Exhibit 99.1

Corporate Presentation January 2019

Legal disclaimers This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases, you can identify forward-looking statements by the words “will,” “expect,” “intend,” “plan,” “objective,” “believe,” “estimate,” “potential,” “continue” and “ongoing,” or the negative of these terms, or other comparable terminology intended to identify statements about the future. These statements are based on management’s current beliefs and expectations. These statements include but are not limited to statements regarding our business strategy, our plans to develop and commercialize our product candidates, our plans to enter into transactions with commercial or strategic partners, the safety and efficacy of our product candidates, our expectations regarding timing, design and results of clinical trials of our product candidates, our plans and expected timing with respect to regulatory filings and approvals, the size and growth potential of the markets for our product candidates, and our ability to serve those markets, and our plans and expected timing with respect to regulatory filings and approvals. These statements involve substantial known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking statements we make. Factors that may cause our actual results to vary from current expectations are discussed in our prospectus relating to our initial public offering and our other filings with the U.S. Securities and Exchange Commission, including the “Risk Factors” sections therein. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking statements. The forward-looking statements in this presentation represent our views as of the date of this presentation. We anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we have no current intention of doing so except to the extent required by applicable law. You should, therefore, not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this presentation. The trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of such products. 2

TLC’s mission to fulfill unmet medical needs presents a compelling investment opportunity Dual-listed (Nasdaq: TLC & TWO: 4152) Dedicated management team with significant experience in liposomal science Proprietary lipid technology platforms conceiving four lead product candidates in pain management, ophthalmology, and oncology: ï,§ BioSeizer™ sustained release technology with complete pharmacokinetic (PK) control designed for immediate onset and prolonged retention TLC5996-month dexamethasone intraarticular injection Pivotal trial 1H19 TLC59072-hour ropivacaine post-operative injection Phase I/II topline data readout 1Q19; Phase II initiated TLC399>6-month dexamethasone intravitreal injection Ph II trial ongoing ï,§ NanoX™ active drug loading to alter systemic exposure, reduce dosing frequency, and improve efficacy due to tissue-targeted delivery TLC178 liposomal vinorelbine Rare Pediatric Disease Designation and Orphan Drug Designation by FDA Phase I/II in adults ongoing Eligible for streamlined 505(b)(2) regulatory pathway for expedited approval Strong intellectual property: 114 patents worldwide - 50 issued / 64 applications 3

Experienced and dedicated management team with extensive drug developmentknow-how Name and Title Experience Prior Experience Keelung Hong, PhD >20 years research at UCSF Cancer Research Institute, biopharm consultant Founder, Chairman, CEOCo-inventor, NanoX George Yeh, MBA Joined TLC in 2002 CFO, Hermes Biosciences President AsiaWired Group, General Bank, George Spencer- Vice President, Biosimilars, C. Pfizer Inc. Green, MD Chairman and CEO, Abbott International, CMO USA Nicole Lin, MBA MA Labs Inc., NextGen Communications, CFO & Vice President Taiwan Securities Company Yunlong Tseng, PhD Founding member of R&D team; 19 Research and publications on liposome research DevelopmentCo-inventor, NanoX Wenji Chen, PhD, >25 years experience at GlaxoSmithKline MBA as head of Worldwide Business Corporate Development Development Hung Wei Chih, PhD >13 years at Genentech/Roche as head of CMC Product Quality & Development Sheue Fang Shih, Joined TLC in 2002 PhD >15 years drug development experience Product Development Inventor, BioSeizer Wendy Wu, PhD >16 years experience at Exelixis, Pfizer, Clinical Affairs Ionis, Prometheus, and Amylin; CCRP Carl Brown, PhD >15 years clinical development and medical research experience in biotech & Medical Sciences pharma at Twi Biotechnology, Amylin Cynthia Lin >19 years in pharma; >14 years in global Clinical Operations & clinical research management at Quintiles Development Terry Tai, MD >10 years regulatory science experience as Portfolio & Strategy medical reviewer and consultant at Taiwan Center for Drug Evaluation Bella Kuo >19 years pharma project management Quality Assurance experience at PharmaEngine, TTY Biopharm Board Member Affiliation Keelung Hong, PhD ChairmanHong-Jen Chang, MD Anupam Dalal, MD, MBA Del Mar May Kang, MBA Technologies Beatrice Liu, PhD Tom Chen, PhD Xiang Investment Chan Lee Company Moun-Rong Lin, MBA 4

Lipid-based drug delivery platforms designed to create innovative products Sustained Release 1995 Sequus/Alza/J&J Doxil Targeted Delivery 2011 Pacira Exparel 2015 Hermes/Merrimack/Ipsen Onivyde 2015 FDA Liposome Guidance ï,§ Controlled density of multi-layers for TLC599 immediate & sustained release TLC590 ï,§ Ability to deliver biologics or small molecules ï,§ Fully biodegradable components TLC399 ï,§ Sterile filtration at thenear-end stage instead of entirely aseptic process ï,§ Possibility for robust BioSeizerscale-up NanoX ï,§ More options for payload selection ï,§ Greater stability for longer shelf life ï,§ Prolonged circulation ï,§ Efficient particle size TLC178 for enhanced delivery to tumor site ï,§ Reduced dose frequency ï,§ Applied with >50 compounds ï,§ Robust, scalable & replicable manufacturing 5
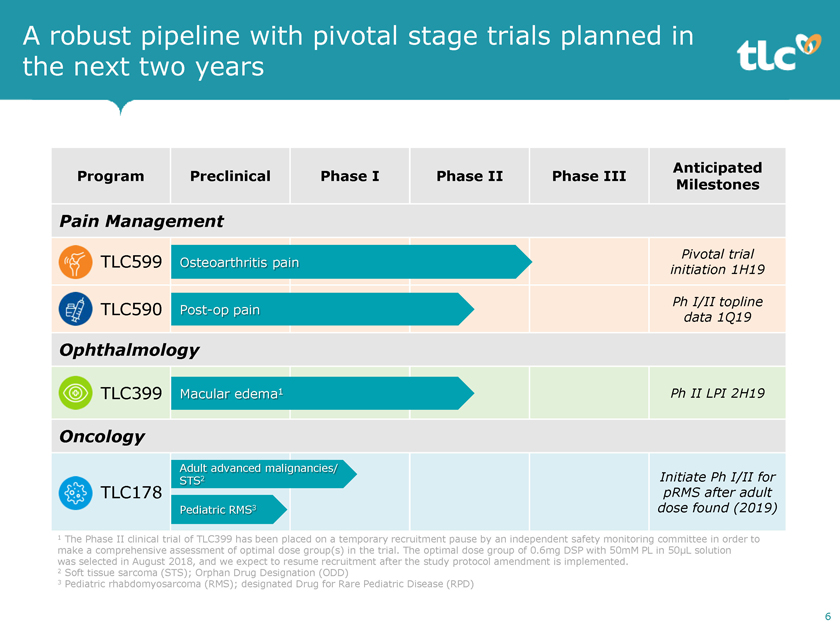
A robust pipeline with pivotal stage trials planned in the next two years Anticipated Program Preclinical Phase I Phase II Phase III Milestones Pain Management Pivotal trial TLC599 Osteoarthritis pain initiation 1H19 Ph I/II topline TLC590Post-op pain data 1Q19 Ophthalmology TLC399 Macular edema1 Ph II LPI 2H19 Oncology Adult advanced malignancies/ STS2 Initiate Ph I/II for TLC178 pRMS after adult Pediatric RMS3 dose found (2019) 1 The Phase II clinical trial of TLC399 has been placed on a temporary recruitment pause by an independent safety monitoring committee in order to make a comprehensive assessment of optimal dose group(s) in the trial. The optimal dose group of 0.6mg DSP with 50mM PL in 50μL solution was selected in August 2018, and we expect to resume recruitment after the study protocol amendment is implemented. 2 Soft tissue sarcoma (STS); Orphan Drug Designation (ODD) 3 Pediatric rhabdomyosarcoma (RMS); designated Drug for Rare Pediatric Disease (RPD) 6

