
Exhibit 99.2 Presentation # 197 Oral long-acting menin inhibitor normalizes type 2 diabetes in two rat models *Thomas Butler, Sanchita Mourya, Weiqun Li, Tenley Archer, Taisei Kinoshita, Mini Balakrishnan, Priyanka Somanath EASD Annual Meeting 2022 Oral Presentation Golgi Hall Session: OP 33 Therapy outside the box 1 September 22, 2022

Our understanding of diabetes has evolved; Progression of type 1 and type 2 diabetes are both driven by beta cell loss Progression of Type 2 Diabetes “Understanding of Diabetes has evolved” Pipeline-in-a-Pill – Single Agent for Multiple Indications MLL Fusion & NPM1 Driven Tumors Initial clinical validation in r/r acute leukemias with MLL fusions in addition to NPM1 mutations MYC Addicted and Driven Tumors Expansion into r/r diffuse large b cell lymphoma and r/r/ multiple myeloma *Both Type 1 Diabetes and Type 2 Diabetes disease *Insulin Resistance leads to an increase in Beta Cell Workload, RAS/RAF Driven Solid Tumors results in Beta Cell loss and reduction in Beta Cell which ultimately leads to Beta Cell Failure and Death and the Mass progression of Type 2 Diabetes. Further expansion into KRAS and RAS mutant colorectal, lung, and pancreatic cancerVMF Disease Modifying Potential: BMF-219 drives Beta Cell Proliferation 2 *Int. J. Mol. Sci. 2016, 17, 744; doi:10.3390/ijms17050744

Menin: A key checkpoint for beta cell homeostasis; an important target for type 1 and type 2 diabetes Menin Activation • Menin functions in a histone methyltransferase protein complex containing MLL • This complex promotes Suppress Beta Cell trimethylation of histone H3 on Homeostasis lysine 4 (H3K4), which is (Prevent Beta Cell Recovery) associated with transcriptionally active chromatin and.. Inhibition of Menin • Menin dependent histone methylation maintains expression of p27 and p18, two key members of cyclin-dependent kinase (CDK) inhibitor family that prevent β-cell Support Beta Cell proliferation. Homeostasis (Permit Beta Cell Recovery) Sources: Karnik et al., Science, Nov 2007, Vol 318 P806-809 3

BMF-219, A potent & selective covalent menin inhibitor BMF-219 exerts transient changes in Menin Protein menin Menin Half Life Varies By Compartment S C Half Life in Cytoplasm: <1hr Half Life in Nucleus: 6-8 hrs Menin’s half-life in nucleus is MEN1 Gene Expression Decreases most relevant for pharmacological intervention w/ BMF-219 Treatment • BMF-219 produces robust decrease in expression of ~90% target protein (Menin) @6 hours Reduction • Effect continues beyond established nuclear half-life of menin, indicating robust effect that is not impacted by protein turnover 4

Zucker Diabetic Fatty Rat –A model of insulin resistance THE ZDF RAT Study Design Day • The ZDF rat is a model of pancreatic exhaustion, thus Treatment groups (n=10/group): mimicking some aspects of human diabetes. 1. Vehicle • Pioglitazone and metformin provide therapeutic efficacy in 2. BMF-219 175 mg/kg 3. Pioglitazone 30 mg/kg this model. • The ZDF rat is a translatable model for studying the development of T2D. 5

BMF-219 significantly reduces blood glucose by Oral Glucose Tolerance Test (OGTT) in ZDF rats OGTT, Day 15 600 * p<0.05 vs Vehicle A ** p<0.01 vs Vehicle OGTT, Area Under the Curve (AUC) *** p<0.001 vs Vehicle 80000 *** p<0.001 vs Vehicle 54% reduction 400 60000 ** ** 40000 *** ** *** ** ** 200 * 20000 ** *** ** ** * ** 0 Vehicle Pioglitazone BMF-219 0 (30 mg/kg) (175mg/kg) 0 15 30 60 90 120 Minutes Vehicle Pioglitazone (30 mg/kg) BMF-219 (175mg/kg) BMF-219 significantly reduces blood glucose levels by OGTT at Day 15 of treatment 6 Blood Glucose (mg/dL) Mean± SEM

