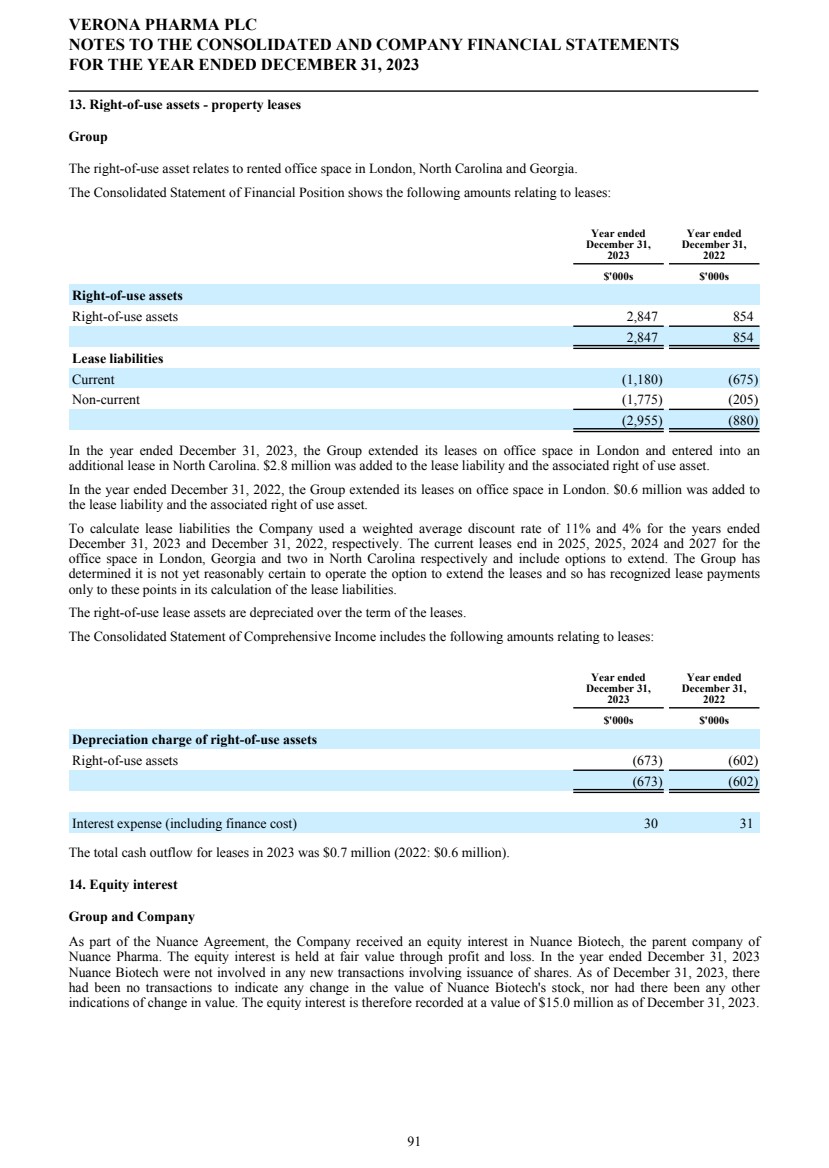
SECURITIES AND EXCHANGE COMMISSION
SCHEDULE 14A INFORMATION
Securities Exchange Act of 1934 (Amendment No. )
of Shareholders
ONE CENTRAL SQUARE
CARDIFF, CF10 1FS
UNITED KINGDOM
![[MISSING IMAGE: lg_veronapharma-4c.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/lg_veronapharma-4c.jpg)
Incorporated in England and Wales with registered no. 5375156
![[MISSING IMAGE: lg_veronapharma-4c.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/lg_veronapharma-4c.jpg)
Incorporated in England and Wales with registered no. 5375156
TO BE HELD ON FRIDAY, APRIL 26, 2024
| | By order of the Board: Ben Harber Company Secretary March 20, 2024 | | | Registered Office: One Central Square Cardiff, CF10 1FS, U.K. | |
| | | | Page | | |||
| | | | | 1 | | | |
| | | | | 2 | | | |
| | | | | 8 | | | |
| | | | | 8 | | | |
| | | | | 8 | | | |
| | | | | 9 | | | |
| | | | | 9 | | | |
| | | | | 10 | | | |
| | | | | 10 | | | |
| | | | | 13 | | | |
| | | | | 14 | | | |
| | | | | 14 | | | |
| | | | | 15 | | | |
| | | | | 15 | | | |
| | | | | 16 | | | |
| | | | | 18 | | | |
| | | | | 19 | | | |
| | | | | 20 | | | |
| | | | | 20 | | | |
| | | | | 22 | | | |
| | | | | 22 | | | |
| | | | | 22 | | | |
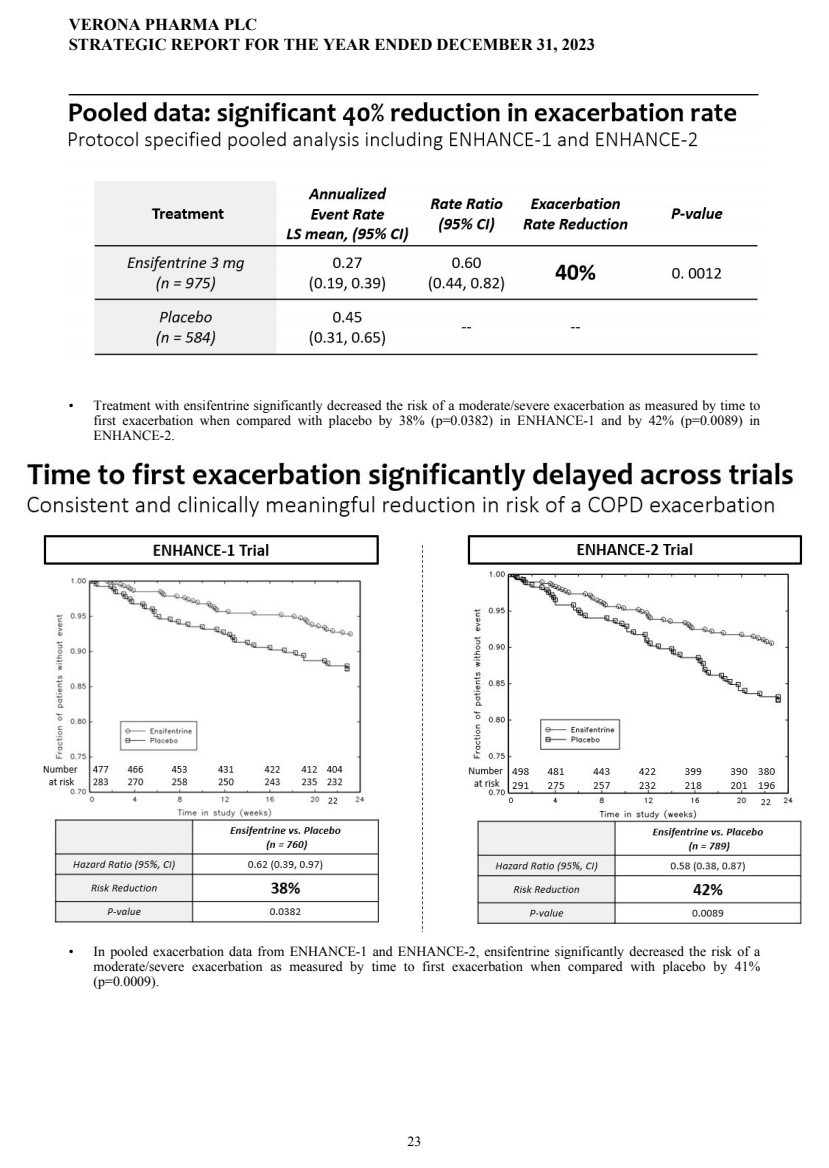
| | | | | 23 | | | |
| | | | | 23 | | | |
| | | | | 23 | | | |
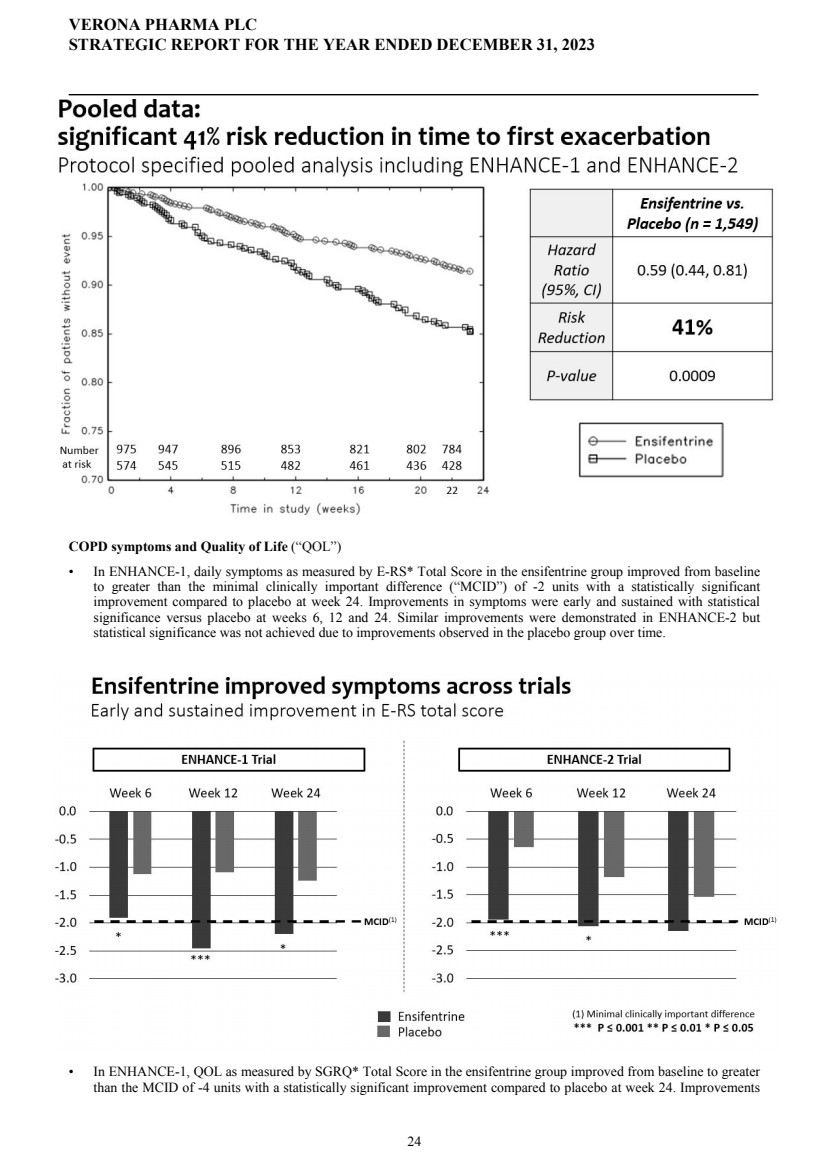
| | | | | 24 | | | |
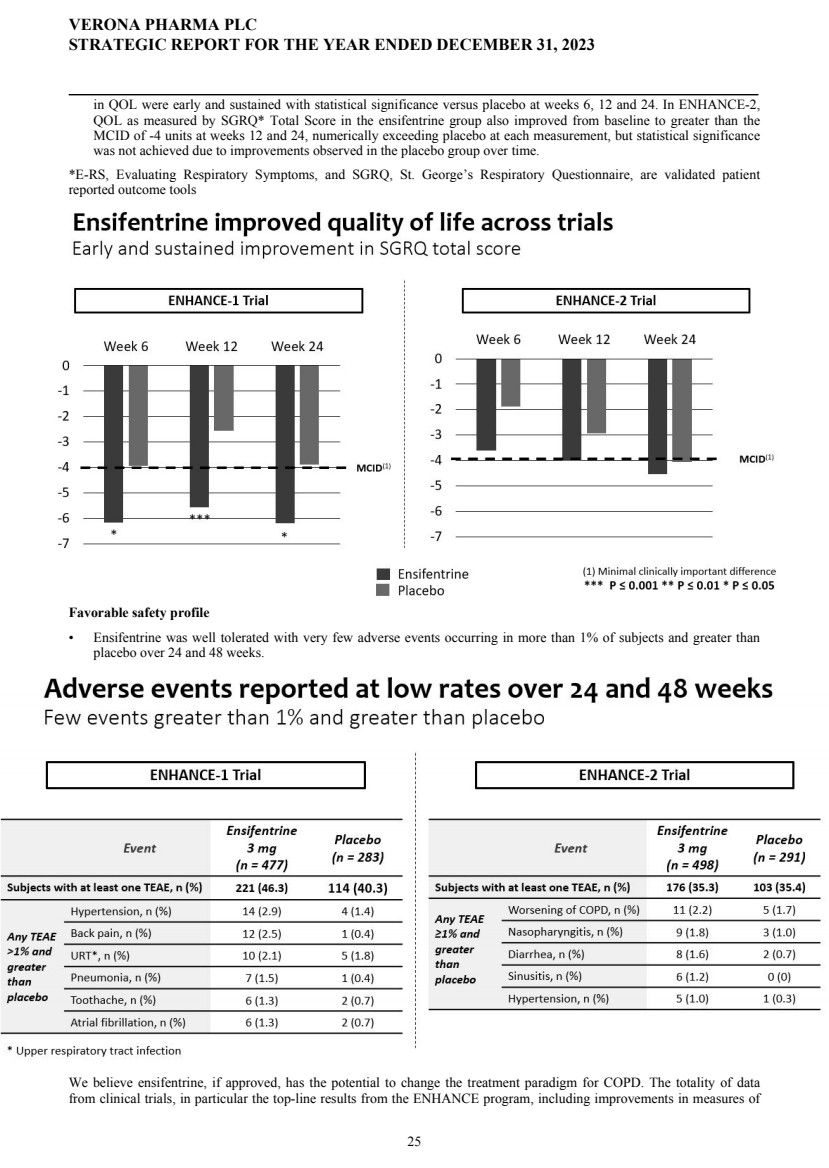
| | | | | 25 | | | |
| | | | | 25 | | | |
| | | | | 25 | | | |
| | | | | 26 | | | |
| | | | | 26 | | | |
| | | | | 26 | | | |
| | | | | 27 | | | |
| | | | | 28 | | | |
| | | | | 28 | | | |
| | | | | 29 | | | |
| | | | | 29 | | | |
| | | | | 31 | | | |
| | | | | 36 | | | |
| | | | | 38 | | | |
| | | | Page | | |||
| | | | | 38 | | | |
| | | | | 41 | | | |
| | | | | 41 | | | |
| | | | | 41 | | | |
| | | | | 42 | | | |
| | | | | 42 | | | |
| | | | | 43 | | | |
| | | | | 43 | | | |
| | | | | 43 | | | |
| | | | | 43 | | | |
| | | | | 43 | | | |
| | | | | 44 | | | |
| | | | | 44 | | | |
| | | | | 44 | | | |
| | | | | | | ||
One Central Square
Cardiff, CF10 1FS
United Kingdom
| Proposal | | | Description of Proposal | | | Board’s Recommendation | |
| 1 | | | To re-elect Ms. Lisa Deschamps as a director of the Company | | | FOR | |
| 2 | | | To re-elect Dr. Martin Edwards as a director of the Company | | | FOR | |
| 3 | | | To re-elect Dr. Sven Anders Ullman as a director of the Company | | | FOR | |
| 4 | | | To re-elect Ms. Christina Ackermann as a director of the Company | | | FOR | |
| 5 | | | To re-elect Mr. Michael Austwick as a director of the Company | | | FOR | |
| 6 | | | To receive and adopt the 2023 U.K. Annual Report | | | FOR | |
| 7 | | | To receive and approve, as a non-binding advisory resolution, the U.K. statutory Directors’ Remuneration Report | | | FOR | |
| 8 | | | To receive and approve the U.K. Directors’ Remuneration Policy | | | FOR | |
| 9 | | | To approve an increase in the maximum aggregate level of Non-Executive Director annual remuneration to £750,000 | | | FOR | |
| 10 | | | To re-appoint EY as the Company’s auditors, to hold office until the conclusion of the next annual general meeting of shareholders | | | FOR | |
| 11 | | | To authorize the Audit and Risk Committee to determine the auditors’ remuneration for the year ending December 31, 2024 | | | FOR | |
| 12 | | | To approve, on an advisory (non-binding) basis, the compensation of the Company’s named executive officers | | | FOR | |
| 13 | | | To authorize the Board of Directors to exercise all the powers of the Company to allot shares in the Company or grant rights to subscribe for or to convert any security into shares in the Company up to an aggregate nominal amount of £8,345,745 (being up to a maximum of 166,914,908 shares representing 25% of the Company’s existing ordinary share capital as at the close of business on March 13, 2024 (being the latest practicable date prior to publication of this document)) | | | FOR | |
| 14 | | | To authorize the Board of Directors to allot equity securities for cash and/or to sell ordinary shares held by the Company as treasury shares for cash as if Section 561(1) of the Companies Act does not apply to any such allotment | | | FOR | |
To re-elect Lisa Deschamps as a Director of the Company
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the re-election of Lisa Deschamps as a Director of the Company | |
To re-elect Martin Edwards as a Director of the Company
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the re-election of Martin Edwards, M.D. as a Director of the Company | |
To re-elect Sven Anders Ullman as a Director of the Company
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the re-election of Sven Anders Ullman, M.D., PhD., as a Director of the Company | |
To re-elect Christina Ackermann as a Director of the Company
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the re-election of Christina Ackermann as a Director of the Company | |
To re-elect Michael Austwick as a Director of the Company
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the re-election of Michael Austwick as a Director of the Company | |
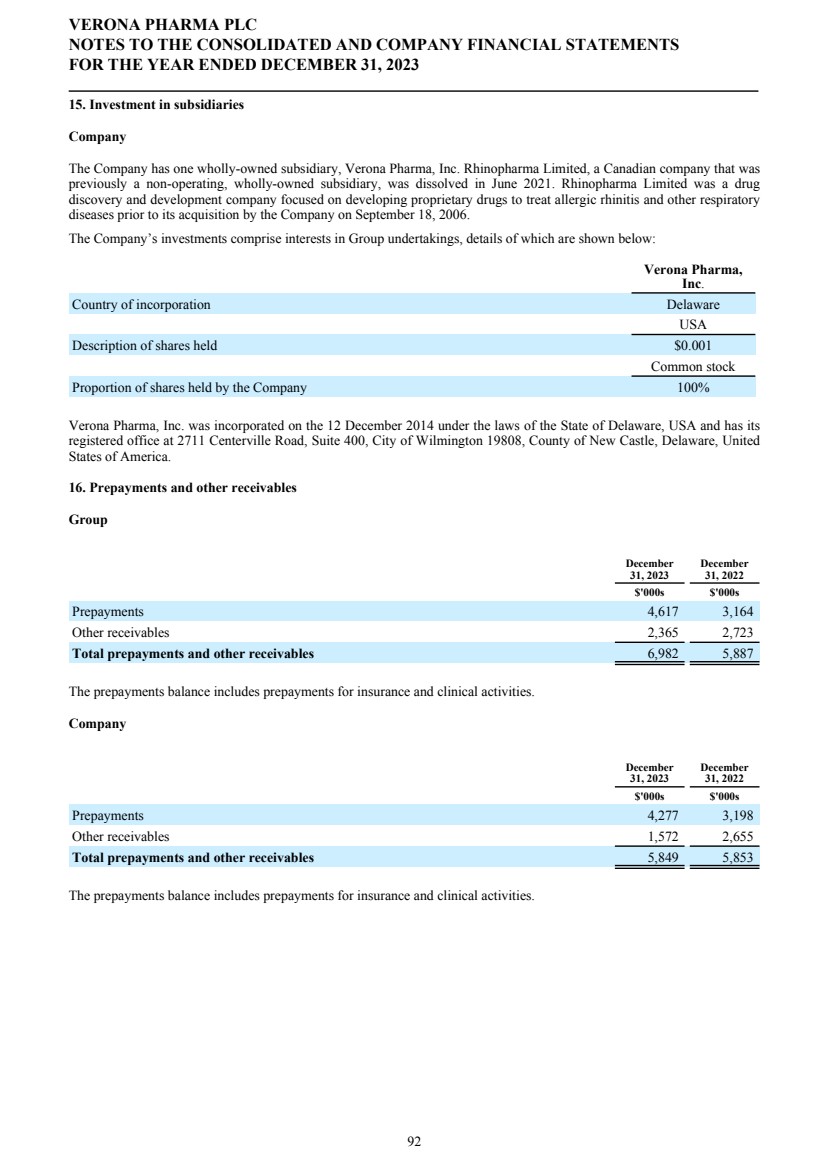
| Name | | | Age | | | Director Since | | | Position(s) | |
| David Zaccardelli, Pharm.D. | | | 59 | | | 2020 | | | Chief Executive Officer and Director | |
| David Ebsworth, Ph.D. | | | 69 | | | 2014 | | | Non-Executive Chairperson of the Board of Directors | |
| Christina Ackermann | | | 59 | | | 2023 | | | Non-Executive Director | |
| Michael Austwick | | | 54 | | | 2024 | | | Non-Executive Director | |
| James Brady | | | 61 | | | 2022 | | | Non-Executive Director | |
| Ken Cunningham, M.D. | | | 71 | | | 2015 | | | Non-Executive Director | |
| Lisa Deschamps | | | 52 | | | 2021 | | | Non-Executive Director | |
| Martin Edwards, M.D. | | | 68 | | | 2019 | | | Non-Executive Director | |
| Mahendra Shah, Ph.D. | | | 79 | | | 2016 | | | Non-Executive Director | |
| Vikas Sinha | | | 60 | | | 2016 | | | Non-Executive Director | |
| Anders Ullman, M.D., Ph.D. | | | 68 | | | 2015 | | | Non-Executive Director | |
| | Board Diversity Matrix (As of March 20, 2024) | | |||
| | Country of Principal Executive Offices | | | United Kingdom | |
| | Foreign Private Issuer | | | No | |
| | Disclosure Prohibited Under Home Country Law | | | No | |
| | Total Number of Directors | | | 11 | |
| | | | Female | | | Male | | | Non-Binary | | | Did Not Disclose Gender | | ||||||||||||
| Part I: Gender Identity | | | | | | | | | | | | | | | | | | | | | | | | | |
| Directors | | | | | 2 | | | | | | 9 | | | | | | — | | | | | | — | | |
| Part II: Demographic Background | | | | | | | | | | | | | | | | | | | | | | | | | |
| Underrepresented Individual in Home Country Jurisdiction | | | | | | | | | | | | | | | | | 6 | | | | | | | | |
| LGBTQ+ | | | | | | | | | | | | | | | | | 1 | | | | | | | | |
| Did Not Disclose Demographic Background | | | | | | | | | | | | | | | | | — | | | | | | | | |
To receive and adopt the 2023 U.K. Annual Report
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the receipt and adoption of the 2023 U.K. Annual Report | |
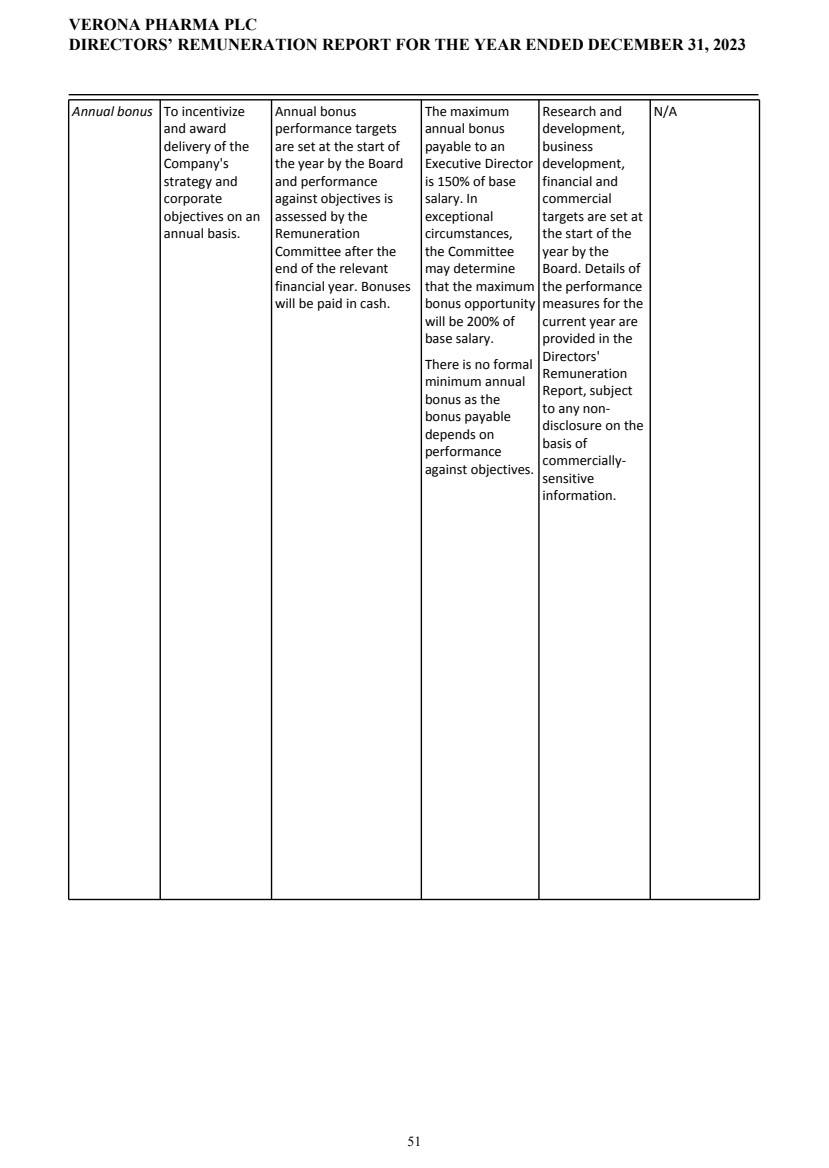
To receive and approve, as a non-binding advisory resolution, the U.K. statutory Directors’ Remuneration Report
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the receipt and approval, as a non-binding advisory resolution, of the U.K. statutory Directors’ Remuneration Report | |
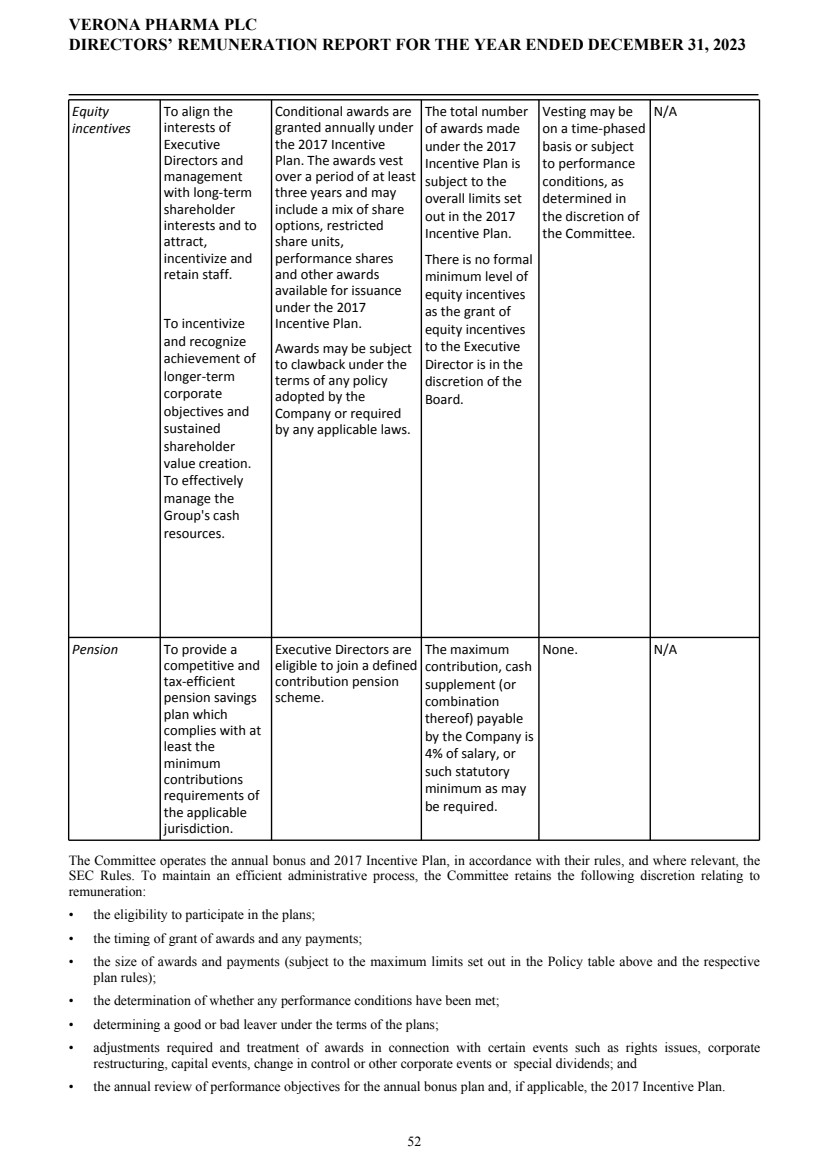
To receive and approve the U.K. Directors’ Remuneration Policy, as set out on pages 47 to 57 of the U.K. Annual Report, which, if approved, will take effect upon conclusion of the AGM.
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the receipt and adoption of the U.K. Directors’ Remuneration Policy | |
To approve an increase in the maximum aggregate level of Non-Executive Director annual remuneration to £750,000 per annum
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the increase in the maximum aggregate level of Non-Executive Director annual remuneration of £750,000 per annum | |
To appoint EY as the Company’s auditors, to hold office until the conclusion of the next annual general meeting of shareholders
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the appointment of EY as the Company’s auditor | |
Christina Ackermann
David Ebsworth, Ph.D.
James Brady
| Fee Category | | | 2023 | | | 2022 | | ||||||
| Audit Fees | | | | $ | 1,058,000 | | | | | $ | 420,000 | | |
| Audit-Related Fees | | | | $ | 158,000 | | | | | $ | 201,000 | | |
| Tax Fees | | | | | — | | | | | | — | | |
| All Other Fees | | | | $ | 97,000 | | | | | $ | 403,000 | | |
| Total Fees | | | | $ | 1,313,000 | | | | | $ | 1,024,000 | | |
To authorize the Audit and Risk Committee to determine the auditors’ remuneration for the year ending December 31, 2024
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR authorization of the Audit Committee to determine EY’s remuneration for the year ending December 31, 2024 | |
To approve, on an advisory (non-binding) basis, the compensation of the Company’s named executive officers
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the approval, on an advisory (non-binding) basis, of the compensation of the Company’s named executive officers | |
To authorize the Board of Directors to exercise all the powers of the Company to allot shares in the Company or grant rights to subscribe for or to convert any security into shares in the Company up to an aggregate nominal amount of £8,345,745 (being up to a maximum of 166,914,908 shares representing 25% of the Company’s existing ordinary share capital as at the close of business on March 13, 2024 (being the latest practicable date prior to publication of this document)).
