 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. The Myriad Transformation: Pioneering Personalized Medicine on a Global Scale 09/14/2015 1 Exhibit 99.1 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Forward Looking Statements 2 Some of the information presented here today may contain projections or other forward-looking statements regarding future events or the future financial performance of the Company. These statements are based on management’s current expectations and the actual events or results may differ materially and adversely from these expectations. We refer you to the documents the Company files from time to time with the Securities and Exchange Commission, specifically, the Company’s annual reports on Form 10-K, its quarterly reports on Form 10-Q, and its current reports on Form 8-K. These documents identify important risk factors that could cause the actual results to differ materially from those contained in the Company’s projections or forward-looking statements. |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Today’s Agenda 3 • The Myriad Transformation: Pioneering Personalized Medicine on a Global Scale – Mark Capone, CEO • Hereditary Cancer Leadership Now and in the Future – Alec Ford, President of Myriad Genetic Laboratories • Pioneering Companion Diagnostics for DNA Damaging Agents – Lloyd Sanders, General Manager Oncology • Autoimmune Market Represents Next Frontier in Personalized Medicine – Bernie Tobin, President of Crescendo Biosciences • Making Prolaris Standard of Care in Urology – Nicole Lambert, General Manager Urology • Break • Transforming Melanoma Diagnosis Through Pioneering Science – Vicki Fish, General Manager Dermatology • Industry Leading Pipeline to Ensure Growth Opportunities – Jerry Lanchbury, CSO • Expanding our Horizons in International Markets – Gary King, Executive Vice President of International Operations • Five Year Outlook: Increased Growth and Financial Leverage – Bryan Riggsbee, CFO • Q&A |
 4 Mark Capone Chief Executive Officer The Myriad Transformation: Pioneering Personalized Medicine on a Global Scale |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 20 The Myriad Transformation 5 years…. FIRST 1 product 1 country in 5 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. A trusted advisor transforming patients’ lives worldwide with pioneering molecular diagnostics 6 the future… 6 |
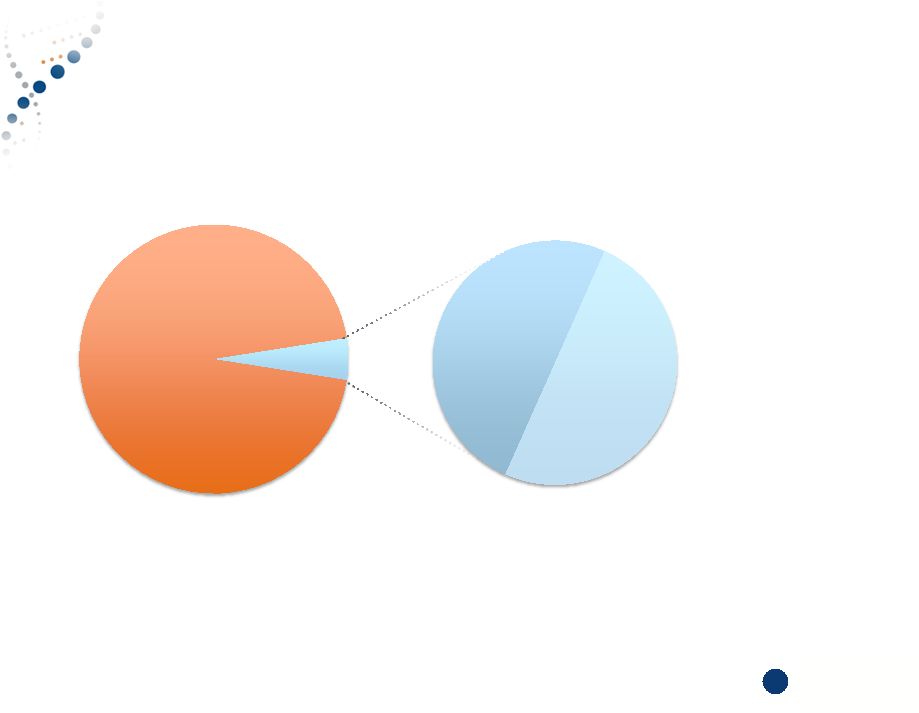 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. U.S. Healthcare System Remains Highly Inefficient 7 95% 5% 50% 50% 5% of patients represent 50% of healthcare costs* *Agency for Healthcare Research and Quality U.S. Population Healthcare Costs Drivers of Cost: • Focus on treatment not prevention • Late or incorrect diagnosis • Undifferentiated treatments • Trial and error approach to pharmaceuticals 7 |
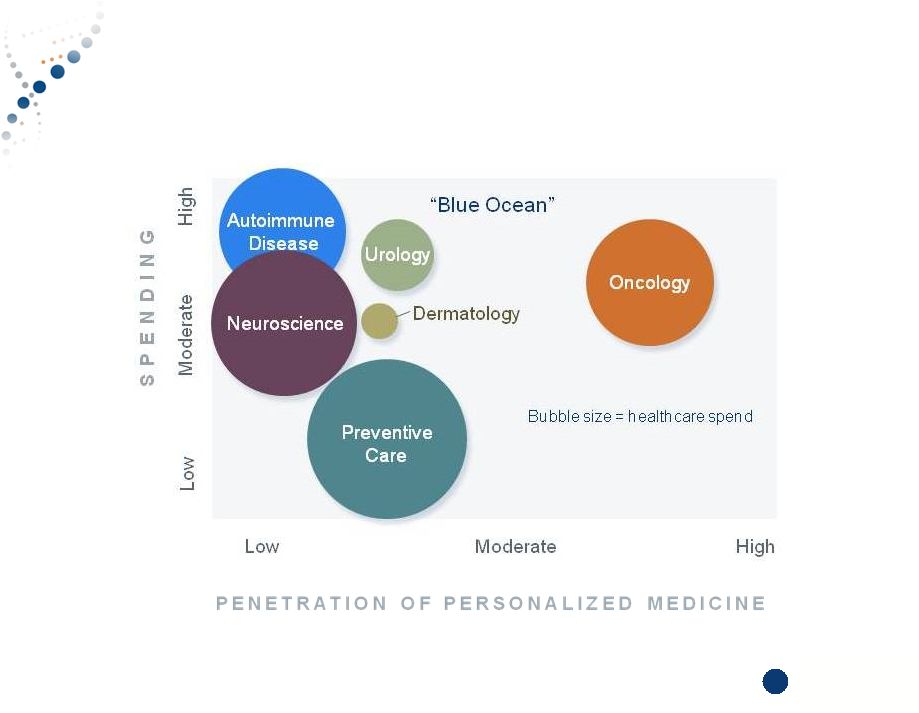 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Large Drivers of Inefficiencies Represent Blue Ocean Opportunities 8 8 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Myriad’s Mission Statement 9 9 9 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Unmatched Competitive Advantages in Personalized Medicine • Profitable R&D driven molecular diagnostic company • Expertise in DNA, RNA and proteins • Strong research capabilities: extensive collaborations (>50 institutions and >20 pharma/bio companies) • Broad regulatory experience (CLIA, FDA, CE mark) • Deep physician relationships (>90,000 ordering physicians since inception) • Extensive managed care contracts (>600) • Reputation for best-in-class quality for high- complexity tests (>2 million performed) 10 10 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Million >$50 Products with Revenue 7 Our Strategic Goals By 2020 11 Revenue Growth CAGR >10% Operating Margin >30% International Revenue >10% 11 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 12 12 * * * |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Our Strategic Imperatives to Achieve Our FIVE-Year Goals 13 Transition & Expand the Hereditary Cancer Market Increase International Contribution Diversify the Portfolio |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Significant Accomplishments Since Last Investor Day 14 STRATEGIC IMPERATIVE ACCOMPLISHMENTS Transition and Expand Hereditary Cancer Market • 6% hereditary cancer revenue CAGR since advent of competition • 80% of incoming samples ordered as myRisk™ Hereditary Cancer • Long-term pricing arrangements 45% of revenue • Expanded guidelines for colon & endometrial cancer (+75,000 patients per year) • Breast and pancreatic expansion studies underway and will be completed in FY16 (+90,000 patients per year) Diversify the Portfolio • Launched 7 new products (5 internal and 2 acquired) • Published and presented 250 studies • First FDA approved laboratory developed test (BRACAnalysis CDx™) • Obtained Medicare reimbursement for Prolaris ® • Completed 37 companion diagnostic deals Increase International Contribution • * • Launched first kit-based product (EndoPredict ® ) • Direct presence in 11 countries and distribution in 50 countries Grew international revenue by over 300%; exiting FY15 at 4% of revenue |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Our Strategic Imperatives to Achieve Our FIVE-Year Goals 15 Transition & Expand the Hereditary Cancer Market Increase International Contribution Diversify the Portfolio |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Our Strategic Imperatives to Achieve Our FIVE-Year Goals 16 Transition & Expand the Hereditary Cancer Market Increase International Contribution Diversify the Portfolio |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Hereditary Cancer Market Has Been More Durable Than Investors Anticipated 17 Revenue has grown at a 6% CAGR since FY13 • Medicare price reduction • Higher Medicaid mix • Small private payer reductions Share loss concentrated in the academic/genetic segment |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Future Landscape for Hereditary Cancer Market 18 Rapid transition to panels Panels are standard of care with minimal gene additions Used primarily for breast cancer patients Broader guidelines and additional cancers CLIA regulated market FDA regulated market Public databases fraught with errors; Myriad has substantial variant classification advantage Oncology Preventive care Preventive care >> Oncology Oncology CDx first then reflex Marginal improvement in public databases; Myriad dramatically expands informatics advantages CURRENT STATE: FY20 STATE: |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. What are the Modeling Assumptions? 19 Market Growth? Pricing? Market Share? |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Sensitivity Analysis Predicts Continued Growth 20 Revenue in millions Monte Carlo Simulation Factor Assumptions Market Growth • 7%-15% growth rate Price • Price decline 0%-40% • Long-term contracts until FY18 followed by… • FDA regulation Market Share • Incremental share loss of 10% to 40% • Share increase with price decline |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Our Strategic Imperatives to Achieve Our FIVE-Year Goals 21 Transition & Expand the Hereditary Cancer Market Increase International Contribution Diversify the Portfolio |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Pipeline Represents Two-Thirds of Opportunities 22 Under Development Risk? Diagnosis? Prognosis? Therapy? Oncology Preventive Care Urology Dermatology Neuroscience Autoimmune Currently Marketed |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Industry Leading Pipeline Facilitates Long-Term Growth 23 $10B $8B $10B+ Stage 3 REIMBURSEMENT Stage 2 VALIDATION Stage 1 DISCOVERY • myRisk Hereditary Cancer • Prolaris ® • Vectra DA ® • EndoPredict ® • BRACAnalysis CDx™ 1 • Tumor BRACAnalysis CDx ® • myPath ® Melanoma • myPlan ® Lung Cancer • myChoice ™ HRD (Platinum) 2 • myChoice HRD ™ (PARP) 3 • myPlan ® Renal Cancer • myPath ® Bipolar • myPath ® Pancreatic Cancer • myPath ® Psoriatic Arthritis • myPath ® Prostate Cancer • myPath ® Endometriosis Total Addressable Market (TAM) 1 Ovarian Cancer, Breast Cancer, Pancreatic Cancer 2 Triple Negative Breast Cancer, HER2- Breast Cancer 3 Ovarian Cancer, Breast Cancer, Pancreatic Cancer, Metastatic Prostate Cancer |
