 June 2015 1 Exhibit 99.2 |
 BioReference Laboratories Snapshot 2 Division Key Products Markets Physician Offices, Health Facilities, Correctional Facilities Flow Cytometry, IHC, FISH, ISH, Microarray, Morphology Hematologists, Oncologists, Hospital Pathologists Obstetricians, Gynecologists, Maternal-Fetal Medicine NextGen & Sanger Sequencing, Array Comparative Genomic Hybridization (aCGH) Geneticists, Medical Centers, Children’s Hospitals, Physicians managing genetic disease Automated, high volume IVD, Health Informatics, HIV, HCV, other Molecular Dx Physicians’ offices in Spanish- Speaking Communities Oncology Women’s Health Automated, high volume IVD, Health Informatics, HIV, HCV, other Molecular Dx Image-Directed Cytology (PAP) HPV, STI |
BioReference Laboratories + OPKO Health = Strong Customer and Product Benefits • BioReference Laboratories – The 3 rd largest clinical laboratory in the US (after Quest and LabCorp) with scale to reach large markets in the Northeastern US, Florida, California, and Texas – Fast growing, innovative molecular diagnostics business with expertise in technologies enabling – Full service routine testing menu for primary care physicians and specialists – High value testing capabilities serving key specialties (Women’s Health, Oncology) and other clinicians managing genetic disease – Team of about 400 sales, customer service and marketing personnel – Patient Service Centers with blood draw capability at over 170 locations • OPKO Health – Specialists in Uropathology, including anatomic pathology, IHC, cytology and FISH, serving the diagnostic testing needs of urologists – The 4Kscore Test for aggressive prostate cancer – • Combined Strengths – Nation wide insurance contract coverage and large payer relationships – Access to the Primary Care Physician market for 4Kscore and Claros platform – Access to over 170 BRL patient service centers for 4Kscore blood draw – Blood specimen transportation network and high volume testing operation – Addition of urology to BRL specialty diagnostic services capability 3 meaningful molecular medicine (NextGen sequencing, aCGH) Claros 1 for in-office immunoassay results in 10 minutes from a fingerstick blood sample |