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the authorization of Directors to exercise all the powers to the Company to allot shares in the Company or grant Rights up to an aggregate nominal amount of £8,345,745 (being up to a maximum of 166,914,908 shares representing 25% of the Company’s existing ordinary share capital as at the close of business on March 13, 2024 (being the latest practicable date prior to publication of this document)) | |
To authorize the Board of Directors to allot equity securities for cash and/or to sell ordinary shares held by the Company as treasury shares for cash as if Section 561(1) of the Companies Act does not apply to any such allotment
| | ![[MISSING IMAGE: ic_arrow-bw.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/ic_arrow-bw.jpg) | | | The Board of Directors unanimously recommends a vote FOR the authorization of Directors to allot equity securities for cash and/or to sell ordinary shares held by the Company as treasury shares for cash as if Section 561(1) of the Companies Act does not apply to any such allotment | |
| Name | | | Age | | | Position | |
| David Zaccardelli, Pharm.D.(1) | | | 59 | | | President, Chief Executive Officer and Executive Director | |
| Andrew Fisher(3) | | | 54 | | | General Counsel | |
| Mark W. Hahn(2) | | | 61 | | | Chief Financial Officer | |
| Kathleen Rickard, M.D.(4) | | | 65 | | | Chief Medical Officer | |
| Name | | | Audit and Risk | | | Remuneration | | | Nomination and Corporate Governance | |
| Christina Ackermann | | | X | | | | | | | |
| Ken Cunningham, M.D. | | | | | | Chair | | | | |
| James Brady | | | X | | | | | | | |
| Lisa Deschamps | | | | | | | | | X | |
| David Ebsworth, Ph.D. | | | X | | | X | | | Chair | |
| Mahendra Shah, Ph.D. | | | | | | X | | | | |
| Vikas Sinha | | | Chair | | | | | | X | |
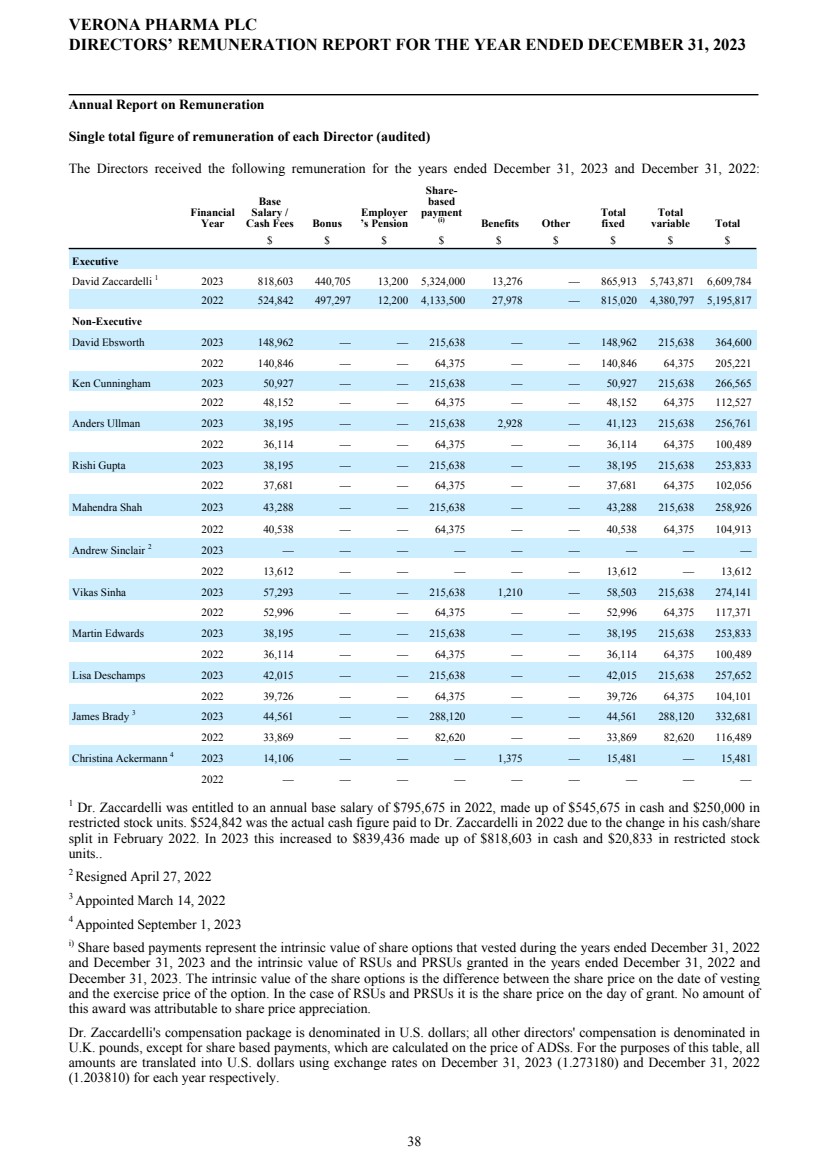
| Name and Principal Position | | | Year | | | Cash Base Salary ($) | | | Stock Awards ($)(1) | | | Non-Equity Incentive Plan Compensation ($) | | | All Other Compensation ($) | | | Total ($) | | ||||||||||||||||||
| David Zaccardelli, Pharm.D. | | | | | 2023 | | | | | | 819,568 | | | | | | 5,324,000 | | | | | | 440,705 | | | | | | 13,200(2) | | | | | | 6,597,473 | | |
| President and Chief Executive Officer (PEO) | | | | | 2022 | | | | | | 524,842 | | | | | | 4,133,500 | | | | | | 497,297 | | | | | | 27,501 | | | | | | 5,183,140 | | |
| Mark W. Hahn | | | | | 2023 | | | | | | 551,291 | | | | | | 5,324,000 | | | | | | 293,803 | | | | | | 13,200(2) | | | | | | 6,182,294 | | |
| Chief Financial Officer | | | | | 2022 | | | | | | 430,450 | | | | | | 3,983,498 | | | | | | 331,531 | | | | | | 22,401 | | | | | | 4,767,880 | | |
| Kathleen Rickard, M.D. | | | | | 2023 | | | | | | 458,333 | | | | | | 1,331,000 | | | | | | 192,500 | | | | | | — | | | | | | 1,981,833 | | |
| Chief Medical Officer | | | | | 2022 | | | | | | 434,439 | | | | | | 863,000 | | | | | | 217,220 | | | | | | 3,342 | | | | | | 1,518,001 | | |
| Name | | | 2023 Annual Base Salary ($) | | | 2024 Annual Base Salary ($) | | ||||||
David Zaccardelli(1) | | | | | 839,437 | | | | | | 873,015 | | |
Mark W. Hahn(1) | | | | | 559,625 | | | | | | 582,010 | | |
| Kathleen Rickard | | | | | 458,333 | | | | | | 504,167 | | |
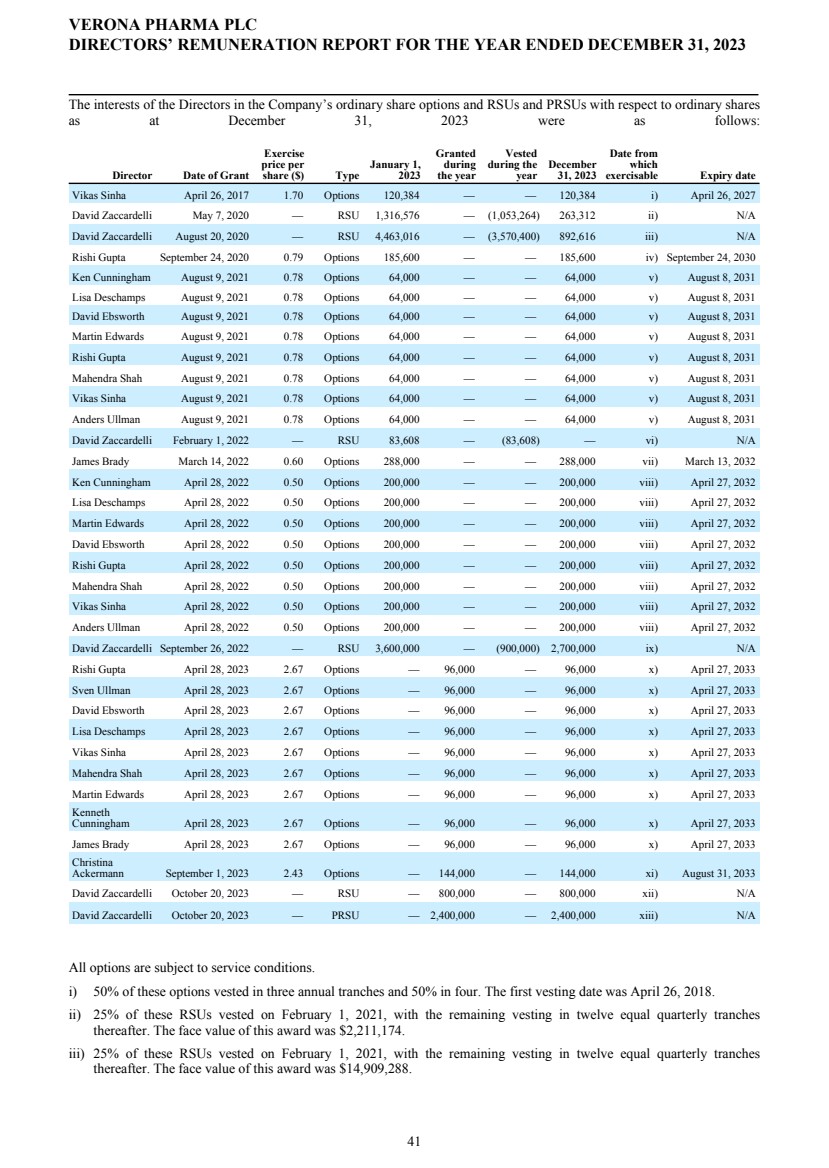
| | | | | | | | | | Option Awards | | | Stock Awards | | ||||||||||||||||||||||||||||||||||||||||||
| Name | | | Grant Date | | | Number of Securities Underlying Unexercised Options (#) Exercisable | | | Number of Securities Underlying Unexercised Options (#) Unexercisable | | | Option Exercise Price ($) | | | Option Expiration Date | | | Number of Shares or Units of Stock That Have Not Vested (#) | | | Market Value of Shares or Units of Stock That Have Not Vested ($) | | | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Other Rights That Have Not Vested (#) | | | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Other Rights That Have Not Vested ($) | | |||||||||||||||||||||||||||
| David Zaccardelli | | | | | 5/7/2020(1) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 32,914 | | | | | | 654,330 | | | | | | — | | | | | | — | | |
| | | | 8/20/2020(1) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 111,577 | | | | | | 2,218,151 | | | | | | — | | | | | | — | | | ||
| | | | 9/26/2022(2) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 337,500 | | | | | | 6,709,500 | | | | | | — | | | | | | — | | | ||
| | | | 10/20/2023(3) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 100,000 | | | | | | 1,988,000 | | | | | | | | | | | | | | | ||
| | | | 10/20/2023(4) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | | | | | | | | | | | | | 300,000 | | | | | | 5,964,000 | | | ||
| Mark W. Hahn | | | | | 5/7/2020(1) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 24,685 | | | | | | 490,738 | | | | | | — | | | | | | — | | |
| | | | 8/20/2020(1) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 119,802 | | | | | | 2,381,664 | | | | | | — | | | | | | — | | | ||
| | | | 9/26/2022(2) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 337,500 | | | | | | 6,709,500 | | | | | | — | | | | | | — | | | ||
| | | | 10/20/2023(3) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 100,000 | | | | | | 1,988,000 | | | | | | | | | | | | | | | ||
| | | | 10/20/2023(4) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 300,000 | | | | | | 5,964,000 | | | ||
| Kathleen Rickard | | | | | 4/1/2019 | | | | | | 70,000 | | | | | | — | | | | | | 5.99 | | | | | | 3/29/2029 | | | | | | — | | | | | | — | | | | | | | | | | | | | | |
| | | | 3/3/2020 | | | | | | 11719 | | | | | | 781(5) | | | | | | 5.64 | | | | | | 3/3/2030 | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | ||
| | | | 8/20/2020(6) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 71,887 | | | | | | 1,429,114 | | | | | | — | | | | | | — | | | ||
| | | | 9/26/2022(2) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 75,000 | | | | | | 1,491,000 | | | | | | — | | | | | | — | | | ||
| | | | 10/20/2023(3) | | | | | | — | | | | | | — | | | | | | — | | | | | | — | | | | | | 25,000 | | | | | | 497,000 | | | | | | | | | | | | | | | ||
| | | | 10/20/2023(4) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | 75,000 | | | | | | 1,491,000.00 | | | ||
| Year | | | Summary Compensation Table Total for PEO ($) | | | Compensation Actually Paid to PEO ($)(1) | | | Average Summary Compensation Table Total for Non-PEO NEOs ($) | | | Average Compensation Actually Paid to Non-PEO NEOs ($)(1) | | | Value of Initial Fixed $100 Investment Based on: | | | Net Income ($) | | ||||||||||||||||||
| | Total Shareholder Return ($) | | |||||||||||||||||||||||||||||||||||
| 2023 | | | | | 6,559,277 | | | | | | 558,317 | | | | | | 4,082,064 | | | | | | 207,291 | | | | | | 284.00 | | | | | | (54,369,000) | | |
| 2022 | | | | | 5,183,140 | | | | | | 27,578,945 | | | | | | 3,142,941 | | | | | | 17,462,808 | | | | | | 373.29 | | | | | | (68,701,000) | | |
| 2021 | | | | | 1,123,304 | | | | | | 823,718 | | | | | | 650,336 | | | | | | 425,254 | | | | | | 96.00 | | | | | | (55,569,000) | | |
| Year | | | PEO | | | Non-PEO NEOs | |
| 2023 | | | David Zaccardelli | | | Mark W. Hahn and Kathleen Rickard | |
| 2022 | | | David Zaccardelli | | | Mark W. Hahn and Kathleen Rickard | |
| 2021 | | | David Zaccardelli | | | Mark W. Hahn and Kathleen Rickard | |
| | | | 2023 | | |||||||||
| Adjustments | | | PEO | | | Average Non-PEO NEOs | | ||||||
| Deduction for Amounts Reported under the “Stock Awards” and “Option Awards” Columns in the Summary Compensation Table for Applicable FY | | | | | (5,324,000) | | | | | | (3,327,500) | | |
| Increase based on ASC 718 Fair Value of Awards Granted during Applicable FY that Remain Unvested as of Applicable FY End, determined as of Applicable FY End | | | | | 7,952,000 | | | | | | 4,970,000 | | |
| Increase based on ASC 718 Fair Value of Awards Granted during Applicable FY that Vested during Applicable FY, determined as of Vesting Date | | | | | — | | | | | | — | | |
| Increase/deduction for Awards Granted during Prior FY that were Outstanding and Unvested as of Applicable FY End, determined based on change in ASC 718 Fair Value from Prior FY End to Applicable FY End | | | | | (3,012,444) | | | | | | (2,185,934) | | |
| | | | 2023 | | |||||||||
| Adjustments | | | PEO | | | Average Non-PEO NEOs | | ||||||
| Increase/deduction for Awards Granted during Prior FY that Vested During Applicable FY, determined based on change in ASC 718 Fair Value from Prior FY End to Vesting Date | | | | | (5,616,516) | | | | | | (3,331,338) | | |
| Deduction of ASC 718 Fair Value of Awards Granted during Prior FY that were Forfeited during Applicable FY, determined as of Prior FY End | | | | | — | | | | | | — | | |
| Increase based on Dividends or Other Earnings Paid during Applicable FY prior to Vesting Date | | | | | — | | | | | | — | | |
| Increase based on Incremental Fair Value of Options/SARs Modified during Applicable FY | | | | | — | | | | | | — | | |
| TOTAL ADJUSTMENTS | | | | | (6,000,960) | | | | | | (3,874,772) | | |
![[MISSING IMAGE: bc_totalshare-4c.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/bc_totalshare-4c.jpg)
![[MISSING IMAGE: bc_netincome-4c.jpg]](https://capedge.com/proxy/DEF 14A/0001104659-24-036465/bc_netincome-4c.jpg)
| Name | | | Fees Earned or Paid in Cash ($)(1) | | | Option Awards ($)(2) | | | Total ($) | | |||||||||
| David Ebsworth | | | | | 148,962 | | | | | | 181,540 | | | | | | 330,502 | | |
| Christina Ackermann | | | | | 14,106 | | | | | | 253,807 | | | | | | 267,913 | | |
| James Brady | | | | | 44,561 | | | | | | 181,540 | | | | | | 226,101 | | |
| Kenneth Cunningham | | | | | 50,927 | | | | | | 181,540 | | | | | | 232,467 | | |
| Lisa Deschamps | | | | | 42,015 | | | | | | 181,540 | | | | | | 223,555 | | |
| Martin Edwards | | | | | 38,195 | | | | | | 181,540 | | | | | | 219,735 | | |
| Rishi Gupta | | | | | 38,195 | | | | | | 181,540 | | | | | | 219,735 | | |
| Mahendra Shah | | | | | 43,288 | | | | | | 181,540 | | | | | | 224,828 | | |
| Vikas Sinha | | | | | 57,293 | | | | | | 181,540 | | | | | | 238,833 | | |
| Sven Ullman | | | | | 38,195 | | | | | | 181,540 | | | | | | 219,735 | | |
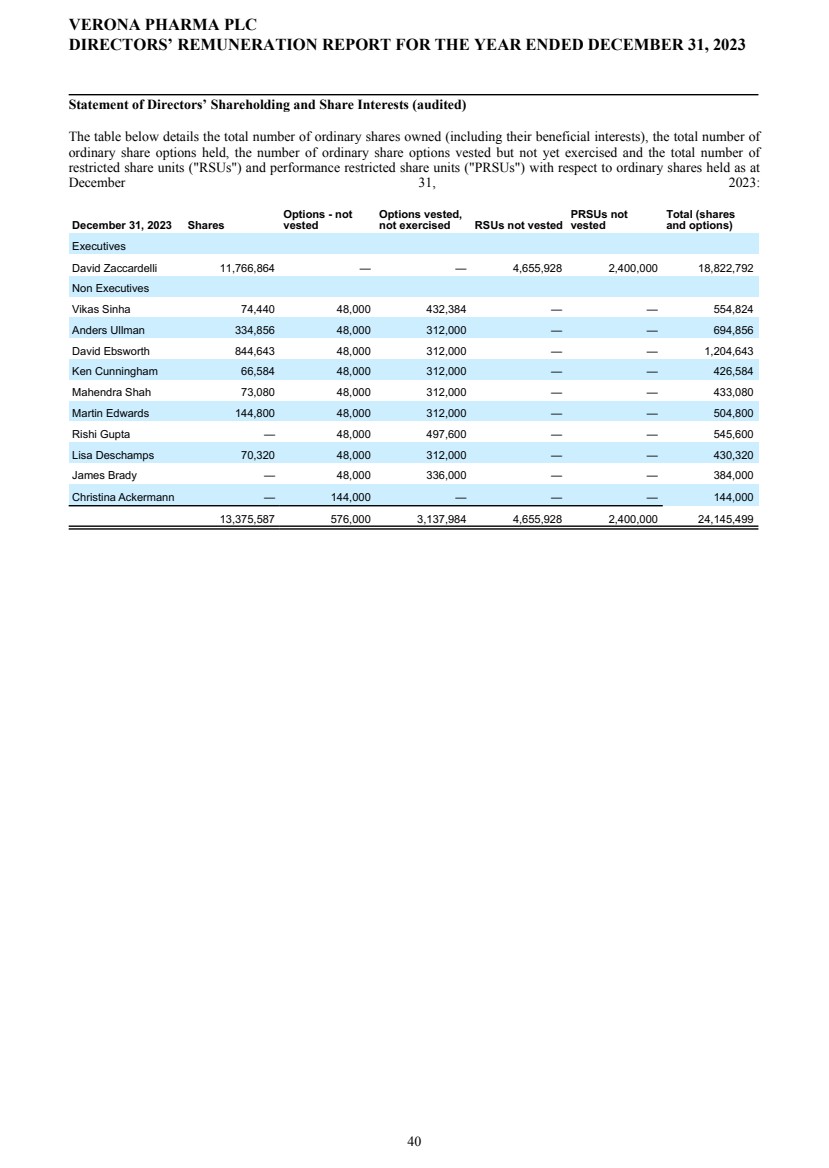
| Name | | | Option Awards Outstanding at 2023 Fiscal Year End | | | Unvested Stock Awards Outstanding at 2023 Fiscal Year End | | ||||||
| David Ebsworth | | | | | 39,000 | | | | | | 6,000 | | |
| Christina Ackermann | | | | | — | | | | | | 18,000 | | |
| James Brady | | | | | 42,000 | | | | | | 6,000 | | |
| Ken Cunningham | | | | | 39,000 | | | | | | 6,000 | | |
| Lisa Deschamps | | | | | 39,000 | | | | | | 6,000 | | |
| Martin Edwards | | | | | 39,000 | | | | | | 6,000 | | |
| Rishi Gupta | | | | | 62,200 | | | | | | 6,000 | | |
| Mahendra Shah | | | | | 39,000 | | | | | | 6,000 | | |
| Vikas Sinha | | | | | 54,048 | | | | | | 6,000 | | |
| Anders Ullman | | | | | 39,000 | | | | | | 6,000 | | |
| Plan Category | | | Number of Ordinary Shares to be Issued Upon Exercise of Outstanding Options, Warrant and Rights (a) | | | Weighted- Average Exercise Price of Outstanding Options, Warrant and Rights (b) | | | Number of Ordinary Shares Remaining Available for Future Issuance Under Equity Compensation Plans (Excluding Securities Reflected in Column (a)) (c) | | |||||||||
| Equity compensation plans approved by shareholders | | | | | 54,922,392(1) | | | | | $ | 1.56(2) | | | | | | 2,899,715(3) | | |
| Equity compensation plans not approved by shareholders | | | | | — | | | | | | — | | | | | | — | | |
| Total | | | | | 54,922,392 | | | | | $ | 1.56 | | | | | | 2,899,715 | | |
| | | | Shares beneficially owned | | |||||||||
| Name of beneficial owner | | | Number | | | Percent | | ||||||
| 5% or greater shareholders: | | | | | | | | | | | | | |
RA Capital Management affiliates(1) | | | | | 60,031,808 | | | | | | 9.3% | | |
New Enterprise Associates affiliates(2) | | | | | 46,447,651 | | | | | | 7.2% | | |
FMR LLC(3) | | | | | 39,219,016 | | | | | | 6.1% | | |
| Named Executive Officers, Directors and Director Nominees: | | | | | | | | | | | | | |
David Zaccardelli, Pharm.D.(4) | | | | | 12,667,592 | | | | | | 2.0% | | |
| Christina Ackermann | | | | | — | | | | | | * | | |
| Michael Austwick | | | | | — | | | | | | * | | |
Ken Cunningham, M.D.(5) | | | | | 426,584 | | | | | | * | | |
Lisa Deschamps(6) | | | | | 430,320 | | | | | | * | | |
David Ebsworth, Ph.D.(7) | | | | | 1,204,643 | | | | | | * | | |
Martin Edwards, M.D.(8) | | | | | 504,800 | | | | | | * | | |
Mahendra Shah, Ph.D.(9) | | | | | 433,080 | | | | | | * | | |
Anders Ullman, M.D., Ph.D.(10) | | | | | 694,856 | | | | | | * | | |
Vikas Sinha(11) | | | | | 554,824 | | | | | | * | | |
James Brady(12) | | | | | 384,000 | | | | | | * | | |
Mark W. Hahn(13) | | | | | 11,756,576 | | | | | | 1.8% | | |
Kathleen Rickard, M.D.(14) | | | | | 2,273,224 | | | | | | * | | |
All directors and executive officers as a group (14 persons)(15) | | | | | 31,330,499 | | | | | | 4.8% | | |
Attention: General Counsel
Email: legal@veronapharma.com
3 More London Riverside
London SE1 2RE
United Kingdom
STATEMENT. PROMPTLY VOTING YOUR SHARES WILL ENSURE THE PRESENCE OF A
QUORUM AT THE ANNUAL GENERAL MEETING OF SHAREHOLDERS AND WILL SAVE US
THE EXPENSE OF FURTHER SOLICITATION.
VOTING INSTRUCTIONS TO CITIBANK, N.A., THE DEPOSITARY FOR THE ADSs.
President and Chief Executive Officer
| Company Number 05375156 VERONA PHARMA PLC ANNUAL REPORT AND ACCOUNTS YEAR ENDED DECEMBER 31, 2023 |
| Directors, Secretary and Advisors 2 Directors' Report 3 Corporate Governance Report 7 Strategic Report 16 Directors' Remuneration Report 36 Independent Auditors' Report 58 Consolidated Statement of Comprehensive Income 65 Consolidated Statement of Financial Position 66 Company Statement of Financial Position 67 Consolidated Statement of Changes in Equity 68 Company Statement of Changes in Equity 69 Consolidated Statement of Cash Flows 70 Company Statement of Cash Flows 71 Notes to the Financial Statements 72 to 102 VERONA PHARMA PLC CONTENTS 1 |
| Directors Dr. David Ebsworth (Non-Executive Chairperson) Dr. David Zaccardelli (President & Chief Executive Officer) Ms. Christina Ackermann Mr. Michael Austwick Mr. James Brady Dr. Ken Cunningham Ms. Lisa Deschamps Dr. Martin Edwards Dr. Mahendra Shah Mr. Vikas Sinha Dr. Anders Ullman Company Secretary Mr. Ben Harber Registered Office One Central Square Cardiff CF10 1FS Company Number 05375156 Independent Auditors PricewaterhouseCoopers LLP 4th Floor, One Reading Central 23 Forbury Road Reading Berkshire RG1 3JH Solicitors Latham & Watkins LLP 99 Bishopsgate London EC2M 3XF Registrars Computershare Investor Services plc The Pavilions Bridgewater Road Bristol BS99 6ZZ VERONA PHARMA PLC DIRECTORS, SECRETARY AND ADVISORS 2 |
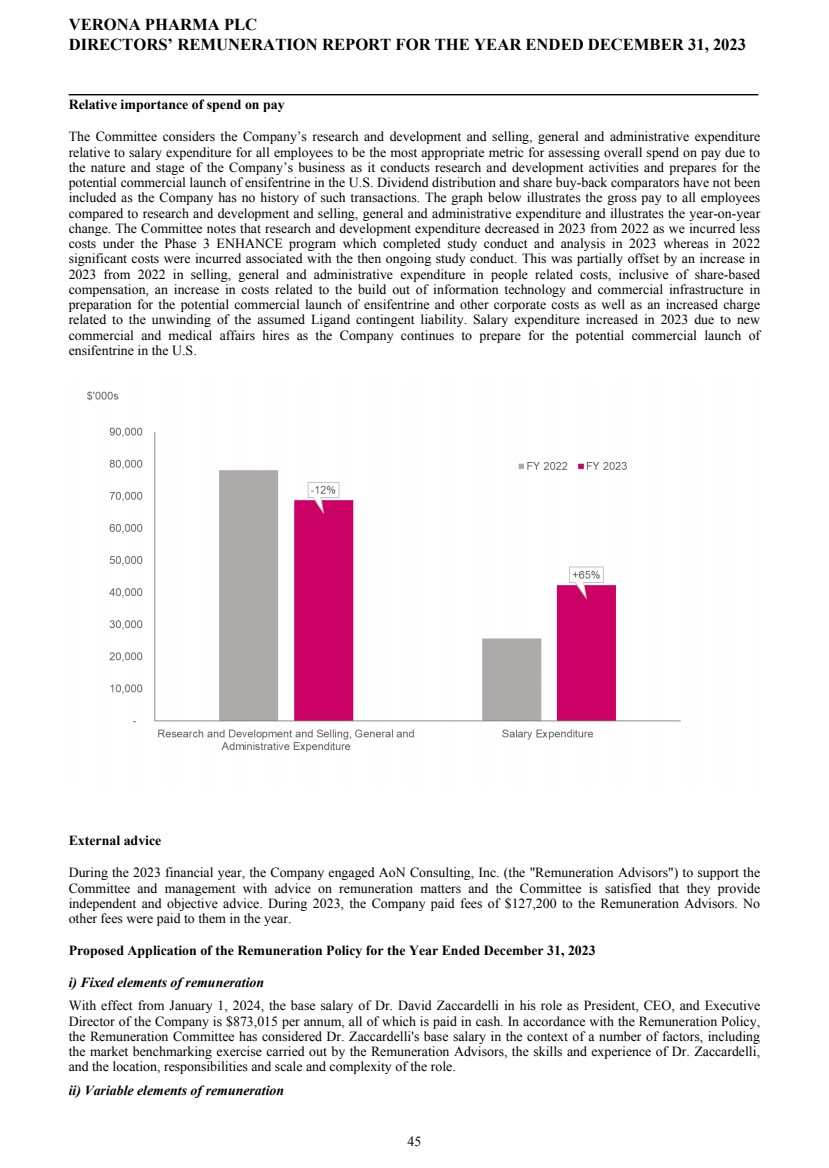
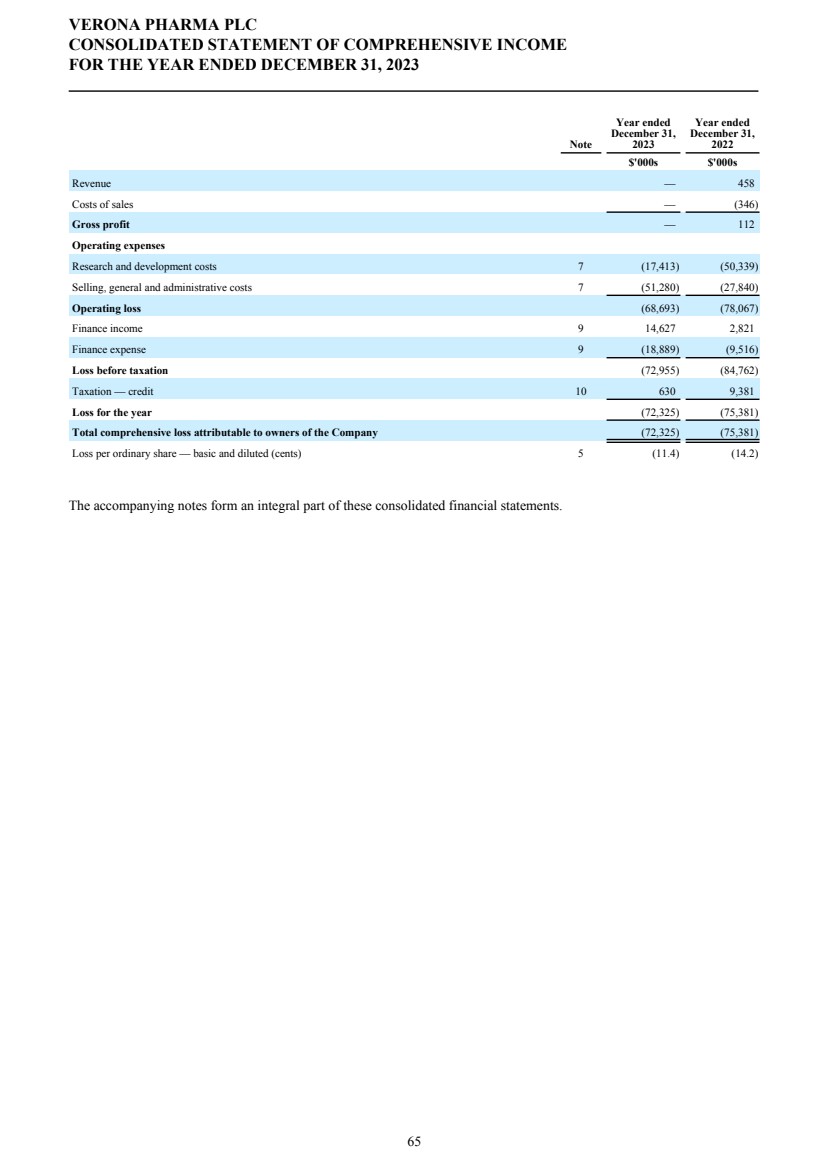
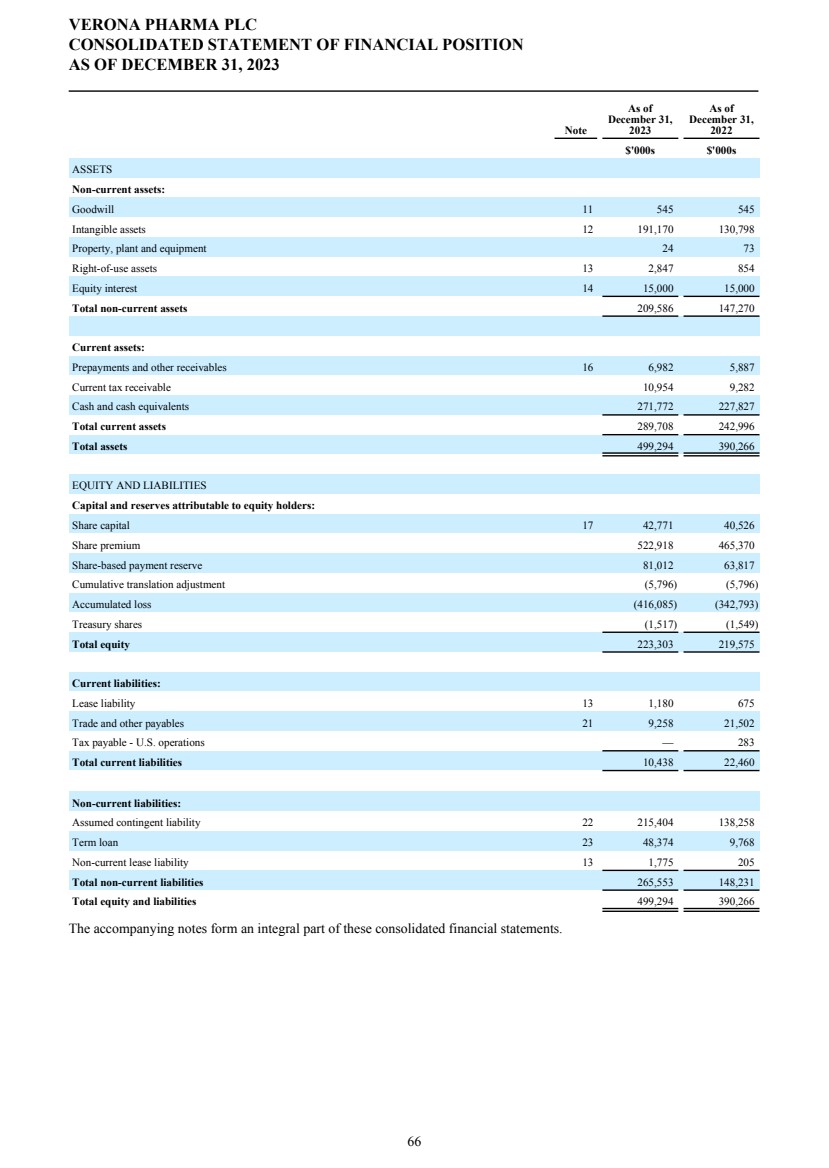
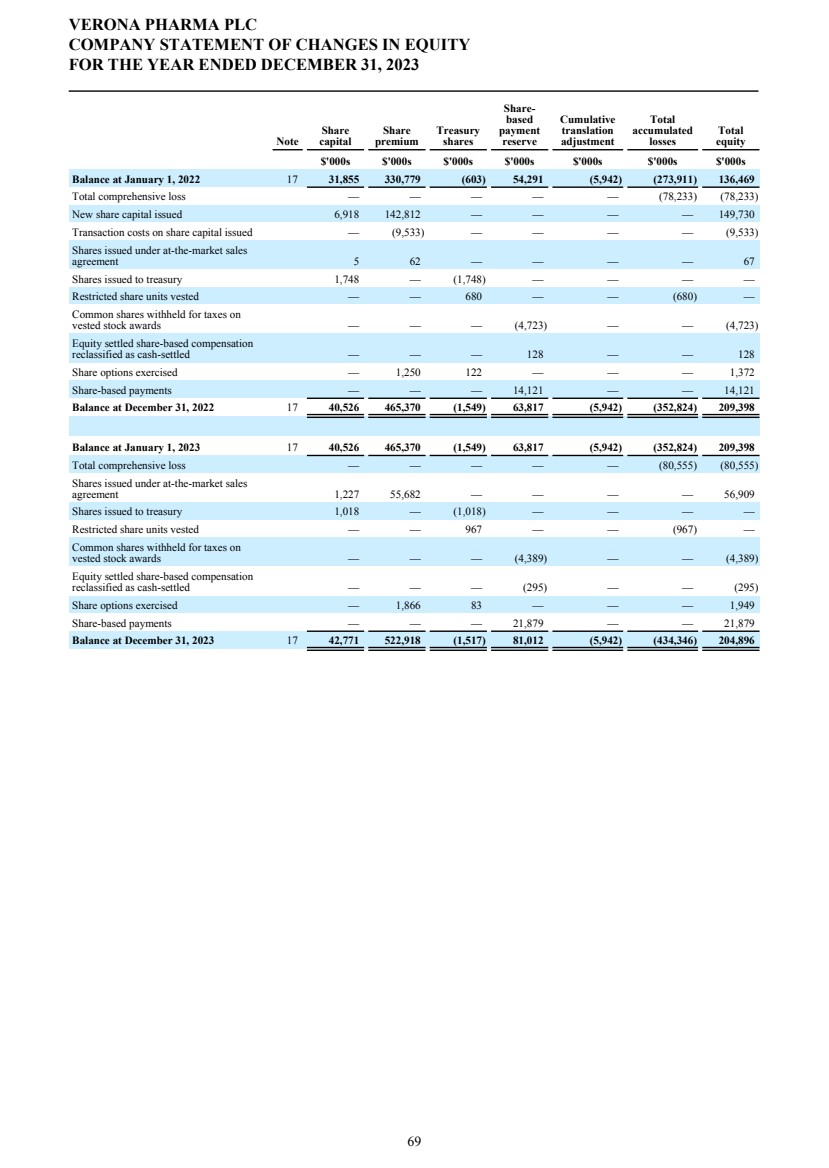
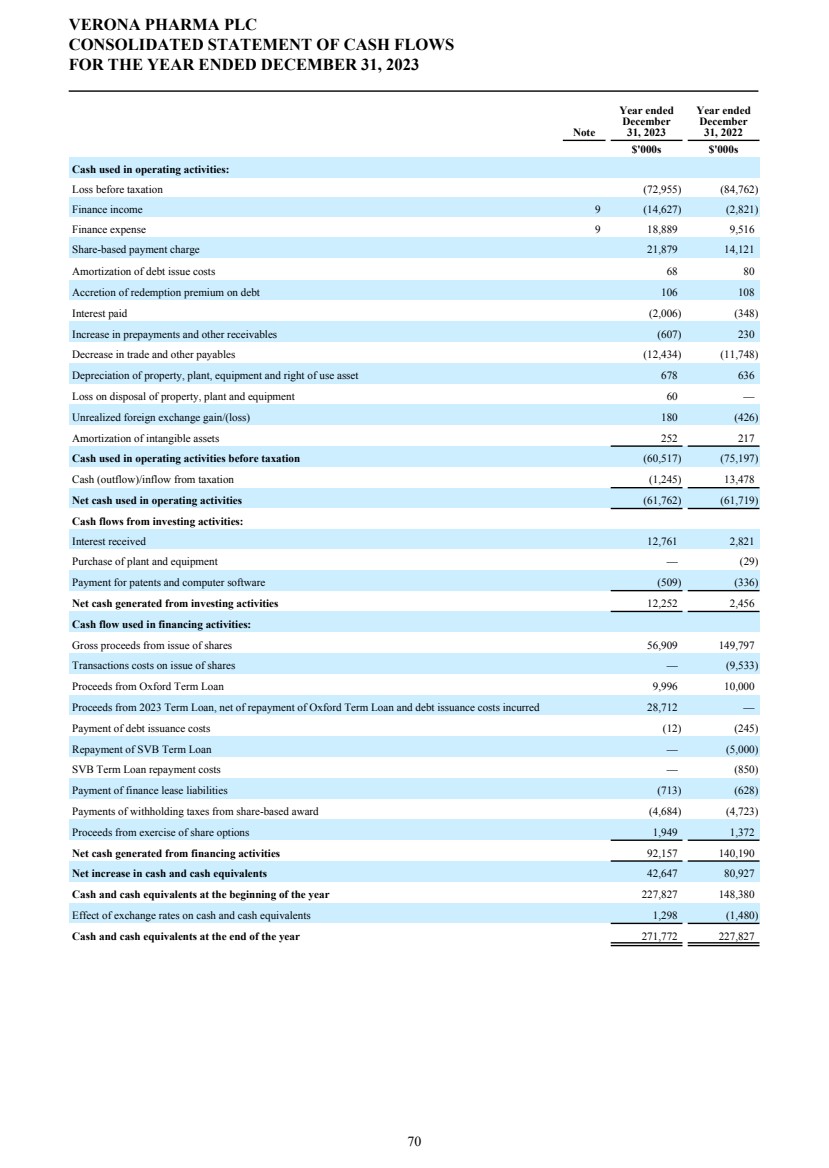
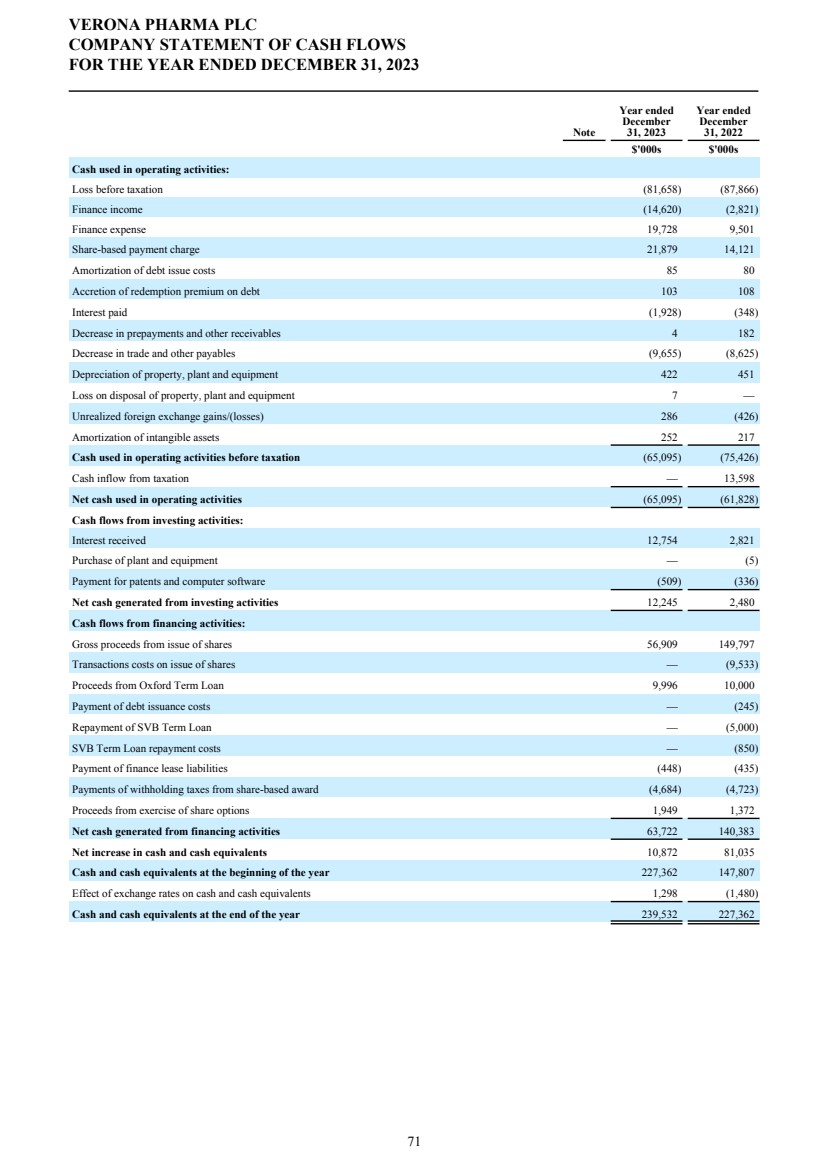
| DIRECTORS' REPORT The Directors present their report together with the audited consolidated financial statements, audited company financial statements and auditors’ report for the year ended December 31, 2023. Results and dividends The Group results for the year are set out on page 65. The loss after taxation for the year was $72.3 million (2022: $75.4 million). The loss was lower in 2023 primarily due to a decrease in research and development costs from 2022 as we incurred less costs under the Phase 3 ENHANCE program which completed study conduct and analysis in 2023 whereas in 2022 significant costs were incurred associated with the then ongoing study conduct. This was partially offset by an increase in people related costs, inclusive of share-based compensation, an increase in costs related to the build out of information technology and commercial infrastructure in preparation for the potential commercial launch of ensifentrine and other corporate costs as well as an increased charge related to the unwinding of the assumed Ligand contingent liability. The Company has no distributable reserves so the Directors cannot recommend the payment of a dividend (2022: $nil). Cash and cash equivalents at December 31, 2023 increased to $271.8 million from $227.8 million at December 31, 2022 primarily due to cash inflows from financing activities including proceeds from the issuance of shares under the at-the-market offering program entered into in March 2021, at an average price of approximately $2.88 per share (equivalent to $23.08 per ADS), raising aggregate net proceeds of approximately $56.9 million after deducting issuance costs. Further, the Group received $27.8 million from the proceeds of the first tranche of a term loan facility entered into in December 2023 with Oxford Finance LLC, ("Oxford") as collateral agent and certain funds managed by Oxford and Hercules Capital, Inc., pursuant to which a term loan facility in an aggregate amount of up to $400.0 million with a term loan advance of $50.0 million funded on the closing date and four additional term loan advances aggregating up to $350.0 million, subject to certain terms and conditions. A portion of the proceeds were used to repay, in full, the outstanding indebtedness owed by the Group under the prior loan and security agreement with Oxford Finance Luxembourg S.À R.L. The Strategic Report describes the Group’s research and development strategy and activities. Directors The Directors of the company who were in office during 2023 and up to the date of signing of the financial statements unless otherwise stated were: Executive Directors Dr. David Zaccardelli Non-executive Directors Dr. David Ebsworth Ms. Christina Ackermann (appointed September 1, 2023) Mr. Michael Austwick (appointed February 1, 2024) Mr. James Brady Dr. Ken Cunningham Ms. Lisa Deschamps Dr. Martin Edwards Mr. Rishi Gupta (resigned January 31, 2024) Dr. Mahendra Shah Mr. Vikas Sinha Dr. Anders Ullman To the extent permitted by the U.K. Companies Act 2006, we are empowered to indemnify our directors against any liability they incur by reason of their directorship. We have also entered into a deed of indemnity with each of our directors and executive officers, in accordance with the Companies Act. These deeds of indemnity were in place during the year ended December 31, 2023, and up to the date of signing of the financial statements. In addition to such indemnification, we provide our directors and executive officers with directors’ and officers’ liability insurance. Pensions Verona Pharma plc operates defined contribution pension plans open to all executive directors and employees. Political and charitable contributions There were no political or charitable contributions made by the Company during the years ended December 31, 2023, or 2022. VERONA PHARMA PLC DIRECTORS' REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 3 |
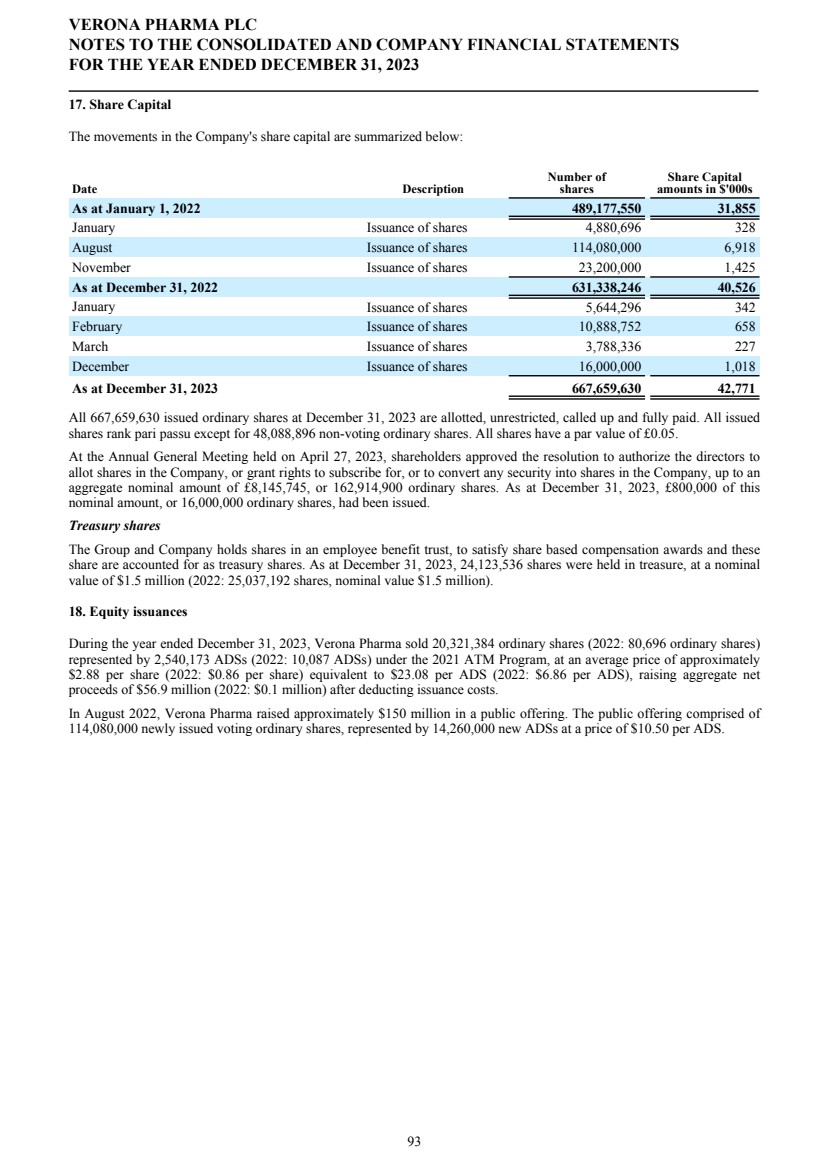
| Future developments The Strategic Report describes the Group’s activities, strategy and future prospects. Capital Structure As at December 31, 2023, the Company had 667,659,630 ordinary shares of 5p nominal value each, of which 48,088,896 are non-voting. In all other respects they rank pari passu. The Company is listed on the Nasdaq Global Market ("Nasdaq") and American Depositary Shares (“ADSs”) are traded on Nasdaq. One ADS represents eight ordinary shares. VERONA PHARMA PLC DIRECTORS' REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 4 |
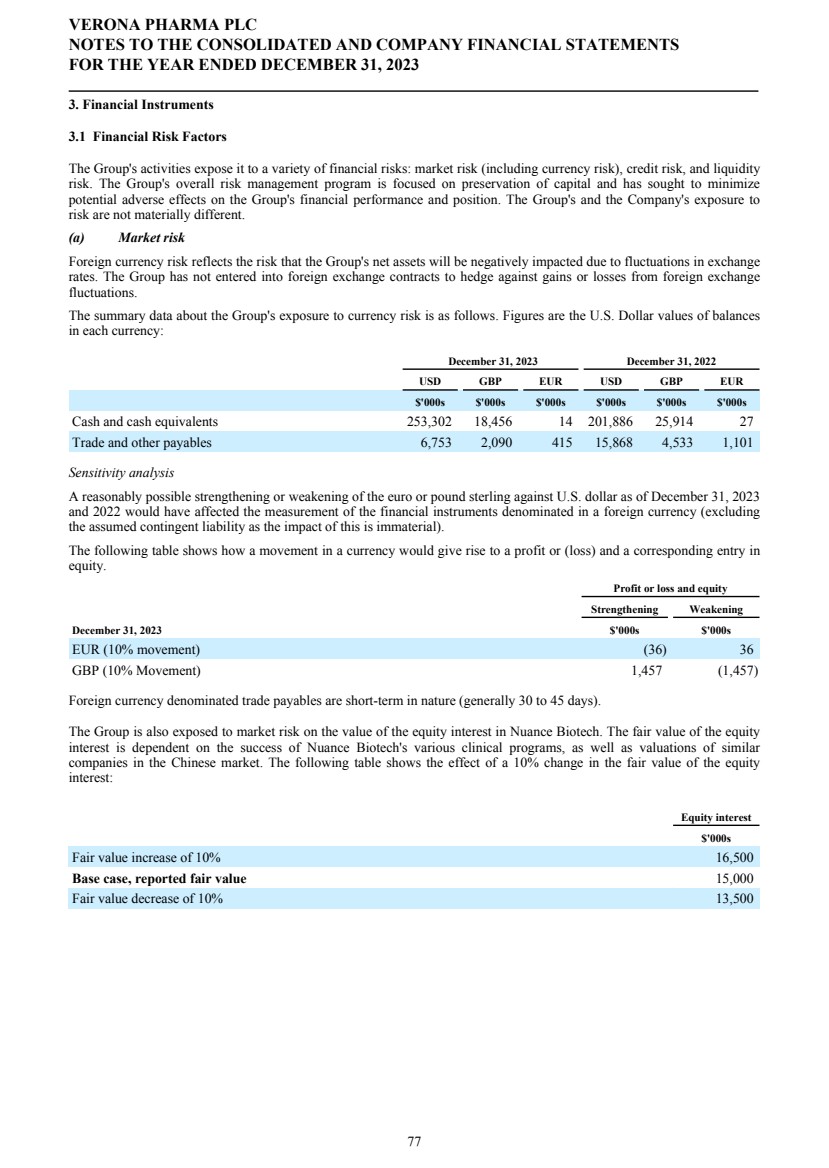
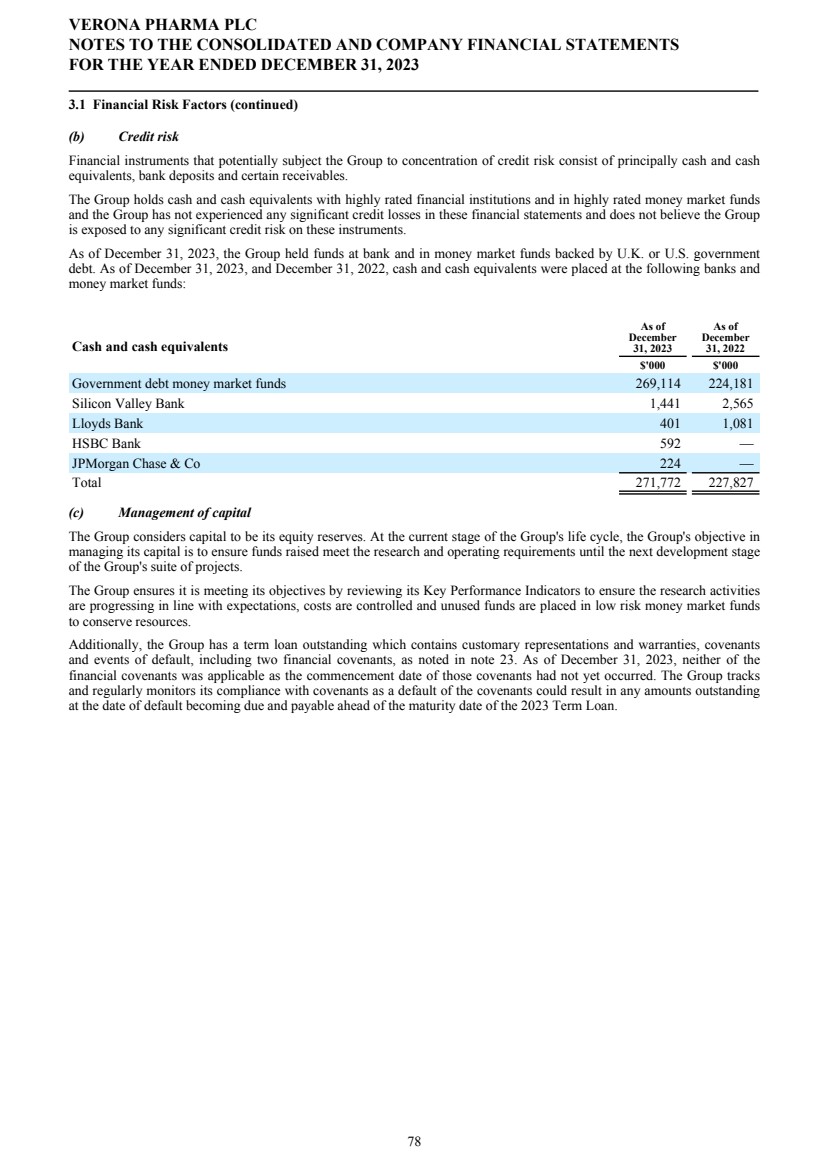
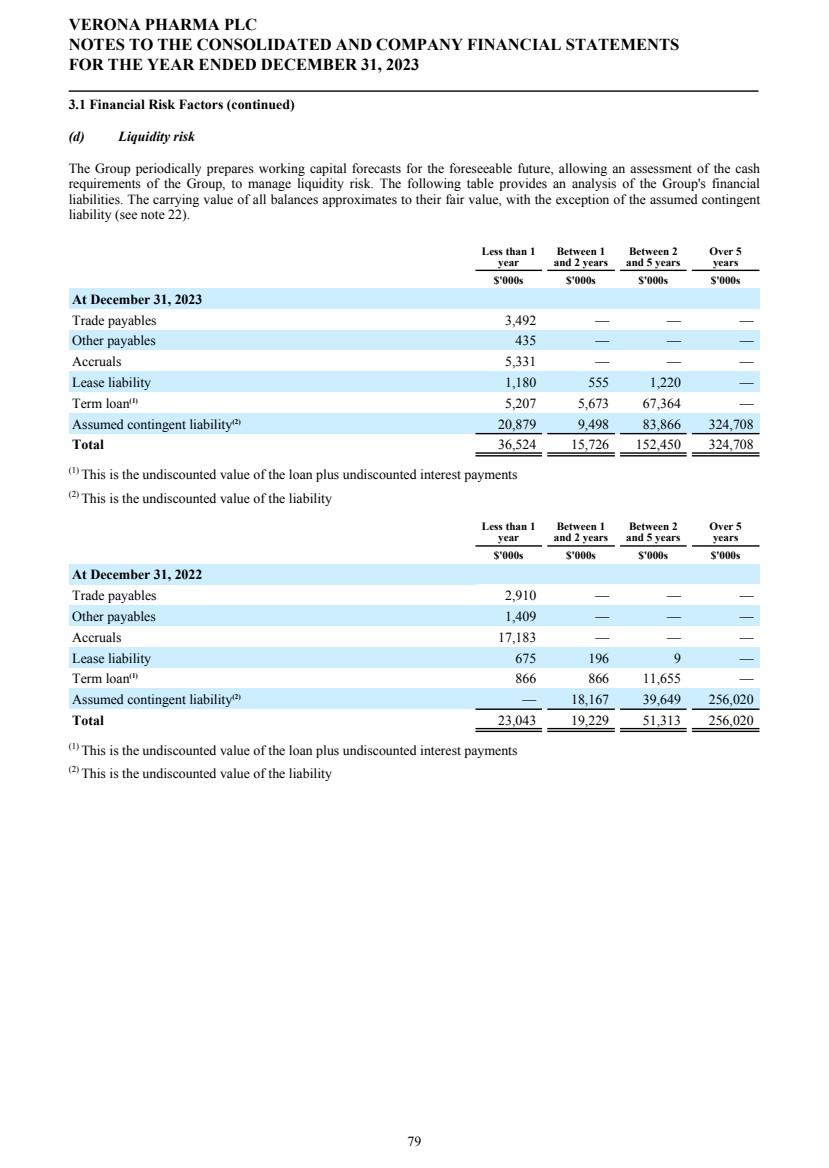
| Corporate Governance The Company's statement on corporate governance can be found in the corporate governance report of these financial statements. The corporate governance report forms part of this Directors' Report and is incorporated into it by cross-reference. Principal Risks and Uncertainties See the Strategic Report for a discussion of risks facing the Group. Financial risk management We are exposed to a variety of financial risks. Our overall risk management program seeks to minimize potential adverse effects of these financial risks on our financial performance. Credit Risk Financial instruments that potentially subject us to concentration of credit risk consist of principally cash and cash equivalents, bank deposits and certain receivables. We hold cash and cash equivalents with highly rated financial institutions and in highly rated money market funds and we have not experienced any significant credit losses in these financial statements and do not believe we are exposed to any significant credit risk on these instruments. Liquidity Risk We manage our liquidity risk by investing surplus cash in funds with highly liquid money market funds investing in U.S. and U.K. government securities. Market Risk Foreign currency risk reflects the risk that the value of a financial commitment or recognized asset or liability will fluctuate due to changes in foreign currency rates. Our financial position, as expressed in U.S. dollars, is exposed to movements in foreign exchange rates against pounds sterling and the euro. Our main trading currencies are the U.S. dollar, pounds sterling, and the euro. We are exposed to foreign currency risk as a result of operating transactions and the translation of foreign bank accounts. We monitor our exposure to foreign exchange risk; sensitivity analysis and exposure is described further in note 3.1 in the financial statements. We have not entered into foreign exchange contracts to hedge against gains or losses from foreign exchange fluctuations. Locations The Company’s principal place of business is in London, U.K., and it operates subsidiary offices in Raleigh, North Carolina, and Savannah, Georgia, USA. Hiring policy The Company's hiring policy with regards to disability, belief, sex and sexual orientation is discussed in the Corporate Governance Report. Carbon dioxide emissions The Strategic Report discusses the Company's carbon dioxide emissions. Post Period Events On January 31, 2024, Mr. Rishi Gupta resigned as a non-executive director of the Board, effective as of January 31, 2024, and the Board appointed Mr. Michael Austwick as a non-executive director, effective as of February 1, 2024. Independent auditors At the Company's Annual General Meeting held on April 27, 2023, the Company's shareholders appointed PricewaterhouseCoopers LLP ("PwC") as the Company's independent auditors for the year ended December 31, 2023. On December 14, 2023, the Audit and Risk Committee of the Board did not re-appoint PwC and approved the engagement of Ernst & Young LLP ("EY") to serve as the Company's independent auditors for the year ending December 31, 2024, contingent upon the appointment of EY as the Company's independent auditors by the Company's shareholders at the Annual General Meeting to be held on April 26, 2024. In accordance with Section 489 of the Companies Act 2006, a resolution proposing that EY be appointed as independent auditors of the Company and that the Directors be authorized to approve their remuneration will be proposed at the Annual General Meeting. Annual General Meeting A notice of Annual General Meeting of the Company will be sent out in due course, setting out time, date and location of the meeting, together with the resolutions relating to the business which the Company proposes to conduct at such meeting. VERONA PHARMA PLC DIRECTORS' REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 5 |
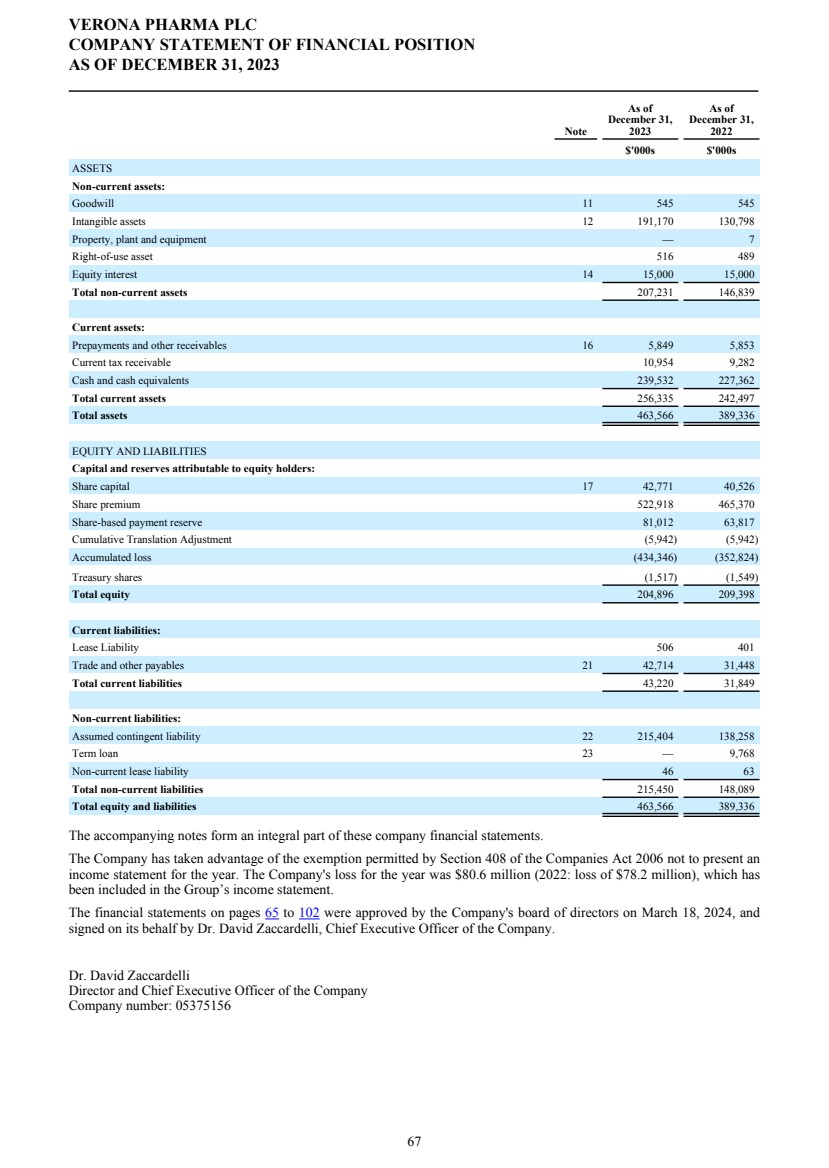
| Statement of Directors’ responsibilities The Directors are responsible for preparing the Annual Report and the financial statements in accordance with applicable laws and regulations. Company law requires the Directors to prepare financial statements for each financial year. Under that law, the Directors have prepared the group and company financial statements in accordance with U.K.-adopted international accounting standards. Under company law, the Directors must not approve the financial statements unless they are satisfied that they give a true and fair view of the state of affairs of the group and company and of the profit or loss of the group for that period. In preparing the financial statements, the Directors are required to: • select suitable accounting policies and then apply them consistently; • state whether applicable U.K.-adopted international accounting standards have been followed, subject to any material departures disclosed and explained in the financial statements; • make judgements and accounting estimates that are reasonable and prudent; and • prepare the financial statements on the going concern basis unless it is inappropriate to presume that the Group and Company will continue in business. The Directors are also responsible for safeguarding the assets of the Group and Company and hence for taking reasonable steps for the prevention and detection of fraud and other irregularities. The Directors are responsible for keeping adequate accounting records that are sufficient to show and explain the Group’s and Company’s transactions and disclose with reasonable accuracy at any time the financial position of the Group and Company and enable them to ensure that the financial statements and the Directors’ Remuneration Report comply with the Companies Act 2006. The Directors are responsible for the maintenance and integrity of the Company’s website. Legislation in the U.K. governing the preparation and dissemination of financial statements may differ from legislation in other jurisdictions. Directors’ confirmations In the case of each Director in office at the date the Directors’ Report is approved: • so far as the Director is aware, there is no relevant audit information of which the Group’s and Company’s auditors are unaware; and • they have taken all the steps that they ought to have taken as a director in order to make themselves aware of any relevant audit information and to establish that the Group’s and Company’s auditors are aware of that information. On behalf of the Board. Dr. David Zaccardelli Chief Executive Officer March 18, 2024 VERONA PHARMA PLC DIRECTORS' REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 6 |
| CORPORATE GOVERNANCE REPORT It is the Board's belief that good corporate governance is integral to a successful business, and the Company seeks to apply the highest standards of corporate governance appropriate to its size and stage of development. THE BOARD OF DIRECTORS At December 31, 2023, the Board comprised ten non-Executive Directors, and one Executive Director. The Board, through its Nomination and Governance Committee, regularly reviews its composition to ensure that it has a sufficiently wide range of skills and experience to enable it to pursue its strategic goals and to address anticipated issues in the foreseeable future. As part of this process, the Board appointed Ms. Christina Ackermann, on September 1, 2023, and Mr. Michael Austwick, post period end on February 1, 2024, as non-Executive Directors. Mr. Rishi Gupta stepped down from the Board as a Non-Executive Director on January 31, 2024. Ms. Ackermann brings over 25 years of legal and management experience across the pharmaceutical, device and consumer products industries. Most recently, Ms. Ackermann served as Executive Vice President, General Counsel and Global President Ophthalmic Pharmaceuticals at Bausch + Lomb Corporation where she was responsible for strategic planning and worldwide commercialization of the pharmaceutical prescription assets across the portfolio as well as global legal affairs. During her tenure as President Ophthalmic Pharmaceuticals several new products were launched globally. Previously, Ms. Ackermann was part of the Novartis group of companies for 14 years where she served in multiple leadership roles of increasing responsibility including Global Head, Legal and General Counsel of Alcon and Sandoz. Ms. Ackermann currently serves as a Non-Executive Director on the board of Oculis Holding AG. She holds a Post Graduate Diploma in EC Competition Law from King’s College at the University of London, UK, and a LLB from Queen’s University, Kingston, Canada. Mr. Austwick has a wealth of strategic and operational experience in the biopharmaceutical industry including more than 15 years of respiratory expertise in leadership roles across the US, China, Europe and Japan. Most recently, he served as CEO at Vectura, and previously as Nordic General Manager and Head of the Global Respiratory Franchise at Novartis and as Head of US Respiratory and Vice President Global Inhaled Respiratory at AstraZeneca. Mr. Austwick has led the development and commercialization of more than 10 brands, covering inhaled and biologic assets at a country and global level. He has an MSc in Information Management from the University of Hull and a BSc in Management and Systems from City University, London. The Board has also considered and concluded that the appointment of a Senior Independent Director is not necessary at this time, but keeps this issue under review. The Board typically has four scheduled meetings per year (approximately once every quarter), with additional Board meetings and Board committee meetings convened as circumstances and business needs dictate. The Board is responsible to the shareholders for the proper management of the Company and sets the overall direction and strategy of the Company, and reviews scientific, operational and financial performance. All key operational and investment decisions are subject to Board approval. There is a clear separation of the roles of Chief Executive Officer and non-Executive Chairperson. The non-Executive Chairperson is responsible for overseeing the running of the Board, ensuring that no individual or group dominates the Board’s decision-making and ensuring the non-Executive Directors are properly briefed on matters. The Chief Executive Officer has the responsibility for implementing the strategy of the Board and managing the day to day business activities of the Company. In accordance with our Articles of Association, one third of our directors retire from office at every annual general meeting of shareholders. However, if the number of directors serving on our Board is not divisible by three, then the number nearest but not exceeding 33.3% shall retire from office at each annual general meeting of shareholders. Retiring directors are eligible for re-election and, if no other director is elected to fill his or her position and the director is willing, shall be re-elected by default. The Board has considered the guidelines on independence and regards Dr. David Ebsworth, Ms. Christina Ackermann, Mr. Michael Austwick, Mr. James Brady, Dr. Ken Cunningham, Ms. Lisa Deschamps, Dr. Martin Edwards, Mr. Rishi Gupta, Dr. Mahendra Shah, Mr. Vikas Sinha and Dr. Anders Ullman as independent directors. Although the non-Executive Directors have been awarded equity awards under the Company's 2017 Incentive Plan, the Board considers that the grant of equity awards is aligned with U.S. best practice for comparable Nasdaq-listed companies. The Board is also satisfied that each non-executive director continues to demonstrate independence of character and judgement with respect to his or her non-executive directors duties. VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 7 |
| BIOGRAPHIES David Zaccardelli, Pharm.D. Dr. Zaccardelli has served as our President and Chief Executive Officer and on our board of directors since February 2020. From December 2018 until its acquisition by Swedish Orphan Biovitrum for up to $915 million in November 2019, Dr. Zaccardelli served as President and CEO of Dova Pharmaceuticals, a U.S. company developing therapeutics for rare diseases. Previously, he was Acting CEO of Cempra, from December 2016 until the company’s merger with Melinta Therapeutics in November 2017. From 2004 until 2016, Dr Zaccardelli served in several senior management roles at United Therapeutics Corporation, including Chief Operating Officer, Chief Manufacturing Officer and Executive Vice President, Pharmaceutical Development and Operations. Prior to United Therapeutics, he founded and led a start-up company focused on contract research positions and held a variety of clinical research positions at Burroughs Wellcome & Co, Glaxo Wellcome, and Bausch & Lomb Pharmaceutical. Dr. Zaccardelli received a Pharm.D. from the University of Michigan. David Ebsworth, Ph.D. Dr. Ebsworth has served as the Non-Executive Chairperson of our board of directors since December 2014. From October 2009 to August 2014, Dr. Ebsworth served as Chief Executive Officer of Vifor Pharma, based in Zürich, the specialty pharma division of Galenica AG Group, a pharmaceutical wholesaler and retailer, and as a member of Galenica's Executive Committee. In 2012, Dr. Ebsworth was also named as Chief Executive Officer of Galenica and as Chair of Galenica's Executive Committee, positions he held until August 2014. In his earlier career, Dr. Ebsworth worked with Bayer AG for over 19 years, heading the Canadian, North American and global pharmaceutical business. He also served as Chief Executive Officer of Oxford Glycosciences, a biotech company, listed on the London Stock Exchange and Nasdaq, which was acquired by Celltech plc (now part of UCB) in 2003. Dr. Ebsworth currently serves on the boards of Synlab AG and Sartorius AG and Kyowa Kirin International. He received a Ph.D. in industrial relations from the University of Surrey. Christina Ackermann. Ms. Ackermann was appointed to the board as a Non-Executive Director in September 2023. She brings over 25 years of legal and management experience across the pharmaceutical, device and consumer products industries. Most recently, Ms. Ackermann served as Executive Vice President, General Counsel and Global President Ophthalmic Pharmaceuticals at Bausch + Lomb Corporation where she was responsible for strategic planning and worldwide commercialization of the pharmaceutical prescription assets across the portfolio as well as global legal affairs. During her tenure as President Ophthalmic Pharmaceuticals several new products were launched globally. Previously, Ms. Ackermann was part of the Novartis group of companies for 14 years where she served in multiple leadership roles of increasing responsibility including Global Head, Legal and General Counsel of Alcon and Sandoz. Ms. Ackermann currently serves as a Non-Executive Director on the board of Oculis Holding AG. She holds a Post Graduate Diploma in EC Competition Law from King’s College at the University of London, U.K., and a LLB from Queen’s University, Kingston, Canada. Michael Austwick. Mr. Austwick was appointed to the board as a Non-Executive Director in February 2024. He has a wealth of strategic and operational experience in the biopharmaceutical industry including more than 15 years of respiratory expertise in leadership roles across the U.S., China, Europe and Japan. Most recently, he served as CEO at Vectura, and previously as Nordic General Manager and Head of the Global Respiratory Franchise at Novartis and as Head of U.S. Respiratory and Vice President Global Inhaled Respiratory at AstraZeneca. Mr. Austwick has led the development and commercialization of more than 10 brands, covering inhaled and biologic assets at a country and global level. He has an MSc in Information Management from the University of Hull and a BSc in Management and Systems from City University, London. James Brady. Mr. Brady was appointed to the board as a Non-Executive Director in March 2022. Mr. Brady has extensive experience in the biopharmaceutical industry, serving in multiple leadership roles of increasing responsibility across the US, Europe and China, during his 30-plus-year career at AstraZeneca. Most recently, Mr. Brady served as Chief Financial Officer of MedImmune, the biologics discovery and development division of AstraZeneca. During his tenure at MedImmune, biologics grew to represent more than half of the product development portfolio of AstraZeneca and five biologics were successfully brought to market. Mr. Brady currently serves as a Non-Executive Director on the board of Panavance Therapeutics. He is a Certified Public Accountant, holds an MBA from Drexel University and a BSc in accounting from St. Joseph University. Ken Cunningham, M.D. Dr. Cunningham has served as a Non-Executive Director on our board of directors since September 2015. Dr. Cunningham has over 30 years’ experience in the pharmaceutical industry including leadership roles at several companies focused on developing respiratory medicines. Between 2008 and 2010, he was at SkyePharma plc (now part of Vectura Group plc), initially as Chief Operating Officer and subsequently as Chief Executive Officer where he was involved in the late-stage development of flutiform® for asthma. Earlier in his career, Dr. Cunningham held a variety of clinical development and commercial strategy roles at Glaxo Wellcome plc and Warner-Lambert. Dr. Cunningham serves as Non-Executive Chairperson of the board of directors of Medherant Ltd. Dr. Cunningham received a degree in medicine from St. Mary’s, Imperial College, London University. Lisa Deschamps. Ms. Deschamps has served as a Non-Executive Director since March 2021. Ms. Deschamps is CEO and executive board member of AviadoBIO Ltd, a private gene therapy company. Prior to joining AviadoBIO Ltd, she was VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 8 |
| Senior Vice President and Chief Business Officer of Novartis Gene Therapies and previously was Head of Novartis’ Global Neuroscience Franchise. During her 25-year career at Novartis AG, Ms. Deschamps gained significant global and U.S. experience in bringing respiratory and other specialized therapeutic area products from the clinic to commercialization. Ms. Deschamps has an MBA in General Management from NYU Stern School of Business and a BBA in marketing from IONA College, Hagan School of Business. Martin Edwards, M.D. Dr. Edwards has served as a Non-Executive Director on our board of directors since April 2019. Previously, he was Senior Partner at Novo Ventures, life sciences investment firm, from 2003-2020, and Corporate VP and Corporate VP and Global Head of Drug Development for Novo Nordisk, where he led all aspects of pre-clinical and clinical drug development. Dr. Edwards currently serves on the boards of directors of Inozyme Pharma Inc and Morphic Therapeutic Inc. Dr. Edwards trained in physiology and medicine at the University of Manchester. He is a Member of the Royal College of Physicians, a Member with distinction of the Royal College of General Practitioners, a Fellow of the Faculty of Pharmaceutical Medicine and holds a MBA from the University of Warwick. Rishi Gupta. Mr. Gupta has served as a Non-Executive Director on our board of directors since July 2016. Mr. Gupta was designated for appointment to our board of directors by OrbiMed Private Investments VI, LP, or OrbiMed, pursuant to our relationship agreement with OrbiMed. Since 2002, Mr. Gupta has held various positions at OrbiMed Advisors LLC, an investment firm, where he is currently a Partner. Prior to that, he was a healthcare investment banker at Raymond James & Associates and served as manager of corporate development at Veritas Medicine. Mr. Gupta currently is a member of the board of directors of Enliven Therapeutics, Inc and several private companies. Mr. Gupta received an A.B. in biochemical sciences from Harvard College and a J.D. from Yale Law School. Mahendra Shah, Ph.D. Dr. Shah has served as a Non-Executive Director on our board of directors since July 2016. Dr. Shah was designated for appointment to our board of directors by funds affiliated with Vivo Capital pursuant to our relationship agreement with such funds. Dr. Shah is a successful pharmaceutical entrepreneur and executive and Senior Fellow of Vivo Capital, a healthcare investment firm, where he was formerly a Managing Partner. Dr. Shah previously served as a member of the board of directors of Soleno Therapeutics Inc, Crinetics Pharmaceuticals Inc, Aadi Bioscience Inc and Homology Medicines Inc. He currently serves as a member of the board of directors of several private companies in the biopharmaceutical and biotechnology industries. Dr. Shah received his Ph.D. in industrial pharmacy from St. John’s University and a Master’s Degree in Pharmacy from L.M. College of Pharmacy in Gujarat, India. Vikas Sinha. Mr. Sinha has served as a Non-Executive Director on our board of directors since September 2016. Mr. Sinha has over 20 years’ experience working in executive finance roles in the life sciences industry. Mr. Sinha is co-founder and Chief Financial Officer of ElevateBio, Inc, a holding company focused on building cell and gene therapy companies. He also serves as President and Chief Financial Officer of AlloVir, Inc, an ElevateBio portfolio company. From 2005 to 2016, Mr. Sinha was the Chief Financial Officer of Alexion Pharmaceuticals, Inc, a biotechnology company. Prior to joining Alexion, Mr. Sinha held various positions with Bayer AG in the U.S., Japan, Germany and Canada, including Vice President and Chief Financial Officer of Bayer Pharmaceuticals Corporation in the U.S. and Vice President and Chief Financial Officer of Bayer Yakuhin Ltd. in Japan. Mr. Sinha holds a master's degree in business administration from the Asian Institute of Management. He is also a qualified Chartered Accountant from the Institute of Chartered Accountants of India and a Certified Public Accountant in the U.S. Anders Ullman, M.D., Ph.D. Dr. Ullman has served as a Non-Executive Director on our board of directors since September 2015. Since May 2023, Dr. Ullman has served as a Non-Executive Director on the board of Sobi, where he was Head of Research and Development and Chief Medical Officer from January 2016 to March 2023. From 2016 to 2021, he was Head of the COPD Centre at Sahlgrenska University Hospital, Sweden, and from 2013 to 2014, was Executive Vice President and Head of Research and Development in the BioScience business unit of Baxter International Inc, a healthcare company, which became Baxalta Inc. From 2007 to 2013, Dr. Ullman was Executive Vice President, Head of Research and Development at Nycomed Pharma Private Limited (now part of Takeda Pharmaceuticals Company Limited), where he led the development and approval of Daxas, the PDE4 inhibitor used to prevent COPD exacerbations. Earlier in his career, he held a number of roles in AstraZeneca. Dr. Ullman received a M.D. and a Ph.D. in clinical pharmacology from the University of Gothenburg. VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 9 |
