 Tosedostat Exclusive Marketing License and Co-Development Agreement Exhibit 99.2 |
 Forward looking statements 2 This presentation contains forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. The forward-looking statements contained in this presentation include statements about future financial and operating results, and risks and uncertainties that could affect Cell Therapeutic Inc.’s (“CTI”) products under development. These statements are based on management’s current expectations and beliefs and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. These statements are not guarantees of future performance, involve certain risks, uncertainties and assumptions that are difficult to predict, and are based upon assumptions as to future events that may not prove accurate. Therefore, actual outcomes and results may differ materially from what is expressed herein. In any forward-looking statement in which CTI expresses an expectation or belief as to future results, such expectation or belief is expressed in good faith and believed to have a reasonable basis, but there can be no assurance that the statement or expectation or belief will result or be achieved or accomplished. The following factors, among others, could cause actual results to differ materially from those described in the forward-looking statements: risks associated with preclinical, clinical and sales and marketing developments in the biopharmaceutical industry in general and in particular including, without limitation, the potential failure of Tosedostat to prove safe and effective (including complete and overall response rates) for the treatment of blood related cancers and selected tumors as determined by the U.S. Food and Drug Administration (the “FDA”) or other regulatory agencies in the territories where CTI has exclusive rights, that the FDA may not accept the