
Frequency Therapeutics Corporate Overview May, 2020 Exhibit 99.3

FORWARD-LOOKING STATEMENTS AND OTHER DISCLAIMERS This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this presentation that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements regarding the treatment potential of FX-322, the design and enrollment of the Phase 2a clinical trial of FX-322, the timing of top-line data from the Phase 2a clinical trial and timing of presentation of results of the exploratory study in Germany, the novelty of the exploratory study in Germany, the implications of the results of the exploratory Germany study in combination with our other trials, the therapeutic levels of FX-322 predicted in the exploratory Germany study, the ability of our technology platform to provide patient benefit, the impact of COVID-19 on the Company’s on-going and planned clinical trials and business, future milestone and royalty payments under the license and collaboration agreement with Astellas Pharma Inc. (“Astellas”), the sufficiency of the Company’s cash, cash equivalents and short-term investments, estimates of the size of the hearing loss population and population at risk for hearing loss, the timing of the remyelination program, and the potential application of the PCA platform to other diseases. These forward-looking statements are based on management’s current expectations. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: Frequency Therapeutics (the “Company”) has incurred and will continue to incur significant losses and is not and may never be profitable; need for additional funding to complete development and commercialization of any product candidate; the Company’s dependence on the development of FX-322; the unproven approach of the PCA platform; the lengthy, expensive and uncertain process of clinical drug development and regulatory approval; limited experience successfully obtaining marketing approval for and commercializing product candidates; the results of earlier clinical trials not being indicative of the results from later clinical trials; differences between preliminary or interim data and final data; adverse events or undesirable side effects; disruptions at the FDA and other regulatory agencies; the impact of the COVID-19 impact; failure to identify additional product candidates; new or changed legislation; failure to maintain Fast Track designation for FX-322 and such designation failing to result in faster development or regulatory review or approval; costly and damaging litigation, including related to product liability, intellectual property or brought by stockholders; dependence on Astellas for the development and commercialization of FX-322 outside of the United States; misconduct by employees or independent contractors; reliance on third parties, including to conduct clinical trials and manufacture product candidates; compliance with laws and regulations, including healthcare and environmental, health, and safety laws and regulations; failure to obtain, maintain and enforce protection of patents and other intellectual property; security breaches or failure to protect private personal information; attracting and retaining key personnel; and ability to manage growth. These and other important factors discussed under the caption “Risk factors” in the Company’s Form 10-Q filed with the Securities and Exchange Commission (SEC) on May 14, 2020 and its other reports filed with the SEC could cause actual results to differ materially from those indicated by the forward-looking statements made in this presentation. Any such forward-looking statements represent management’s estimates as of the date of this presentation. While the Company may elect to update such forward-looking statements at some point in the future, it disclaims any obligation to do so, even if subsequent events cause its views to change. These forward-looking statements should not be relied upon as representing the Company’s views as of any date subsequent to the date of this presentation.

Frequency Today FX-322 program for sensorineural hearing loss in Phase 2a clinical development Phase 1/2 results provided evidence of hearing restoration; favorable safety profile Phase 2a study ongoing; continue to enroll at a number of sites; some sites have temporarily stopped new enrollment due to COVID-19 Recent clinical study data confirms FX-322 delivery to the cochlea FDA Fast Track designation Additional opportunities across multiple degenerative diseases Multiple sclerosis program advancing toward IND; target submission H2 2021 Broad IP portfolio in hearing and additional applications Operating runway into 2022 $206.1 million in cash, cash equivalents and short-term investments (as of March 31, 2020) Potential development milestones related to Astellas ex-US FX-322 collaboration including $90M at first 2b dosing in Europe & Asia and $140M for the first Phase 3 dosing in Europe & Asia
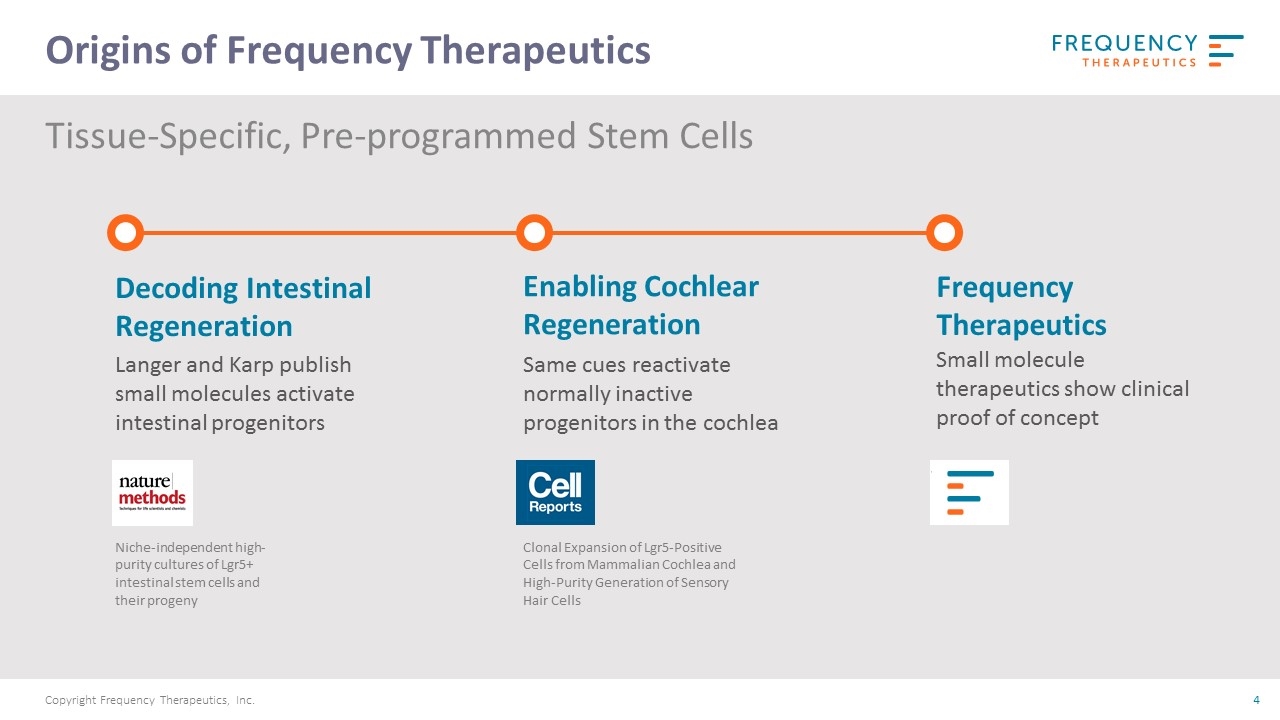
Langer and Karp publish small molecules activate intestinal progenitors Decoding Intestinal Regeneration Same cues reactivate normally inactive progenitors in the cochlea Enabling Cochlear Regeneration Frequency Therapeutics Small molecule therapeutics show clinical proof of concept Niche-independent high-purity cultures of Lgr5+ intestinal stem cells and their progeny Clonal Expansion of Lgr5-Positive Cells from Mammalian Cochlea and High-Purity Generation of Sensory Hair Cells Tissue-Specific, Pre-programmed Stem Cells Origins of Frequency Therapeutics
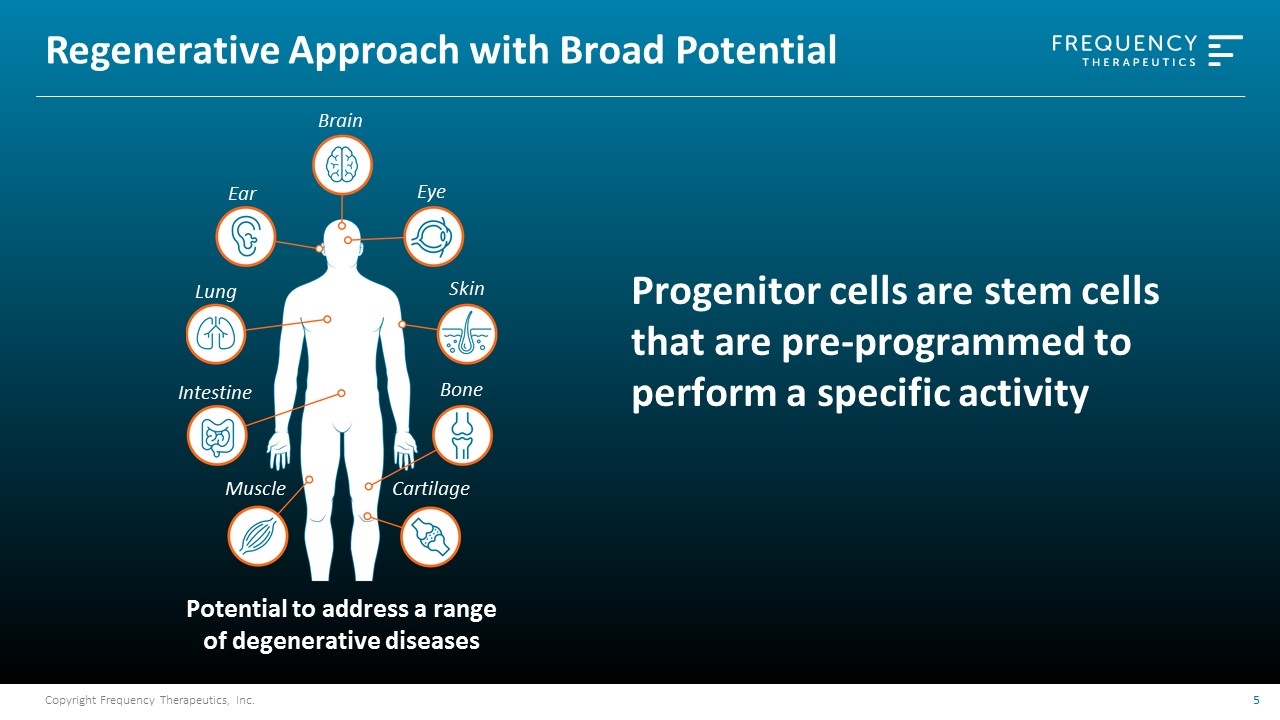
Regenerative Approach with Broad Potential Potential to address a range of degenerative diseases Brain Ear Lung Intestine Cartilage Skin Bone Muscle Eye Progenitor cells are stem cells that are pre-programmed to perform a specific activity