| Committees of our Board of Directors Our Board has three standing committees: an Audit and Risk Committee, a Remuneration Committee and a Nomination and Corporate Governance Committee. The composition and scope of the Audit and Risk Committee of the Board is described further below, within the Audit and Risk Committee Report. Remuneration Committee of the Board The Remuneration Committee, which consists of Dr. Ken Cunningham, Dr. David Ebsworth and Dr. Mahendra Shah, assists the Board in determining directors’ and executive officers’ compensation. Dr. Cunningham serves as Chairperson of the Committee. The Remuneration Committee's responsibilities include, among other things: • identifying, reviewing and proposing policies relevant to the compensation of the Company’s directors, executive officers and senior executives; • evaluating each executive officer's performance in light of such policies and reporting to the Board; • analyzing the possible outcomes of the variable remuneration components and how they may affect the remuneration of the executive officers; • recommending any equity long-term incentive component of each executive officer's compensation in line with the remuneration policy and reviewing our executive officer compensation and benefits policies generally; • appointing and setting the terms of engagement for any remuneration consultants who advise the Committee and obtain benchmarking data with respect to the directors' and executive officers’ compensation; and • reviewing and assessing risks arising from our compensation policies and practices. The Directors' Remuneration Report is presented on pages 36 to 57. Nomination and Corporate Governance Committee of the Board The Nomination and Corporate Governance Committee, which consists of Dr. David Ebsworth, Lisa Deschamps and Vikas Sinha, assists our Board in identifying individuals qualified to become executive and non-executive directors of our Company consistent with criteria established by our Board and in developing our corporate governance principles. Dr. Ebsworth serves as Chairperson of the Committee. The Nomination and Corporate Governance Committee's responsibilities include, among other things: • reviewing and evaluating the structure, size and composition of our Board and making recommendations with regard to any adjustments considered necessary; • drawing up selection criteria and appointment procedures for Board members; • identifying and nominating, for the approval of our Board, candidates to fill vacancies on the Board and its corresponding committees; • keeping under review the leadership needs of the Company, both executive and non-executive, and planning the orderly succession of such appointments; and • assessing the functioning of our Board and individual members and reporting the results of such assessment to the Board. VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 10 |
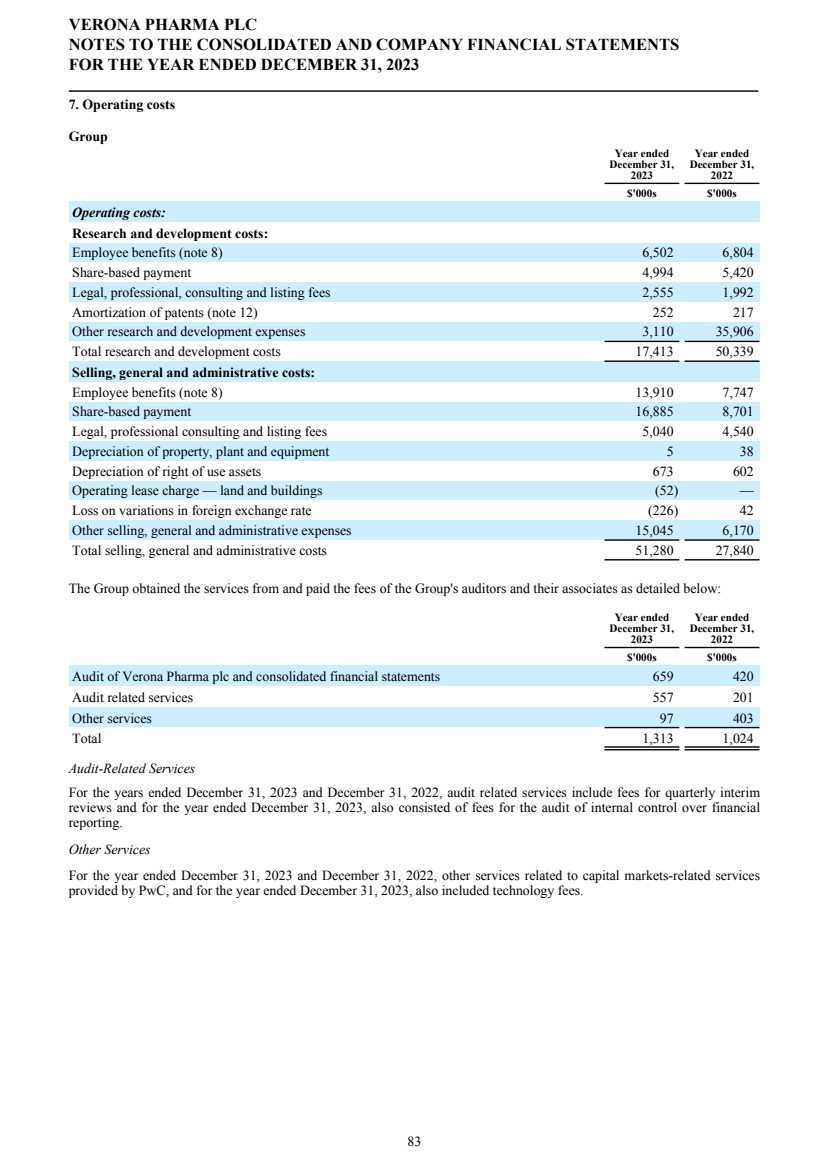
| AUDIT AND RISK COMMITTEE REPORT In this Report, we describe the work of the Audit and Risk Committee and the significant issues considered in 2023. Audit and Risk Committee of the Board The Audit and Risk Committee, which consists of Mr. Vikas Sinha, Dr. David Ebsworth, Ms. Christina Ackermann and Mr. James Brady, assists the Board in overseeing our accounting and financial reporting processes and the audits of our financial statements. Mr. Sinha serves as Chairperson of the Audit and Risk Committee. The Audit and Risk Committee consists of members of our Board who are financially literate and are also considered to be "audit committee financial experts" as defined by applicable SEC rules and have the requisite financial sophistication as defined under the applicable Nasdaq rules and regulations. Our Board has determined that all of the members of the Audit and Risk Committee satisfy the "independence" requirements set forth in Rule 10A-3 under the Securities Exchange Act of 1934. The Audit and Risk Committee is governed by a charter that complies with Nasdaq rules. The Audit and Risk Committee's responsibilities include, among other things: • recommending the appointment of the independent auditors to the general meeting of shareholders; • the appointment, compensation, retention and oversight of the independent auditors; • pre-approving the audit services and non-audit services to be provided by the independent auditors before the auditors are engaged to render such services; • evaluating the independent auditors' qualifications, performance and independence, and presenting its conclusions to our Board on at least an annual basis; • reviewing and discussing with the executive officers, our Board and the independent auditors our financial statements and our financial reporting process; • considering and recommending to our Board whether the audited financial statements be approved; • monitoring and reviewing the Company’s internal control over financial reporting and disclosure controls and procedures; and • monitoring our review and mitigation of corporate and operational risks, including the Company’s financial and cybersecurity risks. The Audit and Risk Committee meets as often as one or more members of the Committee deem necessary, but in any event must meet at least four times per year. The Audit and Risk Committee must meet at least once per year with our independent auditors, without our executive officers being present. Risk Identification and Management The Audit and Risk Committee monitors the Company’s approach to risk management. Management review the Company's risks on an ongoing basis and consider both corporate and project risk, which is risk relating the Company's sole product candidate, ensifentrine. Management reports their risk assessment to the Committee analyzing risk by severity and probability of occurrence. They also discuss mitigation strategies that have been or are intended to be implemented. Independent Auditors PwC has been the Group’s auditors since 2016. PwC operates procedures to safeguard against the possibility of their objectivity and independence being compromised. This includes the use of quality review partners, consultation with internal compliance teams and the carrying out of an annual independence procedure within their firm. PwC report to the Audit Committee on matters including independence and non-audit fees on an annual basis. The audit partner changes every five years. The amount charged by the external auditors for the provision of services during the twelve month period under review is set out in note 8 to the Financial Statements. The Committee assesses the performance of the auditors and is comfortable that PwC has operated effectively. On December 14, 2023, the Audit and Risk Committee did not re-appoint PwC and approved the engagement of EY to serve as the Company’s auditors for the year ending December 31, 2024, contingent upon the appointment of EY as the Company’s independent auditor by the Company’s shareholders at its Annual General Meeting. Subject to the Shareholder Appointment, EY will replace PwC as the Group’s auditor, which is not being nominated for re-appointment by the shareholders and whose term as independent auditor is expected to end following the Company’s Annual General Meeting. Internal Control The Audit and Risk Committee reviews the Group's internal control framework. The Group does not have an internal audit function and so the Committee has engaged an external firm of accountants to test management's systems of internal control. Any significant control deficiencies and mitigation strategies are reported to the Committee for review. The Board is responsible for the systems of internal control and for reviewing their effectiveness. The internal controls are designed to manage rather than eliminate risk and provide reasonable but not absolute assurance against material VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 11 |
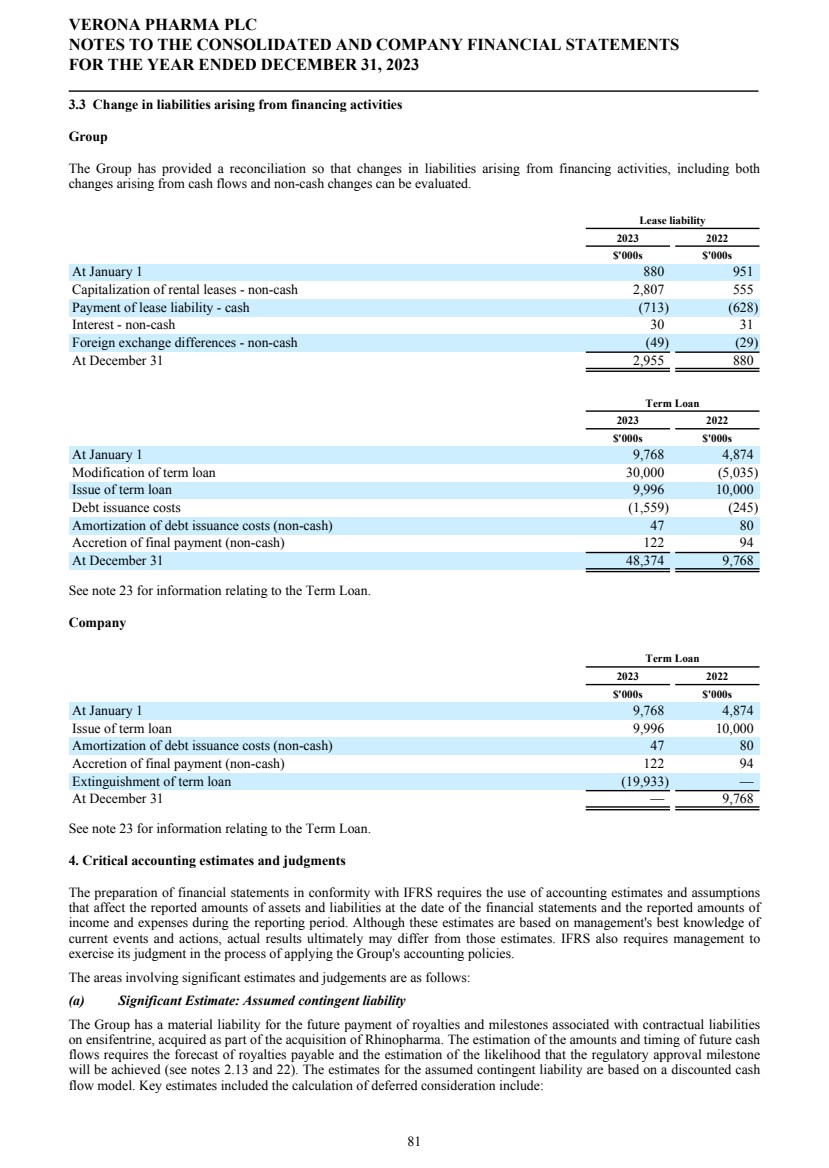
| misstatement or loss. The Board reviews the effectiveness of these systems quarterly by considering the risks potentially affecting the Group. Significant financial reporting issues considered by the Committee in 2023 The Audit and Risk Committee considers risk areas in the financial statements throughout the year and before the audit commences. The Committee considered the following items to be areas of risk: Ligand contingent liability The Group has a material liability for the future payment of a milestone and royalties associated with contractual liabilities over ensifentrine, its development product acquired as part of the acquisition of Rhinopharma. The liability is measured at amortized cost. At each reporting date the liability is re-measured where there are changes in estimated cashflows or probabilities of success. The contingent liability therefore requires quarterly re-assessment for any such triggering event. In the year-ended December 31, 2021, the Group entered into the Nuance Agreement. Consequently, the Group estimated potential cashflows from that agreement and the related royalties payable to Ligand, and remeasured the liability accordingly. For the years ended December 31, 2023 and 2022, management also reviewed the timing of expected royalties from the maintenance treatment of COPD in the U.S. and amended the sales forecasts to reflect the Group's expected timelines and expected sales. Management also amended the probability of success in the U.S. market as a result of the U.S. FDA accepting for review the Group's NDA seeking approval of ensifentrine for the maintenance treatment of COPD. The Committee reviewed and agreed with management's estimates of potential royalties payable, the timing of the expected sales that drive them and the update to probability of success. Accounting for the modification of a term loan facility On December 27, 2023 (the “2023 Effective Date”) Verona Pharma, Inc. entered into a term loan facility of up to $400.0 million (the “2023 Term Loan” or “Loan Agreement”), consisting of a term loan advance in an aggregate amount of $50.0 million funded on the 2023 Effective Date (the “Term A Loan”) and four additional term loan advances subject to certain terms and conditions. The 2023 Term Loan replaced the Group's existing $150.0 million facility. Verona Pharma, Inc. received net proceeds from the Term A Loan of $28.4 million which primarily consisted of the Term A Loan proceeds of $50.0 million partially offset by the repayment, in full, of the existing outstanding indebtedness owed by the Company under the Oxford Term Loan of $20.0 million, lender and third-party fees related to the Loan Agreement of $1.4 million and interest amounts of $0.2 million. Under IFRS 9, debt may be considered extinguished when it has been modified and the terms of the new debt instruments and old debt instruments are “substantially different". Group Based upon management's accounting evaluation of the Loan Agreement, as well as the Oxford entities involved and terms of both the 2023 Term Loan and the Oxford Term Loan, management has applied modification accounting to the portion of the Term A Loan associated with Oxford. The portion of the Term A Loan associated with Hercules Capital, Inc. has been accounted for as the issuance of new debt with the applicable accounting applied. Company As the Loan Agreement was entered into by Verona Pharma, Inc. and settled the prior term debt outstanding under the Oxford Term Loan, extinguishment accounting was applied to the prior term debt held by the Company. VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 12 |
| RISK MANAGEMENT AND INTERNAL CONTROL In connection with Verona Pharma plc's Annual Report on Form 10-K filed with the U.S. Securities and Exchange Commission pursuant to the Securities Exchange Act of 1934, as amended, management is required to assess and report on the effectiveness of its internal controls over financial reporting under Section 404(a) of the Sarbanes-Oxley Act. As Verona Pharma plc no longer qualifies as an ‘emerging growth company,’ as defined in the Jumpstart Our Business Start-Ups Act of 2012, or as a ‘smaller reporting company,’ we are now also required to comply with Section 404(b) of the Sarbanes-Oxley Act, which requires Verona Pharma plc’s auditor to attest to, and report on, management’s assessment of its internal controls. The Group has hired a third party firm to perform certain internal audit functions to, among other things, further assist with management’s assessment on the effectiveness of the Group’s internal controls. The need for the Group to have an internal audit function is evaluated on an annual basis. A comprehensive budgeting process is completed once a year, shortly prior to the start of each new financial year, which is reviewed and approved by the Board. Internal financial statements are produced on a monthly basis, with all significant variances investigated. These financial statements are reviewed and commented on by the Board at board meetings and are reviewed on a monthly basis by management and budget holders. The Group maintains appropriate insurance cover, including in respect of actions taken against the Directors because of their roles, as well as against material loss or claims against the Group. The insured values and type of cover are comprehensively reviewed on an annual basis. VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 13 |
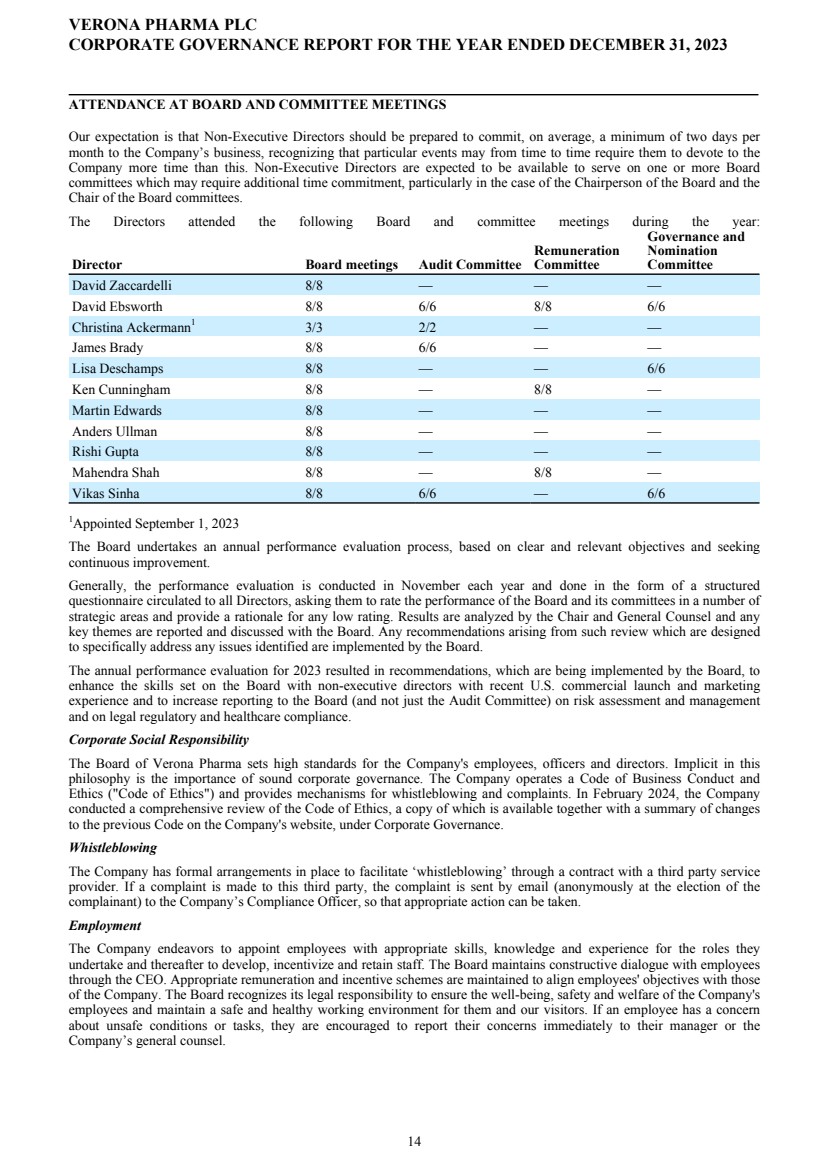
| ATTENDANCE AT BOARD AND COMMITTEE MEETINGS Our expectation is that Non-Executive Directors should be prepared to commit, on average, a minimum of two days per month to the Company’s business, recognizing that particular events may from time to time require them to devote to the Company more time than this. Non-Executive Directors are expected to be available to serve on one or more Board committees which may require additional time commitment, particularly in the case of the Chairperson of the Board and the Chair of the Board committees. The Directors attended the following Board and committee meetings during the year: Director Board meetings Audit Committee Remuneration Committee Governance and Nomination Committee David Zaccardelli 8/8 — — — David Ebsworth 8/8 6/6 8/8 6/6 Christina Ackermann1 3/3 2/2 — — James Brady 8/8 6/6 — — Lisa Deschamps 8/8 — — 6/6 Ken Cunningham 8/8 — 8/8 — Martin Edwards 8/8 — — — Anders Ullman 8/8 — — — Rishi Gupta 8/8 — — — Mahendra Shah 8/8 — 8/8 — Vikas Sinha 8/8 6/6 — 6/6 1Appointed September 1, 2023 The Board undertakes an annual performance evaluation process, based on clear and relevant objectives and seeking continuous improvement. Generally, the performance evaluation is conducted in November each year and done in the form of a structured questionnaire circulated to all Directors, asking them to rate the performance of the Board and its committees in a number of strategic areas and provide a rationale for any low rating. Results are analyzed by the Chair and General Counsel and any key themes are reported and discussed with the Board. Any recommendations arising from such review which are designed to specifically address any issues identified are implemented by the Board. The annual performance evaluation for 2023 resulted in recommendations, which are being implemented by the Board, to enhance the skills set on the Board with non-executive directors with recent U.S. commercial launch and marketing experience and to increase reporting to the Board (and not just the Audit Committee) on risk assessment and management and on legal regulatory and healthcare compliance. Corporate Social Responsibility The Board of Verona Pharma sets high standards for the Company's employees, officers and directors. Implicit in this philosophy is the importance of sound corporate governance. The Company operates a Code of Business Conduct and Ethics ("Code of Ethics") and provides mechanisms for whistleblowing and complaints. In February 2024, the Company conducted a comprehensive review of the Code of Ethics, a copy of which is available together with a summary of changes to the previous Code on the Company's website, under Corporate Governance. Whistleblowing The Company has formal arrangements in place to facilitate ‘whistleblowing’ through a contract with a third party service provider. If a complaint is made to this third party, the complaint is sent by email (anonymously at the election of the complainant) to the Company’s Compliance Officer, so that appropriate action can be taken. Employment The Company endeavors to appoint employees with appropriate skills, knowledge and experience for the roles they undertake and thereafter to develop, incentivize and retain staff. The Board maintains constructive dialogue with employees through the CEO. Appropriate remuneration and incentive schemes are maintained to align employees' objectives with those of the Company. The Board recognizes its legal responsibility to ensure the well-being, safety and welfare of the Company's employees and maintain a safe and healthy working environment for them and our visitors. If an employee has a concern about unsafe conditions or tasks, they are encouraged to report their concerns immediately to their manager or the Company’s general counsel. VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 14 |
| Diversity Policy The Company is fully committed to the elimination of unlawful and unfair discrimination and values the differences that a diverse workforce brings to the organization. The Company endeavors to not discriminate because of age, disability, gender reassignment, marriage and civil partnership, pregnancy and maternity, race (which includes color, nationality and ethnic or national origins), religion or belief, sex or sexual orientation. The Company will undertake an annual review of its policies and procedures to establish its position with regard to compliance and best practice, and monitor and promote a healthy corporate culture. Relations with shareholders The Board values good relations with the Company’s shareholders and understands the importance of effectively communicating the Company’s operational and financial performance as well as its future strategy. The Company’s website provides financial information as well as historical news releases and matters relating to corporate governance. The Chairperson of the Board and the CEO and CFO maintain ongoing dialogue with shareholders and communicate their views to the Board. The Board recognizes it is accountable to shareholders and ensures that their views are taken into account in agreeing the Company’s strategy and other operational matters. The Board also recognizes the importance of treating all shareholders equally. Annual and interim results are filed with the Securities and Exchange Commission and communicated by news services as are ad hoc operational and regulatory releases. Shareholders may also attend the Annual General Meeting where they can ask questions to the Board. Relations with suppliers The Company endeavors to maintain good relationships with its suppliers by contracting them on reasonable business terms and paying them promptly, within agreed terms. Management report to the Board on the performance of significant suppliers engaged for the development, manufacturing, sales and distribution of the Company's drug product to ensure that our research and development program and commercialisation activities are planned and delivered effectively in a timely and cost-efficient manner. This ensures interests are aligned between the Company and our significant suppliers. VERONA PHARMA PLC CORPORATE GOVERNANCE REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 15 |
| STRATEGIC REPORT The Directors present their strategic report together with the audited consolidated financial statements, audited company financial statements and auditors’ report for the year ended December 31, 2023. Principal activity The Company was incorporated on February 24, 2005. On September 18, 2006, the Company successfully acquired all the shares of Rhinopharma Limited, a private company incorporated in Canada, and changed its name from Isis Resources plc to Verona Pharma plc ("Verona Pharma", the “Company” or the “Parent”). On December 12, 2014, the Company established a U.S. subsidiary, Verona Pharma, Inc., in the state of Delaware. In June 2021, Rhinopharma Limited was dissolved. The Company, Rhinopharma Limited (until June 2021) and Verona Pharma, Inc. are collectively referred to as the “Group”. The principal activity of the Group is the development and commercialization of innovative therapeutics for the treatment of respiratory diseases with significant unmet medical need. Section 172(1) Companies Act 2006 The Directors are required by law to act in good faith to promote success of the Company for the benefit of the shareholders as a whole and are also required to have regard to the following: • the principal decisions made by the Board and the likely long-term consequences of any decision; • the interests of the Company's employees; • the need to foster the Company's business relationships with suppliers, customers and others; • the impact of the Company's operations on the community and the environment; • the desirability of the Company maintaining a reputation for high standards of business conduct; and • the need to act fairly as between shareholders of the Company. A discussion on how the Board has regard to these matters can be found on pages 14 and 15 of the Corporate Governance Report. The impact of the Company's operations on the environment is discussed further within "Greenhouse Gas Emissions" on page 33 in this Strategic Report. Outlook and Strategy in this Strategic Report describes the Group’s activities, strategy and future prospects, including the considerations for long-term decision making. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 16 |
| OUTLOOK AND STRATEGY We are a clinical-stage biopharmaceutical company focused on developing and commercializing innovative therapeutics for the treatment of chronic respiratory diseases with significant unmet medical needs. Our product candidate, ensifentrine, is an investigational, first-in-class, inhaled, selective, dual inhibitor of the enzymes phosphodiesterase 3 and 4 (“PDE3 and “PDE4”), combining bronchodilator and non-steroidal anti-inflammatory activities in one molecule. Initially, we are developing inhaled ensifentrine for the maintenance treatment of chronic obstructive pulmonary disease (“COPD”), a common, chronic, progressive, and life-threatening respiratory disease without a cure. If approved, ensifentrine is expected to be the first inhaled therapeutic with a novel mode of action for the maintenance treatment of COPD in over 20 years. In August 2023, the U.S. Food and Drug Administration (“FDA”) accepted for review our New Drug Application (“NDA”) seeking approval of ensifentrine for the maintenance treatment of COPD and assigned a Prescription Drug User Fee Act (“PDUFA”) target action date of June 26, 2024. The FDA stated it is not currently planning to hold an advisory committee meeting to discuss the application. Based on the results from our successful Phase 3 ENHANCE (“Ensifentrine as a Novel inHAled Nebulized COPD thErapy”) program, we believe ensifentrine, if approved, has the potential to change the treatment paradigm for COPD. Ensifentrine met the primary endpoint in both the ENHANCE-1 and ENHANCE-2 trials demonstrating statistically significant and clinically meaningful improvements in measures of lung function. In key secondary endpoints, ensifentrine demonstrated early and sustained improvements in symptoms and quality of life. In addition, other endpoint data demonstrated that ensifentrine substantially reduced the rate and risk of COPD exacerbations in ENHANCE-1 and ENHANCE-2. Ensifentrine was well tolerated in both trials. During 2023, we presented additional analyses of data from the ENHANCE trials at international scientific conferences and the data were published in peer reviewed publications: • In May 2023, we presented 12 abstracts and a symposium on expanded pre-specified and post hoc analyses of the ENHANCE trials including subgroup and pooled data covering exacerbations, use of rescue medication and healthcare utilization, at the American Thoracic Society International Conference ("ATS") 2023. An overview of the ENHANCE trial results was presented as part of the Clinical Trial Symposium reserved for highlighting new innovative medicines. In summary, ensifentrine demonstrated highly consistent results across all the clinically relevant subgroups and pooled analyses assessed including improvements in lung function and reductions in exacerbation rate and risk. Other key analyses demonstrated improvements with ensifentrine in symptoms and quality of life measures including SGRQ* subdomains, as well as reductions in the use of rescue medication and healthcare utilization. Furthermore, ensifentrine was shown to be well tolerated in an expanded safety analysis. *St. George’s Respiratory Questionnaire is a validated patient reported outcome tool • The abstracts were published on the ATS website and in the American Journal of Respiratory and Critical Care Medicine ("AJRCCM"); • In June 2023, the ENHANCE results were published in AJRCCM; • In September 2023, we presented an analysis of the ENHANCE-1 24-week exacerbation data at ERS International Congress 2023, which demonstrated treatment with ensifentrine substantially decreased the rate and risk of moderate and severe COPD exacerbations. The abstract was published in the European Respiratory Journal; and • In October 2023, we gave four presentations on pooled and subgroup post-hoc analyses from ENHANCE-1 and ENHANCE-2 covering data related to exacerbations, lung function, symptoms and quality of life endpoints and use of daily rescue medication, at CHEST Annual Meeting 2023. The data demonstrated treatment with ensifentrine substantially reduced the rate and risk of COPD exacerbations regardless of recent exacerbation history and was well tolerated. In addition, subgroup analyses showed treatment with ensifentrine resulted in improvements in lung function, symptoms, and quality of life measures, reductions in the rate and risk of exacerbations regardless of background therapy as well as reductions in daily rescue medication use. The data were published in the CHEST Annual Meeting online supplement. Also, at CHEST, we launched a disease awareness campaign highlighting how many COPD patients struggle to talk about their condition. While we remain focused on the US commercialization of ensifentrine, if approved, we are developing a fixed-dose combination formulation with ensifentrine and glycopyrrolate, a long-acting muscarinic antagonist (“LAMA”), for the maintenance treatment of patients with COPD via delivery in a nebulizer. Following development activities to confirm a feasible formulation, in the second half of 2024, the Company plans to submit an Investigational New Drug application (“IND”) to the FDA and, subject to clearance, initiate a Phase 2 clinical trial assessing the safety and efficacy of the fixed-dose combination formulation in COPD patients. Also in the second half of 2024, the Company plans to commence a Phase 2 clinical trial to assess the efficacy and safety of nebulized ensifentrine in patients with non-cystic fibrosis bronchiectasis (“NCFBE”), subject to clearance by the FDA. In Phase 2 clinical trials, ensifentrine has demonstrated positive results in patients with COPD, asthma and cystic fibrosis (“CF”). Two additional formulations of ensifentrine have been evaluated in Phase 2 studies for the treatment of COPD: dry VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 17 |
| powder inhaler (“DPI”) and pressurized metered-dose inhaler (“pMDI”). Ensifentrine has shown positive Phase 2 data in COPD trials when delivered by each of these formulations. We believe the development of ensifentrine in cystic fibrosis and asthma as well as the additional formulations of ensifentrine provides pipeline expansion and lifecycle opportunities as well as potential for collaborations outside the US. If approved, we intend to commercialize inhaled ensifentrine for the maintenance treatment of COPD in the United States (“U.S.”). Although we believe ensifentrine will not be regulated as a drug device combination, patients use a readily available standard jet nebulizer to take ensifentrine. Outside the U.S., we intend to license ensifentrine to companies with expertise and experience in developing and commercializing products in those regions. To that end, we have entered into a strategic collaboration with Nuance Pharma Limited, a Shanghai-based specialty pharmaceutical company ("Nuance Pharma"), to develop and commercialize ensifentrine in Greater China. In 2023, Nuance enrolled the first subject in its pivotal Phase 3 trial evaluating ensifentrine for the maintenance treatment of COPD in China. Senior executives bring substantial expertise as we prepare for potential commercialization During 2023, the Company continued to build its commercial capabilities and launch readiness in preparation for the potential approval of ensifentrine. Key pre-commercialization activities included the addition of experienced executives, launch of a disease awareness campaign, initial development of a branded campaign and extensive market research. We significantly expanded our headcount to approximately 80 employees adding key leadership positions across medical affairs, compliance, manufacturing, finance and IT and deepened our commercial teams in marketing, market access and commercial operations. These appointments included Senior Vice President, Medical Affairs, Vice President, Compliance, and Vice President, Pharmacovigilance. Overview of COPD and current treatments COPD is a common, progressive, life-threatening respiratory disease without a cure. It causes loss of lung function, leading to debilitating breathlessness, hospitalizations, and death. COPD has a major impact on everyday life. Patients struggle with basic activities such as getting out of bed, showering, eating, and walking. Worldwide, COPD affects approximately 392 million people and is the third leading cause of death, according to the Global Initiative for Chronic Obstructive Lung Disease. The goal of COPD pharmacological therapy is to improve patients’ quality of life by reducing symptoms, decreasing the quantity and severity of exacerbations (often an escalation of symptoms) and to improve patients’ ability to function (GOLD 202). For approximately 40 years, the treatment of COPD has been dominated by three classes of inhaled therapies approved for use by the FDA and the European Commission based on the European Medicines Agency's (“EMA”) opinion: anti-muscarinics, beta-agonists and inhaled corticosteroids (“ICSs”). COPD patients are frequently treated with bronchodilators, including LAMAs and long-acting beta-agonists (“LABAs”), to relieve airway constriction and make it easier to breathe. In addition, patients at risk for exacerbations may be prescribed ICSs to prevent them. Certain COPD patients are treated with the oral PDE4 inhibitor, roflumilast (Daliresp®), which has demonstrated a reduction in exacerbation risk in patients with severe chronic bronchitis. However, oral PDE4 therapy results in systemic exposure which has been associated with unfavorable gastrointestinal side-effects such as nausea, emesis, diarrhea, abdominal pain, loss of appetite and weight loss. Approximately 8.6 million COPD patients in the U.S. receive LAMA, LABA or ICS treatments alone or in combination regardless of COPD severity. Despite these medication and the earlier use of dual (LAMA / LABA) and triple (LAMA / LABA / ICS) therapies, many patients continue to suffer debilitating symptoms. According to a December 2022 study by Phreesia, 49% of patients continue to have symptoms more than 24 days a month. This burden leaves a significant opportunity for new inhaled therapies that offer additional benefit added to the three main classes of treatment. New treatment options are urgently needed to help improve lung function and symptoms, reduce exacerbations and improve overall quality of life in these patients. Ensifentrine Ensifentrine is an investigational, first-in-class, inhaled small molecule and selective dual PDE3 and PDE4 inhibitor. This dual inhibition enables it to act as a bronchodilator and a non-steroidal anti-inflammatory agent in a single compound. Importantly, ensifentrine's therapeutic profile differentiates it from existing classes of bronchodilator and anti-inflammatory treatments. We are not aware of any other single compound in clinical development in the U.S. or Europe or approved by the FDA nor the European Commission for the treatment of respiratory diseases that acts both as a bronchodilator and anti-inflammatory agent. If successfully developed and approved, ensifentrine has the potential to be the first novel class of inhaled therapeutic in COPD in over 20 years and to become the only bronchodilator option that could be added to existing classes of therapies including LAMA, LABA and ICS. Safety profile Ensifentrine has been well tolerated in clinical trials involving approximately 3,000 subjects to date. Additionally, ensifentrine did not prolong the QT interval or impact other cardiac conduction parameters in a thorough QT study in healthy volunteers. It is delivered directly to the lungs by inhalation to maximize pulmonary exposure to ensifentrine while VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 18 |
| minimizing systemic exposure. This feature minimizes any systemic side-effects such as the gastrointestinal disturbance associated with oral PDE4 inhibitors. In addition, in non-clinical trials ensifentrine has demonstrated high selectivity for PDE3 and PDE4 over other enzymes and receptors, which is believed to minimize off-target effects. Differentiated profile By selectively inhibiting PDE3 and PDE4, ensifentrine impacts three key mechanisms in respiratory disease: bronchodilation, inflammation and mucociliary clearance. Ensifentrine is designed to increase the levels of cellular cAMP and cGMP in smooth muscle cells and inflammatory cells, resulting in bronchodilator and anti-inflammatory effects. Ensifentrine has also be shown to stimulate the cystic fibrosis transmembrane conductance regulator (“CFTR”), which is an ion channel in the epithelial cells lining the airways. Mutations in the CFTR protein result in poorly or non-functioning ion channels, which cause CF. CFTR dysfunction is also potentially important in COPD. CFTR stimulation leads to improved electrolyte balance in the lung and thinning of the mucus, which facilitates mucociliary clearance and leads to improved lung function and potentially a reduction in lung infections. Dual inhibition of PDE3 and PDE4 has shown enhanced or synergistic effects compared with inhibition of either PDE alone on contraction of airway smooth muscle and suppression of inflammatory mediator release in several preclinical studies. We believe these enhanced effects may increase the utility of ensifentrine in the treatment of respiratory diseases including COPD, NCFBE, asthma and CF. The Company believes ensifentrine has the potential to address the large unmet need in treating COPD with its improvements in lung function, COPD symptoms and quality of life. Development of ensifentrine Clinical development of ensifentrine in COPD Phase 3 ENHANCE program Ensifentrine has successfully met the primary endpoints in two randomized, double-blind, placebo-controlled Phase 3 trials, ENHANCE-1 and ENHANCE-2, demonstrating statistically significant and clinically meaningful improvements in measures of lung function in moderate to severe COPD patients. Improvements in symptoms and quality of life measures were shown in both trials, which reached statistical significance in ENHANCE-1. Ensifentrine substantially reduced the rate and risk of moderate to severe COPD exacerbations in both trials. Ensifentrine was well tolerated in both trials. The ENHANCE trials were designed to evaluate ensifentrine as monotherapy and added onto a single bronchodilator with approximately 50% of subjects receiving either a LAMA or a LABA. Additionally, approximately 20% of subjects received ICSs with their concomitant LAMA or LABA. Each trial enrolled approximately 800 subjects, for a total of approximately 1,600 subjects, at sites primarily in the U.S. and Europe. The two trials provided replicate evidence of efficacy and safety data over 24 weeks and ENHANCE-1 also evaluated longer-term safety in approximately 400 patients over 48 weeks. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 19 |
| Subject demographics and disease characteristics were well balanced between treatment groups in both trials. • In ENHANCE-1 approximately 69% of subjects received background COPD therapy, either LAMA or a LABA. Additionally, approximately 20% of all subjects received ICS with concomitant LAMA or LABA. • In ENHANCE-2 approximately 55% of subjects received background COPD therapy, either a LAMA or a LABA. Additionally, approximately 15% of all subjects received ICS with concomitant LAMA or LABA. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 20 |
| We reported positive top-line results from ENHANCE-2 and ENHANCE-1, in August and December 2022, respectively. Ensifentrine successfully met the primary endpoints in both trials, demonstrating statistically significant and clinically meaningful improvements in measures of lung function in moderate to severe COPD patients. Improvements in symptoms and quality of life measures were shown in both trials, which reached statistical significance in ENHANCE-1. Ensifentrine substantially reduced the rate and risk of moderate to severe COPD exacerbations and was well tolerated in both trials. Highlights Primary endpoint met (FEV1 AUC 0-12 hr) • Placebo corrected, change from baseline in average FEV1 area under the curve 0-12 hours post dose at week 12 was 87 mL (p<0.0001) for ensifentrine in ENHANCE-1 and 94 mL (p<0.0001) for ensifentrine in ENHANCE-2. • Demonstrated consistent improvements with ensifentrine in all subgroups including gender, age, smoking status, COPD severity, background medication, ICS use, chronic bronchitis, FEV1 reversibility and geographic region. Secondary endpoints evaluating lung function met: • Placebo corrected, increase in peak FEV1 of 147 mL (p<0.0001) 0-4 hours post dose at week 12 in ENHANCE-1 and 146 mL (p<0.0001) in ENHANCE-2. • Placebo corrected, increase in morning trough FEV1 of 35 mL (p=0.0413) at week 12 in ENHANCE-1 and 49 mL (p=0.0016) in ENHANCE-2, supporting twice daily dosing regimen. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 21 |
| Exacerbation rate and risk reduced • Subjects receiving ensifentrine demonstrated a 36% reduction in the rate of moderate to severe COPD exacerbations over 24 weeks (p=0.0503) compared to those receiving placebo in ENHANCE-1 and a 43% reduction (p=0.0090) in ENHANCE-2. • In pooled exacerbation data from ENHANCE-1 and ENHANCE-2, ensifentrine demonstrated a 40% reduction in the rate of moderate to severe COPD exacerbations over 24 weeks (p=0.0012) compared to those receiving placebo. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 22 |
| • Treatment with ensifentrine significantly decreased the risk of a moderate/severe exacerbation as measured by time to first exacerbation when compared with placebo by 38% (p=0.0382) in ENHANCE-1 and by 42% (p=0.0089) in ENHANCE-2. • In pooled exacerbation data from ENHANCE-1 and ENHANCE-2, ensifentrine significantly decreased the risk of a moderate/severe exacerbation as measured by time to first exacerbation when compared with placebo by 41% (p=0.0009). VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 23 |
| COPD symptoms and Quality of Life (“QOL”) • In ENHANCE-1, daily symptoms as measured by E-RS* Total Score in the ensifentrine group improved from baseline to greater than the minimal clinically important difference (“MCID”) of -2 units with a statistically significant improvement compared to placebo at week 24. Improvements in symptoms were early and sustained with statistical significance versus placebo at weeks 6, 12 and 24. Similar improvements were demonstrated in ENHANCE-2 but statistical significance was not achieved due to improvements observed in the placebo group over time. • In ENHANCE-1, QOL as measured by SGRQ* Total Score in the ensifentrine group improved from baseline to greater than the MCID of -4 units with a statistically significant improvement compared to placebo at week 24. Improvements VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 24 |
| in QOL were early and sustained with statistical significance versus placebo at weeks 6, 12 and 24. In ENHANCE-2, QOL as measured by SGRQ* Total Score in the ensifentrine group also improved from baseline to greater than the MCID of -4 units at weeks 12 and 24, numerically exceeding placebo at each measurement, but statistical significance was not achieved due to improvements observed in the placebo group over time. *E-RS, Evaluating Respiratory Symptoms, and SGRQ, St. George’s Respiratory Questionnaire, are validated patient reported outcome tools Favorable safety profile • Ensifentrine was well tolerated with very few adverse events occurring in more than 1% of subjects and greater than placebo over 24 and 48 weeks. We believe ensifentrine, if approved, has the potential to change the treatment paradigm for COPD. The totality of data from clinical trials, in particular the top-line results from the ENHANCE program, including improvements in measures of VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 25 |
| lung function, symptoms, quality of life measures, and exacerbation reductions, coupled with the consistent safety results, support our belief. Formulations Verona Pharma has developed formulations of ensifentrine for the three most widely used inhalation devices: nebulizer, DPI and pMDI. The nebulized formulation of ensifentrine is designed to be suitable for use in a standard jet nebulizer, not a proprietary device. Delivery of COPD medications by nebulizer is important because such medications can be used by adults of almost any age and dexterity and regardless of peak inspiratory flow, offering advantages to patients who struggle to operate handheld inhaler devices or have low peak inspiratory flow. DPI and pMDI handheld inhaler formats are relatively portable and convenient and are also important delivery mechanisms. While we continue to focus on development of the nebulized formulation of ensifentrine, we believe the development of pMDI and DPI formulations of ensifentrine provides additional lifecycle opportunities including new potential indications, formulation combinations and collaborations. In February 2021, we reported positive results from the second, multiple dose part of a Phase 2 trial with pMDI ensifentrine in patients with moderate to severe COPD. Ensifentrine delivered by pMDI met all of the primary and secondary lung function endpoints. The improvement in lung function was dose-ordered and statistically significant at peak and over the 12-hour dosing interval compared with placebo, and supports twice-daily dosing of ensifentrine via pMDI for the treatment of COPD. Data from the single dose part of the study were reported in March 2020. Verona Pharma has successfully demonstrated proof of concept in Phase 2 COPD trials with all three formulations. In addition, the data from Phase 2 trials were consistent across the three formulations. All three dosage forms have demonstrated statistically significant and clinically meaningful improvements in lung function and duration of action, supporting twice-daily dosing and a safety profile similar to placebo. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 26 |
