State of Israel | 2000 | Not Applicable | ||
(State or Other Jurisdiction of Incorporation or Organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification No.) |
Gary Emmanuel, Esq. David Huberman, Esq. Greenberg Traurig, P.A. One Azrieli Center Round Tower, 30th floor 132 Menachem Begin Rd Tel Aviv 6701101 Tel: +972 3-636-6000 | Shachar Hadar, Adv. Matthew Rudolph, Adv. Meitar | Law Offices 16 Abba Hillel Silver Rd. Ramat Gan 5250608, Israel Tel: +972-3-610-3100 | Ron Ben-Bassat, Esq. Eric Victorson, Esq. Sullivan & Worcester LLP 1633 Broadway New York, NY 10019 Tel: 212-660-3000 | Ronen Kantor, Esq. Doron Tikotzky Kantor Gutman & Amit Gross B.S.R. 4 Tower, 33 Floor 7 Metsada Street, Bnei Brak 5126112 Israel +972 3 613 3371 |
| PRELIMINARY PROSPECTUS | SUBJECT TO COMPLETION | DATED JANUARY 5, 2023 |

Per ADS(1) | Per Pre-Funded Warrant(1) | Total | ||||||||||
| Public offering price | $ | $ | ||||||||||
Underwriting discounts and commissions (2) | $ | $ | ||||||||||
| Proceeds to us (before expenses) | $ | $ | ||||||||||
| (1) | The public offering price and underwriting discounts and commissions correspond to a public offering price per ADS of $1.43, a public offering price per warrant of $0.01 and a public offering price per pre-funded warrant of $1.4299. |
| (2) | For a description of the additional compensation to be received by the underwriters, see “Underwriting” beginning on page 122 for additional information regarding the underwriters’ compensation. |
Sole Book-Running Manager |
| A.G.P. |
Lead Manager |
| Maxim Group LLC |
| 1 | |
| 11 | |
| 40 | |
| 41 | |
| 42 | |
| 43 | |
| 44 | |
| 45 | |
| 55 | |
| 75 | |
| 95 | |
| 97 | |
| 98 | |
| 105 | |
| 114 | |
| 115 | |
| 123 | |
| 128 | |
| 129 | |
| 129 | |
| 129 | |
| 130 | |
| F - 1 |
INDUSTRY AND MARKET DATA
| • | Perfect the development of our cultivated meat manufacturing technology and processes. We intend to continue developing and refining our processes, procedures and equipment until we are in a position to commercialize our technologies, whether by manufacturing final products for consumers (B2C and B2B2C models) or ingredients for industrial use, as well as in outlicensing (B2B models). We are continuing to tackle the technological challenges involved in scaling up both our biological and printing processes to industrial-scale levels. |
| • | Commercialize our technologies for use in consumer and business markets. We intend to provide ingredients to business customers for use in consumer products in order to help meet the growing demand for sustainable, slaughter-free cultivated meat products. For example, manufacturers of meat alternatives, such as vegetarian sausages, may choose to include our cultivated fat biomass in their products in order to deliver the signature meaty flavors, aromas and textures of the meat that is otherwise provided by the conventional meat of species such as chicken, beef and pork. We believe that this combination has the potential to unlock a new level of meat experience. |
| • | Develop additional alternative proteins to meet growing industry demand. There are substantial technological challenges inherent in expanding our offering beyond our current cultivated beef, pork and avian technologies to additional alternative proteins and cell lines. However, we believe that our experience, know-how and intellectual property portfolio form an excellent basis from which to surmount such challenges. Our first step in this direction was the 2021 acquisition of Peace of Meat BV, or Peace of Meat, with the aim of developing avian fat for the alternative meat industry by applying proprietary technology to mimic the cellular composition of conventional poultry. In addition, in January 2023, we announced a collaboration with Singaporean cultivated seafood developer, Umami Meats Pte Ltd., or Umami Meats, to develop 3D-printed structured eel and grouper products pursuant to a grant from the Singapore-Israel R&D Foundation. |
| • | Acquire synergistic and complementary technologies and assets. We intend to optimize our processes and diversify our product range to expand the cultivated meat technologies upon which marketable products can be based. We intend to accomplish this through a combination of internal development, acquisitions and collaborations, with a view to complementing our own processes and diversifying our product range along the cultivated meat production value chain in order to introduce cultivated products to the global market as quickly as possible. See also “- Additional Technologies” below. |
| • | We expect to continue incurring significant losses for the foreseeable future. |
| • | We will require substantial additional funds to complete our research and development activities. |
| • | There is substantial doubt as to whether we can continue as a going concern. |
| • | Raising additional capital may cause dilution to our existing shareholders or restrict our operations. |
| • | We have a limited operating history to date. |
| • | The research and development associated with cultured meat manufacturing is a lengthy process. |
| • | We intend to engage in future acquisitions, joint ventures or collaborations, which may not be successful. |
| • | We may not be able to successfully manage our planned growth, and if the market does not grow as we expect, we may not achieve sustainable revenues. |
| • | Business or economic disruptions may have an adverse impact on our business. |
| • | We may not fully realize the anticipated benefits of the acquisition, and we may have operational challenges in managing the business and staff of Peace of Meat BV, or Peace of Meat. |
| • | We may have to take significant charges against earnings. |
| • | We are an early-stage company with an unproven business model. |
| • | We may suffer reputational harm due to issues with products manufactured by our licensees. |
| • | Failure to improve our technologies may adversely affect our ability to continue to grow. |
| • | We may face difficulties if we expand our operations into new geographic regions. |
| • | Consumer preferences for alternative proteins in general are difficult to predict and may change. |
| • | We have no manufacturing experience or resources, and we may have issues in obtaining raw materials. |
| • | Litigation or legal proceedings, government investigations or other regulatory enforcement actions could subject us to civil and criminal penalties or otherwise expose us to significant liabilities. |
| • | We expect that a small number of customers will account for a significant portion of our revenues, and we may be exposed to the credit risks of our customers. |
| • | If we are unable to attract and retain qualified employees, our ability to implement our business plan may be adversely affected, and we may not be able to enforce covenants not to compete under applicable employment laws. |
| • | Insurance policies may not fully cover the risk of loss to which we are exposed. |
| • | Our business, reputation and operations could suffer in the event of information technology system failures or a cybersecurity incident. |
| • | Food safety and food-borne illness incidents may materially adversely affect our business. |
| • | Products utilizing our technologies will be subject to regulations that could adversely affect our business and results of operations. |
| • | Any changes in, or failure by our supplier to comply with, applicable laws, regulations or policies could adversely affect our business. |
| • | If we are unable to obtain and maintain our intellectual property rights, we may not be able to compete effectively in our markets. |
| • | Intellectual property rights of third parties could adversely affect our ability to successfully commercialize our products and may prevent or delay our development and commercialization. |
| • | Patent policy and rule changes could increase uncertainties and costs. |
| • | We may be involved in lawsuits to protect or enforce our or third party intellectual property rights. |
| • | Our articles of association provide that unless we consent to an alternate forum, the federal district courts of the United States shall be the exclusive forum of resolution of any claims arising under the Securities Act. |
| • | Political, economic and military conditions in Israel could have an adverse impact on our business. |
| • | We are exposed to fluctuations in currency exchange rates. |
| • | Enforcing a U.S. judgment against us and our executive officers and directors, or asserting U.S. securities law claims in Israel, may be difficult. |
| • | Our articles of association provide that unless we consent otherwise, the competent courts of Tel Aviv, Israel shall be the sole and exclusive forum for substantially all disputes between the Company and its shareholders under the Companies Law and the Israeli Securities Law. |
| • | Your rights and responsibilities as our shareholder will be governed by Israeli law, which may differ in some respects from the rights and responsibilities of shareholders of U.S. corporations. |
| • | Our articles of association and Israeli law could prevent a takeover that shareholders consider favorable and could also reduce the market price of our ADSs. |
| • | You will experience immediate and substantial dilution in the net tangible book value of the ADSs. |
| • | The ADS price may be volatile, and you may lose all or part of your investment in this offering. |
| • | The Pre-Funded Warrants and Warrants are speculative in nature and there is no established market for the Warrants and Pre-Funded Warrants being offered in this offering. |
| • | The Warrants and the Pre-Funded Warrants will be treated as a derivative liability, which could cause us to recognize certain adverse changes to our financial statements. |
| • | We have broad discretion as to the use of the net proceeds from this offering. |
| • | A lack of, inaccurate or unfavorable research coverage could cause the price of the ADSs and trading volume to decline. |
| • | The market price of the ADSs could be negatively affected by future sales of the ADSs. |
| • | We have never paid, and do not intend to pay for the foreseeable future, dividends on our Securities. |
| • | ADS holders may not receive the same distributions or dividends as those we make to the holders of our Ordinary Shares. |
| • | ADS holders do not have the same rights as holders of our Ordinary Shares. |
| • | ADS holders may be subject to limitations on the transfer of their ADSs. |
| • | As a foreign private issuer whose ADSs are listed on Nasdaq, we follow certain home country corporate governance practices instead of certain Nasdaq requirements. We are not subject to U.S. proxy rules and are exempt from certain Exchange Act reporting requirements. If we were to lose our foreign private issuer status, our costs to modify our practices and maintain compliance under U.S. securities laws and Nasdaq rules would be significantly higher. |
| • | If we are a “passive foreign investment company,” or PFIC, or a controlled foreign corporation for U.S. federal income tax purposes, there may be adverse U.S. federal income tax consequences to U.S. investors. |
| • | a requirement to present only two years of audited financial statements in addition to any required interim financial statements and correspondingly reduced Management’s Discussion and Analysis of Financial Condition and Results of Operations disclosure; |
| • | to the extent that we no longer qualify as a foreign private issuer, (i) reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and (ii) exemptions from the requirement to hold a non-binding advisory vote on executive compensation, including golden parachute compensation; |
| • | an exemption from the auditor attestation requirement in the assessment of our internal control over financial reporting pursuant to the Sarbanes-Oxley Act of 2002; and |
| • | an exemption from compliance with the requirement that the Public Company Accounting Oversight Board has adopted regarding a supplement to the auditor’s report providing additional information about the audit and the financial statements. See “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Emerging Growth Company Status.” |
| • | the sections of the Exchange Act regulating the solicitation of proxies, consents or authorizations with respect to a security registered under the Exchange Act; |
| • | the requirement to comply with Regulation FD, which restricts selective disclosure of material information; |
| • | the sections of the Exchange Act requiring insiders to file public reports of their share ownership and trading activities and liability for insiders who profit from trades made in a short period of time; and |
| • | the rules under the Exchange Act requiring the filing with the SEC of quarterly reports on Form 10-Q containing unaudited financial and other specified information, or current reports on Form 8-K upon the occurrence of specified significant events. |
| ADSs offered by us in the offering | Up to 5,594,406 ADSs, each representing ten (10) Ordinary Shares. | |
| Pre-Funded Warrants offered by us | We are also offering to each purchaser, if any, whose purchase of ADSs in this offering would otherwise result in the purchaser, together with its affiliates and certain related parties, beneficially owning more than 9.99% of our outstanding Ordinary Shares immediately following the consummation of this offering, the opportunity to purchase, if the purchaser so chooses, Pre-Funded Warrants in lieu of ADSs that would otherwise result in the purchaser's beneficial ownership exceeding 9.99% of our outstanding Ordinary Shares. Each Pre-Funded Warrant will be immediately exercisable for one ADS and may be exercised at any time until all of the Pre-Funded Warrants are exercised in full. The purchase price of each Pre-Funded Warrant will equal the price per share at which the ADSs are being sold to the public in this offering, minus $0.0001, the exercise price of each Pre-Funded Warrant. For each Pre-Funded Warrant we sell, the number of ADSs we are offering will be decreased on a one-for-one basis. | |
| Warrants | Each warrant will have an assumed exercise price of $1.43 (based on an assumed public offering price of $1.43 per ADS, the last reported sale price of our ADSs on Nasdaq on January 4, 2023) per ADS (100% of the public offering price per ADS), will be immediately exercisable and will expire five years from the date of issuance. To better understand the terms of the warrants, you should carefully read the “Description of the Offered Securities” section of this prospectus. You should also read the form of warrant, which is filed as an exhibit to the registration statement that includes this prospectus. | |
| Total number of Ordinary Shares outstanding immediately before this offering | 146,471,677 Ordinary Shares. | |
| Total number of Ordinary Shares outstanding immediately after this offering | Up to 202,415,733 Ordinary Shares, assuming no exercise of the Pre-Funded Warrants, or 210,807,327 Ordinary Shares if the underwriters exercise in full the over-allotment option to purchase additional ADSs, assuming no exercise of the Pre-Funded Warrants. | |
| The ADSs | Each ADS represents ten (10) Ordinary Shares. The depositary will hold Ordinary Shares underlying our ADSs. You will have rights as provided in the deposit agreement among us, the depositary and owners and beneficial owners of ADSs from time to time. To better understand the terms of our ADSs, you should carefully read the “Description of the Offered Securities” section of this prospectus. You should also read the deposit agreement, which is filed as an exhibit to the registration statement that includes this prospectus. | |
| Over-allotment option | We have granted the underwriters an option for a period of up to 45 days to purchase, at the public offering price, up to additional ADSs and/or 839,161 Warrants, less underwriting discounts and commissions, to cover over-allotments, if any. | |
| Use of proceeds | We expect to receive approximately $7.1 million in net proceeds from the sale of ADSs and/or Pre-Funded Warrants offered by us in this offering (approximately $8.2 million if the underwriters exercise their over-allotment option in full), based upon an assumed public offering price of $1.43 per ADS, the last reported sale price of our ADSs on Nasdaq on January 4, 2023. The actual offering price per share in this offering will be determined between us and the underwriters at the time of pricing, and may be at a discount to the current market price. We will use the net proceeds that we receive from the sale of the Securities offered by this prospectus for general corporate purposes, which may include operating expenses, working capital, future acquisitions or share repurchases, general capital expenditures and satisfaction of debt obligations. We have not determined the amount of net proceeds to be used specifically for such purposes. See “Use of Proceeds” for additional information. | |
| Risk factors | Investing in our Securities involves a high degree of risk. You should read the “Risk Factors” section starting on page 11 of this prospectus for a discussion of factors to consider carefully before deciding to invest in our Securities. | |
| Listing | Our ADSs are traded on Nasdaq under the symbol “STKH.” We do not intend to apply for a listing of the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system. | |
| Depositary | The Bank of New York Mellon. |
| • | 1,303,002 ADSs issuable upon the exercise of options and restricted share units to purchase ADSs outstanding as of such date, at a weighted average exercise price of $6.72 per ADS; |
| • | a total of 1,607,728 of our ADSs reserved for future issuance under our 2022 Share Incentive Plan, as of such date; |
| • | 704,454 ADSs issuable upon exercise of options and restricted share units outstanding as of such date at an exercise price to be determined at the time of exercise using a pre-determined formula; |
| • | 3,539,982 ADSs issuable upon the exercise of investor warrants to purchase ADSs outstanding as of such date at a weighted average exercise price of $7.42 per ADS, which warrants are expected to remain outstanding at the consummation of this offering; and |
| • | 139,020 ADSs issuable upon exercise of rights to investors that had been granted and remained outstanding as of such date with no exercise price, vesting based on milestones yet to be achieved. |
| • | no exercise or forfeiture of the outstanding options or warrants or settlement of restricted share units after January 4, 2023; |
| • | no sale of Pre-Funded Warrants in this offering; |
| • | no exercise by the underwriters of their over-allotment option; and |
| • | no exercise of Underwriter Warrants. |
| Year Ended December 31, | Six Months Ended June 30, | Nine Months Ended September 30, | ||||||||||||||||||||||
| 2021 | 2020 | 2022 | 2021 | 2022 | 2021 | |||||||||||||||||||
| (U.S. Dollars, in thousands, except per share and weighted average shares data) | ||||||||||||||||||||||||
| Consolidated Statements of Loss and Other Comprehensive Loss | ||||||||||||||||||||||||
| Research and development expenses | 7,594 | 2,491 | 4,427 | 2,910 | 7,219 | 4,928 | ||||||||||||||||||
| Marketing expenses | 1,628 | 506 | 1,959 | 605 | 2,426 | 872 | ||||||||||||||||||
| General and administrative expenses | 8,010 | 5,380 | 3,687 | 4,159 | 4,982 | 5,961 | ||||||||||||||||||
| Public listing expenses | - | 10,164 | - | - | - | - | ||||||||||||||||||
| Operating loss | 17,232 | 18,541 | 10,073 | 7,674 | 14,627 | 11,761 | ||||||||||||||||||
| Financing income | (509 | ) | (110 | ) | (1,062 | ) | (401 | ) | (3,258 | ) | (457 | ) | ||||||||||||
| Financing expenses | 1,299 | 93 | 145 | 493 | 262 | 434 | ||||||||||||||||||
| Total financing (income) expenses | 790 | (17 | ) | (917 | ) | 92 | (2,996 | ) | (23 | ) | ||||||||||||||
| Loss for the period | $ | 18,022 | $ | 18,524 | $ | 9,156 | 7,766 | $ | 11,631 | 11,738 | ||||||||||||||
| Capital reserve for financial assets at fair value that will not be transferred to profit or loss | - | 334 | - | - | - | - | ||||||||||||||||||
| Currency translation differences loss (income) that will not be transferred to profit or loss over NIS | (1,942 | ) | (758 | ) | 2,326 | (293 | ) | 2,301 | (456 | ) | ||||||||||||||
Currency translation differences loss (income) that might be transferred to profit or loss over EUR | 1,447 | - | 847 | 216 | 1,745 | 717 | ||||||||||||||||||
| Total comprehensive loss for the period | $ | 17,527 | $ | 18,100 | $ | 12,329 | 7,689 | $ | 15,677 | 11,999 | ||||||||||||||
| Basic and diluted loss per ordinary share | $ | 0.155 | $ | 0.308 | $ | 0.072 | 0.072 | $ | 0.088 | 0.104 | ||||||||||||||
Weighted average number of Ordinary Shares outstanding used to compute-basic and diluted: | 115,954,501 | 60,112,197 | 126,385,924 | 107,189,837 | 132,652,822 | 112,891,686 | ||||||||||||||||||
| As of September 30, 2022 | ||||||||
| Actual | As Adjusted(1) | |||||||
| (U.S. Dollars in thousands) | ||||||||
| Consolidated Balance Sheet Data: | ||||||||
| Cash and cash equivalents | $ | 11,203 | $ | 18,332 | ||||
| Total assets | 33,226 | 40,355 | ||||||
| Derivative instrument | (2,450 | ) | (8,044 | ) | ||||
| Total non-current liabilities | (3,195 | ) | (3,195 | ) | ||||
| Accumulated deficit | 48,602 | 49,212 | ||||||
| Total shareholders’ equity | (24,439 | ) | (25,973 | ) | ||||
| (1) | As adjusted gives further effect to the assumed issuance and sale in this offering of 5,594,406 ADSs and/or Pre-Funded Warrants representing 55,944,056 Ordinary Shares (assuming the full exercise of the Pre-Funded Warrants) at the assumed public offering price of $1.43 per ADS, which was the last reported sale price of our ADSs on Nasdaq on January 4, 2023, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
| • | our progress with current research and development activities; |
| • | the number and characteristics of any products or manufacturing processes we develop or acquire; |
| • | the expenses associated with our marketing initiatives; |
| • | the timing, receipt and amount of milestone, royalty and other payments from future customers and collaborators, if any; |
| • | the scope, progress, results and costs of researching and developing future products or improvements to existing products or manufacturing processes; |
| • | any lawsuits related to our products or commenced against us; |
| • | the expenses needed to attract, hire and retain skilled personnel; |
| • | the costs associated with being a public company in the United States; and |
| • | the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing intellectual property claims, including litigation costs and the outcome of such litigation. |
| • | increased operating expenses and cash requirements; |
| • | the assumption of additional indebtedness or contingent liabilities; |
| • | assimilation of operations, intellectual property and products of an acquired company, including difficulties associated with integrating new personnel; |
| • | the diversion of our management’s attention from our existing programs and initiatives in pursuing such a strategic merger or acquisition; |
| • | retention of key employees, the loss of key personnel, and uncertainties in our ability to maintain key business relationships; |
| • | risks and uncertainties associated with the other party to such a transaction, including the prospects of that party and their existing technologies; and |
| • | our inability to generate revenue from acquired technologies or products sufficient to meet our objectives in undertaking the acquisition or even to offset the associated acquisition and maintenance costs. |
| • | collaborators may not perform or prioritize their obligations as expected; |
| • | collaborators may not pursue development and commercialization of any of our cultured meat manufacturing technologies or may elect not to continue or renew development or commercialization, changes in the collaborators’ focus or available funding, or external factors, such as an acquisition, that divert resources or create competing priorities; |
| • | collaborators may provide insufficient funding for the successful development or commercialization of our cultured meat manufacturing technologies; |
| • | collaborators could independently develop, or develop with third parties, products or technologies that compete directly or indirectly with our products or cultured meat manufacturing technologies if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours; |
| • | cultured meat manufacturing technologies developed in collaborations with us may be viewed by our collaborators as competitive with their own products or technologies, which may cause collaborators to cease to devote resources to the development or commercialization of our products; |
| • | a collaborator with marketing and distribution rights to one or more of our products or technologies that achieve regulatory approval may not commit sufficient resources to the marketing and distribution of any such product; |
