Exhibit 99.1

NASDAQ: SNGX … Rising to the Challenges of Rare Disease Treatment

Forward - Looking Statements 2 This presentation contains forward - looking statements . All statements other than statements of historical facts contained in this presentation, including statements regarding our future results of operations and financial position, business strategy, prospective products and product candidates and their development, regulatory approvals, ability to commercialize our products and product candidates and attract collaborators, reimbursement for our product candidates, research and development costs, timing and likelihood of success, plans and objectives of management for future operations, our ability to obtain and maintain intellectual property protection for our product candidates and their development, competing therapies, and future results of current and anticipated products and product candidates, are forward - looking statements . These statements involve known and unknown risks, uncertainties, and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward - looking statements, many of which are disclosed in detail in our reports and other documents filed with the Securities and Exchange Commission . Because forward - looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward - looking statements as predictions of future events . The events and circumstances reflected in our forward - looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward - looking statements . Except as required by applicable law, we do not plan to publically update or revise any forward - looking statements contained herein, whether as a result of any new information, future events, changed circumstances, or otherwise . Certain information contained in this presentation and statements made orally during this presentation relate to or are based on studies, publications, surveys and other data obtained from third - party sources . In addition, no independent source has evaluated the reasonableness or accuracy of Soligenix, Inc . internal estimates and no reliance should be made on any information or statements made in this presentation relating to or based on such internal estimates .

Company Highlights » Diversified product portfolio in advanced development, targeting rare disease indications in inflammation, oncology and biodefense with $200M+ markets worldwide o Phase 3 clinical program for the treatment of cutaneous T - cell lymphoma (SGX301) - Orphan drug and fast track designations - Pivotal study actively enrolling patients with results second half 2017 o Phase 3 clinical program for the treatment of oral mucositis in head & neck cancer (SGX942) - Fast track and promising innovative medicine (UK) designations - Pivotal study to begin first half 2017 with results second half 2018 o Phase 3 clinical