
Will Charlton1, Daniela Rogoff1, Eric Humphriss1, Kelly DiTrapani1, Philippe F. Backeljauw2 1Versartis, Inc, Menlo Park, CA; 2Cincinnati Children's Hospital Medical Center, Cincinnati, OH, USA HbA1c over Two Years of Treatment with Somavaratan (VRS-317) in Children with Growth Hormone Deficiency (GHD) in the VISTA Study Exhibit 99.1

Disclosures Will Charlton, MD, Daniela Rogoff, MD, PhD, Eric Humphriss, MBA, and Kelly Di Trapani, RN, BSN, BA, are employees of Versartis, Inc Philippe F. Backeljauw, MD, is a consultant for Versartis, Inc Somavaratan (VRS-317) is an investigational agent
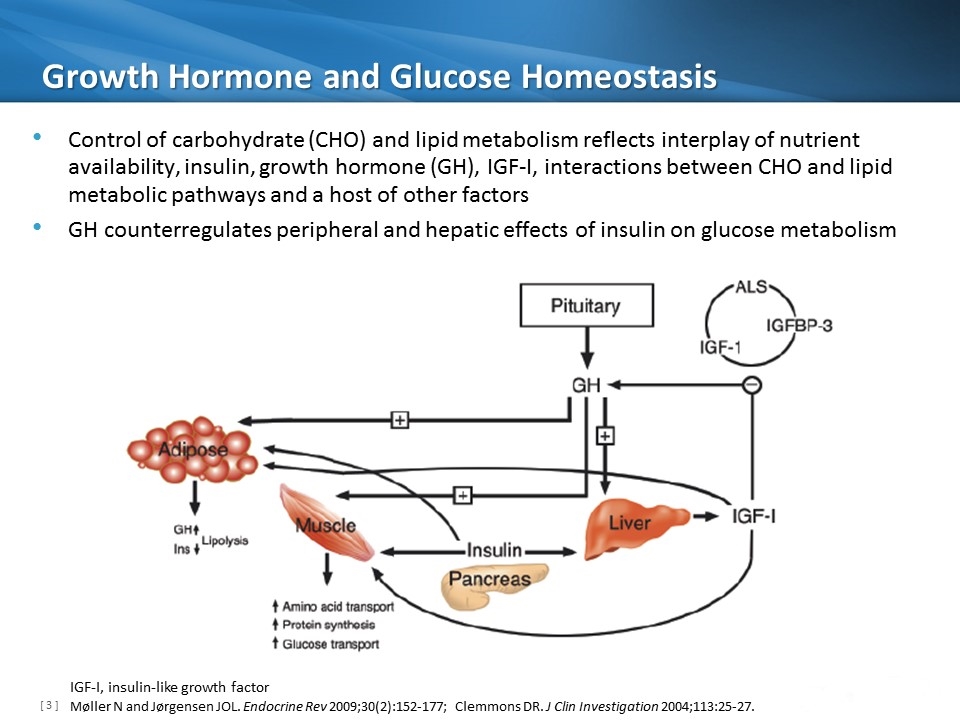
Growth Hormone and Glucose Homeostasis Møller N and Jørgensen JOL. Endocrine Rev 2009;30(2):152-177; Clemmons DR. J Clin Investigation 2004;113:25-27. Control of carbohydrate (CHO) and lipid metabolism reflects interplay of nutrient availability, insulin, growth hormone (GH), IGF-I, interactions between CHO and lipid metabolic pathways and a host of other factors GH counterregulates peripheral and hepatic effects of insulin on glucose metabolism IGF-I, insulin-like growth factor

Endogenous GH Exerts Different Effects during Fasting & Fed States In the fed state, GH-induced stimulation of IGF-I and insulin is important for anabolic storage and growth of: Lean Body Mass (LBM) Adipose tissue Glycogen reserves During fasting/catabolic states, GH predominantly stimulates the release and oxidation of free fatty acids (FFA), which leads to: Decreased glucose and protein oxidation Preservation of LBM and glycogen stores GH-induced insulin resistance protects against hypoglycemia: Development of “stress diabetes” during fasting and inflammatory diseases “Dawn phenomenon” Møller N and Jørgensen JOL. Endocrine Rev 2009;30(2):152-177. GH, growth hormone; IGF-I, insulin-like growth factor