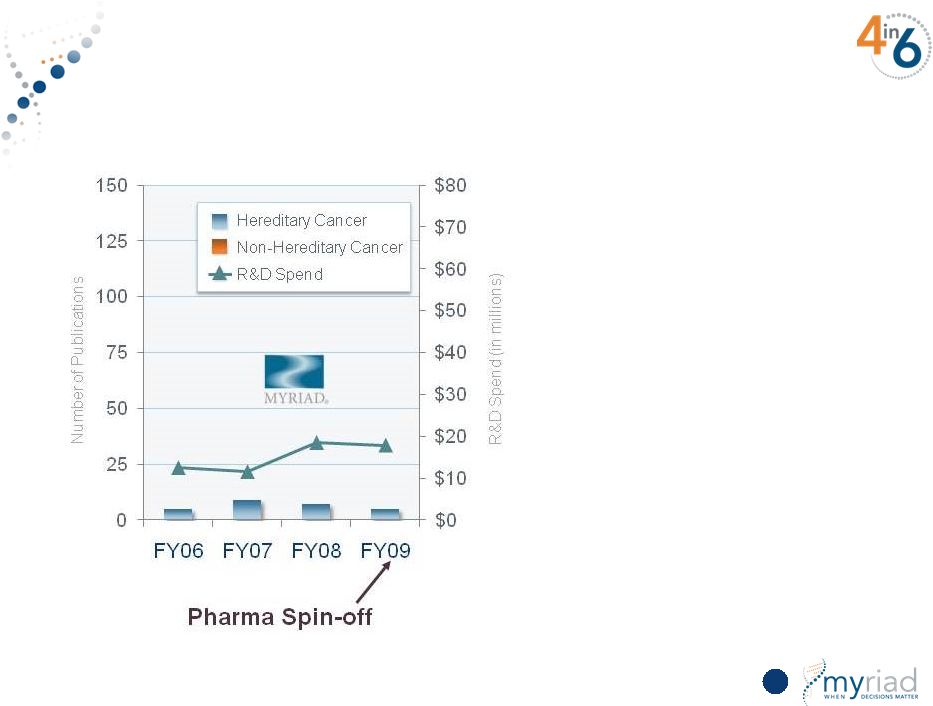 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Increasing Research Investment Yields Substantial Scientific Output 24 |
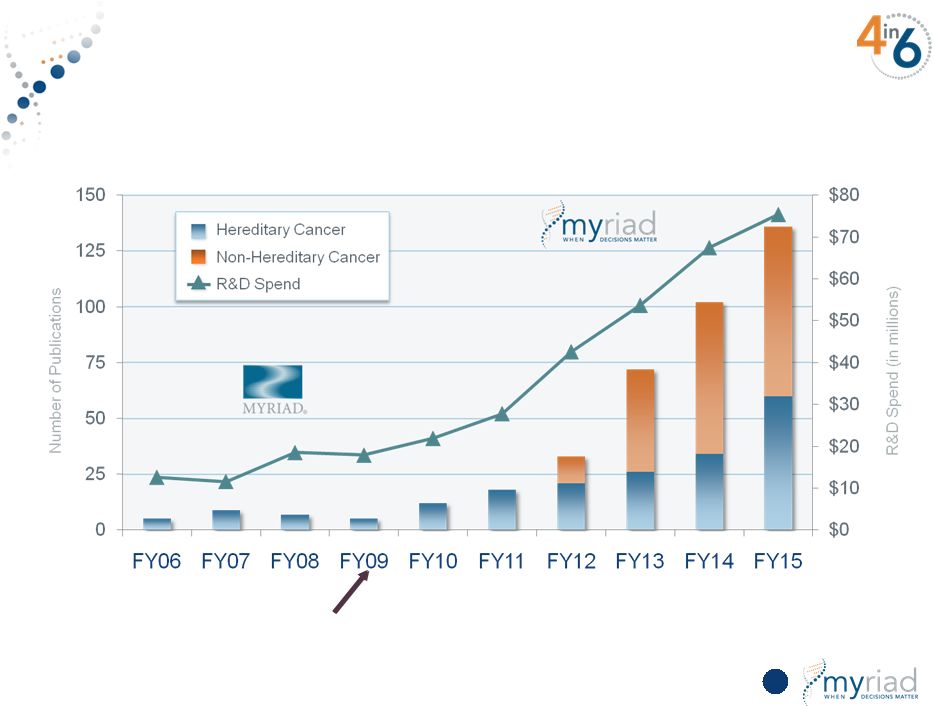 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Increasing Research Investment Yields Substantial Scientific Output Pharma Spin-off 25 |
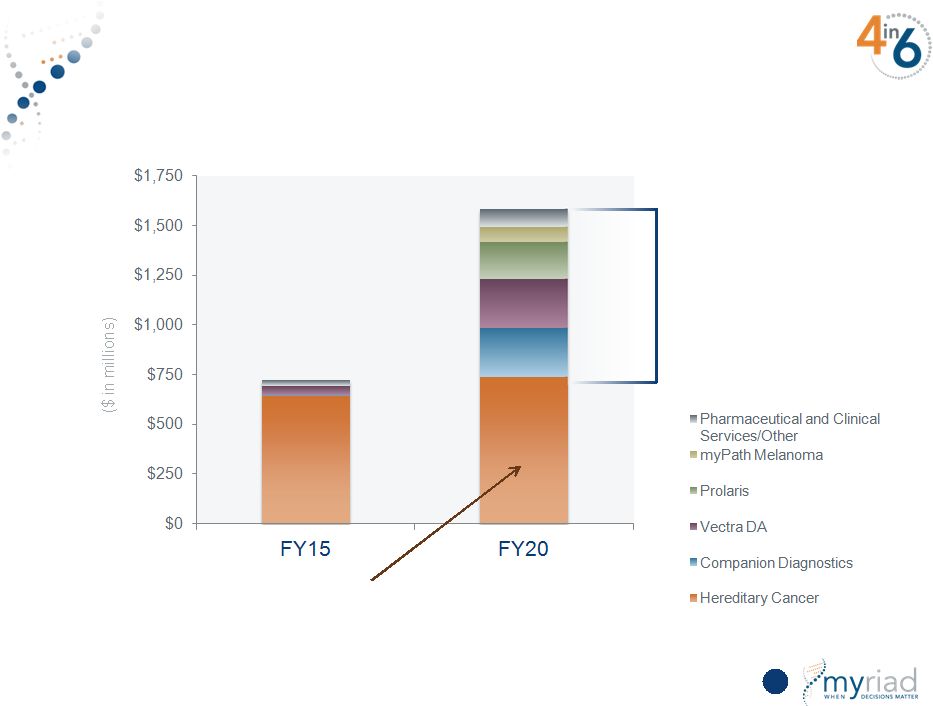 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. New Product Revenue >50% In FY20 26 Hereditary Cancer = 46% of Sales Stage 2 & 3 Products Only |
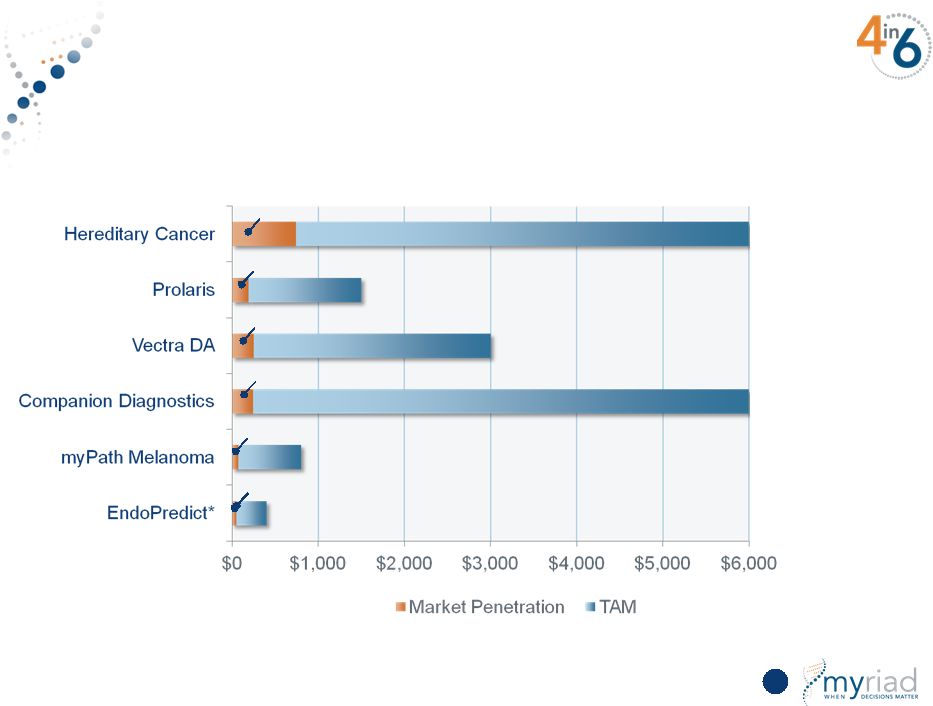 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Realistic Market Penetration Will Meet FY20 Goals 27 Global TAM $6.0B $1.5B $3.0B $6.0B $0.8B $0.4B 12% 13% 8% 4% 10% 13% Market Penetration in FY20 *Assumes only markets outside the United States |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Our Strategic Imperatives to Achieve Our FIVE-Year Goals 28 Transition & Expand the Hereditary Cancer Market Increase International Contribution Diversify the Portfolio |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Refined Strategy to Reflect Unique International Market 29 COUNTRIES Near-Term Growth: EU6 + Canada Long-Term Growth: Japan, China, and Brazil REFERENCE TESTS DNA (multiple platforms): Companion Diagnostics KITS RNA (platform partner): • EndoPredict • Prolaris • myPlan Lung • myPath Melanoma • myPlan Renal Protein (platform partner): • Vectra DA • myPath Bipolar • myPath Pancreatic |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Worldwide Leader in Personalized Medicine • We are entering the golden age for personalized medicine • We are pioneers of “research-driven” and “education-centric” business modeling for diagnostics • No company is better positioned to lead this revolution in healthcare than Myriad • Our finest hour will be discovered in the days ahead 30 |
 31 Alec Ford President, Myriad Genetic Laboratories Hereditary Cancer Leadership Now and into the Future |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Maintain Strong Market Leadership Position 32 Small impact from competition two years post SCOTUS decision Continued market leadership Clinical Accuracy: • Analytical accuracy • Interpretation accuracy • Regulatory capability Continued market leadership FUTURE DIFFERENTIATION Product Leadership: • Most clinically actionable panel • User friendly report Commercial Breadth: • Community physician education and support |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Growth has Continued Post-Competition 33 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Market Leader in Growing Areas of Hereditary Cancer Market 34 1% CAGR 20% CAGR (28%) CAGR >95% Market Share 50% Market Share |
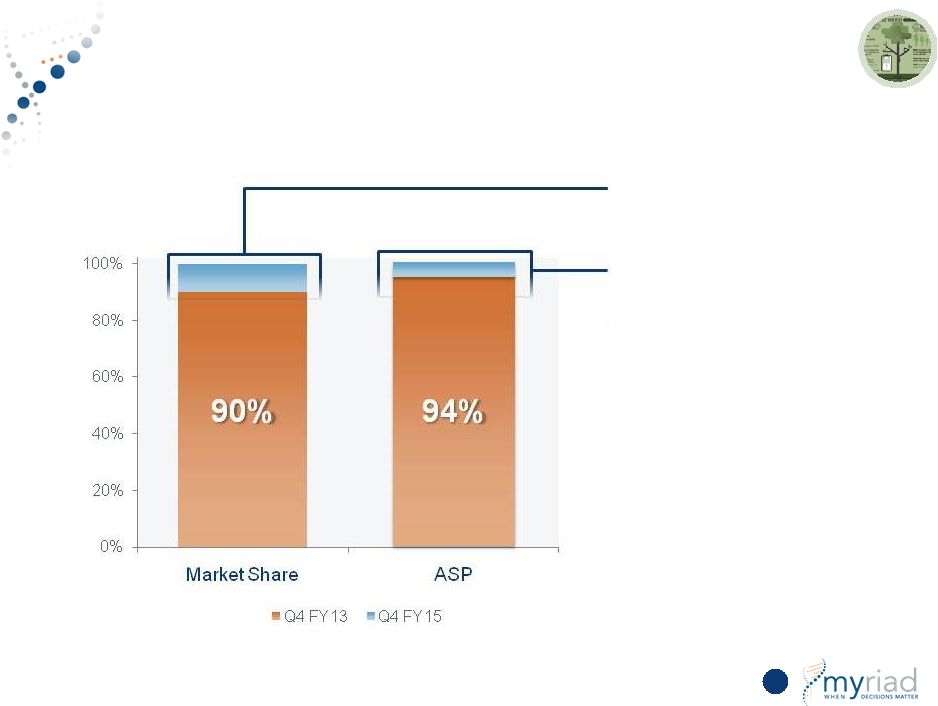
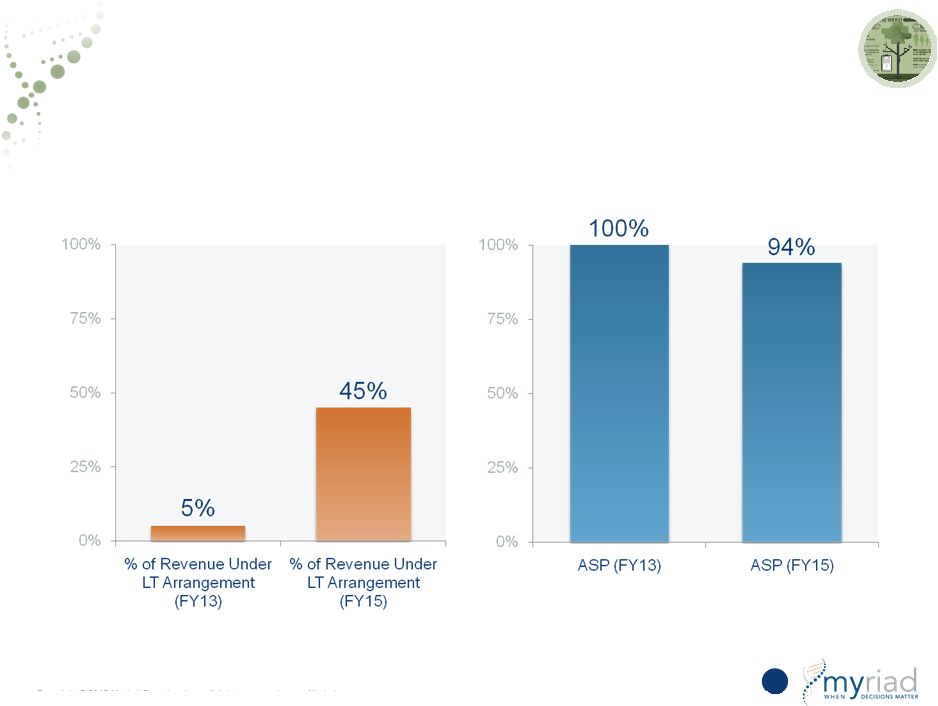 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Increased Pricing Visibility With Long-Term Pricing Arrangements 35 % of HC Business Under LT Contract Change in ASP FY13 to FY15 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Future Landscape for Hereditary Cancer Market 36 Rapid transition to panels Panels are standard of care with minimal gene additions Used primarily for breast cancer patients Broader guidelines and additional cancers CLIA regulated market FDA regulated market Public databases fraught with errors; Myriad has substantial variant classification advantage Oncology Preventive care Preventive care >> Oncology Oncology CDx first then reflex Marginal improvement in public databases; Myriad dramatically expands informatics advantages CURRENT STATE: FY20 STATE: |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Excellent Progress on myRisk Conversion and Market Expansion 37 Indication Added Market Potential Guidelines Colon @5% risk +$100M 40% All Endometrial +$150M 40% Breast <60 yrs +$150M FY16 FY17 All Pancreatic +$120M FY17 FY17 MyRisk Conversion Sep. 2013 Sep. 2014 Sep. 2015 Sep. 2016 0% 50% 80% Complete Market Expansion >$500M Contracting |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Differentiated Value in Hereditary Cancer 38 Clinical Accuracy Commercial Breadth Clinical Accuracy Product Leadership |
       Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 100% Analytical Accuracy Requires Tremendous Investment POST AMPLIFICATION PROCESS DNA EXTRACTION AND BATCH BUILD PROCESS TARGETED GENE SELECTION 39 • 85,000 base pairs that need to be 100% correct • 0% of samples meet quality threshold after first run on NGS platform • 23 major pieces of equipment from 10 vendors • 856 distinct steps required in testing process • 100 proprietary software applications |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Interpretation Accuracy Impossible With Public Databases 40 24,650 sequentially tested patients at MYGN 2,017 unique variants 34% of variants were not present in any of the five major public databases 3%-14% conflicting classification rate within individual public databases 3% concordance with deleterious variants in all five databases VAIL STUDY |
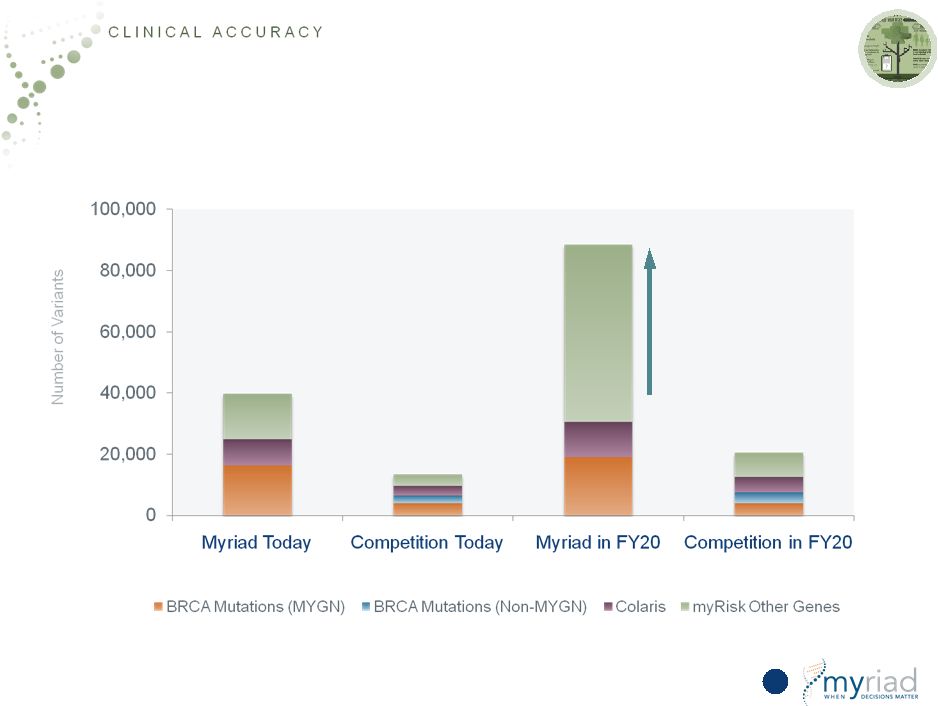 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Expanding Source of Competitive Advantage 41 Size of database more than doubles again Number of unique variants has almost doubled in last two years Informatics Advantage Expands Over the Next Five Years 2x 4.5x |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Economics of Inaccuracy: Potential Cost >$100K Case Study: 42 MYRIAD RESULT: Favors polymorphism based upon Pheno ® (MYGN method validated to be >99.5% accurate) Unnecessary Procedures Breast MRIs: $33,950 Mastectomy: $61,573 Bilateral salpingo-oophorectomy: $6,300 VARIANT BRCA2 C.9006A>T LAB 1 LAB 2 SCENARIO ONE Negative RESULTS: SCENARIO TWO POSITIVE RESULTS: COMPETITIVE LAB: Likely pathogenic based upon no evidence (ClinVar entry) |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Analytical & Interpretation Standards Increase With Additional Regulation 43 MYGN Requirements for BRACAnalysis CDx FDA Approval Analytical Validation >4,500 pages submitted to FDA Clinical Validation 9 major studies consisting of >6,000 patients Quality Systems 1,000 standard operating procedures Informatics 100 software applications with 50,000 work hours of validation for FDA FDA Experience First ever laboratory developed test FDA approved; planning multiple IDE submissions |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Differentiated Value in Hereditary Cancer 44 Clinical Accuracy Commercial Breadth Product Leadership Product Leadership |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Product Leadership Through the Most Clinically Actionable Panel 45 19 of 25 genes in NCCN guidelines Prevalence • Prevalence of actionable mutations in the tested population of 1 in 200 patients per gene Clinically Actionable • Four or more peer reviewed publications • Published data on medical management changes Penetrance • 2- to 3-fold risk vs. general population • Absolute cancer risk >5% |
    Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 46 >50% of Patients Missed With Single Syndrome Testing 41% - Other breast cancer genes 10% - Genes historically not associated with breast cancer 49% - BRCA1 and BRCA2 50% of Patients Missed Data based upon 28,000 patients |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Clear, Accurate and Trusted Reports Actionable to Physician and Patient • Clear plan for each patient based upon personal and family history and genetic testing results • Proprietary informatics power report; 420,000 work hours required to develop 47 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Differentiated Value in Hereditary Cancer 48 Clinical Accuracy Product Leadership Commercial Breadth |
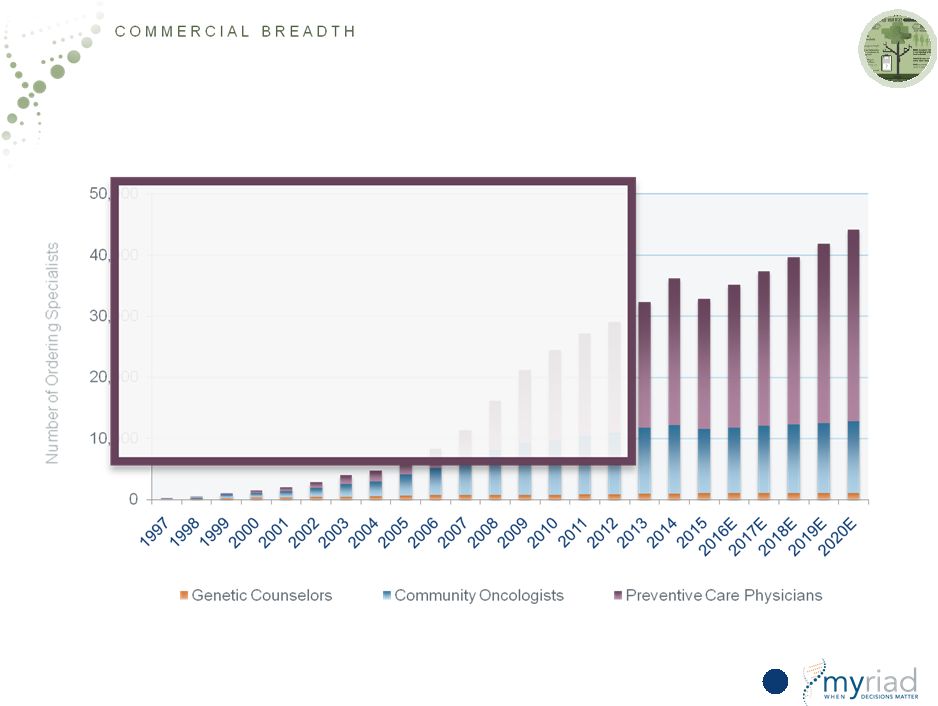 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 100% of Future Growth Derived From Community Physicians • Market increasingly moving to community setting • Community physicians need: — Substantial education and sales support — Extensive customer service and billing — Clinical support team — Only clinically actionable material — Easy to interpret report 49 |
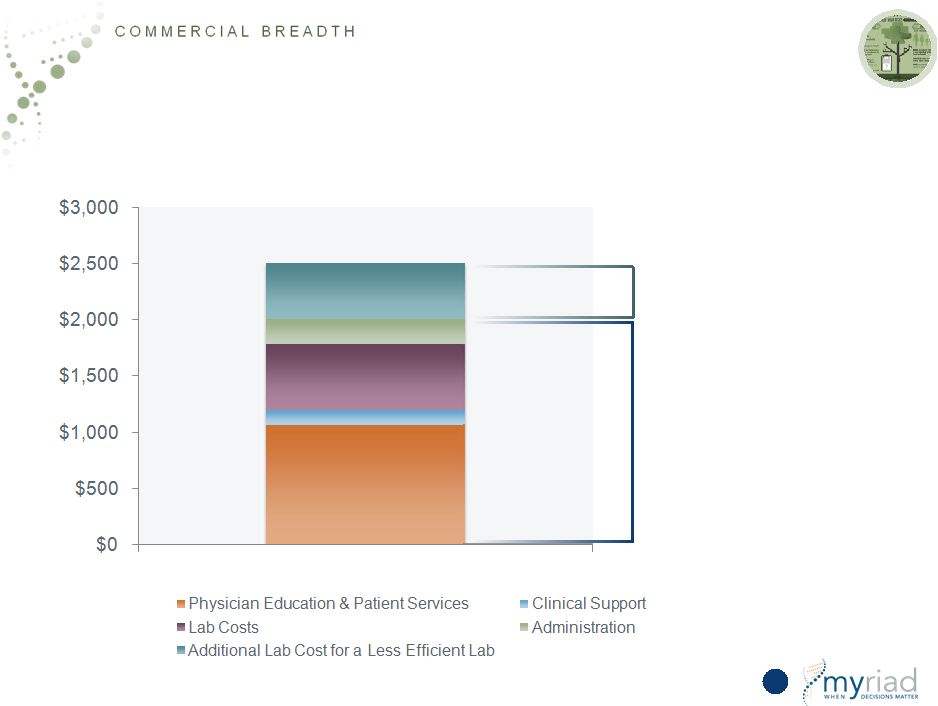 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Expenses Necessary for Community Market Total Cost >$2,500 per test True cost of providing a high-quality test in the community segment for the most efficient provider Additional cost for a less efficient, lower volume lab 50 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Maintain Strong Market Leadership Position 51 Small impact from competition two years post SCOTUS decision Clinical Accuracy: • Analytical accuracy • Interpretation accuracy • Regulatory capability Continued market leadership Product Leadership: • Most clinically actionable panel • User-friendly report Commercial Breadth: • Community physician education and support |
 52 Lloyd Sanders General Manager of Oncology Pioneering Companion Diagnostics for DNA Damaging Agents |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Pioneering Companion Diagnostics for DNA Damaging Agents 53 $6B Global Market Opportunity • FY16-FY20 Positioned to be the market leader in CDx for DNA damaging agents Key Advantages in CDx Market • Reimbursement • Adoption Curve • Barriers to Entry • Co-Promotion Proven Capability • BRACAnalysis CDx in Ovarian Cancer Pioneering Discoveries • myChoice HRD |
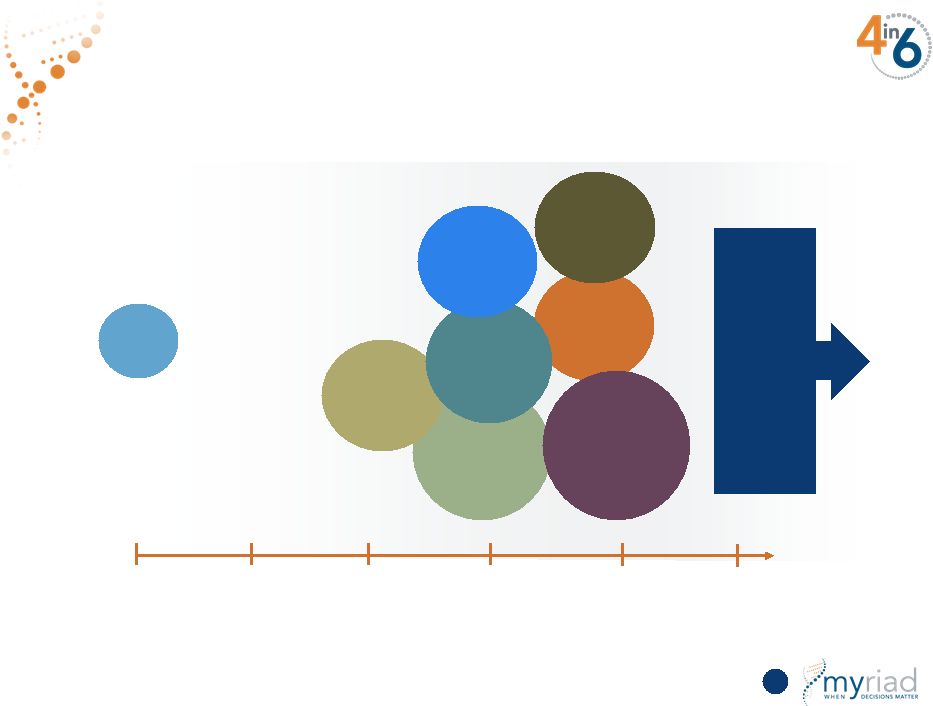 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. $6B Global Market Developing Over Next 5 Years Global Market = 1.4M patients or $6.0b* Neoadjuvant BC (143,000) Neoadjuvant BC (143,000) Pancreatic (102,000) Pancreatic (102,000) Ovarian (58,000) Ovarian (58,000) Platinum in TNBC (95,000) Platinum in TNBC (95,000) Platinum in Her2-BC (120,000) Platinum in Her2-BC (120,000) Metastatic BC (107,000) Metastatic BC (107,000) Metastatic Prostate (80,000) Metastatic Prostate (80,000) Adjuvant BC (194,000) Adjuvant BC (194,000) Colon (278,000) Gastric (114,000) Head & Neck (50,000) Colon (278,000) Gastric (114,000) Head & Neck (50,000) FY15 FY16 FY17 FY18 FY19 FY20 54 *Includes U.S., Canada and EU6 |
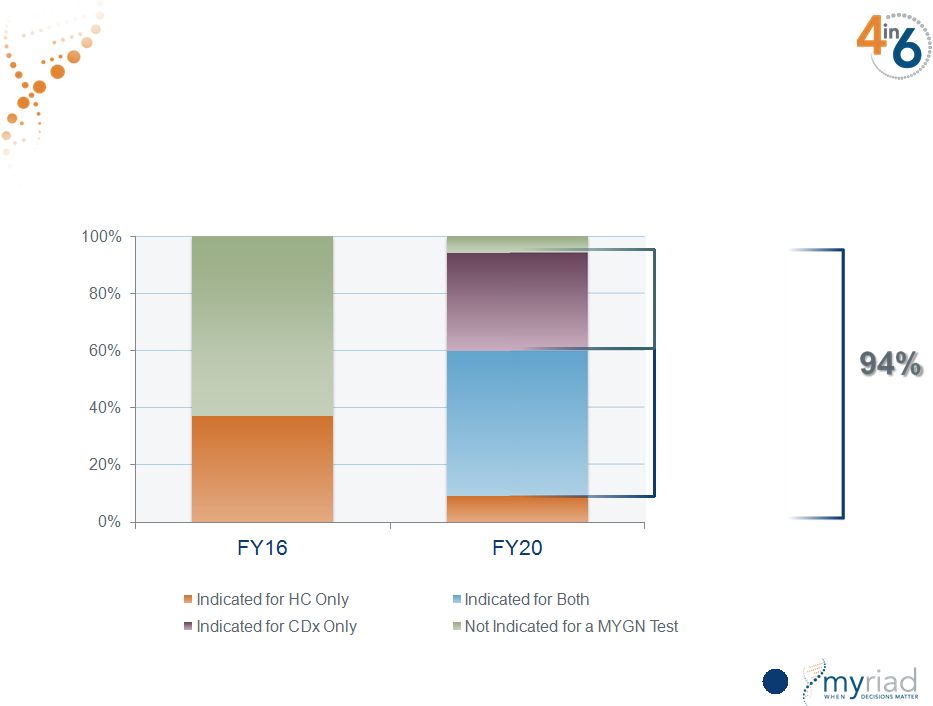 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Uniquely Positioned To Provide Comprehensive Testing 55 37% Breast Cancer Example Indicated for both HC and CDx Indication Indicated for a Companion Diagnostic |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Companion Diagnostic Market Dynamics Offer Advantages Barrier to Market Entry • FDA approval demonstrates high quality; supports FDA test utilization Speed to Market • Reimbursement is very quick following FDA approval • Increased promotional activity – pharma partner • Adoption curve is “pharmaceutical-like” vs. traditional diagnostic 56 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Unique Core Competencies Provide Sustainable Advantage 57 MYGN Unique Capabilities Intellectual Property Proven Regulatory Capabilities Global Distribution Complete Portfolio of Products Key Partnerships in Pharma & Biotech Proprietary Informatics & Database |
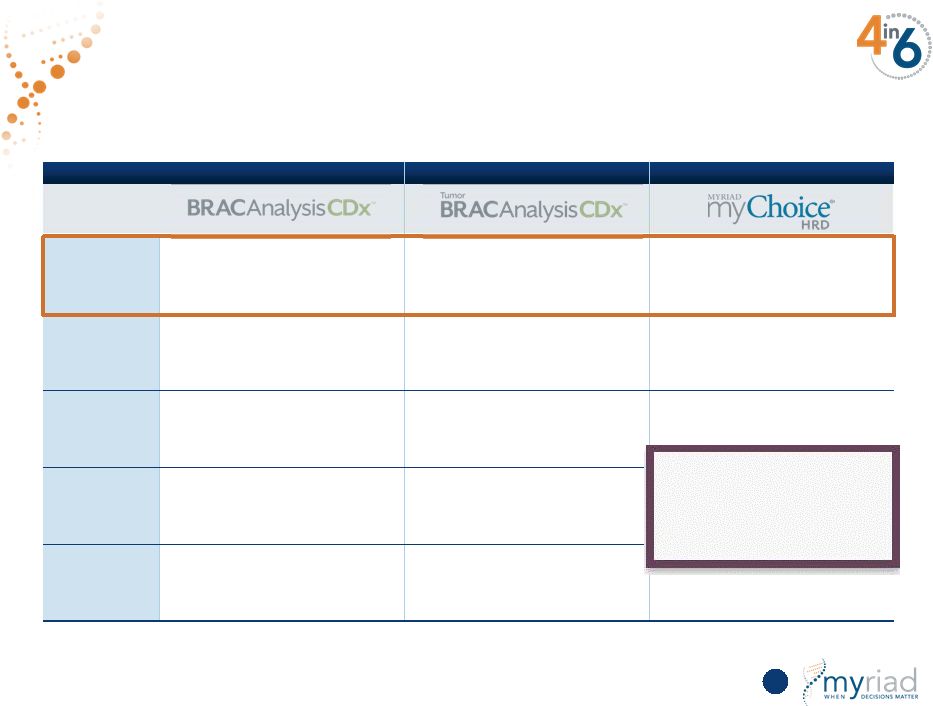 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. % Positive (Ovarian) 15% 22% 48% Sample Blood Tumor Tumor Biomarkers BRCA1&2 Tumor BRCA1&2 Genome-wide assessment of DNA scar associated Intellectual Property Database, process, bioinformatics Database, process, bioinformatics Currently Marketed FDA approved Yes, marketed in Europe only platinum drugs planned in Fall CY16 Only Company With a Complete Suite of Products 58 MYGN has IP on three proprietary technologies (LOH, TAI, & LST) |
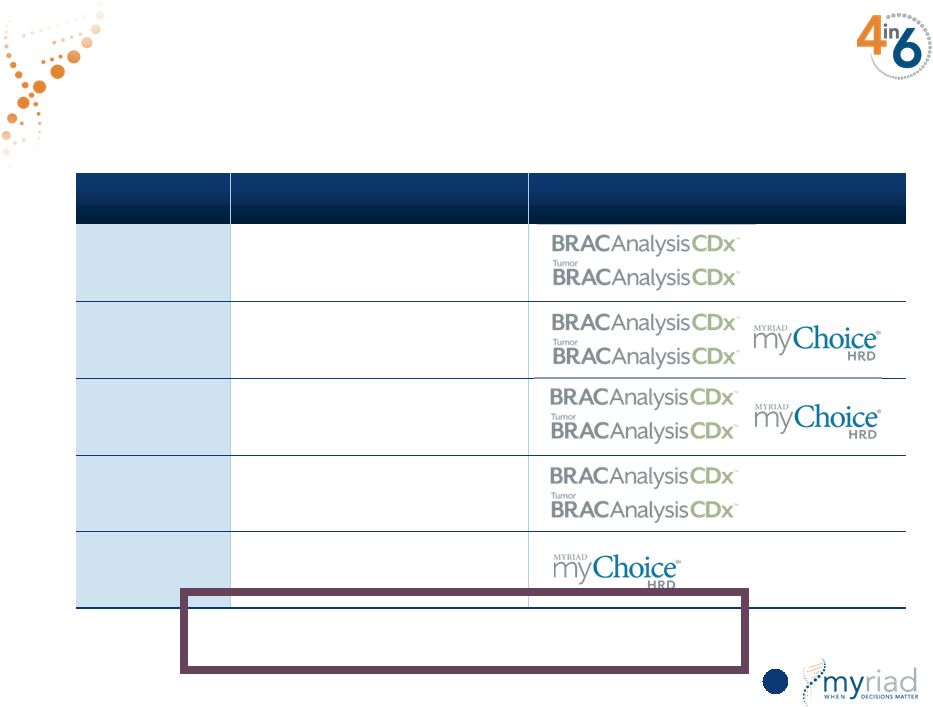       Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Extensive Collaborations With >22 Clinical Studies 59 Partner Indications Tests AstraZeneca (Lynparza™) Ovarian, Metastatic BC, Neoadjuvant BC, Adjuvant BC, Pancreatic, Prostate, Gastric Medivation (talazoparib) Metastatic BC, Pancreatic TESARO (niraparib) Ovarian, Metastatic BC AbbVie (veliparib) Metastatic BC, Ovarian Platinum Drugs Ovarian, TNBC, HER2- BC More than 22 clinical studies underway at key academic centers |
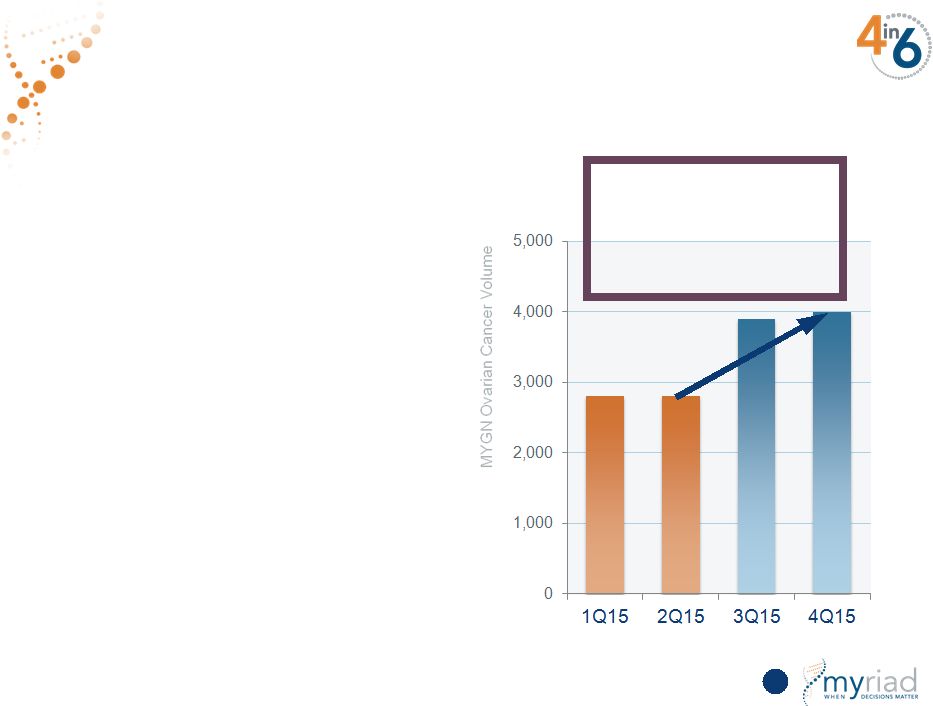       Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Highly Successful Launch of BRACAnalysis CDx in Ovarian Cancer • FDA approval Dec. 19, 2014 for BRACAnalysis CDx as a companion diagnostic • AZN and MYGN sales forces co-promoting the test 60 > 40% growth in ovarian cancer volume since the launch of BRACAnalysis CDx 60 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 61 Pioneering Discoveries For Assessing Genomic Instability |
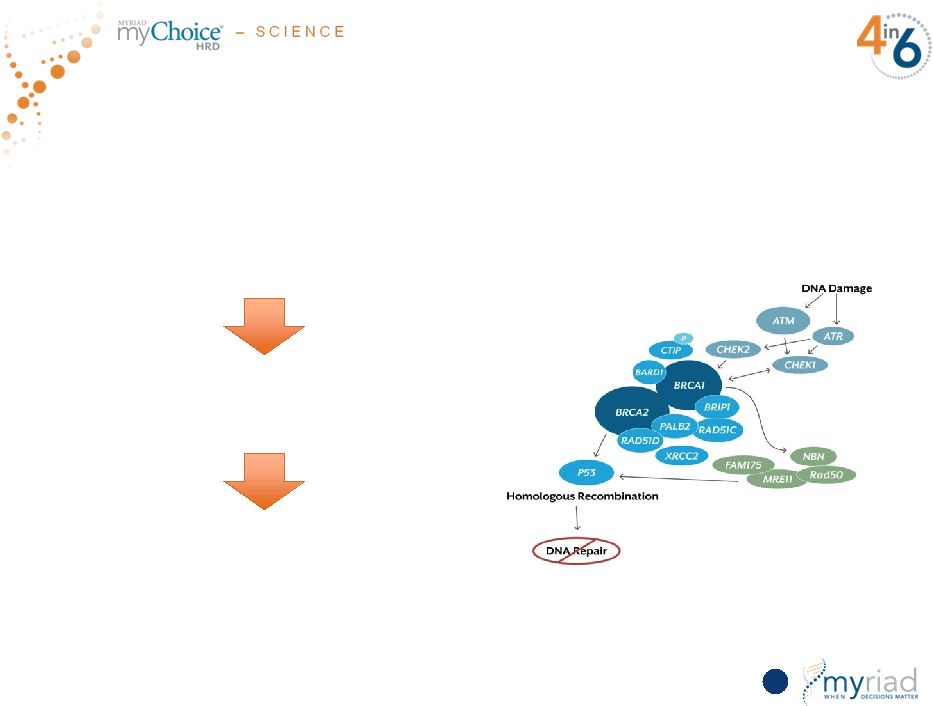 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Pioneering Discovery For Assessing Genomic Instability DNA Repair Pathway 54,000 snapshots of tumor DNA Three proprietary technologies (LOH, TAI, LST) Produces a quantifiable score: Each component is derived from an algorithmic calculation. The final score is the sum of the LOH+TAI+LST scores: (0-100) 62 |
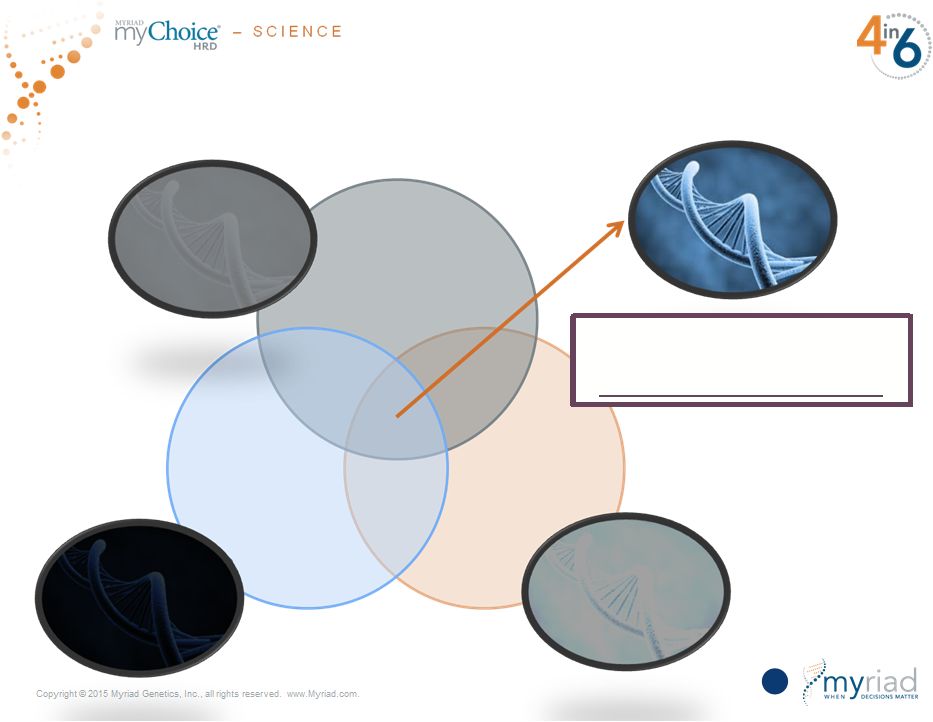 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Only the Combination of all Three Technologies Gives you a Complete Picture 63 Odds ratio of predicting pCR for myChoice HRD is 5.5 All 3 Technologies Required LST odds ratio = 4.7 TAI odds ratio = 2.8 LOH odds ratio = 3.3 |
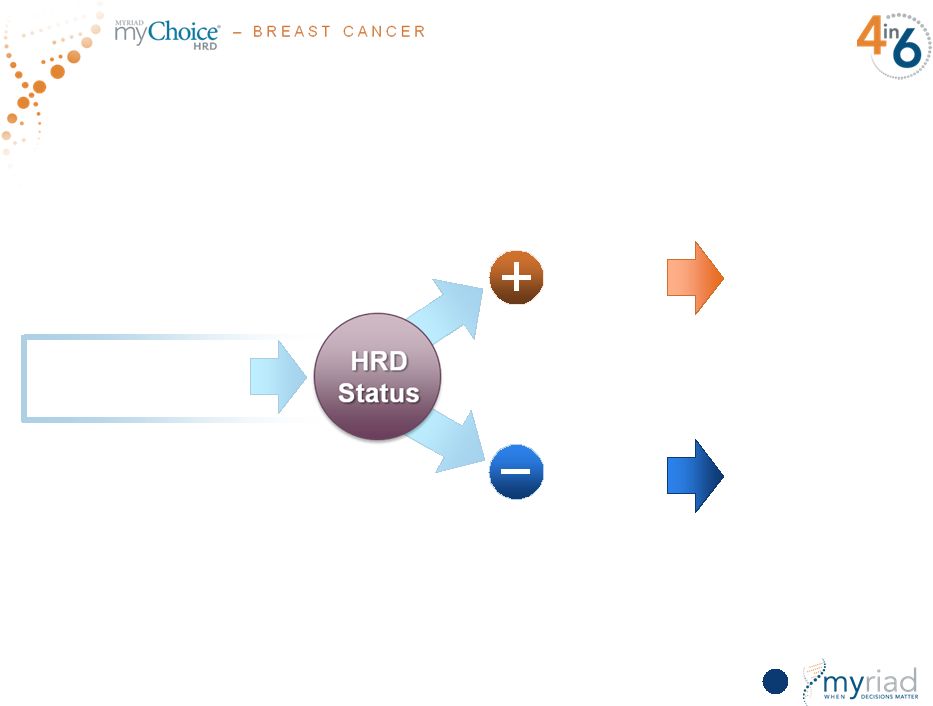 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. myChoice HRD TNBC Platinum Indication Represents $400 Million Global TAM 64 Treatment with Platinum Agents Treatment with Standard Chemo TNBC: 95,000 patients globally HRD NEGATIVE HRD POSITIVE |
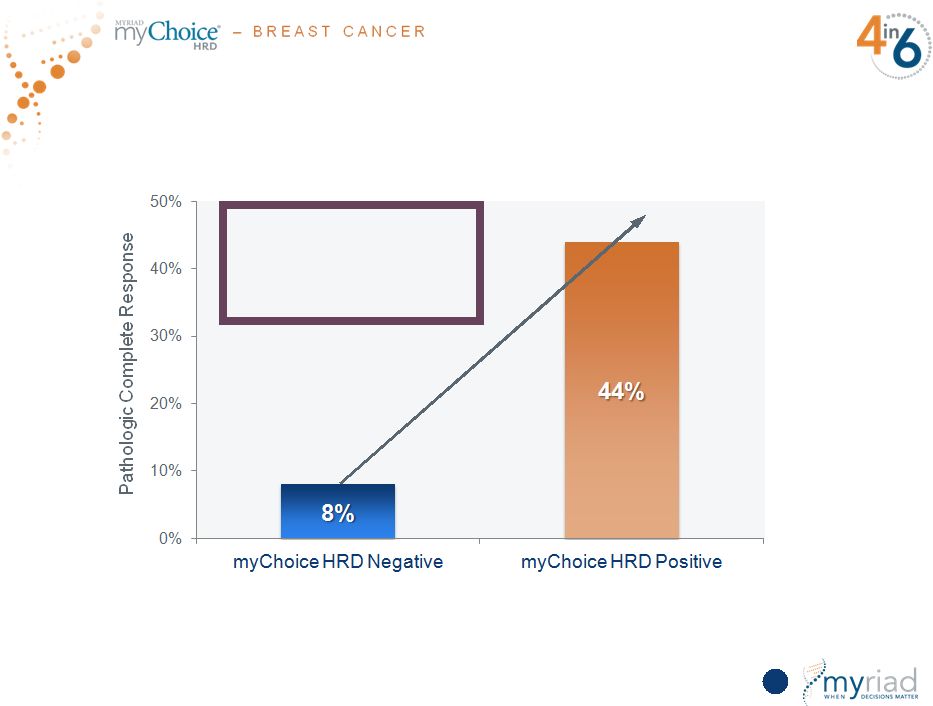     Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Five Studies Demonstrate myChoice HRD Clinical Utility in TNBC Based upon pooled analysis from five statistically significant studies comprising a total of 267 patients 65 Over 5-fold increase in patients with pathological complete response |
    Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Major Milestones Occurring in FY16 • Laboratory: – Completion of FDA laboratories for Tumor BRACAnalysis CDx and myChoice HRD • Regulatory: – IDE submissions for Tumor BRACAnalysis CDx and myChoice HRD – PMA submission for myChoice HRD • Clinical: – TESARO NOVA study results: myChoice HRD and niraparib – Final validation study completed for myChoice HRD for platinum in TNBC – Additional trials in new cancer indications with PARP inhibitors 66 Early access launch for myChoice HRD in Fall of 2015 |
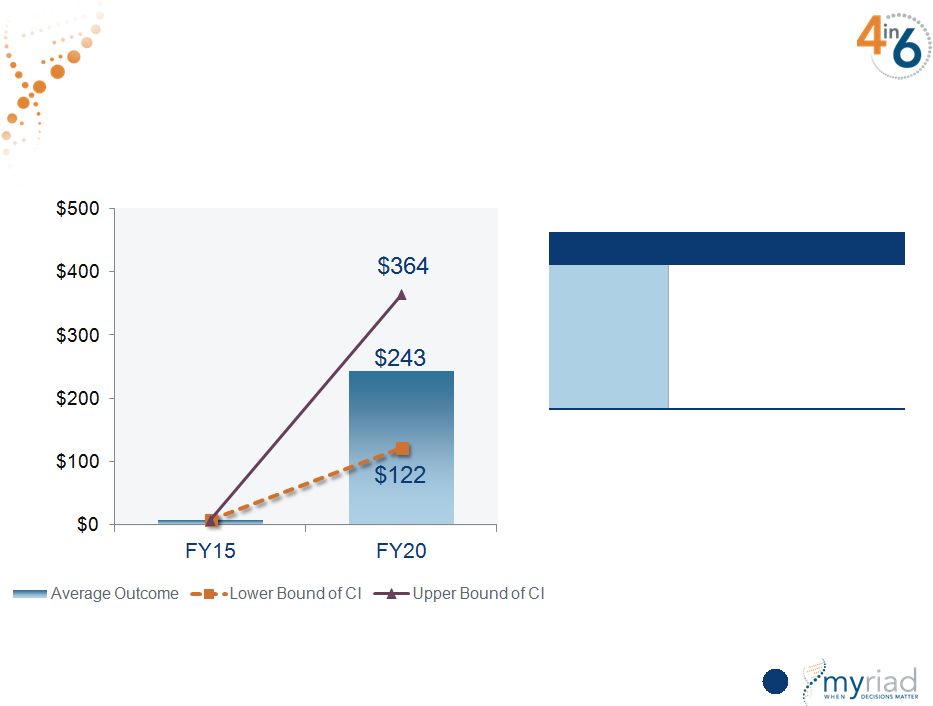 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Sensitivity Analysis Predicts Strong Growth 67 Revenue in millions Monte Carlo Simulation Factor Assumptions Growth • U.S. TAM between $500M and $1.5B • International mix between 5% and 20% • Market penetration between 15% and 25% |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Pioneering Companion Diagnostics for DNA Damaging Agents 68 $6B Global Market Opportunity • FY16-FY20 Positioned to be the market leader in CDx for DNA damaging agents Key Advantages in CDx Market • Reimbursement • Adoption Curve • Barriers to Entry • Co-Promotion Proven Capability • BRACAnalysis CDx in Ovarian Cancer Pioneering Discoveries • myChoice HRD |