Reducing the Complexity of Regenerative Medicine Harnessing Innate Biology Progenitors already located within target tissue No Change To Genome Activating native programs, reducing safety concerns Ease of Manufacturing Use of small molecules: no need to remove or grow cells ex vivo
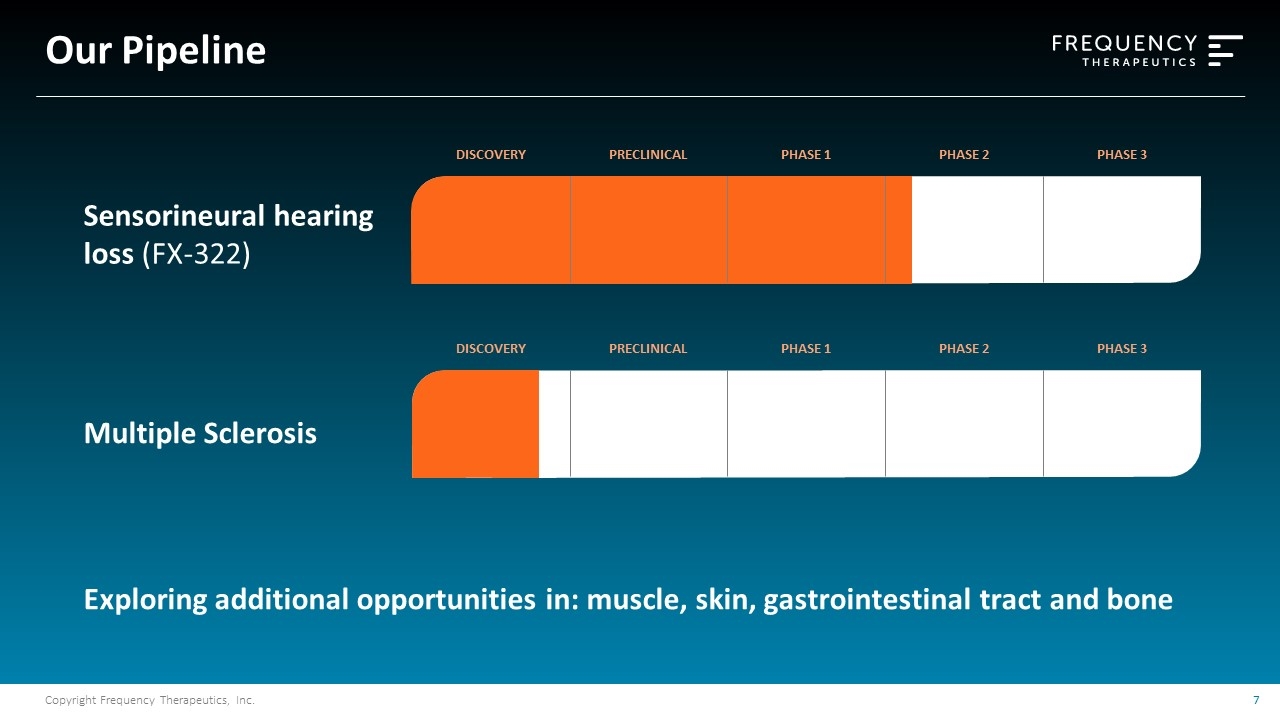
Our Pipeline Sensorineural hearing loss (FX-322) DISCOVERY PRECLINICAL PHASE 1 PHASE 2 PHASE 3 DISCOVERY PRECLINICAL PHASE 1 PHASE 2 PHASE 3 Exploring additional opportunities in: muscle, skin, gastrointestinal tract and bone Multiple Sclerosis

Hearing Restoration Program: Preclinical Validation
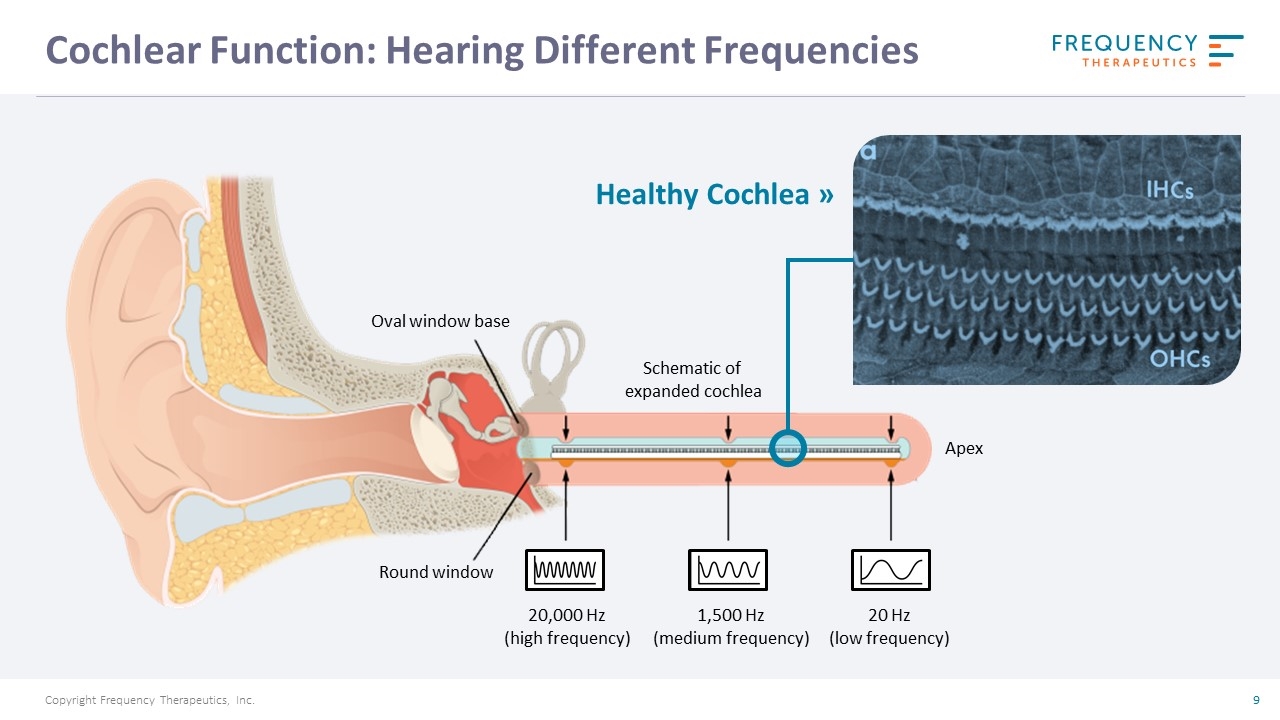
Cochlear Function: Hearing Different Frequencies Healthy Cochlea » Oval window base Round window 20,000 Hz (high frequency) 1,500 Hz (medium frequency) 20 Hz (low frequency) Schematic of expanded cochlea Apex
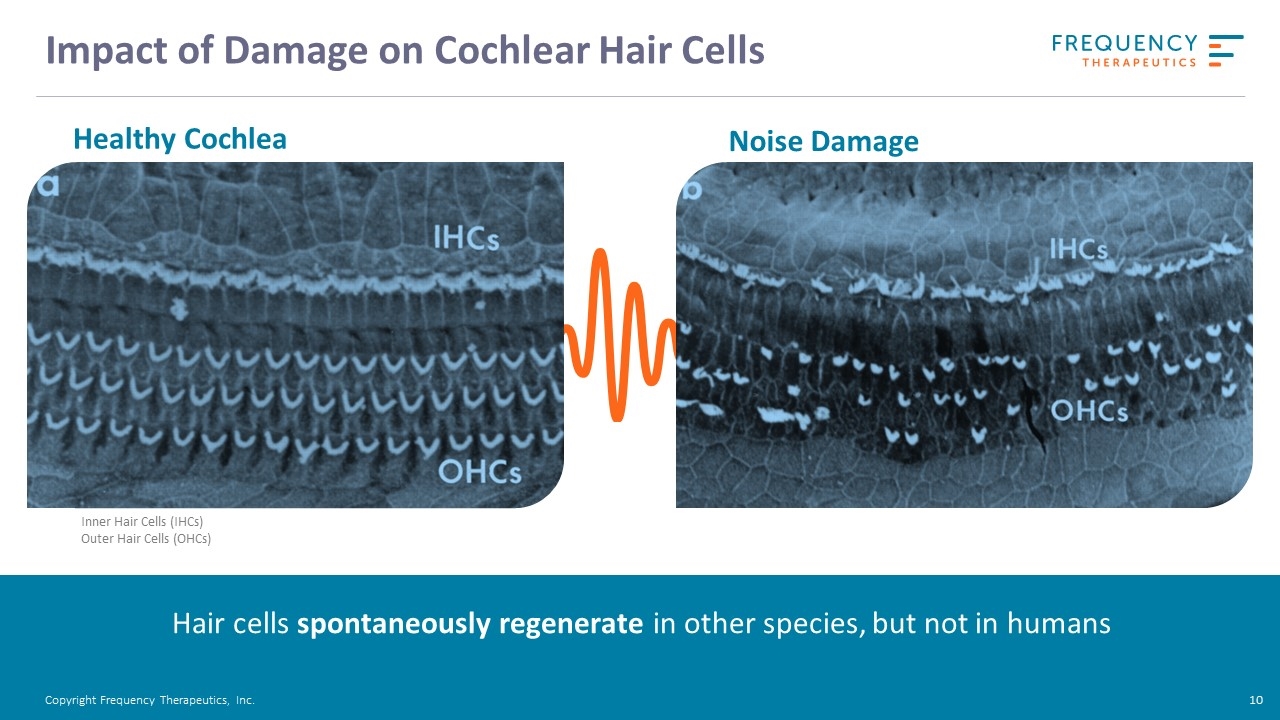
Impact of Damage on Cochlear Hair Cells Healthy Cochlea Inner Hair Cells (IHCs) Outer Hair Cells (OHCs) Hair cells spontaneously regenerate in other species, but not in humans Noise Damage Copyright Frequency Therapeutics, Inc.