program for the treatment of pediatric Crohn’s disease (SGX203) - Orphan drug and fast track designations - Pivotal study to begin first half 2017 with results second half 2018 » Five development candidates supported in whole or in part by government funding » Robust news flow potential over next 24 months across multiple development programs » Collaborations established with biotech, academia and government agencies » Non - dilutive contract/grant funding provided by the government, including o NIAID contract award of up to $24.7M supporting the development of RiVax™ for pre - exposure to ricin toxin o BARDA and NIAID contract awards of up to $26.7M and $7.0M, respectively, supporting the development of OrbeShield ® for gastrointestinal acute radiation syndrome 3
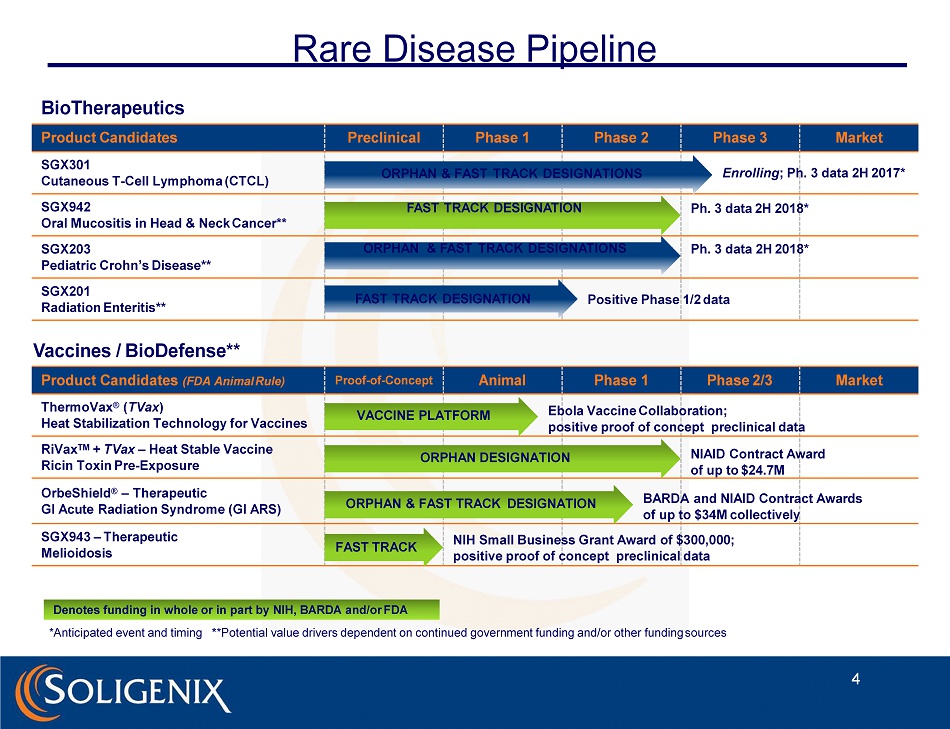
Rare Disease Pipeline SGX301 Cutaneous T - Cell Lymphoma (CTCL) SGX942 Oral Mucositis in Head & Neck Cancer** SGX203 Pediatric Crohn’s Disease** SGX201 Radiation Enteritis** BioTherapeutics P rod u ct C a n didates P reclinical P h a se 1 P h a se 2 P h a se 3 Market Vaccines / BioDefense** Product Candidates (FDA Animal Rule) Proof - of - Con c e pt Animal Phase 1 Phase 2/3 Market ThermoVax ® ( TVax ) Heat Stabilization Technology for Vaccines RiVax TM + TVax – Heat Stable Vaccine Ricin Toxin Pre - Exposure OrbeShield ® – Therapeutic GI Acute Radiation Syndrome (GI ARS) SGX943 – Therapeutic Melioidosis Denotes funding in whole or in part by NIH, BARDA and/or FDA *Anticipated event and timing * *Potential value drivers dependent on continued government funding and/or other funding sources NIAID Contract Award of up to $24.7M BARDA and NIAID Contract Awards of up to $34M collectively Ebola Vaccine Collaboration; positive proof of concept preclinical data NIH Small Business Grant Award of $300,000; positive proof of concept preclinical data FAST TRACK ORPHAN DESIGNATION VACCINE PLATFORM ORPHAN & FAST TRACK DESIGNATION ORPHAN & FAST TRACK DESIGNATIONS Enrolling ; Ph. 3 data 2H 2017* FAST TRACK DESIGNATION Ph. 3 data 2H 2018* ORPHAN & FAST TRACK DESIGNATIONS Ph. 3 data 2H 2018* FAST TRACK DESIGNATION Positive Phase 1/2 data 4
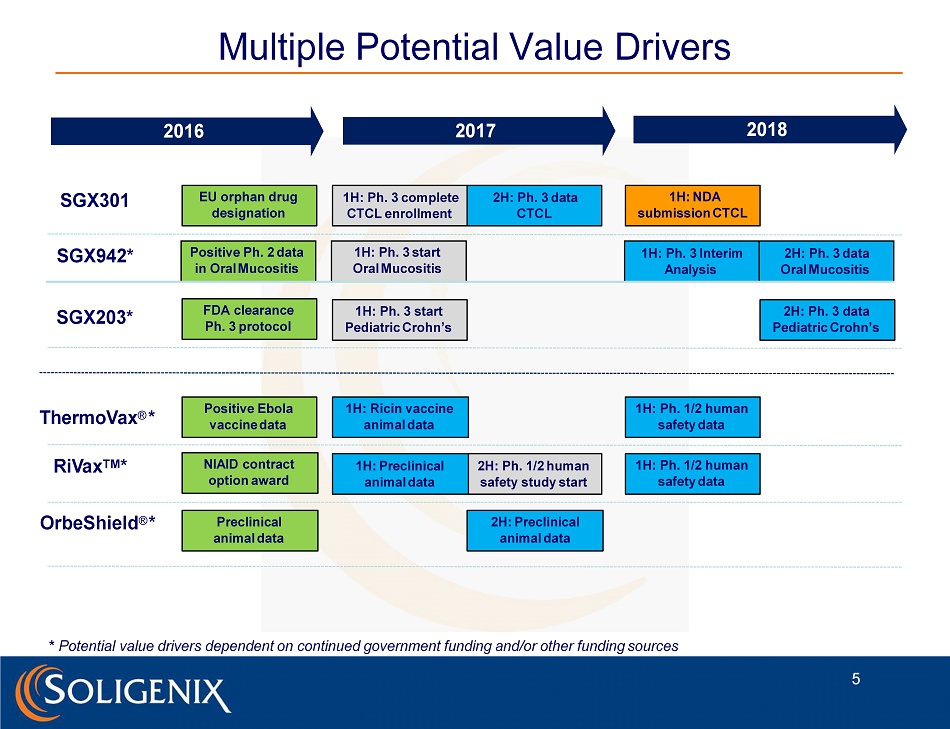
1H: Ph. 1/2 human safety data 2H: Ph. 1/2 human safety study start Multiple Potential Value Drivers SGX203* ThermoVax ® * 2016 SGX301 2017 R i V ax T M * OrbeShield ® * 2018 1H: Ph. 3 start Pediatric Crohn’s * Potential value drivers dependent on continued government funding and/or other funding sources Preclinical animal data EU orphan drug designation 1H: Ph. 3 complete CTCL enrollment 1H: Preclinical animal data FDA clearance Ph. 3 protocol Positive Ebola vaccine data 1H: NDA submission CTCL 2H: Preclinical animal data NIAID contract option award 1H: Ricin vaccine animal data 1H: Ph. 1/2 human safety data 2H: Ph. 3 data CTCL 5 SGX942* Positive Ph. 2 data in Oral Mucositis 1H: Ph. 3 start Oral Mucositis 1H: Ph. 3 Interim Analysis 2H: Ph. 3 data Oral Mucositis 2H: Ph. 3 data Pediatric Crohn’s