BMF-219 significantly reduces blood glucose, insulin, and c-peptide levels in ZDF Rats (After 2 Weeks of Dosing) Blood Glucose (4hr fasting) Insulin (4hr fasting) * p<0.05 ** p<0.01 10 400 *** p<0.0001 8 300 6 ** * 200 * 4 *** ** 100 2 0 0 C-peptide (4hr fasting) Non-fasting glucose 600 6000 400 4000 * ** ** ** *** ** 200 ** 2000 0 0 Day -3 Day 1 Day 8 Day 14 BMF-219 significantly reduces blood glucose levels and alters serum insulin and C-peptide levels in ZDF rats @ Day 17 7 Mean±SE (mg/dL) Blood glucose (mg/dL) Mean ± SEM C-peptide (pM),Mean ± SEM Serum insulin (ng/mL)

BMF-219 increases insulin sensitivity in ZDF rats A HOMA-IR (4hr fasting) B HOMA-IR (non-fasting) 150 400 *** p<0.001 vs Vehicle *** p<0.001 vs Vehicle 300 100 200 *** *** *** *** 50 *** 100 *** 0 0 Day 1 Day 8 Day 14 Measurement of HOMA-IR in rats treated with BMF-219 for 16 Days 8 (ng/mL)

BMF-219 significantly reduces blood lipemic levels and reduces body weight at day 17 Cholesterol Body Weight (g) * p<0.05 ** p<0.01 250 600 * p<0.05 vs Vehicle ** p<0.01 vs Vehicle *** p<0.0001 200 550 * 150 ** 500 100 450 50 400 0 350 Triglycerides 300 1500 -7 -5 -3 1 4 6 8 11 13 15 18 20 22 25 27 29 ** p<0.01 vs Vehicle Days 1000 Vehicle Pioglitazone (30 mg/kg) BMF-219 (175mg/kg) 500 ** ** 0 15% reduction after 2 weeks dosing Measurement of cholesterol, triglycerides and body weight in BMF-219 treated rats for 16 days. (Animals continued to eat a high caloric diet until Day 31). 9 Serum Triglycerides (mg/dL) Serum Cholesterol (mg/dL) Mean ± SEM Mean ± SEM Body weight (g)
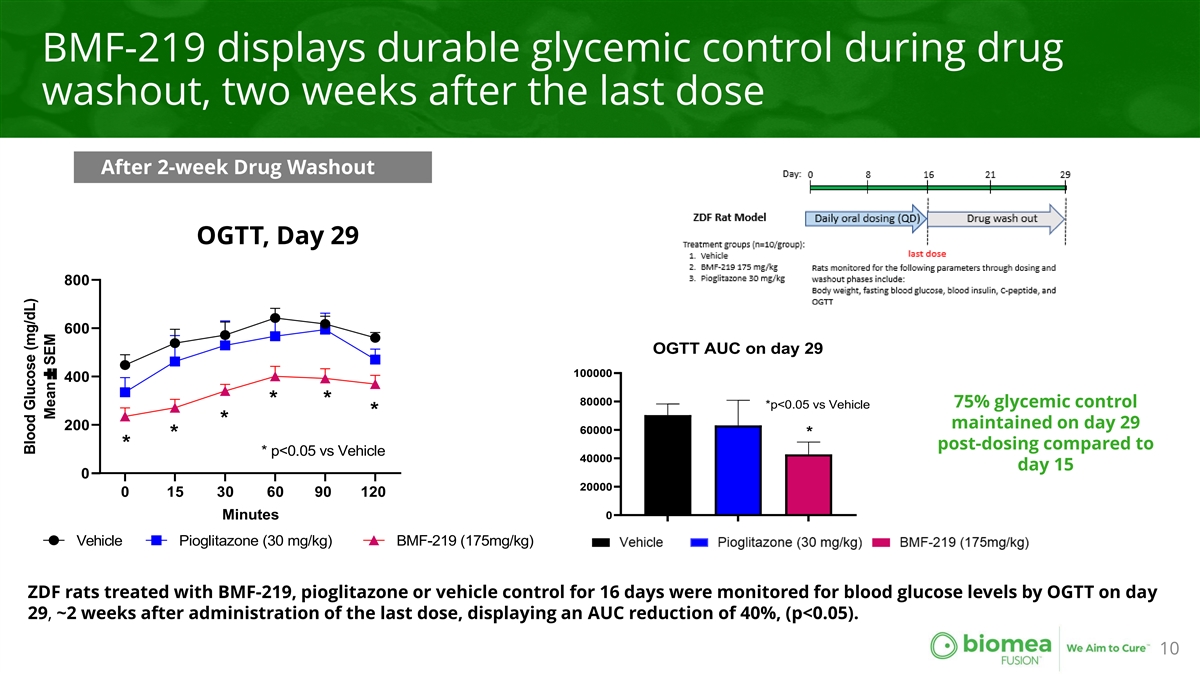
BMF-219 displays durable glycemic control during drug washout, two weeks after the last dose After 2-week Drug Washout OGTT, Day 29 800 600 OGTT AUC on day 29 100000 400 * * 80000 75% glycemic control *p<0.05 vs Vehicle * * maintained on day 29 200 60000 * * post-dosing compared to * * p<0.05 vs Vehicle 40000 day 15 0 20000 0 15 30 60 90 120 0 Minutes Vehicle Pioglitazone (30 mg/kg) BMF-219 (175mg/kg) ZDF rats treated with BMF-219, pioglitazone or vehicle control for 16 days were monitored for blood glucose levels by OGTT on day 29, ~2 weeks after administration of the last dose, displaying an AUC reduction of 40%, (p<0.05). 10 Blood Glucose (mg/dL) Mean± SEM

