
Eric Humphriss1, F. Naureen Sheikh1, Morgan Seaman2, David Ng2, George Bright1 1Versartis, Inc., Menlo Park, CA; 2ResearchPoint Global, Inc., Austin, TX Treatment Adherence with Weekly, Twice-Monthly and Monthly Dosing of Somavaratan (VRS-317), a Long-Acting Growth Hormone Treatment for Children with Growth Hormone Deficiency (GHD), After 24 Months of At-Home Dosing in the VISTA Study Exhibit 99.3

Disclosures Eric Humphriss, MBA, and F. Naureen Sheikh, PhD, are employees of Versartis, Inc David Ng, PhD, and Morgan Seaman are employees of ResearchPoint Global, a CRO receiving funding from Versartis, Inc George Bright, MD, is a consultant to Versartis and holds equity interests in Versartis, Inc Somavaratan (VRS-317) is an investigational agent

Challenges With Adherence to Daily Growth Hormone Treatment Children with growth hormone deficiency (GHD) are often treated for multiple years with daily injections of recombinant human growth hormone (rhGH) replacement therapy, currently the only treatment available in North America and Europe Treatment adherence to daily subcutaneous rhGH is a known burden for these patients, with poor adherence negatively impacting treatment outcome1 Noncompliance to daily injections has been reported in up to 66%–77% of adults and children with GHD,2 and is significantly associated with reduced annual height velocity3 1. De Pedro et al, Gr Horm IGF Res. 2016; 26:32-35; 2. Rosenfeld and Bakker. Endocr Pract. 2008;14:143-54; 3. Cutfield et al, PLOS One. 2011;6:e16223.
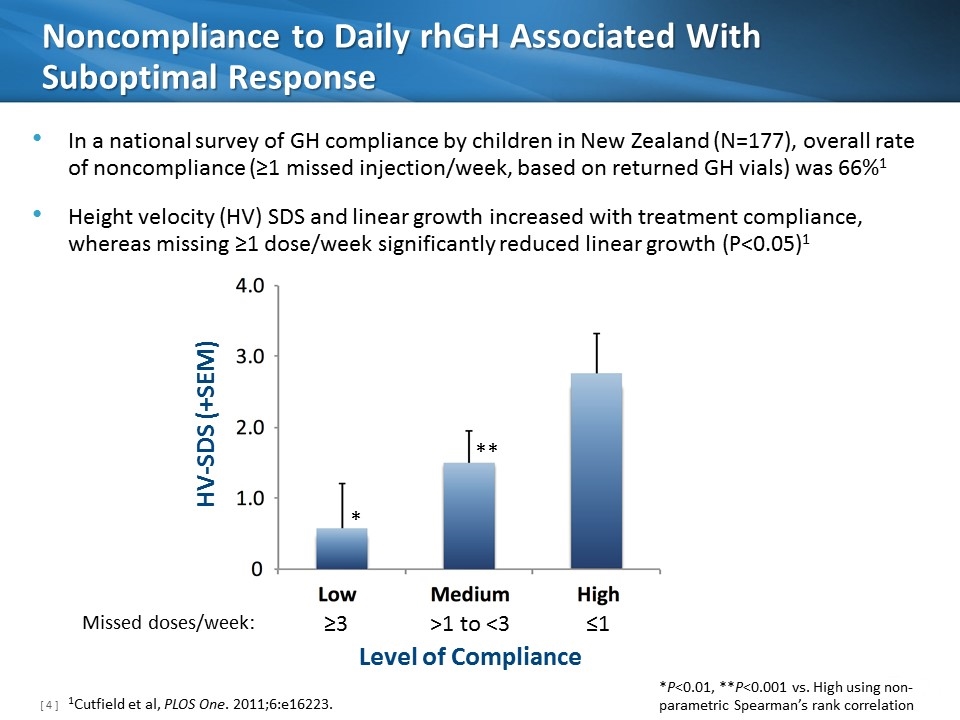
Noncompliance to Daily rhGH Associated With Suboptimal Response HV-SDS (+SEM) Level of Compliance ≥3 ≤1 >1 to <3 Missed doses/week: In a national survey of GH compliance by children in New Zealand (N=177), overall rate of noncompliance (≥1 missed injection/week, based on returned GH vials) was 66%1 Height velocity (HV) SDS and linear growth increased with treatment compliance, whereas missing ≥1 dose/week significantly reduced linear growth (P<0.05)1 *P<0.01, **P<0.001 vs. High using non-parametric Spearman’s rank correlation * ** 1Cutfield et al, PLOS One. 2011;6:e16223.

