 Tosedostat Exclusive Marketing License and Co-Development Agreement Exhibit 99.3 |
 Forward looking statements This presentation contains forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. The forward-looking statements contained in this presentation include statements about future financial and operating results, and risks and uncertainties that could affect Cell Therapeutic Inc.’s (“CTI”) products under development. These statements are based on management’s current expectations and beliefs and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. These statements are not guarantees of future performance, involve certain risks, uncertainties and assumptions that are difficult to predict, and are based upon assumptions as to future events that may not prove accurate. Therefore, actual outcomes and results may differ materially from what is expressed herein. In any forward-looking statement in which CTI expresses an expectation or belief as to future results, such expectation or belief is expressed in good faith and believed to have a reasonable basis, but there can be no assurance that the statement or expectation or belief will result or be achieved or accomplished. The following factors, among others, could cause actual results to differ materially from those described in the forward-looking statements: risks associated with preclinical, clinical and sales and marketing developments in the biopharmaceutical industry in general and in particular including, without limitation, the potential failure of Tosedostat to prove safe and effective (including complete and overall response rates) for the treatment of blood related cancers and selected tumors as determined by the U.S. Food and Drug Administration (the “FDA”) or other regulatory agencies in the territories where CTI has exclusive rights, that the FDA may not accept the proposed clinical trial design of Tosedostat and/or may request additional clinical trials, that clinical trials may not demonstrate the safety and effectiveness of Tosedostat, that CTI cannot predict or guarantee the pace or geography of enrollment of clinical trials of Tosedostat, including whether or not the majority of the patients will be enrolled in the U.S., that CTI cannot predict or guarantee the outcome or results of clinical trials of Tosedostat, that a new drug application and/or marketing authorization application for Tosedostat may not be approved as early as 2013, that even if approved, CTI may not achieve the anticipated sales, that CTI cannot predict or guarantee whether the exclusive marketing license and co-development agreement with Chroma Therapeutics Ltd (the “Agreement”) will strengthen CTI’s business, financial condition, operating results and prospects or the trading price of CTI’s securities, that CTI cannot predict or guarantee whether milestones will be achieved pursuant to the Agreement, that the FDA may not approve pixantrone and/or Opaxio, CTI’s ability to continue to raise capital as needed to fund its operations, including the anticipated development costs for Tosedostat pursuant to the Agreement, determinations by regulatory, patent and administrative governmental authorities, competitive factors, technological developments, costs of developing, producing and selling CTI’s products