Osteoarthritis (OA) Pain Program TLC599: BioSeizer sustained release dexamethasone sodium phosphate (DSP) intraarticular injection for OA pain

TLC599 target product profile Fast acting, long lasting, low toxicitynon-opioid intraarticular injection for OA Current treatment landscape for OA ï,§ Estimated 30.8 million OA patients in US1 ï,§ Estimated 20% of people >65 years will be at risk for OA by 20302 ï,§ knee Treatment OA include for moderate NSAIDs, degeneration steroids, viscosupplements (hyaluronic acid), and opioids TLC599 Our strategic solution: TLC599 ï,§ Designed for prolonged duration up to 24 weeks with pain relief within 3 days ï,§ Minimized cartilage damage and toxicity ï,§ Improved drug retention in joint ï,§ Flexibility of needle size to allow for future expanded indications (small joints) TLC599 development stage ï,§ Randomized, secondary endpoints double-blind, placebo-controlled Phase II trial complete; met primary and key ï,§ EOP2 meetings: CMC complete / clinical scheduled 1Q19 ï,§ Planned Pivotal trial initiation 1H19 1 Arthritis Foundation. Arthritis By the Numbers / Book of Trusted Facts & Figures. 2 National Institutes of Health. FACT SHEET – Osteoarthritis., 2010 3 Intra-articular steroid injections for painful knees. Can Fam Physician2004;50:241-248. 4State-of-the-Art management of knee osteoarthritis. World J 8 Clin Cases 2015; 3(2):89-101. 5 The chondrotoxicity of single-dose corticosteroids. Knee Surg Sports Traumatol Arthrosc. 2012Sep;20(9):1809-14.

In vitro study of human chondrocytes TLC599’s API dexamethasone caused the least cell death among steroids Dexamethasone, the API of TLC599, is 5 times more potent than TA, with cell death comparable to negative control (Dexamethasone) (TA, triamcinolone acetonide) Source: Dragoo et al. Knee Surg Sports Traumatol Arthrosc (2012) 20:1809–14. 9

