| > | | Continued pressure on the price of medicines. |
| > | | Higher regulatory hurdles for new medicines and new indications. |
| > | | Competition from research-based and, increasingly, generic pharmaceutical companies. |
PRICING PRESSURE
The growing demand for healthcare means ever-increasing pressure on healthcare budgets and, whilst payers recognise the need to reward innovation, they have a duty to spend their limited financial resources wisely. Cost-containment, including pharmaceutical spending, therefore continues to be a fundamental consideration. The current global economic downturn is likely to further constrain healthcare providers and those patients who pay directly for their medicines, and additional challenges may arise if suppliers and distributors face credit-related difficulties.
The research-based pharmaceutical industry’s challenge is to manage the associated downward pressure on the price of its products, whilst continuing to invest in the discovery, development, manufacturing and marketing of new medicines.
Most of our sales are generated in highly regulated markets where governments exert various levels of control on price and reimbursement. The network of pricing systems creates a complex matrix that must be managed to optimise revenues. This may be further complicated by currency fluctuations within regions. The principal aspects of price regulation in the major markets are described more in the Geographical Review from page 48.
Payers also increasingly require demonstration of the economic as well as therapeutic value of medicines. Meeting these needs across a diverse range of national and local reimbursement systems requires significant additional resources.
REGULATORY REQUIREMENTS
The pharmaceutical industry is one of the most regulated of all industries and, whilst efforts to harmonise regulations globally are increasing, the number and impact of these regulations continue to grow. Regulatory drug review and approval is a complex and time consuming process, typically taking between six months and two years. In recent years, regulatory processes have become subject to more conditions including patient risk management plans, patient registries, post-marketing requirements, and conditional and limited approvals.
Traditional clinical trials designed to establish safety and efficacy remain a core component of drug development programmes but regulators are increasingly requiring that programmes also clearly demonstrate the benefits and risks of new medicine in the context of other available therapies, as well as demonstrating long-term medical outcomes, such as survival and quality of life improvements.
In addition to safety and efficacy, pre-approval regulation covers every aspect of the product including the chemical composition, manufacturing, quality controls, handling, packaging, labelling, distribution, promotion and marketing. Post approval and launch, all aspects relating to a product’s safety, efficacy and quality must continue to meet regulatory requirements. See also Ensuring Product Quality (page 27).
COMPETITION
Our main competitors are other international, research-based pharmaceutical companies that sell innovative, patent-protected, prescription medicines. Following patent expiry, our products also compete with generic pharmaceuticals. Since generic manufacturers do not bear the same high costs of R&D, nor do they typically invest as significantly in safety monitoring or marketing, they typically adopt lower prices for their products.
The generic industry is increasingly challenging innovators’ patents and in the US, the world’s largest pharmaceutical market, many leading medicines have faced or are facing patent challenges from generic manufacturers. The research-based industry is also experiencing increased challenges elsewhere in the world, for example in Europe, Canada, Asia and Latin America. It is increasingly complex to enforce patent rights and other intellectual property in certain markets, especially those where practices are in place to encourage broad access to medicines. While there are few established regulatory systems for biosimilars of biological products, several markets, including the US, are considering regulatory structures that might allow for an abbreviated marketing approval mechanism akin to that for generic pharmaceuticals. Further information about the risk of the early loss and expiry of patents is explained in the Intellectual Property section on page 26.
Competition also comes from collaborations and partnerships between traditional pharmaceutical companies and smaller biotechnology and vaccine companies. Increasingly, as pharmaceutical companies seek to expand their pipeline, they are able to gain access to promising new product candidates by partnering with these smaller companies that may lack some of the infrastructure for growth that a larger company can provide. Competition for high quality collaborations is increasingly fierce as the major pharmaceutical companies frequently focus on the same opportunities to enhance their in-house capabilities.
Further information about the principal risks and uncertainties we face can be found in the Risk section from page 74.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| > | | Despite a continually challenging environment, including generic pressure, combined sales ofArimidex,Crestor,Nexium,SeroquelandSymbicortup 5% in the US – 65% of our total US sales. |
| > | | Maintained market position as the second largest brand name pharmaceutical company in Canada. |
| > | | Solid sales performance in the Rest of World, up 5%. |
| > | | Strong brand performance in Western Europe but intense competition and governmental controls over healthcare expenditure. |
| > | | Strong volume growth from key products,Crestor,LosecandSeroquelin Japan. |
| > | | Emerging Rest of World delivers strong sales growth, up 16% with Emerging Europe sales up 10% and Emerging Asia sales up 10%. |
| > | | Continued expansion in China, including continued sales growth up 31%. |
| > | | EU Commission of a Sectoral Inquiry into the pharmaceutical industry continues, with a final report expected in Spring 2009. |
For more information regarding our products please refer to the relevant sections of the Therapy Area Review from page 53. Details of relevant continuing litigation can be found in Note 25 to the Financial Statements (from page 144) and details of relevant risks are set out in the Principal Risks and Uncertainties section (from page 76).
For the AstraZeneca definition of markets please see the Glossary on page 199.
US
Despite full generic competition toToprol-XLand the growth in generic omeprazole, sales in the US increased 1% in 2008 to $13,510 million (2007: $13,366 million). Combined sales ofArimidex,Crestor,Nexium,Seroquel, andSymbicortwere up 5% to $8,803 million (2007: $8,414 million) – 65% of our total US sales. AstraZeneca is currently the third largest pharmaceutical company in the US, with a 5.6% share of US prescription pharmaceutical sales. Sales for Aptium Oncology and Astra Tech fell by 2% and rose by 33% to $395 million (2007: $402 million) and $80 million (2007: $60 million), respectively.
Nexiumcontinues to lead the branded proton pump inhibitor (PPI) market for new prescriptions, total prescriptions and total capsules dispensed. Generic pantoprazole showed strong growth after being introduced late in 2007 and together with generic omeprazole captured most of the market growth, resulting in price and share erosion across the entire branded PPI market. In the face of generic pressure,Nexiumcontinued to fare better than its branded competitors with sales in 2008 down 8% to $3,101 million (2007: $3,383 million). During the year, the US Food and Drug Administration (FDA) approved the use ofNexiumin children ages one to 11 years old for the short-term treatment of gastroesophageal reflux disease.
Seroquelmaintained its strong position as the number one prescribed atypical anti-psychotic on the market, with sales up 5% to $3,015 million (2007: $2,863 million). Seroquelposted total prescription growth of 6.6% with an increase of one million prescriptions, outpacing the rate of market growth for anti-psychotics by almost two points, leading the market in absolute total prescription growth. During the year, the FDA approvedSeroquelfor the maintenance of bipolar disorder as adjunct therapy to lithium or divalproex. The FDA also approvedSeroquel XRfor the depressive episodes of bipolar disorder, the manic or mixed episodes associated with bipolar I disorder (as either monotherapy or adjunct therapy to lithium or divalproex), and for maintenance treatment of bipolar disorder as adjunct therapy to lithium or divalproex.
Supplemental new drug applications (sNDAs) were submitted to the FDA for use ofSeroquel XRin adult patients for major depressive disorder (MDD) and generalised anxiety disorder (GAD). In December 2008, we received a Complete Response Letter from the FDA related to the MDD submission, while the GAD submission remains under review. We also submitted an sNDA to the FDA for use ofSeroquelfor treatment of schizophrenia in 13 to 17 year olds and for treatment of acute manic episodes of bipolar I disorder for 10 to 17 year olds. The US Prescribing Information forSeroquel andSeroquel XRis being updated to include new safety information regarding use in children and adolescents.SeroquelandSeroquel XRare not approved currently for use in paediatric patients under 18 years of age.
Crestorsales were up 18% to $1,678 million (2007: $1,424 million) with a total prescription growth of 10.8%, and was the only branded statin to grow in total prescriptions throughout 2008 despite generic pressure. The new indication to slow the progression of atherosclerosis in adult patients with elevated cholesterol, an important differentiator from other products in the cholesterol-lowering market, was successfully introduced and awareness amongst physicians is high. Under the terms of an agreement executed in November 2008, Abbott obtained the non-exclusive right to promoteCrestoralongside AstraZeneca in the US (excluding Puerto Rico) increasingCrestor‘s profile and share of voice. New data presented in November 2008 from the JUPITER study demonstrated thatCrestor20mg significantly reduced major cardiovascular (CV) events – defined by the study as the combined risk of myocardial infarction, stroke, arterial revascularisation, hospitalisation for unstable angina, or death from CV causes – by 44% compared to placebo among men and women with elevated hsCRP (high-sensitivity C-reactive protein) but low to normal cholesterol levels. hsCRP is a recognised marker of inflammation that is associated with an increased risk of atherosclerotic CV events. The JUPITER results also showed that for patients in the trial takingCrestor, the combined risk of heart attack, stroke or CV death was reduced by nearly half. We expect to file a regulatory submission with the FDA that includes the JUPITER data in the first half of 2009 and, if approved, will begin promotional activities within the approved labelling.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
OUR FINANCIAL PERFORMANCE
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | 2008 | | 2007 | | 2006 | | 2008 compared to 2007 | | 2007 compared to 2006 | |
| | | | | | | | | | | Growth due | | | | | | | | | | | Growth due | | | | | | | | | | | | | | | | | |
| | | | | | | CER | | | to exchange | | | | | | | CER | | | to exchange | | | | | | | CER | | | Reported | | | CER | | | Reported | |
| | | Sales | | | growth | | | effect | | | Sales | | | growth | | | effect | | | Sales | | | growth | | | growth | | | growth | | | growth | |
| | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | % | | | % | | | % | | | % | |
| | |
| US | | | 13,510 | | | | 142 | | | | 2 | | | | 13,366 | | | | 917 | | | | – | | | | 12,449 | | | | 1 | | | | 1 | | | | 7 | | | | 7 | |
| | |
| Canada | | | 1,275 | | | | 95 | | | | 35 | | | | 1,145 | | | | 54 | | | | 60 | | | | 1,031 | | | | 8 | | | | 11 | | | | 5 | | | | 11 | |
| | |
North America | | | 14,785 | | | | 237 | | | | 37 | | | | 14,511 | | | | 971 | | | | 60 | | | | 13,480 | | | | 2 | | | | 2 | | | | 7 | | | | 8 | |
| | |
| Western Europe | | | 9,743 | | | | 55 | | | | 573 | | | | 9,115 | | | | 282 | | | | 760 | | | | 8,073 | | | | 1 | | | | 7 | | | | 3 | | | | 13 | |
| | |
| Japan | | | 1,957 | | | | 73 | | | | 223 | | | | 1,661 | | | | 170 | | | | (12 | ) | | | 1,503 | | | | 4 | | | | 18 | | | | 11 | | | | 11 | |
| | |
| Other Established ROW | | | 843 | | | | 107 | | | | 21 | | | | 715 | | | | 83 | | | | 77 | | | | 555 | | | | 15 | | | | 18 | | | | 15 | | | | 29 | |
| | |
Established ROW | | | 12,543 | | | | 235 | | | | 817 | | | | 11,491 | | | | 535 | | | | 825 | | | | 10,131 | | | | 2 | | | | 9 | | | | 5 | | | | 13 | |
| | |
| Emerging Europe | | | 1,215 | | | | 102 | | | | 85 | | | | 1,028 | | | | 102 | | | | 95 | | | | 831 | | | | 10 | | | | 18 | | | | 12 | | | | 24 | |
| | |
| China | | | 627 | | | | 136 | | | | 54 | | | | 437 | | | | 91 | | | | 18 | | | | 328 | | | | 31 | | | | 43 | | | | 28 | | | | 33 | |
| | |
| Emerging Asia Pacific | | | 802 | | | | 72 | | | | (19 | ) | | | 749 | | | | 62 | | | | 41 | | | | 646 | | | | 10 | | | | 7 | | | | 10 | | | | 16 | |
| | |
| Other Emerging ROW | | | 1,629 | | | | 247 | | | | 39 | | | | 1,343 | | | | 223 | | | | 61 | | | | 1,059 | | | | 18 | | | | 21 | | | | 21 | | | | 27 | |
| | |
Emerging ROW | | | 4,273 | | | | 557 | | | | 159 | | | | 3,557 | | | | 478 | | | | 215 | | | | 2,864 | | | | 16 | | | | 20 | | | | 17 | | | | 24 | |
| | |
Total sales | | | 31,601 | | | | 1,029 | | | | 1,013 | | | | 29,559 | | | | 1,984 | | | | 1,100 | | | | 26,475 | | | | 3 | | | | 7 | | | | 7 | | | | 12 | |
| | |
In another agreement with Abbott, we are investigating the fixed dose combination of the active ingredients inCrestor(rosuvastatin calcium) and Trilipix™ (fenofibric acid) for the treatment of mixed dyslipidaemia. A Phase III trial in 2008 demonstrated that a combination of rosuvastatin calcium and fenofibric acid delivers greater improvements in treating all three key lipids (LDL, HDL and triglycerides) than the pre-specified monotherapy comparators. Currently, dyslipidaemia affects more than 100 million US residents and has been shown to play a pivotal role in the development of atherosclerosis and consequently, cardiovascular disease. Patients with mixed dyslipidaemia are expected to become more prominent segments of the dyslipidaemic population. Abbott obtained approval of Trilipix™ in December 2008 as the first and only fibrate labelled for use with a statin.
To maximise the value ofMerrem IVduring the year we announced an agreement with Cubist, who will provide promotional and scientific affairs support forMerrem IVin the US and Puerto Rico.
Arimidexcontinued to perform well with sales up 9% to $754 million (2007: $694 million) for the full year. Arimidexcontinues to be the market leader in new prescriptions for branded hormonal treatments for breast cancer in the US.
In September 2008, an additional six-month period of exclusivity was granted to marketCasodexfor its licensed advanced prostate cancer indication until 1 April 2009.