proposed clinical trial design of Tosedostat and/or may request additional clinical trials, that clinical trials may not demonstrate the safety and effectiveness of Tosedostat, that CTI cannot predict or guarantee the pace or geography of enrollment of clinical trials of Tosedostat, including whether or not the majority of the patients will be enrolled in the U.S., that CTI cannot predict or guarantee the outcome or results of clinical trials of Tosedostat, that CTI’s cannot predict or guarantee whether the exclusive marketing license and co-development agreement with Chroma Therapeutics Ltd (the “Agreement”) will strengthen CTI’s business, financial condition, operating results and prospects or the trading price of CTI’s securities, that CTI cannot predict or guarantee whether milestones will be achieved pursuant to the Agreement, CTI’s ability to continue to raise capital as needed to fund its operations, determinations by regulatory, patent and administrative governmental authorities, competitive factors, technological developments, costs of developing, producing and selling CTI’s products under development; and other economic, business, competitive, and/or regulatory factors affecting CTI’s business generally, including those set forth in CTI’s filings with the U.S. Securities and Exchange Commission, including its Annual Report on Form 10-K for its most recent fiscal year and its most recent Quarterly Report on Form 10-Q, especially in the “Factors Affecting Our Operating Results” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” sections, and its Current Reports on Form 8-K. Except as may be required by law, CTI does not intend to update or alter its forward-looking statements whether as a result of new information, future events, or otherwise. |
