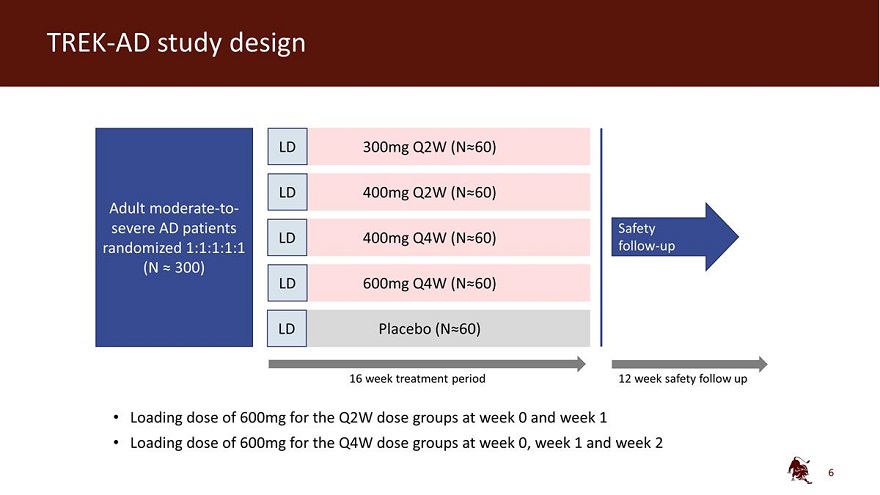
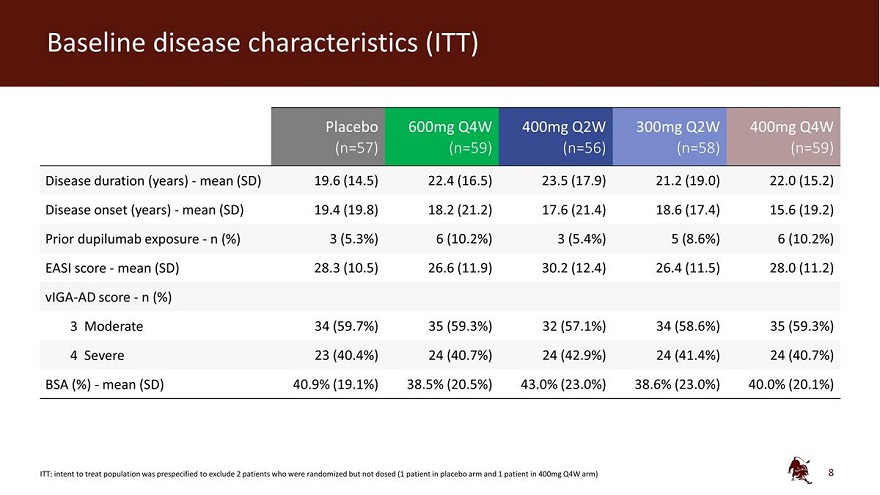
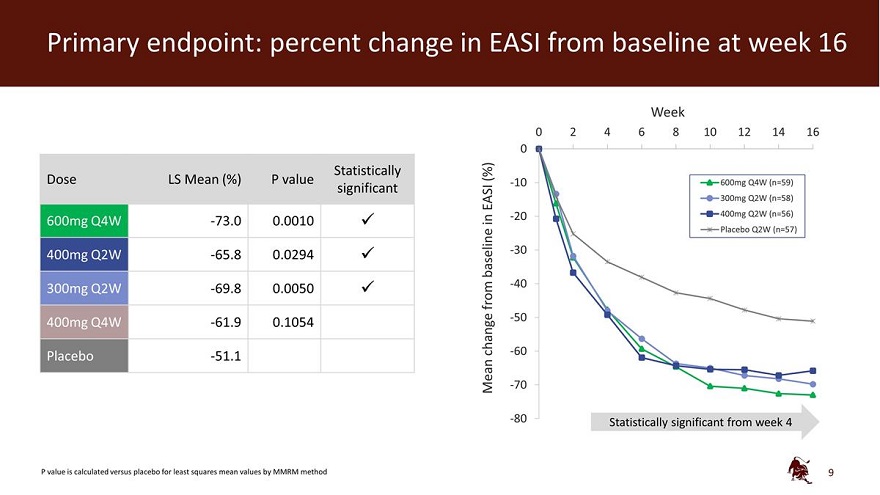
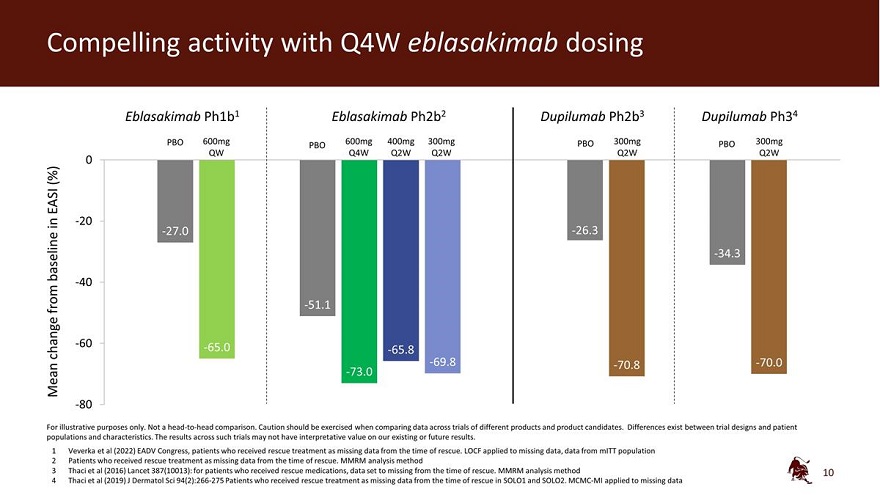
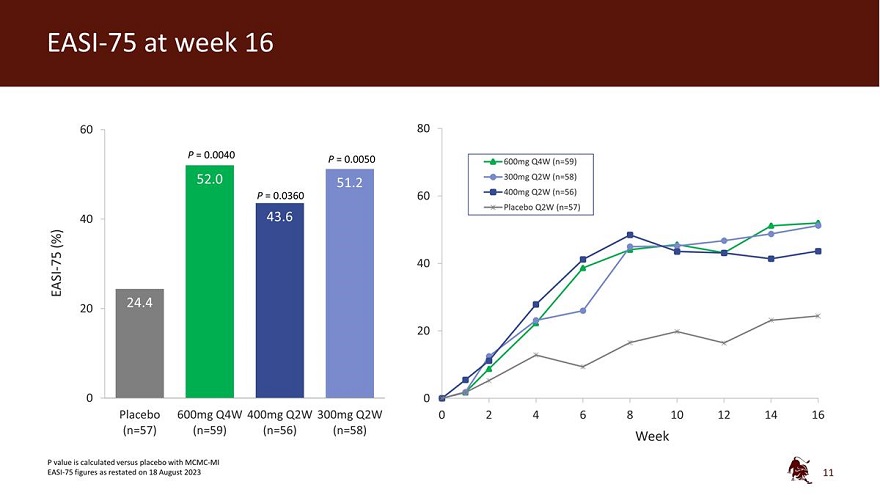
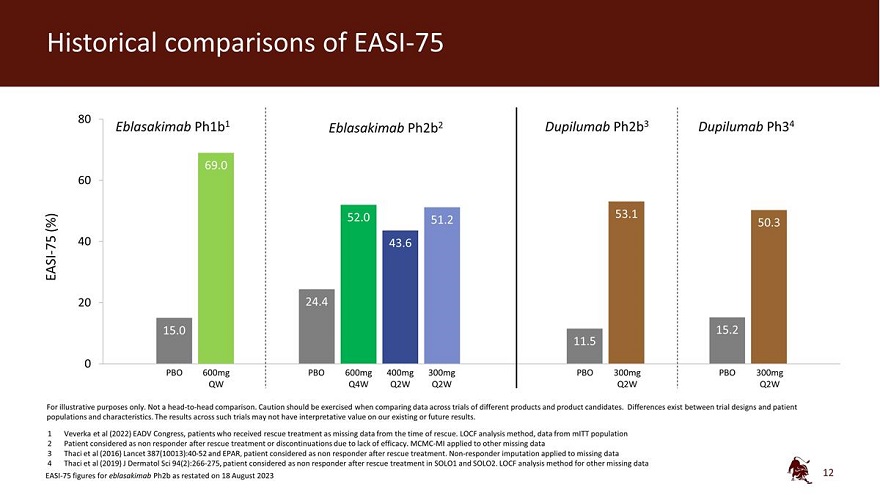
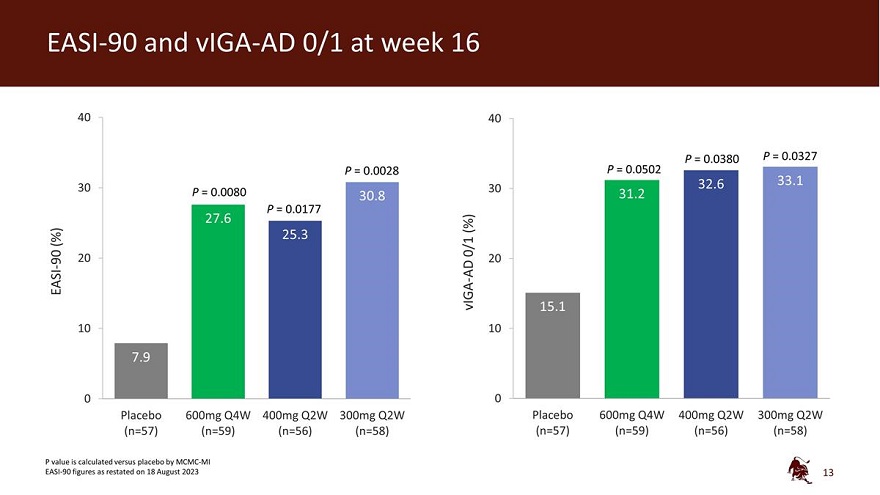
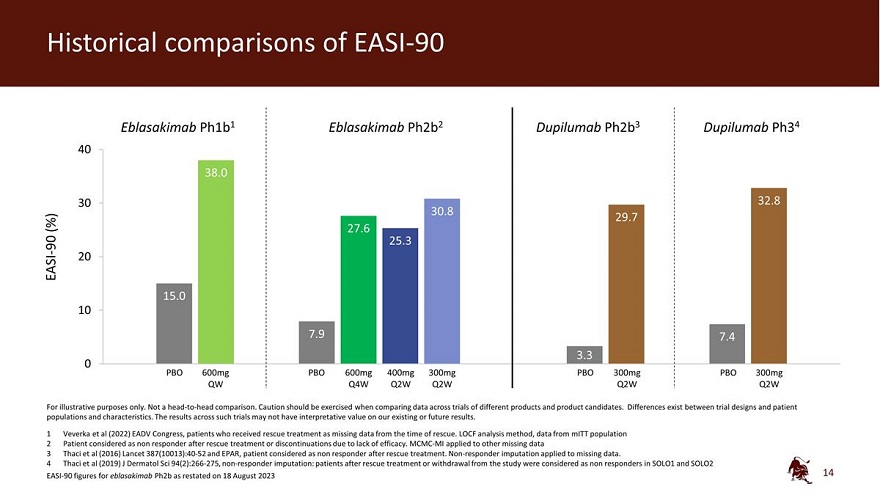
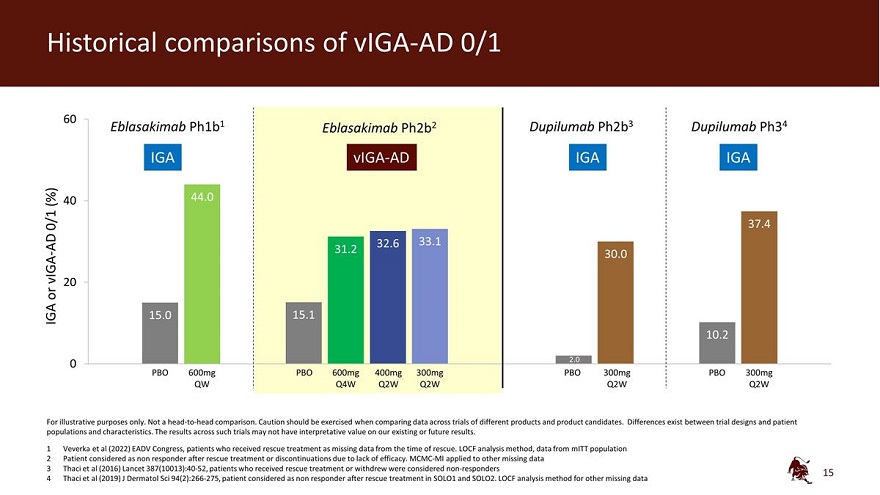
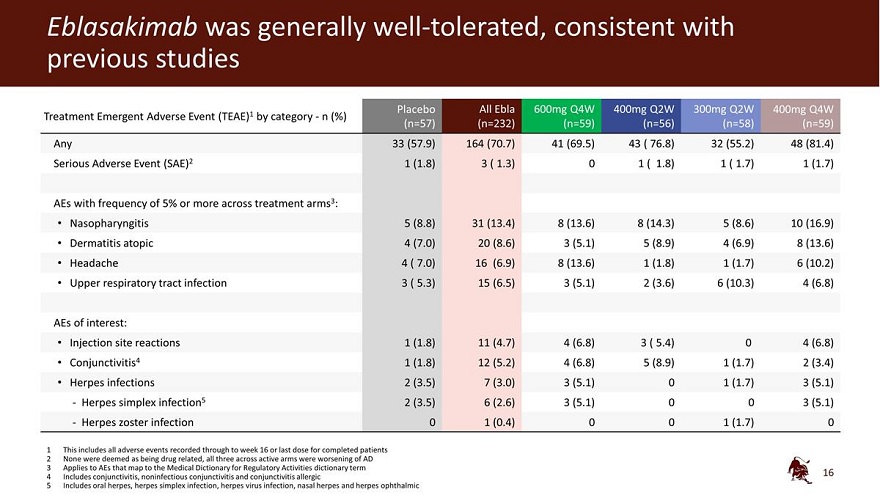
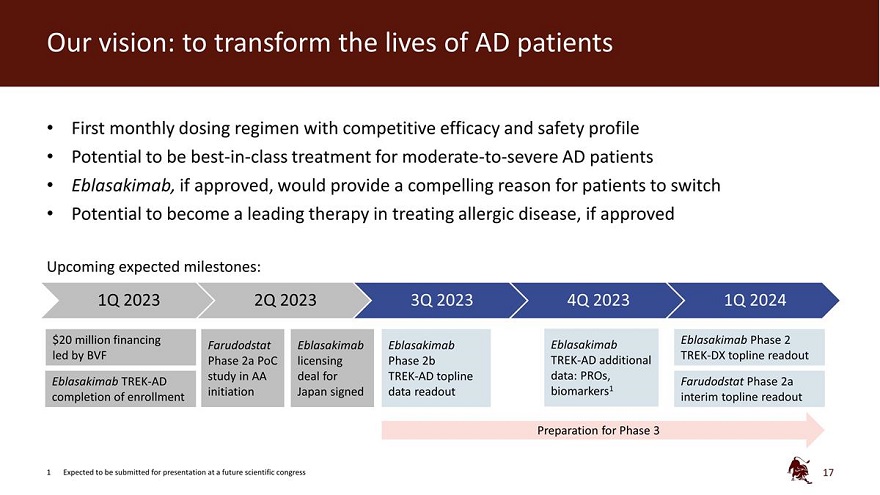
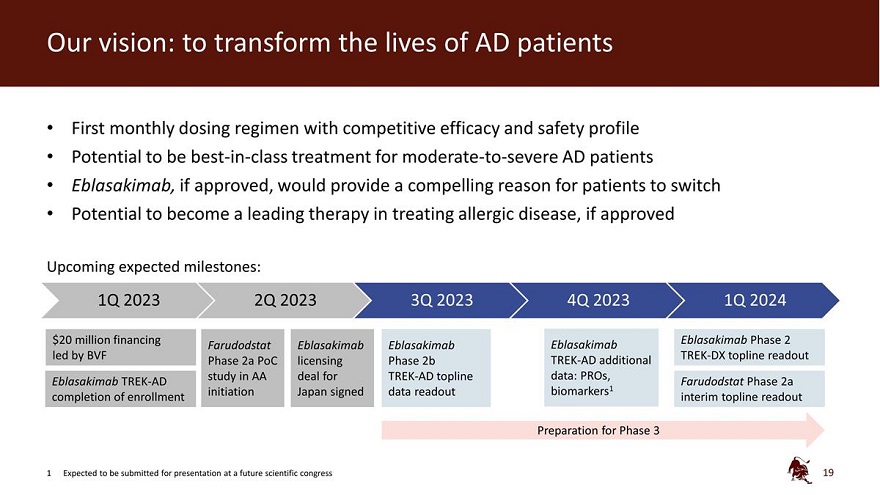
Legal disclaimer
This presentation contains forward-looking statements. These statements are based on the current beliefs and expectations of the management of ASLAN Pharmaceuticals Limited (the “Company”). These forward-looking statements may include, but are not limited to, statements regarding the