 Bernie Tobin 69 President, Crescendo Bioscience Autoimmune Market Represents the Next Frontier in Personalized Medicine |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Autoimmune Market Is An Incredible Opportunity 70 Market Opportunity • Autoimmune market represents blue ocean • Medicare reimbursement • Solid plan for expanding private coverage Positioned for market success in autoimmune Key Advantages • Vectra DA validation • Highly predictive Commercial Breadth and Depth • Re-accelerating growth • Physician adoption • Improved logistics • Practice integration |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Substantial Opportunity in Blue Ocean Autoimmune Market • Vectra DA for rheumatoid arthritis is initial foray into autoimmune market • Additional segments of this market are equally compelling • Initiated discovery work on psoriatic arthritis 71 Juvenile Arthritis 600,000* Psoriatic Arthritis 2,500,000* Ankylosing Spondylitis 2,200,000* Rheumatoid Arthritis (current market) 3,000,000 patients* Companion Dx 15 current collaborations with major pharmaceutical companies *Prevalence numbers for U.S., Canada and EU6 markets only (Source: Datamonitor); |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Clinical Validity and Utility of Vectra DA Demonstrated In Numerous Published Studies 72 |
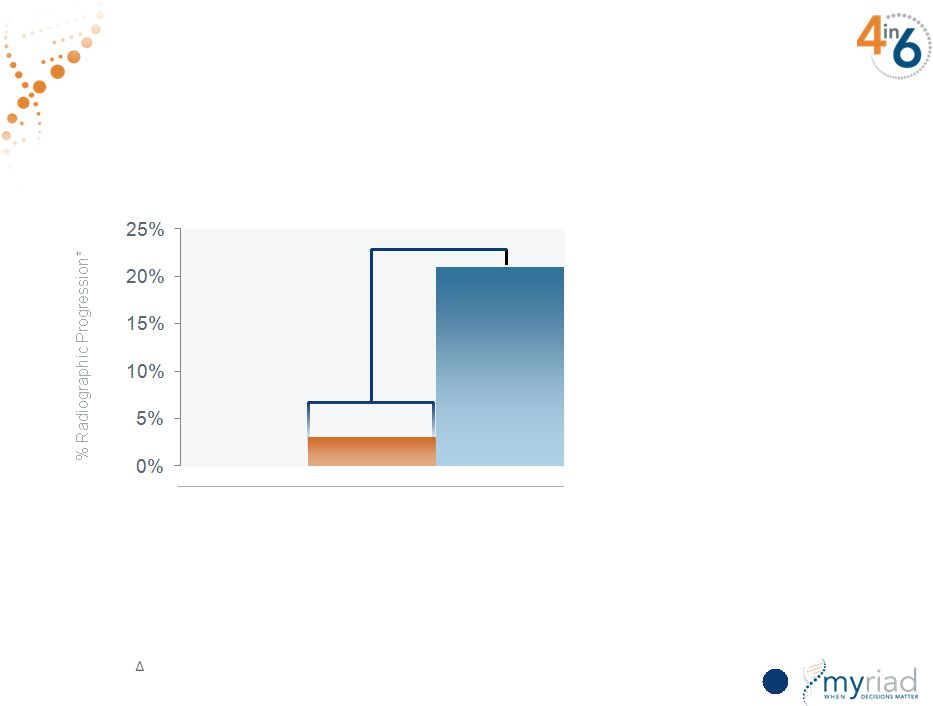 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 7x Relative Risk Low (<30) p = 0.008 Vectra DA Score at Baseline SWEFOT: Vectra DA Score Highly Predictive of Radiographic Progression • 235 patient SWEFOT study • Gold standard endpoint of radiographic progression at one year • Vectra score highly correlated to patient outcomes High (>44) Moderate (30-44) 73 Hambardzumyan, K, et al, Annals of the Rheumatic Diseases 2014. doi:10.1136/annrheumdis-2013-204986 *% Patients with SHS >5 BL to year 1 |
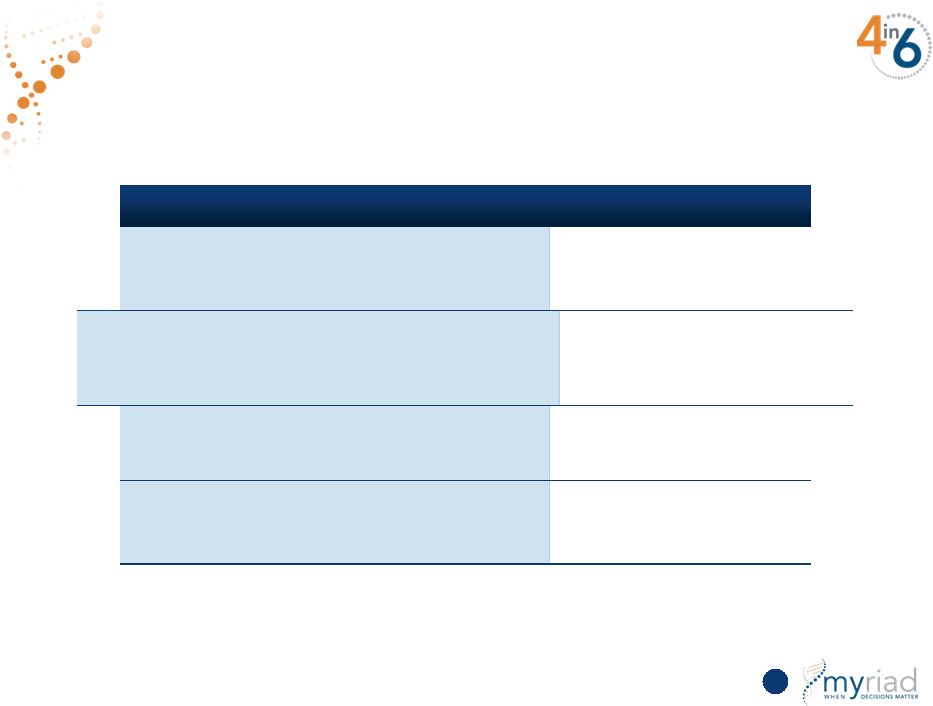 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Strategy to Reaccelerate Growth INITIATIVE PROGRESS TO DATE Clarity and focus on selling message Initiated in Q3 FY15 Increased focus on direct-to-patient marketing Initiated in Q1 FY16 Studies to provide additional data on clinical interpretation Completion in FY16 Driving depth and breadth in Medicare market Initiated in Q4 FY15 74 |
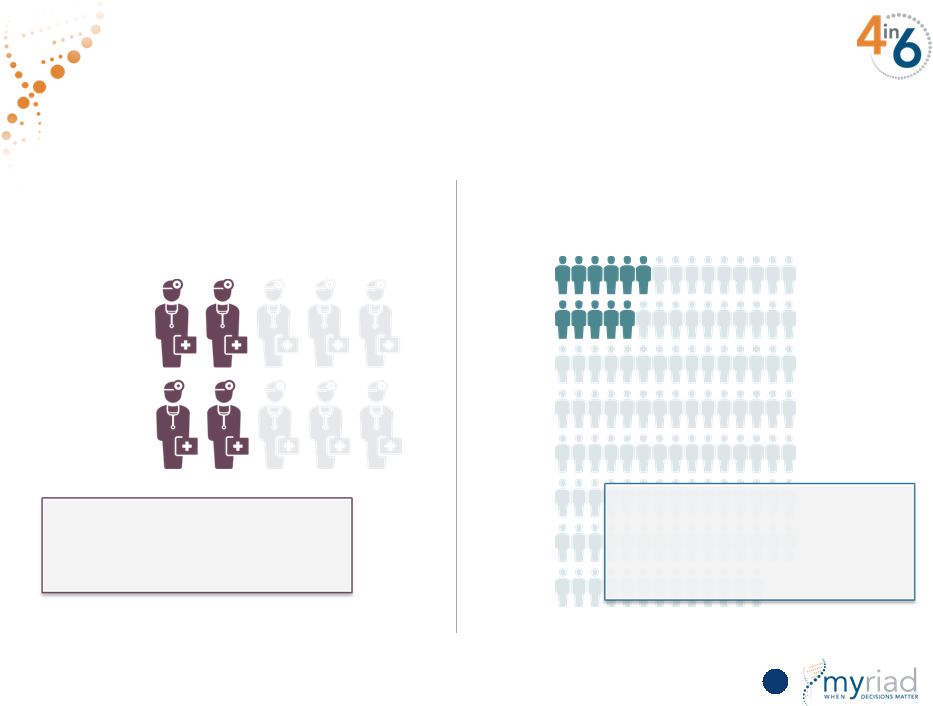 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Driving Depth and Breadth in Medicare Market 75 Breadth of Use Among Doctors Depth in the Patient Population Per Using Physician ~40% of rheumatologists use the test 11 patients tested per using physician per month (~9% of patients seen) |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Driving Breadth: Easier Logistics via National Lab Contract • We have contracted with LabCorp to use their national network of 1,700 Patient Service Centers (PSCs) to collect patient blood samples beginning Aug. 31, 2015 – 24% of the rheumatology market does not have in-office blood collection facilities – 28% of rheumatologists cite blood draw logistical issues as a major barrier to ordering more Vectra DA tests – 37% of rheumatologists said they would order more Vectra DA tests, if their patients could go to a LabCorp PSC 76 1,700 Patient Service Centers |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Driving Depth: Practice Integration Pilot • Multi-specialty clinic in North Carolina with 342 rheumatoid arthritis patients • Physician goal: minimize staff time on the phone, communicating lab results • 86% patient opt-in rate for PI, and 100% patient satisfaction rate • Quarterly increase of 37% in volume; 33% of incremental assays came from new patients 77 Case Study: Practice Integration PI a trial program at certain doctor’s offices to have test results prior to patient visit TEST RESULTS PATIENT VISIT Critical to expanding utilization in certain practices PHYSICIAN REVIEW |
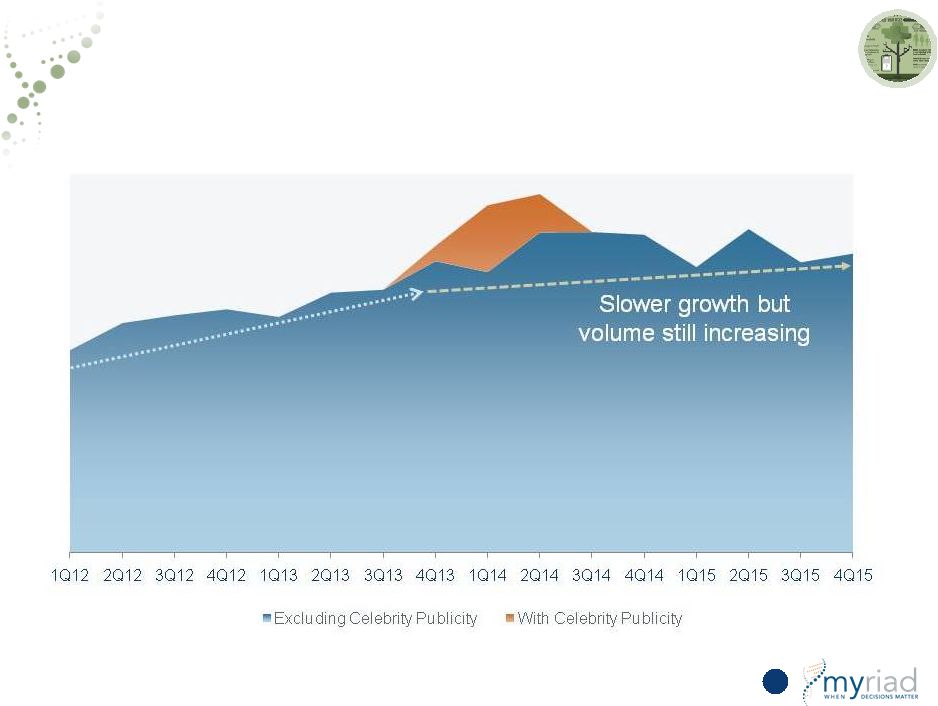
  Vectra DA Has Returned to Growth In the Last Two Quarters 78 12% sequential volume growth Prior to initiation of current strategy Post initiation of current strategy Tests in Thousands 33% CAGR Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
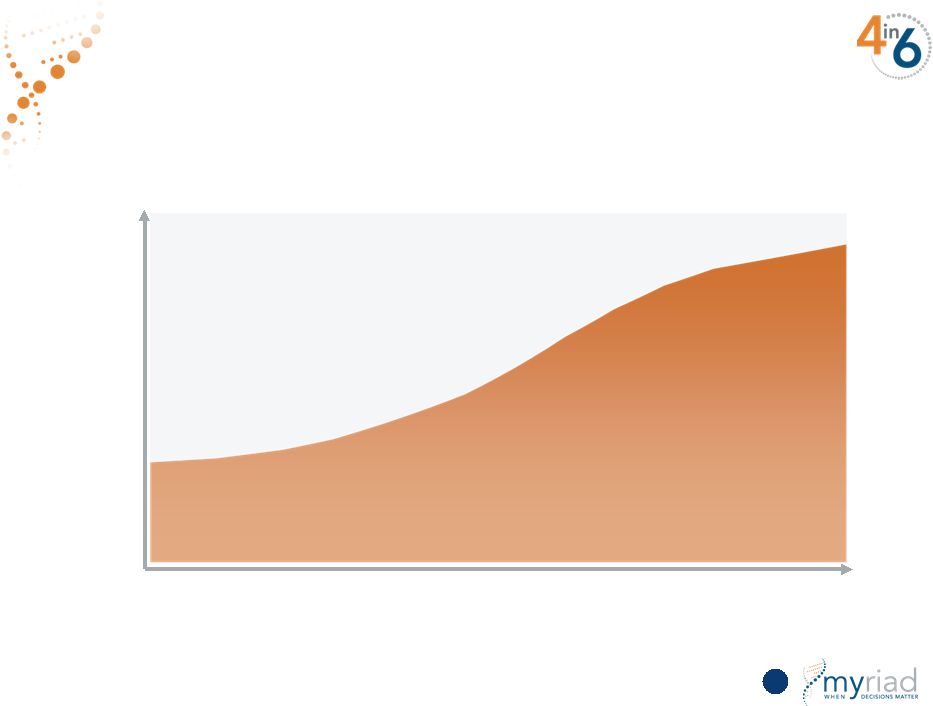 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Executing Plan to Expand Private Payer Coverage in the Future 79 Data Mining FY16 Retrospective Study FY17 Prospective Study FY18 % of private payer covered lives Private Payer Feedback Suggests More Clinical Utility Data Required Distribution of private payers’ data requests |
   Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Expanded Coverage Will Drive Significant Leverage 80 Current Medicare reimbursement level ~$570 Gross profit per test Gross profit per test COGS per test COGS per test Expanded private payer coverage Increased efficiencies |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Sensitivity Analysis Predicts Strong Growth 81 Revenue in millions Monte Carlo Simulation Factor Assumptions Growth • 15%-30% growth rate • 4%-10% market penetration Coverage • 80%-100% payer coverage |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Autoimmune Market Represents the Next Frontier in Personalized Medicine • Autoimmune market is an incredible opportunity • Vectra DA is an outstanding product, supported by robust science • Current Vectra DA reimbursement under Medicare supports over $600M in sales • Penetration of Medicare market and expansion of private payer coverage will be two key drivers of growth 82 |
 83 Nicole Lambert General Manager of Urology Making Prolaris Standard of Care in Urology |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Significant Unmet Need In Prostate Cancer Treatment 84 Market Opportunity • Substantial unmet clinical need • Expanded Medicare coverage based on additional clinical data Key Advantages • Pioneering science differentiation based on gold standard endpoints • Definitive active surveillance threshold • Unmatched clinical utility data Commercial Breadth and Depth • Largest urology sales force in diagnostics • Increased physician adoption • Increased test utilization Positioned to become standard of care in the prostate cancer prognostic market |
    Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Most Prostate Cancer Patients Inappropriately Treated 85 AUA Low-Risk Patients Overtreatment Problem AUA High-Risk Patients Undertreatment Problem TREATMENT PARADIGM RESULTS WITHOUT IMPROVED TOOLS Most low-risk patients have a low risk of prostate- specific mortality Yet the vast majority are treated upfront More than half will experience biochemical recurrence with single- modality treatment alone Yet the majority do not receive multi-modality treatment AUA Intermediate Risk Patients |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 86 Prolaris is Best Positioned to Solve This Need |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Prolaris Is the Only Test Validated Against Meaningful Clinical Endpoints 87 PUBLICATION SAMPLE TYPE PATIENTS OUTCOME Cuzick 2011 RP 353 HR = 1.89 per unit Prolaris score, p=5.6x10 -9 Cuzick 2011 (cohort 2) TURP 337 HR = 2.92, p=6.1x10 -22 Cuzick 2012 Biopsy 349 HR = 2.02, p=8.6x10 -10 Cooperberg 2013 RP 413 HR = 2.10, p=2.2x10 -6 Freeland 2013 Biopsy 141 HR = 2.55, p=0.0017 Bishoff 2014 Biopsy 582 HR = 1.6, p=2.4x10 -7 Bishoff 2014 Biopsy 582 HR = 5.35, p=2.1x10 -8 Cuzick 2015 Biopsy 757 HR = 2.32, p<10 -17 PROCEDE 500 Biopsy 305 Changed treatment plans 65% of the time 40% 25% PROCEDE 1,000 Biopsy 1,206 Changed treatment plans 48% of the time 35% 13% |
    Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Prolaris Has Substantially Stronger Prognostic Power Than Traditional Pathology 88 (p<1.5x10-4) (p<2.1x10-7) (p<3.7x10-15) Gleason PSA Prediction of Prostate Cancer Death of the prognostic power in the Prolaris CCR score is derived by the independent predictive power of the Prolaris test Prolaris >60% Predicting Gleason score is not enough |
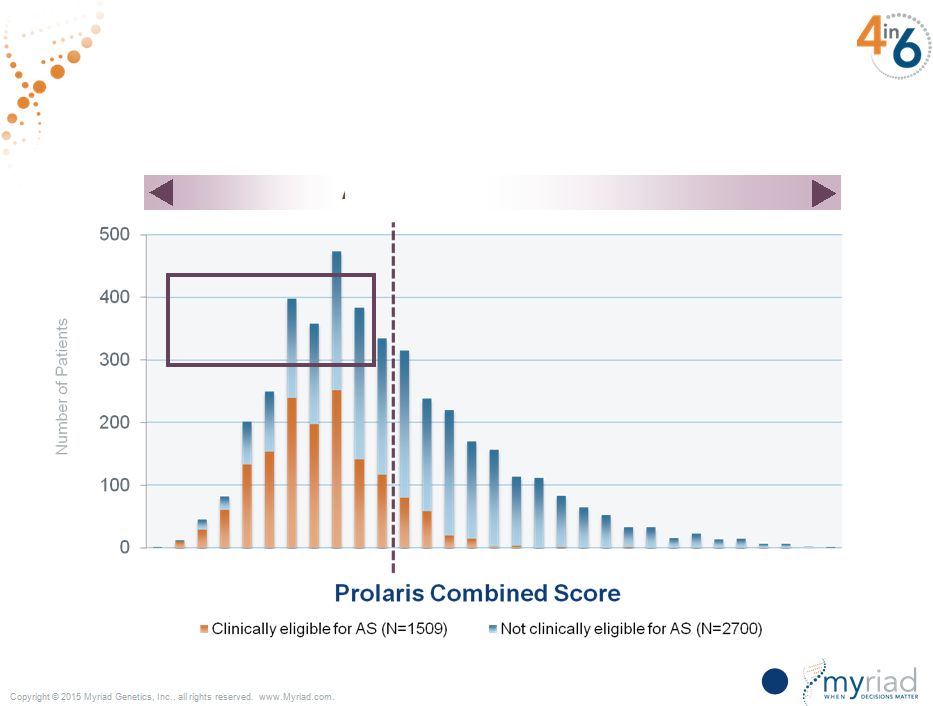       The Definitive Active Surveillance Threshold Is a Unique Differentiator Only Myriad Can Provide 89 AS Threshold 60% of patients below the active surveillance threshold 3% risk of PCSM 20% risk of PCSM |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Redesigned Report Provides Clear and Actionable Results 90 Modification of pathology assessment Definitive cut-off for which patients are eligible for active surveillance |
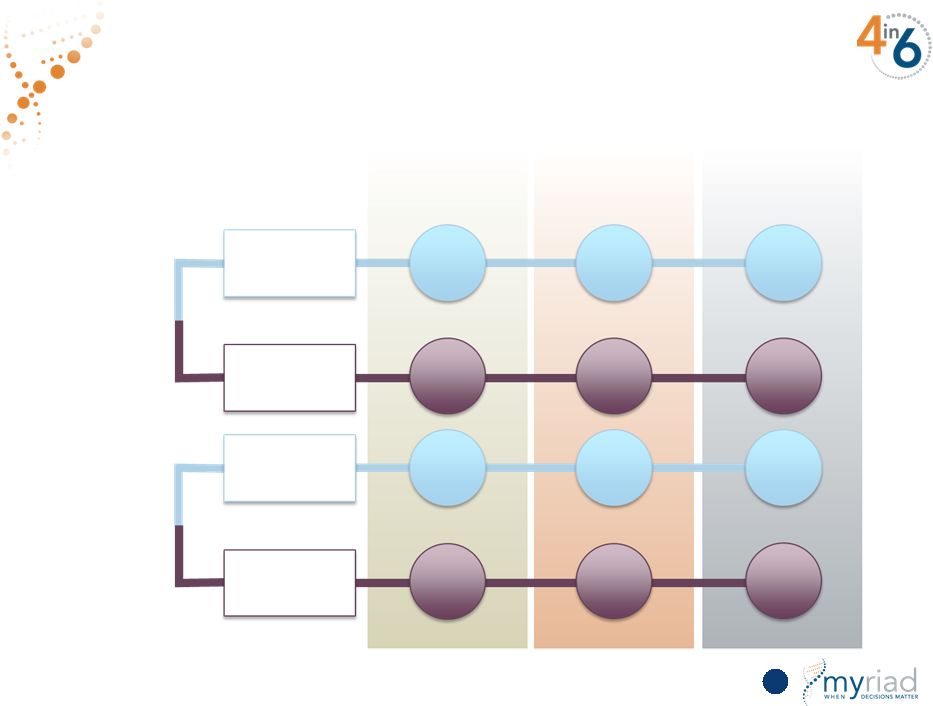 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. W I T H P R O L A R I S A S T H R E S H O L D I N I T I A L W I T H P R O L A R I S Unmatched Impact on Physician Treatment Decisions 91 AUA Low Active Surveillance Treatment 40% 60% 69% 31% 92% 8% AUA Intermediate Active Surveillance Treatment 8% 92% 27% 73% 27% 73% |
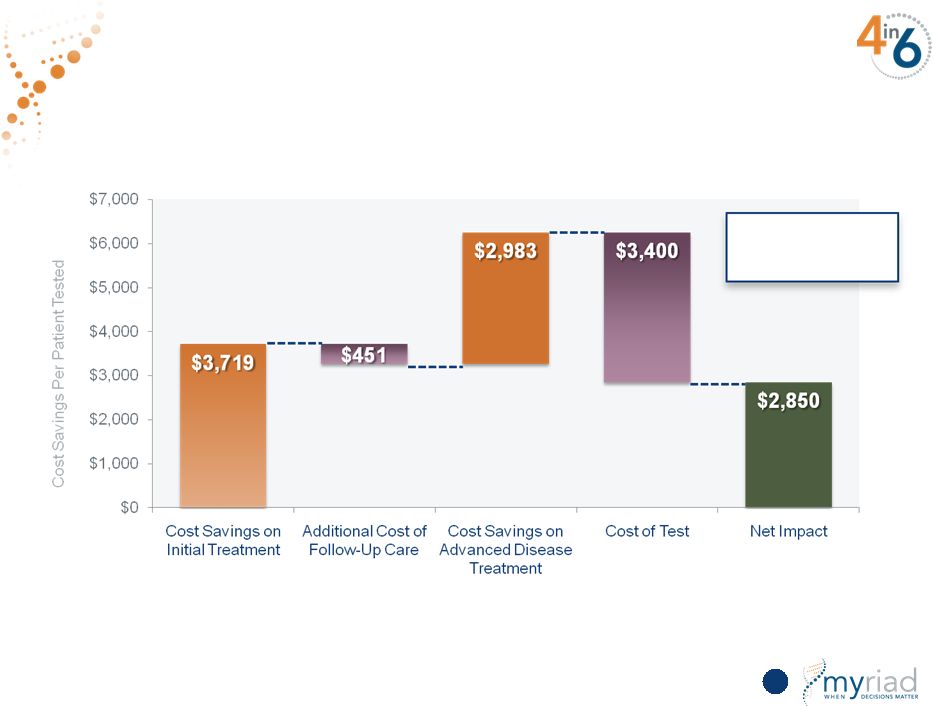 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Drives Substantial Economic Value for the Healthcare System 92 $2,850 saved per patient tested |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 93 Superior Data Are Driving Market Expansion and Growth |
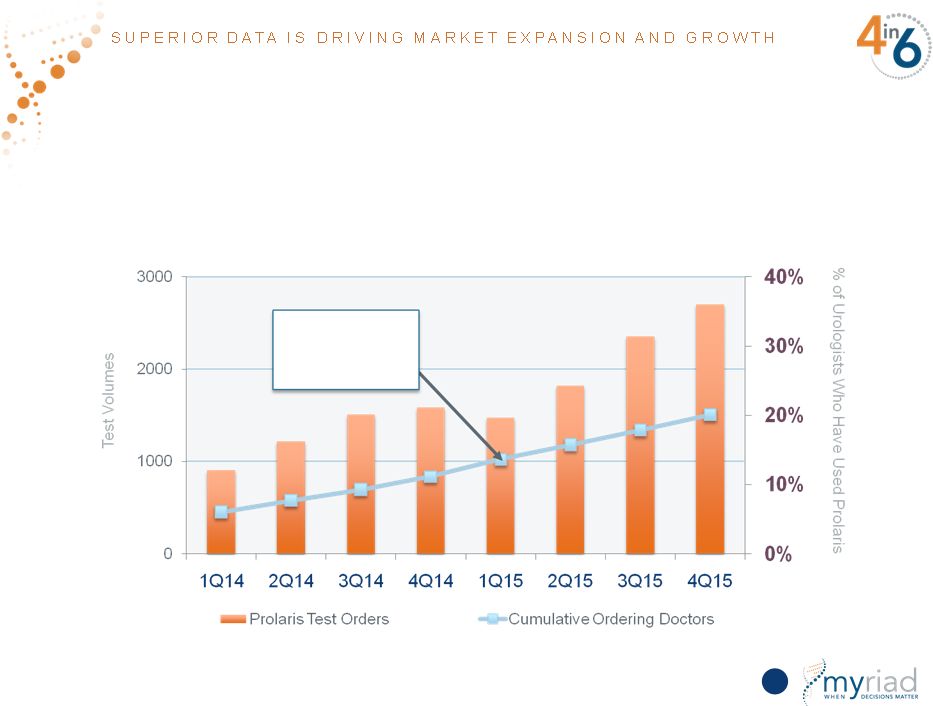 Significant Volume Growth From Breadth and Depth of Customer Base • Test utilization increased 20% throughout FY16 while ordering physicians increased 47% 94 PROCEDE 1,000 Stops Enrolling Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
   Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Focused Plan to Increase Coverage for Prolaris 95 CURRENT MEDICARE COVERAGE: Professional guidelines Retrospective safety data Prospective clinical data Medicare precedent NCCN guidelines Health economic data Low/Very Low Medicare Medicare Advantage EXPAND MEDICARE COVERAGE: EXPAND PRIVATE PAYER COVERAGE: |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Focused Plan to Increase Coverage for Prolaris 96 • Positive coverage decision from Tufts Health Plan • Payer thought-leader in the Northeast represents over one million lives • Covers all patients with localized prostate cancer • Value-based agreement • Assessed on % of patients choosing Active Surveillance • Takes advantage of the unique Prolaris Active Surveillance threshold • Approach can not be matched by other competitors and provides template for additional contracts |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Sensitivity Analysis Predicts Strong Growth 97 Revenue in millions Monte Carlo Simulation Factor Assumptions Market Growth • 30%-50% growth rate • 30%-60% market penetration • 5%-20% international mix Coverage • 80%-100% payer coverage Market Share • 50%-80% market share |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Significant Unmet Need In Prostate Cancer Treatment 98 Market Opportunity • Substantial unmet clinical need • Expanded Medicare coverage based on additional clinical data Key Advantages • Pioneering science differentiation based on gold standard endpoints • Definitive active surveillance threshold • Unmatched clinical utility data Commercial Breadth and Depth • Largest urology sales force in diagnostics • Increased physician adoption • Increased test utilization Positioned to become standard of care in the prostate cancer prognostic market |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. BREAK 99 1 1 2 2 3 3 4 4 5 5 6 6 7 7 8 8 9 9 10 10 |
 Vicki Fish 100 General Manager of Dermatology Transforming Melanoma Diagnosis Through Pioneering Science |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Revolutionizing Melanoma Diagnosis 101 Positioned to become market leader in melanoma diagnostics Market Opportunity • Significant unmet clinical need • Ability to impact physician decision making • Substantial economic value Key Advantages • Pioneering science • Extensively validated approach • One of the most accurate cancer diagnostics ever developed Commercial Breadth and Depth • Significant physician adoption • Increasing utilization |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Early and Accurate Diagnosis Critical to Survival 102 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Traditional Melanoma Diagnosis is Highly Subjective 15% to 47% discordance in peer reviewed literature 103 STUDY N DISCORDANCE Cerroni et al 57 47% Hawryluk et al 478 35% Piepkorn et al 149 46% Gerami et al 24 30% Veenhuizen et al 1,069 15% Shoo et al 392 15% Lodha et al 178 25% Farmer et al 37 35% Overall 2,384 31% |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 430,000 Patients Receive Indeterminate Results 104 Possible Under- Treatment Possible Over- Treatment 3.0 Million Skin Biopsies for Melanoma Per Year* 15% Discordance 430,000 Indeterminate Cases *Includes major European countries, U.S. and Canada |
   Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Costs Increase Dramatically for Later Stage Disease 105 Localized Melanoma Regional Melanoma Distant Melanoma Average 10-Year Cost per Patient $15,612 $29,033 $210,281 >10x increase in costs Undertreatment • Patient only receives monitoring • Recurrence as later stage melanoma • Reduction in survival as disease progresses • Significantly higher cost to treat Overtreatment • Psychological implications • Highly invasive surgery • Follow-on care • Permanent scarring • Unnecessary comorbidities (infection, nerve damage, etc.) INACCURATE DIAGNOSIS |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. myPath Melanoma: The Solution • 23 gene mRNA expression panel (13 genes tied to immune function, 1 gene tied to cell differentiation, 5 genes tied to cell signaling) • Unique approach uses information from inside and outside the cell • Validated in sample cohorts containing all melanoma and benign nevus subtypes • Demonstrated to be highly accurate at differentiating melanoma from benign skin lesions 106 Melanoma Diagnosis Score 1 Gene Cell Differentiation 5 Genes Cell Signaling 8 Genes Immune Group 9 Genes Housekeepers |
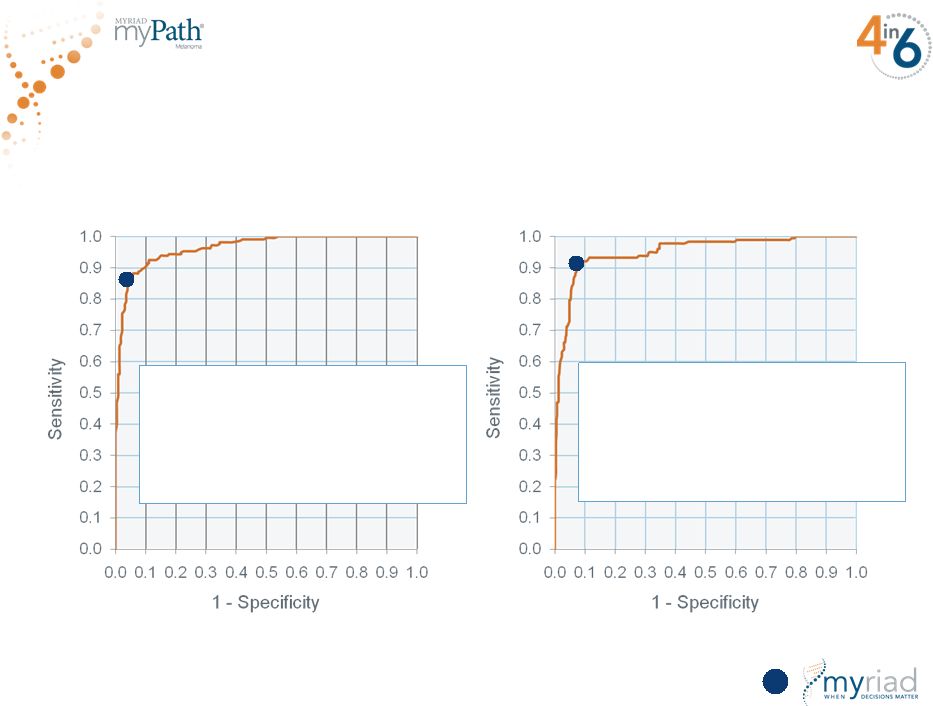   Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. One of the Most Accurate Cancer Diagnostics Ever 107 Clinical Validation 1 (N=437) Clinical Validation 2 (N=736) Diagnostic Accuracy = 90% 90% sensitivity 91% specificity AUC = 0.96 Diagnostic Accuracy = 92% 92% sensitivity 93% specificity AUC = 0.95 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. myPath Melanoma Led to a Significant Increase in Diagnostic Confidence 108 80% 38% Pre-Test Post-Test 9% 22% Pre-Test Post-Test 11% 41% Pre-Test Post-Test Indeterminate Diagnosis Malignant Diagnosis Benign Diagnosis >50% reduction in indeterminate results 2.4x increase in diagnostic confidence 3.7x increase in diagnostic confidence |
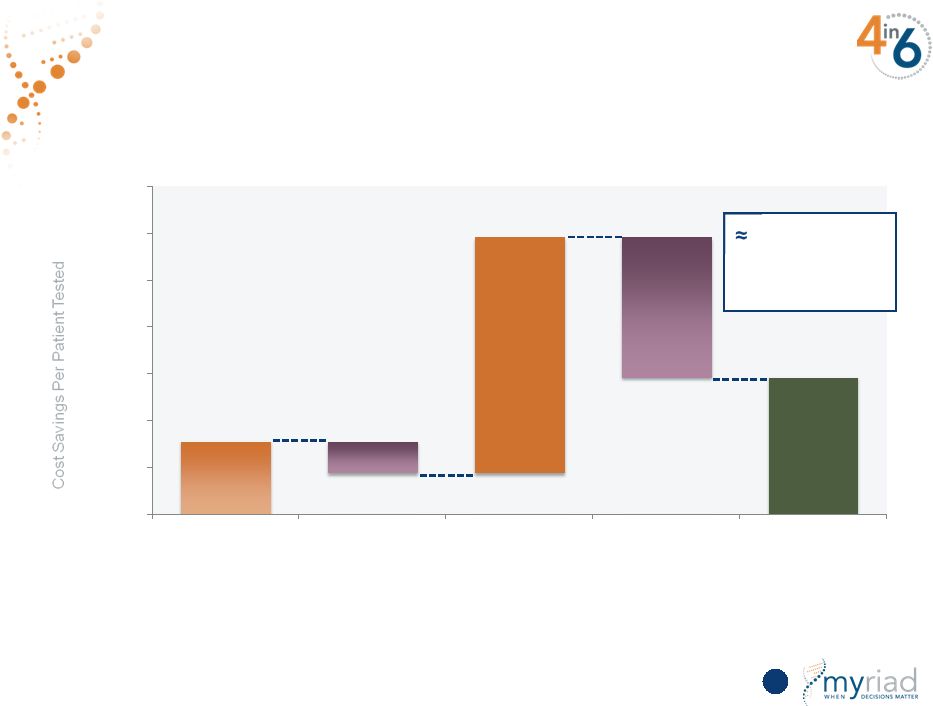  Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Drives Substantial Economic Value for the Healthcare System 109 $776 $1,462 $333 $2,519 $1,500 $0 $500 $1,000 $1,500 $2,000 $2,500 $3,000 $3,500 Initial Pathology Cost Savings Additional Initial Treatment Costs Cost Savings on Advanced Treatment Cost of Test Net Impact $1,500 saved per patient tested |