Pulmicort Respules, the only inhaled corticosteroid for the treatment of asthma approved in the US for children as young as 12 months, showed strong sales growth with sales up 2% to $982 million (2007: $964 million). On 23 September 2008, the US District Court for the District of New Jersey denied a motion filed by Teva Pharmaceuticals Ltd. for summary judgment of no infringement in thePulmicort Respulespatent litigation. On 19 November 2008, the same court awarded a temporary restraining order against Teva Pharmaceuticals after Teva launched its generic product ‘at risk’ on 18 November 2008. On 25 November 2008, the parties settled the matter and AstraZeneca granted Teva a licence to launch its generic product in late 2009.
In its first full year after launch in June 2007,Symbicort Rapihaler(pMDI) continued to deliver steady growth with sales up 410% to $255 million (2007: $50 million). Widespread physician experience and growing appreciation of the differentiating feature of control plus fast onset has led to the product surpassing a 10% new prescription share of the inhaled corticosteroid/long acting beta agonist market.Symbicortis now prescribed to one in five of all patients that are new to combination therapy.
In 2008, two sNDAs were submitted to the FDA: one for the use ofSymbicortin chronic obstructive pulmonary disease and another for its use in paediatric asthma for ages six to 12. In October 2008, the pMDI device was enhanced with an actuation counter.
Synagisis the only FDA-approved monoclonal antibody (MAb) to help protect high-risk babies against severe Respiratory Syncytial Virus (RSV) disease. In its first full year in AstraZeneca, sales in the US were $923 million.
In 2008, distribution agreements continued with Par Pharmaceutical for all available strengths of generic metoprolol succinate. Also, Ranbaxy Pharmaceuticals began distribution of authorised generics of both felodipine and 40mg omeprazole.
Currently, there is no direct government control of prices for commercial prescription drug sales in the US. However, some publicly funded programmes – such as Medicaid and TRICARE (Department of Veterans Affairs) – have statutorily mandated rebates that have the effect of price controls for these programmes. Additionally, pressure on pricing, availability and utilisation of prescription drugs for both commercial and public payers continues to increase, driven by, among other things, an increased focus on generic alternatives. Primary drivers of increased generic use are budgetary policies within healthcare systems and providers, including the use of “generics only” formularies, and increases in patient co-insurance or co-payments. While it is unlikely that there will be widespread adoption of a broad national price control scheme in the near term, there will continue to be increased attention to pharmaceutical prices and their impact on healthcare costs for the foreseeable future.
ASTRAZENECA ANNUAL REPORT AND FOR M 20-F INFORMATION 2008
In the current political climate, policymakers are likely to consider healthcare reform a top priority. The reauthorisation of the State Childrens’ Health Insurance Program (SCHIP), a joint federal-state programme to expand healthcare coverage (including prescription drug coverage) for qualifying children, is poised to be one of the first healthcare reform proposals debated in the 111th Congress. A sustained focus on containing prescription drug costs is also likely, which could include proposals to allow the government to negotiate Medicare Part D prices directly with the pharmaceutical industry, increase manufacturers’ Medicaid drug rebate payments under the Medicaid drug rebate statute, and/or expand Medicaid rebates for patients who qualify for both Medicaid and Medicare (so-called ‘dual eligibles’). Additionally, there could be efforts to pass legislation implementing comparative effectiveness research requirements and/or legislation allowing for the commercial importation of drugs into the US from selected countries by certain individual consumers, pharmacies and drug wholesalers. Finally, proposals that would require disclosure of payments to healthcare professionals (eg for speaker contracts) are also being considered at the state and federal levels.
In its third year of operation, the Medicare Part D prescription drug programme maintained high levels of enrollment and beneficiary satisfaction, achieved prescription volume growth similar to other mature markets and provided access to our medicines for a large segment of the patient population. Through the AZ&Me Prescription Savings Programme for Patients with Medicare Part D, AstraZeneca provides prescription access to financially needy Medicare D beneficiaries. Although difficult to quantify, Medicare Part D has had an indirect effect on pricing in the broader US market. Despite the pricing challenges, overall access in key accounts was maintained or improved in 2008. It is difficult to predict fully the longer-term effects of this initiative on our business.
We continue to support My Medicare Matters, a community based outreach and education programme, in partnership with the National Council on Aging. Funding from AstraZeneca also supports MyMedicareCommunity.org, an on-line community for grass roots organisations serving people with Medicare. During 2007 and 2008, we supported a pilot grant programme focused on testing new approaches to finding and enrolling eligible people in the Medicare’s Low-Income Subsidy (LIS) programme. Over 40,000 LIS applications were submitted as a result of these demonstration projects.
Additionally, AstraZeneca has been providing patient assistance to the uninsured for 30 years and, in the last six years, has provided more than $3 billion in savings to more than one million patients in the US and Puerto Rico. Last year alone, we provided more than $612 million in savings to approximately 440,000 people without drug coverage (approximately 2.7 million prescriptions).
CANADA
Despite the entry of the generic forms ofSeroquel IR, sales in Canada increased by 8% (+11% reported) to $1,275 million (2007: $1,145 million). Combined sales ofCrestor,Nexium,SeroquelandAtacandwere up 18% to over $864 million (2007: $713 million) withCrestor,SeroquelandNexiumamong the top 20 prescription products in Canada by sales.
We remain the second largest brand name pharmaceutical company in Canada.Crestormaintained its number two ranking in the statin market and was the fastest-growing product in both new and total prescription segments (25.9% and 32.0% growth respectively).Crestoris also the third largest product in Canada by sales. Together,Seroquel XRandSeroquel IRremain the leaders in new and total prescriptions within the atypical anti-psychotics market.Atacandcontinues to outperform the anti-hypertensive market, with total prescription growth of over 15.0% compared with market growth of only 7.4%. Several key regulatory approvals were achieved in Canada in 2008. Canada was the first country in which we gained regulatory approval forSeroquel XRfor the treatment of bipolar mania, withSeroquel XRandSeroquel IRalso approved for the treatment of bipolar depression (approvals were received eight months and five months respectively ahead of standard Health Canada review timelines). In addition, a new tablet strength forAtacand(32mg) was approved by Health Canada.
Organisational efficiencies were gained with the closure of the Canadian packaging plant and transfer of product packaging to the Newark, Delaware facility, and further efficiencies were obtained through the movement to common North American technology platforms.
The Canadian government has instituted a Health Technology Assessment appraisal system through their Common Drug review process which rejects almost six out of 10 new medications. The Patented Medicine Prices Review Board has the role of setting
the maximum non-excessive price in the market. For patients to gain optimal access to medicines, they then need to be listed on provincial formularies. This long process means patients in Canada can typically wait over two years following the regulatory approval for access to be granted.
The different provinces have adopted different approaches to pharmaceutical funding, from one end of the continuum in Quebec with more open access to more restricted access, therapeutic substitution and price tendering on the horizon in British Colombia. The trend in Canada indicates provinces will increase their access restrictions and drive prices down while the complex reimbursement system will continue to result in access delays.
Sales in the rest of the world performed strongly in 2008, up 5% (+12% reported) to $16,816 million (2007: $15,048 million). Key products (Arimidex,Crestor,Nexium,SeroquelandSymbicort) delivered a strong performance, up 14% (+20% reported) with sales of $7,413 million (2007: $6,189 million). Latin America, the Middle East and Africa and Asia Pacific regions delivered particularly strong sales, up 13% (+19% reported) with sales of $5,858 million (2007: $4,906 million).
ESTABLISHED REST OF WORLD
Sales in our Established Rest of World Markets increased by 2% (+9% reported), with good growth fromCrestor,SeroquelandSymbicortand our oncology products, together withSynagis,offsetting declines in sales of our proton pump inhibitors in Western Europe.
WESTERN EUROPE
In Western Europe, we saw a flat market with overall growth of 1% (+7% reported). This reflected decreasing sales in France (down 1%, +7% reported), Germany (down 2%, +6% reported), Italy (down 6%, +2% reported) and Spain (down 8%, -1% reported), partly offset by strong growth in the UK (up 8%, +2% reported). Sales in established European markets were mainly impacted by the loss of patent/marketing exclusivity onCasodex, by government initiatives to contain drug expenditures and by the loss of sales due to an ageing portfolio of mature brands. These impacts were partly offset by continued strong performance of key products (mainlyCrestorandSeroquel).
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
We have continued with our programme of resource management in Western Europe and reduced costs by $159 million and headcount by 618 during 2008.
Overall our sales in France were down 1% (+7% reported) to $1,922 million (2007: $1,794 million). The strong performance ofCrestorandNexium, which gained significant market share from competitors, was offset by the loss of patent/marketing exclusivity expiry forCasodex.
In Germany, sales were down 2% (+6% reported) to $1,307 million (2007: $1,233 million), mainly due to theCasodexpatent/marketing exclusivity expiry and the government restriction on access toNexiumleading to a reduction in sales of 34% over last year. Seroquelcontinued to grow well with 27% growth (+38% reported) reaching 29.5% of the market for atypical anti-psychotics.
In the UK, sales were up 8% (+2% reported) to $1,020 million (2007: $1,004 million) driven bySymbicort(+34%, +25% reported),Seroquel(+32%, +22% reported), andArimidex(+8%, +1% reported). Many of our other brands also performed well withMerrem(+13%, +6% reported) being of particular note. Competition remained intense but our key brands gained market share in their respective segments. Especially strong wereSeroquelandSymbicortachieving gains of 2.4 and 1.3 percentage points respectively. The UK Government and pharmaceutical industry have entered into ‘terms of reference’ discussions concerning potential changes to the pricing and reimbursement scheme. The impact of these changes is likely to be $90 million in 2009.
In Italy,Crestorperformed strongly increasing its sales by 12% (+22% reported). The specialty care brands also showed good growth withSeroquelincreasing sales by 19% (+28% reported) with 32.9% of the market for atypical anti-psychotics andArimidexincreasing sales by 12% (+21% reported) with 32.0% of the market for aromatase inhibitors and tamoxifen. However, overall sales declined by 6% (+2% reported) to $1,323 million (2007: $1,294 million) as a result of reference pricing at the regional level on PPIs and measures to control their prescribing by physicians, combined withCasodexpatent/marketing exclusivity expiry.
In Spain, sales were down 8% (-1% reported) to $863 million (2007: $868 million) due toSymbicort (-7%, +1% reported) and generic competition forCasodexandArimidex.
Synagissales outside the US are undertaken through a subsidiary of Abbott Laboratories with revenue of $307 million ($169 million in the seven months from June to December 2007). We estimate that about 36% of sales arise in Western Europe, about 32% in Japan and over 7% in Canada. Strong growth has been recorded in Latin America in 2008.
Most governments in Europe directly intervene to control the price and reimbursement of medicines. The decision-making power of prescribers in Europe has been eroded in favour of a diverse range of payers. While the systems to control pharmaceutical spending vary, they all have had a noticeable negative impact on the uptake and availability of innovative medicines. Several governments have imposed price reductions and increased the use of generic medicines as part of healthcare expenditure control. Several countries are applying strict tests of cost-effectiveness of medicines, which has reduced access of European patients to medicines in areas of high unmet need. These and other measures all contribute to an increasingly tough environment for branded pharmaceuticals in Europe. However, the anticipated radical change in the UK pharmaceutical system towards direct government control of prices was abandoned. Parallel trading of branded pharmaceuticals continues to challenge the European pharmaceutical market; a report commissioned for the EU Commission acknowledged this and also highlighted the negative impact of parallel trading on patient safety.
In January 2008 AstraZeneca, together with several other companies, was the subject of an unannounced inspection simultaneous with the launch by the EU Commission (Commission) of a Sectoral Inquiry (Inquiry) into the pharmaceutical industry. The Inquiry relates to the introduction of innovative and generic medicines and covers commercial and other practices, including the use of patents. On 28 November 2008 the Commission published its preliminary report. The report does not identify wrongdoing by any individual companies but is stated to provide a factual basis for further consideration. The Commission has stated that it will commence individual investigations where there are indications that competition rules have been breached. The preliminary report focuses on a number of issues relating to competition in the EU, referring to strategies which the Commission believes pharmaceutical companies use to block or delay generic entry. Such strategies include: patent filings and enforcement;
patent settlement agreements and other agreements; interventions before national regulatory authorities; and life-cycle management strategies.
A final report is expected in Spring 2009. AstraZeneca has been co-operating fully with the Commission and participating in European Federation for Pharmaceutical Industries and Associations activities.
JAPAN
In Japan, we were the fifth fastest-growing company amongst the top 15 pharmaceutical companies, maintaining our market ranking of number 12 in 2008. Strong volume growth from key products offset the biennial government review of downward drug prices to deliver sales up 4% (+18% reported) to $1,957 million (2007: $1,661 million). The key drivers of growth being the continued success ofCrestor(+93%, +118% reported), the continued growth ofLosec(+5%, +18% reported) and the increased penetration ofSeroquel(+10%, +24% reported).
In Japan, there is formal central government control of prices by the Ministry of Health, Labour and Welfare (MHLW) and the pricing and reimbursement system has remained largely stable in the last few years. As expected, pharmaceuticals were subject to price reductions in April 2008. The long-term objective of the Japanese government is to raise generic volume share from 18.7% in 2007 to 30% by 2012; recent reforms have supported this goal by making substitution of a generic product for a branded product easier.
In 2008, the MHLW continued their push towards the acceptance of non-Japanese Asian data as part of the regulatory approval package for Japanese patients. Despite increasing budgetary pressures associated with an ageing population, they also publicly recognised the importance of the pharmaceutical industry and their own drive to reward innovation better in the future.
OTHER ESTABLISHED REST OF WORLD
In Australia and New Zealand, we delivered a strong sales performance with sales up by 15% (+18% reported) to $843 million (2007: $715 million). Both our primary care and specialist care portfolios continued to grow, driven mainly by sales growth forCrestor,AtacandandNexiumin primary care and bySeroquelandArimidexin specialist care. On a CER basis, these five brands,Arimidex,Crestor,Seroquel,AtacandandNexium, grew by 33% (+37% reported).