| • | disagreements with collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development of cultured meat manufacturing technologies, may cause delays or termination of the research, development or commercialization of such technologies, may lead to additional responsibilities for us with respect to such technologies, or may result in litigation or arbitration, any of which would be time-consuming and expensive; |
| • | collaborators may not properly maintain, protect, defend or enforce our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation; |
| • | disputes may arise with respect to the ownership of intellectual property developed pursuant to our collaborations; |
| • | collaborators may infringe, misappropriate or otherwise violate the intellectual property rights of third parties, which may expose us to litigation and potential liability; |
| • | collaborations may be terminated for the convenience of the collaborator and, if terminated, the development of our cultured meat manufacturing technologies may be delayed, and we could be required to raise additional capital to pursue further development or commercialization of the cultured meat manufacturing technologies; |
| • | future relationships may require us to incur non-recurring and other charges, increase our near- and long-term expenditures, issue securities that dilute our existing shareholders, or disrupt our management and business; and |
| • | we could face significant competition in seeking appropriate collaborators, and the negotiation process is time-consuming and complex. |
| • | Israeli corporate law regulates mergers and requires that a tender offer be effected when more than a specified percentage of shares in a company are purchased; | |
| • | Israeli corporate law requires special approvals for certain transactions involving directors, officers or significant shareholders and regulates other matters that may be relevant to these types of transactions; |
| • | Israeli corporate law does not provide for shareholder action by written consent for public companies, thereby requiring all shareholder actions to be taken at a general meeting of shareholders; | |
| • | our articles of association divide our directors into three classes, each of which is elected once every three years; | |
| • | our articles of association generally require a vote of the holders of a majority of our outstanding ordinary shares entitled to vote present and voting on the matter at a general meeting of shareholders (referred to as simple majority), and solely the amendment of the provision relating to the removal of members of our board of directors, require a vote of the holders of 65% of our outstanding ordinary shares entitled to vote at a general meeting; | |
| • | our articles of association provide that director vacancies may be filled by our board of directors. |
| • | changes in the prices of our raw materials or the products manufactured in factories using our technologies; |
| • | the trading volume of the ADSs; |
| • | the effects of the COVID-19 pandemic; |
| • | general economic, market and political conditions, including negative effects on consumer confidence and spending levels that could indirectly affect our results of operations; |
| • | actual or anticipated fluctuations in our financial condition and operating results, including fluctuations in our quarterly and annual results; |
| • | announcements by us or our competitors of innovations, other significant business developments, changes in distributor relationships, acquisitions or expansion plans; |
| • | announcement by competitors or new market entrants of their entry into or exit from the alternative protein market; |
| • | overall conditions in our industry and the markets in which we intend to operate; |
| • | market conditions or trends in the packaged food sales industry that could indirectly affect our results of operations; |
| • | addition or loss of significant customers or other developments with respect to significant customers; |
| • | adverse developments concerning our manufacturers and suppliers; |
| • | changes in laws or regulations applicable to our products or business; |
| • | our ability to effectively manage our growth and market expectations with respect to our growth, including relative to our competitors; |
| • | changes in the estimation of the future size and growth rate of our markets; |
| • | announcements by us or our competitors of significant acquisitions, strategic partnerships, joint ventures or capital commitments; |
| • | additions or departures of key personnel; |
| • | competition from existing products or new products that may emerge; |
| • | issuance of new or updated research or reports about us or our industry, or positive or negative recommendations or withdrawal of research coverage by securities analysts; |
| • | variance in our financial performance from the expectations of market analysts; |
| • | our failure to meet or exceed the estimates and projections of the investment community or that we may otherwise provide to the public; |
| • | fluctuations in the valuation of companies perceived by investors to be comparable to us; |
| • | disputes or other developments related to proprietary rights, including patents, and our ability to obtain intellectual property protection for our products; |
| • | litigation or regulatory matters; |
| • | announcement or expectation of additional financing efforts; |
| • | our cash position; |
| • | sales and short-selling of the ADSs; |
| • | our issuance of equity or debt; |
| • | changes in accounting practices; |
| • | ineffectiveness of our internal controls; |
| • | negative media or marketing campaigns undertaken by our competitors or lobbyists supporting the conventional meat industry; |
| • | the public’s response to publicity relating to the health aspects or nutritional value of products to be manufactured in factories using our technologies; and |
| • | other events or factors, many of which are beyond our control. |
| • | our estimates regarding our expenses, future revenue, capital requirements and needs for additional financing; |
| • | our expectations regarding the success of our cultured meat manufacturing technologies we are developing, which will require significant additional work before we can potentially launch commercial sales; |
| • | our research and development activities associated with technologies for cultured meat manufacturing, including three-dimensional meat production, which involves a lengthy and complex process; |
| • | our expectations regarding the timing for the potential commercial launch of our cultured meat technologies; |
| • | our ability to successfully manage our planned growth, including with respect to our recent acquisition of Peace of Meat, and any future acquisitions, joint ventures, collaborations or similar transactions; |
| • | the potential business or economic disruptions caused by the COVID-19 pandemic; |
| • | the competitiveness of the market for our cultured meat technologies; |
| • | our ability to enforce our intellectual property rights and to operate our business without infringing, misappropriating, or otherwise violating the intellectual property rights and proprietary technology of third parties; |
| • | our ability to predict and timely respond to preferences for alternative proteins and cultured meats and new trends; |
| • | our ability to predict and timely respond to preferences for alternative proteins and cultured meats and new trends; and |
| • | other risks and uncertainties, including those listed under the heading “Risk Factors” in this prospectus and our Annual Report on Form 20-F for the year ended December 31, 2021, filed with the SEC on March 24, 2022. |
| • | on an actual basis; and | |
| • | on an as adjusted basis, to give effect to the assumed issuance and sale in this offering of 5,594,406 ADSs representing 55,944,056 Ordinary Shares at the assumed public offering price of $1.43 per ADS, the last reported sales price of our ADSs on Nasdaq on January 4, 2023, and assuming no sale of any Pre-Funded Warrants, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
| As of September 30, 2022 | ||||||||
| Actual | As adjusted | |||||||
(U.S. Dollars, in thousands) | ||||||||
| Cash and cash equivalents | 11,203 | 18,332 | ||||||
| Derivative liability | (2,450 | ) | (8,044 | ) | ||||
| Shareholders’ equity: | ||||||||
| Equity: | ||||||||
| Ordinary Shares, no per share: 1,000,000,000 ordinary shares authorized (actual and as adjusted); 135,767,137 Ordinary Shares issued and outstanding (actual); 199,542,510 Ordinary Shares outstanding (as adjusted) | ||||||||
| Share capital and premium on shares | (72,231 | ) | (74,234 | ) | ||||
| Capital reserves | (3,581 | ) | (3,721 | ) | ||||
| Currency translation differences reserve | 2,771 | 2,771 | ||||||
| Accumulated deficit | 48,602 | 49,212 | ||||||
| Total shareholders’ capital equity | (24,439 | ) | (25,973 | ) | ||||
Each $0.10 increase (decrease) in the assumed public offering price of $1.43 per share, which is the last reported sale price of our ADSs on Nasdaq on January 4, 2023, would increase (decrease) cash and cash equivalents and short term bank deposits by $520,280, and our total shareholders’ equity on an as adjusted basis by approximately $129,063, assuming the number of shares offered, as set forth on the cover page of this prospectus, remains the same, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
Each 100,000 increase (decrease) in the number of ADSs offered in this offering would increase or decrease cash and cash equivalents and short term bank deposits by approximately $132,990, and our total shareholders’ equity on an as adjusted basis by approximately $32,990, assuming that the price per ADS for the offering remains at $1.43, which is the last reported sales price of our ADSs on Nasdaq on January 4, 2023, and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
| • | 1,303,002 ADSs issuable upon the exercise of options and restricted share units to purchase ADSs outstanding as of January 4, 2023, at a weighted average exercise price of $6.72 per ADS; |
| • | a total of 1,607,728 of our ADSs reserved for future issuance under our 2022 Share Incentive Plan, as of January 4, 2023; |
| • | 704,454 ADSs issuable upon exercise of options and restricted share units outstanding as of January 4, 2023, at an exercise price to be determined at the time of exercise using a pre-determined formula; |
| • | 3,539,982 ADSs issuable upon the exercise of investor warrants to purchase ADSs outstanding as of January 4, 2023, at a weighted average exercise price of $7.42 per ADS, which warrants are expected to remain outstanding at the consummation of this offering; and |
| • | 139,020 ADSs issuable upon exercise of rights to investors that had been granted and remained outstanding as of January 4, 2023, with no exercise price, vesting based on milestones yet to be achieved. |
| • | no exercise or forfeiture of the outstanding options or warrants or settlement of restricted share units after January 4, 2023; |
| • | no sale of Pre-Funded Warrants in this offering; |
| • | no exercise by the underwriters of their over-allotment option; and |
| • | no exercise of Underwriter Warrants. |
| Assumed offering price per ADS | $ | 1.43 | ||||||
| Net tangible book value per ADS as of September 30, 2022 | $ | 0.94 | ||||||
| Decrease in net tangible book value per ADS attributable to the offering | $ | 0.20 | ||||||
| As-adjusted net tangible book value per ADS after giving effect to the offering | $ | 0.74 | ||||||
| Dilution in net tangible book value per ADS to new investors | $ | 0.69 | ||||||
| • | 1,303,002 ADSs issuable upon the exercise of options and restricted share units to purchase ADSs outstanding as of January 4, 2023, at a weighted average exercise price of $6.72 per ADS; |
• | a total of 1,607,728 of our ADSs reserved for future issuance under our 2022 Share Incentive Plan, as of January 4, 2023; |
• | 704,454 ADSs issuable upon exercise of options and restricted share units outstanding as of January 4, 2023, at an exercise price to be determined at the time of exercise using a pre-determined formula; |
• | 3,539,982 ADSs issuable upon the exercise of investor warrants to purchase ADSs outstanding as of January 4, 2023, at a weighted average exercise price of $7.42 per ADS, which warrants are expected to remain outstanding at the consummation of this offering; and |
• | 139,020 ADSs issuable upon exercise of rights to investors that had been granted and remained outstanding as of January 4, 2023, with no exercise price, vesting based on milestones yet to be achieved. |
no exercise or forfeiture of the outstanding options or warrants or settlement of restricted share units after January 4, 2023; |
| no sale of Pre-Funded Warrants in this offering; |
| no exercise by the underwriters of their over-allotment option; and |
| no exercise of Underwriter Warrants. |
| • | employee-related expenses, such as salaries and share-based compensation; |
| • | expenses relating to outsourced and contracted services, such as external laboratories and consulting, research and advisory services; |
| • | supply and development costs; |
| • | expenses, such as materials, incurred in operating our laboratories and equipment; and |
| • | costs associated with regulatory compliance. |
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
| USD in thousands | ||||||||
| Operating expenses: | ||||||||
| Research and development expenses | $ | 7,594 | $ | 2,491 | ||||
| Marketing expenses | 1,628 | 506 | ||||||
| General and administrative expenses | 8,010 | 5,380 | ||||||
| Public listing expenses | - | 10,164 | ||||||
| Loss from operations | $ | 17,232 | $ | 18,541 | ||||
| Finance income | 509 | 110 | ||||||
| Finance expense | 1,299 | 93 | ||||||
| Finance expense (income), net | 790 | (17 | ) | |||||
| Net loss | $ | 18,022 | $ | 18,524 | ||||
| Six months Ended June 30, | ||||||||
| 2022 | 2021 | |||||||
| USD in thousands | ||||||||
| Operating expenses: | ||||||||
| Research and development expenses | $ | 4,427 | $ | 2,910 | ||||
| Marketing expenses | 1,959 | 605 | ||||||
| General and administrative expenses | 3,687 | 4,159 | ||||||
| Loss from operations | $ | 10,073 | $ | 7,647 | ||||
| Finance income | (1,062) | (401 | ||||||
| Finance expense | 145 | 493 | ||||||
| Finance expense (income), net | (917) | 92 | ||||||
| Net loss | $ | 9,156 | $ | 7,766 | ||||
| Nine months Ended September 30, | ||||||||
| 2022 | 2021 | |||||||
USD in thousands | ||||||||
| Operating expenses: | ||||||||
| Research and development expenses | $ | 7,219 | $ | 4,928 | ||||
| Marketing expenses | 2,426 | 872 | ||||||
| General and administrative expenses | 4,982 | 5,961 | ||||||
| Loss from operations | $ | 14,627 | $ | 11,761 | ||||
| Finance income | (3,258 | ) | (457 | ) | ||||
| Finance expense | 262 | 434 | ||||||
| Finance expense (income), net | (2,996 | ) | (23 | ) | ||||
| Net loss | $ | 11,632 | $ | 11,738 | ||||
| Year Ended December 31, | ||||||||
| 2021 | 2020 | |||||||
USD in thousands | ||||||||
| Net cash used in operating activities | $ | (13,960 | ) | $ | (3,832 | ) | ||
| Net cash used in investing activities | (9,340 | ) | (1,875 | ) | ||||
| Net cash provided by financing activities | 29,023 | 17,345 | ||||||
| Net increase in cash and cash equivalents | $ | 5,723 | $ | 11,638 | ||||
| Six months Ended June 30, | ||||||||
| 2022 | 2021 | |||||||
| USD in thousands | ||||||||
| Net cash used in operating activities | $ | (7,448 | ) | $ | (5,048 | ) | ||
| Net cash used in investing activities | (2,476 | ) | (6,381 | ) | ||||
| Net cash provided by financing activities | (314) | 29,059 | ||||||
| Net increase in cash and cash equivalents | $ | (10,238) | $ | 17,630 | ||||
| Nine months Ended September 30, | ||||||||
| 2022 | 2021 | |||||||
| USD in thousands | ||||||||
| Net cash used in operating activities | $ | (9,845 | ) | $ | (9,612 | ) | ||
| Net cash used in investing activities | (2,711 | ) | (7,402 | ) | ||||
| Net cash provided by financing activities | 5,330 | 28,965 | ||||||
| Net increase in cash and cash equivalents | $ | (7,226 | ) | $ | 11,951 | |||
| • | the progress and costs of our research and development activities; |
| • | the costs of development and expansion of our operational infrastructure; |
| • | the costs and timing of developing technologies sufficient to allow food production equipment manufacturers and food manufacturers to product products compliant with applicable regulations; |
| • | our ability, or that of our collaborators, to achieve development milestones and other events or developments under potential future licensing agreements; |
| • | the amount of revenues and contributions we receive under future licensing, collaboration, development and commercialization arrangements with respect to our technologies; |
| • | the costs of filing, prosecuting, enforcing and defending patent claims and other intellectual property rights; |
| • | the costs of contracting with third parties to provide sales and marketing capabilities for us or establishing such capabilities ourselves, once our technologies are developed and ready for commercialization; |
| • | the costs of acquiring or undertaking development and commercialization efforts for any future products or technology; |
| • | the magnitude of our general and administrative expenses; and |
| • | any additional costs that we may incur under future in- and out-licensing arrangements relating to our technologies and futures products. |
• | Environmental: At least 18% of the greenhouse gases entering the atmosphere today are from the livestock industry. Research shows that the expected environmental footprint of cultivated meat includes approximately 78% to 96% fewer greenhouse gas emissions, 63%-95% less land use, 51% to 78% less water use, and 7% to 45% less energy use than conventionally-produced beef, lamb, pork and poultry. This suggests that the environmental consequences of switching from large-scale, factory farming to lab-grown cultivated meat could have a long-term positive impact on the environment. |
| • | Cost: While the precise economic value of harvested cells has yet to be determined, the potential to harvest large numbers of cells from a small number of live donor animals gives rise to the possibility of considerably higher returns than traditional agriculture, with production cycles potentially measured in months rather than years. By comparison, raising a cow for slaughter generally takes an average of 18 months, over which period 15,400 liters of water and 7 kilograms of feed will be consumed for every kilogram of beef produced. While the original cultivated burger is thought to have cost around $330 thousand, consulting firm CE Delft estimates that economies of scale combined with technological improvements will bring the cost of cultivated meat down to less than $8 per kilogram by 2035. |
| • | Animal Suffering: More and more people are grappling with the ethical question of whether humanity should continue to slaughter animals for food. There is a growing trend of opposition to the way animals are raised for slaughter, often in small, confined spaces with unnatural feeding patterns. In many cases, such animals suffer terribly throughout their lives. This consideration is likely a factor in many consumers choosing to incorporate more flexitarian, vegetarian and vegan approaches to their diets in recent years. |
| • | Controlled Growing Environment: Another potential benefit of cultivated meat is that its growth environment is designed to be less susceptible to biological risk and disease, through standardized, tailored production methods consistent with good manufacturing practices that are controls to contribute to improved nutrition, health and wellbeing. |
| • | Alternate Use of Natural Resources: Eight percent of the world’s freshwater supply and one third of croplands are currently used to provide for livestock. The development of cultivated meat is expected to free up many of these natural resources, especially in developing economies where they are most needed. |
| • | Food Waste: The conventional meat industry’s largest waste management problem relates to the disposal of partially-used carcasses, which are usually buried, incinerated, rendered or composted, with attendant problems such as land, water or air pollution. Cultivated meat offers a potential solution for this problem, with only the desired cuts of meat being produced for consumption and only minimal waste product generated with no leftover carcass. |
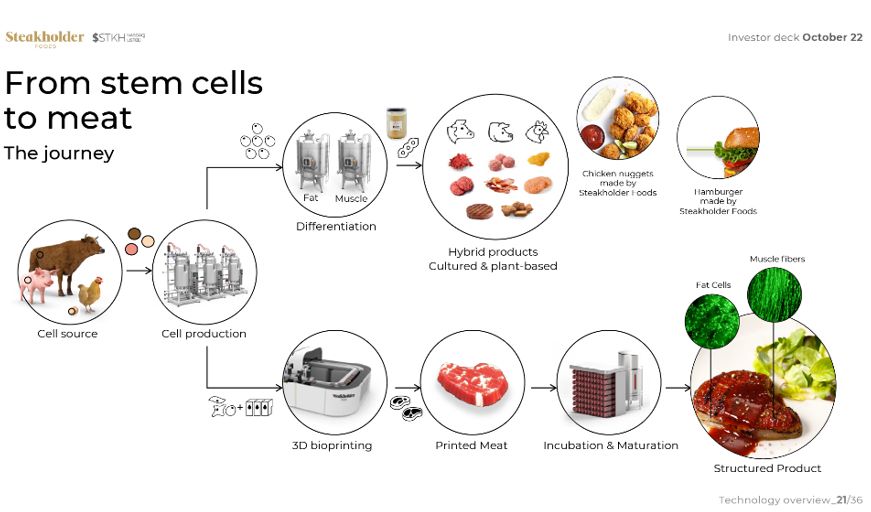
| • | Perfect the development of our cultivated meat manufacturing technology and processes. We intend to continue developing and refining our processes, procedures and equipment until we are in a position to commercialize our technologies, whether by manufacturing final products for consumers (B2C and B2B2C models) or ingredients for industrial use, as well as in outlicensing (B2B models). We are continuing to tackle the technological challenges involved in scaling up both our biological and printing processes to industrial-scale levels. |
| • | Commercialize our technologies for use in consumer and business markets. We intend to provide ingredients to business customers for use in consumer products in order to help meet the growing demand for sustainable, slaughter-free cultivated meat products. For example, manufacturers of meat alternatives, such as vegetarian sausages, may choose to include our cultivated fat biomass in their products in order to deliver the signature meaty flavors, aromas and textures of the meat that is otherwise provided by the conventional meat of species such as chicken, beef and pork. We believe that this combination has the potential to unlock a new level of meat experience. In addition, we intend to license our production technology as well as provide associated products, such as cell lines, printheads, bioreactors and incubators, and services, such as technology implementation, training, and engineering support, whether directly or through contractors, to food processing and food retail companies. We intend to charge our customers a production license fee, based upon the amount of meat printed. We expect that each production facility will periodically require us to provide them with our proprietary materials, such as fresh sets of starter cells, for a fee. In addition, other materials used in the production process, such as cell-culture media and additives in our bio-inks may be sourced from third parties. Whether these materials are customized for the specifics of our production processes, “white-labelled” generic materials or proprietary materials that we have developed, we may charge a fee for restocking such materials; however, we have not yet reached the stage where it would be possible to estimate to what extent this would contribute to any future revenue stream. Finally, we intend to provide paid product implementation and guidance services to our customers looking to establish cultivated meat manufacturing facilities. We expect that each facility licensing our technologies will need to deal with novel challenges and, as a result, will require the assistance of our expert knowledge in order to set up and implement our licensed technologies. |
| • | Develop additional alternative proteins to meet growing industry demand. There are substantial technological challenges inherent in expanding our offering beyond our current cultivated beef, pork and avian technologies to additional alternative proteins and cell lines. However, we believe that our experience, know-how and intellectual property portfolio form an excellent basis from which to surmount such challenges. Our first step in this direction was the 2021 acquisition of Peace of Meat BV, or Peace of Meat, with the aim of developing avian fat for the alternative meat industry by applying proprietary technology to mimic the cellular composition of conventional poultry. In addition, in January 2023, we announced a collaboration with Singaporean cultivated seafood developer, Umami Meats, to develop 3D-printed structured eel and grouper products pursuant to a grant from the Singapore-Israel R&D Foundation. The initiative is being funded by a grant from the Singapore-Israel Industrial R&D Foundation (SIIRD), a cooperation between Enterprise Singapore (ESG) and the Israel Innovation Authority (IIA). The collaboration aims to develop a scalable process for producing structured cultivated fish products and will involve the use of our newly-developed technology for mimicking the flaky texture of cooked fish which was the subject of a recent patent application. By the end of the first quarter of 2023, we intend to complete the project’s first prototype, a structured hybrid grouper product printed using our proprietary three-dimensional bio-printing technology and bio-inks, customized for cells provided by Umami Meats. |
| • | Acquire synergistic and complementary technologies and assets. We intend to optimize our processes and diversify our product range to expand the cultivated meat technologies upon which marketable products can be based. We intend to accomplish this through a combination of internal development, acquisitions and collaborations, with a view to complementing our own processes and diversifying our product range along the cultivated meat production value chain in order to introduce cultivated products to the global market as quickly as possible. See also “- Additional Technologies” below. |