Preclinical in vivo study – chondrotoxicity TLC599 is cartilage sparing compared to ER TA Proteoglycan Staining Study in Beagles Day 30 Darker color = more staining = more proteoglycan presence = less cartilage damage TA = triamcinolone acetonide / ER TA = extended release triamcinolone acetonide 10
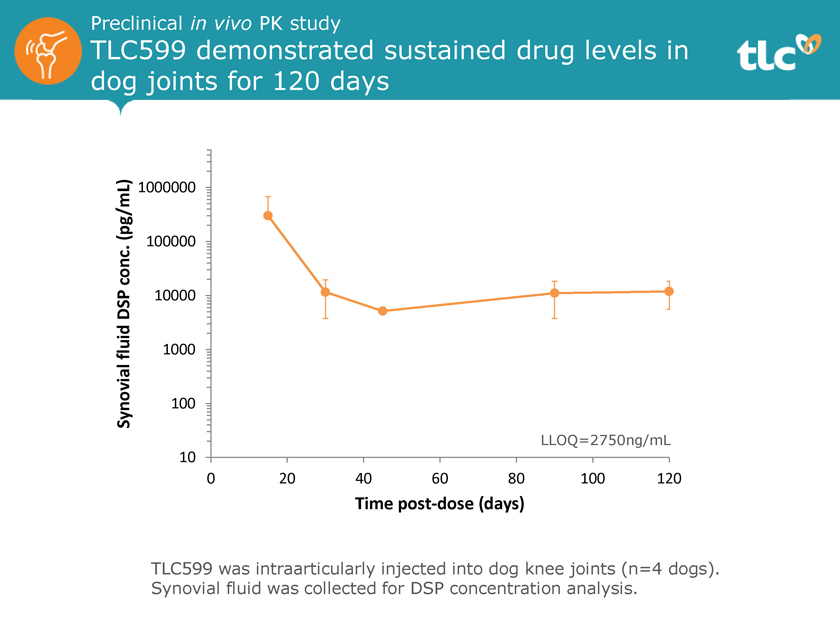
Preclinical in vivo PK study TLC599 demonstrated sustained drug levels in dog joints for 120 days (pg/mL) 1000000 conc. 100000 DSP 10000 fluid 1000 Synovial 100 LLOQ=2750ng/mL 10 0 20 40 60 80 100 120 Time post-dose (days) TLC599 was intraarticularly injected into dog knee joints (n=4 dogs). Synovial fluid was collected for DSP concentration analysis.
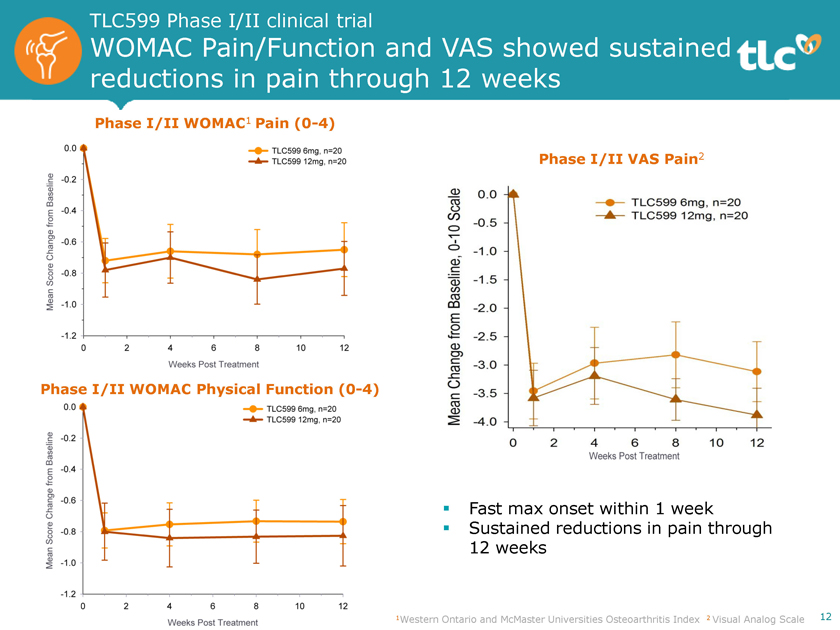
TLC599 Phase I/II clinical trial WOMAC Pain/Function and VAS showed sustained reductions in pain through 12 weeks Phase I/II WOMAC1 Pain(0-4) Phase I/II VAS Pain2 Phase I/II WOMAC Physical Function(0-4) ï,§ Fast max onset within 1 week ï,§ Sustained reductions in pain through 12 weeks 1Western Ontario and McMaster Universities Osteoarthritis Index 2 Visual Analog Scale 12
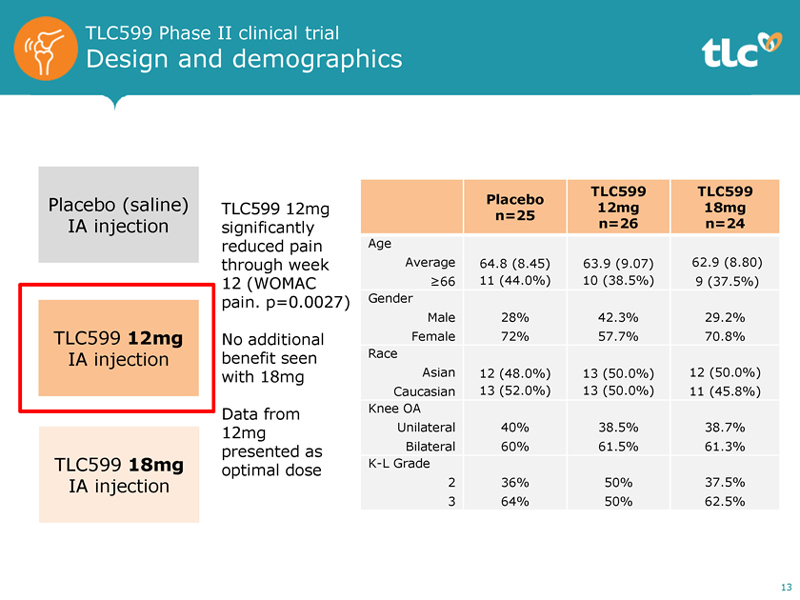
TLC599 Phase II clinical trial Design and demographics Placebo (saline) IA injection TLC599 12mg IA injection TLC599 18mg IA injection TLC599 12mg significantly reduced pain through week 12 (WOMAC pain. p=0.0027) No additional benefit seen with 18mg Data from 12mg presented as optimal dose TLC599 TLC599 Placebo 12mg 18mg n=25 n=26 n=24 Age Average 64.8 (8.45) 63.9 (9.07) 62.9 (8.80) 66 11 (44.0%) 10 (38.5%) 9 (37.5%) Gender Male 28% 42.3% 29.2% Female 72% 57.7% 70.8% Race Asian 12 (48.0%) 13 (50.0%) 12 (50.0%) Caucasian 13 (52.0%) 13 (50.0%) 11 (45.8%) Knee OA Unilateral 40% 38.5% 38.7% Bilateral 60% 61.5% 61.3% K-L Grade 2 36% 50% 37.5% 3 64% 50% 62.5% 13
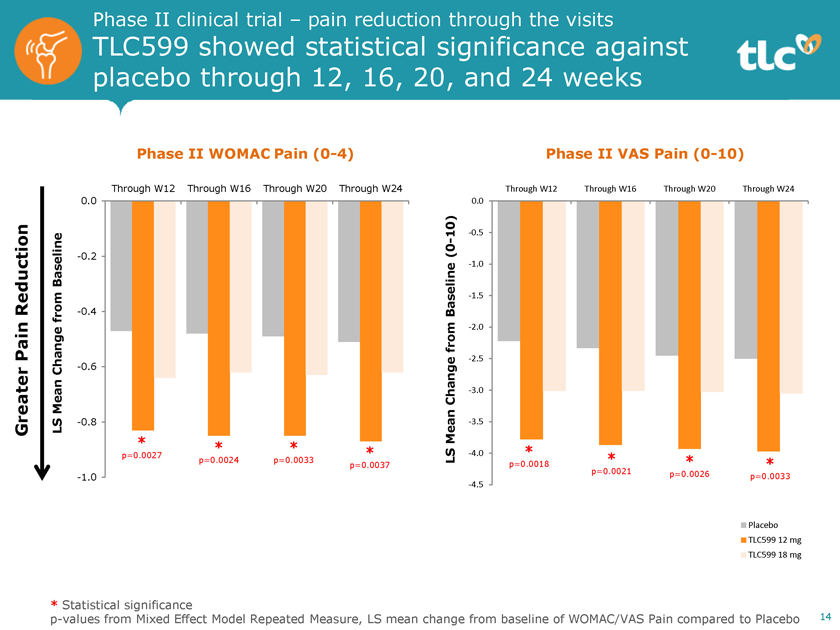
Phase II clinical trial – pain reduction through the visits TLC599 showed statistical significance against placebo through 12, 16, 20, and 24 weeks Phase II WOMAC Pain(0-4) Through W12 Through W16 Through W20 Through W24 0.0 Baseline -0.2 Reduction -0.4 from Pain Change -0.6 Mean Greater LS -0.8 * * * p=0.0027 * p=0.0024 p=0.0033 p=0.0037 -1.0 Phase II VAS Pain(0-10) Through W12 Through W16 Through W20 Through W24 0.0 10) -0.5 (0—-1.0 Baseline -1.5 from -2.0-2.5 Change -3.0 Mean -3.5 * LS -4.0 * * p=0.0018 * p=0.0021 p=0.0026 p=0.0033 -4.5 Placebo TLC599 12 mg TLC599 18 mg * Statistical significancep-values from Mixed Effect Model Repeated Measure, LS mean change from baseline of WOMAC/VAS Pain compared to Placebo 14