| Pipeline The following table summarizes our development programs. Planned Clinical Development Activities Ensifentrine / LAMA fixed-dose combination Fixed-dose combination therapies such as LABA / LAMA, LABA / ICS and LABA / LAMA / ICS are commonly used in the treatment of COPD and, based on our market research, an unmet need exists for a nebulized fixed-dose combination therapy. We believe the combination of ensifentrine with a LAMA could provide COPD patients with the first nebulized fixed-dosed combination with the potential to provide bronchodilation through a dual mechanism and also non-steroidal anti-inflammatory effects via PDE inhibition. We are developing a fixed-dose combination formulation with ensifentrine and glycopyrrolate, a LAMA, for the maintenance treatment of patients with COPD via delivery in a nebulizer. We have filed patent applications in multiple jurisdictions including the US. If a feasible formulation is developed, in the second half of 2024, the Company plans to submit an IND application to the FDA and, if allowed to proceed, initiate a Phase 2 clinical trial assessing the safety and efficacy of the fixed-dose combination formulation in COPD patients. Non-cystic fibrosis bronchiectasis NCFBE is a chronic lung disease characterized by persistent cough, excess sputum production and frequent respiratory infections with more severe patients suffering exacerbations. The condition affects up to 500,000 adults in the US and no therapies are specifically approved to treat it. Physicians currently use bronchodilators, antibiotics, steroids, mucus thinners and surgery. Based on the clinical results of ensifentrine observed in patients with COPD, including improvements in lung function and symptoms of cough and sputum, we believe that ensifentrine could potentially be an effective treatment for NCFBE. We plan to commence a Phase 2 clinical trial to assess the efficacy and safety of nebulized ensifentrine in patients with NCFBE in the second half of 2024, if allowed to proceed by the FDA. Potential additional indications for ensifentrine Cystic fibrosis and asthma In addition to COPD and NCFBE, we believe ensifentrine has potential applications in other respiratory diseases including NCFBE, CF and asthma providing pipeline expansion opportunities and the potential for collaborations outside the US.. CF is a progressive, fatal genetic disease without a cure and a median age of death of 46 years. The condition is characterized by thick, sticky mucus that damages many of the body’s organs. It causes repeat and persistent lung infections that result in frequent exacerbations and hospitalizations. Other symptoms include malnutrition, constipation and diarrhea, and some adults develop diabetes, arthritis and liver problems. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 27 |
| CF is the most common fatal inherited disease in the U.S. and Europe. Approximately 40,000 people in the U.S. and an estimated 105,000 people worldwide have been diagnosed with CF across more than 90 countries and approximately 1,000 new cases are diagnosed each year, according to the Cystic Fibrosis Foundation. The U.S. and European regulatory authorities consider CF to be a rare, or orphan, disease and provide incentives to encourage development of effective new treatments. CF patients endure multiple daily medications, frequent exacerbations and hospitalizations. Ultimately, selected patients have lung transplants. In a Phase 2a clinical trial, a single dose of nebulized ensifentrine demonstrated an improvement in lung function in patients with CF. In addition, in preclinical studies, ensifentrine activated the cystic fibrosis transmembrane conductance regulator ("CFTR"), which is beneficial in reducing mucous viscosity and improving mucociliary clearance. We believe these data support the continued development of ensifentrine as a potential therapy for CF. Asthma is a common chronic inflammatory lung condition that causes sporadic breathing difficulties. The disease causes narrowing and swelling of the airways leading to symptoms including difficulty breathing, wheezing, coughing and tightness in the chest. Exposure to triggers such as allergens or irritants can lead to asthma attacks. Asthma attacks vary in severity and frequency. More than 260 million people worldwide suffer from asthma and it is the most common chronic disease among children, according to estimates from the World Health Organization. Approximately 60% of adult asthmatics in the U.S. have uncontrolled asthma despite taking regular medication. Although there is no cure, symptoms may be prevented by avoiding triggers and through established maintenance therapies including bronchodilators, ICS, anti-IgE agents and leukotriene inhibitors. Ensifentrine has shown potential in a Phase 2a clinical trial in asthma. The data from this trial, published in October 2019 in the journal Pulmonary Pharmacology & Therapeutics, demonstrated that ensifentrine produced dose-dependent improvements in lung function that were comparable to current rescue medication, high dose nebulized albuterol. Importantly, ensifentrine was well tolerated and patients experienced fewer systemic effects than those receiving albuterol. Our team Our expert team has decades of experience in developing and commercializing respiratory therapeutics including the following COPD therapeutics: Advair® ; Anoro Ellipta® ; Breo® ; Flovent® ; Flutiform® ; Incruse Ellipta® ; Serevent® ; Symbicort® ; Tudorza Pressair® and Ventolin® .. MANUFACTURING We do not have manufacturing facilities and rely on, and expect to continue to rely on, third-party contract manufacturing organizations (“CMOs”) for the supply of current good manufacturing practices (“cGMP”) compliant clinical trial materials of ensifentrine, and any future product candidates, as well as for commercial quantities of ensifentrine and any future product candidates, if approved. We currently do not have any agreements for the long-term commercial production of ensifentrine. While we may contract with other CMOs in the future, we currently have one CMO for the manufacture of ensifentrine drug substance and one CMO for each formulation of ensifentrine. All of our current CMOs have commercial scale manufacturing capabilities. We believe that the ensifentrine drug substance and drug product manufacturing processes can be transferred to other CMOs to produce clinical and commercial supplies in the ordinary course of business. COMMERCIALIZATION During 2023, we continued to build our commercial capabilities and launch readiness in preparation for the potential approval of ensifentrine. Key pre-commercialization activities included the addition of experienced executives, launch of a disease awareness campaign, continued refinement and implementation of our patient support and distribution strategy as well as beginning development of our ensifentrine launch materials all supported by extensive market research, We significantly expanded our headcount to 78 employees adding key leadership positions across medical affairs, compliance, manufacturing, finance and IT and deepened our commercial teams in marketing, market access and commercial operations. These appointments included Senior Vice President, Medical Affairs, VP, Compliance, VP, Pharmacovigilance and Area Executive Directors of Sales. In addition, we launched a disease awareness campaign, titled "Unspoken COPD". This campaign highlights how many patients still suffer from persistent symptoms that effect everyday life. The campaign encourages healthcare professionals (“HCPs”) to enquire further to understand how their patients are coping with COPD. Looking ahead, we will continue to progress our go-to-market strategy with the finalization of many key tactics including pricing, distribution and patient support services, HCP and patient engagement plans and the continued rollout of our disease awareness campaign. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 28 |
| United States In the United States, we are preparing to commercialize nebulized ensifentrine ourselves, if approved. Current maintenance COPD treatments in the U.S. generate approximately $10 billion in sales. In the U.S., approximately 8.6 million patients receive chronic maintenance treatment for COPD. These patients receive LAMAs, LABAs, and ICS products alone or in combination across all COPD severities. Despite the use of these therapies, approximately 50% of patients report having symptoms for more than 24 days a month. This burden is significant and highlights the need for new and novel mechanisms of actions to treat COPD patients. These patients need therapies that can help improve their lung function and symptoms. In addition to the number of patients that remain symptomatic, COPD places a tremendous burden on the U.S. healthcare system with approximately $50 billion in direct and indirect costs. Based on our market research, conducted with U.S. healthcare providers and payers, we believe ensifentrine would be widely adopted with use as an add on therapy across all symptomatic patients regardless of COPD severity and treatment. Most of ensifentrine’s use would be as an add on therapy to current patients who are on LAMA, LABA / ICS, LAMA / LABA, or triple therapy. This is due to the urgent unmet need for new therapies to help improve lung function, symptoms and quality of life in these patients. Our market research also suggests the majority of ensifentrine usage would be initially commenced by pulmonologists. Due to this focused prescriber base, we anticipate a field sales force of approximately 100 representatives would be able to reach the potential ensifentrine opportunity. International COPD affects approximately 392 million people worldwide with many patients remaining undiagnosed. Our strategy outside of the U.S. including Asia, Europe and Latin America, is to establish partnerships with leading companies that can support the further development and commercialization of ensifentrine in those regions. In June 2021, we executed on this strategy by entering into a strategic collaboration with Nuance Pharma, a Shanghai-based specialty pharmaceutical company, with a potential value of up to $219.0 million to develop and commercialize ensifentrine in Greater China. Under the terms of the agreement, we granted Nuance Pharma the exclusive rights to develop and commercialize ensifentrine in Greater China. In return, we received an aggregate $40.0 million upfront payment consisting of $25.0 million in cash and an equity interest valued at $15.0 million, as of June 9, 2021, in Nuance Biotech, the parent company of Nuance Pharma. We are eligible to receive further milestone payments of up to $179.0 million that are triggered upon achievement of certain clinical, regulatory and commercial milestones as well as tiered double-digit royalties on net sales in Greater China. Nuance Pharma is responsible for all costs related to clinical development and commercialization in Greater China. A joint steering committee has been established to ensure ensifentrine’s clinical development in the region aligns with our global development and commercialization strategy. In April 2023, Nuance Pharma announced it had enrolled the first subject in its pivotal Phase 3 trial evaluating ensifentrine for the maintenance treatment of COPD in mainland China. Nuance Pharma initiated a Phase 1 trial with ensifentrine in healthy volunteers in March 2023. These studies follow clearance from China’s Center for Drug Evaluation for Nuance Pharma to begin Phase 1 and Phase 3 studies of ensifentrine for COPD in mainland China. COMPETITION The pharmaceutical industry is characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary drugs. We face potential competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions, governmental agencies and public and private research institutions. If successfully developed and commercialized, ensifentrine will compete with existing treatments and new treatments that may become available in the future. Ensifentrine is a unique, first-in-class therapeutic candidate with both bronchodilator and non-steroidal anti-inflammatory properties in a single molecule. As far as we are aware, no other dual PDE3 and PDE4 inhibitor is on the market nor in clinical development in the U.S. or Europe. Based on our market research, we expect ensifentrine to be used across the patient spectrum regardless of severity. We expect it will mainly be used as an add on therapy in symptomatic patients across all existing classes of therapies (LAMA, LABA, ICS). Some healthcare providers have indicated that they would use ensifentrine as a monotherapy based on ensifentrine’s clinical profile. Consequently, we believe that, if approved, nebulized ensifentrine’s unique profile will enable it to compete with all approved COPD therapies including nebulized and handheld inhaler formulations, DPI and pMDI. Furthermore, because ensifentrine’s mechanism of action is complementary to available therapies, we believe it could be used in addition to these treatments. Within the currently approved nebulizers for the maintenance treatment of COPD, we consider ensifentrine's potential competitors in the U.S. market to be LABAs (Brovana® and Perforomist® ) and LAMAs (Yupelri® ). In the DPI/pMDI maintenance treatment of COPD market, ensifentrine’s current closest potential competitors are Symbicort® , a combination of a long-acting beta2-agonist bronchodilator and ICS marketed by AstraZeneca plc, Spiriva® , a long-acting anti-muscarinic bronchodilator marketed by Boehringer Ingelheim GmbH, Advair® , a combination of a long-acting beta2-agonist bronchodilator and ICS marketed by GlaxoSmithKline plc, Breo® , a combination of a long-acting beta2-agonist bronchodilator and ICS marketed by GlaxoSmithKline, and Anoro® , a combination of a long-acting beta2- VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 29 |
| agonist bronchodilator and long-acting anti-muscarinic bronchodilator marketed by GlaxoSmithKline. A triple-combination therapy of a LAMA, a LABA and ICS, developed by GlaxoSmithKline, Trelegy Ellipta® , has been approved in the U.S. and the European Union and AstraZeneca also has a triple-therapy combination product (LAMA / LABA / ICS), Breztri Aerosphere® that was approved in the U.S. in July 2020, in the European Union in December 2020 and in China in December 2019. In addition, Chiesi’s triple-therapy combination product, Trimbow® , was approved in the European Union in 2017 and is in Phase 3 trials in the US. Other potential therapies in clinical development for the prevention of COPD exacerbations include injectable biologics. Sanofi’s anti-IL4, Dupixent® , has successfully completed a Phase 3 program and submitted a supplemental Biologics License Application for COPD in the US. AstraZeneca’s anti-IL33, tozorakimab, GlaxoSmithKline’s anti-IL5, Nucala® and Chiesi’s PDE4 inhibitor, Tanimilast, are in Phase 3 trials. We are also aware of several anti-inflammatories and bronchodilators that are in Phase 2 clinical trials for the treatment of COPD. INTELLECTUAL PROPERTY We strive to protect and enhance the proprietary technologies, inventions and improvements that we believe are important to our business, including seeking, maintaining and defending patent rights, whether developed internally or licensed from third parties. Our policy is to seek to protect our proprietary position by, among other methods, pursuing and obtaining patent protection in the U.S. and in jurisdictions outside of the U.S. related to our proprietary technology, inventions, improvements, platforms and our product candidates that are important to the development and implementation of our business. As of December 31, 2023, our patent portfolio included eleven issued U.S. patents, seven pending U.S. patent applications (including four U.S. provisional patent applications), seventy-nine issued foreign patents and seventy-five pending foreign applications (including six international PCT applications). These patents and patent applications include claims directed to certain respirable formulations comprising ensifentrine, a crystalline form of ensifentrine, combinations of ensifentrine with certain respiratory drugs, certain salts of ensifentrine, ensifentrine for use in the treatment of cystic fibrosis and non-cystic fibrosis bronchiectasis and for use in the treatment of certain aspects of some other respiratory disorders, and a method of making ensifentrine, with expected expiry dates up to 2044. Individual patents extend for varying periods depending on the date of filing of the patent application or the date of patent issuance and the legal term of patents in the countries in which they are obtained. Generally, patents issued for regularly filed applications in the U.S. are granted a term of 20 years from the earliest effective non-provisional filing date. In addition, in certain instances, a patent term can be extended to recapture a portion of the U.S. Patent and Trademark Office, or the USPTO, delay in issuing the patent as well as a portion of the term effectively lost as a result of the FDA regulatory review period. However, as to the FDA component, the restoration period cannot be longer than five years and the total patent term including the restoration period must not exceed 14 years following FDA approval. The duration of foreign patents varies in accordance with provisions of applicable local law, but typically is also 20 years from the earliest effective filing date. However, the actual protection afforded by a patent varies on a product-by-product basis, from country to country and depends upon many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patent. Furthermore, we rely upon trade secrets and know-how and continuing technological innovation to develop and maintain our competitive position. We seek to protect our proprietary information, in part, using confidentiality agreements with our collaborators, employees and consultants and invention assignment agreements with our employees. We also have confidentiality agreements or invention assignment agreements with our collaborators and selected consultants. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with a third party. These agreements may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our collaborators, employees and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions. Our commercial success will also depend in part on not infringing upon the proprietary rights of third parties. It is uncertain whether the issuance of any third-party patent would require us to alter our development or commercial strategies, or our drugs or processes, obtain licenses or cease certain activities. Our breach of any license agreements or failure to obtain a license to proprietary rights that we may require to develop or commercialize our future drugs may have an adverse impact on us. If third parties have prepared and filed patent applications prior to March 16, 2013 in the U.S. that also claim technology to which we have rights, we may have to participate in interference proceedings in the USPTO, to determine priority of invention. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 30 |
| FINANCIALS Comparison of Operations for the Years ended December 31, 2023 and 2022 The operating loss for the year ended December 31, 2023 was $68.7 million (2022: $78.1 million) and the loss after tax for the year ended December 31, 2023 was $72.3 million (2022: $75.4 million). Research and Development Costs Research and development costs were $17.4 million for the year ended December 31, 2023 compared to $50.3 million for the year ended December 31, 2022, a decrease of $32.9 million. This decrease was primarily due to a $32.7 million decrease in clinical trial and other development costs as we incurred less costs under the Phase 3 ENHANCE program which completed study conduct and analysis in 2023 whereas in 2022 significant costs were incurred associated with the then ongoing study conduct. The 2023 clinical trial and other development costs also include the impact of $2.2 million of credits received related to the final financial reconciliation of a Phase 3 ENHANCE program supplier. Selling, General and Administrative Costs Selling, general and administrative costs were $51.3 million for the year ended December 31, 2023 compared to $27.8 million for the year ended December 31, 2022, an increase of $24.5 million. This increase was driven primarily by a $17.1 million increase in people related costs, inclusive of share-based compensation, an increase of $9.7 million related to the build out of information technology and commercial infrastructure in preparation for a potential commercial launch and other corporate costs. These increases were partially offset by a non-recurring $2.0 million charge related to the modification of the assignment and license agreement with Ligand UK Development Limited, which was incurred in the year ended December 31, 2022. Finance Income and Expense Finance income was $14.6 million for the year ended December 31, 2023 and $2.8 million for the year ended December 31, 2022, an increase of $11.8 million. The increase was attributable to an increase in interest income from a higher average cash balance and higher interest rates and $1.9 million relating to foreign exchange gain. Finance expense was $18.9 million for the year ended December 31, 2023, compared to $9.5 million for the year ended December 31, 2022. In the year ended December 31, 2023, there was a $16.8 million expense relating to the unwind of the discount factor on the assumed contingent liability, $2.1 million relating to interest expense on our debt instruments. In the year ended December 31, 2022, there was a $4.3 million expense relating to the unwind of the discount factor on the assumed contingent liability, $3.8 million relating to foreign exchange loss and $0.8 million for the loss on extinguishment of debt. Cash and cash equivalents As at December 31, 2023, the Group held $271.8 million in cash and cash equivalents (2022: $227.8 million). Taxation Taxation for the year ended December 31, 2023 amounted to a credit of $0.6 million compared to a credit of $9.4 million for the year ended December 31, 2022, a decrease of $8.8 million. This decrease is primarily attributable to our decreased qualifying expenditure on research and development, on which a credit is obtained at a rate of 10% of 186% on expenditure incurred after April 01, 2023, and at a rate of 14.5% of 230% on expenditure incurred on or before March 31, 2023. Treasury shares The Group holds shares in an employee benefit trust, to satisfy share based compensation awards and these share are accounted for as treasury shares. As at December 31, 2023, 24,123,536 shares were held in treasury, at a nominal value of $1.5 million (2022: 25,037,192 shares, nominal value $1.5 million). Going concern We have incurred recurring losses and negative cash flows from operations since inception and have accumulated loss of $416.1 million as of December 31, 2023. We expect that our cash and cash equivalents, together with additional funding expected to become available under the 2023 Term Loan, will be sufficient to fund our operating expenses and capital expenditure requirements, including the planned commercial launch of nebulized ensifentrine for COPD maintenance treatment in the U.S, for at least the next 12 months from the date of approval of these financial statements. Future advances under the 2023 Term Loan are contingent upon achievement of certain regulatory and commercial milestones as well as other specified conditions. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 31 |
| Key Performance Indicators (“KPIs”) The Company is a development stage business and does not yet generate revenues or other operating cash inflows. The Company therefore uses a mix of Financial and Non-financial KPIs to monitor its activities. Financial KPIs can typically be compared over a period of years; Non-financial KPIs may change from year to year depending on the development stage of the Company’s programs. 1. Research and development spend during the year Strategic objective: Investment in R&D to generate future revenue for the Group. Key Performance Indicator: R&D expenditure of $17.4 million (2022: $50.3 million). Definition: Costs including labor, materials and other expenditure incurred by the Group on research and development. $’m Year ended December 31, 2019 2020 2021 2022 2023 Research and development 42.4 44.6 79.3 50.3 17.4 In the year ended December 31, 2023 R&D expenditure has reduced as expected due to the Company's ENHANCE phase 3 clinical trials coming to a end. 2. Cash and short-term investments held at year end Strategic objective: Availability of financial resources to progress the development of the Group’s research and development activities. Key Performance Indicator: Year-end cash of $271.8 million (2022: $227.8 million). Definition: Cash and cash equivalents. $’m As at December 31, 2019 2020 2021 2022 2023 Cash and equivalents 40.8 188.0 148.4 227.8 271.8 3. FDA approval and commercial launch of ensifentrine in the U.S. Strategic objective: Obtain FDA approval of the ensifentrine NDA and commercial launch of ensifentrine in the U.S. in the second half of 2024. Key Performance Indicator: FDA approval and first commercial sale of ensifentrine. Definition: Revenues In the year ended December 31, 2023, the company's NDA was accepted for review by the FDA. Gender of Directors and employees We recruit individuals who have the skills, experience and integrity needed to perform the roles to make Verona Pharma a successful company. We recruit without regard to sex or ethnic origin, appointing and thereafter promoting staff based upon merit. The profile of the Group’s employees at December 31, 2023, was as follows: Male Female Total December 31, 2023 December 31, 2023 December 31, 2023 Number of persons who were Directors of the Company 9 2 11 Number of persons who were executive officers of the Company 1 2 3 Number of persons who were other employees of the Company 36 39 75 Total employees at December 31, 2023 46 43 89 VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 32 |
| Environmental matters We currently outsource our research, development, testing and manufacturing activities. These activities are subject to various environmental, health and safety laws and regulations, which govern, among other things, the controlled use, handling, release and disposal of and the maintenance of a registry for, hazardous materials and biological materials. If we or our partners fail to comply with such laws and regulations, we could be subject to fines or other sanctions. As with other companies engaged in activities similar to ours, we face a risk of environmental liability inherent in our current and historical activities, including liability relating to releases of or exposure to hazardous or biological materials. Environmental, health and safety laws and regulations are becoming more stringent. We may be required to incur substantial expenses in connection with future environmental compliance or remediation activities, in which case, our production and development efforts may be interrupted or delayed. Greenhouse Gas Emissions We have used the Greenhouse Gas (“GHG”) Protocol Corporate Accounting and Reporting Standard (revised edition) data gathered to fulfil our requirements under the CRC Energy Efficiency scheme, and emission. Our greenhouse gas emission estimates for 2023 and 2022 have been prepared in accordance with the U.K. government's Department for Environment, Food and Rural Affairs (DEFRA) guidance document Environmental Reporting Guidelines: Including Mandatory GHG emissions reporting guidance from June 2013. Tonnes carbon dioxide equivalent (tCO2-e) 2023 2022 Estimated greenhouse gas emissions from our own activities, including the combustion of fuel and the operation of our facilities — — Estimated greenhouse gas emissions from purchased electricity, heat, steam or cooling for own use — — Total estimated greenhouse gas emissions — — Intensity ratio: N/A N/A We are a company with a small number of employees. We have serviced offices and we currently outsource our research, development, testing and manufacturing activities. As a result, we do not emit greenhouse gases from our own activities, nor do we purchase electricity, heat or steam for our own use (Scope 1 and Scope 2 disclosures). However, we are aware that our activities do have an impact on GHG emissions through the work of our partners and our activities such as business travel (Scope 3 disclosures). We have discussed with our partners the impact of our operations on emissions but they have not been able to provide the information for us to provide a meaningful analysis. We have activities in the U.S. and Europe and we need to fly our employees, directors and consultants to effectively manage our business and operations and these scope 3 emissions from business travel have not been quantified in the table above given the limited nature of these activities. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 33 |
| Approach to Risk Drug development and commercialisation is inherently risky. There is no certainty that ensifentrine will progress successfully through development, obtain regulatory approval and become a marketable product. All of the Group’s activities involve an ongoing assessment of risks and the Group seeks to mitigate such risks where possible. The Board has undertaken an assessment of the principal risks and uncertainties facing the Group, including those that would threaten its business model, future performance, solvency and liquidity. In addition, the Board has considered the longer-term viability of the Group including factors such as the prospects of the Group and its ability to continue in operation for the foreseeable future. On a regular cadence the Board reviews the level of risks that the Group is taking in pursuit of its strategy. Based upon this review the Board is satisfied that the level of retained risk is appropriate and commensurate with the financial rewards that should result from achievement of its strategy. The principal risks and uncertainties have been identified as follows: • We have a limited operating history and have never generated any product revenue; • We may need additional funding to complete development of and commercialize ensifentrine and any future product candidates, if approved, or develop and commercialize other formulations or target indications of ensifentrine, if approved; • The advances under the $400 million 2023 Term Loan are contingent upon achievement of certain clinical and regulatory milestones and other specified conditions. If we fail to meet those conditions, we will need to find alternative sources of funding; • Changes in our tax rates, unavailability of certain tax credits or reliefs or exposure to additional tax liabilities or assessments could affect our profitability, and audits by tax authorities could result in additional tax payments for prior periods; • We depend solely on the success of ensifentrine, our only product candidate under development; • We may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our product candidates; • Ensifentrine may have serious adverse, undesirable or unacceptable side effects which may delay or prevent marketing approval; • If we are unable to enroll patients in our clinical trials for other indications, or enrollment is slower than anticipated, our research and development efforts could be adversely affected; • We may become exposed to costly and damaging liability claims, either when testing ensifentrine in the clinic or at the commercial stage, and our product liability insurance may not cover all damages from such claims; • Regulatory approval processes are lengthy, time consuming and inherently unpredictable, and if we are ultimately unable to obtain regulatory approval for ensifentrine, our business will be substantially harmed; • Enacted and future legislation may increase the difficulty and cost for us to obtain marketing approval of and commercialize ensifentrine and may affect the prices we may set; • Our business operations and current and future relationships with investigators, healthcare professionals, consultants, third-party payors and customers will be subject to applicable healthcare regulatory laws, which could expose us to penalties; • We operate in a highly competitive and rapidly changing industry, which may result in others discovering, developing or commercializing competing products before or more successfully than we do; • We rely, and expect to continue to rely, on third parties, including independent clinical investigators and clinical research organizations, to conduct our pre-clinical studies and clinical trials; • The collaboration and license agreement with Nuance Pharma is important to our business. If Nuance Pharma is unable to develop and commercialize products containing ensifentrine in Greater China, if we or Nuance Pharma fail to adequately perform under the Nuance Agreement, or if we or Nuance Pharma terminate the Nuance Agreement, our business would be adversely affected. • If we fail to enter into new strategic relationships for ensifentrine, our business, research and development and commercialization prospects could be adversely affected; • We rely, and expect to continue to rely, on third party manufacturers and suppliers for production of the active pharmaceutical ingredient ensifentrine and formulated drug products derived therefrom. Our dependence on these third parties may impair the advancement of our research and development programs and the development of ensifentrine; • We rely, and expect to continue to rely, on third parties for the sales, marketing, reimbursement and distribution of our drug products, and a failure by these third parties to adequately perform would adversely affect our business; • Our and our manufacturers’, suppliers’ and other critical third parties’ cybersecurity risk management program and processes may not be effective in protecting our systems, networks and Confidential Information. VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 34 |
| • We rely on patents and other intellectual property rights to protect ensifentrine, the enforcement, defense and maintenance of which may be challenging and costly; • We may not identify relevant third-party patents or may incorrectly interpret the relevance, scope or expiration of a third-party patent which might adversely affect our ability to develop, manufacture and market ensifentrine; • We may be involved in lawsuits to protect or enforce patents covering ensifentrine, which could be expensive, time consuming and unsuccessful, and issued patents could be found invalid or unenforceable if challenged in court; • Our future growth and ability to compete depends on our ability to retain our key personnel and recruit additional qualified personnel; • We expect to expand our development, regulatory and sales and marketing capabilities, and as a result, we may encounter difficulties in managing our growth, which could disrupt our operations; • The price of our American Depositary Shares may be volatile and may fluctuate due to factors beyond our control; and • We will continue to incur increased costs as a result of operating as a public company in the United States, and our senior management are required to devote substantial time to new compliance initiatives and corporate governance practices. On behalf of the Board Dr. David Zaccardelli Chief Executive Officer March 18, 2024 VERONA PHARMA PLC STRATEGIC REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 35 |
| Letter from the Chair of the Remuneration Committee Dear Shareholders, As Chair of the Remuneration Committee (the "Committee"), I am pleased to present, on behalf of the board of directors (the "Board") of Verona Pharma, the Directors’ Remuneration Report for the year ended December 31, 2023 (the "Remuneration Report"). Shareholders will be invited to approve the Remuneration Report, which will be subject to a non-binding advisory vote, at the Annual General Meeting of shareholders (“AGM”) to be held on April 26, 2024 ("2024 AGM"). Shareholders will also be invited to approve the Directors’ Remuneration Policy, which will be subject to a binding Shareholder vote, at the 2024 AGM (the “Remuneration Policy”). If approved, the Remuneration Policy would be expected to be effective from April 26, 2024 and remain in force until the AGM in 2027 with no requirement to vote again on the Policy in the intervening years provided that no substantive changes are proposed. The notice and accompanying materials for the 2024 AGM will be sent out in due course. The Remuneration Committee The Committee is responsible for reviewing and establishing our executive and non-executive remuneration policy and philosophy, including making recommendations to the Board for its approval with respect to the remuneration of our President and CEO, who is our sole Executive Director, and our Non-Executive Directors. The Committee is also responsible for determining and approving the remuneration of senior executive officers. The composition and terms of reference of the Committee can be found on our website at www.veronapharma.com. Remuneration philosophy The aim of the Remuneration Policy is to enable the Company to offer remuneration packages that are designed to promote the long-term success of the Company by: ▪ being sufficiently competitive to enable the Company to attract, incentivize and retain the Executive Directors and management it needs to operate its business; ▪ supporting and rewarding the delivery of the Company's strategy and corporate objectives and ultimately creating value for shareholders; ▪ aligning Executive Directors and management with the long-term interests of shareholders and helping to retain them by delivering a significant element of remuneration in shares; ▪ effectively managing the Company’s cash resources; and ▪ being flexible enough to cope with the Company's changing needs as it grows and the strategy evolves. It is the belief of the Committee that these objectives are best achieved through a greater emphasis on variable rather than fixed remuneration, comprised of a mix of base salary and benefits, along with the flexibility to appropriately reward and incentivize with variable pay and longer term incentives, as described within the Remuneration Policy. Whilst the Company is headquartered in the U.K, given that a number of the Company's senior executives are based in the U.S., where the market for experienced directors and biopharmaceutical executive talent is very competitive, and given that the Company is listed on a U.S. stock exchange and that its shareholder base is primarily U.S. based, the Committee references U.S. benchmarks and practices in designing its remuneration programs and policies. Notwithstanding, the Committee exercises its discretion in determining the various elements of cash and equity compensation and is mindful of the general U.K. compensation framework, including investor bodies guidance, and has considered these when determining the remuneration programs and policies where it believes they best serve the long-term interests of shareholders. Currently the Company has only one Executive Director, but the Remuneration Policy will apply equally to any additional Executive Directors who may be appointed in the future. The Committee annually reviews the operation of the remuneration programs and policies to ensure they are operating within an acceptable risk profile and that they do not inadvertently encourage any economic, social or governance issues. Key activities and decisions in the year ended December 31, 2023 In June 2023, the Company filed with the FDA an NDA seeking approval of ensifentrine for the maintenance treatment of COPD, and in August 2023, the FDA accepted the NDA for review and assigned a PDUFA target action date of June 26, 2024. During 2023, the Company presented numerous additional analyses of data from its Phase 3 ENHANCE clinical trials at international scientific conferences and the data were published in peer reviewed publications. In addition, the Company continued to build its commercial capabilities and launch readiness in preparation for the potential approval of ensifentrine. An important component of this was expanding the Company’s headcount to approximately 80 employees adding key leadership positions across medical affairs, compliance, manufacturing, finance and IT and building commercial teams in marketing, market access and commercial operations. These appointments included Senior Vice President, Medical Affairs, Vice President, Compliance, and Vice President, Pharmacovigilance. In December 2023, the Company significantly strengthened its financial position through a $400 million debt financing facility. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 36 |