Cochlear Progenitor Proliferation (Lgr5-GFP) Culture Media Wnt Activation (glycogen synthase kinase-3 (GSK3) Inhibitor; NCE) HDAC Inhibition (sodium valproate) Wnt Activation and HDAC inhibition HIGH SYNERGY Cell Rep. 2017 Feb 21;18(8):1917-1929. doi: 10.1016/j.celrep.2017.01.066 Strong synergy seen between pathways activated by small molecules HDAC = Histone deacetylase In vitro mouse model testing NCE = new chemical entity Profound, Unexpected Synergy

FX-322 Program: Aiming to Repair Underlying Pathology Progenitors in place despite loss of hair cells FX-322 molecules aim to activate genetic pathways to regenerate missing hair cells
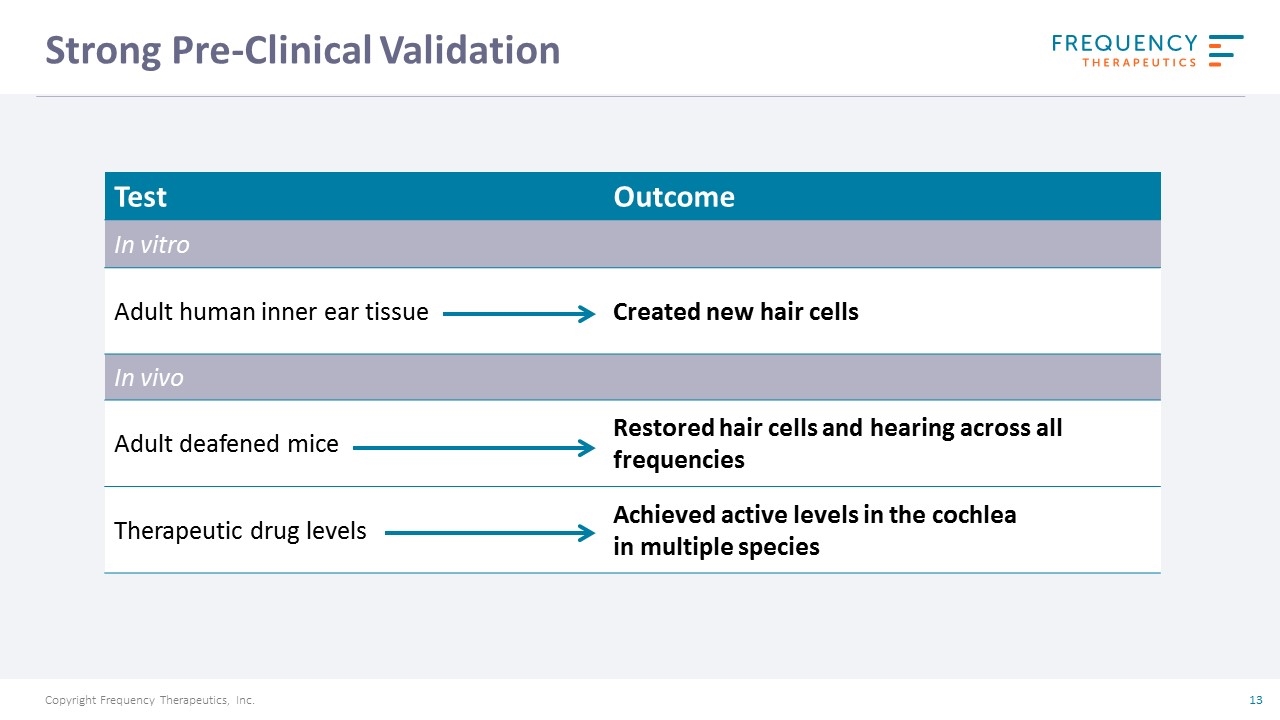
Strong Pre-Clinical Validation Test Outcome In vitro Adult human inner ear tissue Created new hair cells In vivo Adult deafened mice Restored hair cells and hearing across all frequencies Therapeutic drug levels Achieved active levels in the cochlea in multiple species

Clinical Program
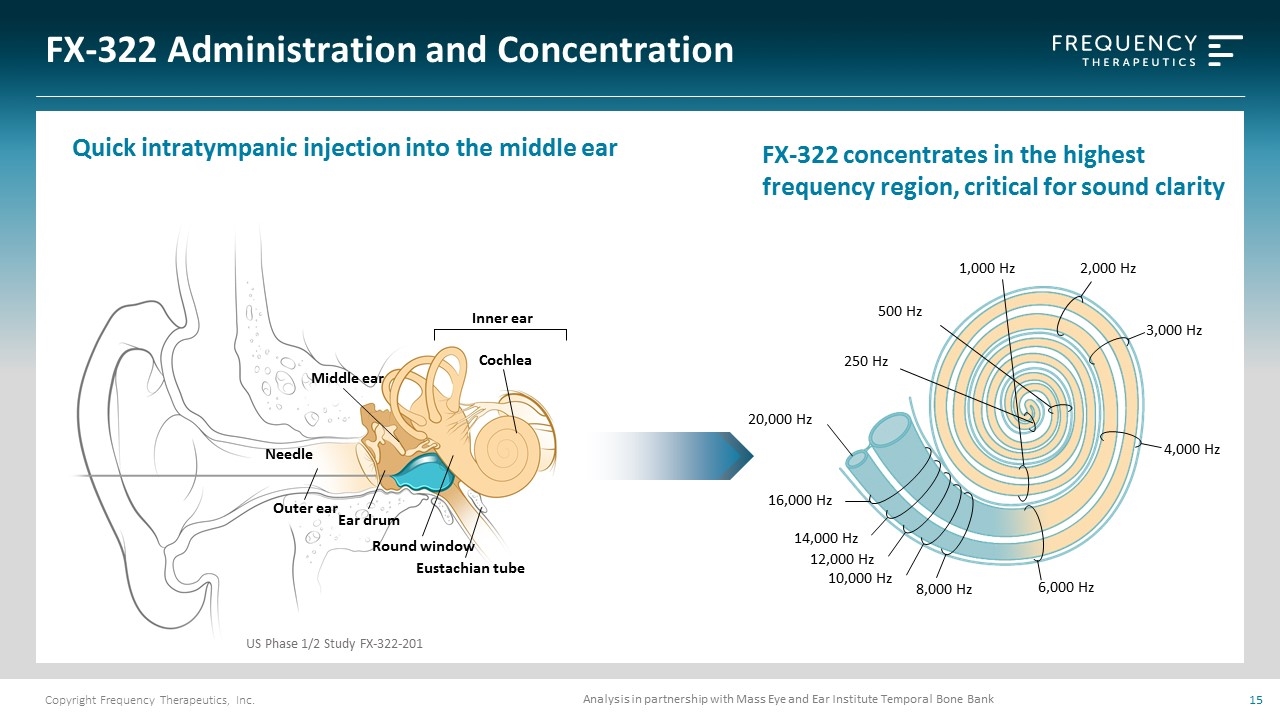
FX-322 Administration and Concentration Middle ear Cochlea Inner ear Outer ear Ear drum Round window Needle Eustachian tube US Phase 1/2 Study FX-322-201 Quick intratympanic injection into the middle ear Analysis in partnership with Mass Eye and Ear Institute Temporal Bone Bank 2,000 Hz 16,000 Hz 14,000 Hz 12,000 Hz 8,000 Hz 6,000 Hz 250 Hz 20,000 Hz 1,000 Hz 10,000 Hz 3,000 Hz 4,000 Hz 500 Hz FX-322 concentrates in the highest frequency region, critical for sound clarity