ASTRAZENECA ANNUAL REPORT AND FOR M 20-F INFORMATION 2008
EMERGING REST OF WORLD
In the Emerging Rest of World regions, sales increased 16% (+20% reported) to $4,273 million for the full year (2007: $3,557 million), accounting for nearly 63% of total sales growth outside the US. Sales in Emerging Europe were up 10% (+18% reported) to $1,215 million (2007: $1,028 million). Sales in China increased 31% (+43% reported) to $627 million (2007: $437 million) and sales in Emerging Asia Pacific increased 10% (+7% reported) to $802 million (2007: $749 million).
As the pharmaceutical markets in Asia Pacific, Latin America and elsewhere develop, reforms in pricing and reimbursement will inevitably follow. As these markets become more important to our business, we have to consider carefully such factors when we develop brands. In many of the major markets, such as China, Brazil and Mexico, the patient pays directly for prescription medicines, and this will be an increasingly important issue for our business. Other growing markets, such as South Korea and Turkey are seeing more direct government intervention in pricing and reimbursement, more in line with the systems in Europe, Canada and Australia.
EMERGING EUROPE
Russia continued to enjoy strong sales growth driven bySymbicort,MerremandCrestorin 2008. Our business in Romania performed particularly well, almost doubling its size, primarily driven bySeroquel,NexiumandCrestor. Our continued expansion included the establishment of a local marketing presence in Ukraine and Kazakhstan.
CHINA
In China, in line with our growth and expansion strategy of the past four years, we have continued to build our presence and sales (including Hong Kong) were up 31% (+43% reported) to $627 million (2007: $437 million). We are the largest multinational pharmaceutical company in the prescription market in China, as surveyed by the Hong Kong Association of the Pharmaceutical Industry, with a growth rate for prescription sales of 28.8% (+40.2% reported). Our investment in China increased with further growth in the number of sales representatives, and continued to support our innovation discovery research centre in Shanghai and our several external collaborations, including a new clinical pharmacology unit in Peking University and a translational science laboratory in Guangdong Province People’s Hospital.
EMERGING ASIA PACIFIC
In the rest of the Emerging Asia Pacific region, overall sales were up 10% (+7% reported) to $802 million (2007: $749 million) by achieving strong growth in India, Indonesia, Singapore, Thailand and Vietnam, where market dynamics continue to be positive.
LATIN AMERICA
Latin America enjoyed strong sales performance up 18% (+22% reported) to $1,159 million (2007: $947 million), mainly driven by Mexico, Brazil, Venezuela, Central America and the Caribbean. As a result, our market share grew to 3% in the prescription market, improving our position from tenth to eighth in the prescription market ranking.
This reflects the investment made to develop our key products in fast-growing markets.Atacand,Crestor,Nexium,SeroquelandSymbicortall showed strong performance with overall sales up 33% (+38% reported) to $516 million (2007: $372 million). Nexiumis our best selling prescription product in Latin America with overall sales up 25% (+28% reported) to $185 million (2007: $144 million) and the fifth best selling prescription product in the Latin American market.Crestoris now our second best selling prescription product with overall sales up 46% (+52% reported) to $128 million (2007: $84 million) and is the eleventh best selling prescription product in the Latin American market.
Our top three largest markets in the region are now Brazil, Mexico and Venezuela. Brazil sales were up 21% (+33% reported) to $440 million (2007: $330 million), Mexico sales were up 6% (+6% reported) to $353 million (2007: $334 million) and Venezuela sales were up 37% (+37% reported) to $142 million (2007: $103 million).
MIDDLE EAST AND AFRICA
In 2008, the region again delivered a very strong double-digit growth, driven mainly byAtacand,Crestor,SeroquelandSymbicort, with particularly good performances in Gulf States, Levant, Egypt, South Africa and Maghreb. We have recently established a new marketing company in Israel as part of our investment plan in the region.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| > | | Crestorsales up 26% to $3.6 billion andCrestor is now approved in every EU country. |
| > | | Crestorstudy demonstrates significant reduction in major cardiovascular events (44% compared to placebo in men and women with elevated hsCRP but low/normal cholesterol levels). |
| > | | Atacandsales up 10% to $1.5 billion. |
| > | | Worldwide collaboration with Bristol-Myers Squibb to develop and commercialise dapagliflozin expanded to include Japan. |
| > | | US submission for fixed dose combination ofCrestorand Abbott’s Trilipix™, for the treatment of mixed dyslypidaemia, anticipated for third quarter 2009. |
| > | | Toprol-XLUS sales down 70% for the full year. |
OUR MARKETED PRODUCTS
Crestor1 (rosuvastatin calcium) is a statin for the treatment of dyslipidaemia and hypercholesterolemia, and to slow the progression of atherosclerosis.
Atacand2 (candesartan cilexetil) is an angiotensin II antagonist for the first-line treatment of hypertension and symptomatic heart failure.
Seloken/Toprol-XL(metoprolol succinate) is a once daily tablet for 24-hour control of hypertension and for use in heart failure and angina.
Tenormin(atenolol) is a cardioselective beta-blocker for hypertension, angina pectoris and other cardiovascular disorders.
Zestril3 (lisinopril dihydrate), an ACE inhibitor, is used for the treatment of a wide range of cardiovascular diseases, including hypertension.
Plendil(felodipine) is a calcium antagonist for the treatment of hypertension and angina.
| |
| 1 | Licensed from Shionogi & Co. Ltd. |
| |
| 2 | Licensed from Takeda Chemicals Industries Ltd. |
| |
| 3 | Licensed from Merck & Co., Inc. |
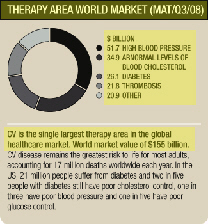
Backed by over 40 years’ experience, AstraZeneca is a world leader in cardiovascular (CV) medicines. We aim to build on our strong position, focusing on the growth areas of atherosclerosis (hardening of the arteries), thrombosis (blood clotting), diabetes and atrial fibrillation.
HYPERTENSION, ATHEROSCLEROSIS AND DYSLIPIDAEMIA
High blood pressure (hypertension) and abnormal levels of blood cholesterol (dyslipidaemia) are well known to damage the arterial wall and thereby lead to atherosclerosis. CV events driven by atherosclerotic disease remain the leading cause of death in the western world. Lipid-modifying therapy, primarily statins, is a cornerstone of treating atherosclerotic risk. Within the lipid-modifying market, generics are taking a significant share of the market and it is anticipated that generic atorvastatin will be available late 2011. Recent studies of some competitor products created uncertainty about clinical efficacy leading to reduced sales of these products, whilst AstraZeneca’s study (see below) provided positive data on the effect of rosuvastatin.
OUR FOCUS
Our key marketed products
Since its launch in 2003, our statin,Crestor, has continued to gain market share, based on its differentiated profile in managing cholesterol levels and its unique recent label indication for treating atherosclerotic disease. Following new approvals during 2008 in Germany, Spain, Poland, Norway and Malta,Crestoris now approved in every EU country.
Less than half of the people thought to have high levels of low-density lipoprotein cholesterol (LDL-C) ‘bad cholesterol’ get diagnosed and treated and of those people, only about half reach their physician’s recommended cholesterol target using existing treatments.Crestoris the most effective statin in lowering LDL-C and the majority of patients reach their LDL-C goals using the usual 10mg starting dose.Crestoralso produces an increase in high-density lipoprotein cholesterol (HDL-C) ‘good cholesterol’, across a range of doses. At its usual 10mg starting dose,Crestorhas been shown, versus placebo, to reduce LDL-C by up to 52% and raise HDL-C by up to 14% with eight out of 10 patients reaching their lipid goals.
In the US,Crestoris also approved for use as an adjunct to diet for slowing the progression of atherosclerosis in patients with elevated cholesterol.Crestoris the only statin with an
atherosclerosis indication in the US which is not limited by disease severity or restricted to patients with coronary heart disease.
Atacand, first launched in 1997, is approved for the treatment of hypertension in over 100 countries and for symptomatic heart failure in over 70 countries. Angiotensin II antagonists are the fastest growing sector of the global hypertension market. Available as a once a day tablet, launches of the 32mg dosage strength outside the US continued during the year, and this dosage is now available in most Established Markets. In July 2008, we sought approval in Europe for two dose strengths ofAtacand Plus(candesartan cilexetil/hydrochlorothiazide) which is a fixed combination ofAtacandand the diuretic hydrochlorothiazide (HCTZ), indicated for the treatment of hypertension in patients who need more than monotherapy.
Clinical trial developments
GALAXY, our long-term global clinical research programme forCrestorinvestigating links between optimal lipid control, atherosclerosis and CV morbidity and mortality, has completed a number of studies involving over 69,000 patients in over 55 countries.
Data from the latest study, JUPITER, published in November 2008, demonstrated thatCrestor20mg significantly reduced major CV events (defined in this study as the combined risk of myocardial infarction, stroke, arterial revascularisation, hospitalisation for unstable angina, or death from CV causes) by 44% compared to placebo among men and women with elevated high-sensitivity C-reactive protein (hsCRP) (and other risk factors) but low to normal cholesterol levels. Results also showed that for patients takingCrestor, the combined risk of heart attack, stroke or CV death was reduced by nearly half, risk of heart attack was cut by more than half, risk of stroke was cut by nearly half and total mortality was significantly reduced by 20%.Crestor20mg was well tolerated in nearly 9,000 patients during the course of the study. There was no difference between treatment groups for major adverse events, including cancer or myopathy. There was a small increase in physician reported diabetes consistent with data from other large placebo controlled statin trials.
GISSI-HF, an investigator sponsored study published in September 2008, evaluatedCrestor10mg and placebo in a heart failure population and confirmed the results of our CORONA study in showing no difference between the treatments in the primary endpoints of death or CV hospitalisation in patients with heart failure, over and above optimised heart failure treatment. Both studies
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
ARRHYTHMIA AND THROMBOSIS
Atrial fibrillation (AF) is the most common cardiac arrhythmia. Rhythm-control therapy to control the symptoms of AF is dominated by generic amiodarone, which is effective at maintaining patients in normal heart rhythm, but very poorly tolerated. There remains an unmet need for a safe and tolerated therapy with effective symptom relief. Two competitor products are in late development for use in AF and recent data from an outcome study of one of them versus placebo in AF patients showed clinical benefit in addition to symptom relief – the first time for an anti-arrhythmic agent.
Patients surviving an acute coronary event are at increased risk from further thrombosis and treatment guidelines advocate anti-platelet therapy. New guidelines issued in 2007 by the European Society of Cardiology for the treatment of acute coronary syndrome (ACS), have highlighted the negative consequences of drug induced bleeding in conjunction with the treatment of ACS, reinforcing the need for new anti-thrombosis drugs with acceptable bleeding risk.
During the year, two new anti-coagulants (dabigatran and rivaroxaban) were approved in Europe for use in prevention of deep vein thrombosis in conjunction with orthopaedic surgery. No Phase III data are yet available for the ability of new anti-coagulants to prevent strokes in AF, the major chronic indication for anti-coagulants, without the risks and repeated monitoring of warfarin or other vitamin K antagonists.
OUR FOCUS
In the pipeline
Brilinta(ticagrelor AZD6140), the first reversible, oral, adenosine diphosphate (ADP) receptor antagonist, is being developed to reduce the risk of blood clots and thrombotic events in patients diagnosed with ACS. Ticagrelor is currently being studied in the Phase III PLATO clinical trial, involving over 18,000 ACS patients in 43 countries, to determine if it is superior to clopidogrel for reducing the risk of thrombotic events in ACS patients.
The effectiveness of AZD0837 (an oral, direct thrombin inhibitor) in preventing strokes and other embolic events in AF patients will be studied in more than 35 countries, using a once-daily extended release formulation that provides sustained anti-coagulation effect throughout the dosing interval. We anticipate starting these Phase III studies in 2009.
Our lead compound in the treatment of AF is AZD1305, a combined ion channel blocker, which has progressed into Phase IIa testing in both the IV and oral form.
In December 2007, we filed patent infringement actions against seven generic drug manufacturers in the US following receipt of notices of their intent to market generic copies ofCrestorbefore the 2016 expiry of our licensed patent covering the active ingredient inCrestor. In July 2008, we filed a patent infringement action against Teva Pharmaceuticals in the US following receipt of its notice of its intent to market generic copies ofCrestorbefore the 2016 expiry of our licensed patent covering the active ingredient inCrestor. These eight cases are proceeding as a consolidated action in US District Court, District of Delaware.
Also in the US, Teva Pharmaceuticals (Teva’s Israeli parent company) filed a patent infringement lawsuit concerningCrestoron 6 October 2008. Teva alleges thatCrestortablets infringe a recently re-issued Teva US patent that claims stabilised pharmaceutical compounds.
AstraZeneca has full confidence in itsCrestorproduct and the intellectual property protecting it, and will vigorously defend and enforce it.
Further information is set out in Note 25 to the Financial Statements on page 148.
FINANCIAL PERFORMANCE 2008/2007
PERFORMANCE 2008
Reported performance
CV sales were up 4% as reported to $6,963 million (2007: $6,686 million). Strong growth fromCrestor, fuelled by the promotion of the atherosclerosis indication and increased sales ofAtacand offset the continuing significant declines inSeloken/Toprol-XL.
Performance – CER growth rates
CV sales were unchanged from 2007 at CER.Crestorsales increased by 26% to $3,597 million. US sales forCrestorfor the full year increased by 18% to $1,678 million. Crestortotal prescription share in the US statin market increased to 9.9% in December 2008 from 8.6% in December 2007, and was the only branded statin to gain market share.Crestorsales in the Rest of World were up 34% for the full year to $1,919 million, over half of global sales for the product. Sales were up 16% in Western Europe to $836 million and 93% in Japan driving sales growth in the Established Markets and the Rest of World up 33% in total. Sales in Emerging Markets increased by 41%.
Toprol-XLand authorised generic sales of the drug in the US were down 70% for the full year to $295 million. For the full year,Selokensales in the Rest of World were up 1% to $512 million.
