
George Bright1, Wayne V. Moore2, Huong Jil Nguyen3, Gad B. Kletter4, Bradley S. Miller5, Patricia Y. Fechner6, David Ng7, Eric Humphriss1, Jeffrey L. Cleland1 1Versartis, Inc, Menlo Park, CA; 2Children's Mercy Hospital and University of Missouri-Kansas City, Kansas City, MO; 3Sierra Medical Research, Clovis, CA; 4Mary Bridge Children's Hospital, University of Washington, Tacoma, WA; 5University of Minnesota Masonic Children's Hospital, Minneapolis, MN; 6Seattle Children's Hospital, University of Washington, Seattle, WA; 7ResearchPoint Global, Inc., Austin, TX Somavaratan (VRS-317) Treatment for Pediatric Growth Hormone Deficiency (GHD): VISTA Study Results at 2.5 Years (NCT02068521) Exhibit 99.2

Disclosures George Bright, MD, and Jeffrey Cleland, PhD, are consultants and hold equity interests in Versartis, Inc Huong Jil Nguyen, MD, has received research funding from Versartis, Inc Bradley S. Miller, MD, PhD, has received grants from Alexion Pharmaceuticals, Inc., Endo Pharmaceuticals, Genentech, Inc., Novo Nordisk A/S, Orphan Reach/Tolmar, Inc., Sandoz International GmbH, Shire Human Genetic Therapies, Inc. and Versartis, Inc. He has also received honoraria from Ferring Pharmaceuticals, Inc., Genentech, Inc., Novo Nordisk A/S, Sandoz International GmbH, and Versartis, Inc Patricia Y. Fechner, MD, has