 What is the 4Kscore Test? 4 + Age, DRE, and prior biopsy status % risk of having aggressive prostate cancer for an individual patient Components Components OPKO { 4 kallikrein levels: Total PSA Free PSA Intact PSA hK2 Results Results OPKO ALGORITHM |
 |
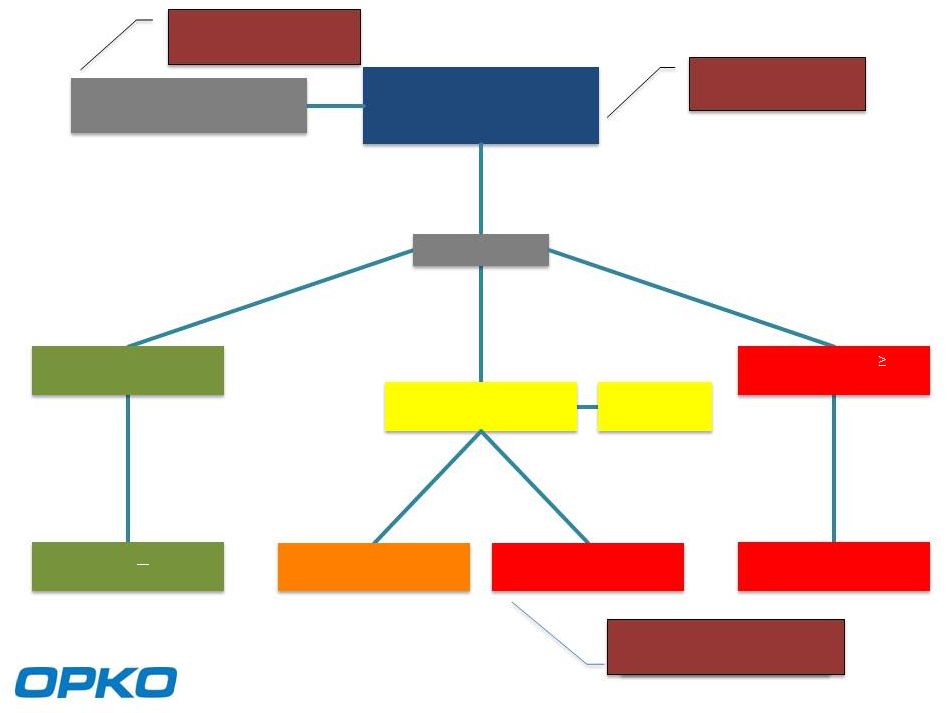 Monitor w PSA or PCA3, ConfirmMDx Monitor w PSA or PCA3, ConfirmMDx OncotypeDx or Prolaris OncotypeDx or Prolaris 100,000 Standard TRUS or mp MRI-Guided Biopsy 100,000 Standard TRUS or mp MRI-Guided Biopsy Negative Biopsy Histopathology Negative Biopsy Histopathology 55,000 25,000 Gleason score 6 Histopathology Gleason score 6 Histopathology Biopsy Result Biopsy Result Primary Treatment Surgery / Radiation Primary Treatment Surgery / Radiation Gleason score 7 Histopathology Gleason score 7 Histopathology 20,000 140,000 Abnormal PSA or DRE 140,000 Abnormal PSA or DRE 700,000 PSA Screenings 700,000 PSA Screenings 4,000 Serious Infections 4,000 Serious Infections 12,000 with Confirmed Indolent Disease 12,000 with Confirmed Indolent Disease Active Surveillance Active Surveillance 7,500 Primary Treatment Surgery / Radiation Primary Treatment Surgery / Radiation 17,500 6 |
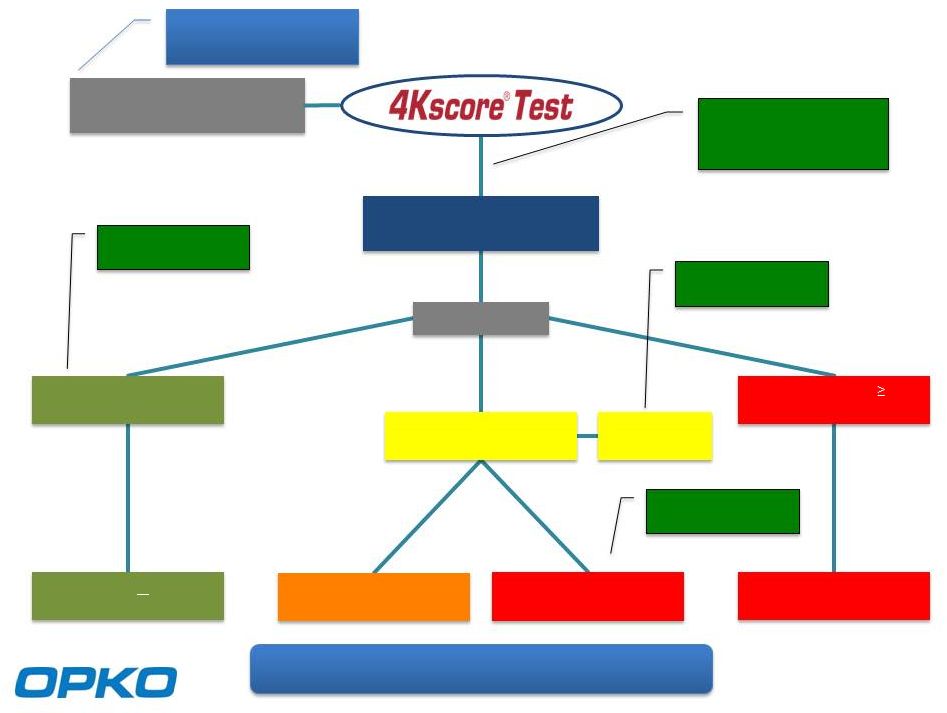 Monitor w PSA if abnormal, 4Kscore Monitor w PSA if abnormal, 4Kscore Biopsy Result Biopsy Result Primary Treatment Surgery / Radiation Primary Treatment Surgery / Radiation Active Surveillance Active Surveillance Primary Treatment Surgery / Radiation Primary Treatment Surgery / Radiation Negative Biopsy Histopathology Negative Biopsy Histopathology Gleason score 7 Histopathology Gleason score 7 Histopathology Gleason score 6 Histopathology Gleason score 6 Histopathology OncotypeDx or Prolaris OncotypeDx or Prolaris Standard TRUS or mp MRI-Guided Biopsy