Filing under Rule 425 under
the U.S. Securities Act of 1933
Filing by: Daiichi Pharmaceutical Co., Ltd.
Subject Company: Daiichi Pharmaceutical Co., Ltd.
and Sankyo Company, Limited
SEC File No. 132-02290

To Our Shareholders
We are providing this pamphlet to explain the key points of the business integration of Daiichi
Pharmaceutical Co., Ltd. and Sankyo Company, Limited.
We would sincerely appreciate your understanding of the objectives of the business integration and ask that you give us your support by voting in favor of the integration proposal at the 127th General Meeting of Shareholders, to be held on June 29, 2005.
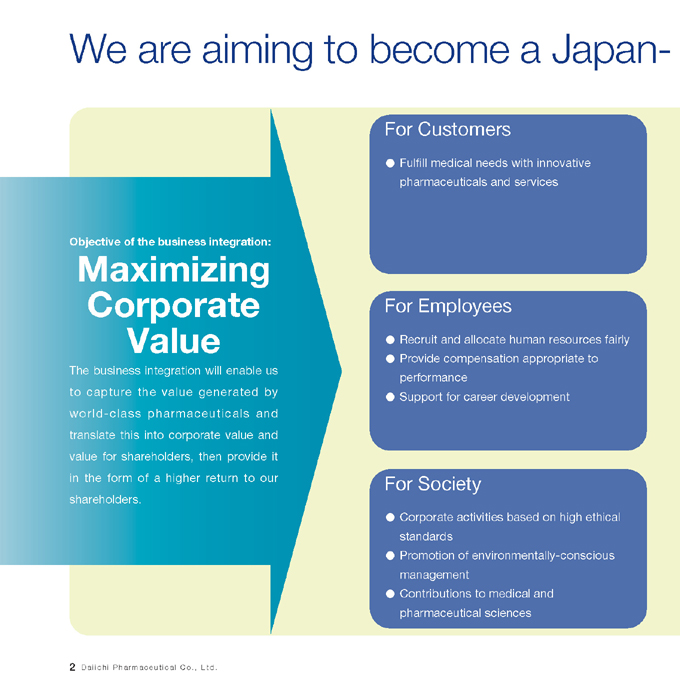
We are aiming to become a Japan-
Objective of the business integration:
Maximizing Corporate Value
The business integration will enable us to capture the value generated by world-class pharmaceuticals and translate this into corporate value and value for shareholders, then provide it in the form of a higher return to our shareholders.
For Customers
Fulfill medical needs with innovative pharmaceuticals and services
For Employees
Recruit and allocate human resources fairly Provide compensation appropriate to performance Support for career development
For Society
Corporate activities based on high ethical standards Promotion of environmentally-conscious management Contributions to medical and pharmaceutical sciences
2 Daiichi Pharmaceutical Co., Ltd.

based Global Pharma Innovator.
For Our Shareholders
Acceleration of Growth in Profits and Shareholders’ Equity
By consolidating our competitive strengths through the business integration, we plan to attain higher growth in profits and accelerated expansion in shareholders’ equity. Our target is to attain 15% annual growth in operating income and expand it to approximately ¥255 billion by fiscal 2009, two times the level for fiscal 2004.
Operating Income
(¥ billion)
127
Fiscal 2004
255
Target
Fiscal 2009
Raise Return to Shareholders
To return profits we generate to our shareholders, we will aim to achieve a ratio of dividends on equity (DOE) of 5% by fiscal 2009. In addition, for fiscal 2005, we plan to secure cash dividends above the level for the previous fiscal year and to make steady increases in dividends in the years ahead.
DOE Ratio
As a result of the business integration, growth in profits will show marked acceleration
DOE target
5%
Fiscal 2009
Note: The figure of operating income for fiscal 2004 is combined data obtained by adding figures for the two companies. The DOE ratio is cash dividends per share divided by shareholders’ equity per share. The dividend per share in fiscal 2003 was ¥30, and the dividend scheduled for fiscal 2004 will be ¥40. These cash dividends represent DOE ratios of 2.0% and 2.6%, respectively.
Daiichi Pharmaceutical Co., Ltd . 3

Increasing Shareholder Value
The objective of the business integration is the creation of a Japan-based Global Pharma Innovator which will have the mission of providing a steady stream of innovative products and services that satisfy unmet medical needs and which will occupy a unique competitive position in the world’s major markets.