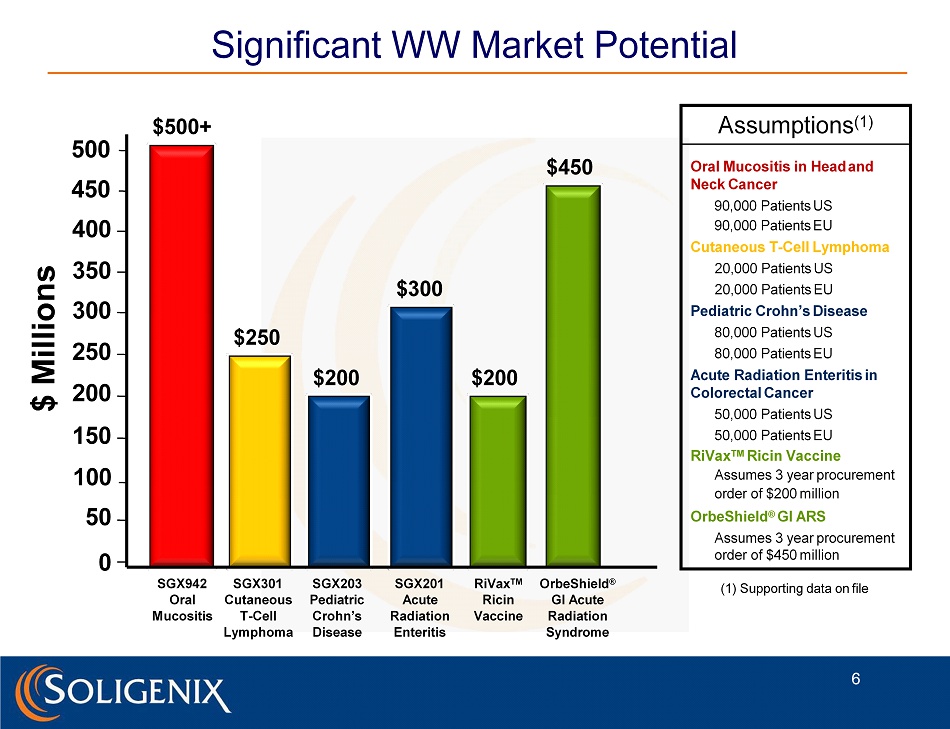
Significant WW Market Potential $300 $ M i l l ions $250 O r be S hie l d ® GI Acute Radiation Syndrome SGX301 Cutaneous T - Cell L y m p homa Assumptions (1) Oral Mucositis in Head and Neck Cancer 90,000 Patients US 90,000 Patients EU Cutaneous T - Cell Lymphoma 20,000 Patients US 20,000 Patients EU Pediatric Crohn’s Disease 80,000 Patients US 80,000 Patients EU Acute Radiation Enteritis in Colorectal Cancer 50,000 Patients US 50,000 Patients EU RiVax TM Ricin Vaccine Assumes 3 year procurement order of $200 million OrbeShield ® GI ARS Assumes 3 year procurement order of $450 million SGX201 Acute Radiation Enteritis $200 Ri V a x TM Ricin V a c cine $200 $450 500 450 400 350 300 250 200 150 100 50 0 $500+ 6 SGX942 Oral M ucositis (1) Supporting data on file SGX 203 P ediat r ic Crohn’s Disease

Experienced Management and Board of Directors Christopher J. Schaber, PhD President & CEO • 27 years of experience • Discovery Laboratories (COO) • Acute Therapeutics (Co - Founder) • Ohmeda Pharmaceuticals • The Liposome Company • Wyeth Ayerst Oreola Donini, PhD Chief Scientific Officer • 15 years of experience • Inimex Pharmaceuticals • ESSA Pharma, Inc • Kinetek Pharmaceuticals Karen Krumeich Chief Financial Officer Rasappa Arumugham, PhD VP, Biopharm Development • 26 years of experience • Merck • Pfizer • Wyeth Keith Brownlie, CPA • 35 years of experience • Ernst & Young Gregg Lapointe, CPA, MBA • 20 years of experience • Cerium Pharmaceuticals (CEO) • Sigma - Tau Pharmaceuticals • AstenJohnson • Price Waterhouse Coopers Robert Rubin, MD • 36 years of experience • The Lewin Group • Georgetown School of Medicine • Former Assistant Surgeon General of the United States Jerome Zeldis, MD, PhD • 33 years of experience • Celgene Corporation (CMO) • Sandoz • Janssen Research Institute Marco Brughera, DVM • 30 years of experience • Sigma - Tau SpA (Global Head, Rare Disease Franchise) • Pfizer • Pharmacia Richard Straube, MD Chief Medical Officer • 30 years of experience • Stealth Peptides Inc. • INO Therapeutics • Ohmeda Pharmaceuticals • Centocor • 25 years of experience • Cerecor • Mela Sciences • Bristol - Myers Squibb 7

Targeted Approach to Treating Oncology & Inflammation BioTherapeutics 8
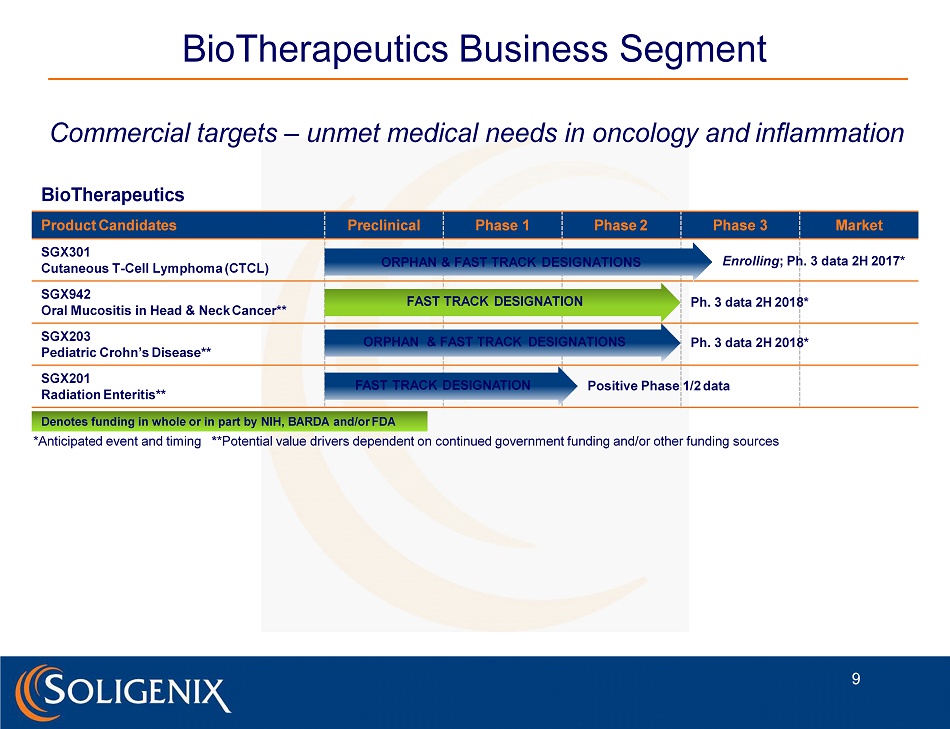
BioTherapeutics Business Segment Commercial targets – unmet medical needs in oncology and inflammation BioTherapeutics Product Candidates SGX301 Cutaneous T - Cell Lymphoma (CTCL) SGX942 Oral Mucositis in Head & Neck Cancer** SGX203 Pediatric Crohn’s Disease** SGX201 Radiation Enteritis** Preclinical Phase 1 Phase 2 ORPHAN & FAST TRACK DESIGNATIONS Phase 3 Market Enrolling ; Ph. 3 data 2H 2017* FAST TRACK DESIGNATION Ph. 3 data 2H 2018* ORPHAN & FAST TRACK DESIGNATIONS Ph. 3 data 2H 2018* FAST TRACK DESIGNATION Positive Phase 1/2 data Denotes funding in whole or in part by NIH, BARDA and/or FDA *Anticipated event and timing * *Potential value drivers dependent on continued government funding and/or other funding sources 9