Effects of Growth Hormone Deficiency on Glucose Homeostasis In children and adults with GHD, loss of counterregulatory effects of GH results in dysregulated glucose homeostasis1 GHD Children GHD Adults Hyperresponsiveness to insulin1 Proneness to hypoglycemia1 Delayed recovery from hypoglycemia following IV insulin1 Reduced elevation in glucose and insulin following exposure to oral glucose and IV arginine, respectively1 Increased rates of abnormal or impaired glucose tolerance2 Loss of insulin sensitivity despite normal or elevated levels of insulin3 Decreased insulin-stimulated glycogen synthase activity in skeletal muscle4 1Møller N and Jørgensen JOL. Endocrine Rev 2009;30(2):152-177; 2Beshyah SA, et al. Clin Sci (Lond) 1995; 89:321-28; 3Johansson JO, et al. Metabolism 1995;44:1126-29; 4Hew FL, et al. J Clin Endocrinol Metab 1996;81:555-564.

General Effects of Exogenous Growth Hormone Change from an insulin sensitive state to a more insulin resistant state Elevated fasting glucose and insulin (INS > GLU) Elevated glucose and insulin responses to meals (INS > GLU) Increased IGF-I and IGFBPs Changes usually modest and within the normal range Effects may be transient Individuals with glucose intolerance may become overtly diabetic if given rhGH modulation of insulin sensitivity by rhGH complex and influenced by multiple factors IGF, insulin-like growth factor; IGFBP, IGF binding protein; rhGH, recombinant human growth hormone

Consensus from 2016 Growth Hormone Safety Workshop: Metabolic Risk Pediatric GHD Very low incidence of developing glucose intolerance or overt type 2 diabetes mellitus (DM) during rhGH treatment Lifetime risk of glucose intolerance and type 2 DM higher than background population in a number of conditions treated with rhGH, including Turner syndrome and in short children born SGA GH treatment does not increase the incidence of type 2 DM in these conditions in the short term Adult GHD rhGH therapy may be associated with the development of glucose intolerance or type 2 DM in the first year of therapy in high risk adult GHD patients (obesity, family history of type 2 DM) Monitoring with HbA1c is important Allen DB, et al. Euro J Endocrinol 2016 Feb;174(2):P1-9 GHD, growth hormone deficiency; HBA1c, hemoglobin A1c; rhGH, recombinant human growth hormone; SGA, small for gestational age

Effects of rhGH on Insulin Sensitivity in GHD Treatment of GHD with daily rhGH reduces insulin sensitivity, with effects correlating positively to dose and inversely with duration of treatment1 While rhGH-treated GHD adults predisposed to DM are prone to impaired glucose tolerance or overt DM, children on rhGH rarely develop either, but may develop modest insulin resistance2 Long-acting forms of rhGH are being developed to improve treatment outcomes by reducing treatment burden and improving adherence issues; effects of these agents on glucose homeostasis are not well understood3 To determine whether somavaratan, a long-acting form of rhGH, has distinct metabolic effects by evaluating its effects on HbA1c in pre-pubertal GHD children OBJECTIVE 1Møller N and Jørgensen JOL. Endocrine Rev 2009;30(2):152-177; 2Allen DB, et al. Euro J Endocrinol 2016 Feb;174(2):P1-9; 3Saenger PH and Mejia-Corletto J. Endocr Dev 2016;30:79-97. DM, diabetes mellitus; GHD, growth hormone deficiency; rhGH, recombinant human growth hormone