We believe positive sales and cost synergies resulting from the business integration will substantially outweigh the possible negative synergies and integration costs, and will increase the shareholder value of Daiichi and Sankyo.
The most important objective of the business integration will be to secure potential synergies from the Global Pharma Innovator platform. Specifically, the integration will combine the R&D capabilities of Daiichi, which created the broad-spectrum oral antibacterial agent Cravit® (levofloxacin), and Sankyo, which created the antihyperlipidemic agent Mevalotin® (pravastatin). We believe this combination will yield true synergies and enable the combined companies to bring promising drugs in their pipelines to market as quickly as possible.
As a result of the business integration, we are confident that the corporate value of Daiichi will expand on a sustained and stable basis and that this will lead to the maximization of shareholder value.
Aggregate value of Daiichi shares
Aggregate value of Sankyo shares
Possible negative synergies*
Integration costs
Sales and cost synergies
Potential synergies from Global Pharma Innovator platform
Estimated shareholder value after integration (Former Daiichi)
Estimated shareholder value after integration (Former Sankyo)
Targeted increase in shareholder value
Note: The aggregate values of Daiichi and Sankyo shares are calculated based on the closing prices on February 18, 2005.
* Risks associated with the merger, such as a decline in sales and/or profits due to changes in corporate control or product cannibalization
4 Daiichi Pharmaceutical Co., Ltd.

Capabilities for Creating Innovative New Drugs
Daiichi has managed its activities based on the policy of concentrating its management resources in priority domains, focusing especially on infectious disease and cardiovascular fields. The proposed business integration will represent a continuation and extension of this management policy. Sankyo is a leading pharmaceutical company in Japan conducting an active R&D program focusing on the cardiovascular field in which Daiichi is also active. We are confident that integrating the management of the product portfolios and drug development pipelines will lead to the realization of major synergies and to significant improvements in pharmaceutical development capabilities and profitability.
Strong R&D Franchise Categories
Daiichi
Infectious diseases
Cancer
Immunological/allergic diseases
Cardiovascular diseases/Thrombosis and arteriosclerosis
Bone/joint diseases
Diabetes and metabolic disorders
Sankyo
Total R&D expenditure target for fiscal 2009 ¥170 billion
Global Drug Development Pipeline: Example of Activities in the United States
Development candidates Patient category Phase 1 Phase 2 Phase 3
CS-3030 SUN N8075 DU-176b CS-505 CS-747
DZ-697b Cardiovascular diseases SUN N4057 HGF
DX-9065a
CS-011 SNK-860 WelChol diabetes Diabetes CS-917
D100 DX-619 DU-6859a Infectious diseases CS-8958 CS-758
CS-1008 DJ-927 Cancer CS-7017
CS-0777 Immunological/allergic diseases DW-908e CS-003 Bone/joint diseases CS-706
Note: Development candidates include those for which U.S. clinical trials have been scheduled.
5 Daiichi Pharmaceutical Co., Ltd.

Competitive Strengths in the Japanese Market
As a result of the integration, domestic marketing capabilities will be bolstered substantially to give the integrated company a strong presence in key areas, particularly in terms of the number of MRs and sales details. We are making plans for initiatives that will realize sales synergies drawing on this marketing strength. In specific terms, beginning in October of this year, collaboration between Daiichi and Sankyo is expected to boost sales of Olmetec—which has been named “Best in Class” among angiotensin II receptor blockers (ARBs)—to the level of the three previously launched products. By fiscal 2009, we plan to develop Olmetec to establish “Best in Class” status in Japan, with domestic sales of ¥100 billion (based on current drug prices)
Number of MRs in Japan
(Persons)
Pfizer Daiichi Sankyo Astellas AstraZenica Novartis Chugai Takeda GlaxoSmithKlein Banyu Mitsubishi Pharma Shionogi Yamanouchi Daiichi Sankyo Fujisawa
0 500 1,000 1,500 2,000 2,500 3,000 3,500
Source: Special edition of MIX
Trends in Sales of ARBs1
Three previously launched products2
Raise to the level of the three previously launched products
Olmetec
Sales
Trends
Notes:
1. Angiotensin II receptor blockers (blood pressure lowering agents) 2. Nu-Lotan, Blopress, Diovan
6 Daiichi Pharmaceutical Co., Ltd.
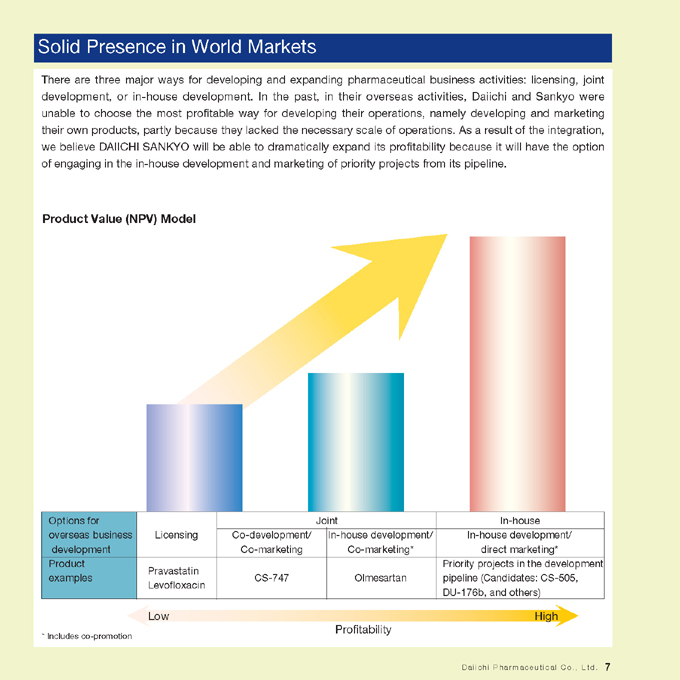
Solid Presence in World Markets
There are three major ways for developing and expanding pharmaceutical business activities: licensing, joint development, or in-house development. In the past, in their overseas activities, Daiichi and Sankyo were unable to choose the most profitable way for developing their operations, namely developing and marketing their own products, partly because they lacked the necessary scale of operations. As a result of the integration, we believe DAIICHI SANKYO will be able to dramatically expand its profitability because it will have the option of engaging in the in-house development and marketing of priority projects from its pipeline.
Product Value (NPV) Model
Options for overseas business development
Licensing
Co-development/ Co-marketing
Joint In-house development/ Co-marketing*
In-house development/ In-house direct marketing*
Produc examples
Pravastatin Levofloxacin
CS-747
Olmesartan
Priority projects in the development pipeline (Candidates: CS-505, DU-176b, and others)
Low High Profitability
* Includes co-promotion
Daiichi Pharmaceutical Co., Ltd. 7
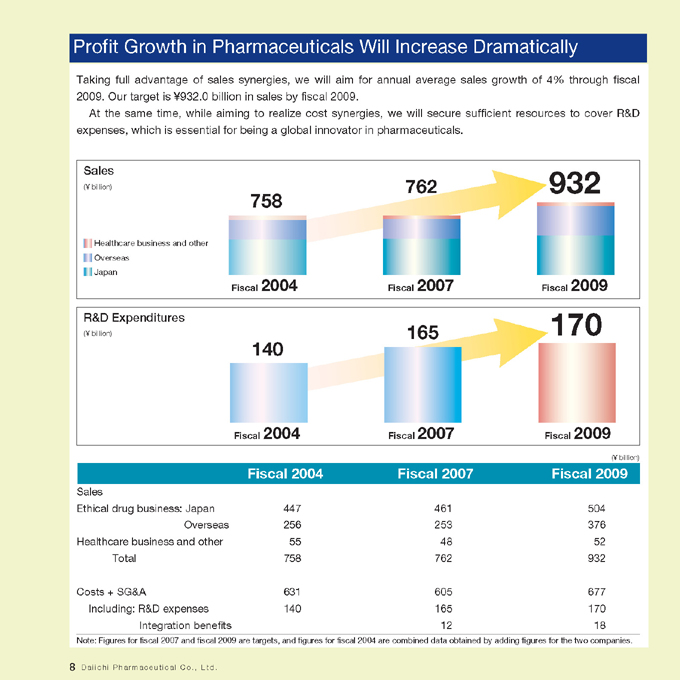
Profit Growth in Pharmaceuticals Will Increase Dramatically
Taking full advantage of sales synergies, we will aim for annual average sales growth of 4% through fiscal 2009. Our target is ¥932.0 billion in sales by fiscal 2009.