BMF-219 maintains a strong impact on blood glucose, insulin and c- peptide levels during drug washout, two weeks after the last dose Day 31: After 2-week Drug Washout Insulin (4hr fasting) C-peptide (4hr fasting) Blood Glucose (4hr fasting) * p<0.05 ** p<0.01 600 *** p<0.0001 ** 15 400 6000 ** ** * 10 4000 * ** 200 * 5 ** 2000 * 0 0 0 on Day 17 on Day 31 on Day 17 on Day 31 on Day 17 on Day 31 Two weeks after Two weeks after Two weeks after last dose last dose last dose BMF-219 maintains significant impact on blood glucose, insulin and c-peptide levels during drug washout (two weeks after last dose) (Animals continued to eat a high caloric diet until Day 31) 11 Blood glucose (mg/dL) Mean ± SEM Insulin (ng/mL) Mean±SEM C-peptide (pM) Mean ± SEM

BMF-219 increases β-islets in pancreas sections of ZDF diabetic model A. Vehicle; Day 31 B. Pioglitazone; Day 17 D. BMF-219; Day 31 C. BMF-219; Day 17 A) Vehicle-treated animal, Day 31. Beta islets display low congregation and growth, while alpha cells dominate. B) Pioglitzaone-treated animal, Day 17. Beta islets display congregation and growth. C) BMF-219 treated animal, Day 17. In contrast to the pioglitazone-treated animal shown in Panel B, note that BMF-219 treatment results in high congregation and growth of the beta islets. D) BMF-219 treated animal, Day 31. Beta islets display high congregation and continue to increase and mature. Red is insulin-beta islets, brown is glucagon-alpha cells. 12