Methods 64 subjects received somavaratan for 6 months in the Phase 1b/2a study 60 elected to remain on somavaratan in the open-label, long-term safety study (13VR3, VISTA) All patients transitioned to 3.5 mg/kg twice-monthly by the 6-month time point in VISTA (after 12 months total somavaratan exposure) HbA1c was measured at VISTA study entry and yearly thereafter, correlating to months 6, 18, and 30 of somavaratan exposure Baseline Characteristics are Consistent with a Pediatric Population with Moderate GHD
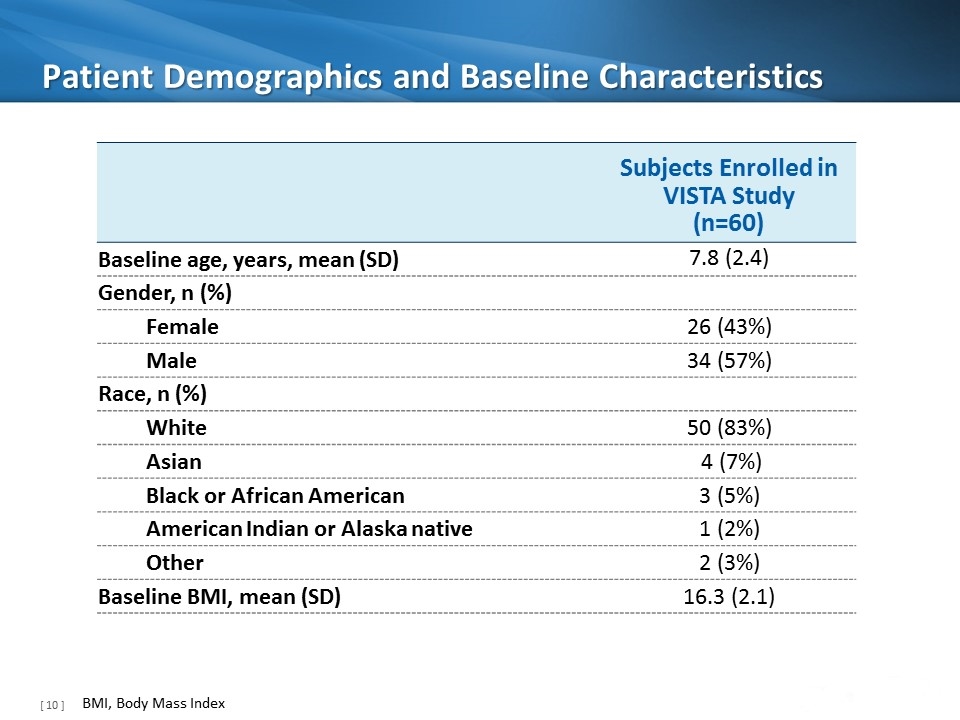
Patient Demographics and Baseline Characteristics Subjects Enrolled in VISTA Study (n=60) Baseline age, years, mean (SD) 7.8 (2.4) Gender, n (%) Female 26 (43%) Male 34 (57%) Race, n (%) White 50 (83%) Asian 4 (7%) Black or African American 3 (5%) American Indian or Alaska native 1 (2%) Other 2 (3%) Baseline BMI, mean (SD) 16.3 (2.1) BMI, Body Mass Index
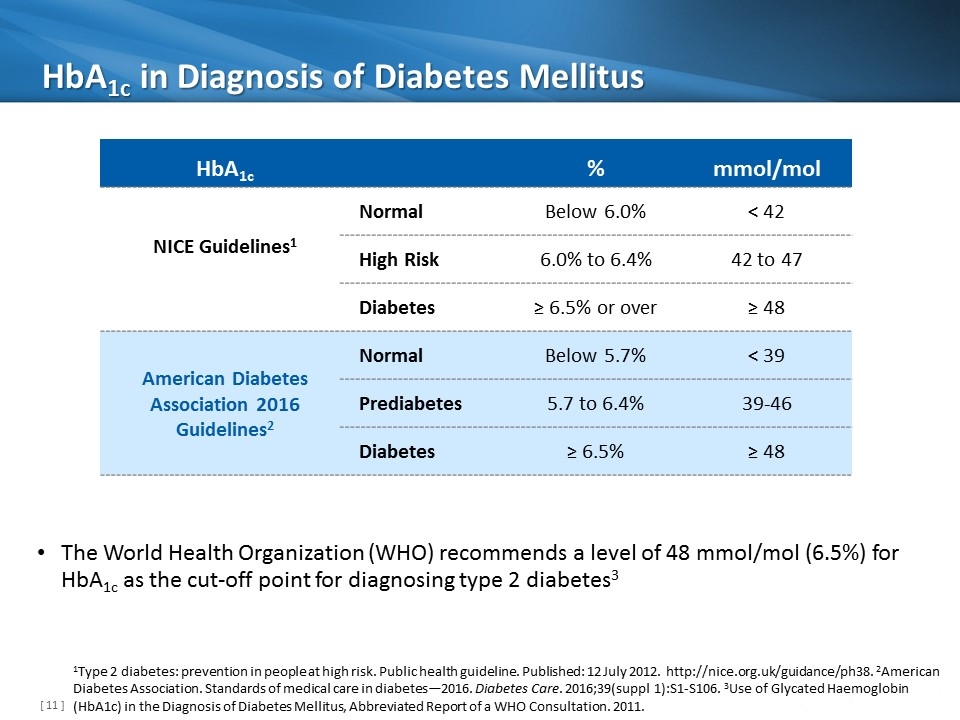