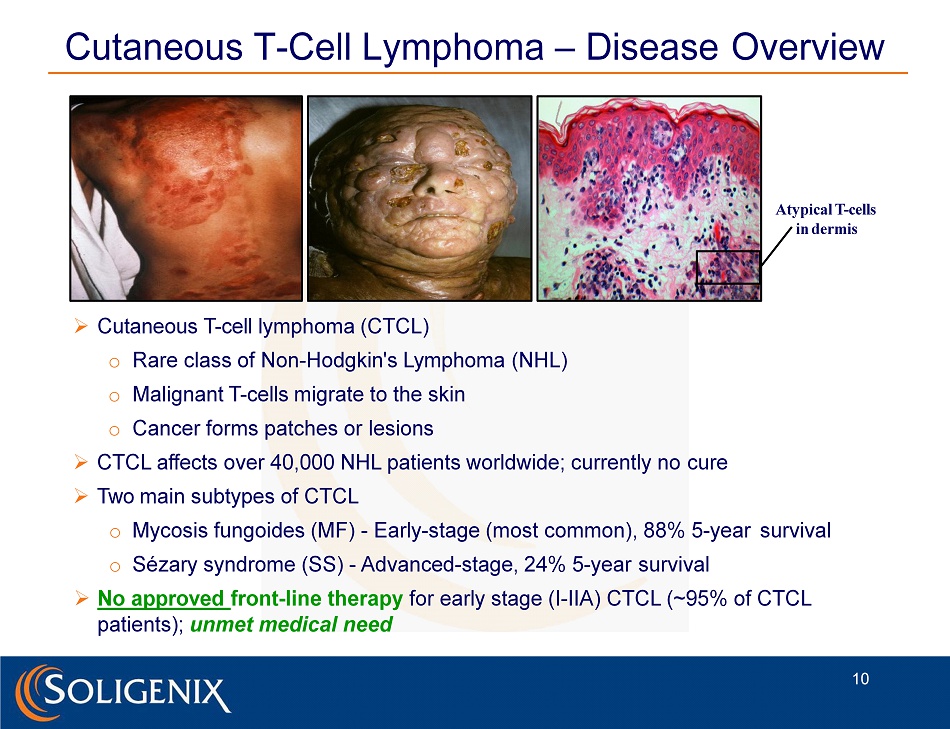
Cutaneous T - Cell Lymphoma – Disease Overview » Cutaneous T - cell lymphoma (CTCL) o Rare class of Non - Hodgkin's Lymphoma (NHL) o Malignant T - cells migrate to the skin o Cancer forms patches or lesions » CTCL affects over 40,000 NHL patients worldwide; currently no cure » Two main subtypes of CTCL o Mycosis fungoides (MF) - Early - stage (most common), 88% 5 - year survival o Sézary syndrome (SS) - Advanced - stage, 24% 5 - year survival » No approved front - line therapy for early stage (I - IIA) CTCL (~95% of CTCL patients); unmet medical need Atypical T - cells in dermis 10

SGX301 – Synthetic Hypericin SGX301 is a first - in - class, topical drug applied to CTCL skin lesions followed by activation with safe, visible, fluorescent light to kill malignant T - cells » Affects over 40,000 patients annually worldwide » No approved front - line therapy for early stage (I - IIA) CTCL (~95% of CTCL patients); unmet medical need » Most common (unapproved) therapy used for early - stage disease is psoralen given with ultraviolet A (UVA) light, referred to as PUVA » PUVA contains Black Box warning for potential malignancies (melanoma) due to psoralen being mutagenic and light source (UVA) being carcinogenic Market O p p ort u ni t y De v elo p me n t Status » FDA Orphan Drug and Fast Track designations granted » Phase 1 study demonstrated safety and tolerability » Phase 2 double - blind, placebo - controlled, multi - center study demonstrated significant (p < 0.04) response » Pivotal Phase 3 trial actively enrolling ~120 subjects » Results expected 2H 2017 11
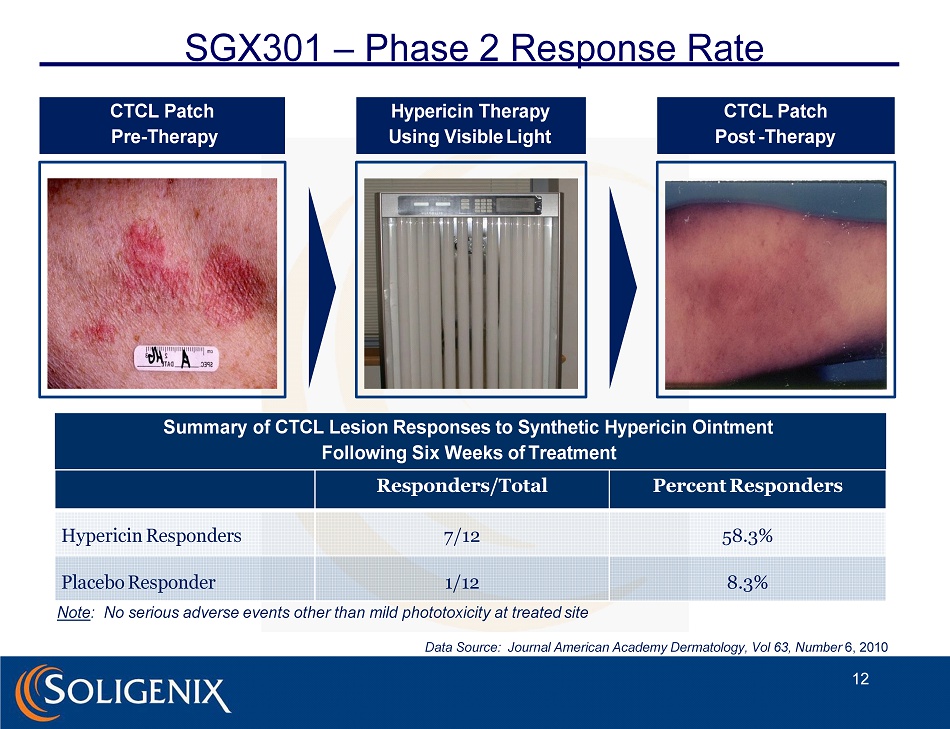
CTCL Patch P r e - Th era p y Hypericin Therapy Using Visible Light CTCL Patch Post - Therapy SGX301 – Phase 2 Response Rate 12 Summary of CTCL Lesion Responses to Synthetic Hypericin Ointment Following Six Weeks of Treatment Responders/Total Percent Responders Hypericin Responders 7/12 58.3% Placebo Responder 1/12 8.3% Note : No serious adverse events other than mild phototoxicity at treated site Data Source: Journal American Academy Dermatology, Vol 63, Number 6, 2010
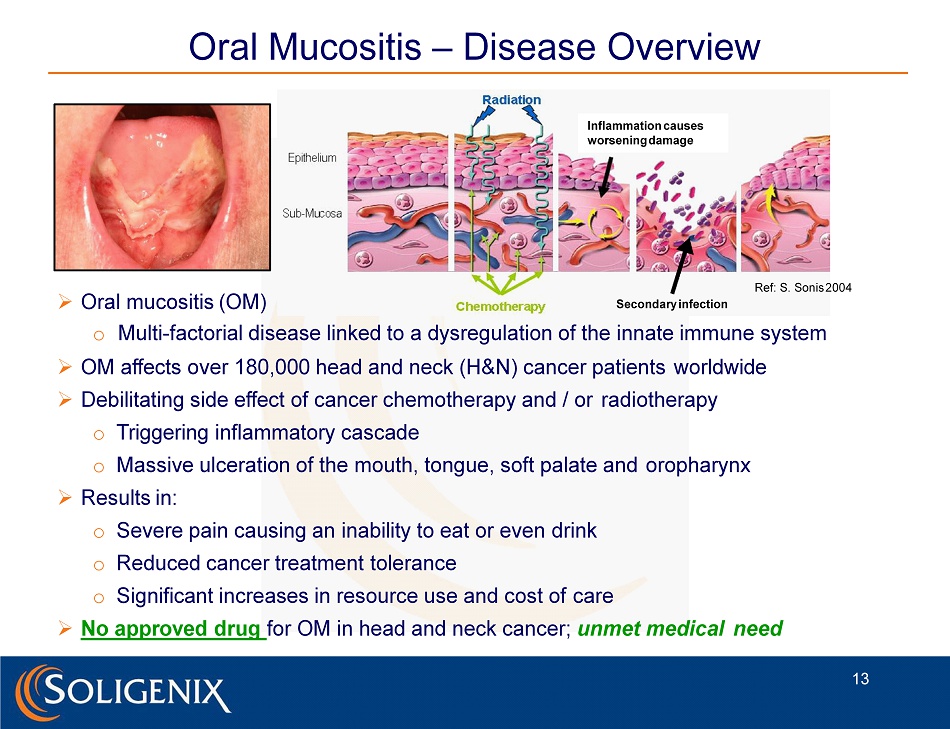