Company’s business strategy, the Company’s plans to develop and commercialize its product candidates, the safety and efficacy of the Company’s product candidates, including their potential to be best-in-class, the Company’s plans and expected timing with respect to clinical trials, clinical trial enrolment and clinical trial results for its product candidates, the Company’s plans and expected timing with respect to regulatory filings and approvals, the size and growth potential of the markets for the Company’s product candidates, and the potential for eblasakimab as a treatment for atopic dermatitis. The Company’s estimates, projections and other forward-looking statements are based on management’s current assumptions and expectations of future events and trends, which affect or may affect the Company’s business, strategy, operations or financial performance, and inherently involve significant known and unknown risks and uncertainties, which include, unexpected safety or efficacy data observed during preclinical or clinical studies; the fact that results of earlier studies and trials may not be predictive of future trial results; clinical site activation rates or clinical trial enrolment rates that are lower than expected; the impact of the COVID-19 pandemic on the Company’s business and the global economy; general market conditions; changes in the competitive landscape; and the Company’s ability to obtain sufficient financing to fund its strategic and clinical development plans. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of these risks and uncertainties, which include, without limitation the risk factors described in the Company’s US Securities and Exchange Commission filings and reports (Commission File No. 001-38475), including the Company’s Form 20-F filed with the U.S. Securities and Exchange Commission (the “SEC”) on March 24, 2023. This presentation discusses product candidates that are under clinical study, and which have not yet been approved for marketing by the US Food and Drug Administration. No representation is made as to the safety or effectiveness of these product candidates for the use for which such product candidates are being studied. Caution should be exercised when comparing data across trials of different products and product candidates. Differences existing between trial designs and patient populations and characteristics. The results across such trials may not have interpretative value on our existing or future results. All statements other than statements of historical fact are forward-looking statements. The words “believe,” “view,” “may,” “might,” “could,” “will,” “aim,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “plan,” or the negative of those terms, and similar expressions that convey uncertainty of future events or outcomes are intended to identify estimates, projections and other forward-looking statements. Estimates, projections and other forward-looking statements speak only as of the date they were made, and, except to the extent required by law, the Company undertakes no obligation to update or review any estimate, projection or forward-looking statement. 2