received research support from Eli Lilly & Company, Novo Nordisk A/S, and Versartis, Inc David Ng, PhD, is an employee of ResearchPoint Global, a CRO receiving funding from Versartis, Inc Eric Humphriss, MBA, is an employee of Versartis, Inc Somavaratan (VRS-317) is an investigational agent

Recombinant Human Growth Hormone (rhGH) for Treatment of Pediatric Growth Hormone Deficiency (GHD) Therapeutic potential of daily rhGH is well established and has been the primary treatment for pediatric GHD for three decades1,2 Current challenges with daily rhGH preparations include burden of daily subcutaneous (SC) injections3 Noncompliance has been reported in up to 77% of adults and children with GHD4,5 Reduced efficacy (decreasing HV-SDS) is significantly associated with number of missed doses per week3,5,6 HV-SDS, height velocity-standard deviation score 1Ergun-Longmire and Wajnrajch. Endotext. http://www.ncbi.nlm.nih.gov/books/NBK279142; 2Ranke et al. J Clin Endocrinol Metabol. 2005;90:1966; 3Saenger and Mejia-Corletto. Adv Ther Ped Endocrinol Diabetes. 2016;30:79-97; 4Rosenfeld and Bakker. Endocr Pract. 2008;14(2)143-54; 5Cutfield et al. PloS One. 2011;6(1):e16223. 6De Pedro et al. Growth Hormone & IGF Research.2016; 26:32-35 Introduction of a safe and effective long-acting form of rhGH that reduces injection frequency, while maintaining long-term growth response, may improve clinical outcomes while reducing the burden of daily SC injections of rhGH