Oral Mucositis – Disease Overview » Oral mucositis (OM) o Multi - factorial disease linked to a dysregulation of the innate immune system » OM affects over 180,000 head and neck (H&N) cancer patients worldwide » Debilitating side effect of cancer chemotherapy and / or radiotherapy o Triggering inflammatory cascade o Massive ulceration of the mouth, tongue, soft palate and oropharynx » Results in: o Severe pain causing an inability to eat or even drink o Reduced cancer treatment tolerance o Significant increases in resource use and cost of care » No approved drug for OM in head and neck cancer; unmet medical need Ref: S. Sonis 2004 Inflammation causes worsening damage Secondary infection 13

SGX942 – Innate Defense Regulator SGX942 (dusquetide) is a first - in - class, injectable drug, called an Innate Defense Regulator (IDR), that modulates the body’s innate immune system to reduce inflammation » OM affects over 180,000 H&N cancer patients worldwide » No approved drug for OM in H&N cancer; unmet medical need Market O p p ort u ni t y De v elo p me n t Status 14 » Only approved drug for OM is palifermin in transplantation; contra - indicated for patients with solid tumors like H&N cancer » Exclusive commercial collaboration with SciClone in China » FDA Fast Track designation granted » UK MHRA Promising Innovative Medicine designation granted » Phase 1 study in 84 healthy volunteers demonstrated safety » Phase 2 double - blind, placebo - controlled, multi - center study in 111 H&N cancer patients with OM demonstrated significant (p=0.04) response o 50% reduction in duration of severe OM in overall population o 67% reduction in duration of severe OM in highest risk population receiving at least 55 Gy radiation and more aggressive (80 - 100 mg/m 2 every 3 rd week) chemotherapy » Pivotal Phase 3 protocol clearance from FDA and EMA with study initiation targeted 1H 2017
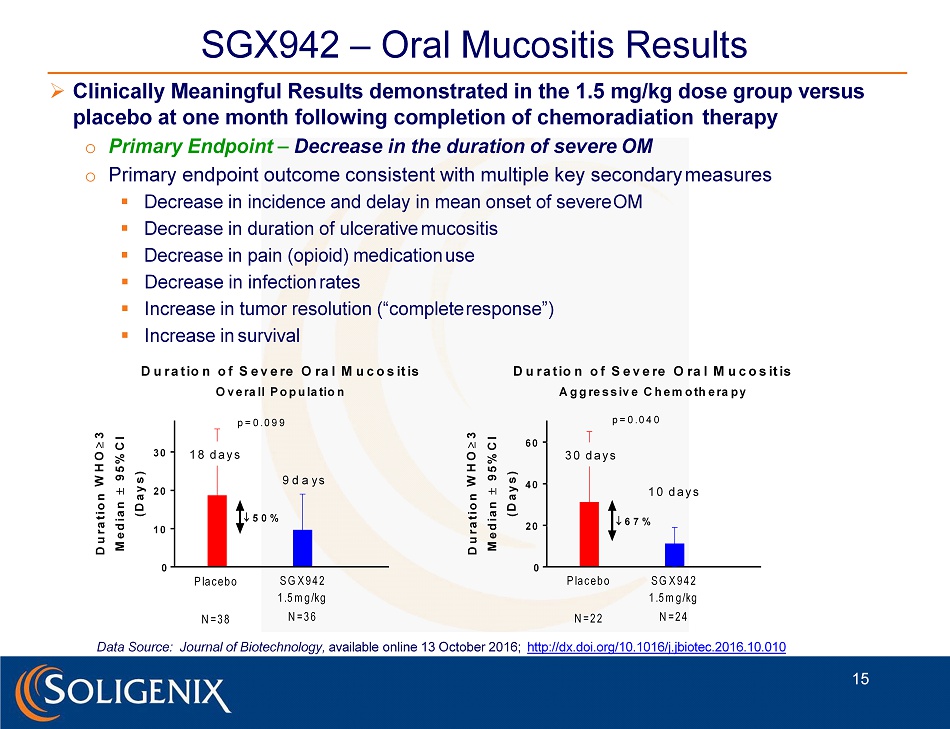
0 1 0 2 0 3 0 D u r a t io n o f S e v e re O ra l M u c o s it is O v e ra ll P o p u la tio n D u r a t i o n W H O 3 M e d i a n 9 5 % C I ( D a y s ) P la c e b o N = 3 8 S G X 9 4 2 1 .5 m g /kg N = 3 6 1 8 d a y s 9 d a y s p = 0 . 0 9 9 5 0 % 0 2 0 4 0 6 0 D u r a t io n o f S e v e re O ra l M u c o s it is A g g re s s iv e C h e m o th e ra p y D u r a t i o n W H O 3 M e d i a n 9 5 % C I ( D a y s ) P la c e b o N = 2 2 S G X 9 4 2 1 .5 m g /kg N = 2 4 3 0 d a y s 1 0 d a y s p = 0 . 0 4 0 6 7 % SGX942 – Oral Mucositis Results » Clinically Meaningful Results demonstrated in the 1.5 mg/kg dose group versus placebo at one month following completion of chemoradiation therapy o Primary Endpoint – Decrease in the duration of severe OM o Primary endpoint outcome consistent with multiple key secondary measures ▪ Decrease in incidence and delay in mean onset of severe OM ▪ Decrease in duration of ulcerative mucositis ▪ Decrease in pain (opioid) medication use ▪ Decrease in infection rates ▪ Increase in tumor resolution (“complete response”) ▪ Increase in survival 15 Data Source: Journal of Biotechnology, available online 13 October 2016; http://dx.doi.org/10.1016/j.jbiotec.2016.10.010