At the same time, while aiming to realize cost synergies, we will secure sufficient resources to cover R&D expenses, which is essential for being a global innovator in pharmaceuticals.
Sales
(¥ billion) 762 932
758
Healthcare business and other
Overseas
Japan
Fiscal 2004 Fiscal 2007 Fiscal 2009
R&D Expenditures 170
(¥ billion) 165
140
Fiscal 2004 Fiscal 2007 Fiscal 2009
Fiscal 2004 Fiscal 2007 Fiscal 2009
Sales
Ethical drug business: Japan 447 461 504
Overseas 256 253 376
Healthcare business and other 55 48 52
Total 758 762 932
Costs + SG&A 631 605 677
Including: R&D expenses 140 165 170
Integration benefits 12 18
Note: Figures for fiscal 2007 and fiscal 2009 are targets, and figures for fiscal 2004 are combined data obtained by adding figures for the two companies.
8 Daiichi Pharmaceutical Co., Ltd.
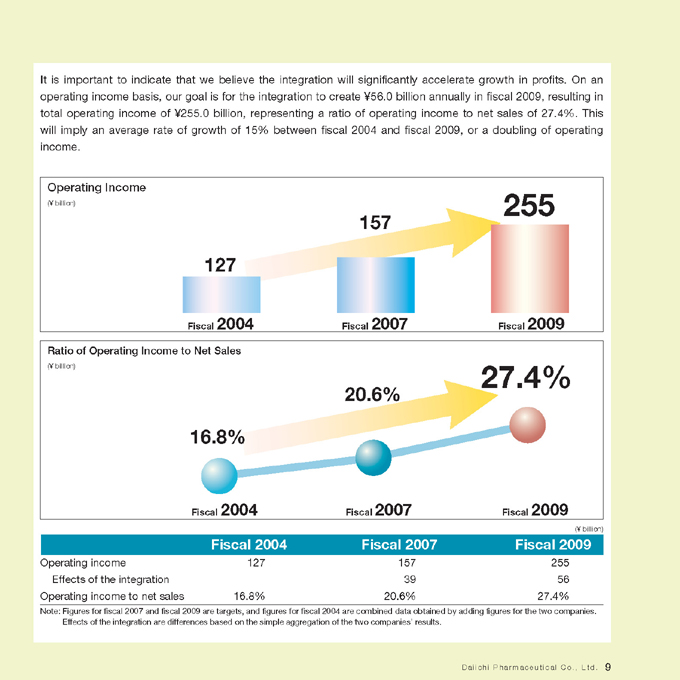
It is important to indicate that we believe the integration will significantly accelerate growth in profits. On an operating income basis, our goal is for the integration to create ¥56.0 billion annually in fiscal 2009, resulting in total operating income of ¥255.0 billion, representing a ratio of operating income to net sales of 27.4%. This will imply an average rate of growth of 15% between fiscal 2004 and fiscal 2009, or a doubling of operating income.
Operating Income
(¥ billion)
127
Fiscal 2004
157
Fiscal 2007
255
Fiscal 2009
Ratio of Operating Income to Net Sales
(¥ billion)
16.8%
Fiscal 2004
20.6%
Fiscal 2007
27.4%
Fiscal 2009
(¥ billion)
Fiscal 2004 Fiscal 2007 Fiscal 2009
Operating income 127 157 255
Effects of the integration 39 56
Operating income to net sales 16.8% 20.6% 27.4%
Note: Figures for fiscal 2007 and fiscal 2009 are targets, and figures for fiscal 2004 are combined data obtained by adding figures for the two companies.
Effects of the integration are differences based on the simple aggregation of the two companies’ results.