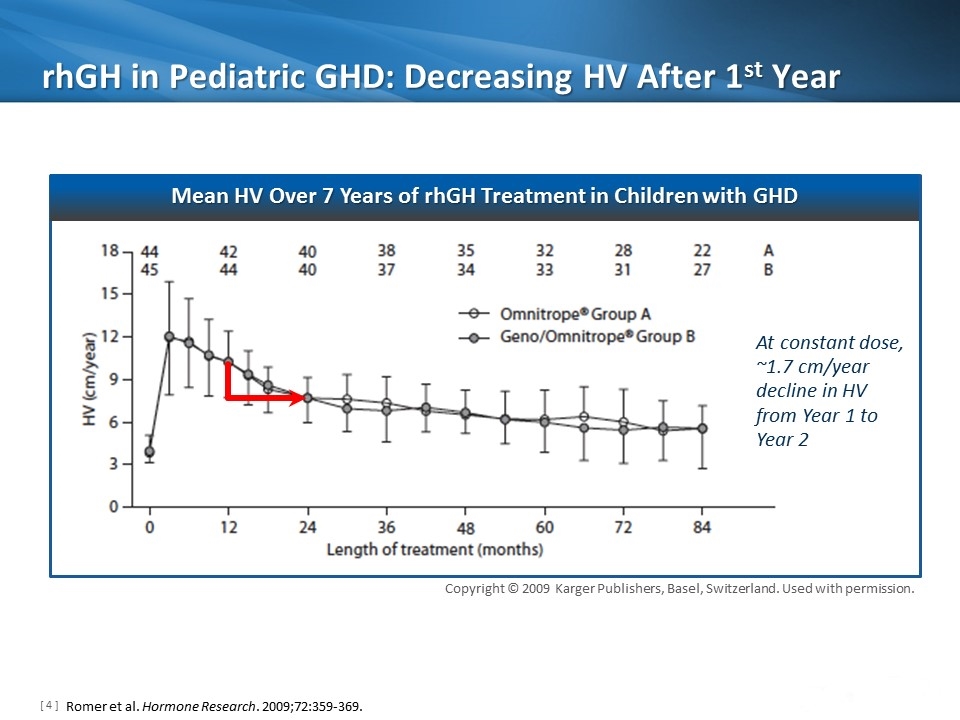
rhGH in Pediatric GHD: Decreasing HV After 1st Year Romer et al. Hormone Research. 2009;72:359-369. Copyright © 2009 Karger Publishers, Basel, Switzerland. Used with permission. Mean HV Over 7 Years of rhGH Treatment in Children with GHD At constant dose, ~1.7 cm/year decline in HV from Year 1 to Year 2
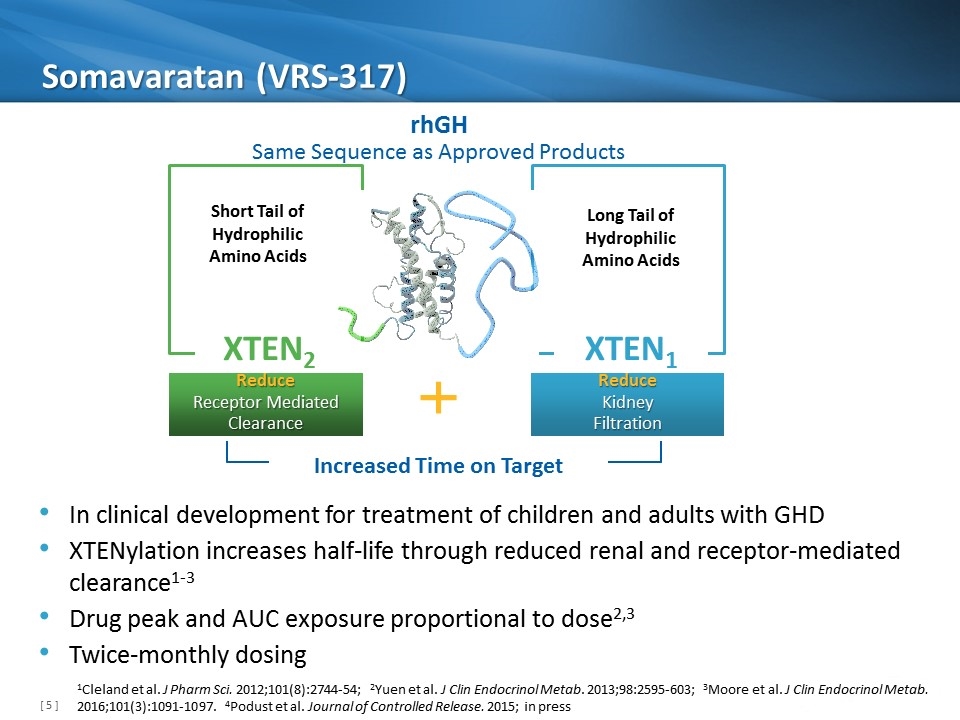
Somavaratan (VRS-317) Increased Time on Target XTEN1 Long Tail of Hydrophilic Amino Acids Reduce Kidney Filtration XTEN2 Short Tail of Hydrophilic Amino Acids Reduce Receptor Mediated Clearance rhGH Same Sequence as Approved Products In clinical development for treatment of children and adults with GHD XTENylation increases half-life through reduced renal and receptor-mediated clearance1-3 Drug peak and AUC exposure proportional to dose2,3 Twice-monthly dosing 1Cleland et al. J Pharm Sci. 2012;101(8):2744-54; 2Yuen et al. J Clin Endocrinol Metab. 2013;98:2595-603; 3Moore et al. J Clin Endocrinol Metab. 2016;101(3):1091-1097. 4Podust et al. Journal of Controlled Release. 2015; in press