US sales forAtacandfor the full year increased 1% to $262 million. Sales in other markets were up 12% to $1,209 million, on a 10% increase in Established Markets and an 18% increase in Emerging Markets.
PERFORMANCE 2007
Reported performance
CV sales rose by 9% from $6,118 million in 2006 to $6,686 million in 2007. Continued strong growth fromCrestormore than offset the significant declines inSeloken/Toprol-XL.
Performance – CER growth rates
CV sales grew by 5% at CER.Crestorsales increased by 33% to $2,796 million. In the US,Crestor sales for the full year were $1,424 million, a 24% increase over 2006. Total prescriptions in the US statin market increased 8% for the year;Crestorprescriptions were up 22%. Sales outside the US for the full year increased 45% to $1,372 million, nearly half the total worldwide sales for the product. Sales were up 26% in Western Europe with good growth in France and Italy. Sales in Canada increased 43%.
Global sales ofSeloken/Toprol-XLfell by 22% to $1,438 million. US sales of theToprol-XLproduct range, which includes sales of the authorised generic were down 30% for the full year, as the full range of dosage strengths were subject to generic competition from August 2007. Sales ofSelokenin other markets were up 5% for the full year as a result of growth in Emerging Markets.
Atacandsales in the US were unchanged for the full year whilst sales in other markets increased 12%.
Continued small declines were seen inZestril(down 10% to $295 million) andPlendil(down 7% to $271 million), with general global falls compensated by increases in discrete markets.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| > | | Sales ofNexium$5.2 billion, down 2%. |
| > | | Nexiumsubmissions in the EU for the short-term maintenance of haemostasis and prevention of re-bleeding in patients with peptic ulcer bleeding following therapeutic endoscopy and in the US for use in patients with peptic ulcer bleeding following therapeutic endoscopy. |
| > | | In late 2008, a Complete Response Letter received from the FDA in connection with ourNexiumsubmission for peptic ulcer bleeding. |
| > | | Losec/Prilosec sales $1.05 billion declining in the EU and US due to continuous generic pressure including the recent patent expiry in Italy. Overall sales down 14%; Japan sales still growing, up 5%. |
| > | | Settlement of patent litigation in the US brought by AstraZeneca against Ranbaxy, with enforceability of disputedNexiumpatents conceded and an agreement for licensed sales of genericNexiumfrom May 2014. |
| > | | Other patent litigation continuing in the US against generic manufacturers following abbreviated new drug applications relating toNexium. |
Nexium(esomeprazole) is the first proton pump inhibitor (PPI) for the treatment of acid-related diseases to offer clinical improvements over other PPIs and other treatments.
Losec/Prilosec(omeprazole) is used for the short-term and long-term treatment of acid-related diseases.
Entocort(budesonide) is a locally acting corticosteroid for the treatment of inflammatory bowel disease (IBD).
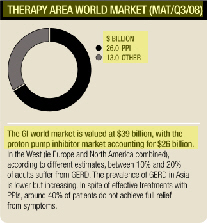
We aim to maintain our strong position in gastrointestinal (GI) treatments by continuing to focus on PPIs. NewNexiumline extensions include prevention of re-bleeding in patients with peptic ulcer bleeding, and prevention of low dose aspirin associated peptic ulcer. Our research and development is focused on finding new, innovative ways for treating acid reflux related disease.
GASTRO-OESOPHAGEAL
REFLUX DISEASE (GERD)
OUR FOCUS
Our key marketed products
Nexiumis an effective, long-term therapy for patients with GERD. For the treatment of active peptic ulcer disease, seven-dayNexiumtriple therapy (in combination with two antibiotics for the eradication of H.pylori) heals most patients without the need for follow-up anti-secretory therapy. Since it was first launched in 2000,Nexiumhas been used in the treatment of acid-related diseases in over one billion patient treatments.
Nexiumis available in approximately 100 countries for the treatment of acid-related diseases. In the US and EU,Nexiumis also approved for the treatment of children aged 12 to 17 years with GERD and in 2008 was approved for use in these countries in children aged one to 11 years old.Nexiumis also approved in the US, the EU, Canada and Australia for the treatment of patients with the rare gastric disorder, Zollinger-Ellison syndrome. In Europe,Nexiumis approved for the healing and prevention of ulcers associated with non-steroidal anti-inflammatory drug (NSAID) therapy including Cox2 inhibitors. In the US,Nexiumis approved for reducing the risk of gastric ulcers associated with continuous NSAID therapy in patients at risk of developing gastric ulcers.
Nexium IV, which is used when oral administration is not suitable for the treatment of GERD and upper GI side effects induced by NSAIDs, is approved in 86 countries including the US and all EU countries.
During 2008, we announced the submission of a supplemental new drug application (sNDA) to the FDA forNexium IV(esomeprazole sodium) injection, seeking approval for use in patients with peptic ulcer bleeding following therapeutic endoscopy. This was followed by an EU submission forNexium IV and tablets, seeking approval for the short-term maintenance of haemostasis and prevention of re-bleeding in patients with peptic ulcer bleeding following therapeutic endoscopy.
In late November 2008, we received the FDA Complete Response Letter regarding ourNexium IVsNDA for peptic ulcer bleed. The application has not received FDA approval in its present form. We are reviewing their comments and will respond in due course. The EU submission is still being reviewed by the European regulatory authorities.
Since its launch in 1988, we estimate that patients have benefited from over 900 million treatments withLosec/Prilosec. We continue to maintain patent property coveringLosec/ Prilosec. Further information about the status of omeprazole patents and patent litigation, including details of generic omeprazole launches, is set out in Note 25 to the Financial Statements on page 150.
Entocorthas better tolerability than other corticosteroids in inflammatory bowel disease and greater efficacy than aminosalicylic acid medicines. It is prescribed as first-line therapy for both acute treatment and maintenance of clinical remission of mild to moderate, active Crohn’s disease and is approved in more than 40 countries.
Clinical trial developments
Data from theNexium IVPeptic Ulcer Bleed study (a multinational, randomised trial of 767 patients with peptic ulcer bleeding) is the basis for submissions in the US and EU referred to above. The study shows that use ofNexium IVfor three days, followed by oralNexiumtherapy for 27 days, was statistically more effective in reducing gastric ulcers compared to placebo after both three and 30 days.
In the pipeline
Our activities focus on reflux inhibitors and hypersensitivity therapy. Our lead compound, AZD3355, is undergoing clinical trials. Follow-up compounds are in Phase I testing.
Non-GERD related GI projects were successfully transferred to the spin-out company Albireo, in which AstraZeneca holds a large minority stake.
In the US, we are continuing to pursue patent litigation against various generic manufacturers who have filed abbreviated new drug applications (ANDAs) and are seeking to market esomeprazole magnesium products before the expiration of certain of our patents relating toNexium.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
OUR FINANCIAL PERFORMANCE
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | 2008 | | 2007 | | 2006 | | 2008 compared to 2007 | | 2007 compared to 2006 | |
| |
| | | | | | | | | | | Growth due | | | | | | | | | | | Growth due | | | | | | | | | | | | | | | | | |
| | | | | | | CER | | | to exchange | | | | | | | CER | | | to exchange | | | | | | | CER | | | Reported | | | CER | | | Reported | |
| | | Sales | | | growth | | | effect | | | Sales | | | growth | | | effect | | | Sales | | | growth | | | growth | | | growth | | | growth | |
| | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | % | | | % | | | % | | | % | |
| | |
Nexium | | | 5,200 | | | | (121 | ) | | | 105 | | | | 5,216 | | | | (104 | ) | | | 138 | | | | 5,182 | | | | (2 | ) | | | – | | | | (2 | ) | | | 1 | |
| | |
Losec/Prilosec | | | 1,055 | | | | (156 | ) | | | 68 | | | | 1,143 | | | | (277 | ) | | | 49 | | | | 1,371 | | | | (14 | ) | | | (8 | ) | | | (20 | ) | | | (17 | ) |
| | |
| Other | | | 89 | | | | 2 | | | | 3 | | | | 84 | | | | 2 | | | | 4 | | | | 78 | | | | 2 | | | | 6 | | | | 3 | | | | 8 | |
| | |
Total | | | 6,344 | | | | (275 | ) | | | 176 | | | | 6,443 | | | | (379 | ) | | | 191 | | | | 6,631 | | | | (4 | ) | | | (2 | ) | | | (6 | ) | | | (3 | ) |
| | |
On 15 April 2008, AstraZeneca announced it had settled itsNexiumpatent infringement litigation against Ranbaxy Pharmaceutical Industries and affiliates (Ranbaxy). As a consequence of the settlement, the patent litigation filed by AstraZeneca following Ranbaxy’s submission to the FDA of an ANDA for a generic version ofNexiumhas been dismissed. Under the settlement Ranbaxy concedes that all six patents asserted by AstraZeneca in the patent litigation are valid and enforceable. Ranbaxy also accepts that four of the patents would be infringed by the unlicensed sale of Ranbaxy’s proposed generic product. The settlement agreement allows Ranbaxy to commence sales of a generic version ofNexiumunder a licence from AstraZeneca from 27 May 2014, the expiry date of US Patent Numbers 5,877,192 and 6,875,872. We are co-operating fully with the Federal Trade Commission inquiry regarding this settlement.
AstraZeneca’sNexiumpatent infringement litigation against Teva/IVAX and Dr Reddy’s Laboratories remains ongoing. No trial date has been set in either case.
During 2008, we received additional notices that patent challenges had been filed by generic drug manufacturers in respect of 20mg and 40mg delayed-release esomeprazole magnesium capsules. Details of these filings and of new and continuing litigation are set out in Note 25 to the Financial Statements on page 153.
The European Patent Office ruled in 2007 that the European process patent forNexiumand the European patent for the multiple unit pellet (MUPS) formulations of PPI, which expire in 2015, are valid in amended form following post-grant oppositions. These decisions are now subject to appeal proceedings.
Further, the European Patent Office granted a new European patent on 19 November 2008 for the MUPS formulations of esomeprazole and omeprazole, which expires in 2015.
We continue to have full confidence in our intellectual property protectingNexiumand will vigorously defend and enforce it.
The decision of the European Court of First Instance on our appeal against the European Commission’s Decision in 2005 to impose fines on us totalling€60 million ($75 million) for alleged infringements of European competition law relating to certain omeprazole intellectual property and regulatory rights is still pending. Further information about this case is set out in Note 25 to the Financial Statements on page 151.
In 2008 we filed complaints for patent infringement against two generic manufacturers (Barr Laboratories and Mylan Pharmaceuticals) in response to notices of ANDA submissions in respect of generic forms ofEntocort EC.
FINANCIAL PERFORMANCE 2008/2007
PERFORMANCE 2008
Reported performance
Sales for 2008 were down 2% on a reported basis to $6,344 million from $6,443 million in 2007.
Performance – CER growth rates
GI sales fell by 4% at CER. GlobalNexiumsales were down 2%, excluding the effects of exchange, to $5,200 million from $5,216 million the previous year. The decline was driven by the decrease in the US of 8% to $3,101 million, however this was largely mitigated by sales in other markets increasing by 9% to $2,099 million. In the US, dispensed retail tablet volumes increased by 2% andNexiumwas the only major PPI brand to do so in 2008 In the Rest of World, growth in Canada (9%), Japan (5%) and Emerging Markets (20%) more than offset the 5% decline in Western European sales.
For the full year, sales ofLosecfell 14% to $1,055 million.Prilosecsales in the US were down 25% as a result of generic competition for the 40mg dosage form in the second half of the year. In the Rest of World, sales declined by 11%, despite increases in China (19%) and Japan (5%).
PERFORMANCE 2007
Reported performance
GI sales fell by 3% to $6,443 million in 2007 from $6,631 million in the previous year.
Performance – CER growth rates
GI sales fell by 6% at CER. Worldwide,Nexiumsales fell by 2% to $5,216 million. In the US,Nexium sales for the full year were $3,383 million, down 4%. Estimated volume growth was 2% for the year. Nexiummarket share in the branded segment of the PPI market increased by 1.5 percentage points in 2007; however, generic omeprazole share of the prescription PPI market increased to 27.4% by December 2007, an increase of nearly 7 percentage points since December 2006. Realised prices declined by around 8% for the year.Nexiumsales in other markets were up 2% for the full year to $1,833 million, as growth in Emerging Markets more than offset the declines in Western Europe.
For the full year,Losecsales declined by 20% to $1,143 million.Prilosecsales in the US were down 3% to $226 million.Losecsales in other markets were down 24%, although sales increased in Japan and China; sales in these two markets accounted for almost 30% of the brand’s performance.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| > | | Merremsales of $897 million, up 13%. |
| > | | Strong reported growth forMerremof 16% globally; 39% in the US. |
| > | | Synagissales of $1.23 billion; in the US $923 million. |
| > | | Biologics Licence Application submitted for motavizumab, an improved anti-respiratory syncytial virus monoclonal antibody. A Complete Response Letter subsequently received from the FDA. |
| > | | Market authorisation application submitted to European Medicines Agency for Live Attenuated Influenza Vaccine. |
OUR MARKETED PRODUCTS
Synagis(palivizumab) is a humanised monoclonal antibody used for the prevention of serious lower respiratory tract disease caused by RSV in paediatric patients at high risk of acquiring RSV disease.
Merrem/Meronem1 (meropenem) is a carbapenem anti-bacterial used for the treatment of serious infections in hospitalised patients.
FluMist(Influenza Virus Vaccine Live, Intranasal) is a live, attenuated, trivalent influenza virus vaccine licensed in the US for active immunisation of people two to 49 years of age against influenza disease caused by influenza virus subtypes A and type B contained in the vaccine.
| |
| 1 | Licensed from Dainippon Sumitomo Pharma Co., Ltd. |
We aim to build a leading franchise in the treatment of infectious diseases through continued commercialisation of the in-line brands such asSynagis,MerremandFluMist, effective use of our structural and genomic-based discovery technologies and antibody platforms, and through continued research of novel approaches in areas of unmet medical need.