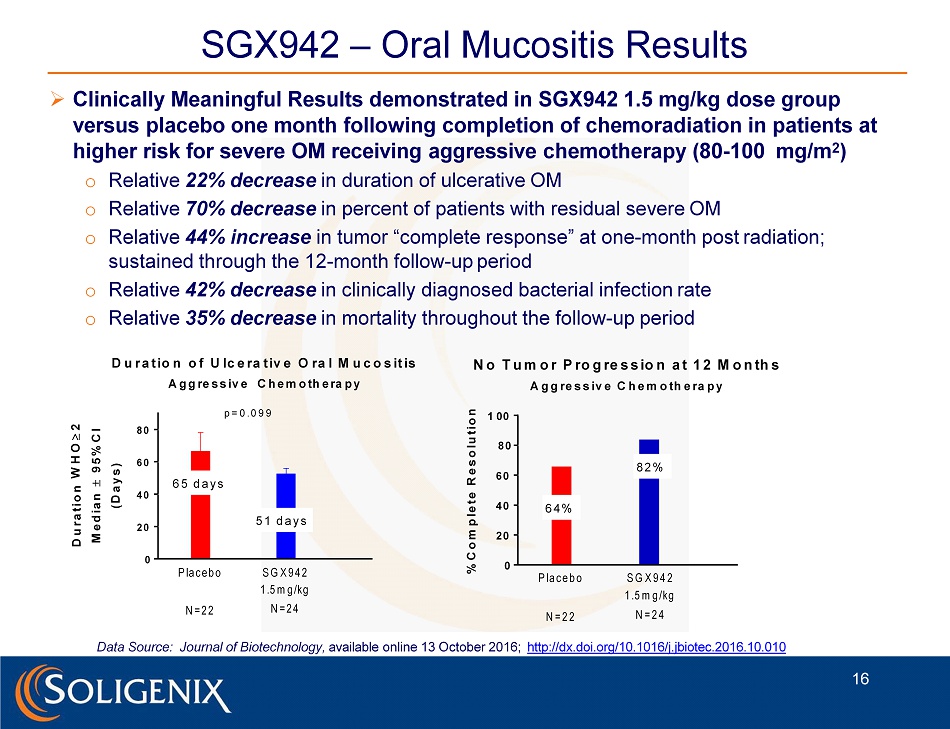
SGX942 – Oral Mucositis Results » Clinically Meaningful Results demonstrated in SGX942 1.5 mg/kg dose group versus placebo one month following completion of chemoradiation in patients at higher risk for severe OM receiving aggressive chemotherapy (80 - 100 mg/m 2 ) o Relative 22% decrease in duration of ulcerative OM o Relative 70% decrease in percent of patients with residual severe OM o Relative 44% increase in tumor “complete response” at one - month post radiation; sustained through the 12 - month follow - up period o Relative 42% decrease in clinically diagnosed bacterial infection rate o Relative 35% decrease in mortality throughout the follow - up period Data Source: Journal of Biotechnology, available online 13 October 2016; http://dx.doi.org/10.1016/j.jbiotec.2016.10.010 0 2 0 4 0 6 0 D u r a t io n o f U lc e ra t iv e O ra l M u c o s it is A g g re s s iv e C h e m o th e ra p y D u r a t i o n W H O 2 M e d i a n 9 5 % C I ( D a y s ) P la c e b o N = 2 2 S G X 9 4 2 1 .5 m g /kg N = 2 4 6 5 d a y s 5 1 d a y s p = 0 . 0 9 9 0 2 0 4 0 6 0 8 0 8 0 1 0 0 N o T u m o r P ro g re s s io n a t 1 2 M o n th s A g g re s s iv e C h e m o th e ra p y % C o m p l e t e R e s o l u t i o n P la c e b o N = 2 2 S G X 9 4 2 1 .5 m g /kg N = 2 4 6 4 % 8 2 % 16

Pediatric Crohn’s Disease – Disease Overview » Pediatric Crohn’s disease o Chronic inflammatory disorder of the gastrointestinal (GI) tract o Diarrhea, rectal bleeding and abdominal pain » Resulting in growth failure, malnutrition, pubertal delay and bone demineralization » Over 160,000 children/adolescents with Crohn’s disease worldwide » Location of disease o Adult predominantly lower GI tract o 50% of children have involvement in the upper GI tract » No approved drug for mild - to - moderate pediatric Crohn’s disease in the US; unmet medical need Normal mucosa under endoscopy 17

SGX203 – Beclomethasone Dipropionate SGX203 is a proprietary oral formulation of immediate and delayed release beclomethasone 17,21 - dipropionate (BDP) tablets to treat GI inflammation with less toxicity than the current standard systemic steroid therapy » 160,000 children/adolescents with Crohn’s disease worldwide – 50% have inflammation of upper GI » No approved drug for mild - to - moderate; unmet medical need » Currently unapproved systemic steroids used front - line cause adrenal suppression, growth impairment, bone demineralization Market O p p ort u ni t y Development Status » Oral BDP delivers high GI steroid effects with minimal (~35%) systemic side effects » Remicade and Humira only approved products in Pediatric Crohn’s in US – generally used after steroids fail » Both contain Black Box warning for increased risk of infection and potential malignancy (T cell lymphoma) » FDA Orphan Drug and Fast Track designations granted » Active ingredient BDP FDA approved for over 40 years in other delivery forms (e.g., aerosol) to treat diseases such as asthma » ~350 subjects treated with oral BDP to date in multiple trials » Pivotal Phase 3 adaptive trial in ~150 subjects targeted 1H 2017 » Results expected 2H 2018 18