Poor Adherence to rhGH Therapy Results in Suboptimal Growth Height Velocity SDS & IGF-I Levels Impacted by Lack of Adherence one of the primary causes of GH therapy failure is patient non-adherence to the prescribed drug therapy Longitudinal study with 158 children between 4-16 years with GHD or SGA 33.5% Moderate-to-poor adherence Defined as taking <92% of prescribed rhGH medication Decreased adherence significantly associated with Treatment duration (p=0.001) Mother’s educational level (p=0.007) Decreased HV (p=0.002) IGF-I levels (p<0.0001) De Pedro, S. et al. Growth Hormone & IGF Research 26 (2016) 32-35 Good Adherence Moderate-Poor Adherence P Age (years) 10.5 ± 2.7 11.5 ± 2.8 0.041 Sex Male 62 (61.4) 39 (38.6) 0.072 Female 43 (75.4) 14 (24.6) Pubertal stage N (%) Pre-pubertal 56 (73.7) 20 (26.3) 0.064 Pubertal 49 (59) 33 (40.2) Duration of treatment (yrs) 2.78 ± 1.71 3.78 ±1.80 0.001 Daily Dose (mg/day) 0.98 ± 0.29 1.08 ± 0.32 0.038 IGF-1 SDS 1.3 ± 1.03 0.48 ± 1.09 <0.0001 HV-SDS 1.57 ± 1.90 0.61 ± 1.50 0.002

Clinical Need for Long-Acting Growth Hormone “Long-acting growth hormone compounds may represent an advance over daily GH injections because of increased convenience and differing pharmacodynamic properties, providing the potential for improved adherence and outcomes.” Growth Hormone Research Society Consensus Statement1 There is a clinical need for a safe and effective long-acting growth hormone treatment for which improved treatment adherence is attainable Development of a long-acting form of rhGH with long-term effectiveness may potentially reduce treatment burden, improve adherence issues, and improve overall treatment outcomes 1. Christiansen et al. Eur J Endocrinol. 174;C1-C8