Standard TRUS or mp MRI-Guided Biopsy 36% Biopsy Reduction $78M Savings 36% Biopsy Reduction $78M Savings 29% Reduction $16 M Savings 29% Reduction $16 M Savings 29% Reduction $146 M Savings 29% Reduction $146 M Savings $143 M net savings / $1,400 per patient $143 M net savings / $1,400 per patient 100,000 Prostate Biopsy Patients 100,000 Prostate Biopsy Patients 700,000 PSA Screenings 700,000 PSA Screenings 7 51% Reduction $22 M Savings 51% Reduction $22 M Savings |
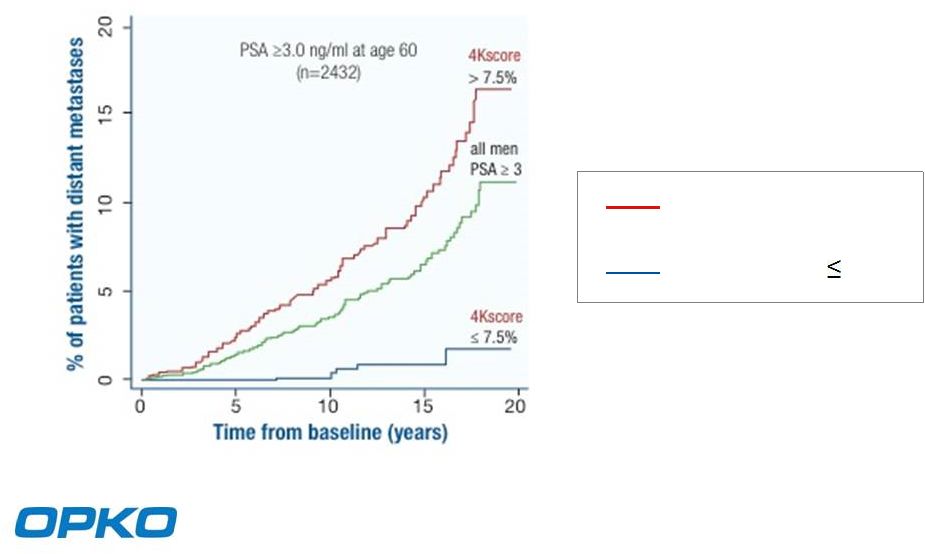 The 4Kscore Test Predicts Metastases Within 20 years 1 16% < 2% 4Kscore Test > 7.5% 4Kscore Test 7.5% ¹ Improving the Specificity of Screening for Lethal Prostate Cancer Using Prostate-specific Antigen and a Panel of Kallikrein Markers: A Nested Case–Control Study European Urology (in press) 8 |
 4Kscore Commercial Update • 4Kscore ProtecT study published – over 6,000 subjects • Total dossier for the 4Kscore Test now includes clinical data on over 22,000 subjects in 12 peer reviewed publications • Over 900 US urologists have used the 4Kscore test in routine practice • American Urological Society Meeting in New Orleans May, 2015 – six podium or poster presentations • Next milestones: – NCCN Early Detection Guidelines 3Q2015 – CPT Code active July 1, 2015 – billing Medicare and Private Insurance – Medicare and private insurance coverage for test: 2015/16 9 |
 Claros ® 1: Rapid Testing in the Physician Office 10 |
 Claros 1 Platform Addresses Large Testing Markets Commercial Leverage with Bio-Reference Sales Team • Testosterone – US test volume: 15 million tests, $525 M – 510(k) filing in 1H2016 • PSA – US test volume: 30 million tests, $750 M – Intended use to focus on detection claim – Modular PMA filing in 1H2106 • Renal panel in Development – Vitamin D • US test volume: 70 million tests, $3.5 B – iPTH – Cystatin C • Lyme Disease – Claros 1 selected as platform for an NIH funded diagnostic development – OPKO has all rights to diagnostic test ($120M US Market) 11 |