under development; and other economic, business, competitive, and/or regulatory factors affecting CTI’s business generally, including those set forth in CTI’s filings with the U.S. Securities and Exchange Commission, including its Annual Report on Form 10-K for its most recent fiscal year and its most recent Quarterly Report on Form 10-Q, especially in the “Factors Affecting Our Operating Results” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” sections and its Current Reports on Form 8-K. Except as may be required by law, CTI does not intend to update or alter its forward-looking statements whether as a result of new information, future events, or otherwise. |
 Product/Deal Highlights Tosedostat: Novel tumor selective targeted therapy • Oral, once daily dosing, well tolerated Encouraging phase I/II data • 27% mono-therapy response rate elderly r/r AML, • Ongoing trials provide clinical news flow through 2012 Potentially rapid registration timeline: pivotal study estimated to start Q4-2011 Leverages CTI’s access to and expertise in blood-related cancer market • Potential development cost and sales synergies with pixantrone Exclusive rights in Americas on reasonable deal terms Targeting billion dollar market applications 3 |
 Tosedostat: Unlike current cytotoxic chemotherapy, Tosedostat does not interfere with genetic machinery of replicating cells • No hair loss, no damage to mouth and stomach lining, no depression of infection fighting white blood cells Tosedostat deprives cancer cells of the building blocks they need to make the proteins required for their survival Demonstrated synergy in combination with currently approved targeted therapies • Bortezomid, decitabine, azacytidine, HDAC inhibitors Potentially applicable across a variety of blood-related cancers • AML/MDS 24,000 pts/yr – Myeloma 26,000 pts/yr (US incidence) |
 Summary Deal Terms & Structure CTI gets exclusive development and marketing rights in the Americas; Chroma retains ROW Cost-sharing • CTI bears 75% of agreed-upon direct development expenses • Trials aimed at US/EU regulatory approval Development budget capped at $50mm over 3 years • 75% CTI - 25% Chroma Payments • $5mm upon execution • $5mm upon initiation of r/r AML phase III pivotal trial • Additional milestone payments based on development and sales milestones • Royalty on net sales |
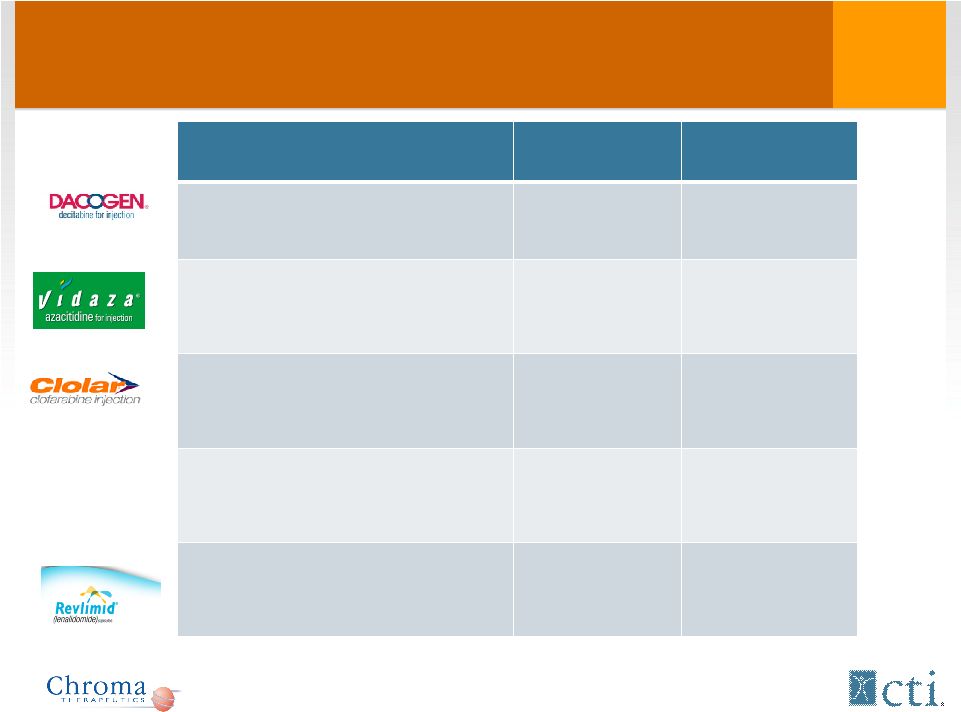 Indication WW 2010 sales* Patent expiration MDS $200MM 2013 MDS $534MM 2011 Ped. ALL $103MM 2018 Multiple Myeloma/Mantle Cell $1.5B 2017 Multiple Myeloma/Deletion 5q MDS $2.5B 2019 Market Opportunity: Current agents VELCADE *Information from SEC filings. |
 Summary Late-stage opportunity addressing significant unmet medical needs in large markets • Potential 1 st oral non-cytotoxic treatment for AML • Synergistic activity with “blockbuster” products (Velcade®, Dacogen®, Vidaza®, Revlimid®) in MDS, myeloma Diversifies CTI’s product portfolio with 3 potential product approvals by 2014 • Pixantrone 2011 – Tosedostat 2013 – Opaxio 2014 Modest ~$12mm increase to annual burn rate x3 years* Provides development and commercial synergies with pixantrone, which make deal accretive at product launch Targeting billion dollar market applications *Direct costs based on 3-year budget cap and cost share |