RESISTANT BACTERIAL INFECTIONS
World demand for antibiotics remains high, due to escalating resistance and the increased risk of serious infections in both immuno-suppressed patients and ageing populations. Many bacterial infections currently have few satisfactory treatment options prompting demand for new and better therapies.
OUR FOCUS
Our key marketed products
Merrem/Meronem(meropenem) is a carbapenem antibiotic, which is active against most bacteria that cause serious infections in hospitalised patients. Merremis the leading carbapenem and has a growing share of the intravenous antibiotic market because of its activity against bacteria resistant to many other agents. To meet the high and growing need for new and better therapies for resistant bacterial infections we have built an anti-bacterials discovery capability that places AstraZeneca among the industry leaders with the capability to create novel mechanism anti-bacterials.
RESPIRATORY SYNCYTIAL VIRUS (RSV)
Approximately half of all infants are infected with RSV during the first year of life and nearly all children in the US have been infected by the time they reach their second birthday. Unlike other viral infections, there is no complete and durable immunity created by RSV, so repeated infection is likely and common. Premature babies (earlier than 36 weeks gestational age, especially those less than 32 weeks) or babies with chronic lung disease or congenital heart disease are at an even greater risk of contracting severe RSV disease than full-term babies.
OUR FOCUS
Our key marketed products
Synagisis used for the prevention of serious lower respiratory tract disease caused by RSV in children at high risk of the disease. It was the first monoclonal antibody (MAb) approved in the US for an infectious disease and since its launch in 1998 it has become the standard of care for RSV prevention.Synagisremains the only immunoprophylaxis in the marketplace
indicated for the prevention of RSV in paediatric patients at high risk of RSV.Synagisis administered by intra-muscular injection.
In the pipeline
During 2008, we filed a biological licence application with the FDA for an improved anti-RSV MAb, motavizumab. We recently completed a Phase III study with motavizumab as a prophylaxis in infants with haemodynamically significant congenital heart disease. We are also conducting a Phase IIb study with motavizumab as a treatment for children hospitalised with severe RSV disease. In November 2008 we received a Complete Response Letter from the FDA asking for additional information on motavizumab which we are confident we can respond to and does not lead us to believe it is necessary to conduct further clinical trials.
In addition, three intranasal vaccines are being developed for the prevention of lower respiratory tract illness caused by RSV and parainfluenza virus-3 (PIV3): MEDI-559 (RSV), MEDI-560 (PIV3) and MEDI-534 (RSV-PIV3). We are conducting several Phase I and Phase I/II studies for these vaccines alone and in collaboration with the US National Institute of Allergy and Infectious Diseases under a Co-operative Research and Development Agreement.
Influenza is the most common vaccine-preventable disease in the developed world. According to World Health Organization estimates, seasonal influenza results in three to five million cases of severe illness and up to half a million deaths globally each year, primarily among the elderly. Rates of infection are highest among children, with school-aged children significantly contributing to the spread of the disease. Influenza also has socio-economic consequences related to both direct and indirect healthcare costs, including hospitalisations, work absence and loss of work productivity when either a caregiver or child is sick with influenza.
OUR FOCUS
Our key marketed products
FluMistis the first live, attenuated nasally delivered vaccine approved in the US for the prevention of disease caused by influenza A and B viruses in eligible children and adults, ages two to 49 years. In 2008, the US Centres for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted to expand recommendations for routine seasonal influenza vaccination to include all school-age children up to the age of 18 years as soon as feasible but no later than the 2009/2010 influenza season. During the year,
ASTRAZENECA ANNUAL REPORT AND FOR M 20-F INFORMATION 2008
OUR FINANCIAL PERFORMANCE
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | 2008 | | | 2007 | | | 2006 | | | 2008 compared to 2007 | | | 2007 compared to 2006 | |
| | | | | | | | | | | Growth due | | | | | | | | | | | Growth due | | | | | | | | | | | | | | | | | |
| | | | | | | CER | | | to exchange | | | | | | | CER | | | to exchange | | | | | | | CER | | | Reported | | | CER | | | Reported | |
| | | Sales | | | growth | | | effect | | | Sales | | | growth | | | effect | | | Sales | | | growth | | | growth | | | growth | | | growth | |
| | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | % | | | % | | | % | | | % | |
| | |
Merrem | | | 897 | | | | 97 | | | | 27 | | | | 773 | | | | 121 | | | | 48 | | | | 604 | | | | 13 | | | | 16 | | | | 20 | | | | 28 | |
| | |
Synagis1 | | | 1,230 | | | | 612 | | | | – | | | | 618 | | | | 618 | | | | – | | | | – | | | | n/m | | | | n/m | | | | n/m | | | | n/m | |
| | |
FluMist1 | | | 104 | | | | 51 | | | | – | | | | 53 | | | | 53 | | | | – | | | | – | | | | n/m | | | | n/m | | | | n/m | | | | n/m | |
| | |
| Other | | | 220 | | | | (54 | ) | | | 4 | | | | 270 | | | | (12 | ) | | | 11 | | | | 271 | | | | (20 | ) | | | (19 | ) | | | (4 | ) | | | – | |
| | |
Total | | | 2,451 | | | | 706 | | | | 31 | | | | 1,714 | | | | 780 | | | | 59 | | | | 875 | | | | 41 | | | | 43 | | | | 89 | | | | 96 | |
| | |
we began rolling out the international marketing plan for our nasal spray influenza vaccine. The first milestone was the filing of a market authorisation application to the European Medicines Agency in late 2008.
HCV infects an estimated 170 million people worldwide and the current market for HCV therapy exceeds $2 billion annually. However, therapy for the strains that predominate in the US and Western Europe require 12 months’ treatment and produces a durable cure in only 50% of patients. Key opinion leaders expect the current standard of treatment (interferon plus ribavirin) to change to a form of combination therapy involving one or more new mechanism of action direct-acting anti-virals and there are several small and large pharmaceutical companies with varying HCV pipelines focused on such therapies. A future paradigm of combinations of anti-virals as standard care offers the opportunity for several new therapies to be widely used.
OUR FOCUS
In the pipeline
Projects in development include AZD7295, a novel HCV compound, currently in Phase II.
Sepsis is a life-threatening condition resulting from uncontrolled severe infections, which affects an estimated three million people a year worldwide. Few industry pipelines are focused on the development of products specifically for registration for the treatment of sepsis or septic shock.
OUR FOCUS
In the pipeline
The development programme for CytoFab™, our potential treatment for severe sepsis licensed from Protherics, continues in Phase II development. CytoFab™ has the potential to be one of a limited number of medicines specifically developed for such patients.
TB remains a worldwide threat and is newly diagnosed in over eight million people worldwide every year. It is one of the greatest causes of death from infectious disease in the developing world.
OUR FOCUS
As part of our commitment to making a contribution to improving health in the developing world, we are working to find a new, improved treatment for TB. We have a dedicated research facility in Bangalore, India that is focused on finding a treatment for TB that will act on drug-resistant strains, simplify the treatment regime (current regimes are complex and lengthy, meaning many patients give up before the infection is fully treated) and be compatible with HIV/AIDS therapies (TB and HIV/AIDS form a lethal combination, each speeding the other’s progress). Over 80 scientists in Bangalore work closely with our infection research centre in Boston, US as well as with academic leaders in the field, and they have full access to all AstraZeneca’s platform technologies, such as high throughput screening and compound libraries. It is a complex area of research, but we hope to have identified a candidate drug for testing in man within the next two to three years.
FINANCIAL PERFORMANCE 2008/2007
PERFORMANCE 2008
Reported performance
Total Infection sales increased on a reported basis by 43% to $2,451 million as a full year ofSynagisandFluMistsales were taken in the Group for the first time, andMerremsales enjoyed another year of good growth.
Performance – CER growth rates
Infection sales were up 41% at CER. For the full year,Synagissales were $1,230 million. Sales in 2007 were $618 million, but this only reflected sales since the acquisition of MedImmune in June 2007. Worldwide sales ofSynagisin the fourth quarter were $506 million, a 5% increase over the same period in 2007 when the product was included in sales.
FluMistsales were $104 million for the full year. In contrast to 2008, all of 2007FluMistsales of $53 million were realised in the fourth quarter as a result of the timing of regulatory approvals for the new formulation and expanded label.
PERFORMANCE 2007
Reported performance
Infection sales grew by 96% to $1,714 million from $875 million in 2006, driven by the inclusion of seven months ofSynagisandFluMistsales and an increase inMerremsales of 28%.
Performance – CER growth rates
Infection sales grew by 89%, after excluding the effect of exchange. CER growth of 20% fromMerrem, with sales of $773 million, and the inclusion ofSynagisandFluMistwere the principal drivers of this growth. Sales ofSynagistotalled $618 million for the period post-acquisition of MedImmune, with $480 million arising in the fourth quarter.Synagissales are highly seasonal, with the majority of sales recorded in the fourth and first quarters.
Sales ofFluMistwere $53 million for the full year, all of which were recorded in the fourth quarter. As withSynagis, there were no corresponding sales in the prior year period.
Sales ofMerremincreased by 20% to $773 million, with strong growth in the US (sales up 32% to $149 million) and Western Europe (sales up 20% to $307 million).
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| > | | Seroquelsales up 9% to over $4.45 billion.
|
| |
| > | | Seroquel XRapproved in the US for acute bipolar depression, acute bipolar mania and bipolar maintenance.
|
| |
| > | | Seroquel XRapproved under the European Mutual Recognition Procedure for the treatment of acute bipolar depression and acute bipolar mania in October.Seroquelalso approved at the same time for the treatment of acute bipolar depression.
|
| |
| > | | FDA Complete Response Letter received onSeroquel XRfor Major Depressive Disorder in December.
|
| |
| > | | Regulatory submissions made forSeroquel XRfor the treatment of Major Depressive Disorder and for Generalised Anxiety Disorder in both the US and EU.
|
| |
| > | | Summary Judgment Motion granted to AstraZeneca in the patent infringement actions commenced against two generic drug manufacturers in the US following abbreviated new drug applications relating toSeroquel.
|
| |
| > | | Separate lawsuits filed in the US against third party manufacturers relating to infringement of theSeroquel XRpatents.
|
| |
| > | | Personal injury actions in the US and Canada involvingSeroquelbeing defended vigorously.
|
Seroquel(quetiapine fumarate) is an atypical anti-psychotic drug approved for the treatment of adult schizophrenia and bipolar disorder (mania, depression and maintenance).
Zomig(zolmitriptan) is for the treatment of migraine with or without aura.
Diprivan(propofol) is an intravenous general anaesthetic used in the induction and maintenance of anaesthesia, light sedation for diagnostic procedures and for intensive care sedation.
Naropin (ropivacaine) is a long-acting local anaesthetic, replacing the previous standard treatment of bupivacaine.
Xylocaine(lidocaine) is a widely used short-acting local anaesthetic.
EMLA(lidocaine + prilocaine) is a local anaesthetic for topical application.
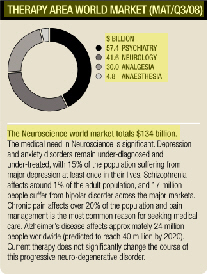
We aim to strengthen our position in neuroscience through further growth of
SeroquelandSeroquel XRand by the successful introduction of a range of new medicines aimed at significant medical need in psychiatry, analgesia (pain control) and cognition (including Alzheimer’s disease and cognitive disorders in schizophrenia).
Most branded schizophrenia products will face generic competition in the period 2012 to 2015, with all current atypical anti-psychotic patents expiring by 2018. Future demand will be for products with significantly improved efficacy and tolerability.
The depression and anxiety markets are currently dominated by generic selective serotonin re-uptake inhibitors and serotonin norepinephrine re-uptake inhibitors. As growth in the US slows, the Japanese market continues to grow. Generic growth is anticipated over the next five years as patents expire.
OUR FOCUS
Our key marketed products
Seroquelis a leading atypical anti-psychotic treatment for adult schizophrenia and bipolar disorder.Seroquelremains the most commonly prescribed atypical anti-psychotic in the US, where it is the only atypical anti-psychotic approved as monotherapy treatment for both bipolar depression and bipolar mania as well as the leading atypical brand globally by sales value. Its clinical development programme was substantially completed during 2008 resulting in worldwide launches ofSeroquel XRfor schizophrenia. We have also made the associated regulatory submissions and data presentations in bipolar disorder, major depressive disorder (MDD) and generalised anxiety disorder (GAD).
First launched in 1997,Seroquelis now approved in 92 countries.Seroquel XR, an extended release formulation that offers patients and doctors a once-daily treatment, was launched in the US for the treatment of schizophrenia in 2007 and is now approved in 45 countries for schizophrenia, 12 countries for bipolar mania, seven countries for bipolar depression and four countries, including the US, for bipolar maintenance, in one market for MDD, and in one market for GAD.
In 2008, the FDA approvedSeroquel XRfor the treatment of depressive episodes associated with bipolar disorder, the manic and mixed episodes associated with bipolar 1 disorder and bothSeroquelandSeroquel XRfor the maintenance treatment of bipolar 1 disorder as adjunctive therapy to lithium or divalproex. In addition,Seroquel XRandSeroquelwere approved in the EU for the treatment of major depressive episodes in bipolar disorder.Seroquel XRwas also licensed in the EU for moderate to severe manic episodes in bipolar disorder.
During 2008, regulatory submissions were made in both the US and in the EU for GAD and for MDD. AstraZeneca received a Complete Response Letter from the FDA for its sNDA forSeroquel XRfor the treatment of MDD in adult patients. AstraZeneca is continuing discussions with the FDA. A separate regulatory submission was made to the FDA for the treatment of schizophrenia in adolescents (13 to 17 year olds) and for the treatment of acute manic episodes in children and adolescents (10 to 17 year olds) with bipolar 1 disorder. The US prescribing information forSeroquel andSeroquel XRis being updated to include new safety information regarding use in children and adolescents.SeroquelandSeroquel XRare not approved currently for use in paediatric patients under 18 years of age.