Hearing is Measured Using Validated Tests Intelligibility (Clarity) measured with word recognition and words-in-noise tests Audibility (Loudness) measured with pure tones Word Recognition Test: List of 50 monosyllabic words Single words played in quiet Words-in-Noise Test: Standard, clinically accepted measure Background noise from multiple voices Played at different signal-to-noise ratios
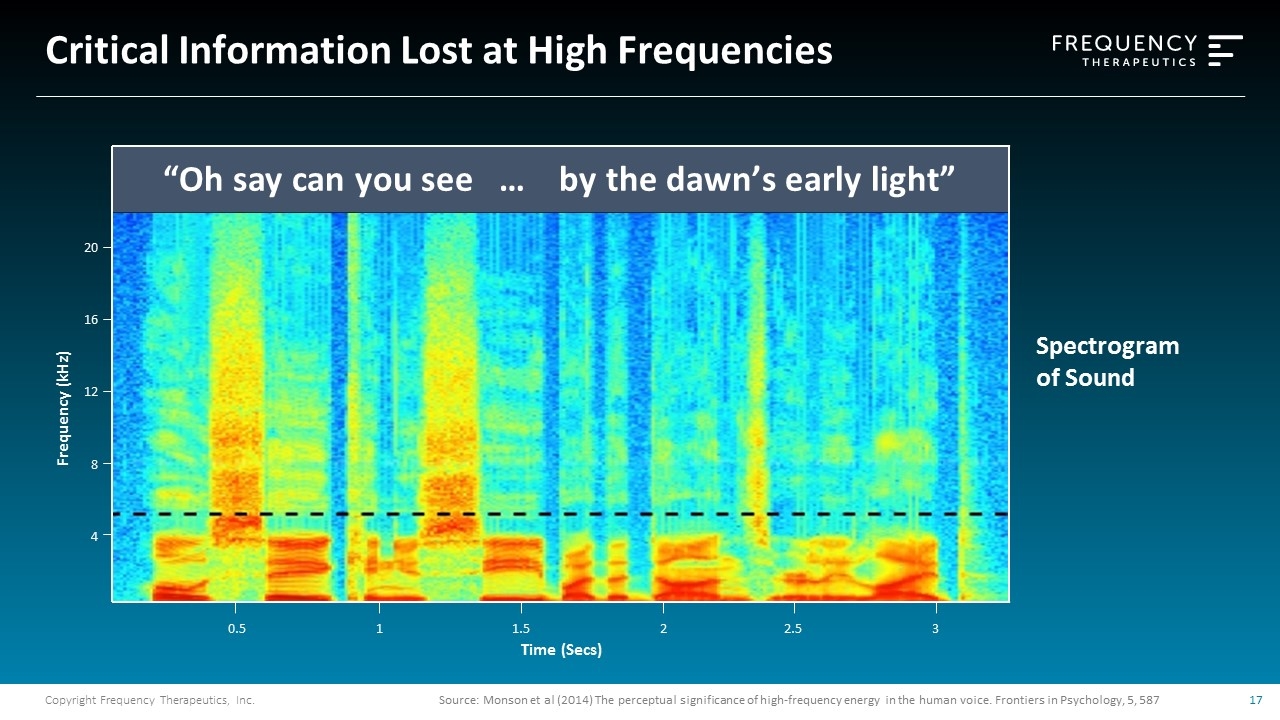
Source: Monson et al (2014) The perceptual significance of high-frequency energy in the human voice. Frontiers in Psychology, 5, 587 20 16 12 8 4 Frequency (kHz) “Oh say can you see … by the dawn’s early light” Spectrogram of Sound Critical Information Lost at High Frequencies 0.5 1 1.5 2 2.5 3 Time (Secs)
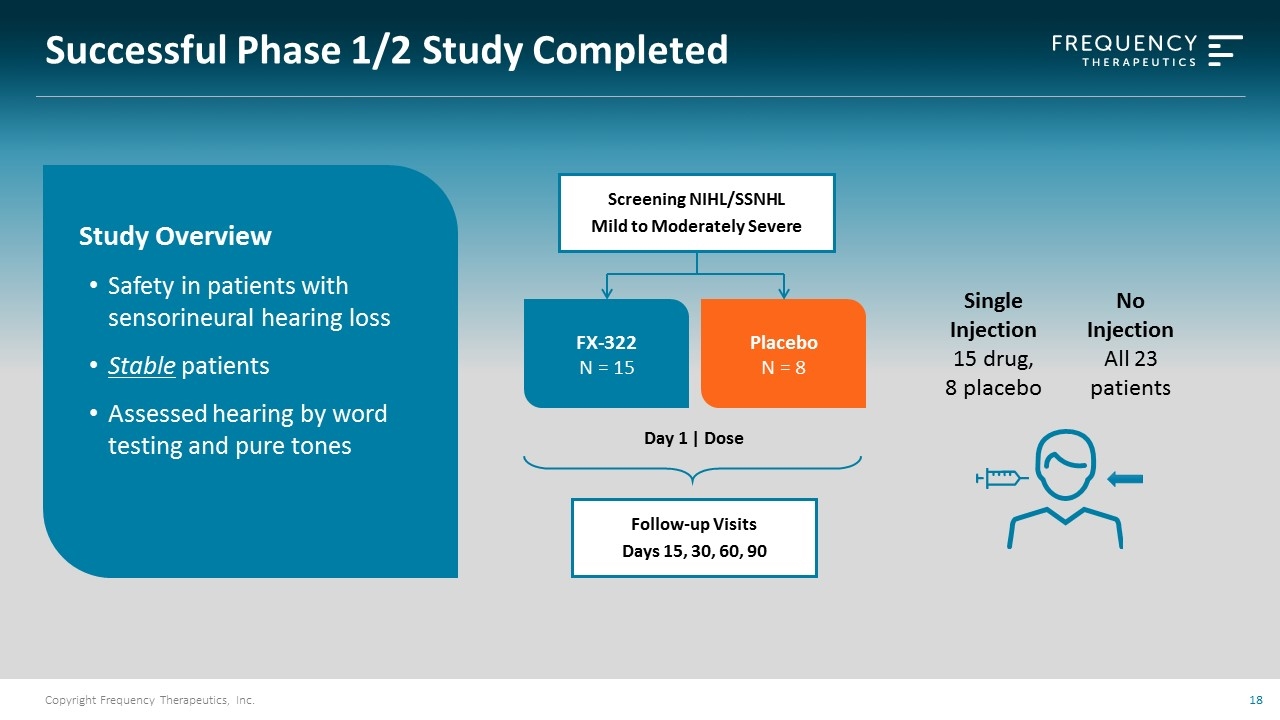
Successful Phase 1/2 Study Completed Single Injection 15 drug, 8 placebo No Injection All 23 patients Study Overview Safety in patients with sensorineural hearing loss Stable patients Assessed hearing by word testing and pure tones Follow-up Visits Days 15, 30, 60, 90 Day 1 | Dose Screening NIHL/SSNHL Mild to Moderately Severe Placebo N = 8 FX-322 N = 15