| • | Fully-plant-based meat-like offerings that are already commercially available but lack the organoleptic properties of meat, primarily flavor, aroma, texture and color; |
| • | Hybrid meat products of the type that we are developing, which combines real cultivated fat with plant-based protein to offer meatier products with enhanced organoleptic properties; |
| • | Unstructured meat products, such as hamburgers and minced meat; and |
| • | Fully-cultivated structured meat products, such as 3D-printed steaks. |




| • | Replacing expensive, animal-derived components in cell growth media with chemical replacements, including through in-house production, with a view to completing animal-free growth media and bio-ink by the first half of 2023; |
| • | Cell line optimizations, such as through high-throughput analyses of evolved isolates; |
| • | Bioprocess optimization and media recycling; |
| • | Upscaled growth factor production, such as through hollow fiber bioreactors; and |
| • | Long-term market optimization as a result of expected increased demand. |


| Name | Jurisdiction of Incorporation | Parent | % Ownership (direct or otherwise) | |||||||
| Steakholder Foods USA, Inc. | Delaware, U.S. | Steakholder Foods Ltd. | 100 | % | ||||||
| Steakholder Innovation Ltd. | Israel | Steakholder Foods Ltd. | 100 | % | ||||||
| Steakholder Foods Europe BV | Belgium | Steakholder Foods Ltd. | 100 | % | ||||||
| Peace of Meat BV | Belgium | Steakholder Foods Europe BV | 100 | % | ||||||
| Name | Age | Position | ||
| Executive Officers: | ||||
| Arik Kaufman | 42 | Chief Executive Officer | ||
| Guy Hefer | 41 | Chief Financial Officer | ||
| Dan Kozlovski | 38 | Chief Technologies Officer | ||
| Non-Employee Directors: | ||||
| Yaron Kaiser | 44 | Chairman of the Board of Directors | ||
| David Gerbi(1)(2)(3) | 43 | Director | ||
| Eli Arad(1)(2)(3) | 50 | Director | ||
| Sari Singer(1)(2)(3) | 42 | Director |
| Name and Principal Position | Salary(1) | Bonus(2) | Equity-Based Compensation(3) | Other Compensation(4) | Total | |||||||||||||||
(USD in thousands) | ||||||||||||||||||||
| Mr. Arik Kaufman | ||||||||||||||||||||
| Chief Executive Officer | $ | 235 | $ | - | $ | 88 | $ | 8 | $ | 331 | ||||||||||
| Mr. Guy Hefer | ||||||||||||||||||||
| Chief Financial Officer | 210 | 34 | 83 | - | 327 | |||||||||||||||
| Mr. Dan Kozlovski | ||||||||||||||||||||
| Chief Technologies Officer | 201 | 42 | 51 | - | 294 | |||||||||||||||
| Mr. Yaron Kaiser | ||||||||||||||||||||
| Chairman of the Board of Directors | 161 | - | 62 | 8 | 231 | |||||||||||||||
Mr. Steven H. Levin (5) | ||||||||||||||||||||
Former Chairman of the Board of Directors(5) | $ | 57 | $ | - | $ | 108 | $ | - | $ | 165 | ||||||||||
| (1) | Salary includes the officer’s gross salary plus payment by us of social benefits on behalf of the officer. Such benefits may include payments, contributions and/or allocations for savings funds (e.g., Managers’ Life Insurance Policy), pension, severance, risk insurance (e.g., life, or work disability insurance), payments for social security and tax gross-up payments, vacation, medical insurance and benefits, convalescence or recreation pay and other benefits and perquisites consistent with our policies. |
| (2) | Represents annual bonuses paid in 2022 with respect to 2021. |
| (3) | Represents the equity-based compensation expenses, based on the options’ fair value on the grant date, calculated in accordance with applicable accounting guidance for equity-based compensation. For a discussion of the assumptions used in reaching this valuation, see Note 10(B) to our annual consolidated financial statements included elsewhere in this prospectus. |
| (4) | Represents consulting services provided prior to commencement of the aforementioned current position. |
| (5) | Mr. Levin resigned his position as Chairman on January 24, 2022. |
| • | Quorum. As permitted under the Companies Law, pursuant to our articles of association, the quorum required for an ordinary meeting of shareholders consists of at least two shareholders present in person or by proxy who hold or represent between them at least 25% of the voting power of our shares (and, with respect to an adjourned meeting, generally one or more shareholders who hold or represent any number of shares), instead of 33 1∕3% of the issued share capital provided under Nasdaq Marketplace Rule 5260(c). |
| • | Shareholder Approval. Although the Nasdaq Marketplace Rules generally require shareholder approval of equity compensation plans and material amendments thereto, we follow Israeli practice, which is to have such plans and amendments approved only by the board of directors, unless such arrangements are for the compensation of chief executive officer or directors, in which case they also require the approval of the compensation committee and the shareholders. In addition, rather than follow the Nasdaq Marketplace Rule requiring shareholder approval for the issuance of securities in certain circumstances, we follow Israeli law, under which a private placement of securities requires approval by our board of directors and shareholders if it will cause a person to become a controlling shareholder (generally presumed at 25% ownership) or if: (a) the securities issued amount to 20% or more of our outstanding voting rights before the issuance; (b) some or all of the consideration is other than cash or listed securities or the transaction is not on market terms; and (c) transaction will increase the relative holdings of a shareholder that holds 5% or more of our outstanding share capital or voting rights or will cause any person to become, as a result of the issuance, a holder of more than 5% of our outstanding share capital or voting rights. |
| • | Executive Sessions. While the Nasdaq Marketplace Rules require that “independent directors,” as defined in the Nasdaq Marketplace Rules, must have regularly scheduled meetings at which only “independent directors” are present. Israeli law does not require, nor do our independent directors necessarily conduct, regularly scheduled meetings at which only they are present. |
| • | the Class I directors are Messrs. Eli Arad and David Gerbi and their respective terms will expire at the Company’s annual general meeting of shareholders to be held in 2023; |
| • | the Class II director is Ms. Sari Singer and her term will expire at the Company’s annual general meeting of shareholders to be held in 2024; and |
| • | the Class III director is Mr. Yaron Kaiser his term will expire at the Company’s annual general meeting of shareholders to be held in 2025. |
| • | refrain from any act involving a conflict of interest between the performance of the office holder’s duties in the company and the office holder’s other duties or personal affairs; | |
| • | refrain from any activity that is competitive with the business of the company; |
| • | refrain from exploiting any business opportunity of the company for the purpose of gaining a personal advantage for the office holder or others; and | |
| • | disclose to the company any information or documents relating to the company’s affairs which the office holder received as a result of the office holder’s position. |
| • | a majority of the shares held by shareholders who are not controlling shareholders and have no personal interest in the transaction and are voting at the meeting must be voted in favor of approving the transaction, excluding abstentions; or |
| • | the shares voted by shareholders who are non-controlling shareholders and who have no personal interest in the transaction who vote against the transaction represent no more than 2% of the voting rights in the company. |
| • | an amendment to the articles of association; |
| • | an increase in the company’s authorized share capital; |
| • | a merger; and |
| • | the approval of related party transactions and acts of office holders that require shareholder approval. |
| • | financial liability that was imposed upon him in favor of another person pursuant to a judgment, including a compromise judgment or an arbitrator’s award approved by a court; |
| • | reasonable litigation expenses, including attorneys’ fees paid by an officeholder following an investigation or proceeding conducted against him by an authority authorized to conduct such investigation or proceeding, and which ended without the filing of an indictment against him and without any financial obligation being imposed on him as an alternative to a criminal proceeding, or which ended without the filing of an indictment against him but with the imposition of a financial obligation as an alternative to a criminal proceeding for an offense which does not require proof of mens rea or in connection with a financial sanction; |
| • | reasonable litigation expenses, including attorneys’ fees paid by the officeholder or which he was required to pay by a court, in a proceeding filed against him by the Company or on its behalf or by another person, or in criminal charges from which he was acquitted, or in criminal charges in which he was convicted of an offense which does not require proof of mens rea; |
| • | certain compensation payments made to an injured party imposed on an office holder by an administrative proceeding, pursuant to certain provisions of the Israeli Securities Law; |
| • | expenses incurred by an officeholder in connection with an administrative proceeding conducted in such officer holder’s regard, including reasonable litigation expenses, and including attorneys’ fees; and |
| • | any other liability or expense in respect of which it is permitted or shall be permitted by Law to indemnify an officeholder. |
| • | Breach of the duty of care to the Company or to a third party, including a breach arising out of the negligent conduct of the office holder; |
| • | Breach of the duty of care to the Company, provided that the office holder acted in good faith and had reasonable grounds to believe that the act would not prejudice the company ; |
| • | financial liability imposed upon an office holder in favor of a third party; |
| • | financial liability imposed on the office holder in favor of a third-party harmed by a breach in an administrative proceeding, pursuant to certain provisions of the Israeli Securities Law; |
| • | expenses incurred or to be incurred by an office holder in connection with an administrative proceeding, instituted against him or her, pursuant to certain provisions of the Israeli Securities Law, including reasonable litigation expenses, and including attorneys’ fees; and |
| • | any other event in respect of which it is permitted and/or shall be permitted by Law to insure the liability of an officeholder. |
Under the Companies Law, a company may not indemnify, exculpate or insure an office holder against any of the following:
| • | a breach of the duty of loyalty, except for indemnification and insurance for a breach of the duty of loyalty to the company to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not prejudice the company; |
| • | a breach of duty of care committed intentionally or recklessly, excluding a breach arising out of the negligent conduct of the office holder; |
| • | an act or omission committed with intent to derive illegal personal benefit; or |
| • | a fine, monetary sanction or forfeit levied against the office holder. |
| ● | each person or entity known by us to own beneficially 5% or more of our outstanding Ordinary Shares; |
| ● | each of our directors and executive officers individually; and |
| ● | all of our executive officers and directors as a group. |
Shares Beneficially Owned Prior to Offering | Shares Beneficially Owned After Offering | |||||||||||||||
| Name of Beneficial Owner | Number | Percentage | Number | Percentage | ||||||||||||
| 5% or greater shareholders | ||||||||||||||||
Shimon Cohen(1) | 12,175,320 | 8.3 | % | 12,175,320 | 6.0 | % | ||||||||||
| Directors and executive officers | ||||||||||||||||
Arik Kaufman(2) | 449,930 | * | 449,930 | * | ||||||||||||
Guy Hefer(3) | 291,670 | * | 291,670 | * | ||||||||||||
Dan Kozlovski(4) | 133,340 | * | 133,340 | * | ||||||||||||
Yaron Kaiser(5) | 1,551,940 | 1.1 | % | 1,551,940 | * | |||||||||||
David Gerbi(6) | 199,640 | * | 199,640 | * | ||||||||||||
Eli Arad(7) | 148,140 | * | 148,140 | * | ||||||||||||
Sari Singer(8) | 152,100 | * | 152,100 | * | ||||||||||||
| All directors and executive officers as a group (7 persons) | 2,926,760 | 2.0 | % | 2,926,760 | 1.4 | % | ||||||||||
| (1) | This information is based solely on a Schedule 13D filed with the SEC on September 22, 2022, pursuant to which Shimon Cohen reported that he is the direct and beneficial owner of 12,175,320 Ordinary Shares, which represents (i) 305,616 ADSs held by Mr. Cohen in his individual capacity and (ii) 437,245 ADSs, 222,068 ADSs and 252,603 ADSs held indirectly by Mr. Cohen through S.C. Ma’agarei Enosh Ltd., Reshet Bitachon Ltd. and Ma’agarim Proyektim Ltd., respectively, each of which Mr. Cohen is the sole owner, manager and shareholder. The address for Shimon Cohen is 20 Derech HaShalom, Tel Aviv, 61250 Israel. |
| (2) | Consists of 283,270 Ordinary Shares and options to purchase 166,660 Ordinary Shares exercisable within 60 days of the date of this prospectus, with an exercise price of $0.519. These options expire on March 16, 2026. |
| (3) | Consists of options to purchase 166,670 Ordinary Shares exercisable within 60 days of the date of this prospectus, with an exercise price of NIS 3.49 ($0.99), expiring on March 24, 2025, and options to purchase 125,000 Ordinary Shares exercisable within 60 days of the date of this prospectus, with an exercise price of $0.716, expiring on July 20, 2025. |
| (4) | Consists of options to purchase 133,340 Ordinary Shares exercisable within 60 days of the date of this prospectus, with an exercise price of NIS 1.90 ($0.54). These options expire on August 5, 2024 |
| (5) | Consists of 1,435,280 Ordinary Shares based on information provided to us by Mr. Kaiser, and options to purchase 116,660 Ordinary Shares exercisable within 60 days of the date of this prospectus, with an exercise price of $0.519. These options expire on March 16, 2026. |
| (6) | Consists of 93,950 Ordinary Shares, RSUs vesting into 7,490 Ordinary Shares within 60 days of the date of this prospectus and options to purchase 98,200 Ordinary Shares within 60 days of the date of this prospectus with an exercise price of $0.716. These options expire on July 20, 2025. |
| (7) | Consists of 42,450 Ordinary Shares, RSUs vesting into 7,490 Ordinary Shares within 60 days of the date of this prospectus and options to purchase 98,200 Ordinary Shares within 60 days of the date of this prospectus with an exercise price of $0.716. These options expire on July 20, 2025. |
| (8) | Consists of 44,910 Ordinary Shares, and RSUs vesting into 8,990 Ordinary Shares within 60 days of the date of this prospectus and options to purchase 98,200 Ordinary Shares within 60 days of the date of this prospectus with an exercise price of $0.716. These options expire on July 20, 2025. |
| • | amendments to our articles of association; |
| • | appointment, terms of service or and termination of service of our auditors; |
| • | appointment of directors, including external directors (if applicable); |
| • | approval of certain related party transactions; |
| • | increases or reductions of our authorized share capital; |
| • | a merger; and |
| • | the exercise of our board of directors’ powers by a general meeting, if our board of directors is unable to exercise its powers and the exercise of any of its powers is required for our proper management. |
| • | the Class I directors are Messrs. Eli Arad and David Gerbi and their respective terms will expire at the Company’s annual general meeting of shareholders to be held in 2023; |
| • | the Class II director is Ms. Sari Singer and her term will expire at the Company’s annual general meeting of shareholders to be held in 2024; and |
| • | the Class III director is Mr. Yaron Kaiser his term will expire at the Company’s annual general meeting of shareholders to be held in 2025. |
| Persons depositing or withdrawing ordinary shares or ADS holders must pay | | For |
| $5.00 (or less) per 100 ADSs (or portion of 100 ADSs) | | Issuance of ADSs, including issuances resulting from a distribution of ordinary shares or rights or other property cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates |
| $.05 (or less) per ADS | | Any cash distribution to ADS holders |
| A fee equivalent to the fee that would be payable if securities distributed to you had been ordinary shares and the ordinary shares had been deposited for issuance of ADSs | | Distribution of securities distributed to holders of deposited securities (including rights) that are distributed by the depositary to ADS holders |
| $.05 (or less) per ADS per calendar year | | Depositary services |
| Persons depositing or withdrawing ordinary shares or ADS holders must pay | | For |
| Registration or transfer fees | | Transfer and registration of ordinary shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw ordinary shares |
| Expenses of the depositary | | Cable (including SWIFT), telex and facsimile transmissions (when expressly provided in the deposit agreement) Converting foreign currency to U.S. dollars |
| Taxes and other governmental charges the depositary or the custodian has to pay on any ADSs or ordinary shares underlying ADSs, such as stock transfer taxes, stamp duty or withholding taxes | | As necessary |
| Any charges incurred by the depositary or its agents for servicing the deposited securities | | As necessary |
| • | 60 days have passed since the depositary told us it wants to resign but a successor depositary has not been appointed and accepted its appointment; |
| • | we delist the ADSs from an exchange in the United States on which they were listed and do not list them on another exchange in the United States or list our Ordinary Shares on an exchange outside the United States and make arrangements for trading of ADSs on the U.S. over the counter market within a reasonable time; |
| • | the depositary has reason to believe the ADSs have become, or will become, ineligible for registration on Form F-6 under the Securities Act; |
| • | we appear to be insolvent or enter insolvency proceedings; |
| • | all or substantially all the value of the deposited securities has been distributed either in cash or in the form of securities; |
| • | there are no deposited securities underlying the ADSs or the underlying deposited securities have become apparently worthless; or |
| • | there has been a replacement of deposited securities. |
| • | are only obligated to take the actions specifically set forth in the deposit agreement without negligence or bad faith, and the depositary will not be a fiduciary or have any fiduciary duty to holders of ADSs; |
| • | are not liable if we are or it is prevented or delayed by law or circumstances beyond our or its control from performing our or its obligations under the deposit agreement; |
| • | are not liable if we exercise or it exercises discretion permitted under the deposit agreement; |
| • | are not liable for the inability of any holder of ADSs to benefit from any distribution on deposited securities that is not made available to holders of ADSs under the terms of the deposit agreement, or for any special, consequential or punitive damages for any breach of the terms of the deposit agreement; |
| • | have no obligation to become involved in a lawsuit or other proceeding related to the ADSs or the deposit agreement on your behalf or on behalf of any other person; |
| • | are not liable for the acts or omissions of any securities depository, clearing agency or settlement system; |
| • | may rely upon any documents we believe or it believes in good faith to be genuine and to have been signed or presented by the proper person; and |
| • | the depositary has no duty to make any determination or provide any information as to our tax status, or any liability for any tax consequences that may be incurred by ADS holders as a result of owning or holding ADSs or be liable for the inability or failure of an ADS holder to obtain the benefit of a foreign tax credit reduced rate of withholding or refund of amounts withheld in respect of tax or any other tax benefit. |
| • | payment of stock transfer or other taxes or other governmental charges and transfer or registration fees charged by third parties for the transfer of any Ordinary Shares or other deposited securities; |
| • | satisfactory proof of the identity and genuineness of any signature or other information it deems necessary; and |
| • | compliance with regulations it may establish, from time to time, consistent with the deposit agreement, including presentation of transfer documents. |
| • | when temporary delays arise because: (i) the depositary has closed its transfer books or we have closed our transfer books; (ii) the transfer of Ordinary Shares is blocked to permit voting at a shareholders’ meeting; or (iii) we are paying a dividend on our shares; |
| • | when you owe money to pay fees, taxes and similar charges; or |
| • | when it is necessary to prohibit withdrawals in order to comply with any laws or governmental regulations that apply to ADSs or to the withdrawal of Ordinary Shares or other deposited securities. |
| • | 1% of the number of Ordinary Shares or ADSs then outstanding; or | |
| • | the average weekly trading volume of our or ADSs on Nasdaq during the four calendar weeks preceding the filing of a notice on Form 144 with respect to the sale; provided that current public information about us is available and the affiliate complies with the manner of sale requirements imposed by Rule 144. |
Capital gains
| • | an individual who is a citizen or resident of the United States; |
| • | a domestic corporation (or other entity taxable as a corporation); |
| • | an estate the income of which is subject to United States federal income taxation regardless of its source; or |
| • | a trust if (1) a court within the United States is able to exercise primary supervision over the trust’s administration and one or more United States persons have the authority to control all substantial decisions of the trust or (2) a valid election under the Treasury regulations is in effect for the trust to be treated as a United States person. |
| • | such gain is effectively connected with your conduct of a trade or business in the United States (or, if required by an applicable income tax treaty, the gain is attributable to a permanent establishment or fixed base that such holder maintains in the United States); or |
| • | you are an individual and have been present in the United States for 183 days or more in the taxable year of such sale or exchange and certain other conditions are met. |
| Name of Underwriter | Number of ADSs and Warrants | Number of Pre-Funded Warrants and Warrants | ||||||
| A.G.P./Alliance Global Partners | ||||||||
| Maxim Group LLC | ||||||||
| Per ADS | Per Pre-Funded Warrant | Total without over-allotment option | Total with full over-allotment option | |||||||||||||
| Public offering price | $ | $ | $ | |||||||||||||
Underwriting discounts and commissions to be paid by us (7.0%)(1) | $ | $ | $ | |||||||||||||
| Non-accountable expense allowance | ||||||||||||||||
| Proceeds, before expenses, to us | $ | $ | $ | |||||||||||||
| (1) | We have agreed to pay a non-accountable expense (“NAE”) allowance to the underwriter up to an amount that shall not exceed $25,000, including, but not limited to, IPREO software related expenses, background check(s), tombstones, marketing related expenses; i.e. roadshow, travel, et al. and any other expenses incurred by the underwriter in connection with the offering. The total NAE allowance shall not exceed $25,000. |
| • | to legal entities which are qualified investors as defined under the Prospectus Directive; |
| • | by the underwriter to fewer than 150, natural or legal persons (other than qualified investors as defined in the Prospectus Directive), as permitted under the Prospectus Directive, subject to obtaining the prior consent of the representatives of the underwriter for any such offer; or |
| • | in any other circumstances falling within Article 3(2) of the Prospectus Directive, provided that no such offer of our common stock shall result in a requirement for us or any underwriter to publish a prospectus pursuant to Article 3 of the Prospectus Directive. |
| Itemized expense | Amount | |||
| SEC registration fee | $ | 2,053 | ||
| FINRA filing fee | $ | 4,867 | ||
| Legal fees and expenses | $ | 120,200 | ||
| Printing and engraving expenses | $ | 10,000 | ||
| Accounting fees and expenses | $ | 40,000 | ||
| Miscellaneous | $ | 10,000 | ||
| Total | $ | 187,120 | ||

Financial Information of MeaTech 3D Ltd. | Page |
Report of Independent Registered Public Accounting Firm (PCAOB ID No. 1057) | F-2 |
Consolidated Financial Statements: | |
F-3 | |
F-4 | |
F-5 | |
F-6 | |
F-7 |
To the Shareholders and Board of Directors
MeaTech 3D Ltd.