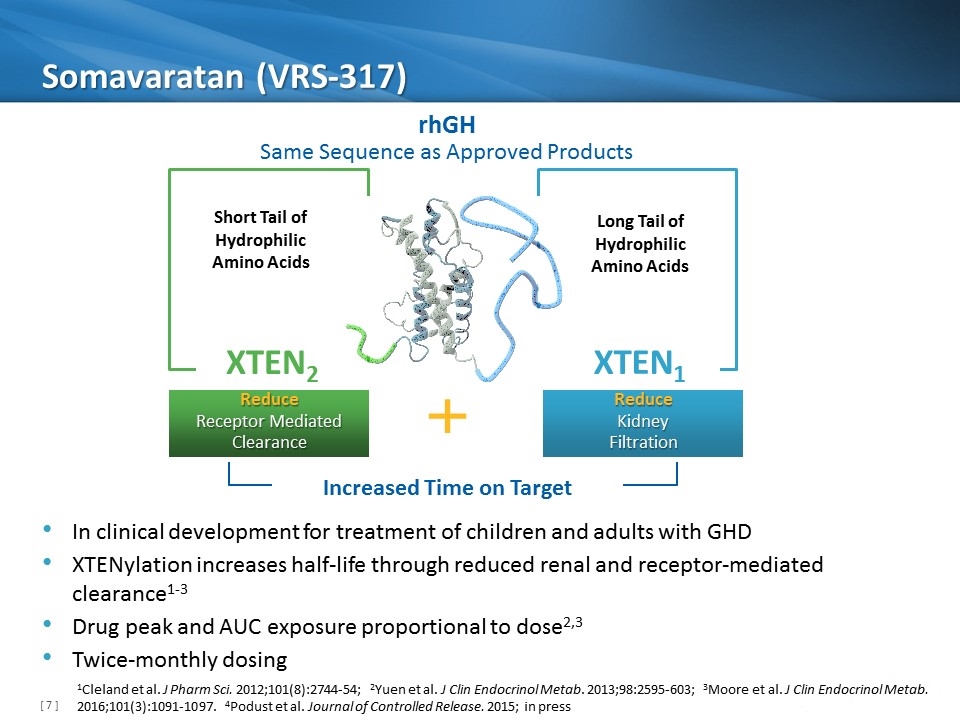
Somavaratan (VRS-317) Increased Time on Target XTEN1 Long Tail of Hydrophilic Amino Acids Reduce Kidney Filtration XTEN2 Short Tail of Hydrophilic Amino Acids Reduce Receptor Mediated Clearance rhGH Same Sequence as Approved Products In clinical development for treatment of children and adults with GHD XTENylation increases half-life through reduced renal and receptor-mediated clearance1-3 Drug peak and AUC exposure proportional to dose2,3 Twice-monthly dosing 1Cleland et al. J Pharm Sci. 2012;101(8):2744-54; 2Yuen et al. J Clin Endocrinol Metab. 2013;98:2595-603; 3Moore et al. J Clin Endocrinol Metab. 2016;101(3):1091-1097. 4Podust et al. Journal of Controlled Release. 2015; in press

Somavaratan: Clinical Experience Phase 1 - Adults1 30- to 60-fold increase in elimination half-life and longer IGF-I responses, compared with daily rhGH (N=50) Dose-proportional increases in magnitude and duration of IGF-I responses for up to 1 month after a single dose No study drug related serious adverse events Phase 1b/2a - Pre-Pubertal Children2 Weekly, twice-monthly, and monthly dosing enabled by dose-proportional increases in magnitude and duration of IGF-I responses Clinically meaningful improvements in height velocity (HV) and IGF-I with all 3 dosing schedules (Phase 2a; N=64) No study drug related serious adverse events 1Yuen et al. J Clin Endocrinol Metab. 2013;98:2595-603; 2Moore et al. J Clin Endocrinol Metab. 2016;101(3):1091-1097. The Open-Label, Long-Term Safety Study (VISTA Study, 13VR3) continues with subjects approaching 3 years somavaratan exposure