| During 2023, the Committee engaged AoN Consulting, Inc. to review the peer companies used for the Company’s compensation benchmarking and conducted an independent benchmarking review of the compensation of the Non-Executive Directors and the senior executive officers, including the Executive Director. This review resulted in a change in the peer companies to better align with the Company in terms of market capitalization, stage of development and other factors, and also resulted in an increase in the fees paid to the Non-Executive Directors, effective as of January 1, 2024. The benchmarking also showed that the retentive value of equity incentives held by employees, including the Executive Director, is less than the peer group and that the incentives held by the Executive Director do not include performance conditions, compared to the peer group which has a mix of time-based and performance-based equity incentives. Based on the review, recognizing the importance of employee retention and incentivization as the Company prepares for the commercial launch of ensifentrine in the U.S., if approved, in October 2023 the Committee approved the grant of equity incentives to all employees, including the Executive Director, comprising a combination of 25% Restricted Stock Units (“RSUs”) and 75% Performance Restricted Stock Units (“PRSUs”) under the Company’s 2017 Long Term Incentive Plan. The Committee also awarded a 4% base salary increase for the Executive Director, with effect from January 1, 2024. During 2023, the Committee's other activities included monitoring and assessing performance against the annual bonus objectives for the senior executives, including the Executive Director. In December 2023, the Committee determined the level of bonus awards payable in respect of the 2023 performance period. The awards recognized that 105% of the Company’s corporate objectives for 2023 were achieved. The Board accepted the Committee's recommendation and such amounts have been included in this 2023 annual report and accounts. In December 2023, the Committee approved the annual bonus objectives to be achieved by the senior executives, including the Executive Director, for the year ending December 31, 2024. These objectives, which were approved by the Board, are considered to be commercially sensitive and will not be disclosed in detail, but are designed to support achievement of the Company's strategic objectives to develop and commercialize innovative therapies for the treatment of chronic respiratory diseases with significant unmet medical needs. The Company has made significant progress during 2023 with the FDA’s acceptance for review of the ensifentrine NDA, preparing for commercial launch of ensifentrine, if approved, and strengthening of the Company’s financial position. The compensation approved by the Committee for 2024, including the bonus objectives for the Executive Director and other senior executive officers, is designed to support achievement of the Company’s strategic objectives and core focus during 2024 to obtain approval of the ensifentrine NDA and progress the commercial launch of ensifentrine in the U.S. in the second half of 2024. We hope that you remain supportive of our remuneration approach and will vote in favor of the Directors' Remuneration Report. Yours faithfully, Dr Ken Cunningham Chair of the Remuneration Committee March 18, 2024 VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 37 |
| Annual Report on Remuneration Single total figure of remuneration of each Director (audited) The Directors received the following remuneration for the years ended December 31, 2023 and December 31, 2022: Financial Year Base Salary / Cash Fees Bonus Employer ’s Pension Share-based payment (i) Benefits Other Total fixed Total variable Total $ $ $ $ $ $ $ $ $ Executive David Zaccardelli 1 2023 818,603 440,705 13,200 5,324,000 13,276 — 865,913 5,743,871 6,609,784 2022 524,842 497,297 12,200 4,133,500 27,978 — 815,020 4,380,797 5,195,817 Non-Executive David Ebsworth 2023 148,962 — — 215,638 — — 148,962 215,638 364,600 2022 140,846 — — 64,375 — — 140,846 64,375 205,221 Ken Cunningham 2023 50,927 — — 215,638 — — 50,927 215,638 266,565 2022 48,152 — — 64,375 — — 48,152 64,375 112,527 Anders Ullman 2023 38,195 — — 215,638 2,928 — 41,123 215,638 256,761 2022 36,114 — — 64,375 — — 36,114 64,375 100,489 Rishi Gupta 2023 38,195 — — 215,638 — — 38,195 215,638 253,833 2022 37,681 — — 64,375 — — 37,681 64,375 102,056 Mahendra Shah 2023 43,288 — — 215,638 — — 43,288 215,638 258,926 2022 40,538 — — 64,375 — — 40,538 64,375 104,913 Andrew Sinclair 2 2023 — — — — — — — — — 2022 13,612 — — — — — 13,612 — 13,612 Vikas Sinha 2023 57,293 — — 215,638 1,210 — 58,503 215,638 274,141 2022 52,996 — — 64,375 — — 52,996 64,375 117,371 Martin Edwards 2023 38,195 — — 215,638 — — 38,195 215,638 253,833 2022 36,114 — — 64,375 — — 36,114 64,375 100,489 Lisa Deschamps 2023 42,015 — — 215,638 — — 42,015 215,638 257,652 2022 39,726 — — 64,375 — — 39,726 64,375 104,101 James Brady 3 2023 44,561 — — 288,120 — — 44,561 288,120 332,681 2022 33,869 — — 82,620 — — 33,869 82,620 116,489 Christina Ackermann 4 2023 14,106 — — — 1,375 — 15,481 — 15,481 2022 — — — — — — — — — 1 Dr. Zaccardelli was entitled to an annual base salary of $795,675 in 2022, made up of $545,675 in cash and $250,000 in restricted stock units. $524,842 was the actual cash figure paid to Dr. Zaccardelli in 2022 due to the change in his cash/share split in February 2022. In 2023 this increased to $839,436 made up of $818,603 in cash and $20,833 in restricted stock units.. 2 Resigned April 27, 2022 3 Appointed March 14, 2022 4 Appointed September 1, 2023 i) Share based payments represent the intrinsic value of share options that vested during the years ended December 31, 2022 and December 31, 2023 and the intrinsic value of RSUs and PRSUs granted in the years ended December 31, 2022 and December 31, 2023. The intrinsic value of the share options is the difference between the share price on the date of vesting and the exercise price of the option. In the case of RSUs and PRSUs it is the share price on the day of grant. No amount of this award was attributable to share price appreciation. Dr. Zaccardelli's compensation package is denominated in U.S. dollars; all other directors' compensation is denominated in U.K. pounds, except for share based payments, which are calculated on the price of ADSs. For the purposes of this table, all amounts are translated into U.S. dollars using exchange rates on December 31, 2023 (1.273180) and December 31, 2022 (1.203810) for each year respectively. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 38 |
| Annual performance bonus The Company operates a discretionary bonus scheme for all employees including the CEO. Bonus awards are granted as a percentage of base salary and based on objectives signed off by the Remuneration Committee each year. For 2023, the CEO’s maximum bonus opportunity was 50% of base salary. The Remuneration Committee assessed performance against the objectives determining that 105% of the objectives were achieved. This resulted in a 2023 bonus award equating to 52.5% of base salary for the CEO. The performance objectives achieved by the Executive Director included the following: • FDA Pre-NDA meeting allowing NDA submission in the second quarter of 2023; • ensifentrine NDA accepted for review in the third quarter of 2023; • nebulized ensifentrine/glycopyrrolate combination product formulation feasibility in the third quarter of 2023; • key pre-commercial activities to support commercial launch of ensifentrine, if approved, including: - launch of a disease awareness campaign; - initial development of a branded campaign; - sales force strategy, including size and territories; - develop distribution model and initiate contracting discussions; and • operated within the approved budget. Long term incentive awards The Executive Director was granted 800,000 restricted share units ("RSUs") with respect to ordinary shares and 2,400,000 performance restricted share units ("PRSUs") with respect to ordinary shares during the 2023 performance period. Payments to past Directors (audited) There were no payments to past Directors during the financial year ended December 31, 2023 or the financial year ended December 31, 2022. Payments for loss of office (audited) There were no payments to directors for loss of office in the financial year ended December 31, 2023 or the financial year ended December 31, 2022. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 39 |
| Statement of Directors’ Shareholding and Share Interests (audited) The table below details the total number of ordinary shares owned (including their beneficial interests), the total number of ordinary share options held, the number of ordinary share options vested but not yet exercised and the total number of restricted share units ("RSUs") and performance restricted share units ("PRSUs") with respect to ordinary shares held as at December 31, 2023: December 31, 2023 Shares Options - not vested Options vested, not exercised RSUs not vested PRSUs not vested Total (shares and options) Executives David Zaccardelli 11,766,864 — — 4,655,928 2,400,000 18,822,792 Non Executives Vikas Sinha 74,440 48,000 432,384 — — 554,824 Anders Ullman 334,856 48,000 312,000 — — 694,856 David Ebsworth 844,643 48,000 312,000 — — 1,204,643 Ken Cunningham 66,584 48,000 312,000 — — 426,584 Mahendra Shah 73,080 48,000 312,000 — — 433,080 Martin Edwards 144,800 48,000 312,000 — — 504,800 Rishi Gupta — 48,000 497,600 — — 545,600 Lisa Deschamps 70,320 48,000 312,000 — — 430,320 James Brady — 48,000 336,000 — — 384,000 Christina Ackermann — 144,000 — — — 144,000 13,375,587 576,000 3,137,984 4,655,928 2,400,000 24,145,499 VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 40 |
| The interests of the Directors in the Company’s ordinary share options and RSUs and PRSUs with respect to ordinary shares as at December 31, 2023 were as follows: Director Date of Grant Exercise price per share ($) Type January 1, 2023 Granted during the year Vested during the year December 31, 2023 Date from which exercisable Expiry date Vikas Sinha April 26, 2017 1.70 Options 120,384 — — 120,384 i) April 26, 2027 David Zaccardelli May 7, 2020 — RSU 1,316,576 — (1,053,264) 263,312 ii) N/A David Zaccardelli August 20, 2020 — RSU 4,463,016 — (3,570,400) 892,616 iii) N/A Rishi Gupta September 24, 2020 0.79 Options 185,600 — — 185,600 iv) September 24, 2030 Ken Cunningham August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 Lisa Deschamps August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 David Ebsworth August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 Martin Edwards August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 Rishi Gupta August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 Mahendra Shah August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 Vikas Sinha August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 Anders Ullman August 9, 2021 0.78 Options 64,000 — — 64,000 v) August 8, 2031 David Zaccardelli February 1, 2022 — RSU 83,608 — (83,608) — vi) N/A James Brady March 14, 2022 0.60 Options 288,000 — — 288,000 vii) March 13, 2032 Ken Cunningham April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 Lisa Deschamps April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 Martin Edwards April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 David Ebsworth April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 Rishi Gupta April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 Mahendra Shah April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 Vikas Sinha April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 Anders Ullman April 28, 2022 0.50 Options 200,000 — — 200,000 viii) April 27, 2032 David Zaccardelli September 26, 2022 — RSU 3,600,000 — (900,000) 2,700,000 ix) N/A Rishi Gupta April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 Sven Ullman April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 David Ebsworth April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 Lisa Deschamps April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 Vikas Sinha April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 Mahendra Shah April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 Martin Edwards April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 Kenneth Cunningham April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 James Brady April 28, 2023 2.67 Options — 96,000 — 96,000 x) April 27, 2033 Christina Ackermann September 1, 2023 2.43 Options — 144,000 — 144,000 xi) August 31, 2033 David Zaccardelli October 20, 2023 — RSU — 800,000 — 800,000 xii) N/A David Zaccardelli October 20, 2023 — PRSU — 2,400,000 — 2,400,000 xiii) N/A All options are subject to service conditions. i) 50% of these options vested in three annual tranches and 50% in four. The first vesting date was April 26, 2018. ii) 25% of these RSUs vested on February 1, 2021, with the remaining vesting in twelve equal quarterly tranches thereafter. The face value of this award was $2,211,174. iii) 25% of these RSUs vested on February 1, 2021, with the remaining vesting in twelve equal quarterly tranches thereafter. The face value of this award was $14,909,288. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 41 |
| iv) 50% of these RSUs or options vested on November 1, 2020, with the remainder in two equal quarterly installments. The face value of each award was $121,075. v) These options vest in four equal installments. The first vesting date was August 9, 2021, with the remaining quarterly from November 1, 2021. The face value of each award was $49,600. vi) These RSUs vest in four equal quarterly tranches. The first vesting date was May, 1 2022. The face value of this award was $250,000. vii) These options vest in four equal quarterly installments. The first vesting date was August 1, 2022. The face value of the award was $173,520. viii)These options vest in four equal quarterly installments. The first vesting date was July 28, 2022. The face value of each award was $100,750. ix) 25% of these RSUs vest on November 1, 2023, with the remaining vesting in twelve equal quarterly tranches thereafter. The face value of this award was $3,883,500. x) These options vest in four equal quarterly installments. The first vesting date was July 28, 2023. The face value of each award was $256,320. xi) 33% of these options will vest on September 1, 2024, with the remaining vesting in eight equal quarterly tranches thereafter. The face value of the award was $349,740. xii) 25% of these RSUs vest on November 1, 2024, with the remaining vesting in twelve equal quarterly tranches thereafter. The face value of this award was $1,331,000. xiii) 33% of these PRSUs vest upon achievement of a performance condition related to the commercialization of ensifentrine in the U.S., with the remaining vesting in eight equal quarterly tranches thereafter, subject generally to continued employment. The face value of this award was $3,993,000. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 42 |
| Directors’ interests (audited) The beneficial and non-beneficial interests of the Directors in the Company’s ordinary shares as at December 31, 2023, were as follows: Name Held at December 31, 2023 Held at December 31, 2022 David Zaccardelli 11,766,864 8,312,600 David Ebsworth 844,643 684,643 Vikas Sinha 74,440 74,440 Anders Ullman 334,856 334,856 Ken Cunningham 66,584 66,584 Mahendra Shah 73,080 73,080 Martin Edwards 144,800 111,064 Rishi Gupta — — Lisa Deschamps 70,320 70,320 James Brady — — Christina Ackermann — — Between December 31, 2023 and March 8, 2024, the only change in the interests of the Directors was the vesting of Dr. Zaccardelli's 900,728 ordinary shares of RSUs. Total shareholder return The graph below shows the Company's performance, measured by total shareholder return, compared with the value if the same investment had been made in the FTSE AIM All Share and NASDAQ / Biotechnology (NBI) indices on the same date. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 43 |
| CHIEF EXECUTIVE OFFICER TOTAL REMUNERATION HISTORY 2017 was the first year that Verona Pharma prepared a Directors' Remuneration Report, and took the exemption not to disclose 5 years of history of remuneration. The Company has chosen to disclose remuneration history from 2017 onwards. 2023 2022 2021 2020 (1) 2019 2018 2017 Total CEO remuneration ($'000s) 6,610 5,196 1,140 18,390 901 1,073 1,452 Annual variable element award rates against maximum opportunity 105% 125% 85% 110 % 40 % 57% 66% Long-term incentive vesting rates against maximum opportunity 100% 100% 100% 100% 100% 100% 100% 1) this includes one month of the remuneration of Dr. Karlsson and eleven months of Dr. Zaccardelli. All pound sterling amounts have been translated into U.S. dollars using exchange rates on December 31, 2020 (1.366312), December 31, 2019 (1.326752), December 31, 2018 (1.276021) and December 31, 2017 (1.350291) for each year respectively. PERCENTAGE CHANGE OF DIRECTORS' REMUNERATION The table below shows the percentage change in remuneration of the directors and the Group’s employees as a whole for the period January 01 to December 31 for the following years: Percentage increase/(decrease) for: 2023 compared to 2022 2022 compared to 2021 2021 compared to 2020 2020 compared to 2019 Director Average Employee Director Average Employee Director Average Employee Director Average Employee Base salary David Zaccardelli 5% 6% 3% 10% 3% 11% 71% 9% Short-term incentives David Zaccardelli (11)% (3)% 51% 53% (20)% (2)% 78% 28% Taxable benefits David Zaccardelli (53)% 6% 2% 16% (33)% 1% 10% 4% Base salary David Ebsworth —% 6% —% 10% 4% 11% 4% 9% Base salary Ken Cunningham —% 6% —% 10% —% 11% —% 9% Base salary Anders Ullman —% 6% (9)% 10% 5% 11% 4% 9% Base salary Rishi Gupta (4)% 6% (8)% 10% 7% 11% 6% 9% Base salary Mahendra Shah 1% 6% 2% 10% 5% 11% 4% 9% Base salary Andrew Sinclair 1 (100)% 6% (68)% 10% 9% 11% 7% 9% Base salary Vikas Sinha 2% 6% 5% 10% —% 11% —% 9% Base salary Martin Edwards —% 6% —% 10% —% 11% 33% 9% Base salary Lisa Deschamps —% 6% 20% 10% N/A 11% N/A 9% Base salary James Brady 2 24% 6% N/A 10% N/A 11% N/A 9% Base salary Christina Ackermann 3 N/A 6% N/A 10% N/A 11% N/A 9% 1 Resigned in April 27, 2022 2 Appointed March 14, 2022 3 Appointed September 01, 2023 VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 44 |
| Relative importance of spend on pay The Committee considers the Company’s research and development and selling, general and administrative expenditure relative to salary expenditure for all employees to be the most appropriate metric for assessing overall spend on pay due to the nature and stage of the Company’s business as it conducts research and development activities and prepares for the potential commercial launch of ensifentrine in the U.S. Dividend distribution and share buy-back comparators have not been included as the Company has no history of such transactions. The graph below illustrates the gross pay to all employees compared to research and development and selling, general and administrative expenditure and illustrates the year-on-year change. The Committee notes that research and development expenditure decreased in 2023 from 2022 as we incurred less costs under the Phase 3 ENHANCE program which completed study conduct and analysis in 2023 whereas in 2022 significant costs were incurred associated with the then ongoing study conduct. This was partially offset by an increase in 2023 from 2022 in selling, general and administrative expenditure in people related costs, inclusive of share-based compensation, an increase in costs related to the build out of information technology and commercial infrastructure in preparation for the potential commercial launch of ensifentrine and other corporate costs as well as an increased charge related to the unwinding of the assumed Ligand contingent liability. Salary expenditure increased in 2023 due to new commercial and medical affairs hires as the Company continues to prepare for the potential commercial launch of ensifentrine in the U.S. External advice During the 2023 financial year, the Company engaged AoN Consulting, Inc. (the "Remuneration Advisors") to support the Committee and management with advice on remuneration matters and the Committee is satisfied that they provide independent and objective advice. During 2023, the Company paid fees of $127,200 to the Remuneration Advisors. No other fees were paid to them in the year. Proposed Application of the Remuneration Policy for the Year Ended December 31, 2023 i) Fixed elements of remuneration With effect from January 1, 2024, the base salary of Dr. David Zaccardelli in his role as President, CEO, and Executive Director of the Company is $873,015 per annum, all of which is paid in cash. In accordance with the Remuneration Policy, the Remuneration Committee has considered Dr. Zaccardelli's base salary in the context of a number of factors, including the market benchmarking exercise carried out by the Remuneration Advisors, the skills and experience of Dr. Zaccardelli, and the location, responsibilities and scale and complexity of the role. ii) Variable elements of remuneration VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 45 |
| Short-term incentives The target bonus for Dr. Zaccardelli for the 2024 performance period will be 50% of base salary. The performance objectives for Dr. Zaccardelli against which the Committee will determine the annual bonus were approved by the Board in December 2023. The detail behind the performance objectives is currently considered to be commercially sensitive as it relates to the Company's strategy for the advancement of the ensifentrine clinical development program and its financial and commercial goals. To the extent that the objectives do not comprise commercially sensitive information, the Company expects to disclose both the objectives and performance against those objectives in next year’s Directors’ Remuneration Report. Long-term incentive awards The Company anticipates awarding a long term incentive grant to the Executive Director in 2024 under the Company's 2017 Incentive Plan, subject to Board approval. iii) Chairperson and Non-Executive Director fees (audited) Chairperson fees The Chairperson is paid a basic fee and a fee for chairing and membership of Board Committees. The basic fee and the fee for chairing and membership of Board Committees were reviewed in 2023 following a benchmarking exercise undertaken by the Company’s external Remuneration Advisors. This review resulted in an increase in the fees paid to the Chairperson. The Chairperson is also awarded equity incentives under the 2017 Incentive Plan. Non-Executive Director cash fees Non-Executive Directors are paid a basic fee and a fee for chairing or membership of Board committees. The basic fee and the fee for chairing and membership of Board Committees were reviewed in 2023 following a benchmarking exercise undertaken by the Company’s Remuneration Advisors. This review resulted in an increase in the fees paid to the Non-Executive Directors. Non-Executive Directors are also awarded equity incentives under the 2017 Incentive Plan. The table below shows the total of the annual basic fee and fees for chairing and membership of Board Committees currently payable to our Chairperson and Non-Executive Directors. Name Annual Fees (£) David Ebsworth 141,000 Ken Cunningham 52,000 Anders Ullman 40,000 Mahendra Shah 46,000 Vikas Sinha 62,000 Martin Edwards 40,000 Lisa Deschamps 44,000 James Brady 50,000 Christina Ackermann 50,000 Michael Austwick 40,000 The Remuneration Policy provides that Executive Directors may have contracts with an indefinite term provided the contracts have a notice period which does not exceed twelve months. Ms. Christina Ackermann, Mr. Michael Austwick, Mr. Jim Brady, Dr. Ken Cunningham, Ms. Lisa Deschamps, Dr. Martin Edwards, Mr. Vikas Sinha and Dr. Anders Ullman have letters of appointment which are subject to a three-month notice period. Dr. Mahendra Shah and Mr. Rishi Gupta were designated as Non-Executive Directors of our Board under relationship agreements we entered into in June 2016 with entities affiliated with each of Vivo Capital and OrbiMed, respectively. The appointment rights under these relationship agreements automatically terminated on the Company delisting from AIM in October 2020. Notwithstanding, the Board resolved that Dr. Shah and Mr. Gupta continue to be appointed to the Board pursuant to letters of appointment, which are also subject to a three-month notice period. Mr. Gupta resigned from the Board on January 31, 2024. The Non-Executive Directors' remuneration is reviewed by the Board annually. In accordance with the Company's Articles of Association, one third of Directors are subject to retirement by rotation at each AGM. Mr. Michael Austwick, Ms. Christina Ackermann, Ms. Lisa Deschamps, Dr. Martin Edwards and Dr. Anders Ullman will be retiring by rotation at the 2024 AGM and, being eligible, will seek re-election. Pursuant to our Articles of Association, if no other director is elected to fill their respective positions and the directors are willing, they shall be re-elected by default. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 46 |
| Details of Directors’ service contracts or letters of appointment for the year ended December 31, 2023 are as follows: Director Date of Contract Executive David Zaccardelli February 1, 2020 Non-Executive David Ebsworth December 1, 2014 Ken Cunningham September 10, 2015 Anders Ullman September 10, 2015 Rishi Gupta July 29, 2016 Mahendra Shah July 29, 2016 Andrew Sinclair July 29, 2016 Vikas Sinha September 12, 2016 Martin Edwards April 1, 2019 Lisa Deschamps March 1, 2021 James Brady March 14, 2022 Christina Ackermann September 1, 2023 Mr. Michael Austwick was appointed as a Non-Executive Director after December 31, 2023 pursuant to a contract dated February 1, 2024. Directors' service contracts are available in the Company’s SEC filings at https://www.veronapharma.com/investors/news-sec-filings. The information in this part of the Directors’ Remuneration Report is not subject to audit. Directors’ Remuneration Policy The current Remuneration Policy was approved by the Company’s shareholders at the 2021 Annual General Meeting and will remain in force for three years from that date until the Annual General Meeting in 2024, or until a new Remuneration Policy is approved by shareholders. The Remuneration Policy on pages 48 to 55 will be subject to a binding Shareholder vote at the 2024 Annual General Meeting, and if approved, would be expected to be effective from April 26, 2024 and remain in force until the Annual General Meeting in 2027 with no requirement to vote again on the Policy in the intervening years provided that no substantive changes are proposed. Statement of voting on the Remuneration Policy at the 2022 Annual General Meeting At the Annual General Meeting held on April 27, 2022, votes cast by proxy at the meeting in respect of the Directors’ Remuneration Report were as follows: In favor votes Against votes Total votes cast Votes withheld To approve the Remuneration Report 444,784,340 64,993 444,849,333 23,048 % of votes cast 99.99 % 0.01 % 100 % — Statement of voting on the Remuneration Report at the 2023 Annual General Meeting At the Annual General Meeting held on April 27, 2023, votes cast by proxy at the meeting in respect of the Directors’ Remuneration Report were as follows: In favor votes Against votes Total votes cast Votes withheld To approve the Remuneration Report 581,723,878 20,054,712 601,778,590 902,840 % of votes cast 96.67 % 3.33 % 100 % — VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 47 |
| Directors’ Remuneration Policy The Policy will be subject to a binding Shareholder vote at the 2024 AGM, and if approved, would be expected to be effective from April 26, 2024 and remain in force until the AGM in 2027 with no requirement to vote again on the Policy in the intervening years provided that no substantive changes are proposed. The Committee may make minor amendments to the Policy (for example for tax, regulatory, exchange control or administrative purposes) without obtaining shareholder approval. The Remuneration Committee of the Board of Directors of the Company (the “Committee”) followed a robust process when reviewing and considering amendments to the Policy, considering both the strategic objectives of the business and evolving market practices. Input was also sought from management, while ensuring that conflicts of interest were suitably mitigated. Remuneration philosophy The aim of the Policy is to enable the Group to offer remuneration packages that are designed to promote the long-term success of the Group by: ▪ being sufficiently competitive to enable the Group to attract, incentivize and retain the Executive Directors and management it needs to operate its business; ▪ supporting and rewarding the delivery of the Group's strategy and corporate objectives and ultimately creating value for shareholders; ▪ aligning Executive Directors and management with the long-term interests of shareholders and helping to retain them by delivering a significant element of remuneration in shares; ▪ effectively managing the Group’s cash resources; and ▪ being flexible enough to cope with the Group's changing needs as it grows and the strategy evolves. Currently the Group has only one Executive Director, but the Policy will apply equally to any additional Executive Directors who may be appointed in the future. The Committee annually reviews the operation of the remuneration packages to ensure they are operating within an acceptable risk profile and that they do not inadvertently encourage any economic, social or governance issues. Remuneration Policy Remuneration Policy for Executive Directors The total remuneration for the Executive Director is made up of the following elements: • Salary; • Benefits; • Annual bonus; • Long-term incentive awards; and • Pension. The Company adopted the 2017 Incentive Plan on completion of the Nasdaq IPO in April 2017, and since January 1, 2017 the Company has only granted equity incentives under the 2017 Incentive Plan. A copy of the employment agreement for the Executive Director and the letters of appointment for the non-Executive Directors are available in the Company’s SEC filings at https://www.veronapharma.com/investors/news-sec-filings. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 48 |
| Element of remuneration Purpose and link to strategy Operation Maximum and minimum potential value Performance metrics Change to 2021 Policy Base salary Provides market competitive fixed remuneration that reflects the responsibilities of the role undertaken, the experience of the individual and performance in the role over time. Reviewed annually taking into account individual responsibilities, experience, performance, inflation and market rates. The Committee will also consider the pay and employment conditions in the wider workforce when determining Executive Directors’ salaries. Salary increases are normally effective from 1 January each year. Salaries are periodically benchmarked against a relevant peer group of life sciences companies, many of which are listed on Nasdaq, with a similar stage of development, and similar market capitalization and financial profile. Salaries are typically aligned with the 50th percentile of peer group comparator data but the Committee may vary from this general rule where it considers that special circumstances apply or where recruitment or retention of a particular role is required. Salaries may be paid in a combination of cash and equity. The current base salary of the Executive Director is set out in the application of policy section of the Directors' Remuneration Report. There is no formal maximum level of base salary. Larger increases may be permitted to reflect a change in responsibilities or a significant increase in the scale or complexity of the role, or increases in line with the remuneration of the Group’s wider workforce. The overall performance of the individual and Group is a key determinant for salary increases. (i) Peer companies no longer limited to clinical development stage companies. (ii) Peer companies no longer benchmarked according to net assets alone. Benchmarking considers other financial measures, such as revenues, R&D expense, and cash. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 49 |
| Benefits Provides market competitive, yet cost-effective employment benefits. For Executive Directors, this includes private insurance covering medical, dental vision, as well as life and disability insurance. Directors and Officers liability insurance is also covered as well as either reimbursement of external advisor fees or external adviser assistance up to a maximum amount determined by the Remuneration Committee from time to time in connection with filing UK tax returns related to remuneration received in connection with the role. Other employment benefits may be provided from time to time on similar terms as those of other employees. If an Executive Director is based outside the UK additional benefits and assistance with relocation may be provided which reflect local market norms or legislation. There is no formal maximum level of benefits as the value of insured benefits will vary from year to year based on the cost from third-party providers. None. Executive Directors may receive either reimbursement of external adviser fees or external adviser assistance in connection up to a maximum amount determined by the Remuneration Committee with filing UK tax returns related to remuneration received in connection with the role. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 50 |
| Annual bonus To incentivize and award delivery of the Company's strategy and corporate objectives on an annual basis. Annual bonus performance targets are set at the start of the year by the Board and performance against objectives is assessed by the Remuneration Committee after the end of the relevant financial year. Bonuses will be paid in cash. The maximum annual bonus payable to an Executive Director is 150% of base salary. In exceptional circumstances, the Committee may determine that the maximum bonus opportunity will be 200% of base salary. There is no formal minimum annual bonus as the bonus payable depends on performance against objectives. Research and development, business development, financial and commercial targets are set at the start of the year by the Board. Details of the performance measures for the current year are provided in the Directors' Remuneration Report, subject to any non-disclosure on the basis of commercially-sensitive information. N/A VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 51 |
| Equity incentives To align the interests of Executive Directors and management with long-term shareholder interests and to attract, incentivize and retain staff. To incentivize and recognize achievement of longer-term corporate objectives and sustained shareholder value creation. To effectively manage the Group's cash resources. Conditional awards are granted annually under the 2017 Incentive Plan. The awards vest over a period of at least three years and may include a mix of share options, restricted share units, performance shares and other awards available for issuance under the 2017 Incentive Plan. Awards may be subject to clawback under the terms of any policy adopted by the Company or required by any applicable laws. The total number of awards made under the 2017 Incentive Plan is subject to the overall limits set out in the 2017 Incentive Plan. There is no formal minimum level of equity incentives as the grant of equity incentives to the Executive Director is in the discretion of the Board. Vesting may be on a time-phased basis or subject to performance conditions, as determined in the discretion of the Committee. N/A Pension To provide a competitive and tax-efficient pension savings plan which complies with at least the minimum contributions requirements of the applicable jurisdiction. Executive Directors are eligible to join a defined contribution pension scheme. The maximum contribution, cash supplement (or combination thereof) payable by the Company is 4% of salary, or such statutory minimum as may be required. None. N/A The Committee operates the annual bonus and 2017 Incentive Plan, in accordance with their rules, and where relevant, the SEC Rules. To maintain an efficient administrative process, the Committee retains the following discretion relating to remuneration: • the eligibility to participate in the plans; • the timing of grant of awards and any payments; • the size of awards and payments (subject to the maximum limits set out in the Policy table above and the respective plan rules); • the determination of whether any performance conditions have been met; • determining a good or bad leaver under the terms of the plans; • adjustments required and treatment of awards in connection with certain events such as rights issues, corporate restructuring, capital events, change in control or other corporate events or special dividends; and • the annual review of performance objectives for the annual bonus plan and, if applicable, the 2017 Incentive Plan. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 52 |
| In certain exceptional circumstances, such as a material acquisition/divestment of a Group business or a change in the broader business environment, which mean the original performance conditions are no longer appropriate, the Committee may adjust the objectives, alter weightings or set different measures as necessary, to ensure the conditions achieve their original purpose and are not materially less difficult to satisfy. Historical equity incentive awards Awards which were granted prior to January 1, 2017 are disclosed separately in this Remuneration Report. These awards remain eligible to vest, based on their original terms which are described separately in the Directors' Report on Remuneration. Annual bonus The annual bonus is designed to drive the achievement of the Company’s strategic and corporate objectives. These targets are agreed by the Board and selected because of their importance in value creation for shareholders. Remuneration on recruitment The remuneration package for any new Executive Director will be determined by the Remuneration Committee in accordance with the terms of the Policy at the time of appointment (including salary, benefits, annual bonus, long-term incentive awards and pension). It is recognised that in order to attract and recruit talented individuals the Policy needs to allow sufficient flexibility with respect to remuneration on recruitment. The following policies apply to the remuneration on recruitment of new Executive Directors: • Salary: Base salary will be determined based on the responsibilities of the role, experience of the individual and current market rates. It may be considered necessary to appoint a new Executive Director on or below market rates (e.g. to reflect limited board experience). In such circumstances, phased increases above those of the wider workforce may be required over an appropriate time period, to bring the salary to the desired market level, subject to the continued development in the role. • Annual bonus: The ongoing annual bonus maximum will be in line with that outlined in the Policy table for existing Executive Directors, pro-rated to reflect the period of service. Depending on the timing or nature of an appointment it may be necessary to set different initial performance measures and targets for the first year of appointment. • Long-term incentive awards: 2017 Incentive Plan awards are granted in line with the policy outlined for existing Executive Directors. An award may be made shortly following an appointment (provided the Company is not in a closed period under its Insider Trading Compliance Policy). For internal appointments, existing awards will continue on their original terms. • Benefits: Benefits provided should be in line with those of existing Executive Directors. For external and internal appointments, where required to meet business needs, reasonable relocation support will be provided. In addition, if it becomes necessary to appoint a new Executive Director from outside the UK, additional benefits may be provided to reflect local market norms or legislation. • Pension: A company contribution or cash supplement up to the maximum as outlined for existing Executive Directors. • Sign-on payments and buy-out awards: To enable the recruitment of exceptional talent, the Committee may offer additional cash and/or share-based remuneration to attract such talent and/or to take account of and compensate for remuneration that the Director is required to relinquish when leaving a former employer. The Committee will seek to structure any such replacement awards to be no more generous overall in terms of quantum or vesting than the award to be forfeited from the previous employer and will take into account the timing, form and performance requirements of the awards forgone. Where appropriate, any long-term incentive awards will be granted under the 2017 Incentive Plan, however, the Remuneration Committee will have discretion to make use of the flexibility to make awards under any relevant exemptions in the SEC Rules. • For an internal Executive Director appointment, any variable pay element awarded in respect of the prior role will be allowed to pay out according to its terms. In addition, any other contractual remuneration obligations existing prior to appointment may continue. • The fees for any new Chairperson and non-Executive Director appointments will be set in accordance with the prevailing policy and at a level that is consistent with those of the existing Chairperson and non-Executive Directors. Policy for payments on loss of office The Company does not have a policy of fixed term employment contracts, however, in accordance with the Company’s Articles of Association, one third of Directors put themselves forward for re-election at each Annual General Meeting. The existing Executive Director’s employment contract may be terminated by either party at any time and for any reason. The existing Chairperson’s and non-Executive Directors’ letters of appointment may be terminated by either party at any time and for any reason upon three months’ notice from either party. The Committee’s approach to payments in the event that an Executive Director’s employment is terminated is to take account of the individual circumstances including the reason for termination, individual performance, contractual obligations and the terms of the equity incentive plans in which the Executive Director participates. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 53 |