Phase II clinical trial – pain reduction at every visit TLC599 also showed statistical significance against placebo at every scheduled visit Phase II WOMAC Pain(0-4) 0.0 Placebo (n=25) -0.2 Baseline TLC599 12mg (n=26) -0.4 from Change-0.6-0.8 Mean * * * LS * -1.0 * * * -1.2 0 4 8 12 16 20 24 Phase II VAS Pain(0-10) 0.0 Placebo (n=25) -0.5 TLC599 12mg (n=26) Baseline-1.0 from -1.5-2.0 Change-2.5-3.0 Mean-3.5 LS * -4.0 * * * -4.5 * * * -5.0 0 4 8 12 16 20 24 Week * Statistical significancep-values from Mixed Effect Model Repeated Measure, LS mean change from baseline of WOMAC/VAS Pain compared to Placebo 15
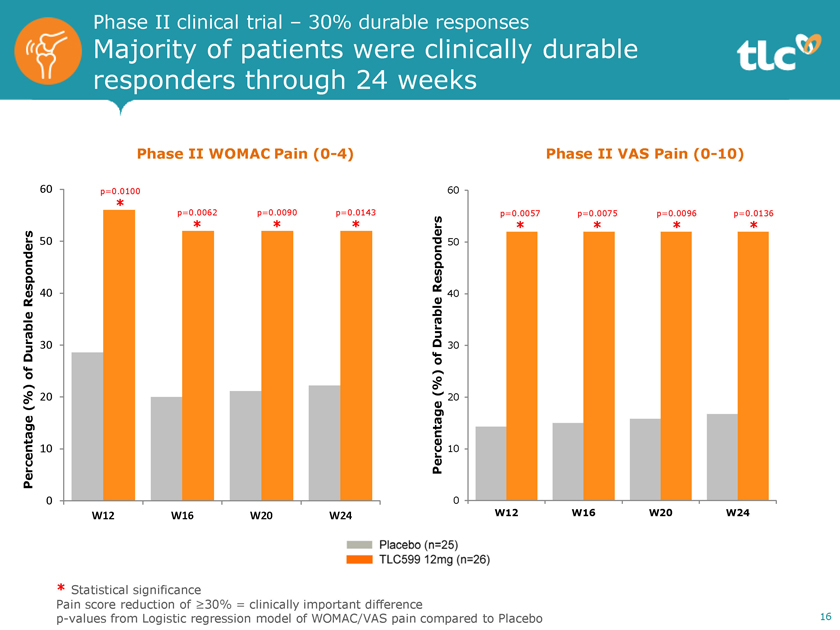
Phase II clinical trial – 30% durable responses Majority of patients were clinically durable responders through 24 weeks Phase II WOMAC Pain(0-4) 60 p=0.0100 * p=0.0062 p=0.0090 p=0.0143 * * * 50 Responders 40 Durable 30 of (%) 20 Percentage 10 0 W12 W16 W20 W24 Phase II VAS Pain(0-10) 60 p=0.0057 p=0.0075 p=0.0096 p=0.0136 * * * * Responders 50 Durable 40 30 of (%) 20 Percentage 10 0 W12 W16 W20 W24 * Statistical significance Pain score reduction of³30% = clinically important differencep-values from Logistic regression model of WOMAC/VAS pain compared to Placebo
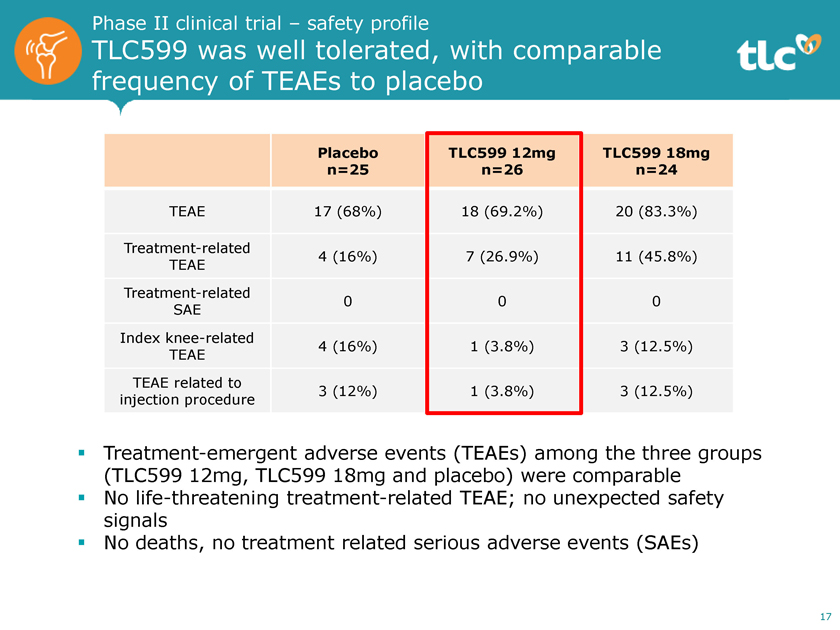
Phase II clinical trial – safety profile TLC599 was well tolerated, with comparable frequency of TEAEs to placebo Placebo TLC599 12mg TLC599 18mg n=25 n=26 n=24 TEAE 17 (68%) 18 (69.2%) 20 (83.3%) Treatment-related 4 (16%) 7 (26.9%) 11 (45.8%) TEAE Treatment-related 0 0 0 SAE Index knee-related 4 (16%) 1 (3.8%) 3 (12.5%) TEAE TEAE related to 3 (12%) 1 (3.8%) 3 (12.5%) injection procedure ï,§ Treatment-emergent adverse events (TEAEs) among the three groups (TLC599 12mg, TLC599 18mg and placebo) were comparable ï,§ No life-threatening treatment-related TEAE; no unexpected safety signals ï,§ No deaths, no treatment related serious adverse events (SAEs) 17

Phase II clinical trial Key takeaways  Immediate reductions in WOMAC and VAS Pain which were sustained • Placebo response generally similar to other trials, with no increase to baseline late in trial  TLC599 12mg was statistically significantly better than placebo for reductions in pain from Day 3 through Week 12 (met primary endpoint)  TLC599 12mg vs placebo, the following were statistically significant: • WOMAC and VAS Pain through Week 12, 16, 20, 24 • WOMAC and VAS Pain at Week 1, 4, 8, 12, 16, 20, 24 • WOMAC and VAS Pain durable response in majority of subjects (maintain >30% reduction) at each visit from Week 1 through Week 12, 16, 20, and 24  Clinically meaningful pain relief • Over half of subjects treated with TLC599 12mg maintained at least 30% reduction in pain throughout the6-month study (>2x as many as placebo) “I am impressed with TLC599’s ability to consistently provide fast and durable pain relief in the majority of patients for the entirefollow-up period of six months.” - Dr. David Hunter A principal investigator on our TLC599 Phase II trial 18

Market research study with16-week pain control TLC599 could achieve usage in 26% of all US knee OA patients TLC599 Optimal Profile Fast pain Better relief as >3mo WOMAC quickly as sustained Pain 1wk pain relief Reduction Safer21G- for repeat 30G dosing needle size Source: ZS Associates Market Study 19