Clinically Meaningful Improvement in Word Recognition Mild Hearing Loss 14 patients (5 placebo) Typically very high word recognition scores (45+ words) Ceiling effect: Limited room for improvement Moderate to Moderately Severe Hearing Loss 9 patients (3 placebo) All treated subjects showed increases in word recognition 4 of 6 showed statistically significant and clinically meaningful improvement No change in placebo No change in untreated ear
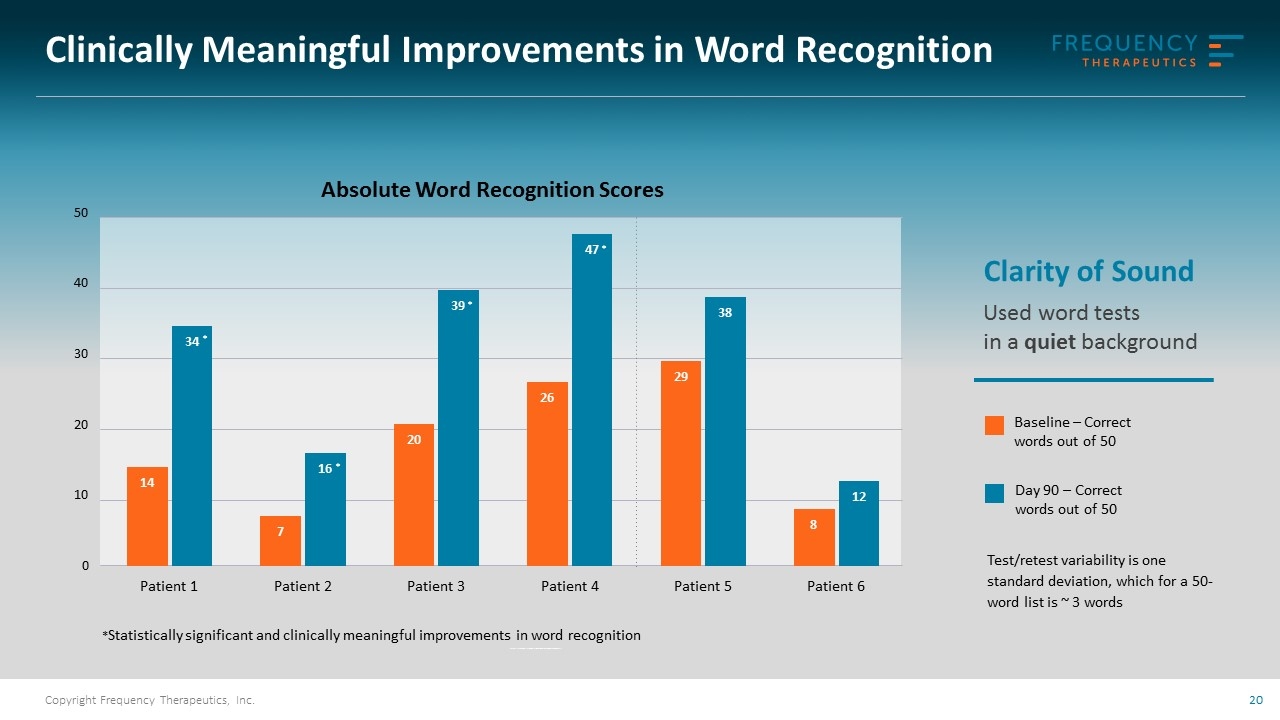
Baseline – Correct words out of 50 Day 90 – Correct words out of 50 Clinically Meaningful Improvements in Word Recognition Clarity of Sound Used word tests in a quiet background Absolute Word Recognition Scores *Statistically significant and clinically meaningful improvements in word recognition * * * * Test/retest variability is one standard deviation, which for a 50-word list is ~ 3 words Patient 1 14 34 patient 2 7 16 patient 3 20 39 patient 4 26 47 patient 29 38 patient 8 12
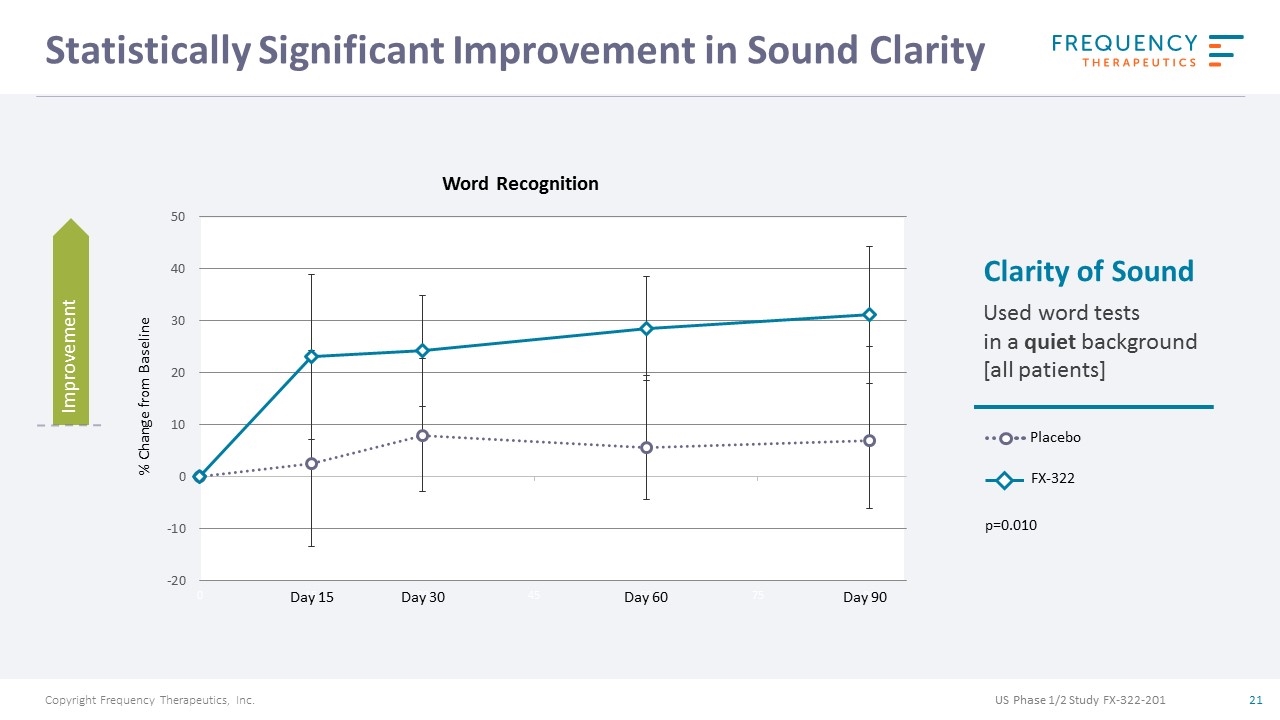
Statistically Significant Improvement in Sound Clarity Placebo FX-322 Clarity of Sound Used word tests in a quiet background [all patients] Word Recognition Day 15 Day 30 Day 60 Day 90 p=0.010 % Change from Baseline Improvement US Phase 1/2 Study FX-322-201
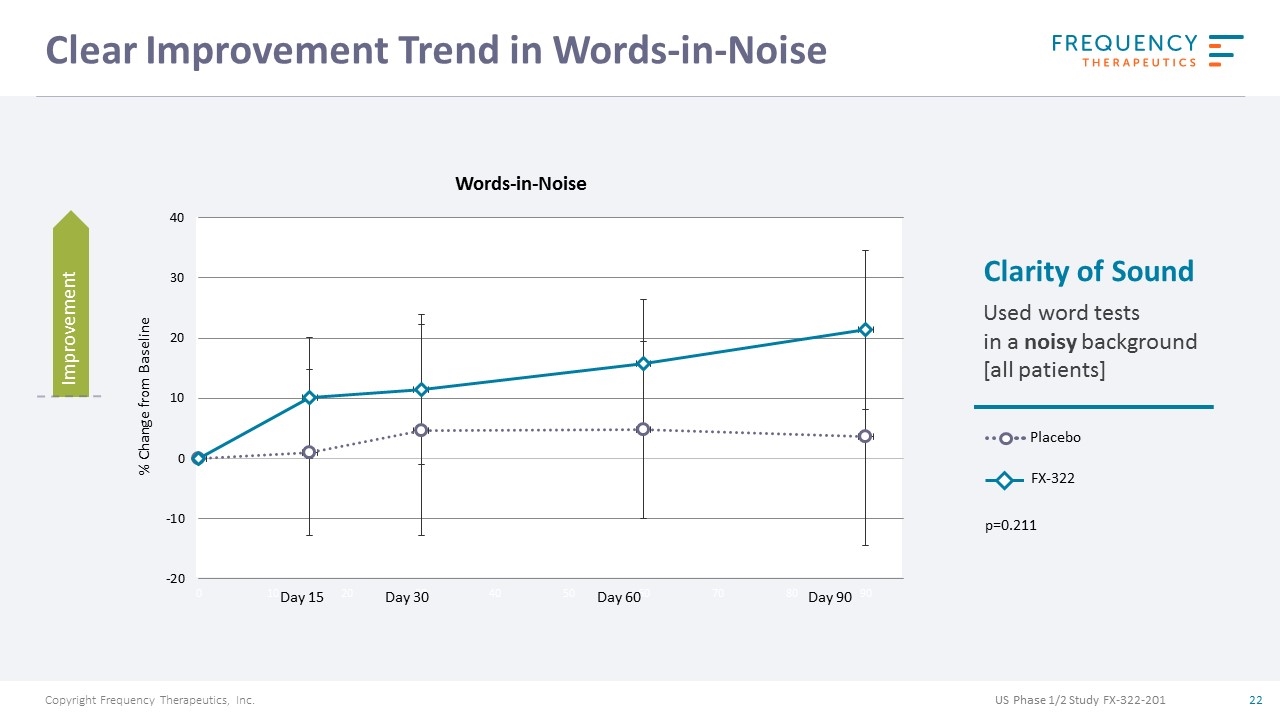
Clear Improvement Trend in Words-in-Noise Placebo FX-322 Clarity of Sound Used word tests in a noisy background [all patients] Words-in-Noise Day 15 Day 30 Day 60 Day 90 p=0.211 % Change from Baseline Improvement US Phase 1/2 Study FX-322-201
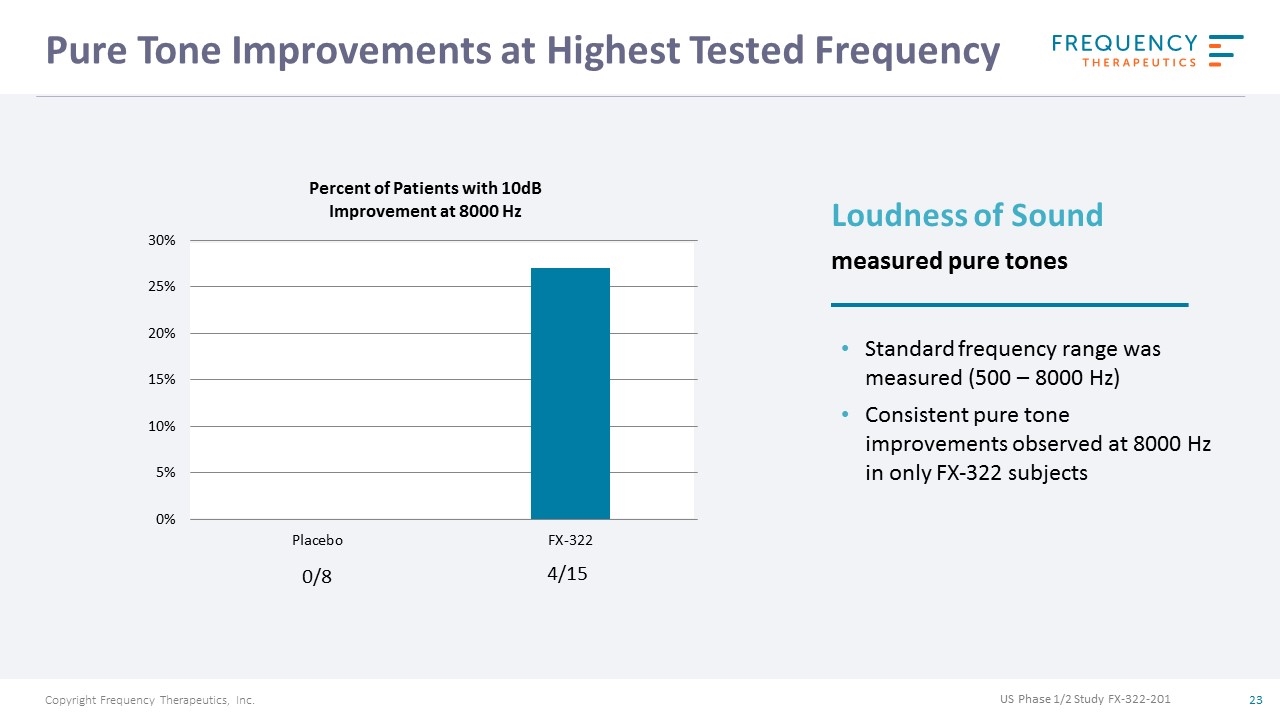
Pure Tone Improvements at Highest Tested Frequency 0/8 4/15 US Phase 1/2 Study FX-322-201 Loudness of Sound measured pure tones Standard frequency range was measured (500 – 8000 Hz) Consistent pure tone improvements observed at 8000 Hz in only FX-322 subjects