| Termination of the Executive Director’s employment agreement by the Company “without cause” or by the Executive Director for “good reason” (as those terms are defined in the Executive Director’s employment agreement): payment of up to 150% of base salary, maximum annual bonus and health insurance for 18 months. Long-term incentives: whether any long-term incentive awards would vest and be exercisable upon loss of office would be subject to the contractual agreement with the Executive Director and the relevant plan rules under which such award was granted, which allow vesting and exercise of awards in the event of death, retirement, ill-health, injury, redundancy and any other reason at the discretion of the Remuneration Committee. Subject to any contractual agreement, the Committee retains discretion to determine the extent to which the award will vest, taking into consideration the circumstances. Unvested awards normally lapse, although the Committee retains the power to determine, in accordance with the “good leaver” provisions of the relevant plan rules, what proportion of unvested awards will be retained and what proportion will lapse. In determining this, the Committee will give consideration to the reason for leaving, the extent of achievement of performance objectives at the date of leaving and may decide to time pro-rate awards. On a change of control, all unvested awards vest on the date of change of control. Additional payments: The Committee reserves the right to make payments it considers reasonable under a compromise or settlement agreement, including payment or reimbursement of reasonable legal and professional fees, untaken holiday and any payment in respect of statutory rights under employment law in the UK or other jurisdictions. Payment or reimbursement of reasonable outplacement fees may also be provided. Remuneration Policy for Non-Executive Directors The Remuneration Committee is responsible for evaluating and making recommendations to the Board on fees payable to the Chairperson. The Chairperson does not participate in discussions in respect of fees. The Chairperson and Chief Executive Officer are responsible for evaluating and making recommendations to the Board on the fees payable to the Company’s non-Executive Directors. Element of Remuneration Purpose and link to strategy Operation and Maximum Change to 2021 Policy Chairperson’s fee To attract and retain a high calibre individual with the requisite experience and knowledge. The current fee is set out in the implementation of policy section of the Directors' Remuneration Report. There is no formal maximum. Fees are periodically benchmarked against a relevant peer group of life sciences companies, many of which are listed on Nasdaq, with a similar stage of development, and similar market capitalization and financial profile to ensure they remain competitive and adequately reflect the time commitments and scope of the role. Supplemental fees may be paid for chairpersonship and membership of Committees to recognize the additional time commitments and responsibilities of these roles. Any increase in fee levels may be above that of the wider workforce in a particular year to reflect the periodic nature of any review and/or any change in responsibilities/time commitments. The Chairperson may also receive limited travel and/or hospitality related benefits and either reimbursement of external advisor fees or external adviser assistance up to a maximum amount determined by the Remuneration Committee from time to time in connection with filing UK tax returns related to fees received in connection with the role. The Chairperson may not receive any consultancy or other payments outside the Chairperson's fee. The Chairperson may be paid in a combination of cash and equity. (i) Benchmarking is against a peer group of life sciences companies, many of which are listed on Nasdaq, with a similar stage of development, and similar market capitalization and financial profile. (ii) The Chairperson may receive either reimbursement of external adviser fees or external adviser assistance up to a maximum amount determined by the Remuneration Committee in connection with filing UK tax returns related to fees received in connection with the role. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 54 |
| Non-Executive Director fee To attract and retain high calibre individuals with the requisite experience and knowledge. The current fee levels are set out in the implementation of policy section of the Directors' Remuneration Report. There is no formal maximum. Fees are periodically benchmarked against a relevant peer group of life sciences companies, many of which are listed on Nasdaq, with a similar stage of development, and similar market capitalization and financial profile to ensure they remain competitive and adequately reflect the time commitments and scope of the role. A Board fee is paid to each non-Executive Director. Supplemental fees may be paid to the Senior Independent Director and for chairpersonship and membership of Committees to recognize the additional time commitments and responsibilities of these roles. Any increase in fee levels may be above that of the wider workforce in a particular year to reflect the periodic nature of any review and/or any change in responsibilities/time commitments. If business needs arise, non-Executive Directors may also be engaged to provide limited consulting services outside their director responsibilities and receive fees for those services. Non-Executive Directors may also receive limited travel and/or hospitality related benefits and either reimbursement of external advisor fees or external adviser assistance up to a maximum amount determined by the Remuneration Committee from time to time in connection with filing UK tax returns related to fees received in connection with the role. Non-Directors may be paid in a combination of cash and equity. (i) Benchmarking is against a peer group of life sciences companies, many of which are listed on Nasdaq, with a similar stage of development, and similar market capitalization and financial profile. (ii) The non-Executive Director may receive either reimbursement of external adviser fees or external adviser assistance up to a maximum amount determined by the Remuneration Committee in connection with filing UK tax returns related to fees received in connection with the role. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 55 |
| Illustrations of Minimum, Expected, and Maximum remuneration for the Executive Director Scenarios The charts set out for illustrative purposes only, what annual remuneration the Company expects the Executive Director, Dr. David Zaccardelli, to obtain at minimum, expected and maximum achievement of performance targets with respect to the financial year ending December 31, 2024. The assumptions used in the calculations are set out below: Fixed base salary includes: • base salary of $873,015 per annum, all of which is paid in cash; and • benefits. Minimum: this illustration assumes fixed base salary, as set out above, and as the annual bonus is discretionary, no annual bonus. Expected: this illustration assumes the fixed base salary, as set out above, plus achievement of the full discretionary annual bonus of 50% of base salary, being $436,507 for the financial year ending December 31, 2024. This illustration assumes no additional grant is made under the 2017 Incentive Plan. Maximum: this illustration assumes the fixed base salary, as set out above, and as the annual bonus is discretionary, we make the assumption that the Executive Director receives the maximum bonus permitted under the Remuneration Policy of 150% of base salary, being $1,309,522 for the financial year ending December 31, 2024. This illustration assumes no additional grant is made under the 2017 Incentive Plan. VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 56 |
| Consideration of shareholder views The views of shareholders and their advisory bodies are also important to the Company and the Committee takes its responsibility to shareholders seriously. The Committee will consider any shareholder feedback received at the AGM and throughout the year when reviewing and applying the Remuneration Policy, and the guidance from shareholder advisory bodies will be considered on an ongoing basis. The Committee will continue to seek to build and maintain an open and constructive dialogue with shareholders on our approach to remuneration. Consideration of employment conditions elsewhere in the Company The Committee generally considers pay and employment conditions elsewhere in the Company when considering the Directors’ remuneration. While the Company gave consideration to these factors there was no formal consultation with employees when developing the Remuneration Policy. The remuneration arrangements for Executive Directors outlined in this Policy are broadly consistent with those for other senior executives in the Company although award opportunities and quantum may vary by seniority and responsibility. The Committee is fully informed of, and considers wider employee remuneration and related policies including salary increases, operation of incentive plans, bonus opportunities and total remuneration levels apply across the wider workforce, when determining director remuneration. On behalf of the Board Dr. Ken Cunningham Chair of the Remuneration Committee VERONA PHARMA PLC DIRECTORS’ REMUNERATION REPORT FOR THE YEAR ENDED DECEMBER 31, 2023 57 |
| Report on the audit of the financial statements Opinion In our opinion, Verona Pharma Plc's group financial statements and company financial statements (the "financial statements"): give a true and fair view of the state of the group's and of the company's affairs as at 31 December 2023 and of the group's loss and the group's and company's cash flows for the year then ended; have been properly prepared in accordance with UK-adopted international accounting standards as applied in accordance with the provisions of the Companies Act 2006; and have been prepared in accordance with the requirements of the Companies Act 2006. We have audited the financial statements, included within the Annual Report and Accounts (the "Annual Report"), which comprise: the consolidated and company statements of financial position as at 31 December 2023; the consolidated statement of comprehensive income, the consolidated and company statements of cash flows and the consolidated and company statements of changes in equity for the year then ended; and the notes to the financial statements, which include a description of the significant accounting policies. Basis for opinion We conducted our audit in accordance with International Standards on Auditing (UK) ("ISAs (UK)") and applicable law. Our responsibilities under ISAs (UK) are further described in the Auditors' responsibilities for the audit of the financial statements section of our report. We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our opinion. Independence We remained independent of the group in accordance with the ethical requirements that are relevant to our audit of the financial statements in the UK, which includes the FRC's Ethical Standard, as applicable to listed entities, and we have fulfilled our other ethical responsibilities in accordance with these requirements. Our audit approach Overview Audit scope Verona Pharma plc has one wholly-owned subsidiary, Verona Pharma, Inc. Both entities were deemed to be significant components and required a full scope audit based on their size. The group audit team conducted all necessary audit procedures with no component auditors supporting the group audit team. Both entities together represent 100% of the group loss before tax and 100% of the group's total assets. Key audit matters Valuation of the assumed contingent liability (group and parent) Accounting for the modification of a term loan facility (group) Materiality Overall group materiality: $2.8 million (2022: $4.0 million) based on 5% of loss before tax excluding the impact of discount unwind on the assumed contingent liability. Overall company materiality: $3.2 million (2022: $3.7 million) based on 5% of loss before tax excluding the impact of discount unwind on the assumed contingent liability. Performance materiality: $2.1 million (2022: $3.0 million) (group) and $2.4 million (2022: $2.8 million) (company). The scope or our audit As part of designing our audit, we determined materiality and assessed the risks of material misstatement in the financial statements. VERONA PHARMA PLC INDEPENDENT AUDITORS’ REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 58 |
| Key audit matters Key audit matters are those matters that, in the auditors' professional judgement, were of most significance in the audit of the financial statements of the current period and include the most significant assessed risks of material misstatement (whether or not due to fraud) identified by the auditors, including those which had the greatest effect on: the overall audit strategy; the allocation of resources in the audit; and directing the efforts of the engagement team. These matters, and any comments we make on the results of our procedures thereon, were addressed in the context of our audit of the financial statements as a whole, and in forming our opinion thereon, and we do not provide a separate opinion on these matters. This is not a complete list of all risks identified by our audit. Accounting for the modification of a term loan facility is a new key audit matter this year. Accuracy, cut off and rights and obligations of R&D expenses, which was a key audit matter last year, is no longer included because of the finalisation of the Phase 3 clinical trials during early 2023, which has reduced the estimation uncertainty required in determining the amount of R&D expense incurred during the year. Otherwise, the key audit matters below are consistent with last year. VERONA PHARMA PLC INDEPENDENT AUDITORS’ REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 59 |
| VERONA PHARMA PLC INDEPENDENT AUDITORS' REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 Key audit matter How our audit addressed the key audit matter Valuation of the assumed contingent liability (group and parent) On 19 September 2006, Verona Pharma pie acquired We obtained management's model calculating the estimated RhinoPharma Ltd which held contingent liabilities relating liability and performed the following procedures: to future potential milestone and royalty payments now due to Ligand relating to the acquisition of rights to certain - Assessed the appropriateness of the model used in patents and patent applications for ensifentrine and related estimating the projected cashflows; compounds. - Verified the mathematical accuracy of the model; - Tested the completeness and accuracy of the model as Per IFRS 3, the existing contingent payments of the well as the underlying data used, including agreeing key acquiree are an assumed liability of the buyer. inputs to market research performed by management's Consequently, Verona Pharma pie fair valued the expert; contingent liability on the date of acquisition and recorded - Substantiated the probability of success applied within the it on the balance sheet. At each subsequent period end, calculation back to publicly available industry data regarding the liability is required to be re-measured when there is a the average success of drugs moving from successful change in the estimated future payments such as an phase 3 clinical trials to submission and subsequent improved probability of success. acceptance of the New Drug Application to the FDA for approval; and The submission and subsequent acceptance of the New - Assessed the reliability, objectivity and competence of Drug Application to the FDA following receipt of positive management's experts utilised in developing the model and clinical trial data in 2023 was deemed to represent a agreed the inputs used in the model to the reports from change in the probability of success. Management have these experts. We further utilised our in-house valuation also updated their revenue forecasts, which combined with experts to assess the valuation techniques used and to the above events have resulted in a re-measurement of assist with the evaluation of key assumptions made and the the contingent liability leading to an increase to the liability sources of data used. of $60.1 million, with a corresponding increase to the Subsequent to the remeasurement triggered in the year, we associated IP R&D intangible asset. Management obtained management's confirmation that there were no assessed that there had been no further triggers to re- further changes to the expected cash flows at year end and measure the liability in the period. verified the reasonableness of this by performing the below procedures: The process involved in the valuation of the contingent - Inquired of management whether there were any further liability is complex and subject to estimation uncertainty. changes to the market or probability of success; The value of the contingent consideration was $215.4 - Reviewed the minutes of meetings of the Board of million at 31 December 2023 (31 December 2022: $138.3 Directors for any indication of changes in the expected million) following discount unwind and foreign currency cashflows and probabilities of success; and movements. Refer also to Note 22 to the consolidated financial statements. - Conducted independent research into whether there were any material changes to the underlying COPD market including new competitor drugs. Tested the mathematical accuracy of the finance charge arising from the unwinding of the discount rate; and Considered the disclosures in Note 22 of the Annual Report and Accounts, including sensitivity analyses based on reasonably possible changes. We are satisfied that these disclosures are appropriate. Based on the work performed, we have concluded that management's assumptions are reasonable. Accounting for the modification of a term loan facility (group) As described in Note 23 to the consolidated financial We obtained management's calculation and accounting statements, on 27 December 2023 (the "2023 Effective paper in respect of the modification, and performed the Date"), Verona Pharma, Inc. entered into a term loan following procedures: facility of up to $400.0 million (the "2023 Term Loan" or "Loan Agreement"), consisting of a term loan advance in - Tested the completeness and accuracy of management's an aggregate amount of $50.0 million funded on the 2023 calculation as well as underlying data used; Effective Date ("the Term A Loan") and four additional term - Evaluated management's assessment regarding the loan advances subject to certain terms and conditions, accounting for the new loan facility, in particular with respect The 2023 Term Loan replaced the Group's existing $150.0 to their assessment of modification and extinguishment in million facility. Verona Pharma, Inc. received net proceeds accordance with IFRS 9; and from the Term A Loan partially offset by the repayment, in full, of the existing outstanding indebtedness owed by the - Evaluated the sufficiency of the disclosures in Note 23 of Group under the previous Term Loan of $20 million. the Annual Report and Accounts. Where the terms of the new debt instruments and old debt Based on the work performed, we have concluded that instruments are not "substantially different", the debt may management's accounting treatment is reasonable. be considered modified rather than extinguished, requiring modification accounting. Based upon management's evaluation of the accounting for the Loan Agreement, management has applied modification accounting to a portion of the Term A Loan in accordance with IFRS 9. VERONA PHARMA PLC INDEPENDENT AUDITORS’ REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 60 |
| How we tailored the audit scope We tailored the scope of our audit to ensure that we performed enough work to be able to give an opinion on the financial statements as a whole, taking into account the structure of the group and the company, the accounting processes and controls, and the industry in which they operate. We tailored the scope of our audit to ensure that we have gained sufficient audit evidence to be able to give an opinion on the financial statements as a whole, taking into account the structure of the group and the company, the accounting processes and controls, and the industry in which they operate. No component auditors supported the group audit team, which conducted all necessary audit procedures. We agreed with the Audit and Risk Committee that we would report to them misstatements identified during our audit above $0.14 million (group audit) (2022: $0.20 million) and $0.16 million (company audit) (2022: $0.18 million) as well as misstatements below those amounts that, in our view, warranted reporting for qualitative reasons The impact of climate risk on our audit As part of our audit we made enquiries of management to understand the extent of the potential impact of climate risk on the group's and company's financial statements, and we remained alert when performing our audit procedures for any indicators of the impact of climate risk. Our procedures did not identify any material impact as a result of climate risk on the group's and company's financial statements. Materiality The scope of our audit was influenced by our application of materiality. We set certain quantitative thresholds for materiality. These, together with qualitative considerations, helped us to determine the scope of our audit and the nature, timing and extent of our audit procedures on the individual financial statement line items and disclosures and in evaluating the effect of misstatements, both individually and in aggregate on the financial statements as a whole. Based on our professional judgement, we determined materiality for the financial statements as a whole as follows: Financial statements - group Financial statements - company Overall materiality $2.8 million (2022: $4.0 million). $3.2 million (2022: $3.7 million). How we determined it 5% of loss before tax excluding the impact of discount unwind on the assumed contingent liability 5% of loss before tax excluding the impact of discount unwind on the assumed contingent liability Rationale for benchmark applied Based on the benchmarks used in the annual report, loss before tax is the primary measure used by the shareholders in assessing the financial performance of the group and is a generally accepted auditing benchmark. We have adjusted this to exclude the impact of the annual discount unwind on the Assumed contingent liability as this is non-cash and can vary significantly each period. As a result of this, it can cause significant movements in the loss before tax. Although large in size, this is a non-cash item which we assess has limited impact on a user of the financial statements. Based on the benchmarks used in the annual report, loss before tax is the primary measure used by the shareholders in assessing the financial performance of the group and is a generally accepted auditing benchmark. We have adjusted this to exclude the impact of the annual discount unwind on the Assumed contingent liability as this is non-cash and can vary significantly each period. As a result of this, it can cause significant movements in the loss before tax. Although large in size, this is a non-cash item which we assess has limited impact on a user of the financial statements. For each component in the scope of our group audit, we allocated a materiality that is less than our overall group materiality. The range of materiality allocated across components was $2.0 million to $2.5 million. We use performance materiality to reduce to an appropriately low level the probability that the aggregate of uncorrected and undetected misstatements exceeds overall materiality. Specifically, we use performance materiality in determining the scope of our audit and the nature and extent of our testing of account balances, classes of transactions and disclosures, for example in determining sample sizes. Our performance materiality was 75% (2022: 75%) of overall materiality, amounting to $2.1 million (2022: $3.0 million) for the group financial statements and $2.4 million (2022: $2.8 million) for the company financial statements. In determining the performance materiality, we considered a number of factors - the history of misstatements, risk assessment and aggregation risk and the effectiveness of controls - and concluded that an amount at the upper end of our normal range was appropriate. We agreed with those charged with governance that we would report to them misstatements identified during our audit above $0.14 million (group audit) (2022: $0.2 million) and $0.16 million (company audit) (2022: $0.18 million) as well as misstatements below those amounts that, in our view, warranted reporting for qualitative reasons. VERONA PHARMA PLC INDEPENDENT AUDITORS’ REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 61 |
| Conclusions relating to going concern Our evaluation of the directors' assessment of the group's and the company's ability to continue to adopt the going concern basis of accounting included: Testing the mathematical integrity of the cash flow forecast and model and reconciling these to the Board approved budget; Understanding and assessing the impact of the 2023 Term Loan and considering the completeness and timing of costs included within the cash flow forecast over the going concern assessment period; and Assessing management's ability to forecast by comparing the budget for the year ended 31 December 2023 against the actuals and understanding the cause of key variances. Based on the work we have performed, we have not identified any material uncertainties relating to events or conditions that, individually or collectively, may cast significant doubt on the group's and the company's ability to continue as a going concern for a period of at least twelve months from when the financial statements are authorised for issue. In auditing the financial statements, we have concluded that the directors' use of the going concern basis of accounting in the preparation of the financial statements is appropriate. However, because not all future events or conditions can be predicted, this conclusion is not a guarantee as to the group's and the companys ability to continue as a going concern. Our responsibilities and the responsibilities of the directors with respect to going concern are described in the relevant sections of this report. Reporting on other information The other information comprises all of the information in the Annual Report other than the financial statements and our auditors' report thereon. The directors are responsible for the other information. Our opinion on the financial statements does not cover the other information and, accordingly, we do not express an audit opinion or, except to the extent otherwise explicitly stated in this report, any form of assurance thereon. In connection with our audit of the financial statements, our responsibility is to read the other information and, in doing so, consider whether the other information is materially inconsistent with the financial statements or our knowledge obtained in the audit, or otherwise appears to be materially misstated. If we identify an apparent material inconsistency or material misstatement, we are required to perform procedures to conclude whether there is a material misstatement of the financial statements or a material misstatement of the other information. If, based on the work we have performed, we conclude that there is a material misstatement of this other information, we are required to report that fact. We have nothing to report based on these responsibilities. With respect to the Strategic report and Directors' Report, we also considered whether the disclosures required by the UK Companies Act 2006 have been included. Based on our work undertaken in the course of the audit, the Companies Act 2006 requires us also to report certain opinions and matters as described below. Strategic report and Directors' Report In our opinion, based on the work undertaken in the course of the audit, the information given in the Strategic report and Directors' Report for the year ended 31 December 2023 is consistent with the financial statements and has been prepared in accordance with applicable legal requirements. In light of the knowledge and understanding of the group and company and their environment obtained in the course of the audit, we did not identify any material misstatements in the Strategic report and Directors' Report. Directors' Remuneration In our opinion, the part of the Directors' Report to be audited has been properly prepared in accordance with the Companies Act 2006. VERONA PHARMA PLC INDEPENDENT AUDITORS’ REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 62 |
| Responsibilities for the financial statements and the audit Responsibilities of the directors for the financial statement As explained more fully in the Statement of Directors' responsibilities, the directors are responsible for the preparation of the financial statements in accordance with the applicable framework and for being satisfied that they give a true and fair view. The directors are also responsible for such internal control as they determine is necessary to enable the preparation of financial statements that are free from material misstatement, whether due to fraud or error. In preparing the financial statements, the directors are responsible for assessing the group's and the company's ability to continue as a going concern, disclosing, as applicable, matters related to going concern and using the going concern basis of accounting unless the directors either intend to liquidate the group or the company or to cease operations, or have no realistic alternative but to do so. Andifnrs' reSponnihilifinn fnr fha audit of the financial statements Our objectives are to obtain reasonable assurance about whether the financial statements as a whole are free from material misstatement, whether due to fraud or error, and to issue an auditors' report that includes our opinion. Reasonable assurance is a high level of assurance, but is not a guarantee that an audit conducted in accordance with ISAs (UK) will always detect a material misstatement when it exists. Misstatements can arise from fraud or error and are considered material if, individually or in the aggregate, they could reasonably be expected to influence the economic decisions of users taken on the basis of these financial statements. Irregularities, including fraud, are instances of non-compliance with laws and regulations. We design procedures in line with our responsibilities, outlined above, to detect material misstatements in respect of irregularities, including fraud. The extent to which our procedures are capable of detecting irregularities, including fraud, is detailed below. Based on our understanding of the group and industry, we identified that the principal risks of non-compliance with laws and regulations related to tax legislation and Companies Act 2006, and we considered the extent to which non-compliance might have a material effect on the financial statements. We evaluated management's incentives and opportunities for fraudulent manipulation of the financial statements (including the risk of override of controls), and determined that the principal risks were related to misappropriation of cash. Audit procedures performed by the engagement team included: Inquiries with management and internal legal counsel, including consideration of known or suspected instances of non-compliance with laws and regulations and fraud; Inspecting meeting minutes of the; Board of Directors, Audit and Risk, Disclosure, Remuneration, Nominations and Corporate Governance committees; Identifying and testing journal entries based on our risk assessment and evaluating whether there was evidence of management bias that represents a risk of material misstatement due to fraud; Consideration of assumptions and judgements made by management in their significant accounting estimates and judgements, particularly in relation to the key audit matters; and Incorporating elements of unpredictability into the audit procedures performed. There are inherent limitations in the audit procedures described above. We are less likely to become aware of instances of non-compliance with laws and regulations that are not closely related to events and transactions reflected in the financial statements. Also, the risk of not detecting a material misstatement due to fraud is higher than the risk of not detecting one resulting from error, as fraud may involve deliberate concealment by, for example, forgery or intentional misrepresentations, or through collusion. Our audit testing might include testing complete populations of certain transactions and balances, possibly using data auditing techniques. However, it typically involves selecting a limited number of items for testing, rather than testing complete populations. We will often seek to target particular items for testing based on their size or risk characteristics. In other cases, we will use audit sampling to enable us to draw a conclusion about the population from which the sample is selected. A further description of our responsibilities for the audit of the financial statements is located on the FRC's website at: www.frc.org.uk/auditorsresponsibilities. This description forms part of our auditors' report. This report, including the opinions, has been prepared for and only for the company's members as a body in accordance with Chapter 3 of Part 16 of the Companies Act 2006 and for no other purpose. We do not, in giving these opinions, accept or assume responsibility for any other purpose or to any other person to whom this report is shown or into whose hands it may come save where expressly agreed by our prior consent in writing. VERONA PHARMA PLC INDEPENDENT AUDITORS’ REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 63 |
| Other required reporting Companies Act 2006 exception reporting Under the Companies Act 2006 we are required to report to you if, in our opinion: we have not obtained all the information and explanations we require for our audit; or adequate accounting records have not been kept by the company, or returns adequate for our audit have not been received from branches not visited by us; or certain disclosures of directors' remuneration specified by law are not made; or the company financial statements and the part of the Directors' Report to be audited are not in agreement with the accounting records and returns. We have no exceptions to report arising from this responsibility. David Farmer (Senior Statutory Auditor) for and on behalf of PricewaterhouseCoopers LLP Chartered Accountants and Statutory Auditors Reading 18 March 2024 VERONA PHARMA PLC INDEPENDENT AUDITORS’ REPORT TO THE MEMBERS OF VERONA PHARMA PLC FOR THE YEAR ENDED DECEMBER 31, 2023 64 |
| Note Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Revenue — 458 Costs of sales — (346) Gross profit — 112 Operating expenses Research and development costs 7 (17,413) (50,339) Selling, general and administrative costs 7 (51,280) (27,840) Operating loss (68,693) (78,067) Finance income 9 14,627 2,821 Finance expense 9 (18,889) (9,516) Loss before taxation (72,955) (84,762) Taxation — credit 10 630 9,381 Loss for the year (72,325) (75,381) Total comprehensive loss attributable to owners of the Company (72,325) (75,381) Loss per ordinary share — basic and diluted (cents) 5 (11.4) (14.2) The accompanying notes form an integral part of these consolidated financial statements. VERONA PHARMA PLC CONSOLIDATED STATEMENT OF COMPREHENSIVE INCOME FOR THE YEAR ENDED DECEMBER 31, 2023 65 |
| Note As of December 31, 2023 As of December 31, 2022 $'000s $'000s ASSETS Non-current assets: Goodwill 11 545 545 Intangible assets 12 191,170 130,798 Property, plant and equipment 24 73 Right-of-use assets 13 2,847 854 Equity interest 14 15,000 15,000 Total non-current assets 209,586 147,270 Current assets: Prepayments and other receivables 16 6,982 5,887 Current tax receivable 10,954 9,282 Cash and cash equivalents 271,772 227,827 Total current assets 289,708 242,996 Total assets 499,294 390,266 EQUITY AND LIABILITIES Capital and reserves attributable to equity holders: Share capital 17 42,771 40,526 Share premium 522,918 465,370 Share-based payment reserve 81,012 63,817 Cumulative translation adjustment (5,796) (5,796) Accumulated loss (416,085) (342,793) Treasury shares (1,517) (1,549) Total equity 223,303 219,575 Current liabilities: Lease liability 13 1,180 675 Trade and other payables 21 9,258 21,502 Tax payable - U.S. operations — 283 Total current liabilities 10,438 22,460 Non-current liabilities: Assumed contingent liability 22 215,404 138,258 Term loan 23 48,374 9,768 Non-current lease liability 13 1,775 205 Total non-current liabilities 265,553 148,231 Total equity and liabilities 499,294 390,266 The accompanying notes form an integral part of these consolidated financial statements. VERONA PHARMA PLC CONSOLIDATED STATEMENT OF FINANCIAL POSITION AS OF DECEMBER 31, 2023 66 |
| Note As of December 31, 2023 As of December 31, 2022 $'000s $'000s ASSETS Non-current assets: Goodwill 11 545 545 Intangible assets 12 191,170 130,798 Property, plant and equipment — 7 Right-of-use asset 516 489 Equity interest 14 15,000 15,000 Total non-current assets 207,231 146,839 Current assets: Prepayments and other receivables 16 5,849 5,853 Current tax receivable 10,954 9,282 Cash and cash equivalents 239,532 227,362 Total current assets 256,335 242,497 Total assets 463,566 389,336 EQUITY AND LIABILITIES Capital and reserves attributable to equity holders: Share capital 17 42,771 40,526 Share premium 522,918 465,370 Share-based payment reserve 81,012 63,817 Cumulative Translation Adjustment (5,942) (5,942) Accumulated loss (434,346) (352,824) Treasury shares (1,517) (1,549) Total equity 204,896 209,398 Current liabilities: Lease Liability 506 401 Trade and other payables 21 42,714 31,448 Total current liabilities 43,220 31,849 Non-current liabilities: Assumed contingent liability 22 215,404 138,258 Term loan 23 — 9,768 Non-current lease liability 46 63 Total non-current liabilities 215,450 148,089 Total equity and liabilities 463,566 389,336 The accompanying notes form an integral part of these company financial statements. The Company has taken advantage of the exemption permitted by Section 408 of the Companies Act 2006 not to present an income statement for the year. The Company's loss for the year was $80.6 million (2022: loss of $78.2 million), which has been included in the Group’s income statement. The financial statements on pages 65 to 102 were approved by the Company's board of directors on March 18, 2024, and signed on its behalf by Dr. David Zaccardelli, Chief Executive Officer of the Company. Dr. David Zaccardelli Director and Chief Executive Officer of the Company Company number: 05375156 VERONA PHARMA PLC COMPANY STATEMENT OF FINANCIAL POSITION AS OF DECEMBER 31, 2023 67 |
| Note Share capital Share premium Treasury shares Share-based payment reserve Cumulative translation adjustment Total accumulated losses Total equity $'000s $'000s $'000s $'000s $'000s $'000s $'000s Balance at January 1, 2022 17 31,855 330,779 (603) 54,291 (5,796) (266,732) 143,794 Total comprehensive loss — — — — — (75,381) (75,381) New share capital issued 6,918 142,812 — — — — 149,730 Transaction costs on share capital issued — (9,533) — — — — (9,533) Shares issued under at-the-market sales agreement 5 62 — — — — 67 Shares issued to treasury 1,748 — (1,748) — — — — Restricted share units vested — — 680 — — (680) — Common shares withheld for taxes on vested stock awards — — — (4,723) — — (4,723) Equity settled share-based compensation reclassified as cash-settled — — — 128 — — 128 Share options exercised — 1,250 122 — — — 1,372 Share-based payments — — — 14,121 — — 14,121 Balance at December 31, 2022 17 40,526 465,370 (1,549) 63,817 (5,796) (342,793) 219,575 Balance at January 1, 2023 17 40,526 465,370 (1,549) 63,817 (5,796) (342,793) 219,575 Total comprehensive loss — — — — — (72,325) (72,325) Shares issued under at-the-market sales agreement 1,227 55,682 — — — — 56,909 Shares issued to treasury 1,018 — (1,018) — — — — Restricted share units vested — — 967 — — (967) — Common shares withheld for taxes on vested stock awards — — — (4,389) — — (4,389) Equity settled share-based compensation reclassified as cash-settled — — — (295) — — (295) Share options exercised — 1,866 83 — — — 1,949 Share-based payments — — — 21,879 — — 21,879 Balance at December 31, 2023 17 42,771 522,918 (1,517) 81,012 (5,796) (416,085) 223,303 VERONA PHARMA PLC CONSOLIDATED STATEMENT OF CHANGES IN EQUITY FOR THE YEAR ENDED DECEMBER 31, 2023 68 |
| Note Share capital Share premium Treasury shares Share-based payment reserve Cumulative translation adjustment Total accumulated losses Total equity $'000s $'000s $'000s $'000s $'000s $'000s $'000s Balance at January 1, 2022 17 31,855 330,779 (603) 54,291 (5,942) (273,911) 136,469 Total comprehensive loss — — — — — (78,233) (78,233) New share capital issued 6,918 142,812 — — — — 149,730 Transaction costs on share capital issued — (9,533) — — — — (9,533) Shares issued under at-the-market sales agreement 5 62 — — — — 67 Shares issued to treasury 1,748 — (1,748) — — — — Restricted share units vested — — 680 — — (680) — Common shares withheld for taxes on vested stock awards — — — (4,723) — — (4,723) Equity settled share-based compensation reclassified as cash-settled — — — 128 — — 128 Share options exercised — 1,250 122 — — — 1,372 Share-based payments — — — 14,121 — — 14,121 Balance at December 31, 2022 17 40,526 465,370 (1,549) 63,817 (5,942) (352,824) 209,398 Balance at January 1, 2023 17 40,526 465,370 (1,549) 63,817 (5,942) (352,824) 209,398 Total comprehensive loss — — — — — (80,555) (80,555) Shares issued under at-the-market sales agreement 1,227 55,682 — — — — 56,909 Shares issued to treasury 1,018 — (1,018) — — — — Restricted share units vested — — 967 — — (967) — Common shares withheld for taxes on vested stock awards — — — (4,389) — — (4,389) Equity settled share-based compensation reclassified as cash-settled — — — (295) — — (295) Share options exercised — 1,866 83 — — — 1,949 Share-based payments — — — 21,879 — — 21,879 Balance at December 31, 2023 17 42,771 522,918 (1,517) 81,012 (5,942) (434,346) 204,896 VERONA PHARMA PLC COMPANY STATEMENT OF CHANGES IN EQUITY FOR THE YEAR ENDED DECEMBER 31, 2023 69 |
| Note Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Cash used in operating activities: Loss before taxation (72,955) (84,762) Finance income 9 (14,627) (2,821) Finance expense 9 18,889 9,516 Share-based payment charge 21,879 14,121 Amortization of debt issue costs 68 80 Accretion of redemption premium on debt 106 108 Interest paid (2,006) (348) Increase in prepayments and other receivables (607) 230 Decrease in trade and other payables (12,434) (11,748) Depreciation of property, plant, equipment and right of use asset 678 636 Loss on disposal of property, plant and equipment 60 — Unrealized foreign exchange gain/(loss) 180 (426) Amortization of intangible assets 252 217 Cash used in operating activities before taxation (60,517) (75,197) Cash (outflow)/inflow from taxation (1,245) 13,478 Net cash used in operating activities (61,762) (61,719) Cash flows from investing activities: Interest received 12,761 2,821 Purchase of plant and equipment — (29) Payment for patents and computer software (509) (336) Net cash generated from investing activities 12,252 2,456 Cash flow used in financing activities: Gross proceeds from issue of shares 56,909 149,797 Transactions costs on issue of shares — (9,533) Proceeds from Oxford Term Loan 9,996 10,000 Proceeds from 2023 Term Loan, net of repayment of Oxford Term Loan and debt issuance costs incurred 28,712 — Payment of debt issuance costs (12) (245) Repayment of SVB Term Loan — (5,000) SVB Term Loan repayment costs — (850) Payment of finance lease liabilities (713) (628) Payments of withholding taxes from share-based award (4,684) (4,723) Proceeds from exercise of share options 1,949 1,372 Net cash generated from financing activities 92,157 140,190 Net increase in cash and cash equivalents 42,647 80,927 Cash and cash equivalents at the beginning of the year 227,827 148,380 Effect of exchange rates on cash and cash equivalents 1,298 (1,480) Cash and cash equivalents at the end of the year 271,772 227,827 VERONA PHARMA PLC CONSOLIDATED STATEMENT OF CASH FLOWS FOR THE YEAR ENDED DECEMBER 31, 2023 70 |