Vaccine/BioDefense Addressing Critical Concerns for Industry and Government 19

Vaccine/BioDefense Business Segment Proof - of - Con c e pt Product Candidates (FDA Animal Rule) Animal Phase 1 Phase 2/3 Market ThermoVax ® ( TVax ) Heat Stabilization Technology for Vaccines RiVax TM + TVax – Heat Stable Vaccine Ricin Toxin Pre - Exposure OrbeShield ® – Therapeutic GI Acute Radiation Syndrome (GI ARS) SGX943 – Therapeutic Melioidosis NIH Small Business Grant Award of $300,000; positive proof of concept preclinical data FAST TRACK ORPHAN DESIGNATION VACCINE PLATFORM Ebola Vaccine Collaboration; positive proof of concept preclinical data ORPHAN & FAST TRACK DESIGNATION Denotes funding in whole or in part by NIH, BARDA and/or FDA *Anticipated event and timing * *Potential value drivers dependent on continued government funding and/or other funding sources 20 Funded by Government – Medical Countermeasures for Civilian and Military Use Vaccines / BioDefense** NIAID Contract Award of up to $24.7M BARDA and NIAID Contract Awards of up to $34M collectively
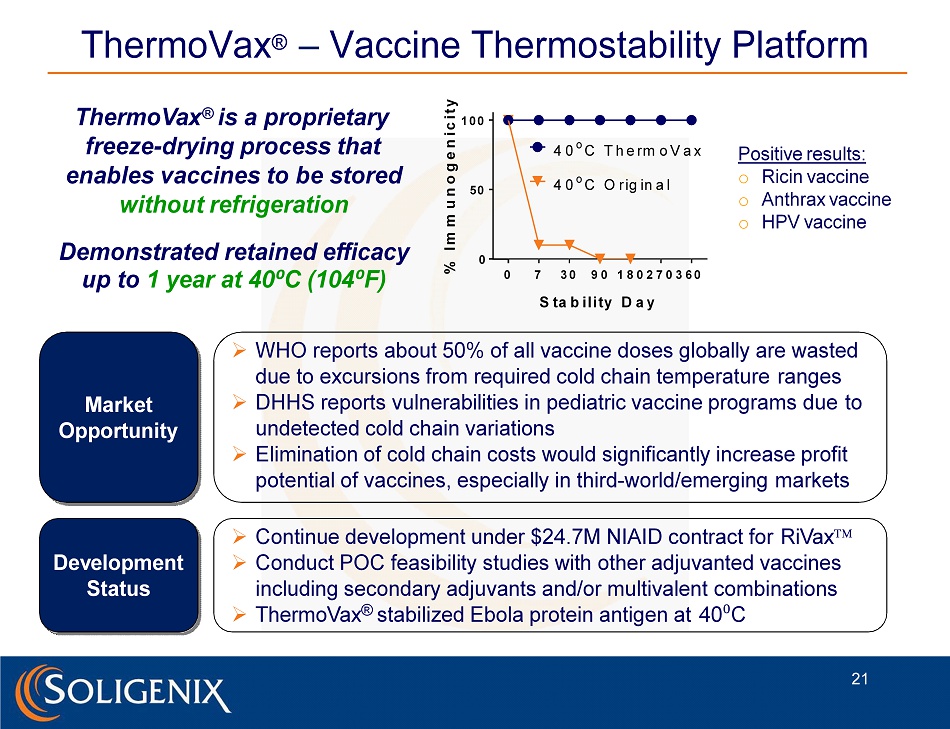
ThermoVax ® – Vaccine Thermostability Platform ThermoVax ® is a proprietary freeze - drying process that enables vaccines to be stored without refrigeration Demonstrated retained efficacy up to 1 year at 40 ⁰ C (104 ⁰ F) Market O p p ort u ni t y De v elo p me n t Status » WHO reports about 50% of all vaccine doses globally are wasted due to excursions from required cold chain temperature ranges » DHHS reports vulnerabilities in pediatric vaccine programs due to undetected cold chain variations » Elimination of cold chain costs would significantly increase profit potential of vaccines, especially in third - world/emerging markets » Continue development under $24.7M NIAID contract for RiVax » Conduct POC feasibility studies with other adjuvanted vaccines including secondary adjuvants and/or multivalent combinations » ThermoVax ® stabilized Ebola protein antigen at 40 ⁰ C Positive results: o Ricin vaccine o Anthrax vaccine o HPV vaccine 0 7 3 0 9 0 1 8 0 2 7 0 3 6 0 0 5 0 1 0 0 S ta b i l i ty D a y % I m m u n o g e n i c i t y 4 0 o C T h e rm o V a x 4 0 o C O r ig in a l 21