Data Presented at AAO-HNS, September 2019 Phase 1/2 results delivered by principal investigator First ever clinical data to show hearing improvement Significant enthusiasm amongst community Results since shared broadly with ENT and auditory science KOLs
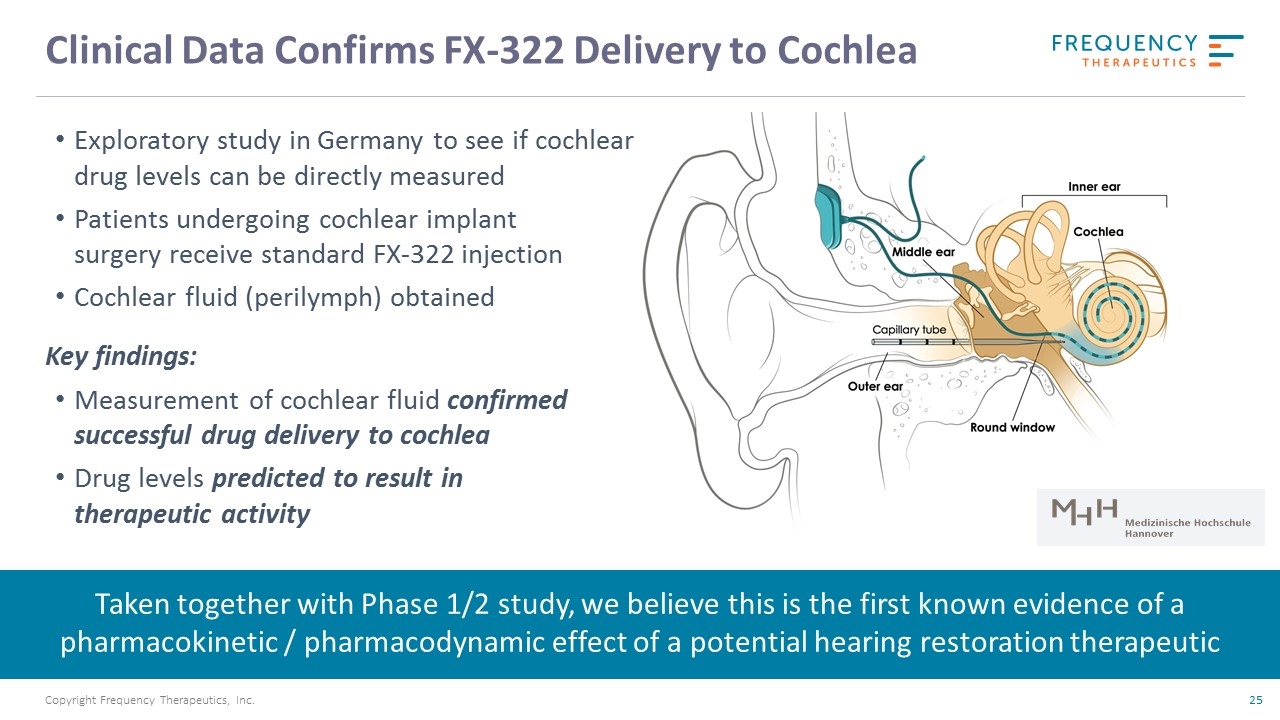
Clinical Data Confirms FX-322 Delivery to Cochlea Exploratory study in Germany to see if cochlear drug levels can be directly measured Patients undergoing cochlear implant surgery receive standard FX-322 injection Cochlear fluid (perilymph) obtained Key findings: Measurement of cochlear fluid confirmed successful drug delivery to cochlea Drug levels predicted to result in therapeutic activity Taken together with Phase 1/2 study, we believe this is the first known evidence of a pharmacokinetic / pharmacodynamic effect of a potential hearing restoration therapeutic
Leadership in Hearing Drug Development Key elements in FX-322 development: Effective delivery to the target tissue Favorable safety profile Clinically meaningful improvements in hearing function

Phase 2a Study – Objectives Evaluate repeat dosing Clarify endpoints and patient population Further establish hearing signal
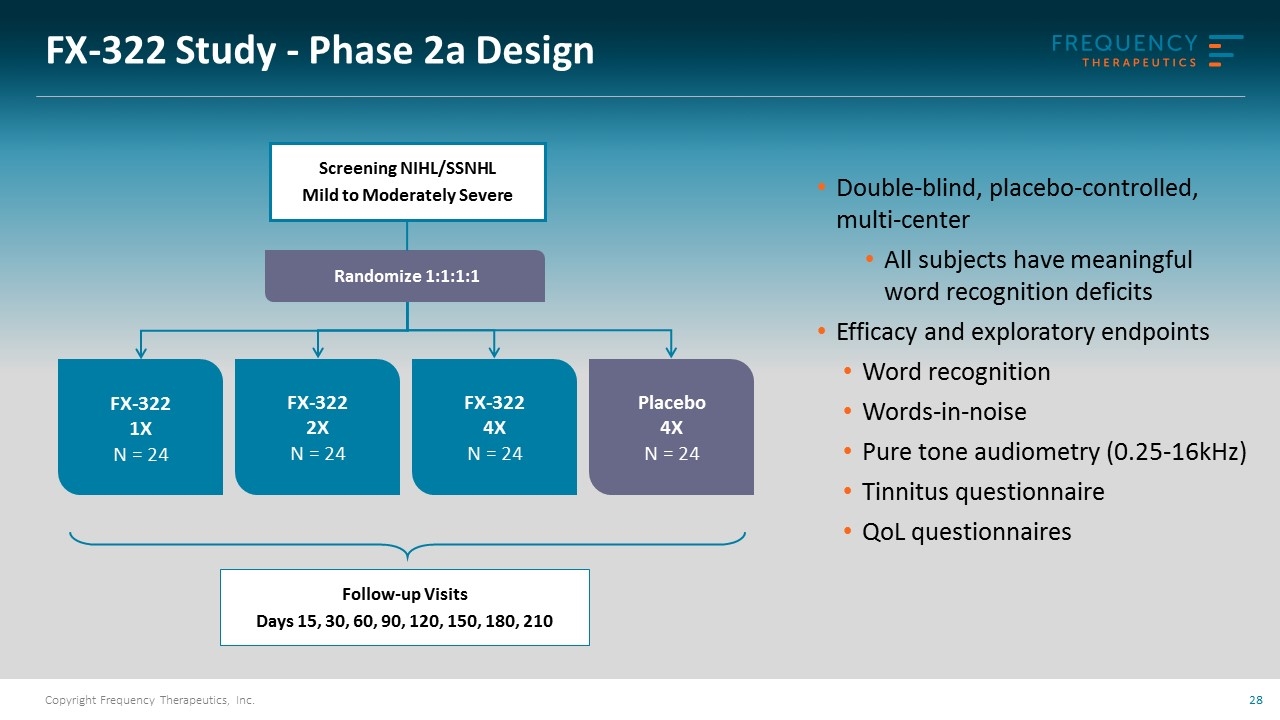
FX-322 Study - Phase 2a Design Placebo 4X N = 24 Follow-up Visits Days 15, 30, 60, 90, 120, 150, 180, 210 Randomize 1:1:1:1 FX-322 4X N = 24 FX-322 2X N = 24 FX-322 1X N = 24 Double-blind, placebo-controlled, multi-center All subjects have meaningful word recognition deficits Efficacy and exploratory endpoints Word recognition Words-in-noise Pure tone audiometry (0.25-16kHz) Tinnitus questionnaire QoL questionnaires Screening NIHL/SSNHL Mild to Moderately Severe

Stringent Enrollment Criteria Only double-blind, placebo-controlled hearing restoration studies for SNHL – greater rigor over past approaches Stable Sensorineural Hearing Loss: Careful screening of all study subjects Working to ensure study subjects have disease etiology that FX-322 may help treat Aiming to reduce endpoint variability

Hearing Loss Overview
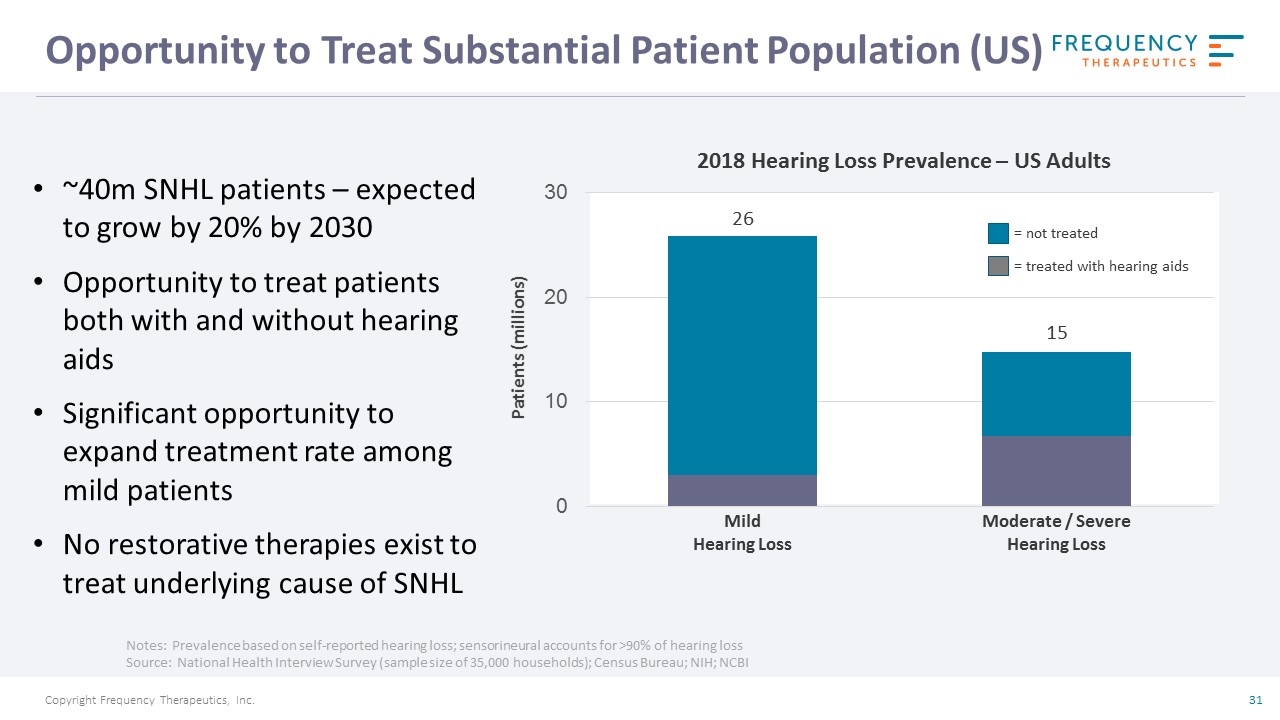
Opportunity to Treat Substantial Patient Population (US) Notes: Prevalence based on self-reported hearing loss; sensorineural accounts for >90% of hearing loss Source: National Health Interview Survey (sample size of 35,000 households); Census Bureau; NIH; NCBI Moderate / Severe Hearing Loss Mild Hearing Loss 2018 Hearing Loss Prevalence – US Adults Patients (millions) = not treated = treated with hearing aids 15 26 ~40m SNHL patients – expected to grow by 20% by 2030 Opportunity to treat patients both with and without hearing aids Significant opportunity to expand treatment rate among mild patients No restorative therapies exist to treat underlying cause of SNHL
“I can hear you… I just can’t understand you.” Improvement in Hearing Clarity is the Major Unmet Need Among Hearing Loss Patients