Daiichi Pharmaceutical Co., Ltd. 9
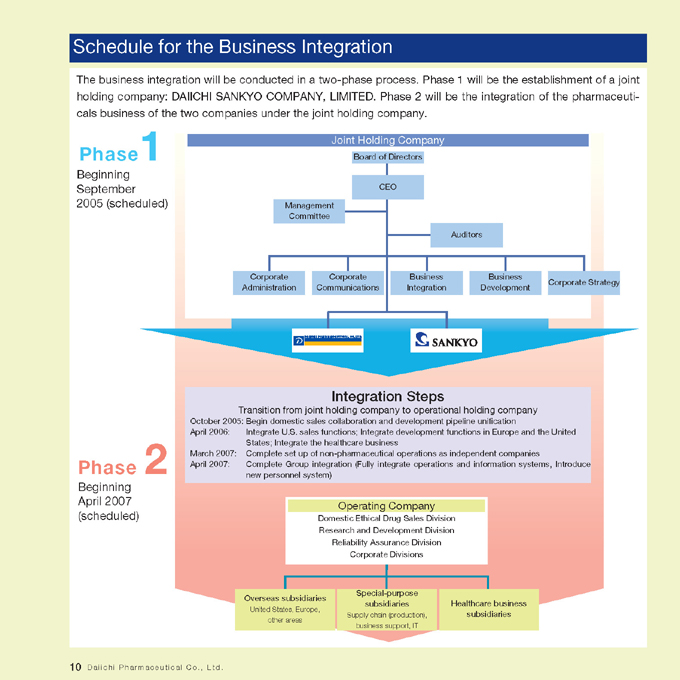
Schedule for the Business Integration
The business integration will be conducted in a two-phase process. Phase 1 will be the establishment of a joint holding company: DAIICHI SANKYO COMPANY, LIMITED. Phase 2 will be the integration of the pharmaceuticals business of the two companies under the joint holding company.
Phase 1
Beginning September 2005 (scheduled)
Joint Holding Company
Board of Directors
CEO
Management Committee
Auditors
Corporate Administration
Corporate Communications
Business Integration
Business Development
Corporate Strategy
Integration Steps
Transition from joint holding company to operational holding company
October 2005:
Begin domestic sales collaboration and development pipeline unification
April 2006:
Integrate U.S. sales functions; Integrate development functions in Europe and the United
States; Integrate the healthcare business
March 2007:
Complete set up of non-pharmaceutical operations as independent companies
April 2007:
Complete Group integration (Fully integrate operations and information systems, Introduce
new personnel system)
Phase 2
Beginning April 2007 (scheduled)
Operating Company
Domestic Ethical Drug Sales Division Research and Development Division Reliability Assurance Division Corporate Divisions
Overseas subsidiaries
United States, Europe, other areas
Special-purpose subsidiaries
Supply chain (production), business support, IT
Healthcare business subsidiaries
10 Daiichi Pharmaceutical Co., Ltd.

Outline of DAIICHI SANKYO COMPANY LIMITED (planned)
Name in Japanese
Daiichi Sankyo Kabushiki Kaisha
Name in English
DAIICHI SANKYO COMPANY, LIMITED
Head office
5-1, Nihonbashi-honcho 3-chome, Chuo-ku, Tokyo, Japan
Date of delisting of predecessor companies
September 21, 2005
Date of stock transfer
September 28, 2005
Date of new listing
September 28, 2005
Stock transfer ratio
New company 1.159 : Daiichi Pharmaceutical 1 New company 1 : Sankyo 1
Stock exchange listings
Tokyo, Osaka, Nagoya
Board of Directors
Representative Director and Chairman
Kiyoshi Morita
(Current President & Representative Director of Daiichi Pharmaceutical)
Representative Director, President and Chief Executive Officer
Takashi Shoda
(Current President & Representative Director of Sankyo)
Directors
Hiroyuki Nagasako
(Current Executive Vice President of Daiichi Pharmaceutical)
Hideho Kawamura
(Current Executive Vice President & Representative Director of Sankyo)
Yasuhiro Ikegami
(Current Executive Vice President & Representative Director of Sankyo)
Tsutomu Une
(Current Managing Director of Daiichi)
Outside Directors
Kunio Nihira
(Current Director of Sankyo)
Yoshifumi Nishikawa
(Current President and Chief Executive Officer of Sumitomo Mitsui Banking Corporation)
Jotaro Yabe
(Current Professor of Jissen Women’s University)
Katsuyuki Sugita
(Current Director of Sankyo)
Outline of Sankyo Co., Ltd.