In January 2009, the FDA granted an additional six-month period of market exclusivity toSeroquelfor its licensed indications, based on studies we conducted in adolescents with schizophrenia and children and adolescents with bipolar mania. TheSeroquelpatent expires in September 2011. The allowed six-month paediatric exclusivity period, which takes effect upon expiration of the patent, will extend the exclusivity ofSeroquelto March 2012.
In the pipeline
We have progressed AZD8529 into Phase I and AZD2624 into Phase II for the treatment of schizophrenia, with AZD2327 entering Phase Ila and AZD6765 and AZD7325 entering into Phase IIb clinical development for the treatment of anxiety and/or depression.
We also continued to build our alliance/ partnership network in 2008 by entering into a collaboration with the Columbia University Medical Center to examine further the relevance of adult neurogenesis in anti-depressant action and novel approaches to treat depression and anxiety.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
OUR FINANCIAL PERFORMANCE
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | 2008 | | | 2007 | | | 2006 | | | 2008 compared to 2007 | | | 2007 compared to 2006 | |
| | | | | | | | | | | Growth due | | | | | | | | | | | Growth due | | | | | | | | | | | | | | | | | |
| | | | | | | CER | | | to exchange | | | | | | | CER | | | to exchange | | | | | | | CER | | | Reported | | | CER | | | Reported | |
| | | Sales | | | growth | | | effect | | | Sales | | | growth | | | effect | | | Sales | | | growth | | | growth | | | growth | | | growth | |
| | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | $m | | | % | | | % | | | % | | | % | |
| | |
Seroquel | | | 4,452 | | | | 346 | | | | 79 | | | | 4,027 | | | | 526 | | | | 85 | | | | 3,416 | | | | 9 | | | | 11 | | | | 15 | | | | 18 | |
| | |
Diprivan | | | 278 | | | | (3 | ) | | | 18 | | | | 263 | | | | (53 | ) | | | 12 | | | | 304 | | | | (1 | ) | | | 6 | | | | (17 | ) | | | (13 | ) |
| | |
Zomig | | | 448 | | | | (3 | ) | | | 17 | | | | 434 | | | | 18 | | | | 18 | | | | 398 | | | | (1 | ) | | | 3 | | | | 5 | | | | 9 | |
| | |
| Local anaesthetics | | | 605 | | | | 13 | | | | 35 | | | | 557 | | | | (6 | ) | | | 34 | | | | 529 | | | | 2 | | | | 9 | | | | (1 | ) | | | 5 | |
| | |
| Other | | | 54 | | | | (7 | ) | | | 2 | | | | 59 | | | | (1 | ) | | | 3 | | | | 57 | | | | (12 | ) | | | (8 | ) | | | (2 | ) | | | 4 | |
| | |
Total | | | 5,837 | | | | 346 | | | | 151 | | | | 5,340 | | | | 484 | | | | 152 | | | | 4,704 | �� | | | 6 | | | | 9 | | | | 10 | | | | 14 | |
| | |
ANALGESIA AND ANAESTHESIA
(PAIN CONTROL)
Significant unmet need remains for efficacy and tolerability in the neuropathic pain market. Several novel compounds are in development but recent disappointments highlight continuing uncertainty regarding market approval.
The osteoarthritis (OA) market is steadily growing, due to ageing populations and novel agents entering the market. However, the established use of generic treatment makes market entry more difficult. Biologics are an emerging treatment option for OA.
OUR FOCUS
Our key marketed products
ZomigNasal Spray was approved for the acute treatment of cluster headache in 14 member states in the EU in 2008.
Diprivanis the world’s best-selling intravenous general anaesthetic. A complete change over toDiprivanEDTA, a microbial-resistant formulation, is expected in 2009, following the approval of this formulation in the last major territory (UK) in 2008.
Naropinapprovals continue for extended use in paediatric patients to include neonates and infants aged below one year old.
EMLAsubmissions/approvals of patch presentation have continued, particularly in Eastern European countries. In Japan,EMLAis out-licensed to SATO who expect to file their Japanese NDA in July 2009.
In the pipeline
PN400 is a fixed-dose combination tablet of enteric-coated naproxen and immediate release esomeprazole which uses proprietary technology licensed from POZEN Inc. through a collaboration established in August 2006. It is being developed for the relief of signs and symptoms of OA, rheumatoid arthritis and ankylosing spondylitis in patients at risk for developing non-steroidal anti-inflammatory drug (NSAID)-associated gastric ulcers. Approximately half of the 121 million chronic arthritis patients in the US and the five largest European countries are at risk of developing NSAID-associated ulcers based on their age, prior history of ulceration, or use of low dose aspirin. The Phase III trial programme, which was initiated in the third quarter of 2007, has now completed enrolment. The two Phase III ulcer risk reduction studies, comparing PN400 against enteric-coated naproxen 500mg in subjects with chronic pain and who are at risk for NSAID-associated ulcers, achieved their primary endpoints. Subjects taking PN400 experienced statistically significantly fewer endoscopically confirmed gastric ulcers than those taking naproxen. Two additional Phase III studies are still ongoing. Upon completion of the entire PN400 Phase III clinical programme, AstraZeneca will make a final determination regarding regulatory filing. A regulatory submission for PN400 in the US is currently planned for mid 2009.
We progressed three other early development compounds during the year: AZD2516 into Phase I clinical development and AZD1386 and AZD1940 in Phase II clinical development for the treatment of nociceptive (caused by tissue damage) and/or neuropathic (caused by nerve damage) pain.
Alzheimer’s disease remains one of the largest areas of unmet need and also one of high risk for neuroscience product development, due in part to the challenges of establishing efficacy in clinical trials. Current treatments, which physicians consider inadequate, target the symptoms not the underlying cause of the disease. Achieving disease modification is very difficult evidenced by recent late stage product development failures. Growth in this area is strong (20% to 40% across the world) but all existing agents will face patent expiry by 2013.
There are currently no products approved to treat cognitive dysfunction associated with schizophrenia. The first product to market will face the challenge of disease education and the establishment of treatment guidelines.
OUR FOCUS
In the pipeline
We have expanded the portfolio of potential medicines in this area to five development programmes, of which three are in clinical evaluation, in Alzheimer’s disease, cognitive disorders in schizophrenia (CDS) and other cognition disorders. In addition to developing molecules for cognitive disorders, we continue to progress two development phase molecules for the treatment of other neurodegenerative diseases.
Through our collaboration with, amongst others, the Karolinska Institute in Sweden, our research capabilities in positron emission tomography, which provides early signalling of potential efficacy for our Alzheimer’s compounds, continue to progress. We now have two C-11 diagnostic compounds and one F-18 compound in development.
Compounds in clinical evaluation include products deriving from our relationship with Targacept (AZD3480, TC-5619 and AZD1446).
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
AZD3480, a neuronal nicotinic receptor agent, is currently in Phase IIb clinical testing in Alzheimer’s disease and TC-5619 is in Phase II clinical testing for CDS. AZD3480 did not meet the Phase IIb trial primary endpoint for CDS and is not expected to progress to Phase III studies in this indication. AstraZeneca and Targacept previously announced top-line results from a Phase IIb study of AZD3480 in mild to moderate Alzheimer’s disease and are currently evaluating AZD3480 in a Phase II exploratory study in attention deficit/hyperactivity disorder (ADHD) in adults. A decision by AstraZeneca with respect to potential further development of AZD3480 in Alzheimer’s disease or ADHD is now expected in the first half of 2009, pending completion of the adult ADHD study and other ongoing evaluations.
AstraZeneca is defending approximately 9,210 served or answered lawsuits involving approximately 15,461 plaintiff groups who have filedSeroquel-related product liability claims in the US and Canada. Although the nature of the alleged injuries is not clear from the face of most of the complaints and discovery of the cases is continuing, plaintiffs generally contend that they developed diabetes and/or other related injuries as a result of takingSeroqueland/or other atypical anti-psychotic medications. Further information can be found in Note 25 to the Financial Statements on page 155. Trials of these cases are expected to commence in 2009.
In July 2008 AstraZeneca was granted a Summary Judgment Motion from the US District Court for the District of New Jersey in the ongoing patent infringement action against Teva Pharmaceuticals USA Inc and Sandoz, Inc. Teva and Sandoz have filed appeals.
Separate lawsuits have been filed in the US against third party manufacturers relating to infringement of theSeroquel XRpatents.
We continue to have full confidence in our intellectual property protectingSeroqueland will vigorously defend and enforce it. Details of the litigation against generic drug manufacturers in respect ofSeroquelare set out in Note 25 to the Financial Statements on page 155.
FINANCIAL PERFORMANCE 2008/2007
PERFORMANCE 2008
Reported performance
Neuroscience sales grew by 9% to $5,837 million in 2008 from $5,340 million in 2007. All geographic areas experienced growth andSeroquelgrew strongly by 11%.
Performance – CER growth rates
Sales in the Neuroscience therapy area grew by 6% to $5,837 million from $5,340 million last year.
US sales forSeroquelfor the full year were $3,015 million, 5% ahead of last year. Seroquelremains the market leader in the US anti-psychotic market, with a total prescription share of 31.6% in December 2008.
For the full year,Seroquelsales in the Rest of World increased by 17% to $1,437 million, with value and volume growth well ahead of the market in all regions.
Sales ofZomigfor the full year were up 6% in the US to $187 million. Sales in the Rest of World were down 5% to $261 million.
PERFORMANCE 2007
Reported performance
Sales in the Neuroscience therapy area rose by 14% in 2007, up to $5,340 million from $4,704 million in 2006.Seroquelwas the principal driver of performance, recording an 18% increase in sales.
Performance – CER growth rates
Neuroscience sales grew by 10% at CER. AnnualSeroquelsales exceeded $4 billion for the first time in 2007. Full year sales were $4,027 million, up 15% over last year. In the US,Seroquelsales increased by 15% to $2,863 million.Seroquelsales in other markets were up 16% for the full year, as a result of market share gain in most markets.
Zomigsales for the full year increased 5% in the US (to $177 million) and 4% in other markets, totalling $434 million.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| > | | Arimidexsales up 4% to $1.86 billion and is the leading branded hormonal breast cancer therapy in the US, Japan and France.
|
| |
| > | | Casodexsales $1.26 billion, down 12%. Expiry of EU marketing exclusivity in 2008.
|
| |
| > | | Zoladexsales $1.14 billion, down 3%.
|
| |
| > | | Results from three Phase IIIZactimatrials in non-small cell lung cancer (NSCLC) showed thatZactima, in combination with standard chemotherapy, brings clinical benefits to patients with previously treated NSCLC.
|
| |
| > | | Results from theIressaPhase III INTEREST study underpin a marketing authorisation application in the EU and the pan-Asian IPASS study met its primary objective showing superior progression-free survival forIressacompared with two chemotherapies in clinically selected patients.
|
| |
| > | | ZD4054 progressed into Phase III development for hormone-resistant prostate cancer.
|
| |
| > | | Registration trials ongoing ofRecentinin first line colorectal cancer and recurrent glioblastoma multiforme.
|
Arimidex(anastrozole) is an aromatase inhibitor for the treatment of breast cancer.
Casodex(bicalutamide) is an anti-androgen therapy for the treatment of prostate cancer.
Zoladex(goserelin acetate implant), in one- and three-month depots, is an LHRH agonist for the treatment of prostate cancer, breast cancer and certain benign gynaecological disorders.
Iressa(gefitinib) is an EGFR-TKI that acts to block signals for cancer cell growth and survival in non-small cell lung cancer.
Faslodex(fulvestrant) is an injectable oestrogen receptor antagonist for the treatment of breast cancer, with no known agonist effects, that down-regulates the oestrogen receptor.
Nolvadex(tamoxifen citrate) remains a widely prescribed breast cancer treatment outside the US.
Ethyol(amifostine) is used to help prevent certain side effects of specific types of chemotherapy and radiotherapy that are used to treat head and neck and ovarian cancer.
Abraxane® (paclitaxel protein-bound particles for injectable suspension) (albumin-bound) for the treatment of breast cancer1.
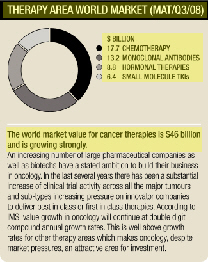
We aim to build on our position as a world leader in cancer treatment through continued growth ofArimidex, further launches and line extensions of newer products, such asFaslodex, and the successful introduction of novel therapeutic approaches currently in development, including both small molecule and biological drugs, targeted at high unmet need.
OUR FOCUS
Our key marketed products
During 2008, our breast cancer treatment,Arimidexmaintained its position as market leader in sales of branded hormonal agents, with approximately four million patient years of clinical experience. This success is largely based on the extensive long-term efficacy and safety results of the ATAC study, which showedArimidexto be significantly superior to tamoxifen at preventing breast cancer recurrence during and beyond the five-year treatment course. (Breast cancer recurrence is defined as loco-regional recurrence, distant recurrence or contra-lateral breast cancer).
Arimidexcontinues to be the leading branded hormonal therapy for new patients in the US, Japan and France, and is also approved in a number of markets in Europe for a switch indication for patients who have already received two to three years of tamoxifen.
Faslodex, now approved in more than 60 markets, offers an additional hormonal therapy for patients with hormone-sensitive, advanced breast cancer, delaying the need for cytotoxic chemotherapy. It is a once-monthly injection approved for the second-line treatment of hormone-receptor positive, advanced breast cancer in post-menopausal women.