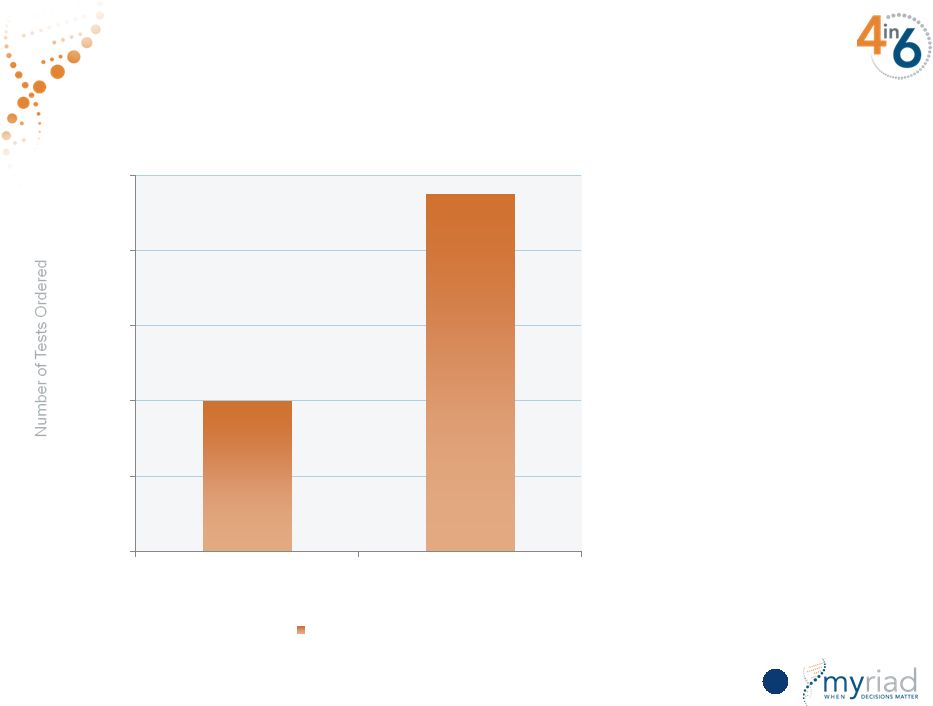 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 18% of Dermatopathologists Have Used myPath Melanoma 110 0 2,000 4,000 6,000 8,000 10,000 FY14 FY15 Tests Performed 18% of U.S. dermatopathologists have ordered myPath Melanoma |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Reimbursement Dossier Complete and Awaiting Publication 111 DOSSIER REQUIREMENT # OF STUDIES COMPLETE PUBLICATION STATUS Analytical Validation 1 Published in Biomarkers in Medicine Clinical Validation 5 1 Published in Journal of Cutaneous Pathology 4 Pending publication Clinical Utility 3 3 Pending publication Health Economic Model 1 Published in Journal of Medical Economics Guidelines/ Consensus Statement 1 1 Pending publication |
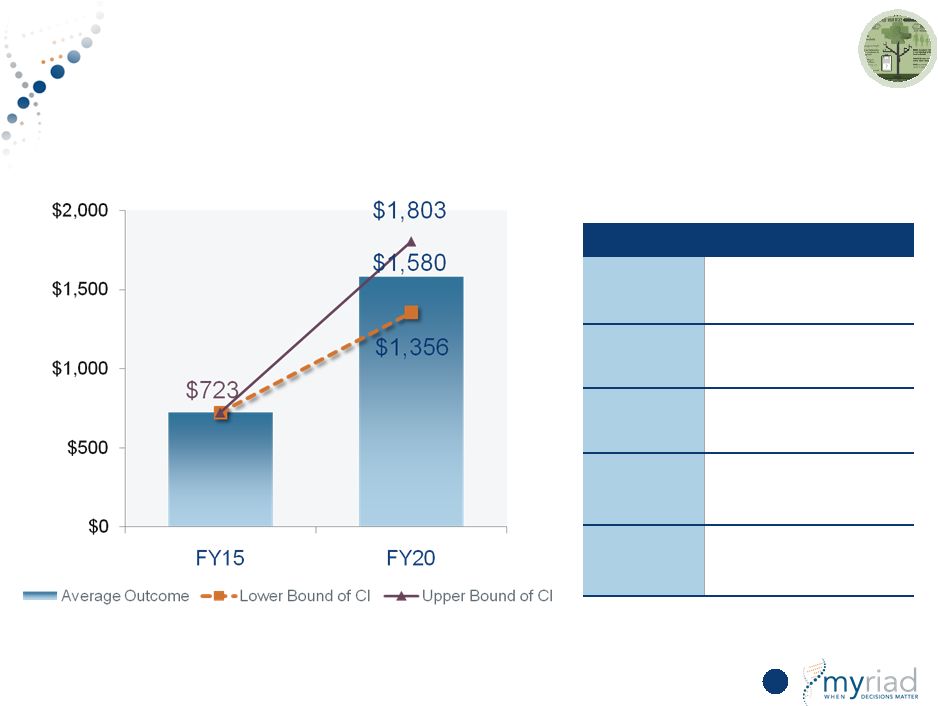
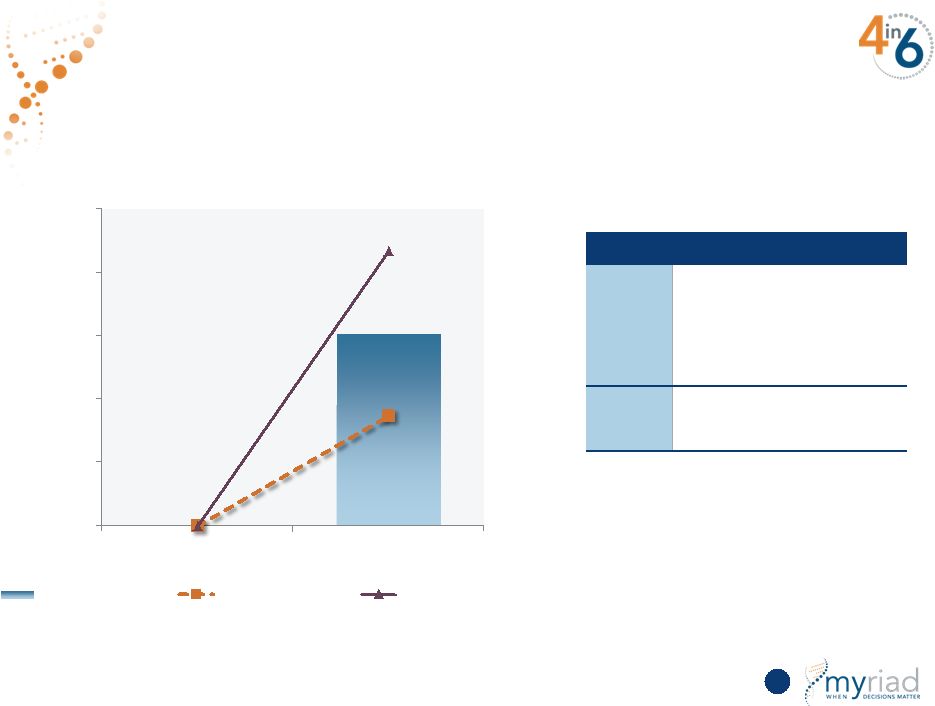 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Sensitivity Analysis Predicts Strong Growth 112 Revenue in millions Monte Carlo Simulation Factor Assumptions Market Growth • 30%-50% growth rate • 5%-13% market penetration • 5%-15% international mix Price • 80%-100% payer coverage $0 $76 $44 $108 $0 $25 $50 $75 $100 $125 FY15 FY20 Average Outcome Lower Bound of CI Upper Bound of CI |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Revolutionizing Melanoma Diagnosis 113 Positioned to become market leader in melanoma diagnostics Market Opportunity • Significant unmet clinical need • Ability to impact physician decision making • Substantial economic value Key Advantages • Pioneering science • Extensively validated approach • One of the most accurate cancer diagnostics ever developed Commercial Breadth and Depth • Significant physician adoption • Increasing utilization |
 Jerry Lanchbury 114 Chief Scientific Officer Industry Leading Pipeline to Ensure Growth Opportunities |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Promising Early-Stage Pipeline Opportunities 115 Substantial market opportunity Ability to leverage existing sales channels Outstanding early discovery data Strong clinical need for the product Utilize existing DNA, RNA and protein expertise myPath Bipolar myPath Prostate Cancer myPath Pancreatic Cancer myPlan Renal Cancer |
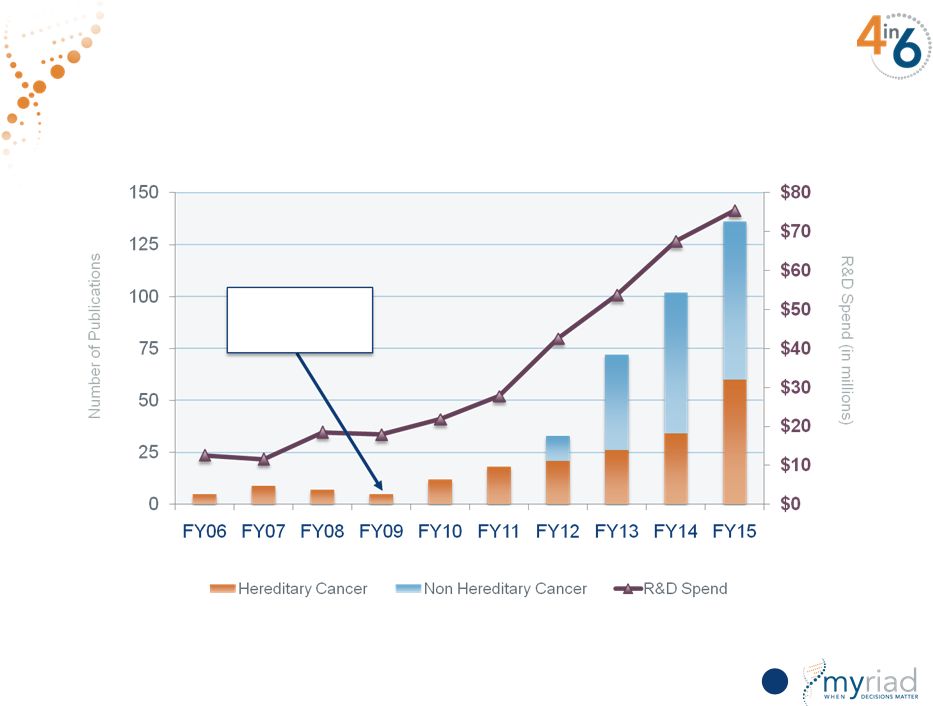 Increasing Research Investment Yields Substantial Scientific Output 116 Pharma Spin-off Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Industry Leading Pipeline Facilitates Long-Term Growth $10B $8B $10B+ Stage 3 REIMBURSEMENT Stage 2 VALIDATION Stage 1 DISCOVERY • myRisk • Prolaris • Vectra DA • EndoPredict • BRACAnalysis CDx 1 • Tumor BRACAnalysis CDx • myPath Melanoma • myPlan Lung Cancer • myChoice HRD (Platinum) 2 • myChoice HRD (PARP) 3 • myPlan Renal Cancer • myPath Bipolar • myPath Pancreatic Cancer • myPath Psoriatic Arthritis • myPath Prostate Cancer • myPath Endometriosis Total Addressable Market (TAM) 1 Ovarian Cancer, Breast Cancer, Pancreatic Cancer 2 Triple Negative Breast Cancer, HER2- Breast Cancer 3 Ovarian Cancer, Breast Cancer, Pancreatic Cancer, Metastatic Prostate Cancer 117 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 118 Renal Cancer |
 |
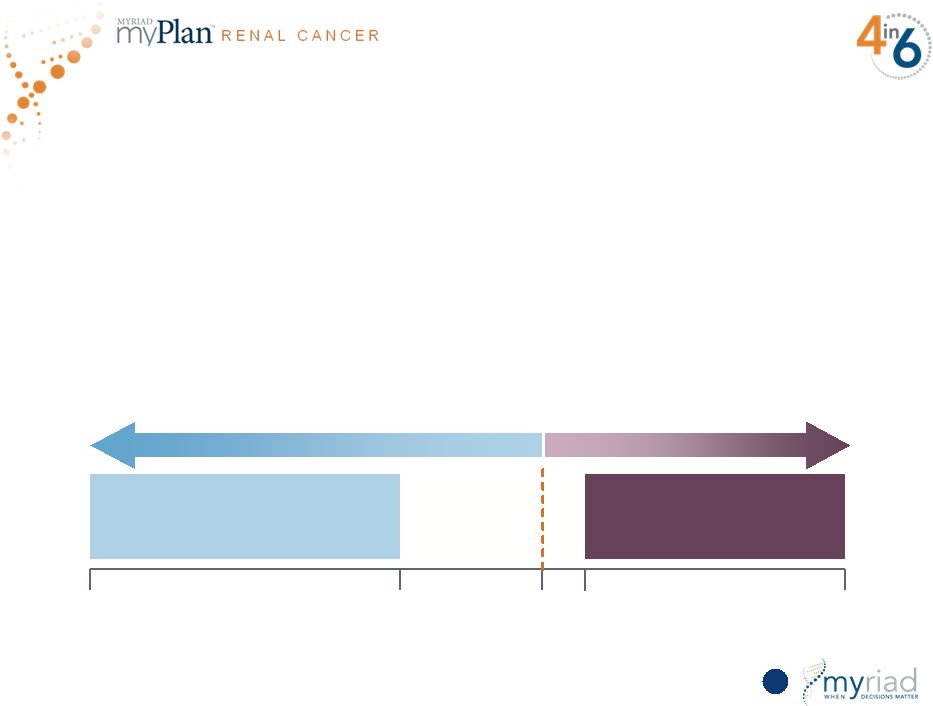 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Groundbreaking Science Uncovers Broadly Applicable Signature for Cancer Prognosis • Cell cycle progression (CCP) genes have demonstrated broad utility as a cancer prognostic • Now validated in prostate, lung, breast and renal cancers • Whole transcriptome analysis shows CCP genes are the only relevant expression targets for multiple cancers 120 *Corrected for the impact of CCP Non-CCP Genes* CCP Genes p Value 1.0 0.34 0.05 0.04 2x10 -7 Ability to Predict Metastases in Renal Cancer STATISTICALLY INSIGNIFICANT STATISTICALLY SIGNIFICANT |
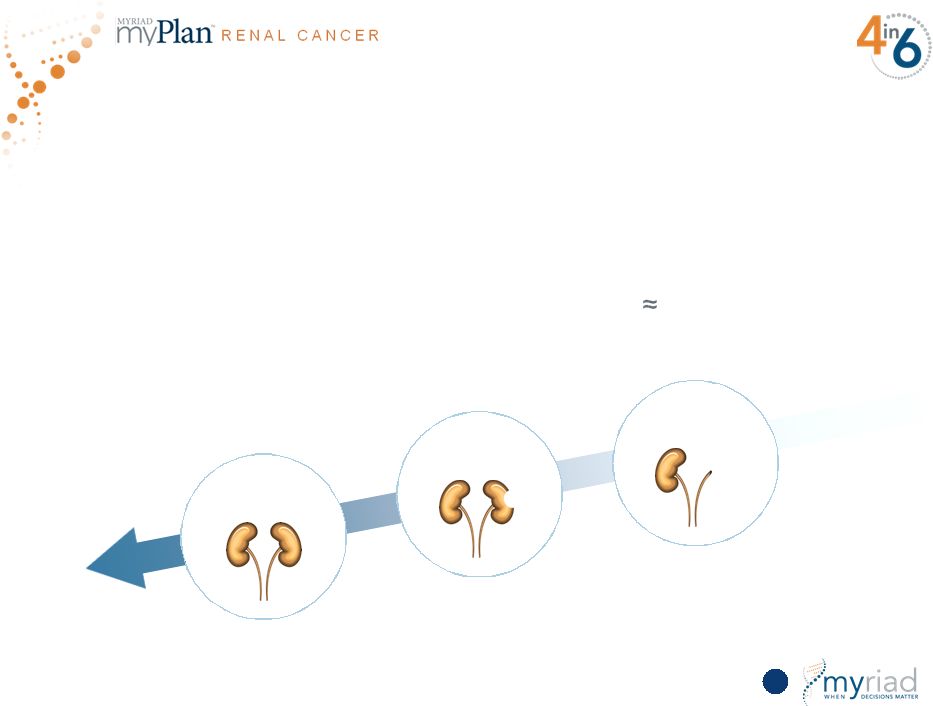 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Substantial Clinical Need for Renal Cancer Prognostic 121 Higher risk of significant complications including ESRD, need for dialysis, etc. NEPHRECTOMY PARTIAL NEPHRECTOMY ACTIVE SURVEILLANCE • Trend toward less invasive therapy • Five-year survival for stage 1 and stage 2 disease >75% • No existing tools to differentiate low/high-risk patients • 90,000 patients diagnosed with localized disease $300M global market opportunity |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Excellent Prognostic Power Shown in Training Data • Panel of 31 cell cycle progression genes and 15 control genes (same as Prolaris) • Training study completed with localized renal cancer • Panel was highly statistically significant at predicting 5-year risk of metastases; odds ratio of 3.89, p value of 0.0072 122 * Case control study in high risk population |
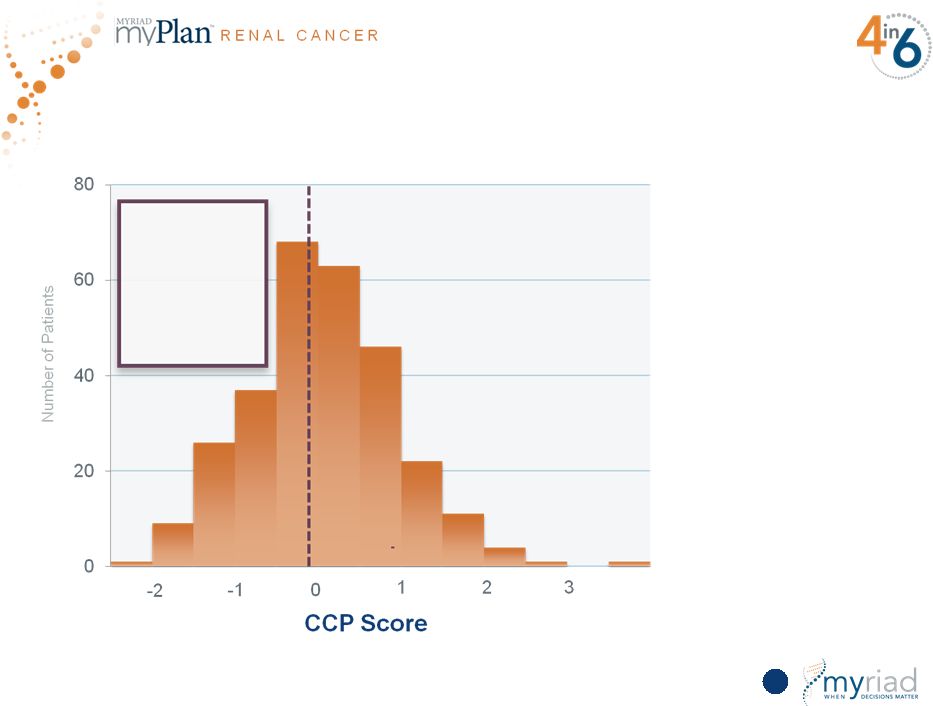 First Look at myPlan Renal Validation Data 123 44% of patients have less than a 3% risk of 5-year disease mortality • Hazard ratio = 3.00, p value = 1.4x10 -6 • Next steps: present and publish first validation; second validation will be completed and presented by 4Q16 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 124 Bipolar |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 70% of Bipolar Patients Initially Misdiagnosed Symptoms of bipolar disorder and major depression are almost exactly the same 125 BIPOLAR DISORDER • Fatigue • Feelings of worthlessness • Impaired concentration • Insomnia or hypersomnia • Diminished interest in activities • Thoughts of suicide • Weight gain/loss • Mania MAJOR DEPRESSIVE DISORDER • Fatigue • Feelings of worthlessness • Impaired concentration • Insomnia or hypersomnia • Diminished interest in activities • Thoughts of suicide • Weight gain/loss |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Cost of Misdiagnosis is Substantial • Over 20 million patients per year present with symptoms consistent with major depressive disorder (MDD) or bipolar disorder (BP) • 90% of patients are diagnosed by primary care physicians • Bipolar patients do not respond to first-line or subsequent therapeutics for MDD • MDD anti-depressive therapeutics can trigger manic psychosis in a subset of BP patients • BP symptoms are 6th leading cause of disability in 15-44 age group • Economic impact of improperly treated BP in U.S. is $72B annually 126 |
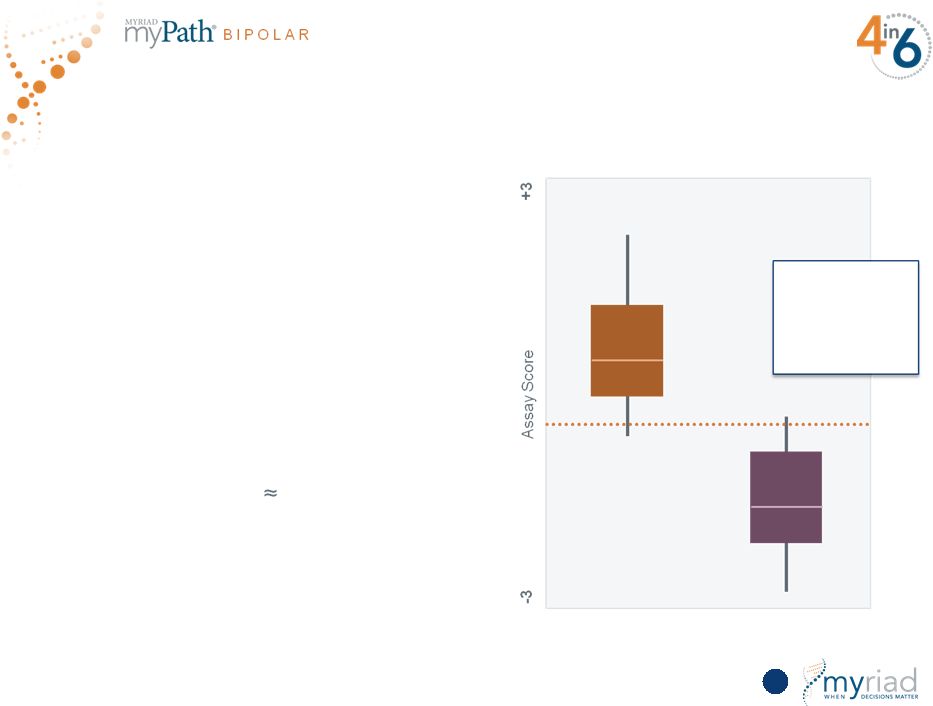 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Outstanding Early Data Show Ability to Differentiate Bipolar from Major Depression 127 96% AUC in Training Set • Multimarket proteomic assay run on Myriad-RBM Luminex platform • Consists of 18 protein analytes from blood • Discovery completed in 150 well- characterized BD1 and MDD samples • Next steps: Validation study sponsored by Myriad and partnered with 3 major medical centers; will enroll 300 patients beginning mid-FY16 BIPOLAR DISORDER MAJOR DEPRESSIVE DISORDER |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 128 Pancreatic |
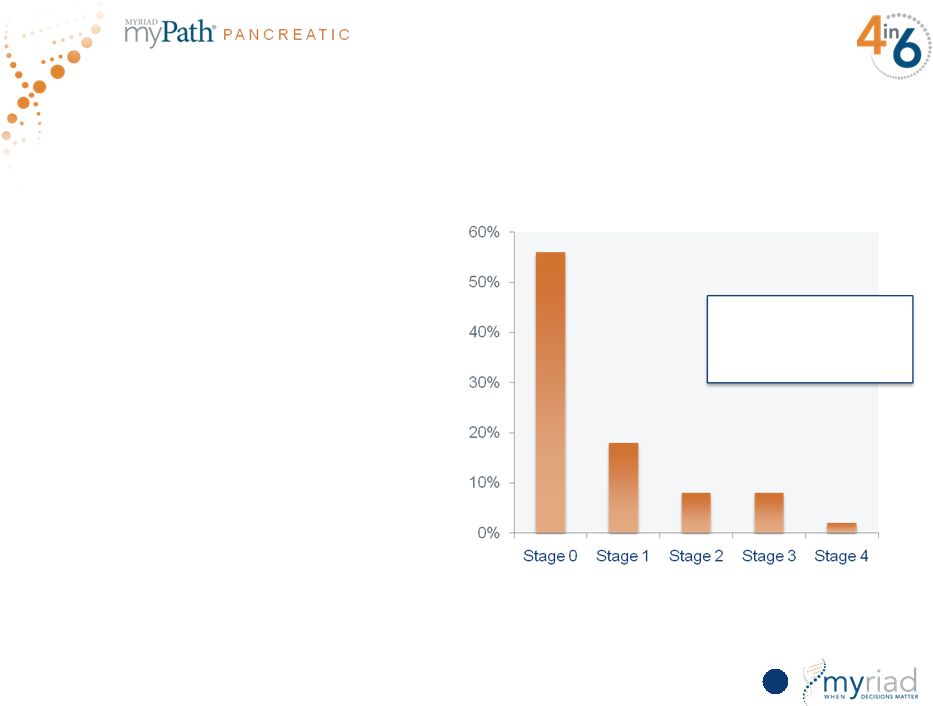 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Early Detection of Pancreatic Cancer Crucial to Survival • 5-year survival only 7% • Symptoms are vague and often misconstrued with other health issues • Most early-stage patients diagnosed through unrelated imaging procedures • Only 15% of patients diagnosed with localized/resectable disease • 102,000 new diagnoses annually 129 Urgent need for an early detection diagnostic Five-Year Survival by Stage for Pancreatic Cancer Most patients currently diagnosed with late- stage disease |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Promising Early Data on myPath Pancreatic Cancer • 136 patient study comparing 42 early- stage pancreatic cancer cases to 94 healthy controls and patients with chronic pancreatitis • 92% AUC when diagnosing early stage pancreatic cancer • Next steps: finalize biomarker set in FY16 130 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 131 Prostate Cancer |