 Product/Deal Highlights Tosedostat: Novel tumor-selective targeted therapy • Oral, once daily dosing, well tolerated Encouraging phase I/II data • 27% mono-therapy response rate elderly r/r AML • Ongoing trials provide clinical news flow through 2012 Potentially rapid registration timeline: pivotal study estimated to start Q4-2011 Leverages CTI’s access to and expertise in blood-related cancer market • Potential development cost and sales synergies with pixantrone Exclusive rights in Americas on reasonable deal terms Targets billion dollar market applications 3 |
 Chroma Therapeutics Public company expertise in a private company • Seasoned management and Board of Directors • Facilitates co-development interactions and synergies Investor group • Abingworth, Wellcome Trust, Essex Woodlands, Nomura, GSK, Gilde Innovators in new targeted therapies harnessing chromatin biology and novel cell accumulation (ESM*) technology Validating partnership with GlaxoSmithKline 4 * Esterase Sensitive Motifs |
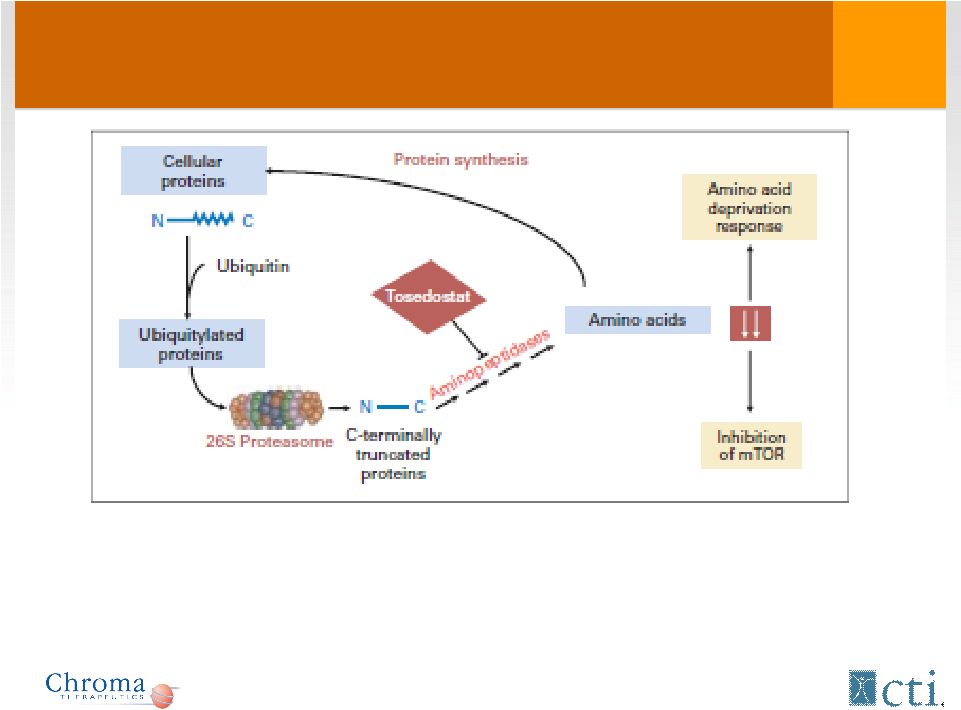 Tosedostat: Mechanism of Action 5 - Targets members of aminopeptidase family - Induces genes characteristic of Amino Acid Deprivation response (AADR) - AADR changes only seen in transformed (tumor) cells and not normal cells |
 Consequences of amino acid deprivation in leukemic cells Cells attempt to solve deficiency (up-regulation of amino acid transporters – AADR response) Cells switch off protein synthesis (e.g. remove mTOR drive) Cells switch on stress-related pathways (NFKB, etc.) Cells produce pro-apoptotic regulators, CHOP, NOXA, etc., that lead to cell death Studies indicate that these effects are not seen in either normal human cells treated with Tosedostat or in insensitive tumor cells. 6 |
 Tosedostat Clinical Program Blood-Related Cancers 7 |