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated statements of financial position of MeaTech 3D Ltd. (“the Company”) as of December 31, 2021 and 2020, the related consolidated statements of income and comprehensive loss, changes in equity, and cash flows for each of the years in the three-year period ended December 31, 2021, and the related notes (collectively, the consolidated financial statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2021 and 2020, and the results of its operations and its cash flows for each of the years in the three-year period ended December 31, 2021, in conformity with International Financial Reporting Standards, as issued by the International Accounting Standards Board.
Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1c to the consolidated financial statements, the Company has incurred recurring losses from operations that, together with other matters described in the aforesaid note, raise substantial doubt about its ability to continue as a going concern Management’s plans in regard to these matters are also described in Note 1c. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty
Basis for Opinion
These consolidated financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Somekh Chaikin
Somekh Chaikin
Member Firm of KPMG International
We have served as the Company’s auditor since 2020.
Tel Aviv, Israel
March 24, 2022
December 31 | December 31 | ||||||||||
2021 | 2020 | ||||||||||
USD thousands | USD thousands | ||||||||||
Current assets | |||||||||||
Cash and cash equivalents | 4 | 19,176 | 13,556 | ||||||||
Other investment | 6 | 154 | 149 | ||||||||
Receivables and prepaid expenses | 5 | 2,782 | 131 | ||||||||
Total current assets | 22,112 | 13,836 | |||||||||
Non-current assets | |||||||||||
Restricted deposits | 405 | 51 | |||||||||
Other investment | 6 | 1,355 | 2,513 | ||||||||
Right-of-use asset | 19 | 407 | 168 | ||||||||
| Intangible assets | 16 | 13,453 | - | ||||||||
Fixed assets, net | 7 | 2,922 | 906 | ||||||||
Total non-current assets | 18,542 | 3,638 | |||||||||
Total Assets | 40,654 | 17,474 | |||||||||
Current liabilities | |||||||||||
Trade payables | 382 | 351 | |||||||||
Other payables | 8 | 2,239 | 996 | ||||||||
Current maturities of lease liabilities | 19 | 165 | 180 | ||||||||
Derivative instrument | - | 316 | |||||||||
Total current liabilities | 2,786 | 1,843 | |||||||||
Non-current liabilities | |||||||||||
Long-term lease liabilities | 19 | 246 | - | ||||||||
Total non-current liabilities | 246 | - | |||||||||
Equity | |||||||||||
Share capital and premium on shares | 69,610 | 30,481 | |||||||||
Capital reserves | 3,708 | 3,319 | |||||||||
Currency translation differences reserve | 1,275 | 780 | |||||||||
Accumulated deficit | (36,971 | ) | (18,949 | ) | |||||||
Total Equity | 37,622 | 15,631 | |||||||||
Total liabilities and Equity | 40,654 | 17,474 | |||||||||
F - 3
MeaTech 3D Ltd.
Year ended December 31, | Year ended December 31, | Year ended December 31, | |||||||||||||
2021 | 2020 | 2019 | |||||||||||||
USD thousands, except share data | USD thousands, except share data | USD thousands, except share data | |||||||||||||
Research and development expenses | 11 | 7,594 | 2,491 | 166 | |||||||||||
Marketing expenses | 12 | 1,628 | 506 | - | |||||||||||
General and administrative expenses | 13 | 8,010 | 5,380 | 256 | |||||||||||
Public listing expenses | - | 10,164 | - | ||||||||||||
Operating loss | 17,232 | 18,541 | 422 | ||||||||||||
Financing income | 14 | 509 | 110 | - | |||||||||||
Financing expenses | 14 | 1,299 | 93 | 1 | |||||||||||
Loss for the year | 18,022 | 18,524 | 423 | ||||||||||||
Capital reserve for financial assets at fair value that will not be transferred to profit or loss | - | 334 | - | ||||||||||||
Currency translation differences loss (income) that will not be transferred to profit or loss over ILS | (1,942 | ) | (758 | ) | (22 | ) | |||||||||
Currency translation differences loss (income) that might be transferred to profit or loss over EUR | 1,447 | - | - | ||||||||||||
Total comprehensive loss for the year | 17,527 | 18,100 | 401 | ||||||||||||
Loss per ordinary share, no par value (USD) | |||||||||||||||
Basic and diluted loss per share (USD) | 0.155 | 0.308 | 0.022 | ||||||||||||
Weighted-average number of shares outstanding - basic and diluted (shares) | 115,954,501 | 60,112,197 | 19,484,478 | ||||||||||||
F - 4
Share capital and capital premium | Fair value of financial assets reserve | Transactions with related parties reserve | Currency translation differences reserve | Share-based payments reserve | Accumulated deficit | Total | ||||||||||||||||||||||
USD thousands | ||||||||||||||||||||||||||||
Balance as at January 1, 2021 | 30,481 | (334 | ) | 14 | 780 | 3,639 | (18,949 | ) | 15,631 | |||||||||||||||||||
Share-based payments | - | - | - | - | 3,965 | - | 3,965 | |||||||||||||||||||||
Issuance of shares and warrants, net | 32,330 | - | - | - | - | - | 32,330 | |||||||||||||||||||||
Exercise of options | 6,799 | - | - | - | (3,576 | ) | - | 3,223 | ||||||||||||||||||||
Other comprehensive income | - | - | - | 495 | - | - | 495 | |||||||||||||||||||||
Loss for the year | - | - | - | - | - | (18,022 | ) | (18,022 | ) | |||||||||||||||||||
Balance as at December 31, 2021 | 69,610 | (334 | ) | 14 | 1,275 | 4,028 | (36,971 | ) | 37,622 | |||||||||||||||||||
Balance as at January 1, 2020 | 1,880 | - | 14 | 22 | - | (425 | ) | 1,491 | ||||||||||||||||||||
| Share-Based Payment | - | - | - | - | 3,958 | - | 3,958 | |||||||||||||||||||||
| Reverse acquisition | 11,439 | - | - | - | - | - | 11,439 | |||||||||||||||||||||
Issuance of shares and warrants, net | 14,067 | - | - | - | - | - | 14,067 | |||||||||||||||||||||
Exercise of options – Investors | 2,753 | - | - | - | - | - | 2,753 | |||||||||||||||||||||
Exercise of options – Share-Based Payment | 342 | - | - | - | (319 | ) | - | 23 | ||||||||||||||||||||
Other comprehensive income (loss) | - | (334 | ) | - | 758 | - | - | 424 | ||||||||||||||||||||
Loss for the year | - | - | - | - | - | (18,524 | ) | (18,524 | ) | |||||||||||||||||||
Balance as at December 31, 2020 | 30,481 | (334 | ) | 14 | 780 | 3,639 | (18,949 | ) | 15,631 | |||||||||||||||||||
| Balance as at January 1, 2019 | - | - | - | - | - | (2 | ) | (2 | ) | |||||||||||||||||||
Issuance of shares and warrants, net | 1,880 | - | - | - | - | - | 1,880 | |||||||||||||||||||||
Other comprehensive income | - | - | - | 22 | - | - | 22 | |||||||||||||||||||||
| Transaction with a related party | - | - | 14 | - | - | - | 14 | |||||||||||||||||||||
Loss for the year | - | - | - | - | - | (423 | ) | (423 | ) | |||||||||||||||||||
Balance as at December 31, 2019 | 1,880 | - | 14 | 22 | - | (425 | ) | 1,491 | ||||||||||||||||||||
F - 5
Year ended December 31, 2021 | Year ended December 31, 2020 | Year ended December 31, 2019 | |||||||||||
USD thousands | USD thousands | USD thousands | |||||||||||
Cash flows - operating activities | |||||||||||||
Net Loss for the period | (18,022 | ) | (18,524 | ) | (423 | ) | |||||||
Adjustments: | |||||||||||||
Depreciation and amortization | 680 | 213 | 21 | ||||||||||
Change in fair value of derivative | (316 | ) | (36 | ) | 14 | ||||||||
Change in fair value of other investment | 6 | (193 | ) | (74 | ) | - | |||||||
Changes in net foreign exchange expenses | 1,279 | - | - | ||||||||||
Share-based payment expenses | 3,965 | 3,958 | - | ||||||||||
Public listing expenses | - | 10,164 | - | ||||||||||
Changes in asset and liability items: | |||||||||||||
Decrease (increase) in receivables | (2,351 | ) | 5 | (36 | ) | ||||||||
Increase (decrease) in trade payables | (97 | ) | 126 | 66 | |||||||||
Increase in other payables | 1,095 | 336 | 185 | ||||||||||
Net cash used in operating activities | (13,960 | ) | (3,832 | ) | (173 | ) | |||||||
Cash flows - investment activities | |||||||||||||
Acquisition of fixed assets | (1,828 | ) | (681 | ) | (126 | ) | |||||||
Increase in restricted deposit | (337 | ) | (6 | ) | (41 | ) | |||||||
Loan provided | (367 | ) | - | (86 | ) | ||||||||
Acquisition of other investments, net of cash acquired | 16 | (6,808 | ) | (1,188 | ) | - | |||||||
| |||||||||||||
Net cash used in investing activities | (9,340 | ) | (1,875 | ) | (253 | ) | |||||||
Cash flows - financing activities | |||||||||||||
Proceeds from issuance of shares and warrants | 29,281 | 14,887 | 1,670 | ||||||||||
Issuance costs | (3,283 | ) | (819 | ) | (8 | ) | |||||||
Repayment of liability for lease | (346 | ) | (140 | ) | (14 | ) | |||||||
Proceeds on account of other investment | 149 | 71 | - | ||||||||||
Proceeds on account of capital issuance | - | 222 | - | ||||||||||
Proceeds with regard to derivative | - | 348 | - | ||||||||||
Proceeds from exercise of share options | 3,222 | 2,776 | - | ||||||||||
Net cash from financing activities | 29,023 | 17,345 | 1,648 | ||||||||||
Increase in cash and cash equivalents | 5,723 | 11,638 | 1,222 | ||||||||||
Effect of exchange differences on cash and cash equivalents | (103 | ) | 644 | 21 | |||||||||
Cash and cash equivalents at the beginning of the period | 13,556 | 1,274 | 31 | ||||||||||
Cash balance and cash equivalents at end of period | 19,176 | 13,556 | 1,274 | ||||||||||
Non cash activities | |||||||||||||
Purchase of fixed assets | 57 | 143 | 1 | ||||||||||
Issue of shares and options against intangible asset | 6,332 | - | 222 | ||||||||||
| A. | Reporting entity |
MeaTech 3D Ltd. (formerly Ophectra Real Estate and Investments Ltd. and Meat-Tech 3D Ltd.) (the “Company”) was incorporated in Israel on July 22, 1992 as a private company limited by shares in accordance with the Companies Ordinance, 1983, and later a publicly-traded company whose ordinary shares were listed for trade on the Tel Aviv Stock Exchange (TASE). The Company’s official address is 5 David Fikes St., Rehovot, Israel.
The Company’s foodtech activities were commenced in July 2019 by a company called MeaTech Ltd., which merged with the Company in January 2020 and became a fully-owned subsidiary, now called MeaTech MT Ltd. As the Company was the surviving entity of the merger, and continued the pre-merger business operations, utilizing the pre-merger management and employees, of MeaTech Ltd., the transaction was treated as a reverse acquisition that does not constitute a business combination.
The Company is developing a suite of advanced high-throughput manufacturing technologies to produce cell-based alternative protein products for cultivated, sustainable meat production, and focused on developing premium, center-of-plate meat products, including development of high-throughput bioprinting systems.
| B. | Material events in the reporting period |
| (1) | Acquisition of subsidiary |
In February 2021, the Company acquired Belgian cultured fat developer Peace of Meat BV. For further details, see Note 16 below.
| (2) | Initial public offering |
On March 12, 2021, the Company completed its Initial Public Offering on the Nasdaq of 2,721,271 ADSs, each representing ten ordinary shares of the Company (in total, 27,212,710 ordinary shares), at an offering price of USD 10.30 per ADS, resulting in gross proceeds of USD 28 million and net proceeds of USD 24.7 million. The ADSs trade on the Nasdaq Capital Market under the symbol “MITC.” On August 5, 2021, the Company completed the voluntary de-listing of its ordinary shares from the TASE. A number of ordinary shares remain traded over the counter (OTC:MTTCF). Additionally, the vesting of 1,374,998 investor share rights was triggered by the IPO, and these shares were issued in return for USD 1.25 million following the IPO.
| (3) | Effects of the spread of COVID-19 |
To date, the impact of the COVID-19 pandemic on the Company’s operations has been mainly limited to a temporary facility closure in the context of a government-mandated general lockdown, which temporary delayed certain development activities. The Company estimates that as of the date of approval of the financial statements, the COVID-19 pandemic is not expected to affect the Company's operations. However, the Company is unable to assess with certainty the extent of future impact, in part due to the uncertainty regarding the duration of the COVID-19 pandemic, its force and its effects on the markets in which the Company operates and the effects of possible government measures to prevent the spread of the virus. |
F - 7
Note 1 – General (cont.)
| C. | Going Concern |
Since inception, the Company has incurred significant losses and negative cash flows from operations and has an accumulated deficit of USD 37.0 million. The Company has financed its operations mainly through fundraising from various investors.
The Company’s management expects that the Company will continue to generate losses and negative cash flows from operations for the foreseeable future. Based on the projected cash flows and cash balances as of December 31, 2021, management is of the opinion that its existing cash will be sufficient to fund operations until Q4 2022. As a result, there is substantial doubt about the Company’s ability to continue as a going concern.
Management’s plans include the continue securing sufficient financing through the sale of additional equity securities or capital inflows from strategic partnerships. Additional funds may not be available when the Company needs them on terms that are acceptable to it, or at all. If the Company is unsuccessful securing sufficient financing, it may need to cease operations.
The financial statements include no adjustments for measurement or presentation of assets and liabilities, which may be required should the Company fail to operate as a going concern.
| D. | Definitions: |
(1) The Company - MeaTech 3D Ltd.
(2) The Group – The Company and its subsidiaries, MeaTech MT Ltd., formerly known as MeaTech Ltd, Meatech Europe BV and Peace of Meat B.V. (hereafter “Peace Of Meat” OR “POM”)
(3) Related Party - as defined in IAS 24 (revised).
(4) USD - United States Dollar
(5) NIS – New Israeli Shekel
(6) EUR – Euro
(7) ADS – American Depositary Shares
F - 8
| A. | Statement of compliance with IFRS |
The financial statements were authorized for issue by the company’s board of directors on March 24, 2022.
| B. | Functional currency and presentation currency |
Currency | USD - ILS | USD - EUR | ||||||||||||
Period | 2021 | 2020 | 2019 | 2021 | ||||||||||
December 31 | 3.110 | 3.215 | 3.446 | 0.883 | ||||||||||
Year Average | 3.230 | 3.479 | 3.442 | 0.845 | ||||||||||
| C. | Basis of Measurement |
| D. | Operating Cycle |
| E. | Use of Estimates and Judgments |
F - 9
| E. | Use of Estimates and Judgments (cont.) |
The preparation of accounting estimates used in the preparation of the Company’s financial statements requires that the Company’s management makes assumptions regarding circumstances and events that involve considerable uncertainty. The Company’s management prepares the estimates on the basis of past experience, various facts, external circumstances, and reasonable assumptions according to the pertinent circumstances of each estimate. Estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimates are revised and in any future periods affected.
Further information about the assumptions that were used to determine fair value is included in the following notes:
• | Note 6, on other investments; |
• | Note 10, on share-based payments; |
• | Note 16, on intangible assets; |
Determination of fair value
• | Level 1: quoted prices (unadjusted) in active markets for identical assets or liabilities. |
• | Level 2: inputs other than quoted prices included within Level 1 that are observable, either directly or indirectly |
• | Level 3: inputs that are not based on observable market data (unobservable inputs). |
F - 10
| A. | Financial Instruments: |
| (1) | Non-derivative financial assets |
| - | It is held within a business model whose objective is to hold assets so as to collect contractual cash flows; and |
| - | The contractual terms of the financial asset give rise to cash flows representing solely payments of principal and interest on the principal amount outstanding on specified dates. |
| (2) | Non-derivative financial liabilities |
F - 11
| (3) | Share capital |
| (4) | Issuance of securities |
| B. | Impairment |
Non-derivative financial assets
See note 6.
| C. | Financing income and expenses |
F - 12
Note 3 – Significant Accounting Policies (cont.)
| D. | Loss per share |
| E. | Intangible Assets |
Subsequent expenditure
Subsequent expenditure is capitalized only when it increases the future economic benefits embodied in the specific asset to which it relates. All other expenditure, including expenditure on internally generated goodwill and brands, is recognized in profit or loss as incurred.
Other intangible assets
Other intangible assets, that are acquired by the Company, are measured at cost less accumulated amortization and accumulated impairment losses. See Note 16.
| F. | Provisions |
| G. | Fixed assets |
| (1) | Recognition and measurement |
| (2) | Depreciation |
F - 13
Note 3 – Significant Accounting Policies (cont.)
| G. | Fixed assets (cont.) |
An asset is depreciated from the date it is ready for use, namely, the date on which it reaches the location and condition required for it to operate in the manner intended by Management.
| ● | Computers | 3 years | |
| ● | Leasehold improvements | 2 years | |
| ● | Laboratory equipment | 5-7 years | |
● | Machinery and Equipment | 6-10 years | |
● | Office furniture, equipment and accessories | 14 years |
| H. | Leases |
| (a) | The right to obtain substantially all the economic benefits from use of the identified asset; and |
| (b) | The right to direct the identified asset’s use. |
F - 14
Note 3 – Significant Accounting Policies (cont.)
| H. | Leases (cont.) |
| I. | Employee benefits |
| (1) | Post-employment benefits |
| (2) | Short-term benefits |
| J. | Share-based compensation |
F - 15
Note 3 – Significant Accounting Policies (cont.)
| K. | Basis of Consolidation |
Acquisition of a subsidiary
Upon the acquisition of a subsidiary, the Company exercises discretion when examining whether the transaction constitutes the acquisition of a business or acquisition of an asset, for the purpose of determining the accounting treatment of the transaction. Transactions in which the acquired company is not considered a business are accounted for as the acquisition of a group of assets and liabilities. In such transactions, the cost of acquisition, which includes transaction costs, is allocated proportionately to the acquired identifiable assets and liabilities, based on their proportionate fair value on the acquisition date. Furthermore, no goodwill is recognized and no deferred taxes are recognized in respect of the temporary differences existing on the acquisition date.
Consideration paid partly in the form of equity instruments, based on the quoted share price. Any additional consideration will be capitalized upon the achievement of defined milestones, which constitutes the variable consideration.
When the variable consideration depends on performance conditions, the Company has elected not to recognize the contingent consideration at the time of purchase, but rather if and when the contingent conditions occur and when the consideration is transferred or obliged to be transferred.