Somavaratan: Clinical Experience Phase 1 - Adults1 30- to 60-fold increase in elimination half-life and longer IGF-I responses, compared with daily rhGH (N=50) Dose-proportional increases in magnitude and duration of IGF-I responses for up to 1 month after a single dose No study drug related serious adverse events Phase 1b/2a - Pre-Pubertal Children2 Weekly, twice-monthly, and monthly dosing enabled by dose-proportional increases in magnitude and duration of IGF-I responses Clinically meaningful improvements in height velocity (HV) and IGF-I with all 3 dosing schedules (Phase 2a; N=64) No study drug related serious adverse events 1Yuen et al. J Clin Endocrinol Metab. 2013;98:2595-603; 2Moore et al. J Clin Endocrinol Metab. 2016;101(3):1091-1097. The Open-Label, Long-Term Safety Study (VISTA Study, 13VR3) continues with subjects approaching 3 years of somavaratan exposure
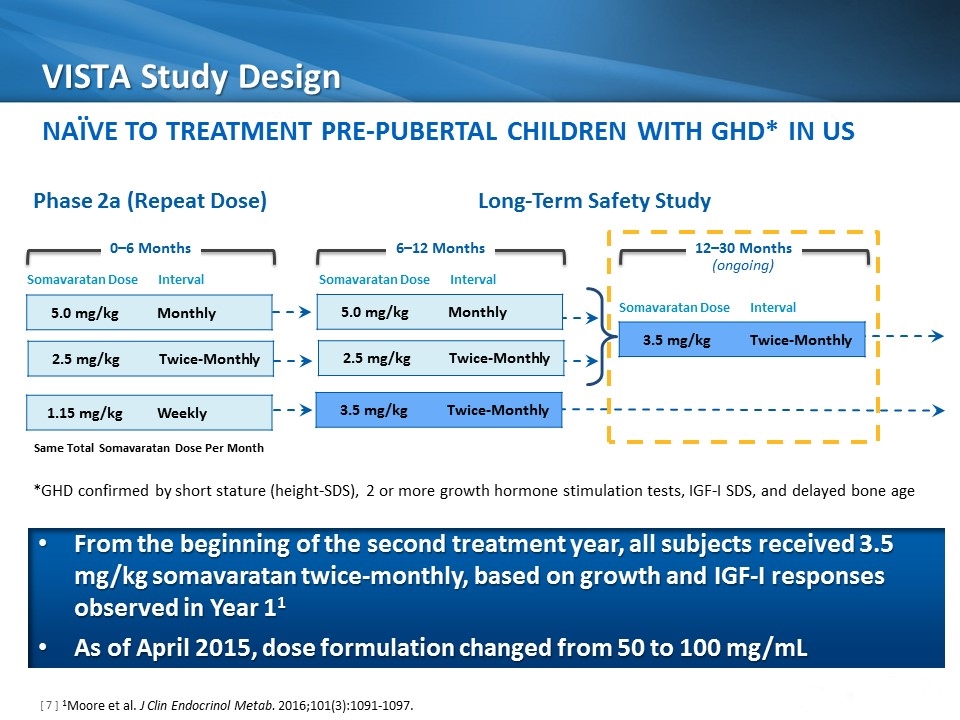
VISTA Study Design 1Moore et al. J Clin Endocrinol Metab. 2016;101(3):1091-1097. 5.0 mg/kg Monthly 3.5 mg/kg Twice-Monthly 6–12 Months 12–30 Months (ongoing) 0–6 Months Somavaratan Dose Interval Phase 2a (Repeat Dose) Long-Term Safety Study 2.5 mg/kg Twice-Monthly 1.15 mg/kg Weekly 5.0 mg/kg Monthly 2.5 mg/kg Twice-Monthly 3.5 mg/kg Twice-Monthly Somavaratan Dose Interval Somavaratan Dose Interval Naïve to Treatment Pre-Pubertal Children with GHD* IN US Same Total