 Acute Myeloid Leukemia (AML) Overview One of the most common types of leukemia in adults U.S. Incidence >12,000/yr Median age at diagnosis – 67 years Poor prognostic factors include • Elderly (>60 yo.) • Elevated WBC counts (>100,000/mm 3 ) • Secondary AML resulting from other primary cancers • History of MDS or another antecedent hematologic disorder Response rates and survival in elderly r/r disease is grim (10%, <3months) 8 |
 Acute Myeloid Leukemia (AML) Overview Cytarabine (Ara-C) and anthracyclines mainstay 1 st line • ~75% relapse within 1 year Approximately 2/3 of elderly patients do not tolerate intensive chemotherapy • 70% of AML patients are >60yrs old • Up to 35% treatment related mortality Significant unmet medical need in elderly AML for • Better tolerated agents • Oral, chronic dosing • Better efficacy 9 |
 Phase I/II (002) Study 10 |
 Phase I/II Study (002) Objectives • Define MTD and anti-leukemic activity of oral Tosedostat Study population • Elderly (>60yrs), r/r AML, MDS, MM not eligible for chemo Phase I (3+3) designed for up to 84 days of dosing • 60mg, 90mg, 130mg, 180mg (oral, 1xdaily) • MTD determined during first 28 days Phase II determines activity in AML, high risk MDS • Bone marrow assessment days 28, 56, 84 • Extended dosing >84 days permissible 11 |
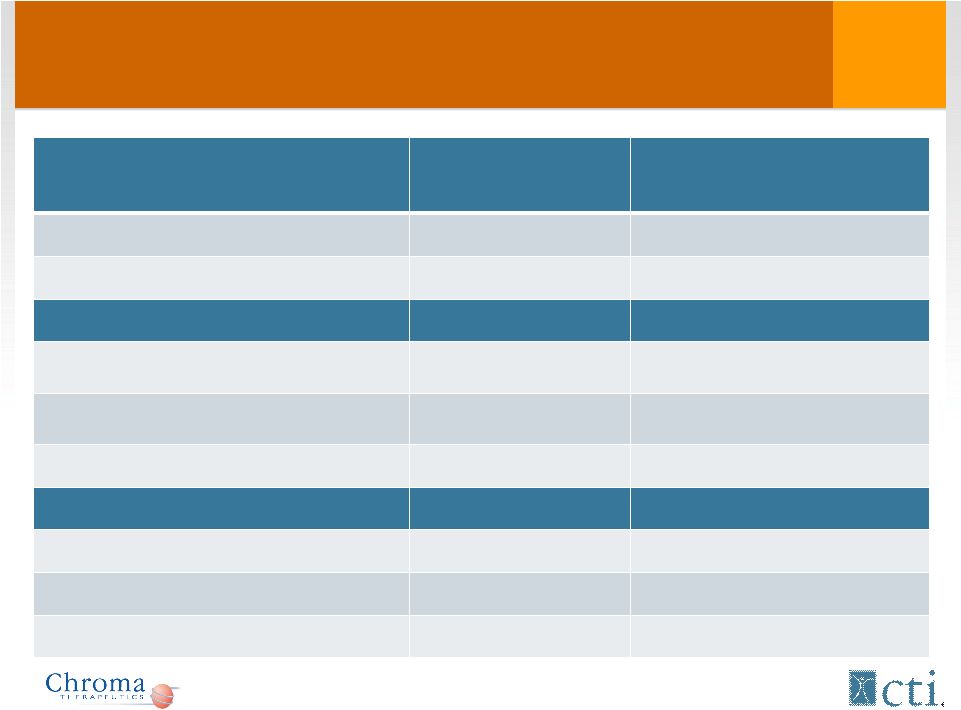 Phase I/II: Patient characteristics * Not reported for 1 patient Phase I (n = 16) Phase II (n = 43) Median age, years (range) 68.4 (45-84) 68 (35-83) Male/Female 13/3 28/15 Diagnosis No. (%) No. (%) AML 13 (81.3) 40 (93) MDS 1 (6.3) 3 (7) MM 2 (12.5) 0 (0) ECOG at baseline No. (%) No. (%)* 0 8 (50.0) 16 (37.2) 1 6 (37.5) 18 (41.9) 2 2 (12.5) 8 (18.6) 12 |
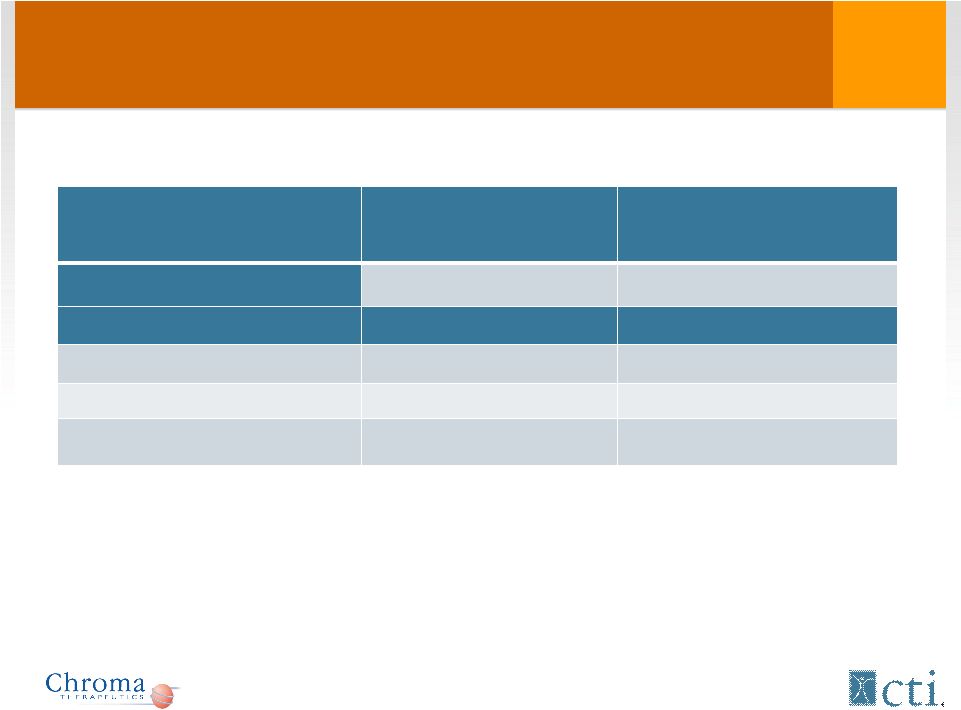 Phase I/II: Patient characteristics (cont) * Excluding the 2 patients that did not receive CHR-2797 in phase II Phase I (n = 16) Phase II (n = 43) Median time from diagnosis 3.1 years (0.1-8.8) 7 months (0-39) Previous treatment* Untreated 4 (25%) 12 (29.3%) Cytotoxic Therapy 12 (75%) 29 (70.7%) Median # priors 2 (range 0-8) 1 (range 1-8) 13 |