Hearing Health Impacts Brain Health “Hearing loss is the largest modifiable risk factor for developing dementia” DEC 30, 2019 Risks Associated with Untreated Hearing Loss DEMENTIA 50% 41% DEPRESSION JAMA Nov 8, 2018 Deal J, et al. Incident Hearing Loss and Comorbidity. A Longitudinal Administrative Claims Study.

Broad Potential of Progenitor Cell Activation Approach

Building on strong progenitor biology foundation Scripps Institute applied PCA approach to brain progenitors Validated in multiple animal models Clinically validated using a single agent (Lancet Dec ’17) Target IND submission in 2H21 Building on expertise in discovering and developing synergistic combinations of small molecules Worldwide license and ongoing research partnership with Scripps Remyelination Program for Multiple Sclerosis

Moving Forward
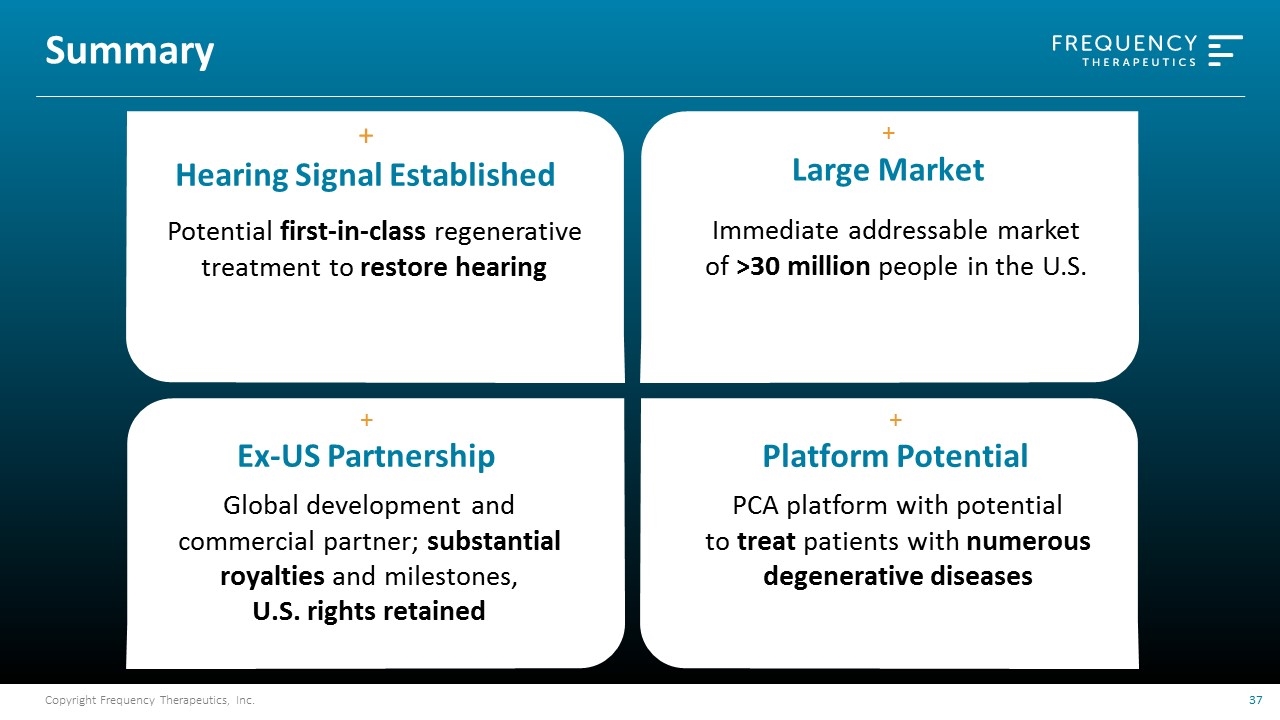
Summary + Hearing Signal Established Potential first-in-class regenerative treatment to restore hearing + Large Market Immediate addressable market of >30 million people in the U.S. + Ex-US Partnership Global development and commercial partner; substantial royalties and milestones, U.S. rights retained + Platform Potential PCA platform with potential to treat patients with numerous degenerative diseases