Somavaratan Dose Per Month From the beginning of the second treatment year, all subjects received 3.5 mg/kg somavaratan twice-monthly, based on growth and IGF-I responses observed in Year 11 As of April 2015, dose formulation changed from 50 to 100 mg/mL *GHD confirmed by short stature (height-SDS), 2 or more growth hormone stimulation tests, IGF-I SDS, and delayed bone age
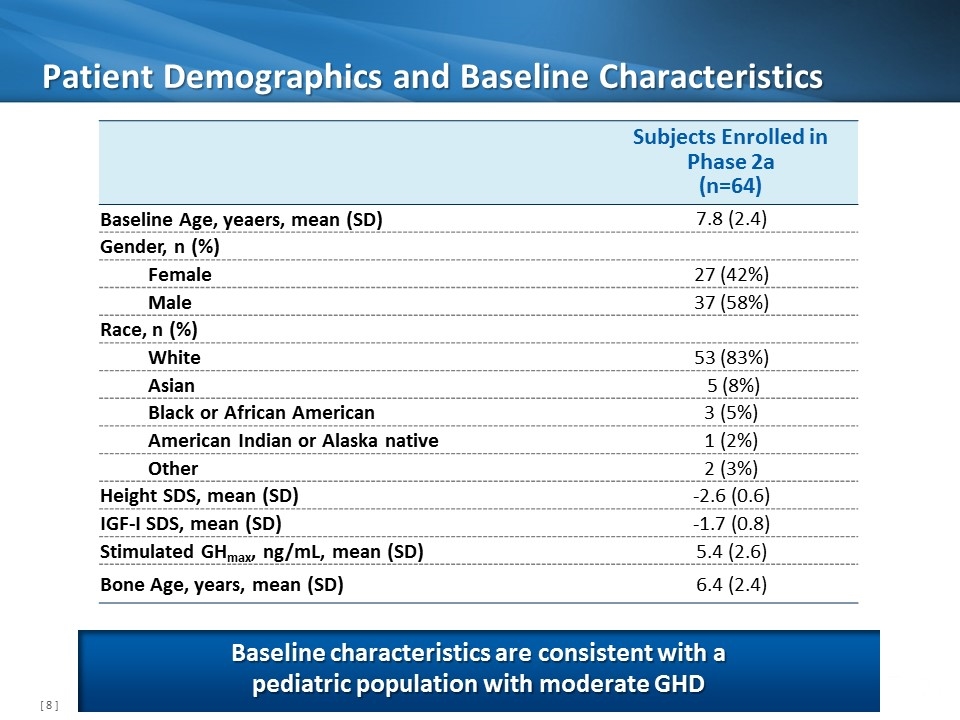
Patient Demographics and Baseline Characteristics Subjects Enrolled in Phase 2a (n=64) Baseline Age, yeaers, mean (SD) 7.8 (2.4) Gender, n (%) Female 27 (42%) Male 37 (58%) Race, n (%) White 53 (83%) Asian 5 (8%) Black or African American 3 (5%) American Indian or Alaska native 1 (2%) Other 2 (3%) Height SDS, mean (SD) -2.6 (0.6) IGF-I SDS, mean (SD) -1.7 (0.8) Stimulated GHmax, ng/mL, mean (SD) 5.4 (2.6) Bone Age, years, mean (SD) 6.4 (2.4) Baseline characteristics are consistent with a pediatric population with moderate GHD
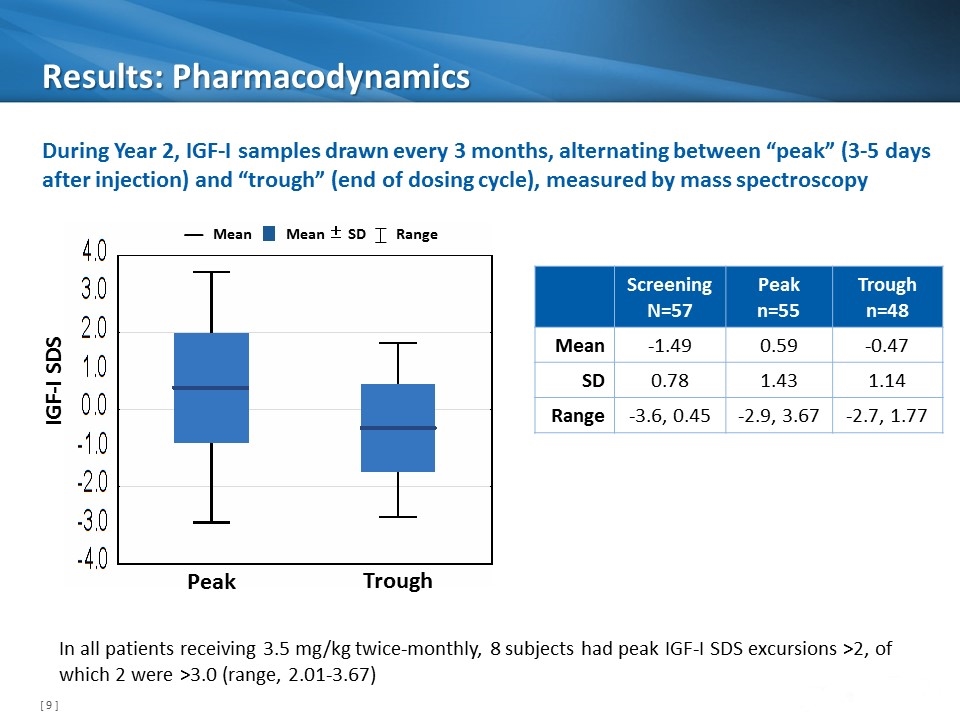
Results: Pharmacodynamics Mean Mean ± SD Range During Year 2, IGF-I samples drawn every 3 months, alternating between “peak” (3-5 days after injection) and “trough” (end of dosing cycle), measured by mass spectroscopy In all patients receiving 3.5 mg/kg twice-monthly, 8 subjects had peak IGF-I SDS excursions >2, of which 2 were >3.0 (range, 2.01-3.67) Screening N=57 Peak n=55 Trough n=48 Mean -1.49 0.59 -0.47 SD 0.78 1.43 1.14 Range -3.6, 0.45 -2.9, 3.67 -2.7, 1.77 Peak Trough IGF-I SDS