Post-Surgical Pain Program TLC590: BioSeizer sustained release ropivacaine injection for post-operative pain management
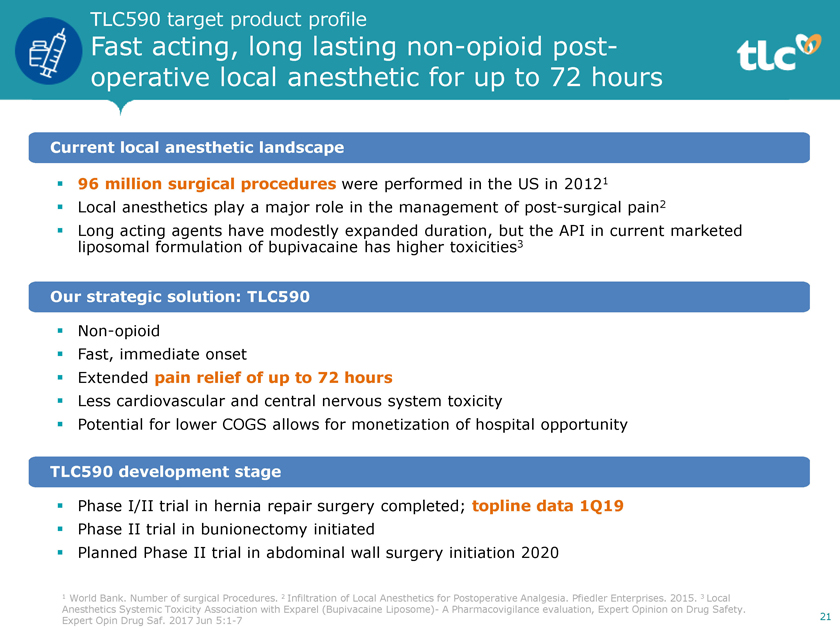
TLC590 target product profile Fast acting, long lastingnon-opioid postoperative local anesthetic for up to 72 hours Current local anesthetic landscape ï,§ 96 million surgical procedures were performed in the US in 20121 ï,§ Local anesthetics play a major role in the management of post-surgical pain2 ï,§ Long acting agents have modestly expanded duration, but the API in current marketed liposomal formulation of bupivacaine has higher toxicities3 Our strategic solution: TLC590 ï,§Non-opioid ï,§ Fast, immediate onset ï,§ Extended pain relief of up to 72 hours ï,§ Less cardiovascular and central nervous system toxicity ï,§ Potential for lower COGS allows for monetization of hospital opportunity TLC590 development stage ï,§ Phase I/II trial in hernia repair surgery completed; topline data 1Q19 ï,§ Phase II trial in bunionectomy initiated ï,§ Planned Phase II trial in abdominal wall surgery initiation 2020 1 World Bank. Number of surgical Procedures. 2 Infiltration of Local Anesthetics for Postoperative Analgesia. Pfiedler Enterprises. 2015. 3 Local Anesthetics Systemic Toxicity Association with Exparel (Bupivacaine Liposome)- A Pharmacovigilance evaluation, Expert Opinion on Drug Safety. Expert Opin Drug Saf. 2017 Jun5:1-7 21
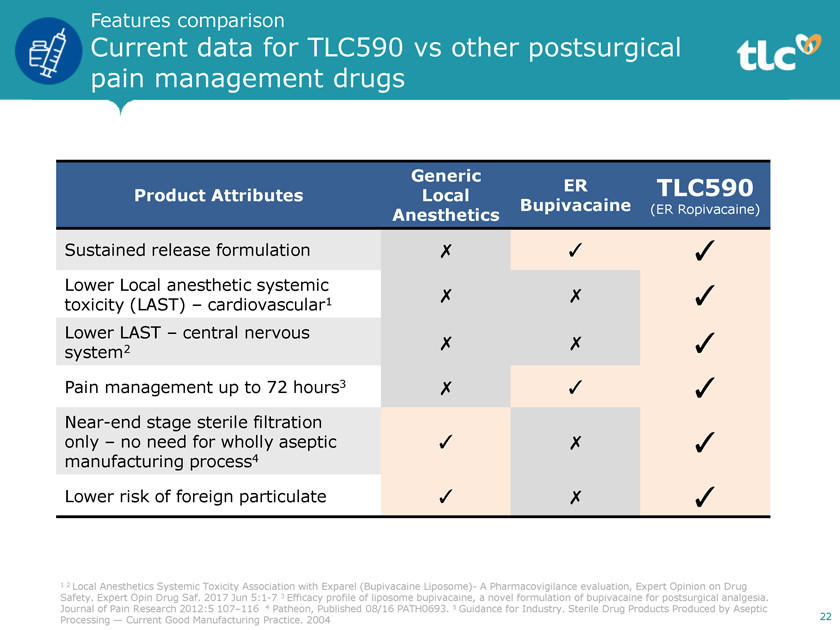
Features comparison Current data for TLC590 vs other postsurgical pain management drugs Generic ER TLC590 Product Attributes Local Bupivacaine (ER Ropivacaine) Anesthetics Sustained release formulation ✗ ✓ ✓ Lower Local anesthetic systemic toxicity (LAST) – cardiovascular1 ✗ ✗ ✓ Lower LAST – central nervous system2 ✗ ✗ ✓ Pain management up to 72 hours3 ✗ ✓ ✓Near-end stage sterile filtration only – no need for wholly aseptic ✓ ✗ ✓ manufacturing process4 Lower risk of foreign particulate ✓ ✗ ✓ 1 2 Local Anesthetics Systemic Toxicity Association with Exparel (Bupivacaine Liposome)- A Pharmacovigilance evaluation, Expert Opinion on Drug Safety. Expert Opin Drug Saf. 2017 Jun5:1-7 3 Efficacy profile of liposome bupivacaine, a novel formulation of bupivacaine for postsurgical analgesia. Journal of Pain Research 2012:5 107–116 4 Patheon, Published 08/16 PATH0693. 5 Guidance for Industry. Sterile Drug Products Produced by Aseptic Processing — Current Good Manufacturing Practice. 2004 22
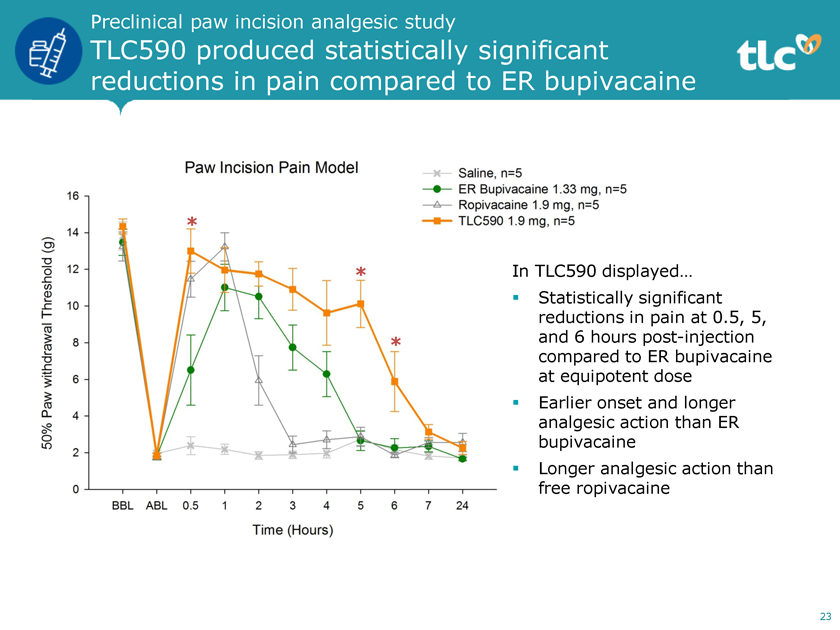
Preclinical paw incision analgesic study TLC590 produced statistically significant reductions in pain compared to ER bupivacaine * * In TLC590 displayed… ï,§ Statistically significant reductions in pain at 0.5, 5, * and 6 hours post-injection compared to ER bupivacaine at equipotent dose ï,§ Earlier onset and longer analgesic action than ER bupivacaine ï,§ Longer analgesic action than free ropivacaine 23

Preclinical nerve block study TLC590 exhibited more robust magnitude and duration of analgesia ï,§ TLC590 exhibited larger magnitude of analgesia which persisted to the 9th hour ï,§ Analgesic action of ER bupivacaine diminished and was comparable to saline by the 8th hour 24