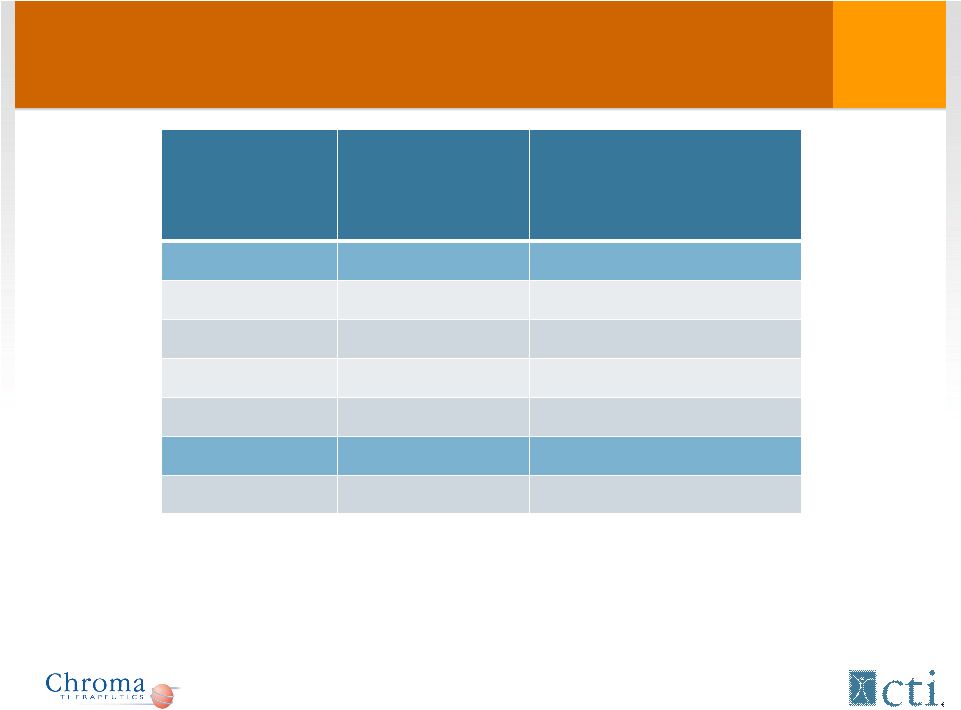 Tosedostat: Treatment duration* * Including extended treatment Dose No. of patients Median duration of therapy, days (range) Phase I 60 mg 3 140 (119-196) 90 mg 4 40 (13-174) 130 mg 6 69 (42-222) 180 mg 3 21 (9-77) Phase II 130 mg 41 72 (1-471) 14 |
 Phase I/II: Dose Limiting Toxicities The recommended dose of 130 mg for the phase II part was determined during phase I and was based on the following DLT’s: DLT Dose No. of patients No. of occurrences ALT elevation (Grade 3) 130 mg 1 1 Thrombocytopenia (Grade 4) 180 mg 2 2 15 |
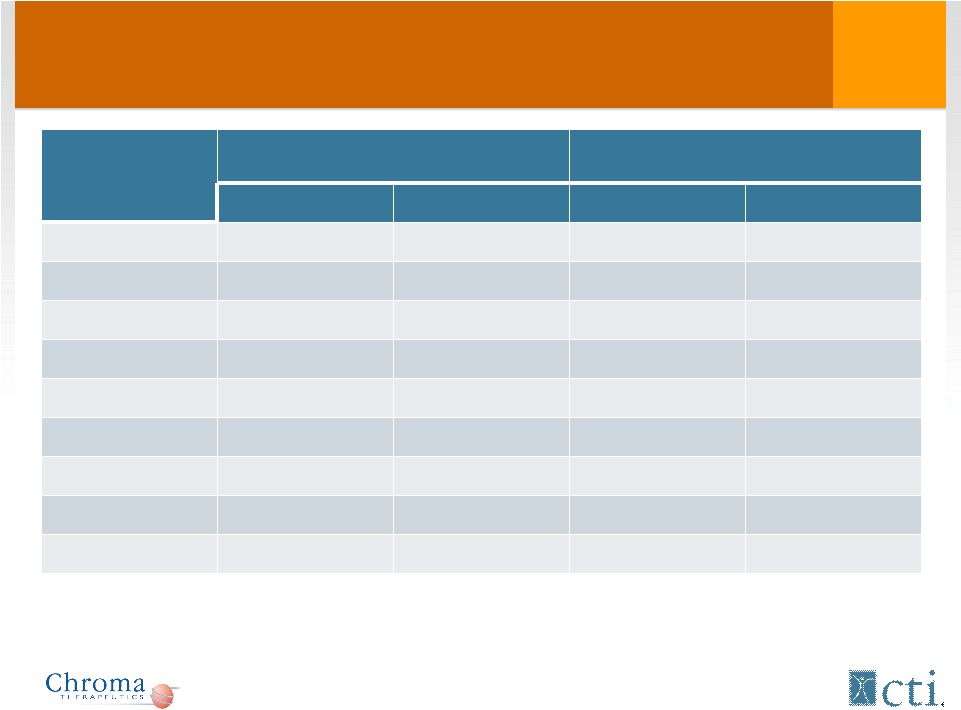 Most common adverse events in phase I-II: time of occurrence Adverse Event (Preferred Term) Incidence Day 1 - 28 Incidence Day 28 - End Patients (n=57) % Patients (n=57) % Fatigue 11 19 19 33 Thrombocytopenia 23 40 4 7 Edema – peripheral 15 26 7 12 Diarrhea 11 19 9 16 Pyrexia 9 16 11 19 Cough 3 5 12 21 Dyspnea 10 18 5 9 Epistaxis 8 14 7 12 Anemia 11 19 3 5 16 |
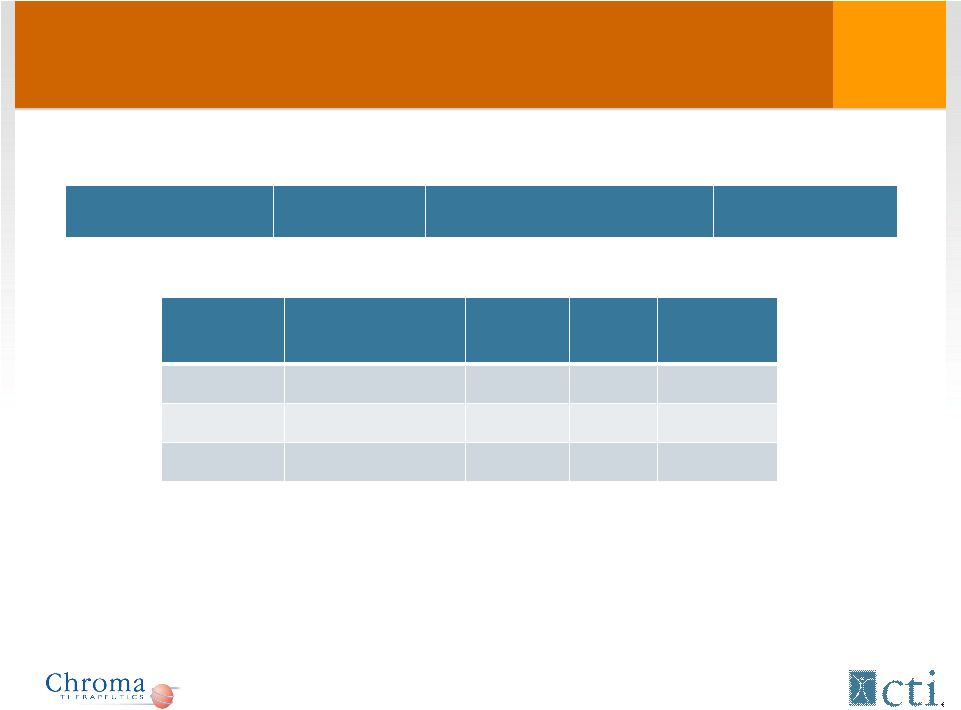 Phase I/II: Efficacy (response rates) Disease Total # patients CR PR SD AML 51 7* 7** MDS 4 1 1 Myeloma 2 1 Phase I/II AML ORR (CR+PR) 27.5% * 4 went into remission (duration 4-9 months); 1 cytogenetic response; 1 patient (0101) had a de facto CR (low volume BM tap) ** 1 PR was short 17 |
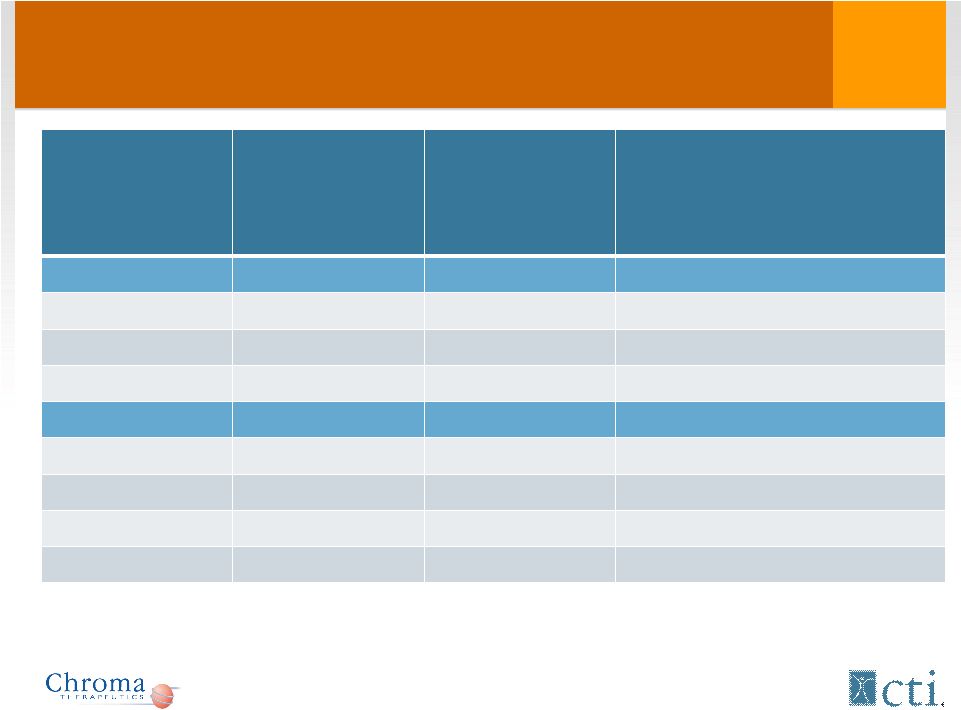 Overall Survival For AML Patients* NR – non responders *Overall survival data for 2 out of the 38 AML patients is not known **Overall survival data for 1 patient unknown JCO 28:4333-4338 Disease Number of patients Median Overall Survival from first treatment with Tosedostat to death, days (range) Phase I All patients AML 13 135 (40-420) • CR AML 4 334 (73-420) • NR AML 9 121 (40-402) Phase II All patients AML 38* 126 (8-696) • CR AML 3 272 (73-835) • PR AML 7** 170 (31-505) • NR AML 27** 100 (8-941) 18 |