Methods: Monitoring Treatment Adherence Dosing events were reported by the caregiver using a smartphone-compatible electronic patient-reported outcome diary (eDiary) Prior to at-home dosing (initiated at beginning of extension study), caregivers were trained on: Preparation of somavaratan Subcutaneous injection technique eDiary use In-clinic visits were conducted quarterly for patient follow-up, eDiary reprogramming, and re-supply of somavaratan and ancillary supplies The eDiary was programmed to provide both assigned injection volume and timing of injection. Caregivers used the eDiary to report injection volume administered and date of administration Notifications sent to research nurse for late or missed injections
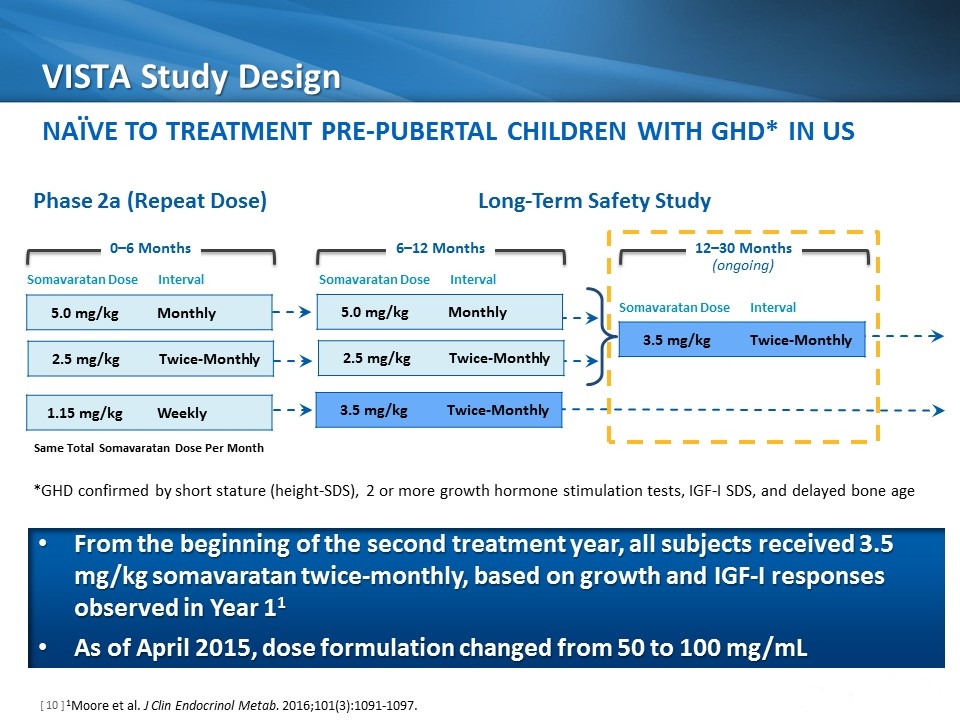
VISTA Study Design 1Moore et al. J Clin Endocrinol Metab. 2016;101(3):1091-1097. 5.0 mg/kg Monthly 3.5 mg/kg Twice-Monthly 6–12 Months 12–30 Months (ongoing) 0–6 Months Somavaratan Dose Interval Phase 2a (Repeat Dose) Long-Term Safety Study 2.5 mg/kg Twice-Monthly 1.15 mg/kg Weekly 5.0 mg/kg Monthly 2.5 mg/kg Twice-Monthly 3.5 mg/kg Twice-Monthly Somavaratan Dose Interval Somavaratan Dose Interval Naïve to Treatment Pre-Pubertal Children with GHD* IN US Same Total Somavaratan Dose Per Month From the beginning of the second treatment year, all subjects received 3.5 mg/kg somavaratan twice-monthly, based on growth and IGF-I responses observed in Year 11 As of April 2015, dose formulation changed from 50 to 100 mg/mL *GHD confirmed by short stature (height-SDS), 2 or more growth hormone stimulation tests, IGF-I SDS, and delayed bone age
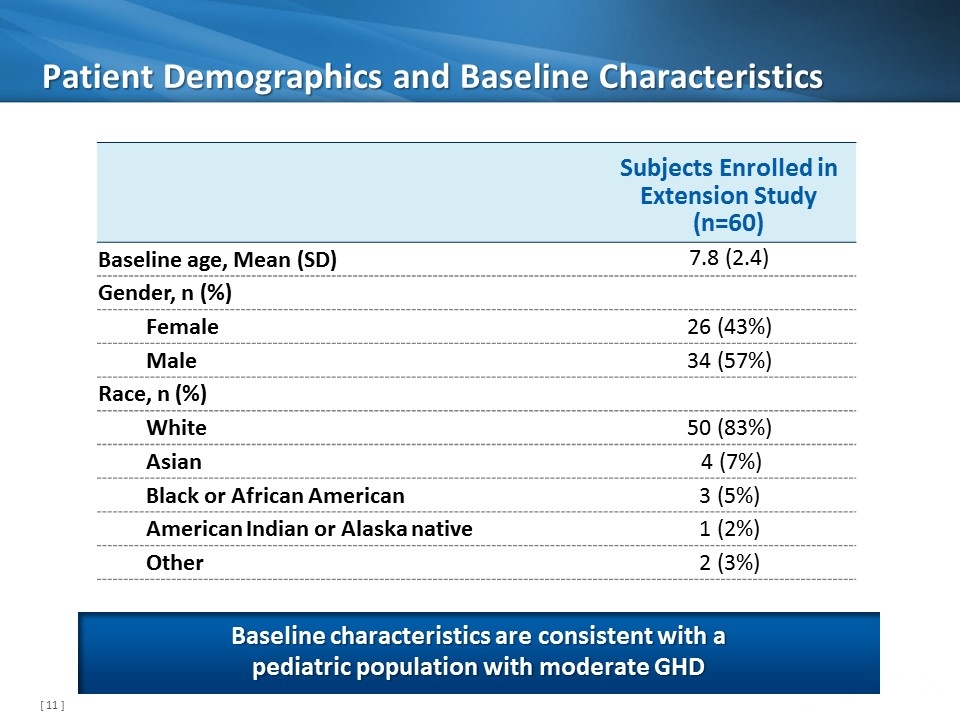