Casodexis used as a 50mg tablet for the treatment of advanced prostate cancer, and as a 150mg tablet for the treatment of locally advanced prostate cancer. European sales declined due to generic erosion following patent and/or marketing exclusivity expiries in July 2008. Sales growth continued in Japan, whereCasodex is available as an 80mg tablet and is approved for all stages of prostate cancer. In the US, the FDA granted an additional six months’ paediatric extension providing marketing exclusivity in the US to April 2009.
| |
| 1 | In November 2008, we entered into an agreement with Abraxis under which Abraxis re-acquired exclusive rights to market Abraxane® in the US. |
Zoladexis approved in 120 countries. It is approved for the treatment of prostate cancer, breast cancer and gynaecological disorders. In non-metastatic prostate cancer,Zoladexis the only luteinising hormone-releasing hormone (LHRH) agonist shown to improve overall survival both when used in addition to radical prostatectomy and when used in addition to radiotherapy. The 10-year follow-up results of a study for the European Organisation for Research and Treatment of Cancer confirmed the long-term survival benefits ofZoladexwhen used as adjuvant to radiotherapy in patients with locally advanced prostate cancer.
In breast cancer,Zoladexis widely approved for use in advanced breast cancer in pre-menopausal women. In a number of countries,Zoladexis also approved for the adjuvant treatment of early stage pre-menopausal breast cancer as an alternative to and/or in addition to chemotherapy.Zoladexoffers proven survival benefits for breast cancer patients with a favourable tolerability profile.
Competition in the LHRH agonist market is expected to increase in Europe during 2009, with the anticipated launches of generic goserelin. This follows the announcement of the approval of generic goserelin (one-month depot) in Germany in December.
Iressais approved in 36 countries and is the leading epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI) in the Asia Pacific region where it continues to be marketed for pre-treated advanced NSCLC. Based on data from the Phase III INTEREST study comparingIressawith docetaxel, a marketing authorisation application forIressahas been submitted to the European Medicines Agency.
Outside the US, we have various distribution and marketing arrangements for brandedEthyol. As of June 2008, our two main distribution partners are Pinnacle Biologics for Western Europe, Turkey and Israel, and Schering-Plough International for Rest of World.
Clinical trial developments
Results from the Phase III pan-Asian IPASS study evaluating the efficacy ofIressa as first-line treatment of NSCLC, were also announced. The IPASS study exceeded its primary objective, demonstrating superior
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
efficacy in metastatic HRPC, both as monotherapy and in combination with docetaxel, and in non-metastatic HRPC.
In December 2008, we ceased our collaboration with Infinity Pharmaceuticals for the development and commercialisation of Infinity’s drug candidates IPI-504 (MEDI-561) and IPI-493 for the treatment of cancer and related conditions. This decision was taken after reviewing the potential opportunity for these projects and to take account of competing R&D investment priorities.
Our early oncology pipeline includes a range of novel compounds that target signalling pathways believed to be pivotal in cancer cell growth, invasion DNA repair and survival, with nine products in Phase II and 15 others in Phase I development. Phase II data from AZD6244, a potent MEK inhibitor licensed from Array BioPharma, showed biological activity in lung cancer and melanoma and studies will now focus on its use in combination with standard and other novel therapies, rather than its development as monotherapy. Phase II studies with the poly-ADP-ribose-polymerase (PARP) inhibitor AZD2281 have started and will initially focus on BRCA-mutated breast and ovarian cancer as well as other cancers where DNA repair could be defective.
The dual-specific Src/Abl kinase inhibitor, AZD0530, has shown a dramatic effect on biomarkers of cell motility and bone resorption and has started Phase II studies in ovarian cancer with others to follow. Among the compounds from the early portfolio continuing in development are AZD4877, a novel inhibitor of cell cycle; AZD7762, a tumour-selective chemosensitiser; and AZD8931. AZD1152, an aurora kinase inhibitor, has shown activity in acute myelogenous leukaemia and will commence Phase II/III studies in 2009. We are also developing potential new cancer treatments using biological approaches with highly defined molecular targets for patient populations with unmet medical needs.
CAT-8015 is an immunotoxin fusion protein that targets CD22, which is a receptor expressed on the surface of a wide variety of B-cell malignancies. CAT-8015 has orphan drug designation for hairy cell leukaemia in the US and EU. In 2008, the enrolment for studies continued in the CAT-8015 Phase I development programme.
Blinatumomab (MEDI-538) is a recombinant single-chain bi-specific T-cell engager (BiTE™) molecule that is being studied for use in certain
patients suffering from certain lymphomas and leukaemias. Exclusive rights to develop and commercialise blinatumomab in North America have been granted from Micromet.
The US Phase I programme with blinatumomab was suspended during 2008 in order to make appropriate modifications to the dosing regimen based on preliminary results from the EU studies.
In April 2008, Sun Pharmaceuticals launched generic amifostine in the US. In response, we extended an agreement with Bedford Laboratories to launch an authorised generic amifostine for oncology in the US. We have ceased all active promotion of brandedEthyolin the US. We have an active infringement action against Sun Pharmaceuticals regarding certainEthyolpatents.
Abraxane®, discovered, developed and owned by Abraxis, uses a novel technology to deliver paclitaxel for the treatment of breast cancer. During 2008, we co-promoted Abraxane® in the US under an agreement with Abraxis. In November 2008, we entered into an agreement with Abraxis under which Abraxis re-acquired exclusive rights to market Abraxane® in the US. Under the agreement, the board of Abraxis’ parent ended the Co-Promotion Agreement. Upon termination, Abraxis will pay AstraZeneca a $268 million fee on 31 March 2009.
FINANCIAL PERFORMANCE 2008/2007
PERFORMANCE 2008
Reported performance
Sales in Oncology increased by 3% on a reported basis to $4,954 million up from $4,819 million in 2007.
Performance – CER growth rates
Sales in the Oncology therapy area were down 2% at CER.Arimidexsales were up 4% to $1,857 million. In the US,Arimidexsales were up 9% to $754 million. In other markets, sales increased by 1% to $1,103 million.
Casodexsales decreased by 12% to $1,258 million, with sales in the US down by 2% and sales in other markets down by 15%.
Iressasales for the year were up 3% as growth in China and other Emerging Markets more than offset the 3% decline in sales in Japan.
Faslodexsales were up 12% with a 5% increase in the US and 18% in other markets.
PERFORMANCE 2007
Reported performance
Oncology sales increased by 13% to reach $4,819 million in 2007, compared with $4,262 million in 2006.
Performance – CER growth rates
Oncology sales grew by 8% at CER.Arimidexsales reached $1,730 million, up 10%. In the US, sales ofArimidexrose by 13% to $694 million. Total prescriptions forArimidexincreased nearly 5.3% compared with 1.3% growth in the market for hormonal treatments for breast cancer.Arimidexsales in other markets increased by 8% to $1,036 million. Sales for the full year were up 6% in Western Europe and increased 9% in Japan.
Casodexsales increased by 6% to $1,335 million. Sales in the US for the full year were up 1% to $298 million. Sales in other markets, which accounted for more than 75% of product sales, were up 8%, on 6% growth in Western Europe and 13% sales growth in Japan.
Iressasales were unchanged for the full year. Sales in Japan increased 4% for the year; sales in China were up 24%.
Faslodexsales increased 10% to $214 million for the full year, on growth of 3% in the US and 18% sales growth in other markets.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| | | | | | | |
| | | RESPIRATORY AND INFLAMMATION | | | 67 | |
| > | | Symbicortsales over $2 billion, up 22%.
|
| |
| > | | Symbicort Rapihaler(pMDI) licensed for long-term maintenance treatment of asthma in the US. Submissions made for use in COPD and paediatric asthma.
|
| |
| > | | Outside the US,Symbicort Turbuhaler SMARTnow approved for use in managing asthma in over 90 countries.
|
| |
| > | | Symbicort Turbuhalernow approved in COPD in over 80 countries.
|
| |
| > | | The Joint Advisory Committee of the FDA concluded that the benefits ofSymbicortoutweigh the risks in adult and adolescent asthma patients.
|
| |
| > | | Continued growth forPulmicortwith sales of $1.49 billion.
|
| |
| > | | Settlement of AstraZeneca’sPulmicort Respulespatent infringement litigation against Teva including an exclusive licence to Teva to sell genericPulmicort Respulesfrom 15 December 2009 with significant royalties for AstraZeneca.
|
Symbicort Turbuhaler(budesonide/formoterol in a dry powder inhaler) is a combination of an inhaled corticosteroid and a fast onset, long-acting bronchodilator for the treatment of asthma and COPD.
Symbicort SMARTis licensed for maintenance therapy as well as for maintenance and reliever therapy in persistent asthma.
Symbicort Rapihaler(pMDI) (budesonide/ formoterol in a pressurised metered-dose inhaler) for the maintenance treatment of asthma.
Pulmicort(budesonide) is a corticosteroid anti-inflammatory inhalation drug that helps prevent symptoms and improves the control of asthma.
Pulmicort Respules(budesonide inhalation suspension) is the first and only nebulised corticosteroid in the US for the treatment of asthma in children as young as 12 months.
Rhinocort(budesonide) is a nasal steroid treatment for allergic rhinitis (hay fever), perennial rhinitis and nasal polyps.
Oxis(formoterol) is a fast onset, long-acting beta-agonist for treating asthma and COPD.
Accolate(zafirlukast) is an oral leukotriene receptor antagonist for the treatment of asthma.
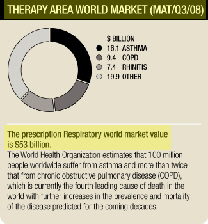
We aim to build on our strong position in asthma treatment through the growth of key products, particularlySymbicort, with new indications and market launches as well as developing novel approaches to other areas of inflammatory disease such as chronic obstructive pulmonary disease (COPD) and rheumatology.
COPD is expected to become the world’s third biggest health threat by 2020. Current treatment has recently demonstrated some survival benefit but the prognosis of the COPD patient remains poor. In asthma, morbidity and mortality remain important issues and disease normalisation is not achieved by any treatment.
The typical treatment across COPD and asthma is a fixed-dose combination of an inhaled corticosteroid (ICS) with a long-acting beta-agonist (LABA) or for COPD specifically, inhaled long-acting muscarinic agonist (LAMA). Other major asthma treatments include oral leukotriene receptor antagonists and oral steroids for severe disease and (in combination with antibiotics) for exacerbations. Significant new product classes impacting the asthma market up to 2015 are unlikely. First novel anti-inflammatory compounds aimed mainly at prevention and/or treatment of COPD exacerbations, such as oral phosphodiesterase 4 inhibitors, may appear on the market before 2015.
Symbicort SMARTflexible dosing introduced a step change to asthma care in Europe resulting in lower ICS and oral steroid use. Novel ICS/LABA combination products for this area are expected from 2009 and generic ICS/LABA combinations may be available from the early part of the next decade. Several companies are developing new biologics for severe asthma, including improved versions of anti-IgE and differentiated anti-cytokine antibodies. Post-2015, immune response modifiers could deliver intermittent therapy for moderate to severe asthma.
A number of novel COPD combinations in industry pipelines may change the way in which COPD is managed. Combinations of LABAs, LAMAs and triple-combinations with existing and new anti-inflammatories, may become future treatments of choice. There are also agents in early development with the potential to change the course of the disease by targeting the immune and inflammatory response that results in lung damage.
OUR FOCUS
Our key marketed products
Symbicort Turbuhalerprovides rapid, effective control of asthma and effective reduction of exacerbations, improving symptoms and providing a clinically important improvement in the health of patients with severe COPD.
Symbicort Rapihaler(pMDI) approved for the long-term maintenance treatment of asthma in patients 12 years of age and older, was launched in the US in 2007. Further information about the progress ofSymbicortsince its launch in the US is set out in the Geographical Review on page 49. In December 2008, the Joint Advisory Committees of the FDA completed a review of the benefits and risks of asthma medications containing LABAs. This concluded that the benefits ofSymbicort outweigh the risks in adult and adolescent asthma patients.
Symbicort SMARTprovides increased asthma control and simplifies asthma management through use of only one inhaler for both maintenance and relief of asthma symptoms. It is also a cost-effective treatment option for many healthcare payers.Symbicort SMARTis included in the Global Initiative for Asthma, the international treatment guidelines.
The US sNDAs forSymbicort Rapihaler(pMDI) in COPD and paediatric asthma in the US were submitted as planned during the second quarter of 2008. Our existing regulatory filings forSymbicort Rapihaler(pMDI) in the EU for asthma and COPD were supplemented with data supporting two additional strengths in the second half of 2008.
Pulmicortremains one of the world’s leading asthma medicines and is available in several forms.Pulmicort pMDIis now approved in 98 countries.
Information about our settlement of the patent infringement action against IVAX in the US, which began in October 2005, in relation to IVAX’s ANDA for a budesonide inhalation suspension is set out in Note 25 to the Financial Statements from page 154.
Oxisis added to the treatment regime when corticosteroid treatment alone is not adequate.Oxisis also indicated for symptom relief in COPD.
Rhinocortcombines powerful efficacy with rapid onset of action and minimal side effects and is available as a once-daily treatment in theRhinocort Aqua(nasal spray) and theTurbuhalerdry powder inhaler forms.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
The RA market has grown from $1.3 billion in 1998 to over $10 billion in 2008, driven largely by the introduction of biologic tumour necrosis factor alpha (TNFa) blockers (first Amgen/ Wyeth’s Enbrel™, followed by Centocor/ Schering-Plough’s Remicade™ and Abbott’s Humira™), which together account for over $8 billion in this disease alone. Launches of additional TNF blockers are imminent, and use of other biologic approaches, currently reserved for TNF failures, is expected to increase. Targeted novel oral drugs aimed at patients that currently choose not to take, are ineligible for or don’t respond to biologics, are in development to provide anti-TNF-like efficacy with safety benefits and more convenient dosing.
Current treatment of systemic lupus erythematosus (SLE) focuses on controlling disease flares, preventing renal failure and suppressing symptoms to an acceptable level while minimising toxicity. Despite considerable recent development activity, no targeted disease-modifying agents have yet been successfully launched for SLE. Most emerging biologic agents will likely be used initially in combination with corticosteroids or immunosuppressants to provide incremental benefit and/or allow reduced doses or numbers of these agents.