| Note Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Cash used in operating activities: Loss before taxation (81,658) (87,866) Finance income (14,620) (2,821) Finance expense 19,728 9,501 Share-based payment charge 21,879 14,121 Amortization of debt issue costs 85 80 Accretion of redemption premium on debt 103 108 Interest paid (1,928) (348) Decrease in prepayments and other receivables 4 182 Decrease in trade and other payables (9,655) (8,625) Depreciation of property, plant and equipment 422 451 Loss on disposal of property, plant and equipment 7 — Unrealized foreign exchange gains/(losses) 286 (426) Amortization of intangible assets 252 217 Cash used in operating activities before taxation (65,095) (75,426) Cash inflow from taxation — 13,598 Net cash used in operating activities (65,095) (61,828) Cash flows from investing activities: Interest received 12,754 2,821 Purchase of plant and equipment — (5) Payment for patents and computer software (509) (336) Net cash generated from investing activities 12,245 2,480 Cash flows from financing activities: Gross proceeds from issue of shares 56,909 149,797 Transactions costs on issue of shares — (9,533) Proceeds from Oxford Term Loan 9,996 10,000 Payment of debt issuance costs — (245) Repayment of SVB Term Loan — (5,000) SVB Term Loan repayment costs — (850) Payment of finance lease liabilities (448) (435) Payments of withholding taxes from share-based award (4,684) (4,723) Proceeds from exercise of share options 1,949 1,372 Net cash generated from financing activities 63,722 140,383 Net increase in cash and cash equivalents 10,872 81,035 Cash and cash equivalents at the beginning of the year 227,362 147,807 Effect of exchange rates on cash and cash equivalents 1,298 (1,480) Cash and cash equivalents at the end of the year 239,532 227,362 VERONA PHARMA PLC COMPANY STATEMENT OF CASH FLOWS FOR THE YEAR ENDED DECEMBER 31, 2023 71 |
| 1. General information Verona Pharma plc (the "Company") and its subsidiaries (together the "Group") are a clinical-stage biopharmaceutical group focused on developing and commercializing innovative therapeutics for the treatment of respiratory diseases with significant unmet medical needs. The Company is a public limited company, which is listed on the Nasdaq Global Market ("Nasdaq"). The company is incorporated and domiciled in the United Kingdom. The address of the registered office is One Central Square, Cardiff, CF10 1FS, United Kingdom. The Company has one subsidiary, Verona Pharma, Inc. which is wholly-owned. Rhinopharma Limited ("Rhinopharma"), a Canadian company that was previously a non-operating, wholly-owned subsidiary, was dissolved in June 2021. The Company listed its American Depositary Shares ("ADS") on Nasdaq in April 2017 ("the 2017 Global Offering") and they trade on the Nasdaq symbol "VRNA". 2. Accounting policies A summary of the material accounting policies, all of which have been applied consistently throughout the year, is set out below. 2.1 Basis of preparation The consolidated financial statements of the group have been prepared in accordance with UK-adopted international accounting standards and with the requirements of the Companies Act 2006 as applicable to companies reporting under those standards. The consolidated financial statements of the Group and the financial statements of the Company have been prepared under the historical cost convention, with the exception of the derivative financial liability and the equity interest, which have been measured at fair value. The preparation of financial statements in conformity with IFRS requires the use of certain critical accounting estimates. It also requires management to exercise its judgment in the process of applying the Group's accounting policies. The areas involving a higher degree of judgment or complexity, or areas where assumptions and estimates are significant to the consolidated financial statements are disclosed in note 4. Going concern The Group has incurred recurring losses since inception, including net losses of $72.3 million, $75.4 million and $59.3 million for the years ended December 31, 2023, 2022, and 2021, respectively. In addition, as of December 31, 2023, the Group had an accumulated loss of $416.1 million. The Group expects to continue to generate operating losses for the foreseeable future. As of the issuance date of the annual consolidated financial statements, the Group expects that its cash and cash equivalents, together with additional funding expected to become available under the 2023 Term Loan, will be sufficient to fund its operating expenses and capital expenditure requirements for at least the next 12 months from the date of approval of these finance statements. Accordingly, the consolidated financial statements have been prepared on the going concern basis. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 72 |
| 2.1 Basis of preparation (continued) Business combinations The Group applies the acquisition method to account for business combinations. The consideration transferred for the acquisition of a subsidiary is the fair value of the assets transferred, the liabilities incurred to the former owners of the acquiree, and the equity interests issued by the Group. The consideration transferred includes the fair value of any asset or liability resulting from a contingent consideration arrangement. The excess of the cost of acquisition over the fair value of the Group's share of the identifiable net assets acquired is recorded as goodwill. Goodwill arising on acquisitions is capitalized and is subject to impairment review, both annually and when there are indications that the carrying value may not be recoverable. Identifiable assets acquired and liabilities and contingent liabilities assumed in a business combination are measured initially at their fair values at the acquisition date. Acquisition-related costs are expensed as incurred and included in administrative expenses. Basis of consolidation These consolidated financial statements include the financial statements of Verona Pharma plc and its wholly owned subsidiary Verona Pharma, Inc., as well as the Verona Employee Benefit Trust ("EBT"). The EBT is accounted for under IFRS 10 and is consolidated on the basis that the Company has control, and the assets and liabilities of the EBT are included on the Company balance sheet and shares held by the EBT in the Company are presented as a deduction from equity. Inter-company transactions, balances and unrealized gains on transactions between group companies are eliminated. Verona Pharma, Inc. adopts the same accounting policies as the Company. 2.2 Foreign currency translation Items included in the Group's consolidated financial statements are measured using the currency of the primary economic environment in which the entity operates ("the functional currency"). The consolidated financial statements are presented in United States Dollar, which became the functional currency of the Company in the year ended December 31, 2020. Transactions in foreign currencies are recorded using the rate of exchange ruling at the date of the transaction. Monetary assets and liabilities denominated in foreign currencies are translated using the rate of exchange at the balance sheet date and the gains or losses on translation are included in the Consolidated Statement of Comprehensive Income. Non-monetary items that are measured in terms of historical cost in a foreign currency are translated using the exchange rates at the dates of the original transactions. Non-monetary items measured at fair value in a foreign currency are translated using the exchange rates at the date when the fair value was determined. 2.3 Cash and cash equivalents Cash and cash equivalents includes deposits held at call with banks with original maturities of three months or less that are readily convertible to known amounts of cash, and money market funds. Money market funds have been classified as cash and cash equivalents as they are low risk instruments, readily convertible to a known amount of cash and are subject to an insignificant risk of change in value. Management's intention is to manage these funds as cash and to use them to meet short-term cash requirements. 2.4 Deferred taxation Deferred tax is provided in full, using the liability method, on temporary differences arising between the tax bases of assets and liabilities and their carrying amounts in the consolidated financial statements. Deferred tax is determined using tax rates and laws that have been enacted or substantively enacted by the balance sheet date and expected to apply when the related deferred tax is realized or the deferred liability is settled. Deferred tax assets are recognized to the extent that it is probable that the future taxable profit will be available against which the temporary differences can be utilized. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 73 |
| 2.5 Research and development costs Capitalization of expenditure on product development commences from the point at which technical feasibility and commercial viability of the product can be demonstrated and the Group is satisfied that it is probable that future economic benefits will result from the product once completed. No such costs have been capitalized to date. Expenditure on research and development activities that do not meet the above criteria is charged to the Consolidated Statement of Comprehensive Income as incurred. 2.6 Property, plant and equipment Property, plant and equipment are stated at cost, net of depreciation and any provision for impairment. Depreciation is calculated to write off the cost less their estimated residual values, on a straight-line basis over the expected useful economic lives of the assets concerned. Computer hardware is depreciated over three years and office equipment over the term of the lease. 2.7 Intangible assets and goodwill (a) Goodwill Goodwill arises on the acquisition of subsidiaries and represents the excess of the consideration transferred over the fair value of the identifiable net assets acquired. (b) Patents Patent costs associated with the preparation, filing, and obtaining of patents are capitalized and amortized on a straight-line basis over the estimated useful lives of ten years. Amortization of patents is included in research and development costs. (c) Computer software Amortization is calculated so as to write off the cost less estimated residual values, on a straight-line basis over the expected useful economic life of two years. Amortization is included in selling, general and administrative costs. (d) In-process research & development ("IP R&D") The IP R&D asset, acquired through a business combination, which had not reached technical feasibility, was initially recognized at fair value. Subsequent movements in the assumed contingent liability (see note 2.13) that relate to changes in estimated cashflows or probabilities of success are recognized as additions to the IP R&D asset that it relates to. The asset is subject to impairment testing until completion, abandonment of the project or when the research findings are commercialized through a revenue generating project. 2.8 Impairment of intangible assets, goodwill and non-financial assets The Group holds intangible assets relating to acquired IP R&D, patent costs and goodwill. Goodwill and indefinite intangible assets are tested annually for impairment or if there is an indication of impairment. The Group is a single cash generating unit ("CGU") so all intangibles are allocated to the Group as one CGU. The Group initially compares the market capitalization of the Group to the book value of its assets. If the value of the market capitalization does not support the valuation of the assets, the Group reviews estimates of the cash flows over the remaining lives of its other intangible assets, or related group of assets where applicable, in measuring whether the assets to be held and used will be realizable. In the event of impairment, the Group would discount the future cash flows using its estimated weighted average cost of capital to estimate the amount of the impairment. As at 31 December 2023 and 2022 the Company carried out impairment reviews with reference to its market capitalization. The Group also monitors for any triggering events for finite-lived intangible assets and non-financial assets and would test for impairment if a triggering event was determined to have occurred. No impairment was identified for any of the assets in the years ended December 31, 2023 and 2022. 2.9 Equity interest As part of the Nuance Agreement, the Group received an equity interest in Nuance Biotech, the parent company of Nuance Pharma. The equity interest was recognized at fair value and is subsequently measured at fair value through profit and loss. Management applies judgement in determining the change in fair value. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 74 |
| 2.10 Employee Benefits (a) Pension The Group operates defined contribution pension plans for its employees. Contributions payable for the year are charged to the Consolidated Statement of Comprehensive Income. The Group has no further liability once the contributions have been paid. (b) Bonus plans The Company recognizes a liability and an expense for bonus plans if contractually obligated or if there is a past practice that has created a constructive obligation. 2.11 Share-based payments The Company operates a number of equity-settled, share-based compensation schemes. The fair value of share based payments is determined using the Black-Scholes model and requires several assumptions and estimates, disclosed in note 19. The fair value of share-based payments under these schemes, other than performance restricted stock units ("PRSUs"), is expensed on a straight-line basis, using the graded-vesting method, over the share based payments' vesting periods, based on the Company's estimate of shares that will eventually vest. The fair value of PRSUs, which are subject to certain performance and service conditions, will be recognized over the remaining service period using the graded-vesting method once the performance conditions are determined to be probable of occurring 2.12 Provisions Provisions are recognized when the Company has a present legal or constructive obligation as a result of past events, it is probable that an outflow of resources will be required to settle the liability, and the amount can be reliably estimated. Provisions are measured at the present value of the expenditures expected to be required to settle the liability using a pre-tax rate that reflects current market assessments of the time value of money and the risks specific to the liability. 2.13 Assumed contingent liability related to the business combination In 2006 the Company acquired Rhinopharma and assumed contingent liabilities owed to Vernalis Pharmaceuticals Limited, which was subsequently acquired by Ligand Pharmaceuticals, Inc. (“Ligand”). The Company refers to the assignment and license agreement as the Ligand Agreement. Ligand assigned to the Company all of its rights to certain patents and patent applications relating to ensifentrine and related compounds (the "Ligand Patents") and an exclusive, worldwide, royalty-bearing license under certain Ligand know-how to develop, manufacture and commercialize products (the "Licensed Products") developed using Ligand Patents, Ligand know-how and the physical stock of certain compounds. The assumed contingent liability comprises a milestone payment on obtaining the first approval of any regulatory authority for the commercialization of a Licensed Product, low single digit royalties based on the future sales performance of all Licensed Products and a portion equal to a mid-twenty percent of any consideration received from any sub-licensees for the Ligand Patents and for Ligand know-how. The liability was initially recognized at fair value and subsequently measured at amortized cost using the effective interest rate method. The assumed contingent liability is estimated as the expected value of the milestone payment and royalty payments, including royalties from the Nuance Agreement. This expected value is based on estimated future royalties payable, derived from sales forecasts, and an assessment of the probability of success using standard market probabilities for respiratory drug development. The risk-weighted value of the assumed contingent arrangement is discounted back to its net present value applying an effective interest rate of 12%. Royalties payable are based on the future sales performance so the amount payable is unlimited. Sales that may be achieved are difficult to predict and subject to estimate, which is inherently uncertain. The assumed contingent liability is re-measured for changes in estimated cash flows or when the probability of success changes. Remeasurements relating to changes in estimated cash flows and probabilities of success are recognized in the IP R&D asset it relates to (see note 2.7). The unwind of the discount is recognized in finance expense. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 75 |
| 2.14 Financial instruments — initial recognition and subsequent measurement The Group classifies a financial instrument, or its component parts, as a financial liability, a financial asset or an equity instrument in accordance with the substance of the contractual arrangement and the definitions of a financial liability, a financial asset and an equity instrument. A financial instrument is any contract that gives rise to a financial asset of one entity and a financial liability or equity instrument of another entity. (a) Financial assets, initial recognition and measurement and subsequent measurement All assets are initially recognized at fair value plus transaction costs. The Group's equity interest in Nuance Biotech is subsequently measured at fair value through profit or loss ("FVPTL") and fair value gains and losses are recognized in profit or loss. All other assets are subsequently measured.at amortized cost using the effective interest method. (b) Financial liabilities, initial recognition and measurement and subsequent measurement Financial liabilities are classified as measured at amortized cost or FVTPL. Financial liabilities are initially recognized at fair value and subsequently measured at amortized cost using the effective interest method. Interest expense and foreign exchange gains and losses are recognized in profit or loss. Any gain or loss on derecognition is also recognized in profit or loss. The Group's financial liabilities include debt, trade and other payables, the Company's warrants and the assumed contingent liability. Debt may be considered extinguished when it has been modified and the terms of the new debt instruments and old debt instruments are “substantially different” (as defined in the debt modification guidance in IFRS 9). Under the applicable guidance, when determining if debt instruments are substantially different, a 10% quantitative test comparing the discounted cash flows before and after the modification is required to be performed. Additionally, there is an accounting policy choice to perform only the 10% test or the 10% test as well as an additional qualitative analysis of any modification of terms when the change in discounted cash flows is less than 10%. The Group's policy with respect to the applicable guidance is to only perform the 10% quantitative test. 2.15 Transaction costs Qualifying transaction costs might be incurred in anticipation of an issuance of equity instruments and may cross reporting periods. The entity defers these costs on the balance sheet until the equity instrument is recognized. Deferred costs are subsequently reclassified as a deduction from equity when the equity instruments are recognized, as the costs are directly attributable to the equity transaction. If the equity instruments are not subsequently issued, the transaction costs are expensed. Any costs not directly attributable to the equity transaction are expensed. Transaction costs that relate to the issue of a compound financial instrument are allocated to the liability and equity components of the instrument in proportion to the allocation of proceeds. Where the liability component is held at fair value through profit or loss, the transaction costs are expensed to the Consolidated Statement of Comprehensive Income. For liabilities held at amortized cost, transaction costs are deducted from the liability and subsequently amortized. The amount of transaction costs accounted for as a deduction from equity is disclosed separately in accordance with International Accounting Standard. 2.16 Employee benefit trust In the year ended December 31, 2020, the Group incorporated a trust to facilitate the acquisition of shares, by or for the benefit of employees and former employees. The Group issued 16.0 million ordinary shares in the year ended December 31, 2023 to cover expected share awards to employees under the 2017 Incentive Plan. Management have determined that the Group has the indirect ability to control the trust as trustees are required to act in accordance with the trust deed that the Group drew up and because the Group controls the issuance of shares to cover awards. As a consequence the trust is included within the Company’s financial statements. The shares that were issued to the trust that have not been transferred to employees to cover share awards are included in the Consolidated Statement of Financial Position as treasury shares. 2.17 Investments in subsidiaries Investments in subsidiaries are shown at cost less any provision for impairment. 2.18 New standards, amendments and interpretations issued but not effective for the financial year beginning January 1, 2023 and not early adopted There are no IFRS standards or interpretations not yet effective that are expected to have a material impact on the Group. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 76 |
| 3. Financial Instruments 3.1 Financial Risk Factors The Group's activities expose it to a variety of financial risks: market risk (including currency risk), credit risk, and liquidity risk. The Group's overall risk management program is focused on preservation of capital and has sought to minimize potential adverse effects on the Group's financial performance and position. The Group's and the Company's exposure to risk are not materially different. (a) Market risk Foreign currency risk reflects the risk that the Group's net assets will be negatively impacted due to fluctuations in exchange rates. The Group has not entered into foreign exchange contracts to hedge against gains or losses from foreign exchange fluctuations. The summary data about the Group's exposure to currency risk is as follows. Figures are the U.S. Dollar values of balances in each currency: December 31, 2023 December 31, 2022 USD GBP EUR USD GBP EUR $'000s $'000s $'000s $'000s $'000s $'000s Cash and cash equivalents 253,302 18,456 14 201,886 25,914 27 Trade and other payables 6,753 2,090 415 15,868 4,533 1,101 Sensitivity analysis A reasonably possible strengthening or weakening of the euro or pound sterling against U.S. dollar as of December 31, 2023 and 2022 would have affected the measurement of the financial instruments denominated in a foreign currency (excluding the assumed contingent liability as the impact of this is immaterial). The following table shows how a movement in a currency would give rise to a profit or (loss) and a corresponding entry in equity. Profit or loss and equity Strengthening Weakening December 31, 2023 $'000s $'000s EUR (10% movement) (36) 36 GBP (10% Movement) 1,457 (1,457) Foreign currency denominated trade payables are short-term in nature (generally 30 to 45 days). The Group is also exposed to market risk on the value of the equity interest in Nuance Biotech. The fair value of the equity interest is dependent on the success of Nuance Biotech's various clinical programs, as well as valuations of similar companies in the Chinese market. The following table shows the effect of a 10% change in the fair value of the equity interest: Equity interest $'000s Fair value increase of 10% 16,500 Base case, reported fair value 15,000 Fair value decrease of 10% 13,500 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 77 |
| 3.1 Financial Risk Factors (continued) (b) Credit risk Financial instruments that potentially subject the Group to concentration of credit risk consist of principally cash and cash equivalents, bank deposits and certain receivables. The Group holds cash and cash equivalents with highly rated financial institutions and in highly rated money market funds and the Group has not experienced any significant credit losses in these financial statements and does not believe the Group is exposed to any significant credit risk on these instruments. As of December 31, 2023, the Group held funds at bank and in money market funds backed by U.K. or U.S. government debt. As of December 31, 2023, and December 31, 2022, cash and cash equivalents were placed at the following banks and money market funds: Cash and cash equivalents As of December 31, 2023 As of December 31, 2022 $'000 $'000 Government debt money market funds 269,114 224,181 Silicon Valley Bank 1,441 2,565 Lloyds Bank 401 1,081 HSBC Bank 592 — JPMorgan Chase & Co 224 — Total 271,772 227,827 (c) Management of capital The Group considers capital to be its equity reserves. At the current stage of the Group's life cycle, the Group's objective in managing its capital is to ensure funds raised meet the research and operating requirements until the next development stage of the Group's suite of projects. The Group ensures it is meeting its objectives by reviewing its Key Performance Indicators to ensure the research activities are progressing in line with expectations, costs are controlled and unused funds are placed in low risk money market funds to conserve resources. Additionally, the Group has a term loan outstanding which contains customary representations and warranties, covenants and events of default, including two financial covenants, as noted in note 23. As of December 31, 2023, neither of the financial covenants was applicable as the commencement date of those covenants had not yet occurred. The Group tracks and regularly monitors its compliance with covenants as a default of the covenants could result in any amounts outstanding at the date of default becoming due and payable ahead of the maturity date of the 2023 Term Loan. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 78 |
| 3.1 Financial Risk Factors (continued) (d) Liquidity risk The Group periodically prepares working capital forecasts for the foreseeable future, allowing an assessment of the cash requirements of the Group, to manage liquidity risk. The following table provides an analysis of the Group's financial liabilities. The carrying value of all balances approximates to their fair value, with the exception of the assumed contingent liability (see note 22). Less than 1 year Between 1 and 2 years Between 2 and 5 years Over 5 years $'000s $'000s $'000s $'000s At December 31, 2023 Trade payables 3,492 — — — Other payables 435 — — — Accruals 5,331 — — — Lease liability 1,180 555 1,220 — Term loan(1) 5,207 5,673 67,364 — Assumed contingent liability(2) 20,879 9,498 83,866 324,708 Total 36,524 15,726 152,450 324,708 (1) This is the undiscounted value of the loan plus undiscounted interest payments (2) This is the undiscounted value of the liability Less than 1 year Between 1 and 2 years Between 2 and 5 years Over 5 years $'000s $'000s $'000s $'000s At December 31, 2022 Trade payables 2,910 — — — Other payables 1,409 — — — Accruals 17,183 — — — Lease liability 675 196 9 — Term loan(1) 866 866 11,655 — Assumed contingent liability(2) — 18,167 39,649 256,020 Total 23,043 19,229 51,313 256,020 (1) This is the undiscounted value of the loan plus undiscounted interest payments (2) This is the undiscounted value of the liability VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 79 |
| 3.2 Fair value estimation Group and Company The carrying amounts of cash and cash equivalents, receivables, accounts payable and accrued liabilities approximate to fair value due to their short-term nature. The carrying amount of the assumed contingent liability is $215.4 million compared to the approximate fair value $204.5 million. The underlying assumptions are similar, the primarily driver of the difference relates to the discount rate. For financial instruments that are measured in the Consolidated Statement of Financial Position at fair value, IFRS 7 requires disclosure of fair value measurements by level of the following fair value measurement hierarchy: • Quoted prices (unadjusted) in active markets for identical assets or liabilities (level 1); • inputs other than quoted prices included within level 1 that are observable for the asset or liability, either directly or indirectly (level 2); and • inputs for the asset or liability that are not based on observable market data (level 3). For the year ended December 31, 2023, and 2022, fair value adjustments to financial instruments measured at fair value through profit and loss resulted in the recognition of no gain or loss in 2023 and 2022. The fair value of financial instruments that are not traded in an active market is determined by using valuation techniques. These valuation techniques maximize the use of observable market data where it is available and rely as little as possible on entity specific estimates. If all significant inputs required to ascertain the fair value of an instrument are observable, the instrument is included in level 2. If one or more of the significant inputs are not based on observable market data, the instrument is included in level 3. The derivative financial instrument is classified at level 3 in the fair value hierarchy. Movements in Level 3 items during the years ended December 31, 2023, and 2022 are as follows: Equity Interest $'000s At January 1, 2023 15,000 At December 31, 2023 15,000 Equity Interest $'000s At January 1, 2022 15,000 At December 31, 2022 15,000 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 80 |
| 3.3 Change in liabilities arising from financing activities Group The Group has provided a reconciliation so that changes in liabilities arising from financing activities, including both changes arising from cash flows and non-cash changes can be evaluated. Lease liability 2023 2022 $'000s $'000s At January 1 880 951 Capitalization of rental leases - non-cash 2,807 555 Payment of lease liability - cash (713) (628) Interest - non-cash 30 31 Foreign exchange differences - non-cash (49) (29) At December 31 2,955 880 Term Loan 2023 2022 $'000s $'000s At January 1 9,768 4,874 Modification of term loan 30,000 (5,035) Issue of term loan 9,996 10,000 Debt issuance costs (1,559) (245) Amortization of debt issuance costs (non-cash) 47 80 Accretion of final payment (non-cash) 122 94 At December 31 48,374 9,768 See note 23 for information relating to the Term Loan. Company Term Loan 2023 2022 $'000s $'000s At January 1 9,768 4,874 Issue of term loan 9,996 10,000 Amortization of debt issuance costs (non-cash) 47 80 Accretion of final payment (non-cash) 122 94 Extinguishment of term loan (19,933) — At December 31 — 9,768 See note 23 for information relating to the Term Loan. 4. Critical accounting estimates and judgments The preparation of financial statements in conformity with IFRS requires the use of accounting estimates and assumptions that affect the reported amounts of assets and liabilities at the date of the financial statements and the reported amounts of income and expenses during the reporting period. Although these estimates are based on management's best knowledge of current events and actions, actual results ultimately may differ from those estimates. IFRS also requires management to exercise its judgment in the process of applying the Group's accounting policies. The areas involving significant estimates and judgements are as follows: (a) Significant Estimate: Assumed contingent liability The Group has a material liability for the future payment of royalties and milestones associated with contractual liabilities on ensifentrine, acquired as part of the acquisition of Rhinopharma. The estimation of the amounts and timing of future cash flows requires the forecast of royalties payable and the estimation of the likelihood that the regulatory approval milestone will be achieved (see notes 2.13 and 22). The estimates for the assumed contingent liability are based on a discounted cash flow model. Key estimates included the calculation of deferred consideration include: VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 81 |
| 4. Critical accounting estimates and judgments (continued) • revenue; and ▪ probabilities of success. The revenue assumption includes, but is not limited to, timing of first commercial sale, revenue growth rates and net selling price, which includes estimated reductions for gross-to-net sales adjustments. When there is a change in the expected cash flows or probabilities of success, the assumed contingent liability is re-measured with the change in value recognized in the IP R&D asset it relates to. The assumed contingent liability is measured at amortized cost with the discount unwinding in finance expense throughout the year. Actual outcomes could differ significantly from the estimates made. A sensitivity analysis is provided in note 22. In August 2023, the U.S. Food and Drug Administration (“FDA”) accepted for review the Group's New Drug Application (“NDA”) seeking approval of ensifentrine for the maintenance treatment of chronic obstructive pulmonary disease (“COPD”) and assigned a Prescription Drug User Fee Act (“PDUFA”) target action date of June 26, 2024. As a consequence, the probability of success changed, reducing the risk-weighting adjustment applied to estimated cash flows. Additionally, in 2023 the Group carried out further market research and updated its forecasts for ensifentrine's revenue for the maintenance treatment of chronic obstructive pulmonary disorder using a nebulized formulation in the U.S. On June 9, 2021 Verona signed an agreement granting Nuance Pharma the exclusive rights to develop and commercialize products containing ensifentrine in Greater China (the "Nuance Agreement"). The assumed contingent liability was calculated using the same methodology as stated above. Management used judgment to determine that Nuance had also entered the Phase 3 stage of ensifentrine's clinical development plan. (b) Significant Estimate: Research and development costs Research and development (“R&D”) costs are expensed as incurred. Research and development expenses include salaries, share-based compensation and benefits of employees, and other costs related to the Group’s R&D activities, including contracts with clinical research organizations and contract manufacturers. As part of the process of preparing financial statements the Group is required to estimate its expenses resulting from its obligations under contracts with vendors and consultants and clinical site agreements in connection with its R&D efforts. The financial terms of these contracts are subject to negotiations which vary contract to contract and may result in payment flows that do not match the periods over which materials or services are provided to the Group under such contracts. The Group’s objective is to reflect the appropriate clinical trial expenses in its financial statements by matching those expenses with the period in which services and efforts are expended. The Group accounts for these expenses according to the progress of the trials and other development activities measured by patient progression and the timing of various aspects of the trial. The Group determines prepaid and accrual estimates through discussions with applicable personnel and outside service providers as to the progress of clinical trials, or other services completed. During the course of a clinical trial, the Group adjusts its rate of clinical trial expense recognition if actual results differ from its estimates. The Group makes estimates of its prepaid and accrued expenses as of each balance sheet date in its financial statements based on facts and circumstances known at that time. Although the Group does not expect its estimates to be materially different from amounts actually incurred, its understanding of the status and timing of services performed relative to the actual status and timing of services performed may vary and may result in the Group reporting amounts that are too high or too low for any particular period. The Group’s clinical trial prepaid and accrual expense is dependent upon the timely and accurate reporting of study recruitment from contract research organizations and activities carried out by other third-party vendors as well as the timely processing of any change orders from the contract research organizations. As of December 31, 2023 and 2022, accrued expenses related to clinical trial and other development costs was $0.7 million and $12.3 million. 5. Loss per ordinary share - basic and diluted (cents) Basic loss per ordinary share of 11.4 cents (2022: 14.2 cents) for the Group is calculated by dividing the loss for the year ended December 31, 2023 by the weighted average number of ordinary shares in issue of 634,142,660 as of December 31, 2023 (2022: 529,071,526). During the years ended December 31, 2023 and 2022, outstanding share options, RSUs and PRSUs of 54,922,392 and 53,818,840 respectively, were not included in the computation of diluted earnings per ordinary share, because to do so would be antidilutive. 6. Segmental reporting The Group’s activities are covered by one operating and reporting segment: Drug development. There have been no changes to management’s assessment of the operating and reporting segment of the Group during the year. All non-current assets are based in the United Kingdom apart from right-of-use assets relating to a property leases, and associated fixtures and fittings, in the United States. Total assets held by Verona Pharma, Inc. approximate $35.7 million. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 82 |
| 7. Operating costs Group Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Operating costs: Research and development costs: Employee benefits (note 8) 6,502 6,804 Share-based payment 4,994 5,420 Legal, professional, consulting and listing fees 2,555 1,992 Amortization of patents (note 12) 252 217 Other research and development expenses 3,110 35,906 Total research and development costs 17,413 50,339 Selling, general and administrative costs: Employee benefits (note 8) 13,910 7,747 Share-based payment 16,885 8,701 Legal, professional consulting and listing fees 5,040 4,540 Depreciation of property, plant and equipment 5 38 Depreciation of right of use assets 673 602 Operating lease charge — land and buildings (52) — Loss on variations in foreign exchange rate (226) 42 Other selling, general and administrative expenses 15,045 6,170 Total selling, general and administrative costs 51,280 27,840 The Group obtained the services from and paid the fees of the Group's auditors and their associates as detailed below: Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Audit of Verona Pharma plc and consolidated financial statements 659 420 Audit related services 557 201 Other services 97 403 Total 1,313 1,024 Audit-Related Services For the years ended December 31, 2023 and December 31, 2022, audit related services include fees for quarterly interim reviews and for the year ended December 31, 2023, also consisted of fees for the audit of internal control over financial reporting. Other Services For the year ended December 31, 2023 and December 31, 2022, other services related to capital markets-related services provided by PwC, and for the year ended December 31, 2023, also included technology fees. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 83 |
| 8. Directors' emoluments and staff costs Group Year ended December 31, 2023 Year ended December 31, 2022 The monthly average number of employees (excluding directors) of the Group during the year: Research and development 21 16 Selling, general and administrative 37 13 Total 58 29 Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Employee benefits expenses Wages and salaries 15,067 10,258 Social security costs 515 4,113 Share-based payment expense 11,094 14,121 Other pension costs 494 178 Total employee benefits expense 27,170 28,670 Directors' emoluments Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Aggregate emoluments of directors: Aggregate emoluments 1,784 1,541 Other pension costs 13 12 Directors' emoluments 1,797 1,553 Directors aggregate amounts receivable under long-term incentive schemes, made up of long-term RSU and PRSU grants was $5.3 million at December 31, 2023 (2022: $3.9 million). No share options were exercised by directors in the year ended December 31, 2023 and 2023. Executive officers compensation Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Aggregate executive officers costs: Wages and salaries 2,034 1,962 Share-based payment expense 5,517 5,018 Other pension costs 135 22 Total executive officers costs 7,686 7,002 Executive officers' aggregate amounts receivable under long-term incentive schemes, made up of long-term RSU and PRSU grants was $8.0 million at December 31, 2023 (2022: $5.6 million). No share options were exercised by executive officers in the year ended December 31, 2023 and 2022. The Group considers key management personnel to be the aggregate of directors and executive officers. The executive officers are the chief financial officer, chief medical officer and legal counsel. The Group operates defined contribution pension schemes for its employees and executive director. There were $321 thousand of accrued pension contributions to the scheme at December 31, 2023 (2022: $75 thousand). VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 84 |
| 8. Directors' emoluments and staff costs (continued) Company Year ended December 31, 2023 Year ended December 31, 2022 The average number of employees (excluding directors) of the Company during the year: Research and development 3 5 Selling, general and administrative 7 7 Total 10 12 Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Employee benefits expenses Wages and salaries 1,549 2,779 Social security costs (250) 2,841 Share-based payment expense 11,094 14,121 Other pension costs 52 70 Total employee benefits expense 12,445 19,811 Directors' emoluments Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Aggregate emoluments of directors: Wages and salaries 549 528 Directors' emoluments 549 528 Directors' aggregate amounts receivable under long-term incentive schemes, made up of long-term RSU and PRSU grants was $nil at December 31, 2023 (2022: $nil). No share options were exercised by directors in the year ended December 31, 2023 and 2022. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 85 |