IFRS 3 includes a distinction between a transaction to acquire an operation is the acquisition of a "business" and the acquisition of a group of assets that according to the standard is not considered the acquisition of a "business". The aforementioned standard offers the optional concentration test so that if substantially all of the fair value of the acquired assets is attributable to a group of similar identifiable assets or to a single identifiable asset, this will not be the acquisition of a business
Transactions eliminated on consolidation
Intra-group balances and transactions, and any unrealized income and expenses arising from intra-group transactions, are eliminated in preparing the consolidated financial statements.
| L. | Transactions with controlling shareholder |
Assets and liabilities included in a transaction with a controlling shareholder are measured at fair value on the date of the transaction. As the transaction is on the equity level, the Company includes the difference between the fair value and the consideration from the transaction in its equity.
| M. | Government grants |
Government grants are recognized initially at fair value when there is reasonable assurance that they will be received and the Group will comply with the conditions associated with the grant. Unconditional government grants are recognized when the Group is entitled to receive them. Grants that compensate the Group for expenses incurred are presented as a deduction from the corresponding expense. Grants that compensate the Group for the cost of an asset are presented as a deduction from the related assets and are recognized in profit or loss on a systematic basis over the useful life of the asset.
F - 16
December 31 | December 31 | |||||||
2021 | 2020 | |||||||
USD thousands | USD thousands | |||||||
Cash in USD | 15,596 | 1,021 | ||||||
Cash in NIS | 1,688 | 7,627 | ||||||
Cash in Euro | 1,892 | 4,908 | ||||||
Total cash and cash equivalents | 19,176 | 13,556 | ||||||
December 31 | December 31 | |||||||
2021 | 2020 | |||||||
USD thousands | USD thousands | |||||||
Institutions | 301 | 102 | ||||||
Prepaid expenses | 743 | 20 | ||||||
Other | 1,738 | 9 | ||||||
2,782 | 131 | |||||||
December 31 | December 31 | |||||||
2021 | 2020 | |||||||
USD thousands | USD thousands | |||||||
Separation Agreement from Therapin (A) (1) | 1,509 | 1,414 | ||||||
Investment in Peace of Meat (B) | - | 1,248 | ||||||
Total Other Investments | 1,509 | 2,662 | ||||||
Developments in Other Investment | USD thousands | |||
| ||||
As at January 1, 2021 | 2,662 | |||
Initial investment in POM transferred to intangible asset | (1,223 | ) | ||
Proceeds from Therapin asset | (149 | ) | ||
Profit from increase in fair value | 193 | |||
Effect of changes in exchange rates | 26 | |||
As at December 31, 2021(1) | 1,509 |
(1) USD 154 thousand are classified as a current asset.
F - 17
| A. | Separation Agreement from Therapin |
| 1. | Therapin committed to pay to the Company an amount of USD 13 thousand (NIS 40 thousand) per month, thereafter as of August 1, 2020 over a period of 119 months (the “Payment Period”), for an aggregate total amount of USD 1.4 million (NIS 4.8 million). During the first two years from the date of the separation agreement, 50% of the payments from Therapin will be transferred to a restricted deposit and form an additional resource of the Merger settlement fund. After two years, the contents of the restricted deposit, will be released to the Company, subject to court approval. |
| 2. | The rest of the Payment Amount will be paid to the Company if, during the Payment Period, Therapin or a subsidiary completes an exit event, including listing on a stock exchange pursuant to a merger or IPO, and the Company will be given the option to receive shares in such merged company/issue, or payment of the remaining balance in cash. |
| 3. | During the Payment Period, if Therapin has not completed one of the transactions as set out in Section 2, then in the event that Therapin generates a distributable surplus, Therapin will pay the Company an amount equivalent to 14.74% of the surplus balance as repayment on account of the outstanding balance (but in any case no more than the outstanding balance). |
| 4. | In the event that, during or subsequent to the end of the Payment Period, Therapin distributes a dividend to its shareholders, and on that date there is a remaining outstanding balance of the Payment Amount, Therapin will pay the Company an amount equivalent to 14.74% of the dividend distributed to shareholders as repayment on account of the outstanding balance (but in any case no more than the outstanding balance). |
| 5. | As a result of the separation agreement, the Company is no longer a shareholder in Therapin, but rather a debtholder. |
The Company re-measured the asset using a fair value measurement at approximately USD 1.3 million (NIS 4.5 million). The fair value was assessed by capitalization of future cash flows (proceeds) at interest rates that reflect the level of risk (based on the duration of the debt) of these proceeds and were classified as Level 3 in the fair value hierarchy. The estimated capitalization interest was based on Therapin's financial statements, cash balances and liabilities, repayment dates, and analysis of the market in which Therapin operates. The expected additional payment event is 4.2 years, and the interest rate for capitalization of the debt is 10.23%-10.72%.
The revaluation was accounted for in other comprehensive income in the amount of USD 0.3 million (NIS 1.2 million). Any change in the fair value following the separation date, will be recognized through profit or loss.
During 2021, the Company received USD 149 thousand based on the agreement detailed above and recorded USD 193 thousand as re-valuation financing income in profit and loss.
| B. | Investment in Peace Of Meat (‘POM’) |
F - 18
| Computers | Leasehold improvements | Laboratory equipment | Machinery and equipment | Office furniture, equipment and accessories | Total | ||||||||||||||||||||||
USD thousands | ||||||||||||||||||||||||||||
Cost |
|
|
|
|
|
| ||||||||||||||||||||||
Balance as at January 1, 2020 | 29 | 11 | 89 | - | 1 | 130 | ||||||||||||||||||||||
Additions during the year | 43 | 46 | 466 | 243 | 27 | 825 | ||||||||||||||||||||||
Effect of changes in exchange rates | 2 | 1 | 18 | - | 1 | 22 | ||||||||||||||||||||||
Dispositions in the year | - | - | - | 1 | 1 | |||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Cost as at December 31, 2020 | 74 | 58 | 573 | 243 | 28 | 976 | ||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Accumulated depreciation | ||||||||||||||||||||||||||||
Balance as at January 1, 2020 | 2 | - | 1 | - | - | 3 | ||||||||||||||||||||||
Depreciation during the year | 15 | 10 | 37 | 4 | 1 | 67 | ||||||||||||||||||||||
Dispositions in the year | - | - | - | - | - | - | ||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Accumulated depreciation as at December 31, 2020 | 17 | 10 | 38 | 4 | 1 | 70 | ||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Depreciated balance as at December 31, 2020 | 57 | 48 | 535 | 239 | 27 | 906 | ||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Balance as at January 1, 2021 | 74 | 58 | 573 | 243 | 28 | 976 | ||||||||||||||||||||||
Additions through acquisition of a subsidiary | 14 | 3 | 556 | - | - | 573 | ||||||||||||||||||||||
Additions during the year | 98 | 75 | 1,608 | 77 | 27 | 1,885 | ||||||||||||||||||||||
Effect of changes in exchange rates | 2 | (1 | ) | (56 | ) | 13 | 2 | (40 | ) | |||||||||||||||||||
| ||||||||||||||||||||||||||||
Cost as at December 31, 2021 | 188 | 135 | 2,681 | 333 | 57 | 3,394 | ||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Accumulated depreciation | ||||||||||||||||||||||||||||
Balance as at January 1, 2021 | 17 | 10 | 38 | 4 | 1 | 70 | ||||||||||||||||||||||
Depreciation during the year | 42 | 22 | 296 | 31 | 3 | 394 | ||||||||||||||||||||||
Effect of changes in exchange rates | 2 | 1 | 4 | 1 | - | 8 | ||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Accumulated depreciation as at December 31, 2021 | 61 | 33 | 338 | 36 | 4 | 472 | ||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Depreciated balance as at December 31, 2021 | 127 | 102 | 2,343 | 297 | 53 | 2,922 | ||||||||||||||||||||||
During the year ended December 31, 2021, the Company acquired fixed assets on credit in the amount of USD 57 thousand (2020: USD 143 thousand). The cost of acquisition had not yet been paid at the reporting date.
F - 19
December 31 | December 31 | ||||||||||
2021 | 2020 | ||||||||||
USD thousands | USD thousands | ||||||||||
Accrued expenses | 459 | 263 | |||||||||
Employee benefits | 1,122 | 503 | |||||||||
Contingent liability | 217 | 217 | |||||||||
| Subsidiary government grant | 218 | - | |||||||||
Others | 223 | 13 | |||||||||
2,239 | 996 | ||||||||||
Number of Ordinary Shares (thousand) | ||||||||||||
2021 | 2020 | 2019 | ||||||||||
Issued and paid-in share capital as at January 1 | 79,866 | 19,870 | 15,447 | |||||||||
Issued in reverse merger | - | 30,526 | - | |||||||||
Exercise of share options during the period – Investor-related | 3,010 | 11,302 | 2,255 | |||||||||
Exercise of share options during the period – Share-Based Payment-related | 2,218 | 294 | - | |||||||||
Issued not for cash during the period (1) | 12,088 | - | - | |||||||||
Issued for cash during the period (2) | 28,588 | 17,874 | 2,168 | |||||||||
Issued and paid-in share capital as at December 31, 2021 | 125,770 | 79,866 | 19,870 | |||||||||
Authorized share capital | 1,000,000 | 1,000,000 | 1,000,000 | |||||||||
| (1) | In February 2021, the Company completed a purchase of all of the outstanding share capital not yet owned by the Company, of Belgian cultured fat developer Peace of Meat BV. See Note 16 for information regarding the issuance of shares as part of the consideration. As part of the IPO, options and rights previously held by the Company's founders and Series A Investors were issued to 6,359,480 shares. |
| (2) | On March 12, 2021, the Company completed its Initial Public Offering (IPO) at the Nasdaq of 2,721,271 ADSs, each representing ten ordinary shares of the Company (in total, 27,212,710 ordinary shares), at an offering price of USD 10.30 per ADS, resulting in gross proceeds of USD 28 million and net proceeds of USD 24.7 million. Additionally, the vesting of 1,374,998 investor share rights was triggered by the IPO, and these shares were issued in return for USD 1.25 million following the IPO. |
F - 20
Date of grant and eligible recipients | Terms of the instrument | No. of ordinary shares (thousands) | Vesting Conditions | Contractual duration of the instrument (years) | ||||
Options awarded to employees of the Company and subsidiaries on March 25, 2021 | Options exercisable for ordinary shares | 2,600 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
RSUs awarded to directors on March 25, 2021 | The RSUs vest automatically at no exercise price | 90 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Options awarded to employees of the Company and subsidiaries on April 19, 2021 | Options exercisable for ordinary shares | 800 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Options awarded to employees of the Company and subsidiaries on July 22, 2021 | Options exercisable for ordinary shares | 1,362.5 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
RSUs awarded to directors on September 14, 2021 | The RSUs vest automatically at no exercise price | 287.5 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Options awarded to directors on September 14, 2021 | Options exercisable for ordinary shares | 785.6 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Options awarded to the deputy CEO on September 14, 2021 | Options exercisable for ordinary shares | 250 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Options awarded to BlueSoundWaves on October 6, 2021 | Options exercisable for ordinary shares | 6,215.8 | 1/3 after one year and the balance in 8 quarterly tranches, subject to milestone-based acceleration | 10 years | ||||
RSUs awarded to BlueSoundWaves on October 6, 2021 | The RSUs vest automatically at no exercise price | 1,243.1 | 1/3 after one year and the balance in 8 quarterly tranches, subject to milestone-based acceleration | 10 years | ||||
Options awarded to employees of the Company and subsidiaries on November 24, 2021 | Options exercisable for ordinary shares | 925 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Total options/RSUs exercisable/vesting into shares granted in the year ended December 31, 2021 | 14,559.5 |
F - 21
A. | Number and weighted average exercise prices of options and RSUs |
Number of options and RSUs 2021 | Weighted average exercise price 2021 NIS | |||||||
Outstanding at January 1 | 9,505,140 | 2.60 | ||||||
Granted during the year | 14,559,520 | 2.06 | ||||||
Forfeited during the year | 194,673 | 1.63 | ||||||
Exercised during the year(1) | 4,834,730 | 2.55 | ||||||
Outstanding at December 31 | 19,035,257 | 2.20 | ||||||
Exercisable at December 31 | 3,562,192 | 3.07 | ||||||
(1) Partly executed through cashless mechanism
Besides incentive options and RSUs, as of the balance sheet date the Company has issued securities exercisable into 22,866,787 ordinary shares to investors, former shareholders of Peace of Meat (see Note 16) and former Ophectra Real Estate and Investments Ltd. employees prior to the reverse merger (see Note 1A), including investor warrants exercisable into 20,224,191 ordinary shares with exercise prices between NIS 3.03 and NIS 6.00, earn-out rights of former shareholders of Peace of Meat, exercisable into 2,412,596 ordinary shares with no exercise price and prior Ophectra Real Estate and Investments Ltd. employees options exercisable into 230,000 ordinary shares with exercise prices between NIS 1.72 and NIS 2.50.
F - 22
B. | Information on measurement of fair value of share-based payment plans |
Options/RSUs | ||
Fair value at date awarded | NIS 23.0 million (USD 7.0 millions) | |
Parameters taken into account in the fair value calculation: | ||
Share price (NIS at date awarded) | 2.05 - 3.53 | |
Exercise price (NIS unlinked) | 0 - 3.68 | |
| Expected volatility | 73.84% - 93.10% | |
Expected useful life | 4 - 10 years | |
Risk-free interest rate | 0.23% - 1.97% | |
Expected rate of dividend | 0% |
F - 23
Year ended December 31, | Year ended December 31, | Year ended | ||||||||||
2021 | 2020 | 2019 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Salaries, wages and related expenses(1) | 3,425 | 1,369 | 117 | |||||||||
Share-based payment(1) | 911 | 476 | - | |||||||||
Materials | 1,875 | 319 | 20 | |||||||||
Professional services | 403 | 89 | 13 | |||||||||
Registration, drafting and filing of patents | 0 | 25 | 10 | |||||||||
Maintenance, office and software fees | 145 | 116 | - | |||||||||
Depreciation and amortization | 400 | 59 | - | |||||||||
Insurance | 332 | - | - | |||||||||
Others | 103 | 38 | 6 | |||||||||
Total Research and Development Expenses | 7,594 | 2,491 | 166 | |||||||||
(1) | Including expenses in respect of related parties - see Note 18. |
Year ended December 31, | Year ended December 31, | Year ended | ||||||||||
2021 | 2020 | 2019 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Salaries, wages and related expenses | 494 | 255 | - | |||||||||
Share-based payment(1) | 570 | 139 | - | |||||||||
PR, advertisement and professional services | 507 | 91 | - | |||||||||
Maintenance, office and software fees | 22 | 13 | - | |||||||||
Depreciation and amortization | 17 | 3 | - | |||||||||
Others | 18 | 5 | - | |||||||||
Total Marketing Expenses | 1,628 | 506 | - | |||||||||
F - 24
Note 13 – General and Administrative Expenses
Year ended December 31, | Year ended December 31, | Year ended December 31, | ||||||||||
2021 | 2020 | 2019 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Salaries, wages and related expenses(1) | 1,328 | 556 | 107 | |||||||||
Share-based payment(1) | 2,484 | 3,343 | - | |||||||||
Legal and professional services(1) | 1,499 | 991 | 112 | |||||||||
Contingent liability expenses | - | 217 | - | |||||||||
Insurance | 1,837 | - | - | |||||||||
Corporate costs | 343 | 60 | - | |||||||||
Maintenance, office and software fees | 149 | 38 | 10 | |||||||||
Depreciation and amortization | 263 | 151 | 20 | |||||||||
Others | 107 | 24 | 7 | |||||||||
Total General and Administrative Expenses | 8,010 | 5,380 | 256 | |||||||||
| (1) | Including expenses in respect of related parties - see Note 18. |
Year ended December 31, | Year ended December 31, | Year ended December 31, | ||||||||||
2021 | 2020 | 2019 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Financing Income | ||||||||||||
Net change in fair value of financial instruments mandatorily measured at fair value through profit or loss | 509 | 110 | - | |||||||||
Financing Expenses | ||||||||||||
Net foreign exchange loss | 1,279 | 85 | - | |||||||||
Interest expense on lease liabilities | 9 | 5 | 1 | |||||||||
Bank interest and commission expenses | 11 | 3 | - | |||||||||
Total Financial expenses | 1,299 | 93 | 1 | |||||||||
Financing expenses (income), net | 790 | (17 | ) | 1 | ||||||||
F - 25
| A. | Details regarding the tax environment of the Company |
| (1) | Corporate tax rate |
The tax rates applicable to the companies operating in Israel for the years 2020-2021 are 23%.
The tax rates applicable to the companies operating in Belgium for the years 2020-2021 are 25%.
| B. | Tax Assessments |
| C. | Unrecognized carryforward losses and deferred taxes |
As at December 31, 2021, the Group has estimated business losses carried forward in the amount of USD 18.2 million. Under current tax legislation in Israel and Belgium, tax losses do not expire. Deferred tax assets have not been recognized in respect of these items, nor in respect of timing differences for research and development expenses carried forward in the amount of USD 4.3 million, since the Company has not yet established the probability that future taxable profit will be available against which the Company can utilize the benefits.
In February 2021, the Company completed a purchase of all of the outstanding share capital not yet owned by the Company of Belgian cultured fat developer Peace of Meat BV for total consideration of up to EUR 16.3 million (USD 19.9 million). The total consideration payable by the Company in the acquisition consists of both cash and equity instruments to be paid to Peace of Meat shareholders and in legal and finder’s fees. The total consideration is to be paid part as of the closing of the acquisition and part upon the achievement of the defined milestones and sub-milestones. Substantially all of the fair value of the gross assets acquired is concentrated in a single identifiable asset or group of similar identifiable assets (the “intangible asset” or “IPR&D”), thus the subsidiary is not considered a business and the acquisition is accounted as an asset acquisition. Contingent consideration, dependent upon the achievement of technological milestones will be recognized at the time of the achievement of each milestone on the basis of the shares and cash that are payable.
USD thousands | ||||
Cash consideration at closing date | 4,799 | |||
Initial cash investment in acquiree | 1,223 | |||
Equity instruments issued (4,070,766 ordinary shares) (1) | 4,359 | |||
Acquisition-related costs (2) | 254 | |||
Total consideration as of consolidation date | 10,635 | |||
Contingent consideration (3) | 9,308 | |||
Total consideration subject to achievement of all milestones | 19,943 | |||
F - 26
Note 16 - Subsidiaries (cont.)
| (1) | The fair value of the ordinary shares issued was based on the share price of the Company at the closing date (February 10, 2021) of NIS 3.986 per share. |
| (2) | Acquisition-related costs include legal expenses and finder’s fees |
| (3) | Contingent consideration The Company agreed to pay the selling shareholders and the finder an additional 4,070,766 rights to ordinary shares with a value of USD 4.4 million and cash consideration of USD 4.9 million upon the achievement of defined milestones related to Peace of Meat’s biomass and bioreactor size, density, capacity and production. The acquisition agreement specified that each milestone must be reached within a six-month period, over a total period of two years, which can be extended by up to nine additional months under circumstances set forth in the acquisition agreement. As of the date of approval of these financial statements, Peace of Meat had fully achieved the first two such milestones. No liability is being provisioned before milestones achievement. |
Identifiable assets acquired and liabilities assumed:
Peace Of Meat condensed Balance Sheet | USD thousands | |||
Current assets | 425 | |||
Non-current assets | 588 | |||
Current liabilities | (578 | ) | ||
Non-current liabilities | (16 | ) | ||
Tangible assets net | 419 | |||
F - 27
Note 16 - Subsidiaries (cont.)
Peace Of Meat initial consolidation effect | USD thousands | |||
Closing cash consideration and related acquisition costs | 5,053 | |||
Shares consideration | 4,359 | |||
Initial cash investment in acquiree | 1,223 | |||
Tangible assets, net | (419 | ) | ||
10,216 | ||||
Additional contributions post acquisition date according to milestone achievement: | ||||
Cash consideration | 1,960 | |||
Payment liabilities | 194 | |||
Shares consideration (1,852,730 ordinary shares) | 1,973 | |||
Total | 14,343 | |||
FX rate effect | (890 | ) | ||
Period end intangible asset balance | 13,453 | |||
The aggregate cash flows for the Group as a result of the acquisition in the Year ended December 31,2021 | USD thousands | |||
Cash and cash equivalents paid | (5,053 | ) | ||
Cash and cash equivalents of the subsidiary | 205 | |||
Cash consideration for milestone achievement during the period | (1,960 | ) | ||
Net reduction of cash flow as of acquisition date | (6,808 | ) | ||
As of December 31, 2021, the recoverable amount of the in-process IPR&D was based on its value in use and was determined by discounting the future cash flows to be generated from it by using the discounted cash flows method, on the annual year test. The recoverable amount of the IPR&D exceeds their carrying amount, thus no impairment loss was recognized. The discount rate used for calculating intangible assets recoverable amount is 23.0%, in addition to taking into consideration the risks associated in small stock premium companies.