OUR FOCUS
In the pipeline
In 2008, we invested in several novel multi-functional MAbs that allow simultaneous inhibition of either two secreted proteins or surface receptors. Our first disease being studied is RA, where TNF inhibitors with other molecules may improve both the efficacy and prevent the establishment of TNF refractory disease while maintaining an acceptable safety profile.
MEDI-545 is a MAb targeting interferon-alpha, which regulates processes involved in autoimmune diseases. In 2008, we initiated a Phase IIa trial in patients with SLE and a Phase I study in patients with active dermatomyositis.
CAM-3001 is a MAb with potential to help patients with RA. The antibody targets the alpha sub-unit of the granulocyte-macrophage colony stimulating factor receptor. In September 2008, preliminary results were reviewed from the first Phase I study of CAM-3001, which had been initiated to evaluate the safety profile and tolerability of single doses in patients with RA. AstraZeneca, through its acquisition of MedImmune, acquired exclusive development rights to the CAM-3001 programme from CSL Limited in 2007.
AZD9056 and AZD5672 are novel oral compounds being primarily developed as a new generation of disease modifying anti-rheumatoid arthritis drugs. Currently in Phase IIb, their different mechanisms of action (a P2X7 antagonist and a CCR5 antagonist) provide multiple chances of success to provide significant new choice in the management of RA.
We also have an ongoing alliance with Bayer Schering in respiratory and rheumatology indications with the objective of identifying novel compounds without steroid side effects.
Patent infringement litigation filed by AstraZeneca against IVAX Pharmaceuticals (a wholly owned subsidiary of Teva Pharmaceuticals USA (Teva)) following the submission of an ANDA with the FDA for genericPulmicort Respuleswas settled in November 2008. Under the terms of the settlement, Teva concedes that the patents asserted by AstraZeneca in the litigation are valid and enforceable and that its generic version ofPulmicort Respulesinfringes AstraZeneca’s patents. See Note 25 for further details.
FINANCIAL PERFORMANCE 2008/2007
PERFORMANCE 2008
Reported performance
Sales in Respiratory and Inflammation therapy area (R&I) increased by 11% to $4,128 million from $3,711 million in 2007.
Performance – CER growth rates
R&I sales grew by 7% at CER.
Sales ofSymbicortgrew by 22% to $2,004 million. In the US, sales of the product were $255 million, up 410%. Product trial rate among target specialist physicians is now approaching 90%; these specialists are now starting more than 30% of patients new to combination therapy onSymbicort. More than half of target primary care physicians have prescribedSymbicort, and share of new patient starts is just under 18%. Overall,Symbicortshare of new prescriptions for fixed combinations reached 11.7% in the week ending 16 January, with market share among patients newly starting combination treatment at 18.3%.Symbicortsales in other markets in the year were $1,749 million, up 9%.Symbicort SMARThas now been approved in 91 markets.
Pulmicortsales were flat at $1,495 million, with US sales up 2% as the generic competition from the Teva product affected quarter four sales. US sales forPulmicortwere down 15% to $260 million in the fourth quarter andPulmicort Respulessales were down 18% as a result of the “at risk” launch of generic budesonide inhalation suspension (BIS) on 18 November. The patent litigation between Teva and AstraZeneca was subsequently settled on 26 November. The agreement allows Teva to commence sales of BIS under an exclusive licence from AstraZeneca beginning 15 December 2009. The agreement also provided that any product already shipped by Teva would remain in the market to be further distributed and dispensed. As a result, Teva products accounted for nearly 15% of total prescriptions for BIS products dispensed during the fourth quarter, including a 40% share in December 2008. US sales forPulmicortfor the full year were $982 million.Pulmicort Respulesaccounted for around 90% of totalPulmicort sales in the US. Sales ofPulmicortin Rest of World were down 2% for the full year to $513 million.
PERFORMANCE 2007
Reported performance
Continued growth fromSymbicortdrove the increase in reported sales for R&I, which grew by 18% from $3,151 million in 2006 to $3,711 million in 2007.
Performance – CER growth rates
Sales in R&I increased by 12% at CER.
Symbicortsales for the full year were up 22% to $1,575 million. Sales in Western Europe were up 16%, with market share up another point in the last 12 months, aided by the roll-out of theSymbicort SMARTregime. Good growth for the year was achieved in Canada (up 25%) and in Emerging Markets (up 26%). Sales in the US were $50 million since launch at the end of June 2007. Specialist physicians rapidly adopted the product; nearly 75% of allergists and more than 60% of pulmonary specialists in our target audience have prescribedSymbicort.Pulmicortsales increased by 10% to $1,454 million. US sales increased 15% for the full year to $964 million.Pulmicort Respulessales in the US were up more than 20% for the full year, on estimated volume growth of 15%. Of the approximately six million children under the age of eight who are treated for asthma, more than one million benefit from treatment withPulmicort Respules. Sales in other markets were unchanged for the year.
Rhinocortsales fell by 4% to $354 million, with a 9% decline in the US being compensated by small gains elsewhere.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
DEFINITIONS AND INTERPRETATION
Figures in parentheses in tables and in the Financial Statements are used to represent negative numbers.
Except where otherwise indicated, figures included in this Report relating to pharmaceutical product market sizes and market shares are obtained from syndicated industry sources, primarily IMS Health (IMS), a market research firm internationally recognised by the pharmaceutical industry. The 2008 market share figures included in this report are based primarily on data obtained from an online IMS database.
IMS data may differ from that compiled by the Group with respect to its own products. Of particular significance in this regard are the following: (1) AstraZeneca publishes its financial results on a financial year and quarterly interim basis, whereas IMS issues its data on a monthly and quarterly basis; (2) the online IMS database is updated quarterly and uses the average exchange rates for the relevant quarter; (3) IMS data from the US is not adjusted for Medicaid and similar state rebates; and (4) IMS sales data are compiled using actual wholesaler data and data from statistically representative panels of retail and hospital pharmacies, which data are then projected by IMS to give figures for national markets.
References to prevalence of disease have been derived from a variety of sources and are not intended to be indicative of the current market or any potential market for AstraZeneca’s pharmaceutical products since, among other things, there may be no correlation between the prevalence of a disease and the number of individuals who are treated for such a disease.
| | | |
| Terms used in the Annual Report | | |
| and Form 20-F Information | | US equivalent or brief description |
| |
| Accruals | | Accrued expenses |
| |
| Allotted | | Issued |
| |
| Bank borrowings | | Payable to banks |
| |
| Called-up share capital | | Issued share capital |
| |
| Creditors | | Liabilities/payables |
| |
| Current instalments of loans | | Long term debt due within one year |
| |
| Debtors | | Receivables and prepaid expenses |
| |
| Earnings | | Net income |
| |
| Finance lease | | Capital lease |
| |
| Fixed asset investments | | Non-current investments |
| |
| Freehold | | Ownership with absolute rights in perpetuity |
| |
| Interest receivable | | Interest income |
| |
| Interest payable | | Interest expense |
| |
| Loans | | Long term debt |
| |
| Prepayments | | Prepaid expenses |
| |
| Profit | | Income |
| |
| Profit and loss account | | Income statement/consolidated statement of income |
| |
| Reserves | | Retained earnings |
| |
| Short term investments | | Redeemable securities and short term deposits |
| |
| Share premium account | | Premiums paid in excess of par value of Ordinary Shares |
| |
| Statement of recognised income and expense | | Statement of comprehensive income |
| |
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008
| | | | | | | | | | | | | | | | | | | | | | | | | |
NORTH AMERICA | | | ESTABLISHED REST OF WORLD | | | EMERGING REST OF WORLD |
US | | | Canada | | | Western Europe | | | Japan | | | Other Established | | | Emerging Europe | | | China | | | Emerging | | | Other Emerging |
| | | | | | | | | | | | | | | | | | | | | | Asia Pacific | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | |
| US | | | Canada | | | Austria | | | Japan | | | Australia | | | Czech Republic | | | China | | | India | | | Algeria |
| | | | | | | Belgium | | | | | | New Zealand | | | Estonia | | | Hong Kong | | | Malaysia | | | Argentina |
| | | | | | | Denmark | | | | | | | | | Latvia | | | | | | Philippines | | | Brazil |
| | | | | | | Finland | | | | | | | | | Poland | | | | | | South Korea | | | Central America |
| | | | | | | France | | | | | | | | | Slovakia | | | | | | Taiwan | | | Chile |
| | | | | | | Germany | | | | | | | | | Turkey | | | | | | Thailand | | | Colombia |
| | | | | | | Greece | | | | | | | | | | | | | | | | | | Egypt |
| | | | | | | Holland | | | | | | | | | | | | | | | | | | Lebanon |
| | | | | | | Italy | | | | | | | | | | | | | | | | | | Mexico |
| | | | | | | Norway | | | | | | | | | | | | | | | | | | Morocco |
| | | | | | | Portugal | | | | | | | | | | | | | | | | | | Peru |
| | | | | | | Spain | | | | | | | | | | | | | | | | | | Saudi Arabia |
| | | | | | | Sweden | | | | | | | | | | | | | | | | | | South Africa |
| | | | | | | UK | | | | | | | | | | | | | | | | | | UAE |
| | | | | | | | | | | | | | | | | | | | | | | | | Venezuela |
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | Iceland | | | | | | | | | Albania | | | | | | Afghanistan | | | |
| | | | | | | Luxembourg | | | | | | | | | Belarus | | | | | | Bangladesh | | | |
| | | | | | | | | | | | | | | | Bosnia-Herzegovina | | | | | | Brunei | | | |
| | | | | | | | | | | | | | | | Bulgaria | | | | | | Cambodia | | | |
| | | | | | | | | | | | | | | | Croatia | | | | | | Indonesia | | | |
| | | | | | | | | | | | | | | | Georgia | | | | | | Laos | | | |
| | | | | | | | | | | | | | | | Hungary | | | | | | Myanmar | | | |
| | | | | | | | | | | | | | | | Macedonia | | | | | | Nepal | | | |
| | | | | | | | | | | | | | | | Romania | | | | | | Papua New Guinea | | | |
| | | | | | | | | | | | | | | | Russia Serbia/Montenegro | | | | | | Singapore | | | |
| | | | | | | | | | | | | | | | | | | | | | Sri Lanka | | | |
| | | | | | | | | | | | | | | | | | | | | | Vietnam | | | |
Emerging Markets means Emerging Rest of World.
Established Markets means North America and Established Rest of World.
IMS Data is not included for those countries listed in the lower table.
The following abbreviations and expressions have the following meanings when used in this report:
abbreviated new drug application (ANDA)
A marketing approval application for a generic drug submitted to the US Food and Drug Administration.
abbreviated new drug submission (ANDS)
A marketing approval application for a generic drug submitted in Canada.
adjuvantAn agent that modifies the effect of other agents (for example drugs and vaccines) while having few if any direct effects when given by itself; it operates like a catalyst in chemical reactions.
ADRAmerican Depositary Receipt evidencing title to an ADS.
ADSAmerican Depositary Share representing one underlying Ordinary Share.
agonistA substance capable of binding to a molecular target to trigger a response.
angiotensin IIA hormone that causes blood vessels to narrow and thereby raises blood pressure.
antagonistA substance capable of binding to a molecular target to neutralise or counteract a reaction.
anti-cytokine monoclonal antibodies (MAbs)A group of large molecule compounds which can block interactions between cytokines and their receptors. Cytokines are a class of signalling molecule, used in cellular communication, similar to hormones and neurotransmitters preventing them from functioning
AstraZenecaAstraZeneca PLC and its subsidiaries.
biomarkerA characteristic that can be measured objectively and evaluated as an indicator of normal biological, or pathogenic processes, or pharmacological responses to a therapeutic intervention.
biopharmaceuticals/biologicsA class of medicines derived from proteins usually produced naturally by living organisms in response to disease, for example antibodies.
bipolar disorderAny of several mood disorders, usually characterised by alternating episodes of depression and mania or by episodes of depression alternating with mild non-psychotic excitement.
bipolar I disorderA sub-set of bipolar patients who have experienced one or more manic or mixed episodes.
biosimilarsFollow-on biopharmaceuticals that are biologically similar to an existing medicine.
BLA (Biological Licence Application)Granting of the licence certifies that biological product is safe, pure and potent; and the facility in which it is manufactured meets standards designed to ensure that it continues to be safe, pure and potent.
BoardThe Board of Directors of AstraZeneca PLC.
CATCambridge Antibody Technology Group plc.
CER Constant exchange rates.
CompanyAstraZeneca PLC.
Complete Response LetterA complete response is now issued by the FDA when it has completed its evaluation of a regulatory submission.
cost growth ratesPercentage growth of a particular cost category over the comparable cost category for the previous year.
depositaryJPMorgan Chase Bank, as depositary under the deposit agreement pursuant to which the ADRs are issued.
DirectorA director of the Company.
earnings per share (EPS)Profit for the year after tax and minority interests, divided by the weighted
average number of Ordinary Shares in issue during the year.
efficacyThe beneficial effect of a drug refers to the potential maximum therapeutic response that a drug can produce.
EFPIAThe European Federation of Pharmaceutical Industries and Associations.
EMEAThe European Medicines Agency.
Emerging MarketsEmerging Rest of World, see table above.
Established MarketsNorth America and Established Rest of World, see table above.
EUEuropean Union.
finance income and expenseThis includes interest earned and payable, and similar items.
first good laboratory practice (FGLP)The point at which a compound undergoes the first pre-clinical study that is required for regulatory approval; this marks entry into the development pipeline.
first-line therapyTreatment given to a newly diagnosed patient, who has therefore not yet been treated; it is the initial treatment of a disease, sign or symptom.
Food and Drug Administration (FDA)Part of the US Department of Health and Human Services Agency, which is the regulatory authority for all pharmaceuticals (including biologics and vaccines) and medical devices in the US.
generalised anxiety disorder (GAD)A neurotic illness characterised by chronic and persistent apprehension and tension.
gross marginThe margin, as a percentage, by which sales exceed the cost of sales, calculated by dividing the difference between the two by the sales figure.
ASTRAZENECA ANNUAL REPORT AND FORM 20-F INFORMATION 2008