Phase I/II clinical trial – inguinal hernia repair surgery Trial complete – topline data readout 1Q19 Phase I/II, randomized, double-blind, comparator-controlled, dose-escalation study of TLC590 compared with free ropivacaine via a single infiltrative local administration in adult subjects following inguinal hernia repair surgery. TLC590 190mg Primary objective: n=12 ï,§ Evaluate the safety and tolerability of TLC590 for postsurgical pain management in subjects following inguinal hernia repair surgery 150mg TLC590 380mg n=12 Secondary objectives: n=16ï,§ Evaluate PK profile and dose-exposure TLC590 475mg relationship and bioavailability compared to n=12 free ropivacaine Ropivacaineï,§ Evaluate analgesic efficacy compared with free ropivacaine TLC590 570mg ï,§ Evaluate exposure-response relationship n=12 between PK parameters and pain management  LPI end of Dec 2018  No SAEs 25

Phase II clinical trial – bunionectomy Design and objectives Phase 2, randomized, double-blind, comparator- and placebo-controlled study of TLC590 compared with free ropivacaine and placebo via a single infiltrative local administration in adult subjects following bunionectomy 152mg 190mg 228mg 50mg Part 1 TLC590 TLC590 TLC590 Ropivacaine (8mL) (10mL) (12mL) (10mL) n=12 n=12 n=12 n=12 50mg Saline Low dose High dose Ropivacaine Placebo Part 2 TLC590 TLC590 (10mL) (10mL) n=50 n=50 n=50 n=25 Primary Objective: ï,§ Evaluate the analgesic efficacy of TLC590 for postsurgical pain management in subjects following bunionectomy. Secondary Objectives: ï,§ Evaluate PK profile and dose-exposure relationship and bioavailability compared to free ropivacaine ï,§ Evaluate safety and tolerability of TLC590 ï,§ Evaluate the exposure-response relationship between PK parameters and pain intensity 26

Ophthalmic Disease Program TLC399: BioSeizer sustained release dexamethasone sodium phosphate (DSP) intravitreal injection for macular edema (ME) due to retinal vein occlusion (RVO)
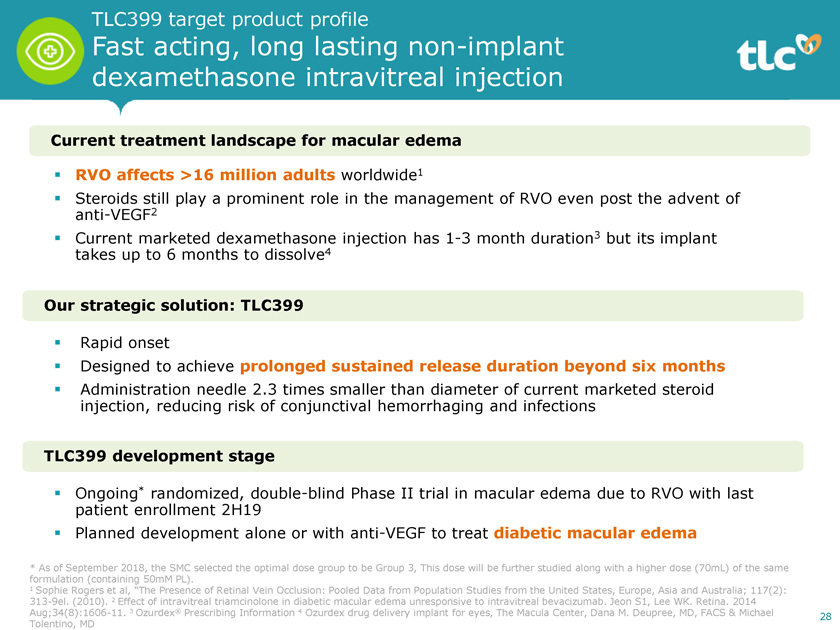
TLC399 target product profile Fast acting, long lastingnon-implant dexamethasone intravitreal injection Current treatment landscape for macular edema ï,§ RVO affects >16 million adults worldwide1 ï,§ Steroids still play a prominent role in the management of RVO even post the advent of anti-VEGF2 ï,§ Current marketed dexamethasone injection has1-3 month duration3 but its implant takes up to 6 months to dissolve4 Our strategic solution: TLC399 ï,§ Rapid onset ï,§ Designed to achieve prolonged sustained release duration beyond six months ï,§ injection, Administration reducing needle risk 2.3 of conjunctival times smaller hemorrhaging than diameter and of infections current marketed steroid TLC399 development stage ï,§ Ongoing* randomized, double-blind Phase II trial in macular edema due to RVO with last patient enrollment 2H19 ï,§ Planned development alone or with anti-VEGF to treat diabetic macular edema * As of September 2018, the SMC selected the optimal dose group to be Group 3, This dose will be further studied along with a higher dose (70mL) of the same formulation (containing 50mM PL). 1 Sophie Rogers et al, “The Presence of Retinal Vein Occlusion: Pooled Data from Population Studies from the United States, Europe, Asia and Australia; 117(2):313-9el. (2010). 2 Effect of intravitreal triamcinolone in diabetic macular edema unresponsive to intravitreal bevacizumab. Jeon S1, Lee WK. Retina. 2014Aug;34(8):1606-11. 3 Ozurdex® Prescribing Information 4 Ozurdex drug delivery implant for eyes, The Macula Center, Dana M. Deupree, MD, FACS & Michael 28 Tolentino, MD
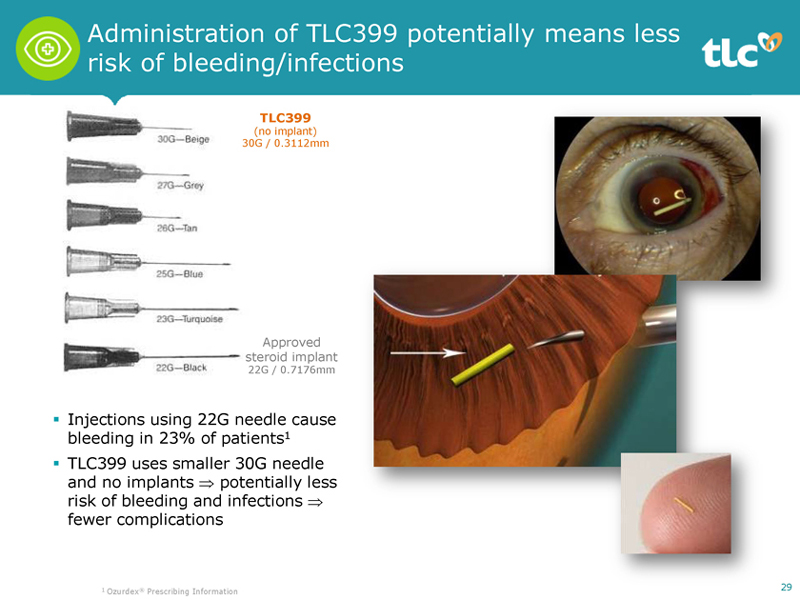
Administration of TLC399 potentially means less risk of bleeding/infections TLC399 (no implant) 30G / 0.3112mm Approved steroid implant 22G / 0.7176mm ï,§ Injections using 22G needle cause bleeding in 23% of patients1 ï,§ TLC399 uses smaller 30G needle and no implants ⇒ potentially less risk of bleeding and infections ⇒ fewer complications 1 Ozurdex® Prescribing Information 29