F - 28
| A. | In November 2020, the Israeli Securities Authority, or ISA, initiated an administrative proceeding claiming negligent misstatement regarding certain immediate and periodic reports published by the Company’s predecessor (Ophectra) during the years 2017 and 2018, prior to the merger with MeaTech and prior to establishment of the settlement fund in connection with the Merger. In February 2021, the trustee of the settlement fund informed the Company that the ISA views the Company as a party to this proceeding, notwithstanding the settlement and establishment of the settlement fund. This proceeding is of an administrative nature and carries a potential penalty in the form of a monetary fine which, under applicable Israeli law, could be as high as NIS 5 million. In April 2021, following negotiations with the ISA, the Company agreed to settle the matter for $0.2 million (NIS 0.7 million), for which the Company recorded a provision. The settlement is subject to approval of the ISA’s Enforcement Committee. |
| B. | In February 2021, a civil claim was lodged against the settlement fund, relating to Ophectra's activities prior to establishment of the settlement fund, in an amount of USD $0.8 million (NIS 2.5 million). The Company believes that the probability is low of a final ruling against the settlement fund. |
F - 29
| A. | Balances with related parties |
Year ended | Year ended | Year ended | ||||||||||
December 31, | December 31, | December 31, | ||||||||||
2021 | 2020 | 2019 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Related companies receivables | - | - | 87 | |||||||||
Trade and other payables | 261 | 117 | 52 | |||||||||
| B. | Expense amounts with respect to related parties |
Year ended December 31, | Year ended December 31, | Year ended December 31, | ||||||||||
2021 | 2020 | 2019 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
General and administrative expenses | ||||||||||||
Salaries, wages and related expenses | 588 | 316 | 89 | |||||||||
Legal and professional services | 301 | 281 | 58 | |||||||||
Share-based payments | 777 | 488 | - | |||||||||
Research & Development expenses | ||||||||||||
Salaries, wages and related | 338 | 121 | - | |||||||||
Share-based payments | 66 | 64 | 15 | |||||||||
| 1. | Key Management Personnel The Company recognizes four key management personnel as related parties, namely Mr. Sharon Fima – former Chief Executive Officer (CEO), served as CEO until January 24 2022, Mr. Omri Schanin - Deputy CEO, Mr. Guy Hefer – Chief Financial Officer (CFO) and Mr. Dan Kozlovski – Chief Technologies Officer (CTO), who served as Vice President of Research and Development (VP R&D) until February 2022.
Mr. Sharon Fima, the previous CEO and CTO, who also served as a director, was employed by the Company (including MeaTech Ltd. prior to the merger described in Note 1A above) between September 1, 2019 and January 24, 2022. Until July 2021, Mr. Fima was entitled to a gross annual salary of NIS 0.5 million (USD 0.1 million) plus generally accepted social benefit contributions for senior executives and the use of a company car, including a related tax gross-up. Commencing August 1, 2021, Mr. Fima was entitled to an annual gross salary of NIS 0.6 million (USD 0.2 million). Mr. Fima also received options valued at NIS 0.2 million (USD 0.1 million) to be recognized over three-year vesting period commencing March 2020, some of which were forfeited subsequent to the balance sheet date following the CEO replacement. |
F - 30
Note 18 – Related and Interested Parties (cont.)
| B. | Expense amounts with respect to related parties (cont.) |
The Deputy CEO, who also served as a director until January 2022, has been employed by the Company (including MeaTech Ltd. prior to the merger described in Note 1A above) since September 1, 2019. Mr. Schanin was entitled to a gross annual salary of NIS 0.4 million (USD 0.1 million) plus generally accepted social benefit contributions for senior executives. Commencing August 1, 2021, Mr. Schanin is entitled to an annual gross salary of NIS 0.5 million (USD 0.2 million). Mr. Schanin also received options valued at an aggregate of NIS 0.8 million (USD 0.25 million) to be recognized over three-year vesting periods commencing August 2021.
The CFO, has been employed by the Company since October 18, 2020. Mr. Hefer was entitled to a gross annual salary of NIS 0.4 million (USD 0.1) plus generally accepted social benefit contributions for senior executives. Commencing August 1, 2021, Mr. Hefer is entitled to an annual gross salary of NIS 0.5 million (USD 0.2 million). Mr. Hefer also received options valued at an aggregate of NIS 0.75 million (USD 0.23 million) to be recognized over three-year vesting periods commencing in 2021.
The CTO (previously VP R&D), has been employed by the Company (including MeaTech Ltd. prior to the merger described in Note 1A above) since December 5, 2019. Mr. Kozlovski was entitled to a gross annual salary of NIS 0.4 million (USD 0.1 million) plus generally accepted social benefit contributions for senior executives. Commencing August 1, 2021, Mr. Kozlovski is entitled to an annual gross salary of NIS 0.5 million (USD 0.2 million). Mr. Kozlovski also received options valued at of NIS 0.07 million (USD 0.02 million)to be recognized over three-year vesting period commencing in 2019. |
| 2. | Directors |
Mr. Steve H. Lavin served as active chairman of the Company's Board of Directors between May 2020 and January 2022, and he was entitled to an annual compensation of USD 0.2 million as well as share-based compensation.
Mr. Danny Ayalon served as director between May 2020 and January 2022, and was entitled to an annual compensation of USD 0.03 million as well as share-based compensation.
Additional non-executive directors were compensated in accordance with the terms of the Israeli Companies Regulations (Rules Regarding Payment and Expenses for External Directors), 2000, as amended until July 31, 2021 and are since entitled to annual compensation of USD 0.03 million as well as share-based compensation.
F - 31
Leases in which the Group is the lessee | ||
| 1. | Under an office leasing agreement dated November 1, 2019, MeaTech leased office space and parking spaces, for a monthly fee of USD 10 thousand (NIS 32 thousand), including management fees, for a period of two years, with an option to extend the term of the lease by one more year. The Company initially recognized a long-term lease liability and a right-of-use asset in the amount of USD 214 thousand (NIS 743 thousand). The incremental interest rate used for estimating the liability is 2.25%. On November 2021 the Company extended the agreement for an additional period of 2.3 years. | |
| 2. | Under an office leasing agreement dated August 9, 2020, the Company leased office space and parking spaces, for a monthly fee of USD 8 thousand (NIS 27 thousand), including management fees, for a period of one year, with an option to extend the term of the lease by one more year. The Company initially recognized a long-term lease liability and a right-of-use asset in the amount of USD 102 thousand (NIS 348 thousand). The incremental interest rate used for estimating the liability is 4.3%. This agreement has ended during 2021. |
| 3. | Under an office leasing agreements dated between March and October 2021 for periods of 1.5-2 years, POM and Meatech Europe BV are leasing several spaces from a shared spaces provider for a monthly aggregated fee of USD 11 thousand (EUR 10 thousand). The Company initially recognized a long-term lease liability and a right-of-use asset in the amount of USD 259 thousand (EUR 220 thousand). The incremental interest rate used for estimating the liability is 3%. |
| 4. | Right-of-Use Asset |
USD thousands | ||||
Balance as at January 1, 2020 | 197 | |||
Additions during the year | 102 | |||
Amortization during the year | (146 | ) | ||
| Effect of changes in exchange rates | 15 | |||
Balance as at December 31, 2020 | 168 | |||
Additions following the acquisition of POM | 16 | |||
Additions during the year | 512 | |||
Amortization during the year | (286 | ) | ||
Effect of changes in exchange rates | (3 | ) | ||
Balance as at December 31, 2021 | 407 | |||
| 5. | Maturity analysis of for the Company’s lease liabilities |
December 31, | December 31, | |||||||
2021 | 2020 | |||||||
USD thousands | USD thousands | |||||||
Up to one year | 165 | 180 | ||||||
1-5 years | 246 | - | ||||||
Total | 411 | 180 | ||||||
F - 32
Note 19 – Leases (cont.)
| 6. | Amounts recognized in the statement of operation |
Year ended December 31, | Year ended December 31, | |||||||
2021 | 2020 | |||||||
USD thousands | USD thousands | |||||||
Amortization of ROU asset | 286 | 146 | ||||||
Interest expenses on lease liability | 9 | 5 | ||||||
F - 33
Year ended December 31, 2021 | Year ended December 31, 2020 | Year ended December 31, 2019 | ||||||||||
Issued and paid-in share capital as at January 1 | 79,866,264 | 19,870,337 | - | |||||||||
Weighted average of the number of ordinary shares of MeaTech 3D Ltd. issued during the year | 36,088,237 | 40,241,860 | - | |||||||||
Weighted average of the number of ordinary shares used to calculate basic earnings per share | 115,954,501 | 60,112,197 | 19,484,478 | |||||||||
In prior periods, the weighted average number of the ordinary shares of MeaTech (now known as MeaTech MT Ltd.) was multiplied by the exchange ratio according to which ordinary shares of MeaTech 3D Ltd. were issued in return for ordinary shares of MeaTech in the 2020 reverse acquisition.
At December 31, 2021, 41,902,044 options, warrants and RSUs (in 2020 and 2019, 45,768,424 and 9,839 options respectively) were excluded from the diluted weighted average number of ordinary shares calculation, as their effect would have been anti-dilutive.
F - 34
| A. | To secure its undertakings in connection with its lease agreements as described in Note 19, MeaTech provided a bank guarantee in the amount of USD 27 thousand (NIS 85 thousand) For which there's a restricted deposit. MeaTech also restricted a deposit of USD 26 thousand (NIS 80 thousand) in favor of a bank to secure its liabilities with respect to credit cards. The guarantee and deposit were assigned to MeaTech 3D Ltd. upon the Merger. |
| B. | To secure its undertakings in connection with its future lease agreement, MeaTech 3D Ltd. provided a bank guarantee in the amount of USD 334 thousand (NIS 1,040 thousand) For which there's a restricted deposit. |
| C. | To secure its undertakings in connection with its lease agreements as described in Note 13, POM provided a bank guarantee in the amount of USD 18 thousand (EUR 15 thousand) For which there's a restricted deposit. |
| A. | Framework for risk management |
| B. | Credit risk |
| C. | Liquidity risk |
| D. | Market risk |
F - 35
Note 23 – Financial Instruments (cont.)
| E. | Fair value |
In connection with the Company’s Nasdaq public offering, all existing price protection mechanisms were eliminated, as a result of which financing income was recorded.
Note 24 – Subsequent Events
| A. | Management Updates
In January 2022, Mr. Sharon Fima stepped down from the positions of Chief Executive Officer, Chief Technology Officer and Director, citing the Company’s current stage of development. Messrs. Steven H. Lavin (Chairman) and Danny Ayalon also stepped down from the Board of Directors, citing the Company’s current stage of development and to pursue other ventures, and Mr. Omri Schanin stepped down from the Board of Directors and continues to serve as MeaTech’s Deputy CEO.
The Company’s Board of Directors appointed Mr. Arik Kaufman to the position of Chief Executive Officer and Mr. Yaron Kaiser to the position of Chairman of the Board of Directors. | |
| B. | Move to New Premises In March 2022, the Company moved to its new headquarters at 5 David Fikes St., Rehovot, Israel, and terminated the lease at its previous headquarters. The laboratory and office space total approximately 18,300 square feet. The lease for this facility will expire in January 2026, although the Company has an option to renew it for four years. The annual rent (including parking fees) is approximately USD 0.7 million, linked to the Israeli CPI. This move is expected to affect the Company’s estimates regarding lease maturities and right-of-use assets in future reporting periods. |
F - 36

Steakholder Foods Ltd. (formerly known as MeaTech 3D Ltd.)
Unaudited Consolidated Interim Financial Statements As At June 30, 2022
June 30 | June 30 | December 31 | |||||||||||||
2022 | 2021 | 2021 | |||||||||||||
USD thousands | USD thousands | USD thousands | |||||||||||||
Current assets | |||||||||||||||
Cash and cash equivalents | 4 | 8,434 | 30,637 | 19,176 | |||||||||||
Other investment | 137 | 147 | 154 | ||||||||||||
Receivables | 5 | 2,237 | 1,174 | 2,782 | |||||||||||
Total current assets | 10,808 | 31,958 | 22,112 | ||||||||||||
Non-current assets | |||||||||||||||
Restricted deposits | 380 | 388 | 405 | ||||||||||||
Other investment | 1,209 | 1,264 | 1,355 | ||||||||||||
Right-of-use asset | 15 | 3,612 | 306 | 407 | |||||||||||
Intangible assets | 12,360 | 9,930 | 13,453 | ||||||||||||
Fixed assets, net | 4,653 | 2,206 | 2,922 | ||||||||||||
Total non-current assets | 22,214 | 14,094 | 18,542 | ||||||||||||
Total Assets | 33,022 | 46,052 | 40,654 | ||||||||||||
Current liabilities | |||||||||||||||
Trade payables | 779 | 359 | 382 | ||||||||||||
Other payables | 6 | 2,174 | 1,573 | 2,239 | |||||||||||
Current maturities of lease liabilities | 15 | 428 | 210 | 165 | |||||||||||
Total current liabilities | 3,381 | 2,142 | 2,786 | ||||||||||||
Non-current liabilities | |||||||||||||||
Long-term lease liabilities | 15 | 3,247 | 102 | 246 | |||||||||||
Total non-current liabilities | 3,247 | 102 | 246 | ||||||||||||
Capital | |||||||||||||||
Share capital and premium on shares | 70,476 | 65,283 | 69,610 | ||||||||||||
Capital reserves | 3,943 | 4,383 | 3,708 | ||||||||||||
Currency translation differences reserve | (1,898 | ) | 857 | 1,275 | |||||||||||
Accumulated deficit | (46,127 | ) | (26,715 | ) | (36,971 | ) | |||||||||
Total capital | 26,394 | 43,808 | 37,622 | ||||||||||||
Total liabilities and capital | 33,022 | 46,052 | 40,654 | ||||||||||||
F - 2
Six months ended June 30, | Six months ended June 30, | Year ended December 31, | |||||||||||||
2022 | 2021 | 2021 | |||||||||||||
USD thousands, except share data | USD thousands, except share data | USD thousands, except share data | |||||||||||||
Research and development expenses | 9 | 4,427 | 2,910 | 7,594 | |||||||||||
Marketing expenses | 10 | 1,959 | 605 | 1,628 | |||||||||||
General and administrative expenses | 11 | 3,687 | 4,159 | 8,010 | |||||||||||
Operating loss | 10,073 | 7,674 | 17,232 | ||||||||||||
Financing income | 12 | (1,062 | ) | (401 | ) | (509 | ) | ||||||||
Financing expenses | 145 | 493 | 1,299 | ||||||||||||
Total financing (income) expenses | (917 | ) | 92 | 790 | |||||||||||
Loss for the period | 9,156 | 7,766 | 18,022 | ||||||||||||
Currency translation differences loss (income) that might be transferred to profit or loss over ILS | 2,326 | (293 | ) | (1,942 | ) | ||||||||||
Currency translation differences loss (income) that might be transferred to profit or loss over EUR | 847 | 216 | 1,447 | ||||||||||||
Total comprehensive loss for the period | 12,329 | 7,689 | 17,527 | ||||||||||||
Loss per ordinary share, no par value (USD) | |||||||||||||||
Basic and diluted loss per share (USD) | 0.072 | 0.072 | 0.155 | ||||||||||||
Weighted-average number of shares outstanding - basic and diluted (shares) | 126,385,924 | 107,189,837 | 115,954,501 | ||||||||||||
F - 3
Unaudited Condensed consolidated interim of Income and of Comprehensive Loss
Three months ended June 30, | Three months ended June 30, | ||||||||||
2022 | 2021 | ||||||||||
USD thousands, except share data | USD thousands, except share data | ||||||||||
Research and development expenses | 9 | 2,285 | 1,783 | ||||||||
Marketing expenses | 10 | 908 | 285 | ||||||||
General and administrative expenses | 11 | 1,569 | 1,399 | ||||||||
Operating loss | 4,762 | 3,467 | |||||||||
Financing income | 12 | (678 | ) | (24 | ) | ||||||
Financing expenses | 85 | 664 | |||||||||
Total financing (income) expenses | (593 | ) | 640 | ||||||||
Loss for the period | 4,169 | 4,107 | |||||||||
Currency translation differences loss (income) that might be transferred to profit or loss over ILS | 1,811 | (898 | ) | ||||||||
Currency translation differences loss (income) that might be transferred to profit or loss over EUR | 600 | 33 | |||||||||
Total comprehensive loss for the period | 6,580 | 3,242 | |||||||||
Loss per ordinary share, no par value (USD) | |||||||||||
Basic and diluted loss per share (USD) | 0.033 | 0.033 | |||||||||
Weighted-average number of shares outstanding - basic and diluted (shares) | 126,533,200 | 123,665,142 | |||||||||
F - 4
Premium and Capital Share | Fair value of financial assets reserve | Transactions with related parties reserve | Currency translation differences reserve | Share- based payments reserve | Accumulated deficit | Total | ||||||||||||||||||||||
USD thousands | ||||||||||||||||||||||||||||
Balance as at January 1, 2022 | 69,610 | (334 | ) | 14 | 1,275 | 4,028 | (36,971 | ) | 37,622 | |||||||||||||||||||
Share-based payments | - | - | - | - | 1,048 | - | 1,048 | |||||||||||||||||||||
Exercise of options | 866 | - | - | - | (813 | ) | - | 53 | ||||||||||||||||||||
Other comprehensive income (loss) | - | - | - | (3,173 | ) | - | - | (3,173 | ) | |||||||||||||||||||
Loss for the period | - | - | - | - | - | (9,156 | ) | (9,156 | ) | |||||||||||||||||||
Balance as at June 30, 2022 | 70,476 | (334 | ) | 14 | (1,898 | ) | 4,263 | (46,127 | ) | 26,394 | ||||||||||||||||||
Balance as at January 1, 2021 | 30,481 | (334 | ) | 14 | 780 | 3,639 | (18,949 | ) | 15,631 | |||||||||||||||||||
Share-Based Payment | - | - | - | - | 2,313 | - | 2,313 | |||||||||||||||||||||
Issuance of shares and warrants, net | 30,357 | - | - | - | - | - | 30,357 | |||||||||||||||||||||
Exercise of options | 4,445 | - | - | - | (1,249 | ) | - | 3,196 | ||||||||||||||||||||
Other comprehensive income (loss) | - | - | - | 77 | - | - | 77 | |||||||||||||||||||||
Loss for the period | - | - | - | - | - | (7,766 | ) | (7,766 | ) | |||||||||||||||||||
Balance as at June 30, 2021 | 65,283 | (334 | ) | 14 | 857 | 4,703 | (26,715 | ) | 43,808 | |||||||||||||||||||
Balance as at January 1, 2021 | 30,481 | (334 | ) | 14 | 780 | 3,639 | (18,949 | ) | 15,631 | |||||||||||||||||||
Share-based payments | - | - | - | - | 3,965 | - | 3,965 | |||||||||||||||||||||
Issuance of shares and warrants, net | 32,330 | - | - | - | - | - | 32,330 | |||||||||||||||||||||
Exercise of options | 6,799 | - | - | - | (3,576 | ) | - | 3,223 | ||||||||||||||||||||
Other comprehensive income (loss) | - | - | - | 495 | - | - | 495 | |||||||||||||||||||||
Loss for the period | - | - | - | - | - | (18,022 | ) | (18,022 | ) | |||||||||||||||||||
Balance as at December 31, 2021 | 69,610 | (334 | ) | 14 | 1,275 | 4,028 | (36,971 | ) | 37,622 | |||||||||||||||||||
F - 5
Six months ended June 30, 2022 | Six months ended June 30, 2021* | Year ended December 31, 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Cash flows - operating activities | ||||||||||||
Net Loss for the period | (9,156 | ) | (7,766 | ) | (18,022 | ) | ||||||
Adjustments: | ||||||||||||
Depreciation and amortization | 722 | 278 | 680 | |||||||||
Change in fair value of derivative | - | (311 | ) | (316 | ) | |||||||
Change in fair value of other investment | (80 | ) | (90 | ) | (193 | ) | ||||||
Changes in net foreign exchange expenses | (982 | ) | 485 | 1,279 | ||||||||
Expenses of interest over liability of lease | 137 | 1 | 9 | |||||||||
Share-based payment expenses | 1,048 | 2,313 | 3,965 | |||||||||
Changes in asset and liability items: | ||||||||||||
Decrease (increase) in receivables | 257 | (782 | ) | (2,351 | ) | |||||||
Increase (decrease) in trade payables | 224 | 3 | (97 | ) | ||||||||
Increase (decrease) in other payables | 382 | 821 | 1,095 | |||||||||
Net cash from (used in) operating activities | (7,448 | ) | (5,048 | ) | (13,951 | ) | ||||||
Cash flows - investment activities | ||||||||||||
Acquisition of fixed assets | (2,341 | ) | (902 | ) | (1,828 | ) | ||||||
Increase in restricted deposit | (20 | ) | (337 | ) | (337 | ) | ||||||
Loan provided | - | (367 | ) | (367 | ) | |||||||
Proceeds on account of other investment* | 73 | 73 | 149 | |||||||||
Acquisition of other investments, net of cash acquired | (188 | ) | (4,848 | ) | (6,808 | ) | ||||||
| ||||||||||||
Net cash used in investing activities | (2,476 | ) | (6,381 | ) | (9,191 | ) | ||||||
Cash flows - financing activities | ||||||||||||
Proceeds from issuance of shares and warrants | - | 29,281 | 29,281 | |||||||||
Issuance costs | - | (3,283 | ) | (3,283 | ) | |||||||
Repayment of liability for lease | (230 | ) | (134 | ) | (346 | ) | ||||||
Repayment of interest over liability of lease | (137 | ) | (1 | ) | (9 | ) | ||||||
Proceeds from exercise of share options | 53 | 3,196 | 3,222 | |||||||||
Net cash from financing activities | (314 | ) | 29,059 | 28,865 | ||||||||
Increase in cash and cash equivalents | (10,238 | ) | 17,630 | 5,723 | ||||||||
Effect of exchange differences on cash and cash equivalents | (504 | ) | (549 | ) | (103 | ) | ||||||
Cash and cash equivalents at the beginning of the period: | 19,176 | 13,556 | 13,556 | |||||||||
Cash balance and cash equivalents at end of period | 8,434 | 30,637 | 19,176 | |||||||||
Non-cash activities | ||||||||||||
Purchase of fixed assets yet to be paid | 237 | 21 | 57 | |||||||||
Issue of shares and options against intangible asset | - | 4,359 | 6,332 | |||||||||
*Reclassified
F - 6
Steakholder Foods Ltd. (formerly Ophectra Real Estate and Investments Ltd., Meat-Tech 3D Ltd. and MeaTech 3D Ltd.) (the “Company”) was incorporated in Israel on July 22, 1992 as a private company limited by shares in accordance with the Companies Ordinance, 1983, and later a publicly-traded company whose ordinary shares were listed for trade on the Tel Aviv Stock Exchange (TASE). In March 2021, the Company completed an initial public offering on the Nasdaq Capital Market, listing American Depositary Shares (ADSs) for trade under the ticker STKH, and later voluntarily de-listed its ordinary shares from the TASE. The Company’s official address is 5 David Fikes St., Rehovot, Israel. In August 2022, the Company changed its name from MeaTech 3D Ltd. to Steakholder Foods Ltd.