HbA1c in Diagnosis of Diabetes Mellitus HbA1c % mmol/mol NICE Guidelines1 Normal Below 6.0% < 42 High Risk 6.0% to 6.4% 42 to 47 Diabetes ≥ 6.5% or over ≥ 48 American Diabetes Association 2016 Guidelines2 Normal Below 5.7% < 39 Prediabetes 5.7 to 6.4% 39-46 Diabetes ≥ 6.5% ≥ 48 1Type 2 diabetes: prevention in people at high risk. Public health guideline. Published: 12 July 2012. http://nice.org.uk/guidance/ph38. 2American Diabetes Association. Standards of medical care in diabetes—2016. Diabetes Care. 2016;39(suppl 1):S1-S106. 3Use of Glycated Haemoglobin (HbA1c) in the Diagnosis of Diabetes Mellitus, Abbreviated Report of a WHO Consultation. 2011. The World Health Organization (WHO) recommends a level of 48 mmol/mol (6.5%) for HbA1c as the cut-off point for diagnosing type 2 diabetes3
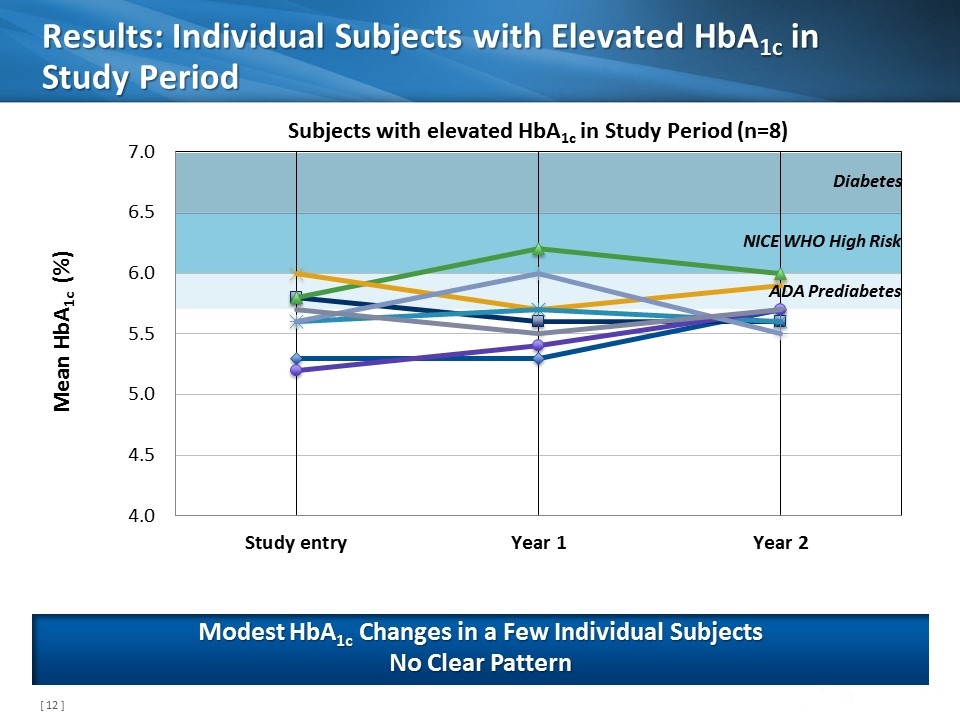
Results: Individual Subjects with Elevated HbA1c in Study Period Modest HbA1c Changes in a Few Individual Subjects No Clear Pattern NICE WHO High Risk Diabetes ADA Prediabetes Subjects with elevated HbA1c in Study Period (n=8)