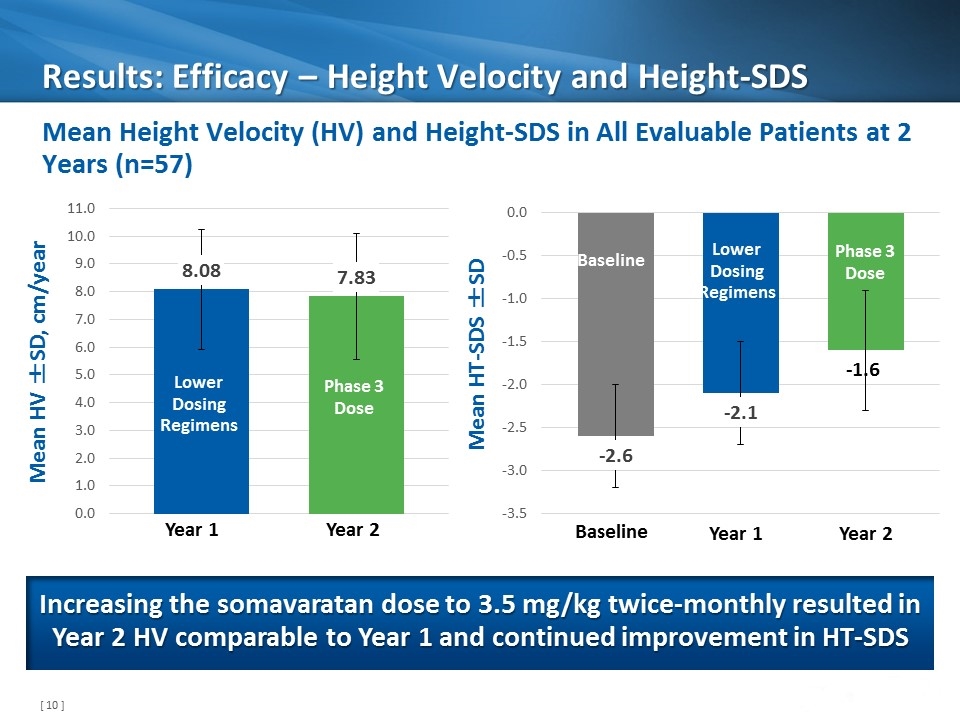
Results: Efficacy – Height Velocity and Height-SDS Mean Height Velocity (HV) and Height-SDS in All Evaluable Patients at 2 Years (n=57) Mean HV ±SD, cm/year Increasing the somavaratan dose to 3.5 mg/kg twice-monthly resulted in Year 2 HV comparable to Year 1 and continued improvement in HT-SDS Lower Dosing Regimens Phase 3 Dose Year 1 Year 2 Mean HT-SDS ±SD Lower Dosing Regimens Phase 3 Dose Year 1 Year 2 Baseline Baseline
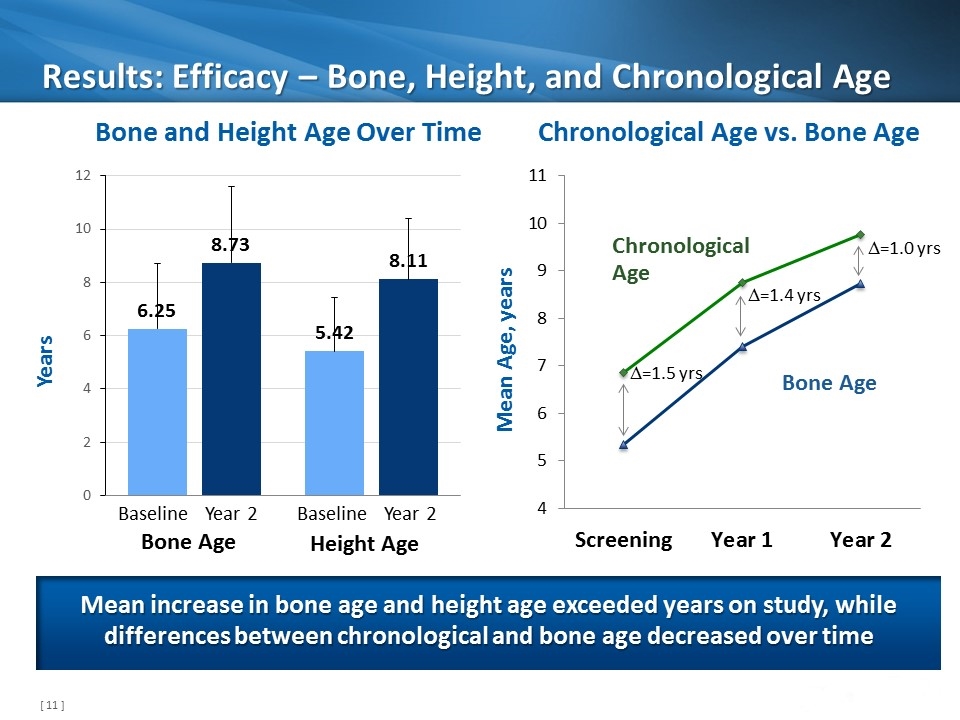
Results: Efficacy – Bone, Height, and Chronological Age Mean increase in bone age and height age exceeded years on study, while differences between chronological and bone age decreased over time Years Bone and Height Age Over Time Chronological Age vs. Bone Age Bone Age Height Age Baseline Year 2 Baseline Year 2 Bone Age Chronological Age Mean Age, years D=1.5 yrs D=1.4 yrs D=1.0 yrs
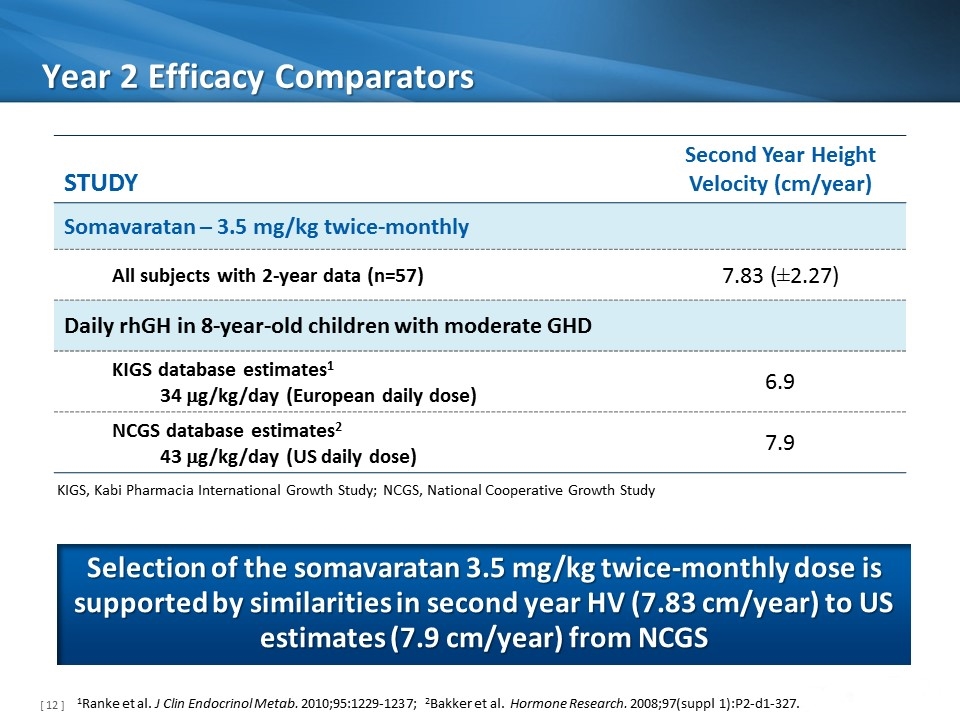
Year 2 Efficacy Comparators Selection of the somavaratan 3.5 mg/kg twice-monthly dose is supported by similarities in second year HV (7.83 cm/year) to US estimates (7.9 cm/year) from NCGS Study Second Year Height Velocity (cm/year) Somavaratan – 3.5 mg/kg twice-monthly All subjects with 2-year data (n=57) 7.83 (±2.27) Daily rhGH in 8-year-old children with moderate GHD KIGS database estimates1 34 mg/kg/day (European daily dose) 6.9 NCGS database estimates2 43 mg/kg/day (US daily dose) 7.9 1Ranke et al. J Clin Endocrinol Metab. 2010;95:1229-1237; 2Bakker et al. Hormone Research. 2008;97(suppl 1):P2-d1-327. KIGS, Kabi Pharmacia International Growth Study; NCGS, National Cooperative Growth Study
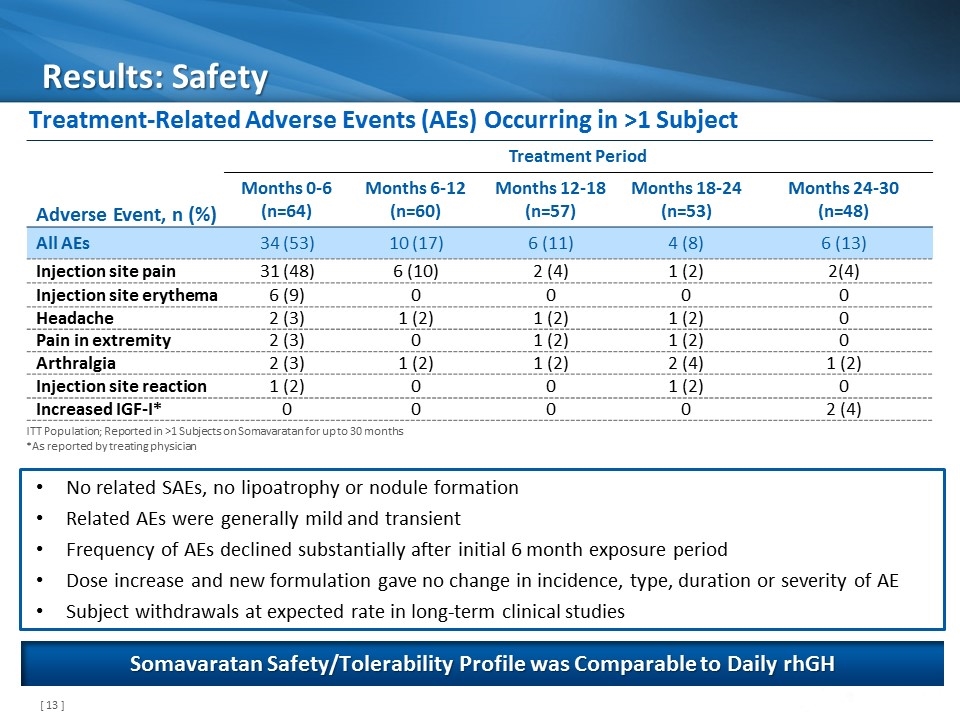
Results: Safety Treatment-Related Adverse Events (AEs) Occurring in >1 Subject Adverse Event, n (%) Treatment Period Months 0-6 (n=64) Months 6-12 (n=60) Months 12-18 (n=57) Months 18-24 (n=53) Months 24-30 (n=48) All AEs 34 (53) 10 (17) 6 (11) 4 (8) 6 (13) Injection site pain 31 (48) 6 (10) 2 (4) 1 (2) 2(4) Injection site erythema 6 (9) 0 0 0 0 Headache 2 (3) 1 (2) 1 (2) 1 (2) 0 Pain in extremity 2 (3) 0 1 (2) 1 (2) 0 Arthralgia 2 (3) 1 (2) 1 (2) 2 (4) 1 (2) Injection site reaction 1 (2) 0 0 1 (2) 0 Increased IGF-I* 0 0 0 0 2 (4) ITT Population; Reported in >1 Subjects on Somavaratan for up to 30 months *As reported by treating physician Somavaratan Safety/Tolerability Profile was Comparable to Daily rhGH No related SAEs, no lipoatrophy or nodule formation Related AEs were generally mild