 Phase I/II Study (002): Conclusions Well-tolerated mono-therapy • Oral once-a-day dosing • Transient reduction in platelets Encouraging phase I/II mono-therapy data • 27% response rate in elderly r/r AML • All responders >60yrs, 79% with r/r disease • Overall survival data encouraging Similar results in MDS warrant separate studies with hypo-methylating agents 19 |
 Tosedostat Clinical Program Blood Related Cancers - AML Randomized Controlled Trials 20 |
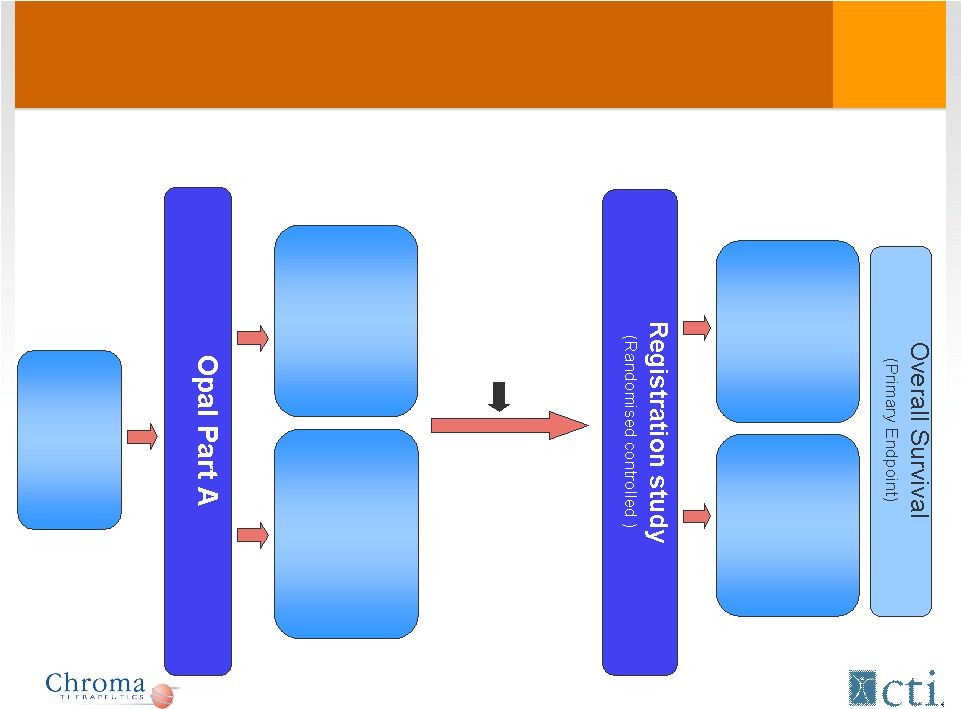 Proposed Pivotal Study Design Tosedostat + Ara-C 6 months Ara-C 6 months Relapsed/ refractory AML (>60yrs) 35 patients 120 mg 6 months 35 patients 240 mg (2 months) 120 mg (4 months) 120 mg 240/120 mg Complete: Q1 2011 Est. start: Q4 2011 Risk/benefit review, Dose selection |
 1 st Line AML (HOVON) Phase I Assessment of optimal induction dosing regimen Dose-finding Tosedostat + dauno/ara-C Phase II Optimum induction regimen and maintenance Optimal Tosedostat + dauno/ara-C regimen Tosedostat maintenance Standard dauno/ara-C regimen consolidation Arm A Arm B • Patients with AML, RAEB or RAEB-t, previously untreated, age >60 yrs, fit for standard chemo • N=~270 • Study open 22 |
 AML Competitive Landscape 7 candidates in Phase III mostly cytotoxics Elderly AML life-threatening disease with poor survival (~3 months) low tolerability to chemotherapy • clofarabine iv NDA and laromustine iv NDA: Rejected • Mylotarg® iv approval in relapsed AML: Withdrawn • decitabine iv: Failed primary O.S. endpoint in front-line AML • lintuzumab iv: Failed primary O.S. endpoint in front-line AML • sapcitibine : 1 st line +/-decitabine elderly AML • Flt-3 kinase inhibitor Oral, well-tolerated, chronic treatments improving survival addresses a major unmet medical need Tosedostat highly differentiated profile addressing unmet medical need 23 |