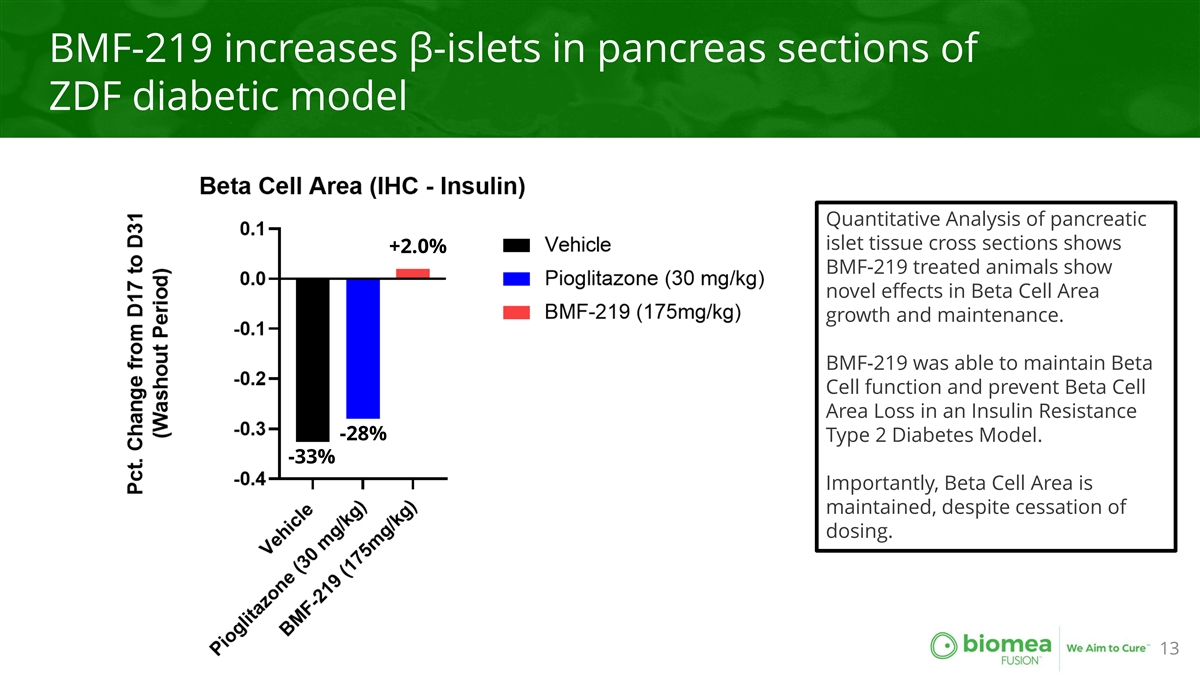
BMF-219 increases β-islets in pancreas sections of ZDF diabetic model Quantitative Analysis of pancreatic islet tissue cross sections shows +2.0% BMF-219 treated animals show novel effects in Beta Cell Area growth and maintenance. BMF-219 was able to maintain Beta Cell function and prevent Beta Cell Area Loss in an Insulin Resistance -28% Type 2 Diabetes Model. -33% Importantly, Beta Cell Area is maintained, despite cessation of dosing. 13
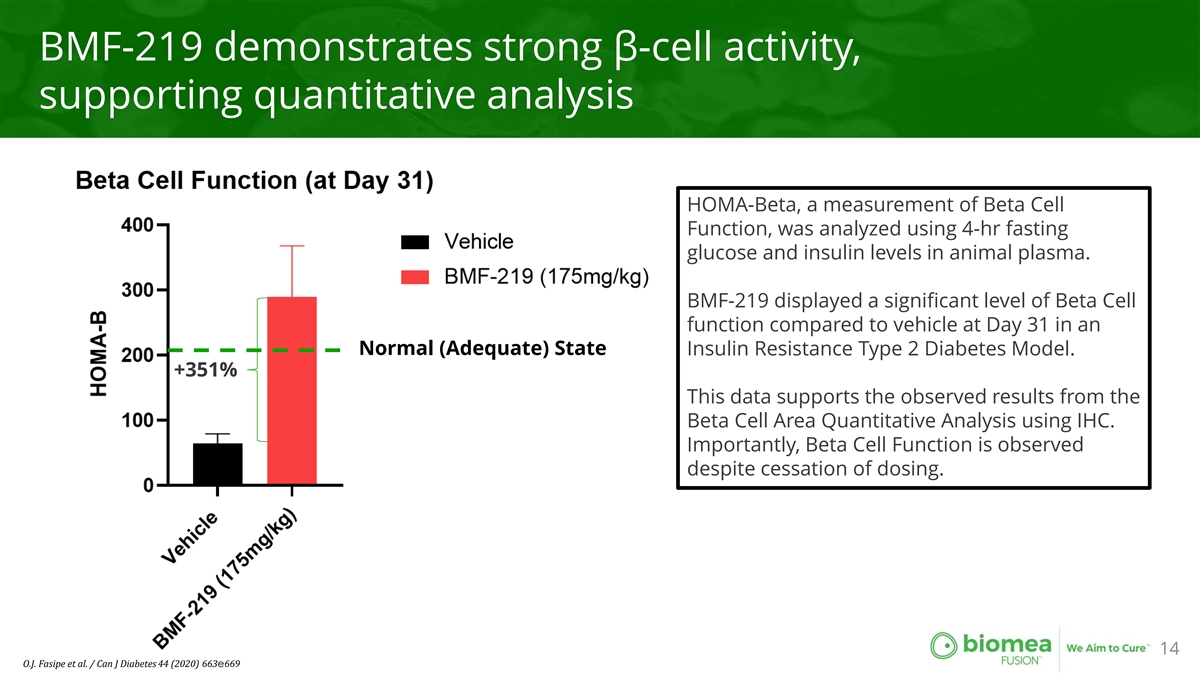
BMF-219 demonstrates strong β-cell activity, supporting quantitative analysis HOMA-Beta, a measurement of Beta Cell Function, was analyzed using 4-hr fasting glucose and insulin levels in animal plasma. BMF-219 displayed a significant level of Beta Cell function compared to vehicle at Day 31 in an Normal (Adequate) State Insulin Resistance Type 2 Diabetes Model. +351% This data supports the observed results from the Beta Cell Area Quantitative Analysis using IHC. Importantly, Beta Cell Function is observed despite cessation of dosing. 14 O.J. Fasipe et al. / Can J Diabetes 44 (2020) 663e669

