| Item 7.01. | Regulation FD Disclosure. |
On September 27, 2021, Evelo Biosciences, Inc. (Evelo) announced data from its Phase 2 clinical trial of EDP1815 in psoriasis and hosted a corporate update conference call with a live webcast. A copy of the slide presentation from the webcast is furnished as Exhibit 99.1 to this Current Report on Form 8-K and a copy of the press release issued in connection with the announcement is furnished as Exhibit 99.2 to this Current Report on Form 8-K. The information contained in this Item 7.01 shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (Exchange Act), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly provided by specific reference in such filing.
Phase 2 Clinical Data with EDP1815 in Psoriasis
On September 27, 2021, Evelo announced data from its Phase 2 clinical trial of EDP1815 in psoriasis. A statistically significant reduction in the Psoriasis Area and Severity Index (PASI) score, as measured by the proportion of patients achieving at least 50% improvement in PASI from baseline at the week 16 timepoint, was observed in the study. EDP1815 is an investigational oral biologic currently in development for the treatment of a broad range of inflammatory diseases, including clinical programs in psoriasis, atopic dermatitis, and COVID-19.
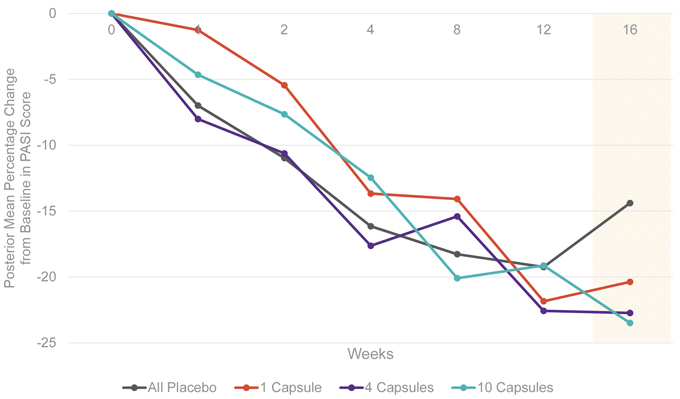
In the Phase 2 study, the PASI scores were assessed by both mean changes from baseline and responder rates. The primary endpoint was the mean percentage change in PASI between treatment and placebo and was prespecified as a Bayesian analysis. The Bayesian approach provides an estimate of the probability that EDP1815 is superior to placebo. The 16-week primary endpoint gave probabilities that EDP1815 is superior to placebo ranging from 80% to 90% across the prespecified analyses and cohorts.
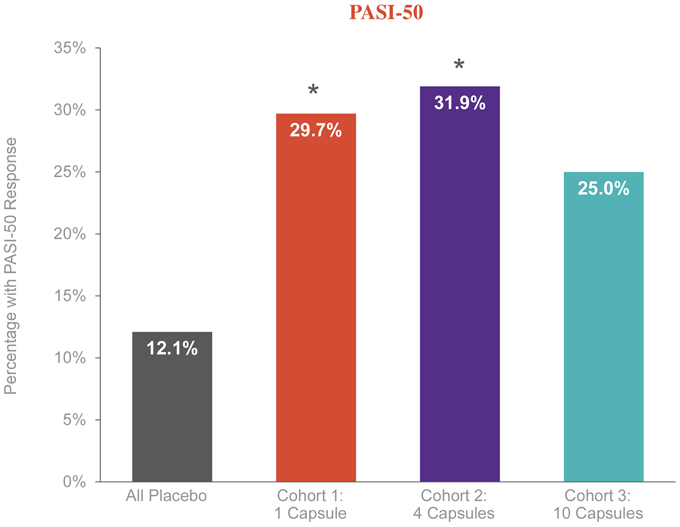
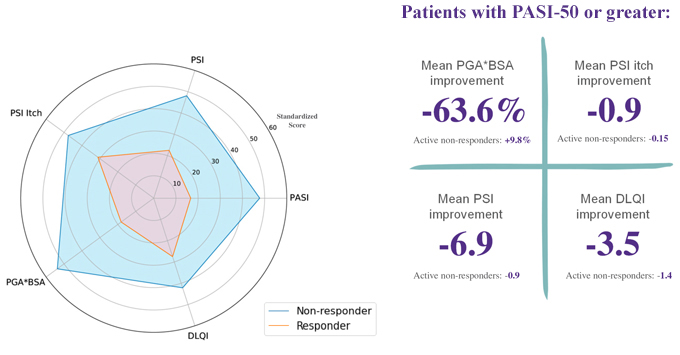
The responder endpoint reports the proportion of patients who had a meaningful clinical response, which is defined as PASI-50 or greater. 25% to 32% of patients across the three cohorts who were treated with EDP1815 achieved a PASI-50 at week 16 compared to 12% on placebo. In cohorts 1 and 2 this difference in response rate was statistically significant (p <0.05). Cohort 3 was directionally similar (25% vs. 12%). The pooled PASI-50 response across all three EDP1815 cohorts, an exploratory analysis, was 29% vs. 12% for placebo and was also statistically significant with a p-value of 0.027. An increase in the number of capsules of EDP1815 did not lead to a dose response.