 Tosedostat Clinical Program Other Blood-Related Cancers Phase I/II 24 |
 Myelodysplastic Syndrome (MDS) Disease of elderly : • Lower-risk patients- Lenalidomide; • Higher-risk patients - HSCT (<10%)/ De-methylating agents • Age (>60yrs) – high-intensity therapies not generally offered Unmet medical need: low-intensity therapies with improved efficacy and toxicity profiles Tosedostat: ‘High risk MDS’ is attractive target Two MDS (RAEB) patients did very well in study 002 Two AML patients were ex-MDS (AHD) Phase I/II combination with decitabine planned 25 |
 Multiple Myeloma (MM) Strong biologic and preclinical rationale • Tosedostat inhibits myeloma cell proliferation and induces apoptosis in cell lines and primary patient cells, with minimal effects on bone marrow stroma (Faith Davies) • Tosedostat is synergistic with other anti-myeloma therapies (dexamethasone, bortezomib) in vitro. HDAC inhibitors being explored in MM • Potential synergy between Tosedostat and HDAC inhibitors 2 Planned phase I/II studies • Combination trial between Tosedostat and CHR-3996 (HDAC inhibitor) planned in MM (Royal Marsden) • Combination with bortezomib 26 |
 Tosedostat: Conclusion Tosedostat is a novel aminopeptidase inhibitor with tumor selective anti-proliferative activity Well-tolerated oral agent (once daily) Demonstrated synergy in combination with currently approved targeted therapies • Bortezomib, decitabine, azacytidine, HDAC inhibitors Potentially applicable across a variety of blood related cancers and select solid tumors Pivotal study (AML) estimated to begin in Q4-2011 27 |