| 8. Directors' emoluments and staff costs (continued) Executive officers' compensation Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Aggregate executive officers costs: Wages and salaries 503 456 Share-based payment expense 5,517 5,018 Other pension costs 122 10 Total executive officers costs 6,142 5,484 Executive officers' aggregate amounts receivable under long-term incentive schemes, made up of long-term RSU and PRSU grants was $1.3 million at December 31, 2023 (2022: $0.9 million). No share options were exercised by executive officers in the year ended December 31, 2023 and 2022. The Company considers key management personnel to be the aggregate of directors and executive officers. The executive officer employed by the Company is the Company's legal counsel. The Company operates a defined contribution pension scheme for its employees. There were $173 thousand of accrued pension contributions to the scheme at December 31, 2023 (2022: $73 thousand). In respect of Directors’ remuneration, the Company has taken advantage of the permission in Paragraph 6(2) of Statutory Instrument 2008/410 to omit aggregate information that is capable of being ascertained from the detailed disclosures in the audited sections of the Directors’ Remuneration Report on pages 36 to 57, which form part of these Consolidated Financial Statements. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 86 |
| 9. Finance income and expense Group Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Finance income: Interest received on cash balances 12,761 2,821 Foreign exchange gain on translating foreign currency denominated balances 1,866 — Total finance income 14,627 2,821 Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Finance expense: Extinguishment of debt — 815 Interest on term loan 2,057 521 Interest on discounted lease liability 30 31 Foreign exchange loss on translating foreign currency denominated balances — 3,817 Unwinding of discount factor related to the assumed contingent arrangement (note 22) 16,802 4,332 Total finance expense 18,889 9,516 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 87 |
| 10. Taxation Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Analysis of tax credit for the year: Current tax: U.K. tax credit (2,191) (9,634) U.S. tax charge 666 253 Adjustment in respect of prior periods 895 (10) Total tax credit (630) (9,391) The difference between the total tax shown above and the amount calculated by applying the standard rate of tax to the loss before tax is as follows: Factors affecting the tax credit for the year: Loss on ordinary activities before taxation (72,955) (84,762) Multiplied by standard rate of corporation tax of 23.5% (2022: 19%) (17,144) (16,105) Effects of: Non-deductible expenses 5,256 3,033 Research and development incentive 36 (4,145) Temporary differences not recognized (339) 1 Difference in overseas tax rates (13) 42 Share options exercised (2,926) (1,404) Tax losses carried forward not recognized 13,605 9,197 Adjustment in respect of prior periods 895 (10) Total tax credit (630) (9,391) U.K. corporation tax is charged at 23.5% (2022: 19%) and U.S. federal and state tax at 23.5% (2022: 27.6%). Factors that may affect future tax charges The Group has U.K. tax losses available for offset against future profits in the United Kingdom. However an additional deferred tax asset has not been recognized in respect of such items due to uncertainty of future profit streams. As of December 31, 2023, the unrecognized deferred tax asset at 25% is estimated to be $62.0 million (2022: $51.0 million at 25%). Unrecognized deferred tax assets related to tax losses and potential tax deductions on potential issuance of shares under employee share programs. These losses and deductions have an indefinite life. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 88 |
| 11. Goodwill Group and Company The goodwill balance of December 31, 2023 and 2022 was $0.5 million. Goodwill represents the excess of the purchase price over the fair value of the net assets acquired in connection with the acquisition of Rhinopharma in September 2006. Goodwill is not amortized, but is tested annually for impairment. The Group has one CGU so goodwill is tested for impairment together with its intangible assets. It was tested with reference to the Group's market capitalization as of December 31, 2023, the date of testing of IP R&D and goodwill impairment. The market capitalization of the Group was approximately $1.6 billion as of December 31, 2023, (2022: $2.0 billion) compared to the Group's net assets of $223.3 million (2022: $219.6 million). Consequently, no impairment was required. 12. Intangible assets Group and Company IP R&D Computer software Patents Total $'000s $'000s $'000s $'000s Cost At January 1, 2022 31,527 23 2,171 33,721 Additions — — 336 336 Disposal — (14) (75) (89) Re-measurement 97,833 — — 97,833 At December 31, 2022 129,360 9 2,432 131,801 Accumulated amortization At January 1, 2022 — 23 852 875 Charge for year — — 217 217 Disposals — (14) (75) (89) At December 31, 2022 — 9 994 1,003 Net book value At December 31, 2022 129,360 — 1,438 130,798 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 89 |
| 12. Intangible assets (continued) IP R&D Computer software Patents Total $'000s $'000s $'000s $'000s Cost At January 1, 2023 129,360 9 2,432 131,801 Additions — — 509 509 Disposal — (9) — (9) Re-measurement 60,115 — — 60,115 At December 31, 2023 189,475 — 2,941 192,416 Accumulated amortization At January 1, 2023 — 9 994 1,003 Charge for year — — 252 252 Disposals — (9) — (9) At December 31, 2023 — — 1,246 1,246 Net book value At December 31, 2023 189,475 — 1,695 191,170 Intangible assets comprise patents, computer software and an IP R&D asset that arose on the acquisition of Rhinopharma and investment in patents to protect ensifentrine. The IP R&D asset acquired through the business combination was initially recognized at fair value. Subsequent movements in the assumed contingent liability that relate to changes in estimated cash flows or probabilities of success are recognized as additions to the IP R&D asset that it relates to. The asset is not amortized and is tested annually for impairment. Patents are amortized over a period of ten years and are tested annually for impairment. Intangible assets are tested for impairment with goodwill, as the Group has only one cash generating unit. See note 11 for information about the impairment review. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 90 |
| 13. Right-of-use assets - property leases Group The right-of-use asset relates to rented office space in London, North Carolina and Georgia. The Consolidated Statement of Financial Position shows the following amounts relating to leases: Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Right-of-use assets Right-of-use assets 2,847 854 2,847 854 Lease liabilities Current (1,180) (675) Non-current (1,775) (205) (2,955) (880) In the year ended December 31, 2023, the Group extended its leases on office space in London and entered into an additional lease in North Carolina. $2.8 million was added to the lease liability and the associated right of use asset. In the year ended December 31, 2022, the Group extended its leases on office space in London. $0.6 million was added to the lease liability and the associated right of use asset. To calculate lease liabilities the Company used a weighted average discount rate of 11% and 4% for the years ended December 31, 2023 and December 31, 2022, respectively. The current leases end in 2025, 2025, 2024 and 2027 for the office space in London, Georgia and two in North Carolina respectively and include options to extend. The Group has determined it is not yet reasonably certain to operate the option to extend the leases and so has recognized lease payments only to these points in its calculation of the lease liabilities. The right-of-use lease assets are depreciated over the term of the leases. The Consolidated Statement of Comprehensive Income includes the following amounts relating to leases: Year ended December 31, 2023 Year ended December 31, 2022 $'000s $'000s Depreciation charge of right-of-use assets Right-of-use assets (673) (602) (673) (602) Interest expense (including finance cost) 30 31 The total cash outflow for leases in 2023 was $0.7 million (2022: $0.6 million). 14. Equity interest Group and Company As part of the Nuance Agreement, the Company received an equity interest in Nuance Biotech, the parent company of Nuance Pharma. The equity interest is held at fair value through profit and loss. In the year ended December 31, 2023 Nuance Biotech were not involved in any new transactions involving issuance of shares. As of December 31, 2023, there had been no transactions to indicate any change in the value of Nuance Biotech's stock, nor had there been any other indications of change in value. The equity interest is therefore recorded at a value of $15.0 million as of December 31, 2023. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 91 |
| 15. Investment in subsidiaries Company The Company has one wholly-owned subsidiary, Verona Pharma, Inc. Rhinopharma Limited, a Canadian company that was previously a non-operating, wholly-owned subsidiary, was dissolved in June 2021. Rhinopharma Limited was a drug discovery and development company focused on developing proprietary drugs to treat allergic rhinitis and other respiratory diseases prior to its acquisition by the Company on September 18, 2006. The Company’s investments comprise interests in Group undertakings, details of which are shown below: Verona Pharma, Inc. Country of incorporation Delaware USA Description of shares held $0.001 Common stock Proportion of shares held by the Company 100% Verona Pharma, Inc. was incorporated on the 12 December 2014 under the laws of the State of Delaware, USA and has its registered office at 2711 Centerville Road, Suite 400, City of Wilmington 19808, County of New Castle, Delaware, United States of America. 16. Prepayments and other receivables Group December 31, 2023 December 31, 2022 $'000s $'000s Prepayments 4,617 3,164 Other receivables 2,365 2,723 Total prepayments and other receivables 6,982 5,887 The prepayments balance includes prepayments for insurance and clinical activities. Company December 31, 2023 December 31, 2022 $'000s $'000s Prepayments 4,277 3,198 Other receivables 1,572 2,655 Total prepayments and other receivables 5,849 5,853 The prepayments balance includes prepayments for insurance and clinical activities. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 92 |
| 17. Share Capital The movements in the Company's share capital are summarized below: Date Description Number of shares Share Capital amounts in $'000s As at January 1, 2022 489,177,550 31,855 January Issuance of shares 4,880,696 328 August Issuance of shares 114,080,000 6,918 November Issuance of shares 23,200,000 1,425 As at December 31, 2022 631,338,246 40,526 January Issuance of shares 5,644,296 342 February Issuance of shares 10,888,752 658 March Issuance of shares 3,788,336 227 December Issuance of shares 16,000,000 1,018 As at December 31, 2023 667,659,630 42,771 All 667,659,630 issued ordinary shares at December 31, 2023 are allotted, unrestricted, called up and fully paid. All issued shares rank pari passu except for 48,088,896 non-voting ordinary shares. All shares have a par value of £0.05. At the Annual General Meeting held on April 27, 2023, shareholders approved the resolution to authorize the directors to allot shares in the Company, or grant rights to subscribe for, or to convert any security into shares in the Company, up to an aggregate nominal amount of £8,145,745, or 162,914,900 ordinary shares. As at December 31, 2023, £800,000 of this nominal amount, or 16,000,000 ordinary shares, had been issued. Treasury shares The Group and Company holds shares in an employee benefit trust, to satisfy share based compensation awards and these share are accounted for as treasury shares. As at December 31, 2023, 24,123,536 shares were held in treasure, at a nominal value of $1.5 million (2022: 25,037,192 shares, nominal value $1.5 million). 18. Equity issuances During the year ended December 31, 2023, Verona Pharma sold 20,321,384 ordinary shares (2022: 80,696 ordinary shares) represented by 2,540,173 ADSs (2022: 10,087 ADSs) under the 2021 ATM Program, at an average price of approximately $2.88 per share (2022: $0.86 per share) equivalent to $23.08 per ADS (2022: $6.86 per ADS), raising aggregate net proceeds of $56.9 million (2022: $0.1 million) after deducting issuance costs. In August 2022, Verona Pharma raised approximately $150 million in a public offering. The public offering comprised of 114,080,000 newly issued voting ordinary shares, represented by 14,260,000 new ADSs at a price of $10.50 per ADS. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 93 |
| 19. Share-based payments charge Group and Company The Company operates various share based payment incentive schemes for its staff. In accordance with IFRS 2 "Share Based Payments," the cost of equity-settled transactions is measured by reference to their fair value at the date at which they are granted. For transactions with employees fair value is determined using the Black-Scholes model. The cost of equity-settled transactions is recognized over the period until the award vests. No expense is recognized for awards that do not ultimately vest. At each reporting date, the cumulative expense recognized for equity-based transactions reflects the extent to which the vesting period has expired and the number of awards that, in the opinion of the Directors at that date, will ultimately vest. The costs of equity-settled share-based payments to employees are recognized in the Statement of Comprehensive Income, together with a corresponding increase in equity during the vesting period. During the twelve months ended December 31, 2023, the Company recognized a share-based payment expense of $21.9 million (2022: $14.1 million). The charge is included in selling, general and administrative costs as well as in research and development costs and represents the current year's allocation of the share based payment expense. The Company operates an Unapproved Share Option Scheme under which options were issued before 31 December 2016. The Company also operates a tax efficient EMI Option Scheme under which options were issued before 31 December 2016. In 2017 the Company commenced the 2017 Incentive Award Plan under which the Company grants share options, restricted stock units ("RSUs") and performance restricted stock units ("PRSUs") to employees and directors. All options and RSUs vest over terms of between one and four years. In the year ended December 31, 2019, the Company modified the terms of all the RSUs issued prior to January 1, 2019, to include a market condition that the Company's share price must be maintained above of £2 per ordinary share for thirty days, in addition to the service condition. As at December 31, 2022, this approximated to $21.90 per ADS. The RSUs vest after a five year term irrespective of whether the £2 market condition was met. This modification did not result in an increase in the fair value of the RSUs. The RSUs issued in the year ended December 31, 2019, also include the same market condition and five year term. In the year ended December 31, 2023, the market condition was met and all of the RSUs vested. In the year ended December 31, 2023, under the 2017 Incentive Award Plan, the Company granted 7,376,000 (2022: 9,024,000) share options, 3,596,872 RSUs (2022: 12,877,864) and 10,790,144 PRSUs (2022: nil). The total fair values of all outstanding options, RSUs and PRSUs were estimated using the Black-Scholes option-pricing model for equity-settled transactions and amounted to $66.3 million (2022: $49.1 million). The cost is amortized over the vesting period of the options and RSUs on a straight-line basis, using the graded-vesting method. The following assumptions were used for the Black-Scholes valuation of share options and RSUs granted in 2022 and 2023: Issued in 2022 Options RSUs PRSUs Number granted 9,024,000 12,877,864 Risk-free interest rate 2.09% - 4.20% Expected life of options 5 - 7 years Annualized volatility 82.50% - 84.27% Dividend rate 0.00 % Vesting period 0 - 4 years 0 - 4 years Issued in 2023 Options RSUs PRSUs Number granted 7,376,000 3,596,872 10,790,144 Risk-free interest rate 3.40% - 4.69% Expected life of options 5 - 7 years Annualized volatility 80.64% - 87.26% Dividend rate 0.00 % Vesting period 0 - 4 years 0 - 4 years 0 - 3 years VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 94 |
| 19. Share-based payments charge (continued) The Company had the following share options movements in the year ended December 31, 2023: Year of issue Exercise price ($) At January 1, 2023 Options granted Options exercised Options forfeited Options expired At December 31, 2023 Expiry date 2013 3.06 80,000 — — — (80,000) — April 15, 2023 2013 3.07 160,000 — — — (160,000) — July 29, 2023 2014 2.94 160,000 — — — — 160,000 May 15, 2024 2015 1.88 342,000 — (300,000) — — 42,000 January 29, 2025 2016 2.90 122,000 — — — — 122,000 February 9, 2026 2016 2.40 610,000 — — — — 610,000 August 3, 2026 2016 2.49 200,000 — — — — 200,000 September 13, 2026 2016 2.65 300,000 — — — — 300,000 September 26, 2026 2017 1.70 3,330,520 — (683,024) — — 2,647,496 April 26, 2027 2018 2.02 955,864 — (43,728) — — 912,136 March 8, 2028 2019 0.75 1,433,800 — — — — 1,433,800 March 29, 2029 2019 0.76 226,000 — — — — 226,000 June 11, 2029 2019 0.56 100,000 — — — — 100,000 August 22, 2029 2020 0.71 1,094,712 — (23,440) (4,680) — 1,066,592 March 3, 2030 2020 0.79 305,600 — — — — 305,600 September 24, 2030 2021 0.62 320,000 — (40,000) (160,000) — 120,000 October 4, 2031 2021 0.78 512,000 — — — — 512,000 August 8, 2031 2022 0.60 288,000 — — — — 288,000 March 13, 2032 2022 0.64 320,000 — (80,000) (240,000) — — March 29, 2032 2022 0.50 1,600,000 — — — — 1,600,000 April 27, 2032 2022 0.57 160,000 — — — — 160,000 May 31, 2032 2022 0.54 2,800,000 — (88,000) — — 2,712,000 July 4, 2032 2022 1.30 1,000,000 — — — — 1,000,000 September 7, 2032 2022 1.27 600,000 — — — — 600,000 September 19, 2032 2022 1.19 120,000 — — — — 120,000 September 28, 2032 2022 1.28 856,000 — — — — 856,000 October 2, 2032 2022 1.45 600,000 — — — — 600,000 October 26, 2032 2022 1.61 40,000 — — — — 40,000 October 30, 2032 2022 1.74 400,000 — — — — 400,000 December 4, 2032 2022 1.64 240,000 — — — — 240,000 December 18, 2032 2023 3.27 — 320,000 — — — 320,000 January 2, 2033 2023 2.76 — 680,000 — — — 680,000 January 31, 2033 2023 2.68 — 320,000 — — — 320,000 February 28, 2033 2023 2.51 — 880,000 — — — 880,000 April 2, 2033 2023 2.67 — 864,000 — — — 864,000 April 27, 2033 2023 2.62 — 840,000 — (60,000) — 780,000 April 30, 2033 2023 2.69 — 240,000 — — — 240,000 May 31, 2033 2023 2.64 — 616,000 — — — 616,000 June 30, 2033 2023 2.76 — 480,000 — — — 480,000 July 31, 2033 2023 2.43 — 304,000 — — — 304,000 August 31, 2033 2023 2.04 — 416,000 — — — 416,000 September 30, 2033 2023 1.74 — 840,000 — — — 840,000 October 31, 2033 2023 1.69 — 576,000 — — — 576,000 November 30, 2033 Total 19,276,496 7,376,000 (1,258,192) (464,680) (240,000) 24,689,624 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 95 |
| 19. Share-based payments charge (continued) The Company had the following RSU movements in the year ended December 31, 2023: Year of issue At January 1, 2023 Units granted Units vested Units forfeited At December 31, 2023 Expiry date 2017 182,680 — (182,680) — — April 26, 2027 2018 47,264 — (47,264) — — March 8, 2028 2019 283,720 — (283,720) — — March 29, 2029 2020 2,304,008 — (1,843,216) — 460,792 May 7, 2030 2020 17,657,984 — (12,205,760) (133,656) 5,318,568 August 20, 2030 2021 1,560,000 — (520,000) — 1,040,000 November 14, 2031 2021 40,000 — (17,512) — 22,488 December 13, 2031 2022 117,048 — (117,048) — — January 31, 2032 2022 57,640 — (57,640) — — September 7, 2032 2022 12,292,000 — (3,058,104) (149,992) 9,083,904 September 25, 2032 2023 — 3,596,872 — (20,000) 3,576,872 October 19, 2033 Total 34,542,344 3,596,872 (18,332,944) (303,648) 19,502,624 Outstanding and exercisable share options by scheme as of December 31, 2023: Plan Share options outstanding Share options exercisable Weighted average exercise price in $ for Outstanding Weighted average exercise price in $ for Exercisable 2017 Incentive Award Plan 23,255,624 11,470,640 1.50 1.14 EMI 114,000 114,000 2.54 2.54 Unapproved 1,320,000 1,320,000 2.55 2.55 Total 24,689,624 12,904,640 1.56 1.30 The options outstanding at December 31, 2023, had a weighted average remaining contractual life of 7.4 years (2022: 7.2 years). For 2022 and 2023, the number of options granted and expired and the weighted average exercise price of options were as follows: Number of options Weighted average exercise price ($) At January 1, 2022 12,695,200 1.38 Options granted in 2022: Employees 7,136,000 0.98 Directors 1,888,000 0.52 Options exercised in the year (1,822,688) 0.75 Options forfeited (620,016) 1.04 At December 31, 2022 19,276,496 1.22 Exercisable at December 31, 2022 10,382,256 1.48 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 96 |
| 19. Share-based payments charge (continued) Number of options Weighted average exercise price ($) At January 1, 2023 19,276,496 1.22 Options granted in 2023: Employees 6,368,000 2.43 Directors 1,008,000 2.64 Options exercised (1,258,192) 1.55 Options forfeited (464,680) 0.89 Options expired (240,000) 3.07 At December 31, 2023 24,689,624 1.56 Exercisable at December 31, 2023 12,904,640 1.30 The weighted average share price at the date of exercise of options exercised during the year ended 31 December 2023 was $3.03 (2021: $2.08). The following table shows the number of RSUs issued, vested and forfeited in 2022. Number of RSUs At January 1, 2022 38,347,352 Granted: Employees 8,943,416 Directors 3,934,448 RSUs vested in the year (15,676,608) RSUs forfeited in the year (1,006,264) At December 31, 2022 34,542,344 The following table shows the number of RSUs issued, vested and forfeited in 2023. Number of RSUs At January 1, 2023 34,542,344 Granted: Employees 2,796,872 Directors 800,000 RSUs vested in the year (18,332,944) RSUs forfeited in the year (303,648) At December 31, 2023 19,502,624 The following table shows the number of PRSUs issued, vested and forfeited in 2023. Number of PRSUs At January 1, 2023 — Granted: Employees 8,390,144 Directors 2,400,000 PRSUs forfeited in the year (60,000) At December 31, 2023 10,730,144 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 97 |
| 20. Derivative financial liability Group and Company On May 2, 2022 all remaining warrants expired. No warrants were exercised or forfeited in the year ended 2022. On July 29, 2016, the Company issued 31,115,926 units to new and existing investors at the placing price of £1.4365 per unit. Each unit comprises one ordinary share and one warrant. The warrant holders could subscribe for 0.4 of an ordinary share at a per share exercise price of £1.7238. The warrant holders could opt for a cashless exercise of their warrants, whereby the warrant holders can choose to exchange the warrants held for reduced number of warrants exercisable at nil consideration. The reduced number of warrants is calculated based on a formula considering the share price and the exercise price of the warrants. The warrants were therefore classified as a derivative financial liability, since their exercise could result in a variable number of shares to be issued. The warrants entitled the investors to subscribe in aggregate, a maximum of 12,401,262 shares. 21. Trade and other payables Group As of December 31, 2023 As of December 31, 2022 $'000s $'000s Trade payables 3,492 2,910 Other payables 435 1,409 Accruals 5,331 17,183 Total trade and other payables 9,258 21,502 Company As of December 31, 2023 As of December 31, 2022 $'000s $'000s Trade payables 1,560 2,909 Other payables 133 1,063 Amount due to group undertakings 37,507 11,319 Accruals 3,514 16,157 Total trade and other payables 42,714 31,448 Amounts due to group undertakings are unsecured, interest free and repayable on demand. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 98 |
| 22. Assumed contingent liability related to the business combination The value of the assumed contingent liability as of December 31, 2023 is $215.4 million (2022: $138.3 million). The increase in value of the assumed contingent liability during 2023 amounted to $77.1 million (2022: $101.8 million). In August 2023, the U.S. Food and Drug Administration (“FDA”) accepted for review the Group's New Drug Application (“NDA”) seeking approval of ensifentrine for the maintenance treatment of chronic obstructive pulmonary disease (“COPD”) and assigned a Prescription Drug User Fee Act (“PDUFA”) target action date of June 26, 2024. As a consequence, the probability of success changed, reducing the risk-weighting adjustment applied to estimated cash flows. Additionally, in 2023 the Group carried out further market research and updated its forecasts for ensifentrine's revenue for the maintenance treatment of chronic obstructive pulmonary disorder using a nebulized formulation in the U.S. On June 9, 2021 Verona signed an agreement granting Nuance Pharma the exclusive rights to develop and commercialize products containing ensifentrine in Greater China (the "Nuance Agreement"). The assumed contingent liability was calculated using the same methodology as stated above. As at December 31, 2023 Nuance are still conducting their phase 3 trial, resulting to no change in the probability of success from the previous year. The expected cash flows are based on estimated future royalties payable, derived from sales forecasts, including expected timings of these sales, and an assessment of the probability of success using standard market probabilities for respiratory drug development. The risk-weighted value of the assumed contingent arrangement is discounted back to its net present value applying an effective interest rate of 12%. 2023 2022 $'000s $'000s January 1 138,258 36,490 Re-measurement of contingent obligation 60,115 97,833 Foreign exchange differences recognised in loss for the period 229 (397) Unwinding of discount factor 16,802 4,332 December 31 215,404 138,258 The fair value of the contingent obligation is approximately $204.5 million. This is calculated using a discount rate of 13%. Because of the unobservable inputs in the model, the fair value is classified under Level 3 of the fair value hierarchy. For the amount recognized as at December 31, 2023, of $215.4 million, the effect if underlying assumptions were to deviate up or down is presented in the following table (assuming the probability of success does not change): Revenue (up / down 10 % pts) Probability of success (up / down 4 % pt) $'000s $'000s Variable up 234,812 224,020 Base case 215,404 215,404 Variable down 195,995 206,787 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 99 |
| 23. Term loan In November 2020, the Group entered into a term loan facility of up to $30.0 million (the “SVB Term Loan”), consisting of advances of $5.0 million funded at closing and $10.0 million and $15.0 million contingent upon achievement of certain clinical development milestones and other specified conditions. On October 14, 2022 (the “2022 Effective Date”), the Group entered into a loan and security agreement with Oxford Finance Luxembourg S.À R.L. for an aggregate amount of up to $150.0 million (the “Oxford Term Loan”). The Oxford Term Loan provided for an initial term loan advance in an aggregate amount of $10.0 million funded on the 2022 Effective Date (the “Oxford Term A Loan”), and up to four additional term loan advances in an aggregate amount of $140.0 million, contingent upon the achievement of certain clinical and regulatory development milestones as well as other specified conditions. The proceeds from the Oxford Term Loan were used for general corporate and working capital purposes, and a portion of the proceeds of the Oxford Term A Loan were used to repay in full the existing outstanding indebtedness owed to SVB. On March 24, 2023, the Group received $10.0 million under the second term loan advance (the “Oxford Term B Loan”). On December 27, 2023 (the “2023 Effective Date”), Verona Pharma, Inc. (the "Borrower") entered into a term loan facility of up to $400.0 million (the “2023 Term Loan” or “Loan Agreement”), consisting of a term loan advance in an aggregate amount of $50.0 million funded on the 2023 Effective Date (the “Term A Loan”) and four additional term loan advances subject to certain terms and conditions, as discussed below, in the amounts of $100.0 million (the “Term B Loan”), $75.0 million (the “Term C Loan”), $75.0 million (the “Term D Loan”) and $100.0 million (the “Term E Loan”). The 2023 Term Loan was entered into with Oxford Finance LLC, a Delaware limited liability company (“Oxford”), as collateral agent, and certain funds managed by Oxford and Hercules Capital, Inc. party thereto (collectively, the “Lenders”). The proceeds of the 2023 Term Loan will be used for general corporate and working capital purposes, and a portion of the proceeds of the Term A Loan was used by the Group on the 2023 Effective Date to repay in full the existing outstanding indebtedness owed by the Group under the Oxford Term Loan. The Group received net proceeds from the Term A Loan of $28.4 million which primarily consisted of the Term A Loan proceeds of $50.0 million partially offset by the repayment, in full, of the existing outstanding indebtedness owed by the Company under the Oxford Term Loan of $20.0 million, lender and third-party fees related to the Loan Agreement of $1.4 million and interest amounts of $0.2 million. Based upon the Group's accounting evaluation of the Loan Agreement, as well as the Oxford entities involved and terms of both the 2023 Term Loan and the Oxford Term Loan, the Group has applied modification accounting to the portion of the Term A Loan associated with Oxford. No gain or loss has been recorded upon the modification with the unamortized debt issuance costs at the 2023 Effective Date from the Oxford Term Loans included as amounts the outstanding amount under the 2023 Term Loan. The portion of the Term A Loan associated with Hercules Capital, Inc. has been accounted for as the issuance of new debt. As the Loan Agreement was entered into by Verona Pharma, Inc. and settled the prior term debt outstanding under the Oxford Term Loan, extinguishment accounting was applied to the prior term debt held by the Company. This resulted in a loss on extinguishment of $0.9 million was recognized. As such, with respect to the Company financial statements, no cash flow impacts are recorded as the debt previously outstanding under the Oxford Term Loan was effectively settled by Verona Pharma, Inc. as part of the Loan Agreement. The Term B Loan will be available, subject to customary terms and conditions, during the period commencing on the date the Group receives approval from the United States Food and Drug Administration for its New Drug Application for ensifentrine through and including the earliest of (i) the date that is 30 days immediately following the date the Group receives such approval and (ii) September 15, 2024. The Term C Loan will be available, subject to customary terms and conditions (including the prior borrowing of the Term B Loan), during the period commencing on the later of (i) September 15, 2025 and (ii) prior to September 30, 2025, the achievement by the Group of a specified net sales milestone. The Term D Loan will be available, subject to customary terms and conditions (including the prior borrowing of the Term C Loan), during the period commencing on the later of (i) February 15, 2026 and (ii) prior to March 31, 2026, the achievement by the Group of a specified net sales milestone. The Term E Loan will be available, subject to customary terms and conditions (including the prior borrowing of the Term D Loan) prior to June 1, 2028 at the Lenders sole discretion and upon the Group’s request. The 2023 Term Loan will mature on December 1, 2028. Each advance under the Loan Agreement accrues interest at a floating per annum rate (the “Basic Rate”) equal to (a) the greater of (i) the 1-Month CME Term SOFR (as defined in the Loan Agreement) reference rate on the last business day of the month that immediately precedes the month in which the interest will accrue and (ii) 5.34%, plus (b) 5.85%. Notwithstanding the foregoing, (i) in no event shall the Basic Rate (x) for the Term A Loan be less than 11.19% and (y) for each other 2023 Term Loan be less than the Basic Rate on the business day immediately prior to the funding date of such 2023 Term Loan, (ii) the Basic Rate for the Term A Loan for the period from the Effective Date through and including December 31, 2023 was 11.19% and (iii) the Basic Rate for each 2023 Term Loan shall not increase by more than 2.00% above the applicable Basic Rate as of the funding date of each such 2023 Term Loan. The 2023 Term Loan provides for interest-only payments on a monthly basis until the payment date immediately VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 100 |
| 23. Term loan (continued) preceding June 1, 2028. Thereafter, amortization payments will be payable monthly in equal installments of principal plus monthly payments of accrued interest. Upon repayment (whether at maturity, upon acceleration or by prepayment or otherwise), the Borrower shall make a final payment to the Lenders in the amount of 2.50% to 3.50% of the aggregate 2023 Term Loans advanced, depending on when a 2023 Term Loan is repaid (the “Final Payment”). The Borrower may prepay the 2023 Term Loan in full or in part provided that the Borrower (i) provides ten (10) days’ prior written notice to Oxford and the Lenders, (ii) pays on the date of such prepayment (A) all outstanding principal plus accrued and unpaid interest, (B) a prepayment fee of 2.00% of the 2023 Term Loans advanced if paid on or before December 27, 2025; 1.50% of the 2023 Term Loans advanced if paid after December 27, 2025 and before December 27, 2026; 1.00% of the 2023 Term Loans advanced if paid after December 27, 2026, (C) the Final Payment and (D) all other sums, if any, that shall become due and payable under the Loan Agreement, including interest at the default rate with respect to any past due amounts. Amounts outstanding during an event of default are payable upon the Required Lenders’ (as defined in the Loan Agreement) demand and shall accrue interest at an additional rate of 5.00% per annum and (iii) any partial prepayment of the 2023 Term Loans shall be in a denomination that is a whole number multiple of $5.0 million. The 2023 Term Loan is secured by a lien on substantially all of the assets of the Group, other than intellectual property, provided that a lien on intellectual property will be granted on the earlier of (i) the funding date of any 2023 Term Loan that would cause the aggregate principal amount of outstanding 2023 Term Loans drawn pursuant to the Loan Agreement to exceed $50.0 million and (ii) prior to the Borrower or the Group entering into a Permitted Royalty Financing (as defined in the Loan Agreement). The Group has also granted Oxford and the Lenders a negative pledge with respect to its intellectual property. The Loan Agreement contains customary representations and warranties, covenants and events of default, including two financial covenants: (i) commencing on July 1, 2025, the Borrower is required to maintain certain levels of cash in the United States subject to control agreements in favor of Oxford; provided that such liquidity covenant shall not apply at any given time if the market capitalization of the Group at such time is at least $3.0 billion and (ii) commencing on September 30, 2025, the Borrower and the Group are required to maintain quarterly trailing six-month net product revenue from the sale of ensifentrine; provided that such revenue covenant will be waived at any time (x) the Group's unrestricted cash balance on the last calendar day of each month during such quarter is equal to or greater than the product of 1.25 multiplied by the aggregate principal amount of outstanding 2023 Term Loans on such date, (y)(1) the Group’s unrestricted cash balance on the last calendar day of each month during such quarter is equal to or greater than the product of 0.5 multiplied by the aggregate principal amount of outstanding 2023 Term Loans on such date and (2) the average of the daily VWAP of the Company’s American Depositary Shares for each of the five trading days preceding the last trading day of each month during such quarter multiplied by the total number of issued and outstanding American Depositary Shares of the Group is at least $1.5 billion, or (z) the average of the daily VWAP of the Company’s American Depositary Shares for each of the five trading days preceding the last trading day of each month during such quarter multiplied by the total number of issued and outstanding American Depositary Shares of the Group is at least $3.0 billion. The Loan Agreement also contains other customary provisions, such as expense reimbursement, as well as indemnification rights for the benefit of Oxford and the Lenders. As of December 31, 2023 the interest rate was approximately 11% per annum and there was no material difference between the carrying value and the estimated fair value of the 2023 Term Loan. Future principal payments, which exclude the end of term charge, in connection with the 2023 Term Loan as of December 31, 2023 are as follows (in thousands): 2024 — 2025 — 2026 — 2027 — 2028 50,000 Total 50,000 VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 101 |
| 24. Related parties transactions and other shareholder matters (i) Related party transactions The Directors have authority and responsibility for planning, directing and controlling the activities of the Company and they therefore comprise key management personnel as defined by IAS 24, ("Related Party Disclosures"). Directors and key management personnel remuneration is disclosed in note8. (ii) Other shareholder matters Year ended December 31, 2023 During the year ended December 31, 2023, Dr. Zaccardelli and Mr. Hahn were granted 100,000 ADS RSUs and 300,000 ADS PRSUs each. During the year ended December 31, 2023, each member of the board of directors was awarded share options. Ms. Ackermann was awarded 18,000 ADS share options. Dr. Ebsworth, Dr. Cunningham, Dr. Edwards, Dr. Shah, Mr. Sinha, Dr. Ullman, Mr. Gupta, Ms. Deschamps and Mr. Brady were each awarded 12,000 ADS share options. Year ended December 31, 2022 During the year ended December 31, 2022, 41,806 and 16,722 ADS RSUs that were issued to Dr. Zaccardelli and Mr. Hahn, respectively, vested. These shares were paid in lieu of salary and were issued on February 1, 2022. During the year ended December 31, 2022, Dr. Zaccardelli and Mr. Hahn were granted an additional 450,000 ADS RSUs each. During the year ended December 31, 2022, each member of the board of directors was awarded share options. Mr. Brady was awarded 36,000 ADS share options. Dr. Ebsworth, Dr. Cunningham, Dr. Edwards, Dr. Shah, Mr. Sinha, Dr. Ullman, Mr. Gupta, and Ms. Deschamps were each awarded 25,000 ADS share options. VERONA PHARMA PLC NOTES TO THE CONSOLIDATED AND COMPANY FINANCIAL STATEMENTS FOR THE YEAR ENDED DECEMBER 31, 2023 102 |
FORM OF PROXY
| | I/We | | | |
| | of | | | |
| | being a member of the Company, hereby appoint | | | |
| | | | | | | Ordinary Resolutions | | | | FOR | | | | AGAINST | | | | WITHHELD | | | | DISCRETION | | | | | | |
| | | Resolution 1 | | | | To re-elect Lisa Deschamps as a director of the Company. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 2 | | | | To re-elect Martin Edwards as a director of the Company. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 3 | | | | To re-elect Sven Anders Ullman as a director of the Company. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 4 | | | | To re-elect Christina Ackermann as a director of the Company. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 5 | | | | To re-elect Michael Austwick as a director of the Company. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 6 | | | | To receive and adopt the U.K. statutory annual accounts and Directors’ report for the year ended 31 December 2023. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 7 | | | | To receive and approve, as a non-binding advisory resolution, the U.K. statutory Directors’ Remuneration Report for the year ended 31 December 2023. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 8 | | | | To receive and approve the U.K. Directors’ Remuneration Policy. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 9 | | | | To approve an increase in the maximum aggregate level of Non-Executive Director annual remuneration (by way of fee) in accordance with Article 21.4 of the Company’s Articles of Association to £750,000 per annum on an ongoing basis. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 10 | | | | To appoint Ernst & Young LLP as the Company’s auditors, to hold office until the conclusion of the next annual general meeting of shareholders. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 11 | | | | To authorize the Audit and Risk Committee to determine the auditors’ remuneration for the year ending December 31, 2024. | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | Ordinary Resolutions | | | | FOR | | | | AGAINST | | | | WITHHELD | | | | DISCRETION | | | | | | |
| | | Resolution 12 | | | | To approve, on an advisory (non-binding) basis, the compensation of the Company’s named executive officers. | | | | | | | | | | | | | | | | | | | | | | |
| | | Resolution 13 | | | | To authorize the Board of Directors to exercise all the powers of the Company to allot shares in the Company or grant rights to subscribe for or to convert any security into shares in the Company up to an aggregate nominal amount of £8,345,745 (being up to a maximum of 166,914,908 shares representing 25% of the Company’s existing ordinary share capital as at the close of business on March 13, 2024 (being the latest practicable date prior to publication of this document)). | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | Special Resolutions | | | | FOR | | | | AGAINST | | | | WITHHELD | | | | DISCRETION | | | | | | |
| | | Resolution 14 | | | | To authorize the Board of Directors to allot equity securities for cash and/or to sell ordinary shares held by the Company as treasury shares for cash as if Section 561(1) of the Companies Act does not apply to any such allotment. | | | | | | | | | | | | | | | | | | | | | | |