Appendix
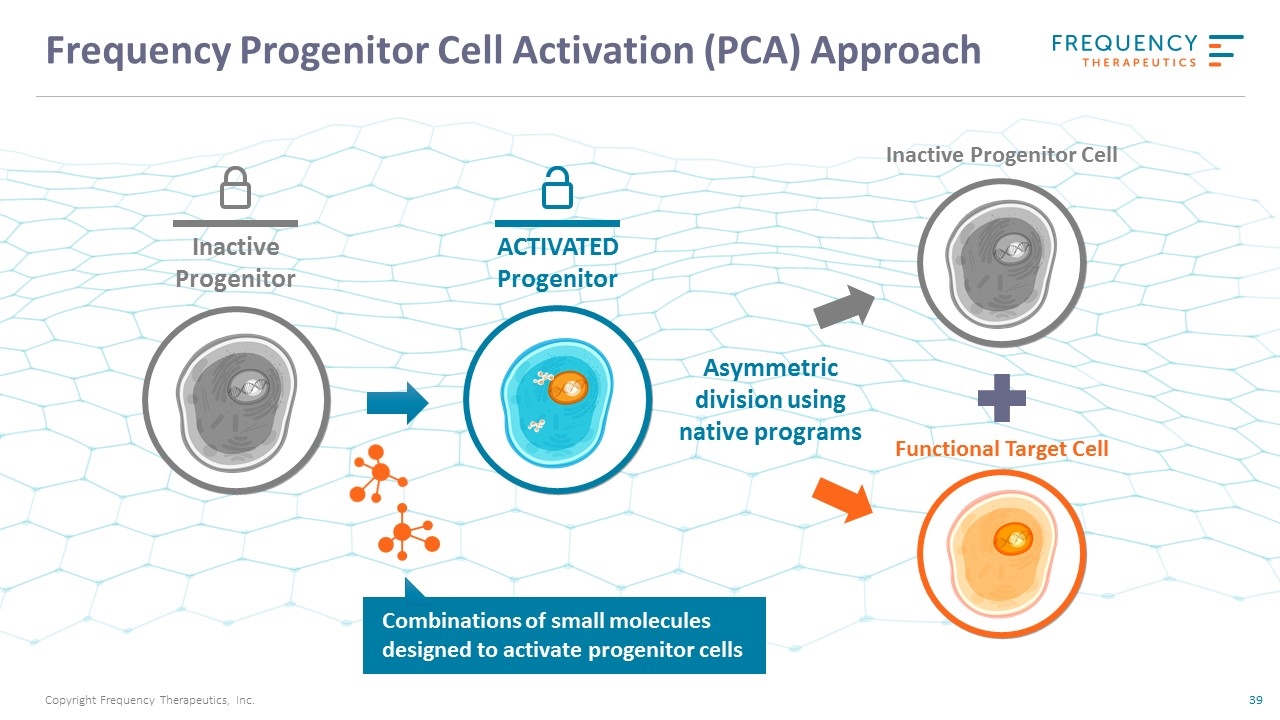
Frequency Progenitor Cell Activation (PCA) Approach Inactive Progenitor ACTIVATED Progenitor Asymmetric division using native programs Combinations of small molecules designed to activate progenitor cells Inactive Progenitor Cell Functional Target Cell
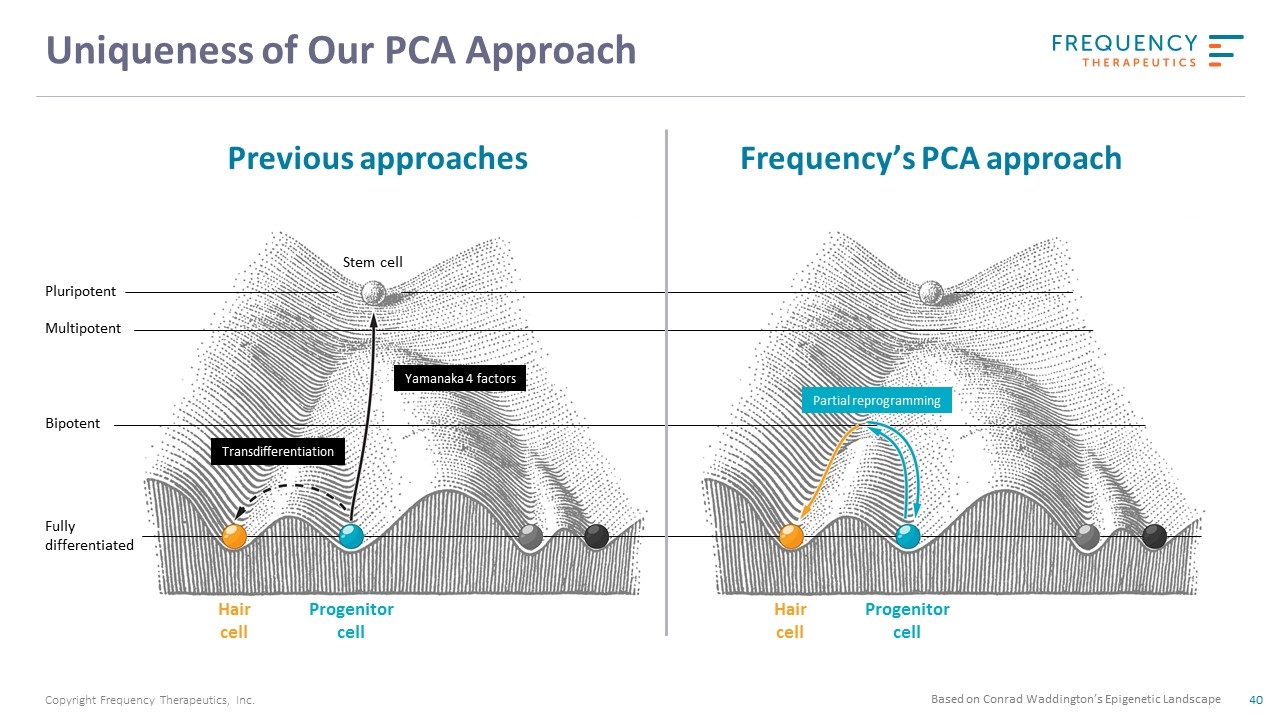
Uniqueness of Our PCA Approach Pluripotent Multipotent Bipotent Fully differentiated Hair cell Progenitor cell Transdifferentiation Yamanaka 4 factors Stem cell Partial reprogramming Hair cell Progenitor cell Based on Conrad Waddington’s Epigenetic Landscape Previous approaches Frequency’s PCA approach
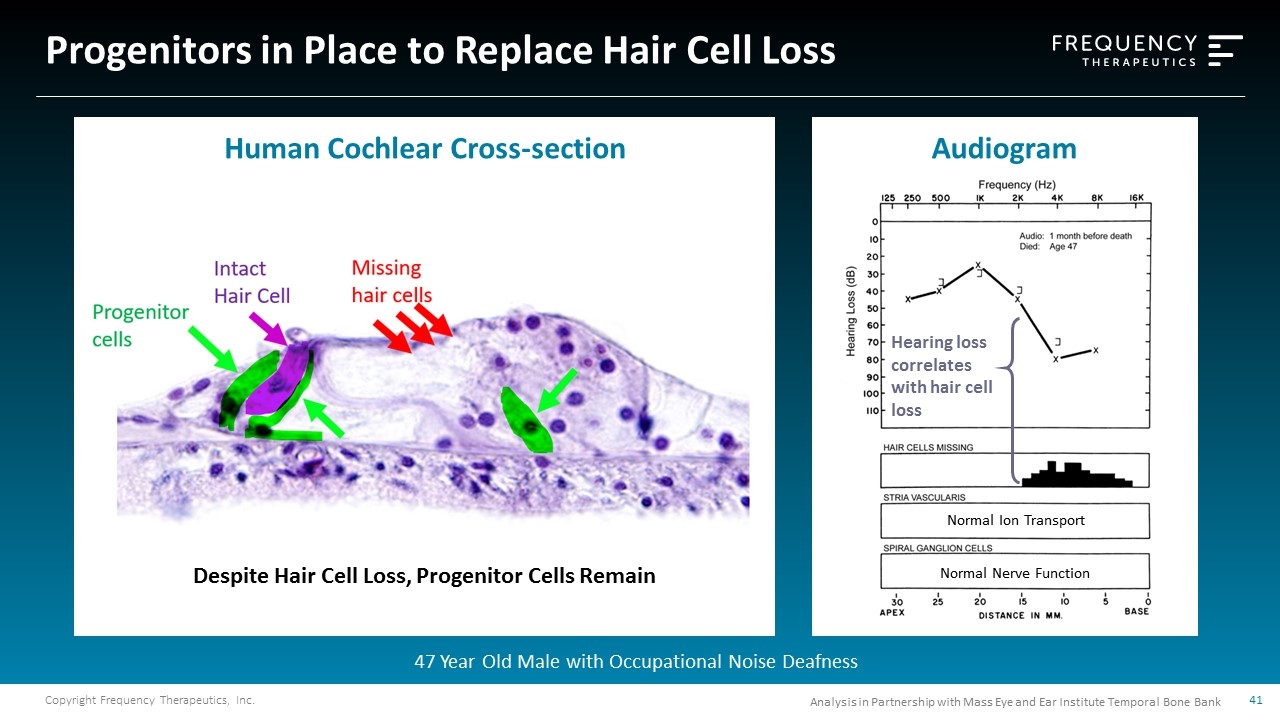
Progenitors in Place to Replace Hair Cell Loss Hearing loss correlates with hair cell loss Normal Ion Transport Normal Nerve Function Despite Hair Cell Loss, Progenitor Cells Remain 47 Year Old Male with Occupational Noise Deafness Analysis in Partnership with Mass Eye and Ear Institute Temporal Bone Bank Human Cochlear Cross-section Audiogram

Astellas Collaboration Development and commercialization collaboration for FX-322, including lifecycle improvements Astellas has ex-US rights; Frequency retains US rights to FX-322 Payments of up to $625mm which included $80mm upfront Development milestone payments to Frequency of $65.0 million and $25.0 million upon the first dosing of a patient in a Phase 2b clinical trial for SNHL in Europe and Asia, respectively $100.0 million and $40.0 million upon the first dosing of a patient in a Phase 3 clinical trial for SNHL in Europe and Asia, respectively Development & commercialization: Astellas responsible for execution and costs of ex-US clinical development and commercialization Strategic commitment to invest in ENT as a therapeutic area Research focus in regenerative medicine Global footprint in major markets and distributorship model in Africa/ME and LATAM

Proven Leadership Team David Lucchino President, CEO & Co-Founder Chris Loose, Ph.D. Chief Scientific Officer & Co-Founder Carl Lebel, Ph.D. Chief Development Officer Former CEO of Entrega Bio (PureTech). Co-founder/CEO of Semprus BioSciences (acquired), Polaris Partners. MIT Sloan Fellow. Co-founder/CTO of Semprus BioSciences through FDA/CE clearance and acquisition. Princeton, MIT, Hertz Fellow and Yale Faculty. Chief Scientific Officer of Otonomy (2009 to 2016). Executive Director, Amgen. Scientific fellow of the American Academy of Otolaryngology. Dana Hilt, M.D. Chief Medical Officer William Chin, M.D. EVP, Clinical & Translational Science Will McLean, Ph.D. VP, Biology & Regen. Med, Co-Founder Neurologist and neuroscientist with two decades in biopharma and CNS drug development. Amgen, Lysosomal, Forum Pharma. SVP, Discovery Research and Clinical Investigation at Eli Lilly, EVP at PhRMA, Executive Dean for Research at Harvard Medical School. 15 years experience in inner ear biology. Discovered specific stem cells that make hair cells and neurons. Harvard-MIT Health Sciences and Technology Program.

Hearing Clinical Advisory Board Dan Lee, M.D. | Mass Eye & Ear As the Director of the Pediatric Ear, Hearing and Balance Center, Dan Lee enjoys the challenges that come with diagnosing and treating children with complex ear conditions. David Friedland, M.D., Ph.D. | Wisconsin Board-certified otolaryngologist head and neck surgeon with specialized training in surgery for hearing restoration. He is Vice-Chair of the Department of Otolaryngology and Communications Sciences. Rene Gifford, Ph.D. | Vanderbilt As Director of the cochlear implant program, Rene Gifford’s research interests include combined electric and acoustic stimulation, hearing preservation, preoperative prediction of outcomes, speech perception and spatial hearing abilities with cochlear implants. Steve Rauch, M.D. | Mass Eye & Ear As an Otologist and Director of the Vestibular Division, Steve Rauch divides his time between clinical care and studying disorders that affect hearing and balance, such as Meniere's disease. Chris Runge, Ph.D. | Wisconsin As Chief of the Division of Communication Sciences, Chris Runge’s research interests include pediatric hearing disorders, genetics of hearing loss, speech and music understanding and the effects of aging on cochlear implant performance. Joni Doherty, MD, Ph.D. | USC Board Certified in both Otolaryngology-Head and Neck Surgery and Neurotology, with exceptional clinical skills in both general otology and skull base surgery. She is Assistant Professor of Clinical Otolaryngology-Head and Neck Surgery at the Keck School of Medicine of USC.

Regenerative Medicine Advisory Board Sheng Ding, Ph.D. Senior Investigator, Gladstone Institute of Cardiovascular Disease Robin Franklin, Ph.D. Professor of Stem Cell Medicine, Wellcome Trust-MRC Cambridge Stem Cell Institute Robert Langer, SC.D. David H. Koch Institute Professor at the Massachusetts Institute of Technology Amy Wagers, Ph.D. Forst Family Professor of Stem Cell and Regenerative Biology, Harvard University Lee Rubin, Ph.D. Professor and lead Investigator for the Rubin Laboratory, Harvard Stem Cell Institute Siddhartha Mukherjee, M.D., D.Phil. Assistant Professor of Medicine, Columbia University Medical Center Sean J. Morrison, Ph.D. Director of the Children's Medical Center Research Institute, UT Southwestern Jeff Karp, Ph.D. Associate Professor at Brigham and Women’s Hospital, Harvard Medical School

Frequency Therapeutics Corporate Overview May, 2020