  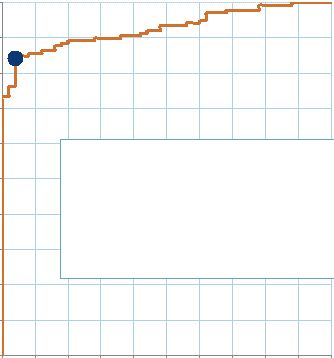  Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Proof of Principle Established for Urine-Based Cancer Detection • Significant need to diagnose urological cancers at an earlier stage (prostate, kidney and bladder cancer) • Discovery study evaluated 139 tumor samples • Proprietary assay was able to differentiate patients with cancer from healthy controls • Next steps: application of technology to prostate cancer 132 0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 1- Specificity Diagnostic Accuracy = 90% 85% sensitivity 96% specificity AUC = 0.93 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Pioneering Research Fuels Industry Leading Pipeline • Uniquely positioned to use all molecular diagnostic tools (DNA, RNA, proteins) for research and development • Scientific output has increased >20x since FY09 140 publications per year • Breakthrough thinking drives discovery engine – Broadly applicable cancer prognosis signature – Proprietary cancer pathway (myChoice HRD) test vs. gene panels – Addition of immune response genes for diagnosis – Complex multiplex protein signatures – Signatures combining DNA, RNA and proteins 133 |
 134 Gary King Executive Vice President of International Operations Expanding Our Horizons in International Markets |
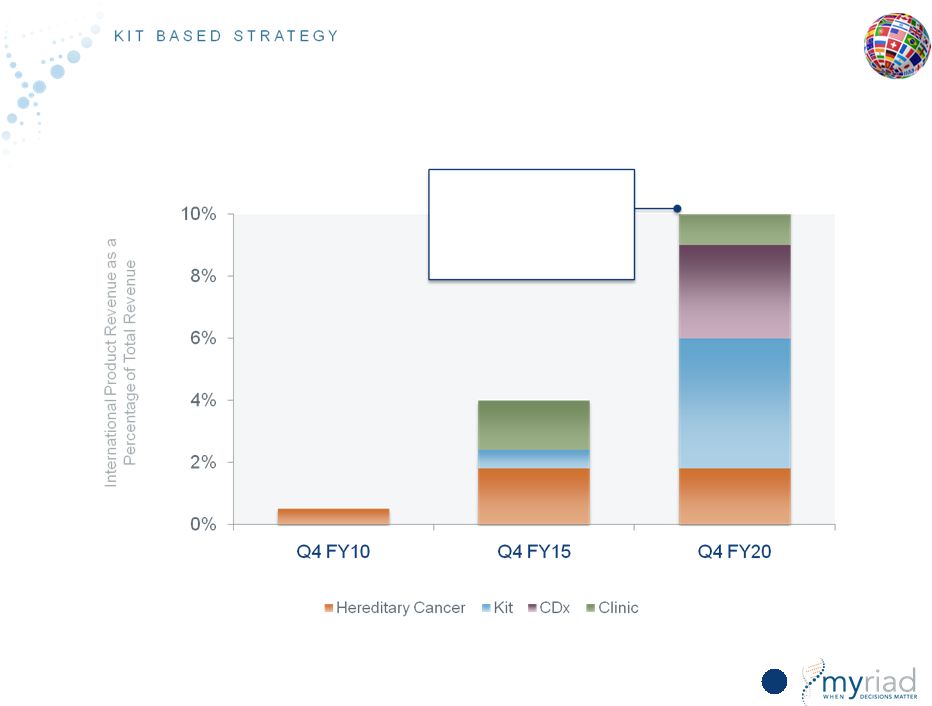
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Incredible Growth Opportunity in International Markets 135 Complex reference laboratory tests, companion diagnostics and high- value kits are most significant long-term growth drivers Market Opportunity • 60% of the global market is outside of the United States (O.U.S.) • 4% of revenue O.U.S. today; goal to reach 10% by FY20 Commercial Breadth and Depth • Expanding reimbursement • Expand current products to kits Key Advantages • High complexity reference lab tests • Companion diagnostic partnerships • Kit-based strategy |
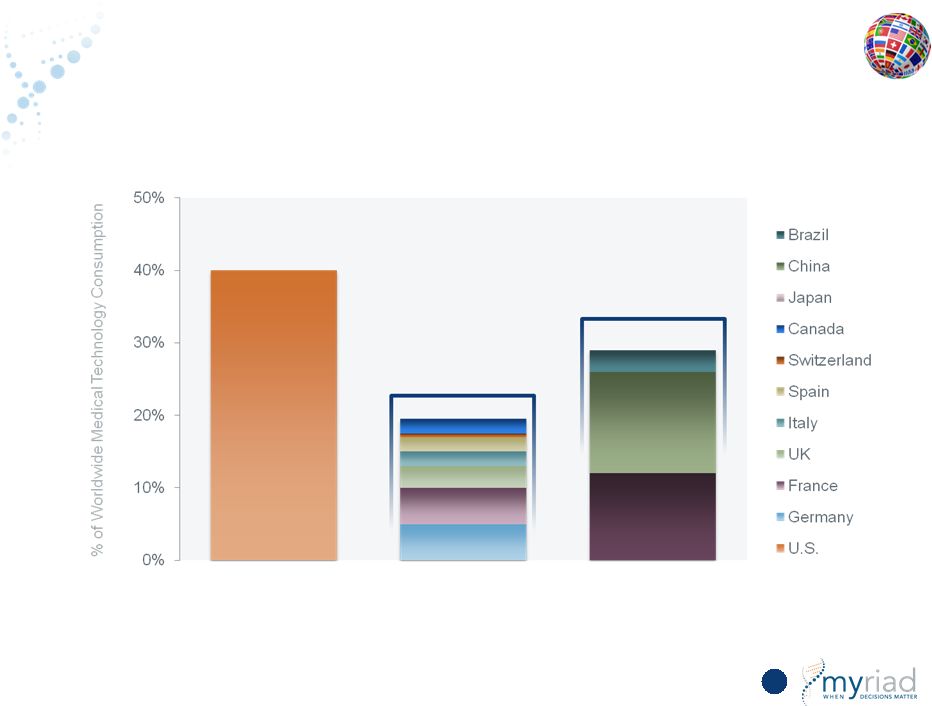 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Total Available Market (TAM) in 10 Major International Markets > U.S. 136 29% 20% 40% Long-Term Opportunity: >70% of U.S. Market Source: European Federation for Pharmaceutical Industry IFPMA Facts & Figures 2012 EU6 + Canada Near Term Opportunity: 50% of U.S. Market |
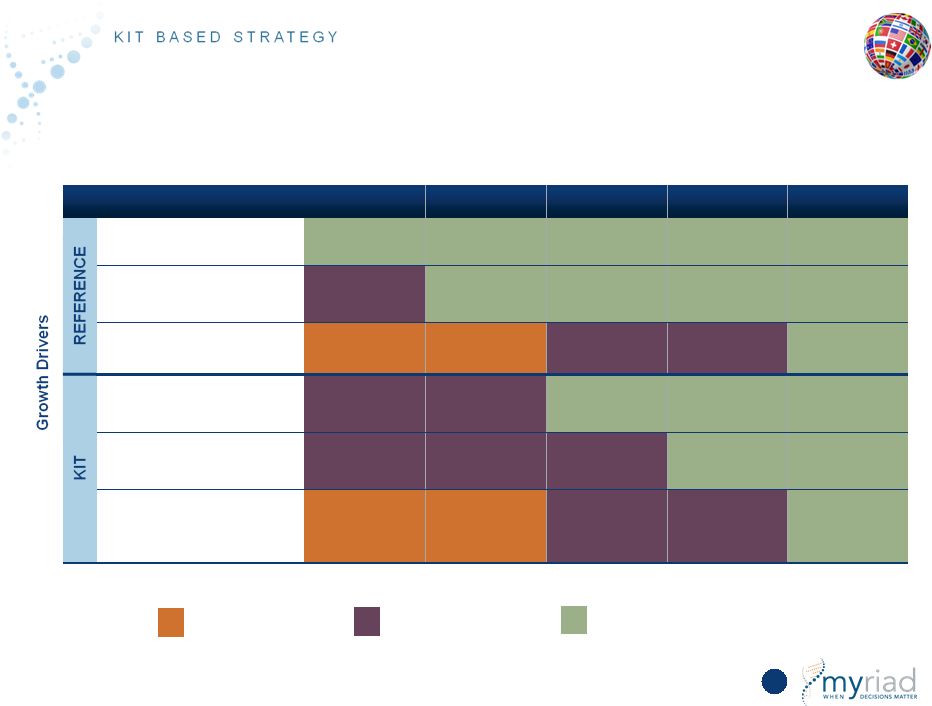
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Key Learnings Drive Strategic Review 137 KEY LEARNING STRATEGY Laboratories are captive within institutions 1. Emphasize reference tests that are too complex for institutional laboratories (myRisk and CDx) 2. Develop proprietary test kits for distribution to institutional laboratories Lengthy reimbursement throughout Europe 1. Acquire German clinic 2. Emphasize health economic studies 3. Incentivize KOL involvement with kit format |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Refined Strategy to Reflect Differences in International Market 138 COUNTRIES Near-Term Growth: EU6 + Canada Long-Term Growth: Japan, China, and Brazil REFERENCE TESTS DNA (multiple platforms): Companion Diagnostics KITS RNA (platform partner): • EndoPredict • Prolaris • myPlan Lung • myPath Melanoma • myPlan Renal Protein (platform partner): • Vectra DA • myPath Bipolar • myPath Pancreatic |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Hereditary Cancer Expansion in Europe 139 • European market increasingly moving to myRisk Hereditary Cancer; myRisk was 32% of hereditary cancer revenue in the 4Q15 compared to 23% in the 4Q14 • Complexity of large panel tests is beyond the capability of most small, decentralized labs • Beginning discussions with German hospital/physician networks following the acquisition of MVZ clinic in 3Q15; potential for positive impact in German market in 2H16 and beyond • Many private healthcare systems in major European countries now cover myRisk Hereditary Cancer |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. International Companion Diagnostic Opportunity 140 |
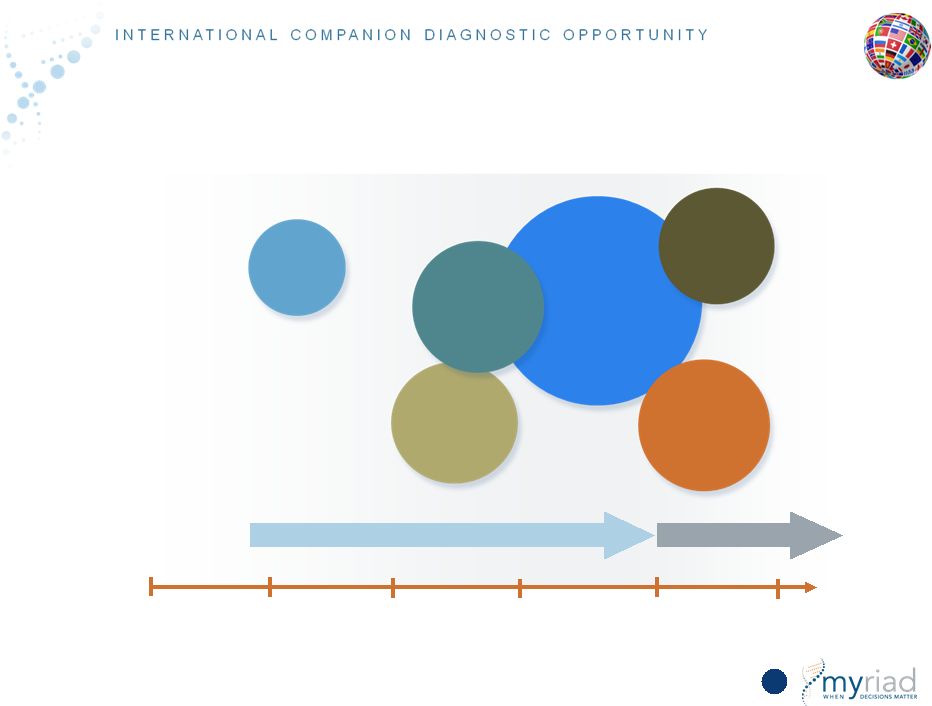 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Companion Dx Opportunity O.U.S. = $3B TAM 141 FY15 FY16 FY17 FY18 FY19 FY20 Tumor BRACAnalysis CDx Launch Japan, China & ROW Ovarian Cancer (30,000) Major country reimbursement for Lynparza Platinum in TNBC (55,000) Platinum in HER2- BC (65,000) PARPs in Breast Cancer (200,000) PARPs in Metastatic Prostate Cancer (40,000) PARPs in Pancreatic Cancer (57,000) Focus on EU6 and Canada |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Progress with Lynparza Launch in Europe 142 Country Status France Launched and reimbursed using temporary system starting in March, final negotiations underway Germany Launched in June; G-BA assessment ongoing Denmark/Luxembourg Launched and reimbursed since June Sweden/Netherlands National reimbursement review completed; awaiting decision Italy, UK, Spain, Belgium, Portugal, Norway National reimbursement review process ongoing |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Kit-Based Strategy 143 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 14.6% 49.4% 80.6% 4.9% 31.2% EndoPredict Increases Low-Risk Group by 340% Without Increasing Risk 144 1,702 Patients Classified By Pathology Intermediate Patients Re-Classified With EndoPredict HIGH - 45% INT. - 10% LOW - 5% LOW - 5% HIGH - 20% |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. EndoPredict Validates Kit-Based Model • Over 40 worldwide sites using EndoPredict • Substantial preference for in-house testing and economic sharing • Faster path to reimbursement; ability to utilize local stakeholders as advocates • Attractive financial model given stickiness of testing once account is established 145 7 additional installations ROW |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. A Number of Myriad Products Are Conducive to Kits 146 RNA Expression Protein In discussions with potential partners |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Expanded Reimbursement Will Drive Increased International Growth 147 YEAR FY16 FY17 FY18 FY19 FY20 Hereditary Cancer Tumor BRACAnalysis CDx Major country reimbursement throughout FY16 myChoice HRD First PARP using HRD EndoPredict Germnay PMI UK PMI/NHS Switzerland Germany GBA Canada France Prolaris Switzerland UK PMI Germany PMI UK NHS France Germany GBA Canada myPath Melanoma Germany PMI UK PMI Germany GBA Canada Switzerland France No Reimbursement Low Reimbursement Broad Reimbursement |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 10% of Global Revenue From International Markets by FY20 148 Goal to have international product revenue comprise 10% of sales by FY20 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Incredible Growth Opportunity in International Markets 149 Complex reference laboratory tests, companion diagnostics and high- value kits are most significant long-term growth drivers Market Opportunity • 60% of the global market is outside of the United States (O.U.S.) • 4% of revenue O.U.S. today; goal to reach 10% by FY20 Commercial Breadth and Depth • Expanding reimbursement • Expand current products to kits Key Advantages • High complexity reference lab tests • Companion diagnostic partnerships • Kit-based strategy |