Patient Demographics and Baseline Characteristics Subjects Enrolled in Extension Study (n=60) Baseline age, Mean (SD) 7.8 (2.4) Gender, n (%) Female 26 (43%) Male 34 (57%) Race, n (%) White 50 (83%) Asian 4 (7%) Black or African American 3 (5%) American Indian or Alaska native 1 (2%) Other 2 (3%) Baseline characteristics are consistent with a pediatric population with moderate GHD
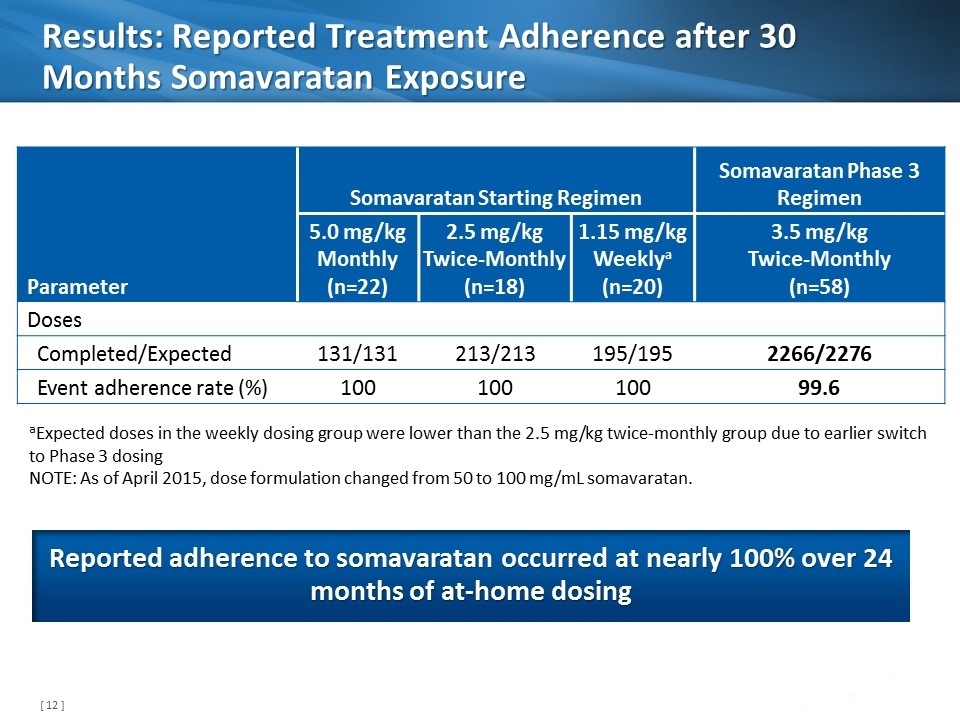
Results: Reported Treatment Adherence after 30 Months Somavaratan Exposure Parameter Somavaratan Starting Regimen Somavaratan Phase 3 Regimen 5.0 mg/kg Monthly (n=22) 2.5 mg/kg Twice-Monthly (n=18) 1.15 mg/kg Weeklya (n=20) 3.5 mg/kg Twice-Monthly (n=58) Doses Completed/Expected 131/131 213/213 195/195 2266/2276 Event adherence rate (%) 100 100 100 99.6 aExpected doses in the weekly dosing group were lower than the 2.5 mg/kg twice-monthly group due to earlier switch to Phase 3 dosing NOTE: As of April 2015, dose formulation changed from 50 to 100 mg/mL somavaratan. Reported adherence to somavaratan occurred at nearly 100% over 24 months of at-home dosing

Conclusions Use of an eDiary device/application simplifies parent tracking of timing for and recording of at-home dosing events in this clinical trial With at-home dosing and over 2200 doses administered at the Phase 3 dose, treatment adherence rate was 99.6% With nearly 100% reported adherence for all dosing schedules over 24 months of at-home therapy, this study provides evidence that long-term adherence to treatment may be improved with long-acting growth hormone preparations in children with GHD A Phase 3 study of somavaratan using the eDiary to monitor treatment adherence is ongoing (NCT02339090)

Acknowledgments Investigators & Study Coordinators Patients and their families ResearchPoint Global Bracket, Inc. (eDiary)