and transient Frequency of AEs declined substantially after initial 6 month exposure period Dose increase and new formulation gave no change in incidence, type, duration or severity of AE Subject withdrawals at expected rate in long-term clinical studies

Conclusions Phase 3 dose selection supported by VISTA study results for subjects switched to 3.5 mg/kg twice-monthly Mean peak IGF-I SDS at Phase 3 dose was in upper half of normal range Catch-up growth supported by mean increase in bone age and height age exceeding years on study, with gap between chronological and bone age closing over the course of the study Improvement in HT-SDS continued in Year 2 Year 2 HV comparable to US daily dosing data from NCGS Phase 3 dose (3.5 mg/kg, twice-monthly) was safe and well tolerated in this study Frequency and severity of treatment-related adverse events indicate no safety concerns Somavaratan, 3.5 mg/kg twice-monthly, is now under study in a randomized, Phase 3, non-inferiority trial versus daily rhGH in pre-pubertal children with GHD (NCT02339090) - The VELOCITY Trial

Acknowledgments Investigators & Study Coordinators Patients and their Caregivers Clinical Operations Leslie Jones, ResearchPoint Global Morgan Seaman, ResearchPoint Global Naureen Sheikh, PhD, Versartis, Inc. Lynda Chaplin, Versartis, Inc.