 Summary Deal Terms & Structure CTI gets exclusive development and marketing rights in the Americas; Chroma retains ROW Cost-sharing • CTI bears 75% of agreed-upon direct development expenses • Trials aimed at US/EU regulatory approval Joint Governance (Development/Marketing Committees) • CTI lead party in America’s/Chroma lead in ROW Development budget capped at $50mm over 3 years • 75% CTI - 25% Chroma Initial Payments • $5mm upon execution • $5mm upon initiation of r/r AML phase III pivotal trial 28 |
 Summary Deal Terms & Structure Success-based Milestones • Scaled by number of potential patients in a given indication taking into account line of approval (1 st line Vs r/r) • Successful clinical trial results and NDA approvals • Blood-related cancers - AML, MDS, or Myeloma • Solid Tumor indication <10,000 pts/yr >50,000 pts/yr • Sales Thresholds Standard royalty rate scaled to increasing net sales 29 |
 Potential Market Opportunity Blood-related Cancers is large US market opportunity • AML ~12,000 pts/yr • MDS ~12,000 pts/yr • Myeloma ~26,000 pts/yr Estimated 2010 WW annual sales for drugs approved/used in AML/MDS or Myeloma • AML/MDS (clofarabine, decitabine, azacytidine) >$ 900 million • Myeloma (lenalidomide/bortezomib) >$2.4 billion 30 |
 Indication WW 2010 sales* Patent expiration MDS $200MM 2013 MDS $534MM 2011 Ped. ALL $103MM 2018 Multiple Myeloma/Mantle Cell $1.5B 2017 Multiple Myeloma/Deletion 5q MDS $2.5B 2019 Market Opportunity: Current agents VELCADE *Information from SEC filings. 31 |
 Summary Late-stage opportunity potentially addressing unmet medical needs in large markets • Potential 1 st oral non-cytotoxic treatment for AML • Synergistic activity with “blockbuster” products (Velcade®, Dacogen®, Vidaza®, Revlimid®) in MDS, myeloma • Potential rapid registration timeline- pivotal study to start Q4-2011 Good data visibility and news flow over next 18-24 months while phase III trial completes Development and sales synergies with pixantrone makes deal accretive at product launch Partnership enhanced by Chroma’s seasoned management and expertise 32 |