Preclinical study Dexamethasone levels of TLC399 in the eye maintained for >200 days TLC399 Rabbit Vitreous Dex Implant Rabbit Vitreous Maintained effective to Day 200 Concentration below LoQ by Day 35 Source: Ozurdex presentation When injected into eyes of animals… ï,§ DSP was undetectable within one day ï,§ Dexamethasone implants sustained therapeutic levels for ~40 days before reaching limit of quantitation (LoQ=2.5ng/mL) ï,§ TLC399 maintained therapeutic levels over a period of >200 days 30 *Nohead-to-head trials have been performed between TLC399 and dex implant

Phase I clinical trial Decrease in CST up to 12 months after single injection (0.6mg DSP) Day 0 Day 90 Day 180 Day 360 CST* 386 ìm 253 ìm 264 ìm 206 ìm * Central Subfield Thickness ï,§ Improved/stabilized vision for 6 to 12 months ï,§ Improved optical coherence tomography (OCT) results for 6 to 12 months 31

Phase II clinical trial Updated TLC399 trial design with optimized formulation Patient Population Inclusion criteria: ï,§ M/F, >18 years old ï,§ Macular edema due to CRVO or BRVO ï,§ BCVA score 20/40 to 20/400 ï,§ CST³350 um ScreeningDay-14 toDay-1 (n=66) Randomization Treatment Visit Groups Structure Part 1 TLC399 Group 1 0.36mg DSP/100mM PL 30µL (n=10) TLC399 Group 2 0.6mg DSP/100mM PL 50µL (n=10) Part 2 50µL (n=15) TLC399 Group 3 0.6mg DSP/50mM PL Day 0 2W 1M 2M 3M 4.5M 6M 7.5M 9M 12M (n=11) 70µL (n=15) 32
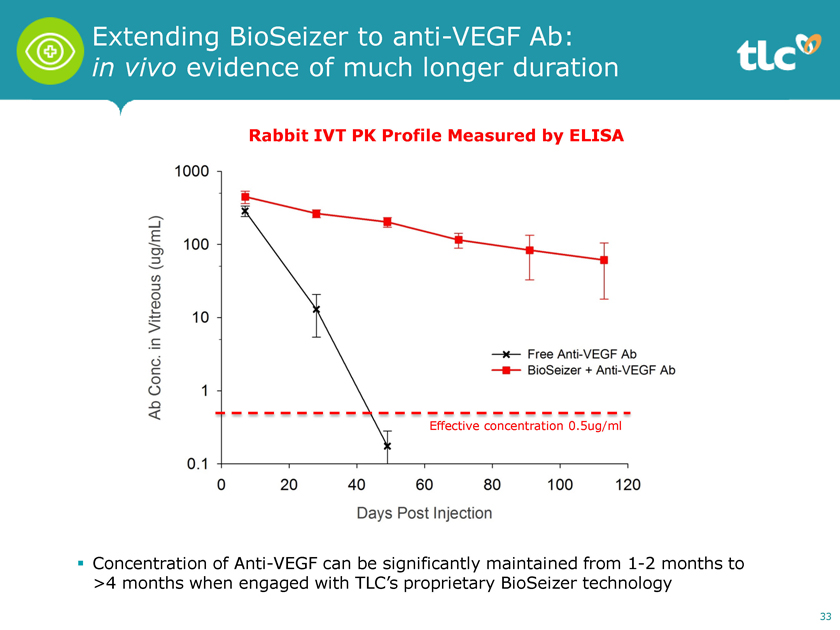
Extending BioSeizer to anti-VEGF Ab: in vivo evidence of much longer duration Rabbit IVT PK Profile Measured by ELISA Effective concentration 0.5ug/ml ï,§ Concentration of Anti-VEGF can be significantly maintained from1-2 months to >4 months when engaged with TLC’s proprietary BioSeizer technology 33

Soft Tissue Sarcoma Program TLC178: NanoX tumor-concentrated delivery of vinorelbine for rhabdomyosarcoma (RMS) and potentially for soft tissue sarcomas (STS) andnon-small cell lung carcinoma (NSCLC)

TLC178 target product profile Safer, less toxic, more durable anticancer drug with RPD and ODD designations Current treatment landscape in cancers ï,§ Vinorelbine Guidelines as (VNB) therapy is listed agent by with the activity National in Comprehensive RMS in combination Cancer with Network cyclophosphamide, (NCCN) or as a single agent only for palliative therapy1, but with significant dose limiting myelosuppression2 3 ï,§ Vinorelbine and gemcitabine (Gem) combo is active regimen in STS and NSCLC4 5 Our strategic solution: TLC178 ï,§ Improve selective delivery to tumor versusnon-tumor tissue ï,§ Higher drug concentration at tumor confers higher activity ï,§ Less drug tonon-tumor reduces myelosuppression, enabling higher dose intensity ï,§ Efficacy improvement in treatment response rate and duration of response TLC178 development stage ï,§ Ongoing Phase I/II dose-escalation trial in adults ï,§ IND cleared for pediatric RMS (US FDA Rare Pediatric Disease Designation, RPD) ï,§ Phase I/II trial in pediatric RMS initiation once suitable dose in adults established ï,§ Planned Designation, further ODD) expansion and NSCLC in gemcitabine combo into STS (US FDA and EU Orphan Drug 1 National Comprehensive Cancer Network, NCCN Clinical Practice Guidelines in Oncology – Soft Tissue Sarcoma, Version 1.2018, October 31, 2017. 2 Phase II Evaluation of Intravenous Vinorelbine (Navelbine) in Recurrent or Refractory Pediatric Malignancies: A Children’s Oncology Group Study. Pediatric Blood Cancer. 2009 October ; 53(4): 590–93. 3 Vinorelbine in Previously Treated Advanced Childhood Sarcomas .Cancer 2002;94:3263–68. 4 Gemcitabine and Vinorelbine Combination Chemotherapy for Patients With Advanced Soft Tissue Sarcomas. Cancer2007;109:1863-69. 5 The Novel and EffectiveNon-platinum, Nontaxane Combination of 35 Gemcitabine and Vinorelbine in AdvancedNon-small Cell Lung Carcinoma. Cancer2002;95(2)340-53.