The Company’s foodtech activities commenced in July 2019 by a company called MeaTech Ltd., which merged with the Company in January 2020 and became a fully-owned subsidiary, now called Steakholder Innovation Ltd. As the Company was the surviving entity of the merger, and continued the pre-merger business operations, utilizing the pre-merger management and employees, of MeaTech Ltd., the transaction was treated as a reverse acquisition that does not constitute a business combination.
The Company is developing a suite of advanced high-throughput manufacturing technologies to produce cell-based alternative protein products for cultivated, sustainable meat production, and focused on developing premium, center-of-plate meat products, including development of high-throughput bioprinting systems. |
Since inception the Company has incurred significant losses and negative cash flows from operations and has an accumulated deficit of USD 46.1 million. The Company has financed its operations mainly through fundraising from various investors.
The Company’s management expects that the Company will continue to generate losses and negative cash flows from operations for the foreseeable future. Based on the projected cash flows and cash balances as of June 30, 2022, management is of the opinion that its existing cash will be sufficient to fund operations until Q2 2023. As a result, there is substantial doubt about the Company’s ability to continue as a going concern.
|
Definitions: |
In these financial statements: |
| (1) | The Company - Steakholder Foods Ltd. |
| (2) | The Group – The Company and its subsidiaries, Steakholder Innovation Ltd. (formerly known as MeaTech Ltd.), Steakholder Foods Europe BV, Peace of Meat BV (hereafter “Peace Of Meat”) and Steakholder Foods USA, Inc. |
| (3) | Related Party - as defined in IAS 24 (revised). |
(4) | USD - United States Dollar | |
(5) | NIS – New Israeli Shekel | |
(6) | EUR – Euro | |
(7) | ADS – American Depositary Shares | |
| (8) | GBP - British pound sterling |
F - 7
| A. | Statement of compliance with IFRS |
| B. | Use of estimates and judgments |
| C. | Functional currency and presentation currency |
Currency | USD - ILS | USD - EUR | ||||||||||||||
2022 | 2021 | 2022 | 2021 | |||||||||||||
June 30 | 3.500 | 3.260 | 0.962 | 0.841 | ||||||||||||
Average for six months ended June 30 | 3.272 | 3.265 | 0.915 | 0.830 | ||||||||||||
Average for three months ended June 30 | 3.341 | 3.265 | 0.938 | 0.830 | ||||||||||||
December 31 | - | 3.110 | - | 0.883 | ||||||||||||
Average for twelve months ended December 31 | - | 3.230 | - | 0.845 | ||||||||||||
Note 3 – Material Events in the reporting period
In January 2022, Messrs. Steven Lavin, Daniel Ayalon, Sharon Fima and Omri Schanin stepped down as directors of the Company, and Mr. Fima also resigned as Chief Executive Officer and Chief Technology Officer. At that time, Mr. Yaron Kaiser was appointed to the Board as its Chairman, and Mr. Arik Kaufman was appointed as the Company’s Chief Executive Officer. In April 2022, Mr. Schanin resigned as Deputy Chief Executive Officer.
In March 2022, the Company’s headquarters were relocated to new, more spacious premises with state-of-the-art laboratories in Rehovot, Israel. The new space, allows the Company to enhance its cultured meat research and development, including three-dimensional bioprinting technology, and to continue growing its biology and engineering teams.
For more information, see Note 15. |
F - 8
June 30 | June 30 | December 31 | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Cash in USD | 4,260 | 22,756 | 15,596 | |||||||||
Cash in NIS | 3,059 | 6,225 | 1,688 | |||||||||
Cash in Euro | 1,110 | 1,656 | 1,892 | |||||||||
| Cash in GBP | 5 | - | - | |||||||||
Total cash and cash equivalents | 8,434 | 30,637 | 19,176 | |||||||||
| June 30 | June 30 | December 31 | ||||||||||
| 2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Institutions | 320 | 548 | 301 | |||||||||
Prepaid expenses | 854 | 605 | 743 | |||||||||
Other | 1,063 | 21 | 1,738 | |||||||||
| 2,237 | 1,174 | 2,782 | ||||||||||
| June 30 | June 30 | December 31 | |||||||||
| 2022 | 2021 | 2021 | |||||||||
| USD thousands | USD thousands | USD thousands | |||||||||
|
|
|
| |||||||||
Accrued expenses | 626 | 513 | 459 | |||||||||
Employee benefits | 1,081 | 532 | 1,122 | |||||||||
Provision - see note 13 (a) | 200 | 217 | 217 | |||||||||
Subsidiary government grant advances | 252 | 311 | 218 | |||||||||
Others | 15 | - | 223 | |||||||||
| 2,174 | 1,573 | 2,239 | |||||||||
F - 9
Thousands of Ordinary Shares | ||||
June 30, 2022 | ||||
Issued and paid-in share capital as at the beginning of the period | 125,770 | |||
Exercise of share options during the period – Share-Based Payment-related | 589 | |||
Issued not - for cash during the period* | 176 | |||
Issued and paid-in share capital as at June 30 | 126,535 | |||
Authorized share capital | 1,000,000 | |||
* Allocated to POM founders as part of original SPA
Date of grant and eligible recipients | Terms of the instrument | No. of ordinary shares (thousands) | Vesting Conditions | Contractual duration of the instrument (years) | ||||
Options awarded to CEO and Chairman on March 15, 2022 | Options exercisable for ordinary shares | 850 | 12 quarterly tranches | 4 years | ||||
Options awarded to employees of the Company and subsidiaries on March 24, 2022 | Options exercisable for ordinary shares | 775 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Options awarded to employees of the Company on June 10, 2022 | Options exercisable for ordinary shares | 2,180 | 1/3 after one year and the balance in 8 quarterly tranches | 4 years | ||||
Total options exercisable into shares | 3,805 |
F - 10
Options | |||
Fair value at date awarded | USD 1,107 thousand | ||
Parameters taken into account in the fair value calculation: | |||
Share price (USD at date awarded) | 0.43-0.51 | ||
Exercise price (USD unlinked) | 0.40-0.52 | ||
Expected volatility (weighted average) | 94.87%-100.74% | ||
Contractual term | 4 years | ||
Risk-free interest rate | 2.03%-3.24% | ||
Expected rate of dividend | 0% |
The expected volatility (standard deviation) was determined on the basis of share price volatility in similar companies, due to the Company’s limited historic share price performance since the date of the merger in January 2020 described in Note 1A above. The estimated Contractual Term of the options is the full contractual life of the option. The risk-free interest rate was based on US bonds, with time to maturity equivalent to the expected useful life of the options. Share price was used according to quoted share prices on Nasdaq.
F - 11
Six months ended June 30, | Six months ended June 30, | Year ended | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Salaries, wages and related expenses(1) | 2,356 | 1,354 | 3,425 | |||||||||
Share-based payment(1) | 218 | 463 | 911 | |||||||||
Materials | 689 | 514 | 1,875 | |||||||||
Professional services | 315 | 132 | 403 | |||||||||
Maintenance, office and software fees | 197 | 79 | 145 | |||||||||
Depreciation and amortization | 468 | 174 | 400 | |||||||||
| D&O insurance | 112 | 119 | 332 | |||||||||
Others | 72 | 75 | 103 | |||||||||
Total Research and Development Expenses | 4,427 | 2,910 | 7,594 | |||||||||
Three months ended June 30, | Three months ended June 30, | |||||||
2022 | 2021 | |||||||
USD thousands | USD thousands | |||||||
Salaries, wages and related expenses(1) | 1,206 | 830 | ||||||
Share-based payment(1) | 193 | 200 | ||||||
Materials | 295 | 342 | ||||||
Professional services | 190 | 86 | ||||||
Maintenance, office and software fees | 78 | 42 | ||||||
Depreciation and amortization | 250 | 149 | ||||||
| D&O insurance | 32 | 66 | ||||||
Others | 41 | 68 | ||||||
Total Research and Development Expenses | 2,285 | 1,783 | ||||||
F - 12
Six months ended June 30, | Six months ended June 30, | Year ended | ||||||||||
| 2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Salaries, wages and related expenses(1) | 445 | 243 | 494 | |||||||||
Share-based payment(1) | 528 | 76 | 570 | |||||||||
PR and advertisement | 817 | 269 | 507 | |||||||||
Maintenance, office and software fees | 41 | 7 | 22 | |||||||||
Depreciation and amortization | 54 | 5 | 17 | |||||||||
D&O insurance | 43 | 0 | - | |||||||||
Others | 31 | 5 | 18 | |||||||||
Total Marketing Expenses | 1,959 | 605 | 1,628 | |||||||||
(1) Including expenses in respect of related parties - see Note 14.
Three months ended June 30, | Three months ended June 30, | |||||||
| 2022 | 2021 | |||||||
USD thousands | USD thousands | |||||||
Salaries, wages and related expenses(1) | 235 | 120 | ||||||
Share-based payment(1) | 71 | 27 | ||||||
PR and advertisement | 523 | 127 | ||||||
Maintenance, office and software fees | 14 | 3 | ||||||
Depreciation and amortization | 23 | 7 | ||||||
D&O insurance | 18 | - | ||||||
Others | 24 | 1 | ||||||
Total Marketing Expenses | 908 | 285 | ||||||
(1) Including expenses in respect of related parties - see Note 14.
F - 13
Note 11 – General and Administrative Expenses
Six months ended June 30, | Six months ended June 30, | Year ended December 31, | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Salaries, wages and related expenses(1) | 969 | 556 | 1,328 | |||||||||
Share-based payment(1) | 302 | 1,774 | 2,484 | |||||||||
Legal and professional services(1) | 1,096 | 739 | 1,499 | |||||||||
D&O insurance | 665 | 680 | 1,837 | |||||||||
Corporate costs | 111 | 227 | 343 | |||||||||
Maintenance, office and software fees | 172 | 54 | 149 | |||||||||
Depreciation and amortization | 190 | 97 | 263 | |||||||||
Others | 182 | 32 | 107 | |||||||||
Total General and Administrative Expenses | 3,687 | 4,159 | 8,010 | |||||||||
(1) Including expenses in respect of related parties - see Note 14. | ||||||||
Three months ended June 30, | Three months ended June 30, | |||||||
2022 | 2021 | |||||||
USD thousands | USD thousands | |||||||
Salaries, wages and related expenses(1) | 494 | 227 | ||||||
Share-based payment(1) | 75 | 208 | ||||||
Legal and professional services(1) | 469 | 155 | ||||||
D&O insurance | 203 | 565 | ||||||
Corporate costs | 64 | 135 | ||||||
Maintenance, office and software fees | 79 | 20 | ||||||
Depreciation and amortization | 88 | 78 | ||||||
Others | 97 | 11 | ||||||
Total General and Administrative Expenses | 1,569 | 1,399 | ||||||
F - 14
Six months ended June 30, | Six months ended June 30, | Year ended December 31, | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Financial Income | ||||||||||||
Net change in fair value of financial instruments mandatorily measured at fair value through profit or loss | 80 | 401 | 509 | |||||||||
Net foreign exchange income | 982 | - | - | |||||||||
Total Financial Income | 1,062 | 401 | 509 | |||||||||
Financial Expenses | ||||||||||||
Net foreign exchange loss | - | 485 | 1,279 | |||||||||
Interest expense on lease liabilities | 137 | 1 | 9 | |||||||||
Bank interest and commission expenses | 8 | 7 | 11 | |||||||||
Total Financial Expenses | 145 | 493 | 1,299 | |||||||||
Net financing expenses (income) recognized in profit or loss | (917 | ) | 92 | 790 | ||||||||
Three months ended June 30, | Three months ended June 30, | |||||||
2022 | 2021 | |||||||
USD thousands | USD thousands | |||||||
Financial Income | ||||||||
Net change in fair value of financial instruments mandatorily measured at fair value through profit or loss(1) | 36 | 24 | ||||||
Net foreign exchange income | 642 | - | ||||||
Total Financial Income | 678 | 24 | ||||||
Financial Expenses | ||||||||
Net foreign exchange loss | - | 659 | ||||||
Interest expense on lease liabilities | 81 | - | ||||||
Bank interest and commission expenses | 4 | 5 | ||||||
Total Financial Expenses | 85 | 664 | ||||||
Net financing expenses (income) recognized in profit or loss | (593 | ) | 640 | |||||
F - 15
In November 2020, the Israeli Securities Authority, or ISA, initiated an administrative proceeding claiming negligent misstatement regarding certain immediate and periodic reports published by the Company’s predecessor (Ophectra) during the years 2017 and 2018, prior to the merger with MeaTech and prior to establishment of the settlement fund in connection with the Merger. In February 2021, the trustee of the settlement fund informed the Company that the ISA views the Company as a party to this proceeding, notwithstanding the settlement and establishment of the settlement fund. This proceeding is of an administrative nature and carries a potential penalty in the form of a monetary fine which, under applicable Israeli law, could be as high as NIS 5 million. In April 2021, following negotiations with the ISA, the Company agreed to settle the matter for $0.2 million (NIS 0.7 million), for which the Company recorded a provision. The settlement is subject to approval of the ISA’s Enforcement Committee. Similar proceedings were initiated with several other companies where the verdict, as of now, is that the companies are not to be held liable to the claims. There for, the Company initiated procedures to try and reclaim its innocence. As of now, due to lack of certainty, the Company keeps its provision stated above. |
In February 2021, a civil claim was lodged against the settlement fund, relating to Ophectra's activities prior to establishment of the settlement fund, in an amount of USD $0.7 million (NIS 2.5 million). The Company believes that it is more likely than not that no final ruling will be decided against the settlement fund. |
F - 16
Balances with related parties |
Six months ended | Six months ended | Year ended | ||||||||||
June 30, | June 30, | December 31, | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Trade and other payables | 165 | 106 | 261 | |||||||||
Expense amounts with respect to related parties |
Period ended June 30, | Period ended June 30, | Year ended December 31, | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
General and administrative expenses | ||||||||||||
Salaries, wages and related expenses | 307 | 242 | 588 | |||||||||
Legal and professional services | 222 | 135 | 301 | |||||||||
Share-based payments | 375 | 309 | 777 | |||||||||
| Sales and Marketing expenses | ||||||||||||
| Salaries, wages and related expenses | 52 | - | - | |||||||||
| Professional services | 17 | - | - | |||||||||
| Share-based payments | 185 | - | - | |||||||||
Research & Development expenses | ||||||||||||
Salaries, wages and related | 133 | 138 | 338 | |||||||||
Share-based payments | 10 | 41 | 66 | |||||||||
Key Management Personnel The Company recognizes three key management personnel as related parties, namely Mr. Arik Kaufman – Chief Executive Officer (CEO), Mr. Guy Hefer – Chief Financial Officer (CFO) and Mr. Dan Kozlovski – Chief Technologies Officer (CTO), who served as Vice President of Research and Development (VP R&D) until February 2022. In addition, Mr. Omri Schanin served as Deputy Chief Executive Officer until April 2022.
The CEO has been employed by the Company since January 24, 2022. He previously served as a director until May 2020 (including as interim chairman from March 2020) and a consultant of the Company until his appointment as CEO. Mr. Kaufman is entitled to a gross annual salary of NIS 0.6 million (USD 0.2 million), an annual bonus based primarily on milestone achievement, generally accepted social benefit contributions for senior executives and domestic travel expenses. Mr. Kaufman has also received options valued at an aggregate of NIS 0.46 million (USD 0.14 million) to be recognized over a three-year vesting period commencing in 2022.
|
F - 17
Note 14 – Related and Interested Parties (Cont.)
Mr. Kaufman is a founding partner of BlueOcean Sustainability Fund, LLC, a Delaware LLC doing business as BlueSoundWaves, led by Ashton Kutcher, Guy Oseary and Effie Epstein, which provides the Company with marketing and promotional, consulting, and partner and investor engagement services in the U.S., in return for which BlueSoundWaves received warrants to purchase ordinary shares, as well as restricted share units, the expenses for which are recognized as share-based payments.
The previous CEO and CTO, who also served as a director, Mr. Sharon Fima, was employed by the Company (including MeaTech Ltd. prior to the merger described in Note 1A above) between September 1, 2019 and January 24, 2022. Mr. Fima was entitled to an annual gross salary of NIS 0.6 million (USD 0.2 million). Mr. Fima also received options valued at NIS 0.2 million (USD 0.1 million) to be recognized over three-year vesting period commencing March 2020, some of which were forfeited upon his resignation.
The former Deputy CEO, Mr. Omri Schanin, who also served as a director until January 2022, has been employed by the Company (including MeaTech Ltd. prior to the merger described in Note 1A above) since September 1, 2019. Following his notice of resignation, he no longer serves as Deputy CEO, and his resignation took effect on May 31, 2022. Mr. Schanin was entitled to an annual gross salary of NIS 0.5 million (USD 0.2 million).
The CFO has been employed by the Company since October 18, 2020. Mr. Hefer is entitled to an annual gross salary of NIS 0.5 million (USD 0.2 million). Mr. Hefer has also received options valued at an aggregate of NIS 0.75 million (USD 0.23 million) to be recognized over three-year vesting periods commencing in 2021.
The CTO (previously VP R&D), has been employed by the Company (including MeaTech Ltd. prior to the merger described in Note 1A above) since December 5, 2019. Mr. Kozlovski is entitled to an annual gross salary of NIS 0.5 million (USD 0.2 million). Mr. Kozlovski has also received options valued at of NIS 0.07 million (USD 0.02 million) to be recognized over three-year vesting period commencing in 2019. |
Directors |
Mr. Steve H. Lavin served as active chairman of the Company's Board of Directors between May 2020 and January 2022, and he was entitled to an annual compensation of USD 0.2 million as well as share-based compensation.
Mr. Yaron Kaiser has served as chairman of the Company’s Board of Directors since January 2022. He previously served as legal counsel to the Company and as a consultant until his appointment as Chairman. Mr. Kaiser is entitled to annual compensation of USD 0.15 million, an annual bonus equal to half the bonus awarded to the CEO, and re-imbursement of travel expenses. Mr. Kaiser is a founding partner of BlueSoundWaves, as described above.
F - 18
Note 14 – Related and Interested Parties (Cont.)
Mr. Danny Ayalon served as director between May 2020 and January 2022, and was entitled to annual compensation of USD 0.03 million as well as share-based compensation.
Additional non-executive directors are since entitled to annual compensation of USD 0.03 million for their service on the Board of Directors and USD 0.005 million for service on each Board committee, as well as share-based compensation.