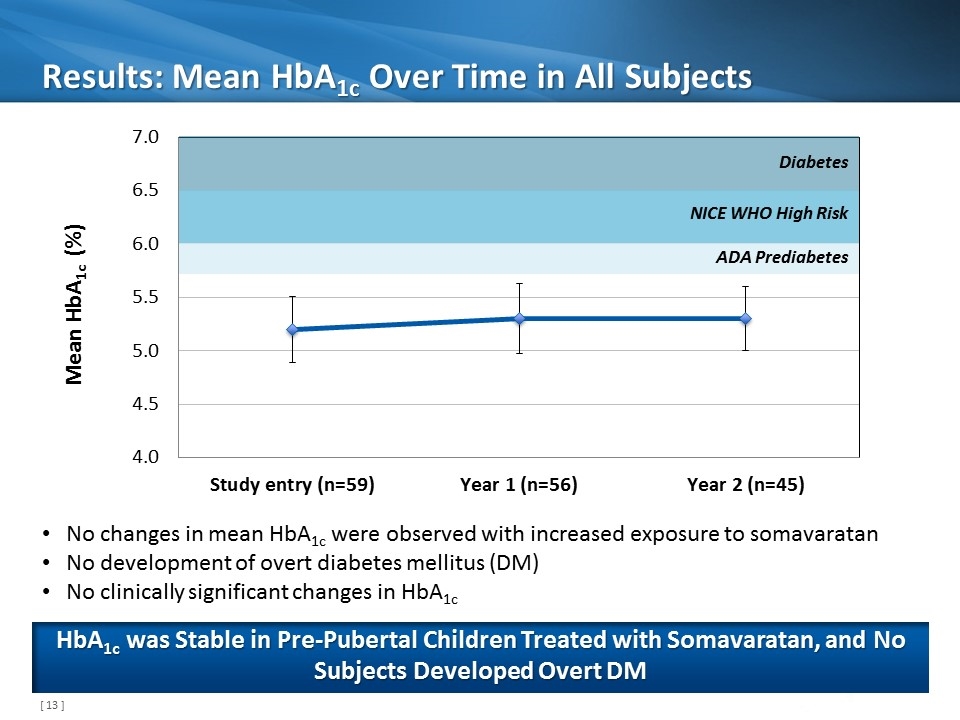
Results: Mean HbA1c Over Time in All Subjects HbA1c was Stable in Pre-Pubertal Children Treated with Somavaratan, and No Subjects Developed Overt DM NICE WHO High Risk Diabetes ADA Prediabetes No changes in mean HbA1c were observed with increased exposure to somavaratan No development of overt diabetes mellitus (DM) No clinically significant changes in HbA1c

Conclusions HbA1c was stable in pre-pubertal GHD children treated with somavaratan Individual subjects (n=8) showed minor changes in HbA1c over time, and no subjects developed diabetes mellitus These findings suggest that somavaratan does not elicit detrimental effects on insulin sensitivity in pre-pubertal GHD children in the current population treated Further characterization of the metabolic effects of somavaratan are warranted and are underway A Phase 3 study of 3.5 mg/kg Twice-Monthly Somavaratan is Ongoing (NCT02339090)

Acknowledgments Investigators & Study Coordinators Patients and their Caregivers ResearchPoint Global