Streptozotocin (STZ) Rat, A type 2 diabetes model of beta cell loss MEN1 Excision Prevents Development of STZ-induced Hyperglycemia ▪ Menin is a scaffold protein, encoded by the geneMEN1, that has been recently recognized for its role in Type 2 Diabetes Mellitus (T2DM) as a key regulator of b-cell proliferation. ▪ Men1 knockout mice demonstrate increased B- cell mass generation (Yang et al., 2010) and menin inhibition has previously been shown to improve glycemic control in high fat induced diabetic mice (Ma et al., 2021). ▪ Men1-excised mice do not develop hyperglycemia in a Streptozotocin-(STZ) Men1-excised mice did not develop hyperglycemia in STZ model, which was observed in the control group induced rat model, which is a model for impaired beta-cell function and insulin production, demonstrating the role of menin in glycemic control. 15

STZ Rat Model- Study Design Study Design The Streptozotocin (STZ)-Induced Rat Model Only direct insulin injection shows an effect in this model with high fat diet STZ treatment typically results in ~50% Beta Cell Loss 16

BMF-219 significantly reduced blood glucose by Oral Glucose Tolerance Test (OGTT) in an STZ-induced rat model Oral Glucose Tolerance Test (Day 17) Non-Fasting Glucose STZ + HFD model Vehicle Non-fasting Glucose Pioglitazone (30 mg/kg) 800 800 BMF-219 (175mg/kg) 600 600 400 400 * * 200 200 * * * * p<0.05 vs Vehicle 0 0 -7 -6 -5 -4 -3 -2 -1 1 8 14 0 15 30 60 90 120 Days Minutes TA dosing Vehicle Pioglitazone (30 mg/kg) BMF-219 (175mg/kg) start Only BMF-219 significantly reduces blood glucose by Only BMF-219 lowered non-fasting glucose in STZ rats OGTT in STZ rats 17 Blood Glucose (mg/dL) Mean± SEM Mean±SE (mg/dL)
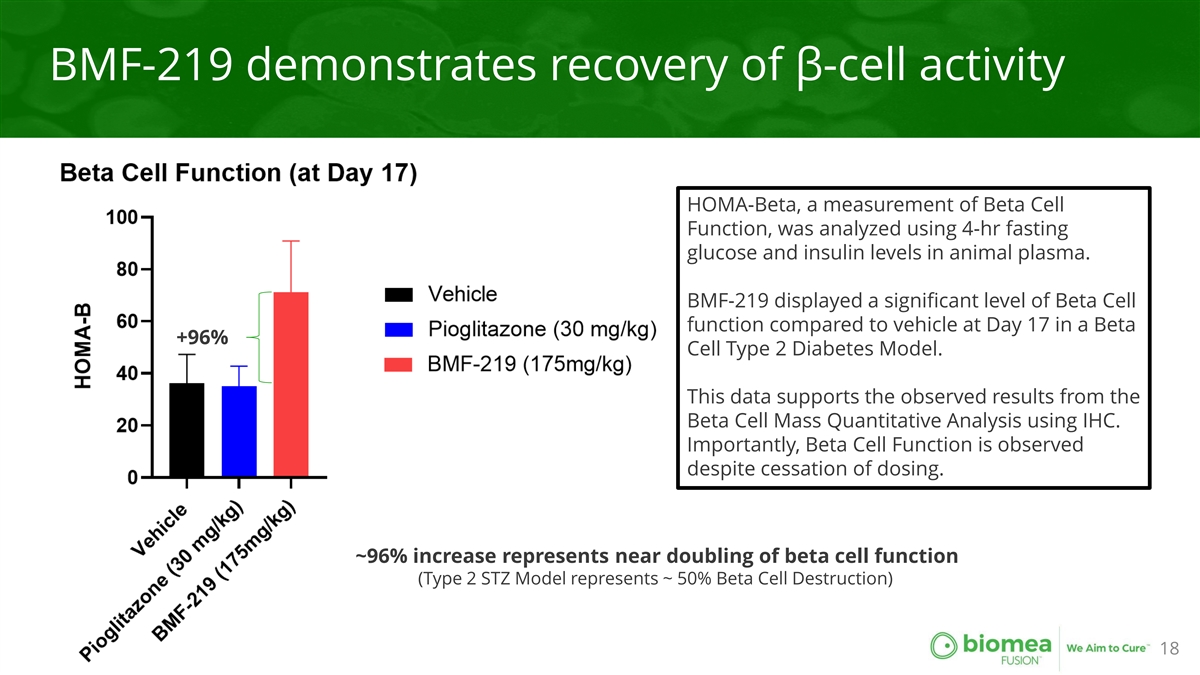
BMF-219 demonstrates recovery of β-cell activity HOMA-Beta, a measurement of Beta Cell Function, was analyzed using 4-hr fasting glucose and insulin levels in animal plasma. BMF-219 displayed a significant level of Beta Cell function compared to vehicle at Day 17 in a Beta +96% Cell Type 2 Diabetes Model. This data supports the observed results from the Beta Cell Mass Quantitative Analysis using IHC. Importantly, Beta Cell Function is observed despite cessation of dosing. ~96% increase represents near doubling of beta cell function (Type 2 STZ Model represents ~ 50% Beta Cell Destruction) 18