Rhabdomyosarcoma (RMS) background RMS: rare, aggressive, most common soft tissue cancer in kids with about 350 cases per year Incidence Annually 350 new cases in the US1 Cure rate 70% for multimodality treatment2 Vincristine, vinorelbine (although not approved by FDA for Common practice use in RMS) and cyclophosphamide regimen3 The remaining 30% RMS patients will suffer from relapsed Unsatisfactory treatment or refractory RMS with only 20% of5-year survival rate; options 4 most relapsed patients die in the first year Neither established guideline nor meaningful improved Guideline 5 6 7 therapy in relapsed or refractory RMS within 30 years 1 “What Are the Key Statistics About Rhabdomyosarcoma?” American Cancer Society. 2 Survival After Relapse in Children and Adolescents With Rhabdomyosarcoma: A Report From the Intergroup Rhabdomyosarcoma Study Group. Journal of Clinical Oncology, Vol 17, No 11, 1999: pp 3487-3493. 3 Relapse After Localized Rhabdomyosarcoma: Evaluation of the Efficacy of Second-Line Chemotherapy. Pediatr Blood Cancer 2015;62:1935–1941. 4 Survival after relapse in children and adolescents with rhabdomyosarcoma: A report from the Intergroup Rhabdomyosarcoma Study Group. Journal of Clinical Oncology, Vol 17, No 11, 1999: pp 3487-3493. 5 Relapse After Localized Rhabdomyosarcoma: Evaluation of the Efficacy of Second-Line Chemotherapy. Pediatr Blood Cancer 2015;62:1935–1941. 6 Treatment of Nonmetastatic Rhabdomyosarcoma in Childhood and Adolescence: Third Study of the International Society of Paediatric Oncology—SIOP Malignant Mesenchymal Tumor 89. Journal of Clinical Oncology. Vol. 23, No 12, 2005: pp 2618-2628. 7 Long-Term Medical Effects of Childhood and Adolescent Rhabdomyosarcoma: A Report From the 36 Childhood Cancer Survivor Study. Pediatr Blood Cancer 2005;44:643–653.

Preclinical study – fibrosarcoma model TLC178 showed significant tumor inhibition response compared to doxorubicin (p<0.05) Antitumor Efficacy of TLC178 in Fibrosarcoma Model 2000 Placebo (D5W) Doxorubicin 3.4mg/kg TLC178 5mg/kg 1500 3 ) (mm volume 1000 Tumor 500 0 0 5 10 15 20 25 30 Post drug injection day 37

Preclinical study – RMS model TLC178 demonstrated more effective control of tumor growth than free vinorelbine Antitumor Efficacy of TLC178 in RMS Model Saline 3000 Vinorelbine 5mg/kg TLC178 5mg/kg TLC178 superior to vinorelbine (p<0.05) from day 18 onwards 2500 3 ) Compared to free vinorelbine, (mm 2000 TLC178 potentially has… ï,§ Better pharmacokinetics volume 1500 Reduced myelosuppressive side- ï,§ Tumor effects 1000ï,§ Longer dosing intervals ï,§ Higher vinorelbine concentration at neovascular-rich and 500 subcutaneous tumor sites ï,§ Capability to broaden indications 0 0 10 20 30 40 Post drug injection day 38
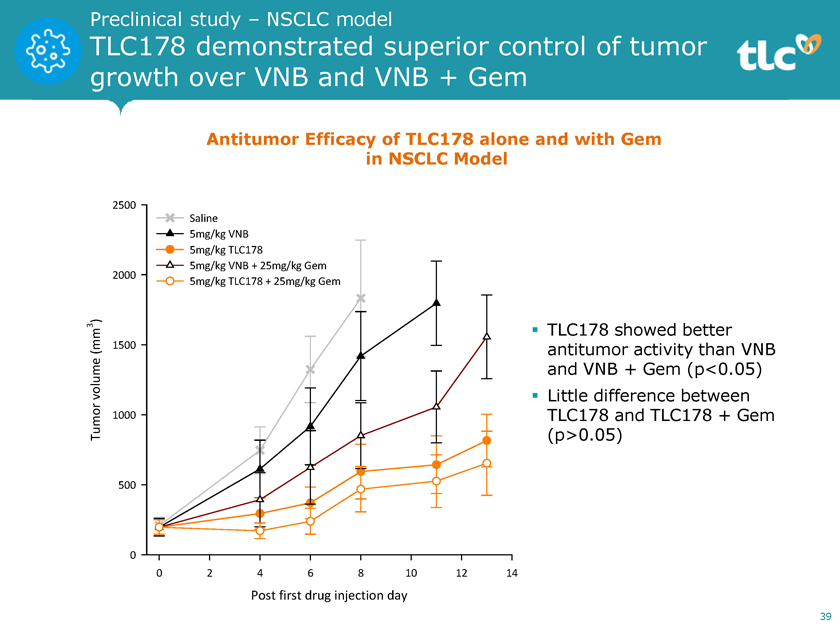
Preclinical study – NSCLC model TLC178 demonstrated superior control of tumor growth over VNB and VNB + Gem Antitumor Efficacy of TLC178 alone and with Gem in NSCLC Model 2500 Saline 5mg/kg VNB 5mg/kg TLC178 5mg/kg VNB + 25mg/kg Gem 2000 5mg/kg TLC178 + 25mg/kg Gem 3 )ï,§ TLC178 showed better (mm 1500 antitumor activity than VNB volume and VNB + Gem (p<0.05) ï,§ Little difference between 1000 TLC178 and TLC178 + Gem Tumor (p>0.05) 500 0 0 2 4 6 8 10 12 14 Post first drug injection day 39

Clinical development plan for TLC178 in pRMS with extended indications in STS and NSCLC 3+3 Dose Escalation Mono TLC178 MTD(n=30-50) Current trial status: ,§ N=23 (current dose: 34mg/m2) ï,§ 1 treatment-related hematological toxicity (neutropenia) which was not a DLT Pediatric Rhabdomyosarcoma (pRMS) Combo TLC178+ CyclophosphamideSingle-arm NDA (N=40, ORR/ DoR)Non-Small Cell Lung Cancer (NSCLC) Soft Tissue Sarcoma (STS) NSCLC Combo TLC178+Gem Phase II/III Adaptive Random STS sNDA Combo TLC178+GemSingle-arm Phase II(n=100-200) 40 * Anemia that was not related to the trial has been observed in three patients.

TLC Summary

Anticipated short- and long-term milestones All programs expected to be in late or pivotal stage within two years Phase TLC599 II PK topline data TLC599 EOP2 meeting (CMC) TLC590 (bunionectomy) Phase II topline data TLC599 Fast track designation TLC399 Pivotal TLC599 trial topline data submission Phase II last patient enrollment 1H19 2H19 1H20 EOP2 TLC599 meeting (clinical) Special TLC599 protocol assessment submission Pivotal TLC599 trial initiation Phase TLC590 I/II topline data (hernia repair) TLC178 EU orphan designation Pivotal TLC590 trial initiation TLC590 Phase II initiation (abdominal wall surgery) Phase TLC178 I/II trial Part 2 – pivotal initiation TLC178 Phase I/II initiation (pediatric RMS) after suitable dose found in adults 42

TLC—Key takeaways  Experienced management team ï,§ CEO Dr. Keelung Hong’s third liposome company; President George Yeh’s second ï,§ Over 150 years of experience on management team  Robust and diverse pipeline ï,§ Programs targeting areas of unmet need in pain management (TLC599 & TLC590), ophthalmology (TLC399) and oncology (TLC178) in their late or pivotal stage within the next two years  Strong global IP protection ï,§ 50 granted patents worldwide ï,§ 64 patent applications under review  Global presence and partners ï,§ 8 offices worldwide ï,§ Partnerships with Sandoz, Pfizer (Hospira), Evonik, Baxter, Asian big pharmas  Distinguished reputation—Top 5% in corporate governance ï,§ Listed on Nasdaq (TLC) and Taipei Exchange (TT:4152) ï,§ Ranked top 5% five years running in corporate governance evaluation among all Taiwan-listed companies 43

Thank YouDelivering Hope for life