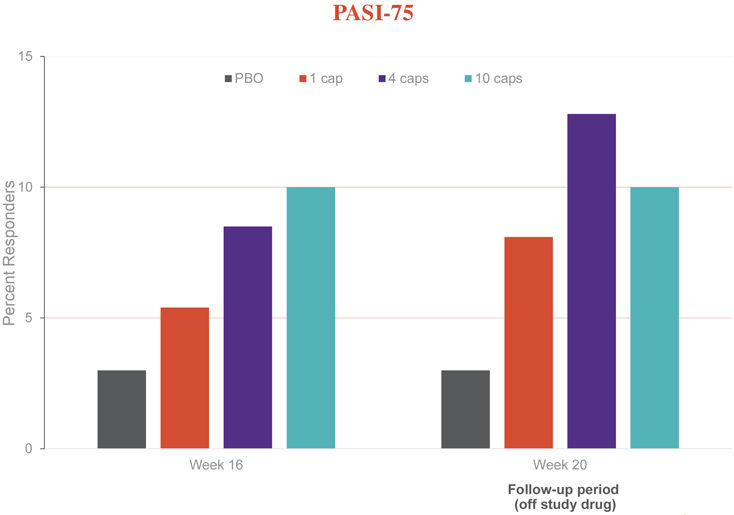
Additionally, several patients on EDP1815 achieved a PASI-75 or better, which was sustained or improved post treatment. For individuals who had a PASI-50 response or better, consistent effects in secondary and exploratory endpoints, including improvements in patient reported outcomes such as Dermatology Life Quality Index (DLQI) and Psoriasis Symptom Inventory (PSI), were observed.
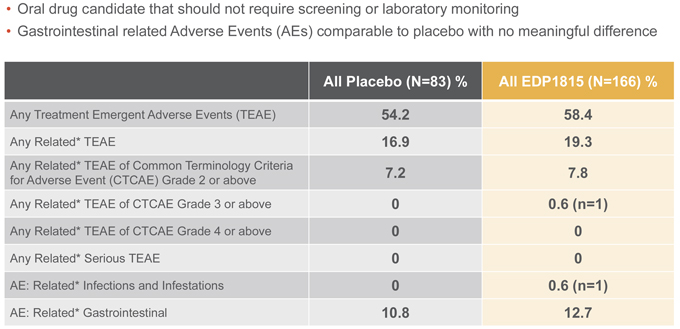
EDP1815 was observed to be well tolerated in the Phase 2 study. The safety data were comparable to placebo and consistent with what was previously reported in a Phase 1b study. Adverse events (AEs) classified as “gastrointestinal” were comparable between active and placebo groups, with no meaningful differences in rates of diarrhea, abdominal pain, nausea, or vomiting. There were no related serious adverse events.
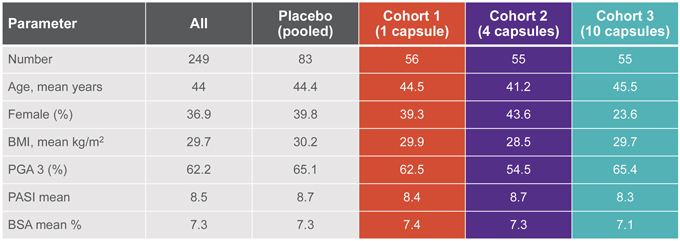
EDP1815-201 is a double-blind, placebo-controlled, dose-ranging Phase 2 study designed to evaluate three doses of an enteric capsule formulation of EDP1815 versus placebo in 249 patients with mild and moderate psoriasis over a 16-week treatment period. In the study, the PASI scores were assessed by both mean changes from baseline and responder rates. The primary endpoint is mean percentage reduction in PASI score at 16 weeks. Secondary endpoints include the proportion of study participants who achieve a PASI-50 response or greater and other clinical measures of disease such as Physicians Global Assessment (PGA), Body Surface Area (BSA), PGA x BSA, PSI, and DLQI.
The corporate update included the following information:
Baseline characteristics are well-balanced across treatment groups