Name in Japanese
Sankyo Kabushiki Kaisha
Name in English
SANKYO COMPANY, LIMITED
Commencement of operations
1899
Company established
March 1913
Representative
Takashi Shoda, President & Representative Director
Capital
¥68,793 million (As of March 31, 2005)
Lines of business
Manufacturing, marketing, exporting, importing of pharmaceuticals, quasi- pharmaceuticals, and medical devices
Number of employees
11,444 (On a consolidated basis, as of March 31, 2005)
Outline of consolidated
Net sales: ¥587,830 million
financial results in fiscal 2004
Operating income: ¥84,925 million
Ordinary income: ¥82,506 million
Daiichi Pharmaceutical Co., Ltd. 11

Filings with the U.S. SEC Daiichi Pharmaceutical Co., Ltd. and Sankyo Company, Limited have filed a registration statement on Form F-4 with the U.S. SEC in connection with the proposed business integration of Daiichi and Sankyo under a new holding company by way of a joint stock transfer. The Form F-4 contains a prospectus and other documents. After the Form F-4 has been declared effective, Daiichi and Sankyo plan to mail the prospectus contained in the Form F-4 to their U.S. shareholders prior to the shareholders’ meetings at which the share exchange will be voted upon. The Form F-4 and prospectus contain important information about Daiichi and Sankyo, the joint stock transfer and related matters. U.S. shareholders of Daiichi and Sankyo are urged to read the Form F-4, the prospectus and the other documents filed with the U.S. SEC in connection with the joint stock transfer carefully before they make any decision at the shareholders’ meeting with respect to the joint stock transfer. The Form F-4, the prospectus and all other documents filed with the U.S. SEC in connection with the joint stock transfer are available, free of charge, on the U.S. SEC’s web site at www.sec.gov. In addition, the prospectus and all other documents filed with the U.S. SEC in connection with the business integration are available to shareholders, free of charge, by calling, writing or e-mailing:
Sankyo Company, Limited
Daiichi Pharmaceutical Co., Ltd.
Mr. Shigemichi Kondo
Mr. Toshio Takahashi
Corporate Communications Department
Corporate Communications Department
3-5-1, Nihonbashi Honcho
14-10 Nihonbashi, 3-chome
Chuo-ku, Tokyo 103-8426, Japan
Chuo-ku, Tokyo 103-8234, Japan
Telephone: 81-3-5255-7034
Telephone: 81-3-3273-7107
E-mail: shige-k@sankyo.co.jp
E-mail: andokb5o@daiichipharm.co.jp
You may read and copy any reports and other information filed with, or submitted to, the U.S. SEC at the U.S. SEC’s public reference rooms at 100 F Street, N.E., Washington, D.C. 20549 or at the other public reference rooms in New York, New York and Chicago, Illinois. Please call the U.S. SEC at 1-800-SEC-0330 for further information on public reference rooms. Filings with the U.S. SEC also are available to the public from commercial document-retrieval services and at the web site maintained by the U.S. SEC at http//www.sec.gov.
Forward-Looking Statements This communication contains forward-looking information and statements about Daiichi and Sankyo and their combined businesses after completion of the joint stock transfer. Forward-looking statements are statements that are not historical facts. These statements include financial projections and estimates and their underlying assumptions, statements regarding plans, objectives and expectations with respect to future operations, products and services, and statements regarding future performance. Forward-looking statements are generally identified by the words “expects,” “anticipates,” “believes,” “intends,” “estimates” and similar expressions. Although the management of Daiichi and Sankyo believe that the expectations reflected in such forward-looking statements are reasonable, investors and holders of Daiichi and Sankyo securities are cautioned that forward-looking information and statements are subject to various risks and uncertainties, many of which are difficult to predict and generally beyond the control of Daiichi and Sankyo, that could cause actual results and developments to differ materially from those expressed in, or implied or projected by, the forward-looking information and statements. These risks and uncertainties include those discussed or identified in the public filings with the SEC and the local filings made by Daiichi and Sankyo, including those listed under “Cautionary Statement Concerning Forward-Looking Statements” and “Risk Factors” in the prospectus included in the registration statement on Form F-4 that Daiichi and Sankyo have filed with the U.S. SEC. Other than as required by applicable law, neither Daiichi nor Sankyo undertakes any obligation to update or revise any forward-looking information or statements.