Heat - stable ricin vaccine provided 100% protection in a non - human primate aerosol challenge model (right) Demonstrated safety in Phase 1 studies Market O p p ort u ni t y De v elo p me n t Status » Ricin toxin vaccine of rising interest to US due to recent terrorist threats and ease of castor bean procurement and production » Government has placed priority on development activities » Potential to be first approved ricin toxin vaccine » FDA Orphan Drug designation granted » Development agreement with Emergent BioSolutions for manufacturing » NIAID contract award of up to $24.7M over 6 years RiVax TM – Ricin Toxin Vaccine % S u r v i v a l 0 5 1 0 D a y s P o s t - In to x ic a t io n 1 5 0 2 5 5 0 7 5 1 0 0 S h a m ( N = 6 ) V a c c in e ( N = 1 2 ) p < 0 .0 0 0 1 22 * L a te d e a th n o t d ire c tly a ttrib u te d to r ic in in to x ic a tio n
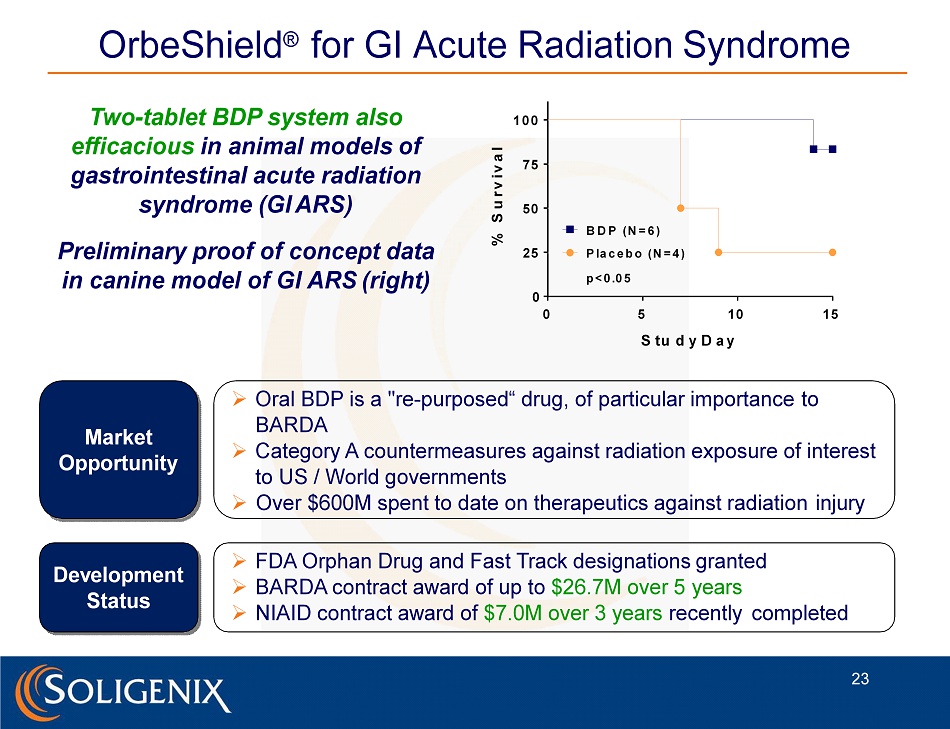
Two - tablet BDP system also efficacious in animal models of gastrointestinal acute radiation syndrome (GI ARS) Preliminary proof of concept data in canine model of GI ARS (right) Market O p p ort u ni t y De v elo p me n t Status » Oral BDP is a "re - purposed“ drug, of particular importance to BARDA » Category A countermeasures against radiation exposure of interest to US / World governments » Over $600M spent to date on therapeutics against radiation injury » FDA Orphan Drug and Fast Track designations granted » BARDA contract award of up to $26.7M over 5 years » NIAID contract award of $7.0M over 3 years recently completed OrbeShield ® for GI Acute Radiation Syndrome 0 5 1 0 1 5 0 2 5 5 0 7 5 1 0 0 S tu d y D a y % S u r v i v a l p < 0 .0 5 B D P ( N = 6 ) P la c e b o ( N = 4 ) 23

Investment Highlights » Diversified product portfolio in advanced development, targeting rare disease indications in inflammation, oncology and biodefense with $200M+ markets worldwide o Phase 3 clinical program for the treatment of cutaneous T - cell lymphoma (SGX301) - Orphan drug and fast track designations - Pivotal study actively enrolling patients with results second half 2017 o Phase 3 clinical program for the treatment of oral mucositis in head & neck cancer (SGX942) - Fast track and promising innovative medicine (UK) designations - Pivotal study to begin first half 2017 with results second half 2018 o Phase 3 clinical program for the treatment of pediatric Crohn’s disease (SGX203) - Orphan drug and fast track designations - Pivotal study to begin first half 2017 with results second half 2018 » Five development candidates supported in whole or in part by government funding » Robust news flow potential over next 24 months across multiple development programs » Collaborations established with biotech, academia and government agencies » Non - dilutive contract/grant funding provided by the government, including o NIAID contract award of up to $24.7M supporting the development of RiVax™ for pre - exposure to ricin toxin o BARDA and NIAID contract awards of up to $26.7M and $7.0M, respectively, supporting the development of OrbeShield ® for gastrointestinal acute radiation syndrome 24

THANK YOU www.soligenix.com Rising to the Challenges of Rare Disease Treatment