BMF-219 significantly reduces blood lipemic levels and body weight in STZ rats Body Weight 550 Triglyceride Total Cholesterol 500 (at Day 17) (at Day 17) 150 250 450 200 400 100 150 * 350 * 100 300 7% reduction after 2 weeks dosing 50 250 50 200 0 0 150 Days *p<0.05 TA dosing start Vehicle Pioglitazone (30 mg/kg) BMF-219 (175mg/kg) 19 -42 -40 -38 -35 -33 -31 -28 -26 -24 -21 -19 -17 -14 -12 -10 -7 -5 -3 1 3 5 8 10 12 15 BW (g) Mean ± SEM Serum Cholesterol (mg/dL) Mean ± SEM Serum Triglyceride (mg/dL) Mean ± SEM
BMF-219 in ex-vivo Human Islet Microtissue (Sneak Peak) 20

Human Donor Islets (Ex-Vivo): Statistically significant increase in beta cells with BMF-219 Solvent BMF-219 BMF-219 (Conc. 2) (Conc. 1) 21 21 Do Not Copy Threshold 375 Threshold 325

Conclusions & Next Steps ▪ BMF-219 was tested in two type 2 diabetic in-vivo models at clinically relevant exposures. BMF-219 treatment in STZ-induced diabetic rat model: ➢ Improved glucose metabolism (OGTT AUC reduction 41%, p<0.05) and reduction in blood lipemic levels and body weight. Minimal efficacy for pioglitazone. ➢ BMF-219 but not pioglitazone increased HOMA-B. BMF-219 treatment in ZDF rat model: ➢ Significant reduction (~50%) in fasting and non-fasting blood glucose levels, ➢ Significant reduction in serum insulin, C-peptide (p<0.05), and HOMA-IR (p<0.001) after two weeks of treatment. ➢ Prolonged glycemic control as evidenced by decreased OGTT glucose levels on day 15 (AUC reduction of 54%, p<0.001) and on day 29 (AUC reduction of 40%, p<0.05, ~2 weeks after the last dose). ➢ Significant reductions in blood lipemic levels (p<0.01) and body weight. ▪ Next Steps: Type 1 Model– Can BMF-219 re-establish Beta Cells from a severely diminished pool? 22

THANK YOU 23