Leases in which the Group is the lessee |
| 1. | Under the office leasing agreement dated May 18, 2021, the Company leased office space and parking spaces, for a monthly fee of USD 51 thousand (NIS 178 thousand) linked to the CPI, including management fees and insurance, for a period of 4 years, with an option to extend the term of the lease by an additional term of 4 years. On February 2022, after receiving the facilities, the Company initially recognized a long-term lease liability and a right-of-use asset in the amount of USD 3,625 thousand (NIS 12,687 thousand). The incremental interest rate used for estimating the liability is 8.77%. |
As part of leasing these new facilities the company decided to terminate its previous leasing agreements in Israel. |
| 2. | Right-of-Use Asset |
USD thousands | ||||
Balance as at January 1, 2020 | 168 | |||
| Additions following the acquisition of POM | 16 | |||
Additions during the year | 512 | |||
Amortization during the year | (286 | ) | ||
| Effect of changes in exchange rates | (3 | ) | ||
Balance as at December 31, 2021 | 407 | |||
Additions during the year | 4,127 | |||
Terminations during the year | (239 | ) | ||
Amortization during the year | (281 | ) | ||
Effect of changes in exchange rates | (402 | ) | ||
Balance as at June 30, 2022 | 3,612 | |||
F - 19
Note 15 – Leases (cont.)
| 3. | Maturity analysis of for the Company’s lease liabilities |
June 30, | June 30, | December 31, | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Up to one year | 428 | 210 | 165 | |||||||||
1-8 years | 3,247 | 102 | 246 | |||||||||
Total | 3,675 | 312 | 411 | |||||||||
| 4. | Amounts recognized in the statement of operation |
Six months ended June 30, | Six months ended June 30, | Year ended December 31, | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Amortization of ROU asset | 281 | 131 | 286 | |||||||||
Interest expenses on lease liability | 137 | 1 | 9 | |||||||||
Note 16 – Subsequent Event
On July 5, 2022, the Company consummated a securities purchase agreement with a single U.S. institutional investor for the purchase and sale of 600,000 American Depositary Shares (“ADSs”), each representing ten (10) ordinary shares of no par value, at a price of $3.50 per ADS, pre-funded warrants to purchase 1,257,143 ADSs at a price of $3.4999 (that were already paid) with an exercise price of $0.0001 per ADS to be paid once exercised, and warrants to purchase 1,857,143 ADSs for five years with an exercise price of $3.50 per ADS. The securities were offered in the framework of a registered direct offering. The gross proceeds were approximately $6.5 million, and the net proceeds were approximately $5.8 million.

Financial Information of Steakholder Foods Ltd. | Page | |
Contents: | ||
2 | ||
3 | ||
5 | ||
6 | ||
September 30 | September 30 | December 31 | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Current assets | ||||||||||||
Cash and cash equivalents | 11,203 | 25,441 | 19,176 | |||||||||
Other investment | 135 | 149 | 154 | |||||||||
Receivables | 764 | 1,890 | 2,782 | |||||||||
Total current assets | 12,102 | 27,480 | 22,112 | |||||||||
Non-current assets | ||||||||||||
Restricted deposits | 374 | 391 | 405 | |||||||||
Other investment | 1,180 | 1,287 | 1,355 | |||||||||
Right-of-use asset | 3,478 | 328 | 407 | |||||||||
Intangible assets | 11,706 | 11,362 | 13,453 | |||||||||
Fixed assets, net | 4,386 | 2,467 | 2,922 | |||||||||
Total non-current assets | 21,124 | 15,835 | 18,542 | |||||||||
Total Assets | 33,226 | 43,315 | 40,654 | |||||||||
Current liabilities | ||||||||||||
Trade payables | 581 | 313 | 382 | |||||||||
Other payables | 2,154 | 1,837 | 2,239 | |||||||||
Current maturities of lease liabilities | 407 | 251 | 165 | |||||||||
Derivative instrument | 2,450 | - | - | |||||||||
Total current liabilities | 5,592 | 2,401 | 2,786 | |||||||||
Non-current liabilities | ||||||||||||
Long-term lease liabilities | 3,195 | 81 | 246 | |||||||||
Total non-current liabilities | 3,195 | 81 | 246 | |||||||||
Capital | ||||||||||||
Share capital and premium on shares | 72,231 | 68,348 | 69,610 | |||||||||
Capital reserves | 3,581 | 2,653 | 3,708 | |||||||||
Currency translation differences reserve | (2,771 | ) | 519 | 1,275 | ||||||||
Accumulated deficit | (48,602 | ) | (30,687 | ) | (36,971 | ) | ||||||
Total capital | 24,439 | 40,833 | 37,622 | |||||||||
Total liabilities and capital | 33,226 | 43,315 | 40,654 | |||||||||
Nine months ended September 30, | Nine months ended September 30, | Year ended December 31, | ||||||||||
2022 | 2021 | 2021 | ||||||||||
USD thousands, except share data | USD thousands, except share data | USD thousands, except share data | ||||||||||
Research and development expenses | 7,219 | 4,928 | 7,594 | |||||||||
Marketing expenses | 2,426 | 872 | 1,628 | |||||||||
General and administrative expenses | 4,982 | 5,961 | 8,010 | |||||||||
Operating loss | 14,627 | 11,761 | 17,232 | |||||||||
Financing income | (3,258 | ) | (457 | ) | (509 | ) | ||||||
Financing expenses | 262 | 434 | 1,299 | |||||||||
Total financing (income) expenses | (2,996 | ) | (23 | ) | 790 | |||||||
Loss for the period | 11,631 | 11,738 | 18,022 | |||||||||
Currency translation differences loss (income) that might be transferred to profit or loss over ILS | 2,301 | (456 | ) | (1,942 | ) | |||||||
Currency translation differences loss (income) that might be transferred to profit or loss over EUR | 1,745 | 717 | 1,447 | |||||||||
Total comprehensive loss for the period | 15,677 | 11,999 | 17,527 | |||||||||
Loss per ordinary share, no par value (USD) | ||||||||||||
Basic and diluted loss per share (USD) | 0.088 | 0.104 | 0.155 | |||||||||
Weighted-average number of shares outstanding - basic and diluted (shares) | 132,652,822 | 112,891,686 | 115,954,501 | |||||||||
Three months ended September 30, | Three months ended September 30, | |||||||
2022 | 2021 | |||||||
USD thousands, except share data | USD thousands, except share data | |||||||
Research and development expenses | 2,792 | 2,018 | ||||||
Marketing expenses | 467 | 267 | ||||||
General and administrative expenses | 1,295 | 1,802 | ||||||
Operating loss | 4,554 | 4,087 | ||||||
Financing income | (2,196 | ) | (130 | ) | ||||
Financing expenses | 117 | 15 | ||||||
Total financing (income) expenses | (2,079 | ) | (115 | ) | ||||
Loss for the period | 2,475 | 3,972 | ||||||
Currency translation differences loss (income) that might be transferred to profit or loss over ILS | (25 | ) | (163 | ) | ||||
Currency translation differences loss (income) that might be transferred to profit or loss over EUR | 898 | 501 | ||||||
Total comprehensive loss for the period | 3,348 | 4,310 | ||||||
Loss per ordinary share, no par value (USD) | ||||||||
Basic and diluted loss per share (USD) | 0.017 | 0.032 | ||||||
Weighted-average number of shares outstanding - basic and diluted (shares) | 145,116,113 | 124,048,833 | ||||||
Premium and Capital Share | Fair value of financial assets reserve | Transactions with related parties reserve | Currency translation differences reserve | Share-based payments reserve | Accumulated deficit | Total | ||||||||||||||||||||||
USD thousands | ||||||||||||||||||||||||||||
Balance as at January 1, 2022 | 69,610 | (334 | ) | 14 | 1,275 | 4,028 | (36,971 | ) | 37,622 | |||||||||||||||||||
Share-based payments | - | - | - | - | 1,092 | - | 1,092 | |||||||||||||||||||||
Issuance of shares and warrants, net | 1,348 | - | - | - | - | - | 1,348 | |||||||||||||||||||||
Exercise of options | 1,273 | (1,219 | ) | 54 | ||||||||||||||||||||||||
Other comprehensive income (loss) | - | - | - | (4,046 | ) | - | - | (4,046 | ) | |||||||||||||||||||
Loss for the period | - | - | - | - | - | (11,631 | ) | (11,631 | ) | |||||||||||||||||||
Balance as at September 30, 2022 | 72,231 | (334 | ) | 14 | (2,771 | ) | 3,901 | (48,602 | ) | 24,439 | ||||||||||||||||||
Balance as at January 1, 2021 | 30,481 | (334 | ) | 14 | 780 | 3,639 | (18,949 | ) | 15,631 | |||||||||||||||||||
0 | 0 | |||||||||||||||||||||||||||
Share-Based Payment | - | - | - | - | 2,829 | - | 2,829 | |||||||||||||||||||||
Issuance of shares and warrants, net | 31,176 | - | - | - | - | - | 31,176 | |||||||||||||||||||||
Exercise of options | 6,691 | (3,495 | ) | 3,196 | ||||||||||||||||||||||||
Other comprehensive income (loss) | - | - | - | (261 | ) | - | - | (261 | ) | |||||||||||||||||||
Loss for the period | - | - | - | - | - | (11,738 | ) | (11,738 | ) | |||||||||||||||||||
0 | ||||||||||||||||||||||||||||
Balance as at September 30, 2021 | 68,348 | (334 | ) | 14 | 519 | 2,973 | (30,687 | ) | 40,833 | |||||||||||||||||||
Balance as at January 1, 2021 | 30,481 | (334 | ) | 14 | 780 | 3,639 | (18,949 | ) | 15,631 | |||||||||||||||||||
Share-based payments | - | - | - | - | 3,965 | - | 3,965 | |||||||||||||||||||||
Issuance of shares and warrants, net | 32,330 | - | - | - | - | - | 32,330 | |||||||||||||||||||||
Exercise of options | 6,799 | (3,576 | ) | 3,223 | ||||||||||||||||||||||||
Other comprehensive income (loss) | - | - | - | 495 | - | - | 495 | |||||||||||||||||||||
Loss for the period | - | - | - | - | - | (18,022 | ) | (18,022 | ) | |||||||||||||||||||
Balance as at December 31, 2021 | 69,610 | (334 | ) | 14 | 1,275 | 4,028 | (36,971 | ) | 37,622 |
Nine months ended September 30, 2022 | Nine months ended September 30, 2021 | Year ended December 31, 2021 | ||||||||||
USD thousands | USD thousands | USD thousands | ||||||||||
Cash flows - operating activities | ||||||||||||
Net Loss for the period | (11,631 | ) | (11,738 | ) | (18,022 | ) | ||||||
Adjustments: | ||||||||||||
Depreciation and amortization | 1,099 | 479 | 680 | |||||||||
Change in fair value of derivative | (2,100 | ) | (312 | ) | (316 | ) | ||||||
Change in fair value of other investment | (99 | ) | (138 | ) | (193 | ) | ||||||
Net foreign exchange expenses | (901 | ) | 411 | 1,279 | ||||||||
Interest expense over lease liabilities | 250 | 7 | 9 | |||||||||
Interest income over short term deposits | (73 | ) | - | - | ||||||||
Share-based payment expenses | 1,092 | 2,829 | 3,965 | |||||||||
Changes in asset and liability items: | ||||||||||||
Decrease (increase) in receivables | 1,790 | (1,523 | ) | (2,351 | ) | |||||||
Increase (decrease) in trade payables | 315 | (309 | ) | (97 | ) | |||||||
Increase (decrease) in other payables | 413 | 682 | 1,095 | |||||||||
Net cash from (used in) operating activities | (9,845 | ) | (9,612 | ) | (13,951 | ) | ||||||
Cash flows - investment activities | ||||||||||||
Acquisition of fixed assets | (2,686 | ) | (1,124 | ) | (1,828 | ) | ||||||
Increase in restricted deposit | (20 | ) | (337 | ) | (337 | ) | ||||||
Loan provided | - | (367 | ) | (367 | ) | |||||||
Proceeds on account of other investment* | 110 | 111 | 149 | |||||||||
Interest received over short term deposits | 73 | - | - | |||||||||
Acquisition of other investments, net of cash acquired | (188 | ) | (5,685 | ) | (6,808 | ) | ||||||
Net cash used in investing activities | (2,711 | ) | (7,402 | ) | (9,191 | ) | ||||||
Cash flows - financing activities | ||||||||||||
Proceeds from issuance of shares and warrants | 2,005 | 29,281 | 29,281 | |||||||||
Issuance costs | (657 | ) | (3,283 | ) | (3,283 | ) | ||||||
Repayment of liability for lease | (316 | ) | (222 | ) | (346 | ) | ||||||
Repayment of interest over liability of lease | (250 | ) | (7 | ) | (9 | ) | ||||||
Proceeds with regard to derivative | 4,495 | - | - | |||||||||
Proceeds from exercise of share options | 53 | 3,196 | 3,222 | |||||||||
Net cash from financing activities | 5,330 | 28,965 | 28,865 | |||||||||
Increase (Decrease) in cash and cash equivalents | (7,226 | ) | 11,951 | 5,723 | ||||||||
Effect of exchange differences on cash and cash equivalents | (747 | ) | (66 | ) | (103 | ) | ||||||
Cash and cash equivalents at the beginning of the period: | 19,176 | 13,556 | 13,556 | |||||||||
Cash balance and cash equivalents at end of period | 11,203 | 25,441 | 19,176 | |||||||||
Non-cash activities | ||||||||||||
Purchase of fixed assets yet to be paid | 4 | 188 | 57 | |||||||||
Issue of shares and options against intangible asset | - | 4,359 | 6,332 | |||||||||

Sole Book-Running Manager |
| A.G.P. |
Lead Manager |
| Maxim Group LLC |
| • | a financial liability imposed on him or her in favor of another person pursuant to a judgment, including a settlement or arbitrator’s award approved by a court. However, if an undertaking to indemnify an office holder with respect to such liability is provided in advance, then such an undertaking must be limited to events which, in the opinion of the board of directors, can be foreseen based on the company’s activities when the undertaking to indemnify is given, and to an amount or according to criteria determined by the board of directors as reasonable under the circumstances, and such undertaking shall detail the above-mentioned foreseen events and amount or criteria; |
| • | reasonable litigation expenses, including attorneys’ fees, incurred by the office holder: (i) as a result of an investigation or proceeding instituted against him or her by an authority authorized to conduct such investigation or proceeding, provided that (A) no indictment was filed against such office holder as a result of such investigation or proceeding; and (B) no financial liability, such as a criminal penalty, was imposed upon him or her as a substitute for the criminal proceeding as a result of such investigation or proceeding, or, if such financial liability was imposed, it was imposed with respect to an offense that does not require proof of criminal intent; and (ii) in connection with a monetary sanction; and |
| • | reasonable litigation expenses, including attorneys’ fees, incurred by the office holder or imposed by a court in proceedings instituted against him or her by the company, on its behalf, or by a third party, or in connection with criminal proceedings in which the office holder was acquitted, or as a result of a conviction for an offense that does not require proof of criminal intent. |
| • | a breach of the duty of loyalty to the company, provided that the office holder acted in good faith and had a reasonable basis to believe that the act would not harm the company; |
| • | a breach of duty of care to the company or to a third party, to the extent such a breach arises out of the negligent conduct of the office holder; and |
| • | a financial liability imposed on the office holder in favor of a third party. |
| • | any breach of duty of care to us or to a third party; |
| • | any other action which is permitted by law to insure an office holder against; |
| • | any expenses incurred and/or paid by the office holder in connection with an administrative enforcement procedure under any applicable law, including the Efficiency of Enforcement Procedures in the Securities Authority Law (legislation amendments), 5771-2011, and the Israeli Securities Law, which we refer to as an Administrative Enforcement Procedure, and including reasonable litigation expenses and attorney fees; and |
| • | any financial liability in favor of a victim of a felony pursuant to Section 52ND of the Israeli Securities Law. |
| • | a breach of the duty of loyalty, except for indemnification and insurance for a breach of the duty of loyalty to the company to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not harm the company; |
| • | a breach of duty of care committed intentionally or recklessly, excluding a breach arising solely out of the negligent conduct of the office holder; |
| • | an act or omission committed with intent to derive illegal personal benefit; or |
| • | a civil or administrative fine or forfeit levied against the office holder. |
• | In September and October 2019, we issued 19,681 Ordinary Shares to certain investors at a price of $95.94 per share and 9,839 warrants exercisable into Ordinary Shares at an exercise price of $353 per warrant, for aggregate gross proceeds of $1.89 million. |
• | In May 2020, we issued to certain investors 1,391,794 Ordinary Shares and warrants to purchase 8,040,382 Ordinary Shares at an exercise price of NIS 3.36 per warrant, for aggregate gross proceeds of $1 million. |
• | In May 2020, we issued to certain investors 4,398,570 Ordinary Shares and 4,398,570 options to purchase Ordinary Shares at an exercise price of NIS 3.03, for aggregate gross proceeds of $2.4 million. |
• | In August 2020, we issued to certain investors 5,292,160 Ordinary Shares and warrants exercisable into 7,409,021 Ordinary Shares at an exercise price of NIS 3.95 per share, for aggregate gross proceeds of $5.8 million, as well as rights exercisable into 1,374,998 Ordinary Shares at an exercise price of NIS 3.00 per share, and rights for warrants exercisable into 1,925,000 Ordinary Shares at an exercise price of NIS 3.95 per share. |
• | In December 2020, we issued to certain investors 6,791,600 Ordinary Shares, warrants exercisable into 3,395,800 Ordinary Shares at an exercise price of NIS 5.00 per share and warrants exercisable into 3,395,800 Ordinary Shares at an exercise price of NIS 6.00 per share, for aggregate gross proceeds of $7.3 million. |
• | In February 2021, we issued 4,070,766 Ordinary Shares and rights to purchase 4,070,766 additional Ordinary Shares, vesting with no exercise price upon the achievement of certain milestones, to certain shareholders of Peace of Meat, in the course of purchasing the entire outstanding equity of Peace of Meat in return for a combination of cash and our securities. |
• | In October 2021, we issued options to purchase 6,251,770 Ordinary Shares exercisable at a price at or above the closing price at the time of allocation (the precise price to be determined by the amount of increase in the ADS price by the time of exercise), and restricted share units vesting into 1,243,150 Ordinary Shares with no exercise price, all vesting between one and three years from the date of allocation and subject to acceleration upon milestone achievement, to our partner BlueSoundWaves in return for services related to accelerating our growth in developing and commercializing our proprietary cultured meat production technologies. |
(a) | The undersigned Registrant hereby undertakes: | ||||
(1) | To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: | ||||
i. | To include any prospectus required by section 10(a)(3) of the Securities Act of 1933; | ||||
ii. | To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; | ||||
iii. | To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement. | ||||
(2) | That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. | ||||
(3) | To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering. | ||||
(4) | To file a post-effective amendment to the registration statement to include any financial statements required by Item 8.A. of Form 20-F at the start of any delayed offering or throughout a continuous offering. Financial statements and information otherwise required by Section 10(a)(3) of the Act need not be furnished, provided that the registrant includes in the prospectus, by means of a post-effective amendment, financial statements required pursuant to this paragraph (a)(4) and other information necessary to ensure that all other information in the prospectus is at least as current as the date of those financial statements. Notwithstanding the foregoing, with respect to registration statements on Form F-3, a post-effective amendment need not be filed to include financial statements and information required by Section 10(a)(3) of the Act or Rule 3-19 of this chapter if such financial statements and information are contained in periodic reports filed with or furnished to the Commission by the registrant pursuant to section 13 or section 15(d) of the Securities Exchange Act of 1934 that are incorporated by reference in the Form F-3. | ||||
(5) | That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser: | ||||
i. | If the registrant is relying on Rule 430B: | ||||
A. | Each prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the date the filed prospectus was deemed part of and included in the registration statement; and | ||||
B. | Each prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) as part of a registration statement in reliance on Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii), or (x) for the purpose of providing the information required by section 10(a) of the Securities Act of 1933 shall be deemed to be part of and included in the registration statement as of the earlier of the date such form of prospectus is first used after effectiveness of the date of the first contract or sale of securities in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities in the registration statement to which that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract sale prior to such effective date, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such effective date; or | |||
ii. | If the registrant is subject to Rule 430C, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use. | |||
(6) | That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell securities to such purchaser: | |||
i. | Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424; | |||
ii. | Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant; | |||
iii. | The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and | |||
iv. | Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser. | |||
(1) | That for purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4), or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective. | |
(2) | That for the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
Steakholder Foods Ltd. | ||
By: | /s/ Arik Kaufman | |
Name: | Arik Kaufman | |
Title: | Chief Executive Officer | |
Signatures | Title | Date | |||
/s/ * | Chairman of the Board of Directors | January 5, 2023 | |||
Yaron Kaiser | |||||
/s/ Arik Kaufman | Chief Executive Officer and Director (Principal Executive Officer) | January 5, 2023 | |||
Arik Kaufman | |||||
/s/ Guy Hefer | Chief Financial Officer (Principal Financial and Accounting Officer) | January 5, 2023 | |||
Guy Hefer | |||||
/s/ * | Director | January 5, 2023 | |||
Eli Arad | |||||
/s/ * | Director | January 5, 2023 | |||
David Gerbi | |||||
/s/ * | Director | January 5, 2023 | |||
Sari Singer | |||||
/s/ Arik Kaufman | |||||
Arik Kaufman | |||||
Attorney in Fact | |||||
Steakholder Foods USA, Inc. | ||
Authorized U.S. Representative | ||
By: | /s/ Arik Kaufman | |
Name: | Arik Kaufman | |
Title: | President | |