 150 Bryan Riggsbee Chief Financial Officer Five-Year Outlook: Increased Growth and Financial Leverage |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 5-Year Outlook: Increased Growth and Financial Leverage 151 • Hereditary cancer growing low single digits • Significant diversification from product pipeline • International becomes larger contributor >30% Op Margins 7 Products >$50M >10% Revenue Growth CAGR >10% of Revenue from International • Majority of investments are completed • Meaningful operating margin improvement as new products obtain reimbursement • Prioritize internal R&D • Pursue accretive M&A • Continue opportunistic share repurchase Maximizing LT Shareholder Value Revenue Growth Operating Leverage |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 5-Year Outlook: Increased Growth and Financial Leverage 152 Maximizing LT Shareholder Value Revenue Growth Operating Leverage |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Assumptions for FY16 Guidance DOWNSIDE RISKS BASE CASE FOR GUIDANCE UPSIDE POTENTIAL • Hereditary cancer market losses > market growth • Hereditary cancer revenue of $638 to $649 million • Hereditary cancer market losses < market growth • Impact from expanded payer coverage for colon and endometrial cancer • Vectra DA revenue of $50 to $55 million • Expanded private payer coverage • Medicare reimbursement starting later than October 1, 2015 • Prolaris revenue of $10 to $12 million • Private payer coverage • Medicare reimbursement prior to October 1, 2015 • Expanded Medicare coverage • Pharmaceutical and Clinical Services revenue of $40M • Other revenue of $12 to $14 million • Reimbursement for EndoPredict, Tumor BRACAnalysis CDx, myPath Melanoma or myPlan Lung Cancer FY16 Guidance Covered in August 153 |
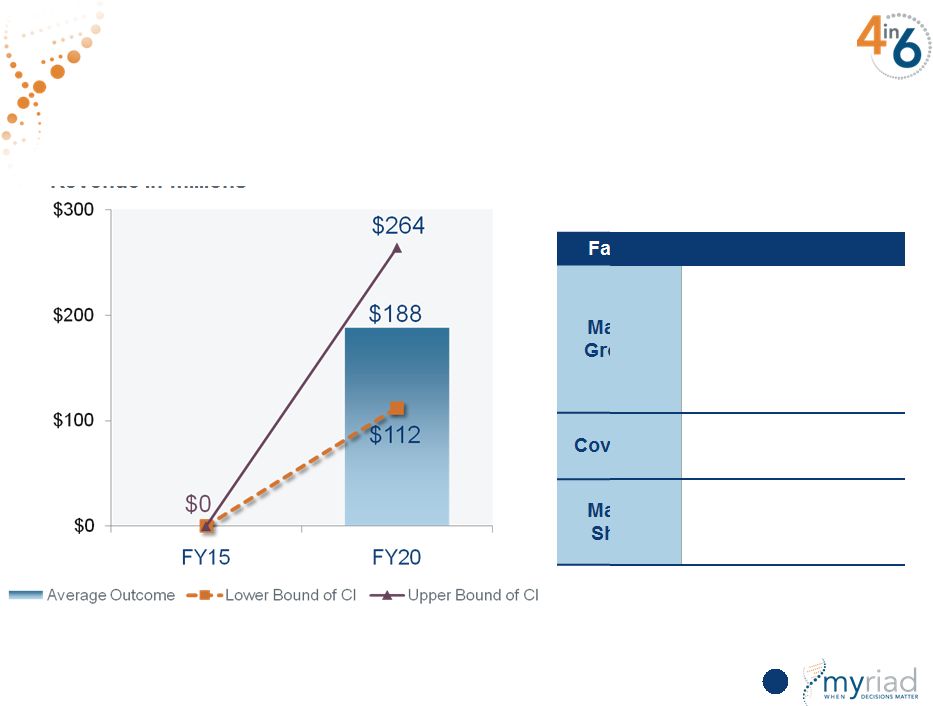
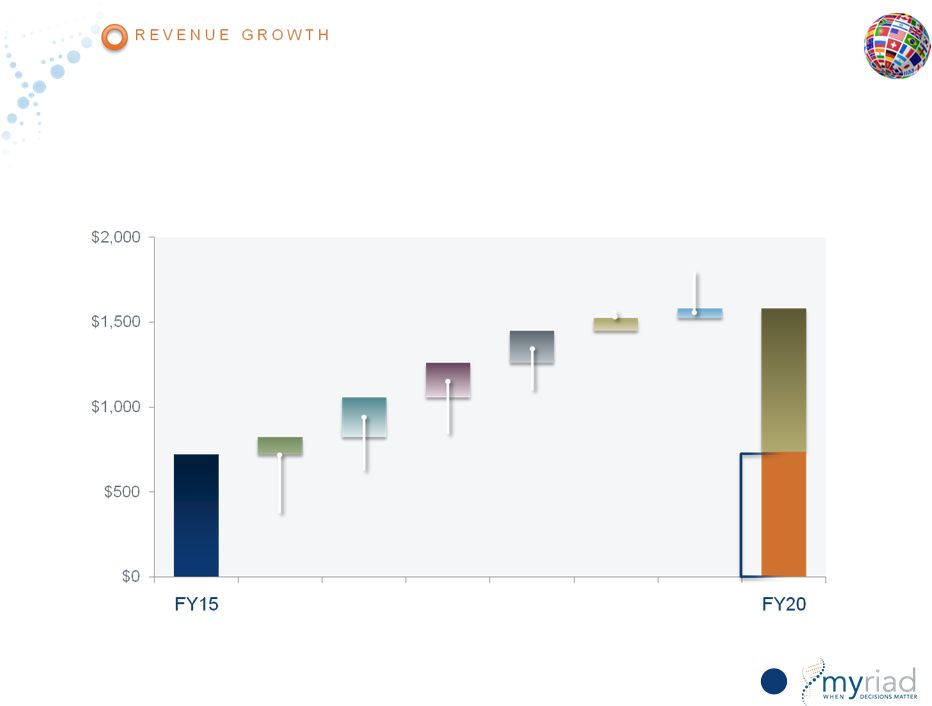
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Myriad Meets 5-Year Revenue Growth Target At Lower End of Sensitivity Analysis 154 Revenue in millions Monte Carlo Simulation Factor Assumptions Hereditary Cancer • Revenue of $554 to $922 million CDx • Revenue of $122 to $364 million Vectra DA • Revenue of $161 to $335 million Prolaris • Revenue of $112 to $264 million myPath Melanoma • Revenue of $44 to $108 million |
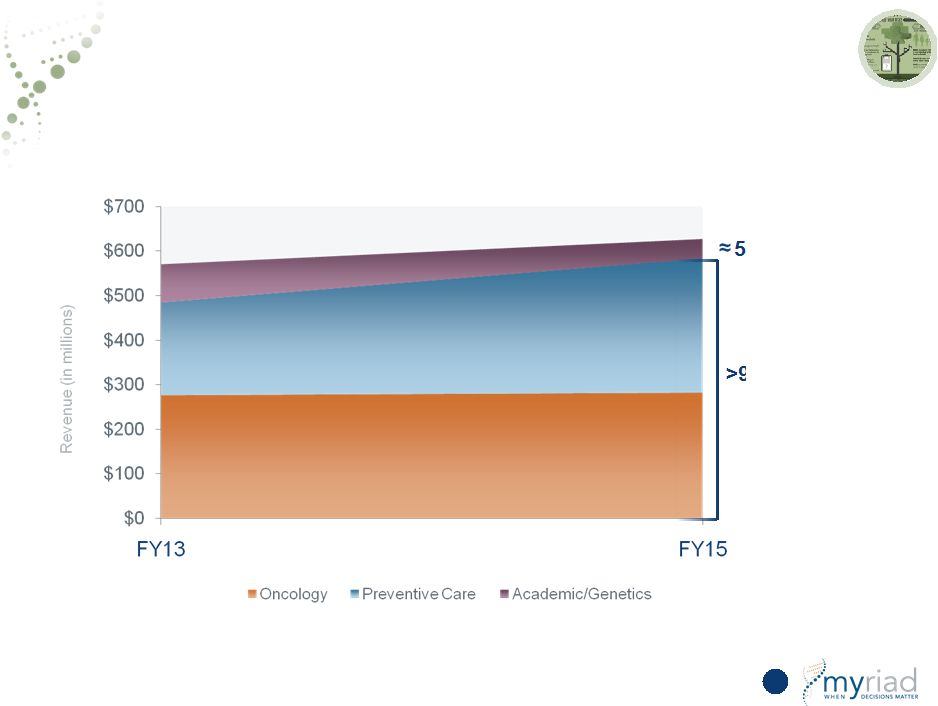
 New Products Represent >50% of FY20 Revenue 155 HEREDITARY CANCER MYPATH MELANOMA <50% of revenue from hereditary cancer COMPANION DX VECTRA DA PROLARIS PHARMA & CLIN SERVICES/OTHER Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 5-Year Outlook: Increased Growth and Financial Leverage 156 Operating Leverage |
 Operating Margin Component Changes FY13-FY15 157 (5.0%) (1.6%) (1.0%) (0.5%) (0.5%) 0.8% (6.2%) Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Operating Margin Component Changes FY15-FY16 158 0.4% 0.3% 0.2% 0.2% 0.8% |
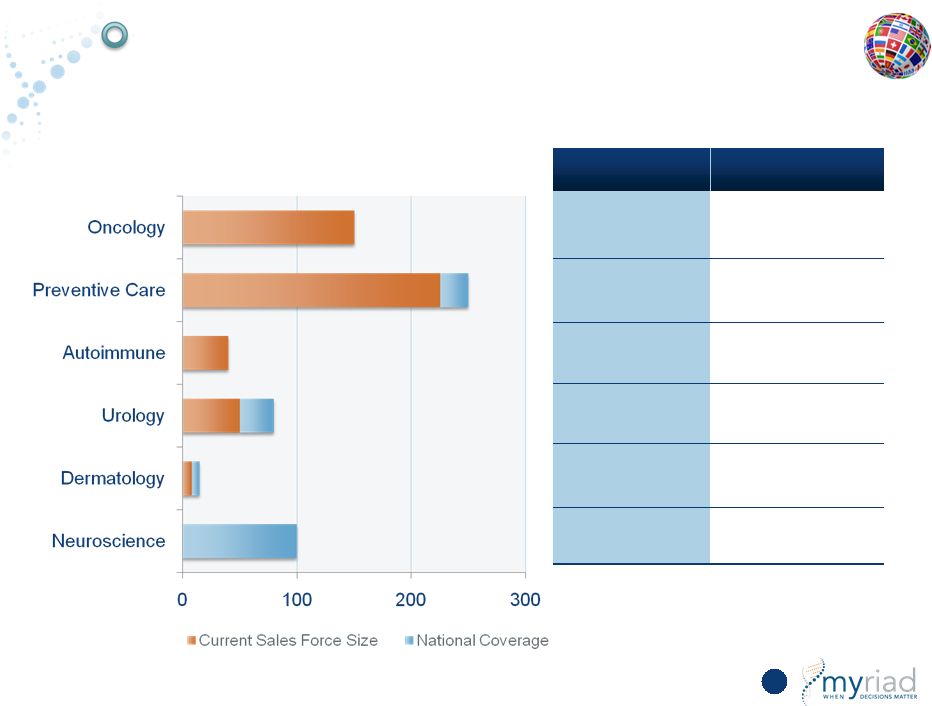 Meaningful Opportunity to Leverage Existing Sales Infrastructure 159 OPERATING LEVERAGE CURRENT PRODUCTS FUTURE PRODUCTS myRisk BRCA CDx EndoPredict myChoice HRD myRisk myPath Bipolar Vectra DA Psoriatic Arthritis Prolaris myPlan Renal Cancer myPath Prostate myPath Melanoma Psoriatic Arthritis None myPath Bipolar Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
 Increased Profitability In Pipeline Products Supports >30% Operating Margins 160 OPERATING LEVERAGE Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Five-Year Outlook: Increased Growth and Financial Leverage 161 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. CAPITAL ALLOCATION PRIORITY CAPITAL DEPLOYMENT SINCE JUNE 2010 GOAL R&D 9% of revenue 8% to 10% of revenue M&A $340M 43% of FCF • Use cash on hand to fund smaller deals (<$100M) • Use cash and leverage to fund larger deals ($100M-$600M) • Use equity to fund strategic deals (beyond borrowing capacity) Share Repurchase >$1B 127% of FCF • Target 100% of FCF • Reduce share repurchases based upon M&A visibility • Maintain cash at $100M to $200M Dividend None No plans for dividend given more attractive uses of capital Capital Deployment Strategy 162 |
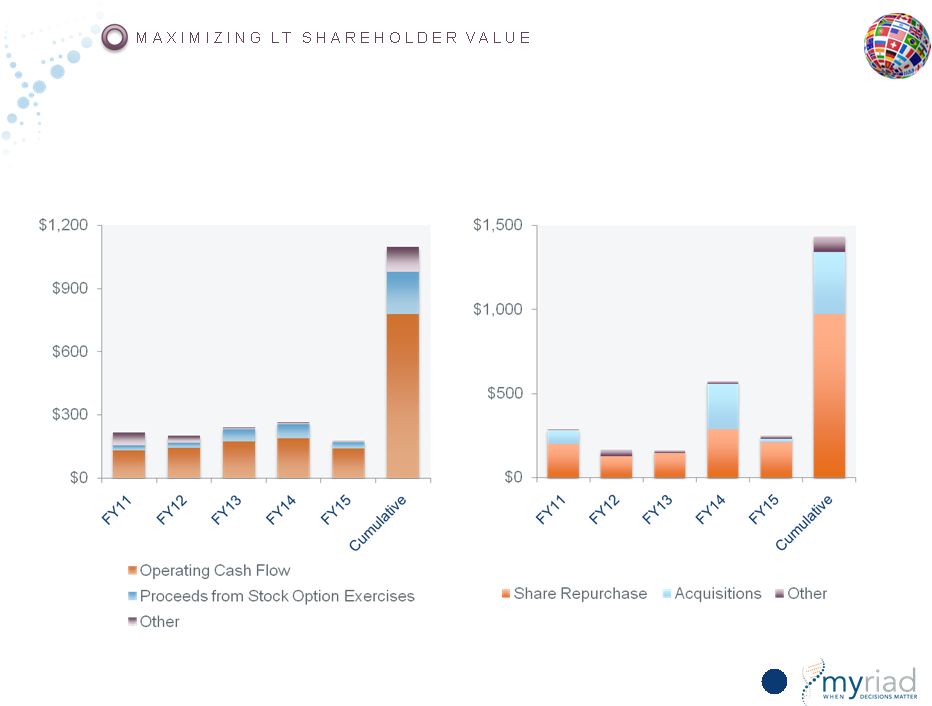 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Historical Cash Generation/Uses of Cash Cash Generation Uses of Cash 163 |
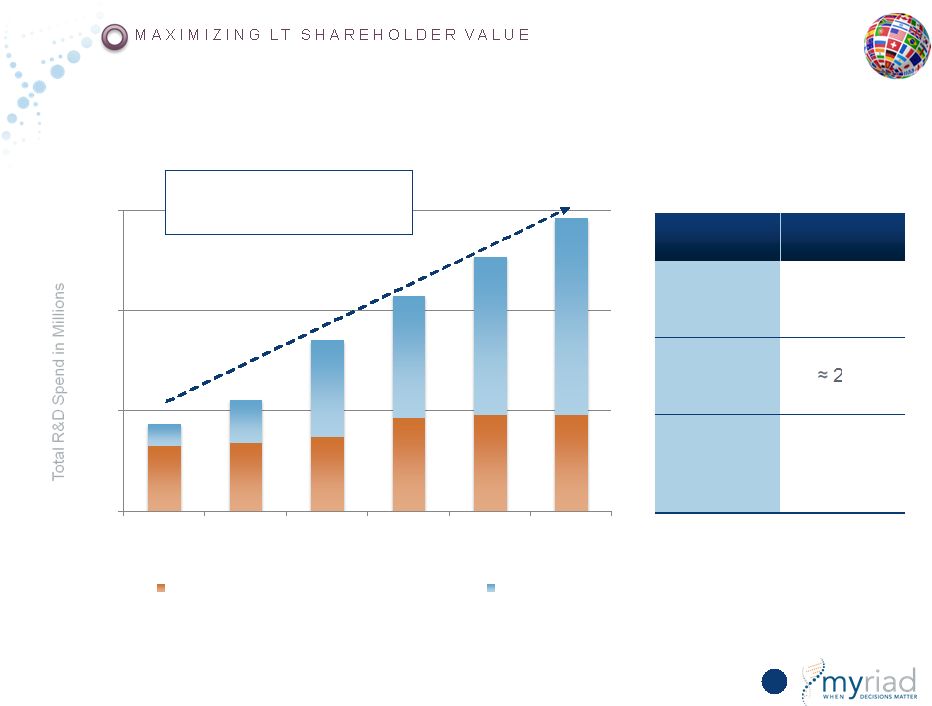  Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Internal R&D Represents Our Best Investment $0 $25 $50 $75 FY10 FY11 FY12 FY13 FY14 FY15 New Product Development (Stage 1 & 2) Stage 3 INVESTMENT EXPECTED ROIC Internal R&D 33% MYGN historical ROIC 20% Acquisitions Dependent on deal and market conditions 164 R&D Grew at 27% CAGR vs. Revenue at 15% CAGR |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Acquisitions – Opportunity For MYGN to be a Consolidator In a Diffuse Industry 165 Short-term visibility to the deal being accretive Strategic fit; ability to leverage existing commercial infrastructure Meaningful revenue and large market opportunity Facilitates international expansion; tax benefits Characteristics of a Myriad acquisition 1 2 3 4 |
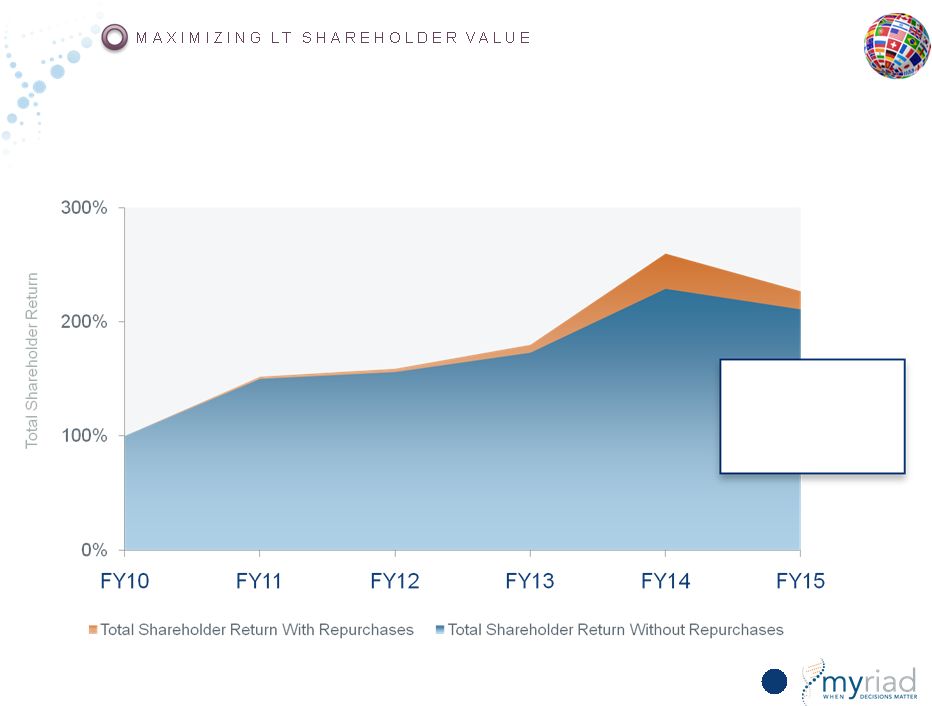 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Historical Share Repurchase Activity Has Increased Shareholder Returns 166 Increasing benefit in future as profitability grows |
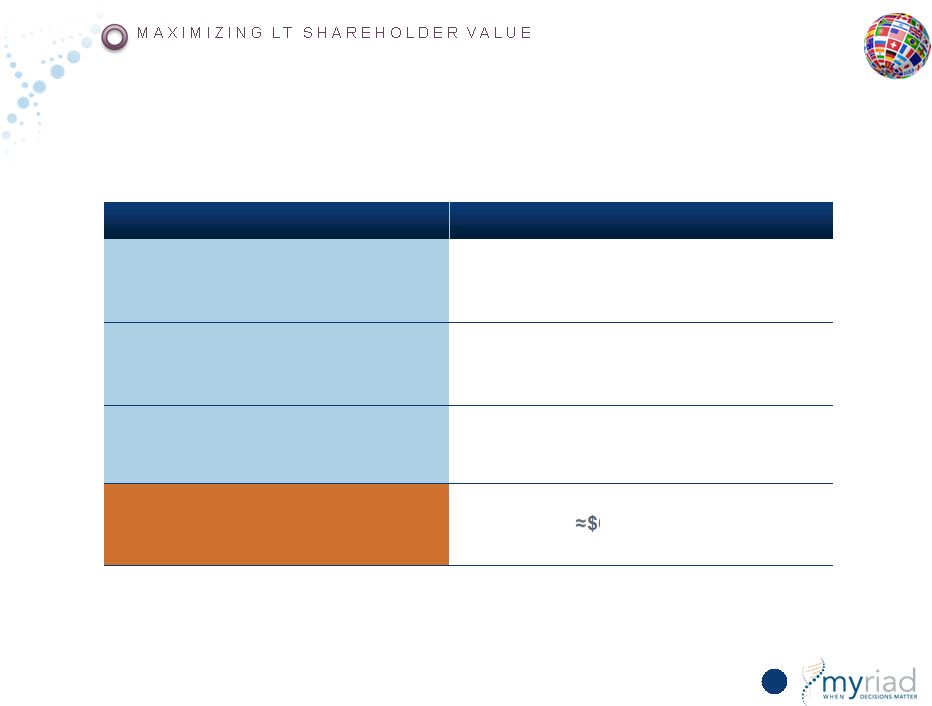 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Present Value Calculation Based on 5-Year Forecast – Supports Continued Repurchases 167 Variable Assumption Capital Deployment Share repurchases = FCF Discount Rate 15% P/E Terminal Multiple Used 20x Present Value $60 per share |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. 5-Year Outlook: Increased Growth and Financial Leverage 168 • Hereditary cancer growing low single digits • Significant diversification from product pipeline • International becomes larger contributor >30% Op Margins 7 Products >$50M >10% Revenue Growth CAGR >10% of Revenue from International drivers • Majority of investments are completed • Meaningful operating margin improvement as new products obtain reimbursement • Prioritize internal R&D • Pursue accretive M&A • Continue opportunistic share repurchase Maximizing LT Shareholder Value Revenue Growth Operating Leverage |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Closing Comments 169 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Million >$50 Products with Revenue 7 What was New Today? Our Strategic Goals 170 Revenue Growth CAGR >10% Operating Margin >30% International Revenue >10% |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. What was New Today? • Transition and Expand Hereditary Cancer – myRisk 80% conversion and 45% of revenue covered by long-term arrangements – Modeling demonstrates 5-year revenue CAGR = 3% revenue – Variant database now over 40,000 and will grow to 80,000 by FY2020 – Pricing floor based upon costs associated with high accuracy and complexity, extensive service and increased regulation 171 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. What was New Today? (continued) • Diversify the portfolio – 22 clinical studies with proprietary companion diagnostics for DNA damaging agents – Early access launch for myChoice HRD in Fall 2016 – Signed LabCorp agreement to increase access to Vectra DA – Prolaris’ unique active surveillance threshold facilitates value-based contracting with TUFTS Health Plan – Successful second validation for myPath Melanoma – Successful validation for myPlan Renal Cancer – myPath Bipolar demonstrates 96% AUC in training set; beginning enrollment in prospective study – Proprietary technology developed for cancer detection in urine 172 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. What was New Today? (continued) • Increase International Contribution – Revised strategy defines countries, reference tests, and kit products – RNA-based tests already under development with Thermo Fischer Scientific for kit strategy 173 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Worldwide Leader in Personalized Medicine • We are entering the golden age for personalized medicine • We are the pioneers of “research-based” and “education-centric” business modeling for diagnostics • No company is better positioned to lead this revolution in healthcare than Myriad • Our finest hour will be discovered in the days ahead 174 |
 Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. Copyright © 2015 Myriad Genetics, Inc., all rights reserved. www.Myriad.com. & 175 Q A |